This content is distributed under the following licence: Attribution-NonCommercial CC BY-NC  View Creative Commons Licence details here
View Creative Commons Licence details here

Introduction
Pulmonary oedema and emphysema are non-specific changes associated with many infectious, parasitological and toxicological conditions in livestock. However, in most diseases these changes are usually mild, develop terminally, are generally not associated with overt and prolonged signs of respiratory distress, and, apart from oedema and emphysema, no other significant changes are usually discernible macro- or microscopically in the lungs.
On the other hand, respiratory distress associated with marked oedema and/or emphysema, coupled with noteworthy microscopical changes in the lungs, rarely occurs in stock in South Africa and has been associated with poisoning by only a few plants, such as Crotalaria spp. and Hertia pallens; also with a variety of lush green pastures containing L-tryptophan, which is responsible for acute bovine pulmonary oedema and emphysema or fog fever; or with an ephemeral fever virus.
Crotalaria spp. (Fabaceae)
C. dura Wood & Evans
Wild lucerne, wilde lusern, jaagsiektebossie
C. globifera E. Mey.
Wild lucerne, wilde lusern, jaagsiektebossie
C. juncea L.
Sunn-hemp, Sunn-hennep
C. spartioides DC.
Dune bush, duinebos, besembossie
Crotalaria spp., such as C. dura,19, 47, 49, 50, 55, 58 C. globifera31, 49, 53 and C. juncea,53, 58 have been associated in South Africa with a chronic respiratory disease called jaagsiekte (Crotalariosis equorum) in horses and mules. The disease is characterized by fever, polypnoea, dyspnoea, subcutaneous and pulmonary emphysema, interstitial pneumonia and sometimes also fibrosis or cirrhosis of the liver.19, 31, 47, 49, 50, 53, 55, 58
Crotalaria dura (Figures 1 and 2) is a low, spreading bush with trifoliate leaves and typical yellow pea-flowers that are borne in terminal racemes, 28 mm long. The fruits are subglobose, densely hairy pods, 10 mm long and 8 mm in diameter.36 The leaves of C. globifera differ from those of C. dura in that the former are minutely hairy. The yellow pea-flowers are arranged in terminal racemes 60–80 mm long and the plant bears pods c.6 mm in diameter with a dense covering of fine hairs.36
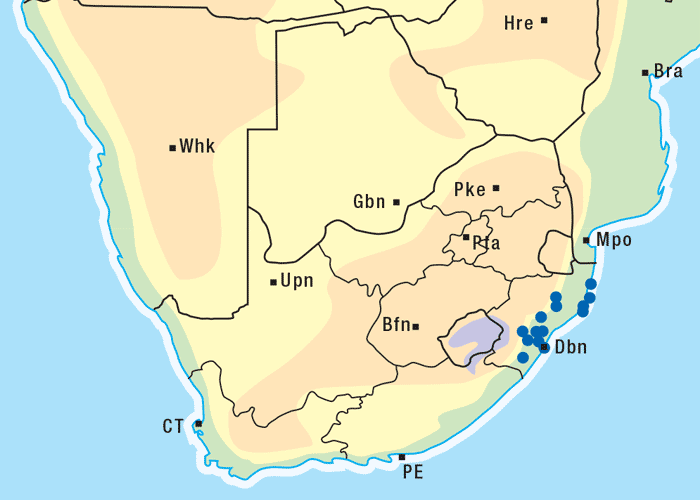
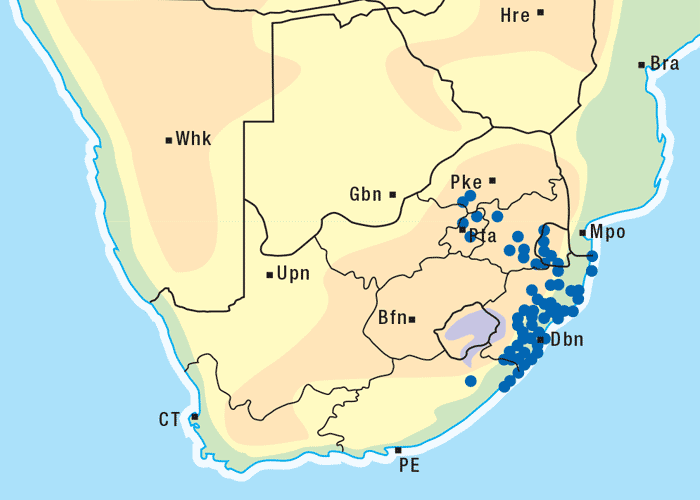
Crotalaria dura and C. globifera occur in certain parts of KwaZulu-Natal, where the disease was first noticed55 and still occurs today,27 and the former Transvaal (Figures 3 and 4).19 In the original elucidation of the aetiology of jaagsiekte, Theiler55 described a number of feeding experiments on horses using C. dura at the Allerton Laboratory and at Onderstepoort. During November 1915, a farmer from Singletree in KwaZulu-Natal submitted hay contaminated with C. dura to the Allerton Laboratory for toxicity trials. A horse was fed with 126 kg of this hay in 16 days, namely an average of 7,8 kg/day. The animal died 109 days after the commencement of the trial or 93 days after feeding had ceased. Another horse, at Onderstepoort, consumed 20,9 kg of C. dura (collected at Singletree) mixed in food over a period of 23 days. Ten days after feeding had started the horse developed a fever which lasted about 12 days. On the seventy-fifth day, the horse was again feverish and died six days later of jaagsiekte. Theiler55 conducted many feeding trials with C. dura and found the minimal quantity of plant material required to induce jaagsiekte to be c.21 kg of C. dura consumed over 23 days. When larger quantities were fed for shorter periods the disease likewise made its appearance. The disease developed after a period of apparent normality which varied among individual horses and in different experiments. Taking into account only those experiments that gave positive results after the shortest feeding trials, Theiler55 found that this period might be fixed at c.50 days. The disease lasted for 6–29 days and fever was in some instances present before the signs of jaagsiekte were evident.
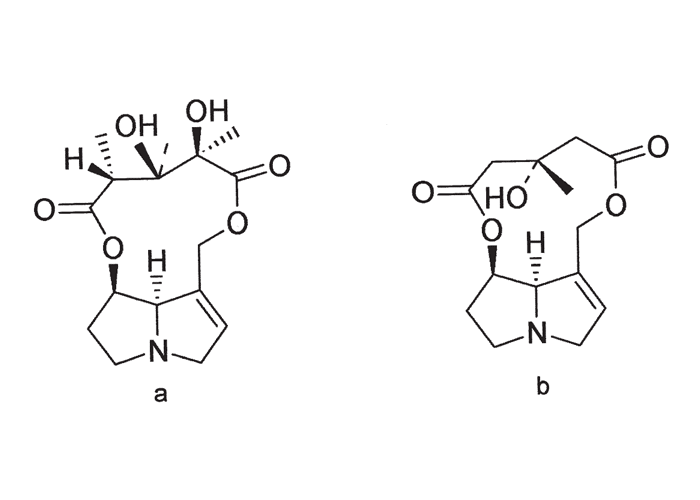
Some years later Steyn47 found evidence to support Theiler’s findings that C. dura is the cause of jaagsiekte. He induced the disease in a horse by feeding it with dry C. dura plants that had been collected in the late seeding stage, and had been stored for five years. According to Steyn49 and Marais,30 fresh C. globifera in the flowering and seeding stages, fed at a level of 90–180 g daily over 116 days, caused the death of a horse from jaagsiekte on Day 128 (Allerton Laboratory, Experiment 141, 27/1127). Though no South African experimental data could be traced on the toxicity of C. juncea, Steyn and Van der Walt53 and Vahrmeijer58 referred to it as an additional cause of jaagsiekte in horses58 and the condition has been induced experimentally with the plant abroad. Loss of wool has been reported in sheep after consuming large quantities of C. juncea53 (see The skin and adnexa). The toxic principle of both C. dura and C. globifera has been shown to be a pyrrolizidine alkaloid, dicrotaline (Figure 5).30
Jaagsiekte was at first thought to be an infectious disease, mainly on account of the lung lesions and the fever. However, the fever that was noticed during the period the horses received the plant was not present in all animals. In some horses the fever started a few days after commencement of feeding of C. dura and lasted for 10–14 days. The body temperature then returned to normal for a varying period, but was interrupted by intermittent attacks of fever, after feeding with the plant had ceased and before the disease was manifested in full. In most cases, the final fever, which was accompanied by severe respiratory signs, reached the maximum on the evening preceding death.
