- Infectious Diseases of Livestock
- Part 1
- Trichomonosis
- Vectors: Ticks
- Vectors: Tsetse flies
- Vectors: Muscidae
- Vectors: Tabanidae
- Vectors: Culicoides spp.
- Vectors: Mosquitoes
- Classification, epidemiology and control of arthropod-borne viruses
- Special factors affecting the control of livestock diseases in sub-Saharan Africa
- The control of infectious diseases of livestock: Making appropriate decisions in different epidemiological and socioeconomic conditions
- Infectious diseases of animals in sub-Saharan Africa: The wildlife⁄livestock interface
- Vaccination: An approach to the control of infectious diseases
- African animal trypanosomoses
- Dourine
- Trichomonosis
- Amoebic infections
- GENERAL INTRODUCTION: COCCIDIA
- Coccidiosis
- Cryptosporidiosis
- Toxoplasmosis
- Besnoitiosis
- Sarcocystosis
- Balantidiosis
- Leishmaniosis
- Neosporosis
- Equine protozoal myeloencephalitis
- GENERAL INTRODUCTION: BABESIOSES
- Bovine babesiosis
- Equine piroplasmosis
- Porcine babesiosis
- Ovine babesiosis
- GENERAL INTRODUCTION: THEILERIOSES OF CATTLE
- East Coast fever
- Corridor disease
- Zimbabwe theileriosis
- Turning sickness
- Theileria taurotragi infection
- Theileria mutans infection
- Theileria annulata theileriosis
- Theileriosis of sheep and goats
- Theileria buffeli⁄orientalis infection
- Non-pathogenic Theileria species in cattle
- GENERAL INTRODUCTION: RICKETTSIAL, CHLAMYDIAL AND HAEMOTROPIC MYCOPLASMAL DISEASES
- Heartwater
- Lesser known rickettsial infections in animals and humans
- Chlamydiosis
- Q fever
- Eperythrozoonosis
- Bovine Haemobartonellosis
- Potomac horse fever
- GENERAL INTRODUCTION: ANAPLASMOSES
- Bovine anaplasmosis
- Ovine and caprine anaplasmosis
Trichomonosis
This content is distributed under the following licence: Attribution-NonCommercial CC BY-NC  View Creative Commons Licence details here
View Creative Commons Licence details here
NJ Maclachlan and M-L Penrith (Editors). C H Annandale, D E Holm and P Irons, Trichomonosis, 2018.
Trichomonosis
Previous authors: P C IRONS, A P SCHUTTE, S HERR* AND J P KITCHING
Current authors:
C H ANNANDALE - Associated Professor, Veterinary Specialist in Reproduction, BCom, BVSc (Tons), MMedVet (Gyn) MBA Dip ACT, Department of Production Animal Studies, Faculty of Veterinary Science, University of Pretoria, Private Bag X04, Onderstepoort, Pretoria, Gauteng, 0110, South Africa
D E HOLM - Deputy Dean: Teaching and Learning, BVSc, MSc, PhD, Faculty of Veterinary Science, Private Bag X04, University of Pretoria, Onderstepoort, Pretoria, Gauteng, 0110, South Africa
P C IRONS - Veterinary Program Director, Faculty of Veterinary Science, Murdoch University, Perth, Australia
Introduction
Trichomonosis is a venereal disease of cattle associated with aberrant oestrus cycles, infertility, embryonal and foetal death, a low percentage of early abortions, and pyometra. It is caused by the protozoan, Tritrichomonas foetus, a parasite of the genital tract.
Although self-limiting and often unnoticed, the economic losses associated with trichomonosis can be severe. These are associated with increased intercalving periods due to delayed pregnancies, decreased calving percentage, reduced calf weaning weight, costs of replacement and culling of animals, as well as the cost of treatment.44, 74, 95, 96 Modelling of the economic effects of trichomonosis in a herd has shown that the losses increase exponentially with an increase in prevalence; up to 35 per cent of cow revenue can be lost if confined to a herd with infected bulls and a herd prevalence of trichomonosis of 40 per cent.96 Non-inflation adjusted losses in a dairy herd was estimated at US $665 per cow, more than 30 years ago.44
The geographical distribution of trichomonosis is vast – in fact, it can be considered to occur in all the major cattle-farming regions globally. It has been reported in North,5, 82, 106 Central and South America,29, 123 Europe,73, 76 Africa,32, 60 Asia71, 87 and Australia.19, 20 There is consensus that it can be considered endemic in regions where cattle are raised extensively.129 It can be found in dairy and beef herds alike, although it seems to be more prevalent in beef herds. The reduced prevalence of trichomonosis in dairy herds can be ascribed to the extensive use of artificial insemination with frozen semen subjected to rigorous biosanitary measures.12, 44, 107
Aetiology
There is some confusion in the literature regarding the first description of T. foetus. Mazanti is generally credited with the discovery of T. foetus in Italy, in 1900, but according to Morgan,78 it was first identified by Kunstles in France. Honigberg contends that the organism was first discovered in Europe by Riedmüller, who described the organism in 1928 and is recognized as the author of the name T. foetus.54
Tritrichomonas foetus is a commensal of the porcine intestine, stomach and nose,79 but is pathogenic in cats in association with chronic large intestinal involvement and diarrhoea68 and in cattle as a parasite of the reproductive tract that causes infertility.70, 129
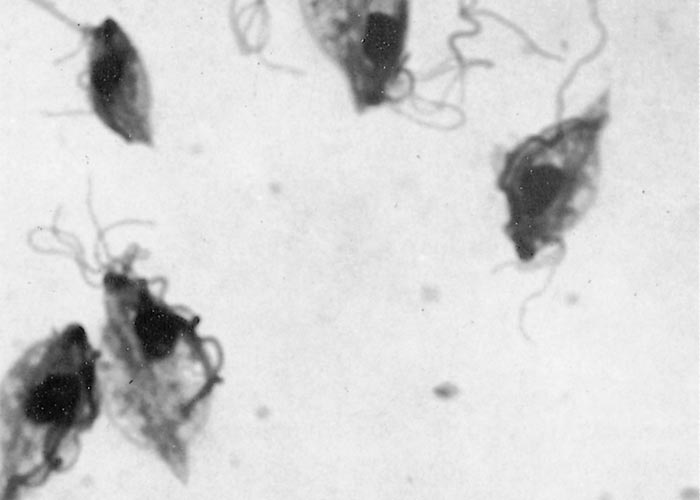
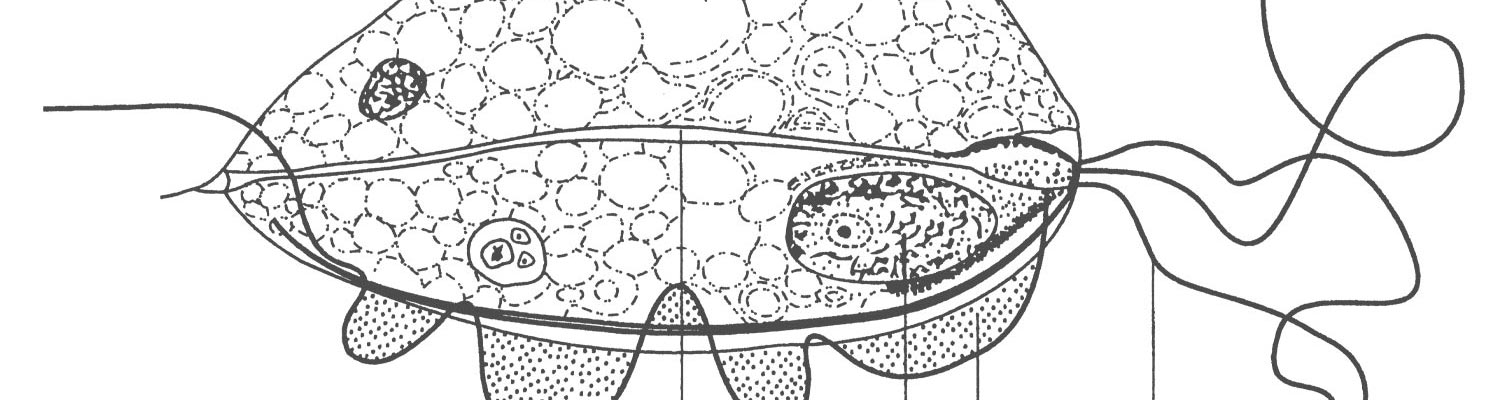
The protozoa can occur in a motile, flagellated form or an immobile, rounded form.72 Characteristic features (Figure 1) of the motile parasite (trophozoite form) under phase contrast or darkfield microscopy (Figure 2) are the three anterior flagellae and an undulating membrane.10 The undulating membrane is easily recognised in viable organisms due to its characteristic wave-like motion on one side of the organism, is typically shorter than the body and its external margin is supported by a recurrent flagellum that continues beyond the posterior end as a free posterior flagellum.106 The protozoan is generally pyriform in shape in fresh specimens, but tends to display pleomorphism and a spherical shape in culture. Its size is reported to be slightly smaller than polymorphonuclear cells at 9-25 X 3-15µm and it shows a characteristic jerky or rolling motion in wet preparations.61, 82, 121
Even though the organism is easy to observe in wet preparations under phase contract microscopy at 100X to 250X magnification, definitive diagnosis should always be pursued by PCR or electron microscopy due to the presence of non-pathogenic trichomonads in preputial cavities of bulls.8, 51
The immotile, rounded form is often termed a pseudocyst, and is associated with unfavourable conditions in the prepuce, in culture or in transport medium.72, 92 The pseudocyst form is not the most common form, but when present in higher numbers, can complicate microscopic diagnosis by obscuring the appearance of characteristic features of the flagellated form.93
Different serotypes64 and genotypes116 are recognized. Three serotypes were first recognized by Kerr and Robertson,64 namely Brisbane, Belfast and Manley and subsequently confirmed and elaborated on by others.14, 30 The serotypes appear not to be of much significance since heifers immunized with one serotype develop immunity to homologous and heterologous serotypes.9 Tritrichimonas foetus antigens are a highly complex set of proteins, ranging in molecular weight from as small as 25 to 220 kD and consist of 55 to 60 protein bands.48 There is strong antigenic cross-reactivity among diverse T. foetus isolates, indicated by similar antigenic and total protein profiles.56
Currently, a bovine and a feline genotype are recognized,79, 116 with a recent report of a novel genotype associated with South African isolates.17 The genotypes appear genetically similar across 10 DNA loci, with most diversity around the cysteine protease-2 (CP-2) gene family, involved in host-parasite interactions at a cellular level.116 The investigation indicated that T. suis is identical to the bovine genotype.116
Epidemiology
