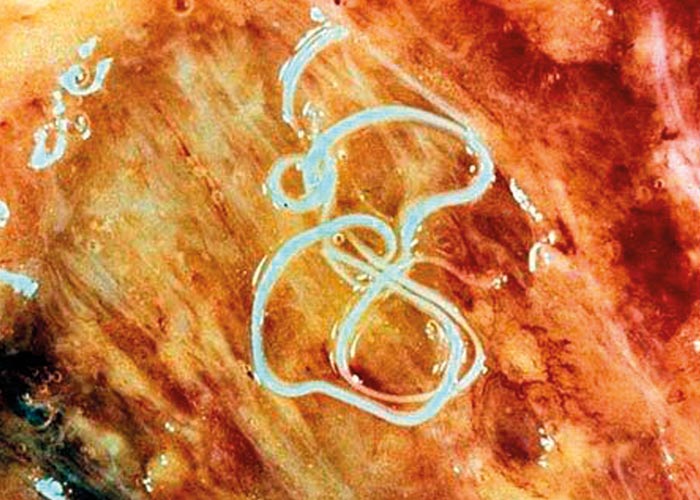Helminths of ruminants
This content is distributed under the following licence: Attribution-NoDerivs CC BY-ND  View Creative Commons Licence details here
View Creative Commons Licence details here

Helminths of ruminants
Author: J BOOMKER
Respiratory system
With the exception of two Dictyocaulus species –Muellerius capillaris and two Mammomonogamus species –no adult helminths occur in the lungs or trachea of ruminants. Several others, however, may pass through the lungs as larvae on their way to their favoured sites elsewhere in the body. Infections with lungworms are not always obvious, clinically speaking. However, in cattle in Europe and sheep in Africa they invariably cause disease in na ïve animals, and, when not treated, often have fatal consequences.
Lungworms
Dictyocaulus species
The Dictyocaulus species belong to the superfamily Trichostrongyloidea, family Dictyocaulidae –and are colloquially known as lungworms. They are large, milky-white worms that are easily seen in the trachea of their hosts. Males are 3 to 8 cm long and females 5 to 10 cm long. The spicules are stout, boot-shaped, and intricately sculptured. Females are ovoviviparous and lay eggs with a thin shell containing a fully developed first stage larva.
Distribution
Dictyocaulus species occur worldwide and are particularly important in temperate climates.
Hosts
Sheep, goats and occasionally some antelope are the hosts of D. filaria , and cattle, deer, reindeer, water buffaloes and camels are the hosts of D. viviparus .
Life cycle
The life cycle of Dictyocaulus species is direct (monoxenous) and the helminths are sometimes referred to as 'geohelminths'. Eggs may hatch in the lungs, but are usually coughed up and swallowed, and the first stage larvae then hatch when they pass through the intestinal tract. The first stage larva of D. filaria has a small cuticular knob at the anterior extremity, which is lacking in D. viviparus . Brown food granules are present in the intestinal cells of the larvae of both species, and the free-living stages live off the food granules. After a few days, the larvae reach the second stage but retain the cuticle of the first stage. Once they reach the third (infective) stage, the cuticle of the first stage is cast off, but that of the second stage is retained. The infective stage is reached after six or seven days.
Infection of the host occurs per os. Larvae penetrate the intestinal wall and pass to the mesenteric lymph nodes –where the third moult takes place. The now fourth stage larvae pass via the lymph and blood vessels to the lungs where they are trapped in the capillaries and break through into the alveoli. They then migrate to the bronchi and trachea to mature into the adult male and female worms. The pre-patent period is about 28 days for D. filaria , and 22 days for D. viviparus .
Transmission
Although D. filaria is cosmopolitan in its distribution, it is only responsible for sporadic outbreaks –even in warmer areas such as parts of Africa and the Mediterranean. Larvae are extremely sensitive to heat and desiccation, but are resistant to cold and can overwinter in colder areas. Carrier animals are important as a source of pasture contamination in Africa, where the climate is often unsuitable for larval survival. Outbreaks of disease are usually seen after prolonged rain around the time that lambs and kids are weaned. Fountains, marshes, streams and rivers provide adequate moisture for the survival of free-living stages, and irrigated pastures can be potential foci of infection.
D. viviparus is similar to D. filaria in many respects, but its epidemiology is largely unknown. Isolated foci of infection occur wherever the climate is suitable for the survival of free-living stages –usually in the cool, moist parts of a region. The parasites are highly prevalent on irrigated pastures. In temperate regions like Europe and North America, these parasites are particularly important (see Urquhart et al . (1991) and http://www.merckmanual.com for a detailed description of the epidemiology in these countries).
Socio-economic importance
Outbreaks of parasitic bronchopneumonia occur sporadically, and then mostly on irrigated pastures, or in the cool, moist areas of a country. Animals lose condition, and unless treated, some deaths may occur.
Pathogenesis
The pathogenesis of dictyocaulosis can be divided into four phases:
- The penetration phase (days 1 to 7) during which the larvae migrate from the intestine to the lungs via the mesenteric lymph nodes and lymphatic system. Neither clinical signs nor pulmonary lesions are seen.
- The pre-patent phase (days 8 to 25) starts when the larvae arrive in the lungs. They cause alveolitis, followed by bronchiolitis, and finally bronchitis as they reach the bronchi. Cellular infiltrates (neutrophils, eosinophils, and macrophages) temporarily block the bronchioli –causing atelectasis when groups of alveoli collapse. The first clinical signs –tachypnoea and coughing –are now observed. Heavily infected animals may start dying from day 15 onwards, due to respiratory failure after the development of severe interstitial emphysema and lung oedema. Epithelialisation and the formation of hyaline membranes in the alveoli commence at this stage.
- The patent phase (days 26 to 60) is associated with two main lesions –a parasitic bronchitis and a parasitic pneumonia respectively. The former is characterised by the presence of many adult worms in the bronchi and distal trachea, which are embedded in a frothy white mucus. There is severe damage to these tissues, which is manifested by a hyperplastic epithelium infiltrated by inflammatory cells, especially eosinophils. The parasitic pneumonia –detectable as collapsed areas around infected bronchi –is the result of the presence of aspirated eggs and first stage larvae that act as foreign bodies. Pronounced polymorph, macrophage and multinucleated giant-cell infiltrations are provoked by the presence of the eggs and larvae. Varying degrees of oedema and emphysema may be seen, and alveolar epithelialisation and hyaline membrane formation become more obvious with progression and increased severity of the lesions.
- The post-patent phase (days 60 to 90) is the recovery phase, after the adult worms have been expelled. The inflammatory exudate in the lungs undergoes organisation, and, clinically, the respiratory rate decreases, coughing is less frequent, and weight gain is resumed. Severe epithelialisation may persist in some animals, 90 days after infection when the worms are usually absent. The remaining lesions consist of peri-bronchial fibrosis and epithelialisation of a few alveoli surrounding some bronchi.
Clinical signs
Animals mildly affected with Dictyocaulus cough intermittently –especially during exercise. Those moderately affected have frequent bouts of coughing, even at rest, and tachypnoea and hyperpnoea are evident. On auscultation, moist r âles are heard over the posterior lung lobes. Severely affected animals show severe tachypnoea and dyspnoea. They often stand with their head with neck outstretched and elbows held away from the chest. A deep, harsh cough is present and r âles are heard over the posterior lung lobes.
Pathology
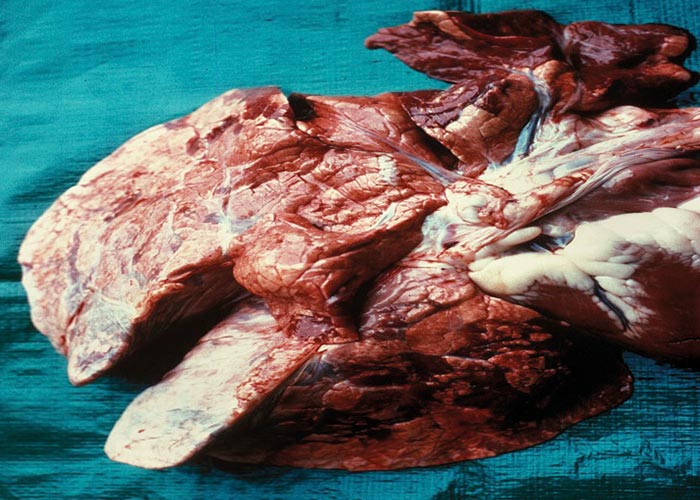
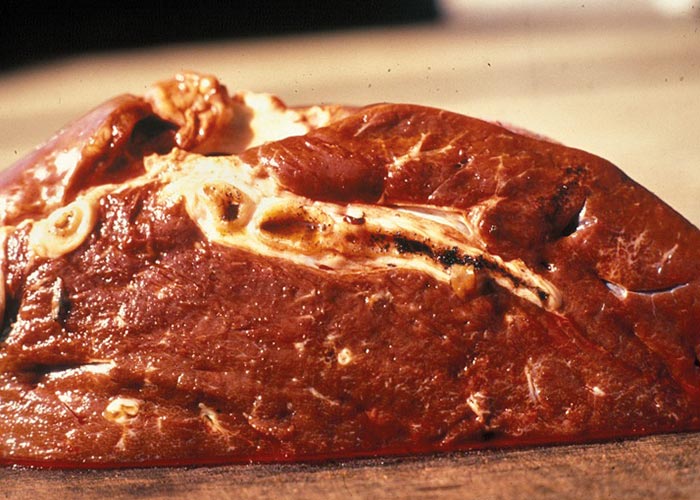
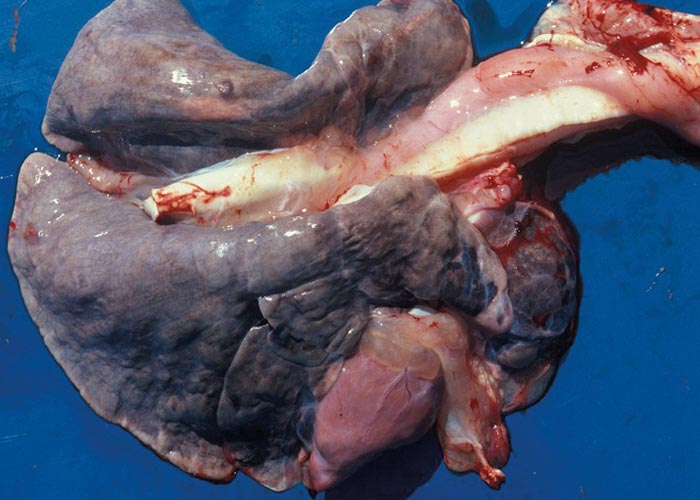
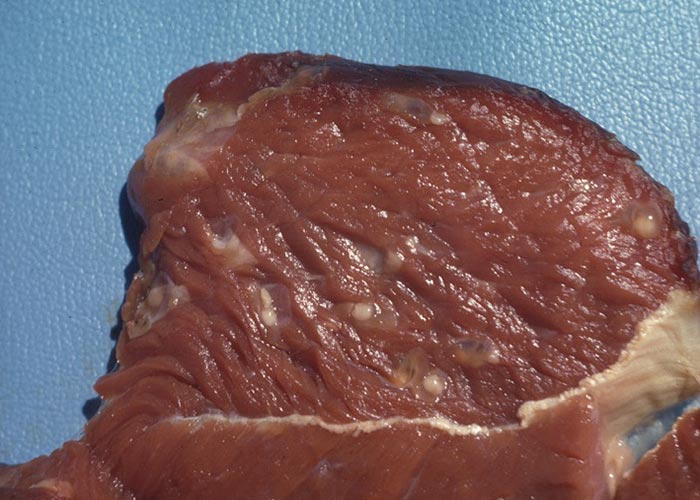
With very heavy infections there may be enteritis resulting from the infective larvae burrowing through the intestinal wall. In Dictyocaulus infections, depending on the severity, lung lesions may be pronounced. Usually there are small foci of pneumonia, resulting from the fifth-stage worms breaking through the alveoli. From about the 10th day after infection, frothy fluid is present in the alveoli and terminal bronchioles, and there may be oedema and emphysema of the interlobular septa. Most of the bronchioles contain plugs of exudate. As the worms mature and move to the bronchi, these plugs may resolve. The mature worms in the bronchi are easy to see. They are surrounded by a frothy bronchial exudate and cause atelectasis and emphysema secondary to the bronchitis –the latter being provoked by aspirated eggs and larvae. The lung lesions appear as large dark-red or grey, wedge-shaped areas slightly sunken below the surface of the surrounding tissue, and are usually situated in the posterior border of the diaphragmatic lobes ( Figure 19 ). In cases where bacterial infections took place, the lesions are complicated purulent pneumonia. There is no pleuritis.
Diagnosis
The diagnosis of lungworm infection is based on the presence of rapid breathing, bronchitis and coughing, and the demonstration of larvae in fresh faeces by the Baermann method. To avoid contamination with soil nematodes, faecal specimens should be collected from the rectum. Eggs may be found in nasal or oral discharges, but their absence is not an indication that the worms are absent.
Differential diagnosis
Other causes of bronchitis and pneumonia, such as Pasteurella , contagious bovine pleuropneumonia, and jaagsiekte, and causes of a purulent nasal discharge such as Oestrus ovis –should be considered.
Control
In the case of Dictyocaulus species, control is difficult as it involves water and grazing management. Because of the rapid build-up of larvae, wet areas, especially in the cooler parts of a country, should not be used for grazing unless all animals have been treated and are free of the worms. In temperate regions, the only reliable method of control is vaccination with irradiated larvae. The vaccine is only available in Europe.
Muellerius capillaris
Mammomonogamus
Muellerius capillaris is a member of the superfamily Metastrongyloidea, and family Protostrongylidae. Males are 12 to 14 mm and females 19 to 23 mm long. The posterior end of the male is spirally coiled and the spicules are 0.15 mm long. Each spicule consists of a proximal alate part and two serrated distal 'arms'. The eggs measure 0.1 by 0.02 mm. Cystocaulus, Spiculocaulus and Neostrongylus are minor related genera. Mammomonogamus belongs to the superfamily Strongyloidea, and family Syngamidae . They are red in colour, and males and females are permanently joined. Males are 4 to 10 mm long and females 8.5 to 23 mm long. Eggs are ellipsoidal and measure 0.075 to 0.098 by 0.042 to 0.054 mm.
Distribution
Muellerius also occurs worldwide, with the exception of the arctic and subarctic regions, and is possibly the commonest lungworm of sheep in Europe, the eastern USA and the winter rainfall regions of Australia. In South Africa, it occurs in the winter rainfall area of the Western Cape Province. Mammomonogamus is a parasite of the tropical and subtropical regions and occurs in India, Malaysia, Vietnam, South America, and parts of Africa. Cattle in the Ethiopian lowlands are often infected with this worm.
Hosts
Muellerius capillaris is a heteroxenous parasite or 'biohelminth' that uses snails and slugs as intermediate hosts. The eggs develop in the lungs of the host and the first stage larvae pass out with the faeces. The tail of the larva has a spine and an undulating tip. These larvae can resist a fair amount of desiccation, are not killed by freezing, and are most active at temperatures of about 17-27 °C. For further development, they must enter a mollusc –the intermediate host –by penetrating the foot of the mollusc. The infective stage (L3) is reached after 12 to 14 days. These larvae can live in the snail/slug for as long as it lives –and for a week after its death. The final host becomes infected when it consumes the mollusc with its food. Larvae pass through the intestinal wall into the mesenteric lymph nodes, where they moult to the fourth stage. They then go to the lungs via the lymph and blood vessels. Trans-placental transmission takes place and larvae have been found in the liver and lungs of foetuses and new-born lambs. The pre-patent period is 6 to 10 weeks. The life cycle of Mammomonogamus is unknown.
Transmission
The epidemiology of Muellerius capillaris and Mammomonogamus is largely unknown. However, Muellerius is usually not found in animals younger than 6 months. Its prevalence increases with age and 100% of animals older than 3 years may be infected. The ability of the L1 to survive for months in faecal pellets, together with the survival of L3 in the snail or slug for the duration of the mollusc's lifetime, ensure the endemicity of this worm. Muellerius is not considered to be pathogenic and even in severe infections clinical signs are rare. Occasionally, heavily infected goats show clinical signs varying from moderate dyspnoea and a persistent cough, through to frank pneumonia. All infected animals are predisposed to secondary bacterial infection because of the damage done to the lungs by the parasite.
Mammomonogamus is not considered to be a serious pathogen, although coughing and some loss of condition are seen when several worms are attached to the larynx. In humans infected with this parasite, coughing and haemoptysis may occur. Adult Muellerius are contained in nodules varying from 2 to 3 mm to 200 mm in diameter, and within the nodules they are contained within an inflammatory reaction characterised by the presence of leukocytes, a few giant cells, and a connective-tissue capsule.
lives in the alveoli and lung parenchyma and produces nodules up to 20 mm in diameter. These consist of necrotic material, leukocytes, and a few giant cells surrounded by a connective-tissue capsule. The content of the nodules may calcify. The eggs cause the formation of smaller nodules due to a leukocyte and epithelioid cell reaction; the reaction usually subsides once the eggs have hatched. In some (rare) cases, an adenoma-like proliferation of the bronchial epithelium occurs. However, despite the nature of the lesions, clinical cases are rarely seen. Muellerius infection is associated with the presence of small, spherical nodules that usually occur near or on the lung surface. On palpation, these feel like lead shot. Nodules in which single worms occur are small and cannot be seen macroscopically, while the larger ones contain several adult worms and eggs, larvae, and some cellular infiltrate.
The pathogenesis of Mammomonogamus is unknown.
Treatment and control
Snails and slugs must be controlled where Muellerius is a problem. However, due to the worm's low pathogenicity, snail/slug control is seldom applied. Both lungworms may be treated with levamisole, any of the benzimidazoles, or the macrocyclic lactones at the recommended dose.
Oesophagus
With the exception of a single nematode, Gongylonema, helminths do not occur in the oesophagus of ruminants.
Zigzag worm
Gongylonema
Gongylonema is a nematode in the Order Spirurida. It is a large worm that inhabits the sub-epithelial tissue mucosa or submucosa of the oesophagus –where it lies in a characteristic zigzag pattern. It is non-pathogenic and is usually an incidental finding at necropsy.
Distribution
Gongylonema is distributed worldwide, wherever dung beetles –the intermediate hosts –occur.
