- Infectious Diseases of Livestock
- Part 1
- Vectors: Muscidae
- Vectors: Ticks
- Vectors: Tsetse flies
- Vectors: Muscidae
- Vectors: Tabanidae
- Vectors: Culicoides spp.
- Vectors: Mosquitoes
- Classification, epidemiology and control of arthropod-borne viruses
- Special factors affecting the control of livestock diseases in sub-Saharan Africa
- The control of infectious diseases of livestock: Making appropriate decisions in different epidemiological and socioeconomic conditions
- Infectious diseases of animals in sub-Saharan Africa: The wildlife⁄livestock interface
- Vaccination: An approach to the control of infectious diseases
- African animal trypanosomoses
- Dourine
- Trichomonosis
- Amoebic infections
- GENERAL INTRODUCTION: COCCIDIA
- Coccidiosis
- Cryptosporidiosis
- Toxoplasmosis
- Besnoitiosis
- Sarcocystosis
- Balantidiosis
- Leishmaniosis
- Neosporosis
- Equine protozoal myeloencephalitis
- GENERAL INTRODUCTION: BABESIOSES
- Bovine babesiosis
- Equine piroplasmosis
- Porcine babesiosis
- Ovine babesiosis
- GENERAL INTRODUCTION: THEILERIOSES OF CATTLE
- East Coast fever
- Corridor disease
- Zimbabwe theileriosis
- Turning sickness
- Theileria taurotragi infection
- Theileria mutans infection
- Theileria annulata theileriosis
- Theileriosis of sheep and goats
- Theileria buffeli⁄orientalis infection
- Non-pathogenic Theileria species in cattle
- GENERAL INTRODUCTION: RICKETTSIAL, CHLAMYDIAL AND HAEMOTROPIC MYCOPLASMAL DISEASES
- Heartwater
- Lesser known rickettsial infections in animals and humans
- Chlamydiosis
- Q fever
- Eperythrozoonosis
- Bovine Haemobartonellosis
- Potomac horse fever
- GENERAL INTRODUCTION: ANAPLASMOSES
- Bovine anaplasmosis
- Ovine and caprine anaplasmosis
Vectors: Muscidae
This content is distributed under the following licence: Attribution-NonCommercial CC BY-NC  View Creative Commons Licence details here
View Creative Commons Licence details here

Vectors: Muscidae
E M NEVILL
Introduction
In the Afrotropical Region (previously referred to as the Ethiopian or sub-Saharan Region) the family Muscidae embraces 870 species in 60 genera and six subfamilies.42 However, only three subfamilies, the Stomoxyinae, Fanniinae, and Muscinae contain genera of veterinary importance, these being Haematobia, Haematobosca, Stomoxys, Musca, Hydrotaea, Morellia, Muscina and Fannia. Species in these genera are important not only because they may cause irritation to livestock, resulting in production losses,46 but also because of their potential as mechanical vectors of various disease organisms, a fact that has been generally neglected in southern Africa. Greenberg13 listed a multitude of organisms that have been isolated directly from various synanthropic fly species and which have the potential to be transmitted mechanically under the right circumstances. He reviewed the studies made throughout the world on fly transmission of a wide variety of diseases, such as brucellosis, anaplasmosis and summer mastitis.14
The literature is saturated with articles on the control of these flies. In North America much effort has gone into the chemical control of the horn fly (Haematobia irritans irritans) and the face fly (Musca autumnalis), while in Australia a biological control programme to control the buffalo fly (Haematobia irritans exigua) and the bush fly (Musca vetustissima) by the introduction of exotic dung beetles was started in the early 1970s.3 However, the stable fly (Stomoxys calcitrans), the house fly (Musca domestica), the lesser house fly (Fannia canicularis) and the false stable fly (Muscina stabulans) continue to flourish in conditions created by humans, especially in intensive farming situations such as dairies, feedlots and chicken farms.23 The efforts and ingenuity of humans to produce more milk, meat and eggs do not always include efforts to avoid a greater fly problem!
