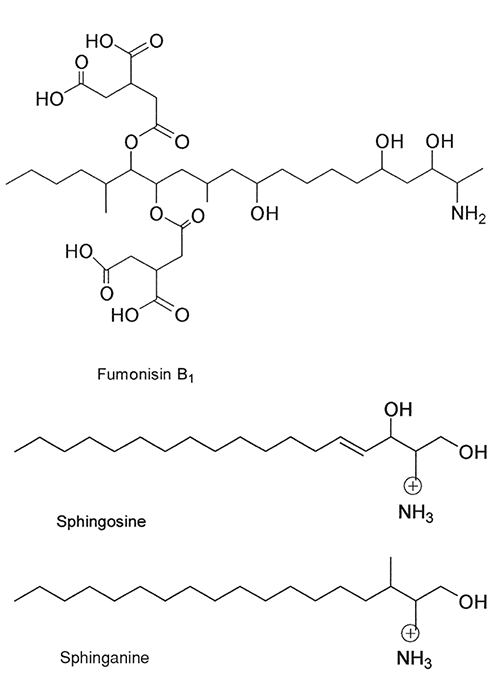
- Plant Poisonings and Mycotoxicoses of Livestock in Southern Africa, 2nd Edition
- Central nervous system
Central nervous system
| Without pathological lesions | Stenocarpella maydis | Albizia spp. | Cynanchum spp. | Sarcostemma viminale | Euphorbia mauritanica | Dipcadi glaucum | Annual ryegrass toxicosis | Melica decumbens | Kweek tremors | Paspalum staggers | Perennial ryegrass staggers | Lupine poisoning | Melia azedarach | Datura spp. | Pteridium aquilinum | Nicotiana glauca | Ficus ingens | Ficus salicifolia | Nierembergia linariifolia |
| With pathological lesions | Solanum tettense | Ipomoea carnea | Aspergillus clavatus | Valsiekte | Trachyandra spp. | Phalaris staggers | Helichrysum argyrosphaerum | Cotula nigellifolia | Fusarium verticillioides |
This content is distributed under the following licence: Attribution-NonCommercial CC BY-NC  View Creative Commons Licence details here
View Creative Commons Licence details here

Introduction
A number of poisonous plants and fungi can cause nervous signs in livestock in southern Africa. Since no chemical analyses are available for the diagnoses of these neurotoxicoses, the only clues to the identity of the aetiological agents involved may be provided by the lesions in the central nervous system (CNS).
Where no pathological lesions are discernible, a diagnosis must be made merely on circumstantial evidence, such as the presence of a grazed toxic plant/fungus capable of causing the observed nervous signs. Sometimes recognizable fragments of plants, such as the pods of Albizia tanganyicensis or pieces of Cynanchum ellipticum in the rumen, may aid in the making of a diagnosis.
Since pathological lesions are among the most reliable criteria on which diagnoses can be made, the neurotoxicoses have been divided into two groups, depending on whether or not notable lesions are present in the CNS.
Neurological disorders without notable pathological lesions
Stenocarpella maydis (Berk.) Sacc. (= Diplodia maydis) (Fungi: Coelomycetes)
Diplodiosis is a southern African neuromycotoxicosis of cattle and sheep, grazing on harvested maize lands in winter. The disease, caused by the ingestion of maize (Zea mays) infected with S. maydis, is characterized by ataxia, paresis and paralysis.111, 124, 125, 135, 136, 202 In addition, the offspring of dams that had been exposed to infected maize may be stillborn or non-viable.106, 109, 110, 160
Diplodiosis is one of the commonest nervous disorders of cattle and sheep in southern Africa where, in South Africa alone, it is believed to be responsible for 2% of all mortalities from plant poisonings and mycotoxicoses.106 Together with facial eczema in New Zealand and lupinosis in Australia, diplodiosis is one of the most important mycotoxicoses of ruminants in the world.110
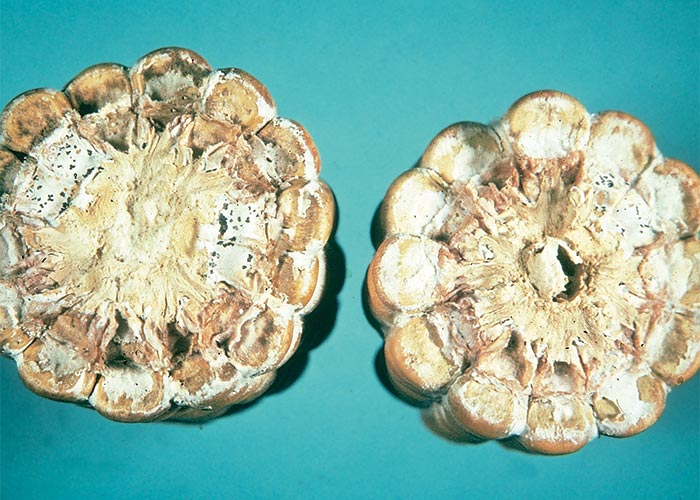
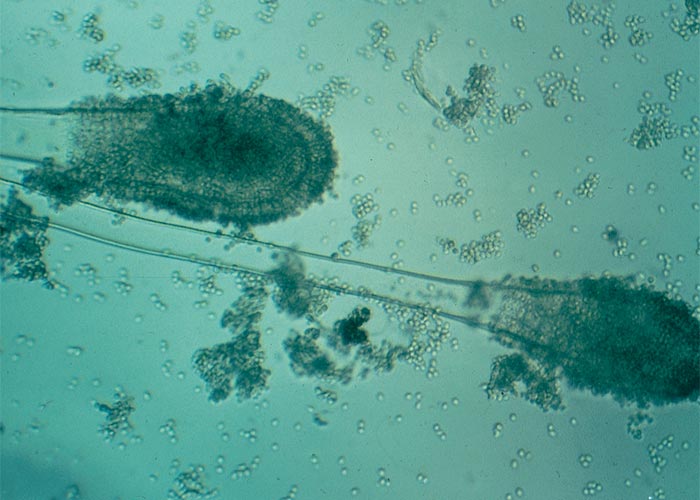
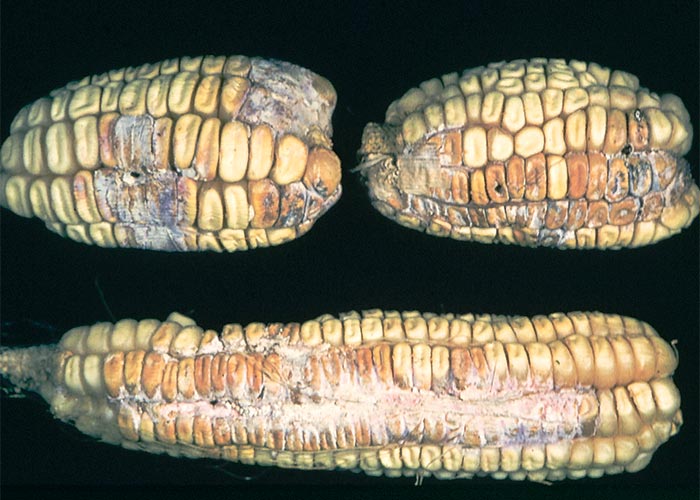
The fungus causes a stem and ear rot of maize. The ear rot often starts at the base of the cob where a coarse white mycelial mat is formed that turns the invaded kernels a greyish-brown colour. Characteristic black fruiting bodies or pycnidia, seen as pinhead-sized spots, are produced on the affected parts towards the end of the growing season (Figures 1 and 2). Without these black spots S. maydis is difficult to distinguish macroscopically from Fusarium verticillioides or other fungi on the cobs. The fungus overwinters in the pycnidial form on maize residues. Air or insect-borne conidia infect the new crop in summer, usually from the flowering stage onwards, when the plants become senescent.89, 123, 207
Stenocarpella maydis is encountered throughout the world wherever maize is grown,123 but apart from a rare report of a suspected outbreak in Australia,49 diplodiosis is known to occur only in southern Africa. The isolation of toxigenic strains of S. maydis from bulk consignments of maize imported from North and South America is, therefore, puzzling in the light of the absence of diplodiosis in these continents.111 Marasas124 suggested that the lack of recorded outbreaks outside southern Africa could be attributed to a combination of factors including agricultural practices (only in southern Africa are reaped lands extensively grazed for winter forage), variability in toxicity of naturally infected maize, variability in the toxigenic potential of S. maydis strains and differences in the susceptibility of animals.
Toxic compounds have been isolated from S. maydis cultures191 but these have not been tested for neurotoxicity in ruminants. Until the neurotoxin is known, diplodiosis will continue to be experimentally reproduced only by feeding naturally infected maize or pure cultures of the fungus or their extracts to ruminants.111, 135, 136, 178, 202 Cultures can be prepared by incubating sterile moistened maize seeds, inoculated with conidia, for at least eight weeks at 28 °C.111, 123, 124, 162 The duration of incubation is of critical importance as cultures grown for less than eight weeks apparently are non-toxic.111, 123, 124, 162 However, not all isolates cultured for eight weeks are neurotoxic,109, 111 e.g. 63 kg of culture, lethal to ducklings, failed to induce ill effects in a 320 kg cow (C.J. Rabie and T.S. Kellerman, VRI, Onderstepoort, unpublished data, 1980). This lack of correlation between mortality in ducklings and diplodiosis casts doubts on the suitability of ducklings as a model for the bioassay of chemical fractions in the isolation of the bovine neurotoxin.
Among the reasons offered for only ruminants being affected by diplodiosis is that the nervous systems of monogastric animals are refractory to the toxin or that a precursor is bioactivated to a toxic substance in the rumen. The recent induction of diplodiosis in laboratory animals with extracts of S. maydis cultures showed that the active principle was a primary toxin and removed a major impediment to the extraction of the neurotoxic component, namely the lack of a biological assay for testing the toxicity of chemical fractions. Using a guinea-pig bio-assay, with typical paretic signs as guide, the neurotoxic principle(s) of a culture has now been purified by column chromatography to a few components (L.D. Snyman, ARC-OVI, unpublished data, 2003).
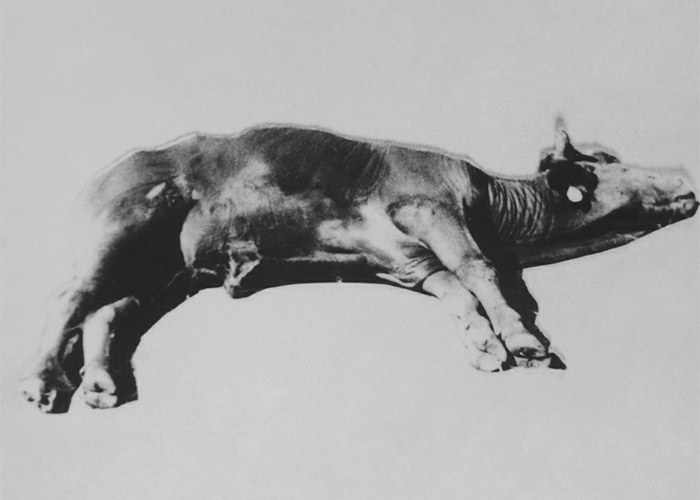
Cattle, sheep and goats usually develop signs 2–8 days after receiving 10–40 g/kg culture in doses of c.5–10 g/kg/day.109, 111 The clinical signs, typically lasting for 1–4 days, included reluctance to move, standing with a wide-based stance, incoordination (walking with a stiff-legged, high-stepping gait), falling, paresis/paralysis, constipation, salivation and occasional tremors (Figure 3). A small percentage (10%) of cases may become almost totally paralysed and lie in lateral recumbency for a week or more. Under extensive farming conditions such animals may die of gassy bloat, hunger or thirst, but with good nursing most can be expected to recover. Although the mortality in the field at times can be high,135 the prognosis is excellent if stock are removed from the toxic lands soon after the signs appear. None of the 35 intoxicated animals in an experiment succumbed to diplodiosis111 (T.S. Kellerman and C.J. Rabie, VRI, Onderstepoort, unpublished data, 1984). Recovery is complete and lesions in the central nervous system are rare.
An unusual feature of the disease is that new cases may appear up to 14 days after stock has been moved from toxic fields.3, 189 In a recent dosing trial a steer ostensibly suffered a relapse 22 days after dosing had ceased.111 Should this observation be correct, some of the new cases appearing after stock have been removed from the toxic lands may well be animals suffering from such relapses.
During 1987 when cob rot was rife in South Africa, a new and disturbing aspect of diplodiosis came to light. Sporadic reports were received of perinatal losses in flocks and herds that had been exposed to diplodiosis. The affected calves and lambs were either stillborn or died soon after birth.
Dosing trials at Onderstepoort Veterinary Institute involving 82 ewes revealed that 66% of the offspring of ewes exposed to cultures of Stenocarpella maydis in the second trimester of pregnancy and 87% of the lambs of those exposed during the third trimester were either born dead or died soon after birth. The lambs of ewes that had shown nervous signs and those of ewes that had not shown such signs were equally affected. Perinatal losses, therefore, can and do occur in healthy flocks that had never shown signs of diplodiosis.109
Foetuses are clearly more susceptible to diplodiosis than adults. In dosing trials, only 44% of the dams exposed to cultures in the third trimester were affected compared to 87% of their lambs.109 Perinatal losses have also been experimentally induced with cultures of S. maydis in cattle (T.S. Kellerman and R.A. Schultz, OVI, unpolished data, 1993).
Gross pathological changes are not normally present in ruminating animals suffering from diplodiosis. In a recent dosing trial with pure cultures, a laminar subcortical status spongiosus was evident in the cerebrum and cerebellum of a sheep that had been paralysed for a number of days and in a steer with irreversible ataxia.111 Save for these two rare instances, microscopical lesions have not been reported in the disease.
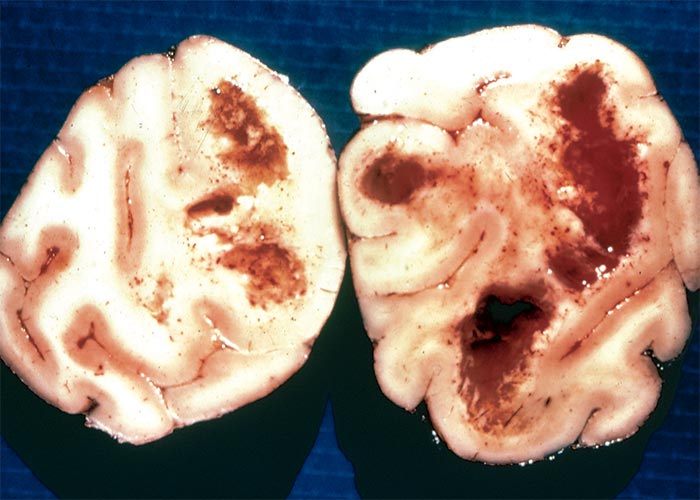
In contrast to post-natally affected animals, stillborn and non-viable lambs have pronounced microscopical lesions in their central nervous systems.160 Spongiform degeneration of myelin of varying degree was present in the brains of all the affected lambs in these trials. Myelin lamellae were noticed to be separated at the intrapod line. The spongiform degeneration occurred throughout the central nervous system in severely affected animals, whereas in mildly affected lambs the lesions had a predilection for the white matter, particularly of the cerebellum and cerebrum. In more than half the lambs, spongiform degeneration progressed to lytic necrosis and two showed hydrocephalus. A status spongiosus similar to that of lambs in the trial was evident in the white matter of the brains of naturally poisoned neonates (L. Prozesky, OVI, personal communication, 1993). The nature of the lesions, that the foetus is susceptible when myelin is being formed and lack of lesions in post-natally affected animals, suggest that the unidentified toxin acts primarily on myelin.160
A diagnosis of diplodiosis in post-natally affected animals is made on circumstantial evidence, such as the clinical signs and a history of exposure to maize stover. Diplodiosis in stillborn and non-viable lambs, on the other hand, is confirmed histopathologically by the presence of a status spongiosus in the white matter of their brains.106, 160
There is no antidote for diplodiosis. When treating diplodiosis one must remember that the salivation is ascribed by some clinicians to paresis of the deglutition muscles. Although experimental evidence could not be found for this,109, 111 the possibility of swallowing impairment must be borne in mind when affected stock are drenched.
The disease is controlled by removing stock from the toxic lands as soon as the first signs appear. According to some observers, diplodiosis seldom if ever occurs in cattle fed on processed maize, e.g. when milled on the cob or ground and incorporated into rations. If no other roughage is available, therefore, the toxic maize litter can be fed to stock after being passed through a hammer mill. The milled plants are thought to be less toxic because the mouldy cobs are broken up and distributed throughout the material, thus diluting the toxin.
The finding109 that ewes became significantly resistant to intoxication after initial exposure to cultures of S. maydis may find practical application in the utilization of toxic lands. Note that the resistance applies only to the neuro- and not the foetotoxic aspects of the condition. During years of severe cob rot, therefore, male animals and empty females rather than heavily pregnant cows and ewes should be put to graze on the harvested maize lands.
Since S. maydis on maize residues is a prime source of infection for the next crop, removal of the residue by deep early ploughing or burning, for instance, should decrease the prevalence of diplodiosis in the following winter.89 Other cultivation practices (or even the choice of cultivar) might influence the occurrence of the disease, e.g. the incidence of diplodiosis can be expected to rise on lands harvested by hand, such as seed-lands, where spoilt cobs are discarded on the ground (T.S. Kellerman, VRI, Onderstepoort, personal observations, 1986). Dry, warm conditions, in the early growth stage of the crop, followed by damp, cloudy periods at f lowering, are especially conducive to multiplication of this phytopathogenic fungus.89
Diplodiosis can be distinguished from Aspergillus clavatus poisoning by the low mortality, the lack of tremors and the absence of CNS lesions. Superficially, diplodiosis bears some resemblance to many nervous disorders, including heartwater, but in practice it is most easily confused with botulism, kweek tremors and Paspalum staggers. Whenever cattle develop nervous signs on harvested maize lands in southern Africa, diplodiosis must be one of the first diagnosis to be considered.
Albizia spp. (Fabaceae)
A. versicolor Welw.
Poison pod Albizia, grootblaar valsdoring
A. tanganyicensis Bak. f.
Paper bark Albizia, papierbas valsdoring
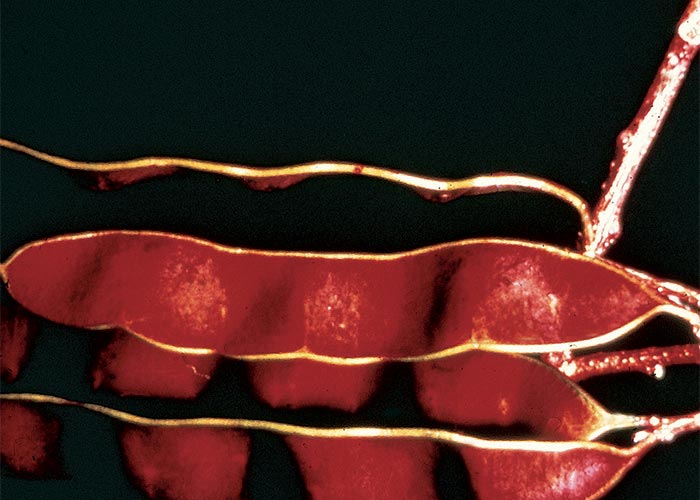
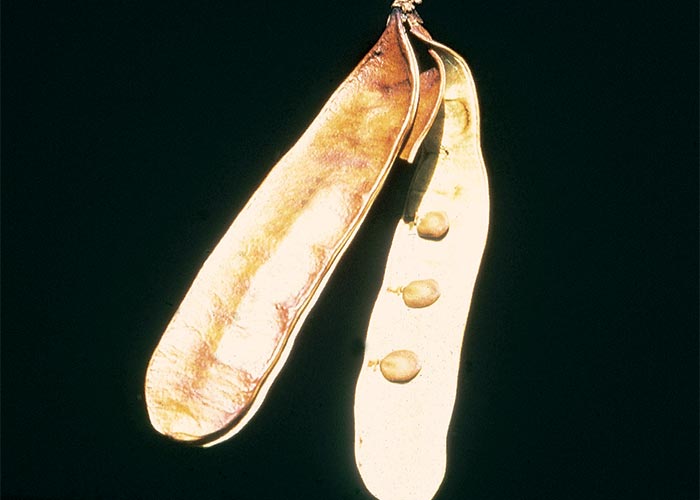
The pods of Albizia versicolor and A. tanganyicensis are associated with hypersensitivity, intermittent tetanic convulsions and mortality, mainly of cattle in southern Africa.10, 144
Albizia versicolor (Figure 4) is a medium to large tree, usually c.10 m in height, with a rounded or spreading crown. Albizia tanganyicensis, on the other hand, is a medium- sized, sparingly branched, more upright tree of only c.3–8 m, with characteristic brownish-red, thin, papery bark. The bark often peels in broad papery strips revealing a pearly, creamy-grey under-surface (Figures 7 and 8).
The flowers of both species consist of half-spherical fluffy heads with stamens of up to 30 mm long (Figure 5). Flowering time is from August to November. The large broad, thin, chestnut-brown pods (Figures 6 and 9) of up to 250 mm x 65 mm in size contain several flat seeds which ripen only in September to October of the following year.45, 205
Both species are deciduous, with large twice compound (bipinnate) leaves, having opposite leaf lets.
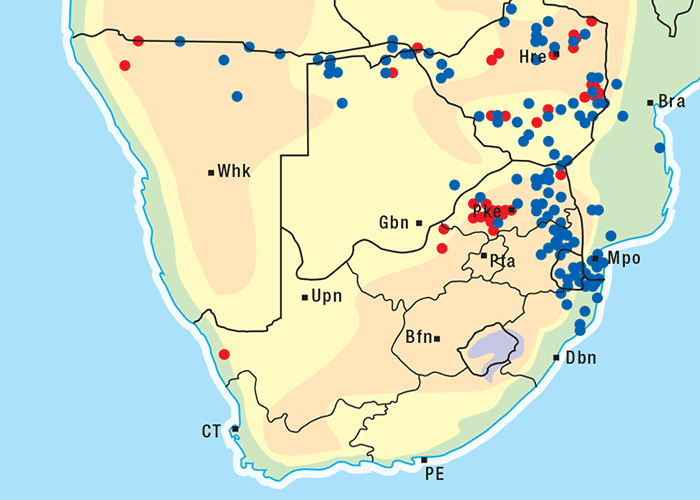
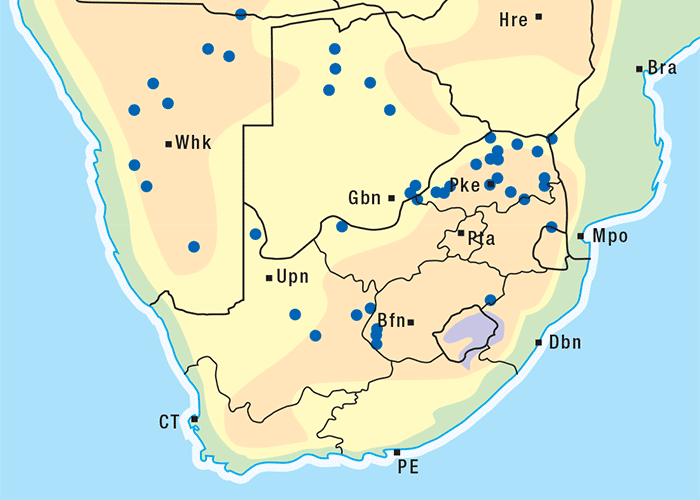
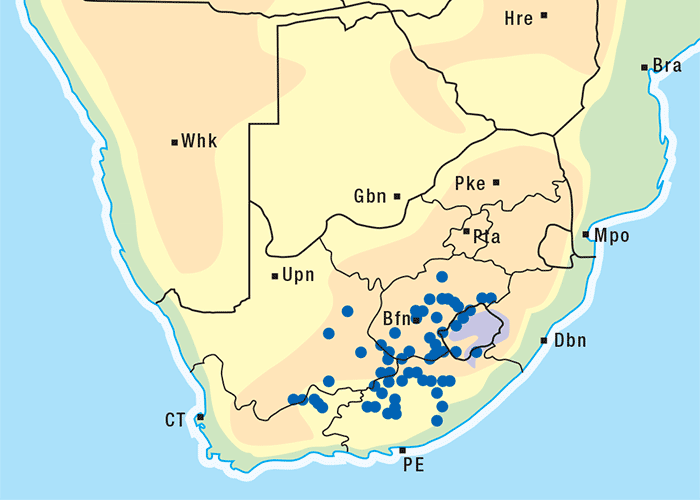
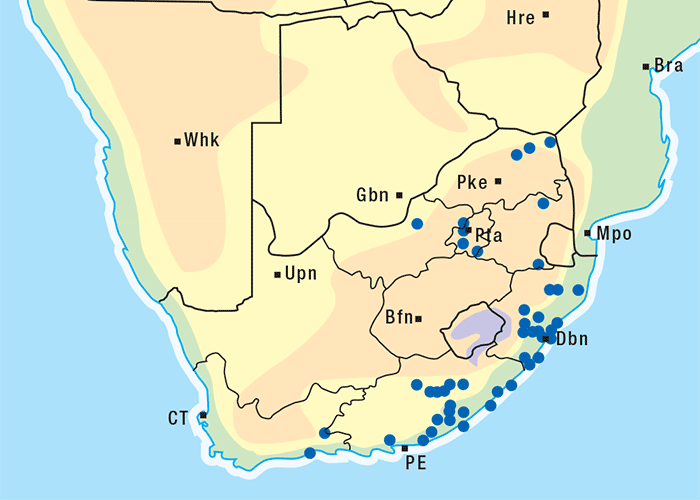
Albizia versicolor and A. tanganyicensis are fairly well represented in the subcontinent north of the Limpopo, and both occur together in the far Limpopo Province of South Africa and in Zimbabwe. Generally speaking, A. versicolor has a more easterly distribution, stretching along eastern Mpumalanga through Swaziland into KwaZulu-Natal and beyond. Albizia tanganyicensis, in turn, occupies the northwestern parts of the Limpopo Province (Figure 10).
Albizia tanganyicensis usually grows on the slopes of koppies, especially those with granite or quartzite rock formations.45, 205 Albizia versicolor is often associated with riverbanks, flood plains and flats (G. Germishuizen, Botanical Research Institute, personal communication, 1985).
