- Infectious Diseases of Livestock
- Part 1
- Equine protozoal myeloencephalitis
- Vectors: Ticks
- Vectors: Tsetse flies
- Vectors: Muscidae
- Vectors: Tabanidae
- Vectors: Culicoides spp.
- Vectors: Mosquitoes
- Classification, epidemiology and control of arthropod-borne viruses
- Special factors affecting the control of livestock diseases in sub-Saharan Africa
- The control of infectious diseases of livestock: Making appropriate decisions in different epidemiological and socioeconomic conditions
- Infectious diseases of animals in sub-Saharan Africa: The wildlife⁄livestock interface
- Vaccination: An approach to the control of infectious diseases
- African animal trypanosomoses
- Dourine
- Trichomonosis
- Amoebic infections
- GENERAL INTRODUCTION: COCCIDIA
- Coccidiosis
- Cryptosporidiosis
- Toxoplasmosis
- Besnoitiosis
- Sarcocystosis
- Balantidiosis
- Leishmaniosis
- Neosporosis
- Equine protozoal myeloencephalitis
- GENERAL INTRODUCTION: BABESIOSES
- Bovine babesiosis
- Equine piroplasmosis
- Porcine babesiosis
- Ovine babesiosis
- GENERAL INTRODUCTION: THEILERIOSES OF CATTLE
- East Coast fever
- Corridor disease
- Zimbabwe theileriosis
- Turning sickness
- Theileria taurotragi infection
- Theileria mutans infection
- Theileria annulata theileriosis
- Theileriosis of sheep and goats
- Theileria buffeli⁄orientalis infection
- Non-pathogenic Theileria species in cattle
- GENERAL INTRODUCTION: RICKETTSIAL, CHLAMYDIAL AND HAEMOTROPIC MYCOPLASMAL DISEASES
- Heartwater
- Lesser known rickettsial infections in animals and humans
- Chlamydiosis
- Q fever
- Eperythrozoonosis
- Bovine Haemobartonellosis
- Potomac horse fever
- GENERAL INTRODUCTION: ANAPLASMOSES
- Bovine anaplasmosis
- Ovine and caprine anaplasmosis
Equine protozoal myeloencephalitis
This content is distributed under the following licence: Attribution-NonCommercial CC BY-NC  View Creative Commons Licence details here
View Creative Commons Licence details here
NJ Maclachlan and M-L Penrith (Editors). JP Dubey, Equine protozoal myeloencephalitis, 2018.

Equine protozoal myeloencephalitis
Previous authors: J P DUBEY
Current authors:
J P DUBEY - Senior scientist, MVSc, PhD, DSc, Animal Parasitic Diseases Laboratory, U.S. Department of Agriculture, Building 1001, Beltsville Agricultural Research Center, 10300 Baltimore Avenue, Beltsville, Maryland, 20705, United States of America
Introduction
Equine protozoal myeloencephalitis (EPM) is a neurological disease of horses in the Americas caused by Sarcocystis neurona. The clinical syndrome was recognized in the USA by Rooney125 in the 1960s and protozoa were first demonstrated in lesions in 1974.2, 19, 29 Initially, the protozoa were considered to be Toxoplasma gondii.2, 19, 29 Dubey et al. in 197429 reported lesions and protozoa in horses and distinguished the parasite from T. gondii. He also re-examined29, 30 the original cases of 19742, 19 and concluded that the parasite that causes EPM was not T. gondii but probably a species of Sarcocystis, which was later confirmed by ultrastructural examination.140 The protozoan was first cultured in 1991 from the spinal cord of an affected horse and was named Sarcocystis neurona.39 The development of a serological test in 199383 based on antigen prepared from in vitro cultured S. neurona merozoites led to the discovery that S. neurona infections are common in horses in the Americas.3, 4, 7, 148 The opossum was proposed as its definitive host74 and the life cycle was completed when the sarcocyst stage was identified.31 The development of an interferon gamma gene knock out (KO) mouse model for S. neurona made it possible to study its biology and chemotherapy.33, 50, 53, 54 Its genome was sequenced and annotated, potentially leading to development of better diagnostic methods and therapies.6 The history of EPM and its biology have been fully reviewed.33, 37, 48
Aetiology and life cycle
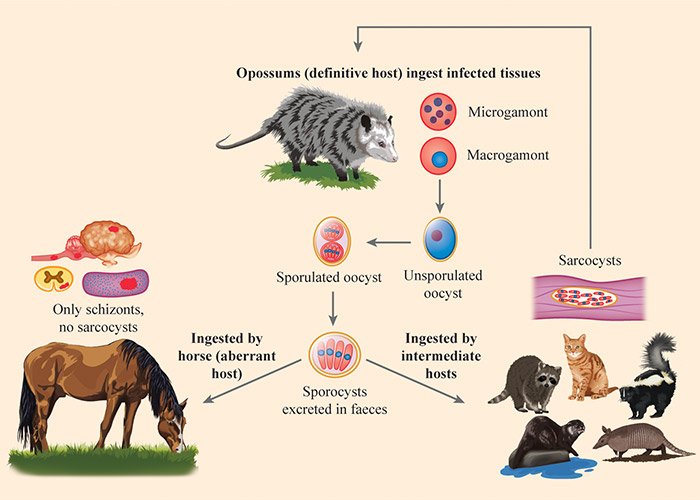
Sarcocystis neurona is a coccidian belonging to the family Sarcocystidae. Opossums of the genus Didelphis (D. virginiana and D. albiventris) are the definitive hosts (Figure 1).31, 50, 53, 55, 72, 74 Many other mammals are aberrant or intermediate hosts.48 Aberrant hosts are those in which only part of the asexual cycle (only schizonts) is completed; horses are aberrant hosts. Intermediate hosts are those in which the full asexual cycle (schizonts and sarcocysts) takes place. Raccoons (Procyon lotor), skunks (Mephitis mephitis), armadillos, domestic cats, and sea otters (Enhydra lutris) are its proven intermediate hosts as evidenced by excretion of S. neurona sporocysts by laboratory-raised opossums fed infected tissues.16, 60, 62, 145, 150 Sarcocystis neurona-like sarcocysts have also been reported in two dogs,152 one horse,114 one mink (Neovison vison),120 one bobcat (Lynx rufus),153 and one fisher (Pekania pennanti). There is a report of brown-headed cowbirds (Molothrus afer) as intermediate host of S. neurona102 but it needs confirmation because S. neurona is usually not infectious to birds.50
The sarcocysts are microscopic in size (about 700 μm long) with a 1 - 2 μm thick cyst wall. They have villar protrusions that are up to 2.8 µm long that do not reach bradyzoites.48, 52, 135 The bradyzoites are slender and tiny (about 5 μm long). The sporocysts are about 10 × 8 μm.93
A sexual cycle occurs in the lamina propria of the intestines of opossums. After gametogony, oocysts sporulate in the lamina propria of the small intestine and are excreted in the faeces. Sporocysts contain four sporozoites and a residual body.
