- Infectious Diseases of Livestock
- Part 1
- Theileria annulata theileriosis
- Vectors: Ticks
- Vectors: Tsetse flies
- Vectors: Muscidae
- Vectors: Tabanidae
- Vectors: Culicoides spp.
- Vectors: Mosquitoes
- Classification, epidemiology and control of arthropod-borne viruses
- Special factors affecting the control of livestock diseases in sub-Saharan Africa
- The control of infectious diseases of livestock: Making appropriate decisions in different epidemiological and socioeconomic conditions
- Infectious diseases of animals in sub-Saharan Africa: The wildlife⁄livestock interface
- Vaccination: An approach to the control of infectious diseases
- African animal trypanosomoses
- Dourine
- Trichomonosis
- Amoebic infections
- GENERAL INTRODUCTION: COCCIDIA
- Coccidiosis
- Cryptosporidiosis
- Toxoplasmosis
- Besnoitiosis
- Sarcocystosis
- Balantidiosis
- Leishmaniosis
- Neosporosis
- Equine protozoal myeloencephalitis
- GENERAL INTRODUCTION: BABESIOSES
- Bovine babesiosis
- Equine piroplasmosis
- Porcine babesiosis
- Ovine babesiosis
- GENERAL INTRODUCTION: THEILERIOSES OF CATTLE
- East Coast fever
- Corridor disease
- Zimbabwe theileriosis
- Turning sickness
- Theileria taurotragi infection
- Theileria mutans infection
- Theileria annulata theileriosis
- Theileriosis of sheep and goats
- Theileria buffeli⁄orientalis infection
- Non-pathogenic Theileria species in cattle
- GENERAL INTRODUCTION: RICKETTSIAL, CHLAMYDIAL AND HAEMOTROPIC MYCOPLASMAL DISEASES
- Heartwater
- Lesser known rickettsial infections in animals and humans
- Chlamydiosis
- Q fever
- Eperythrozoonosis
- Bovine Haemobartonellosis
- Potomac horse fever
- GENERAL INTRODUCTION: ANAPLASMOSES
- Bovine anaplasmosis
- Ovine and caprine anaplasmosis
Theileria annulata theileriosis
This content is distributed under the following licence: Attribution-NonCommercial CC BY-NC  View Creative Commons Licence details here
View Creative Commons Licence details here
Theileria annulata theileriosis
Previous Authors: E PIPANO AND V SHKAP
Current Authors:
J A LAWRENCE - Extraordinary Professor, DPhil, BSc, MRCVS (ret.), DTVM, Department of Paraclinical Veterinary Science, University of Zimbabwe, Harare, Zimbabwe
B J MANS - Principal Researcher, BSc, BSc (Hons) Biochemistry, MSc (Biochemistry), PhD (Biochemistry), Agricultural Research Council, Onderstepoort Veterinary Research, 100 Old Soutpan Road, Pretoria, Gauteng, 0110, South Africa
Introduction
Tropical theileriosis is an infectious disease of cattle caused by the protozoan Theileria annulata, which is transmitted by ticks of the genus Hyalomma. Invasion by the parasite of cells of the macrophage and lymphoid series results in their replication or destruction, and invasion of erythrocytes results in anaemia. Peracute, acute, subacute, mild and chronic forms of the diseases are recognized. Animals that recover remain latently infected.103
Theileria annulata was first described by Dschunkowsky and Luhs in Transcaucasian cattle in 1904;25 they named it Piroplasma annulatum. During the following two decades fatal theileriosis caused by T. annulata in the Mediterranean area and India was often mistakenly attributed to Theileria mutans infections.121
Tropical theileriosis occurs in northern Africa, including the sub-Saharan territories,46 Sudan and Eritrea, southern Europe, the Near and Middle East, Central Asia, India and northern China. The extent of its distribution in the Far East, where it overlaps with Theileria buffeli/orientalis infection,48 is not yet clearly delimited.24
Aetiology and life cycle
The life cycle of T. annulata is similar to that of T. parva65 (see East Coast fever: Figure 29.2) and only some characteristics specific to T. annulata are discussed here.
Three of the developmental stages of T. annulata are infective for cattle: the sporozoites, which develop in the salivary glands of ticks and, in the bovine host, the schizonts in macrophages (monocytes) and the merozoites in erythrocytes. However, in nature, the disease is transmitted only by sporozoites.
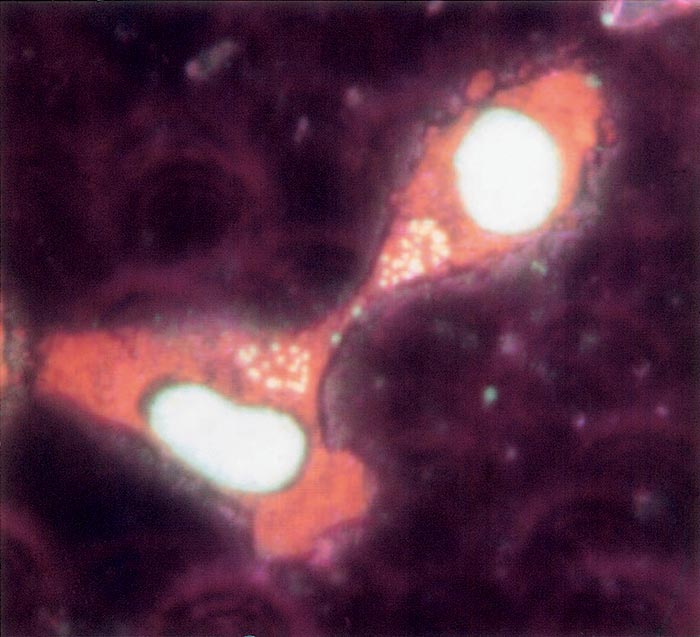
Mature sporozoites are oval-shaped, measuring about 1 μm in length. Schizonts are multinucleated, round, varying in diameter from 1 to 15 μm (in some cases up to 27 μm), with an average of about 8 μm. Each nucleus measures about 1,5μm in diameter 65, 68. In cultures, schizonts contain an average of 12 nuclei, but some with 80 or more may be found.73 Schizonts develop and grow in size as a result of binary fission of their nuclei. Schizonts are associated with the host cell microtubules, enabling the schizont to divide in synchrony with the host cell during mitosis and maintain the infection rate (Figure 37.1)73. The schizont is able to regulate host cell function by secretion of biologically active proteins direct into the cytoplasm that modify nuclear function and transform the cell. Transformation enables proliferation, immortalization and dissemination of the host cell by means of immune evasion and resistance to apoptosis, using mechanisms similar to those of neoplastic cells 15, 67, 116. Unlike neoplastic cells, there is no genomic mutation, and the cells return to normal if the parasite is killed. Host cell proliferation is reduced once merogony commences. Some of the schizonts develop merozoites, which penetrate erythrocytes.65 Schizonts containing micromerozoites (microschizonts) are not as common in T. annulata infections as they are in those of Theileria parva. This is true both in infected cattle and in cultures.
Intra-erythrocytic merozoites (also referred to as ‘piroplasms’) are generally spherical, oval- or comma-shaped, but anaplasma-like forms measuring 0,5 μm may also be encountered; each is bounded by a single-layered cell membrane and possesses an oval nucleus.65 Spherical forms vary in diameter from 0,5 to 1,5 μm, oval forms are 0,6 × 2,0 μm and comma-shaped forms 0,5 × 1,6 μm in size.68 During multiplication of merozoites, nuclear division precedes cytoplasmic division, in a process characteristic of schizogony.16
Theileria annulata is transmitted transstadially by two and three-host ticks of the genus Hyalomma. After the pre-imaginal stages ingest blood containing intra-erythrocytic merozoites, spindle-shaped bodies (considered to be microgamonts, which give rise by fission to microgametes), and spherical forms (which are macrogametes) develop in the lumen of the gut.65 The zygote-like stages which later develop in the gut wall transform into club-shaped kinetes that migrate to the salivary glands of the adult ticks after the moult. After the moulted ticks have begun to feed, the kinetes become polymorphic and undergo repeated nuclear divisions until sporozoites are formed. Generally, infective sporozoites appear in the salivary glands after the tick has been feeding for 48 to 72 hours.109 However, ‘activation’ of ticks by close proximity to cattle or by being warmed to mammalian body temperatures 64, 96 may also trigger the transformation, so that some ticks may be infective before feeding on their host. Ticks can only become infected by ingesting intra-erythrocytic merozoites.97
After inoculation into cattle, sporozoites penetrate mononuclear cells and develop into schizonts. These are detectable in smears made from superficial lymph nodes and the liver 7 to 28 days after infection.103 Merozoites are detected in erythrocytes one to three days after the first appearance of schizonts, and may persist for years in infected cattle. Schizonts also readily infect cattle by needle inoculation. Following their successive passages in cattle or culture, schizonts may lose their ability to produce merozoites, although they retain their infectivity for cattle 72, 78, 103.
Isolates differing in virulence and cross-protection ability have been reported from various geographical areas.2, 80, 83, 101 Fifty-three North African isolates of T. annulata have been characterized by antiparasitic monoclonal antibody reactivity, isoenzyme electrophoresis and Southern blotting. No identical isolates were detected by these methods and the majority of isolates contained more than one parasite population. 7 It appears, however, that the reported diversity in field T. annulata populations is not necessarily related to immunological strain differences and, therefore, it has a limited impact on immunization against tropical theileriosis.
