- Infectious Diseases of Livestock
- Part 1
- Dourine
- Vectors: Ticks
- Vectors: Tsetse flies
- Vectors: Muscidae
- Vectors: Tabanidae
- Vectors: Culicoides spp.
- Vectors: Mosquitoes
- Classification, epidemiology and control of arthropod-borne viruses
- Special factors affecting the control of livestock diseases in sub-Saharan Africa
- The control of infectious diseases of livestock: Making appropriate decisions in different epidemiological and socioeconomic conditions
- Infectious diseases of animals in sub-Saharan Africa: The wildlife⁄livestock interface
- Vaccination: An approach to the control of infectious diseases
- African animal trypanosomoses
- Dourine
- Trichomonosis
- Amoebic infections
- GENERAL INTRODUCTION: COCCIDIA
- Coccidiosis
- Cryptosporidiosis
- Toxoplasmosis
- Besnoitiosis
- Sarcocystosis
- Balantidiosis
- Leishmaniosis
- Neosporosis
- Equine protozoal myeloencephalitis
- GENERAL INTRODUCTION: BABESIOSES
- Bovine babesiosis
- Equine piroplasmosis
- Porcine babesiosis
- Ovine babesiosis
- GENERAL INTRODUCTION: THEILERIOSES OF CATTLE
- East Coast fever
- Corridor disease
- Zimbabwe theileriosis
- Turning sickness
- Theileria taurotragi infection
- Theileria mutans infection
- Theileria annulata theileriosis
- Theileriosis of sheep and goats
- Theileria buffeli⁄orientalis infection
- Non-pathogenic Theileria species in cattle
- GENERAL INTRODUCTION: RICKETTSIAL, CHLAMYDIAL AND HAEMOTROPIC MYCOPLASMAL DISEASES
- Heartwater
- Lesser known rickettsial infections in animals and humans
- Chlamydiosis
- Q fever
- Eperythrozoonosis
- Bovine Haemobartonellosis
- Potomac horse fever
- GENERAL INTRODUCTION: ANAPLASMOSES
- Bovine anaplasmosis
- Ovine and caprine anaplasmosis
Dourine
This content is distributed under the following licence: Attribution-NonCommercial CC BY-NC  View Creative Commons Licence details here
View Creative Commons Licence details here
Introduction
Dourine is a chronic contagious disease of horses, mules and donkeys. It is characterized by swelling of the external genitalia and subcutaneous tissues of the ventral abdomen, cachexia and, in some cases, involvement of the nervous system. The disease is caused by the venereally transmitted protozoan parasite Trypanosoma equiperdum.
Trypanosoma equiperdum is essentially a tissue parasite which has developed the ability to invade and survive in the genital tract, thus enabling direct transmission from one host to another. The disease consequently differs from other African trypanosomoses in that it does not require an arthropod vector for transmission.
Dourine was described as early as AD 400 by Chiron, a Byzantine veterinarian,60 and is probably one of the oldest recognized diseases of horses. The disease was known to the Arabs and horsemen of North Africa for centuries before it was reported in Europe in the late eighteenth century.30 The dissemination of Arab horses for breeding purposes may have introduced dourine to Europe, from where it spread to many parts of the world. Rouget, in 1894, was the first to demonstrate a trypanosome in the blood of an infected horse,35 while the first experimental transmission of the parasite occurred five years later.56 The organism was named Trypanosoma equiperdum by Doflein in 1901.18
Control measures based on clinical identification,35 and later by serological testing,66 have eliminated the disease from many countries. Intensive campaigns led to its eradication in Canada66 and the final pockets of infection in the USA were eliminated in 1949.36 By 1960 most European countries were free of the disease.19 Outside southern Africa dourine still occurs in certain parts of North Africa, Russia, the Middle East, and Burma.19 Recent outbreaks have occurred in Italy.9
Dourine was known to farmers in Griqualand West in South Africa for several years before it was officially recorded for the first time in 1914.64 During the same year the disease was reported from Namibia by Maag as cited by Schulz.60
A shipment of horses from Germany to Namibia may have brought the disease to the subcontinent.33 Schulz believed that dourine was brought into South Africa by donkey jacks imported from Canada or by transport horses returning from Namibia during the Herero wars.58 Dourine has a scattered distribution in Namibia, Botswana, Swaziland, Lesotho and South Africa.8, 19, 20, 68
Although the disease is not of great economic importance in South Africa, its continued presence necessitates strict surveillance of horses intended for breeding and export purposes.
Aetiology
Trypanosoma equiperdum is a salivarian trypanosome of the subgenus Trypanozoon, which includes Trypanosoma evansi and the Trypanosoma brucei complex.17 The mammalian blood forms of the three species have similar morphology and share common somatic antigens, which prevent their distinction by most serological tests.52, 70 Trypanosoma equiperdum is closely related to T. evansi, the causative organism of surra, which is endemic in North Africa,28 and it is possible that they are derived from a common ancestor descended from T. brucei.12 Evidence that they might belong to the same species comes from isoenzyme characterization that has shown isolates of T. equiperdum and T. evansi from different geographical regions form a homogeneous group,39 and analysis of kinetoplast DNA, nuclear DNA and molecular karyotyping has confirmed the closeness of their phylogenetic relationships. 39, 40, 41, 71 A major difference between the two species is in the presence of maxicircles in the DNA of T. equiperdum and their absence in T. evansi,51 so it is unlikely that T. equiperdum could have originated from T. evansi, as suggested by Hoare,28 as this would have required the reacquisition of the maxicircles.
