- Infectious Diseases of Livestock
- Part 1
- Coccidiosis
- Vectors: Ticks
- Vectors: Tsetse flies
- Vectors: Muscidae
- Vectors: Tabanidae
- Vectors: Culicoides spp.
- Vectors: Mosquitoes
- Classification, epidemiology and control of arthropod-borne viruses
- Special factors affecting the control of livestock diseases in sub-Saharan Africa
- The control of infectious diseases of livestock: Making appropriate decisions in different epidemiological and socioeconomic conditions
- Infectious diseases of animals in sub-Saharan Africa: The wildlife⁄livestock interface
- Vaccination: An approach to the control of infectious diseases
- African animal trypanosomoses
- Dourine
- Trichomonosis
- Amoebic infections
- GENERAL INTRODUCTION: COCCIDIA
- Coccidiosis
- Cryptosporidiosis
- Toxoplasmosis
- Besnoitiosis
- Sarcocystosis
- Balantidiosis
- Leishmaniosis
- Neosporosis
- Equine protozoal myeloencephalitis
- GENERAL INTRODUCTION: BABESIOSES
- Bovine babesiosis
- Equine piroplasmosis
- Porcine babesiosis
- Ovine babesiosis
- GENERAL INTRODUCTION: THEILERIOSES OF CATTLE
- East Coast fever
- Corridor disease
- Zimbabwe theileriosis
- Turning sickness
- Theileria taurotragi infection
- Theileria mutans infection
- Theileria annulata theileriosis
- Theileriosis of sheep and goats
- Theileria buffeli⁄orientalis infection
- Non-pathogenic Theileria species in cattle
- GENERAL INTRODUCTION: RICKETTSIAL, CHLAMYDIAL AND HAEMOTROPIC MYCOPLASMAL DISEASES
- Heartwater
- Lesser known rickettsial infections in animals and humans
- Chlamydiosis
- Q fever
- Eperythrozoonosis
- Bovine Haemobartonellosis
- Potomac horse fever
- GENERAL INTRODUCTION: ANAPLASMOSES
- Bovine anaplasmosis
- Ovine and caprine anaplasmosis
Coccidiosis
This content is distributed under the following licence: Attribution-NonCommercial CC BY-NC  View Creative Commons Licence details here
View Creative Commons Licence details here
Coccidiosis
Previous Authors: C G STEWART AND B L PENZHORN
Current Authors:
A L, JOACHIM - Professor for Parasitology, Head of the Institute of Parasitology, DVM, DipEVPC, Department of Pathobiology, University of Veterinary Medicine, Vienna, Austria, Veterinaerplatz 1, A-1210 Vienna, Austria
B L PENZHORN - Emeritus Professor, BVSc, DSc, Department of Veterinary Tropical Diseases, Faculty of Veterinary Science, University of Pretoria, 100 Old Soutpan Road, Onderstepoort, Pretoria, 0110, South Africa
Introduction
Coccidiosis is a protozoal disease that occurs in many mammalian and all domestic livestock species. It is caused by infection with species of the genera Eimeria or Cystoisospora (syn. Isospora) and is characterized clinically in livestock by enteritis, following the invasion of cells of the gastrointestinal tract. Subclinical infections are frequent. The disease is relatively common in sheep, goats and cattle and occurs mainly in young animals, although all age groups can become infected and shed parasite stages.
Coccidiosis occurs universally, but is most prevalent where animals are subjected to overcrowding, unhygienic environments, or where other stress factors are present. Eimeria spp. are important causes of the disease in poultry, cattle, sheep and goats. Coccidiosis is rare in horses52 and in Europe and North America is an important disease of pigs.98 Although a number of Eimeria spp. are commonly found in pig faeces, clinical disease is caused only by infection with Cystoisospora suis (syn. Isospora suis). Coccidiosis due to C. suis has not yet been described in southern African countries, but it is associated with clinical disease of very young piglets in many other parts of the world.
Coccidia are generally host-specific, and, in the gut, specific to a particular location. Host specificity is stringent in both Eimeria and Cystoisospora infections. Economic losses from coccidiosis due to mortality, poor performance, and the cost of treatment and prevention may be considerable, especially on stud farms and in intensive breeding and rearing systems.
Aetiology and life cycle
Diagnosis of an infection with coccidia is accomplished during the phase of oocyst shedding; numerous stages can be found in the faeces upon coproscopy. Since differences in the pathogenicity of the species can be of clinical significance, differentiation of coccidial species is necessary in mixed infections.78 Both morphological and biological characteristics are used to identify the species of coccidia.52 The most important stage for species differentiation is the oocyst (see below), but reliable differentiation is often only possible at the fully infective sporulated stage and can be cumbersome and in some cases unreliable and subjective. For more accurate identification computer-assisted image analyses have been used in cattle91 and pigs.81 PCR-based differentiation is possible and has been developed for several host species,49, 68, 88, 110 but it is not routinely available in diagnostic laboratories. In poultry Eimeria species are not readily differentiated by oocyst morphology, and in these host species post- mortem examination of intestinal lesions is used to diagnose coccidiosis instead.42
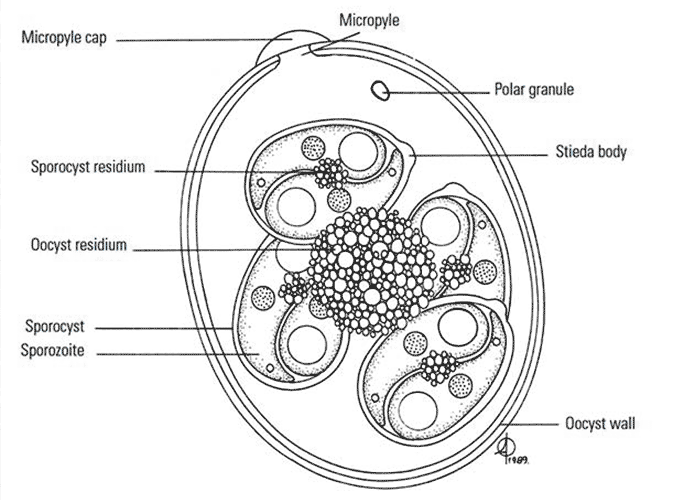
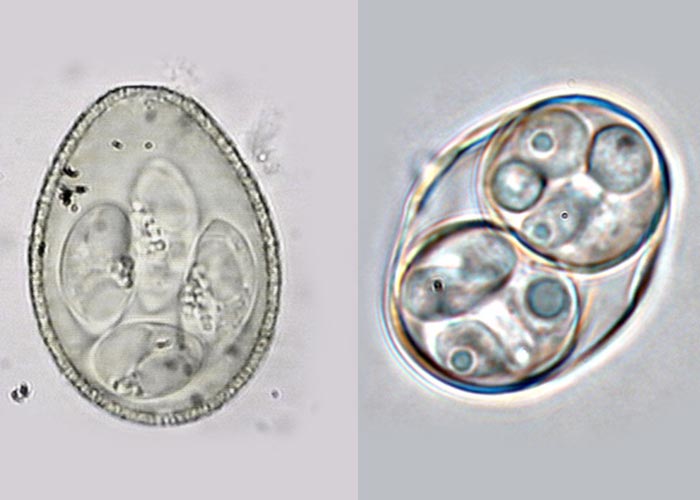
The developmental cycle of the parasite occurs intracellularly within the host (see below), while the final infective form, the oocyst, is passed into the external environment. Oocysts of mammalian livestock can be distinguished by their shape and size, the morphology of their contents, the appearance of their surface (particularly the colour and texture), and the thickness of the wall.57, 81 Variation of oocyst morphology occurs within species, and in heavy infections oocyst formation can result in aberrant forms.11 In a typical sporulated oocyst of the genus Eimeria (Figure 1), the oocyst wall is composed of one or two layers and may have a micropyle, which in turn may be covered by a micropyle cap. An oocyst or sporocyst residium, consisting of material remaining after the formation of these structures, may be present. A sporocyst may have a knob, the Stieda body, at one end.44, 52 At sporulation oocysts of the genus Eimeria develop into four sporocysts, each containing two sporozoites, while those of the genus Cystoisospora have two sporocysts, each of which contains four sporozoites (Figure 2).
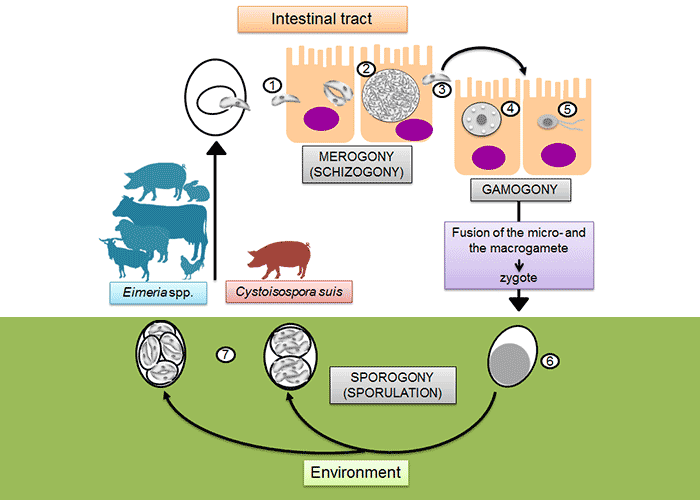
Coccidia of livestock are primarily intracellular parasites of the gastrointestinal tract, but in some animal species they may be found in other organs such as the liver or kidneys,52 or even the uterus.63 Development in the coccidia mentioned in this chapter occurs by means of an initial asexual cycle (merogony), followed by a sexual one (gamogony). Both take place in the same host (Figure 3). The final phase of development is the sporogony. Which in Eimeria and Cystoisospora occurs in the environment. The life cycle from ingestion of infectious oocysts to shedding of stages (prepatent phase) usually takes 5-17 days (rarely >20 days) depending on species.52 It is temperature and humidity-dependent. Following ingestion of infective sporulated oocysts by a susceptible host animal, the sporozoites are released through the action of the digestive juices, and, depending on the parasite species, enter epithelial or other cells of the mucosa of the gut. Here they become meronts and multiply by merogony (schizogony) to produce merozoites. These enter nearby epithelial cells or, in the more pathogenic species, invade the subepithelial tissue and and destroy endothelial cells during repeated replication.27 The number of asexual generations that occur remains constant for each species of Eimeria, while in Cystoisospora meronts develop into different types which can exist in parallel62 with sexual stages.
