- Infectious Diseases of Livestock
- Part 1
- Cryptosporidiosis
- Vectors: Ticks
- Vectors: Tsetse flies
- Vectors: Muscidae
- Vectors: Tabanidae
- Vectors: Culicoides spp.
- Vectors: Mosquitoes
- Classification, epidemiology and control of arthropod-borne viruses
- Special factors affecting the control of livestock diseases in sub-Saharan Africa
- The control of infectious diseases of livestock: Making appropriate decisions in different epidemiological and socioeconomic conditions
- Infectious diseases of animals in sub-Saharan Africa: The wildlife⁄livestock interface
- Vaccination: An approach to the control of infectious diseases
- African animal trypanosomoses
- Dourine
- Trichomonosis
- Amoebic infections
- GENERAL INTRODUCTION: COCCIDIA
- Coccidiosis
- Cryptosporidiosis
- Toxoplasmosis
- Besnoitiosis
- Sarcocystosis
- Balantidiosis
- Leishmaniosis
- Neosporosis
- Equine protozoal myeloencephalitis
- GENERAL INTRODUCTION: BABESIOSES
- Bovine babesiosis
- Equine piroplasmosis
- Porcine babesiosis
- Ovine babesiosis
- GENERAL INTRODUCTION: THEILERIOSES OF CATTLE
- East Coast fever
- Corridor disease
- Zimbabwe theileriosis
- Turning sickness
- Theileria taurotragi infection
- Theileria mutans infection
- Theileria annulata theileriosis
- Theileriosis of sheep and goats
- Theileria buffeli⁄orientalis infection
- Non-pathogenic Theileria species in cattle
- GENERAL INTRODUCTION: RICKETTSIAL, CHLAMYDIAL AND HAEMOTROPIC MYCOPLASMAL DISEASES
- Heartwater
- Lesser known rickettsial infections in animals and humans
- Chlamydiosis
- Q fever
- Eperythrozoonosis
- Bovine Haemobartonellosis
- Potomac horse fever
- GENERAL INTRODUCTION: ANAPLASMOSES
- Bovine anaplasmosis
- Ovine and caprine anaplasmosis
Cryptosporidiosis
This content is distributed under the following licence: Attribution-NonCommercial CC BY-NC  View Creative Commons Licence details here
View Creative Commons Licence details here
Cryptosporidiosis
Previous authors: C G STEWART AND B L PENZHORN
Current authors:
B BANGOURA - Assistant Professor, DVM, PhD, DipEVPC, University of Wyoming, Department of Veterinary Sciences, 1174 Snowy Range Road, Laramie, Wyoming, WY 82070, USA
A DAUGSCHIES - Professor, Centre for Infectious Diseases, Institute for Parasitology, Leipzig University, Leipzig, Germany
Introduction
Infections with parasites of the genus Cryptosporidium are a common finding in a variety of mammals, birds, fishes, and reptiles. The infections are often subclinical, but they may give rise to clinical disease characterized by lesions in the gastrointestinal tract followed by the development of diarrhoea with an occasional fatal outcome. This is particularly so in neonatal animals or humans or immunocompromised hosts.
Cryptosporidia are very small coccidia that were first described by Tyzzer in 190754 from the intestine of normal mice. Until 1955, when they were incriminated as a cause of disease in turkeys, they were thought to be commensals,50 but since then, diarrhoea associated with Cryptosporidium spp. has been reported in lambs,7 kids,58 piglets,59 immunologically deficient foals,51 and humans.42 Bovine intestinal cryptosporidiosis was first described in 1971 in the USA,44 and subsequent reports from the UK,57 Canada,40 Australia,28 and Germany24 indicate a wide geographical distribution of the parasite.26, 37
Cryptosporidiosis has also been identified as one of the five most important causes of diarrhoea in young children in Africa, Asia, and South America.45 Livestock infections are a common source of water- or food-borne infections in humans.14
Aetiology and life cycle
Cryptosporidium spp. are protozoa assigned to the phylum Apicomplexa, class Coccidia, order Eucoccidiorida, and suborder Eimeriorina of the family Cryptosporidiidae.13 In livestock, most of the parasites complete their life cycle in gastrointestinal epithelial cells.56 The life cycle of Cryptosporidium spp.resembles that of other intestinal coccidia but differs in that the parasites develop just under the surface plasmalemma of the host cell.30 In young animals the life cycle is completed in three to four days, which is shorter than the life cycle of most other coccidia.