Members of the Muscidae are not biological transmitters of infectious diseases. Although they have been shown to be capable of mechanically transmitting numerous diseaseproducing organisms, they are seldom controlled for this reason. A second reason for southern African farmers’ apparent apathy about these flies is that they are unaware that so many species exist, each with its own requirements for survival and its own potential for disease transmission. For example, at least 20 species of Musca are found on farms in various parts of South Africa, but are often confused with the house fly.31
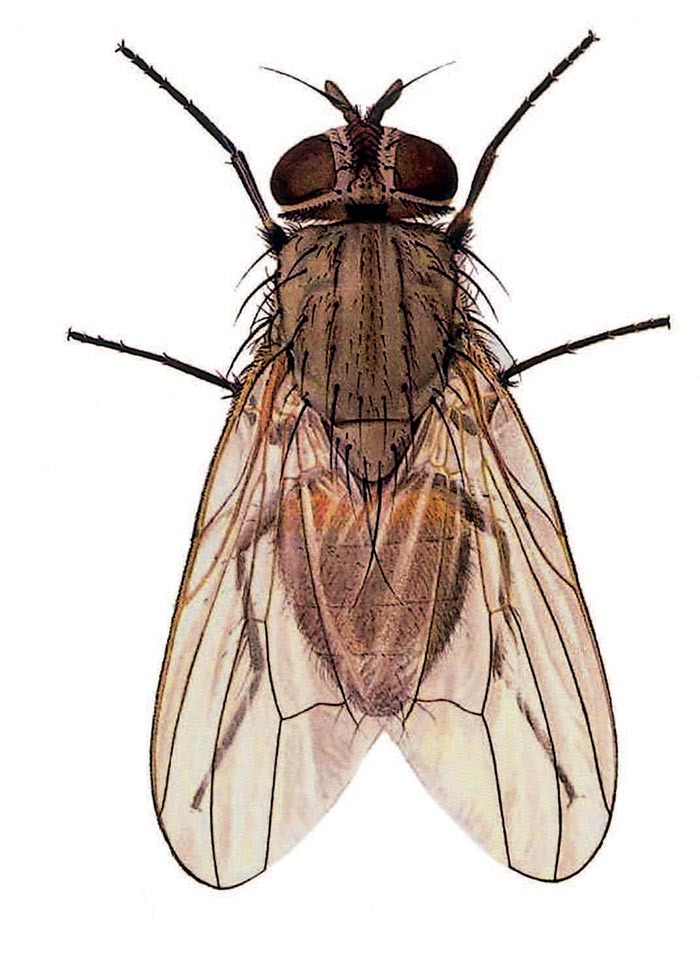
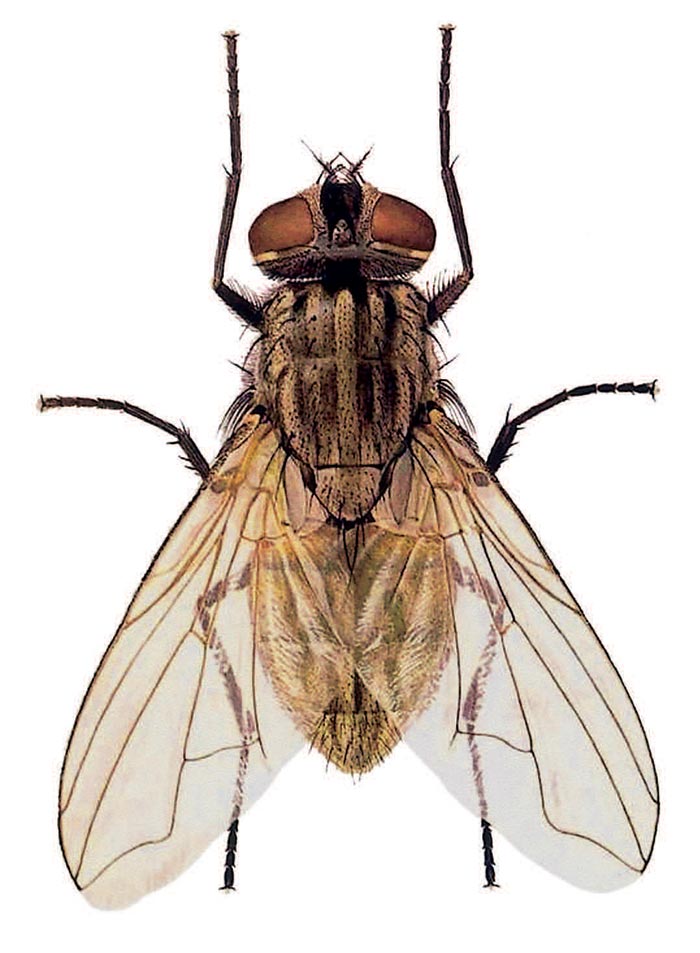
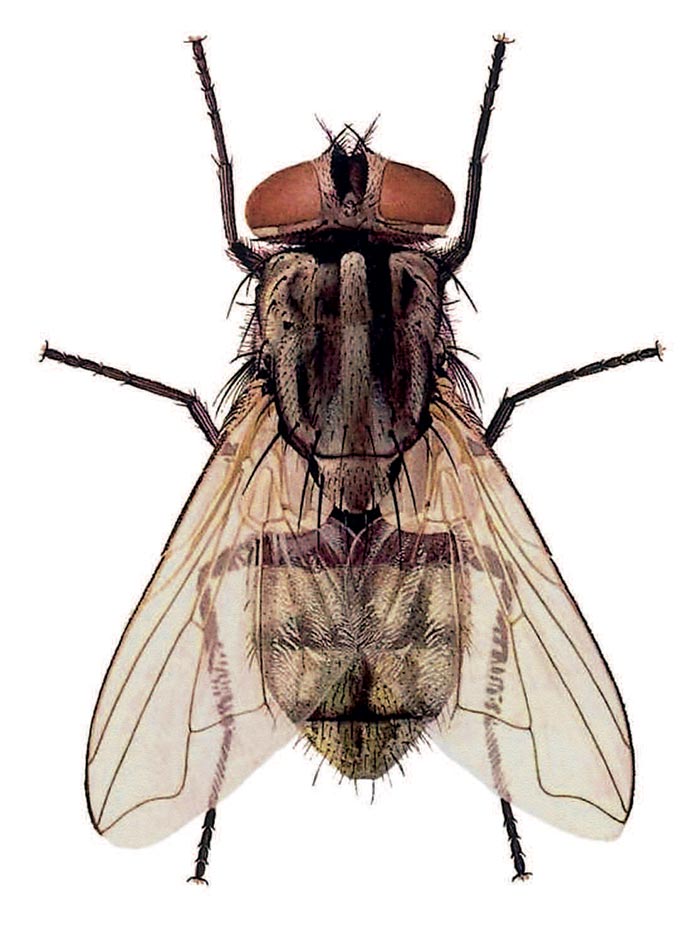
The purpose of this account is to provide an insight into the life cycles, disease transmission potentials and control of the muscid flies in the subcontinent. Identification of families, sub-families, genera and certain species can be facilitated by reference to publications by Walker,51 Greenberg13 and Zumpt.55 These publications also provide information on biology and distribution. For a visual comparison of six common species of Muscidae, see Figures 3.1 to 3.6.
Stomoxyinae
In southern Africa this subfamily is represented by eight genera, the three main livestock-associated genera being Haematobia, Haematobosca and Stomoxys.55 The adults of these flies do not feed on nectar, but are obligatory blood-suckers. They possess a mostly slender, rigid and chitinized proboscis that enables them to puncture the skin of animals to obtain a blood meal. The proboscis cannot be retracted, projects forward and can often be seen from above (Figures 3.1 and 3.3).
Haematobia and Haematobosca spp.
Common names: Horn flies, horingvlieë (Afrik.)
These are obligatory blood-sucking muscid flies that seldom leave the host except to lay eggs in recently deposited dung. Female Haematobia irritans irritans flies (Figure 3.3) have been found to average 38,4 feeds in 24 hours and males 24 feeds.16 This semi-permanently parasitic way of life ensures a food supply, and a larval habitat close at hand. It also leaves them very vulnerable to many insecticides applied to animals for the control of ticks.
Figures 3.1 to 3.4 Common synanthropic flies, namely Stomoxys calcitrans (Figure 3.1); Fannia canicularis (Figure 3.2); Haematobia irritans (Figure 3.3); and Muscina stabulans (Figure 3.4). Their size, type of mouthparts and shape of the fourth wing vein M1+2 are important diagnostic characters. (Illustrations by F. Gregor in Greenberg, Bernard; Flies and Disease, Volume 1. Copyright 8 1971 PUP. Reprinted by permission of Princeton University Press)
Three species of Haematobia have been recorded from southern Africa, namely H. thirouxi potans, H. meridiana and H. spinigera, all of which are primarily associated with game, especially African buffaloes (Syncerus caffer).55 In 1937/38 H. thirouxi potans was reported to be a problem on cattle along the coast from the Eastern Cape Province to southern KwaZulu-Natal.9, 55 The problem disappeared after synthetic organo-chlorine insecticides were used at the end of the Second World War.18 Today, horn flies have again become a pest on farms, either where non-insecticidal tickdetaching agents are used for tick control, or where no tick control is practised.