Merozoites of the last generation infect new host cells and become gamonts, thus initiating the sexual stage of the life cycle. Some of the gamonts develop into macrogametes without further multiplication, while others divide by multiple fission to form a large number of flagellated microgametes. These microgametes are released and penetrate the macrogametes to form zygotes. A thick encompassing wall is formed around each zygote, which is then known as an oocyst. When the host cell is ruptured, the oocysts are released into the lumen of the gut, and are excreted in the faeces in an unsporulated state. If environmental conditions of temperature, oxygen supply and humidity are suitable, oocysts sporulate and sporocysts containing infective sporozoites are formed. This process takes between one and five days under optimal external conditions, which include a temperature of 15 to 30 °C.
Coccidia are haploid throughout their life cycle, except in the zygote stage. The first sporogonous division is meiotic, re-establishing the haploid state. Oocysts administered to aberrant hosts will excyst and begin merogony, but they disappear after 48 hours.61 By contrast, some Cystoisospora species utilise paratenic hosts in which they produce monozoic cysts in tissue that can infect the definitive host upon ingestion.56 However, this phenomenon has not been demonstrated for porcine coccidia.
Sheep
In sheep 15 Eimeria species are described (Table 1). The most pathogenic species that cause clinical disease are E. ovinoidalis and E. crandallis.34, 105 Other species are less pathogenic but may play an augmentative role as mixed coccidial infections are the rule in field outbreaks.82
The characteristic features of the sporulated oocysts of the most common coccidia occurring in sheep are shown in Table 1. Oocysts of E. weybridgensis are similar in size to E. crandallis, but the sporozoites of E. weybridgensis are more elongate and lie lengthwise in the sporocyst, while those of E. crandallis are more ovoid and lie at either end of the sporocyst.57
The meronts of E. (Globidium) gilruthi, a poorly described species with uncertain taxonomic status,are visible to the naked eye during necropsy as whitish nodules (300 to 900 μm in diameter) in the mucosa of the abomasum of sheep and goats.52 It is generally considered to be of low pathogenicity but has been described anecdotally as a cause of abomasitis.59
Table 1 Oocyst characteristics of ovine Eimeria species.78, 106
|
| Form | Oocyst size [µm] | Micropyle | Micropyle cap | Oocyst residual body | Sporocyst residual body | Stieda body | Other characteristics |
| E. ahsata | Ovoid | 30-39 x 19-30 | + | + (distinct) | - | + | - | Yellow-green |
| E. bakuensis | Elliptic | 23-36 x 15-24 | + | + | - | + | + | Green to brownish |
| E. crandallis | round | 17-23 x 17-22 | + | + | - | + | ? |
|
| E. faurei | Ovoid | 22-33 x 19-24 | + | - | - | - | + |
|
| E. granulosa | Urn-shaped | 22-35 x 17-25 | + | + (distinct) | - | + | + | Yellow-green |
| E. intricata | Elliptic | 40-56 x 30-41 | + | + | - | + | + | Yellow to dark brown |
| E. marsica | Elliptic | 15-22 x 11-14 | + | + | - | + | + |
|
| E. ovinoidalis | Oval | 17-25 x 13-20 | - | - | - | + | - | greenish |
| E. pallida | Oval | 12-20 x 8-15 | + | - | - | + | - |
|
| E. parva | Round | 13-23 x 10-19 | - | - | - | + | - |
|
| E. punctata | Elliptic-subspherical | 18-25 x 16-21 | + | + | + | + | + | Surface with cone-shaped pits |
| E. weybridgensis | Long-oval | 17-30 x 14-19 | + | + | - | - | - | Dark inner layer |
Goats
Coccidia species that infect goats are, in spite of being morphologically similar, distinct from those of sheep.74 Currently, seven species are described as valid, E. arloingi, E. christenseni, E. hirci, E. alijevi, E. ninakohlyakimovae, E. caprina, and E. caprovina (Table 2); E. kocharli is not considered valid. Other species are described with limited geographical distribution.78 Due to their high similarity with ovine species, cross transmission with caprine Eimeria was previously considered13 but this could not be proven. The most pathogenic species are E. ninakohlyakimovae and E. arloingi.78
Table 2 Oocyst characteristics of caprine Eimeria species.78, 106
|
| Form | Oocyst size [µm] | Micropyle | Micropyle cap | Oocyst residual body | Sporocyst residual body | Stieda body | Other characteristics |
| E. alijevi | Round | 12-22 x 10-18 | - | - | - | + | - | Yellow-brownish |
| E. arloingi | Elliptic | 17-42 x 13-27 | + | +, distinct | - | + | - |
|
| E. aspheronica | Ovoid | 30-40 18-27 | + | - | - | - | - |
|
| E. caprina | Elliptic | 27-40 x 19-26 | + | - | - | + | - | Wall thick and dark brown |
| E. caprovina | Elliptic | 26 x 21-28 | + | - | - | + | + |
|
| E. christenseni | Ovoid | 34-41 x 23-28 | + | + distinct | - | + | - | brown |
| E. hirci | Round | 18-23 x 14-19 | + | + | - | + | ? |
|
| E. jolichijevi | Urn-shaped | 22-35 x 17-25 | + | + distinct | - | ? | - |
|
| E.nina-kohlyakimovae | Oval | 19-25 x 14-21 | +/- | - | - | + | - |
|
Cattle
At present between 19 and 22 species of Eimeria have been identified in cattle although not all species are validated (Table 3). Most of these are relatively non-pathogenic, and field outbreaks of coccidiosis are generally caused by E. bovis or E. zuernii and both can frequently be found in combination.
Eimeria bovis develops in the endothelium of the intestinal lymphatic vessels in the distal part of the small intestines. Second generation macromeronts of E. bovis and E. zuernii, containing hundreds of thousands of merozoites,20 occur in the large intestine in the lamina propria near the muscularis mucosae and are primarily responsible for the severe destruction of the intestinal lining in heavy infections, together with the gamonts.94
Table 3 Oocyst characteristics of bovine Eimeria species.15, 106
|
| Form | Oocyst size [µm] | Micropyle | Micropyle cap | Oocyst residual body | Sporocyst residual body | Stieda body | Other characteristics |
| E. alabamensis | Ovoid | 13-24 x 11-16 | - | - | - | - | + |
|
| E. auburnensis | Long-oval-ovoid | 32-46 x 20-25 | + | - | - | + | + | Orange-green |
| E. bovis | Ovoid | 23-34 x 17-23 | + | - | + | + | + | Wall thick and orange-brown |
| E. brasiliensis | Elliptic | 33-43 x 24-30 | + | - | - | + | + |
|
| E. bukidnonesis | Pear-shaped | 47-50 x 33-38 | + | - | - | - | - |
|
| E. canadensis | Ovoid | 28-37 x 20-27 | + | - | - | + | ? |
|
| E. cylindrica | Long-elliptic | 16-27 x 12-15 | - | - | - | + | - |
|
| E. ellipsoidalis | Elliptic | 20-26 x 13-17 | - | - | - | + | + | Very dark |
| E. illinoisensis | Elliptic-ovoid | 24-29 x 19-22 | - | - | ? | ? | ? |
|
| E. pellita | Ovoid | 36-41 x 26-30 | + | - | - | + | + |
|
| E. subspherica | Round-subspherical | 9-14 x 8-13 | - | - | - | - | + | Wall thick and bilayered |
| E. wyomingensis | Ovoid | 37-45 x 26-31 | deutlich + | - | - | - | + |
|
| E. zuernii | Subspherical | 15-22 x 13-18 | - | - | - | + | +, |
|
Pigs
Cystoisospora suis infects primarily the jejunum of pigs and is the causative agent of porcine neonatal coccidiosis.55 Excretion of oocysts in the faeces starts four to five days after infection and continues for one to three weeks.55
Eimeria spp. that commonly occur in pigs are E. debliecki, E. neodebliecki, E. porci and E. suis, E. scrabra, E. polita, E. perminuta and E. spinosa (which are rough-walled).98 All Eimeria spp. of pigs are considered to be either non- or only mildly pathogenic and are not significant causes of disease in pigs.55 The morphology of the oocysts of porcine coccidia is described in Table 4.
Table 4 Oocyst characteristics of porcine Eimeria species and Cystoiospora suis.17, 48, 53, 78
|
| Form | Oocyst size [µm] | Micropyle | Micropyle cap | Oocyst residual body | Sporocyst residual body | Stieda body | Other characteristics |
| C. suis | Round -elliptic | 20-24 x 18-21 | - | - | - | + | - | Unilayered, thin, smooth wall |
| E. debliecki | Elliptic-ovoid | 18-24 x 18-25 | - | - | - | + | + | Smooth wall |
| E. neodebliecki | Elliptic-ovoid | 17-26 x 13-20 | - | - | - | - | distinct + | Smooth wall |
| E. perminuta | Elliptic-round | 11-16 x 10-13 | - | - | - | + | + | Rough wall |
| E. polita | Elliptic-ovoid | 22-31 x 17-22 | - | - | - | + | + | Rough wall |
| E. porci | Ovoid | 18-27 x 13-18 | - | - | - | + | + | Smooth wall |
| E. scabra | Ovoid-asymmetric | 22-36 x 16-26 | + | - | - | + | + | Rough wall, brownish |
| E. spinosa | Elliptic-ovoid | 16-22 x 13-16 | - | - | - | + | distinct + | Spiny, rough wall |
| E. suis | Elliptic-ovoid | 15-23 x 12-18 | - | - | - | + | + | Smooth wall |
Horses
Eimeria leuckarti, which is frequently encountered in horses, may be pathogenic5 but is usually an incidental finding. The very large (70-90 x 46-69 µm) pear-shaped oocyst is surrounded by a thick brown layer.4 The macromeronts are 300 × 170 μm in size and so are visible to the naked eye. They occur in the small intestine.52
Epidemiology
Coccidiosis is essentially a disease that is prevalent in young animals under intensive management systems. Older animals are rarely affected although they may serve as clinically inapparent shedders.