A characteristic clinical sign of the cases of jaagsiekte described by Theiler,55 was a sudden increase in the respiratory rate to levels of 100–120/min. The respiration in the affected animals tended to be very superficial and was either costo-abdominal or abdominal in character. In some horses a double movement was apparent, and the groove along the costal arch was more pronounced, especially in horses where the expiratory movements were accentuated. Dyspnoea in horses suffering from jaagsiekte was never as pronounced as it has been found to be, for instance, in cases of horse sickness. Although a dry cough was rarely noticed in affected animals, some farmers claim this to be one of the first signs. Other signs included rales on auscultation of the lungs, stridor, increased heart rate and restlessness. Terminally there was loss of appetite and loss of condition, yet, although it appeared dull and listless, the animal remained on its feet until death. When sufficient plants had been eaten, or once the disease had manifested itself, death was inevitable.
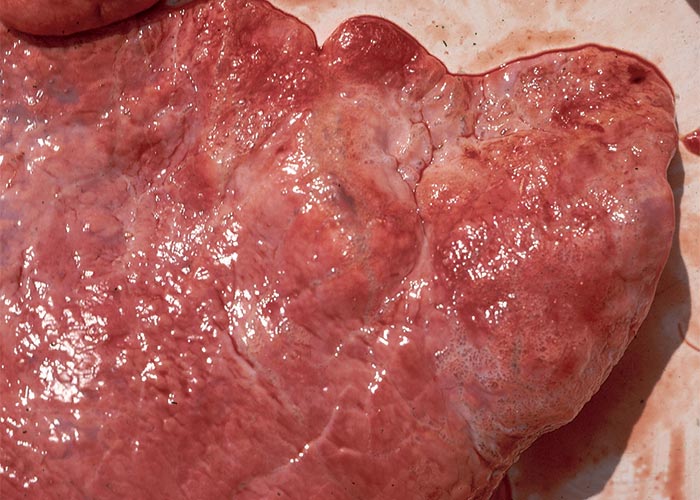
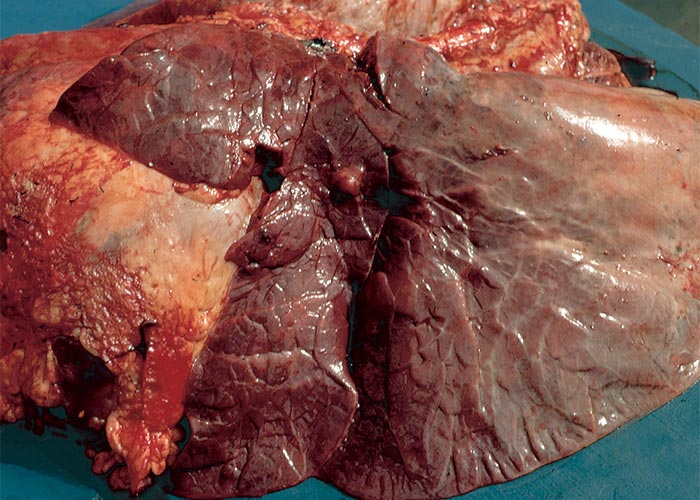
At necropsy, pulmonary emphysema was a striking feature in the majority of the cases studied by Theiler. The lungs were severely inflated, did not collapse when the thorax was opened, rib-markings were evident on the surface of the lungs and bullae, 30–50 mm in diameter, were sometimes evident, especially at the apices of the lungs. Generally, the lungs had a mottled appearance as a result of areas of atelectasis, and coalescing, pneumonic foci of various sizes throughout the organ. The foci had an increased consistency, appeared flesh-like, had thickened gelatinous and oedematous septae, and were either greyish or reddish in colour (Figure 6). Apart from the lung lesions, some horses had emphysema of the subcutaneous tissues in the neck, shoulder, back, loin, peritracheal and perioesophageal regions, subpleurally around the pericardium and in the mediastinum, as well as subperitoneally in the abdominal cavity. The mediastinal and bronchial lymph nodes were also emphysematous. Hydrothorax and hydropericardium sometimes completed the necropsy picture.
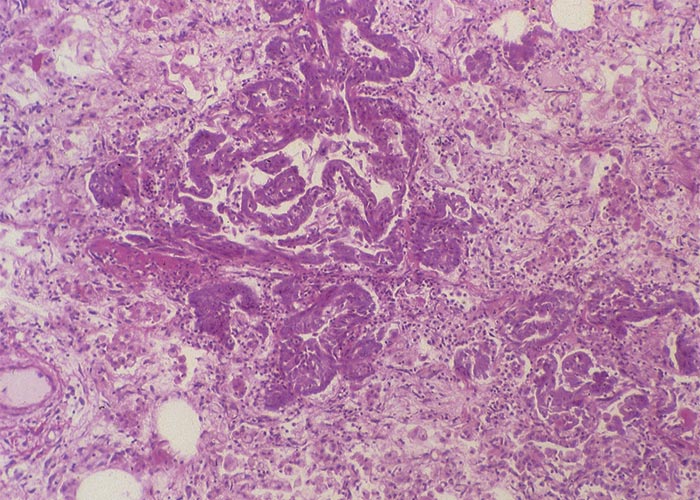
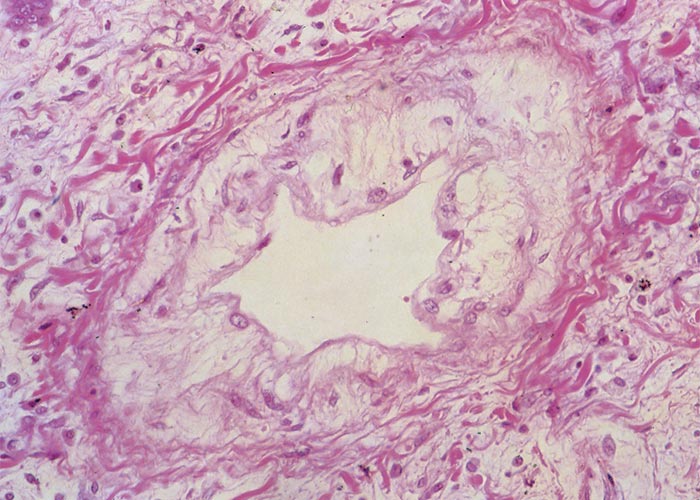
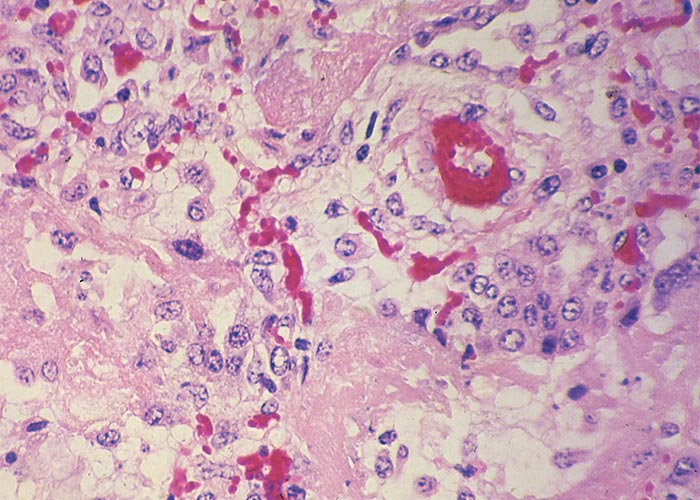
Microscopically, the lumina of the larger bronchi were distended and the lining epithelium flattened, while in the smaller bronchioles desquamation of the epithelial cells was evident, and their lumina were filled with plugs of mucus, epithelial cells and sometimes also neutrophils. Theiler55 described desquamation of epithelial cells in the alveoli as a very striking feature. Some alveoli were filled with these desquamating cells, oedematous fluid and sometimes also fibrin and giant cells. Connective tissue, lymphocytes and polymorphonuclear cells were evident in consolidated areas of the lung. He regarded proliferation of the epithelium of the bronchioles (Figure 7) as a pathognomonic lesion of jaagsiekte. According to him, two types of bronchioles could be distinguished, namely, budding ducts formed by a column of cuboidal cells without any lumen and the differentiated bronchioles with lumens which were lined with several layers of cubic polyhedral epithelial cells. Their lumina were often plugged with epithelial cells and polymorphonuclear cells, while the bronchioles were surrounded by an increased amount of connective tissue (Figure 7). In all cases, the epithelial lining had a peculiar brownish tinge. The newly formed bronchioles frequently underwent necrosis. Re-examination of historic jaagsiekte specimens revealed that many of the blood-vessel walls in the lungs were thickened by oedema (Figure 8) and that hypertrophy of the muscle fibres in the tunica media had occurred (J.A.W. Coetzer, VRI, Onderstepoort, personal observations, 1986).
