This content is distributed under the following licence: Attribution-NonCommercial CC BY-NC  View Creative Commons Licence details here
View Creative Commons Licence details here

Introduction
In southern Africa, there are relatively few non-infectious conditions of plant and fungal origin that affect the skin and adnexa of livestock. The integument can be injured by contact irritants, such as the latex of Euphorbia spp.; systemic intoxications which also involve the skin, as in Vicia poisoning; and mechanical injury by sharp-pointed grass or other seeds. Two toxic plants, Crotalaria burkeana and Chrysocoma ciliata (= C. tenuifolia), although lacking contact irritants or overtly systemic poisons, selectively affect certain components of the skin. For instance, Crotalaria burkeana specifically causes abnormal hoof growth in cattle, and the unidentified toxin of Chrysocoma ciliata in the milk of sheep and goats is responsible for alopecia in newborn lambs and kids.
Please note that the most important plant or mycotoxin-induced dermatitis of stock on the subcontinent, hepatogenous photosensitivity, is dealt with in Liver.
Crotalaria spp. (Fabaceae)
C. burkeana Benth.
Rattle bush, stywesiektebossie
C. barkae Schweinf. (= C. geminiflom Dinter ex Bak. f.)
C. juncea L.
Sunn hemp, Sunnhennep
In South Africa, the Afrikaans term stywesiekte, literally translated as stiffsickness, is used to describe diseases in cattle with diverse aetiology. The term has been coined for an aphosphorosis syndrome,22 a viral disease referred to as drie-dae-stywesiekte (three-day-stiffsickness) or ephemeral fever,3, 57 and a condition caused by the plant Crotalaria burkeana, the stywesiektebossie.8, 22, 50, 52, 56
Crotalaria burkeana is a woody, much-branched, perennial herb 300-600 mm tall. A feature of the plant is that all the aerial parts are covered with long hairs (Figure 1). The three, rarely five, foliate leaves are carried on 15-30 mm long leaf-stalks, and the leaflets are linear to lanceolate in shape. The white, occasionally yellow-tinged or mauve, pea-like flowers, veined brown or dark blue, are borne in terminal racemes (Figure 2) during November through to March. The fruits are inflated pods c.40 mm long and 15 mm in diameter (Figure 3), containing brown kidney-shaped seeds.8, 22, 52, 61 The stywesiektebossie most often frequents wooded grassland or open sandy areas and spreads rapidly in old cultivated lands, trampled veld and disturbed soil.
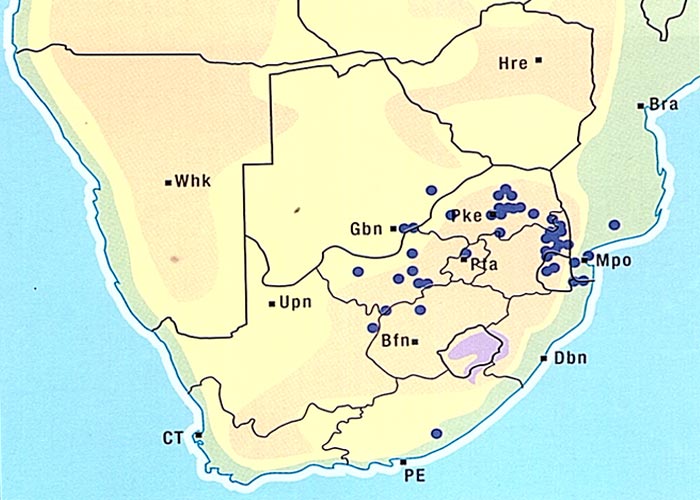
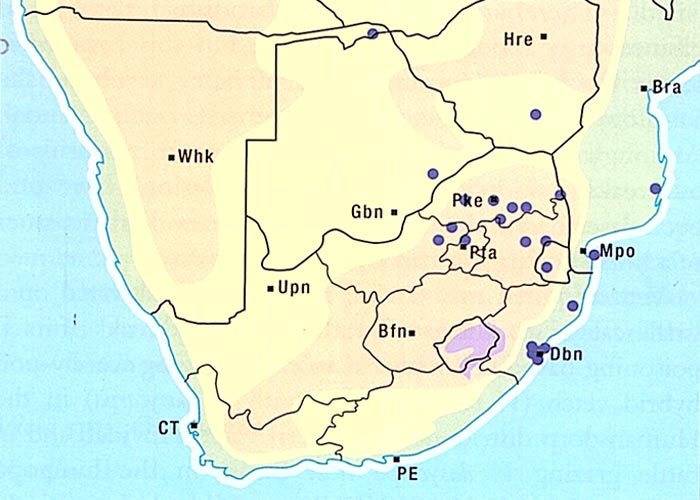
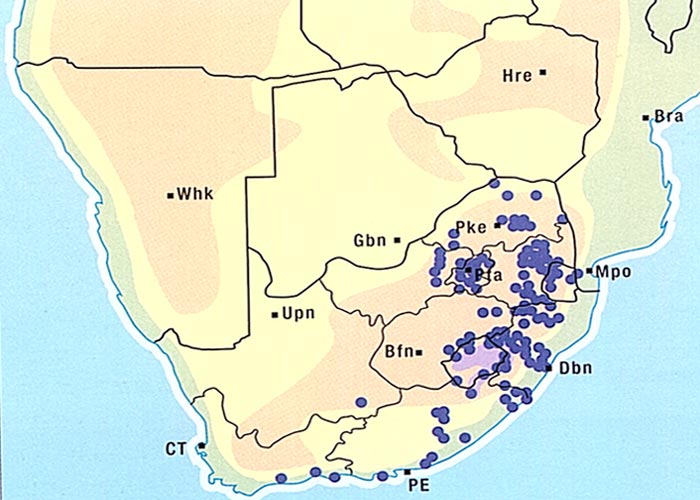
The aerial parts of the plant are sensitive to frost and die off in winter.8, 61 Crotalaria burkeana is widely distributed especially in Mpumalanga, the western and northern parts of the former Transvaal, the western Free State, the northern parts of the Northern Cape Province and Botswana (Figure 4).5, 50, 52, 61
Steyn52 reported that C. rhodesiae - a soft, slender, prostrate species - causes stywesieke in the Limpopo Province; while in the central and northern parts of Namibia, during bad years, other Crotalaria spp. allegedly give rise to thousands of cases of stywesiekte. Outbreaks of the disease have occurred in the districts of Klerksdorp, Zeerust, Rustenburg, Pietersburg and Barberton.8, 22, 56 Stywesiekte also affects cattle in Zimbabwe, where various Crotalaria spp. inhabit the veld. The disease can take on alarming proportions in certain parts of the former Matabeleland. It is said that in bad years more than half of the cattle in the Inyati Tribal Trust Land may suffer from stywesiekte29 where C. barkae (= C. geminifora) is suspected of being the causal plant. The botanical features and habitat of C. barkae are very similar to those of C. burkeana, the chief difference being that C. barkae is usually an annual or short-lived perennial. Other annual Crotalaria spp. are also suspected of causing stywesiekte in Zimbabwe, e.g. C. steudneri (= C. hispida) which has been found abundantly on the veld where cattle had become affected with the condition.29
No mention is made of liver damage in cattle poisoned by either C. burkeana or C. barkae, Steyn52 on the other hand, reported hepatic involvement, particularly in horses and donkeys, suffering from C. burkeana poisoning. Many affected donkeys in Namibia apparently had to be killed as a result of induration of the liver. Steyn50 also referred to Theiler, who fed 15 kg of dried C. burkeana in 17 days to a horse that developed staggers on the eighteenth day and died two days later, showing what he described as 'acute parenchymatous hepatitis'.
The active principles of the majority of Crotalaria spp. worldwide are the pyrrolizidine alkaloids, which are mainly responsible for severe liver and/or lung damage in stock (see Liver and Respiratory system). The toxin of C. burkeana is not known, but the disease caused by the stywesiektebossie differs markedly from that of hepatotoxic Crotalaria species.
Steyn50 mentions a report by Burtt-Davy (1903/4) describing the signs of stywesiekte in cattle supposedly poisoned by C. burkeana. Later Theiler56 confirmed that this plant was a cause of stywesiekte in cattle. He showed in a feeding trial that daily ingestion of 4-5 kg of fresh plant material resulted in acute laminitis on Day 6. However, this latent period increased with smaller daily intakes of C. burkeana: two cows, fed 1,8 kg fresh plants daily, respectively developed laminitis on the twentieth and twenty-ninth days of the experiment. In Zimbabwe, Jackson et al.29 fed a total of 12,7 kg of air-dried C. barkae over a seven-day period to a 182 kg bovine animal. The typical acute laminitis appeared on the fourth day, and the feet only returned to normal c.15 months later.
