Helminths of horses
This content is distributed under the following licence: Attribution-NoDerivs CC BY-ND  View Creative Commons Licence details here
View Creative Commons Licence details here

Helminths of horses
Author: E VOLKER SCHWAN
Equine helminth infections are probably less economically important now than they were several decades ago when horses were used extensively for transport and farming. In South Africa, helminth infections are now of importance mainly in horses owned by rural communities which are dependent on work horses, and also horses used for sport and leisure activities.
The use of macrocyclic lactones has resulted in the large strongyles becoming a less important cause of colic in horses –while small strongyles are now of greater importance. Apart from the strongyles and helminths that cause mortalities in foals, most of the other helminth infections either occur sporadically, or have a limited pathogenic impact.
Nematodes
Large strongyles (Strongylosis)
Strongylus species
General
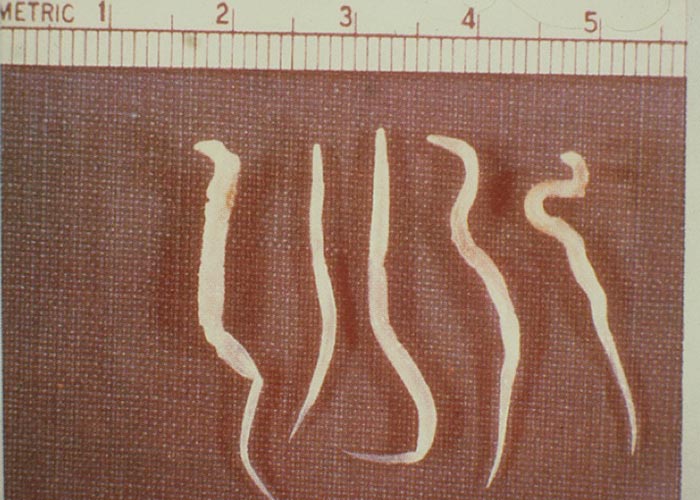
Strongylosis of horses is caused by Strongylus vulgaris, Strongylus edentatus, and Strongylus equinus . These species are collectively also referred to as ‘large strongyles ’, which, in contrast to the ‘small strongyles ’of horses –are migratory (termed ‘migratory strongyles ’). The genus name derives from the Greek word ‘strongylos ’ (= rounded) –referring to the cylindrical shape of the worms. Adult worms are medium-sized and stoutbodied. Microscopically, the species have a large buccal capsule, which can be armed with teeth. Typically, the buccal capsule is surrounded by leaf crowns. The posterior end of males terminates in a typical, well-developed, strongylate bursa with spicules.
Strongylus vulgaris male worms are 10 to 19 mm long and female worms 13 to 25 mm long. Both are 1.0 to 1.4 mm wide. The oval buccal capsule is armed with two typically ear-shaped teeth at its base.
Strongylus edentatus male worms are 19 to 28 mm long and female worms 28 to 45 mm long. Both are 1.3 to 2.2 mm wide. As the species name indicates, Latin ‘e ‘(= without) and ‘dens ’(= tooth) –the cupshaped buccal capsule is ‘toothless ’.
Strongylus equinus male worms are 24 to 35 mm long and female worms 35 to 50 mm long. Both are 1.1 to 2.3 mm wide. The oval buccal capsule is armed with one dorsal tooth with a bifid tip and also two sub-ventral teeth at its base.
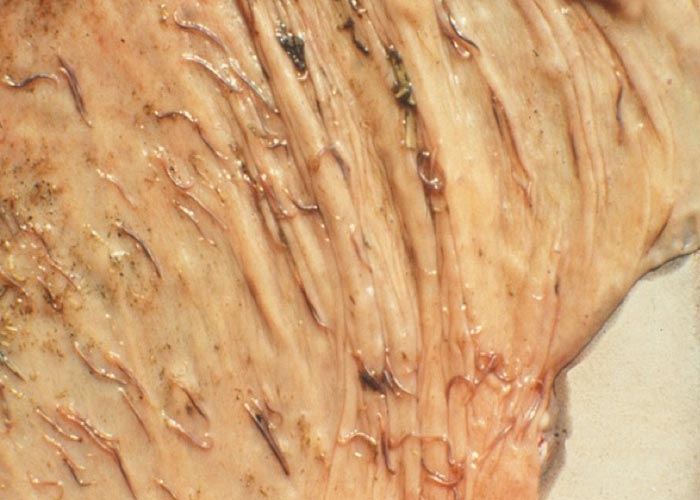
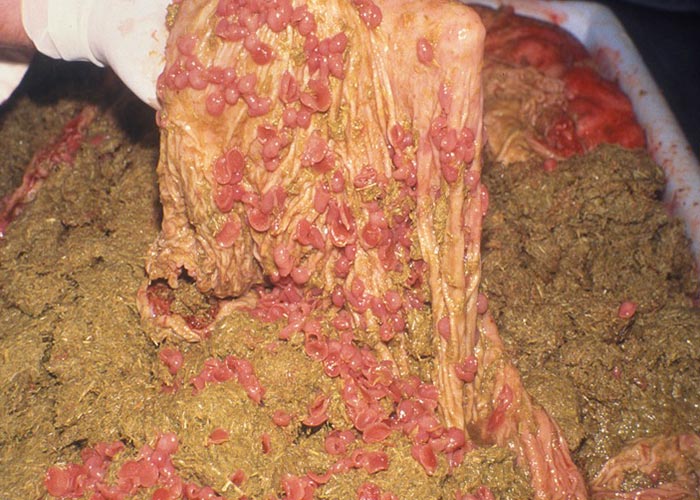
Fresh adult strongyles are dark red, and are sometimes almost black in colour. The predilection site in the definitive host is the caecum and colon ( Figure 52 ).
The eggs of the large strongyles are of the strongylid-type, which are oval and thin-shelled. The surface of the egg is smooth. They contain 4 to 8 blastomeres when laid, and these measure 64-99 x 36-58 μm. The eggs are indistinguishable morphologically and morphometrically from those of other equine GIT strongylids.
Hosts
Apart from the horse, the definitive host range includes donkeys, mules and zebra species.
Distribution
The large strongyles have a cosmopolitan distribution; Strongylus vulgaris is the most common species.
Transmission
The large strongyles have a direct life cycle. Sexually mature females are oviparous. Eggs are shed with the faeces into the environment. Depending on the prevailing climatic conditions, there is a typical pre-parasitic development from egg to free-living L1, L2, and then finally the sheathed infective L3 –this all in as little as 3 days.
Ingestion of sheathed free-living larvae is the only mode of infection for definitive hosts. Following uptake, the larvae of the large strongyles typically undergo extensive intra-abdominal migration –which differs markedly in the three species.
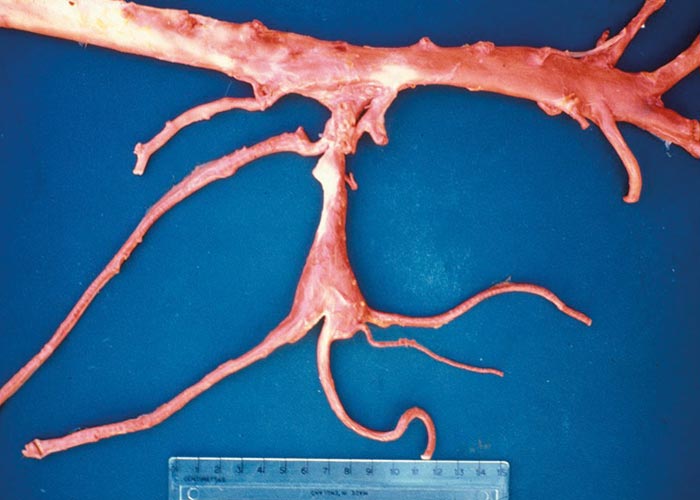
Strongylus vulgaris: Infective larvae exsheath in the stomach and undergo a histotropic phase in the mucosa of the caecum and colon –where they develop to L4. The L4 stage then enters arterioles of the gut and migrates on the endothelium to the cranial mesenteric artery and its main branches, where development to pre-adult stages takes place. Pre-adult stages are carried back via the arterial bloodstream to the wall of the caecum and colon –where they form nodules which eventually rupture and release the worms into the lumen of the caecum and colon. The pre-patent period is 6 to 7 months and the patent period is up to 18 months.
Strongylus edentatus: Infective larvae pass into veins of the intestinal mucosa and migrate via the portal system to the liver, where they migrate into the parenchyma –becoming encapsulated in granulomas in which they develop to L4. L4-stages emerge from granulomas and migrate to the liver capsule, which they cannot penetrate. Eventually, they enter the hepatic ligaments and migrate retroperitoneally of the abdominal wall –where they are found in large numbers in the flanks. Aberrant migration of larvae is, however, common. Under the peritoneum, L4 develop to pre-adult stages. Probably via the hepatorenal ligament, pre-adult stages migrate back to the colon, where they eventually penetrate the lumen. Final maturation takes place in the caecum and colon. The pre-patent period is 10 to 11 months and the patent period up to 18 months.
Strongylus equinus: Infective larvae exsheath in the stomach and undergo a histotropic phase in the mucosa of the caecum and colon, where they develop to L4. The L4-stage migrates via the peritoneal cavity into the liver parenchyma –where extensive migration continues. Eventually, L4 enter the pancreas, and partially also the peritoneal cavity. In the pancreas, larvae develop to pre-adult stages which enter the lumen of the gut via the connective-tissue adhesion of the pancreas and head of the caecum. Final maturation takes place in the caecum and colon. Aberrant migration of L4 is common. The pre-patent period is 8-9 months and the patent period is up to 18 months.
Large strongyles occur concurrently with cyathostomins. However, in contrast to the cyathostomins, large strongyle worm burdens are small, and do not exceed a few hundred worms.
Ssocio-economic importance
Together with the small strongyles, the large strongyles were once regarded as the most common and most important helminth parasites of horses. Both worm groups are major causes of colic. However, with the introduction and wide use of macrocyclic lactones the importance of large strongyles has largely diminished.
Pathogenesis and pathology
The pathogenesis of the large strongyles is mainly associated with the intra-abdominal migration of the larval stages. Strongylus vulgaris is regarded as the most pathogenic nematode of horses.
