- Infectious Diseases of Livestock
- Part 1
- East Coast fever
- Vectors: Ticks
- Vectors: Tsetse flies
- Vectors: Muscidae
- Vectors: Tabanidae
- Vectors: Culicoides spp.
- Vectors: Mosquitoes
- Classification, epidemiology and control of arthropod-borne viruses
- Special factors affecting the control of livestock diseases in sub-Saharan Africa
- The control of infectious diseases of livestock: Making appropriate decisions in different epidemiological and socioeconomic conditions
- Infectious diseases of animals in sub-Saharan Africa: The wildlife⁄livestock interface
- Vaccination: An approach to the control of infectious diseases
- African animal trypanosomoses
- Dourine
- Trichomonosis
- Amoebic infections
- GENERAL INTRODUCTION: COCCIDIA
- Coccidiosis
- Cryptosporidiosis
- Toxoplasmosis
- Besnoitiosis
- Sarcocystosis
- Balantidiosis
- Leishmaniosis
- Neosporosis
- Equine protozoal myeloencephalitis
- GENERAL INTRODUCTION: BABESIOSES
- Bovine babesiosis
- Equine piroplasmosis
- Porcine babesiosis
- Ovine babesiosis
- GENERAL INTRODUCTION: THEILERIOSES OF CATTLE
- East Coast fever
- Corridor disease
- Zimbabwe theileriosis
- Turning sickness
- Theileria taurotragi infection
- Theileria mutans infection
- Theileria annulata theileriosis
- Theileriosis of sheep and goats
- Theileria buffeli⁄orientalis infection
- Non-pathogenic Theileria species in cattle
- GENERAL INTRODUCTION: RICKETTSIAL, CHLAMYDIAL AND HAEMOTROPIC MYCOPLASMAL DISEASES
- Heartwater
- Lesser known rickettsial infections in animals and humans
- Chlamydiosis
- Q fever
- Eperythrozoonosis
- Bovine Haemobartonellosis
- Potomac horse fever
- GENERAL INTRODUCTION: ANAPLASMOSES
- Bovine anaplasmosis
- Ovine and caprine anaplasmosis
East Coast fever
This content is distributed under the following licence: Attribution-NonCommercial CC BY-NC  View Creative Commons Licence details here
View Creative Commons Licence details here
East Coast fever
Previous Authors: J A LAWRENCE, B D PERRY AND S M WILLIAMSON
Current Authors:
J A LAWRENCE - Extraordinary Professor, DPhil, BSc, MRCVS (ret.), DTVM, Department of Paraclinical Veterinary Science, University of Zimbabwe, Harare, Zimbabwe
K P SIBEKO-MATJILA - Senior Lecturer, BSc, PhD, Department of Veterinary Tropical Diseases, Faculty of Veterinary Science, Private Bag X04, University of Pretoria, Gauteng, 0110, South Africa
B J MANS - Principal Researcher, BSc, BSc (Hons) Biochemistry, MSc (Biochemistry), PhD (Biochemistry), Agricultural Research Council, Onderstepoort Veterinary Research, 100 Old Soutpan Road, Gauteng, 0110, South Africa
Introduction
East Coast fever in its classical form is a usually fatal disease of cattle caused by Theileria parva. It is transmitted principally by the brown ear tick Rhipicephalus appendiculatus and is characterized by the proliferation of lymphoblasts infected with theilerial schizonts throughout the body, particularly in the lymph nodes, spleen and lymphoid aggregatesin the kidneys, liver and lungs. The disease occurs widely through the range of its main vector in eastern, central, and southern Africa north of the Zambezi River (see Vectors: Ticks). It was introduced into the region south of the Zambezi River in 1901/02 and spread through most of the range of its vector but was subsequently eradicated (Figure 29.1).
The classical disease is seen in cattle of European origin which have been exposed to infected ticks. Cattle of African origin have a very variable response to infection and the disease may be insignificant or subclinical in Zebu calves born from immune dams and raised in endemically infected areas.
The disease was first described as an unusually virulent form of babesiosis by Gray and Robertson57 in 1902, following its introduction into Zimbabwe. It was initially named Rhodesian redwater. Koch69 identified the parasite with one that he had seen in 1897 in cattle on the coast of Tanzania. He named the disease African Coast fever, the name being subsequently modified to East Coast fever in South Africa. The causal organism was named Piroplasma kochi by Stephens and Christophers in 1903, but the name was changed to Piroplasma parvum by Theiler159 and subsequently to Theileria parva by Bettencourt, França and Borges in 1907 (as quoted by Henning59). Uilenberg168 proposed that the organism be named Theileria parva parva to distinguish it from other subspecies of T. parva which cause diseases that can be differentiated from East Coast fever on epidemiological grounds. However, it has since been agreed that East Coast fever be known as cattle-derived theileriosis and that it is caused by the same parasite as that causing Corridor disease and January disease.3, 135
East Coast fever has long been endemic in eastern Africa. The parasite probably originated from African buffalo (Syncerus caffer) populations in eastern Africa and became adapted to cattle following their introduction and dissemination in the region, especially in the Lake Victoria Basin and along the coastal strip.128 It spread widely through the region during the early part of the nineteenth century as a result of European settlement, which involved changes in patterns of movement, extensive use of ox-drawn transport, and importation of susceptible cattle breeds from overseas. The First World War (1914 to 1918), during which the campaign in what was then German East Africa, now Tanzania, took place, also resulted in unusual cattle movements and further dissemination of the disease.
East Coast fever was introduced into the area south of the Zambezi River in the period 1901 to 1903 by cattle which were imported from Kenya and Tanzania for the purpose of restocking the region after the ravages of the rinderpest epidemic of 1896 and the South African War (1899 to 1902).71 The disease initially appeared in Zimbabwe, southern Mozambique and the northern parts of South Africa, and then spread southward along the east coast of southern Africa, through Swaziland, KwaZulu-Natal, and the Eastern Cape Province. In the period up to 1914 it is estimated to have killed one and a quarter million of the four million cattle that were present in the affected territories at the beginning of the outbreak. The southward advance was halted in the region of East London in the Eastern Cape Province, and the disease was subsequently eradicated in a prolonged campaign consisting of movement control, tick control, destocking of infected pastures, and slaughter. It was eradicated from southern Mozambique by 1917, from Zimbabwe by 1954, and from South Africa by 1955. The last case south of the Zambezi River occurred in Swaziland in 1960. The disease has persisted in the region north of the Zambezi River to the present day and is a major constraint on the development of cattle production.
East Coast fever, if uncontrolled, may cause over 90 per cent mortality of the susceptible cattle following its introduction into a region. In an area where it is endemic, mortality among locally adapted Zebu-type cattle may be negligible but there is evidence that the disease causes a significant reduction in growth and productivity. Susceptible cattle introduced into an endemic area are very vulnerable to infection and the cost of the control measures required is a continuous financial burden on both livestock owners and state veterinary services.44, 106
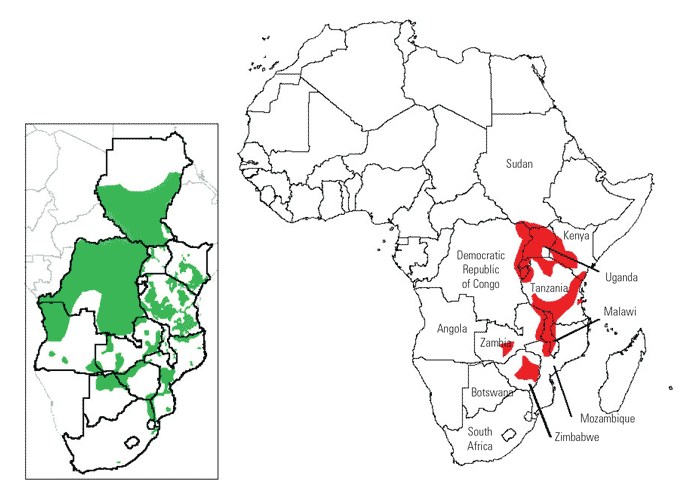
Figure 29.1 The approximate distribution of cattle-associated Theileria parva infection, namely East Coast fever and Zimbabwe theileriosis, in eastern and southern Africa, from 75. Buffalo-associated infection, commonly referred to as Corridor disease, occurs in areas in which cattle and African buffalo (Syncerus caffer) have contact. It has a similar but wider distribution that includes South Africa, Botswana and Angola INSET: The approximate distribution of the African buffalo, modified from 75.
Aetiology and life cycle
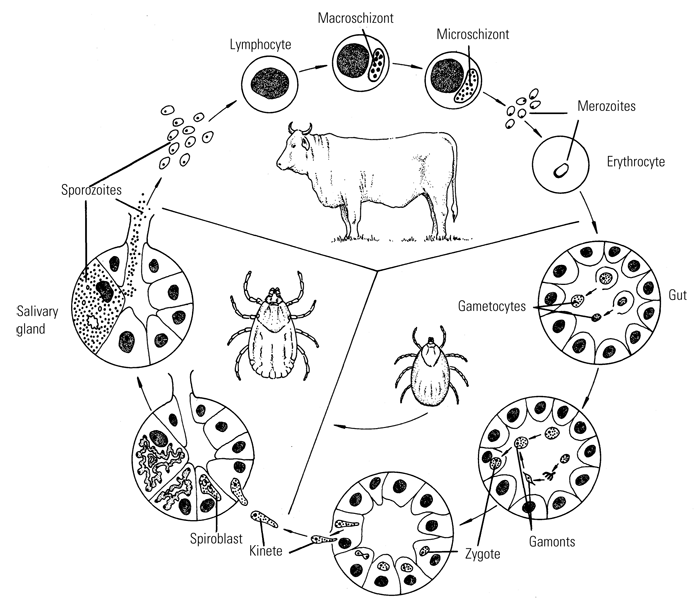
Theileria parva, the causal organism of East Coast fever, alternates between ox and tick in its life cycle (Figure 29.2). The sexual stage of development occurs in the gut of the tick.35 Piroplasms in the erythrocytes of the ox are ingested with the blood meal of the tick and develop into thread-like microgametes and globular macrogametes. These fuse to form zygotes which invade the epithelial cells of the gut of the tick. Genetic recombination between different parasite strains during the sexual stage of development leads to a high level of genetic diversity and a high frequency of cattle with mixed genotypes. This in turn leads to great variability in the immune response to field challenge after immunization 119. Completion of development occurs in the immature stages of the tick only after they have detached from the host.
As the tick moults, club-shaped motile kinetes forms within the zygote. These are liberated into the body cavity and migrates to the salivary glands via the haemolymph. The kinetes invade epithelial cells of the salivary gland, almost exclusively E cells of type III acini,52 and develops into a large syncytial sporoblast within which many thousands of minute, elongated sporozoites develop during the early part of the next feeding stage of the tick. Acini of salivary glands containing mature sporozoites can be identified easily in whole mount preparations of tick salivary glands stained with methyl green pyronin. 173 A polymerase chain reaction (PCR) and DNA probes have also been used to detect T. parva in ticks32, 176 and to discriminate T. parva from T. taurotragi.14
The sporozoites are liberated into the saliva and inoculated through the skin of the ox as the tick feeds. They enter lymphocytes, usually but not exclusively T lymphocytes of CD4+ and CD8+ subsets, and develop within them, becoming multinucleate schizonts after a period of about three days.155 Sporozoite entry is rapid and energy dependent, and is effected by a unique zippering process between sporozoite and lymphocyte plasma membranes 150. The parasite escapes from its vacuole within a few minutes and becomes associated with the host microtubules, enabling the schizont to divide in synchrony with the host cell during mitosis and maintain the infection rate.
The schizont is able to regulate host cell function by secretion of biologically active proteins directly into the cytoplasm that modify nuclear function and transform the cell into a lymphoblast. Transformation enables proliferation, immortalization and dissemination of the host cell by means of immune evasion and resistance to apoptosis, using mechanisms similar to those of neoplastic cells 29, 98, 167. Unlike neoplastic cells, there is no genomic mutation, and the cells return to normal if the parasite is killed. Host cell proliferation is reduced once merogony commences.
It is believed that schizonts have the ability to transfer from one host cell to another, but the mechanism by which they do so is not known and the frequency with which transfer occurs is probably limited.101
The kinetics of schizont replication have been described in detail.100 Schizont-infected cells proliferate exponentially, becoming distributed throughout the lymphoid system, with metastasis of infected cells to non-lymphoid tissues also occurring. Initially the schizonts have large chromatin particles and are called macroschizonts. Later, merogony results in the appearance of microschizonts with small chromatin particles. Merozoites liberated from the microschizonts invade the erythrocytes, in which they are frequently referred to as piroplasms, thus completing the life cycle. Merozoites have the ability to divide within erythrocytes to produce four daughter merozoites33 but intra-erythrocytic division is uncommon.
