- Infectious Diseases of Livestock
- Part 1
- Toxoplasmosis
- Vectors: Ticks
- Vectors: Tsetse flies
- Vectors: Muscidae
- Vectors: Tabanidae
- Vectors: Culicoides spp.
- Vectors: Mosquitoes
- Classification, epidemiology and control of arthropod-borne viruses
- Special factors affecting the control of livestock diseases in sub-Saharan Africa
- The control of infectious diseases of livestock: Making appropriate decisions in different epidemiological and socioeconomic conditions
- Infectious diseases of animals in sub-Saharan Africa: The wildlife⁄livestock interface
- Vaccination: An approach to the control of infectious diseases
- African animal trypanosomoses
- Dourine
- Trichomonosis
- Amoebic infections
- GENERAL INTRODUCTION: COCCIDIA
- Coccidiosis
- Cryptosporidiosis
- Toxoplasmosis
- Besnoitiosis
- Sarcocystosis
- Balantidiosis
- Leishmaniosis
- Neosporosis
- Equine protozoal myeloencephalitis
- GENERAL INTRODUCTION: BABESIOSES
- Bovine babesiosis
- Equine piroplasmosis
- Porcine babesiosis
- Ovine babesiosis
- GENERAL INTRODUCTION: THEILERIOSES OF CATTLE
- East Coast fever
- Corridor disease
- Zimbabwe theileriosis
- Turning sickness
- Theileria taurotragi infection
- Theileria mutans infection
- Theileria annulata theileriosis
- Theileriosis of sheep and goats
- Theileria buffeli⁄orientalis infection
- Non-pathogenic Theileria species in cattle
- GENERAL INTRODUCTION: RICKETTSIAL, CHLAMYDIAL AND HAEMOTROPIC MYCOPLASMAL DISEASES
- Heartwater
- Lesser known rickettsial infections in animals and humans
- Chlamydiosis
- Q fever
- Eperythrozoonosis
- Bovine Haemobartonellosis
- Potomac horse fever
- GENERAL INTRODUCTION: ANAPLASMOSES
- Bovine anaplasmosis
- Ovine and caprine anaplasmosis
Toxoplasmosis
This content is distributed under the following licence: Attribution-NonCommercial CC BY-NC  View Creative Commons Licence details here
View Creative Commons Licence details here
Toxoplasmosis
Previous Authors: J P DUBEY AND C G STEWART
Current Authors:
J P DUBEY - Senior scientist, M.V.Sc., Ph.D., DSc., Animal Parasitic Diseases Laboratory, U.S. Department of Agriculture, Building 1001, Beltsville Agricultural Research Center, 10300 Baltimore Avenue, Beltsville, Maryland, 20705, United States
B BANGOURA - Assistant Professor, DVM, PhD, DipEVPC, Department of Veterinary Sciences, University of Wyoming, 1174 Snowy Range Road, Laramie, Wyoming, WY 82070, USA
A DAUGSCHIES - Professor, Centre for Infectious Diseases, Institute for Parasitology, Leipzig University, Leipzig, Germany
Introduction
The protozoan parasite Toxoplasma gondii is a common parasite of many animal species and humans and is distributed throughout the world. Although most of the infections of livestock caused by the parasite are subclinical, toxoplasmosis is a major cause of abortion in sheep in several countries.3, 26, 39, 64, 65, 99 It is also known to cause reproductive losses in goats.29, 44, 54 Cattle are susceptible to experimental T. gondii-induced abortion100 although abortion has not been reported in natural infections. In southern Africa there is a high prevalence of serologically positive animals,67 but reports of clinical toxoplasmosis in domestic animals are rare.93, 94 Toxoplasmosis has been reported in ferrets7 and chinchillas.19 As toxoplasmosis can affect virtually every warm-blooded animal it is impossible to summarize information for each animal species in this chapter. The disease in animals has been reviewed by.39, 91
Aetiology and life cycle
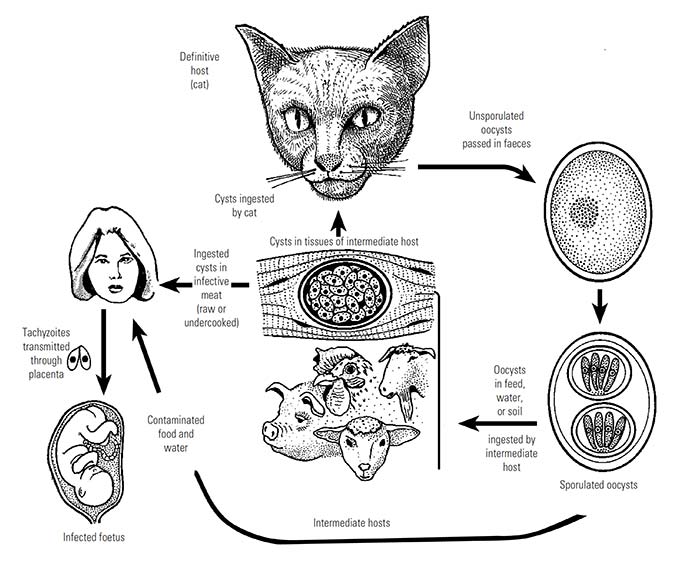
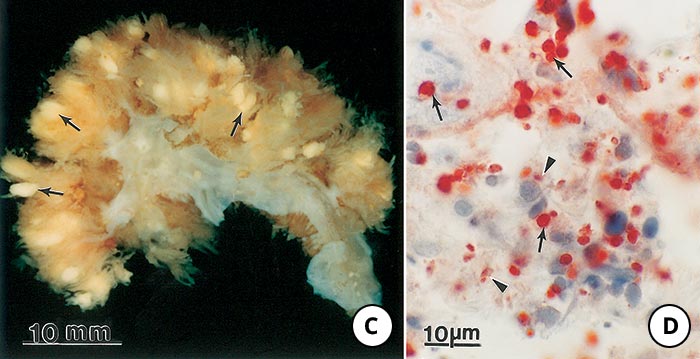
Toxoplasma gondii is a coccidian parasite of domestic cats (Nagel et al. 2013)19 and members of the Felidae, with other warm-blooded animal species, comprising essentially all mammals and birds, as intermediate hosts (Figure 1) It is classified in the phylum Apicomplexa Levine, 1970, class Coccidea Leuckart, 1879, order Eucoccidiorida, and family Sarcocystidae, Poche, 1913. The name Toxoplasma (toxon = arc, plasma = form) is derived from the crescent shape of the tachyzoite stage. There are three infectious stages of T. gondii: the tachyzoites (in groups), the bradyzoites (in tissue cysts), and the sporozoites (in oocysts).39, 58
The tachyzoite is often crescent-shaped and is approximately 2 × 6 μm in size in tissue smears (Figure 2 A) and globular to oval in sections (Figure 2 B). Its anterior (conoidal) end is pointed and its posterior end is round. It has a pellicle (outer covering), a conoid, rhoptries, micronemes, a well-defined nucleus in the central area of the cell (Figure 3) and other organelles found in coccidian merozoites.28, 45
The tachyzoite enters a host cell in a variety of organs and tissues by active penetration of the cell membrane. After entering the cell the tachyzoite becomes surrounded by a parasitophorous vacuole (Figure 3) Numerous intravacuolar tubules connect the parasitophorous vacuolar membrane to the parasite pellicle (Figure 3) The tachyzoite multiplies asexually within the host cell by repeated endodyogeny. Endodyogeny (endo = inside, dyo = two, geny = progeny) is a specialized form of reproduction in which two progeny form within the parent parasite, thereby consuming it (Figure 3) Tachyzoites continue to divide by endodyogeny until the host cell is filled with parasites,45 which is the stage associated with acute toxoplasmosis in animals and humans.
