- Infectious Diseases of Livestock
- Part 1
- The control of infectious diseases of livestock: Making appropriate decisions in different epidemiological and socioeconomic conditions
- Vectors: Ticks
- Vectors: Tsetse flies
- Vectors: Muscidae
- Vectors: Tabanidae
- Vectors: Culicoides spp.
- Vectors: Mosquitoes
- Classification, epidemiology and control of arthropod-borne viruses
- Special factors affecting the control of livestock diseases in sub-Saharan Africa
- The control of infectious diseases of livestock: Making appropriate decisions in different epidemiological and socioeconomic conditions
- Infectious diseases of animals in sub-Saharan Africa: The wildlife⁄livestock interface
- Vaccination: An approach to the control of infectious diseases
- African animal trypanosomoses
- Dourine
- Trichomonosis
- Amoebic infections
- GENERAL INTRODUCTION: COCCIDIA
- Coccidiosis
- Cryptosporidiosis
- Toxoplasmosis
- Besnoitiosis
- Sarcocystosis
- Balantidiosis
- Leishmaniosis
- Neosporosis
- Equine protozoal myeloencephalitis
- GENERAL INTRODUCTION: BABESIOSES
- Bovine babesiosis
- Equine piroplasmosis
- Porcine babesiosis
- Ovine babesiosis
- GENERAL INTRODUCTION: THEILERIOSES OF CATTLE
- East Coast fever
- Corridor disease
- Zimbabwe theileriosis
- Turning sickness
- Theileria taurotragi infection
- Theileria mutans infection
- Theileria annulata theileriosis
- Theileriosis of sheep and goats
- Theileria buffeli⁄orientalis infection
- Non-pathogenic Theileria species in cattle
- GENERAL INTRODUCTION: RICKETTSIAL, CHLAMYDIAL AND HAEMOTROPIC MYCOPLASMAL DISEASES
- Heartwater
- Lesser known rickettsial infections in animals and humans
- Chlamydiosis
- Q fever
- Eperythrozoonosis
- Bovine Haemobartonellosis
- Potomac horse fever
- GENERAL INTRODUCTION: ANAPLASMOSES
- Bovine anaplasmosis
- Ovine and caprine anaplasmosis
The control of infectious diseases of livestock: Making appropriate decisions in different epidemiological and socioeconomic conditions
This content is distributed under the following licence: Attribution-NonCommercial CC BY-NC  View Creative Commons Licence details here
View Creative Commons Licence details here
The control of infectious diseases of livestock: Making appropriate decisions in different epidemiological and socioeconomic conditions
B D PERRY, J J MCDERMOTT AND T F RANDOLPH
Introduction
This book describes the many infectious diseases that affect livestock in sub-Saharan Africa, and indeed in many other regions of the world, and discusses in detail the technical aspects of their diagnosis, treatment and control/eradication. In an environment with unlimited resources, and with the technical capacity to undertake the appropriate measures, many of the diseases described here could be brought under control or even eradicated. Indeed, in the past, a few of them have been eradicated, but usually at enormous cost. Remarkably, in many cases, these costs are not available in the records, indicating that they may not have been of prime importance in the decision to eradicate. Take for example the eradication of East Coast fever (ECF) from southern Africa. In his detailed account of the eradication programme that spanned the years 1901 to 1960, Lawrence114 estimated, from what he called ‘fragmentary data’, that a total of 1,4 million cattle died as a result of the disease, and an additional 100 000 were slaughtered. However, he noted that data on the costs of control were ‘non-existent’. One cannot help asking the question: would such an intensive control and eradication programme, involving compulsory weekly dipping of all cattle, strict movement controls, quarantine of affected herds, compulsory examination of spleen smears from all animals that were slaughtered or died, and slaughter of infected animals with compensation, all sustained over decades, be carried out if ECF were to break out in certain southern African countries today?
It is in the answering of this question that one confronts the stark interaction between the technical feasibility and the socioeconomic feasibility of infectious disease control/ eradication. There are several important differences in circumstances that would affect the nature of a decision made today. These include changed political and socioeconomic circumstances, different disease-control institutions and authorities, and different decision criteria for allocating public and private funds and other resources to infectious disease control, to name but a few.
Why would we today expect the answer to be ‘probably not’? Much has changed in the intervening years since the initiation of ECF control in southern Africa in 1904. The magnitude of the threat presented by even the most virulent of infectious diseases is different, and is probably much smaller now due to our increased understanding of the risks and impacts of different infections, and a greater inventory of measures at our disposal to control them. It is doubtful that ECF would threaten the very existence of the entire agricultural sector today as it did at the beginning of the previous century for settler agricultural development. At the time, the region was recovering from the devastation caused by the rinderpest pandemic, and in an era when the world was preoccupied by the perils of infectious diseases, South Africa was also struggling to recover from the ravages of the Anglo-Boer War (1899 to 1902).
Furthermore, it is difficult to imagine the determined application of a technical solution being successfully implemented today given the political and organizational realities of much of the developing world. Part of this doubt is related to the more complicated nature of decisions on resource allocation and how (and by whom) these decisions are made today. It appears that an extraordinary degree of authority was given to the highly successful group of scientists active in investigating the causes and control of ECF, and this outweighed many of the bigger economic questions. There have been substantial changes in the seat of authority for disease control in virtually all corners of the world, from a very centralized, powerful and autocratic government involvement to a decentralized and often impoverished public sector, and this change has accelerated in most countries of the African continent in the last few years. The change has been complicated by a deterioration in the veterinary infrastructures of many African countries, and thus the feasibility of delivering even highly technically efficacious control measures is increasingly questionable. In addition, several decisionsupport tools (such as cost-benefit analysis, for example) to aid in decision-making are now available and widely used, but did not exist in the early part of the twentieth century.
In summary, decisions regarding disease control have changed radically due to a combination of improved knowledge about infectious diseases and of the need to take into account a whole range of socioeconomic considerations that were not previously considered.
This chapter evaluates how decision-making for the control of infectious diseases can be improved at all levels (e.g. farm, community, country, region) by the consideration of both epidemiological and socioeconomic issues. We review what the impacts of diseases and their control are, how these can be quantified, how such information can be used in the choice of disease control interventions, and how such interventions can be best implemented under different sets of circumstances.
Livestock production and use in Africa
An important perspective on the socioeconomic issues affecting infectious disease control can be gained through an understanding of the concept of different production systems in which diseases have different impacts. These impacts vary in terms of the production losses and control costs incurred, and whether the control costs are borne by private or public coffers. During the colonial era, and for a variable period after depending on the country and region, there were possibly two major production systems (although such terminology was not applied to these) in much of Africa. The first of these was the ‘commercial’ sector, comprising different scales of beef, dairy, sheep and poultry production on medium-sized to large farms with fairly high levels of inputs (in terms of feed, veterinary drugs and vaccines, and genetically improved breeds), in which many of the financial aspects of disease control were the responsibility of the individual farmer, as currently occurs in Europe, for example. There was some financial support provided by the state to farmers, such as loans for the construction of dips, access to vaccines and remedies at government-subsidized prices, and public veterinary services that were generally state-financed and -run, and delivered at no or a highly subsidized cost to farmers. Socioeconomic issues in this sector very much boiled down to the cost efficacy of different approaches to successful infectious disease control.
