- Infectious Diseases of Livestock
- Part 1
- Sarcocystosis
- Vectors: Ticks
- Vectors: Tsetse flies
- Vectors: Muscidae
- Vectors: Tabanidae
- Vectors: Culicoides spp.
- Vectors: Mosquitoes
- Classification, epidemiology and control of arthropod-borne viruses
- Special factors affecting the control of livestock diseases in sub-Saharan Africa
- The control of infectious diseases of livestock: Making appropriate decisions in different epidemiological and socioeconomic conditions
- Infectious diseases of animals in sub-Saharan Africa: The wildlife⁄livestock interface
- Vaccination: An approach to the control of infectious diseases
- African animal trypanosomoses
- Dourine
- Trichomonosis
- Amoebic infections
- GENERAL INTRODUCTION: COCCIDIA
- Coccidiosis
- Cryptosporidiosis
- Toxoplasmosis
- Besnoitiosis
- Sarcocystosis
- Balantidiosis
- Leishmaniosis
- Neosporosis
- Equine protozoal myeloencephalitis
- GENERAL INTRODUCTION: BABESIOSES
- Bovine babesiosis
- Equine piroplasmosis
- Porcine babesiosis
- Ovine babesiosis
- GENERAL INTRODUCTION: THEILERIOSES OF CATTLE
- East Coast fever
- Corridor disease
- Zimbabwe theileriosis
- Turning sickness
- Theileria taurotragi infection
- Theileria mutans infection
- Theileria annulata theileriosis
- Theileriosis of sheep and goats
- Theileria buffeli⁄orientalis infection
- Non-pathogenic Theileria species in cattle
- GENERAL INTRODUCTION: RICKETTSIAL, CHLAMYDIAL AND HAEMOTROPIC MYCOPLASMAL DISEASES
- Heartwater
- Lesser known rickettsial infections in animals and humans
- Chlamydiosis
- Q fever
- Eperythrozoonosis
- Bovine Haemobartonellosis
- Potomac horse fever
- GENERAL INTRODUCTION: ANAPLASMOSES
- Bovine anaplasmosis
- Ovine and caprine anaplasmosis
Sarcocystosis
This content is distributed under the following licence: Attribution-NonCommercial CC BY-NC  View Creative Commons Licence details here
View Creative Commons Licence details here
NJ Maclachlan and M-L Penrith (Editors). J P Dubey, Sarcocystosis, 2019.
Sarcocystosis
Previous authors: M B MARKUS, J J VAN DER LUGT AND J P DUBEY
Current authors:
J P DUBEY - Senior scientist, MVSc, PhD, DSc, Animal Parasitic Diseases Laboratory, U.S.Department of Agriculture, Building 1001, Beltsville Agricultural Research Center, 10300 Baltimore Avenue, Beltsville, Maryland, 20705, United States of America
Introduction
The protozoan Sarcocystis is ubiquitous worldwide. While most infections are subclinical, a few species can cause acute clinical disease in animals and even death. Some Sarcocystis species are zoonotic.
The coccidial nature of Sarcocystis first became apparent in 1970s from studies by FAYER, 197062 and 197263 in the USA and by Heydorn and Rommel90, 130 in Germany. The classification, history and nomenclature of Sarcocystis have been reviewed elsewhere.35, 51, 105 Much of the information in this chapter is extracted from a recent book on Sarcocystis.35
Aetiology and life cycle
Unlike its relatives, Toxoplasma and Neospora, Sarcocystis has many species. A recent book, listed 200 named and many unnamed species and more than 2400 references.35 However, full life cycles are known for only a few species.
The protozoan belongs to the phylum Apicomplexa (Sporozoa); class Sporozoasida; subclass Coccidiasina; order Eucoccidiorida; suborder Eimeriorina; family Sarcocystidae;subfamily Sarcocystinae; genus Sarcocystis. This generic name is derived from the cyst stage in muscle (‘sarco’ = muscle, from the Greek word for flesh). Sarcocystis spp. have the organelles that are characteristic of the phylum Apicomplexa such as apical rings (also called conoidal or preconoidal rings), polar rings, conoid, pellicle, subpellicular microtubules, micropores and micronemes. However, unlike other coccidians, Sarcocystis schizonts are located directly in the host cytoplasm and merozoites lack rhoptries.
Many species of Sarcocystis are found in domestic animals (Table 1). A two-host life cycle is involved (Figure 1): the intermediate hosts (herbivores) harbour the asexual stages of the parasite, whereas a sexual cycle takes place in carnivorous definitive hosts (Table 1). The time required for completion of the life cycle varies considerably, depending upon the species of Sarcocystis.35 The life cycle of S. cruzi will serve as an example in this chapter.30
The definitive host of S. cruzi becomes infected by ingesting muscular or neural tissue containing mature sarcocysts. Bradyzoites (cystozoites) are liberated from the ingested sarcocyst in the stomach and gut, and then penetrate the mucosa of the small intestine and transform into male (micro-) and female (macro-) gamonts. Within six hours of ingestion of infected tissue, gamonts are found within a parasitophorous vacuole (PV) in goblet cells near the tips of villi. Macrogamonts are ovoid to round, 10 to 20 μm in diameter, and contain a single nucleus. Microgamonts are ovoid to elongated and contain one or more nuclei. The microgamont’s nucleus divides and, as the microgamont matures, the daughter nuclei migrate towards the periphery of the gamont. Mature microgamonts of S. cruzi measure about 7 × 5 μm and contain 3 to 11 slender gametes. The microgametes are about 4 × 0.5 μm in size and have a compact nucleus and two flagella. Microgametes liberated from the microgamont actively move to the periphery of the macrogamont. After fertilization, a wall develops around the zygote and the oocyst is formed. The entire process of gametogony and fertilization can be completed within 24 hours and gamonts and oocysts may be found at the same time. The location of gametogony and the type of cell parasitized vary with the species of Sarcocystis and stage of gametogenesis. Sarcocystis differs from other coccidia in that no asexual multiplication takes place in the intestine prior to sexual reproduction.
In contrast to other coccidians (see Coccidiosis), Sarcocystis oocysts sporulate in the lamina propria of the gut. The inner mass of the oocyst (sporont) divides into two sporocysts. Four sporozoites are formed in each sporocyst. Sporulated oocysts are generally colourless and thin-walled and break so that free sporocysts are excreted in faeces (Figure 2). The prepatent and patent periods vary (the latter is usually long), but oocysts of most Sarcocystis species first appear in faeces between 7 and 14 days after ingestion of sarcocysts.
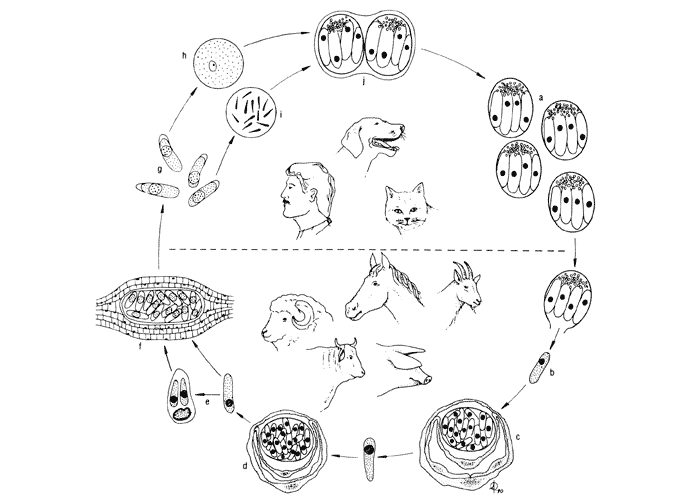
Figure 1 Life cycle of Sarcocystis cruzi. Dog, coyote and other canids are its definitive hosts and cattle and the American bison (Bison bison) are the only proven intermediate hosts.
- a = sporocysts
- b = sporozoite
- c and d = parasites undergoing schizogony in endothelial cells of blood vessels
- e = tachyzoic merozoites (free or inside mononuclear cells, in the bloodstream)
- f = sarcocyst in muscle
- g = bradyzoic merozoites
- h = macrogametocyte
- i = microgametocyte
- j = oocyst
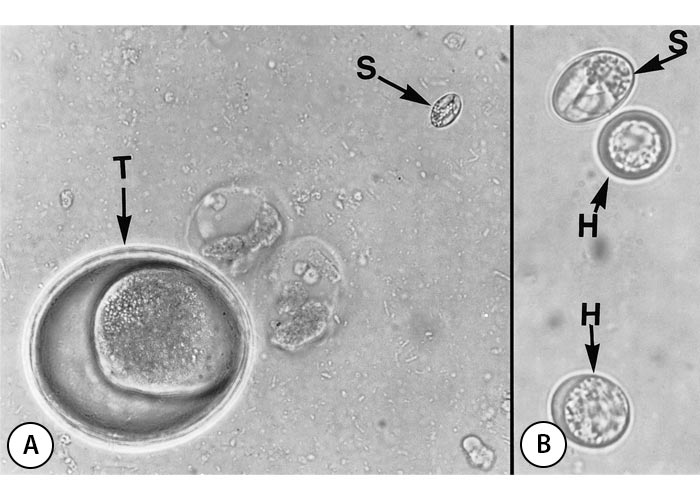
Figure 2 Faecal flotation from a dog naturally infected with Sarcocystis. A: Low magnification, to show the size of a Sarcocystis sporocyst (S) compared with a Toxascaris egg (T), x 430. B: High magnification, showing a Sarcocystis sporocyst (S) and two Neospora or Hammondia-like oocysts (H), x 1 250. (From Dubey, 197629 with permission)
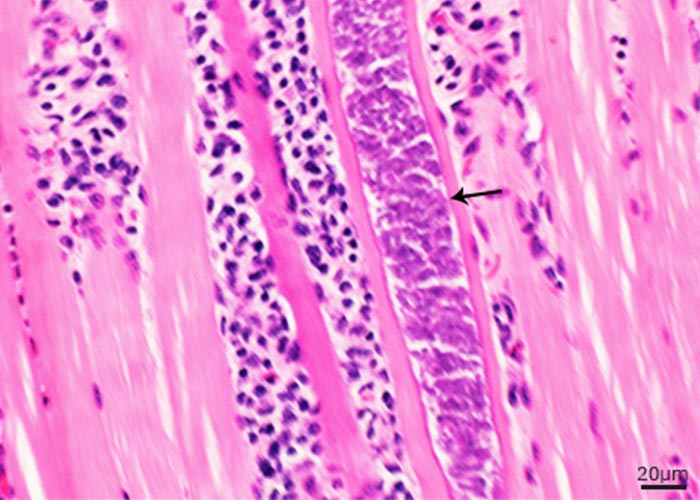
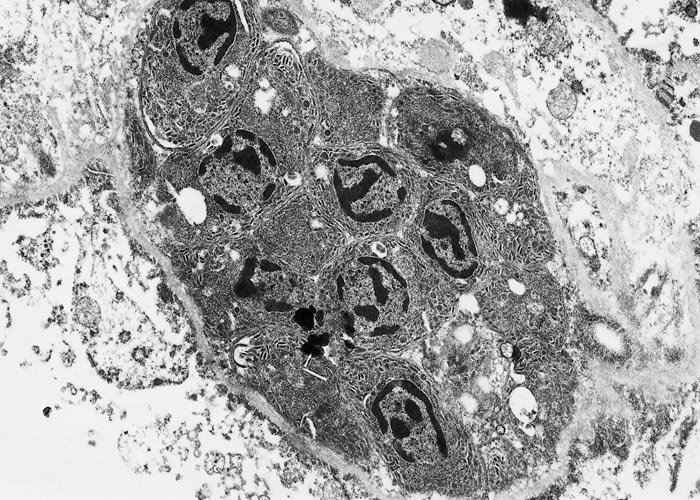
The intermediate host becomes infected by ingesting sporocysts (Figures 2 and 3). Sporozoites excyst from sporocysts in the small intestine. The fate of the sporozoites from the time of ingestion of the sporocysts until initial development in blood vessels in the mesenteric lymph nodes is not known. First-generation schizogony (merogony) begins in endothelial cells of arteries as early as seven days post-inoculation (dpi) and may be completed as early as 15 dpi (Figure 3A). Second-generation schizonts have been seen in vascular endothelium from 19 to 46 dpi, predominantly in capillaries but also in small arteries virtually throughout the body (Figures 3B and C). These schizonts seem to be most numerous in the glomeruli of the kidneys. Cell division in schizonts is by endopolygeny, which is a specialized form of schizogony. The nucleus becomes lobulated and divides into several daughter nuclei (up to 37). Merozoites are formed at the periphery (Figure 4). The shape and size of schizonts vary considerably. Both first- and second-generation schizonts develop within the host cell’s cytoplasm and are not surrounded by a PV. Merozoites of Sarcocystis do not have rhoptries.51 Merozoites are found in peripheral blood from 24 to 46 dpi (Figure 3D and E), and coincide with the maturation of second-generation schizonts. Merozoites in blood are extracellular or located within unidentified mononuclear cells. Intracellular merozoites contain one or two nuclei and some divide into two, apparently by endodyogeny. The number of generations of schizogony and the type of host cell in which it occurs varies, depending on the species of Sarcocystis concerned. All species of Sarcocystis of large domestic livestock (cattle) form first- and second-generation schizonts in vascular endothelium, whereas only a single precystic generation of schizogony has been found in Sarcocystis species of small mammals (rodents), and this is usually in hepatocytes. Merozoites that are liberated following the final cycle of schizogony initiate development of sarcocysts in muscles (Figure 3F). Cysts are rare in extra-muscular tissues. The intracellular merozoite surrounded by a PV becomes circular to ovoid, forming a metrocyte which undergoes repeated divisions. Eventually the sarcocyst is filled with bradyzoites, which are the infective stage for the predator. Sarcocysts generally become infectious at about 75 dpi but in this there is considerable variation among species of Sarcocystis. Sarcocysts may disappear from a host with passing of time.32 Neither schizonts nor immature sarcocysts containing only metrocytes give rise to intestinal infection in the definitive host. There is no lactogenous transmission.