After a few divisions, tachyzoites induce the formation of a cyst wall in a host cell and transform into bradyzoites contained within the wall (Figures 2, 4 and 5). Tissue cysts grow and remain intracellular (Figures 2 and 4) as the bradyzoites (encysted zoites) slowly divide by endodyogeny. Tissue cysts vary in size. Young tissue cysts may be as small as 5 μm in diameter and contain only two to four bradyzoites (Figure 4), while older ones may contain hundreds of crescent-shaped slender organisms (Figures 2 and 5). The tissue cyst wall is elastic, thin (< 0,5 μm) and argyrophilic. Cysts in the brain are round and up to 75 μm in diameter,20 whereas intramuscular cysts are elongated and may be 100 μm long; live tissue cysts in unstained squash preparations vary in size depending on the pressure applied to the coverslip. Although they may develop in visceral organs, including lungs, liver, and kidneys, they are more prevalent in the brain, skeletal muscles and myocardium. Tissue cyst formation can occur as early as one to two weeks after infection and cysts are probably innocuous for their host and can persist for the length of its life. The development of immunity is suspected to contribute significantly to cyst formation, while immunodeficiency may lead to reactivation into dividing tachyzoites.74
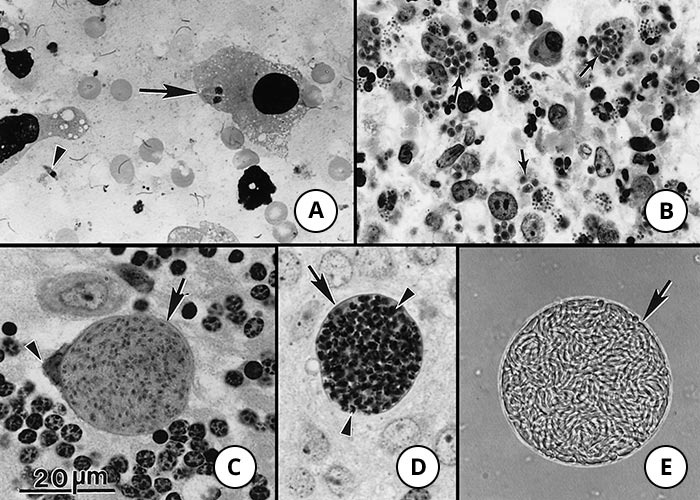
Figure 2 Tachyzoites and tissue cysts of Toxoplasma gondii. Bar = 20 μm and applies to all photographs in the composite figure
- Extracellular single (arrowhead) and paired tachyzoites (arrow) in a macrophage. Impression smear of lung. Giemsa stain
- Tachyzoites in a section of a lymph node. Note numerous oval-shaped tachyzoites (arrows). Haematoxylin and eosin stain
- Intracellular tissue cyst in a neural cell in section of cerebellum. The host cell nucleus is indented (arrowhead). Note thin cyst wall (arrow) enclosing numerous bradyzoites
- Tissue cyst in section of brain. Note darkly stained bradyzoites (arrowheads) and unstained tissue cyst wall (arrow). Periodic acid-Schiff haematoxylin
- Tissue cyst freed from mouse brain. Note thin cyst wall (arrow). Unstained
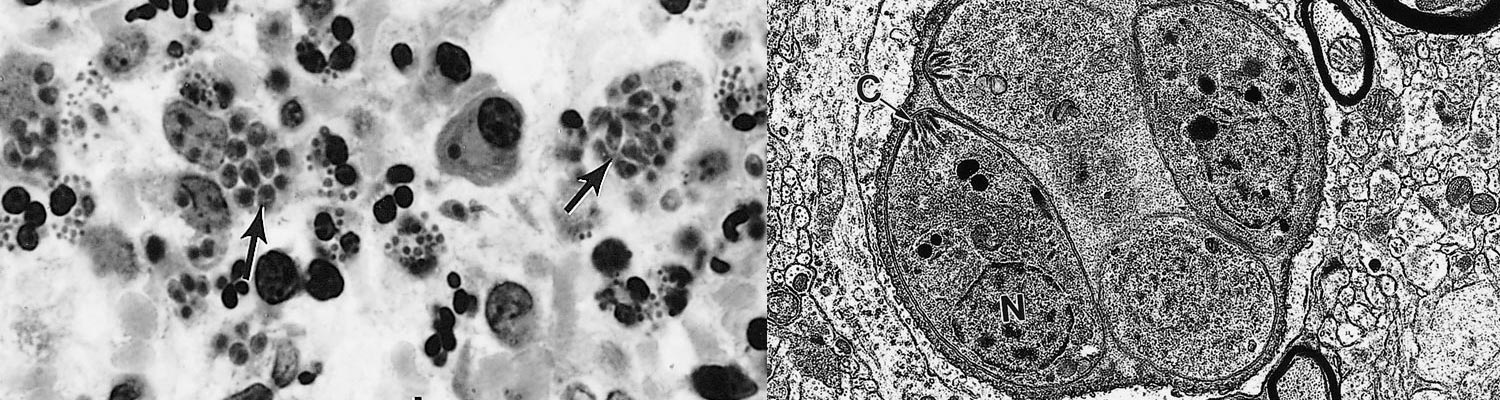
The bradyzoites are approximately 7 × 1.5 μm in size. Bradyzoites differ structurally only slightly from tachyzoites. The former have a nucleus situated towards the posterior end (Figure 5 B) whereas the nucleus in tachyzoites is more centrally located (Figure 5 B) The contents of rhoptries in bradyzoites in older tissue cysts are electron-dense (Figure 3).45 Bradyzoites contain several amylopectin granules that stain red with periodic acid-Schiff (PAS). These particles are absent in tachyzoites.
Cats shed oocysts after ingesting any of the three infectious stages of T. gondii, i.e. tachyzoites, bradyzoites, and sporozoites.22, 23, 43 Prepatent periods (time to the shedding of oocysts after initial infection) and frequency of oocyst shedding vary according to the stage of T. gondii ingested. Prepatent periods are 3 to 10 days after ingesting tissue cysts and generally 19 days or more after ingesting tachyzoites or oocysts.23, 38, 59 Less than 50 per cent of cats shed oocysts after ingesting tachyzoites or oocysts, whereas nearly all cats shed oocysts after ingesting tissue cysts. Oocysts are less infectious for the definitive host (cat) than for non-feline hosts.36, 49
After the ingestion of tissue cysts by cats, the cyst wall is dissolved by the proteolytic enzymes in the stomach and small intestine. The released bradyzoites penetrate the epithelial cells of the small intestine and initiate development of numerous generations of T. gondii.22 Five morphologically distinct types of meronts (A to E) of T. gondii develop in intestinal epithelial cells before gametogony begins. Meront types A to E divide asexually by endodyogeny, endopolygeny or schizogony (division into more than two organisms). The origin of gamonts has not been determined, but it is probable that the merozoites released from schizonts of types D and E initiate gamete formation. Gamonts occur throughout the small intestine but most commonly in the ileum (Figure 6). Gamonts and schizonts are located in surface epithelial cells, usually above the host cell nucleus (Figure 6).
The female gamete is subspherical and contains a single centrally located nucleus (Figure 6) Mature male gamonts are ovoid to ellipsoidal in shape and contain 10 to 21 nuclei (Figure 6). Each microgamete has two flagella. The microgametes swim to and penetrate a mature macrogamete. After penetration, oocyst wall formation begins around the fertilized gamete. When they are mature, the oocysts are discharged into the intestinal lumen following rupture of the infected intestinal epithelial cells (Figure 7)22
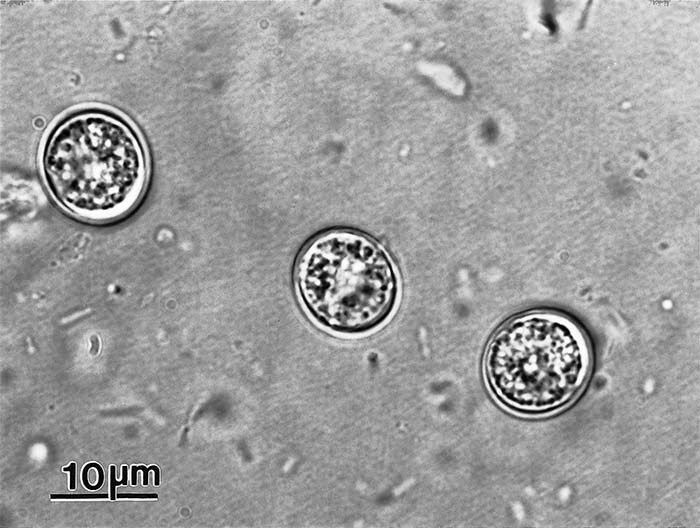
Unsporulated oocysts are subspherical to spherical and are 10 × 12 μm in diameter (Figure 7) Sporulation occurs outside the cat within one to five days depending upon aeration and temperature. Sporulated oocysts are subspherical to ellipsoidal and each sporulated oocyst contains two ellipsoidal sporocysts, each with four sporozoites.
As the enteroepithelial cycle progresses, bradyzoites penetrate the lamina propria of the feline intestine and multiply as tachyzoites.22
Within a few hours after infection of cats, T. gondii may disseminate to extraintestinal tissues. Toxoplasma gondii persists in intestinal and extraintestinal tissues of cats for several months, if not for the life of the cat. Thus, a cat is both the intermediate and the definitive host.
Intermediate hosts become infected after the ingestion of sporulated oocysts or tissue cysts containing bradyzoites. In the lumen of the small intestine, sporozoites released from oocysts and bradyzoites liberated from tissue cysts penetrate the mucosa to reach the submucosa and regional lymph nodes where they first multiply asexually (Figure 8) and are then disseminated via the blood and lymph streams37, 49 to infect a variety of cell types in which they multiply by endodyogeny to form tissue cysts. Sporozoites and bradyzoites of T. gondii can be transmitted to the foetus via the placenta. This occurs in humans, sheep, goats and several higher mammals when infection occurs during pregnancy.39, 45, 58 However, in mice, infection can be transmitted to the foetus from the dam infected before pregnancy and for many generations thereafter.4 In birds, the transmission of T. gondii to eggs has been reported.8, 81
Epidemiology
Infections of animals and humans are acquired from different sources.
