- Plant Poisonings and Mycotoxicoses of Livestock in Southern Africa, 2nd Edition
- Gastrointestinal tract
Gastrointestinal tract
This content is distributed under the following licence: Attribution-NonCommercial CC BY-NC  View Creative Commons Licence details here
View Creative Commons Licence details here

Introduction
Plants that affect the gastrointestinal tract can be roughly divided into three groups, namely, those that (a) induce signs other than diarrhoea, like vermeersiekte vide infra; (b) cause diarrhoea; or (c) are responsible for bloat, impaction or gastroenteritis due to mechanical injury. Although the latter three conditions are not poisonings, they can be regarded as falling more or less within the ambit of the toxicologist. The most important plants affecting the gastrointestinal tract are the vermeersiekte-inducing Geigeria spp.
Poisonings without notable diarrhoea
Vermeersiekte
Geigeria spp.
Geigeria ornativa
Geigeria aspera
Vermeersiekte is a condition mostly of sheep caused by Geigeria spp., characterized by regurgitation of ruminal contents, stiffness, paresis and paralysis. It is one of the most important plant poisonings of sheep on the subcontinent, accounting in South Africa alone for c.13% of all stock deaths from plant poisonings and mycotoxicoses.76 Massive outbreaks periodically occur when weather conditions favour the growth of vermeerbos at the expense of pasture grasses. More than a million sheep died of the disease in the former Griqualand West region of the Northern Cape Province between 1929 and 1930,164 15 000 of these in a small area comprising only 20 farms.40 During 1954, an outbreak claimed 55 000 sheep in the same area.55 About 34 000 sheep are estimated to die annually of this poisoning in South Africa.76 Cattle ingesting Geigeria spp. primarily develop stiffness.
Geigeria ornativa O. Hoffm. (Asteraceae)
(= G. passerinoides Harv.)
(= G. africana Griess. subsp. ornativa (O. Hoffm.) Merxm.)
Vermeerbos
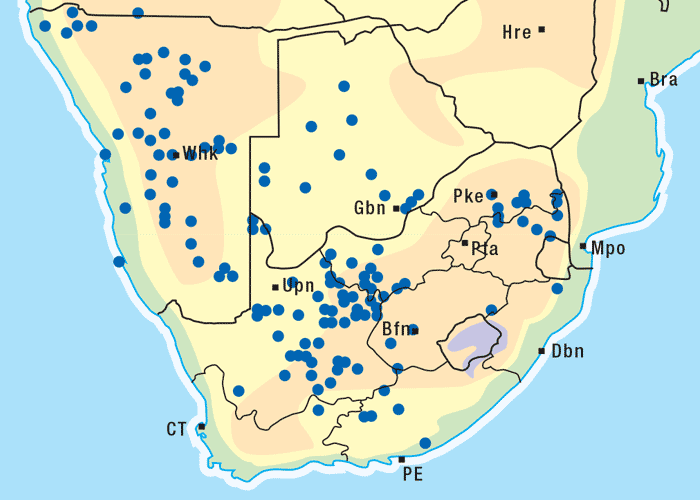
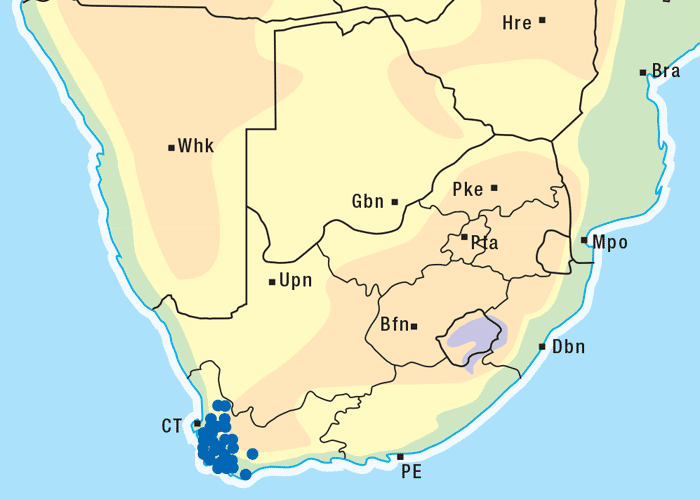
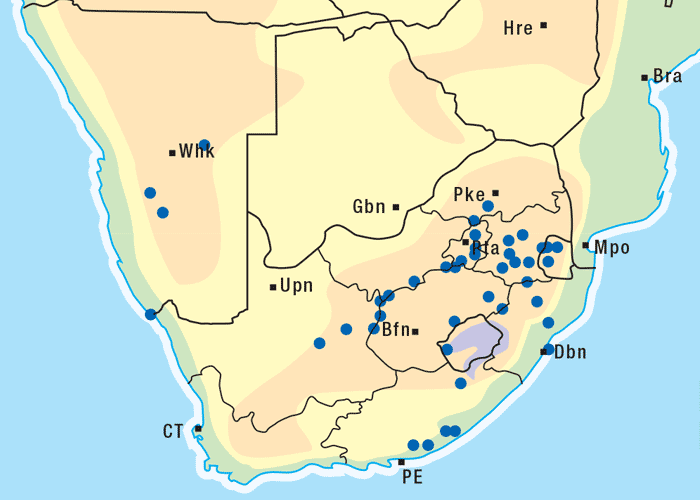
Vermeerbos is a woody annual or weak perennial, usually about 100 mm tall, but varying greatly in size. The roughly textured, narrow, ribbon-like leaves of about 100 mm in length and 1–5 mm wide, emerge close together on the branches (Figures 1 and 2). Yellow, sessile flowerheads with unisexual (female) ray florets and bisexual disc florets, are borne in the forks of the branches. The daisy-like flowerheads (Figures 2 and 3) persist for many months until the seeds are shed.118, 164 The seeds are distributed by wind and water, and in the droppings of animals.164 Geigeria ornativa is found in the drier southern, south-western and western parts of South Africa, and in the neighbouring parts of Namibia (Figure 4).118, 164 Massive outbreaks of vermeersiekte are periodically caused by this species in the former Griqualand West area of the Northern Cape Province.
Geigeria aspera Harv. var. aspera (Asteraceae)
Vermeerbos
Vahrmeijer164 describes G. aspera as a dense, much-branched, woody shrublet of about 200 mm in height. The aerial parts are weakly perennial and the stem-base and root system usually perennial. The leaves are rough-textured and ribbon-like, but they are usually shorter than those of G. ornativa, measuring about 60 mm long and about 4 mm wide. The yellow flowerheads, which are similar to those of G. ornativa, arise from the leaf axils or the ends of branches (Figures 5 and 6). The narrow, oblong seeds are released once the bracts of the flowerheads have decayed.164
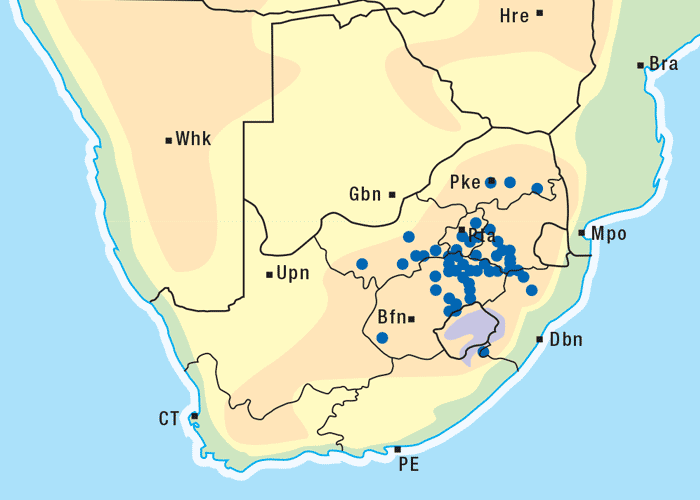
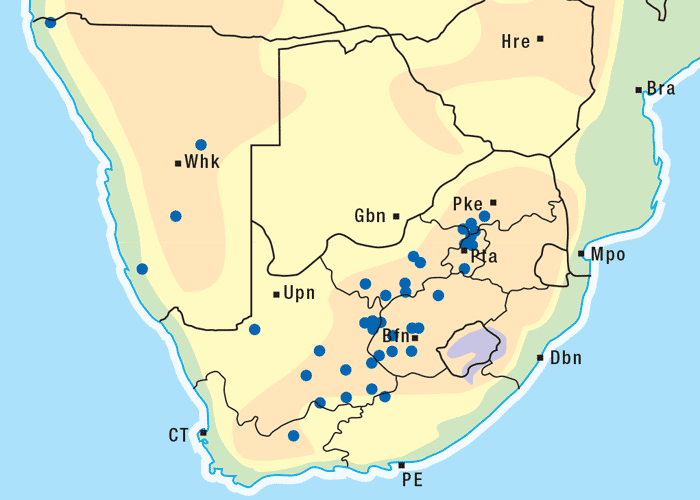
This vermeerbos chiefly frequents open savannah where the rainfall is high (Figure 7). According to Vahrmeijer,164 G. aspera favours the loam and clay soils often found near swamps or on river banks.
Vermeersiekte was recorded30 for the first time by Brandford in 1878.14 As early as 1884, Hutcheon63 referred to G. ornativa (= G. passerinoides) as vermeerbos, thus implying that farmers already then associated the disease with the plant. They suspected that the plant was involved in the aetiology of the condition because the incidence of vermeersiekte was highest where G. ornativa was abundant; large outbreaks were recorded on veld denuded by locusts of all vegetation but vermeerbos; the disease never occurred in the absence of the plant.40, 43, 65, 146 Despite all the circumstantial evidence incriminating it, many negative experiments were carried out37, 38, 43, 63, 64, 67 before Du Toit,40 in the early twenties, unequivocally established that Geigeria ornativa caused vermeersiekte.17, 40 The failure of the earlier experiments can probably be ascribed to the dosing of insufficient plant material to bring about intoxication.40
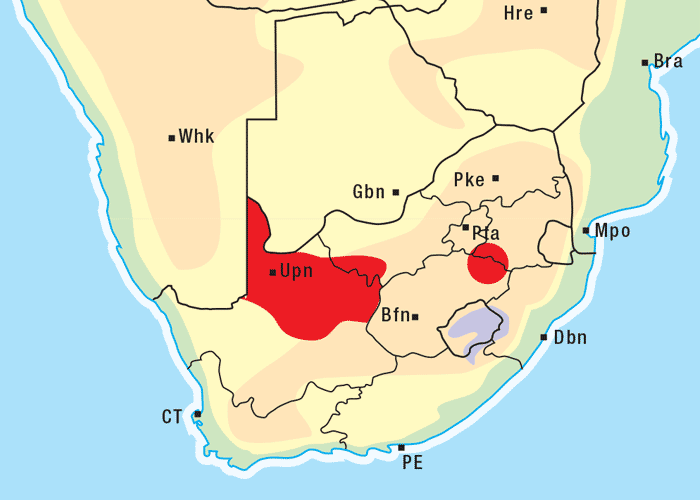
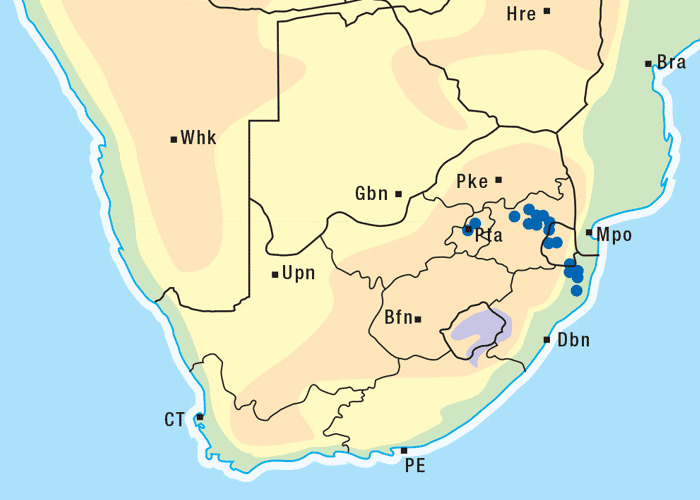
Apart from G. ornativa, a further two species have been incriminated in the disease, namely, G. aspera var. aspera154 and G. pectidea. A fourth species, G. burkei subsp. burkei var. zeyheri (= G. zeyheri), elicited vermeersiekte under experimental conditions, although it had not been linked with natural outbreaks of the disease. Recently, a variety of this species, G. burkei Harv. subsp. burkei var. hirtella Merxm., was positively incriminated in vermeersiekte of sheep in the bushveld of the Limpopo Province of South Africa.13 Geigeria ornativa, the most important of the vermeersiekte-inducing plants, periodically causes severe stock losses especially in an area of the Northern Cape Province, previously known as Griqualand West. Geigeria pectidea plays a relatively minor role in the induction of the disease in Griqualand West, while G. aspera var. aspera is responsible for localized, but often severe, outbreaks in the western and northern Free State as well as in eastern Mpumalanga, particularly in the Standerton and the southern part of the Ermelo districts40 (Figure 8). Geigeria aspera var. aspera is the most poisonous of the Geigeria species, with a toxicity c.10 times greater than that of G. ornativa, while G. pectidea and G. burkei var. burkei subsp. zeyheri are intermediate in toxicity, being about three times more toxic than G. ornativa.130, 157
Vermeerbossie collected in various places, or at different times of the year, can vary considerably in toxicity.55 Geigeria ornativa can apparently cause the disease at any stage of its development,40 but opinions differ as to when it is the most toxic. Some believe that all stages of growth are equally toxic,40 others that toxicity in the preflowering stage is highest.130 Sun-drying or wilting does not destroy the toxicity of G. aspera var. aspera, but the toxicity of dried plants is said to deteriorate on storage.40, 128 In contrast, Pienaar et al.113 reported that dried G. ornativa was still toxic after 15 years of storage.
The toxicity of G. ornativa is said to diminish after rains, although the effect of rain may be merely to promote the pasture growth, resulting in less vermeerbos being eaten.55 Small, stunted vermeerbos, growing in shallow soil overlying limestone, is reported by farmers to be the most dangerous. Shallow soils and below average rainfall make for poor grass cover and high intake of vermeerbos.55
Sheep fed exclusively on G. ornativa consumed at most about 0,9 kg of fresh flowerheads, twigs and leaves per day. The smallest amount of fresh plant to induce ovine vermeersiekte in the experiment of Du Toit40 was 2,3 kg over three days, but in another trial 36,3 kg had to be taken in 42 days before signs appeared. Van Heerden et al.171 reported that sheep, fed 5 g/kg/d dry, flowering G. ornativa mixed in lucerne and some dairy meal, developed clinical signs of vermeersiekte within c.19–33 days.
Fresh, preflowering G. aspera var. aspera administered in a single dose of 300 g154, 155 or 600 g on two successive days,156 was fatal to sheep. On the other hand, considerably more of the dry material had to be given to produce even a mild effect. A total of 2,6 kg of dry plant (= c.6,5 kg of fresh material) caused only transient signs of vermeersiekte, whereas 1,2 kg fresh plant was fatal.156 Variation in toxicity of G. aspera makes estimating a toxic dose difficult. Generally speaking, about 100 g/kg dry G. aspera material given in divided doses of 5–7,5 g/kg per day can be expected to cause vermeersiekte in two to five weeks (N. Fourie, OVI, unpublished data, 1991). Fresh G. burkei var. burkei subsp. zeyheri drenched at the rate of 2,5 kg over three days, resulted in bloat and death of a sheep.154, 155
Four different forms of vermeersiekte have been recognized, namely, that characterized by (a) regurgitation of ruminal contents, (b) bloat, (c) stiffness, and (d) paralysis. Any one or more of these forms may be manifested at the same time by a particular animal, and some species are more prominently affected by certain forms than others (vide infra).40 Sheep and goats are more susceptible to vermeersiekte than are cattle. Claims by farmers that horses and donkeys are affected have not been corroborated by experimental evidence. Donkeys, fed on significant amounts of G. ornativa for a month, showed no ill effects.40 Vermeersiekte has reportedly been seen in wild springbok.55
The first signs of vermeersiekte may appear a few days to a few weeks after commencement of grazing on Geigeria spp. Affected sheep lag behind the flock,65 tire easily, develop a stiff gait, and frequently lie down.40 Eventually, they may become too weak/paretic to support themselves in a standing position. If at this stage they are lifted up they may stand trembling with the feet together and back arched, sometimes taking a few steps before collapsing again. Paralysis may set in after a few days.40
Besides stiffness and paralysis, intoxicated sheep can develop chronic regurgitation (often accompanied by dilatation of the oesophagus), a cough, diarrhoea and, occasionally, bloat.40, 65, 66, 157 Affected sheep may be identified by a green discolouration around the nose and mouth caused by regurgitation of ruminal content. The first sign may be salivation that increases in profusion until the animal actually regurgitates (Figure 9). Animals that ‘vomit’ and purge often undergo a dramatic drop in condition. Immediately after ‘vomition’ the sheep – which may appear to be quite healthy, apart possibly from a cough – can start grazing normally again.157 ‘Vomition’ occurs at short intervals and can be stimulated by drinking or exercise.40, 65 After ‘vomition’, or independently of it, spasmodic contractions of the oesophagus have been observed. Death may occur within a few days, but sheep suffering from chronic pneumonia sometimes take weeks to die. The tympanitic form of vermeersiekte is very rare.40 According to Steyn, those that take in large amounts of vermeerbos may suddenly die without showing the usual signs of vermeersiekte.155
Sheep may exhibit the stiff, paralytic or vomiting forms separately or together.55, 157 Goats develop signs similar to those of sheep, but they are apparently more susceptible to the paralytic form of the disease.40, 55, 157 Cattle usually contract only the stiff or paralytic form. The signs often start with salivation followed by a stiff gait, sometimes accompanied by paralysis and a conspicuous loss of condition. Although they seldom regurgitate,55, 157 dilatation of the oesophagus and/or pneumonia have been observed in cattle that apparently had not ‘vomited’ (J.J. van Niekerk, State Veterinarian, Vryburg, personal communication, 1987).
The causes of death in vermeersiekte are listed as asphyxiation as a result of choking on inhaled ingesta or paralysis of the respiratory centre, exhaustion from ‘vomition’ and purgation, heart failure, and acute or chronic foreign body pneumonia.55, 130, 157 The mortality rate will depend on the constitution of the affected stock, their nutritional status, the amount and the toxicity of the Geigeria ingested, the stress to which the animals are subjected, and the presence of pneumonia.55 Foreign body pneumonia is probably the most important cause of death.55 Up to 80% of a flock may die157 and the morbidity can be high; Du Toit40 averred in 1928 that if sheep are left on Geigeria veld sufficiently long 100% may be affected. The prognosis is usually good if stock are removed early from the source of intoxication.55, 158
The mechanism responsible for the ‘vomition’ has not been satisfactorily explained. ‘Vomition’ has variously been ascribed to stimulation of the vomition centre in the medulla oblongata, persistent local irritation of the rumen and abomasum,130 and uncontrolled rumination reflexes owing to disturbance in the sensitivity of the pharyngeal area.55 On two occasions non-specific lesions have been reported in the central nervous system of vermeersiekte sheep, namely, degeneration, perivascular oedema and focal necrobiosis in the thalamus (Smit, 1958, cited by Grosskopf55) and chromatolysis of neurons in the thalamus, as well as extensive myelin degeneration of the spinal cord (J.W. Nesbit, Faculty of Veterinary Science, University of Pretoria, personal communication, 1982). However, a recent more comprehensive investigation of vermeersiekte failed to reveal significant changes either in the CNS or peripheral nerves of the oesophagus and hind limb. In the light of these findings, the authors, Van der Lugt and Van Heerden (1993),168 concluded that the ‘vomition’ and locomotory disturbances were muscular rather than central nervous in origin.
The most notable clinical pathological changes in experimentally induced vermeersiekte are elevation in the activities of aspartate transaminase and γ-glutamyltransferase in the sera of affected sheep171 (N. Fourie, OVI, unpublished data, 1992) indicating liver involvement.