Cryptosporidium spp. infect a broad range of vertebrate hosts.19 Cryptosporidium parvum is associated with disease in a wide range of mammals including ruminants, pigs, mice, and humans. Some Cryptosporidium spp.have a higher host specificity for certain mammalian hosts e.g. C. canis (dogs), C. felis (cats), C. hominis (humans), C. muris (mice), and C. wrairi (guinea pigs), causing pathogenic effects similar to C. parvum in their respective hosts. In cattle, there is an additional species residing in the epithelium of the peptic glands of the abomasum, C. andersoni, which has not been associated with disease. Birds are commonly parasitized by C. meleagridis (turkeys mainly, but infection may spread to humans), C. galli (chickens), or C. baileyi (gallinaceous birds, water fowl, and others). Reptiles often harbour C. saurophilum or C. serpentis (snakes, lizards). Fish may be infected by C. molnari.62
Cryptosporidium parvum generally parasitizes intestinal epithelial cells,2 and in ruminants its oocysts can be differentiated from those of C. andersoni by their smaller size: C. parvum oocysts measure 4.5–5.4 × 4.2–5 μm and those of C. andersoni 6.6 –7.9 × 5.3–6.5 μm.60
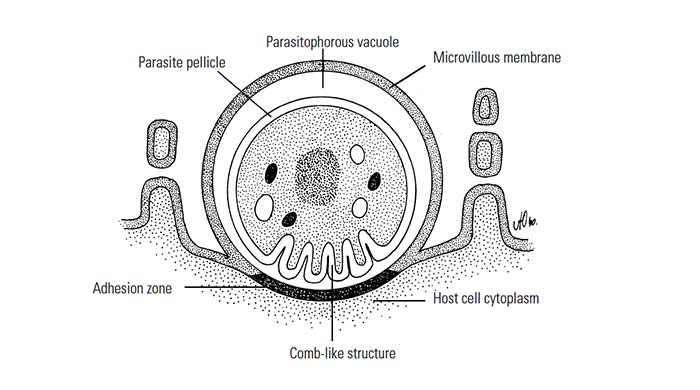
Following ingestion of oocysts by the host, sporozoites are released into the intestine where they penetrate the brush borders of the epithelial cells to develop into meronts. Although organisms appear to be attached superficially to cells, all stages are intracellular and are contained within a parasitophorous vacuole situated between the cytoplasm and outer cell membrane.19 The parasite is surrounded by the microvillous membrane, which fuses with the epithelial plasma membrane to form a thickened adhesion zone at the point of attachment to the host cell (Figure 1) There is also an adjacent comb-like structure formed by a folding of the parasite pellicle.43 These specialized structures are thought to facilitate the intake of nutrients by the parasite from the host cell. The meronts are 2 to 5 μm in diameter and contain eight merozoites that are released into the lumen of the gut. Second-generation meronts may occur, and these will contain four merozoites when mature. These merozoites invade new host cells where they undergo gamogony by forming macro- and microgamonts. The microgamont has sperm-like microgametes which, when released, fertilize the macrogamonts, which then develop into oocysts. Oocysts are round or ellipsoidal, and contain four sporozoites. It is generally accepted that sporocysts do not occur.32 Sporulation takes place within the host cell and is followed by the development of a resistant thick wall around some of the oocysts, which are shed in the faeces.19 Other oocysts, however, do not form a thick wall and the single unit membrane surrounding the sporozoites ruptures when these oocysts are released from the host cell. These forms are invasive and can infect epithelial cells and so reinitiate the development cycle, resulting in auto-infection mainly in immunocompromised hosts.17
Epidemiology
In livestock, clinical cryptosporidiosis occurs most frequently in calves, but it has also been recognized in pigs, lambs, kids and immunocompromised foals.5, 38, 46, 51, 58, 59
Outbreaks of clinical disease are most common in animals one to four weeks of age. Diarrhoeic calves may shed as many as 10 million oocysts per gram of faeces, resulting in heavy contamination of the environment.4 Cryptosporidia may also be found in the faeces of subclinically infected cattle of all ages.
