- Infectious Diseases of Livestock
- Part 1
- Chlamydiosis
- Vectors: Ticks
- Vectors: Tsetse flies
- Vectors: Muscidae
- Vectors: Tabanidae
- Vectors: Culicoides spp.
- Vectors: Mosquitoes
- Classification, epidemiology and control of arthropod-borne viruses
- Special factors affecting the control of livestock diseases in sub-Saharan Africa
- The control of infectious diseases of livestock: Making appropriate decisions in different epidemiological and socioeconomic conditions
- Infectious diseases of animals in sub-Saharan Africa: The wildlife⁄livestock interface
- Vaccination: An approach to the control of infectious diseases
- African animal trypanosomoses
- Dourine
- Trichomonosis
- Amoebic infections
- GENERAL INTRODUCTION: COCCIDIA
- Coccidiosis
- Cryptosporidiosis
- Toxoplasmosis
- Besnoitiosis
- Sarcocystosis
- Balantidiosis
- Leishmaniosis
- Neosporosis
- Equine protozoal myeloencephalitis
- GENERAL INTRODUCTION: BABESIOSES
- Bovine babesiosis
- Equine piroplasmosis
- Porcine babesiosis
- Ovine babesiosis
- GENERAL INTRODUCTION: THEILERIOSES OF CATTLE
- East Coast fever
- Corridor disease
- Zimbabwe theileriosis
- Turning sickness
- Theileria taurotragi infection
- Theileria mutans infection
- Theileria annulata theileriosis
- Theileriosis of sheep and goats
- Theileria buffeli⁄orientalis infection
- Non-pathogenic Theileria species in cattle
- GENERAL INTRODUCTION: RICKETTSIAL, CHLAMYDIAL AND HAEMOTROPIC MYCOPLASMAL DISEASES
- Heartwater
- Lesser known rickettsial infections in animals and humans
- Chlamydiosis
- Q fever
- Eperythrozoonosis
- Bovine Haemobartonellosis
- Potomac horse fever
- GENERAL INTRODUCTION: ANAPLASMOSES
- Bovine anaplasmosis
- Ovine and caprine anaplasmosis
Chlamydiosis
This content is distributed under the following licence: Attribution-NonCommercial CC BY-NC  View Creative Commons Licence details here
View Creative Commons Licence details here
NJ Maclachlan and M-L Penrith (Editors). N Borel and D Longbottom, Chlamydiosis, 2019.
Chlamydiosis
Previous authors: A A ANDERSEN
Current authors:
N BOREL - Dipl. ECVP, FVH Pathology, Institute of Veterinary Pathology, Vetsuisse Faculty, University Zurich, Winterthurerstrasse 268, Zurich, 8057, Switzerland
D LONGBOTTOM - Head of Department of Diagnostics, Head of Chlamydia Research, BSc (Hons), PhD, Moredun Research Institute, Pentlands Science Park, Bush Loan, Edinburgh, Midlothian, EH26 0PZ, Scotland
Introduction
The taxonomic diversity of the order Chlamydiales has been significantly expanded over the last 20 years resulting in the addition of thirteen families of genetically related organisms collectively referred to as “Chlamydia-related bacteria”.46, 105, 157 In this chapter, we will focus on the family Chlamydiaceae containing the single genus Chlamydia and 12 currently accepted species (C. abortus, C. avium, C. caviae, C. felis, C. gallinacea, C. ibidis, C. muridarum, C. pecorum, C. pneumoniae, C. psittaci, C. suis and C. trachomatis). All of these species are able to infect animals (main hosts are non-human mammals and birds) and some also have the ability to infect humans, with C. trachomatis being the only exception as solely a human pathogen. Chlamydiosis in non-avian livestock is caused by three species, C. abortus, C. pecorum and C. suis, while in birds infection is caused by C. psittaci, C. avium, C. ibidis and C. gallinacea, all of which are the focus of this chapter. The terms chlamydiosis and chlamydia(e) will be used here to refer generally to the diseases and members of the Chlamydiaceae family, except when a reference to specific diseases and species is necessary to avoid confusion.
Chlamydiosis is characterized by the development of a variety of clinical syndromes depending on the species and strain of Chlamydia, the host species, and the affected organ system. A wide range of hosts may be affected by chlamydial diseases, including horses, cattle, pigs, sheep, goats, birds and poultry. Chlamydial strains can be fairly host specific, primarily infecting a given species or type of animal and causing specific clinical diseases, but recent evidence has demonstrated that many chlamydial species are not restricted to one particular host and host species jumping may occur.156 These diseases may affect the gastrointestinal, respiratory, reproductive and nervous systems, as well as the joints and eyes. The severity of disease can vary from subclinical to life-threatening, but the usual presentation is a mild to moderate clinical disease leading to persistent infection. Recurrence of the disease and/or shedding of the organism can occur following stress or physiological changes such as oestrus. What is currently unknown is the extent of synergistic effects these persistent chlamydial infections may have with concurrent infections with other pathogens.
Avian chlamydiosis is the disease caused by Chlamydia psittaci in birds.4 It has been called psittacosis and ornithosis depending on the type of bird it was infecting or the type of bird from which it was contracted. Psittacosis and ornithosis are now considered to be the same disease, the term avian chlamydiosis being preferred. Chlamydia psittaci is distributed worldwide and has been reported in nine domestic fowl species and at least 460 free-living bird species frequently kept as pets in 30 different orders.66 In birds, infections caused by C. psittaci can be subclinical or result in respiratory, enteric and ocular signs and it is the most important veterinary chlamydial agent in terms of economic and zoonotic impacts. Recently, avian chlamydial species have been extended to include the novel species C. avium, C. gallinacea, and C. ibidis.120, 165 Chlamydia avium and C. gallinacea have been reported in pigeons, psittacine birds and mallards (C. avium) and domestic poultry, guinea fowl, turkeys and ducks (C. gallinacea) in Europe, Asia, China, Argentina and more recently in North America, while C. ibidis has been isolated from feral Sacred Ibis. Assessment of their pathogenicity, potential hosts, transmission routes and zoonotic potential warrants further research. More information on avian chlamydiosis due to C. psittaci is available in several reviews.121, 161
Chlamydiosis was first recognized in 1879 by Ritter as a disease that could spread from parrots to humans, when a Swiss physician observed respiratory illness in a household with a sick parrot.108 Avian chlamydiosis gained world prominence during a pandemic in 1929 to 1930 that involved over 12 countries in Europe and North America. The outbreak led to strict regulations on the importation of parrots from South America. In 1930, Bedson and colleagues, Coles, Levinthal, and Lillie independently observed for the first time very small basophilic bodies in the tissues of infected birds and humans, and suggested that they were the causative agent.10, 24, 76, 77 In the same year, Bedson and colleagues also established the aetiological relationship between the basophilic bodies and the disease.8, 9 It soon became clear that chlamydial infections were not limited to psittacine birds, but that they were widespread in almost all avian species, and that chlamydiae from other birds were transmissible to humans. Chlamydial infection in pigeons was first reported in South Africa in 1940 by J.D.W. Coles after a pigeon fancier who had been experiencing mortalities in his flock sent two affected birds to the Onderstepoort Veterinary Institute.25 For a full historical overview of the first descriptions of chlamydial infections in animals see.82
Enzootic abortion in ewes was first described in 1936 by Greig following abortions occurring in sheep in Scotland in the UK,50 although it was not until 1950 that it was shown by Stamp and his colleagues to be caused by chlamydial infection.143 Since then chlamydial abortion has been recognized worldwide. It was first seen in South Africa, in 1972, when a severe epidemic of abortions swept through the sheep-raising areas within a single lambing season, resulting in up to 60 per cent abortions in some flocks.133 In cattle, Schoop and Kauker131 furnished serological and cytological proof that incriminated Chlamydia as the cause of abortions in a dairy herd in 1956. In California, Kennedy and co-workers in 196070 described the pathological lesions of aborted calves and, from some of them, Storz et al. in the same year149 isolated the chlamydial agent.
Because of the sporadic occurrence of outbreaks, the disease was named epizootic bovine abortion. The agent is similar to or the same as that causing abortion in sheep, in which the disease is called enzootic abortion of ewes (EAE), ovine enzootic abortion (OEA) or ovine chlamydiosis.
Thereafter, chlamydiae in ruminant livestock were also found to be involved in causing enteritis, pneumonia, arthritis, conjunctivitis, and encephalomyelitis.82 These diseases are usually caused by Chlamydia pecorum strains, which are different from the abortion strain (C. abortus), and outbreaks are usually sporadic, primarily affecting young animals. Morbidity may be high, but mortality is usually low.
Aetiology and life cycle
Historical literature on Chlamydia and its taxonomy contain many later-abandoned terms that are, however, still important in order to understand the literature. Chlamydiae were once classified as viruses and grouped with other filterable organisms that required living cells in which to multiply. Some of the names in the literature include the psittacosis-lymphogranuloma-trachoma (PLT) group, Miyagawanella, Bedsonia, Prowazekia, Rakeia, and trachoma inclusion conjunctivitis.4, 82, 103 However, the use of a chlamydia-like name dates back to 1907, when Halberstaedter and von Prowazek53, 54 described characteristic intracytoplasmic microorganisms within vacuoles in conjunctival scrapings from a human patient with trachoma. They thought that the organisms were protozoa and named them Chlamydozoa after the Greek word ‘chlamys’, which means mantle. The term Chlamydia was proposed in 1945 by Jones63 and later reaffirmed by Page.96, 97
The early classification of the family Chlamydiaceae has been filled with controversy,96, 97 much of which subsided when Page proposed that all chlamydiae be grouped into one genus (Chlamydia)96 containing two species (C. trachomatis and C. psittaci).97 The division was based on susceptibility to sulphadiazine and accumulation of glycogen in the vacuoles when grown in non-human vertebrate tissues. It also effectively separated the known human strains from the animal strains, with only a few exceptions. At the time it was recognized that there was great diversity in C. psittaci. This classification remained unchanged until 1989 when C. pneumoniae was proposed as a third species.49 At that time it was thought to be solely a human respiratory pathogen. However, isolates have since been made from koalas (Phascolarctus cinereus)48, 171 and other marsupials145 and a horse,145 as well as reptiles and amphibians.12 In 1992, C. pecorum was proposed as a fourth species.47 It was originally considered to include isolates from cattle and sheep that caused pneumonia, polyarthritis, encephalomyelitis, and enteritis. Since then, C. pecorum strains have been isolated or identified from other ruminants as well as from pigs130 and koalas.59, 171
More recently, a reclassification of the family Chlamydiaceae based on sequence analysis of 16S and 23S rRNA genes was proposed in 1999.46 This resulted in the division of the original genus Chlamydia into two genera Chlamydia and Chlamydophila, as well as the creation of five new species (Chlamydophila abortus, Chlamydophila felis, Chlamydophila caviae, Chlamydia muridarum and Chlamydia suis), as well as a change in genus for three of the existing species (Chlamydophila psittaci, Chlamydophila pecorum and Chlamydophila pneumoniae). The resulting division reflected a high degree of correlation with host range and disease syndrome, and thus particularly helped in the understanding of the epidemiology of the various strains affecting animals. However, while there was general acceptance of the new species designations, there was great resistance to accept or adopt the new genus Chlamydophila, and ultimately, following a proposal in 2011 in Bergey’s Manual of Systematic Bacteriology, the family Chlamydiaceae has since reverted to a single genus, Chlamydia.58, 127, 144 The currently recognized species classification, together with the primary host ranges and disease syndromes, is shown in Table 1.Table 1 Current classification of the family Chlamydiaceae (as of March 2019)