Studies on the fine structure of the developmental forms of the organism have been reviewed.90
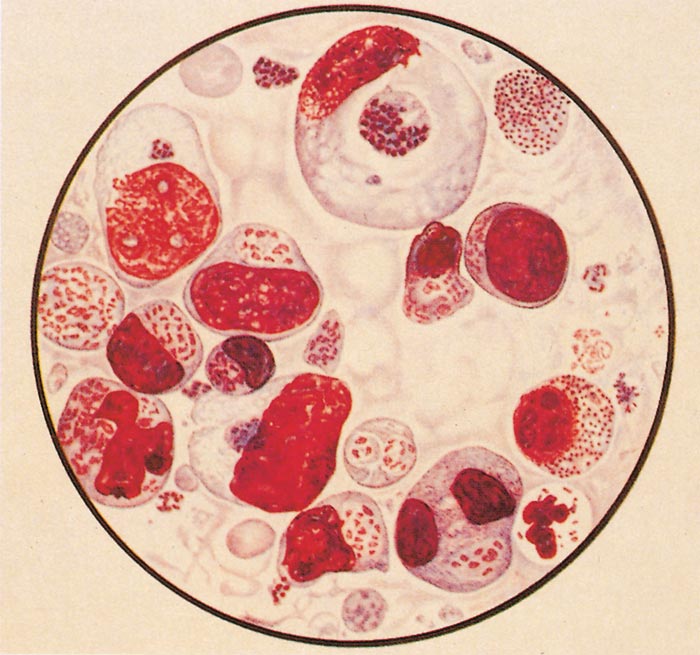
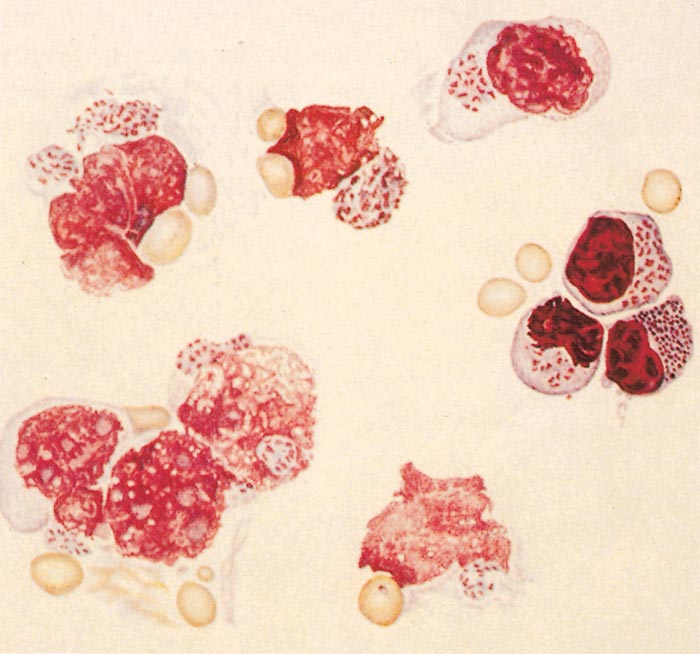
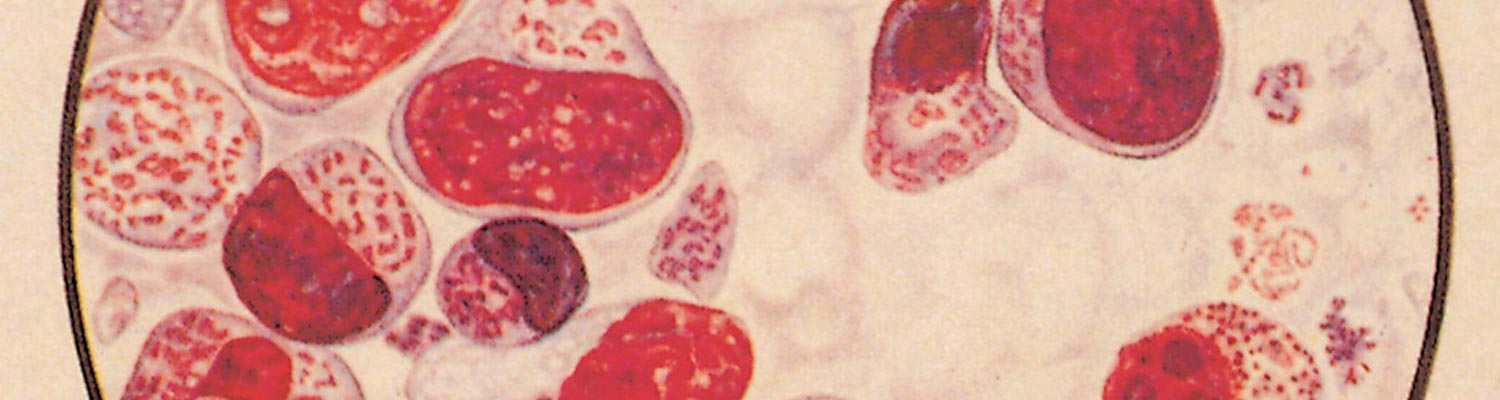
Two stages in the development of T. parva are readily identified in the infected ox, namely the schizont in lymphoblasts and the piroplasm in erythrocytes. The schizont is a mass of protoplasm contained within a thin membrane which is situated intracytoplasmically in the infected lymphoblast (Figure 29.3 to Figure 29.7). It varies in diameter from about 1 to 16 μm with a mean diameter of 5 μm and contains within it between 1 and 85 chromatin bodies, with a mean of eight.9 In macroschizonts the chromatin granules average 1 μm in diameter, while in microschizonts they average 0,5 μm.118 The size of the schizonts and the number of chromatin bodies that they contain increase as the disease progresses.11 With Romanowsky stains, e.g. Giemsa, the cytoplasm stains pale blue, typically of a lighter shade than that of the host cell, and the chromatin granules stain red (Figure 29.3 to Figure 29.7). In haematoxylin and eosin stained histological preparations the chromatin is basophilic and the cytoplasm neutral. The macroschizont was first described by Koch in 190369 and is often known as a ‘Koch’s body’ or a ‘Koch’s blue body’, the latter name emphasizing its appearance when stained by a Romanowsky technique. Schizonts are found intracellularly in lymphoblasts in lymphoid tissues throughout the body (including lymph nodes, spleen, thymus, Peyer’s patches and the lymphoid aggregations in parenchymatous organs which are a feature of theilerial infections) as well as in the blood.Figures 29.3 (top) and 29.4 (bottom) Impression smears from prescapular lymph node: drawings of lymphoblasts and lymphocytes containing macro- and microschizonts. Note several schizonts are extracellular after disintegration of lymphocytic cells. (By courtesy of the Editor of the Onderstepoort Journal of Veterinary Research. Printed in Volume 27, Number 3, 1957)
They frequently appear extracellularly in smears as a result of rupture of the host cell. The number of lymphoblasts infected with schizonts is variable and ranges from less than 1 per cent to over 70 per cent (in fatal cases).9 The infection rate increases as the disease progresses. There may be more than one schizont in a single infected cell.
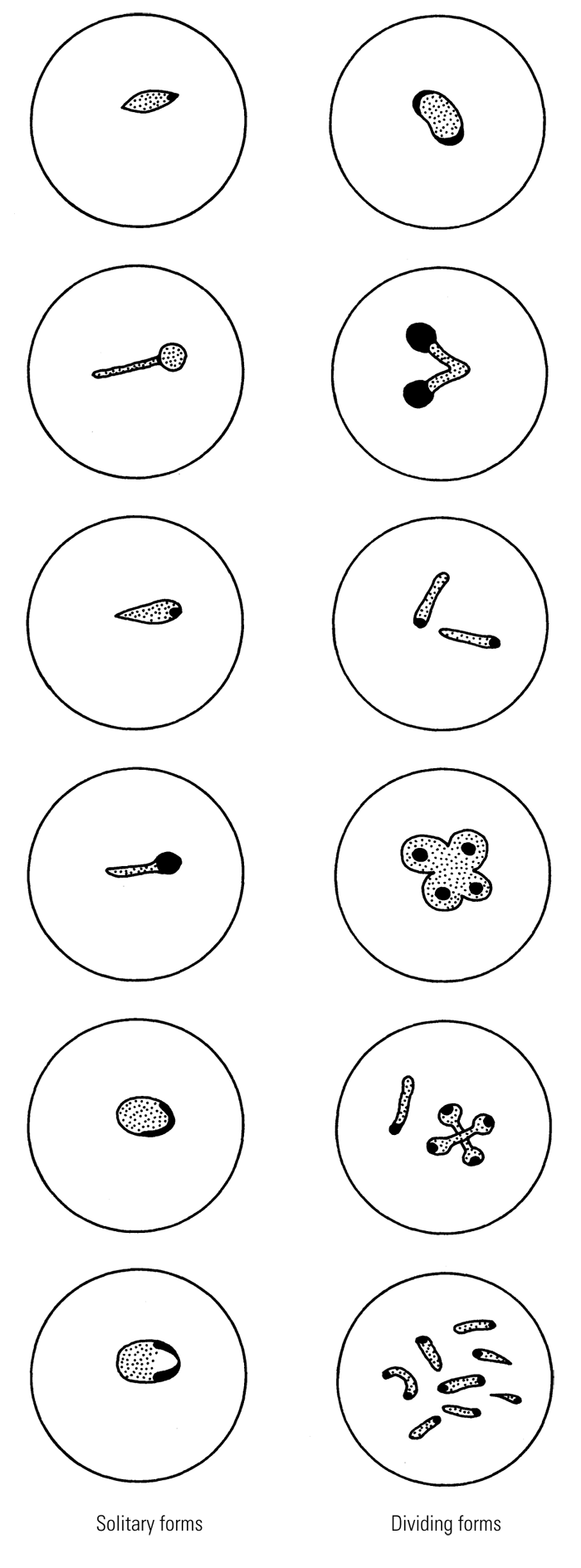
The intra-erythrocytic piroplasms were first described by Koch68 in 1898, although he mistook them for a developmental form of Babesia. The majority are rod-shaped, but round or oval forms are also seen (Figures 29.5 to 29.8). They measure 1 to 2 μm at their greatest dimension, with a neutral or pale blue cytoplasm and a red chromatin granule at one pole in Romanowsky-stained blood smears.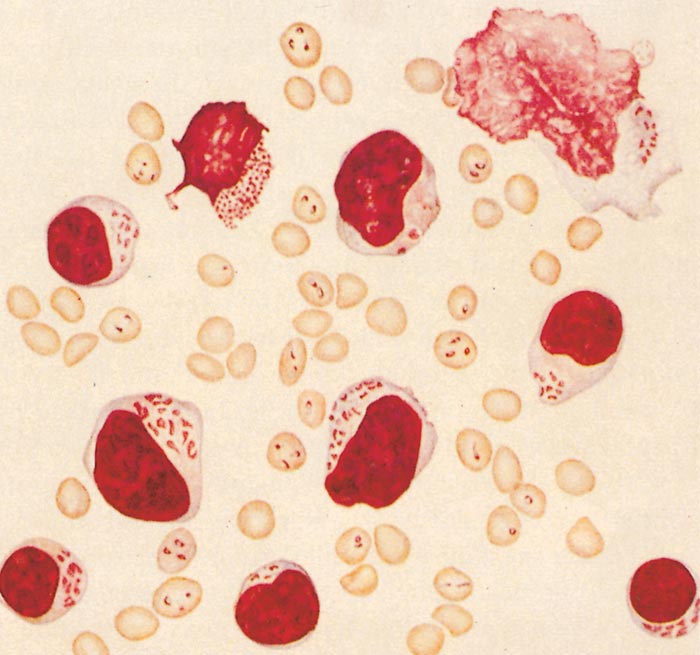
Figure 29.5 Blood smear: drawing of lymphocytes and lymphoblast containing macro- and microschizonts and red blood cells infected with piroplasms. (By courtesy of the Editor of the Onderstepoort Journal of Veterinary Research. Printed in Volume 27, Number 3, 1957)
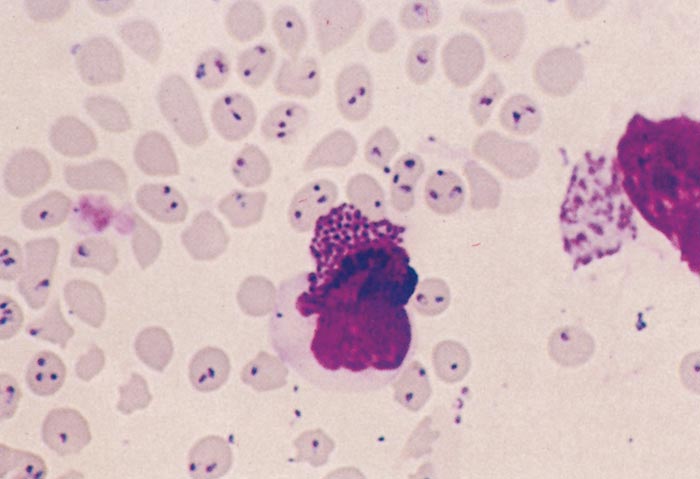
Figure 29.6 Blood smear of a bovine infected with Theileria parva
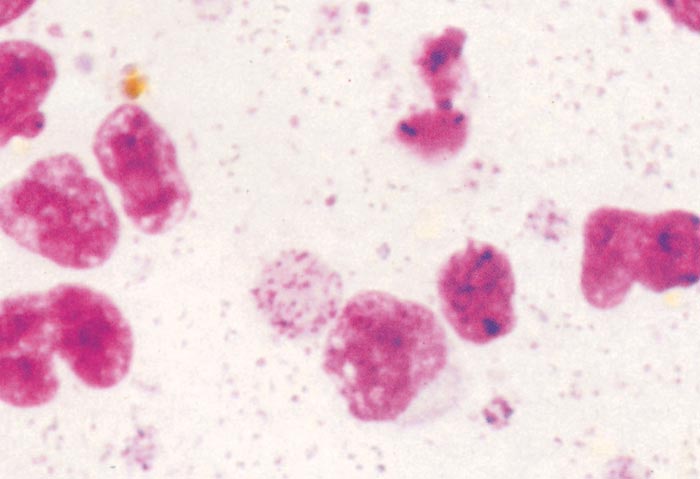
Figure 29.7 Spleen smear of a bovine infected with Theileria parva
Although usually single, there may be several parasites in one erythrocyte in heavy infections. Numbers vary considerably and as many as 80 per cent of the erythrocytes may be infected.118
Following experimental infection, schizonts may be detected between 5 and 15 days after attachment of the tick,118 and they persist for a variable length of time. Piroplasms are likely to appear four to five days later and usually disappear about 10 days after final recovery of the animal, but may persist indefinitely.
The schizont of T. parva was first cultivated in vitro by Hulliger, Wilde, Brown and Turner 61 in 1964 and was subsequently established in bovine lymphoblastoid cell lines by Malmquist, Nyindo and Brown78 in 1970. Lymphoid tissue or circulating lymphocytes from infected cattle can be cultured directly or seeded onto a feeder layer of bovine embryo spleen cells. Schizont-infected lymphoblasts divide and become established as a self-perpetuating line of cells growing in suspension. The schizonts separate into daughter schizonts in association with the mitotic apparatus of the host cell during division and are distributed one to each daughter cell, with the result that almost all the cells in the culture are infected. Cultures can also be established by the inoculation of uninfected lymphocytes with sporozoites derived from infected ticks.24 Schizont cultures have proved extremely useful as antigens for use in serological tests to detect serum antibodies in cattle,26 as antigens for monoclonal antibody characterization of isolates of the parasite,141 as a source of DNA for molecular studies,34 as target material for screening chemotherapeutic agents,84 and as a source of infective material for transmission to cattle.23 Techniques for the propagation of theilerial parasites in culture are described in detail by Brown 18, 20.
In the field, transmission of East Coast fever takes place only through the medium of the tick vector. The natural vector is R. appendiculatus, a common parasite principally infesting the ears of cattle and other herbivores in the more humid areas of southern and eastern Africa (See Vectors: Ticks).77 Rhipicephalus zambeziensis and R. duttoni act as vectors of Corridor disease in arid areas of southern and eastern Africa and Angola respectively 128. Other members of the genus Rhipicephalus and several Hyalomma spp. have been shown to be capable of transmitting T. parva in laboratory conditions (Table 29.1).16, 17, 72, 118 There is, however, no evidence to suggest that any tick other than R. appendiculatus plays any significant role in transmission of East Coast fever in nature.
Table 29.1 Natural and experimental vectors of Theileria parva
| NATURAL VECTORS | EXPERIMENTAL VECTORS |
| Rhipicephalus appendiculatus | R. capensis |
| R. zambeziensis (Corridor disease) | R. carnivoralis |
| R. duttoni (Corridor disease) | R. compositus |
| R. evertsi evertsi | |
| R. kochi | |
| R. pravus | |
| R. pulchellus | |
| R. simus | |
| Hyalomma anatolicum | |
| H. dromedarii | |
| H. impressum |
Rhipicephalus appendiculatus is a three-host tick, and transmission of T. parva is achieved only by nymphs infected during the preceding larval stage or by adults infected during the preceding nymphal stage. Transovarial transmission does not occur, nor is there transmission between larva and adult if the intervening nymph feeds on a non-susceptible host. The tick acquires infection when it feeds on an ox at a stage of clinical disease or recovery when piroplasms are present in the blood. The succeeding stage transmits infection as it feeds when the sporozoites in its salivary glands have matured.
At moderate to low ambient temperatures maturation of sporozoites in adult ticks is not complete until three to four days after feeding commences;129, 144 maturation in nymphs is more rapid.142 At ambient temperatures of 37°C or above, sporozoite maturation can occur in the unfed tick,185 and infection may be transmitted as soon as the tick begins to feed.
Artificial transmission of T. parva can be achieved in two ways. The first is by the inoculation of a suspension of sporozoites prepared from homogenized tick salivary glands or whole ticks, the so-called GUTS (ground-up tick supernatant) preparation.37, 163 This method mimics natural transmission and obviates possible variations in tick attachment and feeding behaviour. By pooling a number of ticks before homogenization it is also possible to eliminate the variation in infectivity between batches of ticks. The method is widely used for the characterization of isolates of T. parva in cattle and for the infection and treatment method of immunization (see Control: Immunization, below). Stabilates of infective sporozoites can be stored in liquid nitrogen and will maintain their viability and biological characteristics for long periods.38
The second method is by the inoculation of schizonts. Suspensions of schizonts in spleen and lymph nodes from infected animals were administered intravenously by Theiler 161, 162 to transmit T. parva as a method of immunization against East Coast fever, but the technique was crude and the results erratic. Suspensions of infected lymphoblasts in culture provide a far safer and more reliable source of schizonts. Infection can be transmitted regularly to recipient cattle when 108 infected donor cells are administered subcutaneously.19 However, when lower cell doses are used, results are inconsistent and some recipients do not become infected, although an inoculum as small as 102 cells will produce infection in the recipient animal if the recipient’s own cells infected in vitro are used.99 The critical factor appears to be the ability of the inoculated cells to survive for a sufficient period for the parasite to transfer to the recipient host cells — this depends on the degree of histocompatibility between donor and recipient.
