Helminth infections of wildlife
This content is distributed under the following licence: Attribution-NoDerivs CC BY-ND  View Creative Commons Licence details here
View Creative Commons Licence details here

Helminth infections of wildlife
Author: J BOOMKER
Introduction
Considering the diversity of the wildlife of Africa –the second largest continent –we really know very little about the helminths that affect them and even less about the diseases caused by helminths. Animals that die of helminthoses are quickly devoured by scavengers, especially in the larger nature reserves, and data on the cause of death and the necropsy findings are therefore usually not available. Another complicating factor is that the study of helminth biodiversity is an invasive process that is frowned upon by ecologists, game reserve managers, and animal rights ’activists. Because parasites are internal, it is not possible to remove them and leave the host alive, and artificial media for maintaining parasitic larval and adult stages are not in common usage. For many years helminths of mammals have been collected incidentally, usually during hunting expeditions and incidental postmortem examinations, and also from road kills. Until about 1940, numerous helminths new to science were described and the life cycles of several elucidated. During the Second World War and for a considerable period thereafter, the emphasis shifted to investigations of the pathogenic effects of helminths of domestic animals –and thus away from the helminths themselves. Helminths of wildlife received little attention and only a few new species or isolated and interesting cases were reported. From about 1973 onwards there was, however, a renewed interest in the helminths of wildlife. Conservation authorities made material that would otherwise have been discarded or ignored available to scientists of various disciplines, who then advised the conservation authorities of their results –to assist them with better management of existing conservation areas.
Round ’s Check list of the helminth parasites of African mammals of the orders Carnivora, Tubulidentata, Proboscidea, Hyracoidea, Artiodactyla and Perissodactyla, published in 1968, is still the only relatively complete and fully annotated check-list, but, particularly in East and South Africa, numerous additions have since been made.
Helminths of wild ruminants
Biology
Geographic distribution
In the same way that many host species have well-defined geographic distributions, so do several parasitic species. For example, eland are widespread in South Africa. Consequently, because several of the parasites infecting eland have specific geographic distributions, the composition of the parasitic fauna of these antelope in the Western Cape Province will differ from that of eland in the Karoo, which in turn will differ from that of eland in the Mpumalanga Lowveld or the Kalahari. Conversely, Trichostrongylus falculatus , which is widespread in South Africa, will infect blue wildebeest in the Mpumalanga Lowveld, springbok in the Karoo, and bontebok in the Western Cape Province. All these antelope also have a defined geographic range.
Gemsbok translocated to Langebaan Nature Reserve in the south-western part of the Western Cape Province acquired 20 times as many worms as their counterparts in the arid Etosha Game Reserve. Sheep introduced into the North West Province are exposed to Gaigeria pachyscelis, probably of blue wildebeest origin, and may die. Springbok introduced into the Bontebok National Park at Swellendam brought with them the lungworm, Bronchonema magna , which caused clinical disease in the indigenous bontebok.
Climate directly influences parasites by its effect on the free-living stages and on the vegetation, which in turn determines the distribution of the antelope hosts.
Because of climatic differences, it is important to give the regional distribution of the parasites when compiling parasite lists for the country.
The climatological regions of southern Africa are illustrated in Figure 87, and the helminths are classified as host specific, definitive, occasional or accidental parasites of their respective hosts in Tables 12 to 15. At the same time, their geographic distribution according to climate is presented.

Figure 87 The climatic regions of South Africa (redrawn from Horak (1981), and published with kind permission of the Journal of the South African Veterinary Association) .
| A | Temperate, warm and moist, occasional hot and dry bergwinds |
| D | Warm, temperate, monsoonal type of climate |
| E | Warm and moist |
| H | Warm, temperate, monsoonal type of climate, dry winter |
| K | Desert and transition zone from winter to summer rains |
| L | Subtropical, warm and muggy, except in winter |
| M | Winter rains, and a hot, dry summer |
| NT | Subtropical, semi-arid |
| SE | Warm, temperate and moist |
| SS | Semi-arid, summer rain |
| SN | Semi-arid, summer rain |
| W | Desert |
| B | Climate similar to SS and SN |
| NAM | Climate similar to SS and SN |
From the tables it can be seen that only a few species qualify as definitive parasites. Most are accidental parasites, which are acquired indirectly from other ruminants –domestic or wild. The definitive parasites, however, generally make up the bulk of the total nematode burden, with only a small contribution coming from the occasional and accidental parasites.
Some interesting observations emerge from these tables. Firstly, it appears that certain parasites are absent from some localities and are replaced by other species. A case in point is that no definitive parasites of grey duikers were recovered from these animals in Valley Bushveld. The probable reason is that this vegetation type is unfavourable for the free-living stages of the definitive parasites of these antelopes –because of the extremes in temperature and the frequently low rainfall during the summer months.
Another example is Cooperia neitzi , which is a common parasite of kudus in the Lowveld of Mpumalanga and Limpopo, but is absent in the Eastern Cape. Cooperia rotundispiculum is abundant in nyala in the moist, warm regions of KwaZulu-Natal and in kudus in the Eastern Cape Province –but is infrequently encountered in the Lowveld of Mpumalanga and in Limpopo Province.
The reason why the latter species is present in habitats with almost opposing climates is unknown, and reflects the dearth of knowledge about the distribution, epidemiology and ecological requirements of the nematodes of wildlife in general.
Table 12 Definitive and occasional helminths of impala and their distribution in the South Africa according to climate. For the distribution code, see Figure 87 .
| Helminth species | Distribution |
|---|---|
| Definitive | |
| Cooperia fuelleborni | NT, L, E |
| Cooperia hungi | NT, L, E |
| Cooperioides hamiltoni | NT, L, E |
| Cooperioides hepaticae | NT, L, E |
| Gaigeria pachyscelis | L, E |
| Haemonchus bedfordi | L, E |
| Impalaia tuberculata | NT, L, E |
| Longistrongylus sabie | NT, L |
| Oesophagostomum columbianum | NT, L |
| Pneumostrongylus calcaratus | L, E |
| Strongyloides papillosus | NT, L, E |
| Trichostrongylus colubriformis | NT, L, E |
| Moniezia expansa | NT |
| Occasional | |
| Cooperia connochaeti | L |
| Haemonchus placei | NT |
| Trichostrongylus axei | NT, E |
| Trichostrongylus falculatus | NT |
| Moniezia benedeni | L |
| Stilesia hepatica | L, E |
| Accidental | |
| Bunostomum trigonocephalum | L |
| Fasciola gigantica | NT |
Table 13 Definitive, occasional, and accidental parasites of blesbok, and their distribution in South Africa according to climate. For the distribution code, see Figure 87 .
| Helminth species | Distribution |
|---|---|
| Definitive | |
| Cooperia hungi | H |
| Cooperia yoshidai | H, E |
| Bronchonema magna | H |
| Haemonchus bedfordi | H |
| Haemonchus contortus | NT, H |
| Impalaia nudicollis | NT, H |
| Impalaia tuberculata | H |
| Longistrongylus albifrontis | H |
| Skrjabinema alata | NT, H |
| Trichostrongylus thomasi | H |
| Occasional | |
| Oesophagostomum columbianum | H |
| Trichostrongylus axei | NT, H |
| Trichostrongylus falculatus | NT, H |
| Avitellina spp. | NT, H |
| Accidental | |
| Agriostomum equidentatum | H |
Table 14 Definitive, occasional and accidental parasites of nyala, and their distribution in South Africa according to climate. For the distribution code, see Figure 87 .
| Helminth species | Distribution |
|---|---|
| Host specific | |
| Paracooperia horaki | E |
| Definitive | |
| Cooperia rotundispiculum | E, L |
| Ostertagia harrisi | E |
| Occasional | |
| Haemonchus vegliai | E, L |
| Accidental | |
| Dictyocaulus viviparus | E |
| Gaigeria pachyscelis | E |
| Impalaia tuberculata | E |
| Oesophagostomum sp. | E |
| Trichostrongylus deflexus | E |
| Trichostrongylus falculatus | E |
Table 15 Definitive, occasional and accidental parasites of kudus and their distribution in South Africa according to climate. For the distribution code, see Figure 87.
| Helminth species | Distribution |
|---|---|
| Definitive | |
| Cooperia neitzi | L, NAM |
| Cooperia rotundispiculum | K |
| Haemonchus vegliai | L, NAM, NT |
| Trichostrongylus deflexus | NT, L |
| Occasional | |
| Agriostomum gorgonis | L |
| Cooperia acutispiculum | L, NAM |
| Impalaia tuberculata | L, NAM, NT |
| Paracooperia devossi | L, NAM |
| Strongyloides papillosus | L |
| Accidental | |
| Agriostomum sp. | NAM |
| Cooperia fuelleborni | L |
| Cooperia hungi | L |
| Cooperia pectinata | NT |
| Cooperia punctata | NT |
| Cooperia yoshidai | L |
| Cooperioides hamiltoni | NAM |
| Dictyocaulus sp. | K |
| Impalaia nudicollis | NAM |
| Nematodirus helvetianus | K |
| Ostertagia ostertagi | K |
| Trichostrongylus falculatus | L, NAM |
| Trichostrongylus thomasi | NAM |
| Trichuris sp. | L |
Table 16 Worms recovered in surveys conducted during the 1980s ranked here in ascending sequence, according to the number of host species infected. Figures in parentheses indicate the number of animals that were infected.