The second production system was the peasant sector,which was often in geographically distinct areas known by names such as ‘tribal trust lands’ or ‘African reserves’, which have evolved into the so-called ‘traditional’, ‘communal’ or ‘smallholder sectors’, depending on the country and the nomenclature used. These low-producing, often subsistenceorientated systems, relied very much on the state for the provision of veterinary services, which in general were provided at no cost to farmers. This was done for many reasons, both political and technical.
A principal concern, not always stated publicly, was the belief that infectious diseases could spread from the peasant sector into the more commercially orientated livestock enterprises, but the responsibility of governments for the welfare of the poorer sectors of society was clearly also an important driving force see (Table 9.1).
Both of these systems have evolved substantially, forming a much wider set of production systems in which socioeconomics play a critical role in the choice and efficacy of interventions to control infectious diseases, and as to who pays, and who benefits. Overall, the commercial sectors have shrunk in size considerably, although less so in southern Africa than in other parts of the continent. In most cases, state support in the form of loans and subsidized veterinary services has vanished. In many cases, their efficiency of production has improved, despite the economic and political pressures on them.
The changes in the ‘peasant’ sector have been much more profound, and it is here that socioeconomic issues usually outweigh technical issues in the optimal control of infectious diseases. Firstly, there has been recognition of the unique nature of certain livestock-centred systems in Africa characterized by pastoralism. These include those of the Maasai, Rendile, Samburu, Boran and others in eastern Africa, the Tuareg and Fulani of West Africa, and the Herero of Namibia, among others. These groups are characterized by their extreme scarcity of financial resources, but by a high priority to disease control for their ‘walking bank accounts’. They are highly demanding of veterinary interventions, in terms of the ‘priority’ of the disease it addresses and the appropriateness of the solution to their circumstances, but if these are correct, they are higher adopters of technologies to improve animal health than other smallholder groups in Africa.
Secondly has been the evolution of market-orientated smallholder systems that are commercial, often intensive in nature, but very small-scale. These include the smallholder dairy system of the Kenyan central highlands, the ‘emergent’ small-scale commercial farmers of South Africa and Zambia, the ‘small-scale commercial’ sector of Zimbabwe, and the peri-urban dairies of Addis Ababa, Cairo, and Dakar. Some of these have achieved significant success in terms of their productivity. In the Kenyan context, the smallholder dairy sector now supplies somewhere in the region of 90 per cent of the milk consumed in the country,145 a dramatic change from the domination of large commercial dairies some 30 years ago. In these sectors there has been a changing pattern of disease priorities, and a tendency for risk avoidance management practices to develop.
Again using the example of the smallholder dairy system of Kenya, the use of stall-feeding (known locally as zero-grazing) has dramatically reduced the risk of tick-borne diseases, with most of the fodder being cut and carried to the confined animals, thus avoiding heavy tick infestation derived from communal grazing. This has brought greater attention in this production system to the so-called infectious diseases of intensification, such as mastitis.
The other major peasant production system, widely prevalent in southern Africa, is not orientated to the livestock product market, but to the crop market. This sector, characteristic of the ‘traditional sector’ in Zambia and the ‘communal lands’ of Zimbabwe, is primarily a staple crop (mainly maize) system in which livestock play many roles. The use of oxen for tilling the land is probably one of the most important functions of cattle in this system, followed by the social (or networking) value of animals. The sale of cattle into the beef sector usually comes in third place, and formal off-takes from this sector have always been considered remarkably low (in the region of 3 to 5 per cent; see for example Perry and associates). 184 As a result, this system has suffered considerably from infectious diseases with the dramatic decline in public sector veterinary services, and the fragile socioeconomic role of cattle in the livestock component of the agricultural sector.
These are very broad production system groupings. Attempts have been made to develop more structured classifications of livestock production that further disaggregate and that can be used for analytical purposes and for priority setting. In 1996, Seré and Steinfeld223 produced the first global livestock production classification system associated with a detailed data set. For this reason, it has been widely used, and served for example as the basis for the International Livestock Research Institute’s (ILRI) recent prioritysetting exercise.203, 238 Seré and Steinfeld’s classification is based on the main food source for livestock, and on the agro-ecology of the region. They used the Food and Agriculture Organization’s (FAO) agro-ecological zoning classification and produced detailed country tables with disaggregated data by area, human population, livestock numbers, and livestock outputs for each livestock production system category. Thornton and co-workers237 have since refined and updated the Seré and Steinfeld classification system and database, and plotted a global distribution of the different production systems.