Disease usually occurs when susceptible, naive animals are infected for the first time (usually in the first days or weeks of life) and the parasite can develop and multiply without being controlled by the immune system. High numbers of oocysts are shed for one to several days. Depending on the host and parasite species, prolonged low-level shedding can occur. Although infections are self-limiting, high numbers of oocysts that are present in the environment and viable for months under suitable conditions support the continuation of the infection on a farm. After the first infection immunity builds up, which is constantly boosted by the intake of oocysts from the animals’ surroundings. Immunity prevents disease but shedding of oocysts still occurs in most species, so that older animals maintain the life cycle of the parasite.
Coccidia are practically ubiquitous. Risk factors for the development of clinical disease include the presence of susceptible young stock, especially in crowded conditions, stress from inadequate feeding or housing or inclement weather, lack of colostrum intake, poor sanitary conditions (which are often accompanied by a high level of environmental oocyst contamination), high humidity, which supports oocyst survival in the surroundings, and imbalances of the gastrointestinal microbiota, e.g. from change of feed (weaning) or bacterial/viral infections. Contaminated feed (e.g. when feeding animals from the ground), and wet floors (e.g. from leaking drinkers) or wet/irrigated pasture can also contribute to the survival, dissemination and transmission of oocysts.
Sporulated oocysts are very resistant to adverse environmental conditions and may ‘overwinter’ on pastures, which then act as a source of infection when favourable climatic conditions return.24
Although hygiene and good management contribute to the control of coccidiosis, outbreaks have also been observed on well-managed farms, and frequently chemotherapeutic intervention is required to prevent economic losses.
Sheep and goats
The disease is most severe in lambs two to eight weeks of age37 and in lambs two to three weeks after weaning. Sheep entering feedlots, or after experiencing severe stress such as that caused by transport or inclement weather are prone to developing severe disease.24
Most sheep and goats pick up oocysts as young animals from contaminated surroundings, possibly also from faecal contamination of the teats and udder of their dams. The parasite population is increased enormously during the first passage in susceptible lambs, putting later-born lambs at greater risk.10 Sheep and goats usually become infected with a number of Eimeria spp. and oocyst excretion can extend over several months. Mixed infections prolong the patency and increase the number of oocysts produced.12 Faecal oocyst counts may be high in apparently healthy lambs.71
The presence of the more pathogenic species, such as E. crandallis and E. ovinoidalis, in high numbers plays an important role in the production of overt disease in sheep.34, 105
Coccidiosis is often a serious problem in feedlots. Species of coccidia to which animals in the feedlots have not previously been exposed may be introduced together with newly purchased animals. Conversely, non-immune animals may be placed in a pen contaminated with oocysts shed by previous or concurrent stock. Clinical signs may develop 10 to 20 days after a new lot of lambs are introduced. Mortalities in feedlots may be high and are often associated with sudden outbreaks of the disease. Once a pen has become infected, oocysts may remain there for a considerable period (weeks to months depending on temperature and humidity), creating a hazard for any new introductions as well as for animals already in the pens. Feed troughs may become contaminated with oocysts from faeces that are inadvertently consumed and, in this way, the production of massive infections or reinfections is facilitated. High concentrations of oocysts in the pens may result when animals are penned at night.
Very wet summers are known to result in a high prevalence of coccidiosis because of the survival of larger numbers of oocysts and stressing of the animals caused by the weather conditions.
Cattle
Clinical coccidiosis is primarily a disease of calves between three weeks and six months of age, although older animals may show signs of infection. Overt disease occurs when large numbers of oocysts are ingested, while the ingestion of smaller numbers of oocysts leads to subclinical infection followed by the development of immunity. In the summer rainfall areas of South Africa the practice of removing calves from their dams during the winter months of June and July and placing them in pens to facilitate weaning commonly results in outbreaks of coccidiosis.93 Outbreaks occur despite the fact that under the dry winter conditions oocyst contamination is likely to be fairly low. Outbreaks of coccidiosis in winter have also been reported from countries where animals are kept indoors during the cold months.73 It would appear that stress, such as that produced by weaning or cold weather, may be important in initiating outbreaks.77 Another predisposing factor for disease outbreaks is the accumulation of oocysts in the bedding when deep litter is used. In such cases disease is typically seen by the end of the prepatent period of the pathogenic species, around three weeks after moving calves to the deep litter pens. The disease may also occur in young cattle kept under extensive farming conditions when high oocyst contamination occurs around watering points.
Eimeria alabamensis has been described as a cause of pasture coccidiosis but in experimental infections very high doses were required to induce clinical disease39, 101 so the clinical significance of natural infections is doubtful. E. auburnensis and E. ellipsoidalis have produced diarrhoea in experiments.20
The presence of subclinical coccidiosis may interfere with the response to vaccination against other diseases.76
Pigs
Unsporulated oocysts of C. suis sporulate very rapidly (within 16 hours at 30 °C) under suitable moisture and temperature conditions, whereas Eimeria spp. take from 8 to 13 days to sporulate.98 Day-old piglets are more susceptible to first infections than month-old piglets maintained free of C. suis infection.47 After the first infection immunity and a rapidly increasing age-resistance largely prevent re-shedding. Adult sows do not appear to shed C. suis, so contaminated farrowing crates are the most likely source of infection.55 Under intensive management conditions in modern piglet production the parasite reaches very high prevalences.107
Pathogenesis
Factors that play an important role in the pathogenesis of the disease include the pathogenicity of the coccidia species concerned, amount of oocysts ingested, age and disposition of the host, location of the parasite in the tissues and the presence or absence of acquired immunity.22
Some species of coccidia are highly pathogenic whereas others are of low pathogenicity, which is correlated with the number and location of endogenous stages arising from oocyst ingestion. Infection is self-limiting provided that reinfection does not occur.52 Nevertheless, coccidiosis can have a lasting effect on performance, and growth retardation and poor weight gain may last beyond the acute phase of infection.50, 69
Many of the affected cells are destroyed or their function impaired.52 Depending on the species, meronts and / or gamonts are responsible for most of the tissue destruction. In severe infections with the most pathogenic species there is an acute inflammatory reaction. Capillaries in the denuded lamina propria may haemorrhage, leading to hypoproteinaemia and anaemia. Less pathogenic species lead to mild inflammation with influx of fluid into the gut lumen. Secondary bacterial infections may exacerbate the clinical picture. All these changes stimulate an increased rate of peristalsis and result in diarrhoea. The reduced absorption capacity of the parasitized mucosa results in loss of fluids via the intestine and decreased absorption of electrolytes such as sodium and potassium, as well as a loss of nutrients due to the impaired gut function. Compensatory increase of absorption in normal portions of the gut may redeem this loss.11 Dehydration and acidosis due to the loss of bicarbonate ions in the faeces, as well as anaemia, hypoproteinaemia, and shock combined with secondary bacterial infection, are the main causes of death in the severest cases.
The effects of coccidiosis due to E. zuernii and E. bovis are mainly the result of damage to the mucosa of the large intestine caused by the second-generation meronts and sexual stages.86 Second-generation meronts and gamonts of E. zuernii are simultaneously present in the caecum, colon and rectum. Maximal destruction of the epithelium of the large intestine occurs in cattle just before the peak output of oocysts.95 This results in a reduction in the absorption of water and sodium and chlorine ions with a consequent rapid loss in body weight. Animals dying at this stage probably do so from dehydration. The severity of the infection is reflected in changes in haematological parameters.2, 95
Immunity develops after infection, but the determinants of protective immunity have yet to be elucidated.87 CD 4+ and CD 8+ T cells as well as gammadelta TCR+ T cells seem to play a role in the local immune response to E. bovis99 and C. suis.28 Subclinical infection at a young age results in the development of specific immunity early in life. This immunity may, however, wane under conditions of stress such as during transportation, exposure to climatic extremes, changes of diet and overcrowding, during the periparturient period or when massive infections occur.21 Drugs may also interfere with the development of immunity. Lambs exposed to an aureomycin-sulphamezathine supplement did not develop immunity when fed 500 mg/lamb/day for 35 days, but were able to develop immunity when fed a lower dose of 100 mg/lamb/day.1 Intermittent or continual reinfection enhances the level of immunity. The immunity that develops in response to an Eimeria infection is species-specific.87 Protection against infection has been obtained by immunization of lambs with E. crandallis and E. ovinoidali 10, 32 and in calves with E. alabamensis.102 A ‘trickle-infection’ appears to be the most effective means of producing immunity. In swine where C. suis infections occur at a very early phase of life, maternally derived partial protection of the piglets could be induced by superinfection of sows before farrowing with high doses of oocysts.89
Clinical signs
Sheep and goats
The onset of diarrhoea is usually the first sign of illness. Affected animals pass soft, unpelleted faeces and later watery diarrhoea. Blood may be present in the faeces but it is less common in sheep and goats than in cattle. The perineum and hind limbs become soiled and this may precipitate fly strike. Anorexia and dehydration develop a few days after the commencement of the diarrhoea, with resultant loss of body mass. These are followed in severe cases by depression, recumbency and death.
The morbidity ranges from 10 to 50 per cent and the mortality may be up to 10 per cent in ruminants. In Angora goat flocks in the Eastern Cape Province of South Africa morbidity can reach 100 per cent with mortality as high as 50 per cent.109 In the sandy, arid eastern parts of Namibia a disease known as ‘boksiekte’ (goat disease) is due to a combined effect of coccidiosis and infestation with the worm Strongyloides papillosus.109 The course varies from one to two weeks and the disease is manifested by the clinical signs of ataxia, opisthotonus and convulsions, in conjunction with various degrees of enteritis.