Domestic cats play a central role in the dissemination of T. gondii in nature. Not only domestic cats (Felis domesticus) but other members of the Felidae can shed oocysts.39 Oocysts of T. gondii were reported from faeces of naturally infected jaguar (Panthera onco),10 ocelots (Felis pardalis),83 cheetah (Acinonyx jubatus), bobcats (Lynx rufus),76 Canadian lynx (Lynx canadensis),1 and mountain lion (Puma concolor).98 Outbreaks of acute toxoplasmosis in humans were attributed to contamination of a water reservoir by oocysts shed by feral domestic cats 9 and oocysts inhaled in a riding stable.92 A cat may shed millions of oocysts after ingesting a few tissue cysts or bradyzoites.22, 38 Oocysts are shed in large numbers for two to three weeks following infection. Cats usually develop a solid immunity after primary infection, so that although they may shed oocysts on reinfection, fewer oocysts are shed than after primary infection.53 Oocysts are not present on a cat’s fur, therefore petting a cat is not the source of T. gondii infection for humans, but handling faecally contaminated material, such as sand-boxes or garden soil, is.53 Sporulated oocysts are resistant to environmental factors and can survive freezing and drying.39
Herbivorous animals mainly become infected as a result of faecal contamination of pastures, water and feed with oocysts from cats35 and other felids, and pasture contamination may be of more importance than conserved feed. Sporulated oocysts remain infectious for more than a year and, because of their small size, are easily dispersed by wind and rain. Transmission of T. gondii to ewes by contact with infected placentas and aborted foetuses is not of practical significance.
Toxoplasma gondii may be transmitted to pigs not only as a result of the ingestion of oocysts, but also of uncooked or undercooked swill, infected rodents or other intermediate hosts, or through cannibalism.
There is possibly also an active sylvatic cycle in which wild felids act as definitive hosts, and their prey, such as rodents and birds, are intermediate hosts. In southern Africa, in which a large population of wild felids and a wide range of potential intermediate hosts exist, such a sylvatic cycle may play an important role in the transmission of T. gondii to herbivores, free-range pigs, and possibly humans.
Very little information is available on the prevalence of toxoplasmosis in animals in countries in southern Africa.68, 82, 90, 93 However, notwithstanding a high prevalence of serologically positive animals, the occurrence of disease in domestic animals is very rare and only a few cases of toxoplasmosis have been reported in dogs,79, 89, 96 although these might have been due to neosporosis (see Neosporosis). A cat with severe clinical toxoplasmosis was reported.78 Additionally, T. gondii has been reported in a Cape hunting dog (Lycaon pictus)66 and a cheetah97 in southern Africa. The finding of antibodies to T. gondii in 50 (74 per cent) of 68 sera from wild felids from South Africa — 37 of 41 lions (Panthera leo), three of four leopards (Panthera pardus), and 10 of 23 cheetahs — has been reported.16
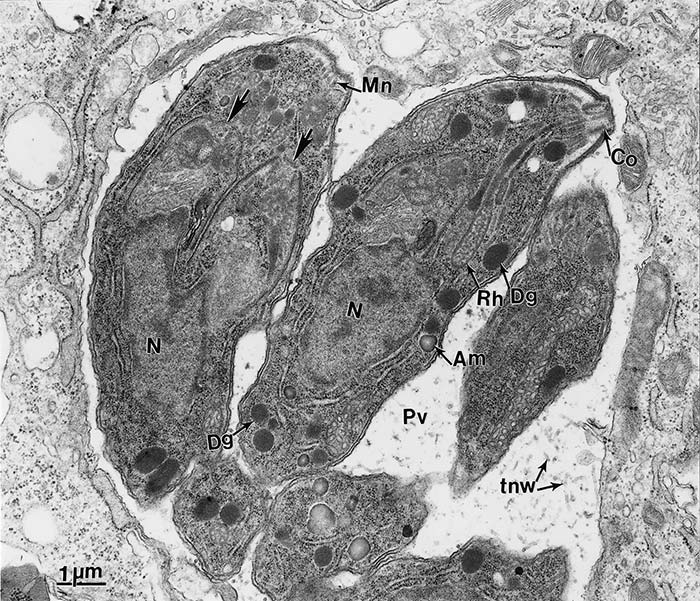
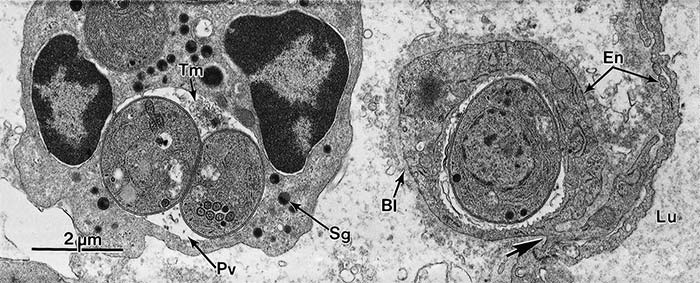
Figure 3 Transmission electron micrograph of tachyzoites of Toxoplasma gondii in a parasitophorous vacuole (Pv) of host cytoplasm in the peritoneal exudate of a mouse. Note conoid (Co), electron-lucent rhoptries (Rh), dense granules (Dg), amylopectin (Am), a centrally located nucleus (N), and the intravacuolar tubular network (tnw) in Pv. One tachyzoite is dividing into two by endodyogeny. Note conoids of the daughter organisms (arrows) are present above the parent nucleus. (Prof C. A. Speer, College of Agricultural Sciences and Natural Resources, University of Tennessee, Knoxville, and Drs D. Fritz, K. Kuehl and D. Austing, USAMRIID, Fort Deterick, Maryland, USA, are acknowledged with thanks for providing this figure and Figures 4, 5 and 8)
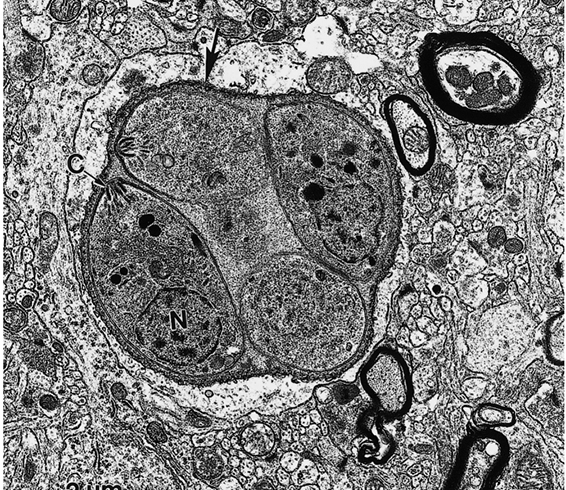
Figure 4 Transmission electron micrograph of a young tissue cyst in a mouse brain. Note well-developed cyst wall (arrow) enclosing four bradyzoites. Note conoid (C) and posteriorly located nucleus (N) in one bradyzoite. (This figure was provided by Prof. C.A. Speer and Drs D. Fritz, K. Kuehl and D. Austing; vide Figure 3)
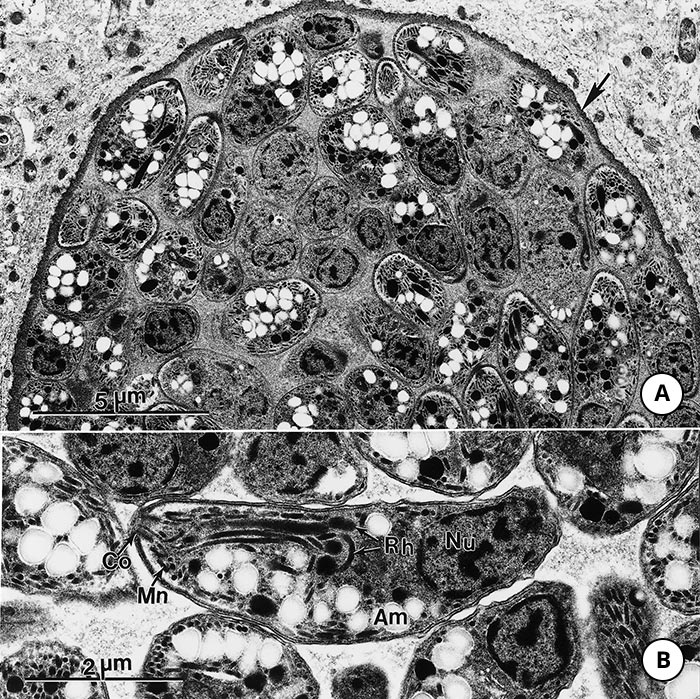
Figure 5 Transmission electron micrograph of a large tissue cyst of Toxoplasma gondii in a mouse brain.
