Helminths of companion animals
This content is distributed under the following licence: Attribution-NoDerivs CC BY-ND  View Creative Commons Licence details here
View Creative Commons Licence details here

Helminths of companion animals
Author: E VOLKER SCHWAN
Nematodes
Hookworms (ancylostomosis)
Ancylostoma
General
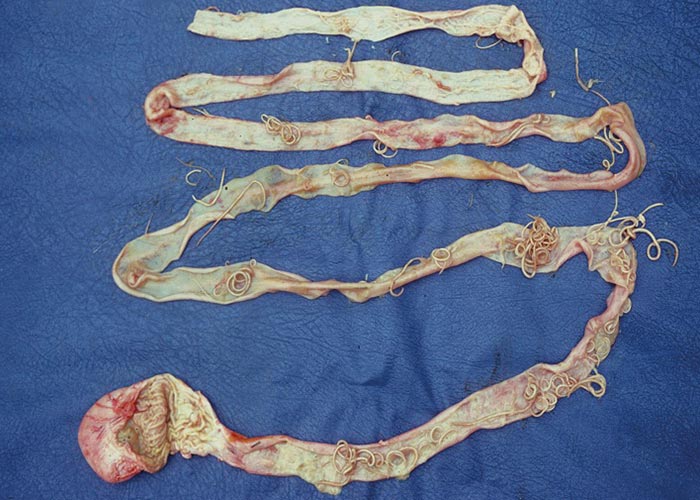
Ancylostomosis of dogs and cats is caused by the nematodes Ancylostoma caninum, Ancylostoma braziliense and Ancylostoma tubaeforme . The genus name derives from the Greek words ‘ankylos ’ (= bent) and ‘stoma ’ (= mouth), which, like the common name ‘hookworm ’, are descriptive terms referring to their dorsally bent (‘hook posture ’) anterior end. Hookworms are medium-sized, with a length of 5-25 mm. They have a large buccal capsule, which is armed with teeth. The posterior end of the males terminates in a strongylate bursa. The spicules are equal. The female ’s caudal end tapers gradually and the vulva is located in the posterior third of the body.
Male Ancylostoma caninum worms are 10- 13 mm long and the female worms 14-21 mm long. Both are about 0.5 mm wide. The worms have a large buccal capsule, which is armed with 3 pairs of ventral teeth. Spicules are equal and measure 800-950 μm in length.
Male Ancylostoma braziliense are 5-8 mm long and the females 7-11 mm long. Both are about 0.3 mm wide. The worms have a large elongated buccal capsule armed with 2 pairs of ventral teeth, of which the lateral ones are large and the medial ones small. Spicules are equal and measure 700-1 000 μm in length.
Ancylostoma tubaeforme closely resembles A. caninum but is smaller. Male worms are 9.5-11.0 mm long and female worms 12- 15 mm long. Both are about 0.4 mm wide. Their spicules are larger than those of A. caninum and measure 1 100-1 700 μm in length.
As for all hookworms, the small intestine is the site of predilection of the three species in their definitive hosts.
The eggs of the ancylostomatids of dogs and cats are of the strongylid-type. They are oval, thin-shelled, contain 4-8 blastomeres when laid, and measure 55-95 by 32-58 μm. The eggs of the three species are indistinguishable.
Hosts
- Ancylostoma caninum: In addition to dogs, the definitive host range includes sylvatic canids, and some of the sylvatic felids, ursids, procyonids and mustelids.
- Ancylostoma braziliense: In addition to dogs and cats, the definitive host range includes sylvatic canids, felids and mustelids.
- Ancylostoma tubaeforme: In addition to cats, the definitive host range includes a wide range of sylvatic felids.
Distribution
Ancylostoma caninum is mainly encountered in subtropical and tropical areas, but its range extends into the temperate zones of all continents. Ancylostoma braziliense is found exclusively in tropical and subtropica l areas, while Ancylostoma tubaeforme has a cosmopolitan distribution.
Transmission
All hookworm species have a direct life cycle. Eggs are shed with the faeces and –depending on the environmental temperature and humidity –there is typically a pre-parasitic development from egg to free-living L1, L2 and then finally, a sheathed infective L3, in as few as 4-5 days. Free-living infective larvae can remain viable for 3-4 months in damp soil.
There are various modes of infection for dogs and cats that vary depending on the species of hookworm involved:
- Percutaneous infection (A. caninum, A. braziliense, A. tubaeforme)
- Transmammary (lactogenic) infection (A. caninum)
- Infection by predation on paratenic hosts (A. caninum, A. braziliense, A. tubaeforme)
- Infection by ingestion of free-living infective larvae (A. caninum, A. braziliense, A. tubaeforme)
- Autoinfection (A. caninum)
Ancylostoma caninum: Following percutaneous infection, infective larvae follow a blood-tracheal migration route. However, when larvae are ingested orally –for example during transmammary ingestion, ingestion of free-living larvae or ingestion of paratenic hosts, –there appears to be no larval migration. The larvae remain in the intestinal tract and develop directly in the small intestine. Transmammary infection in dogs is regarded as the most important mode of infection.
Infection induces immunity with the result that larvae acquired by older dogs largely adopt a somatic migratory route. These larvae do not develop further and eventually become arrested in the musculature and fat tissue. Arrested larvae remain viable for several years and are reactivated during pregnancy. Reactivated larvae are the source for transmammary infection of litters and autoinfection of bitches. Also, in paratenic hosts, larvae do not develop further and become arrested during the course of their somatic migration. Depending on the mode of infection, the prepatent period is 15-26 days.
Ancylostoma braziliense: Little is known about the modes of infection in dogs and cats. The prepatent period is 13-27 days.
Ancylostoma tubaeforme: Percutaneous infection and infection by ingestion of paratenic hosts and free-living infective larvae appear to be equally important. The prepatent period is 19-22 days.
Socio-economic importance
With the Toxocara spp., A. caninum and A. tubaeforme are the most common and important nematodes of young dogs and cats, and, as the main causes of clinical verminosis, deworming programmes in cats and dogs focus mainly on these two species.
Ancylostoma braziliense is only mildly pathogenic and is of lesser importance.
All hookworm species of dogs, cats and other domestic animals have zoonotic implications and they are one of the causes of cutaneous larva migrans in humans. Cutaneous larva migrans is also colloquially known as ‘creeping eruption ’, ‘plumber ’s itch ’or ‘duckhunter ’s itch ’. Humans are infected percutaneously by free-living infective larvae. Infection is acquired following direct contact of the skin with damp ground which is contaminated with dog or cat faeces.
Larval migration in humans is restricted to the subcutaneous tissue and manifests clinically as itching and the appearance of serpiginous, tunnel-like skin lesions. Larvae migrate 3-5 cm a day and can remain active for several weeks –after which they eventually die off. Many of the cases resolve spontaneously without treatment.
Pathogenesis and pathology
Ancylostoma caninum and A. tubaeforme can be highly pathogenic –particularly when large worm burdens occur. In contrast, Ancylostoma braziliense is only mildly pathogenic. The pathogenesis varies according to the developmental phase of the parasites (migratory and lumenal).
Larval migratory phase: Penetration of the skin by large numbers of invading larvae occurs particularly in the interdigital spaces of the paws, on the limbs, the ventral part of chest, and on the abdomen. Continued exposure elicits a hypersensitivity –causing severe itching. Larval migration of A. caninum through the lungs in puppies can cause alveolar haemorrhage, oedema, and bronchopneumonia, following secondary bacterial infection.
Lumenal phase: Hookworm larvae and adults in the small intestine are haematophagous and histophagous. The volume of blood consumed by the worms differs between species; of the hookworms, A. caninum (80-200 μl/worm/day) and A. tubaeforme consume the most blood. The worms continuously change their site of attachment every 4-6 hours. As a result of the trauma inflicted by attaching to the mucosa, small petechial haemorrhages develop and blood oozes into the intestinal lumen and mucosal lesions. Heavy worm burdens cause a haemorrhagic enteritis ( Figure 62 ). Progressive blood loss over time causes anaemia, metabolic acidosis, and hypoproteinaemia.
Ancylostoma braziliense does not cause anaemia in their hosts, since individual worms consume only a minute amount of blood (1-2 μl/worm/day).
Clinical signs
The severity of clinical signs depends on the size of the worm burden, the age, and the nutritional and immunological status of the infected animal. Mild infections in both puppies and kittens and in older dogs and cats can be subclinical.
Puppies with heavy A. caninum burdens, –especially those acquired via the transmammary route –are prone to develop severe and often fatal clinical infections. Bronchopneumonia is the most common clinical picture in these cases. Coughing, a nasal discharge, and rarely epistaxis, are the result of tracheal migration in puppies. Heavy intestinal burdens of Ancylostoma caninum and A. tubaeforme cause progressive development of anaemia, diarrhoea containing mucus and sometimes blood, and also dehydration. Other signs observed during this phase are impaired development, weight loss, a dull coat and inappetence.
Following severe percutaneous infections, localized pruritus with urticaria and alopecia develop in the interdigital spaces of the paws, on the limbs, and on the ventral part of the chest and abdomen. This skin condition is also known as ‘moist summer eczema ’.
Diagnosis
Presumptive clinical diagnosis: The concurrent presence of anaemia, mucoid or haemorrhagic diarrhoea, weight loss, inappetence, anorexia, and retarded development –in particularly puppies and kittens –is a strong indication of hookworm infection.
Parasitological diagnosis: The parasitological diagnosis in live animals is confirmed by demonstrating worm eggs in faecal material, by means of direct flotation. In the early stages of heavy infections it is difficult to make an etiological diagnosis since the parasites are still in their prepatent period, and the diagnosis cannot be confirmed by faecal flotation.
Differential diagnosis
There are numerous other causes of anaemia and diarrhoea that should be considered as a differential diagnosis for hookworm infection. These are canine and feline babesiosis, canine leishmaniosis, canine monocytic ehrlichiosis, other helminth infections such as toxocarosis, trichuriosis, feline immunodeficiency virus and feline leukaemia virus infections, viral gastroenteritis caused by parvovirus and coronavirus, giardiasis, bacterial enteritis, poor nutrition, and drug reactions. For puppies with respiratory signs, other causes such as toxocarosis, kennel cough, and oslerosis should be excluded.
Treatment and control
With the exception of the piperazines, the same spectrum of anthelmintics listed for the treatment of toxocarosis is applicable for the treatment of ancylostomosis in dogs and cats. For details, see under Control section of Toxocara ( below ).
Ascarid worms (Toxocarosis)
Toxocara
General
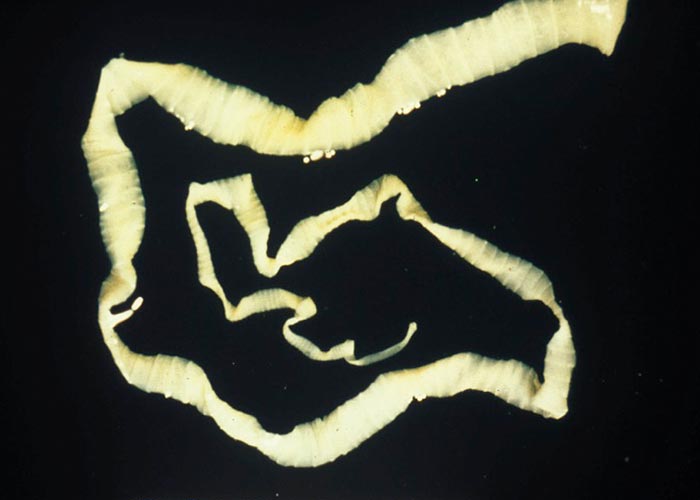
Toxocarosis of dogs is caused by infections with Toxocara canis, and toxocarosis of cats by Toxocara cati (syn Toxocara mystax) . The genus name is derived from the Greek words ‘toxon ’(= arrow, projectile, arch) which refers to the lanceolate or arrow-like appearance of the parasite ’s head and ‘ascaris ’ (= intestinal worm). Adult worms are easily recognized by their large size and typical ascarid appearance ( Figure 63 ).
In contrast to the related ascarid species Toxascaris leonina , the oesophagus of Toxocara spp. has a prominent posterior muscular bulb. The posterior end of the male tail of Toxocara has a finger-like process. In females, the vulva is located in the anterior fourth of the body.
Male Toxocara canis worms are 10-12 cm long and females 12-18 cm long. Both are about 2-3 mm wide. A pair of prominent, lanceolate cervical alae is visible microscopically. The spicules are slightly unequal and they are 750-1 300 μm long.