Table 9.1 Examples of general livestock production system categories currently in use in southern Africa and their animal population numbers
The general groupings used for reporting of livestock population numbers in many southern African countries still fall in the broad categories of traditional (communal, sector familiar, or Swazi National Lands) and commercial. In these, the ‘traditional’ sectors dominate in terms of livestock numbers, except in South Africa
| COUNTRY | CATTLE (GENERAL) | DAIRY | GOATS | SHEEP | |||||
| TRADITIONAL | COMMERCIAL | TRAD. | SMALL-SCALE | COMM. | TRAD. | COMM. | TRAD. | COMM. | |
| Angola | 291 213 | 59 546 | 0 | 0 | 0 | 1 685 6001 | 34 4001 | – | – |
| Botswana | 1 562 200 | 258 500 | 0 | 0 | 0 | 1 809 400 | 28 300 | 233 300 | 16 800 |
| Malawi | 579 700 | 23 500 | 11 600 | 0 | 4 000 | 1 583 400 | 14 100 | 98 000 | 4 800 |
| Mozambique | 291 213 | 59 546 | 0 | 0 | 1 400 | 518 5251 | 19 8741 | – | – |
| South Africa | 4 090 791 | 6 153 720 | 0 | 0 | 1 433 465 | 4 485 953 | 1 433 547 | 3 162 068 | 23 188 499 |
| Swaziland | 491 428 | 142 367 | 3 997 | 0 | 4 187 | 428 4751 | 30 8871 | – | – |
| Tanzania | 15 374 119 | 58 351 | 0 | 187 687 | 24 645 | 10 682 434 | 12 343 | 3 484 957 | 8 071 |
| Zambia | 1 700 000 | 970 0002 | 0 | 20 000 | 50 0002 | 646 257 | 71 8062 | 27 038 | 50 2122 |
Note: 1 Sheep and goats reported together
2 Also includes ‘semi-commercial sector’
Sources: References 8–16, 28
In Zimbabwe, the livestock populations of the smallholder sector are reported in three categories, communal, resettlement and small-scale commercial
| SPECIES | LIVESTOCK SECTOR | |||
| LARGE-SCALE COMMERCIAL | COMMUNAL | RESETTLEMENT | SMALL-SCALE COMMERCIAL | |
| Cattle | 1 679 652 | 3 689 640 | 468 056 | 231 416 |
| Goats | 77 667 | 2 582 463 | 188 263 | 61 477 |
| Sheep | 136 566 | 455 774 | 28 247 | 19 588 |
| Pigs | 135 216 | 123 095 | 8 826 | 11 674 |
| Donkeys | 14 468 | 323 550 | 26 726 | 8 378 |
| Horses | 9 047 | 67 | 22 | 74 |
Source: Department of Veterinary Services, Zimbabwe, 2001
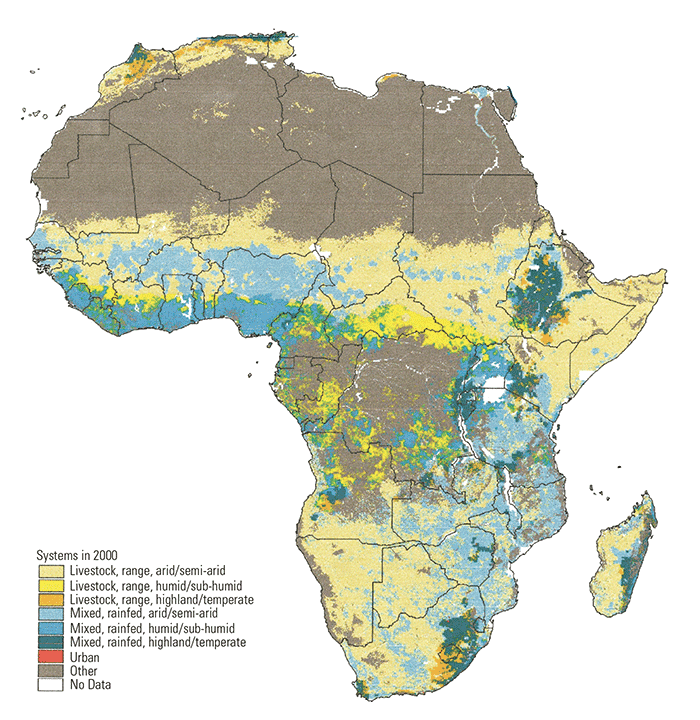
In this production system classification, four main production categories are identified: landless systems (typically found in peri-urban settings), livestock/rangeland-based systems (areas with minimal cropping, often corresponding to pastoral systems), mixed rainfed systems (mostly rainfed cropping combined with livestock, i.e. agro-pastoral systems), and mixed irrigated systems (significant proportion of cropping in which irrigation is used and is interspersed with livestock). All but the landless systems were further disaggregated by agro-ecological potential, as defined by the length of growing period. Three different agro-ecological zones are used: highland/temperate, arid/semi-arid and humid/subhumid. In summary, the following 10 livestock systems have been defined and mapped across the globe:
- LGA – Livestock only, rangeland-based, arid/semi-arid
- LGH – Livestock only, rangeland-based, humid/sub-humid
- LGT – Livestock only, rangeland-based, highland/temperate
- MIA – Mixed, irrigated, arid/semi-arid
- MIH – Mixed, irrigated, humid/sub-humid
- MIT – Mixed, irrigated, highland/temperate
- MRA – Mixed, rainfed, arid/semi-arid
- MRH – Mixed, rainfed, humid/sub-humid
- MRT – Mixed, rainfed, highland/temperate
- LL – Landless (peri-urban)
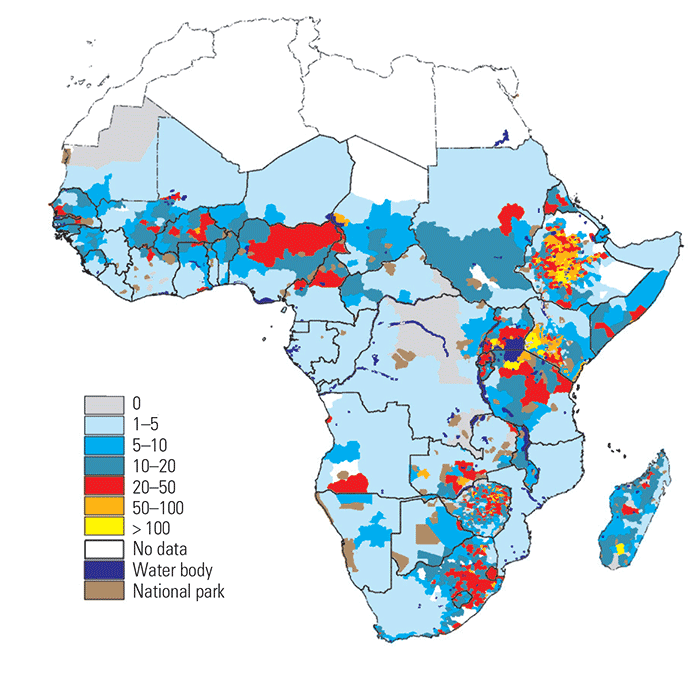
The geographical coverages for each livestock production system have been mapped237 and the distribution of systems for Africa are shown in (Figure 9.1).
In each of these systems, disease-control priorities are different, and the efficacy and viability of interventions also differ. Optimal success will be achieved by serious consideration of the socioeconomic issues influencing, indeed favouring, optimal delivery, adoption and impact in the particular target production system.
The role of livestock in African societies
Livestock play many differing roles in agriculture and the livelihoods of virtually all sectors of society in Africa (Table 9.2). They are important sources of animal protein in the form of meat, milk and eggs, their hides and skins contribute to clothing people, the fat from their bones contributes to the manufacture of soap, their work contributes to ploughing maize fields and moving products to and from markets, and their manure fertilizes the soil used for crops. They also play an important role in social networking in many societies, and as a ‘walking bank account’. Livestock and their products contribute to the incomes, health and livelihoods of livestock keepers, traders in livestock and their products, labourers on farms and in abattoirs, and of course to the great majority of the human population who consume livestock products in their diet. The export of livestock and their products generates valuable foreign exchange, so contributing to national economies. The health of livestock thus has a crucial influence on the health and well-being of Africa’s peoples.
Livestock are particularly important to the poor. It has been estimated that livestock form acomponent of the livelihoods of 70 per cent of the world’s poor.123 Animal diseases are an every- day occurrence to these people, as animals of the poor are particularly vulnerable to disease (due to many reasons, including lack of knowledge about their management and control, and lack of access to—and resources for—animal health and production inputs and services). Furthermore, poor farmers usually have few animals, so the loss of an individual animal has proportionally greater significance. They also have few reserves on which to survive during lean times, livestock being one of them, which they can use for recovery.