| Mean burdens of infected animals | ||||||||
|---|---|---|---|---|---|---|---|---|
| Helminth species | Blue duiker (n=4) | Red duiker (n=27) | Bushbuck (n=29) | Grey duiker (n=45) | Nyala (n=79) | Kudu (n=151) | Grey rhebuck (n=47) | Suni (n=4) |
| one host species infected | ||||||||
| Fasciola hepatica | 0 | 0 | 0 | 0 | 0 | 0 | 2 (1) | 0 |
| Echinococcus sp. larvae | 0 | 0 | 0 | 0 | 0 | 1 (1) | 0 | 0 |
| Agriostomum gorgonis | 0 | 0 | 0 | 0 | 0 | 52 (42) | 0 | 0 |
| Cooperia fuelleborni | 0 | 0 | 0 | 0 | 0 | 89 (4) | 0 | 0 |
| Cooperia punctata | 0 | 0 | 0 | 0 | 0 | 275 (1) | 0 | 0 |
| Cooperioides hamiltoni | 0 | 0 | 0 | 0 | 0 | 73 (4) | 0 | 0 |
| Haemonchus horaki | 0 | 0 | 0 | 0 | 0 | 0) | 202 (19) | 0 |
| Hyostrongylus rubidus | 0 | 68 (20) | 0 | 0 | 0 | 0 | 0 | 0 |
| Impalaia nudicollis | 0 | 0 | 0 | 0 | 0 | 207 (3) | 0 | 0 |
| Longistrongylus curvispiculum | 0 | 0 | 0 | 0 | 0 | 0 | 25 (15) | 0 |
| Longistrongylus namaquensis | 0 | 0 | 0 | 0 | 0 | 0 | 32 (11) | 0 |
| Longistrongylus sabie | 0 | 0 | 41 (2) | 0 | 0 | 0 | 0 | 0 |
| Megacooperia woodfordi | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 22 (3) |
| Nematodirus abnormalis | 0 | 0 | 60 (1) | 0 | 0 | 0 | 0 | 0 |
| Nematodirus helvetianus | 0 | 0 | 0 | 0 | 0 | 275 (3) | 0 | 0 |
| Onchocerca spp. | 0 | 0 | 0 | 0 | 0 | 3 (9) | 0 | 0 |
| Ostertagia hamata | 0 | 0 | 0 | 0 | 0 | 0 | 303 (41) | 0 |
| Ostertagia ostertagi | 0 | 0 | 0 | 0 | 0 | 63 (2) | 0 | 0 |
| Paracooperia horaki | 0 | 0 | 0 | 0 | 95 (35) | 0 | 0 | 0 |
| Paracooperioides peleae | 0 | 0 | 0 | 0 | 0 | 0 | 196 (37) | 0 |
| Pneumostrongylus calcaratus | 0 | 0 | 0 | 1 (1) | 0 | 0 | 0 | 0 |
| Setaria africana | 0 | 0 | 0 | 2 (8) | 0 | 0 | 0 | 0 |
| Skrjabinodera kueltzii | 0 | 0 | 1 (1) | 0 | 0 | 0 | 0 | 0 |
| Trichostrongylus colubriformis | 0 | 0 | 2 (3) | 0 | 0 | 0 | 0 | 0 |
| Trichostrongylus colubriformis | 0 | 0 | 2 (3) | 0 | 0 | 0 | 0 | 0 |
| Two host species infected | ||||||||
| Schistosoma mattheei | 0 | 0 | 0 | 0 | 5 (1) | 21 (18) | 0 | 0 |
| Avitellina spp. | 0 | 0 | 4 (3) | 0 | 0 | 9 (2) | 0 | 0 |
| Moniezia benedeni | 0 | 3 (6) | 0 | 0 | 0 | 2 (13) | 0 | 0 |
| Moniezia expansa | 0 | 0 | 11 (2) | 0 | 0 | # (1) | 0 | 0 |
| Stilesia hepatica | 0 | # (4) | # (2) | 0 | 0 | 0 | 0 | 0 |
| Taenia hydatigena larvae | 5 (1) | 0 | 1 (5) | 0 | 0 | 0 | 0 | 0 |
| Cooperia acutispiculum | 0 | 0 | 66 (2) | 0 | 0 | 348 (87) | 0 | 0 |
| Cooperia pectinata | 0 | 0 | 328 (1) | 0 | 0 | 78 (1) | 0 | 0 |
| Cooperia yoshidai | 0 | 204 (2) | 0 | 0 | 0 | 118 (1) | 0 | 0 |
| Gaigeria pachyscelis | 0 | 0 | 0 | 25 (2) | 25 (2) | 0 | 0 | 0 |
| Longistrongylus schrenki | 0 | 13 (1) | 0 | 0 | 0 | 0 | 6 (2) | 0 |
| Nematodirus spathiger | 0 | 0 | 148 (5) | 0 | 0 | 0 | 117 (17) | 0 |
| Paracooperia devossi | 0 | 0 | 0 | 150 (19) | 0 | 91 (5) | 0 | 0 |
| Setaria scalprum | 0 | 2 (3) | 3 (4) | 0 | 0 | 0 | 0 | 0 |
| Teladorsagia circumcincta | 0 | 36 (2) | 12 (2) | 0 | 0 | 0 | 0 | 0 |
| Trichostrongylus angistris | 0 | 11 (3) | 704 (22 | 0 | 0 | 0 | 0 | 0 |
| Trichostrongylus rugatus | 0 | 1 (1) | 0 | 0 | 0 | 0 | 3 (1) | 0 |
| Three host species infected | ||||||||
| Thysaniezia spp. | 0 | 0 | 2 (2) | 0 | # (1) | # (1) | 0 | 0 |
| Cooperia hungi | 0 | 0 | 254 (5) | 0 | 0 | 193 (8) | 0 | 1 (1) |
| Cooperia neitzi | 0 | 0 | 121 (2) | 58 (3) | 0 | 1 230 (100) | 0 | 0 |
| Elaeophora sagitta | 0 | 0 | 0 | 3 (3) | 6 (6) | 15 (70) | 0 | 0 |
| Dictyocaulus viviparus | 0 | 7 (4) | 0 | 12 (3) | 3 (4) | 0 | 0 | 0 |
| Gongylonema spp. | 1 (1) | 0 | 0 | 4 (3) | 9 (3) | 0 | 0 | 0 |
| Haemonchus contortus spp. | 0 | 27 (11) | 13 (4) | 0 | 0 | 0 | 68 (4) | 0 |
| Strongyloides papillosus spp. | 0 | 11 (2) | 0 | 0 | 0 | 742 (6) | 0 | 26 (1) |
| Oesophagostomum spp. | 0 | 0 | 4 (3) | 10 (1) | 1 (1) | 0 | 0 | 0 |
| Ostertagia harrisi | 0 | 105 (17) | 0 | 233 (21) | 463 (72) | 0 | 0 | 0 |
| Setaria cornuta | 0 | 2 (2) | 3 (8) | 0 | 0 | 0 | 0 | 1 (1) |
| Trichostrongylus anomalus | 1 (1) | 281 (16) | 0 | 0 | 0 | 0 | 0 | 44 (4) |
| Trichostrongylus axei | 410 (1) | 1 060 (1) | 319 (24) | 0 | 0 | 0 | 0 | 0 |
| Trichostrongylus thomasi | 0 | 3 (1) | 41 (3) | 0 | 0 | 38 (2) | 0 | 0 |
| Trichuris spp. | 0 | 38 (5) | 18 (6) | 0 | 0 | 25 (5) | 0 | 0 |
| Four host species infected | ||||||||
| Taenia spp. larvae | 0 | 0 | 3 (4) | 2 (3) | 1 (1) | 2 (11) | 0 | 0 |
| Impalaia tuberculata | 0 | 13 (6) | 247 (15) | 0 | 50 (1) | 194 (30) | 0 | 0 |
| Five host species infected | ||||||||
| Calicophoron spp. | 0 | 258 (8) | 69 (9) | 14 (1) | 306 (28) | 96 (32) | 0 | 0 |
| Haemonchus vegliai | 0 | 0 | 14 (7) | 105 (10) | 44 (14) | 252 (107) | 0 | 19 (4) |
| Setaria spp. | 1 (2) | 2 (3) | 3 (11) | 0 | 3 (16) | 1 (3) | 0 | 0 |
| six host species infected | ||||||||
| Trichostrongylus deflexus | 0 | 0 | 2 (1) | 368 (2) | 74 (6) | 631 (45) | 740 (1) | 36 (1) |
| Trichostrongylus falculatus | 1 (1) | 0 | 22 (2) | 222 (3) | 20 (5) | 123 (7) | 160 (11) | 0 |
| seven host species infected | ||||||||
| Cooperia rotundispiculum | 16 (1) | 842 (25) | 51 (9) | 795 (4) | 422 (36) | 664 (13) | 0 | 1 (1) |
Seasonal abundance
Many internal and external parasites display distinct periods of seasonal abundance. It is thus probable that animals of a particular species examined during summer will not only harbour different levels of infection to those examined in winter, but the actual species composition of the parasites may also differ. As with parasites of domestic stock, the patterns of seasonal abundance are brought about by the parasites employing survival strategies so that their most sensitive stages of development –usually the free-living stages –are protected against regularly occurring unfavourable environmental conditions. Therefore, hypobiosis takes place in one or more of the stages of a parasite ’s life cycle, and life cycles last approximately one year –ensuring that favourable climatic conditions for the parasites are encountered at some time in the future (Horak, 1978).
Seasonal abundance may also be influenced by competition for a limited resource. Thus peak adult burdens of Haemonchus bedfordi and Trichostrongylus thomasi –which both occur in the abomasum of blue wildebeest –are staggered. Trichostrongylus thomasi reaches peak abundance one or two months after the larger nematode H. bedfordi has passed its peak. Peak burdens of the tapeworm Moniezia benedeni , which is a large species, have been encountered in the small intestines of blue wildebeest calves aged six to eight months, while Avitellina sp., which is a smaller tapeworm, only peaked once the calves had reached ten months of age (Horak, De Vos &Brown, 1983).
Stress
Winter
Most wild herbivores suffer stress during winter because of the paucity of quality grazing or browse. This type of stress is generally accompanied by increased parasitic burdens. However, even though the burdens are increased, the parasites themselves are simultaneously employing strategies to escape the unfavourable external winter climate (Horak, 1978). Thus, herbivores may harbour large parasite burdens during winter, but many of these parasites will be in a state of hypobiosis and thus pose little threat to the health of the host. Many Haemonchus spp., Longistrongylus spp. and Cooperia spp. will be arrested in the fourth larval stage.
Drought
The most severe effects of drought on herbivores are generally apparent in spring. By that time the animals have been through a previous spring, summer and autumn with little or poor quality grazing and browse –followed by a winter in which very little feed of any kind was available. Nutritional stress is thus severe and their immune status compromised. At the same time many of the nematodes which have overwintered in the host as larvae, in an arrested stage of development, develop to become adults. Animals may concentrate around green patches or waterholes where contamination levels with parasites become high. If these animals die they are generally cachectic and harbour large burdens of both helminth and arthropod parasites.
Prolonged droughts, lasting two or more years, can have a number of unexpected results. The vegetation and surfacesoil microhabitat in which the free-living stages of the parasitic nematodes develop and survive, may be destroyed –with a concomitant reduction in free-living parasite levels. This is reflected in reduced parasite loads of host animals. Many of the hosts may have died because of the drought or migrated to a more favourable habitat. This in turn leads to a reduction in contamination by host animals of the original habitat. Animals may thus harbour reduced parasite loads for several years, until the microhabitat recovers and host numbers increase again.
Gender
Peri-parturient relaxation of resistance (PPRR) in female antelope could be responsible for an increase in the number of helminths in these animals and in the previous year ’s yearlings (Horak, 1978; Horak, McIvor &Greeff, 2001). Some helminths, such as Strongyloides spp., are transmitted through the milk.
During the rutting season, male animals continuously defend their territories and can be severely stressed. This is reflected in increased parasite burdens.
Disease, injury or age
Diseased, injured or aged animals are all stressed animals with compromised immune systems –and therefore usually harbour large parasite burdens. In addition, their mobility may be impaired and consequently they contaminate their own immediate environment, from which they will then in turn become reinfected.
General
Stressed animals will have larger nematode burdens than normal animals and a greater proportion of female nematodes is likely to mature and lay eggs.
Epidemiology
Most wild animal species are distributed in fairly well-defined geographic regions. Within these regions particular species will have preferred habitats. Animal species, geographic distribution and habitat preference, will each contribute towards determining the species composition of parasite burdens, as well as their numerical magnitude in a given host. Normal, healthy wild animals in large ecosystems frequently harbour nematode burdens exceeding several thousand. Many of these nematodes are in an immature stage of development and cause few pathogenic effects. It is generally only when adult nematodes exceed several hundred or, in some cases several thousand, that problems can be expected.
Dispersion of Parasites
According to Petney, Van Ark and Spickett (1990), parasites are generally overdispersed within host populations ( see Figure 88 ). This means that most hosts have only a few parasites, but some have many. This implies that a few hosts harbour a high proportion of the total population of a particular parasite within a specific environment.
The reasons for overdispersion are:
- Free-living parasites are not randomly dispersed within the host ’s environment (dung pats contain large numbers of worm eggs).
- Variation within habitat (thickets, stream, dam).
- Host ’s feeding preference (grazer, browser, mixed feeder).
- The presence of an intermediate host in the life cycle, and the number of intermediate hosts present.
- Variation in the host ’s ability to reduce or limit parasites by immunity or other means.
- Some of the host ’s behavioural traits (communal dung heaps, spreading dung, pellets or pats).
The role of parasites
In large ecosystems free from human interference, parasites and predators drive an adaptive selection process. Young animals –usually until the age of 12 to 18 months –are often subject to large parasite infections (Horak, 1978; Horak et al., 1983). Weaker individuals and those that do not develop an effective immune response often succumb, and are caught by predators before they can contribute to the gene pool. Diseased, injured, stressed or maladapted young or older animals and aged animals frequently have compromised immune systems. They become heavily infected with parasites –resulting in a further deterioration in their condition and their rapid removal from the environment by predators.
Human intervention
Human activities have interfered with the host-parasite balance. Not only have humans translocated wild animals to regions in which they did not originally occur, but they have eliminated predators –as they have perceived these to be competition for a limited resource. The humans have themselves then failed to assume the role of the predators, or even worse have selectively taken out the fittestlooking individuals for consumption, sale or as trophies.
