Haemopoietic system
| Prussic acid poisoning | Sorghum spp. | Cynodon spp. | Dimorphotheca and Osteospermum spp. | Eucalyptus cladocalyx | Acacia spp. | Manihot esculenta | Jatropha multifida |
This content is distributed under the following licence: Attribution-NonCommercial CC BY-NC  View Creative Commons Licence details here
View Creative Commons Licence details here

Introduction
Plant poisonings and mycotoxicoses in stock affecting the haemopoietic system are not of great economic importance in southern Africa, apart possibly from nitrate and prussic acid toxicities which at times can inflict severe losses in cattle and sheep. Nevertheless, it is often necessary to consider some of these toxicoses as differential diagnoses for important tick-borne and infectious diseases which threaten livestock in our region.
The poisonings under discussion can be grouped roughly under plants and fungi which cause either haemolysis and/or biochemical alterations of mature red blood cells, or those which are responsible for bone-marrow and lymphoid tissue suppression. In animals suffering from intoxication by the former group of plants and fungi, tissue respiration is mostly affected, and kidney and liver function are often disturbed, while in the latter group, affected animals are frequently subjected to impaired immunological response, bleeding tendencies and sometimes also anaemia.
Enzootic icterus
Geelsiekte
Enzootic icterus, also referred to locally as geelsiekte, is a chronic copper poisoning of sheep and goats, following prolonged ingestion of relatively high levels of phytogenous copper in large parts of the semi-arid Karoo and the southern Free State Province.3, 28, 29, 77, 78
The disease was first noticed in 1924, when sheep transported from the Karoo to the Veterinary Research Institute, Onderstepoort, died acutely 10–30 days after arrival, showing severe icterus and anaemia.29 Since then enzootic icterus has been noticed regularly and many theories have been put forward regarding its aetiology. Brown6, 8, 9 suggested that enzootic icterus and geeldikkop were different manifestations of the same disease entity. Both conditions basically arose from aberrations in the selective permeability of cell membranes and the active transport mechanisms across cell membranes.9 Key dehydrogenases were inactivated with catastrophic effects on metabolism.6, 7
As the liver copper content of sheep suffering from enzootic icterus was conspicuously elevated,9, 35 copper was investigated as a possible aetiological factor in the genesis of the disease. Neethling et al.,87 however, concluded that it was unlikely, under conditions prevailing in the Karoo (including those parts where the plants contain up to 28 ppm of copper or more) that chronic copper poisoning would occur if the molybdenum status of the animal was adequate. As a result of the high copper values and the similarities of the necropsy findings of enzootic icterus to those of chronic copper poisoning, Erasmus35 was tempted to equate enzootic icterus with chronic copper poisoning and to consider them as basically the same condition. Another factor, apart from copper and molybdenum, that affects the metabolism of sheep in the Karoo, is selenium. Brown and de Wet10 demonstrated the presence of potentially dangerous amounts of selenium in various plants from geeldikkop and enzootic icterus areas. Sheep emanating from such areas had higher levels of selenium in the body tissues than those of control sheep.9 A theory was put forward that selenium may predispose sheep to geeldikkop and enzootic icterus by disrupting enzyme systems, especially those connected with transfer mechanisms and the glycolytic cycle.6, 76 It was also suggested that geeldikkop and enzootic icterus were autoimmune diseases precipitated by aberrant selenoprotein antigens.86, 87 Recently, it was shown that geeldikkop and enzootic icterus are two distinct entities with different aetiologies. Geeldikkop is a complex disease caused by Tribulus terrestris plants,72 and enzootic icterus is a chronic copper poisoning.3
Bath3 discussed at length many of the epizootiological factors playing a role in enzootic icterus. He, as well as other authorities,28, 77, 78 reported relatively high copper levels in certain plants on farms where enzootic icterus occurs regularly. Bath3 reviewed the literature on enzootic icterus and chronic copper poisoning and came to the conclusion that the type and pH of the soil may influence copper uptake by plants. For example, low pH levels inhibit both phytogenous selenium and molybdenum, and it is well known that the former enhances copper accumulation in the liver and the latter depresses it.98, 99
According to Bath,3 enzootic icterus would appear to occur more frequently on farms with doleritic ridges, and he concluded that the high copper and low pH levels in these ridges may be important contributory factors in the epizootiology of the disease. He, as well as Pope,99 pointed out that other elements, such as sulphur, zinc, manganese, iron, calcium, phosphorus and cobalt can be involved in this complex imbalance.99
Copper analyses, done on the livers of 36 adult Merino sheep originating from Middelburg in the Karoo and used in geeldikkop and facial eczema experiments, showed marked elevation, some of which had values of up to 544 ppm on a wet basis.72 Apart from slight khaki-brown discolouration of the liver and kidneys, no clinical signs or other macroscopical changes compatible with fatal enzootic icterus were seen in these animals. Microscopically, the hepatic changes of these subclinical cases were characterized by infiltration of mild to moderate numbers of macrophages laden with a homogenous khaki-brown lipofuscinous pigment in the portal triads; slight bile ductular proliferation and portal fibroplasia; anisonucleosis and karyomegaly of scattered hepatocytes and pigmentation of some liver and Kupffer cells.72 Bath3 likewise reported that many of the healthy, aged sheep at the Grootfontein Agricultural College at Middelburg have liver copper levels of above 300 ppm on a wet basis. High liver copper values for Karoo sheep are also given by Brown9 and Erasmus.35 It is thus clear that a diagnosis of enzootic icterus cannot be made solely on high liver copper values, but that the clinical signs and especially changes in the carcase compatible with enzootic icterus must be taken into consideration (J.A.W. Coetzer, VRI, Onderstepoort, unpublished observations, 1986).
Of all livestock, sheep show the greatest tendency to accumulate copper in the liver. Hepatocytes in sheep have an avidity for copper, which is excreted at a limited rate in their bile.70 Copper is a strong oxidizing agent, but relatively high levels of copper can be stored in lysosomes with little effect on the integrity of the cell. However, if the concentration rises to very high levels, the lysosomal membranes are damaged, releasing copper and hydrolases which have a noxious effect on the cytoplasm. This leads to necrosis of scattered hepatocytes throughout the liver.70 Copper reaches the circulation, and eventually the concentration in the blood is high enough to cause intravascular haemolysis. The haemolysis in turn results in anoxia and necrosis especially of the centrilobular hepatocytes with the release of more copper in the circulation. When the excess copper exceeds the capacity of the carrier proteins in the plasma and presumably exhausts the antioxidant glutathione system of the red blood cells, it gives rise to denaturation of the haemoglobin and the formation of Heinz-bodies, while the damage to the cell membranes leads to haemolysis.70 In chronic copper poisoning, the ceruloplasmin is reported to rise two to three times the normal level during haemolytic crises; the red cell copper increases 15–20 times; plasma copper increases three to six times; and whole blood copper increases up to ten times the normal level.70
Marked elevations in plasma and red blood cell copper levels have been reported in sheep suffering from enzootic icterus.3 The liver copper value of enzootic icterus cases is always significantly elevated.3, 9, 34, 35 Bath3 found that the mean copper values for the liver and kidneys of fatal cases were 701 μg/g and 107 μg/g, respectively, while the kidney iron level was 534 μg/g. All measurements were done on a wet basis. High liver copper values have been reported for healthy Karoo sheep3, 8, 9, 35 and are thus not diagnostic for chronic copper poisoning, but are indicative of potential toxicity, if the copper is released into the bloodstream. It would seem that determination of the kidney copper level is more useful in making a diagnosis of poisoning and levels of above 20 μg/g on a wet mass should be considered toxic (normal kidney copper level is seldom above 10 μg/g on a wet mass).3, 83, 89, 99, 104 Pope99 reviewed the literature on chronic copper poisoning in sheep and came to the conclusion that most fatal cases will have copper values higher than 500 ppm and 80 ppm in the liver and kidneys on a dry matter basis, respectively.
Enzootic icterus is a stress-related disease brought about by a variety of factors, for instance, transportation or trekking over several days; poor nutrition, such as is often the case in winter months or during droughts; and other factors which include changes in the weather, dipping, dosing, inoculation, shearing, lambing, pregnancy, etc. Sometimes intercurrent diseases, especially those that affect the liver, such as geeldikkop, Rift Valley fever and Wesselsbron disease, may trigger enzootic icterus and confuse the clinical and pathological picture.3, 28, 75, 110
Enzootic icterus occurs in all breeds of sheep, but Dorpers seem more prone to develop the disease than Merinos. Goats are occasionally affected, and no cases have yet been reported in cattle.3 Most animals suffering from enzootic icterus are old, worn-mouth animals, while the disease is rarely seen in sheep less than six-tooth.3, 110
In cases of enzootic icterus, there is a preclinical or incipient stage during which copper accumulates in the liver and the animal suffers mild haemolytic attacks that are generally not noticed by the farmer.3 This stage is usually soon followed by a severe haemolytic episode that may be fatal within 12 hours, or the animal can survive for up to four days, during which more crises may occur. The duration of illness would seem to be primarily influenced by the severity and rapidity of haemolysis.3
Apart from subclinical or unapparent cases in a flock, the morbidity usually ranges between 0,5 and 5% but can be as high as 10 or even 20%.3 Skinner110 claimed that between 1957 and 1958 approximately 15 000 sheep died of enzootic icterus in the Karoo. Almost all animals that are clinically affected may die. Sick sheep are often in a poor condition, anorectic, apathetic and lag behind the flock when driven. They also show signs of anaemia, dirty brownish and icteric mucous membranes and reddish-brown to almost black discolouration of the urine. There is ruminal atony and the faeces is hard and dry, dark brown and covered by mucus that is sometimes tinged with blood.3, 28, 29 However, often the disease is precipitously expressed and animals are found dead without revealing any premonitory signs.
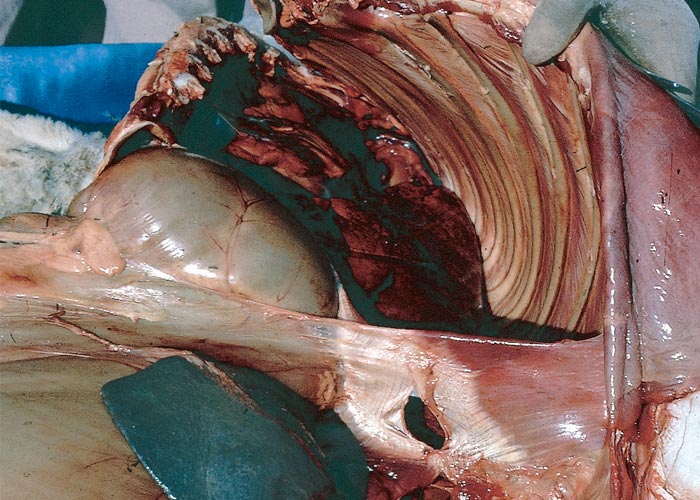
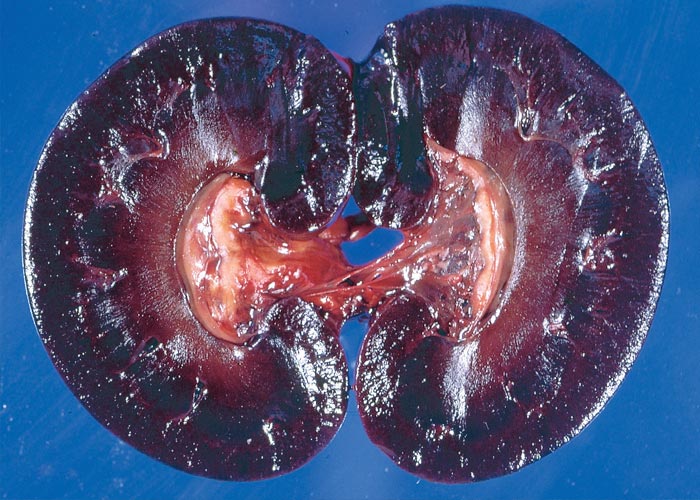
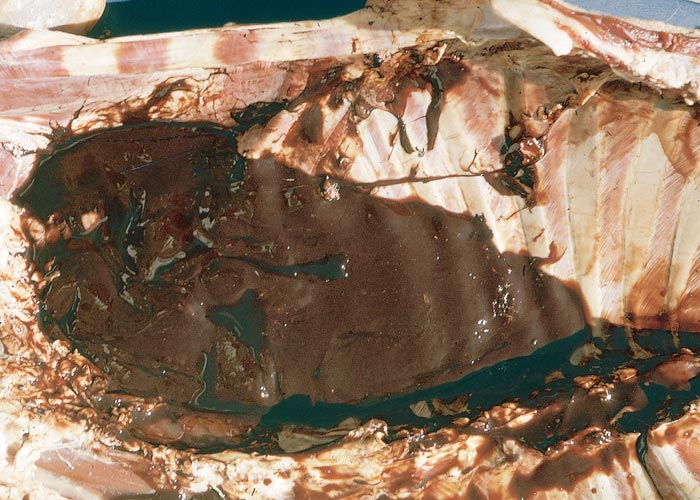
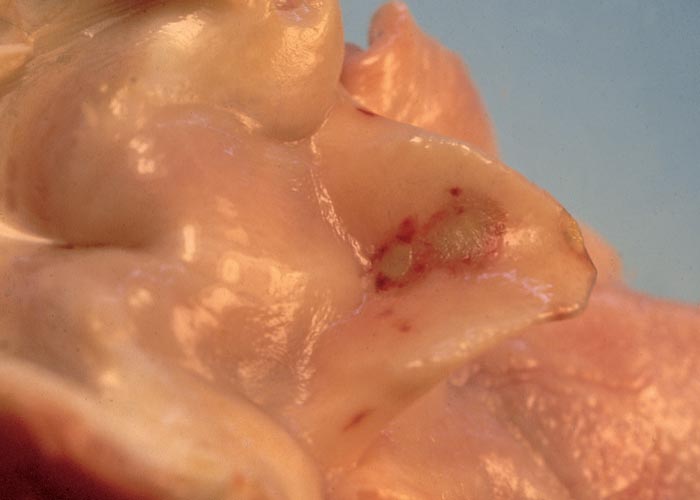
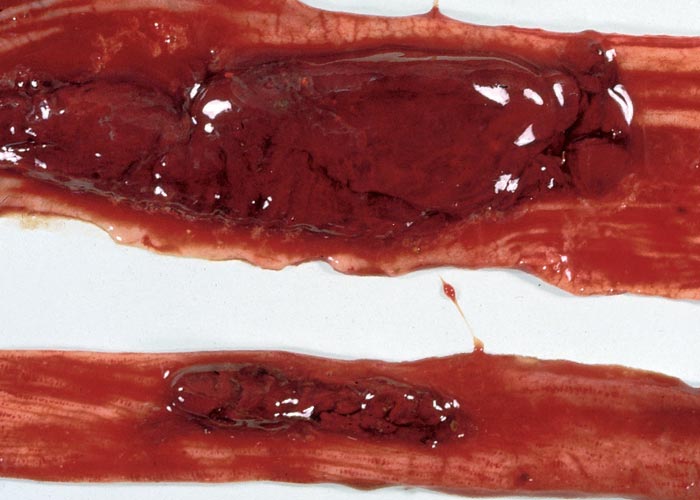
At necropsy, the most noteworthy changes include a dirty brown jaundice (Figure 1) which becomes more visible some time after the carcase has been opened; methaemoglobinaemia, haemoglobinaemia, a watery blood, as well as other signs of anaemia; severely swollen, gun metal to almost black pigmentation of the kidneys (Figure 2); stasis and atrophy of the gastrointestinal tract (especially of the rumen and small intestines) with the accumulation of mucus-covered, dry faecal balls in the caecum and colon; enlargement of the spleen, and on cut section the red pulp appears dark, reddish-brown and mushy; yellowish-brown friable to somewhat enlarged liver, revealing distinct lobulation; dark reddish-brown urine as a result of haemoglobinuria and bilirubinuria; atrophy of the lymphoid tissues and enlargement of the adrenal glands; and mild effusion of the body cavities. The carcases decompose rapidly within hours of death3, 28, 29 (J.A.W. Coetzer, VRI, Onderstepoort, unpublished observations, 1985).