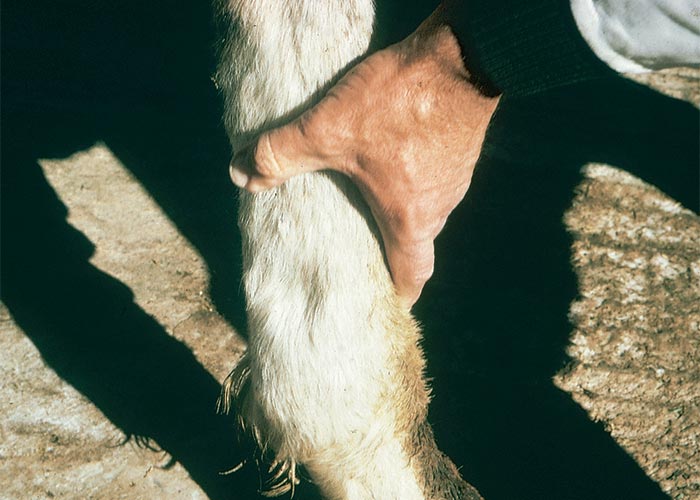
Outbreaks of albiziosis in cattle usually occur in late winter or early spring when the pods are blown from the trees by high winds. Young pods are the most toxic, the toxin being concentrated in the pod cases and to a lesser extent in the seeds. Drying of seeds in the air can apparently reduce their toxicity by half. As little as 0,57–1,14 kg of A. versicolor pods have proved fatal to cattle with body masses of c.230 kg,144 and sheep that received 5 g/kg ripe pods of A. tanganyicensis developed severe clinical signs and died10. Although in South Africa natural outbreaks are rarely reported in sheep, they are thought to be twice as susceptible as cattle to the poisoning.144 Drinking large quantities of water can apparently precipitate the clinical signs.144
In South Africa bovine albiziosis occurs sporadically mostly in the Limpopo Province (T.S. Kellerman, personal observation, 1997), large numbers of cattle purportedly died of this poisoning in Zimbabwe early in the seventies (S.K. Hargreaves, Department of Veterinary Services, Causeway, Zimbabwe, personal communication, 1992) and in 1996 the first outbreak in small stock was recorded in sheep and goats poisoned by A. versicolor in Malawi.180
The Malawi outbreak was described as follows: ‘Approximately 800 animals are estimated to have died over a 9-year period on a government farm near Lake Malawi. Deaths occurred exclusively from August to December when ripe pods that fell to the ground were ingested. The major clinical signs were hyperaesthesia, wild running, lateral recumbency with rapid leg movements, nystagmus and rapid blinking. Approximately 75% of clinical cases made a full and rapid recovery. Sheep more often showed signs of poisoning than goats which was attributed to inherent susceptibility rather than selective feeding. The majority of animals affected were under 1 year of age. A series of experiments was conducted and all the animals dosed with 6.4 g/kg or more of dry pods died with typical clinical signs’.180
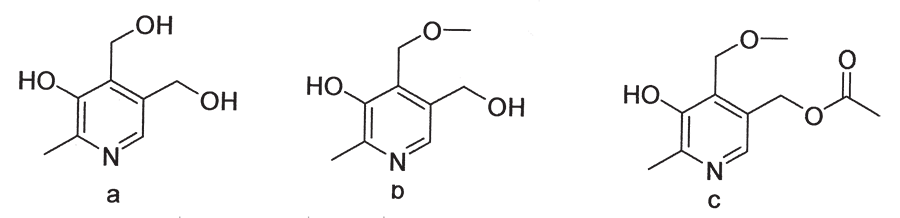
Steyn, Vleggaar and Anderson (1987) extracted the toxic principle, 4ᶦ -methoxypyridoxine and its 5ᶦ -acetoxymethyl derivative,192 from the pods of A. tanganyicensis (Figure 11). Noting the similarity between this toxin and vitamin B6, they surmised that it acted as an antagonist to this vitamin. Trials with guinea-pigs confirmed that vitamin B6 administered subcutaneously at 10 mg/kg was remarkably efficacious in the treatment of Albizia poisoning.84 The treatment was equally successful in sheep, the recommended dose of pyridoxine hydrochloride being 20–25 mg/kg given intravenously or subcutaneously eight hours apart. Two treatments are necessary because relapses occur and the route of administration depends on the severity of the signs.85
Contrary to expectations, pyridoxal was found to be ineffective as a therapeutic agent. This compound, an intermediate between pyridoxine and pyridoxal phosphate (the active or intracellular form of vitamin B6), was expected to act more quickly and effectively than its precursor. The lack of activity of pyridoxal suggests that the toxin has an atypical site of action with regard to the normal pathways requiring vitamin B6 as a co-factor.85
The majority of animals are found dead with signs of severe struggling evident on the ground around them. Those that survive are hypersensitive, walk with a staggering gait and intermittently fall down in tetanic convulsions. A wild expression in the eyes, nystagmus and dyspnoea may accompany these signs. Death apparently results from heart failure. All the experimentally intoxicated sheep had markedly elevated body temperatures, probably as a result of the violent convulsions.10
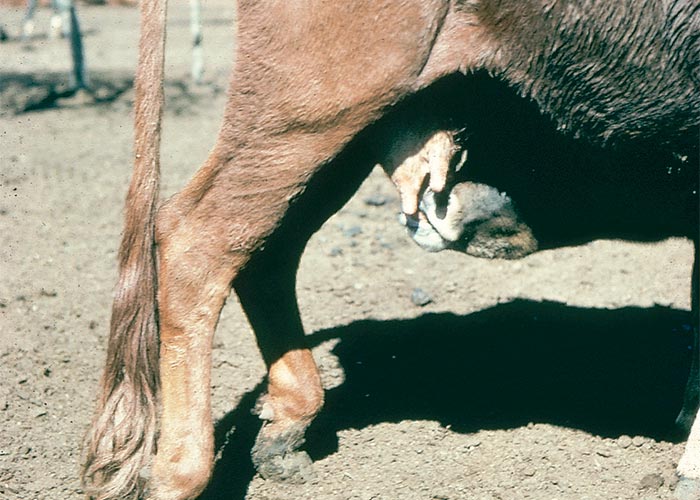
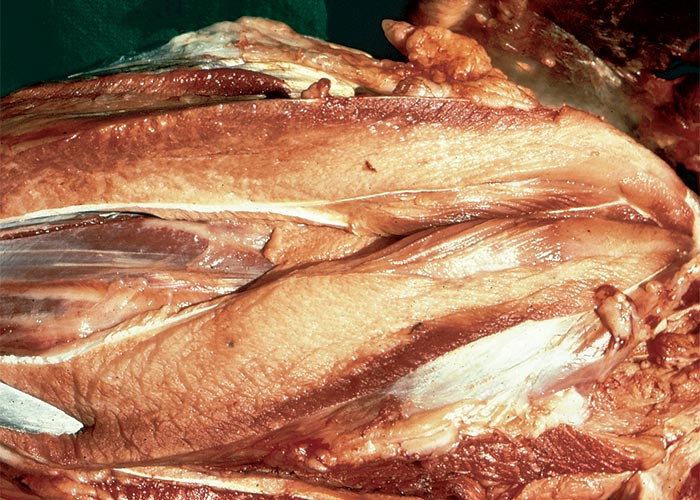
At necropsy, portions of pods are invariably found in the rumen as well as seeds in the reticulum of cattle (Figure 12).10, 144 The carcases are congested and often cyanotic, with petechiae in various tissues and organs, such as the subcutis, skeletal muscles, myocardium, lymph nodes, thymus, trachea and abomasum. Lung congestion and oedema occur in some animals. In sheep experimentally poisoned with the pods of A. tanganyicensis, the myocardium had a prominent par-boiled appearance.10
In the two reports on the microscopical lesions – both involving sheep10, 85 – the most significant changes occurred in the CNS and myocardium. These animals showed a mild to moderate congestion of the brain and spinal cord, with chromatolysis of many neurons. There was also slight microcavitation, thought to be oedema, especially in the white matter.10 Gummow et al., 1992 reported only subdural haemorrhages in the ventral region of the midbrain and spinal cord and microhaemorrhages at various sites in the CNS85. Cloudy swelling, Zenker’s changes, fragmentation and lysis of myocytes were diffusely distributed in the myocardium, while non-specific, degenerative changes occurred in the skeletal muscles, liver, kidneys, lungs and lymph nodes. It is suspected that the markedly elevated body temperature is responsible for the changes in most organs and tissues.
The only preventive measure that can be taken is to graze the animals in camps where there are no Albizia trees during the critical months of August to November.
Albiziosis can be confused with poisoning by chlorinated hydrocarbon pesticides and Sarcostemma viminale. The distribution of the trees coincides to a large extent with endemic heartwater areas and care should be taken not to confuse these two diseases.
Cynanchum spp. (Apocynaceae)
C. africanum L. Hoffmanns.
C. ellipticum (Harv.) R.A. Dyer (= C. capense)
C. obtusifolium L. f.
Monkey rope, klimop, bobbejaantou
Cynanchosis is a nervous disorder of domestic ruminants and more rarely of horses, that clinically resembles chlorinated hydrocarbon poisoning. The condition is characterized by hypersensitivity, incoordination, recurrent tetanic seizures and paralysis.48, 61, 92, 93, 95, 178, 185, 216, 220
Cynanchum ellipticum is a climber with a slender many-jointed stem and branches. The broadly elliptical leaves each have a point at the end of the midrib, and arise opposite each other in pairs. At the junction of the leaves and stem, the stem has a ring-like thickening. The pungent, white to greenish f lowers (Figure 13), have a distinctive tubular corolla. Without their flowers with the characteristic corolla, C. ellipticum cannot easily be distinguished from other Cynanchum spp. (Figure 14). The fruits, shaped like a pair of horns are packed with club-shaped plumed seeds. All Cynanchum species contain a milky, bitter, apparently non-irritant latex.93, 205, 216 The plants are palatable to livestock and some animals even have a predilection for them.
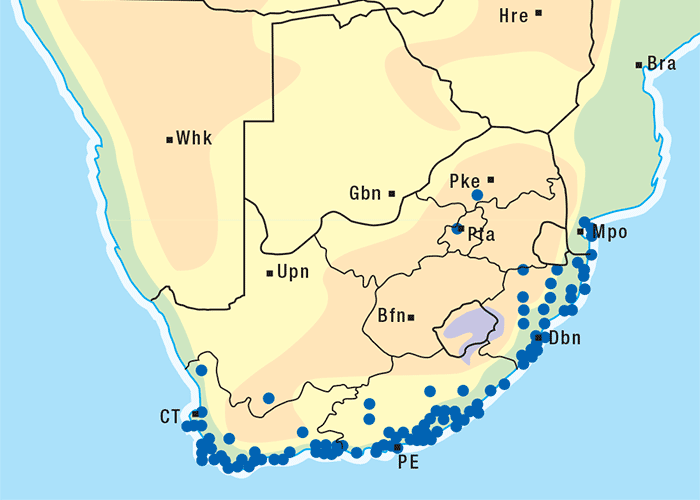
Cynanchum ellipticum festoons trees and shrubs in the coastal bush along the eastern seaboard (Figure 15) and in certain wooded inland valleys, e.g. of the Soutpansberg Range in the Limpopo Province.92, 205 The distribution of C. obtusifolium is similar to that of C. ellipticum.189 Cynanchosis, however, has not been recorded in the interior provinces of South Africa. Cynanchum africanum generally occurs along the coast from about Isipingo to the vicinity of Clanwilliam, being most common in the Eastern and Western Cape provinces.92, 189
Cynanchum ellipticum,92 C. africanum48 (T.S. Kellerman and L.A.P. Anderson, VRI, Onderstepoort, unpublished data, 1984) and C. obtusifolium185, 189 have been shown to be toxic. Dosing trials with sheep, cattle and horses revealed that clinical signs developed approximately 12–40 hours after they were dosed with about 5–14 g/kg of fresh or dried Cynanchum plants.Six toxic pregnane glycosides (cynafoside C-H) have been isolated from C. africanum plants collected near Mossel Bay and Swellendam (Figure 16).193 The lethal oral dose of the cynafosides for sheep is c.4–12 mg/kg.
Cynanchosis can occur at any time of the year. However, some farmers believe that the poisoning is commonest in winter, while others claim the toxicity of the plants is highest in summer, especially if wilted.48
In the early stage of intoxication, which lasts for half an hour to several hours, sheep may assume a wide-based stance, sometimes with the front legs crossed. They also tremble, stagger about, and fall with increasing frequency until recumbent. Cattle usually walk with the back arched, on stiff legs, taking short steps. The contraction of the leg muscles may be so great that they are forced to walk on tip-toe or even on the anterior surfaces of the front fetlocks.
In the next phase, lasting one to seven days, the animals lie in lateral recumbency, periodically undergoing tetanic seizures. During seizures (Figure 17) the head may be held between the rigidly extended legs (orthotonus) or thrown back (opisthotonus). The mouth is tightly closed, the lips are drawn back, the eyes are wide, and nystagmus is evident. The victims are hypersensitive and a slight tap on the head may precipitate violent seizures. Between seizures they may make ineffectual paddling motions with their legs, as observed in heartwater. Death is thought to result from respiratory arrest during seizures.
The spasmodic stage goes over into a period of protracted paralysis, often lasting a week or more. Animals that do not die in the spasmodic stage often recover. Some of the affected animals become bloated48, 92, 93, 185 (T.S. Kellerman and L.A.P. Anderson, VRI, Onderstepoort, unpublished data, 1984).
Erasmus61 reported hyperglycaemia and metabolic alkalosis even in subclinically affected animals or before signs appeared in overtly intoxicated ones.
Rigor mortis may set in almost immediately after death. No consistent macroscopical or microscopical pathological lesions have been described, but fragments of the plant in the rumen can be of assistance in making a diagnosis.
The paralytic stage of cynanchosis should not be confused with krimpsiekte, a nervous disorder of sheep and goats caused by the ingestion of Tylecodon spp., Cotyledon spp. and Kalanchoe spp. (Crassulaceae) containing cumulative neurotoxic cardiac glycosides.4, 61, 92 Poisoning with Cynanchum species is characterized by initial tetanic seizures, while krimpsiekte is primarily a paretic syndrome. The diseases that most closely resemble cynanchosis are tetanus, heartwater and poisoning with either Sarcostemma viminale or chlorinated hydrocarbon pesticides.
Sarcostemma viminale (L.) R. Br. (Apocynaceae)
Caustic bush, caustic creeper, melktou, spantoumelkbos
The clinical signs of poisoning resemble those of cynanchosis. Sarcostemma viminale is a leaf less, succulent climber that grows into or over trees, sometimes smothering them (Figure 18). Without support it forms a bush about 1 m in height. The frequently branched, pencil-thin, greyish-green stems contain a white non-irritant latex. The flowers are yellowish, star-shaped and scented (Figure 19). The fruits (Figure 20), resembling goat’s horns (about 80 mm long and 150 mm thick), are packed with white, plumed seeds.205
Melktou is widely distributed in southern Africa (Figure 21) and is particularly plentiful in the hot, drier parts.154, 178, 186
Doses of 10–17 g/kg of fresh S. viminale, collected at Enkeldoorn (Zimbabwe) and in Namaqualand, fatally poisoned sheep in 6 to 31 hours.154, 178 Terblanche and Van Straten200 found 2 g/kg of dried plant from Kuruman to be lethal for sheep, while 1 g/kg caused only clinical signs. Clinical signs appeared 4–8 hours after dosing and death occurred after 12–28 hours. Steyn,185 on the other hand, reported that sheep, dosed with 14 g/kg of dried plant from Uitenhage, could live for more than a week. The toxicity of S. viminale is so variable that it can sometimes be grazed39, 178 or dosed185 with impunity. The factors that govern its toxicity are not known.Three related pregnane glycosides, sarcovimiside A, B and C, have been isolated from S. viminale (Figure 22),214 which accounts for the similarity in the clinical signs induced by this plant and Cynanchum spp.
Ruminants, especially Angora goats, and horses are mostly affected during times of grazing shortage. When trees in which the plant grows are felled, animals often gain access to sufficient S. viminale to induce intoxication.
Affected animals are hypersensitive, exhibit muscular tremors/spasms of increasing intensity, and walk with an unsteady, stiff-legged, uncoordinated gait. Eventually, they lie on their sides, making ineffectual, galloping motions. Tonic spasms rack their bodies, during which time the head is drawn back or pulled down, the back is arched and the legs are extended, as in strychnine poisoning. Spasms can be elicited by external stimuli such as handling the animal or by tapping it on the head. A case has been described where an animal, suffering a seizure while being chased, leapt into the air with all four legs rigidly extended and fell.
The above signs may be accompanied by tachycardia, polypnoea, bloat and elevation of body temperature.154, 178, 185, 187, 189, 200 Death in the acute phase results from respiratory arrest during spasms or from bloat following ruminal stasis.200
More chronically intoxicated animals may become paralysed after the acute signs have subsided and can lie in lateral recumbency for a week or more.187, 189
In Australia, a similar poisoning is induced in cattle and sheep by S. australe.80, 81, 87
No noteworthy lesions have been reported.
Euphorbia mauritanica L. (Euphorbiaceae)
Yellow milkbush, melkbos
This is a relatively unimportant poisonous plant that seldom causes stock losses.
Euphorbia mauritanica is a bush approximately 1 m in height (Figure 23 a), the branches yellowish-green, pencil-thin and cylindrical. They arise from near the base of the stem and seldom form side branches. A white latex exudes where the plant is injured. Small, sessile, narrow, oblong leaves are formed at the tips of branches (Figure 23 b), but these soon die and fall off, leaving alternate leaf-scars. Yellowish-green flowers are arranged at the ends of young branches (Figure 23 c). The fruit is a capsule c.50 mm in diameter, divided into three compartments, each containing a mottled seed.201, 205
The species is common in the Western and Northern Cape Provinces, especially in the drier karoid areas such as Namaqualand. Euphorbia mauritanica extends also into Namibia, the Free State and KwaZulu-Natal (Figure 24).
Reports of its toxicity have been conflicting.184, 201 Remnants of E. mauritanica were identified in the rumen of cattle that had died of a nervous disorder near Kuruman in 1963. The majority of sheep dosed with milled, dried E. mauritanica from the toxic camp, died within c.12–92 hours. Clinical signs appeared 4–12 hours after dosing, and lasted for 3–5 days.201
The muscles of affected animals showed increased tone and continuous tremors. They were reluctant to stand or stood stiffly with the back arched and legs splayed. True tetanic spasms did not occur as the extended limbs and neck could be easily flexed manually, and spasms could not be induced by external stimuli. These clinical signs were accompanied by foaming at the mouth, salivation, ruminal atony or bloat, diarrhoea and elevated body temperatures.201
The pathological lesions, though fairly unspecific, included changes such as oedema, hyperaemia and emphysema of the lungs, signs of tympany and hyperaemia of the gastrointestinal tract.201
Since E. mauritanica poisoning somewhat resembles Sarcostemma viminale poisoning and since the plants have similar characteristics and distributions, the two conditions should be distinguished from each other.200, 201 In both instances, remnants of the plants may be recovered from the rumens of poisoned stock. When camps are examined for the plants, it must be borne in mind that, although S. viminale is usually a climber, it can also occur in bush form. The presence of leaves at the ends of branches or alternately arranged leaf-scars on the branches will distinguish E. mauritanica from S. viminale, which is leaf less. In the case of S. viminale the side branches arise opposite each other whereas in E. mauritanica branches come off randomly.201
The clinical signs caused by the two plants differ in that S. viminale induces strychnine-like tetanic spasms while in E. mauritanica, despite increased extensor tonus, the limbs can be flexed manually with comparative ease.200, 201
Diarrhoea is not a feature of S. viminale poisoning.201
Dipcadi glaucum (Ker-Gawl.) Bak. (Hyacinthaceae)
Wild onion, malkop-ui
Dipcadi glaucum is responsible for nervous signs and diarrhoea in sheep, cattle and goats. Abortions have been reported in small stock.189
The whitish bulbs, c.10–15 mm in diameter, are buried 50–100 mm under the ground. The greyish-green leaves, which are 150–400 mm long and about 30 mm wide, are arranged in a basal rosette (Figure 25). The inf lorescence is an erect raceme with greenish-white flowers (Figure 26 a). The black, flat seeds, c.3 mm in diameter, are contained in globose capsules (Figure 26 b). After good rains, innumerable wind-blown seeds germinate freely (Figure 27).205
Dipcadi glaucum prefers deep sandy soil and sunny spots between trees. The plant is particularly common in the Vryburg and Kuruman districts of the North-West and Northern Cape provinces and in the western parts of the Limpopo Province (Figure 28), becoming especially troublesome in camps denuded by drought (Figure 29). Immediately after early spring and summer rains, the leaves will appear above ground level and within five to six days the plants can reach a height of approximately 150 mm.
The toxicity of D. glaucum seems to vary according to locality and growth stage of the plant. The toxin(s) of D. glaucum has not yet been identified, but preliminary tests indicate that it is not a cardiac glycoside, the active principle of many genera of the Hyacinthaceae (L.A.P. Anderson, VRI, Onderstepoort, personal communication, 1983)
Dipcadi glaucum poisoning is more common in sheep than in either cattle or goats. In sheep the clinical signs include aimless wandering, pushing against objects for long periods and a severe diarrhoea. Some animals may also show dyspnoea, a weak and accelerated pulse, fever and abortion.183, 184
In October 1982, a herd of mature beef cattle near Dwaalboom in the Limpopo Province, was put out to graze in a camp that had been denuded of grass as a result of drought. Some weeks after the cattle had been introduced into the camp, drenching rains fell. Soon after the showers, D. glaucum bulbs sprouted in such numbers that the veld was tinged a green colour. Densities of up to 15 D. glaucum plants per square metre were recorded in some places.
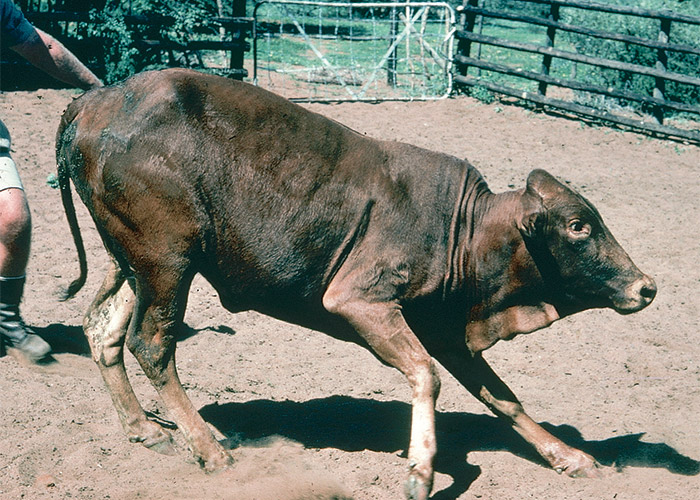
According to the farmer, the cattle became sick about eight days after the rainstorms. Acutely affected animals apparently walked about drunkenly, sometimes circling, often stumbling into trees and occasionally pushing aimlessly against objects. Typically, they walked with the fetlocks in f lexion, with the rump down and the hind legs tucked under the body, frequently knuckling over and brushing with their feet (Figures 30 and 31). They were apathetic, often lay down and had difficulty in standing up as a result of posterior paresis. Some cows lay in sternal recumbency with their hind legs disposed behind them in a peculiar, flexed fashion (Figure 32). Even in far-advanced cases the prostrate animals still ate and drank normally. Twenty-five out of the 200 were affected and, of these, 12 died.
The rather non-specific, pathological changes include general congestion and cyanosis, congestion and oedema of the lungs, epi- and endocardial haemorrhages and catarrhal enteritis.
Annual ryegrass toxicosis (ARGT)
ARGT is a highly fatal neurotoxicosis of cattle, sheep and horses, resulting from the ingestion of ryegrass seedheads containing nematode (Anguina sp.) galls infected by a bacterium, Rathayibacter toxicus.114, 171 The bacterium produces corynetoxins related to the tunicaminyluracil antibiotics. In South Africa, the disease is known to occur only in the Winter Rainfall Area of the Western Cape Province.171
According to Australian reports the corynetoxin poisoning is not limited to stock grazing on annual ryegrass pastures.29, 33, 37, 59 Pigs fed spoilt wheat29 and cattle grazing on pastures dominated either by Agrostis avenacea33, 37, 59 or Polypogon monspeliensis 59 have developed signs consistent with ARGT. Tunicaminyluracil toxins were identified in both the toxic wheat and material from the incriminated pastures.29, 33, 46, 59
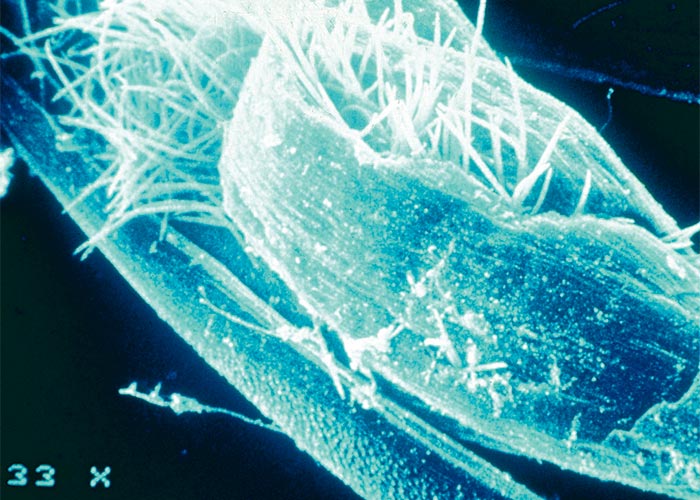
The life cycle of the nematode (Anguina agrostis) has been described by Australian workers, who also elucidated the aetiology of the disease.21, 68, 114, 158, 194, 196 Resistant second stage larvae of the nematode remain dormant in the galls on the ground over the hot, dry summers. After the first autumn rains they emerge from the galls (Figure 33) and migrate onto annual ryegrass (Lolium spp.) seedlings, where they lodge at the growing point. The larvae are carried up passively by the plant as it grows, to a position which enables them to penetrate the floret primordia. Instead of seeds, galls are formed in which the adult nematodes lay eggs, that soon hatch into larvae, ready to begin a new cycle.
