- Infectious Diseases of Livestock
- Part 1
- Leishmaniosis
- Vectors: Ticks
- Vectors: Tsetse flies
- Vectors: Muscidae
- Vectors: Tabanidae
- Vectors: Culicoides spp.
- Vectors: Mosquitoes
- Classification, epidemiology and control of arthropod-borne viruses
- Special factors affecting the control of livestock diseases in sub-Saharan Africa
- The control of infectious diseases of livestock: Making appropriate decisions in different epidemiological and socioeconomic conditions
- Infectious diseases of animals in sub-Saharan Africa: The wildlife⁄livestock interface
- Vaccination: An approach to the control of infectious diseases
- African animal trypanosomoses
- Dourine
- Trichomonosis
- Amoebic infections
- GENERAL INTRODUCTION: COCCIDIA
- Coccidiosis
- Cryptosporidiosis
- Toxoplasmosis
- Besnoitiosis
- Sarcocystosis
- Balantidiosis
- Leishmaniosis
- Neosporosis
- Equine protozoal myeloencephalitis
- GENERAL INTRODUCTION: BABESIOSES
- Bovine babesiosis
- Equine piroplasmosis
- Porcine babesiosis
- Ovine babesiosis
- GENERAL INTRODUCTION: THEILERIOSES OF CATTLE
- East Coast fever
- Corridor disease
- Zimbabwe theileriosis
- Turning sickness
- Theileria taurotragi infection
- Theileria mutans infection
- Theileria annulata theileriosis
- Theileriosis of sheep and goats
- Theileria buffeli⁄orientalis infection
- Non-pathogenic Theileria species in cattle
- GENERAL INTRODUCTION: RICKETTSIAL, CHLAMYDIAL AND HAEMOTROPIC MYCOPLASMAL DISEASES
- Heartwater
- Lesser known rickettsial infections in animals and humans
- Chlamydiosis
- Q fever
- Eperythrozoonosis
- Bovine Haemobartonellosis
- Potomac horse fever
- GENERAL INTRODUCTION: ANAPLASMOSES
- Bovine anaplasmosis
- Ovine and caprine anaplasmosis
Leishmaniosis
This content is distributed under the following licence: Attribution-NonCommercial CC BY-NC  View Creative Commons Licence details here
View Creative Commons Licence details here

Introduction
Leishmaniosis is caused by obligatory intracellular parasitic protozoa of the genus Leishmania that affect various mammalian hosts, but disease occurs most commonly in humans and dogs. Cutaneous, mucocutaneous and visceral forms of the disease are recognized; the visceral form being chronic and often fatal in humans and dogs.
Leishmania spp. are widely distributed in Central and South America, East and West Africa, Asia, the Mediterranean basin and some neighbouring European countries, but they are rare in southern Africa. The cutaneous form of the disease has recently been diagnosed in two sheep from different localities in South Africa.15, 22
Aetiology, life cycle and epidemiology
Members of the genus Leishmania, which comprises a considerable number of species, belong to the order Kinetoplastida and family Trypanosomatidae.11 Identification of species within the genus is complex and is based on clinical and pathological features of the disease, geographical distribution, morphology and behaviour of the parasite, and the results of biochemical and immunological tests.10
Diseases caused by Leishmania spp. are transmitted by blood-sucking sandflies of the genera Lutzomyia in the New World and Phlebotomus in the Old World. The disease in dogs is caused by L. infantum.
