- Plant Poisonings and Mycotoxicoses of Livestock in Southern Africa, 2nd Edition
- Cardiovascular system
Cardiovascular system
| Acute poisoning by cardenolide-containing plants | Nerium oleander | Strophanthus spp. | Acokanthera spp. | Gomphocarpus spp. | Cryptostegia grandiflora | Adenium multiflorum |
| Acute poisoning by non-cumulative, bufadienolide-containing plants | Moraea spp. | Moraea pallida | Moraea miniata | Moraea polystachya | Moraea bipartita | Moraea carsonii |
| Chronic poisoning with bufadienolides that have a cumulative, neurotoxic effect | Tylecodon wallichii | Tylecodon ventricosus | Tylecodon grandiflorus | Cotyledon orbiculata | Kalanchoe lanceolata |
This content is distributed under the following licence: Attribution-NonCommercial CC BY-NC  View Creative Commons Licence details here
View Creative Commons Licence details here

Introduction
Plant-induced cardiotoxicoses are among the best-known and most important plant poisonings in southern Africa. Some of the earliest toxicological research in South Africa, for instance, the investigation of krimpsiekte and tulp poisoning, was done on plants that induce cardiotoxicoses and has continued without interruption until today. In this chapter the progress that has been made in research on these poisonings will be discussed.
Cardiac Glycosides
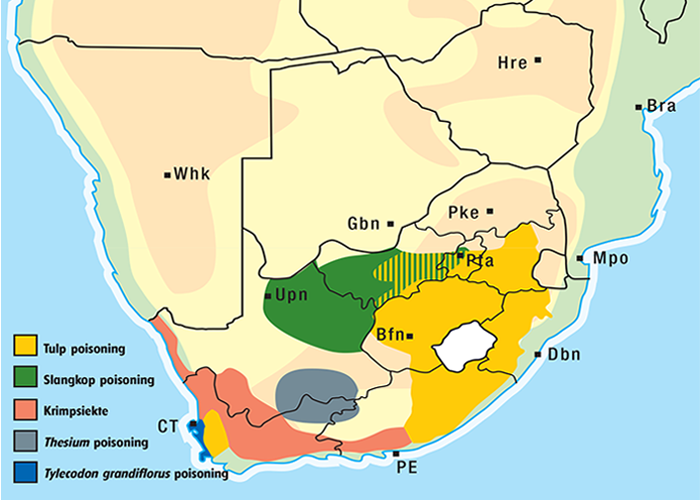
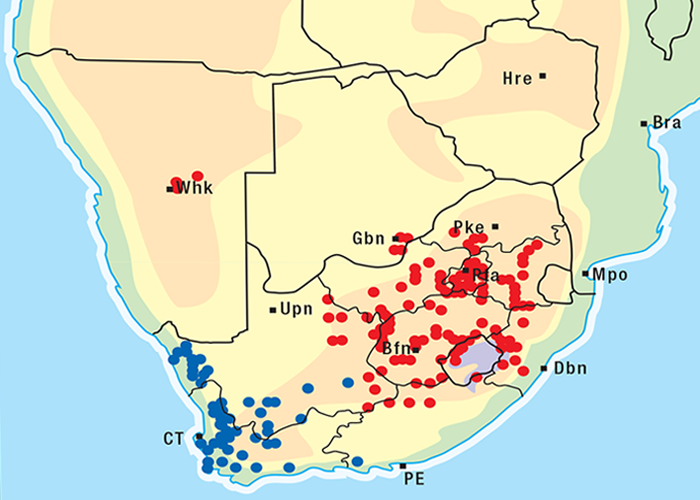
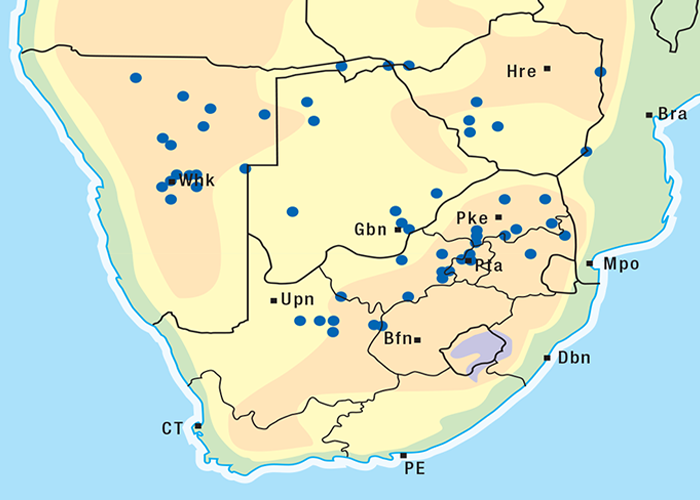
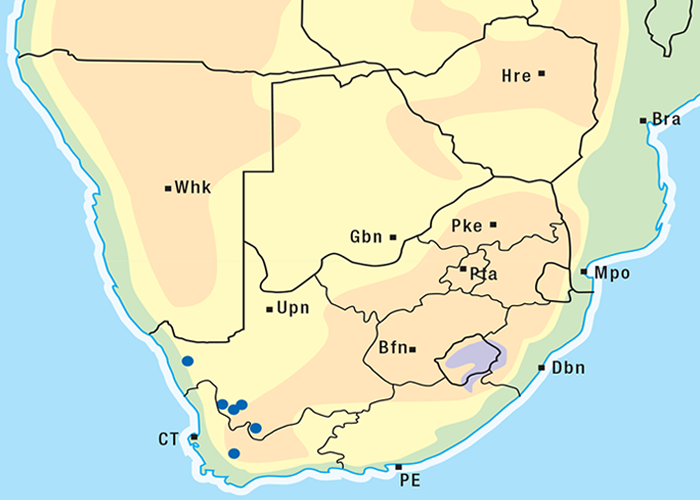
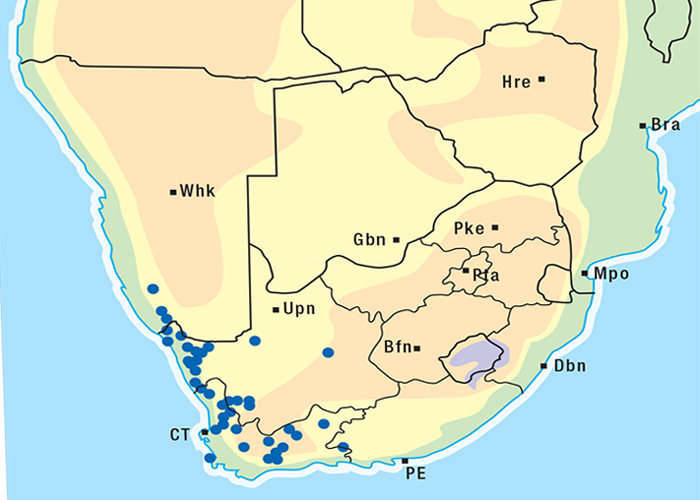
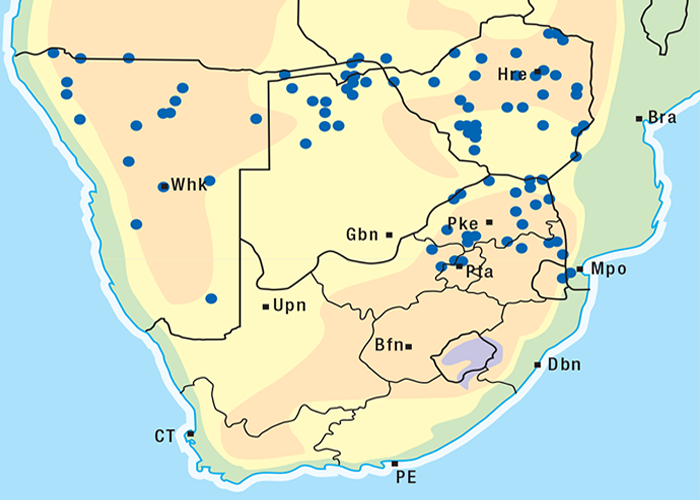
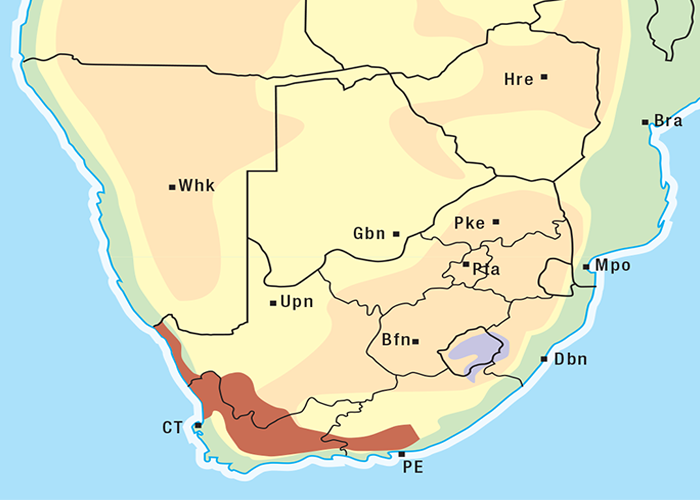
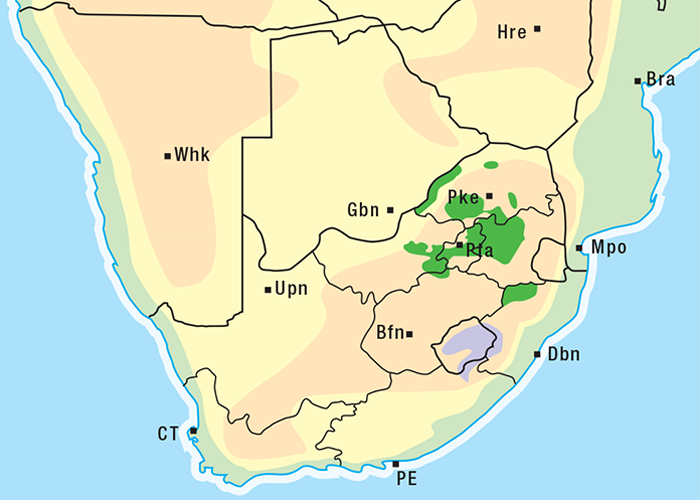
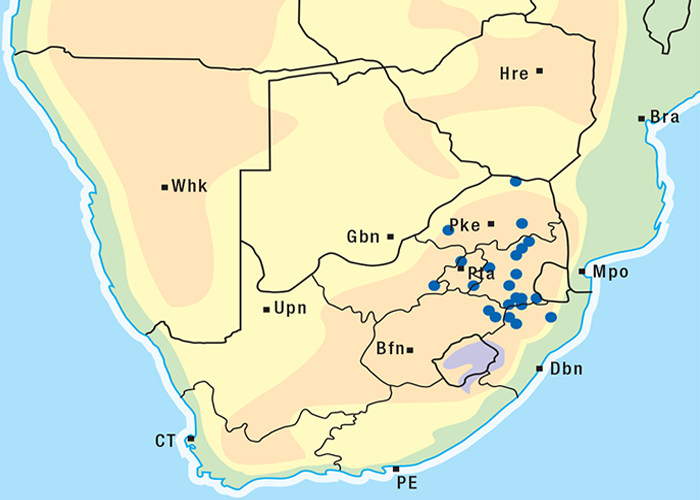
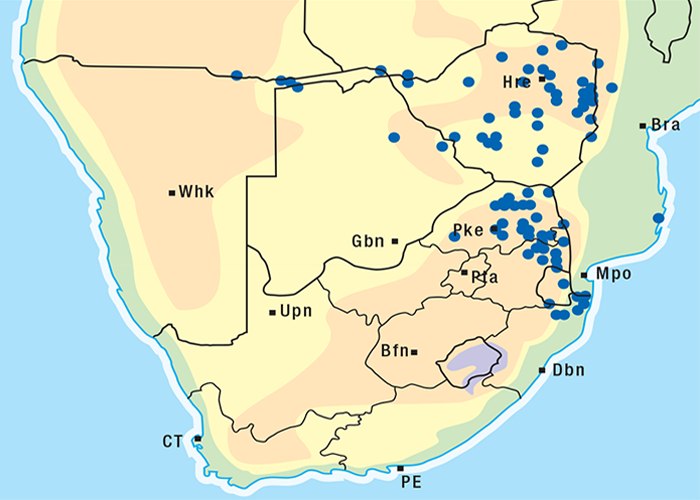
Cardiac glycoside-containing plants have a worldwide distribution, yet poisoning of stock with these plants is of significance only in southern Africa126 and to a much lesser extent in Australia.217 Collectively they are the most important plant poisoning of southern Africa. In South Africa, c.33% of mortalities in cattle from plant poisonings and mycotoxicoses and c.10% of those in small stock are attributed to these plants.126 Heavy stock losses from phytogenous cardiac glycoside poisoning are recorded almost every year in most provinces of this country (Figure 1).
Cardiac glycosides are found in a wide variety of indigenous and imported plants (a number of which cause incidental intoxications), but only poisoning by tulp (Iridaceae), slangkop (Hyacinthaceae) and the succulent plakkies (Crassulaceae) is of economic significance.
Chemical structure
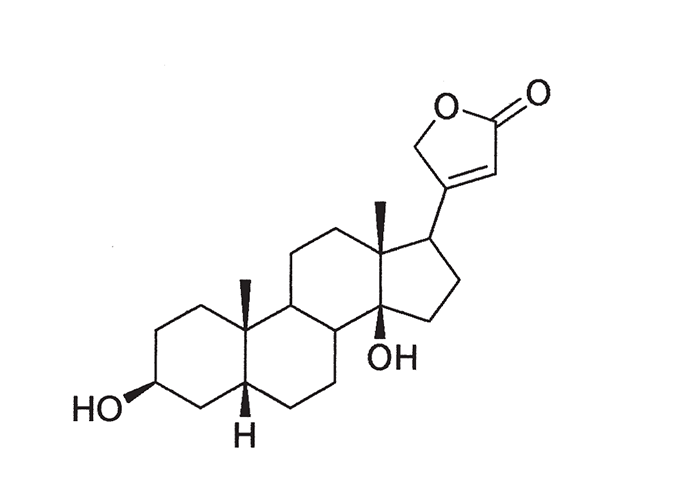
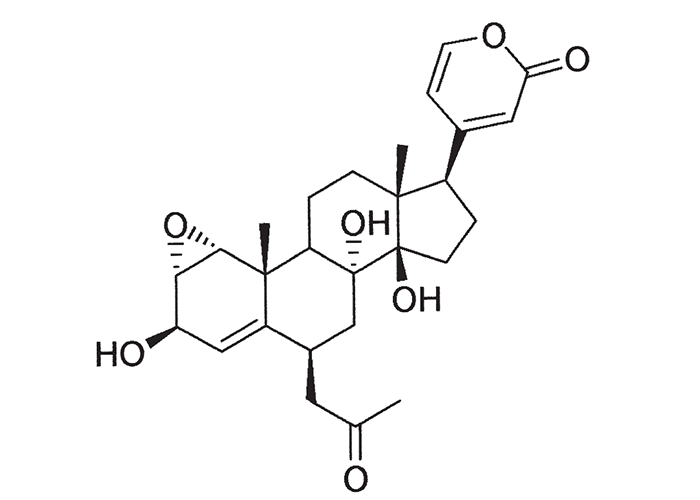
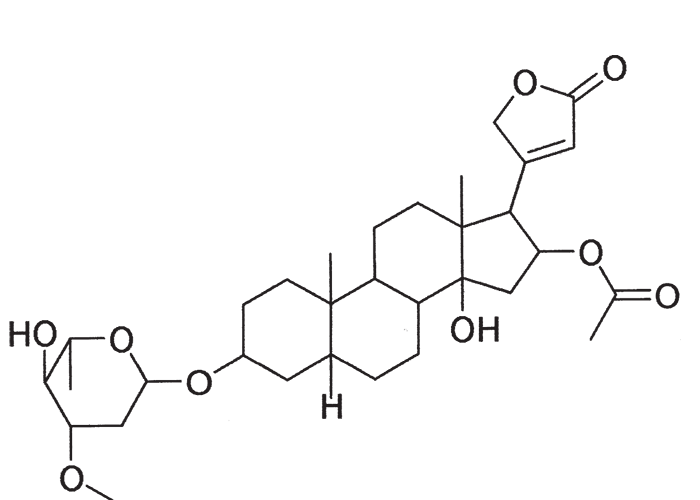
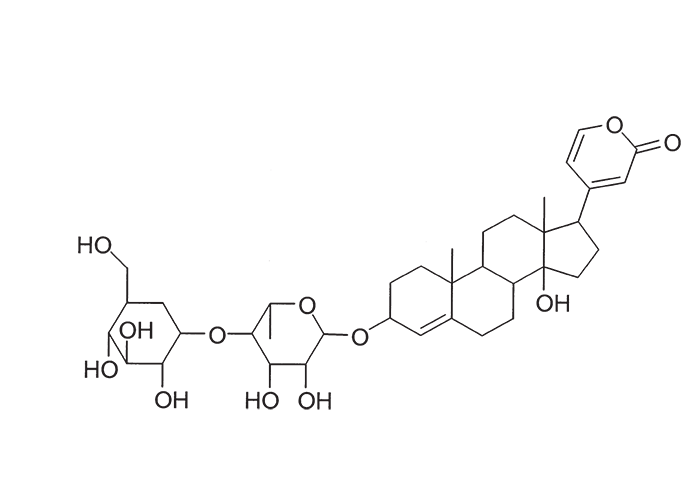
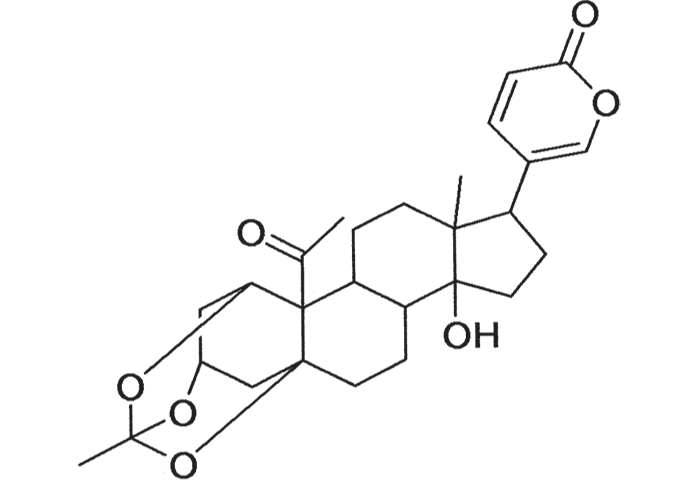
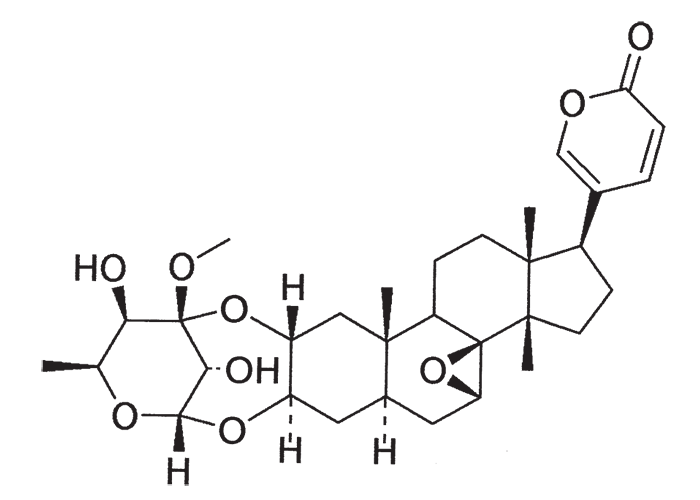
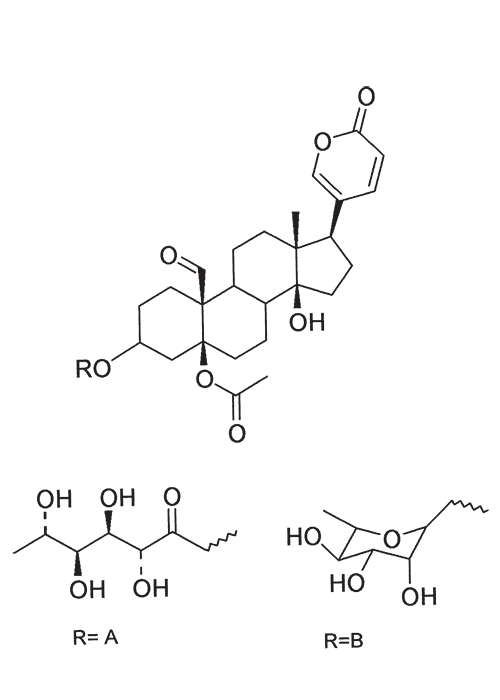
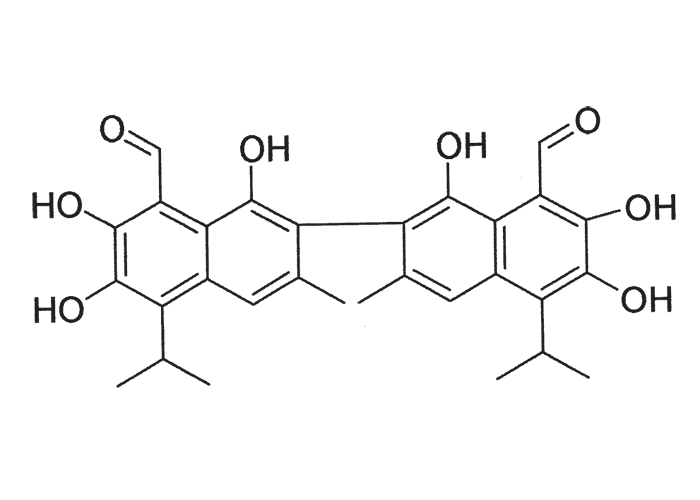
A cardiac glycoside molecule consists of a steroidal aglycone or genin coupled to a sugar-portion. The pharmacological action of the molecule is vested in the aglycone; the sugar portion having little effect apart perhaps from influencing lipid solubility. The aglycones can be chemically divided into cardenolides and bufadienolides. Cardenolides have an unsaturated, five-membered (butenolide) lactone ring on C-17 of the steroid molecule (Figure 2), while bufadienolides have a doubly unsaturated, six-membered (pentadienolide) lactone ring in that position (Figure 3).163 The naturally occurring cardenolides in South Africa are apparently non-cumulative, while bufadienolides can be either cumulative or non-cumulative in effect, depending on the plant species and cardiac glycoside involved.
Clinical signs
The animals most commonly poisoned by cardiac glycosides are cattle, sheep, goats and donkeys, in that order. Horses are seldom affected because they are fastidious grazers and hence avoid cardiac glycoside-containing plants.168, 231, 234 However, recently a number of horses were reported to have died of colic after eating tulp-infested hay. Cardiac glycosides could be demonstrated in the hay and organs of the affected horses using a fluorescent polarisation immunoassay vide infra (R.A. Schultz and N. Fourie, OVI, personal communication, 1996).
Intoxicated animals are generally apathetic and tend to stand with their heads down and stomachs tucked in, sometimes grinding their teeth or making groaning noises. Other common signs include tachycardia, cardiac arrhythmia, atony of the rumen, bloat, diarrhoea and weakness of the hindquarters. Acutely affected animals may die suddenly of cardiac arrest, often after even the slightest exertion.106, 160, 163, 231, 234
Cardiac glycosides basically affect four systems, namely the cardiovascular, gastrointestinal, nervous and respiratory systems. At therapeutic doses these substances have a positive inotropic effect which results in stronger myocardial contraction, and a negative chronotropic effect manifested as bradycardia. Higher doses exert a negative dromotropic effect, causing atrio-ventricular (AV) dissociation and consequent first, second and third degree heart block. During intoxication, initial bradycardia92, 163, 231, 232, 234 is followed by sinus tachycardia, interrupted by runs of ventricular tachycardia of AV nodal origin. Later, multifocal, ectopic, ventricular impulses are generated with increasing frequency, leading to severe arrhythmia, which often culminates in complete AV dissociation and fibrillation. Runs of normal sinus rhythm typically separate episodes of conduction disturbances.197, 215
The gastrointestinal signs are marked by colic, ruminal stasis, bloat, diarrhoea and dehydration. Since the bloat is of a gassy type, it can be relieved by trochar. Diarrhoea is an almost constant feature of poisoning by cardiac glycoside-containing plants. The faeces are liquid, often slimy and sometimes blood stained.67, 92, 106, 160, 161, 168, 231, 232, 234, 244, 246 Notable exceptions where constipation is evident, are poisoning by Homeria glauca (now classified as Moraea pallida) in the provinces of Mpumalanga, Free State and KwaZulu-Natal163 and M. miniata in the Western Cape (D.J. Schneider, Regional Veterinary Laboratory, Stellenbosch, personal communication, 1984).
Nervous involvement occasionally takes the form of hypersensitivity, a stiff-legged or high-stepping gait, muscular tremors or spasms, and incoordination. The most usual and outstanding nervous sign is posterior paresis (Figure 4). Affected animals typically walk with swaying hindquarters160, 232, 234, 241 and can become terminally paralysed. In the case of chronic poisoning by plants that contain cumulative bufadienolides, the paretic component of the symptom complex is greatly accentuated, while the respiratory, cardiac and gastrointestinal signs are much suppressed or absent. The clinical features of the krimpsiekte syndrome will be discussed in the section dealing with chronic intoxication by bufadienolides.
Varying degrees of polypnoea and dyspnoea are usually evident in animals poisoned by cardiac glycoside-containing plants. In acutely intoxicated experimental cases severe respiratory crises or bouts of apnoea can occur concurrently with or even preceding electrocardiographic changes. The respiratory distress is thought to be neuromuscular in origin.163
Pathological changes
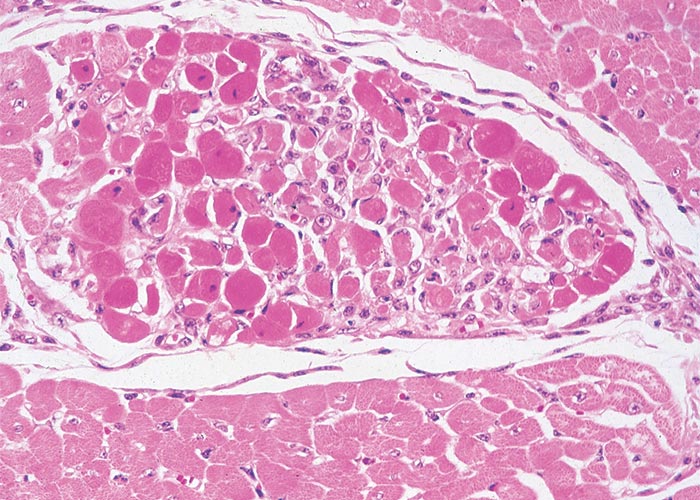
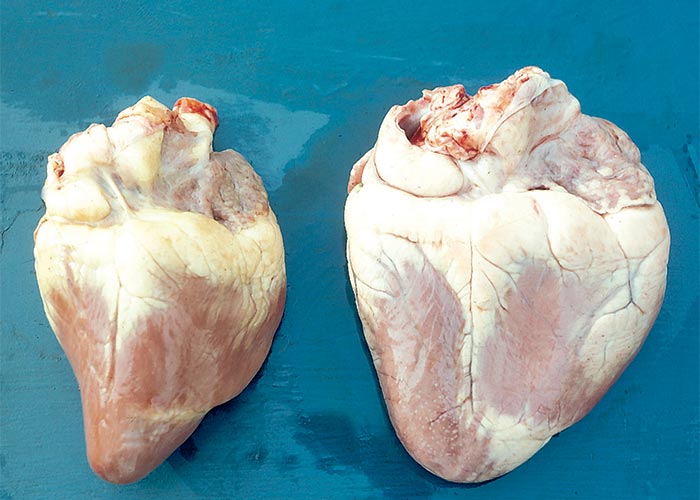
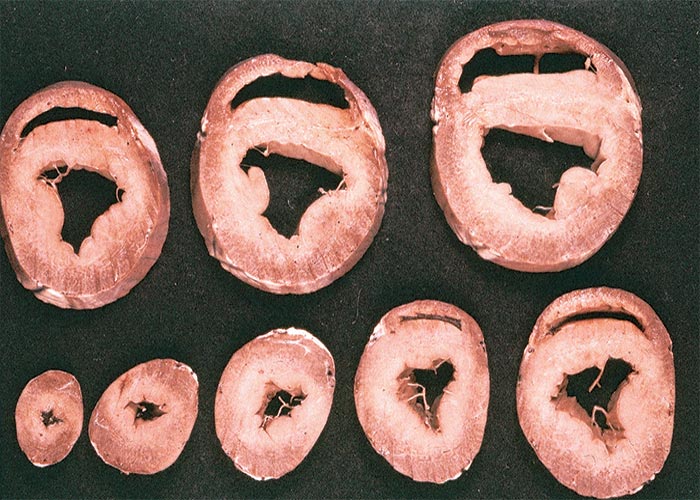
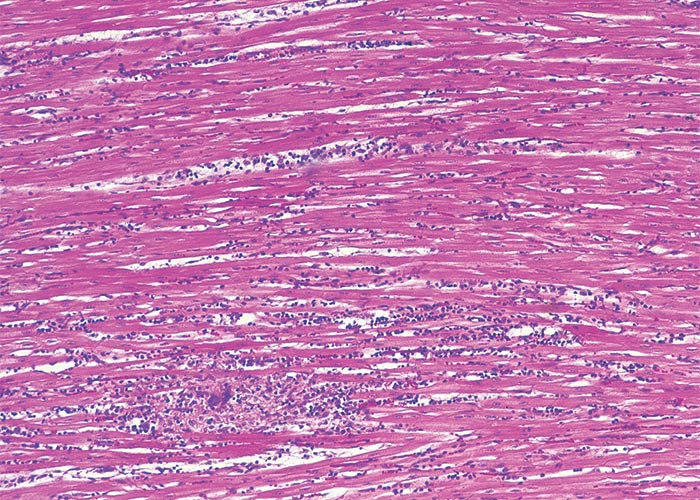
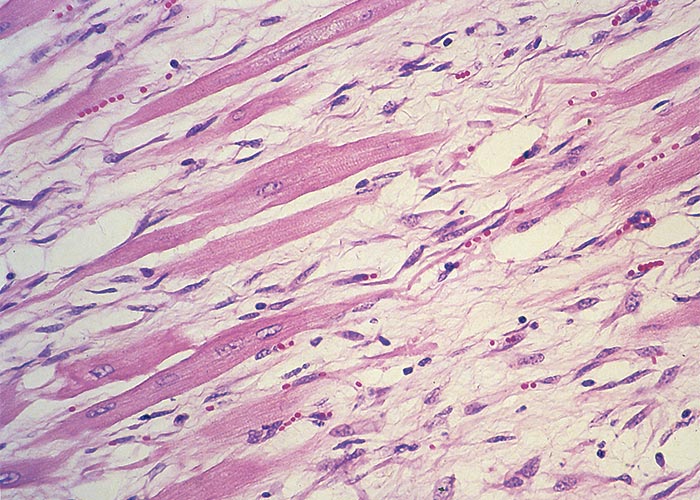
Extracardiac manifestations of heart dysfunction may be grossly evident in the carcases of animals that have died of cardiac glycoside poisoning. These lesions include congestion, oedema of the lungs and/or thoracic, pericardial or peritoneal effusion. Pronounced subcutaneous haemorrhages are evident. Sometimes the mucosa of the gastrointestinal tract may be hyperaemic, the perineum may be soiled by diarrhoea, and signs of dehydration may be present. Scattered foci of myocardial necrosis, sometimes with mononuclear inflammatory cell infiltrates, and evidence of early fibroplasia (Figure 5) have been reported in cases that live for a few days.172, 173
Acute poisoning by cardenolide-containing plantsPlants of this group are extremely toxic but of little veterinary consequence, as they are seldom eaten by stock.
Nerium oleander L. (Apocynaceae)
Oleander, selonsroos
Oleanders are large shrubs (c.5 m in height) originally from Europe and Asia, which are widely grown as ornamental plants in South Africa. The stiff, 100–150 mm long, dark green, lanceolate leaves are arranged in whorls of three and have a conspicuous, closely parallel venation. The flowers are borne terminally on the branches (Figure 6). The flowers of Nerium oleander are dark red, pink, or white and fragrant. All parts of the plant contain a watery latex. In the cooler, moist parts of the country, N. oleander has become a weed.262
The dearth of information on the toxicity of oleanders in South Africa reflect their unimportance as poisonous plants for stock. Various authors have referred to the fatal poisoning of soldiers in Corsica by meat roasted on skewers of N. oleander.44, 107 Hutcheon (1903) described the death of a Clydesdale stallion that nibbled on the plant107 and Henning (1932) quotes American reports that 15–30 g dry leaves were lethal to horses and cattle, while 1–5 g killed sheep.101 Since desiccation has little effect on toxicity,44 hay contaminated by garden clippings can be hazardous to stock.
A sheep at the Veterinary Research Institute, Onderstepoort, died 17 hours after being dosed with 0,25 g/kg fresh N. oleander plant; 0,5 g/kg was lethal in four hours, and 2–4 g/kg in two and a half hours (T.W. Naudé, VRI, Onderstepoort, unpublished data, 1963). Dogs were supposedly poisoned by water from a bird-bath into which oleander leaves had fallen.
Several cardenolides have been isolated from N. oleander, including oleandrin (Figure 7) and adigosid.102
At necropsy, the leathery leaf fragments of N. oleander, with lateral veins running closely parallel, may help to confirm the diagnosis.
Thevetia peruviana, an ornamental shrub of the same family, causes poisoning under similar circumstances. The plant is characterized by shiny, narrow, alternately arranged leaves and fragrant bright yellow flowers, the petals of which are twisted spirally to the right at the distal end (Figure 8). A cardenolide, thevetin,290 has been isolated from it.
