- Infectious Diseases of Livestock
- Part 1
- Vectors: Mosquitoes
- Vectors: Ticks
- Vectors: Tsetse flies
- Vectors: Muscidae
- Vectors: Tabanidae
- Vectors: Culicoides spp.
- Vectors: Mosquitoes
- Classification, epidemiology and control of arthropod-borne viruses
- Special factors affecting the control of livestock diseases in sub-Saharan Africa
- The control of infectious diseases of livestock: Making appropriate decisions in different epidemiological and socioeconomic conditions
- Infectious diseases of animals in sub-Saharan Africa: The wildlife⁄livestock interface
- Vaccination: An approach to the control of infectious diseases
- African animal trypanosomoses
- Dourine
- Trichomonosis
- Amoebic infections
- GENERAL INTRODUCTION: COCCIDIA
- Coccidiosis
- Cryptosporidiosis
- Toxoplasmosis
- Besnoitiosis
- Sarcocystosis
- Balantidiosis
- Leishmaniosis
- Neosporosis
- Equine protozoal myeloencephalitis
- GENERAL INTRODUCTION: BABESIOSES
- Bovine babesiosis
- Equine piroplasmosis
- Porcine babesiosis
- Ovine babesiosis
- GENERAL INTRODUCTION: THEILERIOSES OF CATTLE
- East Coast fever
- Corridor disease
- Zimbabwe theileriosis
- Turning sickness
- Theileria taurotragi infection
- Theileria mutans infection
- Theileria annulata theileriosis
- Theileriosis of sheep and goats
- Theileria buffeli⁄orientalis infection
- Non-pathogenic Theileria species in cattle
- GENERAL INTRODUCTION: RICKETTSIAL, CHLAMYDIAL AND HAEMOTROPIC MYCOPLASMAL DISEASES
- Heartwater
- Lesser known rickettsial infections in animals and humans
- Chlamydiosis
- Q fever
- Eperythrozoonosis
- Bovine Haemobartonellosis
- Potomac horse fever
- GENERAL INTRODUCTION: ANAPLASMOSES
- Bovine anaplasmosis
- Ovine and caprine anaplasmosis
Vectors: Mosquitoes
This content is distributed under the following licence: Attribution-NonCommercial CC BY-NC  View Creative Commons Licence details here
View Creative Commons Licence details here
Mosquito vectors: a Southern African context
Previous author: P G JUPP
Current authors: A J CORNEL, L E O BRAACK and A P G ALMEIDA
Introduction
For the purposes of this chapter we consider South Africa, Lesotho, Eswatini (Swaziland), Botswana, Namibia, Zimbabwe, and southern regions of Angola and Mocambique as southern African because they share areas of similar subtropical and temperate humid, dry and plains, plateau and mountainous ecological zones (http://www.aag.org/galleries/publications-files/africa_ecosystems_booklet.pdf -Accessed April 2019) and diversity of mosquito and other arthropod vector species on animal and human diseases.
The “cosmopolitanizing” of Ae. aegypti and associated global outbreaks of dengue (DENV), Chikungunya (CHIKV), Zika (ZIKV) and yellow fever (YFV) virus infections has stoked a “fire” in arboviral interest and reviews have recently been published that discuss the resurgence of interest and risks of infection by mosquito-borne arboviruses in Africa, including southern Africa.13, 14, 46, 148 These reviews provide summaries of the multi-continent re-emerging viruses such as West Nile (WNV), ZIKV, YFV, CHIKV and dengue viruses in the last two decades. Recent enhancements in surveillance in southern Africa have now revealed considerable annual arboviral activity of the better known arboviruses such as Rift Valley fever (RVFV), Wesselsbron (WESV) and WNV but also the previously lesser known viruses such as Sindbis (SINV), Shuni (SHUNV), Middelburg (MIDV), and Usutu (USUV) viruses that infect both livestock and wild animals.145 A list of these mosquito-borne arboviruses in southern Africa is given in Table 1. Unfortunately a good understanding of the natural cycles of many of these viruses, especially concerning the role of mosquito vectors and reservoir host vertebrates is lacking and is occasionally quite speculative.
Mosquito-borne arboviruses characteristically multiply in both the vertebrate host and the mosquito vector. Successful transmission is dependent upon the virus producing viraemia in the vertebrate, while the mosquito that feeds on the vertebrate must develop a salivary gland infection followed by excretion of virus in the saliva. The arboviruses belong to several families and genera and their taxonomic categories and host relations are discussed elsewhere. Apart from virus isolation from wild-caught mosquitoes, the lines of evidence incriminating a mosquito species as a vector are its susceptibility to infection with, and its ability to transmit, the virus concerned (vector competence) plus its relative density and ecological characteristics. Vector competence is assessed quantitatively in laboratory tests while data on density and ecology are obtained by field observations. Relevant aspects of bioecology are relative density, seasonal dynamics, feeding behaviour, including time of biting activity, host preferences, feeding frequencies, larval habitat associations and biology of the immature stages. Together, the aspects of adult bioecology that affect and condition transmission are usually expressed as vectorial capacity, a concept which was developed by Garrett-Jones29 after the mathematical modelling for malaria by MacDonald85
Climatic factors influence arboviruses both directly and indirectly. Temperature conditions directly affect the multiplication of the virus in the mosquito vector, low temperatures slowing down viral replication and lowering vector competence.56, 128 However, more importantly, climatic factors operate through their influence on the geographical distribution and prevalence of the viral hosts, both mosquito and vertebrate. In the case of mosquitoes, high rainfall and temperatures can lead to higher population densities of species belonging to all genera, particularly Aedes. Climate therefore has a considerable effect on the epidemiology of arbovirus infections.
Apart from some work in the temperate, moist highlands and subtropical, moist lowlands of Zimbabwe, the areas of southern Africa lying outside South Africa have largely only been studied for the presence of mosquito-borne viruses by means of serology, with little virus isolation from wild-caught mosquitoes being attempted. There is considerable arbovirus activity in the tropical region of Mocambique and the Zambezi valley and in the subtropical, moist northern belt that extends westwards from Zimbabwe and embraces the Okavango swamps in northern Botswana, and the Zambezi Region (formerly Caprivi strip) and Etosha pan in north-eastern Namibia.11, 35, 76, 80
Arbovirus activity in southern Africa is more or less confined to the subtropical and tropical summer rainfall areas. The inland plateau has virtually no rain from May to November, while in the winter, from June to August, night temperatures are often near freezing and periodically fall below this. In eastern (northern South Africa and Mocambique) and western coastal areas (Angola) winters are milder with little to no frost .
In South Africa, there has been a change in the complexity of rangeland animal rearing, especially since the 1990s, and many private farmers rear wildlife for ecotourism, conservation, trophy hunting and game meat production. The game ranging industry is now worth more than $700 million annually and game animals are considered a valuable form of “livestock” that succumb to mosquito-borne arboviral infections. Many farmers have mixed livestock and game animal rangelands that create opportunistic interactions between different arboviral reservoir host animal populations and bridge mosquito vectors.41, 136 This in turn creates added risks of viral transmission for farm workers, neighbouring farm personnel and veterinarians, especially via exposure to infected carcasses.143 Out of all the mammals mentioned, horses and donkeys may serve as excellent sentinel animals for certain arbovirus infections. These species are used in South Africa as sentinels for monitoring African horse sickness and equine encephalosis viruses and are approved for use for WNV surveillance by the World health Organization. Sera from these sentinels can also be used to screen for other arboviruses.151
Of the mosquito-borne viruses that occur in southern Africa, RVFV has received most attention because it is periodically responsible for serious disease especially in sheep and humans in the subcontinent (see Rift Valley fever). In fact, RVFV is an emerging pathogen and is included in the World Health Organization “Blueprint” list of ten prioritized viruses that will likely cause future epidemics for which there are insufficient counter-measures in place.123 West Nile virus37 and others including RVFV10, 113 SHUNV, WESV, MIDV and SINV [92, 139] are not always associated with epidemics and there is growing evidence that transmission of some of these viruses occurs cryptically annually in the inter-epidemic period, and that they are geographically quite widely spread in South Africa. Some of these viruses may regularly cause serious neurological disease in humans158 and horses,49, 50, 144, 145, 147, 155 and less commonly in wild animals such as giraffe, rhino, warthog, African buffalo and crocodiles.152 Banzi virus(BANV) and WESV have also been incriminated as causes of congenital abnormalities in small stock and cattle in South Africa.9
Other mosquito-borne viruses of the families Togaviridae and Flaviviridae that do not occur in Africa, but which cause important diseases mainly in horses and humans outside Africa, include the alphaviruses of Eastern, Western and Venezuelan encephalomyelitides and the flaviviruses of Murray Valley, St. Louis and Japanese B encephalitides (see the chapters on the specific diseases).
Table 1 Mosquito-borne viruses causing disease in livestock and/or humans in southern Africa. Species names follow that of the composite classification.
| VIRUS | MOSQUITO VECTOR | WILD VERTEBRATE HOST | HOSTS SHOWING DISEASE | ||
|---|---|---|---|---|---|
| HUMANS | LIVESTOCK | ||||
| Togaviridae | |||||
| Alphavirus | Chikungunya | Ae. (Dic.) furcifer | Primates | + | - |
| Middelburg | Ae. (Och.)caballus, Ma. africana | Ruminants? | + | + | |
| Ndumu | Ae. (Neo.) mcintoshi, Ae. (Adm.) ochraceus, Ae. (Neo.) circumluteolus, Cx. (Eum.) rubinotus and Ma. uniformis | unknown | + | ? | |
| Sindbis | Cx. (Cux.) univittatus | Birds | + | - | |
| Flaviviridae | |||||
| Flavivirus | Banzi | Cx. (Eum.) rubinotus | Rodents | + | +a |
| Spondweni | Ae. (Neo.) circumluteolus, Ma. uniformis, Ma. africana, Ae. (Adm.) cumminsii, Cx. (Cux.) neavei, Cx. (Cux). univittatus, Er. silvestris | unknown | + | ? | |
| Usutu | Ae. (Steg.) albopictus, An. maculipennis, Coq. aurites, Cx. (Cux.) neavei, Cx. (Cux.) perexiguus, Cx. (Cux) perfuscus, Cx. (Cux.) pipiens, Cx. (Cux.) univittatus | Birds and Bats | + | + | |
| Wesselsbron | Ae. (Neo.) circumluteolus, Ae. (Neo.) mcintoshi, Ae. (Och.) caballus/juppi | Rodents? | + | + | |
| West Nile | Cx. (Cux.) univittatus | Birds | + | + | |
| Zika | Ae. (Steg). aegypti, Ae. (Steg.) albopictus, Ae. (Steg.) luteocephalus, Ae. (Steg.) metallicus, Ae. (Dic.) furcifer, Ae. (Fre) vittatus, Ae. (Adm.) tarsalis, Ae. (Adm.) dalziella, Ae. (Adm.) fowleri, An. (Cel.) gambiae ss, Eret. quinquevittatus, Ma. uniformis | Primates | + | - | |
| Bunyaviridae | |||||
| Phlebovirus | Rift Valley fever | Cx. (Cux.) theileri, Cx. (Cux.) zombaensis, Ae. (Neo.) circumluteolus, Ae. (Neo.) mcintoshi, Ae. (Och.) caballus/juppi | Antelope? | + | + |
| Orthobunyavirus | Bunyamwera | Ae. (Neo.) circumluteolus, Ae. (Neo.) mcintoshi, An. (Cel.) funestus s.l | Rodents+ | + | ? |
| Bwamba | Ae. (Dic.) furcifer, An. (Ano.) coustani, An. (Cel.) funestus s.l., An. (Cel.) gambiae s.l, Ma. uniformis. | unknown | + | ? | |
| Germiston | Cx. (Eum.) rubinotus, Cx. (Cux.) theileri? | rodents | + | ? | |
| Lumbo | Ae. (Sku.) pembaensis | Non-human primates? | + | unknown | |
| Pongola | Ae. (Neo.) circumluteolus, Ae. (Neo.) mcintoshi | unknown | + | unknown | |
| Shuni | Cx. (Cux) theileri? | In brains of rhino, warthog, buffalo, crocodile | + | + | |
| Witwatersrand | Cx. (Eum) rubinotus | rodents | + | - | |
| Rhabdoviridae | Mossuril | Cx. spp. (Cux) sitiens? | ? | + | ? |
a Disease produced after inoculation with virus in laboratory- Banzi virus
Notes on mosquito classification
There are currently two Aedini classification systems, the multi-generic system proposed by Reinert et al 2009129 and the other more practical traditional or composite system proposed by Wilkerson et al 2015154 The composite classification follows much more closely the traditional classification system that until about 2010 was most generally accepted globally. Several species within the Aedini are vectors of arboviruses in southern Africa and these have been listed in Table 2 under the two classification systems. Table 2 also includes species name changes that have been proposed when using the multi-generic classification to agree in gender with the name of genus with which they are combined.39 While it is not the purpose of this chapter to discuss mosquito classification, we forewarn that the present state of affairs will result in further confusion. For example, a newly described mosquito species from Madagascar was named using the multi-generic classification system as Paulianus hirsutus. Under the composite classification this species would be known as Aedes (Paulianus) hirsutus. However, there is already a species, under the composite system, known as Aedes (Aedimorphus) hirsutus, a floodwater species common in Africa, and hence there are two different mosquito species with the same genus and species. Proponents of the composite system have not yet listed the new species from Madagascar,117 we guess largely because they do not know how to deal with this confusion that has been created. In this chapter the abbreviations for genera and subgenera of Culicidae follow the revised recommendations.154
Table 2. Multi-generic and traditional/composite classifications of arboviral vector Aedes species of southern Africa.