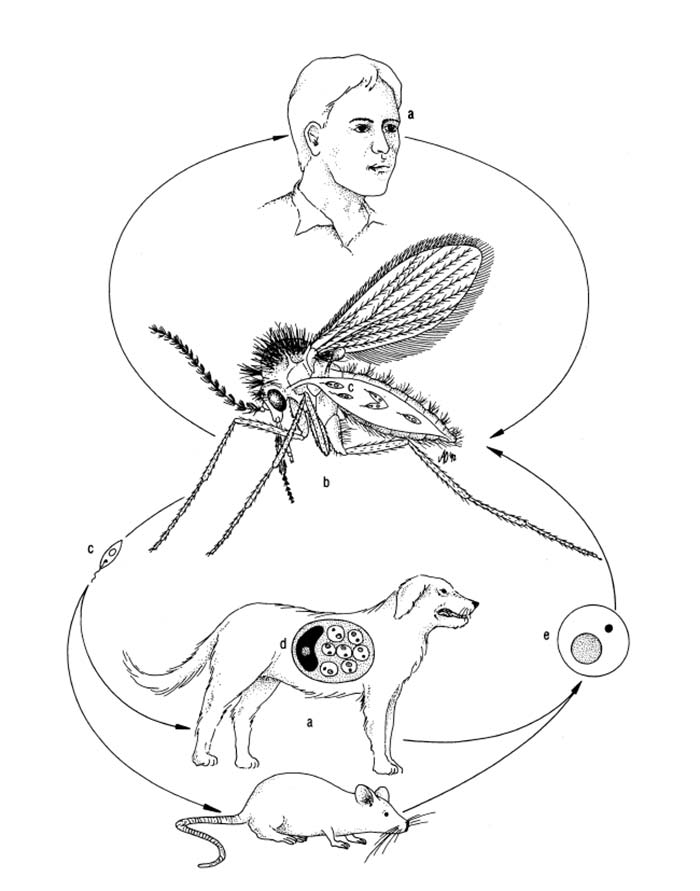
There is usually a primary reservoir host for a given Leishmania sp. in a particular area where the parasite is maintained by a cycle between sandflies and the mammalian hosts (Figure 22.1). Secondary and incidental mammalian hosts also become infected.2 Both domestic and wild dogs (Lycaon pictus), as well as rodents and other wild animals, are reservoir hosts, but occasionally humans may act as the main reservoir.
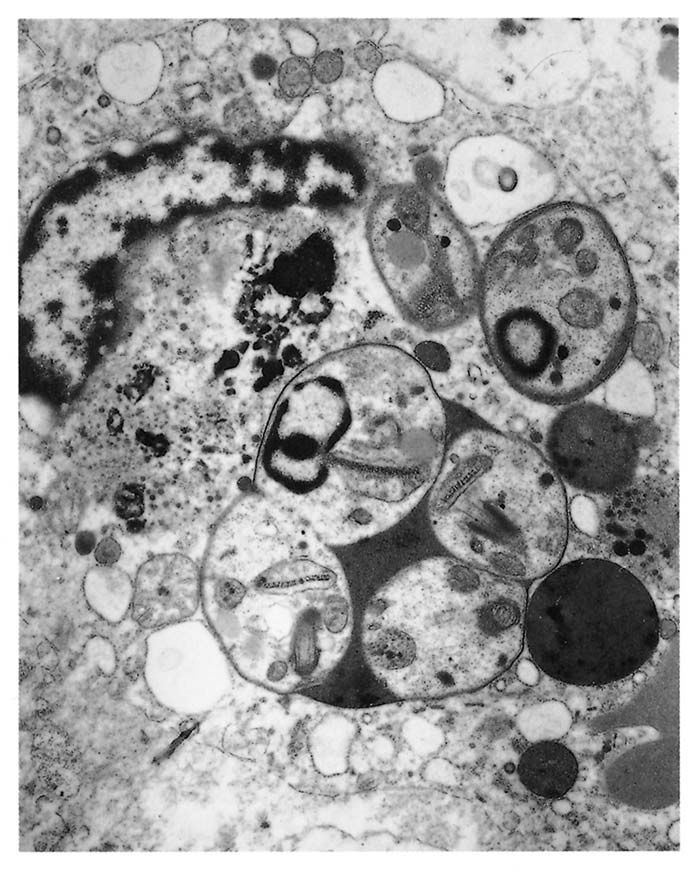
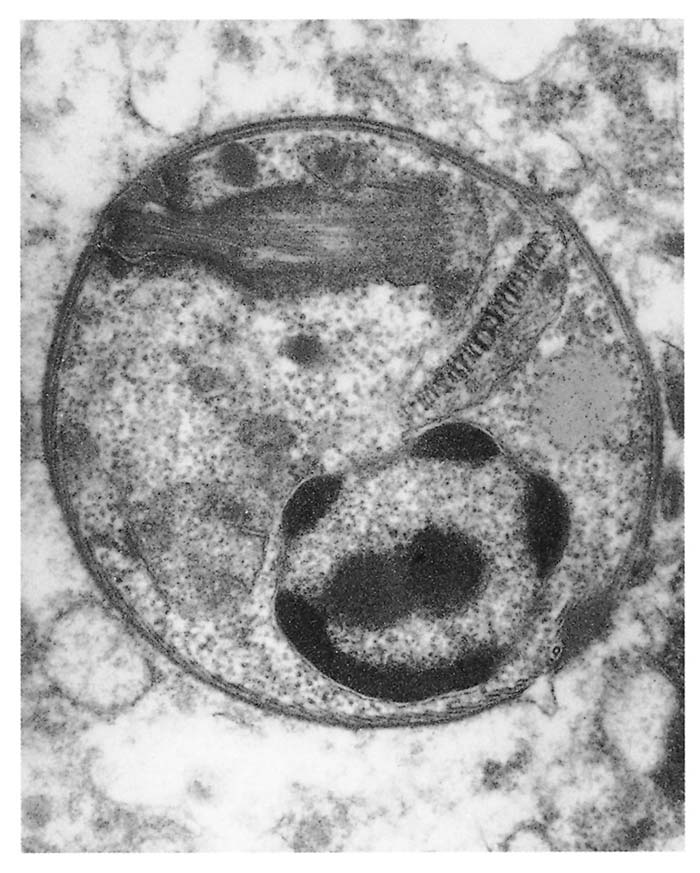
The parasite occurs in two forms: a promastigote stage which is found in the sandfly; and an amastigote stage which occurs in the mammalian host. During feeding, the infected sandfly injects promastigotes into the dermis of the mammalian host and these are engulfed by phagocytic cells before transforming into amastigotes which multiply by binary fission. Heavily parasitized host cells eventually rupture, and the liberated amastigotes invade other macrophages in which they may be spread locally or systemically.17 In tissues, amastigotes of all Leishmania spp. are morphologically similar (Figure 22.2 and Figure 22.3): they are round to oval bodies, 1,5–3 × 2,5–6,5 μm in diameter, containing a single nucleus and a rod-shaped kinetoplast.17
Leishmaniosis has rarely been reported in livestock. There have been reports of infection in calves from Africa in the early literature, but no details are given.13 The cutaneous form of the disease occurs frequently in horses, donkeys and mules in endemic areas of human and canine leishmaniosis in Venezuela and Brazil.3, 4 In 1989 and 1990 cutaneous leishmaniosis was diagnosed in sheep in South Africa — in one instance in two adult ewes in a flock near Barberton in Mpumalanga, and in the other in one sheep near Greytown in KwaZulu-Natal.15, 22
Canine leishmaniosis is endemic in the Mediterranean countries, and is occasionally seen in North Europe and in the USA in dogs that have previously been in endemic areas.20
In South Africa, visceral leishmaniosis has been diagnosed twice in dogs: in 1964 in a dog from Durban whose life history is uncertain;18 and in 1987 in an animal from the Free State which had never left the country.23 In Namibia, 34 cases of cutaneous leishmaniosis in humans have been diagnosed since 1970,9 while only a single case of the cutaneous form was reported from the Northern Cape Province in 1979.1
