- Infectious Diseases of Livestock
- Part 1
- Neosporosis
- Vectors: Ticks
- Vectors: Tsetse flies
- Vectors: Muscidae
- Vectors: Tabanidae
- Vectors: Culicoides spp.
- Vectors: Mosquitoes
- Classification, epidemiology and control of arthropod-borne viruses
- Special factors affecting the control of livestock diseases in sub-Saharan Africa
- The control of infectious diseases of livestock: Making appropriate decisions in different epidemiological and socioeconomic conditions
- Infectious diseases of animals in sub-Saharan Africa: The wildlife⁄livestock interface
- Vaccination: An approach to the control of infectious diseases
- African animal trypanosomoses
- Dourine
- Trichomonosis
- Amoebic infections
- GENERAL INTRODUCTION: COCCIDIA
- Coccidiosis
- Cryptosporidiosis
- Toxoplasmosis
- Besnoitiosis
- Sarcocystosis
- Balantidiosis
- Leishmaniosis
- Neosporosis
- Equine protozoal myeloencephalitis
- GENERAL INTRODUCTION: BABESIOSES
- Bovine babesiosis
- Equine piroplasmosis
- Porcine babesiosis
- Ovine babesiosis
- GENERAL INTRODUCTION: THEILERIOSES OF CATTLE
- East Coast fever
- Corridor disease
- Zimbabwe theileriosis
- Turning sickness
- Theileria taurotragi infection
- Theileria mutans infection
- Theileria annulata theileriosis
- Theileriosis of sheep and goats
- Theileria buffeli⁄orientalis infection
- Non-pathogenic Theileria species in cattle
- GENERAL INTRODUCTION: RICKETTSIAL, CHLAMYDIAL AND HAEMOTROPIC MYCOPLASMAL DISEASES
- Heartwater
- Lesser known rickettsial infections in animals and humans
- Chlamydiosis
- Q fever
- Eperythrozoonosis
- Bovine Haemobartonellosis
- Potomac horse fever
- GENERAL INTRODUCTION: ANAPLASMOSES
- Bovine anaplasmosis
- Ovine and caprine anaplasmosis
Neosporosis
This content is distributed under the following licence: Attribution-NonCommercial CC BY-NC  View Creative Commons Licence details here
View Creative Commons Licence details here
NJ Maclachlan and M-L Penrith (Editors). JP Dubey, Neosporosis, 2018.
Neosporosis
Previous authors: J P DUBEY
Current authors:
J P DUBEY - Senior scientist, M.V.Sc., PhD.,DSc, Animal Parasitic Diseases Laboratory, U.S.Department of Agriculture, Building 1001, Beltsville Agricultural Research Center, 10300 Baltimore Avenue, Beltsville, Maryland, 20705, USA
Introduction
Neospora caninum is a relatively recently recognized protozoan parasite that occurs in a wide range of animals species. Until 1988 it was misdiagnosed as Toxoplasma gondii because of structural and biological similarities.52 The disease was first described in dogs by Bjerkås et al.25 in Norway in 1984 but the parasite was not named at that time. Dubey and his co-workers in 198852 described the parasite and proposed a new genus, Neospora, with N. caninum as the type species. In vitro isolation of the parasite in cell culture inoculated with tissues of paralysed dogs contributed to a better understanding of its biology.57 In 1988, neosporosis was recognized as a cause of abortion in cattle163 and in the subsequent three years infections were induced experimentally in most species of livestock.63 The genome of N. caninum was sequenced in 2012,146 suggesting that N. caninum and T. gondii diverged from their common ancestor approximately 28 million years ago. A book authored by Dubey58 devoted solely to neosporosis listed more than 2000 references: much of the information in this chapter is extracted from that book and only selected references are cited here.58
Aetiology and life cycle
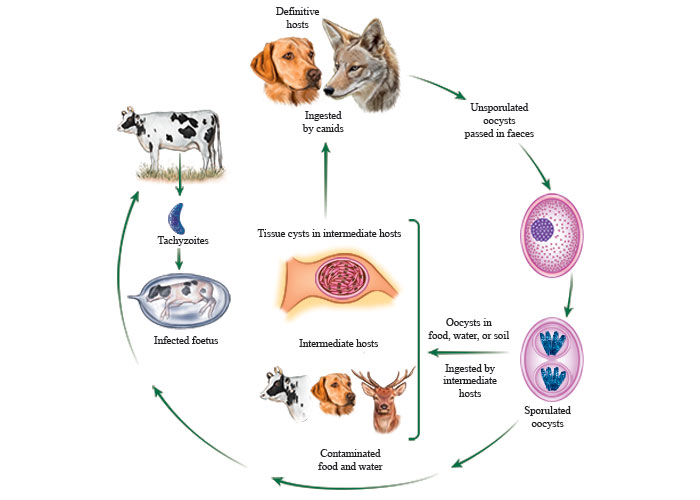
Neospora caninum is a coccidian parasite closely related to T. gondii (Figure 1). Dogs (both domestic and feral or wild, Canis familiaris), coyote (Canis latrans), and gray wolf (Canis lupus) are intermediate and definitive hosts.61, 78, 104, 122 Cattle, sheep, white- tailed deer, European bison and water buffalo are intermediate hosts, as proven by isolation of viable parasites.58 Clinical neosporosis has been reported in numerous hosts including alpaca (Vicugna pacos), captive antelope (Tragelaphus imberbis), Axis deer (Axis axis), black-tailed deer (Oedocoileus hemionus columbianus), cattle, dog, Eld’s deer (Panolia eldii siamensis), goat, llama (Lama glama), Parma wallaby (Macropus parma), Pine marten (Martes martes), red fox (Vulpes vulpes), white rhinoceros (Ceratotherium simum), and sheep.58, 97-101, 171 Antibodies and DNA to N. caninum without evidence of disease have been detected in many mammalian and avian species.58
Only an asexual cycle occurs in the intermediate hosts that consists of tachyzoites and tissue cysts. Both tachyzoites and tissue cysts are microscopic in size and intracellular. Tachyzoites are ovoid, lunate or globular and measure 3–7 μm × 1–5 μm, depending on the stage of division (Figures 2 and 3). Tachyzoites are found in the cytoplasm in many cells including neurons, macrophages, fibroblasts, vascular endothelial cells, myocytes, hepatocytes and dermal cells. Tachyzoites are usually located in a parasitophorous vacuole in the host cell. Ultrastructurally, the tachyzoites of N. caninum resembles the tachyzoites of T. gondii except that the rhoptries in N. caninum are electron-dense and more numerous, with some extending the entire length of the parasite, whereas in T. gondii, they are electron-lucent, few, and located anterior to the nucleus.58
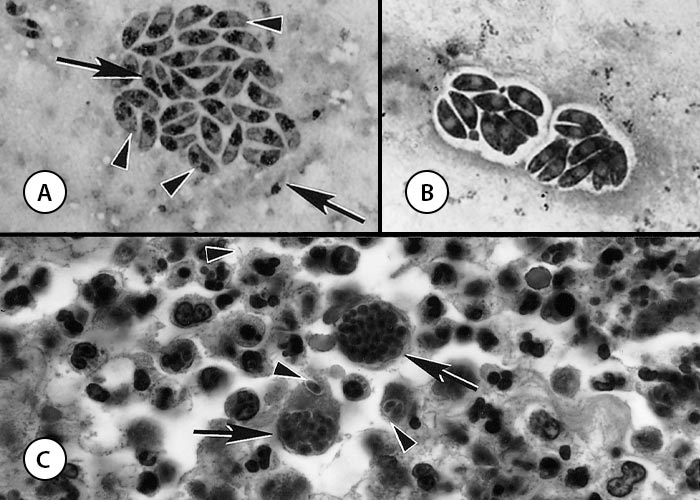
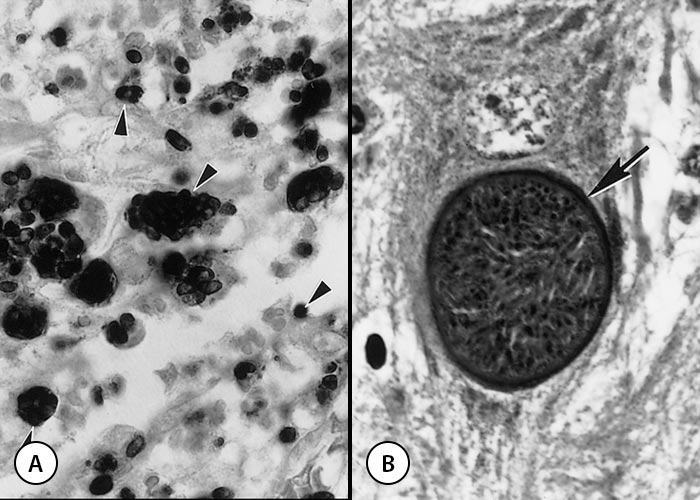
Figure 2 Tachyzoites of Neospora caninum
- Impression smear. Note organisms dividing (arrowheads) into two are bigger than single tachyzoites (arrows). Giemsa stain