| SPECIES | PRIMARY HOST(S) | PRIMARY DISEASE(S) |
|---|---|---|
| Chlamydia trachomatis | Humans | Trachoma, lymphogranuloma venereum, conjunctivitis |
| Chlamydia muridarum | Mice, hamsters | Pneumonia, ileitis |
| Chlamydia suis | Pigs | Pneumonia, conjunctivitis, enteritis |
| Chlamydia pneumoniae | Humans, horses, koalas, reptiles | Pneumonia, systemic disease |
| Chlamydia pecorum | Sheep, goats, cattle, swine, koalas | Encephalomyelitis, conjunctivitis, enteritis, polyarthritis, pneumonia |
| Chlamydia caviae | Guinea pigs | Conjunctivitis, genital tract infection |
| Chlamydia felis | Cats | Rhinitis, conjunctivitis, pneumonia |
| Chlamydia abortus | Sheep, goats, cattle | Abortion |
| Chlamydia psittaci | Psittacine birds, poultry | Conjunctivitis, pneumonia, systemic disease |
| Chlamydia avium | Psittacine birds, pigeons, mallards |
|
| Chlamydia gallinacea | Poultry, guinea fowl |
|
| Chlamydia ibidis | Feral Sacred Ibis |
|
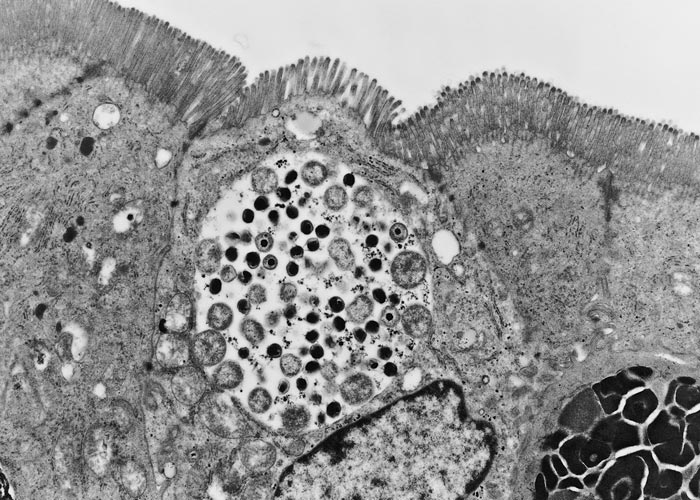
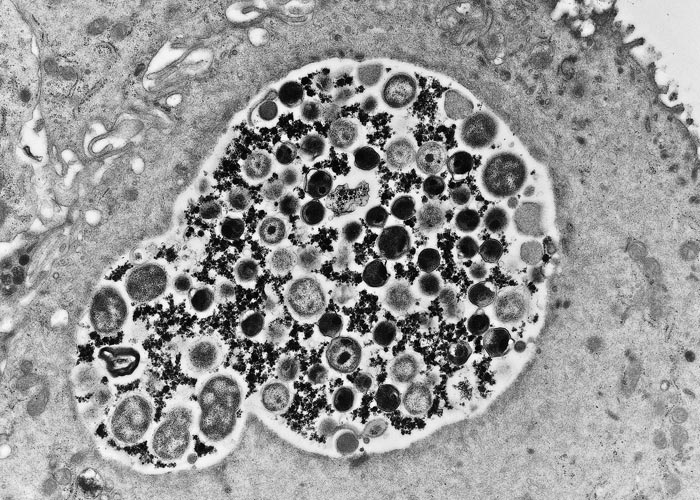
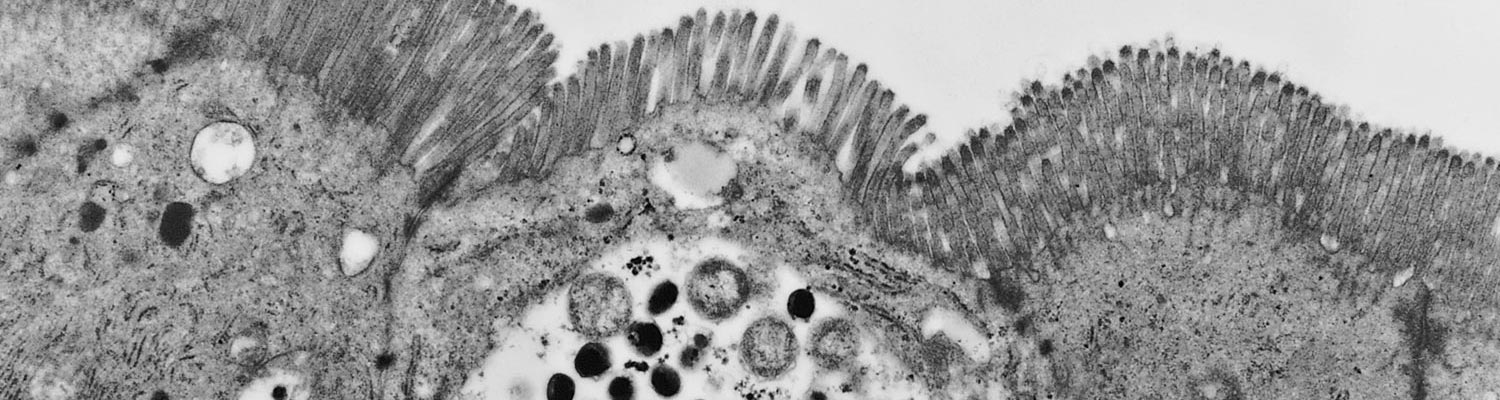
Chlamydiae are obligate intracellular bacteria that multiply in inclusions in the cytoplasm of eukaryotic cells. They have a unique growth cycle consisting of two developmental forms. The elementary body (EB) is the extracellular infectious condensed form of 200 to 300 nm in diameter that is suited for survival in the environment, which in the case of animal pathogens may mean many months outside the natural host. This resistance to physical and chemical extracellular factors is a consequence of the rigidity of the EB cell envelope and the greatly reduced EB surface area. The reticulate body (RB) is the metabolically active replicating form, ranging in size from 0.5 to 1.6 µm, and found in a cytoplasmic inclusion within the host cell (Figures 1 and 2). Replication is by binary fission typical of other bacteria, with the exception that chlamydiae largely rely on the host cell for energy. Intermediate forms between the RB and the EB are usually seen in the inclusion.169
The developmental cycle of chlamydia consists of five major phases:
- the attachment and entry of the EB into the host cell;
- the transition of the metabolically less-active EB into the metabolically active RB;
- multiplication of the RB by binary fission using energy from the host cell;
- maturation of the metabolically active RB into the stable infectious EB; and
- the releaseof the EB from the host cell.93, 150
A critical stage in the chlamydial developmental cycle is the phase following entry into the host cell. The EB must transform into the metabolically active RB and escape from the host cell defences while transforming. The normal cell processes endosomes containing foreign material by acidification and fusion with lysosomes. The digestion and processing of foreign antigens by lysosomes is critical for the processing and presentation of antigens during the immune response. It has been shown that live chlamydiae prevent the acidification of the endosomes and the subsequent fusion with lysosomes.132, 154
Epidemiology
Chlamydiae are among the more successful pathogens known. It was once thought that chlamydial isolates are all similar and that each infected a wide host range. New molecular and serological techniques now show that there are numerous identifiable chlamydial strains and that the isolates from a particular avian or mammalian species tend to be of the same species. This pattern indicates a triangular relationship among the chlamydial strain, the host and the disease syndrome. Research is increasingly showing that when a strain infects its natural host persistent infections are common170 and might play a role in the maintenance of the chlamydial organism in nature. It should be remembered that a number of the avian and mammalian strains can infect other hosts, including humans, with severe consequences, but secondary spread from these infections appears to be limited.
The role played by wild animals in the transmission of chlamydiae needs to be fully assessed. Natural infections have been reported in opossums (Didelphis paraquayensis), northern fur seals (Collorhinus ursinus), muskrats (Ondatra zibethica), snowshoe hares (Lepus americanus), koalas (Phascolarctus cinereus), springbok (Antidorcas marsupialis), ibex (Capra pyrenaica), and bighorn sheep (Ovis canadensis).103
Molecular and serological typing now make it possible to determine whether these and other wild animals are reservoirs of chlamydiae for domestic animals, or whether these species have their own host-strain relationships. Molecular studies of isolates from koalas have shown these animals to harbour both Chlamydia pneumoniae and Chlamydia pecorum strains.59, 171 These strains, however, are distinct from those found in other animals and humans.46 The snowshoe hare and muskrat isolates are identical (strain M56) and found to be similar to the avian strains.46, 141 Conversely, the M56 strain found in snowshoe hares and muskrats has never been isolated from birds. On the other hand, a C. pecorum isolate from bighorn sheep appears the same as the strain causing polyarthritis and conjunctivitis in domestic sheep.2
Transmission of chlamydiae probably occurs via several routes, as chlamydiae have been found in all body secretions, including faeces, as well as in biological products associated with foetal death (including placenta, dead foetuses, vaginal excretions). The faecal–oral route may be the most common for most species.43, 44 Aerosol and contact transmission may be more common in respiratory tract and eye infections, respectively. Insect transmission, especially by flies, must be considered.
The importance of vertical transmission is not fully understood, but generally this route has not been thought to be a major factor41, 82 in the transmission of chlamydial infections. There are, however, previous reports of infections of the male accessory sex glands and of the presence of chlamydiae in the semen of bulls, suggesting that venereal transmission may play a role.148 However, in contrast to the human pathogen C. trachomatis, there is very little evidence supporting a role for sexual transmission for the livestock pathogen C. abortus.7, 99, 158, 174 In ewes that have aborted, chlamydiae have been reported to be shed during the following periovulation period and at the subsequent lambing; however, evidence from molecular analysis of vaginal swabs taken at these points suggests that any such shedding poses a minimal risk of transmission to naïve animals.52, 75, 80, 100 Thus, horizontal transmission appears to provide the greatest risk of transmission from one animal to another.82
Pathogenesis
Chlamydial infections are responsible for a wide variety of syndromes in domestic animals, including enteritis, pneumonia, polyserositis, lymphadenopathy, keratoconjunctivitis, mastitis, polyarthritis, meningoencephalomyelitis, epididymitis, orchitis, seminal vesiculitis, and placentitis. The chlamydial species and strains each have a predilection for different tissues and therefore cause different disease syndromes. The route of infection may play a role in the type of disease syndrome; nevertheless, oral or parenteral inoculation of a given strain usually results in the development of the same disease syndrome from which the strain was originally isolated.
The oral route of exposure is considered the primary means of transmission for most strains.42, 44 It has been shown that most strains readily infect the epithelial cells of the gastrointestinal mucosa. The organisms localize in the epithelial cells of the mucosa in the jejunum and ileum, and then spread via the blood and lymph vessels of the lamina propria to the mesenteric lymph nodes and the liver. Thereafter, infection may be established in other organs or tissues.
With abortion strains, evidence indicates that infection occurs principally by the systemic route, causing a chlamydaemia that results in spread to the placenta and the subsequent extensive placental damage. The extent of placental damage may determine whether the lamb is born apparently healthy or weak, or is stillborn or aborted.21, 82, 111 Placental infection can impair maternal-foetal transfer of nutrients, resulting in impaired growth or death of the foetus. Placental infection may also alter placental steroid and prostaglandin release, resulting in premature labour.75 In some cases, a ewe bearing twins or triplets may deliver one or more healthy and one or more weak or dead lambs, with one or more affected and unaffected placentas. It is likely that differences in the immune response, hormonal imbalance resulting from the destruction of the cells producing progesterone and placental insufficiency resulting from destruction of the placental tissue by the chlamydiae ultimately determine pregnancy outcome.82 However, the reasons why infection can occur in one placenta and not the other, resulting in one infected and one uninfected placenta and thus one dead and one live animal, respectively, is currently unknown.
Abundant evidence exists that most chlamydial species are capable of producing latent infection that can endure for months or years in the absence of treatment.170 The site and mechanism of latency is not known. Persistent infections with intermittent shedding of viable organisms have been identified following chlamydiosis in humans, animals and birds. Immunosuppression with corticosteroids frequently reactivates shedding in these cases.
Chlamydiae can infect and multiply within macrophages. This ability has attracted attention because the chlamydial inclusion is capable of avoiding lysosomal fusion,132, 154 which is necessary in the normal pathways of processing an antigen in order to evoke an immune response. It is apparent that live chlamydiae are needed to initiate the avoidance mechanism, as inactivated chlamydiae are processed by macrophages.
Clinical signs and pathology
Clinical signs and related pathology in affected animals vary according to the chlamydial species or strain and the syndromes from which the animals are suffering. Information on the prevalence, pathogenesis and pathology for many of the disease syndromes is limited. This discussion focuses primarily on those diseases considered to be of major importance.
Sheep, goats and cattle
Chlamydiae in ruminants have been primarily associated with abortions, polyarthritis, conjunctivitis, encephalomyelitis, enteritis, pneumonia, mastitis, and genital infections such as epididymitis and orchitis. Abortions are probably the most dramatic clinical outcome, having both economic and public health significance. They are most commonly caused by Chlamydia abortus, but an abortion in a cow caused by infection with Chlamydia psittaci serovar B has been documented.26 This occurred in a dairy herd where pigeons were common in the barnyard. In addition, a bovine abortion caused by the chlamydia-like organism Waddlia chondrophila WSU86-1044 has been reported.38, 71, 119The C. abortus strain has also been associated with mastitis67 and other forms of genital infection.151, 163, 176 Chlamydia abortus strains appear serologically and genetically similar worldwide, with the exception of a couple of isolates from Greece that appear more distinct and have been designated as ‘variant strains’.79, 134, 137, 166 There is also growing genotypic evidence for the detection of C. abortus strains of avian origin that may be more distantly related and are classified as evolutionary intermediate strains between C. psittaci and C. abortus.153, 160 The polyarthritis, conjunctivitis, encephalomyelitis, enteritis and pneumonia disease syndromes are considered to be caused primarily by various strains of Chlamydia pecorum.82, 167There is some indication that there are strain differences within the C. pecorum group, and that each strain tends to produce a specific type of disease or disease syndrome.