The epidemiology of leishmaniosis in southern Africa is not known. In Namibia, apart from humans, Leishmania parasites have also been isolated from the skin of hyraxes (Procavia capensis) and from naturally infected Phlebotomus rossi sandflies (which are possible vectors of the human disease). The isolates made from humans and sandflies are identical, but are distinctly different from those recovered from the hyraxes. The two Namibian strains of parasite are also dissimilar to L. tropica and L. major which cause cutaneous leishmaniosis in the Old World and are afforded separate status.9
Pathogenesis, clinical signs and pathology
Leishmaniosis in humans comprises several syndromes depending primarily on the species of Leishmania concerned. The clinical spectrum ranges from asymptomatic infections to those with a high mortality. Lesions may be confined to the skin (cutaneous leishmaniosis) and result, inter alia, in nodule and ulcer formation, known in some parts of the world as the ‘Oriental sore’; to the mucocutaneous tissues (mucocutaneous leishmaniosis, sometimes referred to as espundia), in which the organisms are found in endothelial and mononuclear cells of the skin and mucous membranes of the nose, mouth, pharynx and elsewhere and may result in severe disfigurement of the invaded tissues; or they may involve several tissues especially the liver, spleen, bone marrow and lymph nodes (the highly fatal visceral leishmaniosis or ‘Kala azar’).21
In dogs, the parasites multiply within macrophages and other cells of the mononuclear-phagocyte-system and cause chronic inflammatory processes.5, 6 Canine leishmaniosis is a systemic disease, and, in contrast to the situation in human cutaneous leishmaniosis, the skin of dogs is affected in the course of a generalized dissemination of the parasite. Chronic renal failure due to an immune-mediated glomerulonephritis is the main cause of death in affected dogs.20
Clinically, canine leishmaniosis is a chronic wasting disease accompanied by anaemia, intermittent pyrexia and generalized lymphadenopathy. Skin lesions, of which four forms have been described, are common: symmetrical alopecia with dry desquamation; a generalized nodular disease; ulcerative dermatosis; and a form characterized by the presence of multiple pustules. Keratoconjunctivitis, epistaxis and diarrhoea may also be present.8, 12
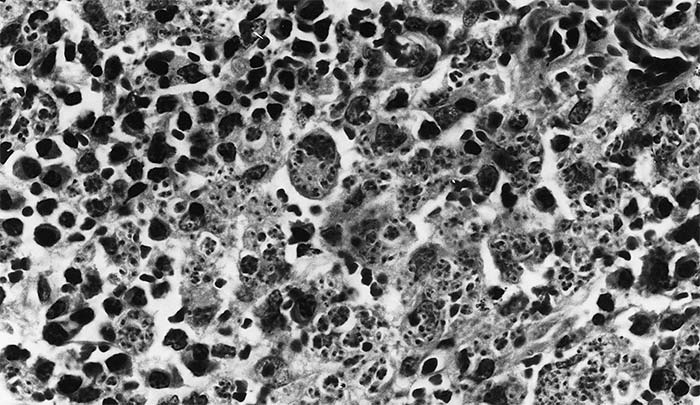
At necropsy, in addition to the lesions visible clinically in dogs, there is generalized lymphadenopathy, and hepato- and splenomegaly. Microscopically, large numbers of macrophages filled with the amastigote form of Leishmania are present in the spleen, bone marrow and lymphoid tissues. In non-lymphoid tissues, a granulomatous inflammatory infiltrate composed of parasitized macrophages, plasma cells and lymphocytes is evident.12