- Tachyzoites in parasitophorous vacuoles in cell culture. Giemsa stain
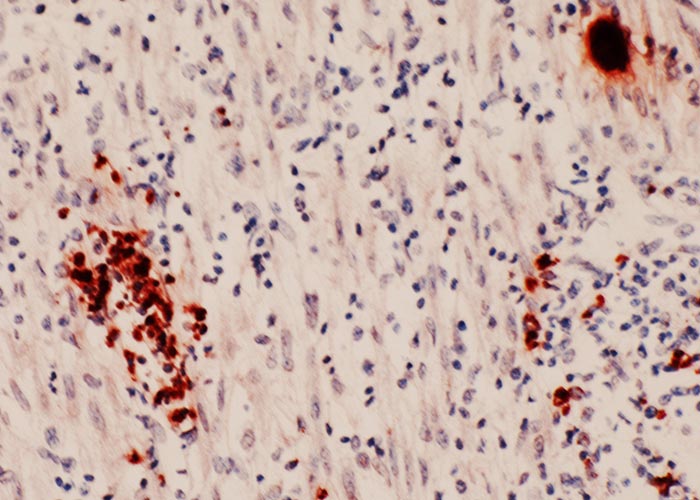
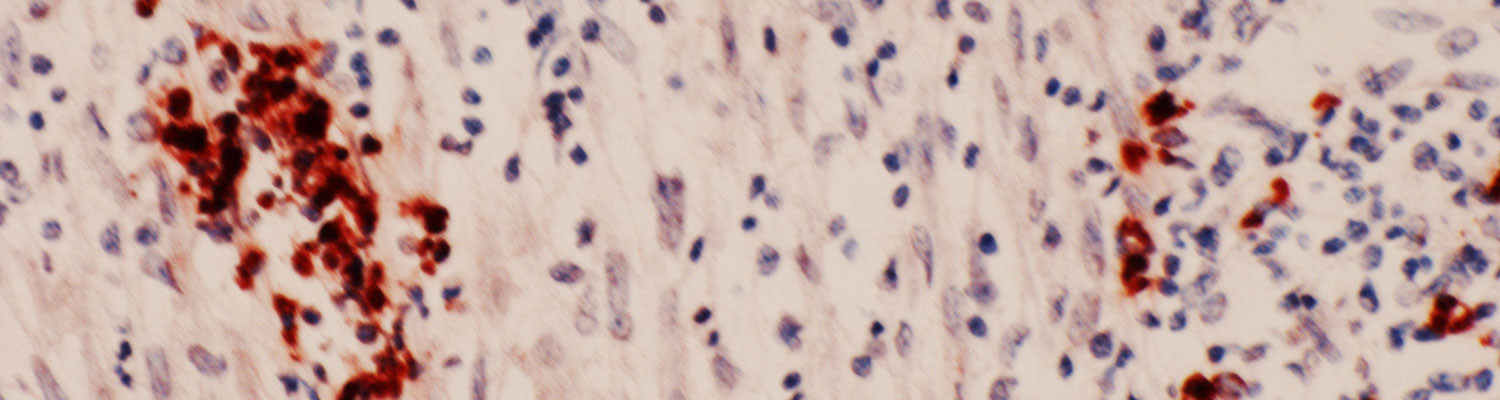
- Sections of the skin of a dog. Note suppurative inflammation associated with two groups of tachyzoites (arrows) and individual tachyzoites (arrowheads). Tachyzoites in sections are much smaller than those in smears. Haematoxylin and eosin stain
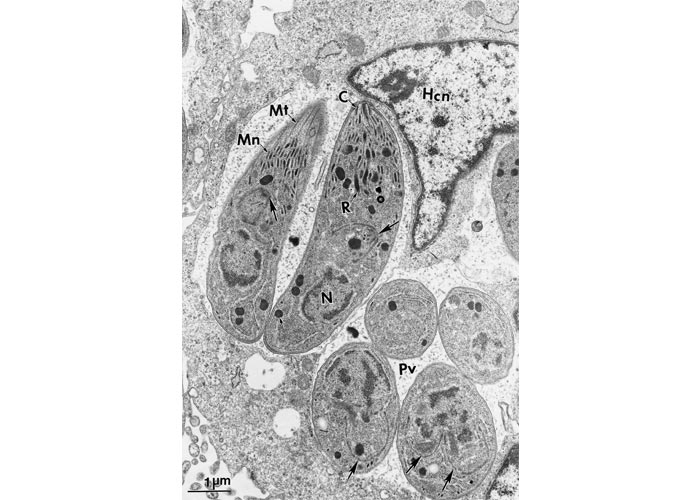
Figure 3 Transmission electron micrograph of dividing tachyzoites in two parasitophorous vacuoles (Pv). The Pv have many tubular networks. Arrows point to the conoidal end of daughter tachyzoites forming inside the mother tachyzoites. Note electron-dense rhoptries (R), micronemes (Mn), microtubules (Mt) or subpellicullar tubules, conoid (C), host cell nucleus (Hcn). Cardiopulmonary artery endothelial cell culture. (Courtesy of Dr C.A. Speer, Montana State University, Bozeman, Montana, USA)
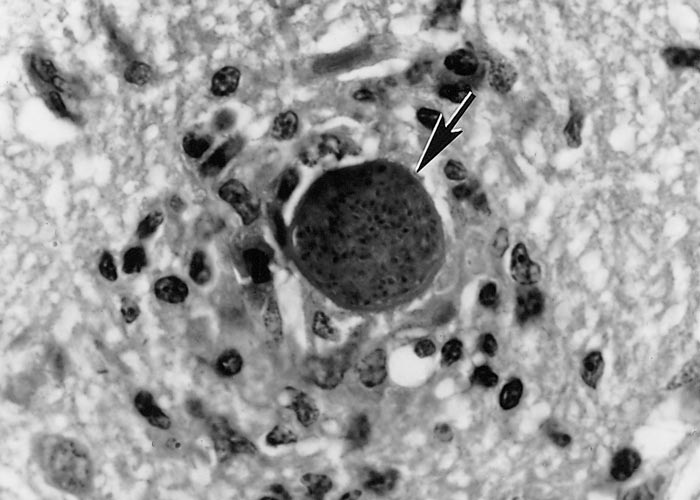
Tissue cysts are often round to oval in shape (Figure 4). Most tissue cysts studied were from the brain and spinal cord, the majority from naturally infected dogs. Tissue cysts in the brain are round to oval and up to 107 µm in diameter. The cyst wall is 0.5 to 4.0 µm thick, both in live, unstained preparations and in histological sections.51 The cysts in myocytes are elongated and may be up to 100 µm long, and have a thin cyst wall.71, 141 Histologically tissue cysts are rare in other organs or tissues of naturally infected animals. The tissue cyst wall is smooth and up to 4 µm thick, presumably depending on the duration of the infection. Septa are absent in the cysts and there is no secondary cyst wall (Figure 5). The tissue cyst wall is elastic, argyrophilic, has a wavy contour, and stains negative with Periodic-acid Schiff (PAS). Bradyzoites are slender and contain the same organelles as tachyzoites except that there are fewer rhoptries and more PAS-positive (amylopectin) granules in the bradyzoites. Tissue cysts may degenerate and cause an inflammatory reaction by the host (Figure 6).
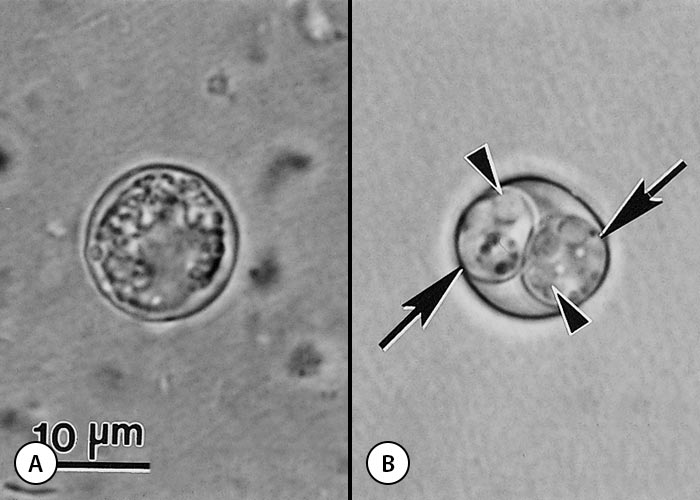
Oocysts in freshly passed faeces are unsporulated and sporulate outside the host within 24 hours. Sporulated oocysts contain two sporocysts each with four sporozoites. Neospora caninum oocysts are 10-11 µm in diameter (Figure 7) and are morphologically indistinguishable from Hammondia heydorni found in canine faeces and T. gondii and Hammondia hammondi in cat faeces.51, 153 Sporulated oocysts are slightly bigger in size than the unsporulated oocysts, namely 11.7 x 11.3 mm (10.6-12.4 x 10.6-12.0) with a length-to-width ratio of 1.04. The oocyst wall is colourless, and 0.6-0.8 mm thick. The developmental stages (ie. schizonts, gamonts) in the intestines of dogs preceding the formation of oocysts are unknown. Oocysts have been found in faeces of naturally infected dogs9, 18, 20, 21, 153 and gray wolf.61
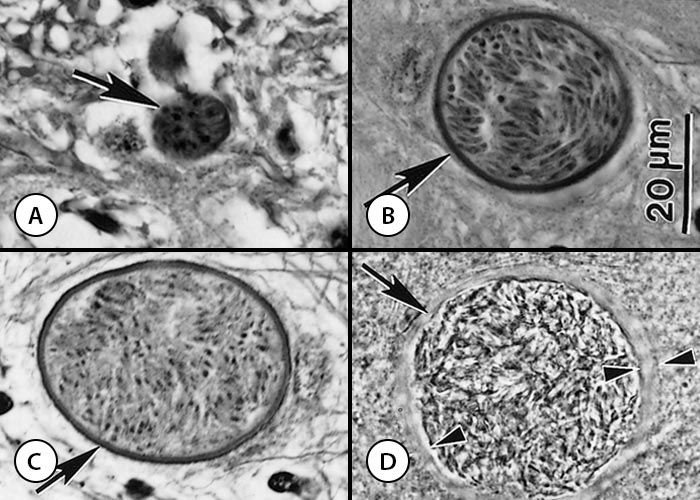
Figure 4 Tissue cysts (arrows) of Neospora caninum
- Section of a calf brain. Note small tissue cyst with thin cyst wall. Haematoxylin and eosin stain
- Intraneuronal tissue cyst in a section of the spinal cord of a calf. Haematoxylin and eosin stain
- Intraneuronal tissue cyst in a section of spinal cord of a calf. Note silver-positive cyst wall. Silver stain
- Tissue cyst in a brain squash of an experimentally infected mouse. Note thick cyst wall (opposing arrowheads). Unstained
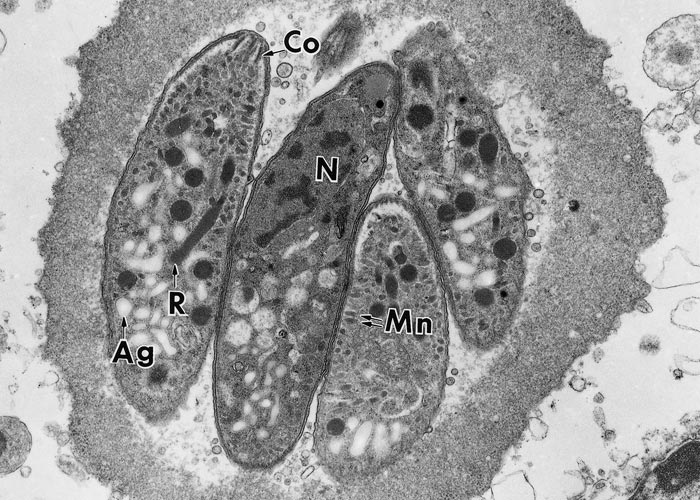
Figure 5 Transmission electron micrograph of dividing tachyzoites in two parasitophorous vacuoles (Pv). The Pv have many tubular networks. Arrows point to conoidal end of daughter tachyzoites forming inside the mother tachyzoites. Note electron-dense rhoptries (R), micronemes (Mn), microtubules (Mt) or subpellicullar tubules, conoid (C), host cell nucleus (Hcn). Cardiopulmonary artery endothelial cell culture. (Courtesy of Dr C.A. Speer, Montana State University, Bozeman, Montana, USA)
Susceptible hosts can become infected by ingesting food and water contaminated with N. caninum oocysts from canid faeces. Transplacental transmission occurs when tachyzoites from the dam cross the placenta during the second week after infection. Neospora tachyzoites initially multiply in the maternal part of the placenta and then spread to the foetal part, and eventually to foetal tissues. Transplacental transmission is common in cattle. The two terms “exogenous transplacental transmission” and “endogenous transplacental transmission” have been proposed to describe more precisely the origin of the transplacental infection of the foetus.165 “Exogenous transplacental transmission” occurs after a primary, oocyst-derived infection of a pregnant dam, while “endogenous transplacental transmission” occurs in a persistently infected dam after reactivation (recrudescence) of the infection during pregnancy.
Venereal or lactogenic transmission may occur rarely.58, 133, 167
Neospora caninum is efficiently transmitted vertically in cattle ; horizontal transmission is necessary to introduce new infections in a herd.5, 27, 48 It can be transmitted transplacentally also in naturally infected sheep, goats, deer and horses, and experimentally in several other species of animals.58 Transplacental infection can occur repeatedly in the same animal and through its progeny for several generations. The mechanisms of primary and repeat congenital transmission of infection are unknown.
