- Ticks and Tick-Borne Diseases Monographs
- Control of ticks and tick-borne diseases in Southern Africa
Control of ticks and tick-borne diseases in Southern Africa
This content is distributed under the following licence: Attribution CC BY  View Creative Commons Licence details here
View Creative Commons Licence details here

INTRODUCTION
The deleterious effects of ticks on domestic and wild animals in Southern Africa are well known and have been extensively reviewed in Monograph 1 of this series, and elsewhere. We briefly summarise here the effects of ticks on livestock – including small stock and game animals.
Heavy infestations of certain tick species such as the blue ticks Rhipicephalus (Boophilus) spp. which secrete salivary substances that have an anorectic effect on the host animal, result in significant weight loss or production. This effect is seen especially in exotic cattle breeds, which show better weight gain when tick infestation is controlled. Tampans or soft-bodied ticks which occur in the more arid western areas of the country can cause extreme irritation, blood loss, toxicoses and death in calves and small stock.
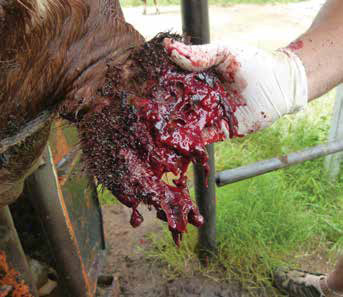
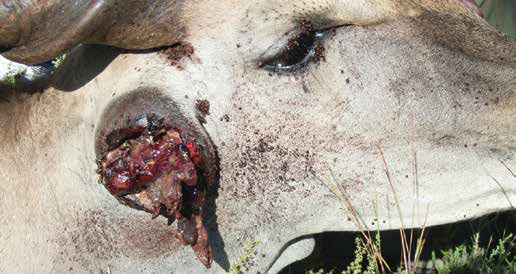
Tick species which have long mouthparts such as Amblyomma and Hyalomma cause considerable discomfort as well as tissue damage in cattle, leading to scarring of hides, and damage to testes and the udder which may lead to the loss of teats. These wounds may become secondarily infected with bacteria, resulting in the development of abscesses or fly strike. In small stock, the socalled foot ticks or those which are found around the claws of sheep and goats, either between or around the claws, cause wounds which can lead to secondary infection with resulting lameness. Tick damage due to the long mouthparts of Amblyomma ticks and irritation due to various Rhipicephalus spp. such as brown ear ticks, cause problems in susceptible species. Eland and gemsbok may lose ears due to necrosis with secondary myiasis. Heavy infestations on the eyelids can cause eye irritation, with subsequent infection. Abscesses may be seen in cattle and various antelope and other game species due to secondary infection of wounds caused by ticks.
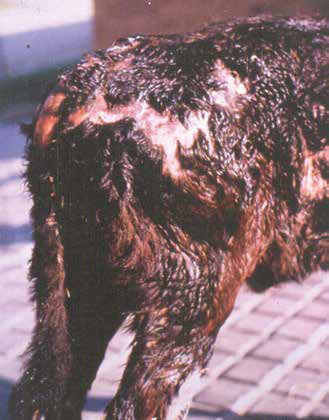
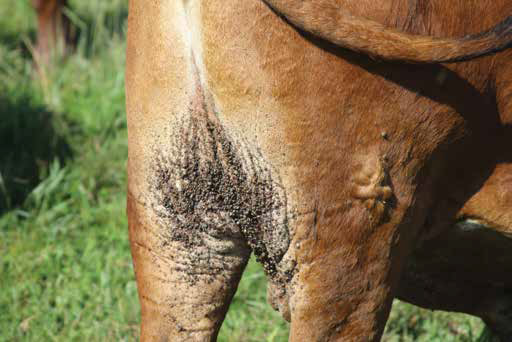
Some tick species have strains which secrete toxic substances in the saliva, which can cause a variety of syndromes; some tick toxins cause paralysis in cattle and small stock (spring lamb paralysis, Karoo paralysis, angora kid paralysis). A different toxin found in Hyalomma truncatum causes dermatosis and immune suppression in calves, called sweating sickness, and which can lead to fatalities. Advanced cases may be irreversible – even if the ticks have been removed. Angora goat kids develop a paralysis on exposure to heavy burdens of Rhipicephalus warburtoni, which cluster inside the ears of newborns.
Certain tick species are the vectors of various bloodborne organisms, including Rickettsia ruminantium, Babesia spp., and Anaplasma marginale. The Theileria infection known as corridor disease, and which is harboured in African buffalo, still remains a threat in KwaZulu-Natal if ticks infected with Theileria mutans infect cattle farms adjoining the Hluhluwe/Umfolozi parks. Other Theileria spp. can cause severe losses in game animal species such as roan and sable (see Monograph 3). Many of these tickborne diseases cause fatal infections and/or considerable economic loss.
Domestic ruminant livestock are susceptible to heartwater, and the disease is transmitted by Amblyomma hebraeum. It affects cattle in the wetter parts of the country, and is a very big problem in some commercial sheepand goat-farming areas like the Eastern Cape.
Tick conditions seen in cattle
Tick conditions in small stock
Selection of tick control methods in livestock and game
Deciding what tick control strategy to use in South Africa is complex, because of the many factors involved: the host species, the species of tick, the predominant tick-borne diseases, and the type of farming.
Part 1 is dedicated to a general discussion of tick control methods, as well as a summary of the problems of tick-borne diseases. Acaricides are still the mainstay of tick control in animal health, and therefore much of this section is dedicated to their use, management, efficacy and resistance. In the absence of a specific tick vaccine for South African conditions, acaricides will continue to be used as the main control measure – but need to be used judiciously to maximise their useful life.
Part 2 presents a brief discussion of tick-borne diseases, which will be dealt with in much more detail in Monograph 3 of the series.
Part 3 approaches tick control methods based on the main target tick species, because of the very different conditions and also the different tick-borne diseases. In livestock the authors adopt a geographic approach based on the distribution of tick species and the tick-borne diseaseswhich occur in various parts of South Africa.
GENERAL TICK CONTROL METHODS
ACARICIDES
For an overview of the history of acaricide use in South Africa, see Monograph 1 of this series. Currently there are six main chemical groups of acaricides that are registered for use in South Africa, in terms of the Fertilizers, Farm Feeds, Agricultural Remedies and Stock Remedies Act (36 of 1947). Some combinations of specific actives from different groups are also available. Acaricides for tick control are available in various formulation types, depending on their chemical nature. Table 1 summarises the main characteristics of each of the chemical groups and their different actives. A discussion of each follows, with reference to the specific effect on ticks.
The action of acaricides
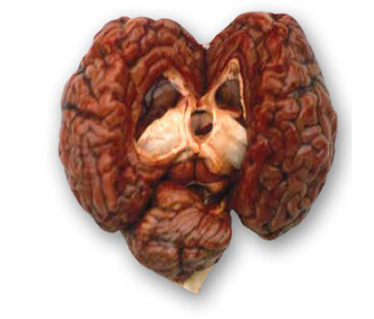
Most currently used acaricides act on the neurological system of the tick – at the neural synapses of the central nervous system (CNS), the neuromuscular junction (NMJ) or the receptors in the brain (see Figure 9) – producing paralysis and death. The stimulatory neurotransmitters at the synapses in invertebrates are acetylcholine in the CNS and glutamate in the NMJ, while gamma-amino butyric acid (GABA) is an inhibitory neurotransmitter in the CNS. Current knowledge no longer places any importance on the speculated effect of the macrocyclic lactones (MLs) on GABA. This differs from the mammalian nervous system, which does not have chloride-gated glutamate channels or GABA.
Insect growth regulators (IGRs) are currently the single exception among the acaricides, as they control ticks by interfering with the development of the larvae into adults. There are two groups of growth regulators: juvenile hormone mimics (which mimic juvenile hormone and prevent immature insects and ticks from moulting and becoming adults) and chitin inhibitors (which interfere with the formation of chitin during the moulting process, thus preventing successful moulting to the next stage of development).
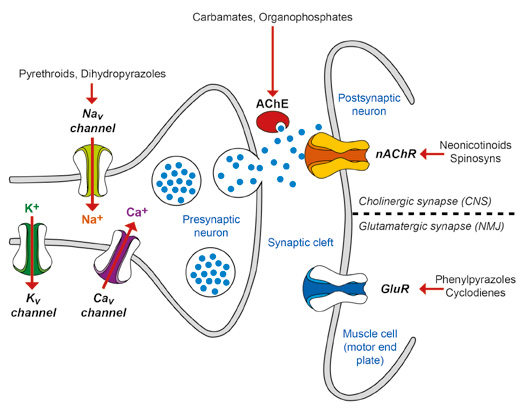
Figure 9 A diagram of an invertebrate synapse showing the site of action of some acaricides (modified from Ikonomopoulos and King, 2013).
Table 1. Summary of receptors targeted by acaricides and the effects on ticks.
| Acaricide | Receptors targeted | Effect on ticks |
|---|---|---|
| Organophosphates Carbamates | Inhibits acetylcholine esterase, and so stimulates the acetylcholine receptors | Stimulation of nerves resulting in spasms |
| Macrocyclic lactones (MLs) | Stimulates chloride-gated glutamate receptors | Stimulation of nerves resulting in spasms |
| Pyrethroids | Causes sodium (Na) influx into nerves | Stimulation of nerves resulting in spasms |
Table 2. Selective toxicity of different groups of actives.
| Active | LD50 Mammal mg/kg | LD50 Insect mg/kg | Ratio of selectivity (safety index) |
|---|---|---|---|
| Carbamate | 45 | 2,8 | 16 |
| Organophosphate | 67 | 2,0 | 33 |
| Organochlorine | 230 | 2,6 | 91 |
| Pyrethroid | 2 000 | 0,45 | 4 500 |
Unlike the conventional acaricides (listed above), which affect the CNS causing almost immediate death, the effect of growth regulators on the tick population is not immediate. Rather it takes time, depending on the life cycles of the different ticks/insects, until it begins to control the tick population (the immature stages cannot develop into the next stage and eventually die off).
Acaricides currently registered in South Africa
The acaricides currently used in South Africa are summarised below – with reference to their action, use and limitations. Please note that the withdrawal times are discussed in general, as they may vary depending on the delivery, formulation, dose and local regulations. The label recommendations should always be consulted for all acaricides, in this regard.
Oganophosphates (OPs)
The organophosphates are a large group of chemicals which are all neutral esters of phosphoric acid. The action of OPs is due to them mimicking the action of acetylcholine esterase. The most commonly used OPs for tick control are chlorfenvinphos for ticks on cattle, and diazinon and triazophos for lice and maggots on sheep and goats.
Action: OPs act by inhibiting the action of acetylcholine esterase, so allowing a buildup of acetylcholine at synaptic junctions. Ticks exposed to OPs experience nerve spasms, which results in death and detachment from the host. The chemical is ingested by the “tarsal effect” – which means that the tick absorbs the chemical by contact with it. There is some residual action two to three days after the OP-containing dip is deposited onto the skin of a treated animal. All species of ticks are controlled.
Efficacy: OPs are effective against all tick species.
Formulations: OPs are available as emulsifiable concentrates used in plunge dips and sprays. There are also formulations for local dressing. This group is not used in pour-ons, due to their narrow safety margin.
Toxicity: OPs are the most toxic acaricides, both for host animals and operators. Intoxication in livestock as a result of overdosing causes increased salivation, mydriasis and muscle tremors. In early cases of poisoning, the symptoms may be reversed by administering atropine. OPs are highly toxic for invertebrates, mammals, birds and aquatic animals. Their widespread use in farming areas contributed to the disappearance of oxpeckers in large areas of the country.
Resistance: The resistance to OPs in ticks, mainly the group referred to as blue ticks – Rhipicephalus (Boophilus) spp. – was prevalent by the 1970s. Cessation of OP use because of resistance and the emergence of newer and safer products, has led to them being effective once again in many places. There is cross-resistance between this group and the carbamates.
Residues: OP residues are considered a human health hazard, and so their use is contraindicated in lactating animals. Withdrawal periods of 30 days are imposed on treated animals intended for slaughter. Fleece residues of OPs are not permitted in EU countries – and this precludes OP use on sheep whose wool is intended for export to these countries.
Carbamates
The carbamates developed after 1960 are closely related to the organophosphates, as they have a similar action. The two actives used in veterinary science are carbaryl and propoxur.
Action: The carbamates have an anticholinesterase action which causes paralysis of the parasite.
Efficacy: Carbamates are effective against all species of ticks.
Formulations: Carbaryl is available as a wettable powder, which is used as a spray or hand-dressing solution for tick control in cattle.
Toxicity: The carbamates have a low toxicity compared with the organophosphates.