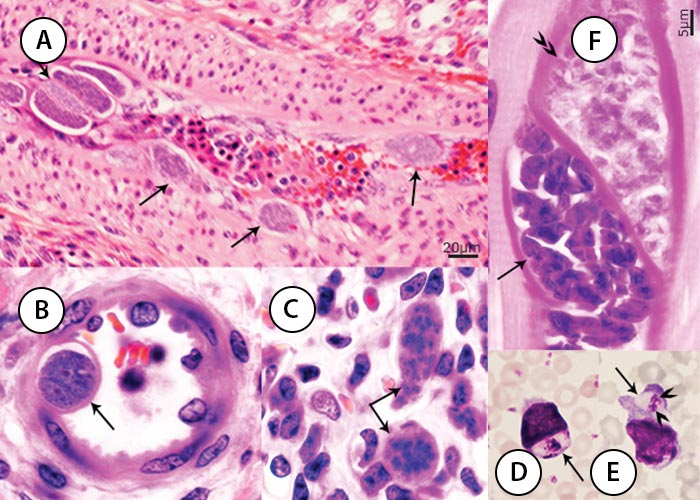
Figure 3 Endogenous stages of S. cruzi in tissues of experimentally infected cattle. A, B, C and F, haematoxylin and eosin stain, D and E, Giemsa stain. (A) First generation schizonts (arrows) in a renal artery. (B) Immature second generation schizont (arrow) protruding in the lumen of an arteriole. (C) Immature and mature second generation schizonts (arrows) in a renal glomerulus. (D) Uninucleate merozoite in a circulating monocyte. (E) Binulceate merozoite in a circulating leukocyte. (F) Immature (double arrow heads) and mature (arrow) sarcocysts apparently within one myocyte. Immature sarcocyst contain metrocytes that stain faintly with haematoxylin and eosin, compared with mature sarcocysts containing deeply stained bradyzoites.
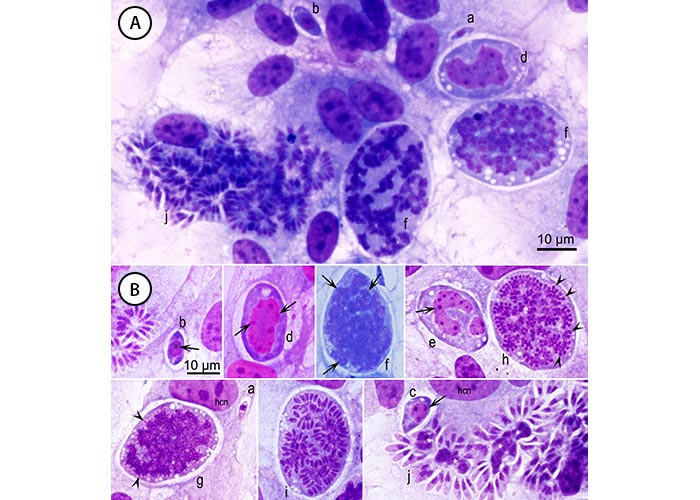
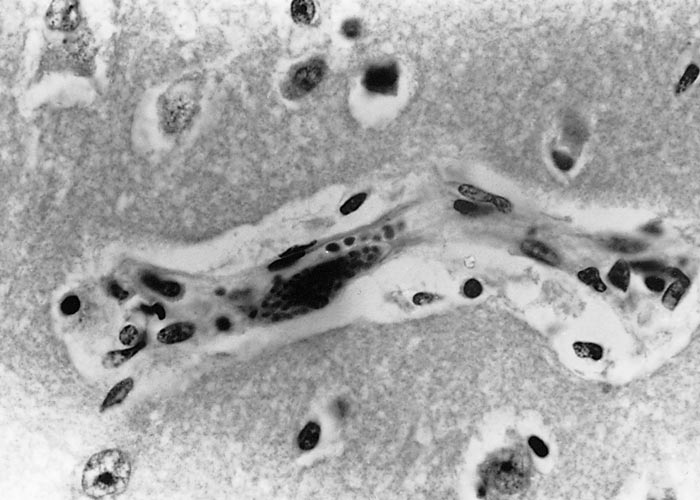
Figure 4 Development of Sarcocystis schizonts by endopolygeny. Sarcocystis falcatula of birds in bovine turbinate cells are used as example. Giemsa stain. hcn = host cell nucleus. There is considerable variability in staining of different stages of the parasite. (A) Different developmental stages in one field. (B) Stages selected from different fields to show details. Stages (a-k), in presumed order of development. Intracellular merozoite (a) with a single nucleus, earliest schizont (b) with a prominent nucleus and 1 nucleolus (arrow), an immature schizont with 3 nucleoli (c), and a schizont with 7 nucleoli (d). Thereafter the nucleus has become lobulated (e, f). In (g, h) ends of merozoites are forming. In (i) the merozoites are arranged in different groups. In (j) merozoites have formed around residual bodies (*). Note different shapes and sizes of merozoites, some are more slender than others. (From Dubey et al., 201635)
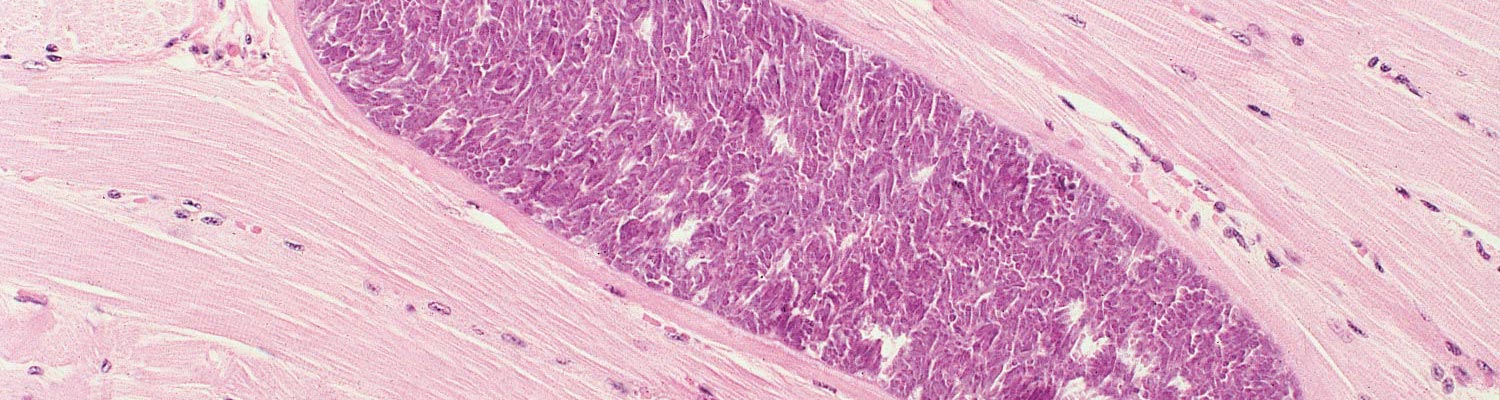
Sarcocysts vary in size and shape, depending on the species of the parasite. Some remain microscopic, whereas others become macroscopic (Figures 5 and 6). Sarcocysts usually take a long time to develop to a macroscopic size: for instance it tends to take more than a year for S. gigantea to develop fully. In general, species transmitted by cats can become macroscopic but that may take years.69, 123 It is probably for this reason that the cat-transmitted Sarcocystis may become extinct in the modern livestock management practices, where animals less than one year old are sent to slaughter. Microscopic sarcocysts vary from long and narrow to short and wide. Macroscopic sarcocysts appear filamentous, like rice grains, or globular. Sarcocysts are always located within a PV in the cytoplasm of the host cell. More than one sarcocyst may be found in the same host cell. A cyst wall surrounds the parasite’s metrocyte or bradyzoite stages. The structure and thickness of the cyst wall differ among species of Sarcocystis and within each species as the sarcocyst matures.
A connective tissue wall (secondary sarcocyst wall) surrounds sarcocysts of some species (eg. S. gigantea). Histologically, the sarcocyst wall is smooth, striated or hirsute, or it may possess complex, branched protrusions (Figure 6D). Internally, groups of bradyzoites are frequently seen to be divided into compartments by septa that originate from the sarcocyst wall. Septa are present in all but a few species of Sarcocystis. The structure of the parasites within the sarcocysts changes with the maturation of the sarcocyst. Immature sarcocysts contain metrocytes (mother cells). Each metrocyte divides into two by endodyogeny. After what appear to be several such divisions, some of the metrocytes, through the process of endodyogeny, produce banana-shaped parasites that are now termed bradyzoites. They contain prominent amylopectin granules that stain bright red in the periodic acid-Schiff (PAS) reaction. Even mature sarcocysts may contain some peripherally arranged metrocytes.