| Multi-generic classification129 | Composite classification154 | ||||
|---|---|---|---|---|---|
| Genus | Subgenus | species | Genus | Subgenus | species |
| Aedimorphus | cumminsii | Aedes | Aedimorphus | cumminsii | |
| dalziella | dalziella | ||||
| fowleri | fowleri | ||||
| ochraceus | ochraceus | ||||
| tarsalis | tarsalis | ||||
| Diceromyia | furifer | Aedes | Diceromyia | furifer | |
| Fredwardsius | vittatus | Aedes | Fredwardsius | vittatus | |
| Neomelanoconion | circumluteolum | Aedes | Neomelanoconion | circumluteolus | |
| mcintoshi | mcintoshi | ||||
| Ochlerotatus | juppius | caballus | Aedes | Ochlerotatus | caballus |
| juppi | juppi | ||||
| Skusea | pembaensis | Aedes | Skusea | pembaensis | |
| Stegomyia | Stegomyia? | aegypti | Aedes | Stegomyia | aegypti |
| uncertain | albopicta | albopictus | |||
| uncertain | luteocephala | luteocephalus | |||
| uncertain | metallica | metallicus | |||
Virus isolations from mosquitoes
Unfortunately, there is still considerable paucity of data on southern African arboviral mosquito vectors, especially in Mocambique, Botswana, Angola and Namibia. The implicated vector species in these countries are, for the most part, considered to be the same species as those from which isolates of virus have been made from in South Africa and in Zimbabwe. However, there are also multiple mosquito species in these countries that do not occur in South Africa and from which viruses have been isolated from other parts of Africa. The mosquito catalogue117 website serves as a useful resource for determining the distributions of mosquito species and a review by Braack et al 201813 provides a comprehensive list of all mosquito species from which mosquito-borne arboviruses have been isolated from in Africa. As mentioned above, isolation of virus from a wild caught mosquito is in itself not enough evidence for concluding its role as a vector, especially if virus isolates have been made from whole mosquito bodies, as viruses may not be efficiently transmitted by the saliva despite being in other body parts, as has been found for Ae. albopictus from the south of France for ZIKV.51 However, clues to potential vector species can be gained if multiple isolates of the same virus are made from a single species. Follow-up vector competence studies and virus isolations from salivary glands of wild caught mosquitoes for those species should then be performed to confirm vector status. Inability to maintain colonies of many specific mosquito species and lack of expertise and facilities to conduct these follow-up studies, especially in Africa, adds to the reasons for this paucity of confirmed African arboviral vector mosquito species.
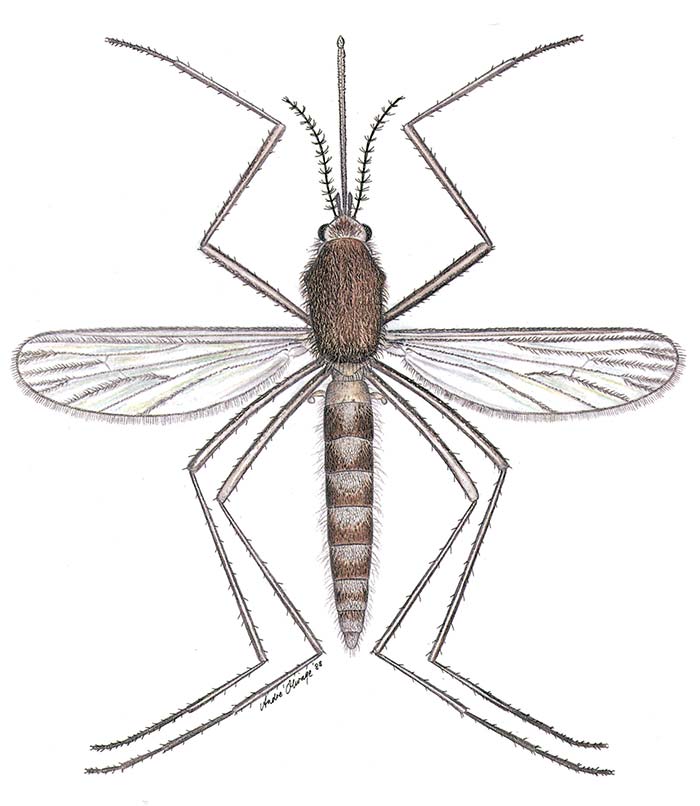
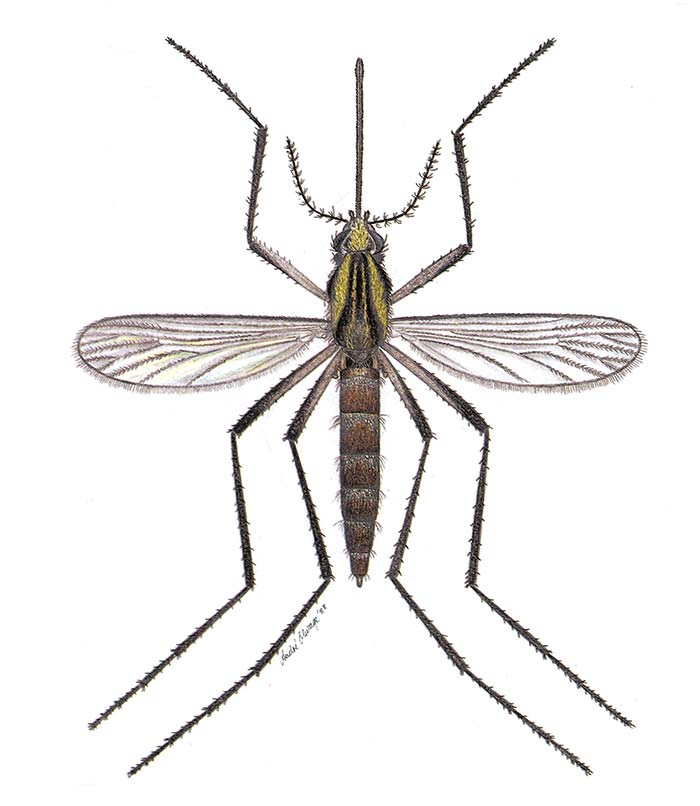
Table 3 lists the six mosquito species which yielded multiple isolations of RVFV when collected on farms during RVF epidemics among sheep and cattle in South Africa and Zimbabwe. The largest number of isolations on the inland plateau came from Cx. theileri (Figure 1) and in the KwaZulu-Natal lowlands from Cx. zombaensis. Fewer isolations were made from Aedine mosquitoes because collecting only began once an epidemic was already well under way, by which time the Aedine populations were waning. All the Aedes spp. given in Table 2 are floodwater mosquitoes belonging to the subgenera Neomelaniconion (Neo.) and Ochlerotatus (Och). Aedes (Neo.) mcintoshi and Ae. (Och.) juppi are illustrated in Figure 2 and Figure 3 respectively.
Table 3 Southern African mosquito species yielding multiple isolations of Rift Valley fever virus from 1953 to 1981: number of isolations and results of transmission experiments
| REGION | SPECIES | NO. ISOLATONS | 50% INFECTION THRESHOLDa LOG10LD50/ml | TRANSMISSION RATEb (DAYS POST-INFECTION) | REFERENCES |
|---|---|---|---|---|---|
| SOUTH AFRICA: | |||||
| KwaZulu-Natal | Cx. (Cux.) zombaensis | 7 | c. 7,5 | 3%, 41%, 42%, (14, 22, 29) | 72, 105 |
| Coastal lowlands | Ae. (Neo.) circumluteolus | 3 | c. 7,5 | 32-38% (16-31) | 68, 69, 72, 105 |
| SOUTH AFRICA: | |||||
| Inland plateau | Cx. (Cux.) theileri | 22 | c. 5,7 | 13-35% (15-22) | 31, 59, 89, 98, 103 |
| ` | Ae. (Och.) juppi | 3 | 8,7 | 6% (17) | 103 |
| ` | Ae. (Och.) caballus s.l. | 6 | 8,0c | 0%c (12, 13) | 31, 103 |
| ` | Ae. (Neo.) mcintoshi d | 2 | 8,1 | 17% (10,17) | 103 |
| ZIMBABWE HIGHLANDS | Ae. (Neo.) mcintoshi d | 2 | 89 |
a The titre of virus needed to infect 50 per cent of the mosquitoes
b The proportion of infected mosquitoes transmitting virus to hamsters
c Experiments done with Ae. (Och) caballus s.s.
d Reported as Ae. (Neo) lineatopennis
From 1956 to 1980, 139 isolations of WNV were made from mosquitoes in South Africa. Of these, 136 came from mosquitoes collected on the inland plateau and three from the KwaZulu-Natal coastal lowlands. On the plateau, 128 of the isolations were from Culex univittatus, which occurs throughout this region.97 There was a large epidemic of WNV infection in the Karoo and Northern Cape Province in 1974, when the infection rate of Cx. univittatus populations reached 39 infected mosquitoes per 1 000 at Upington.104 The low incidence of WNV in the KwaZulu-Natal coastal lowlands is apparently due to the poor susceptibility of Culex neavei to infection.67 Culex neavei replaces Cx. univittatus allopatrically in the KwaZulu-Natal lowlands, where it is apparently the main vector.54, 100, 157 The lack of isolations of WNV from Cx. pipiens group mosquitoes in southern Africa, unlike its proven role as a major vector in Europe and the New World ( see West Nile virus infection), is interesting.26, 125, 126 This may be related to the fact that South African Cx. pipiens and Cx. quinquefasciatus are for genetic reasons refractory to WNV infection. There is growing evidence suggesting that Cx. pipiens from South Africa be considered a separate species proposed to be named Culex juppi but for now is considered a nomen nudum because there is no official morphological description for this “new species.”22 Numerous isolations of SINV have also been made from Cx. univittatus and a few from Cx. pipiens and Cx. theileri in the inland plateau region of South Africa.64, 67, 102 Highveld Cx. pipiens 19 feeds predominantly on birds in southern Africa and so is less likely to transmit the viruses to humans than Cx. univittatus, which feeds on humans more frequently. West Nile virus and SINV have also been isolated, albeit rarely, from Cx. theileri, another species often collected in highveld grassland and Cape fynbos regions that feeds on birds and humans and can be experimentally infected with these viruses.58 The reason for the much lower infection rates of wild Cx. theileri than Cx. univittatus remains unknown, although it has been postulated that this is because that Cx. theileri possibly feeds on bird species less susceptible to infection with these viruses. Because Cx. univittatus and Cx. theileri blood feed on birds and humans, they serve as bridge vectors for bird-host zoonotic viruses.
As shown in Table 4, 43 isolations of WESV have been made from mosquitoes collected in South Africa and 15 from mosquitoes collected in Zimbabwe. Apart from two species, all these isolations were made from floodwater Aedine mosquitoes (Table 2). The most important Aedes spp. involved with WESV were Ae. (Neo.) circumluteolus (KwaZulu-Natal lowlands), Ae. (Och.) caballus/ juppi (inland plateau), Ae. (Neo.) mcintoshi/luridus (inland plateau) and Ae. (Neo.) mcintoshi (Zimbabwe highlands).