Livestock have not always been the flavour of the moment. Not so many years ago, much publicity was given in the media of the developed world to the woes of consuming meat, and the negative environmental and animal welfare impacts of keeping large numbers of pigs and poultry under intensive management. While these views might have been relevant to the conditions in many developed nations, they totally misrepresented the conditions and demands of the poor in the developing world. In the consumer societies of the developed world, per capita consumption of meat and many other livestock products is predicted to decline.49 In these high-income societies, a wide variety of readily available food products, low levels of malnutrition, and increasing levels of cardiovascular disease, diabetes and other complications of overindulgence have led to a negative view of animal protein. These have been coupled with concern over the environmental threats posed by the need to dispose of large amounts of animal effluent from large-scale and intensive animal production units.
Table 9.2 Domestic livestock species kept in Africa, and their contribution to national economics and household assets (from Perry et al. 2002190)
| SPECIES | CONTRIBUTION TO HOUSEHOLD ASSETS | ||||
| FINANCIAL | SOCIAL | PHYSICAL | NATURAL | HUMAN | |
| Cattle | Sales of milk, meat, hides, animals, draught power services, transport Savings instrument | Networking mechanism Social status indicator | Draught power for crop cultivation Draught power for transport | Manure for maintaining soil fertility | Household consumption of milk, meat |
| Camels | Sales of milk, meat, hides, animals, transport services Savings instrument | Networking mechanism Social status indicator | Draught power for transport | Household consumption of milk, meat | |
| Donkeys and horses | Sales of animals, draught services, transport (esp. water) | Draught power for crop cultivation Draught power for transport (esp. water) | Manure for maintaining soil fertility | Provision of household water supplies | |
| Goats and sheep | Sales of milk, meat, hides, animals Savings instrument | Networking mechanism Social status indicator | Manure for maintaining soil fertility | Household consumption of milk, meat | |
| Pigs | Sales of meat, animals Savings instrument | Manure for maintaining soil fertility | Household consumption of meat | ||
| Poultry | Sales of eggs, meat, fowl | Networking mechanism | Manure for maintaining soil fertility | Household consumption of eggs, meat | |
Ironically, these two factors are reversed in much of the developing world. Malnutrition is rife in many regions, and livestock products, particularly meat and milk, provide an important means to overcome this by providing protein, micronutrients and vitamins. The demand for meat and milk is predicted to grow by 2,8 and 3,3 per cent, respectively, per year, dwarfing the growth rates of 0,6 and 0,2 per cent predicted in the developed world.49 Furthermore, livestock are a powerful means of enhancing the purchasing power of the poor through the sale of livestock products, generating income that can be used for the purchase of food, education and health care. When it comes to the environment, livestock are a most valuable asset, providing the essential fertilizer for crop production, often unavailable or unaffordable in any other form.
The realization of the starkly different roles of livestock in the developed and developing worlds, and the predictions as to how these differences are likely to continue and intensify, has reversed the negative attitudes to supporting agricultural development that involve livestock.
On a global basis, precise estimates of the numbers of poor livestock keepers, traders, labourers and consumers by region and system, and the types of livestock that contribute to their livelihoods, have not been made. Thus, it is necessary to rely on crude aggregate estimates from summary reports. These do provide a picture of the relative importance of livestock to poor people. The Livestock in Development (LID)123 has developed global estimates of numbers of poor livestock keepers; these are presented in (Table 9.3).
Table 9.3 Numbers (in millions) of poor livestock keepers by livestock production system (from Livestock in Development, 1999123)
| AGRO-ECOLOGICAL ZONE | CATEGORY OF POOR LIVESTOCK-KEEPERS | |||
| EXTENSIVE GRAZIERS | POOR RAINFED MIXED FARMERS | LANDLESS LIVESTOCK KEEPERS | ||
| Arid or semi-arid | 63 | 213 | 156* | |
| Temperate (including tropical highlands) | 72 | 85 | ||
| Humid, sub-humid and sub-tropical | 89 | |||
| Total | 135 | 387 | 156 | |
* Predominately in irrigated agricultural areas, but also in other densely populated regions
The impact of infectious diseases and of their control
Disease classification based on disease impact, transmission and control
Diseases can be classified in a variety of ways, based on their aetiology, mode of transmission, impacts on animals and humans, and epidemiology, among other criteria. In terms of their impact, a number of factors are important in determining the scale of impact. These are:
- the number of species affected;
- the proportion of animals affected;
- the size of the geographical area affected or under threat;
- the ability of the disease to spread;
- the losses associated with the disease in both animals and humans; and
- the availability, affordability and efficacy of measures to control it.
We propose a classification of infectious diseases based on the main characteristics of their transmission and on their different impacts. This is presented in (Table 9.4.) This classification contains the following categories:
- Epidemic diseases, mainly corresponding to the list A diseases of the World Organization for Animal Health or Office International des Epizooties (OIE). These diseases are considered to be an international threat and are usually directly transmitted micro-parasites, for which the risks of epidemics accompanied by high production losses are the main impacts.
- Diseases from the OIE list B that are directly transmitted. In general these occur sporadically or endemically. They have important impacts on livestock production, and on lost production opportunities.
- Diseases from the OIE list B that are indirectly transmitted (mainly vector-borne). In general these occur sporadically or endemically. They also have important impacts on livestock production and lost production opportunities.
- Farm-level endemic diseases—termed ‘other diseases’ in the OIE classification. Their main impacts are production effects on the farm, but these have an aggregate influence on national productivity.
- Zoonotic diseases or infections in livestock in which a major impact is the risk of human diseases, this element of risk being important in control decisions (e.g. brucellosis, tuberculosis, Trypanosoma rhodesiense sleeping sickness).
- Food-borne infections and intoxications, such as salmonellosis, and Escherichia coli O157 infections. Such infections are increasingly a problem of intensive livestock production and marketing systems in which there are significant opportunities for cross contamination of, for example, carcasses and milk supplies.