The ultrastructure of the mature sarcocyst wall is of taxonomic value. Sarcocyst development begins when a merozoite enters a myocyte or neural cell, as explained above. The merozoite resides in a PV and is surrounded by a parasitophorous vacuolar membrane (PVM). The PVM has villar protrusions that vary in shape and size (Figure 6). Below the PVM is a granular layer. Sarcocyst walls have been categorized into more than 42 types with subdivisions within each type.35
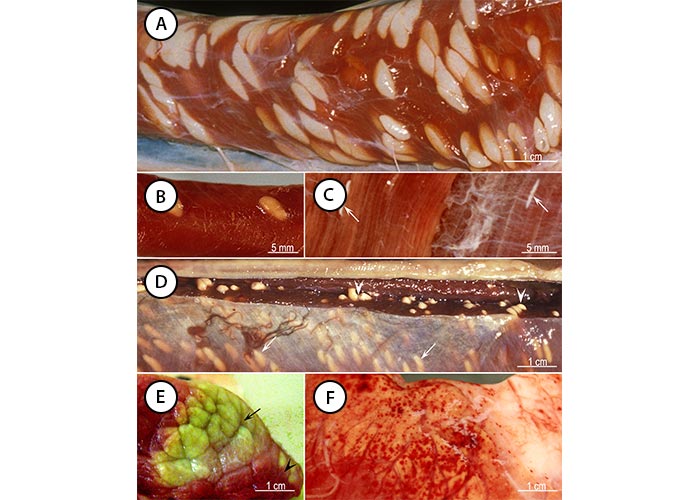
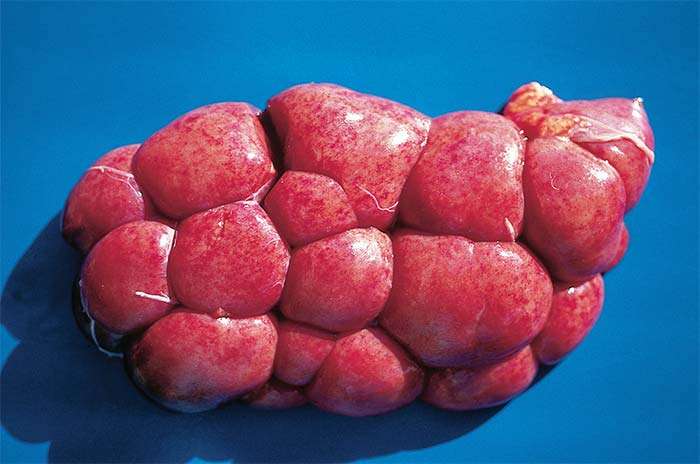
Figure 5 Sarcocystis-associated gross lesions in animals. (A) Sarcocysts of S. fusiformis outer surface of the oesophagus of a naturally infected water buffalo in Vietnam. (B) Sarcocysts of S. gigantea outer surface of the oesophagus of a naturally infected sheep in Sweden. (C) Sarcocysts (arrows) of S. hirsuta in muscles of the flank of a naturally infected cow in USA. (D) Sarcocysts of S. rileyi in pectoral muscles of a naturally infected mallard (Anas platyrhynchos) in Sweden. Arrowheads point to sarcocysts. Arrows point to sarcocysts visible through the connective tissue. (E) Eosinophilic myositis in skeletal muscles of a naturally infected bovine in Belgium. Arrow points to a large greenish area and arrowhead points to smaller lesions. (F) Haemorrhages in the epicardium of a calf experimentally infected with S. cruzi. (For sources see Dubey et al., 201635).
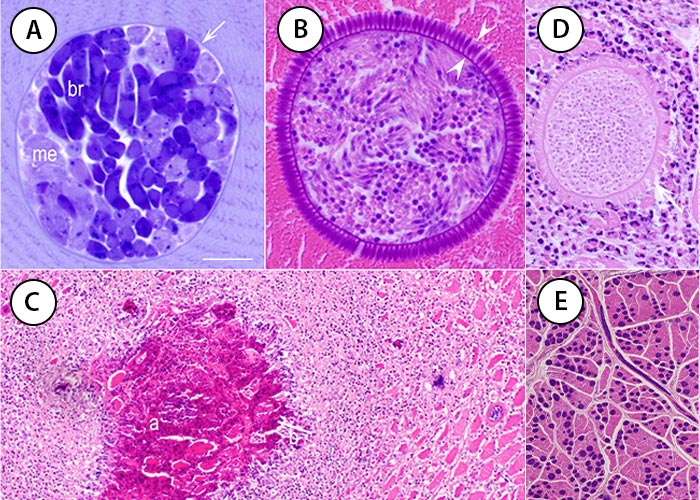
Figure 6 Sarcocysts and associated lesions. A, toluidine blue stain. B-E, haematoxylin and eosin stain. Bar applies to all parts; bar in A and B = 10 μm, in C = 200 μm, in D = 25 μm, and in E = 350 μm. (A) S. muris in a muscle of an experimentally infected Mus musculus muscle. Note the thin sarcocyst wall (arrow), faintly stained metrocytes (me), and intensely stained bradyzoites (br). (B) Sarcocystis sp. in a skeletal muscle of a naturally infected turkey. (C) Eosinophilic myositis in a naturally infected bovine. Note an area of central necrosis (a), a peripheral rim of inflammatory cells (b) and an intact sarcocyst (c). (D) A degenerating S. hominis-like sarcocyst in an area of eosinophilic myositis. The bradyzoites degenerated but the sarcocyst wall is mostly intact. Numerous S. cruzi sarcocysts in the tongue of an experimentally infected asymptomatic calf. (For sources see Dubey et al., 201635).
Epidemiology
Sarcocystis occurs commonly in the muscles of cattle, sheep, pigs, horses and goats worldwide.35
Species of Sarcocystis of domestic animals (Table 1) are generally more specific for their intermediate hosts than for their definitive hosts.35 As an example, for S. cruzi, only cattle and the American bison (Bison bison) are intermediate hosts whereas several canids are its definitive hosts, including dogs, coyotes, raccoon, foxes, wolves, and jackals.
The long patent period of several months in definitive hosts contributes to the high prevalence of Sarcocystis infection in livestock, as it provides ample opportunity for contamination of pastures with sporocysts that are resistant to environmental conditions.35, 110, 111, 136 Long-term production of sporocysts by definitive hosts is enhanced by the fact that the latter are susceptible to repeated reinfection.
Sarcocysts occur in many species of wild mammals and avian species.108, 124, 141, 153 Recently, a new species, S. cafferi (Figure 7) was described from the African buffalo (Syncerus caffer) and distinguished from the species in the water buffalo; its life cycle is unknown but lions are suspected as the definitive host.43 Sarcocysts were found previously in African buffalo but not named. Sarcocysts were detected in histological sections of 22 (66.6 per cent) of 33 African buffalo in the Kruger National Park in South Africa but not in any of the seven young (7-10- months old) animals.127 Two new species of Sarcocystis, S. dubeyella and S. phacochoeri, were reported from warthog (Phacochoerus aethiopicus),141 and two new species, S. klaseriensis, and S. camelopardalis, were described in giraffe (Giraffa camelopardalis)6 in the Kruger National Park.
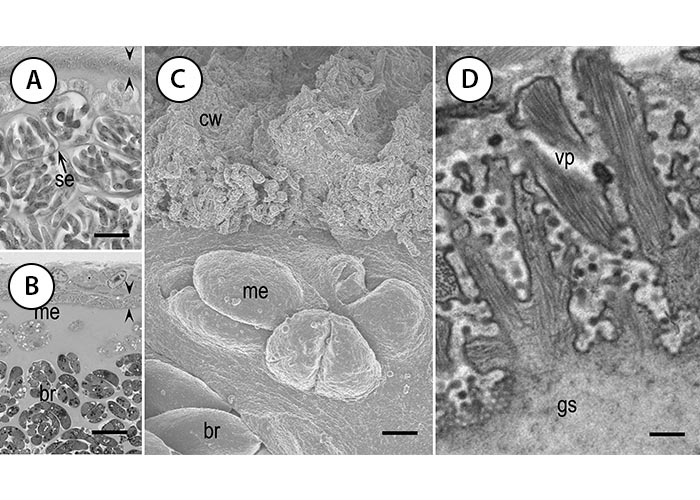
Figure 7 Structure of sarcocyst wall of S. cafferi from the African buffalo (Syncerus caffer). A and B, light microscopy; C, SEM; and D, TEM. Bar in A and B = 10 μm, in C = 2 μm, and in D = 300 nm. Note thin cyst wall (opposing arrowheads) in A and B. (A) Haematoxylin and eosin stain, 5 μm section. (B) Toluidine blue stain, 1 μm section. (C) Surface and side views of the sarcocyst wall. (D) Branching of vp. Also note metrocytes (me), bradyzoites (br), and septa (se). (From Dubey et al., 201443).
Pathogenesis
The pathogenicity of the different species of Sarcocystis in livestock varies. After ingestion of large dose (100 000 or more), animals can develop acute clinical sarcocystosis associated with the intravascular (schizogonic) phase of the life cycle.49 Direct or indirect vascular damage can lead to haemorrhages, hypoproteinaemia, anaemia, and low packed cell volume.49, 51 The serum bilirubin level is elevated during the acute stage of the infection, and the blood urea nitrogen levels are elevated in cases with terminal disease. Serum levels of enzymes such as aspartate aminotransferase, creatine kinase and lactic dehydrogenase tend to be elevated in acute and subacute sarcocystosis.
