Helminths of pigs
This content is distributed under the following licence: Attribution-NoDerivs CC BY-ND  View Creative Commons Licence details here
View Creative Commons Licence details here

Helminths of pigs
Author: J BOOMKER
Introduction
Commercial piggeries seldom have problems with helminths because animals are raised on concrete or slatted floors which are regularly cleaned. There is, however, a growing trend to raise pigs under semi-intensive conditions –which may result in a resurgence of helminthiasis. In rural areas, pigs infected with Taenia solium via human faeces are a threat to human health since they can develop a severe form of infection called neurocysticercosis. Pigs can also be intermediate hosts of canine tapeworms such as E. granulosus granulosus and Taenia hydatigena , which can also be human pathogens. The domestic pig is a potential host of Trichinella spiralis , which is a serious human pathogen.
The helminths which occur in the various organ systems of pigs will be discussed in this section.
Stomach
Stomach worms
Ascarops strongylina
Physocephalus sexalatus
Importance
These nematodes cause only minor pathology –with gastritis as an occasional finding.
Distribution
Cosmopolitan.
Life cycle
These species have an indirect life cycle –using dung beetles as an intermediate host. Pigs become infected by ingesting the larval stage as a result of eating dung beetles or paratenic hosts.
The worms are non-migratory and develop in the mucosa of the stomach under the mucous layer. The pre-patent period is 6 weeks.
Appearance
The adult worms of both species are similar –being small (up to 22 mm) and filiform in shape. The eggs are up to 22 mm long.
The eggs of the two species can be differentiated by their appearance and size: Ascarops eggs are small, oval and roughly 45-51 x 22-26 μm; Physocephalus eggs are thick-shelled, larvated, and roughly 34-39 x 15-17 μm in size.
Diagnosis (patent infections)
The eggs can be seen in faecal samples using the flotation method.
Trichostrongylus axei
This nematode is mainly a helminth of ruminants, but is occasionally found in the stomach of pigs (see under helminths of ruminants ) .
Small intestine
Ascarid worm
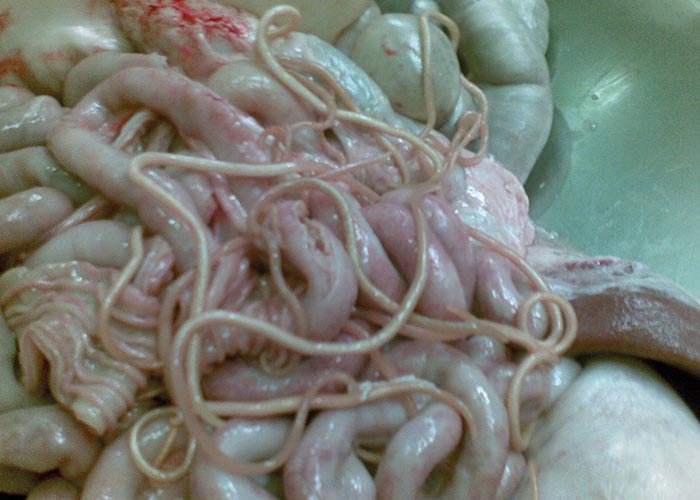
Ascaris suum
Ascaris suum is a highly host specific species which infects pigs.
Importance
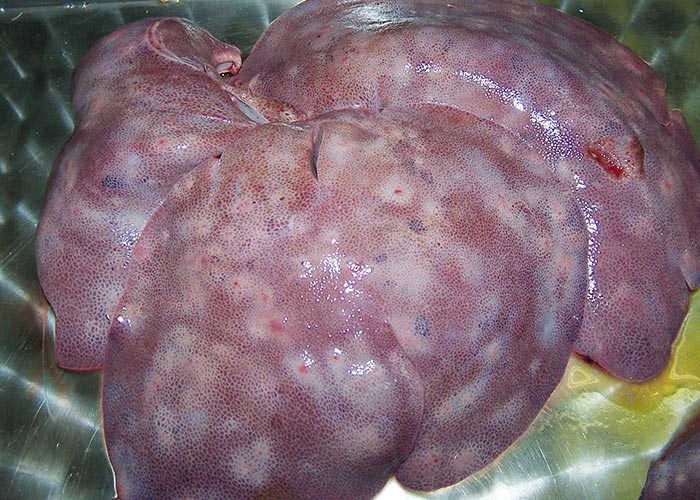
Ascaris suum is one of the commonest roundworms of pigs, but is, however, much over-rated as a pathogen. There is a high incidence in young pigs, and repeated infections induce partial immunity. ‘Milk spots ’–or multiple parasitic interstitial hepatitis, may if extensive, lead to entire livers being condemned during meat inspection.