In culture, T. annulata sporozoites invade and transform macrophages, and also B cells characterized by the CD14+ antigen.13, 44, 112 Inoculation of schizont-infected cells into cattle results in the transfer of schizonts to cells of recipient animals. This was demonstrated by the fact that when schizont-infected cells from a cow were inoculated into a male calf, the schizonts were later found within the cells of the recipient.118 The mechanism by which parasites are transferred from infected cells to host cells is not known. Inoculation with T. annulata-infected cells leads to an allogeneic graft rejection response, resulting in release of the schizonts.
Epidemiology
The occurrence of tropical theileriosis is dependent on the presence of infected ixodid ticks. Not all Hyalomma spp. that transmit T. annulata under experimental conditions are vectors in nature, since the pre-imaginal stages of some species have a preference for hosts other than cattle.11, 77 Fifteen Hyalomma spp. have been incriminated as vectors,88 although some of these may be synonyms for identical species. Among the most important are H. scupense, H. anatolicum, H. excavatum and H. dromedarii.68 The infection is generally transmitted transstadially, but infected male ticks that accidentally become detached from one host may reattach to another and, thus, transmit the disease to more than one animal.103 Transplacental transmission of infection has been demonstrated but is probably rare 36, 114.
Theileria annulata is maintained in nature by a cattle–tick–cattle cycle. The water buffalo (Bubalus bubalis) is considered to be the natural host of this parasite and to be the one in which it evolved.117 Theileria annulata usually causes no apparent infection in water buffalo, but the latter may act as a reservoir for infection of ticks. The yak (Bos grunniens) is highly susceptible to T. annulata theileriosis and suffers severe clinical signs and mortality when infected.61 Infection of sheep with T. annulata has been demonstrated by polymerase chain reaction (PCR) in Sudan and Iran 85, 115, 123, but piroplasms have been shown not to develop after experimental infection 59 , so sheep are unlikely to act as reservoirs of infection. Infection of dogs has been reported on rare occasions 9.
There is a significant difference in susceptibility of different cattle breeds to infection 34, 35. Resistant breeds exhibit a lower, shorter-lived febrile response, with fewer clinical signs and lower mortality. Bos indicus breeds are generally more resistant than Bos taurus breeds and cross-breds, though some local taurine breeds are also resistant. Differences in susceptibility have been shown to depend on differences in cytokine production by macrophages, which are dependent on differential expression of various genes in the different breeds 35, 56.
Tropical theileriosis has a seasonal occurrence, which is related to the ecology and biology of its vectors. In most endemic areas, the majority of cases occur between June and September, 11, 28, 77, 103, 104 although sporadic cases may be found throughout the year.
Theileria annulata may complete its life cycle in ticks on pasture lands or in barns. Engorged nymphs of H. scupense, one of the main vectors of T. annulata, hide in cracks and crevices of clay or stone barn walls and, after moulting, adults infect cattle introduced into these barns. 103 Susceptible cattle on zero grazing in tick-free premises are, in theory, protected from infection, but infected ticks can be introduced on hay or by stray cattle that have grazed on tick-infested pastures.77
Engorged adult Hyalomma ticks produce large numbers of sporozoites; c. 50 000 have been estimated to be present in a single cell of the type III acinus of the tick salivary glands. Since many such cells are generally infected, a bovine host may receive an enormous inoculum from a single tick feeding to repletion.84 This high infectivity has been demonstrated by infecting cattle experimentally with a 1:100 dilution of a single macerated tick.79 In contrast to the situation in some other tick-borne infections, such as babesiosis, where many ticks are needed to infect a single animal,111 in the case of T. annulata, a single tick is capable of transmitting a fatal infection.79 Examination of the salivary glands of Hyalomma ticks showed that a high percentage of ticks harvested in barns or in the field were infected with T. annulata.29, 49, 90, 120 Thus, a small number of infected ticks may cause extensive outbreaks in cattle herds.
Calves are considered by some authors to be more resistant than adult cattle, but others consider them equally susceptible. 68, 104 The situation differs in different geographical regions. In the Mediterranean area, where the two-host tick H. scupense plays the major role in the transmission of T. annulata, the disease in young calves is relatively rare. During the first tick season that calves are exposed, the level of infestation with adult ticks is low compared to that in older cattle, and they are therefore exposed to a limited natural challenge.11, 30. In contrast, in India where the main vector is H. anatolicum, calf morbidity and mortality represent most of the theileriosis cases which occur, since the infestation rate of calves with nymphs is high; and nymphs as well as adults of H. anatolicum can transmit T. annulata infection.8, 69 Antitheilerial antibodies have been detected in colostrum of infected dams,76 but there is no evidence that colostral immunity can engender significant protection against sporozoite infection in young calves. Endemic (enzootic) stability develops in herds that show a seroconversion rate of 100 per cent in calves in the first season that they are exposed to theileriosis.19 Older cattle become more heavily infested with ticks28 and if they escape infection in their first theileriosis season they are more likely than young calves to be infected by T. annulata.11, 30 Recovery from T. annulata primary infection results in the development of a persistent carrier state in cattle. This carrier state is of cardinal importance in maintaining immunity, and the accurate identification of carrier animals could be useful in assessing their immune status.45
Pathogenesis
Sporozoites introduced into susceptible cattle through the saliva of infected ticks migrate to the drainage lymph node and invade macrophages that express major histocompatibility complex (MHC) class II antigens; they may also infect a small subpopulation of B lymphocytes13. Sporozoite entry is rapid and energy dependent, and is effected by a unique zippering process between sporozoite and macrophage plasma membranes 106. The parasite escapes from its vacuole within a few minutes and becomes associated with the host microtubules. The sporozoites transform to trophozoites and transform the host cells leading to uncontrolled proliferation. The trophozoites develop into schizonts, which divide in synchrony with the host cell during mitosis and maintain the infection rate. Theileria annulata schizonts subvert the host immune response in a number of ways. Theileria-infected macrophages present antigen to CD4+ T lymphocytes to activate them and induce proliferation, through a combination of cytokines and contact between infected cells and T cells 13. The vast majority of the activated T cells are non-specific, and their presence apparently blocks the specific immune response. Proliferating infected macrophages and activated T cells invade the medulla of the lymph node, instead of remaining in the paracortex, as they would in a normal T cell response. CD8+ cytotoxic lymphocytes are activated but are blocked from attacking infected cells. Several macrophage functions are impaired in T. annulata-infected cells, including phagocytosis, nitric oxide generation and oxidative burst activity. Parasitised macrophages and proliferating lymphocytes emigrate from the infected lymph node and disperse widely through the body to more distant lymph nodes, spleen and thymus, and to non-lymphoid organs, such as the liver, kidneys, lungs, abomasum, adrenal glands, and later brain and myocardium31. Infected macrophages produce excessive quantities of pro-inflammatory cytokines, interleukin (IL)-1β, IL-6 and tumour necrosis factor-α (TNFα), in a so-called “cytokine storm” and this in turn leads to the generation of pro-inflammatory cytokine-dependent acute phase proteins (APP). Pro-inflammatory cytokines are thought to be responsible for many of the clinical signs of infection, namely fever, cachexia, leukopaenia and anaemia, and levels of APP correlate with the severity of disease 34. They are also thought to contribute to tissue damage, leukopaenia, disseminated haemorrhages and ulcerative lesions.31
In general, the pathogenesis of the lesions which occur in organs such as the liver, spleen, lungs and kidneys is poorly understood. It is possible that lymphohistiocytic vasculitis is responsible for tissue destruction and pulmonary oedema, as in East Coast fever 32, but the detailed histopathology of the disease has not yet been described.
Merozoites released from schizonts infect red blood cells, and in severe infections more than 95 per cent of red blood cells are parasitized, each containing one or more merozoites.68
Significant reductions in erythrocyte count, haemoglobin concentration and packed cell volume, with evidence of severe aplastic anaemia,57 occur during the course of the disease. Anaemia varies in degree, and icterus develops as a result of erythrocyte destruction and liver damage. It appears that not only merozoites are involved in erythrocyte destruction; cattle infected by needle inoculation with strains of T. annulata schizonts that do not produce erythrocytic merozoites may also suffer from a marked anaemia. This suggests that schizonts alone can trigger erythrocyte destruction. The pathogenesis of anaemia is incompletely understood. Factors such as haematopoietic precursor cell destruction, changes in membrane glycolipid components, activated complement products, binding of autoantibody to erythrocytes and removal of infected and noninfected erythrocytes by phagocytosis have been proposed. Most recently, oxidative damage to the erythrocyte membrane protein and lipid contents and to haemoglobin has been shown to be linked to morphological changes in the erythrocyte and osmotic fragility leading to lysis and phagocytosis 84, 91. Increases of calcium, chloride and sodium ions and of total protein in the contents of the abomasum and duodenum during the acute stage of T. annulata infection have been reported. It has been suggested that the loss of these ions and proteins from the interstitial fluid into the lumen of the digestive tract may explain the rapid debilitation of diseased cattle.70
The severity of infection (lethal or sublethal) following tick transmission depends upon the number of sporozoites inoculated. The proportions of schizont-infected cells and parasitized erythrocytes, the duration and severity of pyrexia, and the size of enlarged lymph nodes are directly related to the size of the inoculum, while the times to onset of fever and to the appearance of schizonts and erythrocytic merozoites are inversely proportional to the number of sporozoites inoculated 81, 95.