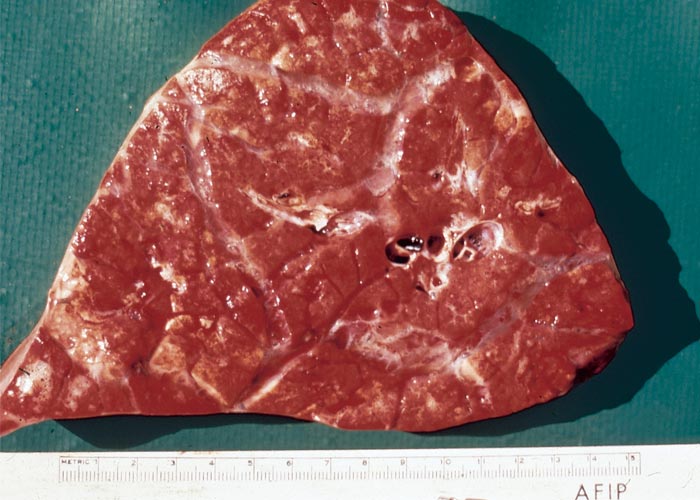
In some instances the liver was reduced in size, pale-brown in colour and of firm consistency. Microscopically, the extent of involvement varied among the animals. A conspicuous change was the proliferation of bile ductules at the periphery of the lobules, some of which were found intermingled with hepatocytes. In addition to fibroplasia of the portal triads and fibrosis around some central veins, megalocytosis and karyomegaly of hepatocytes were also evident. Theiler55 interpreted the hepatic changes as compatible with those seen with ‘parenchymatous hepatitis and of a commencing cirrhosis’, which he ascribed to seneciosis or pyrrolizidine alkaloidal poisoning.
Apart from his work on jaagsiekte in horses, Theiler also fed C. dura to cattle. A bull died on its own and an ox was killed in extremis on Days 65 and 99 respectively of the experiment, after each had received 0,9 kg plant material daily. Clinical signs included anorexia, a black liquid diarrhoea and reduction in mass, but there were no signs of respiratory involvement. In the case of the ox, death was preceded by a fever. The most notable lesions were found in the liver which, according to Theiler, resembled ‘certain stages of cirrhosis of “dunsiekte” (seneciosis) in horses’ and were also very similar to those seen in horses poisoned with C. dura. The lungs, however, did not show any lesions.
Steyn and De Kock52 drenched dry C. dura in the late stage of seeding to sheep at a daily rate of 100–400 g. Depending on the level of dosage, animals died between 10 and 135 days after commencement of the experiment. Clinical signs and lesions in the sheep were very similar to those reported in cases of jaagsiekte in horses.55
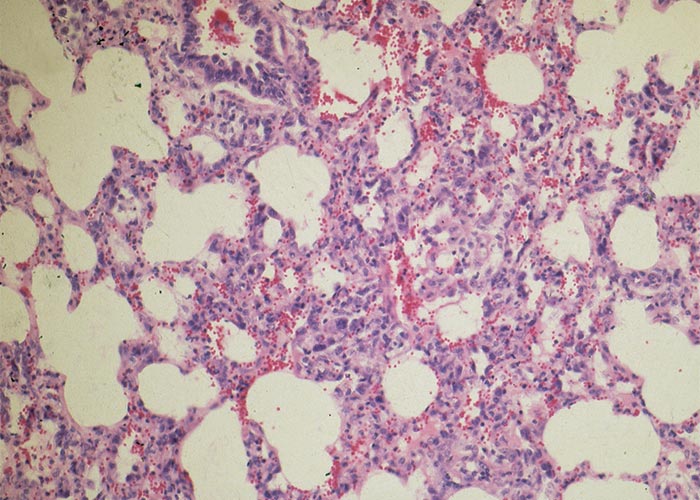
Crotalaria spartioides, or the dune bush, which grows particularly abundantly in the Kuruman district of the Northern Cape Province, is another toxic species that has occasionally been associated with losses in cattle. Apart from a few unpublished field observations and pilot feeding experiments in cattle with C. spartioides, no authentic reports of poisoning by this plant could be traced. The botanical features and distribution of the plant, as well as information on these field outbreaks and experiments, are given in Liver. Recently, it was found for the first time that, apart from hepatic lesions compatible with poisoning by the pyrrolizidine alkaloids, the plant can also be responsible for severe lung lesions in cattle almost identical with those described in cases of jaagsiekte (J.A.W. Coetzer and M. Silkstone, VRI, Onderstepoort, unpublished observations, 1985). The microscopical lesions in the lungs include (Figure 9) a mild to moderate interstitial pneumonia and emphysema, characterized by thickening of the alveolar septae by what appeared to be fibroblasts, macrophages, lymphocytes and some connective tissue; hyperplasia of the epithelium of especially the smaller bronchioles; hypertrophy and sometimes also hyaline degeneration of the walls of the arterioles and smaller arteries; and karyomegaly of some of the alveolar epithelial cells. Although the toxic principle of C. spartioides has not been isolated, it is assumed to be a pyrrolizidine alkaloid on account of the liver lesions induced by it.
It is known that a wide variation in susceptibility exists between animal species and that the target organ affected may also vary in response to different pyrrolizidine alkaloids.20, 21, 31, 44 Generally, the alkaloids or their metabolites affect primarily the liver, lungs and kidneys, and one or more of these organs may be involved.