Distribution
Cosmopolitan.
Life cycle
The pre-parasitic life cycle takes 3 weeks to be completed. In the host, the larvated eggs hatch and the larvae migrate through the liver and then into the lungs via the trachea. The pre-patent period is 6-8 weeks.
Appearance
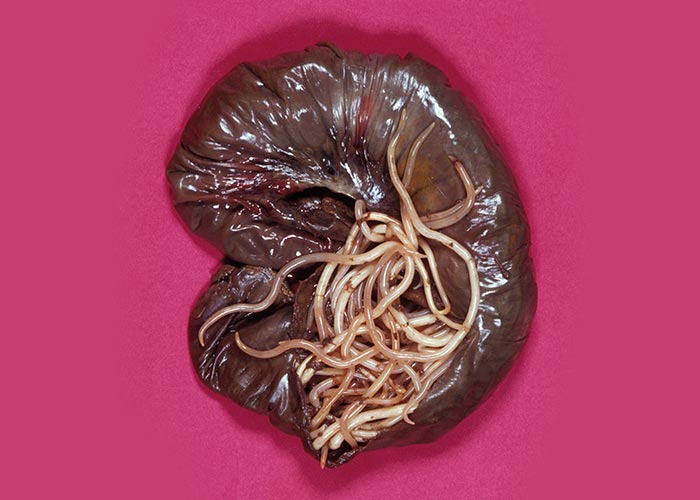
The adult worms are pinkish, large and stout (up to 2.5 mm thick) ( Figure 74 ). The sexes can be distinguished since males are up to 25 cm long with a slightly curled tail, while females can grow up to 40 cm long and are 2-5 mm thick. The eggs are medium sized (56-87 x 46-57 μm), subspherical, and with a thick, irregularly mammillated, yellowish-brown shell.
Diagnosis (patent infections)
Eggs can be seen in faecal samples using faecal flotation or faecal smears.
Epidemiology
A. suum has a one-host lifecycle with no intermediate hosts. Most infected animals develop immunity.
Pathogenesis and pathology
After ingestion the eggs hatch in the intestine, penetrate the gut wall and the L2 larvae infect the liver. Here they undergo a second moult to L3. They reach the lungs via tracheal migration and establish themselves in the small intestine where the L4 and adult worms are found.
Liver: The migration of larvae causes multiple parasitic interstitial hepatitis (‘milk spots ’) ( Figure 75 ) –which is seen as greyish-white, pinhead-sized foci (1 cm) on the surface of liver, with reticular marking. Other aetiologies for ‘milk spots ’are the nematodes Toxocara canis and Toxocara mystax .
Lung: Multiple lesions may be seen in the lungs.
Intestinal phase: Gastroenteritis, intestinal obstruction (rare). The worms may also sometimes migrate to aberrant sites like the bile ducts.
Clinical picture
Clinical signs are seen mainly in piglets. During the migratory phase, piglets may have coughing, dyspnoea (‘thumps ’or ‘heaves ’) and nasal discharge. In the intestinal phase, piglets have one or more of the following signs: a distended abdomen sensitive to pressure, constipation accompanied by light colic, inappetence, weakness, and retarded growth.
Treatment
- Piperazine and benzimidazoles (fenbendazole and flubendazole).
- Macrocyclic lactones (abamectin, doramectin, ivermectin).
Control
Infected piglets must be treated to prevent clinical signs. Breeding stock must be treated to prevent massive contamination, which results even from light infestation. Pens must have concrete or slatted floors, which will allow thorough and regular mechanical/steam cleaning and chemical disinfection.