Oocysts are resistant to most environmental factors, but the time of survival outside the host is unknown. Chlorination of water has little effect on viability of the organisms, hence water-borne outbreaks of the disease are an important public health problem.63 Under experimental conditions infectivity is observed for at least seven months after storage at 15 °C.27
Transmission is by ingestion of oocysts in food or water contaminated with faeces, while aerosol transmission may also occur in both humans and animals.25, 55
Calves that consume adequate colostrum and have high serum gammaglobulin concentrations are less likely to become infected.36 Recovery usually occurs within 30 days.1 There are reports in which Cryptosporidium has been incriminated as the only cause of diarrhoea, but cryptosporidia infections often occur together with other enteric pathogens, such as rotavirus, coronavirus, Salmonella serovars and enterotoxogenic Escherichia coli. These multiple infections act synergistically and increase the mortality rate. Severe stress, such as exposure to cold temperatures, may also increase mortality. Immunodeficiency may be important in the development of clinical disease. Severe infections have been reported in Arabian foals suffering from the combined immunodeficiency syndrome51 and in humans with acquired immune deficiency syndrome (AIDS). In foals the disease is more prevalent in the first three to four weeks of life but can occur in animals up to one year of age.16
Persistent infection in athymic mice and AIDS patients demonstrated that T cells are necessary for immunity to develop.63 In calves, a strong cell-mediated immunity develops after infection, with an increase in CD8+ T cells.1 As oocysts are produced in the sporulated state, auto-infection is particularly important in immunologically compromised hosts, resulting in persistent infection.56
In humans, Cryptosporidium spp. infections may be acquired by contact with infected individuals or from animal sources, and it is a common cause of travellers’ diarrhoea.52 The low host specificity, excretion of large numbers of oocysts and low numbers of oocysts required to initiate infection in susceptible hosts imply that animal species can be potential reservoirs of C. parvum for human infections.11 Besides infections with the anthropozoonotic C. parvum acquired from animals, humans are most frequently infected with C. hominis, a zooanthroponotic species circulating in human populations. Other Cryptosporidium spp. are rarely transmitted from animals to humans.47
Pathogenesis and clinical signs
Cryptosporidium spp. have a marked effect on membrane-bound digestive enzymes, resulting in poor digestion. Malabsorption may occur as a result of villous atrophy.56
Except when it occurs in very young animals, livestock infection is inapparent or, at most, produces only mild disease.3, 30 The incubation period varies between two and seven days.39 Diarrhoea occurs at between one and four weeks of age and lasts for one to two weeks. Although the morbidity rate may be high the mortality rate is generally very low.55 The onset of overt disease is usually sudden and clinical signs are very similar in all affected livestock species. Depression and anorexia are followed by a profuse yellow watery diarrhoea with associated dehydration. Tenesmus may be present. After a few days the diarrhoea becomes intermittent and the faeces pasty for up to 10 days.30 Loss of body weight may be quite marked. Relapses after apparent recovery may occur.55 Vomiting may occur in pigs.59 Most affected animals recover spontaneously unless the disease is complicated by infection with other enteropathogens. Severe chronic infections may occur in immunocompromised hosts.2
Pathology
Generally, the extent of the mucosal injury in the gastrointestinal tract and the severity of the disease are directly correlated.30 Enteric lesions increase in severity towards the terminal portion of the ileum, and the caecum and colon may also be infected.55 The mucosa of affected parts of the intestine, which often contains watery yellow material, is intensely hyperaemic. The carcass may be emaciated and dehydrated.