A number of Crotalaria spp. have been associated worldwide with poisoning in animals. Crotalaria retusa and C. spectabilis, which contain monocrotaline (Figure 5), are primarily responsible for a severe nephrosis and interstitial pneumonia in pigs, liver and lung lesions in cattle, and liver and kidney damage in chickens.21, 34, 43 The alkaloids, fulvine and crispatine of C. crispata, which are closely related to dicrotaline, affect the lungs of horses and the liver and lungs of rabbits;12 and C. pallida (= C. mucronata) has been associated with liver and lung lesions in sheep.28
Finally, it should be emphasized that, although C. spartioides poisoning still occurs from time to time in cattle, jaagsiekte of horses now is rather rare, as horses are mostly stall fed on commercial rations and good quality forage.
Lasiospermum bipinnatum (Thunb.) Druce (Asteraceae)
Ganskweek
In addition to the zonally arranged liver lesions and hepatogenous photosensitization typical of ganskweek poisoning (see Liver), Williams64 described pulmonary lesions in lambs dosed with a specific consignment of the plant. Macroscopically these included pulmonary, mediastinal and subcutaneous emphysema, congestion and oedema of the lungs. Clara cell hypertrophy and hyperplasia and interstitial pneumonia were the most important microscopical features noted.64 Penrith and Van Vollenhoven35 confirmed these findings in a field outbreak of ganskweek poisoning of cattle in the Molteno district of the Eastern Cape Province; where, in addition to hepatic changes, they also observed pulmonary lesions such as emphysema, bronchiolar dilatation, proliferation of non-ciliated epithelial (Clara) cells and interstitial pneumonia.35
Acute bovine pulmonary oedema and emphysema (ABPE)
A typical interstitial pneumonia, fog fever, cow asthma, green grass sickness
ABPE, or fog fever as it is often referred to in Great Britain because of the occurrence of acute respiratory distress in cattle on green sward (foggage), develops within 1 to 14 days after animals have been changed from sparse, poor-quality forage to lush, rapid growing pastures.3, 4, 11, 46 In South Africa, the disease is occasionally reported in cattle grazing on artificial pastures (J.A.W. Coetzer, VRI, Onderstepoort, unpublished observations, 1985).
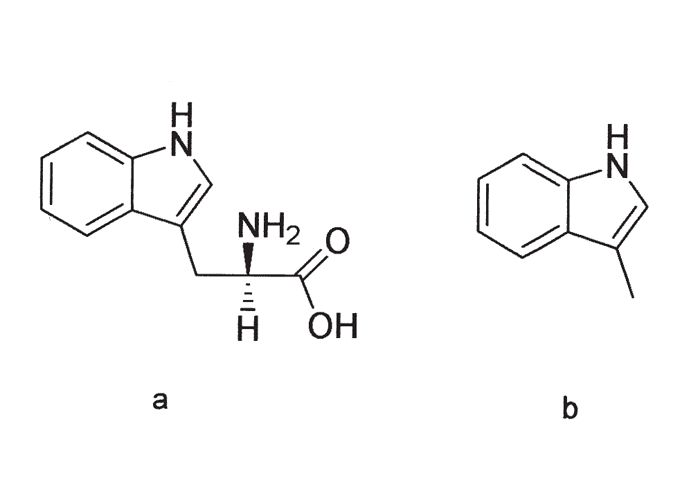
ABPE is, strictly speaking, not a plant poisoning nor is it associated with the consumption of any particular plant species. A variety of plants, mostly considered to be nontoxic, such as kale, rape, turnip tips, lucerne, Bermuda grass (Cynodon spp.), mixed meadow grass, reed canary grass, grains, ryegrass and many others, which contain the amino acid, L-tryptophan (Figure 10), have been linked with outbreaks of the disease.3, 4 A similar condition has also been reported with perilla ketone, the lung toxin in the weed purple mint (Perilla frutescens), and with mouldy sweet potatoes in cattle in Japan and the USA.65 Several 3-substituted furans, such as 4-ipomeanol (which is most abundant and most toxic), 1-ipomeanol and 1,4-ipomeadiol have been isolated from sweet potatoes (Ipomoea batatas) infected with the black-rot fungus pathogen Ceratocystis fimbriata, or damage by other fungi, e.g. Fusarium solani or chemicals such as mercuric chloride.65 These stress metabolites are formed by the host tissue in response to several exogenous stimuli. Contrary to what was believed previously, ABPE does not result from hypersensitivity to lung worms (Dictyocaulus viviparus) or mouldy hay containing Micropolyspora faeni, which results in extrinsic allergic alveolitis similar to farmer’s lung disease.4
There is clear evidence that L-tryptophan is required in the diet for the induction of ABPE.3, 4 It has been demonstrated that L-tryptophan is fermented in the rumen by different bacteria. First, L-tryptophan is deaminated and decarboxylated to indolacetic acid (IAA), then by decarboxylation of the IAA to 3-methylindole (3-MI) (Figure 10), which is pneumotoxic.3
The latter is rapidly absorbed from the rumen and within 30 minutes ultrastructural changes can be evident in the non-ciliated bronchiolar and alveolar type l cells.2, 3 It is interesting to note that D-tryptophan is not degraded to 3-MI and is thus not associated with ABPE. The production of 3-MI from L-tryptophan is influenced by factors that affect ruminal fermentation, such as type of diet, pH, etc.3 It has been pointed out that, because ruminal fermentation is variable, the L-tryptophan content of pastures where ABPE occurs and pastures where the disease is absent, does not always differ significantly.4
Carlson and Dickinson4 showed that pulmonary lesions could be induced in 50% of cows within four days after being dosed with 0,35 g L-tryptophan per kg body mass. ABPE was also induced by a single intraruminal dose of 3-MI (0,1–0,2 g/kg body mass) and by intravenous infusion of 3-MI (0,06 g/kg body mass) over 12 hours.5, 6 It was demonstrated that the incidence and severity of pulmonary lesions are not necessarily dependent on the size of the dose. It is not known how much tryptophan is ingested during an outbreak of ABPE. Although this condition occurs mostly in cattle, Carlson and Breeze3 referred to various workers who studied the effects of 3-MI on the lungs of sheep, goats and horses.2, 5, 22
ABPE occurs in cattle of both sexes but would seem to affect cows over two years most frequently.3, 4 Clinical signs include increased respiration, laboured breathing and, in severe cases, an expiratory grunt, an extended and lowered head, frothing at the mouth and a protruding tongue. Morbidity ranges between zero and 50%, while 30% of affected animals die within two to four days.