Strongylus vulgaris : Larvae migrating in the cranial mesenteric artery cause verminous arteritis with subsequent thrombosis and embolism ( Figure 53 ). Depending on the extent of the vascular lesions, the blood flow is impaired or can become interrupted which can result in infarction of parts of the small and large intestine. Aberrant larval migration in other arteries –such as the aorta, coronary arteries or external iliac artery –is well documented and involves similar vascular changes and lesions in their areas of blood supply.
Strongylus edentatus: Migrating larvae in the liver cause extensive haemorrhage, and larvae accumulating in the flank region cause oedema. Aberrant larval migration results in the formation of granulomatous lesions, in, for example, the diaphragm, lungs and omentum.
Strongylus equinus: The pathogenic effects are similar to those of S. edentatus. The lumenal pre-adult and adult stages are mainly histophagic and to a lesser extent haematophagic, and are regarded as insignificantly pathogenic.
Clinical signs
The appearance and severity of clinical signs are directly related to the number and period over which larvae were acquired –as well as the immune status of the host.
Vulgaris strongylosis : In this type of strongylosis, two main syndromes –acute and chronic verminous arteritis –are encountered. Acute verminous arteritis follows strong primary or superinfections of foals and yearlings, which present with anorexia, colic (thrombo-embolic colic), cyanosis, constipation, fever, dilated bowel, sweating, tachycardia and weight loss. In chronic verminous arteritis, intermittent (sporadic) colic attacks, inappetence, weight loss and a rough hair coat are observed. Aberrant larval migration can clinically manifest in far less common conditions like ‘intermittent lameness ’.
Edentatus and equinus strongylosis: Clinical signs seen in the acute form are colic and diarrhoea. The chronic form presents with similar clinical signs as listed for chronic verminous arteritis caused by S. vulgaris .
Diagnosis
The diagnosis in live animals is problematic, since clinical signs are observed during the pre-patent period.
A presumptive clinical diagnosis can be based on : Careful evaluation of the case history –considering epidemiological factors and the clinical picture.
Discolouration of the peritoneal fluid (dark yellow), serum biochemistry (hypoproteinaemia, hypoalbuminaemia, increased alkaline phosphatase and γ-glutamyl transpeptidase), and haematological findings (leukocytosis, eosinophilia, and anaemia) are indicative.
Parasitological diagnosis : The parasitological diagnosis in live animals is based on the identification of infective larvae harvested from faecal cultures. Identification of larvae in faecal cultures should be entrusted to an experienced diagnostician. Mere demonstration of strongylid-type eggs in faecal samples is inappropriate to derive a definitive diagnosis.
Differential diagnosis
Cyathostominosis is the most important GIT helminth infection of horses to consider when finding strongylid-type eggs in faecal samples.
Other more important helminths to consider in the syndrome of colic are the cyathostomins, Parascaris equorum (particularly in foals and yearlings), Anoplocephala perfoliata , and Gastrodiscus aegyptiacus .
Control
Treatment: There are several anthelmintics belonging to various chemical groups which are indicated for the control of strongylosis in horses.
The drugs differ in their duration of activity –measured by the egg reappearance period (ERP) and the spectrum of developmental stages that they control (adults ‘A ’, migratory larval stages ‘L ’):
- Tetrahydropyrimidines: Pyrantel (A)
- Benzimidazoles: Fenbendazole (L [7.5 mg/kg for 5 consecutive days] + A)
- Macrocyclic lactones: Abamectin, ivermectin, moxidectin (L + A)
Lesions caused by intra-abdominal migrating stages are mostly reversible –following treatment with larvicidal anthelmintics.
Prevention: See under cyathostomins.
Cyathostomins or small strongyles (Cyathostominosis)
General
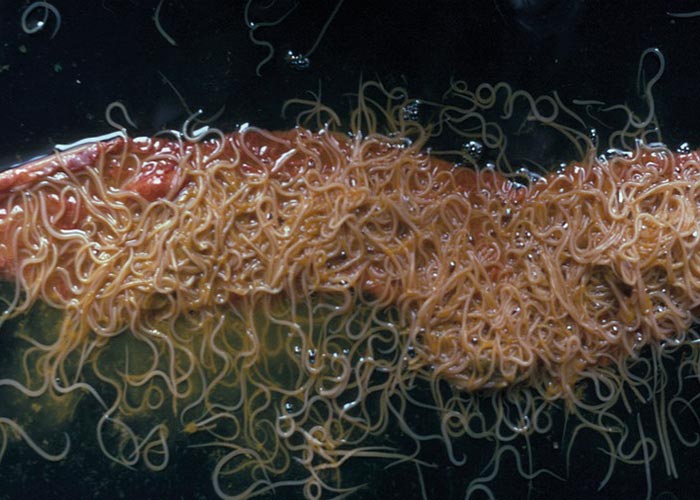
Cyathostominosis of horses is caused by ‘small strongyles ’. In contrast to the ‘large strongyles ’, the ‘small strongyles ’of horses are non-migratory (‘non-migratory strongyles ’) and comprise a group of about 60 different nematode species –the most important of which belong to the subfamily Cyathostominae. Other subfamilies of less importance are Strongylinae and Gyalocephalinae. In the clinical literature the ‘small strongyles ’are often collectively referred to as ‘cyathostomins ’or ‘cyathostomes ’, although strictly speaking the term only comprises members of the subfamily Cyathostominae. The group name ‘cyathostomin ’is derived from the Latin word ‘cyathus ’ (beaker) and the Greek word ‘stoma ’ (mouth), which is highly descriptive as it refers to the well developed, small to medium-sized, beakershaped buccal capsule of the worms. Teeth are present in some species. Typically, the buccal capsule is surrounded by leaf crowns. Adult worms are medium- sized, 0.5 to 3.0 cm long and 1.5 mm wide. Fresh specimens vary in colour from yellowish-white to dark red and are therefore sometimes also referred to as ‘blood worms ’or ‘red worms ’. The posterior end of the males has a strongly developed bursa, with bursal rays and spicules. The predilection sites in the definitive host are the caecum and colon ( Figure 54 ).
The eggs of the cyathostomins are of the strongylid-type. Strongylid-type eggs are oval and medium-sized (60- 125 x 30-60 μm). They have a thin and smooth shell and contain 4 to 8 blastomeres when laid. The eggs are indistinguishable morphologically and morphometrically from those of other equine GIT strongylids.
Hosts
Apart from horses, the definitive host range includes donkeys, mules and zebras.
Distribution
The cyathostomins have a cosmopolitan distribution.
Transmission
All cyathostomins have a direct life cycle. Females are oviparous. Eggs are shed with the faeces into the environment. Depending on prevailing climatic conditions, there is a typical pre-parasitic development from egg to free-living L1, L2, and finally an infective, sheathed L3 in as little as 3 days.
Ingestion of sheathed, free-living L3 is the only mode of infection for definitive hosts. Infective larvae exsheath in the stomach and undergo a histotropic phase in the mucosa of the caecum and colon –where L3 develop into L4. The usual duration of the histotropic phase is 1 to 2 months. Under certain conditions (e.g. unfavourable climate, established adult population in the caecum and colon) L3 can become hypobiotic, which results in a prolonged histotropic phase of up to 3 years.
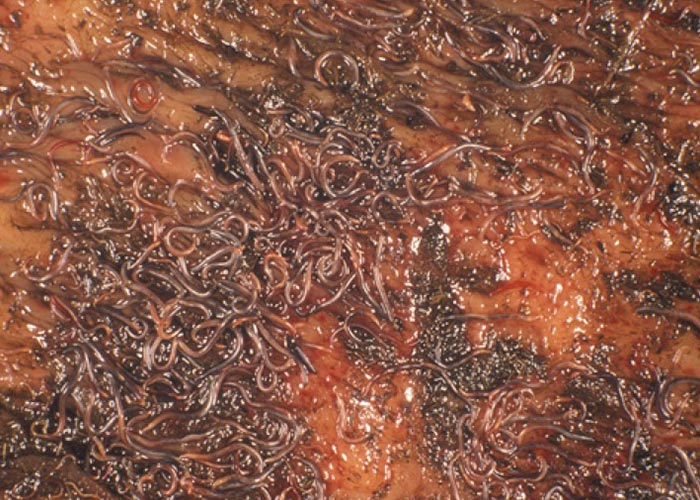
The final moult and maturation take place in the caecal and colonic lumen. The usual pre-patent period varies from 5.5 to 14 weeks. The patent period is for up to 2.5 years. Cyathostomins always occur as a mixed population of several species. Adult worm burdens of 500 000 are common in horses.
Socio-economic importance
The cyathostomins are the commonest and clinically most significant helminth parasites of horses, as they are regarded as a major contributing factor of diarrhoea, colic, sudden weight loss and debilitation. Colic is the most important cause of morbidity and mortality in horses. With the introduction of the macrocyclic lactones, the significance of the previously equally important large strongyles has diminished considerably compared to the cyathostomins. The emergence of anthelmintic resistance in the cyathostomins is another factor leading to their increase in importance. Managing of cyathostomin infections requires considerable input in terms of expert advice, diagnostics and anthelmintics –which is often very expensive.
Pathogenesis and pathology
The pathogenesis is associated with the two developmental phases of the parasite in the definitive host.
Histotropic phase: The larval stages are the most pathogenic. Depending on the infection rate, immunological status of the host, and age, exsheathed L3 entering the mucosa cause a catarrhal, haemorrhagic or fibrinous inflammation of the colon and caecum –known as typhlocolitis. Eventually, L3 larvae provoke a granulomatous inflammatory reaction in the mucosa, resulting in the formation of nodular lesions (0.5 to 5.0 mm in diameter) that significantly affect nutritional metabolism. There can be as many as 60 nodules per cm2. Subsequent synchronised mass excystation and migration of L4 into the lumen leads to mucosal ulceration and haemorrhages. This is usually seen in late winter or early spring. The damaged mucosa allows endotoxins originating from the intestinal contents to enter, which then cause endotoxaemia. The seepage of protein and fluids into the intestine eventually results in hypoalbuminaemia.
Lumenal phase: Adult stages are mainly histophagic and to a lesser extent haematophagic, and feed largely on mucosa. Extensive mucosal damage can be the consequence of high worm burdens, which manifests as diarrhoea, debilitation and weight-loss.
Clinical signs
Several clinical forms of cyathostominosis have been described in the literature –of which larval cyathostominosis is the most important.