Table 9.4 Classification of infectious diseases based on their transmission, types of impact, and level of impact
| CATEGORY OF INFECTIOUS DISEASE (see text) | EXAMPLES OF DISEASES | MAJOR IMPACTS | LEVEL OF IMPACTS | IMPACT INDICATORS | |
|---|---|---|---|---|---|
| 1 | Epidemic, directly transmitted, international importance (OIE list A) | Rinderpest, foot-andmouth disease, African swine fever, classical swine fever | Costs to minimize epidemic risk Production losses in susceptible animals if epidemic Restrictions to markets and international trade | International, regional, national, local area, farmer | Incidence of disease Control costs |
| 2 | Endemic/sporadic, directly transmitted, farm and national importance (OIE list B) | Haemorrhagic septicaemia, infectious bovine rhinotracheitis (IBR), dermatophilosis | Production losses Control costs Restrictions to marketing and trade | National, local area, farmer | Disease occurrence Production losses Control costs |
| 3 | Endemic/sporadic, indirectly transmitted, farm and national importance (OIE list B) | Babesiosis, cowdriosis, theileriosis, African animal trypanosomosis | Production losses Production opportunities lost Restrictions to marketing and trade Control costs | National, local area, farmer | Disease occurrence Vector abundance Production losses Control costs |
| 4 | Endemic/sporadic, directly transmitted, mainly farm importance (OIE other diseases) | Mastitis(Staphylococcus aureus), pneumonia (pasteurellosis), foot rot | Production losses Production opportunities lost Control costs | Farmer (national, local area less important) | Disease occurrence Production losses Control costs |
| 5 | Zoonotic diseases transmitted directly or indirectly from animals to humans | Brucellosis,tuberculosis, T. rhodesiense sleeping sickness | Impacts on human health Production losses Control costs | National, local area, farmer | Disease occurrence— animals and humans Production losses in animals Burden of disease (DALYs)* in humans Control costs |
| 6 | Food-borne infections | E. coli O157 infections, salmonellosis, campylobacteriosis | Impacts on human health Costs of food quality control systems | Local area, national | Burden of disease (DALYs)* in humans Control costs |
* DALY = Disability Adjusted Life Year, a measure of the cost of human disease
Multiple impacts of animal diseases and their control
Livestock play an important role in many agricultural systems and thus animal diseases that impair their function have multiple impacts at animal, farm, ecological zone, national, regional and international levels. Decisions on disease control depend on a good understanding of these multiple impacts. Focusing only on direct losses of overt disease can lead to a poor appreciation of the relative importance of different diseases and of what should be done, or not done, to control them.
The major impacts of different classes of infectious diseases are greatly influenced by the production system, infrastructure and other features of the socioeconomic milieu in different settings. The impacts of endemic diseases are mainly felt at farm level, while broader economic impacts can occur with epidemic diseases that restrict marketing and trade in livestock and livestock products. The occurrence of epidemic diseases affects both poor and richer livestock producers by marginalizing them from higher-price livestock markets and restricting their capacity for value-added trade.135 The overall benefits of the control of epidemic diseases are great188 and in rich countries, where resources are available for their eradication, this option is invariably pursued. However, numerous recent examples highlight how the maintenance of diseasefree status, when the disease still occurs elsewhere, is risky (e.g. foot-and-mouth disease (FMD) introductions worldwide, and classical swine fever reoccurrence in the Netherlands). The specific impacts of epidemic diseases and their control include both direct disease effects and potential trade benefits. For the latter, there are usually differential benefits and costs to different livestock sectors.135, 188
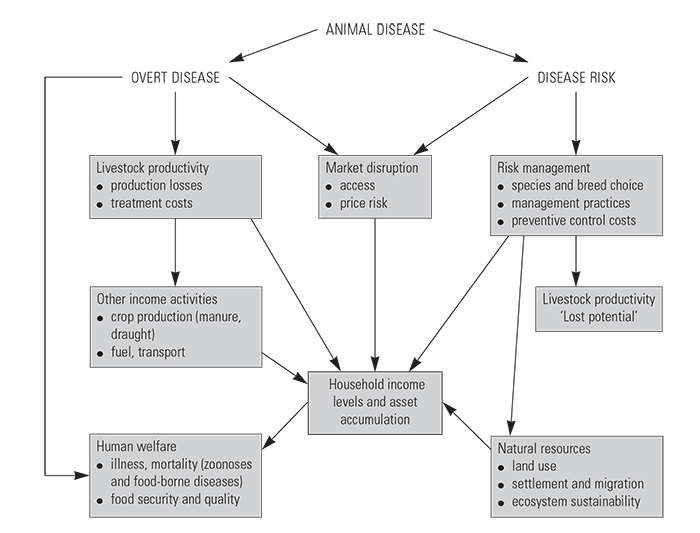
In recent years, a wider perspective on disease impacts has been adopted that considers both the direct effects, as well as a number of non-livestock (indirect) effects.Animportant consideration is to differentiate impacts associated with the occurrence of disease from those associated with the avoidance of disease risk. These are illustrated in (Figure 9.2)
The framework in the figure is particularly useful for considering the more profound impacts, beyond direct productivity effects, that livestock diseases have on African production systems. These include the impacts of disease on control costs, the constraints on livestock management imposed by disease risk, including limitations on species and breed choices, as well as impacts on the production of agricultural and other products, natural resource use, and human welfare. The following sections briefly describe the major types of impact.
Direct effects on livestock productivity
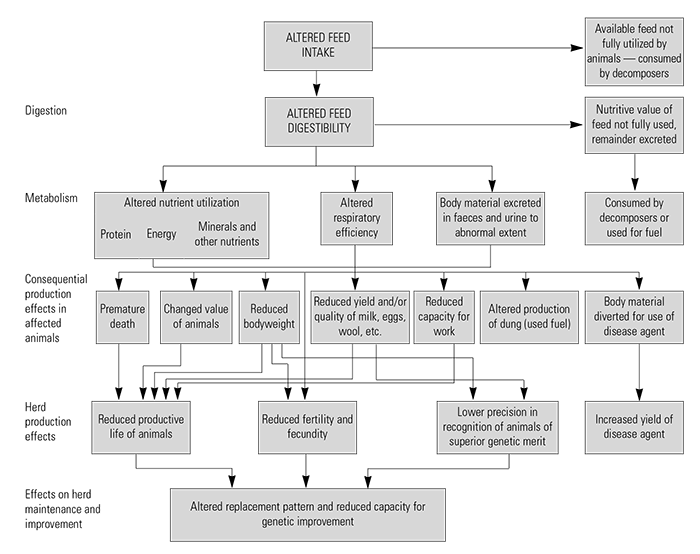
The direct effects of animal diseases on livestock productivity have been comprehensively described by Morris153 and are illustrated in (Table 9.3). These range from reduced feed intake and changes in digestion and metabolism to increased morbidity and mortality, and decreased reproduction, weight gain and milk production. They influence product quality at animal level, and their aggregate effects influence herd productivity. In addition, they limit economically important herd management decisions such as animal selection and optimal longevity. The interactions between disease, nutrition and genetic selection are important and emphasize the necessity to control the effects of key epidemic and endemic diseases before enhanced nutrition and genetic programmes can make an impact. Likewise, substantial productivity and economic gains will not necessarily be achieved by disease control alone.