- Note thin cyst wall (arrow) and many bradyzoites
- Higher magnification of a bradyzoite in longitudinal section.
Note conoid (Co), micronemes (Mn), electron-dense rhoptries (Rh) with convoluted necks, and a terminally located nucleus (Nu). (This figure was provided by Prof. C.A. Speer and Drs D. Fritz, K. Kuehl and D. Austing; vide Figure 3)
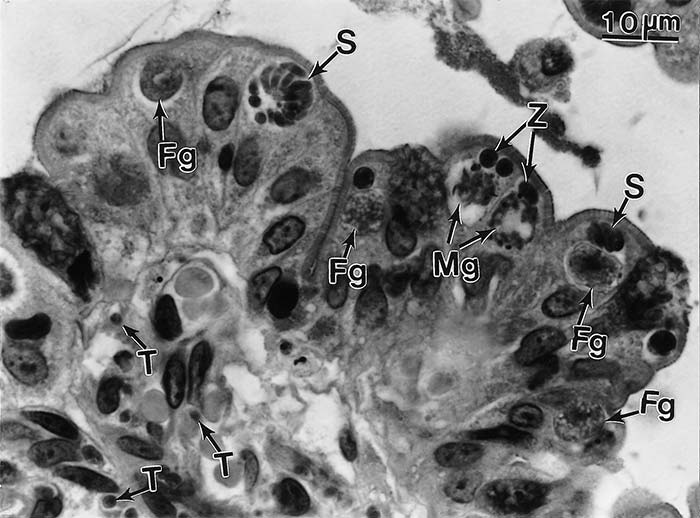
Figure 6 Enteroepithelial stages of Toxoplasma gondii in ileum of a cat six days after feeding tissue cysts. Note mature schizonts (S), male gamonts (Mg), and female gamonts (Fg) are located in epithelial cells above the host cell nucleus. Zoites (probably developing schizonts) are also present toward the brush border. Tachyzoites (T) are present in the lamina propria. Haematoxylin and eosin stain
Toxoplasma gondii is an important cause of abortion and related problems in sheep in several countries including the USA, Australia, New Zealand, the UK, and Norway.3, 12, 26, 39, 45, 48, 64, 65, 66 In these countries toxoplasmosis is a primary cause of loss in 10 to 20 per cent of flocks with abortion/ stillbirth problems. Similarly, toxoplasmosis can cause severe losses in goats.29, 44, 54
The consumption of undercooked meat containing tissue cysts appears to be the most common route of infection in humans. Tissue cysts have been isolated from mutton, pork, chicken, and horse meat,73 and molecular detection has also been successful in beef.75 Nonetheless, cattle are highly resistant to T. gondii infection and the consumption of beef is probably not of significant importance in the transmission of the infection.39 Goat milk containing tachyzoites has been proven to cause human T. gondii infections.86 In addition, as stated above, human infection can also occur following the accidental ingestion of oocysts from cat faeces.
In Central America and other countries with a moist tropical climate, the prevalence of serologically positive humans increases from about one year of age to adolescence. This is considered to be due to children in these areas habitually playing in oocyst-contaminated soil.39 Generally, due to the chronic character of toxoplasmosis, increasing seropositivity is observed in human populations with increasing age even in adults.101 Toxoplasma gondii is an opportunistic pathogen in humans with immune system dysfunction. In human immunodeficiency virus infections toxoplasmosis usually presents as a meningoencephalitis. It is also important for pregnant women as tachyzoites can cross the placental barrier and cause defects in the developing foetus.
A wide variation in the occurrence of T. gondii antibodies has been recorded in humans in tropical Africa.39 A limited number of studies concerning human toxoplasmosis have been conducted in southern Africa. In South Africa, in one study, 37 per cent of 806 human sera from Gauteng and in another study 21 per cent of 10,228 sera (Joubert and Evans 1997) were positive,77 while, in a third study, 29 per cent of the general population in Zimbabwe were serologically positive.90 However, 47 per cent of the slaughtermen in Zimbabwe (n = 90) were serologically positive, the source of infection possibly being associated with the handling or consumption of raw or undercooked meat.
Pathogenesis
After ingestion of T. gondii by a non-feline host (although the following sequence of events can also occur in cats), it first multiplies in the submucosa of the small intestine and its associated lymph nodes37, 49 (Figure 9). Rarely, the host may die of enteritis and lymphadenitis before other organs are affected.39 Parasitaemia occurs during the first week after infection, tachyzoites being disseminated in lymphocytes, macrophages and granulocytes, and as free forms via the lymph and blood to the tissues and organs in which they actively invade, or are actively phagocytosed by, host cells.25 They multiply and, in doing so, destroy the host cell and cell-to-cell transmission may occur within infected organs25 (Figure 9). In this way small necrotic foci are formed that elicit an inflammatory reaction in which mononuclear cells predominate. In most animals protective immunity develops before tachyzoites have had time to cause serious deleterious effects. The infection, however, continues to develop subclinically and tissue cysts containing bradyzoites form. Tissue cysts do not usually evoke any host reaction as encystment protects the bradyzoites from both the humoral and cellular immune systems. In immunocompetent hosts, occasional cyst activation and rupture with release of bradyzoite stages occurs, which is generally handled by the immune system and does not lead to massive replication or clinical illness.20 Inflammation may follow cyst rupture and reactivation of infection may occur in immunosuppressed animals or humans, such as those suffering from the acquired immunodeficient disease syndrome (AIDS).39
In susceptible pregnant animals, transplacental infection causes major economically relevant lossess. Parasitaemia and foetal infection occur during the second week after ingestion of oocysts by pregnant sheep and goats.50 Severe foetal damage is observed as a result of either placentitis or foetal infection, or both.
The severity of the lesions in the foetus generally exceeds that in the dam, the effect of the foetal infection largely being determined by the age of the conceptus. Infection during the early stages of gestation may lead to embryonic death and resorption, while infection of ovine foetuses at about 70 to 90 days of gestation is responsible for the death of a large proportion of the foetuses, and they are either aborted or become mummified and are then expelled at a later stage.26 Some survive the infection for several weeks but are born dead close to term, or else they are born alive but weak and die soon thereafter. Infection of ewes during the last 30 days of gestation generally results in subclinical infection of new-born lambs. Sheep in particular are highly susceptible to reproductive losses.55
The pathogenicity of T. gondii is closely related to the virulence of the strain of the organism and the susceptibility of the host species. New World monkeys, Australian marsupials and Pallas cats are the most susceptible to toxoplasmosis, whereas Old World monkeys, rats, cattle, horses, buffaloes, and chickens72 are highly resistant to clinical toxoplasmosis.24, 39 Other unknown factors vaguely classified as stress may affect the outcome of T. gondii infection in a host. More severe infections are found in pregnant or lactating mice than in non-pregnant or nonlactating mice. Concomitant infections may make a host more susceptible or resistant to T. gondii infection. Clinical toxoplasmosis in dogs is often associated with canine distemper, ehrlichiosis and lymphosarcoma.39, 71
Figure 8 Transmission electron micrograph of cells in the ileal lamina propria of the small intestine of a mouse 48 hours after feeding oocysts. Note the cell on the left is a neutrophil showing specific granules (Sg) and a parasitophorous vacuole (Pv) containing two tachyzoites and a well-developed tubulovesicular membranous network (Tm); on the right side a single tachyzoite is located within an endothelial cell (En) that has become displaced but is still connected by a narrow cytoplasmic bridge (arrow) with the capillary endothelium (En); basal lamina of capillary endothelium (BI); lumen (Lu) of capillary; specific granule (Sg) of neutrophil. (This figure was provided by Prof. C.A. Speer and Drs D. Fritz, K. Kuehl and D. Austing; vide Figure 3)
Clinical signs
In livestock, clinical signs are most commonly encountered after transplacental infection has occurred. Infection of sheep and goats during the early stages of pregnancy results in resorption, abortion or mummification of the foetus. Infection between 50 and 120 days of gestation results in abortion or the birth of premature, weak lambs or kids, or in the birth of offspring suffering from subclinical disease. Apart from aborting, ewes and does do not generally manifest any other evidence that they are infected. Clinical toxoplasmosis has, however, been observed in adult goats,34 pigs31 and occasionally chickens39, 61 but is rare. Blindness and torticollis have been described in canaries.102 There is no well-documented case of naturally acquired clinical toxoplasmosis in cattle and horses.33, 39 Clinical toxoplasmosis occurs in dogs, cats and Australian marsupials.24, 39 The clinical signs manifested by 100 cases of fatal toxoplasmosis in cats from one hospital in the USA21 and in wild and zoo animals24 have been reviewed. Acute systemic toxoplasmosis in animals induces a wide variety of clinical signs because of the many organs and tissues that may be affected. The most common signs observed are those resulting from encephalitis, pneumonia and retinitis.10 However, clinical signs in gallinaceous birds like chickens and turkeys are rarely observed.2, 46, 60 Toxoplasma gondii features a neurotrope and myotrope distribution pattern in many hosts.18 In spite of the reproduction of the parasite in the intestine of cats, clinical evidence of enteric disease has not been recorded.