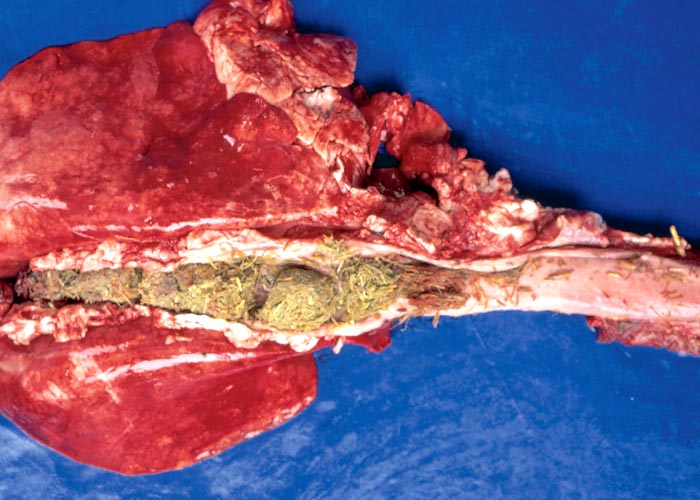
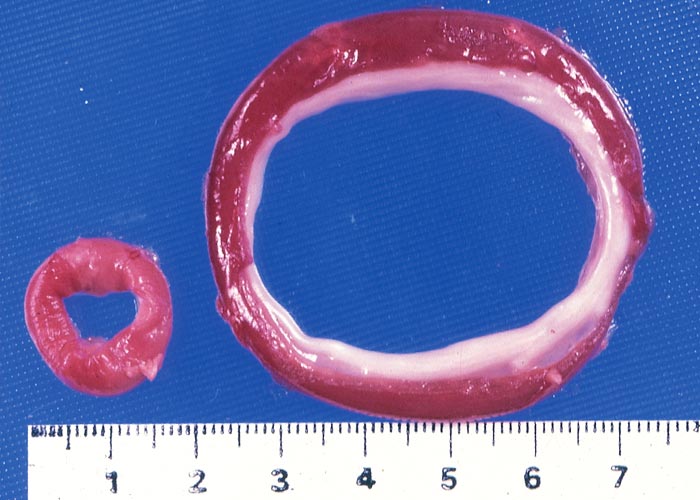
Lips, stained green by ingesta, the presence of rumen content in the air passages and/or a foreign body pneumonia are strongly indicative of vermeersiekte. Among the lesions described is fairly consistent dilatation of the oesophagus (Figures 10 and 11),113 diagnosed in the living animal by ballotment of the neck113 or contrast radiography.171 Using contrast radiography, Van Heerden et al. were able to diagnose oesophageal dilatation in 8 out of 13 affected animals in a feeding trial, where only one regurgitated.171
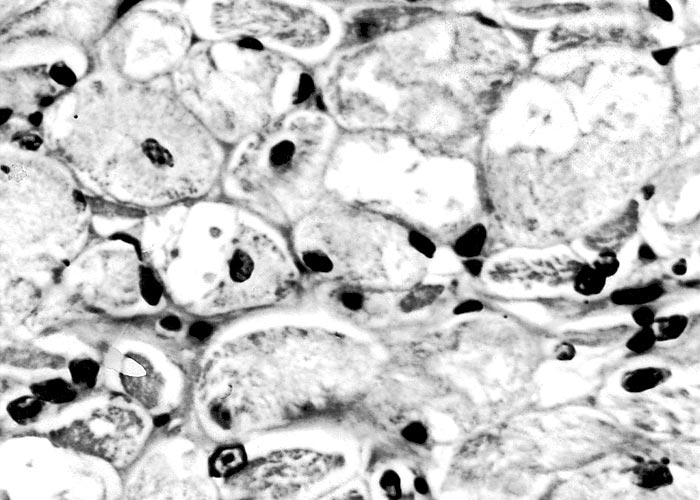
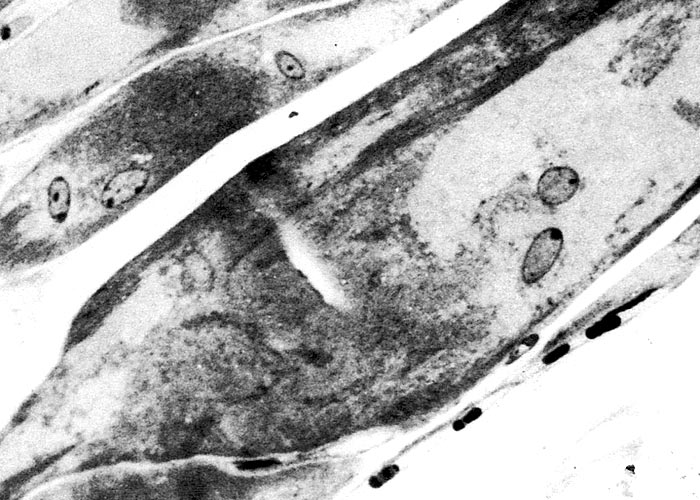
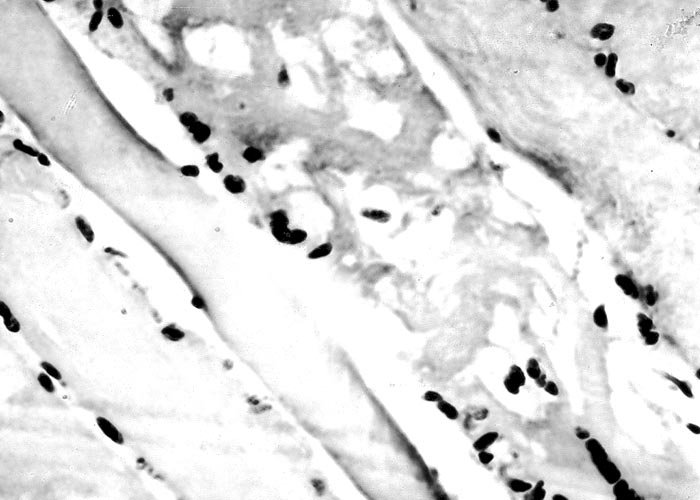
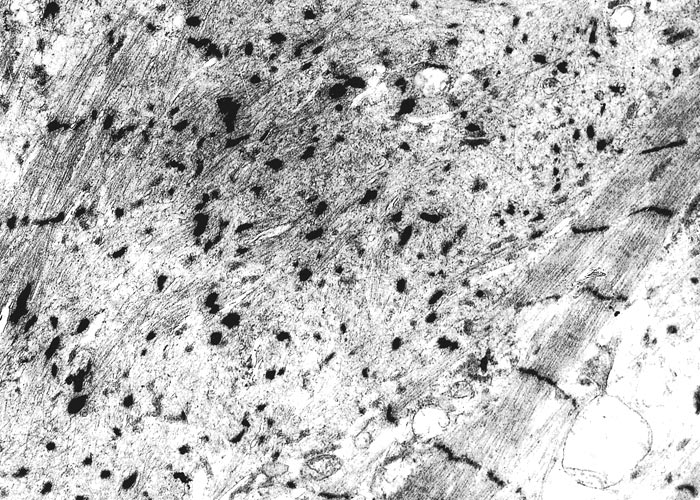
The most complete account of the lesions in vermeersiekte is given by Pienaar et al.113 (1973), who described changes in both the skeletal muscles and oesophagus. Light microscopical examination revealed lesions in randomly distributed individual myocytes or groups of muscle fibres. Affected fibres contained vacuoles of different size into which one or more sarcolemmal nuclei had often intruded (Figures 12 and 13). The sarcoplasm surrounding these vacuoles was in some instances hyalinized (Figure 14). In more chronic experimental cases, the size of the muscle fibres varied, some of the hyalinized fibres being smaller and showing proliferation and centralization of sarcolemmal nuclei. Ultramicroscopical studies showed that the vacuoles in the sarcoplasm were caused by the degeneration of myofibrils in otherwise intact-muscle fibres. The thick myofibrils were first to disappear, resulting in a loss of the A-band (Figure 15), followed by shredding of the remaining filaments, until ultimately all the myofibrils were destroyed (Figure 16). Fine granular material, containing remnants of myofilaments, Z-band material (Figure 16) and swollen vacuolated mitochondria, were evident in such fibres. As a result of the loss in myofibrils the diameter of some of the muscle fibres was reduced.113
Van der Lugt and Van Heerden168 described similar microscopical and ultrastructural lesions in the myocardium.168 In addition, they noted dissociation and fragmentation of intercalated discs.
The histopathology in the liver was marked by diffuse mild to marked degeneration of hepatocytes, with slight bile duct proliferation and fibrosis in the portal tracts.168
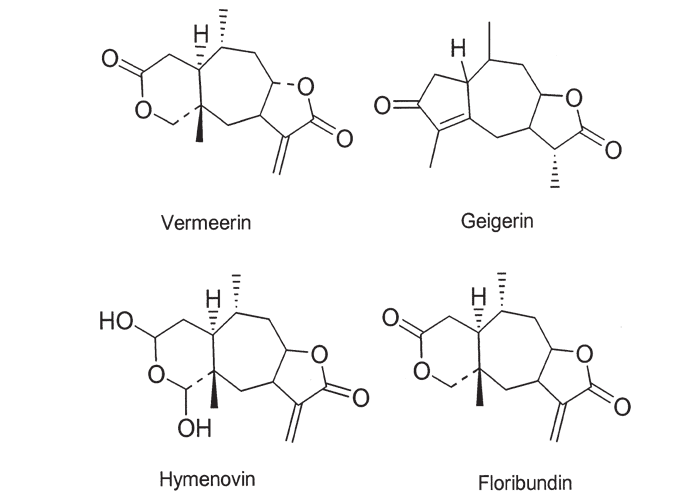
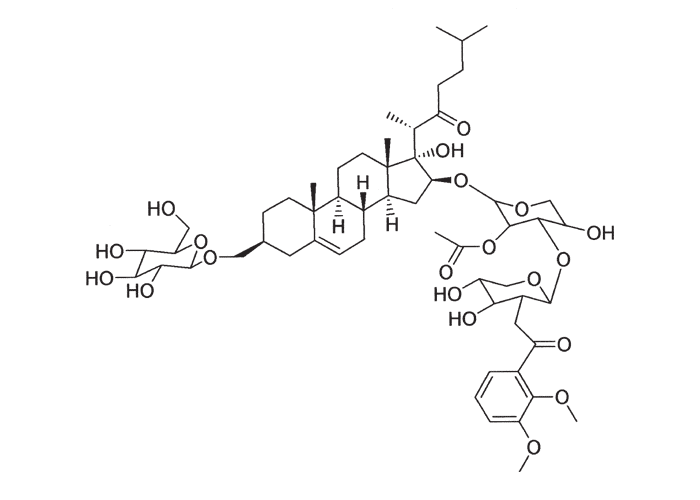
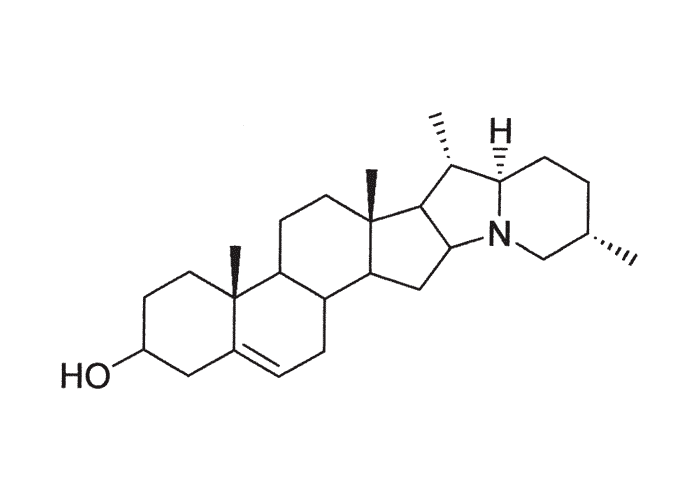
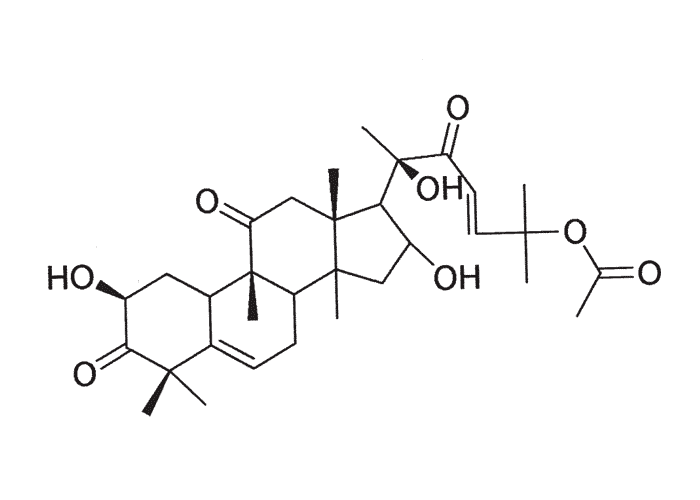
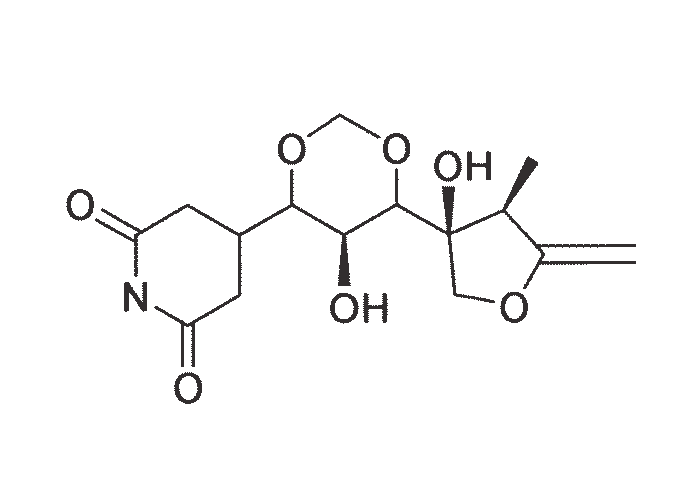
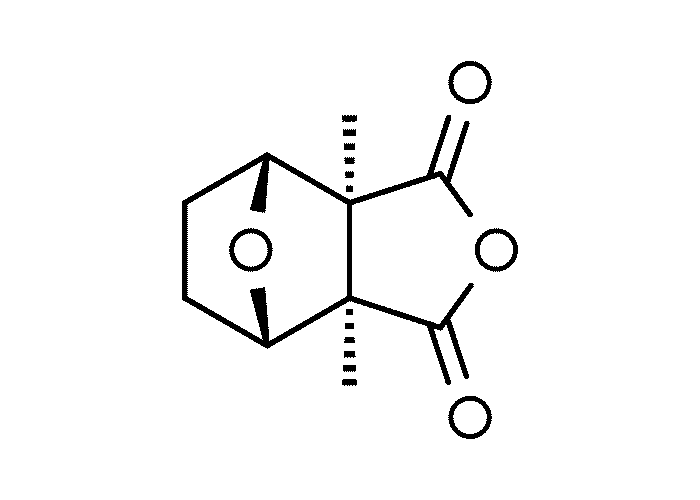
The active principles of Geigeria spp. have been identified as α,ß-unsaturated-γ-sesquiterpene lactones.2, 32, 33, 117, 128, 131, 173, 175 Structural formulae of two of them are given in Figure 17.
Rimington and Roets130 isolated geigerin, a neutral lactonic bitter principle (C15H20O4, anhydrous)35, 130 and its corresponding acid, geigeric acid, from G. aspera var. aspera. Although toxic to cats, geigerin produced no ill effects in the rabbit and the sheep.130 In the same year, Rimington and his co-workers recovered the true active principle from G. aspera var. aspera, a dibasic acid, vermeeric acid (C18H28O7), which passed over readily into the dilactone, vermeerin (C18H24O4)2, 130 Ten to 15 g of vermeeric acid given orally to sheep caused death from acute vermeersiekte within five to 24 hours.131 Vermeeric acid, inexplicably, has since defied all attempts to re-isolate it.55 In view of the association of vermeerin and floribundin with hymenovin, the toxic sesquiterpene lactone70 of Hymenoxys spp. in the USA, it has been suggested by a source cited by Herz60 that vermeeric acid might be identical with hymenovin, or that vermeeric acid and vermeerin could bear the same relationship to each other as hymenovin to floribundin (Figure 17).60 Several sesquiterpenoid lactones have been isolated from Geigeria spp., namely, vermeeric acid,131 vermeerin,131 geigerin,7, 128, 175 geigerinin,32 ivalin175 and dihydrogriesenin173, 175 from G. aspera var. aspera; vermeerin,2 gafrinin,34 griesenin and dihydrogriesenin26 from G. ornativa. Save for vermeeric acid, no experimental evidence has yet been produced that any of these sesquiterpene lactones can cause vermeersiekte. Von Jeney de Boresjenö et al.176 have reported on the detection by thin layer chromatography of sesquiterpene lactones from G. aspera var. aspera.
Laboratory animals, such as rabbits, guinea-pigs and rats, are reported to be fairly resistant to poisoning by sesquiterpenoid lactones55, 128, 130, 131 but recent findings have indicated that they may be more susceptible than originally suspected. When geigerin, vermeerin, gafrinin, dihydrogriesenin, ivalin and tetrahydrogriesenin were injected subcutaneously into mice, all the compounds having an α,ß-unsaturated-γ-lactone ring were toxic at doses of c.0,25 g/kg. Geigerin and tetrahydrogriesenin, without the α, ß-double bond, on the other hand, were non-toxic at 0,5 g/kg. This finding is consistent with reports in the literature that the activity of the sesquiterpenes resides mainly in the α,ß-unsaturated lactone ring. Similar results were obtained with guinea-pigs. The signs of sesquiterpenoid lactone poisoning in laboratory animals took the form of seizures, paresis and paralysis. Vermeersiekte was induced in a sheep by oral administration of an alcoholic extract of G. aspera var. aspera that contained dihydrogriesenin, geigerin and ivalin (N.M.J. Vermeulen et al., University of Pretoria, unpublished data, 1982). Frogs reportedly suffered fatal paralysis and respiratory distress after the injection of small quantities of a vermeeric acid-containing ether extract of G. aspera var. aspera into their dorsal lymph sacs.131
Some progress has been made in the treatment of ovine sesquiterpene lactone poisoning induced by composite plants, such as Helenium spp. and Hymenoxys spp. the cause of so-called spewing disease in the USA.136 The cytotoxic and antitumour activity of many sesquiterpenes have been linked with the α,ß-unsaturated ring as well as other functions potentially able to alkylate sulphydryl groups of key enzymes.57, 60, 85 Exocyclic methylene groups of the α,ß-methylene lactone moiety is believed to react irreversibly with the sulphydryl group of L-cysteine residues of enzymes or other nucleophiles.57, 77, 84 This mechanism of action prompted the experimental use of cysteine as treatment for sesquiterpene lactone poisoning in sheep.77, 136 Cysteine administered i/v significantly increased the survival time of sheep injected i/p with hymenoxin,136 but in South Africa, cysteine, sulphur or sodium thiosulphate, given orally with daily doses of G. aspera var. aspera, did not prevent ovine vermeersiekte.72
More promising results have been obtained in the prophylactic treatment of experimental Hymenoxys odorata DC. (bitter-weed) poisoning of sheep in the USA by including an antioxidant, ethoxyquin in their diets.77 Ethoxyquin (EQ) is thought to exert its antidotal effect by the induction of certain enzymes and acid-soluble thiols in the liver. The reported adverse effects of EQ treatment, namely, lowering of serum albumin and calcium levels and alkaline phosphatase activity, can be countered by the addition of a methionine hydroxy analogue (MHA) (= 2- hydroxy-4-(methylthio) butyric acid) in the food. EQ was once held to be the first antidote to bitter-weed poisoning with potential application in the fiel.78 However, in limited trials with G. ornativa (n=3)171 and G. aspera (n=2) (N. Fourie , OVI, unpublished data. 1991) ethoxyquin given in conjunction with MHA failed to prevent the development of clinical signs. Neither did the administration of either thioctic acid or vitamin E and selenium have a demonstrable therapeutic effect in the treatment of experimental G. aspera poisoning (N. Fourie, OVI, unpublished data, 1991). Fourie suggested that ethoxyquin and MHA may not have been effective because Geigeria spp. are more toxic than Hymenoxys spp.