Larval cyathostominosis: An acute clinical syndrome, which is mostly seen in horses younger than 6 years. A history that horses were dewormed during the previous 2 weeks is typical and indicative of the infection. Onset is highly seasonal –mostly during winter and spring –and is the result of a synchronised reactivation, excystation and emergence of previously hypobiotic stages from the mucosa. There is a sudden onset of diarrhoea, which may be intermittent or persistent. This is accompanied with rapid weight-loss and deterioration of body condition. In severe cases, muscle wasting is apparent. Animals are very weak and lethargic, but their appetite and water intake are unaffected. On the ventral abdomen and limbs a marked subcutaneous oedema develops. There is fever and intermittent attacks of colic. The mortality rate is up to 60%.
Diagnosis
To make a diagnosis in live animals is problematic as clinical signs are mostly observed during the pre-patent period.
A presumptive clinical diagnosis is based on:
- Careful evaluation of the case history considering epidemiological factors (season) and the clinical picture (chronic/acute diarrhoea, weight-loss, oedema, lethargy, fever, colic) –can provide evidence to suspect clinical cyathostominosis.
- Serum biochemistry (hypoalbuminaemia, hyper-β-globulinaemia, increased alkaline phosphatase) and haematological findings (neutrophilia, anaemia with or without eosinophilia and/ or lymphocytosis) are highly indicative that cyathostomins are involved.
- Large numbers of dark red L4 and preadult stages in the faeces are sometimes noticed.
Parasitological diagnosis: The parasitological diagnosis in live animals is based on the identification of infective larvae harvested from faecal cultures. Identification of larvae in faecal cultures should be entrusted to an experienced diagnostician. Mere demonstration of strongylid-type eggs in faecal samples is inappropriate to derive to a definitive diagnosis.
Differential diagnosis
Other causes of typhlocolitis that should be considered are fungal and bacterial infections and drug-related side effects caused by, for example, non-steroidal anti-inflammatory drugs, antibiotics and sulphonamides.
Other important helminth infections to consider in the syndrome of colic include Parascaris equorum particularly in foals and yearlings, Anoplocephala perfoliata, Gastrodiscus aegyptiacus and large strongyles.
Control
Treatment: There are several anthelmintics belonging to various chemical groups which are used for the control of cyathostomins in horses. The drugs differ in their duration of activity –measured by the egg reappearance period (ERP) and the spectrum of developmental stages they control (adults ‘A ’, developing larval stages ‘L ’, hypobiotic larval stages ‘H ’):
- Piperazines:
Piperazine salts (A) - Tetrahydropyrimidines:
Pyrantel (A) - Benzimidazoles:
Fenbendazole (L + A [5 mg/kg], H [10 mg/kg for 5 consecutive days]) - Macrocyclic lactones:
Abamectin, ivermectin (L + A), moxidectin (L + A + H)
The largely anthelmintic-based control programmes of the last few decades have strongly selected for resistant cyathostomin strains which affect all anthelmintic groups used in horses. The faecal egg count reduction test (FECRT) is the most appropriate and practical means to detect anthelmintic resistance.
Prevention: The aim is to manage the level of cyathostomins in order to prevent the syndrome of larval cyathostominosis. Control programmes need to be adapted to individual farms/studs. In the past, control programmes relied predominantly on the repeated use of anthelmintics. However, anthelmintic resistance has developed and is spreading fast in the cyathostomins, meaning that a situation in which no anthelmintics remain to treat infections is approaching. Therefore, it is imperative to take cognisance of the dilemma and act in a proactive manner to slow down the development of anthelmintic resistance.
Control programmes should aim to reduce infective larval stages on pastures and to reduce the number of anthelmintic treatments as far as possible (see selective dosing ) .
Current control measures comprise environmental, non-chemical and chemical procedures which can be summarised as follows:
Environmental/non-chemical control procedures:
- Pasture hygiene by implementing regular collection and proper disposal of faeces (‘clean pasture approach ’) can reduce the availability of infective L3 on the ground.
- Pasture management –taking into consideration ‘stocking density ’, ‘alternate grazing ’(grazing different host species on a rotational basis) and ‘rotational grazing ’where applicable.
Anthelmintic dosing practices:
- Interval dosing (synchronised treatment of an entire group of animals or single animal at intervals): This dosing practice is particularly aimed at yearlings. The intervals are based on the egg reappearance periods after treatment (these are different for each anthelmintic group). A strong selective pressure is exerted on the parasite population.
- Strategic dosing (treatment of an entire group of animals or single animal at set times based on epidemiological information). This dosing practice usually consists of seasonal treatments before winter [May] and after winter [October].
- Selective dosing (treatment only of those animals with significant faecal egg counts; the cut-off point is 100 or 200 eggs per gram [epg] of faeces). This dosing practice requires regular and individual faecal egg counts. Selective dosing allows establishment of naturally acquired protective immunity, while minimising anthelmintic use. However, it is not recommended in yearlings because of inadequate acquired immunity.
- Continuous in-feed dosing consists of year-round daily treatment. Pyrantel was mostly used in this approach. Since there is a very high selective pressure exerted on the helminth population, this dosing practice should no longer be considered.
Strongyloides or thread worms (Strongyloidosis)
Strongyloides westeri
General
Strongyloidosis of horses is caused by Strongyloides westeri. Strongyloides species are also known as ‘threadworms ’. The genus name is derived from the Greek word ‘strongylos ’(= rounded) and ‘-eides/- oides ’ (= similar) –referring to the cylindrical shape of the worms. The parasitic worm population consists of parthenogenetic females only, which are small and slender, with a tapering anterior end. Females are 8 to 9 mm long and 80 to 95 μm wide. Strongyloides species have a cylindrical oesophagus that extends through the anterior third of the body. The posterior end is conical. Fresh adult specimens are colourless.
The predilection site in the definitive host is the small intestine –especially the duodenum and jejunum. The eggs of S. westeri are oval and thin-shelled. The surface of the egg is smooth. They measure 40 to 50 μm by 30 to 40 μm and are embryonated when laid.
Hosts
Apart from horses, the definitive host range includes donkeys and zebras.
Distribution
Strongyloides westeri has a cosmopolitan distribution.
Transmission
Strongyloides westeri has a direct life cycle. Parthenogenetic females are ovoviviparous –i.e. they produce embryonated eggs. Eggs are shed with the faeces into the environment. Depending on prevailing climatic conditions, there is a typical pre-parasitic development from embryonated egg to free-living L1, L2 and finally unsheathed infective L3 –in as little as 3 days. This scenario is part of what is known as the homogonic life cycle. Alternatively, a heterogonic life cycle is followed, in which L1 develop via L2, L3 and L4 to free-living male and female worms reproducing on the ground.
With sufficient moisture provided, infective larvae can survive for about 3 weeks on the ground.
There are various modes of infection for definitive hosts:
- Trans-mammary (lactogenic) infection following ingestion of reactivated arrested (somatic) larvae from the lactating mare, is the most important mode of infection of foals. The pre-patent period is only 8 to 10 days.
- Percutaneous infection and ingestion of free-living infective L3 is less important.
Infective larvae undergo lymphatic-tracheal migration in non-immune hosts –before developing into females in the small intestine. Infection induces immunity, with infective larvae becoming dormant (arrested, hypobiotic) in the udder and other parts of the body. Depending on the mode of infection, the pre-patent period varies from 3 to 14 days. The patent period is 2 to 9 months.
Socio-economic importance
Strongyloidosis of horses seldom requires veterinary intervention. However, Strongyloides westeri is the most significant parasitic aetiological factor of diarrhoea in foals. Humans can act as paratenic hosts for all animal Strongyloides species. Infection is percutaneous, and the developing clinical picture is that of cutaneous larva migrans. This, however, is more commonly caused by animal hookworm species.
Pathogenesis and pathology
Strongyloides species are mucosal dwellers. Depending on the immunological status and worm burden, infection causes villous atrophy, cellular infiltration, and destruction of the epithelium –which results in malabsorption, hypo- proteinaemia, hypo- albuminaemia and eventually subcutaneous oedema.
Clinical signs
Clinical strongyloidosis is only seen in very young foals –mostly 9 to 16 days after birth - harbouring heavy worm burdens. Intermittent watery diarrhoea (‘scouring ’), inappetence, exsiccosis (insufficient intake of water) and diarrhoea are common clinical signs. Death is possible, but is not characteristic for equine strongyloidosis.
Diagnosis
Presumptive clinical diagnosis: Severe diarrhoea of foals during the first 3 weeks post partum –as well as hypoproteinaemia, hypoalbuminaemia, anaemia, and sub cutaneous oedema –are highly indicative.
Parasitological diagnosis: The parasitological diagnosis is based on the demonstration of embryonated eggs in faecal samples, by means of flotation. Preparing faecal cultures with subsequent identification of L3 is more sensitive, and should be considered if faecal flotation is negative.
Differential diagnosis
Other causes of diarrhoea in nursing foals –to be considered include rotavirus, Cryptosporidium and Clostridium perfringens infections.
Control
Treatment: The benzimidazole fenbendazole (50 mg/kg) and the macrocyclic lactones –abamectin, ivermectin and moxidectin –are effective against the lumenal stages. Lactogenic infections can be reduced by treating mares on the day of foaling with a macrocyclic lactone.
Prevention: Hygiene is the most important consideration in the successful control of equine strongyloidosis. Boxes should be cleaned regularly and thoroughly. Disinfection of floors with 2% sodium hydroxide solution, once a week, is an inexpensive and highly effective choice. Daily removal of faeces from boxes and paddocks is another important measure to reduce the number of free-living infective larvae. Depending on availability and feasibility, alternate grazing is a worthwhile consideration.
Oxyuris or pin worms (oxyuriosis)
Oxyuris species
General
Oxyuriosis of horses is caused by Oxyuris equi. The genus name is derived from the Greek words ‘oxys ’ (= pointed) and ‘ura ’ (= tail), which –like the common name ‘pinworm ’–are descriptive terms referring to the long, pointed tail of the female worm. Quite unusual for the group of the oxyuroids, Oxyuris equi is a small to large, thick-bodied nematode. Male worms are only 9 to 12 mm long, and female worms are 40-150 mm long. Microscopically, the worms have a small buccal capsule which leads into a typical oxyuroid oesophagus. The posterior end of the male is truncate with a pair of caudal alae. There is a single, needleshaped spicule, which is 120 to 200 μm long. Females have a typical long, gradually tapering pointed tail –which is up to twice the length of the body. Fresh adult specimens of O. equi are whitish in colour ( Figure 56 ).
The typical predilection site of O. equi is the caecum and colon.