Cattle
The incubation period is usually about three weeks (E. bovis and E. zuernii), but may vary between a week and more than a month depending on species composition.38
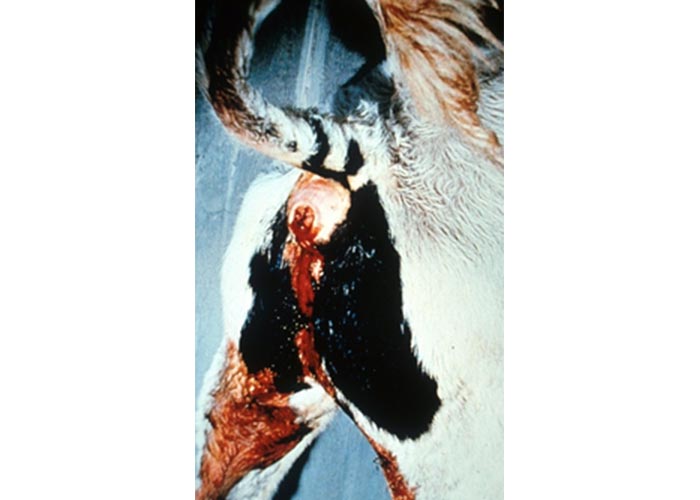
The predominant clinical sign is diarrhoea, which may be poorly developed at first, but becomes more watery and finally haemorrhagic. As the disease progresses there is an increase in the amount of fresh blood in the faeces, and blood clots may be present. After a few days the amount of blood in the faeces may diminish, being replaced by mucus and muco-fibrinous exudate. Later the discharges become blackish-red to blackish, or greenish-black and foul-smelling; shreds of mucosa may be present. The diarrhoea, which may be profuse and foetid, is often accompanied by tenesmus (Figure 4) and pronounced borborygmi. The animal may constantly grind its teeth. Abdominal pain is usually present and, if the tenesmus is intense, prolapse of the rectum may occur. There is often a fever, an increase in the respiratory rate and loss of appetite.38 Ocasionally animals become immobilised with signs of neurological diseases such as opisthotonus.
During the course of the disease, severely affected animals become extremely emaciated and anaemic. After about 8 to 12 days, they either show signs of improvement, or deteriorate and die.38 Severely affected animals often remain unthrifty.
Pigs
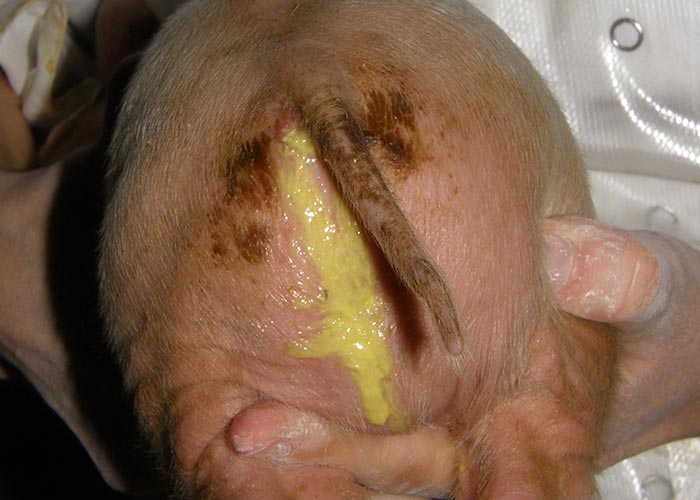
Coccidiosis usually occurs in 5- to 10-day-old piglets, and is characterized by the development of creamy to watery yellow diarrhoea (Figure 5), dehydration and loss of condition. Faeces are yellow to grey and do not contain visible blood.55 Vomiting may occur. The mortality is usually less than 20 per cent,98 but can rise significantly in cases where bacterial pathogens are present. The most severe outcome of piglet coccidiosis is the uneven weight development in affected litters which can still be present during weaning.
Pathology
Sheep and goats
Decreases in the levels of serum albumin and plasma alkaline phosphatase coincide with the onset of diarrhoea.8 The animal may be cachectic or even anaemic.
The location of the lesions in the gut, and to some extent their appearance, varies with the Eimeria spp. concerned, but most infections are mixed.12
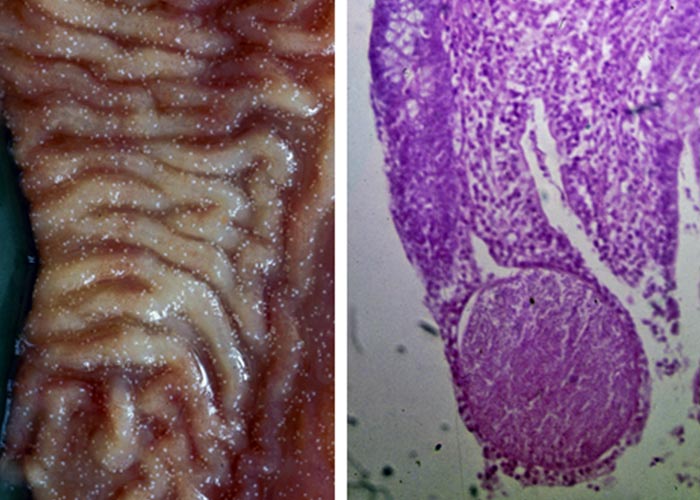
In most natural coccidial infections small, flat to dome-shaped, white foci, 1 to 2 mm in diameter, each consisting of an area where there is severe infection of epithelial cells, occur in the mucosa of the small intestine (Figure 6). These foci, which have a diameter of up to 300 μm, may also be discernible from the serosal surface of the gut31 and are due to the schizont stage ofpathogenic Eimeria speciesin sheep and goats.43 Macroscopically visible, discrete opaque patches may also be seen in the mucosa in E. bakuensis infections because of the presence of groups of heavily parasitized intestinal villi. These may appear as raised patches or even develop into polyps.35 Affected areas of the intestine of severely affected animals are oedematous and thickened, and there may be focal or diffuse congestion or haemorrhage in the mucosa.
Sloughing of the intestinal mucosa may occur, sometimes with ulceration. Haemorrhagic enteritis of the caudal part of the small intestine occurs in the case of E. ovinoidalis in sheep and E. ninakohlyakinovae in goats, and blood may be present in the faeces.49
Microscopically there is disorientation of the villous architecture, a reduction in epithelial cell height, and a decrease in the width of the brush border.84 The villi may be short or absent and may contain proliferative epithelia, with various stages of coccidia being present (Figure 7). Malabsorption may occur if these lesions are widespread.43
Cattle
The sodium and chloride ion levels in the blood decrease about 18 days after experimental infection and reach their lowest levels seven days later.95
The macromeronts of E. bovis may be seen as small white spots in the mucosa of the ileum, but those of E. zuernii are not visible macroscopically.20
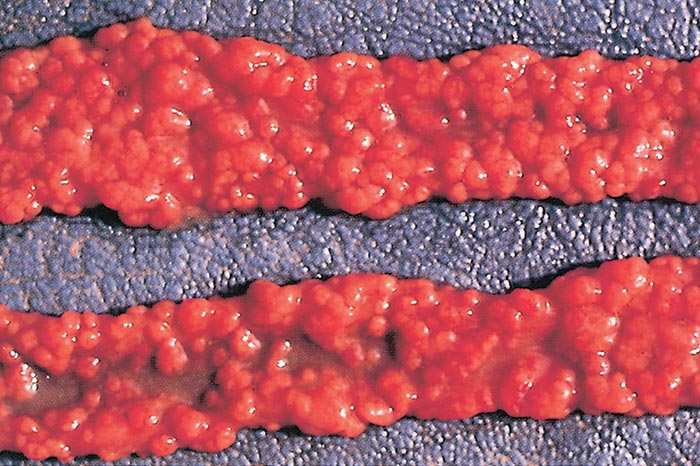
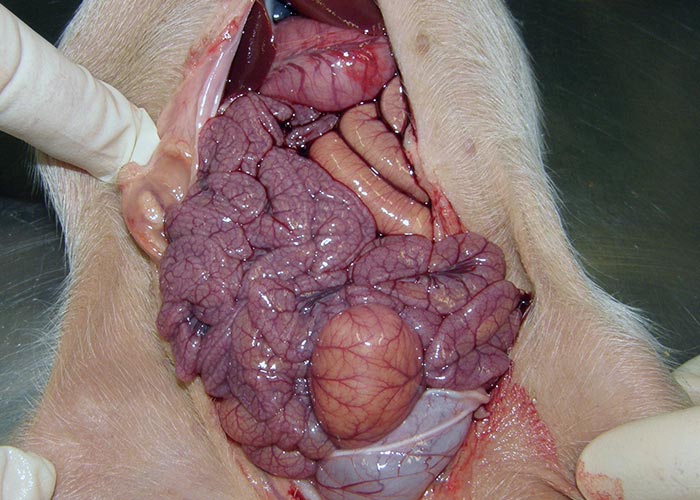
In severe cases, fibrino-haemorrhagic typhlocolitis is typically present as is, in many cases, proctitis (Figure 8 and Figure 9). The terminal ileum may be affected.43 The content of the caecum, colon and rectum becomes semi-fluid and bloody in the early stages of infection. During the later stages sloughed mucosa and copious amounts of blood and fibrin may form fibrino-necrotic pseudomembranes.
The content of the large intestines has an unpleasant smell. The mucosa may contain erosions, and both it and the submucosa are oedematous and congested and contain petechiae. The mesenteric lymph nodes are often enlarged and oedematous.