Strophanthus spp. (Apocynaceae)
S. speciosus (Ward. & Harv.) Reber
S. petersianus Klotzsch
S. luteolus Codd
Poison rope, giftou
These are indigenous shrubs or woody vines60, 183, 190 found mostly in the wooded eastern parts of the country. The corolla lobes of some species are elongated or filiform (Figure 9), giving the flowers a distinctive spidery appearance.183 Apart from their use as arrow poisons136 Strophanthus spp. are not of veterinary importance. Glycosides such as strophanthin and ouabain (G-strophanthin)109, 136 are contained by them.290
Acokanthera spp. (Apocynaceae)
A. oppositifolia (Lam.) Codd
A. oblongifolia (Hochst.) Codd
Bushman’s poison bush, boesmangif, gifboom
Acokanthera oblongifolia is an evergreen shrub with broad, elliptical, shiny, dark-green, leathery leaves that end in sharp points. The white or pink-tinged, sweet-scented flowers are borne in dense clusters between the leaves. The dark-purple, plum-like fruits contain two seeds.46, 101, 190, 262
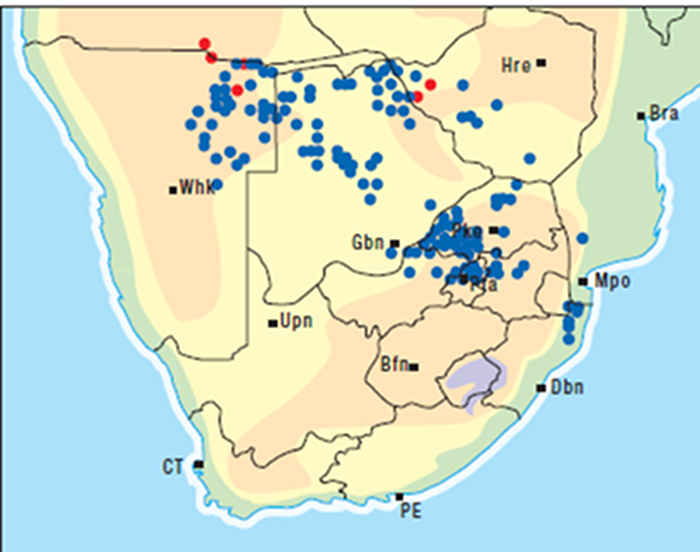
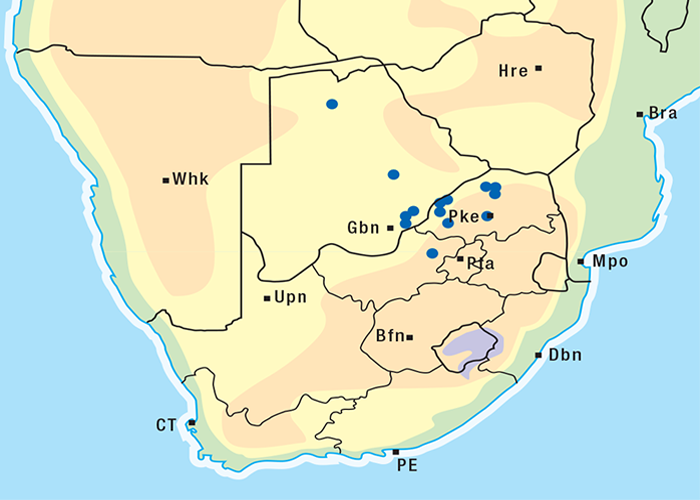
A. oppositifolia (= A. venenata) (Figure 10), a similar but smaller bush than A. oblongifolia, grows in the dry bushveld of the former Transvaal and along the east coast, where it extends inland along river beds. A. oblongifolia is confined to the eastern seaboard from Mozambique to the Eastern Cape Province (Figure 11).101, 190
Acokanthera poisoning is very rare. Although sheep, goats, donkeys and ostriches, and supposedly humans, are said to have been killed by the plants, cattle are reputed to be the species most commonly affected.43, 67, 69, 157 Cattle moved from grassland to coastal bushveld in winter when green herbage is scarce are thought to be particularly at risk.69 Fragments of the leathery leaves may be found in the rumens of poisoned animals.
Members of the genus contain well-known cardiac glycosides, such as ouabain, acokantherin14, 67, 211 and acovenoside A (= venenatin).211, 281 Acovenoside A is regarded as being cumulative58 but krimpsiekte-like symptoms have not been reported in poisoning with it. Like many other cardiac glycoside-containing plants, Acokanthera spp. have been used as arrow poisons.
Gomphocarpus spp. R.Br. (= Asclepias spp.) (Apocynaceae)
G. fruticosus
G. physocarpus
Milkweed, wild cotton, melkbos, kapokbos
G. fructicosus is a shrub, c.1,5 m in height. Like many members of the Apocynaceae family, this species contains a white latex. The opposite leaves are narrow, long and pointed, with revolute margins. The distinctive balloon-like fruits are covered with numerous hair-like processes (Figure 12). The fruit of the more easterly distributed G. physocarpus does not have a pear-shaped appearance. This endemic genus99 is common in most parts of southern Africa (Figure 13), especially on disturbed soil, trampled veld, and along roadsides and waterways.262
Despite containing cardenolides, such as gofrusid and frugosid (Figure 14),103 G. fruticosus is not of particular veterinary importance because it is unpalatable to stock. Recently it came to our attention that the plant is sometimes utilized by browsers and outbreaks of G. fructicosus poisoning in ostriches and goats have been diagnosed by fluorescent polarization immunoassay using digoxin-specific antibodies (R.A. Schultz, OVI, personal communication, 1995). Burtt-Davy (1912)45 fatally poisoned a beast with G. fruticosus and Steyn dosed G. physocarpus (E. Mey.) Schltr. to rabbits236 and sheep.239 A dose of 300 g G. physocarpus was lethal for a sheep.
Cryptostegia grandiflora (Roxb.) R. Br. (Apocynaceae)
Rubber vine
A suspected outbreak of poisoning in young elephants by an exotic garden plant (Figure 15) was reported from Namibia.36 The elephants had recently been translocated to another game reserve where two died within hours of one another. Examination of their tracks of the previous day revealed that they had entered the staff compound and browsed on a vine, C. grandiflora, fragments of the leaves of which were found in their stomachs. C. grandiflora is palatable to stock154 and contains glycosides closely related to oleandrin from Nerium oleander.4 The lesions in the affected animals were consistent with those of cardiac glycoside poisoning.
Adenium multiflorum Klotsch (Apocynaceae)
(= A. obesum (Forssk.) Roem. & Schult. var. multiflorum (Klotsch) Codd)
Impala lily
The beautiful impala lily of Mpumalanga (Figure 16) and A. boehmianum of Namibia are not of toxicological importance save for their use as arrow poisons. They contain cardenolides as their active principles.290
Acute poisoning by non-cumulative, bufadienolide- containing plants
Moraea spp. (Iridaceae)
Tulp
The name tulp (= tulip), a term used for Moraea spp. in English and Afrikaans vernacular, is misleading because this genus does not even belong to the same family as Dutch tulips (Tulipa gesneriana).262 This is the only genus of the Iridaceae to have been implicated in cardiac glycoside poisoning in stock. The known toxic tulp are M. pallida, M. miniata, M. flaccida, M. polystachya and M. bipartita; nevertheless, until proven otherwise, all species of tulp found in South Africa18 should be regarded as potentially toxic. M. flaccida and M. miniata have been introduced into Australia where they cause significant stock losses.217
The star-shaped flowers of yellow and red tulp have petals of about equal size, arranged on the same plane (Figure 17). The flowers of blue tulp, on the other hand, resemble irises: the large outer petals are partially reflexed, the three inner petals are smaller and often less highly coloured, and the petal-like branches of the style are larger (Figure 18).231, 234, 262 All species flower in spring except for M. polystachya, which blooms in autumn and winter.262 The plants most commonly confused with yellow tulp are the non-toxic, Hypoxis spp. (Hypoxidaceae). They have similar six-merous yellow star-shaped flowers, but their leaves are much larger and usually V-shaped in cross-section.276
Moraea pallida Bak.
(= Homeria pallida)
Yellow tulp, geeltulp
This is the tulp most often incriminated in the poisoning of stock; cattle being the species most commonly affected.126
The corm (c.30 mm in diameter) is white, covered by a dark-brown, fibrous tunic and buried deep (up to 200 mm) in the ground. A single leaf, seldom more, is formed at the base of the stem. The leaf emerges from the ground protected by a spear-like tip. In the young stage this tip serves to differentiate tulp from surrounding grass sprouts. The leaf, ensheathing the stem, is long (c.600 mm), narrow (c.20 mm wide), tough and strongly ribbed. Senescent leaves die off retrogressively from the tip. Numerous cormlets may form at the base of the stem. The stem (c.400 mm long) may be branched or unbranched and bears 6–10 flowers. The flowers are usually yellow, but can be apricot-coloured or orange-red (Figure 19). Club-shaped, three-celled capsules contain numerous angled seeds.190, 231, 262
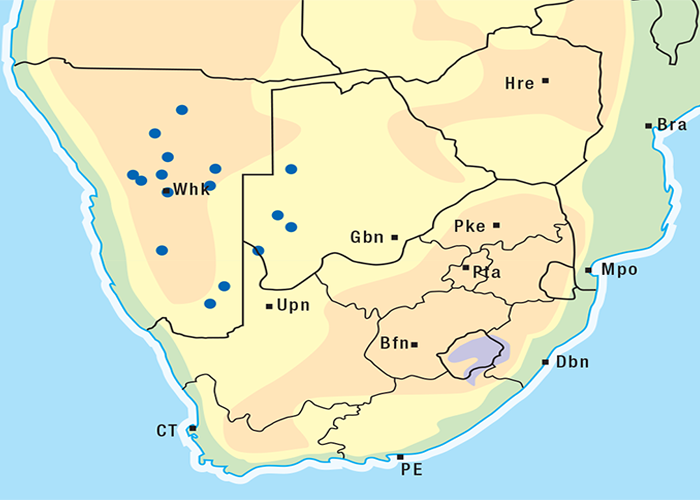
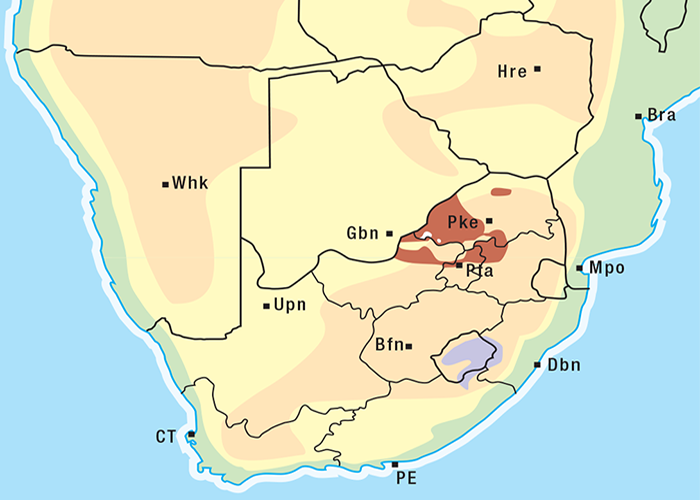
M. pallida is exceptionally invasive, rapidly colonising disturbed soil such as mealie lands. It is widely distributed occurring under a variety of climatic conditions, topographical situations and soil types in most provinces of South Africa and in Botswana (Figure 20).262
Moraea miniata (Andr.) Sweet
(= Homeria miniata)
Red tulp, rooitulp
The corms are much like those of Moraea pallida, and numerous cormlets may be formed on them. The leaves (one to four per plant) also resemble those of M. pallida, but may be slightly wider. Branched stems bear clusters of flowers that are usually pink, but can be yellow, orange or red. A star-shaped, yellow marking in the throat of the flower262 is a distinguishing characteristic of this species (Figure 21). M. miniata is found in the Western and Northern Cape provinces, where it grows under a variety of conditions.262
Moraea polystachya (Thunb.) Ker-Gawl.
Blue tulp, bloutulp
The corm of Moraea polystachya is similar to that of yellow tulp, in that it is covered by a network of dark, rigid fibres. Like other tulp species, contracting roots draw the corms deep into the soil (up to 300 mm). Usually, four narrow (5–10 mm), long (200–900 mm), strongly ribbed leaves are formed. The flowers are bright blue-mauve in colour with a typically iris-like structure (Figure 22). Each outer petal has a yellow spot at the base. The stem is unbranched. The fruit is a three-valved capsule (c.10 mm long) containing numerous black, angled seeds.101, 231, 262
M. polystachya is widely distributed, covering an area from the Kalahari to the southern and south eastern seaboard (Figure 23).101, 262 This highly invasive species also occurs in Botswana and Namibia.
Moraea bipartita L. Bol.
(= Moraea polyanthos L.f.)
Blue tulp, bloutulp
This species was previously known as Moraea polyanthos; however, the name M. polyanthos now applies only to a species formerly referred to as Homeria lilacina (Clare Reid, Botanical Research Institute, Pretoria, personal communication, 1986). Moraea bipartita (Figure 18) is found in the coastal belt of the Eastern and the Western Cape provinces (Figure 24).
Moraea carsonii Bak.
This is a delicate, iris-like plant with a small fibrous-coated corm. The slender stem bears one to three long, narrow leaves and about four clusters of flowers. The perianth is pale blue with yellow guide markings. Flowers tend to open only in the afternoon. M. carsonii occurs in grasslands in Zimbabwe and is particularly common in soil pockets on granite hills.218 It has recently been recorded in Botswana.
Toxicity and chemistry of tulp
Although tulp poisoning frequently occurs, comparatively little has been published on the toxicity of these plants234, 235, 266, 267, 271 since Borthwick and Dixon, cited by Hutcheon,106 first demonstrated tulp (Moraea polystachya and another, probably M. unguiculata) to be poisonous to stock. In a recent therapeutic trial with activated charcoal,114 sheep developed clinical signs four to six hours after being dosed with 1–2 g/kg dry, milled, flowering M. polystachya. The controls and unsuccessfully treated sheep died 9–70 h after the plant was dosed. Twelve steers given 1,25 g/kg of the same material died within 22–48 hours.115 Hungry cattle from a non-tulp area developed signs within 24 hours of being driven onto a Moraea pallida-infested pasture. Death generally intervenes 24–48 h after ingestion of tulp plants and non-fatally poisoned stock usually recover within three to four days.252
M. pallida is regarded as being one of the most toxic tulps234 and, therefore, most of the recent research has been done on this species. Like the other species it is toxic in both the dry and fresh states.204, 231, 234 This persistence of toxicity during desiccation accounts for the frequent poisoning of stabled animals with hay contaminated by tulp.168, 231 Toxicity is supposed to vary according to locality, climatic conditions and growth stage. When the seeds germinate in spring, grass-like leaves with characteristic arrow-like tips emerge, followed by the stems which bear fragile flowers. The seedlings, with their fine, thread-like leaves and small corms, are popularly believed to be more toxic than the older plants.234 Corms are purportedly less toxic than the other parts of the plant since they are uprooted and eaten with impunity by pigs234 and spring hares, but experimental evidence confirming this observation is lacking. Tulp poisoning is commonest in winter or spring before the onset of the rains, when sprouting Moraea spp. might be the only greenery on the barren veld.163, 246
One of the most important features of tulp poisoning is that newly introduced or hungry stock are most at risk, while animals that grow up on tulp-infested veld learn to avoid it (Figure 25).168 The fact that newly weaned calves with no previous exposure to M. pallida no longer became sick four days after being introduced onto heavily infested pastures indicates that this is the period required for adaptation to take place.
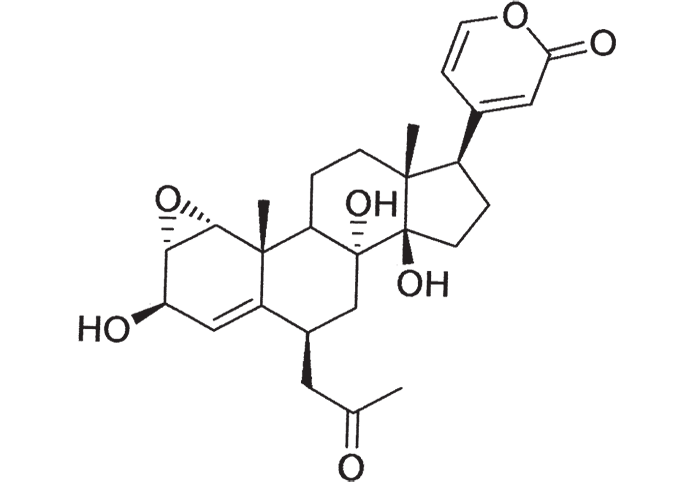
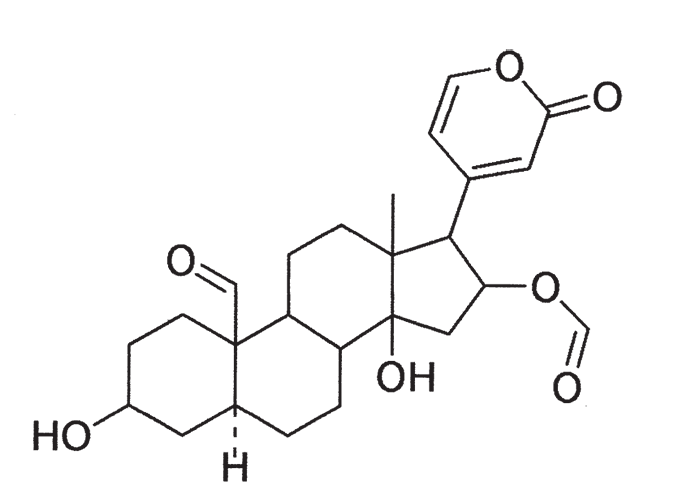
Early chemical investigations by MacKenzie143 in 1910 on a plant then known as Homeria collina (= M. collina), by Rindl204 in 1924 on H. pallida (= M. pallida), and by Dry72 in 1950 on Moraea polystachya, variously suggested that the toxic principles of tulp were glycosides or alkaloids with digitalis-like actions. The speculation on the nature of tulp poisons was laid to rest in 1966 by Naudé and Potgieter167 who, working on Moraea pallida (= Homeria glauca), confirmed the suspicions expressed by Gunn,93 and Gunn and Brown94 thirty-four years earlier, that tulp (H. ochroleuca and H. flaccida (= M. ochroleuca and M. flaccida respectively) contained cardio-active glycosides (bufadienolides). The main toxic principles of M. pallida, 1α, 2α-epoxyscillirosidin (Figure 26)78 and another 1α, 2α-epoxy-12ßhydroxyscillirosidin279 were isolated by mild techniques in which the various extraction steps were monitored by semi-quantitative toxicity determinations in guinea-pigs.167, 168 The subcutaneous LD50, of the main toxic principle for guinea-pigs was 0,194 mg/kg and for mice 3,6 mg/kg. In guinea-pigs the compound induced curare-like paralysis, while mice suffered convulsive seizures. The compound also had a potent local anaesthetic effect.168 The two main toxic principles of Moraea polystachya and M. graminicola have been identified as 16ß-formyloxy- and 16ßhydroxy- derivatives of bovigenin A (Figure 27).278, 280 The bufadienolides of the Iridaceae have most recently been reviewed by Steyn and Van Heerden.250
Drimia spp. (Hyacinthaceae)
(= Urginea spp.)
The vernacular name slangkop is used for some Drimia spp., as well as certain other members of the Hyacinthaceae such as Lindneria clavata (= Pseudogaltonia clavata) and even Ornithoglossum viride of the Colchicaceae. The name describes the snake-like appearance of the newly emerged flower heads. Drimia spp. are typical bulbous plants with a single flowering spike that emerges before the leaves in spring. The aerial parts die off in winter, but the perennial bulbs survive. Each flower is subtended at the junction of the flower and inflorescence stalks by a characteristic spurred bract. The spurs may be inconspicuous and are best seen in young flowers with the aid of a magnifying glass.
Slangkop may be distinguished from similar looking non-toxic plants by the following characteristics276 (Clare Archer, NBI, Pretoria, personal communication, 1996):
Albuca spp.: The flowers are erect with the inner three perianth lobes converging and hooded at the tips while the outer three are spreading.
Merwilla spp.: The upper part of the bulb is covered by fibrous sheaths. Apart from M. plumbea this genus is not of veterinary importance.
Ledebouria spp.: The leaves usually have a more horizontal arrangement and are marked on one or both surfaces by purplish spots.
Ornithogalum spp.: This often highly toxic genus (see Gastrointestinal tract) is not easily differentiated except by the bracts subtending the flower stalks (again best seen in young flowers) which are not spurred.
Dipcadi spp.: The leaves (see Central nervous system) are generally arranged in rosettes and are twisted.
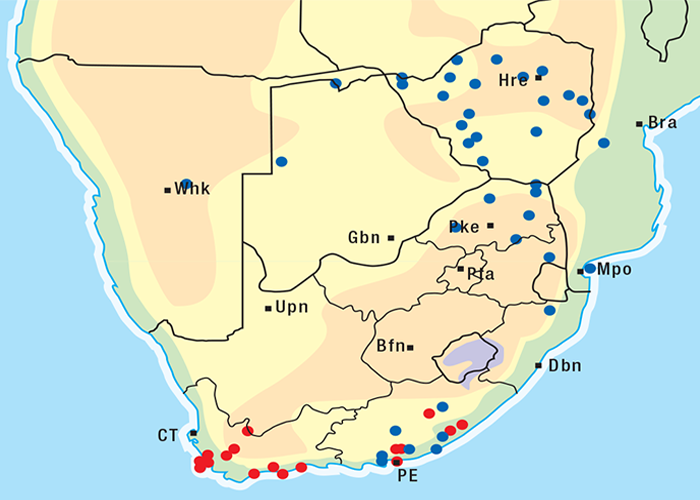
For the distribution of slangkop poisoning see Figure 31.
Drimia sanguinea (Schinz) Jessop
(= Urginea sanguinea)
Transvaal slangkop, Transvaalse slangkop
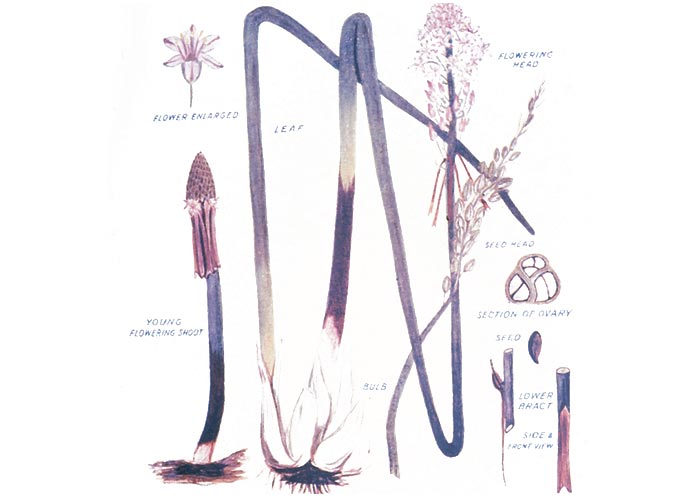
The large deep wine-red, pear-shaped, onion-like bulbs, c.150 mm in diameter, are usually buried just below the surface of the soil. They have fleshy scales that have a sharp, bitter taste when applied to the tip of the tongue. The single flowering stem c.300 mm long emerges before the leaves in spring and has a snakelike appearance (Figure 28). Numerous white or cream flowers are borne on the peduncle. Each petal is marked with a brown stripe down the middle (Figure 29). Propagation is by means of flat, black, winged seeds released from a tightly packed three-celled fruit, and by division of the bulb. The smooth, grey-green leaves, c.300 mm long and 25 mm wide, appear after flowering (Figure 30).44, 73, 101, 190, 218, 262
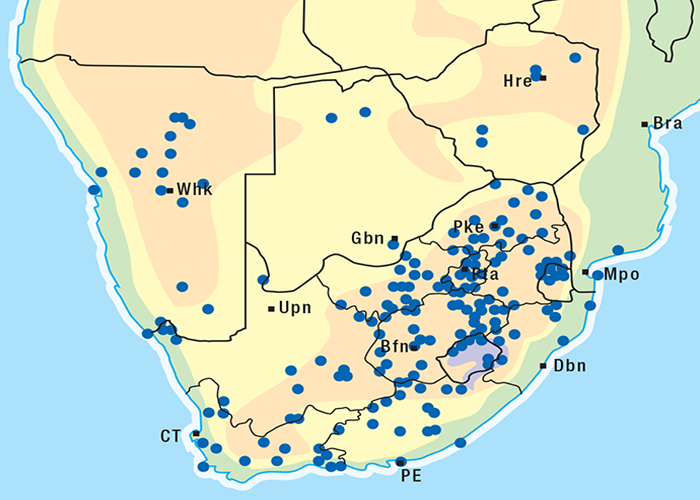
This highly invasive plant is widely distributed in Griqualand West, the western, northern and many other parts of the former Transvaal, Botswana, Namibia and Zimbabwe (Figure 31)101, 262 where it grows on a variety of soil types. D. sanguinea can be eradicated easily with a pick-axe since the bulbs are near the surface.44
Drimia macrocentra (Baker) Jessop
(= Urginea macrocentra)
Natal slangkop, Natalse slangkop
The bulbs are white, covered by fleshy scales and are smaller (25–50 mm diameter) than those of D. sanguinea. Long (up to 1,2 m) tapering flower heads appear before the leaves in spring. The flowers are fragrant, lilac coloured and star shaped. Bracts at the base of the lower flowers have a very conspicuous bifid spur. A single, long, cylindrical, shiny dark green leaf is produced (Figure 32).101, 160, 232, 294 The fruits are three valved.
D. macrocentra is found along the coast of KwaZulu-Natal and the former Transkei and inland up to an altitude of c.1 000 m.160 The plant is associated with periodically waterlogged conditions. Since it does not grow readily in well drained soil or tolerate too much moisture, it favours the margins of vleis160 where it is found in localized patches.
Drimia depressa (Baker) Jessop
(= Urginea capitata)
Bergslangkop
The bulbs, c.35–50 mm in diameter, are spherical in shape. Six to eight leaves, 150–300 mm long and 10 mm wide, develop when the flowers fade. The flowers are claret-purple outside and white inside.161
The plant occurs in KwaZulu-Natal (especially in the vicinity of the Drakensberg Mountains), Mpumalanga, Lesotho and the Eastern Cape province.161, 240, 247
Drimia altissima (L.f.) Ker-Gawl.
(= Urginea altissima)
Maerman
As the species name implies, this is the tallest of the slangkop group (Figure 33). The maerman (thin man) plants are often found in colonies and the large bulbs (120–150 mm in diameter) lie just below the surface of the soil. The bulbs are white fleshed with a brown outer tunic of dead scales. Like D. sanguinea the inflorescences appear before the leaves. In this species, the stem is exceptionally long, reaching 2,5 m, the white flowers have a green streak down the middle of each petal and the subtended spurred bracts are clearly visible in young flowers. The fruits are three chambered and the seeds are glossy, black, flattened and winged.218, 262
D. altissima is common in the Eastern Cape Province whence it extends northwards mainly through KwaZulu- Natal, Mpumalanga and the Limpopo Province into Zimbabwe, Zambia, Kenya, Uganda and beyond (Figure 34). It grows on a variety of soil types, often in black peat vleis or anthills in Zimbabwe, on sandy soils, and on the slopes of koppies.218, 247, 262
Two different plants were called D. altissima in the past.
The species in the winter rainfall area has now been reclassified as Drimia capensis (Brum. f.) Wijnands. The distributions of the two species apparently overlap in the Port Elizabeth district, but the specimens have not been critically examined. It is not known whether D. capensis is also toxic (Clare Reid, Botanical Research Institute, Pretoria, personal communication, 1986). Recently, however, it was determined through the FPIA test that this genus also exhibits cardiac glycoside activity (A.E. van Wyk, Department of Botany, University of Pretoria in conjunction with R.A. Schultz, OVI: unpublished results, 1996). As the differences between these two genera are minimal, they must for all practical purposes both be regarded as toxic.
Drimia physodes (Jacq.) Jessop
(= Urginea physodes)
The bulbs of this slangkop (c.40–50 mm in diameter) are rounded and white fleshed with a brown papery exterior. The inflorescences are c.50–150 mm in height and bear many small, white, purplish-keeled flowers. The flowers last only a day, but since very few open at one time the flowering time is protracted. Initially the young inflorescence is pyramid-shaped (Figure 35), but in time it becomes cylindrical. Six to twelve dark-green, sometimes curled, leaves (c.100–140 mm long) are produced (Figure 36). The fruit is three chambered and contains oblong, smooth black seeds.170
D. physodes is distributed in the Western and Northern Cape Provinces and western parts of the Free State (Figure 37). It favours alluvial washes at mountain bases or compacted gravel on pan margins.170
Pseudogaltonia clavata (Mast.) E. Phillips (Hyacinthaceae)
(= Lidneria clavata)
South West Africa slangkop, groenlelie
This is a relatively unimportant poisonous plant with a large cream-coloured bulb of up to 150 mm in diameter. The grey leaves (100 mm wide and up to 600 mm long) are generally produced together with or after the flowers. Tubular, white flowers with light-green, longitudinal bands are borne at the top of the stem (Figure 38).
It is found in the western Kalahari and in most parts of Namibia (Figure 39).262
Merwilla plumbea (Lindl.) Speta (Hyacinthaceae)
(= Scilla natalensis)
Blue hyacinth, blue squill, blouberglelie, blouslangkop
The blue squill is not a typical slangkop. The bulb (c.60 mm in diameter) is covered by a brown, membranous tunic and the leaves are uniformly green, c.140 mm long and c.40 mm broad at the base (Figure 40). The inflorescence, which is up to 350 mm long, is erect and rigid, the flowers deep blue, rarely pink or white.110, 142 Merwilla plumbea is an unimportant toxic plant which is common in KwaZulu-Natal.
Toxicity and chemistry of slangkop plants
Because they are bulbous, the various slangkop species are less dependent on rainfall than many other plants on the veld232 and therefore can draw on their own reserves to sprout in spring before it rains. In drought years they might be the only greenery for stock to eat on the barren veld during the critical months of September to November.160, 161, 232, 246, 294 The inflorescences and palatable young leaves are the most dangerous246, 294 as mature plants are less often eaten.294 After frost, slangkop-infested veld is said to be relatively safe. As is the case with tulp, stock that grow up on slangkop veld learn to avoid the plants241 with the possible exception of Drimia macrocentra. According to Mitchell160 local animals die as readily from D. macrocentra poisoning as newly introduced animals, but experimental evidence for this observation is lacking.
The whole plant, including the bulb, is toxic and dried plants retain their toxicity,232 the leaves of D. sanguinea reportedly being less toxic than the flowers.246 D. sanguinea has been shown to have a mildly cumulative effect in the rabbit241 and, rare instances of krimpsiekte-like signs vide infra have been recorded in stock poisoned by slangkop in the field.246
D. sanguinea is the most important and best known of all the slangkop species. Information on its toxicity has been provided by Dunphy,73 Shone and Drummond,218 and Joubert and Schultz.116 Milled, dried bulbs (stored at 4 °C) administered orally at 2 g/kg fatally poisoned 32 out of 36 sheep, but in a similar subsequent experiment two out of four sheep survived. At a higher dose of 2,5 g/kg, the material killed all the sheep to which it was dosed. The clinical signs appeared after about 17–24 hours, and the sheep died two to three days after commencement of the experiment.116
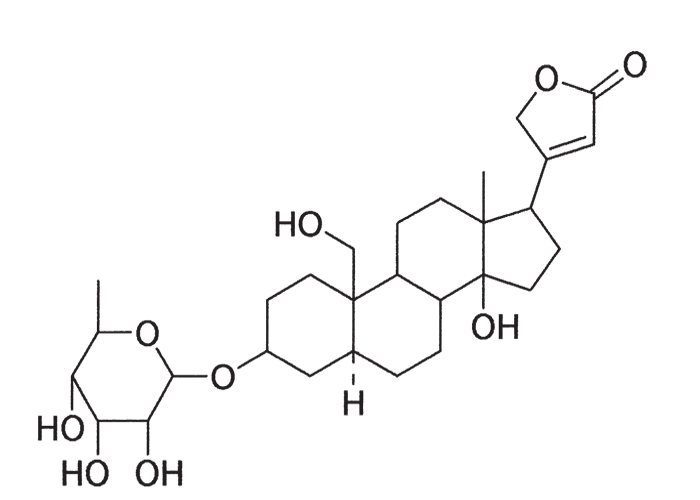
Early workers showed that the active principle of Drimia sanguinea was a glycosidal substance with a digitalis- like action.85, 91, 92 A cardiac glycoside, transvaalin, with a molecular formula C36H52O3, (Figure 42) was subsequently extracted from D. sanguinea bulbs by Louw.139 Hydrolysis of transvaalin yielded scillaridin A, and scillabiose, which indicated that transvaalin was either isomeric with scillaren A or a stable complex of scillaren A and scilliroside,139 a well-known rodenticide. The oral MLD of transvaalin for rats was 40 mg/kg.
Comparatively little has been published on the toxicity of other slangkop plants since Dunphy proved D. sanguinea to be poisonous in 1906.73 Flowering stems of D. macrocentra were lethal to sheep, goats, and cattle at doses of c.2,3–3,0 g/kg, and the leaves at c.3–5 g/kg. In all three species of livestock, the flowering stems were more toxic than the leaves.160 Whole D. capitata plants were fatal to cattle at c.2,67–3,6 kg, while c.0,5–1,8 kg caused only signs. The signs appeared after c.48 hours and the animals died in about two to eight days after commencement of dosing.161 A four-tooth wether of 43,5 kg live mass lived for only five and a half hours after being dosed with 5 g/kg of the bulbs (T.S. Kellerman and T.F. Adelaar, VRI, Onderstepoort, unpublished data, 1970). The toxicity of D. altissima was demonstrated in rabbits242, 243 and sheep.218, 238 Various parts of D. physodes (= U. pusilla) killed sheep within one to three days at doses of 1–5 g/kg (M. Terblanche, 1960; T.F. Adelaar, 1969; T.S. Kellerman, VRI, Onderstepoort, unpublished data, 1970); subsequently, 3 g/kg bulbs caused clinical signs in 17 hours and death in 22 hours, while 5 g/kg was fatal in less than 12 hours.170 The toxic principles of this species, physodine A and B are hellebrigenin derivatives. The non-toxic physodines C and D were also isolated and are the first naturally ocurring 14-deoxybufadienolides to be identified.274 D. calcarata (= U. rubella), given at doses of c.2,2–23,5 g/kg mortally poisoned a sheep within c.16–24 hours, the clinical signs appearing within approximately 9–16 hours.268 Some data is also available on the poisoning of sheep with an unnamed slangkop species, probably D. delagoensis.266
In addition to Drimia spp., Pseudogaltonia clavata bulbs, ingested at the rate of 4,9 kg (138 g/kg) over two days killed a sheep in 54 hours, while 9 kg (210 g/kg) ingested over eight days induced only signs.249 The mortally poisoned sheep showed signs consistent with heart failure, but the second sheep developed signs unusual for bufadienolide poisoning, such as haematuria and icterus. In a subsequent trial, 72,5 g/kg of dry bulbs administered to a sheep in daily doses of 2,5–10 g/kg over 17 days failed to induce any ill effects. Leaves of P. clavata collected in the late flowering stage, also failed to intoxicate a sheep which was dosed at 5–10 g/kg/day (total dose 50 g/kg) over 12 days (T.S. Kellerman and Margaret Wolf, VRI, Onderstepoort, unpublished data, 1971).