Translocated animals often suffer severe stress and may never adapt to the new habitat or to the resident parasites, which are foreign to them. They thus become a source of infection, not only for themselves, but for the wildlife endemic to the region. Because of the cost involved in the translocated wild animals ’purchase and transportation, it is unlikely that they will be purposely exposed to predators, nor will their owners destroy them if they become heavily parasitised –and consequently they persist as reservoirs of infection.
| Under-dispersed |
| Random |
| Over-dispersed | ||||||
| xx | xx | xxx | xxxx | Xx |
| xxxxx |
|
| ||
| xx | xx | xx |
| Xxx | X | x |
|
| ||
| xx | xx | Xx | xx | Xxx | xx |
| X |
| ||
| xxx | xx | X | xxx | X | xxx |
| xxxxx | xxxx | ||
s ²/x=Variance to mean ratio
Figure 88 Parasite dispersion pattern theory
Humans have also introduced domestic livestock into wildlife regions and reintroduced wildlife into regions now used for stock farming. This has led to the introduction of parasites foreign to either one of these host groups and to crossinfection taking place. In some cases the parasites have adapted to the new host species with little visible reaction, while in others morbidity or mortality may be high.
The erection of fences has not only interfered with wildlife movement but also with their migration, and has also placed a finite size on the area available. Movement, and more particularly migration, allows animals to leave areas of high parasite contamination, while containment ensures their confinement –often at high stocking densities –in highly contaminated localities. In the latter type of environment, cross-infection with parasites between host species is very likely.
Host specificity
Host specificity implies the unique occurrence of a helminth species in a particular host species, and studies have shown that host specificity is not present to any great extent amongst the ruminants (antelopes). Helminths are often shared amongst the different species occurring in a geographic region. Certain helminths occurring in a subfamily of antelopes are largely limited to that subfamily, and are rarely found in other hosts. When the wild ruminants share pastures with domestic stock, both groups often acquire each other ’s worms. However, there are a number of helminths that occur only in a particular host species, while others are generalists and occur in hares, warthogs, grazers and browsers, mixed-feeding wild ruminants and even in zebra (e.g. Trichostrongylus thomasi) . To determine host specificity, large numbers of animals from various localities must be examined, and both the immature and adult stages of the parasite must be recovered, counted and identified (Horak 1981).
The various helminths that have been recovered from sheep, cattle, impala and blesbok in South Africa have been listed by Horak (1980) as definitive, occasional or accidental parasites of their respective hosts. He suggests that definitive parasites are present in a large percentage of a host population, often occur in large numbers, and can reproduce and survive for long periods in these hosts. Occasional parasites are present in varying numbers in some of the hosts only. They may be capable of reproduction, but survive only for a limited period. Accidental parasites are present in small numbers in a small percentage of hosts. They may not be able to develop into adults and, even if they do, they may not be able to reproduce. Their survival period in the host may also be short.
The worms recovered in the surveys conducted during the 1980s (Boomker, 1990) are ranked in Table 16 in ascending sequence according to the number of host species infected. None of the worms listed occurred in all eight host species. Cooperia rotundispiculum occurred in seven host species, and T. falculatus and Trichostrongylus deflexus each occurred in six host species. This situation could be due to variable host specificity or whether the worms are definitive, occasional or accidental parasites of their respective hosts.
Host specificity of helminths within groups of antelope species that have similar ecological requirements and habits seems to be more indicative of the adaptation of a particular worm species to an environment, and thus indirectly to specific hosts. This adaptation is an ongoing evolutionary process, which –when host species become geographically isolated and the gene flow within the helminth species is reduced or cut off –eventually leads to the differentiation of new helminth species.
Table 17 Some helminths of domestic ruminants and their counterparts in wild ruminants.
| Helminths of domestic ruminants | Counterpart in wild ruminants |
|---|---|
| Nematodes (Roundworms) | |
| Bunostomum | Bunostomum |
| Agriostomum gorgonis | |
| Gaigeria pachyscelis | Gaigeria pachyscelis |
| Chabertia ovina | |
| Cooperia mcmasteri | |
| Cooperia pectinata | Cooperia pectinata |
| Cooperia punctata | Cooperia punctate |
| Cooperia spatulata | |
| Cooperia oncophora | |
| Cooperia acutispiculum | |
| Cooperia hungi | |
| Cooperia yoshidai | |
| Impalaia tuberculata | |
| Paracooperia horaki | |
| Paracooperioides peleae | |
| Dictyocaulus filarial | |
| Dictyocaulus viviparus | Dictyocaulus viviparus |
| Dictyocaulus africana | |
| Bronchonema magna | |
| Pneumostrongylus calcaratus | |
| Protostrongylus capensis | |
| Muellerius capensis | |
| Elaeophora sagitta | Elaeophora sagitta |
| Elaeophora poeli | |
| Gongylonema spp. | Gongylonema spp. |
| Haemonchus contortus | Haemonchus contortus |
| Haemonchus bedfordi | |
| Haemonchus placei | |
| Ostertagia ostertagi | Ostertagia ostertagi |
| Ostertagia hamata | |
| Ostertagia harrisi | |
| Teladorsagia circumcincta | Teladorsagia circumcincta |
| Longistrongylus albifrontis | |
| Longistrongylus schrenki | |
| Longistrongylus thalae | |
| Nematodirus helvetianus | Nematodirus helvetianus |
| Nematodirus spathiger | Nematodirus spathiger |
| Oesophagostomum columbianum | Oesophagostomum columbianum |
| Oesophagostomum radiatum | |
| Oesophagostomum venulosum | |
| Oesophagostomum africanum | |
| Oesophagostomum walkeri | |
| Setaria labiatopapillosa | Setaria labiatopapillosa |
| Setaria africana | |
| Setaria bicoronata | |
| Setaria boulengeri | |
| Strongyloides papillosus | Strongyloides papillosus |
| Trichostrongylus axei | Trichostrongylus axei |
| Trichostrongylus thomasi | |
| Trichostrongylus colubriformis | Trichostrongylus colubriformis |
| Trichostrongylus deflexus | |
| Trichostrongylus rugatus | Trichostrongylus rugatus |
| Trichostrongylus angistris | |
| Trichostrongylus anomalus | |
| Trichostrongylus falculatus | Trichostrongylus falculatus |
| Trichinella spiralis | Trichinella nelsoni |
| Cestodes (Tapeworms) | |
| Avitellina spp. | Avitellina spp. |
| Echinococcus sp. larvae spp. | Echinococcus sp. larvae spp. |
| Moniezia benedeni | Moniezia benedeni |
| Moniezia expansa | Moniezia expansa |
| Moniezia pallida | |
| Stilesia hepatica | Stilesia hepatica |
| Taenia hydatigena larvae | Taenia hydatigena larvae |
| Taenia crocutae larvae | |
| Taenia hyaenae larvae | |
| Thysaniezia sp. | Thysaniezia sp. |
| Taenia regis larvae | |
| Trematodes (Flukes) | |
| Fasciola hepatica | Fasciola hepatica |
| Fasciola gigantica | Fasciola gigantica |
| Fasciola jacksoni | |
| Fasciola tragelaphi | |
| Calicophoron microbothrium | Calicophoron microbothrium |
| Calicophoron calicophorum | Calicophoron calicophorum |
| Calicophoron bothriophoron | |
| Carmyerius mancupatus | |
| Carmyerius spatiosus | |
| Cotylophoron cotylophorum | Cotylophoron cotylophorum |
| Cotylophoron indicum | |
| Cotylophoron jacksoni | |
| Schistosoma mattheei | Schistosoma mattheei |
| Schistosoma margrebowiei | |
| Schistosoma leiperi | |
From the foregoing discussion it follows that host specificity in the broad sense is largely absent in browsers. The term ‘host specificity ’should therefore be disregarded in favour of the classification suggested by Horak (1980). This classification should be slightly modified by adding ‘host specific ’, and the categories should thus be host specific, definitive, occasional, and accidental. This modification has become necessary to accommodate parasites like Megacooperia woodfordi, Paracooperioides peleae and Paracooperia horaki from suni, grey rhebuck and nyala respectively, that have been recorded from these hosts only (which makes them host specific), and in sufficient numbers to qualify them as being definitive parasites.
Only nematodes with a direct life cycle and that enter the host per os can be classified this way. The utilisation of an intermediate host in the life cycle almost automatically classifies a parasite as an accidental antelope parasitic, since many of the intermediate hosts are accidentally consumed. For example, dung beetles or cockroaches –the intermediate hosts of the spirurid nematodes –are not a ‘normal ’part of the final host ’s diet and are only consumed when they are unable to move away from the final host when it is feeding.
Similarly, biting flies –the vectors of some filarid nematodes –do not feed exclusively on the final hosts of a particular helminth species. The intermediate hosts are not necessarily present on grazing or browse, and are able to leave at will. The infective larvae of nematodes with a direct life cycle depend on vegetation for protection and survival. The larvae are attracted to diffuse light and actually migrate onto the vegetation –provided there is sufficient moisture and temperatures exceed 15 °C –and then await the arrival of a host. The presence of helminths with an indirect life cycle is therefore merely an indication of the abundance of the intermediate host.
The presence of trematodes in a final host not only indicates that host ’s dependence on water, but also its habitat preference. Trematodes should also be more abundant in those antelope that drink water regularly, than in those that do not. Similarly, trematodes should also be more abundant in antelope that prefer a moist habitat, such as sitatunga, than in those that prefer an arid habitat, like gemsbok. This possibly explains the relative abundance of Schistosoma mattheei in kudu in the Kruger National Park (KNP), which regularly drink water, and the paucity of the species in the other browsing antelope examined (Boomker, Du Plessis &Boomker, 1983; Boomker, Horak &De Vos, 1989; Boomker, Horak &Flamand, 1991). The presence of Calicophoron spp. in many of the antelope examined during the numerous surveys conducted in many regions in the country, is an indication that the hosts regularly drink water and also consume the usually green vegetation on which the metacercariae of the trematode may be found, around the watering place.
The lists of host specific, definitive, occasional and accidental parasites are not complete and do not adequately reflect the distribution of the helminths within climatic regions. A possible exception is kudu in the KNP, where the helminths of 100 animals were counted and identified. For the scarcer antelope the data will probably remain incomplete for a long time to come. As a case in point, because of their conservation status in the Limpopo, Gauteng and Mpumalanga Provinces, it is almost impossible to obtain red and blue duikers for worm surveys. It is therefore more than likely that when more material from the different regions becomes available, the status of some of these helminths will change.
General Comments
The effect of feeding habits on nematode burdens
Two factors should be borne in mind when attempting to relate epidemiological trends in the parasites of antelope to those which are already known for domestic stock. Firstly, with the exception of goats in certain habitats and cattle which may also occasionally browse, domestic ruminants are grazers. Hence, one cannot really compare the epidemiology of the worms of domestic grazers with that of the helminths of wild browsers, since the feeding habits of their hosts are entirely different. The ground-cover of most of the nature reserves consists mostly of grass, interspersed with herbs and forbs, and, because of its physical structure and relative abundance, more infective larvae will occur on the grass than on the forbs. Because grass has a lower nutritional value than browse, grazers need to eat more, which in turn results in grazers acquiring more worms than browsers –as is evident from previous studies on grazing antelope. Secondly, most of the epidemiological work on the helminths of wild ruminants has been done on the grazing antelope species. The epidemiological trends of their parasites can probably be compared with those of domestic animals, but not with those of the browsing antelope. Little is known about the life cycles and the ecology of the freeliving stages of many of the nematodes that infect wild ruminants.
Many of these helminth species do not occur in domestic ruminants and one cannot assume that the free-living stages of these worms behave in the same way, as representatives of the same genus, in domestic ruminants. Furthermore, because the worms of antelope have evolved along with their hosts and therefore in the same habitat, there may be small but significant adaptations in their ability to survive, and also in the longevity of their free-living stages.
The feeding habits of the browsing antelope vary, although the diet of each consists of more than 75% browse. Duiker are small antelope that browse at a height of less than 1 metre and they seldom eat grass. Bushbuck and female nyala browse up to a height of approximately 1.5 metres, while male nyala and kudu are large antelopes that browse up to a height of 2.5 metres. Analysis of the rumen content of 100 nyala has shown that they often graze, as well as browse.
Novellie (1983) indicated that kudu utilise different types of browse during different times of the year. They feed on forbs at ground level from summer to spring (December to September), a period which includes the rainy season, and when infective larvae are usually present on the vegetation. Despite this, the largest individual helminth burden recorded in a kudu consisted of only 8 040 worms and that in a nyala of 13 600 worms –which suggests that other factors may also play a role in limiting helminth burdens. Although such detailed food preference and helminthological studies have not been made for the other browsers, mainly because of the limited number of antelope that were available, one could assume that the pattern would be similar. Those browsers that consume grass as well as browse, such as nyalas, may be expected to have larger worm burdens than those that browse exclusively (blue and red duikers and suni), but, nevertheless, the individual burdens remain well below those that are considered pathogenic in sheep and cattle.