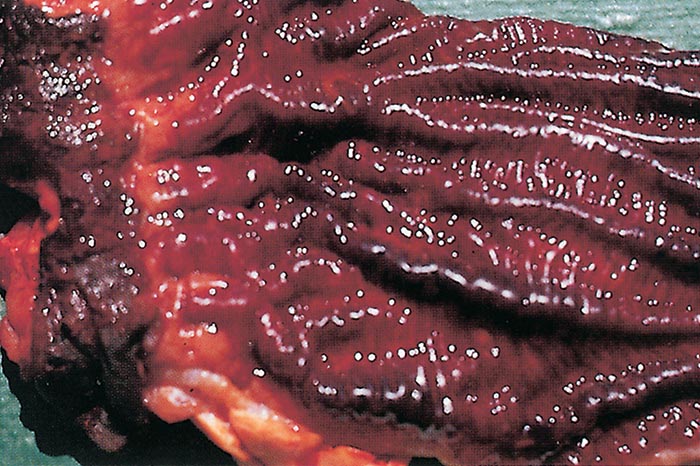
Microscopically, the mucosa of the caudal part of the small intestine is invaded by first-generation meronts and may be infiltrated by a few lymphocytes and eosinophils, but, in general, the lesions are mild.20 The lesions caused by second-generation meronts and gamonts, on the other hand, are characterized by extensive changes.20 In the case of E. zuernii and E. bovis infections these are present in the large intestine (Figure 10). In severe infections almost all the epithelial cells of the affected part contain parasites in various stages of development. Many affected cells lose their integrity and become detached, while the crypts become distended by oocysts and cellular debris and the lamina propria is congested and infiltrated with plasma cells, lymphocytes, neutrophils and eosinophils. Extensive haemorrhage occurs in the tissues. Capillaries in the lamina propria denuded of epithelium may be exposed to the intestinal content because of the sloughing of the epithelial cells.
Pigs
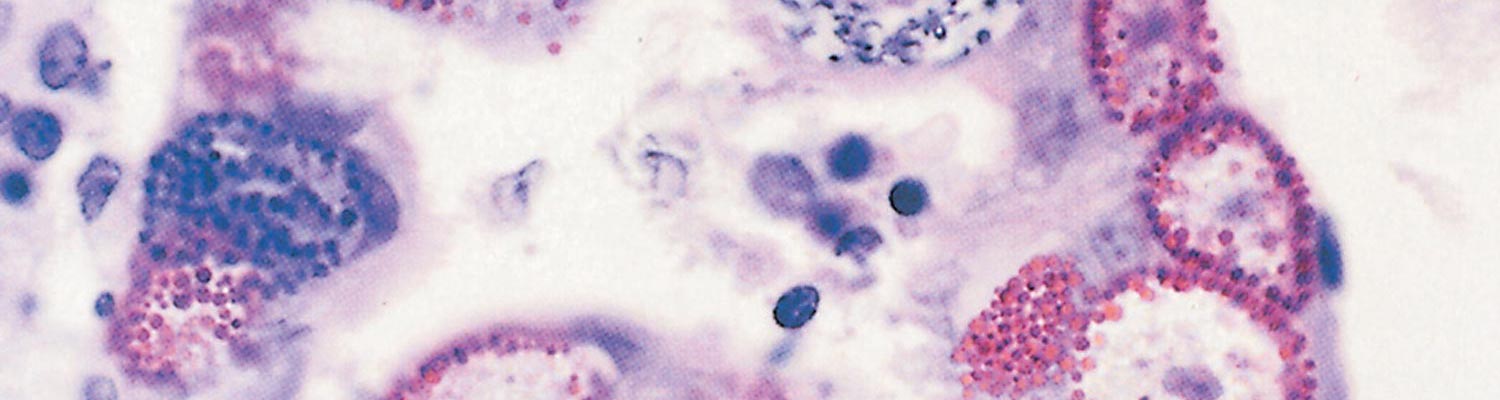
Fibrinous or fibrino-necrotic enteritis in the caudal part of the small intestine is present in some fatal cases, and the small intestines have a rope-like appearance (Figure 11). This is presumably due to the simultaneous presence of Clostridium perfringens and not primarily due to the C. suis infection. Microscopically, severe destruction of the villous epithelium of the jejunum and ileum, and marked atrophy of the villi are evident. Endogenous stages of C. suis can be seen in the villous epithelium.98
Diagnosis
The history of the case, clinical signs and necropsy findings, as well as the demonstration of the parasite, are all relevant when diagnosing coccidiosis.
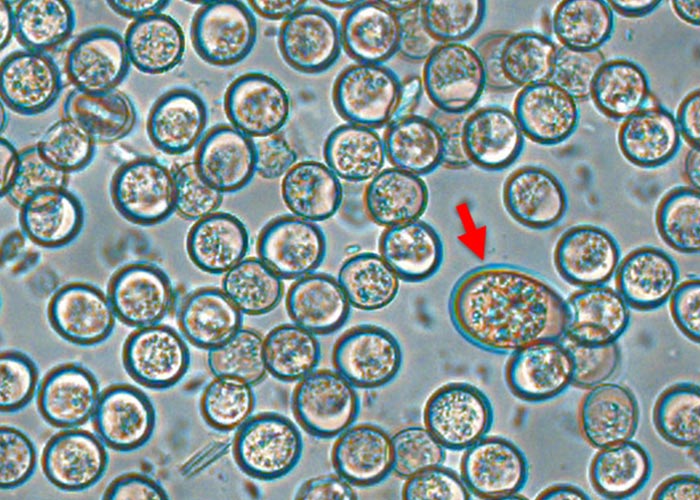
In order to visualize oocysts, a small quantity of faeces from an affected animal is mixed with water and examined microscopically, but, in some cases, concentration of the oocysts is required before they can be demonstrated. This is achieved by flotation of the oocysts in a fluid with a specific gravity between 1,1 and 1,3. The most commonly used is either a 40 per cent sugar or a saturated sodium chloride solution, the former being preferred as it is less detrimental to the oocysts. Approximately 2 to 3 g of faeces are first mixed with about 10 ml of water. The mixture is then passed through a sieve to remove gross debris, placed in a test tube and centrifuged. The supernatant fluid is decanted and the sediment mixed with about 10 ml of the flotation solution, and again centrifuged. The oocysts ‘float’ to the surface and many of them can be removed in a droplet of fluid obtained by touching the surface of the supernatant fluid with a flat object such as the end of a glass rod. The droplet is placed on a glass microscope slide, covered with a cover slip, and examined under a microscope.
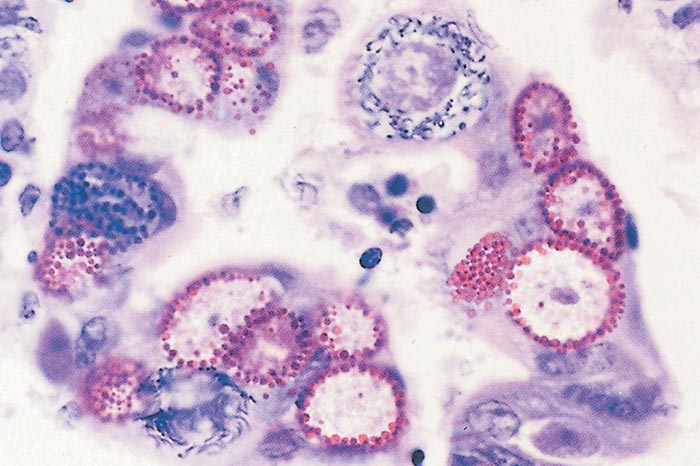
In piglets where a high fat content of the faeces precludes the use of flotation or the examination of faecal smears under the light microscope, autofluorescence microscopy (Figure 12) can be used to detect oocyst in faeces.16 In very liquid faeces, oocysts are often not detectable.
Species identification of the coccidia requires some experience, but by comparison with textbook drawings, some of the more common oocysts of sheep and cattle can be distinguished.7 Oocysts may be more readily differentiated after sporulation has occurred. Sporulation can be induced by mixing a small sample of faeces with a 2 per cent aqueous potassium dichromate solution and allowing it to stand at room temperature for three to four days.67 Accurate identification is a task for a parasitologist.
Estimates of the number of oocysts in faeces can be carried out by the modified McMaster method.67 However, it may be difficult to interpret the results as high counts can occur in apparently healthy animals. Low counts may be present in diseased animals as the majority of the parasites may not have progressed to the oocyst stage at the time of the examination.83 In these cases, smears of faeces should be made and stained with Giemsa to demonstrate the presence of merozoites.52 Sheep tend to have higher oocyst counts than goats.44 At necropsy, either scrapings from lesions or tissue sections of the intestine may be examined microscopically for the presence of meronts, gamonts or oocysts.52 Mixed infections usually occur.
Healthy lambs between four and eight weeks of age may have very high faecal oocyst counts (of the order of 500 000/g or higher), but after this age there is a progressive reduction in oocyst numbers so that by five months, oocyst numbers are similar to those in adult sheep. This makes the interpretation of oocyst counts in relation to their clinical significance very difficult.83
Coccidiosis should be suspected in young cattle with haemorrhagic diarrhoea. As most field outbreaks of coccidiosis are due to E. zuernii,20 the oocyst should be identified (Figure 13). In some cases diarrhoea may begin a day or so before oocysts can be detected in the faeces. Microscopic examination of the faeces of these animals for oocysts should be repeated.
In piglets the demonstration of oocysts of C. suis in the faeces (see Figure 2 and Figure 12) may be difficult as they may be present in low numbers and excretion can occur before, during or after the onset of diarrhoea. It is thus advisable to take individual samples from half of the litter to examine a pooled sample, or to sample repeatedly.65 Death may occur before this stage, in which case it is necessary to perform histological examinations of sections of the intestinal tract in order to detect the organisms. In some animals, microscopic examination of smears made from the gut contents may reveal merozoites and meronts.72
Differential diagnosis
A number of conditions of sheep, including colibacillosis, lamb dysentery, salmonellosis, cryptosporidiosis and rotavirus infection, which usually occur in young lambs under four weeks of age,68 should be considered in the differential diagnosis of ovine coccidiosis.
Mixed infections may, however, occur. Helminth infestations, either on their own or in conjunction with coccidia infections, may result in diarrhoea, but the eggs of helminths are much larger than oocysts and can readily be differentiated.
Clinically, bovine coccidiosis should be differentiated from conditions in which diarrhoea is a major presenting sign. Foremost among these are infectious diseases, such as salmonellosis, colibacillosis, rotavirus and coronavirus infections, bovine viral diarrhoea-mucosal disease, and cryptosporidiosis; intoxications caused by the ingestion of plants, such as the chincherinchee (Ornithogalum spp.), tulp (certain o Moraea spp.) and slangkop (Ornithoglossum and Drimia (Urginea) spp.),45 and helminth infestations, should also be considered.