Table 4 Number of isolations of Wesselsbron virus from mosquitoes collected in southern Africa
| REGION | SPECIES | NO. ISOLATIONS | 50% INFECTION THRESHOLDa LOG10LD50/ml |
|---|---|---|---|
| South Africa: | |||
| KwaZulu-Natal coastal lowlands | Ae. (Neo.) circumluteolus | 13 | 4, 103, 137, 157 |
| Ae. (Adm.) minutus | 1 | 157 | |
| Ae. (Adm.) veeniae | 1 | 8 | |
| Mansonia (Mnd.) uniformis | 1 | 78, 157 | |
| Cx. (Cux.) neavei a | 1 | 157 | |
| South Africa: | |||
| Inland plateau | Ae. (Neo.) mcintoshi b/albothorax c | 3 | 78 |
| Ae. (Neo) mcintoshi/luridus | 5 | 63 | |
| Ae. (Och.) caballus s.l.d | 13 | 78 | |
| Ae. (Och.) juppi/caballus | 5 | 63 | |
| Zimbabwe highlands | Ae. (Neo.) mcintoshi b | 15 | 6, 91 |
a Reported as Cx. (Cux.) univittatus
b Reported as Ae. (Neo.) lineatopennis
c Probably Ae. (Neo.) luridus
d Could be Ae. (Och) caballus s.s. or Ae. (Och) juppi
Isolations of multiple other viruses such as MIDV, Ndumu (NDUV), Bunyamwera (BUNV), Spondweni (SPOV) and Pongola (PGAV) viruses have been obtained from floodwater Aedes species. Floodwater Aedes, however, do not mate in insectary conditions, preventing them from forming colonies, and the few vector competence assays performed on them have been done on individuals reared from field collected larvae, which is not ideal. So for now these isolations provide tentative evidence for their role as vectors.
Banzi virus has been isolated almost exclusively from Culex (Eumelanomyia) rubinotus in Harare in Zimbabwe and in Port Shepstone, Empangeni and Mtubatuba on the KwaZulu-Natal coast of South Africa. Infection rates in this species have often been high.104 Culex theileri has been implicated as a potential vector for several bunyaviruses148 and may be a vector of SHUNV virus but this will obviously require further clarification, especially as the evidence of Culicoides biting midges as vectors of SHUNV seems more compelling.114, 143
There are two species of Mansonia in Africa and they are both found close to edges of permanent to semi-permanent waters that support vegetation to which the immatures attach to breathe. In some locations, they reach high numbers and blood-feed opportunistically and consequently have opportunities to imbibe virus from multiple vertebrates. Attempts to establish colonies of African Mansonia have been unsuccessful and until more convincing vector competence trials have been performed the role of Mansonia as arbovirus vectors should be viewed with some caution.
Vector competence
Values for the 50 per cent infection threshold and the transmission rate determined in horizontal biological transmission tests for six species of mosquito infected with RVFV are given in Table 3. Data indicating the approximate length of the extrinsic incubation period for each species are also shown. On the basis of these results the order of vectorial importance is Cx. theileri, Ae. circumluteolus, Cx. zombaensis, Ae. mcintoshi and Ae. juppi. Table 6gives the results for preliminary tests on three more mosquito species infected with RVFV, all of which had high infection rates after feeds of high titres of virus, and which subsequently transmitted the virus. Comparison of the results for Aedes unidentatus (Table 6) with those for Ae. mcintoshi shows that Ae. unidentatus is a more efficient vector.
Field studies during RVF epidemics have shown only low infection rates in the mosquito vectors, often no more than one infected mosquito per 1 000.89, 103 In addition, high vector competence evidently requires high levels of viraemia in the vertebrate host.62, 98, 103 Despite this, a striking feature of RVF epidemics among sheep on the inland plateau of South Africa is the rapidity with which infection spreads through flocks, causing heavy losses. To account for the high level of infection, biological infection by mosquitoes is almost certainly augmented by mechanical transmission by various biting flies.
Table 6 Results of vector competence tests with Rift Valley fever virus and three mosquito species; infective feeds on viraemic hamsters but transmission by the in vitro capillary method Jupp Cornel 1988
| SPECIES | TITRE OF INFECTIVE FEED IN LOG10CPD50 PFU/mlc | MOSQUITO INFECTION | TRANSMISSION | ||
|---|---|---|---|---|---|
| DAYS AFTER INFECTIVE MEAL | RATEa | DAYS AFTER INFECTIVE MEAL | RATEb | ||
| Ae. (Neo.) unidentatus | 6,8–7,8 | 11 | 12/14 (86%) | 11 | 7/12 (58%) |
| Ae. (Adm.) dentatus | 6,8–7,8 | 11 | 28/31 (90%) | 11 | 9/28 (32%) |
| 17 | 6/8 (75%) | 18 | 3/6 (50%) | ||
| Cx. (Cux.) poicilipes | 7,8 | 15 | 26/29 (90%) | 15 | 4/26 (15%) |
| 30 | 24/30 (80%) | ||||
a Numerator = no. mosquitoes infected; denominator = no. mosquitoes tested
b Numerator = no. mosquitoes transmitting; denominator = no. infected mosquitoes feeding
c Cytopathic dose (CPD); Plaque forming units (PFU)
In mechanical transmission there is no viral replication in the vector, only a passive transfer of virus. Experimental mechanical transmission of RVFV has been accomplished by various haematophagous Diptera: Glossina morsitans, Stomoxys calcitrans, Lutzomyia longipalpis, Culicoides variipennis, and several species of Aedes and Culex.42, 70 In interrupted transmission tests from viraemic to susceptible hamsters by the bite of Aedes aegypti formosus,70 transmission was achieved by groups of mosquitoes for up to 25 minutes after the infective feed but not after 5.5 hours. After shorter intervals of 3 to 5 minutes, single mosquitoes were also shown to transmit the virus on three occasions.
The results of earlier vector competence tests with WESV are shown in Table 7. They indicate that Cx. theileri can be excluded as a vector, while Ae. circumluteolus, Ae. caballus s.l. and Cx. zombaensis are potential vectors.
Table 7 Results of vector competence tests with various mosquito species and WESV virus
| SPECIES | VIRAEMIC INFECTING HOST | BLOODMEAL TITRE LOG10LD50/ml | INFECTION RATE | SUSCEPTIBLE HOST/S | TRANSMISSION ATTEMPTS | REFERENCE |
|---|---|---|---|---|---|---|
| Ae. (Neo.) circumluteolus | Artificial | 7,2 | 7/9 | Mice | 16/18a | 119 |
| Ae. (Och.) caballus s.l. | Mosquitoes collected in epidemic | NT | Lambs | 1/1a | 75 | |
| Ae. (Och.) caballus s.l. | Mosquitoes collected in epidemic | NT | Mice | 1/31a | 57 | |
| Ae. (Och.) caballus s.l. | Lambs | NT | NT | Mice | 2/2a | 75 |
| Cx. (Cux.) zombaensis | Hamsters | 8,4 | 13/26 | Hamsters | 1/2b | 7 |
| Cx. (Cux.) theileri | Hamsters | 6,7–7,2 | 21/52 | Mice | 0/15 | 7 |
a Numerator = no. successful transmissions; denominator = no. attempts with groups of mosquitoes
b Transmission rates
NT Not tested
Experimental vector competency evaluations of common Culex mosquito species Cx. pipiens, Cx. univittatus and Cx. theileri showed that Cx. univittatus is the most efficient WNV and SINV vector, with a low 50 per cent infection threshold of about 2,3 log LD50/ml and a high transmission rate of 97 per cent.56, 58 Culex pipiens and Cx. theileri have similar infection dose thresholds as Cx. univittatus but have much lower WNV and SINV transmission rates (<25%), which would suggest that Cx. pipiens and Culex theileri are of lesser importance as vectors of these viruses.58 West Nile virus vector competency trials conducted on Cx. univittatus held at various temperatures, including fluctuating ambient highveld temperatures experienced in South Africa, revealed that once they reach the extrinsic incubation period they continue to transmit high doses until they die.17 Conversely, Cx. univittatus transmitted oscillating titres of SINV after reaching the extrinsic incubation period, especially when the mosquitoes were held in cages exposed to outside temperatures ranging from night-time lows of 14°C and day-time highs of 34°C.15 Dissections revealed that SINV, and not WNV, caused cytopathy of the salivary gland tissues and that SINV infected Cx. univittaus did not survive as long as WNV infected mosquitoes at all temperatures including ambient temperatures.15 This shows that under natural South African highveld temperature conditions Cx. univittatus is not as efficient a vector of SINV as WNV. Furthermore, it was shown that WNV infected Cx. univittatus kept for 49 days at 14°C were capable of infecting birds the day after they were transferred to 26°C.15 Culex univittatus adult females survived up to 114 days at 14°C, which further supports the notion of overwintering of WNV in quiescent adult females. Although not done on African Culex, it has been proven that WNV can be vertically transmitted in Cx. pipiens/quinquefasciatus hybrids, a major vector of WNV in North America,86 and it would be interesting to perform vertical WNV trials with Cx univittatus.
Owing to the high infection rates of BANV in natural populations of Cx. rubinotus at Port Shepstone,South Africa, it was possible to conduct a transmission experiment in the field in which individual hamsters were exposed to 16 groups of between 4 and 19 wild-caught mosquitoes. Banzi virus isolates were made from the mosquito groups that had fed on 11 out of 16 hamsters, which suggested a high degree of vector efficiency.104
Ecology of arbovirus mosquito vectors of veterinary importance
Brief notes on the ecology and distributions are provided in this section of the mosquito species most commonly mentioned and proven to act as major and minor arboviral vectors in southern Africa.
Aedes (Neo.) circumluteolus
Aedes circumluteolus (a savannah group member of subgenus Neomelaniconion) is widespread in Africa.25, 44 It is the predominant member of Neomelaniconion in the tropical coastal lowlands from northern KwaZulu-Natal in South Africa northwards into Mocambique. In Tongaland in northern KwaZulu-Natal it is often one of the most prevalent of all mosquitoes during the summer.20, 65, 88, 99, 100, 157 It has been collected in much smaller numbers in other parts of northern KwaZulu-Natal at river margins or in marshes after rain, although it can apparently become quite prevalent at Lake St Lucia in South Africa.23 Aedes circumluteolus also commonly occurs in savannah biomes in Angola.132 It is a floodwater species that oviposits at the edge of rivers and pans within the flood plain. Aedine eggs are resistant to desiccation as long as a high level of humidity is maintained within their microhabitat under the soil where, like Ae. juppi, they are probably deposited within the top 50 mm.28
Aedes circumluteolus eggs do not hatch until they are inundated at the next river flooding. Local rainfall apparently has to be heavy and concentrated to cause surface flooding similar to that associated with river flooding.65
A study at Ndumu in KwaZulu-Natal in South Africa showed that during the dry winter and spring months (June to October) Ae. circumluteolus declined to very low numbers, but it never completely disappeared. Blood-feeding and ovarian development occurred throughout the year.65 Aedes circumluteolus is a ground feeder foraging during the day and also at night from about half an hour after sunset. It feeds opportunistically on mammals such cattle, sheep, antelopes and humans.65, 100, 124
Aedes (Neo.) unidentatus and Aedes (Neo.) mcintoshi
In 1971 McIntosh revised the subgenus Neomelaniconion in southern Africa.88 He named two new species, Ae. unidentatus and Ae. luridus, making a total of eight species including Ae. circumluteolus and Ae. lineatopennis. Prior to this revision Ae. unidentatus may for example have been called Ae. lineatopennis in Johannesburg and at Lake Chrissie,64 while Ae. luridus may have been confused with Ae. albothorax. In 1985 Huang47 described a new species, Ae. mcintoshi, and showed that African mosquitoes previously reported as Ae. lineatopennis really belonged to this new species.47 Aedes lineatopennis itself was shown to be confined to the Philippines. Thus, reports of Ae. lineatopennis in the literature after 1971 refer to Ae. mcintoshi but before 1971 could have been either Ae. unidentatus or Ae. mcintoshi.
Large numbers of Ae. mcintoshi have recently been collected at Villiers in the north-eastern highveld, where it was about as common as Ae. unidentatus. In Rooipoort (Northern Cape Province,South Africa) Ae. mcintoshi represented 7 per cent of mosquito numbers and Ae. unidentatus was not collected.18 In southern Africa, the distribution of Ae. mcintoshi indicates that it is probably best adapted to regions of intermediate temperature. In fact, of all the “Savanna” group Neomelaniconion species, Ae. mcintoshi is the most widespread in southern and eastern Africa and was formally named Aedes lineatopennis.25
Aedes unidentatus is probably more common in the moister eastern part of the highveld and the middle veld of the Free State Province, where it is probably the numerically dominant species of the subgenus.88 Aedes mcintoshi is a more widely distributed species adapted to regions with markedly different climates: it occurs in the Karoo, highveld, Limpopo–Mpumalanga lowveld, and the KwaZulu-Natal coastal lowlands of South Africa, and in the Zimbabwe highlands. In the northern part of the KwaZulu-Natal lowlands this species appears to be dominated by Ae. circumluteolus. Its prevalence relative to Ae. unidentatus and Ae. luridus in the highveld and Karoo respectively is not yet properly known because of insufficient sampling.