Migrating larvae often carry with them on their cuticles a bacterium, Rathayibacter toxicus, previously known as Corynebacterium rathayi. Should the inoculum of R. toxicus be sufficiently large, the rapidly multiplying bacteria can take over the nematode gall, forming a yellow slime in the process. Evidence of slime on flowerheads and leaves serves as a warning that a pasture may be toxic,21, 158 but this is not often seen in South Africa.
The toxicity of annual ryegrass pastures is exclusively associated with the bacterial galls. The nematode galls, by contrast are harmless.114, 196
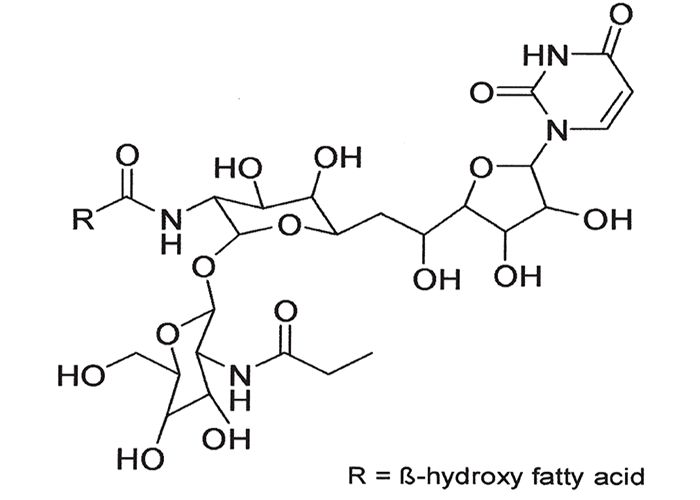
The cumulative active principles, corynetoxins, isolated from bacterial galls, are closely related to the highly toxic, tunicaminyluracil antibiotics produced by certain Streptomyces species.58, 65, 118 Both are composed of uracil, tunicamine, N-acetylglucosamine and a fatty acid. Corynetoxins differ from tunicamycins, however, in having longer fatty acid chains and, sometimes, in having other methyl side chains (Figure 34). They exert their cumulative toxic effect by the inhibition of a specific step in the synthesis of certain sugar-linked proteins.13, 99, 118, 215
It is thought that the toxin may be produced by either the bacterium or the grass in reaction to bacterial infection.196 Cultures of R. toxicus have so far been proven to be non-toxic,196 but toxicity has been induced by the infection of a cell culture of endosperm from L. multiflorum with R. toxicus.195
Schneider171 reproduced the disease experimentally in sheep in South Africa by separately feeding them hay, chaff, which contained numerous seedheads, and seed from a pasture of annual ryegrass (hybrids of L. temulentum) on which sheep had developed nervous signs (Figure 35).
As little as 2 kg of the seed was sufficient to induce fatal nervous intoxication within six days. Galls filled with numerous second stage larvae of an Anguina sp., as well as bacterial galls containing a Corynebacterium sp., were identified in the toxic material.
The toxic seed originated in a vetch (Vicia villosa)/oat field overgrown by Lolium hybrids (possibly of L. temulentum) near Caledon. The sheep began dying early in December 1979, when c.95 out of 120 lambs succumbed four to nine days after being introduced into the toxic camp. Later, a further 226 mortalities were recorded and 200 ewes aborted after eating hay from the toxic pasture. The sheep became sick 8–16 days after feeding commenced, and mortality ceased four to six days after removal of the hay. Virtually all the affected sheep died.171
Three outbreaks of ARGT, two near Bredasdorp and the third near Prince Albert Hamlet, were also reported during October and November 1980, in cattle grazing on seeding pastures of L. multiflorum (Italian ryegrass) and L. rigidum (Wimmera ryegrass) hybrids. No age, breed, or sex predisposition could be found and mortality was uniformly high.171
Since the corynetoxins can survive desiccation and are relatively heat stable, many outbreaks can be attributed to the ingestion of toxic hay.
The clinical signs14, 129, 171 in less severely affected animals may appear only when the animals are disturbed. These signs typically include excitability, muscular twitching, locomotory disturbance such as unsteadiness, a saw-horse and/or high-stepping gait and convulsions (Figure 36). During the tonic/clonic seizures, which initially last about one to three minutes, the animals tend to lie on their sides in opisthotonus, with their legs rigidly extended. Eventually they may become permanently recumbent and, apart from convulsions, they may twitch, tremble, show nystagmus and froth at the mouth.129, 171 Clinical signs usually appear about three days after stock have been introduced onto a toxic pasture and death ensues c.4–24 hours after the onset of signs. Mortalities may continue for four or five days after removal from the camp (D.J. Schneider, personal communication, 1992).
Among the macroscopical changes are rapid rigor mortis, oedema of the lungs, endo- and epicardial haemorrhages, hepatosis and icterus16, 17, 129, 171
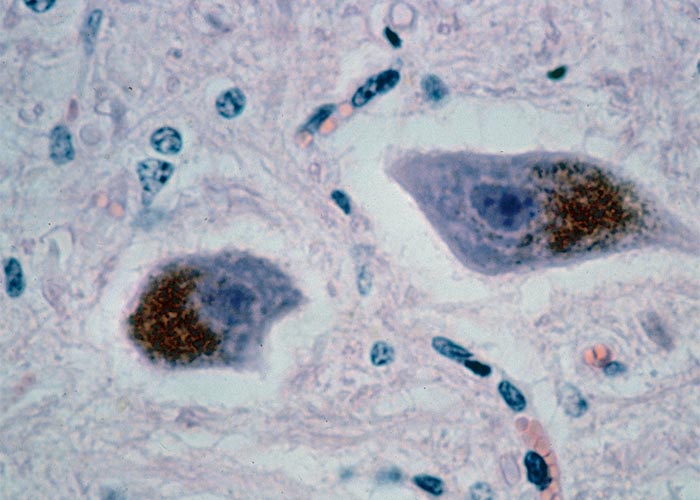
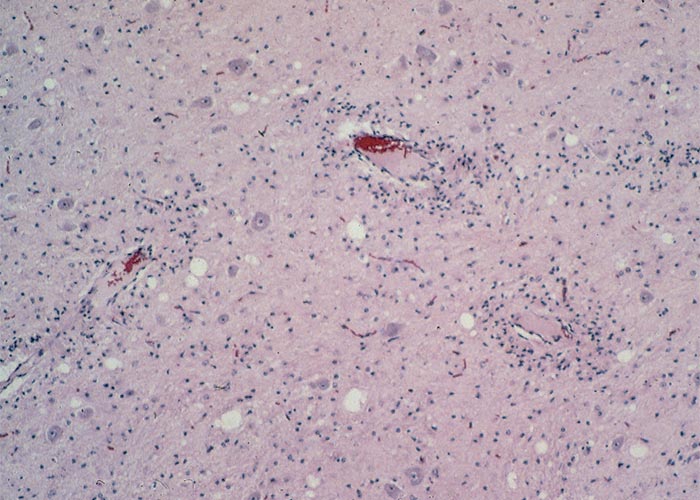
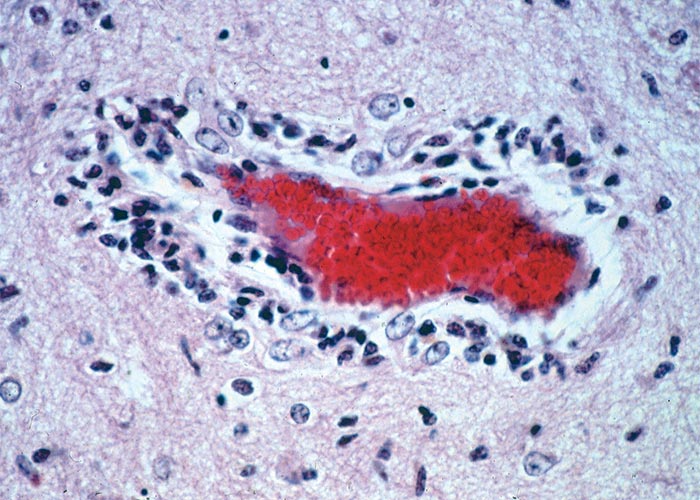
Corynetoxins apparently exert their main effect on the endothelial cells of blood vessels, notably of the central nervous system leading to capillary obstruction and inadequate local perfusion. Permeability is increased as a result of this damage and perivascular oedema is common and occasionally small haemorrhages are present in the brain and meninges. In the longer surviving animals, focal or diffuse degenerative changes, sometimes accompanied by a glial or macrophage response, are evident in the neuropil15, 18, 66, 67
Berry et al.16 reported elevations of liver-specific enzymes and bilirubin in experimentally induced ovine ARGT poisoning. Newer work, however, has shown inhibition of the GlcNAc-1-phosphate transferase to be a more sensitive marker for ARGT.182 Affected livers are pale and often enlarged as a result of hydropic and fatty degeneration of the hepatocytes. Isolated necrotic hepatocytes are seen in some lobules. After the onset of nervous signs, serum creatinine kinase activity rises, probably as a result of the abnormal muscular contractions during convulsions.16, 17, 129, 171
Since the nematode is relatively safe from nematocides in the galls,194 ARGT is controlled by eradicating the ryegrass or by preventing the ingestion of the seedheads by stock. Little is known about the control of the disease in South Africa. Eradication of ryegrass on a large scale would be difficult in practice because annual ryegrass is a common weed on stubble or fallow lands in the Winter Rainfall Area. Better results would probably be obtained by heavily grazing infested pastures before the seedheads mature, or by either applying low levels of herbicides to prevent seeding or mowing the pasture at this stage. The most practical method of control is probably to burn the toxic pasture and then to crop it in the following year, using selective herbicides to control the ryegrass. As soon as an outbreak occurs, livestock should be moved to a clean camp or a camp that has been artificially freed of seeding ryegrass.
There is no specific antidote for ARGT. Some success was achieved with the sedative chlordiazepoxide in Australian pen trials.164 The survival rate of treated sheep under field conditions was not as good and those that survived needed supportive treatment.149 Magnesium sulphate, administered parenterally, prevented ARGT convulsions for up to 12 hours in Australia.163 In South Africa, magnesium sulphate, injected i/m as a 50% aqueous solution at 0,2 g/kg twice daily for 2–5 days or until the clinical signs abate, has given promising results (D.J. Schneider, RVL, Stellenbosch, personal communication, 1993). A variety of sedatives and tranquillizers have been tried without signal effect163, 171 (D.J. Schneider, Regional Veterinary Laboratory, Stellenbosch, personal communication, 1992).
ARGT is diagnosed from the typical nervous signs, the lack of significant pathological lesions and the presence of bacterial galls in the seedheads. One method is to beat a bag of hay from a suspected toxic pasture with a stick to separate the seeds. The fine material at the bottom of the bag is then passed through a series of sieves to remove the larger particles and dust. Stereomicroscopic examination of the siftings reveals the galls, distinguished from healthy seeds by their pointed apices and generally shorter length. Nematode galls are black, while bacterial galls are tinted yellow (Figure 37).132 The former may also be soaked in water for a few hours and examined under a light microscope for emerging larvae, while bacterial galls may be squashed to reveal bacteria.
Diagnosis is confirmed by feeding bacterial galls to mice or sheep and demonstrating the toxins in foodstuffs of poisoned stock.
Annual rye grass toxicosis must be distinguished from other nervous disorders of ruminants on grass pastures, such as Phalaris staggers, perennial ryegrass (Lolium perenne L.) staggers,141 Bermuda grass tremors in the United States of America,52 kweek tremors and Paspalum- and Melica decumbens staggers in southern Africa. These last four conditions lack notable central nervous lesions and are not highly fatal.
Note that annual ryegrass infected with Claviceps purpurea has been implicated in serious outbreaks of ergotism (summer syndrome) of cattle in South Africa (see Cardiovascular system).
Melica decumbens Thunb. (Poaceae)
Staggers grass, dronkgras
Melica decumbens (Figure 38) causes paresis and ataxia in cattle (Figure 39) and occasionally in sheep in the eastern Karoo and drier parts of the former central and eastern Cape Province (Figure 40). It is a tufted grass, c.300 mm in height, with thin culms and dark narrow leaves. The seedheads are shiny and feathery (Figure 38) with distinctive dark glumes.78, 189, 216
The grass becomes troublesome in late winter and early spring following good winter rains. Fresh, fast-growing grass is more palatable and toxic than the mature grass, which is hard and coarse. Large quantities must be consumed for poisoning to occur.
Although the plant has long been known to be toxic,189, 216 information on this poisoning is scant, probably because outbreaks are sporadic and mortality is low.
Under field conditions, cattle 5–18 months of age are mainly affected. Poisoning has been experimentally produced in a three-month-old Jersey calf of c.55 kg that received c.20–30 kg of grass in seed ad libitum for 14 days (G.F. Bath, Regional Veterinary Laboratory, Allerton, Pietermaritzburg, personal communication, 1984). The grass, collected in December, was kept in a refrigerator and fed to the calf in February. The first signs were slight swaying, unsteadiness and a high-stepping gait (Figure 39). The calf lay down regularly and was reluctant to rise. With prodding it could only rise with difficulty, and walked on tip-toe. The clinical signs became progressively worse after a week of continued feeding. The calf walked and ran with a goose-stepping gait and had difficulty in changing direction. It sometimes moved sideways in a forward direction and was often found lying down. When chased, the animal tripped over its front feet, fell down and was unable to rise. It also showed general muscle tremor, especially in the muscles of the limbs. Throughout the course of the disease the habitus and appetite of the animal remained good.
No lesions were observed in the brain or other organs of the calf. With good care affected animals almost invariably recover. Mortality is extremely rare.
The nervous signs, lack of lesions and low mortality of M. decumbens poisoning are in keeping with Paspalum staggers, perennial ryegrass staggers or kweek tremors. Furthermore, endophyte(s) have been identified in South African specimens of M. decumbens 221, 222 (Margaret E. di Menna & C.O. Miles, Ruakura Agricultural Centre, Hamilton, New Zealand, unpublished data, 1996) lending credence to the belief that the active principle of dronkgras is probably an indole-diterpenoid tremorgen. In an enigmatic historical footnote, Marloth in 1913128 speculated that M. decumbens ‘is said to possess toxic effects. Possibly this is due to an infection by a fungus, as in the European Lolium temulentum (drabok, darnel)’.
Kweek tremors
Cynodon dactylon (L) Pers. (Poaceae) Quick grass, kweek
This relatively common non-fatal tremorgenic condition of cattle and sheep is associated with the grazing of Cynodon dactylon (kweek). Kweek tremors were first reported in July 1977, when calves developed neurological signs on a small predominantly kweek grass pasture near Tweespruit, in the Free State Province (Figure 41). Sheep that had been put on the pasture in place of the affected calves promptly developed similar clinical signs. During the following winter and sporadically in subsequent cold seasons, outbreaks occurred, mostly in cattle, in the Parys/Potchefstroom/Ventersdorp areas of the Highveld.
The characteristic clinical signs of the disease are pronounced muscular tremors, ataxia and paresis. In acute cases, almost all the voluntary muscles may tremble, even when the animal is at rest. Typically affected animals walk with stiff hind legs, take short steps, often lift the legs high, frequently stumble and sometimes fall (Figure 42). In a small proportion of cases, the paresis may progress to recumbency in a sternal position. Even though the paresis/ paralysis may be advanced, animals still eat, drink and ruminate normally. The condition is seldom if ever fatal. Clinical signs may last for several weeks, but most cases recover within a day or two.
In some instances the entire herd may be affected, but the morbidity usually varies from 5–35%, with an average of c.10%.
The condition may be confused with Paspalum staggers (Claviceps paspali poisoning), Melica decumbens poisoning and diplodiosis (Stenocarpella maydis poisoning). The clinical signs also bear some resemblance to those of maroek poisoning, a highly fatal, tremorgenic mycotoxicosis caused by Aspergillus clavatus.
The only common factor in the various outbreaks is that they occurred soon after stock had been moved on to mature rested pastures where C. dactylon was abundant. The possible involvement of this grass in the disease was highlighted by the occurrence of outbreaks on pastures consisting almost exclusively of C. dactylon. Moreover, on mixed pastures, C. dactylon was sometimes seen to be selectively grazed before the neurological signs appeared.
The mycological examinations carried out on the pastures revealed that some of the seedheads were infested by Cerebella andropogonis and a Claviceps sp. (Figure 43). The former is a non-parasitic fungus that grows saprophytically on the sugary honeydew secreted by the conidial stage of Claviceps spp. The presence of Cerebella prevents the formation of ergots and is indicative of Claviceps infection. It should be noted though that Claviceps cynodontis Langdon has been identified on kweek in South Africa (E. J. van der Linde, Biosystematics division: Mycology, ARC-Plant Protection Research Institute, personal communication, 1998).
American workers some years ago isolated toxic Claviceps, as well as the indicator C. andropogonis from C. dactylon incriminated in Bermuda grass tremors. The latter disease is a neurological disorder of bovines in the southern states of the USA that is, as far as can be ascertained, indistinguishable from kweek tremors.
Attempts to establish whether an endophyte was involved in the aetiology of the kweek tremors failed because the organism could not be demonstrated in C. dactylon from toxic camps (Margaret E. di Menna & C.O. Miles, Ruakura Agricultural Centre, Hamilton, New Zealand, unpublished data, 1996). Kweek tremors could also not be induced by feeding small quantities of ergot-infected seedheads to sheep and cattle (T.S. Kellerman, OVI, unpublished data, 1978). Nevertheless, despite these equivocal findings, the weight of the evidence points at indole diterpenoid tremorgens (paspalitrems) from the ergots being responsible for the disease.
Paspalum staggers
Claviceps paspali Stev. & Hall (Fungi: Ascomycetes)
Paspalum staggers is a rarely fatal nervous disorder particularly of cattle grazing on the seedheads of Paspalum dilatatum (Dallis grass) and P. distichum (couch Paspalum) infected with ergots of Claviceps paspali.24, 60, 119, 121, 134, 137, 161 Paspalum dilatatum is a moisture loving grass, mostly confined to vleis and marshy areas or highly irrigated pastures (Figure 44). It is a tufted perennial 0,3–1,5 m in height with inf lorescence of three to five racemes (30–120 mm long) provided with spikelets (3–4 mm long) arranged in pairs (Figure 45).42, 43 P. distichum has very much the same characteristics except that the inflorescence usually has only two racemes.
The fungus colonizes the unfertilized ovaries of Paspalum spp. to form roughly spherical, hard, brownish bodies, known as sclerotia (Figure 45). These sclerotia or ergots constitute a resting form of the fungus in which it overwinters. In spring or summer, when temperatures and humidity allow, the sclerotia on the ground germinate to release ascospores which are borne by insects to infect new flowerheads. In its turn the mycelium (sphacelial stage), growing in the infected f loret, produces conidia, borne in honeydew, a sweet sticky secretion of the fungus. Conidia are transmitted from plant to plant by various agencies, such as by insects attracted by the honeydew, by grazing animals, and by infected and healthy seedheads brushing against each other in the wind.119, 121, 134, 137, 161, 181
Approximately 1 g/kg/day121 ergots over four days induced severe staggers in cattle,120 and 2 g/kg Paspalum seeds, the majority of which were infected, caused mild signs in a steer.60 While the ergots contain alkaloids (mainly D-lysergic acid, α-hydroxyethylamide),20 their neurotoxic effects are attributed to tremorgenic indole derivatives of tryptophan and a diterpene, geranylgeraniol.47, 120, 121, 122 The mechanism of action of the tremorgenic mycotoxins has not been fully elucidated. However, four fungal tremorgens (paspalinine, paxilline, aflatrem and verruculogen) have been shown to inhibit the GABAA-receptor by binding at or near its chloride channel, thus preventing inf lux of chloride.75
Claviceps paspali is a cosmopolitan fungus known to cause staggers in New Zealand, Australia, USA, Europe and South Africa.120 Four outbreaks of staggers involving P. dilatatum60, 134, 137, 161, 189 and one with P. distichum24 have been described in southern Africa. This syndrome occurs mostly in cattle, occasionally in sheep, and very rarely in horses.60, 121, 134, 137, 161 Clinical signs, which usually appear two to seven days after stock have been exposed to toxic pastures, include hypersensitivity, tremors and incoordination. Severely affected animals may become recumbent, sometimes lying on their sides making heartwater-like paddling motions. Appetite remains good and animals almost invariably recover after removal from toxic pastures. A notable feature of the disease is that the signs become more pronounced with exercise.
No significant pathological changes have been described.
There is no specific treatment for Paspalum staggers. The condition can be avoided either by not grazing ergotized pastures or by heavily grazing Paspalum grass in spring and summer, thereby preventing seedhead-formation. Alternatively, ergotized seedheads can be mechanically removed after high-mowing the toxic pasture.161
Claviceps africana (= C. sorghi) is a widely occurring pathogen of sorghum70 in South Africa. Although a minor outbreak of tremors in cattle grazing on infected sorghum has come to our attention, the fungus is not regarded as being of veterinary importance. Large quantities of seedheads from the toxic pasture induced only mild transient (lasting an hour or so) tremors in a steer at Onderstepoort Veterinary Institute (T.W. Naudé & T.S. Kellerman, OVI, unpublished data, 1991).
Perennial ryegrass staggers
Neotyphodium lolii (= Acremonium lolii)
Perennial ryegrass staggers (PRS) potentially occurs wherever in the world Lolium perenne is grown, but is particularly common in New Zealand and Australia.41 A presumptive diagnosis of perennial ryegrass staggers (PRS) has been made in cattle grazing on Lolium perenne grown from imported New Zealand seed in KwaZulu-Natal. Microscopical examination of leaf specimens (stained with Rose Bengal) from the grazed-down pasture revealed the presence of an endophyte. The clinical signs, such as severe incoordination, a high-stepping gait, loss of balance, falling, tremors, head nodding or weaving, could be exacerbated/ elicited by exercise. When not disturbed, almost all the signs, save for some head-weaving, disappeared. No mortalities were recorded. About 10% of 342 weaners were affected (J. Kitching & T.S. Kellerman, Allerton Regional Laboratory, unpublished data, 1991). A second outbreak, involving horses and cattle, was recently clinically diagnosed in the Western Cape Province (A. van Halderen, Regional Veterinary Laboratory, Stellenbosch, personal communication, 1997).
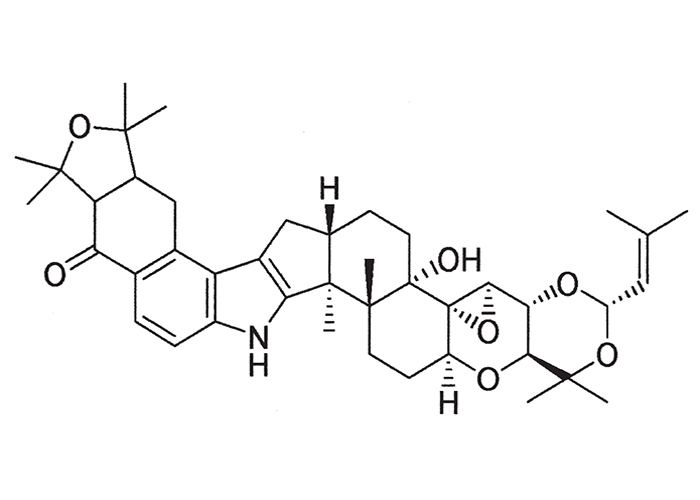
Perennial ryegrass staggers is associated with the ingestion of Lolium perenne infected by an endophytic fungus, Neotyphodium (= Acremonium) lolii.69 As the fungus is most prevalent in the leaf sheaths at the base of the tuft, stock grazing on short sward are most at risk.53, 69, 145 The endophyte is apparently disseminated only through the ryegrass seeds, hence infection does not spread directly from one plant to another on the same pasture.69, 115, 145 Seed may be sterilized of the endophyte by application of fungicides, heat treatment or storage at room temperature for a protracted period, e.g. two years.115, 116, 145 The active principles are believed to be indole-diterpene tremorgens of which lolitrem B is the most important (Figure 46).72, 73, 74 The endophyte is also responsible for improving the vigour of the ryegrass by synthesizing an insect-deterring alkaloid, peramine, which protects the tillers from the Argentine stem weevil.169
Lupine poisoning
Lupinus angustifolius L. (Fabaceae)
Lupines are spreading, erect, annual legumes growing about 600 mm high, with compound leaves comprising 7–11 finger-like leaf lets. Depending on the variety, the f lowers can be yellow, white or blue. The flat pods are slightly pubescent and contain four to five seeds.76 In southern Africa lupines are found only as cultivated plants.
Three distinct disease entities have been associated with lupines, namely, lupinosis, which is a mycotoxicosis caused by the fungus Phomopsis leptostromiformis and is responsible for liver damage in animals (see Liver); lupine (alkaloidal) poisoning, caused by the alkaloids in some of the bitter lupines and giving rise to nervous signs in animals and a crooked calf syndrome characterized by arthrogryposis, torticollis and scoliosis, in cows ingesting Lupinus sericeus, especially between 40–70 days of gestation.103, 179
Lupine poisoning has purportedly occurred in pigs, cattle and sheep in the Swartland of the Western Cape Province. There are also reports of outbreaks of the disease in Australia and in the western states of America.76
Impaction of the rumen with seeds and stasis of the large intestine has been reported in animals that overeat lupines (D.J. Schneider, Regional Veterinary Laboratory, Stellenbosch, personal communication, 1984).
Lupinus angustifolius and L. digitatus are most commonly associated with nervous poisoning. The green plants are usually safe to feed, but the dried, mature plants, especially the seeds, are highly toxic. The alkaloids, such as d-lupanine, sparteine, lupinine, sparthalatine and hydroxylupinine,76 are concentrated mostly in the seeds, but significant amounts of alkaloids are present in the pods, less so in the leaves and even less in the stalks and stems. Seeds retained by the standing plant may be eaten readily by sheep and cattle while seeds on the ground are mostly inaccessible.
Clinical signs usually appear within one to two hours, and often only after exercise, but can be delayed for as long as 24 hours after the lupine seeds have been eaten. As a result of stimulation followed by paralysis of the central nervous system, the affected animals show violent muscle tremors, convulsions, confused dashing about, staggering, dyspnoea and, ultimately, paralysis of especially the respiratory and vasomotor centres. Death is by asphyxia and occurs mostly within the first three hours.