Transmission of T. parva by the inoculation of blood can be achieved but the results are very erratic164 as success depends on the presence of sufficient numbers of infected lymphoblasts in the circulation. Techniques have been described whereby blood from infected animals can be transferred to nymphal ticks as a means of isolating the parasite from blood. This can be achieved either by inoculation into a rabbit on which the ticks are feeding,143 or by direct inoculation into engorged nymphs.149
Epidemiology
The epidemiology of East Coast fever and the other theilerial infections has been reviewed extensively by Norval, Perry and Young.128 Theileria parva is probably originally a parasite of African buffalo which has become adapted to cattle. It has been found to infect waterbuck (Kobus defassa)154 under natural conditions, and the Asiatic buffalo (Bubalus bubalis) under experimental conditions.94 It is not infective to other ungulates, nor to any species of laboratory animal. In East Coast fever infected areas north of the Zambezi River the African buffalo may act as a source of infection of T. parva to cattle, where there is contact, and no distinction is drawn between infection with cattle-derived and buffalo-derived parasites for control purposes. South of the Zambezi, infection in cattle originating from buffalo is called Corridor disease.
Theileria parva depends on its principle vector tick R. appendiculatus for transmission from host to host. The potential distribution of East Coast fever is thus restricted to those areas of eastern, central and southern Africa where ox and tick coexist. 75 In eastern and central Africa, this includes much of Kenya, Uganda, Rwanda, Burundi, the eastern part of the Democratic Republic of Congo, areas of southern Sudan bordering Uganda and much of Tanzania. In southern Africa its range is more limited, and it is confined to the northern and central regions of Malawi, the northern, eastern and central regions of Zambia, and the Tete Province of Mozambique, all lying to the north of the Zambezi River (Figure 29.1). The disease caused by T. parva in the Southern Province of Zambia was previously referred to as Corridor disease, but is now recognized as being predominantly cattle-associated, rather than buffalo-associated. The disease was introduced into the Comoros in 2002 by cattle imported from Tanzania that were infected with one of the components of the Muguga cocktail 39.
The tick vector R. appendiculatus is widely distributed throughout the warmer, more humid areas of eastern, central and Southern Africa (See Vectors: Ticks). Within its range its abundance varies considerably, being governed by soil moisture and temperature, presence of suitable habitat and availability of hosts. The distribution of climatic suitability has been plotted on the basis of ecoclimatic indices.137, 138, 157 Interestingly, areas of ecoclimatic suitability exist where the presence of the tick has yet to be recorded,127 illustrating that other factors also play a role in determining distribution. Among these is vegetation cover, which influences the microclimate of the habitat of the tick; overgrazing has a marked adverse effect on the tick population. 124, 183 Also important is the livestock production system employed, particularly with respect to how management affects exposure to ticks. Thus in Kenya there is a considerable difference in tick infestation levels and corresponding infection prevalence between open grazing and stall-feeding (zero grazing) management systems.55 Transmission of East Coast fever may be expected to occur freely in areas where ecological factors permit an average total population of not less than 10 to 15 adult ticks per ox.158, 182 It is important to note that on the margins of R. appendiculatus distribution, the distribution may expand and contract with seasonal or secular changes in climatic suitability.126
The seasonality of East Coast fever varies throughout its distribution, but is generally dependent on rainfall and the abundance of adult stages of the tick vector.151 In the highland and coastal regions of eastern Africa, with its two rainy seasons, seasonality is not very marked, and in areas where all stages occur simultaneously throughout the year, so does East Coast fever. In southern Africa from southern Tanzania southwards, seasonality is much more marked, with clinical disease occurring predominantly during the wet months of January to March, when adult ticks are most active.179
East Coast fever is maintained by a continuous cycle of transmission between cattle and ticks. Ticks will transmit infection if, during the preceding stage of development, they have fed on an ox with circulating piroplasms. Infective cattle may be clinically ill, recently recovered, or persistent carriers. The severity of disease is parasite dose-dependent 36, 43. In general terms, the infective dose the ticks receive will be higher from clinically affected animals than from recovered cases and carriers. For many years it was considered that cattle recovering from infection had a solid, sterile immunity and that ticks could only acquire infection from recently infected animals. There was thought to be no carrier state, and maintenance of infection was thought to depend on a cycle of rapid transmission. This is certainly true of the classical laboratory stocks studied intensively in Kenya (known as ‘Muguga’) and in South Africa (known as ‘Schoonspruit’), and appears to have been true of the disease as it occurred south of the Zambezi River in the first half of the twentieth century.49 However, investigations in Kenya have demonstrated that the carrier state appears to be widely prevalent in endemic East Coast fever areas.66, 184
Within the infected areas, the incidence of the disease can vary widely depending on numerous factors, including the abundance of the vector, the susceptibility of the host, and the presence of African buffalo. There is a marked variation in the reaction of cattle of different breeds and from different epidemiological situations to infection. Zebu cattle from ECF-endemic areas are more resistant to disease in the field than Zebus from non-endemic areas and Taurine breeds128. Though they are equally susceptible to infection they show a less severe and shorter clinical response and are less likely to succumb 115, 116. The severity of disease has also been found to be reduced when animals are already infected with other Theileria species, T.mutans and T.velifera 180.
Three major epidemiological forms or states of East Coast fever are recognized.128, 136 These are endemic stability, endemic instability and epidemic. Epidemiological states are based on geographical region and/or livestock production systems and are a result of interaction of broad environment and animal genetic factors 54, 64. However, these factors are not static as they are part of the dynamic socio-demographic and environmental processes. For example, fragmentation of large commercial farms and traditional grazing areas, and changing smallholder market-oriented production systems disturb the ecological habitats of ticks and thereby alter the frequency and distribution of the disease. The situation is further complicated by the variability of cross-protective immunity between parasite populations.
Endemic stability describes an ecological balance between ox, tick and parasite in which regular transmission of T. parva occurs in all age groups of the cattle population and high levels of population immunity are achieved. 7, 96 This situation was first recognized by Bruce in Uganda25 and was characterized by Mettam and Carmichael.92 Endemic stability is seen where indigenous Zebu cattle are kept on pasture in areas highly suitable for the vector, and where major seasonal fluctuations in vector abundance do not occur. Infection prevalence in ticks may be in the region of 1 or 2 per cent,74, 97 infection intensity in infected ticks is low, infection of calves takes place early in life, and reinfection occurs throughout life. It is most common in the smallholder systems prevalent in the humid areas surrounding the Lake Victoria basin, the coast of Kenya and Tanzania and an area encompassing southern Tanzania, northern Malawi and eastern Zambia, where T. parva infection prohibits or severely limits the keeping of more productive Taurine breeds of cattle.128 Endemic stability for East Coast fever, unlike most other tick-borne diseases, does not imply absence of clinical disease 5, 96, 97. Up to 50% of calves may show evidence of clinical infection within the first six months, during which there is a setback to growth and probably increased susceptibility to other parasites and diseases in calves that survive, and up to 10% may die from uncomplicated or complicated East Coast fever, despite treatment.
Endemic instability is the situation in the majority of infected areas, where a combination of environmental effects on the tick and acaricide use inhibits regular transmission of infection. Mortality varies widely from property to property and year to year and may be very high where susceptible cattle are introduced into endemic areas without proper precautions, or where tick control breaks down.
Epidemic East Coast fever occurs when the disease is introduced to areas previously free of the disease where the vector is already present, such as southern Africa and Comoros, and often occurs on a seasonal or secular basis at the margins of R. appendiculatus distribution.181, 182
Pathogenesis
Theileria parva primarily affects lymphoid cells and the course of infection in the ox may be divided into three stages: a prepatent stage; a stage of lymphoid proliferation; and a stage of lymphoid disorganization and depletion.63
The prepatent stage covers the period between the inoculation of sporozoites by the tick and the appearance of schizonts in the drainage lymph node. The sporozoites invade lymphocytes and develop into recognizable schizonts after two to three days. There is evidence to suggest that, although sporozoites can infect a similar proportion of both T and B lymphocytes,6 it is the subsequent rapid multiplication of infected T cells that is responsible for the pathogenicity of the parasite.101 Different isolates of T.parva have been shown to infect different subsets of T lymphocytes, and this has been suggested as a possible reason for variations in virulence 165.
The schizonts stimulate transformation and proliferation of both infected 29, 98, 167 and non-infected 60 T lymphocytes. Lymphocyte proliferation commences at the site of inoculation, but from five days after infection parasitized cells begin to appear in the draining lymph node.
The appearance of parasitized cells in the lymph node coincides with the onset of fever and stimulates active lymphoid proliferation and the release of large numbers of infected and non-infected lymphoblasts into the efferent lymph.100 After a further two to three days, more distant lymph nodes in the chain become infected and hyperplastic, and parasitized and non-parasitized lymphoblasts enter the peripheral circulation. These establish in other lymph nodes and lymphoid tissue (thymus, spleen), as well as in many parenchymatous organs (notably liver, kidneys, lungs, myocardium, adrenals), bone marrow, gastrointestinal tract mucosa and sometimes the brain. The distribution of parasites in non-lymphoid organs is variable and may be associated with incidental pre-existing lymphocyte infiltration.
Four to five days after the initial appearance of schizonts, the process of lymphocyte proliferation is succeeded by a process of lymphocyte destruction, and lymphoid organs and lymphoid foci in parenchymatous organs may show evidence of necrosis and depletion of lymphocytes. Both parasitized and non-parasitized lymphocytes are destroyed and this appears to be the result of activation of specific protective cytotoxic T-lymphocytes (CTL) and non-specific CTL (‘natural killer’ cells).51 Lymphocyte destruction causes a fall in serum immunoglobulin levels152 and immunosuppression. 172 This may enhance the progress of the disease and facilitate secondary respiratory infections.
In non-fatal cases, development of specific protective immune responses terminates schizont proliferation. Recovery from infection is thought to depend on the survival of a sufficient number of effector cells over the first 14 days and on their capacity to achieve a specific protective response.51 A proportion of recovered animals remains unthrifty and unproductive, and foci of infected lymphoid cells may be found persisting in a variety of organs.45 Immunosuppression may predispose such animals to secondary infections which may eventually prove fatal.
Lymphocyte proliferation and destruction were thought previously to be responsible in a variety of ways for the lesions which characterize East Coast fever. Recent investigations have revealed that the infiltrating lymphocytes are accompanied by large numbers of histiocytic cells that are responsible for severe lymphohistiocytic vasculitis 53. This results in tissue destruction in lungs, lymph nodes, spleen, liver, and other organs, together with severe pulmonary oedema that is often the final cause of death. The specific subset of histiocytes that predominates in lesions is CD163+, which is associated with macrophage activation syndrome, a syndrome observed in a wide variety of neoplastic, autoimmune and infectious diseases, including bovine trypanosomosis. The CD163+ macrophages inhibit proliferative and cytotoxic T-cell responses. They also produce large quantities of IL-17, a pro-inflammatory cytokine, which binds to endothelial cells and is responsible for the vasculitis.
The invasion of erythrocytes by piroplasms is a striking feature of the disease, but the parasite in this form does not have any consistent pathogenic effect. Non-regenerative anaemia and icterus are sometimes described in East Coast fever, but no evidence of a haemolytic process has been presented.
Immunity to T. parva is considered to be principally cell- mediated 89, 98, the most important effector cells responsible for clearance of infection and recovery being CTL, which belong to the CD8+ subset of T cells. These recognize specific Theileria antigen combined with major histocompatability complex (MHC) antigens on the surface of schizont-infected lymphoblasts. DNA markers have revealed great antigenic heterogeneity between and within isolates and continuous change with passage through cattle and ticks as a result of genetic recombination 119. Immunity is highly strain specific and is focused on a small number of immunodominant antigens that vary between animals with different class 1 MHC genotypes, which explains the variable degree of protection to heterologous challenge observed in cross-immunity trials in cattle. Other elements of the immune response also contribute to protection, notably parasite-specific CD4+ T lymphocytes. Re-exposure of recovered animals to homologous challenge stimulates activation of memory T lymphocytes which, in turn, stimulate activation of CTL, which eliminate the infection, although some development of schizonts and piroplasms may occur and cause a mild clinical response.
Humoral responses to schizonts are not thought to be important in the immunity of recovered animals, but circulating antibodies which develop against schizonts do provide a useful indication of exposure and are the basis for serological diagnostic tests for T. parva (see Diagnosis, below). Cattle repeatedly exposed to sporozoites, as occurs in the field in areas of high T. parva challenge, develop antisporozoite antibodies capable of neutralizing sporozoite infectivity in vitro.109 In these situations, antisporozoite immunity may play a role in protection. Significantly, the neutralizing activity of antisporozoite antibodies is active against a wide range of T. parva isolates, unlike the antischizont immunity described above.110
Clinical signs
East Coast fever in the classical form, as first described by Gray and Robertson57 in 1902, is characterized by pyrexia, enlargement of the superficial lymph nodes, severe pulmonary oedema and wasting. It usually terminates in death. The incubation period is generally about 15 days from the time of attachment of the infected tick, but may range from 8 to 25 days. The period and the course of the disease become shorter as the challenge is increased.63 The first clinical signs are fever and increases in pulse and respiration rates. There may be a sharp decline in milk production. The parotid lymph nodes, which drain the ear to which the infected tick has attached, are enlarged. After a few days the animal becomes depressed and lethargic. The temperature continues to rise, often to 41 or 42 °C. Anorexia may develop but is not inevitable. Lachrymation commonly occurs together with oedema of the eyelids, and may be accompanied by photophobia. The animal is often constipated. There is a generalized enlargement of the superficial lymph nodes; the prescapular and precrural nodes become very prominent. The disease usually progresses over a period of about 15 days, but may terminate after five days or be prolonged to 25 days. The fever remains high, although in a small proportion of cases there may be a temporary remission for one or two days. Appetite and rumination become increasingly depressed and there is a severe loss of body condition, increasing weakness and ataxia, and frequent recumbency. Constipation is succeeded by diarrhoea and there may be blood and mucus in the faeces. Opacity of the cornea may develop and petechiae may be detected in the mucous membranes, especially those beneath the tongue and in the vulva. Anaemia and icterus have been reported but are inconsistent. Nodular skin lesions have been described in a small number of experimental cases.171 Pregnant cows may abort. In the terminal stages of the disease dyspnoea develops, with an increased respiratory rate, a watery cough and the discharge of frothy fluid from the nostrils. The discharge is copious both when the animal is recumbent and immediately before death. Evidence of pulmonary oedema and hydropericardium may be detected on auscultation. Sternal and submandibular oedema may be present. The enlarged superficial lymph nodes begin to regress, the rectal temperature falls to subnormal levels, and the animal becomes recumbent and dies in a coma.
A small number of animals, usually about 5 per cent, may recover, but convalescence is prolonged and the animals may remain emaciated and unproductive for months.
The disease may assume a less severe form in animals with partial immunity or inherent resistance, but pyrexia and enlargement of superficial lymph nodes remain constant features and in calves may be accompanied by persistent unthriftiness.97 Mild disease has also been reported following infection of fully susceptible animals with a number of strains of T. parva of reduced virulence 9, 166. An atypical cerebral form of disease attributed to T. parva infection is described in Turning Sickness.