Rumen
Although most helminths pass through the rumen at some stage in their life –few live there. The nematode Gongylonema can occasionally be found at the oesophagoruminal opening, but it prefers the oesophagus itself. Trematodes belonging to the family Paramphistomatidae are virtually the only ones that colonise the rumen, but only as adults, as the immature stages occur elsewhere.
Conical fluke (Amphistomes)
Calicophoron microbothrium
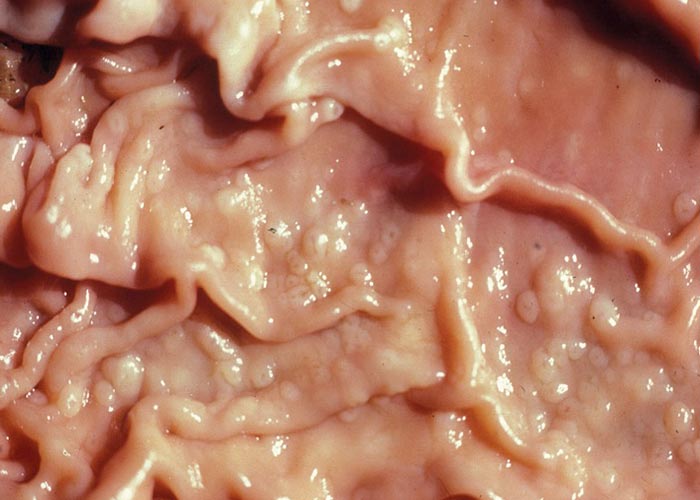
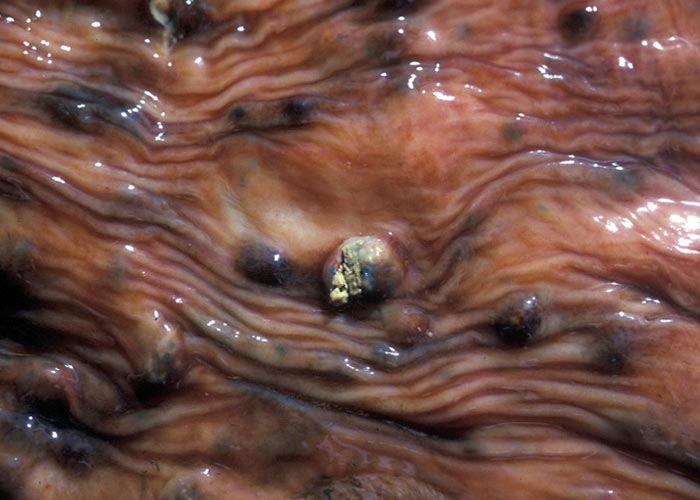
Calicophoron microbothrium (previously Paramphistomum microbothrium) , the conical fluke, is found in the small intestines (immatures) ( Figure 21 ) and the rumen (mature flukes) ( Figure 20 ) of cattle, sheep, goats and most wild ruminants in the tropical and subtropical regions of the world. Other genera are also involved.
Hosts/reservoirs
The genus occurs in sheep, goats, cattle, buffalo and pigs –and less frequently in horses, donkeys, and camels. It also occurs in the crop of domestic chickens, and has also been found in the oesophagus of humans.
Transmission
Dung beetles of the genera Aphodius, Onthophagus, Blaps (and others) transmit Gongylonema. It has also been shown that the cockroach, Blatella germanica , can act as an intermediate host. Infection of the final host takes place by ingestion of infected beetles.
The life cycle, as described below, is the same for all genera, with the exception of the developmental times which vary in different genera and species. Eggs are passed with the faeces and hatch in water 12 to 26 days later. Miracidia enter the young of the aquatic snail Bulinus tropicus (or any other suitable snail intermediate host) at birth, and up to the age of 3 weeks. Older snails are not infected. Sporocysts are found after one day and may persist for up to 11 days. The first rediae are present 14 days after the snail has been infected, and daughter rediae occur after 20 to 28 days. Cercariae are present after 30 days and start emerging from the snail by the 43rd day. Snails may remain infected and shed cercariae for up to 1 year. The cercariae encyst on vegetation to form the metacercariae. They will die if desiccated or completely submerged, but remain viable for 2 months under cool, moist conditions. Once ingested, the metacercariae excyst in the first three metres of the small intestine, and the young flukes attach to the mucosa. After about 15 to 56 days in the small intestine, the young flukes start migrating to the rumen. Their entire life cycle from the time that the eggs are laid until the next generation eggs are laid, takes a minimum of 110 days.
Distribution
Conical flukes occur worldwide wherever ruminants occur or are farmed –with the possible exception of North America. They are of little significance in the northern parts of the northern hemisphere (Europe, Canada, Russia and the United States), although a species of Calicophoron occurs in Scotland, Ireland and Holland. They only occasionally cause disease in the tropics and subtropics.
Apart from Calicophoron, amphistomosis is caused by several other genera in different parts of the world. Cotylophoron and Calicophoron are responsible for outbreaks of amphistomosis in a variety of ruminants all over the world. Ceylonocotyle occurs in water buffaloes and cattle in Asia and cattle in Australasia, and Bilatorchis in cattle in Indonesia. Several genera of lesser importance are sometimes encountered; these include Homologaster in Asia, Carmeyerius in India and Africa, and Gastrothylax in India, Ceylon and China. In short, members of the Paramphistomatidae occur all over the world, but abound in the tropics and subtropics –probably because of environmental factors which favour the snails and the survival of the metacercariae.
Hosts
Many different ruminants are the hosts of the various genera of Paramphistomatidae –or amphistomes as they are known colloquially. This includes the domesticated ones and a number of wild ruminants and hippopotami.
Transmission
The flukes are ingested as metacercariae that adhere to vegetable matter or any other solid object at the water's edge. Metacercariae may be transmitted over short distances by waterfowl, and by irrigation. Clinical cases have been encountered in cattle that have grazed irrigated pastures and had no access to open water.
Socio-economic importance
Adult amphistomes have little socio-economic effect per se. Tripe could be condemned as "aesthetically objectionable" –but once cleaned and cooked it can be consumed with safety.
Pathogenesis
Any pathogenic effect associated with amphistomes is associated with the immatures in the intestine. Adults are thus of little consequence in causing disease.
Clinical signs
No clinical signs are associated with adult amphistome infections.
Pathology
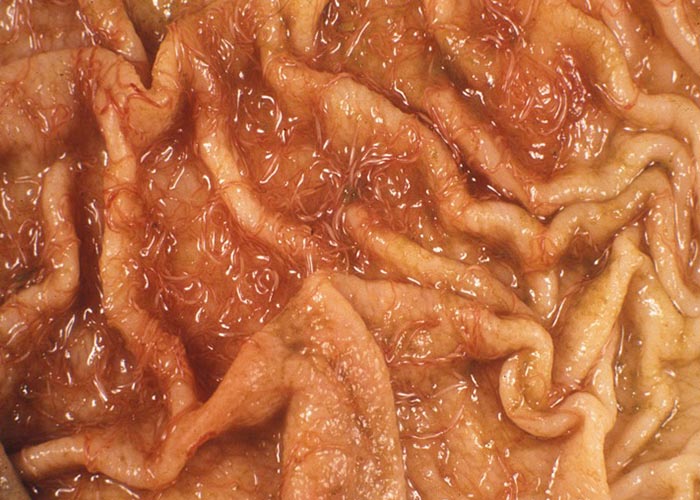
The adult flukes are well tolerated and cause no lesions in the rumen, even when thousands are present. Occasional discolouration of the rumen papillae –to which the flukes attach –may be encountered.
Diagnosis
Since clinical signs are absent with adult fluke infections, a faecal trematode egg count should be done –either by sedimentation or by using the Visser filter. The presence of large, operculated eggs, approximately 0.170 x 0.1 mm, and grey-white in appearance, is indicative. The eggs must be differentiated from those of Fasciola , which are slightly smaller, approximately 0.150 x 0.09 mm in size, and yellow.
Differential diagnosis
Since the condition is asymptomatic, differential diagnoses are not applicable. However, the eggs of Fasciola species could cause confusion. Also refer to "Diagnosis".
Control
Control of adult amphistomes is readily achieved with anthelmintics. Biological control methods such as water supply, snail control and rotational grazing should be employed, and are discussed in greater detail under "Parasitic gastro-enteritis" –specifically the section dealing with trematodes.
Abomasum
There are only a few genera of nematodes that occur in the abomasum of ruminants. Haemonchus, Ostertagia and Trichostrongylus axei are probably the most important causes of disease in cattle, and Haemonchus, Teladorsagia and Trichostrongylus axei in sheep. The genera Ashworthius , Marshallagia and Mecistocirrus are occasionally encountered in cattle and camels.
The nematodes of cattle can also be classified as those of primary, secondary and tertiary importance, but it is generally accepted that they do not cause problems to the same extent as those of sheep. Immunity to the parasites develops faster in cattle –and calves are able to reduce their parasite burdens significantly after 6 months of age.
Occasionally, due to poor management and malnutrition, cattle –particularly calves –may show clinical signs of helminthoses.
It should be noted that all these worms thrive on irrigated pastures and this makes them ideal candidates for the development of anthelmintic resistance.
The socio-economic impact of the helminths of the abomasum of ruminants is severe, not only because of direct losses through death of the animals (as is often the case with H. contortus and the ostertagiids), but also because of the erosive effects of the disease caused by T. axei (hence the colloquial name "stomach bankrupt worm"). The rapidly developing resistance to anthelmintics is an additional socio-economic factor and is discussed in more detail under anthelmintic resistance.
The various abomasal worms all have a direct life cycle; infection is acquired per os, and the free-living third stage larva is the infective stage. Some species of antelope can act as reservoir hosts and may thus be responsible for the dissemination of various species.
Barber ’s pole worm or wireworm
Haemonchus contortus
Wireworm of cattle
Haemonchus placei
Haemonchus are known as barber's pole worms or wireworms. The adults are easily identified by their presence in the abomasum and their large size (2 to 3 cm). Fresh female specimens are conspicuous in having a large vulvar flap and white ovaries twisted spirally around the blood-filled intestine –giving them a barber's pole appearance. The male has a large, asymmetrical, Y-shaped dorsal ray.
The life cycle is direct. The females produce about 10 000 eggs per day. The L1 hatch on the pastures and infective stages can occur within 5 days –during warm, moist weather. However, under adverse conditions development of the L4 stage may be retarded for weeks or even months (hypobiosis). Adults move freely across the abomasal mucosa and suck blood wherever they are. The developmental period is 18 to 21 days. Although Haemonchus placei –the wireworm of cattle –is difficult to distinguish from H. contortus of sheep ( Figure 22 ), its ecological requirements, epidemiology and general behaviour differ so much that –according to many authors –it warrants classification as a separate species. Not all helminthologists, support this, however, and the name H. contortus of cattle may still be found in the literature. The species is widely distributed throughout the world. The developmental period is 23 to 28 days.
Distribution
Haemonchus contortus, Haemonchus placei, Haemonchus similis, Teladorsagia circumcincta, Ostertagia ostertagi and Trichostrongylus axei have a wide distribution wherever cattle, sheep and goats are kept in the temperate, subtropical and tropical regions of the world. This wide distribution is due to the large host spectrum. In Africa, and perhaps in the Middle East and central Asia, Haemonchus longistipes is an important parasite of camels. Longistrongylus elongata and Mecistocirrus are limited to East and North Africa. The temperate regions –such as Europe and the north Americas –are probably the least favoured by Haemonchus .
Transmission
Infections develop in two ways. Firstly, infective larvae develop from eggs deposited by ruminants, especially sheep, in late summer or autumn. These larvae become arrested in the abomasal mucosa as fourth stages (hypobiosis) and will only complete their development the following spring. This spring rise is associated with pregnancy –especially when the ewes are about to lamb or shortly thereafter –but it is not limited to female animals. The spring rise is also referred to as the peri-parturient relaxation of resistance (PPRR) and has an immunological basis. Clinical signs may be seen during maturation of the larvae. Secondly, the worms that have overwintered are responsible for the summer burden, which –depending on the rainfall –may be significant. In the subtropical and tropical regions, haemonchosi s is the primary helminth infection in sheep, and outbreaks are largely seen during summer. Both temperature and humidity are high –facilitating larval growth and development and survival on the pastures. The disease in cattle that is sometimes caused by H. similis and H. placei in tropical and subtropical regions, is similar to the disease in sheep. Severe outbreaks, however, are usually seen during seasonal rains, but have also been recorded at the end of long, dry spells due to the maturation of hypobiotic larvae.
Pathogenesis
The pathogenesis of haemonchosis is essentially that of haemorrhagic anaemia, due to the blood-sucking habits of the worms ( Figure 23 ). Each worm can remove up to 0,05 ml of blood per day by ingestion and seepage from the lesion. There are 3 different forms of haemonchosis:
- Peracute: After infection with 20 000 to 35 000 L3, the resulting L4 cause petechiae, while the 5th stages and adults cause frank haemorrhage and erosions at their attachment sites. Sheep can lose 1 000 to 1 750 ml of blood per day. Death in apparently healthy sheep occurs suddenly as a result of severe blood loss and anaemia.
- Acute: A burden of 2 000 to 20 000 adult worms cause a daily blood loss of 100 to 1 000 ml. Anaemia becomes apparent from about 2 weeks after infection, and is accompanied by a progressive and dramatic fall in the PCV. Subsequently, the haematocrit stabilises and intense compensatory erythropoiesis (visible as hyperplasia of bone marrow) at the expense of the iron reserves occurs. Together with the continual loss of iron and protein (albumin) into the gastrointestinal tract, the bone marrow eventually becomes exhausted, and shortly before death the PCV falls even further.
- Chronic: About 100 to 2 000 adult worms can cause blood loss of about 5 to 100 ml per day. Chronic haemonchosis usually develops during winter when reinfection is negligible, but the pastures become deficient in nutrients, notably protein and iron. The continual blood loss depletes the iron reserves completely, causing marked anaemia to develop before death.
Pathology
- Per-acute cases show few changes, as death occurs too rapidly for lesions to become established. The carcass is anaemic, and there are many petechiae and small erosions in the abomasal mucosa. Coagulated blood –which is often dark brown due to blood seepage –may be present in the abomasal contents. Masses of worms are usually present in the watery, blood-stained abomasal content.
- Acute cases are the most frequently encountered manifestation of the infection. The outstanding features of the acute disease are the extreme anaemia, emaciation, and oedema of the carcass. Compensatory erythropoiesis is seen as hyperplasia of the bone marrow. The abomasal mucosa is hyperaemic, many petechiae and focal erosions are present, and a few small ulcers may occur. The sub-mucosa is thickened and oedematous. The abomasal content is scant, watery and slightly brown, and semi-digested blood clots are sometimes present. The worms are easily seen, but if the necropsy has been delayed for 24 hours or more, the worms may no longer be detectable because of post-mortem changes. However, upwards of 10 000 epg are usually encountered if a faecal examination is done.
- Chronic cases are generally emaciated and pale. The abomasal mucosa shows hyperplasia and metaplasia, and the folds are opaque and thickened. There is evidence of chronic, red bone marrow hyperplasia combined with reversion to white bone marrow –the latter being the result of iron depletion.
Clinical signs
Haemonchosis in cattle shows the same clinical signs as those seen in sheep –but they are generally milder, especially in animals older than 2 years.
Haemonchosis in sheep shows the following clinical signs:
- Animals suffering from the hyper-acute disease die suddenly, with few signs except anaemia and dark brown to black faeces. The hyper-acute form is rarely seen.
- Animals suffering from the acute disease manifest anaemia, bottle jaw, weight loss despite increased food intake, and dry, dark brown to black faeces. Ewes stop producing milk and suckling lambs die of starvation. Lethargy and a break in the wool set in before death. This form of the disease is commonly seen.
- In the chronic from of the disease, animals lose weight progressively over several months –but show neither severe anaemia nor submandibular oedema. Eventually the animal becomes weaker and anorexia sets in. Anaemia is present shortly before death, as is submandibular oedema.
Brown stomach worm of small stock
Teladorsagia
Brown stomach worm of cattle
Ostertagia
Teladorsagia , the brown stomach worm of small stock, occurs worldwide in the abomasum of sheep and goats. The life cycle is direct. Infective larvae enter the gastric pits where they develop and moult to the young adult worms that start emerging from the gastric glands after 18 to 21 days (developmental or pre-patent period). L4 may still be found in the mucosa 8 to 12 weeks after infection; this is a prolonged histotropic phase and should not be confused with hypobiosis.
Ostertagia , the brown stomach worm of the abomasum of cattle, occurs world-wide, and in South Africa they are common in the Eastern and Western Cape Provinces. The life cycle is direct and the developmental period is 18 to 24 days. Like Teladorsagia , infective larvae also enter the gastric pits where they moult to young adults after 9 to 11 days. Adult worms start emerging from the gastric glands after 18 days (pre-patent period). However, L4 may still be found in the mucosa 8 to 12 weeks after infestation –the histotropic phase.
Epidemiology and distribution
Ostertagia and Teladorsagia both prefer cooler climates. In South Africa, the former occurs mainly in the Eastern and Western Cape Provinces from April to September –i.e. autumn to spring –and the latter in the areas adjacent to the former Transkei (Eastern Cape) and Lesotho, from February to June, and again from October to December. In the Highveld of Gauteng and the Free State Provinces, adult worms occur in peak numbers in April, and as hypobiotic L4 from May to October.
Pathogenesis
The L3 and L4 of Ostertagia and Teladorsagia , and presumably of other ostertagiids, cause pressure necrosis in the glandular epithelium and destroy the function of the parietal and zymogen cells. The result is that the pH of the abomasal content rises from 2 to 7 –in which environment pepsinogen is not activated to pepsin (above pH 5 pepsin activity is negligible), meaning that digestion of food cannot take place. In addition, protein is not denatured and bacteriostatic activity is lost, causing an increase in the number of bacteria in the abomasum. The pepsinogen output is further reduced, again resulting in reduced pepsin activity. Blood serum albumin levels remain low until recovery or death. Adult worms suck much blood, as is evidenced by the fall in the haematocrit and haemoglobin values 3 to 4 weeks after infection. However, haemorrhage into the abomasal lumen does not occur.