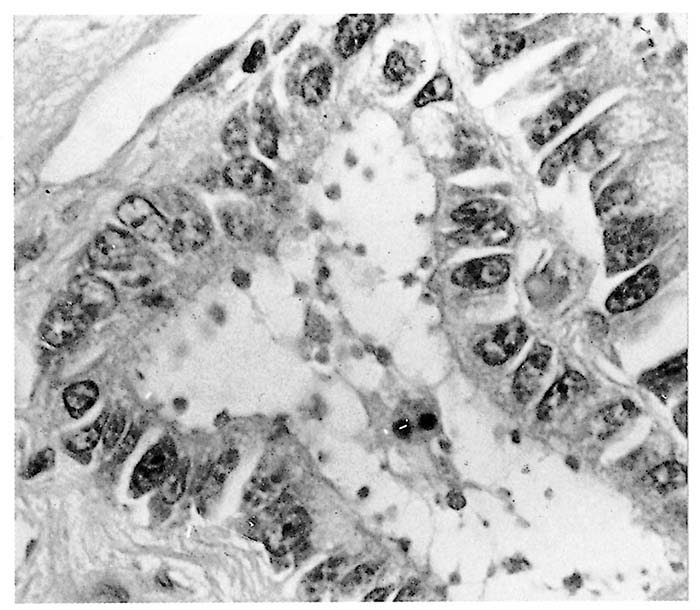
Stunting, fusion and cross-bridging of adjacent intestinal villi are seen histologically.4 In affected parts of the intestine, mucosal epithelial cells are often low columnar, cuboidal or even squamous and, in calves, there may be focal necrosis of epithelial cells in some areas.19, 23 The very small basophilic organisms, generally appearing as small dots, although ring or crescent-shaped forms may also be identified, are found embedded in the microvilli or just inside the distal cell membrane of epithelial cells (Figure 2) They are more easily demonstrated in tissue sections that are stained with Giemsa.5, 53 Extra-intestinal forms in the gall-bladder, mesenteric lymph nodes, trachea, lungs and uterus have been identified in immunocompromised humans49 as well as in pigs and sheep.20 In the bird parasite, C. baileyi, intestinal or respiratory infections are common.10
Diagnosis and differential diagnosis
Demonstration of numerous oocysts in the faeces of young animals manifesting clinical signs of enteritis, or in preparations of intestinal tissue, are the most reliable means of diagnosis. However, the finding of Cryptosporidium does not necessarily mean that the organism is involved in the disease process, and it is therefore important to associate the relevant clinical signs and histological lesions to the pathogen or other pathogens isolated before making a diagnosis.
In formalin-fixed tissue sections, endogenous stages can be demonstrated in the brush border of intestinal epithelial cells.55 Care must be taken to distinguish organisms in tissue sections from small droplets of mucus adhering to the surface of epithelial cells.15
Cryptosporidium spp. oocysts are very small but can be recognized in faecal smears fixed in methanol and stained with modified Ziehl-Neelsen4 or safranin. The latter stain may be the most sensitive.8 Oocysts appear as red spheres against a dark background and need to be differentiated from yeasts, which do not stain red. High sensitivity has been reported using fast staining methods like carbol-fuchsine stain31 or immunofluorescent staining techniques, but specificity of the latter may vary. However, the fluorescent in situ hybridization (FISH) test is highly efficient in determining not only the presence of Cryptosporidium parasites but also the species and viability of oocysts.18
Techniques for concentrating oocysts in faecal specimens, such as those used for the diagnosis of coccidiosis, may facilitate identification. The final preparation can be examined directly, or stained by a safranine-methylene blue method.21, 37
The most sensitive tools for Cryptosporidium diagnosis are nucleid acid detection by polymerase chain reaction (PCR) or antigen detection e.g. by enzyme-linked immunosorbent assay (ELISA) in clinical faecal or tracheal lavage samples.22, 31 However, not all ELISAs may detect all Cryptosporidium spp., depending on the targeted antigen.