Bulls are the main source of infection due to their life-long carrier state. Within herd infection rates in bulls may vary from 1 – 40 per cent.1, 5, 22, 30, 47, 94 There are numerous studies reporting prevalence rates with estimates ranging from 0 – 25 per cent in samples and 0 – 46 per cent in herds.80, 83, 94, 96, 97, 118, 128
Infection occurs predominantly via coitus during natural service, but iatrogenic infection through improper biosecurity44 and the use of contaminated semen for artificial insemination 55 have been reported. After infection, mature / older bulls remain life-long carriers and perpetuate infection from breeding season to breeding season.11 Younger bulls generally do not become infected but if they do may be transient carriers.11 Bulls of all breeds are susceptible to infection, with conflicting reports indicating different infection rates, dependent on breed susceptibility, breeding activity94 and management.4, 7, 96, 111
The epidemiology of trichomonosis is very similar to that of genital campylobacteriosis in cattle caused by Campylobacter fetus subsp. venerealis (see Genital campylobacteriosis in cattle). Due to the common risk factors, mixed infection with C. fetus subsp. venerealis is common,77, 91, 94 although no causal links for such co-infection have been demonstrated.15, 74 While spontaneous recovery has been reported in bulls,11 it remains unclear how commonly it occurs or whether its occurrence is linked to false negative diagnostic tests. It is not prudent to rely solely on self-cure in a management strategy aimed to control or eradicate trichomonosis.
Homosexual transmission of T. foetus between yearling and other bulls is theoretically possible, but unlikely and not supported by strong evidence currently.66, 107, 111 As many non-pathogenic trichomonads can be found in the faeces of bulls, and some faecal material is frequently found in the preputial cavities of yearling bulls, there is the potential to incorrectly identify trichomonads from preputial washes as T. foetus. Mechanical transfer by negative yearling bulls from infected to susceptible cows has been described, but requires two services within 30 minutes of each other.22 Transmission of infection between cows by flies or by other mechanical means is unlikely.22
All cows of breeding age are equally susceptible to infection, but mostly clear themselves of infection over two to three oestrus cycles within seven months under conditions of normal cyclicity.44, 107 A minority of females can become persistently infected with the organism. Cows that develop pyometra after infection do not show regular oestrus cycles and therefore remain persistently infected; they are important reservoirs of the organism within the population. Cows that retain a pregnancy despite being infected with the organism retain the organism and would similarly need a period of normal oestrus cycles after parturition to rid themselves of the infection. During abortion, large numbers of the organisms are shed in the lochia.115
A partial immunity develops in infected cows; this does not prevent reinfection, but does enable the cow to rid herself of the infection in a shorter time period.23 Such immunity from natural infection does not occur in bulls.
The transmission rate of the organism within a herd following service by an infected bull, varies between 42 and 88 per cent, dependent on the number of services and the age of the susceptible females.6, 23
Risk factors associated with higher prevalence rates:
- Type of herd e.g. whether it is a beef or dairy herd
- Herds with bigger numbers of breeding age cows97
- Lower bull-to-cow ratios97
- Older bulls (>4 years of age).19, 23, 97 This can either be ascribed to older bulls being more assertive and confident breeders, or to the deeper and more abundant preputial crypts associated with older bulls.10, 90
- Evidence seems equivocal, but some studies associate a higher prevalence with Bos taurus breeds7, 97
- Not providing for a period of sexual rest94
- Commingled grazing42, 62
Pathogenesis and clinical signs
Most bulls display no visible clinical signs, though if observed closely, some degree of preputial swelling due to an accumulation of mucopurulent exudate may be seen early in the weeks following infection.106 Trichomonosis is characterized by an infection of the preputial and penile mucosa, and rarely, the urethral orifice,10, 89, 104 although it has been isolated from other areas of the male urogenital tract, such as the seminal vesicles.89 Older bulls become life-long carriers of the organism and it is thought that this can be attributed to deeper penile epithelial crypts or more regular breeding activity.10
Cows and heifers are infected through coitus when initially the vagina is colonized by T. foetus. From the colonized vagina, the organisms migrate through the cervix into the uterus and ultimately, the Fallopian tubes, causing cervicitis and in some cases, endometritis and salpingitis by 60 days post-infection.90 Sampling from any of these sites, typically by aspiration of the mucoid exudate, can yield positive cultures of T. foetus. 6, 107 Only in cases where salpingitis develops, would fertilization failure occur, but more commonly, embryonal death is seen.90 The cause of embryonal death has not been established, but it is known to be associated with histological evidence of endometritis2, 21, 90 and most likely interferes with nidation or embryonal nutrition. In a mouse model, the hypothesis was tested that embryonal death is the result of an excessive maternal immune response and consequently attributed to a regulatory T cell response that leads to persistent infection.127 Embryonal and foetal death have been reported from as early as 14-18 days until five months of gestation, with occasional later abortions.6, 10 When embryonal death occurs after day 17 (recognition of pregnancy), irregular oestrus cycles will be observed. Cows develop pyometra when embryonal death occurs after recognition of pregnancy (d 17) and the corpus luteum persists, causing the foetuses to be retained. This occurs in about 5 per cent of cases and may take many months to resolve.10, 77 However, the bulk of infections will spontaneously resolve over two to three oestrus cycles, although reported clearance times vary between two to seven months.11, 21, 24, 44 All the affected parts of the genital tract, i.e. vagina, cervix and uterus are simultaneously cleared of the infection.112 The involvement of T. foetus-specific bovine IgG1 and IgA in uterine and vaginal secretions is thought to play an important role in clearance of the infection, while the role of IgE and consumption (through degranulation) of mast cells has also been demonstrated.28
Control measures can be complicated by the prolonged carrier status44, 112 and the occurrence of persistence of infection through pregnancy and into the postpartum period in some cows.115 After ridding themselves of the infection, partial immunity develops in some animals and can reduce the extent of pregnancy loss in the subsequent breeding season.23 This partial immunity is of short duration, resulting in return to full susceptibility to infection after 15 months of age. Infertility is rarely seen, even following chronic endometritis and pyometra.106
Host-parasite interaction involve several mechanisms unique to the parasite itself, local immunity factors and hormonal influences. Evasive properties of T. foetus include production of extracellular proteinases and the ability to bind non-specifically with immunoglobulins.120 The organisms bind to vaginal epithelial cells in a species-specific fashion thought to be receptor-ligand in type, where they cause apoptotic cell death through the involvement of caspases.110 It has also been shown that the pseudocyst form of T. foetus can adhere to vaginal epithelial cells in a contact-dependent manner.72 The binding to vaginal epithelial cells is enhanced by exposure of the vaginal cells to progesterone.