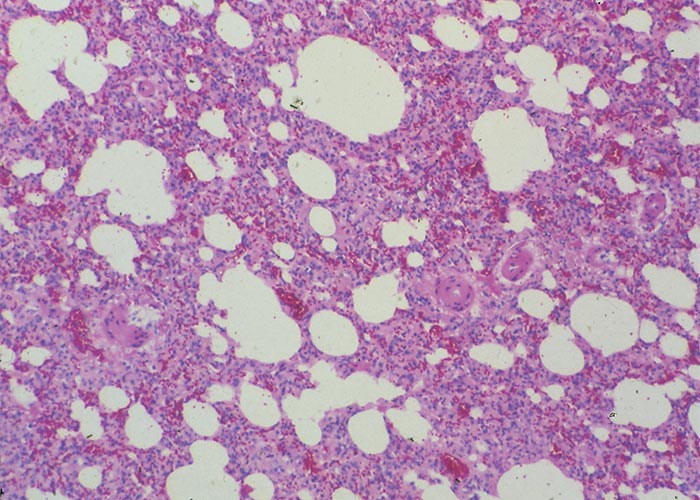
Macroscopically, the lungs are heavier than normal and dark red (Figures 11 and 12); the consistency is firm and rubbery; on cut surface, a protein-rich, almost gelatinous fluid oozes from the lung and is also present in the airways; and there is emphysema and bullae-formation. Microscopically, the pulmonary lesions are characterized by a protein-rich lung oedema, hyaline membrane formation in the alveoli, thickening of the alveolar septae, diffuse alveolar and bronchiolar epithelium hyperplasia and infiltration of some eosinophils, neutrophils and macrophages in the parenchyma and lumina of the alveoli (Figure 13).3, 11, 29, 38
Although there is no effective treatment for animals suffering from ABPE, the incidence of the disease can be reduced by management practices and by administering compounds which inhibit the production of 3-MI in the rumen.14 Ranchers claim to have reduced the incidence of ABPE by gradually exposing cattle to lush pastures, using movable fences or by supplementing hay for the first 10–14 days after pasture changes in an effort to reduce the intake of lush forage and to allow for ruminal adaptation to new conditions. The iodonium compounds in particular, but also monensin15–17 and chlortetracycline, have been shown to reduce the formation of 3-MI.3, 4
Gnidia spp. (Thymelaeaceae)
G. burchellii (Meisn.) Gilg. (= Lasiosiphon burchellii Meisn.)
Harpuisbos, repuisbossie
G. anthylloides (L.f.) Gilg.
G. polycephala (C.A. Mey.) Gilg. (= Arthrosolen polycephalus C.A. Mey.)
Januariebos, besembossie
Gnidia burchellii is responsible for acute deaths in sheep, as well as pulmonary and subcutaneous emphysema, and a malodorous diarrhoea in more protracted cases.48, 54, 58
The plant is described and additional information given concerning its effect on the gastrointestinal tract in Gastrointestinal tract.
The harpuisbos grows in stony and sandy soil on hill and valley slopes, especially in trampled areas in the south-western Mpumalanga, north-eastern Free State, north-western KwaZulu-Natal and the Northern Cape Province.58 Gnidia burchellii poisoning occurs sporadically, and field outbreaks have been reported only in sheep. Adelaar referred to an outbreak of poisoning in the Dundee district, where 68 sheep died, showing marked emphysema of the lungs, mediastinum and the subcutaneous tissues (see Gastrointestinal tract). The toxic principle of G. burchellii is a 12-hydroxy analogue (C27H30O9) of daphnetoxin,8, 9, 37 a phorbol-derived diterpenoid ester (see Gastrointestinal tract).
A furanosesquiterpene and potent antileukaemic diterpenoid esters have been isolated from G. latifolia found in Kenya.26 Van der Walt and Steyn59 found G. burchellii negative for prussic acid. A severe burning in the nose, throat and chest, as well as dyspnoea, were experienced by a worker milling the dry plant1. He also described a burning sensation of the tongue and throat when the ground plant was applied to the tip of the tongue.
Terblanche et al.54 found the MLD of an acetone extract of G. burchellii for sheep to be c.40 mg/kg, which is equivalent to 1,8 g/kg of dried plant. They administered the extract per ruminal fistula to nine sheep at a dosage level that ranged from 20–200 mg/kg body mass. Some animals died acutely within 24–48 hours after dosing without showing any clinical signs, while in others the respiration was accelerated and characterized by a short inspiration and a prolonged forced expiration. These respiratory signs were sometimes accompanied by groaning sounds and, on auscultation, sharp, dry alveolar sounds were heard. Other signs included anorexia, haemorrhagic nasal discharge and head pushing, and one animal collapsed in spasms. The subacutely affected animals showed signs for a week or more before they recovered or died. In addition to dyspnoea and nasal catarrh, they also showed anorexia, ruminal atony, tachycardia and a fetid diarrhoea. In some animals, the diarrhoea persisted for weeks after the dyspnoea had disappeared.
According to Terblanche et al.,54 guinea-pigs are highly susceptible to this poisoning. The clinical signs in this species differ somewhat from those observed in sheep in that they neither showed dyspnoea nor groaned. Most fatal cases died within five to 20 hours or up to three days after dosing. The majority of the guinea-pigs showed paresis within 10 minutes of dosing, a reluctance to move and an incoordination which progressed to paralysis when the animals were forced to move. Diarrhoea and a staring coat were observed in the longer standing cases. These workers also produced signs of poisoning in rabbits, pigs and dogs by dosing them with extracts of the plant.
The necropsy findings in the sheep experimentally poisoned with G. burchellii included marked pulmonary emphysema, and moderate congestion and oedema of the lungs in the acute cases.54 In the subacutely affected animals, congestion and oedema of the lungs were more pronounced and the emphysema less marked, and there were also necrosis of lymphoid tissues, focal myocarditis, hyperaemia, sometimes focal haemorrhages in the alimentary tract and signs of diarrhoea.54
The Thymelaeaceae family, to which Gnidia belongs, is represented in South Africa by nearly 170 species.1 Several members of this family are used in tribal medicine and G. anthylloides and G. burchellii have been shown to have an irritating effect on the nasal and buccal mucosa of people working with the plant.1 Milled, air-dried, G. anthylloides in the flowering stage was dosed to sheep, cattle and a horse.1 The minimal toxic dose for the sheep was c.28 g and 148 g for the cattle and the horse. At the higher dosage level (112 g), a sheep died without showing any clinical signs of poisoning. At the lower levels (28–56 g) of dosing, the most important clinical signs and lesions in sheep included increased body temperature, polypnoea, costo-abdominal respiration, abdominal pain, diarrhoea, catarrhal gastroenteritis and haemorrhagic colitis. A bovine with a body mass of 180 kg received 336 g of this plant material and died 12 hours later, after it had showed salivation, distress and signs of abdominal pain (groaning and grinding of the teeth). Two bovines of c.273 and 455 kg body mass, each of which had been fed 168 g of G. anthylloides, died on Days 11 and 12 of the experiment respectively. They developed abdominal pain, salivation, profuse diarrhoea, increased respiration (mainly costal in nature with sharp inspiration and prolonged expiration) and subcutaneous emphysema. At necropsy, a haemorrhagic gastroenteritis, colitis, proctitis, focal ulcerative rumenitis and congestion, oedema and emphysema of the lungs were evident in both animals. A horse drenched with 560 g of G. anthylloides suffered extreme abdominal pain 20 hours after dosing and died four hours later. The most notable lesion in this animal was hyperaemia of the gastrointestinal tract.
Although the literature on the effect of G. polycephala or Januariebos on livestock is scant, poisoning of sheep and cattle has mainly been associated with severe diarrhoea, abomasitis and enteritis49, 58 (see Gastrointestinal tract).
Hertia pallens (DC.) Kuntze (Asteraceae)
Springbokbush, springbokbossie, malkopharpuis
Hertia pallens has been shown to be both hepatotoxic (see Liver) and pneumotoxic to sheep.39, 49 In some animals, respiratory distress was associated with emphysema of the subcutaneous tissues and lungs, with severe lung oedema, diffuse, mononuclear, interstitial pneumonia and hyperplasia of the epithelium of the smaller bronchi and bronchioli (Figure 14).39, 51
The botanical features and distribution of the plant and details from toxicity trials, clinical signs and lesions in sheep poisoned with this plant are given in Liver.