In the Asiatic buffalo the disease resembles that in the ox,70 but in the African buffalo infection with cattle-derived parasites is invariably subclinical or mild.10
Pathology
East Coast fever is characterized by a severe panleukopenia which coincides with the onset of the febrile reaction.156, 177 In fatal cases, leukocyte counts fall precipitously until the animal dies. Non-regenerative, normocytic, normochromic anaemia, in which erythrocyte counts may fall to 50 per cent of normal, together with thrombocytopenia and icterus, have also been reported on occasion as prominent features of the disease. There is a moderate hypoproteinaemia, and in the terminal stages there may be clinical pathological changes in serum indicative of renal and hepatic dysfunction.
Schizonts may be present in lymphoblasts and piroplasms in erythrocytes in smears made from peripheral blood, depending on the stage of the disease.
At necropsy the most prominent feature is often severe pulmonary oedema.57 There is a copious amount of froth in the bronchi and trachea and often also in the nasal passages. Emphysema of the lungs may develop, and a moderate hydrothorax and hydropericardium may also be present. There is a variable degree of enlargement of peripheral and visceral lymph nodes, which results from hyperplasia and necrosis of cells of the lymphocyte series, as well as oedema, giving the lymph nodes a greyish-white, fleshy appearance. The nodes are sometimes discoloured by hyperaemia and haemorrhage. Splenomegaly may be present but is inconsistent. In young animals the thymus may be enlarged.
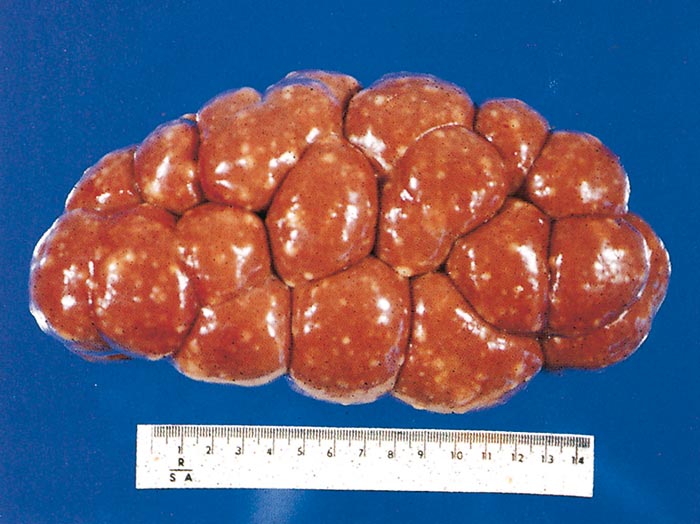
The kidneys frequently exhibit one or more raised red or whitish foci or nodules up to 20 mm in diameter within the cortex (Figure 29.9). Traditionally, but incorrectly, these are known as ‘infarcts’ but they are simply aggregations of large lymphoid cells.
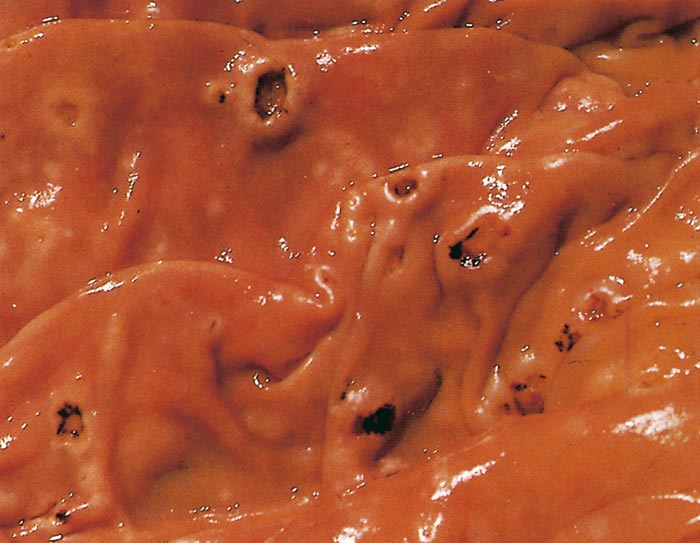
The mucosa of the abomasum is generally thickened, due to lymphoid hyperplasia, and congested. Petechiae or ecchymoses may be present and a few ulcers or superficial erosions up to 10 mm in diameter may be found, particularly in the pyloric region. The ulcers are associated with the mucosal lymphoid hyperplasia (Figure 29.10).
Similar lesions may occur in the intestinal mucosa, particularly in the Peyer’s patches of the terminal ileum.
Opacity of the cornea of the eye is often noted, especially at the limbus, and results from infiltrations of lymphocytic cells and from oedema.
Other changes may include emaciation; subcutaneous and intramuscular oedema; ascites; serous atrophy of fat; pallor of the liver as a result of mild degeneration; petechiae in the mucous membranes of the oral cavity, serosal membranes, and the epi- and the endocardium; anaemia; icterus; and grayish-brown foci of hyaline degeneration and necrosis of muscles, particularly in the larger muscle groups of the hindquarters. Hyperplasia of lymphoid tissue in organs and tissues in which it is normally present, or lymphoid infiltration of other organs, such as the kidneys (Figure 29.11), the liver and the eyes, may be recognized macroscopically and confirmed histologically.156
Histologically, corticomedullary distinction in lymph nodes is largely lost, and normal architecture replaced by sheets of macrophages and lymphoblasts separated by large aggregates of necrotic cells, fibrin and haemorrhage 53. Small to medium calibre blood and lymphatic vessels are often disrupted by mononuclear vasculitis, characterized by fibrinoid degeneration, necrotic cellular debris, oedema, and macrophage and lymphocyte infiltration. In the lungs, small to medium calibre blood and lymphatic vessel walls show mononuclear vasculitis. Alveoli and interlobular septae are distended by oedema, fibrin, haemorrhage, and alveolar macrophages. Hepatic portal triads are rimmed by moderate to large numbers of macrophages and lymphoblasts, and portal vessels are mildly disrupted by vasculitis. Within the spleen, the distinction between the red and white pulp is often inapparent. Periarteriolar lymphatic sheaths are often greatly expanded by monomorphic populations of lymphoblasts or replaced by sheets of macrophages. Numerous macrophages are filled with haemosiderin, suggesting increased erythrocyte turnover and erythrophagocytosis. Lymphoid infiltration is often present in other parenchymatous organs, notably the myocardium, the adrenals, the bone marrow, and the perivascular spaces in the brain. Lymphoblasts are characterized by their large size, a large hyperchromatic nucleus, and relatively extensive hyperchromatic cytoplasm. Mitotic figures are common. 8, 40 Schizonts may be seen in some of the cells, the characteristic feature in a haematoxylin-and-eosin-stained section being a cluster of basophilic granules within a lightly staining vacuole in the cytoplasm. In the terminal stages of infection, necrosis of lymphocytes supervenes and the lymphoid tissue becomes rather depleted of cells, with cellular debris being scattered through the tissue.
Piroplasms in erythrocytes and schizonts in lymphoblasts may be recognized with the transmission electron microscope. Ultrastructural pathological changes in lymph nodes have been described by De Martini and Moulton.40
Diagnosis
The diagnosis of classical East Coast fever is based on the characteristic clinical signs and lesions, and may be confirmed by demonstration of schizonts and, in the later stages of the disease, piroplasms (Figures 29.3 to 29.8). The presence of R. appendiculatus on the animal provides supportive evidence, but tick infestation may be very light and the ticks may have detached by the time the animal is examined. Theilerial parasites are generally numerous. In the live animal they can be demonstrated in smears prepared from peripheral blood and fine-needle aspirates of superficial lymph nodes (Figures 29.3 and 29.4). Parotid, prescapular or precrural lymph nodes may be sampled. In the dead animal, smears prepared from the cut surface of the spleen and any enlarged lymph nodes should be examined. Smears must be of good quality and should be stained with Giemsa or any other Romanowsky stain. The presence of large hyperplastic lymphoblasts should be an incentive to continue the search for schizonts if they are scanty. Care must be taken to differentiate between schizonts and normal azurophilic granules. When smears have not been taken, the diagnosis may still be confirmed if infiltration with infected lymphoid cells can be demonstrated in routine histological preparations. The kidneys, liver, lungs and brain are particularly rewarding tissues for examination.
Intercurrent infection with other haemoparasites may complicate the clinical and pathological picture, and these parasites may be detected microscopically. In those areas where it occurs, East Coast fever is generally the most virulent of the haemoparasitic diseases of cattle, and its possible presence should be given priority over any other disease when making a diagnosis.
Methods of retrospective confirmation of the diagnosis of Theileria have been reviewed by Mans et al., (2015). Circulating antibodies to T. parva are detectable 2-4 weeks after infection and persist for a variable period, depending on the persistence of the parasite in the host. Rising titres can be detected using a variety of serological tests. More usually, serology is used in cross-sectional surveys or longitudinal studies to detect the presence of T. parva infection and to determine the prevalence, incidence and seasonality of infection and disease. Circulating antibodies were first demonstrated in recovered animals by the complement fixation test 76 and since then various techniques have been developed using schizonts or piroplasms as antigen. The indirect fluorescent antibody test (IFAT), which employs cultured T. parva schizont-infected lymphoblasts as the antigen, is widely used but suffers from subjectivity in interpretation and lack of specificity due to cross-reactivity with some other Theileria spp., most importantly T. taurotragi, which is widespread throughout the range of the vector tick. Following the discovery that there are single molecules that can be employed as species-specific antigens the serological test of choice for T. parva-specific antibody detection became the enzyme-linked immunosorbent assay (ELISA) based on the use of a recombinant polymorphic immunodominant molecule (PIM). The PIM based ELISA detects antibody of IgG1 isotype, is more sensitive than the IFAT, and antibody levels remain detectable after a single infection with T. parva for longer than with the IFAT. 27 This assay was commercialized but later discontinued due to problems associated with specificity 130; currently its use is limited to research.
An alternative means of retrospective diagnosis is the detection of infection in recovered carrier cattle (see Epidemiology, above). Morphologically, the piroplasms of different Theileria spp. are difficult to differentiate especially when present in small numbers. Thus, the examination of Giemsa- stained thin blood smears to detect piroplasms lacks specificity and is also relatively insensitive. Tick transmission (xenodiagnosis) is not practical on a large scale. The need for an improved method of detection of T. parva carrier animals has led to the development of a number of molecular diagnostic assays that employ oligonucleotide primers targeting T. parva genes 80 : Reverse Line Blot (RLB) hybridization assay targeting the 18S rRNA gene; conventional and nested polymerase chain reaction (PCR) techniques for p104 gene as well as a repetitive gene sequence (TpR); real-time PCR targeting the 18S rRNA gene; loop mediated isothermal amplification (LAMP) for PIM and p150 genes; and the Pan-FRET real-time PCR targeting the Cox III gene. Real-time and nested PCRs are used for routine diagnostics. Reverse line blot assays are valuable for surveys of the infection status of a herd, as they screen for 24 Babesia and Theileria species at once, but suffer from low sensitivity in mixed infections. Each assay has advantages and disadvantages, and the assay of choice depends on the question to be answered.
Differential diagnosis
Clinically, East Coast fever must be differentiated from other febrile conditions such as mucosal disease and bovine malignant catarrhal fever which usually terminate fatally after a course of one to two weeks and which feature progressive emaciation, respiratory distress, diarrhoea, corneal opacity, and lymphoid hyperplasia. These two diseases may be distinguished by the presence of ulcerating or necrotizing inflammatory reactions in buccal and nasal mucosae.and contagious bovine pleuropneumonia and other pneumonic conditions are characterized by coughing and evidence of pain on respiration. Acute trypanosomosis does not exhibit diarrhoea or severe respiratory distress. In babesiosis and anaplasmosis, anaemia and icterus are the major presenting signs. The clinical picture in East Coast fever may be complicated, however, by secondary respiratory infections or by concurrent infection with other haemoparasites. It is important for the investigator to look beyond the immediate presenting signs for the primary lesions of lymphoid hyperplasia and subsequent depletion.
A definitive diagnosis of East Coast fever is possible if microscopic examination of blood and lymphoid tissues reveals large numbers of theilerial parasites. If only small numbers of schizonts and piroplasms are present in association with R. appendiculatus ticks and severe clinical illness, the possibility exists that the disease under investigation is not classical East Coast fever but is caused by infection with buffalo-derived T. parva (See Corridor disease). Where schizonts are few and a variable number of piroplasms are encountered in mildly affected animals, the presence of another species of Theileria should be considered, or it may be that the T. parva infection is incidental. For example, Zebu-type calves in areas endemic for East Coast fever tend to undergo moderate, mild or inapparent reactions on infection which do not require treatment. The presence of piroplasms in association with anaemia, minimal lymph node enlargement and few, if any, schizonts is likely to indicate T. mutans infection.
To distinguish between the different species of Theileria microscopically may be extremely difficult when relatively mild infections are occurring, particularly as mixed infections are frequent in the field,62 the most common of these being combinations of T. parva, T. taurotragi and T. mutans. Morphologically the piroplasms are difficult to differentiate, except for those of T. velifera, and the schizonts are indistinguishable, except for those of T. mutans.
Serological examination of recovered animals may permit a retrospective differentiation at the species level with a species-specific ELISA available for T. mutans 67 and standard IFAT for others, although the test for T. parva cross-reacts with T. taurotragi at low titres. Biological transmission of the parasite may also provide a means of differentiation at the species level, but is expensive and requiresthe availability of cattle for experimental purposes. Theileria mutans and T. velifera are readily transmitted by intravenous inoculation of heparinized blood to a susceptible ox — an inoculum of 10 ml should be adequate. Theileria taurotragi can sometimes be transmitted in the same way but may require a larger inoculum (100 to 200 ml), while T. parva cannot readily be transmitted by blood inoculation. Tick transmission may also be helpful; T. mutans and T. velifera are transmitted by Amblyomma spp. but not by Rhipicephalus spp., while the converse is true for T. parva and T. taurotragi.
A number of molecular, biochemical and immunological tools are available that enables differentiation between Theileria spp.. Polymerase chain reaction (PCR) combined with oligonucleotide DNA probes detecting ribosomal RNA sequences can be applied to lymph node, blood and culture material, even when only small quantities are available.1 Where larger amounts of DNA can be purified from schizont-infected cultures or piroplasm-infected blood, two additional methods are useful: restriction fragment length polymorphism (RFLP) patterns obtained when an extrachromosomal fragment is used as the DNA probe;65, 103 and glucose phosphate isomerase isoenzyme patterns.2, 91 Species- specific monoclonal antibodies used in the IFAT are also valuable for recognizing piroplasms56, 67 and, where available, schizonts in cell cultures.15, 93, 141
Differentiation of Theileria sp. (buffalo) is of particular importance, as it is similar to T. parva, based on the 18S rRNA gene sequence, and has been found to be widespread in buffalo populations in Kenya, Uganda and South Africa 12, 28, 81, 131, 132, 140. This species usually co-infects with T. parva and has occasionally been detected in cattle,although it seems to be transmitted by a different vector 12. In areas where cattle co-graze with T. parva-infected buffalo, a species-specific assay is required to confirm T. parva infections. This can be done using some of the conventional and real-time PCR assays already mentioned, the conventional PCR for p104 and p67 120 as well as the real-time PCR targeting the 18S rRNA gene. Furthermore, there is a potential to develop differential diagnosis assays of these parasites based on the nucleotide substitutions in Tp6, Tp7 and Tp8 gene orthologues unique to T. sp. (buffalo) 12.
The differentiation between the various disease manifestations caused by T. parva is based on epidemiological, clinical, pathological and parasitological factors, and on the implications of the diagnosis in relation to the animal health control policy of the country. Veterinarians in countries that have eradicated (or are on the verge of eradicating) East Coast fever are far more careful to make a distinction between the forms of disease than those in regions where East Coast fever is endemic.
Control
Treatment
Progress in the chemotherapy of East Coast fever has been reviewed by Dolan.44 The disease proved refractory to treatment for many years until Neitz,117 in 1953, found that certain tetracyclines had a marked suppressive effect on the schizonts if administered during the incubation period.