Leishmaniosis was diagnosed in two adult sheep born and reared on a farm in Mpumalanga. The clinical signs in both cases were similar.
The pinna of one ear of each animal was severely swollen and the overlying skin diffusely thickened and covered with a crusty exudate. Small crusts also occurred in the skin around the external nares and eyes, and in that of the upper lip, muzzle and lateral aspects of the face. Both animals recovered spontaneously.
Another case was diagnosed in an adult sheep from KwaZulu-Natal. It showed alopecia, hyperpigmentation, hyperkeratosis and crust formation of the skin of the dorsum of the nose and both ears, and around the eyes. It also displayed a focally disseminated alopecia and the presence of nodules in the skin of the ventral aspects of the limbs, abdomen and base of the tail. Its recovery was spontaneous.
Diagnosis
The diagnosis of leishmaniosis is based on identification of the agent and serological tests. Demonstration of the amastigotes in Giemsa-stained smears of splenic, bone marrow and lymph node aspirates, and of skin scrapings, gives a positive diagnosis, while an indirect immunoperoxidase staining technique of paraffin-embedded section is also available. If the infection is of low grade, detection is possible only by attempting in vitro orin vivo isolation. Isolated Leishmania organisms are characterized by isoenzyme, DNA and antigen methods at present. Serology is the preferred method for the diagnosis of visceral and canine leishmaniosis with the indirect fluorescent antibody test and the enzyme-linked immunosorbent assays the most suitable. Serology is of less value for the cutaneous and mucocutaneous forms of the disease.7, 14, 16, 19, 20
Control
Pentavalent antimonials, especially meglumine antimonate, are the most effective drugs for the treatment of canine leishmaniosis. With therapy, the majority of cases in dogs show remarkable improvement, provided renal glomerular filtration is not seriously decreased. Treatment is, however, expensive and relapses are the rule, with the development of complications such as iridocyclitis or endstage renal failure being common. Effective vaccines are not available.20
References
- ANON., 1984. The Leishmaniases. World Health Organization, Geneva. Technical Report Series 701.
- ANON., 1990. Control of the Leishmaniosis. World Health Organization, Geneva. Technical Report Series 793.