No lesions have been described. Treatment with acetic acid is said to be effective.
Melia azedarach L. (Meliaceae)
Syringa berry tree, seringboom
Melia azedarach is an attractive tree with heavily scented mauve flowers (Figure 47). Originally from India, it has become naturalized in many parts of southern Africa.90 The ripe drupes are responsible for nervous signs, respiratory distress and diarrhoea in animals and humans.181, 189, 190
It is a popular shade tree with dense foliage which grows vigorously even with little water. The drupes are spread by birds and water, with the result that trees are often found along river beds. The flesh of the ripe berries is most toxic, but the green berries as well as the leaves and flowers also contain some toxin.189, 190 Poisoning rarely occurs in animals in South Africa. Although pigs and poultry are particularly susceptible, outbreaks have also been reported in sheep and cattle. The berries have been associated with vomition, gastroenteritis, diarrhoea, dyspnoea, muscle tremors and convulsions in children.178, 188-190
Affected pigs, sheep and cattle show restlessness, muscle tremors (especially of the large muscle groups in the hindquarters), dyspnoea, cyanosis and ultimately paralysis, constipation and death after eating relatively large quantities of berries. The course of intoxication is short. At necropsy, cyanosis, congestion and oedema of the lungs and sometimes also a catarrhal to haemorrhagic enteritis are encountered. Large amounts of berries and their undigested characteristically fluted, woody pips are usually present in the stomach or rumen.189, 190
Four toxic tetranortriterpenes, meliatoxins A1, A2, B1 and B2, have been isolated from the fruit of M. azedarach L. var. australasica. While responsible for the acute nervous signs and death, they do not cause all the signs of M. azedarach poisoning. Some trees lack these toxins.152
No microscopical lesions have been recorded.
Datura spp. (Solanaceae)
D. ferox. L.
D. stramonium L.
Thorn apple, moon flower, stinkblaar, olieboom
Stinkblaar, a cosmopolitan pioneer, is a common annual weed in southern Africa (Figure 48). It grows to a height of c.1,5 m and is easily recognized by the characteristic 200 mm long, ovate, sub-lobed, irregularly dentate leaves, large funnel-shaped whitish flowers (Figure 48) and distinctive hard, spiny fruits (Figure 49). The small (c.3 mm long) kidney-shaped brown seeds are finely pitted.90
D. ferox differs only from D. stramonium in having larger (up to 30 mm) and more sparsely distributed spines on the fruit capsule (Figure 50).90
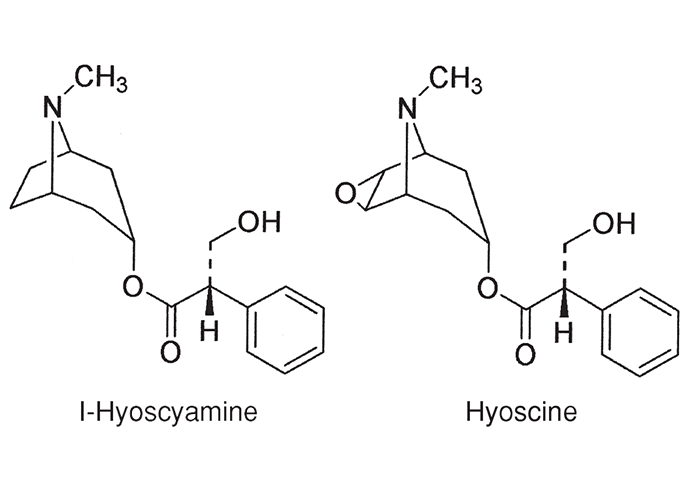
The plant contains the parasympatholytic alkaloids, atropine, hyoscine and hyoscyamine (Figure 51), all of which exert mainly an antimuscarinic effect. High doses block transmission of autonomic impulses at ganglia and neuromuscular junctions.2, 189
Datura species are not regarded as being important toxic plants in South Africa. Horses are nevertheless extremely susceptible to this poisoning and several outbreaks of impaction colic induced by the plant have come to our attention. Among these, two involved horses fed respectively on maize screenings (H.E. van de Pypekamp, State Veterinarian, Rustenburg, personal communication, 1985) and a sunf lower-based feed supplement heavily contaminated by Datura seeds172. In yet another incident, horses were poisoned by hay contaminated with Datura plant material (T.W. Naudé, R. Gerber, R.J. Smith and C.J Botha, Faculty of Veterinary Science, University of Pretoria, unpublished data, 1999). An outbreak of Datura poisoning also occurred in cattle kept in a large camp bare of vegetation save for a large heap of dry Datura plants (T.S. Kellerman and T.W. Naudé, VRI, Onderstepoort, personal observations, 1985). Poisoning of ostrich poults is apparently more common.189
The clinical signs include mydriasis and cycloplegia, dryness of the mouth, colic, tremors, convulsions, respiratory paralysis and coma.2 Excessive doses of atropine may cause mania and excitement in animals.2, 189 In horses, gastric dilatation followed by rupture and unresponsive paralytic ileus have been described.172
Livestock are generally more refractory to atropine poisoning than humans. Naudé (VRI, Onderstepoort, unpublished data, 1969) found that at least 50 mg/kg atropine per os was necessary to induce slight signs of intoxication in sheep. A sheep of c.30 kg survived the intravenous administration of 1 g atropine sulphate after exhibiting only transient signs of intoxication. Pigs and cattle were mildly affected by 5 mg/kg and horses by 3–5 mg/kg orally. Datura seeds containing 0,02% total alkaloid were dosed at the rate of 50 g/kg (a total dose of c.1,75 kg) per ruminal fistula to sheep without ill effect and pigs ate a ration containing 5 g/kg with impunity. Eight-week-old chickens tolerated a ration contaminated by 130 seeds/400 g or the equivalent of 0,12 g seeds/day for a week. If c.5 mg/kg atropine per os induces mild signs in the most susceptible species, then c.25 g/kg of the Datura seeds would have to be ingested to have a similar effect. This amounts to c.1 850 seeds/kg live mass.
Humans, unlike livestock, are highly susceptible to atropine poisoning, the therapeutic dose in humans being 1 mg for an adult. The maximum permissible contamination of products for human consumption, therefore, is one Datura seed/10 kg maize kernels, five Datura seeds/400 g soya beans and three Datura seeds/400 g shelled ground nuts. In the light of the pilot experiments with atropine and Datura seeds and the paucity of confirmed outbreaks of Datura poisoning in the field, it is improbable that all but the most grossly contaminated cereals would poison livestock. As a precaution, contaminated feed should be either diluted with wholesome material or tested for toxicity in a few inferior animals before being fed on a large scale. Poisoning by contaminated grain can occur in humans.189 Datura plants often grow on lands where their ripe seeds are mechanically harvested with grain from which they cannot easily be separated.
Neostigmine and related acetylcholinesterase inhibitors are antidotal.
Pteridium aquilinum (L.) Kühn (Dennstaediaceae)
Bracken fern, adelaarsvaring
Pteridium aquilinum (Figure 52) has a world-wide distribution and grows over large parts of the high rainfall, eastern mountainous areas of southern Africa. Neurointoxication with this mostly unpalatable fern nevertheless rarely occurs in livestock.
The toxicity varies with different parts of the plant and the stage of growth. Fresh and dried fronds, rhizomes, and to a lesser extent stems, are toxic.6 Large quantities of the plant must be consumed over a long period for poisoning to occur. The toxic principles are thiaminase I and a radiomimetic compound which induces aplastic anaemia (see Haemopoietic system) in cattle.204 In South Africa, P. aquilinum and Equisetum ramosissimum have been shown to have more thiaminase activity and lower thiamine content than Malva parviflora, Pennisetum clandestinum and Medicago sativa.131 Neoplasia of the urinary bladder (see Urogenital system) and gastrointestinal tract has also been reported in cattle exposed to small doses of the plant in other parts of the world.153
Neurointoxication of horses occurs most frequently in stabled animals receiving fodder contaminated with bracken. Clinical signs often develop 3–6 weeks after continual exposure to the plant and is characterized by drowsiness, dyspnoea, unsteady gait, pronounced staggers, tremors and an awkward stance (often with the forelegs crossed-over or the feet planted wide apart) and arching of the back. As the disease progresses, the staggers become more pronounced until the animal finally becomes recumbent, showing convulsive seizures, opisthotonus and somnolence.6, 8, 62, 63, 64, 189
Noteworthy pathological changes have not been described with bracken poisoning in horses.
Prognosis is good if the horses are attended to early in the course of the disease, but grave if the animals are recumbent and undergoing convulsions. Apart from symptomatic treatment, the horses should be given approximately 100 mg of thiamine parenterally per day and plenty of bran mash.63
In Australia, sheep that consumed P. aquilinum with a high thiaminase I activity developed polioencephalomalacia.6 Also in Australia, ‘bright blindness’, a progressive retinal degeneration, has been induced in sheep with P. aquilinum.9, 219
Apart from P. aquilinum, two other plants in southern Africa, Equisetum ramosissimum (horse-tail, drilgras or perdestert) (Figure 53) and supposedly Malva parviflora (mallow, kiesieblaar) (Figure 54), contain thiaminases.181, 189 The latter plant so rarely causes poisoning that it is only of academic interest.
Nicotiana glauca R.A. Grah. (Solanaceae)
Wild tobacco, wildetabak
Nicotiana glauca (Figure 55), though widely occurring and highly toxic, seldom causes poisoning of stock in South Africa. The plant, a cosmopolitan pioneer, often found on disturbed soil along river banks, has blue-green leaves and yellow tubular flowers. The toxic principle, anabasine, an alkaloid closely related to nicotine, is responsible for signs of acute toxicity and teratology in sheep, cattle and pigs. Toxic signs include salivation, irregular gait, wobbling while walking or standing, tremors, convulsions and dyspnoea.
Death is thought to be due to respiratory paralysis. Teratogenic defects, such as fixed excessive carpal f lexure, lordosis, cleft palate and deformed head, have been induced in lambs of sheep that received the plant between 30 and 60 days of gestation.102, 189
Tobacco residues cause identical acute signs caused by the nicotine content. Nicotine exerts its effect at all nicotinic receptor sites, namely, the ganglia of the autonomic nervous system (resulting in mixed autonomic response), muscle end plates (resulting in tremors) and the CNS.
Ficus ingens (Miq.) Miq. var. ingens (Moraceae)
Red leaved fig, rooiblaarrotsvy
Ficus salicifolia Vahl (= F. cordata subsp. salicifolia)
Wonderboom fig tree, wonderboomvy
Only these two, of the 25 indigenous species of wild figs, have been implicated in stock poisoning. They cause a rare nervous disorder of cattle during times of drought in the bushveld of the North-West and Limpopo provinces of South Africa.
The red-leafed fig (Figure 56) usually occurs as a low-growing shrub on rock faces in the Limpopo Province. It is deciduous and the ‘fruits’ (syconia) of 9–12 cm in diameter are pink when ripe.
The wonderboom fig tree, an evergreen reaching a height of 9 m on deep loamy soil, may assume a shrub form when growing on hill slopes and rocky outcrops.
The clinical signs of this highly fatal condition include hyperaesthesia, ataxia, tremors and ineffectual paddling motions while lying lateral in recumbency.142
Light microscopical examination of the central nervous system reveals lesions such as oedema and focal demyelination of localized areas of the brain and spinal cord. The changes in the liver range from mild degeneration to focal disseminated necrosis of hepatocytes.142
Nierembergia linariifolia Graham var. glabriuscula (Dunal) Coccucci & Hunziker (Solanaceae)
(= N. hippomanica var. violacea) Nierembergia, purple robe
Nierembergia linariifolia (Figure 57) has been incriminated in field outbreaks of neurotoxicity in calves in the Free State Province.25 This perennial, diffusely branched herb forms a rounded bush up to 0,2 m in height and spread. The simple, linear leaves are arranged alternately. Dark, bluish-purple or violet, cup-shaped flowers are borne in spring and summer. Nierembergia linariifolia is a garden escapee which has become naturalized in the Free State and Eastern Cape provinces.25
Nervous signs such as chewing or licking, protrusion of the tongue, dysphagia, head nodding, rotation of the eye ball, circling, stiffness, hypermetria, ataxia, falling, paresis and lateral recumbency with leg paddling and opisthotonus were observed during natural outbreaks and experimental reproduction of the disease. Salivation, dehydration and cardiac irregularities completed the clinical picture.25
Although no meaningful macroscopical lesions were present, histologically, mild cerebral oedema of the white matter in periventricular areas was evident.25
Neurological disorders with distinct pathological lesions
Solanum tettense Klotzsch var. renschii (Vatke) A.E.Gonç. (Solanaceae)
(= Solanum kwebense) Rooibessie, bitterappel
Solanum tettense is the cause of maldronksiekte (literally translated as mad-drunk-disease) in cattle, a disease characterized by epileptiform seizures and signs of cerebellar dysfunction.156, 208
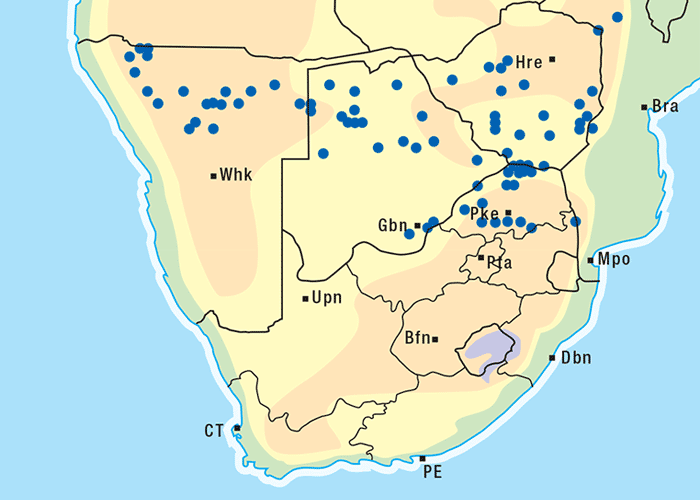
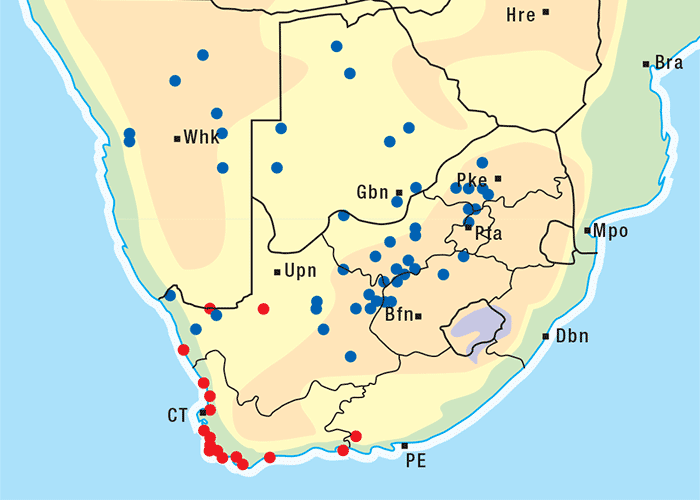
The plant is an erect, somewhat woody, laxly branched shrub, up to 2 m in height (Figure 58). The greyish-green leaves are covered with minute hairs. The light blue predominantly 4-merous flowers and orange-red to scarlet berries (Figures 59 and 60) are borne more or less simultaneously from about October to May. The plant can be found on flats or hill slopes and on sandy, loamy or stony soil. Although S. tettense occurs in Namibia, Botswana and South Africa, the disease has been reported only in the northern and north-western parts of the Limpopo Province, particularly in the vicinity of the conf luence of the Crocodile and Marico rivers (Figure 61). The vegetation in this area is classified as geelhaak (Acacia erubescens) veld.1 One of the reasons for the localized occurrence of poisoning is probably that the plant is not normally grazed in significant quantities when other food is available.
During dry years and when the veld is overgrazed by cattle or game, the palatable Panicum maximum grass, growing predominantly in the shade under the A. erubescens trees, is replaced by S. tettense (Figure 58). A definite correlation exists between the occurrence of the disease and the camps with severely disturbed vegetation.Once S. tettense becomes established, no grass will grow in its vicinity, with the result that animals are forced to feed on it.156
The toxic principle of S. tettense is not known. Epileptiform seizures were experimentally induced in 6–12-month-old Afrikaner calves by dosing them with a total of c.43–64 kg of dried plant material at a rate of 5,0–7,5 g/kg/day over c.60 days, the lowest total dose being 280 g/kg. Mild signs could appear after approximately one month but typical seizures were evident only after two to three months of dosing.156
The morbidity rate varies from one to 70% and the mortality is usually very low, but animals may die as a result of drowning during dipping or have to be destroyed on account of severe injuries sustained during falling episodes. It should be pointed out that affected animals may survive for many years and still show the typical signs. Badly affected animals may sometimes be in poorer condition than the rest of the herd156 and show subtle behavioural or coordinational changes even at rest. Such animals may have to be separated from the herd to prevent them from being bullied by the healthy ones (T.S. Kellerman & T.W. Naudé, OVI, personal observations, 1995).
It is usually necessary to disturb, frighten or chase affected free-ranging animals to elicit signs of the disease. Clinical signs can often also be induced by merely tapping a horn or by forcibly raising the head of an animal for about 60 seconds, while at the same time closing its eyes, and then suddenly releasing the head (Figure 62). Just after an attack or when the animals are stabled and being handled regularly, it becomes increasingly difficult to produce signs with the usual stimuli, but these can often be precipitated by forcibly lifting the head. The clinical signs during a typical attack were described by Pienaar et al. (Figures 63, 64 and 65)156 as follows:
Affected animals appear to graze normally, with normal posture and gait, until disturbed, when signs varying from gross epileptiform seizures with collapse to only a mild lateral head tilt with slight ataxia and muscle tremors may be seen. Mild cases may show only a tilted head or rigid neck, often accompanied by slight degrees of hypermetria and dysmetria. Other animals adopt an attitude of star-gazing with the head raised and swaying from side to side, and a rigidly outstretched neck. They stumble sidelong in a crouching posture with stiff forelegs and a wide-based stance, often stumbling or crashing into objects. Severely affected animals fall and land with their full mass on their muzzles, knees or briskets. After falling, they struggle to regain their feet, frequently with the head folded in under the body or in a position of opisthotonus. A peculiar staring expression is seen in the eyes at the commencement of or during an attack. The eyes are also rotated upwards, showing the white of the sclera with lateral nystagmus, both before the animal falls and while it is down. Continual urination and defaecation may be seen during or following an attack. After regaining their feet, animals show generalized muscle tremors, most conspicuously over the flank and shoulder areas. A remarkable feature of the disease is the rapid recovery following an attack. Fallen animals quickly re-orientate themselves, regain their feet and appear normal within a few minutes. As a result of frequent falling, secondary trauma, e.g. broken horns, fractured teeth and jaws, bruising of the mouth, brisket and legs, are commonly present.
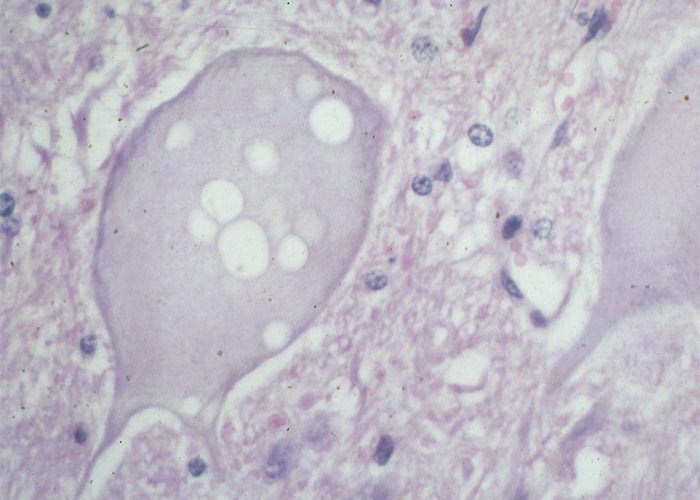
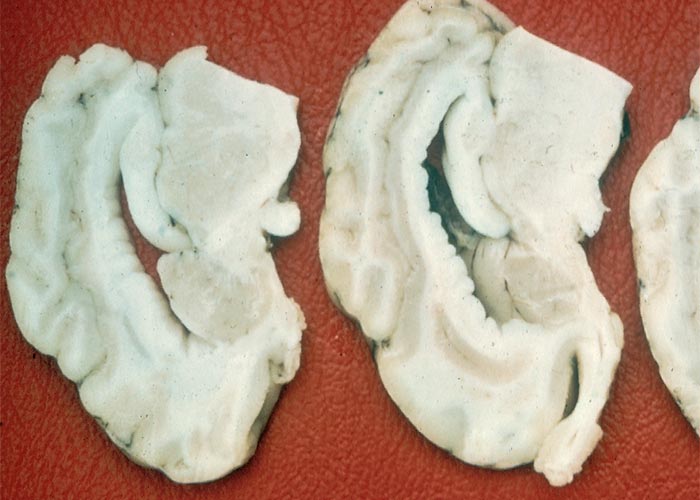
Apart from the trauma, the only other gross lesion is atrophy of the cerebellum. In severely affected animals the cerebellum is conspicuously reduced in size, while in milder cases it is often necessary to compare the affected cerebellum with that of a normal animal to appreciate the degree of atrophy. Atrophy occurs uniformly throughout all lobes and numerous folia are markedly smaller.
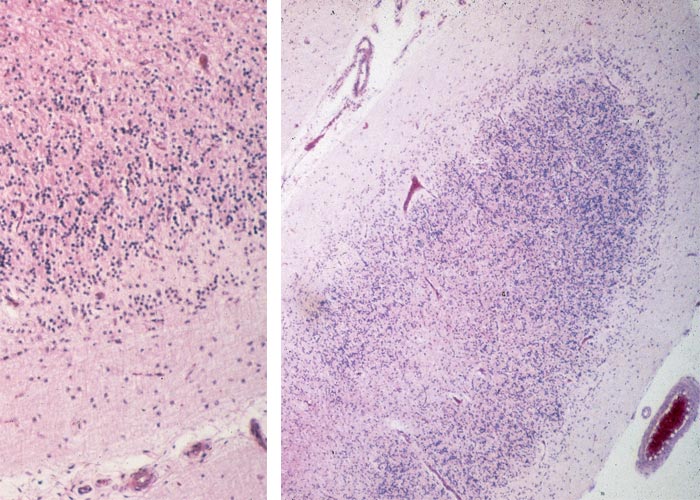
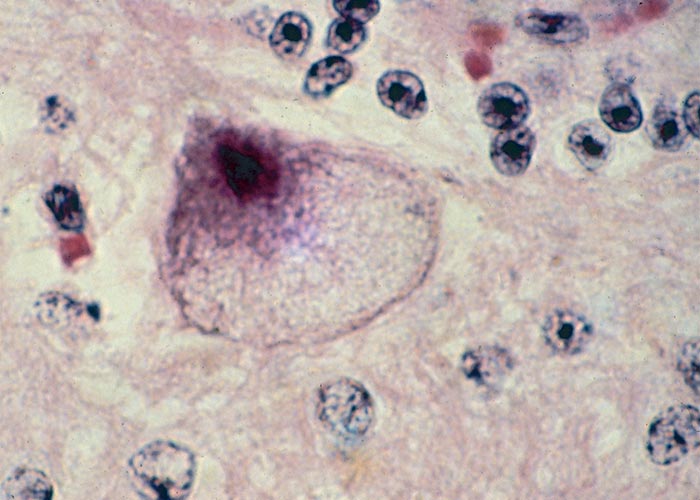
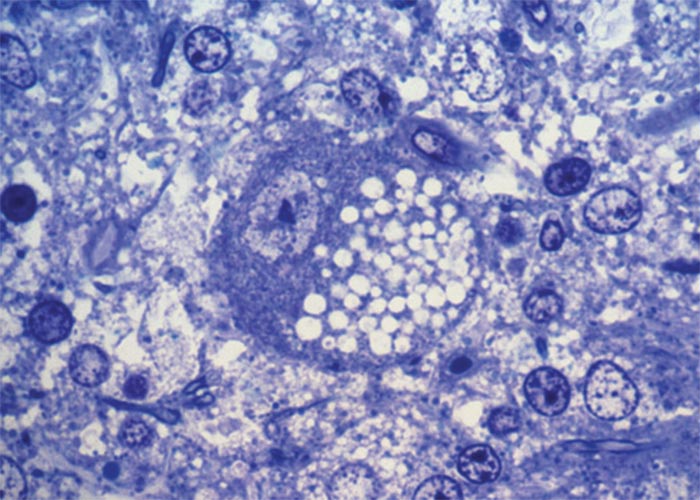
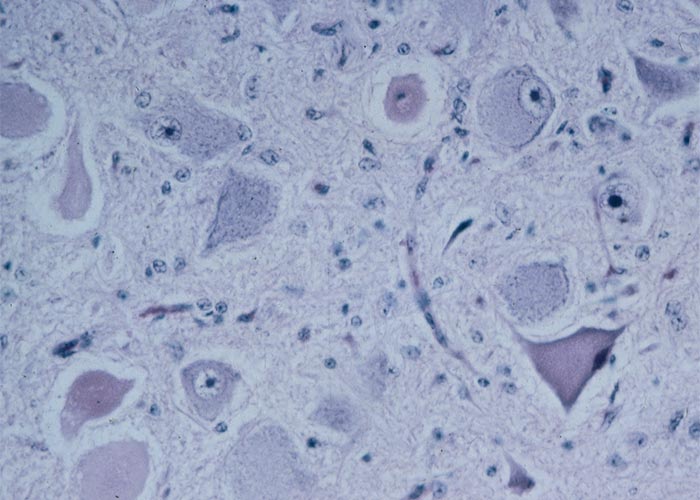
The most striking microscopical lesions are found in the cerebellum (Figure 66), where, in advanced cases the Purkinje cell layer is almost completely absent in most of the folia.130, 156, 165 A few normal Purkinje cells usually remain, but as a rule the majority are affected. In animals with early signs of the disease there is usually no obvious diminution of Purkinje cells, but most of them are affected to a greater or lesser extent.