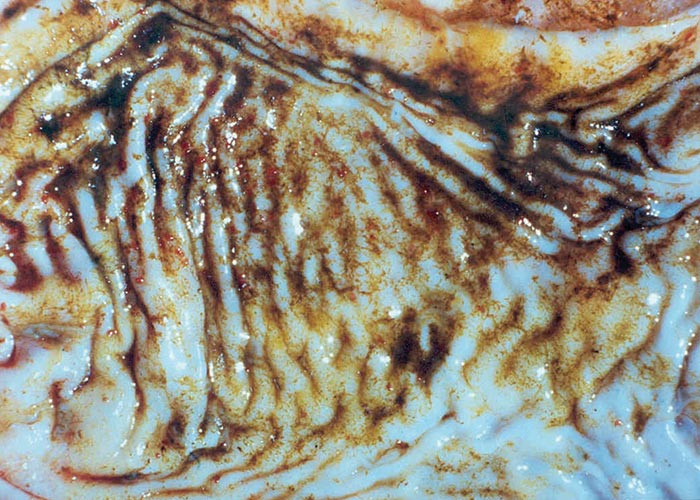
Anaemia, submandibular and sternal oedema, and emaciated and dehydrated carcasses with evidence of diarrhoea –are typical changes seen in teladorsagiosis. There is nodular abomasitis (the nodules sometimes being confluent and covered with tenacious mucus), hyperaemic abomasal mucosa, sometimes small abscesses in the gastric pits where the larvae live, and sometimes –depending on the duration –scar tissue where the gastric pits used to be. The entire mucosa has an 'ostrich leather' appearance ( Figure 24 ) –each nodule having a small, central opening through which the worms can be seen. The picture is one of severe diffuse hyperplastic abomasitis. Angora goats in South Africa develop anasarca and ascites.
Ostertagiosis in cattle presents as a profuse watery diarrhoea, anorexia, loss of mass, severe chronic diarrhoea, dehydration, thirst, emaciation, and death. In the northern hemisphere, a distinction is made between Type I and Type II ostertagiosis. Type I occurs in calves in summer and the larvae ingested develop normally into adults –with the resulting pathogenic effects. Type II is seen in yearlings. The animals are heavily infected, but most larvae remain inhibited as L4. These larvae remain in stabled calves, but cause no clinical signs until they emerge as adults.
In teladorsagiosis in sheep there is anorexia, a marked loss of weight, anaemia, submandibular oedema, occasional diarrhoea and death. In Angora goats there is a marked oedematous subcutaneous swelling of the abdomen and limbs –known as 'waterpens' (literally “water stomach ”) in Afrikaans.
Stomach bankrupt worm
Trichostrongylus axei
T. axei is found in all the domestic ruminants, pigs, horses and occasionally in humans. They occur worldwide, wherever ruminants are kept, and are present in the abomasum or stomach. The life cycle is direct, and the developmental period in ruminants is 24 days. They are very small, thin worms that are not easily seen with the naked eye. Under the microscope, both sexes have a ventral cervical notch into which the excretory pore opens, and the female tail is sharply pointed. Because of the size of the worms, few eggs are present in the uteri. Males are readily recognized by the unequal spicules –of which the right one bears a distinct spine.
Epidemiology and distribution
Trichostrongylus axei occurs from about March to September in winter and nonseasonal rainfall areas, and from March to September in the Gauteng and Free State Highveld.
Pathology
Fair numbers of infectiveTrichostrongylus larvae are necessary to cause clinical disease, e.g. 40 000 larvae cause death in Dorper sheep –but not in Merinos. The worms cause an increase in the abomasal pH, abomasal and serum pepsinogen, and a decrease in the available nitrogen –similar to that seen in ostertagiosis/teladorsagiosis, but not nearly as severe. In abomasal or stomach trichostrongylosis, the carcass is emaciated, the abomasal or stomach content is filled with foul-smelling ingesta, and there is oedema of the stomach mucosa. Catarrhal abomasitis or gastritis is due to larval action, whereas mature worms cause thickening of the mucosa, which resembles wart-like plaques or "ringworm-like" lesions. If these thickenings are removed, an erosion with an intensely hyperaemic base remains. In heavy infections, the thickened areas coalesce to produce a diffuse hypertrophic gastritis.
Infection with Trichostrongylus axei is no longer as common as it used to be. This is probably because of the frequent drenching of ruminants to control Haemonchus . In all animals where T. axei occurs, the clinical signs develop quickly and include rapid loss of body mass –leading to emaciation, diarrhoea and anorexia, which are then followed by weakness and death.
Diagnosis of abomasal worms
When attempting to make a diagnosis of helminth infection, one must consider the age and sex of the animals, and the season, rainfall and the geographic locality of the farm. A detailed history should also be taken. The diagnosis of helminthoses is best done by necropsy, as this is the only way to determine the size and cause of infection. If dead animals are not available or are too decomposed, ask the owner to slaughter an animal. However, this is becoming increasingly difficult because of the value of the animals. Egg counts can be carried out on 10% of the herd or flock. In large flocks of over 500 animals, one should select the tops (fat animals) and the tails (skinny animals) –and then do the egg counts on 10% of each group. Otherwise, one could look for animals with soiled breeches and bottle jaws, and do egg counts on them. Reinecke (1983) stated that "All sheep will be infected as well as young calves and yearlings. The mere detection of infection must therefore be treated with great reserve. No faecal examination can detect immature worms which may have marked pathogenic effects... .”Examples are the L4 of Ostertagia and Teladorsagia. It is clear that egg counts are not the most accurate method to establish a diagnosis as the number of eggs present per gram of faeces depends on a variety of factors; one may have to rely on the identification of L3 larvae obtained by faecal culture.
Differential diagnosis
In the case of haemonchosis, the first differential diagnosis that comes to mind is fasciolosis (because of the anaemia) –followed by other helminth infections, infections with protozoa that cause anaemia, and poisonings that cause anaemia. For the other two abomasal worms, the differential diagnoses would include infection with other helminths and certain poisonings. Chronic cases or mild infections with either of the three genera mentioned here are often difficult to distinguish from malnutrition.
Treatment and control
Small stock
Because anthelmintic resistance is widespread in Haemonchus contortus –and some strains of Teladorsagia and Trichostrongylus in sheep are also resistant to certain anthelmintics –a programme must be implemented which will control clinical disease and limit the development of resistance.
The following is suggested:
- Quarantine treatment: since resistant worms are often acquired by bringing new animals onto a farm, treatment in quarantine is essential. The animals should be dosed sequentially with two different remedies. The use of new actives such as derquantel or monepantel is suggested to ensure that resistant worms are destroyed.
- Identify the worms present on the farm: this can be done by sampling animals at necropsy or using faecal samples for analysis.
- Select a suitable remedy: refer to the MIMS IVS Desk Reference which lists all commercial products currently registered for use against the endoparasites of livestock in South Africa. It is updated annually to include newly registered products. The spectrum of the selected remedy must be carefully considered as it may have to include activity against other gastrointestinal nematodes.
- Use a Targeted Selected Treatment (TST): as discussed under the general chapter on control, the FAMACHA method identifies anaemic animals for treatment for H. contortus infection. For non-bloodsucking worms, animals for treatment can be selected using Body Condition Scoring.
- Vaccination: for additional control of H. contortus , use vaccination to improve the immune status of young animals, in particular.
- Monitor anthelmintic efficacy: using a regular FEC, the efficacy of the anthelmintic in use can be assessed.
- Ensure refugia: dosed animals should not be placed on clean pastures ( see discussion of Refugia under Control, in Chapter 1 ).
- Environmental management: to reduce the worm burdens on pastures, use other species to graze contaminated pastures, as discussed in the section on control.
Cattle
Outbreaks of these helminthoses occur only sporadically in cattle and are best controlled with one of the anthelmintics. Apart from some anthelmintic resistance of Ostertagia in cattle in South Africa, most remedies are effective.
Small intestine
By far the majority of helminths occur in the small intestine of ruminants; this includes members of all the classes –including the Acanthocephala. One can only speculate on why such a diversity of parasites occurs in the intestines, and it is possible that the state of digestion of the food plays a role. The small intestine is also much less harsh an environment than the acidity of the abomasum. Many of the helminths that inhabit the intestine are quite pathogenic (Nematodirus, Trichostrongylus species, Gaigeria, Bunostomum and Calicophoron ), while some are considered to be apathogenic (Cooperia species and the tapeworms). For the purpose of this section, the worms are grouped into the trichostrongylids (Trichostrongylus, Nematodirus and Cooperia) , the hookworms (Bunostomum and Gaigeria), Strongyloides, and Toxocara. The other groups are the tapeworms and Calicophoron.
Trichostrongylids (Roundworms)
Trichostrongylus, Nematodirus, Cooperia
Hookworms
Bunostomum, Gaigeria, Strongyloides, Toxocara
Tapeworms
Moniezia
Fluke
Calicophoron
Several nematodes, cestodes, and a trematode occur in the small intestine of sheep, goats, cattle, and a variety of antelope species:
- The bankrupt worm, Trichostrongylus colubriformis, Trichostrongylus rugatus, Trichostrongylus falculatus and Trichostrongylus vitrinus . All these species are known by the same common name and occur worldwide. They tend to occur in the first 7 m of the small intestine, and rarely in the abomasum or in the stomach. The parasites behave similarly in cattle and sheep, but the condition is usually less severe in cattle and only occurs in young calves. Humans have also been shown to become infected with some of the Trichostrongylus species, as are antelope species, pigs, hares, and rodents. The life cycle is direct and the developmental period is 18 to 20 days.
- The long-necked bankrupt worm, Nematodirus spathiger, is found in the small intestines of sheep, goats and various antelope –and Nematodirus helvetianus in cattle. They occur worldwide wherever sheep and cattle are kept. The life cycle is direct. The first three stages of this worm develop inside the egg, and the infective L3 hatches. The developmental period is 14 to 21 days.
- The white bankrupt worm, Strongyloides papillosus –of sheep, goats, cattle and wild ruminants –occurs all over the world. The life cycle may be homo- or heterogonic. The developmental period is 8 to 14 days. - Bunostomum trigonocephalum, also known as the grassveld hookworm, affects sheep, goats and certain antelope species. The life cycle is direct, and the developmental period is about 30 to 60 days. Infection occurs either percutaneously or per os. The cattle hookworm, Bunostomum phlebotomum –as its name implies –occurs mostly in cattle, but water buffaloes are probably also susceptible. The life cycle is direct. The developmental period is 52 to 56 days and infestation only takes place percutaneously.
- The sandveld hookworm, Gaigeria pectinata, occurs in sheep and goats worldwide, and in impala and wildebeest in Africa. The life cycle is direct. Only percutaneous infection takes place and the developmental period is 70 days.
- The Cooperia species involved with ruminants are C. pectinata, C. punctata, C. spatulata, C. oncophora, C. mcmasteri and C. curticei –and they occur in a wide range of ruminants worldwide. In severe infestations, i.e. animals receiving 300 000 larvae within 10 days, there are clinical signs –but generally these worms are of little importance. The prepatent period is 11 to 19 days.
- Toxocara vitulorum –known as the ascarid of cattle –is seen in suckling calves only. It is morphologically very similar to Toxocara canis . The eggs have a thick wall and are finely pitted. Infection takes place via the milk of the cow and adult worms are present after 33 days.
- The tapeworms that parasitize ruminants are Moniezia expansa and Moniezia benedeni, Avitellina and Thysaniezia . All have indirect life cycles that involve an arthropod –usually one of the oribatids (soil mites) or a psocid (book louse).
- Calicophoron is the only trematode of veterinary importance, as it causes quite severe disease of the small intestine. It has an indirect life cycle and freshwater snails of the genus Bulinus are the intermediate hosts.
Epidemiology and distribution
The helminths occurring in the small intestine occur worldwide, wherever ruminants are present or are kept –with the possible exception of North America.
Hosts/reservoirs
A large variety of ruminants are the hosts of the various genera of intestinal helminths. This includes the domesticated ones and a number of wild ruminants and hippopotami.
Transmission
The trichostrongylids (Trichostrongylus, Cooperia, Nematodirus) and the hookworms (Bunostomum and Gaigeria) have direct life cycles, and transmission is thus per os in the former and either per os or percutaneous in the latter. Strongyloides has a homogonic and a heterogonic life cycle, and infects either percutaneously or through the milk –while Toxocara is transmitted mainly through the milk. Calicophoron has an indirect life cycle ( see “rumen ”) and infects per os, as do the tapeworms which utilise oribatid mites or psocid insects as intermediate hosts.
Socio-economic importance
The socio-economic impact of the helminths of the small intestine of ruminants is severe. This is not only in terms of direct losses through death of the animals, but also through the erosive diseases caused by Trichostrongylus species in sheep and Strongyloides in goats –hence the colloquial names "bankrupt ’’and “white bankrupt worms" respectively. Treatment is expensive and few farmers can afford it, which adds to the impact that these worms have.
Pathogenesis
The larval stages of the trichostrongylids and Strongyloides appear to be the most pathogenic, and, with minor differences, all cause an enteritis, diarrhoea, dehydration, and eventually death with heavy infections. The L3 of Trichostrongylus species burrow underneath the mucosa of the duodenum and the first 6 or 7 m of the jejunum. When these tunnels rupture to release the young adult worms, there is oedema and haemorrhage and a loss of plasma proteins. The Cooperia species have a similar pathogenesis. The L4 and fifth stages of Nematodirus severely damage the villi of especially the ileum –and this leads to villous atrophy and erosion of the mucosa. The larvae of Strongyloides cause erythematous lesions where they penetrate the skin and petechiae in the lungs, and the adults produce a catarrhal enteritis of the duodenum and anterior jejunum. The hookworm larvae cause skin lesions on penetrations and anaemia; hypoalbuminaemia and occasionally diarrhoea are caused by the adults. Initial infection causes swelling at the site of skin penetration and within 24 hours the formation of small isolated scabs. Repeated infections cause severe swelling that may persist for several days. The large mouths of the adults that cut the intestinal villi at their bases, cause intestinal lesions. The crypts of Lieberk ühn and the lamina propria are infiltrated by large numbers of eosinophils, monocytes and lymphocytes. Exposed, haemorrhaging ulcers remain when the worms move to a new feeding site. Anaemia develops gradually and haemoglobin levels drop to as low as 0.35 g/l. The anaemia is of the progressive aplastic type –with no regenerative changes occurring in circulating red cells. The young Calicophoron in the small intestine are “plug feeders ”(Urquhart et al., 1993), which causes erosions on the mucosa.
In heavy infections, enteritis characterised by oedema, haemorrhage and ulceration occurs.
The tapeworms are considered to be nonpathogenic, and even in heavy infections there is little damage to the intestine. They do, however, cause unthriftiness by competing with the host for available nutrients, and may cause the deaths of young lambs and malnourished pregnant sheep.
Pathology
In acute cases of trichostrongylosis the carcass is emaciated and there is atrophy of the fatty tissues. The intestines show catarrhal inflammation with numerous small petechiae in the first few metres of the small intestine. The intestinal walls are thickened and the mesenteric lymph nodes are enlarged. Adult parasites are found beneath the greyish white film that covers the mucosa, but their presence can only be determined by examining a scraping of the mucosa against the light. Changes such as fluid accumulations in the serous cavities, ruminal atony and food retention in the rumen and abomasum, dry ingesta, and distension of the small intestine by fluid may also occur. Carcasses from chronic cases are markedly emaciated, and there is muscular and myocardial atrophy. Mucous membranes are generally pale and the intestinal walls may be thickened.
In nematodirosis the carcass has a dehydrated appearance, and there is loss of the intestinal villi and necrosis of the lamina propria. It is seldom seen in cattle –because they are rarely infected.
In strongyloidosis the carcass is emaciated and wet, and most of the skeletal muscles are atrophied. There are serous effusions in the body cavities and widespread serous atrophy of fat. Intestines are red and the mucosa is lacking in severe infections. The worms can sometimes be seen clumped together and they resemble pieces of cotton wool.
Bunostomum causes emaciation, and bleeding ulcers occur in the mucosa of the small intestines of sheep. In cattle, a typical anaemic carcass with pale mucous membranes and watery blood, is seen. Lesions are present in the mucosa, but free blood in the lumen is seldom seen. The haemopoietic tissues may show signs of compensation –i.e. metaplasia to red bone marrow. Young animals show skin lesions due to larval penetration. Gaigeria causes the same lesions as Bunostomum species.
Cooperia is more of a problem in cattle –in which necrotic enteritis with parasites penetrating the mucosa, haemorrhages in the first 3 m of the small intestine, and catarrhal exudate in the posterior half of the small intestine, are seen on necropsy.
The carcass of a Calicophoron -infected animal is emaciated, dehydrated and there is severe fibrino-catarrhal inflammation of the small intestine, and occasionally the abomasum. Large numbers of small, immature flukes can be found on the mucosa. Ulceration and haemorrhage of the small intestinal mucosa is also evident.
Clinical signs
The clinical signs caused by any one of the Trichostrongylus species appear in acute and chronic forms. The acute disease develops when approximately 250 000 infective larvae are ingested. Pain caused by the parasite causes anorexia, closure of the pyloric sphincter, and retention of food in the abomasum and rumen. Sheep become listless, signs of submandibular oedema develop, and there is a yellow foetid diarrhoea. Sheep die 16 to 17 days after infection. Death is due to the combined effects of starvation, liver impairment, circulatory failure, and pulmonary oedema. Acute disease is rarely seen.
The chronic disease is more commonly seen and develops when about 100 000 larvae are ingested. Anorexia gets progressively worse, with concomitant loss of body mass. Anaemia is caused by a lack of available protein to form haemoglobin. The overall result is emaciation, atrophy of muscles, hydrothorax, hydropericardium and ascites. The clinical signs may be aggravated by poor grazing –especially during winter. Dorper sheep seem to be more susceptible to trichostrongylosis and develop a slight transient bottle jaw. Faeces become putty-like –but not fluid –and animals become weak and listless. Mucous membranes become pale. Merino sheep show slightly pale mucous membranes and putty-like faeces. On lush green pastures, the sheep may have a dark diarrhoea.
The clinical signs in Nematodirus infection are depression, listlessness, anorexia, fluid faeces and death 10 to 14 days after infection. The clinical signs are caused by the L4 rather than by the adults. Animals with a severe infestation of Cooperia species –i.e. those receiving 300 000 larvae within 10 days –have a fluid foetid diarrhoea, selective anorexia, bottle jaw and eventually death resulting from starvation, dehydration and exhaustion.