Horses become infected by ingesting food and water contaminated with sporocysts. The only stages of S. neurona definitively identified in horses are schizonts and merozoites, which are confined to the brain and spinal cord.39, 48 (Figure 2) The precise time period and the route of dissemination of the parasite in horses are unknown.69 After oral inoculation of ponies with S. neurona sporocysts, evidence of invasion of brain occurred as early as seven days post- inoculation. In an experimental aberrant host, the KO mouse, sporozoites excyst in the small intestine and can be found in peripheral blood within one day of feeding sporocysts. Sarcocystis neurona first multiplies in visceral tissues and eventually resides in the central nervous system (CNS).32, 33, 52-54, 76 In the natural intermediate host (raccoon) schizogony occurred 7 to 22 days post- inoculation and mature sarcocysts were detected by 77 days post- infection.143 Sarcocystis bradyzoites are not infectious for intermediate hosts.65
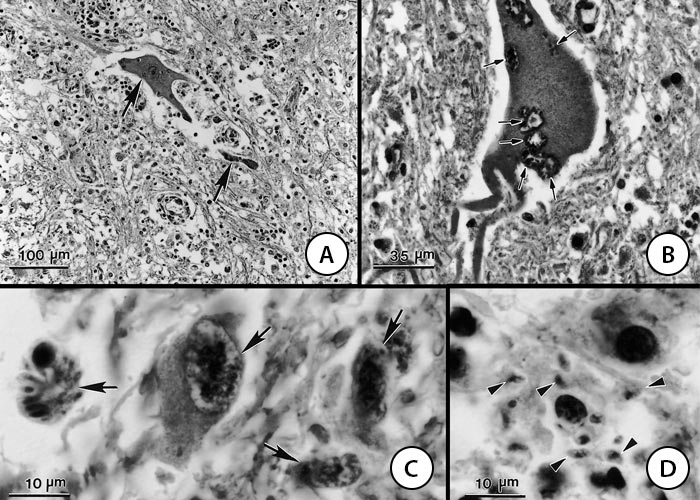
Figure 2 Sarcocystis neurona in sections of the spinal cord of a horse. Haematoxylin and eosin stain
- Perivascular infiltration and necrosis. Two infected neurons (arrows) but parasites are barely visible at this magnification
- Neuron containing several schizonts (arrows)
- Four schizonts (arrows) in various stages of development
- Intra- and extracellular individual merozoites (arrow heads)
Attempts to reproduce EPM after oral inoculation of horses with sporocysts were largely unsuccessful, including in immune-deficient foals.20, 21, 72, 98, 113, 129, 130, 137-139, 141
Sarcocystis neurona schizonts are found in neurons, various glial cells and resident macrophages in the brain and spinal cord.40, 48, 76, 142 They divide by endopolygeny:142 the nucleus of the parasite becomes multilobed and eventually each nuclear lobe is incorporated into two budding merozoites (Figure 3 and Figure 4). Schizonts in tissue sections are approximately 5 -35 × 5 -20 μm in size and contain up to 40 merozoites, sometimes arranged in a rosette around a residual body.55 (Figure 4) Merozoites are approximately 5 × 1 μm in size. Ultrastructurally, S. neurona schizonts are located in the host cell cytoplasm without a parasitophorous vacuole (Figure 4). Merozoites contain a conoid, micronemes, a nucleus, a prominent lipid body posterior to the nucleus, but no rhoptries.55, 142 Schizonts have been identified in extraneural tissues of some hosts including the muscles of a dog,36 turbinates of a ferret11 and retina of a dog36 and a sea otter.66 (Figure 4)
There are no reports of congenital infection in any terrestrial mammal. However, S. neurona DNA was detected in foetal tissues of eight marine mammals.1
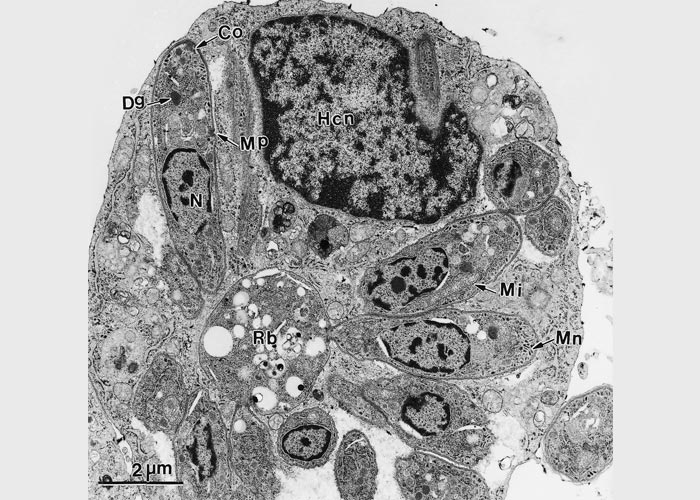
Figure 3 Transmission electron micrograph of a schizont of Sarcocystis neurona in a bovine monocyte showing merozoites budding from residual body (Rb). Some of the merozoites are still attached (arrowheads) to the residual body; conoid (Co); dense granule (Dg); host cell nucleus (Hcn); mitochondrion (Mi); microneme (Mn); micropore (Mp); nucleus of merozoite (N)
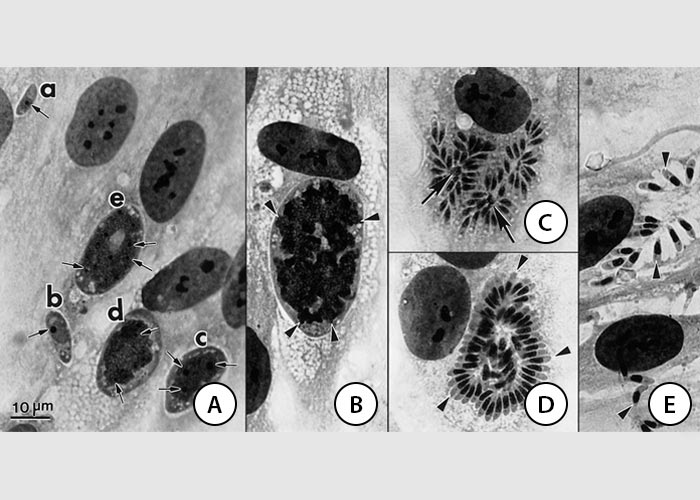
Figure 4 In vitro developmental stages of Sarcocystis neurona in bovine turbinate cells. Giemsa stain
- Immature schizonts with progressively differentiating nuclei from a-b. The nucleus in a and in b contains a single nucleolus whereas in c-e the nucleus has become lobed and has several nucleoli (arrows)
- Immature schizont with multilobed (arrrowheads) nucleus
- Three schizonts with merozoites arround a residual body (arrows)
- Mature schizont with peripherally situated merozoites (arrowheads). Some merozoites are at the centre of the schizont
- Free merozoites (arrowheads)
Epidemiology
Equine protozoal myeloencephalitis has been reported only in horses born and raised in the Americas, including Canada, USA, Panama and Brazil.8-10, 12, 15, 17, 24, 40, 56, 68, 72, 73, 77, 82, 84, 90, 94, 97, 99-101, 104-106, 112, 113, 131, 133, 149 Equine protozoal myeloencephalitis has been diagnosed on post-mortem in other parts of the world, including South Africa;48, 81, 89, 124 it occurred only in horses imported from the Americas. The distribution of EPM follows the geographic range of the opossum (D. virginiana) in North America and its relative D. albiventris in Central and South America.48
Serological surveys indicated that up to 89 per cent of horses in the USA,3, 4, 7, 58, 119, 127, 128, 148, 151 Brazil49, 86, 117 Canada,58 Mexico,156 Costa Rica23 and Argentina67, 111 have been exposed to S. neurona.37, 48 Although antibodies to S. neurona have been found in horses, donkeys, and mules,132 bona fide clinical disease has not been reported in equids other than horses, with the exception of a pony and a Grant’s zebra (Equus burchelli bohmi).57, 103 The prevalence of S. neurona antibody increases with age and is not related to breed, sex, and category (race/draught) of horses.131-133 Although horses of any breed or age may suffer from EPM, clinical disease occurs most frequently in three- to five-year-old Thoroughbreds and Standardbreds; the youngest horse affected was six months old. There are no documented cases of congenital S. neurona infection in terrestrial mammals.
Sarcocystis neurona antibodies have been reported in many other hosts, which were recently summarized.48
Sarcocystis neurona-like organisms have been detected in the CNS of raccoons,43, 45, 63, 85, 144, 147 (Figure 5 and Figure 6), mink,46 skunk (Mephitis mephitis),13, 44 domestic cats,5, 34, 47 domestic dogs,18, 36, 38, 41, 64, 79, 152 ferrets (Mustela putorius furo),11 Canada lynx (Lynx canadensis),75 fisher,80 and a bald eagle (Haliaeetus leucocephalus).116
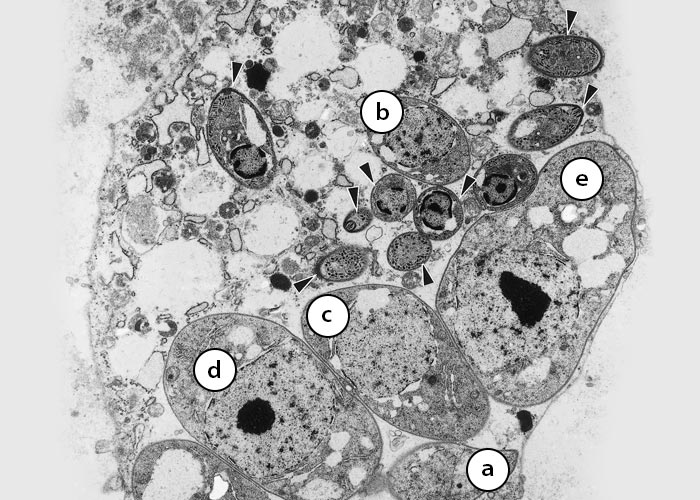
Figure 5 Degenerating host cell with eight individual merozoites and five developing schizonts (a to e) with single large nucleus. Sarcocystis neurona merozoites can form new schizonts without leaving the host cell. (From Dubey et al., 199139)
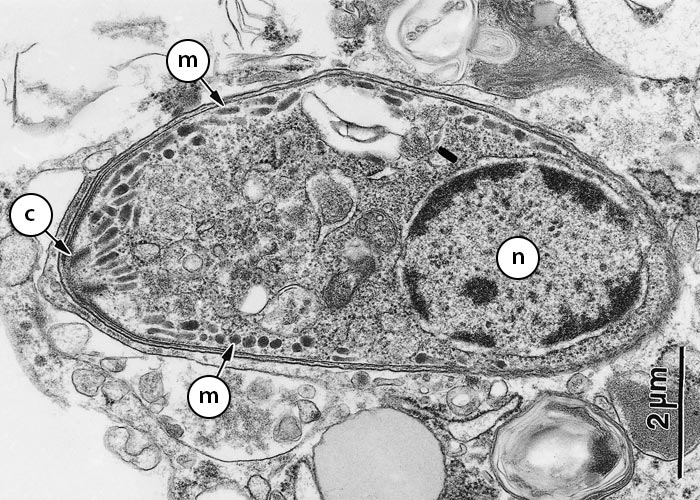
Figure 6 Transmission electron micrograph of a Sarcocystis neurona merozoite in the brain of the raccoon in Figure 5. Longitudinal section of a merozoite. Note the conoid (c), micronemes (m) and nucleus (n) but no rhoptries. (From Dubey et al., 199163, 64)
Sarcocystis neurona is also a major cause of mortality and encephalitis in several marine mammals in particular North American Southern sea otters.66, 95, 96, 107-109, 126, 146, 154, 155 Cases have been reported in Pacific harbour seals (Phoca vitulina).1, 91, 110, 115 However, viable S. neurona has been isolated only from horses, sea otters, harbour porpoise (Phocoena phocoena)1 and a California sea lion (Zalophus californianus).14 In most instances, the diagnosis was confirmed by immunohistochemical examination.
Opossums are also definitive hosts for at least three other species of Sarcocystis, namely, S. falcatula, S. speeri.31, 35, 50, 51 and S. lindsayi.59 Sporocysts of all three species are difficult to distinguish morphologically and progress has been made to develop genetic markers to distinguish them.48
Sarcocystis neurona sporocysts were reported in 6 to 31 per cent of opossums in USA.31, 42, 71, 122, 123 Sporocysts are found in the lamina propria of the intestines of opossums and can be excreted in their faeces for months. The excretion of sporocysts is irregular and they may be absent in faeces while present in the intestines.