The influence of the difference in feeding behaviour on worm burdens is well illustrated by the results of the survey of the helminths of grey duiker, grysbok, and Angora and Boer goats in Valley Bushveld. The mean total helminth burden of the grey duikers was less than those of the grysbok or Angora goats –both mixed feeders –and considerably less than the burdens of Boer goats, which are predominantly grazers (Boomker, Horak &McIvor, 1989).
The effect of antelope behaviour on nematode burdens
With the exception of grey rhebuck and possibly nyala and kudus during certain times of the year, the browsers are solitary animals that, at most, occur as small family groups. They would thus not contaminate their territories with worm eggs to any significant degree, which would in turn limit the size of the infection. Steenbok, which occur singly or in pairs, and impala which sometimes occur in large herds, are both mixed feeders, and, on average, harbour more species and larger burdens than browsers from a region with a similar climate. These antelope species contaminate their environment to a much larger degree, and impala, particularly because of their gregarious habits, may acquire heavy burdens.
A factor which may further limit the magnitude of the worm burden is that all the browsing antelopes produce faecal pellets, which, unlike cattle ’s dung pats, are not good reservoirs for infective larvae (Reinecke, 1960). Despite the protection afforded by the vegetation, faecal pellets tend to dry out rapidly –especially in the more arid regions –and thus the antelope will not accumulate significant helminth burdens.
Red duiker make use of communal dung heaps, which confine the freeliving infective larvae to a particular area. Despite their visits to these heaps, it is unlikely that they acquire large burdens, since the composting effect will kill many nematode eggs and free-living stages. During the wet season, the remaining infective larvae will move laterally and horizontally from the dung heap, and natural curiosity or hunger could entice a red duiker –particularly one that has recently arrived in a territory –to examine the dung heaps and feed on vegetation nearby, so becoming infected. It is also entirely possible that the duiker may simply prefer not to browse in the immediate vicinity of these heaps.
Droppings left by browsers that utilise the same type of habitat may also be a source of infection for other browser species. It is probably for this reason that the helminth fauna of these antelopes is very similar.
The effects of geography on nematode burdens
Most antelope that have been culled and processed for worm recovery were from the eastern part of the country, where, during the years of the surveys, the climate was generally favourable for the survival of the free-living stages. The total and mean nematode burdens of those browsing antelope species that were examined in several localities, apparently differed –but because of the few animals available, statistical comparisons could not be made. In the more arid regions, free-living infective larvae are exposed to high day-time temperatures and desiccation. These environmental factors and the feeding habits of the hosts, in all probability account for the small nematode burdens of kudu and grey duiker in the Valley Bushveld of the Eastern Cape Province, and of kudu in Namibia.
The effect of overpopulation on nematode burdens
Overpopulation is generally considered as one of the major factors in the dissemination of helminths between animals, and if overpopulation occurs on permanent pastures –which is often the case –the situation is further aggravated (Dunn, 1968; Urquhart, Armour, Duncan, Dunn &Jennings, 1987). Although overpopulation with antelopes seldom occurs in well-managed game reserves, their numbers do fluctuate. When environmental conditions are optimal, abnormally large populations may sometimes be present. This was the case with kudu in the main study area of the KNP (De Vos, V., personal communication, 1980) and nyala in the northern KwaZulu- Natal game reserves, where as many as 1 500 nyala were culled annually (Flamand, J.R.B., personal communication, 1983). The results of the helminth surveys of the above-mentioned antelopes may be biased in that –because of the large host populations –the individual and mean total helminth burdens are larger than would be the case with ‘normal ’populations.
Helminth burdens can be indicative of increasing host populations. Bontebok in the old Bontebok National Park (BNP), near Bredasdorp in the Western Cape Province, had large helminth burdens, and this –together with the unsuitable area in which the Park was situated –led to their translocation to the current BNP near Swellendam (Barnard &Van Der Walt, 1961). The breeding herd that consisted of 61 animals at the time has increased to the current estimated 15 000 animals now present in several localities in the country (Bain, 2003). Reedbuck from Charters Creek in northern KwaZulu-Natal also had large helminth burdens (Boomker, Horak, Flamand &Keep, 1989), and the helminth burdens of grey rhebuck in the BNP appear to be increasing –as is evident from the increase in the mean helminth burden.
Because of the ecological disaster created by tropical storm Domoina in 1987, which severely depleted the nyala population in the Umfolozi Game Reserve –now part of the Hluhluwe-Imfolozi Game Reserve –a follow-up survey could not be done. A follow-up of reedbuck from Charters Creek, after some culling had taken place, indicated that the individual and mean total helminth burdens were considerably reduced (Boomker et al., 1989).
Twenty-seven helminth species that are transmissible from impala to other antelope species –as well as to cattle, sheep and goats –are listed by Anderson (1983), and those helminths transmissible from blesbok to cattle, sheep and goats are listed by Horak (1979). No such records are available for the helminths of browsing antelope. With intensification or greater population densities, as is often the case with game farming, one would expect a similar situation, as outlined above, to develop. The parasites of ruminants are not notably host specific (Dunn, 1968) and browsing antelopes could act as reservoir hosts for the parasites of domestic ruminants and vice versa. In this manner, burdens that could cause clinical disease may be acquired by either host. Attributions of serious helminthoses have been recorded in North America, where heavy losses of deer due to infections with Ostertagia spp., Trichostrongylus spp. and Haemonchus spp. were encountered (Longhurst, Leopold &Dasman, 1952, cited by Dunn, 1968). It should be pointed out that deer are predominantly grazers. Several instances of contact between browsers and domestic ruminants have been reported, and in some of these the nematode burdens of the browsers were larger and more helminth species were recovered than in browsers that had no contact with domestic stock (Boomker, 1990). As stated previously, however, the individual helminth burdens remained well below what is considered pathogenic for domestic ruminants, and the helminth burdens in the browsing antelope examined showed no visible deleterious effects.
The effect of nematode burdens on the host
Little information on the numbers of nematodes necessary to produce clinical disease in antelope is available –and none as far as the browsing antelope are concerned. Anderson (1983) in KwaZulu-Natal and Meeser (1952) in the KNP and adjoining Sabie Sand Game Reserve, found that impala with a total worm burden of 30 000 to 50 000 manifested clinical signs of helminthosis, including submandibular oedema. None of the impala examined during 1980 harboured burdens sufficiently high to produce clinical signs (Horak &Boomker, unpublished data, 1983).
However, during winter when browser grazing is not freely available, and what is available is of poor nutritional value, animals may suffer from helminthosis –even with small helminth burdens (Dunn, 1968). Although the effects may not be clinically evident, additional drain on protein and iron reserves may lower the animal ’s resistance, so causing it to succumb to larger helminth infections or infectious diseases.
Conclusion
The data accumulated during all the surveys clearly indicate that –in spite of possibly faulty techniques and probable bias due to unnatural situations –the nematode burdens harboured by antelope in relatively undisturbed nature reserves are numerically and pathogenically insignificant and do not constitute any danger to the respective antelope, particularly when the helminth species diversity is also taken into account. The cestodes and trematodes they harbour are even less significant, and although adult worms were recovered from many antelope that were examined, they were present in small numbers –and then only in young animals. A hydatid cyst of Echinococcus spp., which can be acquired by humans (a zoonosis), was recovered from only one kudu of 96 examined, and kudu can therefore not be considered as particularly dangerous or important from the zoonotic point of view.
Selected helminthoses of antelope
Introduction
In many regions of South Africa sheep, goats or cattle graze the same pastures as various antelope species. Many of the helminths recovered from the antelope are those usually encountered in domestic ruminants –especially sheep and cattle –while other helminths of cattle, sheep and antelope are more host specific and are rarely encountered in other species. Horak (1979) was able to artificially infect sheep with Haemonchus contortus, Trichostrongylus axei, Trichostrongylus falculatus and Impalaia nudicollis cultured from the faeces of blesbok (Damaliscus pygarthus dorcas) , which were naturally infected with these worms. Haemonchus placei, Longistrongylus sabie, Trichostrongylus colubriformis, Trichostrongylus falculatus, Impalaia tuberculata and Cooperia hungi likewise became established in sheep, goats and cattle infected with larvae cultured from the faeces of impala (Aepyceros melampus) . However, H. contortus, T. axei, T. colubriformis and T. falculatus are known to occur in sheep, and H. placei and T. axei in cattle, and it is impossible to determine what role cross-infection plays in maintaining the helminth populations in all four of the host species.