Some farmers and veterinarians claim that piracetam (‘Nootropil’, UCB Pharmaceuticals), injected i/v, can enhance the recovery rate of sheep suffering from vermeersiekte. Piracetam (2-oxo-1-pyrrolidine acetamide) can be regarded as a cyclic derivative of the inhibitory neurotransmitter, γ-aminobutyric acid (GABA) in the brain. The therapeutic effect of piracetam, however, could not be confirmed by Joubert72 in a pilot trial involving the treatment of four sheep with experimentally induced signs of vermeersiekte.
In addition to alkylation of sulphydryl groups, sesquiterpene lactones of G. aspera var. aspera have been shown to inhibit mitochondrial respiration165, 166 and the in vitro activity of key glycolytic enzymes.51
Certain Geigeria spp. contain non-toxic flavones28, 131 which, although apparently not involved in the pathogenesis of vermeersiekte,131 are responsible for inhibition of respiration and uncoupling of oxidative phosphorylation. The unexpected lack of toxicity of the flavanoids for mice can probably be ascribed to their efficient excretion from the body as glucuronides. Two of the flavanoids have been characterized.28
Some believe that treatment of stock with vermeersiekte is futile, since cured animals cannot be prevented from taking in more vermeerbossie on extensively infested veld. In their view, the solution to the vermeersiekte problem lies not in therapy but in improved pasture management, since outbreaks of the disease are virtually confined to veld denuded of grass cover by overgrazing, trampling, injudicious burning and poor management.105 Subdivision of farms into uneconomic units has exacerbated the situation.55
Certain characteristics of G. ornativa may help to explain the correlation between vermeersiekte and veld mismanagement. Vermeerbossies on the average produce 1 855 seeds with a germination potential of 80 to 90%, and the seeds can remain viable for up to 13 years, although they must undergo a post-ripening of 18 months before germination. Geigeria ornativa seeds need as much moisture for germination as those of the grasses, but the plants are less drought resistant than Themeda triandra, the climax grass of Griqualand West. They also do not germinate in a dense grass cover. Geigeria ornativa, therefore, cannot maintain itself in a vigorous stand of grass (Schijff, cited by Grosskopf55). Prevention of vermeersiekte in the long term can best be achieved by good farm planning that provides, among others, for the subdivision of farms into the optimal number of camps, sufficient strategically placed watering points, elimination of overgrazing and burning, and the institution of an appropriate rotational grazing system.105 Rotational grazing systems, however, cannot always be easily applied everywhere, particularly on the limestone soils of the Ghaap plateau in South Africa where the soil is shallow and the substratum consists of a hard calcrete formation. Here the perennial grasses and shrubs are limited to a few areas with deep soil, while pioneer plants such as Geigeria spp. occupy the majority of the plateau. Taken in small amounts, vermeerbos, being high in protein, can be very nutritious.55
In the short term, vermeerbossie can be controlled by the tactical grazing of infested pastures by sheep. In one variation of this system a large camp is set aside which is relatively free of G. ornativa or has been cleared of the plant. The other camps are then non-selectively grazed by a large flock of sheep at about four to five times the carrying capacity of the veld, until all the vermeerbos has been removed. The objective is to force the sheep to eat the vermeerbossie without individuals taking in a toxic dose. Should some of the sheep become poisoned, the flock is temporarily moved to the cleared camp for a week or so to recuperate. The procedure is then repeated camp by camp until all the vermeerbossie has been cleared. Vermeerbossie should preferably be eradicated in the preflowering stage before the seeds are formed.157 Alternatively, a short-term rotational grazing system can be practised in which a flock is kept approximately two weeks ‘on’ and two weeks ‘off’ an infested pasture. The aim is to move the sheep from the Geigeria before signs of vermeersiekte can appear and allowing them to ‘recover’ from possible subclinical intoxication before returning (T.W. Naudé & T.S. Kellerman, OVI, personal observation, 1991).
Snyman and co-workers144 demonstrated that cognitive aversion to vermeerbos can be induced and maintained in sheep by continuous exposure to an aversive mixture. This conditioned feed aversion was not abolished even by cohabitation with non-averted sheep invoking peer group pressure.144
Cattle farmers are in a less favourable position with regard to the eradication of vermeerbos; for without sheep to eradicate the bush, vermeersiekte can be a problem, especially in cattle with high nutritional requirements such as cows on dairy ranches.157
The prospect of eradicating vermeerbos by means other than high pressure grazing with sheep is not very promising. Pulling out the plants by hand is costly and of temporary benefit, as the cleared areas quickly become re-infested by the seeds that have lain dormant on the ground for years. Hormonal weed-killers do not kill the seed or plants growing in the shelter of bushes. And although G. ornativa is parasitized by several indigenous insects, including a weevil (Larinus peregrinus), a scale insect (Monophlebus sp.), a mealie bug (Pseudococcus sp.) and two fruit flies (Urophora hemixantha and Terellia planiscutelata), there is little hope that any of them will become an effective agent for the control of vermeerbos.55
Geigeria aspera var. aspera, the vermeerbossie on overgrazed Highveld areas of South Africa, is relatively easy to control, as the high rainfall of this region allows the grasses to respond quickly to improved management.55
Vermeersiekte is mainly a disease of sheep, and all breeds, sexes and ages (barring suckling lambs) are affected.40, 55 Despite some evidence to the contrary,40 it is generally accepted today that the Merino and the Karakul are the breeds most frequently affected, followed by Dorpers, Persians and nondescripts, more or less in that order.55 The variation in susceptibility of breeds probably arises from differences in their grazing habits and nutritional requirements. Sheep with the highest nutritional demands are those most at risk, e.g. young actively growing stock, ewes with suckling lambs, pregnant ewes or heavy wool producers, because they eat the most.55 Du Toit40 found no relationship between wireworm infestation and the incidence of vermeersiekte, but under natural conditions verminous sheep can be expected to be less able to resist the onslaught of the disease than non-infested ones.55 Imported and local sheep are equally susceptible,40 probably because the stand of vermeerbos is usually too dense for them to avoid ingesting the plant.55 The prevalence of vermeersiekte might be lower in goats, since they utilize herbage unavailable to sheep and thus taking in less vermeerbos.55
The death toll does not reflect the true losses inflicted by vermeersiekte as hidden costs such as diminished milk production, ill-thrift, reduced wool production and a poor lambing percentage, are not taken into account.55, 76
Kikuyu poisoning
Pennisetum clandestinum Chiov. (Poaceae)
Kikuyu grass
This condition predominantly of cattle grazing on kikuyu grass pastures is characterized by anorexia, depression, copious drooling, ruminal atony and distention, dehydration, sham-drinking, constipation and incoordination. Kikuyu poisoning is classified according to whether or not it is associated with army worm (Spodoptera exempta)108 infestation. In South Africa, army worm-associated kikuyu poisoning has been diagnosed only in cattle, whereas the non-associated form also affects sheep.
Kikuyu poisoning associated with army worm infestation
All published accounts of kikuyu poisoning in South Africa15, 16, 108, 170 have been associated with previous invasion of kikuyu grass by army worm. A similar association between the occurrence of outbreaks and previous invasion by army worm, Mythimna separata (= Pseudoletia separata), has been reported in New Zealand.143
The adult army worm moth is about 38 mm across the outstretched wings, with a dark-brown head, thorax and forewings, and white hind wings (Figure 18 a).142 They are believed to breed regularly in East African countries, such as Kenya, Tanzania and the Sudan. The eggs, laid on succulent grass by the female, hatch in two to 11 days depending on the weather. The larvae pass through five to six instars before entering the soil to pupate in fragile cocoons about 40 mm below the surface.5, 15 Larvae are said to require a temperature of 24 to 32 °C to develop,15 and a life cycle is completed in two to four weeks.5, 15
Annecke and Moran5 describe a fully grown larva or caterpillar as being 25 mm long. Initially greyish-green, they become darker in later instars with longitudinal blue lines in the middle, greenish lines on either side, and blackish lateral lines (Figure 18 b).5 Army worm caterpillars can be distinguished also by their large numbers and active disposition. In South Africa, almost the entire army worm population dies out between outbreaks, probably leaving only small islands of surviving insects in certain areas, e.g. along waterways in the Mpumalanga lowveld.5
The sudden explosive outbreaks of army worms in South Africa are generally believed to arise, not from increases in the size of the small incipient surviving local populations, but from massive migrations of moths from the north.5 Joubert speculated that:
‘successive generations of alates move southward with the aid of wind around high-pressure systems. They travel as far as the winds will allow, then breed, feed, pupate, eclode and wait in the grass for northerly winds. Thus, through November to March they move across Tanzania, Zambia, Zimbabwe and finally into Mozambique. From the uninhabited marshes and other green areas of the Mozambican coastal plains (or parts of Zimbabwe) they move into the Transvaal, Swaziland and Natal, again assisted by the northerly warm winds of a high-pressure system. Those that breed in Natal continue the southerly migration and end up in the lndian Ocean.73’
Moths are said to be blown for hundreds of kilometres at a stretch by these winds.
Cattle usually become affected on pastures that have been damaged by army worm some ten or more days previously.15 One of the earliest clinical signs that can be observed is drooling of strings of saliva from the mouth. Affected individuals tend to congregate around drinking troughs, but do not actually drink. Careful observation will show that, although they dip their mouths into the water, they do not swallow (sham-drinking). Eventually their eyes become sunken and other signs of dehydration are apparent (Figure 19). Moderate ruminal distention and ruminal atony are regularly present in affected animals. A marked feature in most cases of kikuyu poisoning is the accumulation of watery fluid in the rumen which may be so voluminous that it can be heard to slosh about as the animal walks. Signs of colic, such as grinding of teeth, kicking and looking at their flanks, are common. Faeces are seldom voided, but constipation is not invariable. Field outbreaks have been reported where moderate diarrhoea was recorded. Some animals might be hypersensitive, have a muscular twitch and walk with a high-stepping gait. Others develop limb weakness and apparent incoordination, characterized by a swaying gait, dragging of the feet and knuckling over. The more seriously affected animals become recumbent and often lie with their legs awkwardly disposed and their heads turned back to the flanks. Gushing of watery ruminal content from the nostrils and mouth is sometimes seen at death.15, 16, 100, 108, 170
Clinical signs have been observed within 24 hours of commencement of grazing on pastures15 or the ingestion of toxic, fresh kikuyu grass in feeding trials (R. Mapham, Veterinary Laboratory, Grahamstown, personal communication, 1977). The course of the disease is usually two to seven days, with most deaths occurring 48 hours after onset of signs.108 Up to 80% of the affected animals can be expected to die.15, 108
According to Bryson,15 opinions differ as to the reason for the inability to swallow. Van Heerden et al.170 reported that the tongues of the affected cattle examined were paretic. Bryson and Newsholme,16 however, found the tonus of the lingual muscles of the cases examined by them to be normal and concluded that either pain in the pharynx or bulbar paralysis could be responsible for the aphagia/adipsia. Detailed examination of the pharynx revealed no apparent lesions.16
The gross pathological changes of kikuyu poisoning include distension of the forestomachs and abomasum by watery ingesta, necrosis and ulceration of the mucosa of the forestomachs (especially of the omasum) (Figures 20 a and b), and constipation.15, 108 The necrosis of the mucosa of the forestomachs is the most consistent and most extensive lesion of the disease. Observation of epithelial necrosis in all the sections of the rumen, reticulum and omasum examined by Newsholme et al.108 in one outbreak indicated that the lesion was extensive and not confined to the grossly recognizable necrotic areas. They further reported that although complete ulceration had occurred in some sections, necrosis was most extensive in the superficial layers. The stratum spinosum and stratum granulosum were selectively involved, while the stratum basale was generally preserved. Electron microscopical examination of ruminal and omasal epithelium from two cattle revealed cytopathological features in the stratum spinosum and stratum granulosum consistent with stages in acute anoxic types of injury.108
The aetiology of kikuyu poisoning remains obscure. Various agents have been suggested as possible causes of kikuyu poisoning: Steyn158 proposed that the saliva of Spodoptera exempta caterpillars on infested pastures might be involved in the toxicity; Bryson15 that army worms altered the composition of grass rendering it toxic. This toxicity, he suggested, could be exacerbated by fungi. Evidence was presented by Martinovitch et al.101 that kikuyu poisoning in New Zealand could be a mycotoxicosis. When dosed to cattle and sheep, cultures of a Myrothecium sp., a pastoral fungus, induced clinical and pathological signs similar to those of kikuyu poisoning.36, 107 Cultures of M. verrucaria, isolated from kikuyu grass which had been incriminated in an outbreak of kikuyu poisoning, and cultures of Phoma herbarum from lucerne, had a similar effect.36
In South Africa potentially toxic fungi, such as Rhizoctonia, Myrothecium, Fusarium and Phoma sp., have been isolated from leaves of kikuyu grass and from the faecal pellets of army worms collected on toxic pastures during outbreaks of kikuyu poisoning. Since there was little visible evidence of fungal growth on the pastures, the colonies on the cultures are believed to have developed from spores that had adhered to the grass. The faecal pellets were present in such small quantities that they were difficult to find and lay mostly on the ground where they were unavailable to cattle. Considering all the evidence at hand, therefore, it seems improbable that fungi could have played a significant part in the aetiology of these particular outbreaks of kikuyu poisoning in South Africa.108
Army worm carcases have been fed to sheep (T.S. Kellerman, OVI, unpublished data, 1973) and cattle (J.G. Tremlett, Veterinary Research Laboratory, Kabete, Kenya, personal communication, 1973) without ill effect. Moreover, since army worms are generally absent from pastures during outbreaks, they could not be directly incriminated in the intoxication.108
Migrating caterpillars can be prevented from travelling from one land to another by drawing a furrow across their line of advance with the flat, mould-board side of the share facing towards them. The caterpillars crawl along the bottom of the furrow and fall into pits, dug at intervals along its length, where they can be dusted with insecticide and buried (R.A. Bell and R.W. Bryson, Entomology Section, Cedara Agricultural College, personal communication, 1981). Chemical control should be carried out as promptly as possible as the first and second instars are more susceptible to poisoning than subsequent ones. The insecticides that have been registered for use against army worm in South Africa are carbaryl, chlorpyrifos, alpha-cypermethrin, cypermethrin, deltamethrin, mercaptothion, triclorfon, methomyl and trichlorfon.12, 82 Care should be taken to apply the insecticides as directed by the manufacturer, as some of them are very dangerous to livestock.
From the point of view of the control of kikuyu poisoning, it is important to note that the chemical eradication of army worm on a pasture does not necessarily render it safe for grazing, and that lightly infested pastures can be as dangerous as heavily infested ones. Pastures do not become toxic immediately upon invasion by army worm, but a variable period of about ten days or so has to elapse for toxicity to develop. North-facing, partially shaded pastures, situated on inclines, are supposed to be particularly hazardous.
Cattle must be removed from affected pastures as soon as the first signs appear. Before a poisonous pasture is re-utilized a few less valuable, ‘tracer’ cattle should be allowed to graze on it for at least 96 hours. If no signs develop, the numbers can be gradually increased to optimal levels. The only fairly sure way of preventing deaths is to withdraw cattle from army worm-damaged pastures for at least 40 days.
Sheep have not been positively linked with army worm-associated kikuyu poisoning involving S. exempta in South Africa,15 although they have been known occasionally to contract the disease abroad where a different species of army worm is present. The rarity of kikuyu poisoning in sheep may be attributed at least in part to their more selective grazing habits.99
To the best of our knowledge, kikuyu poisoning has never been induced by hay or silage prepared from toxic pastures.
In New Zealand, a parasitic wasp, Apanteles ruficrus, which was introduced from Pakistan, reduced the army worm (Mythimna separata) population to some extent. The biological control provided by this wasp, however, did not completely prevent the occurrence of kikuyu poisoning.98 In South Africa, the caterpillars of Spodoptera exempta can be heavily attacked by local parasitic wasps and flies.5
While it is true that kikuyu poisoning has not been diagnosed on army worm-damaged pastures other than kikuyu grass, it must be emphasized that not all invaded kikuyu pastures are toxic. Given the high production potential of kikuyu, it would be unwise to discourage the establishment of this grass in order to prevent the sporadic, localized, though highly destructive, outbreaks of the disease.
Kikuyu poisoning not associated with army worm infestation
A disease, virtually indistinguishable from kikuyu poisoning, has been described in stock grazing on kikuyu pastures not affected by insect damage in Australia50 and South Africa (vide infra).74 Several reports have been received from various parts of South Africa of both cattle and sheep developing signs of kikuyu poisoning on pastures not previously infested by army worm.74
The possibility has been mooted that this syndrome was a metabolic disorder induced by the accumulation of ammonia ions in the blood (A. Immelman, Faculty of Veterinary Science, University of Pretoria, personal observation, 1984) as a result of some ruminal disturbance. Although acute ruminal indigestion and alkalosis have been suspected of being involved in the pathogenesis of an army worm-related outbreak in New Zealand,29 alteration in rumen pH is not a feature of kikuyu poisoning in South Africa. In South Africa heavily fertilized lush-growing formerly drought-stricken kikuyu grass, rich in ammonium salts and nitrates and deficient in energy, is thought to be a predisposing factor for the non-army worm-associated form of the disease74 (D.J. Schneider, Regional Veterinary Laboratory, Stellenbosch, personal communication, 1994). Very similar conditions have been implicated in the occurrence of kikuyu poisoning in Australia.180
Investigations into the reduction in animal performance on fertilized kikuyu pastures in late summer (or ‘autumn slump’) revealed high levels of soluble nitrogen, nitrates, and oxalates in the grass.114, 115 This was particularly evident in young actively growing sward. Moreover, relative to the requirements of most classes of cattle and sheep, kikuyu herbage was found to be very high in K+, while being deficient in Ca2+ and Na+104. The veterinary implications of these findings for stock grazing on kikuyu are several. Nitrate poisoning is indeed occasionally diagnosed in stock grazing on heavily fertilized kikuyu pastures in South Africa. Although overt oxalate poisoning has not been recorded (T.S. Kellerman, OVI, personal observation, 1997), oxalates could have a negative impact on Ca absorption by stock grazing on kikuyu, a grass already deficient in this mineral. According to Miles et al.,104 the Ca concentrations and Ca:P ratios of kikuyu frequently fall below published critical limits. Among the expected detrimental effects of the low Ca:P ratios on livestock is the formation of urinary calculi in sheep and induction of milk fever in cows. Excessively high K levels in herbage will, by inhibiting absorption of Mg, also predispose animals to hypomagnesaemic grass tetany.
Poisonings with diarrhoea
Chinkerinchee poisoning
Chinkerinchee poisoning is characterized by severe purgation accompanied specifically in cattle by blindness.