The pathogenesis of eosinophilic myositis is considered to be immune- mediated.59, 75, 81, 82, 150, 151 Immune responses, growth regulating harmones, prostaglandins and cytokine regulation are thought to be involved in the pathogenesis of chronic sarcocystosis.24-26, 66
Clinical signs and pathology
Cattle
Table 1 Sarcocystis species in livestock
| INTERMEDIATE HOSTS | SARCOCYSTIS SPECIES | DEFINITIVE HOSTS | CYST TYPE IN LONGSTANDING INFECTIONS | LIGHT MICROSCOPIC APPEARANCE OF CYST WALL | ULTRA-STRUCTURAL WALL TYPE | PERTINENT REFERENCES |
| Cattle | S. cruzi* | Dog, coyote, fox, jackal, raccoon, wolf | Microscopic | Thin | 7a | 30, 35 |
| S. hirsuta** | Cat | Macroscopic | Thick | 28 | 13, 31, 51 | |
| S. hominis | Human, other primates | Not known | Thick | 10b | 38, 51, 113 | |
| S. heydorni | Human | Microscopic | Thin | 29 | 56, 94 | |
| S. rommeli | Unknown | Microscopic | Thick | Unclassified | 44, 93 | |
| Sheep | S. arieticanis | Dog | Microscopic | Thin | 7b | 89 |
| S. tenella* | Dog, coyote, fox | Microscopic | Thick | 14 | 48, 60, 87 | |
| S. gigantea | Cat | Macroscopic | Thick | 21a | 119-121 | |
| S. medusiformis | Cat | Macroscopic | Thin | 20 | 21, 121, 123 | |
| S. mihoensis | Dog | Macroscopic | Thick | 39 | 133 | |
| Goats | S. capracanis* | Dog, coyote, fox | Microscopic | Thick | 14 | 50, 58, 86, 121 |
| S. hircicanis | Dog | Microscopic | Thin | 7a | 91 | |
| S. moulei | Cat | Macroscopic | Thick | 21 | 1, 88 | |
| Pigs | S. miescheriana | Dog, fox, jackal, raccoon | Microscopic | Thick | 10b | 80 |
| S. suihominis | Human, other primates | Microscopic | Thick | 31 | 92, 114 | |
| S. porcifelis (uncertain) | Cat | Macroscopic | Not known | Unknown | 29, 35 | |
| Horses and donkeys | S. bertrami | Dog | Microscopic | Thin | 11c | 35, 57, 109, 155 |
| S. fayeri | Dog | Macroscopic | Thick | 11a | 53, 65, 135 | |
| Water buffalo*** |
| |||||
| S. buffalonis | Cat | Macroscopic | Thick | 28 | 95 | |
| S. dubeyi | Unknown | Macroscopic | Thick | 10c | 97 | |
| S. fusiformis | Cat | Macroscopic | Thin | 21b | 41 | |
| S. levinei | Dog | Microscopic | Thin | 7a | 28, 96 | |
| S. dehongensis | Macroscopic | Thick | 24 | 17 | ||
| Camels | S. cameli | Dog | Macroscopic | Thin | 9j | 33, 40 |
| S. ippeni | Dog | Microscopic | Thin | 32 | 40 |
*Considered pathogenic.
** Described a new species S. bovini, and resurrected S. bovifelis based on review of literature of cat transmitted Sarcocystis species in cattle; These reports need confirmation.78, 79
*** There is considerable confusion concerning identity of some Sarcocystis species in buffalo in China.37
The earliest report of acute bovine sarcocystosis was in 1961 in a dairy herd,23 before the discovery of the life cycle of Sarcocystis. Within eight weeks a total of 25 animals were affected and 17 died. Acutely ill cows had intermittent anorexia, drop in milk yield, diarrhoea, transient fever, nasal discharge, hypersalivation, and haemorrhagic vaginitis. Of the 17 pregnant cows, 10 aborted in the last trimester of pregnancy and only one of the 10 survived. Erosions were found on the tongue and buccal mucosa of the salivating cows. Chronic cases were marked by emaciation, pale or icteric mucous membranes, mandibular oedema, exophthalmos, cessation of lactation, and sloughing of the tail switch. Cachectic animals became recumbent, with muscle tremors over the body resembling those of hypocalcaemia. Numerous schizonts were present in the endothelium of many organs in 11 of 16 animals. Because neither the stage nor clinical signs of sarcocystosis had been described at that time, this outbreak was called Dalmeny disease, after the small Canadian town in which it occurred. In retrospect, however, it was an outbreak of acute sarcocystosis.23
Subsequently, cases of natural clinical sarcocystosis in cattle have been reported in Canada,112 England,18 Ireland,19 Norway,103 Australia,11 and the USA.72, 73, 77, 101, 116 Unlike Dalmeny disease, these were infections in yearlings or younger calves. A well-documented outbreak was reported in Kentucky, USA.77 Approximately 30 of 41 heifers in a one hectare lot had diarrhoea, were losing weight and consuming less feed, and had rough coats. The condition worsened to include severe laminitis, hypersalivation, extreme nervousness, and marked hair loss from the ears, lower limbs, and the tail switch, giving a "rat tail" appearance. Within a month, more than half of the animals were severely debilitated and eight died. In the next five weeks, two thirds died or were euthanized, only a third were salvaged for market. Contamination of hay with faeces of farm dogs was implicated as the source of infection.72, 77
Some species of Sarcocystis can be transmitted transplacentally resulting in abortion, stillbirth, or birth of weak animals.However, transplacental transmission of Sarcocystis is rare.
Sporadic cases of bovine abortion and neonatal mortality have been reported in New Zealand,117, 146, 152 Australia,101, 117, 118 Canada,23 the USA4, 31, 34, 102, 138 and Cuba,22 in which foetal lesions and or protozoa were found (Figure 9). Foetal encephalitis, myocarditis, and hepatitis were often associated with the outbreaks. Intravascular schizonts resembling those of Sarcocystis were found in some of the foetuses in all reported outbreaks. Sarcocystis-associated abortion has been recognized in less than one per cent of abortions.4 An unusual necrotizing encephalitis was found in two cattle in South Africa.149 A 2-year-old heifer developed convulsions and died suddenly. Eleven of 15 young calves developed severe clinical signs over five days, and one calf died with neurological signs. Two of these animals were necropsied. Multiple areas of necrosis and vasculitis were found in the brains in both animals. Schizonts, considered to be first generations S. cruzi, were identified in blood vessels in affected areas. In all other reports, clinical infections were associated with second generation schizonts of S. cruzi.
Isolated cases of fatal protozoal encephalitis were reported from Canada,45 Germany36, 145 and England83 but details are scanty. The case from Germany reported in 1984 was restudied 32 years later by Dubey 2016.36 A nine- month-old bull was suspected to have rabies because of neurological signs. It had high fever and oedema of eyes. The bull died after a 10-day illness; only brain was removed at necropsy. Severe leptomeningoencephalitis was detected histologically and it had many merozoites free in the neural parenchyma and some in neurons (Figure 10) and many intravascular schizonts.
Gross lesions at necropsy of acute to subacute sarcocystosis include serous atrophy and necrosis of fat, pallor of the mucous membranes, an icteric appearance of tissues and organs, hydropericardium, hydrothorax, ascites, and widely distributed petechiae and ecchymoses especially on serosal surfaces of the viscera (Figure 10), and throughout the skeletal and cardiac musculature, which may also show multiple pale foci of variable size or haemorrhagic streaks. Skeletal muscles are often pale pink with dark red mottling. Lymph nodes are enlarged and oedematous and often contain petechiae. The mononuclear cell infiltrate is frequently accompanied by haemorrhage, oedema, multifocal degeneration and necrosis (especially in the cardiac and skeletal musculature), vasculitis and, less commonly, thrombosis. Muscular lesions in acute sarcocystosis vary in severity from muscle to muscle and within individual muscles.51
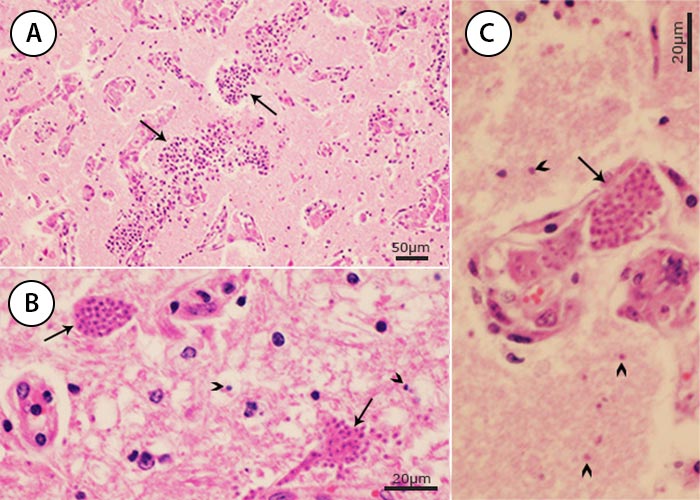
Figure 10 Sarcocystis encephalitis in a bull calf from Germany. (A) Neovascularization and perivascular cuffings. Many organisms are present among inflammatory cells but not visible at this magnification. (B) Merozoites in neurons (arrows) and dispersed in neural parenchyma (arrowheads). (C) Intravascular (arrow) and extravascular schizonts. (From case reported by Dubey et al., 2016).36
In chronically infected livestock animals, sarcocysts occur within myocytes of the myocardium and skeletal muscles, and sometimes in the brain.7, 52 A tissue response is not normally apparent in the muscles.
Eosinophilic myositis (EM) is a subclinical inflammatory condition that is sometimes encountered in livestock (particularly cattle, occasionally sheep and rarely pigs and horses) at abattoirs.2, 9, 14, 15, 27, 35, 51, 55, 59, 68, 75, 76, 81, 82, 98, 100, 104, 128, 129, 132, 150 The affected animals usually show no clinical signs. Sex and breed have no influence on the prevalence and it has been reported with equal frequency in steers, cows, and heifers. Virtually all striated muscles, including skeletal muscles, muscles of the eye, larynx, and heart can be affected.100 In the study in Belgium of 97 cattle samples condemned because of this condition, sarcocysts were found in 27 cases and most (82 per cent) of the sarcocysts were S. hominis.150
The macroscopical lesions of EM consist of greenish (Figure 5E) or yellowish areas or granulomas (measuring up to 5–15 × 1–3 mm) in myocardial and skeletal muscles. These lesions often result in condemnation of affected carcasses. Early microscopic lesions are characterized by infiltration of numerous eosinophils, which are responsible for the green colour, and degeneration and necrosis of myocytes and fibrosis in the more chronic lesions (Figure 6C). The granulomas have a central area containing necrotic cellular debris that is surrounded by and infiltrateconsisting of eosinophils, epithelioid cells, fibroblasts, giant cells and lymphocytes.
Sheep
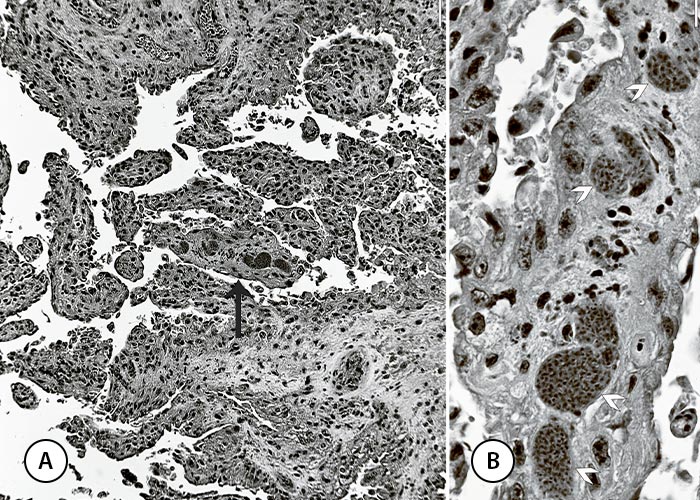
There are three reports of neonatal sarcocystosis in naturally-infected sheep from Brazil, Australia, and Denmark. In Brazil, Sarcocystis-associated mortality was reported in sheep.126 In a flock of Corriedale sheep eight ewes aborted or had stillborn lambs. One stillborn lamb was necropsied. Intravascular schizonts were associated with encephalitis and myocarditis and were also present in the kidneys. A similar caseof Sarcocystis-associated mortality was observed in a stillborn lamb in Denmark.3 The predominant lesions were encephalitis (Figure 12a and b) and placentitis associated with schizonts. The encephalitis was characterized by multiple inflammatory foci throughout the brain but predominantly in the cerebrum. Foci of necrosis were surrounded by a zone of microglia and mononuclear cells. Immature and mature schizonts were seen in vascular endothelium, sometimes occluding the lumen. Placental lesions consisted of multiple foci of necrosis, inflammation, and mild calcification; numerous schizonts and merozoites were seen within the lesions. In Australia, neonatal sarcocystosis was found in a three-week-old lamb with generalized gliosis, and immature and mature schizonts in the brain.39
Severe encephalomyelitis has been reported in sheep from Australia, Bulgaria, Canada, England, Hungary, Turkey, and the USA and it is not clear if these were caused by S. tenella or another unknown species of Sarcocystis because schizonts were located extravascularly in neural cells.10, 39, 52, 67, 85, 115, 122, 125, 134, 140, 142, 154 Another unususal case of sarcocystosis associated with pneumonitis in a lamb of five to seven months old was reported in England.139
Severe Sarcocystis-associated myositis was reported in sheep in England.5, 8, 46, 51, 70, 84, 99, 140, 156 A case of laryngeal hemiplegia in an 18-month-old ram with respiratory distress and poor body condition associated with Sarcocystis sp. occurred in Spain.131
Whole sheep carcasses or parts of them were condemned because of the presence of macroscopic sarcocysts of S. gigantea and S. medusiformis.20 Carcasses have also been condemned for eosinophilic myositis in lambs and in adult sheep.61, 100
Goats
In Australia, 8 of 38 Saanen goats aborted or gave birth to stillborn kids over a period of three months.106, 107 The only stillborn that was necropsied showed non-suppurative encephalitis characterized by necrosis, perivascular cuffing, and schizonts in some blood vessels. Occasional schizonts were also identified in the lungs and kidneys.107 Markus et al., 2004108 reported acute sarcocystosis in a goat from South Africa (Figure 12a).