Trypanosoma equiperdum is monomorphic and typically occurs as slender forms 15,5 to 36 μm long, with a distinct free flagellum and a subterminal kinetoplast.Occasional stumpy forms are seen.29
The organism is only found naturally in the definitive host and multiplies asexually by binary fission.Trypanosoma equiperdum strains have been maintained for experimental purposes in the laboratory in animals such as mice, rats and guinea pigs for many generations.49, 66 These strains have enhanced virulence and produce acute disease when used to infect horses.49 They have a tendency to produce dyskinetoplastic strains, particularly in rodents.30
Although T. equiperdum is essentially a tissue parasite, parasitaemia does occur 21 to 23 days after natural infection and may persist for three to four months. Thereafter it is difficult to demonstrate the parasite in the blood of infected horses.6, 7
For many years, T. equiperdum could only be maintained by serial passage in laboratory animals or developing chick embryos.44 Recently an in vitro culture system has been developed in which a laboratory strain ofT. equiperdum can be continuously maintained in a semi-defined medium, whilst still retaining its morphological and biochemical characteristics. 5, 47 These in vitro systems have been utilized in studies on the efficacy of trypanocidal drugs11, 72 and could be used also in the study of the physiology, biochemistry and antigenic variation of the parasite.
Epidemiology
Dourine occurs in horses, mules and donkeys. Improved breeds of horses such as Thoroughbreds tend to be more susceptible to the disease than native horses, donkeys and mules, which are often subclinically or only mildly affected. 66 It is a true venereal disease. Natural transmission of the organism occurs during genital contact between animals, and infection may occur from stallion to mare or vice versa. Biological transmission of T. equiperdum by tsetse flies has not been reported but the disease can be transmitted by subinoculating large volumes of blood from infected horses to susceptible horses and donkeys.6, 49 The disease is not limited to certain environmental conditions,62 as is usually the case with the tsetse fly-transmitted trypanosomoses.
The presence of dourine in a herd can usually be traced to an infected animal which showed no clinical signs at the time of introduction. Due to the insidious nature of the disease, it may take several months before clinical signs appear and by this time the disease may already be widespread in a herd. Many infected animals are subclinical carriers26, 58 and they can only be reliably detected by serological methods. Sexual activity is impeded only in cases showing extreme emaciation or severe oedema of the genitalia.
The disease is more likely to spread in herds or areas where indiscriminate breeding of horses and mules occurs. Donkeys used for the breeding of mules may be particularly important in this respect. Mule foals may become infected in the same way as horse and donkey foals (see below).
Transmission of the disease is usually limited to the breeding season when the parasite is transmitted from an infected stallion to a number of mares.58 Trypanosomes may be numerous in the vaginal discharges of affected mares or in the urethras of stallions, particularly during the early stages of the disease. Although transmission is erratic and will not occur at every mating, it is more likely to occur when a recently infected animal is involved.6, 49 In South Africa, the disease is seldom seen in Thoroughbred horses, as breeding of these animals is usually done under strict supervision. Great care is taken to test breeding animals before or during the breeding season.
Occasionally foals become infected — possibly during parturition (from vaginal discharges) or by drinking the milk of an infected dam.58 These foals remain serologically positive and may transmit the disease when sexually mature.54 Non-infected foals may also become serologically positive by ingesting colostrum of seropositive dams, but they lose the antibodies within five to six months and are not infective.15
Pathogenesis
Most pathological effects of T. equiperdum infection in the host are characteristic of trypanosome infections in general, particularly those of the T. brucei complex (see African animal trypanosomoses). Natural transmission of the disease occurs when the parasite is deposited on the mucous membranes of the genitalia during coitus. Like the other members of the T. brucei complex, T. equiperdum has the ability to invade intact mucous membranes and lymphatic vessels but also has a predilection for connective tissue.22
Experimental infection of laboratory animals has shown that the major route taken by a laboratory strain of T. equiperdum from an intradermal inoculation site to other locations was via the peripheral lymphatic vessels. It appears that organisms reach the regional lymph node via the afferent lymphatic vessels, from where they reach the bloodstream. Only after some time do they leave the node via the efferent lymphatic vessels.63
Trypanosoma equiperdum multiplies predominantly in extracellular tissue spaces. It is seldom found in peripheral blood and probably only uses the bloodstream as a means of transport from one site to another. York, as cited by Goodwin, 22 reported that the nutrient content of extracellular fluid may favour the growth of the parasites. To some extent this site may also aid their evasion of the action of antibodies present in circulating blood.