The immune response to the schizont stage of T. annulata in primary infections is both innate (NK-cells, macrophages) and adaptive (cytotoxic T cells, T-helper cells) 3. Both T- and B-lymphocytes are activated during infection. Antibodies to parasite antigens, which develop during primary infection,37 may inhibit in vitro infection of cells by sporozoites, 42 but are thought not to play a role in protection, because they have not been shown to interact with the surfaces of either schizont-infected cells or merozoite-infected erythrocytes.82 Cell- mediated immunity to T. annulata is mediated by cytotoxic T lymphocytes (CTLs) and T-helper lymphocytes. T-helper cells produce the cytokines IL-2, which is required for the clonal expansion of CTLs, and IFN-γ, which activates macrophages to produce nitric oxide. The latter destroys the schizonts within the infected cells. Cytokines are also required for the induction of parasite specific antibodies. At low levels, cytokines facilitate the immune response, but at high levels they inhibit it and are responsible for clinical disease 35. Differences in cytokine production are thought to be the basis of breed differences in disease resistance. CTLs kill their infected target cells in an MHC class I-restricted manner. Innate immunity involves suppressor or regulatory macrophages which are capable of inhibiting both the proliferation of T. annulata infected cells and the polyclonally activated T cells which are induced by infected cells 35. It is not clear what the mechanisms for such inhibition are. The role of NK-cells is not clear. They may act as effector cells in controlling the infection by non-specific lysis of parasite-containing cells or they may activate macrophages via production of IFN-γ.
The mechanisms that control intra-erythrocytic merozoites are unknown, but since nitric oxide stops invasion of peripheral blood macrophages by sporozoites,119 it may also prevent invasion of erythrocytes by merozoites.82
Immunosuppression has been demonstrated in tropical theileriosis and may lead to activation of latent infections with various agents during the acute stage of disease.105
Clinical signs
In natural infections the incubation period varies from 9 to 25 days, with an average of 15 days. However, the prepatent period may be as short as seven days in cattle inoculated with a suspension of macerated ticks. The prepatent period following inoculation of infected blood varies from 12 to 30 days, with an average of 17 days; on rare occasions it may extend to as long as 10 months.103
Initially, there is fever, swelling of the superficial lymph nodes and increases in the pulse and respiratory rates. Anorexia and decreased rumen movements are followed by dehydration and constipation. Diarrhoea often occurs in young animals, possibly because of exacerbation of latent infections. Emaciation is rapid. The eyelids may be swollen and hyperaemic, and lachrymation and nasal discharge occur. Anaemia, which becomes progressively more severe during the course of the disease, causes mucous membranes to become pale. Icterus may be present. Petechiae on the visible mucosae usually indicate an unfavourable prognosis.
The urine is dark brown as a result of the presence of bilirubin. In dairy cows there is a sudden and marked drop in milk production. Abortion often occurs. Rarely, nervous signs may appear in acute T. annulata infections one to three days before death.53 Peracute, acute, subacute, chronic and mild forms of the disease have been reported.68
The peracute form is most likely to occur in highly susceptible cattle breeds, after a massive infection. The clinical period lasts for between three and five days and ends with the death of the animal. Typical gross pathological changes generally do not have time to develop, but numerous schizonts may be present.
The acute form is the type most frequently seen in susceptible breeds. Clinical signs are well defined, and the disease lasts for one or two weeks. Recovery is slow, especially in adult cattle, and mortality is high.
The subacute form resembles the acute form, but clinical signs are less intense. Most animals recover if complications from secondary infections, such as bacterial enteritis, particularly in calves, do not occur.
The chronic form is rarely encountered. Affected cattle are recumbent, emaciated, and show intermittent fever. In most cases death occurs after two to three weeks, rarely after four weeks.
The mild form is encountered in naturally resistant cattle, in cattle protected by passive immunity, in vaccinated cattle with only partial immunity, or when the infecting strain is of low virulence. Clinical signs are generally unnoticed and cattle recover spontaneously.
In protracted cases, skin lesions consisting of lenticular haemorrhagic spots or skin nodules, which are generated in the perivascular lymphoid tissues of the dermal arterioles, are sometimes observed. Ulceration of the nodules may occur and Giemsa-stained touch preparations reveal numerous schizont-infected cells.62, 103
Rarely, infection may occur, even with an otherwise virulent strain of T. annulata, in which there are no clinical signs and often no detectable parasites, because the animal is strongly resistant to infection. This condition has been referred to as an immediate latent infection (infection latente d’emblee).100
In practice it is not always feasible to classify a field case into one of the above forms when it is first diagnosed.
Mortality rates of 20 to 90 per cent have been reported from various geographical areas.68 This range is apparently due to variations in susceptibility of cattle and in the virulence of the parasites involved. Adult animals which have apparently recovered may die two to three weeks after the disappearance of schizonts as a result of complications, such as liver dysfunction.58, 108 Growth may be impaired in young calves that recover from the disease.
Cerebral theileriosis has been recorded on occasion 18, 52, 113. (See Turning sickness)
Pathology
Haematological changes consist of severe anaemia, anisocytosis, and basophilic stippling. Leukopenia occurs in lethal and sublethal infections because of a decline in circulating lymphocytes and neutrophils. It coincides with the onset of fever, which is accompanied by initial evidence of schizonts in lymph nodes. An initial brief period of monocytopenia is followed by marked monocytosis in both fatal cases and recovering animals. The monocytosis peaks between three and four weeks after infection.81, 98.
Significant increases in the levels of serum glutamic oxaloacetic transaminase, sorbitol dehydrogenase, isocitric dehydrogenase,108 alanine aminotransferase, alkaline phosphate, creatinine kinase and γ-glutamyltransferase, as well as increases in the concentrations of uric acid, blood urea nitrogen and bilirubin 98 have been found in cattle suffering from tropical theileriosis.
In animals which have died, the severity of lesions is primarily dependent upon the course of the disease. In fulminant cases, lasting only a few days, lesions are limited mainly to hyperplasia of the lymphoid tissues and to disseminated haemorrhages. According to Sergent and collaborators, 103 the lesions in T. annulata infection are similar to those observed in haemorrhagic septicaemias, being characterized by haemorrhages and congestion.
In typical cases, the carcass is emaciated, anaemic and icteric, with yellow gelatinous connective tissues. Numerous petechiae or larger haemorrhages are seen in the subcutaneous tissues and in the mucous and serous membranes. All superficial and internal lymph nodes are swollen and often haemorrhagic. The spleen is enlarged, mainly as a result of hyperplasia of lymphoid tissue. The liver is swollen, brownish or orange-yellow in colour, and friable because of parenchymatous degeneration. Subcapsular haemorrhages are often observed. The gall bladder is distended and contains dark-green, thick bile.
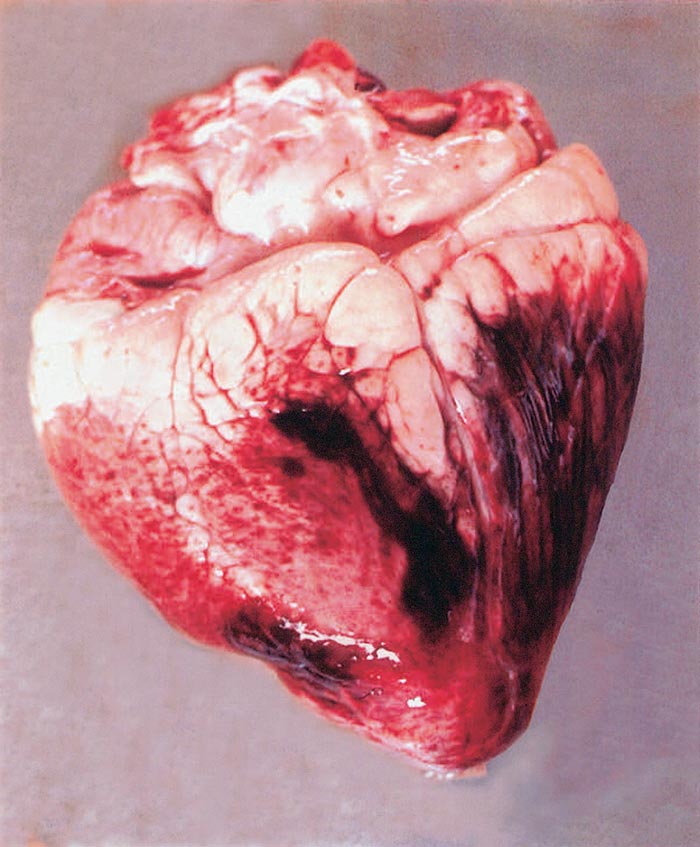
Epi- and endocardial petechiae and ecchymoses are found. The myocardium is often degenerated and shows extensive haemorrhagic areas (Figure 37.2). Excessive amounts of serous fluids may occur in the pericardial, thoracic and other body cavities. The lungs are frequently oedematous. The kidneys are congested and may show haemorrhages or small (1 to 3 mm), whitish-red foci of lymphoid hyperplasia. Depending upon the duration of the infection before death, red patches or ulcers may be found in the mucous membranes of the abomasum and small and large intestines. The urinary bladder contains urine pigmented dark brown because of the presence of bile pigments. Lymphoid hyperplasia has been described in spleen, lymph nodes, liver and kidneys 68, 102.
Exceptionally, papular and pustular skin lesions (mainly in the non-pigmented areas), which may be ulcerated or necrotic, may be observed. These lesions are infiltrated by neutrophils and lymphocytes, as well as cells containing schizonts. 12, 62 described the presence of numerous white nodules (sometimes haemorrhagic or with a haemorrhagic halo) 0,2-3 cm in diameter with a compact and uniform cut surface in the skin and subcutaneous tissue of young calves infected with T. annulata. Similar nodules were observed in skeletal muscles (particularly in the tongue, intermandibular muscles and cervical muscles), abdominal fat, omentum and intestinal serosa. Some calves exhibited these nodules in the pharynx, trachea, myocardium, pituitary gland, thymus and retrobulbar tissues. The nodules were found to be formed by T lymphocytes and macrophages; some of the latter contained schizonts. In the lymph nodes and spleen, moderate infiltration by these cells was accompanied by hypoplasia of the lymphoid tissue. Some lymph nodes also showed heavy diffuse lymphocytic infiltration with no clear cortico-medullary demarcation. In the skin, the infiltration rarely reached the superficial dermis and did not infiltrate the epidermis, being mostly limited to the middle and deep dermis.