Risk management—control costs and lost potential
Considerable costs are often incurred in controlling animal diseases and investments in control measures paid for may not always be effective. This is particularly true for smallholder farmers, who often lack information and have limited diagnostic support to make appropriate disease control and reatment decisions.132 The overall impact of control measures may be constrained by non-compliance of a significant proportion of the community, emphasizing the need for regulatory veterinary services supported by legislation and incentives to comply. An important principle, emphasized by several authors134, 188 is that it is not the total losses associated with diseases that are important, but rather the benefits relative to costs that can be obtained from different control options. An important loss associated with animal diseases is the cost of not investing in production inputs and adopting less profitable livestock management strategies to avoid disease risk 232 Such strategies may include a reduction in livestock numbers (for example in tsetse-infested versus non-infested areas in sub-Saharan Africa111), variations in grazing practices to avoid tsetse challenge,248 and limitations in breed choices (such as the necessary use of trypanotolerant breeds of cattle and small ruminants in West Africa).224
Impacts on crops and other farm enterprises
Given the key role of livestock in mixed farming systems, there are numerous effects of animal diseases on other agricultural enterprises beyond livestock themselves. These include impacts on crop production through decreased nutrient cycling and draught power, and loss of other products such as fuel and transport. Intensive smallholder farming systems in many regions of the developing world have integrated livestock into all other agricultural practices to such an extent that livestock diseases, when they occur, will have multiple agricultural production impacts.Good examples of such crop–livestock systems in Africa include the smallholder dairy production system in the East African highlands130 and the mixed crop–livestock systems in sub-humid West Africa.130 The multiple impacts of trypanosomosis in this latter system are highlighted in (Table 9.5) The integration of livestock into mixed farming systems is now widely considered to be an essential component for sustainable agricultural development.244
Ecological impacts
Another key category of livestock disease impacts is on the utilization of natural resources and ecological impacts. Livestock have been considered harmful to the environment and implicated in a host of environmental sins including desertification, deforestation, global warming, and pollution. Thus, the control of livestock diseases, particularly large-scale disease control efforts, have been the subject of much debate and environmental concerns have had a negative effect on investment in livestock disease control. This has been especially true of tsetse fly and trypanosomosis control in Africa. However, recent evidence suggests that the environmental impacts of disease control efforts are not invariably negative, but can be also positive or neutral, depending largely on how people choose to manage their livestock in response to reduced disease risk (see Reid et al.204 for a fuller discussion related to tsetse fly and trypanosomosis control and its effect on land use, settlement and migration, ecological habitat and biodiversity). Considering such inter-linkages is crucial in arid and semi-arid environments, where livestock are usually the key means for utilizing available natural resources and where the risk of animal diseases (among other risks) complicates rationale utilization of available natural resources. This has important implications for poor livestock keepers, who rely almost exclusively on common-property natural resources. These common-property resources are declining in most areas of Africa.
Human welfare and human disease
Animal diseases have multiple, important and measurable effects on human welfare. Many of these have been described in considerable detail by Schwabe,220 particularly the importance of animal products in human nutrition, the social benefits of livestock, and the impact of food-borne infections and zoonotic diseases. Livestock products have been shown to have considerable impacts on improving child nutrition,113 and animal disease control has also been shown to benefit child nutrition and welfare.44, 97 This is particularly so in pastoralist communities, in which 75 per cent or more of general and child nutrition is from milk and other livestock products.37
Traditional zoonotic diseases, such as brucellosis, bovine tuberculosis, hydatid disease and anthrax, for example, have impacts on both animal and human health. While many of the zoonotic diseases are not among the top impact human diseases globally159 — where the impact is measured by the Disability Adjusted Life Years (DALYs) lost, they are very important among livestock keepers and workers handling livestock products. A recent study in Mongolia has shown cost:benefit ratios of approximately 10:1 for the control of brucellosis in livestock and 2:1 for the direct health costs such as hospital care and drugs.212 In addition, it was estimated that one year of human disability (DALY) could be avoided for US$ 34. This study highlights the importance and benefits of policy changes to enhance inter-sectoral assessment and action for controlling zoonotic diseases.
The intensification of livestock systems in many areas of the developing world49 is expected to increase the transmission and impacts of food-borne infections and intoxications. However, because of poor surveillance and diagnostic facilities, their impacts on consumers, labourers and traders of livestock products in many African and other developing countries are likely to be poorly understood and ineffectively controlled. Currently, low-income consumers face greater risk from food-borne infections such as anthrax and hydatid disease, because they often buy livestock products in parallel markets in which meat inspection is non-existent. In addition to these classic diseases, new zoonotic risks are likely to develop, particularly in the area of food-borne infections in the more industrialized production and processing systems of developing countries. These food-borne infections include Salmonella serovars, Camplyobacter spp. and enterotoxigenic coliforms. It is also likely that public health will be further compromised by increasing levels of drug residues and antibiotic resistance. For low-income consumers, this will be a serious problem, as they will confront many of the multiple antibiotic-resistant infections occurring in Western countries but without the benefit of newer generation antibiotic treatments.
Impacts of disease on market access
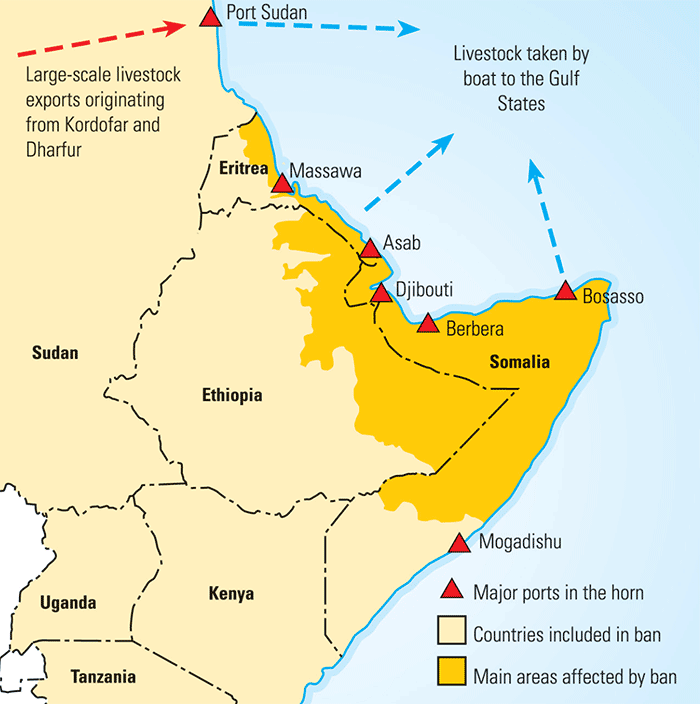
Outbreaks of infectious diseases in an area or country may result in local and/or international market disruptions as the movements of animals and livestock products are restricted. Two important examples in Africa in recent years have been the trade restriction on livestock movements from the Horn of Africa to the Arabian Peninsula associated with outbreaks of Rift Valley fever and the restriction of meat exports from southern Africa associated with FMD. The impact of trade restrictions on livestock trade in the Horn of Africa has been particularly devastating and is highlighted below.