Neosporosis in cattle
Neosporosis occurs in dairy and beef cattle worldwide.56, 58, 92, 138, 163, 169 It is a major cause of abortion in dairy cattle including in the USA,2-4, 13, 38, 163 New Zealand,164 Argentina,34, 127 The Netherlands,174, 176, 177 UK countries, and Australia.33, 132
Abortion is the only clinical sign observed in cattle. Cows of all ages may abort from three months of gestation to term. Most Neospora-induced abortions occur at five to six months of gestation.3, 174 Foetuses may die in utero and be resorbed, mummified or aborted, or may be stillborn, born alive but diseased, or born clinically normal but chronically infected. Isolated or sporadic abortions or abortion storms (epidemic) may occur in herds58, 121, 130, 163, 174, 178 and may occur all year round.3 Cows with N. caninum antibodies (seropositive) are more likely to abort than seronegative cows.126, 139, 174 Congenital deformities are rare:50 exophthalmia or an asymmetrical appearance of the eyes may occur.
Neospora caninum-infected calves may be born underweight, unable to rise and with neurological signs such as ataxia. Hind limbs and/or forelimbs may be flexed or hyperextended and neurological examination may reveal decreased patellar reflexes and loss of conscious proprioception.5, 16, 17, 32, 49, 53, 56, 62, 65, 84, 107, 125, 141 Neosporosis rarely occurs in calves older than two weeks old.
Neosporosis in goats and sheep
Historically, congenital neosporosis was first reported in a lamb in 197589 before the discovery of N. caninum and retrospectively confirmed as neosporosis in 1990.65 The affected lamb was born weak, partially ataxic, and died at one week of age. The most prominent findings were in the spinal cord that showed myelitis, unilateral reduction of grey matter in the ventral horn and focal cavitation, associated with intact and degenerated N. caninum tissue cysts; tachyzoites were not seen.
Neospora caninum infections are less common in goats than in cattle and sheep.58 Neosporosis-associated abortion and neonatal mortality have been reported from Brazil,42, 124, 129, 168 Costa Rica,66 Italy,72 and USA.15 The encephalomyelitis is characterized by necrosis and glial nodules with numerous tissue cysts (predominantly smaller than 20 µm in diameter); only a few tachyzoites were seen. Teratology of the brain including hydrocephalus, hypoplasia of the cerebellum, and porencephaly have also been described.
Although antibodies to N. caninum in sheep have been reported from many countries, the economic, clinical, and epidemiologic importance of N. caninum infection in sheep remains uncertain.58 In one of the largest investigations of protozoal abortion in England, there was no evidence of congenital N. caninum infection in sheep.135 In that investigation, antibodies to N. caninum were not found in pleural fluid of 179 foetuses and 141 lambs. Neospora caninum was also not identified in tissues from 281 foetuses/lambs by immunohistochemistry (IHC). In contrast, N. caninum was identified in some sheep flocks in Italy, New Zealand and Spain.79, 81, 94, 118, 128
Occasionally, N. caninum can cause abortion,7, 90, 94, 95, 105, 118, 128, 152, 166 birth of weak lambs,65, 89 neonatal mortality81 and even clinical signs in adult sheep.24
Neosporosis in dogs
Serological surveys indicate a worldwide prevalence.58 There are numerous reports of clinical neosporosis in dogs.97, 98 Clinical neosporosis is most common in young, very old, and immunosuppressed dogs.11, 96 The most severe signs are seen in transplacentally infected puppies that develop ascending rear limb paresis and paralysis, with atrophy and rigid contracture of the muscles of the pelvic limbs (Figure 8). Initial clinical signs often start in the first weeks of life but most are diagnosed when they are two months old. Infection occurs most likely prenatally. Apart from the brain, N caninum has a tropism for the lumbosacral spinal nerve roots, resulting in meningoencephalomyelitis and polyradiculoneuritis and consequently neurogenic muscle atrophy. Although stillbirths and neonatal deaths have been reported in littermates of pups with neosporosis, the youngest pups with neosporosis were eight and 12 days old.143

Figure 8 Neospora caninum infected dog with hind limb paralysis. The first isolate of N. caninum was obtained from the neural tissues of this dog in 1988. Immunohistochemical staining, however, is required to differentiate the parasite from T. gondii because tachyzoites of the two parasites are indistinguishable from each other by light microscopy. Cerebrospinal fluid should be examined for tachyzoites in cases manifesting hind limb paralysis
Adult dogs may suffer from non-generalized neosporosis and may develop a wider range of manifestations, including encephalitis or meningoencephalomyelitis. Paraparesis is the most common neurological sign, but seizures, abnormal behaviour, and vestibular dysfunction may also occur.43 Signs of myositis include stiff gait, weakness, and pain or atrophy of skeletal muscles. Almost any other organ can be involved, including the lungs, liver, eyes, and heart. Dermatitis is another common presentation of neosporosis in dogs.31, 49, 52, 64, 76, 106, 108, 111, 115, 123, 140, 142, 162 Lesions may ulcerate with a purulent discharge. Most of these cases were in aged dogs or those that are immunosuppressed, but pups can also be affected. Severe hepatitis was reported in five dogs.39, 52, 77, 93, 98
Clindamycin, sulfadiazine, and pyrimethamine alone or in combination with other drugs have been administered to treat canine neosporosis.49, 54, 64, 71, 119 In adult dogs with acute paralysis from myositis, dysfunction is often more amenable to early treatment.
Subclinically infected bitches can transmit the parasite to their foetuses, and successive litters from the same bitch may be born infected.12, 49, 55, 63 There are no drugs that can eliminate tissue cysts. Thus, prophylactic treatment of bitches to prevent transplacental infection is not beneficial.
Neosporosis in horses
Unlike other species, neosporosis in horses would seem also to be caused by another species of Neospora, N. hughesi.117 To date, N. hughesi has been reported only from horses. Morphologically, there are minor differences between N. hughesi and N. caninum, and these species are distinguished primarily by molecular characterization.58 Antigenically, N. caninum cross-reacts with N. hughesi and most serological surveys in horses used N. caninum as antigen.
Clinical neosporosis has been reported in seven horses, all from the USA.36, 44, 67, 68, 82, 88, 113, 116, 117 and one horse from Canada.36, 44, 68, 82, 88, 116, 117, 172 The first case was described in 1985 in a two-months aborted foetus. Only lung tissue was available from this case and numerous N. caninum tachyzoites were observed in the lesions.68 More recently, N. caninum infection was diagnosed by PCR in an aborted foetus in France.144
Neonatal neosporosis was diagnosed in a one-month-old Quarter horse filly in Wisconsin.113 She had circled her dam since birth and frequently tripped over obstacles. If separated from her dam she could not find her way back. Encephalitis associated with tissue cysts was revealed on histopathological examination.
An unusual case of visceral neosporosis has been reported in a 10-year-old Appaloosa mare with a history of weight loss and anaemia. Lesions associated with N. caninum tachyzoites were confined to the intestines and mesenteric lymph nodes. This is the first reported case of Neospora-associated enteritis in any animal species.82
Neosporosis has been diagnosed in an aged Pinto mare suffering from a pituitary adenoma with a history of hind limb paralysis, bizarre behaviour and masticatory difficulty.44 Lesions were present in the central nervous system, peripheral nerves and myocardium, and consisted of multifocal non-suppurative encephalomyelitis, polyradiculoneuritis and multifocal myocarditis. Neospora tachyzoites and tissue cysts were demonstrated by IHC examination in the brain, spinal cord and peripheral nerves. Neosporosis has also been diagnosed in a 20-year-old horse with a pituitary tumour.88
All of the abovementioned cases were diagnosed on post-mortem examination. Ante-mortem diagnosis was reported in four equids from California, USA.73, 74 The diagnosis was based on clinical signs, serology, and improvement with treatment with ponazuril.
Neosporosis in other animal species
Although antibodies to N. caninum have been detected in water buffalo (Bubalus bubalis) sera, and viable parasites have been isolated from adult buffaloes,147 little information is available regarding clinical neosporosis in this species. Neospora caninum DNA was detected in one out of nine foetuses tested from buffaloes slaughtered at an abattoir in Brazil,37 indicating congenital transmission. Suggestive evidence of neonatal neosporosis in buffaloes has been reported from Italy.8, 83 In the first report, encephalitis and myocarditis were found in two of four foetuses examined histologically,83 but the parasite identity was not confirmed. Additional evidence was provided in the second report describing three foetuses from a seropositive buffalo herd that were examined at necropsy: N caninum DNA was detected in the brains of all three foetuses by pNc5 gene-based PCR , all three foetuses had encephalitis and myocarditis, but N. caninum was not demonstrable.8
There are no reports of clinical neosporosis in camels (Camelus spp.) but abortion has been reported in seven of 18 llama and 12 of 32 alpacas from Peru.155, 156
White-tailed deer (Odocoileus virginianus ) are considered an important reservoir host of N. caninum in the USA.58-60 Antibodies to N. caninum were found in 162 of 400 (40,5 per cent) white-tailed deer in Illinois, USA.59 Fatal neosporosis has been diagnosed in a wild two-month-old female black-tailed deer that was found dead in California.173 Lesions and N. caninum were present histologically in sections of the lungs, liver, and kidneys. Tachyzoites were most numerous in the lungs and were associated with interstitial pneumonia, while in the kidneys interstitial nephritis associated with parasites in tubular epithelium and lumens was noted. Tachyzoites within hepatocytes and Kupffer cells were found in the liver. Congenital neosporosis was diagnosed in captive deer, namely, the fawns of Axis deer from Argentina,19 an Eld’s deer in France,69 a fallow deer in Switzerland (Dama dama)158 and three lesser kudu (Tragelaphus imberbis) in Germany. Neosporosis was also been diagnosed in a full-term stillborn brown-antlered deer (Panolia eldii siamensis) from the Paris Zoo, France:66 N. caninum tissue cysts associated with non-suppurative encephalitis were present. Neosporosis was thought to be the main disease involved in the decline of the population of this endangered species in the zoo.
Fatal neosporosis was diagnosed in a white rhinoceros in South Africa that died of massive myocardial necrosis171 (Figures 9 and 10), and another case in Thailand with severe hepatic necrosis.159 An aborted rhinoceros in Australia had extensive hepatic necrosis.150 Numerous tachyzoites were found in lesions in all three animals.
Diagnosis
In congenitally infected calves, precolostral serum collected from live or dead calves, as well as brain and spinal cord from dead calves or aborted foetuses are the specimens of choice. Ideally the entire aborted foetus should be submitted for diagnostic purposes but if this is not possible then samples from the brain, heart and liver should be collected in 10 per cent bufferedformalin for histopathological and immunohistochemical (IHC) examinations3, 112 (Figures 10 and 11), and body fluids or serum for serological evaluation.
Several serological tests can be used to detect N. caninum antibodies, including enzyme-linked immunosorbent assay (ELISA), indirect fluorescent antibody test (IFAT), and direct agglutination test.22, 26, 28, 41, 60, 102, 136, 137, 149, 154, 170, 174 Reagents for some of these tests are available commercially. An IgG avidity ELISA with the potential to discriminate between recent and chronic N. caninum infections in cattle has been developed.29, 30 The value and pitfalls of the different serological tests were recently reviewed.58
Levels of specific antibodies may persist for life but are influenced by age of the animal and stage of gestation.35 In cattle antibody levels fluctuate especially in pregnant dams and sometimes are below the detection limits.131 Recrudescence of chronic infections in cattle as a putative factor responsible for “endogenous transplacental transmission” has been substantiated by several studies that showed an increase of antibody levels during gestation.6, 41, 45, 75, 79, 85, 131, 145, 148, 161 The observation that a rise in antibody levels usually occurs in the second half of gestation79, 85, 131 is suggestive of recrudescence of infection in these cows.