Merwilla plumbea at a high dose (18,3 g/kg) caused the death of a sheep in 12 hours.270, 271 Fresh bulbs dosed at 10 g/kg were fatal to a sheep in 12 hours, and a similar dose of dry bulbs killed two sheep respectively in 5 and 48 hours, signs appearing 20–55 minutes after administration. Electrocardiographic changes akin to those of cardiac glycoside poisoning were recorded, though cardiac glycosides could not be demonstrated by FPIA (Anitra R. Schultz, VRI, Onderstepoort, unpublished data, 1997). Schizocarphus nervosus (= Scilla rigidifolia) is also known to be toxic.265
Rubellin, a cardiac glycoside with a probable molecular formula C36H48O16, was isolated from D. calcarata (= U. rubella) in yields of 0,015– 0,045%.138, 140 Rubellin was reported to be more potent on a frog’s heart than ouabain, standard strophanthidin (BP 1932), lanatoside C, digoxin and digitoxin.210 The intraperitoneal LD50, of rubellin for rats was 0,692 mg/kg, and the oral MLD for rabbits c.10 mg/kg. Rubellin apparently acts as a cardiac poison in rats, while transvaalin acts on the central nervous system.138, 290 A bufadienolide, altoside,137 has also been isolated from D. altissima.
For additional information on the bufadienolides of slangkop plants, the review article by Steyn & Van Heerden250 should be consulted.
Bowiea volubilis Harv. ex Hook. f. (Hyacinthaceae)
The large globose tubers (100–150 mm in diameter) composed of thick, green scales grow partially above the ground (Figure 43). Long, many-branched, usually leafless green stems climb into trees or overgrow rocks (Figure 44). The greenish flowers are borne on long curved pedicils arising from the main stem.68, 101, 190
Bowiea volubilis has been shown in dosing trials to be toxic to dogs, sheep and goats.68, 118, 247 The plant is not of veterinary importance. A large body of work has been done on its bufadienolides, notably, by Katz123 who isolated from it highly active compounds, such as bovisid A, B and C. The chemistry of B. volubilis has been adequately reviewed by Watt and Breyer-Brandwijk.290
Thesium spp. (Santalaceae)
T. lineatum L.f.
Vaalstorm, witstorm
T. namaquense Schltr.
Namaqua Thesium
Thesium namaquense is a small, hemi-parasitic perennial shrub of up to 1 m in height, with many straight, greenish-coloured branches. The leaves are much reduced, small triangular scales, and the flowers are borne in the leaf-axils near the tips of the branches.262 The branches of T. lineatum are prominently longitudinally grooved (Figures 45–47).
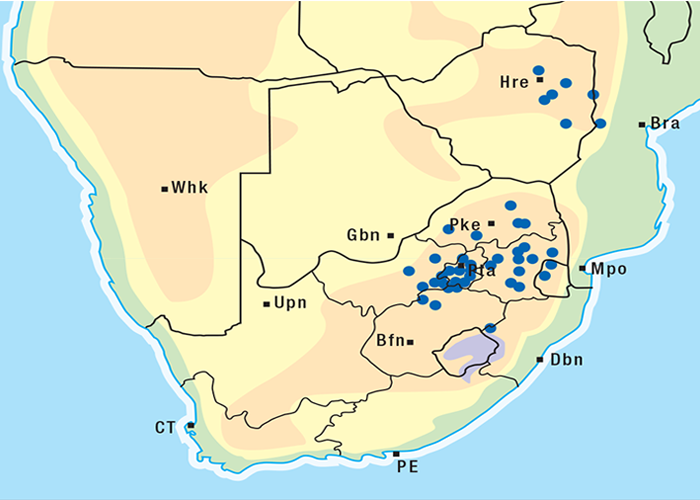
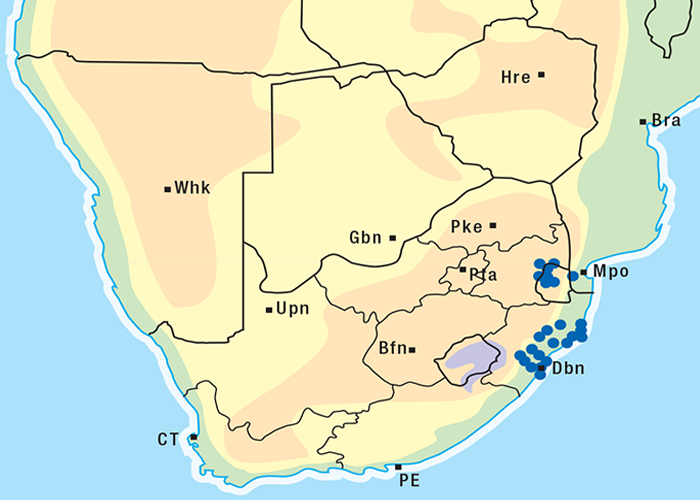
Thesium spp. are root parasites on plants such as Felicia muricata, F. filifolia, Chrysocoma tenuifolia, Pteronia sordida, Melianthus comosus and Lycium spp.247, 262 Joubert reported losses of sheep from T. lineatum poisoning in the districts of Beaufort West and Murraysburg. Most deaths occurred in winter on the southern slopes of hills, but losses can occur in other seasons and on other topographical situations as well. Affected sheep, goats and cattle usually die suddenly, but longer-lived ones develop diarrhoea and dyspnoea. Dosing trials with sheep confirmed that T. lineatum was involved (J.P.J. Joubert, Regional Veterinary Laboratory, Middelburg, Cape Province, personal communication, 1984). A bufadienolide, thesiuside, 5-O-acetyl- 3-O-ß-D-glucosylhellebrigenin, has been isolated from the plant.273 In addition to the usual lesions of cardiac glycoside poisoning, a nephrosis was often present.
T. namaquense, suspected of causing mortality of sheep in the Middelburg district, Eastern Cape Province, was toxic when dosed to rabbits and sheep. Sheep developed signs after being drenched with 50 g of the dried plant and another succumbed eight hours after receiving 200 g of the material. A dose of 100 g, administered on successive days, killed a sheep three hours after the second dose. Similar trials with T. triflorum failed to induce intoxication.240, 242, 243
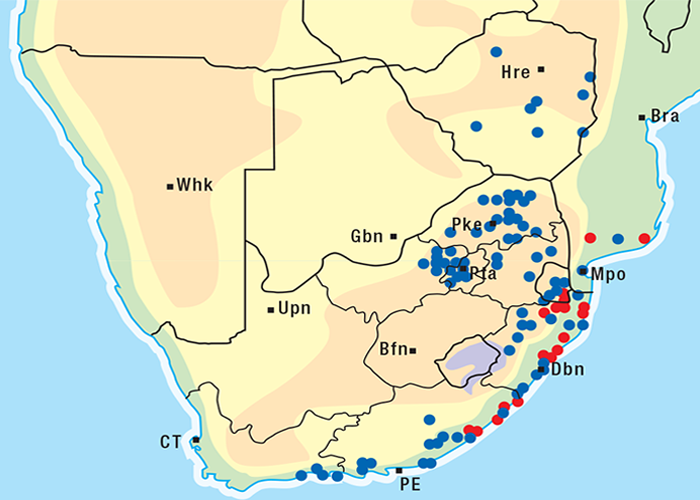
The approximate distribution of Thesium poisoning is given in Figure 1.126
Melianthus spp. (Melianthaceae)
M. comosus Vahl
M. major L.
Honey flower, kruidjie-roer-my-nie
This genus, represented by six species in South Africa, is widely distributed in both dry and humid areas.
Melianthus comosus is a sturdy, woody shrub standing c.1,5–3,0 m high, with leaves grouped at the ends of branches. The yellowish-green leaves are pinnately compound with approximately five pairs of toothed leaflets. A distinctive feature of the leaves is that the leaf stalks and midribs are winged. Brownish-red flowers, heavy with nectar, are borne in clusters below the leaves (Figure 47). The fruit is a membranous, inflated, four-chambered capsule, each chamber of which contains a round glossy seed.190, 262.
M. major is a large shrub that can reach a height of almost 2 m. The compound leaves are glaucous (covered with a fine bloom like a cabbage leaf) and quite glabrous. They are up to 450 mm long with the leaflets up to 180 mm long. The reddish-brown flowers occur in whorls of two to four on the upper half of the peduncle at some distance above the leaves. This is significantly different from the inflorescence of M. comosus, where the flowers are solitary at the nodes on the peduncle. The conspicuous bracts are glabrous, very shiny and also reddish-brown.
Both species are found throughout the North West and Eastern Cape Provinces, especially in the Karoo.262 M. major, the less commonly known one, is distributed from Sutherland and Calvinia, southwards through Clanwilliam and Caledon to Bredasdorp and eastward along the coast as far as King William’s Town.
The six main toxic principles, extracted from the root bark of M. comosus by Anderson and Koekemoer, were hellibrigenin 3-acetate and five new bufadienolides, melianthugenin, melianthusigenin, 6ß-acetoxymelianthugenin, 6ß-acetoxy-14-deoxy-15ß-epoxymelianthugenin and 14-deoxy-15ß-16ß-epoxymelianthugenin. A novel feature of these compounds is the presence of an orthoacetate group on ring A (Figure 48). Hydrolysis of the methanolic extract of the seeds with acetone containing 1% hydrochloric acid (gas) yielded scillaridine A, anhydroscillaridine A and 3-anhydroscilliglaucosidine.5, 8–10, 130, 131
The plants seldom cause poisoning, although some mortalities have been reported in equines and ruminants when grazing is scarce.69 Marloth, in 1913, recorded the observation of farmers that, although stock avoid M. comosus, they can be poisoned by the palatable mistletoe or voëlent, Tapinanthus sp. (= Loranthus sp.), growing parasitically on it. This indicated to him that the poisonous principle could pass from the host to the parasite.151 Some 50 years later, T.F. Adelaar and M. Terblanche (OVI, unpublished data, 1964) fatally poisoned a sheep at the laboratory with a Tapinanthus sp. removed from Melianthus comosus associated with the death of cattle in the Hay district. Similar specimens of Loranthaceae collected from Acacia spp. were non-toxic.
This transference of toxicity from the host to the parasite has been lately confirmed with the aid of fluorescence polarization immunoassay (FPIA). Cardiac glycoside activity of the same order as that of the host plant was registered in the hemiparasitic bird limes, Tapinanthus leendertzii (Loranthaceae) and Viscum rotundifolium (Viscaceae), growing on the very toxic Nerium oleander (R.A. Schultz & T.W. Naudé, OVI, unpublished data, 1995).
Ornithogalum toxicarium Archer & Archer (Hyacinthaceae)
This is a small plant with 4–10 filiform leaves; of which the solitary, ovoid bulb (15–25 mm in diameter) attenuates into a long neck below the ground (Figure 49). Small whitish flowers, with a central brownish to green stripe on the reverse side, are produced during a short f lowering period in spring.17
O. toxicarium is suspected of inducing cardiac glycoside intoxication in small stock. Poisoning of sheep and goats has been recorded in the mountainous Karas region of Keetmanshoop (Namibia),20, 32 as well as the Districts of Beaufort West and Merweville.32 Ornithogalum toxicarium has also been collected in the Northern Cape and Eastern Cape Provinces.17, 32
Positive FPIA results in some of the O. toxicarium plant parts assayed suggested the presence of cardiac glycosides in the samples.32Chronic poisoning with bufadienolides that have a cumulative, neurotoxic effect
Some Tylecodon, Cotyledon and Kalanchoe spp. (plakkies) contain cumulative neurotoxic bufadienolides that cause krimpsiekte, a paretic syndrome of sheep and goats, regarded as being one of the most important poisonings of small stock in South Africa (Figure 1).126 While chronic intoxication with cumulative bufadienolides results in krimpsiekte, acute poisoning may resemble that induced by other bufadienolides.169 Between the two extremes of acute intoxication and krimpsiekte, gradients of clinical signs are possible depending on dose, length of exposure, the particular bufadienolides involved and predisposing factors. Generally speaking, the cardiac and intestinal signs diminish and the paresis increases with chronicity. Intermediate cases, showing cardiac and gastrointestinal signs together with paresis, may be diagnosed either as subacute cardiac glycoside poisoning or krimpsiekte, depending on the clinician’s perception of the two conditions.11
Henning100, 101 distinguishes between acute ‘opblaas’ or bloat and chronic forms of krimpsiekte. According to him acute intoxication occurs in hungry, newly introduced stock that take in large amounts of Tylecodon spp. on veld heavily infested by the plants. These animals may die suddenly or show such signs as dullness, drooping of the lower jaw, paralysis (protrusion) of the tongue, salivation and the accumulation of half-chewed plugs of food at the back of the throat. Some of them may be bloated (opblaaskrimpsiekte) and in these instances regurgitation of ingesta can take place.100, 101 Longer-lived cases frequently stand with the back arched and neck extended, unable to masticate or swallow, drooling saliva and showing signs of dehydration. No mention is made of diarrhoea. If the stock are harassed at this stage they may collapse and die. The prognosis is poor.100, 105, 228, 245
The chronic form of krimpsiekte either follows on a protracted acute attack or is brought about by the ingestion of small quantities of the plant/bufadienolide over a long period. Animals suffering from typical krimpsiekte lag behind the flock, tire easily, walk with the head dangling loosely, and often lie down, usually with the neck stretched flat on the ground. Many will assume a characteristic posture, with the feet together, back arched, head down and neck sometimes twisted to one side (torticollis) (Figures 50 a, b and c). The torticollis can last for months or even years. In goats, the tail may hang.100, 228, 247 The name krimpsiekte, or shrinking disease, is derived from this characteristic stance.
Nervous stimulation has been reported in krimpsiekte100, 101 but the authors of this volume have not seen it and modern observers make no specific mention of it (J.P.J. Joubert, Regional Veterinary Laboratory, Middelburg, Eastern Cape Province, personal communication, 1985). Affected sheep, if disturbed, are said to show signs of hyperaesthesia and trembling or may even undergo tetaniform convulsions. Spasms of the head and neck muscles together with torticollis are common in lambs that chance to eat the plant. The wryness of the neck and paresis combine to make suckling difficult, and even if lambs are manually assisted to reach the teats, the muscles of the neck may be too weak to maintain the head in a suckling position. Spasms of the throat and tongue may further bedevil suckling.100, 101
Fatally poisoned sheep suffering from krimpsiekte become paralysed and lie fully conscious on their sides until they either die or are destroyed. Consciousness is maintained throughout the course of intoxication, which can vary from two to four days (acute) to many weeks (chronic). Mortality may be as high as 90% of the flock.101, 247
The clinical signs are aggravated by exercise, especially on hot sunny days101. Horses with krimpsiekte manifest pronounced torticollis (Figure 51) and hyperaesthesia. Other signs include restlessness, striking, kicking or biting at the abdomen, and paralysis. Unlike sheep, they lie down only towards the end of the disease.100, 101
Krimpsiekte in dogs resembles that in other animals. In addition, dogs often develop a high-stepping gait with the head bent down and the muzzle pointing at the chest (Figure 52). The lower jaw can be slack and the animals may salivate.100, 101
The heads of affected fowls droop, or the necks may be twisted so that their beaks point backwards (Figure 53). Eventually they become paralysed and lie prostrate for days until they die.46, 115, 240
Krimpsiekte has been caused reportedly by Tylecodon ventricosus105, 228 T. wallichii,100, 101 T. grandiflorus7 and Cotyledon orbiculata,255 as well as by the bufadienolides isolated from T. wallichii,27, 169 T. grandiflorus7 and Kalanchoe lanceolata.13 Under certain circumstances, K. paniculata, K. rotundifolia and K. thyrsiflora can cause paralysis.269
For comments on the relationship between structure and cumulative effects of bufadienolides, consult the discussion at the end of this chapter.
In Australia, stock have been poisoned by Bryophyllum spp. containing bufadienolides.145 Only acute cardiac glycoside intoxication and not krimpsiekte, was described.147 This exotic genus is commonly cultivated on rockeries in South Africa from where it readily escapes into adjoining veld,18 yet poisoning of stock by this plant has not been reported in this country (vide infra).
It is essential from a diagnostic viewpoint to realize that innocent succulents can be confused with the highly poisonous krimpsiekte-inducing plakkies. Typical examples of commonly encountered fleshy-leaved confuser species are the five-merous Crassula arborescens (round-leaved crassula, beestebal) and C. ovata (narrow-leaved crassula, kerkeibos). These are both shrubs with respectively either almost round to broadly ovate leaves with a waxy bloom and often reddish-rimmed margins, or oblongate comparatively narrow leaves with a sharp tip, without the waxy bloom. Another is the well-known fodder plant, Portulacaria afra (porkbush, spekboom), of the Portulacaceae which can be easily differentiated by its obovate to round, 1,5 to 2,5 cm leaves and fine, pink to purplish flowers borne in dense sprays at the ends of short, lateral branches.60
In all cases of suspected poisoning by plakkie-like plants the plants should be regarded with suspicion until a proper botanical identification has been made.
Tylecodon wallichii (Harv.) Tölken (Crassulaceae)
Nenta, kandelaarbos, krimpsiektebos, Wallich Cotyledon
(= Cotyledon wallichii)
This is a succulent bush up to 500 mm in height with short thick stems (Figure 54) and branches covered by woody protuberances (old leaf scars). The finger-shaped, fleshy, greyish-green leaves (c.100 mm x 10 mm) are crowded at the end of the branches (Figure 55). The flowers, which appear in December-January after the leaves have dropped off, are borne on upright, 300 mm long, terminally branched peduncles arising from the tips of branches (Figure 54). The pendant, greenish-yellow, bell-shaped flowers have five corolla lobes (Figure 56). The fruit is a papery five-chambered capsule containing numerous small brown seeds.101, 258, 262
Tylecodon wallichii is a xerophyte, native to the arid karoid areas of the Western and Northern Cape (Figure 57). The plant grows well on sandy or stony soils at the foot of hills262 on the lower slopes of mountains.100
Tylecodon ventricosus (Burm.F.) Tölken (Crassulacaeae)
Klipnenta or nenta
This perennial (up to 0,3 meters in height, when not flowering), usually has a branched, tuberous base, from which emerge one or more stems. The spirally arranged, linear to oblanceolate, non-waxy, herbaceous leaves (without a basal abscission layer), remain attached to the stems – old leaves just gradually wear away (Figure 58). The five-merous radially symmetrical (actinomorphic) flowers have yellowish green petals with purplish brown veins (Figure 59). They are borne upright on stalks (up to 24 mm long) in a contracted panicle, on a terminally branched peduncle (up to 0,38 m tall).29
T. ventricosus is widespread in the Northern, Western and Eastern Cape provinces (Figure 60), where it occurs in isolated patches.29
Tylecodon grandiflorus (Burm. f.) Tölken (Crassulaceae)
(= Cotyledon grandiflora Burm. f.)
Rooisuikerboom
This is a semi-succulent, perennial bush (Figure 61) with sparingly branched decumbent stems covered with leaf scars in the form of tubercles. As in Tylecodon wallichii, the leaves (80 mm x 10 mm) are produced seasonally at the ends of branches. The inflorescence, composed of an erect, unbranched peduncle (150–400 mm), is produced after the leaves have withered and have fallen off. The tubular flowers, orange to red, or yellow streaked with red, appear from January to March (Figure 62).7, 258
Tylecodon grandiflorus is confined to the Winter Rainfall Area where it grows on the western slopes of the Cape Peninsula and in the mountains northwards along the coast to Clanwilliam (Figure 63).7
Cotyledon orbiculata L. (Crassulaceae)
Pigs ears, hondeoor-plakkie, kouterie, plakkie, varkiesblaar
This plakkie is a low, shrubby, succulent plant with thick, fleshy, broad, rounded, grey-green, glaucous leaves, usually with red margins (Figure 64). The dark purple-red flowers are bell-shaped and pendulous (Figure 65).101, 262
According to Vahrmeijer, this highly ornamental but exceptionally variable species now includes several varieties which were previously regarded as separate, e.g. Cotyledon orbiculata var. oblonga (= C. leucophylla) and C. orbiculata var. dactylopsis (finger plakkie).262
The species is widespread in South Africa (Figure 66) and usually grows on rocky slopes in open or scrubby vegetation, often very exposed, rarely in sheltered, rocky outcrops.
In the northern bushveld the niche of C. orbiculata is filled by the very similar looking, but non-glaucous, C. barbeyi.
Kalanchoe lanceolata Forssk. (Pers.) (Crassulaceae)
(= K. pentheri Schltr.)
This is an erect, usually unbranched, annual, occasionally perennial (L. Dryers, NBI, personal communication, 1995) succulent plant standing c.1 m in height.262 According to Vahrmeijer,262 the lower stem is squarish and smooth, while the upper part is rounded and covered by hairs. The boat-shaped, stalkless leaves are smooth on the lower stem and minutely hairy towards the crown. The leaves, arranged in opposite pairs (Figure 67), become paper-thin, brittle and translucent in winter when the plant dies off (T.W. Naudé, OVI, personal observation, 1992). Star-shaped apricot or yellow flowers, with four corolla lobes (an obvious characteristic) that, are borne on inflorescences arising from the axils of the upper leaves (Figure 68). The fruit is a papery, four-chambered capsule with many seeds.262 Like many other plakkies it grows in the shade of trees and bushes, often in dense communities.262 Kalanchoe spp. are four-merous (i.e. the floral parts are grouped in fours and eights) as opposed to Cotyledon and Tylecodon spp., which are five-merous. The four corolla lobes or petals of Kalanchoe flowers are an easily recognized feature that distinguishes this genus from Cotyledon and Tylecodon spp. which have five-lobed flowers. The distribution of K. lanceolata in southern Africa is given in Figure 69; however, this is a ubiquitous species occurring widely in tropical Africa from Mali and Ghana in the west to Ethiopia in the east down to 27 degrees south. It has also been recorded in Madagascar, Yemen and India.80
Toxicity and chemistry of plakkies
As early as 1884 Hutcheon induced krimpsiekte in a dog by feeding it with livers of affected goats, and in two goats by drenching them with the strained stomach content of another suffering from the disease.105 Seven years later Soga228 resolved the aetiology of krimpsiekte by dosing Tylecodon ventricosus to caprines. He found that as little as 56,7 g of fresh leaves on three consecutive days caused typical signs of the disease in four days and death in six days of commencement of dosing. The results of his experiments were greeted with some scepticism, however, because the trials were carried out with local goats on a farm where krimpsiekte was endemic, and because no member of the Crassulaceae had previously been known to be toxic. Later, Tomlinson and Dixon independently confirmed Soga’s100 findings by feeding T. ventricosus to goats reared in the non-krimpsiekte areas of Sutherland and Victoria West. Some reservations were nonetheless expressed about the validity of both these experiments101 as the plants apparently had not been officially identified. Any doubts about the involvement of T. ventricosus in the aetiology of krimpsiekte was finally removed by Borthwick in a series of dosing trials at Somerset East, away from localities where T. ventricosus occurred, using plants from the farm where Soga had carried out his trials.101, 105 Botha et al. (1998)27, 29 noted that T. ventricosus induced severe respiratory distress in two penned sheep without significant electrocardiographic abnormalities being recorded. These findings suggested that T. ventricosus predominantly induced the paretic neuromuscular condition, krimpsiekte. A single large intraruminal dose of the plant (10 g/kg) resulted in paresis lasting two and a half weeks. A cumulative bufadienolide, tyledoside D, was subsequently isolated from the semi-dried plant material.27, 29
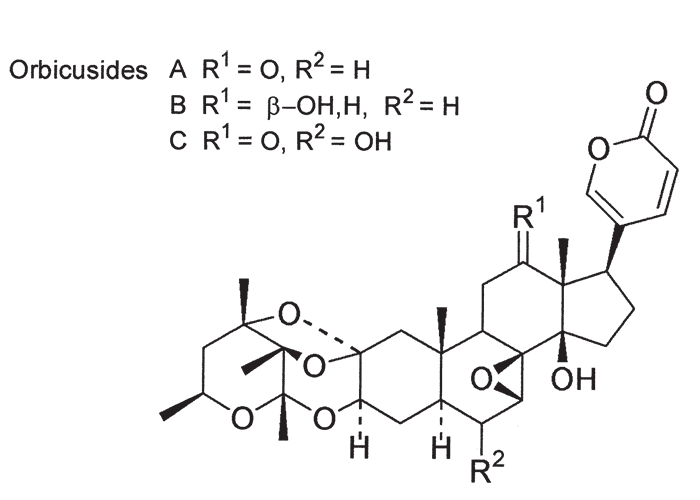
The second plakkie to be implicated in the aetiology of krimpsiekte was Tylecodon wallichii. Henning in 1926100, showed that it was highly toxic to goats, sheep, horses and fowls. Dogs that ate large amounts of the liver and meat of affected herbivores were themselves poisoned. Large doses of the plant (120 g or more) could kill sheep and goats within 24 hours, that is before typical signs had time to develop. Smaller doses of about 15–20 g/day precipitated krimpsiekte within a few days.100, 101 Also in 1926, Kamerman extracted from Cotyledon orbiculata and other plakkies, a non-glycosidal, amorphous substance, cotyledontoxin, which acted on the nervous system like picrotoxin.120, 121 Ten years later Sapeika suggested that, besides this neurotoxin, plakkies contained a substance, probably a glycoside, with the pharmacological properties of digitalis.209 Subsequently, a bufadienolide glycoside, namely, cotyledoside (Figure 70), was isolated from T. wallichii by Van Rooyen and Pieterse275 the structure of which was elucidated by Van Wyk.277 It is now clear that cotyledontoxin isolated by Kamerman in 1926, the structure of which was then not fully elucidated, in all probability was cotyledoside or a mixture of bufadienolides. The oral and subcutaneous 48h LD50 of cotyledoside for guinea-pigs was 0,173 mg/kg and 0,116 mg/kg, respectively.169 Naudé and Schultz169 reported typical signs of acute cardiac glycoside poisoning in a sheep following an intravenous injection of c.0,05 mg/kg cotyledoside, predominantly respiratory involvement after 0,025 mg/kg, and krimpsiekte after repeated doses of 0,01 mg/kg at intervals of 24 hours or more. That cotyledoside was the neurotoxic principle in T. wallichii causing krimpsiekte in small stock was confirmed by Botha et al., in 1997.27, 33, 34 Krimpsiekte was induced in two sheep by repeated intravenous administration of 0,01–0,015 mg/kg cotyledoside isolated from the plant by using paresis in guinea-pigs as criterion for identifying the toxic fractions. Both animals developed clinical signs typical of krimpsiekte without significant ECG abnormalities, attesting that cotyledoside at low doses does not overtly affect the electrical activity of the heart.33 Convincing evidence has been presented that Cotyledon orbiculata can cause both acute intoxication260 and krimpsiekte255 under natural conditions. However, of the three plants already discussed, C. orbiculata is probably the least important. In 1908 Burtt-Davy and Stent first drew attention to its toxicity for chickens and mentioned positive feeding trials carried out by Theiler.46 In 1912 Kehoe125 confirmed the toxicity of C. orbiculata to poultry and goats; 240 g of the plant was reportedly sufficient to induce krimpsiekte in the goat. C. orbiculata from a camp near Maltahöhe in Namibia, where sheep had developed signs of krimpsiekte in October 1963, has also been tested for toxicity.255 The lethal dose of this particular batch of plant material (semi-dried stems and leaves) for sheep and goats was 0,67–1,0 g/kg. Strong indications were found that the toxic principle had a cumulative effect; for instance, while 700 mg/kg was toxic but not lethal, two doses of 300 mg/kg on successive days caused mortality and as little as 50 mg/kg/day produced signs in 13 days. Both acute intoxication and krimpsiekte were observed.255 Besides ‘cotyledontoxin’, which has been isolated only by Kamerman,121 C. orbiculata has now been shown to contain digitalis-like compounds, as predicted by Sapeika209 and these are the principal toxins. In 1985 Anderson and his co-workers12 isolated four bufadienolides from C. orbiculata, namely, cotyledoside A, B, C and D. Cotyledoside A and B had a mild cumulative effect after four daily subcutaneous injections of 25–50% of the acute LD50, to guinea-pigs. Intravenous injection of 12–25% of the LD50 of cotyledoside A to a sheep induced clinical signs and electrocardiographic changes consistent with either subacute cardiac glycoside poisoning or krimpsiekte.12 Steyn and co-workers251 elucidated the structures of the cotyledosides and renamed cotyledosides A, C and D to orbicusides A, B and C. Cotyledoside B was found to be synonymous with tyledoside C.251 The proposed structures of the orbicusides are given in Figure 71.
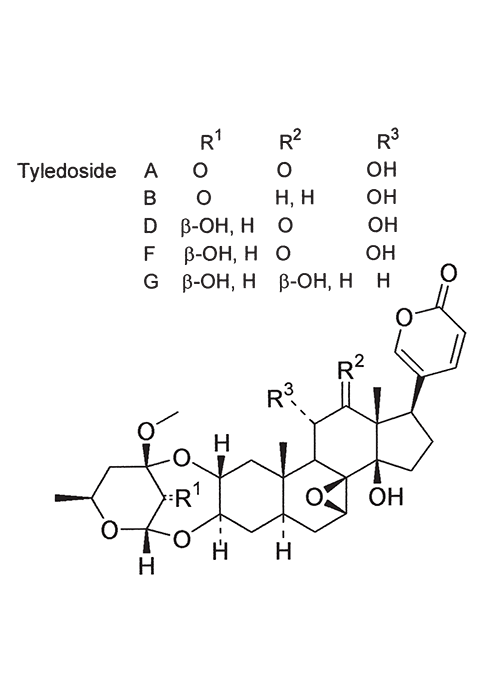
The toxicological properties and toxins of Tylecodon grandiflorus (Burm. f.) Tölken and Kalanchoe lanceolata Forssk. have been thoroughly investigated by Anderson and his co-workers.7, 11, 13 T. grandiflorus usually affects young cattle in the winter rainfall area of the Western Cape Province, where it causes acute intoxication, marked by ruminal stasis, salivation and death (D.J. Schneider, Regional Veterinary Laboratory, Stellenbosch, personal communication, 1985). Six bufadienolides have been isolated from T. grandiflorus.7 The ability of three of these bufadienolides, namely, tyledoside A, D and F (Figure 72), to initiate clinical signs or death after subcutaneous injection of 3 x 50% or 5–6 x 25% of the acute LD50, attest to their cumulative effect in guinea-pigs, but no such cumulative effects were evident with tyledoside C and E. Signs of krimpsiekte could be induced in sheep by the repeated oral administration of small quantities of the plant material or the repeated intravenous injection of small quantities (0,012 mg/kg) of tyledoside A and D.7
Kalanchoe spp. are widely suspected of causing botulism-like signs and haemorrhagic diarrhoea in cattle in South Africa; however, although highly toxic when dosed to sheep,11 Kalanchoe lanceolata has been positively incriminated only in poisoning of cattle in Zimbabwe.152 The clinical signs observed in the field over three successive seasons included haemorrhagic diarrhoea, tachycardia, tremors, aggression, terminal weakness, and collapse 24–48 hours after onset of signs. Pathological changes were consistent with those of cardiac glycoside poisoning. The condition was experimentally induced in a cow forcefed with 7 kg of the plant. As the plant is widely distributed in the continent, diagnosis of K. lanceolata poisoning may have been missed, especially in cattle.
Bryophyllum tubiflorum (= Kalanchoe tubiflorum) and five other Bryophyllum spp. naturalized in Australia, contain mainly the bufadienolides bryotoxin A, B and C. Intoxication of cattle with these plants resulted in persistent diarrhoea, which may be mucoid, blood-flecked, or melaenic.145, 179 The pure bufadienolides induced severe gastroenteritis characterized by ulceration of the omasal folds.148 Despite several species of this genus being naturalized in southern Africa, Bryophyllum poisoning has never been reported here.
Three bufadienolides were isolated and characterized from K. lanceolata11, 13 Hellebrigenin acetate was one of the bufadienolides isolated, and the other two were designated lanceotoxin A (5-O-acetyl-3-O-(2,3,4,5-tetrahydroxyhexanoyl) hellebrigenin) and lanceotoxin B (5-O-acetyl-3- O-α-L-rhamnosylhellebrigenin) (Figure 73). Acute cardiac glycoside poisoning could be induced by drenching milled, dried plant to a sheep and by injecting the bufadienolides intravenously to sheep or subcutaneously to guinea-pigs. Repeated intravenous injection of small quantities of lanceotoxin A or lanceotoxin B to sheep resulted in krimpsiekte. The acute subcutaneous LD50 of lanceotoxin A for guinea-pigs was c.0,20 mg/kg, that for lanceotoxin B c.0,10 mg/kg and for hellebrigenin 3-acetate c.0,36 mg/kg. Hellebrigenin 3-acetate proved to be non-cumulative.11, 13
The chemistry of bufadienolides in the Crassulaceae has been reviewed by Steyn and Van Heerden (1998).250
Numerous other species of the genera Tylecodon, Cotyledon and Kalanchoe, have been suspected of toxicity240, 247, 249, 290 but their importance in the field is not known.