In southern Africa no Haematobia spp. have so far been proven to be vectors of any organism that causes disease in game or livestock. In many other parts of the world Haematobia spp. are biological vectors of Stephanofilaria spp.17 The presence of stephanofilariasis in cattle has not yet been definitely confirmed in southern Africa, but in the 1980s and early 1990s stephanofilariasis-like lesions were seen in cattle in the KwaZulu-Natal Lowveld and Venda in the Limpopo Province of South Africa, and in Swaziland,31 where they occurred in association with large numbers of H. meridiana.37 Haematobia thirouxi potans is found in large numbers on African buffalo in South Africa. Since buffalo are carriers of the foot-and-mouth disease virus, the possibility exists that this horn fly could transmit the virus mechanically from buffalo to nearby cattle. Two attempts at the Onderstepoort Veterinary Institute to transmit the virus by this means from infected to susceptible cattle (after two hours) were unsuccessful.48
Figures 3.5 to 3.6 Two species of the genus Musca, namely M. domestica (Figure 3.5); and M. sorbens (Figure 3.6). Note the similar shape of the fourth wing vein M1+2, which is diagnostic for this genus. Also compare this vein with those of the four other genera of synanthropic flies depicted in Figures 3.1 to 3.4 (Illustrations by F. Gregor in Greenberg, Bernard; Flies and Disease, Volume 1. Copyright 8 1971 PUP. Reprinted by permission of Princeton University Press)
A second genus of horn fly, Haematobosca, was listed by most early authors under Haematobia or Bdellolarynx.55 Four species of Haematobosca have been recorded from southern Africa, namely H. uniseriata, H. latifrons, H. angustifrons and H. zuluensis.42 Only the first two species are common.55 Haematobosca uniseriata was first recorded in the then Transvaal Province (now comprising Limpopo, Gauteng, North West and Mpumalanga provincies) in 1933, when Nieschulz and Du Toit38 found it feeding in relatively large numbers on the belly and legs of horses near the Onderstepoort Veterinary Institute. Zumpt55 caught specimens on a freshly killed warthog (Phacochoerus aethiopicus). This fly was also collected off cattle on farms in the Kuruman area in the eastern part of the Northern Cape Province during an unusually wet year (1974). In subsequent drier years it was present in much lower numbers.29
Stomoxys spp.
Common names: Stable flies, stalvlieë (Afrik.)
Eight species of Stomoxys have been recorded in southern Africa42 with S. calcitrans (Figure 3.1) being the major pest species. In South Africa Stomoxys niger (= S. nigra) is seldom collected, although it can, on occasion, occur in pest proportions in the wetter and hotter parts such as the Lowveld of Mpumulanga and north-eastern KwaZulu-Natal.31 By contrast, S. niger was rated in Mauritius to be a serious pest of cattle and deer while S. calcitrans was considered to be of negligible importance.27
Stable flies are compulsory blood-feeders, but they differ from horn flies in that they only spend short periods feeding on their hosts, digesting their meals while resting on nearby surfaces. They feed on average twice a day,16 usually on whichever host happens to be available at the time. If interrupted they will feed more often. This behaviour makes them ideal mechanical transmitters of any disease organisms present in the blood of their host.
Worldwide there are numerous examples where Stomoxys spp. (particularly S. calcitrans) have been shown to be either involved in, or suspected of, mechanically transmitting disease organisms such as Brucella spp., Borrelia spp. and equine infectious anaemia.55 In Britain it has been proved experimentally that S. calcitrans can act as an efficient mechanical vector of capripox and African swine fever viruses.25 It is also believed that this fly may be capable of mechanically transmitting foot-and-mouth disease virus. These findings are particularly significant for southern Africa where lumpy skin disease, African swine fever and foot-andmouth disease are endemic. In South Africa it has been shown experimentally that S. calcitrans can mechanically transmit Anaplasma marginale within a group of cattle,43 and that it can, in the field, act as a mechanical vector of Besnoitia besnoiti, the cause of bovine besnoitiosis or elephant skin disease. 2 These flies are therefore a very real means of disease transmission between animals in a herd or feedlot.
Members of the Stomoxyinae differ widely in their breeding requirements. Haematobia and Haematobosca spp. breed in freshly dropped dung from animals grazing in the veld, whereas S. calcitrans prefers fermenting organic material, such as rotting heaps of discarded animal feed or bedding, that may or may not contain dung. In 1997, at the Johannesburg Zoo in South Africa, the source of a severe S. calcitrans problem was found to be rotting kikuyu (Pennisetum clandestinum) lawn clippings lying in windrows on the lawns in the public area and adjacent Zoo Lake Park. For these reasons S. calcitrans is usually associated with intensive farming and suburban situations, while Haematobia and Haematobosca spp. are more commonly found where cattle are free-ranging.
While control of horn flies is relatively easy where facilities exist for treating livestock with insecticides, Stomoxys control is more difficult and must rely on a combination of farm hygiene and treatment with residual insecticides of the resting places of the adult flies and the animals themselves. A variation on some of these approaches to control utilizes the preference of adult stable flies to rest on certain surfaces.
It was found that translucent white fibreglass panels, tradenamed ‘Alsynite’, could be used to construct an attractive sticky sampling trap for these flies.54 If treated with a pyrethroid this device could also be used to control them,24 but the value of this approach in their control has not yet been investigated in southern Africa. Traps baited with synthetic odour attractants, developed to lure and catch tsetse flies, also attract stable flies (and flies belonging to the Tabanidae). These traps are predominantly bright blue (pthalogen blue) and may be made of cloth, e.g. Nzi26 and H traps,20 or of hardboard painted with a highly sticky substance such as polybutene or ‘Temoocid’.21
Fanniinae
The main nuisance fly in this subfamily is Fannia canicularis (Figure 3.2), the so-called lesser house fly. It is often confused with the house fly (Musca domestica) but is slightly smaller and more slender in build. It is a cosmopolitan fly associated primarily with intensive poultry production, where it breeds in the litter. Its sheer numbers make it a nuisance and it often infests neighbouring properties. These flies are commonly seen in daytime swarms circling in midair between cage aisles on poultry ranches, in work alleys in milking parlours, and under trees or other sheltered places. Unlike house flies, Fannia are less important as household pests or disease vectors. 23 In high-density poultry houses in South Africa the main control approach for it, as well as for the house fly, is the addition of a cyromazine-based additive to the feed. Cyromazine inhibits the formation of chitin so that development to the following larval stage is prevented.31
Muscinae
Although this subfamily contains 18 genera,42 only species of the genus Musca are commonly found on livestock in southern Africa. However, low numbers of other genera such as Muscina and Morellia may sometimes be collected off livestock.
Muscina stabulans
Common name: False stable fly
The false stable fly (Figure 3.4) is also often mistaken for the house fly because of its similarity in colour, size, and food preference, and its habit of entering homes. In appearance it falls in between Stomoxys and Musca in that its wing venation is similar to Stomoxys but mouthparts resemble those of M. domestica. It feeds on and deposits its eggs in a wide variety of animal excrement (poultry, horse, cow, domestic pets ) and decaying vegetable matter.23 Its larvae have been known to prey on those of other flies.13
Morellia spp.