Male Toxocara cati worms are 6-7 cm long and females up to 10 cm long. Both are about 2 mm wide. A pair of prominent alae is visible microscopically, and they have an arrowhead-like shape.
The eggs of both Toxocara spp. are subspherical and have a thick, finely pitted, brown shell, which gives them a golf balllike appearance. Toxocara canis eggs are 75-90 μm in diameter and T. cati eggs 65-75 μm in diameter.
Hosts
Toxocara ascarids are intestinal parasites, and in companion animals they have a direct life cycle. Intestinal migration, somatic migration (prenatal infection in dogs) and transmammary infection are the most important modes of infection. Tracheal migration is not very common in Toxocara spp.
In addition to dogs, the definitive host range of T. canis includes a wide range of sylvatic canids. The definitive host range of T. cati includes domestic cats and a wide range of sylvatic felids, martens, mongooses, coatis, and civets.
Distribution
Toxocara canis and T. cati have a cosmopolitan distribution.
Transmission
Both species have a direct life cycle. The eggs are shed with the faeces into the environment. The eggs are highly resistant to the effects of the environment and can remain viable for up to 4 years. Depending on the ambient temperature, the infective L3 develops within the egg in as little as 2 weeks. Depending on the route of infection, the prepatent period ranges between 3 and 5 weeks.
There are various routes and methods of infection in dogs and cats:
- Infection by ingestion of larvated eggs (T. canis, T. cati)
- Transmammary (lactogenic) infection (T. canis, T. cati)
- Prenatal infection (T. canis)
- Infection by predation on paratenic hosts (T. canis, T. cati)
- Post-partum infection of bitches (T. canis)
The predilection site in ascarids of dogs and cats is the small intestine. Following ingestion of larvated eggs, infected paratenic hosts or milk in the case of transmammary infection, infective larvae enter the small blood vessels in the small intestine and migrate via the hepatotracheal route in non-immune hosts. L4 stages settle in the small intestine, where the final moult takes place. Infection induces immunity with the result that newly acquired larvae do not develop; they migrate somatically and eventually become arrested to remain as dormant larvae in various organs and in the musculature, liver, kidneys and CNS. Arrested larvae remain viable for several years and can become reactivated during pregnancy to become the source of larvae for transmammary and prenatal infections of litters. Infective larvae in paratenic hosts do not develop and become arrested in granulomas during the course of somatic migration. Post-partum infection of bitches occurs by ingestion of larvae passed in the faeces of heavily infected puppies.
Socio-economic importance
Hookworms and the Toxocara ascarid worms are the most common and important nematodes of puppies and kittens. Deworming of adult dogs and cats mainly focuses on these helminths.
Toxocara canis and T. mystax have zoonotic implications and are one of the causes of visceral larva migrans in humans. Humans become infected by ingesting larvated eggs. Clinical visceral larva migrans mainly features in young children, who contract infection by playing in contaminated soil or playgrounds through geophagia or by direct contact with infected dogs. Hatched larvae do not develop further and localise –as in other paratenic hosts –in the musculature, liver, CNS, eyes and other organs where they become entrapped in granulomas elicited by their presence. Most infections are sub-clinical.
The clinical signs of Toxocara visceral larval migrans are non-specific and include fever, respiratory signs, lymphadenopathy, hepato- splenomegaly, and indistinct neurological signs. Ocular larval migrans is a specific form of visceral larva migrans in which the eye is affected. Larvae migrating in the eye can cause endophthalmitis and formation of granulomas mostly in the retina, which can lead to irreversible, partial or complete blindness.
Most cases resolve without treatment. Treatment of problematic cases is usually unsatisfactory.
Pathogenesis and pathology
The development and locality of the lesions are associated with the following two developmental phases of the parasites:
Larval migratory phase: Larval migration through the liver and lungs causes the formation of widely disseminated microgranulomas in the liver, lungs, kidneys, and heart.
Lumenal phase: In the small intestine the worms are responsible for mucosal defects, intestinal obstruction, and occasional intestinal perforation. Migration of worms to aberrant sites such as the bile and pancreatic ducts may occur where they cause obstruction of the ducts and related clinical signs.
Metabolic products of the ascarids can interfere with parathormone production, which may result in rickets.
Clinical signs
Mild infections in both puppies and kittens, and older dogs and cats, are often clinically inapparent.
Puppies and kittens that become heavily infected via the transmammary route or in the case of T. canis, the prenatal route, show clinical signs that are dependent on the developmental phases of the parasite in the definitive host. During larval migration, coughing and nasal discharge are common clinical signs. Intestinal parasitism results in frequent vomiting, the development of a ‘pot-bellied ’abdomen which is sensitive to pressure, colic, and constipation alternating with diarrhoea. Sometimes intact worms are found in vomit and stools. Other signs that may be observed include anorexia, weight loss, impaired development, a dull coat, nervous disorders, and bone deformities.
Diagnosis
Presumptive clinical diagnosis: Clinical signs in puppies and kittens as listed are indicative of a Toxocara infection. Respiratory signs are initially seen during the prepatent period in animals with heavy infections. Haematology will show a marked eosinophilia.
Parasitological diagnosis: The parasitological diagnosis in live animals is based on the demonstration of eggs in faecal samples by means of direct flotation and faecal smears. Towards the end of the prepatent period, immature worms may be passed in stools and vomitus. Worms passed in stools and vomit should be differentiated from the related Toxascaris leonina.
Differential diagnosis
The differential diagnosis will differ depending on the stage and therefore site of the infection
During the respiratory phase prepatent hookworm infections, kennel cough, and oslerosis should be considered.
In animals with the intestinal phase, the differential diagnosis should consider other causes of diarrhoea and vomiting such as viral gastroenteritis (parvovirus and coronavirus infections), other helminth infections (toxocarosis, trichuriosis), giardiosis, bacterial enteritis, poor nutrition, and side effects of drugs.
Control
The control of hookworm and ascarid roundworms in dogs and cats is very important, not only from an animal health and welfare point of view, but also because of the zoonotic potential –as discussed above.
The eggs of both Toxocara spp. are very resistant to environmental conditions, thus making it impractical to control dog and cat roundworms via environmental decontamination. Toxocara eggs can also survive composting and sewage treatment. Environmental contamination –the removal of dog and cat faeces on residential properties and the prevention of dogs defecating in public areas –is very important for controlling the spread of roundworm eggs. Cleaning of bedding and floors in kennels is critical for preventing contamination of kennels. The floors of kennels must preferably be concrete to make cleaning of the kennels easier. Overcrowding in kennels and catteries and long-term confinement must also be avoided. The prevention of environmental contamination must be combined with the treatment of infected dogs and cats by an effective anthelmintic.
Suitable remedies: There are several anthelmintics belonging to various chemical groups that can be used for the treatment of hookworm and toxocarosis in dogs and cats. Some registered products contain two nematocides and may include a cestocide such as praziquantel. Note that a few of the actives mentioned below are only suitable for use in dogs. The following are registered for use in the treatment of roundworm infections in dogs and cats:
- Piperazines: Piperazine salts
- Benzimidazoles: Fenbendazole
- Probenzimidazoles: Febantel (D)
- Imidazothiazoles: Levamisole
- Tetrahydropyrimidines: Pyrantel
(hookworm only), oxantel (D) - Isothyocyanates: Nitroscanate (D)
- Macrocyclic lactones: milbemycin,
moxidectin, selamectin, ivermectin - Cyclic octadepsipeptides: Emodepside (C)
Treatment and control:
- Pregnant and lactating animals: Suckling puppies and kittens are very susceptible to roundworm infection from hookworm (Ancylostoma spp.) and roundworm (Toxocara spp.). Bitches and queens therefore need to be treated with an anthelmintic at mating time and again at whelping –to decrease prenatal infections. During lactation the dam needs to be treated every two weeks to prevent reactivated larvae from being established in the dam ’s gut and infecting the puppies/kittens, and to prevent the dam from getting re-infected from the environment. The most common route of infection in suckling puppies is via the transmammary and prenatal infection route from the reactivation of encysted larvae in the dam. Treatment of these arrested larvae will be beneficial in decreasing the incidents of prenatal infections. Most anthelmintics given at the recommended dose will not treat these dormant larvae present in the tissues of the bitch. Fenbendazole given to a pregnant bitch at 25mg/kg daily from the 40th day of pregnancy –until 2 days post-partum –has been shown to substantially reduce postnatal infection. This is, however, a time consuming and expensive treatment regime for owners to follow. Alternatively, macrocyclic lactones, iver mectin and doramectin (injected at 1mg/kg subcutaneously), given once on day 55 or 56 of pregnancy, or moxidectin (injected at 1mg/kg subcutaneously) given on days 40 and 50 has been known to decrease vertical infection of roundworms from the dam to puppies, while still in utero. Note, however, that the treatment of a pregnant dam as described above is extralabel, as none of the canine and feline anthelmintic products are registered for the treatment regimens described above. Certain herding type dog breeds, primarily Collies, Shetland Sheep Dogs and Australian Shepherds are known to be highly sensitive to macrocyclic lactones, and treatment in these breeds must be done with extreme caution.
- Puppies and kittens: Intestinal parasite infections in neonates may cause serious illness or even death before a diagnosis is possible via faecal examination –thus making the control of roundworms in neonates very important. Neonates can get infected with roundworms by picking up larvae in the environment or via the transmammary route from an infected dam. The selection of the correct anthelmintic for use in neonates is important. The products that can be safely used in neonates are those containing piperazine, fenbendazole, febantel, pyrantel and oxantel(D), nitroscanate(D) and milbemycin. It takes 2 weeks for hookworms to mature and to start laying eggs and therefore effective control in neonates will require treatment every two weeks starting from 2 weeks of age in puppies and 6 weeks of age in kittens –until weaning at 12 weeks. After weaning, puppies and kittens need to be treated monthly until they are 6 months of age –after which they can be treated every 3 months.
- Dog and cats over 6 months of age: The aim of treatment of dogs and cats over 6 months of age is to lower environmental contamination of roundworms, prevent zoonotic transmission, and to prevent clinical signs of infection in dogs and cats. Most companies that market worm treatments advise deworming adult cats and dogs every 3 months, 4 times a year. This can be increased to monthly treatment in areas where the risk of infection is higher. Ideally, this should be combined with regular faecal examination to ascertain the response to treatment.
- Cost of treatment for hookworm and ascarid prevention: The frequency of treatment required for adequate control of these roundworms can be expensive, in particular the macrocyclic lactones including milbemycin and selamectin, as well as the newer remedy emodepside. Pyrantel and oxantel are cheaper, while nitroscanate and generic piperazines are even more economical. Fenbendazole intended for livestock use can be used extra-label by veterinarians as another cheaper option.
- Pyrantel resistance: Pyrantel resistance in A. caninum has been confirmed in Brisbane in Australia and it is suspected that this may occur in other subtropical cities. The situation in South Africa is unknown.
Toxascaris (Toxascariosis)
General
Toxascariosis of dogs and cats is caused by infection with the nematode Toxascaris leonina . The genus name is derived from the Greek word ‘toxon ’ (= arrow, projectile, arch) which refers to the lanceolate appearance of the head, and ‘ascaris ’ (= intestinal worm). Its predilection site is the small intestine. Being a typical ascarid, adult worms are easily recognisable because of their comparatively large size. In contrast to the related Toxocara spp., the oesophagus of T. leonina , which can be visualised in cleared specimens, lacks a posterior bulb. Male worms are 6-7 cm long and female worms 6-10 cm long –and both are about 2 mm wide. A pair of prominent lanceolate cervical alae can be seen microscopically. Eggs of T. leonina are spherical, 70-85 μm in diameter and have a thick, smooth and colourless shell.
Hosts
Apart from dogs and cats, the definitive host range includes a wide range of sylvatic carnivores.
Distribution
Toxascaris has a cosmopolitan distribution.
Transmission
Toxascaris leonina has a direct life cycle. Female worms lay a large number of eggs which are shed with the faeces into the environment. The eggs are highly resistant to adverse environmental conditions and can remain viable for several years. Depending on the ambient temperature, an infective L3 can develop in the egg within 8 days.
The only modes of infection in dogs and cats are ingestion of larvated eggs or of infected paratenic hosts (eg mice). Infective larvae undergo a histotropic phase in the mucosa of the small intestine during which time the third moult takes place. Larvae re-enter the intestinal lumen where they develop to adults. The prepatent period is 7-10 weeks.
Socio-economic importance
Toxascaris leonina is less common and less important than the Toxocara spp. There are no reports of infections in humans.
Pathogenesis and pathology
Since there is no prenatal or transmammary transmission, worm burdens are smaller and their pathogenic effects minor compared to those of Toxocara infections in puppies and kittens.
