- Infectious Diseases of Livestock
- Part 1
- Ovine babesiosis
- Vectors: Ticks
- Vectors: Tsetse flies
- Vectors: Muscidae
- Vectors: Tabanidae
- Vectors: Culicoides spp.
- Vectors: Mosquitoes
- Classification, epidemiology and control of arthropod-borne viruses
- Special factors affecting the control of livestock diseases in sub-Saharan Africa
- The control of infectious diseases of livestock: Making appropriate decisions in different epidemiological and socioeconomic conditions
- Infectious diseases of animals in sub-Saharan Africa: The wildlife⁄livestock interface
- Vaccination: An approach to the control of infectious diseases
- African animal trypanosomoses
- Dourine
- Trichomonosis
- Amoebic infections
- GENERAL INTRODUCTION: COCCIDIA
- Coccidiosis
- Cryptosporidiosis
- Toxoplasmosis
- Besnoitiosis
- Sarcocystosis
- Balantidiosis
- Leishmaniosis
- Neosporosis
- Equine protozoal myeloencephalitis
- GENERAL INTRODUCTION: BABESIOSES
- Bovine babesiosis
- Equine piroplasmosis
- Porcine babesiosis
- Ovine babesiosis
- GENERAL INTRODUCTION: THEILERIOSES OF CATTLE
- East Coast fever
- Corridor disease
- Zimbabwe theileriosis
- Turning sickness
- Theileria taurotragi infection
- Theileria mutans infection
- Theileria annulata theileriosis
- Theileriosis of sheep and goats
- Theileria buffeli⁄orientalis infection
- Non-pathogenic Theileria species in cattle
- GENERAL INTRODUCTION: RICKETTSIAL, CHLAMYDIAL AND HAEMOTROPIC MYCOPLASMAL DISEASES
- Heartwater
- Lesser known rickettsial infections in animals and humans
- Chlamydiosis
- Q fever
- Eperythrozoonosis
- Bovine Haemobartonellosis
- Potomac horse fever
- GENERAL INTRODUCTION: ANAPLASMOSES
- Bovine anaplasmosis
- Ovine and caprine anaplasmosis
Ovine babesiosis
This content is distributed under the following licence: Attribution-NonCommercial CC BY-NC  View Creative Commons Licence details here
View Creative Commons Licence details here

Ovine babesiosis
Previous authors: I YERUHAM AND A HADANI
Current authors:
D T DE WAAL - Associate Professor, BVSc, PhD, DipDatMet, HDipUTL, DipEVPC, MRCVS, School of Veterinary Medicine, University College Dublin, Room 034, UCD Veterinary Sciences Centre, Belfield, Dublin, D04 W6F6, Ireland
Introduction
Ovine babesiosis is a tick-borne disease caused by the intraerythrocytic protozoan parasites Babesia ovis10, Babesia motasi70 and several uncharacterized Babesia spp.12, 33, 36, 59 The acute form of the disease is characterized by fever, progressive anaemia, icterus and bilirubinaemia. Haemoglobinuria is rare but may occur in the late stage of the disease. Pregnant animals may abort. Susceptible flocks may suffer a high rate of mortality. Recovered animals that are latently infected are usually immune for a certain period. There is no cross-immunity between the parasites.
Ovine babesiosis caused by B. ovis is of considerable economic importance in the southern part of the Palaearctic zone (Mediterranean basin, Balkans, Kazakhstan, Kirghizstan, Turkmenistan, Iraq and Iran) overlapping the distribution area of the vector tick, Rhipicephalus bursa.16 The presence of B. ovis in sheep in Madagascar97 is questionable.
Rhipicephalus bursa is widely distributed between the 31 and 45° parallels north, including the Mediterranean basin,45, 47, 55, 58, 87, 110 the Balkans,24, 69, 75 southern former USSR,81 Iraq85 and Iran.48 It has not been found in the southern hemisphere.
The geographical distribution of B. motasi is very wide and includes western and northern Europe, the Mediterranean basin, Asia (Turkey, India, China) and Africa (Somalia, Nigeria, Madagascar and southern Africa) and is transmitted mainly by Haemaphysalis spp. ticks. Babesia motasi was first reported in southern Africa in 1902,49 and subsequently ovine babesiosis was diagnosed in other countries in the African continent (Somalia, Nigeria, Madagascar).20, 26, 56, 63, 95, 97 Since the report of Hutcheon and Robertson in 1902,49 there are no other records on the occurrence of ovine babesiosis in southern Africa. Babesia motasi causes a severe disease and mortality in sheep in Asia,13, 50 Africa20 and China.12 It is regarded as a benign parasite in western Europe,21, 57, 82, 98 but occasionally causes anaemia in lambs.17
Babesiosis caused by B. ovis and B. motasi affects all breeds of sheep and, more rarely, goats, with B. ovis being the more pathogenic. It appears that some isolates of B. motasi are infective for sheep and goats while others only infect sheep.6
More recently several large Babesia strains (Babesia sp. BQ1 (Lintan), Babesia
sp. BQ1 (Ningxian), Babesia sp. Tianzhu, Babesia sp. Hebei, Babesia sp. Madang, Babesia sp. Liaoning and Babesia sp. Xinjiang) have been isolated in China with variable degree of virulence to sheep and goats.34 One isolate in particular Babesia sp. Xinjiang is morphologically very different from B. motasi and B. ovis.33 Based on the 18S rRNA gene sequences these Babesia isolates can be broadly divided into two groups – Babesia motasi-like; and a second group, consisting of Babesia sp. Xinjiang, that falls between B. ovis and B. orientalis.33
Three other Babesia species have been reported to occur in sheep—B. sergenti,102 B. foliata84 and B. crassa42 (in Algeria, India and Iran respectively). These have a restricted local importance and their validity is still debated.
Aetiology and life cycle
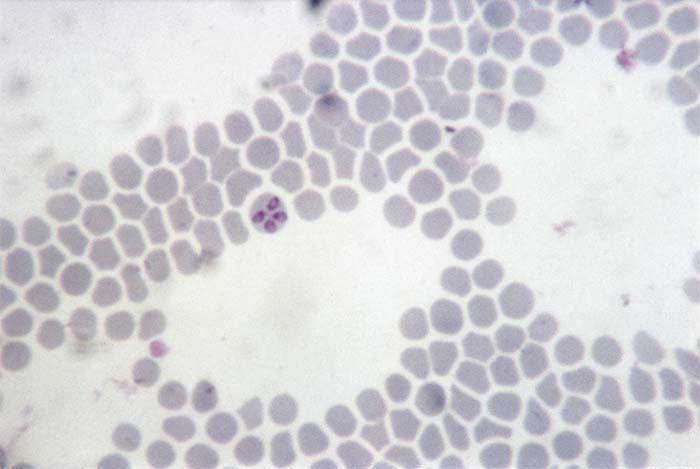
Babesia ovis is a small parasite of 1,0 to 2,5 μm in length. The majority of parasites are oval or pyriform, tending to occur at the periphery of the erythrocytes.40, 93, 106 The angle between the paired pyriform parasites is obtuse. Cross-forms are uncommon112 (Figure 1). Babesia crassa, described in Iran,42 has a similar morphology.