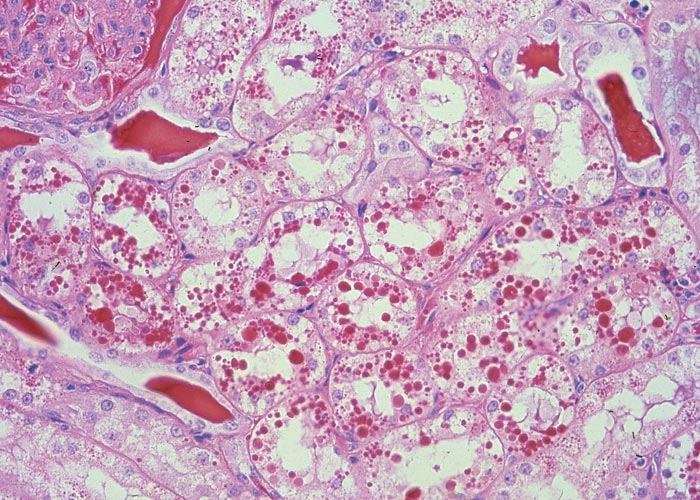
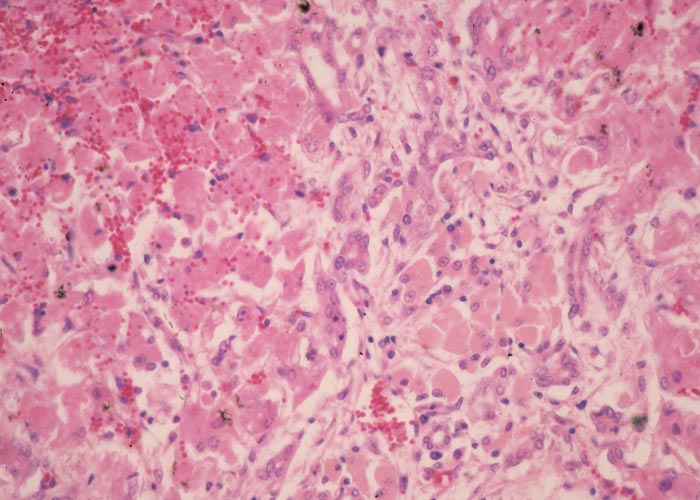
Microscopically, there is evidence of a haemoglobinuric/ischaemic nephrosis characterized by severe tubular degeneration and necrosis; the accumulation of haemoglobin and protein droplets of varying size (seen as hyaline droplets), bile pigments, lipofuscin and haemosiderin especially in the proximal convoluted tubular epithelium (Figure 3). Numerous haemoglobin and proteinaceous casts in tubuli occur in the cortex and medulla. The hepatic lesions are typified by severe bile stasis; portal reactions, comprising infiltration of many characteristic khaki-brown lipofuscinous-laden macrophages (Figure 4), mild to moderate bile ductular proliferation, fibroplasia and mononuclear cell infiltration; moderate Kupffer cell proliferation and pigmentation (lipofuscin and haemosiderin); diffuse hepatocellular degeneration (cloudy swelling, hydropic degeneration and fatty metamorphosis); acidophilic degeneration and necrosis of individual hepatocytes, and in some instances, centrilobular coagulative necrosis; and anisonucleosis and karyomegaly of hepatocytes, some of which contain intranuclear inclusion (parts of invaginated cytoplasm)29, 96 (J.A.W. Coetzer, VRI, Onderstepoort, unpublished observations, 1986). A spongiform leucoencephalopathy, characterized ultrastructurally by the formation of intramyelinic vacuoles with splitting of myelin lamellae at the intraperiod line may also be evident in sheep suffering from chronic copper poisoning.83
The prognosis of sheep suffering from clinical enzootic icterus is poor and notwithstanding vigorous treatment most animals will die.3 Treatment of these cases is thus of little avail and instigation of preventive measures is therefore preferable. Bath3 obtained encouraging results by the prophylactic treatment of sheep with ammonium or sodium molybdate on farms where enzootic icterus is a problem. He stressed that the goal in prevention of the disease is to reduce copper uptake and storage in the liver over a long period by regular dosing or the supplementation of molybdate in licks and drinking water. Farmers in this area are advised to dose 5–10 ml of a 2% solution of ammonium or sodium molybdate to their sheep every second day or weekly (this supplies 100–200 mg/dose/day). According to Bath,3 in most instances further deaths and clinical cases were prevented after this procedure. The addition of ZnSO4 at a level of 250–300 mg/sheep/day to the drinking water for at least six weeks also gave excellent results (G.F. Bath, Allerton Veterinary Laboratory, personal communication, 1986). Sheep have a high tolerance to zinc and a diet of 100 ppm on a dry matter basis reduces liver copper storage and can directly reverse the lesions induced by copper.99 Pope99 summarized different methods, subsequently implemented in various countries, of the treatment and prevention of copper toxicoses in sheep. He concluded that treatment must be carried out daily for three weeks or longer, and both molybdate and sulphate should be used to form thiomolybdates. He also gave different formulae that can either be drenched, supplemented in licks, incorporated in pelleted feed or sprayed on hay. Gooneratne et al.52 found that the intravenous injection of 100 mg of ammonium tetrathiomolybdate twice a week prevented the occurrence of a haemolytic crisis in sheep repeatedly dosed with CuSO4. It also minimized tissue damage and prevented further crises when given to sheep already suffering from haemolysis. They also showed that thiomolybdate prevented excessive accumulation of Cu in the liver of sheep on a diet high in Cu and reduced liver Cu levels in animals that were not given additional Cu.
Mobilization and excretion of copper stored in the liver can be accelerated by the use of chelating drugs, such as D-penicillamine at high doses of 52 mg/kg daily in divided doses.5, 112 This is, however, expensive and it can consequently only be used in very valuable stock.
Although it is beneficial to eliminate various stress factors, it is often difficult to carry out in practice. Sound veld management is also of value in the prevention of enzootic icterus.3, 110
Allium cepa L. (Alliaceae)
Onion, ui
A. sativum L. var. sativum
Garlic, knoffel
A. sativum var. othioscorodon
Spanish garlic
Restricted quantities of culled onions are regularly fed, especially in Europe, to cattle and sheep without any toxic effect. However, high intakes of Allium cepa by stock give rise to severe haemolytic anaemia, haemoglobinuria, haemoglobinuric/ischaemic nephrosis and icterus.66, 73, 74, 122 In our part of the world, culled onions are rarely used as feed and, to the best of our knowledge, outbreaks of poisoning with this commodity have only occurred in the Caledon district of the Cape Province (R.A. Wilson, private veterinarian, Caledon, personal communication, 1986).
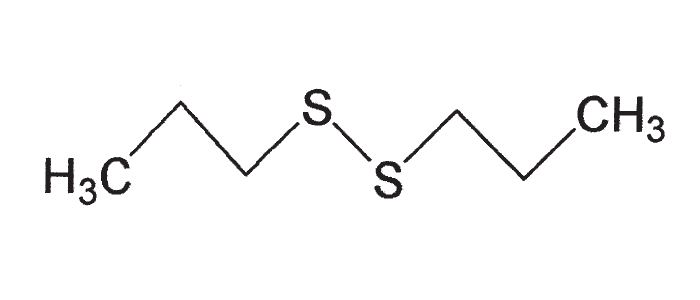
The toxic principle of A. cepa is n-propyl disulphide (Figure 5), which is a non-nitrogenous, pungent, volatile oil. The enzyme glucose-6-phosphate dehydrogenase (G-6-PD) within the red blood cells is affected by the toxin resulting in interference with the hexose monophosphate pathway.111, 122 In normal circumstances, adequate levels of reduced glutathione (which is a sulphydryl-containing tripeptide) within red blood cells are necessary to maintain haemoglobin in its reduced state and thus protect erythrocytes against oxidative injury.54 With low glutathione, or with impaired G-6-PD, the reducing power in red blood cells is deficient, and oxidative changes in the haemoglobin and damage to the cell membranes result. The denaturated haemoglobin precipitates in the red blood cells to form Heinz-bodies. The affected cells may be taken up by the reticulo-endothelial system or haemolysis may take place as a result of damage to the cell membranes.111, 122 Heinz-bodies often project from the outer edge of red blood cells or may be released into the plasma. These spherical, refractile bodies do not stain with Wright’s stain, but with methylene blue they take on a dark blue colour against red blood cells which are unstained.70, 111
The susceptibility of stock to onion and Brassica poisonings (vide infra) is not only dependent on the G-6-PD levels in red blood cells, but is also influenced by the amount of glutathione reductase in these cells.111 For example, it has been demonstrated that red blood cells of cattle are generally very low in glutathione reductase, but if animals are selected with higher levels of this enzyme they will be more resistant to Brassica poisoning.111
Verhoeff et al.122 described an outbreak of poisoning in the Netherlands, where cattle 6–22 months old each consumed c.8–15 kg of onions per day for five days before they became sick. New cases continued to occur for five days after withdrawal of the onions. Blood smears of affected animals revealed hypochromasia, anisochromasia, polychromasia and basophilic stippling of red blood cells, and some corpuscles contained Heinz-bodies. At necropsy, many pieces of onions were found in the rumen, all the tissues smelled of onions and there were changes compatible with a haemolytic anaemia and icterus. The meat and milk of cattle on a culled onion diet may be tainted.21, 73, 74, 124
Apart from poisoning cattle, sheep and horses, onions have also been reported as the cause of poisoning of cats and dogs,54 in the last instance after cooking.
Recently an outbreak of typical Heinz-body haemolytic anaemia in which 13 of 243 cattle died one week after being fed excessive quantities of unmarketable garlic, occurred in the Limpopo Province of South Africa (J.C. Stroebel, Veterinary Investigation Centre, Potgietersrust, personal communication, 1999). Clarke et al. (1981)21 also refer to n-propyl-disulphide as the active principle in garlic. The possibility of this intoxication should be borne in mind when stock are exposed to any member of the Alliaceae.
Brassica spp. (Brassicaceae) (= Cruciferae)
Kale, rape and other Brassica spp. are known to cause haemolysis in animals in the Northern Hemisphere, when cruciferous plants are fed almost exclusively to stock.1, 16, 73, 111 However, an outbreak of cabbage poisoning has now been described in South Africa, where poor production and reproductive performance, as well as anaemia, were observed in cattle and sheep fed unmarketable cabbage for 5–6 months per year, over a number of years.116
The majority of toxic cruciferous plants are cultivated. These species include plants in which the tubers are usually utilized, e.g. B. rapus (turnip), B. napus (rape) and B. sativus cult. longipinnatus (Japanese radish), as well as species cultivated for their leaves, e.g. B. oleracea, varieties of which include the well-known cabbage, kale (beeskool), Brussels sprouts and cauliflower.
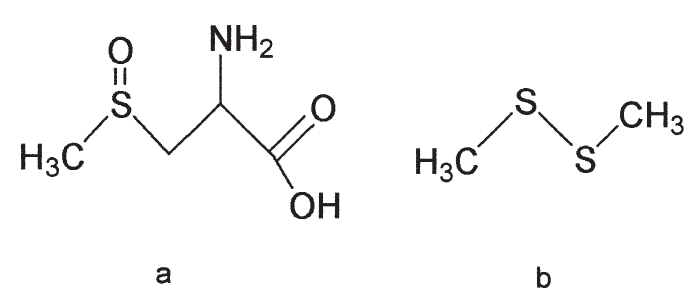
Various disease syndromes have been associated with Brassica spp. Some species contain a relatively non-toxic substance, S-methylcysteine sulphoxide [SMCO] (Figure 6), which is converted by ruminal micro-organisms to a haemolytic factor, dimethyl disulphide.1, 16, 111 This factor is similar to the toxic principle, n-propyl disulphide, in Allium cepa.122 The haemolytic response to SMCO intake by the animal could be influenced by other dietary components having an effect on the production and absorption of dimethyl disulphide.111 The SMCO content of Brassica spp. rises as the plant matures, becomes damaged by frost, or after the application of nitrogenous fertilizer.111
The pathogenesis of the anaemia and the formation of Heinz-bodies in some Brassica toxicities are discussed under onion poisoning. Heinz-bodies are more numerous, and haemolytic anaemia appears to be more severe in cattle and goats than in sheep after feeding kale.16 Affected animals are initially anaemic, but later also become icteric and show haemoglobinuria and bilirubinuria.73, 111 Apart from degenerative changes in the liver and kidneys there are also haemosiderin deposits in the liver, kidneys and spleen.111 Large quantities (40–50 kg per animal per day) of B. oleracea (Marrowstem kale or beeskool), fed fresh or ensiled to cattle for prolonged periods, have been identified as the cause of anaemia and haemoglobinuria.21 Depending on the Brassica spp. involved, the result may be acute bovine pulmonary oedema and emphysema (fog fever) (see Respiratory system); gastrointestinal irritation or constipation; a nervous syndrome and blindness. Glucosinolate-containing seeds of Brassica spp. are goitrogenic and may cause resorption, abortion or the birth of weak offspring, showing signs of cretinism. Cruciferous plants tend to taint milk and should be fed after milking.21, 73 Poor growth and conception rates in stock may be part of Brassica poisoning and have been attributed to the antithyroid factors in the plant, acting most probably synergistically with the anaemic factor.111 In the South African outbreak, abortions, increased inter-calving periods and repeated inseminations were recorded in the cows and swayback and goitre in lambs.116 Brassica leaves can contain as much as 2% nitrates on a dry basis, which, if ingested, is sufficient to cause methaemoglobinaemia.111 The tubers are similarly toxic.
According to Smith,111 Brassica poisoning may be prevented by cultivating new varieties low in SMCO.