Bulls show a weak circulating antibody response, but antibodies are present in preputial secretions, suggesting local processing of antigens.16, 104 More recent work, comparing vaccinated and unvaccinated bulls has shown that unvaccinated bulls mount no detectable immune response in serum or preputial secretions. The vaccinated bulls mounted a humoral (systemic and local IgG1 and IgA) and cell-mediated response (MHC II, CD3+ and CD8+ T cells, dendritic cell and B cells, including IgA and IgG1 plasma cells).26
Heifers and cows mount a significant antibody response by 11 weeks after infection.112 The immune response is characterized by transient IgG2 and IgM presence and longer-lasting IgA and IgG1 response in vaginal mucus,41, 58, 112, 117 while the uterine response was found to be slower and of shorter duration. A substantial rise in systemic IgG levels can also be demonstrated following infection.18 From vaccine trials, it has been established that the IgG1 response is directed against the surface antigen of T. foetus.18, 35, 86
It is almost a hallmark of trichomonosis that pregnancy rates are in the 35-40 per cent range in susceptible herds, accompanied by a spread in gestational ages of pregnancies4, 11, 23 in herds making use of restricted breeding seasons. In chronically infected herds pregnancy rates typically are higher, reportedly 75-80 per cent, a level still suggestive of pregnancy loss. Analysis of the calving distribution over the calving season typically reveals a shift in calving towards the latter part of the season. Trichomonosis appears to be of a more insidious nature in dairy herds.111
Pathology
Gross lesions are not commonly observed in bulls, but occasionally a mucopurulent preputial exudate can be seen, accompanied by a mild preputial swelling.21 Debris can be seen in the crypts of the preputial mucosa, but the organism cannot be demonstrated in sections.89 The histology is characterized by inflammation with a mixed cellular infiltrate.
In cows, few lesions are observed in the first 50 days after infection, but after that varying degrees of cervicitis, endometritis and salpingitis can be seen.2, 90 A silver stain can be used to identify the organisms on the surface and in the lumen of the endometrial glands. In cows with pyometra due to T. foetus infection, large volumes of pus with abundant organisms can be found.10
Aborted foetuses can be fresh or autolysed and their placentas are often oedematous, although this is not a very specific finding.101 While most foetuses show few gross changes, some may exhibit pyogranulamotous bronchopneumonia and necrotizing enteritis characterized by thrombotic lesions with tissue invasion by tritrichomonads. Placental lesions consist of stromal oedema, mixed inflammatory cell infiltration and focal necrosis of the chorionic epithelium.101, 102 Organisms may be found randomly scattered in chorionic stroma and in necrotic exudate adherent to the villi.103
Diagnosis
One is usually led to suspect a herd is infected with T. foetus when calving percentage or pregnancy rate is low to moderate, aberrant oestrus cycles are observed, prolonged intercalving periods are recorded and there is a higher than normal occurrence of post-coital pyometra.10, 98, 111 Sporadic abortions and failure to conceive are often seen in combination with the clinical picture described above.
A positive diagnosis of trichomonosis requires demonstration of live T. foetus organisms from diagnostic samples, which can include preputial material of bulls, genital secretions of cows and heifers and aborted foetuses and placentae. Since preputial material is readily available for collection from bulls, they are often deemed the most suitable animals for testing. It is generally recommended that samples from bulls are collected once a week for three weeks. This takes into account the fluctuation of organism numbers, peaking every 5-10 days,50 as well as the imperfect diagnostic sensitivity of the currently available tests.88, 95 It is also generally recommended that sampling takes place outside of the breeding season, preferably before the onset of the breeding season. Sampling from bulls during the breeding season can lead to false -negative results since the number of organisms tend to be markedly reduced when bulls are actively breeding.23
Preputial material from bulls can be collected either via sheath wash or sheath scraping; both methods offer similar sensitivity60, 109 and have been well described.107, 119 Recently, a technique utilizing gauze swabs was described and found to be diagnostically more sensitive, while at the same time offering safety advantages over the traditional techniques.31 Rinses taken from artificial vagina liners immediately after use constitute another sample that can be considered to provide similar sensitivity.46
Sampling technique
Sheath washes are collected by infusing 50 ml of sterile phosphate-buffered saline into the preputial cavity of a bull through a funnel and silicone tube, vigorously massaging the fluid in the preputial cavity and retrieving it via the tube and funnel into a clear glass bottle. A good sample is opaque in colour and contains visible cellular material. Urine contamination of the sample does not kill the organisms.89
Sheath scraping is performed with the aid of a plastic artificial insemination pipette, attached to a syringe. The tip of the pipette is inserted into the preputial cavity, advanced to the caudal end of the lamina interna reflection onto the penis, and moderate suction is applied on the syringe. While maintaining the negative pressure on the syringe and pipette, and applying external pressure onto the sheath, the tip of the pipette is directed onto the preputial mucosa and the pipette is moved forwards and backwards, scraping against the mucosa and collecting cellular material into the lumen of the pipette. When the pipette is withdrawn, thick mucoid material should be visible in its lumen; this material is then flushed into a sterile vial by aspirating end ejecting PBS through the pipette to loosen the cellular material. Blood is sometimes seen and is taken to indicate adequate depth of sampling.4
The technique for obtaining a sample of the preputial mucosa by swabbing involves the use of 16-ply gauze sponges. These are used to swab the penile and preputial mucosa during full extension of the penis.31
In females, five consecutive negative results are needed to certify an animal free from the infection44 and it is suggested that samples are collected from those with pyometra or that have recently aborted to maximise chances of a positive culture. Vaginal, cervical or uterine samples may be collected via a pipette attached to a syringe, or by a swab, douche or scraping method.6 Use of a sanitary sleeve is advised to prevent contamination of the sample. A small volume of Ringer’s lactate solution can be infused to facilitate collection of thicker secretions.36
Suitable samples from aborted material include foetal abomasal contents and foetal fluids. Foetal and placental tissues should also be collected for histopathological examination.