Common name: Sweat flies
These flies have been collected off cattle where they scratch the skin surface and lap up exuding blood. In South Africa they have not been found in high numbers. They are also of interest because, like M. stabulans, their larvae prey on other larvae in the dung in which they develop.
Hydrotaea spp.
Common names: Sheep head flies; plantation flies (Hydrotaea irritans)
Although species of Hydrotaea have not yet been recorded as pests in southern Africa, certain species elsewhere in the world have warranted serious attention. In Europe, especially the Netherlands, Denmark, Germany, Belgium and the UK, Hydrotaea irritans is regarded as the mechanical vector of summer mastitis.45 In Czechoslovakia, H. armipes is suspected of being a mechanical vector of infectious bovine keratoconjunctivitis.8
Musca spp.
Common names: House flies, huisvlieë (Afrik.); face flies, gesigsvlieë (Afrik.)
Except for the cosmopolitan M. domestica (Figure 3.5), the veterinary importance of other species of the genus Musca has been largely overlooked in southern Africa, and there has been a tendency to attribute problems caused by them to M. domestica. In the 1970s and 1980s, however, various studies in South Africa on the transmission of the filarial worm, Parafilaria bovicola, in the Limpopo Province Bushveld,30 a fly problem in the Northern Cape Province,34 the transmission of bovine malignant catarrhal fever in the Limpopo Province Bushveld,35 and in Namibia on the epidemiology of anaplasmosis in cattle,32 have led to an increased interest in the Musca spp. associated with cattle and, to a lesser extent, wild-living game animals (Table 3.1). Six Musca spp. (Table 3.1) were common to most of the farming situations examined, namely M. xanthomelas, M. lusoria, M. domestica, M. confiscata (= M. fasciata), M. nevilli and M. sorbens (Figure 3.6). The presence of certain other specieswasdependentonthe geographical area, the season and climatological factors. It was only near homesteads that the synanthropic M. domestica and M. sorbens were important. In the veld, away from the homesteads, there is an interaction between various symbovine Musca spp., and the dominant species change according to both local and the broader climatic factors. With the exception of M. domestica which breeds in dung and decaying organic matter and refuse, all the other Musca spp. mentioned above breed in fresh dung in the veld and their larvae all have to feed and survive in this medium. Over thousands of years, evolution has resulted in the development of a variety of species, each with its own preferences for food, temperature, moisture and other requirements.
Twenty-five Musca spp. have so far been recorded in southern Africa.22, 42 They belong to the subgenera Eumusca, Byomya, Musca, Philaematomyia, Viviparomusca and Lissosterna.42 The 12 Musca spp. listed in Table 3.1 belong to the first four subgenera. Only five species of the last two subgenera have been recorded in southern Africa42 and these have seldom, if ever, been collected in South Africa.31 The observations on Musca spp. below are therefore restricted to 13 South African species.
Subgenus Eumusca
Common names: African face flies, Afrika gesigsvlieë (Afrik.)
Musca xanthomelas is often the commonest African face fly associated with cattle in the veld in South Africa. It is found throughout the year in the Limpopo Province Bushveld but is less common in the South African Highveld. It lays an average of four batches of 33 eggs each and may survive for up to 35 days at 27 °C.33 Musca xanthomelas has only once been collected south of latitude 29°.28
Musca lusoria is the largest of the African face flies. It iscommon throughout the year in the Limpopo Province Bushveld, but, as is the case with M. xanthomelas, is also less common on the Highveld. The female is ovoviviparous, depositing one late first stage larva at a time on fresh dung, and is able to produce up to 27 larvae in her life-time of up to 75 days.33
Musca nevilli closely resembles M. xanthomelas in appearance but produces fewer eggs.22, 33 It differs greatly, however, in its seasonal prevalence, being common only from late January to April.
Musca aethiops is rarely collected off cattle, but has been reared from the dung of both cattle and African buffalo.
Musca munroi was never collected in the surveys summarized in Table 3.1. On occasion it is quite common on cattle farms in parts of KwaZulu-Natal and the Eastern Cape provinces of South Africa.
Subgenus Byomya
Musca confiscata (= M. fasciata) is a very versatile species in that it may be found in reasonable numbers in both the Bushveld and Highveld areas of South Africa. It breeds in both cattle and wildebeest (Connochaetes spp.) dung, and feeds on cattle and presumably also on game. Its ability to utilize various situations would therefore well qualify this fly to act as a mechanical transmitter of disease organisms.
Musca sorbens (Figure 3.6), like M. confiscata, may be found throughout the year in the Limpopo Province Bushveld but, like M. domestica, it is associated with human habitations and does not breed in animal dung in the veld.
Two forms of M. sorbens have been recognized in southern Africa.41 Both are attracted to fresh human faeces, which appears to be their preferred breeding medium.41
Musca tempestatum is only encountered in the vicinity of wildebeest, in whose dung it prefers to breed.
Musca freedmani is a lesser-known fly whose preferences have not as yet been observed.
Musca lasiophthalma is occasionally collected in the Bushveld in winter, but has been collected in large numbers on the Highveld in both winter and early spring.
Musca conducens has been reared from cattle dung samples collected in the Bushveld but has seldom been collected off cattle. It is possible that the fly identified as M. conducens in southern Africa is different from the nominate species, which is found in Asia.40
Subgenus Musca
Musca domestica is divided into three subspecies in the Afrotropical Region.42 Two of these subspecies are indigenous to Africa, including southern Africa. These are M. d. calleva (an outdoor fly) and M. d. curviforceps (more domestic in habits). Hybrids may also be found.39 During the writer’s studies in the Bushveld, M. domestica was never identified to subspecies level. It was common near homestead kraals in the first half of summer but was seldom reared from cattle dung samples collected in the veld.
Subgenus Philaematomyia
Musca crassirostris is a ‘veld-fly’, apparently common only in dry years.
Feeding habits of Musca spp.
All Musca spp. require water, sugars and protein in order to survive and reproduce. Flies that live in the veld obtain these basic requirements from fresh dung, eye and nasal secretions, wounds and sores on animals, carrion, plant and flower secretions, and the excretions of sap-sucking insects. 33, 49 Protein is essential for the development of the ovaries of the flies. Protein hunger in female face flies (M. autumnalis) has been directly related to the stage of oogenesis and is significantly diminished sometime before the end of each gonotrophic cycle.50 All Musca spp. possess a sponge-like lapping mouth-part or labellum made up of numerous medially directed channels or pseudotracheae which radiate out from a central oral opening. Fluid is sucked up along these pseudotracheae into a central cavity and then via the labial-gutter epipharyngeal tube into the digestive system.10 Excess fluid is stored in a large sac-like crop from which it is either passed to the mid-gut for digestion or regurgitated by the fly if more attractive food is found. This fluid is also used to dissolve or dilute food that is too coarse or viscous to take up through the pseudotracheae. 53 The fly mixes the regurgitated fluid with the food, often using its prestomal teeth for the purpose.