Eggs of O. equi are slightly asymmetrical, thick-shelled, and have an operculum at one pole. They measure 85 to 95 μm by 40 to 45 μm and contain a larva (L1 stage) when shed in the faeces.
Hosts
Apart from horses, the definitive host range includes donkeys, mules and zebra.
Distribution
Oxyuris equi has a cosmopolitan distribution.
Transmission
Oxyuris equi has a direct life cycle. Females only oviposit once. They partially migrate out of the anus to deposit the eggs in strings in the perianal area. In only 3 to 7 days, an infective L3 develops within the egg. Egg strings –on drying off –detach from the skin and fall to the ground as small flakes. Eggs are highly sensitive to desiccation and are viable for only a few days on the ground. However, in moist surroundings, eggs can remain viable for several weeks. Ingestion of eggs containing infective larvae is the only mode of infection. Infective larvae hatch in the small intestine and undergo a histotropic phase for 3 to 10 days in the mucosal crypts of the caecum and colon –during which time the third moult takes place. The emerging L4 stages migrate to the dorsal colon, where they are found attached to the mucosa. After 2 months the final moult takes place, and sexual maturity is reached after another 2 to 3 months. The pre-patent period is 4.5 to 5 months and the patent period of females is 6 months, after migrating back into the rectum. Male worms die soon after fertilisation has taken place.
Socio-economic importance
Oxyuris equi is a very common nematode parasite of horses, but is only mildly pathogenic. The species does not have any zoonotic implications. The apparent zoonotic potential of O. equi is a common misconception –not only among horse owners, but also with health professionals. Oxyuroids in general are highly host specific. Pinworm infection of humans, commonly seen in children, is caused by Enterobius vermicularis .
Pathogenesis and pathology
Pathogenesis is associated with the emerging L4-stages in the caecum and ventral colon feeding on mucosa and the mature female worms: large numbers of L4 stages in the caecum and ventral colon can cause inflammatory reactions and ulceration of the mucosa, which are clinically inapparent. Females partially migrating out of the anus to deposit eggs cause intense pruritus in the perianal area.
Clinical signs
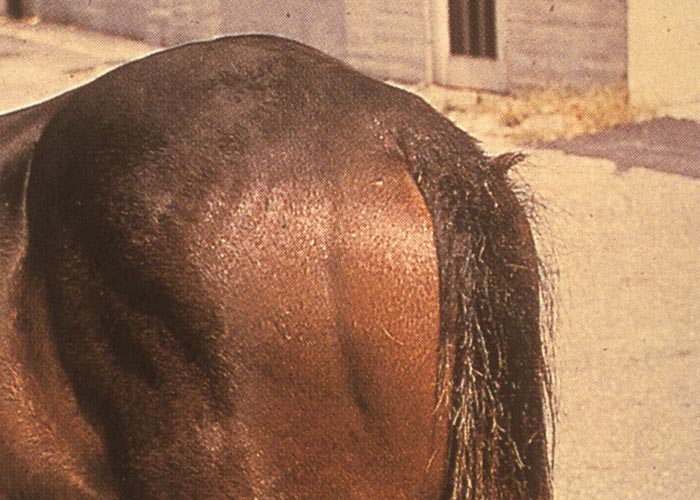
Oxyuriosis is typically seen in stabled horses. As a result of intense pruritus in the perianal area, horses are restless –which also impacts on feeding and subsequently body condition. Horses rub their tail-base against walls, fence posts, tree trunks and other objects, causing hairs to break off which eventually gives the tail an untidy or “rat tailed ”appearance in severe cases ( Figure 55 ).
Diagnosis in live animals
Presumptive clinical diagnosis: Clinical signs and the presence of whitish-greyish streaks or smudges in the perianal area, are indicative of infection.
Parasitological diagnosis: The parasitological diagnosis in live animals is based on the demonstration of eggs in the perianal area –by means of the adhesive tape-swab technique. Since eggs are deposited outside the gut, they are unlikely to be found in faecal samples. Female worms are sometimes voided with the faeces and are easily recognised by their size, thick-bodied appearance, and long pointed tail.
Differential diagnosis
Infections with the oxyuroid Probstmayria vivipara should be taken into consideration. However, the species is regarded as nonpathogenic and infected horses –even with high worm burdens –do not develop clinical signs. Diagnosis in live horses is difficult, since females are viviparous.
Control
Treatment: Several anthelmintics belonging to various chemical groups are indicated for the treatment of oxyuriosis in horses:
- Piperazines: Piperazine salts
- Tetrahydropyrimidines: Pyrantel
- Benzimidazoles: Fenbendazole
- Macrocyclic lactones: Abamectin, ivermectin, moxidectin
Control: Infections can be prevented by regular check-ups of the perianal area and by wiping off egg masses with a wet cloth. Other important measures that will assist in control are frequent changing of bedding and the provision of proper cribs and troughs installed off the ground.
Summer sores (Habronematidosis)
Habronema species
Draschia species
General
Habronematidosis of horses is caused by Habronema muscae, Habronema majus and Draschia megastoma . The genera Habronema and Draschia belong to the family Habronematidae –a name derived from the Greek words ‘habros ’ (= delicate) and ‘nema ’ (= thread). Habronematids are slender and 7 to 25 mm long. The species have a mediumsized, thick-walled buccal capsule with large pseudolabia. The posterior end of the male is spirally coiled and has caudal alae. The spicules are unequal. The female posterior end is conical. The vulva is located near the middle of the body.
Habronema muscae male worms are 8 to 14 mm long and female worms 12-22 mm long. The worms have a cylindrical buccal capsule, which is unarmed. Spicules are unequal and measure 2.5 mm (left) and 0.5 mm (right) in length, respectively.
Habronema majus male worms are 9 to 22 mm long and female worms are 15 to 35 mm long. The worms have a cylindrical buccal capsule, which is armed with two teeth. Spicules are unequal and measure 750 to 880 μm (left) and 350 to 380 μm (right) in length, respectively.
Draschia megastoma male worms are 7 to 10 mm long and female worms 10 to 13 mm long. The worms have a funnelshaped buccal capsule, which is unarmed. Spicules are unequal and measure 460 μm (left) and 240 to 280 μm (right) in length, respectively.
Fresh adult specimens of H. muscae are orange in colour, whereas H. majus and D. megastoma are whitish in colour.
The typical predilection site of the Habronema species is the mucosa of the fundus region of the stomach –where they are covered by a thick mucus layer.
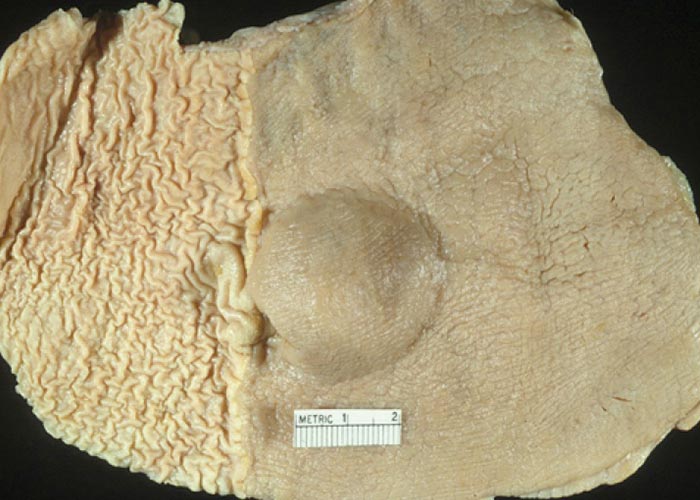
Draschia megastoma is found in colonies embedded in granulomatous, chickenegg- sized nodules in the fundus region of the stomach. Nodules protrude into the lumen of the stomach.
Eggs of the Habronema species are cigar- shaped and thin-shelled. They measure 40 to 50 μm by 10 to 16 μm, and are embryonated when laid.
Female worms of D. megastoma are viviparous. The L1 stages are 330 to 350 μm long and 8 μm wide.
Hosts
Apart from horses, the definitive host range includes donkeys, mules and zebra.
Distribution
The habronematids have a cosmopolitan distribution.
Transmission
The habronematids have an indirect life cycle. Females of H. muscae and H. majus are ovoviviparous, whereas females of D. megastoma are viviparous. Embryonated eggs and larvae are shed with the faeces into the environment.
Non-biting and biting muscid flies act as intermediate hosts (vectors). Musca domestica and other Musca species are known vectors for H. muscae, H. majus and D. megastoma. Stomoxys calcitrans has been identified as a vector for H. majus only. Embryonated eggs and larvae are ingested by the dung-inhabiting larvae of the muscids. The development to infective L3 stages is synchronised with the development of the vectors, and is completed when the flies are emerging from the pupae. The incubation period in muscid vectors is largely temperature dependent and can last as little as 7 days.
Horses become infected by ingestion of infective larvae breaking out of the fly ’s mouthparts while feeding on the lips and nostrils, or by ingestion of dead infected flies in water and feed. There is no migration involved and larvae will develop to adults in the stomach –leading to ‘gastric habronematidosis ’. The pre-patent period is 2 months.
Flies also commonly settle on skin wounds and the conjunctivae of the eyes, in which case emerging larvae invade tissues, but do not complete the life cycle and succumb after a few weeks. Migration of larvae will result in granulomatous lesions referred to as summer sores (‘cutaneous/ophthalmic habronematidosis ’).
Socio-economic importance
Habronematids are common and important nematode parasites of horses –responsible for granulomatous skin lesions.
Pathogenesis and pathology
Gastric habronematidosis ( Figure 58 ): Habronema species can cause a chronic catarrhal gastritis with excessive mucous production. Draschia megastoma causes a typical granulomatous reaction in the fundic region of the stomach –resulting in the formation of chicken-egg-sized nodular lesions which protrude into the gastric lumen. Gastric perforation with subsequent peritonitis has been reported, but is extremely rare.
Cutaneous habronematidosis (‘summer sores ’) ( Figure 57 ): Infective larvae entering skin wounds cause the formation of pruritic, reddish-brown, granulomatous lesions that protrude in a cauliflower-like fashion above the surface of the skin.
Clinical signs
Despite a high prevalence of gastric habronematidosis, it very rarely manifests clinically as chronic gastritis. However, cutaneous habronematidosis is a common clinical observation in horses worldwide, and is known as ‘summer sores ’. Common parts of the body affected are those subjected to superficial injury, such as the head region (head, eyelids, conjunctivae), withers, inner parts of the lower limbs, and the coronet, prepuce and penis. Secondary bacterial infections can result in further aggravation. Lesions are typically seasonal and coincide with the fly season –after which larvae slowly die off in the wounds, and self healing with formation of scar tissue takes place.