Ornithogalum thyrsoides Jacq. (Hyacinthaceae)
Chinkerinchee, Star-of-Bethlehem, viooltjie
The small, spherical, white bulbs, 20–40 mm in diameter, are covered by papery scales. Four to five strap-shaped leaves, 150–450 mm long and 10–40 mm broad, are arranged in a basal rosette (Figure 21). The peduncle is usually single, stiffly erect, and 150–450 mm long. Beautiful, star-shaped, white flowers with brownish-green centres are grouped at the top of the peduncle (Figure 22) and the floral bracts have no appendages or spurs. The fruits are three chambered and contain many seeds.31, 118, 164
Ornithogalum thyrsoides is endemic to the Winter Rainfall Area of the Western Cape Province (Figure 23) but is widely cultivated throughout southern Africa for its long-lasting, beautiful pyramid-shaped flowerheads.164 In its natural habitat, O. thyrsoides favours damp places, such as vleis or the banks of streams (Figure 24).164
Ornithogalum saundersiae Baker (Hyacinthaceae)
Transvaal chinkerinchee
This chinkerinchee is a much larger plant than O. thyrsoides. According to Vahrmeijer,164 O. saundersiae has a white bulb about 60 mm in diameter which is surrounded by soft papery scales. The leaves are 30–60 mm wide and up to 500 mm long (Figure 25). The single, erect inflorescence is about 1 m tall with the flowers arranged in an inverted pyramid (corymb) at the top (Figure 26). The striking pale-yellow flowers are star shaped, with prominent distinctive black ovaries that turn green after fertilization (Figure 26). The three-chambered fruits contain many flat, black seeds.164
Ornithogalum saundersiae is found in the Mpumalanga Province, Swaziland, and northern KwaZulu-Natal (Figure 27),152, 164 where it grows on mountain slopes, in vleis, on stream banks, in the shade of trees and on open veld.164 Like O. thyrsoides, it is often grown in gardens for its flowers.
Ornithogalum spp. are among the most poisonous plants in South Africa, the dried material having toxicities measured in mg/kg rather than g/kg live mass.
Several Ornithogalum spp. have been identified as toxic, including O. conicum Jacq. subsp. conicum (= O. lacteum Jacq.),156 O. flexuousum (Thunb.) U.Müll.-Doblies & D.Müll.-Doblies (= O. ornithogaloides),156 O. prasinum Lindl. (H.E. van der Pypekamp, T.S. Kellerman and N. Fourie, OVI, unpublished data, 1986), O. saundersiae Bak.,121 a species identified as O. pilosum L.f. (= O. tenellum),30, 31 and O. thyrsoides Jacq.68, 69 The chinkerinchee species that usually poison stock in the Winter Rainfall Area are O. thyrsoides and O. conicum,159 while in the summer rainfall areas O. saundersiae, O. flexuousum and O. prasinum are potentially important. The two most beautiful chinkerinchees, O. thyrsoides and O. saundersiae, are widely used as ornamental plants and cause losses throughout the country wherever stock has access to garden waste.
Ornithogalum thyrsoides came to the fore as a toxic plant in 1904 when cart horses died after eating oat hay contaminated with it near Wellington in the Western Cape Province.68, 111 Field outbreaks of chinkerinchee poisoning were reported shortly afterwards from Kimberley in horses that had been fed on O. thyrsoides contaminated hay imported from the Wellington area.69 Feeding trials with suspect material revealed the remarkable toxicity of this plant: horses were fatally poisoned, respectively, by eight dried flowerheads,68 less than 680 g of fresh flowerheads, and 170 g of half-dried leaves.69
Ornithogalum saundersiae was shown to be toxic by Quin,121 the dried, powdered bulbs having been administered to different animals with the following results: guinea-pigs, 0,125 g induced death in three to four days; rabbit, 0,5 g was fatal in 24–48 hours; dog, 0,5 g was fatal in four days; sheep, 1 g induced signs lasting more than a week; horse, 5 g was fatal in three days; cattle, 10 g caused marked persistent signs without death.121
Poisoning induced by Ornithogalum toxicarium, which differs from the above in being krimpsiekte-like, is discussed in Cardiovascular system.
Toxic cholestane glycoside have been isolated from O. saundersiae, O. thyrsoides and O. prasinum (Figure 28).83, 169
In a typical outbreak of chinkerinchee poisoning during August/September 1962 near Malkerns in Swaziland, 70 out of 126 head of cattle developed signs, 15 with fatal results. The herd had grazed on broken veld heavily infested with O. saundersiae that grew abundantly in the shade of trees or between rocks. According to the owner, almost all the severely intoxicated cattle became temporarily blind. Fresh, green leaves of O. saundersiae from the toxic camp were dosed at the rate of 2,5 g/kg on two successive days to a young bull at the Veterinary Research Institute, Onderstepoort. On the third day, the animal developed a severe watery to haemorrhagic diarrhoea that persisted for 13 days, despite symptomatic treatment. The bull then went blind, its sight gradually returning to normal between the fourth and eighth weeks.
A single dose of 5 g/kg of green O. saundersiae was fatal for a bull, while 5 g/kg on two successive days killed a sheep (M. Terblanche and T.F. Adelaar, OVI, unpublished data, 1963). Limited dosing trials with rabbits suggested that the toxicity of the aerial parts of O. saundersiae diminishes in the post-flowering stage (M. Terblanche, OVI, unpublished data, 1964).
Sporadic outbreaks of diarrhoea and blindness in cattle near Rustenburg in the North West Province prompted an investigation of the toxicity of some of the Ornithogalum spp. of the bushveld. Ornithogalum seineri and O. tenuifolium were dosed to sheep with negative results, but O. prasinum (Figures 29 and 30) proved to be very toxic (H.E. van der Pypekamp and T.S. Kellerman, OVI, unpublished data, 1986).
Fresh O. prasinum leaves and bulbs from a toxic camp induced diarrhoea and blindness in a heifer at a dose of 1 g/kg. Both the diarrhoea (which developed within 24 hours) and the blindness (which was noticed after 14 days) persisted until the animal was destroyed on the eighteenth day. During the course of the experiment the heifer lost 61 kg in body mass. Sheep could be fatally poisoned by as little as 1 g/kg of the fresh material (H.E. van der Pypekamp, N. Fourie and T.S. Kellerman, OVI, unpublished data, 1986).
Very little specific information is available on the toxicity of O. flexuousum (Figure 31), the vlei or grass chinkerinchee of the summer rainfall area (Figure 32). The leaves of this little chinkerinchee are not easily distinguished from those of the surrounding grasses of the swampy areas in which they grow. To identify this plant when it is not in flower, a few sods should be turned to expose the small (20 mm diameter) bulbs.155
Half a kilogram of fresh flowers and stems of O. pilosum (vide supra) caused the death of a horse in three days,30, 31 and 900 g of fresh flowering O. conicum (= O. lacteum) was fatal to a sheep.147 Ornithogalum pilosum, like O. flexuousum, particularly favours marshy ground.164 In Kenya, O. longibracteatum Jacq. has been incriminated in the poisoning of stock.103 Several local Ornithogalum spp., including O. multifolium (= O. auranticum), O. longibracteatum (= O. caudatum), O. tenuifolium (= O. ecklonia),147 and O. seineri (H.E. van der Pypekamp and T.S. Kellerman, OVI, unpublished data, 1986), in contrast, have been dosed to animals without ill effect.
The beginning of intoxication is marked by anorexia and depression of such severity that affected animals have been described as ‘drowsy’ or having the appearance of being drugged; followed by a transient febrile reaction, tachycardia, polypnoea, abdominal pain and diarrhoea.30, 31, 68, 69, 121, 154, 158, 159 The most important and most constant feature of chinkerinchee poisoning is a severe, foetid, very watery to slightly haemorrhagic diarrhoea, starting about 24 hours after ingestion of the plant and persisting for up to three weeks. Death usually occurs in about two to five days, but if large amounts of chinkerinchee are taken in, animals may collapse and die without showing any signs. In addition to the usual clinical signs, cattle may become temporarily or permanently blind. The blindness can be described as an amaurosis, since – apart from a reported lack of pupillary reflexes and miosis that has not been experimentally confirmed – no lesions are apparent in the eyes158, 159 (T.S. Kellerman, unpublished data, 1986). Limited experimental evidence based on three animals indicates that the blindness commences about 10–14 days after ingestion of the plants and that vision can remain impaired either permanently or for up to eight weeks (T.S. Kellerman, unpublished data, 1986). It is not known what percentage of intoxicated cattle become blind in the field but, according to farmers, the numbers may be considerable (T.S. Kellerman, T.F. Adelaar, T.W. Naudé and M. Terblanche, OVI, unpublished data, 1963–1986). Van der Lugt (Faculty of Veterinary Science, University of Pretoria, unpublished data, 1997), reports that dogs poisoned by chinkerinchee bulbs can also lose their sight.
The most important lesion of chinkerinchee poisoning is a catarrhal to haemorrhagic enteritis. The enteritis is usually of a catarrhal nature and the intestinal content is fluid and foetid. The lungs are occasionally congested or oedematous, and degenerative changes have been reported in certain organs.68, 69, 154, 159 According to Steyn, damage to the retina, which he did not specify, could be so severe that cattle are permanently blinded.158, 159 In a recent dosing trial with O. prasinum (vide supra), however, microscopical lesions could not be demonstrated in the eyes, optic chiasma or optic nerves of a sightless heifer. The only detectable change in this case was mild cavitation of the white matter of the globus pallidus, putamen, capsula interna and capsula externa. Necrosis of lymphocytes and lymphoid tissue has also been observed.167
Chinkerinchee poisoning has been experimentally treated with some success by the administration of industrial activated charcoal in doses of 2 g/kg live mass. The activated charcoal eliminated all the signs of poisoning, including diarrhoea, but did not prevent blindness (T.S. Kellerman and N. Fourie, OVI, Onderstepoort, unpublished data, 1986). If highly adsorbent activated charcoal is not available the clinical signs can be treated symptomatically with antidiarrhoeal preparations and electrolytes (M. Terblanche and T.F. Adelaar, OVI, Onderstepoort, personal observations, 1963). Vleis in which chinkerinchee plants are likely to grow should be avoided as grazing and hay must not be made on fields infested by the plants.158
The blindness induced by chinkerinchees in cattle can be distinguished from that of Helichrysum argyrosphaerum or lead poisoning, and poisoning by overdosing with certain halogenated salicylamide anthelmintics, by the accompanying violent diarrhoea. Chinkerinchee poisoning may, in addition, be confused with conditions affecting the gastrointestinal tract, such as cardiac glycoside or acute arsenical poisoning.
Livestock in South Africa are most often poisoned, not by wild chinkerinchee plants growing in the veld, but by their highly palatable bulbs in garden waste.
Ricinus communis L. (Euphorbiaceae)
Castor oil plant, kasterolieboom
This well-known cosmopolitan weed is a much-branched shrub or small tree growing up to 4 m in height with grey-green or reddish stems prominently marked by leaf scars (Figure 33). Large hand-shaped leaves, 300 mm or more in diameter, with closely serrated edges, arise alternately from the stem on long petioles. The veins of each leaf radiate from the insertion of the petiole through the middle of each of the lobes or ‘fingers’ (Figure 33). The flowers are unisexual, with the male flowers arranged at the lower and the female flowers at the upper end of the inflorescence. The fruits are spiny (Figure 33), three-lobed capsules about 10–150 mm in diameter, each containing a hard glossy seed somewhat resembling a gravid tick (Figure 34).58, 164
The castor oil plant is a widespread weed in southern Africa along roadsides, cultivated lands and other disturbed places. It is grown as an ornamental or as a crop-plant58, 164 for production of castor oil which is used as an industrial lubricant or for medicinal purposes.
The literature on the toxicity of castor beans has been competently reviewed in several textbooks.23,42, 79, 126, 154, 158, 178 The toxic principle, ricin, is a toxalbumin (lectin), which, unlike snake venoms, may survive the action of proteolytic enzymes in sufficient quantity to be absorbed from the gut.22, 23, 47, 49, 79, 124, 154 Ricin, being a protein, is antigenic and repeated ingestion of small doses causes immunity. Immunized animals reportedly can tolerate up to 800 times the normal lethal dose of ricin.23, 79, 81, 124 Pure ricin is one of the most toxic substances known. Administered intravenously, it can be fatal to experimental animals at doses as low as 0,3 μg/kg.126 The parenteral toxic dose of ricin is conspicuously lower than the oral dose; for example, the intraperitoneal LD100 for mice reportedly is 25 000 times less than that of the oral route.24 The extreme toxicity of parenterally administered ricin has presumably been exploited in at least one homicide. Knight81 described the assassination of a man in London, supposedly by poisoning with ricin. The method of administration was particularly bizarre; it is believed that the ricin, contained in a minute, perforated metallic sphere, was driven under the skin of the thigh of the victim with the point of an umbrella. After carrying out the assault in public, at a bus stop, the assailant made good his escape. The lesser toxicity of ricin along the oral route can probably be attributed to its digestion and/or poor absorption from the gut.81 Ricin is heat labile47 and a powerful agglutinant of erythrocytes even in defibrinated blood.81, 154
According to Olsnes et al.110 ricin is composed of two protein units A and B linked by a disulphide bond. Protein A allows the molecule to penetrate the cell wall, while B disrupts protein synthesis.
All parts of the plant are potentially poisonous, but the seeds are particularly toxic.81, 124 Castor beans have been reported variously to contain 0,2–1% ricin;24, 178 medicinal castor oil, on the other hand, is free of it. After expression of the oil from the seed, the ricin, which is insoluble in the oil, remains in the press cake, or pomace as it is also known.22, 23 The press cake can be detoxified by steam, which causes the toxalbumin to coagulate, thus rendering it less hazardous and suitable for use as an organic fertilizer.23, 79 Despite the extreme toxicity of ricin, castor bean poisoning rarely occurs in stock grazing on pastures infested by the plant.47 Poisonings usually result only from the ingestion of grains contaminated by castor seeds or stock feeds containing press cake.1, 23, 49, 91, 154 Ricinus communis plants often grow in or near lands where the seeds can easily contaminate crops, such as maize or beans. The danger of contamination is especially great when the harvested crop is stacked or threshed beside infested lands.154 Poultry has been poisoned by R. communis growing near their runs,178 the seeds reputedly being propelled up to 7,6 m by the capsules as they dehisce.81
Animal species vary in their susceptibility to poisoning by ricin: horses are most susceptible; sheep, cattle and pigs intermediately so; and ducks and poultry the least.22 Several authors, citing Frohner (1919), state that the lethal doses of castor seed in g/kg for various species are: horses, 0,1 g/kg; geese, 0,4 g/kg; sheep, 1,25 g/kg; pigs, 1,4 g/kg; cattle 2,0 g/kg; goats, 5,5 g/kg; fowls, 14,0 g/kg.23, 154 Individual animals within a species can vary greatly in their response to ricin,22, 49, 154 and the ricin content between seed-lots often differs markedly.124
The clinical signs of ricin poisoning usually appear abruptly a few hours to a few days after ingestion of the beans,23, 52, 91, 124 but high doses can lead to sudden collapse.154 The outstanding feature of castor bean poisoning in all species is a severe watery to haemorrhagic diarrhoea, usually accompanied by abdominal pain, inappetence, dullness, weakness and dehydration. These signs may be associated in cattle by tympany and a drop in milk yield; in horses by sweating, trembling and incoordination; in pigs by vomition; and in poultry by drooping of the wings, emaciation and a drop in egg production.1, 23, 52, 79, 91, 124, 154
The typical necropsy feature of ricin poisoning is a severe haemorrhagic to necrotic gastroenteritis and necrosis of lymphocytes in lymphoid organs (especially the mesenteric lymph nodes), similar to that seen in many viral infections. These lesions can be accompanied by widespread haemorrhage and oedema of the digestive tract and associated lymph nodes.22, 52, 91
Since ricin does not have distinctive chemical properties by which it can be identified easily,22 a laboratory diagnosis of Ricinus poisoning depends on the microscopical examination of feeds or stomach contents for fragments of the seeds,39 agglutination of red blood cells by extracts of suspectedly poisonous material, and a precipitin test. The precipitin test has the advantage of being specific but none of the tests are particularly satisfactory.22, 23 The best confirmation of a diagnosis is a positive response to the administration of immune serum. Although it is the ideal antidote, immune serum is not likely to be at hand under field conditions.23 Clark22 suggested that the administration of arecoline hydrobromide, followed by a saline purgative (as recommended for abrin poisoning), could be efficacious.
In South Africa, ricin poisoning most often results from the inadvertent inclusion of castor seed cake in stock rations. Because it is dark coloured it is readily confused with sunflower or cotton seed cake. Stereo-microscopical examination of contaminated rations reveals castor bean hull fragments with the characteristic porous structure absent in either sunflower or cotton seed hulls (J.A. Minné, OVI, personal communication, 1985).
Jatropha curcas L. (Euphorbiaceae)
Physic nut, purge nut
This is a small spreading tree about 4,5 m high with spear-shaped, slightly-lobed leaves up to 150 mm across. The flowers are inconspicuous; the fruits fleshy with three seeds c.12 mm in length.87
The leaves of Jatropha multifida, the popular ornamental ‘umbrella tree’, are large and deeply cut or ‘fingered’ (Figure 35).
The seeds of both species (Figure 36) contain curcin, a toxalbumin with an action similar to that of ricin.87, 158 According to Makkar and Becker97 these phorbolesters (glucosinolates) are the main toxins of J. curcas, although lectins and trypsin inhibitors may enhance their toxic effects.97 Children sometimes eat the seeds, of which three or fewer can apparently be poisonous.87, 158, 178 Calves have died within 19 hours of being dosed once with 0,25–2,5 g/kg of ground seed, and repeated administration of 0,025 g/kg/day was fatal in 10–14 days.3 Clinical signs appear a few minutes to a few hours after ingestion of the seed and these consist of diarrhoea, dyspnoea, dehydration and loss of condition.87, 158
To the best of our knowledge, authenticated outbreaks of Jatropha poisoning of livestock have not been recorded in South Africa; however, presumptive diagnoses of prussic acid poisoning have occasionally been made in ruminants browsing the leaves of Jatropha multifida, which is rich in cyanogenic glycosides (T.S. Kellerman, OVI, personal observation, 1987).
Abrus precatorius L. subsp. africanus Verde. (Fabaceae)
Love bean, lucky bean, minnie-minnies
Minnie-minnies is a perennial woody climber with compound leaves about 60–80 mm long, each having c.11 pairs of broad, oblong leaflets, 6–15 mm long by 3,5–7 mm broad (Figure 37 a). The most distinctive feature of this plant is the cluster of hairy pods (Figure 37 a), about 30 mm long and 13 mm wide. Ripe pods split, exposing decorative scarlet seeds with a black area at one end (Figure 37 b).118 The plant is common in the northern parts of the Limpopo Province and KwaZulu-Natal and the species has also been introduced into tropical areas abroad, e.g. in India and Australia.
The outer seed-coat is so indigestible that unbroken beans reputedly pass harmlessly through the digestive tract.158, 178 Less than one seed, thoroughly masticated, is said to be fatal for humans. The toxic principle, abrin, is a lectin consisting of two polypeptide chains joined by disulphide bonds.20 Abrin, like ricin, is antigenic, and this property is exploited in India, where stock are immunized with subclinical doses of seed before being put out to graze on Abrus precatorius-infested pastures. Horses have been fatally poisoned by 60 g beans, but cattle, sheep and goats are more resistant,158 probably because abrin is destroyed in the rumen. The clinical signs, resembling those of ricin poisoning, are preceded by a latent period of a few hours to a day or two.158 Post-mortem features include severe gastroenteritis, free blood in the digestive tract, haemorrhages in various organs, ulceration of the abomasal mucosa, haemoglobinuria, nephrosis and degenerative changes in the liver.
The mechanism of action of abrin is similar to that of ricin.110
An outbreak of suspected A. precatorius poisoning has been diagnosed in cattle near Thohoyandou in the Limpopo Province. A number of cows, unfamiliar with minnie-minnies, died after being introduced onto a pasture heavily infested by the plant. The minnie-minnies had been grazed, and the gross and microscopical lesions were consistent with those of abrin poisoning (J.G. Pienaar, Veterinary Laboratory, Potgietersrus, Monthly Report for February, 1986).
Modeccin, a toxalbumin produced by Adenia digitata, is mentioned under the section Discussion at the end of this chapter.