A variety of changes occurs in the affected Purkinje cells. They are swollen and ballooned and contain numerous small vacuoles which are often located at one pole of the cell and which push the nucleus eccentrically (Figure 67). In the first case, a few larger vacuoles are sometimes visible towards the periphery of the cell body in a number of the neurons. There is a loss of Nissl substance and the cytoplasm often has a faintly eosinophilic and foamy to a hyalinized and strongly eosinophilic appearance. The nucleus in many of the cells is pyknotic and karyorrhectic. As a result of the loss of cytoplasmic and nuclear detail the neurons often assume ghost forms. The axons and dendrites of some of the affected cells are swollen. Van der Lugt described a second type of degeneration and necrosis of the Purkinje cells in which the cells were shrunken, hyperchromatic and less prominently vacuolated.208
The molecular, as well as the granular layers of the cerebellum are often diminished in thickness and the cells in the granular layer seem to be reduced. Other less important changes that can occur in some animals include mild status spongiosus of the white matter in the vicinity of the cerebellar peduncles, as well as diffuse gliosis and lymphocytic infiltration around some of the blood vessels of the cerebellum and meninges.156
Vacuolated neurons are also present in the cerebellar peduncles, midbrain, thalamus, medulla oblongata and nuclei in the ventral horns of the spinal cord. Odd neurons in the cerebral cortex too may be affected.
Other Solanum species are also toxic to animals and humans. Solanum dimidiatum in West Texas,130 S. fastigiatum var. fastigiatum in Brazil and S. bonariensis in Uruguay have been associated with clinical signs of seizures and pathology similar to that reported for S. tettense.165
There are about 1 500 species in the genus Solanum, and most of them have solanine as the toxic principle.112 The clinical signs of acute solanine poisoning include dermatitis, apathy, drowsiness, salivation, dyspnoea, trembling, progressive weakness and ultimately paralysis and unconsciousness. It may also have an irritant effect on the gastrointestinal tract, giving rise to anorexia, nausea, abdominal pain, vomiting and constipation or diarrhoea.112, 189 In South America, Solanum malacoxylon is responsible for arteriosclerosis of the aorta and major arteries and metastatic calcification in various organs, such as the heart, lungs, kidneys and tendons.54
A pathogenesis similar to a storage disease has been proposed for Solanum fastigiatum var. fastigiatum poisoning, where membranous cytoplasmic bodies compatible with gangliosidoses in animals and humans have been detected in vacuolated Purkinje cells.165 Although glycosidase inhibitors have been isolated from S. tettense138 and small numbers of cytoplasmic membranous bodies, which may represent storage material, have been demonstrated with electron microscopy,208 evidence that maldronksiekte is a storage disease is not conclusive.
Ipomoea carnea Jacq. subsp. fistulosa (Mart. ex Choisy) D.F. Austin (Convolvulaceae)
Shrubby morning glory
The plant has been implicated in an outbreak of lysosomal storage disease of goats in Mozambique. This was the first report of a plant-induced lysosomal storage disease in Africa.50
It is an erect to scrambling, rarely branched, densely leaved shrub of up to 3 m in height. The ovate to lanceolate leaves are 10–25 cm long, truncated or shallowly indented (cordate) at the base narrowing (long cordate) towards the apex. The leaves are alternately arranged with petioles of 2–20 cm. The deep pink to rose flowers, resembling those of a morning glory, are clustered at the tips of the branches50 (Figure 68).
Ipomoea carnea, an ubiquitous shrub from tropical America, has spread to various parts of the world. In southern Africa it is cultivated in gardens, windbreaks or hedges, from where it sometimes escapes to infest disturbed soil, e.g. road sides, or in moist places, such as river banks. It does not favour open grassland.50
Goats developed nervous signs after feeding on I. carnea hedges around the dwellings and kraals of a village near Maputo. The clinical signs, including ataxia, head tremors and nystagmus, had followed on protracted ingestion of the plant. Light and ultramicroscopical examination of brain tissues of goats naturally and experimentally poisoned by I. carnea revealed vacuolar changes consistent with those of a storage disease (Figure 69). The diagnosis was confirmed by demonstration of swainsonine and two calystegine glycosidase inhibitors in the plant material50 (Figure 70). Significantly, swainsonine and calystegines have also been demonstrated in a toxic Ipomoea sp. from Australia.138, 139
Similar vacuolar changes in neurons and other cells have been reported in sheep and cattle poisoned by Swainsona spp. in Australia,56, 117 Oxytropis and Astragalus spp. (locoweeds) in the United States of America151, 213 and in mannosidosis, a hereditary lysosomal storage disease in Aberdeen Angus cattle.101 The active principles in the plants mimic the genetic defect in cattle. Phytogenous indolizidine alkaloids, such as swainsonine, inhibit α- mannosidase, thereby causing oligosaccharides to accumulate in lysosomes.56, 57, 96 Calystegine B2 and calystegine C1 in I. carnea in their turn inhibit ß-glycosidase and α-galactosidases with similar results.
Aspergillus clavatus Desmazieres (Fungi: Hyphomycetes)
The fungus, Aspergillus clavatus (Figure 71), has been associated with hypersensitivity, muscle tremors, ataxia, paresis and paralysis in cattle grazing on sprouted wheat in France,98, 140 malt sprouts in China,100, 133 malt culms in Scotland,79 sprouted barley grains in Israel176, 177 and sprouted maize (Figure 72) and sorghum beer residues (SBR) (Figure 73) in South Africa.107, 108
Sorghum beer is a mildly intoxicating traditional drink in southern Africa. The fresh wet or dry residue is a nutritious supplementary feed for stock. Sorghum beer residue (SBR) is usually free of mould when it leaves the factory, but it is soon colonized by many fungi, including A. clavatus. Under some circumstances A. clavatus may predominate. On the farm where the first outbreak of A. clavatus poisoning occurred, 130 out of 330 cattle died after eating mouldy SBR and many suffered more or less permanent locomotory disturbance.108 The condition is fairly regularly diagnosed in South Africa, outbreaks of A. clavatus poisoning having variously been reported in dairy cows fed mouldy sprouted maize as a green fodder107 and beef steers on a commercial ration incorporating mouldy sorghum (P.W. Nel, N. Fourie & T.S. Kellerman, ARC-OVI, unpublished data, 1992).
Aspergillus spp., including A. clavatus, are known to produce mycotoxins, such as patulin,12 cytochalasin E,82 escladiol197 and the two tremorgenic metabolites, tryptoquivalone and tryptoquivaline.44, 82 It was not possible, however, to demonstrate these toxic metabolites on thin layer chromatography in methanol extracts of A. clavatus-infested SBR, which had elicited tremors in a sheep.107 A Friesland steer, dosed on two consecutive days with c.43 g/kg of a pure culture of A. clavatus isolated from toxic SBR, showed typical clinical signs 44 hours after the initial dose.108 A bovine, dosed at a level of 5–10 g/kg for five days and which received a total mass of 8,7 kg SBR, developed the tremorgenic syndrome on Day 4108 Sheep have also been experimentally poisoned with A. clavatus.107, 108
The clinical signs of A. clavatus poisoning (Figure 74) include hypersensitivity, tremors, ataxia, progressive paresis, paralysis and constipation.98, 107, 108, 140 The tremors (which are not invariably present) usually start inconspicuously in the flanks and hindquarters prior to spreading to the rest of the body. Affected animals walk with a stiff-legged gait, taking short steps and sometimes knuckling over at the fetlocks. They often stagger about drunkenly and fall repeatedly before paralysis, recumbency and death sets in. Affected animals seem to be well aware of their environment and, although they cannot eat or drink, the tongue, limbs and tail are seldom paralysed (cf. botulism). Saliva often drools from their mouths. Chronically affected cattle can survive for several weeks or even longer.107 Clinical signs are usually not evident when these animals are at rest; however, on exercise, they may tire easily, walk with the back arched, brush and knuckle over at the fetlocks, tremble, show respiratory distress and salivate. Eventually they may slip into a dog-sitting position or go down in sternal recumbency, often with the legs abnormally disposed (Figures 75 and 76). After a variable period of rest they may rise, often with difficulty.
Macroscopical lesions are generally more prominent in severely affected animals that show clinical signs for extended periods.107, 108 In those with pronounced muscle tremors, rigor mortis of the limbs sets in within minutes of death. Although skeletal muscle lesions are the most obvious macroscopical change, they are not visible in mildly affected animals. Greyish-white, almost fish flesh-coloured areas of hyaline degeneration and necrosis occur, particularly near the insertions and origins of the larger muscle groups of the hind- and forequarters (Figure 77).
Petechiae and oedema are frequently present in the muscle, as well as in the intermuscular fasciae. The myocardium often has a parboiled appearance and in longer surviving cases, small foci of necrosis are noted in the papillary muscle and wall of the left ventricle. There can also be stasis of the rumen and large intestines. Apart from meningeal congestion in some animals, the brain is not overtly affected.
The most significant microscopical lesions occur in the brain and spinal cord.107, 108 The larger neurons, especially in the ventral horns of the spinal cord and those in selected nuclei in the medulla oblongata, the midbrain and the thalamus are consistently involved. Affected neurons are swollen and in many instances reveal complete chromatolysis which gives the cytoplasm a finely granular and slightly or markedly eosinophilic appearance (Figure 78). Small cytoplasmic vacuoles may be visible in some affected neurons. Many of the nuclei of degenerated or necrotic neurons are situated eccentrically, are pyknotic and either faded or absent (Figure 79). Satellitosis, neurophagia and focal gliosis are only occasionally seen in the more chronic cases.
According to Van der Lugt (2002), in cases examined by him, the severe bilateral symmetrical degeneration, resembling Wallerian degeneration in the spinal cord, was suggestive of a primary axonopathy with secondary myelin loss.208
Valsiekte
Falling disease
In the late winter of 1979 a new disease of sheep, valsiekte, characterized by ataxia of the hindquarters, broke out in the Bethulie district of South Africa. The disease has since been reported further afield in the districts of Middelburg (Eastern Cape Province) and Jagersfontein (Free State)210.
The clinical signs appear in lambs between the ages of approximately two to four months. Affected lambs drop out of the flock when driven hard. At first they stumble, then the hindquarters give way, causing them to fall (Figure 80) sometimes in a dog-sitting position. In this position they struggle forward, often on bent knees, dragging their hind legs behind them, before collapsing on the sternum (Figure 80). After a short rest the less severely affected ones get up again.
The animals do not appear to be hypersensitive. Affected lambs often die suddenly after a few weeks, without showing signs of deterioration in their condition. They eat normally throughout. Note that the clinical signs of valsiekte are in many respects similar to those of Tribulus terrestris and T. micrococcus staggers of sheep in Australia (see Discussion).
At necropsy, bruised, hairless areas are evident on the carpal joints, dorsal aspects of the hind fetlocks, and the belly, where injury occurs during creeping episodes.
The microscopical lesions closely resemble swayback, which is a result of copper deficiency. The lesions include a status spongiosus, especially in the dorso-lateral and ventral tracts along the entire length of the spinal cord, and, more rarely, in the medulla oblongata. Many of the larger neurons in the spinal cord show changes which include chromatolysis, cytoplasmic vacuolization, nuclear margination and karyolysis.
Answers to a questionnaire submitted to farmers in 1981 revealed that during a typical outbreak of valsiekte the grazing is composed of about 10–30% Chrysocoma ciliata (bitterbos) and 10–80% Rosenia oppositifolia (bekkerbos). Bekkerbos was not invariably present. Two other important facts to emerge were that almost without exception only Dorper sheep were affected and that 13 out of 15 outbreaks of valsiekte occurred concurrently with kaalsiekte.210 Chrysocoma ciliata is known to cause two syndromes in sheep, namely alopecia (kaalsiekte) in new-born lambs whose dams browse large quantities of the plant during late pregnancy and diarrhoea in mature sheep185, 189 (see The skin and adnexa). Although the incidence of valsiekte apparently coincides with that of kaalsiekte, the reverse is not necessarily true.
A limited dosing trial in which C. ciliata and R. oppositifolia were dosed to lambs failed to reproduce the condition, nor did C. ciliata and R. oppositifolia given separately and together, to ewes before and after parturition, and to their lambs, induce valsiekte.
Failure of the dosing trials does not exclude the possible involvement of either of these plants, as the low morbidity rate of valsiekte militates against its experimental reproduction in small numbers of sheep. Of the two plants, C. ciliata would be the more probable causative agent in view of the apparent coincidence of valsiekte and kaalsiekte and the constant presence of the plant during outbreaks.
Although copper deficiency cannot be ruled out as a possible cause of the disease, low copper levels could not be demonstrated in the livers of affected sheep. Further evidence that copper deficiency might not be involved is the fact that high liver copper levels rather than low ones would be expected in this area and that copper supplementation purportedly did not reduce the prevalence of the disease.210
Trachyandra spp. (Asphodelaceae)
T. laxa (N.E.Br.) Oberm.
T. divaricata (Jacq.) Kunth
The plants have been associated with progressive, ascending paresis and paralysis in cattle, sheep, horses and pigs in the Western Cape Province of South Africa and in Namibia (Figure 81).83, 148
Trachyandra spp. are typical geophytes with many soft succulent leaves (Figure 82) arranged in rosettes.83, 148 The leaves of T. divaricata are f lat and strap-like while those of T. laxa are cylindrical. Large, branched inf lorescences bear small white flowers, followed by capsules. Trachyandra laxa is a tumble-weed. The plants are palatable early in the growing season and outbreaks occur in overgrazed areas during the dry months of August to December. The occurrence of the disease ceases soon after the onset of good rains and improvement of pastures.
The disease has been experimentally reproduced in a sheep fed ad libitum for six months on freshly cut plants, and clinical signs of the disease were induced in a horse after it had been fed on fresh plants for six weeks.
Clinical signs in the different species of animals are very similar. During the early stages of the disease the animals move with difficulty, knuckle over at the fetlock joints and show various degrees of hypersensitivity and muscle twitching. In horses, these signs improve after light exercise, and even disappear, although the animals tire easily and show laboured breathing. At times some animals may reveal over-knuckling and over-flexion of the hock and carpal joints and occasionally also spasticity of the hind legs. Progressive paresis and paralysis usually follow and the animals go down in sternal recumbency (Figure 83). Affected animals die as a result of starvation, exposure or secondary complications one to three months after the onset of clinical signs. With proper feeding or with better grazing, total recovery from the early signs may occur after six months, but in other cases animals recover only partially and retain a paretic limb.
At necropsy, no macroscopical changes are usually seen. A khaki-brown pigmentation, especially of the larger nuclear areas in the brain and/or spinal cord, may be visible in some animals.
Microscopically, yellowish-brown to greyish-brown intracytoplasmic pigment, which gives a moderate positive reaction with the Schmorl’s method for lipofuscin, is present, particularly in the larger neurons in the brain, spinal cord and ganglia (Figure 84). The nuclei in the thalamus, putamen, amygdaloid nucleus, midbrain and medulla oblongata, as well as in the neurons in the dorsal horns in the cervical and lumbar enlargements of the spinal cord, are most frequently and most severely affected. The Purkinje cells in the cerebellum are not affected. A mild status spongiosus of the cerebral white matter has been noticed in some animals poisoned by T. laxa in Namibia, but this could be the result of the ingestion of the plant Helichrysum argyrosphaerum (vide infra) or infestation with Strongyloides papillosus (P.A. Basson & J.G. Pienaar, Grootfontein, Namibia, unpublished observations, 1983). A similar pigment can be found in the Kupffer cells of the liver, in the epithelial cells of the kidneys and in the reticulo-endothelial cells of the lymph nodes and spleen.
Intraneuronal lipofuscin pigment is often present in the brain and in other cells of different tissues, such as the myocardium, in older, healthy animals. The mere presence of this pigment in the brain is thus not necessarily indicative of this disease. Intracytoplasmic accumulation of melanin in the neurons in the thalamic area is a common feature of healthy Karakul sheep in areas where Trachyandra poisoning occurs. A melanin-like pigment has also been reported in neurons in Phalaris staggers.
Phalaris staggers
Phalaris poisoning or staggers is a progressive neurological disorder of sheep and cattle induced by the perennial grass, Phalaris aquatica (Tawoomba canary grass) and the annual P. minor (canary grass) (Figure 85). In Australia and New Zealand, P. aquatica22 is usually associated with poisoning. In South Africa sporadic outbreaks involving P. minor have been reported in the Swartland of the Winter Rainfall Area in the Western Cape Province212 (D.J. Schneider, Regional Veterinary Laboratory, Stellenbosch, personal communication, 1984) and an unidentified Phalaris species in Mpumalanga (S.S. Bastianello, ARC-OVI, unpublished data, 1999).
Phalaris minor, characterized by its densely paniculate spike-like inflorescence, is an exotic cultivated grass which occasionally escapes from artificial pastures. In the Swartland, it can become troublesome on reaped lands or on disturbed areas next to roads. Poisoning occurs only when the pasture is dominated by this grass or where it is preferentially grazed. New growth is thought to be the most toxic.
The causative toxins in the grass are thought to be three tryptamine alkaloids which are structurally similar to serotonin. These alkaloids are said to cause both neurological signs and cardiac abnormalities, such as tachycardia and ventricular block because of interference with the chemical transmitter, serotonin, in the spinal cord pathways of the autonomic nervous system.22, 31 However, newer work suggests that neither the methylated tryptamines nor beta-carbolines play a primary role in the aetiology of the ‘sudden death’ syndrome. Bourke and his coworkers could induce only the nervous and not the cardiac (sudden death) form by intravenous and oral administration of three tryptamine alkaloids to sheep. Some evidence was found incriminating phenylethylamines in the cardiac form of the intoxication.35, 36
Three distinct entities or syndromes have been reported in sheep: a sudden death or cardiac syndrome where animals may die as soon as four hours, but usually 12–72 hours, after being exposed to the toxic grass; acute Phalaris poisoning with transient nervous signs; a chronic poisoning or Phalaris staggers which is a slowly progressive and irreversible, fatal neurological disorder that develops in sheep two to three weeks after grazing on the pastures.22, 88
The clinical signs of the sudden death or cardiac syndrome in sheep are characterized by collapse, especially when excited, respiratory distress, cyanosis, arrhythmic tachycardia, ventricular fibrillation and cardiac arrest.22 With the nervous forms of the poisoning, hypersensitivity and generalized tremors develop during the initial stages only when the animals are disturbed. Affected sheep often walk with stiff legs and they drag their hind legs as a result of an inability to bend the hocks. These signs are followed by incoordination and swaying of the hindquarters, while severely affected animals go down in lateral recumbency, showing tetanic convulsions, paddling movements, muscle tremors, nodding of the head, nystagmus and salivation. Some of the sheep may die at this stage, but if left undisturbed others may recover and walk away apparently unaffected. The condition in some animals may worsen and they may die if left on the pasture. On the other hand, mildly affected sheep recover if removed from the pasture in time. Deterioration of the nervous signs can occur even after the animals have been removed from the pastures. Some animals show apparent recovery, but clinical signs can often be elicited by forcing them to exercise. Signs may persist for as long as two months. The morbidity ranges from five to more than 50% in outbreaks.
In cattle, the clinical signs are usually restricted to stiffness of the hocks and dragging of the hind toes. Only severely affected animals may occasionally show nervous signs similar to those in sheep.22, 212
The macroscopical changes in sheep that die of the cardiac syndrome include lesions compatible with acute heart failure such as generalized congestion and cyanosis, oedema of the lungs and a few scattered haemorrhages on serosal and visceral surfaces. More chronically affected animals reveal greyish-green to bluish discolourations, focally symmetrical in certain areas of the brain stem and midbrain, as well as in the ventral horns and dorsal root ganglia of the spinal cord. Pigmentation is commonly seen in the renal medulla and less commonly in the liver. The pigment in the tissues leaches into the formalin after several weeks storage, giving it a pale blue colour. Symmetrical pallor of several hind leg muscles has occasionally been noted in advanced cases.88
Microscopically, a yellowish-brown granular pigment occurs in variable amounts in the cytoplasm of neurons in the affected areas of the brain and spinal cord. These neurons may eventually show degenerative and necrotic changes. Mild to moderate Wallerian degeneration of nerve fibres is sometimes present in the white matter of the spinal cord.88, 212 Ultramicroscopy suggests that the pigment accumulates in and destroys mitochondria, and may then be taken up by lysosomes. The pigment is mostly amorphous, with electron-dense areas in some cells and finger print patterns in others.88
The introduction of heavy cobalt pellets in the rumen prevents the nervous form of the disease.22, 88 Parenteral administration of cobalt or vitamin B12 is not effective. It is believed that cobalt stimulates proliferation of certain micro-organisms in the rumen, which are capable of destroying the causative agent.22
Neuronal pigmentation has also been reported in healthy ageing animals,174 as well as in animals poisoned by Trachyandra.83, 148
Helichrysum argyrosphaerum DC. (Asteraceae)
Wild everlasting, sewejaartjie, poprosie
Ingestion of large quantities of Helichrysum argyrosphaerum causes amaurosis, paresis and paralysis, mainly in sheep. It is a prostrate annual plant, forming mats of up to 500 mm in diameter (Figure 86). The 25 mm long, greyish-green, sessile leaves are borne alternately. The bracts of the f lowerheads are silvery-white to rosy-purple (Figure 87). Flowering time is throughout the year, but mostly from July to October. Not with standing the wide distribution of the plant in southern Africa, outbreaks of the disease have been restricted mostly to Namibia.11 Helichrysum argyrosphaerum is the only one of the several hundred species of this genus in South Africa to be implicated in poisoning of stock.
Helichrysum argyrosphaerum prefers red sand or sandy loams but often grows in stony or even gravelly soils. It is commonly found on exposed, disturbed ground such as along railway lines and roadsides, on cleared ground, old termite mounds and on cultivated or fallow lands. The plant can also occur on flats, dunes and dry river beds. This composite may become abundant whenever the veld is heavily grazed and can be an excellent fodder for antelope, cattle and especially sheep.
The toxin has not yet been isolated and the plant appears to be most toxic in the flowering stage. A sheep, dosed with 15–20 g/kg/day or a total dose of 15,3 kg of dried (400 g/kg) plant material for 41 days, showed progressive paresis and apathy, but not blindness. The microscopical lesions in the brain of this animal were compatible with those seen in field outbreaks of H. argyrosphaerum poisoning .11 Cattle can be affected, but the poisoning is more common in sheep.
The morbidity rate in sheep varies from one to 29% and although mortality is low, practically all the affected animals may become unthrifty. The poisoning is characterized by blindness which is sometimes accompanied by nervous signs such as ataxia, circling, chorea of the head and neck muscles, star-gazing, nystagmus, paresis and paralysis. Two forms of blindness were observed, one marked by amaurosis and the other by cataracts.11
The most notable lesions are present in the eyes, nervous tissue and skeletal muscles. Macroscopically the optic chiasm and optic fasciculi are swollen, with the latter seemingly compressed at the optic foramen. The gyri of the brain may be somewhat flattened as a result of swelling and oedema, and sectioning of the fixed brain reveals that the periventricular white matter is distinctly swollen, gelatinous and off-white in colour. As a result of the swelling, the ventricular surface of the corpus callosum and centrum semiovale is notably undulated (Figure 88).
Microscopically the amaurotic sheep reveal congestion, oedema and mild haemorrhage in the choroid and retina, especially in the region of the fundus and optic papilla. Necrosis commences in the layer of the rods and cones near the optic papilla and eventually leads to the disappearance of various layers, including the inner nuclear layer. The lesion has a patchy distribution. A pronounced bilateral status spongiosus and areas of malacia, with gitter cell mobilization and big cavities, occur in the optic fasciculi and chiasm. The bilaterally symmetrical status spongiosus of the white matter is much more severe in the midbrain and thalamus in fatal cases. This lesion has a higher degree of prevalence and severity in the periventricular area and the white matter in the hippocampus, corpus callosum, subcollosal tract, fornix, centrum semiovale, septum pellucidum, optic tracts, lateral geniculate and certain nerve tracts in the brain. Mild spongy lesions can also be detected in the white matter of the spinal cord in about 40% of amaurotic animals, the highest incidence being in the Lissauer’s tract and the fasciculus cuneatus and fasciculus gracilis. Similar mild lesions occur in some nerves such as the trigeminus, intermuscular and spinal nerves.11
The cataractous sheep show a range of lesions in the lens. These include proliferation, swelling, degeneration and necrosis of the epithelial cells, as well as degenerative and necrotic lesions (homogenization and acidification) in the cortex. Morgagnian globules94 are frequently present in cracks and clefts in the lens. Similar but milder lesions occur in the optic fasciculi, optic chiasm, brain, spinal cord and nerves of the cataractous sheep than in the animals with amaurosis.11 Van der Lugt and his co-workers209 described degenerative retinopathy in five naturally intoxicated sheep. This consisted essentially of degeneration and loss of photoreceptor outer segments, particularly in the nontapetal retina. Other retinal changes included hyperplasia and hypertrophy, accompanied by migration of pigmented epithelium; focal separation; depletion and loss of the nuclear layers.
Apart from the lesions in the eye and nervous tissue, some animals show degeneration and necrosis of individual skeletal muscle fibres.11
Ultramicroscopical examination of three naturally affected small stock revealed oedema of the myelin, as evidenced by intramyelinic vacuoles formed by splitting of the lamellae at the intrapod lines. In addition, the perivascular and extracellular spaces in severely affected areas were mildly distended. Oedema of myelin, associated with degeneration and loss of axons and myelin, as well as astrocyte gliosis, was observed also in the intraorbital and intracranial portions of the optic nerves. More chronic changes in the intracanalicular segments of these nerves, such as fibrosis and atrophy, indicated that the neuropathy was in this case brought about by compression of the nerves as a result of the oedema of the myelin.209
Cattle are occasionally affected with H. argyrosphaerum poisoning. They show stiffness and posterior paresis and/or paralysis, but not blindness. No microscopical lesions were apparent in the eyes, optic fasciculi, brain and spinal cord of one animal studied. The only noteworthy lesions occurred in the skeletal muscles, in which widespread degeneration, necrosis, oedema and haemorrhage, as well as evidence of sarcolemma nuclei proliferation and regeneration occurred.11
Amaurosis in sheep has been reported with the halogenated salicylanilide compounds such as rafoxanide and closantel used as anthelmintics in sheep.150, 159 The clinical signs and pathology in the brain, optic fasciculi, spinal cord and nerves are almost identical to those described in Helichrysum poisoning. However, the changes in the eyes differ somewhat in rafoxanide poisoning. The neurons in the ganglionic layer of the retina show degeneration and necrosis, and, in chronically affected sheep, only a few neurons may be spared. In Helichrysum poisoning the layer of rods and cones is primarily involved. Amaurosis has also been reported with Ornithogalum poisoning in cattle189 (see Gastrointestinal tract).