Opossums can excrete millions of sporocysts in their faeces for months. Sporocysts are resistant to environmental influences, and most commonly used disinfectants do not kill S. neurona sporocysts. Heating to 60˚C for 1 minute will kill sporocysts, but exposure to 55˚C for 5 minutes will not.61 Although survival of sporocysts in different outdoor environmental conditions has not been tested, sporocysts may remain viable at 4˚C for months.
Clinical signs
Because S. neurona can infect any region of the CNS, clinical signs are related to the region affected.48, 55, 99, 100, 136 For example, lesions in the spinal cord may result in demarcated areas of spontaneous sweating or in loss of reflexes and cutaneous sensation.99 There may be foot dragging, difficulty in backing, or ataxia of the hind limbs or of all the limbs. Horses with lesions in the brain stem may be depressed, and have vestibular nerve dysfunction.99 There may be muscular atrophy, particularly of the gluteal muscles. Clinical signs may be sudden or progress over a period of weeks. Some horses have collapsed while racing.
Pathology
Lesions in horses are confined to the CNS, but occur more commonly in the spinal cord than in the brain.48 In the brain, the brain stem is affected more frequently than the cerebrum. Gross lesions consist of focal haemorrhages and areas of discolouration (from red to dark- brown) 1 to 30 mm in size (Figure 7). Any region of the spinal cord may be affected, and lesions may be confined to one segment of the spinal cord or be dispersed.2, 9, 17, 19, 25, 37, 40, 55
Microscopically, encephalomyelitis characterized by haemorrhages in and necrosis of the neuropil, as well as an infiltration of mononuclear cells in the neuropil and perivascularly, is observed. Chronic lesions consist of glial nodules and scattered areas of mononuclear cell infiltrations. Sarcocystis neurona parasites are usually associated with the lesions and may be scant or numerous, but some may be located away from the lesions.37 In most cases of EPM only a few organisms are found in the lesions.
Schizonts and merozoites are detected generally in the cytoplasm of neurons, glial cells and macrophages and rarely in vascular endothelial cells. Immunohistochemistry (IHC) reveals many more organisms than in sections stained only with haematoxylin and eosin (HE) (Figure 8).
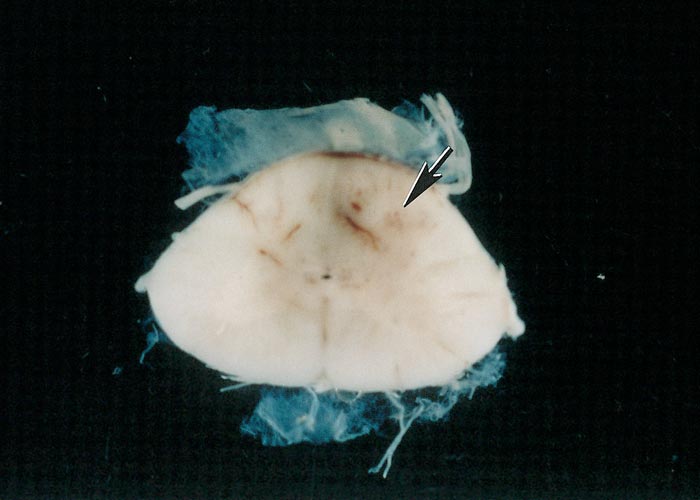
Figure 7 Cross-section of the spinal cord of a horse that had suffered from equine protozoal myeloencephalitis. Note area of discoloration (arrow)
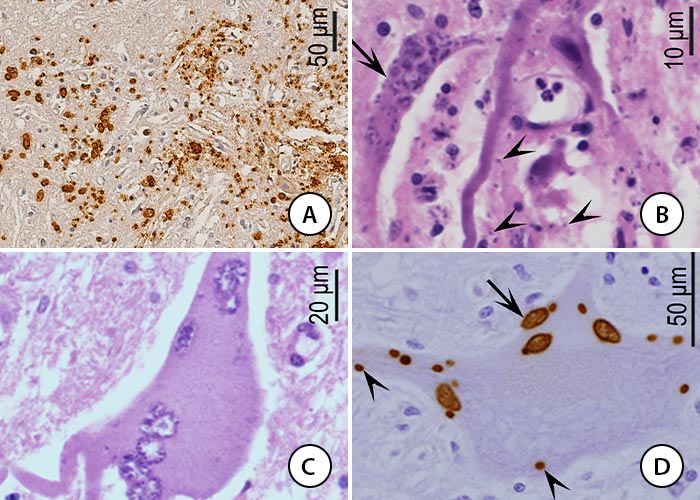
Figure 8 Histological sections of spinal cord of a horse with EPM. A and D, immunostaining with S. neurona antibody, B, C, haematoxylin and eosin stain
- Severe myelitis and numerous brownish stained parasites
- Myelin degeneration, and numerous parasites in an axon (arrow). Merozoites (arrowheads) are 2-3 mm long and difficult to observe
- Neuron with several schizonts
- Intraneuronal merozoites (arrowheads) and schizonts (arrow) in a neuron. (From Dubey et al., 2016)37
Diagnosis and differential diagnosis
The diagnosis of EPM is based on the clinical signs and the detection of S. neurona-specific antibodies in the cerebrospinal fluid (CSF).22, 55, 83, 99, 100, 121 The detection of antibodies in serum only indicates exposure and not necessarily clinical disease. A knowledge of the prevalence of subclinical S. neurona infections in the local equine population assists in the evaluation of serological results.
The diagnostic test used commercially to detect S. neurona-specific antibodies is an immunoblot using culture-derived S. neurona merozoite antigen.55, 83 Subsequently, an indirect fluorescent antibody test,26-28 various ELISAs48, 86-88, 157 and a direct agglutination test92 have been developed for the diagnosis of EPM.
The polymerase chain reaction (PCR) assay may also be used for the detection of S. neurona DNA in CSF.99 However, the sensitivity of the PCR appears to be much lower than initially estimated because intact merozoites rarely enter the CSF.48 Histopathology and IHC of CNS tissue section using S. neurona-specific antibodies can be useful in making a diagnosis.37 As stated earlier, EPM lesions are more common in spinal cord than the brain; however, any part of CNS can be affected (see Pathology).37
Microscopically, S. neurona may be confused with Neospora hughesi (see Neosporosis). Neospora spp. divide by endodyogeny and there is no schizont stage, whereas Sarcocystis spp. schizonts divide by endopolygeny.
As yet, no clinically confirmed cases of toxoplasmosis in horses have been diagnosed (see Toxoplasmosis). Equine protozoal myeloencephalitis should be considered in the differential diagnosis of diseases associated with impairment of function of the CNS, such as neosporosis, neurological involvement as a result of equid herpesvirus-1 infection, West Nile virus infection, rabies, equine encephalitides caused by alphavirus infections, and cervical stenosis.99, 100
Control
Horses with suspected EPM should be treated as soon as possible after the first clinical signs are recognized.48 Treatment results in recovery in 70 to 75 per cent of the affected horses, although these estimates may be somewhat high without prior confirmation of the diagnosis.48 Since it became evident that EPM has a protozoal aetiology, anti-protozoal drugs such as sulphonamides and pyrimethamine have been used to treat clinical cases. Nowadays, ponazuril (Marquis, Bayer), diclazuril (Protazil, Merck), and a sulphadiazine/pyrimethamine combination drug (ReBalance, PRN Pharmacal) are approved for treatment of EPM.42, 48, 118 Ponazuril and diclazuril are safe, with no side effects.78 Different supportive therapies have been used in affected horses70 including anti-inflammatory drugs such as phenylbutazone, flunixin meglumine, dimethylsulphoxide (DMSO) or steroids. Corticosteroids assist in stabilizing horses with serious neurologic involvement during the early period of treatment. Long-term treatment with steroids should be avoided due to their unknown effects in cases with EPM. Immuno-stimulants such as levamisole or Equimune have been recommended by some on the presumption that immunosuppression may play a role in the pathophysiological events of EPM but there is no information to suggest that these have any positive effects.48
The duration of treatment of clinical cases of EPM appears to be important. Horses should be evaluated clinically after one month of treatment with ponazuril or diclazuril. If there has been improvement but some clinical signs remain, then further treatment for at least a month is recommended. Recovered horses should be examined one month after treatment has been discontinued to ensure that there have not been relapses. Some horses may have severe relapses after prolonged therapy. The cause of relapses is unknown because the encysted stage, the sarcocyst, has not been confirmed. Whether schizonts can remain dormant in equine tissues is unknown.48
Preventing contamination of feed and water with opossum faeces is essential to prevent EPM as opossums can excrete millions of sporocysts in their faeces that may remain viable in the environment at 4oC for months (see Epidemiology).
There is no vaccine for EPM. A killed whole S. neurona merozoite vaccine was conditionally marketed by Fort Dodge in the USA but it has been withdrawn because of the difficulty of obtaining efficacy data.134
References
- BARBOSA, L., JOHNSON, C.K., LAMBOURN, D.M., GIBSON, A.K., HAMAN, K.H., HUGGINS, J.L., SWEENY, A.R., SUNDAR, N., RAVERTY, S.A., & GRIGG, M.E, 2015. A novel Sarcocystis neurona genotype XIII is associated with severe encephalitis in an unexpectedly broad range of marine mammals from the northeastern Pacific Ocean. International Journal for Parasitology, 45, 595-603.
- BEECH, J. & DODD, D. C., 1974. Toxoplasma-like encephalomyelitis in the horse. Veterinary Pathology, 11, 87-96.