In cattle that died after having shown cerebral disturbances, clogging of meningeal and cerebral blood vessels by large numbers of lymphocytes has been observed.53
Diagnosis and differential diagnosis
In areas endemic for tropical theileriosis, the non-specific clinical signs of fever, anaemia, icterus and swelling of peripheral lymph nodes justify a presumptive diagnosis of acute T. annulata theileriosis.
For laboratory confirmation, thin smears should be prepared from biopsy material of lymph nodes or the liver. Blood films prepared during the initial stage of the disease show rare or no erythrocytic merozoites, a picture also observed in recovered carrier animals. On the other hand, the presence of schizonts in lymph nodes and the liver is often sufficient to support a diagnosis of acute theileriosis. Smears made from liver tissue are considered by Sergent et al.102 to be the most suitable means for the detection of schizonts and for the evaluation of the severity of the infection.
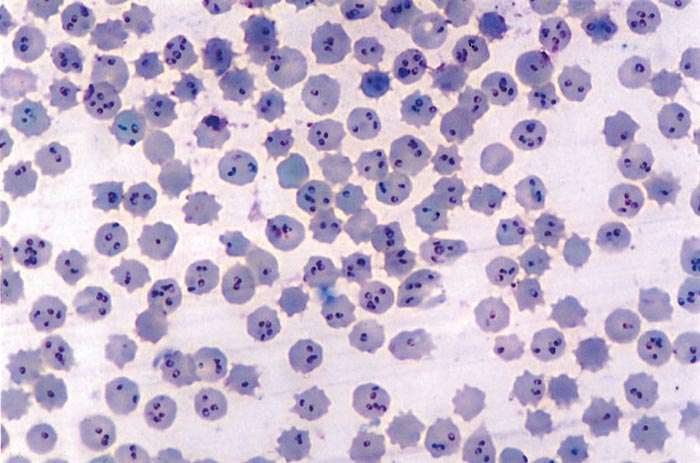
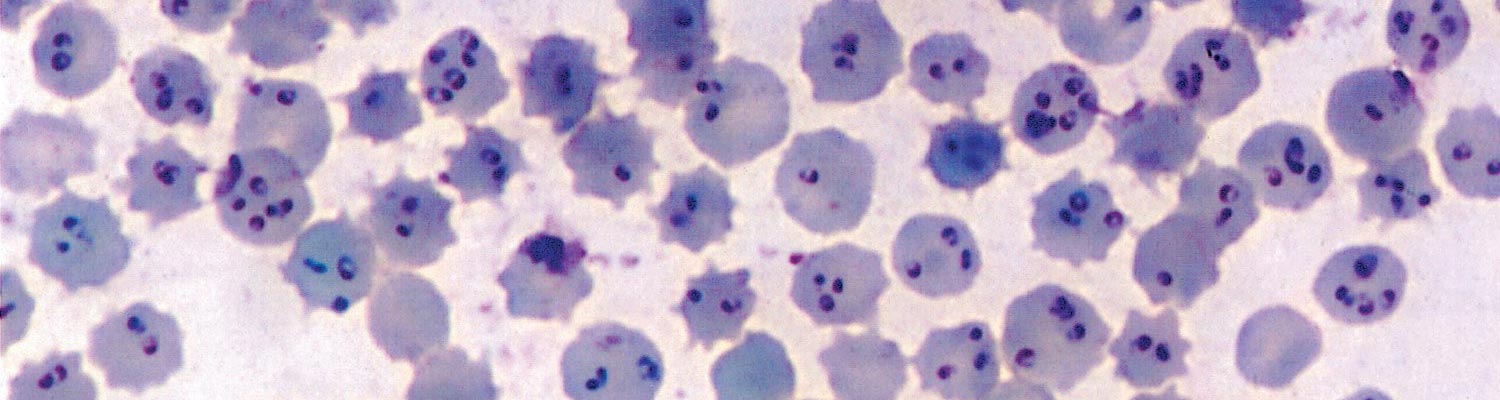
In T. annulata infection, schizonts are rare in the peripheral blood, and their presence in blood smears indicates a poor prognosis. The number of parasitized erythrocytes increases progressively during the course of the disease, and as many as 95 per cent of the red blood cells may be infected (Figure 37.3), but the severity of the disease does not necessarily correspond to the percentage of parasitized erythrocytes. The Giemsa staining method is considered to be the most suitable one for demonstrating parasites in smears.
To detect schizonts and merozoites in fatal cases, impression smears of the liver, spleen and several lymph nodes should be examined. Both intracellular and free-lying schizonts may be detected, the latter having been released from parasitized cells during preparation of the smears. Most of the merozoites in the acute stage of the disease are spherical or oval-shaped and are smaller (1 to 2 μm) than the merozoites observed in the chronic carrier form. Merozoites persist for months or even years in recovered cattle, but they are not always detectable by direct microscopic examination of smears. On the other hand, the presence of schizonts is always associated with the acute stage of the disease.
The most widely used serological test for detecting latent T. annulata infection is the indirect fluorescent antibody test (IFAT). Both schizonts and erythrocytic merozoites are used as antigens.20, 51, 71 The schizont antigen appears to be significantly better than the merozoite antigen for detecting animals carrying T. annulata infection.20 The IFAT is specific only in areas where T. annulata occurs alone, since antibodies to other Theileria spp. may cross-react with the T. annulata antigen. The IFAT is useful for identifying infected herds containing carrier animals but is not sufficiently accurate to detect all infected individuals. Neither the schizont nor merozoite IFAT detects antibodies in every animal with erythrocytic merozoites in its peripheral blood.20, 74
The enzyme-linked immunosorbent assay (ELISA) has been successfully adapted for detection of anti-T. annulata antibodies.38 In contrast to the IFAT, schizont antigen used in ELISA is less sensitive and specific than the antigen prepared from erythrocytic merozoites, since the latter detects antibodies for a longer period.58, 59
More recently, ELISA’s were developed against defined antigens such as the major immunodominant antigens found in the sporozoite and schizont stages (T. annulata macroschizont surface protein, TaSP) or the merozoite stage (30 kDa T. annulata major merozoite surface antigen, Tams1). An indirect ELISA against Tams1 was developed that showed similar sensitivity to the IFAT 40, but showed limited cross-reactivity with T. parva positive antisera, which would exclude its use within areas were the geographic distribution of these parasites overlap, such as may be the case in southern Sudan 27. Significant diversity also exists within the Tams1 gene which may limit its use to detect all T. annulata populations 41. An indirect ELISA has also been developed for TaSP 5, 92 which has a sensitivity of 99% and specificity of 90%, both significantly better than the IFAT 93. TaSP antigen gave superior performance to three other T. annulata derived antigens (TaD, TaSE and TamtHSP70) 99. A competitive ELISA using a mAb against TaSP gave a sensitivity and specificity of 77% and 100% respectively and allowed differentiation of infected and uninfected animals 87. Field validation showed a sensitivity and specificity of 84% and 52% respectively 86, with the implication that a lower number of false positives are detected using the competitive ELISA. To allow rapid detection in the field, a lateral flow device based on the TaSP antigen was developed 1. It compares well with other tests, having a sensitivity and specificity of 96.3% and 87.5% compared to the IFAT, 98.7% and 81.8% compared to the indirect ELISA, and 100% and 47.6% compared to competitive ELISA, respectively. While possibly only useful as a herd test to detect a possible theileriosis outbreak, the speed of testing (10 minutes) makes this test attractive for epidemiological surveys.
Serological tests are mainly used for epidemiological screening for T. annulata infection and assessment of the results of immunization with live vaccines. It appears that immunity to T. annulata theileriosis is mainly cell-mediated, but antibodies can be used as an indication for establishment and multiplication of Theileria parasites which induce a protective immunity. Serological detection allows inference of previous exposure to parasites and diagnosis of a possible carrier state, since T. annulata infection is considered lifelong. Direct detection of parasite DNA by molecular methods allows confirmation of current infection. Various methods have been summarized 63. These include reverse line blot of the 18S rRNA gene and cytochrome b gene 10, 39; conventional PCR's for various protein genes 7, 55, 107; and real-time PCR for the 18S rRNA gene 89. Detection limits vary for different PCR’s, but in general fluctuate around 0.00005% parasitemia. Real-time PCR assays could detect as low as 10 parasites per µl of blood 89. LAMP assays for the 18S rRNA, ITS and TA04795 60, 94 also exist.
A number of PCR’s have been successfully used to detect infected ticks 17, 55, although most can probably be adapted for this purpose.