Most cows that delivered a N. caninum-infected foetus or calf had antibodies against N. caninum at the time of abortion47, 134, 160, 174 or after calving.5, 46 Consequently, a negative serological test result for the dam makes it unlikely that N. caninum was the cause of abortion, stillbirth or neonatal mortality. In cases of abortion antibody levels as determined by two different ELISAs were higher in aborting than in non-aborting dams at risk.1, 60, 70, 80, 91, 103, 114, 120, 121, 145, 151, 154, 161, 169, 175, 179
Some of the commercial ELISAs have been modified for assessment of antibody levels in individual bovine milk samples or bulk milk samples.58
For the IFAT a titre 1:20060 or 1:64041 has been considered indicative of N. caninum infection in adult cattle, whereas much lower values of 1:25 or 1:80 have been selected as cut-off values for samples from bovine foetuses14 due to reduced foetal immunocompetence, especially in bovine foetuses younger than six months.176, 177
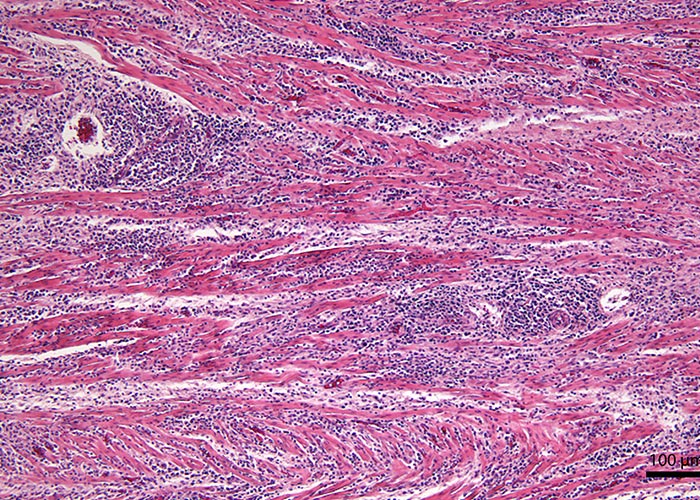
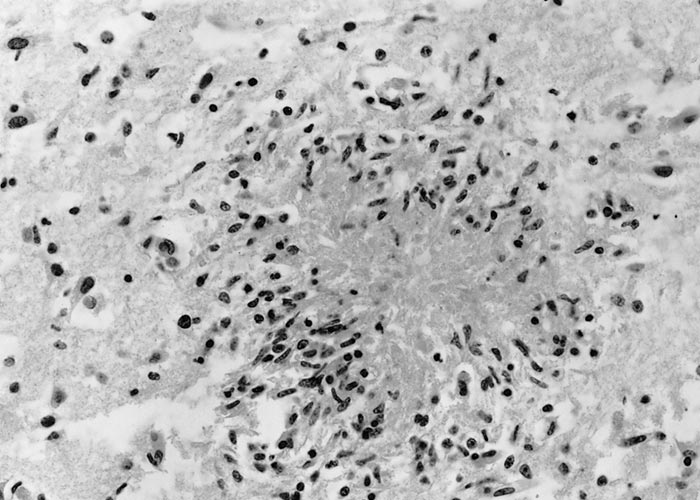
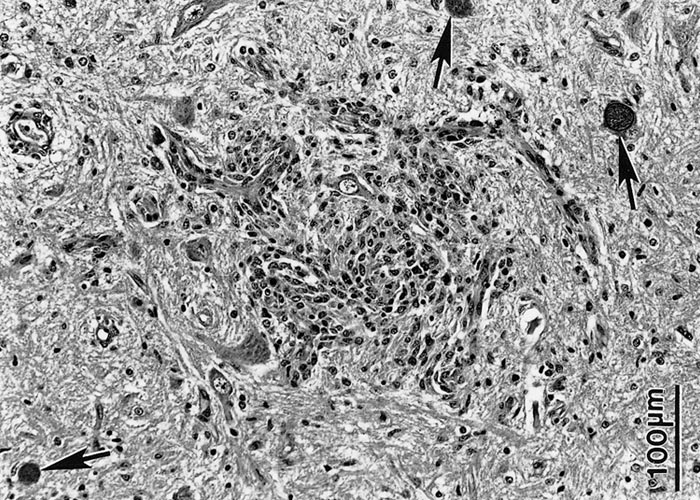
Although N. caninum infection can cause lesions in several organs, foetal brain is most consistently affected,3, 13, 58 characterized by multifocal non-suppurative encephalitis.13 (Figures 12 and 13) Scattered inflammatory foci are also common in the liver,174 heart, tongue and diaphragm and are noticeable even in autolysed foetuses. Usually only a few tachyzoites can be detected in the inflammatory foci. Lesions in the cotyledons consist of non-suppurative focal necrosis and occasional foci of mineralization. Rarely, tachyzoites are demonstrable in trophoblasts.23, 157
Although a presumptive diagnosis can be made by examination of haematoxylin and eosin (HE) stained tissue sections, IHC is necessary to detect the few N. caninum present in autolysed tissues, as these are often not visible in haematoxylin and eosin (HE) stained sections.
Isolation of Neospora in cell culture is rarely possible because most organisms in bovine foetuses die when the host cells become autolysed.40
Several polymerase chain reaction (PCR) methods are available for the detection of N. caninum DNA.58
Control
Prevention of contamination of feed and water with oocysts and not feeding raw meat/tissues to canids are important in the prevention and control of neosporosis.
There are no drugs that can eliminate tissue cysts: thus treatment of dams to prevent congenital transmission is not possible.
Different control measures including embryo transfer from infected cows to uninfected donors,10, 109 artificial insemination1, 114 and test-and-cull86, 87, 110 have been suggested to reduce the economic losses related to transplacental transmission in infected cattle herds but none seems practical.
References
- ALMERÍA, S., LÓPEZ-GATIUS, F., GARCÍA-ISPIERTO, I., NOGAREDA, C., BECH-SÀBAT, G., SERRANO, B., SANTOLARIA, P. & YÁNIZ, J. L., 2009. Effects of crossbreed pregnancies on the abortion risk of Neospora caninum-infected dairy cows. Veterinary Parasitology, 163, 323-329.
- ANDERSON, M. L., ANDRIANARIVO, A. G. & CONRAD, P. A., 2000. Neosporosis in cattle. Animal Reproduction Science, 60-61, 417-431.
- ANDERSON, M. L., BLANCHARD, P. C., BARR, B. C., DUBEY, J. P., HOFFMAN, R. L. & CONRAD, P. A., 1991. Neospora-like protozoan infection as a major cause of abortion in California dairy cattle. Journal of the American Veterinary Medical Association, 198, 241-244.
- ANDERSON, M. L., PALMER, C. W., THURMOND, M. C., PICANSO, J. P., BLANCHARD, P. C., BREITMEYER, R. E., LAYTON, A. W., MCALLISTER, M., DAFT, B., KINDE, H., READ, D. H., DUBEY, J. P., CONRAD, P. A. & BARR, B. C., 1995. Evaluation of abortions in cattle attributable to neosporosis in selected dairy herds in California. Journal of the American Veterinary Medical Association, 207, 1206-1210.
- ANDERSON, M. L., REYNOLDS, J. P., ROWE, J. D., SVERLOW, K. W., PACKHAM, A. E., BARR, B. C. & CONRAD, P. A., 1997. Evidence of vertical transmission of Neospora sp. infection in dairy cattle. Journal of the American Veterinary Medical Association, 210, 1169-1172.
- ANDRIANARIVO, A. G., ANDERSON, M. L., ROWE, J. D., GARDNER, I. A., REYNOLDS, J. P., CHOROMANSKI, L. & CONRAD, P. A., 2005. Immune responses during pregnancy in heifers naturally infected with Neospora caninum with and without immunization. Parasitology Research, 96, 24-31.
- ASADPOUR, R., JAFARI-JOOZANI, R. & SALEHI, N., 2013. Detection of Neospora caninum in ovine abortion in Iran. Journal of Parasitic Diseases, 37, 105-109.
- AURIEMMA, C., LUCIBELLI, M. G., BORRIELLO, G., DE CARLO, E., MARTUCCIELLO, A., SCHIAVO, L., GALLO, A., BOVE, F., CORRADO, F., GIRARDI, S., AMOROSO, M. G., DEGLI UBERTI, B. & GALIERO, G., 2014. PCR detection of Neospora caninum in water buffalo foetal tissues. Acta Parasitologica, 59, 1-4.
- BACIGALUPE, D., BASSO, W., CASPE, S. G., MORÉ, G., LISCHINSKY, L., GOS, M. L., LEUNDA, M., CAMPERO, L., MOORE, D. P., SCHARES, G., CAMPERO, C. M. & VENTURINI, M. C., 2013. Neospora caninum NC-6 Argentina induces fetopathy in both serologically positive and negative experimentally inoculated pregnant dams. Parasitology Research, 112, 2585-2592.
- BAILLARGEON, P., FECTEAU, G., PARÉ, J., LAMOTHE, P. & SAUVÉ, R., 2001. Evaluation of the embryo transfer procedure proposed by the International Embryo Transfer Society as a method of controlling vertical transmission of Neospora caninum in cattle. Journal of the American Veterinary Medical Association, 218, 1803-1806.
- BARBER, J. S. & TREES, A. J., 1996. Clinical aspects of 27 cases of neosporosis in dogs. The Veterinary Record, 139, 439-443.
- BARBER, J. S. & TREES, A. J., 1998. Naturally occurring vertical transmission of Neospora caninum in dogs. International Journal for Parasitology, 28, 57-64.
- BARR, B. C., ANDERSON, M. L., DUBEY, J. P. & CONRAD, P. A., 1991. Neospora-like protozoal infections associated with bovine abortions. Veterinary Pathology, 28, 110-116.
- BARR, B. C., ANDERSON, M. L., SVERLOW, K. W. & CONRAD, P. A., 1995. Diagnosis of bovine fetal Neospora infection with an indirect fluorescent antibody test. The Veterinary Record, 137, 611-613.
- BARR, B. C., ANDERSON, M. L., WOODS, L. W., DUBEY, J. P. & CONRAD, P. A., 1992. Neospora-like protozoal infections associated with abortion in goats. Journal of Veterinary Diagnostic Investigation, 4, 365-367.
- BARR, B. C., CONRAD, P. A., BREITMEYER, R., SVERLOW, K., ANDERSON, M. L., REYNOLDS, J., CHAUVET, A. E., DUBEY, J. P. & ARDANS, A. A., 1993. Congenital Neospora infection in calves born from cows that had previously aborted Neospora-infected fetuses: four cases (1990-1992). Journal of the American Veterinary Medical Association, 202, 113-117.
- BARR, B. C., CONRAD, P. A., DUBEY, J. P. & ANDERSON, M. L., 1991. Neospora-like encephalomyelitis in a calf: pathology, ultrastructure, and immunoreactivity. Journal of Veterinary Diagnostic Investigation, 3, 39-46.
- BASSO, W., HERRMANN, D. C., CONRATHS, F. J., PANTCHEV, N., VRHOVEC, M. G. & SCHARES, G., 2009. First isolation of Neospora caninum from the faeces of a dog from Portugal. Veterinary Parasitology, 159, 162-166.
- BASSO, W., MORÉ, G., QUIROGA, M. A., BALDUCCHI, D., SCHARES, G. & VENTURINI, M. C., 2014. Neospora caninum is a cause of perinatal mortality in axis deer (Axis axis). Veterinary Parasitology, 199, 255-258.