Tetracyclines have been widely used in the treatment of clinical disease, sometimes to good effect, but they have not proved to be completely reliable therapeutic agents. They have been used successfully, however, in immunization against East Coast fever by an infection-treatment technique (see Immunization, below).
An important breakthrough in the control of East Coast fever was achieved in the late 1970s by the development of two highly effective therapeutic agents. The first, parvaquone, 83 a napthoquinone, achieved recovery rates of 90 per cent or better in field trials.31, 107 The drug is effective against both schizonts and piroplasms, but treatment does not achieve a parasitological cure, and recovered animals may remain intermittent carriers and some may take several months to return to normal productivity.45 An analogue, buparvaquone86, has since replaced parvaquone. It is recommended at a dose of 2,5 mg/kg for the treatment of clinical theileriosis,83 although, under field conditions where clinical cases are often detected late in the course of the disease, a two-dose regimen is advised with the doses being given 48 hours apart.46 Supportive treatment with the diuretic frusemide to relieve pulmonary oedema is beneficial in severe cases (Azuba Musoke et al. 2004).
A second drug, halofuginone, a quinazoline derived from febrifugine, achieved recovery rates of 80 per cent or higher in the field30, but the therapeutic index is relatively low and its use has been discontinued.
Buparvaquone has the disadvantage of being relatively expensive and this reduces its application in the field, but it can be used as a back-up to prevent mortality if other forms of control prove ineffective. Experimental work suggests that buparvaquone could be used chemoprophylactically in areas of intense T. parva challenge. The administration of a single dose of buparvaquone at 5 mg/kg seven days after field exposure has commenced may protect susceptible cattle from disease while they become immune.118
To prevent the danger of carrier animals, no affected animal may be treated chemotherapeutically in South Africa without the written authorization of the National Director of Animal Health and Production, but there are no such restrictions in other countries in the region.Immunization
The development of methods of immunization against East Coast fever has been reviewed by Brown.21 Protective immunity to T. parva persists for at least five years after natural infection.118
Immunity will develop only in animals in which infection with schizonts has been established. Effective methods of immunization are achieved by controlled infection of cattle using sporozoites derived from ticks, or schizont-infected cells. The infective material is virulent and the method of administration must be carefully regulated to avoid a fatal outcome.
In the early years of the twentieth century, before the introduction of acaricides, crude techniques of controlled infection were developed for immunization. In Kenya, animals from endemic areas which were thought to be immune were exposed to natural infection on heavily infected pastures. The majority survived and could be used thereafter as transport oxen through infected areas.128 In South Africa, cattle were immunized by the intravenous inoculation of preparations of spleen and lymph node from infected animals, and up to 70 per cent were protected against natural challenge.162 There was a significant mortality from theilerial infection or from bacterial contamination of the inoculum as a direct result of the immunization procedure, but the technique was used to control the first onslaught of the disease in the Transkei region of the Eastern Cape Province. A total of 275 000 cattle was inoculated and it is estimated that 100 000 survived.147
No further progress was made until 1953, when Neitz discovered that the administration of chlortetracycline (Aureomycin) during the incubation period had a marked suppressive effect on the development of infection and permitted the establishment of a solid immunity.117 His observation was followed up by a team of scientists working in Kenya under the auspices of the Food and Agriculture Organization (FAO) of the United Nations, and an infection and treatment method of immunization was developed.145 A crude stabilate of sporozoites is prepared from adult ticks which have fed as nymphs on infected cattle, have moulted, and have been pre-fed on rabbits for three to four days to allow the sporozoites to mature. The suspension is mixed with glycerol as a cryoprotectant and stored, deep-frozen at −70°C or in liquid nitrogen. Because of the variability in the stabilates, aliquots are subjected to infectivity testing and safety and efficacy titration in susceptible cattle, including homologous challenge (Patel et al., 2016). The stabilate is inoculated subcutaneously into the cattle to be immunized and simultaneously an intramuscular injection of 20 or, more recently, 30mg/kg of a long-acting oxytetracycline preparation is administered.
Infection develops to a mild or subclinical level and the animals are thereafter resistant to challenge, both experimental and natural. The mechanism by which the oxytetracycline works to control infection and enhance immunity is not fully understood, but the drug seems to affect the degree of maturation of sporozoites to schizonts after infection of lymphocytes. One mechanism of protective immunity induced by the technique is thought to be based on major histocompatibility complex (MHC) class-I-restricted CD8+ cytotoxic T cells. The duration of immunity appears to be comparable to that which follows natural recovery. Buparvaquone is also effective as a prophylactic and may be administered at the time of stabilate inoculation.86 However, there is a risk that this drug may be so effective that infection fails to establish and the animal is left partially or fully susceptible.122
The infection and treatment technique provides good immunity against the homologous immunizing strain, but is not consistently successful against unrelated strains. On the basis of extensive cross-immunity trials, three isolates, identified as cattle-derived T. parva Muguga and T. parva Kiambu 5 and buffalo-derived T. parva Serengeti transformed, have been combined in the so-called Muguga cocktail.146 Characterization of the components of Muguga cocktail vaccine has generally shown relatively limited genetic diversity (Hemmink et al 2016; Patel et al., 2011; Norling et al., 2015). Nonetheless it provides protection against most isolates of cattle origin from outbreaks of East Coast fever in eastern Africa and is registered as a vaccine in Tanzania, Uganda, and Malawi (Nene et al 2016; Peters 2009). Protection against buffalo-adapted parasites is variable (Nene et al., 2016). Fears that the use of a vaccine in areas other than those from which the components were isolated carries with it the potential danger of introducing parasites of an antigenic type against which local cattle may have no immunity have led to the use of local strains in some countries, as in the Eastern Province of Zambia (T. parva Katete79) and the Coast Province of Kenya (T. parva Marikebuni114). The Muguga and Serengeti transformed components of the ‘Muguga cocktail’ do not induce carrier status in immunized animals but the Kiambu 5 component does, for up to four years, as do both T. parva Marikebuni and T. parva Katete, and has been shown to transfer to non-immunized animals in the field 5, 42, 88. Genetic recombination in ticks occurs between immunizing and indigenous parasites in both immunized and non-immunized animals, presumably establishing a broad-based immunity in both. There has been no evidence of disease outbreaks in non-immunized animals in East Coast fever infected areas attributable to the introduction of immunizing stocks.
The infection and treatment technique is relatively expensive and cumbersome (Di Giulio et al. 2009). Production and testing of stabilates is complex and demanding of time and resources. A liquid nitrogen cold chain is essential in order to maintain parasite viability and, compared to other veterinary immunizations, more training and expertise are required for the vaccine to be delivered safely and effectively. Another important limitation has been the risk of occurrence of clinical reactions after immunization 82. Incorrect drug or vaccine dosing and immunization of cattle under stress from poor nutrition or intercurrent disease are predisposing factors for animals to suffer clinical theileriosis as a result of immunization. Monitoring of animals in the month after immunization may be necessary to allow the identification and treatment of animals that react severely if they are not to die. Serious problems with chronic and cerebral theileriosis in immunized cattle maintained under heavy challenge in traditionally managed herds have been reported in one area of Kenya.95 In spite of these disadvantages, the Muguga cocktail has been used commercially on a wide scale in northern Tanzania since 1998 with a protection rate of >98% (Di Giulio et al. 2009).
The possibilities of immunization against T. parva by inoculation of schizont-infected lymphoblast cultures, which has proved so effective with T. annulata infection, have been studied intensively.19 The technique is possible but impractical, as very large numbers of cells are required to establish infection consistently. Whereas with T. annulata inocula 106 cells will regularly establish infection, with T. parva it requires 108 cells in 100 ml of culture fluid to establish infection reliably in allogeneic animals.47, 111
A number of other approaches to immunization is currently under investigation. The presence of sporozoite neutralizing antibodies in cattle repeatedly exposed to field infection, or to sporozoites by stabilate inoculation, provided the rationale for the development of a potential vaccine based on a recombinant sporozoite surface antigen of T. parva, known as recombinant p67.111 This protects two-thirds of immunized animals in laboratory trials108 and preliminary evidence indicates that it may afford a broad spectrum of immunity against different T. parva isolates.120 Field trials produced varied results; in Kenya, 50% of the immunized cattle and 25% of the control animals exhibited immunity to East Coast fever 113 while immunized and control cattle were equally susceptible to East Coast fever during field trials in Zambia 148. These variations have been attributed to possible differences in antigen doses, adjuvants, and cattle types used in the two trials 119. The definitive subunit vaccine should ideally contain antigens from both the sporozoite and schizont stages, especially as the latter is known to stimulate the main effector mechanism. Identification of the antigens recognized by CTL is the main focus of research towards a vaccine based on the schizont stage.112
Other preventive measures
In the absence of an effective vaccine, control of East Coast fever has depended in the past, and continues to depend, on the prevention of infestation of susceptible cattle with infected ticks. There are three possible approaches:
- Isolation
Susceptible cattle maintained in a closed herd on properly fenced pastures (which exclude all possible contact with other cattle) will be protected from East Coast fever, as infection can only be introduced by ticks which have dropped from infected cattle during the preceding stage of the life cycle. Where the pastures border an area of high risk, such as a public road on which infected cattle may pass, two fences 5 metres apart will provide an effective barrier. Hay and straw from potentially infected areas may introduce unfed ticks mechanically to the property and must be excluded. Other animals, such as horses, sheep and goats, may transport infected ticks mechanically and must be treated with an acaricide if they are introduced. Wildlife may also transport ticks and should be excluded. In situations where fencing is not practicable, cattle may be housed and fodder transported to them (‘zero-grazing’). Isolation as a technique is too prone to breakdown to be relied upon as the sole method of control and is normally used in conjunction with tick control. - Tick control
Susceptible cattle can be maintained in an infected environment provided that they are kept completely free of R. appendiculatus. Dipping of cattle in arsenical preparations at three-, five- or seven-day intervals was widely used in the first half of the twentieth century to control East Coast fever. In conjunction with clipping of hair from ears and tail-brushes to expose ticks at these sites, and local application (‘hand-dressing’) of acaricide to ears and the perineal region, short-interval dipping provided very good control of R. appendiculatus and prevented ticks feeding to engorgement on infected cattle and thus infecting the pasture.175 Control was not sufficiently complete to prevent a proportion of susceptible cattle contracting infection when grazed on infected pastures. 50 Arsenic has been superseded by organochlorines, organophosphates, carbamates, amidines and pyrethroids, and techniques of application with spray races, hand-operated sprays, pour-on formulations and impregnated eartags have been developed as alternatives to the dip tank. Cattle must be treated two or three times a week where the challenge is heavy, and even this may not be sufficient to provide absolute protection. If used in conjunction with isolation, it does reduce the risk of infection to a minimum. Even intensive acaricide use on cattle will not prevent infection where there is contact with buffalo 174. - Destocking
If infected pastures are kept free of cattle for 15 to 18 months, infection will disappear. This period is greater than the limit of survival of the parasite in infected adult ticks in the field.121, 160 Ticks on the pasture will continue to feed on other species but will not pick up parasites for transmission. Destocking was used effectively in the control of East Coast fever in the early part of the twentieth century. Cattle were moved from infected to clean pasture through a series of ‘temperature camps’, animals being checked for evidence of fever daily, and those showing a rise in temperature being culled as potential early cases of infection.59 The infected pasture was fenced and kept cattle-free for 18 months. The technique fell into disuse as the cattle population grew and vacant clean pasture became more difficult to locate. Destocking may also be achieved by the removal of all cattle for slaughter and prohibition of reintroduction of cattle for 18 months. The pasture can be used for sheep or goats in the interim.
Practical approaches to control
Selection of the methods for control of East Coast fever in a given situation will depend largely on the epidemiology of the disease, i.e. whether it is newly established in the area and spreading in an epidemic form, or whether it is endemic 54. Where East Coast fever appears in an area for the first time, every effort should be made to eradicate it. This entails the vigorous application of conventional zoosanitary measures, which include an embargo on all cattle movements into, within, or out of the area; enumeration and identification of all cattle in the area; and regular inspection to detect evidence of infection. In addition, the enforcement of short interval acaricide treatment of all cattle is essential to suppress the tick population and reduce the opportunities for transmission. These measures were applied with considerable success to control the epidemic in southern Africa in the first half of the twentieth century.41
They were supplemented by a programme of intensive surveillance, which involved the examination of blood and spleen smears from every bovine animal which died or was slaughtered other than at a recognized abattoir throughout the infected area, in order to identify residual foci of infection. Infected premises were quarantined for up to two years after the last confirmed or suspected case, and persistent foci of infection were finally cleared by slaughter.
Implementation of measures such as these requires a comprehensive infrastructure including effective legislation, adequate and competent veterinary staff, transport, good communications, fencing, dip tanks or spray races, acaricide and, above all, a stable political environment. Failure to achieve rapid control of the epidemic may well result in the disease establishing permanently in an endemic form.
In an endemic area the greater part of the cattle population is of indigenous cattle that have a measure of resistance to ticks and disease and survives in equilibrium with the disease, though there may well be an increased level of calf mortality and a reduction in productivity. Where intensive regional dipping programmes are introduced, there may be an improvement in the general health of the cattle. However, such programmes are very demanding in terms of finance, organization and effort on the part of owners and veterinary services alike, and in the long run they usually collapse and the cattle revert to their previous stable state. Large production units with improved, susceptible cattle can control the disease effectively by isolation and intensive acaricide treatment, provided they are well managed and have adequate financial resources. However, the costs of short-interval dipping or spraying are very high, both in terms of acaricide and labour and of reduced production resulting from frequent mustering and treatment. The effects of the occasional breakdowns in control which inevitably occur can be minimized with chemotherapy. Small-scale producers, attempting to run small herds of highly susceptible cattle in the midst of an environment of uncontrolled ticks and infection, have little chance of protecting their animals from the disease by conventional zoosanitary methods.
Control of East Coast fever in endemic areas should ideally be based on an integrated programme aimed at reducing the challenge and increasing the resistance of the cattle 54. The use of Zebu or Zebu-cross cattle, which have an increased capacity for the development of resistance to ticks, should be encouraged; they will be subjected to reduced disease challenge and less tick worry. Exposure of calves to moderate tick challenge in early life should be allowed to facilitate the development of tick resistance and to permit infection with tick-borne parasites of all types while the animals are partially resistant.
The use of acaricides should be limited to the minimum required to prevent excessive tick loads; this will reduce production costs substantially. Chemotherapy may be used to treat clinical cases. In addition, current recommendations are that immunization by the infection and treatment method should be implemented throughout except in marginal areas 54. After decades of prevarication while debating the merits of local and universal immunizing stocks and waiting for the development of subunit vaccines, the ‘Muguga cocktail’ of isolates has been accepted as the agent of choice throughout much of eastern Africa 54, and the infection and treatment method of immunization of calves between one and four months of age has been demonstrated to be the most cost-effective method of control, even in indigenous Zebu cattle in an endemic area 5, 42. However, the method only provides variable protection against parasites of buffalo origin where cattle and buffalo co-exist 174.
Whatever the final benefits may be, the conversion from intensive tick control to an integrated control programme based on immunization for East Coast fever is not to be undertaken lightly. In the initial stages of acaricide reduction, cattle of all ages will be at risk and immunization of the whole herd will be necessary before acaricide use is reduced. The herd may also be at risk to other tick-borne diseases. Uilenberg169 has reviewed the results of field immunization trials in Tanzania and Malawi in which, once East Coast fever was controlled, heartwater, babesiosis, anaplasmosis, T. mutans infections and tick toxicosis emerged as significant causes of mortality. In Zimbabwe, a breakdown of dipping services in communal areas in the 1970’s led to severe outbreaks of various tick-borne diseases.125 The inference is that cattle should be immunized against anaplasmosis, babesiosis and cowdriosis as well before the integrated programme is implemented. There are serious drawbacks to this proposal, and it may not always be justified 73. Each case should be judged on its history of tick control and disease and, where there is doubt, a preliminary serological survey of the epidemiological status for each disease should be carried out.