Among the more notable features of plakkie poisoning is that the flowers reputedly exceed the leaves in toxicity100, 228, 247 that the toxicity of plants can vary both within and between localities120, 209, 247 and within seasons.31 Soil-type may also influence toxicity, e.g. Cotyledon orbiculata is less toxic on black clay than on sandy soils.237
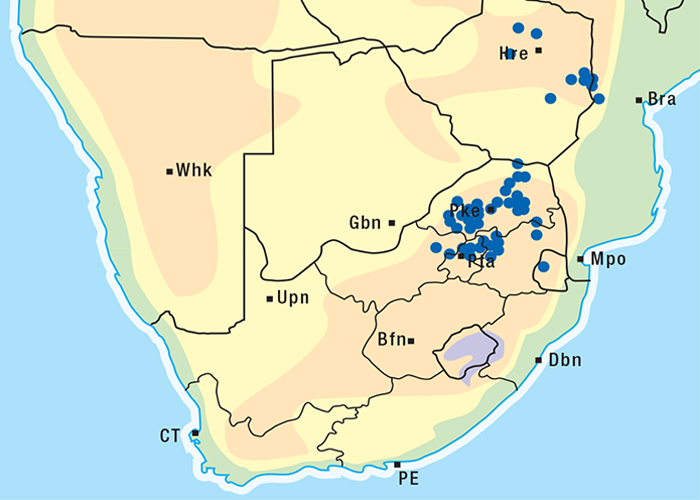
The incidence of krimpsiekte is highest in goats, followed by sheep, cattle and horses.100 Angoras reportedly are more prone to krimpsiekte than boer goats.100 Like any other cardiac glycoside poisoning, newly introduced stock suffer more frequently from krimpsiekte than local animals which tend to avoid the plant. Stock of all ages are susceptible but the young are more often afflicted probably because they graze with less discernment than the older ones.100 Plakkies have the distinction of being the only plants in South Africa that are known to cause secondary intoxication although there is some circumstantial evidence that people have been affected by the meat of sheep poisoned by Argemone.38 Dogs are particularly susceptible and, like humans, can be poisoned by eating the meat of sheep and goats that have died of krimpsiekte. Even cooked meat should be treated with caution as autoclaving for 15 minutes at 120 °C or cooking for 30 minutes does not destroy the toxicity. Krimpsiekte can occur throughout the year, but the incidence is highest in spring or early summer, particularly in the Winter Rainfall Area as then it is dry. Intoxication occurs especially in drought years. Losses diminish in good years when the grazing is abundant.100 Stock grazing on ridges, hills, mountain sides100 or broken veld, particularly on the southern slopes (J.P.J. Joubert, Regional Veterinary Laboratory, Middelburg, Eastern Cape Province, personal communication, 1985) where the plant grows abundantly, are most likely to be poisoned. Krimpsiekte is a problem of arid, karoid areas, especially in the southern parts of the Karoo and the Little Karoo (Figure 74). Acute poisoning by plakkies260 on the other hand, is not confined to the Karoo, and can result from the ingestion anywhere of garden waste containing popular ornamental species such as C. orbiculata. Poisoning of stock with Cynanchum spp. (krampsiekte) is sometimes erroneously referred to as krimpsiekte108 (see Central nervous system).
Absorption and disposition
The absorption, metabolism and excretion of cardiac glycosides vary according to the individual characteristics of the specific molecule(s) involved. All are sufficiently lipid soluble to be absorbed in lethal quantities from the gastrointestinal tract. Some are more rapidly absorbed than others resulting in acute intoxication within a few hours. They are also degraded by ruminal micro-organisms, but at high doses degradation is not fast enough to prevent intoxication.292 Cardiac glycosides are widely distributed in the body and can readily cross the blood-brain barrier into the central nervous system. Some are relatively slowly metabolized and may be excreted unchanged in the urine, while other compounds are fairly readily metabolized and/or excreted.122 Neurotoxic bufadienolides (e.g. cotyledoside) can have prolonged clinical effects.
Physiopathology of poisoning by cardiac glycoside-containing plants
The effect of cardiac glycosides on sodium-potassium adenosine triphosphatase was reviewed by Joubert in 1981.113 Myocardial or other cells depend on higher K+ and lower Na+ concentrations within the cell (relative to the extracellular fluid) for the maintenance of their resting cell membrane potentials. Since Na+ and K+ will diffuse across semi-permeable membranes, an active transport mechanism is required to maintain the intracellular electrolyte status quo against the concentration gradients. This mechanism is a sodium, potassium ATP-ase, known as the sodium-potassium pump. Failure of this pump leads to equalization of the intra- and extracellular ion concentrations, with consequent persistent frequent depolarization and disorderly transmission of nerve impulses. Cardiac glycosides are known to inhibit this pump. Inhibition of the active extrusion of Na+ leads to a rise in intracellular Na+ thus reducing the transmembrane Na+ gradient driving the extrusion of cytosolic Ca2+ during repolarization. Impairment of the sarcolemmal Na+-Ca2+ cation exchange mechanism results in accumulation of Ca2+ in the cell. Elevated cytosolic Ca2+ increases the force of myocardial contraction (the positive inotropic effect)86, 87, 113 at therapeutic doses or causes harmful Ca2+ overload at toxic doses of cardiac glycosides.181
The negative chronotropic effect (decreased heart rate) and negative dromotropic effect (decreased rate of conduction) of cardiac glycosides can be attributed to both the direct and indirect effects on the SA and AV nodes and on the conduction mechanism. The direct effects arise from changes in the trans-membrane potential, whereas the indirect effects (the more important effects) are brought about by a combination of enhanced vagal effects (vagotonic effects) and decreased sympathetic effects.86 At toxic levels various degrees of AV-block and disturbance of rhythm, automaticity and ectopy are encountered.
Hypokalaemia can lower the dose of cardiac glycoside necessary for intoxication, as K+ and cardiac glycosides apparently compete for an extracellular receptor which activates the intracellular sodium pump. Conversely, hyperkalaemia in massive digitalis poisoning, both in humans and sheep49 will intensify AV-block and depress the automaticity of the ventricular pacemakers, sometimes resulting in complete AV-block and cardiac arrest.
Cardiac glycoside intoxication is exacerbated by increased levels of Ca2+ outside the cell. Elevated extracellular Ca2+, in turn, results in higher levels of intracellular Ca2+ which may further inhibit the sodium pump. High Ca2+ may also directly decrease the speed of the excitatory current between cells and thus impair conduction. According to Skou,220 other cations, such as ammonium (NH4+, rubidium (Rb+), cesium (Cs+ and lithium (Li+), can interact with sodium, potassium ATP-ase in the same way as K+. Magnesium ions act as co-factors for the activation of sodium, potassium ATP-ase.95
The physiopathological features of experimental tulp poisoning were investigated by Button and co-workers.47–49 Tulp-intoxicated sheep develop progressive tachycardia and arrhythmias, systolic arterial hypertension, hypoxaemia, hypercarbia and acidaemia with depletion of plasma bicarbonate and rising lactate. The increase in heart rate was initially a sinus and later a ventricular tachycardia. The arrhythmia and eventual fibrillation were ascribed to the inherent arrhythmogenic effects of the bufadienolides, exacerbated by progressive hypoxaemia, acidosis, hyperkalaemia and elevated plasma catecholamine concentrations. The systolic blood pressure rose concomitantly with the heart rate, but the mean and diastolic arterial pressure or central venous pressure did not fluctuate significantly. Failure of the mean and diastolic blood pressure to rise in parallel with the systolic blood pressure was presumably due to the vasodilatory action of catecholamine on ß2- innervated beds.47–49 By stimulating anaerobic metabolism, the hypoxaemia and hypercarbia resulting from pulmonary dysfunction gave rise to increased plasma lactate, depletion of plasma HCO3– and a mixed metabolic/respiratory acidosis. Most of the physiopathological abnormalities seen in the experiments could be explained in the light of this hypoxaemia. Other changes included haemoconcentration, hyperkalaemia, hyperchloraemia and a rise in serum creatinine and plasma glucose levels. The marked hyperkalaemia, thought to be a sequel to the acidemia and glycoside-related inhibition of the sodium pump, could be of therapeutic significance. The cause of death was ventricular fibrillation or respiratory arrest.47–49 The severe pulmonary dysfunction, resulting in hypoxaemia and hypercarbia, when considered in conjunction with other parameters, would suggest that a primary vascular lesion was involved. Such a lesion could theoretically play a central part in the physiopathology of poisoning by at least certain cardiac glycosides in sheep.47–49
The pathophysiology of krimpsiekte is still obscure. In an in vitro study, the neurotoxic cardiac glycosides, cotyledoside and tyledoside D, were shown to be agonists at muscarinic acetylcholine receptors. The neuromuscular signs observed with krimpsiekte could therefore, conceivably, result from binding of these ‘cumulative’ bufadienolides to some of the nicotinic acetylcholine receptors at the neuromuscular junction.28
Diagnosis of plant-induced cardiac glycoside poisoning
Diagnoses of cardiac glycoside poisoning in the field are usually based on circumstantial evidence such as typical clinical signs, the presence of freshly grazed cardiac glycoside-containing plants on the pasture and necropsy features consistent with heart failure. In acutely intoxicated cases, fragments of leaves in the rumen may sometimes help to confirm a diagnosis. The similarity between these fragments and those of forage plants, however, as well as damage caused to the leaves by chewing and digestion, can complicate identification.
Electrocardiographic (ECG) recordings are useful aids for diagnosis, but they can seldom be made under extensive farming conditions. The expected ECG changes are prolongation of the PR-interval, depression or elevation of the RT-segment and an increase in amplitude, as well as inversion of the T-wave. The QRS complex might be widened as a result of delayed AV conduction. In addition, AV dissociation, varying degrees of heart block, evidence of ectopic foci and runs of ventricular tachycardia, are frequently recorded.
Diagnosis in the laboratory of field outbreaks have been hampered by the diversity of cardiac glycoside and their aglycones in the various plants. Several attempts have accordingly been made to develop practical direct or indirect methods for demonstrating cardiac glycosides in plant and animal tissue, e.g.:
- the rubidium test35 was too time consuming for routine use
- thin layer chromatography [TLC]153 high-performance liquid chromatography (HPLC) and quantitative nuclear magnetic resonance (NMR) studies (H.D. Brand, Medical University of Southern Africa, Medunsa, personal communication, 1993) could not be generally applied for lack of standards
- competitive radio-immunoassay (RIA) with antibodies of broad specificity to screen plants and animal tissue for the presence of immunoreactive cardiac glycosides200 held promise.
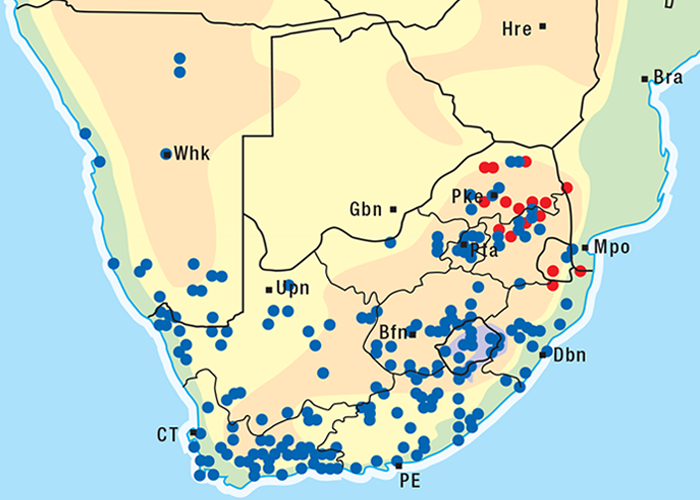
The fluorescence polarization immunoassay (FPIA) is a medical technique utilized for therapeutic monitoring of human patients on digoxin therapy. Digoxin plasma concentrations are monitored by demonstrating these cardenolides in human serum based on antigen/antibody reaction and competitive binding.54 Phytogenous bufadienolides were shown to have cross-reactivity with a commercially available fluorophore for digoxin, a cardenolide. This finding has made it possible to demonstrate cardiac glycosides in tulp (Moraea pallida, M. marlothii, M. polystachya, M. stricta, M. tripetala), slangkop (Drimia sanguinea, D. altissima, D. delagoensis), plakkies (Tylecodon ventricosus, T. wallichii, T. paniculata, T. reticulatum, Cotyledon orbiculata, Kalanhcoe lanceolata, K. rotundifolia, K. thyrsiflora, K. crenata), Gomphocarpus fructicosus (=Asclepia fructicosa), Nerium oleander and Adenium boehmianum. Cardiac glycosides have furthermore also been demonstrated by FPIA in Ornithogalum toxicarium, incriminated in isolated outbreaks of cardiac glycoside-like poisoning, of small stock in Karoid areas of South Africa and Namibia.32 Negative results were obtained in plants not known to contain cardiac glycosides (Merwilla plumbea, Gnidia burchellii, Ornithogalum prasinum, Senecio latifolius and Tribulus terrestris). Positive reactions were obtained in the rumen/stomach contents and organs of cattle, sheep, boer goats, horses and ostriches that had been poisoned by cardiac glycoside-containing plants.
The FPIA has enabled us to diagnose cardiac glycoside poisoning in cattle on the veld; ostriches that supposedly ate Gomphocarpus fructicosus; suni antelope believed to have ingested Kalanchoe lanceolata, and a dog that had mouthed a toad, Schismaderma carens (R.A. Schultz, OVI, unpublished data, 1997).
Treatment and prevention of acute bufadienolide poisoning
(a) Treatment
Joubert and Schultz114, 115, 116 demonstrated that activated charcoal (2 g/kg) was the most effective treatment for plant-induced cardiac glycoside poisoning of livestock in South Africa. Six out of six steers survived a lethal dose of Moraea polystachya after being treated with activated charcoal alone115 and three out of four wethers survived after being given activated charcoal plus potassium chloride.114 The charcoal was dosed to the animals as soon as the first clinical signs appeared, that is, 4–12 hours after a lethal dose of the plant had been administered. Activated charcoal and potassium chloride were equally effective against D. sanguinea poisoning: 80% of the treated sheep survived compared with 20% of the controls.116 Experimental evidence suggests that potassium chloride contributes nothing to the treatment115, 116 and could, in fact, be contraindicated.49 Oils or other substances, such as tannic acid, that can be adsorbed, should not be administered with activated charcoal as they deactivate the charcoal and are themselves rendered unavailable.
Although activated charcoal is an extremely effective treatment,134 it must be borne in mind that the stress of handling animals with hearts about to fibrillate can be fatal. To counter this danger of fatal conduction disturbances induced by cardiac glycosides and catecholamines during tulp poisoning, prior treatment with anti-arrhythmic drugs (to reduce the risk of fibrillation) and atropine (to prevent vagotonic conduction disturbances) seems to be strongly indicated. Swan accordingly tested the efficacy of atropine, lignocaine, acetylpromazine or propranolol administered alone, concurrently or before activated charcoal at the first signs of poisoning. Success of treatment was evaluated in terms of the numbers of animals that survived a toxic dose of Moraea pallida, changes in ECG, haematocrit, and levels of glucose, lactate and potassium in the blood. According to the findings of these rather limited trials, activated charcoal plus anti-arrhythmics or atropine were no better than activated charcoal alone (G.E. Swan, Faculty of Veterinary Science, University of Pretoria, personal communication, 1986). Indications were found that R56865 (Janssen Pharmaceutica, Belgium), a novel Ca2+ antagonist that selectively blocks intercellular Ca2+ overload, might have some clinical therapeutic effect against ovine epoxyscillirosidin and Moraea pallida poisoning.253 Premedication for activated charcoal treatment would probably be a better application for this drug.253
In addition to activated charcoal, other therapies might be considered, such as systemic alkalinizers to combat the metabolic acidosis and measures to normalize hyperkalaemia.47
Diphenylhydantoin, despite countering the electrophysiologic effect of cardiac glycosides,56 was discarded as a potential treatment of tulp poisoning because the therapeutic dose was too close to the toxic one (J.P.J. Joubert, ARC-VRI, Onderstepoort, unpublished data, 1978).
Working on the treatment of dogs poisoned by the cardiac glycoside and catecholamine containing skin secretion of the toad, Bufo marinus, Otani, Palumbo and Read (1969)182 proved that extensive synergism exists between the cardiac glycoside ouabain and the catecholamine epinephrine. They found that total adrenergic blockade with propranolol and dibenamine protected rats against the toxic effects of up to 100 times the LD of crude toad toxins. Propranolol was also used with some degree of success as adjunctive treatment in experimental Bryophyllum poisoning in cattle.146
(b) Prevention
Tulp poisoning is usually prevented by herding stock, fencing off infested areas or eradicating the plants. Where eradication of the plants by lifting the deeply buried corms is impractical, herbicides may be considered as an alternative control measure.234 Since tulp propagates in three ways, i.e. by forming cormlets in the axils of the leaves, forming additional corms under the ground and producing seed, any method of eradication should be carried out before these structures are formed.101 The same general principles of eradication apply for Drimia sanguinea, whose bulbs, lying just below the surface, are more easily lifted.
The above methods of controlling cardiac glycoside-containing plants, whether by mechanical or chemical means, all involve changing the environment; however, changing the environment may not be economically feasible under extensive farming conditions where capital and labour are lacking. Since changing the environment is largely impractical, attempts were made to control plant poisonings by manipulating the animals. Research was, consequently, directed at (a) developing a vaccine against tulp poisoning and (b) exploiting the natural ability of stock to avoid poisonous plants.
Vaccines:
In one attempt at developing a vaccine for the protection of stock against cardiac glycoside poisoning, sheep vaccinated with antigens prepared by the conjugation of digitoxin, G-strophanthin and proscillaridin with bovine serum albumin (BSA), were challenged with pure cardiac glycosides or cardiac glycoside-containing plants. Strong evidence of immunity to homologous cardiac glycosides were found. Joubert reported that 10 out of 14 sheep immunized with digitoxin-BSA survived a dose of digitoxin that killed eight out of eight controls. No immunity to Drimia sanguinea poisoning, however, could be demonstrated in sheep that had been repeatedly inoculated with individual, or combinations of, cardiac glycoside-BSA conjugates. This lack of cross-immunity to various cardiac glycosides is proving to be a serious impediment to the development of effective haptens (J.P.J. Joubert, Regional Veterinary Laboratory, Middelburg, Cape Province, unpublished data, 1982). Another approach was to prepare a vaccine by the conjugation of cardiac glycosides to so-called naked bacteria. In this procedure, the cardiac glycosides are conjugated to bacilli that have been stripped of all antigenic substances on their cell membranes. Antibodies can theoretically then be formed against the cardiac glycosides protruding from the cell membranes of the bacteria. Preliminary trials with sheep immunized with the digitoxin (cardenolide) vaccine and challenged with the homologue, or sheep immunized with proscillaridin (bufadienolide) vaccine (separately or together with the digitoxin vaccine) and challenged with Tylecodon wallichii, have not been encouraging (J.P.J. Joubert and K. van der Merwe, University of Stellenbosch, personal communication, 1985).
Conditioned feed aversion (CFA):
Natural conditioning of stock against tulp poisoning is so effective that (a) stock reared on infested veld seldom eat the plant and (b) poisoning usually occurs only in animals newly introduced from non-infested areas. Traditional and commercial farmers, especially in the eastern parts of South Africa, believe that naive stock can be taught to avoid yellow tulp by pre-dosing them with suspensions or various preparations of the plant. However, Strydom & Joubert (1983)252 – albeit in a limited field trial – could find no experimental evidence that pre-dosing naive cattle, respectively, with suspensions of charred leaves, sub-toxic quantities of chopped leaves or infusions of the plant, induced CFA.
Attention subsequently turned to extracting and identifying the factor(s) that induce CFA in Moraea pallida (yellow tulp). Part of the incentive for doing this was that CFA was a natural mechanism for protecting animals against plant poisonings. Controlling plant poisonings by means of CFA is thus completely environmentally friendly, as it does not disturb the environment in any way. Furthermore, CFA would have equal application in both commercial and communal farming systems.
‘Sniffer sheep’, previously averted to yellow tulp, were used to sense the presence of the aversive substance in fractions of the plant mixed in maize meal. Refusal of the fractions consumed by naive control sheep indicated the presence of an aversive substance, provided that the ‘sniffer sheep’ were still willing to eat pure maize meal. By following the refused fractions – obtained by solvent extraction and chromatographic separation – epoxyscillirosidin, the main toxic principle of yellow tulp, was identified as the aversive substance.224
In practice, cattle averted with epoxyscillirosidin, experienced difficulty in identifying the compound in yellow tulp growing naturally on kikuyu pastures. To obviate the problem, an epoxyscillirosidin-free hexane tulp extract, serving as an ‘identification factor’ for tulp, was dosed in conjunction with epoxyscillirosidin, during induction of aversion. This effectively strengthened the association between epoxyscillirosidin and tulp in animals averted to the bufadienolide.
Some evidence was also found that the epoxyscillirosidin, used in the induction of aversion, could contribute towards poisoning of averted animals grazing on tulp-infested pastures. This problem was addressed by partially replacing epoxyscillirosidin with lithium chloride, a non-lethal aversive agent, that does not affect the heart. Averting cattle with a combination of epoxyscillirosidin and lithium chloride, together with a tulp-hexane extract acting as an identification factor for tulp, effectively restricted severe poisoning of cattle on tulp-infested grazing.224, 225
Apart from M. pallida, CFA was experimentally induced with D. sanguinea and T. wallichii. In these, very limited trials, however, M. polystachya ostensibly was not aversive (T.S. Kellerman, L.D. Snyman & R. Anitra, Schultz, VRI, unpublished data, 1994).
No treatment is available for krimpsiekte.Fluoroacetate Poisoning by African Dichapetalaceae
Eighty-six Dichapetalum spp. have been described in Africa. Twenty-six of these are found in East and southern Africa, sixteen occurring in Kenya and Tanzania, seven in Mozambique, three in Zimbabwe, two in Namibia and one (D. cymosum) in South Africa.37 While some are trees, those involved in livestock poisoning usually are shrubs. Unlike in South Africa, where D. cymosum dies off in winter and losses are strictly seasonal, the species of Central and East Africa (being evergreen), are probably grazed throughout the year, resulting in different mortality patterns and lesions166, 174 (T. Fison, Vetaid Centre for Tropical Veterinary Medicine, Midlothian, Scotland, personal communication, 1993).
In Kenya D. ruhlandii was shown to be lethal among others to calves119 and in Tanzania a Dichapetalum sp. from a camp where D. ruhlandii, D. stuhlmannii and D. braunii grew, fatally poisoned goats.174 D. macrocarpum is also believed to be toxic to stock.283
D. barteri was lethal to goats in Nigeria175 and D. toxicarum, collected during a field outbreak of intoxication in Ndama cattle in Sierra Leone, fatally poisoned calves.272
Despite fluoroacetate (MFA) having been determined in only five species of this genus, there is good toxicological grounds to suspect that this compound occurs in nine others as well.97
All Dichapetalum spp., therefore, should be regarded as toxic until proven otherwise. The highest concentration of MFA recorded in this genus is 8 000 μg/g in D. braunii.176 MFA has been demonstrated in both D. cymosum149 and Tapura fischeri (P.P. Minnaar, Veterinary Faculty, Onderstepoort, personal communication, 1995), the only members of the Dichapetalaceae known to occur in South Africa. Unlike D. cymosum, T. fischeri (a tree) has never been involved in poisoning of stock.
Stock losses from phytogenous MFA in southern Africa have been reported only in South Africa, Botswana, Namibia and Zimbabwe. Although D. rhodesicum occurs together with D. cymosum in all these countries save for South Africa (Figure 78), this plant has never, as far as we know, been implicated in intoxication. A very limited trial revealed that D. rhodesicum was more readily eaten by eland than D. cymosum and water extracts equivalent to 10 g and 15 g fresh D. rhodesicum/kg was not toxic to two domestic goats, whereas extracts of much lower doses of D. cymosum was lethal (T. van der Merwe, A.G. Norval & P.A. Basson, Grootfontein, Namibia, personal communication, 1996).
Gifblaar Poisoning
Dichapetalum cymosum (Hook.) Engl. (Dichapetalaceae)
(= Chailletia cymosa Hook.) (= D. venenatum Engl. & Gilg) Poison leaf, gifblaar, makhouw (Tswana), umkauzaan (Sindebele)
Gifblaar causes sudden death of ruminants (particularly of cattle) in the Limpopo, North-West, Gauteng and Mpumalanga provinces of South Africa, as well as in Zimbabwe, Botswana and Namibia.
Gifblaar poisoning is estimated to be the third most important plant poisoning in South Africa, where it accounts annually for c.8% of all livestock deaths from plant poisonings and mycotoxicoses126 Some of the African Dichapetalum spp. are trees. Dichapetalum cymosum, on the other hand, can be likened to an underground tree of which the tips of the branches protrude from the soil. This growth habit is probably in adaptation to sandy soil and periodic veld fires (R. Archer, NBI, personal communication, 1996). A taproot-like stem, 10–70 mm in diameter, descends obliquely for a great length deep into the ground. Side roots are produced especially from the lower third of the main root. Near Pretoria a gifblaar plant was reportedly excavated for 30 m to a depth of 12 m. Long lateral branches radiate from a rootcrown 150–300 mm below the surface. These subterranean branches follow a tortuous upward path, ramifying near the surface to form a loose network of branchlets. Numerous buds are produced by the subterranean branchlets from which shoots can sprout. The aerial stems are short, usually 150 mm in length, and densely leafy (Figure 75). Fine, almost hairlike stipules are attached to the stem on either side of the short leaf stalks (Figure 76). The leaves are alternate, simple, usually broadly lanceolate, and bright green on both surfaces. The secondary veins are characteristically looped before reaching the leaf-margin, producing an arched effect (Figure 75). Young leaves are brown and hairy, but become green, leathery and usually smooth with age although some individual plants may remain hairy. The small, fragrant, off-white flowers are borne in dense clusters (Figure 75). The fruits are roundish, orange-yellow drupes (Figure 77), containing one to three seeds. Gifblaar rarely produces fruit.37, 101, 135, 162, 203, 230, 240, 262, 291
Mogg162 distinguished three forms of D. cymosum that could be present in the same area, namely, narrow-leaved (lanceolate), broad-leaved (oblong-elliptical) and hairy-leaved (narrow-oblong) gifblaar. The hairy-leaved form was regarded as relatively the most toxic. This variation in leaf form is illustrated in Breteler (1986).37
D. cymosum is a southern African species recorded in South Africa, Botswana, Namibia, Zimbabwe, Angola, Zambia and Mozambique (Figure 78)37 (Robert Archer, NBI, personal communication, 1994). The distribution of gifblaar poisoning in South Africa coincides almost exactly with that of the plant (Figure 79).126 However, this map, charting the distribution only of poisonings, does not include the pockets of gifblaar at Mkuzi and Ndumu (game reserves) east of the Lebombo mountain range in northern KwaZulu-Natal and in adjacent Mozambique at the latitude of Satara (the only record of gifblaar in that country) (Robert Archer, NBI, personal communication, 1994). In Zimbabwe, D. cymosum is found in the ‘gusu’ sands of the Kalahari geological system north-west of Bulawayo in the districts of Nyamandhlovu, Bulalima-Mangwe, Bubi and Sebungwe.254, 291
The distribution of D. rhodesicum overlaps with that of D. cymosum in Zimbabwe, Botswana and Namibia. The two plants are very similar, but D. rhodesicum can be distinguished by its pubescent leaves and branches.
Gifblaar frequents sub-tropical, usually dry bushveld areas where it favours the northern aspect of hills or sandy, well-drained sites.262, 291 According to Vahrmeijer, gifblaar forms part of a plant community known by farmers as gifveld, which typically includes Burkea africana (wild seringa), Ochna pulchra (lekkerbreek), Terminalia sericea (silver clusterleaf or Terminalia), Parinari capensis (grysappel)262 and Fadogia homblei (wild date) (T.W. Naudé, ARC-OVI, Onderstepoort, personal observation, 1985). The trees, Burkea africana and Ochna pulchra, are fairly reliable indicators of the presence of gifblaar. In KwaZulu-Natal, where these trees are absent, the plant is associated with T. sericea. In Zimbabwe, gifblaar variously grows with Acacia erioloba (camel thorn), Acacia erubescens (blue thorn) and Baikiaea plurijuga (Rhodesian teak).291
Gifblaar can be confused with similar local plants, such as the shrublet-form of Ochna pulchra (lekkerbreek), Pygmaeothamnus zeyheri (goorappel) (Figure 80), P. chamaedendrum (small goorappel) (Figure 81), Parinari capensis (grysappel) (Figure 82), Pachystigma pygmaeum (gousiektebossie) and P. thamnus (smooth gousiektebossie).101, 135, 191, 240, 262, 291 The differences between these plants will be discussed later.

Figure 80 Pygmaeothamnus zeyheri or the goorappel, somewhat resembling gifblaar or the gousiektebossie
Chemistry and physiopathology
Monofluoroacetate, the toxic principle of gifblaar, was first isolated in 1943 by Marais at the Onderstepoort Veterinary Institute.149, 150 This compound has subsequently also been recovered in toxicological significant quantities from Palicourea marcgravii St Hill71, in South America and from Acacia georginae21, 180 Gastrolobium spp. and Oxylobium spp.15, 16 in Australia. According to Harper & O’Hagan (1994)97 a number of plants can produce traces of monofluoroacetate in the presence of fluorine. The toxicity of Dichapetalum spp. in Africa has been reviewed by Vickery and Vickery.285
To understand the action of gifblaar it is necessary briefly to discuss the metabolism of acetic acid. Free acetate condenses with coenzyme A (CoA) to form acetyl-CoA, which reacts with oxaloacetic acid to give citric acid in the Krebs (tricarboxylic acid) cycle (Figure 83). Citric acid is converted firstly to cis-aconitic acid in a reaction catalyzed by aconitase and is then systematically decarboxylated through the agency of specific enzymes to carbon dioxide, water, oxaloacetic acid and ATP.
Fluoroacetate, a relatively harmless compound, is activated to fluoroacetyl-CoA before being converted stereospecifically through ‘lethal synthesis’ by citrate synthase to the highly toxic (2R,3R)-2-fluorocitrate (also known as (-)-erythro-fluorocitrate) (Figure 84). The other three possible non-toxic stereoisomers are not formed in vivo.124 The competitive but reversible inhibition of aconitase by fluorocitrate (which prevents the interconversion of citric to isocitric acid and blocks the tricarboxyllic acid cycle) results in a catastrophic loss of cellular respiration.79, 88, 186, 187, 189 Note that there is some evidence that plant aconitase is relatively resistant to inhibition by fluorocitrate.259
The inhibition of aconitase, however, only occurs at micromole levels of fluorocitrate, whereas it was more recently determined128 that at picomole levels fluorocitrate selectively and irreversibly inhibits mitochondrial citrate-dependent ATP and fatty acid synthesis. This occurs through covalent binding to proteins on the inner mitochondrial membrane associated with the mitoplast of liver, kidney, heart and brain tissue of the rat. It is obvious that inhibition of citrate transport would have important metabolic consequences for cellular processes dependent on the production of acetyl-CoA generated from mitochondrial citrate. These authors suggest that the neurotoxicity associated with fluorocitrate poisoning may be related to a deficiency of acetylcholine generated from this source.
The differences in the signs seen could probably be ascribed to variation in response of individual species to this metabolic insult.
Clinical signs and toxicity
Gifblaar poisoning is characterized by a latent period of four to 24 hours before clinical signs appear.77, 101, 233, 247 This is the time taken for the plant to be digested in the rumen and for the monofluoroacetate to be absorbed into the blood stream, to be transported to the target cells, to be converted to monofluorocitrate and to disrupt respiration. Monofluoroacetate is notable for the wide variation in response it induces in various species.52, 53, 187 For instance, the oral lethal dose for the dog is 0,06–0,20 mg/kg; for cattle, 0,15–0,62 mg/kg; for sheep, 0,25–0,50 mg/kg; for goats, 0,30–0,70 mg/kg; for fowls, 10–30 mg/kg.59 The major point of attack can be either the central nervous system (CNS) or the heart. In the case of carnivores it is the CNS187 and in ruminants it is the heart199, 247 that is primarily affected. Cattle that ingest lethal amounts of gifblaar and D. toxicarum typically drop dead, usually after drinking water or during exercise.101, 230, 233, 240, 247, 289 Clinical signs are rarely seen under natural conditions and recovery is exceptional; those that exhibit symptoms may die within hours or linger for days.101, 233, 240 A varied complex of clinical signs has been described, including anxiety, hyperaesthesia/depression, salivation, respiratory distress, ataxia (e.g. a high-stepping gait), peculiar stance, muscle tremors and tachycardia. Some animals may show tenesmus, void slimy faeces, bellow, and urinate frequently73, 101, 230, 233, 240 (H.E. van de Pypekamp, State Veterinarian, Rustenburg, personal communication, 1985).
Natural poisoning by gifblaar of wild game has never been recorded; however, game such as eland and kudu that browse appear to be more resistant to poisoning by gifblaar than grazers such as springbok. Antelope in the same environment as gifblaar may by natural selection have become resistant to it.22, 23 It is not clear whether livestock adapt to gifblaar-infested veld. Some authors believe that stock can learn from experience to avoid gifblaar291 but others hold an opposite view.101 West291 described kraals in the region formerly known as Matabeleland which were surrounded by gifblaar, yet the stock penned in them were rarely poisoned.
Although they are as susceptible to fluoroacetate poisoning as bovines (LD 0,5–1,75 mg/kg)59 horses are not naturally affected by D. cymosum. In fact, the plant is so unpalatable to horses that they are often used to graze gifblaar-infested veld which is lethal to cattle (T.W. Naudé, OVI, personal observation, 1998).
Burtt-Davy recorded the first outbreak of gifblaar poisoning in 1903.44 According to this author, specimens of a ‘highly poisonous plant’ collected in the former northern Transvaal by Brock, had been identified as Dichapetalum cymosum by MacOwan in 1890. Dunphy, in 1906, experimentally confirmed the toxicity of gifblaar73 though Theiler (unpublished data) had apparently poisoned oxen and rabbits with it three years earlier.233 Walsh,289 Steyn,233 Curson69 and others subsequently attested to the extreme toxicity of gifblaar.