Resistance: There is cross-resistance between OPs and carbamates.
Residues: A seven-day withdrawal period is usually imposed for treated animals.
Pyrethroids
The synthetic pyrethroids used for tick control in livestock are analogues of the plant-derived pyrethrum, which has a short period of action due to its low potency and instability/photolability. The later generation synthetics now used are more UV stable than the early analogues such as allethrin, phenothrin and tetramethrin. The isomer content of some pyrethroids is expressed in ratios of cis to trans isomers, e.g.
70:30, where the cis isomer has approximately six-fold better efficacy than the trans isomer. This is sometimes mentioned by manufacturers of cypermethrin, as the cis isomers are generally more active than their trans equivalents. Pyrethroids currently used for tick control in South Africa are flumethrin, cypermethin, deltamethrin, cyfluthrin, and alphamethrin.
Action: Pyrethroids cause an influx of sodium into the nervous system – causing excitation, spasm and death. The ticks, especially the males, may however remain attached after death, which sometimes leads to assumptions that pyrethroid products are not working. The various actives have slightly different actions – for example, permethrin has a repellent effect on ticks, and flumethrin has an ovicidal effect on the eggs of female ticks which contributes to the overall control. Deltamethrin has the added advantage of having a repellent effect and a high activity against flies.
Efficacy: Pyrethroids are effective against all tick species.
Formulations: Pyrethroids are available as liquid concentrates. Emulsifiable concentrates or suspension concentrates must be diluted before applying as dips or sprays, and have a residual effect for roughly five days. Unlike other chemicals used in dip tanks, they do not strip from the dip wash, and therefore the initial fill rate is usually used for replenishment. Pyrethroids are included in many pour-ons which have a residual effect for up to seven days. They are also added to some tick greases and wound oils. Pyrethroids are sometimes combined with piperonyl butonyl to enhance their activity by inhibiting the breakdown by microsomal enzymes which metabolise pyrethroids in the tick.
Toxicity: Pyrethroids have a broad safety margin and are safe for mammals and birds, but are toxic to fish, aquatic vertebrates, and invertebrates. Pyrethroid pour-ons may cause severe peripheral nerve irritation – especially in dairy cattle. They are also known to cause severe irritation in sensitive human individuals.
Residues: There is little problem with residues and therefore pyrethroids can be used in lactating dairy cattle. Pyrethroids registered for use in dairies generally have a zero milk withdrawal period.
Amidines/formamidines
There are only two members of this group used in veterinary science: amitraz and cymiazole. The latter is less potent and is used in combination with other actives, for example pyrethroids.
Action: The amidines have an antagonistic effect on the octopamine receptors of the brain of the parasite, where they inhibit the enzyme monoamine oxidase. On ingestion of the chemical, ticks become hyperexcitable – causing them to hotfoot or wander around aimlessly until they die from exhaustion. There is a moderate repellent effect causing them to leave the host before completing a meal.
Efficacy: Amidines are effective against all tick species.
Formulations: Amitraz is available as wettable powders and liquid concentrates – viz. emulsifiable concentrates – and these are used as dips or sprays which have a registered residual effect (dipping interval) of seven days, and as pour-on formulations which similarly have a residual effect for seven days. Amitraz suspensions in the dip tank are unstable below a pH of 7, and therefore dip wash must be stabilised using lime to prevent breakdown.
Toxicity: Amitraz has a broad safety margin, especially in sheep. Sedation is the only known adverse effect known in animals and humans. It is therefore relatively safe for host animals and operators. Some formulations may, however, become toxic on long storage and must never be used after the expiry date. Amidines are safe for use in areas where oxpeckers are prevalent. They break down rapidly after disposal, but must not be discharged into water bodies such as dams or rivers as they are toxic for aquatic life.
Resistance: There is some resistance to amitraz, especially in the Eastern Cape and KwaZulu-Natal, but this is less prevalent than pyrethroid resistance.
Residues: The withdrawal period is usually seven days for meat and none for milk – depending on the formulation.
Macrocyclic lactones (MLs)
The MLs are chemical fermentation derivatives of soil bacteria in the genus Streptomyces. There are two distinct groups – the
avermectins and milbemycins. Ivermectin is an avermectin which is used as an acaricide with systemic or local action, depending on the route of application.
Action: The mode of action is not clearly understood, but ivermectin is known to stimulate chloride-gated glutamate receptors. This effect is less significant in mammals which do not have glutamate receptors.
Efficacy: The MLs are only registered for the control of the one-host ticks, because these ticks have longer and more frequent exposure to their treated cattle hosts than twoor three-host ticks. The treatment with shortacting MLs must be re-applied at 21-day periods. Long-acting formulations must be repeated every 76 days.
Formulations: The MLs are used as injectables and their particular usefulness is for the control of resistant blue ticks. They are also available in South Africa in dip and jetting formulations, which are registered for sheep and goats.
Toxicity: The MLs have a high safety margin in most livestock. In healthy animals, MLs are deposited in the fat and are released gradually into the system, but in animals in poor condition toxicity can arise – especially with moxidectin. Some MLs may have a negative impact on the biology of dung beetles, and if this is a concern the label must be checked for the extent of the effect. They can be used with less negative impact in winter when dung beetles are not active.
Resistance: Resistance to MLs has been seen in some countries.
Residues: There is a 21-day withdrawal period, and so MLs are not used in dairy animals, except for eprinomectin which has no residues in milk.
Insect growth regulators (IGRs)
The chitin inhibitors and insect juvenile hormone mimics fall under the insect growth regulators.
Action and efficacy: The chitin inhibitors (usually benzoylurea or suphonyl urea compounds including fluazuron (Acatak); novaluron; diflubenzuron and triflumuron), interfere with the formation of chitin which is the carbohydrate needed for the exoskeleton. The only two members of the group currently registered for tick control are fluazuron and novaluron. They have a systemic effect which prevents the development of the eggs and immature stages of blue ticks into adults – Rhipicephalus (Boophilus) spp. Because of this, there is an initial delay before tick control is achieved, and so concurrent application of another acaricide will be necessary in cases of concurrent heavy tick infestation, and multi-host tick treatment has to be continued in conjunction with fluazuron/ novaluron to combat twoand three-host ticks.

Figure 13 A chitin inhibitor is one of the two groups of IGRs that acts on chitin, which is an essential component in the tick exoskeleton and eggs.
Formulation: Fluazuron (the only currently registered product being Acatak), is applied as a pour-on formulation which should be administered at the beginning, middle and end of the season for effective blue tick control. There are injectable formulations alone and/or in combination with ivermectin in development.
Toxicity: Fluazuron is non-toxic, except for aquatic invertebrates, and must not be disposed of in water bodies.
Residues: Fluazuron is not suitable for lactating cattle as it has a six-week withdrawal period after application, because the active ingredient is stored in fat and is slowly released.
New/emerging products
Phenylpyrazoles
Only one of the two representatives of this group, Fipronil, is used on livestock currently and this mainly in Latin America. Topical application results in surface (nonsystemic) translocation over the entire body with significant deposition in the sebum, sebaceous glands and hair follicles. Oral administration results in significant amounts being deposited in body fat which serves as a depot, thus providing residual action against target parasites.
Action: Phenylpyrazoles act on the central nervous system by preventing chloride ions from entering the nerves through modulation of both gamma-amino butyric acid (GABA) and glutamate receptors – but mainly the latter. These are important neurotransmitters in the central nervous system of arthropods. As mammals do not have glutamate-gated chloride channels, this may explain the wide margin of safety in mammals. Oral LD50 in rats = 100 mg/kg and dermal LD50 = >2 000 mg/kg. However, phenylpyrazoles are highly toxic to aquatic life and beneficial insects.
Table 3. Activity and toxicity of acaricide groups currently registered in South Africa.
| Chemical groups | Actives | Efficacy for blue ticks | Efficacy for multi-host ticks | Toxicity for host | Toxicity for environment |
|---|---|---|---|---|---|
| Organophosphates (OPs) | Diazinon | ++ | ++ | 4 | 4 |
| Chlorfenvinphos | ++ | ++ | 3 | 4 | |
| Triazaphos | ++ | ++ | 4 | 4 | |
| Carbamates | Carbaryl | ++ | ++ | 3 | 4 |
| Pyrethroids | Deltamethrin | ++++ | +++ | 1 | 2 |
| Flumethrin | ++++ | ++++ | 1 | 2 | |
| Cypermethrin | +++ | +++ | 1 | 2 | |
| Alphacypermetrin | +++ | +++ | 1 | 2 | |
| Fenvalerate | +++ | ++++ | 1 | 2 | |
| Cyalothrin | +++ | +++ | 1 | 2 | |
| Amidines/ Formamidines | Amitraz | ++++ | ++++ | 2 | 1 |
| Cymiazole | ++ | ++ | 2 | 1 | |
| Macrocyclic lactones (MLs) | Ivermectin | ++ | - | 1 | 2 |
| Moxidectin | ++ | - | 2 | 2 | |
| Doramectin | +++ | - | 1 | 2 | |
| Abamectin | +/td> | - | 1 | 2 | |
| Insect growth regulators (IGRs) | Novaluron | ++ | - | 1 | 2 |
| Fluazuron | +++ | + | 1 | 2 | |
| Phenylpyrazoles* | Fipronil | ++++ | ++++ | 1 | 2 |
| Spinosyns* | Spinosad | +++ | - | 1 | 1 |
Key:
1 – 4: low to very toxic
no effect
+ some effect
++ moderate effect
+++ good effect
++++ very good effect
*Not yet registered for tick control on livestock in South Africa.
Efficacy: Fipronil has a broad spectrum of activity, being very effective against all tick species.
Formulations: In Latin America, pour-ons are used to control ticks and hornflies on cattle. A bolus for use in cattle has been patented, which provides significantly longer protection against ticks than most other currently used acaricides.
Toxicity: Fipronil has a wide safety margin in mammals. Environmentally, it is toxic to many species of beneficial insects such as bees, and is highly toxic for fish and aquatic invertebrates. It is stable in the environment, with a half-life of 125 days in soil.
Residues: Fipronil is excreted mainly through the faeces, but 5% is excreted in milk. The registration restrictions for use in lactating cattle are currently unclear.
Spinosyns
Spinosyns are compounds derived from the fermentation of various soil fungi. They act on the GABA and nicotinic receptors of ectoparasites, have low toxicity, and do not persist in the environment. They are currently only used in small animals for the control
of ectoparasites, but have been shown to be effective against Rhipicephalus (Boophilus) microplus. It is suggested they may be useful as rotation products to combat amitraz resistance.
Synergism and potentiation of acaricide combinations
Synergists are products which, while they do not have an insecticidal/acaricidal effect of their own, improve the efficacy of the pesticide with which they are combined. Sometimes, the administration of two compounds to an insect/tick produces an effect greater than the sum of the individual actives (1 + 1 = 3 effect). This is termed potentiation (Brown et al., 1967).
With the emergence of resistance to most of the available acaricides, various strategies were employed to counter this.
The rotation of acaricides was employed with limited effect in some countries where the emergence of multi-resistant tick strains made all acaricides ineffective. Based on work done on mosquito control and crop protection pesticides, scientists looked – as long ago as in 1937 – at the use of substances like piperonyl butoxide which potentiate the effect of pyrethrum (and more recently various other insecticides like carbamates and pyrethroids) by inhibiting their breakdown in the body by cytochrome P450 monooxygenase (Sparks and Byford, 1988).
Potentiating combinations of acaricides such as OPs and synthetic pyrethroids, and permethrin and amitraz, have been demonstrated (Guerrero et al., 2013). In South Africa, various potentiating combinations of acaricide were developed to address the problem of multi-acaricide resistance. On the basis of data showing synergism/potentiation, these combinations were registered to assist tick control on farms which were severely affected by acaricide resistance. The advantages and disadvantages of synergistic combinations are theoretically:
- A broader spectrum of activity against a wider range of parasites. For example, if amitraz is used alone in a dip tank or as a pour-on, only ticks are controlled while flies and lice of various species could become a serious pr
- It is possible to reduce the amount of each active used, thus making the end product cheaper to produce and also safer for both the host being treated and the envir
- The likelihood of an insect population having the genes for resistance to two or more active groups with different modes of action, decreases exponentially.
- The increased efficacy of synergistic actives is also likely to reduce the chances of resistance occurring, and to reduce the speed at which it dev
- The ratio of the various actives used in combination is crucial if maximum synergistic benefit is to be achieved – g. the optimum pyrethroid:amitraz ratio is 1:7.