Clinical signs
Toxascaris leonina can cause enteritis if dogs and cats carry large worm burdens.
Diagnosis
The parasitological diagnosis in live animals is based on the demonstration of eggs in faecal samples by means of direct flotation and faecal smears.
Differential diagnosis
As for Toxocarosis .
Control
The drug spectrum and hygiene measures listed for toxocarosis are also applicable to the control of toxascariosis.
Spirocerca lupi (spirocercosis)
General
Spirocercosis of dogs is caused by the nematode Spirocerca lupi . The genus name is derived from the Greek word ‘speira ’ (= coil, thread) which refers to the appearance of fresh adult specimens which are blood-red and coiled. Adult worms are medium-sized. Male worms are 3-5 cm long and females 5-8 cm long, and they are about 1 mm wide. The worms have a typically hexagonal mouth opening that can be seen microscopically, while the male ’s posterior end is spiral in shape and bears caudal alae. The spicules are unequal and are 2.4-2.8 mm (left) and 0.47-0.75 mm (right) long. The tail of the female is blunt and the vulva is located in the oesophageal region.
The typical predilection sites are the wall of the caudal part of the thoracic oesophagus and the cardia of the stomach –where they are found embedded in fibrous nodules that protrude into the lumen. To a lesser extent, worms are found in aberrant locations such as in the kidneys and under the skin.
Eggs of S. lupi are small (22-37 x 11-15 μm), elongate with parallel sides, thickshelled, and larvated (embryonated) when laid.
Hosts
Apart from dogs, which are the predominant definitive host, infections have also been reported from a range of sylvatic carnivores, and occasionally from domestic ruminants and equids.
Distribution
Spirocerca lupi is widespread in tropical and subtropical parts of the world. In South Africa, infection is commonly seen in farming areas and even urban areas like Pretoria.
Transmission
Spirocerca lupi has an indirect life cycle. The females are ovoviviparous –shedding embryonated eggs in the faeces and vomitus.
Various dung beetle genera are intermediate hosts of S. lupi . Following ingestion of the eggs, the L1-stages hatch and develop to the infective L3- stage. A wide spectrum of domestic and wild birds, amphibia, reptiles, and small mammals commonly prey on dung beetles and may be paratenic hosts.
Dogs are infected by either ingesting infected dung beetles or paratenic hosts (chicken scraps, birds, lizards and other potential hosts). The migratory route and larval development within the definitive host is complex and determines the distribution of the lesions caused by the parasites. After ingestion, the thirdstage larvae penetrate the gastric mucosa and enter the wall of gastric arteries to eventually reach the abdominal aorta –from where they migrate to the thoracic aorta and where they remain for up to 3 months and develop to L4-stages. From the aorta, fourth-stage larvae migrate through the thoracic cavity and reach the oesophagus 15 weeks after infection. The larvae occur in the oesophageal subepithelial connective tissue, from where they perforate the oesophageal epithelium. Once the worms are mature, eggs are shed through this perforation into the oesophageal lumen –from where they enter the intestinal tract and are shed in the faeces. The final development to the adult stage takes place in fibrous nodules in the oesophageal wall that form as a result of the host ’s inflammatory response to the worm.
The prepatent period is 4-6 months and the patent period lasts about 2 years.
Socio-economic importance
In endemic and particularly in hyperendemic areas, S. lupi is one of the most important helminths of dogs due to the severity of the lesions induced, and because of the expense of diagnosis and treatment.
In addition, the diagnosis in live animals requires specialist diagnostic facilities.
The prognosis of complicated spirocercosis is poor –even with intensive treatment.
The risk of human infection worldwide is low and there is only a single report of an infant in Italy being infected.
Pathogenesis and pathology
The lesions caused by the parasite and their locality are determined by the migratory route and developmental phases of the parasite.
Larval migration phase: Larval migration in the thoracic aorta results in ‘scarring ’, which is recognised as a pathognomonic lesion of S. lupi infection. Depending on the severity of infection, this can lead to extensive internal haemorrhages and consequently death. Larvae migrating in the wall of the thoracic aorta cause degeneration of elastic tissue and replacement of collagen –which is followed by calcification and ossification. The result is the formation of aneurysms and stenosis. Sudden death as a result of rupture of an aortic aneurysm is not uncommon.
The mechanism of malignant transformation into oesophageal fibrosarcomas and osteosarcomas is poorly understood. Dogs with intrathoracic sarcomas often develop hypertrophic osteopathy (Marie ’s disease). Deformative ossifying spondylitis affecting the thoracic vertebrae is another poorly understood sequel to the infection.
Clinical signs
Several clinical forms of spirocercosis can be differentiated.
Subclinical form: Apart from occasional regurgitation or vomiting, usually no clinical signs are observed. This is common in areas of high prevalence.
Acute haemorrhagic form: This occurs due to the invasion of the aortic wall by larvae, resulting in rupture of the aorta and consequent sudden death.
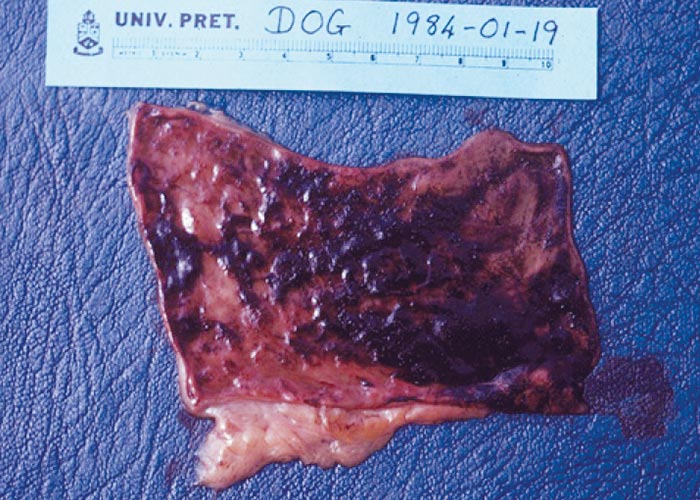
Chronic form: This results from the establishment of parasites in the oesophagus and the development of fibrous nodules ( Figure 64 ). Clinical signs observed are decreased appetite, vomiting, regurgitation, dysphagia, salivation, oedema of the throat, coughing, dyspnoea, thoracic pain, weight loss, anaemia, and weakness.
Neoplastic form: The parasitic nodules undergo neoplastic transformation, which is followed by cachexia, vomiting, regurgitation, and hypertrophic osteopathy.
Cutaneous form: Following aberrant migration, worms may localise in the subcutaneous tissues, causing protruding nodules in the skin.
Diagnosis
The diagnosis in live dogs is problematic because of the long prepatent period of the parasite –which can be up to 6 months long. The detection of the migrating parasites during the prepatent period is difficult. The clinical diagnosis can only be confirmed by the demonstration of adult worms in oesophageal nodules or by demonstration of S. lupi eggs in the eggs using faecal flotation.
Presumptive clinical diagnosis: Animal particulars and history should be considered –for example, breed, age, origin, conditions under which animal is kept and used, changes in behaviour, changes in appetite, contact with and ingestion of lizards and other small vertebrates and insects, and a tendency to coprophagia.
General clinical signs such as chronic weight loss, vomiting, regurgitation, anaemia and weakness are indicative of the infection.
Clinical diagnosis: An initial diagnosis can be made via radiography. A typical oesophageal mass will usually be seen in radiographs. The mass is usually found in the caudal oesophagus. A moderate amount of air may be present, cranial to the mass. The mass can best be seen via a ventrodorsal (VD) or dorsoventral (DV) view as a single midline soft tissue opacity superimposing on the caudal cardiac border and diaphragmatic cupula. However, cases presented before oesophageal nodules have formed cannot be diagnosed via radiography. Small nodules or atypically located nodules may also not be visible on radiographs. Endoscopy is regarded as the technique of choice which allows the demonstration of oesophageal nodules, as it is more sensitive than radiography in diagnosing S. lupi infection where the nodules are too small to be detected on radiographs. It is important to inflate the oesophagus adequately to prevent oesophageal folds being misdiagnosed as small S. lupi nodules. Early nodules are smooth and rounded and have a pink, nipple-like protuberance through which the female lays her eggs. The nodules are typically found caudal to the heart and may extend through the cardia into the stomach. Over time the nodules may enlarge and become roughened, lobulated, cauliflowerlike masses that obliterate the oesophageal lumen. At this stage the nodules have often undergone neoplastic transformation.
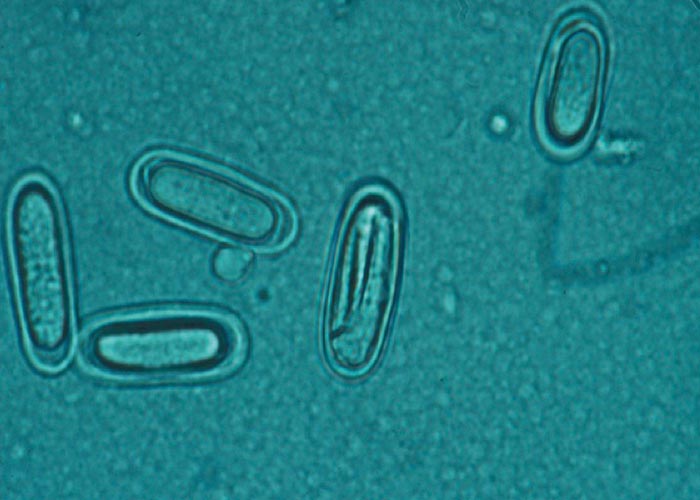
Parasitological diagnosis: The parasitological diagnosis in live dogs is based on the demonstration of eggs in faecal samples by means of direct faecal flotation or combined centrifugal sedimentation/ flotation techniques ( Figure 65 ). It is essential to use flotation fluids with a high specific gravity (SG) of not less than 1.3 (eg saturated MgSO4 ). Because of the relatively small size and the small number of the eggs, faecal examination should be entrusted to the experienced laboratory diagnostician. Failure to detect eggs in a flotation procedure does not necessarily indicate a negative result since the sensitivity of faecal examination is 70-80%.
Differential diagnosis
As a result of the varied and often general clinical signs associated with S. lupi infection, the differential diagnosis list is very long.Control
Treatment: Uncomplicated S. lupi infections (non-neoplastic granulomas) can be treated effectively with macrocyclic lactones (MLs). Topical application of moxidectin 2.5% m/v combined with imidacloprid 10% m/v (Advocate ®Bayer) weekly for 19 weeks –starting 170 days after infection –showed a 98.5% efficacy in the treatment of S. lupi infection in eight dogs. Milbemycin oxime (Milbemax ®Novartis) at a minimum dose of 0.5 mg/kg bodyweight, given as a once-off dose 30 days post infection with S. lupi was 79.8% effective at preventing the establishment of S. lupi in the oesophagus in seven dogs. In a second study, milbimycin oxime was given at a minimum dose of 0.5 mg/kg bodyweight 28 days post-S. lupi infection to 4 dogs. The dose was repeated at 14 or 28 day intervals up to 169 days post infection. The treatment was found to be 100% effective in preventing the formation of S. lupi nodules in the oesophagus. Doramectin (Dectomax ®Zoetis, extra label) has also been used for the treatment of S. infection. A standard dosage of 0.4 mg/kg SC or orally should be given either weekly or every two weeks, until the resolution of clinical signs.
Control: In a study in the Eastern Cape Province of South Africa, 58 puppies from a S. lupi endemic area were selected for treatment. Thirty eight of these puppies received Milbemycin oxime (Milbemax ®Novartis) at a minimum dose of 0.5 mg/ kg bodyweight at 2-6 weeks of age. The puppies received five further treatments at approximately 28-day intervals. The remainder of the puppies were left as untreated controls. Twenty four of the 27 control dogs became infected with S. lupi infection as demonstrated by aortic nodules. This treatment regime reduced the severity of the aortic lesions and significantly reduced the number of oesophageal nodules in the treated dogs.
Use of MLs in dog breeds prone to ABCB1 (formerly MDR1 gene) mutations: MLs must be used with caution in dog breeds that are prone to ABCB1 gene (formerly MDR1 gene) mutations –which predispose them to toxicity.
The mutation is typically seen in the herding type breeds –primarily Collies, Shetland Sheep Dogs and Australian Shepherds. The mutation has also been detected in Longhaired Whippets, Old English Sheepdogs, German Shepherd Dogs, Swiss Shepherd Dogs and some crosses of these breeds. Moxidectin 2.5% m/v combined with imidacloprid 10% m/v (Advocate ®Bayer) has been applied topically to confirmed ivermectin sensitive collies at five times the recommended dose –with no adverse effects.