Chlamydial abortions in sheep and goats, also known as enzootic abortion, can occur as an acute outbreak in flocks or herds (where it is referred to as epizootic), or as an endemic infection. The usual pattern of infection that occurs in a naïve sheep flock is a small number of abortions in the first year following the probable introduction of infected replacement animals, followed in the second and third years by an abortion storm in 30 per cent or more animals, mainly affecting gimmers (sheep of one to two years of age also known as yearlings, hoggets, shearlings, theaves or tegs) and then an enzootic phase where younger ewes infected in the previous season are affected.1, 82, 111 In goats abortion rates of more than 60 per cent can occur. The abortion storm is generally followed by an annual abortion incidence of 5-10 per cent unless measures are put in place to control infection.91 Sheep and goats can become infected at any age and during any season, but the highest period of risk is at lambing time.82 The major sources of infection are the placental membranes, dead foetuses, coats of live lambs/kids born to infected mothers as well as vaginal discharges. Chlamydiae can remain viable in the environment for at least several days, with the potential to spread infection. Survival of chlamydiae at temperatures close to or below freezing is likely to be prolonged for weeks or even months.82
A number of human abortions followed by severe complications have been associated with exposure to infected placentas of ewes and does.19, 27, 64 The prevalence of such abortions is not known, as infectious causes are often not looked for in humans. Chlamydial infections must, however, be considered an occupational hazard for pregnant women who come into contact with domestic ruminants.41, 138
Ewes exposed to C. abortus before they have become pregnant may abort through a process known as latency, during which the organism has been found to be undetectable.84, 99, 174 In a recent study, during the development of a model of latency in non-pregnant sheep, it was observed that this can be achieved using relatively low doses of organisms inoculated intranasally, with the animals aborting late in gestation.84 In another study, seven ewes were exposed by rubbing infected placental material on their noses two days after they had lambed.174 During the following year two of them aborted as a result of chlamydial infection. It has been suggested that in such cases infection is first established in the tonsils, from where it is disseminated via blood and lymph to other organs.52, 62, 84 Whether the infection persists in the tonsils or in other organs of non-pregnant animals is not known, but it is thought that this latent infection eventually results in recrudescence of infection once the animal becomes pregnant, resulting in a low-grade chlamydaemia that ultimately results in the placentitis and abortion that are characteristic of this disease.
Research has shown that regardless of the point of infection, chlamydial growth and pathology are not observed until after around Day 85-90 of gestation.21, 88, 111 The reason for this is not clear but it has been hypothesized that it is due to the chlamydiae being able to access the trophoblast cells lining the foetal chorionic villi following the formation of haematomas in the septal tips of the hilus of the ovine placentome. These haematomas result from the leakage of maternal blood from peripheral capillaries, which occurs around Day 60 of gestation.21, 82 The 25-30-day period between the formation of the haematomas and the first observation of pathological changes in the placenta raises the question as to whether endocrinological and/or immune-related changes also play a role.21 Chlamydial inclusions and lesions can first be detected in the placenta after around Day 85 of gestation. The lesions first involve the limbus of the placentomes in the hilar region. At this time local necrosis occurs and the infection then spreads to involve the cotyledonary and intercotyledonary placental membranes and opposing endometrium. A mixed-cell inflammatory infiltrate and oedema is present, resulting in the characteristic thickened and reddened appearance of the placental membranes and cotyledons.20, 21, 88, 124 Underlying the lesions, fibrin deposits and nonpurulent arteritis are present in the thickened placental mesenchyme. Chlamydial inclusions are present in the epithelial cells of the endometrium where the affected chorion is adjacent to the maternal tissues. Severe necrosis and sloughing of the endometrial epithelium occurs during later stages of pregnancy.
In sheep, infection of the foetus is secondary to the placentitis and is not a major factor in the disease. Focal disseminated necrosis may be evident in the liver and other organs of the foetus and may be embolic in origin.21 Chlamydial antigen, if present in the foci of necrosis, usually occurs only in small amounts. The mesenteric and popliteal lymph nodes may be enlarged, having a demarcated cortex with several follicles and germinal centres.
The placental lesions may lead to death of the foetus and abortion. If the disease process is not extensive it may lead to the premature birth of a weak lamb that dies shortly after birth or to one that may live but remains unthrifty. Ewes are rarely affected clinically and remain fertile, although they can develop unrelated secondary bacterial infections. This is in contrast to the disease in goats and cows in which there is tendency for placentas to be retained and for the development of vaginitis and persistent endometritis. Having once aborted, sheep are solidly immune and will not abort again, even though they may remain persistently infected.100, 101, 111
In cattle, chlamydiosis causes abortions, stillbirths, or the birth of weak calves.31, 32, 40, 51, 56, 147, 149 Abortions are usually sporadic, but may reach 20 per cent in a herd and usually occur during the last trimester, affected cows showing little evidence of disease. The abortion is caused by placentitis, but the aborted foetus may manifest ascites, serofibrinous pleuritis and peritonitis, an enlarged reddish-yellow liver, and hyperplastic lymph nodes. Calves born weak but survive usually remain unthrifty.
In the UK, a syndrome in cows characterized by a drop in milk production, mucoid/mucopurulent oculonasal discharges, depression and anorexia over an eight-week period has been reported. Recovery is slow but appears to be aided by administration of tetracyclines. Abortion is common weeks to months after the event. The only consistent finding is that chlamydial antigen and/or rise in antibody titre to chlamydiae have been demonstrated in affected herds.31, 32
A seminal vesiculitis syndrome (SVS) has been reported in bulls.148 It is characterized as a chronic inflammation of the accessory sex glands, epididymes, and testicles. Infected bulls have inferior semen quality and may develop testicular atrophy. The syndrome is thought to be widespread globally. An isolate from a bull suffering from the syndrome was found to be indistinguishable from the EBA59-795 isolate of Chlamydia, indicating that the strain is the same as that causing abortion. The SVS can possibly be regarded as the counterpart of the abortion syndrome in cows. Similar diseases or syndromes have been reported in sheep.86, 103 In open cows, cervicitis and endometritis with a non-putrid discharge have been associated with chlamydial infections.163, 176
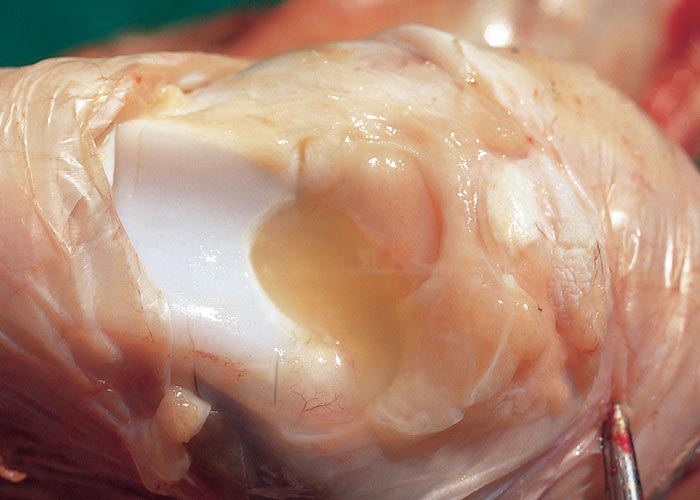
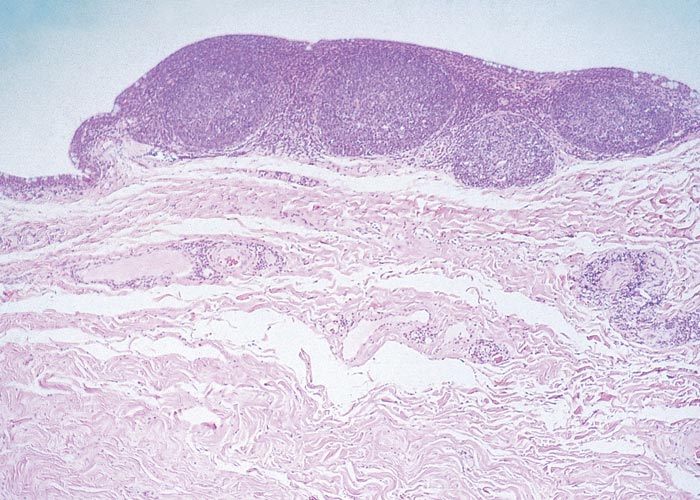
Polyarthritis in sheep, goats and cattle may be caused by strains of C. pecorum.47, 168 The disease can be readily reproduced experimentally by inoculating lambs or calves by oral, intramuscular, subcutaneous, intravenous or intra-articular routes.30, 44 In the natural disease, the route of infection is thought to be oral, the organism multiplying in the mucosa of the small intestine. Diarrhoea may be present during this phase. Following multiplication at the primary site, the organism spreads haematogenously to other organs and tissues of which the diarthrodial joints are usually the most severely affected (Figure 3). Lesions occur in both the peri-articular and articular tissues of the joints,28, 29, 136, 146 which usually are grossly enlarged because of localized subcutaneous oedema and an increase in the amount of fluid in the synovial sacs. The fluid is greenish-yellow and turbid.
In advanced cases, fibrin plaques may be present in the joints. In severe cases excess fluid of a similar nature may be present in the tendon sheaths. Muscle involvement is primarily limited to mononuclear cells in perivascular areas near the point of tendinous attachment. Histopathological changes in the synovium, tendon sheaths, and subsynovial tissues include infiltration of neutrophils, macrophages and lymphoid cells. Changes in cells in the synovial lining can vary from necrosis to proliferation.28-30
Conjunctivitis frequently accompanies polyarthritis in lambs and may be a prodromal sign of a developing outbreak of polyarthritis. Chlamydial conjunctivitis is usually bilateral, and clinical signs vary from a profuse watery to mucopurulent discharge which, in some animals, seals the eyelids together, and hyperaemia of the conjunctiva.57 In severe cases, photophobia and keratitis occur. Secondary bacterial infections of the conjunctiva are common.
Sporadic bovine encephalomyelitis (SBE) is a disease that has been reported worldwide,55, 90, 102, 104 including in South Africa.159 It primarily affects young calves, but has been known to occur in adult cattle and sheep. Generally, the morbidity in cattle is under 10 to 15 per cent, with mortality of up to 30 per cent in affected animals. A generalized chlamydial infection usually precedes or occurs concurrently with central nervous system (CNS) involvement. The principal clinical signs include moderate fever, stiffness, incoordination and depression. Gross lesions are indicative of systemic infection and include serofibrinous peritonitis, pleuritis, and sometimes pericarditis, similar to those seen in other C. pecorum infections. Gross lesions in the CNS are not as evident and are often limited to meningeal congestion and oedema. Histopathologically, the CNS lesion is diffuse non-purulent meningoencephalomyelitis which is especially marked in the cerebellum. The lesions are characterized by perivascular and focal infiltrations of mononuclear cells, focal areas of encephalomalacia and neuronal degeneration.55 In sheep, the clinical signs and lesions are similar to but generally milder than those in cattle.
Enteritis and interstitial pneumonia caused by chlamydial infections have been reported in sheep and cattle.43, 44, 107, 147 The isolates that have been characterized to date from such cases have been identified as C. pecorum. There appear to be overlaps in the clinical signs and lesions with those occurring in the polyarthritis, conjunctivitis and SBE syndromes, indicating different tissue predilections for the various strains of C. pecorum. Both syndromes primarily affect young animals. The enteritis syndrome is characterized by a mucoid to watery or bloody diarrhoea in young calves and lambs. The onset of the disease is rapid and the severity depends on the virulence of the strain and the degree of protection afforded by colostral antibodies.42, 43, 103, 147 On necropsy, focal to generalized peritonitis may be seen. Petechial haemorrhages in the abomasum and intestine occur in infections by highly virulent chlamydial strains. Hyperaemia and congestion of the mucosa are common, and enlargement of the lymph nodes may be seen. The ileum is usually the most severely infected region, but lesions may occur throughout the intestinal tract.42, 43, 147 Histologically, chlamydial inclusions are primarily seen in the villus enterocytes and occasionally in the cryptal enterocytes. Villus atrophy and multifocal necrosis of the villi are often extensive. Peyer’s patches may lose their follicular architecture and contain necrotic centres.42, 43, 103, 147
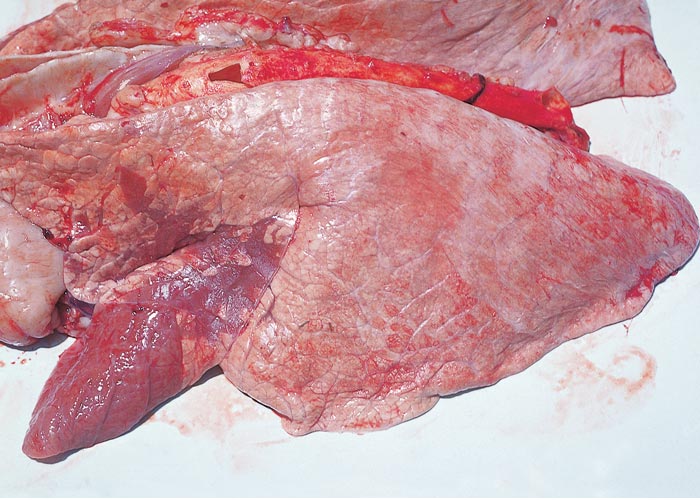
Calves and lambs affected with chlamydial pneumonia have fever and a serous to mucopurulent nasal discharge. They are depressed, cough, and have signs of dyspnoea. Older animals will have similar clinical signs. Conjunctivitis may be seen in acute cases. In severe cases, young animals become prostrate, have laboured respiration, and may die. Most cases are of moderate severity, with recovery taking one to two months.103, 147 The acute pulmonary lesions are characterized by multifocal greyish-purple consolidated areas, usually located in the region of the hilus and the ventral portions of the cranial and middle lobes (Figure 4). Serofibrinous pleuritis may accompany these lesions. In severe cases foci of consolidation extend throughout the lungs. The microscopical lesions in acute cases are characterized by serofibrinous pneumonia and pleuritis, and small haemorrhages are frequently present in the parenchyma of the lungs.