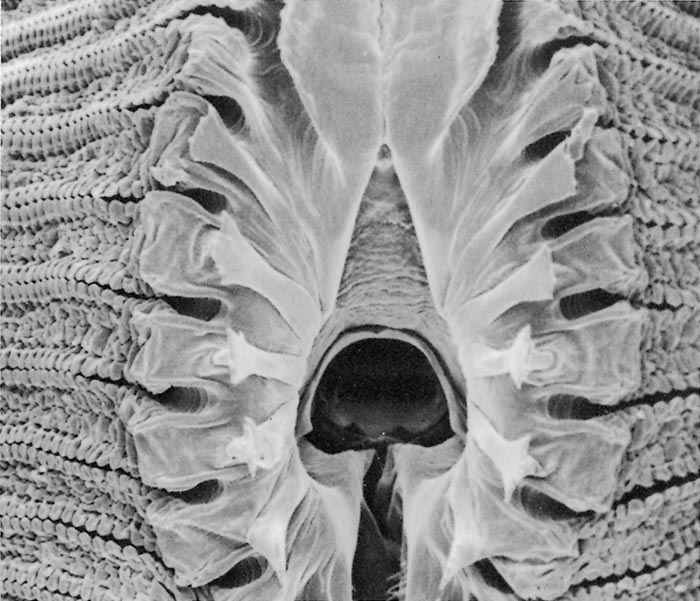
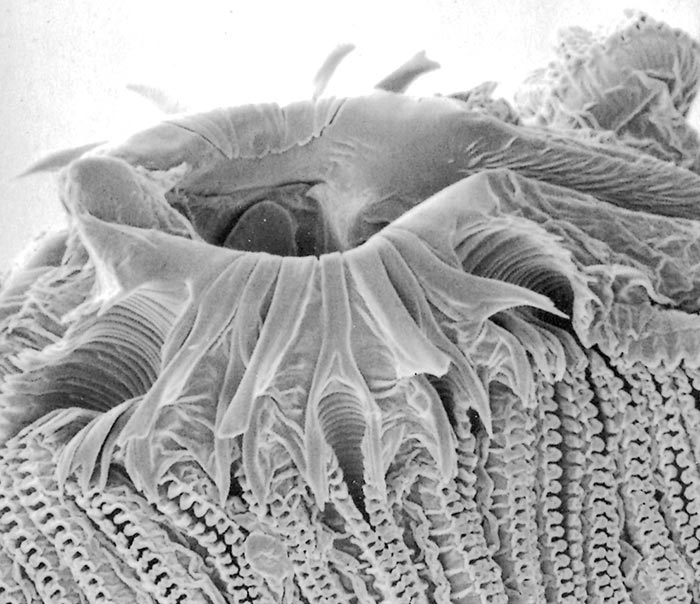
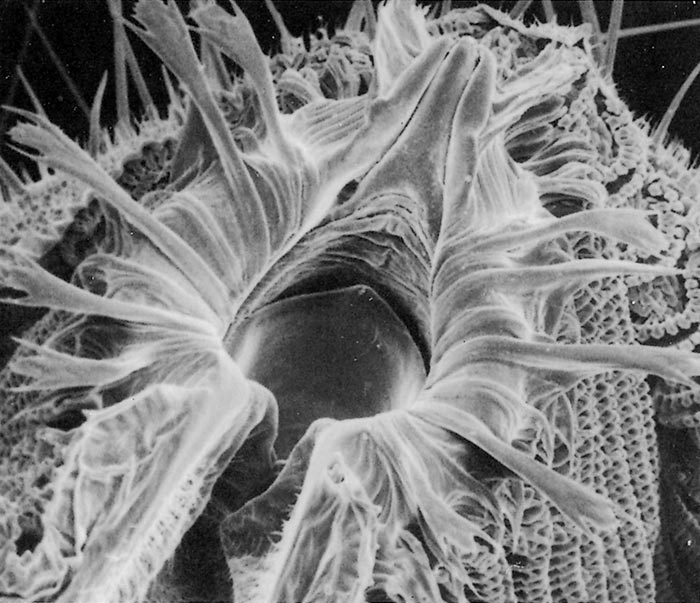
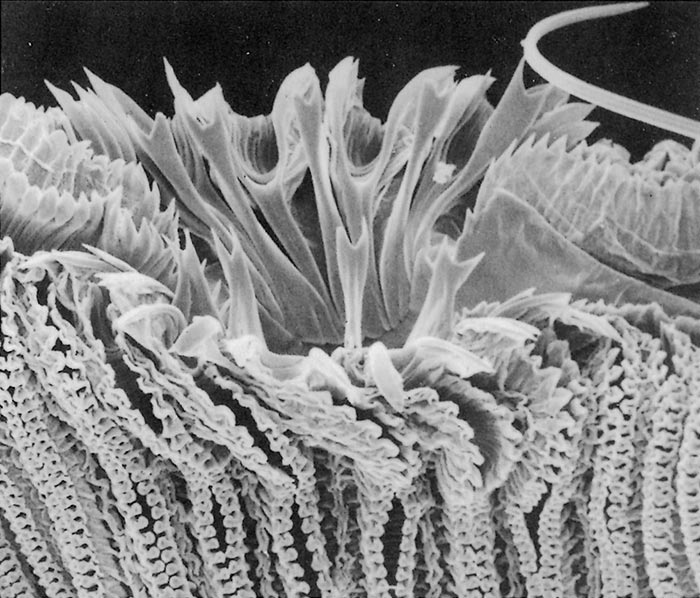
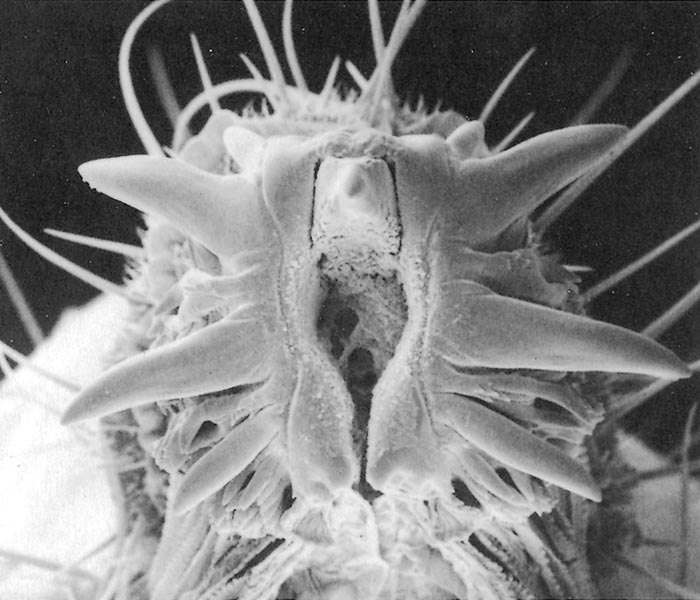
All Musca spp. have one or more circles of prestomal teeth. They are seldom visible as they are only extruded during feeding, and are otherwise hidden by the enveloping labellar lobes of the mouth-parts. The first row of these prestomal teeth ranges in size and form according to the species. For example, short, relatively blunt teeth are present in the house fly (M. domestica) and an African face fly (M. xanthomelas);4 longer sharper teeth in the face fly M. autumnalis, which occurs in Europe and North America and two African face flies (M. nevilli and M. lusoria); and large coarse teeth in M. crassirostris10 (Figure 3.7 to 3.12).