Other tick remedies
Botanical remedies
Many research groups are testing botanical extracts for their acaricidal effects. There are several actives which have shown promise but, to date, none have been registered with regard to efficacy and safety. In general, as in the case of pyrethrin, natural botanical extracts (as opposed to the synthetic version of the plant actives), are less effective because they have a short action, are unstable, and are rapidly broken down by sunlight. However, some such as tobacco extracts, which contain nicotine, and which are used illegally as insecticides – are highly toxic to hosts, operators and the environment.
In South Africa claims have been made for the efficacy of aloe extracts on parasites – which has led to some feed companies including this extract in commercial rations/ licks. Controlled experimental trials have showed that this administration had no effect on tick burdens (Fourie et al., 2005; Spickett et al., 2007).
Diatomaceous earth
Diatomaceous earth (DE) is composed of the calcite shells of marine diatoms, and is sold as an acaricide. The manufacturers claim that DE kills ticks by interfering with their respiration and damaging the chitin coating. The claims for the efficacy of this material as a control measure for ticks are unsubstantiated by data – since no sound scientific or peer reviewed trials have been done. On the contrary, treated animals may develop lung pathology if the dust is inhaled in substantial quantities.
Environmental and safety aspects of chemical control
The newer acaricides on the market are much less toxic than the arsenicals used 50 years ago and the organophosphates, which are now used only in severe cases of resistance. Nevertheless, all acaricides must be used carefully by the operator and when applied to the host. Safe disposal is essential to prevent environmental damage.
Operator safety: All acaricides must be stored under lock and key and kept away from uninformed persons. Staff must be given proper training in the handling of acaricides, and in particular should avoid eating, drinking and smoking during mixing and application. Training courses for operators are available from the South African Animal Health Association (SAAHA) (refer to box on the next page). Protective clothing such as overalls, eyewear and gloves must be used – especially with OPs which are highly toxic and the pyrethroids which, although safer, may cause severe skin irritation. In the event of accidental exposure, wash the skin or eyes, change clothing, and ensure that the person receives medical care if necessary.
Safety of host: Overdosing with organophosphates can cause severe toxicity. Although atropine can be used as an antidote, its efficacy depends on early administration. Home-made pour-ons containing pyrethroids often cause extreme irritation, and in very severe cases the skin may be burnt to the extent that the epidermis and hair are shed. In these cases, the animal must be thoroughly washed with soap and water to remove the pyrethroid from the skin. It should be pointed out to farmers that the use of home-made products is illegal in terms of Act 36 of 1947, is unsafe, contributes to the development of tick resistance, and causes illegal residues in meat and milk.
SOUTH AFRICAN ANIMAL HEALTH ASSOCIATION (SAAHA)
Safe disposal of acaricides and the effects on the environment
Discarding used dip: When dip tanks are emptied for cleaning, it is essential that the dip is discarded safely and without it being a potential hazard to humans or animals – including all forms of wildlife, large or small. The accepted best practice for disposal is to pump used dip wash out onto an allocated area of ground, where the active ingredient is broken down by the combined action of sunlight and soil bacteria.
Select a disposal site for discarding the dip by choosing a level piece of ground near the dip tank and ploughing contour furrows to promote containment and to prevent run-off. Allow 0,5 ha for 15 000 ℓ. The disposal site must be at least 100 m from boreholes, streams, houses and public thoroughfares. The area must be fenced off and should be marked “DANGER SABS WW5” and “KEEP AWAY” in all local languages. There must be no run-off from this area into local watercourses. Alternatively, if sufficient surface area is not available and if the soil drains well, a circular, well-like hole, about 4 m in diameter and 1,5 m to 2 m deep, can be built not too far from the dip. The inside of this hole must not be cemented, because the dip wash is pumped into the pit and must slowly drain down into the soil. The disposal well must also be fenced and marked as described above.
Discarding containers: Empty dip containers should never be re-used for any other purpose, given the danger of poisoning. To render the containers safe before disposal, use the triple-rinse method as follows: Fill the container to a quarter of its volume with water, shake vigorously and empty the contents into the dip tank. Do this three times (e.g. with the first rinse, 4 000 ppm is diluted to 40 ppm, and with the third rinse to 0 ppm). Make holes in the container to render it unusable and bury it in a refuse pit dug especially for the purpose. The pit must be sited away from water sources and must be fenced off. Alternately, the containers can be made available for recycling.
Suspected acaricide inefficacy
Ineffective acaricide application is the most common cause of tick control failures, and is often mistaken for tick resistance (see later). The most common causes of efficacy failure are below strength dip wash and/ or incorrect application. The causes of these two factors are discussed below.
Below strength dip wash
Whether one is filling a dip tank, making up dip for a spray race, hand spraying or applying pour-on, the manufacturers’ instructions must be followed to the letter – to ensure that the concentration of the active ingredient is optimal. This is achieved by correct management of all aspects of dipping: filling the dip tank, dilution of the dip concentrate, correct replenishment, stirring, and overall management of the tank itself. Details on how to do this are available in Afrivet’s Practical guide to dip tank construction and management (Oberem and Oberem, 2014). The dip tank concentration must be monitored at intervals (at least at the beginning of each season and then possibly midway through the season), to ensure that it is up to strength. Below strength dip wash often results from the underestimation of the dip tank capacity, from incorrect replenishment, or from not keeping a head count of animals put through the dip tank.
Other factors that can affect the concentration of the dip wash are flooding of the dip by rain, and not allowing animals to drain – which returns the dip solution into the tank. The important dip wash residual on the animal can be removed or diluted by rain, or by animals swimming across rivers or walking through long, wet grass.
Insufficient control can result from failure to stir the dip in the dip tank by using 30 cattle (which must then be re-dipped later) or from using an expired, faulty or damaged product.
If an amitraz dip is used according to the conventional dipping method, it must be stabilised with lime sulphate or the dip will break down. If the dip is not stabilised, replenishment must be done using the total replenishment rates. This latter method is only economically viable if large numbers of animals are put through the dip every week.
Incorrect application
Poor application of dip solution is most common with hand spraying, because achieving sufficient wetting of the animal – especially the ears and underline – is difficult and labour intensive. Experts recommend applying 12 ℓ of dip per adult animal, in order to achieve proper wetting.
Poor wetting can result from dip tanks that are poorly constructed. The most common problem is failure to wet the head, which occurs when the jump-off area of the dip tank is not properly designed. If the entrance to the dip is not steep enough, the animal is not forced to jump into the tank and submerge its head – and instead swims into the dip tank, leaving the head unwetted. Dip tanks that are too shallow or filled with mud will also result in poor head wetting.
Real tick control failure must be distinguished from apparent failure, as detailed below:
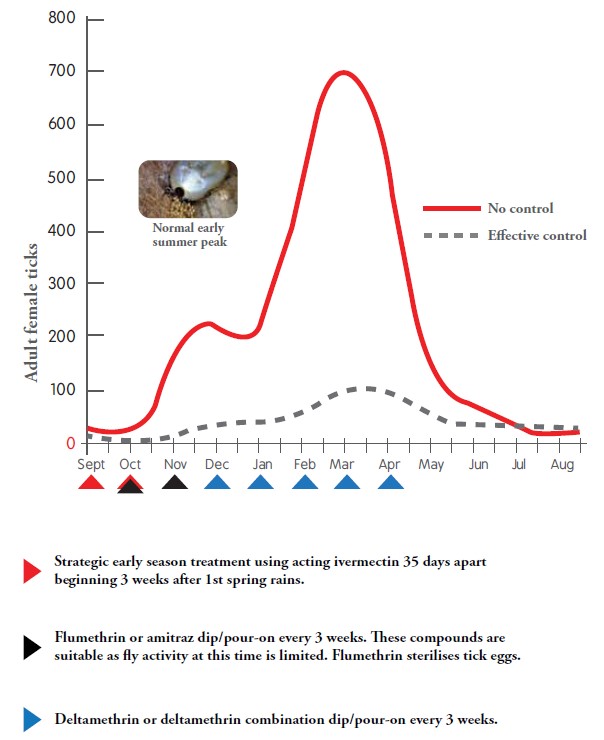
- Pyrethroids can take up to two days to kill ticks – the males may stay attached, despite being dead.
- Macrocyclic lactones take time to act, given the time required for the active to reach a peak concentration – whereafter the ticks have to feed before any effect on tick numbers can be observed. The initial effect will only be seen from eight hours after injection.
- Pour-ons may take two to three days to spread over the animal, and their effect on the lower body areas (belly and legs) will be slower than on the head and neck.
- Ticks may climb on animals after the residual effect is past (after roughly three days). This occurs especially when farmers put animals in new camps where there might be thousands of hungry ticks.
- Growth regulators will not kill adult ticks and treatment with dips that do kill adults (adulticides) might initially be necessary.
- When there is a massive population of ticks due to warm and wet conditions, more frequent dipping is required to reduce the numbers.
A quick acaricide efficacy test
Acaricide resistance and management
Since the use of arsenicals as acaricides, tick populations in Southern Africa – chiefly the blue ticks of cattle – Rhipicephalus (Boophilus) decoloratus – have developed resistance to each new active that has come onto the market (see Monogaph 1). The development of acaricide resistance appears to be an evolutionary inevitability when sufficient selection pressure is exerted on a tick population – regardless of the active involved. The impact of this on livestock farming is serious for a number of reasons. The arsenal of acaricides is shrinking as some resistance has been recorded for almost all of those registered for use currently – and new actives are not being discovered on a regular basis. Multi-acaricide resistant ticks have been collected on some farms in the Eastern Cape, which have made tick control almost impossible on the properties involved. New acaricides are being developed at a slow pace, and so careful management of currently available products must be practised.
Definition: Acaricide resistance in a tick species can be defined as the presence of a population, on a particular property, which is able to survive exposure to formerly lethal levels of a specific acaricide or acaricides. It arises due to genetic selection of naturally occurring mutants in the population, which are selected when they are regularly exposed to an acaricide.
Molecular mechanisms of acaricide resistance: Researchers have postulated three mechanisms for acaricide resistance in ticks: target site resistance, metabolic resistance, and receptor site resistance (Guerrero et al., 2012). Because blue ticks are one-host ticks, they are exposed most frequently to acaricides in places where regular (weekly or biweekly) dipping/treatment is carried out, and are the most likely to be affected by acaricide resistance. Most studies of acaricide resistance have concentrated on these species.
Target site resistance
Studies on R. (B.) microplus have shown that target site resistance arises due to amino acid mutations which confer resistance. This has been studied and observed in Rhipicephalus (Boophilus) spp. against pyrethroids, where the target site is the voltage-gated sodium channels. Three mutations can arise at three different domains (1, 11 and 111). The phenotypic effect of each of these differs significantly. The Domain 111 mutation (phenylalanine to isoleucine) confers a high-level resistance to permethrin, cypermetrin, and flumethrin, while a mutation at Domain
11 confers a lesser degree of resistance. The target site for amitraz resistance is not confirmed, but is thought to be the monoamine oxidase (MAO) receptor, octopamine.
Metabolic resistance
Metabolic resistance causes an increased breakdown or inactivation of an acaricide. In R. (B.) microplus metabolic resistance has not been well defined – but is generally attributed to a variety of enzymes, which are known to play a role in OP resistance.
Prevalence of acaricide resistance in South Africa
Receptor site resistance
Receptor site resistance is when the receptor site is altered (down regulated) and becomes less sensitive to the active ingredient/molecule involved.
Management practices which promote resistance
- Buying in resistant ticks: Buying in animals infected with resistant ticks will result in the rapid establishment of resistance on a farm. This has been responsible for the rapid development of multi-remedy resistance in the Eastern Cape, where some farms can no longer implement tick control effectively. It is therefore essential to quarantine cattle introduced onto a farm and to ensure they are free of ticks, before allowing them into the main her
- Long-term use of the same active: Using a particular active continuously applies a specific selective pressure to a tick population which can result – if the gene for resistance is present on the property or is inadvertently introduced with new animals from elsewhere – in the eventual establishment (survival) of resistant ticks on the farm. These resistant ticks are then not controlled and multiply at the expense of the susceptible population, and ultimately dominate the population. The development of resistance can be effectively delayed by ensuring good dip management practices like optimal application and management of the dip tank to ensure proper control with the chosen active/s.
- Use of pour-ons (high concentrations): The use of pour-ons has increased the prevalence of tick resistance to pyrethr The reason for this is probably the high concentration of acaricide and the consequent higher selection pressure in the pour-on formulation and/or the consequent fall in concentration and the long “tail” of falling concentration – during which time individual ticks, which are resistant at low active concentrations, may be selected (Taylor, personal communication). Home-made pour-ons are a serious cause of resistance, because of the extremely high active concentrations which can also cause very severe irritation. These home-made pour-ons are also dangerous because they cause residues and toxicity.