Control
Successful chemotherapy of acute T. annulata theileriosis using various drugs or combinations of drugs has been claimed for many years. Two doses of long-acting oxytetracycline at 20 mg/kg given 48 hours apart, simultaneously with the infection or during the early prepatent period, prevent death of cattle.33, 47 However, no therapeutic effect has been recorded when this drug is administered during the acute stage of the infection.43
The napthoquinones (menoctone, parvaquone and buparvaquone) and a quinozolinone (halofuginone hydrobromide or lactate) are effective against both schizont and erythrocytic merozoites of Theileria. In experimental trials, one dose of 2,5 mg/kg of buparvaquone administered simultaneously with sporozoite infection, or during the acute stage of the disease, considerably mitigated the clinical signs and prevented death.6, 22 However, in young calves, haematopoiesis-stimulating drugs must be applied together with buparvaquone to avoid the deleterious effects of severe anaemia.23 Menoctone, parvaquone and halofuginone are no longer available, and buparvaquone remains the drug of choice. Repeated treatment may be required if buparvaquone is given 8 to 12 days after the infection. 110 A chemoprophylactic effect against a mild infection (one-tenth of a ground-up, infected tick) has been obtained with 5 mg/kg given seven days before infection, but such a dose did not prevent severe clinical signs when it was injected 14 days before infection.122 Treatment with 5 mg/kg buparvaquone on or before the day of infection did not interfere with the development of immunity to lethal challenge infections.21, 122 However, resistance of the parasite to buparvaquone has begun to emerge 66.
The epidemiology of T. annulata theileriosis indicates that short of a total eradication of the vector, the only effective way to decrease the losses from the infection significantly is through vaccination of susceptible cattle.
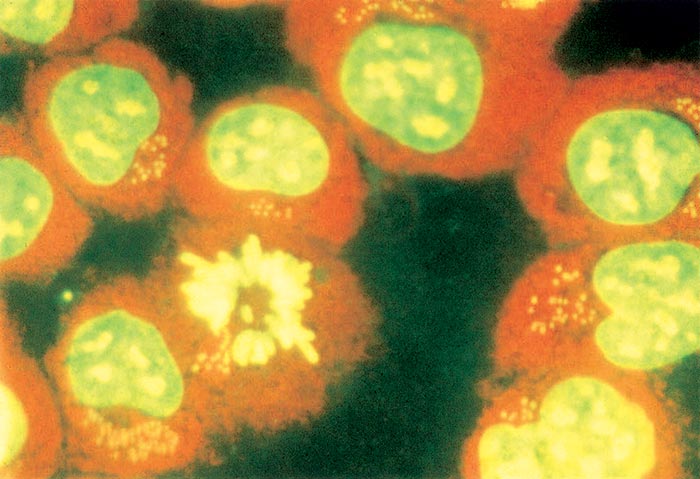
Vaccination is widely practised throughout endemic areas using attenuated schizonts 75. Attenuation was initially achieved by repeated passage of blood through calves, but this has been replaced by passage of schizonts in cell culture (Figure 37.4). Attenuation leads to progressive reduction in the capacity of the schizonts to produce merozoites and to disseminate through the body and is accompanied by progressive loss of virulence. Complete attenuation has been achieved when calves inoculated with cultured parasites do not develop clinical signs, and when schizonts or erythrocytic merozoites cannot be demonstrated in smears made from their blood and tissues. Different field isolates of T. annulata may require three months to three years of continuous cultivation in order to become completely attenuated. Vaccine prepared from fresh suspensions of cultured schizonts has a limited shelf life of only a few days at 4 °C, but storage in a concentrated form in liquid nitrogen extends the effective life of the vaccine to at least two years.
Vaccine containing completely attenuated schizonts induces a marked production of specific, but not necessarily protective, antibodies that can be detected serologically. Some multiplication of such attenuated schizonts occurs in inoculated animals, since equivalent numbers of killed schizonts inoculated without an adjuvant do not induce the formation of significant serum antibody levels. Culture-derived, attenuated schizonts engender an almost total immunity against homologous virulent schizonts and a lesser degree of immunity against virulent heterologous schizonts. The immunity is specifically directed against schizonts and does not prevent invasion by sporozoites. Variable reactions develop after challenge by infected ticks or cryopreserved stabilates of ticks. A proportion of vaccinated cattle remain asymptomatic following the challenge infection, but in most instances transient fever and mild parasitaemia occur. Protection is reduced when cattle are subjected to heavy tick challenge. Immunity lasts for at least three and a half years. Field observations have shown that some vaccinated cattle suffer severe theileriosis four or more years after vaccination, and revaccination may be required for cattle in herds with low tick-infestation rates. The response to primary immunization with anti-theilerial vaccine containing schizont-infected cells is barely influenced by the histocompatibility of the schizont-infected cell and the recipient animals. However, in addition to the immune response to the Theileria schizonts, an allogenic response to the cells carrying the parasites occurs. Upon revaccination with the same culture-derived schizont-infected cell line the allogenic antibody can block the parasite transfer to the recipient animal and prevent the enhancement of immunity. Heterologous schizont-infected cells from low-culture passages may be used to boost the immunity against tick-transmitted infection.
The schizont vaccine induces effective immunity against field-transmitted infection in indigenous susceptible cattle as well as in high-grade exotic stock introduced into theileriosis-endemic regions. There is considerable antigenic variation between and within isolates of T. annulata, but experience from vaccination campaigns has shown that the diversity in field parasite populations detected by laboratory techniques is not necessarily related to immunogenic differences and has a limited impact on immunization within a limited geographical area. Each endemic country produces its own vaccine by isolating a virulent clinical isolate and then growing the infected and transformed macrophages in the laboratory. With time, virulence is progressively lost, and loss is evaluated from time to time by injecting infected macrophages into calves and monitoring any resulting pathology. If the calves demonstrate clinical signs, in vitro culture of vaccine lines continues, and eventually attenuation is achieved after approximately two years. The process is empirical, and with time in culture the infected macrophage lines lose their immunizing potential. One reason is that the infected macrophage becomes less immunogenic with the length of time in culture, combined sometimes with selection of less virulent intracellular parasites better adapted for growth in vitro. A host cell virulence trait (c-Jun) has been identified in schizont-infected cells, and its removal by genetic manipulation induced a complete failure of T. annulata–transformed macrophages to disseminate within three weeks 26. Such procedures provide promise of an alternative to lengthy passage in culture. As with other tick-borne diseases, tropical theileriosis can be prevented by control or eradication of the vectors. The sensitivity to acaricides of some Hyalomma spp. has been evaluated,4, 54, 124 but adequate treatment regimes to control or eradicate these ticks have not yet been critically evaluated. Since a single infected tick can cause fatal theileriosis, 79 tick control must be highly efficient if it is to be effective. The time schedule for acaricide treatment should take into account the effective residual activity of the acaricide and the very short period required for maturation of T. annulata sporozoites in attached ticks.109 Under some environmental circumstances ticks may carry mature sporozoites even before they commence feeding,64 and such ticks may transmit the infection within a few hours after attachment. In tick infested barns, periodic dusting of the floor with acaricide after removal of litter and manure is recommended, in addition to acaricide treatment of the cattle.
There is considerable interest in trying to identify the genetic basis of resistance in breeds, in order to make greater use of their potential 35, 56.
References
- ABDO, J., KRISTERSSON, T., SEITZER, U., RENNEKER, S., MERZA, M. & AHMED, J., 2010. Development and laboratory evaluation of a lateral flow device (LFD) for the serodiagnosis of Theileria annulata infection. Parasitology Research, 107, 1241-1248.
- ADLER, S. & ELLENBOGEN, V., 1936. Remarks on the relationship between the Palestinian and Algerian pathogenic Theileria. Archives de l’Institut Pasteur d’Algerie, 14, 66–68.
- AHMED, J.S., GLASS, E.J., SALIH, D.A. & U SEITZER, U., 2008. Innate immunity to tropical theileriosis. Innate Immunity, 14, 5-12.
- BAGHERWAL, R.K., SISODIA, R.S., SHARMA, A., DHANOTIYA, R.S. & GHOSAL, S.B., 1995. In vitro studies on the susceptibility of the tick Hyalomma anatolicum anatolicum to acaricides using F.A.O. test kit. Indian Veterinary Journal, 72, 332–335.
- BAKHEIT, M.A., SCHNITTGER, L., SALIH, D.A., BOGUSLAWSKI, K., BEYER, D., FADL, M. & AHMED, J.S., 2004. Application of the recombinant Theileria annulata surface protein in an indirect ELISA for the diagnosis of tropical theileriosis. Parasitology Research, 92, 299-302.
- BANSAL, G.C. & SHARMA, N.N., 1989. Prophylactic efficacy of buparvaquone in experimentally induced Theileria annulata infection in calves. Veterinary Parasitology, 33, 219–224.
- BEN MILED, L., DELLAGI, K., BERNARDI, G., MELROSE, T.R., DARGOUTH, M., BOUATTOUR, A., KINNAIRD, J., SHIELS, B., TAIT, A. & BROWN, C.G.D., 1994. Genomic and phenotypic diversity of Tunisian Theileria annulata isolates. Parasitology, 108, 51–60.
- BENIWAL, R.K., NICHANI, A.K., SHARMA, R.D., RAKHA, N.K., SURI, D. & SARUP, S., 1997. Responses in animals vaccinated with the Theileria annulata (Vissar) cell culture vaccine. Tropical Animal Health and Production, 29, 109S–113S.
- BIGDELI, M., RAFIE, S.M., NAMAVARI, M.M., & JAMSHIDI S., 2012. Report of Theileria annulata and Babesia canis infections in dogs. Comparative Clinical Pathology, 21, 375–377.
- BILGIC, H.B., KARAGENÇ, T., SHIELS, B., TAIT, A., EREN, H., WEIR, W., 2010. Evaluation of cytochrome b as a sensitive target for PCR based detection of T. annulata carrier animals. Veterinary Parasitology, 174, 341-347.
- BOUATTOUR, A., DARGHOUTH, M.A. & BEN MILED, L., 1996. Cattle infestation by Hyalomma ticks and prevalence of Theileria in H. detritum species in Tunisia. Veterinary Parasitology, 65, 233–245.