Like most helminths infecting percutaneously, Strongyloides larvae cause marked urticaria at the site of infection. In goats, adult worms cause anorexia, diarrhoea or constipation, sunken eyes with a purulent discharge, a frothy mucous discharge from the nose, muscle atrophy, and paresis just before death.
Bunostomum causes itching of the skin –particularly that of the limbs (often accompanied by foot-stamping) –and wet eczema. Rapid mass loss occurs, with emaciation, anaemia, submandibular oedema and constipation followed by diarrhoea, with the faeces being foetid and tarry. Animals lie down for a few days before they die. A massive dose (4 000 larvae) can kill adult sheep or young calves without any ante mortal clinical signs. However, more commonly, as few as 200 to 300 adults can produce severe anaemia –causing a fall in the haematocrit levels as well as hypoalbuminaemia.
Gaigeria is a voracious bloodsucker in sheep, and severe anaemia that develops over a period of about 13 weeks is seen together with emaciation, weakness, and loss of weight and appetite. It is usually fatal in under-nourished animals.
Unthriftiness and intermittent diarrhoea are the only clinical signs seen in Toxocara infections –and then only in calves less than 6 months old.
Calicophoron immatures cause anorexia, foetid diarrhoea, mass loss and occasionally bottle jaw. In severe cases death occurs 5 to 9 days after the onset of diarrhoea. Sheep are very susceptible, show severe clinical signs, and quickly die of amphistomosis –whereas cattle rapidly develop a good immunity.
Both Moniezia benedeni and M. expansa tend to occur only in young animals, and a good immunity develops after the first infection. They do not cause significant clinical effects –even with large worm burdens. However, clinical signs may include unthriftiness, diarrhoea, respiratory signs and even convulsions –but then only with massive burdens. Thysaniezia giardi and Avitellina are considered to be apathogenic.
Diagnosis
See “abomasal worms ”for general principles of diagnosis, including autopsy and egg detection. Several of the eggs can be identified to genus level, but the eggs of the trichostrongylids and hookworms are difficult to distinguish from one another.
Differential diagnosis
Differential diagnoses for intestinal helminthoses includes such conditions as poisoning with certain plants or metals, malnutrition, and –in the case of the hookworms Bunostomum and Gaigeria –causes of anaemia like fasciolosis or haemonchosis.
Control of nematodes
The anthelmintic selected must be suitable for the endoparasites on the farm. Although no resistance has been detected so far in intestinal nematodes, good basic practice should be implemented as in the discussion under the control of abomasal worms in small stock.
Control of cestodes
Although considered apathogenic, in practice heavy burdens of Moniezia have been shown to cause mortalities in calves, lambs, and pregnant ewes on dry lands. This can be prevented by strategic dosing of ewes on old lands and of young ruminants when they begin to graze. There is anecdotal evidence of niclosamide resistance in the field, and in such cases suitable alternatives like praziquantel can be used.
Large intestine
Few helminths utilise the large intestine, but those that do are often severely pathogenic. Nematodes that occur here are Oesophagostomum species, Chabertia ovina, and the Trichuris species. Trematodes and cestodes are not, as a rule, found in the large intestine of ruminants.
Nodular worm
Oesophagostomum
Whipworm
Trichuris species
Large-mouthed bowel worm
Chabertia ovina
AdultOesophagostomum are also known as nodular worm and occur in the caecum and colon, while immature stages are found in nodules in the wall of the small and large intestines. They are present in ruminants in the tropical and subtropical regions of the world. The life cycle of Oesophagostomum columbianum of sheep and goats, and Oesophagostomum radiatum of cattle, is direct. The developmental period of the former is 35 to 39 days for the first infection, increasing to 46 to 47 days to many months for subsequent infections. The developmental period of Oesophagostomum radiatum is 32 to 34 days. Wet, warm weather, overgrazed camps or unhygienic kraals and pens will predispose calves and they can be expected to show clinical signs. A third species, Oesophagostomum venulosum , seems to be of lesser importance in sheep and goats. Chabertia ovina is the largemouthed bowel worm of sheep, goats and rarely cattle, and also occurs in some antelope species. Their habitat is the colon and they are found wherever sheep are kept and the climate is suitable. The life cycle is direct and the developmental perio d is 49 days or more. The whipworms (Trichuris species) are named for their long, thin anterior end and a short, thicker posterior. They are found in the caecum and colon. The eggs are lemon-shaped and have a plug at each end.
Epidemiology and distribution
Oesophagostomum columbianum is present in the summer rainfall areas, where rainfall is in excess of 360 mm per annum. It is absent in the semi-arid, non-seasonal and winter rainfall areas.
Oesophagostomum radiatum does not occur in the arid, non-seasonal and winter rainfall areas. In semi-arid areas, it is found in modest numbers in calves in May. In the summer rainfall areas, calves are infested throughout the year, with burdens rising to a peak in August and declining in spring. Irrigated pastures are an unsuitable habitat for this parasite.
Chabertia ovina is confined to winter- and non-seasonal rainfall areas, and is present throughout the year in moderate numbers. Trichuris species occur throughout the world –wherever ruminants, domesticated animals and wildlife are present.
Hosts/reservoirs
Ruminants are the main hosts of Oesophagostomum species, although there are about nine other species that occur in domestic pigs, warthogs and bushpigs. These species are not transmissible to ruminants. Chabertia is only known in sheep, and Trichuris species are present in ruminants, carnivores, suids, and even in humans. Wild antelope are often the reservoir host for the nodular worms and whipworms of domesticated ruminants.
Transmission
The life cycle of all the worms of the large intestine is direct, and infection takes place per os. The free-living stages of Oesophagostomum species are susceptible to desiccation and require hot, moist conditions for optimal development to the infective stages. The species are present in the summer rainfall areas where rainfall exceeds 360 mm per annum. They are absent in the semi-arid, non-seasonal and winter rainfall areas. Sheep quickly expel the worms on improved pastures, and the parasite is absent on irrigated pastures. Trichuris also has a direct life cycle, but in this case the eggs contain the infective L1 and can survive for years in dry pellets. The eggs are also resistant to desiccation and survive temperatures of - 20 °C to 50 °C.
Socio-economic importance
The socio-economic impact of the helminths of the large intestine of ruminants is severe –not only in direct losses through death of the animals, but also through the erosive disease caused by Trichuris species in sheep and O. radiatum in cattle. Treatment is expensive and few farmers are able to afford it, which further adds to the impact that these worms have.
Pathogenesis
Anorexia is the most important finding with ovine and caprine oesophagostomosis. It is caused by the intestinal discomfort starting with the larval migration to the intestinal lumen –and persists until death or recovery. A mild mucoid to projectile foetid diarrhoea sets in, and this may lead to intussusception. Death caused by starvation, dehydration and exhaustion may occur from days 18 to 22. Calves pass blood from as early day 16. This becomes more severe from the 19th and 20th day as the worms moult (M4). Like lambs and kids, calves die of dehydration and exhaustion, or recover from the 10th week onwards.
Chabertia browse on the mucosa causing haemorrhage from the 4th week onwards –and faeces become blood-flecked. Larval stages of Trichuris cause haemorrhage and local oedema when they penetrate the intestinal wall. These injuries are considered to predispose to the development of secondary bacterial infections. Unless they are present in large numbers, adult worms are not pathogenic.
Pathology
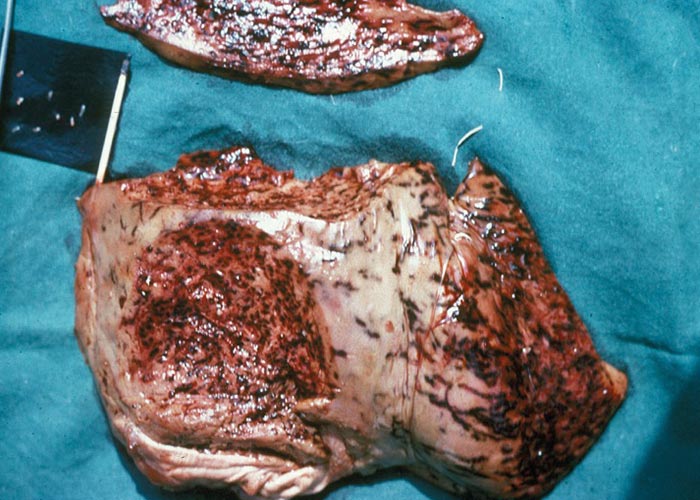
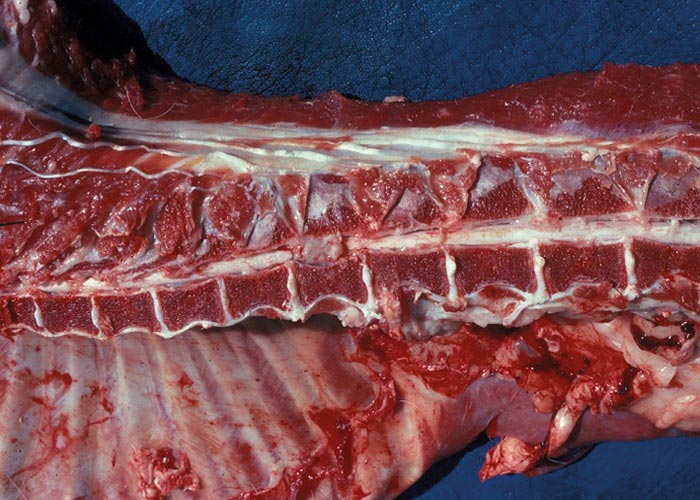
In O. columbianum infections, thickened patches are noted on the intestinal mucosa and, as the larvae leave the mucosa, ecchymoses appear. Extensive nodule formation and thickening of the mucosa of the colon occurs –together with peritonitis and adhesions ( Figure 25 ). There may be a diphtheritic jejunitis, typhlitis and colitis –with numerous perforations and adhesions. At a later stage the nodules may calcify. Once the worms have reached patency, only nodules and a thickened intestinal wall with adhesions may be visible. The lesions caused by O. radiatum are similar to those of O. columbianum, but calcified nodules are rare and haemorrhage is a more common finding.
The mucosa of the caecum and colon of sheep infected with Chabertia contain haemorrhages and inflammatory areas which are produced by the adult worms. The adults cause extensive damage because they frequently move to new sites to feed. Only in heavy infections do the immature stages cause tissue damage –and then throughout the entire intestine from the pylorus to the ileocaecal valve.
Clinical signs
Acute oesophagostomosis is seldom seen in sheep and then only in na ïve animals. It is caused by larvae that have not yet reached patency and is characterised by pain, a rise in temperature, diarrhoea and rapid dehydration. In the chronic disease food and water consumption decreases and then improves again. Diarrhoea starts from the 10th day onwards, and persists until death. The faeces vary in consistency –from putty-like to mucopurulent, green and foul smelling. Intussusception may occur. This form of oesophagostomosis used to be quite common, but, due to intensive treatment aimed mainly at Haemonchus contortus , it has become scarce.
In calves, pain, anorexia, loss in mass, hypoproteinaemia, anaemia and diarrhoea are seen. Submandibular oedema and progressive cachexia occur after 7 weeks. Chabertiosis manifests with weight loss, diarrhoea and faeces that may be flecked with blood.
Adult Trichuris worms are not pathogenic –unless present in large numbers when they may cause abdominal pains, mucoid diarrhoea, anaemia, loss of body mass, and, rarely, death.
Diagnosis
Faecal nematode egg counts are of little value for a diagnosis of oesophagostomosis. Clinical signs –and the presence of nodules in the wall of the small intestine and colon –are diagnostic at necropsy of sheep; nodules are seldom seen in cattle. To make a diagnosis of chabertiosis, consider the clinical signs, the season and the area. Lesions in the intestines and the presence of small numbers of the worms are diagnostic. The eggs of Trichuris are diagnostic. As mentioned above, large numbers of the worms –and thus their eggs –must be present before a diagnosis can be made, as the worms are usually apathogenic.
Differential diagnosis
Causes of diarrhoea such as coccidiosis and nutritional causes should be taken into account for Oesophagostomum and Trichuris species. For Chabertia , consider causes of anaemia like haemonchosis, trichostrongylosis, fasciolosis, coccidiosis and nutritional issues. Also consider causes of diarrhoea like other helminth infections, nutritional deficiencies, poisonings, and coccidiosis.
Control
On commercial farms, where sheep are regularly drenched, oesophagostomosis should not be a problem. The drenching interval for Haemonchus is such that Oesophagostomum is exposed to the anthelmintic twice during its pre-patent period. Sheep also develop a local immunity against the larvae –mainly in the large intestine –and adult worms are expelled in subsequent infections.
Cattle in the field develop a strong immunity after 8 to 12 months of age. The immunity may cause a great reduction in the number of re-infesting larvae developing to adults during the normal pre-patent period. The immunity will persist if adult worms are removed by anthelmintics. Living Chabertia stimulate high levels of reaginic antibody, which is probably linked to cell-mediated immunity. It is almost impossible to control Trichuris in the environment and only in intensive situations where floors can be cleaned daily, is environmental control possible. No anthelmintic resistance is known for any of the worms in the large intestine. Select a suitable anthelmintic using the process discussed under the control of abomasal worms in small stock. Encourage “good practice ”by monitoring the effect of anthelmintics, ensuring refugia, and using environmental controls.
Liver
Apart from the trematodes that migrate through the liver on their way to their predilection sites, the larvae of Taenia hydatigena, Echinococcus granulosus, and Echinococcus multilocularis also cause significant damage. Various Schistosoma species may be found in the blood vessels of the liver. The pentastome genus Linguatula bores tunnels of about 1 mm in diameter in the parenchyma. Nematode larvae, mostly ascarids, may occasionally be found.