Crotalaria-induced stywesiekte occurs most commonly in cattle, but according to Steyn52 sheep, goats, donkeys, horses and wild antelope may also be affected. The acute phase of the disease is characterized by painful and warm hooves with reddish discolouration of the unpigmented parts. The stance and gait of affected cattle are typical of an animal suffering from a painful acute laminitis, i.e. they stand with their backs arched, front legs somewhat to the fore and the hind legs tucked under, alternately lifting their feet. The animals lie down frequently and rise and walk with difficulty. Affected cattle may be feverish and the heart and respiratory rates are sometimes increased.8, 22, 29, 50, 52, 56
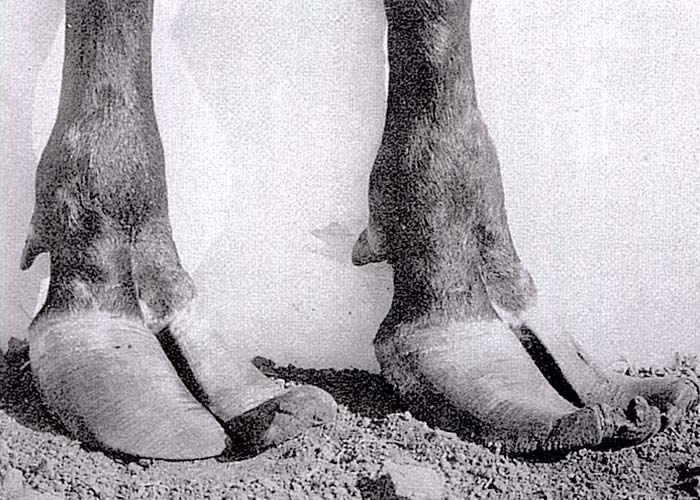
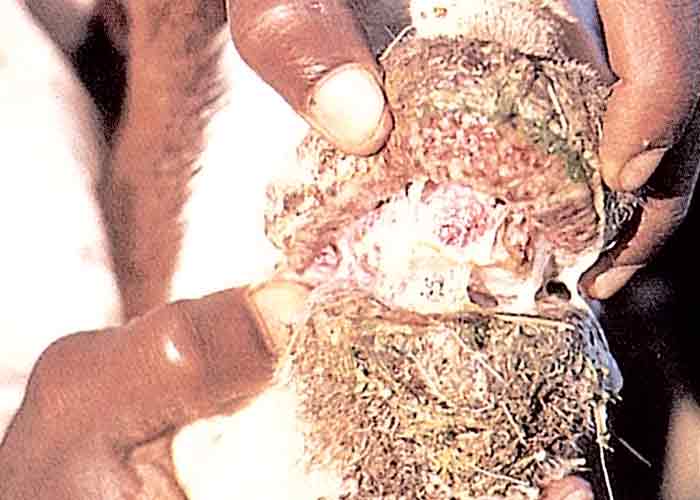
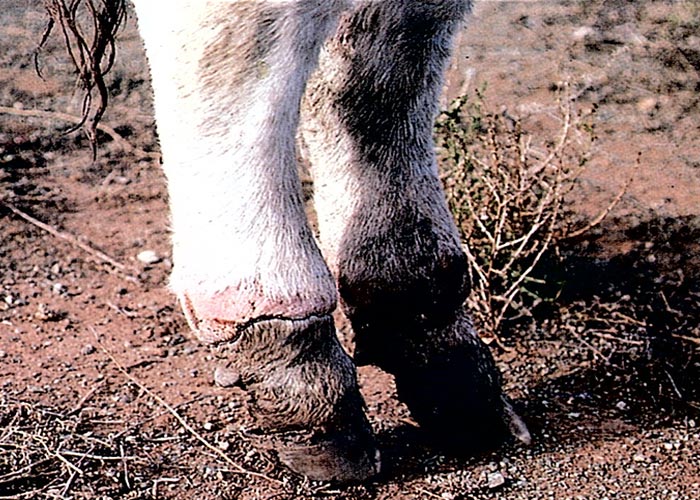
The acute phase lasts a few weeks before the disease gradually becomes chronic.8, 22, 29, 50 During the latter phase, pain decreases and the hooves grow out abnormally, the claws becoming elongated and their points turning upwards (Figure 5). The abnormal hooves cause the animal to walk with difficulty, using an abnormal gait. Rattling sounds from the elongated toes, knocking on the ground and against each other, are heard, especially when affected cattle are driven hard. These sounds give rise to the common name, rattle bush, for C. burkeana.50, 52 Severely affected animals lose weight, cannot rise and may die of starvation, but if stywesiekte cases are carefully nursed, mortality will be negligible. After approximately one year the elongated parts of the hooves may wear off and the feet return to normal.29
According to Theiler,56 Mitchell gave a description of the microscopical features of hoof lesions of a bovine which had suffered for 46 days with stywesiekte. Granulation tissue separated the horny and sensitive laminae; this reaction was particularly marked near the toe and was more severe in the hind claws. The area surrounding the coronopedal articulation was thickened by dense fibrous connective tissue and the articular surface of the os coronae and os pedis were also thickened.
Stywesiekte can be best prevented by proper veld management, because the incriminated plants are uncommon where the grass cover is good.29
Overgrown hooves are not associated only with Crotalaria poisoning. Similar lesions may be evident in stabled animals or animals grazing on sandy soil, where the hooves are not subjected to much wear. The hooves of cattle and even or some game species may likewise be deformed following infection with foot-and-mouth disease. In stall-fed cattle on high energy rations, the hooves often grow out as a result of histamine-induced laminitis.
In South Africa, C. juncea (Figures 6 and 7) is sometimes cultivated as fodder and/or a green manure crop.52 Little is known locally about its toxicity but, according to Steyn,52 suspected poisonings have been reported in horses, sheep and cattle. In horses, C. juncea causes a chronic lung condition, called jaagsiekte (see Respiratory system).52, 53 Nobre et al. (1994)36 describe a recent outbreak where 20 horses died within 30 days of receiving 40% ground C. juncea seed in their diet. No laminitis but severe lung involvement, as in jaagsiekte, were reported.
Loss of wool may apparently occur in sheep (Figure 8) following the ingestion of large quantities of the plant.52, 53 Steyn52 proposed that sunn hemp should not be fed to horses and, if included in the ration of sheep and cattle, it should not constitute more than one-tenth of the daily intake. He also warned that the toxicity of C. juncea may vary in different localities.
Chrysocoma ciliata L. (Asteraceae)
(= C. tenuifolia)
Bitter bush, bitterbos, bitterkaroo
Chrysocoma ciliata is responsible for alopecia (kaalsiekte) in lambs and kids suckled by dams that ingest large amounts of the plant. Adult sheep, goats and cattle, on the other hand, develop lakseersiekte (purging disease) on bitterbos veld.22, 52
Bitterbos have been described by Vahrmeijer61 as a dense, perennial, woody shrublet, about 500 mm high, with a rounded or cushion-shaped form (Figure 9). The numerous leaves are small (5 mm long), narrowly linear, dark green and arranged alternately on the greyish-brown stems (Figure 10). They are usually slightly sticky and bitter, hence the name bitterbos. The flowerheads without ray florets are small, yellow, and arise from the ends of the branches (Figure 10).61
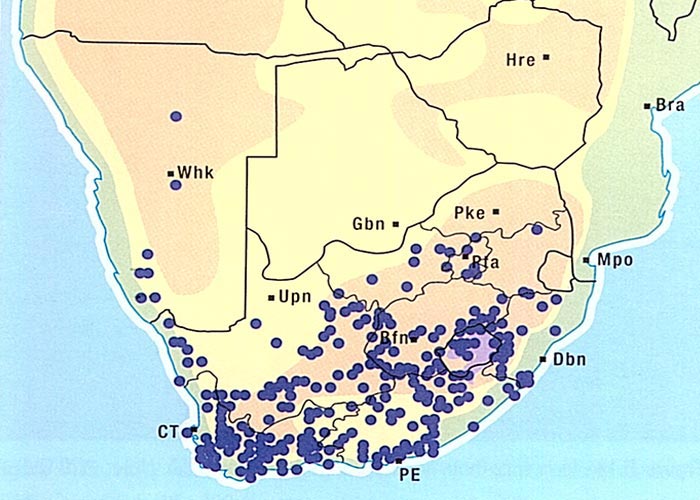
Bitterbos is widely distributed in the Karoo (Figure 11) on a variety of soil types, except brackish areas, where it does not flourish. It is an extremely active invader and is the plant most commonly associated with expansion of the Karoo. Stock will graze large quantities of bitterbos only if more palatable species are not available. Dense stands of C. ciliata are indicative of overgrazed, deteriorating veld.22, 61
The incidence of kaalsiekte is highest in years with good winter and spring rains, when C. ciliata is young and succulent. The disease is generally noticed during the lambing season from August to November, but cases have been recorded at other times of the year.22 The exact quantity of bitterbos that has to be eaten by the dam before kaalsiekte is induced in her offspring is not known. Steyn48 reported that a kid, whose dam received 12,5 kg dried flowering bitterbos in 35 days, developed alopecia and fatal diarrhoea. According to experimental evidence, pregnant sheep or goats have to ingest considerable quantities of the plant for a few weeks before partus for kaalsiekte to occur.22, 48, 50 Desiccation of the plant is said not to destroy its toxicity. The unidentified toxin is transmitted through the milk to the young, and this explains why affected lambs or kids have normal hair coats at birth, why twins are less susceptible to kaalsiekte than single offspring, and why the risk of kaalsiekte can be reduced by partially emptying the udders.22, 50, 52 Steyn48 failed to induce the disease in young kids by drenching them repeatedly with fairly large quantities of the dried plant.48
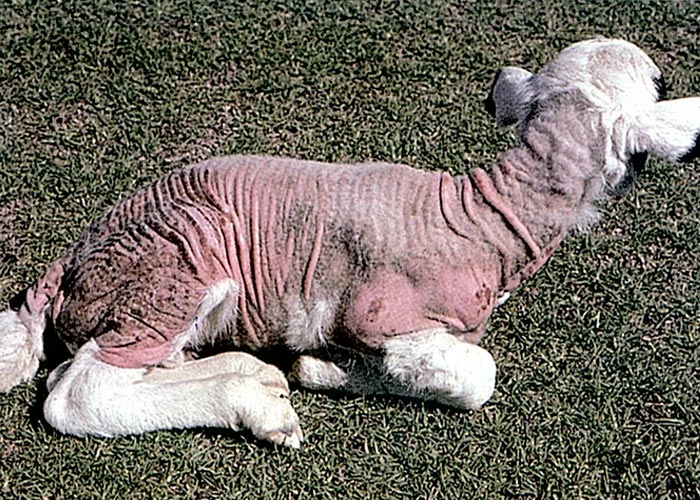
Lambs or kids develop kaalsiekte from about three to 14 days after birth, seldom later. The first signs are pruritus and scratching or biting at the affected areas, often accompanied by the swallowing of hair. The fleece is ruffled and tufts of wool can easily be plucked out. Loss of hair starts over the shoulders and lateral aspects of the upper hind legs, then spreads progressively to other parts of the body until eventually remnants of fleece are left only at the extremities (tail, lower limbs, ears) and on top of the head (Figure 12). No primary macroscopical or microscopical lesions have been reported in skin of the bald areas. Unpigmented skin is notably more readily affected than the pigmented parts, hence in crossbred sheep the white areas may be bald, leaving the dark areas covered by hair. Although black or brown animals are apparently more resistant to kaalsiekte, in severe outbreaks they too may lose their coats.2, 48, 50, 52
As a result of exposure to the sun, the hairless areas become red and sensitive (Figure 12), followed by cutaneous exudation and formation of scabs. Another common complication of kaalsiekte is bronchopneumonia in lambs exposed to cold winds.2, 48, 50, 52