Table 3.1 The relative abundance of various Musca spp. collected off cattle and reared from the dung of cattle and blue wildebeest (Connochaetes taurinus taurinus) in southern Africa
| COLLECTED OFF CATTLE (%) | DUNG-REARED (%) | |||||||
|---|---|---|---|---|---|---|---|---|
| MUSCA SPECIES | RSA* N. CAPE | NAMIBIA | RSA* HIGHVELD | RSA* BUSHVELD | RSA* BUSHVELD | |||
| KURUMAN DISTRICT 1973 | WINDHOEK DISTRICT 1978-1980 | PRETORIA DISTRICT 1974 | LYDENBURG DISTRICT 1974 | VARIOUS DISTRICTS 1973-1976 | THABAZIMBI DISTRICT 1987 | THABAZIMBI DISTRICT | ||
| CATTLE 1987 | WILDEBEEST 1987 | |||||||
| M. lusoria | 11,4 | 25,3 | 6,6 | 4,1 | 45,7 | 12,6 | 14,4 | 1,6 |
| M. xanthomelas | 28,2 | 52,8 | 0,7 | 2,1 | 9,3 | 27,8 | 44,9 | 0,4 |
| M. nevilli | 3,3 | 1,4 | 0,2 | 10,3 | 15,9 | 4,9 | 1,8 | |
| M. domestica | 18,1 | 5,8 | 23,4 | 26,2 | 18,8 | 32,5 | 0,6 | 0,1 |
| M. confiscata** | 6,2 | 12,0 | 7,5 | 36,4 | 7,3 | 7,1 | 12,0 | 41,9 |
| M. sorbens | 2,3 | 0,1 | 1,6 | 14,4 | 7,1 | 3,1 | ||
| M. crassirostris | 20,4 | 0,7 | 1,0 | 0,2 | 9,2 | |||
| M. tempestatum | 0,4 | 0,6 | 51,6 | |||||
| M. freedmani | 8,8 | 0,5 | 0,1 | 0,2 | 0,3 | 0,1 | ||
| M. lasiophthalma | 1,2 | 1,3 | 60,0 | 16,9 | 0,3 | |||
| M. aethiops | 0,1 | 1,6 | ||||||
| M. conducens | 0,2 | 0,2 | 11,6 | 2,5 | ||||
| Fly totals | 3 262 | 18 000 | 889 | 195 | 20 045 | 2 380 | 2 631 | 1 140 |
* Republic of South Africa
** According to Pont42 M. confiscata is the replacement name for M. fasciata
The feeding habits of flies differ according to the type of prestomal teeth present. Musca domestica and M. xanthomelas (Figure 3.7 and 3.8), feed mainly on the lachrymal secretions and wounds already present on an animal; M. autumnalis (Figure 3.9 feeds directly on conjunctival tissue5, 44 and M. lusoria (the southern African counterpart of M. autumnalis) (Figure 3.10)10 may be expected to feed in the same manner. It has also been shown that M. nevilli (Figure 3.11) has prestomal teeth almost identical to those of M. autumnalis (Figure 3.9).6 Musca crassirostris seldom visits the eyes or wounds on cattle, but crawls under the hair of animals to rasp away the skin surface with its large, coarse teeth until blood is drawn, on which it will engorge (Figure 3.12). As an obligatory blood-feeder, M. crassirostris is therefore the Musca equivalent of Stomoxys. Musca conducens has a similar feeding mechanism, although the mouth-parts are not as heavily developed as are those in M. crassirostris.
Disease transmission potential of the Muscinae
Very little is known about the transmission of infectious agents by Musca spp. in southern Africa; that which has been recorded is based largely on circumstantial evidence. Musca confiscata (= M. fasciata) has been suspected of mechanically transmitting lumpy skin disease virus.52 Also, during an outbreak of enzootic abortion in sheep in the Northern Cape Province in 1975, a Chlamydophila sp. was isolated from Musca spp. collected around sheep.19
In the search for the vectors of Parafilaria bovicola,30 20 000 specimens of 11 Musca spp. were collected in the Bushveld of South Africa and identified (Table 3.1). Of these, M. lusoria, M. xanthomelas and M. nevilli, all belonging to the subgenus Eumusca, were shown to be vectors. These flies accounted for 65,3 per cent of all the specimens collected. As African face flies breed in fresh dung deposited in the veld, it is only in these situations that they occur in large enough numbers to act as vectors of the larvae of this worm. Face flies are unable to develop in the dung of cattle which have been fed a supplementary grain diet, as this dung ferments; it is, however, highly suitable for this purpose for house flies. This previously unsuspected predominance of African face flies in the veld has stimulated thinking about the possible involvement of these and other Musca spp. in the transmission of infectious agents, not only between cattle but also between carrier game animals and cattle in southern Africa.
One of the diseases most likely to be transmitted mechanically by flies in the veld is infectious keratoconjunctivitis (‘pinkeye’), which may be caused by a number of different organisms, including Moraxella bovis, Mycoplasma spp., Chlamydophila sp. or rickettsiae (see Moraxella spp. infections). No work has yet been done in southern Africa on the transmission of any of these organisms, but detailed studies have been conducted in the USA on the transmission of M. bovis by the face fly, M. autumnalis. 15 The North American studies proved conclusively that the damage done to the eye surface by the prestomal teeth of M. autumnalis while feeding predisposes the eyes of cattle to infection.44 In addition, experimental studies have shown that M. bovis may be present in material regurgitated from the crops of face flies, and that infection of the eyes of cattle may follow exposure to infected flies.1, 12 Furthermore, a higher prevalence of ‘pinkeye’ in the field has been related to face fly numbers.11 The African face flies fall into the same subgenus as M. autumnalis, and have similar feeding habits and prestomal teeth (especially M. lusoria and M. nevilli). They may therefore transmit M. bovis in the same manner.
In South Africa investigations have been conducted into the possible role of Musca spp. in the field transmission of anaplasmosis, foot-and-mouth disease and malignant catarrhal fever. Unfortunately none of these studies has been sufficiently intensive to prove or disprove the vectorial role of these flies.