In Somaliland alone, the ban associated with Rift Valley fever prevented the export of two million head of sheep and goats, valued at US$ 100 million between February 1998 and May 1999 (Figure 9.4). Because of this, local livestock prices decreased by 30 per cent, although these lower prices did not persist. Unfortunately, resolving the removal of this ban will not be easy.
Table 9.5 Multiple impacts of trypanosomosis in crop-livestock systems (mainly from Swallow, 2000232)
| Direct impacts on livestock productivity
Impacts on livestock risk management
Impacts on agricultural and other products
Effects on natural resource use
Impacts on human welfare
|
Sanitary and phyto-sanitary (SPS) requirements often have an even greater effect on market access in local markets. In many African countries, sanitary regulations for livestock products have been adopted from those put in place for industrialized systems in developed countries. These may or may not be reasonable or appropriate, depending on local conditions. In Table 9.6, the health risks associated with the marketing of raw and pasteurized milk in Kenya are explored in terms of protection of public health versus market access and income-generating activities.
Impacts on household income and asset accumulation
The benefits of livestock as a regular source of ‘income’, both for cash and barter, have been detailed in numerous studies.123 A number of observers (cited in LID, 1999123) have shown that poor farmers (both those with small land size or those who are landless) are increasingly relying on livestock as their main income source. In addition to income, livestock are often the main way in which poor farmers can acquire real assets, providing a safety factor when difficulties strike. The asset acquisition pathway usually begins with poultry, followed by small ruminants and/or pigs, with larger stock such as equids, cattle and water buffalo (Bubalus bubalis) being acquired at later stages. Animal diseases are a key constraint to both income generation and asset acquisition, especially for the poor, since they usually have limited access to cash to pay for animal health services.35, 45
In addition to improving the welfare of livestock keepers, enhancing livestock production through the control of animal diseases can have multiple benefits to consumers, traders and labourers. This is particularly true if disease control and other livestock development benefits are focused on poor livestock keepers such as smallholder mixed farmers and pastoralists. Enhanced livestock production on smallholder farms tends to be labour-intensive, using both surplus family labour and to a lesser extent labour from non-family members.19 Improved smallholder dairy production has also been shown to increase labour opportunities for small-scale milk traders and to lower transaction costs so that milk prices to poor consumers are considerably lower (Table 9.6).173
The importance of animal diseases to the poor in sub-Saharan Africa
Because of the impact that animal diseases can have on poor communities, disease control can be an important tool for improving poverty alleviation and enhancing social equity. This topic has been investigated in some detail in a report commissioned by the Inter-Agency Group of Donors Supporting Research on Livestock Production and Health, and sponsored by the Department for International Development (DFID) of the Government of the United Kingdom.190
Table 9.6 Assessing and managing milk-borne health risks for the benefit of consumers in Kenya (from Omore et al., 2000173)
| In Kenya, approximately 90 per cent of milk is produced by smallholder farmers. Much of this is consumed locally but increasing amounts are being collected, transported and sold in towns and cities by small- and large-scale traders. Omore and colleagues173 assessed a variety of risks associated with different milk marketing systems. Overall, both raw and pasteurized milk rarely met their accepted standards for bacterial counts. Ninety per cent of milk reaching consumers was not pasteurized but 96 per cent of it was boiled before consumption. Worryingly, 5 to 15 per cent of milk sampled had antibacterial residues; however, Salmonella and E. coli O157 were only rarely detected. Zoonoses (brucellosis and bovine-source tuberculosis) were not detected in milk from smallholder farms but antibodies to brucellosis in milk indicate that there is some risk that increases, as milk is collected and bulked from many sources. This study highlights the trade-offs that will need to be considered, in weighing both public health and economic benefits to producers, traders and consumers. Two milk marketing systems are emerging—a formal market for high-quality milk and milk products that will require the highest milk quality standards and an informal milk market involving small-scale producers and traders who sell unpasteurized milk at a third less cost to consumers. The informal sector provides employment to thousands of people. The authours recommend measures to improve the quality and safety of milk from the informal sector (advice to consumers on boiling milk, training and improved handling containers for small-scale traders) and maintain economic benefits for poor farmers, traders and consumers. |
Many of the points made thus far on the multiple impacts of animal disease are particularly relevant to the poor, who are often at the greatest risk of suffering from the deleterious effects of animal and zoonotic diseases and who have the least capacity to control disease or cope with disease risk. Here, we briefly outline the main pathways in which the control of animal disease can benefit poor people and illustrate this with an example of the control of Newcastle disease. Readers are referred to Perry et al.190 for a fuller account of the impact of animal diseases on the poor.
The control of livestock diseases is likely to improve the livelihoods (see Carney32 and DFID,48 for further accounts of livelihoods issues) of the poor in three main ways:
- Secure the current assets (human, financial, social) of poor people who keep livestock, consume livestock products, market livestock and livestock products, and work as wage labourers with livestock, by reducing the risks they experience through animal and zoonotic diseases.
- Enhance the marketing opportunities of the poor by controlling the diseases that affect the movement and marketing of livestock and animal products, primarily locally, but also regionally and internationally.
- Reduce the constraints experienced by the poor to livestock- based pathways out of poverty through intensification (improved productivity and performance efficiency through the use of inputs).
For many diseases, control efforts targeted at poor livestock keepers will simply require the transfer of available knowledge and tools; for other diseases, current tools and delivery systems will need to be adapted and improved so that they are appropriate for local conditions, and in particular for optimal uptake by the poor. In some instances, new technologies, such as vaccines and other disease control tools, will need to be developed based on new scientific methods and approaches.
It is generally acknowledged that poultry are very important to the poorest of the poor. The major disease constraint to poultry keeping throughout the developing world is Newcastle disease.228 However, greater benefits from Newcastle disease control could be enjoyed if other inputs such as the control of other important diseases (e.g. Gumboro, fowl pox, internal parasites) plus improved housing and feeding were also available as a package. Essentially, some benefits in securing poultry assets will be achieved by providing thermostable Newcastle disease vaccine alone but greater intensification and income generation benefits could be obtained if input packages were delivered. These are expanded upon in Table 9.7.
Measuring the impacts of infectious diseases and their control
There is a series of data needs required for the evaluation of disease impacts, and of the impacts of alternative interventions to prevent or control them. These are similar in principle regardless of the scale of assessment (i.e. whether at the level of herd, community, production system, agro-ecological zone, country or region).
Table 9.7 Assessing options for Newcastle disease (ND) control by vaccination in smallholder subsistence poultry systems of the developing world
| Socio-political and environmental context
Acquisition of capital
Evaluation of opportunities
Potential options Assessment of options Option A: Appropriate vaccination strategies to secure assets
Option B: Vaccination and other inputs to improve the intensification of rural poultry keeping
|
These fall into the following broad categories:
- Size, structure and composition of animal population under consideration.
- Characteristics of animal population and its environment, particularly in terms of agro-ecology and feeding management.
- Disease occurrence, in terms of prevalence, incidence, distribution and dynamics.