- BENTZ, B. G., EALEY, K.A., MORROW, J., CLAYPOOL, P.L., & SALIKI, J.T, 2003. Seroprevalence of antibodies to Sarcocystis neurona in equids residing in Oklahoma. Journal of Veterinary Diagnostic Investigation, 15, 597-600.
- BENTZ, B. G., GRANSTROM, D. E. & STAMPER, S., 1997. Seroprevalence of antibodies to Sarcocystis neurona in horses residing in a county of South Eastern Pennsylvania. Journal of the American Veterinary Medical Association, 210, 517-518.
- BISBY, T. M., HOLMAN, P.J., PITOC, G.A., PACKER, R.A., THOMPSON, C.A., & RASKIN, R.E., 2010. Sarcocystis sp. encephalomyelitis in a cat. Veterinary Clinical Pathology, 39, 105-112.
- BLAZEJEWSKI, T., NURSIMULU, N., PSZENNY, V., DANGOUDOUBIYAM, S., NAMASIVAYAM, S., CHIASSON, M.A., CHESSMAN, K., TONKIN, M., SESHADRI SWAPNA, L., HUNG, S.S., BRIDGERS, J., RICKLEFS, S.M., BOULANGER, M.J., DUBEY, J.P., PORCELLA, S.F., KISSINGER, J.C., HOWE, D.K., GRIGG, M.E., & PARKINSON, J., 2015. Systems-based analysis of the Sarcocystis neurona genome identifies pathways that contribute to a heteroxenous life cycle. mBio, 6(1), e02445.
- BLYTHE, L. L., GRANSTROM, D.E., HANSEN, D.E., WALKER, L.L., BARTLETT, J., & STAMPER, S., 1997. Seroprevalence of antibodies to Sarcocystis neurona in horses residing in Oregon. Journal of the American Veterinary Medical Association, 210, 525-527.
- BOWMAN, D. D., CUMMINGS, J.F., DAVIS, S.W., DE LAHUNTA, A., DUBEY, J.P., SUTER, M.M., ROWLAND, P.H., & CONNER, D.L., 1992. Characterization of Sarcocystis neurona from a thoroughbred with equine protozoal myeloencephalitis. Cornell Veterinarian, 82, 41-52.
- BOY, M. G., GALLIGAN, D.T., & DIVERS, T.J., 1990. Protozoal encephalomyelitis in horses: 82 cases (1972-1986). Journal of the American Veterinary Medical Association, 196, 632-634.
- BREWER, B. D. & MAYHEW, I. G., 1988. Multifocal neurologic disease in a horse. Equine Veterinary Science, 8, 302-304.
- BRITTON, A. P., DUBEY, J.P., & ROSENTHAL, B.M., 2010. Rhinitis and disseminated disease in a ferret (Mustela putorius furo) naturally infected with Sarcocystis neurona. Veterinary Parasitology, 169, 226-231.
- BROWN, T. T. & PATTON, C. S., 1977. Protozoal encephalomyelitis in horses. Journal of the American Veterinary Medical Association, 171, 492.
- BURCHAM, G. N., RAMOS-VARA, J.A., & VEMULAPALLI, R., 2010. Systemic sarcocystosis in a striped skunk (Mephitis mephitis). Veterinary Pathology, 47, 560-564.
- CARLSON-BREMER, D. P., GULLAND, F.M.D., JOHNSON, C.K., COLEGROVE, K.M., & VAN BONN, W.G., 2012. Diagnosis and treatment of Sarcocystis neurona-induced myositis in a free-ranging California sea lion. Journal of the American Veterinary Medical Association, 240, 324-328.
- CHEADLE, M. A., LINDSAY, D. S., ROWE, S., DYKSTRA, C. C., WILLIAMS, M. A., SPENCER, J. A., TOIVIO-KINNUCAN, M. A., LENZ, S. D., NEWTON, J. C., ROLSMA, M. D. & BLAGBURN, B. L., 1999. Prevalence of antibodies to Neospora sp. in horses from Alabama and characterisation of an isolate recovered from a naturally infected horse. International Journal for Parasitology, 29, 1537-1543.
- CHEADLE, M. A., YOWELL, C.A., SELLON, D.C., HINES, M., GINN, P.E., MARSH, A.E., DAME, J.B., & GREINER, E.C, 2001. The striped skunk (Mephitis mephitis) is an intermediate host for Sarcocystis neurona. International Journal for Parasitology, 31, 843-849.
- CLARK, E. G., TOWNSEND, H.G.G., & MCKENZIE, N.T, 1981. Equine protozoal myeloencephalitis: a report of two cases from western Canada. Canadian Veterinary Journal, 22, 140-144.
- COOLEY, A. J., BARR, B. & REJMANEK, D., 2007. Sarcocystis neurona encephalitis in a dog. Veterinary Pathology, 44, 956-961.
- CUSICK, P. K., SELLS, D. M., HAMILTON, D. P. & HARDENBROOK, H. J., 1974. Toxoplasmosis in two horses. Journal of the American Veterinary Medical Association, 164, 77-80.
- CUTLER, T. J., MACKAY, R. J., GINN, P. E., GILLIS, K., TANHAUSER, S. M., LERAY, E. V., DAME, J. B. & GREINER, E. C., 2001. Immunoconversion against Sarcocystis neurona in normal and dexamethasone-treated horses challenged with S. neurona sporocysts. Veterinary Parasitology, 95, 197-210.
- CUTLER, T. J., MACKAY, R. J., GINN, P. E., GREINER, E. C., PORTER, R., YOWELL, C. A. & DAME, J. B., 1999. Are Sarcocystis neurona and Sarcocystis falcatula synonymous? A horse infection challenge. Journal of Parasitology, 85, 301-305.
- DAFT, B. M., BARR, B. C., GARDNER, I. A., READ, D., BELL, W., PEYSER, K. G., ARDANS, A., KINDE, H. & MORROW, J. K., 2002. Sensitivity and specificity of western blot testing of cerebrospinal fluid and serum for diagnosis of equine protozoal myeloencephalitis in horses with and without neurologic abnormalities. Journal of the American Veterinary Medical Association, 221, 1007-1013.
- DANGOUDOUBIYAM, S., OLIVEIRA, J. B., VÍQUEZ, C., GÓMEZ-GARCÍA, A., GONZÁLEZ, O., ROMERO, J. J., KWOK, O. C. H., DUBEY, J. P. & HOWE, D. K., 2011. Detection of antibodies against Sarcocystis neurona, Neospora spp., and Toxoplasma gondii in horses from Costa Rica. Journal of Parasitology, 97, 522-524.
- DAVISON, H. C., OTTER, A. & TREES, A. J., 1999. Estimation of vertical and horizontal transmission parameters of Neospora caninum infections in dairy cattle. International Journal for Parasitology, 29, 1683-1689.
- DORR, T. E., HIGGINS, R. J., DANGLER, C. A., MADIGAN, J. E. & WITHAM, C. L., 1984. Protozoal myeloencephalitis in horses in California. Journal of the American Veterinary Medical Association, 185, 801-802.
- DUARTE, P. C., DAFT, B. M., CONRAD, P. A., PACKHAM, A. E. & GARDNER, I. A., 2003. Comparison of a serum indirect fluorescent antibody test with two Western blot tests for the diagnosis of equine protozoal myeloencephalitis. Journal of Veterinary Diagnostic Investigation, 15, 8-13.
- DUARTE, P. C., DAFT, B. M., CONRAD, P. A., PACKHAM, A. E., SAVILLE, W. J., MACKAY, R. J., BARR, B. C., WILSON, W. D., NG, T., REED, S. M. & GARDNER, I. A., 2004. Evaluation and comparison of an indirect fluorescent antibody test for detection of antibodies to Sarcocystis neurona, using serum and cerebrospinal fluid of naturally and experimentally infected, and vaccinated horses. Journal of Parasitology, 90, 379-386.
- DUARTE, P. C., EBEL, E.D., TRAUB-DARGATZ, J., WILSON, W.D., CONRAD, P.A., & GARDNER, I.A., 2006. Indirect fluorescent antibody testing of cerebrospinal fluid for diagnosis of equine protozoal myeloencephalitis. American Journal of Veterinary Research, 67, 869-876.
- DUBEY, J. P., 1974. Toxoplasmosis in horses. Journal of the American Veterinary Medical Association, 165, 668.
- DUBEY, J. P., 1976. A review of Sarcocystis of domestic animals and of other coccidia of cats and dogs. Journal of the American Veterinary Medical Association, 169, 1061-1078.
- DUBEY, J. P., 2000. Prevalence of Sarcocystis species sporocysts in wild caught opossums (Didelphis virginiana). Journal of Parasitology, 86, 705-710.
- DUBEY, J. P., 2001. Migration and development of Sarcocystis neurona in tissues of interferon gamma knockout mice fed sporocysts from a naturally infected opossums. Veterinary Parasitology, 95, 341-351.
- DUBEY, J. P., 2001. Parasitemia and early tissue localization of Sarcocystis neurona in interferon gamma gene knockout mice fed sporocysts. Journal of Parasitology, 87, 1476-1479.
- DUBEY, J. P., BENSON, J. & LARSON, M. A., 2003. Clinical Sarcocystis neurona encephalomyelitis in a domestic cat following routine surgery. Veterinary Parasitology, 112, 261-267.
- DUBEY, J. P., BLACK, S. S., RICKARD, L. G., ROSENTHAL, B. M., LINDSAY, D. S., SHEN, S. K., KWOK, O. C. H., HURST, G. & RASHMIR-RAVEN, A., 2001. Prevalence of Sarcocystis neurona sporocysts in opossums (Didelphis virginiana) from rural Mississippi. Veterinary Parasitology, 95, 283-293.