Diagnosis
The diagnosis in live animals is problematic and is almost exclusively based on the clinical signs. Histopathological examination of biopsy specimens may help differentiate it from other proliferative lesions, including neoplasms such as squamous cell carcinoma that may occur in similar areas of the skin.
Presumptive diagnosis: The clinical diag nosis is based on the seasonal appearance of proliferative and pruritic granulomatous skin lesions
Parasitological diagnosis: The parasitological diagnosis of patent gastric infections is impractical as embryonated eggs or larvae are rarely detected in faecal examinations. Since gastric habronematidosis has very little clinical significance, diagnosis in live animals is irrelevant.
Differential diagnosis
The filarial parasites Parafilaria multipapillosa and Onchocerca reticulata should be considered since both are known to cause open skin lesions, which resemble those observed in cutaneous habronematidosis.
Control
Treatment: Macrocyclic lactones are effective for the treatment of cutaneous and gastric habronematidosis.
Control: Fly control is beneficial in the control of habronematidosis. Skin wounds –especially during the fly season –should be adequately attended to in order to prevent infestation by muscid flies.
Ascarid worms (Parascariosis)
Parascaris species
General
Parascariosis of horses is caused by the nematode Parascaris equorum. The genus name is derived from the Greek word ‘ascaris ’ (= worm of the entrails). Being a typical ascarid, adult worms are easily recognisable because of their large size. Male worms are 15 to 28 cm long and female worms 16 to 50 cm long. Both are about 3 to 8 mm wide.
Microscopically, the species has a small buccal capsule, surrounded by 3 large, heart-shaped lips. The posterior end of the male is conical and has small caudal alae. The spicules are equal and measure 2.0 to 2.5 mm in length. The posterior end of the female terminates in a short conical process. The vulva is located at the posterior end of the anterior quarter of the body.
As for all ascarids the predilection site in the definitive host is the small intestine.
Eggs of P. equorum are subspherical and medium-sized (90 to 100 μm in diameter). They have a thick, pitted, brown shell and contain a single cell when laid.
Hosts
Apart form the horse, the definitive host range includes donkeys, mules and zebra.
Distribution
Parascaris equorum has a cosmopolitan distribution.
Transmission
Parascaris equorum has a direct life cycle. As with all ascarids, females have a characteristically high biotic potential. Eggs are shed with the faeces into the environment. They are highly resistant and remain viable for up to 5 years.
Depending on temperature, an infective L3 develops within the egg in as little as 10 to 15 days.
Ingestion of larvated eggs is the only mode of infection for definitive hosts. Larvae hatch in the small intestine and subsequently burrow into the intestinal wall, where they enter small blood vessels. Following a hepatic-tracheal migration, L4 stages settle in the small intestine, where the final moult takes place ( Figure 59 ). The pre-patent period is 72 to 115 days and the patent period is up to 2 years.
Socio-economic importance
As a significant cause of diarrhoea and colic, Parascaris equorum is the most important endoparasite of foals. The expense of registered products for deworming and veterinary treatment in surgical cases is considerable, and is beyond the reach of resource-poor animal owners.
Pathogenesis and pathology
The pathogenesis is associated with the two developmental phases of the parasite.
Larval migratory phase: Migration of larvae through the liver and lungs causes microlesions and inflammatory reactions, which lead to alveolitis, bronchiolitis and bronchitis.
Lumenal phase: After reaching the small intestine, the worms cause a chronic catarrhal enteritis and hypoalbuminaemia. Because of the large size of the developing worms, intestinal impaction, intussusception and perforation with subsequent peritonitis, are common if high burdens prevail. Migration of worms to aberrant sites such as the bile duct may occur.
Clinical signs
Clinical parascariosis is almost exclusively seen in foals and yearlings. From about 6 months of age, protective immune mechanisms become effective –with the result that occasional infections are mostly clinically inapparent in older horses.
The clinical signs are also associated with the two developmental phases of the parasite in the definitive host.
Larval migratory phase: During this phase coughing and nasal discharge are sometimes noticed.
Lumenal phase: Clinical signs observed are diarrhoea and colic attacks, together with poor growth, weight loss, sometimes a pot-bellied appearance, changing appetite, and a dull hair coat.
Diagnosis
Presumptive clinical diagnosis: Clinical signs in foals and yearlings –as listed above –give reason to suspect Parascaris infection. Respiratory clinical signs are the earliest seen in heavily infected foals during the pre-patent period, in which an aetiological diagnosis cannot be made. However, a marked blood eosinophilia is indicative.
Parasitological diagnosis: The parasitological diagnosis in live animals is based on the demonstration of eggs in faecal samples.
Differential diagnosis
Other causes of diarrhoea that should be considered are improper nutrition, common side effects of drugs (non-steroidal anti-inflammatory drugs, antibiotics, sulphonamides), fungal infections, bacterial infections, and other helminth infections (cyathostominosis, strongyloidosis, anoplocephalosis). Regarding respiratory clinical signs, microbial causes and infections with the horse lungworm Dictyocaulus arnfieldi should be taken into account.
Control
Treatment: There are several anthelmintics belonging to various chemical groups which are used for the control of parascariosis in horses. The drugs differ in their duration of activity, measured by the egg reappearance period (ERP):
- Piperazines: Various piperazine salts
- Tetrahydropyrimidines: Pyrantel
- Benzimidazoles: Fenbendazole
- Macrocyclic lactones: Abamectin, ivermectin, moxidectin
Pyrantel-resistant isolates of P. equorum have only been reported from North America. The emergence of macrocyclic lactone-resistant isolates of P. equorum , however, is of great concern –as macrocyclic lactones are widely relied on in roundworm control for foals and yearlings.
Foals become infected soon after birth and should first be treated at 9 to 10 weeks of age, with subsequent treatments at intervals of 2 months until they become yearlings. The efficacy of the anthelmintic should be monitored annually. Other important considerations in the control are the regular removal of faeces.
Trichostrongylus axei
(Trichostrongylosis)
General
Trichostrongylosis of horses is caused by the nematode Trichostrongylus axei . The genus name is derived from the Greek words ‘trichos ’ (= hair) and ‘strongylos ’ (= rounded). Adult worms are small, slender, and have a tapering anterior end. Male worms are 2.3 to 6.0 mm long and 50 to 70 μm wide. Female worms are 3.0 to 2-8 mm long and 55 to 75 μm wide. The species has no buccal capsule. The posterior end of the male has a strongly developed bursa. Spicules are dissimilar in size and shape; they measure 96 to 128 μm (left) and 74 to 104 μm (right) in length, respectively. The posterior end of the female is cone-shaped. The vulva is located in the posterior sixth of the body. Fresh adult specimens of T. axei are pale reddish-brown.
The predilection site in horses is the stomach –particularly the glandular part.
The eggs of T. axei are of the strongylidtype. Strongylid-type eggs are oval and thin-shelled, and have a smooth surface. They contain 4 to 8 blastomeres when laid and measure 79 to 92 by 31 to 41 μm. The eggs are morphologically and morphometrically indistinguishable from those of other equine GIT strongylids.
Hosts
Apart from horses, the definitive host range includes donkeys, mules, sheep, goats, cattle, deer, antelope, warthogs –and occasionally pigs and humans.
Distribution
Trichostrongylus axei has a cosmopolitan distribution.
Transmission
Trichostrongylus axei has a direct life cycle. Sexually mature females are oviparous. Eggs are voided with the faeces into the environment. Depending on prevailing climatic conditions, there is a typical preparasitic development from egg to freeliving L1, L2 and finally sheathed infective L3 in as little as 4 to 6 days.
Ingestion of sheathed free-living L3 is the only mode of infection for definitive hosts. Infective larvae exsheath in the stomach and enter the mucosa and glandular pits –where they develop after 2 moults into adults. The pre-patent period is 3 weeks, but can be much longer under unfavourable environmental conditions.
Socio-economic importance
Clinical trichostrongylosis can only develop where communal grazing or alternate grazing with ruminants is practised. Diagnosis in live animals requires specialist diagnostic facilities, which are expensive and out of reach for resource-poor animal owners. Furthermore, the choice of effective anthelmintics is limited.
Pathogenesis and pathology
In heavy infections, invasion of the gastric mucosa results in a chronic hyperplastic gastritis –which becomes macroscopically visible as circumscribed proliferative areas in the glandular part of the stomach. At a later stage these areas become eroded and are devoid of epithelium.
Clinical signs
Clinical signs observed in clinical trichostrongylosis are varied and range from a changing appetite, progressive weight-loss, through to general loss of condition. There is no diarrhoea.
Diagnosis
The parasitological diagnosis in live animals is based on the identification of infective larvae harvested from faecal cultures. Identification of larvae in faecal cultures should be entrusted to an experienced diagnostician. However, in most instances, the proof of T. axei infection is clinically insignificant. Mere demonstration of strongylid-type eggs in faecal samples cannot be used to confirm the diagnosis.
Differential diagnosis
Particularly cyathostominosis, and to a lesser extent strongylosis, are more important GIT helminth infections of horses to consider when finding strongylid-type eggs in faecal samples.
Control
Treatment: The macrocyclic lactones abamectin, ivermectin and moxidectin are effective for the treatment of trichostrongylosis in horses.
Prevention: Although advocated as a tool to manage helminths in the environment, communal grazing and alternate grazing with ruminants should be avoided.
Onchocerca species (Onchocercosis)
General
Onchocercosis of horses is caused by Onchocerca cervicalis and Onchocerca reticulata. The genus name is derived from the Greek words ‘onkos ’ (= tumour, lump) and ‘kerkos ’ (= tail) –which refer to the formation of lumps in the connective tissues, which are caused by the presence of the adult worms. Onchocerca species are medium to large-sized filiform nematodes. The cuticle of the body is often transversely striated. They have an atrophied stoma.
The posterior end of the male is spirally coiled. The spicules are dissimilar in size and shape. The posterior end of the female is rounded. The vulva is located in the anterior or posterior oesophageal region of the body.
O. cervicalis typically occurs in the nuchal ligament and surrounding sub-dermal and inter-muscular connective tissue, while O. reticulata occurs in the flexor tendon and suspensory ligament of the fetlocks.