- BRANCO, S., ORVALHO, J., LEITÃO, A., PEREIRA, I., MALTA, M., MARIANO, I., CARVALHO, T., RUI BAPTISTA, R., BRIAN R. SHIELS, B.R. & MARIA C. PELETEIRO, M.C., 2010. Fatal cases of Theileria annulata infection in calves in Portugal associated with neoplastic-like lymphoid cell proliferation. Journal of Veterinary Science, 11, 27-34.
- CAMPBELL, J. & SPOONER, R. 1999. Macrophage behaving badly: infected cells and subversion of immune reponses to Theileria annulata. Parasitology Today, 15, 10–16.
- CAMPBELL, J., NICHANI, A., BROWN, D., HOWIE, S., SPOONER, R. & GLASS, E., 1997. Parasite-mediated steps in immune responses failure during primary Theileria annulata-infection. Tropical Animal Health and Production, 29, 133S–135S.
- CHEESEMAN, K. & WEITZMAN, J.B., 2015. Host–parasite interactions: an intimate epigenetic relationship. Cellular Microbiology, 17, 1121-1132.
- CONRAD, P.A., KELLY, B.C. & BROWN, C.G.D., 1985. Intraerythrocytic schizogony of Theileria annulata. Parasitology, 91, 67–82.
- D’OLIVEIRA, C., VAN DERWEIDE, M., HABELA, M.A., JACQUIET, P., JONGEJAN, F., 1995. Detection of Theileria annulata in blood samples of carrier cattle by PCR. Journal of Clinical Microbiology, 33, 2665–2669.
- DABAK, M., DABAK, D.O. & AKTAS, M., 2004. Cerebral theileriosis in a Holstein calf. The Veterinary Record, 154, 533-534.
- DARGHOUTH, M.E.A., BOUATTOUR, A., BEN-MILED, L., KILANI, M. & BROWN, C.G.D., 1996. Epidemiology of tropical theileriosis (Theileria annulata infection of cattle) in an endemic region of Tunisia: characterization on endemicity states. Veterinary Parasitology, 65, 199–211.
- DARGHOUTH, M.E.A., BOYATTOUR, A., BEN-MILED, L. & SASSI, L., 1996. Diagnosis of Theileria annulata infection of cattle in Tunisia: comparison of serology and blood smears. Veterinary Research, 27, 613–621.
- DHAR, S., MALHOTRA, D.V., BHUSHAN, C. & GAUTAM, O.P. 1990. Chemoimmunoprophylaxis against bovine tropical theileriosis in young calves: a comparison between buparvaquone and long acting oxytetracycline. Research in Veterinary Science, 49, 110–112.
- DHAR, S., MALHOTRA, D.V., BHUSHAN, C. & GAUTAM, O.P., 1987. Chemoimmunoprophylaxis with buparvaquone against theileriosis in calves. Veterinary Record, 120, 375.
- DHAR, S., MALHOTRA, D.V., BHUSHAN, C. & GAUTAM, O.P., 1988. Treatment of experimentally induced Theileria annulata infection in cross-bred calves with buparvaquone. Veterinary Parasitology, 27, 267–275.
- DOLAN, T.T., 1989. Theileriasis: a comprehensive review. Revue Scientifilque et Technique, Office International des Epizooties, 8, 11–36.
- DSCHUNKOWSKY, E. & LUHS, J., 1904. Die piroplasmosen der Rinder. Zentralblatt für Bakteriologie Parasitenkunde Infektionkrankheiten, 35, 486–492.
- ECHEBLI, N., MHADHBI, M., CHAUSSEPIED, M., VAYSSETTES, C., DI SANTO, J.P., DARGHOUTH, M.A. & LANGSLEY, G., 2014. Engineering attenuated virulence of a Theileria annulata–infected macrophage. PLOS Neglected Tropical Diseases https://doi.org/10.1371/journal.pntd.0003183.
- EL HUSSEIN, A.M., HASSAN, S.M. & SALIH, D.A., 2012. Current situation of tropical theileriosis in the Sudan. Parasitology Research, 111, 503-508.
- FLACH, E.J. & OUHELLI, H., 1992. The epidemiology of tropical theileriosis (Theileria annulata infection in cattle) in an endemic area of Morocco. Veterinary Parasitology, 44, 51–65.
- FLACH, E.J., OUHELLI, H., WADDINGTON, D. & EL HASNAOUI, M., 1993. Prevalence of Theileria in the tick Hyalomma detritum detritum in the Doukkala region, Morocco. Medical and Veterinary Entomology, 7, 343–350.
- FLACH, E.J., OUHELLI, H., WADDINGTON, D., OUDICH, M. & SPOONER, R.L., 1995. Factors influencing the transmission and incidence of tropical theileriosis (Theileria annulata infection of cattle) in Morocco. Veterinary Parasitology, 59, 177–188.
- FORSYTH, L. M.G., MINNS, F.C., KIRVAR, E., ADAMSON, R.E., HALL, F.R., MCORIST, S., BROWN, C.G.D. & PRESTON, P.M., 1999. Tissue damage in cattle infected with Theileria annulata accompanied by metastasis of cytokine-producing schizont-infected mononuclear phagocytes. Journal of Comparative Pathology, 120, 39–57.
- FRY, L.M., SCHNEIDER, D.A., FREVERT, C.W., NELSON, D.D., MORRISON, W.I. & KNOWLES, D.P., 2016. East Coast fever caused by Theileria parva is characterized by macrophage activation associated with vasculitis and respiratory failure. https://doi.org/10.1371/journal.pone.0156004.
- GILL, B.S., BHATTACHARYULU, Y KAUR, D. & SINGH, A., 1978. Chemoprophylaxis with tetracycline drugs in the immunization of cattle against Theileria annulata infection. International Journal of Parasitology, 8, 467–469.
- GLASS, E.J. & JENSEN, K., 2007. Resistance and susceptibility to a protozoan parasite of cattle—gene expression differences in macrophages from different breeds of cattle. Veterinary immunology and immunopathology, 120, 20-30.
- GLASS, E.J., CRUTCHLEY, S. & JENSEN, K., 2012. Living with the enemy or uninvited guests: Functional genomics approaches to investigating host resistance or tolerance traits to a protozoan parasite, Theileria annulata, in cattle. Veterinary Immunology and Immunopathology, 148, 178– 189.
- GODARA, R., SHARMA, R.L. & SHARMA, C.S., 2010. Bovine tropical theileriosis in a neonate calf. Tropical Animal Health and Production, 42, 551-553.
- GRAY, M.A. & BROWN, C.G.D., 1981. In vitro neutralization of theilerial sporozoite infectivity with immune serum. In: IRVIN, A.D., CUNNINGHAM, M.P. & YOUNG, A.S., (eds). Advances in the Control of Theileriosis. The Hague: Martinus Nijhoff Publishers.
- GRAY, M.A., LUCKINS, A.G., RAE, P.F. & BROWN, C.G.D., 1980. Evaluation of an enzyme immunoassay for serodiagnosis of infections with Theileria parva and T. annulata. Research in Veterinary Science, 29, 360–366.
- GUBBELS, J.M., DE VOS, A.P., VAN DER WEIDE, M., VISERAS, J., SCHOULS, L.M., DE VRIES, E., & JONGEJAN, F., 1999. Simultaneous detection of bovine Theileria and Babesia species by reverse line blot hybridization. Journal of Clinical Microbiology, 37, 1782–1789.
- GUBBELS, M.J., D'OLIVEIRA, C. & JONGEJAN, F., 2000a. Development of an indirect Tams1 enzyme-linked immunosorbent assay for diagnosis of Theileria annulata infection in cattle. Clinical and Diagnostic Laboratory Immunology, 7, 404-411.
- GUBBELS, M.J., KATZER, F., HIDE, G., JONGEJAN, F. & SHIELS, B.R., 2000b. Generation of a mosaic pattern of diversity in the major merozoite-piroplasm surface antigen of Theileria annulata. Molecular and Biochemical Parasitology, 110, 23-32.
- HALL, F.R., 1988. Antigens and immunity in Theileria annulata. Parasitology Today, 4, 257–261.
- HASHEMI-FESHARKI, R. & SHAD-DEL, F., 1974. The therapeutic value of oxytetracycline hydrochloride (terramycin) in cattle infected experimentally with Theileria annulata. Tropical Animal Health and Production, 6, 119–121.
- HOWARD, C., SOPP, P., PRESTON, P., JACKSON, L. & BROWN, D., 1993. Phenotypic analysis of bovine leukocyte lines infected with Theileria annulata. Veterinary Immunology and Immunopathology, 39, 275–282.
- ILHAN, T., WILLIAMSON, S., KIRVAR, E., SHIELS, B. & BROWN, C.G.D., 1998. Theileria annulata: carrier state and immunity. Annals of New York Academy of Sciences, 849, 109–125.
- JACQUIET, P., COLAS, F., CHEIKH, D., THIAM, E. & LY, B.A., 1994. Epidemiologie descriptive de la theileriose bovine a Theileria annulata en Mauritanie, Afrique de l’Quest sub-saharienne. Revue d’Elevage et de Medicine veterinaire des Pays tropicaux, 47, 147–155.
- JAGDISH, S., SINGH, D.K., GAUTAM, O.P. & DHAR, S., 1979. Chemoprophylactic immunisation against bovine tropical theileriosis. Veterinary Record, 104, 140–142.
- JIANXUN, L. & WENSHUN, L., 1997. Cattle theileriosis in China. Tropical Animal Health and Production, 29, 45–75.
- JONGEJAN, F., MORZARIA, S., MUSTAFA, O.E.H. & LATIF, A., 1983. Infection rates of Theileria annulata in the salivary glands of the tick Hyalomma marginatum rufipes. Veterinary Parasitology, 13, 121–126.