As in other trypanosomoses, T. equiperdum elicits a marked immune response in the host.7 Antigen-antibody reactions are accompanied by the release of a number of physiologically active substances including histamine and kinin. The latter may be responsible for the vascular changes and oedema after the initial histamine release. The exact cause of the often observed anaemia has not yet been determined. A combination of the resultant local hypoxia and the proteolytic and lysosomal enzymes released (in the host’s bid to contain the infection) probably contribute largely to the extensive damage caused to connective tissues.22
Although during the course of infection trypanosomes are constantly exposed to the immune responses of the host, they are able to evade the deleterious consequences of the immune mechanisms by a process known as antigenic variation. Trypanosomes are covered by a surface coat consisting of a single protein known as the variable surface glycoprotein (VSG). This is a powerful immunogen and elicits the production of protective antibodies, but as the host responds to one VSG, trypanosomes expressing another VSG become dominant. The different VSGs are expressed in a general order, with certain VSGs appearing early in infection and some later (see also African animal trypanosomoses). 4, 55
Immunodepression is a characteristic feature of infections with pathogenic trypanosomes in both domesticated livestock and laboratory hosts,59 and it may well be an important mechanism in the induction of disease. Infection results in reduced primary immune responses to nontrypanosomal antigens and diminished T cell proliferation and cytokine production.14, 59 Complement components are reduced, inappropriate antibody responses are made and there are defects in isotype switching and impaired B cell memory responses.2, 67 In experimental infections with T. equiperdum, a rheumatoid factor-like substance destroying IgG antibodies and an unidentified soluble substance causing functional suppression of T and B lymphocytes have been identified.32, 33 These factors probably predispose the host to secondary infections.23, 53
The onset of the nervous form of the disease appears to coincide with the presence of parasites in the cerebrospinal fluid (CSF). The CSF may represent a favourable environment for the parasite as the immunogobulin levels are much lower than in plasma, inflammatory cells are fewer and glucose levels similar.2 The neuropathology of dourine differs from that described for other trypanosomoses, including T. brucei infection in horses.43 The primary changes in T. equiperdum infections comprise cellular infiltrations of spinal nerves and some major nerve trunks, but without much involvement of the central nervous system. A possible explanation for the site of these lesions is the drainage of CSF which moves along the sheaths of the spinal nerves, i.e. antigens of trypanosomes present in CSF may elicit a host response at these sites where nerve tissue is no longer protected by the blood–brain barrier.6, 7
Passive protection against T. equiperdum infection by transfer of immune serum to susceptible animals has been demonstrated,34 and passive transfer of immunity from infected dams to their offspring may thus occur via colostrum. The efficacy of this protection is, however, limited by the ability of the parasite to rapidly change antigenically during the course of infection. Resistance to reinfection is limited only to homologous strains.61
Clinical signs
Dourine in southern Africa6, 26, 58, 64 is similar to but usually milder than the disease in Europe31, 35 and North America.66 The course of infection and clinical signs vary considerably, depending on the virulence of the strain and the susceptibility of the host. After an incubation period ranging from one week to a few months,26, 30, 58, 62 the diseasemayassume one of three forms, namely an asymptomatic, interstitial or oedematous, or nervous form. The nervous form occurs as a sequel to the interstitial or oedematous form and is invariably fatal.
South African strains of T. equiperdum tend to induce asymptomatic or latent infections, particularly in donkeys.26 These animals remain seropositive for several years and are capable of transmitting the infection. Clinical signs may be precipitated in latent cases by stress such as that caused by unfavourable weather, physical exhaustion and malnutrition. 26, 49, 58 A moderate anaemia is often observed in animals which show no apparent clinical signs, as well as in those with chronic oedematous lesions or nervous signs. 6, 62, 66
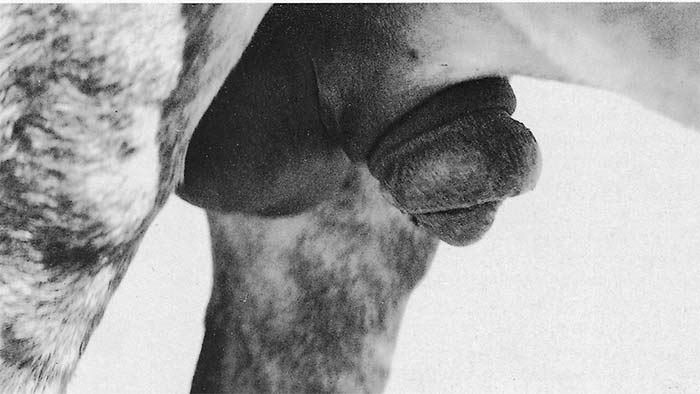
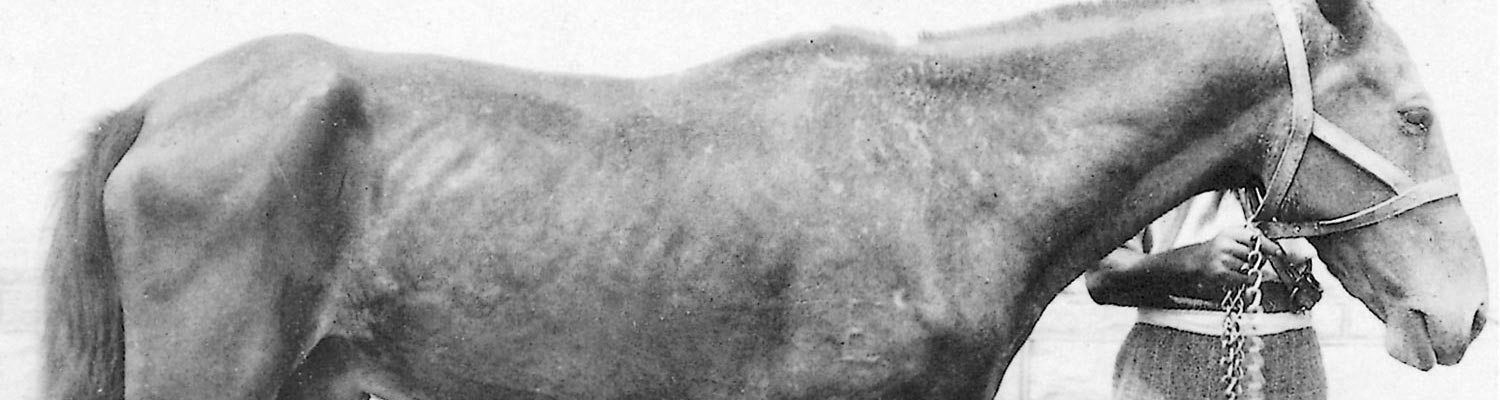