Prussic acid poisoning
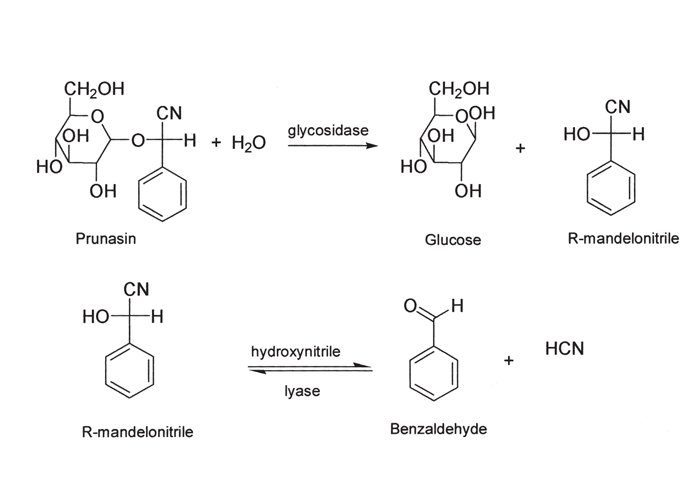
Before enterotoxaemia (bloednier) was recognized as a specific disease entity, many deaths caused by it in sheep were wrongly ascribed to prussic acid (HCN) poisoning (geilsiekte). Nevertheless, geilsiekte still ranks as an important plant poisoning of livestock in southern Africa. According to Conn24 approximately 1000 species of plants worldwide, representing some 250 genera and 80 families, are known to be cyanophoric. Only those plants of significance in the sub-Saharan region will be mentioned here. In plants, prussic acid is present as cyanogenic glycosides of α-hydroxy nitriles or cyanohydrins. These glycosides in the plant cells are compartmentalized separately from the hydrolysing enzymes, ß-glycosidases and hydroxynitrile lyases. Damage to the plant cell by frost, hail, wilting, trampling, the application of herbicides like 2,4-D or digestion by ruminal micro-organisms, causes the enzymes to react with the glycosides, thereby liberating HCN and increasing the toxicity of the plant (Figure 7). Application of nitrogenous fertilizers and stunting of growth may increase the HCN-content; young growth is generally most dangerous; drying decreases the toxicity of plants and ensiling results in complete detoxification.
Ruminants are more prone to intoxication than non-ruminants because liberation of prussic acid in the forestomachs is not stopped by low stomach pH as in monogastric animals. Nevertheless, intoxication of pigs by peach stones has been experienced in South Africa (D.J. Schneider, Regional Veterinary Laboratory, Stellenbosch, personal communication, 1984).
A number of cyanogenic plants are of veterinary importance in southern Africa. These include the following:
Sorghum spp. (Poaceae)
S. bicolor (L.) Moench. subsp. arundinaceum (Desv.) De Wet and Harlan
Grain sorghum, graansorghum
S. bicolor (L.) Moench. subsp. drummondii (Steud.) De Wet
(= S. sudanense (Piper) Stapf)
Sudan grass
S. halepense (L.) Pers.
(= S. almum Parodi)
Columbus grass
Both grain sorghum (Figure 8) and the various feed sorghums, e.g. Columbus grass and Sudan grass, are particularly dangerous when young sprouts under 500 mm in length are ingested. The cyanogenic glycoside in this genus is dhurrin. Certain feed sorghum hybrids have been selected for their low prussic acid potential.
Cynodon spp. (Poaceae)
C. dactylon (L.) Pers.
Quick grass, kweek
C. nlemfuensis Vanderyst var. robustus
Robust or Rhodesian star grass
C. aethiopicus Clayton and Harlan
Nakuru grass
Kweek is an indigenous toxic grass and several cultivated varieties of this species and of other star grasses of the same genus, e.g. robust or Rhodesian star grass and Nakuru grass, are potentially toxic.
Dimorphotheca and Osteospermum spp. (Asteraceae)
This group of plants belonging to the daisy family includes bietou or madeliefies which occur widely in southern Africa as annuals or sometimes as ephemeral growth (opslag) after rain or as perennials. Bietous may cause severe losses in sheep in certain years. Many of these daisies are cultivated as ornamental plants.
Dimorphotheca cuneata (Thunb.) Less. (the Karoo or large bietou, witgousblom) is a typical example of this group. It is a spreading, many-branched, soft woody shrublet growing up to 0,7 m high. The leaves are alternate and are c.20 mm in length and 5–10 mm wide, toothed, sometimes deeply incised, and of variable shape; the young leaves are sticky and strongly aromatic. Single flowerheads are borne at the ends of branches (Figure 9). The petals of the outer ray florets are white above and pale copper coloured or purplish beneath. The disc florets are yellow. This species has flat round and winged seeds, but also bears seeds which are oblong with wart-like tubercles on the sides as in Osteospermum spp.121
Dimorphotheca cuneata is the most widely distributed species of this genus and although it is found in a variety of soil types and habitats it most often grows on hill slopes and invades over-grazed areas. Its main flowering time is early spring, but depending on local conditions, flowering plants can be found from August to February.121
Eucalyptus cladocalyx F. Muell. (Myrtaceae)
Of the many Eucalyptus tree spp. (blue gum, bloekom) introduced from Australia, only E. cladocalyx (sugar gum), which is grown extensively in the Winter Rainfall Area of the Western Cape Province, is of toxicological significance. This species is regarded as the main cause of prussic acid poisoning in this area (D.J. Schneider, Veterinary Regional Laboratory, Stellenbosch, personal communication, 1984). The leaves are low in volatile oils in comparison to other Eucalyptus spp., and this makes them particularly attractive to livestock (R. Poynton, Forestry Research, Department of Environmental Affairs, Pretoria, personal communication, 1985). Young leaves contain up to 0,59% HCN on a dry-matter basis (Finnemore et al., 1935, as cited by Everist46).
Acacia spp. (Fabaceae)
A. erioloba E. Mey
Camel thorn, kameeldoring
A. sieberiana DC. var. woodii (Burtt-Davy) Keay and Brenan
Paper bark thorn, papierbasdoring
A. caffra (Thunb.) Willd.
Common hookthorn, gewone haakdoring
Two species are of special significance, namely, kameeldoring, the dry, large, grey, velvety pods of which are sometimes milled and given to stock as fodder during droughts, and paperbark thorn or papierbasdoring, which has relatively large leaves and soft-thorned young shoots, and may be grazed by stock. Prussic acid poisoning has also been associated with Acacia caffra, especially young wilted twigs brought down by storms.
Manihot esculenta Crantz. (Euphorbiaceae)
The tubers of Manihot esculenta (casava), which are cultivated for human consumption and ethanol production, contain a cyanogenic glycoside, phaseolunatin (Figures 10 and 11). Occasionally feeding of residues of this plant has resulted in stock losses.124
Jatropha multifida L. (Euphorbiaceae)
The palatable leaves of the umbrella tree contain enough HCN to be fatal to stock.124 More information on the toxicity of this plant is given in Gastrointestinal tract.
Pathophysiology, diagnosis and treatment of prussic acid poisoning
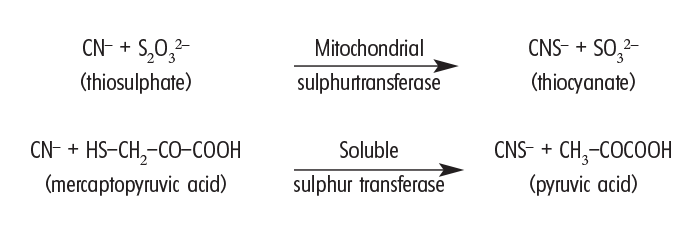
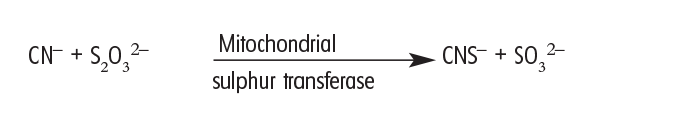
HCN and its alkali salts have a toxicity of c.2 mg/kg in most animal species if taken in at one time, and plant material containing more than 0,02% HCN can be regarded as potentially lethal.21 The liberated cyanide is rapidly absorbed from the gastrointestinal tract and if not present in excessive quantities is efficiently detoxified to thiocyanate by mitochondrial and soluble sulphur transferases (rhodanese) through the following two reactions:51
It is possible for an animal to ingest continually just less than a lethal dose of cyanide over extended periods without any harmful effects. However, when an excess of cyanide is in circulation, it binds stably with Fe3+ in the tissue cytochrome oxidase system, to halt electron transport, and consequently the chain of cellular respiration. This leads to tissue anoxia and death of the animal.
Tissue respiration in the aortic and carotid bodies is also affected and the hypoxic state so induced is reflected in the respiratory system (vide infra). In addition, poisoned animals successively experience rapid depression of cerebral, hypothalamic and midbrain activity, in that order. The brain stem (and hence the respiratory centre) is affected later in the course of the intoxication. The heart may continue to beat several minutes after respiration has ceased.11, 53
Oxygenation of blood is not impaired and the blood consequently remains red. The bright-red colour of the normally dark-red jugular blood is especially noticeable when poisoned animals are treated by intravenous injection (D.J. Schneider, Regional Veterinary Laboratory, Stellenbosch, personal communication, 1984). However, in our experience, the cherry-red mucous membranes and blood at necropsy are not as conspicuous as suggested in most textbooks. The blood is more usually dark-red, probably owing to respiratory depression which precedes cardiac arrest, coupled with deoxygenation of the blood by the non-inhibited portion of the cytochrome oxidase system.
Peracutely poisoned animals are frequently found dead, but some of the signs listed for acute poisoning may sometimes be evident. In acute intoxication, which occurs most often, respiratory distress is manifested as dyspnoea, polypnoea and cyanosis. These signs are accompanied by excitement and tremors which may go over into convulsions and finally paralysis and stupor. Tympany, frothing at the mouth and salivation may be present.
At necropsy signs of asphyxiation can be seen, including generalized congestion and cyanosis, congestion and oedema of the lungs, petechiae in the mucosa of the trachea and bronchi, and haemorrhages in the epi- and endocardium. Coagulation of the blood may be impaired.
Since HCN quickly dissipates or is rapidly degraded, specimens (rumen content and liver) for diagnosis should be taken as soon after death as possible. Although the HCN concentration in the muscle is lower than that in the liver and rumen, degradation in the muscles proceeds at a slower rate, making muscle tissue the specimen of choice where animals have been dead for some time.117 Specimens should either be kept frozen or preserved in 1% HgCl2. In the latter instance, a stable mercuric cyanide complex is formed which allows the specimens to be stored at room temperature (4 CN– + Hg2+ → [Hg(CN)4]2–). For quantitative analytical determination, a measured volume of 1% HgCl2, should be added to an approximately equal volume of ruminal content, liver or muscle. The tissue must be cut into 5 mm strips for better penetration of the HgCl2. However, the use of HgCl2 in the preservation of specimens for HCN determination has now largely fallen in disuse owing to the high toxicity of HgCl2.
As a rule of thumb, a diagnosis is confirmed when the HCN levels in ruminants are 1,4 μg/g in the liver taken two to four hours after death; 10 μg/g in the rumen content taken eight to 12 hours after death; and 6 μg/g in the muscles taken up to 20 hours after death (J.A. Minne, VRI, Onderstepoort, personal communication, 1984).
An old, very useful field test (sodium picrate test) using a stable reagent consisting of 0,5% picric acid and 5% NaHCO3 dissolved in water (Henrici, 1926, cited by Steyn114) is still sometimes employed. Filter paper strips are moistened with this bright yellow solution, blotted and dried. In a test tube a freshly prepared filter paper strip is suspended over either a few grams of ruminal content or crushed leaves, which have been moistened with water. The test tube is then corked and immersed in water at 37 °C. A change in colour of the filter paper from yellow to brick-red denotes the presence of HCN in the specimen. If this colour-change takes place within a few minutes it is significant, but if the change occurs after an hour or longer it is probably meaningless. This test is not specific but in practice is almost certainly indicative of the presence of HCN.
The treatment of cyanide intoxication consists of two basic steps. Firstly, the cyanide is removed from the cytochrome oxidase so that it can be fixed in a harmless form. Although cyanide (CN- has a greater affinity for tissue cytochrome Fe3+ than for methaemoglobin (MetHb), it can be removed from the cytochrome oxidase system by a high concentration of MetHb. This can be achieved by carefully controlled intravenous injection of NaNO2. The reactions are shown as follows:
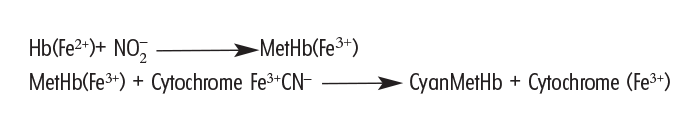
As a result of these reactions the cytochrome oxidase is restored and cyanide is fixed as non-toxic cyanMetHb.
The second step is to assist the mitochondrial sulphur transferase to convert free cyanide, or the cyanide in cyanMetHb, to thiocyanate (which can be excreted in the urine) by supplying sulphur in the form of sodium thiosulphate (‘hypo’) intravenously and per os.51
The recommended therapeutic dosages of NaNO2 and Na2S2O3 vary greatly from 4 and 12 mg/kg respectively to 22 and 660 mg/kg respectively in sheep14 and even 100 mg and 1 g/kg respectively in mice (Chen & Rose, 1952 cited by Burrows & Way14). Burrows and Way14 found that 22 mg NaNO2/kg and 660 mg Na2S2O3/kg increased the lethal dose of NaCN for sheep from 3,17 mg/kg to more than 50 mg/kg. At this therapeutic dose, methaemoglobin formation did not exceed 40% and this level was reached 45 minutes after administration. They are of the opinion that the above dose is the maximum that should be used in sheep.
Similar work in the bovine could not be traced, though this species is quite often involved in cyanide intoxication in South Africa. An apparently feasible treatment regime for all ruminants is described as follows:
An aqueous solution of 1% NaNO2 and 25% Na2S2O3 is administered intravenously at a rate of 1 mg/kg body mass, which is the equivalent of 10 and 250 mg/kg of the two therapeutic agents, respectively. The treatment may be repeated once. At the same time Na2S2O3 may be given orally at a rate of 0,5 g/kg as often as required. This treatment of ruminants is so effective, simple and cheap, that other treatments such as aminopropriophenone4, cobaltous chloride,14 hydroxycobalamine or aquocobalamine and cobalt EDTA, are unnecessary.
Recently the mechanism by which nitrite has its antidotal effect has been questioned, but its vasodilatory action is still regarded as important. The possibility has been mooted that sulphurated albumin would be a more readily available source of sulphur than thiosulphate. However, the nitrite-thiosulphate combination is still thought to be one of the most effective treatments of cyanide poisoning.22, 125
According to Steyn,115 prussic acid poisoning can be prevented by supplying sulphur through licks containing between 5 and 8% flowers of sulphur, by dosing adult goats and sheep c.10 g/day and cattle c.50 g/day, or by supplying as sole source, water containing ‘hypo’ (Na2S2O3) at a rate of 1 kg/2 000 l in summer and 1 kg/1 000 l in winter.
Chronic cyanide intoxication apparently occurs in man,24, 125 tobacco amblyopia and tropical ataxic neuropathy associated with chronic casava consumption being two such syndromes. In veterinary medicine11 a syndrome, referred to as Equine Sorghum Cystitis/Ataxia, in horses on Sorghum spp. or hybrid Sudan pastures, is thought to be related to chronic cyanide intoxication.