Solanum lichtensteinii Willd. (Solanaceae)
(= S. incanum)
Bitter apple, bitterappel
Shone and Drummond141 described S. lichtensteinii as a much-branched greyish, perennial, woody shrub, up to 1,2 m in height, with recurved spines on the branches and leaves (Figure 38). The alternate leaves are hairy beneath. The five-merous flowers are purplish like those of a potato. The unripe fruits are a mottled dark and pale-green. Ripe fruits are yellow (Figure 38), up to 38 mm in diameter and tomato-like, with a spiny pedicel and calyx. Solanum lichtensteinii is widespread in southern Africa.164
Solanum nigrum L. (Solanaceae)
Black nightshade, nastergal
This well-known Solanum is a herbaceous, erect, branched annual or biennial up to 1 m high. The leaves, arising alternately on long petioles, are lanceolate to ovate or elliptical, up to 80 mm long and 50 mm broad, bright green on both surfaces, and smooth or hairy. The drooping, potato-like flowers are white. The fruits are round berries, about 10 mm or less in diameter, that turn black when ripe58. Both S. nigrum and S. lichtensteinii are common throughout South Africa.58, 158
Solanum aculeastrum, S. linnaeanum (= S. sodomeum), S. lichtensteinii, S. nigrum, S. panduriforme and possibly S. pseudocapsicum have been shown to be toxic in South Africa.151, 158 These species display considerable morphological variation, ranging from the typically thorny S. lichtensteinii to the soft and thornless S. nigrum.
The toxicology of Solanaceae-glycoalkaloids has been reviewed by several authors.20, 21, 71, 106, 178 Cases of glycoalkaloid poisoning of livestock after ingestion of Solanum spp. have been reported in South Africa,154, 158, 178 but this type of poisoning is rare. The toxin is contained principally by the fruit of wild Solanum plants, green fruits being particularly toxic. The ripe fruits of S. nigrum are so innocuous that they can be eaten fresh or cooked in jams, cakes and desserts.152, 158, 178 Leaves have little or no glycoalkaloids.158
Solanine (Figure 39) is the commonest, though not the only, glycoalkaloid in Solanum spp. A glycoalkaloid consists of a steroid alkaloid moiety to which is attached a side chain of sugars.106, 178 Glycoalkaloids affect mainly the nervous system and the gastrointestinal tract. The nervous signs are attributed to inhibition of acetylcholinesterase, and the gastrointestinal effects mostly to the saponin-like properties of glycoalkaloids.20, 21, 71, 106
A sheep was fatally poisoned within three days of being dosed with c.19 g/kg of S. lichtensteinii fruit.141 Steyn154, 158 lists the main clinical signs of S. lichtensteinii poisoning in animals as salivation, diarrhoea, colic, bloat, stomatitis, tachycardia, polypnoea, cramps and paralysis. Occasionally, a vesicular exanthema is present.154, 158 The principal necropsy features are catarrhal enteritis,141, 154, 158 hyperaemia and oedema of the lungs, ascites and hydrothorax.141
Cultivated members of the Solanaceae, such as unripe tomatoes (Lycopersicon lycopersicum), which contain the glycoalkaloid tomatine,178 and potatoes (S. tuberosum), which contain solanine, are potentially poisonous. The highest concentration of glycoalkaloids in potatoes usually occurs in the peels, but considerable amounts can also be found in tubers, sprouts, foliage and berries106. Potatoes that are coloured green by exposure to light are particularly dangerous, and so too are tubers that have been bruised, cut or otherwise wounded. Boiling, baking or frying does not necessarily destroy the glycoalkaloids.20, 71, 106
The low prevalence of solanine poisoning is attributed by Nishie et al. (cited by Cheeke20, 21) to the poor absorption of solanine, hydrolysis of solanine to the less toxic aglycone solanidine in the gut, and rapid urinary and faecal excretion of the metabolites.
In South Africa, mechanical obstruction of the oesophagus by tubers is believed to be a greater threat to cattle than solanine (T.W. Naudé & T.S. Kellerman, VRI, personal observation, 1987).
Maldronksiekte, a nervous condition of cattle caused by S. tettense, is discussed in Central nervous system.
Cucumis spp. (Cucurbitaceae)
C. myriocarpus Naud. subsp. myriocarpus
C. myriocarpus Naud. subsp. leptodermis (Schweik.) C. Jeffrey & P. Halliday
C. africanus L.f.
Striped wild cucumber, wildekomkommer, streepwildekomkommer
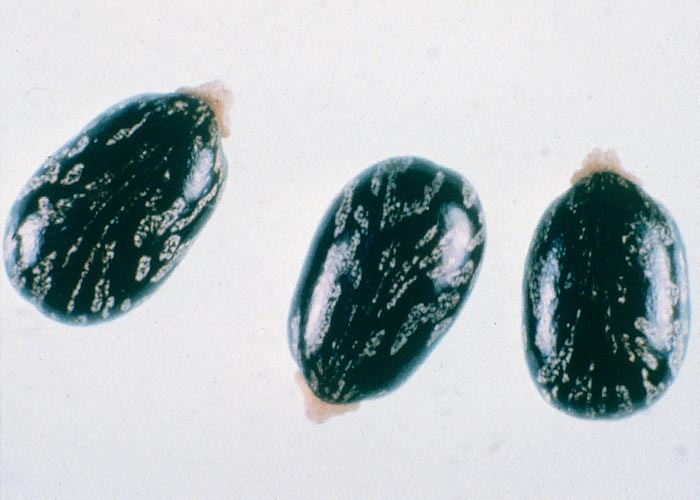
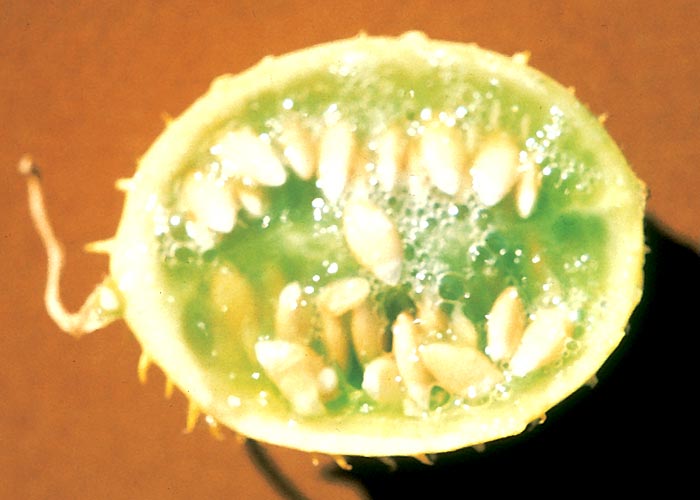
Cucumis myriocarpus is a sprawling or occasionally twining annual creeper with rough, hairy, grooved stems reaching a length of 1,25 m (Figure 40). The alternately arranged leaves up to 100 mm broad and 70 mm long, on long petioles, are ovate/oblong and deeply or shallowly three to seven lobed, with toothed margins (Figure 40). The upper surface of the leaf is darker and smoother than the under surface. Flowers are unisexual, and male and female flowers are borne on the same plant. The small 45 x 30 mm, oval to roundish, cucumber-like fruits are striped and covered with soft fleshy prickles. Young fruits are dark-green (Figure 40) to brownish, with conspicuous, pale-green, longitudinal stripes; older fruits are more or less orange and are less conspicuously striped. The seeds are white, smooth, compressed-oblong, 5 mm long and 2,5 mm broad58 and evenly boat-shaped with sharp tips (Figure 41).
Cucumis africanus L.f. closely resembles the above, but the spiny ovoid fruit, the size of a hen’s egg, is pale-lemon coloured.58
Both species are widely distributed throughout the region on open veld, but C. myriocarpus subsp. myriocarpus prefers disturbed areas, such as cultivated lands. Poisoning usually occurs in times of food scarcity as in winter, when sheep and cattle are turned onto harvested Cucumis-infested maize lands to graze.129, 158, 178Cucumis poisoning has been recorded also in Kenya109 and Australia.19, 163
Cucumis africanus is less toxic than C. myriocarpus subsp. myriocarpus.129 Quin,122, 123 in the first investigation of the chemistry of these plants, extracted an amorphous, bitter-tasting, crude toxin from the pulp of C. myriocarpus subsp. myriocarpus and C. africanus fruit. Later, Rimington127, 129 isolated a bitter substance, cucumin (C27H40O9), from ripe fruits of these two species and a closely related compound, leptodermin (C27H38O8), from ripe fruits of C. myriocarpus subsp. leptodermis (= C. leptodermis). Both were piscicidal and equally toxic to rabbits with an intravenous MLD of c.2 mg/kg.127, 129 Thin layer chromatography of cucumin and leptodermin by Enslin et al. in 1954 revealed them to consist mainly of cucurbitacin A together with some impurities. Cucumin may also have contained a small quantity of cucurbitacin D44. Several of these oxygenated, tetracyclic triterpenes and their glycosides, or cucurbitacins as they are commonly known, have been isolated from wild South African species,44–46, 125 for example, cucurbitacin A (C28H40O8) from C. myriocarpus subsp. myriocarpus and subsp. leptodermis, and cucurbitacin B (Figure 42) (C28H40O7) from C. africanus. Cucurbitacin B occurs most often of the bitter principles.125 The toxicity and cucurbitacin content of the fruits can increase as they ripen.125 The bitter principle content is usually highest in the fruit and roots, while the leaves and stems are not at all, or only slightly, bitter. Cucurbitacins are among the most bitter substances known to humans: a taste panel at the University of California could detect dilutions of cucurbitacin B in water as low as 1 μg/kg.102 One of the functions of toxic cucurbitacins in nature supposedly is to protect the plants against attack by herbivores. In contrast to this protective effect, cucurbitacins also act as kairomone feeding stimulants for certain beetles. Diabroticite beetles of the New World compulsively feed on plants that contain these substances, reportedly responding to as little as 1 ng cucurbitacin B on thin layer plates.102
African peoples often use the fruit as a purgative, sometimes with unfortunate results.154, 158 Cattle and sheep that take in large amounts of ripe fruit die suddenly without showing notable signs. Less acutely intoxicated stock develop severe diarrhoea, anorexia, weakness, tachycardia and polypnoea.123, 158 When large amounts have been eaten, lung oedema is the outstanding necropsy feature. The lesions in less acutely affected animals are located mainly in the gastrointestinal tract and these include severe hyperaemia of the mucosa which can give rise to the formation of ‘croupous’ pseudomembranes over large areas and haemorrhage into the lumen of the intestine. Varying amounts of what appears to be whole, coagulated blood plasma have been reported in the pyloric portion of the stomach and adjoining small intestine. The presence of large amounts of the characteristic, white, boat-shaped, undigested seed in the gut and rumen confirms the diagnosis.123, 158
Bitterness has been recorded in cultivated cucurbits in South Africa, such as Cucurbita pepo (squash and vegetable marrow), Citrullus lanatus (water-melon) and Cucumis metuliferus (jelly melon).44, 125 Steyn150 reported an instance of human poisoning with marancas (Lagenaria leucantha). The heat-stable cucurbitacins are not destroyed by cooking.123
Ornithoglossum vulgare B. Nord. Colchicaceae
(= O. viride)
Cape poison onion, Cape slangkop, Karoo-slangkop
The name slangkop is not really appropriate in this case, as the young inflorescence does not resemble a snake’s head and the flowers do not appear before the leaves are formed. Unlike Drimia sanguinea, the yellowish-white bulbs (20–50 mm in diameter) are buried deep in the soil, making them difficult to lift. Mature plants are about 300 mm tall, with about six bluish-green, boat-shaped leaves (c.100–150 mm long) arranged alternately and clasping the stem. The lower leaves are usually longer than the upper ones. The purplish-green, star-shaped, pendulous flowers, with upwardly reflexed petals, are attached by rather long stalks to the unbranched stem (Figure 43). The fruits are three-chambered and the seeds spherical and fleshy.59, 164
The plant is widely distributed in southern Africa, particularly in the former Cape Province, where it is common in the Karoo and the Kalahari (Figure 44). It prefers alkaline soils.59, 164
The taxon, O. viride, has now been split into a number of localized species, all of which should provisionally be regarded as toxic.
Despite being sporadically incriminated in localized, sometimes quite significant, losses of small stock in the Karoo, very little is known about poisoning with this plant. Anderson isolated a non-cardiac glycoside toxic principle from O. vulgare, which unfortunately was not pure enough for characterization by nuclear magnetic resonance spectroscopy (L.A.P. Anderson, OVI, unpublished data, 1988).
Steyn fatally poisoned a sheep with c.0,65 kg fresh leaves, flowers and seeds, while 30 g fresh leaves killed a rabbit.149 According to this author the clinical signs resemble those of cardiac glycoside poisoning. Peracutely or acutely affected sheep died before diarrhoea could develop, after showing signs such as excitement and imperfect muscular control. In subacute and chronic cases, diarrhoea was the most prominent feature.149
Schultz and Kellerman dosed dried O. vulgare material, consisting of a few bulbs, many leaves and almost no flowers, to a milk-tooth Merino of 42 kg. A dose of 2,5 g/kg was given without effect, followed a week later by 5 g/kg. Within 24 hours the animal developed apathy, diarrhoea and moderate bloating, which persisted for three days. No ECG changes were detected and the CPFI remained within normal limits (R.A. Schultz & T.S. Kellerman, OVI, unpublished data, 1988).
Since the uncharacterized toxic principle was not a cardiac glycoside and as there was no evidence of cardiac involvement, O. vulgare was adjudged to affect primarily the digestive tract.
Gnidia polycephala (C.A. Mey.) Gilg (Thymelaeaceae)
(= Arthrosolen polycephalus C.A. Mey.) Januariebos
Vahrmeijer164 describes Januariebos as a densely tufted bush, about 500 mm tall, much branched from the base, with few side branches (Figure 45). The narrow elliptical leaves, pointed at the tips, are borne only in the young stages, then fall off revealing dark-green stems. The broom-like appearance of the leafless shrub accounts for the alternative colloquial name, besembos (= broom bush). Flowerheads, subtended by papery bracts, arise at the ends of the branches (Figure 46). The flowers are tubular, hairy and yellow; the fruit is plumed, single chambered and single seeded (Figure 46).164 The bush is common on sandy-lime soils and sand dunes of the arid Karoo and northern Kalahari. It often grows on run-down veld158, 164 and has invaded trampled areas in Griqualand West (Figure 47) and southern Botswana.
The earliest reference to the toxicity of Gnidia was by Robertson in 1905, who induced a ‘fatal gastritis’ with it in an ox.135 After initial negative dosing trials, the toxicity of the plant was verified by Steyn.147, 154, 158 Cattle and sheep eat it in times of adversity, for instance, in winter, when the flowering G. polycephala bushes are highly visible on the brown veld. The toxicity of the plant varies with locality and time of the year, and is reputed to be highest in the flowering stage.158
Steyn lists the clinical signs of G. polycephala poisoning as severe diarrhoea, apathy, weakness, tachycardia, polypnoea and fever. The gross lesions mirror the clinical signs, and include severe enteritis and congestion of the lungs and liver.158 More recently, lesions very similar to those of harpuisbos poisoning (vide infra) have been found in sheep poisoned by G. polycephala. These included focal myocardial necrosis, pulmonary oedema and emphysema and lymphoid atrophy (P.A. Basson, State Veterinarian, Grootfontein, Namibia, personal communication, 1986).
A dose of 1,36 kg fresh flowering G. polycephala was fatal for a sheep.158 Rindl (1917) obtained a non-toxic glycoside from Januariebos that was isomeric, but not identical, with daphnin.132–134 Later, Vermeulen and Pieterse isolated umbelliferone and an unidentified active principle from the plant.172
Powdered G. polycephala is extremely irritating to the eyes, nose and skin (ref. Gnidia burchellii).
Gnidia burchellii (Meisn.) Gilg (Thymelaeaceae)
(= Lasiosiphon burchellii Meisn. Harpuisbos)
The harpuisbos is a much-branched, woody shrublet growing c.1 m high (Figure 48), with narrow elliptic leaves which are carried mainly at the upper ends of the branches. Distinct scars are visible on the stems where the leaves have dropped off. Yellow, tubular flowers are borne in dense clusters at the ends of branches (Figure 49), chiefly during summer. Leaf-like bracts, which are green and hairy, surround the flower-clusters. Gnidia burchellii can easily be confused with other Gnidia spp. This plant family, however, can be distinguished from other families by the fine silvery, silky threads which are exposed when a twig is snapped and the tough bark is pulled away from the wood.164
Vahrmeijer164 reports that G. burchellii favours the slopes of hills and grows well on sandy or stony sandstone soils. It is frequently associated with trampled veld in southern Mpumalanga, the north-eastern Free State, and north-western KwaZulu-Natal (Figure 50). Harpuisbos poisoning can be of considerable local importance in this area.76
Figure 51 12-Hydroxydaphnetoxin: the tricyclic phorbol nucleus is responsible for its irritant effect (see The skin and adnexa)
Harpuisbos was first demonstrated to be toxic in South Africa by Steyn (1932).151, 152 Adelaar in 1961 described an outbreak in sheep being trekked from the vicinity of Babanango towards Dundee during February, in which 68 out of 540 died after developing typical signs of harpuisbos poisoning. The diagnosis was confirmed by dosing plants collected along their route to sheep at the Veterinary Research Institute, Onderstepoort. Fresh G. burchellii was fatal at a dose of 3,4 g/kg and dried material at 0,7–1,4 g/kg. Clinical signs were noticed within hours and death occurred within one to eight days (T.F. Adelaar, T.W. Naudé and R.C. Tustin, VRI, unpublished data, 1961). A 12-hydroxy analogue (C27H30O9) of daphnetoxin (Figure 51), a poisonous principle from the bark of Daphne mezereum (Thymelaeaceae), has been isolated from G. burchellii.26, 116 The basic skeleton of this compound is a phorbol diterpene, joined across the cyclohexane ring by the ortho ester of benzoic acid27, 116 (see The skin and adnexa). Potent antileukaemic diterpenoid esters and a furanosesquiterpene have also been isolated from G. latifolia, a species found in Kenya.84, 86
The naturally intoxicated sheep lagged behind the flock and suffered from severe dyspnoea; breathing was laboured, abdominal and noisy. Other signs included a mild febrile reaction, a bloody nasal discharge, walking with the neck extended, trembling, and subcutaneous emphysema over the neck and head. Even the slightest exertion sometimes induced death (T.F. Adelaar et al., OVI, unpublished data, 1961). Acute intoxication of sheep with ethanolic extracts of G. burchellii resulted in accelerated respiration characterized by short, sharp inspiration and prolonged forced expiration, weakness and occasional pushing against objects. Subacutely affected cases manifested dyspnoea, nasal discharge, ruminal atony, anorexia and foetid diarrhoea.162 These experiments are more completely discussed in Respiratory system, which deals with the respiratory system.
The significant lesions of acute experimental poisoning were emphysema and moderate congestion and oedema of the lungs, karyorrhexis of the lymphocytes in lymphoid follicles and focal hyperaemia of the gastrointestinal tract. Subacute intoxication was marked by similar changes in the lungs (but the emphysema was less prominent than the oedema), atrophy of lymphoid tissue in the spleen, nephrosis, myocarditis, hyperaemia of the gastrointestinal tract, and diarrhoea. The myocardial lesions consisted variously of oedema, focal necrosis, and fibrosis, with round-cell infiltration.162 In naturally poisoned animals, emphysema of the lungs was more prominent than in the experimental cases, and emphysema was sometimes present in the mediastinum and subcutaneous tissues162 (T.F. Adelaar et al., OVI, unpublished data, 1961). Lesions similar to those of harpuisbos were described in bovine and ovine G. anthylloides poisoning by Alexander4 (see Respiratory system).
Terblanche et al.162 found the lesions of G. burchellii poisoning to be surprisingly mild in view of the violent symptoms of dyspnoea, ruminal stasis, and diarrhoea observed in most cases. They suggested that the toxic principle primarily affected the heart and lungs and caused irritation of the gastrointestinal tract. Steyn152 and Alexander,4 on the other hand, were of the opinion that the plant contained mainly a gastrointestinal irritant.