Young leaves are believed to be more toxic than older ones101, 233, 240, 254 probably because the incidence of gifblaar poisoning is highest in spring (August-November) and autumn (March)162, 254 when new shoots appear. These rapidly growing new shoots arise mainly from preformed subterranean buds, the apical buds on the aerial parts being virtually functionless.162 Since gifblaar has a deep root system, it can sprout before the rains fall in spring when the veld is bare. At that time of the year gifblaar may be the only succulent forage for stock starved of greenery over the winter. Later (December-February), when the pastures are lush, the incidence of gifblaar poisoning diminishes dramatically, firstly because the plant is not readily taken in when other grazing is abundant and, secondly, because older leaves purportedly are less toxic.162, 291 However, the contention that only young leaves have high fluoroacetate levels was not borne out by recent trials in which mature green leaves were found to be almost equally toxic for several months.159 Although all growth stages of gifblaar are potentially lethal, succulent gifblaar sprouting after autumn rains – when the veld is deteriorating – is particularly dangerous. Pastures usually become safe in winter, after the first frost, when the leaves have dropped off.159, 162
The toxicity of plants, all parts of which are toxic,240 growing in one spot may vary considerably.162, 240, 291
Steyn240 quotes Marloth that the fruits of D. cymosum are eaten by humans in Ovamboland. Apparently, the fleshy portion only is eaten, while the pips are discarded (P.A. Basson, State Veterinarian, Grootfontein, Namibia, personal communication, 1983). Both the fleshy portion and the pips of drupes collected in Namibia, however, were lethal for rabbits at 5 g/kg (P.A. Basson, R.A. Schultz and T.S. Kellerman, ARC-VRI, Onderstepoort, unpublished data, 1983). The apparent non-toxicity of the fruit for humans could be related to differences in the relative amounts taken in or a disparity in the susceptibility of the two species, humans57 probably being much less susceptible than cattle. Under experimental conditions fresh leaves are not readily eaten by animals, even if the plant is mixed in feed or if the animals are starved.233 Sheep and goats rarely voluntarily eat more than 90 g, and cattle 270 g of the plant.233 Steyn found that 20 g fresh leaves was sufficient to kill a sheep, while 90 g was fatal for an ox240. Shaw is quoted by West291 as having killed a sheep and a goat by drenching them with 57 g of leaves; 450 g was fatal to cows, and 170 g caused severe signs in a heifer. In 1986 Egyed et al.77 fatally poisoned five out of five sheep with 1 g/kg of dry, milled gifblaar. West291 cites a report by Shaw that two dogs had died after eating a portion of the intestines of an ox that had been poisoned by gifblaar. Humans partook of the meat with impunity but ‘were careful to avoid the offal and the internal organs’.291 This observation could again be related to disparity in susceptibility, the LD50 for the dog being in the order of only 0.05 mg/kg59 most certainly lower than the c.5.0 mg/kg estimated for a human.57
Pathology
Effusions have been described in the body cavities of stock poisoned by gifblaar233 but this is exceptional. Most animals that died of gifblaar poisoning do not show lesions of any diagnostic significance.172 Small multiple foci of myocardial necrosis, often accompanied by lymphocytic infiltrates and early fibroplasia, have occasionally been noticed in stock that die on gifblaar-infested veld.213 Somewhat similar lesions have been induced experimentally in sheep by dosing them with sublethal amounts of monofluoroacetate over prolonged periods.213 These lesions also bear some resemblance to those induced by Acacia georginae, an Australian plant reportedly containing less monofluoroacetate than gifblaar.21, 25, 177, 293 The myocardial lesions of subacute or chronic monofluoroacetate poisoning, furthermore, must be distinguished from those of gousiekte, as gousiekte-inducing plants sometimes grow together with gifblaar. The lesions of fluoroacetate poisoning tend to be multifocal, less widely spread and more evenly distributed in the myocardium, and the fibrosis is less pronounced.177, 213
The lesions induced by the northern Dichapetalum spp. differ somewhat from those of D. cymosum poisoning. Pulmonary oedema is apparently commoner in poisonings by northern species and the myocardial lesions induced by them are closer to those of gousiekte vide infra than gifblaar poisoning. The pathological features of poisoning with the northern species probably most closely resemble those of A. georginae poisoning, where repeated intake of sublethal concentrations of fluoroacetate results in myocardial scarring.293
Prevention and treatment
West291 pointed out that, because of its well-developed system of underground stems, attempts to get rid of gifblaar by digging it out are seldom successful and often serve only to aggravate the situation. Repeated ploughing over years might eradicate the plant in lands162, 291 but this method cannot be applied to gifblaar growing in unbroken veld.162 Generally speaking, mechanical eradication is not practical.162, 240
Poisoning can be avoided by fencing off gifblaar patches, as the plant hardly spreads at all.101, 240 Few seeds are produced101, 162 and many of these are destroyed by insects. The plant is parasitized by a lace bug (Oncyochilla sp.) which causes leaf-curl, an unidentified leaf miner that leaves brown blotches, and the caterpillar of a moth (Episindris albimaculalis) that attacks the aerial parts, fruit and seeds. The seeds are recalcitrant: they cannot be stored at room temperature for more than about two weeks, or in a refrigerator for more than about four weeks, and freezing kills them. Moreover, seeds require moisture and relatively high temperatures to survive, but under such conditions they germinate. In nature, removal of the fleshy parts by millipedes is believed to prevent decomposition of the seeds before they are buried by some unknown animal (N. Grobbelaar, University of Pretoria, lecture to the South African Association for the Advancement of Science, March 1984).
Unfenced pastures should be avoided, especially in spring and autumn, when gifblaar sprouts. If infested pastures have to be grazed, grazing should take place in midsummer when the pastures are lush, or in winter after frost, when the gifblaar leaves have dropped off.162
Copper sulphate has been used traditionally to eradicate the plant. Leemann135 packed the salt mixed with calcium chloride around the ring-barked underground stem below the crown,135 but this is impractical on a large scale. In later methods, the tips of excavated stems were submerged in solutions of copper sulphate, the objective being for the plant to draw the phytotoxic fluid into its vast underground system (Figure 85). Bottles containing 1,0–2,5% copper sulphate are buried in standing positions near aerial stems; sections of the stems are then excavated for short lengths and the free points are immersed in the solution. According to one farmer the solution can last for about two weeks, after which the bottles are lifted and the process repeated on other aerial stems of the same plant. One bottle can apparently be responsible for the death of 5–15 growing points. In the first season up to 90% of the aerial stems are reputed to die and thereafter a portion of the surviving stems are killed each year until after about five years the entire plant has been destroyed. Copper sulphate is usually applied from January to June. The disadvantage of the system, though cheap, is that it is labour intensive and time consuming.
Several herbicides have been registered and used in South Africa with varying degrees of success for chemical control of gifblaar. Phillips (1993)192 tested granular formulations of ethidimuron and tebuthiuron, soil-applied liquid tebuthiuron and foliar applications of chlorfenac, glyphosate, picloram and imazapyr on this plant in Botswana. Only chlorfenac and imazapyr gave promising results. The former resulted in up to five year’s suppression of regrowth but this has, unfortunately, been withdrawn from the market. Imazapyr prevented regrowth for 3–4 seasons. They recommend a solution of 1,25 g active ingredient as a foliar application to fully developed leaves between November and February. Regrowth in subsequent seasons may have to be resprayed to kill the plant completely. Careful spot application is advised to limit grass damage.192 The directions on the labels must be carefully followed, as over-application will lead to defoliation and thus prevent absorption of sufficient herbicide to kill the plant.
No antidote has yet been found that can remove or inactivate fluorocitrate once it has been formed.187 However, quite promising results have been obtained in the prevention of fluoroacetate poisoning in laboratory animals by administering to them compounds which during their metabolism yield acetate. These acetate donors compete with monofluoroacetate for binding sites if administered early enough, that is, before exposure or in the beginning of latency. The best results have been obtained by two compounds, glycerol monoacetate (monoacetin) and acetamide. While monoacetin is effective only by repeated intramuscular injections, acetamide can be administered orally.77, 187
Egyed et al.77 tested the efficacy of acetamide as therapy for gifblaar poisoning in 10 sheep. Oral doses of 2,5–5,0 g/kg of acetamide administered at various intervals before and sometimes after 5 g/kg gifblaar, resulted in the survival of one out of five sheep while neither of two controls survived. When 2 g/kg of acetamide was dosed before or simultaneously with 1 g/kg of gifblaar, five out of five treated sheep survived compared with none of the five controls. They concluded that acetamide had a demonstrable prophylactic effect under these experimental conditions.77 Prophylaxis in the field would be more difficult to achieve, as gifblaar poisoning manifests itself as sudden death; by the time the first animals die others would probably be in the later stages of latency, making treatment futile. Since affected animals often die after drinking water, animals that have been exposed to gifblaar should be denied water for 48 hours and handled as little as possible. The slightest exertion can precipitate mortality.
Diagnosis
Gifblaar poisoning is usually diagnosed from circumstantial evidence, such as sudden death without notable lesions, on veld infested by sprouting gifblaar. Fragments of undigested gifblaar leaves in the rumen, distinguished by their looped secondary and conspicuous ‘honey comb’ tertiary vein structure, may assist in confirming the diagnosis. Care should be taken with the identification as some of these characteristics may be shared by harmless plants growing in association with gifblaar.
Monofluoroacetate could not previously be easily demonstrated in the laboratory, and changes in clinical pathological parameters, such as blood citrate levels76, 213 have been found to be too inconsistent for diagnostic purposes. However, gas and high-pressure liquid chromatographic techniques are available for the determination of monofluoroacetate. In 1997 Minnaar (1997) developed an uncomplicated method for demonstrating monofluoroacetate in plant and animal specimens. The technique involves single acidic (0,02M H3PO4) aqueous extraction, followed by isocratic high-pressure liquid chromatography (C-610 analytical column for organic acids, mobile phase 0,02M H3PO4, detection by UV at 210 nm). A yield in the order of 95% is achieved. This method has been successfully applied in epidemiological field trials and for the diagnosis of gifblaar and MFA poisoning, respectively, in cattle and pets.159
The distinction between gifblaar, gousiektebossies and similar plants
Plants associated with Dichapetalum cymosum can be divided into two categories, namely, those that are indicators of gifblaar and those that can be confused with it.
The two non-toxic plants most consistently associated with gifblaar and which serve as obvious indicators of its presence, are Burkea africana and Ochna pulchra.
Burkea africana Hook. (Caesalpiniaceae)
Wild seringa, wildesering or sandsering
This is a medium-sized tree up to 10 m in height with a dark-grey, rough bark (Figure 86). Young shoots and tips of branches are densely velvety and rusty-red to maroon. The bipinnate leaves with two pairs of pinnae and five to nine alternate leaflets per pinna are crowded at the ends of the branches. The leaflets are characteristically grey-green to dark-green and measure c.50 x 20 mm.60
Ochna pulchra Hook. (Ochnaceae)
Peeling plane, lekkerbreek
The lekkerbreek is a small tree 3–7 m in height (Figure 87), which can also occur as a shrublet of no more than 250 mm (Figure 88). The bark is pale grey, sometimes roughish near the base, otherwise peeling thinly to reveal a beautiful creamy, opalescent underbark. The spirally alternate leaves are elliptic to oblanceolate, usually 70–80 mm by 250 mm, often brick red in the very young stage, later becoming yellowish-green to dark green. The leaves are many veined and the margins are usually entire, but may be toothed towards the apex. Pale-yellow or greenish-yellow flowers appear in spring and are succeeded by fruit with three separate, black, kidney-shaped carpels and striking pink to coral-red persistent sepals.183
A third tree, Terminalia sericea Burch ex DC. (silver clusterleaf, silver terminalia, vaalboom), is not generally associated with gifblaar except in the northern KwaZulu-Natal sandveld where, in the absence of the two indicators, gifblaar and this tree grow together.
In gifblaar veld the veterinarian is usually confronted with the following plants of similar size and appearance to identify and distinguish between: Dichapetalum cymosum (Figures 75–77); the shrublet form of O. pulchra (Figure 88); Pygmaeothamnus zeyheri var. zeyheri (Figure 80) and P. zeyheri var. rogersii (the smooth and hairy varieties of the large goorappel respectively); and Parinari capensis (grysappel) (Figure 82). In some isolated areas, further confusion exists because of the presence of two alike gousiekte-causing plants, namely, Pachystigma thamnus (smooth gousiektebossie) and P. pygmaeum (hairy gousiektebossie). Although it cannot be confused with gifblaar, the gousiekte-causing Fadogia homblei may also grow in the same areas as Dichapetalum cymosum.
In order to distinguish gifblaar and the two gousiekte species from the somewhat similar but innocuous low-growing plants mentioned earlier, the following key has been adapted from Vahrmeijer.262
Leaves alternate; stipules small and narrow, if absent from old stems leaving a minute scar
GROUP 1 Gifblaar, grysappel and lekkerbreek
Leaves opposite; stipules triangular, short and wide, connecting the bases of the opposite leaf-stalks
GROUP 2 Gousiekte plants, goorappels
GROUP 1 Gifblaar, grysappel and lekkerbreek
* Leaves equally green on both surfaces-
Secondary veins joining to form loops near margin; tertiary veins conspicuous, in ‘honey-comb’ pattern; leaves leathery with smooth margin, usually glabrous but occasionally hairy
Dichapetalum cymosum (gifblaar)
-
Secondary veins ending in the leaf margin; leaves rather thin with margin, particularly near apex, finely toothed and rough to touch
Ochna pulchra (lekkerbreek)
** Leaves dark-green above, grey-felted on lower surface; stems usually with only two to five leaves
- Parinari capensis (Grysappel)
GROUP 2 Gousiekte plants and goorappels
* Plants 100–150 mm high with a fairly upright habit; leaves conspicuously broader in upper third, apex slightly twisted; petiole long and distinct. Young fruit two to three chambered
-
Leaves glabrous
Pygmaeothamnus zeyheri var. zeyheri
-
Leaves hairy
Pygmaeothamnus zeyheri var. rogersii (Large, hairy goorappel)
** Plants 50–100 mm high with the leaves growing out more or less horizontally; leaves not conspicuously broader in upper third, apex not twisted; petiole very short
- Leaves glabrous
-
Flowers green; young fruit four- to five-chambered in cross-section
Pachystigma thamnus (Smooth gousiekte plant)
-
Flowers white; young fruit two- to three-chambered in cross-section; plant not known to be toxic
Pygmaeothamnus chamaedendrum var. chamaedendrum (Small, smooth goorappel)
-
- Leaves hairy
-
Flowers green; leaves densely hairy, with yellow hairs; young fruit four- to five-chambered in crosssection
Pachystigma pygmaeum (Hairy gousiekte plant)
-
Flowers white; leaves sparsely or densely hairy with white hairs; young fruit two- to three-chambered in cross-section; plant not known to be toxic
Pygmaeothamnus chamaedendrum var. setulosus (Small, hairy goorappel)
-
Gousiekte
Gousiekte is a disease of domestic ruminants characterized by acute heart failure without premonitory signs, four to eight weeks after the initial ingestion of certain rubiaceous plants. The underlying lesion is a replacement fibrosis, particularly of the endocardium, accompanied by round cell infiltration.
The condition occurs in other southern African countries, but is most common in South Africa where it is rated respectively the fourth and fifth most important plant poisoning of cattle and small stock. The areas of highest incidence are charted in Figure 89.126
The following plants are involved in the disease:
Pachystigma pygmaeum (Schltr.) Robyns (Rubiaceae)
(= Vangueria pygmaea Schltr.)
Hairy gousiektebossie, harige gousiektebossie
The gousiektebossie is a low-growing shrublet with an extensive underground system of stems and roots. Short aerial stalks, about 80–150 mm in height, bearing four to eight leaves, grow out of a network of irregularly branched subterranean stems (Figure 90). The leaves are broadly elliptical, c.40–100 mm long and c.35 mm wide, uniformly bright green and covered by yellowish hairs. The leaves have short petioles (c.3 mm) and arise from the stem in opposite pairs. This opposite arrangement of the leaves is a useful feature for distinguishing Pachystigma spp. from Dichapetalum cymosum, which has alternate leaves. Well-developed triangular stipules connect the bases of opposite petioles on either side of the stalk at the leaf junctions. Clusters of pale-green, star-shaped flowers on short, branched inflorescences are formed in the axils of the lower leaves. The young ovary is usually four to five chambered with one ovule in each chamber, but some of these ovules ultimately abort, with the result that mature fruit usually contain only one seed. When identifying gousiektebossie, therefore, it is important to examine the ovary either in the flowering or early in the fruiting stage, before the ovules have aborted, because the number of chambers in the ovary is important for distinguishing Pachystigma spp. from the non-toxic Pygmaeothamnus spp. (goorappels). The fruit is round (c.20 mm in diameter), green and drupe-like, with remnants of the sepals attached to the distal end. In winter, the aerial parts die off.61, 62, 156, 193, 262, 282
Pachystigma pygmaeum is distributed in a belt across South Africa from the North-West Province through Gauteng to Mpumalanga and Swaziland in the east, with odd patches occurring in the northern Free State, the Limpopo Province and KwaZulu-Natal (Figure 91). The plant has also been collected in north-eastern Zimbabwe61, 62, 262 and Tanzania (specimens at National Herbarium of Kenya, Nairobi).
The gousiektebossie frequents open grassland in high-lying areas62 and is one of the shrublets of the highveld that remains dormant in the ground during the dry winter months. In spring the plant can utilize reserves of food and moisture to sprout before the grass, after the first rains. Although these tender shoots may seem very tempting for stock starved of greenery over the winter62 most deaths from P. pygmaeum poisoning occur in the latter half of summer (T.W. Naudé, ARC-OVI, Onderstepoort, personal observation, 1982).
The gousiektebossie on the highveld favours red sandy soil and stony veld (klipveld), while in the eastern highveld and the Waterberg district of the Limpopo Province it occurs in dolomite and sandstone areas.62, 262
Pachystigma thamnus Robyns (Rubiaceae)
Natal gousiektebossie, smooth gousiektebossie, gladde gousiektebossie
Pachystigma thamnus resembles P. pygmaeum save that the former is smooth (Figure 92). P. thamnus is most abundant in Mpumalanga and adjoining northern KwaZulu-Natal, the far eastern Free State and Gauteng. Small pockets are scattered in the North West and Limpopo provinces. The plant often grows together with P. pygmaeum, particularly in Gauteng and Mpumalanga.62, 156, 262 It favours poor sandy soils (Figure 93).
Pachystigma latifolium Sond. (Rubiaceae)
This plant is distinguished from all the other Pachystigma spp. in being an underground shrub having massive, woody axes. The annual, short-lived above-ground shoots are unbranched. In contrast to P. pygmaeum and P. thamnus, which are shrublets, the plant grows c.0,5 m tall and has much longer (30–40 mm) internodes. The glabrous leaves have short petioles, but unlike the others, the blade is broadly elliptic or occasionally broadly ovate (c.95x60 mm), its base rounded and the apex obtuse. The five-loculed and five-merous flowers are very similar, but yellow or yellowish-cream in colour. The large green fruit, usually five-seeded, ripens to dark-brown or black (Elizabeth Retief, NBI, Pretoria, personal communication, 1997) (Figure 94).
P. latifolium occurs on open and rocky grassland, the grassy banks of streams, in riverine woodland or on the coastal sand flats. The distribution in South Africa is given in Figure 95. This is the only plant known to cause gousiekte near the coast.
Fadogia homblei de Wild. (Rubiaceae)
(= F. monticola Robyns) Wild date, wildedadel
Like the other gousiekte-inducing shrubs Fadogia homblei has a perennial taproot with subterranean branches from which the aerial stems grow. The erect aerial stems are unbranched, squarish in cross-section, and 300–500 mm in height (Figure 96). Groups of three to five oppositely arranged leaves are borne in whorls at regular intervals along the stem. Young leaves are uniformly light green, while older leaves are dark green, shiny on the upper surface, greyish-white and felted below. Small yellowish star-shaped flowers form in the axils of the leaves (Figure 97), and these are followed by round fruits that blacken with age.62, 104, 262
F. homblei is common on white, grey or pale red soils in northern Gauteng, and the adjoining parts of the Mpumalanga and Limpopo provinces. It also occurs on the mountain ranges of the Magaliesberg and eastern escarpment from Swaziland to Zoutpansberg and northwards to the Limpopo and beyond into Central Africa (Figure 98).62, 104

Figure 96 Fodogia homblei. Note that the leaves are shiny-green on the upper surface and velvety-grey on the lower surface
Pavetta harborii S. Moore (Rubiaceae)
Pavetta, Tonnabossie
This is a perennial woody shrublet with subterranean branches giving rise to groups of aerial stems (Figure 99).
One plant can cover an area of c.2 m in diameter. The erect, smooth, greyish to pale-yellow, woody stems persist through the winter, though the leaves drop off. The opposite, sessile, oblanceolate leaves, measuring 30–45 mm by c.10 mm, are sparsely hairy on the upper surface, and pale and felt-like on the under surface. As in all Pavetta spp., characteristic opaque spots (bacterial galls or nodules) may be visible when the leaves are held up to the light (Figure 100). Clusters of white scented tubular flowers with star-shaped corolla lobes and protruding styles appear in early summer on the previous season’s growth (Figure 101). The common group name for this genus, bride’s bushes, is derived from these floral clusters. The small pea-sized fruits or drupes, becoming shiny-black with age, characteristically bear remnants of the calyx at the distal end (Figure 102).62, 261, 262
Tonnabossie grows on deep sandy soils mostly in south-eastern Botswana (Machudi area) and the north-western part of the Limpopo Province near Rooibokkraal, Ellisras and Soutpan (Figure 103).62, 261
Pavetta schumanniana F. Hoffm. (Rubiaceae)
Poison bride’s bush, gousiekte tree, gifbruidsbos, gousiekteboom
This Pavetta is a deciduous, much-branched shrub or small tree, growing up to 4 m in height, with dark-brown, furrowed bark (Figure 104). The large (75–150 mm by 70 mm), oppositely arranged oval leaves taper to the base and are rounded and broadest towards the tip. The leaves are yellowish-green, leathery with a rough upper surface and a hairy lower surface, conspicuously net-veined and covered with dots. Small, white, fragrant flowers are borne in clusters at the ends of short branchlets, in the axils of fallen leaves on the previous year’s growth. The fruits are small, round, black drupes when mature.62, 183
P. schumanniana extends from Central Africa into northern Namibia, northern Botswana, South Africa and Swaziland. In South Africa, it mainly grows in the sour bushveld of the Limpopo Province, Mpumalanga and northern KwaZulu-Natal, favouring rocky places or hill slopes (Figure 105).62, 183, 262
Clinical signs and cardiodynamics of gousiekte
Animals with gousiekte typically drop dead, usually without premonitory signs. Walker288 in 1908 to 1909 described gousiekte as follows:
Animals while at rest or grazing peacefully will suddenly describe one or two circles, bleat, or leap into the air, and fall dead. If by chance they had been grazing or ruminating, a bolus of food may be found in the mouth after death. In his own words:
‘Death has been observed to occur suddenly when the flock was made to move hurriedly; particularly when attempts were made to catch one of the sheep. Frequently when they are leaving the kraal in the morning one will fall, death occurring after a few minutes struggling. In other cases they have been kraaled for the night, all apparently healthy, and in the morning one or more may be found dead288’
Death can occur spontaneously or, more usually, it can be precipitated by exercise.1, 61, 104, 290 The most healthy-looking animals often succumb.61, 193, 290 In a minority of cases symptoms consistent with congestive heart failure can be observed; these include weakness, lagging behind the flock, staggering, dyspnoea and infrequent subcutaneous oedema.61, 191, 198 Recovery is rare.61
A study of the clinical signs and cardiodynamics of gousiekte by Pretorius and Terblanche revealed considerable variation in the time of onset of the signs that can be detected by auscultation. Not all the clinical signs were invariably manifested, and the signs could occur in various combinations in certain animals. In 10% of cases no signs could be observed before death. Pretorius and Terblanche summarized the sequence of clinical signs in experimental gousiekte induced in 50 sheep and goats as: dull first heart sound, arrhythmia, systolic murmur, split first heart sound, hyperpnoea, gallop rhythm, tachycardia and dyspnoea. All the notable changes appeared terminally, in the last two weeks of latency.195
Pathogenesis of gousiekte
Three types of arrhythmia could be distinguished by Pretorius and Terblanche, namely, runs of tachycardia, a dropped beat, and irregular rhythm. In the absence of evidence of heart block, the ‘dropped’ heart beat rhythm heard on auscultation were attributed to sinus arrhythmia resulting from PP-time alternation and irregular increases in PP-time intervals.195
An early decrease of myocardial contractility was indicated by conspicuous alterations of the ultra-low frequency acceleration ballistocardiogram and aorta blood flow recordings.195
Pretorius and his colleagues195, 196 concluded that the cardiac insufficiency in gousiekte was characterized by:
- functional cardiac dilatation, which causes symptoms of AV-valve insufficiency, gallop rhythm, bundle branch block, and an increase in P-wave duration
- cardiac ischaemia, which causes symptoms of wandering pacemaker, bundle branch block and ectopic ventricular beats
- decreased myocardial contractibility, which causes symptoms of generalized congestion, lung oedema, hydrothorax, hydropericardium and ascites.
From the evidence at hand Pretorius et al. deduced that the primary lesion in gousiekte was inhibition of the contractile mechanism of the entire myocardium, probably on a biochemical level, by the toxic principle of the plant.195, 196
Very little is known about the pathogenesis of gousiekte at cellular level. Pretorius and his co-workers195, 196 suggested that the transformation of energy in the hearts of gousiekte sheep could be defective. This suggestion is supported by evidence of cardiac ischaemia195 and dissolution of myofibrils216 in gousiekte, as well as reports in the literature of defective energy metabolism in other types of heart failure.223 Snyman223–227 cites Olson and Schwart as dividing heart failure at molecular level into two main types, namely, those in which the production of energy is defective and those in which the utilization of energy is inhibited. Studies of energy transformation in isolated heart muscle of gousiekte sheep revealed imbalances in both the production and utilization of energy.223–227 The uptake of oxygen by mitochondria isolated from affected heart muscle was found to be impaired. Any impairment of the function of these organelles can have potentially serious consequences for tissues, such as myocardium, which is almost wholly dependent on aerobic respiration for energy. The myocardium attempts to compensate for the shortfall in energy by increased anaerobic metabolism, as is evidenced by the high lactate levels and small though insignificant increases in the concentration of NADH in the myocardial cells. But these attempts at compensation appear to be inadequate.
Diminished levels of adenosine triphosphate (ATP) and creatine phosphate (CrP) point to a reduction in the total energy reserves, and increases in the ATP:CrP ratios indicate that this depletion is due to lowering of energy production rather than to excessive demand. The activity of n-actomyosin ATP-ase at varying concentrations of Mg2+ and Ca2+ was at the same time reduced, implying that utilization of energy by the contractile system was impaired. Depression of the superprecipitation activity of gousiekte n-actomyosin corroborated this finding and implied that n-actomyosin of sheep suffering from gousiekte dissociated more easily than that of unaffected sheep.
The effect of the biochemical lesions described here, could be exacerbated in vivo by secondary complications, such as reduced coronary circulation and cardiac ischaemia. No signs of cardiac hypertrophy were evident in the sheep observed in this experiment.223–227
The recent isolation81, 82 and characterization (R. Vleggaar, Department of Chemistry, University of Pretoria, unpublished data, 1997) of the toxic principle, pavetamine, together with the recent finding that laboratory animals are susceptible to this toxin, has given fresh impetus to the study of the pathogenesis of gousiekte. Studies in rats have revealed that pavetamine inhibits protein synthesis in the heart.214 This finding might well have bearing on the pathogenesis of a latent period. After administration of the toxin, the heart functions normally until myosin is depleted. Cardiac failure then follows when new myosin is not synthesized to replace that degraded during physiological protein turnover.214 For comment on speculation that gousiekte is an autoimmune disease, consult the discussion at the end of this chapter.
Pathology of gousiekte
Extracardiac signs of heart failure1–3, 155, 256 are present in many, but not all field cases of gousiekte172. These signs include cyanosis, congestion, marked oedema of the lungs, severe hydrothorax, slight hydropericardium and mild ascites. Earlier workers described dilatation of the heart (Figure 106) as commonplace257 but nowadays104, 172, 221 hearts of normal size (sometimes with thin and tough left ventricular walls) are often seen.172 Irregular areas of pallor may be apparent, particularly in the endocardium,172 which may be greyish in colour (Figure 107). Macroscopical cardiac lesions on the other hand, might be very subtle or even entirely absent.104, 172
In cases of natural intoxication, replacement fibrosis and evidence of atrophy of myofibres may often be the only detectable lesions. Gousiekte lesions reportedly are found most consistently in the apex where, as in other parts of the heart, they are prominent in the endocardium. In acute cases, the lesion may be limited to the apex, but eventually the left ventricle, septum, and even the right ventricle may become involved.221 Field cases generally display widespread endocardial lesions (J.A.W. Coetzer, ARC-OVI, Onderstepoort, personal observation, 1985). Note that connective tissue stains may be useful in determining the extent of the fibroplasia.172
The histopathological lesions of experimental cases described in the older literature include focal to fairly diffuse hyaline degeneration and/or necrosis of myofibres3, 104, 155, 221, 256 accompanied by lymphocytic infiltrates of varying intensity and fibrosis (Figure 108).104, 172, 256. More recent work, involving 33 sheep dosed with extracts of P. pygmaeum, F. homblei and P. harborii, however, has shed new light on the pathogenesis, range and nature of both the gross and histological lesions. Gross lesions, contrary to what was previously believed, are commonplace and reflect left-sided heart failure, namely, pulmonary oedema, hydropericardium and hydrothorax. The macroscopical lesions include dilation of the heart and/or subendocardial fibrosis, evident as a white streak below the endocardium (in c.25% of cases) or less commonly, as myocardial mottling. Hypertrophy of the myocardial fibres in the endocardial region of the apex, left ventricular wall and septum, being the initial lesion of gousiekte, is present in all cases. This is then followed by multifocal necrosis and replacement fibrosis. A less common sequel to the hypertrophy is diffuse atrophy (involving the entire width of the myocardium of the left ventricle and frequently also the right ventricle) without any evidence of replacement fibrosis. Sheep that die after a short latent period (less than five weeks) may show only hypertrophy or a few small indistinct foci of necrosis and replacement fibrosis, while their more long-lived counterparts (with a latent period of six weeks or more) manifest the characteristic multifocal to focally extensive replacement fibrosis. Foci of round cell infiltration, usually in association with the foci of necrosis and/or replacement fibrosis, though common, are not diagnostic or specific for gousiekte (S.S. Bastianello & L. Prozesky, ARC-OVI, unpublished data, 1998).
Ultrastructural changes include degeneration of myofibres, fibrosis of the interstitium, multiplication and enlargement of mitochondria, and duplication of nuclei.216 A lack of register between sarcomeres of individual and adjacent myofibres is accompanied by disintegration and necrosis of myofibrils, giving the myofibrils a frayed appearance. The disturbance of the orderly arrangement of myofilaments in the sarcomere is apparently due to a loss of myosin rather than actin. In more advanced stages of the disease, the mitochondria are enlarged and nuclear changes are evident. The nuclear membranes are involuted, and the nuclei are enlarged, irregular and duplicated. The degenerated myofibres are infiltrated by fibroblasts with the result that they often become surrounded by a matrix of collagen fibres.216 The loss of myofibrils is ascribed to deficient synthesis of contractile proteins.214 From the lesions that have been observed, it is clear that the function of the myofibres could be seriously impaired. For instance, reduction in the number of myofibrils could reduce the contractile force of myofibres and the envelopment of degenerated fibres by collagen could interfere with the normal transmission of impulses in the myocardium. The lesions are to a large extent irreversible because myofibres have a limited regenerative capacity.216
Toxicology of gousiekte
Gousiekte was at first confused with gifblaar because of the extreme suddenness with which death takes place in both diseases.61 According to Theiler,256 Mr J. Walker, the Government Veterinary Surgeon, Ermelo, (1904–1906), first reported the appearance of gousiekte, a serious new disease which caused sudden death of sheep.256 As a result of his investigations, Walker became convinced that the causative agent of gousiekte was a plant288 but the matter was not pursued until a severe outbreak in 1915 again brought attention on the disease. In this outbreak, 1 047 out of 1 761 sheep died of gousiekte after trekking across a purportedly toxic farm near Kaalfontein in the Pretoria district. A series of experiments was conducted on the suspect farm by Theiler and his co-workers between 1916 and 1922.257 According to Codd61 despite ‘numerous setbacks and conflicting results’, it was established beyond doubt in these experiments that Pachystigma pygmaeum was the cause of gousiekte. It soon became evident, however, that P. pygmaeum was not the only cause of the disease, as syndromes indistinguishable from gousiekte occurred where the gousiektebossie was scarce or absent. Several members of the Rubiaceae have now been incriminated in the aetiology of the disease, namely Pachystigma thamnus2 in Mpumalanga and northern KwaZulu-Natal, P. latifolium Sond. in Mpumalanga (T.W. Naudé, G.E. Swan and R.C. Tustin, Faculty of Veterinary Science, University of Pretoria, unpublished data, 1987) and KwaZulu-Natal (J. Randles, Provincial Veterinary Laboratory, Pietermaritzburg, personal communication, 1996), Fadogia homblei in areas around Vaalwater (Limpopo Province) and Bronkhorstspruit (Gauteng)104 Pavetta harborii on sandveld in the vicinity of Ellisras (Limpopo Province) and adjacent Botswana261 and Pavetta schumanniana in the eastern Limpopo Province, Mpumalanga, and Zimbabwe.
Toxicity of the plants
Without exception, fairly large quantities of the various plants have to be ingested for intoxication to occur. There is, nevertheless, strong evidence that a single dose can occasionally be fatal. Theiler et al.257 mentioned an outbreak in which 60% of 1 761 sheep died reportedly after they had been exposed to gousiektebossie for less than 24 hours, and in one of their experiments a sheep died 37 days after eating 0,9 kg Pachystigma pygmaeum in a sitting.257
The approximate LD100 of fresh P. pygmaeum, administered per ruminal fistula to sheep at Swartrand, was 175 g/kg and the LD50, was 100 g/kg; however, in this experiment three out of ten sheep receiving a single dose of 47 g/kg, administered in divided portions over one day, also succumbed to gousiekte. Desiccation of the leaves diminished their toxicity three- to four-fold. The estimated LD100 of air-dried leaves was 200–250 g/kg, or the equivalent of 510–640 g/kg fresh plant (1 kg of fresh material = 300–490 g dried). The latent period varied from 32 days to eight weeks or more, with an average of six weeks or so257 (T.W. Naudé, ARC-VRI, Onderstepoort, unpublished data, 1967). The lethal dose of dried Pachystigma thamnus for three goats was 210–500 g/kg with a latent period of 51–93 days.2
Fresh Fadogia homblei, collected in spring and early summer, induced typical gousiekte in cattle at doses of 0,21–0,50 kg/kg, with death occurring 28–54 days after the commencement of dosing. Allowing for a moisture content 75%, the lowest lethal dose was equivalent to c.53 g/kg air-dried material. In an experiment with dried F. homblei administered per stomach tube, four out of five goats died after receiving 133–176 g/kg, two of them with typical histopathological lesions. Two sheep survived doses of c.0,5–0,8 kg/kg of fresh wildedadel administered over 33 and 62 days, while cattle receiving 0,38–0,64 kg/kg of the same material in 31 and 57 days were fatally poisoned104. No correlation could be found between the amount of plant ingested and length of latency.