Babesia motasi is a relatively large parasite measuring 2,2 to 3,8 μm in length by about 2,0 μm in width. It commonly appears as a pair of pear-shaped organisms, separated by an acute angle, occupying two-thirds or more of the affected erythrocyte.5, 98 Ring, oval, elongated and budding forms are less common. Babesia motasi often possesses a double nucleus, which is invariably single in B. ovis. The intraerythrocytic stages include single tropozoites, dividing forms and paired merozoites. The fine structure of the tropozoites and merozoites are apparently similar to those described for other species of Babesia.28
Babesia sp. Xinjiang is also large approx. 2.42 (±0.35) µm in length and 1.06 (±0.22) µm wide and typically occurs in most cases as single or paired pirifom bodies at an acute angle within erythrocytes. A variety of other morphological forms has also been found, including ring, trifoliate, rod- shaped and oval.33
Rhipicephalus bursa, a two-host tick,107 is the only known vector of B. ovis.106 Haemaphysalis punctata, a three-host tick, is the only proven vector of B. motasi in Europe5 while H. bispinosa and H. intermedia are the only proven ones in India.3, 50 In China the large Babesia isolates are transmitted by a number of three-host ticks including H. longicornis, Hyalomma anatolicum anatolicum and H. qinghaiensis33, 34, 36
Markov (cited by Erashov23 and Warnecke101) reported on the transmission of B. motasi by Haemaphysalis otophila, a three-host tick. The possibility that R. bursa may be a vector of B. motasi is extremely doubtful.98 Opasina74 suggested that Amblyomma variegatum, a three-host tick, is a vector of B. motasi in goats in Nigeria.
Babesia ovis, B. motasi and the Chinese Babesia isolates all develop only in erythrocytes and multiply by binary fission into merozoites (merogony).
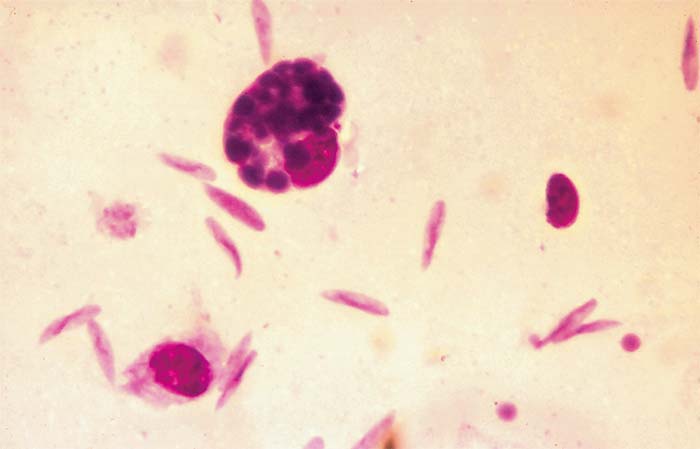
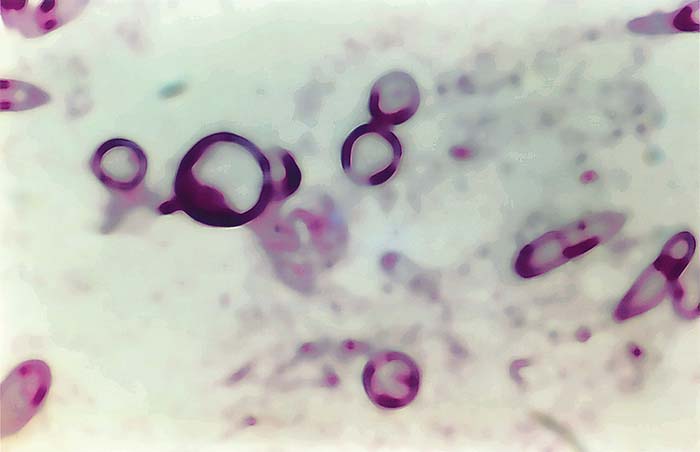
The life cycle of B. ovis in the vector R. bursa has been thoroughly studied2, 14, 27, 29, 67, 68 and consists of gametogony (sexual cycle) in the gut of engorged female ticks, which results in the formation of ookinetes (zygotes). A primary schizogony (= sporogony) produces sporokinetes. A number of schizogonic cycles occur in the gut and various tissues. Schizonts have been observed in the haemolymph, eggs and salivary glands. During the final schizogony in the salivary glands, the infective forms (merozoites) are produced. Young kinetes of B. ovis in haemolymph and eggs of R. bursa are round in shape, but subsequently become elongated and cigar-shaped (Figures 2 and 3).
Adult R. bursa ticks become infected with B. ovis during a blood meal. Transmission to a susceptible host occurs by all instars of the succeeding generation.106 Rhipicephalus bursa can be infected with B. ovis both alimentarily and vertically. Alimentary transmission gives rise to a higher rate and degree of tick infection than that following vertical transmission. Eggs infected with B. ovis appear earlier in horizontally (alimentary) infected ticks than in vertically infected ones.108
Infection rates in ticks can be estimated by examining their haemolymph 7 to 12 days after their detachment, and those of eggs 5 to 10 days after the commencement of oviposition.104 In the salivary glands, infective particles can be observed and are transmitted to the host during a blood meal.