Kaalsiekte is usually associated with diarrhoea, though some cases may scour without loss of hair and vice versa. The diarrhoea is thought to be either a primary effect of the toxin or to result from irritation of the digestive tract by the ingested hair. Sometimes the abomasum is obstructed by hairballs (trichobezoars) lodged in the pylorus, a fairly common complication of kaalsiekte. In such cases, the ingesta in the abomasum decomposes and the stomach becomes distended by foul-smelling gas.22, 52
In addition to the obvious signs of kaalsiekte, affected lambs sometimes manifest conjunctivitis, keratitis, rhinitis and purulent nasal and ocular discharges. Steyn52 noted spasms and posterior paresis in some kaalsiekte lambs, but in the light of our present knowledge the possibility cannot be excluded that these lambs might have suffered from valsiekte.62 The apparent association of C. ciliata with valsiekte (see Central nervous system) and phytobezoars (see Gastrointestinal tract) must be borne in mind when the total effect of the plant on the health of lambs is considered. The morbidity of kaalsiekte on kaalsiekte-prone farms may vary from 1% in dry years to almost 100% in extremely wet years. Unless the lambs are properly cared for, more than 50% can be expected to die.22
The best treatment of kaalsiekte is to provide shelter for afflicted lambs against the sun and wind. Where shelter is unavailable, the use of small 'blankets' has been advocated.52 Bald areas can be dressed with raw linseed oil and limewater (carron oil), lanolin, or some other emollient dressing to prevent hardening. Mild purgatives have been prescribed for hairballs,22, 52 although the wisdom of such a treatment is questionable (see Gastrointestinal tract).
Kaalsiekte is a product of overstocking, and since large amounts of bitterbos are ingested by stock only if they are forced to do so, the best long-term solution to the problem is good pasture management. If ewes are kept off Chrysocoma-infested pastures for about a month prior to and after lambing, the incidence of kaalsiekte can be reduced to negligible levels. Where evasion of the plant is impossible, pregnant ewes might be allowed to graze daily for an hour or so on green oats, wheat, rye, lucerne or some other supplementary feed before being turned out onto the veld. It is preferable for pastures heavily infested with luxuriantly growing bitterbos to be avoided.22, 52
Severe diarrhoea has been reported in sheep and cattle that take in large quantities of bitterbos. The condition, known as lakseersiekte (purging disease), occurs on trampled veld during winter in times of drought, when stock graze almost exclusively on bitterbos. Stock suffering from lakseersiekte may die suddenly or develop a persistent, sometimes haemorrhagic diarrhoea which leads to weakness, dehydration and recumbency. A severe, occasionally haemorrhagic, enteritis is the principal necropsy feature. Some evidence has been presented that conditioning of stock with non-toxic doses of bitterbos can bring about a measure of tolerance to intoxication by the plant. The most effective treatment of lakseersiekte is to provide supplementary feed, or to move stock to bitterbos-free grazing. Under experimental conditions, supplementary feeding has reportedly enabled animals to ingest lethal doses of Chrysocoma without notable ill effects.49, 50, 52
Vicia spp. (Fabaceae)
V. villosa Roth.
Hairy vetch, broad-leaved vetch, breëblaarperswieke
V. villosa spp. dasycarpa
Woolly-pod vetch
V. sativa L.
Common vetch
V. faba L.
Faba bean, broad bean, horse bean
V. sativa L. subsp. nigra (L.) Ehrh . (= V. angustifolia)
Narrow-leafed vetch
V. benghalensis L.
Popany vetch
Vicia spp. (Figure 13) are low or climbing herbs with singly compound leaves, the rachis of which ends in characteristic tendrils (Figure 14) or bristles (Elizabeth Retief. NBI, personal communication, 1999). These legumes are often cultivated with other grasses as artificial pastures or they can be harvested as hay and silage. Poisoning with various vetches has been recorded occasionally in stock and humans worldwide.32
Different forms of poisoning are associated with Vicia spp.7, 32, 39, 40 The seeds of the common vetch, V. sativa, contain cyanogenic glycosides responsible for acute deaths in stock32. Photosensitization and liver lesions similar to those of lupinosis have also been reported in horses feeding on Vicia sativa which contains beta-cyano-L-alanine, a compound related to the toxic principle of some Lathyrus spp.32 Vicia faba or the faba bean is native to the Mediterranean region, where it is cultivated as food for humans and animals.32 Ingestion of the raw or partially cooked beans or inhalation of pollen causes haemolytic anaemia and toxic hepatitis, called 'favism' in humans. It is believed that a genetic, sex-linked factor, which is responsible for low levels of whole-blood glutathione and a deficiency of glucose-6-phosphate dehydrogenase activity, makes certain individuals more susceptible to poisoning.32
Three apparently distinct syndromes have been noticed in cattle grazing on hairy vetch. Cattle may show locomotory disturbances, convulsions, sexual excitement, bellowing and acute deaths after the consumption of the seeds. In the second syndrome, there are subcutaneous swellings of the head, neck and body, alopecia, herpetiform eruptions in the oral mucous membranes, purulent nasal discharge, coughing, dyspnoea, and a few animals may die after about 12-15 days of illness. In the third and most commonly encountered syndrome animals develop pruritus and marked skin lesions two to six weeks after being put on hairy vetch pastures. The skin lesions are characterized by roughened coats, exudative to ulcerative granulomatous dermatitis, thickening of the skin and alopecia, especially of the escutcheon, perineum, udder and teats, tailhead and neck of pigmented and non-pigmented areas. Other signs include loss of appetite and body mass, severe diarrhoea which may be bloody in advanced cases, conjunctivitis, dyspnoea and, occasionally, abortion. The morbidity could be as high as 35% and the mortality is usually 50%, but can range from zero to 100%.7, 39 Most animals die after a course of illness of 10-20 days.
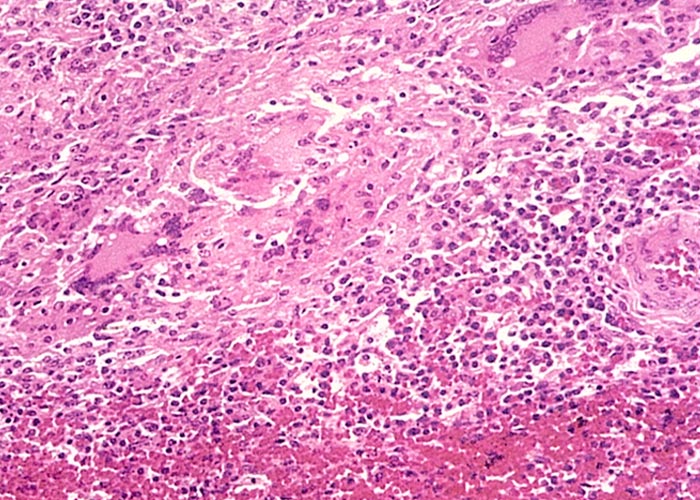
Although multifocal to diffuse greyish-yellow lesions, which are sometimes fairly sharply demarcated, can be found in a wide range of organs and tissues, these changes occur most commonly in the kidneys, myocardium, adrenal and thyroid glands.7, 39, 40 Microscopically, the lesions in the various organs are very similar and comprise multifocal infiltrations of macrophages, lymphocytes, plasma cells, occasional multinucleated giant cells and some eosinophils (Figure 15). These cellular infiltrates can occur in most organs and tissues but are more prevalent in the renal cortices, myocardium, adrenals and the dermis, and are less commonly found in the thyroids, salivary glands, pancreas, spleen, lymph nodes and portal triads of the liver. Occasionally, the trachea, bronchi, gall-bladder, intestines, ovaries and endometrium are involved. Depending on the tissue, degeneration and necrosis of the cells are often associated with these infiltrates. Vascular lesions have been reported in the myocardium and the skin.7, 39 Burroughs et al.7 described subintimal mineralized plaques, proliferation of endothelial cells, as well as oedema and fibroplasia of the intima in a few of the smaller arteries in the heart, while Panciera39 reported nuclear swelling of endothelial cells, and thickening and hyalinization of the walls of many arterioles in the dermis.