Cistoisospora (Isospora) suis infection should be differentiated from other infectious causes of enteritis and diarrhoea in young piglets, such as colibacillosis, clostridiosis, and rotavirus and coronavirus infections. Strongyloidosis is rather rare in intensive piglet production but may also cause diarrhoea in young piglets. Concurrent infections may occur.
Control
Application of the principles of management, hygiene and chemotherapy plays a role in the control of coccidiosis.52 Once clinical signs are apparent, the disease is advanced and considerable contamination of the environment has occurred. Although clinical cases may be successfully treated, treatment may be expensive and the subsequent productivity of the recovered animal is often poor.52 The extent of disease is strongly correlated with management. For these reasons hygiene and metaphylactic treatment are the most effective approach to control.
Management and hygiene
If the intake of sporulated oocysts by young animals is kept at a low level through good management practices, infections may become established at levels that stimulate immunity without causing clinical disease.32 Young, susceptible animals should be kept clean and dry, and feed and water troughs should be constructed and maintained in such a manner that as little feed as possible falls to the ground, leakages are prevented, and faecal contamination is minimized. Proper drainage of paddocks, kraals and feedlot pens is also important in order to prevent the accumulation of large numbers of oocysts and to create an adverse environment for their development. The faeces of animals should, if possible, be removed from pens before oocysts have sporulated. Stress associated with such factors as weaning, sudden changes of diet and transportation should be minimized.
Older animals, although partially immune, may continuously shed oocysts in their faeces and so contaminate pastures. Susceptible animals, when placed on these apparently safe pastures, may suffer severe consequences. This occurs especially when animals are transported from the dry extensive farming areas and are placed in intensive production systems or wet environments. Whenever possible, clinically affected animals should be isolated.
Piglets present an exception to these rules as infection takes place at a very early age and disease severity is increased in infections in the first three days of life, so even small amounts of oocysts ingested straight after birth can lead to disease outbreaks and the fast development time of C. suis leads to immediate infection of litter mates, which is facilitated by the extensive coprophagy in pigs. These factors greatly reduce the effects of good hygiene in pens as even low-grade infections will quickly lead to high morbidity.
In a heavily infected environment, sterilization of bedding and soil with aqueous solutions of 1,25 per cent sodium hypochlorite, 0,5 per cent cresol or phenol, or by fumigation with formaldehyde,92 may be achieved but this is not a practical control measure (except for pigs where all-in-all-out management of farrowing areas can be combined with disinfection). Exposure to sunlight (for at least eight hours), and desiccation (humidity less than 25 per cent) are the most cost-effective methods of killing oocysts.36
Chemotherapy
Clinical coccidiosis is difficult to treat, and success is normally limited. By the time clinical signs are evident, the infection has usually progressed to the sexual stage of the life cycle and oocysts are appearing in the faeces. Extensive destruction of intestinal epithelium will have taken place and recovery will be slow. In addition, the parasites may have passed through the stages against which the applied anticoccidial drugs are effective. Treatment of clinically affected animals should therefore include supportive therapy to aid in the repair of damaged gut and to limit secondary infection.79
Supportive treatment includes the administration of antiprotozoal drugs, antidiarrhoeal compounds and fluids, and, when necessary, haematinics and blood transfusions in exceptional cases.20 Specific treatment of concurrent infestations with helminths or other infections may be indicated.9
Where animals are penned, those showing clinical signs should be removed, treated immediately and examined for oocyst shedding before returning to prevent further contamination of the environment. All other susceptible animals in the pen should be treated metaphylactically, as they may harbour early developmental stages that are susceptible to treatment.
The development of anticoccidial agents for use in ruminants and pigs has largely resulted from an extension of development of these compounds for use in the poultry industry. Relatively few of the compounds registered for use in poultry have been tested and found to be effective in ruminants and pigs. Especially in suckling piglets that receive no other source of feed and drink except mother’s milk, prolonged treatment is laborious and impractical in the field.
Anticoccidial compounds are used either prophylactically or therapeutically. The most important anticoccidial drugs for use in cattle, sheep, goats and pigs are listed in (Table 5).
Table 5 Recommended use and dosage of anticoccidial drugs used in cattle, sheep, goats and pigs
| DRUG | USE | CATTLE | SHEEP | GOATS | PIGS | DOSAGE |
| Sulphonamides | ||||||
| Sulphadimidine | Therapeutic | X | X | X |
| 50–100 mg/kg daily for 4 days |
| Sulphaquinoxaline | Prophylactic | X |
|
| 0.5–3 g/lamb/day for 20 days | |
| Ionophores | ||||||
| Salinomycin | Prophylactic |
| X | X |
| 125-167 g/ton of feed for 30 days (premix) |
| Lasalocid | Prophylactic | X | X | X |
| 0,5–1 mg/kg per day in feed for up to 6 weeks |
| Monensin | Prophylactic | X | X |
|
| 20-30 g/ton of feed for 30 days (premix) |
| Thiamine analogues | ||||||
| Amprolium | Prophylactic |
|
| X |
| 50 mg/kg for 5 days |
| Quniates | ||||||
| Decoquinate | Prophylactic | X |
| X |
| 0,5 mg/kg in feed for at least 28 days |
| Triazine triones | ||||||
| Toltrazuril | Metaphylactic | X | X |
| X | 20 mg/kg (single treatment) |
| Diclazuril | Metaphylactic | X | X |
| 1 mg/kg orally (singel treatment) | |
Sulphonamides are commonly used for treatment of coccidiosis in ruminants but are only partially effective. Their concurrent antibacterial activity may contribute to their efficacy by controlling secondary bacterial invaders. Potentiated sulphonamide mixtures have been found to have synergistic activity against coccidia in poultry, but have not been evaluated in ruminants. Gut-active sulphonamides (e.g. succinylsulphathiazole and phthalylsulphathiazole) should not be used in treating coccidiosis as they become active only in the large intestine. In piglets, treatment with sulphonamides is only effective when applied parenterally (as sulphamethoxypyrimidine, 75 mg/kg of body weight, parenteral application) and needs repeated application41 so it must be considered impractical.
The carboxylic ionophores have broad-spectrum activity, are coccidiocidal in action and have a narrow margin of safety.64 They act by facilitating the movement of inorganic cations across cell membranes, thus upsetting the balance of physiologically important ions such as sodium and potassium. Activity is most marked against the sporozoites, but merozoites are also affected. They are therefore best used prophylactically. Salinomycin is registered for the prophylaxis of coccidiosis in small ruminants. The ionophores have a relatively narrow margin of safety, and errors in dosage or feed mixing procedures may result in toxicity.71, 90 They are not recommended for use in neonatal piglets. Monensin is a feed additive for cattle that also has an effect on coccidiosis.97
Oral treatment with nitrofurazone (10 mg/kg per day for five days) is effective against clinical coccidiosis,84 but nitrofurazone is no longer available for use in food-producing animals in many countries due to persistent residues.
The thiamine analogue amprolium has been used against goat coccidiosis.29, 46
The quinate decoquinate completely controlled E. bovis in calves when given in daily doses for three to four weeks, with no relapse after withdrawal of medication.66
Toltrazuril and diclazuril are symmetric triazinetriones, a novel chemical class of anticoccidials. A single dose given to lambs 7 to 10 days after being put to pasture was highly efficacious in preventing clinical coccidiosis. It reduced oocyst output to low levels, prevented the development of diarrhoea and improved weight gain during the four to five weeks after treatment.30 Calves can alsoeffectively be treated with toltrazuril or diclazuril in a single dose.80, 111 Toltrazuril is highly effective against C. suis upon a single oral application before the onset of excretion, while for diclazuril no such efficacy could be shown.69
The level of drug required to be mixed in feed varies according to the amount of feed consumed by the animal each day. Distinction must also be made between full rations, creep feed and supplementary feed when calculating the inclusion rate.
References
- AJAYI, J.A. & TODD, A.C., 1977. Relationships between two levels of aureomycin-sulphamethazine supplementation and acquisition of resistance to ovine coccidiosis. British Veterinary Journal, 133, 166–174.
- BANGOURA, B. & DAUGSCHIES A., 2007. Parasitological and clinical parameters of experimental Eimeria zuernii infection in calves and influence on weight gain and haemogram. Parasitology Research, 100, 1331–1340.
- BARTA, J.R., SCHRENZEL, M.D., CARRENO, R. & RIDEOUT, B.A., 2005. The genus Atoxoplasma (Garnham 1950) as a junior objective synonym of the genus Isospora (Schneider 1881) species infecting birds and resurrection of Cystoisospora (Frenkel 1977) as the correct genus for Isospora species infecting mammals. Journal of Parasitology, 91, 726–727.
- BAUER, C. & BÜRGER, H. J., 1984: Zur Biologie von Eimeria leuckarti (Flesch, 1883) der Equiden. Berlin und Münchener Tierärztliche Wochenschrift 97, 367–372.
- BEMRICK, W.J., O’LEARY, T.P. & BARNES, D.M., 1979. Eimeria leukarti in five Minnesota horses. Veterinary Medicine/Small Animal Clinician, 74, 77–80.
- BERRIATUA, E., GREEN, L. E. & MORGAN, K. L. 1994. A descriptive epidemiological study of coccidiosis in early lambing housed flocks. Veterinary Parasitology, 54, 337–351.
- CALLOW, L., 1984. Animal Health in Australia. Vol 5. Protozoal and Rickettsial Diseases. Melbourne: Australian Government Publishing Service.
- CATCHPOLE, J. & GREGORY, M.W., 1985. Pathogenicity of the coccidium Eimeria crandallis in laboratory lambs. Parasitology, 91, 45–52.
- CATCHPOLE, J. & HARRIS, T.J., 1989. Interaction between coccidia and Nematodirus battus in lambs on pasture. The Veterinary Record, 124, 603–605.
- CATCHPOLE, J., NORTON, C. C. & GREGORY, W. W., 1993. Immunization of lambs against coccidiosis. The Veterinary Record, 16, 56–59.
- CATCHPOLE, J., NORTON, C.C. & JOYNER, L.P., 1975. The occurrence of Eimeria weybridgensis and other coccidia in lambs in England and Wales. British Veterinary Journal, 131, 329–401.