- The role of combinations in the development of acaricide resistance: Combination products consisting of actives with no potentiation, will immediately begin the selection process for resistance if the gene for resistance to both actives already exists or is introduced onto the property. Once resistance has developed to the combination, there are in effect two less options available to the stock owner to control
On the other hand, where synergism and higher efficacy results from the combination used, combinations can reduce the development of resistance. For example, if a pyrethroid/formamidine combination is used, the formamidine will kill the pyrethroidresistant ticks that may be introduced (and vice versa), thus preventing the development of resistance where the gene does not exist on a property.
Investigating tick resistance
The first step to resolving a problem with acaricide resistance on a property, is to determine whether one is dealing with resistance or poor efficacy (see the quick efficacy test, above). If the quick efficacy test indicates that the acaricide in current use is not effective, other possibilities like an expired
batch, damage of the product during storage and below strength applications, must all be excluded. If these possibilities have been eliminated it can be suspected that acaricide resistance may be present. In this case, ticks should be collected and submitted to a suitable laboratory. Veterinary companies will assist with this by providing suitable containers and standard testing forms.
Tick collection must be done meticulously, as follows:
- Make holes in the lid of the containers and line them with tissue to keep the ticks dry and to prevent fungal growth.
- Collect 40 to 50 engorged female ticks of each species, from the cattle of various stock owners in the case of communal dip tanks.
- Collect only fully-engorged female ticks and do not mix different species of ticks in the same container.
- Wash hands before and after the tick collection, to prevent infection with tickborne organisms and to prevent the ticks from being contaminated with acaricides.
- Complete the tick resistance testing request form in full, wrap it around the specimen holder, and keep it in place with an elastic band.
- Store the container of ticks in a cool place and send it to the laboratory recommended by the supplier, as soon as possible, and preferably by courier.
Various tests for establishing acaricide resistance are available and are reviewed in Monograph 1 of this series.
Managing resistance
Once resistance to a specific dip has been confirmed on a farm, the dip used must be changed to a group against which ticks have no resistance. Use an active from a different group for immediate effect, because there is cross resistance between actives within the same group. The generally recommended principle is to use the selected group meticulously and for as long as possible – while monitoring the dip activity carefully. Using one chemical group only will relieve the selective pressure for the resistant active, and it may eventually be possible to use this group again. Macrocyclic lactones such as ivermectin and IGRs (fluazuron) can be used where multi-resistant blue ticks are a problem.
Changing the dip wash in a dip tank
Preventing resistance
The single most important practice for safeguarding a farm from the scourge of resistant ticks, is to prevent their introduction to a farm (biosecurity). When new cattle are brought in they must be held in quarantine
– i.e. in a camp where they are isolated from the animals on the rest of the farm. They must be treated with at least two different chemical groups with a week interval, and then must be carefully observed to ensure that the ticks have been fully controlled. The chemicals used must preferably not be those used by the previous owner. This is to ensure that all resistant ticks are killed off, and are not allowed to establish themselves on the farm. Failure to practice biosecurity can lead to the presence of multi-resistant ticks on farms – as seen on some Eastern Cape farms where cattle farming has become almost impossible because of this.
Although the danger of developing resistance is less with plunge dipping than, for example, when using pour-on formulations (especially home-made products), the danger of resistance developing increases with the frequency of chemical use. However, managing the dip tank and dipping intervals correctly, and according to label recommendations, will effectively reduce tick numbers and therefore the likelihood of resistance. The strategic use of macrocyclic lactones (ivermectin) or IGRs (fluazuron) can be used as an aid to plunge dipping, to prevent the development of peaks in the blue tick population. However, any change in tick control programmes must be carefully considered because it will have consequences for the control of tick-borne diseases. As discussed in the introduction to this Monograph, being mindful of all factors which promote integrated tick control (ITC) will reduce the usage of acaricides – and therefore reduce the likelihood of acaricide resistance.
TICK RESISTANT/TOLERANT BREEDS
Certain breeds of cattle – specifically the Zebu or Bos indicus breeds – are able to acquire resistance to ticks on exposure. The mechanism is thought to be a local histamine-mediated or cell-mediated immune reaction (see box insert below) which discourages ticks from feeding (Oberem, 1984). This resistance is not absolute, but reduces the number of ticks which attach. This is clearly an advantage as it reduces the need for regular tick control. Because this is an acquired resistance, lack of exposure to ticks, for example under intensive treatment, will render Zebu animals susceptible. Failure to select these animals for their tick tolerance may however lead to the loss of this genetic characteristic. Tick tolerance may also break down when animals are stressed or infected with chronic diseases.
Since Zebu cattle breeds are more tick tolerant than European breeds, they are a valuable resource on extensive cattle farms because of the reduced input costs required and their general hardiness. Nevertheless, some local tick control may be needed to reduce organ and tissue damage.
Various authors have reported that certain small-stock breeds can develop resistance to ticks (Abdul-Amir and Gary, 1987). However, a limitation of tick-resistant animals is that they may not have the required production characteristics needed for commercial farming, such as milk production or adequate carcass weights in the case of beef cattle or mutton sheep. Various breeds are available which can be used as alternatives – for example the Meatmaster which is a better mutton sheep than the Pedi.
The immune mechanisms of acquired tick resistance in animals
TICK VACCINES
The development, application and efficacy of anti-tick vaccines have been extensively reviewed by Willadsen (2004). The reason for developing these vaccines was the numerous advantages they would have over chemical control. A vaccine would be environmentally more acceptable, sustainable, cheaper to produce and probably easier to register with the authorities (limited residue and safety work required) than an acaricide. In addition, the strategy of using tickresistant animals – while notably important in resource poor countries – has some limitations. Not all B. indicus (indigenous) breeds are able to develop sufficient resistance to ticks, and those that do may not retain desirable production characteristics. An additional advantage is that resistance is unlikely to develop to a vaccine, while resistance to acaricides is almost inevitable with their continued usage.

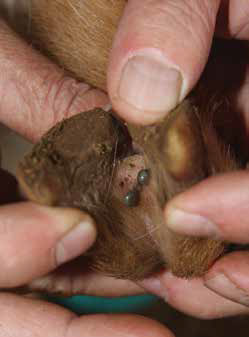
Figure 19 A dissection showing the intestines of a tick. Tick vaccines have been developed using a gut antigen BM86.
Since it was known for many years that a crude tick homogenate could induce partial to solid immunity against ticks, the task of vaccine developers was to identify suitable immunogenic antigens which could be used as a recombinant vaccine. The antigens which developers focused on fell into two categories: exposed and concealed antigens. Exposed antigens are defined as those to which the host is exposed during attachment and feeding. Concealed antigens refer to those not part of the normal host–parasite interaction, and which therefore do not elicit an immune response on natural exposure to ticks. Researchers used different parameters to identify suitable antigens – including immune response, the importance of the antigen to parasite survival, and biochemical fractionation.
Various candidate antigens were investigated with different degrees of success, until the Australian research group at the Commonwealth Scientific and Industrial Research Organisation (CSIRO) isolated the BM86 protein molecule from R. (B.) microplus gut epithelium. This antigen induces a strong antibody response in the vaccinated host; the antibody is taken in by the R. (B.) microplus tick through ingested blood and directly affects the structural integrity of the tick’s gut.
Field trials using the recombinant BM86 produced in an E.coli expression system, showed that the main effect is on adult ticks. The vaccine reduced the number of maturing adults, and also caused adult mortality after engorgement. The main effect occurs in second and subsequent generations when reproductive performance was inhibited and numbers of larvae in the field were reduced. The vaccine is produced and used commercially in Australia under the name TickGARD. This vaccine became part of an integrated tick management system in which acaricide application could be reduced and resistant R. (B.) microplus could be effectively countered.
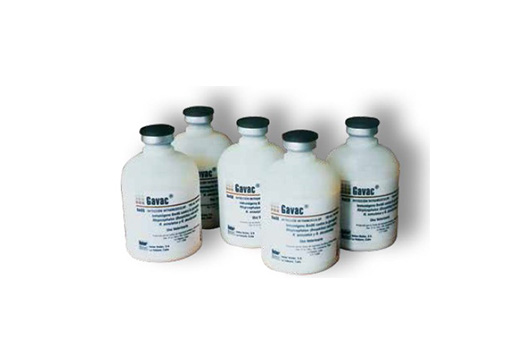
A BM86-based vaccine was developed in South America for local use under the name Gavac. There is sequence variation between BM86 isolates, which may cause the efficacy of these vaccines to vary in different geographic areas.
The currently available tick vaccines are not very effective against the blue tick species in South Africa, and thus the development of a BM86 vaccine against South African tick isolates would considerably enhance the control of Rhipicephalus (Boophilus) species.
BIOLOGICAL CONTROL METHODS
Oxpeckers
There are two species of oxpecker which occur in South Africa. Both the red-billed (Buphagus erythrorynchus) and yellow-billed oxpecker (Buphagus africanus) have evolved adaptations for eating ticks off mammalian hosts. They have sharp claws and short legs which allow them to cling to the hair coat of large mammals, where they use a scissoring action to comb through the hair in search of ticks. Oxpeckers can eat roughly 100 engorged adult ticks and up to 10 000 larvae per day. They remove ticks from all areas of the body and also clean wounds of fly maggots.
Arsenic and the OPs are toxic to birds and their use caused a drastic decline in oxpecker numbers – to the extent that the yellow-billed oxpecker is considered a vulnerable species in South Africa. The introduction of less toxic dips as well as various breeding initiatives have increased the numbers and distribution of the redbilled oxpecker considerably, and farmers in many parts of the country now experience the welcome sight of oxpeckers on their cattle. However, this is a densitydependent form of control, and so it is unlikely that the birds alone can adequately control ticks on a cattle farm or smaller game ranch. Other control methods haveto be used in conjunction with encouraging the presence of the birds. Pyrethroids and amitraz-containing products are entirely safe to use on farms where oxpeckers occur. These products have an “oxpecker compatible” statement on their labels.
Entomopathogenic fungi
Several naturally occurring soil fungi have been shown to cause a disease in insects as well as in ticks. Their use as a means of biological control has been investigated by researchers in various countries. Field trials have shown that application of the fungus to soil causes mortality and reduced fecundity of various tick species (Stafford and Allan, 2010). There are currently no commercially available products for tick control.
Insects
The parasitic wasp Ixodiphagus hookeri has been observed to parasitise ixodid ticks, including Amblyomma variegatum. Field trials conducted in Kenya showed that the release of these parasitoids reduced the A. variegatum burdens on cattle from a mean of 44 to 2. However, the numbers of R. appendiculatus on the cattle increased (Mwangi and Kaaya, 1997). More research needs to be done to determine the usefulness of parasitoids for tick control. This control measure is also density dependent.
Pastures and other plants
Melinis minutiflora (molasses grass) is a grass species which is native to the tropics and has established itself in other regions. It is a fastgrowing pioneer grass which gives good coverage of disturbed land. It is fairly drought resistant, but is intolerant of severe frosts.
The viscous foliage discourages insects and ticks, and has been observed to reduce the burdens of R. appendiculatus and R. (B.) microplus (Mwangi et al., 1995).
See Chapter 7 in Monograph 1 for a short synopsis on other plants with either physical (hooks) or chemical tick deterrents.
PASTURE MANAGEMENT
Management of pastures can help to reduce the number of ticks on cattle. The resting of camps, if done for sufficiently long intervals, may be helpful; if the grass can be burnt, this will help reduce tick numbers, especially blue ticks. Burning must be done in spring after the first rains when the blue tick eggs have hatched (but is less effective for three-host/questing ticks). Planted pastures are much less attractive to ticks than natural pastures, but even under zero-grazing conditions, the use of hay can lead to the introduction of sufficient ticks to cause disease.
TICK-BORNE DISEASES AND VACCINES FOR THEIR CONTROL
It is impossible to deal with the subject of tick control without referring to tick-borne diseases. In this section we provide an overview, but the subject will be discussed in detail in Monograph 3 of the series.
TICK-BORNE DISEASES IN CATTLE IN SOUTH AFRICA
The most important tick-borne diseases in cattle in South Africa are heartwater, babesiosis and anaplasmosis (the latter can also be transmitted by biting flies). Corridor disease caused by Theileria parva lawrencei, which occurs in buffalo in the KwaZulu-Natal national parks, poses a potential threat to cattle farms in this area and elsewhere where the vector occurs.
Depending on the geographic location in the country, one or all of the diseases may be present, and, for this reason, tick and disease control in different areas are dealt with in Part 3 of this Monograph.
Although the epidemiology of the specific tick-borne diseases varies, as a general rule with cattle, exposure to infected ticks immunises the young animals before they become fully susceptible to the disease, and in this way they become immune adults. If natural selection is permitted, animals that have greater genetic resistance will be selected, as occurred with indigenous breeds prior to “modern” farming methods (the same pertains to indigenous wild ruminants (heartwater) and ungulates (babesiosis)). These two factors – natural immunisation and natural selection – result in a so called “stable” disease situation, in which animals rarely develop clinical disease.