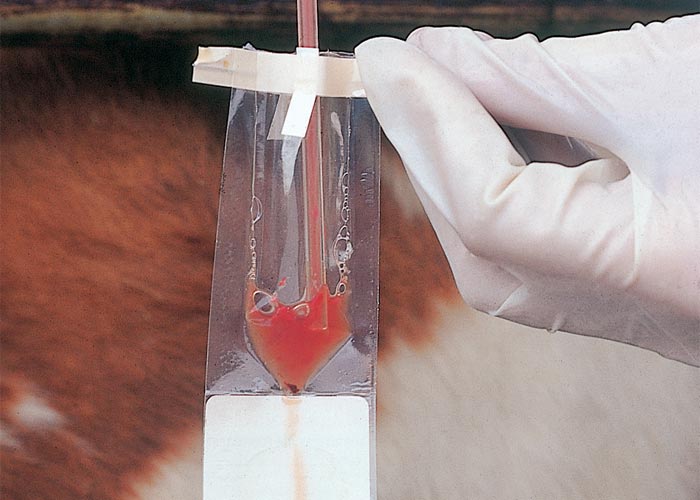
Sample transport and media
Enriched media between sample collection and laboratory processing are essential to achieve satisfactory diagnostic accuracy. After collection of samples, they are placed either in a transport (Figure 3) or culture medium. This is usually done at the crushside.43 In the absence of commercial transport or culture medium, refrigerated buffered saline with foetal bovine serum added can be used. Other suitable media include lactated Ringer’s solution, Kupferberg medium and skimmed milk, all of which could ensure survival of organisms for 48 – 96 hours.100, 111 It is important that samples are kept cool and arrive at the laboratory as soon as possible, since delayed transport can lead to decreased organism viability.65, 114
Commercially available transport media often also include nutrients for culture, allowing diagnosis in situations where laboratory facilities are not available. The best-known of these products is the In-Pouch TF (BioMed Diagnostics), which is a clear pouch with a protease peptone medium.13, 119 The culture of trichomonads other than T. foetus is a potential disadvantage of using In-Pouch TF.9
Various nutrient media have been used for the culture of T. foetus. Most consist of peptones, yeast extract, serum, maltose or dextrose, buffers and antibiotics. Diamond’s medium (trypticase-yeast extract-maltose) is the most commonly used in the USA and has a reported sensitivity of 81.6 – 93.2 per cent.65, 88, 109 Positive cultures are usually identified by 48 hours, but should continue up to seven days. Despite finding similar sensitivities for preputial samples for Diamond’s, Claussen’s and In-Pouch media, the In-Pouch medium is preferred by some researchers due to its more rapid identification of positive cultures.13, 119 Interestingly, sensitivity of culture from cervicovaginal samples of experimentally infected cows was reported to be higher than for naturally infected animals.90, 114
Diagnostic methods and tests
Samples may be subjected to direct examination or to culture, and then examined. The motile organisms are more readily observed under dark field, phase contrast and differential interference microscopy. While the organisms can be readily identified with microscopy, care should be taken that other trichomonads are not mistaken for T. foetus.8 Centrifugation of samples can aid in diagnosis of trichomonads due to concentration of the samples, but sensitivity is nevertheless reported to range between 20 and 73.9 per cent.105, 113 Direct examination of female genital secretions is surprisingly more sensitive than examination of preputial material, and the cervix appears to be the site best suited for sampling and culture.113
Despite initial results finding low sensitivity and specificity for a DNA probe,3 various PCR tests are now commercially available and validated for this purpose.33, 53 Currently, this includes a conventional PCR that uses diagnostic size variants from within the internal transcribed spacer 1 (ITS1) region that is located between the 18S rRNA and 5.8S rRNA subunits, allowing it to identify T. foetus and distinguishing it from other trichomonads that might be present in the preputial cavity of bulls.45 PCR assays utilising these primers have been found to be highly sensitive and specific for diagnosing T. foetus in smegma and mucus samples.75 The conventional PCR can be employed either on samples that have already been cultured or on the collected sample prior to culture.27, 75, 84, 98 While specificity of the conventional PCR is consistently reported to be high (>92 per cent), sensitivity starts off at 76 per cent for a single sample and improves to 92 per cent if 3 consecutive samples, collected over three weeks, are tested.27 The conventional PCR is reported to have a detection limit of 100 organisms, not to be affected by sampling method and to suffer from decreased sensitivity with prolonged storage time, reaching 31 per cent by Day 5 of storage.81 A quantitative real-time PCR has also been developed and is reported to be more tolerant of suboptimal transport conditions.25
Pooling of preputial samples, and then subjecting pools to culture and PCR, or direct PCR testing, has been reported, using either conventional or real-time PCR assays.38, 39, 63 Using conventional PCR following culture, Kennedy63 found that pooling up to five samples did not result in a reduction in sensitivity and specificity of the test. In fact, a higher sensitivity for the test on pools of five samples than a single culture was reported. The authors outlined the benefit of pooled samples in government eradication and surveillance schemes, herd status monitoring and the robustness of PCR technology to sample transport and storage conditions. Using RT-qPCR, Garcia Guerra reported direct testing of samples collected in PCR and pooling of ten samples to have a sensitivity of 96 per cent;39 they later reported pools of up to 25 samples to be feasible without affecting sensitivity,38 provided they were individually collected and cultured before pooling. A multiplex PCR assay for screening of Campylobacter fetus subsp. venerealis and T. foetus has recently been developed and offers promise for surveillance and large-scale epidemiological studies.125 A bead-agglutination test offers another alternative for rapid screening of bulls as part of a breeding soundness evaluation and could also reduce the costs involved with screening of a group of bulls with PCR and culture.108
Vaginal mucus agglutination tests have been well documented but lack sufficient specificity for accurate diagnosis.100, 106 An ELISA assay using the protective surface antigen TF1.17 has been shown to detect longstanding IgA responses in vaginal mucus, starting six weeks after infection.59 While serological assays have been developed,8 there are none that identify infected bulls and their use is somewhat limited given the relative ubiquity and affordability of PCR tests. An intradermal test has been developed and is proposed as a screening method, but needs further research.117
While trichomonads are often visible in haematoxylin and eosin-stained sections of female genital tissues, special staining might be needed to differentiate them form leukocytes.103 Immunohistochemistry using mono- and polyclonal antibodies has also been successful in identifying trichomonads; this technique is especially useful for aborted material that has not yet undergone marked autolysis.101, 103, 104
Differential diagnosis
The main differential diagnosis for the clinical syndromes associated with trichomonosis is genital campylobacteriosis, caused by Campylobacter fetus subsp. venerealis. Laboratory tests are essential to distinguish between the two causative organisms. There are many other causes of infertility, irregular oestrus cycles and occasional abortions, many of which are not infectious.
Control
Control requires a multifactorial managerial approach, taking the epidemiology of the disease, diagnostic tests and their sensitivity and specificity, and unique farm and economic factors into consideration.
Treatment
There are many reports of successful treatment of bulls and cows infected with T. foetus. The treatment is, however, fraught with logistic challenges and complications, cumbersome and time-consuming and is rarely pursued nowadays. Side effects associated with treatment include intractability of bulls due to the discomfort of the treatment and treatment process, rumen stasis, inappetence, abscesses at the injection sites and rapid development of resistance in the organisms.
Early reports included the use of potassium iodide orally, sodium iodide intravenously, topical application of nitrofurazone, iodine, hydrogen peroxide, diminazene or an ointment containing 0.5 per cent diaminomethylacridine and 0.5 per cent aminochinolylcarbamide hydrochloride (Bovoflavin).34, 52, 69, 107, 122 More recent reports of treatment focus on the use of antiprotozoal agents such as metronidazole, ipronidazole and dimetridazole.4, 40, 65, 114, 126 The successful use of these agents often requires intrapreputial infusion and parenteral administration due to the presence of intrapreputial flora that metabolize imidazole compounds. It is advisable to test to determine whether a bull is free of infection after treatment and again before the start of the breeding season, since treatment failure and recurrence of infection is common.114 In many countries the use of these drugs in food animals is prohibited due to residue concerns and the potential impact thereof in the food chain on human health.
Although treatment of cows with ipronidazole has been suggested, it is difficult to evaluate efficacy due to poor diagnostic methods for identifying chronically infected cows.10, 37 Concurrent treatment of cows with pyometra with ecbolics does evacuate the uterus, but infection can persist and lead to more permanent infertility.37
Vaccination
Currently, the only commercially available vaccine contains whole killed T. foetus, alone or in combination with other infectious agents implicated in embryonal loss and infertility (TrichGuard® or TrichGuard V5L HB®, Boehringer). Improved understanding of the immunity associated with T. foetus infection, combined with modern molecular and vaccine methods, has led to advancement of vaccine efficacy against T. foetus in recent years, although many of the newer vaccines are not yet commercially available.18, 26, 35, 86, 124
While the commercial vaccine does not offer complete protection against infection, it has been shown to reduce numbers of organisms that are shed, the number of animals that become infected and the duration of infection.16, 41, 57, 67, 124 It is suggested that vaccination prevents early embryonal death, known to occur between 60 to 120 days after infection.41, 67 Vaccination has the added benefit of reducing or eliminating the cow carrier state in a herd.67
The ability of vaccines to be used as treatment for trichomonosis has been demonstrated,35 and outlined the potential of different adjuvants to enhance the immune response; saponin plus aluminium hydroxide seems the best adjuvant currently.
Management
Many managerial strategies have been developed to control trichomonosis. All are based on a sound understanding of the epidemiology of the disease and have been reviewed extensively.11, 98 A good strategy should additionally also consider unique practicalities and economics on a given farm and would therefore be unique to every farm and situation. Broadly speaking, the following objectives would need to be achieved to control the infection within a herd:
- Removal of infected bulls through treatment or culling
- Ensuring freedom of infection of bulls before the start of the breeding season through testing
- Reduction of infection rate or elimination of infection in the female herd
- Increasing herd immunity by vaccination
- Prevention of reintroduction of the infection
Artificial insemination, provided semen is certified free from pathogens and bio sanitary measures are adhered to during the process of insemination, is a very good measure to prevent transmission of the disease. Its use in many herds is not feasible due to the extensive administration and logistics it entails.37
Sexual rest in cows and heifers allows spontaneous cure to occur in most cases, and with it allows a return to acceptable levels of fertility over a 2 to 6-month period.2, 44 A period of three months is generally considered long enough to establish clinical cure provided cycling occurs regularly through the period. It is very important, however, to note that a small percentage112 of cows and heifers do remain infected for longer periods and would necessitate additional control measures to sexual rest. Pregnant carrier cows also need a similar rest period after calving in order to rid themselves of infection.115
The mainstay of many management strategies is separation of the herd into infected and clean groups. While it can be assumed that young, virgin bulls are clean, in the face of a high infection rate, they should rather be tested.7 Virgin heifers that will be mated for the first time can be safely assumed to be free from T. foetus infection. These two categories of animals are then regarded as part of the clean herd while the previously exposed herd is gradually reduced through culling. Without excellent management and fencing to allow strict separation of the two groups, this strategy is likely to fail.