Little data is available on Pavetta harborii261 or Pavetta schumanniana, but all evidence points to their toxicities being similar to those of Pachystigma pygmaeum and Fadogia homblei. Indications have been found of variation in susceptibility between individuals of a species and between species.104, 257, 282 A sheep survived 152 g/kg of dry F. homblei dosed over 25 days, whereas a goat in a duplicate experiment died after receiving only 140 g/kg of the material. Moreover, when a goat and a sheep were dosed 500 g of dried material per day until they died, the goat succumbed without heart lesions on the nineteenth day while the sheep lived on until the fortieth day before it died of typical gousiekte. Goats, therefore, are probably more susceptible than sheep to F. homblei poisoning.104
Apart from differences in susceptibility of animals to gousiekte the toxicity of the plants is known to vary in different years and even in the same year. In a limited experiment, cattle were fatally poisoned by 20% of their body mass of F. homblei collected in spring and early summer, whereas ingestion of their own body mass of the plant collected in winter had no ill effect.104 This observation is supported by the finding of Fourie (N. Fourie, ARC-OVI, personal communication, 1995) that the concentration of the toxin in F. homblei can drop tenfold within two weeks during the early growing season. Toxicity may also be influenced by locality, soil type, and possibly climatic conditions.257
Isolation of the toxic principle
Gousiekte was an exceptionally difficult toxicological problem to investigate chemically, because the toxicity of the plants is variable and their toxicity diminishes during drying and storage, animals vary in their susceptibility to intoxication and there is a long latent period with which to contend. Since large quantities of the plants have to be dosed daily for weeks at a time in order to induce intoxication, it was impossible (in the absence of premonitory signs during latency) to know when a lethal dose has been given. Studies of the pathogenesis of gousiekte, or even calculation of an LD50, under these circumstances is not a simple matter.
Another major stumbling block in the investigation of gousiekte was that the disease cannot be induced by dosing the plants to laboratory animals.61 Chemical fractions had to be assayed for toxicity in sheep or goats, an expensive procedure requiring large quantities of valuable extracts and involving a lapse of many weeks before a result can be obtained. Nevertheless, in spite of these difficulties, many attempts have been made over three decades to isolate a gousiekte-causing component from the plants.6, 61, 282, 284
Fourie and his coworkers were the first to purify a gousiekte-inducing substance from a plant, namely P. harborii.81, 82 This compound has now been identified as pavetamine, a polyamine (R. Vleggaar, University of Pretoria, personal communication, 1997). Polyamines are much studied highly biologically active substances affecting many functions in the body including cell growth. Because some inhibit protein synthesis, artificial analogues of members of the group are being investigated abroad for the treatment of cancer and HIV/Aids. Pavetamine is the only naturally occurring member of this group to be incriminated in the poisoning of stock. A compound indistinguishable from pavetamine on TLC and NMR was also isolated from other gousiekte-inducing plants, namely Pavetta schumanniana, Fadogia homblei, Pachystigma pygmaeum81 and Pachystigma latifolium (N. Fourie, OVI, personal communication, 1996) indicating that they all contain the same toxic principle.
The effect of environment and the occurrence of gousiekte
The incidence of gousiekte is high when weather conditions or management practices upset the balance between grass and gousiekte plants in favour of the latter.61, 62 If the early summer is dry, Pachystigma pygmaeum may appear in January and February to cause heavy losses late in the season. This situation occasionally arises in the drier parts of the former western Transvaal, e.g. in the Potchefstroom district, where late growth of gousiektebossie during the summer of 1957 to 1958 caused many deaths in the following autumn.61, 62 Generally speaking, the extent to which gousiektebossie is eaten depends on the condition of the veld. Gousiekte is rife on overgrazed veld where the grass cover relative to gousiektebossie is sparse. Conversely, in exceptionally good years, with abundant grazing, stock may selectively graze gousiekte plants in late summer when the sour grasses of the highveld become unpalatable.63, 67 The incidence of gousiekte induced by Fadogia homblei has increased in the Limpopo Province as a result of the too-close settlement and overgrazing brought about by subdivision of farms.61 In the sour bushveld, it was common practice to burn the veld annually to remove the winter cover of dead grass, and this stimulates the growth of numerous dwarf shrubs, including F. homblei, just before or just after the rains in spring.104 By rule of thumb, F. homblei causes stock losses in early summer, while Pachystigma pygmaeum is responsible for deaths later in the season. Pavetta harborii and P. schumanniana can cause gousiekte throughout the year when grazing is sparse (T.W. Naudé and T.S. Kellerman, ARC-OVI, personal observation, 1982). Outbreaks cease after the first frost.193
Eradication and control
Although veld can be heavily infested with Pachystigma pygmaeum, spread by seed or by expansion of existing plants is very slow. Fruit is parasitized by insects and eaten by animals; removal of the soft parts leads to desiccation and mouldiness of the seeds; and seeds must apparently be covered by soil before they can germinate.156 Since gousiekte does not spread easily, it can be eradicated by repeated ploughing on arable land.61, 156 Continuous digging will eventually also destroy the plant on pastures, but mechanical eradication by whatever method is generally too costly to be a viable proposition.61
Gousiekte is best controlled by good pasture management.61, 63 Sheep appear to graze Pachystigma pygmaeum more readily than do cattle and, consequently, by grazing the two species together, theoretically the sheep can protect cattle by selectively removing the gousiektebossie. Intensive rotational grazing systems incorporating high stocking rates have been used to graze the gousiektebossie non-selectively, the underlying principle of this system being that the high grazing pressure will prevent the intake of lethal amounts of the plants by individual animals. As regrowth of gousiektebossie is slow it might be possible by repeated grazing down of P. pygmaeum by sheep to reduce the number of plants to non-toxic levels. And finally, on pastures that are kept young and palatable by judicious rotations and optimal stocking rates, stock are less likely to take in gousiektebossie.61, 63
Very little has been published on the chemical control of gousiektebossies.156, 286 Herbicides that contain fenac or picloram have been shown to control gousiektebossie without damaging the surrounding grass cover. Herbicides have been found to be most effective when applied in autumn, after active growth of the plant has ceased. The reason for this is not clear, but in autumn the surface area of leaves available for absorption of a chemical would be greatest, and if applied then, the herbicide is thought to remain in the system of the plants over winter. The concentration of the solution is important: if the concentration is too high the plant will be defoliated before sufficient herbicide is absorbed; if the concentration of the herbicide is too low the herbicide will not be effective. The most important factor militating against the use of herbicides is the cost of their application; however, since gousiektebossie tends to grow in patches, the actual area covered may be smaller than anticipated. Even on a badly infested pasture the actual area occupied by P. pygmaeum has been estimated to be as low as 5%.63
One of the difficulties in controlling gousiekte is that affected animals cannot be recognized during latency. It is important to identify these covertly affected animals so that they can be slaughtered, at least saving the carcase. Investigation of the clinical pathological changes in gousiekte by Fourie et al.83 revealed that, of the clinical pathological parameters tested, elevation of aspartate transaminase activity in the serum was the most reliable indicator of cardiac damage during latency. This finding has been successfully applied in the diagnosis of gousiekte in clinically normal animals during natural outbreaks. Knowing which animals are affected is a boon to stock owners, who once their animals start dying, have no means of telling when the mortalities will cease.
Other Cardiac Toxicoses
Gossypium spp. (Malvaceae)
G. arboceum L.
G. barbadense L.
G. herbaceum L. var. africanum Watt (Hutch. & Ghose)
G. hirsutum L.
Cotton, katoen
Cottonseed meal has been associated with acute, but more often with chronic, poisoning in non-ruminants (especially pigs) and pre-ruminant calves and lambs, vitamin A deficiency in cattle, reduced hatchability and discolouration of hens’ eggs (dark olive of the yolk and pink of the white) and severe constipation in stock consuming undecorticated meal.
The oil extracted from cottonseeds is used in salad and cooking oil, margarine, shortening, etc. and the meal obtained as a byproduct is a good protein supplement in food and feed for humans and livestock respectively.26, 112, 206, 222 Of the more than twenty recognized species of the genus Gossypium, only the species listed are cultivated worldwide for fibre production.26 The mature cotton bolls or fruits which are divided into three, four or five locks are harvested after five to six months of growth of the plants (Figure 109). Several ovoid seeds, 8–12 mm in length, each with their masses of fibres, are found in each lock. Ginning removes the staple fibres from the seeds. Small spherical or ovoid pigment glands, 100–400 μm on the long axis, are visible as dark specks throughout the tissue of the kernels. The colour of the glands ranges from light-yellow to orange, red and purple in a single kernel when viewed with a microscope. The colours may vary with growth and environmental conditions. Most or all of the gossypol pigments of the seeds are concentrated in the glands. The pigments occur in greater quantities in cotton roots than in seeds. Small amounts can also be found in other parts of the plants. The contents of gossypol pigments differ among the various species and are negatively correlated with environmental temperature, positively correlated with rainfall and also decrease after months of storage of the seeds. Time of storage of the ginned seeds and the type of commercial method used for the processing of the seeds also influence the types of gossypol pigments that will be present in the finished meals and oils.26
There are at least 15 pigments or derivatives in cottonseed extracts, oils and meals26 of which the yellow polyphenolic pigment, gossypol (C30H30O8) occurs predominantly. It is markedly reactive and has been shown to exhibit strong acidic properties, acting as a phenolic and an aldehydic compound (Figure 110).26 Gossypol occurs in cottonseed meal in a free or bound form. It is generally accepted that free gossypol is physiologically active, while that bound to the lysine in proteins is not.26 According to sources cited by Cheeke (1998)50 the tolerance of ruminants can be ascribed to the binding of gossypol to proteins in the rumen. This binding to proteins is promoted by the heat such as that generated by mechanical extrusion of the oil or even pelleting. Solvent extracted cottonseed meal, being subjected to less heat and pressure, contains more free gossypol and, therefore, is more toxic. In whole seed, the gossypol is almost totally in the free form. The methods of determining the free gossypol levels in food and feed have been reviewed by Berardi and Goldblatt.26
The toxicity of gossypol varies with age, breed and species of animal. Swine, guinea-pigs and rabbits are most sensitive, cats and dogs are intermediate and poultry and rats are least sensitive. Ruminants are not usually adversely affected by gossypol, but young calves and lambs, before the rumen is fully functioning, are susceptible. It is believed that gossypol forms complexes with protein in the rumen, rendering gossypol non-toxic. This mechanism is absent or poorly developed in pre-ruminant calves and lambs.26, 206, 222 Intoxication of adult ruminants (haemolysis and haemoglobinuria) by high levels of gossypol has been suspected of occurring in South Africa, but the diagnoses were never authenticated (C.J. Botha & T.W. Naudé, OVI, personal communication, 1997).
Small quantities of the toxin over prolonged periods can have a cumulative effect in animals, usually leading to clinical signs of poisoning one to three months after cottonseed meal has been introduced to the diet. Clinical signs include dyspnoea, anorexia, weakness, unthriftiness, poor growth, weight loss and sometimes diarrhoea. Calves are often emaciated and pot-bellied and have harsh coats. Death sets in after several days as a result, it is thought, of cardiac and circulatory failure.26, 112, 206, 222
At necropsy there is evidence of emaciation, heart failure and hepatic involvement. The most notable changes include serous atrophy of fat, accumulation of fluid in the thoracic, pericardial and abdominal cavities, congestion and oedema of the lungs, an enlarged dilated and greyish-brown mottled heart, hepatomegaly with accentuation of the lobulation and oedema of the gall-bladder wall. Scattered subcutaneous, visceral and serosal haemorrhages, gastroenteritis and whitish discolouration of skeletal muscles have also been reported.26, 112, 206, 222 Microscopically, there is atrophy, multifocal hyaline degeneration and necrosis of myocytes in the heart, accompanied by infiltrations of mononuclear cells and neutrophils and sometimes also evidence of fibroplasia. All the lobules in the liver show centrilobular necrosis and haemorrhage, often sparing only narrow rims of hepatocytes around portal triads.75, 98, 112, 171, 206, 222, 295 It is important that this lesion should be differentiated from a similar lesion seen in many, but not all, the lobules in the livers in cases of hepatosis dietetica associated with vitamin E/selenium deficiency in pigs.112, 117, 222 The cardiac lesions may also be confused with those of ionophore antibiotic poisoning.98
In their review of the influence of gossypol on reproduction, Randel et al. (1992)201 observed that the antifertility effects documented in many non-ruminant species were overshadowed by the other toxic effects. According to the authors cited by them, oestrus cycles, pregnancy and early embryo development were apparently disrupted by gossypol. These disruptions were probably brought about by endocrine disturbances and cytotoxic damage to the uterus or embryos. In males, gossypol induced immobility of sperm and depressed sperm counts. The immobility of sperm was attributed to mitochondrial damage in the tail and the low sperm counts to damage of the germinal epithelium. Fertility in females was less seriously affected than that in males.201
In South Africa, cottonseed and cottonseed meal are fed mainly as a protein source to cattle. There is little evidence of these products having significant deleterious effects on reproduction of female ruminants.39, 64, 84, 90 In bulls, sperm motility may be reduced39, 70, 201, 205 and structural changes in sperm such as abnormalities in the midpiece205 have been noted. Arshami and Ruttle (1988)19 reported larger lumens in seminiferous tubules and a reduction in the number of cell layers in the seminiferous tubules of bulls exposed to gossypol diets. These changes, however, were reversible. Though no difference in sperm motility was noted, the number of sperm with degenerative membranes was relatively greater in pubescent rams receiving regular cottonseed than those on the gland-free variety. Kramer et al., (1991) concluded that even moderate levels of gossypol could be detrimental to testicular function in pubescent ram lambs.132 However, not all experiments with gossypol induced adverse effects on spermatogenesis and semen quality70, 111 and the practical implications of gossypol on the fertility of ruminants in the field has yet to be determined.
It has been recommended that where cottonseed meal is the sole source of supplemented protein, the amount of free gossypol in the ration for non-ruminants should not be greater than 0,01% (100 ppm).26 However, the toxic effect of gossypol to swine and poultry can be reduced or in some cases eliminated by increasing the level of good quality protein and the supplementation of ferrous sulphate to their rations.26, 112 Calcium compounds have also been shown to have a synergistic effect with iron in the inactivation of gossypol.26 The addition of a 1:1 ratio of iron to free gossypol on a weight basis up to 400 ppm of free gossypol in the total ration will reduce the toxicity.26, 206
Rogers et al.206 reviewed the literature on recommendations for the feeding of cottonseed meal to animals and made the following suggestions, which may in some instances be conflicting. According to them, if cottonseed products are fed to adult animals, they should be fed for only short periods and should be withdrawn from the diet 12–14 days before parturition. Some workers feel that cottonseed meal must not be incorporated into the ration of young animals while others suggest that it can be fed without deleterious effects as long as the free gossypol does not exceed certain levels and as long as the meal is fed dry and not mixed with milk.206
Feeding of decorticated meal can either include limiting the amount of meal to be fed per day or limiting the free gossypol content of the total diet. As variations in the free gossypol content in the meal occur, the latter type of feeding is preferable. Nevertheless, it would appear that dairy cows can be fed up to 4 kg/day and adult sheep up to 200 g/day with safety, while young pigs should not be fed any cottonseed products. If the meal is fed to weaner pigs it should not exceed 5–10% of the total diet on a dry matter basis and should be fed only for short periods. The maximum permissible level of free gossypol in cottonseed meal is 20 ppm for animals, except cattle, sheep, goats, pigs, rabbits and poultry. The allowable concentration in feeds for cattle and goats is 500 ppm and for rabbits and pigs (excluding piglets) 60 ppm.
In natural outbreaks pigs,171 preruminant calves295 and adult goats75 have been poisoned by the prolonged feeding of diets containing roughly 200–400 ppm free gossypol.
Argemone spp. (Papaveraceae)
A. mexicana L.
A. ochroleuca Sweet subsp. ochroleuca (= A. subfusiformis)
Prickly poppy, Mexican poppy, bloudissel
Argemone spp. are annual or perennial exotic weeds, up to 1 m tall with prickly branches containing yellow sap and thistle-like leaves. The leaves of A. ochroleuca are glaucous and bluish-green, while those of A. mexicana are less glaucous and dark green. Their flowers are respectively white to pale yellow and bright yellow (Figure 111), the fruits spiny and capsule-like. Both Argemone spp. are common on disturbed soil and can become troublesome on fallowed lands, lucerne pastures and wheat lands,89, 127 and along roadsides. A. ochroleuca is widely distributed in southern Africa while A. mexicana is limited more to the east. As A. ochroleuca was once regarded as a variety of A. mexicana (M. Jordaan, NBI, Pretoria, personal communication, 1996) present toxicity data applies to both species.
The whole plant contains the toxic isoquinoline alkaloids berberine and protopine, while sanguinarine and dihydrosanguinarine are concentrated in the seeds.127
Argemone spp. are highly unpalatable plants and poisoning of livestock occurs rarely. Two suspected outbreaks of poisoning in cattle have come to our attention during the past 20 years. In both instances, cattle fed for several months with lucerne heavily contaminated with Argemone plants developed a severe subcutaneous oedema of the ventral parts of the body and became emaciated before they died (Figure 112). The most noteworthy changes in these animals included a severe ascites, hydrothorax, centrilobular and portal fibroplasia, as well as proliferation of sarcolemma nuclei (some of which were bizarre shaped), and fine vacuolation and loss of striation of some myocytes, especially in the endocardium. Severe dyspnoea was the most prominent clinical sign noted in natural and experimental ovine Argemone poisoning. In humans, wheat contaminated with large amounts of Argemone seeds have been associated with epidemic dropsy and glaucoma.127 The alkaloids in the seeds have been shown to cause dilation of the capillaries and leakage of fluid.127 In 1950 an outbreak of ovine and human epidemic dropsy, apparently resulting from the consumption of seed-contaminated wheat, was reported near Carnavon. The only ostensible contact one family had with Argemone was their consumption, over a three-month period, of meat from sheep that had died of the intoxication. Two of their five children died and three were hospitalized with cardiomyopathy indistinguishable from that of epidemic dropsy.38
Apart from the plakkies causing krimpsiekte (vide supra), this is the only plant suspected of secondary poisoning in South Africa.
Persea americana Mill.
Avocado, avokado
Three races of this well-known commercial fruit tree are recognized, according to their origin, namely Guatemalan, West Indian (or Colombian) and Mexican. These races also readily hybridize to give rise to numerous commercially available cultivars. Although all may be toxic, poisoning is usually associated with the Hass and Fuerte cultivars of the Guatemalan race.42
While their ripe fruit are eaten with impunity by humans, the Hass and Fuerte cultivars are toxic to budgerigars and canaries at excessively high dosages (c.50–100 g/kg).96 One case of putative intoxication of dogs by fruit has also been recorded.41 Intoxication of animals by various parts of the plant have been described, but stock poisonings usually result from browsing of leaves and (occasionally) unripe fruit.42, 65, 89, 144, 208, 229
The toxic principle(s) have been incriminated in cardiomyopathy and aseptic mastitis. An active principle, persin [(Z,Z)-1-(acetyloxy)-2-hydroxy 12,15-heneicosadien- 4-one], has been isolated from avocado leaves.178 At high dosages in goats cardiomyopathy is seen208 and at lower doses (c.20 g/kg) mastitis.66 Damage to the mammary gland results in agalactia, oedema and reddening of the udder, and clots in the large ducts. Microscopically, widespread degeneration and necrosis of the secretory epithelium with necrotic debris sloughing into the lumen, but without significant inflammatory response, are seen.66 Congestive heart failure has been reported in goats,89, 208, 229 sheep,89 horses144 and ostriches,42 resulting in anterior anasarca (involving especially the head, neck and brisket), persistent respiratory distress and sudden death. At necropsy pericardial effusion, hydrothorax and lung oedema were encountered. Severe degeneration and necrosis of myocytes with leucocyte infiltration characterized the cardiomyopathy.
Ergot Alkaloid Poisoning (Ergotism, Summer Syndrome or Fescue Foot)
Claviceps purpurea, C. cyperi
Ergot alkaloid poisoning in South Africa is a vasoconstrictive condition manifesting as dry gangrene of the extremities or hyperthermia (summer syndrome) following ingestion of ergots (sclerotia) of Claviceps purpurea and C. cyperi or endophyte-infected tall fescue (Festuca elatior). The endophytic fungus, Neotyphodium (= Acremonium) coenophialum synthesizes alkaloids similar to those of the Claviceps spp.30, 50
It should be noted that certain endophytic fungi, such as Neotyphodium lollii in perennial rye grass and the ergots of Claviceps paspali, although taxonomically related, produce indole-diterpenoid tremorgenic metabolites responsible for nervous disorders in stock (see Central nervous system).
C. purpurea is an obligate parasite of various grasses and cereals (Poaceae), and C. cyperi141 of the nut sedges, Cyperus esculentus and C. rotundus. The fungus replaces the seeds of the host plant with toxic sclerotia or ergots (Figure 113) which are dark grey or black, horny structures, usually somewhat larger than the host’s seeds (see Claviceps paspali, Central nervous system).
Ergot alkaloids are biogenic amines with varied and complex actions as partial agonists or antagonists at α-adrenergic, dopaminergic and tryptaminergic (serotonin) receptors. Some of these actions are completely unrelated and some even mutually antagonistic.188 The main effects seen in stock intoxication are due to agonistic α-adrenergic, peripheral vasoconstriction and agonistic dopaminergic interference with anterior pituitary function. The latter results in a precipitous reduction in the level of prolactin.
In summer the disease is usually characterized by hyperthermia (heat stress or ‘summer syndrome’) as a result of cutaneous vasoconstriction. Affected animals, especially lactating cows, cannot effectively dissipate heat through the skin. Animals show signs such as elevated temperatures and rapid breathing (often with the mouth open and tongue protruding, Figure 114) accompanied by excessive salivation; they also seek shade and frequent cool, moist places (Figure 115). Hot weather and exercise aggravate the condition, but in cool weather these signs may not be evident. With severe intoxication, especially in winter (particularly in calves), dry gangrenous necrosis of the extremities (ears, tails and lower limbs) is evident.50, 212
A serious economic complication of ‘summer syndrome’ is a sudden drop in milk yield. The severe drop in milk production is probably the result of decreased feed consumption aggravated by diminished prolactin secretion. Prolactin secretion during the peri-parturient period is essential for maximal milk production. It is important in initiating milk secretion and mammogenesis, but not for maintaining milk production.194
Conception is also deleteriously affected and may in the long term be of greater economic significance than the milk loss.212 Owing to melatonin imbalance, an abnormally long and reddish winter coat is also a feature of the disease. Mortalities are rare and after removal of the source several weeks may elapse before the hyperthermia abates and milk production is restored.50, 212
The first suspected outbreak of gangrenous ergotism in South Africa was reported by Schneider et al.212 in 1996 in cattle grazing on infected annual rye grass (Lolium temulentum and L. rigidum hybrids) near Bredasdorp in the Western Cape Province. Subsequently, widespread hyperthermia was manifested during summer and autumn by cattle in that province. The main outbreak, involving 2 600 dairy cows, was linked to the feeding of barley ‘sceenings’ contaminated by ergotized rye grass seeds. Ergotamine, ergonovine, ergosine and ergocornine were demonstrated in the toxic rations.212
During December/January 1996/97 hyperthermia and a drop in milk yield were reported in two dairy herds on the Highveld, one near Greylingstad (Mpumalanga) and the other between Memel and Newcastle (KwaZulu-Natal) (T.W. Naudé & J. Vorster, ARC-OVI and L. van Jaarsveld & A. Lawrence, Greylingstad, personal observations, 1996/7).
Endophytic fungi were initially suspected of being involved, but neither endophytes nor ergot alkaloids could be implicated in the condition (C. Roux & E. van der Linde, ARC-Plant Protection Research Institute, Pretoria and C.O. Miles, Pastoral Agricultural Research Institute, Ruakura Agricultural Research Centre, Hamilton, New Zealand, personal observations, 1997).
The source of the ergot alkaloids was eventually identified by measuring the serum prolactin concentrations of sheep fed on the individual components of the cows’ total mixed ration. Only the group on maize silage registered a statistically significant drop in the level of this hormone (S.I. van der Walt, ARC-Animal Improvement Institute, Irene, personal communication, 1997). The involvement of the maize silage was chemically confirmed by the high levels of ergot alkaloids, particular ergocryptine (Figure 116), found in the silage (115–975 μg/kg), as well as the total mixed ration (65–300 μg/kg) (G. Rottinghaus and S. Casteel, Veterinary Diagnostic Laboratory, University of Missouri, personal communication, 1997). The ergot alkaloid content (again, mainly ergocryptine) of the maize silage on the second affected farm was 875 μg/kg. Withdrawal of the silage resulted in gradual recovery of stock on both farms.
Nut sedge (Cyperus esculentus and C. rotundus of the family Cyperaceae) has a worldwide distribution and is a common weed in annual crops.99 It can be parasitized by Claviceps cyperi141 (C. Roux, Division of Biosystematics, ARC-Plant Protection Research Institute, Pretoria, personal communication, 1997) (Figure 117).
Careful examination of the maize silage from both farms revealed that it was contaminated by ergots, identified as those of Claviceps cyperi growing on the nut sedges from the maize lands.
Nut sedges were abundant on both farms. It is believed that late rain had resulted in mature, heavily ergotized nut sedge being cut with the maize for ensiling. Claviceps cyperi sclerotia, collected on the affected fields in the following autumn (E. van der Linde, Division of Biosystematics, ARC-Plant Protection Research Institute, Pretoria, personal observation, 1997), contained 3 600–4 000 mg/kg ergocryptine (G. Rottinghaus, personal communication, 1997). That ergocryptine was indeed the dominant alkaloid produced by this particular fungus, was confirmed by negative chemical ionization MS/MS (F. Ross, APHIS, USDA, personal communication, 1997).
Subsequent to these two outbreaks, ‘summer syndrome’ was clinically diagnosed on various farms on the eastern Highveld, where maize silage contaminated by ergotized nut sedge had been fed to cattle (T.W. Naudé, OVI, personal communication, 1997). In one outbreak, milled tef hay containing ergotized nut sedge (1 200 μg/kg alkaloid) was incriminated in the condition (C.J. Botha, Department of Pharmacology and Toxicology, Veterinary Faculty, Onderstepoort, and E. van der Linde, ARC-Plant Protection Research Institute, Pretoria, personal communication, 1997). Summer syndrome appears to be widespread in South Africa.
In the case of pigs, reduced body mass and agalactia are most prominent55. Signs of porcine ergotism have been recorded once in Zimbabwe in pigs fed ergotized bullrush millet, Pennisetum typhoides.219
Discussion
The accurate diagnosis of poisonings is of major importance in southern Africa, where cardiotoxic plants annually cause heavy losses of stock. Unfortunately, with the possible exception of gousiekte, which has distinctive fibrotic myocardial lesions, the diagnosis of cardiotoxicoses is not a simple matter. In the longer-lived cases of cardiac glycoside poisoning, signs of congestive heart failure might be evident at necropsy, and on histopathological examination small disseminated foci of necrotizing myocarditis might be seen. The difficulty arises in acutely intoxicated animals that die within a few hours of consuming these plants.172, 173 Newsholme and Coetzer172 reported that in these cases pathological changes are not detected by routine methods, presumably because lesions have not had time to develop to a stage at which they are recognizable. In view of the generally unsatisfactory method currently used to diagnose these plant poisonings, Newsholme and his co-workers173 investigated the veterinary application of certain techniques developed by medical scientists for the diagnosis of cardiac disorders. Limited investigations were carried out mainly on three techniques, namely, haematoxylin-basic fuchsin-picric acid (HBFP) staining of sections of formalin-fixed myocardium, observations for myocardial tissue fluorescence transmitted blue light, and measurement of myocardial sodium:potassium (Na:K) ratios. The HBFP technique is based on the principle that recently injured myocytes retain basic fuchsin dye more strongly than non-injured myocytes when differentiated with picric acid. In their hands, the HBFP method identified necrotic myocytes only in sheep that had lived for more than 15 hours after receiving a lethal dose of slangkop. As lesions of cardiac glycoside poisoning can be discerned in HE-stained myocardial sections at about the same time, HBFP had no particular advantage over the routine method. Tissue fluorescence, likewise, was found not to be more sensitive.173
The ratio of Na:K from specimens of the myocardium of groups of sheep poisoned by gifblaar was slightly, but significantly, lower than those of the controls. The possible use of Na:K ratios for the diagnosis of gifblaar poisoning in outbreaks with multiple deaths, therefore, deserves further investigation. No significant differences could be demonstrated between the Na:K ratios of control hearts and those from sheep poisoned by slangkop. This result was somewhat unexpected, as cardiac glycosides are known to inactivate the sodium pump.173
The most encouraging finding of their investigation was that creatine kinase (CK) and lactic dehydrogenase (LD) appeared to be consistently elevated in the pericardial fluid of sheep poisoned by gifblaar.173 More erratic results were obtained with regard to the activities of these enzymes in the pericardial fluid of sheep poisoned by slangkop.173
However, since the introduction of a method for determining monofluoroacetate in animal and plant tissue the urgency of this research has diminished.
According to Fourie et al.83 elevation of the activities of serum LD in sheep were less useful than that of AST in determining cardiac damage in the latent period of gousiekte. Neither could specific changes in the pattern of LD isoenzyme be discerned by them during this period. This lack of change in the isoenzyme pattern was attributed to the fact that the distribution of LD isoenzymes in the serum of normal sheep approximated that of cardiac tissue. In humans, on the other hand – where the LD isomer pattern in the heart and serum differ considerably – cardiac damage is accompanied by changes in pattern of LD isoenzymes in the serum.74, 133
Although AST and LD are widely distributed in the body, Fourie et al.83 attributed the elevated serum activity of these two enzymes in gousiekte to myocardial damage. In gousiekte, only the heart is affected and injury to other organs arising from congestive heart failure is rare (see Introduction, Liver); moreover, in their experiments the activity of GGT remained within normal limits, indicating that hypoxic damage to the liver was absent. They concluded, therefore, that the elevated activities of AST and LD had resulted from myocardial rather than liver damage.83
The extent to which cardiac function is affected by the changes that are heard on auscultation and recorded on ECG, or seen on histopathological examination, is not always clear. Even in cases where the heart is grossly dilated in gousiekte, little pathological evidence is seen of chronic AV insufficiency, which suggests that, in the unstressed animal at least, cardiac function is maintained until almost the very end. There was thus a great need for a reliable test of cardiac function in research on cardiotoxic plants. This need in our research has been very ably filled by the cardiopulmonary flow index (CPFI), a procedure developed at the Potchefstroom University for Christian Higher Education.263, 264 The CPFI can be defined as the ratio of the cardiopulmonary blood volume to stroke volume. Since this ratio is equivalent to the number of heartbeats necessary to pump blood from the right to the left side of the heart through the lungs, the CPFI can be calculated by counting the number of heartbeats between the two peaks of a radiogram. A bolus of the radio-isotope 99mTc (technetium 99m pertechnetate) is injected into the jugular vein and the flow of radioactivity through the heart is recorded with a sodium iodide crystal and collimator system. Two peaks of radioactivity are manifested during the passage of the bolus through the heart; the first peak is recorded immediately after injection, when the isotope enters the right side of the heart, and the second when the isotope re-enters the heart on the left side after passing through the lungs. The radiocardiogram and ECG are recorded simultaneously by a two-channel writing device. The CPFI, which is c.7 in normal sheep, rapidly rises up to 50 in the acute, terminal phase of gousiekte.263, 264 CPFI has been extensively used to monitor cardiac function in studies of the pathogenesis of gousiekte.
The pathogenesis of gousiekte has not been fully elucidated, though lately some progress has been made in this direction. The clinical signs and haemodynamic changes that appear in the last two weeks of latency have to some extent been investigated195, 196 but little data was available on the changes that take place in the myocardium during the major part of the latent period prior to the appearance of these signs. According to published reports, in gousiekte there is a decrease in the production and utilization of energy by the myocytes223, 226 accompanied by depletion of myosin, both of which detrimentally affect the contractility of the myocardium. Notable decreases in the contractility of the heart muscle of affected sheep have indeed been demonstrated with the aid of ultra-low frequency ballistocardiograms and aorta flow recordings.195, 196 These findings lend credence to the hypothesis that the primary lesion of gousiekte is inhibition of the contractile mechanism of the entire myocardium, a view strongly supported by the fact that pavetamine inhibits the formation of protein in the heart.214
Gousiekte, as it is induced in feeding trials, has been described as a congestive heart failure on account of the clinical signs, haemodynamic changes and certain lesions, notably oedema of the lungs and effusions in the body cavities. The congestive heart failure almost always follows an acute course, overt clinical signs of cardiac dysfunction seldom appearing more than a day or two before death. Chronic effects of congestive heart failure, such as centrilobular necrosis and induration of the liver are, with rare exception, absent both in field and laboratory cases of gousiekte, even when the hearts are grossly dilated. The histopathological features in experimentally induced gousiekte can differ to some extent from those of natural poisoning. For instance, in laboratory cases the fibrosis or round-cell infiltration can sometimes be much reduced or even be absent and in them the only evident changes may be dissociation of myofibrils, atrophy of the myofibres and fibres with enlarged bizarre-shaped nuclei (L. Prozesky, ARC-VRI, Onderstepoort, unpublished data, 1986). Sheep affected by gousiekte in the field, on the other hand, often drop dead without premonitory signs, sometimes without macroscopical lesions of heart failure. Death can be so sudden that a bolus of unchewed grass is found in the mouth. A case has even been reported where an affected steer was resuscitated by cardiac massage after ostensibly falling dead in a crush pen (P.W. Nel, ARC-VRI, Onderstepoort, personal communication, 1986). These signs of gousiekte in naturally affected animals are inconsistent with congestive heart failure and more in line with sudden cardiac arrest. It is thought that the pronounced myocardial fibrosis in natural poisoning may play a part in the genesis of the supposed conduction disturbances leading to the cardiac arrest. According to our current understanding of gousiekte then, the spectrum of clinical signs ranges from congestive heart failure to sudden cardiac arrest, and the lesions from hypertrophy (followed by atrophy) and/or necrosis of heart muscle to pronounced endocardial fibrosis and some round-cell infiltration of the interstitium.