However, with farming practices such as tick control and the importation of animals into disease endemic areas, the situation becomes destabilised: tick control to counter the adverse effects of ticks may reduce the number of ticks below the threshold required for natural immunisation of cattle – resulting in the appearance of disease, even in indigenous breeds. In addition, the movement of disease-susceptible adult cattle into endemic areas results in severe clinical disease. The use of tickborne disease (TBD) vaccines therefore became necessary to immunise young and susceptible animals. This is because achieving a natural or stable situation can be difficult, especially in areas where all three tick-borne diseases and their different tick vectors occur. This is further complicated where both species of Babesia (bovis and bigemina) are present.
Considering the above, the recommendations on tick control and the control of tick-borne diseases in Part 3 are divided into different geographic areas, in order to simplify the discussion.
A TICK-BORNE DISEASE IN SMALL STOCK IN SOUTH AFRICA – HEARTWATER
The only tick-borne disease of importance in small stock in South Africa, is heartwater. Although some indigenous small stock such as Pedi sheep have been shown to be heartwater resistant (Yunker, 1996), most commercial sheep, and particularly angora and boer goats, are susceptible. They do not immunise on natural exposure, and also do not develop solid protective immunity after vaccination with the current Ball 3 blood suspension vaccine. In endemic areas where small stock are traditionally farmed such as the Eastern Cape, sustained tick control is the recommended method of disease control. Angora goats are shorn twice a year and cannot be dipped because of residue problems in exported fleece. Farmers have therefore resorted to treating animals prophylactically with oxytetracycline injections, 5 mg/kg every 11 to 14 days, in order to control heartwater. Newly developed attenuated and recombinant heartwater vaccines have shown promise for being safe and effective in small stock and will hopefully come onto the market without being delayed unduly by government and regulatory authorities.
CURRENT TBD VACCINES
A brief summary is given here for each of the three available TBD vaccines. Monograph 3 of this series will review more thoroughly the composition, mechanism and usage of TBD vaccines.
Heartwater vaccine – Erlichia (Cowdria) ruminantium
Vaccination against heartwater is merely a form of controlled exposure to the disease. This vaccine is essentially an infected blood suspension of live unattenuated organisms. This is produced by infecting susceptible sheep with the Ball 3 heartwater isolate. The vaccine induces a rickettsemia, and in calves younger than three weeks (better less than seven days) this effectively immunises them without causing clinical disease. In older cattle a fever reaction usually results, which must be treated with tetracyclines to prevent the animal developing heartwater.
The blood suspension is stored at -70 °C and is dispatched on dry ice to the farmer. It is then thawed shortly before use and must be given intravenously. Although the vaccine was originally registered for cattle and sheep, veterinarians who work in heartwater areas with small stock report that even young sheep, and particularly angora goats, develop severe clinical disease due to the vaccine. In heartwater endemic areas intensive control of Amblyomma ticks by bellybathing is successful in controlling heartwater, and vaccination with the blood vaccine has been discontinued by commercial farmers – especially for angoras and wool sheep.
Facts about the current Ball 3 unattenuated heartwater blood vaccine:
- Expensive to produce (200 to 300 sheep are used annually for production).
- Costly to store and transport (needs to be kept in liquid nitrogen and transported on dry ice).
- A challenge to keep uncontaminated in respect of extraneous organisms.
- Difficult to administer (it must be given intravenously).
- Except in very young calves, the vaccinated animals develop the disease which must then be treated.
- Treatment is costly (oxytetracycline injections) and labour intensive.
- Animal losses due to heartwater resulting from the vaccination sometimes occur.
- Young sheep and goats cannot be safely vaccinated.
- Pregnant animals cannot be vaccinated.
- Protection is only provided against a limited range of isolates.
Babesiosis vaccines
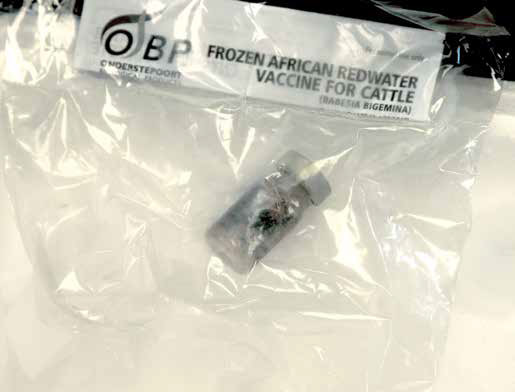
There are two separate live attenuated vaccines available in South Africa for babesiosis. The African redwater vaccine contains B. bigemina and the Asiatic redwater vaccine contains B. bovis. The vaccines are blood suspensions produced by infecting tick-free cattle.
Babesia vaccines are dispatched on dry ice and are thawed carefully just before use. They are administered intramuscularly to calves at six months of age. Vaccinating adult animals is risky as they can develop vaccine reactions which have to be treated with babesicides. This can compromise the vaccine immunity and animals may need revaccination. Pregnant animals cannot be vaccinated.
Both vaccines must be applied in areas which experience both B. bigemina (African babesiosis) and B. bovis (Asiatic babesiosis), as there is no cross-resistance.
Some inefficacy problems have been experienced with the local vaccine – allegedly due to poor viability as a result of inadequate cooling on storage. Australian producers of babesiosis vaccines report that storing the vaccine in AI straws in liquid nitrogen, rather than in vaccine bottles on dry ice, improves the shelf life and results in better efficacy. They produce a three-in-one vaccine containing both Babesia spp. and anaplasmosis organisms.
Anaplasmosis vaccine
The current anaplasmosis vaccine is a blood suspension of the attenuated organism Anaplasma centrale. Vaccination is done between three and nine months, which is the age at which animals will not develop severe vaccine reactions.
The vaccine is supplied frozen on dry ice and must be thawed carefully, like the other blood suspensions. Vaccine reactions develop between the fourth and sixth week after vaccination. During this period animals must be observed for signs of fever and symptoms. Vaccine reactions can be treated with 10 mg/kg oxytetracycline, repeated if necessary after 24 hours, or with a single administration of a long-acting formulation at 20 mg/kg.
Anaplasmosis vaccination can be done simultaneously with redwater vaccination. Immunity develops roughly two months after vaccination and lasts about four years – which is basically the productive lifetime of cattle. It must be borne in mind, however, that even when animals have developed a good immunity, under stress like pregnancy and poor nutrition, the immune system is weakened and cases can occur with heavy challenge.
FUTURE DEVELOPMENTS/ALTERNATIVES TO CURRENT VACCINES
The current blood vaccines for the control of tick-borne diseases in South Africa are all cumbersome to produce, and in addition they need to be stored and transported at very low temperatures. They also have to be thawed carefully to prevent damage of the live organisms. Research and development is urgently required to improve the usefulness of these vaccines.
New generation heartwater vaccines
South African researchers have developed an attenuated heartwater vaccine, which can be produced in cell culture and easily stored and transported (Zweygarth, 2006). In experimental trials it was shown that the vaccine can be given intramuscularly, produces only very mild signs of infection (no treatment needed), and protects animals (including small stock) from challenge from a wide range of isolates (Zweygarth et al., 2008). The vaccine is, however, still in the production development stage, but could be a major advance in animal health technology once it is registered.
American research groups have developed a recombinant vaccine which may offer multi-strain coverage of heartwater organisms in the field. The vaccine is still under development.
Combination of babesiosis and anaplasmosis vaccines
Vaccine producers in Australia have succeeded in producing a combined B. bovis, B. bigemina and Anaplasma vaccine. Just one vaccination is therefore required. The importation of the vaccine could help South African farmers with the control of tickborne diseases, but veterinary authorities have vehemently opposed its importation.
SPECIES APPROACH TO TICK CONTROL
TICK CONTROL STRATEGIES IN CATTLE
Apart from the various methods of tick control available in cattle – there are several strategies or approaches which relate, inter alia, to the type of cattle involved, the level of control desired, the frequency of treatment, and the prevention of resistance.
Levels of tick control
Intensive control
Historically, intensive tick control was applied by commercial farmers in the eastern, north-eastern and south-eastern areas of South Africa, where disease-transmitting ticks are most prevalent. Intensive treatment was applied in the form of weekly dipping in the summer months (as frequently as every four to five days during the East Coast fever control programmes), and at longer intervals (every second week) in winter to protect exotic cattle breeds against tick worry, loss of production, and tick-borne diseases.
For many years intensive control was even applied in areas where indigenous or crossbred animals grazed on communal pastures – for example in previous homelands like Transkei and KwaZulu. However, although intensive treatment is an effective tick control measure, and in many cases is able to break the transmission of an important disease such as East Coast fever, this control method has some attendant problems.
Firstly, acaricides are expensive and need equipment, applicators or labour to deliver the product. In addition, regular use results in the inevitable emergence of resistance to acaricides. An additional disadvantage of intensive tick control in Africa is that it renders treated animals susceptible to tickborne diseases. This is because immunity to tick-borne diseases is reliant on ongoing exposure to infected ticks or regular continued vaccination. Less intensive strategies have in many instances been adopted, as outlined below.
Strategic/less intensive control
This control measure is suitable – especially for Bos indicus and B. indicus/B. taurus crosses. They can be treated at intervals to reduce tick damage, but at the same time allow sufficient tick numbers to immunise the cattle. While this approach is economical and desirable, it is difficult to achieve disease control – in particular with cerebral babesiosis (B. bovis) – because the parasite is not prevalent enough in the vector tick to enable the establishment of enzootic stability (a stable disease situation) and in areas where there are various tick species transmitting more than one tick-borne disease. Vaccines which control tick-borne diseases have to be used in conjunction with strategic treatment to ensure sufficient levels of immunity.
Minimal/threshold tick control
This method is mainly applicable to B. indicus breeds or crosses under conditions where enzootic disease stability has been established and maintained. Minimal tick control can be practised in order to limit massive tick challenge and tick damage – including treating the predilection sites of ticks with long mouthparts, in order to prevent tissue damage.
Integrated control
There is a global tendency to move to the philosophy of integrated control for parasites (Pegram et al., 2015). This approach entails reducing the number of chemical applications while using other non-chemical control methods. The concept of integrated tick control as proposed by Pegram, includes using tick-resistant livestock breeds (including cross-bred dairy animals which are less productive but more tick resistant), and in some countries using tick vaccines. Vaccines against ticks have been developed in Australia and South America to control blue ticks – the group in which acaricide resistance usually develops. These vaccines have been successful for the control of R. (B.) microplus in reducing the number of acaricide applications in South America and Australia. The development of a specific vaccine against local R. (B.) decoloratus would be a significant advance for tick control in South Africa.
Control strategies for different types of farming
Intensive farming (dairy and feedlots)
In dairy cattle and feedlots, tick worry and tick-borne diseases will adversely affect milk production and rapid weight gain. The disease and tick control strategies will depend on whether cattle are exposed to ticks through natural grazing or not (as in where they are kept under zero-grazing conditions), but it should be noted that anaplasmosis can also be transmitted by biting flies. Feedlot cattle are by definition kept on zero-grazing, so tick control is generally unnecessary except during processing when the animals are introduced into the feedlot. Anaplasmosis vaccination and various fly control methods are practised if fly transmission is a problem.
Most large dairies keep in-milk animals on zero-grazing where they are not exposed to tick challenge. However, heifers and dry cows are on semi-intensive or extensive grazing, and thus will be exposed to some tick challenge.
Dairy cattle in multi-host tick areas usually receive intensive tick control using pyrethroids as the dip of choice since they are effective for flies as well as ticks. They are delivered as pour-ons or sometimes by spray race. Vaccination for the tick-borne diseases may be necessary. In areas where multi-host ticks are absent, tick control with pyrethroids is the treatment of choice in order to also control flies. Anaplasmosis and babesiosis vaccination may be necessary.
Organophosphates, and IGRs (fluazuron) are not suitable for use in lactating animals. The situation with fipronil for use in dairy cattle remains unclear.