Lungs and trachea
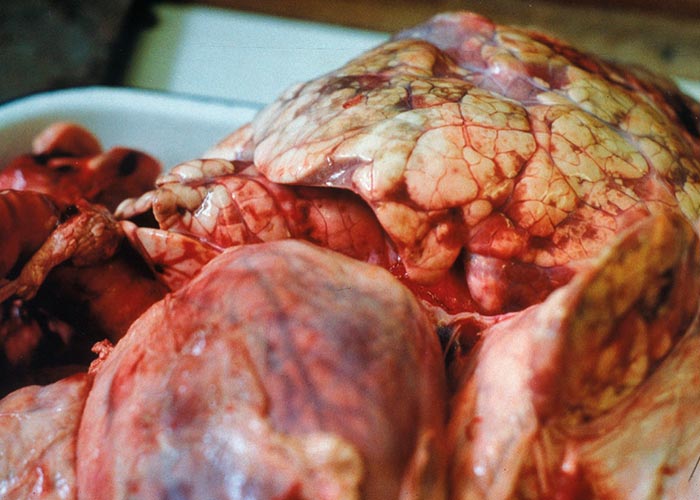
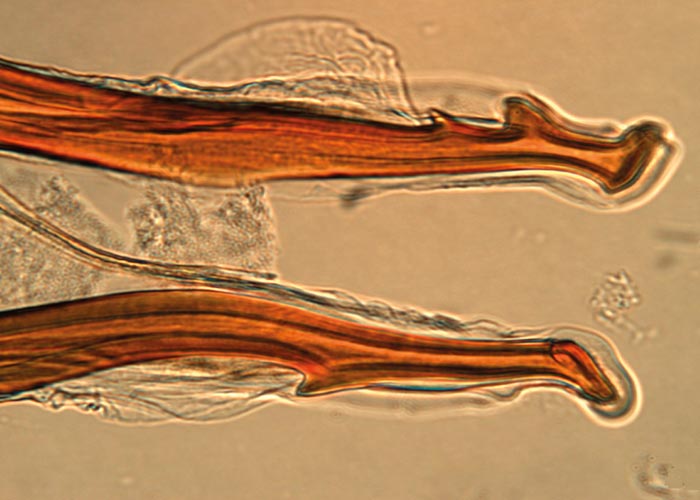
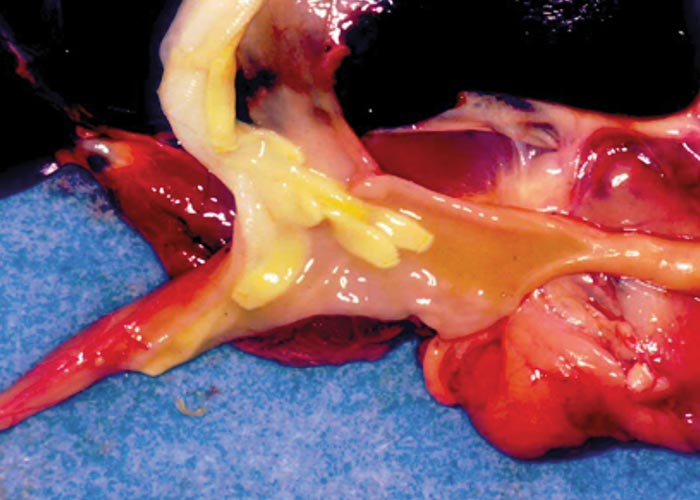
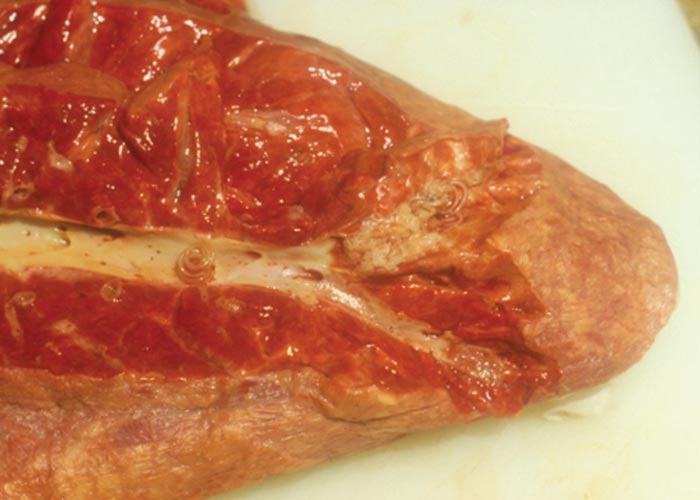
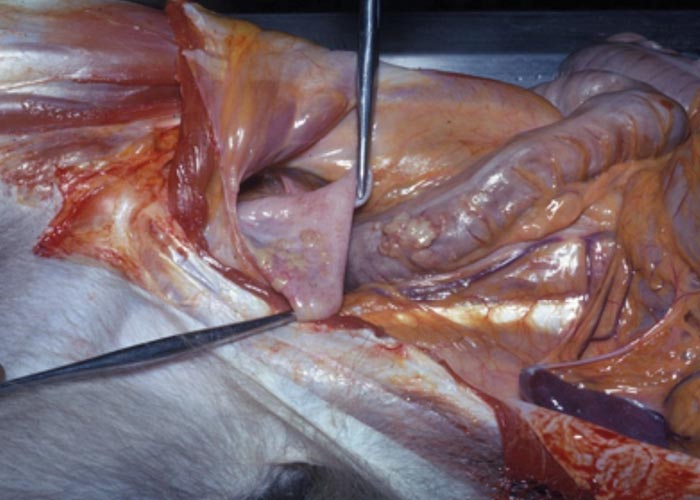
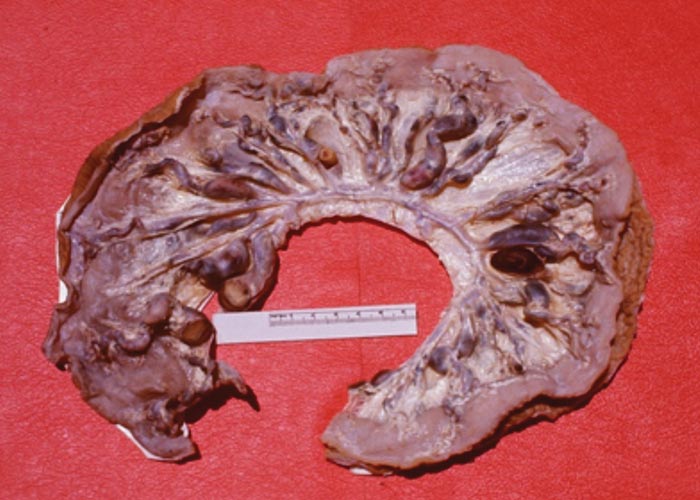
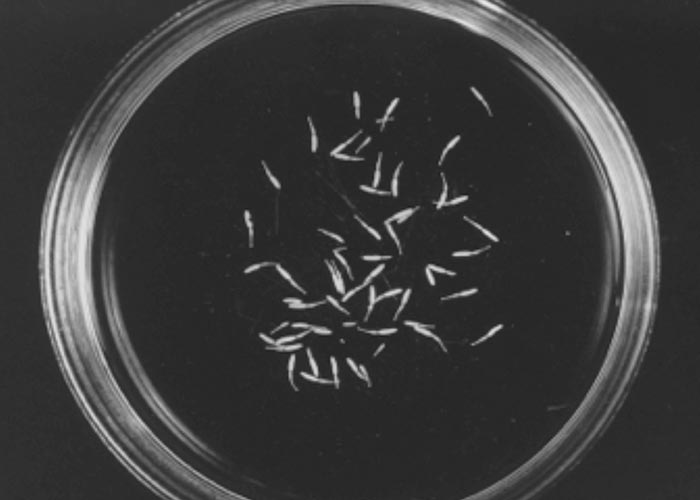
The nematodes Dictyocaulus africana, Dictyocaulus filaria, Dictyocaulus viviparus and Bronchonema magna occur in the bronchi and trachea of a variety of antelopes. The Dictyocaulus species and B. magna in their natural hosts produce lesions similar to, but never as severe as those seen in domesticated ruminants. Initially, the worms cause alveolitis, followed by bronchiolitis and finally bronchitis –as they become mature and move to the bronchi. Cellular infiltrates (neutrophils, eosinophils and macrophages) temporarily plug the bronchioles, causing the collapse of groups of alveoli, leading to the clinical signs of coughing, dyspnoea and breathing with an extended neck. The patent phase is associated with two main lesions. Firstly, a parasitic bronchitis characterised by the presence of many adult worms in a white frothy mucus. Secondly, there is a parasitic pneumonia, characterised by collapsed areas around infected bronchi. The pneumonia is the result of aspirated eggs and L1 which act as foreign bodies and provoke pronounced polymorph, macrophage and multinucleated giant-cell infiltrations. Varying degrees of oedema and emphysema may also be seen ( Figure 89 ). Recovery starts taking place once the adult lungworms have been expelled. The inflammatory reaction resolves and then clinical signs abate.
Horak et al . (1983) found a few blue wildebeest in the KNP with fairly extensive pulmonary lesions caused by Dictyocaulus viviparus ( Figure 90 ). These lesions did not appear to be severe enough to cause death, but may have debilitated the animals sufficiently to make them prone to capture by predators.
Bronchonema magna is considered nonpathogenic for springbok, its natural host. When springbok were introduced into the BNP near Swellendam, bontebok became infected and mortalities occurred. As soon as the springbok were removed, the mortalities stopped. The lesions in the bontebok lungs were similar to those caused by the Dictyocaulus species in other antelope.
As the genus name implies, Pneumostrongylus calcaratus of impala and P. of bontebok both occur in the lung –where they are so intimately associated with the lung parenchyma that it is virtually impossible to obtain intact worms. Pneumostrongylus calcaratus in impala is so common that it is considered ‘normal ’, and apart from a localised parasitic pneumonia sometimes resembling infarcts, the lesions cause no discomfort to both the host species ( Figure 91 ). Gallivan, Barker, Alves, Culverwell and Girdwood (1989) describe P. calcaratus infection in impala from Swaziland. Van der Walt and Ortlepp (1960) recorded mortalities in bontebok as a result of P. cornigerus infection.
Protostrongylus capensis of bontebok and P. etoshai of blue wildebeest and gemsbok, respectively, occur in the lung alveoli where no lesions are produced. Muellerius capillaris is primarily a parasite of the lungs of sheep and goats in the Western Cape Province, and have been recovered from impala, Grant ’s gazelles, and okapi in Kenya (Round, 1968). No lesions have been described.
Oesophagus and forestomach
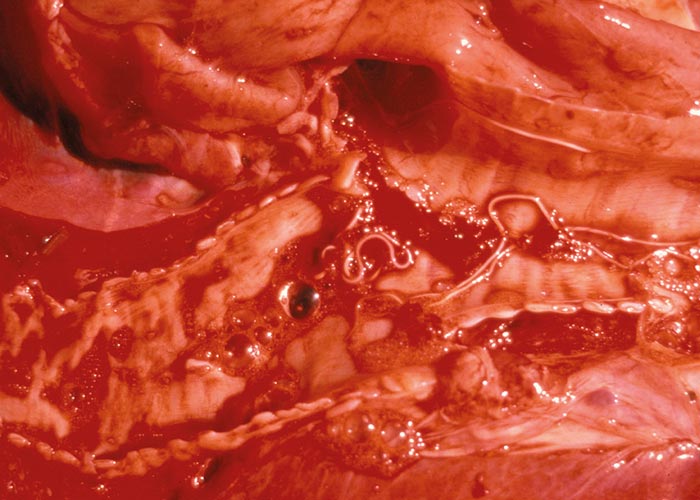
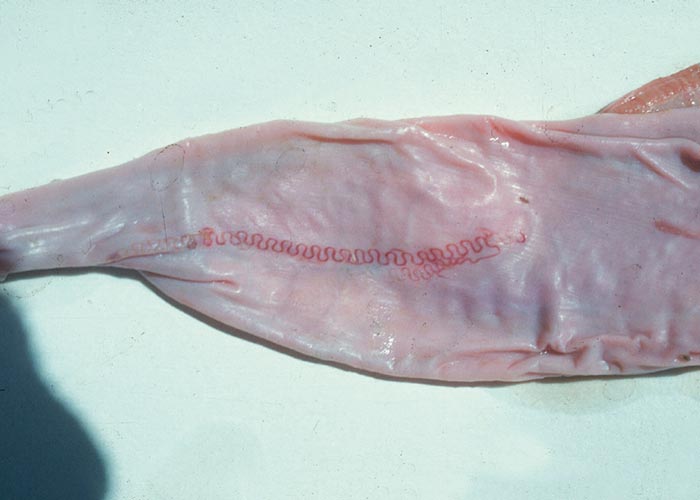
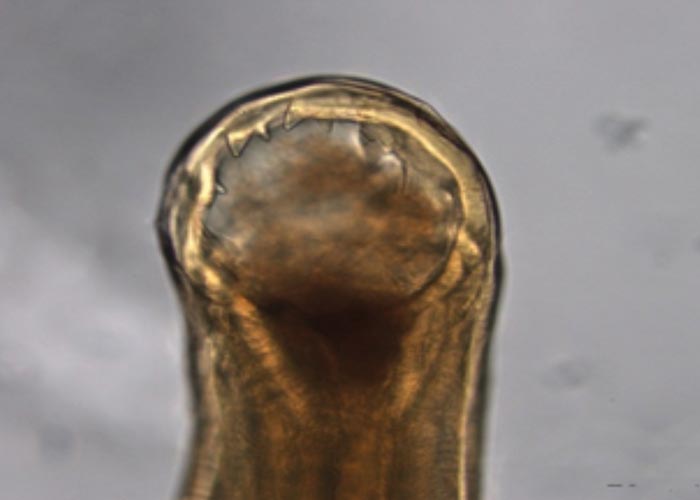
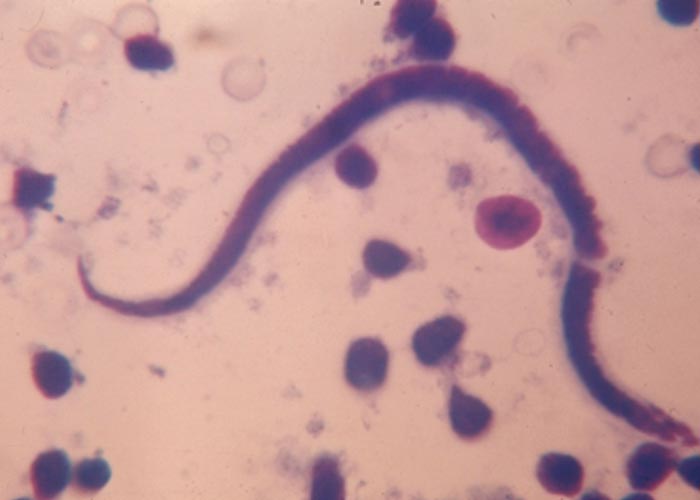
The Gongylonema species –of which there are several –occur in the squamous epithelium of the tongue, oesophagus or the rumen. The worm with its typical zigzag pattern, is the only indication of the presence of the worms. They are non-pathogenic ( Figure 92 ).
Adult Calicophoron in the rumen and reticulum are non-pathogenic ( Figure 93 ). Several species occur in wildlife, all of which have a freshwater snail –usually of the genus Bulinus –as an intermediate host (see Chapter 1 for a description of the life cycle ) .