Treatment schedule for pregnant sows and piglets: Treat pregnant sows 14 days before farrowing; treatment should be completed at least 4 days before entering farrowing pens. Sows should be washed down thoroughly –particularly the udder and teats –before being transferred to farrowing pens. Treat sows and piglets again after weaning, and decontaminate farrowing pens.
Strongyloides ransomi
Importance
- Mainly young piglets are clinically affected, as resistance develops early in life.
- Most important in warmer, humid areas.
- Anthropozoonosis: Cutaneous larval migrans in humans.
Distribution
Cosmopolitan.
Life cycle
Strongyloides species have both sexual and asexual cycles of reproduction. Free living males and females can reproduce and give rise to female worms. These female worms infect the host subcutaneously and then migrate to various organs in the host. They are parthenogenetic –producing eggs which are shed in the faeces. S. ransomi infects pigs of all ages transcutaneously. They migrate to the lung and then up the trachea, where they are swallowed and enter the intestine. Migration to the lactating udder of the sows results in piglets being infected during suckling. The prepatent period is 3-14 days.
Appearance
The parasitic worm population consists of parthenogenetic females only. They are small, ±6 mm long and colourless, and so are only microscopically visible in clear water over a smooth and dark surface.
Females are ovoviviparous. The egg is oval, medium-sized (50-60 x 25-30 μm), thinshelled, and larvated.
Diagnosis (patent infections)
Eggs can be demonstrated in faecal samples by the flotation method. Faecal cultures can yield larvae which can be identified. On postmortem, intestinal stages may be shown in mucosal scrapings, intestinal washings, or squashed samples of small intestine.
Epidemiology
Free-living, infective larvae can survive for up to 4 months, but because they are unsheathed they are susceptible to extreme climatic conditions. Warmth and moisture promote the development and allow a build-up of large numbers of infective larvae –in particularly the summer months. Lactogenic and percutaneous modes of infection favour transmission. Clinical strongyloidosis is mainly seen in stabled piglets, where resting places under infrared lamps are hotspots for infection. Piglets are susceptible up to weaning age, when immunity starts developing. However, the immunity does not incapacitate the hypobiotic somatic larvae.
Pathogenesis and pathology
On reinfection, the migratory phase can cause erythema and urticaria of the skin in the inguinal area and on the coronary bands, petechiae in the lung, and also interstitial pneumonia. In the intestine, a catarrhal enteritis may be seen which is characterised by hyperaemic mucosa, mucosal petechiae, and ecchymoses. Villous atrophy may cause malabsorption, hypoalbuminaemia, and hypoproteinaemia –which give rise to subcutaneous oedema and anaemia.
Clinical picture
Strongyloidosis manifests clinically in very young animals during the first few weeks of life. During the migratory phase, clinical signs caused by the migration are rarely observed. In the intestinal phase, piglets show inappetence, intermittent watery diarrhoea, weight loss, reduced growth rate, anaemia, apathy, sunken eyes with purulent discharge, and frothy discharge from the nose. Strongyloides can be highly pathogenic in suckling pigs; the mortality rate in a litter can be as high as 75%.
Treatment
- Benzimidazoles (fenbendazole and flubendazole).
- Macrocyclic lactones (abamectin, ivermectin and doramectin).
Control
Raising of Strongyloides -free piglets is made possible by removing them from the sow immediately after birth.
Public health
Like other Strongyloides species, S. ransomi can cause cutaneous larval migrans. Percutaneous infection can occur when there is contact of skin with damp ground which is contaminated with faeces of domestic stock.
Caecum and colon
Nodular worm of pigs
Oesophagostomum dentatum
Oesophagostomum quadrispinulatum
Importance
O. dentatum and quadrispinulatum are important pathogens of young pigs as they can cause severe wasting disease –with enteritis and colitis. The nodular lesions that result render the gut unsuitable for processing as sausage casings.
Distribution
Cosmopolitan in free-living pigs.
Life cycle
After the ingestion of infective larvae, there is a non-migratory histotrophic phase in the caecum and colon, which causes formation of nodules (hypobiosis). The prepatent period is 30-40 days.