Aedes unidentatus feeds both by day and night, with peak biting commencing about half an hour after sunset in Johannesburg. It feeds voraciously on goats, cattle, sheep and humans.2, 64, 88 Aedes mcintoshi has the same host preferences but its peak biting in the eastern highveld at Bultfontein in the Free State Province was from late afternoon until sunset, with a lesser peak at dawn.63
Larvae of Ae. unidentatus have been collected near Johannesburg in temporary rainwater pools in flooded pasture.52 Both Ae. unidentatus and Ae. mcintoshi larvae occur in large numbers in flooded pans and, to a lesser extent, in vleis in South Africa.3
Aedes (Och.) juppi and Aedes (Och.) caballus
McIntosh made a taxonomic reassessment of Aedes caballus and found that mosquitoes that had been identified as this species before 1973 could have been either Ae. caballus s.s. or a new species, Ae. juppi, or a mixture of both these species.90 From the limited number of locality records available for these two90 it appears that Ae. juppi is adapted to temperate conditions and is endemic to the temperate regions of southern Africa. Aedes juppi probably outnumbers Ae. caballus in the highveld and seems to be the only species in the western and coastal regions of the Western Cape Province, and represented 44 per cent of mosquitoes collected in Rooipoort (Northern Cape Province-South Africa).18 Both species are sympatric over a large area, including the Karoo and southern Free State, but the relative proportions of the two species require further elucidation. Aedes caballus is apparently adapted to higher temperatures and a lower rainfall than Ae. juppi and is absent from the coastal regions of KwaZulu-Natal and Mozambique.
Aedes juppi and Ae. caballus have very similar ecologies. Both feed avidly in the light and just before and after sunrise on goats, sheep, cattle and humans.62, 68, 103 In the eastern highveld Ae. juppi has its biting peak from late afternoon until sunset with a lesser peak at dawn.63 Larvae of Ae. caballus s.l. have been recorded from small or large temporary pools or pans.118 More recent studies have shown that the eggs of both species are laid in sedge or grassland in low-lying depressions (pans) and hatch to produce enormous numbers of larvae when these pans fill after heavy rainfall.28, 60
Species of the subgenera Ochlerotatus and Neomelaniconion of Aedes are found in similar habitats and often collected together at the same aquatic site on the inland plateau especially in temporary pools overgrown with sedges. Recent work at Bultfontein in the eastern highveld has shown that in the case of eggs of Ae. juppi and Ae. mcintoshi in pans, the development cycle from egg to adult can be as short as five days after inundation.63
Culex (Cux.) univittatus
It feeds nocturnally, particularly in the tree canopy but also at ground level. While this species is only slightly anthropophilic, it has been shown to feed on humans indoors to some extent (endophagy).55
Epidemics of WNV infection are probably partly a result of increased feeding on humans when particularly high densities of Cx. univittatus occur, as they did at Upington in northern Cape Province in South Africa in 1974.104 Precipitin tests on blood meals of Cx. univittatus collected on a cattle farm near Johannesburg showed that 37 per cent of them were of mammalian origin, and 55 per cent of these mammalian meals were from cattle. Culex univittatus will utilize temporary, semi-permanent and permanent ground pools as larval habitats, although it prefers sites with clear water and emergent vegetation.44, 52 Reed beds at the margins of the Gariep (formerly Orange) river were probably utilized at Upington in 1974 and 1976 when densities of Cx. univittatus were high. It appears to be important for Cx. univittatus adults and larvae to enter a state of quiescence to overwinter, although the available evidence indicates that adults pass the winter in a nulliparous rather than a parous condition.53, 57
Culex (Cux.) zombaensis
Culex zombaensis seems more selective in its choice of larval habitat than Cx. theileri and is more confined to cleaner water with emergent vegetation, such as river margins and permanent pans and vernal pools. Culex zombaensis is mainly a ground frequenting species that feeds nocturnally on cattle, sheep, goats, antelope and humans.100, 105, 124
Culex (Cux.) theileri
This species is moderately ornithophilic, blood feeding on birds more at ground level than in the tree canopies. However, it has been recorded blood feeding on a wide range of other hosts such as goats, cattle, sheep and humans and serves as an arboviral bridge vector.2, 55, 64, 68
Culex (Eum.) rubinotus
Culex rubinotus can be collected in reasonable numbers only within or near its preferred habitat. This is dense marshland vegetation, always associated with permanent water such as lagoons, rivers or streams, and dams or lakes. Rank reeds, papyrus or grass are always present but trees are either few or absent. Although this type of habitat occurs on the inland plateau, it is particularly common on the KwaZulu-Natal coast, and large populations of the mosquito have been sampled at Mtubatuba and Empangeni.8Culex rubinotus has also frequently been collected in the western and eastern Cape,118 Gauteng and Limpopo Provinces in South Africa,76 as well in Mocambique and Zimbabwe,67, 93, 106 and has a scant distribution in Angola.131 The mosquito more often than not seeks out dense vegetation because of its sensitivity to wind and low humidity. Large numbers have also been taken in mature sugar-cane fields when these adjoined the natural habitat.66
Culex rubinotus feeds primarily on rodents, to a moderate degree on domestic ungulates and rarely on humans. [66] The feeding rate is high on hamsters in mosquito traps placed on the ground. Female mosquitoes confine their activity almost entirely to ground level and rarely enter traps in the tree canopy.66
Larvae of Cx. rubinotus have been collected in reed beds in shallow water.44, 66 The larval habitats have been recorded as being papyrus swamps and weedy or Pistia-covered borrow-pits.
Viral maintenance and occurrence of epidemics of disease
Records of rainfall in the summer rainfall region of South Africa since 1910 reveal a 20-year oscillation between decades of above and below average rainfall.141 The 1950s was a decade of high average rainfall, the 1960s below average and the 1970s again above average. During the period 1957 to 1979, the major outbreaks of WN, CHIK and RVF virus infections in humans and domestic animals occurred mainly during periods of above average rainfall. More recent outbreaks of RVF in South Africa also coincided with high and widespread rainfall in 2010 and 2011110 with small more geographically restricted waves of virus transmission in 2008, and two in 2009 that were seemingly also triggered by local heavy rainfall and pastoral flood irrigational practices.111 Febrile human cases in Mocambique also occurred after high rainfall and flooding in 2013.36 Multiple risk factors influence the extent (both in time and space) of RVF outbreaks that also extend beyond the vector itself and these include temperature, immune status of host animals (herd immunity influenced by previous outbreaks and vaccination) and vegetation density.111 Increased vegetation provides more shaded resting sites from higher temperatures that improves mosquito survival through the virus extrinsic incubation period.
During times of heavy rain certain parts of the inland plateau in South Africa, notably the panveld area of the western Free State and northern Eastern Cape Provinces, may become so saturated that large pans and areas of pasture are flooded. Such accumulations of water may remain standing on farms for several months, particularly if there is a granite underlay as in the case of the Kaap Plateau. Under these conditions large populations of floodwater Aedes mosquitoes are produced initially, which are subsequently replaced by equally large populations of Culex mosquitoes. At such times epidemics of RVF may occur, but the extremely high densities of mosquitoes and other biting flies are also detrimental to livestock in other ways. These include reduced milk production by dairy cows and the development of a poor general state of health in sheep and cattle. Sheep may even die from loss of blood as a result of the continual biting by vast numbers of insects.
Rainfall trends from 1921 to 2015 indicate an increase in rainfall in the southern interior and west to north-west and decrease in the far northern and north–eastern parts of South Africa.81 As has been observed as a trend globally, short spell intense rainfall events are becoming more frequent, whereas longer term wet spells (soft rain for several hours) especially in the east and north- eastern parts of South Africa are decreasing. How this change in rainfall pattern will influence vector mosquito prevalence and density and virus abundance and distribution will be of interest. Evidence has, however, been obtained in southern Africa and Egypt that transmission by various mosquito species, notably Culex spp., occurs during epidemics in sheep and cattle.31, 43, 103, 105
Culex theileri and Cx. zombaensis are the important species on the inland plateau and KwaZulu-Natal coastal lowlands of South Africa, respectively. There is strong evidence that these species transmit RVFV biologically between sheep and cattle, and that mechanical transmission by several mosquito species and other biting flies is responsible for the maintenance of the epidemic. Humans are also thought to become infected on occasion through transmission by Cx. theileri and Cx. zombaensis, although most human infections during epidemics are undoubtedly caused by contagion during the slaughter of infected animals.
In South Africa, transovarial transmission tests and testing of mosquitoes reared from field-collected eggs have been undertaken to try to incriminate various floodwater Aedes spp. as reservoir vectors of RVFV.28, 62, 103 Such studies have so far been directed mainly at A. juppi and have indicated that this species has a low vector potential, that it is only a subsidiary epidemic vector, and that it is not involved in vertical transmission of the virus. Of the Aedes species, Ae. mcintoshi, Ae. circumluteolus and Ae.caballus have the widest distributions and occur in other southern African countries such as Mocambique, Namibia, Botswana and Zimbabwe. These species, therefore, may be responsible for transmission of RVFV in these countries as well. Based on seroprevalence studies in animals and humans, RVFV occurs more commonly than originally thought, often because of misdiagnosis.12, 21, 27, 36, 48, 115, 116, 121
The paucity of isolations from floodwater Aedes during epidemics is partly because sampling of the mosquito populations for virus assay invariably began late in the course of an epidemic. Despite this, isolations have been made from Ae. mcintoshi and Ae. juppi on the inland plateau and from Ae. circumluteolus and Ae.durbanensis on the KwaZulu-Natal coastal lowlands. Although not proven one or more of these species may be responsible for transovarial transmission as has been proposed for Ae. mcintoshi in Kenya.82 Theoretically it might also be possible for some infected Culex spp. to overwinter and initiate transmission the next spring, especially as small numbers of Cx. theileri adults have been collected in the winter in Johannesburg.53, 57
Abortion as a result of RVF was reported in the late 1990s in African buffalo (Syncerus caffer) that were kept in a boma at Skukuza in the Kruger National Park, Mpumalanga, South Africa.33 Retrospective serological testing of buffalo from the Park and cattle sera from other parts of South Africa and Mocambique indicated that RVFV can circulate silently in more resistant species such as wildlife and cattle at times when no epidemics were occurring in livestock.45 This suggests that RVFV may be endemic in certain areas where climatological and other factors favour the breeding and maintenance of mosquito and vector populations.
Serological surveys in humans and livestock showed that WESV is more common in the moist tropical part of southern Africa such as northern coastal KwaZulu-Natal in South Africa, Mocambique and Namibia than on the inland plateau, the southern KwaZulu-Natal lowlands and the Cape Provinces of South Africa.14, 76, 77, 94, 138 Sequence analysis of isolates from two humans during an outbreak of WESV in 2010 and 2011 and other GenBank deposited sequences suggest that there are two WESV clades with one more widespread in South Africa and Zimbabwe and another more restricted to the KwaZulu-Natal region.14 On the temperate South African plateau the Ae. caballus/juppi and Ae. mcintoshi-luridus-unidentatus groups of mosquitoes are vectors of WESV. Along the tropical, eastern coastal plain of northern KwaZulu-Natal and Mocambique, Ae. circumluteolus is the most likely vector. Because the tropical areas of northern KwaZulu-Natal and Mocambique receive a higher rainfall, and are therefore moister than the drier temperate areas of South Africa, Aedes spp. are probably active there over longer periods. This would account for the higher level of WESV activity in the tropical areas.91
The only evidence for the possible existence of a wild vertebrate host for WESV is the single isolation of virus from a gerbil (Desmodillus auriculatus).5 Only one unsuccessful attempt has so far been made to show the presence of infected floodwater Aedes mosquitoes among those emerging from an inundated pan.63 Domestic ungulates are important incidental hosts with which the vector could probably maintain the virus for quite long periods in the absence of a primary vertebrate maintenance host. Livestock, particularly adult sheep and cattle, are often asymptomatic (see Wesselsbron disease). Humans can be infected either by contagion through handling infected carcasses or by mosquito bite.