- KACHANI, M., FLACH, E.J., WILLIAMSON, S., OUHELLI, H., EL HASNAOUI, M. & SPOONER, R.L., 1996. The use of an enzyme-linked immunosorbent assay for tropical theileriosis research in Morocco. Preventive Veterinary Medicine, 26, 329–339.
- KACHANI, M., SPOONER, R., RAE, P., BELL-SAKYI, L. & BROWN, D., 1992. Stage-specific responses following infection with Theileria annulata as evaluated using elisa. Parasitology Research, 78, 43–47.
- KALEIBAR, M.T., HELAN, J.A. & FATHI, E., 2014. Occurrence of congenital cerebral theileriosis in a newborn twin Holstein calves in Iran: Case report. Veterinary Research Forum, 5, 237–241.
- KHANNA, B.M KHAROLE, M.U., DHAR, S. & GAUTAM, O.P., 1980. Pathogenesis of lesions in bovine cerebral theileriosis. In: GAUTAM, O.P., SHARMA, R.D. & DHAR, S., (eds). Haemoprotozoan Diseases of Domestic Animals. Department of Veterinary Medicine, Haryana Agricultural University, Hissar 125004, India.
- KHURNA, K.L., CHABRA, M.B. & SAMANTRAY, S., 1992. Comparative efficacy of some acaricides against Hyalomma spp. ticks in vitro. Journal of Veterinary Parasitology (India), 6, 7–10.
- Kirvar, E., Ilhan, T., Katzer, F., Hooshmand-Rad, P., Zweygarth, E., Gerstenberg, C., Phipps, P., Brown, C.G., 2000. Detection of Theileria annulata in cattle and vector ticks by PCR using the Tams1 gene sequences. Parasitology, 120, 245-254.
- KUMAR, A., GAUR, G.K., GANDHAM, R.K., PANIGRAHI, M., GHOSH, S., SARAVANAN, B.C., BHUSHAN, B., TIWARI, A.K., SULABH, S., PRIYA, B., MUHASIN ASAF V.N, GUPTA, J.P., WANI, S.A., SAHU, A.R.& SAHOO, A.P., 2017. Global gene expression profile of peripheral blood mononuclear cells challenged with Theileria annulata in crossbred and indigenous cattle. Infection, Genetics and Evolution, 47, 9–18.
- LAIBLIN, C., 1978. Clinical investigations in cattle experimentally infected with Theileria annulata. 1. Haematological investigations. Berliner und Münchener Tierärztliche Wochenschrift, 91, 48–50.
- LAIBLIN, C., BAYSU, N. & MULLER, M., 1978. Clinical investigations in cattle experimentally infected with Theileria annulata. 1. Clinico-chemical investigations. Berliner und Münchener Tierärztliche Wochenschrift, 91, 25–27.
- LEEMANS, I., BROWN, D., HOOSHMAND-RAD, P., KIRVAR, E & UGGLA, A., 1999. Infectivity and cross-immunity studies of Theileria lestoquardi and Theileria annulata in sheep and cattle: I. In vivo responses. Veterinary Parasitology, 82, 179-192.
- LIU, A., GUAN, G., DU, P., LIU, Z., GOU, H., LIU, J., YANG, J., LI, Y., MA, M., NIU, Q., REN, Q., BAI, Q., YIN, H. & LUO, J., 2012. Loop-mediated isothermal amplification (LAMP) assays for the detection of Theileria annulata infection in China targeting the 18S rRNA and ITS sequences. Experimental Parasitology, 131, 125-129.
- LU, W.S. & YIN, H., 1994. Importance and epidemiology of theileriosis in China. Proceedings of the Third European Union Coordination Meeting on Tropical Theileriosis. 4–9 October, Antalya, Turkey.
- MANICKAM, R., DHAR, S., SINGH, R. P. & KHAROLE, M.U., 1984. Histopathology of cutaneous lesions in Theileria annulata infection of calves. Indian Veterinary Journal, 61, 13–15.
- MANS, B.J., PIENAAR, R., LATIF, A.A., 2015. A review of Theileria diagnostics and epidemiology. International Journal for Parasitology: Parasites and Wildlife, 4, 104-118.
- MAZLUM, Z., 1969. Transmission of Theileria annulata by the crushed infected unfed Hyalomma dromedarii. Parasitology, 59, 597–600.
- MEHLHORN, H. & SCHEIN, E., 1984. The piroplasms: life cycle and sexual stages. In: BAKER, J.R. & MULLER, R., (eds). Advances in Parasitology, Vol. 23. London: Academic Press.
- MHADHBI, M., NAOUACH, A, BOUMIZA, A., MOHAMED, A. CHAABANI, F., ABDERAZZAK, S.B., & DARGHOUTH, M.A., 2010. In vivo evidence for the resistance of Theileria annulata to buparvaquone. Veterinary Parasitology, 169, 241-247.
- MORRISON, W. I., 2009. Progress towards understanding the immunobiology of Theileria parasites. Parasitology, 136, 1415–1426.
- NEITZ, W.O., 1957. Theilerioses, gonderioses and ccytauxzoonoses: A review. Onderstepoort Journal of Veterinary Research, 27, 275-430.
- NICHANI, A., MANUJA, A., KHATRI, N., SING, S., BENIWAL, S., GUPTA, S., KUMAR, R. & SHARMA, R., 1998. Epidemiology and control of tropical theileriosis in North India. Proceedings of the 5th International Veterinary Immunology Symposium. Punjab Agricultural University, Ludhiana, India.
- OUHELLI, H., DAKKAK, A. & OUAZZANI, B., 1987. Theileria annulata Difference du potentiel transmural de l’abomasum et du duodenum et permeabilite de leur muqueuse chez le bovin infeste experimentalement. Experimental Parasitology, 63, 253–259.
- PIPANO, E. & CAHANA, M., 1969. Fluorescent antibody test for the serodiagnosis of Theileria annulata. Journal of Parasitology, 55, 765.
- PIPANO, E. & ISRAEL, V., 1971. Absence of erythrocyte forms of Theileria annulata in calves inoculated with schizonts from a virulent field strain grown in tissue culture. Journal of Protozoology, 18, Suppl. 37.
- PIPANO, E. & SHKAP, V., 1990. Observations on the in vitro multiplication of bovine lymphoid cells infected with Theileria annulata schizonts. Revue d’Elevage et de Medicine Veterinaire des Pays Tropicaux, 43, 485–488.
- PIPANO, E. & SHKAP, V., 1995. Kimron Veterinary Institute, Bet Dagan, Israel. Unpublished observations.
- PIPANO, E. & SHKAP, V., 2000. Vaccination against tropical theileriosis. Annals of the New York Academy of Sciences, 916, 484-500.
- PIPANO, E., 1977. Basic principles of Theileria annulata control. In: HENSON, J.B. & CAMPBELL, M., (eds). Theileriosis. Ottawa, Canada: International Development Research Center. pp. 55–65.
- PIPANO, E., 1989. Bovine theileriosis in Israel. Revue Sientifique et Technique de 1’Office International des Epizooties, 8, 79–87.
- PIPANO, E., KLOPFER, U. & COHEN, R., 1973. Inoculation of cattle with bovine lymphoid cell lines infected with Theileria annulata. Research in Veterinary Science, 15, 388–389.
- PIPANO, E., SAMISH, M. & KRIGEL, Y., 1982. Relative infectivity of Theileria annulata (Dschunkowsky and Luhs, 1904) stabilates derived from female and male Hyalomma excavatum (Koch, 1844) ticks. Veterinary Parasitology, 10, 21–27.
- PIPANO, E., WEISMAN, Y. & BENADO, A., 1974. The virulence of four local strains of Theileria annulata. Refuah Veterinarith, 31, 59–63.
- PRESTON, P.M., BROWN, C.G.D., BELL-SAKYI, L., RICHARDSON, W. & SANDERSON, A., 1992. Tropical theileriosis in Bos taurus and Bos taurus cross Bos indicus calves: response to infection with graded doses of sporozoites of Theileria annulata. Research in Veterinary Science, 53, 230–243.
- PRESTON, P.M., HALL, F.R., GLASS, E.J., CAMPBELL, J.D.M., DARGHOUTH, M.A., AHMED, J.S., SHIELS, B.R., SPOONER, R.L., JONGEJAN, F. & BROWN, C.G.D., 1999. Innate and adaptive immune responses co-operate to protect cattle against Theileria annulata. Parasitology Today, 15, 268–274.
- RAFYI, A., MAGHAMI, G. & HOOSHMAND-RAD, P., 1965. Sur la virulence de Theileria annulata (Dschunkowsky et Luhs, 1904) et la premunition contre la theileriose bovine en Iran. Bulletin de l’Office International des Epizooties, 64, 431–446.
- RAZAVI, S.M., S. NAZIFI, S., RAKHSHANDEHROO, E., FIROOZI, P. & FARSANDAJ, M., 2012. Erythrocyte antioxidant systems, lipid peroxidation and circulating lipid profiles in cattle naturally infected with Theileria annulata Revue Médecine Véterinaire, 163, 18-24.
- RAZMI, G. & YAGHFOORI, S., 2013. Molecular surveillance of Theileria ovis, Theileria lestoquardi and Theileria annulata infection in sheep and ixodid ticks in Iran. Onderstepoort Journal of Veterinary Research, 80, 1-5.
- RENNEKER, S., ABDO, J., AHMED, J.S. & SEITZER, U., 2009. Field validation of a competitive ELISA for detection of Theileria annulata infection. Parasitology Research, 106, 47-53.