Canine lungworm (Oslerosis)
Oslerus
General
Oslerosis of dogs is caused by the nematode Oslerus osleri (syn Filaroides osleri) . The genus is named after the Canadian physician W Osler, who first described the species. The typical predilection sites are the trachea and bronchi, where the nematodes are found within typical, flat, up to bean-sized raised nodules in the mucosa.
Outbreaks of clinical oslerosis are seen occasionally, under specific conditions, usually where dogs are kept in crowded and unhygienic conditions –for example in boarding kennels. Infection is most often seen in weaned puppies, not less than 10 weeks old.
Male worms are 4-7 mm long and females 10-15 mm long. Both are slender. The posterior end of the male is rounded and lacks a bursa. The spicules are slightly unequal and are 99-113 μm long. In females, the vulva is located close to the anus.
The eggs of O. osleri are oval and thinshelled and measure 80-120 x 60-70 μm. They contain a larva (L1-stage) when laid. Some of the eggs hatch in the trachea and may be seen in faecal examination. Larvae have a typical S-shaped tail.
Hosts
The definitive host range includes sylvatic carnivores, but O. osleri is not a common parasite of dogs.
Distribution
Oslerus osleri has a cosmopolitan distribution.
Transmission
Oslerus osleri is an unusual nematode since no development is required outside of the definitive host. Oslerus osleri has a direct life cycle. Females are ovoviviparous, i.e . they produce embryonated eggs.
Dogs of all ages are susceptible to the infection. The definitive host is infected by ingesting L1-stage larvae shed in expectorations, saliva, vomitus and faeces (coprophagia). Infection is mainly acquired by young puppies from the bitch through licking and cleaning soon after birth and by regurgitational feeding of puppies. After entering the intestinal wall, infective larvae migrate via the lymphatictracheal route. The prepatent period is 10 weeks and the patent period extends over several years.
Socio-economic importance
Oslerosis in dogs is of minor importance. There are no reports of O. osleri in humans.
Pathogenesis and pathology
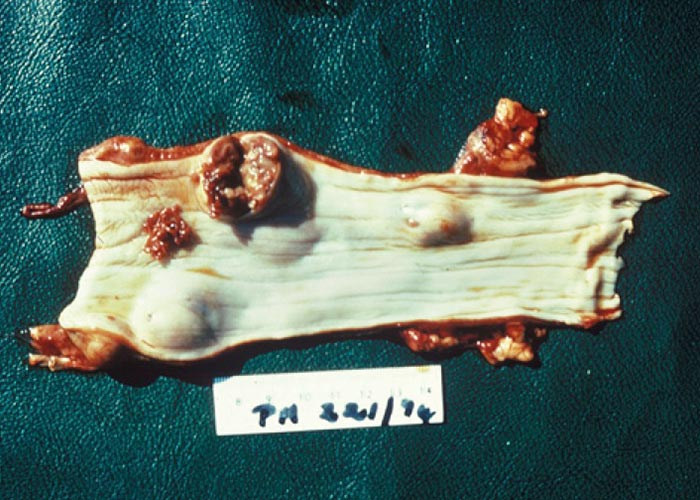
In the trachea and bronchi, worms develop in submucosal nodules which form as a result of an inflammatory host response. Nodules are flat and millet- to beansized. Nodules narrow the lumen of the air passages, which impacts on breathing ( Figure 66 ).
Clinical signs
Many infections are clinically inapparent. The clinical picture is that of chronic tracheobronchitis. Clinical signs usually include respiratory distress and a chronic, dry (non-productive) paroxysmal cough, which can be induced by exercise. Other signs include inappetence, vomiting and weight loss.
Diagnosis
Presumptive clinical diagnosis: A febrile chronic tracheobronchitis not responding to antibiotics is indicative for oslerosis. Important to consider are animal details and history (e.g. age, when and where acquired, and recent stay in boarding kennel).
Clinical diagnosis: Endoscopy is regarded as the technique of choice, which allows the demonstration of nodular lesions in the trachea and bronchi. Radiography is unsatisfactory.
Parasitological diagnosis: Some of the infective L1-stage larvae already hatch in the respiratory tract and occur together with unhatched eggs in sputum and faeces.
The parasitological diagnosis in live dogs is based on the demonstration of larvated eggs and larvae in the sediment of tracheal lavage samples. Demonstration of larvae in faecal samples is less sensitive and can be attempted by flotation with ZnSO4 .
Differential diagnosis
Other causes of chronic tracheobronchitis that should be considered as differential diagnoses include infectious tracheobronchitis (kennel cough), prepatent toxocarosis and ancylostomosis, other metastrongylid infections, dirofilariosis, tracheal collapse, and cardiovascular disease.
Control
Treatment: Fenbendazole and oxfendazole at a dose rate of 50 mg/kg daily for 15 consecutive days and ivermectin have been used successfully extra-label for the treatment of oslerosis in dogs.
Control: Hygiene in boarding and breeding kennels is essential to prevent infections. Regular removal of faeces, washing and disinfection of cages is essential.
Filarial worms of dogs (Acanthocheilonemosis)
Acanthocheilonema
Dipetalonema
General
Acanthocheilonemosis of dogs is caused by the nematodes Acanthocheilonema reconditum, and Acanthocheilonema dracunculoides. Acanthocheilonemosis is of minor clinical importance and therefore only very occasionally might require veterinary intervention. A. reconditum occurs in the subcutaneous fascia of the limbs and back, while A. dracunculoides occurs mainly in the abdominal and to a lesser extent in the thoracic cavity
The genus name is derived from the Greek words ‘acantha ’ (= spiny, thorny), ‘cheilos ’ (= lip) and ‘nema ’ (= thread). Acanthocheilonema species are medium to large nematodes and have an atrophied stoma. The posterior end of males is tapering and spirally coiled, and the spicules are dissimilar in length and shape. The posterior end of females is digitate and the vulva is located in the oesophageal region of the body. Female worms are viviparous and produce unsheathed L1-stages known as microfilariae.
Refer to Table 9 for the comparative morphological details of A. reconditum and A. dracunculoides .
Hosts
In domestic carnivores, A. reconditum , and A. dracunculoides are only found in dogs –but their host range includes some wild canids and hyaenids.
Distribution
Acanthocheilonema reconditum has a cosmopolitan distribution. Only sparse information is available regarding Africa, but it is known to be endemic in South Africa and neighbouring Botswana and Mozambique. In South Africa, A. reconditum is commonly encountered in neglected dogs.
Acanthocheilonema dracunculoides has been reported from Africa, Asia and Europe. In Africa it is widely distributed and is also endemic in South Africa and neighbouring Namibia, Zimbabwe, and Mozambique. Infection is commonly encountered in dogs in Namibia –but rarely in South Africa.
Transmission
All Acanthocheilonema species have an indirect life cycle. Microfilariae produced by females appear in the blood nonperiodically.
The Acanthocheilonema species are transmitted by a number of vectors. The vectors become infected by obtaining blood-meals from microfilaraemic dogs. In the vector, the microfilariae develop to the infective L3-stages (metacyclic larvae). The incubation period for A. reconditum in the cat flea (Ctenocephalides felis) may be as short as 7 days. Fleas (Ctenocephalides canis, Ctenocephalides felis, Pulex irritans) and to a lesser extent lice (Heterodoxus spiniger, Linognathus setosus) act as intermediate hosts (vectors) for A. reconditum. The louse fly (Hippobosca longipennis ) and the kennel tick (Rhipicephalus sanguineus) are the vectors of A. dracunculoides .
Definitive hosts become infected when the vectors feed. The migration pattern after infection with metacyclic larvae, and their further development in definitive hosts, is unknown for both species. The prepatent period for A. reconditum is 2-3 ½months, but it is not known for A. dracunculoides .
Socio-economic importance
Acanthocheilonemosis is important as a differential diagnosis of heartworm infection. Diagnosis of the disease in live animals requires specialist diagnostic procedures –which are expensive.
Acanthocheilonema reconditum and A. dracunculoides do not have zoonotic implications.
Pathogenesis and pathology
Acanthocheilonema reconditum and A. dracunculoides are regarded as largely non-pathogenic.
Clinical signs
Isolated cases of pruritic dermatitis and focal alopecia have been attributed to A. reconditum infections with high microfilaraemias. Similarly, there are reports of animals infected with A. dracunculoides presenting with pruritus, alopecia, erythema of the skin, skin ulcers, ascites, pleural effusion, ataxia, incoordination and cachexia.
Diagnosis
The parasitological diagnosis in live animals is based on the demonstration of unsheathed microfilariae found in blood. Microfilariae can be detected in wet blood films, stained thin blood films and by the capillary haematocrit tube method. For increased sensitivity, concentration techniques such as membrane filtration, preferably, or the modified Knott ’s test should be used. In order to determine the species, histochemical staining for acid phosphatase activity is required ( Table 9 ). Adult worms in the abdominal cavity are commonly encountered during spays.
Differential diagnosis
Infections with filariids of the genus Dirofilaria and Brugia have to be taken into consideration. For a complete discussion of the differential diagnosis and how to distinguish between the different microfilaria, refer to the section dealing with dirofilariosis.
Table 9 Comparative morphology of the Acanthocheilonema spp. of dogs
| A. reconditum | A. dracunculoides | |
|---|---|---|
Adult stages
| 21-36 mm x 0.07-0.2 mm 9-17 mm x 0.07-0.13 mm 220-300 μm (l) and 92-104 μm (r) | 30-60 mm x 0.2-0.37 mm 15-31 mm x 0.1-0.31 mm 320-402 μm (l) and 120-186 μm (r) |
Microfilariae
| Unsheathed 168-292 μm x 4-6.7 μm Uniform with lighter area from cephalic end to excretory vesicle or diffuse denser staining at excretory pore, inner body and anal pore | Unsheathed 121-277 μm x 3.1-7.4 μm Cephalic space, excretory pore, inner body, anal pore |
Control
Treatment: Macrocyclic lactones are suitable for the treatment of microfilaraemic dogs. Refer to the section dealing with dirofilariosis for full detail of treatment regimes
Control: Infection with A. reconditum is commonly seen in neglected dogs in South Africa. Flea control can effectively prevent infection.
Canine Heartworm
(Dirofilariosis)
Dirofilaria spp.
General
Dirofilariosis of dogs and cats is caused by the nematodes Dirofilaria immitis and Dirofilaria repens. Dirofilaria immitis is also known by the common names ‘heartworm ’and ‘canine heartworm ’, and it typically occurs in the right ventricle, right auricle, and the pulmonary artery. Infection is also referred to as cardiovascular dirofilariosis. Dirofilaria repens typically occurs in the subcutaneous connective tissue and the fascial sheaths of the hind legs. Infection is also referred to as cutaneous dirofilariosis. Dirofilaria repens is mainly important as a differential diagnosis of heartworm infection.
The genus name is derived from the Latin words ‘diruo ’ (= to destroy, to ruin) and ‘filum ’ (= thread) and refers to the pathogenic effects of some of these filiform (thread-like) worms. Dirofilaria species are medium to large nematodes and have an atrophied stoma. The posterior end of the male is spirally coiled, rounded and fitted with alae. The spicules are dissimilar in length and shape. The posterior end of the female is rounded and the vulva is located just posterior to the oesophageal region.
Female worms are viviparous and produce unsheathed L1-stages known as microfilariae.
Refer to Table 10 for the comparative morphological details of D. immitis and D. repens .
Hosts
Dogs are the principal definitive hosts of Dirofilaria immitis . Cats are less susceptible and are regarded as an insignificant reservoir. The host range also includes some wild canids, felids, other mammals and humans. However, in most of these hosts infection is amicrofilaraemic and the worms are located in aberrant sites.
Dogs and cats are the principal definitive hosts of Dirofilaria repens. Infection has also been reported in wild canids, felids and humans.Increasing climatic changes and movement of people with their pets are important factors for the continuous spreading of the parasite.
Distribution
Dirofilaria immitis has an almost cosmopolitan distribution and occurs in tropical, subtropical and temperate regions. In Africa it is known to be endemic in Morocco, Algeria, Tunisia, Egypt, Senegal, Ethiopia, Kenya, Tanzania, Malawi, Mozambique, Gabon, DRC, Angola, Madagascar, Mauritius and R éunion. In South Africa heartworm has so far only been reported in imported dogs.
Dirofilaria repens has been reported from Europe, Asia and Africa. In Africa it is widely distributed and is also endemic in South Africa, where it is common –particularly in the coastal areas of KwaZulu-Natal.