Clinically, cryptosporidiosis should be differentiated from other enteric conditions, such as colibacillosis, salmonellosis, rotavirus or coronavirus infections and coccidiosis (see the relevant chapters). Under field conditions, however, diarrhoea is often associated with a simultaneous infection with two or more micro-organisms,30 and this, together with the tendency for the infection to be subclinical, makes diagnosis difficult. For livestock, a herd diagnosis should be made by screening several samples within an age group.
Control
In general, treatment of overt disease is not done in immunocompetent hosts because infection is in most cases self-limiting.
Few chemotherapeuticals are available to treat these infections. Halofuginone lactate at a dose of 120 μg/kg body weight administered orally for seven days is highly effective against cryptosporidiosis and reduces oocyst excretion.29 Administration of fluid therapy and other symptomatic treatment is important in the prevention of dehydration and the correction of the electrolyte imbalance caused by the diarrhoea. If managed correctly, most animals will recover unless the disease is complicated by infection with other enteropathogens.
Prevention should be aimed at reduction of the ingestion of oocysts by raising young animals in a clean and dry environment. Faecal material should be removed from pens on a regular basis, and animals suffering from diarrhoea should be kept in isolation. Although oocysts are resistant to many disinfectants and most prevailing environmental conditions, they may be destroyed by exposure to (chloro-) creosols,29 5 per cent ammonia solution or 10 per cent formalin.12
References
- ABRAHAMSEN, M.S., 1998. Bovine T cell responses in Cryptosporidium parvum infection. International Journal of Parasitology, 28, 1083–1088.
- ANDERSON, B.C., 1987. Abomasal cryptosporidiosis in cattle. Veterinary Pathology, 24, 235–238.
- ANDERSON, B.C., 1998. Cryptosporidiosis in bovine and human health. Journal of Dairy Science, 81, 3036–3041.
- ANGUS, K.W., 1987. Cryptosporidiosis in domestic animals and humans. In Practice, 9, 47–49.
- ANGUS, K.W., TZIPORI, S. & GRAY, E.W., 1982. Intestinal lesions in specific pathogen-free lambs associated with a Cryptosporidium from calves with diarrhea. Veterinary Pathology, 19, 67–78.
- AWAD-EL-KARIEM, F. M., ROBINSON, H. A., PETRY, F., MCDONALD, V., EVANS, D. & CASEMORE, D., 1998. Differentiation between human and animal isolates of Cryptosporidium parvum using molecular and biological markers. Parasitological Research, 84, 297–301.
- BAKER, I.K. & CARBONELL, P.L., 1974. Cryptosporidium agni species nova from lambs, and Cryptosporidium bovis species nova from a calf with observations on the oocyst. Zeitschrift fu¨r Parasitenkunde, 44, 289–298.
- BAXBY, D. & BLUNDELL, N., 1983. Sensitive, rapid, simple methods for detecting Cryptosporidium in faeces. Lancet, 2, 1149.
- BERKOWITZ, F.E., VALLABH, W., BUGWANA, A. & HENEY, C., 1988. Cryptosporidiosis in black South African children. South African Medical Journal, 74, 272–273.
- BLAGBURN, B. L., LINDSAY, D. S., HOERR, F. J., DAVIS, J. F. & GIAMBRONE, J.J., 1991. Pathobiology of cryptosporidiosis (Cryptosporidium baileyi) in broiler chickens. Journal of Protozoology, 38(6), 25S-28S.
- BUKHARI, Z. & SMITH, H.V., 1997. Cryptosporidium parvum: oocyst excretion and viability in experimentally infected lambs. Epidemiology and Infection, 119, 105–108.
- CAMPBELL, J., TZIPORI, S., HUTCHISON, G. & ANGUS, K.W., 1982. Effect of disinfectants on survival of cryptosporidium oocysts. The Veterinary Record, 111, 414–415.