Before the isolation of pavetamine82 the possibility was investigated that gousiekte was an autoimmune disease.81 The peculiar course of the disease, similarities with known immunopathological syndromes such as rheumatic fever, and the presence of anti-heart antibodies all pointed to the involvement of the immune system in the pathogenesis of gousiekte. The results of this research, however, showed that while factors against heart muscle were present in some sheep during latency, in others they were absent. Initial positive results were ascribed to cross-reacting antibodies against Eperythrozoon ovis.
Poisoning by cardiac glycoside-containing plants must be distinguished from poisoning induced by ionophore-antibiotics in stock feeds. Certain ionophores, such as monensin, are widely used as coccidiostats for broilers and growth promotants in ruminants. These compounds are capable of forming lipid-soluble, dynamically reversible complexes with monovalent and divalent cations, a property that enables them to transport cations across biological membranes. The changes in intra- and extracellular ion concentrations and electrical potentials brought about by ionophore-induced transport across cell membranes accounts for their pharmacological or toxicological effects. Obvious parallels can be drawn between ionophore-antibiotics and cardiac glycosides: both owe their activity to disturbance of the transportation of cations across cell membranes, cardiac glycosides by inhibition of sodium, potassium ATP-ase and ionophores by forming complexes with cations. In view of these similarities in their actions, it is not surprising that ECG changes indistinguishable from those of cardiac glycoside poisoning, were recorded in a horse that was acutely poisoned by salinomycin (P.W. Nel, R. Anitra Schultz and T.S. Kellerman, ARC-VRI, Onderstepoort, unpublished data, 1985). The myocardial changes induced by ionophores can be very similar to those of cardiac glycosides, but the lesions in the skeletal muscles of animals poisoned by ionophores are lacking in cardiac glycoside intoxication (J.A.W. Coetzer, ARC-VRI, Onderstepoort, personal observation, 1985).24
The factors that govern the toxicity and cumulativity of bufadienolides can only be speculated upon. Recently, two bufadienolides isolated by Anderson from Drimia physodes were found to be non-toxic to guinea-pigs at doses of up to 100 mg/kg. Nuclear resonance studies on these non-toxic bufadienolides indicated that the hydroxyl group on C-14 was lacking (L.A.P. Anderson, ARC-OVI, unpublished data, 1986). The striking chemical characteristics of the cumulative bufadienolides isolated from the Crassulaceae [cotyledoside (Figure 70), tyledosides (Figure 72), orbicusides (Figure 71), and lanceotoxins (Figure 73)] are that they occur in the glycosidic form and that the sugar moieties cannot be cleaved by acid hydrolysis. In addition, the sugar moieties are invariably laevorotatory or derived from a laevorotatory sugar.165
In conclusion, it is well to remember that not only plants contain cardiotoxic glycosides. Toads of the Bufonidae, notably the central American sea toad, Bufo marinus, secrete, among other active substances, cardioactive toxins in their skin glands to protect themselves against predators51, 129 (Figure 4.118). These toxins are bufadienolides, derived by biotransformation of cholesterol158 but unlike cardiac glycosides from plants, the A-ring of the aglycone is linked to suberyl arginine at the C-3 position. Mortalities occur in dogs that mouth this toad. The toxicity of the toad is ascribed to the additive effect of this toxin and especially catecholamines in the milky secretion of the large parotoid and other skin glands. In South Africa,11 Bufo species have been identified.184, 185 Dogs that play with some of these toads salivate profusely, soon learn to avoid them and occasionally die.164, 184 The greater fatalities experienced in dogs playing with B. marinus abroad, appears to be associated with the amount of toxin secreted by it. Several mortalities, especially in small dogs, have recently been positively linked to Bufo rangeri (C. Deacon, George, personal communication, 1994), B. pardalis (H. Currie, Cape Town, personal communication, 1996) and Schismaderma carens (M. Williams, Faculty of Veterinary Science, Onderstepoort, personal communication, 1996) in South Africa. These and the two other local toads in this category, B. gutturalis and B. garmani, are relatively large animals. Cardiac glycoside activity has been determined by the FPIA technique in the parotoid or the skin gland secretions of all five these toads (R.A. Schultz and T.W. Naudé, ARC-OVI, unpublished data, 1996). Pantonowitz, Naudé and Leisewitz (1998)184 have succinctly reviewed the literature on the noxious toads and frogs of South Africa.
Some insects can accumulate cardiac glycosides in their bodies from the milkweeds and other Apocynaceae on which they feed, as a defence against predators. Since the cardenolides induce severe vomition, birds soon learn to avoid these insects on sight after one or more emetic experiences.40 Appreciable quantities of several cardenolides have been demonstrated in the haemolymph and tissues of Danaus plexippus, the American counterpart of D. chrysippus, our Monarch butterfly, or ‘Melkbosskoenlapper’ (Figure 119), as it is known locally,202 as well as in other species.207 The glycosides corresponded with those of the asclepiad plants on which the caterpillars had fed. Individual butterflies were shown by Reichstein et al.202 to contain 0,2 mg of the glycoside ‘calatin’, or the equivalent of 1,8 times the intravenous LD for cats.
In 1962, Steyn reported the case of a child who had died of heart failure soon after eating an aposematically coloured grasshopper, Phymateus leprosus, the well-known garden ‘stinksprinkaan’. As a similar condition could be induced by administering minced grasshopper to rabbits, he concluded that either the grasshoppers were toxic per se or that they had acquired their toxicity by feeding on cardenolide-containing plants, such as Nerium oleander or Gomphocarpus fruticosus (=Asclepias fruticosa)248 (Figure 120). Poekilocerus bufonius, a North African grasshopper, suspected of feeding exclusively on asclepiad plants, was subsequently shown to contain cardenolides by Von Euw and his co-workers.287 This species is provided with a bilobed poison gland dorsally between the first and second terga, from which a pungent, sticky secretion can be squirted out for up to 0,6 m. The secretion flowing down the sides of the adult grasshopper is also transformed into a venomous foam by bubbles of air passing through it from the spiracles. Both the haemolymph and the secretion contain several cardiac glycosides derived from the plants on which the grasshopper had fed.
References
- ADELAAR, T.F. 1936. Gousiekte in the Waterberg district. Journal of the South African Veterinary Medical Association, 7, 51–53.
- ADELAAR, T.F. & TERBLANCHE, M. 1967. A note on the toxicity of the plant Pachystigma thamnus Robyns. Journal of the South African Veterinary Medical Association, 38, 25–26.
- ADELAAR, T.F., TERBLANCHE, M. & SMIT, J.D. 1966. A report on negative experiments with ferric chloride as a prophylactic agent against gousiekte. Journal of the South African Veterinary Medical Association, 37, 199–201.
- AEBI, A. & REICHSTEIN, T. 1950. Über die Glykoside der Blätter von Cryptostegia grandiflora (Roxb.) R. Br. (Asclepiadaceae). Helvetica Chimica Acta, 33, 1013–1034.
- ANDERSON, L.A.P. 1969. ‘n Chemiese ondersoek van inhoudstowwe van Melianthus comosus Vahl. Tesis vir die graad Doctor Scientae: Fakulteit van Natuurwetenskappe, Universiteit van die Oranje Vrystaat, Bloemfontein.
- ANDERSON, L.A.P. & DE KOCK, W.T. 1959. The occurrence of ursolic acid in several plants reported to be responsible for ‘Gousiekte’ in South Africa. Journal of the South African Chemical Institute, 12, 114–115.
- ANDERSON, L.A.P., JOUBERT, J.P.J., PROZESKY, L., KELLERMAN, T.S., SCHULTZ, R.A., PROCOS, J. & OLIVIER, P.M. 1983. The experimental production of krimpsiekte in sheep with Tylecodon grandiflorus (Burm. f.) Toelken and some of its bufadienolides. Onderstepoort Journal of Veterinary Research, 50, 301–307.
- ANDERSON, L.A.P. & KOEKEMOER, J.M. 1968. The chemistry of Melianthus comosus Vahl. Part II. The isolation of hellibregenin 3–acetate and other unidentified bufadienolides from the rootbark of Melianthus comosus Vahl. Journal of the South African Chemical Institute, 21, 155–158.
- ANDERSON, L.A.P. & KOEKEMOER, J.M. 1969. The chemistry of Melianthus comosus Vahl. Part III. The constitution of melianthugenin and melianthusigenin, new bufadienolides from the rootbark. Journal of the South African Chemical Institute, 22, 191–197.
- ANDERSON, L.A.P. & KOEKEMOER, J.M. 1969. Toxic bufadienolides from Melianthus comosus Vahl. Journal of the South African Chemical Institute, 22, 119–124.
- ANDERSON, L.A.P., SCHULTZ, R.A., JOUBERT, J.P.J., PROZESKY, L., KELLERMAN, T.S., ERASMUS, G.L. & PROCOS, J. 1983. Krimpsiekte and acute cardiac glycoside poisoning in sheep caused by bufadienolides from the plant Kalanchoe lanceolata Forssk. Onderstepoort Journal of Veterinary Research, 50, 295–300.
- ANDERSON, L.A.P., SCHULTZ, R A, KELLERMAN, T.S., KOTZÉ, S.M., PROZESKY, L., ERASMUS, G.L. & LABUSCHAGNE, L. 1985. Isolation and characterization of and some observations on poisoning by bufadienolides from Cotyledon orbiculata L. var. orbiculata. Onderstepoort Journal of Veterinary Research, 52, 21–24.
- ANDERSON, L.A.P., STEYN, P.S. & VAN HEERDEN, F.R. 1984. The characterization of two novel bufadienolides, Lanceotoxins A and B from Kalanchoe lanceolata (Forssk.) Pers. Journal of the Chemical Society Perkin Transactions, 1573–1575.
- ANON. 1915. Report on Acokanthera venenata G. Don, from the Transvaal. Report on Veterinary Research, Union of South Africa, 384, 617–623.
- APLIN, T.E.H. 1967. Poison plants of Western Australia. Toxic species of genera Gastrolobium and Oxylobium. Journal of Agriculture of Western Australia, 8, 42–52, 200–206, 241–243, 408–414.
- APLIN, T.E.H. 1968. Poison plants of Western Australia. Toxic species of genera Gastrolobium and Oxylobium. Journal of Agriculture of Western Australia, 9, 69–74, 356–362.
- ARCHER, C. & ARCHER, R.H. 1999. A new species of Ornithogalum subgenus Urophyllon (Hyacinthaceae) from central South Africa and southern Namibia. South African Journal of Botany, 65, 431–433.
- ARNOLD, T.H. & DE WET, B.C. 1993. Plants of Southern Africa. Names and Distribution. Brummeria: National Botanical Institute.
- ARSHAMI, J. & RUTTLE, J.L. 1988. Effects of diets containing gossypol on spermatogenic tissue of young bulls. Theriogenology, 507–516.
- BAMHARE, C. 1998. Suspected cardiac glycoside intoxication in sheep and goats in Namibia due to Ornithogalum nanodes (Leighton). Onderstepoort Journal of Veterinary Research, 65, 25–30.
- BARNES, J.E. 1958. Georgina poisoning of cattle in the Northern Territory. Australian Veterinary Journal, 34, 281–290.
- BASSON, P.A., NORVAL, A.G., HOFMEYR, J.M., EBEDES, H. & SCHULTZ, R.A. 1982. Antelope and poisonous plants: 1. Gifblaar Dichapetalum cymosum (Hooker) Engler & Prantl containing monofluoroacetate. Madoqua, 13, 59–70.
- BASSON, P.A., NORVAL, A.G., HOFMEYR, J.M., EBEDES, H., SCHULTZ, R.A., KELLERMAN, T.S. & MINNE, J.A. 1984. Antelopes and poisonous plants. Proceedings 13th World Congress on Diseases of Cattle, World Biuatrics Association, Durban, 17–2 September 1984.
- BASTIANELLO, S.S., FOURIE, N., PROZESKY, L., NEL, P.W. & KELLERMAN, T.S. 1995. Cardiomyopathy of ruminants induced by the litter of poultry fed on rations containing the ionophore antibiotic, maduramycin. II. Macropathology and histopathology. Onderstepoort Journal of Veterinary Research, 5–8.
- BELL, A.T., NEWTON, L.G., EVERIST, S.L. & LEGG, J. 1955. Acacia georginae poisoning of cattle and sheep. Australian Veterinary Journal, 31, 249–257.
- BERARDI, L. C. & GOLDBLATT, L. A. 1969. Gossypol. In LIEVER, I.C. (ed.) Constituents of Foodstuff. London: Academic Press.
- BOTHA, C.J. 2003. Krimpsiekte, a paretic/paralytic syndrome of small stock, in South Africa: Studies on the aetiology of krimpsiekte, the toxicokinetics and dynamic effects of cotyledoside and seasonal fluctuation of cotyledoside concentration in plant material. Thesis for the degree Doctor Philosophiae: The Norwegian School of Veterinary Science, Oslo.
- BOTHA, C.J., GEHRING, R., VAN ROOYEN, J.M. & VENTER, D. 2002. The effect of three bufadienolide cardiac glycosides on contraction of isolated rat jejunum. Onderstepoort Journal of Veterinary Research, 69, 243–246.
- BOTHA, C.J., KELLERMAN, T.S., SCHULTZ, R.A., ERASMUS, G.L., VLEGGAAR, R. & RETIEF, E. 1998. Krimpsiekte in sheep following a single dose of Tylecodon ventricosus (Burm.f.) Toelken and the isolation of tyledoside D from this plant species. Onderstepoort Journal of Veterinary Research, 17–23.
- BOTHA, C.J., NAUDÉ, T.W., MOROE, M.L. & ROTTINGHAUS, G.E. 2004. Gangrenous ergotism in cattle grazing fescue (Festuca elatior L.) in South Africa. Journal of the South African Veterinary Association, 75, 45–48.
- BOTHA, C.J., RUNDBERGET, T., WILKINS, A.L., MÜLDERS, M.S.G., FLÅØYEN, A. & VAN AARDT, M.P. 2000. Seasonal variation in cotyledoside concentration of Tylecodon wallichii (Harv.) Tölken subsp. wallichii sampled in a krimpsiekte-prevalent region. Onderstepoort Journal of Veterinary Research, 68, 1–9.
- BOTHA, C.J., SCHULTZ, R.A., VAN DER LUGT, J.J. & ARCHER C. 2000. A krimpsiekte-like syndrome in small stock poisoned by Ornithogalum toxicarium. Journal of the South African Veterinary Association, 71, 6–9.
- BOTHA, C.J., VAN DER LUGT, J.J., ERASMUS, G.L., KELLERMAN, T.S., SCHULTZ, R.A. & VLEGGAAR, R. 1997. Krimpsiekte, associated with thalamic lesions induced by the neurotoxic cardiac glycoside, cotyledoside, isolated from Tylecodon wallichii (harv.) Toelken subsp. wallichii. Onderstepoort Journal of Veterinary Research, 64, 189–194.
- BOTHA, C.J., VAN DER LUGT, J.J., ERASMUS, G.L., KELLERMAN, T.S., SCHULTZ, R.A. & VLEGGAAR, R. 1998. Krimpsiekte, a paretic condition of small stock by bufadienolide- containing plants of the Crassulaceae in South Africa. In: GARLAND, T. & BARR, C.A. (eds.) Plants and other natural toxicants, 407–412: CAB International.
- BOURDON, R. & MERCIER, M. 1969. Dosage des hétérosides cardiottoniques dans les liquides Biologiques par spectrophotometrie d’absorption atomique. Annales de Biologie Clinique, 27, 651–657.
- BRAIN, C. & FOX, V.E.B. 1994. Suspected cardiac glycoside poisoning in elephants (Loxodonta africana). Journal of the South African Veterinary Association, 64, 173–174.
- BRETELER, F.J. 1986. The African Dichapetalaceae. IX. A taxonomical revision. Agricultural School of Wageningen Papers, 86–3, 1–74.
- BRINK, A.J., LEWIS, C.M. & WEBER, H.W. 1965. Myocardiopathy in Argemone mexicana poisoning. South African Medical Journal, 39, 108–114.
- BROCAS, C., RIVERA, R.M., PAULA-LOPEZ, F.F., MC DOWELL, L.R., CALHOUN, M.C., STAPLES, C.R., WILKINSON, N.S., BONING, A.J., CHENOWETH, P.J. & HANSEN, P.J. 1997. Deleterious actions of gossypol on bovine spermatozoa, oocytes, and embryos. Biology of Reproduction, 901–907.
- BROWER, L.P. & MOFFIT, C.M. 1974. Palatability dynamics of cardenolides in the monarch butterfly. Nature, 249, 280–283.
- BUORO, I.B.J., NYAMWANGE, S.B., CHAI, D. & MUNYUA, S.M. 1994. Putative avocado toxicity in two dogs. Onderstepoort Journal of Veterinary Research, 61, 107–109.
- BURGER, W.P., NAUDÉ, T.W., VAN RENSBURG, I.B.J., BOTHA, C.J. & PIENAAR, A.C.E. 1994. Cardiomyopathy in ostriches (Struthio camelus) due to avocado (Persea americana var. gautemalensis) intoxication. Journal of the South African Veterinary Association, 65, 113–118.
- BURTT-DAVY, J. 1904. Plant poisonous and otherwise injurious to stock. Annual Report of the Director of Agriculture, Transvaal, pp. 273–274.
- BURTT-DAVY, J. 1904. The botanical section. 8. Correspondence. Transvaal Agricultural Journal, 3, 303–319.
- BURTT-DAVY, J. 1912. Botanical investigation into galsiekte. Report on Veterinary Research, Union of South Africa, 2, 181–221.
- BURTT-DAVY, J. & STENT, S.M. 1908. Notes from the economic herbarium. Transvaal Agricultural Journal, 7, 652–657.
- BUTTON, C. & MÜLDERS, M.S.G. 1984. Further physiopathological features of experimental Homeria glauca (Wood & Evans) N.E. Br. poisoning in Merino sheep. Onderstepoort Journal of Veterinary Research, 51, 95–96.
- BUTTON, C. & MÜLDERS, M.S.G. 1984. Physiological features of experimental plant-related cardiac glycoside intoxication in sheep. Proceedings 13th World Congress on Diseases of Cattle, World Biuatrics Association, Durban. 17–2 September 1984, 675–680.
- BUTTON, C., REYERS, F., MELTZER, D.G.A., MÜLDERS, M.S.G. & KILLEEN, V.M. 1983. Some physiological features of experimental Homeria glauca (Wood & Evans) N.E. Br. Poisoning in Merino sheep. Onderstepoort Journal of Veterinary Research, 50, 191–196.
- CHEEKE, P.R. 1998. Natural toxicants and feeds, forages and poisonous plants. 2nd edn. Danville, Illinois: Interstate Publishers, Inc.
- CHEN, K.K. & KOVARIKOVA, A. 1967. Pharmacology and toxicology of toad venom. Journal of Pharmaceutical Science, 56, 1535–1541.
- CHENOWETH, M.B. 1949. Monofluor acetic acid and related compounds. Pharmacological Review, 1, 383–424.
- CHENOWETH, M.B. & GILMAN, A. 1946. Studies on the pharmacology of fluoroacetate. 1. Species response to fluoroacetate. Journal of Pharmacology and Experimental Therapeutic, 87, 90–100.
- CHEUNG, K., HINDS, J.A. & DUFFY, P. 1989. Detection of poisoning by plant-origin cardiac glycosides with the Abott TDX analyzer. Clinical Chemistry, 35, 295–297.
- CHRISTENSEN, C.M. 1980. Conference on mycotoxins in animal feeds and grains related to animal health. Rockville, Maryland, USA.
- CHUNG, E.K. 1971. Guide to managing digitalis intoxication. Post-graduate Medicine, 49, 99–101.
- CHUNG, H.M. 1984. Acute renal failure caused by acute monofluoroacetate poisoning. Veterinary and Human Toxicology, 26, 29–32.
- CLARK, R. 1952. Studies on the pharmacology and toxicology of Acovenoside A. Onderstepoort Journal of Veterinary Research, 25, 69–74.
- CLARKE, M.L., HARVEY, D.G. & HUMPHREYS, D.J. 1981. Veterinary Toxicology. 2nd edn. London: Bailliere Tindall.
- COATES-PALGRAVE, K. 1981. Trees of Southern Africa. Cape Town: Struik Publishers.
- CODD, L.E. 1961. Notes on poisonous plants, with special reference to the gousiekte problem. Journal of the South African Biological Society, 2, 8–17.
- CODD, L.E. & VOORENDYK, S. 1966. Plante wat gousiektevergiftiging veroorsaak. Bothalia, 8, 47–58.
- COETZEE, G. 1975. Oorsig oor navorsing wat uitgevoer is op Pachystigma pygmaeum (Gousiektebossie). Vergadering van die Grasveldwerkspan, Potchefstroom, 24–25 Maart, 1975.
- COLLIN-NEGRETE, J., KIESLING, H.E., ROSS, T.T. & SMITH, J.F. 1996. The effect of whole cottonseed on serum constituents, fragility of erythrocyte cells and reproduction of growing Holstein heifers. Journal of Dairy Science, 2016–2023.
- CRAIGMILL, A.L., EIDE, R.N., SHULTZ, T.A. & HEDRICK, K.R.N. 1994. Toxicity of avocado [Persea americana (Guatamalan var.)] leaves: Review and preliminary report. Veterinary and Human Toxicology, 26, 381–383.
- CRAIGMILL, A.L., SEAWRIGHT, A.A., MATILLA, T. & FROST, A.J. 1989. Pathological changes in the mammary gland and biochemical changes in milk of the goat following oral dosing with leaf of the avocado (Persea americana). Australian Veterinary Journal, 66, 206–211.
- CURSON, H.H. 1926. Some little known South African poisonous plants and their effects upon stock. Tropical Veterinary Bulletin, 14, 79–80.
- CURSON, H.H. 1928. Note on Bowiea volubilis Harv. South African Journal of Science, 25, 240–241.
- CURSON, H.H. 1928. Some little known South African poisonous plants and their effect on stock. Report on Veterinary Research, Union of South Africa, 13 & 14, 205–229.
- CUSACK, P.M.V. & PERRY, V. 1995. The effect of feeding whole cotton seed on the fertility of bulls. Australian Veterinary Journal, 463–466.
- DE OLIVEIRA, M.M. 1968. Chromatographic isolation of monofluoroacetic acid from Palicourea marcgravii St. Hil. Experientia, 19, 586–587.
- DRY, L.J. 1950. A preliminary investigation of the toxic principle of Moraea polystachya. MSc thesis, Rhodes University College, Grahamstown.
- DUNPHY, J.T. 1906. Report of experiments carried out to observe effects of certain poisonous plants on sheep and goats. Transvaal Agricultural Journal, 4, 315–323.
- DU PLOOY, W.J. & BROWN, J.M.M. 1984. Lactate dehydrogenase isoenzymes during congestive heart failure in sheep. Proceedings of the Annual Congress of the Pharmacological and Physiological Societies of Southern Africa, Potchefstroom. October 3 to November 2, 1984.
- EAST, N.E., ANDERSON, M. & LOWENSTINE, L.J. 1994. Apparent gossypol-induced toxicosis in adult dairy goats. Journal of the American Veterinary Medical Association, 642–643.
- EGYED, M.N. & MILLER, G.W. 1971. Experimental acute fluoroacetamide poisoning in guinea-pigs and sheep. Fluoride., 4, 137–142.
- EGYED, M.N. & SCHULTZ, R.A. 1986. The efficacy of acetamide for treatment of experimental Dichapetalum cymosum (gifblaar) poisoning in sheep. Onderstepoort Journal of Veterinary Research,53, 231–234.
- ENSLIN, P.R., NAUDÉ, T.W., POTGIETER, D.J.J. & VAN WYK, A.J. 1966. 1–alpha, 2–alpha–Epoxyscillirosidine, the main toxic principle of Homeria glauca (Wood & Evans) N.E. Br. Tetrahedron, 22, 3213–3220.
- FANSHIER, Z.W., GOTTWALD, L.K. & KUN, E. 1964. Studies on specific enzyme inhibitors. VI. Characterization and mechanism of action of the enzyme-inhibitory isomer of monofluorocitrate. The Journal of Biological Chemistry, 239, 425–434.
- FERNANDES, R. 1983. Kalanchoe. Flora Zambesica, 41–66.
- FOURIE, N. 1994. Isolation of the cardiotoxin from gousiekte-inducing plants and investigation of the pathogenesis and diagnosis of the disease. PhD Thesis, University of Pretoria.
- FOURIE, N., ERASMUS, G.L., SCHULTZ, R.A. & PROZESKY, L. 1994. Isolation of the toxin responsible for gousiekte, a plant-induced cardiomyopathy of ruminants in southern Africa. Onderstepoort Journal of Veterinary Research, 77–87.
- FOURIE, N., SCHULTZ, R.A., PROZESKY, L., KELLERMAN, T.S. & LABUSCHAGNE, L. 1989. Clinical pathological changes in gousiekte, a plant-induced cardiotoxicosis of ruminants. Onderstepoort Journal of Veterinary Research, 56, 73–80.
- GAMBILL, M.D. & HUMPHREY, W.D. 1993. Effects of diets containing gossypol on ovarian histology, function and fertility in prepubertal beef heifers. Theriogenology, 585–593.
- GEORGE, E. 1925. The active principles of the African squill Urginea burkei (slangkop) and its suitability or otherwise as a substitute for Urginea maritima in commerce. Journal of the South African Chemical Institute, 8, 14–27.
- GILMAN, R.G., GOODMAN, L.S., RALL, T.W. & MURAD, F. 1985. Goodman and Gilman’s The Pharmacological Basis of Therapeutics. (eds. R.G. Gilman, L.S. Goodman, T.W. Rall & F. Murad.) 7th edn. New York: Macmillan Publishing Co. Inc.
- GLYNN, I.M. 1964. The action of cardiac glycosides on ion movements. Pharmacological Review, 16, 381–407.
- GOLDMAN, P. 1969. The carbonfluorine bond in compounds of biological interest. Science, 164, 1123–1130.
- GRANT, R., BASSON, P.A., BOOKER, H.H., HOFHERR, B. & ANTHONISSEN, M. 1991. Cardiomyopathy caused by avocado (Persea americana Mill.) leaves. Journal of the South African Veterinary Association, 62, 21–22.
- GRAY, M.L., GREENE, L.W. & WILLIAMS, G.L. 1993. Effects of dietary gossypol on metabolic homeostasis and reproductive endocrine function in beef heifers and cows. Journal of Animal Science, 3052–3059.
- GUNN, J.W.C. 1921. The action of Urginea burkei (Baker). Transactions of the Royal Society of South Africa, 9, 197–204.
- GUNN, J.W.C. 1924. Slangkop poisoning. Journal of the Department of Agriculture, Union of South Africa, 9, 141–142.
- GUNN, J.W.C. 1932. The action of Homeria bulbifera and Homeria aurantianica. Suid-Afrikaanse Tydskrif vir Geneeskunde, 6, 461.
- GUNN, J.W.C. & BROWN, H.A. 1932. The action of Homeria lucasii. South African Medical Journal, 6, 459–461.
- GUYTON, A.C. 1981. Textbook of Medical Physiology. 6th edn. Philadelphia, London, Toronto, Tokyo: W.B. Saunders Company.
- HARGIS, A.M., STAUBER, E., CASTEEL, S. & EITNER, D. 1989. Avocado (Persea americana) intoxication in caged birds. Journal of the American Veterinary Medical Association, 62, 64–66.
- HARPER, D.B. & O’HAGAN, D. 1994. The fluorinated natural compounds. Natural Products Report 1994, 123–133.
- HASCHEK, W.M., BEASLEY, V.R., BUCK, W.B. & FINNELL, J.H. 1989. Cottonseed meal (gossypol) toxicosis in a swine herd. Journal of the American Veterinary Medical Association, 613–615.
- HENDERSON, M. & ANDERSON, J.G. 1966. Common weeds in South Africa. Botanical Survey No. 37. Department of Agricultural Technical Services.
- HENNING, M.W. 1926. Krimpsiekte. Report on Veterinary Research, Union of South Africa, 12, 331–365.
- HENNING, M.W. 1932. Animal diseases in South Africa. South African Agricultural Series, XIII Central News Agency, Ltd.
- HOFFMANN, S., WEISS, E. & REICHSTEIN, T. 1966. Adigosid. Strukturbestimmung. Helvetica Chimica Acta, 49, 1855–1872.
- HUNGER, A. & REICHSTEIN, T. 1952. Die Konstitution von Gofrusid und Frugosid. Helvetica Chimica Acta, 35, 1073–1103.
- HURTER, L.P., NAUDÉ, T.W., ADELAAR, T.F., SMIT, J.D. & CODD, L.E. 1972. Ingestion of the plant Fadogia monticola Robyns as an additional cause of gousiekte in ruminants. Onderstepoort Journal of Veterinary Research, 39, 71–82.
- HUTCHEON, D. 1899. Nenta. Agricultural Journal of the Cape of Good Hope, 14, 862–873.
- HUTCHEON, D. 1900. The poisoning of stock by tulp (Moraea polystachya). The Agricultural Journal of the Cape of Good Hope, 17, 84–89.
- HUTCHEON, D. 1903. The poisoning of stock. Agricultural Journal of the Cape of Good Hope, 23, 390–399.
- HUTCHEON, D. 1904. Cattle poisoning by ‘Klimop’ Cynoctomum capense. Agricultural Journal of the Cape of Good Hope, 24, 398–401.
- JACOBS, W.A. & HOFFMANN, A. 1926. The structural relationship of cardiac poisons. Journal of Biological Chemistry, 67, 333–339.
- JESSOP, J.P. 1970. Studies on the bulbous Liliaceae I. Scilla, Schizocarpus and Ledebouria. Journal of South African Botany, 36, 233–266.
- JIMENEZ, D.A., CHANDLER, J.E., ATKINSON, R.W., NIPPER, W.A., BAHMAN, W.A. & SAXTON, A.M. 1989. Effect of feeding gossypol in cottonseed on growth, semen quality and spermatogenesis of yearling Holstein bulls. Journal of Dairy Science, 1866–1875.
- JONES, T.C. & HUNT, R.D. 1983. Veterinary Pathology. 5th edn. Philadelphia: Lea & Febiger.
- JOUBERT, J.P.J. 1981. Die effek van hartglikosiede en elektroliete op die selmembraan se natrium, kalium-adenosientrifosfatase. Journal of the South African Veterinary Association, 52, 225–228.
- JOUBERT, J.P.J. & SCHULTZ, R.A. 1982. The minimal effective dose of activated charcoal in the treatment of sheep poisoned with the cardiac glycoside containing plant Moraea polystachya (Thunb) Ker-Gawl. Journal of the South African Veterinary Association, 53, 265–266.
- JOUBERT, J.P.J. & SCHULTZ, R.A. 1982. The treatment of Moraea polystachya (Thunb) Ker-Gawl (cardiac glycoside) poisoning in sheep and cattle with activated charcoal and potassium chloride. Journal of the South African Veterinary Association, 53, 249–253.
- JOUBERT, J.P.J. & SCHULTZ, R.A. 1982. The treatment of Urginea sanguinea Schinz poisoning in sheep with activated charcoal and potassium chloride. Journal of the South African Veterinary Association, 53, 25–28.
- JUBB, K.V.F., KENNEDY, P.C. & PALMER, N. 1985. Pathology of domestic animals. 3rd edn. Orlando, Florida: Academic Press, Inc.
- JURITZ, C.F. 1915. South African plant poisons and their investigation. Report of the 12th meeting of the South African Association for Advanced Science, Kimberley, 109–145.
- KAMAU, J.A., GACHUHI, D. M., GRYD-HANSEN, N. & GATUMA, J.M. 1978. A study of the toxicity of Dichapetalum ruhlandi (Ludi). Indian Veterinary Journal, 55, 626–630.
- KAMERMAN, P. 1926. The toxicity of the genus Cotyledon, the cause of the disease krimpsiekte. South African Journal of Science, 23, 185–187.
- KAMERMAN, P. 1932. The toxic principle of Cotyledon orbiculata. Thesis in fulfilment of requirements for the degree Doctor Science, Faculty of Science, University of Pretoria.
- KATSUNG, B.G. 1995. Basic and clinical pharmacology. 6th edn. Norwalk, Connecticut: Appleton & Lange.
- KATZ, A. 1954. Über die glykoside von Bowiea volubilis Harvey. Helvetica Chimica Acta, 37, 832–836.
- KECK, R., HAAS, H. & RÈTEY, J. 1980. Synthesis of stereospecifically deuterated fluoroacetic acid and its behaviour in enzymic aldol-type condensations. FEBBS Letters, 114, 287–290.
- KEHOE, D. 1912. Preliminary note on the poisonous properties of Cotyledon orbiculata. Report on Veterinary Research, Union of South Africa, 2, 387–397.
- KELLERMAN, T.S., NAUDÉ, T.W. & FOURIE, N. 1996. The distribution, diagnosis and estimated economic impact of plant poisonings and mycotoxicoses in South Africa. Onderstepoort Journal of Veterinary Research, 63, 65–90.
- KINGSBURY, J.M. 1964. Poisonous plants of the United States and Canada. Englewood Cliffs, New Jersey:Prentice Hall, Inc.
- KIRSTEN, E., SHARMA, K.L. & KUN, E. 1978. Molecular toxicology of (-)-erythrofluorocitrate: selective inhibition of citrate transport in mitochondria and binding of fluorocitrate to mitochondrial proteins. Molecular Pharmacology, 14, 172–184.
- KNOWLES, P.R. 1964. The poison toad and the canine. Veterinary Medicine, 59, 39–42.
- KOEKEMOER, J.M., ANDERSON, L.A.P. & PACHLER, K.G.R. 1970. The chemistry of Melianthus comosus Vahl. Part IV. 6ß-Acetoxymelianthugenin a new toxic principle from the rootbark. Journal of the South African Chemical Institute, 23, 146–153.
- KOEKEMOER, J.M., ANDERSON, L.A.P. & PACHLER, K.G.R. 1971. The chemistry of Melianthus comosus Vahl. Part V. Two novel bufadienolides from the rootbark. Journal of the South African Chemical Institute, 24, 75–78.