Transmission
Dirofilaria species have an indirect life cycle. Microfilariae produced by females appear in the blood. The presence of microfilariae of D. immitis and D. repens in the peripheral blood of dogs and cats is irregular and mainly nocturnal. Only 20% of D. immitisinfected cats become microfilaraemic and then only for a very short period.
Various culicine and anopheline mosquitoes act as intermediate hosts (vectors) for both Dirofilaria spp. Female mosquitoes become infected by obtaining blood meals from microfilaraemic dogs and cats. In the vector, microfilariae develop to infective L3-stages (metacyclic larvae), which eventually accumulate in the proboscis. The incubation period in vectors is largely temperature-dependent and may be as short as 10 days.
Definitive hosts become infected during feeding by the vectors when the metacyclic larvae actively leave the labella of the proboscis and enter the puncture wound on their own.
In the definitive hosts the metacyclic larvae of D. immitis develop further during a complex migration through various body tissues and reach the predilection sites after 70-120 days.
The migratory route of metacyclic larvae of D. repens and their further development in definitive hosts is unknown. The prepatent period is 6-9 months for D. immitis and 6-8 months for D. repens . Filarial helminths of the genus Dirofilaria are long-lived. Similarly microfilariae in the blood remain viable and infective for up to 2 ½years.
Socio-economic importance
In endemic and particularly hyperendemic areas, heartworm is one of the most important helminths of dogs and cats, due to its severe pathogenic effects and the financial implications for owners.
Diagnosis in live animals requires specialist diagnostic facilities, which are expensive as is treatment. The availability of drugs is also problematic in many endemic countries.
Dirofilaria immitis infection of humans is very rare with about 230 cases reported worldwide and it is mostly asymptomatic. Adult worms are mainly found in the lungs, and present incidentally as ‘coin lesions ’during chest radiography. However, Dirofilaria repens infection of humans is more common and probably underdiagnosed and under-reported. In the more common superficial form, worms are found in nodules in the subcutaneous and submucosal connective tissue of the head, chest and upper limbs. The head, eyelids and conjunctivae are most frequently affected. Nodular lesions in the less common visceral form can be misdiagnosed as neoplastic growths.
Pathogenesis and pathology
In cardiovascular dirofilariosis ( Figure 67 ), the pathogenic stages are the pre-adult and adult worms, also referred to as macrofilariae –at the predilection sites. Microfilariae are largely non-pathogenic. Depending on worm burden, duration of infection and host response, cardiovascular dirofilariosis ultimately develops into a multisystemic disorder which affects the lungs, heart, liver and kidneys. Inflammatory reactions in the pulmonary arterial system and associated mechanical effects interfere with the blood flow which eventually results in pulmonary hypertension and right-sided heart failure, causing chronic venous congestion. Chronic venous congestion is often accompanied by ascites, oedema of limbs, hydrothorax and hydropericard.
In the past, Dirofilaria repens has been regarded as non-pathogenic in dogs and cats. However, there are several reports of pruritic dermatitis which are thought to be caused by microfilariae and the movement of adults in the subcutaneous tissue. Protruding nodules in the skin of infected animals are sometimes noticed.
Clinical signs
Cardiovascular dirofilariosis of dogs and cats can go clinically unnoticed. Clinical signs associated with heartworm infection are principally a reflection of the macrofilarial worm burden, duration of infection, and host-parasite interaction. Clinically, cardiovascular dirofilariosis can be classified into various forms:
- Mild, asymptomatic form which is mostly detected incidentally.
- Moderate form, which presents with exercise intolerance, chronic cough, dyspnoea, and progressive weight loss.
- Severe form characterised by rightsided congestive heart failure, syncope, an acute or chronic vena cava syndrome, and sudden death (particularly in cats).
Cutaneous dirofilariosis of dogs and cats, in contrast, is largely asymptomatic and non-pathogenic. Adults can sometimes provoke the formation of protruding nodules in the skin. Microfilariae and migrating adults are known to cause a pruritic pseudo-eczematous dermatitis in some dogs.
Diagnosis
The diagnosis of cardiovascular dirofilariosis in live animals is problematic, due to the long prepatent period of D. immitis .
Presumptive diagnosis: The typical clinical signs, and a history of the animal originating or having stayed temporarily in an endemic or suspected endemic area, are indicative of cardiovascular dirofilariosis.
Parasitological diagnosis: The parasitological diagnosis in live dogs is based on the demonstration of unsheathed microfilariae in blood. Microfilariae can be detected in wet blood films, stained thin and thick blood films, and by the capillary haematocrit tube method. For increased sensitivity, concentration techniques such as membrane filtration, preferably, or the modified Knott ’s test, should be used. To differentiate between the microfilariae of the two Dirofilaria species and microfilariae of the genus Acanthocheilonema , histochemical staining for acid phosphatase activity is required. Acid phosphatase staining is considered the most reliable, consistent and practical technique for species identification of microfilariae in dogs and cats ( Table 10 ).
Dealing with the differential diagnosis and interpretation of blood from infected animals may be challenging and should be done by diagnosticians with specialist training. When interpreting the findings, infections with filariids of the genus Acanthocheilonema and Brugia should also be taken into consideration. Acanthocheilonema reconditum and Acanthocheilonema dracunculoides have unsheathed microfilariae that appear in blood and are similar to the Dirofilaria spp. The microfilariae of Brugia species are sheathed. With the exception of Brugia patei , which is endemic in Kenya and Tanzania, all other Brugia species found in dogs and cats –namely Brugia malayi, Brugia pahangi and Brugia ceylonensis –are endemic and limited to Asia, and should be considered in imported animals presenting with signs of heartworm disease.
Cats infected with heartworm are usually amicrofilaraemic and the confirmation of the final diagnosis largely depends on serology.
Antigen detection methods: enzymelinked immunosorbent (ELISA) and immunochromatographic assays are available. These methods have the advantage of detecting occult (amicrofilaraemic), patent infections. However, their lack of sensitivity poses a problem in areas of low prevalence and in newly-colonised areas. In these cases, the use of concentration techniques is recommended. The tests are also not specific enough to exclude false positives due to D. repens infections.
Serological tests are used in cats due to their amicrofilaraemic infections and low worm burdens.
Clinical diagnosis: Further clinical diagnostic procedures such as radiography and echocardiography might also be required.
Differential diagnosis
The clinical differential diagnosis is dependent on the parasite species and the stage of development of the disease.
Control
Treatment: Cardiovascular dirofilariosis in dogs is treated initially by administering macrofilaricides (adulticides), followed by the application of microfilaricides.
Table 10 Comparative morphology of Dirofilaria spp. of dogs and cats
| D. immitis | D. repens | |
|---|---|---|
Adult stages
| 21-31 cm x 1.0-1.3 mm 12-20 cm x 0.6-0.9 mm 300-355 μm (l) and 175-229 μm (r) | 8.4-17.0 cm x 0.38-0.65 mm 3.9-7.0 cm x 0.27-0.45 mm 338-590 μm (l) and 123-206 μm (r) |
Microfilariae
| Unsheathed 180-340 μm x 5-7 μm Excretory and anal pore | Unsheathed 207-385 μm x 5-9 μm Anal pore or anal pore and inner body |
The arsenical melarsomine is currently the drug of choice in dogs. Treatment may cause systemic reactions precipitated by disintegrating worms, which may necessitate supportive treatment with anti-inflammatories and parenteral fluids. This is a problem –particularly in cats.
The concurrent administration of the antibiotic doxycycline is recommended in conjunction with melarsomine, because it targets the Wolbachia endosymbionts of D. immitis .
Microfilaricidal therapy should be applied 3-4 weeks after completion of the macrofilaricidal therapy in dogs. The macrocyclic lactones ivermectin, milbemycin, moxidectin and selamectin are suitable for this purpose. As with the macrofilarial treatment, severe systemic reactions may occur and therefore treated animals should be hospitalised and closely monitored for 12 hours following treatment.
To prevent establishing cardiovascular dirofilarosis in non-endemic areas, infected dogs should be quarantined in mosquito-proof cages during treatment.
Due to the largely non-pathogenic nature of D. repens , treatment is not recommended.
Control: Prophylactic treatment for heartworm is indicated for those dogs and cats living in or travelling to an endemic area. The macrocyclic lactones ivermectin, milbemycin oxime, moxidectin and selamectin given monthly –are suitable for this purpose.
Cardiovascular dirofilariosis is a notifiable disease in South Africa. To prevent D. immitis from becoming endemic in South Africa, imported dogs are quarantined and screened for infection.
Whipworms (Trichuriosis)
Trichuris species
General
Trichuriosis of dogs is caused by the nematode Trichuris vulpis . Feline trichuriosis is caused by Trichuris campanula and Trichuris serrata , both of which are comparatively rare and have only been described in the Americas, some Caribbean Islands, and Australia. The parasites typically occur in the caecum and colon.
The genus name is derived from the Greek words ‘trichos ’ (= hair) and ‘ura ’ (= tail), which, like the common name ‘whipworm ’, are descriptive and refer to the peculiar filament-like, slender cephalic end and a thick caudal end. Males of T. vulpis are 45-60 mm long and female worms 62-75 mm long. The anterior part, which constitutes ⅔ of the body length, is 0.1-0.2 mm wide, whereas the posterior part is about 0.5 mm wide.
Microscopically, the presence of a stylet protruding from the small buccal capsule can be seen. The buccal capsule leads into a long typical trichuroid oesophagus. The posterior end of the male is coiled and there is a single spicule, 8.5-11.0 mm long. The spicule is surrounded by a protrusible, spinous sheath. The female caudal end is blunt and the vulva is located at the level of the end of the oesophagus.
Eggs of T. vulpis are medium-sized (70- 93 μm by 37-40 μm), characteristically lemon-shaped, yellowish-brown and have a thick, smooth shell with prominent projecting plugs at both poles.
Hosts
Dogs and a range of sylvatic canids are the definitive hosts of T. vulpis. All ages are susceptible. The effects of trichuriosis are more severe under poor husbandry conditions. However, clinical trichuriosis also occurs in well-kept and regularly dewormed dogs, often as the result of being exposed to a heavily infected environment which is a source of continual reinfection.
Distribution
Trichuris vulpis has a cosmopolitan distribution.
Transmission
Trichuris vulpis has a direct life cycle. The females are oviparous and the eggs are voided with the faeces into the environment. Trichuris spp. eggs are highly resistant and can remain viable in the environment for 4-5 years.
Depending on the ambient temperature, infective L1-larvae develop in the egg within 9-10 days.
Dogs become infected by ingesting larvated eggs, either via contaminated food, drinking water, by coprophagia, or by grooming the coat of infected dogs. The ingested larvae hatch and migrate into the mucosa of the small and large intestines (histotropic phase) for 10 days –during which time they develop to L2. Second-stage larvae migrate back into the lumen of the caecum and colon, where they remain attached to the mucosa and mature to adults. The prepatent period is 2-3 months.
Socio-economic importance
Trichuris vulpis is an important nematode of dogs, although they are less commonly encountered than hookworms and ascarids. Because of the long prepatent period, patent infections are only seen in animals older than 3 months.
Trichuris vulpis is not a zoonosis. Trichuriosis of humans is caused by Trichuris trichiura.
Pathogenesis and pathology
The lumenal developmental stages are haematophagic and they burrow with their anterior ends into the caecal and colonic mucosa.
The worms are not stationary, but continuously change their site of attachment. Small petechial haemorrhages with blood oozing into the lumen, catarrhal inflammation with small epithelial defects, hyperaemia, and oedema are caused by the trauma inflicted by the worms attaching to the mucosa. Haemorrhagic inflammation with intestinal contents containing blood can be observed when large worm burdens occur. Although the worms are haematophagic, the amount of blood withdrawn by individual worms (±5 μl blood/worm/day) is negligible. The anaemia seen in these cases is most likely the result of indirect blood loss due to haemorrhage from the injured mucosa where the worms attach.
Clinical signs
Dogs mostly harbour low worm burdens, and the infection remains clinically inapparent. Build-up of large worm burdens, particularly in older dogs, is not uncommon and they may then present with severe clinical signs reflecting typhlitis and colitis. They manifest abdominal pain, chronic diarrhoea alternating with constipation, tenesmus, and vomiting. Stools are mucoid and often contain blood.
There are also signs of chronic weight loss, dehydration, a dull coat, anaemia, and inappetence. The outcome is rarely fatal.
Diagnosis
Presumptive clinical diagnosis: A history of chronic diarrhoea alternating with constipation, the voiding of mucoid bloodtinged stools, and weight loss is indicative of trichuriosis.