Bulls can be ensured to be free from infection through various means. Ideally, all bulls should be tested and certified free from infection or purchased from clean herds with certificates indicating freedom from infection. Such a certificate should only be issued after three negative tests, one week apart. One could consider replacing bulls older than four years of age with virgin bulls on a regular basis, unless this is not economically sustainable due to cost and the marked shortening of the reproductive lifespan of bulls.74 While testing and using negative bulls only can be very effective, ignoring carrier cows and the known difficulty detecting them can derail a control strategy.49
The maintenance of a closed herd is essential to preventing introduction of T. foetus. This involves proper record-keeping, maintenance of good perimeter fences, limited new introductions and testing and quarantine of newly purchased stock. Older cows can only be assumed to be negative if they are from a known negative source or if they have calved normally and have cycled normally for three months without breeding.
Shortening of the breeding season coupled with a stringent culling policy is sometimes advocated for control of the disease, with the rationale of not allowing infected females the chance to become pregnant before the end of the breeding season and thus stay in the herd and be source of infection in subsequent breeding seasons.
References
- AKINBOADE, O.A., 1980. Incidence of bovine trichomoniasis in Nigeria. Revue d"elevage et de medecine veterinaire des pays tropicaux, 33, 381-384.
- ANDERSON, M.L., BONDURANT, R.H., CORBEIL, R.R. & CORBEIL, L.B., 1996. Immune and inflammatory responses to reproductive tract infection with Tritrichomonas foetus in immunized and control heifers. Journal of Parasitology, 82, 594-600.
- APPELL, L.H., MICKELSEN, W.D., THOMAS, M.W., 1993. A comparison of techniques used for the diagnosis of Tritrichomonas foetus infections in beef bulls. Agricultural Practice, 14, 30-34.
- BALL, L., DARGATZ, D.A., CHENEY, J.M. & MORTIMER, R.G., 1987. Control of venereal disease in infected herds. Veterinary Clinics of North America Food Animal Practice, 3, 561-574.
- BARTLETT, D.E., HASSON, E.V. & TEETER, K.G., 1947. Occurrence of Tritrichomonas foetus in preputial samples from infected bulls. Journal of the American Veterinary Medical Association, 110, 114-120.
- BARTLETT, D.E., 1968. Bovine venereal trichomoniasis and bovine abortion. In: FAULKNER, L.C., (ed). Abortion Diseases of Livestock. Illinois: Charles C. & Thomas Publisher.
- BONDURANT, R.H., ANDERSON, M.L., BLANCHARD, P., HIRD, D., ELMI-DANAYE, C., PALMER, C., SISCHO, W.M., SUTHER, D., UTTERBACK, W. & WEIGLER, B.J., 1990. Prevalence of trichomoniasis among California beef herds. Journal of the American Veterinary Medical Association, 196, 1590-1593.
- BONDURANT, R.H., GAJADHAR, A., CAMPERO, C.M., JOHNSON, E., LUN, Z.R., NORDHAUSEN, R.W., VAN HOOSEAR, K.A., VILLANUEVA, M.R. & WALKER, R.L., 1999. Preliminary characterization of a Tritrichomonas foetus-like protozoan isolated from preputial smegma of virgin bulls. Bovine Practitioner, 33, 124-127.
- BONDURANT, R.H., HONIGBERG, B.M., 1994. Trichomonads of veterinary importance. In: KREIER, J.P., (ed). Parasitic protozoa. San Diego: Academic Press, 112-117.
- BONDURANT, R.H., 1985. Diagnosis, treatment and control of bovine trichomoniasis. Compendium of Continuing Education for the Practicing Veterinarian, 7, S179-S186.
- BONDURANT, R.H., 1997. Pathogenesis, diagnosis, and management of trichomoniasis in cattle. The Veterinary clinics of North America Food animal practice, 13, 345-361.
- BONDURANT, R.H., 2005. Venereal diseases of cattle: Natural history, diagnosis, and the role of vaccines in their control. Veterinary Clinics of North America - Food Animal Practice, 21, 383-408.
- BORCHARDT, K.A., NORMAN, B.N., THOMAS, M.W. & HARMON, W.M., 1992. Evaluation of a new culture method for diagnosing Tritrichomonas foetus infection. Veterinary Medicine, 87, 104.
- BURGESS, D.E., 1986. Tritrichomonas foetus: Preparation of monoclonal antibodies with effector function. Experimental Parasitology, 62, 266-274.
- CAMPERO, C.M., BALLABENE, N.C., CIPOLLA, A.C. & ZAMORA, A.S., 1987. Dual infection of bulls with campylobacteriosis and trichomoniasis: treatmet with dimetridazole chlorhydrate. Australian Veterinary Journal, 64, 320-321.
- CAMPERO, C.M., HIRST, R.G., LADDS, P.W., VAUGHAN, J.A., EMERY, D.L. & WATSON, D.L., 1990. Measurement of antibody in serum and genital fluids of bulls by ELISA after vaccination and challenge with Tritrichomonas foetus. Australian Veterinary Journal, 67, 175-178.
- CASTERIANO, A., MOLINI, U., KANDJUMBWA, K., KHAISEB, S., FREY, C.F. & SLAPETA, J., 2016. Novel genotype of Tritrichomonas foetus from cattle in Southern Africa. Parasitology, 143, 1954-1959.
- CHAPWANYA, A., USMAN, A.Y. & IRONS, P.C., 2016. Comparative aspects of immunity and vaccination in human and bovine trichomoniasis: a review. Tropical Animal Health and Production, 48, 1-7.
- CHRISTENSEN, H.R., CLARK, B.L. & PARSONSON, I.M., 1977. Incidence of Tritrichomonas foetus in young replacement bulls following introduction into an infected herd. Australian Veterinary Journal, 53, 132-134.
- CHRISTENSEN, H.R. & CLARK, B.L., 1979. Spread of Tritrichomonas foetus in beef bulls in an infected herd. Australian Veterinary Journal, 55, 205.
- CLARK, B.L. & DUFTY, J., 1986. The frequency of infertility and abortion in cows infected with Trichomonas foetus var. Brisbane. Australian Veterinary Journal, 63, 31-32.
- CLARK, B.L., DUFTY, J.H. & PARSONSON, I.M., 1977. Studies on the transmission of Tritrichomonas foetus. Australian Veterinary Journal, 53, 170-172.
- CLARK, B.L., DUFTY, J.H. & PARSONSON, I.M., 1983. The effect of Tritrichomonas foetus infection on calving rates in beef cattle. Australian Veterinary Journal, 60, 71-74.
- CLARK, B.L., PARSONSON, I.M. & DUFTY, J.H., 1974. Experimental infection of bulls with Tritrichomonas foetus. Australian Veterinary Journal, 50, 189-191.
- CLAVIJO, A., EROL, E., SNEED, L., SUN, F. & SWINFORD, A., 2011. The influence of temperature and simulated transport conditions of diagnostic samples on real-time polymerase chain reaction for the detection of Tritrichomonas foetus DNA. Journal of Veterinary Diagnostic Investigation, 23, 982-985.
- COBO, E.R., CORBEIL, L.B., GERSHWIN, L.J. & BONDURANT, R.H., 2009. Preputial cellular and antibody responses of bulls vaccinated and/or challenged with Tritrichomonas foetus. Vaccine, 28, 361-370.
- COBO, E.R., FAVETTO, P.H., LANE, V.M., FRIEND, A., VANHOOSER, K., MITCHELL, J. & BONDURANT, R.H., 2007. Sensitivity and specificity of culture and PCR of smegma samples of bulls experimentally infected with Tritrichomonas foetus. Theriogenology, 68, 853-860.