- CARRENO. R. A., MARTIN, D. S. & BARTA, J. R., 1999. Cryptosporidium is more closely related to the gregarines than to coccidia as shown by phylogenetic analysis of apicomplexan parasites inferred using small-subunit ribosomal RNA gene sequences. Parasitology Research, 85(11), 899-904.
- CHAKO, C. Z., TYLER, J. W., SCHULTZ, L. G., CHIGUMA, L. & BEERNTSEN, B. T., 2010. Cryptosporidiosis in people: It’s not just about the cows. Journal of Veterinary Internal Medicine, 24, 37-43.
- COETZER, J.A.W., 1990. Faculty of Veterinary Science, University of Pretoria. Personal communication.
- COHEN, N.D. & SNOWDEN, K., 1996. Cryptosporidial diarrhea in foals. Foal Medicine and Surgery, 18, 298–306.
- CURRENT, W.L., 1985. Cryptosporidiosis. Journal of the American Veterinary Medical Association, 187, 1334–1338.
- FAYER, R., MORGAN, U. & UPTON, S.J., 2000. Epidemiology of Cryptosporidium: transmission, detection and identification. International Journal of Parasitology, 30(12-13), 1305-22.
- FAYER, R. & UNGAR, L.P., 1986.Cryptosporidium sub species and cryptosporidiosis. Microbiological Reviews, 50, 458–483.
- FLETA, J., SANCHES-ACEDO, C., CLAVEL, A. & QUILEZ, J., 1995. Detection of Cryptosporidium oocysts in extra-intestinal tissues of sheep and pigs. Veterinary Parasitology, 59, 201–205.
- FRIPP, P.J. & BOTHMA, M.T., 1987. Cryptosporidium associated with diarrhoea at Ga-Rankuwa hospital. The Southern African Journal of Epidemiology and Infection, 2, 45–47.
- HADFIELD, S. J., ROBINSON, G., ELWIN, K. & CHALMERS, R. M., 2011. Detection and differentiation of Cryptosporidium sub species in human clinical samples by use of real-time PCR. Journal of Clinical Microbiology, 49(3), 918-24.
- HALL, G.A., REYNOLDS, D.J., PARSONS, K.R., BLAND, A.P. & MORGAN, J.H., 1988. Pathology of calves with diarrhoea in southern Britain. Research in Veterinary Science, 45, 240–250.
- HEINE, V.J. & BOCH, J., 1981. Kryptosporidien Infektionen beim Kalb. Nachweis, Vorkommen und experimentelle Übertragung. Berliner und Mu¨nchener Tierärztliche Wochenschrift, 94, 289–292.
- HOJLYNG, N., HOLTEN-ANDERSEN, W. & JEPSEN, S., 1987. Cryptosporidiosis: A case for airborne transmission. Lancet, 2(8553), 271–272.
- HOWERTH, E.W., 1981. Bovine cryptosporidiosis. Journal of the South African Veterinary Association, 52, 251–253.
- JENKINS, M., TROUT, J. M., HIGGINS, J., DORSCH, M., VEAL, D. & FAYER, R., 2003. Comparison of tests for viable and infectious Cryptosporidium parvum oocysts. Parasitology Research, 89(1), 1-5.
- JERRETT, I.V. & SNODGRASS, D.R., 1981. Cryptosporidia associated with outbreaks of neonatal calf diarrhoea. Australian Veterinary Journal, 57, 434–435.
- KEIDEL, J. & DAUGSCHIES, A., 2013. Integration of halofuginone lactate treatment and disinfection with p-chloro-m-cresol to control natural cryptosporidiosis in calves. Veterinary Parasitology, 196(3-4), 321-6.
- KIRKPATRICK, C.E. & FARREL, J.P., 1984. Cryptosporidiosis. The Compendium on Continuing Education for Veterinarians, 6, 154–162.
- KUHNERT-PAUL, Y., BANGOURA, B., DITTMAR, K., DAUGSCHIES, A. & SCHMÄSCHKE, R., 2012. Cryptosporidiosis: comparison of three diagnostic methods and effects of storage temperature on detectability of cryptosporidia in cattle faeces. Parasitology Research, 111(1), 165-71.