During summer on the inland plateau of South Africa, WNV and SINV are maintained in a sylvatic cycle between Cx. univittatus and Cx. theileri and various avian species. The same mosquitoes, mostly Cx. univittatus, also infect humans.56, 61, 64, 95, 101, 102 The exposure of sentinel pigeons at Bethulie in the Free State Province indicated the occurrence of such epidemics in April 1970 and April 1971.96 Serological surveys for neutralizing antibodies in livestock showed prevalences of 16 per cent for WNV in sheep and cattle on the highveld generally, and rates of 21 to 47 per cent in older cattle, oxen and horses in Johannesburg.79, 94 The high prevalence of antibodies in horses proves that Cx univittatus also feeds extensively on horses and because of neurological consequences and even death149, 150, 151 a WNV vaccine regimen (see West Nile virus infections) for veterinary use in horses and possibly valuable wild animals is recommended.127, 156 Viraemia with WNV and SINV in vertebrates other than birds is too low to infect mosquitoes and they are considered dead end hosts. These viruses amplify to higher levels in birds, sufficient to infect mosquitoes, which then pass on the virus to other vertebrates in subsequent blood-feeding after the extrinsic incubation period has passed. Consequently, epidemics in horses and humans of WNV and to a lesser extent SINV are usually associated with concurrent epidemics in birds.13
Culex rubinotus maintains BANV among wild rodents. The virus has been isolated and antibodies have been detected in Mastomys natalensis.104 Wild rodents inoculated with the H336 strain became viraemic.83
The occurrence of Cx. rubinotus in other parts of Africa, with associated virus isolations, suggests that it is probably the vector of BANV over a large part of the continent.40, 112 Serological surveys in the KwaZulu-Natal coastal lowlands showed that neutralizing antibodies were present in cattle at rates of 2 to 16 per cent and in sheep at rates of 0 to 7 per cent.77 This evidence, together with the moderate feeding of Cx. rubinotus on domestic ungulates, suggests that pregnant cattle, for example, could become infected, particularly if they were to browse near the marshy reed beds where Cx. rubinotus is prevalent. It would seem worthwhile to examine foetal material arising from natural abortions on the KwaZulu-Natal coast for the presence of the virus and developmental abnormalities. Whether bovine infections ever attain the level of an epidemic is not known. Banzi virus has been isolated from humans and is known to cause at least mild illness,138 but the extent to which it causes morbidity in humans is uncertain.
The maintenance and mosquito derived transmission of other less common viruses that are now known to infect livestock and wildlife in southern Africa remains very enigmatic. These include MIDV, NDUV, USUV, and SHUNV which, based on seroprevalence data, are quite widespread in southern Africa. For example, and most unusual for an alphavirus, NDUV was isolated from larvae of Cx. pipiens in Kenya, suggesting vertical transmission as a means of overwintering.84 Isolation of USUV virus from a pool of male Mansonia uniformis in Uganda may also suggest a vertical transmission pathway for this virus.120 Tissue samples from acutely sick horses positive for MIDV and SHUNV between 2008 and 201313 indicated that these viruses and likely others such as NDUV, SPOV and USUV are not necessarily associated with outbreaks and that yearly sporadic cases usually occur.
Not much is known about the natural cycles of several Bunyaviridae (orthobunyaviruses) such as BUNV, Bwamba (BWAV), Lumbo (LUMV), Witwatersrand (WITV), Pongola (PGAV) and Germiston (GERV) that infect humans, sometimes with severe health consequences and even death.34 Collectively, these viruses are not sufficiently recognized and understood in spite of high seroprevalence rates in humans. The association with mainly self-limiting, non-specific febrile illness in humans is likely the cause for under-reporting.34 BUNV, GERV and WITV have been isolated from a variety of mosquito species in different habitats but mostly from Cx. rubinotus in the temperate plateau and coastal lowlands of South Africa76, 104, 107 and in Zimbabwe.93, 106 Further isolations of these three viruses from sentinel and wild rodents in the inland plateau and eastern coastal South Africa and in central Mozambique93, 106 and human seroprevalance in northern Namibia80, 122 and the Okavango basin in Botswana80 confirm their wide distribution in southern Africa and that rodents are the most likely vertebrate reservoirs for BUNV, GERV and WITV. 93, 106 Neutralizing antibodies to BUNV and PGAV have been detected in cattle, sheep and goats in eastern South Africa80 but their role as reservoir or amplifying hosts is unknown. Lumbo virus may be more restricted to the eastern tropical coast of southern Africa near salt or brackish marshes. This virus has only been isolated from several pools in consecutive years from one mosquito species, namely Ae. pembaensis, which is restricted to breeding in the dwelling holes of brackish water crabs.25 Aedes pembaensis avidly bites people indoors and outdoors25 and neutralizing antibodies to LUMV have been found in humans in Lumbo in Mozambique and in northern KwaZulu-Natal of South Africa73 where these mosquitoes occur. During the virus surveys performed in Lumbo in the late 1950s and early 1960s another virus called Mosssuril was isolated from a pool of Culex with white-ringed proboscides probably consisting of Culex sitiens but possibly also Culex thalassius and Culex tritaeniorhynchus.74
As mentioned earlier, we currently owe rekindling of mosquito arboviral research and interest to multi-continental re-emergence of four arboviruses, CHIKV, ZIKV, YFV and DENV which, although they are of no veterinary importance, are all transmitted by the globally distributed mosquito Aedes aegypti and to lesser extent Ae. albopictus/albopicta. For this reason, we consider it valuable to devote some attention to them. Although CHIKV was traditionally considered to be a sylvatic endemic virus in wooded areas in Africa because of cycling between multiple forest dwelling type Aedes and small mammals (including monkeys), it has emerged as a major global infection.13 Spread of this virus out of Africa was likely facilitated by a viral mutation that increased its vector competence for Ae. albopictus and in global urbanization of Aedes aegypti and global spread of Ae. albopictus1, 32. Proof of infected persons and sporadic outbreaks of this virus have been recorded multiple times in the more subtropical areas in South Africa,14, 30 once in Zimbabwe133 and by circulation based on human seroprevalence in Mocambique.1 Proof of introduction of Ae. albopictus from Asia has been known since the early 1990s16 and it was only a matter of time before this mosquito became established in Africa130 including Mocambique.1 There is no evidence of established Ae. albopictus populations in South Africa but eventual establishment is likely as populations spread southwards from Mocambique. Aedes aegypti is a common domestic mosquito in eastern coastal cities of South Africa and is now abundant in Pretoria. Interestingly, it is also abundant and a profound biting nuisance in isolated reserve camp sites and villages in the dry Kalahari sands regions of South Africa.18 Given this abundance of Ae. aegypti, it is predicted that a significant outbreak of CHIKV in southern Africa is inevitable. No vector competence studies have been performed on southern African Ae. aegypti populations for CHIKV or ZIKV but it has been shown that Ae. aegypti is more vector competent for African than for American strains of ZIKV.71 Aedes aegypti is considered the major vector of ZIKV and an outbreak of this virus in southern Africa is also not out of the question. The occurrence of SPOV, closely related to ZIKV, known to cause serious illness in humans can easily be confused with ZIKV and is possibly considerably underestimated in Africa.146 It is likely also quite contagious as two personnel members handling infected mosquitoes and mice tissue got ill.87 Isolations of SPOV have been obtained from multiple species of mosquitoes in South Africa108 and follow up studies are warranted especially in light of the emergence of ZIKV, its serological cross- reactivity with ZIKV and that SPOV was recently isolated from a pool of Culex quiqnqufasciatus in Haiti.153 SPOV is, however, sufficiently different from ZIKV to render Ae. aegypti and Ae. albopictus refractory to infection with it.38
Mosquito control
To the best of our knowledge there is no organized (government) control of any arboviral vector mosquito species in southern Africa. All organized mosquito control efforts are directed towards Anopheles malaria vectors.
From the point of view of controlling RVFV, WNV and other arboviruses, prior immunization of livestock would be more cost-effective than vector control. In the event of impending current epidemic situations it may be advisable to institute some mosquito control measures to reduce the degree of exposure of stock to mosquito bites. Since the peak period for biting is from just before sunset until sunrise, and because mosquitoes are always active at or close to their breeding sites, stock should, if possible, be moved away from pans and vleis to higher ground before nightfall. In the case of dairy farms, the milking sheds should be made mosquito proof, and perhaps Culicoides-proof, and monthly spraying of their interior walls should be carried out with a residual insecticide other than an organochlorine. Organophosphate insecticides, such as malathion, fenthion (Baytex), fenitrothion (Sumithion), or carbamates such as carbaryl (Sevin) and propoxur (Arprocarb), can be used at rates of 1,5 to 2,0 g/m2.135
Control of adult mosquitoes outdoors would probably be neither effective nor economical. Outside control could, however, be directed at larvae in the main breeding pans, vleis and dams on smallholdings and farms. Larvicides such as Temephos (Abate) can be incorporated into sand granules or pellets and sprinkled into the breeding place. In the case of pans, a dry pan could be seeded with such pellets just before a flood is expected so that the insecticide is released into the water as the aedine eggs hatch. As an alternative larvicide, spores of the bacteria Bacillus thuringiensis israelensis and Lysinibacillus sphaericus (formerly, Bacillus sphaericus) can be incorporated into pellets. This may become practicable if the present high cost is reduced. Application of slow release pellets of insect growth regulators with active ingredients containing methoprene and pyriproxyfen, that inhibit mosquito immature stage development, also offer control possibilities for use in locations of intense domestic Culex and Aedes breeding in urban environments.109, 134 The emphasis should be on a control programme with measures that are selective, integrated and environmentally sound.140
References
- ABILIO, A. P., ABUDIASSE, G., KAMPANGO, A., CANDRINHO, B., SITOI, S., LUCIANO, J., TEMBISE, D., SIBINDY, S., DE ALMEIDA, A. P. G., GARCIA, G. A., DAVID, W. R., DE FREITAS, R. M. & GUDO, E. S., 2018. Distribution and breeding sites of Aedes aegypti and Aedes albopictus in 32 urban/periurban districts of Mozambique: implication for assesing the risk of arbovirus outbreaks. PLoS Neglected Tropical Diseases, 12, e0006692. doi: https:.//doi.org/10.1371/journal.pntd.0006692.
- ANDERSON, D., 1967. Ecological studies on Sindbis and West Nile viruses in South Africa. III. Host preferences of mosquitoes as determined by the precipitin test. South African Journal of Medical Sciences, 32, 34-39.
- ANON., 1966–1988. Arbovirus Unit, National Institute for Virology, Johannesburg. Unpublished data.
- ANON., 1967. Arbovirus Unit, National Institute for Virology, Johannesburg. Unpublished data.
- ANON., 1968. Arbovirus Unit, National Institute for Virology, Johannesburg. Unpublished data.
- ANON., 1969. Arbovirus Unit, National Institute for Virology, Johannesburg. Unpublished data.
- ANON., 1971. Arbovirus Unit, National Institute for Virology, Johannesburg. Unpublished data.
- ANON., 1980. Arbovirus Unit, National Institute for Virology, Johannesburg. Unpublished data.
- BARNARD, B. J. H. & VOGES, S. F., 1986. Flaviviruses in South Africa: Pathogenicity for sheep. Onderstepoort Journal of Veterinary Research, 53, 235–238.
- BEECHLER, B. R., BENGIS, R., SWANEPOEL, R., PAWESKA, J. T., KEMP, A., VAN VUREN, P. J., JOUBERT, J., EZENWA, V. O. & JOLLES, A. E., 2015. Rift Valley Fever in Kruger National Park: Do buffalo play a role in Inter-epizootic circulation of virus. Transboundary and Emerging Disease, 62, 24-32.
- BLACKBURN, N. K. & SWANEPOEL, R., 1980. An investigation of flavivirus infections of cattle in Zimbabwe Rhodesia with particular reference to Wesselsbron virus. Journal of Hygiene, 85, 1-33.
- BLOMSTROM, A. L., SCHARIN, I., STENBERG, H., FIGUEIREDO, J., NHAMBIRRE, O., ABILIO, A., BERG, M. & FAFETINE, J., 2016. Infection Ecology & Epidemiology, 6, 31343. doi: http://dx.doi.org/10.3402/iee.v6.31243.
- BRAACK, L. E. O., DE ALMEIDA, A. P. G., CORNEL, A. J., SWANEPOEL, R. & DE JAGER, C., 2018. Mosquito-borne arboviruses of African origin: review of key viruses and vectors. Parasite and Vectors, 11, 29. doi: 10.1186/s13071-017-2559-9.
- BURT, F. J., GOEDHALS, D. & MATHENGTHENG, L., 2014. Arbovirus in Southern Africa: are we missing something? Future Virology, 9, 993-1008.
- CORNEL, A. J., 1987. The effect of temperature on the vector competence of Culex univittatus Theobald (Diptera: Culicidae) for West Nile and Sindbis viruses. Masters Thesis, University of the Witwatersrand.
- CORNEL, A. J. & HUNT, R. H., 1991. Aedes albopictus in Africa? First records of live specimens in imported tires in Cape Town. Journal of the American Mosquito Control Association, 7, 107-108.
- CORNEL, A. J., JUPP, P. G. & BLACKBURN, N. K., 1993. Effect of environmental temperature on the vector competence of Culex univittatus (Diptera: Culicidae) for West Nile virus. Journal of Medical Entomology, 30, 449-456.
- CORNEL, A. J., LEE, Y., ALMEIDA, A. P. G., MOUATCHO, J., VENTER, M., DE JAGER, C. & BRAACK, L. E. O., 2018. Mosquito community composition in South Africa and some neighboring countries. Parasites & Vectors, 11, 331. doi: https://doi.org/10.1186/s13071-018.2824-6.