Tapeworms
Taenia hydatigena
Echinococcus granulosus
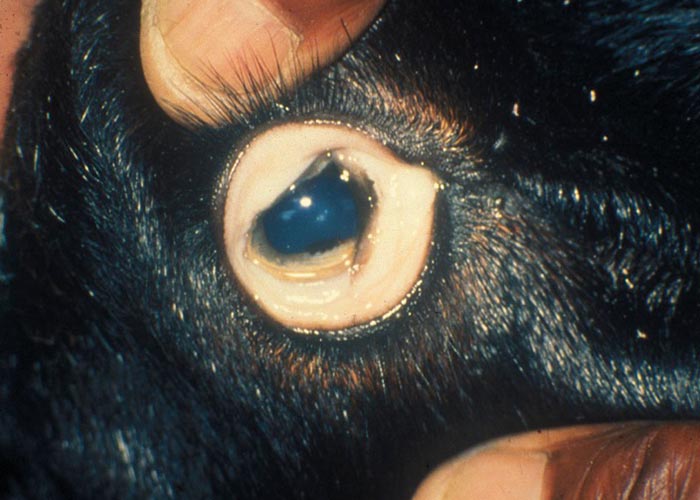
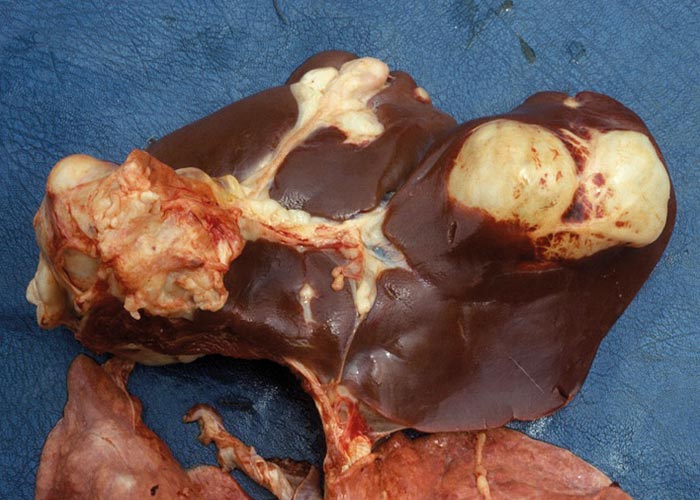
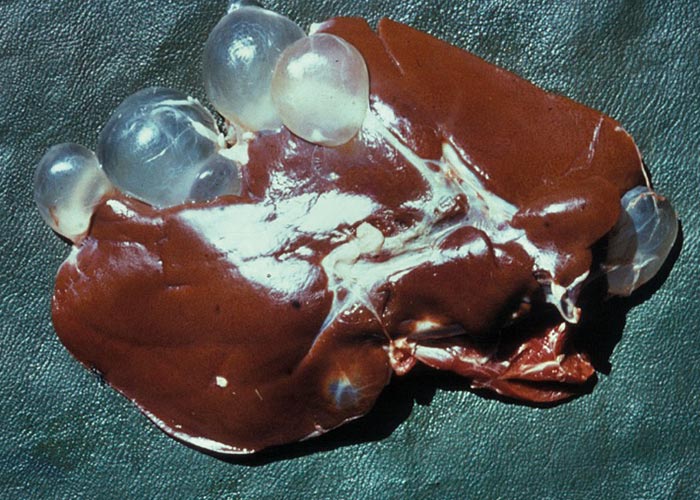
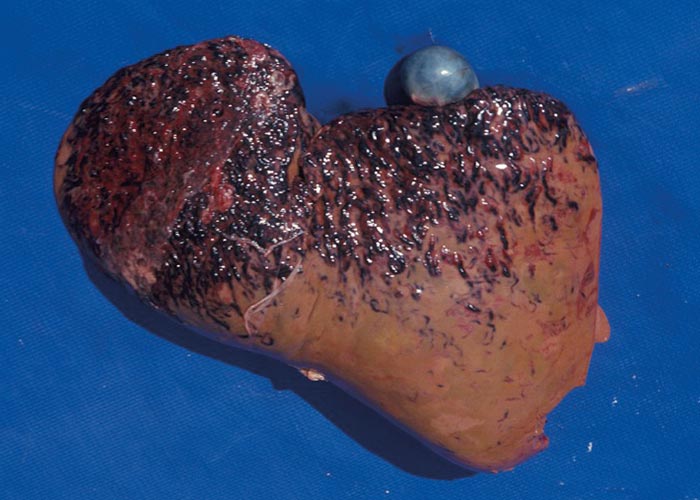
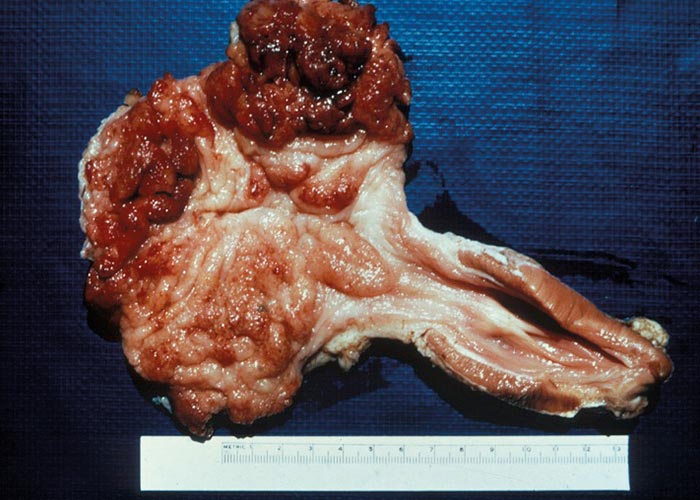
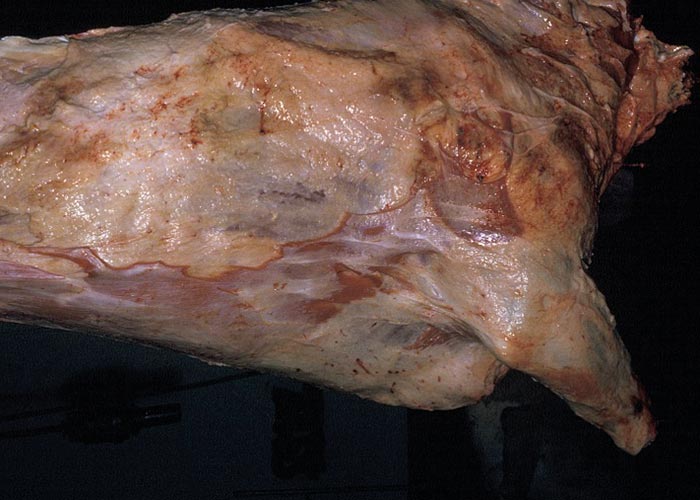
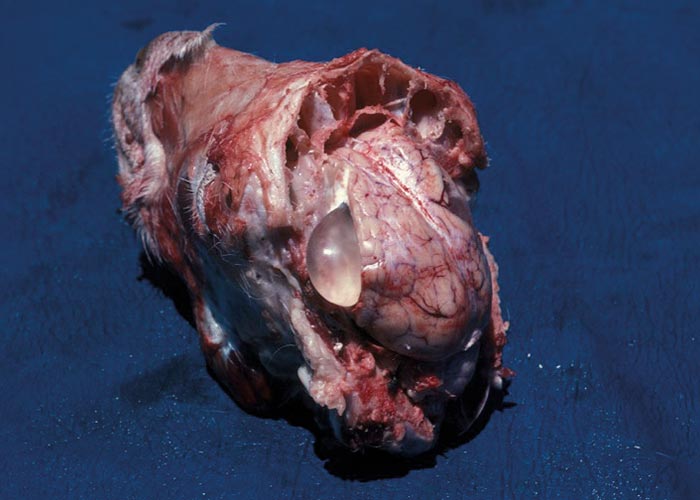
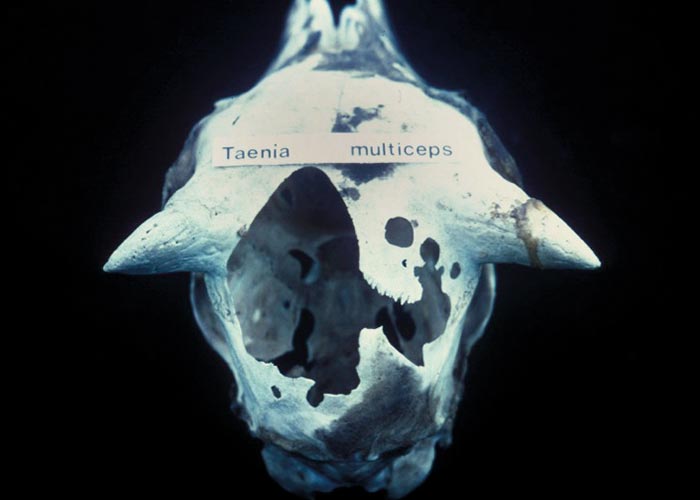
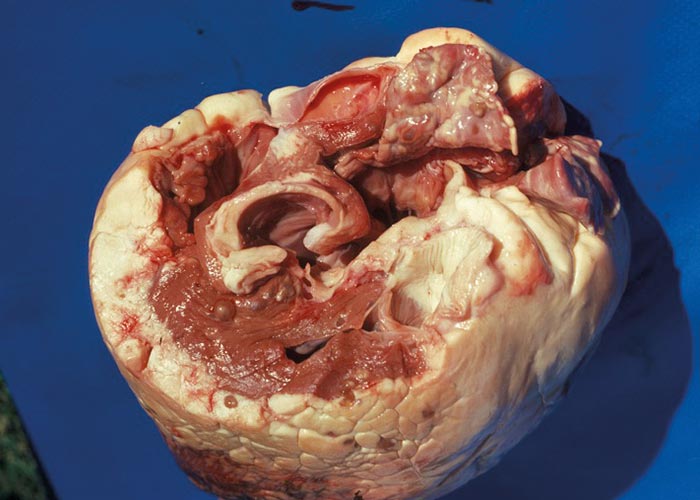
Ruminants are also the intermediate hosts for some tapeworms –of which Taenia hydatigena and Echinococcus granulosus are possibly the most commonly encountered in Africa. The pentastome genera Linguatula and Armillifer are also occasionally found, but their prevalence seems to be higher in wild animals. Eggs of Taenia are eaten with the food and the hexacanth larvae (oncospheres) hatch in the small intestine. These burrow through the wall of the intestine, and, depending on the species, are transported by the blood to the liver, lungs, heart, or striated muscles. In the case of T. hydatigena , the larvae migrate in the liver for about four weeks –before emerging and attaching to the peritoneum. Here they develop to cysticerci up to 8 cm in diameter –the socalled Cysticercus tenuicollis ( Figures 27 and 28 ). The life cycle of E. granulosus is similar. Oncospheres are carried to the liver by the blood or to the lungs by the lymph –the two commonest sites for development –where they form hydatid cysts ( Figure 26 ). The eggs of the pentastomes are also ingested and the primary larvae hatch in the small intestine. In the case of Linguatula , the larvae moult several times to form infective nymphs that keep migrating through the body, and these are often found free in the liver or heart.
Distribution
Cysticercosis is common throughout the world wherever sheep and goats occur, and wherever the cestode Taenia hydatigena is present. Somalia, Sudan, Uganda, Kenya, Nigeria and the Ivory Coast have a prevalence of more than 10% of cystic echinococcosis in domestic stock. In Ethiopia, the Central African Republic, Tanzania, Madagascar, Mozambique, Zambia and South Africa, the prevalence is 1 to 10%, and in Malawi, Namibia, Botswana, Zimbabwe, Angola and the Democratic Republic of the Congo it is less that 1%. It has not been recorded from Benin and Gabon (FAO, 1993). Pentastomes are widely spread but do not occur in large numbers, and are therefore seldom encountered.
Hosts
The definitive hosts of Taenia hydatigena are domestic dogs and other canids and the intermediate hosts are primarily sheep and goats –although cattle and camels are infected on occasion. The definitive hosts of Echinococcus granulosus are domestic dogs and cats and a variety of wild carnivores, e.g. coyote, dingo, wolf, Cape hunting dog, and other canids. The list of intermediate hosts comprises ruminants –including camels, suids, horses, and rodents. Humans are accidental, but entirely suitable, intermediate hosts. The pentastome genera Linguatula and Armillifer use ruminants as an intermediate host. As a final host, Linguatula parasitises the nasal sinuses and respiratory passages of carnivores, while Armillifer is a parasite of the lungs of snakes.
Transmission
In all cases, the acquisition of larval cestodes occurring in the liver is through ingestion of eggs by the intermediate host. Eggs are dispersed in the environment by various agents –notably insects and rain. Adult worms develop after ingestion of the metacestodes by the final host, and in the case of the pentastomes, the infective nymph.
Socio-economic importance
The socio-economic influence of cysticercosis is considerable. Few animals die as a result of acute infection, but chronic infection leads to condemnation of the liver. The cysticerci on the outside of the liver are often just trimmed away and fed to dogs, which further compounds the problem. Cystic echinococcosis has a severe influence on those populations –human or animal –where it occurs, and cost is only one of them. Cost can be categorised as the cost due to the disease in humans (e.g. medical and non-medical costs, lost productivity), the cost due to the disease in animals (e.g. condemnation of edible organs, destruction of rejected organs, reduced yield of milk, meat, wool and hides, and reduced number of viable offspring), and the cost of control programmes (e.g. personnel, equipment, travel, surveillance, education, drugs and destruction of animals). Humans are occasionally infected with pentastomes –but to what extent, is unknown.
Pathogenesis
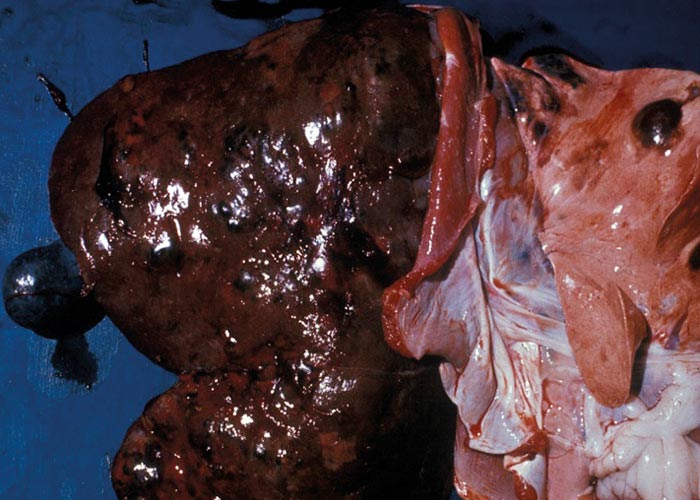
In sheep and goats the hexacanth larvae occasionally cause a severe hepatitis during the four weeks they migrate through and grow in the liver. This resembles acute fasciolosis, and death occurs due to haemorrhage into the abdominal cavity. Once they have attained a certain size, they leave that organ and attach to the peritoneum –usually close to the liver. After another four weeks they have become large, flabby structures –Cysticercus tenuicollis –which are filled with fluid through which the protoscoleces can easily be seen ( Figure 27 ). Hydatids in the liver and lungs are well tolerated and most infections are discovered at necropsy. Where the oncospheres have been carried in the blood to other organs, a variety of clinical signs may present, depending on the organ in which the oncospheres lodged. Pentastomes are also well tolerated, and even with heavy infections there is little haemorrhage into the abdominal cavity.
Clinical signs
In most cases infection with cestode or pentastome larvae goes unnoticed and is detected only at meat inspection or necropsy. Infrequently, however, large numbers of developing cysticerci migrate in the liver, causing hepatitis cysticercosa. This is a condition that is similar to acute fasciolosis and is often fatal (refer to pathogenesis and pathology). Once the cysticerci have developed, there are no clinical signs. Cystic echinococcosis refers to an infection with hydatid cysts, and is a common occurrence in Africa. As a clinical entity, it is rarely suspected –but is frequently diagnosed at necropsy. Pentastomes often encapsulate in the liver or peritoneum of ruminant intermediate hosts, but no clinical signs have been ascribed to them.
Pathology
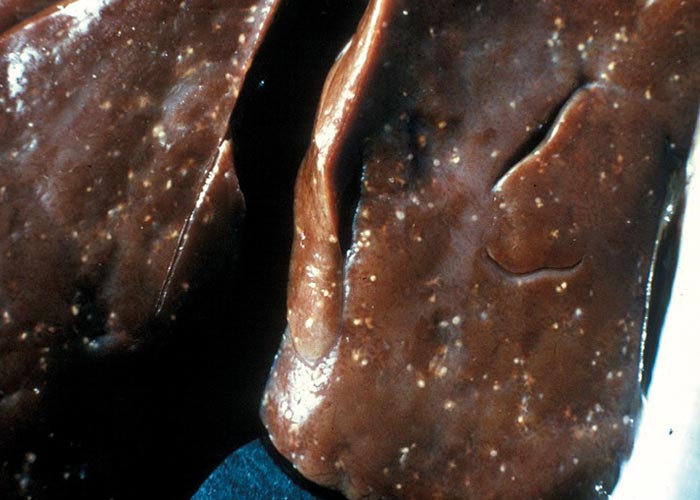
Acute hepatitis cysticercosa in sheep is characterised by an enlarged liver in the parenchyma in which numerous migration tracts are seen. The surface of particularly the ventral lobe is covered with a fibrinous exudate. Subcapsular haemorrhages are common, and these may rupture so that blood is found in the abdominal cavity. Acute hepatitis cysticercosa is seldom seen in cattle. Depending on the number of oncospheres invading, there may or may not be significant damage. In the former case, it will resemble chronic fasciolosis and the large cysticerci can be seen on the peritoneum or close to the gall bladder. In the case of cystic hydatidosis, the lesions are associated with pressure –which in the different organs (i.e. liver, lungs, long bones and others) may cause different clinical signs and gross pathological lesions.
Diagnosis
Acute hepatitis cysticercosa resembles acute fasciolosis –in its sudden onset and rapid death. Chronic cysticercosis is usually only diagnosed at the abattoir or at necropsy, as is infection with pentastomes.
Cystic echinococcosis in humans can be diagnosed by various imaging techniques, e.g. X-ray, ultrasound, CT scan or magnetic resonance, or by serological methods –of which a whole battery is available. Commonly used methods are ELISA tests, immuno-electrophoresis and complement fixation. In animals the diagnosis of cystic echinococcosis is seldom called for, and is usually only diagnosed at the abattoir or at necropsy. The diagnosis of infection in dogs is difficult because of the small size of the tapeworm and few proglottids are shed. In some countries a purgative like arecoline hydrochloride is administered –which results in the expulsion of the entire tapeworm.
Differential diagnosis
Differential diagnoses for acute deaths from acute hepatitis cysticercosa would include acute fasciolosis.
Control
Cysticercosis and cystic hydatidosis are both relatively easy to control, but the lack of knowledge in many parts of Africa still makes for large numbers of infected animals and humans. Firstly, dogs should never be fed uncooked offal –this may contain the cysticerci or hydatid cysts. Secondly, humans should wash their hands thoroughly before eating or drinking, especially if they have been playing with dogs or gardening. Thirdly, where possible, dogs should be dewormed using praziquantel and the faeces disposed of so that the tapeworm eggs cannot contaminate the pastures where ruminants graze. Fourthly, where dogs are found positive for Echinococcus , contamination of vegetables and water is possible, and therefore everything, including the water, should be boiled before consumption. It is not possible to control the pentastomes, since the final hosts are either wild carnivores or snakes.
Liver flukes
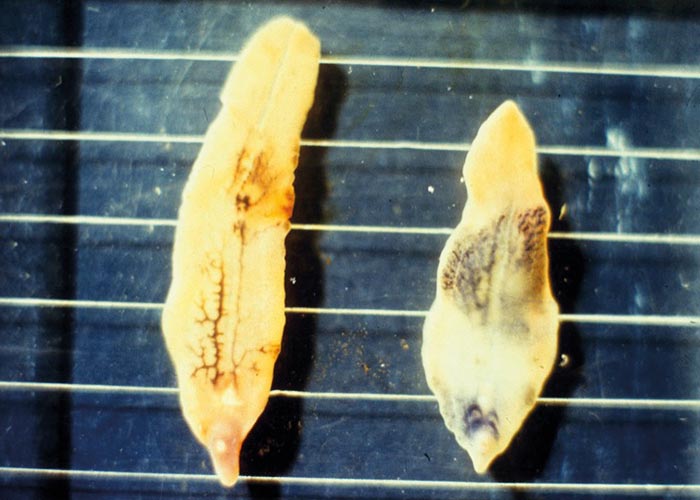
Fasciola hepatica
Fasciola gigantica
Although many helminths pass through the liver during their development in their respective hosts, few adult parasites occur here. The major trematode genera are Fasciola, Fascioloides, and Dicrocoelium. Eurytrema occurs mainly in the pancreatic duct, and more rarely in the bile ducts. The only adult cestode found here is Stilesia hepatica. Apart from the trematodes that migrate through the liver on their way to their predilection sites, the larvae of Taenia hydatigena, Echinococcus granulosus and Echinococcus multilocularis also cause significant damage. Various Schistosoma species may be found in the blood vessels of the liver. The pentastome genus Linguatula bores tunnels of about 1 mm in diameter in the parenchyma. Nematode larvae –mostly ascarids –may occasionally be found.