Appearance
Stout, white worms, 1.0-2.5 cm long, with body often slightly curled. The head is usually bent so that –except for the predilection site –there is a slight possibility that they may be confused with hookworms. They produce strongylid-type eggs.
Diagnosis (patent infections)
Eggs can be demonstrated in faecal samples using flotation, and they can be identified by using subsequent faecal cultures.
Epidemiology
O. dentatum and O. quadrispinulatum produce free living, infective larvae which can survive for as long as 14 months on pastures.
Pathogenesis and pathology
The histotrophic phase produces the most severe effects; the mucosa of the caecum and colon develop localised inflammation around each larva. The inflammatory foci become encapsulated, causing nodule formation (millet seed to pea-sized) in the caecum and colon. Following repeated infections, the nodules become larger and the inflammatory reaction more severe. Extensive formation of nodules interferes with absorption, bowel movement, and digestion. Nodules often show abscessation due to bacterial infection, and may rupture to the peritoneal surface –causing peritonitis and multiple adhesions. Ulcerative colitis may occur due to ulceration of the mucosa when larvae emerge. Widespread haemorrhages and the formation of diphtheritic membranes and regional lymphadenopathy may occur. During the luminal phase, the late larval stages and adults are plug feeders –which can cause colonic mucosa to be hyperaemic, thickened, and covered with mucus when a large number of adult worms are present.
Clinical picture
Blood-stained mucoid diarrhoea, inappetence, general weakness, unthriftiness, progressive weight loss, retarded growth, and anaemia.
Treatment
The following anthelmintic groups are suitable for use in pigs:
- Piperazine.
- Benzimidazoles: fenbendazole and flubendazole.
- Macrocyclic lactones: abamectin, doramectin, ivermectin.
Control
The aim is to limit infections in sows and to produce Oesophagostomum -free piglets. This is achieved by strict hygiene. Pens, open ranges and pastures should be kept dry, and farrowing pens should be cleaned daily and disinfected once every week. Sows should be treated shortly before farrowing and then transferred to the farrowing pen.
Whipworm of pigs
Trichuris suis
Importance
T. suis is a cause of colitis, with haemorrhagic diarrhoea.
Distribution
Cosmopolitan.
Life cycle
The infective L1 develops within the egg. Animals ingest the larvated eggs and a non-migratory histotrophic phase develops in the mucosa of the large intestine ( Figure 76 ). The pre-patent period is 2-3 months.
Appearance
The worms are medium-sized (3.5-8 cm long), whitish, and with a whip-like tail. The eggs are lemon-shaped, mediumsized (70-80 x 30-42 μm), yellow brown, thick-shelled –with a projecting plug at each end.
Diagnosis (patent infections)
Demonstration of eggs in faecal samples (faecal flotation).
Epidemiology
Trichuris eggs can survive extreme temperatures and are resistant to desiccation. In a cool environment the eggs can survive for 4-5 years on pastures.
Pathogenesis and pathology
The pathology is related to worm burden. Adult worms tunnel into intestinal mucosa with their thin anterior ends and cause catarrhal inflammation with small epithelial defects, hyperaemia, and oedema. Whipworms suck blood –but the amount of blood withdrawn (±5 μl/worm/ day) is not regarded as significant enough to cause anaemia. Rather, this is caused indirectly as a result of bleeding from the damaged mucosa. There is colitis and formation of diphtheritic membranes.
Clinical picture
The main symptom is diarrhoea with mucus and blood ( Figure 76 ). Pigs show weight loss and retarded growth.
Treatment
- Benzimidazoles: fenbendazole and flubendazole.
- Macrocyclic lactones: abamectin, ivermectin and doramectin.
Control
The aim is to prevent clinical infection by treatment –as part of a regular control programme.
Brain
Taenia solium larvae
Cysticercus cellulosae
(See T. solium under muscle and tendons ).
Liver and kidney
Hydatid
Echinococcus spp. larvae
These are canine tapeworms which occasionally infect pigs and present a human health risk.
Fasciola hepatica
Infestation with these trematodes is occasionally seen in pigs ( see F. hepatica under ruminants ).