Female worms are viviparous and produce unsheathed L1 stages –known as microfilariae.
See Table 7 for the comparative morphological details of O. cervicalis and O. reticulata.
Hosts
Apart from horses, the definitive host range of the two species includes mules and donkeys.
Distribution
Both species have a cosmopolitan distribution.
Transmission
Onchocerca species follow an indirect life cycle. Microfilariae produced by females appear in the lymph spaces of the skin.
Biting midges of the genus Culicoides act as intermediate hosts (vectors) for O. cervicalis and O. reticulata . Female biting midges become infected by obtaining blood meals from microfilaraemic horses. In the vector, microfilariae develop to infective L3 stages (metacyclic larvae), which eventually assemble in the proboscis. The incubation period in vectors is largely temperature dependent and may take as little as 14 to 15 days. Definitive hosts become infected during blood feeding of vectors, during which metacyclic larvae actively leave the labella of the proboscis onto the skin, and subsequently enter the puncture wound on their own. The pre-patent period is up to 16 months for O. cervicalis , but is unknown for O. reticulata. Onchocerca species are long-lived and microfilariae in the skin remain viable and infective for vectors, for up to 30 months.
Socio-economic importance
Onchocercosis in horses appears to be of minor clinical importance.
Pathogenesis and pathology
Macrofilariae (adult and pre-adult stages) and microfilariae can cause lesions in horses and other equids:
Effects on the ligamentum nuchae (‘fistulous withers ’): Large nodules (1 to 4 cm in diameter) caused by a verminous granulomatous inflammatory reaction, develop in the nuchal ligament. The inflammatory exudate contains fragments of degenerated worms embedded in caseous necrotic material, that eventually becomes calcified.
Effects on the skin (‘cutaneous onchocercosis ’): Microfilariae of O. cervicalis migrate into the epidermis and dermis of the ventral abdomen along the linea alba, head, neck and ventral thorax. These locations coincide with the preferential feeding sites of the vectors. Microfilariae can elicit a hypersensitivity reaction in these skin parts.
Effects on the eye (‘periodic ophthalmia ’): As in human onchocerciasis, microfilariae of O. cervicalis can migrate to particularly the cornea and sclera of the eye. Vascularisation of the cornea is a common feature, which is interpreted as a reaction to dead microfilariae.
Effects on the distal parts of fore and hind limbs (parasitic suspensory desmitis, tendonitis, sesamoiditis, navicular disease): Adults of O. reticulata cause nodular lesions mainly in the suspensory ligament and flexor tendon of the fore and hind limbs. Depending on their location, nodules can exert pressure on nerves –resulting in lameness. Tumour-like granulomatous lesions are sometimes noticed on overlying skin parts.
Clinical signs
Fistulous withers: Calcified nodules in the nuchal ligament can cause considerable irritation in horses used extensively for riding or as pack animals. Localised oedema, seromas, followed by perforation of fistulous channels originating from a lesion in the nuchal ligament can be observed.
Cutaneous onchocercosis: Hypersensitivity reaction to the presence of microfilariae of O. cervicalis can present as pruritic dermatitis –with erythema, alopecia, depigmentation, a papular rash, excoriation and the formation of crusts.
Periodic ophthalmia: Corneal vascularisation is commonly observed in periodic ophthalmia. Severely swollen eyelids, hyperaemic and oedematous conjunctivae –as well as excessive lachrymation –are seen in acute attacks.
Parasitic suspensory desmitis, tendonitis, sesamoiditis, and navicular disease: There is often a history of chronic lameness which is unresponsive to treatment. Nodular lesions in the suspensory ligaments can be highly sensitive to pressure. Eventually, open, tumour-like granulomatous lesions might develop on the bruised overlying skin.
Diagnosis
Presumptive diagnosis: Fistulous lesions in the withers region, periodic ophthalmia, and pruritic dermatitis on the ventral abdomen, ventral thorax, head and neck, can be indicative of onchocercosis caused by O. cervicalis .
A history of chronic lameness unresponsive to treatment, nodular lesions in mainly the suspensory ligament and flexor tendon of the fore and hind limbs, along with open, tumour-like, granulomatous lesions on the bruised overlying skin, can be indicative of onchocercosis caused by O. reticulata .
Parasitological diagnosis: The parasitological diagnosis in live horses is based on the identification of unsheathed microfilariae found in skin biopsies obtained from the ventral abdomen, ventral thorax, head and neck. Skin samples –about 3 mm in diameter –are teased and incubated in saline at 37 °C for 12 hours. Subsequently, the suspension is filtered through gauze and centrifuged at 200 g for 5 minutes. The sediment is examined microscopically for microfilariae. The microfilariae of the two Onchocerca species can easily be differentiated by their size ( see Table 7 ).
Differential diagnosis
Fistulous withers: Apart from bacterial infections, rare infections with the bovine species Onchocerca gutturosa have been reported to cause similar lesions.
Cutaneous onchocercosis: Dermatitis caused by the feeding of blood-sucking insects –such as ceratopogonids, biting muscids and simuliids –presents with the same clinical picture as is observed in cutaneous onchocercosis.
Periodic ophthalmia: Infections with Setaria equina have to be considered in cases of periodic ophthalmia.
Parasitic suspensory desmitis, tendonitis, sesamoiditis, and navicular disease: Apart from O. reticulata, Elaeophora boehmi has to be considered as a possible parasitological cause in cases of suspensory desmitis, tendonitis, sesamoiditis and navicular disease. However, currently, E. boehmi infections have only been reported in Europe and Asia. In concurrently occurring granulomatous lesions, habronematids (Habronema/Draschia species) as the cause of summer sores, should be considered.
Distinguishing between various causes of similar skin lesions is aided by histopathological examination of appropriate skin biopsies taken from the edges of the lesions.
Control
Treatment: There are no effective macrofilaricidal (adulticidal) drugs available in horses. The macrocyclic lactones ivermectin and moxidectin are effective microfilaricides.
Nodulectomy is indicated in some cases of O. reticulata infections affecting the distal parts of the limbs.
Control: Stabling of horses at night could be considered –since biting midges are mostly nocturnal feeders and exophagic.
Table 7 Onchocerca species of horses
| O. cervicalis | O. reticulate | |
|---|---|---|
Adult stages
| 500-700 mm x 260-570 μm 60-103 mm x 100-190 μm 320-380 μm (left) and 97-125 μm (right) | 450-750 mm x 275-400 μm 105-270 mm x 150-185 μm 248-315 μm (left) and 115-130 μm (right) |
Microfilariae
| Unsheathed 178-240 μm x 4-6 μm Skin | Unsheathed 310-395 μm x 6-7 μm Skin |
| Definitive hosts | Horse, donkey, mule | Horse, donkey, mule |
| Intermediate hosts (vectors) | Culicoides species | Culicoides species |
Setaria (setariosis)
General
Setariosis of horses is caused by Setaria equina . The genus name is derived from the Latin word ‘saeta ’ (= thick hair, bristle) –which refers to the appearance of the adult worms. Setaria species are large, filiform nematodes. The cuticle of the body is finely transversely striated. Microscopically, they have a small buccal capsule with an opening that is surrounded by a peri-buccal crown. Male worms are 51 to 66 mm long and 470 to 560 μm wide. The posterior end of the male is spirally coiled. Spicules are dissimilar in size and shape. They measure 280 to 290 μm (right) and 610 to 640 μm (left) in length. The smaller spicule terminates in a claw through which the larger spicule slips. Female worms are 72 to 190 mm long and 1.0 to 1.2 mm wide. The vulva is located in the oesophageal region of the body. The posterior end of the female is conical. Fresh specimens are whitish in colour.
Setaria equina typically occurs mainly in the peritoneal cavity. Sometimes the worms localise in the scrotum and the eye, and underneath the serosal surfaces of the liver, spleen, lungs, and heart.
Female worms are viviparous and produce sheathed L1 stages known as microfilariae. Microfilariae are 240 to 290 μm long and 5 to 8 μm wide.
Hosts
Apart from horses, the definitive host range of S. equina includes mules, donkeys, zebra and dromedaries.
Distribution
Setaria equina has a cosmopolitan distribution.
Transmission
Setaria species have an indirect life cycle. Microfilariae produced by females appear in the blood. The appearance of microfilariae in the peripheral blood of horses is nocturnal sub-periodic, with a maximum microfilaraemia observed at night between 22:00 and 23:00.
Mainly culicine mosquito species belonging to the genera Aedes and Culex act as intermediate hosts (vectors). Obtaining blood meals from microfilaraemic final hosts infects female mosquitoes. In the vector, microfilariae develop to infective L3 stages (metacyclic larvae) –which eventually assemble in the proboscis. The incubation period in vectors is largely temperature-dependent, and may take as little as 15 to 16 days.
Definitive hosts become infected during blood feeding of vectors –during which metacyclic larvae actively leave the labella of the proboscis, are deposited onto the skin, and subsequently enter the puncture wound on their own. The pre-patent period is up to 12 months.
Socio-economic importance
Equine setariosis is only of minor clinical importance and usually does not require veterinary intervention.
Pathogenesis and pathology
Microfilariae are regarded as non-pathogenic. Although rare, adult and pre-adult stages (macrofilariae) lodging in sites other than the peritoneal cavity can have pathogenic effects on the CNS of the host (‘verminous meningoencephalomyelitis ’). Setaria equina is regarded as the most common cause of equine verminous meningoencephalomyelitis in some tropical and subtropical parts of the world. Aberrant migration and definitive development of S. equina in the CNS causes acute focal encephalomyelomalacia –with degeneration of nerve tracts and an eosinophilic inflammatory reaction.
Eye: Infection of the anterior eye chamber and vitreous body can illicit severe inflammatory reactions. The reaction to degenerating microfilariae can cause blindness.
Scrotum: Aberrant migration into the scrotum can result in local inflammatory reactions accompanied by painful swelling of affected tissues.
Clinical signs
Although high prevalence has been reported in some endemic areas, setariosis is mostly clinically inapparent. Clinical signs can be severe in cases of aberrant migration and definitive development of worms in the CNS and eyes.
Verminous meningoencephalomyelitis: Most reports come from Sri Lanka, India and Burma, where setariosis is known as ‘kumri ’ (word derived from Hindustani, meaning ‘weak back ’). The clinical signs include muscular weakness, incoordination, ataxia, and paralysis.