- BASSO, W., SCHARES, S., BÄRWALD, A., HERRMANN, D. C., CONRATHS, F. J., PANTCHEV, N., VRHOVEC, M. G. & SCHARES, G., 2009. Molecular comparison of Neospora caninum oocyst isolates from naturally infected dogs with cell culture-derived tachyzoites of the same isolates using nested polymerase chain reaction to amplify microsatellite markers. Veterinary Parasitology, 160, 43-50.
- BASSO, W., VENTURINI, L., VENTURINI, M. C., HILL, D. E., KWOK, O. C. H., SHEN, S. K. & DUBEY, J. P., 2001. First isolation of Neospora caninum from the feces of a naturally infected dog. Journal of Parasitology, 87, 612-618.
- BASZLER, T. V., KNOWLES, D. P., DUBEY, J. P., GAY, J. M., MATHISON, B. A. & MCELWAIN, T. F., 1996. Serological diagnosis of bovine neosporosis by Neospora caninum monoclonal antibody-based competitive inhibition enzyme-linked immunosorbent assay. Journal of Clinical Microbiology, 34, 1423-1428.
- BERGERON, N., GIRARD, C., PARÉ, J., FECTEAU, G., ROBINSON, J. & BAILLARGEON, P., 2001. Rare detection of Neospora caninum in placentas from seropositive dams giving birth to full-term calves. Journal of Veterinary Diagnostic Investigation, 13, 173-175.
- BISHOP, S., KING, J., WINDSOR, P., REICHEL, M. P., ELLIS, J. & ŠLAPETA, J., 2010. The first report of ovine cerebral neosporosis and evaluation of Neospora caninum prevalence in sheep in New South Wales. Veterinary Parasitology, 170, 137-142.
- BJERKÅS, I., MOHN, S. F. & PRESTHUS, J., 1984. Unidentified cyst-forming sporozoon causing encephalomyelitis and myositis in dogs. Zeitschrift für Parasitenkunde, 70, 271-274.
- BJÖRKMAN, C., HOLMDAHL, O. J. M. & UGGLA, A., 1997. An indirect enzyme-linked immunoassay (ELISA) for demonstration of antibodies to Neospora caninum in serum and milk of cattle. Veterinary Parasitology, 68, 251-260.
- BJÖRKMAN, C., JOHANSSON, O., STENLUND, S., HOLMDAHL, O. J. M. & UGGLA, A., 1996. Neospora species infection in a herd of dairy cattle. Journal of the American Veterinary Medical Association, 208, 1441-1444.
- BJÖRKMAN, C., LUNDÉN, A., HOLMDAHL, J., BARBER, J., TREES, A. J. & UGGLA, A., 1994. Neospora caninum in dogs: detection of antibodies by ELISA using an iscom antigen. Parasite Immunology, 16, 643-648.
- BJÖRKMAN, C., NÄSLUND, K., STENLUND, S., MALEY, S. W., BUXTON, D. & UGGLA, A., 1999. An IgG avidity ELISA to discriminate between recent and chronic Neospora caninum infection. Journal of Veterinary Diagnostic Investigation, 11, 41-44.
- BJÖRKMAN, C. & UGGLA, A., 1999. Serological diagnosis of Neospora caninum infection. International Journal for Parasitology, 29, 1497-1507.
- BOYD, S. P., BARR, P. A., BROOKS, H. W. & ORR, J. P., 2005. Neosporosis in a young dog presenting with dermatitis and neuromuscular signs. Journal of Small Animal Practice, 46, 85-88.
- BRYAN, L. A., GAJADHAR, A. A., DUBEY, J. P. & HAINES, D. M., 1994. Bovine neonatal encephalomyelitis associated with a Neospora sp. protozoan. Canadian Veterinary Journal, 35, 111-113.
- BUXTON, D., CALDOW, G. L., MALEY, S. W., MARKS, J. & INNES, E. A., 1997. Neosporosis and bovine abortion in Scotland. Veterinary Record, 141, 649-651.
- CAMPERO, C. M., MOORE, D. P., ODEÓN, A. C., CIPOLLA, A. L. & ODRIOZOLA, E., 2003. Aetiology of bovine abortion in Argentina. Veterinary Research Communications, 27, 359-369.
- CARDOSO, J. M. S., NISHI, S. M., FUNADA, M. R., AMAKU, M., GUIMARÃES, J. S. & GENNARI, S. M., 2009. Antibody dynamics during gestation in cows naturally infected with Neospora caninum from four dairy herds in Brazil. Brazilian Journal of Veterinary Research and Animal Science, 46, 395-399.
- CHEADLE, M. A., LINDSAY, D. S., ROWE, S., DYKSTRA, C. C., WILLIAMS, M. A., SPENCER, J. A., TOIVIO-KINNUCAN, M. A., LENZ, S. D., NEWTON, J. C., ROLSMA, M. D. & BLAGBURN, B. L., 1999. Prevalence of antibodies to Neospora sp. in horses from Alabama and characterisation of an isolate recovered from a naturally infected horse. International Journal for Parasitology, 29, 1537-1543.
- CHRYSSAFIDIS, A. L., SOARES, R. M., RODRIGUES, A. A. R., CARVALHO, N. A. T. & GENNARI, S. M., 2011. Evidence of congenital transmission of Neospora caninum in naturally infected water buffalo (Bubalus bubalis) fetus from Brazil. Parasitology Research, 108, 741-743.
- CLOTHIER, K. & ANDERSON, M., 2015. Evaluation of bovine abortion cases and tissue suitbility for identification of infectious agents in California diagnostic laboatory cases from 2007 to 2012. Theriogenology, 85, 933-938.
- COCHRANE, S. M. & DUBEY, J. P., 1993. Neosporosis in a golden retriever dog from Ontario. Canadian Veterinary Journal, 34, 232-233.
- CONRAD, P. A., BARR, B. C., SVERLOW, K. W., ANDERSON, M., DAFT, B., KINDE, H., DUBEY, J. P., MUNSON, L. & ARDANS, A., 1993. In vitro isolation and characterization of a Neospora sp. from aborted bovine foetuses. Parasitology, 106, 239-249.
- CONRAD, P. A., SVERLOW, K., ANDERSON, M., ROWE, J., BONDURANT, R., TUTER, G., BREITMEYER, R., PALMER, C., THURMOND, M., ARDANS, A., DUBEY, J. P., DUHAMEL, G. & BARR, B., 1993. Detection of serum antibody responses in cattle with natural or experimental Neospora infections. Journal of Veterinary Diagnostic Investigation, 5, 572-578.
- CORBELLINI, L. G., COLODEL, E. M. & DRIEMEIER, D., 2001. Granulomatous encephalitis in a neurologically impaired goat kid associated with degeneration of Neospora caninum tissue cysts. Journal of Veterinary Diagnostic Investigation, 13, 416-419.
- CUDDON, P., LIN, D. S., BOWMAN, D. D., LINDSAY, D. S., MILLER, T. K., DUNCAN, I. D., DELAHUNTA, A., CUMMINGS, J., SUTER, M., COOPER, B., KING, J. M. & DUBEY, J. P., 1992. Neospora caninum infection in English Springer spaniel littermates: Diagnostic evaluation and organism isolation. Journal of Veterinary Internal Medicine, 6, 325-332.
- DAFT, B. M., BARR, B. C., COLLINS, N. & SVERLOW, K., 1996. Neospora encephalomyelitis and polyradiculoneuritis in an aged mare with Cushing's disease. Equine Veterinary Journal, 28, 240-243.
- DANNATT, L., 1997. Neospora caninum antibody levels in an endemically-infected dairy herd. Cattle Practice, 5, 335-337.
- DAVISON, H. C., OTTER, A. & TREES, A. J., 1999. Estimation of vertical and horizontal transmission parameters of Neospora caninum infections in dairy cattle. International Journal for Parasitology, 29, 1683-1689.
- DE MEERSCHMAN, F., SPEYBROECK, N., BERKVENS, D., RETTIGNER, C., FOCANT, C., LECLIPTEUX, T., CASSART, D. & LOSSON, B., 2002. Fetal infection with Neospora caninum in dairy and beef cattle in Belgium. Theriogenology, 58, 933-945.
- DIJKSTRA, T., 2002. Horizontal and vertical transmission of Neospora caninum. PhD Universiteit Utrecht, Utrecht, Belgium, 1-140.
- DUBEY, J. P., 2013. Neosporosis in dogs. Commonwealth Agricultural Bureau Reviews, 8, 055.
- DUBEY, J. P., ABBITT, B., TOPPER, M. J. & EDWARDS, J. F., 1998. Hydrocephalus associated with Neospora caninum-infection in an aborted bovine fetus. Journal of Comparative Pathology, 118, 169-173.
- DUBEY, J. P., BARR, B. C., BARTA, J. R., BJERKÅS, I., BJÖRKMAN, C., BLAGBURN, B. L., BOWMAN, D. D., BUXTON, D., ELLIS, J. T., GOTTSTEIN, B., HEMPHILL, A., HILL, D. E., HOWE, D. K., JENKINS, M. C., KOBAYASHI, Y., KOUDELA, B., MARSH, A. E., MATTSSON, J. G., MCALLISTER, M. M., MODRÝ, D., OMATA, Y., SIBLEY, L. D., SPEER, C. A., TREES, A. J., UGGLA, A., UPTON, S. J., WILLIAMS, D. J. L. & LINDSAY, D. S., 2002. Redescription of Neospora caninum and its differentiation from related coccidia. International Journal for Parasitology, 32, 929-946.
- DUBEY, J. P., CARPENTER, J. L., SPEER, C. A., TOPPER, M. J. & UGGLA, A., 1988. Newly recognized fatal protozoan disease of dogs. Journal of the American Veterinary Medical Association, 192, 1269-1285.
- DUBEY, J. P. & DE LAHUNTA, A., 1993. Neosporosis associated congenital limb deformities in a calf. Applied Parasitology, 34, 229-233.
- DUBEY, J. P., DOROUGH, K. R., JENKINS, M. C., LIDDELL, S., SPEER, C. A., KWOK, O. C. H. & SHEN, S. K., 1998. Canine neosporosis: clinical signs, diagnosis, treatment and isolation of Neospora caninum in mice and cell culture. International Journal for Parasitology, 28, 1293-1304.
- DUBEY, J. P., HARTLEY, W. J. & LINDSAY, D. S., 1990. Congenital Neospora caninum infection in a calf with spinal cord anomaly. Journal of the American Veterinary Medical Association, 197, 1043-1044.
- DUBEY, J. P., HARTLEY, W. J., LINDSAY, D. S. & TOPPER, M. J., 1990. Fatal congenital Neospora caninum infection in a lamb. Journal of Parasitology, 76, 127-130.
- DUBEY, J. P., HATTEL, A. L., LINDSAY, D. S. & TOPPER, M. J., 1988. Neonatal Neospora caninum infection in dogs: Isolation of the causative agent and experimental transmission. Journal of the American Veterinary Medical Association, 193, 1259-1263.
- DUBEY, J. P., HEMPHILL, A., CALERO-BERNAL, R. & SCHARES, G., 2017. Neosporosis in animals. CRC Press, Boca Raton, Florida, 1-529.
- DUBEY, J. P., HOLLIS, K., ROMAND, S., THULLIEZ, P., KWOK, O. C. H., HUNGERFORD, L., ANCHOR, C. & ETTER, D., 1999. High prevalence of antibodies to Neospora caninum in white-tailed deer (Odocoileus virginianus). International Journal for Parasitology, 29, 1709-1711.