As the lesions progress, moderate numbers of mononuclear cells, particularly lymphocytes, are found in the alveoli and interstitial tissues. Perivascular and peribronchiolar lymphoid hyperplasia, in which scattered plasma cells, macrophages, and a few neutrophils occur, are also evident.103
Chronic pulmonary lesions consist of small greyish-pink areas of pneumonia, depressed slightly below the surface of the pleura. Histopathologically these areas are characterized by atelectasis, infiltration of variable numbers of mononuclear cells and neutrophils, and severe peribronchiolar lymphoid hyperplasia.103, 147
Microscopically, chlamydiae are found in monocytes and in the cytoplasm of epithelial cells, mainly of the bronchioli and alveoli. Organisms are frequently difficult to detect, particularly in chronic lesions.103
Pigs
Disease syndromes in pigs that are associated with chlamydial infections are conjunctivitis, pneumonia, enteritis, reproductive problems, pericarditis, polyarthritis and encephalomyelitis.5, 155 A number of distinct strains have been identified, including those that belong to C. suis, C. pecorum and C. abortus.46, 68, 69, 129, 130 Of these, C. suis appears to be the most common. Chlamydia suis strains have been isolated from the conjunctiva and respiratory and intestinal tracts of pigs, where they were associated with conjunctivitis, pneumonia and enteritis.95, 117 Recent genome analysis of 10 US and 29 Swiss strains revealed very high genetic diversity associated with events of recombination.65, 134 Chlamydia suis is the only chlamydial species for which structural elements encoding antimicrobial (tetracycline) resistance have been found39 The tetracycline-resistant gene has been only retrieved from porcine, not from human C. suis isolates.
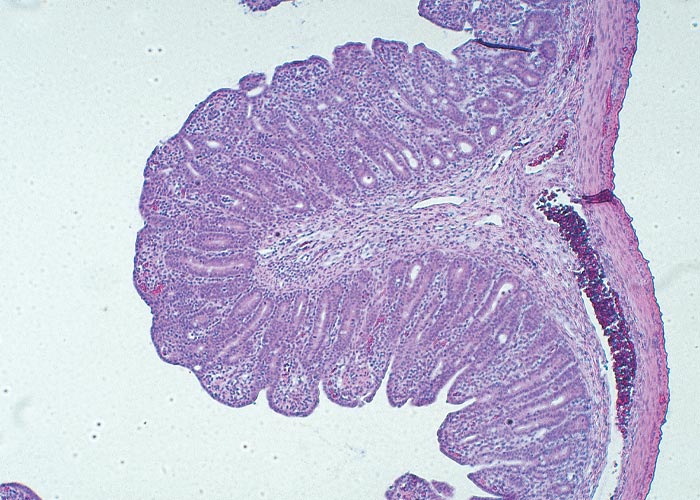
Many C. suis infections are thought to be mild or asymptomatic. Some studies have indicated that C. suis is not a significant pathogen, as it can be isolated from normal and diarrhoeic pigs at the same rate.95, 152 On the other hand, C. suis was the only potential pathogen isolated from four 30-day-old piglets suffering from diarrhoea.94 In another trial, inoculation of gnotobiotic piglets with C. suis resulted in diarrhoea and extensive villous atrophy of the ileum and distal jejunum.114 (Figures 5 and 6). Inoculation of 21-day-old piglets raised conventionally with the same isolates produced only moderate villous atrophy in the absence of clinical diarrhoea.116 The role that colostral antibodies to C. suis may play in moderating intestinal disease in animals of that age is unknown, and it is not clear what synergistic effect chlamydial infection may have with other pathogens. Recent studies with Salmonella typhimurium and Eimeria scaba have shown that the diseases produced by those organisms were more severe when chlamydia was present.72, 106
The same C. suis strains have been isolated from pigs suffering from pneumonia and conjunctivitis, although it is difficult to establish their significance as pathogens because they have also been isolated from clinically normal pigs. When a C. suis isolate from nursery pigs with pneumonia was inoculated experimentally into gnotobiotic piglets, several of them became moribund or severely dyspnoeic, although the majority exhibited only mild dyspnoea.118 The gross lung lesions at 17 to 21 days after infection were typical of bronchopneumonia.152, 177
Similarly, C. suis strains have been isolated both from the eyes of clinically normal pigs and from those of pigs suffering from conjunctivitis.33, 115, 117 In severe cases, lymphofollicular hyperplasia in the palpebral conjunctiva is often present (Figure 7).
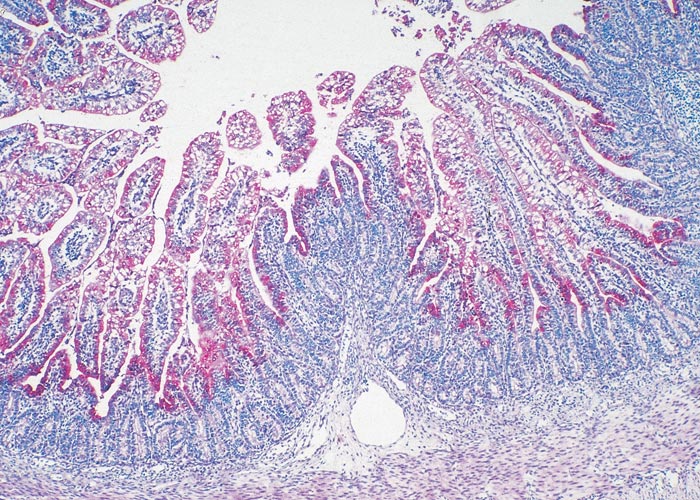
Figure 5 Chlamydial antigen in a villus of the ileum (four days post-infection) of a gnotobiotic piglet infected with C. suis. The chlamydiae are stained red (ABC-alkaline phosphatase stain)
When an isolate obtained from the eye was inoculated experimentally into the conjunctiva of normal pigs, they did not develop clinical signs, although histopathological examination revealed mild conjunctivitis. The mild conjunctivitis may have been the result of a first infection, as is the case in humans in whom it is thought that repeated or chronic infections are required for severe cases to develop. The piglets inoculated in the conjunctiva or trachea developed diarrhoea with villous atrophy similar to that seen in the pigs that developed diarrhoea after being inoculated orally.117, 118
The epidemiology of C. suis strains is of interest because they are ubiquitous in pig populations. Serological and isolation studies indicate that most pigs become infected before six weeks of age, and that virtually all pigs are infected by one or more strains.135 Infection with one strain may not protect against another. Many infections appear to be persistent, permitting the chlamydiae to be passed on from the sow to her young piglets following farrowing. In the USA, many of the C. suis strains are resistant to sulphonamides and tetracyclines, making treatment or control of the disease syndromes very difficult.6 Tetracycline-resistant C. suis strains have been detected in clinical samples of subclinically, infected pigs in European countries such as Italy, Germany, Belgium, The Netherlands and Switzerland.16, 37, 128, 162 Chlamydia suis may potentially be zoonotic based on its detection in Nepalese trachoma patients36 and samples taken from Belgian farmers and slaughterhouse workers.34, 35
Other disease syndromes that have been associated with chlamydiae in pigs include pericarditis, encephalomyelitis, polyarthritis and reproductive problems. These are caused by C. pecorum strains.67, 69
Chlamydia abortus, of the same strain as that isolated from sheep and cattle, was isolated from a case of abortion in a sow,2 and has been detected in retrospective studies in other cases of abortion.129, 130
Horses
Reports of chlamydiosis in horses are limited. However, there is sufficient evidence to indicate that chlamydia should be considered as differential diagnosis in abortion cases and respiratory disease. Because many of the isolations of chlamydiae made from horses were obtained before strains could be identified readily, it is not known whether more than one species or strain of Chlamydia was involved.
During the 1972 to 1973 epidemic of chlamydial abortion in sheep in South Africa, Chlamydia was incriminated as the cause of abortion in mares and of keratoconjunctivitis in foals.103 Abortions in mares due to chlamydial infections have also been reported in Spain81 and Germany.11 Chlamydia abortus or C. psittaci are able to cause abortion in pregnant mares.15 Outbreaks of C. psittaci-induced abortions in mares were recently reported in New South Wales, Australia.17, 23 They were associated with human psittacosis cases in veterinary students, veterinarians and stud staff after exposure to infected foetal membranes. Molecular typing revealed involvement of a highly virulent C. psittaci of the 6BC lineage (genotype A) identical to lineages detected in Australian parrots, but a second case was related to a C. psittaci strain detected in a dove (genotype B) typically associated with infections in pigeons, globally.60, 61
An upper respiratory tract/pneumonia syndrome may be another important chlamydial disease of horses.18, 87, 89, 92 An isolate (N16) from horses was recently characterized as belonging to Chlamydia pneumoniae.145 Molecularly, there are enough differences between this isolate and the human strain of C. pneumoniae to make it unlikely that transmission of C. pneumoniae occurs between horses and humans. Isolations have been made from normal horses, and from those with serous nasal discharges, as well as from the lungs of a mare suffering from pneumonia.18, 87, 89, 92, 173 Most of the equine strains that have been isolated are difficult to grow in cell cultures and embryonated eggs. Attempts to infect other horses with these strains resulted only in mild or subclinical disease.87, 89 This is also characteristic of the C. pneumoniae strains occurring naturally in humans and koalas.
Diagnosis
A definitive diagnosis of chlamydiosis requires demonstration of the chlamydial agent from infected tissues by molecular or antigen detection methods, together with typical clinical signs and/or pathological findings. A retrospective diagnosis can also be made serologically if the aetiological agent cannot be demonstrated. Serum collected after disease (e.g. abortion) will reveal an elevated antibody titre resulting from a current or previous infection.
Nucleic acid amplification tests are considered the method of choice for the diagnosis of chlamydial infections in animals because of their high sensitivity and specificity. Several species-specific protocols for conventional or real-time PCR are published in the literature or commercially available.45, 46, 122 Broad-range PCR methods targeting conserved regions, such as the 16S-23S rRNA specific for the family Chlamydiaceae or order Chlamydiales, are often used as an initial screening method to be followed by chlamydial species determination, using a species-specific PCR,98 by sequencing of the PCR product or by Arraymate Microarray.14 PCR assays in combination with restriction fragment length polymorphism (RFLP) or high resolution melting (HRM) analysis have been developed that can differentiate naturally infected from vaccinated animals (DIVA).74, 164, 172
Direct staining of chlamydiae or chlamydial inclusions in tissues or smears of exudate, or in cell culture cell monolayers, can be performed using histochemical or immunohistochemical techniques. Some of the more commonly used stains are Giemsa, Giménez, modified Ziehl-Neelsen and Macchiavello.3, 6, 103 Tissue specimens or smears can be examined by immunofluoresence and immunohistochemistry, using antibodies to LPS or surface antigens such as MOMP (Major Outer Membrane Protein). However, such antibodies are often not species-specific. Stained smears might be useful as an initial screening test, but confirmation by molecular methods is highly recommended due to the inferior sensitivity of staining and the lack of species specificity.
Historically, isolation in embryonated chicken eggs and later in cell culture was considered the gold standard but this is considered time consuming and expensive, and requires specialist expertise. Most isolates will grow in a range of commonly used cell lines, such as Vero, McCoy, Buffalo Green Monkey (BGM) or baby hamster kidney (BHK)3, 6 Centrifugation (at 1000–2000 x g for 60 minutes at close to 37 °C) of the inoculum onto the cell monolayer can increase infection efficiency for some species.3, 6
Blind passaging at four to six days increases efficiency of isolation. It is important to examine cultures for inclusions three to six days after infection of cell cultures. It should be borne in mind that chlamydiae are relatively sensitive to freezing; the common practice of freeze-thaw to disrupt the cell monolayers must be avoided. A diluent consisting of sucrose-phosphate-glutamate has proved to be satisfactory as a transport medium for chlamydial field samples3, 6, 142 and it can also be used as a diluent when freezing cultures for storage. Antibiotics may be added to it to reduce possible contamination.3, 6
In the case of abortion, the placental membranes are the specimens of choice for diagnosing C. abortus infection as these contain very large numbers of organisms.13, 82, 122 When the placenta is not available, vaginal swabs or swabs from the moist coat of the aborted foetus or from the abomasal content of the aborted or stillborn lamb should be used.82, 125, 140 In general, such preparations contain fewer organisms than placental smears. In other chlamydial diseases, swabs from the infected tissues (e.g. conjunctiva) or exudates, faecal swabs, or samples of infected tissues can be used and processed for DNA extraction prior to investigation by molecular methods.
Serology as a diagnostic test for chlamydiosis is of limited value when rapid diagnosis and treatment are needed. The complement fixation test (CFT) has traditionally been the most widely used procedure for detecting ovine enzootic abortion. However, antigenic cross-reactivity between C. abortus and C. pecorum, which is endemic in small ruminants, as well as with some Gram-negative bacteria (e.g. Acinetobacter), can give rise to low false-positive CFT results. Furthermore, CFT has been shown to be less sensitive than alternative tests. Therefore, CFT is no longer recommended as the method of choice for serological diagnosis of ovine enzootic abortion.