Other hosts
Clinical myositis has been associated with the presence of sarcocysts in histologic sections of horses in Germany,74, 137 a mule in Israel,144 a malnourished horse in Canada,12 an ataxic horse in California,147 and in a horse with pronounced muscle wasting in Colorado,148 both in the USA. In a controlled study of 91 horses with clinical myositis in Belgium, sarcocysts were found in 12 muscle biopsies, but there was no association between the presence of sarcocysts and clinical myositis.71
Clinical sarcocystosis has been reported in chickens, many avian species (particularly in zoos), South American camelids, and mule deer.35, 42 Sarcocysts have been found in muscles of canids, felids, and other carnivores; life cycles of these species are known and infections are considered incidental. However, clinically apparent severe myositis has been associated with two new species, S. svanai and S. caninum, in domestic dogs in Canada and the USA.16, 35, 54, 143
An unusual Sarcocystis species, S. canis, caused severe hepatitis in dogs, polar bear, black bear, sea lion, chinchilla, striped dolphin, Hawaiian monk seal, and steller sea lion.35, 47 Its life cycle is unknown and the schizont, which occurs in hepatocytes, is the only stage known.
Diagnosis
It is difficult to diagnose acute sarcocystosis because the clinical signs of disease are nonspecific and there are, at present, no definitive tests to confirm the diagnosis in the living animal.
Identification of Sarcocystis to species level is usually an academic exercise rather than a matter of practical importance, the reason being that chronic Sarcocystis infections in livestock are invariably subclinical and the prevalence of sarcocysts in the musculature of domestic herbivores is high.
Parasitological confirmation of clinically suspected disease is based on the finding of schizonts in endothelial cells of blood vessels, on microscopy of biopsy material (such as muscle or lymph node), or, more commonly, on histopathology after a necropsy. However, schizonts or free merozoites are not always detected in infected animals, either because they are too few to be found or because schizonts have ruptured by the time that the infection becomes clinically apparent. It is particularly in foetuses that the parasites are not consistently present, and the chances of diagnosing Sarcocystis-induced abortion are improved if a variety of foetal tissues are examined microscopically including samples from the brain and placenta (see Pathology). Schizonts may occur in vascular endothelial cells in the subepithelial connective tissue of placental cotyledons.
Ultrastructurally, the merozoites of Sarcocystis are like other apicomplexan zoites (see Aetiology and life cycle and Differential diagnosis), except that they lack rhoptries. Immunohistochemistry is helpful in identifying schizonts in tissue sections.
Although various serological tests (indirect fluorescent antibody test and enzyme-linked immunosorbent assay) have been described, they are of limited value. Few laboratories keep antigen as it is not commercially available. Results are difficult to interpret because subclinical Sarcocystis infections are very common in livestock.39 Furthermore, there is cross-reaction in serological tests between pathogenic and non-pathogenic Sarcocystis spp., although Sarcocystis is largely antigenically distinct from other coccidia.
A search for the small sporocysts of Sarcocystis in the faeces of carnivores (particularly dogs) by centrifugal flotation, using a super-saturated sodium chloride solution, can help in the aetiological evaluation of suspected acute sarcocystosis. What should be taken into consideration is that the prepatent period in carnivores is one to two weeks, and that the duration of the patent period can be several months.
The internal structure of a sporocyst (containing four sporozoites and a granular sporocystic residuum) can be seen when an oil immersion objective is used.
Differential diagnosis
Known pathogens of livestock that closely resemble Sarcocystis morphologically are Toxoplasma gondii (see Toxoplasmosis) and Neospora spp. (see Neosporosis). In histological sections, Sarcocystis can be differentiated from these protozoa by the presence of schizonts in vascular endothelial cells, whereas Neospora and Toxoplasma can develop in a wide range of cell types. The presence of schizonts in numerous endothelial cells of blood vessels is suggestive of a Sarcocystis infection.
On transmission electron microscopy of Sarcocystis schizonts and merozoites are seen to be free in the host cell cytoplasm (i.e. no PV vacuole is evident). Groups of Toxoplasma organisms, on the other hand, are surrounded by a PVM. Merozoites of Sarcocystis do not have the organelles known as rhoptries that are observed in Neospora and Toxoplasma. Immature schizonts with multiple nuclei not surrounded by discrete cytoplasmic areas are seen in sarcocystosis. Multinucleated stages do not occur in Neospora and Toxoplasma infections because division is by endodyogeny, each parasite giving rise to two daughter organisms.
Tissue cysts of both Neospora and Toxoplasma lack the cyst wall protrusions that are characteristic of sarcocysts (see Aetiology and life cycle). Septa are present in most Sarcocystis species but are absent in Toxoplasma and Neospora tissue cysts. Sarcocysts of some species of Sarcocystis (eg. S. cruzi) are divided into compartments by septa, unlike tissue cysts of Neospora and Toxoplasma. There are metrocytes (ovoid, rapidly multiplying organisms) in young sarcocysts (and sometimes a few in older ones) but these do not occur in cysts of Neospora or Toxoplasma.
Control
The life cycle of Sarcocystis is dependent upon the predator–prey relationship that exists between its definitive and intermediate hosts. Consequently, if meat eaten by the definitive hosts has been cooked, the transmission cycle is broken.
To effectively control Sarcocystis infection on farms, carcasses of dead livestock should be buried or burned. Conventional cooking and freezing in a household freezer (-12°C) kills sarcocysts.64
At present, no vaccine is available to protect livestock against acute sarcocystosis.
Although prophylactic administration of some drugs (amprolium, salinomycin and monensin) can reduce the clinical effects of acute sarcocystosis, none of these regimens is practical because exposure to Sarcocystis is unpredictable.51
References
- ABDEL GHAFFAR, F. A., HEYDORN, A. O. & MEHLHORN, H., 1989. The fine structure of cysts of Sarcocystis moulei from goats. Parasitology Research, 75, 416-418.
- ABDIRAHMAN, O. M., FINAZZI, M., SCANZIANI, E. & SIRONI, G., 1991. Reperti de miosite eosinofilica in bovini regolarmente macellati. Archivo Veterinario Italiano, 42, 97-104.
- AGERHOLM, J. S. & DUBEY, J. P., 2014. Sarcocystosis in a stillborn lamb. Reproduction in Domestic Animals, 49, e60-e63.
- ANDERSON, M. L., BLANCHARD, P. C., BARR, B. C., DUBEY, J. P., HOFFMAN, R. L. & CONRAD, P. A., 1991. Neospora-like protozoan infection as a major cause of abortion in California dairy cattle. Journal of the American Veterinary Medical Association, 198, 241-244.
- BELCHEV, L. & ARNAOUDOV, D., 1987. A rare case of sarcocystic encephalitis in a sheep [in Russian]. Veterinarnomeditsinski, 24, 33-37.
- BENGIS, R. G., ODENING, K., STOLTE, M., QUANDT, S. & BOCKHARDT, I., 1998. Three new Sarcocystis species, Sarcocystis giraffae, S. klaseriensis, and S. camelopardalis (Protozoa: Sarcocystidae) from the giraffe (Giraffa camelopardalis) in South Africa. Journal of Parasitology, 84, 562-565.
- BIGALKE, R. D. & TUSTIN, R. C., 1960. The occurrence of a cyst of Sarcocystis Lankester 1882 in the cerebellum of an ox. Journal of the South African Veterinary Association, 31, 271-274.
- BRUMLOOP, A. & SAGER, M., 1993. Sarkosporidien als Ursache von plötzlichen Todesfällen in der Narkose beim Schaf. Berliner und Münchener Tierärztliche Wochenschrift, 106, 80-83.
- BUNDZA, A. & FELTMATE, T. E., 1989. Eosinophilic myositis/lymphadenitis in slaughter cattle. Canadian Veterinary Journal, 30, 514-516.
- CALDOW, G. L., GIDLOW, J. R. & SCHOCK, A., 2000. Clinical, pathological and epidemiological findings in three outbreaks of ovine protozoan myeloencephalitis. Veterinary Record, 146, 7-10.
- CARRIGAN, M. J., 1986. An outbreak of sarcocystosis in dairy cattle. Australian Veterinary Journal, 63, 22-24.
- CAWTHORN, R. J., CLARK, M., HUDSON, R. & FRIESEN, D., 1990. Histological and ultrastructural appearance of severe Sarcocystis fayeri infection in a malnourished horse. Journal of Veterinary Diagnostic Investigation, 2, 342-345.
- CAWTHORN, R. J., MARKHAM, R. J. F., HITT, N. D. & DESPRES, D., 1990. In vitro cultivation of the vascular phase of Sarcocystis hirsuta (Apicomplexa). Canadian Journal of Zoology, 68, 1068-1070.
- CERETTO, F., JULINI, M., CRAVERO, G. & COSCIA, G., 1987. Reperti ispettivi della sarcosporidiosi in bovini da macello. Obiettivi e Documenti Veterinari, 8, 67-71.
- CHAMBERS, P. G., 1987. Carcase and offal condemnations at meat inspection in Zimbabwe. Zimbabwe Veterinary Journal, 18, 11-18.
- CHAPMAN, J., MENSE, M. & DUBEY, J. P., 2005. Clinical muscular sarcocystosis in a dog. Journal of Parasitology, 91, 187-190.
- CHEN, X., WEN, T., HU, J., LIU, T., ESCH, G. W., LIANG, Y., LI, H. & HUANG, S., 2017. Sarcocystis dehongensis n. sp. (Apicomplexa: Sarcocystidae) from water buffalo (Bubalus bubalis) in China. Parasitology Research, 116, 2145-2150.
- CLEGG, F. G., BEVERLEY, J. K. A. & MARKSON, L. M., 1978. Clinical disease in cattle in England resembling Dalmeny disease associated with suspected Sarcocystis infection. Journal of Comparative Pathology, 88, 105-114.