Extensive farming
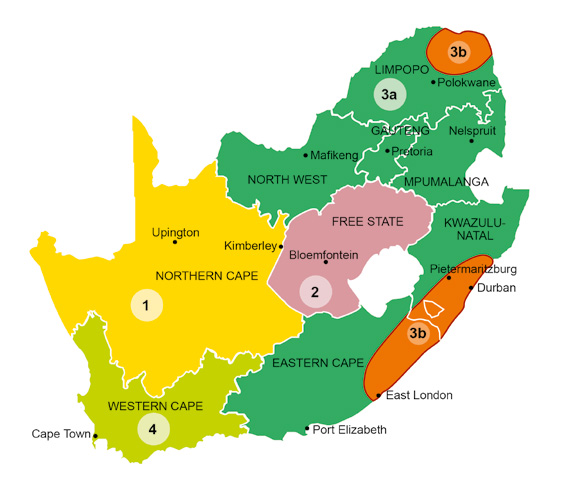
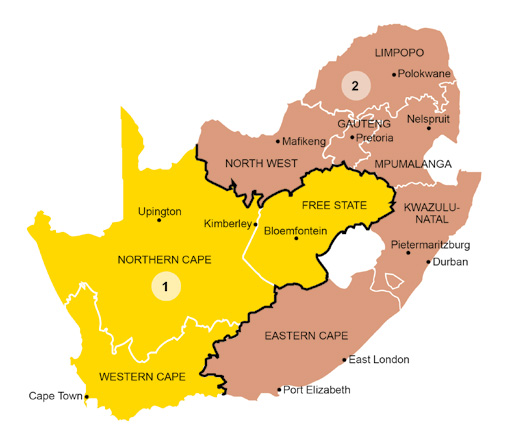
Depending on the geographic location of the cattle farm, various ticks and therefore tick-borne diseases will occur. The map of South Africa has been subdivided into areas according to the predominant tick and tick-borne disease problems, and specific recommendations are made for each area.
Area 1: Arid western areas of South Africa, e.g. the Northern Cape
Ticks: The vector of heartwater A. hebraeum, and also blue ticks are absent from this area. Hyalomma species are present here. Sand tampans can be the cause of severe irritation and the death of heavily infested calves.
Diseases/damage: Hyalomma ticks (and biting flies) are vectors of anaplasmosis. They cause severe wounding of teats, vulvas, and the prepuce. Hyalomma ticks are also responsible for sweating sickness – a toxicosis seen in calves.
Recommended strategy: For low-level tick control (Hyalomma ticks) use spot treatment with tick grease, hand spraying, tactical application of pour-ons, or acaricidecontaining paint balls which have recently been registered for use on game.
Tampan control: The most severe tampan challenge is seen in camps and kraals where livestock are kept for management purposes. These animals can be treated with acaricidal pour-ons on the legs and underline to prevent tampan attacks. The use of bantams and CO2 traps has also been recommended for reducing tampan numbers in the area. CO2 traps can be made by burying a plastic container in the ground (rim level with the soil), and then placing a tin can containing dry ice inside the plastic container. The tampans are attracted to the release of CO2 and will fall into the plastic container. They must then be destroyed using an acaricide.
Vaccination: An anaplasmosis vaccination may be required as even low-intensity tick control could prevent the development of an enzootically stable situation with respect to anaplasmosis.
Area 2: Central areas of South Africa, e.g. the Free State
Ticks: R. (B.) decoloratus, Hyalomma rufipes, Ixodes rubicundus
Diseases: African babesiosis (B. bigemina), anaplasmosis, occasional tick paralysis caused by I. rubicundus (for control, see under small stock).
Control strategy: In the eastern part of the area, strategic control can be done and enzootic stability may be achieved with respect to the diseases. In the western parts of this area where tick populations fluctuate according to annual variation in rainfall (drought), vaccination may be required. MLs can be used effectively in spring in both parts of the area, to dramatically lower the late season peaks of the blue tick. Hyalomma ticks may need to be treated using hand dressing or acaricide-containing paint balls, when their numbers are sufficient to cause damage.
Vaccination: This may need to be done for both diseases if stability cannot be achieved.
Area 3: Northern, north-eastern, eastern and south-eastern areas of South Africa, e.g. North West, Limpopo, Gauteng, KwaZuluNatal and the Eastern Cape
This is the most complex and difficult area for tick control. It includes one-host, two-host and three-host ticks. In addition to the African blue tick, the Asiatic blue tick – R. (B.) microplus – occurs in focal areas (Area 3b), and are said to be increasing their range. Also present is the multi-host tick A. hebraeum which transmits heartwater and because of its long mouthparts and clumping behaviour, causes severe teat, udder, vulva and prepuce damage.
Regular dipping is carried out during the summer months, contributing to the development of resistant blue ticks as they have a shorter life cycle and spend more time on cattle than other species. In addition, cattle in the corridor area adjacent to the Hluhluwe/Umfolozi parks are at risk of exposure to Theileria-infected R. appendiculatus, which occurs in buffalo in these parks. The presence of tsetse fly and nagana in these areas further complicates control measure decisions.
• Area 3a
Ticks: A. hebraeum and R. (B.) decoloratus are widespread. Brown ear ticks (R. appendiculatus) can cause severe infestations. R. (B.) microplus ticks may spread from the areas currently identified in Area 3b, and so landowners should be vigilant. Only tick taxonomists are able to tell the two Boophilus spp. apart; this is done by examining the dentition on the hypostome (more details can be seen in Monographs 1 and 2 of this series).
Diseases: A. hebraeum is the vector of heartwater; R. (B.) decoloratus transmits B. bigemina or African babesiosis and anaplasmosis. Corridor disease may be transmitted by R. appendiculatus from buffalo to cattle in areas adjacent to the Hluhluwe/Umfolozi corridor.
Control strategy: In exotic breeds, regular tick control with acaricides suitable for multihost ticks in summer, will make disease stability difficult to achieve. Less intensive tick control may be possible in Zebu and/or indigenous cattle breeds. The frequency of tick control measures can be reduced in winter, depending on tick prevalence. Additionally, the use of MLs and IGRs in spring, to reduce the initial blue tick population peak, may be useful for limiting the later summer peaks to more manageable levels. Farmers should ensure that good management practices are used in applying acaricides in order to prevent and slow the development of resistance. It is essential that plunge dips are built correctly to ensure head-wetting for the control of – especially brown ear ticks – which transmit corridor disease in areas adjacent to the KwaZulu-Natal parks.
Vaccination: Vaccination against heartwater, anaplasmosis and B. bigemina will be necessary because of the frequency of tick control.
• Area 3b
The ticks and diseases are as for Area 3a, but the presence of R. (B.) microplus which transmits cerebral or Asiatic babesiosis complicates disease control considerably – given that both babesiosis vaccines must be used. Vaccination is advised, as the tick is often not prevalent enough for disease stability to be achieved.
Area 4: The Western Cape
Ticks: R. (B.) decoloratus, Hyalomma spp.
Diseases: Babesiosis (B. bigemina) and anaplasmosis.
Control strategy: Strategic blue tick control with macrocyclic lactones will be sufficient to reduce tick worry and irritation. Hyalomma ticks must be controlled if they are in sufficient numbers to cause tissue damage.
Disease control: Strategic tick control can achieve enzootic stability for both diseases – in which case use of the vaccine is not necessary.
Summary of tick control in cattle
Methods of acaricide application in cattle
1. Plunge dipping
Plunge dipping of cattle is the method of choice for tick control, because if the dip tank is well designed it ensures good wetting with the acaricide. Briefly it entails the animal jumping into the dip tank and swimming through the dip wash. Poor design results in poor wetting, so dip tanks must be planned and built correctly to ensure good tick control. For guidelines on planning, design, construction and management of dip tanks for cattle, see Afrivet’s Practical guide to dip tank construction and management (Oberem and Oberem, 2014)
Replenishment methods for dip tanks
Dip wash is removed during the process of dipping (roughly three litres per animal). This requires that the dip tank be topped up with water and dip concentrate. However, because there is generally a concreted dripoff crush immediately after the dip tank, water runs back into the dip tank. With most dips, active ingredient is trapped in the hair of the animals dipped, and so the run/drip-off is often more diluted than in the dip tank. The topping up of the chemical is referred to as replenishment. There are various methods of replenishment, depending on the product used:
- Conventional replenishment: Under this system the dip is only fresh-filled every two to five years, or after roughly 20 000 animals have been dipped. The dip wash is then drained, the tank cleaned, and it is then filled with the required amount of water. The dip concentrate is then added according to the volume of water and the recommended fresh-fill rate. Once in use, the dip wash must be replenished to compensate for the stripping (removal of active ingredient by the animals) at a rate specified by the manufacturer. Generally, the replenishment rate is higher than the fresh-fill rate. However, as most pyrethroids do not strip much, their replenishment rate is the same as their fresh-fill rate – e.g. a Decatix dip wash analysis should be done every six to 12 months to ensure the correct concentration of dip wash. The best time to sample dips is at the beginning of the tick season (August) or after long periods of disuse. The types of dips used in conventional dipping are organophosphates, pyrethroids and formamidines, or combinations thereof.
- Head count: This system was developed and patented by Coopers so that operators can manage dip tanks without having to do complicated mathematical calculations. Since each animal removes roughly the same amount of active ingredient, the replenishment rate is based on the number of animals that have been through the dip.
- Total replacement (TR): This method is used only for non-lime-stabilised amitraz dips. The dip is fresh-filled with amitraz at each dipping, because the amitraz breaks down with time. This method is only economical if large numbers (>600) are dipped on a single day, but this depends on tank size.
See link to Afrivet Plan A for price comparison of the various methods of replenishment – www.afrivetplana.co.za
Stripping of dip wash
Management of dip tanks
A dip tank can only be a useful tool for tick control if it is correctly managed. Apart from the maintenance of the correct concentration of the chemical (see later) there are various other aspects which must be considered. These include safety and animal welfare, planning to avoid the heat of the day, preparation of the dip tank and dip wash, the dipping procedure, replenishment, and after-dipping management.
Discarding dip
It is essential that when dip tanks are emptied the dip is discarded safely and without causing a potential hazard to humans, animals or wildlife. This is done by running used dip wash out on an allocated area of ground, where the active ingredient will be broken down by the combined action of sunlight and soil bacteria. Choose a level piece of ground near the dip tank and plough contour furrows to promote containment and to prevent run-off. Allow 0,5 ha for 15 000 ℓ (600 m² for a sheep dip tank and 100 m² for a normal spray race). The disposal site must be at least 100 m from boreholes, streams, houses/huts and public thoroughfares. The area must be fenced off and should be marked “DANGER SABS WW5” and “KEEP AWAY” in all local languages. There must be no runoff from this area into local watercourses.
Dip wash sampling and testing
Once or twice a year, dip tank samples should be sent to a laboratory to ensure that the dip wash is still up to strength. Ask the animal health company that supplies your dip to provide the necessary contain- ers and forms for dip sampling, and also to refer you to a suitable laboratory for testing the sample.
The dip wash sample must be taken carefully, since it represents a very small sample of a very large volume – meaning there is a high measure of sampling error. Here are some guidelines:
- Use a clean 250 ml glass bottle with a 15 mm diameter neck, to take the sample.
- Attach the bottle to the bottom of a 1,5 m pole. A pole with a tin or bottle big enough to hold the sample bottle can be made specifically for this purpose.
- The sample should be taken from about 0,5 m below the dip wash surface.
- The best time to take the sample is after replenishment, when the tank has been well stirred (usually by the first 50 cattle passing through it).
- After retrieving the dip-filled sample bottle, the following preservatives must be added to the sample dip wash:
- For pyrethroid and organophosphate dips, add 5 ml of 10% formalin.
- For amitraz dips like Triatix add one teaspoon of lime (the powder supplied with the lime-stabilised dip).
- Seal and dry the bottle.
- Store in a refrigerator.
- Complete and submit the “Dip Wash Analysis Request Form”, as provided by the company supplying the dip.
- Follow the procedures and complete the form fully and correctly, as the quality of the advice you get back will be dependent on this.
- Send to the laboratory as soon as possible.
2. Spray race
A spray race is simply a crush over which an arrangement of metal piping is erected. The dip is circulated from a sump through the piping and is forced through small noz- zles, thus creating a fine spray. Animals are wetted while passing through the race.
The dip wash drains back into the sump and is recirculated through the system. A double footbath at the entrance of the spray race ensures the cleaning of the hooves and helps prevent fouling of the spray and blocking of the nozzles. The choice of loca- tion for a spray race is based on the same principles as for a dip tank, but the wind direction must also be taken into account. The spray race must be diagonal to the pre- vailing winds in order to prevent dispersion and contamination.
Spray race management
The system must be able to maintain 300 ℓ to 500 ℓ of dip wash in circulation and to provide 1,5 ℓ to 2,5 ℓ per animal. The dip wash must be replenished according to directions on the label of the product used. Fresh dip must be used at each dipping. The use of a foot valve to trigger the spray when animals are in the race, will reduce wastage of dip wash. Constant supervision to monitor the wetting of the animals is essential, and also for maintaining a calm and unhurried progress through the spray race.
3. Mechanical/hand spraying
This method is used for very small numbers of animals, where limited holding facilities are available. This can be done using a motorised pump with a lance and nozzles, a high-pressure manual pump, a bucket pump, or a knapsack sprayer.