Several enzyme-linked immunosorbent assay (ELISA) kits are commercially available for diagnosing C. abortus in sheep, goats and cattle. However, none of these tests have been properly evaluated for use with cattle sera due to the lack of defined reference sera for determining sensitivity and specificity. Specific detection of anti-C. abortus antibodies in sheep and goat sera can be accomplished by the use of ELISAs based on synthetic peptides of MOMP, recombinant MOMP123 and recombinant polymorphic membrane protein Pmp12G (aka POMP90) in indirect ELISAs.83, 175 Most recently, a new indirect ELISA based on Pmp12G has been commercialized and shown to be both sensitive and specific for C. abortus, in particular in differentiating animals infected with C. abortus from those infected with C. pecorum.41, 175
Differential diagnosis
Many of the clinical signs and lesions caused by chlamydiae are also caused by other bacterial and viral agents. None of the lesions or signs are pathognomonic, and therefore laboratory confirmation is almost always needed. In the case of abortion, the other organisms most commonly encountered are Brucella spp. and Coxiella burnetii. In smears or sections of infected tissues, it may be difficult for inexperienced personnel to differentiate between Brucella, Chlamydia and Coxiella morphologically, making further typing methods advisable.13, 122
Control
Chlamydiae are ubiquitous in the livestock population, which makes vaccination and antibiotic therapy the only effective means of control. Currently vaccines are only available for the ovine abortion strain and the feline pneumonitis strain. Both attenuated live and inactivated vaccines are available depending on the country concerned. Only the vaccines produced for the control of ovine chlamydial abortion, for which there is most information, are discussed below.
Inactivated vaccines against ovine abortion due to chlamydial infection are usually formalin-inactivated and contain an oil-emulsion adjuvant.103, 111 To be effective they require large amounts of antigen to be incorporated into the vaccine. Most manufacturers use the level of complement fixing antibodies induced in the sera of rabbits as the standard for judging potency. These antibodies are, however, not strain-specific, and are also not considered protective. Because the strain specificity and antigen incorporated into vaccines are often not controlled, variation in efficacy of this type of vaccine is often a problem. Although most of these vaccines do reduce the abortion rate, they do not prevent infection or shedding.103, 111 Annual vaccination of ewes is recommended for these inactivated vaccines.
A vaccine based on a live attenuated strain of C. abortus was developed in the 1980s and first became commercially available in the mid-1990s. When administered to susceptible ewes or goats before mating, it provides protection against abortion for at least three years and reduces post-parturition shedding of chlamydiae.110, 112, 113 It can be administered simultaneously with Brucella Rev 1 and the live Salmonella Rv 6 vaccines.139 When administered with the live Toxoplasma vaccine, it must be given at a different site.22 The C. abortus 1B strain is a temperature-sensitive mutant of the C. abortus strain AB7 and is designated 1B.109, 110 It has been reported to have reduced infectivity and ability to induce death in utero in ewes and mice.109, 112 The vaccine relies on the fact that the growth of the 1B strain is restricted at 39.5°C, which is the body temperature of sheep, thus apparently not able to replicate in the host animal and cause disease. However, following the development of molecular methods for differentiating the vaccine-type strain from wild-type strains74, 164, 172 it became apparent that strains present in ovine enzootic abortion cases in vaccinated sheep flocks also cluster phylogenetically very tightly with the vaccine strain, suggesting that this strain can in fact replicate in sheep and cause disease in some vaccinated animals.73, 78, 85, 126, 172 Genome sequencing of the vaccine strain 1B, its parent strain AB7 and another temperature-sensitive mutant strain (1H), produced at the same time as the 1B strain, but which reverted back to virulence, revealed that 1B has only ten single nucleotide polymorphisms (SNPs) compared to AB7 and that these SNPs are identical to those found in strain 1H. This suggests that there is no genetic basis for a temperature-sensitive phenotype and that protection elicited may result for other reasons.85 Indeed, a study in 2013 demonstrated that when a high dose, such as that given with the commercial vaccines, of a highly virulent C. abortus strain is administered to sheep it does not cause disease, suggesting that it may instead be stimulating protective immunity, which could explain these observations concerning the live vaccines.84 Most recently, evidence has been provided that the vaccine strain can be shed and infect other naïve animals, causing disease.73, 85
After chlamydial abortion, ewes should immediately be isolated. All dead foetuses, placental membranes, and bedding should be carefully disposed of, and lambing pens must be cleaned and disinfected to limit the spread of infection to naïve animals as well as the possibility of zoonotic transmission. Basic hygiene procedures, including thoroughly washing of hands and the use of disposable gloves are essential when handling infected animals or material.82
Some antibiotics are effective both prophylactically and therapeutically in controlling chlamydiosis in most animals, but they must be administered early in the course of the infection, as soon as possible after Day 95-100 of gestation. Tetracyclines are the most widely used antibiotics because of their relatively low cost and ease of administration. They have been used prophylactically with success in ewes under field conditions to prevent abortions. Although recommended to be administered as a single dose, to minimise the possible development of resistance, further doses should ideally be given fortnightly until lambing, to suppress multiplication of the pathogen in the placenta and thus limiting any further damage and possible death of the foetus.1, 41, 82, 103 Chlortetracycline has been used in feeder lambs to control polyarthritis. Tylosin and long-acting oxytetracycline have been used parenterally103 Most of these antimicrobials should be effective in controlling other chlamydial disease syndromes in cattle, sheep, goats and horses, but their use in controlling syndromes in pigs can constitute a problem as many of the current porcine isolates are resistant to tetracycline and sulphadiazine.6
References
- AITKEN, I. D. & LONGBOTTOM, D., 2007. Chlamydial abortion. Diseases of Sheep, 4th edn, Chapter 16, Blackwell Publishing, 105-112.
- ANDERSEN, A. A., 1999. Unpublished data.
- ANDERSEN, A. A., 2000. Avian chlamydiosis. In: BLANCOU, J., (ed.). OIE Manual of Standards for Diagnostic Tests and Vaccines. 4th edn. Paris: Office International des Epizooties. In Press.
- ANDERSEN, A. A., GRIMES, J. E. & WYRICK, P., 1997. Chlamydiosis (psittacosis, ornithosis). In: CALNEK, B. W., (ed.). Diseases of Poultry. 10th edn. Ames, IA: Iowa State University Press.
- ANDERSEN, A. A. & ROGERS, D. G., 1996. Are chlamydiae swine pathogens? Swine Health and Production, 4, 286–288.
- ANDERSEN, A. A. & ROGERS, D. G., 1998. Resistance to tetracycline and sulfadiazine in swine C. trachomatis isolates. In: STEPHENS, R. S., BYRNE, G. I., CHRISTIANSEN, G., CLARKE, I. N., GRAYSTON, J. T., RANK, R. G., RIDGWAY, G. L., SAIKKU, P., SCHACHTER, J. & STAMM, W. E., (eds). Chlamydial Infections: Proceedings of the Ninth International Symposium on Human Chlamydial Infection, Napa, California, USA, June 21–26. San Francisco, CA: International Chlamydia Symposium, 313–316.
- APPLEYARD, W. T., AITKEN, I. D. & ANDERSON, I. E., 1985. Attempted venereal transmission of Chlamydia psittaci in sheep. Veterinary Record, 116, 535–538.
- BEDSON, S. P. & BLAND, J. O. W., 1932. A morphological study of psittacosis virus, with the description of a developmental cycle. British Journal of Experimental Pathology, 13, 461-466.
- BEDSON, S. P. & WESTERN, G. T., 1930. Aetiology experimental observations. In: A Disease of Parrots Communicable to Man (Psittacosis), STURDEE, E. L. & SCOTT, W. M., (eds.). His Majesty's Stationery Office, London, 59-95.
- BEDSON, S. P., WESTERN, G. T. & SIMPSON, S. L., 1930. Observations on the aetiology of psittacosis. Lancet, 1, 235-236.
- BOCKLISCH, V. H., LUDWIG, C. & LANGE, S., 1991. Chlamydien als Abortursache beim Pferd. Berliner und Münchener Tierärztliche Wochenschrift, 104, 119–124.
- BODETTI, T. J., JACOBSON, E., WAN, C., HAFNER, L., POSPISCHIL, A., ROSE, K. & TIMMS, P., 2002. Molecular evidence to support the expansion of the host range of Chlamydophila pneumoniae to include reptiles as well as humans, horses, koalas and amphibians. Systematic and Applied Microbiology, 25, 146-152.
- BOREL, N., FREY, C. F., GOTTSTEIN, B., HILBE, M., POSPISCHIL, A., FRANZOSO, F. D. & WALDVOGEL, A., 2014. Laboratory diagnosis of ruminant abortion in Europe. Veterinary Journal, 200, 218-229.
- BOREL, N., KEMPF, E., HOTZEL, H., SCHUBERT, E., TORGERSON, P., SLICKERS, P., EHRICHT, R., TASARA, T., POSPISCHIL, A. & SACHSE, K., 2008. Direct identification of Chlamydiae from clinical samples using a DNA microarray assay: A validation study. Molecular Cellular Probes, 22, 55-64.
- BOREL, N., POLKINGHORNE, A. & POSPISCHIL, A., 2018. A review on chlamydial diseases in animals: Still a challenge for pathologists? Veterinary Pathology, 55, 374-390.
- BOREL, N., REGENSCHEIT, N., DI FRANCESCO, A., DONATI, M., MARKOV, J., MASSEREY, Y. & POSPISCHIL, A., 2012. Selection for tetracycline-resistant Chlamydia suis in treated pigs. Veterinary Microbiology,156, 143-146.
- BRANLEY, J., BACHMANN, N. L., JELOCNIK, M., MYERS, G. S. & POLKINGHORNE, A., 2016. Australian human and parrot Chlamydia psittaci strains cluster within the highly virulent 6BC clade of this important zoonotic pathogen. Scientific Reports, 6, 30019.
- BURRELL, M. H., CHALMERS, W. S. K. & KEWLEY, D. R., 1986. Isolation of Chlamydia psittaci from the respiratory tract and conjunctivae of thoroughbred horses. Veterinary Record, 119, 302–303.
- BUXTON, D., 1986. Potential danger to pregnant women of Chlamydia psittaci from sheep. Veterinary Record, 118, 510–511.
- BUXTON, D., ANDERSON, I. E., LONGBOTTOM, D., LIVINGSTONE, M., WATTEGEDERA, S. & ENTRICAN, G., 2002. Ovine chlamydial abortion: characterization of the inflammatory immune response in placental tissues. Journal of Comparative Pathology, 127, 133-141.
- BUXTON, D., BARLOW, R. M., FINLAYSON, J., ANDERSON, I. E. & MACKELLER, A., 1990. Observations on the pathogenesis of Chlamydia psittaci infection of pregnant sheep. Journal of Comparative Pathology, 102, 221–237.
- CHALMERS, W. S. K., SIMPSON, J., LEE, S. J. & BAXENDALE, W., 1997. Use of a live chlamydial vaccine to prevent ovine enzootic abortion. Veterinary Record, 141, 63–67.
- CHAN, J., DOYLE, B., BRANLEY, J., SHEPPEARD, V., GABOR, M., VINEY, K., QUINN, H., JANOVER, O., MCCREADY, M. & HELLER, J., 2017. An outbreak of psittacosis at a veterinary school demonstrating a novel source of infection. One Health, 3, 29-33.
- COLES, A. C., 1930. Micro-organisms in psittacosis. Lancet, 1, 1011-1012.
- COLES, J. D. W., 1940. Psittacosis in domestic pigeons. Onderstepoort Journal of Veterinary Science and Animal Industry, 15, 141–148.
- COX, H. U., HOYT, P. G., POSTON, R. P., SNIDER, T. G. R., LEMARCHAND, T. X. & O’REILLY, K. L., 1998. Isolation of an avian serovar of Chlamydia psittaci from a case of bovine abortion. Journal of Veterinary Diagnostic Investigations, 10, 280–282.
- CROSSE, B. A., GOMES, P. & MUERS, M. M., 1991. Ovine psittacosis and sarcoidosis in a pregnant woman. Thorax, 46, 604–606.
- CUTLIP, R. C., 1974. Ultrastructure of the synovial membrane of lambs affected with chlamydial polyarthritis. American Journal of Veterinary Research, 35, 171–176.
- CUTLIP, R. C. & RAMSEY, F. K., 1973. Sequential development of articular lesions in lambs after intra-articular exposure. American Journal of Veterinary Research, 34, 71–75.
- CUTLIP, R. C., SMITH, P. C. & PAGE, L. A., 1972. Chlamydial polyarthritis of lambs: A review. Journal of the American Veterinary Medical Association, 161, 1213–1216.
- DANIEL, R. G., HOLLIMAN, A., DAVID, G. P., KIRBY, F. D., SIMPSON, V. R., CRANDWELL, M. P., DAWSON, M., GRIFFITHS, P. C. & BEVAN, B. J., 1993. Bovine chlamydiosis in the United Kingdom. Veterinary Record, 133, 351–352.