- COLLERY, P. & WEAVERS, E., 1981. An outbreak of Sarcocystis in calves in Ireland. Irish Veterinary Journal, 35, 159-162.
- COLLINS, G. H., 1980. Host reaction to Sarcocystis in goats. New Zealand Veterinary Journal, 28, 244.
- COLLINS, G. H., ATKINSON, E. & CHARLESTON, W. A. G., 1979. Studies on Sarcocystis species III. The macrocystic species of sheep. New Zealand Veterinary Journal, 27, 204-206.
- CORDOVÈS CÉSPEDES, C. O., MERNIO, M. & FERNÁNDEZ ÁLVAREZ, B., 1981. Primer reporte de Sarcocystis en fetos bovinos. Academia Ciencias Cuba de Serologia y Biologia, 155, 2-6.
- CORNER, A. H., MITCHELL, D., MEADS, E. B. & TAYLOR, P. A., 1963. Dalmeny disease. An infection of cattle presumed to be caused by an unidentified protozoon. Canadian Veterinary Journal, 4, 252-264.
- DAUGSCHIES, A., ALTFELD, E. & ROMMEL, M., 1989. Hemostatic alterations in pigs fed sublethal doses of Sarcocystis miescheriana. Veterinary Parasitology, 34, 1-13.
- DAUGSCHIES, A., HINTZ, J., HENNING, M. & ROMMEL, M., 2000. Growth performance, meat quality and activities of glycolytic enzymes in the blood and muscle tissue of calves infected with Sarcocystis cruzi. Veterinary Parasitology, 88, 7-16.
- DAUGSCHIES, A., ROMMEL, M. & HOPPEN, H. O., 1989. Prostanoids during acute sarcocystosis in growing pigs. Parasitology Research, 76, 115-118.
- DE BOSSCHERE, H. & DUCATELLE, R., 2001. Inverse correlation between myositis eosinophilica and number of Sarcocystis cystozoites in heart tissue of cattle. Vlaams Diergeneeskundig Tijdschrift, 70, 118-123.
- DISSANAIKE, A. S. & KAN, S. P., 1978. Studies on Sarcocystis in Malaysia. I. Sarcocystis levinei n. sp. from the water buffalo Bubalus bubalis. Z. Parasitenkd, 55, 127-138.
- DUBEY, J. P., 1976. A review of Sarcocystis of domestic animals and of other coccidia of cats and dogs. Journal of the American Veterinary Medical Association, 169, 1061-1078.
- DUBEY, J. P., 1982. Development of ox-coyote cycle of Sarcocystis cruzi. Journal of Protozoology, 29, 591-601.
- DUBEY, J. P., 1982. Development the ox-cat cycle of Sarcocystis hirsuta. Proceedings of the Helminthological Society of Washington, 49, 295-304.
- DUBEY, J. P., 1983. Immunity to sarcocystosis: Modifications of intestinal coccidiosis, and disappearance of sarcocysts in dairy goats. Veterinary Parasitology, 13, 23-34.
- DUBEY, J. P., A'AIZ, N. N., MOWERY, J. D., VERMA, S. K. & CALERO-BERNAL, R., 2017. Identification of macroscopic sarcocysts of Sarcocystis cameli from one-humped camel (Camelus dromedarius) in Iraq. Journal of Parasitology, 103, 168-169.
- DUBEY, J. P. & BERGERON, J. A., 1982. Sarcocystis as a cause of placentitis and abortion in cattle. Veterinary Pathology, 19, 315-318.
- DUBEY, J. P., CALERO-BERNAL, R., ROSENTHAL, B. M., SPEER, C. A. & FAYER, R., 2016. Sarcocystosis of animals and humans. 2nd edition. CRC Press. Boca Raton, Florida, USA, 1-481.
- DUBEY, J. P., CALERO-BERNAL, R., VERMA, S. K. & MOWERY, J. D., 2016. Pathology, immunohistochemistry, and ultrastructural findings associated with neurological sarcocystosis in cattle. Veterinary Parasitology, 223, 147-152.
- DUBEY, J. P., FAYER, R., ROSENTHAL, B. M., CALERO-BERNAL, R. & UGGLA, A., 2014. Identity of Sarcocystis species of the water buffalo (Bubalus bubalis) and cattle (Bos taurus) and the suppression of Sarcocystis sinensis as a nomen nudum. Veterinary Parasitology, 205, 1-6.
- DUBEY, J. P., FAYER, R. & SPEER, C. A., 1988. Experimental Sarcocystis hominis infection in cattle: lesions and ultrastructure of sarcocysts. Journal of Parasitology, 74, 875-879.
- DUBEY, J. P., HARTLEY, W. J. & BADMAN, R. T., 1989. Fatal perinatal sarcocystosis in a lamb. Journal of Parasitology, 75, 980-982.
- DUBEY, J. P., HILALI, M., VAN WILPE, E., CALERO-BERNAL, R., VERMA, S. K. & ABBAS, I. E., 2015. A review of sarcocystosis in camels and redescription of Sarcocystis cameli and Sarcocystis ippeni sarcocysts from the one-humped camel (Camelus dromedarius). Parasitology, 142, 1481-1492.
- DUBEY, J. P., HILALI, M., VAN WILPE, E., VERMA, S. K., CALERO-BERNAL, R. & ABDEL-WAHAB, A., 2015. Redescription of Sarcocystis fusiformis sarcocysts from the water buffalo (Bubalus bubalis). Parasitology, 142, 385-394.
- DUBEY, J. P. & KISTNER, T. P., 1985. Epizootiology of Sarcocystis infections in mule deer fawns in Oregon. Journal of the American Veterinary Medical Association, 187, 1181-1186.
- DUBEY, J. P., LANE, E. P., VAN WILPE, E., SULEMAN, E., REININGHAUS, B., VERMA, S. K., ROSENTHAL, B. M. & MTSHALI, M. S., 2014. Sarcocystis cafferi, n. sp. (Protozoa: Apicomplexa) from the African buffalo (Syncerus caffer). Journal of Parasitology, 100, 817-827.
- DUBEY, J. P., MORÉ, G., VAN WILPE, E., CALERO-BERNAL, R., VERMA, S. K. & SCHARES, G., 2016. Sarcocystis rommeli, n. sp. (Apicomplexa: Sarcocystidae) from cattle (Bos taurus) and its differentiation from Sarcocystis hominis. Journal of Eukaryotic Microbiology, 63, 62-68.
- DUBEY, J. P., PERRY, A. & KENNEDY, M. J., 1987. Encephalitis caused by a Sarcocystis-like organism in a steer. Journal of the American Veterinary Medical Association, 191, 231-232.
- DUBEY, J. P. & ROSENTHAL, B. M., 2013. Sarcocystis capracanis-associated encephalitis in sheep. Veterinary Parasitology, 197, 407-408.
- DUBEY, J. P. & SPEER, C. A., 1991. Sarcocystis canis n. sp. (Apicomplexa: Sarcocystidae), the etiologic agent of generalized coccidiosis in dogs. Journal of Parasitology, 77, 522-527.
- DUBEY, J. P., SPEER, C. A., CALLIS, G. & BLIXT, J. A., 1982. Development of sheep-canid cycle of Sarcocystis tenella. Canadian Journal of Zoology, 60, 2464-2477.
- DUBEY, J. P., SPEER, C. A. & EPLING, G. P., 1982. Sarcocystosis in newborn calves fed Sarcocystis cruzi sporocysts from coyotes. American Journal of Veterinary Research, 43, 2147-2164.
- DUBEY, J. P., SPEER, C. A., EPLING, G. P. & BLIXT, J. A., 1984. Sarcocystis capracanis: Development in goats, dogs and coyotes. International Goat and Sheep Research, 2, 252-265.
- DUBEY, J. P., SPEER, C. A. & FAYER, R., 1989. Sarcocystosis of animals and man. CRC Press, Boca Raton, Florida, USA, 1-215.
- DUBEY, J. P., SPEER, C. A., MUNDAY, B. L. & LIPSCOMB, T. P., 1989. Ovine sporozoan encephalomyelitis linked to Sarcocystis infection. Veterinary Parasitology, 34, 159-163.
- DUBEY, J. P., STREITEL, R. H., STROMBERG, P. C. & TOUSSANT, M. J., 1977. Sarcocystis fayeri sp. n. from the horse. Journal of Parasitology 63, 443-447.
- DUBEY, J. P., SYKES, J. E., SHELTON, G. D., SHARP, N., VERMA, S. K., CALERO-BERNAL, R., VIVIANO, J., SUNDAR, N., KHAN, A. & GRIGG, M. E., 2015. Sarcocystis caninum and Sarcocystis svanai n. spp. (Apicomplexa: Sarcocystidae) associated with severe myositis and hepatitis in the domestic dog (Canis familiaris). Journal of Eukaryotic Microbiology, 62, 307-317.
- DUBEY, J. P., UDTUJAN, R. M., CANNON, L. & LINDSAY, D. S., 1990. Condemnation of beef because of Sarcocystis hirsuta infection. Journal of the American Veterinary Medical Association, 196, 1095-1096.
- DUBEY, J. P., VAN WILPE, E., CALERO-BERNAL, R., VERMA, S. K. & FAYER, R., 2015. Sarcocystis heydorni n. sp. (Apicomplexa: Sarcocystidae) with cattle (Bos taurus) and human (Homo sapiens) cycle. Parasitology Research, 114, 4143-4147.
- DUBEY, J. P., VAN WILPE, E., VERMA, S. K. & HILALI, M., 2016. Ultrastructure of Sarcocystis bertrami sarcocysts from a naturally infected donkey (Equus asinus) from Egypt. Parasitology, 143, 18-23.
- DUBEY, J. P., WEISBRODE, S. E., SPEER, C. A. & SHARMA, S. P., 1981. Sarcocystosis in goats: Clinical signs and pathologic and hematologic findings. Journal of the American Veterinary Medical Association, 178, 683-699.
- ELY, R. W. & FOX, J. C., 1989. Elevated IgG antibody to Sarcocystis cruzi associated with eosinophilic myositis in cattle. Journal of Veterinary Diagnostic Investigation, 1, 53-56.
- ERBER, M. & BURGKART, M., 1981. Wirtschaftliche Verluste durch Sarkosporidiose (Sarcocystis ovicanis und S. spec.) bei der Mast von Schaflämmern. Praktische Tierarzt, 62, 422-427.
- EZZI, A., GHOLAMI, M. R. & AHOURAI, P., 1992. Eosinophilic myositis associated with Sarcocystis in sheep. Archives of Razi Institute, 42/43, 65-68.
- FAYER, R., 1970. Sarcocystis: Development in cultured avian and mammalian cells. Science, 168, 1104-1105.
- FAYER, R., 1972. Gametogony of Sarcocystis sp. In cell culture. Science, 175, 65-67.
- FAYER, R., 1975. Effects of refrigeration, cooking, and freezing on Sarcocystis in beef from retail food stores. Proceedings of the Helminthological Society of Washington, 42, 138-140.
- FAYER, R. & DUBEY, J. P., 1982. Development of Sarcocystis fayeri in the equine. Journal of Parasitology, 68, 856-860.
- FAYER, R. & ELSASSER, T. H., 1991. Bovine sarcocystosis: How parasites negatively affect growth. Parasitology Today, 7, 250-255.