- CORBEIL, L.B., CAMPERO, C.M., RHYAN, J.C., ANDERSON, M.L., GERSHWIN, L.J., AGNEW, D.W., MUNSON, L. & BONDURANT, R.H., 2005. Uterine mast cells and immunoglobulin-E antibody responses during clearance of Tritrichomonas foetus. Veterinary Pathology, 42, 282-290.
- DE OLIVEIRA, J.M.B., BATISTA FILHO, A.F.B., BORGES, J., DE, M., SOARES, L.B.F., ORTEGA-MORA, L.M., BRANDESPIM, D.F., MOTA, R.A. & PINHEIRO JÚNIOR, J.W., 2016.Tritrichomonas foetus in bulls in the State of Pernambuco, Brazil. Revista Brasileira de Medicina Veterinaria, 38, 449-453.
- DENNETT, D.P., REECE, R.L., BARASA, J.O. & JOHNSON, R.H., 1974. Observations on the incidence and distribution of serotypes of Tritrichomonas foetus in beef cattle in north‐eastern australia. Australian Veterinary Journal, 50, 427-431.
- DEWELL, G.A., PHILLIPS, P.E., DOHLMAN, T.M., HARMON, K.M. & GAUGER, P.C., 2016. Validation of a gauze sponge sampling methodology to detect Tritrichomonas foetus by real-time PCR. Journal of Veterinary Diagnostic Investigation, 28, 595-598.
- ERASMUS, J.A., DE WET., J.A.L., VAN DER MERWE, H.E. & PIENAAR, G.C.J., 1989. Bovine trichomoniasis in the north Western Cape Province, western Transvaal and the Orange Free State. Journal of the South African Veterinary Association, 60, 51-52.
- FELLEISEN, R.S.J., 1997. Comparative genetic analysis of tritrichomonadid protozoa by the random amplified polymorphic DNA technique. Parasitology Research, 84, 153-156.
- FITZGERALD, P.R., 1963. Treatment of genital trichomoniasis in bulls. Journal of the American Veterinary Medical Association, 143, 259-262.
- FUCHS, L., FORT, M., CANO, D., BONETTI, C., GIMÉNEZ, H., VÁZQUEZ, P., BACIGALUPE, D., BRECCIA, J., CAMPERO, C.M. & OYHENART, J.A., 2017. Clearance of Tritrichomonas foetus in experimentally infected heifers protected with vaccines based on killed-T. foetus with different adjuvants. Vaccine, 35, 1341-1346.
- GAINES, J.D., 1990. Exploring the causes of subfertility in natural service dairy herds. Veterinary Medicine, 85, 296-302.
- GAINES, J.D., 1988. Tirchomoniasis in a dairy herd: control by artificial insemination and prostaglandin F2alpha treatment. Theriogenology, 29, 1367-1374.
- GARCÍA GUERRA, A., HILL, J.E., CAMPBELL, J., 2014. Use of pooled protozoal cultures of preputial scraping samples obtained from bulls for the detection of Tritrichomonas foetus by means of a real-time polymerase chain reaction. Journal of the American Veterinary Medical Association, 244, 352-356.
- GARCIA GUERRA, A., HILL, J.E., CAMPBELL, J., WALDNER, C.L. & HENDRICK, S., 2013. Sensitivity of a real-time polymerase chain reaction for Tritrichomonas fetus in direct individual and pooled preputial samples. Theriogenology, 80, 1097-1103.
- GASPARINI, G., VAGHI, M. & TARDAMO, A., 1963.Treatment of bovine trichomoniasis with metronidazole. Veterinary Record, 75, 940-943.
- GAULT, R.A., KVASNICKA, W.G., HANKS, D., HANKS, M., & HALL, M.R.,1995. Specific antibodies in serum and vaginal mucus of heifers inoculated with a vaccine containing Tritrichomonas foetus. American Journal of Veterinary Research, 56, 454-459.
- GAY, J.M., EBEL, E.D. & KEARLEY, W.P., 1996. Commingled grazing as a risk factor for trichomonosis in beef herds. Journal of the American Veterinary Medical Association, 209, 643-646.
- GIVENS, M.D., 2006. A clinical, evidence-based approach to infectious causes of infertility in beef cattle. Theriogenology, 66, 648-654.
- GOODGER, W.J. & SKIRROW, S.Z., 1986. Epidemiologic and economic analyses of an unusually long epizootic of trichomoniasis in a large California dairy herd. Journal of the American Veterinary Medical Association, 189, 772-776.
- GRAHN, R.A., BONDURANT, R.H., VAN HOOSEAR, K.A., WALKER, R.L. & LYONS, L.A., 2005. An improved molecular assay for Tritrichomonas foetus. Veterinary Parasitology, 127, 33-41.
- GREGORY, M.W., ELLIS, B. & REDWOOD, D.W., 1990. Comparison of sampling methods for teh detection of Tritrichomonas foetus infection in bulls. Veterinary Record, 127, 16.
- GROTELUESCHEN, D.M., CHENEY, J., HUDSON, D.B., SCHWEITZER, D.J., KIMBERLING, C.V., TATON-ALLEN, G.F., NIELSEN, K.A. & MARSH, D.J., 1994. Bovine trichomoniasis: Results of a slaughter survey in Colorado and Nebraska. Theriogenology, 42, 165-171.
- HALL, M.R., HUANG, J.C., OTA, R., REDELMAN, D., HANKS, D. & TAYLOR, R.E., 1986. Characterization of Tritrichomonas foetus antigens, using bovine antiserum. American Journal of Veterinary Research, 47, 2549-2553.
- HALL, M.R., KVASNICKA, W.G., HANKS, D., CHAVEZ, L. & SANDBLOM, D., 1993. Improved control of trichomoniasis with Trichomonas foetus vaccine. Agricultural Practice, 14, 29-34.
- HAMMOND, D.M. & BARTLETT, D.E., 1945. Pattern of fluctuation in numbers of Trichomonas foetus in preputial samples from infected bulls. American Journal of Veterinary Research, 6, 94-95.
- HENNING, K. & SAGER, H., 2005. Diagnosis of bovine trichomoniasis. Tierarztliche Umschau, 60, 349-351.
- HESS, E., 1951. Diagnose und Therapie der Trichomonadenseuche beim Zuchtstier. Tierarztliche Umschau, 6, 191-196.
- HO, M.S.Y., CONRAD, P.A., CONRAD, P.J., LEFEBVRE, R.B., PEREZ, E. & BONDURANT, R.H., 1994. Detection of bovine trichomoniasis with a specific DNA probe and PCR amplification system. Journal of Clinical Microbiology, 32, 98-104.
- HONIGBERG, B.M., 1978. Trichomonads of veterinary importance In: KREIER, J.P., (ed). Parasitic protozoa. Boca Raton (FL): Academic Press Inc., 207-273.
- HOWARD, T.H., VASQUEZ, L.A. & AMANN, R.P., 1982. Antibiotic control of Campylobacter fetus by three extenders of bovine semen. Journal of Dairy Science, 65, 1596-1600.
- HUANG, J.C., HANKS, D., KVASNICKA, W., HANKS, M. & HALL, M.R., 1989. Antigenic relationship among field isolates of Tritrichomonas foetus from cattle. American Journal of Veterinary Research, 50, 1064-1068.
- HUDSON, D.B., BALL, L., CHENEY, J.M., MORTIMER, R.G., BOWEN, R.A., MARSH, D.J. & PEETZ, R.H., 1993. Development and testing of a bovine trichomoniasis vaccine. Theriogenology, 39, 929-935.
- IKEDA, J.S., BONDURANT, R.H., CAMPERO, C.M. & CORBEIL, L.B., 1993. Conservation of a protective surface antigen of Tritrichomonas foetus. Journal of Clinical Microbiology, 31, 3289-3295.
- IKEDA, J.S., BONDURANT, R.H. & CORBEIL, L.B., 1995. Bovine vaginal antibody responses to immunoaffinity-purified surface antigen of Tritrichomonas foetus. Journal of Clinical Microbiology, 33, 1158-1163.
- IRONS, P.C., HENTON, M.M. & BERTSCHINGER, H.J., 2002. Collection of preputial material by scraping and aspiration for the diagnosis of Tritrichomonas foetus in bulls. Journal of the South African Veterinary Association, 73, 66-69.
- JENSEN, E.A. & HAMMOND, D.M., 1964. A morphological study of trichomonads and related flagellates from the bovine digestive tract. The Journal of protozoology, 11, 386-394.