- KRAMER, R.Y., GARNER, D.L., ERICSSON, S.A., WESSON, D.A. & DOWNING, T.W. 1991. The effect of cottonseed components on testicular developments in pubescent rams. Veterinary and Human Toxicology, 11–16.
- KUPPER, W. & BLEIFELD, W. 1979. Serum enzyme changes in patients with cardiac disease. In: SMIDT, E., SCHMIDT, F.W., TRAUTSCHOLD, I. & FRIEDLE, R. (eds.) Advances in Clinical Enzymology, 106–123. Basel: S. Krager.
- LALONDE, R.L., DESHPANDE, R., HAMILTON, P.P., MCLEAN, W.M. & GREENWAY, D.C. 1985. Acceleration of digoxin clearance by activated charcoal. Clinical Pharmacology and Therapeutics, 37, 367–371.
- LEEMANN, A.C. 1935. The eradication of gifblaar (Dichapetaum cymosum). Farming in South Africa, June 1935, 233–236.
- LETTY, C.L. 1961. Wild flowers of the Transvaal. Pretoria: Central News Agency Ltd.
- LICHTI, H. & VON WARTBURG, A. 1960. Herzaktive glykoside aus Urginea altissima Baker. Helvetica Chimica Acta, 43, 1666–1681.
- LOUW, P.G.J. 1949. The cardiac glycoside from Urginea rubella Baker. Part I. Isolation and properties of rubellin. Onderstepoort Journal of Veterinary Science and Animal Industry, 22, 313–314.
- LOUW, P.G.J. 1952. Transvaalin, a cardiac glycoside isolated from Urginea burkei Bkr. (Transvaal slangkop). Onderstepoort Journal of Veterinary Research, 25, 123–131.
- LOUW, P.G.J. 1953. A cardiac glycoside from Urginea rubella Baker. Part II. Hydrolysis of rubellin and pre–rubellin. Onderstepoort Journal of Veterinary Research, 26, 141–148.
- LOVELESS, A.R. 1976. A new species of Claviceps on Cyperaceae. Transactions of the British Mycological Society, 50, 19–22.
- LUBKE, R.A. & LUBKE, M.A. 1971. Scilla natalensis versus Ledebouria floribunda. Veterinary Clinician, 6, 14–17.
- MACKENZIE, A.T. 1910. A brief account of the pharmacological (or physiological) action of a Western Province tulp: Homeria collina (Vent.) var. minata. South African Medical Record,8, 94–95.
- MACKENZIE, R.A. & BROWN, O.P. 1991. Avocado (Persea americana) poisoning of horses. Australian Veterinary Journal, 68, 77–88.
- MACKENZIE, R.A. & DUNSTER, P.J. 1986. Hearts and flowers: Bryophyllum poisoning of cattle. Australian Veterinary Journal, 63, 222–227.
- MACKENZIE, R.A. & DUNSTER, P.J. 1987. Curing experimental Bryophyllum tubiflorum poisoning of cattle with activated carbon, electrolyte replacement and antiarrhythmic drugs. Australian Veterinary Journal, 64, 211–214.
- MACKENZIE, R.A., FRANKE, F.P. & DUNSTER, P.J. 1987. The toxicity to cattle and bufadienolide content of six Bryophyllum species. Australian Veterinary Journal, 64, 298–301.
- MACKENZIE, R.A., FRANKE, F.P. & DUNSTER, P.J. 1989. The toxicity for cattle of bufadienolide cardiac glycosides from Bryophyllum tubiflorum flowers. Australian Veterinary Journal, 66, 374–376.
- MARAIS, J.S.C. 1943. The isolation of the toxic principle ‘potassium cymonate’ from ‘Gifblaar’, Dichapetalum cymosum (Hook.) Engl. Onderstepoort Journal of Veterinary Science and Animal Industry, 18, 203–206.
- MARAIS, J.S.C. 1944. Monofluoroacetic acid, the toxic principle of ‘Gifblaar’ Dichapetalum cymosum (Hook.) Engl. Onderstepoort Journal of Veterinary Science and Animal Industry, 20, 67–73.
- MARLOTH, R. 1913. The flora of South Africa. Cape Town: Darter Bross & Co.
- MASVINGWE, C. & MAVENYENGWA, M. 1997. Kalanchoe lanceolata poisoning in Brahman cattle in Zimbabwe: The first field outbreak. Journal of the South African Veterinary Association, 68, 18–20.
- MCVANN, A., HAVLIK, I., JOUBERT, P.H. & MONTEAGUDO, F.S.E. 1992. Cardiac glycoside poisoning involved in deaths from traditional medicines. South African Medical Journal, 81, 139–141.
- MCGAVIN, M.D. 1966. Rubber-vine (Cryptostegia grandiflora) toxicity for ruminants. Queensland Journal of Agricultural Science, 26, 9–19.
- MCKINNEY, B. 1974. Gousiekte: Pathology of the cardiomyopathies. London: Butterworths.
- MES, M.G. & DE VILLIERS, E. 1944. Die gousiektebossie. ‘n Studie van die morfologie, fisiologie en uitroeiing van Pachystigma pygmaeum (Schl.) Robyns. Departement van Landbou en Bosbou, Landbounavorsingsreeks 4, Pamflet 245, 1–52.
- METTAM, R. W.M. 1933. Poisonous plants in East Africa. Kenya Agricultural Journal, Reprint from KAJ.
- MEYER, K. & LINDE, H. 1971. Collection of toad venoms and chemistry of the toad venom steroids, In: BÜCHEREL, W. & BUCKLEY, E.A. (eds.) Venomous animals and their venoms.New York: Academic Press.
- MINNAAR, P.P. 1997. A high performance liquid chromatographic method for the determination of monofluoroacetic acid from Dichapetalum cymosum and biological samples. M. Tech Dissertation, Technikon Pretoria.
- MITCHELL, D.T. 1926. The toxic effects on ruminants. Urginea macrocentra (Baker). Report on Veterinary Research, Union of South Africa, 1 & 12, 303–327.
- MITCHELL, D.T., CANHAM, A.S. & BAYER, A.M. 1934. Urginea capitata Baker – The berg slangkop. Its toxic effect on ruminants. Onderstepoort Journal of Veterinary Science and Animal Industry, 2, 681–689.
- MOGG, A. 1930. An aut-ecological note on the poisonous ‘Gifblaar’ (Dichapetalum cymosum) (Hook.) Engl. South African Journal of Science, 27, 368–376.
- NAUDÉ, T.W. 1977. The occurrence and significance of South African cardiac glycosides. Journal of the South African Biological Society, 18, 7–20.
- NAUDÉ, T.W. 1992. Bufo-intoxication in dogs. Proceedings of a Mini-symposium on Veterinary Aspects of Some Zootoxins in Southern Africa. Department of Pharmacology and Toxicology, Faculty of Veterinary Science, University of Pretoria.
- NAUDÉ, T.W., ANDERSON, L.A.P., SCHULTZ, R.A. & KELLERMAN, T.S. 1992. ‘Krimpsiekte’: A chronic paralytic syndrome of small stock caused by cumulative bufadienolide cardiac glycosides. In: JAMES, L.F., KEELER, R.F., BAILEY, E.M., CHEEKE, P.R. & MEGARTY, M.P. (eds.) Poisonous plants, Proceedings of the Third International Symposium, pp. 392–396. Ames, Iowa: Iowa State University Press.
- NAUDÉ, T.W., KELLERMAN, T.S. & COETZER, J.A.W. 1996. Plant poisonings and mycotoxicoses as constraints in livestock production in East Africa: The southern Africa experience. Journal of the South African Veterinary Association, 67, 8–11.
- NAUDÉ, T.W. & POTGIETER, D.J.J. 1966. A preliminary note on the isolation and pharmacological actions of the toxic principles of Homeria glauca (W & E). N.E. Br. Journal of the South African Veterinary Medical Association, 37, 73–75.
- NAUDÉ, T.W. & POTGIETER, D.J.J. 1971. Studies on South African cardiac glycosides. I. Isolation of toxic principles of Homeria glauca (W & E) N.E. Br. and observations on their chemical and pharmacological properties. Onderstepoort Journal of Veterinary Research, 38, 255–278.
- NAUDÉ, T.W. & SCHULTZ, R.A. 1982. Studies of South African cardiac glycosides. II. Observations on the clinical and haemodynamic effects of cotyledoside. Onderstepoort Journal of Veterinary Research, 49, 247–254.
- NEL, P.W., SCHULTZ, R.A., JORDAAN, P., ANDERSON, L.A.P., KELLERMAN, T.S. & REID, C. 1987. Cardiac glycoside poisoning in sheep caused by Urginea physodes (Jacq.) Bak. and the isolated physodine A. Onderstepoort Journal of Veterinary Science, 641–644.
- NESER, J.A., GRIMBEEK, P.J., LANGFORD, M.J. & NEL, P.W. 1988. Gossypol poisoning in pigs. Proceedings of a Workshop of the Pathology Group, South African Veterinary Association, Sandton, 16 August, 1986. Journal of the South African Veterinary Association, 59, 104.
- NEWSHOLME, S. J. & COETZER, J.A.W. 1984. Myocardial pathology of domestic ruminants in southern Africa. Journal of the South African Veterinary Association, 55, 89–96.
- NEWSHOLME, S.J., SCHULTZ, R.A. & NEL, P.W. 1986. Improved methods to diagnose plant poisonings in ruminants. Research Highlights, Animal Production, Department of Agriculture and Water Supply. ISBN 062 09693 8.
- NGOMUO, A.J., KAMBARAGE, D.M., MWAMENGELE, G.L.M. & MATOVELO, J.A. 1995. Dichapetalum spp. toxicity in cattle at a farm in Tanzania. Veterinary and Human Toxicology, 37, 143–144.
- NWUDE, N., PARSONS, L.E. & ADAUDI, A.O. 1977. Acute toxicity of the leaves and extracts of Dichapetalum barteri (Engl.) in mice, rabbits and goats. Toxicology, 7, 23–29.
- O’HAGAN, D., PERRY, R., LOCK, J.M., MEYER, J.M.M., DASARADHI, L., HAMILTON, J.T.G. & HARPER, D.B. 1993. High levels of monofluoroacetate in Dichapetalum braunii. Phytochemistry, 1043–1045.
- OELRICHS, P.B. 1981. Georgina gidyea poisoning in cattle and sheep. Queensland Agricultural Journal, 300–302.
- OELRICHS, P.B., KRATZMANN, S., MACLEOD, J.K., SCHÄFFELER, L., NG J.C. & SEAWRIGHT, A.A. 1998. A study of persin, the mammary cell necrosis agent from avocado (Persea americana), and its furan derivative in lactating mice. In: GARLAND, T. & BARR, C.A. (eds.) Toxic plants and other natural toxicants. pp 86–90. New York: CAB.
- OELRICHS, P.B., MACLEOD, J.K. & CAPON, R.J. 1992. Isolation and identification of the toxic principle in Bryophyllum tubiflorum (Kalanchoe), In: JAMES, L.F., KEELER, R.F., BAILEY, E.M., CHEEKE, P.R. & HEGARTY, M.P. (eds.) Poisonous plants. Proceedings of the Third Symposium. Ames: Iowa State University Press.
- OELRICHS, P.B. & McEWAN, T. 1962. The toxic principles of Acacia georginae. Queensland Journal of Agricultural Science, 19, 1–16.
- OOI, H. & COLUCCI, W.S. 2001. Pharmacological treatment of heart failure. In: J.G. HARDMAN, J.G., LIMBIRD. L.E. & GILMAN, A.G. (eds.) Goodman and Gilman’s The Pharmacological Basis of Therapeutics. 10th edn. New York: McGraw-Hill Co. Inc.
- OTANI, A., PALUMBO, N. & READ, G. 1969. Pharmacodynamics and treatment of mammals poisoned by Bufo marinus toxin. American Journal of Veterinary Research, 30, 1865–1872.
- PALMER, E. & PITMAN, N. 1972. Trees of Southern Africa. Cape Town: A.A. Balkema.
- PANTANOWITZ, L., NAUDÉ, T.W. & LEISEWITZ, A. 1998. Noxious toads and frogs of South Africa. South African Medical Journal, 88 (11), 1408–1413.
- PASSMORE, N.H. & CARUTHERS, V.C. 1995. South African Frogs. A Complete Guide. 2nd edn. Johannesburg: Witwatersrand University Press.
- PATTISON, F.L.M. & PETERS, R.A. 1959. Industrial toxic agents. Toxic aliphatic fluorine compounds. London: Elsevier Publishing Company.
- PATTISON, F.L.M. & PETERS, R.A. 1959. Toxic aliphatic fluorine compounds. Amsterdam: Elsevier Publishing Company.
- PEROUTKA, S.J. 1996. Drugs effective in the therapy of migraine. In: HARDMAN, J.G., LIMBIRD, L.E., MOLINOFF, P.B., RUDDON, R.W. & GILMAN, A.G. (eds.) Goodman and Gilman’s The Pharmacological Basis of Therapeutics. New York: Mc Graw Hill.
- PETERS, R.A. 1951. Croonian lectures. Proceedings of the Royal Society of Britain, 139, 143.
- PHILLIPS, E.P. 1926. A preliminary list of the known poisonous plants found in South Africa. Botanical Survey of South Africa. Memoir No. 9. Agricultural Department, Pretoria.
- PHILLIPS, E.P. 1927. Gifblaar en gousiektebossie. Boerdery in Suid-Afrika, Maart, 454–455.
- PHILLIPS, M.C., RICHES, C.R. & SWEET, R.J. 1993. Chemical control of the poisonous plant Dichapetalum cymosum (Hook) Engl. International Journal of Pest Management, 39, 358–365.
- POLE EVANS, I.B. 1923. Gouwziekte Veld: Its vegetation and flora. Report on Veterinary Research, Union of South Africa, 9 & 10, 109–119.
- PORTER, J.K. & THOMPSON, F.N. 1992. Effect of fescue toxicosis on reproduction in livestock. Journal of Animal Science, 70, 1694–1603.
- PRETORIUS, P.J. & TERBLANCHE, M. 1967. A preliminary study on the symptomatology and cardiodynamics of gousiekte in sheep and goats. Journal of the South African Veterinary Medical Association, 38, 29–53.
- PRETORIUS, P.J., TERBLANCHE, M., VAN DER WALT, J.D. & VAN RYSSEN, J.C.J. 1973. Cardiac failure in ruminants caused by gousiekte. In: BAJUSZ, E., RONA, G., BRINK, A.J. & LOCHNER, A. (eds.) Cardiomyopathies, 2, 384–624. Baltimore: University Park Press.
- PRETORIUS, P.J., VAN DER WALT, J.J., KRUGER, J.M. & NAUDÉ, T.W. 1969. A comparison between haemodynamics and cellular micro-electrode experiments with the cardiac aglycone epoxyscillirosidin. South African Medical Journal, 1360–1363.
- PROZESKY, L., FOURIE, N., NESER, J.A. & NEL, P.W. 1988. A field outbreak in Ile-de-France sheep of a cardiotoxicosis caused by the plant Pachystigma pygmaeum (Schltr) Robyns (Rubiaceae). Onderstepoort Journal of Veterinary Research, 55, 193–196.
- QUIN, J.I. & CLARK, R. 1947. Studies on the action of potassium monofluoroacetate (CH2FCOOK) [Dichapetalum cymosum (Hook.) Engl.] toxin on animals. Onderstepoort Journal of Veterinary Science and Animal Industry, 22, 77–82.
- RADFORD, D.J., GILLIES, A.D., HINDS, J.A. & DUFFY, P. 1986. Naturally occurring cardiac glycosides. The Medical Journal of Australia, 144, 540–544.
- RANDEL, R.D., CHASE, C.C. & WYSE, S.J. 1992. Effects of gossypol and cottonseed products on reproduction of mammals. Journal of Animal Science, 1628–1638.
- REICHSTEIN, T., VON EUW, J., PARSON, J.A. & ROTHSCHILD, M. 1968. Heart poisons in the monarch butterfly. Science, 161, 861–866.
- RIMINGTON, C. 1935. Chemical investigations of the ‘Gifblaar’ Dichapetalum cymosum (Hook.) Engl. I. Onderstepoort Journal of Veterinary Science and Animal Industry, 5, 81–95.
- RINDL, M.M. 1924. Preliminary note on a poisonous alkaloid from the overground portions of the Transvaal Yellow Tulp (Homeria pallida). Transactions of the Royal Society of South Africa, 11, 251–256.
- RISCO, C.A., CHENOWETH, P.J., LARSON, R.E., VELEZ, J. & SHAW, N. 1993. The effect of gossypol in cottonseed meal on the performance and on hematological and semen traits in postpubertal Brahman bulls. Theriogenology, 629–642.
- ROGERS, P.A.M., HENAGHAN, T.P. & WHEELER, B. 1975. Gossypol poisoning in young calves. Irish Veterinary Journal, 29, 9–13.
- ROTHSCHILD, M., VON EUW, J. & REICHSTEIN, T. 1973. Cardiac glycosides in the polkadot moth, Syntomeida aplais Walk. (Atenudae, Lep.) with some observations on the toxic qualities of Amata (= Syntomis) phega L. Proceedings of the Royal Entomological Society of London (B) , 183, 227–247.
- SANI, Y., ATWELL, R. & SEAWRIGHT, A.A. 1991. The cardiotoxicity of avocado leaves. Australian Veterinary Journal, 68, 150–151.
- SAPEIKA, N. 1936. The pharmacological actions of plants of the genera Cotyledon and Crassula. N.O. Crassulaceae. Archives Internationales de Pharmacodynamic et de Therapie, 54, 307–328.
- SAPEIKA, N. 1944. The action of rubellin, a glycoside from Urginea rubella. South African Journal of Medical Science, 9, 31–32.
- SAPEIKA, N. 1949. The action of venenatin, a glycoside from Acokanthera venenata. South African Journal of Medical Science, 14, 87–88.
- SCHNEIDER, D.J., MILES, C.O., GARTHWAITE, I., VAN HALDEREN, A., WESSELS, J.C. & LATEGAN, H.J. 1996. First report of field outbreaks of ergot-alkaloid toxicity in South Africa. Onderstepoort Journal of Veterinary Research, 63, 97–108.
- SCHULTZ, R.A., COETZER, J.A.W., KELLERMAN, T.S. & NAUDÉ, T.W. 1982. Observations on the clinical, cardiac and histopathological effects of fluoroacetate in sheep. Onderstepoort Journal of Veterinary Research, 49, 237–245.
- SCHULTZ, R.A., FOURIE, N., BASSON, K.M. LABUSCHAGNE, L. & PROZESKY, L. 2001. Effect of pavetamine on protein synthesis in rat tissue. Onderstepoort Journal of Veterinary Research, 68, 325–330.
- SCHULTZ, R.A., NAUDÉ, T.W. & PRETORIUS, P.J. 1975. Cardiac glycoside poisoning in animals. South African Medical Journal, 49, 285–286.
- SCHUTTE, P.J., ELS, H.J., BOOYSENS, J. & PIENAAR, J.G. 1984. Ultrastructure of myocardial cells in sheep with congestive heart failure induced by Pachystigma pygmaeum. South African Journal of Science, 80, 378.
- SEAWRIGHT, A.A. 1989. Animal Health in Australia. Chemical and Plant Poisons. 2nd edn. Canberra: Australian Government Publishing Service.
- SHONE, D.K. & DRUMMOND, R.B. 1965. Poisonous plants of Rhodesia. Rhodesian Agricultural Journal, 62, 7–64.
- SHONE, D.R., PHILLIP, J.R. & CHRISTIE, G.J. 1959. Agalactia of sows caused by feeding the ergot of the bullrush millet, Pennisetum typhoides. The Veterinary Record, 71, 729–756.
- SKOU, J.C. 1965. Enzymatic basis for active transport of Na and K across cell membranes. Physiological Reviews, 45, 596–617.
- SMIT, J.D. 1959. Die histopatologiese diagnose van gousiekte. Journal of the South African Veterinary Medical Association, 30, 447–450.
- SMITH, H.A. 1957. The pathology of gossypol poisoning. American Journal of Pathology, 33, 353–365.
- SNYMAN, L.D. 1978. Biochemiese verandering in die hartspierweefsel van skape met gousiekte onderlede. Proefskrif vir die graad Doctor Scientiae (Biochemie), Potchefstroom Universiteit vir CHO.
- SNYMAN, L.D., KELLERMAN, T.S., SCHULTZ, R.A., JOUBERT, J.P.J., BASSON, K.M. & LABUSCHAGNE, L. 2004. Conditioned feed aversion (CFA) as a means of preventing the intake of yellow-tulp (Homeria pallida) by livestock. In: ACAMOVIC, T, STEWART, C.S. & PENNYCOTT, T.W. (eds.) Poisonous plants and related toxins. 531–539. Wallingford: CAB International.
- SNYMAN, L.D., SCHULTZ, R.A., JOUBERT, J.P.J., BASSON, K.M. & LABUSCHAGNE, L. 2003. Conditioned feed aversion as a means to prevent tulp (Homeria pallida) poisoning in cattle. Onderstepoort Journal of Veterinary Research, 70:43–48.
- SNYMAN, L.D., VAN DER WALT, J.J. & PRETORIUS, P.J. 1982. A study on the function of some subcellular systems of the sheep myocardium during gousiekte. I. The energy production system. Onderstepoort Journal of Veterinary Research, 49, 215–220.
- SNYMAN, L.D., VAN DER WALT, J.J. & PRETORIUS, P.J. 1982. A study on the function of some subcellular systems of the sheep myocardium during gousiekte. II. The contractile protein system. Onderstepoort Journal of Veterinary Research, 49, 221–226.
- SOGA, J.F. 1891. Disease ‘Nenta’ in goats. Agricultural Journal of the Cape of Good Hope, 3, 40–142.
- STADLER, P., VAN RENSBURG, I.B.J. & NAUDÉ, T.W. 1991. Suspected avocado (Persea americana) poisoning in goats. Journal of the South African Veterinary Association, 62, 186–188.
- STENT, S.M. 1916. Gifblaar. Agricultural Journal and Smallholder of South Africa, 70–73.
- STENT, S.M. & CURSON, H.H. 1922. Poisonous plants of South Africa. I. Tulp. Bulletin 6, Department of Agriculture, Union of South Africa.
- STENT, S.M. & CURSON, H.H. 1922. Poisonous plants of South Africa. II. Slangkop. Bulletin 7, Department of Agriculture, Union of South Africa.
- STEYN, D.G. 1928. Gifblaar poisoning. A summary of our present knowledge in respect of gifblaar poisoning. Report on Veterinary Research, Union of South Africa, 13 & 14, 187–194.
- STEYN, D.G. 1928. Tulp poisoning. Report on Veterinary Research, Union of South Africa, 13 & 14, 197–202.
- STEYN, D.G. 1929. Recent investigations into the toxicity of known and unknown poisonous plants in the Union of South Africa. Report on Veterinary Research, Union of South Africa, 15, 777–803.
- STEYN, D.G. 1931. Recent investigations into the toxicity of known and unknown poisonous plants in the Union of South Africa. Report on Veterinary Research, Union of South Africa, 17, 707–722.
- STEYN, D.G. 1932. A study of the factors concerned in the determination of the toxicity of Cotyledon orbiculata. Report on Veterinary Research, Union of South Africa, 18, 899–938.
- STEYN, D.G. 1932. Investigations into the toxicity of known and unknown poisonous plants in the Union of South Africa. Report on Veterinary Research, Union of South Africa, 18, 871–892.
- STEYN, D.G. 1933. Plant poisoning in stock and the development of tolerance. Onderstepoort Journal of Veterinary Science and Animal Industry, 1, 149–156.
- STEYN, D.G. 1934. The toxicology of plants in South Africa. Central News Agency Ltd.
- STEYN, D.G. 1935. Plant poisoning in stock and the development of tolerance. Onderstepoort Journal of Veterinary Science and Animal Industry, 4, 417–420.
- STEYN, D.G. 1935. Recent investigations into the toxicity of known and unknown poisonous plants in the Union of South Africa. Onderstepoort Journal of Veterinary Science and Animal Industry, 4, 399–415.
- STEYN, D.G. 1936. Recent investigations into the toxicity of known and unknown poisonous plants in the Union of South Africa. Onderstepoort Journal of Veterinary Science and Animal Industry, 7, 169–178.
- STEYN, D.G. 1937. Recent investigations into the toxicity of known and unknown poisonous plants in the Union of South Africa. VIII. Onderstepoort Journal of Veterinary Science and Animal Industry, 9, 573–582.
- STEYN, D.G. 1945. Krimpsiekte by vee. Boerdery in Suid Afrika, Herdruk 38, 1–3.
- STEYN, D.G. 1945. Slangkop- en tulpvergiftiging by vee. Boerdery in Suid–Afrika, Herdruk 46, 1–4.
- STEYN, D.G. 1949. Vergiftiging van mens en dier. Pretoria: Van Schaik.
- STEYN, D.G. 1962. Grasshopper (Phymateus leprosus Fabr.) poisoning in a bantu child. South African Medical Journal, 36, 822–823.
- STEYN, D.G. & VAN DER WALT, S.J. 1941. Recent investigations into the toxicity of known and unknown poisonous plants in the Union of South Africa. XI. Onderstepoort Journal of Veterinary Science and Animal Industry, 16, 121–147.
- STEYN, P.S. & VAN HEERDEN, F.R. 1998. Bufadienolides of plant and animal origin. Natural Products Reports, 397–413.
- STEYN, P.S, VAN HEERDEN, F.R., VLEGGAAR, R & ANDERSON, L.A.P. 1986. Bufadienolide glycosides of the Crassulaceae. Structure and stereochemistry of orbicuside A–C, novel toxic metabolites of Cotyledon orbiculata. Journal of the Chemical Society Perkin Transactions, 1633–1636.
- STRYDOM, J.A. & JOUBERT, J.P.J. 1983. The effect of predosing Homeria pallida Bak. to cattle to prevent tulp poisoning. Journal of the South African Veterinary Association, 54, 201–203.
- SWAN, G.E., SCHULTZ, R.A., KELLERMAN, T.S., MÜLDERS, M.S.G., MAARTENS, B.P. & VAN DER WALT, J.J. 1995. In vivo effects of a novel calcium antagonist (R56865) against induced epoxyscillirosidin and tulp poisoning in sheep. Onderstepoort Journal of Veterinary Research, 62, 163–166.
- TANNOCK, J. 1975. Seasonal variation in toxicity of Dichapetalum cymosum (Hook.) Engl. in the Nyamandhlovu area. Rhodesian Journal of Agricultural Research, 13, 67–78.
- TERBLANCHE, M. & ADELAAR, T.F. 1965. A note on the toxicity of Cotyledon orbiculata L. Journal of the South African Veterinary Medical Association, 36, 555–559.
- THEILER, A. 1906. Gouw-Ziekte. Report of the Government Veterinary Bacteriologist of the Transvaal, 1906–1907, 21–22.
- THEILER, A., DU TOIT, P.J. & MITCHELL, D.T. 1923. Gousiekte in sheep. Report on Veterinary Research, Union of South Africa, 9 & 10, 9–105.
- TÖLKEN, H.R. 1978. New taxa and new combinations in Cotyledon and other genera. Bothalia, 12, 377–393.
- TREBLE, D.H., LAMPORT, D.T.A. & PETERS, R.A. 1962. The inhibition of plant aconitate hydratase (aconitase) by fluorocitrate. Biochemical Journal, 85, 113–115.
- TUSTIN, R.C., THORNTON, D.J. & KLEU, C. 1984. An outbreak of Cotyledon orbiculata L. poisoning in a flock of Angora goat rams. Journal of the South African Veterinary Association, 181–184.
- UYS, P.L. & ADELAAR, T.F. 1957. A new poisonous plant. Journal of the South African Veterinary Medical Association, 28, 5–8.
- VAHRMEIJER, J. 1981. Poisonous plants of Southern Africa that cause stock losses. Cape Town: Tafelberg.
- VAN DER WALT, J.J. & VAN ROOYEN, J.M. 1977. Use of technetium-99m to determine haemodynamic changes during the development of ventricular failure with gousiekte. South African Medical Journal, 52, 375.
- VAN DER WALT, J.J., VAN ROOYEN, J.M., CILLIERS, G.D., VAN RYSSEN, J.C.J. & VAN AARD, M.N. 1981. Ratio of cardiopulmonary blood volume to stroke volume as an index of cardiac function in animals and in man. Cardiovascular Research, 15, 580–587.
- VAN DER WALT, S.J. 1944. Recent investigations into the toxicity of known and unknown poisonous plants in the Union of South Africa. Onderstepoort Journal of Veterinary Science and Animal Industry, 20, 75–83.
- VAN DER WALT, S.J. & STEYN, D.G. 1939. Recent investigation into the toxicity of known and unkown poisonous plants in the Union of South Africa. IX. Onderstepoort Journal of Veterinary Science and Animal Industry, 12, 335–366.
- VAN DER WALT, S.J. & STEYN, D.G. 1940. Recent investigations into the toxicity of known and unknown poisonous plants, etc. in the Union of South Africa. X. Onderstepoort Journal of Veterinary Science and Animal Industry, 15, 261–277.
- VAN DER WALT, S.J. & STEYN, D.G. 1941. Recent investigations into the toxicity of known and unknown poisonous plants in the Union of South Africa. XII. Onderstepoort Journal of Veterinary Science and Animal Industry, 17, 211–223.
- VAN DER WALT, S.J. & STEYN, D.G. 1942. Siektes wat met lamsiekte verwar kan word. Boerdery in Suid-Afrika, Herdruk, 63.
- VAN DER WALT, S.J. & STEYN, D.G. 1943. Recent investigations into the toxicity of known and unknown poisonous plants in the Union of South Africa. XIII. Onderstepoort Journal of Veterinary Science and Animal Industry, 18, 207–224.
- VAN DER WALT, S.J. & STEYN, D.G. 1946. Recent investigations into the toxicity of known and unknown poisonous plants in the Union of South Africa. XV. Onderstepoort Journal of Veterinary Science and Animal Industry, 21, 45–55.
- VAN DIJK, C., VICKERY, B. & VICKERY, M.L. 1972. Poisoning in livestock by Dichapetalum toxicarum. Nigerian Journal of Science, 6, 135–138.
- VAN HEERDEN, F.R., VLEGGAAR, R. & ANDERSON, L.A.P. 1980. Structure elucidation of thesiuside, a bufadienolide glycoside from Thesium lineatum. South African Journal of Chemistry, 39–41.
- VAN HEERDEN, F.R., VLEGGAAR, R. & ANDERSON, L.A.P. 1988. Bufadienolide glycosides from Urginea physodes. First report of natural 14-deoxybufadienolides. South African Industrial Chemistry, 41, 145–151.
- VAN ROOYEN, F.G. & PIETERSE, M.J. 1968. Die chemie van Cotyledon wallichii Harv. (kandelaarsbos). II. Die isolering van ‘n bufadienolied. Journal of the South African Chemical Institute, 21, 89–90.
- VAN WYK, A.E. & MALAN, S.J. 1988. Field guide to the wild flowers of the Witwatersrand and Pretoria region. Cape Town: Struik.
- VAN WYK, A.J., 1975. The chemistry of Cotyledon wallichii Harv. III. The partial constitution of cotyledoside, a novel bufadienolide. Journal of the South African Chemical Institute, 28, 281–283.
- VAN WYK, A.J. & ENSLIN, P.R. 1968. Bufadienolides in Moraea polystachya Ker. and M. graminicola Obem. Journal of the South African Chemical Institute, 22, 186–190.
- VAN WYK, A.J. & ENSLIN, P.R. 1969. A new toxic principle of Homeria glauca (Wood & Evans) N.E. Br. Journal of the South African Chemical Institute, 22, 186–190.
- VAN WYK, A.J. & ENSLIN, P.R. 1969. Bufadienolides from Iridaceae. Journal of the South African Chemical Institute, 22, 66–70.
- VELDSMAN, D.P. 1949. On venenatin, the cardiac glycoside from the bark of wood of Acokanthera venenata G. Don. South African Industrial Chemist, 3, 144–147.
- VELDSMAN, D.P. 1952. Chemical investigation of Pachystigma pygmaeum Schlechter (gousiektebossie). Onderstepoort Journal of Veterinary Research, 25, 113–121.
- VERDCOURT, B. & TRUMP, E.C. 1969. Common poisonous plants of East Africa. London: Collins.
- VERSCHOOR, J.A. & POTGIETER, D.J.J. 1984. Preparation and properties of a crude extract toxic to guinea-pigs from Pachystigma pygmaeum, a plant causing heart failure in ruminants. Onderstepoort Journal of Veterinary Research, 51, 245–248.
- VICKERY, B. & VICKERY, M.L. 1973. Toxicity for livestock of organofluorine compounds present in Dichapetalum plant species. The Veterinary Bulletin, 43, 537–542.
- VISSER, J.H. 1964. Iron supplementation as a means of preventing poisoning in livestock by Pachystigma pygmaeum. South African Journal of Agricultural Science, 7, 173–175.
- VON EUW, J., FISHELSON, L., PARSONS, J.A., REICHSTEIN, T. & ROTHSCHILD, M. 1967. Cardenolides (heart poisons) in a grasshopper feeding on milkweeds. Nature, 214, 36–39.
- WALKER, J. 1908. Gouw-Ziekte. A disease of sheep. Report of the Government Veterinary Bacteriologist of the Transvaal, 1908–1909, 74–99.
- WALSH, L.H. 1909. South African poisonous plants. Cape Town: Maskew Miller.
- WATT, J.M. & BREYER-BRANDWIJK, M.G. 1962. The medicinal and poisonous plants of southern and eastern Africa. 2nd edn. Edinburgh & London: E & S Livingstone Ltd.
- WEST, O. 1947. Umkuzanne (Dichapetalum cymosum). The Rhodesian Agricultural Journal, 44, 609–613.
- WESTERMARK, H. 1959. Digitalis inactivation in vitro due to rumen microbes from sheep. Acta Veterinaria Scandinavica, 1, 67–73.
- WHITTEM, J.H. & MURRAY, L.R. 1963. The chemistry and pathology of Georgina River poisoning. Australian Veterinary Journal, 39, 168–173.
- WOOD, J.M. 1914. Note on Natal Slangkop, or poison bulb. Agricultural Journal of the Union of South Africa, 12, 703–705.
- ZELSKI, R.Z., ROTHWELL, J.T., MOORE, R.E. & KENNEDY, D.J. 1995. Gossypol toxicity in preruminant calves. Australian Veterinary Journal, 394–398.