- DUBEY, J. P., BLACK, S.S., VERMA, S.K., CALERO-BERNAL, R., MORRIS, E., HANSON, M.A., & COOLEY, A.J., 2014. Sarcocystis neurona schizonts-associated encephalitis, chorioretinitis, and myositis in a two-month-old dog simulating toxoplasmosis, and presence of mature sarcocysts in muscles. Veterinary Parasitology, 202, 194-200.
- DUBEY, J. P., CALERO-BERNAL, R., ROSENTHAL, B.M., SPEER, C.A., & FAYER, R., 2016. Sarcocystosis of animals and humans. 2nd edition. CRC Press, Boca Raton, Florida, USA, 1-481.
- DUBEY, J. P., CHAPMAN, J.L., ROSENTHAL, B.M., MENSE, M., & SCHUELER, R.L., 2006. Clinical Sarcocystis neurona, Sarcocystis canis, Toxoplasma gondii, and Neospora caninum infections in dogs. Veterinary Parasitology, 137, 36-49.
- DUBEY, J. P., DAVIS, S. W., SPEER, C. A., BOWMAN, D. D., DE LAHUNTA, A., GRANSTROM, D. E., TOPPER, M. J., HAMIR, A. N., CUMMINGS, J. F. & SUTER, M. M., 1991. Sarcocystis neurona n. sp. (Protozoa: Apicomplexa), the etiologic agent of equine protozoal myeloencephalitis. Journal of Parasitology, 77, 212-218.
- DUBEY, J. P., DAVIS, G.W., KOESTNER, A., & KIRYU, K., 1974. Equine encephalomyelitis due to a protozoan parasite resembling Toxoplasma gondii. Journal of the American Veterinary Medical Association, 165, 249-255.
- DUBEY, J. P., DUNCAN, D. E., SPEER, C. A. & BROWN, C., 1992. Congenital sarcocystosis in a two-day-old dog. Journal of Veterinary Diagnostic Investigation, 4, 89-93.
- DUBEY, J. P., FRITZ, D., LINDSAY, D. S., SHEN, S. K., KWOK, O. C. H. & THOMPSON, K. C., 2001. Diclazuril preventive therapy of gamma interferon knockout mice fed Sarcocystis neurona sporocysts. Veterinary Parasitology, 94, 257-263.
- DUBEY, J. P. & HAMIR, A. N., 2000. Immunohistochemical confirmation of Sarcocystis neurona infections in raccoons, mink, cat, skunk and pony. Journal of Parasitology, 86, 1150-1152.
- DUBEY, J. P., HAMIR, A. N., NIEZGODA, M. & RUPPRECHT, C. E., 1996. A Sarcocystis neurona-like organism associated with encephalitis in a striped skunk (Mephitis mephitis). Journal of Parasitology, 82, 172-174.
- DUBEY, J. P., HAMIR, A.N., HANLON, C.A., TOPPER, M.J., & RUPPRECHT, C.E., 1990. Fatal necrotizing encephalitis in a raccoon associated with a Sarcocystis-like protozoon. Journal of Veterinary Diagnostic Investigation, 2, 345-347.
- DUBEY, J. P. & HEDSTROM, O. R., 1993. Meningoencephalitis in mink associated with a Sarcocystis neurona-like organism. Journal of Veterinary Diagnostic Investigation, 5, 467-471.
- DUBEY, J. P., HIGGINS, R. J., BARR, B. C., SPANGLER, W. L., KOLLIN, B. & JORGENSEN, L. S., 1994. Sarcocystis-associated meningoencephalomyelitis in a cat. Journal of Veterinary Diagnostic Investigation, 6, 118-120.
- DUBEY, J. P., HOWE, D.K., FURR, M., SAVILLE, W.J., MARSH, A.E., REED, S.M., & GRIGG, M.E., 2015. An update on Sarcocystis neurona infections in animals and equine protozoal myeloencephalitis (EPM). Veterinary Parasitology, 209, 1-42.
- DUBEY, J. P., KERBER, C. E. & GRANSTROM, D. E., 1999. Serologic prevalence of Sarcocystis neurona, Toxoplasma gondii, and Neospora caninum in horses in Brazil. Journal of the American Veterinary Medical Association, 215, 970-972.
- DUBEY, J. P. & LINDSAY, D. S., 1998. Isolation in immunodeficient mice of Sarcocystis neurona from opossum (Didelphis virginiana) faeces, and its differentiation from Sarcocystis falcatula. International Journal for Parasitology, 28, 1823-1828.
- DUBEY, J. P. & LINDSAY, D. S., 1999. Sarcocystis speeri n. sp. (Protozoa: Sarcocystidae) from the opossum (Didelphis virginiana). Journal of Parasitology, 85, 903-909.
- DUBEY, J. P., LINDSAY, D. S., FRITZ, D. & SPEER, C. A., 2001. Structure of Sarcocystis neurona sarcocysts. Journal of Parasitology, 87, 1323-1327.
- DUBEY, J. P., LINDSAY, D. S., KERBER, C. E., KASAI, N., PENA, H. F. J., GENNARI, S. M., KWOK, O. C. H., SHEN, S. K. & ROSENTHAL, B. M., 2001. First isolation of Sarcocystis neurona from the South American opossum, Didelphis albiventris, from Brazil. Veterinary Parasitology, 95, 295-304.
- DUBEY, J. P., LINDSAY, D. S., KWOK, O. C. H. & SHEN, S. K., 2001. The gamma interferon knockout mouse model for Sarcocystis neurona: comparison of infectivity of sporocysts and merozoites and routes of inoculation. Journal of Parasitology, 87, 1171-1173.
- DUBEY, J. P., LINDSAY, D. S., SAVILLE, W. J. A., REED, S. M., GRANSTROM, D. E. & SPEER, C. A., 2001. A review of Sarcocystis neurona and equine protozoal myeloencephalitis (EPM). Veterinary Parasitology, 95, 89-131.
- DUBEY, J. P., MATTSON, D. E., SPEER, C. A., BAKER, R. J., MULROONEY, D. M., TORNQUIST, S. J., HAMIR, A. N. & GERROS, T. C., 1999. Characterization of Sarcocystis neurona isolate (SN6) from a naturally infected horse from Oregon. Journal of Eukaryotic Microbiology, 46, 500-506.
- DUBEY, J. P. & MILLER, S., 1986. Equine protozoal myeloencephalitis in a pony. Journal of the American Veterinary Medical Association, 188, 1311-1312.
- DUBEY, J. P., MITCHELL, S.M., MORROW, J.K., RHYAN, J.C., STEWART, L.M., GRANSTROM, D.E., ROMAND, S., THULLIEZ, P., SAVILLE, W.J., & LINDSAY, D.S., 2003. Prevalence of antibodies to Neospora caninum, Sarcocystis neurona, and Toxoplasma gondii in wild horses from central Wyoming. Journal of Parasitology, 89, 716-720.
- DUBEY, J. P., ROSENTHAL, B. M. & SPEER, C. A., 2001. Sarcocystis lindsayi n. sp. (Protozoa: Sarcocystidae) from the South American opossum, Didelphis albiventris from Brazil. Journal of Eukaryotic Microbiology, 48, 595-603.
- DUBEY, J. P., ROSYPAL, A. C., ROSENTHAL, B. M., THOMAS, N. J., LINDSAY, D. S., STANEK, J. F., REED, S. M. & SAVILLE, W. J. A., 2001. Sarcocystis neurona infections in sea otter (Enhydra lutris): evidence for natural infections with sarcocysts and transmission of infection to opossums (Didelphis virginiana). Journal of Parasitology, 87, 1387-1393.
- DUBEY, J. P., SAVILLE, W. J., SREEKUMAR, C., SHEN, S. K., LINDSAY, D. S., PENA, H. F., VIANNA, M. C., GENNARI, S. M. & REED, S. M., 2002. Effects of high temperature and disinfectants on the viability of Sarcocystis neurona sporocysts. Journal of Parasitology, 88, 1252-1254.
- DUBEY, J. P., SAVILLE, W. J. A., STANEK, J. F., LINDSAY, D. S., ROSENTHAL, B. M., OGLESBEE, M. J., ROSYPAL, A. C., NJOKU, C. J., STICH, R. W., KWOK, O. C. H., SHEN, S. K., HAMIR, A. N. & REED, S. M., 2001. Sarcocystis neurona infections in raccoons (Procyon lotor): evidence for natural infection with sarcocysts, transmission of infection to opossums (Didelphis virginiana), and experimental induction of neurologic disease in raccoons. Veterinary Parasitology, 100, 117-129.
- DUBEY, J. P., SLIFE, L. N., SPEER, C. A., LIPSCOMB, T. P. & TOPPER, M. J., 1991. Fatal cutaneous and visceral infection in a Rottweiler dog associated with a Sarcocystis-like protozoon. Journal of Veterinary Diagnostic Investigation, 3, 72-75.
- DUBEY, J. P., SPEER, C. A., HAMIR, A. N., TOPPER, M. J., BROWN, C. & RUPPRECHT, C. E., 1991. Development of a Sarcocystis-like apicomplexan protozoan in the brain of a raccoon (Procyon lotor). Journal of the Helminthological Society of Washington, 58, 250-255.
- DUBEY, J. P., SUNDAR, N., KWOK, O. C. H. & SAVILLE, W. J. A., 2013. Sarcocystis neurona infection in gamma interferon gene knockout (KO) mice: Comparative infectivity of sporocysts in two strains of KO mice, effect of trypsin digestion on merozoite viability, and infectivity of bradyzoites to KO mice and cell culture. Veterinary Parasitology, 196, 212-215.