References
- ALLSOPP, B.A., BAYLIS, H.A., ALLSOPP, M.T.E.P., CAVALIER-SMITH, T., BISHOP, R.P., CARRINGTON, D.M., SOHANPAL, B. & SPOONER, P., 1993. Discrimination between six species of Theileria using oligonucleotide probes which detect small subunit ribosomal RNA sequences. Parasitology, 107, 157.
- ALLSOPP, B.A., GIBSON, W.C. & STAGG, D.A., 1985. Characterisation of some East African Theileria species isolates by isoenzyme analysis, with particular reference to T. parva. International Journal for Parasitology, 15, 271–276.
- ANON. 1989. Theileriosis in Eastern, Central and Southern Africa. In: DOLAN, T.T. (ed.) Proceedings of a Workshop on East Coast Fever Immunisation held in Lilongwe, Malawi, 20–22 September 1988.Nairobi, Kenya. International Laboratory for Research on Animal Diseases.
- AZUBA MUSOKE, R., TWEYONGYERE, R., BIZIMENYERA, E., WAISWA, C., MUGISHA, A., BIRYOMUMAISHO, S. & McHARDY, N., 2004. Treatment of East Coast fever of cattle with a combination of parvaquone and frusemide. Tropical Animal Health and Production, 36, 233-245.
- BABO MARTINS, S., DI GIULIO, G., LYNEN, G., PETERS, A. & RUSHTON, J., 2010. Assessing the impact of East Coast fever immunisation by the infection and treatment method in Tanzanian pastoralist systems. Preventive Veterinary Medicine, 97, 175-182.
- BALDWIN, C.L., BLACK, S.J., BROWN, W.C., CONRAD, P.A., GODDEERIS,B.M., KINUTHIA, S.W., LALOR P.A., MACHUGH, N.D., MORRISON, W.I., MORZARIA, S.P., NAESSENS, J &NEWSON,J., 1988. Bovine T cells, B cells and null cells are transformed by the protozoan parasite Theileria parva. Infection and Immunity, 56, 462–467.
- BARNETT, S.F., 1957. Theileriasis control. Bulletin of Epizootic Diseases of Africa, 5, 343–357.
- BARNETT, S.F., 1960. Connective tissue reactions in acute fatal East Coast fever (Theileria parva) of cattle. Journal of Infectious Diseases, 107, 253–282.
- BARNETT, S.F. & BROCKLESBY, D.W., 1961. A mild form of East Coast fever. The Veterinary Record, 73, 43–44.
- BARNETT, S.F. & BROCKLESBY, D.W., 1966. The susceptibility of the African buffalo (Syncerus caffer) to infection with Theileria parva (Theiler, 1904). British Veterinary Journal, 122, 379–386.
- BARNETT, S.F., BROCKLESBY, D.W. & VIDLER, B.O., 1961. Studies on macroschizonts of Theileria parva. Research in Veterinary Science, 2, 11–18.
- BISHOP, R.P., HEMMINK, J.D., MORRISON, W.I., WEIR, W., TOYE, P.G., SITT, T., SPOONER, P.R., MUSOKE, A.J., SKILTON, R.A. & ODONGO, D.O., 2015. The African buffalo parasite Theileria. Sp (buffalo) can infect and immortalize cattle leukocytes and encodes divergent orthologues of Theileria parva antigen genes. International Journal for Parasitology Parasites and Wildlife, 4, 333-342.
- BISHOP, R., KARIUKI, D.P. & YOUNG, A.S., 1992. Detection of a carrier state in Theileria parva-infected cattle by the polymerase chain reaction. Parasitology, 104, 205-232.
- BISHOP, R., SOHANPAL, B., MORZARIA, S.P., DOLAN, T.T., MWAKIMA, F.N. & YOUNG, A.S., 1994. Discrimination between Theileria parva and Theileria taurotragi in the salivary glands of Rhipicephalus appendiculatus ticks using oligonucleotide homologues to ribosomal-RNA sequences. Parasitology Research, 80, 259–261.
- BISHOP, R.P., SPOONER, P.R., KANHAI, G.K., KIARIE, J., LATIF, A.A., HOVE, T., MASAKA, S. & DOLAN, T.T., 1994. Molecular characterisation of Theileria parasites: application to the epidemiology of theileriosis in Zimbabwe. Parasitology, 109, 573–581.
- BROCKLESBY, D.W., 1965. Evidence that Rhipicephalus pulchellus (Gerstäcker 1873) may be a vector of some piroplasms. Bulletin of Epizootic Diseases of Africa, 13, 37–44.
- BROCKLESBY, D.W., BAILEY, K.P. & VIDLER, B.O., 1966. The transmission of Theileria parva (Theiler 1904) by Rhipicephalus carnivoralis Walker 1966. Parasitology, 56, 13–14.
- BROWN, C.G.D., 1979. Propagation of Theileria parva. In: MARAMOROSCH, K. & HIRUMI, H., (eds). Practical Tissue Culture Applications. New York: Academic Press.
- BROWN, C.G.D., 1981. Application of in vitro techniques to vaccination against theileriosis. In: IRVIN, A.D., CUNNINGHAM, M.P. & YOUNG, A.S., (eds). Advances in the Control of Theileriosis. The Hague, Boston, London: Martinus Nijhoff Publishers.
- BROWN, C.G.D., 1983. Theileria parva. In: JENSEN, J.B., (ed.). In Vitro Cultivation of Protozoan Parasites. Boca Raton, Florida: CRC Press Inc.
- BROWN, C.G.D., 1985. Immunization against East Coast fever. In: IRVIN, A.D., (ed.). Immunization against Theileriosis in Africa. Nairobi: International Laboratory for Research on Animal Diseases.
- BROWN, C.G.D., CRAWFORD, J.G., KANHAI, G.K., NJUGUNA, L.N. & STAGG, D.A., 1978. Immunisation of cattle against East Coast fever with lymphoblastoid cell lines infected and transformed by Theileria parva. In: WILDE, J.K.H. (ed.). Tick-borne Diseases and their Vectors. Edinburgh: University of Edinburgh, Centre for Tropical Veterinary Medicine.
- BROWN, C.G.D., MALMQUIST, W.A., CUNNINGHAM, M.P., RADLEY, D.E. & BURRIDGE, M.J., 1971. Immunization against East Coast fever. Inoculation of cattle with Theileria parva schizonts grown in cell culture. Journal of Parasitology, 57, No. 4, Section II, 59–60.
- BROWN, C.G.D., STAGG, D.A., PURNELL, R.E., KANHAI, G.K. & PAYNE, R.C., 1973. Infection and transformation of bovine lymphoid cells in vitro by infective particles of Theileria parva. Nature, London, 245, 101–102.
- BRUCE, D.B., HAMERTON, A.E., BATERMAN, H.R. & MACKIE, F.P., 1910. Amakebe: A disease of calves in Uganda. Proceedings of the Royal Society, B, 82, 256–272.
- BURRIDGE, M.J. & KIMBER, C.D., 1972. The indirect fluorescent antibody test for experimental East Coast fever (Theileria parva infection of cattle). Evaluation of a cell culture schizont antigen. Research in Veterinary Science, 13, 451–455.
- BURRIDGE, M.J. & KIMBER, C.D., 1973. Duration of serological response to the indirect fluorescent antibody test of cattle recovered from Theileria parva infection. Research in Veterinary Science, 14, 270–271.
- CHAISI, M.E., JANSSENS, M.E., VERMEIREN, L., OOSTHUIZEN, M.C., COLLINS, N.E. & GEYSEN, D., 2013. Evaluation of a real-time PCR test for the detection and discrimination of Theileria species in the African buffalo (Syncerus caffer). PLoS ONE 8, e75827.
- CHEESEMAN, K & WEITZMAN, J.B., 2015 Host-parasite interactions: an intimate epigenetic relationship. Cellular Microbiology, 17, 1121-1132.
- CHEMA, S., CHUMO, R.S., DOLAN, T.T., GATHUMA, J.M. IRVIN, A.D., JAMES, A.D. & YOUNG, A.S., 1987. Clinical trial of halofuginone lactate for the treatment of East Coast fever in Kenya. The Veterinary Record, 120, 575-577.
- CHEMA, S., WAGHELA, S., JAMES, A.D., DOLAN, T.T., YOUNG, A.S., MASIGA, W.N., IRVAN, A.D., MULELA, G.H.M. & WEKESA, L.S., 1986. Clinical trial of parvaquone for the treatment of East Coast fever in Kenya. The Veterinary Record, 118, 588–589.
- CHEN, P.P., CONRAD, P.A., OLE-MOIYOIi, O.K., BROWN, W.C. & DOLAN, T.T., 1991. DNA probes detect Theileria parva in the salivary glands of Rhipicephalus appendiculatus ticks. Parasitology Research, 77, 590–594.
- CONRAD, P.A., DENHAM, D. & BROWN, C.G.D., 1986. Intraerythrocytic multiplication of Theileria parva in vitro: An ultrastructural study. International Journal of Parasitology, 16, 223–230.
- CONRAD, P., OLE-MOIYOII, O.K., BALDWIN, C.L, DOLAN, T.T., O’CALLAGHAN, C.J., NJAMUNGGEH, R.E.G., GROOTENHUIS, J.G., STAGG, D.A., LEITCH, B.L. & YOUNG, A.S., 1989. Characterisation of buffalo-derived theilerial parasites with monoclonal antibodies and DNA probes. Parasitology, 98, 179–188.
- COWDRY, E.V. & HAM, A.W., 1932. Studies on East Coast fever. 1. The life cycle of the parasite in ticks. Parasitology, 24, 1–49.
- CUNNINGHAM, M.P., BROWN, C.G., BURRIDGE, M.J., MUSOKE, A.J., PURNELL, R.E., RADLEY, D.E. & SEMPEBWA, C., 1974. East Coast fever: titration in cattle of suspensions of Theileria parva derived from ticks. British Veterinary Journal, 130, 336–345.
- CUNNINGHAM, M.P., BROWN, C.G.D., BURRIDGE, M.J., IRVIN, A.D., PURNELL, R.E. & RADLEY, D.E., 1973. Theileria parva: Comparative infectivity of a ground tick stabilate and a classical 10-tick challenge. Research in Veterinary Science, 15, 263–265.
- CUNNINGHAM, M.P., BROWN, C.G.D., BURRIDGE, M.J. & PURNELL, R.E., 1973 Cryopreservation of infective particles of Theileria parva. International Journal of Parasitology, 3, 583–587.
- DE DEKEN, R., MARTIN, V., SAIDO, A., MADDER, M., BRANDT, J. & GEYSEN, D., 2007. An outbreak of East Coast fever on the Comoros: a consequence of the import of immunised cattle from Tanzania? Veterinary Parasitology, 143, 245-253.
- DE MARTINI, J.C. & MOULTON, J.E., 1973. Responses of the bovine lymphatic system to infection by Theileria parva. 1. Histology and ultrastructure of lymph nodes in experimentally-infected calves.Journal of Comparative Pathology, 83, 281–298.
- DIESEL, A.M., 1948. The campaign against East Coast fever in South Africa. Onderstepoort Journal of Veterinary Science and Animal Industry, 23, 19–31.
- DI GIULIO, G., LYNEN, G., MORZARIA, S., OURA, C. & BISHOP, R., 2009. Live immunization against East Coast fever - current status. Trends in Parasitology, 25, 85-92.
- DOLAN, T.T., YOUNG, A.S., LOSOS, G.J., MCMILIAN, I., MINDER, C.E. & SOULSBY, K., 1984. Dose dependent responses of cattle to Theileria parva stabilate. International Journal of Parasitology, 14, 89-95.
- DOLAN, T.T., 1981. Progress in the chemotherapy of theileriosis. In: IRVIN, A.D., CUNNINGHAM, M.P. & YOUNG, A.S., (eds). Advances in the Control of Theileriosis. The Hague, Boston, London: Martinus Nijhoff Publishers.
- DOLAN, T.T., 1986. Chemotherapy of East Coast fever: The long-term weight changes, carrier state and disease manifestations of parvaquone-treated cattle. Journal of Comparative Pathology, 96, 137–146.
- DOLAN, T.T., INJAIRU, R., GISEMBA, F., MAINA, J.N., MBADI, G., MBWIRIA, S.K., MULELA, G.H.M. & OTHIENO, D.A.O., 1992. A clinical trial of buparvaquone in the treatment of East Coast fever. The Veterinary Record, 130, 536–538.
- DOLAN, T.T., NJUGUNA, L.N. & STAGG, D.A., 1982. The response of Bos taurus and Bos indicus cattle types to inoculation of lymphoblastoid cell lines infected with Theileria parva schizonts. Tropical Medical Parasitology, 33, 57–62.
- DOLAN, T.T. & YOUNG, A.S., 1981. An approach to the economic assessment of East Coast fever in Kenya. In: IRVIN, A.D., CUNNINGHAM, M.P. & YOUNG, A.S., (eds). Advances in the Control of Theileriosis. The Hague, Boston, London: Martinus Nijhoff Publishers.
- DU TOIT, P.J., 1931. Immunity in East Coast fever. Seventeenth Report of the Director of Veterinary Services and Animal Industry, Union of South Africa, 3–25.
- DU TOIT, P.J. & VILJOEN, P.R., 1929. Dipping as a method of eradicating East Coast fever. Fifteenth Annual Report of the Director of Veterinary Services, Union of South Africa, 33–66.
- EMERY, D.L., EUGUI, E.M., NELSON, R.T. & TENYWA, T., 1981. Cell-mediated immune responses to Theileria parva (East Coast fever) during immunization and lethal infections in cattle. Immunology, 43, 323–336.
- FAWCETT, D.W., BUSCHER, G & DOXSEY, S., 1982. Salivary gland of the tick vector of East Coast fever. IV. Cell type selectivity and host cell responses to Theileria parva. Tissue and Cell, 14, 397-414.
- FRY, L.M., SCHNEIDER, D.A., FREVERT, C.W., NELSON, D.D., MORRISON, W.I. & KNOWLES, D.P., 2016. East coast fever caused by Theileria parva is characterized by macrophage activation associated with vasculitis and respiratory failure. https://doi.org/10.1371/journal.pone.0156004.
- GACHOHI, J., SKILTON, R., HANSEN, F., NGUMI, P. & KITALA, P., 2012. Epidemiology of east coast fever (Theileria parva infection) in Kenya: past, present and the future. Parasites & Vectors, 5, 194.
- GITAU, G.K., PERRY, B.D., KATENDA, J.M., MCDERMOTT, J.J., MORZARIA, S.P. & YOUNG, A.S., 1997. The prevalence of tick-borne infections in smallholder dairy farms in Murang’a District, Kenya: a cross-sectional study. Preventive Veterinary Medicine, 30, 95–107.
- GODDEERIS, B.M., KATENDA, J.M., IRVIN, A.D. & CHUMO, R.S.C., 1982. The indirect fluorescent antibody test for experimental and epizootiological studies of East Coast fever (Theileria parva infection in cattle). Evaluation of a cell culture schizont antigen fixed and stored in suspension. Research in Veterinary Science, 33, 360–365.
- GRAY, C.E. & ROBERTSON, W., 1902. Report upon Texas Fever or Redwater in Rhodesia. Cape Town: Argus Printing & Publishing Company Ltd.
- HEMMINK, J.D., WEIR, W., MACHUGH, N.D., GRAHAM, S.P., PATEL, E., PAXTON, E., SHIELS, B., TOYE, P.G., MORRISON, W.I. & PELLE, R., 2016. Limited genetic and antigenic diversity within parasite isolates used in a live vaccine against Theileria parva. International Journal of Parasitology, 46, 495-506.