Stephanurus dentatus
Importance: The worms migrate through the liver and lodge in the kidneys. They are not regarded as particularly pathogenic.
Lungs
Hydatid
Echinococcus spp.
As under liver and kidney.
Metastrongylus apri
Importance: The adult worms are found in the lungs of free-living pigs –as the intermediate hosts are earthworms.
Muscles and tendons
Pork tapeworm
Taenia solium
Importance
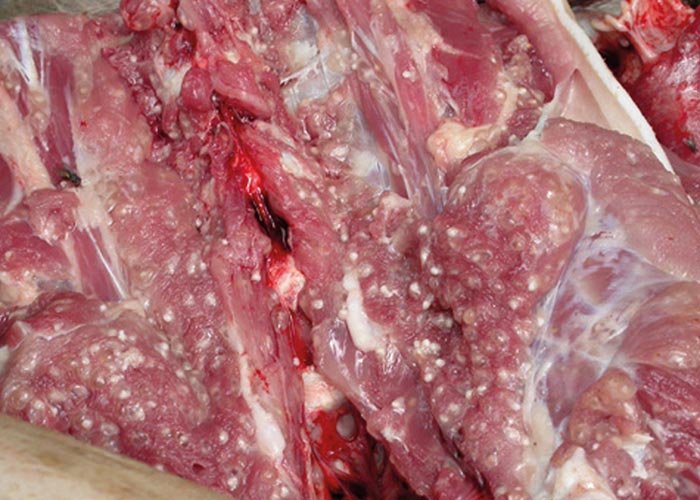
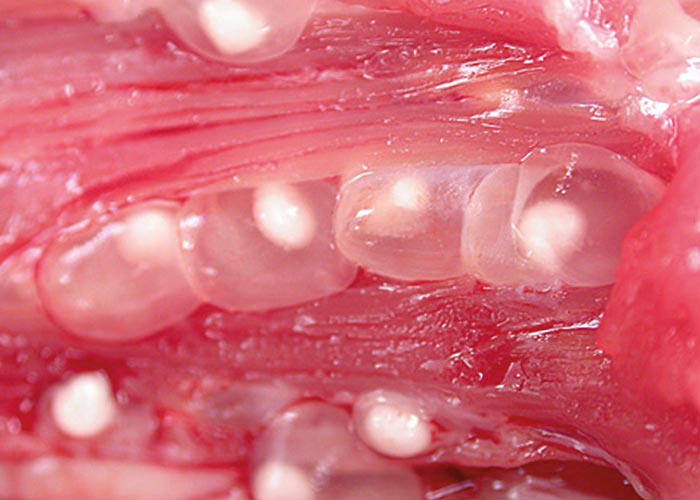
This is a zooanthroponosis, since it can result in neurocysticercosis in humans, which is an important cause of epilepsy. Taenia solium is founds mainly in free-living pigs, and only occasionally in commercial piggeries. Production losses result from the condemnation of meat containing cysticerci, known as ‘measles’, at abattoirs.
Distribution
Worldwide.
Reliable information on the distribution of Taenia solium is scarce, but it is endemic in most countries in South and Central America, Africa, and South and Southeast Asia where pigs are kept, mainly affecting subsistence pig farmers; it is absent from North America, Australia, New Zealand and most of Western Europe (https://www.who.int/taeniasis/epidemiology/en/) but imported cases are experienced sporadically in non-endemic areas.
Life cycle
Humans are the sole final host –in which the adult tapeworm is found in the small intestine. The eggs are expelled with detached proglottids in faeces. When pigs ingest T. solium eggs, they lodge in the muscle in the form of a cysticercoid, which lies dormant until the pork is eaten.
Appearance
The adult tapeworm in humans can be up to several metres in length ( Figure 77 ). The intermediate form or cysticercus is most commonly found in the striated muscles and muscle of the heart in pigs. These appear as small, white nodules, roughly 1-2 mm in diameter and are often referred to as pearly pork or measles ( Figures 78 and 79 ).