- DUBEY, J. P. & THOMAS, N. J., 2011. Sarcocystis neurona retinochoroiditis in a sea otter (Enhydra lutris kenyoni). Veterinary Parasitology, 183, 156-159.
- DUBEY, J. P., VENTURINI, M. C., VENTURINI, L., MCKINNEY, J. & PECORARO, M., 1999. Prevalence of antibodies to Sarcocystis neurona, Toxoplasma gondii, and Neospora caninum in horses from Argentina. Veterinary Parasitology, 86, 59-62.
- DUNIGAN, C. E., OGLESBEE, M. J., PODELL, M., MITTEN, L. A. & REED, S. M., 1995. Seizure activity associated with equine protozoal myeloencephalitis. Progress in Veterinary Neurology, 6, 50-54.
- ELITSUR, E., MARSH, A. E., REED, S. M., DUBEY, J. P., OGLESBEE, M. J., MURPHY, J. E. & SAVILLE, W. J. A., 2007. Early migration of Sarcocystis neurona in ponies fed sporocysts. Journal of Parasitology, 93, 1222-1225.
- ELLISON, S. P. & LINDSAY, D. S., 2012. Decoquinate combined with levamisole reduce the clinical signs and serum SAG 1, 5, 6 antibodies in horses with suspected equine protozoal myeloencephalitis. International Journal of Applied Research in Veterinary Medicine, 10, 1-7.
- ELSHEIKHA, H. M., MURPHY, A. J. & MANSFIELD, L. S., 2004. Prevalence of Sarcocystis species sporocysts in Northern Virginia opossums (Didelphis virginiana). Parasitology Research, 93, 427-431.
- FENGER, C. K., GRANSTROM, D. E., GAJADHAR, A. A., WILLIAMS, N. M., MCCRILLIS, S. A., STAMPER, S., LANGEMEIER, J. L. & DUBEY, J. P., 1997. Experimental induction of equine protozoal myeloencephalitis in horses using Sarcocystis sp. sporocysts from the opossum (Didelphis virginiana). Veterinary Parasitology, 68, 199-213.
- FENGER, C. K., GRANSTROM, D. E., LANGEMEIER, J. L. & STAMPER, S., 1997. Epizootic of equine protozoal myeloencephalitis on a farm. Journal of the American Veterinary Medical Association, 210, 923-927.
- FENGER, C. K., GRANSTROM, D. E., LANGEMEIER, J. L., STAMPER, S., DONAHUE, J. M., PATTERSON, J. S., GAJADHAR, A. A., MARTENIUK, J. V., XIAOMIN, Z. & DUBEY, J. P., 1995. Identification of opossums (Didelphis virginiana) as the putative definitive host of Sarcocystis neurona. Journal of Parasitology, 81, 916-919.
- FOREST, T. W., ABOU-MADI, N., SUMMERS, B. A., TORNQUIST, S. J. & COOPER, B. J., 2001. Sarcocystis neurona-like encephalitis in a Canada lynx (Felis lynx canadensis). Journal of Zoo and Wildlife Medicine, 31, 383-387.
- FRITZ, D. L. & DUBEY, J. P., 2002. Pathology of Sarcocystis neurona in interferon-gamma gene knockout mice. Veterinary Pathology, 39, 137-140.
- FURR, M., MACKAY, R., GRANSTROM, D., SCHOTT, H., & ANDREWS, F., 2002. Clinical diagnosis of equine protozoal myeloencephalitis (EPM). Journal of Veterinary Internal Medicine, 16, 618-621.
- FURR, M., MCKENZIE, H., SAVILLE, W.J.A., DUBEY, J.P., REED, S.M., & DAVIS, W., 2006. Prophylactic admini stration of ponazuril reduces clinical signs and delays seroconversion in horses challenged with Sarcocystis neurona. Journal of Parasitology, 92, 637-643.
- GERHOLD, R., NEWMAN, S.J., GRUNENWALD, G.M., CREWS, A., HODSHON, A., & SU, C., 2014. Acute onset of encephalomyelitis with atypical lesions associaed with dual infection of Sarcocystis neurona and Toxoplasma gondii. Veterinary Parasitology, 205, 697-701.
- GERHOLD, R. W., HOWERTH, E.W., & LINDSAY, D.S., 2005. Sarcocystis neurona-associated meningoencephalitis and description of intramuscular sarcocysts in a fisher (Martes pennanti). Journal of Wildlife Diseases, 41, 224-230.
- GOEHRING, L. S. & SLOET VAN OLDRUITENBORGH-OOSTERBAAN, M. M., 2001. Equine protozoal myeloencephalitis (EPM) in the Netherlands? Tijdschrift voor Diergeneeskunde, 126, 346-351.
- GRANSTROM, D. E., ALVAREZ, O., DUBEY, J.P., COMER, P.F., & WILLIAMS, N.M., 1992. Equine protozoal myelitis in Panamanian horses and isolation of Sarcocystis neurona. Journal of Parasitology, 78, 909-912.
- GRANSTROM, D. E., DUBEY, J.P., DAVIS, S.W., FAYER, R., FOX, J.C., POONACHA, K.B., GILES, R.C., & COMER, P.F., 1993. Equine protozoal myeloencephalitis: antigen analysis of cultured Sarcocystis neurona merozoites. Journal of Veterinary Diagnostic Investigation, 5, 88-90.
- HAMIR, A. N., MOSER, G., GALLIGAN, D.T., DAVIS, S.W., GRANSTROM, D.E., & DUBEY, J.P., 1993. Immunohistochemical study to demonstrate Sarcocystis neurona in equine protozoal myeloencephalitis. Journal of Veterinary Diagnostic Investigation, 5, 418-422.
- HAMIR, A. N. & DUBEY, J. P., 2001. Myocarditis and encephalitis associated with Sarcocystis neurona infection in raccoons (Procyon lotor). Veterinary Parasitology, 95, 335-340.
- HOANE, J. S., GENNARI, S.M., DUBEY, J.P., RIBEIRO, M.G., BORGES, A.S., YAI, L.E.O., AGUIAR, D.M., CAVALCANTE, G.T., BONESI, G.L., & HOWE, D.K., 2006. Prevalence of Sarcocystis neurona and Neospora spp. infection in horses from Brazil based on presence of serum antibodies to parasite surface antigen. Veterinary Parasitology, 136, 155-159.
- HOANE, J. S., MORROW, J.K., SAVILLE, W.J., DUBEY, J.P., GRANSTROM, D.E., & HOWE, D.K., 2005. Enzyme-linked immunosorbent assays for the detection of equine antibodies specific to Sarcocystis neurona surface antigens. Clinical and Diagnostic Laboratory Immunology, 12, 1050-1056.
- HOWE, D. K., GAJI, R.Y., MARSH, A.E., PATIL, B.A., SAVILLE, W.J., LINDSAY, D.S., DUBEY, J.P., & GRANSTROM, D.E., 2008. Strains of Sarcocystis neurona exhibit differences in their surface antigens, including the absence of the major surface antigen SnSAG1. International Journal for Parasitology, 38, 623-631.
- KATAYAMA, Y., WADA, R., KANEMARU, T., SASAGAWA, T., UCHIYAMA, T., MATSUMURA, T., & ANZAI, T., 2003. First case report of Sarcocystis neurona-induced equine protozoal myeloencephalitis in Japan. Journal of Veterinary Medical Science, 65, 757-759.
- KISTHARDT, K. & LINDSAY, D. S., 1997. Equine protozoal myeloencephalitis. Equine Practice, 19, 8-13.
- LAPOINTE, J. M., DUIGNAN, P.J., MARSH, A.E., GULLAND, F.M., BARR, B.C., NAYDAN, D.K., KING, D.P., FARMAN, C.A., HUNTINGDON, K.A.B., & LOWENSTINE, L.J., 1998. Meningoencephalitis due to a Sarcocystis neurona-like protozoan in Pacific harbor seals (Phoca vitulina richardsi). Journal of Parasitology, 84, 1184-1189.
- LINDSAY, D. S. & DUBEY, J. P., 2001. Direct agglutination test for the detection of antibodies to Sarcocystis neurona in experimentally infected animals. Veterinary Parasitology, 95, 179-186.
- LINDSAY, D. S., MITCHELL, S. M., VIANNA, M. C. & DUBEY, J. P., 2004. Sarcocystis neurona (Protozoa: Apicomplexa): Description of oocysts, sporocysts, sporozoites, excystation, and early development. Journal of Parasitology, 90, 461-465.
- LINDSAY, D. S., STEINBERG, H., DUBIELZIG, R. R., SEMRAD, S. D., KONKLE, D. M., MILLER, P. E. & BLAGBURN, B. L., 1996. Central nervous system neosporosis in a foal. Journal of Veterinary Diagnostic Investigation, 8, 507-510.
- LINDSAY, D. S., THOMAS, N. J. & DUBEY, J. P., 2000. Biological characterisation of Sarcocystis neurona from a Southern sea otter (Enhydra lutris nereis). International Journal for Parasitology, 30, 617-624.
- LINDSAY, D. S., THOMAS, N. J., ROSYPAL, A. C. & DUBEY, J. P., 2001. Dual Sarcocystis neurona and Toxoplasma gondii infection in a Northern sea otter from Washington state, USA. Veterinary Parasitology, 97, 319-327.
- LOMBARDO DE BARROS, C. S., DE BARROS, S. S. & DOS SANTOS, M. N., 1986. Equine protozoal myeloencephalitis in southern Brazil. The Veterinary Record, 119, 283-284.