Babesia ovis infection in sheep has been observed 11 to 12 days post-infestation by the tick vector.64 It is readily transmitted by inoculation of infected blood.105 Successive serial passages of infected blood in splenectomized lambs result in a decrease in the degree of parasitaemia and apparent attenuation of the parasite; this feature has been employed in the production of a blood-based vaccine.96, 99, 105 Intra-uterine infection of the foetus in pregnant sheep by B. ovis also occurs.92
Transmission of B. ovis by R. bursa109 and of B. motasi by H. punctata is transovarial as well as transstadial.5, 14, 106 All instars of H. punctata are capable of transmitting B. motasi parasites.5 Little is know about the Chinese Babesia spp. and the transmission of them have only been studied in a few instances. Transmission is transovarial in H. longicornis, Hy. a. anatolicum and H. qinghaiensis with larvae, nymphs and adults being able to transmit the parasite to sheep, although, the ability of adult H. longicornis to transmit Babesia still needs to be confirmed.12, 33, 36 The life cycle of Babesia sp. Xinjiang in its vector Hy. a. anatolicum has also been studied.33
Epidemiology
The larvae, nymphs and adults of R. bursa, a two-host tick, parasitize the same animal species, and their natural hosts are wild sheep and goats.48 This tick is one of the most important species in endemically infected countries (Cyprus, Syria, Israel, the Balkans, Turkey)1, 8, 51, 55, 75, 77, 105, 110 and favours a habitat characterized by rendzinas and terra rossa soils, low average precipitation and a long, dry summer.46 It has a strictly seasonal life cycle with one generation annually. Larvae are found on the pasture, and larvae and nymphs on the animals during the winter period (October to February) in the northern hemisphere, with a peak prevalence in November to December. The adult ticks first appear in April, their numbers reaching a peak in May, and persist in the field and on the animals until July.111
There is a striking seasonal incidence of ovine babesiosis caused by B. ovis. The disease occurs annually in R. bursa infested areas during the activity period of the adult ticks.106 The prevalence of babesiosis reaches a peak in May, corresponding to a peak in the infestation rate of R. bursa on the animals. It thereafter decreases and, by the end of July, disappears.1, 11, 51, 58, 69, 75, 89, 106 Ovine babesiosis occurs two weeks after the first appearance of the R. bursa adult ticks on pasture.113 The disease may reappear annually in the same flocks in endemic areas. This epidemiological pattern differs from the state of enzootic stability known to develop in bovine babesiosis.62 Sporadic clinical cases of ovine babesiosis may occur outside the main season of adult R. bursa activity, concurrently with the rare appearance of few adult ticks.89 Relapses of chronic latent cases due to stress factors, such as heat, food shortage and helminthosis, may also occur in the autumn months.
The incidence of babesiosis in 6- to 12-month-old sheep in their first grazing season may be almost double (10 per cent) that of ewes (5,1 per cent).83, 106 The average rate of parasitaemia in lambs and hoggets (7,5 per cent) may be somewhat higher than in ewes (5,6 per cent).112 These rates are rather low when compared to those recorded in endemic areas of bovine babesiosis.18, 80
It appears that a certain percentage of sheep are not exposed annually to infection and consequently remain susceptible. Endemic stability in bovine babesiosis depends on regular exposure to the parasite, and is influenced by climatic conditions, animal management, and dipping. It appears that endemic stability in ovine babesiosis in R. bursa-infested areas is rarely established.
In general, unstable endemic situations accompanied by outbreaks of ovine babesiosis depend to a large extent on four factors: transhumance (animals are moved to endemic areas), health management (tick control and chemotherapy), especially unfavourable climatic conditions, and chemoprophylaxis. Intensive tick control should be avoided to maintain endemic stability.91
Recovered animals remain patent or latent carriers of B. ovis for varying periods. The parasites persist in the blood of infected sheep for two years37 and ticks feeding on the animals during this period may become infected. The presence of parasites in the circulating blood maintains a state of premunity.88
High annual rates of seroconversion have been observed in both hoggets and ewes (84,5 per cent and 89 per cent, respectively). The ‘autumn-winter rise’ in serological reactions observed in young animals might be caused by larval and nymphal infestations.106
In young animals, immunity to babesiosis may be conferred by colostral antibodies and exposure to immature and/or adult R. bursa ticks. Morbidity in flocks grazing in endemic areas can be attributed to the fact that only 75 per cent of the colostrum samples of the hoggets and 67,5 per cent of those of the ewes may contain maternal antibodies to B. ovis, and these may persist for only three months in lambs,37, 113 as compared to the longer period of resistance known in B. bovis in cattle.86 It therefore appears that lambs must be exposed to B. ovis infection up to the age of three months in order to maintain an immune state. Lambs infested with R. bursa larvae may develop antibody titres after a relatively long post-infestation period (39 days on average) and may suffer from only mild clinical reactions when subsequently challenged with infected R. bursa adult ticks.113 The delay in the serological reaction and the fact that only 85 per cent of the lambs may become serologically positive are probably due to the low B. ovis infection rate of the preimaginal stages of the tick.109
The relationship between antibody levels and the degree of immunity to babesiosis is not clear. Serologically positive animals, when challenged, may develop clinical babesiosis. A similar situation has been reported in bovine babesiosis.15, 31 In addition to the climatic and husbandry factors mentioned above, the precarious enzootic stability in ovine babesiosis caused by B. ovis might be due to the fact that the active period of R. bursa adults is short, resulting in low levels of colostral antibodies, as well as the short lifespan of these antibodies in lambs. The transmission of B. ovis by the pre-imaginal stages during the autumn and winter months, causing seroconversion, might play a major role in pre-immunizing and boosting the immunity in sheep against infections which occur during the spring to summer months.106
Ovine babesiosis caused by B. motasi has not been studied in detail. It has been suggested that Nigerian sheep are resistant to infection with B. motasi.56 Furthermore, certain isolates of B. motasi might be of lower pathogenicity, while others are known to cause severe disease and even death in sheep in India50 and in Somalia.20 It has been proposed that the virulence of different isolates of B. motasi and the severity of the disease might be greatly affected by the tick vector.20, 50 Although B. motasi may be a significant pathogen on its own, it seems more probable that disease in certain regions may be complicated by other tick-borne pathogens, such as Theileria spp., Ehrlichia spp.,60 and Nairobi sheep disease virus.20
Over the last three decades at least seven large Babesia spp. have been isolates from sheep in China. Based on 18S rRNA gene phylogeny it has been shown that six of these isolates (Babesia sp. BQ1 (Lintan), Babesia sp. BQ1 (Ningxian), Babesia sp. (Tianzhu), Babesia sp. (Hebei), Babesia sp. (Madang) and Babesia sp. (Liaoning)) all had a 88-98 per cent identity to the European isolates of B. motasi 59 and referred to as Babesia motasi-like. Although it is still debated if these represent one or more subspecies. The seventh isolate, Babesia sp. Xinjiang, is morphologically different, less pathogenic, less than 82 per cent similarity with other ovine Babesia spp. and forms a sister clade with B. ovis, B. bovis, B. orientalis and B. occultans 53
From recent large scale sero-surveys in China it would appear that these Babesia spp. are widespread throughout China with seroprevalence ranging between 30 – 44 per cent32, 71, 100
In goats, clinical babesiosis cases are rare.78 It seems that the local breeds possess a certain degree of natural resistance to babesiosis.
In the absence of non-ovine susceptible hosts, the tick might serve as a reservoir of B. ovis.65 Babesia ovis and B. motasi have been reported to occur in unusual hosts22 and ticks9, 73 but the role of these, as asymptomatic carriers or true vectors, is still unknown.