- LEVINE, N.D., 1984. Taxonomy and review of the coccidian genus Cryptosporidium (Protozoa, Apicomplexa). Journal of Protozoology, 31, 94–98.
- LEVINE, N.D., 1985. Veterinary Protozoology. Ames, Iowa: Iowa State University Press.
- LINDSAY, D.S., UPTON, S.J., OWENS, D.S., MORGAN, U. M., MEAD, J.R. & BLAGBURN, B.L., 2000. Cryptosporidium andersoni naegleria species (Apicomplexa: Cryptosporiidae) from cattle, Bos taurus. Journal of Eukaryotic Microbiology, 47, 91–95.
- LINDSAY, D.S., BLAGBURN, B.L., SUNDERMANN, C.A. & ERNEST, J.A., 1987. Chemoprophylaxis of cryptosporidiosis in chickens, using halfunginone, salinomycin, lasolocid, or monensin. American Journal of Veterinary Research, 48, 354–355.
- LOPEZ, J.W., ALLEN, S.D., MITCHELL, J. & QUINN, M., 1988. Rotavirus and Cryptosporidium shedding in dairy calf faeces and its relationship to colostrum immune transfer. Journal of Dairy Science, 71, 1288–1294.
- MARKUS, M.B. & BUSH, J.B., 1987. Staining of coccidial oocysts. The Veterinary Record, 121, 329.
- MOON, H.W. & BEMWICK, W.J., 1981. Fecal transmission of calf cryptosporidia between calves and pigs. Veterinary Pathology, 18, 248–255.
- MOORE, D.A., 1989. Minimizing morbidity and mortality from cryptosporidiosis. Veterinary Medicine, 84, 811–815.
- MORIN, M., LARIVIORE, S. & LALLIER, R., 1976. Pathological and microbiological observations made on spontaneous cases of acute neonatal calf diarrhoea. Canadian Journal of Comparative Medicine, 40, 228–240.
- NACIRI, M., 1989. Animal and human cryptosporidiosis: Opportunist infections? Pathogenicity of the genus Cryptosporidium. Coccidia and Intestinal Coccidiomorphs. Proceedings of the Fifth International Coccidiosis Conference, Tours (France), 17–20 October 1989. INRA 147 Rue de P’Unurersite.
- NIME, F.A., BURCK, J.D., PAGE, D.L., HOLSCHER, M.A. & YARDLEY, J.H., 1976. Acute enterocolitis in a human being infected with the protozoan Cryptosporidium. Gastroenterology, 70, 592–598.
- O’DONOGHUE, P.J., 1985. Cryptosporidium infection in man, animals, birds and fish. Australian Veterinary Journal, 62, 253–258.
- PANCIERA, R.J., THOMASSEN, R.W. & GARNER, F.M., 1971. Cryptosporidial infection in a calf. Veterinary Pathology, 8, 479–484.
- PLATTS-MILLS, J.A., BABJI, S., BODHIDATTA, L., GRATZ, J., HAQUE, R., HAVT, A., MCCORMICK, B.J., MCGRATH, M., OLORTEGUI, M.P., SAMIE, A., SHAKOOR, S., MONDAL, D., LIMA, I,F., HARIRAJU, D., RAYAMAJHI, B.B., QURESHI, S., KABIR, F., YORI, P.P., MUFAMADI, B., AMOUR, C., CARREON, J.D., RICHARD, S.A., LANG, D., BESSONG, P., MDUMA, E., AHMED, T., LIMA, A.A., MASON, C.J., ZAIDI, A.K., BHUTTA, Z.A., KOSEK, M., GUERRANT, R.I., GOTTLIEB, M., MILLER, M., KANG, G., HOUPT, E.R. & MAL-ED NETWORK INVESTIGATORS., 2015. Pathogen-specific burdens of community diarrhoea in developing countries: a multisite birth cohort study (MAL-ED). Lancet Global Health, 3:e564–e575.