- DUBEY, J. P., JENKINS, M. C., ADAMS, D. S., MCALLISTER, M. M., ANDERSON-SPRECHER, R., BASZLER, T. V., KWOK, O. C. H., LALLY, N. C., BJÖRKMAN, C. & UGGLA, A., 1997. Antibody responses of cows during an outbreak of neosporosis evaluated by indirect fluorescent antibody test and different enzyme-linked immunosorbent assays. Journal of Parasitology, 83, 1063-1069.
- DUBEY, J. P., JENKINS, M. C., RAJENDRAN, C., MISKA, K., FERREIRA, L. R., MARTINS, J., KWOK, O. C. H. & CHOUDHARY, S., 2011. Gray wolf (Canis lupus) is a natural definitive host for Neospora caninum. Veterinary Parasitology, 181, 382-387.
- DUBEY, J. P., KOESTNER, A. & PIPER, R. C., 1990. Repeated transplacental transmission of Neospora caninum in dogs. Journal of the American Veterinary Medical Association, 197, 857-860.
- DUBEY, J. P. & LINDSAY, D. S., 1996. A review of Neospora caninum and neosporosis. Veterinary Parasitology, 67, 1-59.
- DUBEY, J. P., METZGER, F. L. J., HATTEL, A. L., LINDSAY, D. S. & FRITZ, D. L., 1995. Canine cutaneous neosporosis: clinical improvement with clindamycin. Veterinary Dermatology, 6, 37-43.
- DUBEY, J. P., MILLER, S., LINDSAY, D. S. & TOPPER, M. J., 1990. Neospora caninum-associated myocarditis and encephalitis in an aborted calf. Journal of Veterinary Diagnostic Investigation, 2, 66-69.
- DUBEY, J. P., MORALES, J. A., VILLALOBOS, P., LINDSAY, D. S., BLAGBURN, B. L. & TOPPER, M. J., 1996. Neosporosis-associated abortion in a dairy goat. Journal of the American Veterinary Medical Association, 208, 263-265.
- DUBEY, J. P. & PORTERFIELD, J. S., 1986.
- DUBEY, J. P. & PORTERFIELD, M. L., 1990. Neospora caninum (Apicomplexa) in an aborted equine fetus. Journal of Parasitology, 76, 732-734.
- DUBEY, J. P., RIGOULET, J., LAGOURETTE, P., GEORGE, C., LONGEART, L. & LENET, J. L., 1996. Fatal transplacental neosporosis in a deer (Cervus eldi siamensis). Journal of Parasitology, 82, 338-339.
- DUBEY, J. P. & SCHARES, G., 2006. Diagnosis of bovine neosporosis. Veterinary Parasitology, 140, 1-34.
- DUBEY, J. P., SREEKUMAR, C., KNICKMAN, E., MISKA, K. B., VIANNA, M. C. B., KWOK, O. C. H., HILL, D. E., JENKINS, M. C., LINDSAY, D. S. & GREENE, C. E., 2004. Biologic, morphologic, and molecular characterisation of Neospora caninum isolates from littermate dogs. International Journal for Parasitology, 34, 1157-1167.
- ELENI, C., CROTTI, S., MANUALI, E., COSTARELLI, S., FILIPPINI, G., MOSCATI, L. & MAGNINO, S., 2004. Detection of Neospora caninum in an aborted goat foetus. Veterinary Parasitology, 123, 271-274.
- FINNO, C. J., ALEMAN, M. & PUSTERLA, N., 2007. Equine protozoal myeloencephalitis associated with neosporosis in 3 horses. Journal of Veterinary Internal Medicine, 21, 1405-1408.
- FINNO, C. J., EATON, J. S., ALEMAN, M. & HOLLINGSWORTH, S. R., 2010. Equine protozoal myeloencephalitis due to Neospora hughesi and equine motor neuron disease in a mule. Veterinary Ophthalmology, 13, 259-265.
- FIORETTI, D. P., PASQUAI, P., DIAFERIA, M., MANGILI, V. & ROSIGNOLI, L., 2003. Neospora caninum infection and congenital transmission: serological and parasitological study of cows up to the fourth gestation. Journal of Veterinary Medicine B, 50, 399-404.
- FRITZ, D., GEORGE, C., DUBEY, J. P., TREES, A. J., BARBER, J. S., HOPFNER, C. L., MEHAUT, S., LE NET, J. L. & LONGEART, L., 1997. Neospora caninum: associated nodular dermatitis in a middle-aged dog. Canine Practice, 22, 21-24.
- FRY, D. R., MCSPORRAN, K. D., ELLIS, J. T. & HARVEY, C., 2009. Protozoal hepatitis associated with immunosuppressive therapy in a dog. Journal of Veterinary Internal Medicine, 23, 366-368.
- GONDIM, L. F. P., MCALLISTER, M. M., PITT, W. C. & ZEMLICKA, D. E., 2004. Coyotes (Canis latrans) are definitive hosts of Neospora caninum. International Journal for Parasitology, 34, 159-161.
- GONZÁLEZ-WARLETA, M., CASTRO-HERMIDA, J. A., CARRO-CORRAL, C., CORTIZO-MELLA, J. & MEZO, M., 2008. Epidemiology of neosporosis in dairy cattle in Galicia (NW Spain). Parasitology Research, 102, 243-249.
- GONZÁLEZ-WARLETA, M., CASTRO-HERMIDA, J. A., CARRO-CORRAL, C. & MEZO, M., 2011. Anti-Neospora caninum antibodies in milk in relation to production losses in dairy cattle. Preventive Veterinary Medicine, 101, 58-64.
- GONZÁLEZ-WARLETA, M., CASTRO-HERMIDA, J. A., REGIDOR-CERRILLO, J., BENAVIDES, J., ÁLVAREZ-GARCÍA, G., FUERTES, M., ORTEGA-MORA, L. M. & MEZO, M., 2014. Neospora caninum infection as a cause of reproductive failure in a sheep flock. Veterinary Research, 45, 88.
- GRAY, M. L., HARMON, B. G., SALES, L. & DUBEY, J. P., 1996. Visceral neosporosis in a 10-year-old horse. Journal of Veterinary Diagnostic Investigation, 8, 130-133.
- GUARINO, A., FUSCO, G., SAVINI, G., DI FRANCESCO, G. & CRINGOLI, G., 2000. Neosporosis in water buffalo (Bubalus bubalis) in southern Italy. Veterinary Parasitology, 91, 15-21.
- GUNNING, R. F., GUMBRELL, R. C. & JEFFREY, M., 1994. Neospora infection and congenital ataxia in calves. Veterinary Record, 134, 558.
- GUY, C. S., WILLIAMS, D. J. L., KELLY, D. F., MCGARRY, J. W., GUY, F., BJÖRKMAN, C., SMITH, R. F. & TREES, A. J., 2001. Neospora caninum in persistently infected, pregnant cows: spontaneous transplacental infection is associated with an acute increase in maternal antibody. Veterinary Record, 149, 443-449.
- HADDAD, J. P. A., DOHOO, I. R. & VANLEEWEN, J. A., 2005. A review of Neospora caninum in dairy and beef cattle - a Canadian perspective. Canadian Veterinary Journal, 46, 230-243.
- HALL, C. A., REICHEL, M. P. & ELLIS, J. T., 2005. Neospora abortions in dairy cattle: diagnosis, mode of transmission and control. Veterinary Parasitology, 128, 231-241.
- HAMIR, A. N., TORNQUIST, S. J., GERROS, T. C., TOPPER, M. J. & DUBEY, J. P., 1998. Neospora caninum-associated equine protozoal myeloencephalitis. Veterinary Parasitology, 79, 269-274.
- HARTLEY, W. J. & BRIDGE, P. S., 1975. A case of suspected congenital Toxoplasma encephalomyelitis in a lamb associated with a spinal cord anomaly. British Veterinary Journal, 131, 380-384.
- HÄSSIG, M., SAGER, H., REITT, K., ZIEGLER, D., STRABEL, D. & GOTTSTEIN, B., 2003. Neospora caninum in sheep: a herd case report. Veterinary Parasitology, 117, 213-220.
- HERNANDEZ, J., RISCO, C. & DONOVAN, A., 2002. Risk of abortion associated with Neospora caninum during different lactations and evidence of congenital transmission in dairy cows. Journal of the American Veterinary Medical Association, 221, 1742-1746.
- HOAR, B. R., RIBBLE, C. S., SPITZER, C. C., SPITZER, P. G. & JANZEN, E. D., 1996. Investigation of pregnancy losses in beef cattle herds associated with Neospora sp. infection. Canadian Veterinary Journal, 37, 364-366.
- HOON-HANKS, L. L., REGAN, D., DUBEY, J. P., PORTER, M. C. & DUNCAN, C. G., 2013. Hepatic neosporosis in a dog treated for pemphigus foliaceus. Journal of Veterinary Diagnostic Investigation, 25, 807-810.
- HOWE, L., COLLETT, M. G., PATTISON, R. S., MARSHALL, J., WEST, D. M. & POMROY, W. E., 2012. Potential involvement of Neospora caninum in naturally occurring ovine abortions in New Zealand. Veterinary Parasitology, 185, 64-71.
- HOWE, L., WEST, D. M., COLLETT, M. G., TATTERSFIELD, G., PATTISON, R. S., POMROY, W. E., KENYON, P. R., MORRIS, S. T. & WILLIAMSON, N. B., 2008. The role of Neospora caninum in three cases of unexplained ewe abortions in the southern North Island of New Zealand. Small Ruminant Research, 75, 115-122.
- JACKSON, W., DE LAHUNTA, A., ADASKA, J., COOPER, B. & DUBEY, J. P., 1995. Neospora caninum in an adult dog with progressive cerebellar signs. Progress in Veterinary Neurology, 6, 124-127.
- JACOBSON, L. S. & JARDINE, J. E., 1993. Neospora caninum infection in three Labrador littermates. Journal of the South African Veterinary Association, 64, 47-51.
- JARDINE, J. E. & DUBEY, J. P., 1992. Canine neosporosis in South Africa. Veterinary Parasitology, 44, 291-294.
- JARDINE, J. E. & LAST, R. D., 1993. Neospora caninum in aborted twin calves. Journal of the South African Veterinary Association, 64, 101-102.
- JARDINE, J. E. & LAST, R. D., 1995. The prevalence of neosporosis in aborted bovine foetuses submitted to the Allerton Regional Veterinary Laboratory. Onderstepoort Journal of Veterinary Research, 62, 207-209.
- JARDINE, J. E. & WELLS, B. H., 1995. Bovine neosporosis in Zimbabwe. The Veterinary Record, 137, 223.
- JENKINS, M. C., WOUDA, W. & DUBEY, J. P., 1997. Serological response over time to recombinant Neospora caninum antigens in cattle after a neosporosis-induced abortion. Clinical and Diagnostic Laboratory Immunology, 4, 270-274.
- KASHIWAZAKI, Y., GIANNEECHINI, R. E., LUST, M. & GIL, J., 2004. Seroepidemiology of neosporosis in dairy cattle in Uruguay. Veterinary Parasitology, 120, 139-144.
- KING, J. S., ŠLAPETA, J., JENKINS, D. J., AL-QASSAB, S. E., ELLIS, J. T. & WINDSOR, P. A., 2010. Australian dingoes are definitive host of Neospora caninum. International Journal for Parasitology, 40, 945-950.