Fusarium verticillioides (Sacc.) Nirenberg (=F. moniliforme Sheldon) Fungi: Hyphomycetes
Kriek et al. (1981)25 reported that LEM-inducing isolates were toxic also to non-equidae and that the lesions were variously sited in the different species (Central nervous system). Although certain organs were constantly affected to some degree in all animals, the main target organ was different in each species; for instance, the liver and heart in rats, the liver in baboons, the brain in the horse, the kidneys and liver in sheep, and the lungs in pigs.25 Large outbreaks of porcine lung oedema, associated with the ingestion of F. verticillioides-infected commercial rations, maize or maize screenings, have been reported in the USA.10, 18, 33, 40, 41, 42, 66, 67 The clinical signs of the condition, known as ‘porcine pulmonary oedema syndrome’ (PES), included lethargy; dyspnoea with open-mouthed breathing; cyanosis; posterior paresis; abortions; recumbency and death.33 The onset was rapid, starting about five days after exposure, and death often ensued without observing premonitory signs. Abortions ceased after withdrawal of the incriminated feed.10, 18 Interstitial and interlobular pulmonary oedema, accompanied by hydrothorax, were the principal necropsy features.33, 40
A survey by Osweiler et al., 199233 of 16 field cases revealed that the morbidity ranged from 1–100% (mean 25,9%) and mortality from 2–100% (mean 70,3%). Samples of corn or screenings from 14 of the 16 cases contained 3–330 ppm FB1; five contained > 50 ppm and four contained > 100 ppm. In a feeding trial, screenings from one of the outbreaks containing 92 ppm FB1 and 28 ppm FB2 (total 120 ppm fumonisins) caused fatal PES in four of six weanling pigs.33 In addition to corn screenings, PES has been reproduced with pure FB1.10, 18 PES is suspected of occurring in South Africa (Mary-Louise Penrith, ARC-OVI, personal observation, 1999).
Discussion
While respiratory distress and lung emphysema are part of jaagsiekte and other pyrrolizidine alkaloid related diseases, acute bovine pulmonary oedema and emphysema (ABPE), or fog fever, and the viral disease, ephemeral fever,56 the lesions in the lungs and the mechanisms involved in the development of emphysema in these disorders are strikingly different. Jaagsiekte is a chronic disease in which the lesions develop over several weeks or even months. In addition to hyperplasia of the bronchiolar and alveolar epithelium, interstitial pneumonia and vascular changes are hallmarks of the lung pathology. In contrast to jaagsiekte, acute changes, such as oedema of a protein-rich nature, hyaline membrane formation and hyperplasia of alveolar and bronchiolar epithelial cells, are features of ABPE. In ephemeral fever, the essential lesions include inflammation of the bronchi, the bronchioli, and the tissues surrounding these structures.50
Apart from jaagsiekte in horses, which refers to a chronic interstitial pneumonia caused by different Crotalaria spp., the term jaagsiekte has also been coined for a bronchiolar-alveolar carcinoma in sheep as a result of a retrovirus.21, 57, 60–62
Horses may also develop a condition referred to as chronic bronchiolitis-emphysema complex, heaves or broken wind.23 Although other factors may possibly play a role in the development of this condition, inhaled allergens, such as fungal antigens in mouldy hay, bedding or stable dust, would seem to be the most important aetiological agents.23
Emphysematous lungs are usually moderately to severely inflated, do not collapse when the thoracic cavity is opened, have a puffy consistency and are lighter in colour than normal. In severe cases of pulmonary emphysema, rib-markings may be imprinted on the lungs and bullae of varying size may be evident in the parenchyma. In animals suffering from pulmonary emphysema, air often escapes from the lungs and migrates interstitially and subpleurally to involve the pericardium and mediastinum.56 From here, it progresses anteriorly through the thoracic inlet to the subcutaneous tissues of the neck, shoulder, back, etc. or moves posteriorly through the pillars of the diaphragm to the abdominal cavity, giving rise to subperitoneal emphysema of many abdominal organs.56 The bronchial and mediastinal lymph nodes in particular may also be emphysematous.
Apart from Crotalaria spp. other plants have also been associated from time to time with respiratory distress in livestock here and abroad. Brassica spp., which include weeds and forage crops, such as wild and cultivated mustards, kale, rape, turnips, Brussels sprouts, cabbage, etc. can be harmful to stock. These plants have been associated worldwide with a number of syndromes in animals, namely, digestive problems, haemolysis, haemoglobinuria, nephrosis, nervous signs, blindness, nitrate and isothiocyanate poisonings, goitres, photosensitivity and respiratory distress.7, 24 From the available literature it would seem that Brassica spp. do not cause a specific lung condition but that the L-tryptophan of some plants can give rise to ABPE or fog fever.3, 4, 7 Lasiospermum bipinnatum (ganskweek) is most often responsible for zonal necrosis in the liver and photosensitivity in ruminants, but respiratory distress, coupled with pulmonary oedema and emphysema, and sometimes interstitial pneumonia, have also been reported in cattle and sheep35, 63, 64 (Liver). Similar hepatic lesions and photosensitivity in ruminants, as well as lung lesions in sheep and mice, have been reported with the furanosesquiterpenoid oils of Myoporum laetum, M. tetrandrum and M. deserti growing in Australasia.45, 65 Since Athanasia minuta (= Asaemia axillaris) and Athanasia trifurcata are believed to contain similar toxins such lesions may be expected in poisonings with these plants also. These oils are closely related to the pneumotoxic stress metabolites (phytoalexins) of the sweet potato and those occurring naturally in the weed, Perilla frutescens.65 Myoporum tenuifolium (= M. acuminatum), popularly known as the manatoka, is widely planted as a shade tree in the sandy southern and western coastal areas for its drought- and saline-resistant properties. In addition to the manatoka, M. serratum, also from Australia, has become naturalized mainly in the coastal areas of the Western Cape Province (L. Henderson, National Botanical Institute, personal communication, 1999), where it poses a potential threat to stock (M. Wells, Botanical Research Institute, Pretoria, personal communication, 1986). The crofton weed, Ageratina adenophora (= Eupatorium adenophorum) in Australia and Hawaii affects the lungs of horses, causing infiltration of eosinophils in bronchioles, septae, alveolar spaces and blood vessel walls.13, 32 This plant and related species of the Eupatorium tribe (Ageratum conyzoides, A. houstonianum, Campuloclinium macrocephalum and abundant Chromolaena odorata (trifid weed or parafienbos) have become firmly established in South Africa (L. Henderson, National Botanical Institute, Pretoria, personal communication, 1999).
References
- ALEXANDER, R. 1928. Lasiosiphon anthylliodes as a poisonous plant. 13th & 14th Reports of the Director of Veterinary Education and Research, Onderstepoort, pp. 233–241.
- BRADLEY, B.J. & CARLSON, J.R. 1980. Ultrastructural pulmonary changes induced by intravenously administered 3-methylindole in goats. American Journal of Pathology, 99, 551–560.
- CARLSON, R.J. & BREEZE, R.G. 1984. Ruminal metabolism of plant toxins with emphasis on indolic compounds. Journal of Animal Science, 58, 1040–1049.
- CARLSON, J.R. & DICKINSON, E.O. 1978. Tryptophan-induced pulmonary oedema and emphysema in ruminants. pp. 261–272. In: KEELER, R.F, VAN KAMPEN, K.R. & JAMES, L.F. (eds.) Effects of poisonous plants on livestock. New York, San Francisco, London: Academic Press.