Abomasum
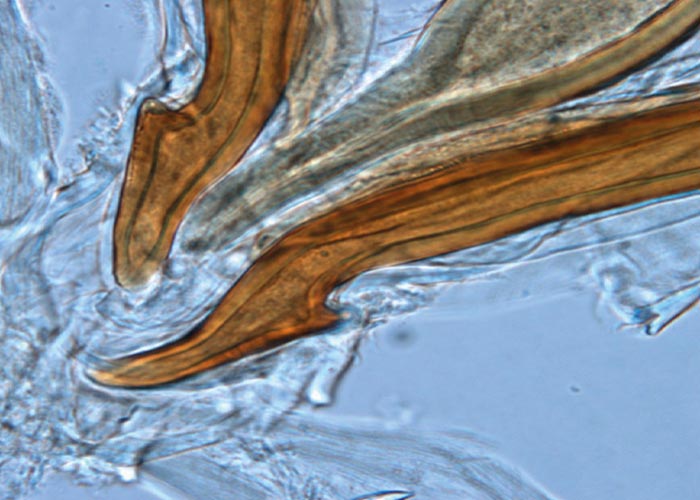
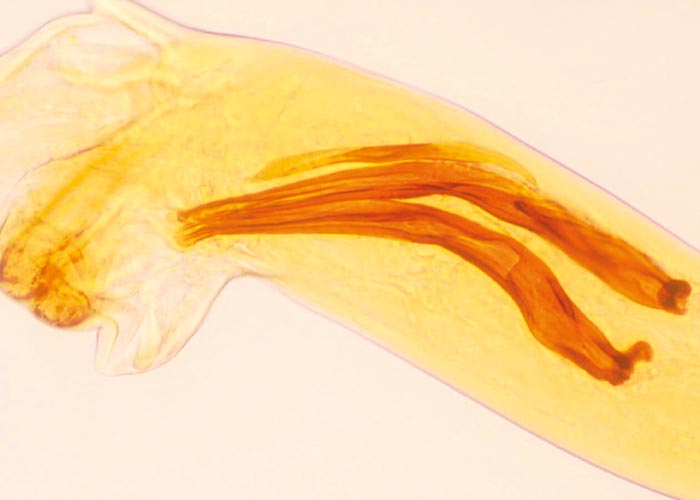
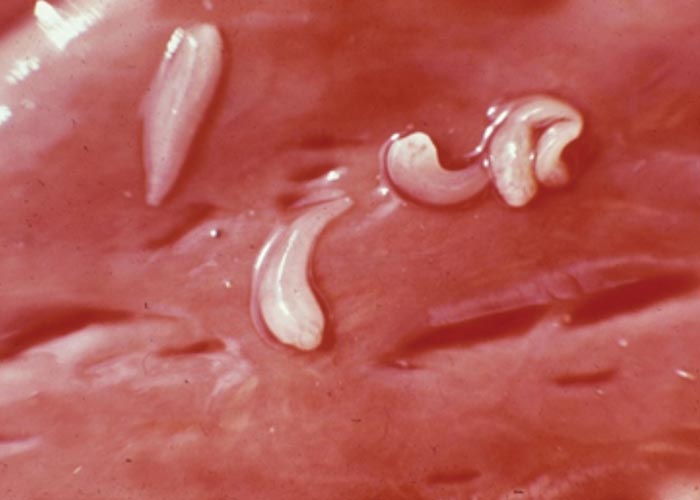
With the exception of the Parabronema spp., the helminths that occur in the abomasum have monoxenous life cycles. A number of Haemonchus species occur in the abomasum of antelope, but their pathogenicity has not been studied. From several surveys in the KNP and in some of the KwaZulu-Natal Parks (KZNP) it became apparent that certain Haemonchus species are associated with certain host groups. For example, in the KNP and KZNP, Haemonchus vegliai ( Figures 94 and 96 ) is associated with the browsing antelope (kudu, nyala and bushbuck), while impala in the KNP harboured Haemonchus krugeri ( Figure 95 ). In areas where game and domesticated ruminants graze the same pastures –for example, sheep and blesbok or impala –the game will harbour Haemonchus contortus , a primary parasite of sheep. This has economic and managerial implications, since the wild ruminants can act as reservoir hosts for resistant H. contortus .
Deaths of sable antelope, roan antelope, and kudu due to anthelmintic-resistant H. contortus infections, are known. In all cases the clinical signs and macroscopic lesions were the same as for similar infections in sheep.
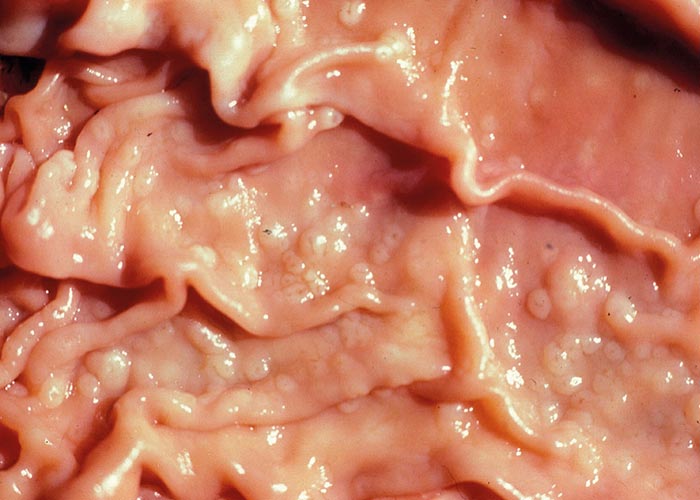
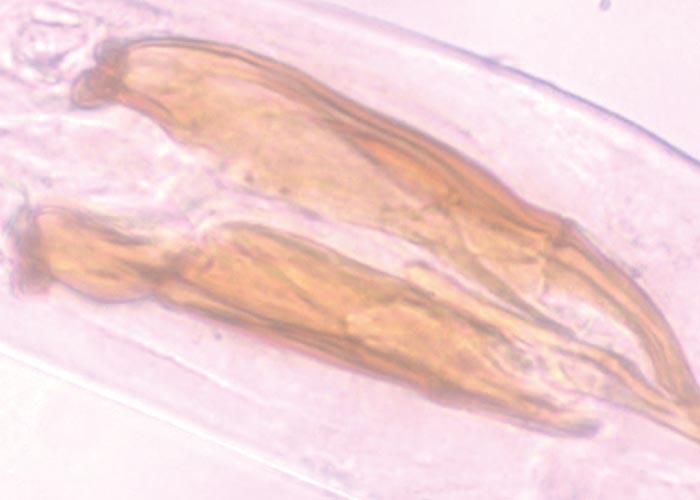
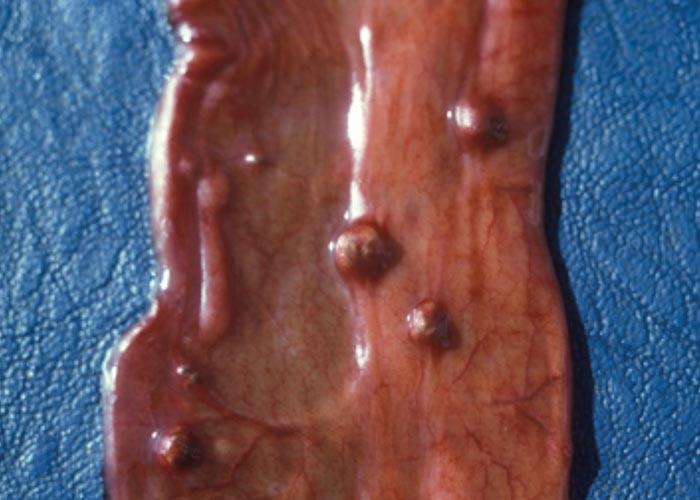
Teladorsagia, Ostertagia and Longistrongylus all belong to the subfamily Ostertagiinae, and produce similar lesions in their antelope hosts ( Figures 97 and 98 ). The lesion is essentially a nodular abomasitis caused by hyperplasia of the mucosa –which in turn is caused by the nematodes that develop and live in the abomasal glands (Pletcher, Horak, De Vos &Boomker, 1984). These helminths have been described from a variety of antelope and no adverse effects were observed. Basson, Kruger and McCully (1968, cited in Basson, McCully, Kruger, Van Niekerk, Young, De Vos, Keep &Ebedes, 1976), however, saw fatal cases of ostertagiosis caused by Ostertagia ostertagi in eland that were kept in small camps.
Trichostrongylus thomasi ( Figure 99 ) is the species usually found in the abomasum of a number of antelope species, and it is the counterpart of Trichostrongylus axei ( Figure 100 ) of domesticated ruminants. No clinical signs have been reported due to the presence of this parasite.
Different species of Parabronema ( Figure 101 ) parasitise elephant, rhinoceros, buffalo and giraffe in South Africa, and camels, sheep, cattle and buffalo in North Africa. All make use of a stomoxid fly –Haematobia or Lyperosia –as intermediate host. The fly larva ingests eggs or first-stage larvae of the nematode, in which they develop to the second stage. They develop to the infective third stage by the time the fly emerges from the pupa. The fly has to be ingested, either with water or food, for the life cycle to continue. Large numbers of worms are often present in the abomasum or stomach, and their presence may cause small ulcers.
Small intestine
Although immature Calicophoron (Paramphistomum) spp. ( Figure 102 ) cause serious disease in susceptible domestic animals –to date only a single case has been reported in nyala. This occurred in the North West Province where the animals were kept in an enclosure and the intermediate hosts were present in a water trough.
The strongylid nematode Agriostomum ( Figure 103 ) occurs in the posterior part of the small intestine, where it, on occasion, produces ecchymoses on the mucosa. Despite their common occurrence, the life cycle of this nematode is unknown, and no clinical signs or specific lesions have been ascribed to it
The family Trichostrongylidae is well represented in all antelope, and Cooperia, Cooperioides, Nematodirus, Impalaia, Paracooperia and Trichostrongylus are the commonly encountered genera. As is evident from Table 16 , there are numerous species of, especially, Cooperia and Trichostrongylus . Large numbers of worms of any or all the genera mentioned above can occur in antelope, but clinical signs, because of their presence, are rarely seen.
Bunostomum trigonocephalum was present in 3 out of 12 impala culled at Pafuri in the KNP, and all three were approximately 8-months old. None showed any clinical disease or macroscopic lesions (Boomker &Horak, unpublished data).
Small numbers of Gaigeria pachyscelis have been recorded from blue wildebeest in the KNP, and mostly in the 4 to 12-month old antelope –again without clinical signs or macroscopic lesions being present (Horak et al ., 1983). Moniezia benedeni, Moniezia expansa and Avitellina are widespread in antelope throughout southern Africa, but no clinical disease has been recorded. These tapeworms are usually seen in young animals.
Large intestine
Oesophagostomum is a large genus of which two species are com monly encountered in antelopes: Oesophagostomum columbianum ( Figure 104 ) and Oesophagostomum walkeri. The former species has been recorded from at least 18 different antelope species, but no mention was made of their pathogenicity in their respective hosts. Oesophagostomum radiatum is fairly common in buffalo in the KNP –but the infection is mild (Basson et al ., 1970). The nodules commonly seen in sheep and goats, and even cattle, are much less conspicuous in antelope.
Several species of the genus Trichuris parasitise wildlife. Trichuris globulosa , one of the more commonly encountered species, occurs in 8 antelope species. It is, however, a rare finding in buffalo and the infection is invariably very mild (Basson et al., 1970) –as it is in most antelope. Because of its monoxenous life cycle and the infective larva that occurs in a thick-walled egg, large numbers can build up in enclosures under intensive conditions. In private collections or zoos, this parasite is one of the most troublesome.