Most postnatally acquired infections in humans are generally asymptomatic, but fever, lymphodenopathy, headache, myalgia and fatigue may occur.39, 58 Severe illness may develop in those suffering from immunodeficiency syndromes or who are undergoing immunosuppressive therapy. Congenital infection, on the other hand, may have serious consequences, chorioretinitis, neurological and neuropsychiatric disturbances57 and hydrocephalus being most commonly manifested in non-abortion cases. Clinical toxoplasmosis in humans has been reported in South Africa.87
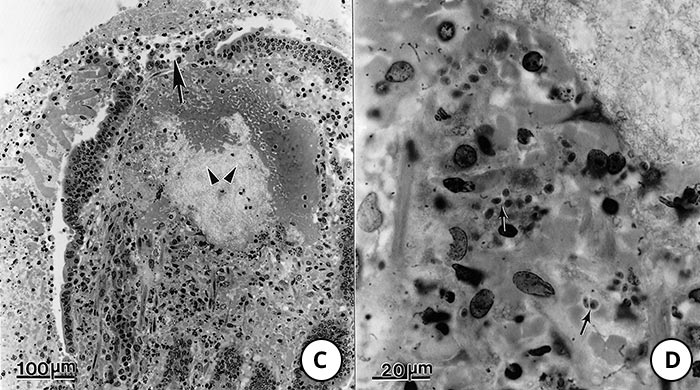
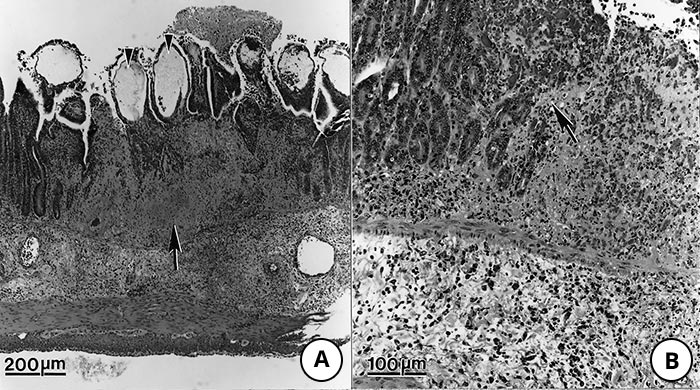
Figure 9 Lesions in the small intestine of pigs fed Toxoplasma gondii oocysts
- Note transmural necrosis (arrow) and ballooning of villi (arrowheads)
- Higher magnification of an area of inflammation and necrosis (arrow) adjacent to relatively unaffected epithelium
- High magnification of a villus showing oedema of lamina propria (arrowheads) and exudation into the lumen from an ulcer (arrow)
- Higher magnification of lamina propria showing tachyzoites (arrows). Haematoxylin and eosin stain
Pathology
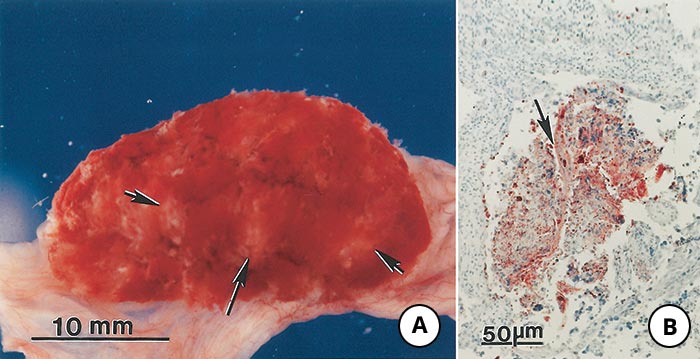
Systemic toxoplasmosis occurs mainly in young animals and immunocompromised hosts. The predominant lesions of systemic toxoplasmosis in animals comprise interstitial pneumonitis, focal hepatic necrosis, lymphadenitis, myocarditis and non-suppurative meningoencephalitis.21, 24, 32, 34, 39, 51, 62, 71, 84 Macroscopic lesions in the lung vary from irregular grey areas of necrosis to haemorrhagic pneumonia. Necrosis is common and appears to be related to the rapid replication of tachyzoites. The most striking and pathognomonic lesions of toxoplasmosis in animals, however, are in the placenta of sheep and goats.5, 6, 12, 29, 39, 44, 45, 50, 63, 85 These consist of foci of necrosis varying in size from microscopic to macroscopic in the cotyledons while intercotyledonary areas are unaffected (Figure 10) The characteristic lesions manifest as white flecks or multiple white, chalky nodules up to 2mm in diameter and are found in approximately half of the confirmed cases. They may be sparse or dense, may occur in any plane of the cotyledon (Figure 10) and may be confluent. Not all cotyledons are affected to the same degree. It is often necessary to wash the cotyledons thoroughly in saline solution in order to expose the deeper lesions that may otherwise be overlooked.
The earliest lesions are necrosis of mesenchymal cells and oedema in the foetal villi with infiltration of mononuclear cells associated with hyperplasia and focal coagulative necrosis of the trophoblastic epithelium. Older lesions consist of foci of caseous necrosis involving foetal and maternal villi that may show mineralization. Most T. gondii present in lesions are degenerated and difficult to find in haematoxylin and eosin-stained sections. lmmunohistochemical staining is useful in locating T. gondii 95 (Figure 10 C) and (D).
Lesions in foetal tissues are microscopic and occur particularly in the brain, liver and lungs.6, 39, 63 Lesions in the brain are found in 90 per cent of cases and consist of leukoencephalomalacia and gliosis.
Placental lesions have also been described in toxoplasmosis in cats42 and pigs.47 Numerous pale foci of necrosis were seen in the allantochorion of the cats while multiple foci of necrosis of the allantochorion were associated with numerous tachyzoites in the pigs.
Diagnosis
The diagnosis of toxoplasmosis can be made by direct biological or histological pathogen detection, serological methods, or by a combination of some of these. Clinical signs are not sufficiently specific to enable a definite diagnosis to be made because the disease resembles several other infectious diseases depending on the affected organs.
Numerous serological procedures are available for the detection of humoral antibodies, including Sabin-Feldman dye test, indirect haemagglutination test (IHT), indirect fluorescent antibody test (IFT), modified direct agglutination test (MAT), latex agglutination tests, enzyme-linked immunoabsorbent assay (ELISA), and immuno absorbent agglutination assay test (IAAT).39
The IFT, IAAT and ELISA have been modified to enable detection of IgM antibodies, which appear sooner after infection than IgG and disappear faster than IgG after recovery. The finding of antibodies to T. gondii in one serum sample from an animal merely establishes that the host has been infected at some time in the past; it is therefore preferable to collect two samples from the same animal, the second two to four weeks after the first. A four-fold increase in antibody titre in the second sample indicates an acute infection. In some animals a high antibody titre persists for months after infection. A rise in antibody titre may not be associated with clinical signs. The fact that titres persist after clinical recovery complicates the interpretation of the results of serological tests. The MAT using formalin-preserved whole tachyzoites and mercaptoethanol has proved very useful in measuring IgG antibodies in livestock, including poultry.39, 40, 46, 48
Finding antibodies in foetal fluids or presuckling serum in ruminants is diagnostic of congenital infection because maternal antibodies do not cross the placenta.27, 40
Toxoplasma gondii can be isolated from the infected tissues, secretions, excretions and body fluids of animals collected either ante- or post-mortally, by inoculating them into laboratory animals and tissue culture.39 Using these methods it is possible not only to isolate T. gondii, but also microscopically to visualize the organism or demonstrate its DNA using the polymerase chain reaction (PCR) test.56, 80 By PCR, T. gondii strains can be genotyped allowing their classification and comparison to other isolated strains in terms of geographical distribution and virulence,88 A rapid diagnosis may be made by the microscopic examination of impression smears of lesions. After drying for 10 to 30 minutes, the smears are fixed in methyl alcohol and stained with a Romanowsky stain, Giemsa being very satisfactory. Well-preserved T. gondii are crescent shaped (Figure 2 A). In tissue sections, the tachyzoites usually appear round to oval (Figure 2 B). Tissue cysts are usually spherical and lack septa, and their walls stain with silver stains. The bradyzoites are strongly PAS positive (Figure 2 D). The immunohistochemical staining of parasites with fluorescent or other types of labelled T. gondii antiserum can assist in the diagnosis95 (Figure 10). Electron microscopy can aid the diagnosis. Toxoplasma gondii tachyzoites are always located in vacuoles; they have few (usually four) rhoptries and often have a honeycomb structure (Figure 3).