- CARSLON, J.R., YOKOYAMA, M.T. & DICKINSON, E.O. 1972. Induction of pulmonary oedema and emphysema in cattle and goats with 3-methylindole. Science, 176, 298–299.
- CARLSON, J.R., DICKINSON, E.O., YOKOYAMA, M.T. & BRADLEY, B.J. 1975. Pulmonary oedema and emphysema in cattle after intraruminal and intravenous administration of 3-methylindole. American Journal of Veterinary Research, 36, 1341–1347.
- CLARKE, MYRA L., HARVEY, D.G. & HUMPHREYS, D.J. 1981. Veterinary toxicology. London: Bailliére Tindall.
- COETZER, J. & PIETERSE, M.J. 1971. The isolation of 12-hydroxy-daphnetoxin, a degradation product of a constituent of Lasiosiphon burchellii. Journal of the South African Chemical Institute, 24, 241–243.
- COETZER, J. & PIETERSE, M.J. 1972. The structure of 12-hydroxy-daphnetoxin, a poisonous constituent of Lasiosiphon burchellii, and X-ray analysis of 12-hydroxydaphnetoxin tribromoacetate. Acta Crystallographica, 28, 620–624.
- COLVIN, B.M. & HARRISON, L.R. 1992. Fumonisin-induced pulmonary edema and hydrothorax in swine. Mycopathologia, 117, 79–82.
- DICKINSON, E.O. & CARLSON, J.R. 1978. Acute respiratory distress of rangeland cattle. pp. 251–259. In: KEELER, R.F., VAN KAMPEN, K.R. & JAMES, L.F. (eds.) The effects of poisonous plants on livestock. New York, San Francisco, London: Academic Press.
- GARDINER, M.R., ROYCE, R. & BOKOR, A. 1965. Studies on Crotolaria crispata, a newly recognised cause of Kimberly horse disease. Journal of Pathology and Bacteriology, 9, 43–55.
- GIBSON, J.A. & O’SULLIVAN, B.M. 1984. Lung lesions in horses fed mist flower (Eupatorium riparium). Australian Veterinary Journal, 61, 271.
- HAMMOND, A.C. & CARLSON, J.R. 1980. Inhibition of ruminal degradation of L-tryptophan to 3-methylindole in vitro. Journal of Animal Science, 51, 207–214.
- HAMMOND, A.C., CARSLON, R.J. & BREEZE, R.G. 1978. Monensin and the prevention of tryptophan-induced acute bovine pulmonary oedema and emphysema. Science, 201, 153–155.
- HAMMOND, A.C., CARLSON, J.R. & BREEZE, R.G. 1980. Prevention of tryptophan-induced acute bovine pulmonary oedema and emphysema (fog fever). The Veterinary Record, 107, 322–325.
- HAMMOND, A.C., CARLSON, J.R., BREEZE, R.G. & SELMAN, I.E. 1979. Progress in the prevention of acute bovine pulmonary emphysema. Bovine Practitioner, 14, 9–14.
- HARRISON, L.R., COLVIN, B.M., GREEN, J.T., NEWMAN, L.E. & COLE, J.R. 1990. Pulmonary edema and hydrothorax in swine produced by fumonisin B1, a toxic metabolite of Fusarium moniliforme. Journal of Veterinary Diagnostic Investigation, 2, 217–221.
- HENNING, M.W. 1932. Animal diseases in South Africa. Vol. II. Virus and deficiency diseases, plant poisons. South Africa: Central News Agency, Limited.
- HOOPER, P.T. 1978. Pyrrolizidine alkaloid poisoning-pathology with particular reference to differences in animal and plant species. pp. 161–176. In: KEELER, R.F., VAN KAMPEN, K.R. & JAMES, L.F. (eds.) The effects of poisonous plants on livestock. New York, San Francisco, London: Academic Press.
- HOOPER, P.T. & SCANLAN, W.A. 1977. Crotolaria retusa poisoning of pigs and poultry. Australian Veterinary Journal, 53, 109–114.
- HUANG, T.W., CARLSON, J.R., BRAY, T.M. & BRADLEY, B.J. 1977. 3-Methylindole-induced pulmonary injury in goats. American Journal of Pathology, 87, 647–666.
- JUBB, K.V.F., KENNEDY, P.C. & PALMER, N. 1985. The respiratory system. In: Pathology of domestic animals. 3rd edn. Vol. 2. Orlando, San Diego, New York, London, Toronto, Montreal, Sydney, Tokyo: Academic Press.
- KINGSBURY, J.M. 1964. Poisonous plants of the United States and Canada. Englewood Cliff, New Jersey: Prentice-Hall, Inc.
- KRIEK, N.P.J., KELLERMAN, T.S. & MARASAS, W.F.O. 1981. A comparative study of the toxicity of Fusarium verticillioides (= F. moniliforme) to horses, primates, pigs, sheep and rats. Onderstepoort Journal of Veterinary Research, 48, 129–131.
- KUPCHAN, S.M., SHIZURI, Y., BAXTER, R.L. & HAYNES, H.R. 1977. Gnididione, a new furanosesquiterpene from Gnidia latifolia. Journal of Organic Chemistry, 42, 348–350.
- LAST, R. D., VORSTER, J. H., BURGOYNE, P., PETERSEN, R. & THOMPSON, M. 2000. Two outbreaks of Crotalaria poisoning ‘jaagsiekte’ in horses in KwaZulu-Natal. Proceedings of the Biennial Congress of the South African Veterinary Association, Durban, South Africa, 20–22 2000: 89–91.
- LAWS, L. 1968. Toxicity of Crotalaria mucronata to sheep. Australian Veterinary Journal, 44, 453–455.
- LEKEUX, P., HAJER, R., VAN DEN INGH, T.S.G.A.M. & BREUKINK, H.J. 1985. Pathophysiologic study of 3-methylindole-induced pulmonary toxicosis in immature cattle. American Journal of Veterinary Research, 46, 1629-1631.
- MARAIS, J.S.C. 1944. Dicrotaline: The toxic alkaloid from Crotalaria dura (Wood & Evans) and Crotalaria globifera (E. Mey). Onderstepoort Journal of Veterinary Science and Animal Industry,20, 61–65.
- McLEAN, ELIZABETH K. 1970. The toxic actions of pyrrolizidine (Senecio) alkaloids. Pharmacological Reviews, 22, 429–483.
- O’SULLIVAN, B.M. 1979. Crofton weed (Eupatorium adenophorum) toxicity in horses. Australian Veterinary Journal, 55, 19–21.
- OSWEILER, G.P., ROSS, P.F., WILSON, T.M., NELSON, P.E., WITTE, S.T., CARSON, T.L., RICE, L.G. & NELSON, H.A. 1992. Characterization of an epizootic pulmonary edema in swine associated with fumonisin in corn screenings. Journal of Veterinary Diagnostic Investigation, 4, 53, 53–59.
- PECKHAM, J.C., SANGSTER, L.T. & JONES, O.H. 1974. Crotalaria spectabilis poisoning in swine. Journal of the American Veterinary Medical Association, 165, 633–638.
- PENRITH, M-L. & VAN VOLLENHOVEN, E. 1994. Pulmonary and hepatic lesions associated with suspected ganskweek (Lasiospermum bipinnatum) poisoning in cattle. Journal of the South African Veterinary Association, 65, 122–124.
- PHILLIPS, E.P. 1926. A preliminary list of the known poisonous plants found in South Africa. Botanical Survey of South Africa, Memoir No. 9. The Government Printing and Stationery Office, Pretoria.