- DANNATT, L., DANIEL, R. G., GRIFFITHS, P. C. & DAWSON, M., 1998. Investigation of a possible role for chlamydia in a new disease syndrome in dairy cattle. Veterinary Record, 143, 691–693.
- DAVIDSON, H. J., ROGERS, D. P., YEARY, T. J., STONE, G. G., SCHONEWEIS, D. A. & CHENGAPPA, M. M., 1994. Conjunctival microbial flora of clinically normal pigs. American Journal of Veterinary Research, 55, 949–951.
- DE PUYSSELEYR, K., DE PUYSSELEYR, L., DHONDT, H., GEENS, T., BRAECKMAN, L., MORRE, S. A., COX, E. & VANROMPAY, D., 2014. Evaluation of the presence and zoonotic transmission of Chlamydia suis in a pig slaughterhouse. BMC Infectious Diseases, 14, 560.
- DE PUYSSELEYR, L., DE PUYSSELEYR, K., BRAECKMAN, L., MORRE, S. A., COX, E. & VANROMPAY, D., 2017. Assessment of Chlamydia suis Infection in Pig Farmers. Transboundary and Emerging Diseases, 64, 826-833.
- DEAN, D., ROTHSCHILD, J., RUETTGER, A., KANDEL, R. P. & SACHSE, K., 2013. Zoonotic Chlamydiaceae species associated with trachoma, Nepal. Emerging Infectious Diseases, 19, 1948-1955.
- DI FRANCESCO, A., DONATI, M., ROSSI, M., PIGNANELLI, S., SHURDHI, A., BALDELLI, R. & CEVENINI, R., 2008. Tetracycline-resistant Chlamydia suis isolates in Italy. Veterinary Record,163, 251-252.
- DILBECK, P. M., EVERMANN, J. F., CRAWFORD, T. B., WARD, A. C., LEATHERS, C. W., HOLLAND, C. J., MEBUS, C. A., LOGAN, L. L., RURANGIRWA, F. R. & MCGUIRE, T. C., 1990. Isolation of a previously undescribed rickettsia from an aborted bovine fetus. Journal of Clinical Microbiology, 28, 814–816.
- DUGAN, J., ROCKEY, D. D., JONES, L. & ANDERSEN, A. A., 2004. Tetracycline resistance in Chlamydia suis mediated by genomic islands inserted into the chlamydial inv-like gene. Antimicrobial Agents and Chemotherapy, 48, 3989-3995.
- EHRET, W. J., SCHUTTE, A. P., PIENAAR, J. G. & HENTON, M. M., 1975. Chlamydiosis in a beef herd. Journal of the South African Veterinary Medical Association, 46, 171–179.
- ESSIG, A. & LONGBOTTOM, D., 2015. Chlamydia abortus: New aspects of infectious abortion in sheep and potential risk for pregnant women. Current Clinical Microbiology Reports, 2, 22–34.
- EUGSTER, A. K., 1980. Chlamydiosis. In: STEELE, J. H., (ed.). CRC Handbook Series in Zoonoses. Section A: Bacterial, Rickettsial, and Mycotic Diseases. Florida: CRC Press, Inc, 2.
- EUGSTER, A. K. & STORZ, J., 1971. Effect of colostral antibodies on the pathogenesis of intestinal chlamydial infections in calves. American Journal of Veterinary Research, 32, 711–718.
- EUGSTER, A. K. & STORZ, J., 1971. Pathogenetic events in intestinal chlamydial infections leading to polyarthritis in calves. Journal of Infectious Diseases, 123, 41–50.
- EVERETT, K. D. E. & ANDERSEN, A. A., 1999. Identification of nine species of the Chlamydiaceae using PCR-RFLP. International Journal of Systematic Bacteriology, 49, 803–813.
- EVERETT, K. D. E., BUSH, R. M. & ANDERSEN, A. A., 1999. Emended description of the order Chlamydiales, proposal of Parachlamydiaceae fam. Nov. and Simkaniaceae fam. Nov., each containing one monotypic genus, revised taxonomy of the family Chlamydiaceae, including a new genus and five new species, and standards for the identification of organisms. International Journal of Systematic Bacteriology, 49, 415–440.
- FUKUSHI, H. & HIRAI, K., 1992. Proposal of Chlamydia pecorum sp. nov. for Chlamydia strains derived from ruminants. International Journal of Systematic Bacteriology, 42, 306–308.
- GIRJES, A. A., CARRICK, F. N. & LAVIN, M. F., 1994. Remarkable sequence relatedness in the DNA encoding the major outer membrane protein of Chlamydia psittaci (koala type I) and Chlamydia pneumoniae. Gene, 138, 139–142.
- GRAYSTON, J. T., KUO, C. C., CAMPBELL, L. A. & WANG, S. P., 1989. Chlamydia pneumoniae sp. nov. for Chlamydia sp. strain TWAR. International Journal of Systematic Bacteriology, 39, 88–90.
- GREIG, J. R., 1936. Enzootic abortion in ewes: a preliminary note. Veterinary Record, 48, 1225-1227.
- GRIFFITHS, P. C., PLATER, J. M., MARTIN, T. C., HUGHES, S. L., HUGHES, K. J., HEWINSON, R. G. & DAWSON, M., 1995. Epizootic bovine abortion in a dairy herd: characterization of a Chlamydia psittaci isolate and antibody response. British Veterinary Journal, 151, 683–693.
- GUTIERREZ, J., WILLIAMS, E. J., O'DONOVAN, J., BRADY, C., PROCTOR, A. F., MARQUES, P. X., WORRALL, S., NALLY, J. E., MCELROY, M., BASSETT, H. F., SAMMIN, D. J. & MARKEY, B. K., 2011. Monitoring clinical outcomes, pathological changes and shedding of Chlamydophila abortus following experimental challenge of periparturient ewes utilizing the natural route of infection. Veterinary Microbiology, 147, 119-126.
- HALBERSTAEDTER, L. & VON PROWAZEK, S., 1907a. Über Zelleinschlüsse parasitärer Natur beim Trachom. Arbeiten aus dem Kaiserlichen Gesundheitsamte, 26, 44-47.
- HALBERSTAEDTER, L. & VON PROWAZEK, S., 1907b. Zur Aetiologie des Trachoms. Deutsche Medizinische Wochenschrift, 33, 1285-1287.
- HARSHFIELD, G. S., 1970. Sporadic bovine encephalomyelitis. Journal of the American Veterinary Medical Association, 156, 466–477.
- HOLLIMAN, A., DANIEL, R. G., PARR, J. G., GRIFFITHS, P. C., BEVAN, B. J., MARTIN, T. C., HEWINSON, R. G., DAWSON, M. & MUNRO, R., 1994. Chlamydiosis and abortion in a dairy herd. Veterinary Record, 134, 500–502.
- HOPKINS, J. B., STEPHENSON, E. H., STORZ, J. & PIERSON, R. E., 1973. Conjunctivitis associated with chlamydial polyarthritis in lambs. Journal of the American Veterinary Medical Association, 163, 1157–1160.
- HORN, M., 2011. Chlamydiae. In: KRIEG, N. R., LUDWIG, W., WHITMAN, W. B., HEDLUND, B. P., PASTER, B. J., STALEY, J. T., WARD, N. & BROWN, D., (ed.). Bergey`s Manual of Systematic Bacteriology. Springer, New York, 4, 843-877.
- JACKSON, M., WHITE, N., GIFFARD, P. & TIMMS, P., 1999. Epizootiology of chlamydia infections in two free-range koala populations. Veterinary Microbiology, 65, 255–264.
- JELOCNIK, M., BRANLEY, J., HELLER, J., RAIDAL, S., ALDERSON, S., GALEA, F., GABOR, M. & POLKINGHORNE, A., 2017. Multilocus sequence typing identifies an avian-like Chlamydia psittaci strain involved in equine placentitis and associated with subsequent human psittacosis. Emerging Microbes and Infections, 6(2), e7. DOI: 10.1038/emi.2016.135.
- JELOCNIK, M., JENKINS, C., O'ROURKE, B., BARNWELL, J. & POLKINGHORNE, A., 2018. Molecular evidence to suggest pigeon-type Chlamydia psittaci in association with an equine foal loss. Transboundary Emerging Diseases, 65(3), 911-915. DOI: 10.1111/tbed.12817.
- JONES, G. E. & ANDERSON, I. E., 1988. Chlamydia psittaci: is tonsillar tissue the portal of entry in ovine enzootic abortion? Research in Veterinary Science, 44, 260-261.
- JONES, H., RAKE, G. & STEARNS, B., 1945. Studies on lymphogranuloma venereum. III. The action of the sulfonamides on the agent of lymphogranuloma venereum. Journal of Infectious Diseases, 76, 55-69.
- JORGENSEN, D. M., 1997. Gestational psittacosis in a Montana sheep rancher. Emerging Infectious Diseases, 3, 191–194.
- JOSEPH, S. J., MARTI, H., DIDELOT, X., READ, T. D. & DEAN, D., 2016. Tetracycline selective pressure and homologous recombination shape the evolution of Chlamydia suis: A recently identified zoonotic pathogen. Genome Biology and Evolution,8, 2613-2623.
- KALETA, E. F. & TADAY, E. M., 2003. Avian host range of Chlamydophila spp. based on isolation, antigen detection and serology. Avian Pathology, 32, 435-461.
- KALTENBOECK, B., KOUSOULAS, K. G. & STORZ, J., 1992. Two-step polymerase chain reactions and restriction endonuclease analyses detect and differentiate ompA DNA of Chlamydia spp. Journal of Clinical Microbiology, 30, 1098–1104.
- KALTENBOECK, B., SCHMEER, N. & SCHNEIDER, R., 1997. Evidence for numerous omp1 alleles of porcine Chlamydia trachomatis and novel chlamydial species obtained by PCR. Journal of Clinical Microbiology, 35, 1835–1841.
- KALTENBOECK, B. & STORZ, J., 1992. Biological properties and genetic analysis of the ompA locus in chlamydiae isolated from swine. American Journal of Veterinary Research, 53, 1482–1487.
- KENNEDY, P. C., OLANDER, H. J. & HOWARTH, J. A., 1960. Pathology of epizootic bovine abortion. Cornell Veterinarian, 50, 417–429.
- KOCAN, K. M., CRAWFORD, T. B., DILBECK, P. M., EVERMANN, J. F. & MCGUIRE, T. C., 1990. Development of a rickettsia isolated from an aborted bovine fetus. Journal of Bacteriology, 172, 5949–5955.
- KOUDELA, B., VIVOVEC, J. & STERBA, J., 1990. Concurrent infection of enterocytes with Eimeria scabra and other enteropathogens in swine. Veterinary Parasitology, 35, 71–77.
- LAROUCAU, K., AAZIZ, R., VORIMORE, F., MENARD, M. F., LONGBOTTOM, D. & DENIS, G., 2018. Abortion storm involving the live vaccine C. abortus 1B strain in a vaccinated sheep flock. Veterinary Microbiology, 225, 31-33.
- LAROUCAU, K., VORIMORE, F., SACHSE, K., VRETOU, E., SIARKOU, V. I., WILLEMS, H., MAGNINO, S., RODOLAKIS, A. & BAVOIL, P. M., 2010. Differential identification of Chlamydophila abortus live vaccine strain 1B and C. abortus field isolates by PCR-RFLP. Vaccine, 28, 5653–5656.
- LEAVER, H. A., HOWIE, A., AITKEN, I. D., APPLEYARD, B. W., ANDERSON, I. E., JONES, G., HAY, L. A., WILLIAMS, G. E. & BUXTON, D., 1989. Changes in progesterone, oestradiol 17Β, and intrauterine prostaglandin E2 during late gestation in sheep experimentally infected with an ovine abortion strain of Chlamydia psittaci. Journal of General Microbiology, 135, 565–573.
- LEVINTHAL, W., 1930. Die Ätiologie der Psittakosis. Klinische Wochenschrift, 9, 654.
- LILLIE, R. D., 1930. Psittacosis: Rickettsia-like inclusions in man and in experimental animals. Public Health Report, 45, 773-778.
- LIVINGSTONE, M., AITCHISON, K. & LONGBOTTOM, D., 2014. Abortion in flocks vaccinated against enzootic abortion. Veterinary Record, 174, 613-614.
- LIVINGSTONE, M., WHEELHOUSE, N., ENSOR, H., ROCCHI, M., MALEY, S., AITCHISON, K., WATTEGEDERA, S., WILSON, K., SAIT, M., SIARKOU, V., VRETOU, E., ENTRICAN, G., DAGLEISH, M. & LONGBOTTOM, D., 2017. Pathogenic outcome following experimental infection of sheep with Chlamydia abortus variant strains LLG and POS. PLoS ONE, 12, e0177653.
- LIVINGSTONE, M., WHEELHOUSE, N., MALEY, S. W. & LONGBOTTOM, D., 2009. Molecular detection of Chlamydophila abortus in post-abortion sheep at oestrus and subsequent lambing. Veterinary Microbiology, 135, 134-141.