- LONG, M. T., MINES, M. T., KNOWLES, D. P., TANHAUSER, S. M., DAME, J. B., CUTLER, T. J., MACKAY, R. J. & SELLON, D. C., 2002. Sarcocystis neurona: parasitemia in a severe combined immunodeficient (SCID) horse fed sporocysts. Experimental Parasitology, 100, 150-154.
- MACKAY, R. J., 1997. Equine protozoal myeloencephalitis. Veterinary Clinics of North America: Equine Practice, 13, 79-96.
- MACKAY, R. J., DAVIS, S. W. & DUBEY, J. P., 1992. Equine protozoal myeloencephalitis. Compendium on Continuing Education for the Practicing Veterinarian, 14, 1359-1367.
- MADIGAN, J. E. & HIGGINS, R. J., 1987. Equine protozoal myeloencephalitis. Veterinary Clinics of North America: Equine Practice, 3, 397-403.
- MANSFIELD, L. S., MEHLER, S., NELSON, K., ELSHEIKHA, H. M., MURPHY, A. J., KNUST, B., TANHAUSER, S. M., GEARHART, P. M., ROSSANO, M. G., BOWMAN, D. D., SCHOTT, H. C. & PATTERSON, J. S., 2008. Brown-headed cowbirds (Molothrus ater) harbor Sarcocystis neurona and act as intermediate hosts. Veterinary Parasitology, 153, 24-43.
- MARSH, A. E., DENVER, M., HILL, F. I., MCELHANEY, M. R., TRUPKIEWICZ, J. G., STEWART, J. & TELL, L., 2000. Detection of Sarcocystis neurona in the brain of a Grant's zebra (Equus burchelli bohmi). Journal of Zoo and Wildlife Medicine, 31, 82-86.
- MARSH, A. E., JOHNSON, P. J., RAMOS-VARA, J. & JOHNSON, G. C., 2001. Characterization of a Sarcocystis neurona isolate from a Missouri horse with equine protozoal myeloencephalitis. Veterinary Parasitology, 95, 143-154.
- MASRI, M. D., LÓPEZ DE ALDA, J. & DUBEY, J. P., 1992. Sarcocystis neurona-associated ataxia in horses in Brazil. Veterinary Parasitology, 44, 311-314.
- MAYHEW, I. G. & GREINER, E. C., 1986. Protozoal diseases. Veterinary Clinics of North America: Equine Practice, 2, 439-459.
- MILLER, M. A., BARR, B. C., NORDHAUSEN, R., JAMES, E. R., MAGARGAL, S. L., MURRAY, M., CONRAD, P. A., TOY-CHOUTKA, S., JESSUP, D. A. & GRIGG, M. E., 2009. Ultrastructural and molecular confirmation of the development of Sarcocystis neurona tissue cysts in the central nervous system of southern sea otters (Enhydra lutris nereis). International Journal for Parasitology, 39, 1363-1372.
- MILLER, M. A., CONRAD, P.A., HARRIS, M., HATFIELD, B., LANGLOIS, G., JESSUP, D.A., MAGARGAL, S.L., PACKHAM, A.E., TOY-CHOUTKA, S., MELLI, A.C., MURRAY, M.A., GULLAND, F.M., & GRIGG, M.E., 2010. A protozoal-associated epizootic impacting marine wildlife: Mass-mortality of southern sea otters (Enhydra lutris nereis) due to Sarcocystis neurona infection. Veterinary Parasitology, 172, 183-194.
- MILLER, M. A., CROSBIE, P. R., SVERLOW, K., HANNI, K., BARR, B. C., KOCK, N., MURRAY, M. J., LOWENSTINE, L. J. & CONRAD, P. A., 2001. Isolation and characterization of Sarcocystis from brain tissue of a free-living southern sea otter (Enhydra lutris nereis) with fatal meningoencephalitis. Parasitology Research, 87, 252-257.
- MILLER, M. A., SVERLOW, K., CROSBIE, P. R., BARR, B. C., LOWENSTINE, L. J., GULLAND, F. M., PACKHAM, A. & CONRAD, P. A., 2001. Isolation and characterization of two parasitic protozoa from a pacific harbor seal (Phoca vitulina richardsi) with meningoencephalomyelitis. Journal of Parasitology, 87, 816-822.
- MORÉ, G., VISSANI, A., PARDINI, L., MONINA, M., MURIEL, M., HOWE, D., BARRANDEGUY, M., & VENTURINI, M.C, 2014. Seroprevalence of Sarcocystis neurona and its association with neurologic disorders in Argentinean horses. Journal of Equine Veterinary Science, 34, 1051-1054.
- MORLEY, P. S., TRAUB-DARGATZ, J.L., BENEDICT, K.M., SAVILLE, W.J.A., VOELKER, L.D., & WAGNER, B.A., 2008. Risk factors for owner-reported occurrence of equine protozoal myeloencephalitis in the US equine population. Journal of Veterinary Internal Medicine, 22, 616-629.
- MORLEY, S., TRAUB-DARGATZ, J., SAVILLE, W., WAGNER, B., GARBER, L., & HILLBERG-SEITZINGER, A., 2001. Equine protozoal myeloencephalitis. Journal of Equine Veterinary Science, 21, 262-270.
- MULLANEY, T., MURPHY, A.J., KIUPEL, M., BELL, J.A., ROSSANO, M.G., & MANSFIELD, L.S., 2005. Evidence to support horses as natural intermediate hosts for Sarcocystis neurona. Veterinary Parasitology, 133, 27-36.
- MYLNICZENKO, N. D., KEARNS, K.S., & MELLI, A.C., 2008. Diagnosis and treatment of Sarcocystis neurona in a captive harbor seal (Phoca vitulina). Journal of Zoo and Wildlife Medicine, 39, 228-235.
- OLSON, E. J., WÜNSCHMANN, A., & DUBEY, J.P., 2007. Sarcocystis sp.-associated meningoencephalitis in a bald eagle (Haliaeetus leucocephalus). Journal of Veterinary Diagnostic Investigation, 19, 564-568.
- PIVOTO, F. L., DE MACÊDO JUNIOR, A.G., DA SILVA, M.V., FERREIRA, F.B., SILVA, D.A.O., POMPERMAYER, E., SANGIONI, L.A., MINEO, T.W.P., & VOGEL, F.S.F., 2014. Serological status of mares in parturition and the levels of antibodies (IgG) against protozoan family Sarcocystidae from their pre colostral foals. Veterinary Parasitology, 199, 107-111.
- PUSTERLA, N., PACKHAM, A., MACKIE, S., KASS, P.H., HUNYADI, L., & CONRAD, P.A., 2015. Daily feeding of diclazuril top dress pellets in foals reduces seroconversion to Sarcocystis neurona. Veterinary Journal, 206, 236-238.
- PUSTERLA, N., TAMEZ-TREVINO, E., WHITE, A., VANGEEM, J., PACKHAM, A., CONRAD, P.A., & KASS, P., 2014. Comparison of prevalence factors in horses with and without seropositivity to Neospora hughesi and/or Sarcocystis neurona. Veterinary Journal, 200, 332-334.
- RAMOS-VARA, J. A., DUBEY, J.P., WATSON, G.L., WINN-ELLIOT, M., PATTERSON, J.S., & YAMINI, B., 1997. Sarcocystosis in mink (Mustela vison). Journal of Parasitology, 83, 1198-1201.
- REED, S. M., HOWE, D.K., MORROW, J.K., GRAVES, A., YEARGAN, M.R., JOHNSON, A.L., MACKAY, R.J., FURR, M., SAVILLE, W.J., & WILLIAMS, N.M., 2013. Accurate antemortem diagnosis of equine protozoal myeloencephalitis (EPM) based on detecting intrathecal antibodies against Sarcocystis neurona using the SnSAG2 and SnSAG4/3 ELISAs. Journal of Veterinary Internal Medicine, 27, 1193-1200.
- REJMANEK, D., VANWORMER, E., MILLER, M.A., MAZET, J.A.K., NICHELASON, A.E., MELLI, A.C., PACKHAM, A.E., JESSUP, D.A., & CONRAD, P.A., 2009. Prevalence and risk factors associated with Sarcocystis neurona infections in opossums (Didelphis virginiana) from central California. Veterinary Parasitology, 166, 8-14.
- RICKARD, L. G., BLACK, S.S., RASHMIR-RAVEN, A., HURST, G., & DUBEY, J.P., 2001. Risk factors associated with the presence of Sarcocystis neurona sporocysts in opossums (Didelphis virginiana). Veterinary Parasitology, 102, 179-184.
- RONEN, N., 1992. Putative equine protozoal myeloencephalitis in an imported Arabian filly. Journal of the South African Veterinary Association, 63, 78-79.
- ROONEY, J. R., PRICKETT, M.E., DELANEY, F.M., & CROWE, M.W., 1970. Focal myelitis-encephalitis in horses. Cornell Veterinarian, 50, 494-501.
- ROSONKE, B. J., BROWN, S.R., TORNQUIST, S.J., SNYDER, S.P., GARNER, M.M., & BLYTHE, L.L., 1999. Encephalomyelitis associated with a Sarcocystis neurona-like organism in a sea otter Journal of the American Veterinary Medical Association, 215, 1839-1842.
- ROSSANO, M. G., KANEENE, J.B., MARTENIUK, J.V., BANKS, B.D., SCHOTT, H.C., & MANSFIELD, L.S., 2001. The seroprevalence of antibodies to Sarcocystis neurona in Michigan equids. Preventive Veterinary Medicine, 48, 113-128.
- ROSSANO, M. G., KANEENE, J.B., MARTENIUK, J.V., BANKS, B.D., SCHOTT, H.C., & MANSFIELD, L.S., 2003. A herd-level analysis of risk factors for antibodies to Sarcocystis neurona in Michigan equids. Preventive Veterinary Medicine, 57, 7-13.