Adult cattle three years or older are more susceptible than young animals, which are usually either mildly affected or not at all.39 Apart from this age predisposition, Holstein-Friesian and Aberdeen Angus breeds appear to be more prone to develop the disease than Hereford and Jersey cattle. Thousands of cattle in the USA are pastured each year on hairy vetch without any ill effects. Most outbreaks worldwide have occurred when hairy vetch pastures were at the zenith of their growth. To date, the temporary toxicity of pastures has not been explained satisfactorily, and attempts to reproduce the disease have failed.39 There are speculations that insect damage of pastures may render V. villosa toxic, but this hypothesis seems doubtful.39 Incrimination of hairy vetch in the aetiology of this disease, therefore, remains circumstantial. Although cattle have been involved in the majority of outbreaks worldwide, granulomatous lesions have also been described in the heart, kidneys, skin and other tissues of a horse grazing on-hairy vetch pastures.1
Vetches, often mixed with ryegrass, are cultivated on a small scale as stock feed in South Africa. Outbreaks of vetch poisoning have been reported in cattle grazing woolly-pod hybrid vetch (V. villosa crossed with V. dasycarpa) in the Humansdorp district of the Eastern Cape Province and in cattle grazing V. dasycarpa near Sibasa in the Limpopo Province (J.A Neser, ARC-OVI, unpublished observations 1983). In Australia this species has been involved in at least 11 outbreaks. V. benghalensis (popany vetch) was associated with V. dasycarpa in two of the outbreaks and once apparently caused the disease on its own.30 In South Africa only one case of V. benghalensis poisoning has been recorded.18
The exudative to ulcerative dermatitis associated with hairy vetch poisoning should be differentiated from similar skin lesions seen in photosensitivity diseases and chlorinated naphthalene poisoning, while the microscopical granulomatous reactions in many organs are very similar to those caused by acid-fast bacteria, fungi, helminths and foreign material.
Euphorbia ingens E. Meyer ex Boiss. (Euphorbiaceae)
Candelabra tree, gewone naboom
The naboom is a much-branched tree about 10 m in height. Unlike other tree Euphorbia species, the lower branches do not fall off and leave a bare stem with a cluster of branchlets at the top. Instead, the branches emerge fairly low down on the trunk from which they grow out fairly upright into a dense rounded crown (Figure 16). The branches are four-angled and divided by constrictions into irregular segments. Along the margins of the angles or wings, paired spines (2 mm long) are present, which may be much reduced or even absent. Small fleshy, red flowers arise in groups of three from the margins of the branches near to the obsolescent, corky spine shields. The fruits are three-lobed spherical capsules up to 10 mm in diameter.12 Euphorbia ingens favours savanna bushveld, its distribution extending from KwaZulu-Natal and the northern provinces of South Africa into Botswana, Zimbabwe and Mozambique (Figure 17).12, 38
Many species of the Euphorbiaceae, including the naboom (E. ingens), the lesser candelabra tree (E. cooperi) and the hedge euphorbia (E. tirucalli), contain highly irritant latices, a drop in the eye of which can lead to severe inflammation and temporary or permanent blindness. Exposure of the skin or buccal mucosa to the latex results in vesicles and ulcers. Steyn51, 52 reported severe burning and destruction of the facial skin (Figure 18) and the eyes, as well as ulceration of the tongues and mouths, of cattle that had run through a thick stand of E. ingens. It was as if caustic soda had been poured onto their heads.
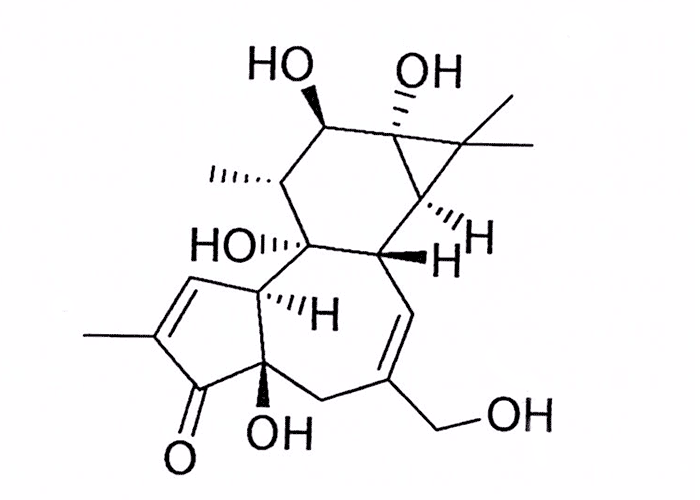
The toxic principles in the latices are esters of the diterpene alcohols phorbol (Figure 19) and closely related resiniferol and ingenol. Apart from their highly irritant properties, many of these widely investigated substances have tumour-promoting, cytotoxic, antileukaemic, insecticidal and piscicidal effects.31
Steyn suggested that affected skin should be gently washed, first with warm soapy water and then with a strong aqueous solution of potassium permanganate. The treatment is completed by covering the injured area with cotton wool soaked in bicarbonate of soda (one teaspoonful in a cup of water).51, 52 Kinghorn,31 on the other hand, recommended that affected skin should not be covered as in experimental cases this aggravated the condition. He also noted that ingestion of the latices of the toxic Euphorbiaceae generally resulted in severe gastrointestinal irritation and diarrhoea.31 Steyn,52 contrastingly, having observed only mild transient diarrhoea in sheep dosed with Synadenium cupulare (= S. arborescens), a species of the Euphorbiaceae which also contains an irritant latex, suggested that the irritant component in the latex was probably destroyed by the ruminal juices.52 Not all of the 288 southern African Euphorbia spp.2 are toxic, in fact many of the noorsdoring are regarded as good fodder for stock.
The latex has long been known also to have piscicidal properties. Wefts of grass soaked in latex are bound to stones and dropped into pools by tribesmen living along the Limpopo River. After about 15 minutes, stupefied fish are said to float to the surface, where they can be harvested.12, 13
Primary photosensitivity
Primary photosensitivity, unlike secondary photosensitivity (see Liver), results from the ingestion of actual photodynamic agents. It can be induced, among others, by drugs, two of which are acridine dyes, such as euflavine (a babesiacide) and tetracyclines. Since the photodynamic agents of primary photosensitivities are not manufactured by microbial degradation of precursors in the rumen (e.g. phylloerythrin from chlorophyll), all classes of stock and also of birds are susceptible to intoxication by them.
Primary photosensitivity caused by Hypericum perforatum (St John's wort) occurs worldwide in all species of stock.10, 11, 66 Despite having become established on c.20 000 ha in the Boland region of the Western Cape Province since it was inadvertently introduced with vetch seed in 1942,54 St John's wort has never been incriminated in any outbreaks of photosensitivity (D.J. Schneider, Regional Veterinary Laboratory, Stellenbosch, personal communication, 1986). According to Stirton,54 there are six indigenous species of Hypericum in southern Africa. Quin41 reported the experimental production of primary photosensitivity with two of these, namely H. aethiopicum (= H. aethiopicum var. glaucescens) and H. revolutum (= H. leucoptychodes), in a limited number of animals. He found the former more toxic than the latter, which grows up to 3 m tall. The photodynamic agent is hypericin, a conjugated quinone derivative of naphthodianthrone (Figure 20). There is no documented evidence of naturally occurring hypericism in southern Africa, but incidents of primary photosensitivity in sheep (T.F. Adelaar, VRI, Onderstepoort, personal communication, 1967) and in cattle (M. van Aardt, private practitioner, Ermelo, personal communication, 1986) have been attributed to this plant. In view of its potential importance, a description is given of H. aethiopicum.61
Hypericum aethiopicum Thunb. (Hypericaceae)
St John's wort, Johanneskruid, vlieëbossie
This herb-like plant, growing 300 mm high, has a perennial underground root system, often forming a dense clump of erect, almost entirely unbranched stems terminating in branched inflorescences (Figure 21). The small ovate, sessile leaves are borne in opposite pairs. The flowers are sulphur yellow and arise from the axils of leaves near the tips of branches. Small, characteristic, dark glands dot the leaves and calyx lobes of both subspecies and also the stems of one subspecies.61
The plant occurs most commonly in stony areas and open grassland or damp areas, as well as on old cultivated lands (Figure 22).61
Primary photosensitization has periodically been reported from the Western and Northern Cape Provinces in sheep ingesting Erodium moschatum (L.) L'Hér. (musk heron's bill), an exotic, naturalised weed growing in disturbed areas such as cultivated lands, orchards and vineyards55 (Figure 23). Stroebel (2002),55 investigating an outbreak at Riebeeck-West, Western Cape, reported that over a period of about two weeks ten sheep out of a flock ±350 were affected, of which one died. The ewes exhibited purulent crusts on their nasal planes and other hairless areas of their faces, vulvas and coronets. The ears were erythematous, but not swollen. At autopsy only skin lesions were observed and no icterus or other lesions were present. The histological examination of affected skin revealed epidermal necrosis with scab formation and severe inflammatory changes in the dermis. Mild photosensitization was experimentally induced in two sheep dosed with E. moschatum plant material.55
Fagopyrum esculentum (= F. sygittum), or buckwheat, is cultivated worldwide for human consumption and as a stock feed. In South Africa it is grown mainly in the eastern and south-eastern Mpumalanga and in the north-eastern Free State. Both the whole plant and the seed are fed to animals.