The control of flies and disease
Under intensive conditions
A multi-faceted approach is necessary for the control of so-called filth flies, the most common of which is the house fly (M. domestica). This should include good management, the judicious use of pesticides, and the encouragement of parasites and predators.
Good management practices are essential to reduce and eliminate conditions that favour fly development.23 These should include:
- a system of manure management focusing on its collection, processing, storage and utilization as a fertilizer or compost,
- proper water management, including supply, drainage, and disposal systems, taking into account above-normal rainfall in the area concerned, and
- general farm sanitation involving the routine disposal of other animal wastes, spilt feed, organic debris, and even household garbage.23
Although certain registered insecticides may be applied directly to livestock, insecticides are best used as residual sprays on resting surfaces and incorporated in attractive baits in feedlots and dairies. In this way only resting and feeding adult flies will be killed and not the wasp parasites searching the breeding medium for fly pupae to parasitize nor other arthropod predators in search of eggs and larvae on which to feed. Certain poultry feed additives such as cyromazine, which inhibits the moulting of fly larvae, are harmless to non-dipteran parasites and predators.
Cyromazine is also available in a formulation that can be applied directly to fly-breeding material.31
Under extensive conditions
Field studies indicate that most fly species which breed in the veld seek out the nearest host animals and do not fly far, unless the animals are removed from the area. Preliminary proof of this has come from insecticidal trials against African face flies in the Limpopo Province Bushveld of South Africa.7 A marked fly reduction could be maintained in an insecticide-treated herd of cattle while the control herd, only 3 km away, had a high incidence of flies, indicating that these flies had not come into contact with the treated animals.
Reduction of fly numbers on cattle in the veld should therefore lead to a reduction in the transmission of certain infective organisms. This has been clearly demonstrated by Nevill et al.,36 who drastically reduced the prevalence of P. bovicola infestation in cattle by weekly to fortnightly dipping with a pyrethroid insecticide. An easier method of face fly control is to attach pyrethroid-impregnated tags to both ears of an animal. In this way, in one British study, a reduction of up to 90 per cent in face fly numbers was achieved during a two- to three-month period, and infectious keratoconjunctivitis was almost completely eliminated. 47 This principle of fly reduction should be applied in situations where losses due to fly-transmitted diseases warrant the effort and expenditure. In South Africa no impregnated ear-tags are registered for fly control in livestock. This is because treatment is aimed primarily at ticks, for which ear-tags are unsuitable. However, pour-on pyrethroids, which are often effective for fly control, have become popular.31
Figures 3.7 to 3.12 Scanning electron micrographs of the prestomal teeth of six Musca spp. namely M. domestica (Figure 3.7), M. xanthomelas (Figure 3.8), M. autumnalis (Figure 3.9), M. lusoria (Figure 3.10), M. nevilli (Figure 3.11) and M. crassirostris (Figure 3.12). (By courtesy of Drs A.B. Broce and R.J. Elzinga, Kansas State University, USA)
References
- ARENDS, J.J., WRIGHT, R.E., BARTO, P.B. & LUSBY, K.S., 1984. Transmission of Moraxella bovis from blood agar cultures to Hereford cattle by face flies (Diptera: Muscidae). Journal of Economic Entomology, 77, 394–398.
- BIGALKE, R.D., 1968. New concepts on the epidemiological features of bovine besnoitiosis as determined by laboratory and field investigations. Onderstepoort Journal of Veterinary Research, 35, 3–138.
- BORNEMISSZA, G.F., 1979. The Australian Dung Beetle Research Unit in Pretoria. South African Journal of Science, 75, 257–260.
- BROCE, A.B., 1988. Kansas State University, USA, personal communication.
- BROCE, A.B. & ELZINGA, R.J., 1984. Comparison of prestomal teeth in the face fly (Musca autumnalis) and the house fly (Musca domestica) (Diptera: Muscidae). Journal of Medical Entomology, 21, 82–85.
- BROCE, A.B. & ELZINGA, R.J., 1988. Kansas State University, USA, personal communication.
- catton, d.g., 1984. Cyhalothrin, a new synthetic pyrethroid—efficacy established against African face flies (subgenus Eumusca) in South Africa. Proceedings of the 13th World Congress on Diseases of Cattle, Durban, 1984, 442–447.
- DUSBABEK, F., SOUKUPOVA, A., GREGOR, F. & KREJCI, J., 1982. The role of Hydrotaea armipes Fall. (Diptera: Muscidae) in the transmission of infectious bovine keratoconjunctivitis. Folia Parasitologica, (Praha), 29, 79–83.
- DU TOIT, R., 1938. The horn fly (Lyperosia minuta) in South Africa. Journal of the South African Veterinary Medical Association, 9, 136–143.
- ELZINGA, R.J. & BROCE, A.B., 1986. Labellar modifications of muscomorpha flies (Diptera). Annals of the Entomological Society of America, 79, 150–209.
- GERHARDT, R.R., ALLEN, J.W., GREENE, W.H. & SMITH, P.C., 1982. The role of face flies in an episode of infectious bovine keratoconjunctivitis. Journal of the American Veterinary Medical Association, 180, 156–159.
- GLASS, H.W. & GERHARDT, R.R., 1984. Transmission of Moraxella bovis by regurgitation from the crop of the face fly (Diptera: Muscidae). Journal of Economic Entomology, 77, 399–401.
- GREENBERG, B., 1971. Flies and Disease. Vol. I. Ecology, Classification, and Biotic Associations. Princeton, New Jersey: Princeton University Press.
- GREENBERG, B., 1973. Flies and Disease. Vol. II. Biology and Disease Transmission. Princeton, New Jersey: Princeton University Press.
- HALL, R.D., 1984. Relationship of the face fly (Diptera: Muscidae) to pinkeye in cattle: A review and synthesis of the relevant literature. Journal of Medical Entomology, 21, 361–365.
- HARRIS, R.L., MILLER, J.A. & FRAZAR, E.D., 1974. Horn flies and stable flies: Feeding activity. Annals of the Entomological Society of America, 67, 891–894.
- HIBLER, C.P., 1966. Development of Stephanofilaria stilesi in the horn fly. Journal of Parasitology, 52, 890–898.
- HOWELL, C.J., 1975. Veterinary entomology in southern Africa. Entomology Memoir, No. 44, Department of Agricultural Technical Services, Republic of South Africa.
- HOWELL, C.J., 1975. Onderstepoort Veterinary Institute, South Africa. Unpublished data.
- KAPPMEIER , K., 2000. A newly developed odour-baited ‘H trap’ for the live collection of Glossina brevipalpis and G. austeni (Diptera: Glossinidae) in South Africa. Onderstepoort Journal of Veterinary Research, 67, 15–26.