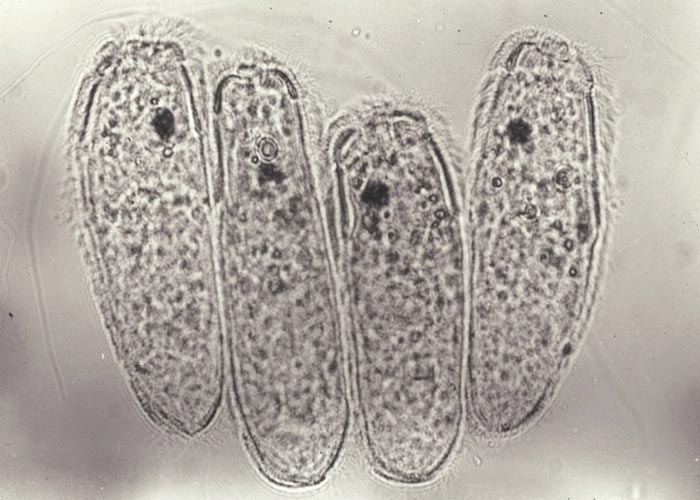
Life cycle
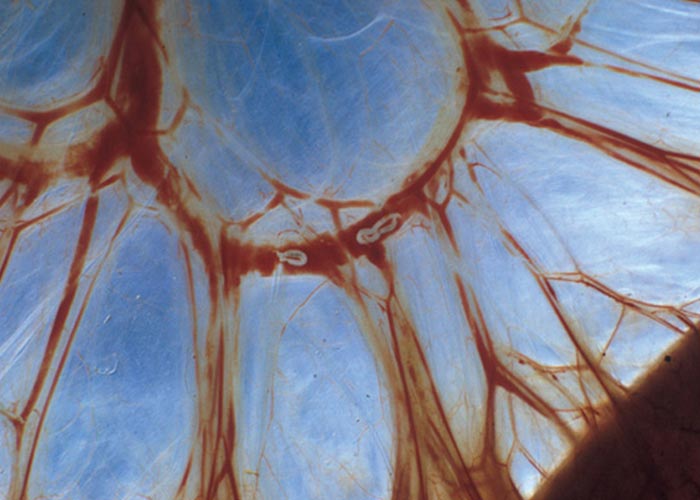
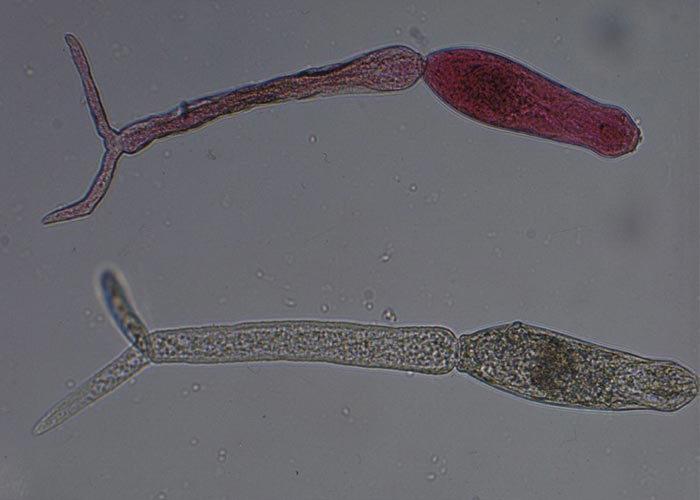
The trematodes have indirect life cycles involving either a terrestrial or an aquatic snail –and in some genera an arthropod intermediate host. The Fasciola life cycle starts with an egg that contains a miracidium ( Figure 30 ) and which must fall in a wet or damp area in order to release the miracidium ( Figure 29 ). The miracidium must enter a suitable snail within three hours of hatching from the egg. In the snail, development proceeds through the sporocyst and redial stages to cercariae that are the final stage of development in the intermediate host. The motile cercariae are shed, and attach to any solid object in the water like vegetation, dead leaves and even twigs and dead insects –where they encyst to the metacercariae. The latter are infective for the final host. Once ingested by the final host, the metacercariae excyst in the small intestine, pass through the intestinal wall, cross the abdominal cavity, and then penetrate the liver. The young flukes tunnel through the parenchyma and enter the small bile ducts after 6 to 8 weeks. They migrate to the larger bile ducts where they develop to adults ( Figure 31 ). The prepatent period of F. hepatica varies from 8 to 13 weeks or 10 to 12 weeks, and that of F. gigantica from 11 to 16 weeks.
The life cycle of Dicrocoelium is different in that a second intermediate host is utilised. The egg –which contains a miracidium –must be eaten by a terrestrial snail in order to hatch. Two generations of sporocysts develop, which then produce cercariae, but there are no rediae. The cercariae are extruded in masses cemented together with mucus. The slime balls of cercariae are ingested by an ant of the genus Formica (D. dendriticum) or Campanotus (D. hospes) in which they develop to metacercariae –usually in the haemocoel, but occasionally in the brain of the ant. Those ants with metacercariae in the brain show aberrant behaviour and climb to the top of vegetation and remain there even after “normal ”ants have returned to their nest. This presumably increases the possibility that infected ants will be eaten by the final host. The metacercariae excyst in the small intestine and the young flukes migrate up the main bile duct into the smaller ones, where they attain sexual maturity. The pre-patent period is 10 to 12 weeks.
The life cycle of Eurytrema is similar, in that a terrestrial snail is the first intermediate host and a tree cricket or grasshopper the second. The metacercariae hatch in the small intestine and enter the pancreatic duct; occasionally the main bile duct is entered and the flukes are found in the smaller bile ducts in the liver. Stilesia hepatica are shed with the faeces, and a soil mite –probably of the family Oribateidae –is used as an intermediate host. A cysticercoid develops in the mite and the mite must be eaten by the final host for the life cycle to continue. In the final host, the cysticercoid evaginates its scolex and moves to the opening of the common bile duct, through which it enters to lodge in the very small bile ducts.
Distribution
In Africa, the major helminths infecting the liver are the trematodes Fasciola hepatica , Fasciola gigantica and Dicrocoelium hospes . In Europe and North America Fascioloides magna is fairly common, while Dicrocoelium dendriticum is widespread. The genus Eurytrema occurs in Brazil and Asia. The small cestode Stilesia hepatica occurs worldwide; it is also common in ruminants in Africa, except cattle. A prerequisite, which ultimately determines the distribution of the trematodes, is the presence of suitable snail intermediate hosts for the trematodes, and a suitable arthropod for Stilesia. The snails can be terrestrial (Eurytrema, Dicrocoelium) or aquatic (Fasciola, Fascioloides) , and, in some cases, a second intermediate host –usually an arthropod –is required. Stilesia requires one of the soil mites, probably of the family Oribateidae.
Hosts
The main hosts of trematodes and the cestode are ruminants. Sheep, goats, cattle and a variety of wild ruminants are affected, but often not to the same degree. Dicrocoelium is present in rabbits and in ruminants, and Stilesia is rare in cattle.
Transmission
In all cases the transmission of helminths parasitising the liver is through ingestion of metacercariae (the trematodes) or cysticercoids (Stilesia) that occur either on vegetation or in an intermediate host. The epidemiology of the snails is also important for the transmission of the parasites. Some snails are aquatic, others are semi-aquatic, and suitable habitats must be present. The aquatic snail Lymnaea truncatula prefers wet mud to free water, and temporary habitats like hoof marks and rain ponds that develop after heavy rain or excessive irrigation, are colonised. Temperatures in excess of 15 ºC are required for the multiplication of both the snails and the developing stages of the parasite in the snails ( Figure 35 ). During winter, the snails hibernate and the development of the fluke larvae is halted. Development resumes when temperatures rise and metacercariae are available during spring. There is some evidence that the prevalence of fasciolosis in warm countries is higher after several months of drought –possibly because animals congregate around watering places, so increasing the chances of snails becoming infected. Rainfall should exceed the transpiration rate of the vegetation in order for the eggs to develop, the miracidia to find the snails, and the cercariae to disperse after having left the snails.
Socio-economic importance
The socio-economic influence of trematode infection of the liver –and, in particular, fasciolosis –can be considerable. Many animals die as a result of infection, and in developing countries this has a major impact on the cash flow and economics of a region. During the acute phase of infection, the immature flukes burrow through the parenchyma and their rapid growth during this period causes substantial damage –especially just before they enter the bile ducts. Depending on the number of flukes present, this causes severe haemorrhage and often the death of the animal. Similarly, the fibrosis in the parenchyma and thickened bile ducts seen in chronic cases lead to condemnation of the liver, which is a further economic loss. It is estimated that 37% of sheep livers are condemned in South Africa because of Stilesia infection. Worldwide, the estimated loss due to fasciolosis is US$2 billion.
Humans are occasionally infected with liver fluke, but it seems to be under-reported in the literature. Recent estimates, based on serodiagnosis, indicate that as many as 17 million people in the world suffer from fasciolosis. In humans, clinical manifestations appear early in the infection, long before parasitologic diagnosis is possible, and the symptoms and clinical signs are as severe as they are in ruminants.
Pathogenesis
The pathogenesis of fasciolosis depends on the host species, the stage of the infection, and the number of infecting flukes. In field situations, animals acquire infections over an extended period, so that flukes of all developmental stages are in the liver. This results in clinical signs of both acute and chronic disease. The passage of the young Fasciola species through the intestinal wall into the peritoneal cavity causes little damage, and the changes that do occur are mostly localized and transient. Once in the liver the young flukes burrow through the parenchyma creating tracks or tunnels, and this is the acute stage. Blood vessels and small bile ducts are damaged –resulting in haemorrhage, thrombosis and disruption of the flow of bile. During their migration, the young flukes feed on hepatic cells and blood, and also secrete proteolytic enzymes. They grow rapidly, causing even more damage, especially during the period just before they enter the bile ducts. Haemorrhage may become severe in the late acute stage, and may cause the death of the host either through haemorrhage into the abdominal cavity or beneath the capsule of the liver ( Figure 32 and 34) . If the host survives, hepatic fibrosis and hyperplastic cholangitis develop. The chronic stage develops slowly and is the result of the presence of the fluke in the bile ducts, where they become sexually mature. Chronic fasciolosis is a combination of cholangitis, biliary obstruction, destruction of hepatic tissue, and the release of a haemolytic substance by the fluke.
The tracts in the parenchyma heal and connective tissue forms in and between tracts. Foci of coagulative necrosis form parallel and adjacent to the healing tracts –which, in turn, also eventually heal by fibrosis ( Figure 33 ). A peri-biliary fibrosis occurs throughout the liver, even around the small bile ducts where there are no flukes. Lastly, monolobular fibrosis develops and the portal canals become linked together by fibrous tissue. In addition, there are changes in the blood supply to the liver. The inlet venules that arise from the portal vein are occluded, and this causes disruption, degeneration and necrosis of the hepatic limiting plate. Bile-duct proliferation occurs with accompanying fibrosis around the portal canals that surround the hepatocytes and small bile ducts. Eventually, phlebitis of the portal vein develops, which leads to oedema and fibrosis, and the occlusion of the vein. Adult Fasciola in the bile ducts cause erosion and necrosis of the biliary mucosa and intense inflammation of the adjacent lamina propria. These changes, however, are less important than the anaemia caused by the adult flukes –which will eventually lead to an iron deficiency. Migration of young Fasciola, and in some areas Dicrocoelium , through a liver that contains spores of Clostridium novyi type B may cause infectious necrotic hepatitis (black disease) to develop in sheep and cattle. Dicrocoelium dendriticum and D. hospes are of low pathogenicity –depending on the number of flukes infecting. In heavy infections, there is a lymphocytic infiltrate around larger bile ducts, which is sometimes accompanied by fibroblastic proliferation around portal ducts, veins and arteries. There is slight hyperplasia of the bile ducts that continues as the infection progresses.
Eurytrema pancreaticum rarely invades the bile ducts of cattle, and its pathogenesis is unknown.
Fascioloides magna is a large, broad fluke that causes little harm in the Cervidae, its natural host. Early infections involve portal areas with extensive zones of fibrosis and an extremely pronounced influx of eosinophils. The flukes encyst rapidly once in the liver parenchyma, and the cyst expands as the fluke grows in size. However, the fibrosis is such that the afferent and efferent bile ducts still have access to the cyst cavity –thereby allowing the fluke eggs to be released into the intestine. In cattle, and in heavy infections in deer, the marked tissue reaction to the fluke produces large fibrous cysts up to 40 mm in diameter. The worms are contained in the cyst, which is completely occluded. Eggs are therefore not found in cattle. The cyst and tissues adjacent to it are black due to extensive pigmentation. The pigment is a haeme compound, which is believed to be produced by the fluke.
Sheep and goats show little host reaction to the flukes, and cysts do not form. This indicates that sheep are not natural hosts. The flukes continue migrating through the parenchyma, destroying it in the process, and leaving black, tortuous tracts. The presence of this fluke in certain areas has made it impossible to farm with sheep.
Clinical signs
Acute fasciolosis occurs when animals consume 2000 or more metacercariae –and manifests as dullness, weakness, lack of appetite, pallor and oedema of the mucosae and conjunctivae, and also pain when pressure is applied over the area of the liver. Deaths occur quickly, often in less than 48 hours, and may be accompanied by blood-stained discharges from the nostrils and anus. Outbreaks are more common in young sheep and are of short duration; most deaths occur within 2 to 3 weeks. Fluke eggs are not found in the faeces.
Subacute fasciolosis results when animals ingest 500 to 1500 metacercariae over an extended period. Some flukes have already reached the bile ducts, where they cause cholangitis. Others are still migrating in the parenchyma where they cause less severe, but similar lesions as are seen in the acute disease. Sheep show clinical signs for 1 to 2 weeks prior to death. There is rapid loss of condition, pallor of the mucous membranes, and an enlarged and palpable liver. Chronic fasciolosis manifests as gradual wasting, severe anaemia with ascites, oedema, bottle jaw, and very high fluke egg counts. Sheep have diarrhoea and shed their wool. The course of the disease is often as long as 2 to 3 months in those animals that die, but many survive and remain emaciated for long periods.
Although acute and subacute clinical disease occasionally occur in heavy infections –especially in calves –the chronic form is, by far, the one most often encountered in cattle. Aberrant migration is more common and flukes are often seen in the lungs. With heavy burdens there is anaemia and bottle jaw, but with smaller burdens the clinical signs are minimal. Diarrhoea is not a feature of fasciolosis in cattle.
The large liver fluke, Fascioloides magna, wanders through the liver parenchyma of cattle for a short while, whereafter it is encapsulated and does little harm. However, in sheep it never gets encapsulated and keeps on wandering through the liver parenchyma –causing severe clinical signs and death.
Infection with Dicrocoelium is usually chronic. The clinical signs are similar to those produced by Fasciola, but are much milder since the flukes do not destroy the liver parenchyma to the same extent. Because of their small size, Dicrocoelium may be found in the smaller bile ducts.
Stilesia is not known to produce clinical signs.
Pathology
An enlarged liver –in the parenchyma of which numerous migration tracts are seen –characterises acute fasciolosis in sheep. The surface of particularly the ventral lobe is covered with a fibrinous exudate. Subcapsular haemorrhages are common, and these may rupture so that blood is found in the abdominal cavity.
Acute fasciolosis is seldom seen in cattle. The lesions of subacute fasciolosis are similar to those of acute fasciolosis, but rupture of subcapsular haemorrhages is rare; this form of the disease is seldom seen in cattle.
With chronic fasciolosis, the liver has an irregular outline, and is firm and pale. The ventral lobe is most affected and is smaller. Hepatic fibrosis and hyperplastic cholangitis are commonly seen. Several types of fibrosis are present. Post-necrotic scarring is associated with the healing of migration tracts. Ischaemic fibrosis is a sequel of infarction caused by damage to and thrombosis of large vessels. Peribiliary fibrosis develops when the young flukes reach the small bile ducts and monolobular fibrosis develops –which can be seen as white strands of fibrous tissue demarcating the hepatic lobule. Chronic fasciolosis is often seen in cattle. The pathology is essentially the same as that in sheep. In addition, the bile ducts tend to calcify and the gall bladder becomes enlarged. Aberrant migration of the flukes is more common in cattle, and encapsulated parasites may be found in the lungs.
The lesions caused by both acute and chronic Fascioloides infection in cattle are milder than those of Fasciola . In sheep, lesions are severe, as the flukes are large, and, as they never encyst as in cattle and deer, they destroy the liver parenchyma. The lesions are also discussed in the section on pathogenesis.
Dicrocoelium does not –as a rule –cause serious lesions to the liver, even though a few thousand may be present. In heavier infections, fibrosis of the smaller bile ducts and extensive cirrhosis can occur. Sometimes the bile ducts become markedly distended.
Although large numbers of Stilesia can often be found in the bile ducts, they cause neither clinical signs nor significant pathology.
Diagnosis
Acute fasciolosis occurs when the flukes are still migrating, and eggs will therefore not be found. Animals often die before any clinical signs are apparent, and those surviving will be severely anaemic (refer to clinical signs for more diagnostic features).
A diagnosis of chronic fasciolosis is made by examining the faeces for eggs. The eggs of F. gigantica are larger than those of F. hepatica –but both have a golden colour imparted by the bile. Liver function tests –particularly GLDH (glutamate dehydrogenase) and GGT (gamma glutamyl transpeptidase) –can give an indication of liver damage, which may be due to fasciolosis. Antibodies of the flukes may be detected in serum or milk with an ELISA (enzyme-linked immunosorbent assay) test and the passive haemagglutination test may also be used.
Differential diagnosis
Differential diagnoses for acute deaths from fasciolosis would include acute hepatitis cysticercosa, acute haemonchosis, anthrax, enterotoxaemia, infectious necrotic hepatitis or black disease (Clostridium novyi) –and for chronic cases Senecio and other hepatotoxic plant poisonings and chronic haemonchosis.
Control
Environmental control and reducing exposure: Decreasing exposure to liver fluke and reducing pasture contamination are critical for limiting liver fluke infestations. This can be done by reducing snail populations through drainage of water-logged areas to eliminate snail habitats. Draining is the best long-term method for managing aquatic snail habitats but may not be feasible due to cost implications. Waterlogged areas can also be fenced off, which prevents animals from grazing in liver fluke-infested pastures during high risk times of the year when the snails are active. Animals should ideally be withheld from high risk pastures during times of peak snail activity ( see Figure 35 ). Vegetation at the edges of standing water can be cut away to make the environment less suitable for snails.
It is important to fix leaking water troughs, as water that accumulates around them provides an ideal environment for aquatic snails to thrive –so increasing the risk of exposure to liver flukes.
Anthelmintics used at carefully chosen periods will lower the number of flukes present in the host, thus reducing pasture contamination. Animals newly introduced into a farm should be quarantined and dewormed with a product which is effective against immature and adult flukes in order to avoid introduction of flukes into uninfected farms.
Strategic control: Strategic control is best applied during late winter (early spring) and autumn (April/May).
Strategic treatment during late winter –when the intermediate host is inactive –is aimed at killing adult flukes present in the liver. The aim is to prevent infection of the intermediate host during the ensuing spring. Animals can be dosed with products containing closantel, nitroxynil, rafoxanide, oxyclozanide or triclabendazole.
Strategic treatment in autumn (April/ May) is aimed at clearing animals of both immature and mature liver fluke stages before winter. The active of choice for strategic treatment in autumn is triclabendazole which should be given 2 weeks after the first frost has occurred or where the minimum temperature is maintained below 10 °C.
Tactical control: Tactical control is aimed at reducing clinical disease and production loss. It is implemented during periods when there is heavy infestation of the pastures. This is to manage the contamination of immature liver flukes in high risk areas when environmental conditions are optimal for the survival of aquatic snails. Treatment during these periods is critical in order to avoid liver damage caused by migration of immature flukes. It is advisable to use a product containing triclabendazole –which is highly effective against immature liver flukes from 2 weeks of age ( Figure 36 ).
Treatment: Acute and subacute disease due to immature flukes may occur from August to May the following year when aquatic snails are most active. Triclabendazole is the treatment of choice, as it removes all developing stages from 2 weeks of age. A single dose of triclabendazole should be given –after which the animals should be moved to a well drained, fluke-free pasture.