Clarke et al.,21 citing Hurst (1940, 1942), were of the opinion that although there is some experimental evidence of chronic cyanide poisoning in monkeys and sheep, there was no reason to believe that this type of poisoning occurs under field conditions.
In certain animal species, cyanogenic glycosides, after being absorbed intact from the gut, are hydrolysed intracellularly to produce their toxic effect. It is thought that chronic cyanide intoxication may be related to this phenomenon. Putative chronic cyanide intoxication has been diagnosed in South Africa in cattle and sheep exhibiting nervous signs such as depression, incoordination and paresis. The affected animals apparently responded favourably to intravenous treatment with sodium thiosulphate (‘Hypo’) (G. Harmse, Faculty of Veterinary Science, University of Pretoria, personal communication, 2004).
The goitrogenic effects of thiocyanate, the main metabolite of cyanide, is discussed in Urogenital system. This condition is sometimes seen in sheep on star grass pastures, especially if the intake of iodine is low, and iodine supplementation may be of therapeutic value.
Nitrate or nitrite poisoning
There are a number of examples where ruminal bacteria act on plant material to produce toxic substances, e.g. the hydrolysis of cyanogenic glycosides resulting in cyanide poisoning; formation of dimethyl disulphide from S-methylcysteine sulphoxide in Brassica poisoning; production of the lung toxin, 3-methylindole, from tryptophan fermentation; and the changing of the isoflavones (especially formononetin) in Trifolium spp. to equol, which is a strong oestrogenic metabolite. Likewise, nitrate is relatively non-toxic, but its conversion to nitrite, by ruminal micro-organisms, renders it toxic.1, 16 Nitrate poisoning has been succinctly reviewed by Pfister (1988).95
Table 1 Some nitrate-containing plants found in southern Africa
| Plant Species | Family | Common Name |
| Amaranthus spp. | Amaranthaceae | Pigweed, cockscomb, hanekam, misbredie |
| Brassica spp. | Brassicaceae | |
| B. napus L. B. oleraceae B. rapa L. | Rape, blaarraap Rape, kale, marrowstem kale Rape, turnip, raap, beeskool | |
| Beta vulgaris L. | Chenopodiaceae | Beetroot, beet, rooibeet, mangelwortel, mangel |
| Sorghum spp. | Poaceae | |
| S. bicolor (L.) Moench. subsp. drummondii (Steud.) De Wet (= S. sudanense (Piper) Stapf.) S. bicolor (L.) Moench. subsp. arundinaceum (Desv.) De Wet and Harlan | Sudan grass, Soedangras grain-sorghum, graansorghum | |
| Avena sativa L. | Oats, hawer | |
| Secale cereale L. | Rye, rog | |
| Triticum aestivum L. | Wheat, koring | |
| Zea mays L. | Maize, mielie |
Nitrate toxicity is a worldwide problem in stock and the terms nitrate or nitrite poisonings are often used synonymously to describe toxicoses caused by nitrate-containing chemicals and fertilizers, weeds, grasses, feeds and water. In South Africa, nitrate-containing plants play a relatively minor role in comparison to intoxication from contaminated water, but the plants listed in Table 1 may be of concern, Amaranthus spp. (Figure 12) being considered the most important of these.
Crop plants, such as cereal grasses (oats, wheat, barley, maize and rye), Beta vulgaris, Sorghum and Brassica spp. are sometimes associated with nitrate poisoning, particularly in ruminants.17, 18, 27, 68, 73, 90 These commodities are not always toxic and the nitrate concentration may vary according to species, growth stage and physiological condition of the plant, soil type and nitrogen content. Meteorological factors, such as temperature and rainfall, may also affect nitrate concentration.11, 27, 57 Toxic amounts of nitrates may be present in cultivated crops in the early growth stage, but may decline rapidly as the plant reaches maturity.11, 27 The conductive tissues of plants usually contain the highest level of nitrate, while the lowest concentrations are found in the leaves and fruits or grains.11, 31 Plants that are inclined to accumulate nitrate are often particularly dangerous after heavy nitrogenous fertilization, during periods of low light intensity, and damage by drought stress or after the application of herbicides, such as 2,4-D.11, 17, 31, 68, 95 Since the activity of nitrate reductase diminishes under low light intensity, enough soil moisture may be present during droughts to allow uptake of dangerous amounts of nitrate at night when activity of this enzyme is low. The situation may be compounded on the following day by slower reduction of nitrates owing to depression of nitrate reductase activity in the wilted plants. Cloudy weather could further inhibit this reduction, allowing toxic amounts of nitrate to accumulate.95 In southern Africa, it is common practice to graze cattle on drought-stricken maize or sorghum fields abandoned for seed production, but this can be hazardous. It is also worthwhile to note that the nitrate concentration of plants in close proximity to each other may vary significantly. Hibbs57 reported a tenfold difference in the nitrate content of Sorghum growing as close as 300 mm apart.
In general, the microbial conversion of nitrate to nitrite occurs mostly in the forestomachs of ruminants and in the gastrointestinal tract of human infants.11, 23, 31, 65 However, under optimal conditions of temperature, moisture and pH, enzymatic reactions in plants and bacterial fermentation in hay and silage may also play a role in this conversion.11, 18, 25, 27, 31, 90, 101 In this connection it should be mentioned that the weed Capsella bursa-pastoris has been implicated in nitrite poisoning in pigs in South Africa.126 In most instances, ensiling of forage tends to reduce the amount of nitrate by 40–60% through the reduction of some of the nitrate to oxides and nitrogen, while stacked or dry, cured forage loses little nitrate.11, 57
Apart from plants and chemicals, water can also be a potential source of poisoning. Nitrate can contaminate streams, dams and underground water by various means: through the application of more nitrate or nitrogenous fertilizer to the soil than plants can utilize; through the conversion to nitrate of ammonia in effluent of feedlots, sewage and decaying organic matter by soil bacteria; and through the fixation of aerial nitrogens by root nodules of leguminous plants, such as soyabeans and lucerne.11, 18, 31, 65, 68 Deep underground water in certain areas of the Springbok Flats (Limpopo Province), the Kalahari and Namaqualand (North West and Northern Cape provinces) in South Africa, and the southern and western parts of Namibia may contain dangerous concentrations of nitrate and nitrite.115
Nitrate is converted by bacterial nitrate reductase to nitrite in the rumen. If this reaction takes place faster than nitrite can be converted by nitrite reductase to ammonia, toxicity results.16 Nitrite is c.10 times more poisonous than nitrate31 and is absorbed into the circulation, where it oxidizes the iron in haemoglobin from the ferrous to the ferric state, thereby changing oxyhaemoglobin to methaemoglobin. The latter, being unable to carry sufficient oxygen, leads to tissue anoxia.2, 11, 18, 21, 70 The haemoglobin of foetuses and neonates is apparently more susceptible to methaemoglobin formation.11, 68 Apart from having an effect on the oxygen transport capacity of red blood cells, nitrite also causes dilatation of blood vessels and a drop in blood pressure.2, 21, 70 However, Asbury and Rhode2 showed experimentally that in calves the fall in blood pressure is not great enough to have a significant effect on clinical signs and death in nitrate toxicosis. Nitrate and nitrite may also have an irritant effect on the mucosa of the gastrointestinal tract and sometimes may be responsible for abdominal pain and diarrhoea in stock.18, 68
Whether the disease will follow an acute, subacute or chronic course in livestock is influenced by a number of factors, such as the nitrate level in the plant; the amount and rapidity of ingestion of the commodity and the rate of conversion in the rumen of nitrate to nitrite. Stock on a good-quality ration would seem to tolerate higher levels of nitrate than animals on a deficient diet.11, 18, 31 Murdock et al.84 also demonstrated that cattle become adapted to higher nitrate concentrations when nitrate is fed continuously.
In stock, acute poisoning occurs most frequently in hungry animals consuming large quantities of nitrate-containing feed in a relatively short time.31 Clinical signs are usually apparent one to four hours after ingestion of high nitrate feed or water, but may take up to five to eight days to develop.11 The signs include hyperpnoea; dyspnoea; rapid, weak pulse; progressive cyanosis; dirty, mucous membranes; incoordination; anxiety; muscle tremors; convulsions and sometimes also signs of abdominal pain and diarrhoea. These signs are often terminated by death in acute poisoning.11, 17, 21, 27, 31, 56, 68, 115 Abortion may occur in surviving pregnant animals11, 17, 27, 109 (see Urogenital system).
At necropsy the blood is dark and somewhat tarry and chocolate-brown, as a result of methaemoglobinaemia (which may only become discernible after exposure to the air); the lungs are congested and oedematous; scattered haemorrhages are often visible subcutaneously, as well as on the serosal and visceral surfaces; and there may be congestion of the mucosa of the gastrointestinal tract, especially of the rumen and abomasum.11, 17, 21, 27, 115
Subclinical or chronic nitrate poisoning has long been a matter of great controversy among veterinarians, but its existence is now more generally accepted. It has been produced in cows taking in sublethal amounts of nitrate over a prolonged period. Some cows appeared to be quite healthy, while others, although not apparently sick, showed signs of lowered milk production, roughened coat and digestive upset. Some of the pregnant animals aborted.18, 68 In the USA, this type of poisoning has been reported in cattle on rations of silage and legume hay (lucerne) and also on chopped fresh forage, especially Sudan grass and maize.18 Case18 stressed that whenever sheep or cattle abort, and if the cause is not infectious in nature, the possibility of subclinical nitrate intoxication should be investigated. Abortion, mostly in the last trimester, in cattle and sheep, has often been associated with nitrate poisoning abroad18, 68, 108, 109 (see Urogenital system). Other associated problems blamed on chronic nitrate intoxication, but which have not yet been proved to be caused by it, include poor growth rate, infertility, vitamin A deficiency, goitre and an increased susceptibility to infections.11, 27, 68
The diphenylamine (DP) test is a most useful qualitative field test for the detection of nitrates and nitrites in water, serum and other fluids in the animal, rumen and abomasal contents and plant material.18, 31, 64 It is based on the oxidation of DP via diphenylbenzidine to blue quinoidal compounds. Nitrates can be detected in the serum of animals even before the development of clinical signs.31 According to workers abroad, the reagent can be prepared by cautiously dissolving 62,5 mg of DP in 100 ml of 80% sulphuric acid and storing it in a brown bottle.18, 31 At the ARC-Onderstepoort Veterinary Institute, 0,5 g of DP is dissolved in 20 ml of distilled water and 80 ml of concentrated sulphuric acid is added. This results in a stable stock solution. For test purposes, one part of stock solution should be diluted with seven parts of 80% concentrated sulphuric acid immediately prior to use (A.T. Basson, ARC-OVI, personal communication, 1999). In the application of the test, personnel and other materials should be protected against the corrosive effects of this reagent. The test is conducted by placing one drop of serum or clear body fluid, such as urine, ascitic, thoracic, pericardial, intraocular or cerebrospinal fluids, on a spot test plate, smooth porcelain or any glass surface and then adding three drops of reagent. A dark-blue colour will develop within a few seconds if nitrates or nitrites are present, but any development of colour after 60 seconds should be disregarded. The amount of nitrate or nitrite present will determine the intensity and rapidity with which the blue colour develops.18, 31 Helwig and Setchell56 made certain observations on the diagnosis of nitrate poisoning in sheep and showed that nitrates can be detected up to 20 hours after death in the serum collected from the heart. They also demonstrated that blood methaemoglobin determination for routine diagnosis of nitrate poisoning is unsuitable when it takes specimens some hours to reach the laboratory. Samples mailed to the laboratory or held overnight should be frozen.11 DP reagent can likewise be applied to a cut surface of suspected plants to determine their nitrate content qualitatively. Contaminated water can be tested by running the heavy DP test solution carefully under a few millilitres of water in an inclined test tube. On light agitation, a purple ring will form at the junction of the two fluids. Nitrates can be measured quantitatively by extraction from plants with distilled water and determination of the concentration with a nitrate specific ion electrode17 or by using other standard procedures. Positive reaction with the DP test can also be obtained with other oxidizing compounds such as chlorates, bromates, chromates, permanganates, selenites, vanadates, molybdates, peroxides, higher metal oxides, antimony and iron.56 The compounds, however, are most unlikely to be present in biological material.
When the nitrate level in forage exceeds 1% on a dry weight basis or 1 500 ppm in water, acute poisoning in stock may be expected, although higher levels in forage and water have been tolerated by ruminants.11
Intravenous treatment of affected animals with a 4% aqueous solution of methylene blue at a level of 4 mg/kg body weight is very successful and animals which are apparently at the point of death will start to take food 10–15 minutes after injection.11, 27 Burrows and Way13 showed experimentally that a dose of 22 mg/kg gives the best results in sheep, and they warned that under-treatment will have a poor response.
According to Goldstein et al.,51 one per cent of the total haemoglobin in the blood is in the form of methaemoglobin. Constant reduction of any excess methaemoglobin takes place via the enzymes methaemoglobin reductases I and II. Methaemoglobin reductase I needs NADH for conversion, and although this process is slow, it is physiologically probably the more important of the two mechanisms. Methaemoglobin reductase II requires NADPH, but can also use glutathione and ascorbic acid as reducing agents. It is with this particular enzyme that methylene blue in its reduced form, leucomethylene blue, can interact as an intermediate electron acceptor between NADPH and methaemoglobin, thus accelerating the conversion of haemoglobin.51
Acacia nilotica (L.) Wild. ex Del. subsp. kraussiana (Benth.) Brenan (Fabaceae)
Scented thorn, stinkpeul, lekkerruikpeul
The pods of Acacia nilotica have been associated with methaemoglobinaemia, anaemia and abortion in goats in South Africa.118 Poisoning occurs especially in times of drought and food shortage, when excessive quantities of the dry pods are eaten by animals.
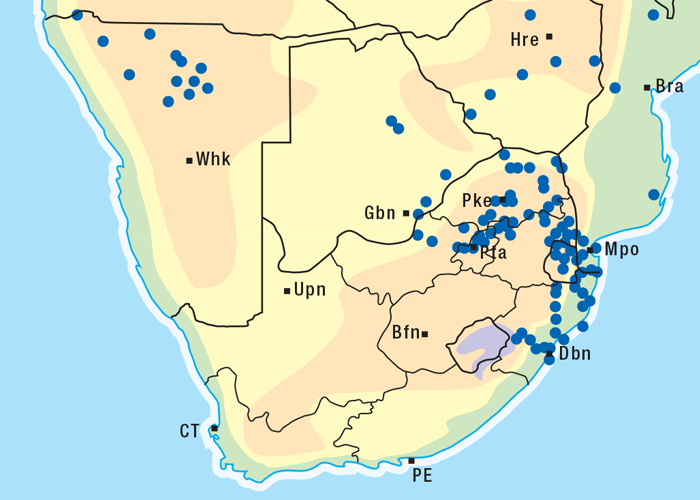
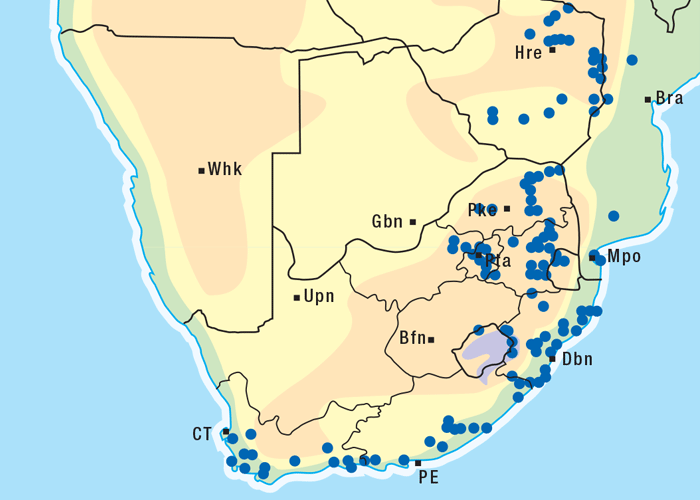
Acacia nilotica, a thorn tree, locally referred to as stinkpeul or lekkerruikpeul, grows up to 8 m high and often has an umbrella-like crown (Figure 13). The spines are weakly developed or robust and may be straight or slightly curved; the inflorescences are globose and yellow; the pods are long, containing five to 15 seeds arranged like a string of beads (Figure 14).118 Acacia nilotica grows in a variety of habitats from plains to mountain slopes, but is most commonly found in valleys. The tree is widely distributed in the northern and eastern parts of the former Transvaal, KwaZulu-Natal and in Namibia118 (Figure 15).