- RENNEKER, S., KULLMANN, B., GERBER, S., DOBSCHANSKI, J., BAKHEIT, M.A., GEYSEN, D., SHIELS, B., TAIT, A., AHMED, J.S. & SEITZER, U., 2008. Development of a competitive ELISA for detection of Theileria annulata infection. Transboundary and Emerging Diseases, 55, 249-256.
- ROBINSON, P.M., 1982. Theileria annulata and its transmission: review. Tropical Animal Health and Production, 14, 3–12.
- ROS-GARCÍA, A., NICOLÁS, A., GARCÍA-PÉREZ, A.L., JUSTE, R.A. & HURTADO, A., 2012. Development and evaluation of a real-time PCR assay for the quantitative detection of Theileria annulata in cattle. Parasites and Vectors, 5, 171.
- SAHOO, P.K. & MISRA, S.C., 1995. Natural infection rate of Theileria annulata in Hyalomma anatolicum anatolicum at Bhubaneswar. Indian Veterinary Journal, 72, 687–689.
- SALEH, M.A., MAHRAN, O.M. & AL-SALAHY, M.B., 2011. Corpuscular oxidation in newborn crossbred calves naturally infected with Theileria annulata. Veterinary Parasitology, 182, 193–200.
- SALIH, D.A., AHMED, J.S., BAKHEIT, M.A., ALI, E.B., EL HUSSEIN, A.M., HASSAN, S.M., SHARIFF, O.E., FADL, M. & JONGEJAN, F., 2005. Validation of the indirect TaSP enzyme-linked immunosorbent assay for diagnosis of Theileria annulata infection in cattle. Parasitology Research, 97, 302-308.
- SALIH, D.A., HASSAN, S.M. & EL HUSSEIN, A.M., 2007. Comparisons among two serological tests and microscopic examination for the detection of Theileria annulata in cattle in northern Sudan. Preventive Veterinary Medicine, 81, 323-326.
- SALIH, D.A., LIU, Z., BAKHEIT, M.A., ALI, A.M., EL HUSSEIN, A.M., UNGER, H., VILJOEN, G., SEITZER, U. & AHMED, J.S., 2008. Development and evaluation of a loop-mediated isothermal amplification method for diagnosis of tropical theileriosis. Transboundary and Emerging Diseases, 55, 238-243.
- SAMANTARAY, S.N., BHATTACHARYULU, Y. & GILL, B.S., 1980. Immunization of calves against bovine tropical theileriosis (Theileria annulata) with graded doses of sporozoites and irradiated sporozoites. International Journal for Parasitology, 10, 355–358.
- SAMISH, M., 1977. Infective Theileria annulata in the tick without a blood meal stimulus. Nature, 270, 51–52.
- SAMISH, M., KRIEGEL, Y., FRANK, M., BIN, C. & PIPANO, E., 1984. The transmission of Theileria annulata to cattle by Hyalomma excavatum infected with Theileria cultivated in vitro. Refuah Veterinarith, 41, 62–63.
- SANDHU, G.S., GREWAL, A.S., SINGH, A., KONDAL, J.K., SINGH, J. & BRAR, R.S., 1998. Haematological and biochemical studies on experimental Theileria annulata infection in crossbred calves. Veterinary Research Communications, 22, 347–354.
- SEITZER, U., BEYER, D., KULLMANN, B., BAKHEIT, M.A. & AHMED, J.S., 2008. Evaluation of Theileria annulata recombinant immunodominant proteins for the development of ELISA. Transboundary and Emerging Diseases, 55, 244-248.
- SERGENT, E., 1963. Latent infection and premunition. Some definitions of microbiology and immunology. In: GARNHAM, P.C.C., PIERCE, A.E. & ROITT, T., (eds). 1. Immunity to Protozoa. Oxford: Blackwell Scientific Publications.
- SERGENT, E., DONATIEN, A., PARROT, L. & LESTOQUARD, F., 1935. Theilerioses bovines de l’Afrique du Nord et du Proche Orient. Archives de l’lnstitut Pasteur d’Algerie, 13, 472–488.
- SERGENT, E., DONATIEN, A., PARROT, L. & LESTOQUARD, F., 1945. Techniques. In: Etudes sur les Piroplasmoses bovines, Institut Pasteur d’Algerie, Alger, 79–118.
- SERGENT, E., DONATIEN, A., PARROT, L. & LESTOQUARD, F., 1945. Theileriose a Theileria dispar. In: Etudes sur les Piroplasmoses bovines. Institut Pasteur d’Algerie, Alger, 243–560.
- SHARMA, R.D., 1980. Some epidemiological observations on tropical theileriosis in India. In: GAUTAM, O.P., SHARMA, R.D. & DHAR, S., (eds). Haemoprotozoan Diseases of Domestic Animals. Proceedings of Seminar on Haemoprotozoan Diseases. Department of Veterinary Medicine, Haryana Agricultural University, Hissar 125004, (Haryana) India.
- SHARPE, R.T. & LANGLEY, A.M., 1983. The effect of Theileria annulata infection on the immune response of cattle to foot-and-mouth disease. British Veterinary Journal, 139, 378–385.
- SHAW, M.K., 2003. Cell invasion by Theileria sporozoites. Trends in Parasitology, 19, 2-6.
- SHAYAN, P., BIERMANN, R., SCHEIN, E., GERDES, J. & AHMED, J.S., 1998. Detection and differentiation of Theileria annulata and Theileria parva using macroschizont-derived DNA probes. Annals of the New York Academy of Sciences, 849, 88-95.
- SHLOSBERG, A., BOGIN, E., PIPANO, E. & KLINGER, I., 1973. Serum enzyme levels in experimental theileriosis in calves. Refuah Veterinarith, 30, 24–27.
- SINGH, D.K., JAGDISH, S., GAUTAM, O.P. & DHAR, S., 1979. Infectivity of ground-up tick supernates prepared from Theileria annulata infected Hyalomma anatolicum anatolicum. Tropical Animal Health and Production, 11, 87–90.
- SINGH, D.K., THAKUR, M., RAGHAV, P.R.S. & VARSHNEY, B.C., 1993. Chemotherapeutic trials with four drugs in crossbred calves experimentally infected with Theileria annulata. Research in Veterinary Science, 54, 68–71.
- SMITH, R.D., 1983. Babesia bovis: Computer simulation of the relationship between the tick vector, parasite and bovine host. Experimental Parasitology, 56, 27–40.
- SPOONER, R., INNES, E., GLASS, E. & BROWN, D., 1989. Theileria annulata and Theileria parva infect and transform different bovine mononuclear cells. Immunology, 66, 284–288.
- SUDAN, V., SHARMA, R.L., YADAV, R. & BORAH, M.K., 2012. Turning sickness in a cross bred cow naturally infected with Theileria annulata. Journal of Parasitic Diseases, 36, 226-229.
- SUDAN, V., SINGH, S.K., JAISWAL, A.K., PARASHAR, R. & SHANKER, D., 2015. First molecular evidence of the transplacental transmission of Theileria annulata. Tropical Animal Health and Production, 47, 1213-1215.
- TAHA, K.M., SALIH, D.A, ALI, A.M., OMER, R.A. & EL HUSSEIN, A.M., 2013. Naturally occurring infections of cattle with Theileria lestoquardi and sheep with Theileria annulata in the Sudan. Veterinary Parasitology, 191, 143-145.
- TRETINA, K., GOTIA, H.T., MANN, D.J., & SILVA, J.C., 2015. Theileria-transformed bovine leukocytes have cancer hallmarks. Trends in Parasitology, 31, 306-314.
- UILENBERG, G., 1981. Theilerial species of domestic livestock, In: IRVIN, A.D., CUNNINGHAM, M.P. & YOUNG, A.S., (eds). Advances in the Control of Theileriosis. The Hague: Martinus Nijhoff Publishers. pp. 4–37.
- VAN DEN ENDE, M. & EDLINGER, E., 1971. Culture de lignees lymphocytaires bovines infectees par Theileria annulata. Archives de l’lnstitut Pasteur de Tunis, 45–54.
- VISSER, A., ABRAHAM, A., BELL-SAKYI, L., BROWN, D. & PRESTON, P., 1995. Nitric oxide inhibits establishment of macroschizont-infected cells lines and is produced by macrophages of calves undergoing bovine tropical theileriosis or East Coast fever. Parasite Immunology, 17, 91–102.
- WALKER, A., LATIF, A., MORZARIA, S. & JONGEJAN, F., 1983. Natural infection rates of Hyalomma anatolicum anatolicum with Theileria in Sudan Research in Veterinary Science, 35, 87–90.
- WENYON, C.M., 1926. Theileria in cattle. In: Protozoology, Vol. II. London: Bailliere, Tindall and Cox.
- WILKIE, G.M., BROWN, C.G.D., KIRVAR, E., THOMAS, M., WILLIAMSON, S.M., BELL-SAKYI, L.J. & SPARAGANO, O., 1998. Chemoprophylaxis of Theileria annulata and Theileria parva infections of calves with buparvaquone. Veterinary Parasitology, 78, 1–12.
- ZAEEMI, M., HADDADZADEH, H., KHAZRAIINIA, P., KAZEMI, B. & BANDEHPOUR, M., 2011. Identification of different Theileria species (Theileria lestoquardi, Theileria ovis, and Theileria annulata) in naturally infected sheep using nested PCR–RFLP. Parasitology Research, 108, 837-843.
- ZIV, M., 1979. The susceptibility to lindane of field strains of Hyalomma ticks (lxodoidea, Isodidae) in Israel. Refuah Veterinarith, 36, 114–116.