- HENNING, M.W., 1956. Animal Diseases in South Africa. 3rd edn. Pretoria, South Africa: Central News Agency Ltd.
- HOUSTON, E.F., TARACHA, E.L., BRACKENBURY, L., MACHUGH, N.D., MCKEEVER, D.J., CHARLESTON, B. & MORRISON, W.I., 2008. Infection of cattle with Theileria parva induces an early CD8 T cell response lacking appropriate effector function. International Journal Parasitology, 38, 1693-1704.
- HULLIGER, L., WILDE, J.K.H., BROWN, C.G.D. & TURNER, L., 1964. Mode of multiplication of Theileria in cultures of bovine lymphocytic cells. Nature, London, 203, 728–730.
- IRVIN, A.D., 1987. Characterization of species and strains of Theileria. Advances in Parasitology, 26, 145–197.
- JARRETT, W.F.H., CRIGHTON, G.W. & PIRIE, H.M., 1969. Theileria parva: Kinetics of replication. Experimental Parasitology, 24, 9–25.
- KABI, F., MASEMBE, C., MUWANIKA, V., KIRUNDA, H. & NEGRINI, R., 2014. Geographic distribution of non-clinical Theileria parva infection among indigenous cattle populations in contrasting agro-ecological zones of Uganda: Implications for control strategies. Parasites & Vectors, 7, 414.
- KAIRO, A., FAIRLAMB, A.H., GOBRIGHT, E. & NENE, V., 1994. A 7.1 KB linear DNA molecule of T. parva has scrambled rDNA sequences and open reading frames for mitochondrially encoded proteins. EMBO Journal, 13, 898–905.
- KARIUKI, D.P., YOUNG, A.S., MORZARIA, S.P., LESAN, A.C., MINING, S.K., OMWOYO, P., WAFULA, J.L.M. & MOLYNEUX, D.H., 1995. Theileria parva carrier state in naturally infected and artificially immunized cattle. Tropical Animal Health and Production, 27, 15–25.
- KATENDE, J.M., GODDEERIS, B.M., MORZARIA, S.P., NKONGE, C.G. & MUSOKE, A.J., 1990. Identification of a Theileria mutans-specific antigen for use in an antibody and antigen detection ELISA. Parasite Immunology, 12, 419–433.
- KOCH, R., 1898. Reiseberichte u¨ber Rinderpest, Bubonenpest in Indien und Afrika, Tsetse—oder Surrakrankheit, Texasfieber, tropische Malaria, Schwarzwasserfieber. Berlin: J. Springer.
- KOCH, R., 1903. Interim report on Rhodesian redwater or ‘African Coast fever’. Journal of Comparative Pathology and Therapeutics, 16, 273–280.
- LAMBELIN, G., ECTORS, F., VAN VAERENBERGH, R. & MAMMERICKX, M., 1960. Sensibilité du buffle d’Asie aux principales maladies à protozoaires du bétail au Congo belge. Essais expérimentaux et observations cliniques. Annales de la Société Belge de Médécine Tropicale, 40, 189–197.
- LAWRENCE, J.A., 1992. History of bovine theileriosis in southern Africa. In: NORVAL, R.A.I., PERRY, B.D. & YOUNG, A.S. (eds). The Epidemiology of Theileriosis in Africa. London, Academic Press.
- LAWRENCE, J.A., NORVAL, R.A.I. & UILENBERG, G., 1983. Rhipicephalus zambeziensis as a vector of bovine theileriae. Tropical Animal Health and Production, 15, 39–42.
- LAWRENCE, J.A., TJORNEHOJ, K. & KAMWENDO, S.P., 1995. The consequences of interruption of intensive dipping of cattle. In: COONS, L. & ROTHSCHILD, M. (eds) The Second International Conference on Tick-borne Pathogens at the Host-Vector Interface: A Global Perspective. Kruger National Park, South Africa, August 28 to September 1, 1995. Proceedings and Abstracts, pp 455-459.
- LEITCH, B.L. & YOUNG, A.S., 1981. Theileria infections in Rhipicephalus appendiculatus ticks collected in the field. In: IRVIN, A.D., CUNNINGHAM, M.P. & YOUNG, A.S., (eds). Advances in the Control of Theileriosis. The Hague, Boston, London: Martinus Nijhoff Publishers.
- LESSARD, P., L’EPLATTENIER, R., NORVAL, R.A.I., KUNDERT, K., DOLAN, T.T., CROZE, H., WALKER, J.B., IRVAN, A.D. & PERRY, B.D., 1990. Geographical information systems for studying the epidemiology of cattle diseases caused by Theileria parva. The Veterinary Record, 126, 255–262.
- LICHTENHELD, G., 1911. Preliminary communication on the fixing of complement in horsesickness and East Coast fever. Report of the Government Veterinary Bacteriologist, Transvaal, 1909–10.
- LOUNSBURY, C.P., 1904. Transmission of African Coast fever. Agricultural Journal, Cape of Good Hope, 24, 428–432.
- MALMQUIST, W.A., NYINDO, M.B.A. & BROWN, C.G.D., 1970. East Coast fever: Cultivation in vitro of bovine spleen cell lines infected and transformed by Theileria parva. Tropical Animal Health and Production, 2, 139–145.
- MANGANI, M., 1999. ECF Immunisation in Zambia. In: MORZARIA, S. & WILLIAMSON. S. (eds). Live vaccines for Theileria parva: Deployment in Eastern, Central and Southern Africa. Nairobi, Kenya: International Livestock Research Institute.
- MANS, B.J., PIENAAR, R. & LATIF, A.A., 2015. A review of Theileria diagnostics and epidemiology. International Journal for Parasitology Parasites and Wildlife, 4, 104-118.
- MANS, B.J., PIENAAR, R., LATIF, A.A. & POTGIETER, F.T., 2011. Diversity in the 18S SSU rRNA V4 hyper-variable region of Theileria in bovines and African buffalo (Syncerus caffer) from Southern Africa. Parasitology, 138, 766-779.
- MBONGO, S., 1999. The infection and treatment method of immunisation: practice and problems. In: MORZARIA, S. & WILLIAMSON, S. (eds). Live vaccines for Theileria parva: Deployment in Eastern, Central and Southern Africa. Nairobi, Kenya: International Livestock Research Institute.
- MCHARDY, N., 1989. Buparvaquone, the new antitheilerial: a review of its efficacy and safety. In: DOLAN, T.T. (ed.). Theileriosis in Eastern, Central and Southern Africa. Nairobi, Kenya: International Laboratory for Research on Animal Diseases.
- MCHARDY, N., HAIGH, A.J.B. & DOLAN, T.T., 1976. Chemotherapy of Theileria parva infection. Nature, London, 261, 698–699.
- MCHARDY, N., HUDSON, A.T., MORGAN, D.W.T., RAE, D.G. & DOLAN, T.T., 1983. Activity of 10 naphthoquinones, including parvaquone (993C) and menoctone, in cattle artificially infected with Theileria parva. Research in Veterinary Science, 35, 347–352.
- MCHARDY, N. & WEKESA, L.S., 1985. Buparvaquone (BW 720C). A new antitheilerial naphthoquinone: Its role in the therapy and prophylaxis of theileriosis. In: IRVIN, A.D., (ed.). Immunization Against Theileriosis in Africa. Nairobi: International Laboratory for Research on Animal Diseases.
- MCHARDY, N., WEKESA, L.S., HUDSON, A.T. & RANDALL, A.W., 1985. Antitheilerial activity of BW-720C buparvaquone. A comparison with parvaquone. Research in Veterinary Science, 39, 29–33.
- MCKEEVER, D.J., 2007. Live immunisation against Theileria parva: containing or spreading the disease? Trends in Parasitology, 25, 269-276.
- MCKEEVER, D.J., 2009. Bovine immunity – a driver for diversity in Theileria parasites? Trends in Parasitology, 23, 565-568.
- MEHLHORN, H. & SCHEIN, E., 1984. The piroplasms: Life cycle and sexual stages. Advances in Parasitology, 23, 37–103.
- MELROSE, T.R. & BROWN, C.G.D., 1979. Isoenzyme variation in piroplasms isolated from bovine blood infected with Theileria annulata and T. parva. Research in Veterinary Science, 27, 379–381.
- METTAM, R.W.M. & CARMICHAEL, J., 1936. Turning sickness, a protozoan encephalitis of cattle in Uganda: its relationship with East Coast fever. Parasitology, 28, 254–283.
- MINAMI, T., SPOONER, P.R., IRVIN, A.D., OCAMA, J.G.R., DOBBELAERE, D.A.E. & FUJINAGA, T., 1983. Characterisation of stocks of Theileria parva by monoclonal antibody profiles. Research in Veterinary Science, 35, 334–340.
- MOHAN, R.N., 1968. Diseases and parasites of buffaloes. Part III. Parasitic and miscellaneous diseases. Veterinary Bulletin, 38, 735–756.
- MOLL, G., AGAN, L. & LOHDING, A., 1985. Bovine cerebral theileriosis in pure Boran and Sahiwal-cross cattle immunized against East Coast fever and kept under continuous field challenge. In: IRVIN, A.D., (ed.). Immunization Against Theileriosis in Africa. Nairobi: International Laboratory for Research on Animal Diseases.
- MOLL, G., LOHDING, A. & YOUNG, A.S., 1984. Epidemiology of theileriosis in the Trans-Mara Division, Kenya. Husbandry and disease background and preliminary investigations on theilerioses in calves. Preventive Veterinary Medicine, 2, 801–832.
- MOLL, G., LOHDING, A., YOUNG, A.S. & LEITCH, B.L., 1986. Epidemiology of theileriosis in calves in an endemic area of Kenya. Veterinary Parasitology, 19, 255–274.
- MORRISON, W.I., 2009. Progress towards understanding the immunobiology of Theileria parasites. Parasitology, 136, 1415-1426.
- MORRISON, W.I., BUSCHER, G., EMERY, D.I., NELSON, R.T. & MURRAY, M., 1981. The kinetics of infection with Theileria parva in cattle and the relevance to the development of immunity. In: IRVIN, A.D., CUNNINGHAM, M.P. & YOUNG, A.S., (eds). Advances in the Control of Theileriosis. The Hague, Boston, London: Martinus Nijhoff Publishers.
- MORRISON, W.I., BUSCHER, G., MURRAY, M., EMERY, D.I., MASAKE, R.A., COOK, R.H. & WELLS, P.W., 1981. Theileria parva: Kinetics of infection in the lymphoid system of cattle. Experimental Parasitology, 52, 248–260.
- MORRISON, W.I., MACHUGH, N.D. & LALOR, P.A., 1996. Pathogenicity of Theileria parva is influenced by the host cell type infected by the parasite. Infection and Immunity, 64, 557–562.
- MORZARIA, S.P IRVAN, A.D., TARACHA, E., SPOONER, P.R., VOIGT, W.P., FUJINAGA, T. & KATENDA, J., 1987. Immunization against East Coast fever: The use of selected stocks of Theileria parva for immunization of cattle exposed to field challenge. Veterinary Parasitology, 23, 23–41.
- MORZARIA, S., NENE, V., MUSOKE, A.J. & BISHOP, R., 1993. Modern biotechnological methods for diagnosis of bovine theileriosis and future trends. Proceedings of the FAO Expert Consultation on the Use of Applicable Biotechnological Methods for Diagnosis of Haemoparasites. October 1993, Merida, Mexico. Rome: Food and Agriculture Organisation of the United Nations.
- MORZARIA, S. & WILLIAMSON, S., (eds)., 1997. Live vaccines for Theileria parva: Deployment in Eastern, Central and Southern Africa. Nairobi, Kenya: International Livestock Research Institute.
- MUSOKE, A.J., ROWLANDS, J., NENE, V., NYANJUI, J., KATENDE, J., SPOONER, P., MWAURA, S., ODONGO, D., NKONGE, C., MBOGO, S., BISHOP, R. & MORZARIA, S., 2005. Subunit vaccine based on the p67 major surface protein of Theileria parva sporozoites reduces severity of infection derived from field tick challenge. Vaccine, 23, 3084-3095.
- MUKHEBI, A.W., PERRY, B.D. & KRUSKA, R., 1992. Estimated economics of theileriosis control in East Africa. Preventive Veterinary Medicine, 12, 73–85.
- MUSISI, F.L., MORGAN, D.W.T. & SCHELS, H.F., 1985. Treatment of theileriosis with parvaquone in Zambia. The Veterinary Record, 117, 338–339.
- MUSOKE, A., MORZARIA, S., NKONGE, C., JONES, E. & NENE, V., 1992. A recombinant sporozoite surface antigen of Theileria parva induces protection in cattle. Proceedings of the National Academy of Science USA, 89, 514–518.
- MUSOKE, A.J., NANTULYA, V.M., BUSCHER, G., MASAKE, R.A. & OTIM, B., 1982. Bovine immune response to Theileria parva: Neutralizing antibodies to sporozoites. Immunology, 45, 663–668.
- MUSOKE, A.J., NANTULYA, V.M., RURANGIRWA, F.R. & BUSCHER, G., 1984. Evidence for a common protective antigenic determinant on sporozoites of several Theileria parva strains. Immunology, 52, 231–238.
- MUSOKE, A., NENE, V. & MORZARIA, S.P., 1993. A sporozoite-based vaccine for Theileria parva. Parasitology Today, 9, 385–388.
- MUSOKE, A.J., PALMER, G.H., MCELWAIN, T.F., NENE, V. & MCKEEVER, D., 1996. Prospects for subunit vaccines against tick-borne diseases. British Veterinary Journal, 152, 621–639.
- MUSOKE, A.J., ROWLANDS, J., NENE, V., NYANJUI, J., KATENDE, J., SPOONER, P., MWAURA, S.,ODONGO, D., NKONGE, C., MBOGO, S., BISHOP, R. & MORZARIA, S., 2005. Subunit vaccine based on the p67 major surface protein of Theileria parva sporozoites reduces severity of infection derived from field tick challenge. Vaccine, 23, 3084–3095.
- MUTUGI, J.J., LAMPARD, D., YOUNG, A.S., NDUNGU, S.G., LINYONI, A., MARITIM, A.C., MINING, S.K., NGUMI, P.N., KARIUKI, D.P., WILLIAMSON, S.M., AWICH, J.R. & LESAN, A.C., 1990. Recent immunisation trials against T. parva parva infections in Kenya. In: YOUNG, A.S., MUTUGI, J.J. & MARITIM, A.C., (eds). Progress towards the Control of East Coast Fever (Theileriosis) in Kenya. Nairobi, Kenya: Kenya Agricultural Research Institute.
- NDUNGU, S.G., BROWN, C.G.D. & DOLAN, T.T., 2005a. In vivo comparison of susceptibility between Bos indicus and Bos taurus cattle types to Theileria parva infection. Onderstepoort Journal of Veterinary Research, 72, 13-22.
- NDUNGU, S.G., NGUMI, P.N., MBOGO, S.K., DOLAN, T.T., MUTUGI, J.J. & YOUNG, A.S., 2005b. Some preliminary observations on the susceptibility and resistance of different cattle breeds to Theileria parva infection. Onderstepoort Journal of Veterinary Research, 72, 7-11.
- NEITZ, W.O., 1953. Aureomycin in Theileria parva infection. Nature, London, 171, 34–35.
- NEITZ, W.O., 1957. Theileriosis, gonderioses and cytauxzoonoses: A review. Onderstepoort Journal of Veterinary Research, 27, 275–430.
- NENE, V., KIARA, H., LACASTA, A., PELLE, R., SVITEK, N. & STEINAA, L., 2016. The biology of Theileria parva and control of east coast fever - Current status and future trends. Ticks and Tick-Borne Diseases, 7, 549-564.