- KOBAYASHI, Y., YAMADA, M., OMATA, Y., KOYAMA, T., SAITO, A., MATSUDA, T., OKUYAMA, K., FUJIMOTO, S., FURUOKA, H. & MATSUI, T., 2001. Naturally-occurring Neospora caninum infection in an adult sheep and her twin fetuses. Journal of Parasitology, 87, 434-436.
- KOUDELA, B., SVOBODA, M., BJÖRKMAN, C. & UGGLA, A., 1998. Neosporosis in dogs: the first case report in the Czech Republic. Veterinarni Medicina, 43, 51-54.
- KUL, O., KABAKCI, N., YILDIZ, K., ÖCAL, N., KALENDER, H. & ILKME, N. A., 2009. Neospora caninum associated with epidemic abortions in dairy cattle: the first clinical neosporosis report in Turkey. Veterinary Parasitology, 159, 69-72.
- LA PERLE, K. M. D., DEL PIERO, F., CARR, R. F., HARRIS, C. & STROMBERG, P. C., 2001. Cutaneous neosporosis in two adult dogs on chronic immunosuppressive therapy. Journal of Veterinary Diagnostic Investigation, 13, 252-255.
- LANDMANN, J. K., JILLELLA, D., O'DONOGHUE, P. J. & MCGOWAN, M. R., 2002. Confirmation of the prevention of vertical transmission of Neospora caninum in cattle by the use of embryo transfer. Australian Veterinary Journal, 80, 502-503.
- LARSON, R. L., HARDIN, D. K. & PIERCE, V. L., 2004. Economic considerations for diagnostic and control options for Neospora caninum-induced abortions in endemically infected herds of beef cattle. Journal of the American Veterinary Medical Association, 224, 1597-1604.
- LEGNANI, S., PANTCHEV, N., FORLANI, A., ZINI, E., SCHARES, G., BALZER, J., ROCCABIANCA, P., FERRI, F. & ZANNA, G., 2016. Emergence of cutaneous neosporosis in a dog receiving immunosuppressive therapy: molecular identification and management. Veterinary Dermatology, 27, 49-e14.
- LINDSAY, D. S. & DUBEY, J. P., 1989. Immunohistochemical diagnosis of Neospora caninum in tissue sections. American Journal of Veterinary Research, 50, 1981-1983.
- LINDSAY, D. S., STEINBERG, H., DUBIELZIG, R. R., SEMRAD, S. D., KONKLE, D. M., MILLER, P. E. & BLAGBURN, B. L., 1996. Central nervous system neosporosis in a foal. Journal of Veterinary Diagnostic Investigation, 8, 507-510.
- LÓPEZ-GATIUS, F., SANTOLARIA, P., YÁNIZ, J. L., GARBAYO, J. M. & ALMERÍA, S., 2005. The use of beef bull semen reduced the risk of abortion in Neospora-seropositive dairy cows. Journal of Veterinary Medicine B, 52, 88-92.
- MANN, T. R., CADORE, G. C., CAMILLO, G., VOGEL, F. S. F., SCHMIDT, C. & ANDRADE, C. M., 2016. Canine cutaneous neosporosis in Brazil. Veterinary Dermatology, 27, 195-197.
- MARSH, A. E., BARR, B. C., MADIGAN, J., LAKRITZ, J., NORDHAUSEN, R. & CONRAD, P. A., 1996. Neosporosis as a cause of equine protozoal myeloencephalitis. Journal of the American Veterinary Medical Association, 209, 1907-1913.
- MARSH, A. E., BARR, B. C., PACKHAM, A. E. & CONRAD, P. A., 1998. Description of a new Neospora species (Protozoa: Apicomplexa: Sarcocystidae). Journal of Parasitology, 84, 983-991.
- MASALA, G., PORCU, R., DAGA, C., DENTI, S., CANU, G., PATTA, C. & TOLA, S., 2007. Detection of pathogens in ovine and caprine abortion samples from Sardinia, Italy, by PCR. Journal of Veterinary Diagnostic Investigation, 19, 96-98.
- MAYHEW, I. G., SMITH, K. C., DUBEY, J. P., GATWARD, L. K. & MCGLENNON, N. J., 1991. Treatment of encephalomyelitis due to Neospora caninum in a litter of puppies. Journal of Small Animal Practice, 32, 609-612.
- MAZUZ, M. L., FISH, L., REZNIKOV, D., WOLKOMIRSKY, R., LEIBOVITZ, B., SAVITZKY, I., GOLENSER, J. & SHKAP, V., 2014. Neosporosis in naturally infected pregnant dairy cattle. Veterinary Parasitology, 205, 85-91.
- MCALLISTER, M., HUFFMAN, E. M., HIETALA, S. K., CONRAD, P. A., ANDERSON, M. L. & SALMAN, M. D., 1996. Evidence suggesting a point source exposure in an outbreak of bovine abortion due to neosporosis. Journal of Veterinary Diagnostic Investigation, 8, 355-357.
- MCALLISTER, M. M., DUBEY, J. P., LINDSAY, D. S., JOLLEY, W. R., WILLS, R. A. & MCGUIRE, A. M., 1998. Dogs are definitive hosts of Neospora caninum. International Journal for Parasitology, 28, 1473-1478.
- MCINNES, L. M., IRWIN, P., PALMER, D. G. & RYAN, U. M., 2006. In vitro isolation and characterisation of the first canine Neospora caninum isolate in Australia. Veterinary Parasitology, 137, 355-363.
- MESQUITA, L. P., NOGUEIRA, C. I., COSTA, R. C., ORLANDO, D. R., BRUHN, F. R., LOPES, P. F. R., NAKAGAKI, K. Y. R., PECONICK, A. P., SEIXAS, J. N., BEZERRA JÚNIOR, P. S., RAYMUNDO, D. L. & VARASCHIN, M. S., 2013. Antibody kinetics in goats and conceptuses naturally infected with Neospora caninum. Veterinary Parasitology, 196, 327-333.
- MICHELOUD, J. F., MOORE, D. P., CANAL, A. M., LISCHINSKY, L., HECKER, Y. P., CANTON, G. J., ODRIOZOLA, E., ODEON, A. C. & CAMPERO, C. M., 2015. First report of congenital Neospora caninum encephalomyelitis in two newborn calves in the Argentinean Pampas. J. Vet. Sci. Technol, 6, 251.
- MOEN, A. R., WOUDA, W., MUL, M. F., GRAAT, E. A. M. & VAN WERVEN, T., 1998. Increased risk of abortion following Neospora caninum abortion outbreaks: a retrospective and prospective cohort study in four dairy herds. Theriogenology, 49, 1301-1309.
- MOORE, D. P., CAMPERO, C. M., ODEÓN, A. C., CHAYER, R. & BIANCO, M. A., 2003. Reproductive losses due to Neospora caninum in a beef herd in Argentina. Journal of Veterinary Medicine B, 50, 304-308.
- MORENO, B., COLLANTES-FERNÁNDEZ, E., VILLA, A., NAVARRO, A., REGIDOR-CERRILLO, J. & ORTEGA-MORA, L. M., 2012. Occurrence of Neospora caninum and Toxoplasma gondii infections in ovine and caprine abortions. Veterinary Parasitology, 187, 312-318.
- NAKAGAKI, K. Y. R., ABREU, C. C., COSTA, R. C., ORLANDO, D. R., FREIRE, L. R., BRUHN, F. R. P., PECONICK, A. P., WOUTERS, F., WOUTERS, A. T. B., RAYMUNDO, D. L. & VARASCHIN, M. S., 2016. Lesions and distribution of Neospora caninum in tissues of naturally infected female goats. Small Ruminant Research, 140, 57-62.
- NIETFELD, J. C., DUBEY, J. P., ANDERSON, M. L., LIBAL, M. C., YAEGER, M. J. & NEIGER, R. D., 1992. Neospora-like protozoan infection as a cause of abortion in dairy cattle. Journal of Veterinary Diagnostic Investigation, 4, 223-226.
- NOGAREDA, C., LÓPEZ-GATIUS, F., SANTOLARIA, P., GARCÍA-ISPIERTO, I., BECH-SÀBAT, G., PABÓN, M., MEZO, M., GONZALEZ-WARLETA, M., CASTRO-HERMIDA, J. A., YÁNIZ, J. & ALMERIA, S., 2007. Dynamics of anti-Neospora caninum antibodies during gestation in chronically infected dairy cows. Veterinary Parasitology, 148, 193-199.
- OBENDORF, D. L., MURRAY, N., VELDHUIS, G., MUNDAY, B. L. & DUBEY, J. P., 1995. Abortion caused by neosporosis in cattle. Australian Veterinary Journal, 72, 117-118.
- ORTEGA-MORA, L. M., FERRE, I., DEL POZO, I., CAETANO DA SILVA, A., COLLANTES-FERNÁNDEZ, E., REGIDOR-CERRILLO, J., UGARTE-GARAGALZA, C. & ADURIZ, G., 2003. Detection of Neospora caninum in semen of bulls. Veterinary Parasitology, 117, 301-308.
- OTTER, A., JEFFREY, M., SCHOLES, S. F. E., HELMICK, B., WILESMITH, J. W. & TREES, A. J., 1997. Comparison of histology with maternal and fetal serology for the diagnosis of abortion due to bovine neosporosis. Veterinary Record, 141, 487-489.
- OTTER, A., WILSON, B. W., SCHOLES, S. F. E., JEFFREY, M., HELMICK, B. & TREES, A. J., 1997. Results of a survey to determine whether Neospora is a significant cause of ovine abortion in England and Wales. Veterinary Record, 140, 175-177.
- PACKHAM, A. E., SVERLOW, K. W., CONRAD, P. A., LOOMIS, E. F., ROWE, J. D., ANDERSON, M. L., MARSH, A. E., CRAY, C. & BARR, B. C., 1998. A modified agglutination test for Neospora caninum: Development, optimization, and comparison to the indirect fluorescent-antibody test and enzyme-linked immunosorbent assay. Clinical and Diagnostic Laboratory Immunology, 5, 467-473.
- PARÉ, J., HIETALA, S. K. & THURMOND, M. C., 1995. An enzyme-linked immunosorbent assay (ELISA) for serological diagnosis of Neospora sp. infection in cattle. Journal of Veterinary Diagnostic Investigation, 7, 352-359.
- PARÉ, J., THURMOND, M. C. & HIETALA, S. K., 1996. Congenital Neospora caninum infection in dairy cattle and associated calfhood mortality. Canadian Journal of Veterinary Research, 60, 133-139.
- PARÉ, J., THURMOND, M. C. & HIETALA, S. K., 1997. Neospora caninum antibodies in cows during pregnancy as a predictor of congenital infection and abortion. Journal of Parasitology, 83, 82-87.
- PERL, S., HARRUS, S., SATUCHNE, C., YAKOBSON, B. & HAINES, D., 1998. Cutaneous neosporosis in a dog in Israel. Veterinary Parasitology, 79, 257-261.
- PETERS, M., LÜTKEFELS, E., HECKEROTH, A. R. & SCHARES, G., 2001. Immunohistochemical and ultrastructural evidence for Neospora caninum tissue cysts in skeletal muscles of naturally infected dogs and cattle. International Journal for Parasitology, 31, 1144-1148.
- POLI, A., MANCIANTI, F., CARLI, M. A., STROSCIO, M. C. & KRAMER, L., 1998. Neospora caninum infection in a Bernese cattle dog from Italy. Veterinary Parasitology, 78, 79-85.