Apart from an outbreak of a nitrate-like poisoning in goats near Brits in the North West Province,118 the ingestion of parts of the tree has generally been considered not to be harmful to stock. Saponins and tannins,118, 124 but no nitrates, nitrites or other strong oxidizing agents, were found in the pods that could explain the development of methaemoglobinaemia.118 Preliminary investigation by Terblanche et al.118 could also not show any inhibition of the methaemoglobin reductase system or triose phosphate dehydrogenase in red blood cells. There was, however, a tendency to increased red cell fragility.118
Abortion was reported in goats that were allowed to graze in a camp containing many A. nilotica trees.118 Some of the affected animals that died showed anaemia, icterus and a slight methaemoglobinaemia. Dry, ripe pods of the thorn tree were collected from the same area of the occurrence of the outbreak of poisoning. The pods were then ground and dosed per stomach tube to six mature goat ewes, five of which were heavily pregnant. Acute toxicity occurred in three pregnant goats receiving increasing daily dosages (5–30 g/kg) over a short time (7–12 days). Two of these animals aborted on the seventh and twelfth days of the experiment and died on the eighth and fourteenth days, respectively. Clinical signs included methaemoglobinaemia, tachycardia, hyperpnoea, and ruminal stasis, and the goats acquired a sweet, putrid (garlic-like) smell. Some animals showed a terminal diarrhoea and dyspnoea. Chronic toxicity was induced in two pregnant goats that received 23 relatively small doses (5 g/kg/day) of the same material over a long period (31 and 32 days). Both animals survived, but one of the ewes aborted on the fourth day of the experiment. Clinical signs included occasional spells of tachycardia and hyperpnoea, and dirty discolouration of the mucous membranes indicated that the two animals were slightly methaemoglobinaemic.118
Pathological changes in the field cases included methaemoglobinaemia (Figure 16), anaemia, slight icterus, haemoglobinuria, nephrosis, varying degrees of centrilobular degeneration and necrosis in the liver and slight hydropericardium and pulmonary oedema. Methaemoglobinaemia was most striking in the experimental cases, but anaemia, icterus, haemoglobinaemia and haemoglobinuria were absent.
Pteridium aquilinum (L.) Kühn (Dennstaedtiaceae)
(= Pteris aquilina L.)
Bracken fern, adelaarsvaring
Bracken poisoning is a condition of cattle characterized by depression of the bone-marrow activity, leukopaenia (especially of granulocytes), thrombocytopaenia and severe haemorrhages. This condition has many features in common with stachybotryotoxicosis (vide infra), as both have a radiomimetic effect on the body.
Bracken is a perennial fern about 1,5 m tall which grows from underground stems covered by reddish hairs (Figure 17). The young leaves are coiled like a watch spring and hairy, but the hairs are lost as the fronds unfold, so that the upper surface becomes glossy.121 Unlike other, possibly confusing, large ferns in which the sori (groups of sporangia) on the back of fronds are often round and situated on the veins of the frond, the sori in Pteridium are arranged linearly and submarginally. Although some other ferns also have this arrangement, the disposition of the sori in conjunction with the hard leathery leaves is typical for Pteridium (Clare Reid, Botanical Research Institute, Pretoria, personal communication, 1986).
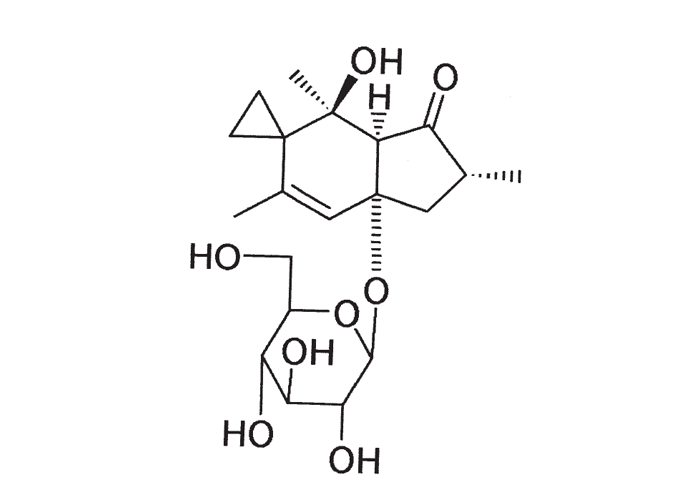
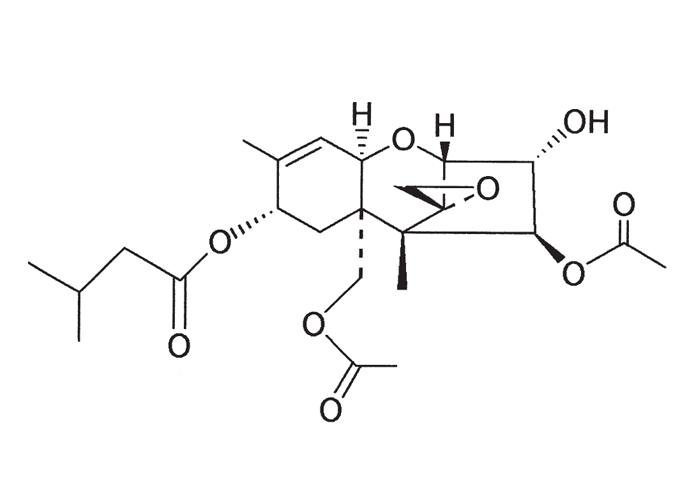
Pteridium aquilinum contains thiaminase I, which gives rise to neurological disorders in horses and sheep36, 37, 46 (see Central nervous system). However, thiaminase I is not the main toxin in the plant, but ptaquiloside, a norsesquiterpene glucoside (Figure 19). Both bracken fern and ptaquiloside have been implicated in a haemorrhagic tendency with bone-marrow suppression in cattle;40, 45, 62, 119 neoplasia particularly of the urinary bladder in cattle91–93, 100 (see Urogenital system) as well as a variety of neoplasms in other species38, 93, 94 and progressive retinal degeneration (night blindness) in sheep.63 Of these, the haemorrhagic syndrome is economically the most important.
Bracken poisoning is reported mostly in hill camps where the ecology is conducive to fern growth.40 In South Africa, outbreaks of poisoning occur every now and then in cattle in the mountainous parts of the Mpumalanga, KwaZulu-Natal and Eastern Cape provinces (Figure 18). However, the only documented outbreak of the disease in cattle in South Africa occurred on a farm in the foothills of the Drakensberg in the Estcourt district of KwaZulu- Natal, during which 55 cattle were affected and 28 animals died.119 Outbreaks of poisoning in cattle in southern Africa are sporadic and are usually associated with a scarcity of food, perhaps as a result of overstocking, drought or after the veld has been denuded by fires.44, 119 Bracken grows vigorously, and the young fronds, which grow faster than grass, may be eaten by hungry animals. Some authorities are of the opinion that cattle may even acquire a taste for the plant.41 By nature, sheep are more tolerant of bracken poisoning than cattle, and because of their selective grazing habits, sheep seldom eat sufficient of the plant to be troubled.40, 81, 82 The green bracken, both fronds and rhizomes of which are turned up by ploughing, is most toxic to cattle,40, 44 but poisoning has also been reproduced by sun-dried bracken or powdered rhizomes.40–43 Dried plants, used as bedding and eaten by calves, have been reported to be toxic.40, 44 The toxin is thought to be cumulative40 and, depending on the degree of toxicity of bracken, large quantities of the plant must usually be ingested for a few weeks before having an effect on the animal.40 There are reports where apparently healthy animals have succumbed to the disease up to eight weeks after being moved from bracken-infested camps to non-infested pastures.41
Bracken poisoning has been reproduced experimentally by a number of workers abroad.41, 42 Fresh, green bracken reportedly reproduces the acute condition in cattle more rapidly than air-dried bracken from which the disease develops within 30–80 days of feeding the plant material.43 Four 16-month-old bovines died 58–73 days after commencement of the experiment, at which stage each had consumed 211–275 kg of grounded bracken ad libitum.41
Three calves, each receiving 95,5 kg of dried bracken, died on the thirty-fifth day of the experiment, showing typical signs of poisoning.41 According to Evans et al.,44 cattle must eat c.1 kg of dry green fronds and rhizomes per day for two to four weeks before signs, which usually appear within one to two months, become evident.
For many years the toxic principle responsible for bone-marrow suppression, and consequently the haemorrhagic tendencies, had eluded researchers.44 In 1984 Hirono and co-workers, however, induced acute bracken fern toxicity in a calf with ptaquiloside (Figure 19) isolated from a boiling water extract of bracken fern.62 A six-month-old calf was drenched at different dosage levels six days out of every seven days for the following periods: 400 mg/day for the first 24 days, 800 mg/day for 14 days and 1 600 mg/day for four days. The calf received a total amount of 27,2 g ptaquiloside during this time and was observed for 39 days after dosing was discontinued. Initially, there was a leucocytosis and it reached a maximum on Day 17 of the experiment. The leucocyte counts then dropped to a level of 4,7 x 109/litre on Day 64 of the experiment. The thrombocyte levels showed a relatively slow decline during the dosage period. No clinical signs were noted during the experiment, and the calf was necropsied 86 days after the start of ptaquiloside administration. Apart from a fatty appearance of the femoral bone-marrow, no haemorrhages or other gross changes were observed. Microscopically, there was severe depression of the bone-marrow activity, characterized by a replacement of most of the haemopoietic cells by fat with only islands of normal marrow remaining. The megakaryocyte numbers were markedly reduced in the sternal bone-marrow.
In dosing trials, involving the separate administration of the plant and ptaquiloside, Hirono et al.63 in 1993 reproduced progressive retinal degeneration in sheep, providing conclusive evidence of the causative role of ptaquiloside also in this aspect of poisoning with bracken.
Poisoning in mature cattle is usually referred to as the ‘enteric’ type and is characterized by depression and anorexia; mucus, discharged from the nose, that may be blood-tinged, or free blood can ooze from the nostrils; haemorrhages, particularly notable in the conjunctiva and vagina; clots of blood in the faeces; and fever which occurs terminally.40, 41, 44, 85, 119 The morbidity is usually low, but most affected animals, notwithstanding therapy, will die.41, 44
As a result of the fever and widespread haemorrhaging associated with bracken poisoning in cattle, a controversy initially existed as to whether the disease was an intoxication or a haemorrhagic septicaemia.40, 41 While Evans40 could find no close correlation between positive bacterial blood cultures and the presence of fever, Naftalin and Cushnie85 noted that a bacteraemia coincided with the rise in temperature.
At necropsy, numerous haemorrhages and sometimes also haematomas are scattered throughout the body, and free blood and many ulcers are present in the digestive tract.41, 44, 119 Acute infarcts in the spleen, liver and kidneys are thought to be the result of bacterial emboli which originate from infected ulcers in the gut.85, 119 The leukocytic infiltrations at the periphery of these infarcts were apparently not as dense as would have been expected, and comprised mainly round cells. The haematological picture of affected cattle reveals thrombocytopaenia and a leukopaenia with almost a complete disappearance of all white blood cells, except the lymphocytes.41, 42, 44, 85, 119 Other changes include prolonged bleeding time; defective clot retraction; increased capillary fragility; suppression of the granulocytic and thrombocytic (megakaryocytes) series and, occasionally, in the late stage of the disease, also suppression of the erythrocytic series in the bone-marrow.40–45 Bone-marrow lesions have also been reproduced in sheep, but this species appears to be more resistant than cattle.81, 82 Evans40 pointed out that there are many similarities between X-ray irradiation and bracken poisoning in adult cattle and speculated that a radiomimetic substance in the plant may be responsible for the lesions in this intoxication.
In contrast to mature cattle, affected calves often show a fever without external signs of bleeding, and oedema of the throat, resulting in difficult breathing and roaring, referred to as the ‘laryngitic’ type.40, 41, 44 Apart from oedema of the throat region and a few petechial haemorrhages that may be present in the carcase, there is little evidence of internal bleeding at necropsy.41 Evans et al.39 showed an increase in abnormal fibrinogen and heparin-like substances in the blood of affected calves. They and other workers speculated that heparin and histamine may be released from mast cells and give rise to shock and sensitivity conditions which may explain the ‘laryngitic’ type of poisoning and also the ulcerations and capillary fragility.40
According to some overseas reports, antibiotic treatment to control secondary infections or any other therapy would seem not to affect the final outcome of the disease.43 Tustin et al.119 found that intravenous injection of 10 ml of 1% protamine sulphate per animal to counteract the anticoagulant effect of the heparin-like substance, together with the administration of citrated blood transfusions, gave outstanding results.