The stiffness, posterior paresis and/or paralysis in sheep and cattle and the associated skeletal muscle lesions closely resemble clinical signs reported for Geigeria poisoning157 (see Gastrointestinal tract).
Cotula nigellifolia DC. K Bremer & Humphries sens. lat. (Asteraceae)
(= Matricaria nigellifolia) Stagger weed, stootsiektebossie
Cotula nigellifolia is responsible for a nervous disorder of cattle known as stootsiekte (pushing disease). The plant is a soft-stemmed, straggling, creeping or decumbent, annual or perennial, growing c.500 mm high, with deeply incised, carrot-like leaves (Figure 89). The ray florets of the daisy-like inf lorescences are short, broad and white, while the disc florets are yellow (Figure 89).147, 205
Cotula nigellifolia will grow on sand or clay, from about sea-level to 1 500 m. It is a water-loving plant which prefers damp or wet soil and shady places, such as the banks of rivers, dams, marshes, vleis, or irrigation furrows, where it sometimes grows semi-submerged, with its stems floating in the water.
In South Africa the plant is found from the Eastern Cape Province, through KwaZulu-Natal to the former Transvaal (Figure 90). Field outbreaks of the disease have generally been confined to KwaZulu-Natal, in recent times particularly to the Vryheid and Ixopo districts.
Andrews5 first established that C. nigellifolia was the cause of stootsiekte. Although the toxic principle is not known, the disease has been reproduced by feeding dried, milled plant material to Afrikaner-type steers at a dosage level of 5 g/kg/day for two to 12 days (i.e. MTD 10 g/kg).147 The clinical signs in these animals appear abruptly after a latent period of 16–44 days. The length of the latent period seems to be inversely related to the dose of plant5, 147 and it would appear that animals that receive less plant material show milder clinical signs. Sheep are apparently resistant to stootsiekte.5, 146
Affected cattle consistently show signs of docility, apathy, clumsiness, aimless wandering and pushing with the head against objects; the pushing seeming to increase in frequency during the course of the disease (Figure 91).5, 147 Even animals that are unaccustomed to being handled can be led without resistance. Other clinical signs are: reduced response to stimuli, such as noise and sudden movement; bumping clumsily against objects; incoordination, which includes scuffing of the feet, knocking on, brushing and occasionally knuckling over at the phalangeal joints. The clinical condition of the affected animals often deteriorates rapidly over eight to 48 hours.
Apart from subcutaneous contusions no other gross lesions are usually seen. Microscopical changes in the brain, however, are usually sufficiently distinctive to substantiate a diagnosis of stootsiekte.147 The lesions are generally confined to the white matter throughout the forebrain and midbrain and may extend into the white matter of the cerebellum. Occasionally, perivascular gliosis can be detected in closely adjacent zones of grey matter.
The most characteristic lesions are perivascular gliosis and inflammatory cell infiltration (Figures 92 and 93). The glial cells are believed to be microglia and are often arranged in palisade patterns with their long axes lying perpendicular to the vessel wall. Lymphocytes, occasionally plasma cells, and cells laden with pigment resembling lipofuscin are sometimes dispersed among these glial cells. Small, periodic acid-positive granules are numerous in the perivascular microglia. Hypertrophied astrocytes, with abundant eosinophilic cytoplasm and enlarged eccentric and reniform or lobulated nuclei, are usually scattered throughout the white matter in the affected areas. The above changes are often accompanied by a mild status spongiosus of white matter in the areas of gliosis.
Although the perivascular changes described here are features usually associated with infections or allergic encephalitides, they are unique for a plant poisoning. The distribution of the lesions, furthermore, suggests that the primary site of toxic insult is the cerebral vasculature.147
Fusarium verticillioides (Sacc.) Nirenberg Fungi: Hyphomycetes
(= F. moniliforme)
Leukoencephalomalacia (LEM) is a highly fatal neuromycotoxicosis of Equidae characterized by multifocal liquefactive necrosis of the white matter of the brain. The disease is caused by the ingestion of maize colonized by Fusarium verticillioides,51, 126, 127, 223, 224 one of the most prevalent moulds on harvested maize in the world23 (C.M.I. Distribution Map of Plant Diseases, Map No. 102 Ed. 4, 1972). Outbreaks of LEM have been recorded among others in the USA40, 86, 173 South Africa,104, 155 Argentina,166 China,97 Egypt7 and New Caledonia.55
The characteristic pinkish mycelium of the F. verticillioides is particularly conspicuous where the maize cobs are damaged by insects (Figure 94). This colour and lack of pycnidia macroscopically distinguishes F. verticillioides from Stenocarpella maydis, arguably the most toxicologically important cob rot fungus in southern Africa.
In the USA, the disease is particularly prevalent in wet seasons following droughts or when late maturing maize is affected by frost. Although the precise conditions favouring outbreaks of LEM in South Africa are not known, the relatively low prevalence of the disease can probably be attributed to the dry winters. In practice, field outbreaks appear to be associated with heavy rains and high humidity during the latter part of the growing season. Damage to cobs by insects such as stalk borer, also plays a part in the promotion of LEM by encouraging growth of the fungus. LEM has been reported in South African horses fed maize chaff, cracked maize, mouldy cobs and even a commercial ration. None of the limited number of field cases examined in South Africa exhibited the liver lesions sometimes seen in experimentally induced LEM,155 but hepatic lesions have been reported in natural cases of LEM in the USA.38 It is possible that a hepatic syndrome associated with mortality of horses fed F. verticillioides-infected bean hay in 1934 may have been the liver component of LEM.211
The overwhelming majority of natural intoxications occurred in horses that had either grazed on harvested maize lands or been fed mouldy, home-grown maize. No outbreaks have been recorded in commercial maize graded according to the standard South African maize grading regulations. The extreme hazard of feeding mouldy maize to horses, therefore, cannot be over-emphasized.
LEM has been experimentally reproduced with pure cultures of F. verticillioides in South Africa113, 126 and abroad.223 The amount of culture material needed to induce LEM roughly varied between 1,0 and 2,5 g/kg per day, administered for about 7 to 144 days,113, 126 or a total dose of c.3,8–63,8 kg per horse, depending on the toxicity of the culture. As can be seen, the toxicity of isolates and cultures varied considerably. One of the cultures that fatally poisoned two horses after administration, respectively, of 2 902 g and 6 365 g in divided doses over 11–12 days, contained c.1 g/kg fumonisin B1. Animals in these experiments variously manifested LEM without other significant lesions;126 LEM with hepatosis; brain oedema with hepatosis 113, 126, 127 and hepatosis alone.104, 113, 126
LEM has also been induced by intravenous127 and oral105 administration of fumonisin B1 (Figure 95). In the first trial,127 a horse was injected intravenously seven times from Day 0–Day 9 with 0,0125 mg of fumonisin B1/kg body mass/day. Clinical signs of neurotoxicosis, which appeared on Day 8, included nervousness followed by apathy, a wide-based stance, trembling, ataxia, reluctance to move, paresis of the lower lip and tongue, and inability to eat or drink. Euthanasia was performed on the horse on Day 10 while it was in tetanic convulsions. The principal lesion was severe oedema of the brain and early, bilaterally symmetrical, focal necrosis in the medulla oblongata.127
The second trial involved the administration of fumonisin B1 per stomach tube to two horses. A filly received 59,9 mg/kg of a 50% preparation of fumonisin B1 administered in 21 doses of 1,25–4 mg/kg over 33 days and a colt 44,3 mg/kg of 95% pure fumonisin B1 in 20 doses of 1–4 mg/kg in 29 days. The total amount given to the colt of live mass 190 kg was 8,417 g fumonisin B1. Both animals developed nervous signs, such as apathy, changes in temperament, incoordination and walking into objects, and one showed paralysis of the lips and tongue. Characteristic lesions of LEM were present in their brains. These trials provided conclusive proof that fumonisin B1 can induce LEM in horses.105
Ross and co-workers167 fed naturally contaminated maize to horses in a trial carried out in two parts separated by a period of 120 days on wholesome (less than 1 ppm fumonisin B1) maize. In the first part of the trial, lasting 98 days, the ponies were given maize containing 44 ppm fumonisin B1. One died acutely on Day 9 and another was euthanazed on Day 45. Both animals had encephalopathy and hepatic lesions. In the second part the surviving ponies ate maize containing 88 ppm fumonisin B1 with the result that one died on the 75th day and the other had to be euthanazed for humane reasons on the 78th. Leukoencephalomalacia and hepatic lesions were evident in both horses.
The following levels of fumonisin B1 were recorded in maize and feed associated with LEM: 37–122 ppm B1 (3 samples),225 7,7 ppm B1 and 3,1 ppm B2 (mean of 14 samples),203 1–126 ppm B1 (98 samples from 44 outbreaks)168 in the USA; 0,2–38,5 ppm B1 and 0,1–12,0 ppm B2 with means of 12,0 and 4,1 ppm, respectively (20 and 18 samples) in Brazil;198 and 8,9 ppm B1 and 3,0 ppm B2 (1 sample) in South Africa.175
Isolation of the toxic principle was in the past hampered by lack of a susceptible laboratory animal for bioassay of the chemical fractions. Wilson and Maronpot,223 for instance, could not induce overt signs of illness by dosing LEM-producing cultures to a variety of animals, including chickens, guinea-pigs, hamsters, rats, mice and rabbits. Nor could moniliformin, the most toxic metabolite of F. verticillioides then known, be demonstrated in LEM-inducing cultures.38, 126
The problem of finding a bioassay was solved by Gelderblom and his co-workers77 using a known histochemical technique to demonstrate foci of elevated γ-glutamyl transferase activity in the livers of exposed rats.170 Using this procedure they isolated a novel mycotoxin, fumonisin B1, from cultures of F. verticillioides. The structure of fumonisin B1 was characterized by Bezuidenhout et al. (1988).19
Leukoencephalomalacia has recently been reproduced both by intravenous injection and intragastric administration of fumonisin B1 to horses.105, 127
Fumonisins were shown by Wang and her co-workers217 to block the synthesis of sphingolipids by inhibiting sphingosine and sphinganine N-acetyltransferases (ceramide synthase). This mode of action217, 226 could probably be ascribed to the structural similarities between fumonisins and sphingosine.217 They suggested that a lack of sphingolipids (highly bioactive molecules) would disrupt the metabolism of cells, thereby affecting their growth, differentiation and behaviour.217, 218 Wang et al. (1992) demonstrated that within days of giving ponies feed contaminated with fumonisin B1 the levels of free sphinganine (and sometimes sphingosine) in their sera rose while that of the complex sphingolipids fell. When toxicosis was evident as indicated by other serum markers, the levels of the complex sphingolipids also increased, probably due to loss of sphingolipids from dying cells. These findings confirmed that consumption of fumonisin-contaminated feed disrupted sphingolipid metabolism. Because the levels of sphinganine and sphingosine rose before liver enzyme activities were noticeably elevated, they may be an early marker of exposure to fumonisins.218
The clinical signs of LEM usually appear abruptly and the course of the disease is short, normally lasting from only a few hours to about five days. The onset and progress of LEM might be so rapid that horses are occasionally found dead in the morning without signs of intoxication having been seen. Affected animals manifest a variety of signs such as changes in temperament, sweating, paralysis of the lips and tongue, locomotory disturbances and convulsions. The change in temperament may range from apathy and docility to frenzy and that of the gait from severe incoordination to almost normality, probably depending on the extent and siting of the lesion. Even when LEM is not accompanied by pronounced hepatic lesions, the terminal nervous crisis is often preceded some days before by bilirubinaemia and elevation in the serum activities of aspartate transaminase and γ-glutamyl transferase.105, 113, 126, 127 Recovery is rare.
The nervous crises induced by cultures of F. verticillioides 113, 126 were typically marked by initial apathy followed by hypersensitivity/agitation, sweating, muscular tremors and locomotory disturbance. Ataxia was manifested as posterior weakness, (swaying hindquarters), walking awkwardly with the rump tucked under, taking short steps, lifting the hind feet high (goose-stepping), brushing, stumbling, circling, pushing and colliding with objects (Figures 96 and 97). Before death, the animals fell down in convulsions or even became frenzied. Some were apparently blind, but, in this instance, the blindness could have been due to incomprehension of visual stimuli rather than impairment of vision (Figure 98). Inability to swallow, paralysis of the lower lip, and protrusion of a flaccid tongue from the mouth completed the clinical picture (Figure 99).113 126
Note that the nervous crisis was not invariably associated with foci of malacia. Examination of the brains of a number of horses that had pronounced nervous signs revealed only oedema.
In those experiments where liver lesions alone were induced, the clinical signs included progressive weakness, apathy, icterus, and oedema of the lips, supraorbital fossae and legs (Figure 100). These signs were accompanied by the previously described chemical pathological changes indicative of liver damage.104
Large malacic lesions were sometimes revealed by a softening of the external surface of the cerebral cortex or by swollen areas that fluctuated on palpation. Gyri over the swollen areas could be slightly flattened and tinged yellow. On coronal section large cavities were visible where the subcortical white matter had undergone liquefactive necrosis (Figure 101). Numerous haemorrhages and a yellow discolouration that turned green on fixation in formalin were evident in the white matter bordering on the cavities. Microscopical examination of the pericavital areas revealed rarefaction of the white matter, perivascular haemorrhages, oedema, and cell infiltrates composed mainly of plasma cells and eosinophils. Lipofuscin-like granules were sometimes present in macrophages or scattered extracellularly in the neuropil. In the cortical layers nearest the malacic areas satellitosis and neurophagia were commonly seen. Foci of green discolouration, grossly visible in the subcortical white matter of some of the horses, proved to consist of perivascular oedema and the intraand extracellular accumulation of lipofuscin-like pigment as described before.126
The most conspicuous lesion in the liver was centrilobular fibrosis that radiated out to form bands between adjacent centrilobular areas. Hepatocytes exhibited fatty changes, multinucleation and large, bizarre, hyperchromatic nuclei and many macrophages contained yellowishbrown pigment.104, 126
Kriek et al.113 provided experimental evidence that LEM-inducing isolates were also toxic to non-equidae and that the lesions were variously sited in the different species. Although certain organs were constantly affected to some degree in all animals, the main target organ was different in each species; for instance, the brain in the horse, the liver and heart in rats, the liver in baboons, the lung in pigs, and the kidney and liver in sheep. These changes included LEM, cirrhosis of the liver, pulmonary oedema and signs of congestive heart failure. For information on naturally occurring outbreaks of lung oedema in pigs induced by F. verticillioides-infected maize screenings in the USA, consult Respiratory system.
A diagnosis of LEM is made on the grounds of nervous signs in horses fed on mouldy maize or maize products, the presence of malacic lesions in the cerebral cortex and toxic levels of fumonisin in the rations. Bilirubinaemia and elevation of aspartate transaminase and γ-glutamyl transferase activity105 in the serum would support such a diagnosis.
Discussion
While convulsions or seizures are easily recognized as being nervous signs, less obvious manifestations of nervous disorders such as changes in temperament, hypersensitivity, or paresis may not be so easy to distinguish from similar signs in non-nervous conditions. The paresis manifested in certain neurotoxicoses, e.g. diplodiosis, can be confused with weakness induced by systemic diseases, aggression may be a normal expression of fear by an animal unable to flee, and hypersensitivity is a subjective concept open to varying interpretations. Nervous signs, therefore, should be treated with circumspection, especially in livestock that are kept under extensive conditions where clinical examination is difficult. Not only can clinical signs be misleading but the clinical signs of various neurotoxicoses may overlap and signs may change drastically during the course of an intoxication, for instance, from excitement to depression as in Sarcostemma viminale poisoning. The making of a diagnosis, furthermore, may be bedevilled by a latent period (in stootsiekte it can be 44 days) during which the offending plant may have died off and disappeared, or during which the affected animals may have been transported out of the area where the toxic plant occurs. Although clinical signs of poisoning may be valuable indicators of the type of plant involved, without corroborative evidence, a diagnosis can seldom be made from them alone.
Stimulation of the CNS, manifested as hypersensitivity, tremors, spasms, tetanic convulsions and nystagmus, occurs among others in albiziosis, cynanchosis, annual ryegrass toxicity (ARGT) and poisoning with S. viminale and Euphorbia mauritanica. In these poisonings, the body temperature is often elevated and rigor mortis sets in almost immediately after death.
Depression of the CNS, variously expressed as apathy, reluctance to move, docility, clumsiness, aimless wandering, pushing, paresis and paralysis, is a feature of stootsiekte, diplodiosis, Helichrysum argyrosphaerum and Trachyandra poisonings, etc. Often, as in cynanchosis or S. viminale poisoning, excitability is followed by depression.
In certain neurotoxicoses the clinical signs can be elicited or exacerbated by exercise, e.g. in ARGT, kweek tremors, Paspalum staggers and Melica decumbens poisoning. Sometimes, too, signs may be precipitated by stimuli, such as fright or forcibly lifting up the head, as in the case of maldronksiekte. With few exceptions, such as cerebellar atrophy in maldronksiekte, leukoencephalomalacia (LEM) in equine Fusarium verticillioides poisoning, and pigmentation of certain areas in the brain as in Trachyandra and Phalaris poisonings, a diagnosis can rarely be made on the macroscopical pathology alone. In contrast, the histopathological lesions in many neurotoxicoses are sufficiently specific to substantiate a diagnosis, e.g. in stootsiekte, maldronksiekte and poisoning with Aspergillus clavatus and Trachyandra spp.
Toxic insult may be directed at various elements in the CNS, e.g. the white matter in LEM, stootsiekte and H. argyrosphaerum poisoning; the neurons in Phalaris staggers, maldronksiekte, valsiekte and poisonings with A. clavatus and Trachyandra spp.; the vasculature, in ARGT as well as, possibly, in stootsiekte and LEM.
In South Africa, Aspergillus clavatus poisoning, Paspalum staggers, kweek tremors and dronkgras and perennial ryegrass staggers are classified as tremorgenic syndromes. However, Aspergillus clavatus poisoning differs from the others in that mortality is high and distinctive lesions are present in the central nervous system. All the remaining conditions involve grasses known or suspected of being infected by either ergots or endophytes. Their clinical signs, such as hypersensitivity, tremors, incoordination and paralysis, are consistent with those induced by indole diterpenes. The lack of mortality, that clinical signs can be elicited or exacerbated by exercise and the persistence of these signs, further incriminate Claviceps paspali or Neotyphodium (= Acremonium) spp. as the probable aetiological agents. Save for A. clavatus poisoning, the tremorgenic disorders in South Africa are almost impossible to differentiate clinically.
Nervous signs may be evident in conditions where the primary lesion is in the liver, e.g. fibrosis or cirrhosis in seneciosis. In chronic hepatic insufficiencies, the accumulation of toxic metabolites and other substances in the circulation may lead to hepatic encephalopathy (Liver).
Owing to the importance of Tribulus terrestris as a forage and toxic plant in the arid karoid areas of South Africa,106 any poisoning induced by this plant, even if it occurs abroad, is potentially significant to the livestock industry. In 1984, Bourke reported a progressive, irreversible, asymmetric, nervous disorder, manifested principally as paresis of the hindquarters, in sheep grazing T. terrestris in Australia.27 Strong evidence was provided that the beta-carboline alkaloids, harmane and norharmane, in T. terrestris, caused the condition. It was suggested that these alkaloids gradually accumulate in tryptamine-associated neurones, where they irreversibly interact with a specific gene DNA sequence.30, 32, 34 Tribulus or Coonabarabran staggers in Australia is of interest because valsiekte in the Karoo is also characterized by paresis of the hindquarters. However, Coonabarabran staggers differs from valsiekte principally in that adult sheep are afflicted, Merinos are frequently involved and significant lesions are not found in the CNS. A similar condition occurs in sheep ingesting Tribulus micrococcus in Australia, with the difference that in this instance the posterior paresis is bilaterally symmetrical and reversible.28 Coonabarabran staggers must always be borne in mind as a differential diagnosis should sheep on T. terrestris develop para- or tetraparesis in the Karoo.
One of the most important nervous disorders of sheep and goats in South Africa is krimpsiekte. Krimpsiekte is basically a paretic syndrome caused by the ingestion of members of the genera Cotyledon, Tylecodon and Kalanchoe (Crassulaceae) that contain cumulative, neurotoxic bufadienolides. Affected animals lag behind the flock. When forced to move, they tire easily and lie down or stand trembling, typically with the feet together, the back arched and the head low. The neck may be twisted (torticollis) and the head may dangle loosely as the animal walks.4, 91, 143,189, 205 Vacuolation of the thalamus has been reported in experimentally induced krimpsiekte.26 The thalamus participates in regulation of motor activity, arising from the cerebral cortex, by relaying cerebellar feedback to the cerebral cortex. This coordinates the intended motor action and lesions in this area may account for some of the locomotory and postural abnormalities seen in this syndrome.26 For a more detailed discussion of krimpsiekte, please consult Cardiovascular system.
Odd incidents of suspected plant-induced stringhalt have occurred in South Africa (D. van Tonder, private practitioner, Wellington, personal communication, 2002; A. de Vos private practitioner, Ladybrand, personal communication, 2003). The outbreaks have been associated with the ingestion of Hypochaeris radicata. Hypochaeris radicata (hairy wild lettuce, flatweed) is a cosmopolitan weed occurring throughout South Africa (Figure 102). There is circumstantial evidence that this plant or fungi associated with this weed induces stringhalt in horses in the United States of America, Australia and New Zealand. This locomotory disorder appears in summer or autumn; usually following a dry summer when the grazing is sparse and the weed is abundantly present in the paddocks.
Although axonal degeneration is present, horses generally recover, albeit over an extended period (several months to years), if removed from the incriminated pastures.71
Hyananche globosa (hyena poison, gifboom, wolwegif) is a small, evergreen tree with a limited distribution in the Vanrhynsdorp and Clanwilliam districts of the Western Cape Province. The sexes are separate and the female plant bears round capsular fruits. In the nineteen-twenties, a toxin, hyananchin, was isolated, mainly from the fruits, which induced strychnine-like convulsive seizures.185
Ingestion of large quantities of Prosopis glandulosa pods (mesquite, Suidwesdoring) has been implicated in excessive rumination, greenish-staining of the lips (Figure 103), continuous chewing and salivation, locally referred to as ‘kougoed’, in Boer goats in the Northern Cape Province.206 The lesions, denervation atrophy of the masseter and other muscles of mastication, are ascribed to neuronal vacuolation of the trigeminal nuclei.199, 206
References
- ACOCKS, J.P.H. 1975. Veld types of South Africa. Memoirs of the Botanical Survey of South Africa, No. 40, 2nd edn. pp. 128.
- ADAMS, R.H. 1982. Cholinergic pharmacology. In: BOOTH, N.H. & McDONALD, L.E. (eds.) Veterinary Pharmacology and Therapeutics. 5th edn. 113–148. Ames: Iowa State University Press.
- ADELAAR, T.F. 1958. Fungus infected and fermented stock feed can be poisonous. Farming in South Africa, 34, 43–44.
- ANDERSON, L.A.P., SCHULTZ, ANITRA, R., JOUBERT, J.P.J., PROZESKY, L., KELLERMAN, T.S., ERASMUS, G.L. & PROCOS, J. 1983. Krimpsiekte and acute cardiac glycoside poisoning in sheep caused by bufadienolides from the plant Kalanchoe lanceolata Forsk. Onderstepoort Journal of Veterinary Research, 50, 295–300.
- ANDREWS, W.H. 1923. The so-called ‘staggers’ or ‘pushing disease’ of cattle in Natal: An intoxication due to the ingestion of Matricaria nigellaefolia DC. Report on Veterinary Research, Union of South Africa, 9 & 10, 121–220.
- APPLING, T.E.H., STEELE, P. & NOTTLE, M.C. 1983. Toxic ferns in Western Australia. Technical Bulletin, No. 63. Department of Agriculture, Western Australia.
- BADIALI, L., ABOU-YOUSSEF, M.H., RADWIN, A.L., HAMDY, F.M. & HILDEBRANDT, P.K. 1968. Moldy corn poisoning as a major cause of encephalomalacia syndrome in Egyptian Equidae. American Journal of Veterinary Research, 29, 2029–2035.
- BAKKER, H.J., DICKSON, J., STEELE, P. & NOTTLE, M.C. 1980. Experimental inductions of ovine polioencephalomalacia. Veterinary Record, 107, 464–466.
- BARNETT, K.C. & WATSON, W.A. 1970. Bright blindness in sheep. A primary retinopathy due to feeding bracken (Pteris aquilina). Research in Veterinary Science, 11, 289–290.
- BASSON, P.A., ADELAAR, T.F., NAUDÉ, T.W. & MINNE, J.A. 1970. Albizia poisoning: Report of the first outbreak and some experimental work in South Africa. Journal of the South African Veterinary Medical Association, 41, 117–130.
- BASSON, P.A., KELLERMAN, T.S., ALBL, P., VON MALTITZ, L.J.F., MILLER, E.S. & WELMAN, WILHELMINA G. 1975. Blindness and encephalopathy caused by Helichrysum argyrosphaerum DC. (Compositae) in sheep and cattle. Onderstepoort Journal of Veterinary Research, 42, 135–148.
- BERGEL, F., MORRISON, A.L., MOSS, A.R., KLEIN, R., RINDERKNECHT, H. & WARD, J.L. 1943. An antibacterial substance from Aspergillus clavatus and Penicillium claviforme and its probable identity with patulin. Nature (London), 152, 720.
- BERRY, P.H. & VOGEL, P. 1982. Toxicity studies of the toxins isolated from annual ryegrass (Lolium rigidum) infected by Corynebacterium rathayi. Australian Journal of Experimental Biology and Medical Science, 60, 129–132.
- BERRY, P.H. & WISE, J.L. 1975. Wimmera ryegrass toxicity in Western Australia. Australian Veterinary Journal, 51, 525–530.
- BERRY, P.H., HOWELL, J. McC. & COOK, R.D. 1980. Morphological changes in the central nervous system of sheep affected with experimental annual ryegrass (Lolium rigidum) toxicity. Journal of Comparative Pathology, 90, 603–617.