- POHLENZ, J., MOON, H.W., CHEVILLE, N.F. & BEMRICK, W.J., 1978. Cryptosporidiosis as a probable factor of neonatal diarrhoea in calves. Journal of the American Veterinary Medical Association, 172, 452–457.
- RYAN, U., FAYER, R. & XIAO, L., 2014. Cryptosporidium species in humans and animals: current understanding and research needs. Parasitology, 141(13), 1667-85.
- SCHAEFER, D.A., BETZER, D.P., SMITH, K.D., MILLMAN, Z.G., MICHALSKI, H.C., MENCHACA, S.E, ZAMBRISKI, J.A., OJO, K.K., HULVERSON, M.A., ARNOLD, S.L., RIVAS, K.L., VIDADALA, R.S., HUANG, W., BARRETT, L.K., MALY, D.J., FAN, E., VAN VOORHIS, W.C. & RIGGS, M.W., 2016. Novel Bumped Kinase Inhibitors Are Safe and Effective Therapeutics in the Calf Clinical Model for Cryptosporidiosis. Journal of Infectious Diseases, 214(12), 1856-1864.
- SHRIVASTAVA, A.K., KUMAR, S., SMITH, W.A. & SAHU, P.S., 2017. Revisiting the global problem of cryptosporidiosis and recommendations. Tropical Parasitology, 7(1), 8-17.
- SLAVIN, D., 1955. Cryptosporidium meleagridis (species nova). Journal of Comparative Pathology, 65, 262–266.
- SNYDER, S.P., ENGLAND, J.J. & MCCHESNEY, A.E., 1978. Cryptosporidiosis in immunodeficient Arabian foals. Veterinary Pathology, 15, 12–17.
- SUN, T. & TEICHBERG, S., 1988. Protozoal infections in the acquired immunodeficiency syndrome. Journal of Electron Microscopy Techniques, 8, 79–103.
- THOMSON, R.G., 1988.Special Veterinary Pathology. Toronto: B.C. Decker Inc.
- TYZZER, E.E., 1907. A sporozoan found in the peptic glands of the common mouse. Proceedings of the Society of Experimental Biology and Medicine, 5, 12–13.
- TZIPORI, S., 1983. Cryptosporidiosis in animals and humans. Microbiological Reviews, 47, 84–96.
- TZIPORI, S., 1985. The relative importance of enteric pathogens affecting neonates of domestic animals. Advances in Veterinary Science and Comparative Medicine, 29, 103–206.
- TZIPORI, S., CAMPBELL, I., SHERWOOD, D. & SNODGRASS, D.R., 1980. An outbreak of calf diarrhoea attributed to cryptosporidial infection. The Veterinary Record, 107, 579–580.
- TZIPORI, S., LARSEN, J., SMITH, M. & LUEFL, R., 1982. Diarrhoea in goat kids attributed to Cryptosporidium infection. The Veterinary Record, 111, 35–36.
- TZIPORI, S., MCCARTNEY, E., LAWSON, G.H.K. & ROWLAND, A.C., 1981. Experimental infection of piglets with Cryptosporidium. Research in Veterinary Science, 31, 358–368.
- UPTON, S.J. & CURRENT, N.L., 1985. The species of Cryptosporidium (Apicomplexa: Cryptosporidiidae) infecting animals. Journal of Parasitology, 71, 625–629.
- WEBSTER, K.A., 1993. Molecular methods for detection and classification of Cryptosporidium. Parasitology Today, 9, 263–266.
- XIAO, L., FAYER, R., RYAN, U. & UPTON, S.J, 2004. Cryptosporidium taxonomy: Recent advances and implications for public health. Clinical Microbiology Reviews, 17(1), 72-97.
- ZU, S-X. FANG, G-D., FAYER, R. & GUERRANT, R.L., 1992. Cryptosporidiosis: pathogenesis and immunology. Parasitology Today, 8, 24–27.