In cats, oocysts in faeces are concentrated by salt or sugar floatation techniques.39 They can be differentiated from the oocysts of Isospora spp. by the fact that T. gondii oocysts are smaller (10 × 12 μm) and are shed in an unsporulated state (Figure 7). Toxoplasma. gondii oocysts are morphologically highly similar to Hammondia hammondi oocysts.For unambiguous differentiation of oocysts in feline faecal samples PCR is a very suitable method.17 Again, genotyping may deliver additional information.
Ewes are usually serologically positive at the time of abortion, and a negative test generally excludes toxoplasmosis as a cause of the abortion. A positive result, however, is not necessarily diagnostic, as high titres may persist into the following breeding season.39
Figure 10 Lesions in the placenta of goats and sheep naturally infected with Toxoplasma gondii
- Cotyledon of a goat with whitish-yellow areas of discolouration (arrows). The intercotyledonary area is normal. Unstained
- Microscopic lesion in the placenta of a goat. Note necrosis, mineralization, and dispersed T. gondii antigen (red coloured areas). Immunohistochemical staining with anti-T. gondii serum
- Cut surface of a sheep cotyledon showing yellowish-white areas of necrosis in villi (arrows). Unstained
- Higher magnification of Figure 10 C showing intact (arrows) tachyzoites and dispersed T. gondii antigens (arrowheads)
Differential diagnosis
Foetal and neonatal losses in sheep resulting from T. gondii infection should be differentiated from other possible causes of abortion, stillbirths or neonatal deaths, such as those caused by Chlamydophila abortus (Chlamydia psittaci), Campylobacter jejuni, Listeria monocytogenes, Salmonella serovars, Brucella spp., Coxiella burnetii, Rift Valley fever and Wesselsbron disease viruses, Neospora caninum, and Sarcocystis spp. (see the relevant chapters on these diseases).
Control
The control of toxoplasmosis in livestock is aimed at the prevention of infection in pregnant animals. The perpetuation of the life cycle of T. gondii in them depends on the availability of susceptible definitive hosts, the most important of which is probably the domestic cat, and of infected intermediate hosts, such as rodents. Cats and rodents should therefore be excluded from areas where livestock are fed, or where their feed is stored. If swill is fed to pigs, it should be cooked, and cannibalism should also be prevented.
Treatment of individual cases is usually limited to those suffering from acute infections in high-risk groups, and to immunologically compromised hosts. The treatment of choice is potentiated sulphonamide mixtures, such as sulphadoxine, and trimethoprim.39 These two drugs act synergistically to inhibit sequential steps in the biosynthesis of folic acid. Monensin at a dose of 5 mg/animal/day incorporated into the feed of pregnant ewes can reduce losses from toxoplasmosis.15 Experimentally, monensin limits but does not prevent infection; ewes therefore develop protective immunity to subsequent challenge. The level of monensin in the feed should not exceed 30 ppm as otherwise refusal of feed or toxicity may occur. Incorporation of monensin (200 ppm) into feed blocks is a safe way of utilizing this drug.13 The addition of decoquinate to the feed of pregnant ewes at the daily rate of 2 mg/kg body weight reduces the deleterious effects of T. gondii.14 A live modified T. gondii vaccine is marketed in some European countries and New Zealand for sheep to reduce the prevalence of abortion.11 Other vaccines are currently not (yet) available.
Pregnant women who are serologically negative should avoid gardening without wearing gloves. Cat sand trays should be cleaned daily as oocysts will not have had time to sporulate.
Pregnant women should not eat raw or undercooked meat and should wash their hands after handling raw meat. Immunity follows infection so that an abortion due to toxoplasmosis during chronic infection is unlikely.
References
- ARAMINI, J.J., STEPHEN, C. & DUBEY, J.P., 1998. Toxoplasma gondii in Vancouver Island cougars (Felis concolor vancouverensis): serology and oocyst shedding. Journal of Parasitology, 84, 438–440.
- BANGOURA, B., ZÖLLER, B., KOETHE, M., LUDEWIG, M., POTT, S., FEHLHABER, K., STRAUBINGER, R.K. & DAUGSCHIES, A, 2013. Experimental Toxoplasma gondii oocyst infections in turkeys (Meleagris gallopavo). Veterinary Parasitology, 196(3-4), 272-277.
- BEVERLEY, J.K.A. & WATSON, W.A., 1961. Ovine abortion and toxoplasmosis in Yorkshire. The Veterinary Record, 73, 6–11.
- BEVERLEY, J.K.A., 1959. Congenital transmission of toxoplasmosis through successive generations of mice. Nature, 183, 1348–1349.
- BEVERLEY, J.K.A., WATSON, W.A. & PAYNE, J.M., 1971. The pathology of the placenta in ovine abortion due to toxoplasmosis. The Veterinary Record, 88, 124–128.
- BEVERLEY, J.K.A., WATSON, W.A. & SPENCE, J.B., 1971. The pathology of the foetus in ovine abortion due to toxoplasmosis. The Veterinary Record, 88, 174–178.
- BIGALKE, R.D., TUSTIN, R.C., DU PLESSIS, J.L., BASSON, P.A. & MCCULLY, R.M., 1966. The isolation of Toxoplasma gondii from ferrets in South Africa. Journal of the South African Veterinary Medical Association, 37, 243–247.
- BOCH, J., ROMMEL, M., WEILAND, G., JANITSCHKE, K. & SOMMER, R., 1966. Experimentelle Toxoplasma-Infektionen bei Legehennen. Berliner und Munchener Tierarztliche Wochenschrift, 79(18), 352-356.
- BOWIE, W.R, KING, A.S, WERKER, D.H., ISAAC-RENTON, J.L., BELL, A., ENG, S.B. & MARION, S.A., 1997. Outbreak of toxoplasmosis associated with municipal drinking water. Lancet, 350, 173–177.
- BURRIDGE, J.J., 1980. Toxoplasmosis. The Compendium of Continuing Education for Veterinarians, 5, 456–462.
- BUXTON, D. & INNES, E.A., 1995. A commercial vaccine for ovine toxoplasmosis. Parasitology, 10, S11–S16.
- BUXTON, D., 1990. Ovine toxoplasmosis: a review. Journal of the Royal Society of Medicine, 83, 509–511.
- BUXTON, D., BLEWETT, D.A., TREES, A.J., MCCOLGAN, C. & FINLAYSON, J., 1988. Further studies in the use of monensin in the control of experimental ovine toxoplasmosis. Journal of Comparative Pathology, 98, 225–236.
- BUXTON, D., BREBNER, J., WRIGHTS, S., MALEY, S.W., THOMSON, K.M. & MILLARD, K., 1996. Decoquinate and the control of experimental ovine toxoplasmosis. The Veterinary Record, 138, 434–436.
- BUXTON, D., DONALD, K.M. & FINLAYSON, J., 1987. Monensin and the control of experimental ovine toxoplasmosis: A systemic effect. The Veterinary Record, 120, 618–619.
- CHEADLE, M.A., SPENCER, J.A., & BLAGBURN, B.L., 1999. Seroprevalence of Neospora caninum and Toxoplasma gondii in nondomestic felids from Southern Africa. Journal of Zoo and Wildlife Medicine, 30, 248–251.
- CHEMOH, W., SAWANGJAROEN, N., NISSAPATORN, V. & SERMWITTAYAWONG, N., 2016. Molecular investigation on the occurrence of Toxoplasma gondii oocysts in cat feces using TOX-element and ITS-1 region targets. The Veterinary Journal, 215, 118-122.
- DISKO, R., BRAVENY, I. & GREUTÉLAERS, M.T., 1978. Experimental studies on the affinity of Toxoplasma gondii to various organs of mice (author's transl). Zentralblatt fuer Bakteriologie, Parasitenkunde, Infektionskrankheiten und Hygiene, Erste Abteilung Originale, Reihe A, 242(4), 565-571.
- DU PLESSIS, J.L., BIGALKE, R.D. & GURNELL, T.A., 1967. An outbreak of toxoplasmosis in chinchillas in South Africa. Journal of the South African Veterinary Medical Association, 38, 79–83.
- DUBEY, J.P., FERREIRA, L.R., ALSAAD, M., VERMA, S.K., ALVES, D.A., HOLLAND, G.N. & MCCONKEY, G.A., 2016. Experimental toxoplasmosis in rats induced orally with eleven strains of Toxoplasma gondii of seven genotypes: tissue tropism, tissue cyst size, neural lesions, tissue cyst rupture without reactivation, and ocular Lesions. Public Library of Science One, 11(5), e0156255.
- DUBEY, J.P. & CARPENTER, J.L., 1993. Histologically confirmed clinical toxoplasmosis in cats—IGO cases (I952–1990). Journal of the American Veterinary Medical Association, 203, 1556–1566.
- DUBEY, J.P. & FRENKEL, J.K., 1972. Cyst induced toxoplasmosis in cats. Journal of Protozoology, 19, 155–177.