Epidemiology
Human faeces are the only source of infection for pigs. This usually occurs where free-living pigs ingest human excreta deposited around dwellings. Human infection usually occurs by ingesting undercooked pork containing cysts or ‘measles’ results in taeniasis. The larvae are released into the large intestine, where they develop into an adult worm. Egg-filled segments of the tapeworm known as proglottids are shed in the faeces and the eggs are released into the environment. After being eaten by a pig the larvae hatch and migrate to the striated muscles or sometimes the brain, where they form cysts. However humans can also inadvertently ingest tapeworm eggs, shed either by themselves (auto-infection) or via food contaminated by another person, and they then assume the role of an intermediate host; in these cases cysts developing in the brain can be a major cause of epilepsy.
In South Africa there is a high prevalence of T. solium infection in pigs kept in rural areas. A number of studies undertaken in the Eastern Cape show that there is a very high incidence of neurocysticercosis (NCC) in humans. Carabin et al. (2004) showed that there were 34 000 NCCassociated epilepsy cases due to T. solium infection, which they estimated to be a burden on the health services to the tune of US$20-30 million. Serrano Ocana (2009) showed that 60% of CT scans to investigate epilepsy cases could be attributed to NCC, with the highest prevalence in the 10-19 year-old age group.
Pathogenesis and pathology
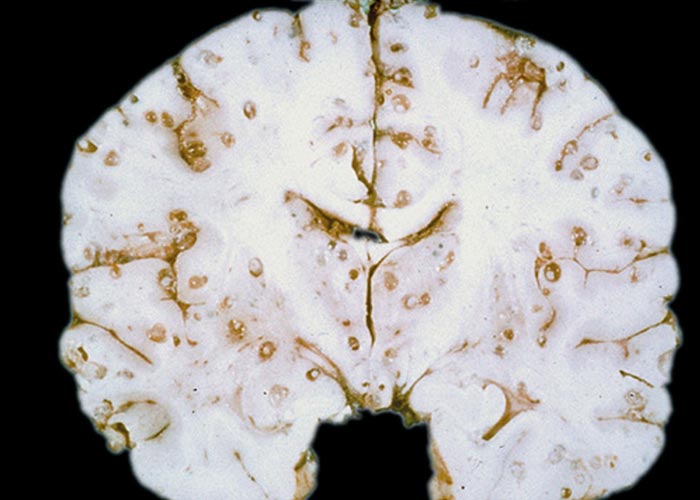
The ingested egg hatches in the intestine of the pig and bores its way through the intestinal wall and into the skeletal muscle –sometimes also affecting the heart. The cysticercus remains dormant here until eaten by the main host. Taenia solium can cause neurocysticercosis (NCC) in humans, dogs and cats ( Figure 80 ).
Clinical picture
The T. solium cysticercus which occurs in the muscle of pigs does not affect them adversely. However, Taenia solium NCC of humans, dogs and cats occurs when the host autoinfects itself, resulting in cysticercosis in the brain tissue. In South Africa this is the most common source of epilepsy in rural human populations.
Curative treatment
Pigs: Treatment of pigs with cysticercosis has been shown to be successful using oxfendazole –an inexpensive benzimidazole. A single oral dose of 30 mg/ kg is almost 100% effective. Although it should be considered as an important, cost-effective addition to the control of cysticercosis, its application has been hampered by the lack of convenient formulations.
Humans: Although it has been applied for several years, the treatment of patients infected with NCC, with cysticidal or anthelmintic drugs such as albendazole or praziquantel, remains controversial because of lack of evidence that the treatment is beneficial. Guidelines for treatment of cysticercosis have been proposed, but no adequate studies on efficacy have been reported. In addition in some patients –for example those with subarachnoidal cysts –therapy might be harmful. The drugs may cause arachnoiditis and arteritis, and consequently hydrocephalus.
Vaccination of pigs
Early research on the immunobiology of Taenia spp. indicated that immunity to re-infection plays an important role in the natural regulation of transmission of this group of parasites. In addition, it was demonstrated that host-protective immune responses are directed towards the oncosphere stage in the early developing embryo.