- CATCHPOLE, J., NORTON, C.C. & JOYNER, L.P., 1976. Experiments with defined multi-specific coccidial infections in lambs. Parasitology, 72, 137–147.
- DAI, Y.B., LIU, X.Y., LIU, M. & TAO, J.P., 2006. Pathogenic effects of the the coccidium Eimeria ninakohlyakimovae in goats. Laboratory Medicine, 30, 149–160.
- DAUGSCHIES, A, BURGER, H-J & AKIMARU, M., 1998. Apparent digestibility of nutrients and nitrogen balance during experimental infection of calves with Eimeria bovis. Veterinary Parasitology, 77, 93–102.
- DAUGSCHIES, A., 2005. Parasitäre Erkrankungen. In: HOFMANN, W. Rinderkrankheiten innere und chirurgische Erkrankungen. 2nd edn. Stuttgart: Eugen Ulmer, 324–326.
- DAUGSCHIES, A., BIALEK, R., JOACHIM, A.& MUNDT, H.C., 2001. Autofluorescence microscopy for the detection of nematode eggs and protozoa, in particular Isospora suis, in swine faeces. Parasitology Research 87, 409–412.
- DAUGSCHIES, A., IMAROM, S. & BOLLWAHN, W.,1999. Differentiation of porcine Eimeria spp. by morphologic algorithms. Veterinary Parasitology 81, 201–210.
- DEPLAZES, P., ECKERT J., MATHIS A., VON SAMSON–HIMMELSTJERNA G, ZAHNER, H., 2016. Parasitology in Veterinary Medicine. Wageningen: Academic Publishers.
- DUSZYNSKI D.W, UPTON S.J. & COUCH, L, 2017. The Coccidia of the World. http://biology.unm.edu/coccidia/table.html
- ERNST, J.V. & BENZ, G.W., 1986. Intestinal coccidiosis of cattle. Veterinary Clinics of North America: Food Animal Practice, 2, 283–291.
- FAYER, R., 1980. Epidemiology of protozoan infections: The coccidia. Veterinary Parasitology, 6, 75–103.
- FERNANDO, M.A., 1982. Pathology and pathogenicity. In: LONG, P.L., (ed.). The Biology of the Coccidia. London: Edward Arnold.
- FITZGERALD, P.R. & MANSFIELD, M.E., 1979. Efficacy of lasalocid against coccidia in cattle. Journal of Parasitology, 65, 824–825.
- FOREYT, W.J., 1986. Epidemiology and control of coccidia in sheep. Veterinary Clinics of North America: Food Animal Practice, 2, 383–388.
- FOREYT, W.J., GATES, N.C. & RICH, J.E., 1981. Evaluation of lasalocid in salt against ovine coccidia. American Journal of Veterinary Research, 42, 54–57.
- FOSTER, A.O., CHRISTENSEN, J.F. & HABERMANN, R.T., 1947. Treatment of coccidial infections in lambs with suphaguanidine. Proceedings of the Helminthological Society of Washington, 8, 33–38.
- FRIEND, S.C. & STOCKDALE, P.H., 1980. Experimental Eimeria bovis infection in calves: a histopathological study. Canadian Journal of Comparative Medicine 44, 129–140.
- GABNER, S., WORLICZEK, H.L., WITTER, K., MEYER, F.R., GERNER, W. & JOACHIM, A., 2014. Immune response to Cystoisospora suis in piglets: local and systemic changes in T–cell subsets and selected mRNA transcripts in the small intestine. Parasite Immunology, 36, 277–291.
- GIBBONS, P., LOVE, D., CRAIG, T. & BUDKE, C., 2016. Efficacy of treatment of elevated coccidial oocyst counts in goats using amprolium versus ponazuril. Veterinary Parasitology, 218, 1-4.
- GJERDE, B. & HELLER, O., 1991. Chemoprophylaxis of coccidiosis in lambs with a single oral dose of toltrazuril. Veterinary Parasitology, 38, 97–107.
- GREGORY, M.W. & CATCHPOLE, J., 1987. Ovine coccidiosis: Pathology of Eimeria ovinoidalis infection. International Journal of Parasitology, 17, 1099–1111.
- GREGORY, M.W. & CATCHPOLE, J., 1989. Ovine coccidiosis: Heavy infection in young lambs increases resistance without causing disease. The Veterinary Record, 124, 458–461.
- GREGORY, M.W. & NORTON, C.C., 1986. Caprine coccidiosis. Goat Veterinary Society Journal, 7, 32–34.
- GREGORY, M.W., 1990. Pathology of coccidial infections. In: Long, P.L. (Ed.), Coccidiosis of Man and Domestic Animals. Boca Raton: CRC Press, 235–261.
- GREGORY, M.W., CATCHPOLE, J., PITTILO, R.M. & NORTON, C.C., 1987. Ovine coccidiosis: observations on ‘oocyst patches’ and polyps in naturally acquired infections. International Journal for Parasitology, 17, 1113– 1124.
- GREGORY, M.W., JOYNER, L.P. & CATCHPOLE, 1982. Medication against ovine coccidiosis—A review. Veterinary Research Communications, 5, 307–335.
- GREGORY, M.W., JOYNER, L.P., CATCHPOLE, J. & NORTON, C.C., 1980. Ovine coccidiosis in England and Wales 1978–1979. The Veterinary Record, 106, 461–462.
- HENNING, M.W., 1956. Animal Diseases in South Africa. Pretoria: Central News Agency.
- HOOSHMAND-RAD, P., SVENSSON, C & UGGLA, A., 1994. Experimental Eimeria alabamensis infection in calves. Veterinary Parasitology 53, 23–32.
- HORTON, G.M.J. & STOCKDALE, P.H.G., 1979. Effects of amprolium and monensin on oocyst discharge, feed utilization and ruminal metabolism of lambs with coccidiosis. American Journal of Veterinary Research, 40, 966–970.
- JOACHIM, A. & MUNDT. H.C., 2011. Efficacy of sulfonamides and Baycox® against Isospora suis in experimental infections of suckling piglets. Parasitology Research, 109, 1653–1659.
- JOHNSON J. & REID W.M., 1970. Anticoccidial drugs: lesion scoring techniques in battery and floor–pen experiments with chickens. Experimental Parasitology, 28, 30–36.
- JUBB, K.V.F., KENNEDY, P.C. & PALMER, N., 1985. Pathology of Domestic Animals. 3rd edn. Orlando: Academic Press.
- KANYARI, W. N., 1993. The relationship between coccidial and helminth infections in sheep and goats in Kenya. Veterinary Parasitology, 51, 137–141.
- KELLERMAN, T.S., COETZER, J.A.W. & NAUDÉ, T.W., 1988. Plant Poisonings and Mycotoxicoses of Livestock in Southern Africa. Cape Town: Oxford University Press Southern Africa.
- KIMBITA, E.N., SILAYO, R.S., MWEGA. E.D., MTAU, A.T. & MROSO, J.B., 2009. Studies on the Eimeria of goats at Magadu Dairy Farm SUA, Morogoro, Tanzania. Tropical Animal Health Production, 41, 1263-1265.
- KOUDELA, B. & KUCEROVÁ, S., 1999. Role of acquired immunity and natural age resistance on course of Isospora suis coccidiosis in nursing piglets. Veterinary Parasitology, 82, 93–99.
- KOUDELA, B. & VÍTOVEC, J., 1998. Biology and pathogenicity of Eimeria neodebliecki, in experimentally infected pigs. Parasitology International 47, 249–256.
- LALONDE, L.F. & GAJADHAR, A.A., 2011. Detection and differentiation of coccidian oocysts by real–time PCR and melting curve analysis. Journal of Parasitology, 97, 725–730.
- LE SUEUR, C., MAGE, C., MUNDT, H.C., 2009. Efficacy of toltrazuril (Baycox 5% suspension) in natural infections with pathogenic Eimeria spp. in housed lambs. Parasitology Research, 104, 1157–1162.
- LEEK, R.G., FAYER, R. & MCLOUGHLIN, K.K., 1976. Effect of monensin on experimental infections of Eimeria ninakohlyakimovae in lambs. American Journal of Veterinary Research, 33, 339–343.
- LEVINE, N.D., 1985. Veterinary Protozoology. Ames: Iowa State University Press.
- LEVINE, N.D., & IVENS, V., 1986. The Coccidian Parasites (Protozoa, Apicomplexa) of Artiodactyla. Illinois Biological Monographs 55. Urbana, Chicago: University of Illinois Press.
- LIMA, J.D., 1979. Eimeria caprina sp. n. from the domestic goat, Capra hircus, from the USA. Journal of Parasitology, 65, 902–903.
- LINDSAY, D. S., BLACKBURN, B. L. & POWE, T. A., 1992. Enteric coccidial infections and coccidiosis in swine. Compendium on Continuing Education for the Practicing Veterinarian, 14, 698–702.
- LINDSAY, D.S., HOUK, A. E., MITCHELL, S.M. & DUBEY, J.P., 2014. Developmental biology of Cystoisospora (Apicomplexa: Sarcocystidae) monozoic tissue cysts. Journal of Parasitology 100, 392–398.
- LONG, P.L. & JOYNER, L.P., 1984. Problems in the identification of species of Eimeria. Journal of Protozoology, 31, 535–541.
- MACPHERSON, J. M. & GAJADHAR, A. A., 1993. Differentiation of seven Eimeria species by random amplified polymorphic DNA. Veterinary Parasitology, 45, 257–266.
- MARATEA, K.A. & MILLER, M.A., 2007. Abomasal coccidiosis associated with proliferative abomasitis in a sheep. Journal of Veterinary Diagnostic Investigation, 19, 118–121.
- MARLOW, C.H.B., 1968. Amprolium as a coccidiostat for Angora goats. Journal of the South African Veterinary Medical Association, 39, 93.
- MARQUARDT, W.C., 1981. Host and site specificity in the coccidia: A perspective. Journal of Protozoology, 28, 243–244.
- MATUSCHKA, F.R. & HEYDORN, A.O., 1980. Die Entwicklung von Isopora suis Biester 1934 (Sporozoa: Coccidia: Eimeriidae) im Schwein. Zoologische Beiträge, 26, 405–476.