Pathogenesis
Upon infection of a susceptible sheep with B. ovis or B. motasi, multiplication of the organisms occurs in erythrocytes with the development of a clinically detectable anaemia which results in hypoxia of tissues that may cause the death of the host. Organs, particularly those with high metabolic activity, such as the liver and kidneys,44 are the most severely affected in ovine babesiosis.39, 41, 55, 94, 104
Severe anaemia may occur in sheep harbouring a low parasitaemia, which implies that additional factors are involved in its pathogenesis. Erythrophagocytosis of both parasitized and non-parasitized erythrocytes has been observed in cases of B. ovis infection. A similar finding has been recorded in infections with other species of Babesia.44, 61
The anaemia following infections with Babesia spp. is considered to be caused by:
- mechanical damage of red blood cells by invading organisms;
- autoimmune phenomena; and
- adsorption of antigens onto normal erythrocytes followed by the formation of immune complexes and the activation of the classical complement pathway resulting in their destruction.6
The pathogenic mechanisms of B. ovis are broadly similar to those of B. bovis, provoking lesions characteristic of a hypovolaemic and endotoxic shock process. The latter is probably the result of a massive release of catabolites from the lysis of erythrocytes and necrosis of tissues.39 The lesions observed generally arise from vascular alterations, such as vasodilation and vascular stasis, leading to hypoxia of the tissues.39 Apart from the changes in the brain, other findings are similar to those reported in B. bovis infection of cattle.103
Clinical signs
Ovine babesiosis, in general, is an acute disease particularly in susceptible animals. Morbidity and mortality due to ovine babesiosis in flocks in endemic areas occur annually. Babesia ovis infections may cause 10 per cent morbidity with 2 per cent mortality,110 but a mortality rate as high as 25 per cent has been reported.83
The clinical signs of ovine babesiosis caused by either B. ovis or B. motasi are similar. In general, the signs observed are typical of those due to an anaemia. An incubation period, generally ranging from seven to nine days, is followed by a febrile period of several days during which inappetence and depression are evident. The febrile response in babesiosis caused by B. ovis precedes a detectable parasitaemia by at least 24 hours.109
Initially, visible mucous membranes are reddened and the urine is normal in colour, but as the disease progresses visible mucous membranes become pale and icteric. Haemoglobinuria is uncommon. Constipation may occur, and faecal pellets may contain blood and be covered by mucus. Respiration and pulse rates are increased. Weakness and prostration develop together with muscular tremors, lachrymation and salivation. The parasitaemia in B. ovis infections is usually low (less than 1 per cent).109
Death may occur at any time after the onset of the clinical signs depending on the virulence of the Babesia strain and the susceptibility of the animal.
Pathology
Infection with either B. ovis or B. motasi leads to a macrocytic hypochromic anaemia which coincides with the peak of parasitaemia. Reticulocytosis, poikilocytosis and anisocytosis, together with hypochromasia, indicate that haemopoetic centres attempt to compensate for the anaemia. There is an initial leukocytosis, largely due to neutrophilia,6 and increased total and conjugated bilirubin levels.
The clinico-pathological findings in babesiosis due to B. motasi infection are considerably milder than those observed in B. ovis infections.6
Affected animals show a decrease in the packed cell volume (PCV), which ranges between 30 and 40 per cent.109 There is an increase in plasma transaminases, particularly aspartate aminotransferase and alanine aminotransferase, dehydrogenase, blood urea nitrogen and creatine, and a decrease in sorbitol dehydrogenase, alkaline phosphatase, total proteins and albumin.38, 66 A proteinuria occurs in the course of the disease.38
In experimental B. ovis infection in sheep39, 109 evidence of anaemia, marked icterus, splenomegaly, a grossly enlarged dark-brown degenerated liver, and nephrosis are noted. Ecchymotic haemorrhages are present in the epicardium, endocardium and subserosa of the viscera.
Erythrocytes parasitized by B. ovis have not been observed to block capillaries in the brain,109 as recorded in B. bovis infections in cattle. This explains the absence of neurological signs in acute ovine babesiosis caused by B. ovis. A disseminated intravascular coagulation syndrome has been observed in sheep suffering from B. ovis infection.39, 109
Diagnosis
Acute ovine babesiosis can be confirmed by microscopic examination of stained blood smears made from capillary blood. The parasitaemia in B. ovis infection is, however, low. Diagnosis of ovine babesiosis at necropsy is possible if decomposition is not too advanced.
Babesia parasites deform within a few hours of death, thus making their morphological identification impossible. Thin blood smears from the peripheral circulation, and impression smears of the spleen, liver and kidneys should be made.
A variety of serological tests has been used to demonstrate the presence of antibodies to B. ovis and B. motasi.7, 19, 25, 37, 52, 57, 76, 79, 98, 109 Cross-reactions have recently been reported within the genus Babesia. A high correlation is found between B. motasi and B. crassa, and a lower one between these two and B. ovis.79 No cross-reactions have been found between B. ovis antigens and B. motasi, B. crassa or Theileria ovis.40 Serological tests are used mainly in epidemiological surveys. The indirect immunofluorescence test (IFAT) is one of the simplest, most sensitive and effective techniques used for this purpose. However, as in the case with other serological techniques, the specificity of the method is not always satisfactory.79 Recombinant ELISAs have been developed for the Chinese Babesia spp.32, 35, 71, 72, 100
Differential diagnosis
Ovine babesiosis should be differentiated from infections caused by other tick-borne blood parasites such as Anaplasma ovis, Theileria ovis, Anaplasma phagocytophilum and Mycoplasma ovis. Copper poisoning resulting in an acute haemolytic crisis with icterus, should also be considered.
Control
The control of babesiosis in sheep is similar to that used for the control of bovine babesiosis (see Bovine babesiosis) and is based on a combination of strategic tick control, vaccination, chemoprophylaxis, when necessary, and chemotherapy.
Immunization can be achieved by inoculation of a whole blood vaccine containing attenuated parasites.54, 96, 99 Attenuation is achieved by serial passage of the parasites in splenectomized lambs. A crude extract of B. ovis parasites in Freund’s complete adjuvant has been also used as a vaccine.4 There is, however, no ovine babesiosis vaccine available commercially. An effective method for the control of the disease in endemic areas might be the exposure of sheep to immature stages of R. bursa in October to February before the main adult tick period in April to July.
Animals suffering from clinical babesiosis should be treated with the babesiacidal drugs used in bovine babesiosis. Diminazene aceturate (Berenil®) is used at a dosage rate of 3 to 5 mg/kg body weight subcutaneously.90, 98 Diminazene can also be used in the chemoprophylaxis of ovine babesiosis with a withdrawal period as recommended by the manufacturers.30 Imidocarb dipropionate (Imizol®) is used at a dosage rate of 1,2 mg/kg body weight intramuscularly. A second treatment may be necessary in case of recrudescence of the infection.43 Amicarbalide isethionate (Pirodia®, Diampron ®) at 5 mg/kg body weight subcutaneously has also been used with success.98
References
- ABO-SHEHADA, M. N., MUWALLA, M. M. & TAWFEEK, F., 1988. Ovine babesiosis in Jordanian sheep mistakenly diagnosed as plant poisoning. Preventive Veterinary Medicine, 6, 235-238.