- FITZGERALD, S. D., JANOVITZ, E. B., KAZACOS, K. R., DUBEY, J. P. & MURPHY, D. A., 1993. Sarcocystosis with involvement of the central nervous system in lambs. Journal of Veterinary Diagnostic Investigation, 5, 291-296.
- FOGGIN, C. M., 1980. Sarcocystis infection and granulomatous myositis in cattle in Zimbabwe. Zimbabwe Veterinary Journal, 11, 8-13.
- FORD, G. E., 1986. Completion of the cycle for transmission of sarcosporidiosis between cats and sheep reared specific pathogen free. Australian Veterinary Journal, 63, 42-44.
- FORMISANO, P., ALDRIDGE, B., ALONY, Y., BEEKHUIS, L., DAVIES, E., DEL POZO, J., DUNN, K., ENGLISH, K., MORRISON, L., SARGISON, N., SEGUINO, A., SUMMERS, B. A., WILSON, D., MILNE, E. & BEARD, P. M., 2013. Identification of Sarcocystis capracanis in cerebrospinal fluid from sheep with neurological disease. Veterinary Parasitology, 193, 252-255.
- FRANSEN, J. L. A., DEGRYSE, A. D. A. Y., VAN MOL, K. A. C. & OOMS, L. A. A., 1987. Sarcocystis und chronische Myopathien bei Pferden. Berliner und Münchener Tierärztliche Wochenschrift, 100, 229-232.
- FRELIER, P., MAYHEW, I. G., FAYER, R. & LUNDE, M. N., 1977. Sarcocystosis: A clinical outbreak in dairy calves. Science, 195, 1341-1342.
- FRELIER, P. F., MAYHEW, I. G. & POLLOCK, R., 1979. Bovine sarcocystosis: Pathologic features of naturally occurring infection with Sarcocystis cruzi. American Journal of Veterinary Research, 40, 651-657.
- FREUDENBERG, F. R., 1956. Zur klinik der myositis sarcosporidica des pferdes. Tierärztliche Umschau, 11, 91-93.
- GAJADHAR, A. A. & MARQUARDT, W. C., 1992. Ultrastructural and transmission evidence of Sarcocystis cruzi associated with eosinophilic myositis in cattle. Canadian Journal of Veterinary Research, 56, 41-46.
- GAJADHAR, A. A., YATES, W. D. G. & ALLEN, J. R., 1987. Association of eosinophilic myositis with an unusual species of Sarcocystis in a beef cow. Canadian Journal of Veterinary Research, 51, 373-378.
- GILES, R. C., TRAMONTIN, R., KADEL, W. L., WHITAKER, K., MIKSCH, D., BRYANT, D. W. & FAYER, R., 1980. Sarcocystosis in cattle in Kentucky. Journal of the American Veterinary Medical Association, 176, 543-548.
- GJERDE, B., 2016. Molecular characterisation of Sarcocystis bovifelis, Sarcocystis bovini n. sp., Sarcocystis hirsuta and Sarcocystis cruzi from cattle (Bos taurus) and Sarcocystis sinensis from water buffaloes (Bulbalus bulbalis). Parasitology Research, 115, 1473-1492.
- GJERDE, B., 2016. The resurrection of a species: Sarcocystis bovifelis Heydorn et al., 1975 is distinct from the current Sarcocystis hirsuta in cattle and morphologically indistinguishable from Sarcocystis sinensis in water buffaloes. Parasitology Research, 115, 1-21.
- GÖBEL, E., KATZ, M. & ERBER, M., 1978. Licht- und elektronenmikroskopische Untersuchungen zur Entwicklung von Muskelzysten von Sarcocystis suicanis in Hausschweinen nach experimenteller infektion. Zentralblatt für Bakteriologie Originale A, Medizinische Mikrobiologie, Parasitologie, 241, 368-383.
- GRANSTROM, D. E., RIDLEY, R. K., BAOAN, Y. & GERSHWIN, L. J., 1990. Immunodominant proteins of Sarcocystis cruzi bradyzoites isolated from cattle affected or nonaffected with eosinophilic myositis. American Journal of Veterinary Research, 51, 1151-1155.
- GRANSTROM, D. E., RIDLEY, R. K., YAO, B., GERSHWIN, L. J. & BRIGGS, D. J., 1990. Immunofluorescent localization of Sarcocystis cruzi antigens, IgG and IgE, in lesions of eosinophilic myositis in cattle. Journal of Veterinary Diagnostic Investigation, 2, 147-149.
- GUNNING, R. F., JONES, J. R., JEFFREY, M., HIGGINS, R. J. & WILLIAMSON, A. G., 2000. Sarcocystis encephalomyelitis in cattle. Veterinary Record, 146, 328.
- HAJTÓS, I., GLÁVITS, R., PÁLFI, V. & KOVÁCS, T., 2000. Sarcocystosis with neurological signs in breeding-sheep stock. Magyar Állatorvosok Lapja, 122, 72-78.
- HARTLEY, W. J. & BLAKEMORE, W. F., 1974. An unidentified sporozoan encephalomyelitis in sheep. Veterinary Pathology, 11, 1-12.
- HEYDORN, A. O. & HARALAMBIDIS, S., 1982. Zur Entwicklung von Sarcocystis capracanis Fischer, 1979. Berliner und Münchener Tierärztliche Wochenschrift, 95, 265-271.
- HEYDORN, A. O. & KARAER, Z., 1986. Zur Schizogonie von Sarcocystis ovicanis. Berliner und Münchener Tierärztliche Wochenschrift, 99, 185-189.
- HEYDORN, A. O. & KIRMSSE, P., 1996. Isolation und experimentelle Übertragung von Sarcocystis moulei Neveu-Lemaire, 1912. Berliner und Münchener Tierärztliche Wochenschrift, 109, 440-445.
- HEYDORN, A. O. & MEHLHORN, H., 1987. Fine structure of Sarcocystis arieticanis Heydorn, 1985 in its intermediate and final hosts (sheep and dog). Zentralblatt für Bakteriologie, Mikrobiologie und Hygiene A, 264, 353-362.
- HEYDORN, A. O. & ROMMEL, M., 1972. Beiträge zum Lebenszyklus der Sarkosporidien. II. Hund und Katze als Überträger der Sarkosporidien des Rindes. Berliner und Münchener Tierärztliche Wochenschrift, 85, 121-123.
- HEYDORN, A. O. & UNTERHOLZNER, J., 1983. Zur Entwicklung von Sarcocystis hircicanis n. sp. Berliner und Münchener Tierärztliche Wochenschrift, 96, 275-282.
- HEYDORN, A. O. & WENIGER, J. H., 1988. Einflub einer akuten Sarcocystis suihominis - Infektion auf die Mastleistung von Absatzferkeln. Berliner und Münchener Tierärztliche Wochenschrift, 101, 307-310.
- HU, J. J., HUANG, S., WEN, T., ESCH, G. W. & LI, H. L., 2017. Morphology, molecular characteristics, and demonstration of a definitive host for Sarcocystis rommeli from cattle (Bos taurus) in China. Journal of Parasitology, 103, 471-476.
- HU, J. J., WEN, T., CHEN, X. W., LIU, T. T., ESCH, G. W. & HUANG, S., 2016. Prevalence, morphology, and molecular characterization of Sarcocystis heydorni sarcocysts from cattle (Bos taurus) in China. Journal of Parasitology, 102, 545-548.
- HUONG, L. T. T., DUBEY, J. P., NIKKILÄ, T. & UGGLA, A., 1997. Sarcocystis buffalonis n. sp. (Protozoa: Sarcocystidae) from the water buffalo (Bubalus bubalis) in Vietnam. Journal of Parasitology, 83, 471-474.
- HUONG, L. T. T., DUBEY, J. P. & UGGLA, A., 1997. Redescription of Sarcocystis levinei Dissanaike and Kan, 1978 (Protozoa: Sarcocystidae) of the water buffalo (Bubalus bubalis). Journal of Parasitology, 83, 1148-1151.
- HUONG, L. T. T. & UGGLA, A., 1999. Sarcocystis dubeyi n. sp. (Protozoa: Sarcocystidae) in the water buffalo (Bubalus bubalis). Journal of Parasitology, 85, 102-104.
- IMES, G. D. & MIGAKI, G., 1967. Eosinophilic myositis in cattle-pathology and incidence. Proceedings, annual meeting of the United States Animal Health Association, 111-112.
- JEFFREY, M., LOW, J. C. & UGGLA, A., 1989. A myopathy of sheep associated with Sarcocystis infection and monensin administration. Veterinary Record, 124, 422-426.
- JENSEN, R., ALEXANDER, A. F., DAHLGREN, R. R., JOLLEY, W. R., MARQUARDT, W. C., FLACK, D. E., BENNETT, B. W., COX, M. F., HARRIS, C. W., HOFFMANN, G. A., TROUTMAN, R. S., HOFF, R. L., JONES, R. L., COLLINS, J. K., HAMAR, D. W. & CRAVANS, R. L., 1986. Eosinophilic myositis and muscular sarcocystosis in the carcasses of slaughtered cattle and lambs. American Journal of Veterinary Research, 47, 587-593.
- JERRETT, I. V., MCORIST, S., WADDINGTON, J., BROWNING, J. W., MALECKI, J. C. & MCCAUSLAND, I. P., 1984. Diagnostic studies of the fetus, placenta and maternal blood from 265 bovine abortions. Cornell Veterinarian, 74, 8-20.
- KUNDE, J. M., JONES, L. P. & CRAIG, T. M., 1980. Protozoal encephalitis in a bovine fetus. Southwestern Veterinarian, 33, 231-232.
- LANDSVERK, T., 1979. An outbreak of Sarcocystis in a cattle herd. Acta Veterinaria Scandinavica, 20, 238-244.
- LERCHE, M. & BROCHWITZ, H., 1957. Sarkosporidienbefall des Rindes und Perimyositis eosinophilica. Deutsche Tierärztliche Wochenschrift, 64, 251-252.
- LEVINE, N. D., 1986. The taxonomy of Sarcocystis (Protozoa, Apicomplexa) species. Journal of Parasitology, 72, 372-382.
- MACKIE, J. T. & DUBEY, J. P., 1996. Congenital sarcocystosis in a Saanen goat. Journal of Parasitology, 82, 350-351.
- MACKIE, J. T., RAHALEY, R. S. & NUGENT, R., 1992. Suspected Sarcocystis encephalitis in a stillborn kid. Australian Veterinary Journal, 69, 114-115.
- MARKUS, M. B., VAN DER LUGT, J. J. & DUBEY, J. P., 2004. Sarcocystosis. In: Infectious Diseases of Livestock, COETZER, J. A. W. & TUSTIN, R. C., (eds.). 2nd edition. Oxford University Press, 1, 360–375.
- MATUSCHKA, F. R., SCHNIEDER, T., DAUGSCHIES, A. & ROMMEL, M., 1986. Cyclic transmission of Sarcocystis bertrami Doflein, 1901 by the dog to the horse. Protistologica, 22, 231-233.
- MCKENNA, P. B. & CHARLESTON, W. A. G., 1992. The survival of Sarcocystis gigantea sporocysts following exposure to various chemical and physical agents. Veterinary Parasitology, 45(1-2), 1-16.