Kiptoon et al.80 reported elevations in blood urea nitrogen and degeneration of lymphocytes in bovine G. latifolia poisoning which were similar in many respects to those described by Terblanche et al.162
Workers exposed to the dust of harpuisbos have complained of a burning sensation in the nose, throat and chest, dyspnoea,152 coughing, headache and nausea. This irritant effect is attributed to the phorbol ester derivatives (see Euphorbia ingens, The skin and adnexa). When milling the plant, contact with the dust must be avoided and respirators should be worn.
Sesbania punicea Cav. (Benth.) (Fabaceae)
Red sesbania, Brazilian glory pea, coffee bean, rattle box, rooi-sesbania
Sesbania punicea is a laxly branched bush or small tree with slender branches, drooping leaves and dense sprays of highly ornamental orange-red flowers (Figure 52). The compound leaves of about 150–200 mm in length have 8–15 pairs of oppositely arranged oblong or inversely oval leaflets (Figure 52). The striking pea-like flowers are borne on racemes, and the four-angled pods, c.100 mm long (Figure 52), contain glossy, rounded, kidney-shaped greyish seeds.58, 161
The rattle box is native to southern Brazil, Argentina and Mexico. A prolific seeder, it has escaped from gardens to become a noxious weed on the open veld161 and along waterways, especially in the high-rainfall eastern and south-western parts of the country. Outbreaks of poisoning are occasionally reported in fowls and pigeons.161
Sesbania poisoning has been experimentally induced in domestic animals and poultry. The whole plant is poisonous, the seeds being most toxic, followed by the flowers and then the leaves. Repeated small doses have a cumulative effect. Fowls and turkeys could be fatally poisoned by a single dose of 2,5 g/kg of seeds (20–70 seeds), with the clinical signs appearing within 24–48 hours, succeeded by death after two to eight days. Pigeons were killed in 10–40 hours by doses of 1 g/kg (six seeds) or more. Ducks were very refractory and as much as 6,5 g/kg of seeds produced no ill effect. The resistance of ducks was possibly related to the speed at which the seeds passed through the digestive tract. In fowls, an average of seven out of 40 seeds was recovered in their droppings within 48 hours of ingestion, while in ducks the figure was 19 seeds in 30 hours.
The clinical signs of Sesbania poisoning in fowls were depression, ruffled feathers, drooping of the wings, ataxia, progressive debility and diarrhoea. Pigeons vomited and the greater individual variation in susceptibility to intoxication of this species can probably be attributed to vomition. Hyperaemia and haemorrhage in the proventriculus were the most constant and characteristic lesion of Sesbania poisoning in birds. These haemorrhages occurred on the crest of the proventriculus in the lamina propria: cross-section of the papillae showed haemorrhages in the lamina propria of the plicae, especially in those nearest the gland opening. In a few cases, round focal necrotic lesions were evident in the mucous membrane of the crop. The kidneys were pale and the lungs congested and oedematous.161
A guinea-pig and a rat died 48 hours after being dosed with 2 g/kg of ground seeds. Rabbits were equally, if not more, susceptible.161 The acute, MLD of the ground seed for sheep was between 0,5 and 1 g/kg, but as little as 0,1 g/kg/day caused death after six doses. The clinical signs included depression, listlessness, weakness, anorexia, ruminal stasis and diarrhoea, hyperaemia of the conjunctiva and occasional dyspnoea. At necropsy the most notable findings were congestion of the lungs, with varying degrees of hyperaemia and haemorrhage in the gastrointestinal tract.161 To the best of our knowledge, natural outbreaks of Sesbania poisoning in mammals have not been recorded in southern Africa.
Two cytotoxic metabolites, sesbanine and drummondol, and a potent anti-tumour agent, sesbanimide (Figure 53), have been isolated from S. drummondii by Powell et al. (cited by Gorst-Allman et al.53). Sesbanimide has now been recovered from S. punicea seed and its structure elucidated.53 A tandem mass spectrometric procedure is available for the detection of sesbanimide A and its isomers in plant material.120
Other conditions of the gastrointestinal tract
Bloat
Bloat induced by plants may be of either the gassy or frothy type; both equally deadly. In southern Africa, gassy bloat is associated most often with paralysis of the rumen caused by phytogenous cardiac glycosides (see Cardiovascular system). Frothy bloat, the subject of this discussion, on the other hand, results from the inability of ruminants to eructate gas trapped in a stable foam.20, 158 Leguminous plants, generally speaking, are particularly bloat-provoking, especially when immature and wilted or frosted.158
Cheeke20, 21 in a succinct review of bloat-producing proteins list lucerne (Medicago sativa), red clover (Trifolium pratense) and white clover (T. repens) as among the most important bloating species in temperate regions. Subterranean clover (T. subterraneaum) is usually less often incriminated in bloat than other clovers or lucerne. The position with regard to the bloating properties of cultivated legumes in South Africa is much the same.
Cattle are as a rule more prone to frothy bloat than other domestic ruminants.
That interference with eructation is hazardous, is evident from the large volumes of gas produced in the rumen. The daily production of rumen gas, principally carbon dioxide and methane, is about 400 litre in cattle and 50 litre in sheep. During moderate/severe bloat, the total volume of the rumen could be 115 litre.177
The bloat-producing principles in plants have been identified as soluble proteins20, 62 and possibly saponins.25 However, the involvement of saponins is in doubt94 as Majak and his co-workers,96 after prolonged research, could not find an association between lucerne saponins and bloat. Soluble protein in the membranes of the gas bubbles stabilizes the foam in the rumen.20, 62
Sant and Wilson137 reported that the cell walls of bloat-causing legumes (lucerne, red clover and white clover) were thinner and disintegrated faster during cellulose digestion than those of non-bloat-causing legumes (sainfoin with tannin and cicer milkvetch without tannin). The reason for the higher bloat potential of legumes with fragile cell walls is that the rapid discharge of their cell contents into the rumen apparently creates favourable conditions for the sudden development of stable foam.61, 137 Immature legumes, similarly, are highly bloat-inducing because their lush foliage is rich in soluble proteins and carbohydrates, and their cells rupture quickly in the rumen.
Howarth et al.62 reported that the total soluble N and total soluble protein were the most practical chemical parameters for predicting the bloat potential of lucerne. Owing to their low coefficients of determination, however, these parameters have poor predictive value on a single analysis basis.
Other surface-active plant constituents, such as polar lipids, possibly play a part in the frothy complex, either as foaming agents or antifoaming agents. Persistent rigid foams are believed to be formed when critical concentrations of several surface-active agents are reached in the rumen.62
The probability of bloating is increased by conditions in the rumen that favour a high fermentation rate (i.e. gas production) such as a vigorous microbial population and the consumption of forage rich in fermentable soluble carbohydrates. Soluble carbohydrates exert an additional indirect effect on bloating by favouring the protozoal component of the ruminal flora. When the protozoa die off in large numbers, their cellular contents may contribute to the production of stable foams.20, 93
Frothiness has also been associated with high chlorophyll levels in the ruminal fluid of the dorsal sac, and these levels, in their turn, reflect the amount of suspended chloroplast material present.93, 96 Chloroplast fragments, among others, act as a substrate for mesophyll cell-digesting bacteria to grow on (thus providing inocula for the rapid digestion of incoming feeds) and can be involved in the entrapment of gases.93, 96
Non-surface active agents too may play a part in the occurrence of bloat, e.g. condensed tannins in plants are believed to prevent foam formation by precipitating soluble proteins.20, 62, 137, 160
Some cattle are more prone to bloat than others and individual animals may vary in their propensity for bloating from one day to another.158 Evidence has been submitted that differences in the saliva constituents might be associated with high or low susceptibility to bloat.92, 95
Since the insertion of a trochar is of no avail in this type of bloat, an antifoaming agent has to be introduced into the rumen to break the froth.158 Any suitable vegetable oil can be used for this purpose in an emergency, if commercial pharmaceutical preparations containing recognized antifoaming agents, such as polymerized methyl silicone or poloxalene, are not available.
Antifoaming agents, especially poloxalene, can be used prophylactically, e.g. by dosing them56 or by incorporating them in intraruminal capsules48 and controlled-intake-blocks. Monensin, an ionophore antibiotic, contained in controlled release capsules, has also been successfully applied in the prevention of bloat in Australia.18, 90
Another precautionary measure is to spray concentrate rations or herbage with vegetable oils or fats before high-risk pastures are grazed. High-energy, high-protein supplement increases the incidence of bloat while low-energy, high-protein reduces it.119 Steyn158 reported that bloat can sometimes be avoided by not feeding lucerne and high-energy concentrate rations together but a few hours apart. Lucerne, which is easily fermented, releases a great deal of gas in the rumen if it is fed concurrently with cereals, sugar, or molasses.158 Slowly fermented forages reduce the risk of bloat.119 Any measure that will reduce the speed of intake of bloat-inducing forage is beneficial. The feeding of roughage in the form of grass hay just before, or during the grazing of lush immature legume pastures will reduce the risk of the occurrence of bloat.158 High-pressure grazing systems have been employed to avoid bloat by forcing stock to consume the entire plant and not just the succulent tops.20
Besides legumes, the following plants have been named by Steyn158 as causing bloat in South Africa: Phytolacca dioica (belhambra tree), Noltea africana, Senna italica subsp. arachoides (= C. arachoides), Indigofera sessilifolia (= I. patens) and Zygophyllum foetidum.
Finally, it should be borne in mind that cardiac glycoside-containing plants can cause bloat in addition to their cardiac, nervous and other effects. The bloat induced by cardiac glycoside-containing plants, like the bloat of animals paralysed by diplodiosis or poisoned by prussic acid, is of the gassy type and hence can be relieved by trochar.
Mechanical obstruction and irritation
Phytobezoariasis
The formation of plant-hair balls or phytobezoars (also known as pappus hair bezoars) in the abomasum of sheep and goats is a sporadic problem in many parts of the Karoo in South Africa.8, 9 Phytobezoars similar to natural cases have been experimentally induced in lambs and kids by feeding with mature flowers of Gnidia polycephala and Chrysocoma ciliata (= C. tenuifolia).8, 10 Balls resembling natural bezoars have also been manufactured in vitro by separately felting the seeds of Gnidia polycephala, Chrysocoma ciliata and Eriocephalus ericoides (= E. glaber).11 Bath8 concluded from these experiments that several Karoo bushes may be involved in the formation of bezoars and that the dominant species responsible for their formation possibly varied from area to area. Besides these three plants, ovine phytobezoars have been reported to result from the ingestion of Stipagrostis obtusa (= Aristida obtusa) and S. ciliata var. capensis (= Aristida ciliata).6, 89, 158 Bath and Bergh9 cite Albl as having seen phytobezoars formed by A. ciliata in the Namib and Lotononis platycarpa in the Kalahari.
Bath and Bergh9 reported that bezoariasis is not limited to specific veld types, though the incidence appears to be highest on the mountains of the southern Karoo. In the north, the condition is associated with the plant communities of the dune ‘streets’ between sand dunes. Poor veld management leading to diminution in the variety of the vegetation and an increase in undesirable plants is believed to promote the condition. Goats are relatively more susceptible than sheep, and Boer goats more than Angoras. Fatalities are highest in the age group 3 to 15 months, with a peak in deaths occurring between the ages of 6 and 12 months. The morbidity varies between one and 10%, rarely rising to 20% or higher. The overall occurrence of phytobezoars is difficult to assess as many animals are not clinically affected.8, 9
The condition is characterized by loss of mass and poor appetite. The abdomen may become so enlarged that it swings from side to side as the animal runs. Bath and Bergh9 reported further that the rumen contents are watery, intestinal movement is sluggish or absent and the faeces are often scanty and foul-smelling. Phytobezoars can be diagnosed by deep palpation of the abdomen caudal to the xiphoid cartilage. This is best done by straddling the animal just ahead of the hind legs, clasping the hands under the belly and then lifting the hands and pressing them together.9
Death results from gastrointestinal complications associated with impediment of the flow of ingesta or from complete blockage of the digestive tract (Figure 55). In the latter instance, death is sudden and can be accompanied by rupture of the intestinal or gastric wall.9
Carcases are usually emaciated; the forestomachs are thin-walled; the papillae of the rumen atrophied; the ruminal contents watery, finely divided, dark-coloured and foul-smelling. No phytobezoars are encountered in the rumen, reticulum or omasum.9
The abomasum is often enlarged and the walls thin, ulcerated or ruptured where bezoars touch the gastric wall. One or several bezoars can be present at one time. Smaller bezoars sometimes escape through the pyloric sphincter into the small intestine to cause obstruction or even rupture lower down. Here again the content is dark and foul-smelling. Below the obstruction the intestine is empty, thin-walled and slightly inflamed.
Bezoars vary between 10 mm and 150 mm in diameter, with a dry mass of 0,5–270 g. They are compact and dense, with a fine velvety texture and vary in shape from spherical to oval, flattened, or bizarre (Figures 56 and 57). Occasionally several bezoars merge to form a large lobulated structure. The bezoars are composed of innumerable fine plant fibres, tightly packed (felted) into layers. The plant fibres can be distinguished from wool, among others, by the presence of annular thickenings and terminal hooks.11 There is no sign in the central core of any structure which could serve as a nucleus for growth. The colour of a typical bezoar is khaki-dun.8, 9
Treatment with purgatives is ineffective or dangerous, and bezoars cannot be broken manually through the stomach wall. Apart from surgery, losses can be reduced by avoiding camps with bezoar-forming bushes (especially if they are in flower), applying good pasture management, identifying affected animals through palpation and marketing or slaughtering them before they lose condition.8, 9
Phytobezoars have been reported at least once in sheep grazing pastures other than natural Karoo veld. Schneider and Hugo139 established that the phytobezoars of suckling lambs on a farm in the Ceres Karoo were composed almost entirely of the hairs covering the stems and leaves of Ornithopus sativus Brot. (serradella) (Figure 54). The lambs, of about four months of age, had grazed for about two and a half months with their dams on a good artificial serradella pasture. A total of 62 out of the 300 lambs died after showing typical signs and lesions of phytobezoariasis.
Phytobezoars must be distinguished from the abomasal hairballs (trichobezoars) in kaalsiekte, (see The skin and adnexa) an alopecic condition induced in lambs by a toxin in the milk of ewes grazing on bitterbos (Chrysocoma ciliata).59, 148, 153, 154
Obstruction by cocoons of moths
Reports have been received of the mechanical impaction of the rumens of cattle by cocoons of the molopo moth (Gonometa postica) (Figure 58) and a related species (Gonometa rufobrunnea)(Figure 59). After their ingestion, the persistent silk threads of the cocoons bind the contents of the rumen into a fibrous mass of up to 37 kg (wet).41, 181 The protein fibres of silk have a very stable three-dimensional structure174 into which digestive enzymes cannot easily penetrate. Doubts have also been expressed whether ruminal flora produce enzymes capable of degrading silk (J. Van der Walt, Faculty of Veterinary Science, University of Pretoria, personal observation, 1995). Cattle are believed to take in the cocoons of G. postica principally in the weaning period when, out of inquisitiveness, they may display aberrant appetites. Parasitism of the pupae, causing the cocoons to dry out and be blown from the trees, might contribute to the occurrence of the condition. The only successful treatment is surgical removal of the ingesta-mass (Figure 60).41, 181 Gonometa rufobrunnea is found in the mopani belt of the Zoutpansberg bushveld41 and G. postica on black thorn (Acacia mellifera) and camel thorn (A. erioloba) in the Northern Cape Province (Figure 61) and the southern parts of Namibia and Botswana.181
Mechanical irritation
An outbreak of diarrhoea has been reported in sheep grazing on a wheat-stubble land infested by a number of weeds including Dittrichia graveolens (= Inula graveolens) (Cape khakiweed). This plant was introduced from the Mediterranean area to the Cape, where it is confined to the south western parts. The Cape khakiweed on this particular stubble-land had reached the end of its growth and was covered by dry flowers bearing ripe seeds.138 Schneider and Du Plessis138 reported that, although they are fairly unpalatable, the tender young plants of D. graveolens are often grazed during the dry summers when green forage is scarce. Seeding Cape khakiweed, on the other hand, is seldom eaten, because it is dry and unattractive in early winter after the first rains have turned the pastures green. On this occasion, the rains had been late and no other food being available, the sheep were forced to eat D. graveolens in the seed-bearing stage. Affected sheep were anorexic and listless, their stomachs were tucked in, and they suffered from a dark, watery diarrhoea. Post-mortem examination revealed that the wall of the entire small intestine was thickened and reddish in colour. Numerous hair-like projections protruded from the mucosal surface and these projections were microscopically identified as the pappus bristles of D. graveolens. Barbs on the bristles prevented their retraction. A total of 10 out of 190 sheep died.138
Discussion
Some plants that primarily affect other organs or systems can exert a considerable secondary effect on the digestive tract. The most notable of these are the cardiac glycoside-containing plants (see Cardiovascular system) which variously cause constipation, gassy bloat and diarrhoea. The purgation can be so persistent in cardiac glycoside poisoning that stock, surviving the cardiac effects, can succumb from the diarrhoea. Significant gastrointestinal lesions, including erosions and ulceration, have also been observed in trichothecene-induced radiomimetic intoxications, such as stachybotryotoxicosis. Likewise, in Pteridium aquilinum poisoning ulcerations have been reported in the digestive tracts of cattle and adenocarcinomas in the small intestines of sheep (see Haemopoietic system).
Less important plant poisonings that primarily or secondarily affect the gastrointestinal tract are Xanthium spp., Ranunculus multifidus, Raphanus raphanistrum, Chrysocoma ciliata and Adenia digitata. Xanthium poisoning has been discussed in some detail in Liver. Ranunculus multifidus (buttercup, botterblom) is a common weed in damp places throughout the region.158 The plant contains a glycoside, ranunculin, that is converted enzymatically into an irritant volatile oil, protoanemonin. Protoanemonin either volatilizes or, being unstable, polymerizes to non-toxic anemonin in the dry plant.20 Raphanus raphanistrum (wild radish, ramenas) seeds, in their turn, contain a glycoside that is apparently broken down by enzymes in the plant to a severely irritant substance.158 A complex of clinical signs is produced, the most important of which is diarrhoea. Wild radish seed is common in badly winnowed grain.158 Chrysocoma ciliata is noted for causing alopecia in suckling lambs (see The skin and adnexa), but ingestion of large amounts can also lead to diarrhoea (lakseersiekte) in adult sheep and cattle.154 This type of diarrhoea is relatively rare, however, since it usually appears only in times of severe drought. Adenia digitata (Figure 62) has the most striking appearance of the plants in this group: twining branches with digitately three- to five-lobed leaves, tendrils and bright-orange flowers grow from a large tuberous underground rootstock.88, 118 The plant contains prussic acid and a toxalbumin, modeccin, that is a gastrointestinal irritant.54, 158 Humans who survive the cyanide in the tubers may develop a severe diarrhoea from the modeccin two or three days later.158 A closely related species, A. glauca, has not been incriminated in poisoning,158 although it contains the cyclopentene cyanogens tetraphyllin B and epitetraphyllin B.145 The first-mentioned cyanogen, tetraphyllin B, has also been isolated from A. digitata.145 Adenia digitata is found in KwaZulu-Natal, the provinces constituting the former Transvaal and in Zimbabwe.88 Tannin-containing plants, such as Elephantorrhiza elephantina (elandsboontjie), have been incriminated in diarrhoea,154 and oaks (Quercus spp.), though primarily nephrotoxic, can cause gastrointestinal signs and lesions (see Urogenital system).