- MCCULLY, R.M., BASSON, P.A., DE VOS, V. & DE VOS, A.J., 1970. Uterine coccidiosis of the impala caused by Eimeria neitzi spec. nov. Onderstepoort Journal of Veterinary Research, 37, 45–58.
- MCDOUGALD, L., 1990. Control of coccidiosis: chemotherapy. In: LONG, P.L. (ed.) Coccidiosis of Man and Domestic Animals. Boca Raton: CRC Press, 307–320.
- MEYER, C., JOACHIM, A. & DAUGSCHIES, A., 1999. Occurrence of Isospora suis in larger piglet production units and on specialized piglet rearing farms. Veterinary Parasitology, 82, 277–284.
- MINER, M.L. & JENSEN J. B., 1976. Decoquinate in the control of experimentally induced coccidiosis of calves. American Journal of Veterinary Research, 37, 1043–1045.
- MINISTRY OF AGRICULTURE, FISHERIES AND FOOD, 1987. Manual of Veterinary Parasitological Laboratory Techniques. Technical Bulletin No. 18. Her Majesty’s Stationary Office, London.
- MITCHELL, G. & LINKLATER, K., 1983. Differential diagnosis of scouring in lambs. In Practice, 5, 4–12.
- MUNDT, H.C., MUNDT-WÜSTENBERG, S., DAUGSCHIES, A. & JOACHIM, A., 2007. Efficacy of various anticoccidials against experimental porcine neonatal isosporosis. Parasitology Research 100, 401–411.
- NARCROSS, M.A., SIEGMUND, O.H. & FRASER, C.M., 1974. Amprolium for coccidiosis in cattle. Veterinary Medicine and Small Animal Clinician, 69, 462–465.
- NATION, P.W., CROWE, S.P. & HARRIS, W.N., 1982. Clinical signs and pathology of accidental monensin poisoning in sheep. Canadian Veterinary Journal, 23, 323–326.
- NIESTRATH, M., TAKLA, M., JOACHIM, A. & DAUGSCHIES, A., 2002. The role of Isospora suis as a pathogen in conventional piglet production in Germany. Journal of Veterinary Medicine Series B: Infectious Diseases and Veterinary Public Health, 49, 176–180.
- NIILO, L., 1970. Bovine coccidiosis in Canada. Canadian Veterinary Journal, 11, 91–98.
- NORTON, C.C., 1986. Coccidia of the domestic goat Capra hircus, with notes on Eimeria ovinoidalis and E. bakuensis (syn. E. ovina) from the sheep Ovis aries. Parasitology, 92, 279–289.
- NORTON, C.C., JOYNER, L.P. & CATCHPOLE, J., 1974. Eimeria weybridgensis sp. nov. and Eimeria ovina from the domestic sheep. Parasitology, 69, 87–95.
- OETJEN,B.D. & DU LAC, F., 1993. Management of coccidiosis in dairy calves and replacement heifers. Compendium on Continuing Education for the Practicing Veterinarian, 15, 891–895.
- PARKER, R.J., JONES, G.W., ELLIS, K.J., HEATER, K.M., SCHROTER, K.L., TYLER, R. & HOLROYD, R.G., 1986. Post-weaning coccidiosis in beef calves in the dry tropics: Experimental control with continuous monensin supplementation via intraruminal devices. Tropical Animal Health and Production, 18, 198–208.
- PELLÉRDY, L. P., 1974. Coccidia and Coccidiosis. 2nd edition, Berlin: Paul Parey.
- PENZHORN, B. L. & SWAN, G. E., 1986. Coccidiosis. In: HOWARD, J.L. (ed.). Current Veterinary Therapy 3 Food Animal Practice. Philadelphia: W. B. Saunders.
- PHILIPPE, P., ALZIEU, J.P., TAYLOR, M.A. & DORCHIES, P., 2016. Comparative efficacy of diclazuril (Vecoxan®) and toltrazuril (Baycox bovis®) against natural infections of Eimeria bovis and Eimeria zuernii in French calves. Veterinary Parasitology, 206, 129-137.
- PLITT, A., IMAROM, S., JOACHIM, A. & DAUGSCHIES, A., 1999. Interactive classification of porcine Eimeria spp. by computer-assisted image analysis. Veterinary Parasitology, 86, 105–112.
- POUT, D.D., 1969. Coccidiosis of sheep. A critical review of the disease. Veterinary Bulletin, 39, 609–618.
- POUT, D.D., 1973. Coccidiosis of lambs. I. Observations on the naturally acquired infection. British Veterinary Journal, 129, 555–566.
- POUT, D.D., 1974. Coccidiosis of lambs. III. The reaction of the small intestinal mucosa to experimental infections with E. arloingi ‘B’ and E. crandallis. British Veterinary Journal, 130, 45–53.
- POUT, D.D., OSTLER, D.C., JOYNER, L.P. & NORTON, C.C., 1966. The coccidial population in clinically normal sheep. The Veterinary Record, 78, 455–460.
- RADOSTITS, O.M. & STOCKDALE, P.H.G., 1980. A brief review of bovine coccidiosis in Western Canada. Canadian Veterinary Journal, 21, 227–230.
- ROSE, M.E., 1987. Immunity to Eimeria infections. Veterinary Immunology and Immunopathology, 17, 333–343.
- RUTTKOWSKI, B., JOACHIM, A. & DAUGSCHIES, A. 2001. PCR-based differentiation of three porcine Eimeria species and Isospora suis. Veterinary Parasitology 95, 17–23.
- SCHWARZ, L., WORLICZEK, H.L., WINKLER, M. & JOACHIM, A., 2014. Superinfection of sows with Cystoisospora suis ante partum leads to a milder course of cystoisosporosis in suckling piglets. Veterinary Parasitology, 204, 158–168.
- SCHWEITZER, D., KIMBERLING, C. & SPRAKER, T., 1984. Accidental monensin sodium intoxication of feedlot cattle. Journal of the American Veterinary Medical Association, 184, 1273–1276.
- SOMER, C., 1998. Quantitative characterization, classification and reconstruction of oocyst shapes of Eimeria species from cattle. Parasitology, 116, 21–28.
- SOULSBY, E.J.L., 1982. Helminths, Arthropods and Protozoa of Domesticated Animals. 7th edition. Philadelphia: Lea & Febiger.
- STEWART, C.G., 1991. Medical University of Southern Africa, Medunsa South Africa. Personal observations.
- STOCKDALE, P.H.G., 1977. Proposed life cycle of Eimeria zuernii. British Veterinary Journal, 133, 471–473.
- STOCKDALE, P.H.G., BAINBOROUGH, C.B., BAILEY, C.B. & NIILO, L., 1981. Some pathophysiological changes associated with infection of Eimeria zuernii in calves. Canadian Journal of Comparative Medicine, 45, 34–37.
- STOCKDALE, P.H.G., SHEARD, A. & TIFFIN, G.B., 1982. Resistance to Eimeria bovis produced after chemotherapy of experimental infections in calves. Veterinary Parasitology, 9, 171–177.
- STROMBERG, B.E., SCHLOTTHAUER, J.C., HAMANN, K.J., SAATARA OZ, H. & BEMRICK, W.J. 1986. Experimental bovine coccidiosis: control with monensin. Veterinary Parasitology, 22, 135–140.
- STUART, B.P. & LINDSAY, D.S., 1986. Coccidiosis of swine. Veterinary Clinics of North America: Food Animal Practice, 2, 455–468.
- SÜHWOLD, A., HERMOSILLA, C., SEEGER, T., ZAHNER, H. & TAUBERT, A., 2010. T cell reactions of Eimeria bovis primary and challenge-infected calves. Parasitology Research, 106, 595–605.
- SVENSSON, C., 1997. The survival and transmission of oocysts of Eimeria alabamensis in hay. Veterinary Parasitology, 69, 211–218.
- SVENSSON, C., HOOSHMAND-RAD, P., PEHRSON, B., TORNQUIST M. & UGGLA, A., 1993. Excretion of Eimeria oocysts in calves during their first three weeks after turn-out to pasture. Acta Vetenaria Scandinavia, 34, 175– 182.
- SVENSSON, C., OLOFSSON, H. & UGGLA, A., 1996. Immunisation of calves against Eimeria alabamensis coccidiosis. Applied Parasitology, 37, 209–216.
- SVENSSON, C., UGGLA, A. & PEHRSON, B., 1994. Eimeria alabamensis infection in calves at pasture. Veterinary Parasitology, 53, 33–43.
- TARLATZIS, C., PANETSOS, A. & DRAGONAS, P., 1957. Further experiences with furacin in treatment of ovine and caprine coccidiosis. Journal of the American Veterinary Medical Association, 131, 474.
- TAYLOR, M.A., COOP, R. & WALL, R.L., 2007. Veterinary Parasitology (3rd edition). Blackwell Publishing, London.
- TENTER, A., 2006. Parasitosen der Wiederkäuer (Rind, Schaf, Ziege). In: SCHNIEDER (ed.): Veterinärmedizinische Parasitologie. 6th edition. Belrn: Paul Parey, 119–165.
- TORRES A., 2004. Prevalence study of Isospora suis in Europe. In: 18th International Pig Veterinary Society Congress. Hamburg, Germany.
- TUBBS, R.C., 1986. A review of porcine neonatal coccidiosis. Modern Veterinary Practice, 67, 899–903.
- VAN TONDER, E.M., 1975. Notes on some disease problems in Angora goats in South Africa. Veterinary Medical Review, 1/2, 109–138.
- YANG, R., JACOBSON, C., GARDNER, G., CARMICHAEL, I., CAMPBELL, A.J. & RYAN, U., 2014. Longitudinal prevalence, oocyst shedding and molecular characterisation of Eimeria species in sheep across four states in Australia. Experimental Parasitology, 145, 14–21.
- ZECHNER, G., BAUER C., JACOBS, J., GOOSSENS, L., VERTENTEN, G. & TAYLOR, M.A., 2015. Efficacy of diclazuril and toltrazuril in the prevention of coccidiosis in dairy calves under field conditions. The Veterinary Record, 176, 126.