Stachybotrys spp. (Fungi: Hyphomycetes)
S. atra Corda (= S. alternans Bonorden, var. Jateli)
(= S. chartarum (Ehrenb. ex Link) Hughes)
Stachybotryotoxicosis is a mycotoxicosis of stock and humans caused by fungal metabolites loosely grouped as the trichothecenes. These have a radiomimetic effect especially on the lymphoid tissues, bone-marrow, epithelium of the gastrointestinal tract and the skin.19, 80, 105, 106, 123 Most reports of this intoxication come from eastern European countries and the former USSR,80 however, the disease has also been documented in sheep in the Western Cape Province.106
Stachybotrys spp. are cosmopolitan, saprophytic, cellulose-degrading fungi and have been isolated especially from straw of cereal grains (wheat, oats, barley, rye), but also from peas, cotton and sugar-cane roots. In straw, black masses of spores are often discernible at the nodes of the plants (Figure 20).58, 80, 106 Stachybotryotoxicosis is most likely caused by one or more of the macrocyclic trichothecenes, including roridin E, verrucarin J, and satratoxins F, G and H, although as yet unidentified toxic fungal metabolites may also play a role in this disease complex.32, 33, 58, 80, 106 Some of the above-mentioned toxins are also produced by the fungi, Myrothecium roridum, M. verrucaria and M. leucotrichum.30, 55, 80 The toxic metabolites of Stachybotrys spp. are resistant to gastric juices, X-ray and ultraviolet irradiation, and infected feed can apparently remain toxic for years.106, 120
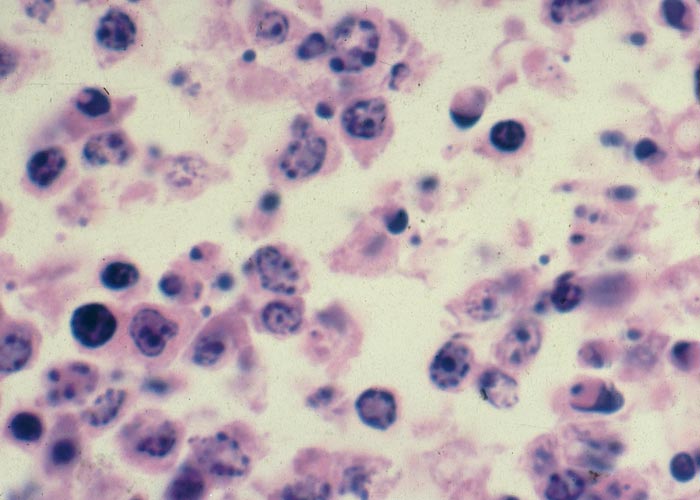
Trichothecenes are cytotoxic, and they inhibit protein synthesis of eukaryotic cells in plants and animals, culminating in atrophy, degeneration and/or necrosis of the lymphoid tissues (Figure 21) (lymph nodes, spleen, thymus, gut-associated lymphoid tissue etc.); the haemopoietic cells including megakaryocytes in the bone-marrow; the lining epithelium of the gastrointestinal tract, but especially that of the mouth (Figure 22) and small intestine; and the skin. Damage to these sites often results in leukopaenia, coupled with suppression of the immunological response; anaemia and thrombocytopaenia, associated with bleeding tendencies (Figure 23) (subcutaneous, intermuscular and serosal haemorrhages, epistaxis, enterorrhagia, etc.); erosions and ulcerations in certain parts of the digestive tract; and erythema, oedema, degeneration and necrosis of the skin. The local application of the toxins to the shaved skin of rabbits or other laboratory animals has in fact become a standard biological test for the trichothecenes. The degree of tissue damage or whether intoxication manifests acutely or chronically depends on the specific trichothecene; the route of administration, dosage level, and intervals between exposures to the toxin; and the species involved. Under field conditions poisoning is usually chronic following low level intake of the toxin over a prolonged period.12, 19, 26, 55, 80, 105, 106, 120, 123
Outbreaks of stachybotryotoxicosis have been associated with the ingestion of, or contact with hay, feed or litter contaminated by toxic strains of the fungus.32, 58, 80, 103, 106 Animals may apparently even develop a taste for Stachybotrys-infected straw.26 The first report of the disease came from Russia during the early 1930s when thousands of horses died and farm labourers handling contaminated hay became ill.47 Since then, field outbreaks of this relatively rare mycotoxicosis have been reported in cattle, sheep, chickens and zoo animals47, 58, 80, 103, 106 and the disease has also been produced experimentally with mouldy feed, toxic Stachybotrys cultures, or various extracts of these materials, in horses, cattle, sheep, pigs, poultry, laboratory animals (mice, guinea-pigs, rabbits) and dogs.47, 48, 80, 106, 107, 123
In horses, stachybotryotoxicosis typically occurs after the ingestion of straw containing low levels of toxin80 and presents as a stomatitis, characterized by deep necrotic bleeding fissures in the angles of the mouth and marked swelling of the lips47, 59, 60 (see also The skin and adnexa). Salivation, rhinitis and conjunctivitis may accompany the mouth lesions. In spite of continued ingestion of the toxin, the lesions regress within approximately two weeks before the disease enters the second stage which is marked by thrombocytopaenia, leukocytopaenia, and prolonged prothrombin time. Terminally, thrombocytopaenia and leukocytopaenia become more severe, the blood doesn’t clot well, fresh necrotic coalescing areas develop in the mouth and the animal shows a fever and a weak arrhythmic pulse before death intervenes.80 Very occasionally horses will develop the atypical or shock form of the disease within 10–12 hours and up to 72 hours after consumption of large quantities of heavily contaminated hay. In this instance, no mouth lesions or blood abnormalities are apparent, but affected horses reveal nervous signs, such as hyperaesthesia, hyperirritability, loss of vision, inco-ordination, tremors, clonic spasms, increased peristalsis, atony of the gut and diarrhoea. Death appears to be due to respiratory failure.47, 59, 80
Cattle are less susceptible than horses to stachybotryotoxicosis, but otherwise the disease is very similar in both.26, 58, 80 Clinical signs include anorexia and weakness, and some animals reveal a haemorrhagic diarrhoea, epistaxis, focal ulcerative stomatitis and purpuric haemorrhages. Hintikka60 referred to Yugoslavian workers who reported that cattle which had been given mouldy hay for bedding, developed skin lesions on the scrotum and around the dewclaws in addition to the more common signs. The majority of cases show thrombocytopaenia and leukocytopaenia. Deaths are mostly restricted to young animals where the mortality ranges between 90 and 100%.26, 58, 80
Stachybotryotoxicosis in sheep is marked by weakness, anorexia, rhinitis, epistaxis, haemorrhagic diarrhoea, leukocytopaenia, thrombocytopaenia and hyperthermia.80, 106 In South Africa, an outbreak of the disease has been reported in a flock of Mutton Merino sheep, during which 109 out of 568 animals died after continual feeding of home-made sheep cubes prepared from visibly mouldy wheat, oats or barley straw or a mixture of the three in combination with lucerne.106 The animals started dying approximately one month after continual feeding of the pellets, and mortalities were still reported three to four weeks after withdrawal of the cubes. Abortion was reported and, as a result of the impaired immunological response many animals suffered from secondary bacterial infections, such as pasteurellosis and colibacillosis. The clinical signs and lesions of affected animals were very similar to those previously reported for stachybotryotoxicosis, save for the low incidence of oral lesions (a single ulcer was found in the pharynx of one animal only) and the very marked anaemia in some of the sheep in the later stages of the outbreak. Mycotoxicological investigations revealed the presence of toxigenic strains of S. atra on the wheat and barley straw; a positive skin test in rats was obtained with diethyl ether extracts of wheat straw, sheep cubes and S. atra culture material; and the presence of 12,13- epoxytrichothecenes (Figure 24) in these extracts was confirmed by thin layer chromatography.
Pigs have fallen victim to the disease after infected straw was used as bedding. Dry necrosis of the skin of the teats occurred in the sows, while the suckling piglets, before they died, developed necrosis of the snout and mouth and haemorrhagic tendencies.61, 80
As with most mycotoxicoses, withdrawal of the suspected feed is most important to prevent further losses. Blood transfusions have been used with some success to treat affected sheep in South Africa.106
Melilotus spp. (Fabaceae)
M. alba Medik.
White sweet clover, witstinkklawer
M. indica (L.) All.
Annual yellow sweet clover, eenjarige geelstinkklawer
The prolonged feeding of spoiled sweet clover (Melilotus spp.) (Figure 25) hay or silage, particularly to cattle, causes a haemorrhagic diathesis, known as ‘sweet clover disease’.49, 69, 70, 102, 113, 127 This condition occurs from time to time in stock abroad, but as far as can be ascertained it has not been diagnosed in South Africa.
Sweet clover hay, stacked with a moisture content of c.50%, spoils readily and becomes toxic. The toxicity may be retained for several years.73 During spoilage of Melilotus spp. hay or silage, the coumarin in the plant is modified to dicoumarol by microbial action.15, 69, 113, 127 Dicoumarin or dicoumarol has a potent anticoagulative effect on blood and many derivatives, e.g. warfarin, are used commercially as rodenticides.69 The action of dicoumarin is to suppress the synthesis in the liver of prothrombin and clotting factors VII, IX and X, all of which require vitamin K for their formation.69, 129 Normally, vitamin K is converted in the liver by microsomal epoxidase to phylloquinone epoxide, which is converted back by microsomal reductase to vitamin K.130 The action of dicoumarol supposedly inhibits the formation of vitamin K from phylloquinone epoxide.130 Apart from its anticoagulative effect, dicoumarin also increases the permeability of capillaries. Kahn et al.71 showed, ultrastructurally, a marked loss of ground substance and decreased organellar content in capillary endothelial cells of rats treated with sodium warfarin. Cattle, pigs and rabbits are most susceptible, while horses and sheep are more resistant to sweet clover poisoning.70, 73 Young calves are often more severely affected than mature cattle, presumably because their vitamin K reserves are lower than those of adults.70 Dicoumarin also crosses the placenta, and new-born calves have been reported to be affected, while their dams were apparently healthy and did not reveal any signs at parturition.49, 69
Large quantities of poisonous hay or silage have to be eaten by cattle for about one month before bleeding tendencies are manifested.69, 73 The onset of bleeding in affected animals is often sudden and includes varying-sized haemorrhages and haematomas, particularly subcutaneously and intermuscularly, in parts of the body often subjected to trauma, such as over bony prominences and the ribs. Haemorrhage may also be noticed on the serosal surfaces, subcapsularly in many organs, in the ventricles in the brain, medullary cavities of long bones, etc.69, 70, 73, 102 Animals may also exhibit epistaxis and melaena, and fatal bleedings may be experienced during surgical procedures, such as dehorning and castration.70, 102 Post-partum haemorrhage is a common occurrence.73
The mortality in stock suffering from sweet clover disease is high, because of internal and external haemorrhage, and most animals will die within three days after the appearance of the first signs.73 Affected animals can be treated successfully with repeated doses of vitamin K1 (phytomenadione), and fresh blood transfusions.49, 127 It is suggested that at least ten days should elapse after withdrawal of the toxic hay or silage before any surgical procedure is attempted.49
Discussion
Haemolytic anaemia is common in stock in the African continent and may have different aetiologies. Normally, a small number of red blood cells is constantly undergoing intravascular destruction in the living animal. The released haemoglobin is rapidly bound in the plasma by three protective proteins of which haptoglobin is the most important.70 The bound haemoglobin is then taken up by the liver and processed. If the free haemoglobin in the circulation exceeds the binding capacity of the haptoglobin, it is filtered in the kidneys. The free haemoglobin, not bound to haptoglobin or passed in the urine, is oxidized to methaemoglobin, because it is not protected by the reductase system of intact red blood cells.70
In South Africa, sheep frequently suffer from a haemolytic anaemia, resulting from chronic copper poisoning, following prolonged intake of relatively high levels of copper in commercial rations or in natural pastures in the semi-arid Karoo. Copper is a strong oxidant and when released in large quantities from the liver into the circulation, it will exceed the capacity of the carrier proteins in the plasma and exhaust the anti-oxidant glutathione system in red blood cells. The oxidative effects of copper give rise to denaturation of the haemoglobin, and consequently the formation of spherical, refractile Heinz-bodies, as well as damage to the cell membranes and haemolysis.70, 111
Similar changes in red blood cells have been reported in poisonings with Brassica spp. and Allium cepa in stock. The relatively non-toxic S-methylcysteine sulphoxide in Brassica spp. is converted by ruminal micro-organisms to the haemolytic factor, dimethyl disulphide,1, 16, 111 which has a similar action to the toxic principle, n-propyl disulphide in A. cepa.122 In both poisonings, the enzyme glucose-6-phosphate dehydrogenase and therefore the hexose monophosphate pathway in red blood cells, is impaired.111, 122 This culminates in deficient reducing power, oxidative changes in the haemoglobin and damage to the cell membranes.
Often in bone-marrow responsive anaemic conditions, such as in chronic copper poisoning and intoxications with Brassica spp. and Allium cepa, the RNA, when stained with methylene blue, is visible as a few basophilic aggregates in newly released red blood cells. These cells are approximately one and a half times the size of normal red blood cells and are referred to as reticulocytes. In all animal species, except the horse, reticulocytes in the peripheral circulation are indicative of erythrocyte production.70
Haemolysis is not a feature of poisoning with nitrate and prussic acid-containing plants: instead, in the former, the oxygen transport capacity of red blood cells is impaired, leading to asphyxiation at the cellular level. Nitrite oxidizes the iron in haemoglobin from the ferrous to the ferric state, thereby changing oxyhaemoglobin to methaemoglobin and hydrocyanic acid inhibits the action of cytochrome oxidase in tissues.
In this chapter, the radiomimetic effect on the bone-marrow of the macrocyclic trichothecenes of the fungus Stachybotrys atra and ptaquiloside, extracted from bracken fern Pteridium aquilinum, have been discussed. In both toxicoses, the bone-marrow is suppressed and the diseases are marked by fever, impaired immunologic response, bleeding tendencies and, in the more chronic cases, also anaemia. Leukocytopaenia and thrombocytopaenia are usually prominent in stachybotryotoxicosis and bracken fern poisoning.12, 40, 44, 80, 106, 119
Immunosuppressive activity also occurs with two other mycotoxins, namely, aflatoxin (see Liver) and ochratoxin A (see Urogenital system). Pier and McLoughlin97 reviewed the literature on mycotoxic-induced immunogenesis and found that protein synthesis is primarily inhibited by all three fungal metabolites (trichothecenes, aflatoxin, ochratoxin A), but that each exerts its action at a different site. Aflatoxin has a suppressive action on complement (C2), interferon, phagocytosis of macrophages and cell-mediated immunity (CMI) and is responsible for impaired immunogenesis without suppressing antibody formation. Only at very high levels of aflatoxin is immunoglobulin (Ig)A and IgG, but not IgM, suppressed.97 Several of the trichothecenes have been associated with alterations in serum proteins and Ig profiles, reduced antibody formation, thymic atrophy and aplasia and generally reduced CMI.97 These fungal metabolites therefore present an insidious threat to animals and humans, since the level of toxin in feed and foodstuffs necessary to exert an immunosuppressive action is below the levels that cause clinical disease of that specific mycotoxin. The true underlying cause may thus go undetected and only the clinical signs of the secondary infection may be apparent in the host.97
Apart from the mycotoxins and bracken fern discussed in this chapter, there are few other plant poisonings that affect the lymphoid tissues. The toxic principles, ricin in Ricinus communis (castor-oil plant), curcin in Jatropha curcas (physic or purge nut), abrin in Abrus precatorius (love or lucky bean, minnie-minnies) and modeccin in Adenia digitata, are all toxalbumins or lectins and therefore antigenic. Of these plants, Ricinus communis is most commonly associated with losses in stock, horses being particularly susceptible.20, 50, 79 Typically, ricin is responsible for a severe gastroenteritis, but also causes pyknosis and karyorrhexis of lymphocytes in lymphoid organs20, 50 (especially the mesenteric lymph nodes) similar to that often reported for many acute viral infections in animals. Ricin, the cause of agglutination of red blood cells, results in thrombosis in capillaries and haemorrhages seen in various organs and on the serosal surfaces of affected animals.50 For further information on these four plants, Gastrointestinal tract should be consulted.