- MCKENNA, P. B. & CHARLESTON, W. A. G., 1994. The outdoor survival of Sarcocystis gigantea sporocysts. Veterinary Parasitology, 55, 21-27.
- MEADS, E. B., 1976. Dalmeny disease - another outbreak - probably sarcocystosis. Canadian Veterinary Journal, 17, 271.
- MEHLHORN, H., HARTLEY, W. J. & HEYDORN, A. O., 1976. A comparative ultrastructural study of the cyst wall of 13 Sarcocystis species. Protistologica, 12, 451-467.
- MEHLHORN, H. & HEYDORN, A. O., 1977. Light and electron microscopic studies of Sarcocystis suihominis. 1. The development of cysts in experimentally infected pigs. Zentralbl. Bakteriol. Orig. A, 239, 124-139.
- MORGAN, G., TERLECKI, S. & BRADLEY, R., 1984. A suspected case of Sarcocystis encephalitis in sheep. British Veterinary Journal, 140, 64-69.
- MUHM, R. L., BARNETT, D., BRYANT, D. T., COLE, J. H. & KADEL, W. L., 1979. Sarcocystosis: a case study. Proceedings of the 22nd Annual Meeting of the American Association of Veterinary Laboratory Diagnosticians, 139-146.
- MUNDAY, B. L. & BLACK, H., 1976. Suspected Sarcocystis infections of the bovine placenta and foetus. Z Parasitenkd, 51, 129-132.
- MUNDAY, B. L., MASON, R. W. & CUMMING, R., 1973. Observations on diseases of the central nervous system of cattle in Tasmania. Australian Veterinary Journal, 49, 451-455.
- MUNDAY, B. L. & OBENDORF, D. L., 1984. Development and growth of Sarcocystis gigantea in experimentally-infected sheep. Veterinary Parasitology ,15, 203-211.
- MUNDAY, B. L. & OBENDORF, D. L., 1984. Morphology of Sarcocystis gigantea in experimentally-infected sheep. Veterinary Parasitology, 16, 193-199.
- O'DONOGHUE, P. J., ADAMS, M., DIXON, B. R., FORD, G. E. & BAVERSTOCK, P. R., 1986. Morphological and biochemical correlates in the characterization of Sarcocystis spp. Journal of Protozoology, 33, 114-121.
- O'TOOLE, D., JEFFREY, M., CHALLONER, D., MAYBEY, R. & WELCH, V., 1993. Ovine myeloencephalitis-leukomyelomalacia associated with a Sarcocystis-like protozoan. Journal of Veterinary Diagnostic Investigation, 5, 212-225.
- OBENDORF, D. L. & MUNDAY, B. L., 1987. Experimental infection with Sarcocystis medusiformis in sheep. Veterinary Parasitology, 24, 59-65.
- ODENING, K., RUDOLPH, M., QUANDT, S., BENGIS, R. G., BOCKHARDT, I. & VIERTEL, D., 1998. Sarcocystis spp. in antelopes from southern Africa. Acta Protozoologica 37, 149-158.
- ÖZMEN, O., SAHINDURAN, S., HALIGÜR, M., YUKARI, B. A. & DORRESTEIN, G. M., 2009. Encephalitic sarcocystosis and its prophylactic treatment in sheep. Turkish Journal of Veterinary and Animal Sciences, 33, 151-155.
- PESCADOR, C. A., CORBELLINI, L. G., DE OLIVEIRA, E. C., BANDARRA, P. M., LEAL, J. S., PEDROSO, P. M. O. & DRIEMEIER, D., 2007. Aborto ovino associado com infecção por Sarcocystis sp. Pesquisa Veterinária Brasileira, 27, 393-397.
- QUANDT, S., BENGIS, R. G., STOLTE, M., ODENING, K. & BOCKHARDT, I., 1997. Sarcocystis infection of the African buffalo (Syncerus caffer) in the Krüger National Park, South Africa. Acta Parasitologica, 42, 68-73.
- REITEN, A. C., JENSEN, R. & GRINER, L. A., 1966. Eosinophilic myositis (sarcosporidiosis; sarco) in beef cattle. American Journal of Veterinary Research, 27, 903-906.
- RIMAILA-PÄRNÄNEN, E. & NIKANDER, S., 1980. Generalized eosinophilic myositis with sarcosporidiosis in a Finnish cow. Nordisk Veterinaermedicin, 32, 96-99.
- ROMMEL, M. & HEYDORN, A. O., 1972. Beiträge zum Lebenszyklus der Sarkosporidien. III. Isospora hominis (Railliet und Lucet, 1891) Wenyon, 1923, eine Dauerform der Sarkosporidien des Rindes und des Schweins. Berliner und Münchener Tierärztliche Wochenschrift, 85, 143-145.
- SÁEZ, T., RAMOS, J. J., DE JALON, J. A., UNZUETA, A. & LOSTE, A., 2003. Laryngeal hemiplegia in a ram associated with Sarcocystis species infection. Veterinary Record, 153, 27-28.
- SAITO, M., MIZUSAWA, K. & ITAGAKI, H., 1993. Chronic Sarcocystis infections in slaughtered cattle. Journal of Veterinary Medical Science, 55, 757-761.
- SAITO, M., SHIBATA, Y., KUBO, M. & ITAGAKI, H., 1997. Sarcocystis mihoensis n. sp. from sheep in Japan. Journal of Veterinary Medical Science 59, 103-106.
- SARGISON, N. D., SCHOCK, A., MACLEAN, I. A., RAE, A., HECKEROTH, A. R. & CLARK, A. M., 2000. Unusual outbreak of sporozoan encephalomyelitis in Bluefaced Leicester ram lambs. Veterinary Record, 146, 225-226.
- SAVILLE, W. J. A., DUBEY, J. P., OGLESBEE, M. J., SOFALY, C. D., MARSH, A. E., ELITSUR, E., VIANNA, M. C., LINDSAY, D. S. & REED, S. M., 2004. Experimental infection of ponies with Sarcocystis fayeri and differentiation from Sarcocystis neurona infections in horses. Journal of Parasitology, 90, 1487-1491.
- SAVINI, G., ROBERTSON, I. D. & DUNSMORE, J. D., 1997. Excystation rates and infectivity of sporocysts of Sarcocystis cruzi exposed to different treatments and storages. Veterinary Parasitology, 73, 17-25.
- SCHEBITZ, H. & HARTWIGK, H., 1950. Myositis sarcosporidica beim Pferd. Tierärztliche Umschau, 5, 351-353.
- SCHMITZ, J. A. & WOLF, W. W., 1977. Spontaneous fatal sarcocystosis in a calf. Veterinary Pathology, 14, 527-531.
- SCHOCK, A., FRENCH, H., CHIANINI, F., BARTLEY, P., KATZER, F. & OTTER, A., 2012. Respiratory disease due to acute Sarcocystis tenella infection in sheep. Veterinary Record, 170, 571.
- SCOTT, P. R. & SARGISON, N. D., 2001. Extensive ascites associated with vegetative endocarditis and Sarcocystis myositis in a shearling ram. Veterinary Record, 149, 240-241.
- STOLTE, M., ODENING, K., QUANDT, S., BENGIS, R. G. & BOCKHARDT, I., 1998. Sarcocystis dubeyella n. sp. and Sarcocystis phacochoeri n. sp. (Protozoa: Sarcocystidae) from the warthog (Phacochoerus aethiopicus) in South Africa. Journal of Eukaryotic Microbiology, 45, 101-104.
- STUBBINGS, D. P. & JEFFREY, M., 1985. Presumptive protozoan (Sarcocystis) encephalomyelitis with paresis in lambs. Veterinary Record, 116, 373-374.
- SYKES, J. E., DUBEY, J. P., LINDSAY, L. L., PRATO, P., LAPPIN, M. R., GUO, L. T., MIZISIN, A. P. & SHELTON, G. D., 2011. Severe myositis associated with Sarcocystis spp. infection in 2 dogs. Journal of Veterinary Internal Medicine, 25, 1277-1283.
- TADMOR, A., NOBEL, T. A. & MINDEL, J. B., 1966. Myositis in a mule possibly due to infestation with Sarcosporidia. Refuah Veterinarith, 23, 106-109.
- TAKLA, M., 1984. Akute Sarkosporidiose beim Rind. Ein Fall einer klinischen natürlichen Meningoenzephalitis durch Sarcocsytis cruzi beim Bullen. Tierärztliche Praxis, 12, 167-172.
- THORTON, R., 1987. Protozoal abortion in cattle. Surveillance, 14, 15-16.
- TINLING, S. P., CARDINET, G. H., BLYTHE, L. L., COHEN, M. & VONDERFECHT, S. L., 1980. A light and electron microscopic study of sarcocysts in a horse. Journal of Parasitology, 66, 458-465.
- TRAUB-DARGATZ, J. L., SCHLIPF, J. W. J., GRANSTROM, D. E., INGRAM, J. T., SHELTON, G. D., GETZY, D. M., LAPPIN, M. R. & BAKER, D. C., 1994. Multifocal myositis associated with Sarcocystis sp in a horse. Journal of the American Veterinary Medical Association, 205, 1574-1576.
- VAN DER LUGT, J. J., MARKUS, M. B., KITCHING, J. P. & DALY, T. J. M., 1994. Necrotic encephalitis as a manifestation of acute sarcocystosis in cattle. Journal of the South African Veterinary Association, 65, 119-121.
- VANGEEL, L., HOUF, K., GELDHOF, P., DE PRETER, K., VERCRUYSSE, J., DUCATELLE, R. & CHIERS, K., 2013. Different Sarcocystis spp. are present in bovine eosinophilic myositis. Veterinary Parasitology, 197, 543-548.
- VANGEEL, L., HOUF, K., GELDHOF, P., NOLLET, H., VERCRUYSSE, J., DUCATELLE, R. & CHIERS, K., 2012. Intramuscular inoculation of cattle with Sarcocystis antigen results in focal eosinophilic myositis. Veterinary Parasitology, 183, 224-230.
- VICKERS, M. C. & BROOKS, H. V., 1983. Suspected Sarcocystis infection in an aborted bovine foetus. New Zealand Veterinary Journal, 31, 166.
- WESEMEIER, H. H., ODENING, K., WALTER, G. & BOCKHARDT, I., 1995. The black-backed jackal (Carnivora: Canidae) in Namibia as intermediate host of two Sarcocystis species (Protozoa: Sarcocystidae). Parasite, 2, 391-394.
- YAZICIOGLU, O. & BEYAZIT, A., 2005. Encephalomyelitis associated with a Sarcocystis-like protozoan in a ten-month-old ewe lamb. Turkish Journal of Veterinary and Animal Sciences, 29, 1209-1212.
- ZENG, W., SUN, L., XIANG, Z., LI, N., ZHANG, J., HE, Y., LI, Q., YANG, F., SONG, J., MORRIS, J., ROSENTHAL, B. M., SUN, L., LIU, H. & YANG, Z., 2018. Morphological and molecular characteristics of Sarcocystis bertrami from horses and donkeys in China. Veterinary Parasitology, 252, 89-94.
- ZOBBA, R., ALBERTI, A., MANUNTA, M. L., EVANGELISTI, M. A. & PARPAGLIA, M. L. P., 2014. What is your diagnosis? Cerebrospinal fluid from a sheep. Veterinary Clinical Pathology, 1-2.