Parasitological diagnosis: The parasitological diagnosis in live animals is based on the detection of eggs in faecal samples by means of direct flotation. Direct flotation is more sensitive when flotation fluids with a high specific gravity of 1,3 is used (saturated MgSO4 or ZnSO4 solution).
Differential diagnosis
Other causes of diarrhoea that should be considered include helminth infections (toxocarosis, trichuriosis), giardiosis, viral gastroenteritis (parvovirus and coronavirus infections), bacterial infections, poor nutrition, and side effects of drugs.
When doing faecal examinations, Trichuris eggs should be distinguished from those of the Capillaria spp. Capillaria eggs are colourless and have less prominent projecting ‘plugs ’at either pole. Depending on the species, they are either barrelshaped with sides almost parallel, or lemon-shaped. False positive faecal flotation results can be obtained when animals ingest trichurid eggs with prey –for example rodents or as a result of coprophagia.
Control
Treatment: There are a number of anthelmintics from the various anthelmintic groups that are effective when used for the treatment of trichuriosis in dogs. Most drugs are used in combinations and also together with specific cestocides such as praziquantel.
The following are effective:
- Benzimidazoles: Fenbendazole
- Probenzimidazoles: Febantel
- Tetrahydropyrimidines: Oxantel
- Macrocyclic lactones: Milbemycin, Moxidectin
Control: See the section on the control of toxocarosis for general control principles.
Cestodes
Tapeworms (Taeniosis)
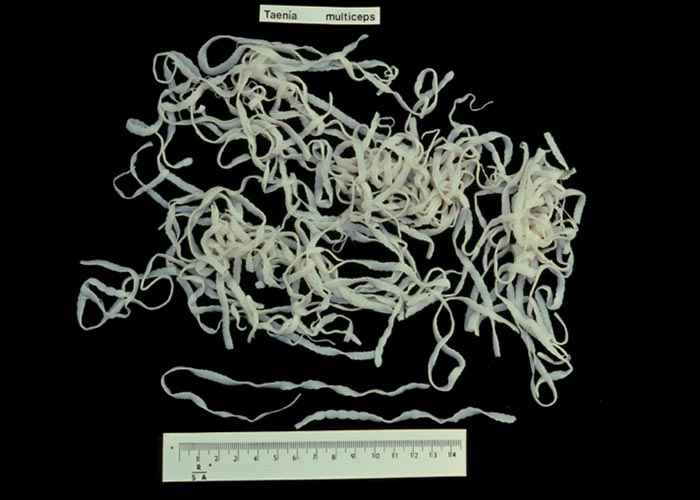
Taenia spp.
General
Taeniosis of domestic carnivores is caused by the strobilar stages of Taenia hydatigena, Taenia pisiformis, Taenia ovis, Taenia multiceps, Taenia serialis and Taenia taeniaeformis. The Taenia spp., together with the Echinococcus spp. in the family Taeniidae , are collectively referred to as taeniids. These parasites share many characteristics –particularly with respect to their morphology, life cycle, and epidemiology. The small intestine is the predilection site of the strobilar stages of Taenia hydatigena, Taenia pisiformis, Taenia ovis, Taenia multiceps, Taenia serialis, and Taenia taeniaeformis .
The genus name Taenia is derived from the Greek word ‘tainia ’ , which was later latinized to ‘taenia ’ , meaning tape. Taenia spp. are typically large tapeworms and the species found in dogs and cats range from 0.2 m to 2.5 m in length. The scolex of the Taenia spp. of dogs and cats has four suckers and a rostellum armed with one row of small, and one row of large hooks. The size and number of the rostellar hooks are of diagnostic importance and allow identification to species level. The strobila is typically large and consists of up to several 100 proglottids. Mature proglottids are rectangular, wider than long and are provided with one set of reproductive organs. Genital pores are marginal and alternate irregularly. Gravid proglottids are also rectangular, but longer (1-2 cm) than wide (0.5-1.0 cm), and contain a uterus with a median stem and lateral diverticula. Depending on the species, gravid proglottids contain between 15 000 and 100 000 eggs each.
Comparative information on the Taenia spp. of dogs and cats is presented in Table 11 .
The eggs of all species in the family Taeniidae (including Echinococcus spp.) are indistinguishable morphologically and morphometrically. Collectively they are referred to as taeniid-type eggs or taeniid eggs. Taeniid eggs are spherical, about 40 μm in diameter and consist of an oncosphere (first larval stage), which is surrounded by a 5 μm-thick striated embryophore.
The types of metacestode (second larval stage developing in intermediate hosts and infective stage for definitive host) in the life cycle of Taenia spp. are the cysticercus, coenurus and strobilocercus. Macroscopically, the metacestode types have no or small similarities with the intestinal strobilar stages. Descriptive scientific names were allocated to the metacestodes of some Taenia spp. developing in intermediate hosts, and although no longer regarded as valid, they are still much in use in the literature and meat-inspection regulations of various countries ( Table 11 ).
Hosts
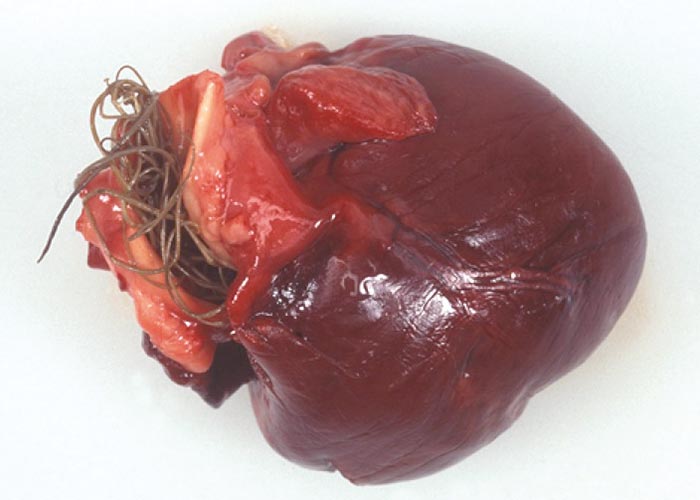
Apart from dogs and cats, the Taenia spp. covered in this chapter have a wide range of definitive hosts, particularly amongst sylvatic canids and to a lesser extent in felids. Regarding domestic carnivores, T. hydatigena, T. ovis and T. multiceps are only found in dogs and T. taeniaeformis is only encountered in cats. Taenia pisiformis is found in dogs and rarely in cats. Taenia serialis is a parasite of both dogs and cats. In South Africa, infections with T. hydatigena, T. multiceps and T. taeniaeformis are most commonly seen ( Figures 68 - 71 ).
Table 11 Comparative characteristics of Taenia spp. of dogs and cats
| T. hydatigena | T. pisiformis | T. ovis | T. multiceps | T. serialis | T. taeniaeformis | |
|---|---|---|---|---|---|---|
| Strobila r stage (length) | 50-250 cm | 36-200 cm | 45-146 cm | 21-120 cm | 20-27 cm | 15-60 cm |
| Scolex (number of large and small hooks) | 26-44 | 34-48 | 24-38 | 11-34 | 26-34 | 26-52 |
| Large hooks (length) | 169-224 μm | 200-294 μm | 156-197 μm | 120-185 μm | 110-177 μm | 294-485 μm |
| Small hooks (length) | 110-160 μm | 114-177 μm | 96-138 μm | 73-160 μm | 63-129 μm | 187-293 μm |
| Number of lateral diverticula of uterus | 5-10 | 8-20 | 11-25 | 9-26 | 8-25 | 5-11 |
| Definitive hosts | Dog | Dog, cat (rare) | Dog | Dog | Dog, cat | Cat |
| Common intermediate hosts | Sheep, goat, pig | Lagomorphs | Sheep, goat | Sheep, goat | Lagomorphs, rodents | Rodents |
| ‘Scientific name ’of metacestode | Cysticercus tenuicollis | Cysticercus pisiformis | Cysticercus ovis | Coenurus cerebralis | Coenurus serialis | Strobilocercus fasciolaris |
| Morphology of metacestode | Semitransparent, chicken eggsized cyst with single invaginated scolex | Pea-sized cyst with single invaginated scolex | Pea-sized cyst with single invaginated scolex | Semitransparent, chicken egg-sized cyst with several invaginated scoleces arranged in random groups | Walnut-sized with several invaginated scoleces arranged in crowded lines | Strobilar-like connected to small terminal bladder, enclosed in cyst about 1 cm in Ø |
| Predilection site of metacestode in intermediate hosts (IH) | Abdominal serous surfaces | Abdominal serous surfaces | Heart, diaphragm, skeletal musculature | Brain (sheep), subcutaneous and intermuscular connective tissue (goat and other IH) | Subcutaneous and intermuscular connective tissue | Liver |
Distribution
All listed Taenia spp. have a cosmopolitan distribution.
Transmission
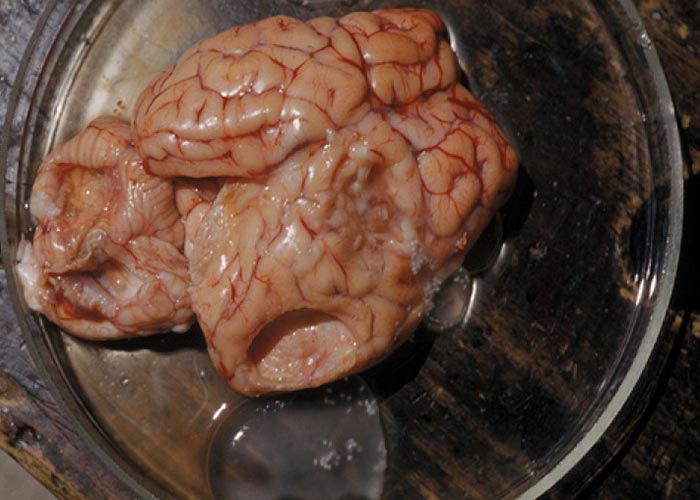
All Taenia spp. have an indirect life cycle, which requires two mammalian hosts for completion. Taenia spp. have a high biotic potential; a single gravid proglottid can contain 15 000 to 100 000 eggs. Gravid proglottids and eggs already released in the gut are voided with the faeces. However, the majority of proglottids appear to leave the host spontaneously –since gravid proglottids of Taenia spp. are motile. This implies that an infected definitive host can contaminate its environment with eggs without defaecation. Insects (especially flies), earthworms, birds, rainfall and sewage are also important for the dispersal of eggs. Embryonated eggs in gravid proglottids are immediately infective for intermediate hosts. In a suitable environment, taeniid eggs are highly resistant to physical factors and can remain infective for up to one year. Intermediate hosts become infected by ingesting eggs. Liberated oncospheres enter small blood vessels in the intestinal wall and eventually reach their predilection sites via the blood stream, and then develop into metacestodes ( Table 11 ). Metacestodes become infective for definitive hosts 2-3 months after ingestion of eggs. Dogs and cats as definitive hosts eventually become infected by ingestion of metacestodes. The prepatent period in dogs and cats is 1-3 months. Strobilar stages of Taenia spp. are long-lived; the patent period can be several years.
Figures 68 and 69 Taenia hydatigena tapeworm and the metacestode (C. tenuicollis) in a herbivore liver.
Figures 70 and 71 Taenia multiceps tapeworm and the metacestode (C. cerebralis) in the brain of a sheep.
Immunity appears not to develop in infections with Taenia spp. –as both dogs and cats become readily re-infected.
Socio-economic importance
The cestode family Taeniidae has the greatest relevance in public health. For aesthetic reasons or because of zoonotic implications, infected carcasses or organs of intermediate hosts (sheep, goat, cattle, water buffalo, pigs, rabbits and game) are downgraded, destroyed or confiscated. Humans can potentially act as accidental intermediate hosts for T. multiceps and T. serialis by ingesting eggs. Sources of infection are direct contact with infected dogs and/or cats as well as contaminated drinking water and food. Coenuri may develop in the brain, eye (T. multiceps) ( Figure 71 ) and the subcutaneous or intermuscular connective tissues (T. serialis, T. multiceps) .
Pathogenesis and pathology
See Echinococcosis.
Clinical signs
See Echinococcosis.
Diagnosis
See Echinococcosis.
Differential diagnosis
Other tapeworm infections such as echinococcosis, dipylidiosis, joyeuxiellosis and mesocestoidosis should be considered.
Control
Treatment: There are several anthelmintics registered for the treatment of taeniosis in dogs and cats. None of the listed drugs has ovicidal properties:
- Isoquinolines: Praziquantel
- Isothyocyanates: Nitroscanate (dogs only)
- Diphenylmethanes: Dichlorophen
- Salicylanilides: Niclosamide
- Benzimidazoles: Fenbendazole
Control: See Echinococcosis .