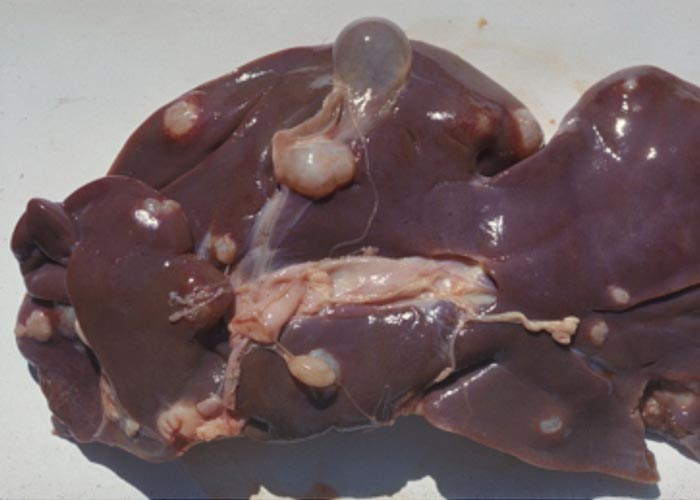
Liver
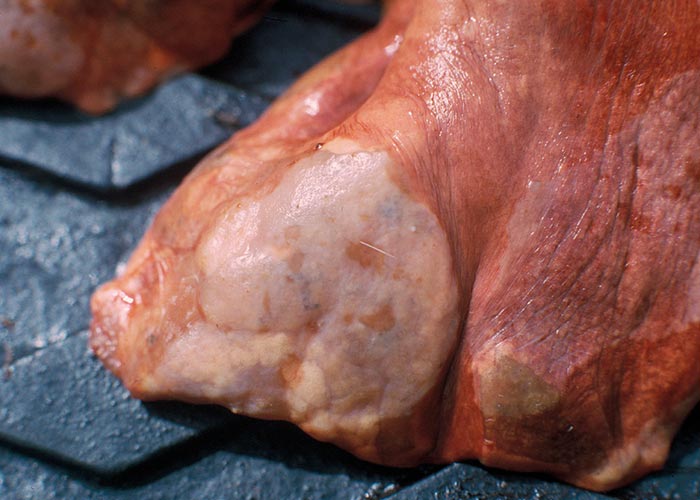
Pletcher, Horak, De Vos and Boomker (1988) describe the lesions caused by Cooperioides hepaticae in impala from the KNP ( Figure 105 ), and concluded that members of this genus are usually of minor pathological significance, unless present in large numbers and in combination with other trichostrongyles. Gallivan, Barker, Culverwell and Girdwood (1996) described lesions caused by hepatic parasites in general, in the same antelope, from Swaziland. Despite these nematodes being present in most impala examined during several surveys, clinical signs have never been observed as a consequence of their presence.
Monodontus giraffae is an extremely common parasite of the bile ducts of giraffes and causes mild to severe cholangitis –depending on the number of worms present (Basson et al ., 1971). Fasciolosis seems to be a rare occurrence in free-living antelope. Basson et al . (1970) did not find a single case in the 100 buffalo examined in the southern part of the KNP. Boomker (1990) examined 386 browsing antelope from all over the country and the northern parts of Namibia and found a single grey rhebuck in the Bontebok National Park which harboured only two Fasciola specimens. Boomker and Horak (unpublished, 1980 –1990) did not find Fasciola spp. in any of the 162 impalas examined from five localities in the KNP, and neither did Heinichen (1973) in the north-eastern part of KwaZulu-Natal. However, Horak (1978) found Fasciola gigantica in one of 36 impala at Nylsvley, Limpopo Province –where they shared pastures with cattle.
Even though antelope seem to be resilient to infections with Fasciola, cases of acute fasciolosis are known. These, however, were present on game farms or where overcrowding occurred due to overstocking. In the dry north-western part of Limpopo Province, metacercariae of Fasciola were found in water troughs –together with the intermediate hosts Lymnaea truncatula .The non-pathogenic liver tapeworm, Stilesia hepatica ( Figure 106 ) is prevalent in various antelopes. Buffalo from the KNP did not show any signs of Stilesia infection (Basson et al. , 1970).
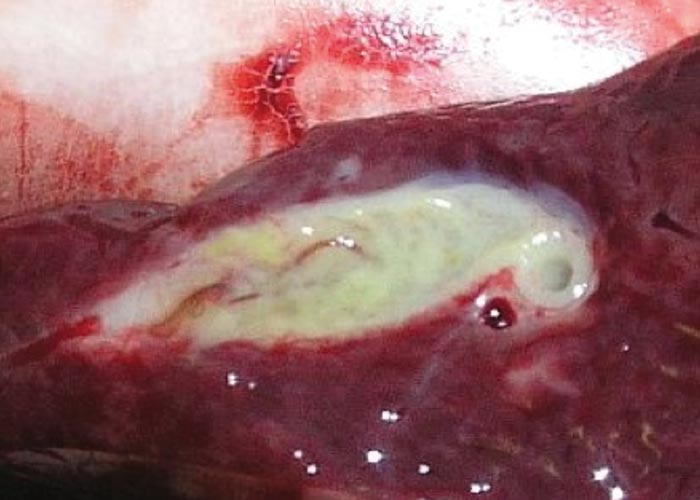
Cysticercosis due to the metacestodes of Taenia hydatigena is a common finding at necropsy of a number of antelope species ( Figure ). After the egg has been eaten, the oncosphere, or hexacanth larva, hatches and burrows through the wall of the small intestine, crosses the abdominal cavity, and then enters the liver. It migrates through the liver parenchyma for a while and leaves the liver in the vicinity of the bile duct. It attaches to the mesenterium in the immediate vicinity of the liver. The infection is dependent on the presence of jackal, Cape hunting dogs or domestic dogs. Boomker (1990) found these cysticerci in blue and grey duiker, but associated lesions were not seen. Round (1968) lists 15 species of intermediate hosts for this tapeworm, including warthogs and bushpigs.
Hydatid cysts of Echinococcus granulosus ( Figure 107 ) were found in one kudu out of the 386 antelope examined by Boomker (1990). Basson et al . (1970) found a 5% prevalence in the buffaloes they processed. Hydatids were not found in the impala examined by Heinichen (1973), Horak (1979) and Boomker and Horak (unpublished data). Hydatidosis, or cystic echinococcosis does not seem to be of importance in the larger nature reserves but could theoretically become problematic on game farms. Infective nymphs of the pentastome genus Linguatula are often encountered in antelope ( Figure 108 ). They utilise antelope as intermediate hosts and the large carnivores –especially lions –as final hosts. The nymphs tunnel in the liver without causing haemorrhage; they were found in 63.2% of kudu (Horak, Boomker, Spickett &De Vos, 1992), in 21.8% of blue wildebeest (Horak, De Vos &Brown, 1983) and in 35.5% of warthog (Horak, Boomker, De Vos &Potgieter, 1988) –all surveyed in the KNP. It is interesting that a browsing species such as kudu has the highest prevalence of this parasite, whereas grazers like blue wildebeest have the least.
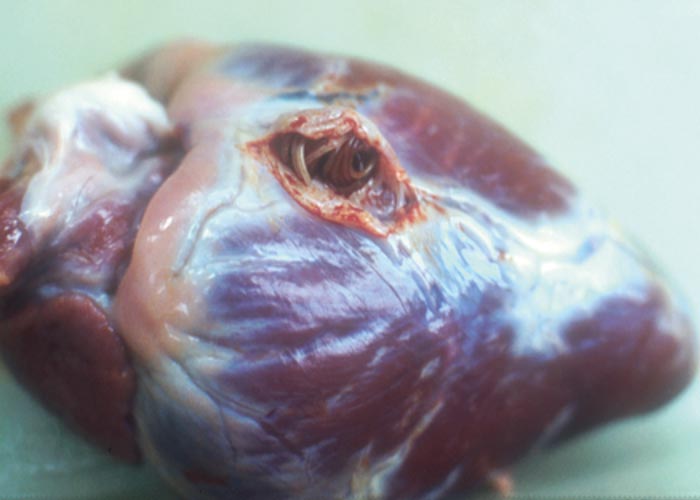
Cardiovascular system
Elaeophora sagitta (Cordophilus sagittus) is found in aneurysms in the coronary arteries ( Figure 109 ), as well as in the small branches of the pulmonary artery –especially in the distal portions of the diaphragmatic lobes ( Figure 110 ). McCully, Van Niekerk and Basson (1967) described the lesions of Elaeophora infections in kudu, bushbuck and buffalo, as did Pletcher, Boomker, De Vos and Gardiner (1989) in kudu from the KNP. Lesions containing live and dead worms were found in bushbuck and kudu from the KNP, and bushbuck and nyala in the northern KZN Parks.
The worm seems to occur primarily in the tragelaphine antelope –i.e. kudu, bushbuck and nyala –and rarely occurs in buffalo and cattle. According to Young and Basson (1976), nearly half of 33 eland transferred from the Addo Elephant National Park to the Kruger National Park died suddenly from acute cardiac arrest due to infection with the parasite. Postmortem examination revealed prominent heart lesions –notably sub-epicardial aneurysms associated with the presence of Elaeophora sagitta . The worms cause a villous proliferation in the pulmonary arteries.
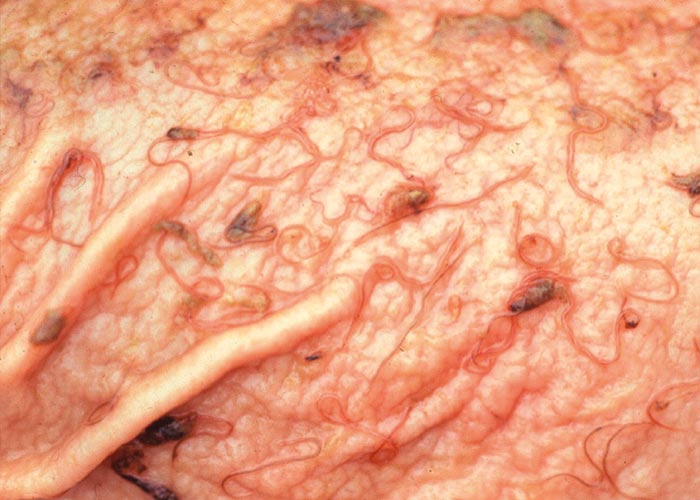
Schistosoma spp. are common in those animals that are dependent on water, and have been recorded from baboons ( Figure 111 ), zebra, hippopotami, giraffe, buffalo and at least 13 species of antelope in southern Africa. According to Basson et al . (1970), lesions are particularly pronounced in ‘river buffalo ’of the KNP, which are the old bulls that have been expelled from their herds. Severe phlebitis and thrombosis of the mesenteric veins were described in one of these buffaloes ( Figure 112 ).
Eighteen of the 96 kudus examined (18.8%) in the KNP had schistosomes in the liver and mesenteric veins (Boomker et al., 1989). The prevalence of Schistosoma in impala from Malelane, KNP was 4.9%, and 11.5% for the same antelope from Skukuza (Boomker &Horak, unpublished data). Conversely, no schistosomes were recovered from impala from Nylsvley (Horak, 1978), impala from a farm in northern KwaZulu-Natal (Anderson, 1983), and reedbuck in the moist St Lucia area of KwaZulu-Natal (Boomker et al ., 1989a).
Skin and adnexa
Approximately 16% of the buffalo in the KNP have lesions caused by one or more of the three species of Onchocerca ( Figure 113 ). The infection manifests as small nodules in the subcutis, mainly in the thoracic, sternal and abdominal regions, but they are also present in the eyelids, prepuce and testis (Basson et al. , 1970). Unidentified Onchocerca spp. occur in 13 species of antelope throughout southern Africa –as well as in leopards in Tanzania (Round, 1968).
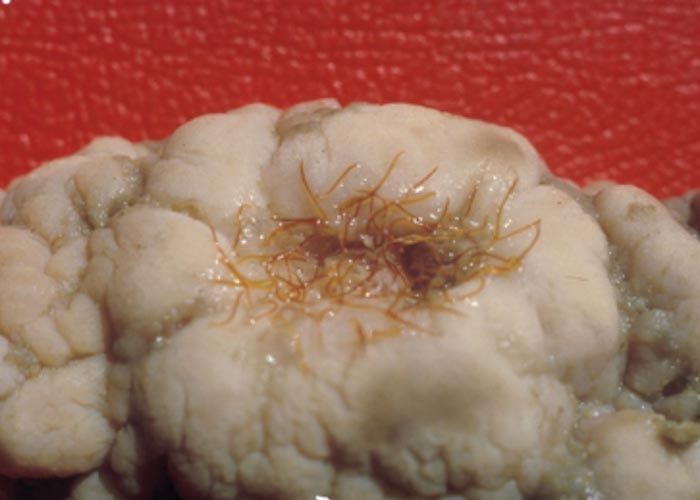
During the late 1980s and early 1990s a skin condition was noticed in buffalo in the KNP, from which Parafilaria bassoni –a filarid nematode previously only recorded from springbuck in Namibia –was recovered. Haemorrhagic perforations or bleeding points were seen dorsally and laterally on the body ( Figure 114 ). Complications due to bacterial infection that caused subcutaneous abscesses, and a type 1 hypersensitivity that caused large ulcers, were seen in a small number of animals. Red-billed oxpeckers often enlarged the bleeding points by feeding on the blood and skin –in the process causing large ulcers ( Figures 115 and 116 ). The oxpeckers played an important part in limiting the spread of the helminths by ingesting blood that contains eggs and first-stage larvae (Keet, Boomker, Kriek, Zakrisson &Meltzer, 1997).