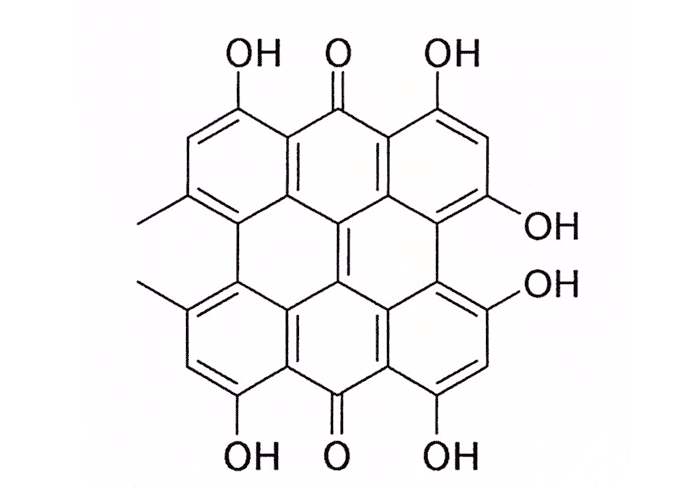
From a toxicological point of view, buckwheat is well known for causing fagopyrism, a primary photosensitization induced by fagopyrin, a photodynamic agent chemically closely related to hypericin (vide supra) (Figure 20). According to Watt and Breyer-Brandwijk,66 fagopyrin is a pigment found in the flowers and seeds, but Steyn50 states that all parts of the plant, whether dry or fresh, are capable of producing photosensitivity, the fresh, flowering plants being the most toxic. Clinical signs may appear in less than 24 hours after commencement of feeding and may still be present up to four weeks after feeding has been discontinued. No published account of fagopyrism in southern Africa could be traced, but the authors are aware of a local outbreak in sheep which had been fed on large quantities of reject seed.
Most textbooks infer that burr clover (Medicago polymorpha = M. denticulata), certain other clovers, and even lucerne (M. sativa), may cause primary photosensitivity, but there is some speculation that this photosensitivity could possibly be of a secondary type.16
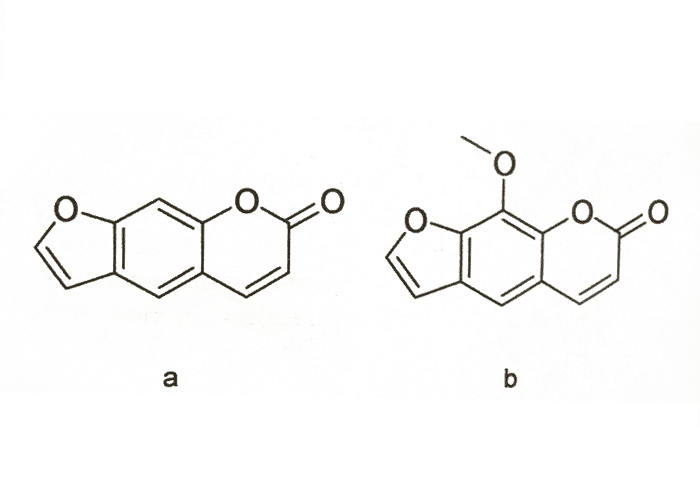
Photodynamic plant furanocoumarins, or psoralens (Figure 24), differ from naphthodianthrone-derived conjugated quinones (see Hypericum spp.) in being able also to cause contact photosensitivity when applied to the skin. These compounds are found in many plants, particularly in the Apiaceae, e.g. Ammi majus (lace flower). Extracts of lace flower was used as early as 1 500 BC for treating loss or deficiency of melanin pigmentation in humans. Psoralens, (notably xanthotoxin or methoxysporalen from A. majus) are extensively used for the treatment of dermatological diorders.19, 42 Notwithstanding extensive research19 the complex mechanism by which furanocoumarins cause photosensitivity has not been fully elucidated. Intercalation into cellular DNA and the epidermal growth factor receptor to form photo adducts, appears to be involved.33
Photodynamic plant furanocoumarins have been linked with photosensitization of domestic animals in various countries abroad28 and in South Africa.63 In the latter instance, cows developed painful skin lesions on their teats apparently after walking through Ammi majus, a common weed in the Eastern Cape Province and certain localities elsewhere in the country.63 There is also strong circumstantial evidence that ostriches in the same province had developed photosensitivity after being fed dried lucerne contaminated with A. majus (W.P. Burger, Oudtshoorn, personal com munication 1996).
Furanocoumarins occur as such or as phytoalexins generated by plants such as parsnips and celery in response to fungal infection. As both cultivated and wild members of the furanocoumarin-containing families, Apiaceae (including Ammi majus var. glaucifolius), Rutaceae, Fabaceae, Moraceae and Orchidaceae, occur in South Africa, furanocoumarins must be borne in mind as possible causes of certain unexplained local primary photosensitizations.
The photodynamic agent of Erodium moschatum is not a furanocoumarin as incubation of ground leaves on a Candida albicans seeded plate and exposed to ultraviolet light (320-380 nm) revealed no zone of inhibition.43, 55
Compared to hepatogenous photosensitivity, the damage caused by primary photosensitizations in South Africa is so small that it is almost negligible. The clinical signs of primary photosensitivity are similar to those of secondary photosensitivity save for the icterus (see Liver).
Mechanical injury of the skin
The burs of weeds such as Xanthium spp. (cocklebur, boetebossie or kankerroos) and the sharp-pointed seeds and awns of grasses such as Aristida and Heteropogon spp. (steekgrasse) can detrimentally affect the quality of wool in which they lodge.50, 64 Apart from the possible hepatotoxic effects of the plant (see Liver), the burs of Xanthium spp. have been known to cause obstruction and inflammation of the prepuce of cattle.50 Sharp seeds may also lodge in the external body orifices, such as the mouth, causing severe tissue damage and abscessation. Foreign bodies with retrorse barbs may even penetrate the skin, particularly of lambs.
Foreign body vesiculitis is caused by a variety of barbed grass seeds and their awns. Among others, the seeds of Aristida congesta14, 15, 64 and Themeda triandra14, 15 have been incriminated as causal agents of this condition in sheep. The offending seed of Heteropogon cortortus (Figure 25), armed with retrorse hairs - has a very sharp-tipped callus, indurated lemma (enclosing the seed), and a long bent and twisted awn (Figure 26). The retrorse hairs allow the cylindrical spikelet to move only in one direction while the awn, twisting as a result of hygroscopic changes, drills the seed into the ground.17 Grass seeds are believed to penetrate the skin in the same fashion.14, 64 After penetration, any movement of either seed or skin, will result in the seed or awn being driven progressively deeper into the body. The seeds/awns settle in the subcutis where they may either become encapsulated or cause foreign body vesiculitis or small granulomas and abscesses or may migrate deeper into the superficial muscles or even into the thoracic or abdominal cavities. At slaughter, grass seeds and awns may be encountered inter- and intramuscularly or even subpleural/peritoneally in sheep (C.M. Veary, Department of Animal and Community Health, Faculty for Veterinary Science, Onderstepoort, personal communication, 1999).
De Wet14 describes penetration by awns and not the grass seeds, as the main problem. In one extraordinary case (Figure 27) an awn, possibly of Heteropogon contortus, was incidentally found lodged in the atrioventricular valve of a sheep (R.C. Tustin, Department of Pathology, Faculty for Veterinary Science, University of Pretoria, personal communication, 1986). It is theorised that the awn had probably migrated into a subcutaneous lymphatic vessel before being transported to the heart.
Apart from Heteropogon, Arisrida and Themeda spp., severe skin problems are also caused by the seeds of Bromus diandrus (predikantsluis). The glumes and awns of several seeds of this plant felted together form large, heavily barbed structures which cause extensive mechanical damage. They even lodged between the toes of dogs from where, after penetrating interdigitally, they may migrate upwards, sometimes re-emerging as high as the fetlock (D.J. Schneider, Stellenbosch, personal communication, 1986).
Several invasive Stipa spp. from South America have large, sharp seeds, known as 'flechilles' (little arrows), which bore into the skin of animals causing painful wounds. Nasella neesiana (= Stipa neesiana) is becoming common in parts of the Eastern Cape Province coastal belt, where it could pose a problem in future (M.J. Wells, Botanical Research Institute, Pretoria, personal communication, 1986).
Foreign body damage to the buccal mucous membrane can result from both grass seed and thorns. De Wet (1983)14 describes an incident where 400 out of 2 000 suckling lambs died in a flock grazing on seeding rooigras, Themeda triandra. The seeds and awns of this species, which are very similar in structure to those of Heteropogon contortus, penetrated the mucosa especially of the faucial area of the mouth but also of the palate, causing severe Corynebacterium/Arcanobacterium abscessation.