- ROSSANO, M. G., SCHOTT, H. C., MURPHY, A. J., KANEENE, J. B., SELLON, D. C., HINES, M. T., HOCHSTATTER, T., BELL, J. A. & MANSFIELD, L. S., 2005. Parasitemia in an immunocompetent horse experimentally challenged with Sarcocystis neurona sporocysts. Veterinary Parasitology, 127, 3-8.
- ROSSANO, M. G., SCHOTT, H.C., KANEENE, J.B., MURPHY, A.J., KRUTTLIN, E.A., HINES, M.T., SELLON, D.C., PATTERSON, J.S., ELSHEIKHA, H.M., DUBEY, J.P., & MANSFIELD, L.S., 2005. Effect of daily administration of pyrantel tartrate in preventing infection in horses experimentally challenged with Sarcocystis neurona. American Journal of Veterinary Research, 66, 846-852.
- SAVILLE, W. J., MORLEY, P.S., REED, S.M., GRANSTROM, D.E., KOHN, C.W., HINCHCLIFF, K.W., & WITTUM, T.E., 2000. Evaluation of risk factors associated with clinical improvement and survival of horses with equine protozoal myeloencephalitis. Journal of the American Veterinary Medical Association, 217, 1181-1185.
- SAVILLE, W. J., REED, S.M., GRANSTROM, D.E., HINCHCLIFF, K.W., KOHN, C.W., WITTUM, T.E., & STAMPER, S., 1997. Seroprevalence of antibodies to Sarcocystis neurona in horses residing in Ohio. Journal of the American Veterinary Medical Association, 210, 519-524.
- SAVILLE, W. J., REED, S.M., MORLEY, P.S., GRANSTROM, D.E., KOHN, C.W., HINCHCLIFF, K.W., & WITTUM, T.E., 2000. Analysis of risk factors for the development of equine protozoal myeloencephalitis in horses. Journal of the American Veterinary Medical Association, 217, 1174-1180.
- SAVILLE, W. J. A., DUBEY, J.P., MARSH, A.E., REED, S.M., KEENE, R.O., HOWE, D.K., MORROW, J., & WORKMAN, J.D., 2017. Testing the Sarcocystis neurona vaccine using an equine protozoal myeloencephalitis challenge model. Veterinary Parasitology, 247, 37-41.
- SAVILLE, W. J. A., DUBEY, J.P., OGLESBEE, M.J., SOFALY, C.D., MARSH, A.E., ELITSUR, E., VIANNA, M.C., LINDSAY, D.S., & REED, S.M., 2004. Experimental infection of ponies with Sarcocystis fayeri and differentiation from Sarcocystis neurona infections in horses. Journal of Parasitology, 90, 1487-1491.
- SAVILLE, W. J. A., REED, S.M., DUBEY, J.P., GRANSTROM, D.E., MORLEY, P.S., HINCHCLIFF, K.W., KOHN, C.W., WITTUM, T.E., & WORKMAN, J.D., 2017. Interobserver variation in the diagnosis of neurologic abnormalities in the horse. Journal of Veterinary Internal Medicine, 31, 1871-1876.
- SAVILLE, W. J. A., SOFALY, C.D., REED, S.M., DUBEY, J.P., OGLESBEE, M.J., LACOMBE, V.A., KEENE, R.O., GUGISBERG, K.M., SWENSEN, S.W., SHIPLEY, R.D., CHIANG, Y.W., CHU, H.J., & NG, T., 2004. An equine protozoal myeloencephalitis challenge model testing a second transport after inoculation with Sarcocystis neurona sporocysts. Journal of Parasitology, 90, 1406-1410.
- SELLON, D. C., KNOWLES, D.P., GREINER, E.C., LONG, M.T., HINES, M.T., HOCHSTATTER, T., TIBARY, A., & DAME, J.B., 2004. Infection of immunodeficient horses with Sarcocystis neurona does not result in neurologic disease. Clinical and Diagnostic Laboratory Immunology, 11, 1134-1139.
- SELLON, D. C. & DUBEY, J. P., 2007. Equine protozoal myeloencephalitis. In: SELLON, D.C., AND LONG, M.T. (eds). Elsevier Inc., St. Louis, USA. Equine Infectious Diseases, 453-464.
- SIMPSON, C.F. & MAYHEW, I.G., 1980. Evidence for Sarcocystis as the etiologic agent of equine protozoal myeloencephalitis. Journal of Protozoology, 27, 288-292.
- SOFALY, C. D., REED, S.M., GORDON, J.C., DUBEY, J.P., OGLESBEE, M.J., NJOKU, C.J., GROVER, D.L., & SAVILLE, W.J.A., 2002. Experimental induction of equine protozoan myeloencephalitis (EPM) in the horse: effect of Sarcocystis neurona sporocyst inoculation dose on the development of clinical neurologic disease. Journal of Parasitology, 88, 1164-1170.
- SPEER, C.A. & DUBEY, J.P., 2001. Ultrastructure of schizonts and merozoites of Sarcocystis neurona. Veterinary Parasitology, 95, 263-271.
- STANEK, J. F., DUBEY, J.P., OGLESBEE, M.J., REED, S.M., LINDSAY, D.S., CAPITINI, L.A., NJOKU, C.J., VITTITOW, K.L., & SAVILLE, W.J.A., 2002. Life cycle of Sarcocystis neurona in its natural intermediate host, the racoon, Procyon lotor. Journal of Parasitology, 88, 1151-1158.
- STOFFREGEN, D. A. & DUBEY, J. P., 1991. A Sarcocystis sp.-like protozoan and concurrent canine distemper virus infection associated with encephalitis in a raccoon (Procyon lotor). Journal of Wildlife Diseases, 27, 688-692.
- TANHAUSER, S. M., CHEADLE, M. A., MASSEY, E. T., MAYER, B. A., SCHROEDTER, D. E., DAME, J. B., GREINER, E. C. & MACKAY, R. J., 2001. The nine-banded armadillo (Dasypus novemcinctus) is naturally infected with Sarcocystis neurona. International Journal for Parasitology, 31, 325-329.
- THOMAS, N. J., DUBEY, J. P., LINDSAY, D. S., COLE, R. A. & METEYER, C. U., 2007. Protozoal meningoencephalitis in sea otters (Enhydra lutris): a histopathological and immunohistochemical study of naturally occurring cases. Journal of Comparative Pathology, 137, 102-121.
- THULIN, J. D., GRANSTROM, D. E., GELBERG, H. B., MORTON, D. G., FRENCH, R. A. & GILES, R. C., 1992. Concurrent protozoal encephalitis and canine distemper virus infection in a raccoon (Procyon lotor). The Veterinary Record, 130, 162-164.
- TILLOTSON, K., MCCUE, P. M., GRANSTROM, D. E., DARGATZ, D. A., SMITH, M. O. & TRAUB-DARGATZ, J. L., 1999. Seroprevalence of antibodies to Sarcocystis neurona in horses residing in northern Colorado. Journal of Equine Veterinary Science, 19, 122-126.
- TRAVER, D. S., COFFMAN, J. R., MOORE, J. N. & NELSON, S. L., 1978. Protozoal myeloencephalitis in sibling horses. Journal of Equine Medicine and Surgery, 2, 425-428.
- TURAY, H. O., BARR, B. C., CALDWELL, A., BRANSON, K. R., COCKRELL, M. K. R. & MARSH, A. E., 2002. Sarcocystis neurona reacting antibodies in Missouri feral domestic cats (Felis domesticus) and their role as intermediate host. Parasitology Research, 88, 38-43.
- VARDELEON, D., MARSH, A. E., THORNE, J. G., LOCH, W., YOUNG, R. & JOHNSON, P. J., 2001. Prevalence of Neospora hughesi and Sarcocystis neurona antibodies in horses from various geographical locations. Veterinary Parasitology, 95, 273-282.
- VASHISHT, K., LICHTENSTEIGER, C. A., MILLER, L. A., GONDIM, L. F. P. & MCALLISTER, M. M., 2005. Naturally occurring Sarcocystis neurona-like infection in a dog with myositis. Veterinary Parasitology, 133, 19-25.
- VERMA, S. K., CALERO-BERNAL, R., LOVALLO, M. J., SWEENY, A. R., GRIGG, M. E. & DUBEY, J. P., 2015. Detection of Sarcocystis spp. infection in bobcats (Lynx rufus). Veterinary Parasitology, 212, 422-426.
- WENDTE, J. M., MILLER, M. A., LAMBOURN, D. M., MAGARGAL, S. L., JESSUP, D. A. & GRIGG, M. E., 2010. Self-mating in the definitive host potentiates clonal outbreaks of the apicomplexan parasites Sarcocystis neurona and Toxoplasma gondii. Plos Genetics, 6, e1001261.
- WHITE, C. L., LANKAU, E. W., LYNCH, D., KNOWLES, S., SCHULER, K. L., DUBEY, J. P., SHEARN-BOCHSLER, V. I., ISIDORO-AYZA, M. & THOMAS, N. J., 2018. Mortality trends in northern sea otters (Enhydra lutris kenyoni) collected from the coasts of Washington and Oregon, USA (2002-15). Journal of Wildlife Diseases, 54, 238-247.
- YEARGAN, M. R., ALVARADO-ESQUIVEL, C., DUBEY, J. P. & HOWE, D. K., 2013. Prevalence of antibodies to Sarcocystis neurona and Neospora hughesi in horses from Mexico. Parasite, 20, 29.
- YEARGAN, M. R. & HOWE, D. K., 2011. Improved detection of equine antibodies against Sarcocystis neurona using polyvalent ELISAs based on the parasite SnSAG surface antigens. Veterinary Parasitology, 176, 16-22.