- DUBEY, J.P. & FRENKEL, J.K., 1976. Feline toxoplasmosis from acutely infected mice and the development of Toxoplasma cysts. Journal of Protozoology, 23, 537–546.
- DUBEY, J.P. & ODENING, K., 2001. Toxoplasmosis and related infections. In: SAMUEL, B., PYBUR, M. & KOCAN, A.M., (eds). Parasitic Diseases of Wild Mammals. Ames: Iowa State University Press, 478–519.
- DUBEY, J.P. & SHARMA, S.P., 1980. Parasitemia and tissue infection in sheep fed Toxoplasma gondii oocysts. Journal of Parasitology, 66, 111–114.
- DUBEY, J.P. & TOWLE, A., 1986. Toxoplasmosis in sheep: A review and annotated bibliography. Miscellaneous Publication No. 10 of the Commonwealth Institute of Parasitology, London, 1–152.
- DUBEY, J.P. & URBAN, J.F., 1990. Diagnosis of transplacentally induced toxoplasmosis in pigs. American Journal of Veterinary Research, 51, 1295–1299.
- DUBEY, J.P., 1977. Toxoplasma, Hammondia, Besnoitia, Sarcocystis, and other tissue cyst-forming coccidia of man and animals. In: KRIER, J.P., (ed.). Parasitic Protozoa. New York: Academic Press, 3, 101–237.
- DUBEY, J.P., 1981. Epizootic toxoplasmosis associated with abortion in dairy goats in Montana. Journal of the American Veterinary Medical Association, 178, 661–670.
- DUBEY, J.P., 1984. Experimental toxoplasmosis in sheep fed Toxoplasma gondii oocysts. International Goat and Sheep Research, 2, 93–104.
- DUBEY, J.P., 1986. A review of toxoplasmosis in pigs. Veterinary Parasitology, 19, 181–223.
- DUBEY, J.P., 1986. Toxoplasmosis in cats. Feline Practice, 16, 12–46.
- DUBEY, J.P., 1986b. A review of toxoplasmosis in cattle. Veterinary Parasitology, 22, 177–202.
- DUBEY, J.P., 1987. Toxoplasmosis in goats. Agri-Practice, 8, 43–52.
- DUBEY, J.P., 1994. Toxoplasmosis. Journal of the American Veterinary Medical Association, 205, 1593–1598.
- DUBEY, J.P., 1996. Infectivity and pathogenicity of Toxoplasma gondii oocysts for cats. Journal of Parasitology, 82, 957–960.
- DUBEY, J.P., 1997. Bradyzoite-induced murine toxoplasmosis: stage conversion, pathogenesis, and tissue cyst formation in mice fed bradyzoites of different strains of Toxoplasma gondii. Journal of Eukaryotic Microbiology, 44, 592–602.
- DUBEY, J.P., 2001. Oocyst shedding by cats fed isolated bradyzoites and comparison of infectivity of bradyzoites of the VEG strain Toxoplasma gondii to cats and mice. Journal of Parasitology, 87, 215–219.
- DUBEY, J.P., BEATTIE, C.P., 1988. Toxoplasmosis of Animals and Man. Boca Raton, Florida: CRC Press, 1–220.
- DUBEY, J.P., EMOND, J.P., DESMONTS, G. & ANDERSON, W.R., 1987. Serodiagnosis of postnatally and prenatally induced toxoplasmosis in sheep. American Journal of Veterinary Research, 48, 1239–1243.
- DUBEY, J.P., LINDSAY, D.S. & SPEER, C.A., 1998. Structure of Toxoplasma gondii tachyzoites, bradyzoites and sporozoites, and biology and development of tissue cysts. Clinical Microbiology Reviews, 11, 267–299.
- DUBEY, J.P., MATTIX, M.E. & LIPSCOMB, T.P., 1996. Lesions of neonatally induced toxoplasmosis in cats. Veterinary Pathology, 33, 290–295.
- DUBEY, J.P., MILLER, N.I. & FRENKEL, J.K., 1970. The Toxoplasma gondii oocysts from cat feces. Journal of Experimental Medicine, 132, 636–662.
- DUBEY, J.P., MILLER, S., DESMONTS, G., THULLIEZ, P. & ANDERSON, W.R., 1986. Toxoplasma gondii-induced abortion in dairy goats. Journal of the American Veterinary Medical Association, 188, 159–162.
- DUBEY, J.P., MILLER, S., POWELL, E.C. & ANDERSON, W.R., 1986. Epizootologic investigations on a sheep farm with Toxoplasma gondii-induced abortions. Journal of the American Veterinary Medical Association, 188, 155–158.
- DUBEY, J.P., RUFF, M.D., CAMARGO, M.E., SHEN, S.K., WILKINS, G.L., KWOK, O.C.H. & THULLIEZ, P., 1993. Serologic and parasitologic responses of domestic chickens after oral inoculation with Toxoplasma gondii oocysts. American Journal of Veterinary Research, 54, 1668–1672.
- DUBEY, J.P., SCHLAFER, D.H., URBAN, J.F., JR. & LINDSAY, D.S., 1990. Lesions in fetal pigs with transplacentally-induced toxoplasmosis. Veterinary Pathology, 27, 411–418.
- DUBEY, J.P., SONN, R.J., HEDSTROM, O., SNYDER, S.P. & LASSEN, E.D., 1990. Serologic and histologic diagnosis of toxoplasmic abortions in sheep in Oregon. Journal of the American Veterinary Medical Association, 196, 291–294.
- DUBEY, J.P., SPEER, C.A., SHEN, S.K., KWOK, O.C.H. & BLIXT, J.A., 1997 Oocyst-induced murine toxoplasmosis: life cycle, pathogenicity, and stage conversion in mice fed Toxoplasma gondii oocysts. Journal of Parasitology, 83, 870–882.
- DUBEY, J.P., 1981. Toxoplasma-induced abortion in dairy goats. Journal of the American Veterinary Medical Association, 178, 671–674.
- DUBEY, J.P., 1989. Lesions in goats fed Toxoplasma gondii oocysts. Veterinary Parasitology, 32, 133–144.
- DUBEY, J.P., 1993. Toxoplasma, Neospora, Sarcocystis, and other tissue cyst-forming coccidia of humans and animals. In: KRIER, J.P., (ed.). Parasitic Protozoa. New York: Academic Press, 6, 1–158.
- DUBEY, J.P., 1995. Duration of immunity to shedding of Toxoplasma gondii oocysts by cats. Journal of Parasitology, 81, 410–415.
- ENGELAND, I.V., WALDELAND, H., ANDRESEN, O., LOKEN, T., BJORKMAN, C. & BJERKAS, I., 1999. Foetal loss in dairy goats: an epidemiological study in 22 herds. Small Ruminant Research, 30, 37–48.
- ESTEBAN-REDONDO, I. & INNES, E.A., 1997. Toxoplasma gondii infection in sheep and cattle. Comparative Immunology, Microbiology, and Infectious Diseases, 20(2), 191-196.
- ESTEBAN-REDONDO, I. & INNES, E.A., 1998. Detection of Toxoplasma gondii in tissues of sheep orally challenged with different doses of oocysts. International Journal for Parasitology, 28, 1459–1466.
- FABIANI S, PINTO B, BONUCCELLI U & BRUSCHI F, 2015. Neurobiological studies on the relationship between toxoplasmosis and neuropsychiatric diseases. Journal of the Neurological Sciences, 351(1-2), 3-8.
- FRENKEL, J.K., 1973. Toxoplasmosis: Parasitic life cycle, pathology and immunology. In: HAMMOND, D.M. & LONG, P.L., (eds). The Coccidia. Baltimore: University Park Press.
- FREYRE, A., DUBEY, J.P., SMITH, D.D. & FRENKEL, J.K., 1989. Oocyst-induced Toxoplasma gondii infections in cats. Journal of Parasitology, 75, 750–755.
- GEUTHNER A C, KOETHE M, LUDEWIG M, POTT S, SCHARES G, DAUGSCHIES A & BANGOURA B, 2014. Persistence of Toxoplasma gondii tissue stages in poultry over a conventional fattening cycle. Parasitology, 141(11), 1359-64.
- GOODWIN, M.A., DUBEY, J.P. & HATKIN, J., 1994. Toxoplasma gondii peripheral neuritis in chickens. Journal of Veterinary Diagnostic Investigation, 6, 382–385.
- HARTLEY, W.J. & DUBEY, J.P., 1991. Fatal toxoplasmosis in some native Australian birds. Journal of Veterinary Diagnostic Investigation, 3, 167–169.
- HARTLEY, W.J. & KATER, J.G., 1963. The pathology of Toxoplasma infection in the pregnant ewe. Research in Veterinary Science, 4, 326–332.
- HARTLEY, W.J. & MARSHALL, S.C., 1957. Toxoplasmosis as a cause of ovine perinatal mortality. New Zealand Veterinary Journal, 5, 119–124.