- AQUILAR, C.M., RANGEL, E.F. & DEANE, L.M., 1986. Cutaneous leishmaniasis is frequent in equines from an endemic area in Rio de Janeiro, Brazil. Memórias do Instituto Oswaldo Cruz, 81, 471–472.
- BONFANTE-GARRIDO, R., MELENDEZ, E.C., TORRES, R.A., MORILLO, N.C., ARREDONDO, C.C. & URDANETA, I., 1981. Enzootic equine cutaneous leishmaniasis in Venezuela. Transactions of the Royal Society of Tropical Medicine and Hygiene, 75, 471.
- CHANG, K.P., FONG, D. & BRAY, R.S., 1985. Biology of Leishmania and leishmaniasis. In: CHANG, K.P. & BRAY, R.S., (eds). Leishmaniasis. Amsterdam: Elsevier Science Publishers.
- CHAPMAN, W. & HANSON, W., 1984. Leishmaniasis. In: GREEN, C., (ed.). Clinical Microbiology and Infectious Diseases of the Dog and Cat. Philadelphia: W.B. Saunders.
- FERRER, L., RABANAL, R.M., DOMINGO, M., RAMOS, J.A. & FONDEVILA, D., 1988. Identification of Leishmania donovani amastigotes in canine tissues by immunoperoxidase staining. Research in Veterinary Science, 44, 194–196.
- FERRER, L., RABANAL, R., FONDEVILA, D., RAMOS, J.A. & DOMINGO, M., 1988. Skin lesions in canine leishmaniasis. Journal of Small Animal Practice, 29, 381–388.
- GROVÉ, S.S., 1989. Leishmaniasis in South West Africa/Namibia to date. South African Medical Journal, 75, 290–292.
- LAINSON, R. & SHAW, J.J., 1987. Evolution, classification and geographical distribution. In: PETERS, W. & KILLICK-KENDRICK, R., (eds). The Leishmaniasis in Biology and Medicine. Orlando: Academic Press
- LEVINE, N.D., CORLISS, J.O., COX, F.E.G. ET AL., 1980. A newly revised classification of the protozoa. Journal of Protozoology, 27, 37–58.
- LONGSTAFFE, J.A. & GUY, M.W., 1985. Leishmaniasis in dogs. Veterinary Annual, 25, 358–367.
- LOSOS, G.J., 1986. Infectious Tropical Diseases of Domestic Animals. Essex, England: Longman Scientific & Technical.
- MANCIANTI, F., FALCONE, M.L. & POLI, A., 1995. Comparison between an enzyme-linked immunosorbent assay using a detergent-soluble Leishmania infantum antigen and direct immunofluorescence for the diagnosis of canine leishmaniosis. Veterinary Parasitology, 59, 13–21.
- MARSHALL, C., 1990. Medical University of Southern Africa, Medunsa, South Africa. Unpublished observations.
- MATHIS, A. & DEPLAZES, P., 1995. PCR and in vitro cultivation for detection of Leishmania spp. in diagnostic samples from humans and dogs. Journal of Clinical Microbiology, 33, 1145–1149.
- MOLYNEUX, D.H. & KILLICK-KENDRICK, R., 1987. Morphology, ultrastructure and life cycles. In: PETERS, W. & KILLICK-KENDRICK, R., (eds). The Leishmaniasis in Biology and Medicine. Orlando: Academic Press
- PIENAAR, J.G., 1964. State Veterinarian, Potgietersrus, South Africa. Unpublished data.
- ROURA, X., SANCHES, A. & FERRER, L., 1999. Diagnosis of canine leishmaniasis by a polymerase chain reaction technique. The Veterinary Record, 144, 262–264.
- SLAPPENDEL, R.J., 1988. Canine leishmaniasis. The Veterinary Quarterly, 10, 1–16.
- SOULSBY, E.J.L., 1969. Helminths, Arthropods and Protozoa of Domesticated Animals (Sixth Edition of Monnig’s Veterinary Helminthology and Entomology). London: Baillière, Tindall & Cassel.
- VAN DER LUGT, J.J., CARLYON, J.F. & DE WAAL, D.T., 1992. Cutaneous leishmaniasis in a sheep. Journal of the South African Veterinary Association, 63, 74–77.
- YEATES, S.V., TUBBESING, U.H., SOLEY, J.T. & BASTIANELLO, S.S., 1990. Faculty of Veterinary Science, University or Pretoria, Pretoria. Unpublished observations.