- CORNEL, A. J., MCABEE, R. D., RASGON, J., STANICH, M. A., SCOTT, T. W. & COETZEE, M., 2003. Differences in extent og genetic introgression between sympatric Culex pipiens and Culex quinquefasciatus (Diptera: Culicidae) in California and South Africa. Journal of Medical Entomology, 40, 36-51.
- DE MEILLON, B., PATERSON, H. E. & MUSPRATT, J., 1957. Studies on the arthropod-borne viruses of Tongaland. II. Notes on the more common mosquitoes. South African Journal of Medical Sciences, 22, 47-53.
- DONDONA, A. C., ASCHENBORN, O., PINONI, C., DI GIALLEONARDO, L., MASEKE, A., BORTONE, G., POLCI, A., SCACCHIA, M., MOLINI, U. & MONACO, F., 2016. Rift Valley Fever among wild ruminants, Etosha National Park, Namibia, 2011. Emerging Infectious Diseases, 22, 128-130.
- DUMAS, E., ATYAME, C. M., MALCOLM, C. A., LE GOFF, G., UNAL, S., MAKOUNDOU, P., PASTEUR, N., WEILL, M. & DURON, O., 2016. Molecular data reveal a cryptic species within the Culex pipiens mosquito complex. Insect Molecular Biology, 25, 800-809.
- ECKARD, D. M., 1984. National Institute for Tropical Diseases, Eshowe. Personal communication.
- ECKARD, D. M., WORTHINGTON, V. & JUPP, P. G., 1988. Mosquito species collected at Richards Bay, Natal. Journal of the Entomological Society of Southern Africa, 51, 140-143.
- EDWARDS, F. W., 1941. Mosquitoes of the Ethiopian region. III Culicine adults and pupae. British Museum (Natural History), London, 499
- ENGLER, E., SAVINI, G., PAPA, A., FIGUEROLA, J., GROSCHUP, M. H., KAMPEN, H., MEDLOCK, J., VAUX, A., WILSON, A. J., WERNER, D., JÖST, H., GOFFREDO, M., CAPELLI, G., FEDERICI, V., TONOLLA, M., PATOCCHI, N., FLACIO, E., PORTMANN, J., ROSSI-PEDRUZZI, A., MOURELATOS, S., RUIZ, S., VÁZQUEZ, A., CALZOLARI, M., BONILAURI, P., DOTTORI, M., SCHAFFNER, F., MATHIS, A. & JOHNSON, N., 2013. European surveillance for West Nile virus in mosquito populations. International Journal of Environmental Research for Public Health, 10, 4869-4895.
- FAFETINE, J. M., COETZEE, P., MUBEMBA, B., NHAMIRRE, O., NEVES, L., COSTZER, J. A. W. & VENTER, E. H., 2016. Rift Valley Fever outbreak in livestock, Mozambique, 2014. Emerging Infectious Diseases, 22, 2165-2167.
- GARGAN, T. P., JUPP, P. G. & NOVAK, R. J., 1988. Rift Valley Fever outbreak in livestock, Mozambique, 2014. Emerging Infectious Diseases, 22, 2165-2167.
- GARRET-JONES, C., 1964. Prognosis for the interruption of malaria transmission through assessment of a mosquito’s vectorial capacity.Nature, 204, 1173-1175.
- GEAR, J. & REID, F. P., 1957. The occurrence of a dengue-like fever in the North-Eastern Transvaal. I. Clinical features and isolations of virus. South African Medical Journal, 31, 253-257.
- GEAR, J. H. S., DE MEILLON, B., LE ROUX, A. F. & KOFSKY, R., 1955. Rift Valley fever in South Africa; a study on the 1953 outbreak in the Orange Free State with special reference to the vectors and possible reservoir hosts. South African Medical Journal, 29, 514-518.
- GOULD, E., PETTERSON, J., HIGGS, S., CHARREL, R. & DE LAMBALLERIE, X., 2017. Emerging arboviruses: Why today? One Health, 4 1-13.
- GROBLER, D. G. & BENGIS, R., 1999. National Parks Board and Department of Veterinary Services, Kruger National Park, Skukuza 1350. Unpublished data
- GROSETH, A., MAMPILLI, V., WEISAN, C., DAHLSTROM, E., PORCELLA, S. F., RUSSELL, B. J., TESH, R. B. & EBIHARA, H., 2014. Molecular characterization of human pathogenic Bunyaviruses of the Nyando and Bwamba/Pongola virus groups leads to the genetic identification of Mojuí dos Campos and Kaeng Khoi Virus. PLoS Negelected Tropical Diseases, 8, e3147. doi:10.1371/journal.pntd.0003147.
- GUDO, E. S., FALK, K. & CLIFF, J., 2018. Historical Perspective of Arboviruses in Mozambique and its implication for Current and Future Epidemics. Advances in Experimental Medical Biology, 1062, 11-18. doi: 10.1007/978-981-10-8727-1.
- GUDO, E. S., PINTO, G., WEYER, J., LE ROUX, C., MANDLAZE, A., JOSE, A. F., MUIANGA, A. & PAWESKA, J. T., 2016. Serological evidence of Rift Valley Fever virus among acute febrile patients in Southern Mozambique during and after the 2013 heavy rainfall and flooding: implication for management of febrile illness. Virology Journal, 1396. doi:10.1186/s12985-016-0542-2./.
- GUTHRIE, A. J., HOWELL, P. G., GARDNER, I. A., SWANEPOEL, R. E., NURTON, J. P., HARPER, C. K., PARDINI, A., GROENEWALD, D., VISAGE, C. W., HEDGES, J. F., BALASURIYA, U. B., CORNEL, A. J. & MACLACHLAN, N. J., 2003. West Nile virus infection of thorough-bred horse in South Africa (2000-2001). Equine Veterinary Journal, 35, 601-605.
- HADDOW, A. D., NASAR, F., GUZMAN, H., PONLAWAT, A., JARMAN, R. G., TESH, R. B. & WEAVER, S. C., 2016. Genetic characterization of Spondweni and Zika viruses and susceptibility of geographically distinct strains of Aedes aegypti, Aedes albopictus and Culex quinquefasciatus (Diptera: Culicidae) to Spondweni virus. PLoS Neglected Tropical Diseases, 10: e0005083. doi: http[://dx.doi.org/10.1371/journal.pntd.0005083.
- HARBACH, R. E., 2018. Culicipedia. Species-group, genus-group and family-group names in Culicidae (Diptera). Oxfordshire, UK, CABI.
- HENDERSON, B. E., MCCRAE, A. W. R., KIRYA, B. G., SENKUBUGE, Y. & SEMPALA, S. D. K., 1972. Arbovirus epizootics involving man, mosquitoes and vertebrates at Lunyo, Uganda 1968. Annals of Tropical Medicine and Parasitology, 66, 343-355.
- HENRICHS, B., OOSTHUIZEN, M. C., TROSKIE, M., GORSICH, E., GONDHALEKAR, C., BEECHLER, B. R., EZENWA, V. O. & JOLLES, A. E., 2016. Within guild co-infections influence parasite community membership: a longitudinal study in African Buffalo. Journal of Animal Ecology, 85(4), 1025-1034.
- HOCH, A. L., GARGAN, T. P. & BAILEY, C. L., 1985. Mechanical transmission of Rift Valley fever virus by hematophagous Diptera. American Journal of Tropical Medicine and Hygiene, 34, 188-193.
- HOOGSTRAAL, H., MEEGAN, J. M., KHALIL, G. M. & ADHAM, F. K., 1979. The Rift Valley fever epizootic in Egypt 1977–78. 2. Ecological and entomological studies. Transactions of the Royal Society of Tropical Medicine and Hygiene, 73, 624-629.
- HOPKINS, G. H. E., 1952. Mosquitoes of the Ethiopian Region. I. Larval bionomics of mosquitoes and taxonomy of culicine larvae. 2nd edn. London: British Museum (Natural History).
- HOWELL, P. G., 1999. Faculty of Veterinary Science, University of Pretoria, Onderstepoort 0110. Unpublished data.
- HUANG, Y. J. S., HIGGS, S. & VANLANDINGHAM, D. L., 2019. Emergence and re-mergence of mosquito-borne arboviruses. Current Opinion in Virology, 34, 104-109.
- HUANG, Y. M., 1985. A new African species of Aedes (Diptera: Culicidae). Mosquito Systematics, 17, 108-120.
- JORI, F., ALEXANDER, K. A., MOKOPASETSO, M., MUNSTERMANN, S., MOAGABO, K. & PAWESKA, J. T., 2015. Serological evidence of Rift Valley Fever virus circulation in domestic cattle and African buffalo in Northern Botswana (2010-2011). Froniters in Veterinary Science, 2, 63. doi:10.3389/fvets.2015.00063.
- JOUBERT, L., 1975. L’arbovirose West Nile, zoonose du midi méditerraneen de la France. Bulletin de l’Académie Nationale de Médicine, 159, 499-503.
- JOUBERT, L., OUDOR, J., HANNOVN, C., BEYTOUT, D., CORNIOU, B., GUILLON, J. C. & PANTHIER, R., 1970. Epidémioloqie du virus West Nile: étude d’un foyer en Camargue. IV. La méningo-encéphalomyélite du cheval. Annales Institute Pasteur, 118, 239-247.
- JUPILLE, M. H., SEIXAS, G., MOUSSON, L., SOUSA, C. A. & FAILLOUX, A. B., 2016. Zika virus, a new threat for Europe? PLoS Neglected Tropical Diseases, 10, e0004901. doi:10.1371/journal.pntd.0004901.
- JUPP, P. G., 1967. Larval habitats of culicine mosquitoes (Diptera: Culicidae) in a sewage effluent disposal area in the South African Highveld. Journal of the Entomological Society of Southern Africa, 30, 242-250.
- JUPP, P. G., 1969. Preliminary studies on the overwintering stages of Culex mosquitoes (Diptera: Culicidae) in the Highveld region of South Africa. Journal of the Entomological Society of Southern Africa, 32, 91-98.
- JUPP, P. G., 1971. The taxonomic status of Culex (Culex) univittatus in South Africa. Journal of the Entomological Society of Southern Africa, 34, 339-357.
- JUPP, P. G., 1973. Field studies on the feeding habits of mosquitoes in the Highveld region of South Africa. South African Journal of Medical Sciences, 38, 69-83.
- JUPP, P. G., 1974. Laboratory studies on the transmission of West Nile virus by Culex (Culex) univittatus Theobald; factors influencing the transmission rate. Journal of Medical Entomology, 11, 455-458.
- JUPP, P. G., 1975. Further studies on the overwintering stages of Culex mosquitoes (Diptera: Culicidae) in the Highveld region of South Africa. Journal of the Entomological Society of Southern Africa, 38, 89-97.
- JUPP, P. G., 1976. The susceptibility of four South African species of Culex to West Nile and Sindbis viruses by two different infecting methods. Mosquito News, 36, 166-173.
- JUPP, P. G., 1983. National Institute for Virology, Johannesburg. Unpublished data.
- JUPP, P. G., 1987. National Institute for Virology, Johannesburg. Unpublished data.
- JUPP, P. G., BLACKBURN, N. K., THOMPSON, D. L. & MEENEHAN, G. M., 1986. Sindbis and West Nile virus infections in the Witwatersrand/Pretoria region. South African Medical Journal, 70, 218-220.
- JUPP, P. G. & CORNEL, A. J., 1988. Vector competence tests with Rift Valley fever virus and five South African species of mosquitoes. Journal of the American Mosquito Control Association, 4, 4-8.
- JUPP, P. G. & KEMP, A., 1998. Studies on an outbreak of Wesselsbron virus in the Free State Province, South Africa. Journal of the American Mosquito Control Association, 14, 40-45
- JUPP, P. G. & MCINTOSH, B. M., 1967. Ecological studies on Sindbis and West Nile viruses in South Africa. II. Mosquito bionomics. South African Journal of Medical Sciences, 32, 15-33.
- JUPP, P. G. & MCINTOSH, B. M., 1987. A bionomic study of adult Aedes (Neomelaniconion) circumluteolus in northern Kwazulu, South Africa. Journal of the American Mosquito Control Association, 3, 131-136.
- JUPP, P. G., MCINTOSH, B. M. & ANDERSON, D., 1976. Culex (Eumelanomyia) rubinotus Theobald as vector of Banzi, Germiston and Witwatersrand viruses. IV. Observations on the biology of Cx. rubinotus. Journal of Medical Entomology, 12, 647-651.
- JUPP, P. G., MCINTOSH, B. M. & BLACKBURN, N. K., 1986. Experimental assessment of the vector competence of Culex (Culex) neavei Theobald with West Nile and Sindbis viruses in South Africa. Transactions of the Royal Society of Tropical Medicine and Hygiene, 80, 226-230.