The interstitial or oedematous form is usually a manifestation of chronic infection, lasting several months to several years. A common clinical sign in both stallions and mares is progressive weight loss, with the animal eventually becoming severely emaciated (Figure 13.1). These debilitated animals are prone to infection by other pathogens and parasites, and many die from secondary causes.53 The interstitial form of the disease may or may not be associated with oedematous swellings in the early stages. Stallions show varying degrees of oedematous swelling of the sheath, glans penis and scrotum (Figure 13.2) and, in severe cases, the oedema may extend to the perineum and ventral abdomen and thorax (Figure 13.1). Such swellings are neither hot nor painful and often disappear after a few days, only to recur more severely within two to three weeks. The oedema may persist for up to 18 months after which the swellings often become permanent due to their organization by fibrous connective tissue.6 The penis may protrude and occasionally there is prolapse of the urethra.26 In mares, the vulva and perineum and occasionally the ventral abdomen and udder are swollen. In some cases, a viscous vaginal discharge is seen which may be copious. Hyperaemia of the mucous membranes of the vulva, vagina and clitoris is sometimes accompanied by a few nodules and ulcers which heal leaving white scars. Some mares show frequent micturition and behave as if continually in oestrus. In other countries, signs such as ulceration of the penis, frequent erections, and ocular manifestations such as photophobia, keratitis and corneal opacity have been reported, but these have not been recorded in southern Africa. Raised urticarial skin plaques, 40 to 50 mm in diameter, referred to as ‘silver dollar’ plaques, have only rarely been recorded in animals suffering from dourine in southern Africa.6, 58, 64
Early reports from Europe, North Africa.50 and India described these plaques as being of common occurrence.25 Although not frequently seen in Canada, Watson66 considered them to be pathognomonic.
Nervous disorders occur in some cases and may set in soon after the emaciation or the oedema appear or, alternatively, some time may elapse before their onset. Initially there is acute generalized or localized hyperaesthesia of the skin.31 Later, sensitivity decreases and is followed by paralysis of motor nerves. The motor involvement originates in the perineum and hindlegs and may eventually extend to affect the forelegs. Affected animals become restless and move uneasily from one hindleg to the other. There may be stiffness, weakness or lameness in one or both hindlegs and the animal may have a swaying or straddling gait.
The hindlegs are often set wide apart and the feet are frequently dragged along the ground with knuckling over of the fetlocks. Animals may have difficulty in rising and lying down. Movement becomes increasingly difficult and affected animals are therefore reluctant to do so. Eventually they go down in sternal or lateral recumbency. Although some cases do show evidence of apparent improvement, the condition of animals suffering from the nervous form deteriorates progressively and they invariably die.26
Early reports from Canada and Europe describe facial or recurrent nerve paralysis affecting the lips, nostrils, ears and throat.31, 66 These findings are far less frequent in southern Africa but they have been reported from the Cape provinces.58
Pregnant mares may very rarely abort64 but usually carry their foals to full term.15, 58
Pathology
Animals that die from dourine are usually severely emaciated and anaemic, and changes similar to those found in other wasting diseases are seen. There is subcutaneous oedema, particularly of the perineum, male and female genitalia, and ventral parts of the abdomen and chest. In longstanding chronic cases the oedema is often accompanied by fibrosis of the scrotum, prepuce and tunica vaginalis. In mares vaginitis, metritis and cystitis may be present.58 Numerous cysts occur in the uterus of some mares, and the ovaries are enlarged and show cystic degeneration.58 Most of the internal lymph nodes, but especially those draining the inguinal and pelvic regions, are enlarged, oedematous and firm on palpation. On cut surfaces the affected nodes are pale and there is little differentiation between cortex and medulla.7
In horses that exhibit neurological disturbances, the perineural connective tissues of the larger nerve trunks, particularly those leading to the extremities, are oedematous,7, 31, 58 or the nerves are transformed into fibrous cords fused with the surrounding fascia.31 There may be a serous infiltration around the lumbar and sacral portions of the spinal cord6, 45, 57 and the cord may be softened.26, 66
Microscopically, variable numbers of lymphocytes, plasma cells and macrophages infiltrate the dorsal and ventral spinal nerve roots, spinal nerves, ganglia and meninges of the lumbar and sacral regions, and in more advanced cases, the larger peripheral nerves, notably the sciatic and obturator nerves.6, 7, 31, 45, 46, 66 In cases of longer duration, spinal nerve roots in the thoracic region and the brachial nerves are also affected, although to a lesser extent.6, 7 Earlier workers referred to ‘degenerative changes’, ‘atrophy’ and demyelination of nerve fibres in the spinal and peripheral nerves and the spinal cord,21, 31, 45, 46, 57, 61 but Barrowman6 stated that the lesions in dourine in southern Africa are largely confined to the peripheral nervous system. Marek42 described inflammatory changes in the facial and trigeminal nerves.