Animals are sometimes poisoned by highly toxic garden plants, such as chinkerinchees or plakkies, and a rare case has been described of the poisoning of stock with Colocasia antiquorum (elephant ear) (Figure 63). Hungry sheep, off-loaded at a station, were accidentally poisoned by potted Colocasia plants to which they had access (H.E. van de Pypekamp, State Veterinarian, Rustenburg, 1982). The active principle in this case is the crystal raphides of calcium oxalate monohydrate. When the leaves are chewed, the highly irritant crystals, tightly packed in special ejector cells, are shot out piercing the mucosa of the mouth and pharynx. Transient buccal irritation has also been described in a black rhinoceros browsing on Xanthosoma mafaffa, of the same family as Colocasia.179 Soluble oxalates from plants can also crystalize as calcium oxalate in various tissues, especially in the kidneys and the rumen wall, causing rumenitis and shock (see Urogenital system). Other plants that contain insoluble calcium oxalates all belong to the family Araceae and include the indigenous Zanthedeschia spp. (arum lilies, varkoor) and ornamentals such as Philodendron, Dieffenbachia and Caladium spp. grown for their striking foliage. Mechanical irritation of the buccal cavity by other plant agencies is discussed in The skin and adnexa.
Insects can sometimes be indirectly involved in plant poisonings that affect the gastrointestinal tract, e.g. monarch butterflies or grasshoppers that accumulate cardiac glycosides in their bodies from the plants on which they feed (see to Cardiovascular system) or army worms that play an unexplained role in the aetiology of kikuyu poisoning. Insects may also directly affect the gastrointestinal tract, as in the case of pollen beetles (Astylus atromaculatus) (Figure 64) that now and then cause diarrhoea and a necrotizing enteritis in ruminants during late summer.75 Since the beetles are attracted to pollen, special care should be taken when stock graze on harvested lands on which volunteer crops in the flowering stage are growing. The most dangerous time for grazing plants infested with A. atromaculatus is in the cooler parts of the day when the beetles are less active and thus most easily ingested. A diagnosis of Astylus poisoning is confirmed by the presence of large numbers of the yellow and black beetles in the rumen (Figure 65). Unlike the CMR or blister beetle, Mylabris oculata (Meloidae), the pollen beetle contains no cantharidin (Figure 66).75 Ingestion of smaller catharidin-containing beetles of this family, Cyanolytta and Cylindrothorax spp., have resulted in fatal gastrointestinal irritation of chickens.112
Circumstantial evidence has been submitted that a toxic fungus might be involved in outbreaks of diarrhoea in stock grazing on green oats pastures in the Western Cape Province. In goats the diarrhoea was occasionally associated with photosensitivity and in cattle by a drop in milk yield. Pure cultures of a phytopathogenic fungus, Drechslera campanulata (teleomorph Pyrenophora semeniperda), isolated from leafspots on the suspectedly poisonous oats, elicited only diarrhoea and necrosis of the forestomachs of goats and sheep to which they were dosed. The necrotic lesions in the forestomachs resembled those of kikuyu poisoning.140
A chapter on gastrointestinal conditions would not be complete without brief mention of trypsin inhibitors. Many plants, notably raw soya beans, contain these heat labile protein substances; heating at 100 °C for 15 minutes destroys 95% of their activity. According to Cheeke and Shull,20 trypsin apparently reacts with the inhibitor protein by tight noncovalent bonds at the active site to form irreversible complexes. Severe digestive disturbance results.
References
- ANDERSON, T.S. 1948. Castor poisoning in Ayrshire cattle. Veterinary Record, 60, 28.
- ANDERSON, L.A.P., DE KOCK, W.T. & PACHLER, K.G.R. 1967. The structure of vermeerin, a sesquiterpenoid dilactone from Geigeria africana Gries. Tetrahedron, 23, 4153–4160.
- AHMED, O.M.M. & ADAM, S.E.I. 1979. Effects of Jatropha curcas on calves. Veterinary Pathology, 16, 476–482.
- ALEXANDER, R. 1928. Lasiosiphon anthylloides as a poisonous plant. Report on Veterinary Research, Union of South Africa, 13 & 14, 233–240.
- ANNECKE, D.P. & MORAN, V.C. 1982. Insects and mites of cultivated plants in South Africa. Pretoria: Butterworths.
- BANTING, L.F. 1972. Abomasal obstruction in Karakul sheep. Veterinary Clinician, 8, 25.
- BARTON, D.H.R. & LEVISALLES, J.E.D. 1958. Sesquiterpenoids. Part XI. The constitution of geigerin. Journal of the Chemical Society, 4518–4523.
- BATH, G.F. 1978. Abomasal phytobezoariasis of goats and sheep. Journal of the South African Veterinary Association, 49, 133.
- BATH, G.F. & BERGH, T. 1979. A specific form of abomasal phytobezoar in goats and sheep. Journal of the South African Veterinary Association, 50, 69–72.
- BATH, G.F., SMITH, F.G., VORSTER, H.J. & CROSS, R.H.M. 1992. Experimental reproduction of phytobezoars. Journal of the South African Veterinary Association, 63, 108–112.
- BATH, G.F., BOTHA, P., VORSTER, H.J. & CROSS, R.H.M. 1992. Physical structure and chemical composition of abomasal phytobezoars of sheep and goats. Journal of the South African Veterinary Association, 63, 103–107.
- BOT, J., SWEET, SEGNET & HOLLINGS, NORA. 1985. A guide to the use of pesticides and fungicides in the Republic of South Africa. Directorate of Agricultural Information, Pretoria, ISBN 0 621 09678 4, Republic of South Africa.
- BOTHA, C.J., GOUS, T.A., PENRITH, MARY-LOUISE, NAUDÉ, T.W., LABUSCHAGNE, LEONIE & RETIEF, ELIZABETH. 1997. Vermeersiekte caused by Geigeria burkei Harv. subsp. burkei var. hirtella Merxm. in the Northern Province of South Africa. Journal of the South African Veterinary Association, 68, 97–101.
- BRANDFORD, W.C. 1878. In reference to vomit sickness in sheep in Victoria West. Report of the Veterinary Surgeon, Cape of Good Hope, (G. 13–78).
- BRYSON, R.W. 1982. Kikuyu poisoning and army worm. Journal of the South African Veterinary Association, 53, 161–165.
- BRYSON, R.W. & NEWSHOLME, S.J. 1978. Kikuyu grass poisoning of cattle in Natal. Journal of the South African Veterinary Association, 49, 19–21.
- BURTT-DAVY, J. 1912. Botanical Investigation into Gal-Lamziekte. Report on Veterinary Research, Union of South Africa, 2, 181–221.
- CAMERON, A.R. & MALMO, J. 1993. A survey of the efficacy of sustained-release monensin capsules in the control of bloat in cattle. Australian Veterinary Journal, 70, 1–4.
- CARTER, G.I. 1990. Prickly paddy melon (Cucumis myriocarpus) poisoning of cattle. Australian Veterinary Journal, 67, 276.
- CHEEKE, P.R. & SHULL, L.R. 1985. Natural toxicants in feeds and poisonous plants. Westport, Connecticut: AVI Publishing Company, Inc.
- CHEEKE, P.R. 1998. Natural toxicants and feeds, forages and poisonous plants. 2nd edn. Danville, Illinois: Interstate Publishers, Inc.
- CLARKE, E.G.C. 1947. Poisoning by castor seed. The Veterinary Journal, 103, 273–278.
- CLARKE, E.G.C. & CLARKE, M.L. 1975. Veterinary toxicology. London: Bailliére Tindall.
- CLARKE, E.G.C. & JACKSON, J.H. 1956. The use of immune serum in the treatment of ricin poisoning. The British Veterinary Journal, 112, 57–62.
- CLARKE, MYRA L., HARVEY, D.G. & HUMPHREYS, D.J. 1981. Veterinary toxicology. London: Bailliére Tindall.
- COETZER, J. & PIETERSE, M.J. 1971. Note. The isolation of 12-hydroxy-daphnetoxin, a degradation product of a constituent of Lasiosiphon burchellii. Journal of the South African Chemical Institute, 24, 241–243.
- COETZER, J. & PIETERSE, M.J. 1972. The structure of 12-hydroxy-daphnetoxin, a poisonous constituent of Lasiosiphon burchellii: An X-ray analysis of 12-hydroxydaphnetoxin tribromoacetate. Acta Crystallographica, B.28, 620–624.
- COLEMAN, P.C., POTGIETER, D.J.J., VAN ASWEGEN, C.H. & VERMEULEN, N.M.J. 1984. Flavanoids of Geigeria. Phytochemistry, 23, 1202–1203.
- CORDES, D.O., COUP, M.R., HARRIS. G.H., DAVENPORT, P.G. & BUSCH, J. 1969. Acute ruminal indigestion, alkalosis and death of cattle grazing kikuyu grass. New Zealand Veterinary Journal, 17, 77–81.
- CURSON, H.H. 1926. Some little known South African poisonous plants and their effects upon stock. Tropical Veterinary Bulletin, 14, 79–80.
- CURSON, H.H. 1928. Some little known South African poisonous plants and their effects upon stock. Report on Veterinary Research, Union of South Africa, 13 & 14, 205–229.
- DE KOCK, W.T., PACHLER, K.G.R., ROSS, W.F. & WESSELS, P.L. 1968. Griesenin and dihydrogriesenin, two new sesquiterpenoid lactones from Geigeria africana Gries-1 Structures. Tetrahedron, 24, 6037–6043.
- DE VILLIERS, J.P. 1959. Isolation and structure of geigerinin, a guaianolide. Journal of the Chemical Society, 2412–2414.
- DE VILLIERS, J.P. 1961. Isolation and structure of gafrinin, a sesquiterpenoid lactone from Geigeria africana. Journal of the Chemical Society, 2049–2052.
- DE WAAL, H.L. 1938. On the constitution of the bitter principle ‘Geigerin’ I. The isolation of various degradation acids. Onderstepoort Journal of Veterinary Science, 10, 395–410.
- DI MENNA, MARGARET E. & MORTIMER, P.H. 1971. Experimental myrotheciotoxicosis in sheep and calves. New Zealand Veterinary Journal, 19, 246–248.
- DIXON, R.W. 1895. Report of the Veterinary Surgeon, Cape of Good Hope. (G.41–96).
- DIXON, R.W. 1899. Report of the Veterinary Surgeon, Cape of Good Hope, (G.35–1900).
- DODD, F.R. 1932. Castor seed in feeding stuffs. Analyst, 57, 488–490.
- DU TOIT, P.J. 1928. Investigations into the cause of vomeersiekte in sheep. Report on Veterinary Research, Union of South Africa, 13 & 14, 109–153.
- EDWARDS, L.T. 1935. Impaction of the rumen of cattle due to the ingestion of the cocoons of the caterpillar Gonometa rufobrunnea Auriv. Journal of the South African Veterinary Medical Association, 6, 188–191.
- ELLENHORN, M.J. & BARCELOUX, D.G. 1988. Medical toxicology. Diagnosis and treatment of human poisoning. New York, Amsterdam, London: Elsevier.
- ELLEY, S. 1905. Reports of the Chief and Assistant Veterinary Surgeons for 1905, Department of Agriculture, Cape of Good Hope, (G.47–1906).
- ENSLIN, P.R., JOUBERT, T.G. & REHM, S. 1954. Bitter principles of the Cucurbitaceae. Part II. Paper chromatography of bitter principles and some applications in horticultural research. Journal of the South African Chemical Institute, 7, 131–138.
- ENSLIN, P.R., JOUBERT, F.J. & REHM, S. 1956. Bitter principles of the Cucurbitaceae. III. Elaterase, an active enzyme for the hydrolysis of bitter principle glycosides. Journal of the Science of Food and Agriculture, 7, 646–655.
- ENSLIN, P.R., REHM, S. & RIVETT, E.A. 1957. Bitter principles of the Cucurbitaceae. VI. The isolation and characterization of six new crystalline bitter principles. Journal of the Science of Food and Agriculture, 8, 673–678.
- EVERIST, S.L. 1974. Poisonous plants of Australia. Sydney: Angus and Robertson Publishers.
- FITZGERALD, R.D., WOLFE, E.C., LABY, R.H. & HALL, D.G. 1980. Beef production from lucerne and subterranean clover pastures. 2. Bloat occurrence and effect of anti-bloat capsules. Australian Journal of Experimental Agriculture and Animal Husbandry, 20, 688–694.
- FOX, M.W. 1961. Castor seed residue poisoning in dairy cattle. Veterinary Record, 73, 885–886.
- GABBEDY, B.J., GWYNN, R., HOPKINSON, W.I. & KAY, B.E. 1974. Kikuyu poisoning of cattle in Western Australia. Australian Veterinary Journal, 50, 369–370.
- GASPAR, A.R.M.D., POTGIETER, D.J.J. & VERMEULEN, N.M.J. 1986. The effect of the sesquiterpene lactones from Geigeria on glycolytic enzymes. Biochemical Pharmacology, 35, 493–497.
- GEARY, T. 1950. Castor bean poisoning. Veterinary Record, 62, 472–473.
- GORST-ALLMAN, C.P., STEYN, P.S., VLEGGAAR, R. & GROBBELAAR, N. 1984. Structure elucidation of sesbanimide using high-field N.M.R. spectroscopy. Journal of the Chemical Society Perkin Transactions, 1, 1311–1131.
- GREEN, H.H. & ANDREWS, W.H. 1923. The toxicity of Adenia digitata Burtt-Davy (Modeca digitata Harv.). Report on Veterinary Research, Union of South Africa, 9 & 10, 381–392.
- GROSSKOPF, J.F.W. 1964. Our present knowledge of vermeersiekte (Geigeria poisoning). Technical communication No. 21. Department of Agriculture Technical Services. Republic of South Africa.
- HALL, J.W., WALKER, I. & MAJAK, W. 1994. Evaluation of two supplements for the prevention of alfalfa bloat. Canadian Veterinary Journal, 35, 702–705.
- HANSON, R.L., LARDY, H.A. & KUPCHAN, S.M. 1970. Inhibition of phosphofructokinase by quinone methide and α-methylene lactone tumor inhibitors. Science, 168, 378–380.
- HENDERSON, MAYDA & ANDERSON, J.G. 1966. Common weeds of South Africa. Memoir of the Botanical Survey of South Africa 37, Department of Agriculture Technical Services, Republic of South Africa.
- HENNING, M.W. 1932. Animal diseases in South Africa. South African Agricultural Series. Vol. XII. Central News Agency, Ltd: South Africa.
- HERZ, W. 1978. Sesquiterpene lactones from livestock poisons. In: KEELER, R.F., VAN KAMPEN, K.R. & JONES, L.F. (ed.) Effects of poisonous plants on livestock. Proceedings of a Joint United States–Australia Symposium on poisonous plants. Utah State University, Logan. Utah, June 19–24, 1977. New York: Academic Press.
- HOWARTH, R.E., GOPLEN, B.P., BRAND, S.A. & CHENG, K.J. 1982. Disruption of leaf tissues by rumen micro-organisms: An approach to breeding of bloat-safe forage legumes. Crop Science, 22, 564–568.
- HOWARTH, R.E., MAJAK, W., WALDERN, D.E., BRANDT, S.A., FESSER, A.C., GOPLEN, B.P. & SPURR, D.T. 1977. Relationships between ruminant bloat and the chemical composition of alfalfa herbage. I. Nitrogen and protein fractions. Canadian Journal of Animal Science, 57, 345–357.
- HUTCHEON, D. 1884. Report of the Veterinary Surgeon. Cape of Good Hope (G.31–85).
- HUTCHEON, D. 1886. Report of the Veterinary Surgeon. Cape of Good Hope (G.14–1887).
- HUTCHEON, D. 1893. Vomeerziekte or vomit sickness. Agricultural Journal of the Cape of Good Hope, 6, 24.
- HUTCHEON, D. 1902. Vomeerziekte of sheep. Agricultural Journal of the Cape of Good Hope, 21, 39.
- HUTCHEON, D. 1903. Vomeerziekte or mistbek in goats. Agricultural Journal of the Cape of Good Hope, 22, 219.
- HUTCHEON, D. 1904. Poisoning of horses from eating the plant Ornithogalum thyrsoides. Agricultural Journal of the Cape of Good Hope, 25, 48–50.
- HUTCHEON, D. 1906. Poisoning of horses by Ornithogalum thyrsoides or ‘Chinkerinchee’. Agricultural Journal of the Cape of Good Hope, 27, 165–171.
- IVIE, G.W., WITZEL, D.A., HERZ, W., RAMAMURTHI KANNAN, NORMAN, J., RUSHING, D.D., JOHNSON, J.H., ROWE, L.D. & VEECH, J.A. 1975. Hymenovin. A major toxic constituent of western bitterweed. Journal of Agriculture and Food Chemistry, 23, 841–845.
- JADHAV, S.J., SHARMA, R.P. & SALUNKHE, D.K. 1981. Naturally occurring toxic alkaloids in foods. CRC Critical Review in Toxicology, 9, 21–104.
- JOUBERT, J.P.J. 1983. Attempted prevention and treatment of Geigeria filifolia Mattf. poisoning in sheep. Journal of the South African Veterinary Association, 54, 255–258.
- JOUBERT, P.C. 1984. Are army worm outbreaks correlated with weather systems? Arena, Newsletter of the Natal Region, Department of Agriculture, 7, 19–20.
- KELLERMAN, T.S. 1983. Kikuyu (Pennisetum clandestinum) or army worm poisoning. Newsletter of the Onderstepoort Veterinary Research Institute, 1, 5.
- KELLERMAN, T.S., ADELAAR, T.F. & MINNE, J.A. 1972. The toxicity of the pollen beetle Astylus atromaculatus Blanch. Journal of the South African Veterinary Association, 43, 377–381.
- KELLERMAN, T.S., NAUDÉ, T.W. & FOURIE, N. 1996. The distribution, diagnosis and estimated economic impact of plant poisonings and mycotoxicoses in South Africa. Onderstepoort Journal of Veterinary Research, 63, 65–90.
- KIM, H.L., ANDERSON, A.C., HERRIG, B.W., JONES, L.P. & CALHOUN, M.C. 1982. Protective effects of antioxidants on bitterweed (Hymenoxys odorata DC.) toxicity in sheep. American Journal of Veterinary Research, 43, 194–195.
- KIM, H.L., HERRIG, B.W., ANDERSON, A.C., JONES, L.P. & CALHOUN, M.C. 1983. Elimination of adverse effects of ethoxyquin (EQ) by methionine hydroxy analog (MHA) protective effects of EQ and MHA for bitterweed poisoning in sheep. Toxicological Letters, 16, 23–29.
- KINGSBURY, J.M. 1964. Poisonous plants of the United States and Canada. Englewood Cliffs, New Jersey: Prentice-Hall, Inc.
- KIPTOON, J.C., MUGERA, G.M. & WAYAKI, P.J. 1982. Haematological and biochemical changes in cattle poisoned by Gnidia latifolia Syn. Lasiosiphon latifolius (Thymelaceae). Toxicology, 25, 129–139.
- KNIGHT, B. 1979. Ricin – a potent homicidal poison. British Medical Journal, 1, 350–351.
- KRAUSE, MARELI, NEL, ANNETTE & VAN ZYL, KATHY. 1996. A guide to the use of pesticides and fungicides in the Republic of South Africa. National Department of Agriculture, Pretoria, ISBN 0 621 15518 7.
- KUBO, S., MIMAKI, Y., SASHIDA, Y., NIKAIDO, T. & OHMOTO, T. 1992. Acylated cholestane glycosides from the bulbs of Ornithogalum saundersiae. Phytochemistry, 31, 3969–3973.
- KUPCHAN, S.M. 1970. Recent advances in the chemistry of tumor inhibitors of plant origin. Transactions of the New York Academy of Sciences, Series II, 32, No. 1, 85–106.
- KUPCHAN, S.M., FESSLER, D.C., EAKIN, M.A. & GIACOBBE, J.J. 1970. Reactions of alpha methylene lactone tumor inhibitors with model biological nucleophiles. Science, 168, 376–377.