- JUPP, P. G., MCINTOSH, B. M. & NEVILL, E. M., 1980. A survey of the mosquito and Culicoides faunas at two localities in the Karoo region of South Africa with some observations on bionomics. Onderstepoort Journal of Veterinary Research, 47, 1-6.
- JUPP, P. G., MCINTOSH, B. M. & THOMPSON, D. L., 1983. Isolation of Rift Valley fever virus from Aedes (Neomelaniconion) circumluteolus and/or luteolateralis collected during an outbreak in cattle in the coastal region of Natal, South Africa. South African Journal of Science, 79, 377.
- JUPP, P. G., MCINTOSH, B. M. & THOMPSON, O. L., 1984. Mechanical transmission of Rift Valley fever virus by mosquitoes. South African Journal of Science, 80, 276.
- KAUFFMAN, E. B. & KRAMER, L. D., 2017. Zika virus mosquito vectors: competence, biology and vector control. Journal of Infectious Diseases, 216(10), S976-S990.
- KOKERNOT, R. H., HEYMANN, C. S., MUSPRATT, J. & WOLSTENHOLME, B., 1957. Studies on arthropod-borne viruses of Tongaland. V. Isolation of Bunyamwera and Rift Valley fever viruses from mosquitoes. South African Journal of Medical Sciences, 22, 71-79.
- KOKERNOT, R. H., MCINTOSH, B. M., WORTH, B., DE MORAIS, T. & WEINBREN, M. P., 1962. Isolation of viruses from mosquitoes collected at Lumbo, Mozambique. I Lumbo virus, a new virus isolated from Aedes (Skusea) pembaensis Theobald. Journal of Tropical Medicine and Hygiene, 11, 678-682.
- KOKERNOT, R. H., MCINTOSH, B. M., WORTH, B. & DE SOUSA, J., 1962. Isolation of viruses from mosquitoes collected at Lumbo, Mozambique. II Mossuril virus, a new virus isolated from the Culex (Culex) sitiens Weidemann group.American Journal of Tropical Medicine and Hygiene, 11, 683-684.
- KOKERNOT, R. H., PATERSON, H. E. & DE MEILLON, B., 1958. Studies on the transmission of Wesselsbron virus by Aedes (Ochlerotatus) caballus (Theo). South African Medical Journal, 32, 546-548.
- KOKERNOT, R. H., SMITHBURN, K. C., GANDARA, A. F., MCINTOSH, B. M. & HEYMANN, C. S., 1960. Provas de neutralizaçao com soros de indivíduos residentes em Moçambique contra determinados vírus isolados em Africa transmitidos por artrópodes. Anals Do Instituto De Medicino Tropical, 17, 201-230.
- KOKERNOT, R. H., SMITHBURN, K. C. & KLUGE, F., 1961. Neutralizing antibodies against arthropod-borne viruses in the sera of domestic quadrupeds ranging in Tongaland, Union of South Africa. Annals of Tropical Medicine and Parasitology, 55, 73-85.
- KOKERNOT, R. H., SMITHBURN, K. C., PATERSON, H. E. & DE MEILLON, B., 1960. Further isolations of Wesselsbron virus from mosquitoes. South African Medical Journal, 34, 871-874.
- KOKERNOT, R. H., SMITHBURN, K. C. & WEINBREN, M. P., 1956. Neutralizing antibodies to arthropod-borne viruses in human beings and animals in the Union of South Africa. Journal of Immunology, 77, 313-323.
- KOKERNOT, R. H., SZLAMP, E. L., LEVITT, J. & MCINTOSH, B. M., 1965. Surveys for antibodies against arthropod-borne viruses in the sera of indigenous residents of the Caprivi Strip and Bechuanaland Protectorate. Transactions of the Royal Society of Tropical Medicine and Hygiene, 59, 553-562.
- KRUGER, A. C. & NXUMALO, M. P., 2017. Historical rainfall trends in South Africa: 1921-2015. Water SA, 43, 285-297. doi: http://dx.doi.org/10.4314/wsa/v43i2.12.
- LINTHICUM, K. J., DAVIES, F. G., KAIRO, A. & BAILEY, C. L., 1985. Rift Valley fever virus (family Bunyaviridae, genus Phlebovirus). Isolations from Diptera collected during an inter-epizootic period in Kenya. Journal of Hygiene, Cambridge, 95, 197-209.
- LOGAN, T. M., LINTHICUM, K. J., DAVIES, F. G., BINEPAL, Y. S. & ROBERTS, C. R., 1991. Isolation of Rift Valley fever virus from mosquitoes (Diptera: Culicidae) collected during an outbreak in domestic animals in Kenya. Journal of Medical Entomology, 28, 293-395.
- LUTOMIAH, J., ONGUS, J., LINTHICUM, K. & SANG, R., 2014. Natural vertical transmission of Ndumu virus in Culex pipiens (Diptera: Culicidae) mosquitoes collected as larvae. Journal of Medical Entomology, 51, 1091-1095.
- MACDONALD, G., 1957. The epidemiology and Control of Malaria. London: Oxford University Press.
- MCABEE, R. D., GREEN, E. N., HOLEMAN, J., CHRISTIANSEN, J., FRYE, N., DEALEY, K., MULLIGAN, F. S., BRAULT, A. C. & CORNEL, A. J., 2008. Identification of Culex pipiens complex mosquitoes in a hybrid zone of West Nile virus transmission in Fresno County, California. American Journal of Tropical Medicine and Hygiene, 303-310.
- MCINTOSH, B. M., 1961. Susceptibility of some African wild rodents to infection with various arthropod-borne viruses. Transactions of the Royal Society of Tropical Medicine and Hygiene, 55, 63-68.
- MCINTOSH, B. M., 1971. The aedine subgenus Neomelaniconion Newstead (Culicidae, Diptera) in southern Africa with descriptions of two new species. Journal of the Entomological Society of Southern Africa, 34, 319-333.
- MCINTOSH, B. M., 1972. Rift Valley fever. I. Vector studies in the field. Journal of the South African Veterinary Association, 43, 391-395.
- MCINTOSH, B. M., 1973. A taxonomic reassessment of Aedes (Ochlerotatus) caballus (Theobald) (Diptera: Culicidae) including a description of a new species of Ochlerotatus. Journal of the Entomological Society of Southern Africa, 36, 261-269.
- MCINTOSH, B. M., 1980. The epidemiology of arthropod-borne viruses in southern Africa. D.Sc. Thesis, University of Pretoria.
- MCINTOSH, B. M., 1986. Mosquito-borne virus diseases of man in southern Africa. South African Medical Journal, 70, 69-72.
- MCINTOSH, B. M., DICKENSON, D. B., MEENEHAN, G. M. & DOS SANTOS, I. S. L., 1976. Culex (Eumelanomyia) rubinotus Theobald as vector of Banzi, Germiston and Witwatersrand viruses. II. Infections in sentinel hamsters and wild rodents. Journal of Medical Entomology, 12, 641-644.
- MCINTOSH, B. M., DICKENSON, D. B. & SERAFINI, E. T., 1962. Antibodies against certain arboviruses in sera from human beings and domestic animals from the South African Highveld. South African Journal of Medical Sciences, 27, 87-94.
- MCINTOSH, B. M., DOS SANTOS, I. S. L. & MEENEHAN, G. M., 1976. Culex (Eumelanomyia) rubinotus Theobald as vector of Banzi, Germiston and Witwatersrand viruses. III. Transmission of virus to hamsters by wild-caught infected C. rubinotus. Journal of Medical Entomology, 12, 645-646.
- MCINTOSH, B. M. & JUPP, P. G., 1979. Infections in sentinel pigeons by Sindbis and West Nile viruses in South Africa, with observations on Culex (Culex) univittatus (Diptera: Culicidae) attracted to these birds. Journal of Medical Entomology, 16, 234-239.
- MCINTOSH, B. M. & JUPP, P. G., 1982. Ecological study on West Nile virus in southern Africa 1965–1980. In: GEORGE, T. D. & KAY, B. H., (eds). Arbovirus Research in Australia. Proceedings of the 3rd symposium of the Commonwealth Scientific and Research Organization and the Queensland Institute for Medical Research, 15–17 February, 1982, Brisbane, Australia.
- MCINTOSH, B. M., JUPP, P. G., ANDERSON, D. & DICKENSON, D. B., 1973. Rift Valley fever: 2. Attempts to transmit virus with seven species of mosquitoes. Journal of the South African Veterinary Association, 44, 57-60.
- MCINTOSH, B. M., JUPP, P. G. & DE SOUSA, J., 1972. Further isolations of arboviruses from mosquitoes collected in Tongaland, South Africa, 1960–1968. Journal of Medical Entomology, 9, 155-159.
- MCINTOSH, B. M., JUPP, P. G. & DE SOUSA, J., 1972. Mosquitoes feeding at two horizontal levels in gallery forest in Natal, South Africa, with reference to possible vectors of chikungunya virus. Journal of the Entomological Society of Southern Africa, 35, 81-90.
- MCINTOSH, B. M., JUPP, P. G., DICKENSON, D. B., MCGILLIVRAY, G. M. & SWEETNAM, J., 1967. Ecological studies on Sindbis and West Nile viruses in South Africa. I. Viral activity as revealed by infection of mosquitoes and sentinel fowls. South African Journal of Medical Sciences, 32, 1-14.
- MCINTOSH, B. M., JUPP, P. G. & DOS SANTOS, I., 1978. Infection by Sindbis and West Nile viruses in wild populations of Culex (Culex) univittatus Theobald (Diptera: Culicidae) in South Africa. Journal of the Entomological Society of Southern Africa, 41, 57-61.
- MCINTOSH, B. M., JUPP, P. G., DOS SANTOS, I. & BARNARD, B. J. H., 1980. Vector studies on Rift Valley fever in South Africa. South African Medical Journal, 58, 127-132.
- MCINTOSH, B. M., JUPP, P. G., DOS SANTOS, I. & MEENEHAN, G. M., 1976. Epidemics of West Nile and Sindbis viruses in South Africa with Culex (Culex) univittatus Theobald as vector. South African Journal of Science, 72, 295-300
- MCINTOSH, B. M., JUPP, P. G., DOS SANTOS, I. & ROWE, A. C., 1983. Field and laboratory evidence implicating Culex zombaensis and Aedes circumluteolus as vectors of Rift Valley fever virus in coastal South Africa. South African Medical Journal of Science, 79, 61-64.
- MCINTOSH, B. M., JUPP, P. G., DOS SANTOS, I. S. L. & MEENEHAN, G. M., 1976. Culex (Eumelanomyia) rubinotus Theobald as vector of Banzi, Germiston and Witwatersrand viruses. I. Isolation of virus from wild populations of Cx. rubinotus. Journal of Medical Entomology, 12, 637-640.
- MCINTOSH, B. M., KOKERNOT, R. H. & PATERSON, H. E., 1960. Witwatersrand virus: an apparently new virus isolated from Culicine Mosquitoes. South African medical Journal of Science, 23, 33-37.
- MCINTOSH, B. M., KOKERNOT, R. H., PATERSON, H. E. & DE MEILLON, B., 1961. Isolation of Spondweni virus from four species of Culicine mosquitoes and a report of two laboratory infections with the virus. South African Medical Journal, 35, 647-650.
- MCMILLAN, J. R., BLAKNEY, R. A., MEAD, D. G., COKER, S. M., MORRAN, L. T., WALLER, L. A., KITROM, U. & VAZQUEZ-PROKOPEC, G. M., 2018. Larviciding Culex spp. (Diptera: Culicidae) populations in catch basins and its impact on West Nile virus transmission in urban parks in Atlanta, GA. Journal of Medical Entomology, 20, 1-11.
- METRAS, R., BAGUELIN, M., EDMUNDS, W. J., THOMPSON, P. N., KEMP, A., PFEIFFER, D. U., COLLINS, L. M. & WHITE, R. G., 2013. Transmission potential of Rift Valley Fever virus over the course of the 2010 epidemic in South Africa. Emerging Infectious Diseases, 19. doi: http://dx.org/10.3201/eid1906.121641.
- METRAS, R., JEWELL, C., PORPHYRE, T., THOMPSON, P. N., PFEIFFER, D. U., COLLINS, L. M. & WHITE, R. G., 2015. Risk factors associated with Rift Valley fever epidemics in South Africa in 2008-11. Scientific Reports, 6, 9492, doi: 10.1038/srep09492.
- METSELAAR, D., HENDERSON, B. E., KIRYA, G. B., TUKEI, P. M. & DE GEUS, A., 1974. Isolation of arboviruses in Kenya, 1966–1971. Transactions of the Royal Society of Tropical Medicine and Hygiene, 68, 114-123.