- ABRAMOV, I. V., DYAKONOV, L. P. & STEPANOVA, N. I., 1973. The ultrastructure and development of Babesia ovis in ticks and in the warm-blooded host. Progress in Protozoology. Abstract of papers read at the 4th International Congress of Protozoology, Clermont-Ferrand, September 2-9.
- ACHAR, S. D. & SRIKANTIAH, G. N., 1934. A note on Babesia motasi, Wenyon (1926) in sheep in Nysore state. Indian Veterinary Journal, 10, 270-273.
- ALABAY, M., DUZGUN, A., CERCI, H., WRIGHT, I. G., WALTISBUHL, D. J. & GOODGER, B. V., 1987. Ovine babesiosis: Induction of a protective immune response with crude extracts of either Babesia bovis or B. ovis. Research in Veterinary Science, 43, 401-402.
- ALANI, A. J. & HERBERT, I. V., 1988. The morphometrics of Babesia motasi (Wales) and its transmission by Haemaphysalis punctata (Canestrini and Fanzago, 1877) to sheep. Veterinary Parasitology, 30, 87-95.
- ALANI, A. J. & HERBERT, I. V., 1988. The pathology of Babesia motasi (Wales) infection in sheep. Veterinary Parasitology, 27, 209-220.
- ALANI, A. J., KONRAD, J. & HERBERT, I. V., 1987. Serodiagnosis of Babesia motasi (Wales), Theileria recondita (Wales) and Cytoecetes phagocytophila infection in sheep. Research in Veterinary Science, 43, 104-108.
- ANGELOVSKI, T., PETROVIC, Z. & TOMOCOVA, D., 1963. Piroplasmosis in sheep in SR Macedonia. Veterinarski Glasnik, 17, 861-867.
- AYDIN, M. F., AKTAS, M. & DUMANLI, N., 2015. Molecular identification of Theileria and Babesia in ticks collected from sheep and goats in the Black Sea region of Turkey. Parasitology Research, 114, 65-69.
- BABES, V., 1892. L’etiologie d’une enzootie des moutons, denomm’ee carceag en Roumanie. Comptes Rendus. Academie des Sciences (Paris), 115, 359.
- BADESCU, C., POPOVICI, I. & MIHI, S., 1968. Contribution to the study of the ecology of the ixodids (six species) of the Mihai Brava pasture of the district of Giurgiu (Rumania). Lucrarile Sesiunii Stiintifice, Institutul Agronomic Nicolae Balcescu (C), 11, 311-325.
- BAI, Q., LIU, G., LIU, D., REN, J. & LI, X., 2002. Isolation and preliminary characterization of a large Babesia sp. from sheep and goats in the eastern part of Gansu Province, China. Parasitology Research, 88, S16-S21.
- BHASKARA RAO, P., SURENDRAN, N. S. & O., R. R. A., 1989. Study on outbreaks of babesiosis in sheep in Andhra Pradesh. Indian Veterinary Journal, 66, 348-351.
- BUSCHER, G., FRIEDHOFF, K. T. & EL-ALLAWY, T. A. A., 1988. Quantitative description of the development of Babesia ovis in Rhipicephalus bursa (haemolymph, ovary, eggs). Parasitology Research, 74, 331-339.
- CALLOW, L. L., MCGREGOR, W., PARKER, R. J. & DALGLIESH, R. J., 1974. The immunity of cattle to Babesia argentina after drug sterilization of infections of varying duration. Australian Veterinary Journal, 50, 6-11.
- CANESTRINI, G. & FANZAGO, F., 1877. Intornoagli acari Italiani. Atti Istituto Veneto di Scienze, Lettere, ed Arti, 4, 69-208.
- CHRISTENSSON, D. & THUNEGARD, E., 1981. Babesia motasi in sheep on the island of Gotland in Sweden. Veterinary Parasitology, 9, 99-106.
- CURNOW, J. A., 1973. Studies on the epizootiology of bovine babesiosis in northeastern New South Wales. Australian Veterinary Journal, 49, 284-297.
- DUZGUN, A., WRIGHT, I. G., WALTISBUHL, D. J., GALE, K. R., GOODGER, B. V., DARGIE, J. D., ALABAY, M. & CERCI, H., 1991. An ELISA for the diagnosis of Babesia ovis infection utilizing a synthetic, Babesia bovis derived antigen. Veterinary Parasitology, 39, 225-231.
- EDELSTEN, R. M., 1975. The distribution and prevalence of Nairobi sheep disease and other tick-borne infections of sheep and goats in Northern Somalia. Tropical Animal Health and Prodution, 7, 29-34.
- ENIGK, K., 1956. Die Schafpiroplasmose in Deutschland. Deutsche Tierärztliche Wochenschrift, 63, 161-162.
- ENIGK, K., FRIEDHOFF, K. T. & WIRAHADIREDJA, S., 1964. Zur Wirtsspezifitat von Babesia motasi und Babesia ovis (Piroplasmidea). Zeitschrift für Parasitenkunde, 24, 309-318.
- ERASHOV, V. S., 1956. Parasitology and Parasitic Diseases of Livestock. Israel Program for Scientific Translations, 1960, (translation of original Russian edition of 1956).
- FEIDER, Z., RAUCHBACH, C. & MIRONESCU, I., 1958. The ticks of Rumania. Ceskoslovenska Parasitologie, 5, 71-87.
- FERRER, D., CASTELLA, J. & GUTIERREZ, J. F., 1998. Seroprevalence of Babesia ovis in sheep in Catalonia, northeastern Spain. Veterinary Parasitology, 79, 275-281.
- FOLKERS, C. & KUIL, H., 1967. Blood parasites in cattle, sheep and goats in northern Nigeria. Bulletin of Epizootiology Diseases in Africa, 15, 121-123.
- FRIEDHOFF, K. T., 1969. Lichtmikroskopische Untersuchungen über die Entwicklung von Babesia ovis (Piroplasmidea) in Rhipicephalus bursa (Ixodoidea). I. Die Entwicklung in Weiblichen Zecken nach der Repletion. Zeitschrift für Parasitenkunde, 32, 191-219.
- FRIEDHOFF, K. T., 1981. Morphologic aspects of Babesia in the tick. In: RISTIC, M. & KREIER, J. P., (eds). Babesiosis. New York: Academic Press.
- FRIEDHOFF, K. T. & SMITH, R. D., 1981. Transmission of Babesia by tick. In: RISTIC, M. & KREIER, J. P., (eds). Babesiosis. New York: Academic Press.
- GEORGESCU, L. & TEUSTEA, V., 1966. Prevention of Babesiella ovis and Babesia motasi infections in sheep by injection of Berenil. Lucrarile Institutului de Cercetari Veterinare si Biopreparate Pasteur, 5, 427-438.