- BERRY, P.H., RICHARDS, R.B., HOWELL, J. McC. & COOK, R.D. 1982. Hepatic damage in sheep fed annual ryegrass, Lolium rigidum, parasitised by Anguina agrostis and Corynebacterium rathayi. Research in Veterinary Science, 32, 148–156.
- BERRY, P.H., COOK, R.D., HOWELL, J. McC., WHITE, R.R. & PURCELL, D.A. 1976. Lesions in sheep and guinea-pigs pen fed parasitised annual ryegrass (Lolium rigidum). Australian Veterinary Journal, 52, 540–541.
- BERRY, P.H., HOWELL, J. McC., COOK, R.D., RICHARDS, R.B. & PEET, R.L. 1980. Central nervous system changes in sheep and cattle affected with natural or experimental ryegrass toxicity. Australian Veterinary Journal, 56, 402–403.
- BEZUIDENHOUT, S. CATHERINE, GELDERBLOM, W.C.A., GORST-ALLMAN, C.P., HORAK, R.M., MARASAS, W.F.O., SPITELLER, G. & VLEGGAAR, R. 1988. Structure elucidation of the fumonisins, mycotoxins from Fusarium moniliforme. Journal of the Chemical Society Communications, 743–745.
- BIANCHI, P., CHAIN, E.B., MANTLE, P.G. & TONOLO, A. 1964. Formation of a lysergic acid derivative during the parasitic development of a strain of Claviceps paspali Stevens and Hall. Nature, 202, 312–313.
- BIRD, A.F. & STYNES, B.A. 1977. The morphology of a Corynebacterium sp. parasitic on annual ryegrass. Phytopathology, 67, 828–830.
- BLOOD, D.C., HENDERSON, J.A. & RADOSTITS, O.M. 1979. Veterinary Medicine, 5th edn. Philadelphia: Lea & Febiger.
- BOOTH, C. 1971. The genus Fusarium. Commonwealth Mycological Institute, Kew, England, pp. 237.
- BOTHA, C.J., KELLERMAN, T.S. & FOURIE, N. 1996. A tremorgenic mycotoxicosis caused by Paspalum distichum (L.) infected by Claviceps paspali. Journal of the South African Veterinary Association, 67, 36–37.
- BOTHA, C.J., SCHULTZ, R. ANITRA, VAN DER LUGT, J.J., RETIEF ELIZABETH & LABUSCHAGNE, LEONIE. 1999. Neurotoxicity in calves induced by the plant, Nierembergia hippomanica Miers var. violacea Millán in South Africa. Onderstepoort Journal of Veterinary Research, 66, 237–244.
- BOTHA, C.J., VAN DER LUGT, J.J., ERASMUS, G.L., KELLERMAN, T.S., SCHULTZ, R.A. & VLEGGAAR, R. 1997. Krimpsiekte, associated with thalamic lesions induced by the neurotoxic cardiac glycoside, cotyledoside, isolated from Tylecodon wallichii (Harv.) Toelken subsp. wallichii. Onderstepoort Journal of Veterinary Research, 64, 189–194.
- BOURKE, C.A. 1984. Staggers in sheep associated with the ingestion of Tribulus terrestris. Australian Veterinary Journal, 61, 360–363.
- BOURKE, C.A. & MACFARLANE, J.A. 1985. A transient ataxia of sheep associated with the ingestion of Tribulus micrococcus (yellow vine). Australian Veterinary Journal, 62, 282.
- BOURKE, C.A. 1987. A naturally occurring tunicamycin-like intoxication in pigs eating water damaged wheat. Australian Veterinary Journal, 64, 127–128.
- BOURKE, C.A. 1987. A novel nigrostriatal dopaminergic disorder in sheep affected by Tribulus terrestris staggers. Research in Veterinary Science, 43, 347–350.
- BOURKE, C.A., CARRIGAN, M.J. & DIXON, R.J. 1990. The pathogenesis of the nervous syndrome of Phalaris aquatica toxicity in sheep. Australian Veterinary Journal, 67, 356–358.
- BOURKE, C.A., CARRIGAN, M.J. & DIXON, R.J. 1990. Upper neurone effects in sheep of some beta-carboline alkaloids identified in zygophyllaceous plants. Australian Veterinary Journal, 67, 248–251.
- BOURKE, C.A., CARRIGAN, M.J. & LOVE, S.C.J. 1992. Flood plain staggers, a tunicaminyluracil toxicosis of cattle in New South Wales. Australian Veterinary Journal, 69, 228–229.
- BOURKE, C.A., STEVENS, G.R. & CARRIGAN, M.J. 1992. Locomotor effects in sheep of alkaloids identified in Australian Tribulus terrestris. Australian Veterinary Journal, 69, 163–165.
- BOURKE, C.A. 1994. The clinico-toxicological differentiation of Phalaris spp. toxicity syndromes in ruminants. In: COLEGATE, S.M. & DORLING, P.R. (eds-in-chief) ALLEN, J.G., HUXTABLE, C.R., MEAD, R.J. & PANTER, K.E. (assoc. eds.) Plant-associated toxins: Agricultural, phytochemical and ecological aspects, 523–528: CAB International.
- BOURKE, C.A., CARRIGAN, M.J. & DIXON, R.J. 1988. Experimental evidence that tryptamine alkaloids do not cause Phalaris aquatica sudden death syndrome in sheep. Australian Veterinary Journal, 65, 218–220.
- BRYDEN, W.L., TRENGROVE, C.L., DAVIS, E.O., GIESECKE, P.R. & CURREN, G.C. 1994. Corynetoxicosis of livestock: A nematode-bacterium disease complex associated with different grasses. In: COLGATE, S.M. & DORLING, P.R. (edsin- chief) ALLEN. J.G., HUXTABLE, C.R., MEAD, R.J. & PANTER, K.E. (assoc. eds.) Plant-associated toxins: Agricultural, phytochemical and ecological aspects, 410–414. CAB International.
- BUCK, W.B., HALIBURTON, J.C., THILSTED, J.P., LOCK, T.F. & VERSONDER, R.F. 1979. Equine leucoencephalomalacia: Comparative pathology of naturally occurring and experimental cases. American Association of Veterinary Laboratory Diagnosticians, 22nd Annual Proceedings, 239–258.
- BURTT–DAVY, J. 1912. Botanical investigations into gal lamsiekte. Report on Veterinary Research, Union of South Africa, 2, 181–265.
- BUTLER, T. 1902. Notes on a feeding experiment to produce leucoencephalitis in a horse with positive results. American Veterinary Review, 26, 748–751.
- CHEEKE, P.R. 1995. Endogenous toxins and mycotoxins in forage grasses and their effects on livestock. Journal of American Science, 73, 909–918.
- CHIPPENDALL, LUCY, K.A. 1955. Part 1. A guide to the identification of grasses in South Africa. In: MERIDITH, D. (ed.)The grasses and pastures of South Africa. 1–527, Pretoria: Central News Agency.
- CHIPPENDALL, LUCY, K.A. & COOK, A.D. 1976. Grasses of southern Africa. Salisbury: Collins.
- CLARDY, J., SPRINGER, J.P. BUCHI, G., MATSUO, K. & WIGHTMAN, R. 1975. Tryptoquivaline and tryptoquivalone, two tremorgenic metabolites of Aspergillus clavatus. Journal of the American Chemical Society, 97, 663–665.
- COATES PALGRAVE, K. 1981. Trees of southern Africa. Cape Town: C. Struik (Pty) Ltd.
- COCKRUM, P.A., CULVENOR, C.C.J., EDGAR, J.A., JAGO, M.V., PAYNE,A.L., & BOURKE, C.A. 1988. Toxic tunicaminyluracil antibiotics identified in water-damaged wheat responsible for the death of pigs. Australian Journal of Agricultural Research, 38, 245–253.
- COLE, R.J., DORNER, J.W., LANDSDEN, J.A., COX, R.H., PAPE, C., CUNFER, B., NICHOLSON, S.S. & BEDELL, D.M. 1977. Paspalum staggers: Isolation and identification of tremorgenic metabolites from sclerotia of Claviceps paspali. Journal of Agricultural Food Chemistry, 25, 1197–1201.
- CURSON, H.H. 1928. Some little known South African poisonous plants and their effect on stock. Report on Veterinary Research, Union of South Africa, 13 & 14, 205–229.
- DARVALL, P.M. 1964. Mouldy corn cobs, a danger to cows. Queensland Agricultural Journal, 90, 692–693.
- DE BALOGH, K.K.I.M., DIMANDE, A.P., VAN DER LUGT, J.J., MOLYNEUX, R.J., NAUDÉ, T.W. & WELMAN, W.G. 1999 . A lysosomal storage disease induced by Ipomoea carnea in goats in Mozambique. Journal of Veterinary Diagnostic Investigation, 11, 266–273.
- DIAZ, G.J. & BOERMANS, H.J. 1994. Fumonisin toxicosis in domestic animals: A review. Veterinary and Human Toxicology, 36, 548–555.
- DIENER, U.L., DAVIS, N.D., MORGAN-JONES, G., CUNNINGHAM, J.P., LANGFORD, J.W. & JOHNSON, W.C. 1975. Bermuda grass tremors in cattle in Alabama. Highlight, 22, 12.
- DI MENNA, M.E., MORTIMER, P.H., PRESTIGE, R.A., HAWKES, A.D. & SPROSEN, J.M. 1992. Lolitrem B concentrations, counts of Acremonium lolii hyphae, and the incidence of ryegrass staggers in lambs on plots of A. lolii-infected perennial ryegrass. New Zealand Journal of Agricultural Research, 35, 211.
- DOBEREINER, J., TOKARMIA, C.H., DA COSTA, J.B.D., CAMPOS, J.L.E. & DAYTEL, M. DE S. 1971. ‘Espichamento’, intoxicacao de bovinos pos Solanum malacoxylon, no pantanal de mato grosso. Pesquisa Agropecuaria Prasileira Srie, Veterinaria, 6, 91–117.
- DOMENECH, J., BOCCAS, B., PELLEGRIN, F., LAURENT, D., KOHLER, F., MAGNOL, J. & LAMBERT, C. 1984. Étude de la fusariose du maïs (Fusarium moniliforme) en Nouvelle Calédonie et de la pathologie equine associée: La leucoencéphalomalacia équine. Reveu d’ Élevage et Médicine Vétérinaire des Pays Tropicaux, 37, 253–259.
- DORLING, P.R., HUXTABLE, C.R. & VOGEL, P. 1978. Lysosomal storage in Swainson a spp. toxicosis: An induced mannosidosis. Neuropathology and Applied Neurobiology, 4, 285–295.
- DORLING, P.R., HUXTABLE, C.R. & COLGATE, S.M. 1980. Inhibition of lysosomal -mannosidase by swainsonine, an indolizidine alkaloid isolated from Swainson a canescens. Biochemical Journal, 191, 649–651.
- EDGAR, J.A., FRAHN, J.L., COCKRUM, P.A., ANDERTON, N., JAGO, M.V. & CULVENOR, C.J. 1982. Corynetoxins, causative agents of annual ryegrass toxicity, are identified as tunicamycin-group antibiotics. Chemical Communications, 4, 222–224.
- EDGAR, J.A., COCKRUM, P.L., STEWART, N.A., ANDERTON, N.A. & PAYNE, A.L. 1994. Identification of corynetoxins as the cause of poisoning associated with annual beardgrass (Polypogon monspeliensis (L) Desf) and blown grass (Agrostis avenacea C. Gemelin). In: COLGATE, S.M. & DORLING, P.R. (eds-inchief) ALLEN. J.G., HUXTABLE, C.R., MEAD, R.J. & PANTER, K.E. (assoc. eds.) Plant-associated toxins: Agricultural, phytochemical and ecological aspects, 393–398. CAB International.
- EHRET, W.J., ADELAAR, T.F. & KRIEK, N.P.J. 1968. An outbreak of Claviceps paspali poisoning (Paspalum staggers) in beef cattle. Journal of the South African Veterinary Medical Association, 39, 100–105.
- ERASMUS, G.L. 1983. Cynanchum gifstowwe geïdentifiseer. Newsletter, Onderstepoort Veterinary Research Institute, 1. No. 3, 10.
- EVANS, E.T.R., EVANS, W.C. & ROBERTS, H.E. 1951. Studies on bracken poisoning in the horse. The British Veterinary Journal, 107, 364–371.
- EVANS, W.C. 1964. Bracken poisoning of farm animals. Veterinary Record, 76, 365–369.
- EVANS, W.C., EVANS, I.A., HUMPHREYS, D.J., LEWIN, B., DAVIES, W.E.J. & AXFORD, R.F.E. 1975. Induction of thiamine deficiency in sheep with lesions similar to those of cerebrocortical necrosis. Journal of Comparative Pathology, 85, 253–267.
- FINNIE, J.W. & JAGO., M.V. 1985. Experimental production of annual rye grass toxicity with tunicamycin. Australian Veterinary Journal, 62, 248–249.
- FINNIE, J.W. & MUKHERJEE, T.M. 1986. Ultrastructural changes in cerebral blood vessels of sheep injected with tunicamycin. Journal of Comparative Pathology, 96, 205–216.
- FINNIE, J.W. 1994. Pathogenesis of corynetoxin poisoning. In: COLGATE, S.M. & DORLING, P.R. (eds-in-chief) ALLEN. J.G., HUXTABLE, C.R., MEAD, R.J. & PANTER, K.E. (assoc. eds.) Plant-associated toxins: Agricultural, phytochemical and ecological aspects, 405–409: CAB International.
- FISHER, J.M. 1977. Annual ryegrass toxicity 1976–77. Waite Plant Agricultural Research Institute, Biennial Report, 7–11.
- FLETCHER, L.R. & HARVEY, I.C. 1981. An association of Lolium endophyte with ryegrass staggers. New Zealand Veterinary Journal, 29, 185–186.
- FREDRICKSON, DEBRA E., MANTLE, P.G. 1991. Claviceps africana sp. nov.: The distinctive ergot pathogen of sorghum in Africa. Mycological Research, 95, 101–107.
- GALEY, F. D., HULLINGER, P. J., McCASKILL, J. 1991. Outbreaks of stringhalt in northern California. Veterinary and Human Toxicology, 33, 176–177.
- GALLAGHER, R.T., CAMPBELL, A.G., HAWKES, A.D., HOLLAND, P.T., MCGAVESTON, D.A. PANSIER, E.A. & HARVEY, I.C. 1982. Ryegrass staggers: The presence of lolitrem neurotoxins in perennial ryegrass seed. New Zealand Veterinary Journal, 30, 183–184.
- GALLAGHER, R.T., HAWKES, A.D., STEYN, P.S. & VLEGGAAR, R. 1984. Tremorgenic neurotoxins from perennial ryegrass causing ryegrass staggers disorder of livestock: Structure elucidation of lolitrem B. Journal of the Chemical Society, Chemical Communications, 9, 614–616.
- GALLAGHER, R.T. & HAWKES, A.D. 1986. The potent tremorgenic neurotoxins lolitrem B and aflatrem: A comparison of the tremor response in mice. Experientia, 42, 823–825.
- GANT, D.B. COLE, R.J., VALDES, J.J., ELDEFRAWI, M.E. & ELDEFRAWI, A.T. 1987. Action of tremorgenic mycotoxins on GABAA receptor. Life Sciences, 41, 2207–2214.
- GARDINER, M.R. 1967. Lupinosis. Advances in Veterinary Science, 11, 85–138.
- GELDERBLOM, W.C.A., JASKIEWICZ, K., MARASAS, W.F.O., THIEL, P.G., HORAK, R.M., VLEGGAAR, R. & KRIEK, N.P.J. 1988. Fumonisins-Novel mycotoxins with cancerpromoting activity produced by Fusarium moniliforme. Applied and Environmental Microbiology, 54, 1806–1811.
- GIBBS RUSSELL, G.E. & ELLIS, R.P. 1982. The genus Melica L. (Poaceae) in southern Africa. Bothalia, 14, 37–44.
- GILMOUR, J.S., INGLIS, D.M., ROBB, J. & MACLEAN, M. 1989. A fodder mycotoxicosis of ruminants caused by contamination of a distillery by-product with Aspergillus clavatus. Veterinary Record, 124, 133–135.
- GILRUTH, J.A. 1931. Caustic Vine (Sarcostemma a ustrale) as a poison plant. Journal of Commonwealth of Australia, Council for Scientific and Industrial Research, 58–60.
- GILRUTH, J.A. & MURANE, D. 1931. Sarcostemma a ustrale (Caustic Vine): A plant that is poisonous to stock. Journal of Commonwealth of Australia, Council for Scientific and Industrial Research, 225–231.
- GLINSUKON, T., YUAN, S.S., WIGHTMAN, R., KITAURA, Y., BCHI, G., SHANK, R.C., WOGAN, G.N. & CHRISTENSEN, C.M. 1974. Isolation and purification of cytochalasin E and two tremorgens from Aspergillus clavatus. Plant Foods for Man, 1, 113–119.
- GRANT, C.C., BASSON, P.A. & KIDD, A.B. 1985. Paralysis and lipofuscin-like pigmentation of stock caused by the plant Trachyandra laxa var. laxa. Onderstepoort Journal of Veterinary Research, 52, 255–259.
- GUMMOW, B. & ERASMUS, G.L. 1990. Pyridoxine (a vitamin B6) and its derivative pyridoxal as treatment for Albizia versicolor poisoning in guinea-pigs. Onderstepoort Journal of Veterinary Research, 57, 109–114.
- GUMMOW, B., BASTIANELLO, STELLA, S., LABUSCHAGNE, LEONIE & ERASMUS, G.L. 1992. Experimental Albizia versicolor poisoning in sheep and its successful treatment with pyridoxine hydrochloride. Onderstepoort Journal of Veterinary Research, 59, 111–118.
- HALIBURTON, J.C., VERSONDER, R.F., LOCK, T.F. & BUCK, W.B. 1979. Equine leucoencephalomalacia (ELEM): A study of Fusarium moniliforme as an etiologic agent. Veterinary and Human Toxicology, 21, 348–351.
- HALL, W.T.K. 1964. Plant toxicoses of tropical Australia. Australian Veterinary Journal, 40, 176–182.
- HARTLEY, W.J. 1970. Chronic Phalaris poisoning or Phalaris staggers. In: Effects of poisonous plants on livestock. New York, San Francisco & London: Academic Press, Inc.
- HEENOP, C.H., McCLAREN, N.W. & BOTHA, ANDRIETTA, J. 1981. Diplodia-kopvrot by mielies. Boerdery in Suid-Afrika. Mielies E.5/1981.
- HENDERSON, MAYDA & ANDERSON, J.G. 1966. Common weeds in South Africa. Botanical Survey, Memoir No. 37, Department of Agriculture Technical Services, Republic of South Africa, 266–267.
- HENNING, M.W. 1926. Krimpsiekte. Report of the Director of Veterinary Education and Research, Onderstepoort, 11 & 12, 331–364.
- HENNING, M.W. 1933. Animal diseases in South Africa. South African Agricultural Series, Vol. XII: Central News Agency, Ltd.
- HENNING, O. 1894. Annual Report of the Colonial Veterinary Surgeon of the Cape of Good Hope for 1893, 22–25.
- HOGAN, M.J. & ZIMMERMAN, L.E. 1962. Ophthalmic pathology. 2nd edn. Philadelphia & London: W.B. Saunders Co.
- HUTCHEON, D. 1904. Kraam ziekte or krimp ziekte. Agricultural Journal of the Cape of Cood Hope, 24, 424–426.
- HUXTABLE, C.R. 1994. Some characteristics of neurones which endow vulnerability to chemical toxins. In: COLEGATE, S.M. & DORLING, P.R. (eds-in-chief), ALLEN, J.G., HUXTABLE, C.R., MEAD, R.J. & PANTER, K.E. (assoc. eds.) Plant-associated toxins: Agricultural, phytochemical and ecological aspects, 357–362. CAB International.
- IWANOFF, X., YUAN, CHANG-KUO & FANG SHIHCHIEH. 1957. Uber die toxishe Enzephalomalazie (Mouldy corn poisoning) der Einhufer in China. Archiv für experimentelle Veterinarmedizin, 11, 1033–1056.
- JACQUET, J., BOUTIBONNES, P. & ACILE, J.P. 1963. Observations sur la toxicite d’Aspergillus clavatus pour les animaux. Bulletin de L’Academie Veterinaire de France, 36, 199–207.
- JAGO, M.V., PAYNE, A. A.E., PETERSON, J.E. & BAGUST, T.J. 1983. Inhibition of glycosylation by corynetoxin, the causative agent of annual ryegrass toxicity: A comparison with tunicamycin. Chemical–Biological Interactions, 45, 223–344.
- JIANG, C.S., HUANG, S.S., CHEN, M.Z., YU, Z., CHEN, L.H. & SANG, Y.Z. 1982. Studies on mycotoxicosis in dairy cattle caused by Aspergillus clavatus from mouldy malt sprouts. Acta Veterinaria et Zootechnica Sinica, 13, 247–254.
- JOLLY, R.D. & HARTLEY, W.J. 1977. Storage diseases of domestic animals. Australian Veterinary Journal, 53, 1–8.
- KEELER, R.F. & CROWE, M.W. 1984. Teratogenicity and toxicity of wild tree tobacco, Nicotiana glauca in sheep. Cornell Veterinarian, 76, 50–59.
- KEELER, R.F., CRONIN, E.H. & SHUPE, J.L. 1976. Lupin alkaloids from teratogenic and non-teratogenic lupins. IV. Concentration of the total alkaloids, individual major alkaloids, and the teratogen anagyrine as a function of plant parts and stage of growth and their relationship to crooked calf disease. Journal of Toxicology and Environmental Health, 1, 899–908.
- KELLERMAN, T.S., MARASAS, W.F.O., PIENAAR, J.G. & NAUDÉ, T.W. 1972. A mycotoxicosis of Equidae caused by Fusarium moniliforme Sheldon. A preliminary communication. Onderstepoort Journal of Veterinary Research, 39, 205–208
- KELLERMAN, T.S., MARASAS, W.F.O., THIEL, P.G., GELDERBLOM, W.C.A., CAWOOD, M. & COETZER, J.A.W. 1990. Leukoencephalomalacia in two horses induced by oral dosing of fumonisin B1. Onderstepoort Journal of Veterinary Research, 57, 269–275.
- KELLERMAN, T.S., NAUDÉ, T.W. & FOURIE, N. 1996. The distribution, diagnosis and estimated economic impact of plant poisonings and mycotoxicoses in South Africa. Onderstepoort Journal of Veterinary Research, 63, 65–90.
- KELLERMAN, T.S., NEWSHOLME, S.J., COETZER, J.A.W. & VAN DER WESTHUIZEN, G.C.A. 1984. A tremorgenic mycotoxicosis of cattle caused by maize sprouts infested by Aspergillus clavatus. Onderstepoort Journal of Veterinary Research, 51, 271–274.
- KELLERMAN, T.S., PIENAAR, J.G., VAN DER WESTHUIZEN, G.C.A., ANDERSON, L.A.P. & NAUDÉ, T.W. 1976. A highly fatal tremorgenic mycotoxicosis of cattle caused by Aspergillus clavatus. Onderstepoort Journal of Veterinary Research, 43, 147–154.
- KELLERMAN, T.S., PROZESKY, L., SCHULTZ, R. ANITRA, RABIE, C.J., VAN ARK, H., MAARTENS, B.P. & LÜBBEN, ANNALIE. 1991. Perinatal mortality in lambs of ewes exposed to cultures of Diplodia maydis (= Stenocarpella maydis) during gestation. Onderstepoort Journal of Veterinary Research, 58, 297–308.
- KELLERMAN, T.S., PROZESKY, L., SCHULTZ, R. ANITRA & RABIE, C.J. 1993. Neuromycotoxicoses of livestock on pastures in South Africa, with special reference to diplodiosis. Proceedings of a symposium on ‘Mycotoxicoses of Grassland Farming’, Hamilton, New Zealand, 22–24 September 1993. New Zealand Veterinary Journal, 41, 212–213.
- KELLERMAN, T.S., RABIE, C.J., VAN DER WESTHUIZEN, G.C.A., KRIEK, N.P.J. & PROZESKY, L. 1985. Induction of the neuromycotoxicosis, diplodiosis, in domestic ruminants with cultures of indigenous and exotic isolates of Diplodia maydis. Onderstepoort Journal of Veterinary Research, 52, 35–42.
- KINGSBURY, J.M. 1964. Poisonous plants of the United States and Canada. Englewood Cliffs, New Jersey: Prentice-Hall Inc.
- KRIEK, N.P.J., KELLERMAN, T.S. & MARASAS, W.F.O.1981. A comparative study of the toxicity of Fusarium verticilloides (= F. moniliforme) to horses, primates, pigs, sheep and rats. Onderstepoort Journal of Veterinary Research, 48, 129–131.
- LANIGAN, G.W., PAYNE, A.E. & FRAHN, J.L. 1976. Origin of toxicity in parasitised annual ryegrass (Lolium rigidum). Australian Veterinary Journal, 52, 244–246.
- LATCH, G.C.M., & CHRISTENSEN, M.J. 1982. Ryegrass endophyte, incidence and control. New Zealand Journal of Agricultural Research, 25, 443–448.
- LATCH, G.C.M. 1985. Endophytes in ryegrass staggers. In: LACEY, J. (ed.) Tri chothecenes and other mycotoxins. 135–140. New York: John Wiley & Sons.
- LAWS, L. & ANSON, R.B. 1968. Neuropathy in sheep fed Swainsona luteola and S. galegifolia. Australian Veterinary Journal, 44, 147–152.
- LEHANE, L. 1982. Annual ryegrass toxins exposed. Rural Research, Winter, 29–30.
- MANTLE, P.G. 1979. The genus Claviceps. In: WYLLIE, T.D. & MOREHOUSE, L.G. (eds.) Mycotoxic fungi, mycotoxins, mycotoxicoses. An encyclopedic handbook. Vol. 1. 83–88. New York: Marcel Decker, Inc.
- MANTLE, P.G. 1978. Ergotism in cattle. In: WYLLIE, T.D. & MOREHOUSE, L.D. (eds.) Mycotoxic fungi, mycotoxins, mycotoxicoses. An encyclopedic handbook. Vol. 11. 145–151. New York: Marcel Decker, Inc.