Echinococcus (Echinococcosis)
General
Echinococcus granulosus is the principal cause of canine echinococcosis worldwide. Together with the Taenia spp., the Echinococcus spp. are grouped in the family Taeniidae and are collectively referred to as taeniids. Many characteristics are shared between the two genera, particularly with respect to morphology, life cycle and epidemiology.
The genus name Echinococcus is derived from the Greek word ‘echinos ’ (= hedgehog, hook) which refers to the rostellar hooks, and the Greek word ‘kokkos ’ (= pip, seed) which refers to the appearance and small size of the tapeworms. Echinococcus granulosus , similar to other species within the genus, does not exceed 7 mm in length. The scolex of Echinococcus spp. has four suckers and a rostellum, which is armed with one row of small hooks and one row of large hooks. The strobila usually consists of 3 proglottids, which have similar morphological features to Taenia spp. The penultimate proglottid is the mature one and the terminal proglottid is the gravid one. Gravid proglottids are longer (2-3 mm) than wide (<1 mm) and are difficult to see macroscopically. Gravid proglottids contain 600-1 500 eggs.
The eggs are typically taeniid and cannot be differentiated morphologically or morphometrically from other Echinococcus spp. and Taenia spp. Taeniid eggs are spherical, about 40 μm in diameter, and consist of an oncosphere (first larval stage), which is surrounded by a 5-μm thick striated embryophore( see taeniosis) .
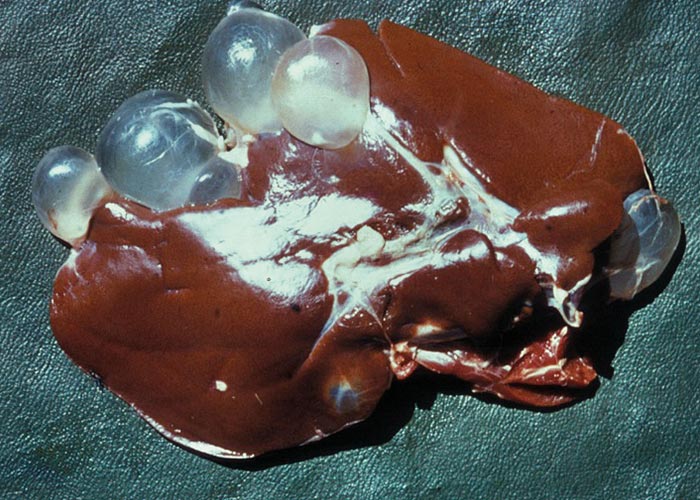
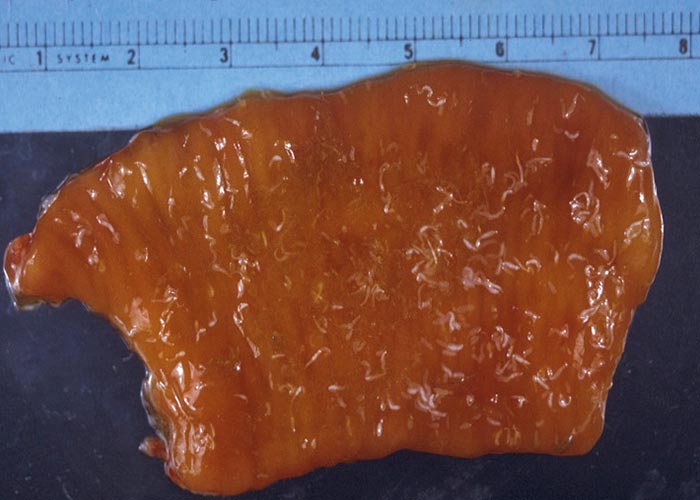
The type of metacestode (second larval stage developing in intermediate hosts and infective stage for definitive host) in the life cycle of E. granulosus is a hydatid (hydatid cyst) which is a unilocular, fluidfilled bladder with a thin inner germinal layer, a thick outer acellular laminated layer, and a host-derived adventitial layer ( Figure 73 ). The germinal layer generates brood capsules which generate protoscoleces asexually. The size of hydatids can range from 1 to 20 cm in diameter; however, the usual size is 5-10 cm. The cyst fluid of fertile hydatids contains hydatid sand which consists of free-floating protoscoleces, brood capsules, detached hooklets, and calcareous bodies.
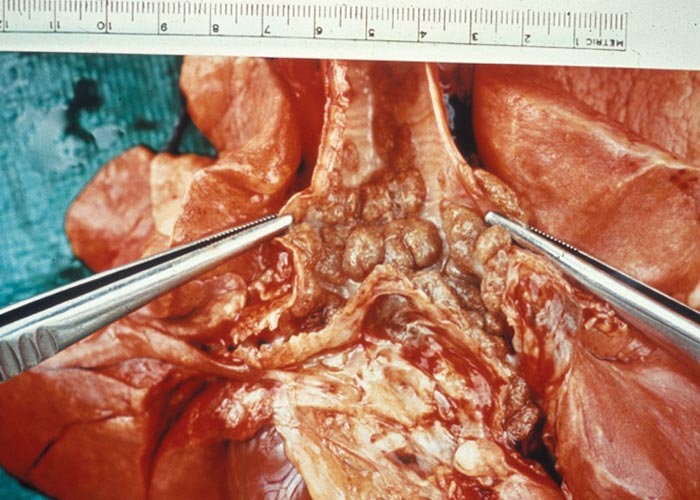
The small intestine, as with most tapeworms, is the predilection site of the strobilar stage of E. granulosus ( Figure 72 ).
Since asexual reproduction occurs in hydatid cysts, high worm burdens with thousands of specimens are common in dogs.
Hosts
E. granulosus has a wide range of definitive hosts amongst sylvatic canids and felids, in addition to domestic dogs. Domestic cats, however, are not susceptible.
Distribution
E. granulosus has a cosmopolitan distribution.
Transmission
Echinococcus spp. have an indirect life cycle which requires two mammalian hosts for completion. Compared to Taenia, Echinococcus spp. have a far lower biotic potential. A single proglottid of E. granulosus contains 600-1 500 eggs and only a few oncospheres eventually develop into fertile hydatid cysts in intermediate hosts. Gravid proglottids contain eggs that are already released in the gut and voided with the faeces. Gravid proglottids of E. granulosus are motile, which implies that a definitive host can contaminate its environment without defaecation. Eggs are dispersed by insects (flies, beetles, ants), earthworms, birds and rainfall. As in Taenia , eggs are already embryonated and immediately infective for intermediate hosts. Echinococcus eggs are highly sensitive to desiccation. At low temperatures (10 °C) and sufficient humidity, they can remain viable for at least 7 months.
Domestic herbivorous and omnivorous ungulates including sheep, goats, cattle, water buffalo, camels, horses, and pigs act as the principal intermediate hosts. Intermediate hosts become infected by ingesting eggs. Liberated oncospheres enter small blood vessels in the intestinal wall. Many oncospheres are retained in the liver while others pass on further to settle in the lungs and less in other organs where they develop into hydatids. Hydatids become infective for definitive hosts no sooner than 6 months after ingestion of eggs. The development of protoscoleces does not depend on cyst size. In horses, fertile hydatids as small as 2 mm in diameter can be found. Dogs as definitive hosts become infected by ingestion of fertile hydatid cysts. The prepatent period is 33-58 days. The patent period is up to one year. In hyperendemic areas, dogs develop a partial immunity –with higher prevalences and worm burdens in young dogs.
Socio-economic importance
Cystic echinococcosis (CE), also known as cystic hydatid disease or hydatidosis, is the result of humans becoming accidentally infected by ingesting eggs of E. granulosus. Cystic echinococcosis can be regarded as the most important and widespread zoonotic disease of canine origin. Humans act as intermediate hosts with the metacestode developing mostly in the liver (>50%), followed by the lungs and less so in other sites such as the spleen, kidneys, central nervous system, subcutaneous connective tissue, and bone. Hydatid cysts typically grow slowly to a large size in humans and can be up to 25 cm in diameter. Infection is often asymptomatic with infections diagnosed on routine examination. Abdominal distension is commonly noticed in advanced cases. Hydatids developing in the central nervous system cause intracranial pressure and paralysis. Sudden rupture of hydatids and spillage of cyst fluid into body cavities may culminate in a fatal anaphylactic reaction. The most common sources of infection are direct contact with infected dogs and contaminated drinking water and food (especially vegetables and fruit). In areas where hydatidosis is prevalent, the financial implications of the expense of diagnosis and particularly treatment can be considerable for a health-care system.
Pathogenesis and pathology
Taenia and Echinococcus spp. are largely non-pathogenic in their definitive hosts. The strobilar stages attach to the mucosa of the small intestine by the rostellar hooks and suckers of the scolex. At these attachment sites, inflammatory infiltration –as well as desquamation and destruction of epithelial cells –can be noticed, although it is insignificant. Very high worm burdens (T. multiceps) can cause impaction of the small intestine, which is rare. Gravid proglottids of taeniids are motile and can leave the host spontaneously, which causes discomfort in the anal area.
Clinical signs
Taeniid infections in dogs and cats are mostly asymptomatic. Pruritus caused by gravid proglottids migrating out of the anus may cause animals to drag and rub their anus over the ground –a behaviour which is referred to as ‘scooting ’.
Diagnosis
Presumptive clinical diagnosis: Dogs and cats dragging and rubbing their anus over the ground can be indicative of tapeworm infection.
Parasitological diagnosis in live dogs and cats: Gravid proglottids of taeniids are motile and can be demonstrated on occasion in faeces, the perianal area or directly emerging from the anus. Gravid proglottids of Taenia spp. are whitish, longer than wide, 1-2 cm long and 0.5- 1 cm wide. Gravid proglottids of E. granulosus are also whitish and longer than wide, but only 2-3 mm long and <1 mm wide, and are very seldom noticed.
Eggs may be demonstrated following direct faecal flotation, on faecal smears or best by means of the adhesive-tape swab technique under the tail and in the perianal area. However, taeniid eggs cannot be differentiated morphometrically or morphologically. Taeniid eggs, particularly in dogs, can be those of Taenia or Echinococcus.
Coproantigen tests are highly specific and sensitive but, unfortunately, are not available commercially.
Diagnostic deworming with arecoline hydrobromide, applied as a drench or enema, is mainly used in surveys and during control campaigns for E. granulosus.
Parasitological diagnosis during necropsy: While Taenia spp. are easy to demonstrate, Echinococcus spp. are often overlooked because of their small size. For stereomicroscopic examination of intestinal content, small pieces of intestine as well as mucosal scrapings are required to positively identify carriers with low worm burdens. Considering the severe zoonotic implications of Echinococcus spp., necropsies conducted on dogs and sylvatic carnivores, as well as implementing the above-mentioned laboratory procedures, should adhere to safety precautions as recommended by WHO.
Differential diagnosis
Other tapeworm infections such as taeniosis, dipylidiosis, joyeuxiellosis and mesocestoidosis should be considered.
Control
Treatment: The isoquinoline praziquantel is the drug of choice for the treatment of echinococcosis in dogs. Although praziquantel is highly effective, treatment should be repeated to prevent residual worm burdens. Since no cestocidal drug, including praziquantel, has ovicidal properties, extreme care should be exercised when opting for treatment of infected dogs. Dogs should be confined to cages that can be cleaned and disinfected easily (concrete or tiled floors). Faeces passed up to 5 days after final treatment should be properly disposed of. After completion of treatment, dogs should be bathed to eliminate eggs attached to the fur, and cages should be disinfected. Personnel involved in the procedure should wear protective clothing.
Figures 72 and 73 Echinococcus granulosus tapeworm in a carnivore intestine and metacestode or hydatid cyst in the liver of a goat.
Control: In principle, control of canine taeniosis/echinococcosis is simple. The access to raw offal must be prevented. Abattoirs should strictly enforce the regulations of meat inspection. Infected organs should be confiscated and destroyed appropriately. Domestic carnivores, and, in particular dogs, should not have access to abattoirs.
Systematic community education in rural endemic areas is critical –where home slaughtering is practised and where dogs are utilised for herding and hunting. Feeding dogs on home-butchered domestic and hunted animals may result in high prevalence rates and massive contamination of the environment with taeniid eggs. Offal frozen at a temperature of -20 °C for at least 3 days, or, preferably cooked offal, is safe. In hyperendemic areas regular chemotherapy (every 5-6 weeks) as an individual prophylaxis can be considered, in order to prevent the establishment of patent infections with particularly Echinococcus in shepherd and hunting dogs. Taeniid eggs can be killed by temperatures of over 80 °C within a few minutes. Common disinfectants, including ethanol, propanol and formalin, have virtually no effect on taeniid eggs. Sodium hypochlorite at a concentration of 3.75% (undiluted household bleach or industrial bleach) is a suitable ovicide. The WHO has published detailed guidelines for the control of echinococcosis/ hydatidoses, which should be consulted thoroughly for the planning and execution of control programmes.