- LOIZELIER, A. B., MARCOTEGUI, M. A., DELGADO, I., DE FRUTOS, I. & ESCRIBANO, M. A., 1979. Chlamydiosis equina. Anales del Instituto Nacional de Investigaciones Agrarias, 4, 11–32.
- LONGBOTTOM, D. & COULTER, L. J., 2003. Animal chlamydioses and zoonotic implications. Journal of Comparative Pathology,128, 217-244.
- LONGBOTTOM, D., FAIRLEY, S., CHAPMAN, S., PSARROU, E., VRETOU, E. & LIVINGSTONE, M., 2002. Serological diagnosis of ovine enzootic abortion by enzyme-linked immunosorbent assay with a recombinant protein fragment of the polymorphic outer membrane protein POMP90 of Chlamydophila abortus. Journal of Clinical Microbiology, 40, 4235–4243.
- LONGBOTTOM, D., LIVINGSTONE, M., MALEY, S., VAN DER ZON, A., ROCCHI, M., WILSON, K., WHEELHOUSE, N., DAGLEISH, M., AITCHISON, K., WATTEGEDERA, S., NATH, M., ENTRICAN, G. & BUXTON, D., 2013. Intranasal infection with Chlamydia abortus induces dose-dependent latency and abortion in sheep. PLoS One, 8, e57950.
- LONGBOTTOM, D., SAIT, M. L., LIVINGSTONE, M., LAROUCAU, K., SACHSE, K., HARRIS, S. R., THOMSON, N. R. & SETH-SMITH, H. M. B., 2018. Genomic evidence that the live Chlamydia abortus vaccine strain 1B is not attenuated and has the potential to cause disease. Vaccine, 36, 3593-3598.
- LOZANO, E. A., 1986. Etiologic significance of bacterial isolates from rams with palpable epididymitis. American Journal of Veterinary Research, 47, 1153–1156.
- MAIR, T. S. & WILLS, J. M., 1992. Chlamydia psittaci infection in horses: results of a prevalence survey and experimental challenge. Veterinary Record, 130, 417–419.
- MALEY, S. W., LIVINGSTONE, M., RODGER, S. M., LONGBOTTOM, D. & BUXTON, D., 2009. Identification of Chlamydophila abortus and the development of lesions in placental tissues of experimentally infected sheep. Veterinary Microbiology, 135, 122-127.
- MCCHESNEY, S. L., ENGLAND, J. J. & MCCHESNEY, A. E., 1982. Chlamydia psittaci induced pneumonia in a horse. Cornell Veterinarian, 72, 92–97.
- MCNUTT, S. H. & WALLER, E. F., 1940. Sporadic bovine encephalomyelitis (Buss disease). Cornell Veterinarian, 30, 437–448.
- MILNE, C. E., GUNN, G. J., ENTRICAN, G. & LONGBOTTOM, D., 2009. Epidemiological modelling of chlamydial abortion in sheep flocks. Veterinary Microbiology 135, 128-133.
- MOORTHY, A. R. S. & SPRADBROW, P. B., 1978. Chlamydia psittaci infection of horses with respiratory disease. Equine Veterinary Journal, 10, 38–42.
- MOULDER, J. W., 1985. Comparative biology of intra-cellular parasitism. Microbiology Review, 49, 298–337.
- NIETFELD, J. C., JANKE, B. H., LESLIE-STEEN, P., ROBISON, D. J. & ZEMAN, D. H., 1993. Small intestinal Chlamydia infection in piglets. Journal of Veterinary Diagnostic Investigation, 5, 114–117.
- NIETFELD, J. C., LESLIE-STEEN, P., ZEMAN, D. H. & NELSON, D., 1997. Prevalence of intestinal chlamydial infection in pigs in the midwest, as determined by immunoperoxidase staining. American Journal of Veterinary Research, 58, 260–264.
- PAGE, L. A., 1966. Revision of the family Chlamydiaceae Rake (Rickettsiales): Unification of the Psittacosis-lymphogranuloma venereum-trachoma group of organisms in the genus Chlamydia Jones, Rake and Stearns, 1945. International Journal of Systematic Bacteriology, 16, 223–252.
- PAGE, L. A., 1968. Proposal for the recognition of two species in the genus Chlamydia Jones, Rake, and Stearns, 1945. International Journal of Systematic Bacteriology, 18, 51–66.
- PANTCHEV, A., STING, R., BAUERFEIND, R., TYCZKA, J. & SACHSE, K., 2010. Detection of all Chlamydophila and Chlamydia spp. of veterinary interest using species-specific real- time PCR assay. Comparative Immunology, Microbiology and Infectious Diseases, 33, 473–484.
- PAPP, J. R. & SHEWEN, P. E., 1996. Pregnancy failure following vaginal infection of sheep with Chlamydia psittaci prior to breeding. Infection and Immunity, 64, 1116–1125.
- PAPP, J. R., SHEWEN, P. E. & GARTLEY, C. J., 1994. Abortion and subsequent excretion of chlamydiae from the reproductive tract of sheep during estrus. Infection and Immunity, 62, 3786–3792.
- PAPP, J. R., SHEWEN, P. E., THORN, C. E. & ANDERSEN, A. A., 1998. Immunocytologic detection of Chlamydia psittaci from cervical and vaginal samples of chronically infected ewes. Canadian Journal of Veterinary Research, 62, 72–74.
- PIENAAR, J. G. & SCHUTTE, A. P., 1975. The occurrence and pathology of chlamydiosis in domestic and laboratory animals: A review. Onderstepoort Journal of Veterinary Research, 42, 77–90.
- PIENAAR, J. G. & SCHUTTE, A. P., 1994. Chlamydiosis. In: COETZER, J. A. W., THOMSON, G. R. & TUSTIN, R. C., (eds.). Infectious Diseases of Livestock with Special Reference to Southern Africa. Cape Town, South Africa: Oxford University Press, 378–389.
- PIERCY, D. W., GRIFFITHS, P. C. & TEALE, C. J., 1999. Encephalitis related to Chlamydia psittaci infection in a 14-week-old calf. Veterinary Record, 144, 126–128.
- PILLONEL, T., BERTELLI, C. & GREUB, G., 2018. Environmental Metagenomic Assemblies Reveal Seven New Highly Divergent Chlamydial Lineages and Hallmarks of a Conserved Intracellular Lifestyle. Frontiers in Microbiology, 9, 79.
- POSPISCHIL, A. & WOOD, R. L., 1987. Intestinal Chlamydia in pigs. Veterinary Pathology, 24, 568–570.
- REGGIARDO, C., FUHRMANN, T. J., MEERDINK, G. L. & BICKNELL, E. J., 1989. Diagnostic features of chlamydial infection in dairy calves. Journal of Veterinary Diagnostic Investigations, 1, 305–308.
- RITTER, J., 1880. Beitrag zur Frage des Pneumotyphus (Eine Hausepidemie in Uster [Schweiz] betreffend). Deutsches Archiv für Klinische Medizin, 25, 53-96.
- RODOLAKIS, A., 1983. In vitro and in vivo properties of chemically induced temperature-sensitive mutants of Chlamydia psittaci var ovis: Screening in a murine model. Infection and Immunity, 42, 525–530.
- RODOLAKIS, A. & BERNARD, F., 1984. Vaccination with temperaturesensitive mutant of Chlamydia psittaci against enzootic abortion of ewes. Veterinary Record, 114, 193–194.
- RODOLAKIS, A., SALINAS, J. & PAPP, J., 1998. Recent advances on ovine chlamydial abortion. Veterinary Research, 29, 275–288.
- RODOLAKIS, A. & SOURIAU, A., 1983. Response of ewes to temperaturesensitive mutants of Chlamydia psittaci obtained by nitrosoguanidine mutagenesis. Annales De Recherches Veterinaires, 14, 155–161.
- RODOLAKIS, A. & SOURIAU, A., 1986. Response of goats to vaccination with temperature-sensitive mutants of Chlamydia psittaci (var ovis) obtained by NTG mutagenesis. American Journal of Veterinary Research, 47, 2627–2631.
- ROGERS, D. G. & ANDERSEN, A. A., 1996. Intestinal lesions caused by two swine chlamydial isolates in gnotobiotic pigs. Journal of Veterinary Diagnostic Investigation, 8, 433–440.
- ROGERS, D. G. & ANDERSEN, A. A., 1999. Conjunctivitis caused by a swine Chlamydia trachomatis-like organism in gnotobiotic pigs. Journal of Veterinary Diagnostic Investigation, 11, 341–344.
- ROGERS, D. G. & ANDERSEN, A. A., 2000. Intestinal lesions caused by a strain of Chlamydia suis in weanling pigs infected at 21 days of age. Journal of Veterinary Diagnostic Investigation, 12, 233–239.
- ROGERS, D. G., ANDERSEN, A. A., HOGG, A., NIELSEN, D. L. & HUEBERT, M. A., 1993. Conjunctivitis and keratoconjunctivitis associated with chlamydiae in swine. Journal of the American Veterinary Medical Association, 203, 1321–1323.
- ROGERS, D. G., ANDERSEN, A. A. & HUNSAKER, B. D., 1996. Lung and nasal lesions caused by a swine chlamydial isolate in gnotobiotic pigs. Journal of Veterinary Diagnostic Investigation, 8, 45–55.
- RURANGIRWA, F. R., DILBECK, P. M., CRAWFORD, T. B., MCGUIRE, T. C. & MCELWAIN, T. F., 1999. Analysis of the 16S rRNA gene of micro-organism WSU 86-1044 from an aborted bovine foetus reveals that it is a member of the order Chlamydiales: proposal of Waddliaceae fam. nov., Waddlia chondrophila gen. nov., sp. nov. International Journal of Systematic Bacteriology, 49, 577–581.
- SACHSE, K., LAROUCAU, K., RIEGE, K., WEHNER, S., DILCHER, M., CREASY, H. H., WEIDMANN, M., MYERS, G., VORIMORE, F., VICARI, N., MAGNINO, S., LIEBLER-TENORIO, E., RUETTGER, A., BAVOIL, P. M., HUFERT, F. T., ROSSELLÓ-MÓRA, R. & MARZ, M., 2014. Evidence for the existence of two new members of the family Chlamydiaceae and proposal of Chlamydia avium sp. nov. and Chlamydia gallinacea sp. nov. Systematic and Applied Microbiology,37, 79-88.
- SACHSE, K., LAROUCAU, K. & VANROMPAY, D., 2015. Avian Chlamydiosis. Current Clinical Microbiology Reports, 2, 10-21.
- SACHSE, K., VRETOU, E., LIVINGSTONE, M., BOREL, N., POSPISCHIL, A. & LONGBOTTOM, D., 2009. Recent developments in the laboratory diagnosis of chlamydial infections. Veterinary Microbiology,135, 2-21.
- SALTI-MONTESANTO, V., TSOLI, E., PAPAVASSILIOU, P., PSARROU, E., MARKEY, B. K., JONES, G. & VRETOU, E., 1997. Diagnosis of ovine enzootic abortion, using a competitive ELISA based on monoclonal antibodies against variable segments 1 and 2 of the major outer membrane protein of Chlamydia psittaci serotype 1. American Journal of Veterinary Research, 58, 228–235.
- SAMMIN, D. J., MARKEY, B. K., QUINN, P. J., MCELROY, M. C. & BASSETT, H. F., 2006. Comparison of fetal and maternal inflammatory responses in the ovine placenta after experimental infection with Chlamydophila abortus. Journal of Comparative Pathology, 135, 83-92.
- SANDERSON, T. P. & ANDERSEN, A. A., 1989. Evaluation of an enzyme immunoassay for detection of Chlamydia psittaci in vaginal secretions, placentas, and fetal tissues from aborting ewes. Journal of Veterinary Diagnostic Investigations, 1, 309–315.
- SARGISON, N. D., TRUYERS, I. G., HOWIE, F. E., THOMSON, J. R., COX, A. L., LIVINGSTONE, M. & LONGBOTTOM, D., 2015. Identification of the 1B vaccine strain of Chlamydia abortus in aborted placentas during the investigation of toxaemic and systemic disease in sheep. New Zealand Veterinary Journal, 63, 284-287. DOI: 10.1080/00480169.2015.1018365.
- SCHACHTER, J., et al., 2001. Radical changes to chlamydial taxonomy are not necessary just yet. International Journal of Systematic and Evolutionary Microbiology, 51, 249-243.
- SCHAUTTEET, K., DE CLERCQ, E., MIRY, C., VAN GROENWEGHE, F., DELAVA, P., KALMAR, I. & VANROMPAY, D., 2013. Tetracycline-resistant Chlamydia suis in cases of reproductive failure on Belgian, Cypriote and Israeli pig production farms. Journal of Medical Microbiology, 62, 331-334. DOI: 10.1099/jmm.0.042861-0.
- SCHILLER, I., KOESTERS, R., WEILENMANN, R., KALTENBOECK, B. & POSPISCHIL, A., 1997. Polymerase chain reaction (PCR) detection of porcine Chlamydia trachomatis and ruminant Chlamydia psittaci serovar 1 DNA in formalin-fixed intestinal specimens from swine. Journal of Veterinary Medicine (B), 44, 185–191.