- MILLER, M., BUSS, P., JOUBERT, J., MASEKO, N., HOFMEYR, M. & GERDES, T., 2011. Serosurvey for selected viral agents in white rhinoceros (Ceratotherium simum) in Kruger National Park, 2007. Journal of Zoo and Wildlife Medicine, 42, 29-32.
- MÖHLMANN, T. W. R., OYMANS, J., SCHREUR, P. J. W., KOENRAADT, J. M., KORTEKAAS, J. & VOGELS, C. B. F., 2018. Vector competence of biting midges and mosquitoes for Shuni virus. PLoS Negelected Tropical Diseases, 12, e0006993. doi: https://doi.org/10.1371/journal.pntd.0006993.
- MOIANE, B., MAPACO, L., THOMPSON, P., BERG, M., ALBIHN, A. & FAFETINE, J., 2017. High seroprevalence of Rift valley Fever phlebovirus in domestic ruminants and African Buffaloes in Mozambique shows need for intensified surveillance. Infection Ecology & Epidemiology, 7, 1416248. doi: https://doi.org/10.1080/230008686.2017.1416248.
- MONACO, F., PINONI, C., COSSEDDU, G. M., KHAISEB, S., CALISTRI, P., MOLINI, U., BISHI, A., CONTE, A., SCACCHIA, M. & LELLI, R., 2013. Rift Valley Fever in Namibia. Emerging Infectious Diseases, 19, 2025-2027.
- MOSQUITO CATALOGUE, http://www.mosquitocatalog.org/. (Accessed 2 January 2019)
- MUSPRATT, J., 1955. Research on South African Culicini (Diptera: Culicidae). Journal of the Entomological Society of Southern Africa, 18, 149-207.
- MUSPRATT, J., SMITHBURN, K. C., PATERSON, H. E. & KOKERNOT, R. H., 1957. The laboratory transmission of Wesselsbron virus by the bite of Aedes (Banksinella) circumluteolus Theo. South African Journal of Medical Sciences, 22, 121-126.
- NDIAYE, E. H., G., F., BA, Y., FAYE, O., DIA, I. & DIALLO, M., 2018. Arboviruses isolated from Barkedji mosquito-based surveillance system, 2012-2013. BMC Infectious Diseases, 18, 642. https://doi.org/10.1186/s12879-018-3538-2.
- NIKLASSON, B., LILJESTRAND, J., BERGSTRÖM, S. & J., P. C., 1987. Rift Valley Fever: a sero-epidemiological survey among pregnant women in Mozambique. Epidemiology and Infection, 99, 517-522.
- NODEN, B. C. & VAN DER COLF, B., 2013. Neglected tropical diseases of Namibia: Unsolved mysteries. Acta Tropica, 125, 1-17.
- ORGANIZATION., W. H., 2018. Annual review of diseases prioritized under the Research and Development Blueprint. doi: http://www/who.int/blueprint/en/. (Accessed 27 December 2018).
- PATERSON, H. E., BRONSDEN, P., LEVITT, J. & WORTH, C. B., 1964. Some culicine mosquitoes (Diptera: Culicidae) at Ndumu, Republic of South Africa; a study of their host preferences and host range. Medical Proceedings, 10, 188-192.
- PETERSEN, L. R. & MARFIN, A. A., 2002. West Nile virus: a primer for the clinician. Annals of Internal Medicine, 137, 173-179.
- RAPPOLE, J. H., DERRICKSON, S. R. & HUBÁLEK, Z., 2000. Migratory birds and spread of West Nile virus in the Western Hemisphere. Emerging Infectious Diseases, 6, 319-328.
- REBELLO, B., SARRASECA, J., LECOLLINET, S., ABOUCHOAIB, N., ALONSO, J., GARVIA-BOCANEGRA, I., SANZ, A. J., VENTEO, A. & JIMENEZ-CLAVERO, M., 2018. Monitoring Anti-NS1 antibodies in West Nuile virus-infected and vaccinated horses. BioMed Research International, 2018, 8309818, 8 pages, https://doi.org/10.1155/2018/8309816.
- REEVES, W. C., BELLAMY, R. E., GEIB, A. F. & SCRIVANI, R. P., 1964. Analysis of the circumstances leading to abortion of a Western equine encephalitis epidemic. American Journal of Hygiene, 80, 205-220.
- REINERT, J. F., HARBACH, R. E. & KITCHING, I. J., 2009. Phylogeny and classification of tribe Aedini (Diptera: Culicidae). Zoological Journal of the Linnean Society, 57,700-794 + two appendices.
- REIS, S., CORNEL, A. J., MELO, M., PEREIRA, H. & LOISEAU, C., 2016. First record of Aedes albopictus(Skuse 1894) on Sao Tomé Island. Acta Tropica, 171, 86-89.
- RIBEIRO, H. & RAMOS, H. D. C., 1980. Research on the mosquitoes of Angola (Diptera: Culicidae). X- The genus Culex L., 1758. Check-list with new records, keys to females and larvae, distribution and taxonomic and bioecological notes. Est. Ens. Doc., n 134, Junta Invest. Cient. Ult., Lisboa.
- RIBEIRO, H. & RAMOS, H. D. C., 1980. Research on the mosquitoes of Angola. VIII. The genus Aedes Meigan, 1818 (Diptera: Culicidae). Check-list with new records, keys to females and larvae, distribution and taxonomic and bioecological notes. Anais do Instituto de Higiene e Medicina Tropical, Lisboa, 1, 107-138.
- RODGER, L. M., 1961. An outbreak of suspected Chikungunya fever in northern Rhodesia. South African Medical Journal 35, 126-128.
- SECCACINI, E., JUAN, L., ZREBA, E. & LICASTRO, S., 2014. Aedes aegypti (Diptera: Culicidae): evaluation of natural long-lasting materials containing pyriproxyfen to improve control strategies. Parasitology Research, 113, 3355-3360.
- SERVICE, M. W., 1980. A Guide to Medical Entomology. London: Macmillan.
- SMITH, E. R. & PARKER, D. M., 2010. Tick communities at the expanding wildlife/cattle interface in the Eastern Cape Province, South Africa: implications for Corridor disease. Journal of the South African Veterinary Association, 81, 237-240.
- SMITHBURN, K. C., KOKERNOT, R. M., WEINBREN, M. P. & DE MEILLON, B., 1957. Isolation of Wesselsbron virus from a naturally infected human being and from Aedes (Banksinella) circumluteolus Theo. South African Journal of Medical Sciences, 22, 113-120.
- SMITHBURN, K. C., PATERSON, H. E., HEYMANN, C. S. & WINTER, P. A. D., 1959. An agent related to Uganda S virus from man and mosquitoes in South Africa. South African Medical Journal, 46, 959-962.
- STORM, N., WEYER, J., MSIMANG, V., NEL, L. H., KEMP, A., MARKOTTER, W. & PAWESKA, J. T., 2014. Epidemiological features of Sindbis fever in South Africa. 16th ICID Abstracts/ International Journal of Infectious Diseases, 21S, 327. doi: http://dx.doi.org/10.1016.j.ijid.2014.03.1096
- STUECKLE, T. A., LIKENS, J. & FORAN, C. M., 2008. Limb regeneration and molting process under chronic methoprene exposure in the mud fiddler crab, Uca pugnax. Comparative Biochemistry and Physiology, Part C, 147, 366-377.
- TYSON, P. D., 1978. Rainfall changes over South Africa during the period of meteorological record. In: WEGER, M. J. A., (ed.). Biogeography and Ecology of Southern Africa. The Hague: W. Junk.
- VAN DER LINDE, T. C., DE, K., HEWITT, P. H., VAN PLETZEN, R., KOK, D. J., FOURIE, S., MOSTERT, D. J. & NEL, A., 1982. Species richness and relative abundance of female mosquitoes at a site in the western Orange Free State. Journal of the Entomological Society of Southern Africa, 45, 57-67.
- VAN EEDEN, C., SWANEPOEL, R. & VENTER, M., 2014. Antibodies against West Nile and Shuni Viruses in Veterinarians, South Africa. Emerging Infectious Diseases, 20, 1409-1411. doi:10.3201/eid2008.131724.
- VAN EEDEN, C., WILLIAMS, J. H., GERDES, T. G. H., VAN WILPE, E., VILJOEN, A., SWANEPOEL, R. & VENTER, M., 2012. Shuni virus as cause of neurologic disease in horses. Emerging Infectious Diseases, 18, 3318-3321.
- VAN NIEKERK, S., HUMAN, S., WILLIAMS, J., VAN WILPE, E., PRETORIUS, M., SWANEPOEL, R. & VENTER, M., 2015. Sindbis and Middleburg old world Alphaviruses associated with Neurologic Disease in Horse, South Africa. Emerging Infectious Diseases, 21, 2225-2229.
- VAN VUREN, P. J., KGALADI, J., PATHAROO, V. & PAWESKA, J. T., 2018. Complete genome sequences of Spondweni viruses isolated between 1958 and 1960. Microbiology Resource Announcement, 7 e01278-18.
- VAN VUUREN, M. & PENZHORN, B. L., 2015. Geographic range of vector-borne infections and their vectors: the role of African wildlife. Technical Review of the Office International des Epizooties, 34, 139-149.
- VENTER, M., 2018. Assessing the zoonotic potential of arboviruses of African origin. Current Opinion in Virology, 26, 74-84.
- VENTER, M., HUMAN, S., VAN NIEKERK, S., WILLIAMS, J., VAN EEDEN, C. & FREEMAN, F., 2011. Fatal neurologic disease and abortion in mare infected with lineage 1 West Nile virus, South Africa. Emerging Infectious Diseases, 17, 1534-1536.
- VENTER, M., HUMAN, S., ZAAYMAN, D., GERDES, G. H., WILLIAMS, J., STEYL, J., LEMAN, P. A., PAWESKA, J. T., SETZKORN, H., ROUS, G., MURRAY, S., PARKER, R., DONNELLAN, C. & SWANEPOEL, R., 2009. Lineage 2 West Nile virus as casue of fatal neurologic disease in horses, South Africa. Emerging Infectious Diseases, 15, 877-884. doi. 10.3201/eid1506.081515.
- VENTER, M., M., P., FULLER, J. A., BOTHA, E., RAKGOTHO, M., STIVAKTAS, V., WEYER, C., ROMITO, M. & WILLIAMS, J., 2017. West Nile Virus Lineage 2 in horses and other animals with neurologic disease, South Africa, 2008-2015. Emerging Infectious Diseases, 23, 2060-2064.
- VENTER, M., VAN EEDEN, C., WILLIAMS, J., VAN NIEKERK, S., STEYL, J., JOOSTE, T. & SWANEPOEL, R., 2014. Arboviruses associated with neurological disease in animals in South Africa and their zoonotic potential in humans. 16th ICID Abstracts/ International Journal of Infectious Diseases, 21S (2014), 184. doi: http://dx.doi.org/10.1016.j.ijid.2014.03.804.
- WHITE, S. K., LEDNICKY, J. A., OKECH, B. A., MORRIS JR., G. & DUNFORD, J. C., 2018. Spondweni virus in field-caught Culex quinquefasciatus mosquitoes, Haiti, 2016. Emerging Infectious Diseases, 24, 1765-1767.
- WILKERSON, R. C., LINTON, Y. M., FONSECA, D. M., SCHULTZ, T. R., PRICE, D. C. & STRICKMAN, D., 2015. Making mosquito taxonomy useful: A stable classification of tribe Aedini that balances utility with current knowledge of evolutionary relationships. PLoS One, 10, e0133602.
- WILLIAMS, J. H., VAN NIEKERK, S., HUMAN, S., VAN WILPE, E. & VENTER, M., 2014. Pathology of fatal lineage 1 and 2 West Nile virus infections in horses in South Africa. Journal of the South African Veterinary Association, 85(1), 1105. doi: http://dx.doi.org/10.4102/jsava.v85Si1.1105.
- WOLF, T. M., GANDOLF, A. R., DOOLEY, J. L., ATKINSON, M. W. & WOLFE, B. A., 2008. Serological response to West Nile virus vaccination in the greater one-horned Rhinoceros (Rhinoceros unicornis). Journal of Zoo and Wildlife Medicine, 39, 537-541.
- WORTH, C. B., PATERSON, H. E. & DE MEILLON, B., 1961. The incidence of arthropod-borne viruses in a population of culicine mosquitoes in Tongaland, Union of South Africa (January, 1956, through April, 1960). American Journal of Tropical Medicine and Hygiene, 10, 583-592.
- ZAAYMAN, D. & VENTER, M., 2012. West Nile Virus Neurologic disease in humans, South Africa, September 2008-May 2009. Emerging Infectious Diseases, 18, 2051-2054.