- HARTLEY, W.J., MOYLE, G.G., 1968. Observations on an outbreak of ovine congenital toxoplasmosis. Australian Veterinary Journal, 44, 105–107.
- HOFMEYR, C.F.B., 1956. Two hundred and eighty-four autopsies at the National Zoological Gardens, Pretoria. Journal of the South African Veterinary Medical Association, 27, 263–282.
- HOVE, T., LIND, P. & MUKARATIRWA. S., 2005. Seroprevalence of Toxoplasma gondii infection in goats and sheep in Zimbabwe. The Onderstepoort Journal of Veterinary Research, 72(4), 267-272.
- HOVE, T. & DUBEY, J.P., 1999. Prevalence of Toxoplasma gondii antibodies in sera of domestic pigs and some wild game species from Zimbabwe. Journal of Parasitology, 85, 372–373.
- JACOBS, M.R. & MASON, P.R., 1978. Prevalence of Toxoplasma antibodies in southern Africa. South African Medical Journal, 53, 619–621.
- JOUBERT, J.J. & EVANS, A.C., 1997. Current status of food-borne parasitic zoonoses in South Africa and Namibia. The Southeast Asian Journal of Tropical Medicine and Public Health, 28 Supplement, 1, 7-10.
- JUBB, K.V.C., KENNEDY, P.C. & PALMER, N., 1992. Pathology of Domestic Animals. 4th edition. London: Academic Press.
- KANETO, C.N., COSTA, A.J., PAULILLO, A.C., MORAES, F.R., MURAKAMI, T.O. & MEIRELES, M.V., 1997. Experimental toxoplasmosis in broiler chicks. Veterinary Parasitology, 69(3-4), 203-10.
- KLUN, I., UZELAC, A., VILLENA, I., MERCIER, A., BOBIĆ, B., NIKOLIĆ, A., RAJNPREHT, I., OPSTEEGH. M., AUBERT, D., BLAGA, R., VAN DER GIESSEN, J. & DJURKOVIĆ-DJAKOVIĆ, O., 2017. The first isolation and molecular characterization of Toxoplasma gondii from horses in Serbia. Parasites & Vectors, 10(1), 167.
- LYONS, R.E., MCLEOD, R. & ROBERTS, C.W., 2002. Toxoplasma gondii tachyzoite-bradyzoite interconversion. Trends in Parasitology, 18(5), 198-201.
- MAHAMI-OSKOUEI, M., MORADI, M., FALLAH, E., HAMIDI, F. & ASL RAHNAMAYE AKBARI, N., 2017. Molecular detection and genotyping of Toxoplasma gondii in chicken, beef, and lamb meat consumed in Northwestern Iran. Iranian Journal of Parasitology, 12(1), 38-45.
- MARCHIONDO, A.A., DUSZYNSKI, D.W. & MAUPIN, G.O., 1976. Prevalence of antibodies to Toxoplasma gondii in wild and domestic animals of New Mexico, Arizona and Colorado. Journal of Wildlife Diseases, 12, 226–232.
- MASON, P.R., JACOBS, M.R. & FRIPP, P.J., 1974. Serological survey of toxoplasmosis in the Transvaal. South African Medical Journal, 48, 1707–1709.
- NAGEL, S.S., WILLIAMS, J.H. & SCHOEMAN, J.P., 2013. Fatal disseminated toxoplasmosis in an immunocompetent cat. Journal of the South African Veterinary Association, 84(1), E1-E6.
- NESBIT, J.W., LOURENS, D.C. & WILLIAMS, M.C., 1981. Spastic paresis in two littermate pups caused by Toxoplasma gondii. Journal of the South African Veterinary Association, 52, 243–246.
- OWEN, M.R., CLARKSON, M.J. & TREES, A.J., 1998. Diagnosis of toxoplasma abortion in ewes by polymerase chain reaction. The Veterinary Record, 142, 445–448.
- PANDE, P.G., SHUKLA, R.R. & SEKARIAH, P.C., 1961. Toxoplasma from the eggs of the domestic fowl (Gallus gallus). Science, 133(3453), 648.
- PANDEY, V.S. & VAN KNAPEN, F., 1992. The seroprevalence of toxoplasmosis in sheep, goats and pigs in Zimbabwe. Annals of Tropical Medicine and Parasitology, 86, 313–315.
- PATTON, S., JOHNSON, S.L., LOEFFLER, D.G., WRIGHT, B.G. & JENSEN, J.M., 1986. Epizootic of toxoplasmosis in kangaroos, wallabies, and potaroos: possible transmission via domestic cats. Journal of the American Veterinary Medical Association, 189, 1166–1169.
- REDDACLIFF, G.L., HARTLEY, W.J., DUBEY, J.P. & COOPER, D.W., 1993. Pathology of experimentally-induced, acute toxoplasmosis in macropods. Australian Veterinary Journal, 70, 4–6.
- RYAN, J.C. & DUBEY, J.P., 1984. Ovine abortion and neonatal death due to toxoplasmosis in Montana. Journal of the American Veterinary Medical Association, 184, 661–664.
- SACKS, J.J., ROBERTO, R.R. & BROOKS, N.F., 1982. Toxoplasmosis infection associated with raw goat’s milk. Journal of the American Medical Association, 248, 1728-1732.
- SAUNDERS, S.A. & THATCHER, G.N., 1963. Toxoplasmosis in the adult. South African Medical Journal, 37, 1026–1029.
- SHWAB, E.K., ZHU, X.Q., MAJUMDAR, D., PENA, H.F., GENNARI, S.M., DUBEY, J.P. & SU, C., 2014. Geographical patterns of Toxoplasma gondii genetic diversity revealed by multilocus PCR-RFLP genotyping. Parasitology, 141(4), 453-61.
- SMIT, J.D., 1961. Toxoplasmosis in dogs in South Africa: Seven case reports. Journal of the South African Veterinary Medical Association, 32, 339–346.
- SWANEPOEL, R., BLACKBURN, N.K. & EFSTRATIOU, S., 1974. An investigation of the relationship between infection with Toxoplasma gondii and contact with animals. The Central African Journal of Medicine, 20, 206–210.
- TENTER, A.M., HECKEROTH, A.R. & WEISS, L.M, 2000. Toxoplasma gondii: from animals to humans. International Journal of Parasitology, 30(12-13), 1217-1258.
- TEUTSCH, S.M., JURANEK, D.D., SULZER, A., DUBEY, J.P. & SIKES, R.K., 1979. Epidemic toxoplasmosis associated with infected cats. New England Journal of Medicine, 300, 695–699.
- TURNER, G.V.S., 1976. Toxoplasmosis as a public health hazard. Journal of the South African Veterinary Association, 47, 227–231.
- TURNER, G.V.S., 1978. Some aspects of the pathogenesis and comparative pathology of toxoplasmosis. Journal of the South African Veterinary Association, 49, 3–8.
- UGGLA, A., SJÖLAND, L. & DUBEY, J.P., 1987. Immunohistochemical diagnosis of toxoplasmosis in fetuses and fetal membranes of sheep. American Journal of Veterinary Research, 48, 348–351.
- VAN HEERDEN, J. & VAN RENSBURG, I.B.J., 1979. Toxoplasmosis in a dog. Journal of the South African Veterinary Association, 50, 211–214.
- VAN RENSBURG, I.B.J. & SILKSTONE, M.A., 1984. Concomitant feline infectious peritonitis and toxoplasmosis in a cheetah (Acinonyx jubatus). Journal of the South African Veterinary Association, 55, 205–207.
- VAN WORMER, E., CONRAD, P.A., MILLER, M.A., MELLI, A.C., CARPENTER, T.E & MAZET, J.A, 2013. Toxoplasma gondii, source to sea: higher contribution of domestic felids to terrestrial parasite loading despite lower infection prevalence. EcoHealth, 10(3), 277-289.
- WADELAND, H., 1976. Toxoplasmosis in sheep. The relative importance of the infection as a cause of reproductive loss in sheep in Norway. Acta Veterinaria Scandinavica, 17, 412–425.
- WIENGCHAROEN, J., THOMPSON, R.C., NAKTHONG, C., RATTANAKORN, P. & SUKTHANA, Y., 2011. Transplacental transmission in cattle: is Toxoplasma gondii less potent than Neospora caninum? Parasitology Research, 108(5), 1235-1241.
- WILKING, H., THAMM, M., STARK, K., AEBISCHER, T. & SEEBER, F., 2016. Prevalence, incidence estimations, and risk factors of Toxoplasma gondii infection in Germany: a representative, cross-sectional, serological study. Scientific Reports, 6, 22551.
- WILLIAMS, S.M., FULTON, R.M., RENDER, J.A., MANSFIELD, L. & BOULDIN, M., 2001. Blindness and torticollis has been described in canaries. Avian Diseases, 45, 262–267.