Dipylidium (Dipylidiosis)
General
Dipylidiosis of dogs and cats is caused by the strobilar stage of Dipylidium caninum. Together with the genus Joyeuxiella, Dipylidium belongs in the family Dipylidiidae and both genera share many characteristics with respect to morphology and life cycle. As for most tapeworms, the small intestine is the predilection site of the strobilar stage of D. caninum. Dogs and cats become infected by ingesting fleas or lice harbouring cysticercoids. The prepatent period is 2-3 weeks. The patent period is up to 3 years.
Dipylidium caninum is usually 0.2-0.5 m long and only rarely reaches 0.8 m. The scolex has four suckers and a conical rostellum, which is armed with 3-5 rows of rose-thorn-shaped hooks. The large hooks are ±13 μm long, and the small hooks ±7 μm. Mature proglottids are provided with 2 sets of reproductive organs, the genital pores are bilateral (Greek ‘pylos ’ = pore, Greek ‘di ’= two, hence the genus and family names Dipylidium and Dipylidiidae) and post-equatorial. Gravid proglottids are longer than wide and typically resemble cucumber seeds. They have a reddish tinge when freshly passed and are 7-12 mm long and 1.5-3 mm wide. Within gravid proglottids, the uterus breaks up and forms egg capsules.
The egg capsules are oval and large (120 x 200 μm), and contain 2-38 eggs. The eggs are spherical, 30-50 μm in diameter, and consist of an oncosphere, which is surrounded by a very thin, striated embryophore. Within the capsule, the eggs are covered by a vitelline membrane.
The type of metacestode (second larval stage developing in an intermediate host and infective stage for the definitive host) in the life cycle of D. caninum is a cysticercoid.
Hosts
In addition to dogs and cats, D. caninum has a wide range of definitive hosts that include wild canids, wild felids, and very occasionally humans
Distribution
Dipylidium caninum has a cosmopolitan distribution. It is the most common tapeworm of dogs and cats in many parts of the world.
Transmission
Dipylidium caninum has an indirect life cycle, which requires an arthropod and a mammalian host for completion.
Gravid proglottids containing egg capsules and egg capsules already released in the gut are voided with the faeces. As in the taeniids, the gravid proglottids of D. caninum are motile and can also leave the host spontaneously. The embryonated eggs contained in the egg capsules are immediately infective for intermediate hosts.
Fleas (Ctenocephalides felis and Ctenocephalides canis) which commonly infest dogs and cats, are the principal intermediate hosts (vectors). The human flea Pulex irritans and the chewing louse Trichodectes canis have also been identified as intermediate hosts. Eggs containing oncospheres are ingested by flea larvae or nymphal stages of T. canis . Released oncospheres penetrate the intestinal wall and develop into cysticercoids in the body fat. The development to cysticercoids is synchronised with the development of the vectors and is completed when fleas emerge from their cocoons. The incubation period in vectors is largely temperature-dependent, as is the flea development, and may be as short as 2-3 weeks.
Socio-economic importance
Humans can act as definitive hosts for D. caninum. Infection in humans is accidental and is caused by swallowing adult fleas, or as a result of dogs and cats licking the owner ’s face and thereby transferring crushed fleas. Infection is very rare and mainly seen in children because of their close contact with pets. Most cases are asymptomatic.
Pathogenesis and pathology
As in the taeniids, D. caninum is largely non-pathogenic in their definitive hosts. Gravid proglottids of D. caninum are motile and can leave the host spontaneously –causing discomfort in the anal area (see Echinococcosis ).
Clinical signs
Dipylidiosis of dogs and cats is mostly asymptomatic. Pruritus caused by gravid proglottids migrating out of the anus may cause animals to drag and rub their anus over the ground.
Diagnosis
Presumptive clinical diagnosis: Dogs and cats dragging and rubbing their anus over the ground can be indicative of tapeworm infection.
Parasitological diagnosis in live dogs and cats: Gravid proglottids of D. caninum are motile and can occasionally be detected in faeces, the perianal area, or directly emerging from the anus. Gravid proglottids of D. caninum have a reddish tinge when freshly passed, and are longer (7-12 mm) than wide (1.5-3 mm). Dried and distorted rice-grain-like proglottids have first to be rehydrated and broken up before the egg capsules (containing several eggs; egg capsules containing a single egg are those of Joyeuxiella spp.) can be demonstrated. Eggs capsules may be demonstrated following direct faecal flotation.
Differential diagnosis
Other tapeworm infections such as taeniosis, echinococcosis, joyeuxiellosis and mesocestoidosis should be considered.
Control
Treatment: The isoquinoline praziquantel is effective for the treatment of dipylidiosis in dogs and cats. Alternatively, the isothyocyanate nitroscanate can be used for treatment of dogs –but not cats.
Control: Control of fleas and lice is essential in order to prevent dipylidiosis of dogs and cats.
Joyeuxiella (Joyeuxiellosis)
General
Joyeuxiellosis of dogs and cats is caused by the strobilar stage of Joyeuxiella pasqualei, Joyeuxiella fuhrmanni and Joyeuxiella echinorhynchoides . The genus Joyeuxiella is named after the French parasitologist Charles Joyeux. Together with the genus Dipylidium, Joyeuxiella belongs in the family Dipylidiidae and both genera share many characteristics with respect to morphology and life cycle. As for most tapeworms, the predilection site of the strobilar stages is the small intestine.
Joyeuxiella spp. are 8-50 cm long. The scolex has four oval-shaped suckers and a conical rostellum (J. pasqualei, J. fuhrmanni) or a cylindrical rostellum with a bulbous anterior part (J. echinorhynchoides). The rostellum is armed with 8-30 rows of rose-thorn-shaped, tiny hooks –which decrease in size from the anterior towards the posterior end of the rostellum. Mature proglottids are provided with 2 sets of reproductive organs, and the genital pores are bilateral and pre-equatorial. Gravid proglottids are longer (1-6 mm) than wide (0.5-2 mm). Within gravid proglottids the uterus breaks up into egg capsules.
The egg capsules are spherical, 35-95 μm in diameter, and contain a single egg. Eggs are spherical, 28-50 μm in diameter, and consist of an oncosphere surrounded by a very thin, striated embryophore.
The type of metacestode (second larval stage developing in intermediate host and infective stage for definitive host) in the life cycle of Joyeuxiella spp. –is a cysticercoid.
Hosts
In addition to dogs and cats, the definitive host range of J. pasqualei and J. echinorhynchoides includes a variety of sylvatic felids and canids. Joyeuxiella fuhrmanni, is only found in cats and in sylvatic felids and canids.
Distribution
Joyeuxiella pasqualei has been reported from several countries in Europe, Asia, Africa (South Africa, Zambia and other countries) and Australia.
Joyeuxiella fuhrmanni has only been reported from Africa (South Africa, Zimbabwe, DRC).
Joyeuxiella echinorhynchoides has been reported from several countries in Europe, Asia and Africa (South Africa).
Transmission
Joyeuxiella spp. have an indirect life cycle, which requires three hosts for completion. Like D. caninum , gravid proglottids are motile and can also leave the host spontaneously. Embryonated eggs contained in gravid proglottids are immediately infective for intermediate hosts.The eggs are ingested by coprophagous beetles. Reptiles and small mammals act as paratenic (transport) hosts. Definitive hosts become infected, mostly by ingesting paratenic hosts.
Socio-economic importance
Joyeuxiella spp. are one of the most common cestode species of cats in some parts of the world. There are no reports of infections in humans.
Pathogenesis and pathology
See dipylidiosis .
Clinical signs
See dipylidiosis .
Diagnosis
See dipylidiosis .
Differential diagnosis
Other tapeworm infections such as taeniosis, echinococcosis, dipylidiosis and mesocestoidosis should be considered.
Control
Treatment: The isoquinoline praziquantel is highly effective for the treatment of the strobilar stage of Joyeuxiella spp. in dogs and cats.
Control: The control is problematic due to the large spectrum of, particularly, paratenic hosts.
Mesocestoides (Mesocestoidosis)
General
Mesocestoidosis of domestic dogs and cats is caused by species of the tapeworm genus Mesocestoides. No reference is made to individual species since their taxonomic status is uncertain. As for most tapeworms, the predilection site of the strobilar stages is the small intestine. Mesocestoides spp. are medium-sized tapeworms ranging from 0.3-1.5 m in length. The scolex has four suckers and no rostellum. Mature proglottids are wider than long, and gravid proglottids are usually longer than wide. There is a single set of reproductive organs in each mature proglottid and an inconspicuous ventromedian genital pore. Gravid proglottids are barrel-shaped or club-shaped and are 4-6 mm long and 2-3 mm wide. They have a conspicuous par-uterine organ, which is 0.5 mm in diameter.
The eggs are referred to as mesocestoidid- type eggs or mesocestoidid eggs. Mesocestoidid eggs are subspherical, medium-sized (60 x 40 μm) and consist of an oncosphere surrounded by a very thin embryophore.
The type of larval stage developing in the assumed first intermediate host is unknown. The metacestode developing in the second intermediate host is known as a tetrathyridium. Tetrathyridia have a tapelike, unsegmented body with an invaginated scolex provided with four suckers. The length ranges from 1 mm to 70 mm.
Hosts
In addition to dogs and cats, the definitive host range includes sylvatic carnivores, birds, and occasionally humans.
Distribution
Mesocestoides spp. have a cosmopolitan distribution.
Transmission
Mesocestoides spp. have an indirect life cycle, which requires three hosts for completion.
Mostly gravid proglottids containing eggs are voided with the faeces. Knowledge of the life cycle is incomplete. A diverse range of vertebrates such as amphibians, reptiles, birds and mammals are known to act as intermediate hosts which, however, cannot become directly infected by eggs. Therefore, the existence of a first intermediate host is assumed –in which the oncosphere develops into an unknown larval stage for the second intermediate host. The metacestode developing in the second intermediate hosts is a tetrathyridium which can reproduce asexually. Tetrathyridia are most commonly found in the abdominal cavity but are also in other body cavities, the liver, lungs and other sites. Infection in second intermediate hosts is also referred to as peritoneal or larval mesocestoidosis.
Definitive hosts become infected by ingestion of tetrathyridia in intermediate hosts –where they develop into strobilar stages in the small intestine. Tetrathyridia can also invade the abdominal cavity by penetrating the gut and continue to reproduce asexually or settle in various organs. The prepatent period in dogs and cats is approximately 3 weeks.
Socio-economic importance
Humans can become infected accidentally by ingesting tetrathyridia in raw organs of various intermediate hosts. Most cases have been described from Asia and seem to have originated from snakes.
Pathogenesis and pathology
The strobilar stages of Mesocestoides spp. are largely non-pathogenic in dogs and cats. Migrating tetrathyridia may illicit inflammatory reactions with severe peritonitis and development of granulomas in affected organs.
Clinical signs
Mesocestoidosis of dogs and cats is mostly asymptomatic. Peritoneal (larval) Mesocestoides infection caused by invading tetrathyridia can be severe, with animals presenting with abdominal distension, anorexia, apathy, fever, vomiting, diarrhoea, and weight loss.
Diagnosis
Parasitological diagnosis of strobilar infections in live dogs and cats: Gravid proglottids are rarely seen in faeces. Gravid proglottids of Mesocestoides spp. are barrel- or club-shaped, and up to 4-6 mm long and 2 to 3 mm wide. Even in unstained gravid proglottids, the characteristic par-uterine organ (diameter 0.5 mm) is clearly visible. Eggs in faeces are only rarely detected successfully.
Parasitological diagnosis of peritoneal (larval) mesocestoidosis in dogs and cats: Diagnosis is based on the demonstration of tetrathyridia in abdominal exudate following abdominocentesis.
Differential diagnosis
Other tapeworm infections such as taeniosis, echinococcosis, dipylidiosis, and joyeuxiellosis should be considered.
Control
Treatment: The isoquinoline praziquantel given at the recommended dose rate is effective for the treatment of mesocestoidosis.
Peritoneal mesocestoidosis requires longterm treatment with the benzimidazole fenbendazole.
Control: No control measures can be suggested due to the incomplete knowledge of the life cycle and the large spectrum of intermediate hosts harbouring tetrathyridia.