Microscopic changes in affected lymph nodes include extensive lymphoid hyperplasia, reticuloendothelial proliferation and varying degrees of lymph stasis, as well as areas of fibrosis in the more chronic cases.7
Diagnosis
The diagnosis of dourine is usually made indirectly, due to difficulty in demonstrating organisms in either the blood or tissues or in discharges from the genitalia. It is generally futile to search for trypanosomes in blood smears because their presence here is transient, although organisms may be found in recently infected animals. If an attempt to demonstrate them is made, a large quantity of citrated blood should be collected and either be centrifuged or allowed to sediment out. The lowest part of the plasma layer is then examined for parasites.6, 49, 64
The clinical signs of dourine are not specific and, as many cases in southern Africa are asymptomatic,6, 7, 49 serology remains the only reliable and practical method of diagnosis. The complement fixation (CF) test has been used for the diagnosis of dourine since 1915 and is still the most commonly used serological technique.27 However, it is only appropriate for making a specific diagnosis in countries in which other members of the T. brucei complex are not present, since antigenic cross-reactions within the group occur. The sera of infected animals give positive CF tests from 20 to 30 days after infection6 and remain positive, with fluctuating titre, for up to 10 years.66 Sera from donkeys and mules are more frequently anticomplementary than are those of horses, which are rarely so.68
Other serological tests for the diagnosis of dourine have been applied experimentally.13 These are the enzymelinked immunosorbent assay (ELISA), and the radio immunoassay, immuno-electro-osmophoresis, and fluorescent antibody tests. The results obtained show that the CF test is still the most reliable although the ELISA has a satisfactory concordance ratio with the CF test. Discrepancies encountered with the other tests are attributed to many causes, including the subjectivity of test interpretation and differences in antibody populations detected by the different tests. Caporale et al.,13 however, consider that the ELISA is the method of choice as the antigen is used in minute quantities and appears to be more stable than the CF test antigen. This was supported by the work of Wassall and his co-workers,65 who concluded that the CF test was less sensitive than either the indirect fluorescent antibody test or the ELISA. They considered that ELISA was the test most likely to lend itself to standardization, allow considerable automation and could be adopted eventually as an internationally approved test for equine health certification for freedom from dourine. ELISAs based on Trypanozoon group-specific antigen have also shown promise for detecting antibodies to pathogenic trypanosomes, including T. equiperdum,10, 38 and tests based on defined antigens could more readily enable standardization than tests based on the use of crude, sonicated antigen preparations. Enzyme- linked immunosorbent assays for antigen detection (Ag-ELISA) have also shown promise experimentally10 and both Ab- and Ag-ELISA tests have been used to investigate the prevalence of T. equiperdum in horses in Ethiopia.1 A commercial card agglutination test for trypanosomosis, using formalin-fixed T. evansi, is available3 and might be useful in the diagnosis of equine trypanosomosis. The test has not been evaluated for use in the diagnosis of T. equiperdum, but a similar test, based on T. brucei gambiense antigen, has been shown to detect antibodies in horses that show clinical signs of dourine.53
Diagnosis by subinoculation of blood into experimental animals is not practical as the southern African strains of T. equiperdum do not transfer readily to dogs, rabbits, rats or other laboratory animals.6, 26, 64 Infection of these laboratory animals would occur more readily after passage in equine animals, but this method is rarely used for the diagnosis of the disease.,25, 49
Differential diagnosis
In southern Africa, oedema of the genitalia in horses may occur in other conditions, e.g. nagana (caused by T. brucei); equine viral arteritis, which was recently diagnosed in South Africa; coital exanthema; purpura haemorrhagica, where, in addition, extensive petechial and ecchymotic haemorrhages may occur on the skin and mucous membranes; and trauma of the genitalia.
Conditions leading to chronic weight loss and emaciation, such as malnutrition, verminosis, dental pathology, other chronic infections (e.g. trypanosomosis) and chronic liver disease (e.g. that occurring in seneciosis) can be confused with, or mask, the signs of dourine. The nervous signs associated with dourine should be differentiated from similar signs recognized in a variety of infectious and non-infectious causes, such as equid herpesvirus-1 encephalomyelitis, cervical stenotic myelopathy (‘wobbler syndrome’), injuries to the back and pelvis, and iliac thrombosis.16
Control
Several chemotherapeutic drugs, particularly homidium, suramin, quinapyramine and some melaminyl antimony compounds may have some beneficial effect in clinically affected animals, but none have been proven effective in the sterilization of the infection.7, 70 The efficacy of most trypanocidal drugs is limited by their inability to cross the blood–brain barrier. The use of chemotherapy for the control of dourine is thus not recommended as treated animals may show clinical improvement but remain carriers of the parasite.
In most countries dourine is a scheduled disease and control measures are enforced through legislation. In countries free of the disease, the issue of import permits for domestic equids is subject to certification by an official laboratory of negative serological results (usually the CF test) for dourine.
There is no legislation restricting the movement of horses within the borders of South Africa and Namibia as far as dourine is concerned. In these countries all outbreaks of the disease are notifiable by law, but compulsory testing of equine animals (all stallions and maiden and barren mares under the jurisdiction of the Jockey Club of South Africa must be tested for dourine prior to the commencement of each breeding season) to control or eradicate the disease is not enforced. Serological testing of animals is performed at the request of the owners and is subject to them signing an indemnity form which requires them to have all positive animals destroyed, castrated or ovariectomized. Neutering may lead to clinical improvement but animals remain infected. 58 Castration of infected stallions is not always effective in preventing them from transmitting the disease, as they may still serve mares for some time.