Ocular setariosis: Ophthalmia with photophobia, excessive lachrymation, conjunctivitis, opacity of the cornea, hypopyon and iridocyclitis in the anterior chamber of the eye, and eventually blindness can be observed. Whitish worms rapidly moving in the anterior chamber of the eye are sometimes noticed.
Diagnosis
Presumptive diagnosis: Neurological clinical signs and ophthalmia can be indicative of setariosis. The definitive diagnosis of parasitic encephalomyelitis and its aetiology is only possible following a post-mortem examination.
Parasitological diagnosis: The parasitological diagnosis in live horses is based on the identification of microfilariae found in blood. Microfilariae can be detected microscopically in wet blood films, and stained thin and thick blood films. For increased sensitivity, concentration techniques like membrane filtration, the modified Knott ’s test, or microhaematocrit centrifugation should be used.
Whitish worms rapidly moving in the anterior chamber of the eye are sometimes visible. Specimens removed surgically can be identified on morphological criteria.
Differential diagnosis
Other microfilariae reported in the blood of horses are Elaeophora boehmi . However, these microfilariae are unsheathed and larger (length: 300 to 330 μm, width: 6 to 7 μm). Elaeophora boehmi has only been reported from Europe and Asia.
Other causes of equine parasitic encephalomyelitis in horses include Strongylus vulgaris, Strongylus equinus, Angiostrongylus cantonensis, Draschia megastoma, Halicephalobus deletrix, Hypoderma species and Sarcocystis neurona.
Control
Treatment: The macrocyclic lactone ivermectin has been reported to have both macrofilaricidal and microfilaricidal properties. Macrofilariae in the anterior chamber of the eye can be removed surgically.
Control: Control is not necessary because of the very minor clinical importance of S. equina.
Cestodes
Horse tapeworm (Anoplocephalidosis)
Anoplocephala species
Anoplocephaloides
General
Anoplocephalidosis of horses is caused by Anoplocephala perfoliata, Anoplocephala magna, and Anoplocephaloides mamillana (syn. Paranoplocephala mamillana) –all members of the family Anoplocephalidae. Also known collectively as anoplocephalids, many characteristics are shared particularly with respect to morphology, life cycle and epidemiology. In South Africa, as in Europe and North America, A. perfoliata is the most commonly encountered tapeworm of horses.
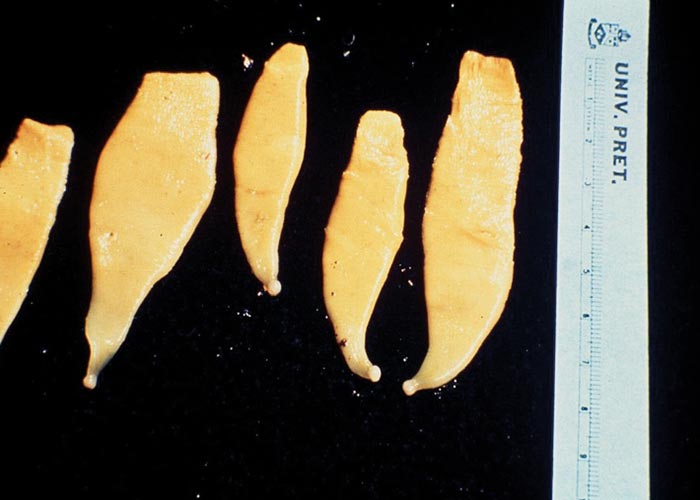
The family and genus names are derived from the Greek words ‘anoplos ’ (unarmed) and ‘kephale ’ (head) –which is descriptive and refers to the scoleces that lack a rostellum and are fitted with 4 unarmed (devoid of hooks) suckers. The anoplocephalids of horses are small to mediumsized tapeworms ranging from 1 to 52 cm in length ( Figure 60 ). Depending on the species, the strobila consists of numerous proglottids of the craspedote type –which are wider than long and are provided with 1 set of reproductive organs. Genital pores are unilateral.
The eggs of all species of Anoplocephalidae are collectively referred to as anoplocephalid- type eggs or anoplocephalid eggs. Anoplocephalid eggs are thick-shelled and have an irregularly spherical shape. Inside the egg, the first larval stage, known as an oncosphere, is covered by the embryophore. The embryophore is typically elongated and pear-shaped, and is known as the pyriform apparatus (Latin ‘pirum ’ - pear, Latin ‘formis ’ - shaped).
The type of metacestode (second larval stage developing in intermediate hosts and infective stage for definitive host) developing in the life cycle of anoplocephalids is a cysticercoid.
The predilection site of the strobilar stages of A. magna and A. mamillana is the small intestine. The predilection site of A. perfoliata is the ileo-caecal valve –which is unusual for strobilar stages of tapeworms. Comparative morphological details regarding the anoplocephalids of horses covered in this chapter can be obtained from Table 8.
Hosts
Apart from horses, the definitive host range for the three species also includes mules, donkeys and zebras.
Distribution
The three species have a cosmopolitan distribution.
Transmission
Anoplocephalids have an indirect life cycle, which requires an oribatid mite as an intermediate host.
Gravid proglottids containing eggs –as well as eggs released from proglottids in the intestines –are shed into the environment with the faeces. The already embryonated eggs are immediately infective for oribatid mites and are ingested by the mites on the ground. Released oncospheres penetrate the gut wall and develop in the haemocoel to cysticercoids. The incubation period in mites is temperature-dependent and may take 2 to 4 months. Definitive hosts become infected by ingesting oribatid mites harbouring cysticercoids while grazing. The pre-patent period is 6 to 10 weeks.
Socio-economic importance
Colic is the most important cause of morbidity and mortality in horses and Anoplocephala perfoliata is regarded as a risk factor for this syndrome. The expense of diagnostic procedures, registered products for deworming, and veterinary costs in surgical colic cases are considerable –and beyond the reach of resource-poor animal owners.
Pathogenesis and pathology
Anoplocephala perfoliata and A. magna are associated with lesions and clinical signs whose severity is related to the worm burden. No pathogenic effects have been ascribed to A. mamillana.
Anoplocephala perfoliata: Attachment of the strobilar stage commonly causes obstructive lesions due to mucosal inflammation, oedema, ulceration and the formation of diphtheritic membranes. Eventually, this may result in catarrhal enteritis, ileal impaction, intussusceptions, caecal torsions and ruptures, with subsequent peritonitis developing.
Anoplocephala magna: Catarrhal or haemorrhagic enteritis –and even rupture of the small intestine –has been described in horses harbouring heavy worm burdens.
Clinical signs
The appearance and severity of clinical signs in horses is related to the worm burden. Apart from indigestion and episodes of diarrhoea, Anoplocephala perfoliata is regarded as a significant risk factor for colic in horses.
Catarrhal and haemorrhagic diarrhoea can be seen in horses harbouring heavy worm burdens of A. magna.
Diagnosis in live animals
Presumptive clinical diagnosis: A history of diarrhoea and recurrent episodes of colic might indicate the involvement of anoplocephalid tapeworms.
Parasitological diagnosis: The parasitological diagnosis is based on the demonstration of eggs in faecal samples. Since most eggs are retained in gravid proglottids, shedding of eggs is erratic and occurs at a very low level. Therefore, the sensitivity of faecal examination –irrespective of the coprological technique used –is unsatisfactory and leads to an underestimation of prevalence. The coprological technique of choice is combined centrifugal sedimentation/ flotation, which is more sensitive than routine faecal flotation techniques. There are numerous variations of the sedimentation/ flotation technique described in the literature.
Diagnostic deworming: Considering the difficulties in making a diagnosis in live animals, treatment with a specific cestocidal drug in suspected anoplocephalidosis should be considered.
Differential diagnosis
Other important helminth infections to consider in the syndrome of colic are cyathostomins, Parascaris equorum in particularly foals and yearlings, Gastrodiscus aegyptiacus , and large strongyles.
Control
Treatment: There are comparatively few anthelmintics that are reported to be effective for the treatment of anoplocephalidosis. As for many other cestode infections of domestic and wild animals, the isoquinoline praziquantel (1 mg/kg) is highly effective for anoplocephalidosis. Also indicated is the salicylanilide niclosamide (80 to 100 mg/ kg), and, although variable efficacy has been reported, also the tetrahydropyrimidine pyrantel (6.6 mg base/kg, 13.2 mg base/ kg, 19.8 mg base/kg).
Prevention: The control of this infection is highly problematic due to the ubiquity of oribatid mites. Regular removal of faeces, rotational grazing, ploughing and veld burning are measures to consider in problem studs –as they affect the oribatid mite populations.
Table 8 Anoplocephalids of horses
| Characteristics | A. perfoliata | A. magna | A. mamillana |
|---|---|---|---|
| Length (cm) | 1.5-8.0 | 52-80 | 1-5 |
| Maximum width of strobila (mm) | 8-14 | 25 | 4-6 |
Scolex
| 2-3 Spherical Ear-shaped lappet posterior to each sucker | 3-5 Circular No lappets | 1 Slit-like No lappets |
Egg
| Irregularly spherical 65-80 8-10 16 | Irregularly spherical 70-84 4 8 | Irregularly spherical 37-51 4 22 |
| Predilection site | Ileocaecal valve (caudal ileum, caecum, proximal colon) | Small intestine | Small intestine |
Trematodes
Conical Fluke (Gastrodiscosis)
Gastrodiscus aegyptiacus
General
Gastrodiscosis of horses is caused by Gastrodiscus aegyptiacus . The genus name is derived from the Greek words ’gastros ‘ (stomach, belly) and ’discus ‘ (disc). The species is a hermaphrodite, dorsoventrally flattened amphistome with a small, anterior conical part (4 mm long and 3 mm wide) and a disc-shaped posterior part that gives the worm the typical ‘tennis-racket ’-shaped appearance ( Figure 61 ). The oral and ventral suckers are situated terminally at the anterior and posterior extremities of the body, respectively. Fresh specimens are pink in colour. Adult worms are 12 to 18 mm long and 10 to 14 mm wide.
The predilection site of G. aegyptiacus is the caecum and colon. The eggs are oval and large-sized (140 to 170 x 90 to 100 μm). They are colourless, thin-shelled with a distinct operculum, and contain coarse granular material.
Hosts
Apart from horses, the definitive host range includes other equids, domestic pigs, and warthogs.
Distribution
Gastrodiscus aegyptiacus occurs through out Africa and there are also isolated records from India, Guyana, Suriname, Cuba and Guadeloupe.