- JIN, Y., SCHUMAKER, B., LOGAN, J. & YAO, C., 2014. Risk factors associated with bovine trichomoniasis in beef cattle identified by a questionnaire. Journal of Medical Microbiology, 63, 896-902.
- KENNEDY, J.A., PEARL, D., TOMKY, L. & CARMAN, J., 2008. Pooled polymerase chain reaction to detect Tritrichomonas foetus in beef bulls. Journal of Veterinary Diagnostic Investigation, 20, 97-99.
- KERR, W.R. & ROBERTSON, M., 1945. A note on the appearance of serological varieties among Trichomonas foetus strains isolated from infected cattle. Veterinary Record, 19, 221-222.
- KIMSEY, P.B., DARIEN, B.J., KENDRICK, J.W. & FRANTI, C.E., 1980. Bovine trichomoniasis: Diagnosis and treatment. Journal of the American Veterinary Medical Association, 177, 616-619.
- KIMSEY, P.B., 1997. Bovine trichomoniasis In: YOUNGQUIST, R.S., (ed). Current Therapy in Large Animal. Tehriogenology. Philadelphia: W. B. Saunders Company, 275-279.
- KVASNICKA, W.G., HANKS, D., HUANG, J.C., HALL, M.R., SANDBLOM, D., CHU, H.J., CHAVEZ, L. & ACREE, W.M., 1992. Clinical evaluation of the efficacy of inoculating cattle with a vaccine containing Tritrichomonas foetus. American Journal of Veterinary Research, 53, 2023-2027.
- LEVY, M.G., GOOKIN, J.L., POORE, M., BIRKENHEUER, A.J., DYKSTRA, M.J., LITAKER, R.W., 2003. Tritrichomonas foetus and not Pentatrichomonas hominis is the etiologic agent of feline trichomonal diarrhea. Journal of Parasitology, 89, 99-104.
- LITTLE, E.W., 1951. The treatment of trichomoniasis in bulls. The Irish Veterinary Journal, 5, 154-157.
- LUN, Z.R., CHEN, X.G., ZHU, X.Q., LI, X.R. & XIE, M.Q., 2005. Are Tritrichomonas suis and Tritrichomonas foetus synonyms? Trends in Parasitology, 21(3), 122-125
- MANDAL, P., CHATTERJEE, A., CHAKRABORTY, M., BIDYANTA, J., DE, B.N. & SEN, G.P., 1983. Cultural isolation and serological typing of Trichomonas foetus from breeding bulls in West Bengal. Indian Veterinary Journal, 60, 791-794.
- MARIANTE, R.M., LOPES, L.C. & BENCHIMOL, M., 2004. Tritrichomonas foetus pseudocysts adhere to vaginal epithelial cells in a contact-dependent manner. Parasitology Research, 92, 303-312.
- MARTIN-GOMEZ, S., GONZALEZ-PANIELLO, R., PEREIRA-BUENO, J. & ORTEGA-MORA, L.M., 1998. Prevalence of Tritrichomonas foetus infection in beef bulls in northwestern Spain. Veterinary Parasitology, 75, 265-268.
- MC COOL, C.J, TOWNSEND, M.P., WOLFE, S.G., SIMPSON, M.A., OLM, T.C., JAYAWARDHANA, G.A. & CARNEY, J.V., 1988. Prevalence of bovine venereal disease in the Victoria River District of the Northern Territory: likely economic effects and practicable control measures. Australian Veterinary Journal, 153-156.
- MC MILLEN, L. & LEW, A.E., 2006. Improved detection of Tritrichomonas foetus in bovine diagnostic specimens using a novel probe-based real time PCR assay. Veterinary Parasitology, 141, 204-215.
- MENDOZA-IBARRA, J.A., ORTEGA-MORA, L.M. & PEDRAZA-DIAZ, J., 2012. Differences in the prevalence of Tritrichomonas foetus in beef cattle farmed under extensive conditions in northern Spain. Veterinary Journal, 196, 547-549.
- MICKELSEN, W.D., PAISLEY, L.G. & ANDERSON, P.B., 1985. Prevalence of postservice pyometra in a herd of beef cows infected with trichomoniasis: A case report. Theriogenology, 25, 741-744.
- MORGAN, B.B., 1946. Bovine trichomoniasis. Minneapolis: Burgess Publishing Company
- MORIN-ADELINE, V., MUELLER, K. & CONESA, A., 2015. Comparative RNA-seq analysis of the Tritrichomonas foetus PIG30/1 isolate from pigs reveals close association with Tritrichomonas foetus BP-4 isolate 'bovine genotype'. Veterinary Parasitology, 212, 111-117.
- MUELLER, K., MORIN-ADELINE, V., GILCHRIST, K., BROWN, G. & ŠLAPETA, J., 2015. High prevalence of Tritrichomonas foetus 'bovine genotype' in faecal samples from domestic pigs at a farm where bovine trichomonosis has not been reported for over 30 years. Veterinary Parasitology, 212, 105-110.
- MUKHUFHI, N., IRONS, P.C., MICHEL, A. & PETA, F., 2003. Evaluation of a PCR test for the diagnosis of Tritrichomonas foetus infection in bulls: Effects of sample collection method, storage and transport medium on the test. Theriogenology, 60, 1269-1278.
- MUNDT, D., 1954. Beitrag zum Problem enes Entwichlungzyklus der Trichomonas foetus unter besonderer Berorcsichtigung vakuoliger Rundformen. Der Praktische Tierarzt, 6, 135-137.
- OKAFOR, C.C., STRICKLAND, L.G., JONES, B.M., KANIA, S., ANDERSON, D.E. & WHITLOCK, B.K., 2017. Prevalence of Tritrichomonas foetus in Tennessee bulls. Veterinary Parasitology, 243, 169-175.
- ONDRAK, J.D., KEEN, J.E., RUPP, G.P., KENNEDY, J.A., MCVEY, D.S. & BAKER, W.D., 2010. Repeated testing by use of culture and PCR assay to detect Tritrichomonas foetus carrier bulls in an infected Nebraska herd. Journal of the American Veterinary Medical Association, 237, 1068-1073.
- PAKANDL, M. & GRUBHOFFER, L., 1994. Some properties of sialic-acid binding systems in Tritrichomonas suis and Tritrichomonas foetus. Comparative Biochemistry and Physiology -- Part B: Biochemistry and Physiology, 10, 529-536.
- PALOMARES, R.A., HURLEY, D.J., CRUM, L.T., ROLLIN, E., COLLOP, T., WILLIARD, A. & CORBEIL, L.S., 2017. Serum, uterine, and vaginal mucosal IgG antibody responses against Tritrichomonas foetus after administration of a commercial killed whole T foetus vaccine in beef cows. Theriogenology, 87, 235-241.
- PAREEK, P.K., SHARMA, G.D., VYAS, U.K. & JATKAR, P.R., 1980. Note on Trichomonas foetus infection in native cattle of Rajasthan. Indian Journal of Animal Sciences, 50, 582-584.
- PARKER, S., CAMPBELL, J. & GAJADHAR, A., 2003. Comparison of the diagnostic sensitivity of a commercially available culture kit and a diagnostic culture test using Diamond's media for diagnosing Tritrichomonas foetus in bulls. Journal of Veterinary Diagnostic Investigation, 15, 460-465.
- PARSONSON, I.M., CLARK, B.L. & DUFTY, J., 1974. The pathogenesis of Tritrichomonas foetus infection in the bull. Australian Veterinary Journal, 50, 421-423.
- PARSONSON, I.M., CLARK, B.L. & DUFTY, J.H., 1976. Early pathogenesis and pathology of Tritrichomonas foetus infection in virgin heifers. Journal of Comparative Pathology, 86, 59-66.
- PEFANIS, S.M., HERR, S., VENTER, C.G., KRUGER, L.P., QUEIROGA, C.C., & AMARAL, L., 1988. Trichomoniasis and campylobacteriosis in bulls in the Republic of Transkei. Journal of the South African Veterinary Association, 59, 139-140.
- PEREIRA-NEVES, A., CAMPERO, C.M., MARTÍNEZ, A. & BENCHIMOL, M., 2011. Identification of Tritrichomonas foetus pseudocysts in fresh preputial secretion samples from bulls. Veterinary Parasitology, 175, 1-8.
- PEREIRA-NEVES, A., RIBEIRO, K.C. & BENCHIMOL, M., 2003. Pseudocysts in Trichomonads - New Insights. Protist, 154, 313-329.