- GOLDMAN, M. & ROSENBERG, A. S., 1974. Immunofluorescence studies of the small Babesia species of cattle from different geographical areas. Research in Veterinary Science, 16, 351-354.
- GUAN, G., MA, M., LIU, A., REN, Q., WANG, J., YANG, J., LI, A., LIU, Z., DU, P., LI, Y., LIU, Q., ZHU, H., YIN, H. & LUO, J., 2012a. A recently identified ovine Babesia in China: serology and sero-epidemiology. Parasitology International, 61, 532-537.
- GUAN, G., MA, M., MOREAU, E., LIU, J., LU, B., BAI, Q., LUO, J., JORGENSEN, W., CHAUVIN, A. & YIN, H., 2009. A new ovine Babesia species transmitted by Hyalomma anatolicum anatolicum. Experimental Parasitology, 122, 261-267.
- GUAN, G., MOREAU, E., LIU, J., HAO, X., MA, M., LUO, J., CHAUVIN, A. & YIN, H., 2010. Babesia sp. BQ1 (Lintan): molecular evidence of experimental transmission to sheep by Haemaphysalis qinghaiensis and Haemaphysalis longicornis. Parasitology International, 59, 265-267.
- GUAN, G., MOREAU, E., LIU, J., MA, M., ROGNIAUX, H., LIU, A., NIU, Q., LI, Y., REN, Q., LUO, J., CHAUVIN, A. & YIN, H., 2012b. BQP35 is a novel member of the intrinsically unstructured protein (IUP) family which is a potential antigen for the sero-diagnosis of Babesia sp. BQ1 (Lintan) infection. Veterinary Parasitology, 187, 421-430.
- GUAN, G. Q., YIN, H., LUO, J. X., LU, W. S., ZHANG, Q. C., GAO, Y. L. & LU, B. Y., 2002. Transmission of Babesia sp to sheep with field-collected Haemaphysalis qinghaiensis. Parasitology Research, 88, S22-S24.
- HABELA, M. A., REINA, D. & NAVARRETE, I., 1990a. Antibody response and duration of latent infection in sheep following experimental infection with Babesia ovis. Veterinary Parasitology, 35, 1-10.
- HABELA, M. A., REINA, D., NAVARRETE, I. & HERNANDEZ RODRIGUEZ, S., 1987. Alterations of the renal function in the ovine experimental babesiosis. Third Mediterranean Conference on Parasitology, 24-27 August, Jerusalem, Israel.
- HABELA, M. A., REINA, D., NAVARRETE, I., REDONDO, E. & HERNANDEZ, S., 1990. Histopathological changes in sheep experimentally infected with Babesia ovis. Veterinary Parasitology, 38, 1-12.
- HABELA, M. A., REINA, D., NIETO, C. & NAVARRETE, I., 1990b. Isolation and identification of Babesia ovis in Extremadura (Spain). Veterinary Parasitology, 35, 233-238.
- HALACHEVA, M. & PAULOV, W., 1977. Histopathological changes in splenectomized sheep infected with Babesia ovis. Veterinary Science, 14, 50-56.
- HASHEMI-FESHARKI, F. & UILENBERG, G., 1981. Babesia crassa n.sp. (Sporozoa, Babesiidae) of domestic sheep in Iran. The Veterinary Quarterly, 3, 1-8.
- HASHEMI-FESHARKI, R., 1991. Ovine and caprine babesiosis in Iran: treatment with imidocarb. Veterinary Record, 129, 383-384.
- HILDEBRANDT, P. K., 1981. The organ and vascular pathology of babesiosis. In: RISTIC, M. & KREIER, J. P., (eds). Babesiosis. New York: Academic Press.
- HOFFMANN, G., HORCHNER, F., SCHEIN, E. & GERBER, H. C., 1971. Seasonal occurrence of ticks and piroplasms in domestic animals in the asiatic provinces of Turkey. Berliner und Münchner Tierärztliche Wochenschrift, 84, 152-156.
- HOOGSTRAAL, H., 1978. Biology ot ticks. In: WILDE J. K. H., (ed.). Tick-borne Diseases and their Vectors. Proceedings of an International Conference, University of Edinburgh, Centre for Tropical Veterinary Medicine, Eastern Bush, Roslin, Midlothian, UK.
- HOOGSTRAAL, H., 1979. The epidemiology of tick borne Crimean-Congo hemorrhagic fever in Asia, Europe and Africa. Journal of Medical Entomology, 15, 307-417.
- HOOGSTRAAL, H. & VALDEZ, R., 1980. Ticks (Ixodoidea) from wild sheep and goats in Iran and medical and veterinary implications. Fieldiana: Zoology, New Series, 1-16.
- HUTCHEON, D. & ROBERTSON, W., 1902. Malarial catarrhal fever of sheep. Veterinary Record, 14, 629.
- JAGANNATH, M. S., HEGDE, K. S., SHIVARAM, K. & NAGARAJA, K. V., 1974. An outbreak of babesiosis in sheep and goats and its control. Mysure Journal of Agricultural Sciences, 8, 441-443.
- KOHLER, G., HOFFMAN, G., JANITSCHKE, K. & WIESENHUTTER, E., 1967. Studies towards knowledge of the ticks found in Syria. Zeitschrift fürTropenmedizin und Parasitologie, 18, 375-381.
- KYURTOV, N., 1976. Use of the immunofluorescence reaction in the diagnosis of Babesia ovis infection. Veterinarno Meditsinski Nauki, 13, 61-66.
- LACK, J. B., REICHARD, M. V. & VAN DEN BUSSCHE, R. A., 2012. Phylogeny and evolution of the Piroplasmida as inferred from 18S rRNA sequences. International Journal for Parasitology, 42, 353-363.
- LAWRENCE, J. A., 1997. Conventional vaccines for tick-borne haemoparasitic diseases of sheep and goats. Parasitologia, 39, 119-121.
- LE RICHE, P. D., ALTAN, Y., CAMPBELL, J. B. & EFSTATHIOU, G. C., 1974. Ticks (Ixodoidea) of domestic animals in Cyprus. Bulletin of Entomological Research, 64, 53-63.
- LEEFLANG, P., 1977. Tick-borne diseases of domestic animals in northern Nigeria. I. Historical review 1923–1966. Tropical Animal Health and Production, 9, 147-152.
- LEWIS, D., HOLMAN, M. R., PURNELL, R. E. & YOUNG, E. R., 1981. Investigation on Babesia motasi isolated from Wales. Research in Veterinary Science, 31, 239-243.
- LIEBISCH, A. & ZUKARI, M., 1978. Biological and ecological studies on ticks of the genera Boophilus, Rhipicephalus and Hyalomma in Syria. In: WILDE, J.K.H., (ed.). Tick-borne Diseases and their Vectors. Proceeding of an International Conference, University of Edinburgh, Centre for Tropical Veterinary Medicine, Eastern Bush, Roslin, Midlothian, UK.