Transmission
Gastrodiscus aegyptiacus has an indirect life cycle, which requires a freshwater snail as intermediate host. Sexually mature worms are oviparous. Eggs are shed with the faeces into the environment, but are highly sensitive to desiccation. Provided there is sufficient ambient moisture, eggs remain viable for several months. Depending on the prevailing temperature and availability of water, a first larval stage, known as miracidium, develops within the egg in as little as 14 to 16 days. The miracidium hatches from the egg and swims actively in the water.
The aquatic pulmonate freshwater snail Bulinus forskalii serves as the only known intermediate host. Typical habitats are permanent collections of water (swamps, lake margins, irrigation systems) –as well as small temporary and permanent water bodies. The snail has an Afrotropical distribution, but is not encountered in the cool highlands and the Western Cape Province of South Africa. The miracidium invades the snail and develops to a sporocyst. Within the sporocyst, several rediae are produced by means of polyembryony. Asexual reproduction continues within the rediae –which results in the formation of cercariae. The incubation period in the snail is temperature dependent and may be as short as 5 weeks. Cercariae leave the snail and encyst within 1 hour on partially inundated vegetation, near the water surface.
Similar to other paramphistomids and the liver flukes, the metacercarial stage appears to be partially resistant to desiccation. Ingesting metacercariae while grazing in swampy areas or by consuming contaminated hay harvested from infested areas, infects definitive hosts. Metacercariae excyst following ingestion. The emerging immature flukes do not undergo any extra-intestinal development, and attach to the mucosa of the colon. The pre-patent period for G. aegyptiacus –in horses –is about 5 months.
Socio-economic importance
Gastrosdiscus aegyptiacus can be an important cause of diarrhoea and colic in horses.
Pathogenesis and pathology
Pathogenicity appears to be related to worm burden. Although generally regarded as non-pathogenic in the past, there is evidence from South Africa and Guyana that a severe haemorrhagic and oedematous colitis could be attributed to the presence of large numbers of the parasite.
Clinical signs
Mild infections in horses are clinically inapparent and mostly undetectable. Recurrent colic attacks, chronic diarrhoea, weight loss, poor performance, and anaemia –as well as incoordination and collapse –were reported in horses with high worm burdens prior to death.
Diagnosis
The diagnosis in live animals is based on the demonstration of eggs in faecal samples, using sedimentation.
Differential diagnosis
Although rarely reported in South Africa, infections with Fasciola hepatica and Fasciola gigantica have to be taken into consideration. Fasciola eggs are also large-sized, oval, thin-shelled and operculate. However, in contrast to G.aegyptiacus, they are yellowish-brown and contain fine granular material.
Other important helminth infections to consider as a cause of colic are particularly cyathostomins, Parascaris equorum in foals and yearlings, Anoplocephala perfoliata , and large strongyles.
Control
Treatment: The organophosphate dichlorvos given at a dose rate of 35-40 mg/kg is highly effective against G. aegyptiacus infection in horses; however, there are no longer registered products available. As the only known alternative, the salicylanilide oxyclozanide at a dose rate of 8.5 mg/kg is used off label. Important to consider, is that oxyclozanide is only available in combination with levamisole in South Africa. Because of the low safety margin in equids and the limited efficacy against many equine nematodes, the use of levamisole is restricted to ruminants, pigs and poultry.
Prevention: Clinical gastrodiscosis in horses can be effectively prevented by proper farm and grazing management. Horses and other equids should ideally not share pastures with free-ranging pigs or be kept on pastures that can be accessed by warthogs. Horses should be watered with fresh water and the origin of hay should be carefully investigated. Banks of perennial and seasonal, stagnant, or flowing water bodies should be avoided as they are typical habitats of B. forskalii. Where possible, fencing off identified snail habitats can be effective.
Liver fluke (Fasciolosis)
Fasciola species
General
Fasciolosis of horses is caused by Fasciola hepatica and Fasciola gigantica, which are also colloquially known as the ‘liver fluke ’and ‘giant liver fluke ’, respectively. The genus name is derived from the Latin word ‘fasciola ’ (small ribbon). Fasciola species are hermaphrodite, dorsoventrally flattened, leaf-like distomes, with a distinct head cone. Microscopically, the cuticle is covered with scale-like spines. The oral sucker and the slightly larger ventral sucker are situated in close proximity at the head cone. Fresh specimens are pale brown.
Fasciola hepatica adult worms are 20 to 50 mm long and 4 to 13 mm wide (length to width ratio is ±2). Fasciola gigantica adult worms have a narrower body and are 25 to 75 mm long and 3 to 11 mm wide (length to width ratio is ±4.5).
The predilection sites of both species in the definitive host are the hepatic bile ducts. The immature and mature flukes are haematophagous and histophagous.
The eggs are oval, large-sized (F. hepatica 130-150 x 63-90 μm, F. gigantica 156-197 x 90-104 μm) and contain fine granular material. They are typically yellowishbrown in colour, thin-shelled, and have an indistinct operculum.
The metacercariae are circular, 310-350 μm in diameter and are attached to herbage in water or flotsam. They can detach and float free in water or settle at the bottom of water bodies.
Hosts
The definitive host range includes a broad spectrum of herbivorous and omnivorous mammals –including humans. The principal definitive hosts are cattle and small stock. Although horses and other equids are regarded as unusual hosts for Fasciola species, a high prevalence has been reported in endemic areas.
Distribution
Fasciola hepatica has a cosmopolitan distribution with a higher prevalence in temperate climates. In Africa it appears to be more restricted to the northern Mediterranean countries, as well as to southern Zimbabwe and South Africa.
Fasciola gigantica is more common in tropical and subtropical regions and is widespread in Africa and Asia.
Transmission
Fasciola species have an indirect life cycle, which requires a freshwater snail as an intermediate host.
Sexually mature worms are oviparous. Eggs are shed with the faeces into the environment, and are highly sensitive to desiccation and temperatures below freezing point. With sufficient ambient moisture, eggs remain viable for several months. Depending on the prevailing temperature and availability of water, a first larval stage, known as a miracidium, develops within the egg within as little as 10 to 15 days. The miracidium hatches from the egg and swims actively in the water.
Various freshwater snail species of the family Lymnaeidae act as intermediate hosts. According to their habitat requirements, lymnaeids can be divided into amphibious and aquatic species. In South Africa the aquatic snail Lymnaea (Pseudosuccinea) columella is an intermediate host for both Fasciola species. Likewise, the amphibious Lymnaea (Galba) truncatula is an intermediate host for F. hepatica and the aquatic (Radix) natalensis for F. gigantica. Lymnaea columella was originally a North American species. Introduced into South Africa, it is widely distributed and found with L. natalensis. The snail is known to move out of the water onto vegetation and objects. The species has an Afrotropical distribution, but in South Africa is not found in the Western Cape Province. Typical habitats are permanent water bodies (streams, impoundments, dams). Lymnaea truncatula has a mainly Holarctic distribution (in the northern hemisphere), and in South Africa is found in the cooler areas and Lesotho. Typical habitats are permanent collections of water (banks and edges of small streams and ponds) –as well as temporary bodies of rainwater such as puddles, hoof marks and wheel ruts. The miracidium invades the snail and develops to a sporocyst. Within the sporocyst, several rediae are produced by means of polyembryony. Under unfavourable conditions a second redial generation might also develop. Asexual reproduction continues within the rediae, which results in the formation of cercariae. The incubation period in the snail is temperature dependent and may take as little as 6 to 10 weeks. Cercariae leave the snail and encyst on aquatic vegetation, flotsam or directly in the water. The metacercarial stage is partially resistant to desiccation and is known to remain infective for 4 to 6 months in contaminated hay. High temperatures of over 40 °C, and excessive dryness, will kill the metacercariae within hours. Definitive hosts become infected by ingesting metacercariae attached to hay, aquatic vegetation, or drinking water from the bottom of infested water bodies. Metacercariae excyst following ingestion. The emerging immature flukes penetrate the wall of the small intestine and migrate intra-abdominally to the liver, where they are found in the parenchyma within 24 hours of infection. Migration in the liver parenchyma continues for 6 to 7 weeks before the flukes settle in the hepatic bile ducts. The pre-patent period for Fasciola species in horses is 2 to 3 months. Not all infections become patent in horses.
Socio-economic importance
In contrast to cattle and small stock, fasciolosis in horses is of minor clinical importance and seldom requires treatment.
Humans can act as definitive host for both Fasciola species. Cases of human fasciolosis have been reported from Africa, the Americas, Asia, Australia, Europe and Oceania. It is believed that human infection is under-reported. Vegetables grown in snail-infested habitats which are grazed by ruminants are a common source of human infection.
Pathogenesis and pathology
Horses demonstrate a pronounced resistance to liver fluke infection. Only a small percentage of ingested metacercariae reach the bile ducts –where they become sexually mature. Nevertheless, a high prevalence has been reported in various endemic areas. A chronic and a subacute form of fasciolosis have been described in horses. The chronic form is characterised by mild cholangitis, interstitial fibrosis, and eosinophilia. The subacute form is characterised by perihepatic fibrosis and peritonitis caused by immature flukes migrating through the liver parenchyma. Infection is easily overlooked during necropsy, as macroscopic lesions are rarely noticed –even in heavily infected horses.
Clinical signs
Most infections in horses are clinically inapparent. Only rarely does fasciolosis clinically manifest in its chronic or subacute form. The clinical signs observed in chronic fasciolosis are unspecific and comprise inappetence, weight loss, diarrhoea, light anaemia, and icterus. Abdominal oedema and colic-like symptoms have also been reported in the subacute form.
Diagnosis
The diagnosis in live animals is based on the demonstration of eggs in faecal samples by using sedimentation. Since only small numbers of eggs are shed, larger faecal samples (50 g) should be examined.
Differential diagnosis
Common infections with Gastrodiscus aegyptiacus have to be taken into consideration. The eggs of G. aegyptiacus are also large, oval, thin-shelled and operculated. However, in contrast to Fasciola species, they are colourless and contain coarse granular material.
Control
Treatment: The benzimidazole triclabendazole (12 mg/kg) is highly effective for adults of Fasciola hepatica; however, immature flukes are not always completely removed.
Prevention: Fasciolosis in horses can be effectively prevented by proper farm and grazing management. Horses should ideally not share pastures with ruminants. Marshy areas, which constitute potential lymnaeid snail habitats, should be avoided –particularly if grazed previously by domestic ruminants. Fencing off identified snail habitats can be effective.