- PIETERSE, M.J. 1971. On the isolation of toxic constituents from Lasiosiphon burchellii Meisn. Agrochemophysica, 3, 63–66.
- PIRIE, H.M., BREEZE, R.G., SELMAN, J.E. & WISEMAN, A. 1974. Fog fever in cattle: Pathology. The Veterinary Record, 95, 479–483.
- PROZESKY, L., KELLERMAN, T.S., JORDAAN, P. & WELMAN, WILHELMINA G. 1986. An ovine hepatotoxicosis caused by the plant Hertia pallens (Asteraceae) Kuntze. Onderstepoort Journal of Veterinary Research, 52, 233–238.
- ROSS, P.F., NELSON, P.E., RICHARD, J.L., OSWEILER, G.D., RICE, L.G., PLATNER, R.D. & WILSON, T.M. 1990. Production of fumonisins by Fusarium moniliforme and Fusarium proliferatum isolates associated with eqiune leukoencephalomalacia and pulmonary edema syndrome in swine. Applied Environmental Microbiology, 56, 3225–3226.
- ROSS, P.F., RICE, L.G., PLATTNER, R.D., OSWEILER, G.D., WILSON, T.M., OWENS, D.L., NELSON, H.A. & RICHARD. J.L. 1991a. Concentrations of fumonisin B1 in feed associated with animal health problems. Mycopathologia, 114, 129–135.
- ROSS, P.F., RICE, L.G., REAGOR, C., OSWEILER, G.D., WILSON, T.M., NELSON, H.A., OWENS, D.L. PLATTNER, R.D., HARLIN, K.A., RICHARD, J.L., COLVIN, B.M. & BANTON, M.I. 1991b. Fumonisin B1 concentrations in feeds from 45 confirmed cases of equine leukoencephalomalacia. Journal of Veterinary Diagnostic Investigation, 3, 238–241.
- SANDERS, D.A., SHEALY, A.L. & EMMEL, M.W. 1936. The pathology of Crotalaria spectabilis froth poisoning in cattle. Journal of the American Veterinary Medical Association, 89, 150–156.
- SCHOENTAL, R. 1968. The toxicology and carcinogenic action of pyrrolizidine alkaloids. Cancer Research, 28, 2237–2246.
- SEAWRIGHT, A.A., LEE, J.S., ALLEN, J.G. & HRDLICKA, J. 1978. Toxicity of Myoporum spp. and their furanosesquiterpenoid essential oils. In: KEELER, R.F., VAN KAMPEN, K.R. & JAMES, L.F. (eds.) The effects of poisonous plants in livestock. New York, San Francisco, London: Academic Press.
- SELMAN, J.E., WISEMAN, A., PIRIE, H.M. & BREEZE, R.G. 1974. Fog fever in cattle: Clinical and epidemiological features. The Veterinary Record, 95, 139–146.
- STEYN, D.G. 1931. Recent investigations into the toxicity of known and unknown poisonous plants in the Union of South Africa. 17th Report of the Director of Veterinary Services and Animal Industry, pp. 707–727.
- STEYN, D.G. 1932. Investigations into the toxicity of known and unknown poisonous plants in the Union of South Africa. 18th Report of the Director of Veterinary Services and Animal Industry of the Union of South Africa, pp. 871–891.
- STEYN, D.G. 1934. The toxicology of plants in South Africa. Central News Agency, Limited.
- STEYN, D.G. 1937. Recent investigations into the toxicity of known and unknown poisonous plants in the Union of South Africa. VIII. Onderstepoort Journal of Veterinary Science and Animal Industry, 9, 119–120.
- STEYN, D.G. 1949. Vergiftiging van mens en dier. Pretoria: Van Schaik.
- STEYN, D.G. & DE KOCK, G. 1932. Crotalariasis in sheep. 18th Report of the Director of Veterinary Services and Animal Industry, Union of South Africa, pp. 947–953.
- STEYN, D.G. & VAN DER WALT, S.J. 1945. Jaagsiekte by perde en Sunn-hennop-vergiftiging by vee. Boerdery in Suid-Afrika, 20, 445–447.
- TERBLANCHE, M., PIETERSE, M.J., ADELAAR, T.F. & SMIT, J.D. 1966. Further studies on the toxicology of Lasiosiphon burchellii Meisn (Harpuisbos). Journal of the South African Veterinary Medical Association, 37, 185–189.
- THEILER, A. 1918. Jaagsiekte in horses. 7th and 8th Reports of the South African Department of Agriculture and Veterinary Services, pp. 5–108.
- THEODORIDIS, A. & COETZER, J.A.W. 1979. Subcutaneous and pulmonary emphysema as complications of bovine ephemeral fever. Onderstepoort Journal of Veterinary Research, 46, 125–127.
- TUSTIN, R.C. 1969. Ovine jaagsiekte. Journal of the South African Veterinary Medical Association, 40, 3–23.
- VAHRMEIJER, J. 1981. Poisonous plants of Southern Africa that cause stock losses. Cape Town: Tafelberg.
- VAN DER WALT, S.J. & STEYN, D.G. 1939. Recent investigations into the toxicity of known and unknown poisonous plants in the Union of South Africa. IX. Onderstepoort Journal of Veterinary Science and Animal Industry, 12, 335–366.
- VERWOERD, D.W. 1985. Biotechnology, viral oncogenesis and jaagsiekte. Onderstepoort Journal of Veterinary Research, 52, 145–148.
- VERWOERD, D.W., WILLIAMSON, A. & DE VILLIERS, E-M. 1980. Aetiology of jaagsiekte: Transmission by means of subcellular fractions and evidence for the involvement of a retrovirus. Onderstepoort Journal of Veterinary Research, 47, 275–280.
- VERWOERD, D.W., PAYNE, ANNALISE, YORK, D.F. & MYER, M.S. 1983. Isolation and preliminary characterization of the jaagsiekte retrovirus (JSRV). Onderstepoort Journal of Veterinary Research, 50, 309–316.
- WILLIAMS, M.C. 1985. Pathology of Lasiospermum bipinnatum (Thunb.) Druce poisoning in sheep. Dissertation: Master of Veterinary Medicine (Pathology), University of Pretoria.
- WILLIAMS, M.C. 1990. The pathology of experimental Lasiospermum bipinnatum (Thunb.) Druce (Asteraceae) poisoning in sheep. II. Pulmonary and miscellaneous lesions. Onderstepoort Journal of Veterinary Research, 57, 263–268.
- WILSON, B.J., GARST, J.E., LINNABARY, R.D. & DOSTER, A.R. 1978. Pulmonary toxicity of naturally occurring 3-substituted furans. In: KEELER, R.F., VAN KAMPEN, K.R. & JAMES, L.F. (eds.) The effects of poisonous plants on livestock. New York, San Francisco, London: Academic Press.
- WILSON, T.M., ROSS, P.F., RICE, L.G., OSWEILER, G.D., NELSON, H.A., OWENS, P.L., PLATTNER, R.D., REGGIARDO, C., NOON, T.H. & PICKERELL, S.W. 1990a. Fumonisin B1 levels associated with an epizootic of equine leukoencephalomalacia. Journal of Veterinary Diagnostic Investigation, 2, 213–216.
- WILSON, T.M., NELSON, P.E., MARASAS, W.F.O., THIEL, P.G., SHEPHARD, G.S., SYDENHAM, E.W., NELSON, H.A., ROSS, P.F. 1990b. A mycological evaluation and in vivo toxicity evaluation of feed from 41 farms with equine leukoencephalomalacia. Journal of Veterinary Diagnostic Investigation, 2, 352–354.