Occasionally the coenuri of Taenia multiceps can be found under the skin or in the intermuscular connective tissue of blue wildebeest, oryx and roan antelope –all three being intermediate hosts of the parasite. The coenuri are recognized by the flabby “sac ”in which numerous protoscoleces are present. Contrary to what is observed in antelope, only those oncospheres of T. multiceps that end up in the central nervous system and spinal cord of sheep will develop into coenuri. The adult tapeworm occurs in dogs and jackals.
A whole host of microfilariae of unknown species have been reported in the literature, from dik-dik in Ethiopia, giant African otter in the Democratic Republic of the Congo, zebra, waterbuck, bushbuck and warthog in South Africa, and steenbuck in Mozambique (Neitz, 1931; Van den Berghe, Chardome &Peel, 1957; Round, 1968; Palmieri, Pletcher, De Vos &Boomker, 1985). These microfilariae may be those of Setaria species, which are quite common in many antelope and warthog in South Africa, or they may represent new species of filarid nematodes. The microfilariae have not been associated with any lesions. However, microfilariae, presumably those of Elaeophora , were associated with a mononuclear myocarditis (Basson et al ., 1971; Boomker et al ., 1989b).
Musculature
The cysticerci of a number of cestodes are known to occur in a variety of the antelopes as well as in warthogs and bushpigs. Most common are those of Taenia crocutae, Taenia hyaenae, Taenia regis and Taenia gonyamai . These can be identified with some accuracy when the hook sizes are compared. Neither the tapeworms themselves in the small intestine of the carnivores nor the cysticerci in the muscles and abdominal cavity of the herbivore intermediate host, seem to have any adverse effect on the tissues or the host.
Basson et al. (1970) found 29% of the buffalo examined in the KNP to be infected with cysticerci. But Boomker et al. (1989) found only 11.3% of kudu in the KNP to be infected and 3% of reedbuck near Himeville, KwaZulu-Natal were infected with Taenia hydatigena larvae (Boomker et al ., 1989).
The sudden appearance of “measles ”(Taenia spp.) was reported in Namibia in kudu after a group of builders ’workers camped on a farm. On slaughter, the meat of the animals was rejected for venison processing (Oberem &Krecek, 2002, unpublished). This is a potentially important source of contamination on game farms.
Nervous system
Setaria labiatopapillosa was found in gemsbok and waterbuck, and both were associated with an eosinophilic cerebrospinal pachymeningitis (Basson et al ., 1971). The cysts of Taenia multiceps have been recorded as causing gid (characterised by an unsteady gait and staggering) in sable antelope and the clinical signs have been recorded on video (Mc Farlane, State Vet).
Helminths of wild suids
Musculature
Trichinella spiralis has a sylvatic cycle which involves lion, spotted hyaena, black-backed jackal, multi-mammate mice, warthog and African civet. South of the Sahara, and especially in East Africa, Trichinella nelsoni appears to be more important in wildlife. Experimental infections of domestic pigs with T. nelsoni and T. spiralis from meat of wild animals in the KNP have indicated that the nematode can adapt, and it may thus become an important zoonosis in future (Young &Kruger, 1967).
Trichinosis is largely asymptomatic in wildlife, but clinical signs are seen in humans. Adult worms in the intestine of humans cause nausea, diarrhoea and vomiting, and when the larvae enter the muscles, oedema of the eyelids and face occurs, and respiratory distress is sometimes seen.
Taenia spp. metacestodes are sometimes seen, depending on how much contact there is with humans and their dogs, or wild carnivores. In large game reserves, the incidence and prevalence of muscle cysticercosis is low. Cysticerci of Taenia solium, Taenia hydatigena, Taenia crocutae, Taenia hyaenae and Taenia regis have been recorded (Round, 1968; Boomker et al ., 1991). As with cysticerci in domestic animals, few lesions are caused by its presence.
Gastro-intestinal tract
Physocephalus sexalatus is a spirurid nematode that infects an intermediate host, usually a dung beetle, as part of its life cycle. It occurs in the stomach of warthog and bushpigs and only when they are present in massive numbers do they cause gastritis.
The genera Oesophagostomum and Murshidia ( Figure 117 ) are large genera that contain numerous species that are particularly abundant in elephant, rhinoceros and wild pigs. Six species of Oesophagostomum –of which Oesophagostomum mocambiquei and Oesophagostomum mwanzae were the most common –and two of Murshidia , have been described from the large intestine of warthogs and bushpigs and were present in huge numbers. An average of 35 000 for the former and 16 725 for the latter species were recovered –a total of almost 52 000 worms per animal (Horak, Boomker, De Vos &Potgieter, 1988; Boomker, Horak, Booyse &Meyer, 1991). Clinical signs were not seen and macroscopic lesions were limited to a few petechiae in the caecum and colon (Boomker, unpublished data, 1989).
Ascaris phacochoeri was constantly found in surveys done in different parts of South Africa, and its prevalence varied from 30.7% to 57%. No reports of this nematode causing disease in free-living warthogs and bushpigs could be found.
The anoplocephalid tapeworms Moniezia mettami and Paramoniezia phacochoeri are regularly encountered in young warthogs, in which they do not cause disease. The trematode Gastrodiscus aethiopicus , is not known to cause disease in warthog but may serve as a source of infection for other species such as horses.
Viscera
A number of wild carnivores have been reported to be hosts of adult Echinococcus spp. worms –including black-backed jackal, Cape hunting dogs, hyaenas and lions (Verster &Collins, 1966; Young, 1975). Sylvatic cycles exist for Echinococcus spp. in the larger game reserves. The larger prey species –which are the antelope, warthog and bushpig –act as intermediate hosts for the tapeworm, but the prevalence is not high. Eight warthogs out of the 52 examined in the KNP had hydatid cysts (a prevalence of 15.4%), while in a nearby Hoedspruit nature reserve where the larger carnivores do not occur, the prevalence was only 3.6% (Horak et al ., 1988; Boomker et al ., 1991). The reason for the low prevalence is the absence of the carnivore final hosts, which interrupts its life cycle. It is also possible that the warthogs that harboured the hydatid cysts were from the KNP –or even neighbouring, large private reserves where the carnivores occur.
Helminths of wild carnivores
As with the antelope and pigs, freeliving carnivores are seldom affected by clinical helminthoses, when in good health and condition. However, it is possible that the high mortality seen in young lion cubs may be due to malnutrition combined with parasite infections –especially the hookworms Ancylostoma and Galonchus . Little has been published on helminth diseases of free-living lions and leopards (Boomker, Penzhorn &Horak, 1997).
Gastro-intestinal tract
Ancylostoma and Galonchus are virulent blood suckers and can cause severe anaemia in a very short time. Spirocerca lupi has been recorded from a nodule in the oesophagus of a lion that was kept at a zoo. Cylicocyclus spp. occur in nodules in the stomach of lions and leopards, and a non-pathogenic Physaloptera spp. in that of cheetahs. Toxocara and Toxascaris probably have the same life cycle as is seen in cats and dogs, and therefore have a more severe influence on the young animals than on the older ones. These ascarids compete with the host for available nutrients. Several Taenia species occur in the small intestine of lion and leopard, and as with similar species in dogs, the tapeworms do not seem to cause significant damage. The species encountered include Taenia regis, Taenia crocutae, Taenia hyaenae and Taenia gonyamai. Echinococcus is one of the most serious helminth zoonoses –so veterinarians and researchers handling wild carnivores should always wear gloves, and should not eat, drink or smoke while working with these species.
There are two subspecies of E. granulosus –E. granulosus granulosus which infects canids and E. granulosus felidis which infects felids ( Figures 118 and 119 ). The tapeworm genera Mesocestoides and Dipylidium have been recorded from lion and leopard, but are of little importance.
Skin
There is a high prevalence of cutaneous dirofilariosis in lion in the KNP. The condition is caused by the filarid nematode Dirofilaria sudanensis . Clinically, it manifests as a large soft subcutaneous lump, but does not seem to cause much discomfort. The nematode is an extremely long one that lies curled up in the subcutis.
Helminths of zebra
Gastro-intestinal tract
A large variety of nematodes occur in the gastro-intestinal tract of zebras. These include the ascarid Parascaris , the spirurids Draschia and Habronema , the strongylid genera Strongylus ( Figure 120 ) and Triodontophorus , and a whole host of cyathostomins –such as Cylicodontophorus, Cylicostephanus, Cyathostomum, Cylicocyclus, Poteriostomum, and Oesophagodontus .
The Habronematidae are represented by Habronema and Draschia, while Oxyuris equi (Oxyuridae) and Trichostrongylus thomasi (Trichostrongylidae) are usually present in small numbers.
The Family Atractidae are tiny worms and occur in tens of thousands rather than tens or hundreds. Millions of worms of the two atractid genera Probstmayria and Crosscephalus have been recovered from healthy zebra –indicating that they are non-pathogenic. Zebra appear to have developed a host tolerance to these huge numbers of worms. Krecek (1984) listed the helminths from 10 zebras shot in the KNP, and a summary of the range in numbers of the helminth families collected, is presented in Table 18 .
Anoplocephala and Anoplocephaloides are tapeworms that occur in the small intestine of zebra, but are not associated with lesions or clinical disease. Gastrodiscus aethiopicus has been recorded from zebras, but its pathogenicity, if any, is unknown.
Table 18 Numbers of worms recovered from zebra from the KNP
| Nematode family | Range |
|---|---|
Strongylidae
Habronematidae Oxyuridae Trichostrongylidae | 1 –137 1 –159 491 2 –24 206 530 4 –1 864 5 –1 515 20 –580 |
Control of helminths in wildlife
Given the sporadic occurrence of helminths in free-living, wild animals and the relatively non-pathogenic nature of these parasites, in general there is no need for the control of helminths in wildlife. From an evolutionary point of view, it is ideal to maintain wild animal populations that are genetically resistant or tolerant to parasites –so treatment is not desirable. However, in captive animals or valuable species being transported or kept under intensive conditions, treatment may be necessary if clinical disease is present. Most anthelmintics are used offlabel, because few anthelmintics have been registered for use in wild animals. Clinicians needing to treat clinical disease should consult veterinarians experienced with the use of anthelmintics in wild species. Wildlife Vetnet provides a forum for discussions/questions on these issues.
It is of cardinal importance that game farms on which valuable species are raised under intensive conditions, make use of quarantine treatment for anthelmintic-resistant helminths such as H. contortus . This species has a high prevalence of resistance and has been recorded as causing problems in sable antelope, roan antelope, and kudu. Similarly, all the other basic principles that apply to the control of helminths in domesticated animals apply to wild animals –namely, preventing overcrowding, ensuring refugia, using remedies judiciously, and, where possible, selecting for worm resistance by eliminating those animals that are particularly susceptible to the infections.
As with infectious diseases, the wildlife/ domestic animal interface must be borne in mind at all times. For example, the wireworm of small stock H. contortus can infect wild antelope and may cause losses in intensively raised species. The situation will be exacerbated if these nematodes are anthelmintic-resistant (see above ) .
The introduction of heminthoses of humans and domestic carnivores which are a potential cause of zoonoses in wildlife, must be prevented. Preventative measures include providing toilet facilities for all humans on game farms and reserves –to prevent infections of Taenia solium and T. saginata . Deworming domestic dogs and cats on a regular basis will reduce the potential of infecting herbivores with the taenid and echinococcal species of carnivores.