- KAPPMEIER-GREEN, K., VENTER, G.J. & NEVILL, E.M., 2001. The evaluation of sticky traps and their improvement as a monitoring tool for Glossina austeni and G. brevipalpis (Diptera: Glossinidae) in South Africa. Unpublished data.
- KLEYNHANS, K.P.N., 1987. Musca nevilli sp. nov. (Diptera: Muscidae), a dung-breeding fly from South Africa. Onderstepoort Journal of Veterinary Research, 54, 115–118.
- LOOMIS, E.C., 1982. Common External Parasites and Pests of Livestock and Poultry in California. University of California, Davis: Davis, California.
- MEIFERT, D.W., PATTERSON, R.S., WHITFIELD, T., LA-BRECQUE, G.C. & WEIDHAAS, D.E., 1978. Unique attractant-toxicant system to control stable fly populations. Journal of Economic Entomology, 71, 290–292.
- MELLOR, P.S., KITCHING, R.P. & WILKINSON, P.J., 1987. Mechanical transmission of capripox virus and African swine fever virus by Stomoxys calcitrans. Research in Veterinary Science, 43, 109–112.
- MIHOK, S. 2001. The NZI Trap—a simple environment-friendly cloth trap for biting flies that does not use insecticides. Available from: URL: http://informatics.icipe.org/ nzi/index.htm (Hosted by the International Centre of Insect Physiology and Ecology, Nairobi, Kenya.)
- MONTY, J., 1972. A review of the stable fly problem in Mauritius. la Revue Agricole et Sucriére de l’Ile Maurice, 51, 13–29.
- NEVILL, E.M., 1980. Studies on Parafilaria bovicola Tubangui in South Africa with particular reference to the role played by insects in its transmission and distribution. D.Sc. thesis, University of Pretoria.
- NEVILL, E.M., 1982. Flies of veterinary importance on cattle. Proceedings of Symposium on ectoparasites of cattle, 15–16 March, 1982, Pretoria. pp. 86–90.
- NEVILL, E.M., 1985. The epidemiology of Parafilaria bovicola in the Transvaal Bushveld of South Africa. Onderstepoort Journal of Veterinary Research, 52, 261–267.
- NEVILL, E.M., 2001. Onderstepoort Veterinary Institute, South Africa, unpublished data.
- NEVILL, E.M., BIGGS, H.C. & LANGENHOVEN, J.W., 1980. Onderstepoort Veterinary Institute, South Africa, unpublished data.
- NEVILL, E.M. & SUTHERLAND, B., 1987. The colonization and life-cycles of Musca lusoria, Musca xanthomelas and Musca nevilli, vectors of Parafilaria bovicola in South Africa. Onderstepoort Journal of Veterinary Research, 54, 607–611.
- NEVILL, E.M. & SWART, J.J., 1977. Onderstepoort Veterinary Institute, South Africa, unpublished data.
- NEVILL, E.M. & VENTER, G.J., 1988. Onderstepoort Veterinary Institute, South Africa, unpublished data.
- NEVILL, E.M., WILKINS, C.A. & ZAKRISSON, G., 1987. The control of Parafilaria bovicola transmission in South Africa. Onderstepoort Journal of Veterinary Research, 54, 547–550.
- NEWSHOLME, S.J., VERSTER, A.J.M. & JACOBS, J.C., 1983. Bovine skin lesions of possible filarial origin associated with heavy horn fly infestations (Haematobia meridiana). Onderstepoort Journal of Veterinary Research, 50, 73–75.
- NIESCHULZ, O. & DU TOIT, R., 1933. The occurrence of Bdellolarynx uniseriatus Malloch, a blood-sucking fly, in the Transvaal. Journal of the South African Veterinary Medical Association, 4, 221–222.
- PATERSON, H.E., 1964. Population genetic studies in areas of overlap of two subspecies of Musca domestica L. In: davis, d.h.s., (ed.). Ecological Studies in Southern Africa. Monographiae Biology. The Hague: W. Junk.
- PATERSON, H.E., 1988. University of Queensland, Australia, personal communication.
- PATERSON, H.E. & NORRIS, K.R., 1970. The Musca sorbens complex: The relative status of the Australian and two African populations. Australian Journal of Zoology, 18, 231–245.
- PONT, A.C., 1980. Family Muscidae. In: CROSSKEY, R.W., (ed.). Catalogue of the Diptera of the Afrotropical Region. London: British Museum (Natural History).
- POTGIETER, F.T., SUTHERLAND, B. & BIGGS, H.C., 1981. Attempts to transmit Anaplasma marginale with Hippobosca rufipes and Stomoxys calcitrans. Onderstepoort Journal of Veterinary Research, 48, 119–122.
- SHUGART, J.I., CAMPBELL, J.B., HUDSON, D.B., HIBBS, C.M., WHITE, R.G. & CLANTON, D.C., 1979. Ability of the face fly to cause damage to eyes of cattle. Journal of Economic Entomology, 72, 633–635.
- SOL, J., 1984. Control methods in summer mastitis: The importance of fly control. Proceedings of the 13th World Congress on Diseases of Cattle, Durban, 1984, 236–242.
- STEELMAN, C.D., 1976. Effects of external and internal arthropod parasites on domestic livestock production. Annual Review of Entomology, 21, 155–178.
- TARRY, D.W., 1985. Cattle fly control using controlled-release insecticides. Veterinary Parasitology, 18, 229–234.
- THOMSON, G.R., DOUBE, B.M., BRAACK, L.E.O., GAINARU, M.D. & BENGIS, R.G., 1988. Failure of Haematobia thirouxi potans (Bezzi) to transmit foot-and-mouth disease virus mechanically between viraemic and susceptible cattle. Onderstepoort Journal of Veterinary Research, 55, 121–122.
- VAN GEEM, T. & BROCE, A.B., 1985. Significance of cattle discharges and secretions as protein sources for ovarian development in the face fly (Diptera: Muscidae). Environmental Entomology, 14, 60–64.
- VAN GEEM, T. & BROCE, A.B., 1986. Fluctuations in the protein and carbohydrate content of the crop related to periodicities in ovarian development of the female face fly (Diptera: Muscidae). Annals of the Entomological Society of America, 79, 1–6.
- WALKER, A., 1994. The arthropods of humans and domestic animals – a guide to preliminary identification. London: Chapman & Hall.
- WEISS, K.E., 1968. Lumpy skin disease virus. Virology Monographs, 3, 111–131.
- WEST, L.S., 1951. The Housefly. Ithaca, New York: Comstock Publishing Company Inc.
- WILLIAMS, D.F., 1973. Sticky traps for sampling populations of Stomoxys calcitrans. Journal of Economic Entomology, 66, 1279–1280.
- ZUMPT, F., 1973. The Stomoxyine Biting Flies of the World. Stuttgart: Gustav Fischer Verlag.