A recombinant vaccine has been developed against cysticercosis caused by T. ovis in sheep. This was the first effective, defined antigen vaccine against a parasitic infection. Based on the homology of host-protective antigens between T. ovis and T. saginata, a recombinant vaccine was developed against T. saginata cysticercosis in cattle. Despite the high level of protection induced by these vaccines, the vaccines were not commercialised because of financial considerations.
Using the same approach as above, one oncospheric antigen (TSOL 18) achieved complete protection against the development of cysticerci in vaccinated pigs. These experiments demonstrate the potential of recombinant oncosphere antigens in the development of a practical vaccine against porcine cysticercosis. Field trials showed protection against naturally acquired infection. The use of this vaccine is therefore expected to reduce the transmission of T. solium and reduce the incidence of NCC in humans.
Control
Since man is the only definitive host of T. solium , the infective cycle is easily broken by preventing faecal contamination of pastures –simply through the provision and use of toilets. Meat inspection must be carried out in abattoirs and infected pig carcasses must be condemned and destroyed. To prevent NCC, strict hand washing after defaecation should be practised stringently.
Trichinella spiralis
Importance
Public health importance: Trichinella spp. are non-pathogenic in animals and hence of no clinical importance in the veterinary field. However, trichinosis is a potentially fatal zoonotic disease in humans, which results from eating inadequately cooked or processed meat. Trichinella spp. have never been demonstrated in domestic pigs in Africa south of the Sahara, but a sylvatic cycle has been shown in the Kruger National Park where the cysts have been detected in the venison of antelope, crocodiles, warthog and bushpig.
Distribution
Cosmopolitan.
Life cycle
A sylvatic cycle occurs in which the encysted larvae are eaten and then develop to adult worms ( Figure 81 ). Adult worms develop within 24-36 hours of infection –the pre-patent period being 5-6 days and the patent period 4-6 weeks. Females are viviparous. Larvae (L1) enter lymphatic vessels and travel via the bloodstream to the skeletal muscles where they become encapsulated by stimulating the host genes to lay down collagen. Larvae (L1) become infective for new hosts 17-21 days after infection. Humans are accidental hosts –ingesting the larval cysts when eating venison or pork.
Appearance
Very small worms (1-4 mm long, up to 70 μm wide); the encapsulated larvae are not visible macroscopically. Microscopically, a lemon-shaped capsule (0.2-1.0 mm long) is shown to contain a single coiled-up larva (L1).
Diagnosis
There is no method of diagnosis in live animals, and infections are uncovered during meat inspection ( Figure 82 ). One method used for detection is an artificial digestion or trichinoscopy –used, for example, on the diaphragm and biceps muscle in crocodiles intended for consumption. Trichinella inspection is not compulsory in South Africa, but is required for meat intended for export.
Epidemiology
While both adults and encysted larvae develop within the same host, two hosts are required to complete the life cycle. South of the Sahara, trichinosis appears to be maintained in sylvatic cycles only –but the potential for introduction into domestic pigs exists.
Pathogenesis and pathology
Non-pathogenic in animals; highly pathogenic in man.
Control
Prevention of human infection, by Trichinella inspection.
Public health aspects (anthropozoonosis)
- Potentially fatal zoonotic disease.
- Infection by ingestion of inadequately cooked or processed pork, horsemeat or venison (drying, curing or smoking does not kill encysted larvae).
- Enteric phase: clinical signs resemble those seen in acute food poisoning (2-6 days after infection).
- Migratory (invasion) phase: myositis accompanied by severe myalgia, periorbital oedema, difficulty in chewing, breathing, and swallowing; muscular paralysis; remittent fever; splinter haemorrhages under nails and conjunctivae.
- Encystment phase: cachexia, oedema and extreme dehydration.
- Treatment only possible during enteric phase.
Subcutaneous tissue
Suifilaria suis
This worm is found in subcutaneous tissue and intramuscular connective tissue. The female is longer than male (32-40 mm). There is an abrupt end to the tail and it is studded with tubercles. The eggs contain L1 when shed. The life cycle is unknown. The worms cause white nodules which may develop into micro-abscesses if they become infected. They may be confused with the larval stage of T. solium .