- LIU, A. H., YIN, H., GUAN, G. Q., SCHNITTGER, L., LIU, Z. J., MA, M. L., DANG, Z. S., LIU, J. L., REN, Q. Y., BAI, Q., AHMED, J. S. & LUO, J. X., 2007. At least two genetically distinct large Babesia species infective to sheep and goats in China. Veterinary Parasitology, 147, 246-251.
- MADHAVA, K. R. A., 1966. Some observations on Babesia motasi infection in sheep in Andhra Pradesh. Indian Veterinary Journal, 43, 785-789.
- MAEGRAITH, B., GILLES, H. M. & DEVAKUL, K., 1957. Pathological processes in Babesia canis infections. Zeitschrift für Tropenmedizin und Parasitologie, 8, 485-514.
- MAHONEY, D. F. & ROSS, D. R., 1972. The epizootiological factors in the control of bovine babesiosis. Australian Veterinary Journal, 48, 292–298.
- MAJARO, O. M. & DIPEOLU, O. O., 1986. Spontaneous infections of tick-borne blood parasites in splenectomized indigenous sheep and goats in Ibadan, Nigeria. Revue d’Elevage Medecine veterinaire Pays tropicax, 39, 189-191.
- MARIN, J., ROL, J. A., NAVARRETE, I. & HABELA, M. A., 1995. Aportaciones al conociniento epidemiologico de la babesiosis ovina Babesia ovis en la provincia de Badajoz. Seroprevalencia Y transmision experimental por ixodidos. Medicina Veterinaria, 12, 460-470.
- MARKOV, A. A. & ABRAMOV, I. V., 1970. Results of twenty years observations on repeated life cycles of Babesia ovis in 44 generations of Rhipicephalus bursa. Trudy Vsesoyznoge Instituta Eksperimental’noi Veterinarii, 38, 5–14 (Ref: Veterinary Bulletin, 1977, 42, 4).
- MILLIC, D., 1964. Ein Beitrag der Kenntnis der Veranderungen in Bult und den Geweben bei den an der Piroplasmose erkrankten Schafen. Acta Veterinaria, Beograd, 14, 227-238.
- MOLTMANN, U. G., MEHLHORN, H. & FRIEDHOFF, K. T., 1982. Electron microscopic study on the development of Babesia ovis (Piroplasmia) in the salivary glands of the vector tick Rhipicephalus bursa. Acta Tropica, 39, 29-40.
- MOLTMANN, U. G., MEHLHORN, H. & FRIEDHOFF, K. T., 1982. Ultrastructural study of the development of Babesia ovis (Piroplasmia) in the ovary of the vector tick Rhipicephalus bursa. Journal of Protozoology, 29, 30-38.
- MONOV, M., PETROV, D. & MILOUSHEV, I., 1997. The occurrence, species composition and seasonal activity of Ixodidae family ticks in northwest Bulgaria. Veterinarno Meditsinski Nauki (Sofia), 14, 48-54.
- MOTAS, C., 1903. La piroplasmose ovine ‘carceag’. Comptes Rendus des Seances Societe de Biologie et de Ses Filiates et Associees (Paris), 54, 1522-1524.
- NIU, Q., LIU, Z., YANG, J., YU, P., PAN, Y., ZHAI, B., LUO, J., GUAN, G. & YIN, H., 2016a. Expression of sheep pathogen Babesia sp. Xinjiang rhoptry-associated protein 1 and evaluation of its diagnostic potential by enzyme-linked immunosorbent assay. Parasitology, 143, 1990-1999.
- NIU, Q., LIU, Z., YANG, J., YU, P., PAN, Y., ZHAI, B., LUO, J., MOREAU, E., GUAN, G. & YIN, H., 2016b. Expression analysis and biological characterization of Babesia sp. BQ1 (Lintan) (Babesia motasi-like) rhoptry-associated protein 1 and its potential use in serodiagnosis via ELISA. Parasites and Vectors, 9, 313. doi: 10.1186/s13071-016-1573-7.
- NIU, Q., LIU, Z., YANG, J., YU, P., PAN, Y., ZHAI, B., LUO, J. & YIN, H., 2016c. Genetic diversity and molecular characterization of Babesia motasi-like in small ruminants and ixodid ticks from China. Infection, Genetics and Evolution, 41, 8-15.
- OPASINA, B. A., 1983. Ctenocephalides canis infection of goats. Tropical Animal Health and Production, 15, 106.
- OSWALD, B., 1939. On Yugoslavian (Balkan) ticks Ixodoided. Parasitology, 31, 271-280.
- OZKOC, U., 1979. Studies in the serological diagnosis of Babesia ovis infection in sheep by the fluorescent antibody technique. Pendik Veteriner Mikrobiyoloji Dergisi, 11, 70-83.
- OZKOC, U., ONAR, E. & DOGRU, C., 1982. An investigation on the relation of seasonal activity of Rhipicephalus bursa (Ixodoidea) with Babesia ovis infection in sheep in the Marmara region. Pendik Veteriner Mikrobiyoloji Dergisi, 14, 44-52.
- PAPADOPOULOS, B., BROSSARD, M. & PERIE, N. M., 1996. Piroplasms of domestic animals in the Macedonia region of Greece. 3. Piroplasms of small ruminants. Veterinary Parasitology, 63, 67-74.
- PAPADOPOULOS, B., PERIE, N. M. & UILENBERG, G., 1996. Piroplasms of domestic animals in the Macedonia region of Greece. 1. Serological cross-reactions. Veterinary Parasitology, 63, 41-56.
- PERRY, B. D., MUSISI, F. L., PEGRAM, R. G. & SCHELS, H. F., 1985. Assessment of enzootic stability to tick borne diseases. World Animal Review, 56, 24-31.
- POMERANTZEV, B. I., 1950. Fauna of the USSR. Arachnida IV No. 2. Ixodid Ticks (Ixodidae). Academy of Sciences Moscow, USSR.
- PURNELL, R. E., LEWIS, D., HOLMAN, M. R. & YOUNG, E. R., 1981. Investigations on a Babesia isolated from Scottish sheep. Parasitology, 83, 347-356.
- RAFI, A. & MAGHAMI, C., 1966. In: Report of FAO/OIE International Conference of Sheep Diseases. FAO. Rome 1966.
- RAY, H. N. & RAGHAVACHARI, K., 1941. Observations on Babesia foliata n. sp. from a sheep. Indian Journal of Veterinary Science, 11, 239-242.
- ROBSON, J., ROBB, J. M. & AL-WAHAYYIB, T., 1968. Ticks (Ixodoidea) of domestic animals in Iraq: Part 2. Summer infestations in the liwas of Hilla, Karbala, Diwaniya and Nasiriya. Journal of the Medical Entomology, 5, 27-31.