- PRANDINI DA COSTA REIS, R., CRISMAN, R., ROSER, M., MALIK, R. & ŠLAPETA, J., 2016. Neonatal neosporosis in a 2-week-old Bernese mountain dog infected with multiple Neospora caninum strains based on MS10 microsatellite analysis. Veterinary Parasitology, 221, 134-138.
- PRONOST, S., PITEL, P. H., ROMAND, S., THULLIEZ, P., COLLOBERT, C. & FORTIER, G., 1999. Neospora caninum: première mise en évidence en France sur un avorton équin, analyse et perspectives. Pratique Véterinaire Equine, 31, 111-114.
- QUINTANILLA-GOZALO, A., PEREIRA-BUENO, J., SEIJAS-CARBALLEDO, A., COSTAS, E. & ORTEGA-MORA, L. M., 2000. Observational studies in Neospora caninum infected dairy cattle: relationship infection-abortion and gestational antibody fluctuations. International Journal for Parasitology, 30, 900-906.
- REID, A. J., VERMONT, S. J., COTTON, J. A., HARRIS, D., HILL-CAWTHORNE, G. A., KÖNEN-WAISMAN, S., LATHAM, S. M., MOURIER, T., NORTON, R., QUAIL, M. A., SANDERS, M., SHANMUGAM, D., SOHAL, A., WASMUTH, J. D., BRUNK, B., GRIGG, M. E., HOWARD, J. C., PARKINSON, J., ROOS, D. S., TREES, A. J., BERRIMAN, M., PAIN, A. & WASTLING, J. M., 2012. Comparative genomics of the apicomplexan parasites Toxoplasma gondii and Neospora caninum: Coccida differing in host range and transmission strategy. PLoS Pathogens, 8, e1002567.
- RODRIGUES, A. A. R., GENNARI, S. M., AGUIAR, D. M., SREEKUMAR, C., HILL, D. E., MISKA, K. B., VIANNA, M. C. B. & DUBEY, J. P., 2004. Shedding of Neospora caninum oocysts by dogs fed tissues from naturally infected water buffaloes (Bubalus bubalis) from Brazil. Veterinary Parasitology, 124, 139-150.
- RODRÍGUEZ, A. M., MARESCA, S., CANO, D. B., ARMENDANO, J. I., COMBESSIES, G., LOPÉZ-VALIENTE, S., ODRIOZOLA, E. R., SPÄTH, E. J. L., ODEÓN, A. C., CAMPERO, C. M. & MOORE, D. P., 2016. Frequency of Neospora caninum infections in beef cow-calf operations under extensive management. Veterinary Parasitology, 219, 40-43.
- ROMAND, S., THULLIEZ, P. & DUBEY, J. P., 1998. Direct agglutination test for serologic diagnosis of Neospora caninum infection. Parasitology Research, 84, 50-53.
- SANGSTER, C., BRYANT, B., CAMPBELL-WARD, M., KING, J. S. & ŠLAPETA, J., 2010. Neosporosis in an aborted southern white rhinoceros (Ceratotherium simum simum) fetus. Journal of Zoo and Wildlife Medicine, 41, 725-728.
- SANHUEZA, J. M., HEUER, C. & WEST, D., 2013. Contribution of Leptospira, Neospora caninum and bovine viral diarrhea virus to fetal loss of beef cattle in New Zealand. Preventive Veterinary Medicine, 112, 90-98.
- SASANI, F., JAVANBAKHT, J., SEIFORI, P., FATHI, S. & HASSAN, M. A., 2013. Neospora caninum as causative agent of ovine encephalitis in Iran. Pathology Discovery, 1, e5.
- SCHARES, G., PANTCHEV, N., BARUTZKI, D., HEYDORN, A.O., BAUER, C., & CONRATHS, F.J., 2005. Oocysts of Neospora caninum, Hammondia heydorni, Toxoplasma gondii and Hammondia hammondi in faeces collected from dogs in Germany. International Journal for Parasitology, 35, 1525-1537.
- SCHARES, G., RAUSER, M., ZIMMER, K., PETERS, M., WURM, R., DUBEY, J. P., DE GRAAF, D. C., EDELHOFER, R., MERTENS, C., HESS, G. & CONRATHS, F. J., 1999. Serological differences in Neospora caninum-associated epidemic and endemic abortions. Journal of Parasitology, 85, 688-694.
- SERRANO-MARTÍNEZ, E., COLLANTES-FERNÁNDEZ, E., CHÁVEZ-VELÁSQUEZ, A., RODRÍGUEZ-BERTOS, A., CASAS-ASTOS, E., RISCO-CASTILLO, V., ROSADIO-ALCANTARA, R. & ORTEGA-MORA, L. M., 2007. Evaluation of Neospora caninum and Toxoplasma gondii infections in alpaca (Vicugna pacos) and llama (Lama glama) aborted foetuses from Peru. Veterinary Parasitology, 150, 39-45.
- SERRANO-MARTÍNEZ, E., COLLANTES-FERNÁNDEZ, E., RODRÍGUEZ-BERTOS, A., CASAS-ASTOS, E., ÁLVAREZ-GARCÍA, G., CHÁVEZ-VELÁSQUEZ, A. & ORTEGA-MORA, L. M., 2004. Neospora species-associated abortion in alpacas (Vicugna pacos) and llamas (Llama glama). Veterinary Record, 155, 748-749.
- SHIVAPRASAD, H. L., ELY, R. & DUBEY, J. P., 1989. A Neospora-like protozoon found in an aborted bovine placenta. Veterinary Parasitology, 34, 145-148.
- SOLDATI, S., KIUPEL, M., WISE, A., MAES, R., BOTTERON, C. & ROBERT, N., 2004. Meningoencephalomyelitis caused by Neospora caninum in a juvenile fallow deer (Dama dama). Journal of Veterinary Medicine A, 51, 280-283.
- SOMMANUSTWEECHAI, A., VONGPAKORN, M., KASANTIKUL, T., TAEWNEAN, J., SIRIAROONRAT, B., BUSH, M. & PIRARAT, N., 2010. Systemic neosporosis in a white rhinoceros. Journal of Zoo and Wildlife Medicine, 41, 165-168.
- SÖNDGEN, P., PETERS, M., BÄRWALD, A., WURM, R., HOLLING, F., CONRATHS, F. J. & SCHARES, G., 2001. Bovine neosporosis: immunoblot improves foetal serology. Veterinary Parasitology, 102, 279-290.
- STENLUND, S., KINDAHL, H., MAGNUSSON, U., UGGLA, A. & BJÖRKMAN, C., 1999. Serum antibody profile and reproductive performance during two consecutive pregnancies of cows naturally infected with Neospora caninum. Veterinary Parasitology, 85, 227-234.
- TARANTINO, C., ROSSI, G., KRAMER, L. H., PERRUCCI, S., CRINGOLI, G. & MACCHIONI, G., 2001. Leishmania infantum and Neospora caninum simultaneous skin infection in a young dog in Italy. Veterinary Parasitology, 102, 77-83.
- THILSTED, J. P. & DUBEY, J. P., 1989. Neosporosis-like abortions in a herd of dairy cattle. Journal of Veterinary Diagnostic Investigation, 1, 205-209.
- THORNTON, R. N., THOMPSON, E. J. & DUBEY, J. P., 1991. Neospora abortion in New Zealand cattle. New Zealand Veterinary Journal, 39, 129-133.
- TREES, A. J. & WILLIAMS, D. J. L., 2005. Endogenous and exogenous transplacental infection in Neospora caninum and Toxoplasma gondii. Trends in Parasitology, 21, 558-561.
- UENO, T. E. H., GONÇALVES, V. S. P., HEINEMANN, M., DILLI, T. L. B., AKIMOTO, B. M., DE SOUZA, S. L. P., GENNARI, S. M. & SOARES, R. M., 2009. Prevalence of Toxoplasma gondii and Neospora caninum infections in sheep from Federal District, central region of Brazil. Tropical Animal Health and Production, 41, 547-552.
- UGGLA, A., STENLUND, S., HOLMDAHL, O. J. M., JAKUBEK, E. B., THEBO, P., KINDAHL, H. & BJÖRKMAN, C., 1998. Oral Neospora caninum inoculation of neonatal calves. International Journal for Parasitology, 28, 1467-1472.
- VARASCHIN, M. S., HIRSCH, C., WOUTERS, F., NAKAGAKI, K. Y., GUIMARÃES, A. M., SANTOS, D. S., BEZERRA, P. S., COSTA, R. C., PECONICK, A. P. & LANGOHR, I. M., 2012. Congenital neosporosis in goats from the State of Minas Gerais, Brazil. Korean Journal of Parasitology, 50.
- WALDNER, C. L., 2005. Serological status for Neospora caninum, bovine viral darrhea virus, and infectious bovine rhinotracheitis virus at pregnancy testing and reproductive performance in beef herds. Animal Reproduction Science, 90, 219-242.
- WILLIAMS, D. J. L., MCGARRY, J., GUY, F., BARBER, J. & TREES, A. J., 1997. Novel ELISA for detection of Neospora-specific antibodies in cattle. The Veterinary Record, 140, 328-331.
- WILLIAMS, J. H., ESPIE, I., VAN WILPE, E. & MATTHEE, A., 2002. Neosporosis in a white rhinoceros (Ceratotherium simum) calf. Journal of the South African Veterinary Association, 73, 38-43.
- WOBESER, B. K., GODSON, D. L., REJMANEK, D. & DOWLING, P., 2009. Equine protozoal myeloencephalitis caused by Neospora hughesi in an adult horse in Saskatchewan. Canadian Veterinary Journal, 50, 851-853.
- WOODS, L. W., ANDERSON, M. L., SWIFT, P. K. & SVERLOW, K. W., 1994. Systemic neosporosis in a California black-tailed deer (Odocoileus hemionus columbianus). Journal of Veterinary Diagnostic Investigation, 6, 508-510.
- WOUDA, W., 1998. Neospora abortion in cattle, aspects of diagnosis and epidemiology. PhD Thesis Utrecht, The Netherlands,
- WOUDA, W., BARTELS, C. J. M. & MOEN, A. R., 1999. Characteristics of Neospora caninum-associated abortion storms in dairy herds in The Netherlands (1995 to 1997). Theriogenology, 52, 233-245.
- WOUDA, W., DUBEY, J. P. & JENKINS, M. C., 1997. Serological diagnosis of bovine fetal neosporosis. Journal of Parasitology, 83, 545-547.
- WOUDA, W., MOEN, A. R., VISSER, I. J. R. & VAN KNAPEN, F., 1997. Bovine fetal neosporosis: a comparison of epizootic and sporadic abortion cases and different age classes with regard to lesion severity and immunohistochemical identification of organisms in brain, heart, and liver. Journal of Veterinary Diagnostic Investigation, 9, 180-185.
- YAEGER, M. J., SHAWD-WESSELS, S. & LESLIE-STEEN, P., 1994. Neospora abortion storm in a midwestern dairy. Journal of Veterinary Diagnostic Investigation, 6, 506-508.
- YÁNIZ, J. L., LÓPEZ-GATIUS, F., GARCÍA-ISPIERTO, I., BECH-SÀBAT, G., SERRANO, B., NOGAREDA, C., SANCHEZ-NADAL, J. A., ALMERÍA, S. & SANTOLARIA, P., 2010. Some factors affecting the abortion rate in dairy herds with high incidence of Neospora-associated abortions are different in cows and heifers. Reproduction in Domestic Animals, 45, 699-705.