This is the most difficult method to get right. Compared with plunge dips and spray races, hand spraying is very time consuming and wasteful as it allows a lot of run-off which cannot be reused. Animals need to be adequately restrained, and they must be thoroughly wetted with at least 5 ℓ to 10 ℓ per animal (12 ℓ is optimal).
For effective application, have the animals in single file in a crush pen. Start spraying from the underline to the topline and tail to head, against the lie of the hair. Pay attention to hooves, tail brush, under the tail, the area between the front and hind legs, the ears, poll, udder, sheath, scrotum and muzzle.
4. Pour-ons
Pour-on formulations are acaricides dis- solved in solvents which aid the spread of the product over the skin. Their application is labour intensive and requires restraint of the animals in order to be done effectively.
A calibrated applicator must be used to ensure delivery of the correct volume. For whole body treatment, the application of the pouron needs to start between the horns – making sure, however, that it does not run down into the eyes and end on the tail root. Pour-on remedies can also be used for patch treatment on tick predilection sites (see below).
Operators must use suitable gloves to avoid contact with the pour-on liquid. Home-made pour-ons contribute to the more rapid development of resistance (selection of resistant ticks), and should be avoided at all costs.
5. Patch/spot treatment
Patch/spot treatment is done as an adjunct to dipping or spraying applied to difficult- to-wet predilection sites. It can also be used as a low-level tick control method. Patch treatment can be done by applying tick oil or grease, dip solution, pour-on formulations, or acaricide-containing paint balls.
6. Injectables (macrocyclic lactones)
MLs are used for control of one-host ticks. They are used particularly for strategic control or when there is resistance to acaricides. This method is often unsuitable for controlling multi-host ticks, as usually the immatures are found on hosts, e.g. scrub hare, guinea fowl and/or reptiles, which cannot be treated.
Figure 27 illustrates how the strategic seasonal treatment of blue ticks (one-host ticks), with a few follow-up treatments later in the year, can keep blue tick numbers low. Initially, two treatments with ivermectin are given at 35-day intervals (September and October – 14 to 21 days after the first good rains). Treatment with ivermectin in November and December is followed by three times weekly treatments with pour- ons or dipping from January to April (Van Rensburg, personal communication).
This method reduces tick infestation sig- nificantly. This eliminates the direct primary and secondary effects, but allows sufficient tick numbers to ensure that an enzootically stable disease situation is maintained for B. bigemina.
7. Bolus administration
Fipronil is used as a bolus, which is dosed to the animals using a specially designed applicator. This should come on the mar- ket soon, as its registration is currently in progress. The bolus is given every six weeks during the tick season or when marker ani- mals or 20% of the animals show more than 60 adult ticks counted in the early morning. Fipronil may not be suitable for dairy cat- tle, depending on the results of residue trials currently being done.
Table 4. A summary of application methods in cattle.
| Application method | Suitability | Advantages | Disadvantages | Cost |
|---|---|---|---|---|
| 1. Plunge dipping | Large numbers of animals for tick and fly control | • Good wetting • Moderate management level | • Sufficient water is required • Labour intensive | High initial outlay – unit cost low with use |
| 2. Spray race | Ideal for tick and fly control in dairies | • Flexibility if remedies have to be changed | • Less effective wetting than plunge dipping • High level of management is needed | Moderately expensive with initial capital outlay |
| 3. Mechani- cal /hand spraying | Used for small numbers of cattle for tick and fly control | • Flexible and convenient for low-intensity dipping and spot treatment | • Less effective wetting • Wastage of active and water | Low capital outlay initially, but very expensive per animal |
| 4. Pour-ons | Tick and fly control; ideal for single treatments for lice/mites | • Convenient | • Irritation with some pyrethroids • High dose of active ingredient per animal accelerates resistance | >Low capital outlay initially, but expensive per animal |
| 5. Patch/spot treatment | Additional to other methods | N/a | N/a | N/a |
| 6. Injectables (MLs) | Control of blue ticks | • Deworming effect for roundworms • No tick resistance yet | • Narrow tick spectrum • Long meat withdrawal • Most are unsuitable for dairy animals | Expensive per treatment |
| 7. Bolus administration (fipronil) | All tick spp. | • Broad spectrum for tick and fly control • Six-week action | • Possible milk residues • Administration requires a good technique | Inexpensive |
TICK CONTROL STRATEGIES IN SMALL STOCK
Control strategies for different areas in South Africa
Area 1: Arid western areas of South Africa, e.g. Northern Cape
The dry western areas of the country are free of heartwater because of the absence of A. hebraeum ticks, but other ticks, e.g. red-legged ticks, foot ticks, brown ticks and I.rubicundus, can cause sporadic problems. I.rubicundus causes Karoo paralysis and becomes active when the first frosts occur, usually in May.
For prevention on affected farms, the bellybathing of sheep and goats every four to six weeks using deltamethrin dip, is effective. Alternately, the use of pour-on formulations applied to the legs, axilla and groin have good residual action. Choose the correct formulation according to the type of animal to be treated. For example, Clout® is an oil-based formulation suitable for non-wool sheep and goats, while the water-based Wipeout® can be used on wool sheep and angora goats. Foot ticks and brown ticks can be treated with pour-on formulations, belly- or footbaths containing suitable acaricides. Sand tampan challenge is most severe in camps or kraals where small stock is kept for management purposes. To prevent infestation, treat animals with the application of acaricide pour-ons, on both the legs and underline.
Area 2: Eastern and south-eastern areas of South Africa, e.g. KwaZulu- Natal and the Eastern Cape
The vector of heartwater is present in the wetter eastern areas of the country, so the disease is prevalent in sheep and goats. The use of heartwater vaccination in small stock has been investigated for many years, but has been found to be generally ineffective in pro- viding immunity against the disease, is labour intensive, and has several safety concerns.
Regular tick control using acaricides in a bellybath is therefore recommended for controlling the vector of heartwater, A. he- braeum, in sheep and goats including an- goras (Taylor, personal communication).
This method will also control brown ticks (Rhipicephalus spp.) such as R. simus which are found between the claws of the hooves.
A new attenuated vaccine for heartwa- ter which will become available in future has been shown to be effective and safe for use in small stock, and will make a major contribution to the control of heartwater in endemic areas.
Table 5. A summary of the most important ticks and the diseases/damage caused in small stock.
| Common name | Scientific name | Disease/damage |
|---|---|---|
| The South African bont tick | Amblyomma hebraeum | • Heartwater in sheep and goats • Wounds and abscesses with secondary screwworm strike |
| The red-legged tick | Rhipicephalus evertsi evertsi | • Spring lamb paralysis |
| The glossy brown tick (foot ticks) | Rhipicephalus simus | • Lameness • Foot abscesses |
| The brown paralysis tick | Rhipicephalus warburtoni | • Angora goat paralysis |
| The bont-legged tick | Hyalomma spp. | • Lameness • Abscesses |
| The Karoo paralysis tick | Ixodes rubicundus | • Tick paralysis |
| The sand tampan | Ornithodoros savignyi | • Anaemia • Toxicosis |
Methods of acaricide application in small stock
The ticks that cause problems in small stock are mostly multi-host ticks – and the most suitable acaricides for control are amitraz, organophosphates and pyrethroids in dip form. The pyrethroids are also available in pour-on/spot-on formulations for local application.
1. Plunge dipping
Although plunge dipping of small stock can be used for tick control, it is mainly done for blow fly and scab control. If plunge dip- ping is used, the tanks must be correctly constructed and managed (Oberem and Oberem, 2014).
2. Belly- and footbathing
This form of dipping is useful for the con- trol of the ticks infesting the lower regions of the body of sheep and goats. As the system uses minimal dip wash, it is an ef- ficient and cost-effective way of managing foot disease caused by tick bites in sheep and goats – for the control of bont tick (A. hebraeum), Karoo paralysis tick (I. rubi- cundus) and the red-legged tick which causes spring lamb paralysis (R. evertsi evertsi).
The infrastructure required consists of a rectangular dip tank 3 m to 5 m long, which is built out of concrete and bricks. Moulded fibre-glass units are also available commer- cially from time to time. It should have a replenishment tank on site, to allow constant replenishment of the dip wash during treatment, in order to maintain the dip-wash level of 30 cm for bellybathing and 15 cm for footbathing. Collecting kraals and a drain- ing race are required. The replenishment tank should have sufficient capacity to deliv- er 0,3 ℓ per animal when footbathing, and
0,5 ℓ to 1,5 ℓ per animal when bellybath- ing – depending on wool length. The object of this form of treatment is to saturate the feet, lower limbs and belly with dip wash. A certain amount of splashing occurs, and as sheep exit the dipping bath the crutch area can become wetted, which can also help control blowfly strike in that region.
For tick control only, amidines or pyre- throids can be used (amitraz or deltamethrin). If tick and blowfly control are required, an organophosphate such as diazinon or chlofen- vinphos is preferred. Dips should be diluted, as per the manufacturers’ instructions.
3. Pour-on/patch treatment
Pour-on remedies for tick control are avail- able for sheep and goats, in particular for ticks which attach at certain predilection sites, e.g. the feet (Rhipicephalus spp.) and the underside (I. rubicundus). As with cattle, the basic principle for application is to ad- minister the correct volume at the correct sites. There are different formulations for wooled (water based) and non-wooled (oil based) sheep and goats, to prevent the con- tamination of fleece/mohair.
Hand dressing with tick grease, oil or pour-ons, is useful for treating ticks with specific predilection sites such as the feet.
Table 6. A summary of application methods in sheep and goats.
| Method | Suitability | Advantages | Disadvantages | Cost |
|---|---|---|---|---|
| 1. Plunge dipping | Only for short hair/wool or for non-wooled stock | One after-shear dip prevents scab, other mites and lice (permanent parasites) | Not suitable for stock with long fleece | Low |
| 2a. Belly- bathing | Karoo paralysis ticks, bont ticks, foot or brown ticks, Rhipicephalus ticks | Economical and suitable for sheep and goats | Not suitable for scab control | Low |
| 2b. Foot- bathing | Foot ticks (see tick table) | Product applied to specific area | N/a | Cheaper than plunge dipping and bellybathing |
| 3. Pour-on/ patch treatment | Paralysis tick, Rhipicephalus spp. on feet | No dip tank needed. Can target predilection sites of ticks | Choose correct formulation for type of animal (wooled/non-wooled). Labour intensive | Moderate |
TICK CONTROL STRATEGIES IN GAME ANIMALS
General principles
The vast majority of the 37 tick species in South Africa feed on wild animals, includ- ing large and small mammals, birds and reptiles. It is therefore not surprising that wildlife species have developed strategies for defence against the ticks to which they have been historically exposed. Some of the de- fence strategies against ticks are behavioural, such as grooming, mud or sand bathing, and migrating to clean areas. Some wild game species have innate immunity and can acquire an immune response, as seen in some livestock, so preventing tick attach- ment. A study of buffalo herds in the Kru- ger National Park showed that animals with innate immunity and younger animals, had lower tick burdens, while those that were older, pregnant or had poor body condition were more likely to have high tick burdens (Anderson et al., 2013).
Game farming creates a situation in which wild species are restricted by fences, albeit sometimes on large properties. Large herbivores like buffalo, rhino and giraffe are considered to be “multipliers” of certain tick species as they are favoured by adult ticks, while small antelope, small mammals and birds are favoured by the immature stages. According to researchers, tick populations can become problematic on small or over- stocked properties, which cause large tick populations to flourish.
Although most healthy large antelope and equids can harbour large burdens of ticks without them causing problems, indi- vidual animals that have injuries or which are debilitated by disease or starvation will become heavily infested. However, even in apparently healthy animals heavy burdens of brown ear ticks, for example, can cause tissue damage on teats and ears in species like eland and even kudu – especially under more intensive farming conditions. Heavy burdens of ticks may cause tick worry and potentially reduce growth and reproductive performance.
The calves of some antelope species like sa- ble, roan and wildebeest become infected with a Theileria species of sable (previously called T. hippotragi – nomenclature not finalised), which can cause fatal infections in animals mainly under three months of age. Animals like kudu, duiker, giraffe and rhino, are sometimes infected with their “own” Theile- ria spp. which are not yet properly identified and named.
Control methods in game animals
The ideal situation on game farms is to have a “hands-off policy” allowing animals to adapt naturally to their environment. However, sometimes intervention is necessary when heavy tick burdens are causing tissue damage or other losses. Intervention is usually done by darting and treating in- dividuals with myiasis. Animals in bomas can be treated by using hand sprays, acari- cidal tick balls (acaricide-containing paint balls), or the application of pour-on for- mulations.