The presence of fences may help to control the spread of the disease, although stallions have been reported to serve mares over fences, even those constructed of barbed wire.
Due to the presence of variable surface antigens on T. equiperdum, the development of an effective vaccine has not been achieved.24
References
- ALEMU, T., LUCKINS, A.G., PHIPPS, L.P., REID, S.W.J. & HOLMES, P.H., 1997. The use of enzyme-linked immunosorbent assays to investigate the prevalence of Trypanosoma equiperdum in Ethiopian horses. Veterinary Parasitology, 71, 239–250.
- AUTHIE, E. & POBEL, T., 1990. Serum haemolytic complement activity and C3 levels in bovine trypanosomiasis under natural conditions of challenge—early indications of individual susceptibility to disease. Veterinary Parasitology, 35, 43–59.
- BAJYANA-SONGA, E., HAMERS-CASTERMAN, C., HAMERS, R., PHOLPARK, M., PHOLPARK, S., LEIDL, K., TANGCHAITRONG, S., CHAICHANOPOONPOL, I., VITOORAKOOL, C. & THIRAPATASKUM, T., 1987. The use of a card aglutination test (Testryp CATT) for use in detection of T. evansi infection: a comparison with other trypanosomiasis diagnostic tests under field conditions in Thailand. Annales de la Societe Belge de Medicine Tropicale, 67, 137–148.
- BALTZ, T., GIROUD, C., BRINGAUD, F., EISEN, H., JACQUEMOT, C. & ROTH, C.W., 1991. Exposed epitopes on a Trypanosoma equiperdum variant surface glycoprotein altered by point mutations. Embo Journal, 110, 1653–1659.
- BALTZ, T., BALTZ, D., GIROUD, C. & CROCKETT, J., 1985. Cultivation in a semi-defined medium of animal infective forms of Trypanosoma brucei, T. equiperdum, T. evansi, T. rhodesiense and T. gambiense. The European Molecular Biological Organisation Journal, 4, 1273–1277.
- BARROWMAN, P.R., 1976. Observations on the transmission, immunology, clinical signs and chemotherapy of dourine (Trypanosoma equiperdum) in horses, with special reference to cerebro-spinal fluid. Onderstepoort Journal of Veterinary Research, 43, 55–66.
- BARROWMAN, P.R., 1977. Studies on Trypanosoma equiperdum infection in horses and the immunopathology of Trypanosoma brucei infection in sheep. DVSc thesis, University of Pretoria.
- BARROWMAN, P.R. & VAN VUUREN, M., 1976. The prevalence of dourine in southern Africa. Journal of the South African Veterinary Association, 47, 83–85.
- BELLANI, L., PAPALIA, S. & CAPORALE, E.P., 1979. Report on dourine epidemiological surveillance and research in Italy. In: FAO Report of the Expert Consultation on Research on Trypanomosiasis, 1–5 October 1979. FAO, Rome.
- BISHOP, S., PHIPPS, P. & LUCKINS, A.G., 1995. Trypanosoma equiperdum detection of trypanosomal antibodies and antigen by enzyme-linked immunosorbent assay. British Veterinary Journal, 151, 715–720.
- BRUN, R. & LUN, Z.R., 1994. Drug sensitivity of Chinese Trypanosoma evansi and Trypanosoma equiperdum isolates. Veterinary Parasitology, 52, 37–46.
- BRUN, R., HECKER, H. & LUN, Z.R., 1998. Trypanosoma evansi and T. equiperdum: distribution, biology, treatment and phylogenetic relationship (a review). Veterinary Parasitology, 79, 95–107.
- CAPORALE, V.P., BIANCIFIORI, F., FRESCURA, F., DI MATTEO, A., NANNINI, D. & URBANI, G., 1981. Comparison of various tests for the serological diagnosis of Trypanosoma equiperdum infection in the horse. Comparative Immunology, Microbiology and Infectious Diseases, 4, 243–246.
- DE BAETSELIER, P., 1996. Mechanisms underlying trypanosome-induced T-cell immunosuppression. In: MUSTAFA, A.S., AL-ATTIYAH, R.J., NATH, I. & CHUGH, T.D., (eds). T-Cell Subsets and Cytokines Interplay in Infectious Diseases. Basel, Switzerland: S. Karger. pp. 124–139.
- DE KOCK, G., ROBINSON, E.M. & PARKIN, B.S., 1939. Some observations on dourine. Journal of the South African Veterinary Medical Association, 10, 44–55.
- DELAHUNTA, A., 1983. Veterinary Neuroanatomy and Clinical Neurology. 2nd edn. Philadelphia, London: W.B. Saunders Company.
- DE RAADT, P. & SEED, J.R., 1977. Trypanosomes causing disease in man in Africa. In: KREIER, J.P., (ed.). Parasitic Protozoa. New York, San Francisco, London: Academic Press.
- DOFLEIN, F., 1901. Die Protozoen als Parasiten and Krankheitserreger: Jena. Fischer.
- FAO, 1986. FAO-WHO-OIE Animal Health Year Book. FAO Animal Production and Health Series. Rome: FAO.