Thrombocytopaenia coupled with defective haemostasis in stock has been associated with the radiomimetic effect on the bone-marrow of the toxic principles of stachybotryotoxicosis and bracken fern poisoning. There are many causes of thrombocytopaenia in animals and humans, including the invasion of the bone-marrow by neoplasms and the result of several bacterial, viral and rickettsial infections. It is often a consequence of some drugs and X-ray irradiation, or has an immunologic origin.67, 69, 70, 88, 128
In most species, thrombocytes can be produced from megakaryocytes within four days.70 Mature platelets are discoid in shape and about one third to one half the size of red blood cells and compared to young platelets, which are usually larger and exhibit greater basophilia, are less granular and lack basophilia.70 Immature thrombocytes, sometimes referred to as shift platelets, are indicative of increased platelet production.70 Apart from its function in coagulation of blood, the endothelial integrity is partly sustained by thrombocytes, and haemorrhage per diapedesis through the intact endothelium of vessels follows, if the circulating platelet numbers are reduced.
Defective haemostasis in animals and humans is not only part of quantitative and qualitative platelet disorders but is also influenced by many other factors85, 128 including acute and chronic liver diseases (see Liver). Although it has not been reported in South Africa and occurs rarely worldwide, the prolonged consumption of spoiled sweet clover (Melilotus spp.) hay may cause excessive bleeding in stock. In this instance, dicoumarol in the spoiled hay or silage is a vitamin K antagonist which suppresses the synthesis of prothrombin and clotting factors VII, IX and X in the liver.129
References
- ALLISON, M.J. 1978. The role of ruminal microbes in the metabolism of toxic constituents form plants. In: KEELER, R.F., VAN KAMPEN, K.R. & JAMES, L.F. (eds.) Effects of poisonous plants on livestock. New York, San Francisco, London: Academic Press.
- ASBURY, A.C. & RHODE, E.A. 1964. Nitrite intoxication in cattle: The effects of lethal doses of nitrite on blood pressure. American Journal of Veterinary Research, 25, 1010–1013.
- BATH, G.F. 1979. Enzootic icterus: a form of chronic copper poisoning. Journal of the South African Veterinary Association, 50, 3–14.
- BLOOD, D.C., RADOSTITS, O.M. & HENDERSON, J.A. 1983. Veterinary Medicine. 6th edn. London: Baillière Tindall.
- BOTHA, C.J., NAUDÉ, T.W., SWAN, G.E., DAUTH, J., DREYER, M.J. & WILLIAMS, M.C. 1993. The cupreretic effect of two chelators following copper loading in sheep. Veterinary and Human Toxicology, 35, 409–413.
- BROWN, J.M.M. 1962. Advances in ‘geeldikkop’ (Tribulosis ovis) research. V. Aetiological factors in ‘geeldikkop’ and enzootic icterus. Journal of the South African Veterinary Medical Association, 33, 493–507.
- BROWN, J.M.M. 1963. Biochemical lesions in the pathogenesis of geeldikkop (Tribulosis ovis) and enzootic icterus in sheep in South Africa. Annals of the New York Academy of Sciences, Art 2, 504–38.
- BROWN, J.M.M. 1964. Advances in geeldikkop (Tribulosis ovis) research. VI. Studies on selected aspects of the biochemistry of geeldikkop and enzootic icterus. Journal of the South African Veterinary Medical Association, 35, 507–530.
- BROWN, J.M.M. 1968. Biochemical studies on geeldikkop and enzootic icterus. Onderstepoort Journal of Veterinary Research, 35, 319–576.
- BROWN, J.M.M. & DE WET, P.J. 1962. A preliminary report on the occurrence of selenosis in South Africa and its possible role in the aetiology of Tribulosis (geeldikkop), enzootic icterus and some other disease conditions encountered in the Karoo areas. Onderstepoort Journal of Veterinary Research, 29, 111–135.
- BUCK, W.B., OSWEILER, G.D. & VAN GELDER, G.A. 1982. Clinical and Diagnostic Veterinary Toxicology. 2nd edn. Iowa, USA: Kendal/Hunt Publishing Company.
- BUNNER, D.L., WANNEMACHER, R.W., NEUFELD, H.A., HASSLER, C.R., PARKER, G.W., COSGRIFF, T.M. & DINTERMAN, R.E. 1985. Pathophysiology of acute T-2 intoxication in the cynomolgus monkey and rat models. In: LACEY, J. (ed.) Trichothecenes and other mycotoxins. Proceedings of the International Mycotoxin Symposium, Sydney, Australia: John Wiley & Sons Ltd.
- BURROWS, G.E. & WAY, J.L. 1975. Methylene blue versus nitrite. Proceedings of the United States Animal Health Association, Annual Meeting. pp. 266–270.
- BURROWS, G.E. & WAY, J.L. 1979. Cyanide intoxication in sheep: Enhancement of efficacy of sodium nitrite, sodium thiosulphate and cobaltous chloride. American Journal of Veterinary Research, 40, 613–617.
- CAMPBELL, H.A. & LINK, K.P. 1941. Studies on the haemorrhagic sweet clover disease. IV. The isolation and crystallization of the haemorrhagic agent. Journal of Biological Chemistry, 138, 21–33.
- CARLSON, J. & BREEZE, R.G. 1984. Ruminal metabolism of plant toxins with emphasis on indolic compounds. Journal of Animal Science, 58, 1040–1049.
- CARRIGAN, M.J. & GARDNER, I.A. 1982. Nitrate poisoning in cattle fed Sudan (Sorghum sp. hybrid) hay. Australian Veterinary Journal, 59, 155–157.
- CASE, A.A. 1957. Some aspects of nitrate intoxication in livestock. Journal of the American Veterinary Medical Association, 130, 323–329.
- CIEGLER, A. 1975. Mycotoxins: Occurrence, chemistry, biological activity. Lloydia, 38, 21–35.
- CLARKE, E.G.C. 1947. Poisoning by castor seed. Veterinary Journal, 103, 273–278.
- CLARKE, MYRA, L., HARVEY, D.G. & HUMPHREYS, D.J. 1981. Veterinary Toxicology. 2nd edn. London: Baillière Tindall.
- COHEN, M.A. & GUZZARDI, L.J. 1984. A letter on the treatment of cyanide poisoning. Veterinary and Human Toxicology, 26, 503–504.
- COMLEY, H.H. 1954. Cyanosis in infants caused by nitrates in well water. Journal of American Medical Association, 129, 112–116.
- CONN, E.E. 1978. Cyanogenesis, the production of hydrogen cyanide, by plants. In: KEELER, R.F., VAN KAMPEN, K.R. & JAMES, L.F. (eds.) Effects of poisonous plants on livestock. Proceedings of a Joint United States-Australia Symposium on poisonous plants, Utah State University, Logan, Utah. June 19–24, 1977. New York: Academic Press.
- CRAWFORD, R.F., KENNEDY, W.K. & DAVIDSON, K.L. 1966. Factors influencing toxicity of forages that contain nitrate when fed to cattle. Cornell Veterinarian, 56, 1–17.
- DANKO, G. 1975. Stachybotryotoxicosis and immunosuppression. International Journal of Environmental Studies, 8, 209–211.
- DAVIDSON, W.B., DOUGHTY, J.L. & BOLTON, J.L. 1941. Nitrate poisoning of livestock. Canadian Journal of Medicine and Veterinary Science, 5, 303–313.
- DE BOOM, H.P.A. & BROWN, J.M.M. 1968. Advances in ‘geeldikkop’ (Tribulosis ovis) research. X. The symptomatology and gross pathology of enzootic icterus. Journal of the South African Veterinary Medical Association, 39, 21–24.
- DE KOCK, G. 1928. A study of the reticulo-endothelial system of the sheep. 13th & 14th Reports of the Director of Veterinary Education and Research, 647–724.
- DI MENNA, MARGARET, E. & MORTIMER, P.H. 1985. Toxigenic Pithomyces and Myrothecium species in pastures. pp. 119–124. In: LACEY, J. (ed.) Trichothecenes and other mycotoxins. Proceedings of the International Mycotoxin Symposium. Sydney, Australia: John Wiley & Sons Ltd.
- DOLLAHITE, J.W. & HOLT, E.C. 1969. Nitrate poisoning. Southwest Vet, 23, 23–28.
- EPPLEY, R.M. & BAILEY, W.J. 1973. 12,13-Epoxy-9- trichothecenes as the probable mycotoxins responsible for stachybotryotoxicosis. Science, 181, 758–76.
- EPPLEY, R.M., MAZZOLA, E.P., HIGHET, R.J. & BAILEY, W.J. 1977. Structure of satratoxin H, a metabolite of Stachybotrys atra. Application of proton and carbon-13 nuclear magnetic resonance. Journal of Organic Chemistry, 42, 240–243.
- ERASMUS, J.A. 1970. Koper en seleen as etiologiese faktore by geeldikkop en enzootiese ikterus in skape in Suid-Afrika. D.V.Sc. Proefskrif, Universiteit van Pretoria, Pretoria.
- ERASMUS, J.A. 1970. Copper metabolism in the merino sheep in South Africa. Normal levels of liver copper in sheep from the Cape midlands. Journal of the South African Veterinary Medical Association, 41, 207–210.
- EVANS, W.C., EVANS, I.A., HUMPHREYS, D.J., LEWIN, B., DAVIES, W.E.J. & AXFORD, R.F.E. 1975. Induction of thiamine deficiency in sheep with lesions similar to those of cerebrocortical necrosis. Journal of Comparative Pathology, 85, 253–267.
- EVANS, E.T.R., EVANS, W.C. & ROBERTS, H.E. 1951. Studies on bracken poisoning in the horse. British Veterinary Journal, 107, 364–371.
- EVANS, I.A. & MASON, J. 1965. Carcinogenic activity of bracken. Nature, 208, 913–914.
- EVANS, I.A., THOMAS, A.J., EVANS, W.C., EDWARDS, C.N. & HOWELL, R.M. 1962. Bovine bracken poisoning. Nature, 194, 584–585.
- EVANS, W.C. 1964. Bracken poisoning of farm animals. The Veterinary Record, 76, 365–369.
- EVANS, W.C., EVANS, E.T.R. & HUGHES, L.E. 1954. Studies on bracken poisoning in cattle. I. British Veterinary Journal, 110, 295–306.
- EVANS, W.C., EVANS, E.T.R. & HUGHES, L.E. 1954. Studies on bracken poisoning in cattle. II. 1950. Bracken poisoning experiments. (Lluest Farm). British Veterinary Journal, 110, 365–380.
- EVANS, W.C., EVANS, E.T.R. & HUGHES, L.E. 1954. Studies on bracken poisoning in cattle. III. Field outbreaks of bovine bracken poisoning. British Veterinary Journal, 110, 426–442.
- EVANS, W.C., PATEL, N.C. & KOOHY, Y. 1982. Acute bracken poisoning in monogastric and ruminant animals. Proceedings of the Royal Society of Edinburgh, 81B, 29–64.
- EVANS, W.C., EVANS, I.A., THOMAS, A.J., WATKINS, J.E. & CHAMBERLAIN, A.G. 1958. Studies on bracken poisoning in cattle. IV. British Veterinary Journal, 114, 180–198.
- EVERIST, S.L. 1981. Poisonous plants of Australia. London, Sydney. Melbourne: Angus and Robertson Publishers.
- FORGACS, J. 1972. Stachybotryotoxicosis. In: KADIS, S., CIEGLER, A. & AJL, S. (eds.) Microbial toxins, Vol. III. New York: Academic Press.
- FORGACS, J., CARLL, W.T., HERRING, A.S. & HINSHAW, W.R. 1958. Toxicity of Stachybotrys atra for animals. Transactions of the New York Academy of Science, 20, 787–808.
- FRAZER, C.M. & NELSON, J. 1959. Sweet clover poisoning in newborn calves. Journal of the American Veterinary Medical Association, 135, 283–286.
- GEARY, T. 1950. Castor bean poisoning. The Veterinary Record, 62, 472–473.
- GOLDSTEIN, A., AROKOW, L. & KALMAN, S.M. 1969. Principles of drug action. New York: Harper & Row.
- GOONERATNE, S.R., HOWELL, J. & GAWTHORNE, J.M. 1981. Intravenous administration of thiomolybdate for the prevention and treatment of chronic copper poisoning in sheep. British Journal of Nutrition, 46, 457–467.
- HATCH, R.C. 1982. Poisons causing respiratory insufficiency. In: BOOTH, N.H. & McDONALD, L.E. (eds.) Veterinary Pharmacology and Therapeutics. 5th edn. Ames: Iowa State University Press.
- HARVEY, J.W. & RACKEAR, D. 1985. Experimental onion-induced haemolytic anaemia in dogs. Veterinary Pathology, 22, 387–392.
- HARVIS, B.B., PAVANASAIVAM, G. & BEAN, G.A. 1985. Mycotoxin production from Myrothecium species. In: LACEY, J. (ed.) Trichothecenes and other mycotoxins. Proceedings of the International Mycotoxin Symposium. Sydney. Australia: John Wiley & Sons Ltd.
- HELWIG, D.M. & SETCHELL, B.P. 1960. Observations on the diagnosis of nitrite poisoning in sheep. Australian Veterinary Journal, 36, 14–17.
- HIBBS, C.M. 1979. Cyanide and nitrate toxicosis of cattle. Proceedings of the 116th Annual American Veterinary Medical Association Meeting. pp. 23–26.
- HINTIKKA, EEVA-LIISA, 1977. Stachybotryotoxicosis as a veterinary problem. In: RODRICKS, J.V., HESSELTINE, C.W. & MEHLMAN, M.A. (eds.) Mycotoxins in human and animal health. Park Forest South, Illinois: Pathotox Publisher.
- HINTIKKA, EEVA-LIISA. 1978. Stachybotryotoxicosis in cattle and captive animals. In: WYLLIE, T.D. & MOREHOUSE, L.G. (eds.) Mycotoxic fungi, mycotoxins, mycotoxicoses. An encyclopedic handbook, Vol. 2. New York and Basel: Marcel Dekker, Inc.
- HINTIKKA, EEVA-LIISA. 1978. Stachybotryotoxicosis in horses. In: WYLLIE, T.D. & MOREHOUSE, L.G. (eds.) Mycotoxic fungi, mycotoxins, mycotoxicoses. An encyclopedic handbook, Vol. 2. New York and Basel: Marcel Dekker, Inc.
- HINTIKKA, EEVA-LIISA. 1978. Stachybotryotoxicosis in swine. In: WYLLIE, T.D. & MOREHOUSE, L.G. (eds.) Mycotoxic fungi, mycotoxins, mycotoxicoses. An encyclopedic handbook, Vol. 2. New York and Basel: Marcel Dekker, Inc.
- HIRONO, I., KONO, Y., TAKAHASHI, K., YAMADA, K., NIWA, H., OJIKA, M., KIGOSHI, H., HIIYAMA, K. & UOSAKI, Y. 1984. Reproduction of acute bracken poisoning in a calf with ptaquiloside, a bracken constituent. The Veterinary Record, 115, 375–378.