Treating free-range animals of vari- ous species to reduce the level of ticks on a farm, is more problematic. Most of the systems developed to deliver acaricides to large game animals, e.g. the Duncan ap- plicator and Oom Gielie’s dipbak, do not apply the correct dose per weight, nor do they regulate the frequency of treatment. Systems like the Tick off apparatus which weighs the animal and squirts the calcu- lated amount, are an improvement on the earlier designs. Nevertheless, they cannot ensure the correct frequency of applica- tion, since animals enter or do not enter the treatment race voluntarily.
Some ranchers have built large, circular footbaths, about 1,5 m to 2 m wide, and which are filled with a registered raw cot- ton and pyrethroid dip around a drinking/ feeding trough. This is very effective for reducing the cases of foot rot and lameness in oryx kept in the Waterberg.
An alternative method is to treat free- ranging, heavily infested individuals, by using pyrethroid-containing tick balls. The tick balls, which have only recently been registered for use on antelope, are deliv- ered by using an applicator or marker (an ordinary paintball gun) to deliver the pyre- throid formulation. This has been shown to be effective, and the dose (number of balls used per animal) can be adjusted per live weight. There are three different pyrethroid- containing tick balls registered: 2,5% for use on smaller antelope up to 62,5 kg live weight; 5% for use on antelope up to 125 kg live weight; and 10% for animals weighing
250 kg. Bigger animals must be treated with more than one ball – e.g. a 350 kg animal can be “shot” using 1 x 5% and 1 x 10% balls. The tick balls are now available from co-operatives or via an Afrivet agent.
This control method is well suited to ensure:
- Only the correct animals are treated (those in need).
- The correct dose is applied.
- That the dose frequency can be well managed.
- The welfare of the animals is taken into account.
Note that feed formulations containing aloe extracts are touted as being effective for tick control in game species. This prod- uct has however been shown by evidence- based trials to be ineffective (Fourie et al., 2005; Spickett et al., 2007). There is also no evidence-based work to show that diatoma- ceous earth-containing products can effect tick control on any species.
Heartwater susceptibility of wild ruminants
Based on observations made on a wide range of wild ruminants in South Africa, a general pattern of heartwater susceptibility can be distinguished, and is divided into four categories:
- Immune/resistant: These species have an innate or genetic resistance to heart- water. They include all species which evolved in bushveld, such as buffalo, kudu, and nyala.
- Susceptible but can acquire immunity: These species are susceptible, but can acquire heartwater immunity when ex- posed naturally, and if vaccinated will often not need “blocking” to treat the vaccine response. They include impala, gemsbok and eland.
- Highly susceptible: These are mainly the species which evolved in grassland, where they had no historic exposure to or evolution with heartwater. They in- clude springbok, bontebok and black wildebeest. This group can be immun- ised by controlled exposure and treat- ment, which is currently the Ball 3 strain blood vaccine that must be administered intravenously. Vaccination can give al- most 100% immunity, but must be blocked with antibiotic treatment.
- Exotic ruminants: Exotic ruminants are totally susceptible to heartwater.
In susceptible species which acquire im- munity, offspring will be protected by ma- ternal immunity, and will then be able to develop active immunity when exposed to infection by ticks. The immunity acquired by susceptible species is thought to be ge- netic, and may involve immunity to ticks as well as the Ehrlichia organism.
Introduction of wild animals into heartwater areas
Susceptible species: Animals which fall under categories three and four, such as springbok,
must either be vaccinated before introduc- tion, which is a cumbersome exercise (see under vaccination), or exposed to low levels of challenge by placing them in camps with mainly short grass and little bush. The latter method, however, makes it difficult to ensure immunity and may incur losses. It should be noted that animals sold as heartwater- resistant springbok may well originate from endemic areas, but may never have been ex- posed because of the absence of ticks in a par- ticular micro-habitat such as farm or camp systems. This can lead to losses, so buyers should be aware of this potential problem.
Immune species: Immune species can be in- troduced into heartwater areas from non- endemic areas without losses.
Vaccination
Heartwater vaccination must be done six weeks before introducing animals into an endemic area. Using the currently available blood vaccine, it is a rather cumbersome process involving intravenous adminis- tration of infected blood which is stored frozen in liquid nitrogen and transported on dry ice. The vaccine contains live viru- lent heartwater organisms, so animals may develop a reaction indicated by the devel- opment of a fever, which must then be blocked by treatment with tetracyclines. This is a very risky and stressful operation, when dealing with wild antelope. However, it has been done with some success by vari- ous game ranchers, as detailed below (Ma- lan, personal communication; Barnard, personal communication):
- Animals held in a camp are hand caught and vaccinated intravenously, as directed on the pamphlet.
- From day eight after vaccination the animals are caught and their tempera- tures are taken until a significant rise in temperature is seen (39,5 °C – 40 °C). This usually occurs by day 12 after vac- cination.
- When the fever reaction develops, the animals are treated with tetracyclines.
It should be noted that because field strains of heartwater vary in pathogenicity, the vaccine does not protect equally well in all animals. Other preventive measures such as reducing stress on capture and adequate tick-control measures should be used.
Immunity certification
Although laboratory tests are available for heartwater antibody determination by the evaluation of enzyme-linked immunosorbent assay/immunofluorescent antibody (ELISA/IFA), the presence of antibody against the organism merely indicates that animals have been exposed, and does not guarantee immunity (Latif, personal communication). Game ranchers have reported that animals which have tested positive with IFA by the Onderstepoort Veterinary Insti- tute, have developed clinical cases of heart- water on introduction into endemic areas (Malan, personal communication). Further research is needed to develop a reliable di- agnostic test. A potential buyer should therefore request written proof that animals purchased originate from an endemic heart- water area, but must be aware that not all these animals are necessarily immune.
Future prospects
A new attenuated vaccine which does not require blocking treatment with antibiotics, has been recently developed at the Onder- stepoort Veterinary Institute and has been shown to be protective in livestock. It has not yet been registered for use or tested in highly susceptible game species such as springbok.
Theilerial susceptibility in wild ruminants
The Theileria species has been reported to occur in roan and sable antelope. A Thei- leria species of sable (previously called T. hippotragi) has been recorded as the cause of deaths in roan antelope, in which almost 100% of the year’s calf crop died. Similarly, it has been identified as the cause of mortalities in young sable calves up to the age of two months. Sometimes clinical cases have been seen in wildebeest calves.
The red-legged tick, R. evertsi evertsi – with the most widespread distribution of all ticks in South Africa – has been shown to be the main vector of the Theileria spp. The immature stages of this two-host tick feed in the ears of antelope and equids such as zebra. The ticks become infected by the an- telope; they have been shown to be benign carriers of Theileria spp. in reedbuck, blue wildebeest, blesbuck, nyala and bushbuck. Roan and sable calves do not have any im- munity to this parasite, and will develop a clinical, usually fatal, disease if they receive a high enough dose of parasites (Steyl, 2015). Adults are less susceptible – possibly because the infective dose is less per body size. There is some indication that theilerial susceptibil- ity in roan and sable has a genetic cause.
Two strategies can be used or combined to prevent the exposure of roan and sable to ticks:
Calving in clean camps: Pregnant cows can be moved in December to camps which have been “tick-cleaned”, in other words, camps which have not be stocked with antelope for 18 months, as the chances of exposure of calves to infected ticks are less under these conditions (Steyl, 2015). The cows then calve in February and remain in the clean camps until April when the young are past the age of susceptibility.
Treatment with tick pour-ons: Roan and sable antelope calves in small breeding camps are usually caught and tagged soon after birth. They can therefore be treated by applying a registered pour-on to the underline instead of the topline, which makes it less likely that the cow will reject her treated calf. Repeat treatments may be needed until the calves are over two months of age (Davey, personal communication).
REFERENCES
- Abdul-Amir, IM and Gray, JS, 1987. Resistance of sheep to laboratory infestations of the tick Ixodes ricinus. Research in Veterinary Science, 43: 266–267.
- Anderson, K, Ezenwa, VO and Jolles, AE, 2013. Tick infestation patterns in free-ranging African buffalo (Syncerus caffer): effects of host innate immunity and niche segregation among tick species. International Journal for Parasitology: Parasites and Wildlife, 2: 1–9.
- Barnard, H, 2016. Personal communication.
- Brown, NC, Chadwick, PR and Wickham, JC, 1967. The role of synergists in the formulation of insecticides. International Pest Control, 10-13. As cited in: Piperonyl butoxide – the insecticide synergist. Edited by Glynne Jones, Academic Press London, 1998.
- Davey, E, 2016. Personal communication.
- Fletcher, WA, [undated]. A guide to practical cattle tick control in Southern Africa. Intervet, South Africa.
- Fourie, JJ, Fourie, LJ and Horak, IG, 2005. Efficacy of orally administered powdered aloe juice (Aloe ferox) against ticks on cattle and ticks and fleas on dogs. Journal of the South African Veterinary Association, Dec, 76(4): 193–196.
- Guerrero, FD, Lovis, L and Martins, JR, 2012. Acaricide resistance mechanisms in Rhipicephalus (Boophilus) microplus. Revista Brasileira de Parasitologia Veterinaria, online version. http//dx.doi.org/10.1590/ ISSN 1984-2961. 12012000100002. Accessed: 2014-04-10.
- Guerrero, FD, Adalberto, A, De Leon, P, Rodriguez-Vivas, RI, Jonsson, N, Miller, RJ and Andreotti, R, 2013. Chapter 13: Acaricide research and development, resistance and resistance monitoring, 353–381. In: Biolog y of ticks, 2. Eds: Soneshine, D and Roe, RM. Oxford University Press, USA.
- Ikonomopoulos, M and King, G, 2013. National born killers: spider venom peptides and their potential for managing arthropod pests. Outlooks on Pest Management. ResearchGate.
- Katsuda, Y, 2012. Progress and future of pyrethroids. In: Pyrethroids: from Chrysanthemum to modern industrial insecticide. Eds: Matsuo, N and Mori, T. Springer-Verlag, Berlin, Heidelberg.
- Latif, AA, 2017. Personal communication.
- Malan, J, 2016. Personal communication.
- Mwangi, EN and Kaaya, GP, 1997. Prospects of using tick parasitoids (Insecta) for tick management. International Journal of Acarology, 23: 215–219.
- Mwangi, EN, Kaaya, GP, Nyandat, E, Munyinyi, D and Kimodo, MG, 1995. Repellence of the tick Rhipicephalus appendiculatus by the grass Melinis minultiflora. Tropical Animal Health and Production, 27(4): 211–216.
- Ntondini, Z, 2009. The extent of acaricide resistance in the eastern region of the Eastern Cape Province. MSc thesis, University of Pretoria.
- Oberem, PT, 1984. The immunological basis of host resistance to ticks: a review. Journal of the South African Veterinary Association, 55 (4): 215–217.
- Oberem, PT and Oberem, P, 2014. Practical guide to dip tank construction and management. Afrivet Training Services.
- Pegram, RG, Tatchell, RJ, De Castro, JJ, Chizyuka, HGB, Creek, MJ, Mc Cosker, PJ, Moran, MC and Nigarura, G, 2015. Tick control: new concepts. FAO publication online. Accessed 2015-01-15.
- Sparks, TC and Byford, RL, 1988. Pyrethroid-synergist mixtures: toxicity, resistance, and field efficacy towards pyrethroid-resistant horn flies (Diptera: Muscidae). Journal of Economic Entomolog y, 81(6): 1567–1574.
- Spickett, AM, Van der Merwe, D and Matthee, O, 2007. The effect of orally administered Aloe marlothii leaves on Boophilus decoloratus tick burdens on cattle. Experimental and Applied Acarolog y, 41(1): 139–146.
- Stafford, KC and Allan, SA, 2010. Field applications of entomopathogenic fungi Beauvaria bassiana and Metarhizium anisopliae F52 (Hypocreales: Clavicipitaceae) for the control of Ixodes scapularis (Acari: Ixodidae). Journal of Medical Entomolog y, 47(6): 1107–1115.
- Steyl, J, 2016. Personal communication. Taylor, R, 2016. Personal communication.
- Van Rensburg, J, 2016. Personal communication.
- Willadsen, P, 2004. Anti-tick vaccines. Parasitolog y, 129: S367–387.
- Yunker, CE, 1996. Heartwater in sheep and goats: a review. Onderstepoort Journal of
- Veterinary Research, 63: 159–170.
- Zweygarth, E, 2006. In vitro cultivation of Erlichia ruminantium and a development of a culture derived attenuated vaccine. PhD thesis, University of Utrecht.
- Zweygarth, E, Josemans, AI and Steyn, HC, 2008. Experimental use of the attenuated
- Erlichia ruminantium (Welgevonden) vaccine in Merino sheep and Angora goats. Vaccine, 2(S6): G34–39.