Numerous fine, but severely barbed, awns enable whole seedheads of Setaria verticillata (bur-bristle grass, klits setaria) to stick to the soft hair, especially of shaggy dogs, badly matting their coats. On self-grooming the awns may penetrate the soft palate of the dogs causing prolonged pharyngitis. Mechanical removal of awns as much as 0,5 cm long with a forceps results in uneventful recovery (M.M.T. Kruse and T.W. Naudé, Faculty for Veterinary Science, Onderstepoort, personal observations, 1984).
Even the very fine, apparently soft and harmless seeds of Chloris pycnothrix can damage the buccal mucous membrane of stock. Numerous lesions packed with clumps of delicate C. pycnothrix seeds have been noticed in the mouths of sheep and goats fed milled lucerne contaminated by this annual grass. Microscopical examination of the offending seed revealed fine retrorse barbs on the callus and awn. The impression was gained that once one of the finely barbed seeds had penetrated the mucous membrane, other seeds packed onto it. Alternatively, the fine dried seeds were believed to clump together during milling, forming large but finely barbed structures which penetrated the mucous membrane causing extensive damage (D.G.A. Nolte, Delmas, P. Stadler, G. Bath and T.W. Naudé, Faculty for Veterinary Science, University of Pretoria, personal observations, 1988). The problem was also experienced in cattle consuming ensiled lucerne contaminated by C. pycnothrix seeds.35
Any of the above-mentioned retrorsely barbed, self-propelling seeds can cause extensive problems if lodged in the eyes or ears, since all attempts by the animal to dislodge them merely propels them deeper. In this respect the large structures of Bromus diandrus are particularly dangerous. The seeds of kakiebos, Tagetes minuta, are finely but densely barbed and although they can migrate to the surface of the skin down hairs, they do not appear to penetrate the epidermis. However, an instance has been recorded where kakiebos seed, having fallen into the ear of a dog, had migrated up to and penetrated the delicate tympanum, resulting in otitis media and permanent deafness (T.W. Naudé and F. Verstraete, Faculty for Veterinary Science, University of Pretoria, personal observations, 1987).
Notwithstanding their relatively low nutritional value, Opuntia spp. (prickly pear, turksvy), especially the spineless varieties, are often cultivated as fodder for stock in semiarid parts of South Africa. These plants make good use of the low rainfall and store large quantities of water in their succulent stems in times of drought.58 Apart from the hard, long, white thorns of the thorny varieties, numerous glochids (minute thorns or bristles) are present on the leaves and fruits of both types of prickly pears. In sheep, the large thorns and the countless minute bristles penetrate the skin of the lips and also damage the mouth, oesophagus and stomach. Particularly at the pyloric ring of the abomasum, many bristles often stud the mucous membrane.58 Small abscesses and granulomas, infected by Arcanobacterium/Corynebacterium spp. and Actinobacillus lingnieresi, may complicate the penetration sites, and similar infections are sometimes found in the lungs and the bronchial, mediastinal and retropharyngeal lymph nodes.
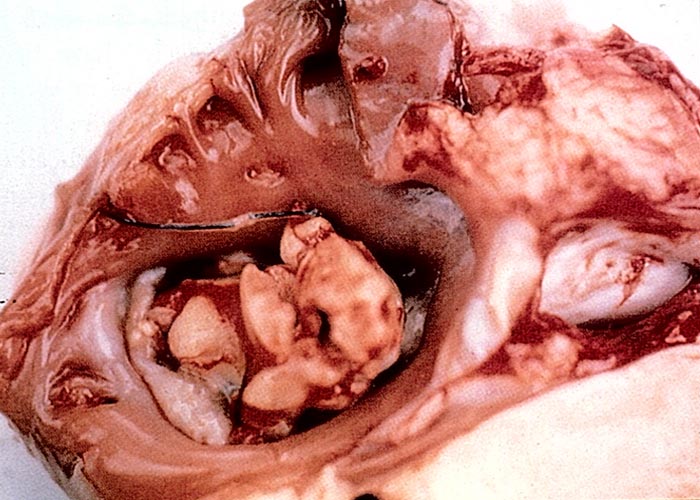
A most bizarre incident occurred involving the enormous, severely armed fruit of the devil's claw, Harpagophytum procumbens (Figure 28), which is usually dispersed by catching on the hoof of an animal and being dropped elsewhere.27 Failure of penile protrusion and coitus in a Brahman bull was investigated. Examination of the painful swollen preputium revealed that one of these fruits had lodged high up in the prepuce. As the bull was of a breed given to prolapse of the prepuce, it was assumed that the fruit had become attached to the prolapsed mucosa before being retracted up the sheath. Removal of the foreign body led to complete recovery.27
Discolouration of the skin and subcutis
Apart from toxins that mainly affect the skin, the integumentum and adjoining tissues may be discoloured as part of toxicoses that primarily involve other body systems. For example, icterus in animals is often reflected in the skin and subcutis, in acute and chronic toxic liver injury (see Liver) or following haemolytic conditions (see Haemopoietic system). Likewise, the blood, as well as the entire carcase may have a distinct brownish tinge in animals suffering from methaemoglobinaemia as a consequence of poisonings with nitrate-containing plants, Acacia nilotica and chronic copper toxicity (see Haemopoietic system).
Carcases of apparently healthy sheep are sometimes condemned at abattoirs for aesthetic, reasons, because of a yellowish to khaki-brown discolouration, which often only becomes visible a few hours after slaughter. The liver, kidneys and lymph nodes of these animals are stained with a lipofuscinous pigment. Although the aetiology of this condition has not been elucidated, the ingestion of certain plants in the Eastern Cape Province and southern parts of Namibia is suspected of playing a role.
Discussion
The most important cause of skin lesions in livestock in southern Africa is hepatogenous photosensitivity induced by hepatotoxic plants, fungi and cyanobacteria (= bluegreen algae). The aetiology, pathogenesis, clinical signs and other aspects of hepatogenous photosensitivity are discussed in detail in the chapter on the liver. In brief, the cutaneous lesions in the acute stage of photosensitivity are characterized by erythema and oedema particularly visible in the unpigmented and more exposed parts of the skin such as the face, ears, perineum and udder. In more chronic cases, the skin becomes leathery or parchment-like, immobilizing the lips or eyelids, before the necrotic parts of the skin slough. Some degree of icterus is almost invariably present and in all but the most acute cases there is some coronitis. A number of conditions that can inflict skin lesions very similar to those seen in the acute and chronic stages of photosensitivity are mentioned in the discussion of Liver.
Classical ergotism (see also 'summer syndrome', Cardiovascular system) characterized by dry gangrene of the extremities, is most often seen in cattle, particularly in calves in winter (Figure 29). Other noteworthy signs include reduced feed intake, loss of weight, cold and insensitive extremities, lameness, tremors and incoordination.9, 34, 67 In pigs, reduced body mass and agalactia are most prominent.9, 47 The disease is caused by Claviceps purpurea growing on cereal grains, such as rye, barley and wheat, and also on a number of cultivated and wild grass species.9, 34, 67 The sclerotia of the fungus contain alkaloids, such as ergotamine and ergometrine, which stimulate the adrenergic receptors. This leads to vasoconstriction, damage to the capillary endothelium, thrombosis and infarction of the skin.68 Under cold conditions, constriction of α-adrenergic blood vessels by the ergot alkaloids may be sufficiently severe to induce dry gangrene of the extremities, especially in calves. Parts of the ears, feet and tail may even drop off.9, 34, 67, 68 Lesions consistent with those of ergotism have been reported in cattle exposed to annual ryegrass ergotized by C. purpurea in the Western Cape Province of South Africa.46
Tall fescue, Festuca arundinacea, is a perennial grass used extensively for grazing as well as cover along roadsides and for lawns in certain parts of the USA.21 Hemken et al.21 reviewed the literature on tall fescue toxicity and found that different syndromes, including fescue foot, summer syndrome (hyperthermia and poor performance) fat necrosis and agalactia in livestock have been associated with this grass. In fescue foot, necrotic skin lesions indistinguishable from those caused by the alkaloids of Claviceps purpurea have been reported.21, 28 Other signs include loss of weight, roughened hair coat and pain in one or both hindclaws.21 As with the neurotoxicity of Lolium perenne (perennial ryegrass staggers), the poisonous effects of tall fescue is associated with the colonization of the grass by an endophytic fungus; in this case, Neotyphordium coenophialum (= Epicloë typhina, Acremonium coenophialium).21 The endophyte produces alkaloids, such as ergovaline, which is chemically closely related to those of C. pupurea.
Gangrenous ergotism or fescue foot has been diagnosed in 50 of 385 adult Brahmans near Standerton in South Africa.5 The cattle, which had grazed on fescue pastures (F. elatior) for less than three weeks in mid-winter, developed claw lesions, lameness and dry gangrene of the tail. Ergovaline concentrations in basal leaf sheaths and grass stems collected during the outbreak ranged from 1 720-8 170 ppb DM.5 It is estimated that fescue toxicosis is induced in cattle when ergovaline concentrations exceed 400-750 ppb5, 59 (see also Cardiovascular system).