- PEREZ, E., CONRAD, P.A., HIRD, D., ORTUNO, A., CHACON, J., BONDURANT, R. & NOORDHUIZEN, J., 1992. Prevalence and risk factors for Trichomonas foetus infection in cattle in north-eastern Costa Rica. Preventative Veterinary Medicine, 14, 155-165.
- PETER, D., 1997. Bovine venereal diseases. In: YOUNGQUIST, R.B., (ed). Current therapy in large animal theriogenology. Philadelphia: WB Saunders Co.
- RAE, D.O., CHENOWETH, P.J., GENHO, P.C., MCINTOSH, A.D., CROSBY, E. & MOORE, S.A., 1999. Prevalence of Tritrichomonas fetus in a bull population and effect on production in a large cow-calf enterprise. Journal of the American Veterinary Medical Association, 214, 1051-1055.
- RAE, D.O., CREWS, J.E., GREINER, E.C. & DONOVAN, G.A., 2004. Epidemiology of Tritrichomonas foetus in beef bull populations in Florida. Theriogenology, 61, 605-618.
- RAE, D.O. & CREWS, J.E., 2006. Tritrichomonas foetus. Veterinary Clinics of North America - Food Animal Practice, 22, 595-611.
- RAE, D.O., 1989. Impact of trichomoniasis on the cow-calf producer's profitability. Journal of the American Veterinary Medical Association, 194, 771-775.
- REECE, R.L., DENNETT, D.P. & JOHNSON, R.H., 1983. Some observations on cultural and transport conditions for Tritrichomonas foetus var. Brisbane. Australian Veterinary Journal, 60, 62-63.
- RHYAN, J.C., STACKHOUSE, L.L. & QUINN, W.J., 1995. Tissue-invasive Tritrichomonas foetus in four aborted bovine fetuses. Journal of Veterinary Diagnostic Investigation, 7, 409-412.
- RHYAN, J.C., STACKHOUSE, L.L. & QUINN, W.J., 1988. Foetal and placental lesions in bovine abortion due to Tritrichomonas foetus. Veterinary Pathology, 25, 350-355.
- RHYAN, J.C., WILSON, K.L., BURGESS, D.E., STACKHOUSE, L.L. & QUINN, W.J., 1995. Immunohistochemical detection of Tritrichomonas foetus in formalin-fixed, paraffin-embedded sections of bovine placenta and fetal lun. Journal of Veterinary Diagnostic Investigation, 7, 98-101.
- RHYAN, J.C., WILSON, K.L., WAGNER, B., ANDERSON, M.L., BONDURANT, R.H., BURGESS, D.E., MUTWIRI, G.K. & CORBEIL, L.B., 1999. Demonstration of Tritrichomonas foetus in the external genitalia and of specific antibodies in preputial secretions of naturally infected bulls. Veterinary Pathology, 36, 406-411.
- RIBEIRO, L.M., 1999. An efficient medium for the isolation of Trichomonas foetus. Onderstepoort Journal of Veterinary Research, 57, 209-210.
- RIEDMÜLLER, L., 1978. Tritrichomonas foetus. In: KREIER J.P., (ed). Parasitic Protozoa. New York: Academic Press
- ROBERTS, S.J., 1971. In: ROBERTS, S., (ed). Veterinary obstetrics and genital diseases. 2nd Edition. Ann Arbor (MI): Edwards Bros.
- SCHAUT, R.G., CORBEIL, L.B., BLAKE, C.N. & BREWER, M.T., 2017. Development of a bead-agglutination assay for rapid detection of Tritrichomonas foetus. Veterinary Parasitology, 243, 188-191.
- SCHÖNMANN, M.J., BONDURANT, R.H., GARDNER, I.A., VAN HOOSEAR, K., BALTZER, W. & KACHULIS, C., 1994. Comparison of sampling and culture methods for the diagnosis of Tritrichomonas foetus infection in bulls. Veterinary Record, 134.
- SINGH, B.N., LUCAS, J.J., HAYES, G.R., KUMAR, I. & BEACH, D.H., 2004. Tritrichomonas foetus induces apoptotic cell death in bovine vaginal epithelial cells. Infection and Immunity, 72, 4151-4158.
- SKIRROW, S.Z. & BONDURANT, R.H., 1988. Bovine trichomoniasis. Veterinary Bulletin, 58, 591-603.
- SKIRROW, S.Z. & BONDURANT, R.H., 1990. Immunoglobulin isotype of specific antibodies in reproductive tract secretions and sera in Tritrichomonas foetus-infected heifers. American Journal of Veterinary Research, 51, 645-653.
- SKIRROW, S.Z. & BONDURANT, R.H., 1990. Induced Tritrichomonas foetus infection in beef heifers. Journal of the American Veterinary Medical Association, 196, 885-889.
- SKIRROW, S.Z. & BONDURANT, R.H., 1988. Treatment of bovine trichomoniasis with ipronidazole. Australian Veterinary Journal, 65, 156.
- SKIRROW, S.Z., 1987. Identification of trichomonad-carrier cows. Journal of the American Veterinary Medical Association, 191, 553-554.
- SLAPETA, J., MÜLLER, N., STACK, C.M., WALKER, G., LEW-TABOR, A., TACHEZY, J. & FREY, C.F., 2012. Comparative analysis of Tritrichomonas foetus (Riedmüller, 1928) cat genotype, T. foetus (Riedmüller, 1928) cattle genotype and Tritrichomonas suis (Davaine, 1875) at 10 DNA loci. International Journal for Parasitology, 42, 1143-1149.
- SOTO, P. & PARMA, A.E., 1989. The immune response in cattle infected with Tritrichomonas foetus. Veterinary Parasitology, 33, 343-348.
- SZONYI, B., SRINATH, I., SCHWARTZ, A., CLAVIJO, A. & IVANEK, R., 2012. Spatio-temporal epidemiology of Tritrichomonas foetus infection in Texas bulls based on state-wide diagnostic laboratory data. Veterinary Parasitology, 186, 450-455.
- THOMAS, M.W., HARMON, W.M. & WHITE, C., 1991. An improved method for the detection of Tritrichomonas foetus infection by culture in bulls. Agricultural Practice, 11, 13-17.
- THOMFORD, J.W., TALBOT, J.A., IKEDA, J.S. & CORBEIL, L.B., 1996. Characterization of extracellular proteinases of Tritrichomonas foetus. Journal of Parasitology, 82, 112-117.
- VAN SOMEREN, V.D., 1946. A simple technique for the fixation and staining of Trichomonas in culture and vaginal mucus. Veterinary Journal, 102.
- VAN DE PLASSCHE, M., 1946. Het bestrijden van de trichomonas infectie bij stieren. Vlaamsch Diergeneeskundige Tijdschrift, 15, 1-15.
- VASQUEZ, R., AVILA, J.D., ROSSANIGO, C.E. & SAGER, R., 1984. Trichomoniasis y campylobacteriosis en la region semiarida Central Argentina. Veterinaria Argentina, 1, 942-947.
- VILLARROEL, A., CARPENTER, T.E. & BONDURANT, R.H., 2004. Development of a simulation model to evaluate the effect of vaccination against Tritrichomonas foetus on productive effieciency in beef herds. American Journal of Veterinary Research, 65, 770-775.
- WALDNER, C.L., PARKER, S., GESY, K.M., WAUGH, T., LANIGAN, E. & CAMPBELL, J.R., 2017. Application of direct polymerase chain reaction assays for Campylobacter fetus subsp. venerealis and Tritrichomonas foetus to screen preputial samples from breeding bulls in cow-calf herds in western Canada. Canadian Journal of Veterinary Research, 81, 91-99.
- WILLIAMS, O.J., MCEWAN, D., BERTRAM, J.D. & GILFEDDER, J., 1987. Treatment of trichomoniasis. Australian Veterinary Journal, 64, 159-160.
- WOUDWYK, M.A., MONTEAVARO, C.E., JENSEN, F., SOTO, P., BARBEITO, C.G. & ZENCLUSSEN, A.C., 2012. Study of the uterine local immune response in a murine model of embryonic death due to Tritrichomonas foetus. American Journal of Reproductive Immunology, 68, 128-137.
- YAO, C., 2013. Diagnosis of tritrichomonas foetus-infected bulls, an ultimate approach to eradicate bovine trichomoniasis in US cattle? Journal of Medical Microbiology, 62, 1-9.
- YULE, A., SKIRROW, S.Z. & BONDURANT, R.H., 1989. Bovine trichomoniasis. Parasitology Today, 5, 373-377.