- NENE, V., MUSOKE, A., GOBRIGHT, E., MORZARIA, S., 1996. Conservation of the sporozoite p67 vaccine antigen in cattle-derived Theileria parva stocks with different cross-immunity profiles. Infection and Immunity, 64, 2056-2061.
- NEWSON, R.M., CHIERA, J.W., YOUNG, A.S., DOLAN, T.T., CUNNINGHUM, M.P. & RADLEY, D.E., 1984. Survival of Rhipicephalus appendiculatus (Acarina: Ixodidae) and persistence of Theileria parva (Apicomplexa: Theileriidae) in the field. International Journal of Parasitology, 14, 483–489.
- NGUMI, P.N., YOUNG, A.S., LAMPARD, D., MINING, S.K., NDUNGU, S.G., LESAN, A.C., WILLIAMSON, S.M., LINYONI, A. & KARIUKI, D.P., 1992. Further evaluation of the use of buparvaquone in the infection and treatment method of immunising cattle against Theileria parva derived from African buffalo (Syncerus caffer). Veterinary Parasitology, 43, 15–24.
- NORLING, M., BISHOP, R.P., PELLE, R., QI, W., HENSON, S., DRABEK, E.F., TRETINA, K., ODONGO, D., MWAURA, S., NJOROGE, T., BONGCAM-RUDLOFF, E., DAUBENBERGER, C.A. & SILVA, J.C., 2015. The genomes of three stocks comprising the most widely utilized live sporozoite Theileria parva vaccine exhibit very different degrees and patterns of sequence divergence. BMC Genomics, 16, 729.
- NORVAL, R.A.I., 1977. Tick problems in relation to land utilization in Rhodesia. Rhodesian Veterinary Journal, 8, 33–38.
- NORVAL, R.A.I., 1978. The effects of partial breakdown of dipping in African areas in Rhodesia. Rhodesian Veterinary Journal, 9, 9–16.
- NORVAL, R.A.I. & PERRY, B.D., 1990. The introduction, spread and subsequent disappearance of the brown ear tick (Rhipicephalus appendiculatus) from the Southern lowveld of Zimbabwe. Experimental and Applied Acarology, 9, 103–111.
- NORVAL, R.A.I., PERRY, B.D., GEBREAB, F. & LESSARD, P., 1991. East Coast fever: a problem of the future for the Horn of Africa? Preventive Veterinary Medicine, 10, 163–172.
- NORVAL, R.A.I., PERRY, B.D. & YOUNG, A.S. (eds.), 1992. The Epidemiology of Theileriosis in Africa. London: Academic Press.
- NUTTALL, G.H.F. & HINDLE, E., 1913. Conditions influencing the transmission of East Coast fever. Parasitology, 2, 208–210.
- O.I.E., 2014 Manual of Diagnostic Tests and Vaccines for Terrestrial Animals, Vol. 1-2, 7th Edition, Office International des Epizooties, Paris.
- OURA, C.A., TAIT, A., ASIIMWE, B., LUBEGA, G.W., WEIR, W., 2011a. Haemoparasite prevalence and Theileria parva strain diversity in Cape buffalo (Syncerus caffer) in Uganda. Veterinary Parasitology, 175, 212-219.
- OURA, C.A., TAIT, A., ASIIMWE, B., LUBEGA, G.W., WEIR, W., 2011b. Theileria parva genetic diversity and haemoparasite prevalence in cattle and wildlife in and around lake Mburo National Park in Uganda. Parasitology Research, 108, 1365-1374.
- PATEL, E.H., LUBEMBE, D.M., GACHANJA, J., MWAURA, S., SPOONER, P., & TOYE, P., 2011.Molecular characterization of live Theileria parva sporozoite vaccine stabilates reveals extensive genotypic diversity. Veterinary Parasitology, 179, 62–68.
- PATEL, E., MWAURA, S., KIARA, H., MORZARIA, S., PETERS, A., TOYE, P., 2016. Production and dose determination of the Infection and Treatment Method (ITM) Muguga cocktail vaccine used to control East Coast fever in cattle. Ticks and Tick Borne Diseases, 7, 306-314.
- PERRY, B.D. & YOUNG, A.S., 1993. The naming game: the changing fortunes of East Coast fever and Theileria parva. The Veterinary Record, 133, 613–616.
- PERRY, B.D. & YOUNG, A.S., 1995. The past and future roles of epidemiology and economics in the control of tick-borne diseases of livestock in Africa: the case of theileriosis. Preventive Veterinary Medicine, 25, 107–120.
- PERRY, B.D., KRUSKA, R., LESSARD, P., NORVAL, R.A.I. & KUNDERT, K., 1991. Estimating the distribution and abundance of Rhipicephalus appendiculatus in Africa. Preventive Veterinary Medicine, 11, 261–268.
- PERRY, B.D., LESSARD, P., NORVAL, R.A.I., KUNDERT, K. & KRUSKA, R., 1990. Climate, vegetation and the distribution of Rhipicephalus appendiculatus in Africa. Parasitology Today, 6, 100–104.
- PETERS, A.R., 2009. Registration and commercialisation of the East Coast fever Muguga cocktail vaccine (IAEA-CN--174). International Atomic Energy Agency (IAEA).
- PIENAAR, R., POTGIETER, F.T., LATIF, A.A., THEKISOE, O.M.M. & MANS, B.J., 2011. Mixed Theileria infections in free-ranging buffalo herds: implications for diagnosing Theileria parva infections in Cape buffalo (Syncerus caffer). Parasitology, 138, 884–895.
- PINDER, M. & HEWETT, R.S., 1980. Monoclonal antibodies detect antigenic diversity in Theileria parva parasites. Journal of Immunology, 124, 1000–1001.
- PURNELL, R.E., BOARER, C.D.H. & PIERCE, M.A., 1971. Theileria parva: Comparative infection rates of adult and nymphal Rhipicephalus appendiculatus. Parasitology, 62, 349–353.
- PURNELL, R.E., IRVAN, A.D., KIMBER, C.D., OMWOYO, P.L. & PAYNE, R.C., 1974. East Coast fever: Further laboratory investigations on the use of rabbits as vehicles for infecting ticks with theilerial piroplasms. Tropical Animal Health and Production, 6, 145–151.
- PURNELL, R.E. & JOYNER, L.P., 1968. The development of Theileria parva in the salivary glands of the tick, Rhipicephalus appendiculatus. Parasitology, 58, 725–732.
- RADLEY, D.E., 1978. Chemoprophylactic immunization against East Coast fever. In: WILDE, J.K.H., (ed.). Tick-borne Diseases and their Vectors. Edinburgh: University of Edinburgh, Centre for Tropical Veterinary Medicine.
- RADLEY, D.E., 1981. Infection and treatment immunisation against theileriosis. In: IRVIN, A.D., CUNNINGHAM, M.P. & YOUNG, A.S. (eds). Advances in the Control of Theileriosis. The Hague, Boston, London: Martinus Nijhoff Publishers.
- RADLEY, D.E., 1985. East Coast fever immunization—field trials in Malawi. In: IRVIN, A.D., (ed.). Immunization Against Theileriosis in Africa. Nairobi: International Laboratory for Research on Animal Diseases.
- SCHETTERS, T.P., ARTS, G., NIESSEN, R., VAN BINSBERGEN, R., SANDERS, L., MOLS-VORSTEMANS, T., STRYDOM, T., GEYSEN, D. & SCHAAP, D., 2014. A recombinant p67 vaccine against Theileria parva in cattle: Conflicting results from experimental and field challenges. ICOPA XIII, Mexico.
- SCHREUDER, B.E.C. & UILENBERG, G., 1976. Studies on Theileriidae (Sporozoa) in Tanzania. V. Preliminary experiments on a new method for infecting ticks with Theileria parva and Theileria mutans. Tropenmedizin und Parasitologie, 27, 422–426.
- SHAW, M.K., 2003. Cell invasion by Theileria sporozoites. Trends in Parasitology, 19, 2-6.
- SHORT, N.J. & NORVAL, R.A.I., 1981. Regulation of seasonal occurrence in the tick Rhipicephalus appendiculatus Neumann, 1901. Tropical Animal Health and Production, 13, 19–26.
- SPOONER, R.L., PENHALE, W.J., BURRIDGE, M.J. & BROWN, C.G.D., 1973. Some serum globulin changes in East Coast fever. Research in Veterinary Science, 15, 368–374.
- SPREULL, J., 1914. East Coast fever inoculation in the Transkeian Territories, South Africa. Journal of Comparative Pathology and Therapeutics, 27, 299–304.
- STAGG, D.D., 1992. Studies of Theilerial Parasites of Waterbuck (Kobus defassa) and their Possible Role in the Epidemiology of East Coast Fever. Ph.D thesis, University of Salford, UK.
- STAGG, D.A., DOLAN, T.T., LEITCH, B.L. & YOUNG, A.S., 1981. The initial stages of infection of cattle cells with Theileria parva sporozoites in vitro. Parasitology, 83, 191–197.
- STECK, W., 1928. Histological studies on East Coast fever. Thirteenth and Fourteenth Reports of the Director of Veterinary Education and Research, South Africa, 243–282.
- SUTHERST, R.W. & MAYWALD, G.F., 1985. A computerized system for matching climates in ecology. Agriculture, Ecosystems and Environment, 13, 281–299.
- TATCHELL, R.J. & EASTON, E., 1986. Tick (Acari: Ixodidae) ecological studies in Tanzania. Bulletin of Entomological Research, 76, 229–246.
- THEILER, A., 1904. East Coast fever. Transvaal Agricultural Journal, 2, 421–438.
- THEILER, A., 1905. Report of the Government Veterinary Bacteriologist, Transvaal, 1903–04.
- THEILER, A., 1911. The artificial transmission of East Coast fever. Report of the Government Veterinary Bacteriologist, Transvaal, 1909–10, 7–55.
- THEILER, A., 1911. Progress report on the possibility of vaccinating cattle against East Coast fever. First Report of the Director of Veterinary Research, South Africa, 47–207.
- THEILER, A. & DU TOIT, P.J., 1928. The transmission of tick-borne diseases by the intrajugular injection of the emulsified intermediary host itself. Thirteenth and Fourteenth Reports of the Director of Veterinary Education and Research, Union of South Africa, Part 1, 17–44.
- THEILER, A. & DU TOIT, P.J., 1929. The transmission of East Coast fever by means of blood. Fifteenth Annual Report of the Director of Veterinary Services, Union of South Africa, 15–31.
- TINDIH, H.S., GEYSEN, D., GODDEERIS, B.M., AWINO, E., DOBBELAERE, D.A.E. & NAESSENS, J., 2012. Theileria parva isolate of low virulence infects a subpopulation of lymphocytes. Infection and Immunity, 80, 1267–1273.
- TINDIH, H.S., MARCOTTY, T., NAESSENS, J., GODDEERIS, B.M. & GEYSEN, D., 2010. Demonstration of differences in virulence between two Theileria parva isolates. Veterinary Parasitology, 168, 223-230.
- TRETINA, K., GOTIA, H.T., MANN, D.J., & SILVA, J.C., 2015. Theileria-transformed bovine leukocytes have cancer hallmarks. Trends in Parasitology, 31, 306-314.
- UILENBERG, G., 1976. Tick-borne livestock diseases and their vectors. 2. Epizootiology of tick-borne diseases. World Animal Review, 17, 8–15.
- UILENBERG, G., 1985. Possible impact of other tick-borne diseases following East Coast fever immunization. In: IRVIN, A.D., (ed.). Immunization against Theileriosis in Africa. Nairobi: International Laboratory for Research on Animal Diseases.
- UILENBERG, G., SCHREUDER, B.E.C., MPANGALA, C., SILAYO, R.S., TONDEUR, W., TATCHELL, R.J. & SANGA, H.J.N., 1978. Immunization against East Coast fever. In: WILDE, J.K.H., (ed.). Tick-borne Diseases and their Vectors. Edinburgh: University of Edinburgh, Centre for Tropical Veterinary Medicine.
- UILENBERG, G. & ZWART, D., 1979. Skin nodules in East Coast fever. Research in Veterinary Science, 26, 243–245.
- WAGNER, G.G., JESSETT, D.M., BROWN, C.G.D. & RADLEY, D.E., 1975. Diminished antibody response to rinderpest vaccination in cattle undergoing experimental East Coast fever. Research in Veterinary Science, 19, 209–211.
- WALKER, A.R., MCKELLAR, S.B., BELL, L.J. & BROWN, C.G.D., 1979. Rapid quantitative assessment of Theileria infection in ticks. Tropical Animal Health and Production, 11, 21–26.
- WALKER, J.G., KLEIN, E.Y. & LEVIN, S.A., 2014. Disease at the wildlife-livestock interface: Acaricide use on domestic cattle does not prevent transmission of a tick-borne pathogen with multiple hosts. Veterinary Parasitology, 199, 206-214.
- WATKINS-PITCHFORD, H. (n.d., c. 1911). An Illustrated Pamphlet on Tick-destruction and the Eradication of East Coast Fever and other S. African Diseases by Dipping. Natal: P. Davis & Sons.
- WATT, D., SPARAGANO, O., BROWN, C.G.D. & WALKER, A.R., 1997. Use of the polymerase chain reaction for identification and quantification of Theileria parva protozoa in Rhipicehalus appendiculatus ticks. Parasitology Research, 83, 359–363.
- WILDE, J.K.H., 1966. Changes in bovine bone marrow during the course of East Coast fever. Research in Veterinary Science, 7, 213–224.
- WILKE, G.M., BROWN, C.G., KIRVAR, B.E., THOMAS, M., WILLIAMSON, S.M., BELL-SAKYI, L.J. & SPARAGANO, O., 1998. Chemoprophylaxis of Theileria annulata and Theileria parva infections of calves with buparvaquone. Veterinary Parasitology, 78, 1–12.
- WILSON, S.G., 1945. Some factors affecting the incidence of East Coast fever in Northern Province, Nyasaland. Journal of the South African Veterinary Medical Association, 16, 47–52.
- WOOLHOUSE, M.E.J., THUMBI, S.M., JENNINGS, A., CHASE-TOPPING, M., CALLABY, R., KIARA, H., OOSTHUIZEN, M.C., MBOLE-KARIUKI, M., CONRADIE, I., HANDEL, I.J., POOLE, E.J., NJIIRI, E., COLLINS, N.E., MURRAY, G.,TAPIO, M., AUGUET, O.T., WEIR, W., MORRISON, W.I., KRUUK, L.E.B., BRONSVOORT, B.M.de C., HANOTTE, O., COETZER, K. & TOYE, P.G., 2015. Co-infections determine patterns of mortality in a population exposed to parasite infection Science Advances 1 (2), e1400026.
- YEOMAN, G.H., 1966. Field vector studies of epizootic East Coast fever. I. A quantitative relationship between R. appendiculatus and the epizooticity of East Coast fever. Bulletin of Epizootic Diseases of Africa, 14, 5–27.
- YEOMAN, G.H., 1966. Field vector studies of epizootic East Coast fever. II. Seasonal studies of R. appendiculatus on bovine and non-bovine hosts in East Coast fever enzootic, epizootic and free areas. Bulletin of Epizootic Diseases of Africa, 14, 113–140.
- YEOMAN, G.H., 1967. Field vector studies of epizootic East Coast fever. III. Pasture ecology in relation to R. appendiculatus infestation rates on cattle. Bulletin of Epizootic Diseases of Africa, 15, 89–113.
- YOUNG, A.S., LEITCH, B.L., NEWSON, R.M. & CUNNINGHAM, M.P., 1986. Maintenance of Theileria parva parva infection in an endemic area of Kenya. Parasitology, 93, 9–16.
- YOUNG, A.S., LEITCH, B.L. & OMWOYO, P.L., 1979. Induction of infective stages of Theileria parva by exposure of host ticks to high temperature. The Veterinary Record, 105, 531–533.