Apart from having a radiomimetic effect on the gastrointestinal tract, lymphoid tissues and bone-marrow of animals and humans (see Haemopoietic system), the topical application of some of the trichothecenes produced by different Fusarium, Myrothecium and Stachybotrys spp. also elicit a dermal inflammatory response, characterized initially by erythema and oedema and followed by degeneration and necrosis of the epidermis.6, 23, 44, 60, 65 In fact, this skin reaction has become a standard biological test for the determination of the toxicity of the trichothecenes. The macrocyclic trichothecenes of Stachybotrys spp. have been associated with severe dermal lesions in horses, cattle and pigs abroad. In horses, the skin of the lips, particularly at the corners of the mouth, becomes necrotic and develops bleeding fissures as a result of contact with the toxin-containing feed. The subcutaneous tissues of the lips and especially the submandibular area is often oedematous.25 Two suspected outbreaks of this condition were diagnosed in horses in South Africa in 1998: one in the Western Cape Province and the other in the Free State.4 In both cases wheat straw bedding with confirmed Stachybotrys atra infestation was suspected of causing the transient superficial lesions on the muzzles of the horses.4
Similar lesions around the mouth and of the skin of the teats, belly and the snout of pigs have been reported when contaminated bedding or feed has been used.26 Cattle suffered from severe skin lesions of the scrotum and around the dew-claws after being given Stachybotrys-infected straw as bedding.24 No skin lesions, however, were noted in the outbreak of stachybotryotoxicosis in sheep in South Africa.45
Schmidtia kalahariensis and to a lesser extent Enneapogon cenchroides (sour grasses, 'suurgrasse') secrete microscopic droplets of an irritant acid during active growth.4 The former occurs in the Kalahari region of Botswana and Namibia, whereas the latter has a wider distribution. Dense stands of these pioneer, annual grasses are usually avoided by livestock during the acid secretory growth stage. These grasses cause severe irritation of the skin of the face and distal limbs. Dermatitis, eczema and alopecia around the muzzle and lower limbs can develop in horses grazing in camps where these grasses are very abundant.4
Some skin disorders of cattle, notably Vicia poisoning, can be confused with similar skin lesions seen in photosensitization, Sencobo disease or dermatophilosis, mange, sweating sickness (see Liver) and poisoning with chlorinated naphthalenes. Recently, an outbreak of feed-related, cutaneous hyperkeratosis in feedlot cattle has been noted near Pretoria, but chlorinated naphthalenes could not be demonstrated in the rations.
Chlorinated naphthalenes and related compounds, such as polychlorinated and polybrominated biphenyls and chlorinated dibenzodioxins, are used in wood preservatives, lubricating oils and electrical insulators, etc. Intoxication with these compounds results in epithelial hypertrophy, hyperplasia and metaplasia, particularly of the skin and gastrointestinal tract. The skin, especially of the neck and withers, is thickened by acanthosis and hyperkeratosis of the epidermis, and the hair follicles are transformed into keratin-filled cysts. Squamous metaplasia of the epithelium of the rumen, salivary and accessory sex glands, secretory ducts, testis and cervix, as well as ulceration of the mouth and abomasum, may also occur. These compounds inhibit the synthesis of vitamin A and low levels of vitamin A have been recorded in affected animals. However, supplementation of this vitamin has little therapeutic effect.30, 37
References
- ANDERSON, C.A. & DIVERS, T.J. 1983. Systemic granulomatous inflammation in a horse grazing hairy vetch. Journal of the American Veterinary Medical Association, 183, 569-570.
- ARNOLD, T.H. & DE WET, B.C. (eds.) 1993. Plants of southern Africa: Names and distribution. National Botanical Institute: Pretoria.
- BASSON, P.A., PIENAAR, J.G. & VAN DER WESTHUIZEN, B. 1970. The pathology of ephemeral fever: A study on the experimental disease in cattle. Journal of the South African Veterinary Medical Association, 40, 385-397.
- BOTHA, C.J. & NAUDÉ, TW. 2002. Plant poisonings and mycotoxicoses of importance in horses in southern Africa. Journal of the South African Veterinary Association, 73, 91-97.
- BOTHA, C.J. & NAUDÉ, TW., MOROE, M.L. & ROTTINGHAUS, G.E. 2004. Gangrenous ergotism in cattle grazing fescue (Festuca elatior L.) in South Africa. Journal of the South African Veterinary Association, 75, -15-48.
- BUNNER, D.L., WANNEMACHER., R.W., NEUFELD, H.A., HASSLER., C.R., PARKER, G.W., COSGRIFF, T.M. & DINTERMAN, R.E. 1985. Pathophysiology of acute T-2 intoxication in the cynomolgus monkey and rat models In: LACEY, J (ed.) Trichothecenes and others mycotoxins. Proceedings of the International Mycotoxin Symposium. Sydney, Australia: John Wiley & Sons, Ltd.
- BURROUGHS, G.W., NESER, J.A., KELLERMAN, T.S. & VAN NIEKERK, F.A. 1983. Suspected hybrid vetch (Vicia villosa crossed with Vicia dasyacarpa) poisoning of cattle in the Republic of South Africa. Journal of the South African Veterinary Association, 54, 74-79.
- BURTT-DAVY, J. 1910. Notes on Crotalaria burkeana and other leguminous plants causing disease in stock. Journal of the South African Veterinary Association, 7, 269-278.
- CHRISTENSEN, C.M. 1980. Ergots. In: Conference on mycotoxins in animal feeds and grains related to animal health. PB80-221773, US Department of Commerce, National Technical Information Service, Food and Drug Administration, Rockville, Maryland, USA.
- CLARE, N.T. 1952. Photosensitization in diseases of domestic animals. Review Series No. 3 of the Commonwealth Bureau of Animal Health. Farnham Royal, Bucks, England: Commonwealth Agricultural Bureau.
- CLARKE, MYRA L., HARVEY, D.G. & HUMPHREYS, D.J. 1981. Veterinary Toxicology. London: Baillière Tindall.
- COATES PALGRAVE, K. 1981. Trees of southern Africa. Cape Town: C. Struik Ltd.
- CODD, L.E.W. & VAN DER SCHYFF, H.P. 1951. Bome en struike van die Nasionale Kruger Wildtuin. Plantkundige opname pamflet No. 26. Departement van Landbou, Unie van Suid-Afrika.
- DE WET, J.A.L. 1983. Rooigras seed awns cause problems in sheep. Journal of the South African Veterinary Association, 54, 141-142.
- DE WET, J.A.L. & BATH. G.F. (eds.) 1994. Kleinveesiektes. Kaapstad: Tafelberg-uitgewers.
- EVERIST, S.L. 1974. Poisonous plants of Australia. Sydney: Angus and Robertson, Publishers.
- GIBBS RUSSELL, G.E., WATSON, L., KOEKEMOER, M., SMOOK, L., BARKER, N.P., ANDERSON, H.M. & DALLWITZ, M.J. 1990. Grasses of Southern Africa. Memoirs of the Botanical Survey of South Africa No. 58. South Africa: National Botanical Research Institute.
- GREEN, J.R. & KLEYNHANS, J.E. 1989. Suspected vetch (Vicia benghalensis L) poisoning in a Friesland cow in the Republic of South Africa. Journal of the South African Veterinary Association, 60, 109-110.
- GUZZO, C.A., LAZARUS, G.S., & WERTH, V.P. 1996. Respiratory system4. Dermatological pharmacology. In: HARDMAN. J.G., LIMBIRD, L.E., MOLINOFF, P.B. RUDDON, R.W. &· GILMAN, A.G. (eds.) Goodman & Gilman's The Pharmacological Basis of Therapeutics. New York: McGraw-Hill.
- HARPER, P.A.W., COOK, R.W., GILL, P.A., FRAZER, G.C., BADCOE, L.M. & POWER, J.M. 1993. Vetch toxicosis in cattle grazing Vicia villosa ssp. dasycarpa and V. benghalensis. Australian Veterinary Journal, 70, 140-144.
- HEMKEN, R.W., JACKSON Jnr. J.A. & BOLING, J.A. 1984. Toxic factors in tall fescue. Journal of Animal Science, 58, 1011-1016.
- HENNING, M.W. 1932. Animal diseases in South Africa. South African Agricultural Series. Vol XII. South Africa: Central News Agency, Ltd.
- HINTIKKA, EEVA-LIISA. 1977. Stachybotryotoxicosis as a veterinary problem. In: RODRICKS, J.V., HESSELTINE, C.W. & MEHLMAN, M.A. (eds.) Mycotoxins in human and animal health. Park Forest South, Illinois: Pathotox Publishers.
- HINTIKKA, EEVA-LIISA. 1978. Stachybotryotoxicosis in cattle and captive animals. In: WYLLIE, T.D. & MOREHOUSE, L.G. (eds.) Mycotoxic fungi, mycotoxins, mycotoxicoses. An encyclopedic handbook, Vol. 2. New York and Basel: Marcel Dekker, Inc.
- HINTIKKA, EEVA-LIISA. 1978. Stachybotryotoxicosis in horses. In: WYLLIE, T.D. & MOREHOUSE, L.G. (eds.) Mycotoxic fungi, mycotoxins, mycotoxicoses. An encyclopedic handbook, Vol. 2. New York and Basel: Marcel Dekker, Inc.
- HINTIKKA, EEVA-LIISA. 1978. Stachybotryotoxicosis in swine. In: WYLLIE, T.D. & MOREHOUSE, L.G. (eds.) Mycotoxic fungi, mycotoxins, mycotoxicoses. An encyclopedic handbook, Vol. 2. New York and Basel: Marcel Dekker, Inc.