- FAUL, A., 1988. Dourine in South Africa. Journal of the South African Veterinary Association, 59, 7.
- FORMAD, J., 1919. Pathology of dourine with special reference to the microscopic changes in nerve tissue and other structures. Journal of Agricultural Research, 18, 145–154.
- GOODWIN, L.G., 1970. The pathology of African trypanosomiasis. Transactions of the Royal Society of Tropical Medicine and Hygiene, 64, 797–817.
- GOODWIN, L.G., GREEN, D.G., GUY, M.W. & VOLLER, A., 1972. Immunosuppression during trypanosomiasis. British Journal of Experimental Pathology, 53, 40.
- GRAY, A.R., 1976. Immunological research and the problem of immunization against African trypanosomiasis. Transactions of the Royal Society of Tropical Medicine and Hygiene, 70, 118–121.
- HAIG, D.A. & LUND, A.S., 1948. Transmission of the South African strain of dourine to laboratory animals. Onderstepoort Journal of Veterinary Science and Animal Industry, 23, 59–61.
- HENNING, M.W., 1956. Animal Diseases in South Africa. 3rd edn. South Africa: Central News Agency, Ltd.
- HERR, S., HUCHZERMEYER, H.F.K.A., TE BRUGGE, L.A., WILLIAMSON, C.C., ROOS, J.A. & SCHIELE, G.J., 1985. The use of a single complement fixation test technique in bovine brucellosis, Johne’s disease, dourine, equine piroplasmosis and Q-fever serology. Onderstepoort Journal of Veterinary Research, 52, 279–282.
- HOARE, C.A., 1967. Evolutionary trends in mammalian trypanosomes. Advances in Parasitology, 5, 47–85.
- HOARE, C.A., 1970. Systematic description of the mammalian trypanosomes of Africa. In: MULLIGAN, H.W., (ed.). The African Trypanosomiases. London: George Allen & Unwin.
- HOARE, C.A., 1972. The Trypanosomes of Mammals. A Zoological Monograph. Oxford: Blackwell Scientific Publications.
- HUTYRA, F. & MAREK, J., 1926. Special Pathology and Therapeutics of the Diseases of Domestic Animals. 3rd edn. London: Baillière, Tindall & Cox.
- KLEIN, F., MATTERN, P. & KORNMAN-VAN DEN BOSCH, H.J., 1970. Experimental induction of rheumatoid factor-like substance in animal trypanosomiasis. Clinical and Experimental Immunology, 7, 851–863.
- KNUTH, P. & DU TOIT, P.J., 1921. Handbuch der Tropen Krankheiten. Leipzig: Barth.
- KUHN, L.R., 1938. The effect of splenectomy and blockade on the protective titre of antiserum against Trypanosoma equiperdum. Journal of Infectious Diseases, 63, 217–224.
- LAVERAN, A. & MESNIL, F., 1904. Trypanosomes and Trypanosomiasis. English translation and enlargement by Nabarro, D., 1907. London: Balliere, Tindall & Cox.
- LEVINE, N.D., 1961. Protozoan Parasites of Domestic Animals and Man. Minneapolis: Burgess.
- LINGARD, A., 1904. The trypanosome of dourine and its life history. Centralblatt fu¨r Bacteriologie, Parasitenkunde und Infektionskrankheiten, 37, 537–547.
- LUCKINS, A.G., 1998. Application of enzyme linked immunosorbent assays to the diagnosis of the pathogenic animal trypanosomoses. In: ‘Toward livestock disease diagnosis and control in the 21st century’, Proceedings of an international symposium on diagnosis and control of livestock diseases using nuclear and related techniques. International Atomic Energy Agency, Vienna, Austria, 1998. ISBN 92-0-102498-3.
- LUN, Z.R., ALLINGHAM, R., BRUN, R. & LANHAM, S.M., 1992. The isoenzyme characteristics of Trypanosoma equiperdum isolated from domestic stocks in China. Annals of Tropical Medicine and Parasitology, 86, 333–340.
- LUN, Z.R., BRUN, R. & GIBSON, W., 1992. Kinetoplast DNA and molecular karyotypes of Trypanosoma evansi and Trypanosoma equiperdum from China. Molecular and Biochemical Parasitology, 50, 189–196..
- MASIGA, D.K. & GIBSON, W., 1990. Specific probes for Trypanosoma (Trypanozoon) evansi based on kinetoplast DNA mini-circles. Molecular and Biochemical Parasitology, 40, 279–284.
- MAREK, J., 1904. Wertere Beitrage zur Kenntniss der Beschalseuche. Zeitschrift fu¨r Tiermedizin, 8, 11–23.
- MCCULLY, R.M. & NEITZ, W.O., 1971. Clinicopathological study on experimental Trypanosoma brucei infections in horses. Onderstepoort Journal of Veterinary Research, 38, 141–176.
- MITCHELL, C.A., WALKER, R.V.L., HEATH, L.M. & MCKERCHER, D.G., 1939. Preliminary note on the growth of Trypanosoma equiperdum in the developing chick embryo. Canadian Journal of Comparative Medicine, 3, 223–224.
- MOTT, F.W., 1906. The microscopic changes in the nervous system in a case of chronic dourine or mal de coit, and comparison of the same with those found in sleeping sickness. Proceedings of the Royal Society, Series B, 78, 1–12.