- SCHILLER, I., KOESTERS, R., WEILENMANN, R., THOMA, R., KALTENBOECK, B., HEITZ, P. & POSPISCHIL, A., 1997. Mixed infections with porcine Chlamydia trachomatis/pecorum and infections with ruminant Chlamydia psittaci serovar 1 associated with abortions in swine. Veterinary Microbiology, 58, 251–260.
- SCHOOP, G. & KAUKER, E., 1956. Infektion eines Rinderbestandes durch ein Virus der Psittakosis-Lymphogranuloma-Gruppe. Gehäufte Aborte im Verlauf der Erkrankungen. Deutsche Tierärztliche Wochenschrift, 63, 233–235.
- SCHRAMM, N., BAGNELL, C. R. & WYRICK, P. B., 1996. Vesicles containing Chlamydia trachomatis serovar L2 remain above pH 6 within HEC-1B cells. Infection and Immunity, 64, 1208–1214.
- SCHUTTE, A. P. & PIENAAR, J. G., 1977. Chlamydiose by skape en beeste in Suid-Afrika. Journal of the South African Veterinary Association, 48, 261–265.
- SETH-SMITH, H. M. B., BUSÓ, L. S., LIVINGSTONE, M., SAIT, M., HARRIS, S. R., AITCHISON, K. D., VRETOU, E., SIARKOU, V. I., LAROUCAU, K., SACHSE, K., LONGBOTTOM, D. & THOMSON, N. R., 2017. European Chlamydia abortus livestock isolate genomes reveal unusual stability and limited diversity, reflected in geographical signatures. BMC Genomics, 18, 344.
- SHARMA, M., ROGERS, D. G. & ANDERSEN, A. A., 1999. Prevalence of Chlamydia trachomatis infection in swine. Abstracts of the 99th General Meeting of the American Society for Microbiology, May 30–June 3, 174.
- SHUPE, J. L. & STORZ, J., 1964. Pathologic study of psittacosis lymphogranuloma polyarthritis of lambs. American Journal of Veterinary Research, 25, 943–941.
- SIARKOU, V. I., VORIMORE, F., VICARI, N., MAGNINO, S., RODOLAKIS, A., PANNEKOEK, Y., SACHSE, K., LONGBOTTOM, D. & LAROUCAU, K., 2015. Diversification and distribution of ruminant Chlamydia abortus clones assessed by MLST and MLVA. PLoS One, 10, e0126433.
- SILLIS, M. & LONGBOTTOM, D., 2011. Chlamydiosis. In: Zoonoses, PALMER, S. R., SOULSBY, L., TORGERSON, P. R. & BROWN, D. W. G., (eds.), 2nd edition, Oxford University Press, 146-157.
- SOURIAU, A., BOSSERAY, N., RODOLAKIS, A., LANTIER, F. & PLOMMET, M., 1988. Anti-chlamydial immunity in ewes conferred by vaccination with a combination of three live chlamydia, brucella and salmonella vaccines. Veterinary Record, 123, 29–32.
- SOURIAU, A. & RODOLAKIS, A., 1986. Rapid detection of Chlamydia psittaci in vaginal swabs of aborted ewes and goats by enzyme-linked immunosorbent assay (ELISA). Veterinary Microbiology, 11, 251–259.
- SPALATIN, J., FRASER, C. E. O., CONNELL, R., HANSON, R. P. & BERMAN, D. T., 1966. Agents of psittacosis-lymphogranuloma venereum group isolated from muskrats and snowshoe hares in Saskatchewan. Canadian Journal of Comparative Medicine and Veterinary Science, 30, 260–264.
- SPENCER, W. N. & JOHNSON, F. W. A., 1983. Simple transport medium for the isolation of Chlamydia psittaci from clinical material. Veterinary Record, 113, 535–536.
- STAMP, J. T., MCEWEN, A. D., WATT, J. A. A. & NISBET, D. I., 1950. Enzootic abortion in ewes. I. Transmission of the disease. Veterinary Record, 62, 251–254.
- STEPHENS, R. S., MYERS, G., EPPINGER, M. & BAVOIL, P. M., 2009. Divergence without difference: phylogenetics and taxonomy of Chlamydia resolved. FEMS Immunology and Medical Microbiology, 55, 115-119.
- STOREY, C., LUSHER, M., YATES, P. & RICHMOND, S., 1993. Evidence for Chlamydia pneumoniae of non-human origin. Journal of General Microbiology, 139, 2621–2626.
- STORZ, J., 1967. Psittacosis agents as cause of polyarthritis in cattle and sheep. Veterinary Medical Review, 2(3), 125–139.
- STORZ, J., 1971. Chlamydia and Chlamydia-induced Diseases. 1st edn. Springfield, IL: Charles C. Thomas.
- STORZ, J., CARROLL, E. J., BALL, L. & FAULKNER, L. C., 1968. Isolation of a psittacosis agent (Chlamydia) from semen and epididymis of bulls with seminal vesiculitis syndrome. American Journal of Veterinary Research, 29, 549–555.
- STORZ, J., MCKERCHER, D. G., HOWARTH, J. A. & STRAUB, O. C., 1960. The isolation of a viral agent from epizootic bovine abortion. Journal of the American Veterinary Medical Association, 137, 509–514.
- STORZ, J. & SPEARS, P., 1977. Chlamydiales: Properties, cycle of development and effect on eukaryotic host cells. Current Topics in Microbiological Immunology, 76, 167–214.
- SURI, A. K., GUERIN, B., HUMBLOT, P. & THIBIER, M., 1986. Effect of infection of the genital tract on the concentration of IgG and albumin in bull serum and semen. Veterinary Immunology and Immunopathology, 13, 273–278.
- SZEREDI, L., SCHILLER, I., SYDLER, T., GUSCETTI, F., HEINEN, E., CORBOZ, L., EGGENBERGER, E., JONES, G. E. & POSPISCHIL, A., 1996. Intestinal Chlamydia in finishing pigs. Veterinary Pathology, 33, 369–374.
- SZYMAŃSKA-CZERWIŃSKA, M., MITURA, A., NIEMCZUK, K., ZARĘBA, K., JODEŁKO, A., PLUTA, A., SCHARF, S., VITEK, B., AAZIZ, R., VORIMORE, F., LAROUCAU, K. & SCHNEE, C., 2017. Dissemination and genetic diversity of chlamydial agents in Polish wildfowl: Isolation and molecular characterisation of avian Chlamydia abortus strains. PLoS One, 12, e 0174599.
- TAPPE, J. & ANDERSEN, A., 1991. Inhibition of endosomal acidification by Chlamydia psittaci. Abstracts of the 91st General Meeting of the American Society for Microbiology, May 5–9, 103.
- TAYLOR, D. J., 1999. Chlamydiae. In: STRAW, B. E., D’ALLAIRE, S., MENGLING, W. L. & TAYLOR, D. J., (eds.). Diseases of Swine. Ames, IA: Iowa State University Press.
- TAYLOR-BROWN, A. & POLKINGHORNE, A., 2017. New and emerging chlamydial infections of creatures great and small. New Microbes and New Infections,18, 28-33.
- TAYLOR-BROWN, A., VAUGHAN, L., GREUB, G., TIMMS, P. & POLKINGHORNE, A., 2015. Twenty years of research into Chlamydia-like organisms: a revolution in our understanding of the biology and pathogenicity of members of the phylum Chlamydiae. Pathogens and Disease, 73, 1-15.
- TEANKUM, K., POSPISCHIL, A., JANETT, F., BRUGNERA, E., HOELZLE, L. E., HOELZLE, K., WEILENMANN, R., ZIMMERMANN, D. R., GERBER, A., POLKINGHORNE, A. & BOREL, N., 2007. Prevalence of chlamydiae in semen and genital tracts of bulls, rams and bucks. Theriogenology, 67, 303-310.
- TUSTIN, R. C., MARÉ, J. & VAN HEERDEN, A., 1961. A disease of calves resembling sporadic bovine encephalomyelitis. Journal of the South African Veterinary Medical Association, 32, 117–123.
- VAN LOOCK, M., VANROMPAY, D., HERRMANN, B., VAN DER STAPPEN, J., VOLCKAERT, G., GODDEERIS, B. M. & EVERETT, K. D. E., 2003. Missing links in the divergence of Chlamydophila abortus from Chlamydophila psittaci. International Journal of Systematic and Evolutionary Microbiology, 53, 761–770.
- VANROMPAY, D., 2013. Avian Chlamydiosis. In: Swanye, D. E., (ed.). Diseases of Poultry. 13th edition Vol Section III: Bacterial Diseases, Wiley-Blackwell Publishing.
- VANROMPAY, D., NGUYEN, T. L. A., CUTLER, S. J. & BUTAYE, P., 2018. Antimicrobial resistance in Chlamydiales, Rickettsia, Coxiella, and other intracellular pathogens. Microbiology Spectrum,6.
- VEZNIK, Z., KUMMER, V., CANDERLE, J., MASKOVA, J., SVECOVA, D., POSPISIL, L., DIBLIKOVA, I. & ZRALY, A., 1996. Detection of Chlamydia in inflammation of the cervix and uterus in cows. Veterinary Medicine (Praha), 41, 297–304.
- VORIMORE, F., CAVANNA, N., VICARI, N., MAGNINO, S., WILLEMS, H., RODOLAKIS, A., SIARKOU, V. I. & LAROUCAU, K., 2012. High-resolution melt PCR analysis for rapid identification of Chlamydia abortus live vaccine strain 1B among C. abortus strains and field isolates. Journal of Microbiological Methods, 90, 241–244.
- VORIMORE, F., HSIA, R. C., HUOT-CREASY, H., BASTIAN, S., DERUYTER, L., PASSET, A., SACHSE, K., BAVOIL, P., MYERS, G. & LAROUCAU, K., 2013. Isolation of a new Chlamydia species from the Feral Sacred Ibis (Threskiornis aethiopicus): Chlamydia ibidis. PLoS One,8, e74823.
- VRETOU, E., LOUTRARI, H., MARIANI, L., COSTELIDOU, K., ELIADES, P., CONIDOU, G., KARAMANOU, S., MANGANA, O., SIARKOU, V. & PAPADOPOULOS, O., 1996. Diversity among abortion strains of Chlamydia psittaci demonstrated by inclusion morphology, polypeptide profiles and monoclonal antibodies. Veterinary Microbiology, 51, 275–289.
- WALKER, E., LEE, E. J., TIMMS, P. & POLKINGHORNE, A., 2015. Chlamydia pecorum infections in sheep and cattle: a common and under-recognised infectious disease with significant impact on animal health. Veterinary Journal, 206, 252-260.
- WALKER, E., MOORE, C., SHEARER, P., JELOCNIK, M., BOMMANA, S., TIMMS, P. & POLKINGHORNE, A., 2016. Clinical, diagnostic and pathologic features of presumptive cases of Chlamydia pecorum-associated arthritis in Australian sheep flocks. BMC Veterinary Research, 12, 193.
- WARD, M. E., 1988. The chlamydial developmental cycle. In: BARON, A. L., (ed.). Microbiology of Chlamydia. Boca Raton, FL: CRC Press.
- WARD, M. E., 1999. Mechanisms of Chlamydia-induced disease. In: STEPHENS, R. S., (ed.). Chlamydia: Intracellular Biology, Pathogenesis, and Immunity. Washington, DC: American Society for Microbiology.
- WARDROP, S., FOWLER, A., O’CALLAGHAN, P., GIFFARD, P. & TIMMS, P., 1999. Characterization of the koala biovar of Chlamydia pneumoniae at four gene loci-ompAVD4, ompB, 16S rRNA, groESL spacer region. Systematic Applied Microbiology, 22, 22–27.
- WHEELHOUSE, N., AITCHSON, K., LAROUCAU, K., THOMSON, J. & LONGBOTTOM, D., 2010. Evidence of Chlamydophila abortus vaccine strain 1B as a possible cause of ovine enzootic abortion. Vaccine, 28, 5657–5663.
- WILLS, J. M., WATSON, G., LUSHER, M., MAIR, T. S., WOOD, D. & RICHMOND, S. J., 1990. Characterization of Chlamydia psittaci isolated from a horse. Veterinary Microbiology, 24, 11–19.
- WILSMORE, A. J., PARSONS, V. & DAWSON, M., 1984. Experiments to demonstrate routes of transmission of ovine enzootic abortion. British Veterinary Journal, 140, 380–391.
- WILSON, K., LIVINGSTONE, M. & LONGBOTTOM, D., 2009. Comparative evaluation of eight serological assays for diagnosing Chlamydophila abortus infection in sheep. Veterinary Microbiology, 135, 38-45.
- WITTENBRINK, M. M., KIRPAL, G., THIELE, D., FISCHER, D., KRAUSS, H. & BISPING, W., 1994. Detection of Chlamydia psittaci in vaginal discharge of cows: a necessary enlargement of bacteriologic diagnosis for the etiologic clarification of fertility disorders in the female cow. Journal of Veterinary Medicine (B), 41, 492–503.
- WITTENBRINK, M. M., WEN, X., BOHMER, N., AMTSBERG, G. & BINDER, A., 1991. Bakteriologische Untersuchungen zum vorkommen von Chlamydia psittaci in Organen von Schweinen und in Abortierten Schweinefeten. Journal of Veterinary Medicine (B), 38, 411–420.