- MANTLE, P.G., MORTIMER, P.H. & WHITE, E.P. 1977. Mycotoxic tremorgens of Claviceps paspali and Penicillum cyclopium: A comparative study of effects on sheep and cattle in relation to natural stagger syndromes. Research in Veterinary Science, 24, 49–56.
- MANTLE, P.G. & PENN, J. 1989. A role for paxilline in the biosynthesis of indole–diterpenoid penitrem mycotoxins. Journal of the Chemical Society, Perkin Transactions, 18, 1539–1540.
- MARASAS, W.F.O. 1977. The genus Diplodia. In: WYLLIE, T.D. & MOREHOUSE, L.G. (eds.) Mycotoxic fungi, mycotoxins, mycotoxicoses. An encyclopedic handbook. Vol. 1. 119–128. New York: Marcel Decker, Inc.
- MARASAS, W.F.O. 1978. Diplodiosis in cattle. In: WYLLIE, T.D. & MOREHOUSE, L.G. (eds.) Mycotoxic fungi, mycotoxins, mycotoxicoses. An encyclopedic handbook. Vol. 11. 163–165. New York: Marcel Decker, Inc.
- MARASAS, W.F.O. 1978. Diplodiosis in sheep. In: WYLLIE, T.D. & MOREHOUSE, L.G. (eds.) Mycotoxic fungi, mycotoxins, mycotoxicoses. An encyclopedic handbook. Vol. 11. 218–219. New York: Marcel Decker, Inc.
- MARASAS, W.F.O., KELLERMAN, T.S., PIENAAR, J.G. & NAUDÉ, T.W. 1976. Leukoencephalomalacia: A mycotoxicosis of Equidae caused by Fusarium moniliforme Sheldon. Onderstepoort Journal of Veterinary Research, 43, 113–122.
- MARASAS, W.F.O., KELLERMAN, T.S., GELDERBLOM, W.C.A., COETZER, J.A.W., THIEL, P.G. & VAN DER LUGT, J.J. 1988. Leukoencephalomalacia in a horse induced by fumonisin B1 isolated from Fusarium moniliforme. Onderstepoort Journal of Veterinary Research, 55, 197–203.
- MARLOTH, R. 1913. Presidential address, Eighth Annual General Meeting of the Cape Chemical Society, Cape Town, 30th May, 1913.
- McINTOSH, G.H. & THOMAS, MARY R. 1967. Toxicity of parasitised Wimmera ryegrass, Lolium rigidum, for sheep and cattle. Australian Veterinary Journal, 43, 349–353.
- MENZIES, J.S., BRIDGES, C.H. & BAILEY, E.M. 1979. A neurological disease of cattle associated with Solanum dimidiatum. The Southwestern Veterinarian, 32, 45–49.
- MEYER, P. 1989. Thiaminase activity and thiamine content of Pteridium aquilinum, Equisetum ramosissimum, Malva parviflora, Pennisetum clandestinum and Medicago sativa. Onderstepoort Journal of Veterinary Research, 56, 145–146.
- MICHELMORE, A., MACKAY, A. & MACKIE, D. 1980. Annual ryegrass toxicity. Department of Agriculture, Fact Sheet No.91/77.
- MINCUINA, V., MITROIV, P., DONTA, C. 1977. Severe intoxication in cows due to ingestion of mouldy malt sprouts. Lucrarile Institutuliu de Cercetari Veterinare si Biopreparate ‘Pasteur’, 13, 81–84.
- MITCHELL, D.T. 1918. The effects produced on cattle by feeding on Paspalum dilatatum infected with a species of ergot Claviceps paspali. Report on Veterinary Research, Union of South Africa, 7 & 8, 441–449.
- MITCHELL, D.T. 1918. A condition produced in cattle feeding on maize infected with Diplodia zeae. Report on Veterinary Research, Union of South Africa, 7 & 8, 425–437.
- MITCHELL, D.T., 1919. Poisoning of cattle by Diplodia-infected maize. South African Journal of Science, 16, 446–452.
- MITCHELL, D.T. 1920. Poisoning of cattle by feeding on ergotized Paspalum. Journal of the Department of Agriculture, Union of South Africa, 20, 3–7.
- MOLYNEUX, R.J., LYNN, J.F., RALPHS, M.H., PFISTER, J.A., PANTER, K.E. & NASH, J.R. 1994. Polyhydroxy alkaloid glycosidase inhibitors from poisonous plants of global distribution: Analysis and identification. In: COLEGATE, S.M. & DORLING, P.R. (eds-in-chief), ALLEN, J.G., HUXTABLE, C.R., MEAD, R.J. & PANTER, K.E. (assoc. eds.) Plant-associated toxins: Agricultural, phytochemical and ecological aspects, 107–112. CAB International.
- MOLYNEUX, R.J., MCKENZIE, R.A., O’SULLIVAN, B.M. & ALBEIN, A.D. 1995. Identification of the glycosidase inhibitors swainsonine and calystegine B2 in weir vine (Ipomoea sp. Q6[aff. Calobra]) and correlation with toxicity. Journal of Natural Products, 58, 878–886.
- MOREAU, C. & MOREAU, M. 1960. Un danger pour le bétail nourri de plantules fourragéres cultivées en germoir; la pollution d’une moisissure toxique, l’Aspergillus clavatus cause des accidents mortels. Compte rendu de l’Academie d’Agriculture de France, 46, 441–445.
- MORTIMER, P.H. 1983. Ryegrass staggers: Clinical, pathological and aetiological aspects. Proceedings of the New Zealand Grassland Association, 44, 230–233.
- MYBURGH, J.G., FOURIE, N., VAN DER LUGT, J.J., KELLERMAN, T.S., CORNELIUS, S.T. & WARD, CAROLINE. 1994. A nervous disorder in cattle caused by the plants Ficus ingens var. ingens and Ficus cordata subsp. salicifolia. Onderstepoort Journal of Veterinary Research, 61, 171–176.
- NAUDÉ, T.W. 1977. The occurrence and significance of South African cardiac glycosides. Journal of the South African Biological Society, 18, 7–20.
- NEEDHAM, A.J.E. & LAWRENCE, J.A. 1966. The toxicity of Albizia versicolor. Rhodesian Agricultural Journal, 63, 137–140.
- NEILL, J.C. 1940. The endophyte of ryegrass (Lolium perenne). New Zealand Journal of Science and Technology, A23, 185–193.
- NEWSHOLME, S.J. & KELLERMAN, T.S. 1984. Resistance of sheep to poisoning by the plant Matricaria nigellifolia DC. Onderstepoort Journal of Veterinary Research, 51, 277–278.
- NEWSHOLME, S.J., KELLERMAN, T.S. & WELMAN, WILHELMINA G. 1984. Pathology of a nervous disorder (pushing disease or ‘stootsiekte’) in cattle caused by the plant Matricaria nigellifolia DC. (Asteraceae). Onderstepoort Journal of Veterinary Research, 51, 119–127.
- NEWSHOLME, S.J., SCHNEIDER, D.J. & REID, CLARE. 1985. A suspected lipofuscin storage disease of sheep associated with ingestion of the plant, Trachyandra divaricata (Jacq.) Kunth. Onderstepoort Journal of Veterinary Research, 52, 87–92.
- NORRIS, R.T., RICHARDS, I.S. & PETTERSON, D.S. 1981. Treatment of ovine annual ryegrass toxicity with chlordiazepoxide: A field evaluation. Australian Veterinary Journal, 57, 302–303.
- O’BRIEN, J. 1970. Toxicological aspects of some modem anthelmintics. Australian Veterinary Journal, 46, 297–300.
- OEHME, F.W., BAILIE, W.E. & HUBERT, L.C. 1968. Astragulus mollissimus (Locoweed) toxicosis of horses in Western Kansas. Journal of the American Veterinary Medical Association, 152, 271–278.
- OELRICHS, P.B., HILL, M.W., VALLEL, P.J., MACLEOD, J.K. & MOLINSKI, T.F. 1985. The chemistry and pathology of meliatoxins A and B constituents from the fruit of Melia azedarach L. var. australasica. Proceedings of a combined Australia–USA Poisonous Plants Symposium, Brisbane, May 14–18, 1984.
- PAMUKCU, A.M., PRICE, J.M. & BRYAN, G.T. 1976. Naturally occurring and bracken-fern-induced bovine urinary bladder tumors. Veterinary Pathology, 130, 110–122.
- PHILIP, J.R., JACKSON, J.J. & SHONE, D.K. 1958. Sarcostemma viminale poisoning in sheep and cattle. Journal of the South African Veterinary Medical Association, 29, 319–320.
- PIENAAR, J.G., KELLERMAN, T.S. & MARASAS, W.F.O. 1981. Field outbreaks of leukoencephalomalacia in horses consuming maize infected by Fusarium verticilloides (= F. moniliforme) in South Africa. Journal of the South African Veterinary Association, 52, 21–24.
- PIENAAR, J.G., KELLERMAN, T.S., BASSON, P.A., JENKINS, W.L. & VAHRMEIJER, J. 1976. Maldronksiekte in cattle: A neuropathy caused by Solanum kwebense N.E. Br. Onderstepoort Journal of Veterinary Research, 43, 67–74.
- PIENAAR, J.G., KRIEK, N.P.J., NAUDÉ, T.W., ADELAAR, T.F. & ELLIS, SHIRLEY, D. 1973. Lesions in sheep skeletal and oesophageal muscle in vermeersiekte (Geigeria ornativa O. Hoffm. poisoning). Onderstepoort Journal of Veterinary Research, 40, 127–138.
- PRICE, P.C., FISHER, J.M. & KERR, A. 1979. An Anguina funesta n.sp. and its association with Corynebacterium sp. in infecting Lolium rigidum. Nemalogica, 25, 76–85.
- PROZESKY, L. & PIENAAR, J.G. 1977. Amaurosis in sheep resulting from treatment with rafoxanide. Onderstepoort Journal of Veterinary Research, 44, 257–260.
- PROZESKY, L., KELLERMAN, T.S., SWART, D. PETRO, MAARTENS B.P. & SCHULTZ, R. ANITRA. 1994. Perinatal mortality in lambs exposed to cultures of Diplodia maydis (= Stenocarella maydis) during gestation. A pathological study of the central nervous system lesions. Onderstepoort Journal of Veterinary Research, 61, 247–253.
- QUINLAN, J. 1956. A note on the occurrence of acute ergot (Claviceps purpurea) poisoning in steers. Journal of the South African Veterinary Medical Association, 27, 113.
- RABIE, C.J., VAN RENSBURG, S.J., KRIEK, N.P.J. & LUBBEN, ANNELIE. 1977. Toxicity of Diplodia maydis to laboratory animals. Applied and Environmental Microbiology, 34, 111–114.
- RICHARDS, I.S. 1982. The effect of magnesium sulphate on convulsions induced by annual ryegrass toxicity. Australian Veterinary Journal, 58, 115–116.
- RICHARDS, I.S., PETTERSON, D.S. & PURCELL, D.A. 1979. Treatment of ovine annual ryegrass toxicity with chlordiazepoxide. Australian Veterinary Journal, 55, 383.
- RIET-CORREA, F., DEL CARMEN MENDEZ, M., SCHILD, A.L., SUMMERS, B.A. & OLIVEIRA, J.A. 1983. Intoxication by Solanum fastigiatum var. fastigiatum as a cause of cerebellar degeneration in cattle. Cornell Veterinarian, 73, 240–256.
- RODRIGUEZ, J.A. 1945. Diferenciation entre la enformedad de los rastrojos y la meningo encefalomielitis enfecciosa de los equinos. Anales de la Sociedad Rural Argentina, 69, 305–307.
- ROSS P.F., LEDET, A.E. OWENS, D.L., RICE, L.G., NELSON, H.A., OSWEILER, G.D. & WILSON, T.M. 1993. Experimental equine leukoencephalomalacia, toxic hepatosis, and encephalopathy caused by corn naturally contaminated with fumonisins. Journal of Veterinary Diagnostic Investigation, 36, 548–555.
- ROSS, P.F., RICE, L.G., PLATTNER, R.D., OSWEILER, G.D., WILSON, T.M.. OWENS, D.L., NELSON, H.A. & RICHARD, J.L. 1991. Concentrations of fumonisin B1 in feeds associated with animal health problems. Mycologia, 114, 129–135.
- ROWAN, D.D. & GAYNOR, D.L. 1986. Isolation of feeding deterrents against Argentine stem weevil from ryegrass infected with the endophyte Acremonium lolii. Journal of Chemical Ecology, 12, 647–658.
- RUTENBURG, A.M., KIM, WHAKYU, FISCHBEIN, J.W., HANKER, J.S., WASSERKRUG, HANNAH L. & SELIGMAN, A.M. 1969. Histochemical and ultrastructural demonstration of γ-glutamyl transpeptidase activity. Journal of Histochemistry and Cytochemistry, 17, 517–526.
- SCHNEIDER, D.J. 1981. First report of annual ryegrass toxicity in the Republic of South Africa. Onderstepoort Journal of Veterinary Research, 48, 251–255.
- SCHULMAN, M.L. & BOLTON, LORNA A. 1998. Datura seed intoxication in two horses. Journal of the South African Veterinary Association, 69, 27–29.
- SCHWARTE, L.R., BIESTER, H.E. & MURRAY, C. 1937. A disease of horses caused by feeding mouldy corn. Journal of the American Veterinary Association, 90, 76–85.
- SEKHON, S.S. & MAXWELL, D.S. 1974. Ultrastructural changes in neurones of the spinal anterior horn of ageing mice with particular reference to the accumulation of lipofuscin pigment. Journal of Neurocytology, 3, 59–72.
- SHEPHARD, G.S., SYDENHAM, E.W., THIEL, P.G. & GELDERBLOM, W.C.A. 1990. Quantitative determination of fumonisin B1 and B2 by high-performance liquid chromatography with fluorescence detection. Journal of Liquid Chromatography, 13, 2077–2087.
- SHLOSBERG, A., ZADIKOV, I., PERL, S., YAKOBSON, Y., VAROD, Y., ELAD, D., RAPOPORT, E. & HANDJI, V. 1991. Aspergillus clavatus as probable cause of a lethal mass neurotoxicosis in sheep. Mycopathologia, 114, 35–39.
- SHLOSBERG, A., ZADIKOV, I., PERL, S., YAKOBSON, Y., ELAD, D. & PAPOPORT, E. 1992. A lethal mass neurotoxicosis in sheep caused by Aspergillus clavatus contaminated feed. In: JAMES, L.F., KEELER, R.F., BAILEY, E.M., CHEEKE, P.R. & MEGARTY, M.P. 1992. Poisonous Plants, Proceedings of the Third International Symposium, 397–402. Ames, Iowa: Iowa State University Press.
- SHONE, D.K. & DRUMMOND, R.B. 1965. Poisonous plants of Rhodesia. Rhodesian Agricultural Journal, 62, 7–64.
- SHUPE, J.L., BINNS, W., JAMES, L.F. & KEELER, R.F. 1967. Lupine, a cause of crooked calf disease. Journal of the American Veterinary Medical Association, 151, 198–203.
- SOLDAN, A.W., VAN INZEN, C. & EDELSTEN, R.M. 1996. Albizia versicolor poisoning of sheep and goats in Malawi. Journal of the South African Veterinary Association, 67, 217–221.
- STENT, S.M. 1931. Some poisonous plants of Southern Rhodesia. Rhodesian Agricultural Journal, 28, 811–821.
- STEWART, P.L. & MAY, C. 1994. Liver GlcNAc-1-phosphate: Dilicol phosphate GlcNAc-1-phosphate transferase activity as an indicator of ARGT. In: COLGATE, S.M. & DORLING, P.R. (eds-in-chief), ALLEN. J.G., HUXTABLE, C.R., MEAD, R.J. & PANTER, K.E. (assoc. eds). Plant-associated toxins: Agricultural, phytochemical and ecological aspects, 149–154. CAB International.
- STEYN, D.G. 1931. Recent investigations into the toxicity of plants. Journal of the South African Veterinary Association, 2, 116.
- STEYN, D.G. 1932. Investigations into the toxicity of known and unknown poisonous plants in the Union of South Africa. Report on Veterinary Research, Union of South Africa, 18, 871–891.
- STEYN, D.G. 1934. The toxicology of plants in South Africa. Central News Agency: South Africa.
- STEYN, D.G. 1937. Recent investigations into the toxicity of known and unknown poisonous plants in the Union of South Africa. VII. Onderstepoort Journal of Veterinary Science and Animal Industry, 9, 111–124.
- STEYN, D.G. 1939. Recent investigations into the toxicity of known and unknown poisonous plants in the Union of South Africa, IX. Onderstepoort Journal of Veterinary Science and Animal Industry, 12, 335–366.
- STEYN. D.G. 1942. Seringbessie-vergiftiging by vee. Boerdery in Suid-Afrika, 5, 1–3.
- STEYN, D.G. 1949. Vergiftiging van mens en dier. Pretoria: Van Schaik.
- STEYN, D.G. & RINDL, M. 1928–29. Preliminary report on the toxicity of the fruit of Melia azedarach (Syringa berries). Transactions of the Royal Society of South Africa, 17, 295–308.
- STEYN, D.G., WESSELS, P.L., HOLZAPFEL, C.W., POTGIETER, D.J.J. & LOUW, W.K.A. 1972. The isolation and structure of a toxic metabolite from Diplodia maydis (Berk.) Sacc. Tetrahedron, 28, 4775–4785.
- STEYN, P.S., VLEGGAAR, R. & ANDERSON, L.A.P. 1987. Structure elucidation of two neurotoxins from Albizia tanganyicensis. South African Journal of Chemistry, 40, 191–192.
- STEYN, P.S., VAN HEERDEN, FANIE R., VLEGGAAR, R. ERASMUS, G.L. & ANDERSON, L.A.P. 1989. Toxic constituents of the Asclepiadaceae. Structure elucidation of the cynafosides, toxic pregnane glycosides of Cynanchum africanum R. Br. South African Journal of Chemistry, 42, 27–29.
- STYNES, B.A. 1980. A study of annual ryegrass toxicity. Report on work done during the tenure of a Reserve Bank of Australia Research Fellowship at CSIRO. Division of Horticultural Research. Adelaide, (May 1979 to May 1980). Western Australian Department of Agriculture.
- STYNES, B.A. & PETTERSON, D.S. 1980. Production of a neurotoxin in a cell culture of endosperm from Lolium multiflorum colonised by Corynebacterium rathayi. Physiological Plant Pathology, 16, 163–167.
- STYNES, B.A., PETTERSON, D.S., LLOYD, J., PAYNE, A.L. & LANIGAN, G.W. 1979. The production of toxin in annual ryegrass, Lolium rigidum, infected with a nematode Anguina sp., and Corynebacterium rathayi. Australian Journal of Agricultural Research, 30, 201–209.
- SUZUKI, T., TAKEDA, M. & TANABE, H. 1971. A new mycotoxin produced by Aspergillus clavatus. Chemical and Pharmaceutical Bulletin, 19, 1786–1788.
- SYDENHAM, E.C., MARASAS, W.F.O., SHEPHARD, G.S., THIEL, P.G. & HIROOKA, E.Y. 1992. Fumonisin concentrations in Brazilian feeds associated with field outbreaks of confirmed and suspected animal mycotoxicoses. Journal of food and Agricultural Chemistry, 40, 994–997.
- TABOSA, I.M., SOUZA, J.C., GRAÇA, D.L., BARBOSAFILHO, J.M., ALMEIDA, R.N. & RIET-CORREA, F. 2000. Neuronal vacuolation of the Trigeminal nuclei in goats caused by ingestion of Prosopis juliflora pods (mesquite beans). Veterinary and Human Toxicology, 42, 155–158.
- TERBLANCHE, M. & VAN STRATEN, A.M.S. 1966. Further studies on Sarcostemma viminale R. Br. poisoning. Journal of the South African Veterinary Medical Association, 37, 317–319.
- TERBLANCHE, M., ADELAAR, T.F. & VAN STRATEN, A.M.S. 1966. Euphorbia mauritanica L. as a poisonous plant in South Africa. Journal of the South African Veterinary Medical Association, 37, 311–315.
- THEILER, A. 1927. Die Diplodiosis der Rinder und Schafe in Süd-Afrika. Deutschen Tierärztlichen Wochenschrift, 35, 395–399.
- THIEL, P.G., SHEPHARD, G.S., SYDENHAM, E.W., MARASAS, W.F.O., NELSON, P.E. & WILSON, T.M. 1991. Levels of fumonisin B1 and B2 in feeds associated with confirmed cases of equine leukoencephalomalacia. Journal of Agricultural Food Chemistry, 39, 109–111.
- TUSTIN, R.C., ADELAAR, T.F. & MELDAL-JOHNSEN, C.M. 1968. Bracken poisoning in cattle in the Natal Midlands. Journal of the South African Veterinary Medical Association, 39, 91–99.
- VAHRMEIJER, J. 1981. Poisonous plants of Southern Africa. Cape Town: Tafelberg.
- VAN AARDT, M.P. & DU PLESSIS, E.C. 2002. Poisoning of Boergoats by the ingestion of pods from Prosopis glandulosa trees. Proceedings of the Annual Congress of the Livestock Health and Production Group of the South African Veterinary Association, Wilderness, May 2002, 117–120.
- VAN DER BIJL, P.A. 1916. A study of ‘dry-rot’ disease of maize. Bulletin of the Department of Agriculture, Science Bulletin, 7, Pretoria.
- VAN DER LUGT, J.J. 2002 The clinicopathology and pathology of selective toxicoses and storage diseases of the nervous system of ruminants in southern Africa. PhD Thesis, Utrecht University.
- VAN DER LUGT, J.J., OLIVIER, J. & JORDAAN, P. 1996. Status spongiosus, optic neuropathy, and retinal degeneration in Helichrysum argyrosphaerum poisoning in sheep and a goat. Veterinary Pathology, 33, 495–502.
- VAN DER VYVER, F.H., KELLERMAN, T.S., BASTIANELLO, STELLA S., DE WET, J.A.L., JOUBERT, J.P.J. & FAUL, ADELE. 1985. Valsiekte (Falling disease): A nervous disorder in lambs suspected of being caused by the plant Chrysocoma tenuifolia. Journal of the South African Veterinary Association, 56, 65–68.
- VAN DER WALT, S.J. & STEYN, D.G. 1934. Recent investigations into the toxicity of plants. XIII. Onderstepoort Journal of Veterinary Science and Animal Industry, 21, 45–55.
- VAN HALDEREN, A., GREEN, JENNIFER R. & SCHNEIDER, D.J. 1990. An outbreak of suspected Phalaris staggers in the Western Cape Province. Journal of the South African Veterinary Association, 61, 39–40.
- VAN KAMPEN, K.R. & JAMES, L.F. 1969. Pathology of locoweed poisoning in sheep. Pathologica Veterinaria, 6, 413–423.
- VLEGGAAR, R., VAN HEERDEN, FANIE R., ANDERSON, L.A.P. & ERASMUS, G.L. 1993. Toxic costituents of the Asclepiadaceae. Structure elucidation of sarcovimiside A-C, pregnane glycosides of Sarcostemma viminale. Journal of the Chemical Society, Perkin Transactions, 1, 483–487.
- VOGEL, P., PETTERSON, D.S., BERRY, P.H., FRAHN, J.L., ANDERTON, N., COCKRUM, P.A., EDGAR, J.A., JAGO, M.V., LANIGAN, G.W., PAYNE, A.L. & CULVENOR, C.J. 1981. Isolation of a group of glycolipid toxins from seedheads of annual ryegrass, Lolium rigidum GAUD., infected by Corynebacterium rathayi. Australian Journal of Experimental Biology and Medical Science, 59, 455–467.
- WALSH, L.H. 1909. South African poisonous plants. Cape Town: L. Maskew Miller.
- WANG, E., NORRED, W.P., BACON, C.W., RILEY, R.T. & MERRIL, A.H. 1991. Inhibition of sphingolipid biosynthesis by fumonisins: Implications for diseases associated with Fusarium moniliforme. Journal of Biological Chemistry, 266, 14486–14490.
- WANG, E., ROSS,P.F., WILSON, T.M., RILEY, R.T. & MERRIL, A.H. 1992. Increases in serum sphinganine and sphingosine and decreases in complex sphingolipids in ponies given feed containing fumonisins, mycotoxins produced by Fusarium moniliforme. Journal of Nutrition, 122, 1706–1716.
- WATSON, W.A., BARNETT, K.C. & TERLECKI, S. 1972. Progressive retinal degeneration (bright blindness) in sheep: A review. Veterinary Record, 91, 665.
- WATT, J.M. & BREYER–BRANDWIJK, M.G. 1962. The Medicinal and Poisonous Plants of Southern and Eastern Africa. 2nd. edn. Edinburgh and London: E & S Livingstone Ltd.
- WHITE, J.F. 1987. Widespread distribution of endophytes in the Poaceae. Plant Disease, 71, 340–342.
- WHITE, J.F. & HALISKY, P.M. 1992. Association of systematic fungal endophytes with stock poisoning in grasses. In: JAMES, L.F., KEELER, R.F., BAILEY, E.M., CHEEKE, P.R. & MEGARTY, M.P. 1992. Poisonous Plants, Proceedings of the Third International Symposium, 574–578. Ames, Iowa: Iowa State University Press.
- WILSON, B.J. & MARONPOT, R.R. 1971. Causative fungal agent of leucoencephalomalacia in equine animals. Veterinary Record, 88, 484–486.
- WILSON, B.J.R. & MARONPOT, R.R. 1971. Causative fungal agent of leucoencephalomalacia. Journal of the American Veterinary Medical Association, 163, 1293–1295.
- WILSON, T.M., ROSS, P.F., RICE, L.G., OSWEILER, G.D., NELSON, H.A., OWENS, DEBRA L., PLATTNER, R.D., REGGIARDIO, C., NOON, T.H. & PICKRELL, J.W. 1990. Fumonisin B1 levels associated with an epizootic of equine leukoencephalomalacia. Journal of Veterinary Diagnostic Investigation, 2, 213–216.
- YOO, H–S., NORRED, W.P., WANG, E., MERRIL, A.H. & RILEY, R.T. 1992. Fumonisin inhibition of de novo sphingolipid biosynthesis and cytotoxicity are correlated in LLC–PK1 cells. Toxicology and Applied Pharmacology, 114, 9–15.