- KUPCHAN, S.M., SHIZURI, Y., BAXTER, L.R. & HAYNES, H.R. 1977. Gnidione, a new furanosesquiterpene from Gnidia latifolia. Journal of Organic Chemistry, 42, 348–350.
- LAMPE, K.F. & FAGERSTROM, R. 1968. Plant toxicity and dermatitis. A manual for physicians. Baltimore: The Williams & Wilkins Company
- LETTY, CYTHNA, L., DYER, R.A., VERDOORN, INEZ, C. & CODD, L.E. 1962. Wild flowers of the Transvaal. Central News Agency.
- LOUW, C.N. & STEENKAMP, E.L. 1965. The occurrence of balls of vegetable origin in the digestive tract of sheep. Proceedings of the South African Society of Animal Production, 4, 134.
- LOWE, L.B., BALL, G.J., CARRUTHERS, V.R., DOBOS, R.C., LYNCHE, G.A., MOATE, P.J., POOLE, P.R. & V ALENTINE, S.C. 1991. Monensin controlled-release intraruminal capsule for control of bloat in pastured dairy cows. Australian Veterinary Journal, 68, 17–20.
- McCUNN, J., ANDREW, H. & CLOUGH, G.W. 1945. Castor bean poisoning in horses. The Veterinary Journal, 101, 136–138.
- McINTOSH, J.T. & COCKREM, F.R.M. 1977. Genetics of the susceptibility to bloat in cattle. II. Preliminary results from saliva samples from cows with high and low susceptibility. New Zealand Journal of Agricultural Research, 20, 263–268.
- MAJAK, W., HOWARTH, R.E., CHENG, K.J. & HALL, J.W. 1983. Rumen conditions that predispose cattle to pasture bloat. Journal of Dairy Science, 66, 1683–1688.
- MAJAK, W., HOWARTH, R.E., FESSER, A.C., GOPLEN, B.P. & PEDERSEN, M.W. 1980. Relationship between ruminant bloat and the composition of alfalfa herbage. II. Saponins. Canadian Journal of Animal Science, 60, 699–709.
- MAJAK, W. & HALL, J.W. 1990. Sodium and potassium concentrations in ruminal contents after feeding bloat-inducing alfalfa to cattle. Canadian Journal of Animal Science, 70, 235–241.
- MAJAK, W., HALL, J.W. & MAGAUGHEY, W.P. 1995. Pasture management strategies for reducing the risk of legume bloat in cattle. Journal of Animal Science, 73, 1493–1498.
- MAKKAR, H.P.S. & BECKER, K. 1997. Jatropha curcas toxicity: Identification of the toxic principles. Proceedings of the Fifth International Symposium on Poisonous Plants, San Angelo, Texas, USA, May 19–23, 1997.
- MARTINOVICH, D. 1979. Kikuyu toxicity. Surveillance, 6, 12–13.
- MARTINOVICH, D. & SMITH, B. 1972. Kikuyu poisoning in sheep. New Zealand Veterinary Journal, 25, 169.
- MARTINOVICH, D. & SMITH, B. 1973. Kikuyu poisoning of cattle. 1. Clinical and pathological findings. New Zealand Veterinary Journal, 21, 55–63.
- MARTINOVICH, D., MORTIMER, P.H. & DI MENNA, MARGARET, E. 1972. Similarities between so-called kikuyu poisoning of cattle and two experimental mycotoxicoses. New Zealand Veterinary Journal, 20, 57–58.
- METCALF, R.L., METCALF, R.A. & RHODES, A.M. 1980. Cucurbitacins as kairomones for diabroticite beetles. Proceedings of the National Acadamy of Science, USA, 77, 3769–3772.
- METTAM, R.W.M. 1933. Poisonous plants in East Africa. Reprint from Kenya Agricultural Journal, Government Press, Uganda.
- MILES, N., DE VILLIERS, J.F. & DUGMORE, T.J. 1995. Macromineral composition of kikuyu herbage relative to the requirements of ruminants. Journal of the South African Veterinary Association, 66, 206–212.
- MORRIS, J.J. 1945. Die vermeerbosvraagstuk in Griekwaland-Wes. Boerdery in Suid-Afrika, Augustus, 489–494.
- MORRIS, S.C. & LEE, T.H. 1984. The toxicity and tetratogenicity of Solanaceae glycoalkaloids particularly those of the potato (Solanum tuberosum): A review. Food Technology in Australia, 36, 118–124.
- MORTIMER, P.H., CAMPBELL, J., DI MENNA, MARGARET E. & WHITE, E.P. 1971. Experimental myrotheciotoxicosis and poisoning in ruminants by verrucarin A and roridin A. Research in Veterinary Science, 12, 508–515.
- NEWSHOLME, S.J., KELLERMAN, T.S., VAN DER WESTHUIZEN, G.C.A. & SOLEY, T. 1983. Intoxication of cattle on kikuyu grass following army worm (Spodoptera exempta) invasion. Onderstepoort Journal of Veterinary Research, 50, 157–167.
- NJOROROGE, GRACE, N. & NEWTON, L.E. 1994. Edible and poisonous species of Cucurbitaceae in the Central Highlands of Kenya. Journal of East African Natural History, 83, 101–115.
- OLSNES, S., REFSNES, K. & PIHL, A. 1974. Mechanism of action of the toxic lectins abrin and ricin. Nature, 249, 627–631.
- PAINE, R. 1904. Chinckerinchee poisoning. Report of Chief Veterinary Surgeon, Cape of Good Hope, 8.
- PENRITH, MARY-LOUISE & NAUDÉ, T.W. 1996. Mortality of chickens associated with blister beetle consumption. Journal of the South African Veterinary Association, 76, 97–99.
- PIENAAR, J.G., KRIEK, N.P.J., NAUDÉ, T.W., ADELAAR, T.F. & ELLIS, SHIRLEY D. 1973. Lesions in sheep skeletal and oesophageal muscle in vermeersiekte (Geigeria ornativa O. Hoffm. poisoning). Onderstepoort Journal of Veterinary Research, 40, 127–138.
- PIENAAR, J.P., TAINTON, N.M., VAN RYSSEN, J.B.J. & SWIEGERS, J.P. 1993 a. Factors affecting the voluntary intakes of sheep grazing Pennisetum clandestinum (kikuyu) pastures: Observations in the animal. African Journal for Range Science,10, 145–150.
- PIENAAR, J.P., TAINTON, N.M. & VAN RYSSEN, J.B.J. 1993 b. Factors affecting the voluntary intakes of sheep grazing Pennisetum clandestinum (kikuyu) pastures: Observations from forage analysis. African Journal for Range Science, 10, 140–144.
- PIETERSE, M.J. 1971. On the isolation of toxic constituents from Lasiosiphon burchellii Meisn. Agrochemophysica, 3, 63–66.
- PEROLD, G.W. 1957. The structure of geigerin. Journal of the Chemical Society, 47–51.
- PHILLIPS, P.E. 1926. A preliminary list of the known poisonous plants found in South Africa. Botanical Survey of South Africa. Memoir No. 9. Agricultural Department, Pretoria.
- PHILLIPS, C.J.C., JAMES, N.L. & MURRAY–EVANS, J.P. 1996. Effects of forage supplements on the incidence of bloat in dairy cows grazing high clover pastures. Veterinary Record, 139, 162–165.
- POWELL, R.G., PLATTNER, R.D. & SUFFNESS, M. 1990. Occurrence of sesbanimide in seeds of toxic Sesbania species. Weed-Science, 38, 148–152.
- QUIN, J.I. 1927. The toxicity of Ornithogalum saundersiae. South African Journal of Science, 24, 431–434.
- QUIN, J.I. 1928. The toxicity of Cucumis myriocarpus Naud. South African Journal of Science, 25, 242–245.
- QUIN, J.I. 1929. The toxic properties of Cucumis myriocarpus Naud. and Cucumis africanus Linn. Report on Veterinary Research, Union of South Africa, 15, 769–775.
- RADELEFF, R.D. 1964. Veterinary toxicology. London: Bailliére Tindal & Cox.
- REHM, S., ENSLIN, P.R., MEEUSE, A.D.J. & WESSELS, J.H. 1957. Bitter principles of the Cucurbitaceae. VII. The distribution of bitter principles in this plant family. Journal of the Science of Food and Agriculture, 12, 679–686.
- REYNOLDS, J.E. & PRASAD, ANNE B. (ed.) 1982. Martindale. The Extra Pharmacopoeia. 28th edn. London: The Pharmaceutical Press.
- RIMINGTON, C. 1933. The toxic principles of Cucumis africanus L.f., Cucumis myriocarpus (Naud). emend., and of new unnamed Cucumis species. South African Journal of Science, 30, 505–514.
- RIMINGTON, C. 1934. The chemical investigation of poisonous plants. Journal of the South African Veterinary Medical Association, 5, 227–244.
- RIMINGTON, C. 1935. Isolation of the toxic principles of Cucumis africanus L.f., Cucumis myriocarpus Naud. emend. Schweikerdt and of Cucumis leptodermis Schweikerdt sp. nov. and characterisation of trilactones belonging to the ‘Bitter Principle’ class. Onderstepoort Journal of Veterinary Science and Animal Industry, 4, 65–90.
- RIMINGTON, C. & ROETS, G.C.S. 1936. Chemical studies upon vermeerbos, Geigeria aspera Harv. Isolation of a bitter principle ‘Geigerin’. Onderstepoort Journal of Veterinary Science and Animal Industry, 7, 485–506.
- RIMINGTON, C., ROETS, G.C.S. & STEYN, D.G. 1936. Chemical studies upon the vermeerbos, Geigeria aspera Harv. II. Isolation of the active principle ‘Vermeeric acid’. Onderstepoort Journal of Veterinary Science and Animal Industry, 7, 507–520.
- RINDL, M. 1917. Note on the occurrence of daphnin in the Arthrosolen. Transactions of the Royal Society of South Africa, 6, 295–296.
- RINDL, M. 1933. The crystalline glucoside of Gnidia polycephala (Januariebossie). South African Journal of Science, 30, 455–457.
- RINDL, M. 1933. Isolation of a glycoside from Gnidia polycephala (Januariebossie). Transactions of the Royal Society of South Africa, 21, 239–244.
- ROBERTSON, W. 1905. Reports of the Chief Veterinary Surgeon and Assistant Veterinary Surgeons for 1905, Department of Agriculture, Cape of Good Hope. (G.47–1906).
- ROWE, L.D., KIM, H.L. & CAMP, B.J. 1980. The antagonistic effect of L-cysteine in experimental hymenoxin intoxication in sheep. American Journal of Veterinary Research, 41, 484–486.
- SANT, F.I. & WILSON, D. 1982. Use of cellulolytic enzyme digestion technique to distinguish bloat-causing from non-bloat causing legumes and to select for speed of mesophyll cell-wall disintegration in red clover (Trifolium pratense L.). Journal of Agricultural Science, Cambridge, 98, 99–102.
- SCHNEIDER, D.J. & DU PLESSIS, J.L. 1980. Enteritis in sheep due to the ingestion of Inula graveolens Desf. (Cape khaki-weed). Journal of the South African Veterinary Association, 51, 159–161.
- SCHNEIDER, D.J. & HUGO, L. 1980. Mortaliteit in lammers as gevolg van verstopping van die abomasum deur Ornithopus sativus Brot. (seradella) haarballe. Journal of the South African Veterinary Association, 51, 245–247.
- SCHNEIDER, D.J., MARASAS, W.F.O., COLLET, M.G. & VAN DER WESTHUIZEN, G.C.A. 1985. An experimental mycotoxicosis in sheep and goats caused by Drechslera campanulata, a fungal pathogen of green oats. Onderstepoort Journal of Veterinary Research, 52, 93–100.
- SHONE, D.K. & DRUMMOND, R.B. 1965. Poisonous plants of Rhodesia. Rhodesian Agricultural Journal, 62, 7–64.
- SKAIFE, S.H. 1953. African insect life. Cape Town: Longmans and Co. Green.
- SMITH, B. & MARTINOVICH, D. 1973. Kikuyu poisoning of cattle. 2. Epizootiological aspects. New Zealand Veterinary Journal, 21, 85–89.
- SNYMAN, L.D., SCHULTZ, R.A., KELLERMAN, T.S. & LABUSCHAGNE, L. 2002. Continuous exposure to an aversive mixture as a means of maintaining aversion to vermeerbos (Geigeria ornativa O. Hoffm.) in the presence of non–averted sheep. Onderstepoort Journal of Veterinary Research, 69, 321–325.
- SPENCER, K.C., SEIGLER, D.S. 1982. Tetraphyllin R and epi-tetraphyllin B from Adenia glauca Schinz. Onderstepoort Journal of Veterinary Research, 49, 137–138.
- SPREULL, J. 1899. Report of the Veterinary Surgeon, Cape of Good Hope, 43.
- STEYN, D.G. 1929. Recent investigation into the toxicity of known and unknown poisonous plants in the Union of South Africa. Report on Veterinary Research, Union of South Africa, 15, 777–803.
- STEYN, D.G. 1931. Investigation into the cause of alopecia (Kaalsiekte) in kids and goats. Report on Veterinary Research, Union of South Africa, 17, 729–756.
- STEYN, D.G. 1931. Recent investigations into the toxicity of known and unknown poisonous plants in the Union of South Africa. Reports on Veterinary Research, Union of South Africa,17, 707–722.
- STEYN, D.G. 1932–1933. The toxicity of ‘maranc’. Journal of the South African Veterinary Medical Association, 3 & 4, 44–45.
- STEYN, D.G. 1932. Known poisonous plants in the Union of South Africa. Journal of the South African Veterinary Medical Association, 3 & 4, 178–189.
- STEYN, D.G. 1932. Investigations into the toxicity of known and unknown poisonous plants in the Union of South Africa. Report on Veterinary Research, Union of South Africa, 18, 871–891.
- STEYN, D.G. 1932. Chrysocoma tenuifolia Berg. poisoning in Angora goats and the development of tolerance. Report on Veterinary Research, Union of South Africa, 18, 893–898.
- STEYN, D.G. 1934. The toxicology of plants in South Africa. Central News Agency: South Africa.
- STEYN, D.G. 1935. Recent investigations into the toxicity of known and unknown poisonous plants in the Union of South Africa. Onderstepoort Journal of Veterinary Research and Animal Science, 4, 399–415.
- STEYN, D.G. 1936. Recent investigations into the toxicity of known and unknown poisonous plants in the Union of South Africa. Onderstepoort Journal of Veterinary Science and Animal Industry, 7, 169–178.
- STEYN, D.G. 1943. Vermeersiekte by veediere. Boerdery in Suid- Afrika, Reprint number 107.
- STEYN, D.G. 1949. Vergiftiging van die mens en dier. Pretoria: Van Schaik.
- STEYN, D.G. & VAN DER WALT, S.J. 1942. Chinkerinchee or star of Bethlehem poisoning of stock. Farming in South Africa. Reprint No. 12.
- TANNER, G.J., MOATE, P.J., DAVIS, L.H., LABY, R.H., LI YUGUANG & LARKIN, P.J. 1995. Proanthocyanidins (condensed tannin) destabilise plant protein foams in a dose dependent manner. Australian Journal of Agricultural Research, 46, 1101–1109.
- TERBLANCHE, M., DE KLERK, W.A., SMIT, J.D. & ADELAAR, T.F. 1966. A toxicological study of the plant Sesbania punicea Benth. Journal of the South African Veterinary Medical Association, 37, 191–197.
- TERBLANCHE, M., PIETERSE, M.J., ADELAAR, T.F. & SMIT, J.D. 1966. Further studies of the toxicology of Lasiosiphon burchellii Meisn. (Harpuisbos). Journal of the South African Veterinary Medical Association, 37, 185–189.
- TUBB, T.F., CREEPER, J.H. & MC KENZIE, R.A. 1995. Poisoning of cattle attributed to Cucumis melo spp. agrestis (Ulcardo melon). Australian Veterinary Journal, 72, 274–275.
- VAHRMEIJER, J. 1981. Poisonous plants of Southern Africa that cause stock losses. Cape Town: Tafelberg.
- VAN ASWEGEN, C.H., POTGIETER, D.J.J. & VERMEULEN, N.M.J. 1982. Site of respiratory inhibition by sesquiterpene lactones from Geigeria. South African Journal of Science, 78, 125–128.
- VAN ASWEGEN, C.H., VERMEULEN, N.M.J. & POTGIETER, D.J.J. 1979. Inhibition of oxidative phosphorylation by sesquiterpene lactones from Geigeria aspera. South African Journal of Science, 75, 84–85.
- VAN DER LUGT, J.J. 2002 The clinicopathology and pathology of selective toxicoses and storage diseases of the nervous system of ruminants in southern Africa. Doctoral thesis, Utrecht University.
- VAN DER LUGT, J.J. & VAN HEERDEN, J. 1993. Experimental vermeersiekte (Geigeria ornativa O. Hoffm. poisoning). II: Histological and ultrastructural lesions. Journal of the South African Veterinary Association, 64, 82–88.
- VAN DER WESTHUIZEN, D. 1996. Structural studies on the toxic principle of Ornithogalum thyrsoides. MSc thesis, University of Pretoria.
- VAN HEERDEN, J., WILLIAMS, M.C., VAN RENSBURG, I.B.J. & IPLAND, F.F. 1978. An outbreak of ‘kikuyu poisoning’ in Western Transvaal. Journal of the South African Veterinary Association, 49, 27–30.
- VAN HEERDEN, J. & VAN DER LUGT, J.J. & DURANTE, E. 1993. Experimental vermeersiekte (Geigeria ornativa O. Hoffm. poisoning) in sheep. I: An evaluation of diagnostic aids and an assessment of the preventive effect of ethoxyquin. Journal of the South African Veterinary Association, 64, 76–81.
- VERMEULEN, N.M.J. & PIETERSE, M.J. 1968. The isolation of umbelliferone and an active principle from Arthrosolen polycephalus (Januariebos). Journal of the South African Chemical Institute, 21, 190–191.
- VERMEULEN, N.M.J., VOGELZANG, M.E. & POTGIETER, D.J.J. 1978. Dihydrogriesenin in Geigeria aspera Harv. Agrochemophysica, 10, 1–3.
- VOET, D. & VOET, J. 1990. Three-dimensional structures of proteins. In: Biochemistry, 158–159. New York: John Willy & Son.
- VOGELZANG, MARION E., VERMEULEN, N.M.J., POTGIETER, D.J.J. & STRAUSS, H.F. 1978. Ivalin in Geigeria aspera. Phytochemistry, 17, 2030–2031
- VON JENEY DE BORESJENÖ, N.L.T.R.M., POTGIETER, D.J.J. & VERMEULEN, N.M.J. 1974. Detection and thin-layer chromatography of sesquiterpene lactones from Geigeria species. Journal of Chromatography, 94, 255–260.
- WAGHORN, D.C. 1991. Relationships between intra-ruminal pressure, distension, and the volume of gas used to stimulate bloat in cows. New Zealand Journal of Agricultural Research, 34, 213–220.
- WATT, J.M. & BREYER–BRANDWIJK, M.G. 1968. The medicinal and poisonous plants of Southern and Eastern Africa. 2nd edn. Endinburgh and London: E & E Livingstone Ltd.
- WOOD, P.A., FOGGIN, D.C. & NAUDÉ, T.W. 1997. Suspected calcium oxalate raphide irritation in a black rhinoceros (Diceros bicornis) due to ingestion of Xanthosoma mafaffa. Journal of the South African Veterinary Association, 68, 2.
- WONG, P.T., ROTH, I.J. & JACKSON, A.R.B. 1987. Kikuyu poisoning of cattle in New South Wales and its relationship to pasture fungi on kikuyu. Australian Veterinary Journal, 64, 229–231.
- ZUMPT, I. 1971. Impaction of the rumen in cattle due to the ingestion of the cocoons of the Molopo caterpillar Gonometa postica (Walker). Journal of the South African Veterinary Medical Association, 42, 315–318.