Subacute fasciolosis occurs when a large number of immature flukes are present in the liver, but the disease is not rapidly fatal. The parasites will have developed further –resulting in a portion of the population being present as adults in the bile ducts. Triclabendazole is the treatment of choice as it kills all stages of liver fluke present in the animal. However, rafoxanide, closantel or nitroxynil may also be used in cases of subacute fasciolosis. It must however be kept in mind that these actives are not effective against the early, immature, liver fluke stages.
It is also advisable –in cases of subacute fasciolosis –to move animals to flukefree, dry pastures following treatment. In cases where animals cannot be moved to clean camps the treatment should be repeated after 4-8 weeks, in order to eliminate maturing flukes.
Chronic fasciolosis is characterised by the presence of adult flukes in the bile ducts. Mature flukes can be successfully treated with products containing closantel, nitroxynil, rafoxanide, oxyclozanide or triclabendazole. Movement to dry, flukefree pastures is important, as any eggs being eliminated will not hatch given that the environment is unfavourable –thus preventing reinfection of treated animals and infection of snails in water-logged areas.
Flukicides
Several products are registered for the treatment of liver flukes ( Figure 36 ). Most flukicides available are effective against adult flukes, with variable activity against immature stages.
Clorsulon is a benzenesulfonamide and is effective against adult stages of F. hepatica , and may be used for the treatment of immature flukes older than 10 weeks. Clorsulon is also effective against Fasciola gigantica and Fascioloides magna . Clorsulon is commonly combined with ivermectin for simultaneous treatment of F. hepatica and nematode infections.
Oxyclozanide is a narrow-spectrum flukicide that belongs to the salicylanide group and is mostly used in livestock. It is effective against adult liver flukes (F. hepatica) from the age of 10 weeks. Oxyclozanide is the only flukicide registered for use in lactating cows, as it has a zero-day milk withdrawal.
Albendazole belongs to the benzimidazole group and is effective against adult liver flukes (Fasciola hepatica and Fascioloides magna) from 10 weeks of age.
Nitroxynil belongs to the nitrophenolic group and is highly effective against adult stages of liver fluke and can be used to treat immature flukes from 6 weeks of age. Nitroxynil is also effective against a few gastrointestinal round worms like Bunostomum spp., Oesophagostomum spp., Haemonchus spp. and Parafilaria bovicola .
Closantel is the most used of the salicylanilides and is highly effective for the treatment of adult liver flukes. It can also be used to treat immature flukes from 6 weeks of age.
Closantel is well absorbed from either parenteral or oral administration routes –in both sheep and cattle. Closantel is also effective against H. contortus and nasal bots (Oestrus ovis) in sheep and other adult nematodes like Oesophagostumum spp., Bunostomum spp. and Ostertagia spp. in cattle and sheep.
Rafoxanide is a halogenated salicylanilide. It has been used extensively against fasciolosis and haemonchosis in cattle and sheep, but its principal use is as an adulticide against F. hepatica and F. gigantica .
Rafoxanide may be used for the treatment of early immature liver flukes from the age of 4 weeks, but is only considered to be highly effective against immature flukes older than 6 weeks. It is also indicated for the treatment of . contortus and Bunostomum spp., as well as nasal bots (Oestrus ovis) in sheep.
Triclabendazole is an anthelmintic flukicide belonging to the benzimidazole group. It is highly effective against adult as well as immature stages. Triclabendazole is the only flukicide effective for the treatment of early immature liver fluke larvae from 2 weeks of age. Treatment of the early stages plays a critical role in preventing liver damage caused by migrating larvae.
Cardiovascular system
The trematode helminths of the genera Schistosoma and Orientobilharzia occur in the cardiovascular system of ruminants. Several Schistosoma species –e.g. S. mattheei, S. margrebowiei, S. curassoni and S. leiperi –have been recorded from the mesenteric veins of cattle and sheep. More species occur in Asia, where domestic stock are the reservoir hosts for S. japonicum of humans. Four species of the related Orientobilharzia occur in livestock in Asia.
Two species of the nematode Elaeophora, E. sagitta and E. poeli occur in the coronary and pulmonary arteries of African buffalo, a number of tragelaphine antelope, and occasionally cattle (for illustrations see Helminths of game).
Table 5 Distribution of Schistosoma spp.
| Major species | |
|---|---|
| Schistosoma bovis | Africa, the Middle East and southern Europe |
| Schistosoma mattheei | Africa |
| Schistosoma japonicum | Far East |
| Minor species | |
| Schistosoma leiperi | Africa |
| Schistosoma spindale | Asia and the Far East |
| Schistosoma incognitum | India and Pakistan |
| Schistosoma mansoni | Africa, South America and the Middle East |
| Schistosoma nasalis | India and Pakistan |
| Schistosoma (syn Orientobilharzia) turkestanica | Asia |
Bilharzia or schistosomosis
Schistosoma species
The Schistosoma species –also known as blood flukes –inhabit a wide variety of host species, including ruminants. The life cycle is heteroxenous, and freshwater snails of the genera Biomphalaria, Bulinus and Physopsis are of particular importance in the transmission to sheep and cattle. The male and female flukes live in permanent association –the female being carried in the gynaecophoric canal of the male ( Figure 37 ). She lays eggs in the mesenteric venules, and, aided by their spines and proteolytic enzymes secreted by the miracidia, the eggs work their way through several layers of tissue to end up in the lumen of the intestine. They are then voided and must reach water for the miracidia to hatch, a process that only takes minutes. The miracidia enter the snail, change into primary and secondary sporocysts and eventually cercariae. The cercariae are furcocercous (forked tails) ( Figure 38 ) and usually enter the host percutaneously. Upon entering the host, the cercariae lose their tails and transform to schistosomulae. There are no redial or metacercarial stages. The schistosomulae reach the lungs via the bloodstream, and after four or five days migrate against the flow of blood to the liver. The flukes become sexually mature in the liver and then migrate to the mesenteric veins. The pre-patent period is 6 to 9 weeks.
The life cycle of Elaeophora is largely unknown, and it is assumed that an intermediate host is involved –probably a tabanid fly.
Distribution
Schistosoma species are widely distributed throughout Africa, the Middle East, Asia, and some countries bordering the Mediterranean –i.e. mostly in the tropics and subtropics –where suitable intermediate snail hosts are present ( Table 5 ).
Elaeophora contains five species. E. poeli and E. sagitta are common in Africa, and the remaining three occur in Europe and North America. Again, the distribution depends very much on the availability of a suitable intermediate host –a horsefly (Diptera: Tabanidae).
Hosts
The host-parasite list for the various Schistosoma species has been adapted from Urquhart et al. (1993). Each of the hosts can act as reservoir host for the other parasite species mentioned ( Table 6 ).
Table 6 Host species of Schistosoma .
| Major species | |
|---|---|
| Schistosoma bovis | Ruminants |
| Schistosoma mattheei | Ruminants and occasionally humans |
| Schistosoma japonicum | Humans and most domestic animals |
| Minor species | |
| Schistosoma leiperi | Cattle |
| Schistosoma spindale | Ruminants, horses and pigs |
| Schistosoma incognitum | Pigs and dogs |
| Schistosoma mansoni | Humans and wild animals |
| Schistosoma nasalis | Ruminants and horses |
| Schistosoma (syn Orientobilharzia) turkestanica | Ruminants |
Transmission
The transmission of Schistosoma is similar to that of Fasciola and Calicophoron , with all three genera being fully dependent on water as a medium for infection of both the intermediate and final hosts. Subclinical carriers release eggs in the water, and new hosts are infected when they wade or swim in the water. Elaeophora is thought to use a tabanid as intermediate host, and its distribution is therefore limited to the distribution of the fly. In South Africa, the fly concerned occurs in the extreme northern and eastern regions.
Socio-economic importance
Schistosomosis is invariably more serious in sheep than in cattle, and sheep die in large numbers. The disseminated granulomata, haematin pigmentation of the lungs, liver and kidneys that is so typical of schistosomosis, and also the emaciated carcass, causes considerable economic loss as the carcass has to be condemned. The effect on cattle is less, but some organs may have to be condemned because of the pathological changes. Since Elaeophora affects mainly the heart and the lungs, the socio-economic effects are not as dramatic. However, it is postulated that deaths in cattle do occur due to ruptured aneurysms or occluded coronary vessels.
Pathogenesis
The eggs work their way through the different layers of blood vessel walls, assisted by proteolytic enzymes secreted by the miracidium and the mechanical action of the spine during normal peristaltic motions of the bowel. The eggs form granulomas that can cause thrombosis of the portal vessels. Many eggs are drained through the portal system to the liver and the lungs, where they also form granulomas ( Figure 39 ). Adult worms are bloodsuckers with blind caecae –which causes them to periodically regurgitate digested and partly digested blood. Haematin is phagocytosed by the macrophages, and many of them end up in the lungs. The lungs of sheep infected with S. mattheei have a grey colour –which is highly indicative of the infection ( Figure 42 ). The adult worms cause considerable damage to the blood-vessel walls, and this may interfere with the normal flow of blood. Dead and dying worms are removed to the portal system. The worms accumulate in the liver on a temporary or permanent basis. This may lead to thrombosis, infarction, and death. Cercariae sensitise the host when penetrating the skin. Subsequent infections cause a severe allergic dermatitis –which traps and destroys the cercariae.
Clinical signs
The major clinical signs associated with acute intestinal schistosomosis in sheep are diarrhoea, sometimes haemorrhagic, anaemia, and also emaciation. These signs only develop after the onset of egg production by the trematode. Severely affected animals rapidly lose condition and usually die a few months after infection. Those less heavily infected develop chronic disease and their growth is retarded. Cattle seem to be more resistant, although the clinical signs in naive animals are often more severe than in sheep. Diarrhoea is severe, with large blood clots being present, eyes are sunken, and animals gnash on their teeth. Older cattle have an effective level of immunity against reinfection, but if they are challenged with massive doses of cercariae, the immunity breaks down and clinical signs resembling Matricaria nigellaefolia poisoning appear over a period of time. These signs include an anxious expression in the eyes, emaciation, knuckling over of the hind fetlocks, disorientation, constipation, and intermittent straining. Cattle tend to lean against any convenient object (“stootsiekte ”) ( Figure 40 ). It must be emphasised, however, that even in areas with a high prevalence, clinical disease in cattle is seldom seen.
Pathology
The worst lesions are produced by the presence of schistosome eggs in the tissues. These work their way through the different layers of the blood vessel walls and many eggs are drained through the portal system to the liver and the lungs, where they cause the development of granulomas. Haematin regurgitated by the worms is phagocytised by the macrophages and many of them end up in the lungs. The lungs of sheep infected with S. mattheei have a distinctive dark greyish colour, which is highly indicative of the infection. In both sheep and cattle the liver, intestine, bladder and kidneys, are greyish-brown ( Figure 41 ). In severely infected animals, atelectasis and emphysema may develop as a result of these granulomas. The adult worms cause considerable damage to the blood vessel walls, which may interfere with the normal flow of blood. Dead and dying worms end up in the portal system and accumulate in the liver, which in turn may lead to embolism, infarction and death. Cercariae sensitise the host when penetrating the skin. Subsequent infections cause a severe allergic dermatitis.
The lesions caused by Elaeophora are usually large and well defined and consist of aneurysms and a proliferative endarteritis in the coronary and pulmonary arteries. The endothelium of the aneurysms on the coronary vessels thickens somewhat –while that of the pulmonary vessels becomes hyperplastic and resembles cotton towelling.
Diagnosis
The diagnosis of schistosomosis is based mainly on the clinical-pathological picture. The demonstration of the characteristic eggs in the faeces and/ or in “squash ”preparations of blood from the faeces is useful in the early stages of the disease –but not in the later stages, as egg production decreases. When schistosomosis is suspected, it is best to perform a detailed necropsy, which will show the lesions, and, if the mesentery is inspected before removal, the numerous worms in the veins. The diagnosis of elaeophorosis is usually incidental, and worms are seen in the coronary and pulmonary vessels at necropsy or after slaughter.
Differential diagnosis
Coccidiosis, fasciolosis and Matricaria poisoning are possible differential diagnoses for cattle –and coccidiosis, fasciolosis and a variety of helminthoses for sheep.
Control
The control of schistosomosis is the same as that described for Fasciola and Calicophoron . The snail population varies according to the temperature and rainfall. Efforts should be made to determine the months during which time snail numbers are highest –when access to dangerous sections of water can be denied to the animals. Treatment should be carried out with care as dislodgement of the flukes causes them to act as parasitic emboli that drain to the smaller vessels of the portal vein: the so-called liver shift. Infarction of parts of the liver is possible, and in heavily infected animals treatment may have fatal consequences. The drugs still widely used are the antimonial compounds that have to be given over a period of days at a high dosage rate. Praziquantel at 25 mg/kg body mass is effective, although it may be necessary to give another treatment 3 to 5 weeks later.
Skin
The nematodes that occur in the skin, subcutis and adnexa of cattle, are Parafilaria bovicola, Onchocerca gibsoni, Onchocerca gutturosa, Onchocerca armillata, Setaria labiatopapillosa and Setaria digitata. Parafilaria occurs in the subcutis, O. gibsoni in nodules in the brisket or limbs, O. gutturosa in the ligamentum nuchae, O. armillata in the wall of the aorta, and Setaria in the abdominal cavity. A Stephanofilaria species has been reported from cattle in the northern parts of South Africa. Apart from Stephanofilaria dedoesi, which occurs in goats, helminths have not been recorded from sheep and goats. None of these nematode infections is life threatening, but they cause much damage to the hide –and carcasses have to be trimmed.
Parafilaria
Stephanofilaria
The nematodes that occur in the skin and adnexa belong to the superfamily Filarioidea. Parafilaria and Stephanofilaria belong to the family Filariidae, and Setaria and Onchocerca to the Onchocercidae. Parafilaria are slender white worms, 3 to 6 cm long, and they occur in the subcutaneous tissues. Anteriorly there are numerous cuticular papillae and circular ridges. The male has large pre- and post-cloacal papillae, and the spicules are unequal. The female has a bluntly rounded posterior end, and the vulva is situated near the mouth. Eggs are thin-walled, 0.04 to 0.05 mm long, 0.023 to 0.033 mm wide, and contain a fully developed first stage larva which is about 0.2 mm long.
The Onchocerca species are of little veterinary importance, but one species –Onchocerca volvulus –is of major concern in human medicine, as it is the cause of river blindness. The worms are 3 to 6 cm (males) to 14 to 50 cm (females) long, and are tightly coiled in nodules in connective or muscle tissue. Microfilariae are present in the skin.
The Setaria species in cattle are S. labiatopapillosa and S. digitata –and both occur free in the abdominal cavity. They are large worms, 4 to 12 cm long, and with complex cephalic structures and a cluster of spikes on the tip of the female tail. The male spicules are unequal. Sheathed microfilariae, 0.14 to 0.23 mm long, are produced and these are found in the blood. The Stephanofilaria species are small worms, 3.2 to 6.8 mm long –that live in tunnels in the dermis. Microfilariae are produced and these move to the dermal papillae. Depending on the species, microfilariae are 0.04 to 0.18 mm long.
Epidemiology and distribution
Parafilaria bovicola occurs in the warmer regions of the world –notably Africa, Asia and southern Europe. It has been introduced into Sweden where it is now well established. In South Africa it occurs in areas that receive an annual rainfall of 400-700 mm, have at least 120 frost-free days per year, and lie at an altitude of 800 to 1200 m. Setaria has a worldwide distribution wherever cattle are kept and mosquitoes occur. Onchocerca gutturosa occurs world-wide, O. gibsoni occurs in Africa, Asia and Australasia, and O. armillata in the middle East, Africa and India. There are five species of Stephanofilaria that parasitise domesticated ruminants. S. stilesi occurs in the USA, S. dedoesi in Malaysia, S. assamensis in India, Malaysia and Japan, S. zaheeri in Asia, and S. dinniki in Africa.
Hosts/reservoirs
Parafilaria bovicola is limited to cattle and water buffaloes, although a closely related species, Parafilaria bassoni , occurs in African buffaloes. Many species of Setaria are known and two species –S. digitata and S. labiatopapillosa –occur in cattle. All four Onchocerca species occur in cattle and most probably in African and water buffaloes. The Stephanofilaria species inhabit the dermis of African and water buffaloes, cattle, goats, the black rhinoceros, hippopotami, and elephants.
Transmission
All these nematodes have a heteroxenous life cycle –Diptera being the intermediate hosts. Musca lusoria and Musca xanthomelas, also known as face flies, are the main intermediate hosts of P. bovicola in Africa. In Europe, Musca autumnalis is the intermediate host. In South Africa, for Setaria , the mosquito genera Aedes and Culex are the intermediate hosts, while the various species of Simulium and Culicoides are the intermediate hosts for Onchocerca species.
The female Parafilaria in the subcutis bores a small opening through the skin to the exterior to lay thin-shelled eggs which contain a fully developed first-stage larva. A small amount of blood exudes from the wound –the so-called 'bleeding point' –to form the blood streaks that are clinically so typical of parafilariosis ( Figure 44 ). Flies are attracted to the blood and ingest the nematode eggs or first stage larvae. The larvae develop to the third or infective stage in several weeks to months, depending on the ambient temperature. Infection of cattle takes place through wounds or the mucous membranes of the eye, where the flies deposit the infective larvae. The pre-patent period is about 7 to 9 months, during which time the larvae migrate through the subcutis to eventually reach their predilection sites.
The haematophagous flies Simulium and Culicoides transmit the Onchocerca species. Unlike Parafilaria , these worms produce live first-stage larvae, known as microfilariae, that migrate to the blood vessels and end up in the capillaries. When the fly sucks blood, the microfilariae are ingested and develop to the third or infective stage in the fly. The infective worm larvae are transmitted to the new host when the fly feeds again. The Setaria species are transmitted in the same way. The culicine mosquitoes Aedes, Anopheles and Culex are the intermediate hosts.
Flies of the genera Lyperosia, Haematobia and Musca transmit Stephanofilaria . Since the infection is a superficial one and the worms occur in tunnels in the dermis, the flies readily access the first stage larvae. In the flies, the larvae develop to the third stage, and are then transmitted to another animal when the flies feed on a wound.
Socio-economic importance
The socio-economic effects of the helminths that affect the skin can be considerable. Not only does infection ruin the hides –but helminth migration in the subcutis (Parafilaria and Stephanofilaria) and the formation of nodules and abscesses (Onchocerca) may lead to excessive trimming of an otherwise good quality carcass.