- MOTT, F.W., 1906. Changes in the nervous system produced in chronic trypanosome infections. British Medical Journal, 300, 1772–1777.
- NIE, H.Y., FANG, Y., YE, W.X., HUANG, F.L. & WANG, Y.L., 1991. Studies on the in vitro culture of Trypanosoma equiperdum. Chinese Journal of Veterinary Medicine, 17, 2–4.
- OYEJIDE, A. & MOULTON, J.E., 1983. Splenic cell immunosuppression in experimental trypanosomiasis. Tropical Veterinarian, 1, 92–97.
- PARKIN, B.S., 1948. The demonstration and transmission of the South African strain of Trypanosoma equiperdum of horses. Onderstepoort Journal of Veterinary Science and Animal Industry, 23, 41–57.
- PEASE, H.T., 1904. Surra and dourine. The Veterinary Journal, 9, 187–200.
- RIOU, G.F. & SAUCIER, J.M., 1979. Characterization of the molecular components in kinetoplast-mitochondrial DNA of Trypanosoma equiperdum. Journal of Cell Biology, 82, 248–263.
- ROBINSON, E.M., 1927. Serological investigations into some diseases of domesticated animals in South Africa caused by Trypanosomes. Eleventh and Twelfth Reports of the Director of Veterinary Education and Research. Union of South Africa, 9–25.
- ROBINSON, E.M., 1948. Notes on serological tests carried out on equine species infected with dourine. Onderstepoort Journal of Veterinary Science and Animal Industry, 23, 33–36.
- ROBINSON, E.M., 1948. Dourine infection in young equines. Onderstepoort Journal of Veterinary Science and Animal Industry, 23, 39–40.
- ROTH, C., JACQUEMOT, C., GIROUD, C., BRINGAUD, F., EISEN, H. & BALTZ, T., 1991. Antigenic variation in Trypanosoma equiperdum. Research in Microbiology, 142, 725–730.
- SCHNEIDER, G. & BUFFARD, M., 1900. La dourine et son parasite. Recuil de Médicine Vétérinaire, 7, 81–105.
- SCHOENING, H.W. & FORMAD, R.J., 1923. A study of the serology, the cerebro-spinal fluid, and the pathological changes in the spinal cord in dourine. Journal of Agricultural Research, 26, 497–505.
- SCHULZ, K., 1935. Dourine or slapsiekte. Journal of the South African Veterinary Medical Association, 6, 4–15.
- SILEGHEM, M., FLYNN, J.N., DARJI, A., DE BAETSELIER, P. & NAESSENS, J., 1994. African Trypanosomiasis. In: KIERSZENBAUM, F., (ed.). Parasitic Infections and the Immune System. San Diego: Academic Press. pp. 1–51.
- SMITH, F., 1919. The Early History of Veterinary Literature. London: Balliere, Tindall and Cox.
- SOLTYS, M.A. & WOO, P.T.K., 1977. Trypanosomes producing disease in livestock in Africa. In: KREIER, J.P., (ed.). Parasitic Protozoa. New York, San Francisco, London: Academic Press.
- STEPHEN, L.E., 1986. Trypanosomiasis: A Veterinary Perspective. Oxford, New York: Pergamon Press.
- THEIS, J.H. & BOLTON, V., 1980. Trypanosoma equiperdum: Movement from the dermis. Experimental Parasitology, 50, 317–330.
- WALKER, J., 1918. The occurrence of dourine (slapsiekte) in South Africa. Fifth and Sixth Reports of the Director of Veterinary Research Services. Union of South Africa, 189–206.
- WASSALL, D.A., GREGORY, R.J.F. & PHIPPS, L.P., 1991. Comparative evaluation of enzyme-linked immunosorbent assay (ELISA) for the diagnosis of Dourine. Veterinary Parasitology, 39, 233–239.
- WATSON, E.A., 1920. Dourine in Canada: History, Research and Suppression. Dominion of Canada, Department of Agriculture, Health of Animal Branch.
- WILLIAMS, D.J.L., TAYLOR, K.A., NEWSON, J. & GICHUKI, B., 1996. The role of anti-variable surface glycoprotein antibody responses in bovine trypanotolerance. Parasite Immunology, 18, 209–218.
- WILLIAMSON, C.C. & HERR, S., 1986. Dourine in Southern Africa 1981—1984: Serological findings from the Veterinary Research Institute, Onderstepoort. Journal of the South African Veterinary Association, 57, 163–165.
- WILLIAMSON, C.C., STOLTSZ, W.H., MATTHEUS, A. & SCHIELE, G.J., 1988. An investigation into alternate methods for the serodiagnosis of dourine. Onderstepoort Journal of Veterinary Research, 55, 117–119.
- WOO, P.T.K., 1977. Salivarian trypanosomes producing disease in livestock outside of sub-saharan Africa. In: KREIER, J.P., (ed.). Parasitic Protozoa. New York, San Francisco, London: Academic Press.
- ZHANG, Z.Q. & BALTZ, T., 1994. Identification of Trypanosoma evansi, Trypanosoma equiperdum and Trypanosoma brucei using repetitive DNA probes. Veterinary Parasitology, 53, 196–208.
- ZHANG, Z.Q., GIROUD, C. & BALTZ, T., 1991. The in vivo and in vitro sensitivity of Trypanosoma evansi and T. equiperdum to diminazene, suramin, MelCy, quinapyramine and isometamidium. Acta Tropica, 50, 101–110.