- ISA, J.S.W., MAGEMBE, S.R. & NAUDÉ, T.W. 2001. Devil's claw (Harpagophytum procumbens) in a Brahman's preputial sheath: A case report from Botswana. Journal of the South African Veterinary Association, 72, 55-56
- IVIE, G.W. 1978. Toxicological significance of plant furocoumarins. In: KEELER, R.F., VAN KAMPEN, K.R. & JONES, L.F. (eds.) Effects of poisonous plants on livestock. Proceedings of a Joint United States-Australia Symposium on poisonous plants, Utah State University, Logan, Utah, June 19-24, 1977. New York: Academic Press.
- JACKSON, J.J., NEEDHAM, A.J.E. & LAWRENCE, J.A. 1968. Some recent investigations into Rhodesian toxic plants. Transactions. Zimbabwe Scientific Association, 53, 9-12.
- JONES, T.C. & HUNT, R.D. 1983. Veterinary Pathology, 5th edn. Philadelphia: Lea & Febiger.
- KINGHORN, A.O. 1985. Skin-irritant and tumour-promoting compounds of the Euphorbiaceae. In: SEAWRIGHT, A.A., HEGARTY, M.P., JAMES, L.F. & KEELER, R.F. (eds.) Plant toxicology: Proceedings of the Australia-USA Poisonous Plant Symposium, Brisbane, Australia. Queensland Poisonous Plant Committee: Yeerongpilly.
- KINGSBURY, J.M. 1964. Poisonous plants of the United States and Canada. Englewood Cliffs, New Jersey: Prentice-Hall. Inc.
- LASKIN, J.D. 1994. Cellular and molecular mechanisms in photochemical sensitization: Studies of the mechanism of action of psoralens. Food and Chemical Toxicology, 32, 119-127.
- MANTLE, P.G. 1978. Ergotism in cattle. In: WYLLIE, T.D. & MOREHOUSE, L.G. (eds.) Mycotoxic fungi, mycotoxins, mycotoxicoses. An encyclopedic handbook, Vol. 2, New York and Basel: Marcel Dekker, Inc.
- MEESKE, R. 1995. Klossiegras kan groot probleme veroorsaak. ARC-Irene Animal Production Institute, Agricultural news, 23 January 1995.
- NOBRE, D., DAGLI, M.L.Z. & HARAGUCHI, M. 1994. Crotalaria juncea intoxication in horses. Veterinary and Human Toxicology, 36, 445-448.
- OLSON, C. 1969. Bovine hyperkeratosis (X-disease, highly chlorinated naphthalene poisoning). Historical review. Advances in Veterinary Sciences and Comparative Medicine, 13, 101-157.
- PALMER, EVE & PITMAN, NORAH, 1972. Trees of Southern Africa. Vol. II. Cape Town: A. Balkema.
- PANCIERA, R.J. 1978. Hairy vetch (Vicia villosa Roth) poisoning in cattle. In: KEELER, R.F., VAN KAMPEN, K.R. & JONES, L.F. (eds.) Effects of poisonous plants of livetock. New York, San Francisco, London: Academic Press.
- PANCIERA, R.J., JOHNSON, L., OSBURN, B.I. 1966. A disease of cattle grazing hairy vetch (Vicia villosa Roth) pasture. Journal of the American Veterinary Medical Association, 148, 804-808.
- QUIN, J.I. 1933. Studies on the photosensitization of animals in South Africa. III. Photodynamic action of Hypericum aethiopicum var. glaucescens Sond. and Hypericum leucoptychodes (Syn. lanceolatum Lam). Onderstepoort Journal of Veterinary Science and Animal Industry, 1, 491-496.
- REYNOLDS, J.E.F. 1993. (ed.) Martindale. The Extra Pharmacopeia. London: The Pharmaceutical Press.
- ROWE, L.D. & NORMAN, J.O. 1989. Detection of phototoxic activity in plant specimens associated with primary photosensitization in livestock using a simple microbiological test. Journal of Veterinary Diagnostic Investigation, 1, 269-270.
- SCHIEFER, H.B. 1985. Dynamics of experimental trichothecene mycotoxicosis. In: LACEY, J. (ed.) Trichothecenes and others mycotoxins. Proceedings of the International Mycotoxin Symposium. Sydney, Australia: John Wiky & Sons, Ltd.
- SCHNEIDER, D.J., MARASAS, W.F.O., KUYS, J.C.D., KRIEK, N.P.J. & VAN SCHALWYK, G.C. 1979. Field outbreak of suspected stachybotryotoxicosis in sheep. Journal of the South African Veterinary Association, 50, 73-8 I.
- SCHNEIDER, D.J., MILES, C.O., GARTHWAITE, I., VAN HALDEREN, A., WESSELS, J.C. & LATEGAN, H.J. 1996. First report of field outbreaks of ergot-alkaloid toxicity in South Africa. Onderstepoort Journal of Veterinary Research, 63, 97-108.
- SHONE, D.R., PHILIP, J.R. & CHRISTIE, G.J. 1959. Agalactia of sows caused by feeding the ergot of the bullrush millet, Pennisetum typhoides. The Veterinary Record, 71, 129-132.
- STEYN, D.G. 1931. Investigation into the cause of alopecia (kaalsiekte) in kids and goats. Report on Veterinary Research, Union of South Africa, 17, 729-756.
- STEYN, D.G. 1934. Chrysocoma tennuifolia Berg. poisoning in Angora goats and the development of tolerance. Report on Veterinary Research, Union of South Africa, 18, 893-898.
- STEYN, D.G. 1934. The toxicology of plants in South Africa. South Africa: Central News Agency, Ltd.
- STEYN, D.G. 1941. Vergiftiging van mense met wilde plante, sierplante en huishoudelike gifstowwe. Boerdery in Suid-Afrika, 16, 11-18.
- STEYN, D.G. 1949. Vergiftiging van mens en dier. Pretoria: Van Schaik.
- STEYN, D.G. & VAN DER WALT, S.J. 1945. Jaagsiekte by perde en Sunn-hennop-vergiftiging by vee. Boerdery in Suid-Afrika, 20, 445-447.
- STIRTON, C.H. (ed.) 1978. Plant invaders, beautiful but dangerous. Publication of the Department of Nature Conservation of the Cape Provincial Administration, Cape Town, ISBN 7984-0092-7.
- STROEBEL, J.C. 2002. Induction of photosensitivity in sheep with Erodium moschatum (L.) L'Hérit. Journal of the South African Veterinary Association, 73, 57-61.
- THEILER, A. 1911. Stiff sickness or stywesiekte in cattle. Agriculture Journal of the Union of South Africa, 1, 10-21.
- THEODORIDIS, A. & COETZER, J.A.W. 1979. Subcutaneous and pulmonary emphysema as complications of bovine ephemeral fever. Onderstepoort Journal of Veterinary Research, 46, 125-127.
- THOMAS, A.D. 1931. Actinobacillosis and other complications in sheep which may arise from the feeding of prickly pear, Opuntia spp. 17th Report of the Director of Veterinary Services and Animal Industry, Union of South Africa, 215-229.
- TOR-AGBIDYE, J., BLYTHE, L. & CRAIG, A.M. 2001. Correlation of endophyte toxins (ergovaline and lolitrem B) with clinical disease: Fescue foot and perennial ryegrass staggers. Veterinary and Human Toxicology, 42, 140-146.
- UENO, Y. & ISHII, K. 1985. Chemical and biological properties of trichothecenes from Fusarium sporotrichioides. In: LACEY, J. (ed.). Trichothecenes and other mycotoxins. Proceedings of the International Mycotoxin Symposium. Sydney, Australia: John Wiley &. Sons Ltd.
- VAHRMEIJER, J. 1981. Poisonous plants of Southern Africa that cause stock losses. Cape Town: Tafelberg.
- VAN DER VYVER, F.H., KELLERMAN, T.S., BASTIANELLO, STELLA, S., DE WET, J.A.L., JOUBERT, J.P.J & FAUL, ADELE 1985. Valsiekte (falling disease): A nervous disorder in lambs suspected of being caused by the plant Chrysocoma tennuifolia. Journal of the South African Veterinary Association, 56, 65-68.
- VAN NIEKERK, F.A. 1994. Suspected Ammi majus toxicity. VetMed, Autumn 1994, 43.
- VILJOEN, P.R. 1918. Preliminary report on the harmful effects of 'steek' grass on the general health and condition of sheep. 5th and 6th Reports of the Director of Veterinary Research, Union of South Africa, 323-333.
- WANNEMACHER, R.W., RUNNER, D.L., PACE, J.G., NEUFELD, H.A., BRENNECKE, L.H. & DINTERMAN, R.E. 1985. Dermal toxicity of T-2 toxin in guinea-pigs, rats and cynomolgus monkeys. In: LACEY, J. (ed.) Trichothecenes and others mycotoxins. Proceedings of the International Mycotoxin Symposium. Sydney, Australia: John Wiley & Sons, Ltd.
- WATT, J.M. & BREYER-BRANDWIJK, M.G., 1968. The medicinal and poisonous plants of Southern and Eastern Africa. 2nd edn. Edinburgh and London: E & E Livingstone, Ltd.
- WOODS, A.J., JONES, J.B. & MANTLE, P.G. 1966. An outbreak of gangrenous ergotism in cattle. The Veterinary Record, 78, 742-7-19.
- YAGER, JULIE, A. & SCOTT, D.W. 1985. The skin and appendages. In: JUBB, K.V.F., KENNEDY, P.C. & PALMER. N. (eds.) Pathology of domestic animals, 3rd edn. Vol. I. Orlando. Florida, USA: Academic Press.