- HIRONO, I., ITO, M., YAGYU, S., HAGA, M. WAKAMATSU, K., KISHIKAWA, K., NISHIKAWA, O., YAMADA, K. OJIKA, M. & KIGOSHI, H. 1993. Reproduction of progressive retinal degeneration (bright-blindness) in sheep by administration of ptaquiloside contained in bracken. Journal of Veterinary Medical Science, 55, 979–983.
- HOUSEHOLDER, G.T., DOLLAHITE, J.W. & HULSE, ROSELYN. 1966. Diphenylamine for the diagnosis of nitrate intoxication. Journal of the American Veterinary Association, 148, 662–665.
- HUNTER, H.C. 1945. Cyanosis in infants caused by nitrates in well water. Journal of the American Medical Association, 129, 112–116.
- HUTCHISON, T.W.S. 1977. Onions as a cause of Heinz body anaemia and death in cattle. Canadian Veterinary Journal, 18, 358–360.
- ISHAK, K.G., WALKER, D.H., COETZER, J.A.W., GARDNER, J.J. & GORELKIN, L. 1982. Viral haemorrhagic fever with hepatic involvement: Pathologic aspects with clinical correlation. In: POPPER, H. & SCHAFFNER, F. (eds.) Progress in liver diseases, Vol . VII. New York: Grune & Stratton, Inc.
- JOHNSON, J.L., SCHNEIDER, N.R., KELLING, C.L. & DOSTER, A.R. 1983. Nitrate exposure in perinatal beef calves. American Association of Veterinary Pathology, 5th edn. Philadelphia: Lea & Febiger.
- JONES, T.C. & HUNT, R.D. 1983. Veterinary Pathology, 5th edn. Philadelphia: Lea & Febiger.
- JUBB, K.V.F., KENNEDY, P.C. & PALMER, N. 1985. Pathology of domestic animals. 3rd edn. Vol. 3. Orlando, Florida: Academic Press.
- KAHN, R.A., JOHNSON, S.A. & DeGRAFF, A.F. 1971. Effects of sodium warfarin on capillary ultrastructure. American Journal of Pathology, 65, 149–156.
- KELLERMAN, T.S., VAN DER WESTHUIZEN, G.C.A., COETZER, J.A.W., ROUX, CECILIA, MARASAS, W.F.O., MINNE, J.A., BATH, G.F. & BASSON, P.A. 1980. Photosensitivity in South Africa. II. The experimental production of the ovine hepatogenous photosensitivity disease geeldikkop (Tribulosis ovis) by the simultaneous ingestion of Tribulus terrestris plants and cultures of Pithomyces chartarum containing the mycotoxin sporidesmin. Onderstepoort Journal of Veterinary Research, 47, 231–261.
- KINGSBURY, J.M. 1964. Poisonous plants of the United States and Canada. Englewood Cliffs, New Jersey: Prentice-Hall Inc.
- KIRK, J.H. & BULGIN, M.S. 1979. Effects of feeding cull domestic onions (Allium cepa) to sheep. American Journal of Veterinary Research, 40, 397–399.
- LE ROUX, J.M.W. 1959. The histopathology of Wesselsbron disease in sheep. Onderstepoort Journal of Veterinary Research, 28, 237–243.
- LOUW, G.N., STEENKAMP, C.W.P. & STEENKAMP, E.L. 1967. Some aspects of the aetiology of enzootic icterus (‘geelsiekte’) and Tribulosis ovis (‘geeldikkop’). South African Journal of Agricultural Science, 10, 215–228.
- LOUW, G.N., STEENKAMP, C.W.P. & STEENKAMP, E.L. 1968. Chemiese samestelling van die vernaamste plantspesies in die westelike bergkaroo in die distrik Fraserburg. Tegniese Mededeling nr. 78. Departement van Landbou-tegniese Dienste, Pretoria, Suid-Afrika.
- LOUW, G.N., STEENKAMP, C.W.P. & STEENKAMP, E.L. 1968. Chemiese samestelling van die vernaamste plantspesies in die dorre, skyn-dorre, skyn-sukkulente en sentraal bo-Karoo. Tegniese mededeling nr. 79, Departement van Landbou-tegniese Dienste, Pretoria, Suid-Afrika.
- McCUNN, J., ANDREW. H. & CLOUGH, G.W. 1945. Castor-bean poisoning in horses. Veterinary Journal, 101, 136–138.
- MIROCHA, C.J. 1980. Rubratoxin, sterigmatocystin and stachybotrys mycotoxins. In: Conference on mycotoxins in animal feeds and grains related to animal health. US Department of Commerce National Technical Information Service. PB80–221773. Food and Drug Administration, Rockville, Maryland. USA.
- MOON, F.E. & RAAFAT, M.A. 1951. The experimental production of bracken poisoning in sheep. Journal of Comparative Pathology, 61, 88–100.
- MOON, F.E., RAAFAT, M.A. & McKEAND, J.M. 1953. Observations on the Vitamin C status and haematology of bracken-fed ruminants. British Veterinary Journal, 109, 321–326.
- MORGAN, K.T. 1973. Chronic copper toxicity of sheep. An ultrastructural study of spongiform leucoencephalopathy. Research in Veterinary Science, 15, 88–95.
- MURDOCK, F.R., HODGSON, A.S. & BAKER, A.S. 1972. Utilization of nitrates by dairy cows. Journal of Dairy Science, 55, 640.
- NAFTALIN, J.M. & CUSHNIE, G.H. 1954. Pathology of bracken poisoning in cattle. Journal of Comparative Pathology and Therapeutics, 64, 54–74.
- NEETHLING, L.P., BROWN, J.M.M. & DE WET, P.J. 1968. Natural occurrence of selenium in sheep blood and tissues and its possible biological effects. Journal of the South African Veterinary Medical Association, 29, 93–97.
- NEETHLING, L.P., BROWN, J.M.M & DE WET, P.J. 1968. The toxicology and metabolic fate of selenium in sheep. Journal of the South African Veterinary Medical Association, 39, 25–33.
- NESER, J.A., PHILLIPS, T., THOMSON, G.R., GAINARU, M.D. & COETZEE, T. 1986. African swine fever. I. Morphological changes at virus replication in blood platelets of pigs infected with virulent haemadsorbing and non-haemadsorbing virus isolates. Onderstepoort Journal of Veterinary Research, 53, 133–142.
- OGILVIE, D.D. 1954. ‘Chronic’ copper poisoning of sheep. The Veterinary Record, 66, 279–282.
- OLSON, O.E. & MOXON, A.L. 1942. Nitrate reduction in relation to oat hay poisoning. Journal of American Veterinary Medical Association, 100, 403–406.
- PAMUCKU, A.M. 1963. Epidemiologic studies on urinary bladder tumours in Turkish cattle. Annals of the New York Academy of Sciences, 108, 938–947.
- PAMUCKU, A.M., GOKSOY, S.K. & PRICE, J.M. 1967. Urinary bladder neoplasms induced by feeding bracken fern (Pteris aquilina) to cows. Cancer Research, 27, 917–924.
- PAMUCKU, A.M., PRICE, J.M. BRYANT, G.T. 1976. Naturally occurring and bracken fern induced urinary bladder tumours. Veterinary Pathology, 13, 110–122.
- PARKER, W.H. & McCREA, C.T. 1965. Bracken (Pteris aquilina) poisoning of sheep in the North York Moors. The Veterinary Record, 77, 861–865.
- PFISTER, J.A. 1988. Nitrate intoxication of ruminant livestock. In: JAMES, L.F., RALPHS, M.H. & NIELSEN, D.B. (eds.) The ecology and economic impact of poisonous plants on livestock production. 233–259. Boulder & London: Westview Press.
- PIENAAR, J.G. & VAN DER MERWE, J.L. DE B. 1966. Intranuclear inclusions in the hepatocytes in enzootic icterus in sheep. Journal of the South African Veterinary Medical Association, 37, 53–60.
- PIER, A.C. & McLOUGHLlN, M.E. 1985. Mycotoxic suppression of immunity. In: LACEY, J. (ed.) Trichothecenes and other mycotoxins. Proceedings of the International Mycotoxin Symposium, Sydney. Australia: John Wiley & Sons Ltd.
- POND, W.G. 1975. Guest editorial: Mineral interrelationships in nutrition. The Cornell Veterinarian, 65, 441–456.
- POPE, A.L. 1975. Mineral interrelationships in ovine nutrition. Journal of the American Veterinary Medical Association, 166, 264–268.
- PRAKASH, A.S., PEREIRA, T.N., SMITH, B.L., SHAW, G. & SEAWRIGHT, A.A. 1996. Mechanism of bracken fern carcinogenesis: Evidence for H-ras activation via initial adenine alkylation by ptaquiloside. Natural-Toxins, 4, 221–227.
- RIGGS, C.W. 1945. Nitrite poisoning from ingestion of plants high in nitrate. American Journal of Veterinary Research, 5, 194–197.
- RODERICK, L.M. 1929. The pathology of sweet clover disease in cattle. Journal of American Veterinary Medical Association, 74, 314–324.
- RODRICKS, J.V. & EPPLEY, R.M. 1974. Stachybotrys and stachybotryotoxicosis. In: PURCHASE, I.F.H. (ed.) Mycotoxins. Amsterdam: Elsevier.
- ROSS, D.B. 1966. The diagnosis, prevention and treatment of chronic copper poisoning in housed lambs. British Veterinary Journal, 122, 279–284.
- SCHIEFER, H.B. 1985. Dynamics of experimental trichothecene mycotoxicosis. pp. 433–439. In: LACEY, J. (ed.) Trichothecenes and other mycotoxins. Proceedings of the International Mycotoxin Symposium. Sydney, Australia: John Wiley & Sons, Ltd.
- SCHNEIDER, D.J., MARASAS, W.F.O., KUYS, J.C.D., KRIEK, N.P.J. & VAN SCHALKWYK, G.C. 1979. A field outbreak of suspected stachybotryotoxicosis in sheep. Journal of the South African Veterinary Association, 50, 73–81.
- SCHUMAIER, G., DE VOLT, H.M., LAFFER, N.C. & GREEK, R.D. 1963. Stachybotryotoxicosis of chicks. Poultry Science, 42, 70–74.
- SIMON, J., SUND, J.M., WRIGHT, M.J. & DOUGLAS, F.D. 1959. Prevention of noninfectious abortion in cattle by weed control and fertilization practices on Lowland pastures. Journal of the American Veterinary Medical Association, 135, 315–317.
- SIMON, J., SUND, J.M., DOUGLAS, F.D., WRIGHT, M.J. & KOWALCZYK, T. 1959. The effect of nitrate or nitrite when placed in the rumens of pregnant dairy cattle. Journal of the American Veterinary Medical Association, 135, 311–314.
- SKINNER, T.E. 1960. Sound veld management may limit the incidence of geelsiekte. Farming in South Africa, 36, 22–24.
- SMITH, R.H. 1980. Kale poisoning: The brassica anaemia factor. The Veterinary Record, 107, 12–15.
- SOLI, N.E., FROSLIE, A. & AASETH, J. 1978. The mobilization of copper in sheep by chelating agents. Acta Veterinaria Scandinavia, 19, 422–429.
- STAHMANN, M.A., HUEBNER, C.L.F. & LINK, K.P. 1941. Studies on the sweet clover disease. V. Identification and synthesis of the haemorrhagic agent. Journal of Biological Chemistry, 138, 513–527.
- STEYN, D.G. 1934. The toxicology of plants in South Africa. Central News Agency, Limited.
- STEYN, D.G. 1949. Vergiftiging van mens en dier. Pretoria: Van Schaik.
- TALJAARD, T.L. 1993. Cabbage poisoning in ruminants. Journal of the South African Veterinary Association, 64, 96–100.
- TERBLANCHE, M., MINNE, J.A. & ADELAAR, T.F. 1964. Hydrocyanic acid poisoning. A note on the HCN content of animal tissue at various stages of decomposition. Journal of the South African Veterinary Medical Association, 35, 503–506.
- TERBLANCHE, M., PIENAAR, J.G., BIGALKE, R.D. & VAHRMEIJER, J. 1967. Acacia nilotica (L) Del. subsp. kraussiana (Benth.). Brenan as a poisonous plant in South Africa. Journal of the South African Veterinary Medical Association, 38, 57–63.
- TUSTIN, R.C., ADELAAR, T.F. & MELDAL-JOHNSON, C.M. 1968. Bracken poisoning in cattle in the Natal Midlands. Journal of the South African Veterinary Medical Association, 39, 91–99.
- UENO, Y. & TSHII, K., 1985. Chemical and biological properties of trichothecenes from Fusarium sporotrichioides. In: LACEY, J. (ed.) Trichothecenes and other mycotoxins. Proceedings of the International Mycotoxin Symposium. Sydney, Australia: John Wiley & Sons, Ltd.
- VAHRMEIJER, J. 1981. Poisonous plants of southern Africa that cause stock losses. Cape Town: Tafelberg Publishers Ltd.
- VERHOEFF, J., HAJER, R. & VAN DEN INGH, T.S.G.A.M. 1985. Onion poisoning in young cattle. The Veterinary Record, 117, 497–498.
- WANNEMACHER, R.W., BUNNER, D.L., PACE, J.G., NEUFELD, H.A., BRENNECKE, L.H. & DINTERMAN, R.E. 1985. Dermal toxicity of T-2 toxin in guinea-pigs, rats and cynomolgus monkeys. In: LACEY, J. (ed.) Trichothecenes and other mycotoxins. Proceedings of the International Mycotoxin Symposium. Sydney, Australia: John Wiley & Sons, Ltd.
- WATT, J.M. & BREYER-BRANDWIJK, M.G. 1962. The medicinal and poisonous plants of southern and eastern Africa. 2nd edn. Edinburgh and London: E & S Livingstone Ltd.
- WAY, J.L. 1984. Cyanide intoxication and its mechanism of antagonism. Annual Review of Pharmacology and Toxicology, 24, 451–481.
- WIESE, W.J. & JOUBERT, J.P.J. 2001. Suspected nitrite poisoning in pigs caused by Capsella bursa-pastoris (L.) Medik. (Herderstassie, shepherd’s purse). Journal of the South African Veterinary Association, 72, 170–171.
- WIGNALL, W.N., BANKS, A.W., HACKETT, E. & IRVING, E.A. 1961. Dicoumarol poisoning of cattle and sheep in South Australia. Australian Veterinary Journal, 37, 456–459.
- WILLIAMS, W.J. 1983. Disorders of hemostasis-classification. In: WILLIAMS, W.J., BEUTLER, E., ERSLEV, A.J. & LICHTMAN, M.A. (eds.) Hematology, 3rd edn. New York: McGraw-Hill Inc.
- WILLIAMS, W.J. 1983. Disorders of hemostasis-thrombosis. In: WILLIAMS, W.J., BEUTLER, E., ERSLEV, A.J. & LICHTMAN, M.A. (eds.) Hematology, 3rd edn. New York: McGraw-Hill Inc.
- WILLIAMS, W.J. 1983. Kinetics of plasma coagulation factors. In: WILLIAMS, W.J., BEUTLER, E., ERSLEV, A.J. & LICHTMAN, M.A. (eds.) Hematology, 3rd edn. New York: McGraw-Hill Inc.