- ROSS, J. P. J. & LOHR, K. F., 1970. Ubertragung und Vetweildauer von Kolostral erworbenan Babesia bigemina und Anaplasma marginale Antikorpern. Zeitschrift für Tropenmedizin und Parasitologie, 21, 401-411.
- SERGENT, E., DONATIEN, A., PARROT, L. & LESTOQUARD, F., 1945. Etudes sur les Piroplasmoses Bovines. Institut Pasteur d’Algerie, Alger.
- SERGENT, E., DONATIEN, A., PARROT, L., LESTOQUARD, F. & PLANTUREUS, E., 1927. Etudes experimentales sur les Piroplasmoses Bovines d’Algerie. Annales Institut Pasteur, (Paris), 41, 721-723.
- SHERKOV, M. S., EL-RABIE, Y. & KOKASH, L., 1976. A survey of parasites blood disease ‘Tick Borne Fever’ in domestic animals in Jordan. Egypt Journal of Veterinary Science, 13, 29-35.
- SIMITCH, C. P., NEVENIC, V. & SIBALIC, S., 1956. Le traitement de la piroplasmose ovine et la piroplasmose bovine par ‘berenil’. Acta Veterinaria, Beograd, 6, 3-13.
- SMITH, R. D., 1983. Babesia bovis: Computer simulation of the relationship between the tick vector, parasite, and bovine host. Experimental Parasitology, 56, 27-40.
- SULEIMANOV, S. A., 1965. Intrauterine infection of sheep fetus with Babesia ovis Dagestan and Turkmenia. Trudy Vsesoyuznogo Instituta Eksperimental’noi Veterinarii, 31, 307-308.
- SULEIMANOV, S. A., 1976. Development and morphology of Babesia ovis (Babes, 1892) in the blood on the invertebrate host. Estestvennie Nauki, 2, 84-86.
- SUTEU, E., VARTIC, N., COSMA, A., IVASCU., ROTARU, O. & GORY, I., 1975. New data and observations on babesiosis in sheep in Transylvania. Bulletin Institutul Agronomic, 29, 107-109.
- THOMSON, J. G. & HALL, G. N., 1933. The occurrence of Babesia motasi Wenyon 1926, in sheep in northern Nigeria, with a discussion on the classification of the piroplasms. Journal of Comparative Pathology, 46, 218-231.
- TSOMAYA, I. V., 1960. Immunization of sheep against ovine babesiosis. Veterinaryia, Moscow, 37, 31.
- UILENBERG, G., 1964. Notes sur les Hematozoaires et tiques des animaux domestiques a Madagascar. Revue de Elevage Medecine veterinaire Pays tropicax, 17, 337-359.
- UILENBERG, G., ROMBACH, M. C., PERIE, N. M. & ZWART, D., 1980. Blood parasites of sheep in the Netherlands. II Babesia motasi (Sporozoa, Babesiidae). Veterinary Quarterly, 2, 3-14.
- VECHERKIN, S. S., 1957. Immunization of sheep against Babesia ovis infection. Trudy vesoyuznogo Instituto eksperimentalnoi Veterinarii, 21, 275-289.
- WANG, J. M., MA, M. L., LIU, A. H., REN, Q. Y., LI, A. Y., LIU, Z. J., LI, Y. Q., YIN, H., LUO, J. X. & GUAN, G. Q., 2013. A sero-epidemiological survey of Chinese Babesia motasi for small ruminants in China. Parasitology Research, 112, 2387-2391.
- WARNECKE, M., 1978. A survey of the infestation rate of ixodid ticks with piroplasms in Turkey. In: WILDE, J. K. H., (ed.). Tick-borne Diseases and their Vectors. Proceedings of an International Conference. Edinburgh, 1976. Edinburgh University Press.
- WENYON, C. M., 1926. Protozoology, a Manual for Medical Men, Veterinarians and Zoologists. London: Bailliére Tindal & Co. 2,
- WRIGHT, I. G., GOODGER, B. V. & MAHONEY, D. F., 1981. Virulent and avirulent strain of Babesia bovis. The relationships between parasite protease content and pathophysiological effect of the strain. Journal of Protozoology, 28, 118-120.
- YERUHAM, I., 1995. Babesiosis in sheep: The ecology of the tick vector Rhipicephalus bursa, transmission of Babesia ovis, immunological, clinical and clinico-pathological aspects. PhD Thesis. The Hebrew University of Jerusalem.
- YERUHAM, I., HADANI, A. & GALKER, F., 1998. Attenuation of Babesia ovis by rapid successive passages in splenectomized lambs. Tropical Animal Health and Production, 30, 53-55.
- YERUHAM, I., HADANI, A. & GALKER, F., 1998. Some epizootiological and clinical aspects of ovine babesiosis caused by Babesia ovis - a review. Veterinary Parasitology, 74, 153-163.
- YERUHAM, I., HADANI, A. & GALKER, F., 2000. The life cycle of Rhipicephalus bursa Canestrini and Fanzago, 1877 (Acarina: Ixodidae) under laboratory conditions. Veterinary Parasitology, 89, 109-116.
- YERUHAM, I., HADANI, A. & GALKER, F., 2001. The effect of the ovine host parasitaemia on the development of Babesia ovis (Babes, 1892) in the tick Rhipicephalus bursa (Canestrini and Fanzago, 1877). Veterinary Parasitology, 96, 195-202.
- YERUHAM, I., HADANI, A., GALKER, F., AVIDAR, Y. & BOGIN, E., 1998. Clinical, clinico-pathological and serological studies of Babesia ovis in experimentally infected sheep. Journal of Veterinary Medicine Ser B. 45, 385-394.
- YERUHAM, I., HADANI, A., GALKER, F., MAURER, E., RUBINA, M. & ROSEN, S., 1985. The geographical distribution and animal hosts of Rhipicephalus bursa (Canestrini and Fanzago, 1877) in Israel. Revue d’Elevage et Medecine veterinare Pays tropicaux, 38, 173-179.
- YERUHAM, I., HADANI, A., GALKER, F. & ROSEN, S., 1996. The seasonal occurrence of ticks (Acari: Ixodidae) on sheep and in the field in the Judean area of Israel. Experimental and Applied Acarology, 20, 47-56.
- YERUHAM, I., HADANI, A., GALKER, F., ROSEN, S. & SCHLEIN, J., 1992. A field study of haemoparasites in two flocks of sheep in Israel. Israel Journal of Veterinary Medicine, 47, 107-111.
- YERUHAM, I., HADANI, A., GALKER, F., ROSEN, S. & SCHLEIN, J., 1995. A study of an enzootic focus of sheep babesiosis (Babesia ovis, Babes 1892). Veterinary Parasitology, 60, 349-354.