Pathogenesis
The pathogenesis of parafilariosis consists of two phases: one during which the L3 migrates in the subcutis and attains maturity ( Figure 43 ), and, secondly, the egg-laying phase. Once the L3 have escaped from the proboscis of the intermediate fly host, they penetrate various tissues to eventually reach the subcutaneous tissue. Inflammation and oedema occur, and the migration tracts are filled with masses of eosinophils. Lesions are slimy to the touch, yellowish-green in colour, and resemble superficial bruising ( Figure 45 ). Lesions may extend into the inter-muscular fascia. When the gravid female punctures the skin, a small amount of blood exudes from the lesion and mats the surrounding hair. These lesions bleed only for a short time and then heal quickly.
Onchocerca , depending on the species, are associated with different changes.
O. gutturosa in the large ligaments is of little consequence, and O. gibsoni causes a fibrous reaction in the muscles.
The Setaria species are apathogenic. Dead worms may sometimes be encountered on the surface of abdominal organs where they become encapsulated and eventually become calcified there.
Lesions caused by Stephanofilaria appear about 2 weeks after infection. Small skin nodules are seen and these rapidly form papules that rupture. Papules may coalesce to form large areas that exude a haemorrhagic exudate.
Pathology
The macroscopic lesions that are produced by Parafilaria are the bleeding points and the discoloration of the subcutis and superficial muscle layers. Live or dead worms can be encountered under the skin. During the development period of the larvae, the discolouration is more yellowish to yellowish-green. The older lesions, once the female has produced a bleeding spot, are yellowish-brown to brown. The lesions are slimy with a characteristic coppery odour. Histologically, the lesions are characterised by a massive influx of eosinophils –accompanied by a mild to moderate oedema. In older lesions, pigment-laden macrophages are present in large numbers and moderate numbers of lymphocytes, some plasma cells, and also histiocytes are present.
The adult Onchocerca cause a chronic inflammatory response. This leads to the formation of dense fibrous nodules adjacent to the ligamentum nuchae or fascia next to the major bones of the limbs (O. gutturosa) or in the subcutaneous tissue of the brisket and hind limbs (O. gibsoni) .
Other than these, few lesions are caused by the infection.
Apart from the calcification of dead worms, no lesions are seen with setariosis. However, occasionally, the immatures of Setaria digitata cause cerebrospinal setariosis. See "Nervous system" for a detailed description.
The lesion caused by Stephanofilaria stilesi is located near the mid-ventral line, while the other species cause lesions in the skin on the sides of the neck, withers, dewlap, shoulders, and around the eyes. Irrespective of the species, the lesion is essentially a circumscribed focal dermatitis. Histologically, the nematodes are present in the dermis near the epidermis, and are surrounded by a zone of inflammation characterised by the presence of eosinophils, lymphocytes, some neutrophils, and histiocytes. Hyperkeratosis and parakeratosis can be seen and crusts of exuded serum and detritus collect on the surface. Sensitisation to, and death of, the worms causes a severe dermatitis.
Clinical signs
Clinical signs of parafilariosis are limited to actively bleeding spots which occur especially on the withers, but may also be present on the flanks, back and rump. These spots –referred to as bleeding points –appear during warm weather, in order to coincide with fly activity.
No clinical signs are apparent with setariosis. Onchocercosis presents as nodules and lumps of varying sizes in the brisket and external surfaces of the limbs.
Stephanofilaria in the skin usually presents as papules that erupt with a slightly bloody exudate. In the centre of the lesion, the skin tends to slough, and there usually is some hyperkeratosis around the edges. It can best be described as an exudative dermatitis that is often haemorrhagic.
Diagnosis
A diagnosis of parafilariosis is made on the presence of bleeding points during the warmer months of the year, and also during the warm hours of the day. A smear made from the blood that oozes out will –on microscopic examination –reveal thinwalled eggs and larvae.
The microfilariae of Onchocerca are concentrated where the intermediate hosts, Simulium and Culicoides , prefer to feed –which is the shady, ventral parts of the host. For diagnostic purposes, a piece of skin about 5 mm square, is removed and placed in warm saline. It is carefully teased apart to help the microfilariae emerge, and is then incubated for 6 hours or more. The microfilariae can be seen moving at the bottom of the container. The diagnosis can also be confirmed by making a blood smear in which microfilariae may be found.
Stephanofilaria adults and microfilariae are present in the skin lesions, but are difficult to find in skin scrapings. The diagnosis, therefore, is mostly presumptive and based on the site of the lesions. Removing a piece of affected skin and carefully dissecting the parasites from the tunnels they create in the dermis can reveal the presence of adult Stephanofilaria .
Differential diagnosis
Differential diagnoses for all of the helminths that affect the skin include injuries caused by thorns or barbed wire, contact with Euphorbia latex, tick and insect bites, abscesses, and focal proliferative neoplastic conditions of the skin.
Control
Parafilaria should be controlled by controlling face flies. This can be achieved by dipping with or applying pyrethroids –preferably deltamethrin –which has the best action against flies. The macrocyclic lactones are effective in killing the adult worms, but there is uncertainty about their efficacy against the immature stages. Cattle should not be slaughtered for at least 70 days after treatment, in order to allow the lesions caused by the worms to heal fully.
Since both Culicoides and Simulium are intermediate hosts of the Onchocerca species, it may be of value to control these flies. This can be achieved by destroying breeding places and by dipping animals. Occasional cases can be treated with one of the macrocyclic lactones –which seem to be quite effective.
Setaria is transmitted by mosquitoes (Aedes and Culex) and, theoretically, breeding places of the flies should be destroyed and cattle dipped with residual insecticides. Because the nematodes are non-pathogenic, however, control is seldom resorted to and routine tick control seems to reduce the incidence of the worms.
Stephanofilaria can be treated with the topical application of pyrethroids, macrocyclic lactones and levamisole.
Nervous system
Taenia multiceps
Echinococcus granulosus
With the exception of the larvae of Taenia multiceps , and rarely Echinococcus , no helminths primarily target the brain of ruminants –although several may end up there following aberrant migration. The final hosts of Taenia multiceps are dogs and wild carnivores, and the intermediate hosts are sheep, goats and man. When the oncosphere hatches in the intermediate host, it bores through the wall of the intestines and is carried by the blood stream to all parts of the body. In sheep, only those that settle in the spinal cord or brain will develop into the larval stage coenurus cerebralis. In goats the coenuri occur in the brain, in the muscles, and subcutaneously. In wild ruminants they predominantly occur subcutaneously.
Distribution
Coenurosis in South Africa occurs in Western and Eastern Cape Provinces, the Free State Province, and at two localities in Gauteng and Northwest Province (1979 data). The distribution in the rest of Africa is unknown –but it is assumed to be worldwide.
Hosts/reservoirs
Carnivores like the various species of jackals and foxes, and hunting and domestic dogs, are the usual hosts of Taenia multiceps –and the foregoing species and wild and domestic cats, of Echinococcus granulosus .
Transmission
Transmission to the intermediate host is by the faecal-oral route and the final host acquires the infection by eating the infective larval stage in the intermediate host.
Socio-economic importance
The socio-economic impact of this condition is moderate, as the overall infection rate of T. multiceps is about 1%. However, when valuable animals are affected, the situation changes considerably. The treatment of coenurosis and cerebral echinococcosis is expensive, and a successful outcome is not guaranteed. Certain cases are untreatable because of the localisation of the parasite.
Clinical signs
The presence of oncospheres in the central nervous system (CNS) cause a syndrome known as ‘gid ’or ‘turning disease ’; the clinical manifestations depend on the location of the coenuri in the brain ( Figure 46 and 47 ). Thus, circling, visual defects, peculiarities in the gait, hyperaesthesia, or paraplegia can be seen. Coenuri may grow so large that they cause pressure atrophy of the surrounding bone of the cranium or the vertebral column ( Figure 48 and 49 ).
Pathogenesis
The oncospheres are eaten by the intermediate host. When the oncospheres hatch, they bore through the wall of the small intestine and are carried by the blood stream to all parts of the body. Only those in the spinal cord and brain will develop into the larval stage, which is known as coenurus cerebralis. The coenuri grow slowly, taking months to mature. The pathogenesis of the lesions in the CNS is essentially that of gradual pressure atrophy and malacia of the nervous tissue –which progressively interferes with a particular cerebral function depending on where the cyst is located.
Pathology
The gross lesion of Taenia multiceps is compression of the nervous tissue and sometimes atrophy of the cranial bones –by the space-occupying larva. When mature the larva is easily seen, as it is large, often larger than 5 cm in diameter, and has a fluidfilled cyst bearing clusters of protoscoleces on its internal wall. The hydatid cyst of Echinococcus is as large as or even larger –but does not have the thin, transparent wall of a coenurus. Secondary lesions, which may not be present, include abrasions and other traumatic lesions incurred by the abnormal behaviour of the ruminant.
Diagnosis
The diagnosis is based on positive identification of the tapeworms in carnivores, and the clinical signs. The presence of taeniid eggs in the faeces of carnivores is of no particular value, as the eggs of the taeniids all look the same.
Differential diagnosis
Differential diagnoses would include many infective, toxic and traumatic conditions affecting the CNS.
Control
Control of these tapeworms depends entirely on preventing ruminants from consuming the taeniid eggs. This includes regular deworming of the carnivore final hosts where possible, feeding them only well-cooked offal, and fencing off areas contaminated with tapeworm eggs.
Surgical removal of the coenurus is possible only when it is on the surface of the brain.
Treatment
To salvage valuable animals the following treatment can be attempted: administer praziquantel 100mg/kg once (or 50mg/ kg twice). At the same time, give the normal dose of cortisone and aspirin 100 mg/kg, twice a day. Continue the treatment with the anti-inflammatory drugs for 5 days. Thirty six hours after having given the praziquantel, start giving a diuretic. Depending on the reaction of the sheep, give it at 12 or 24- hour intervals. Obviously, clean water should be available at all times and the animal should not be stressed in any way.
Muscle
Taeniosis occurs in humans after ingestion of under-cooked pork (Taenia solium) or beef (Taenia saginata) infected with metacestodes –the larval stages of these parasites. Pigs and cattle acquire cysticercosis following the ingestion of eggs or proglottids of T. solium and T. saginata respectively. Following the accidental ingestion of eggs, humans can get cysticercosis, or, more specifically, neurocysticercosis (NCC). (For more discussions on T. solium see under pigs).
Taenia saginata
Several species of can infect humans as definitive hosts –primarily T. saginata (the beef tapeworm) and T. solium (the pork tapeworm). In addition, the larval stage of other Taenia species (e.g. T. multiceps, T. serialis, T. brauni, T. taeniaeformis and T. crassiceps) can infect humans in various sites of the body.
Recently, a new species of taeniid tapeworm that appeared to be T. saginata was described from people in Asian countries –even though they had eaten only pork (the so-called Asian Taenia ). This organism is now known as T. asiatica , and has been found in Taiwan, Korea, China, Vietnam, and Indonesia. Differentiation between T. saginata acquired from beef and the Asian Taenia from pork, is difficult.
Morphology
The eggs of Taenia species are characteristically ellipsoid, approximately 0.04 mm in diameter, and with a thick, smooth radially striated embryophore. The egg contains a hexacanth embryo with larval hooks. Eggs of all the Taenia species look alike. Usually only a few eggs are found in the stools, but, more often, a few are found on the perianal skin.
Morphologically, the adult Taenia species can be differentiated on the number of sacculations arising from the uterus. In T. solium there are 7 to 15 lateral branches on each side, whereas in T. saginata there are 15 to 20 on each side. In addition, T. solium has a scolex with four suckers and a double crown of hooks (26 to 36 in number) of different sizes (small hooks of 0.12 to 0.15 mm and large hooks of 0.16 to 0.18 mm), and a narrow neck. The scolex of T. saginata is slightly larger and has four suckers, and no hooks. The adult of T. saginata generally is longer than T. solium –attaining an average length of 5 m. However, occasionally it may grow to more than 15 m.
Life cycle
The adult tapeworm of T. saginata occurs in the definitive host ’s small intestine. Proglottids –which contain eggs –break off the posterior end of the tapeworm, and are then voided with the host ’s faeces. They can be either intact or, if broken up in the host intestines, only eggs will be found in the faeces. The intermediate host becomes infected by the ingestion of eggs. Once ingested, the eggs hatch to release the hexacanth larva, which migrates through the intestinal wall to reach the blood and lymphatic systems –which carry it to the tissues, particularly the heart and other muscles of cattle, and the muscles and brain of pigs and/or humans. Here the larval stage develops to a cysticercus, with an invaginated scolex.
Epidemiology/distribution
Taenia saginata has a cosmopolitan distribution, with an estimated 50 million cases of infection reported annually.
Hosts/reservoirs
Taenia saginata uses cattle or related animals as intermediate hosts, although other animals such as camels, llamas and antelope may also occasionally be infected. Up to now, only humans have been described as a definitive host.
Transmission
Cattle are infected with T. saginata by ingestion of grass, water or soil which is contaminated with eggs that have likely been disseminated through the faeces of infected humans.
Socio-economic importance
T. saginata infections are seldom associated with clinical disease in both the intermediate and definitive host. Hence, socio-economic implications of the disease relate mainly to the costs of condemnation of contaminated carcasses, meaning that the impact can best be evaluated in terms of financial losses to the farmers.
Pathology
The cysticerci are commonly found in the heart muscle ( Figure 50 ) and relatively few occur in the skeletal muscles ( Figure 51 ). However, in either location they cause no pathology.
Clinical signs
Larvae: Infection with the larval stage of T. saginata (cysticercus bovis) is restricted to cattle –but is completely asymptomatic.
Adults: Most patients infected with T. saginata notice the passage of proglottids that are motile, more numerous, and larger than those of T. solium. The lesions of T. saginata infection are highly variable. Infections are often completely asymptomatic, but in some cases they may cause intestinal obstruction. Vitamin deficiency may be the result of excessive absorption of nutrients by the parasite –although this is more a feature of infection with the fish tapeworm Diphyllobotrium latum. Finally, a broad range of non-specific clinical signs such as abdominal pains, digestive disturbance, excessive appetite or loss of appetite, weakness, and loss of weight can accompany infection.
Diagnosis
Taeniosis
Microscopic identification of eggs and proglottids in faeces is diagnostic for taeniosis –but is not possible during the first 3 months following infection, prior to development of adult tapeworms. The intermittent nature of egg excretion often leads to underestimation of the prevalence of taeniosis. Repeated examination and concentration techniques will increase the likelihood of detecting light infections. Nevertheless, species differentiation of Taenia species is impossible based solely on microscopic examination of their eggs –because all Taenia species produce morphologically identical eggs. Eggs of Taenia species are also indistinguishable from those produced by cestodes of the genus Echinococcus (tapeworms of dogs and other canid hosts). Microscopic identification of gravid proglottids (or, more rarely, examination of the scolex) allows species determination. Immunodiagnostic tests for the differentiation of taeniid eggs were developed using either monoclonal or affinity-purified polyclonal antibodies in an immunofluorescent assay. These techniques were successfully applied to the identification of T. solium eggs. However, this relies on the ability to recover oncospheres, which leaves the issue of test sensitivity unresolved. The detection of parasite antigens in host faeces has been used for the diagnosis of taeniosis.
The following features were consistent across the different studies:
- Antigen detection in faecal samples is genus-specific, with T. saginata and T. solium both reacting in the assay, but with no cross reaction with other parasite infections –including the cestode Hymenolepis .
- Coproantigen detection is possible prior to patency of the worms, and the sensitivity of the tests is independent of the presence or the number of eggs.
- Coproantigens are no longer detectable within a week after treatment.
- Coproantigens are stable for weeks in unfixed faecal samples kept at room temperature, and for years in frozen samples or samples fixed in, e.g. formalin, and kept at room temperature.
These tests are available as a standard ELISA, as well as in a more easily applicable dipstick ELISA form. The sensitivity and specificity of the latter are lower compared with the standard ELISA –but remains higher than normal coprological methods. Currently, researchers are investigating the nature of antigens detected, with the aim of improving the test performance and evolving to a species-specific test that allows diagnosis of T. solium and T. saginata separately.
Recently, new PCR-based techniques have been developed that enable a speciesspecific diagnosis of T. saginata and T. solium in human faecal samples. Although very promising in terms of sensitivity and specificity, these tests are still not widely applied. Furthermore, the implementation of PCR-based technologies in areas where taeniosis/cysticercosis is endemic, does not occur –given the required infrastructure and cost associated with this technology.
Cysticercosis and neurocysticercosis (NCC)
The definitive diagnosis of cysticercosis is dependent on demonstrating the cysticercus in the tissue concerned. Demonstration of eggs and proglottids in the faeces is useful in the diagnosis of taeniosis, but not cysticercosis. Some of the diagnostic tests used in humans are antibody detection, electro-immunotransfer blot (EITB), and CT or MRI imaging.
Molecular tools
Differentiation of T. and T. saginata based on DNA analysis has been possible for several years. The main advantage of this technique is the ability to differentiate between T. saginata and T. asiatica –which is impossible when solely based on morphology. Differentiation of these human host species may be important, especially in epidemiological surveys, since Taenia species may travel around the world in their human definitive hosts.
Differential diagnosis
In cattle small abscesses in the muscles could be a differential diagnosis. In humans, the eventual differential diagnosis will depend on the localisation of the cysticercus in the brain.
Control
To prevent infection of cattle on farms, defecation on pastures must be prevented by supplying toilets to workers. To prevent human infections, South African public health legislation specifies that heavily infected meat be condemned at controlled/ licensed abattoirs, in order to prevent it entering the food chain. Carcasses with low levels of infection may be kept for 14 days at -14 ºC, to ensure the death of the larvae and to reduce the risk of infection.
Preventive measures (vaccination)
Early research on Taenia species showed that immunity to re-infection plays an important role in the natural regulation of transmission of these parasites. In addition, it was demonstrated that host-protective immune responses are directed towards the oncosphere stage in the early developing embryo. A recombinant vaccine has been developed against T. ovis cysticercosis in sheep. This was the first effective, definedantigen vaccine against a parasitic infection. Based on the homology of host-protective antigens between T. ovis and T. saginata , a recombinant vaccine was subsequently developed against T. saginata cysticercosis in cattle. Despite the high level of protection induced by these vaccines, the vaccines have not been commercialised due to financial considerations.