- Effects of disease (or of interventions) on productivity (or other key indicators).
- Value of productivity losses of disease/incremental gain from intervention.
- Value of control costs of disease/incremental savings from intervention.
- Impacts of zoonotic diseases (or of interventions) on the human population.
- Environmental impacts of disease (or of interventions).
This series combines demographic, epidemiological, productivity and socioeconomic data, illustrating the increasing trend towards a multi-disciplinary approach to disease and disease control impact assessments. Clearly, as a general principle, the broader the range of data included, and the better the quality of the data, the better the resulting decision. However, the collection and processing of data also have a cost in terms of money, time and communication, which must be taken into consideration, and may have a significant effect on the quality and value of any impact assessment study.
For each of these categories of data, there is a set of techniques that can be used for data collection and assembly, and these are listed and discussed in general terms below.
Use of intuitive response
Many impact assessments use intuitive valuations of some or all of the parameters listed. This might sound very non-scientific and obvious, but is the rule rather than the exception at the farm level regardless of the production system. Even at the national level, where resources are inadequate and when impact assessments are demanded within a short time, this technique is common. As a general rule, when made by farmers, this tends to be based on a combination of personal experience and financial commonsense, when made by economists it tends to be on the basis of sound economic criteria regardless of technical considerations,and when made by veterinarians it tends to be on the basis of technical considerations regardless of economics!
Passive surveillance systems
The standard source of quantitative data on livestock populations and infectious disease occurrence in most countries is still from national passive reporting systems. In these, livestock population sizes by administrative boundary (district, province) and cases of a given disease observed by the veterinary department are compiled in periodic reports. The advantage of using such data in an impact assessment is that they are available, and generally at no cost to the user (but of course often at a significant cost to the state in their compilation and publication). However, there are numerous disadvantages, which usually far outweigh the advantages. The data are often long out of date, with some annual reports of veterinary departments in Africa published 10 years late. Furthermore, there is a tremendous variation in the method of reporting used. As far as livestock population sizes are concerned, there are only a few countries that carry out regular livestock censuses. In many cases, populations seem to mysteriously increase annually by a standard index. There is also a large variation in the categories of livestock quantified, some taking species, some dividing within species (such as beef and dairy), and very few quantifying on the basis of production system.
The continuation by most national veterinary authorities in Africa to report livestock populations on the basis of species rather than production system severely hampers impact assessments. We have, for example, experienced this recently in studies of the impact of FMD and its control in the countries of south-east Asia. The pig populations of many countries in the region are reported as a single entity. Hidden within this are at least two contrasting production systems, in which both the infection dynamics of FMD and the economic impact of its control are entirely different.188 Medium- and large-scale pig production present in Thailand, the Philippines and to a lesser extent Vietnam is similar to commercial production enterprises in many parts of the world, with intensive hygiene measures, regular preventive FMD vaccination, and little in the way of FMD occurrence. In contrast, the vast village and ‘backyard’ sectors are highly vulnerable with little vaccination, hygiene and movement control being applied, and as a consequence, frequent outbreaks of the disease occur.
As far as disease reporting in passive surveillance systems is concerned, there is much variation from country to country, and several different indices are used. For example, some use number of cases, while others use number of outbreaks. In both instances, there is often a mixture of clinical and laboratory records contributing to this index, which may go undifferentiated. This is particularly important with infections in which the mere presence of an organism does not necessarily mean it has anything to do with the mortality, disease or production loss under investigation. Mere identification of haemo- or gastrointestinal parasites, for example, is often taken as diagnosticwhentheir presencemayhave been merely coincidental.
Generally, passively derived systems have been at their best when not applied nationally, but rather strategically and demand-driven for specific diseases. The overall decline of passively derived disease data has probably been less in developed countries and in the more commercial livestock enterprises of southern Africa. In these areas, there has been a significant shift in data collection techniques, through greater focus and more use of active methods. These changes have been supported by improved technologies for animal identification and more effective trace-back procedures, enhanced by the information and communications explosion, and the Internet. There has been a greater focus on priority diseases or disease complexes, on greater privatization of the data collection and analysis, which then become a marketable product, as well on the greater use of active surveillance techniques.
The decision as to whether or not to use official reports of disease occurrence based on a passive reporting system will depend on a deeper knowledge of the quality of the data, and on whether standard reporting procedures are being followed. This can vary dramatically from place to place, and from year to year, depending on the enthusiasm and diligence of individuals, and of course on the resources available for such a service.
In passive surveillance, consideration is rarely given to reporting the productivity effects of a disease (or a control intervention), productivity losses/incremental gains from an intervention, or control costs/incremental savings from an intervention.
Active surveillance systems
As a result of an expert consultation, FAO60 developed a framework for animal health information systems in developing countries that could serve the varying needs these countries face. A key component of this report was a recognition that the passive disease information systems that have been in place for so many decades are often of little value, frequently being out of touch and highly inaccurate. Strategic active surveillance, and strategic sample surveys and studies with focus, were considered to be much more useful. It has been encouraging to see this approach adopted in some regions; for example, the southern African countries recently held a workshop in Pretoria on demand- driven animal health information systems in collaboration with the OIE, FAO and ILRI.
Rabies surveillance in Kenya provides a good illustrative example of the problems of passive collection of data and the potential opportunities for strategic active data collection. For many years, passive data collection for rabies was considered to provide a reasonable indication of rabies occurrence in Kenya, albeit an underestimated one, particularly in non endemic rabies areas. Potential rabies exposures were reported to veterinary offices for follow-up and any humans considered exposed were referred to government medical officers for free/subsidized post-exposure treatment. However, over time, the latter form of post-exposure treatment was withdrawn, without alternative suppliers being established, and many people gradually stopped reporting cases as there was no incentive to do so. In order to better assess the rabies situation with a view to improving control, Kitala et al.106 established an active surveillance system using stratified random sampling for human and animal rabies, linked to a network of private sector vaccine suppliers. Active surveillance through community rabies workers uncovered 40 times more rabies cases than the passive surveillance system, as well as providing data on dog demographics and transmission parameters for the development of models to assess rabies transmission and the potential impact of control measures.107 Depending on resource availability and priorities, countries could consider mixes of passive surveillance (if it can be linked to animal or human health service provision) and active surveillance from sites chosen using probability-based sampling methods.
Observational studies
There is a large number of different types of observational surveys and studies that can be used to gain more precise data on livestock populations, their characteristics, infection and disease occurrence, and disease causality. Below is outlined the general characteristics of such studies, but for more detailed discussion of their relative merits, and design and analysis issues, consult the numerous texts on the subject.143, 180, 201, 221, 227, 239, 240
Surveys
The tool used to gather further information from the field on a particular subject is often termed a ‘survey’. Thrusfield,239 a stickler for detail, differentiated between surveys and studies as follows: He defines surveys as counting the occurrence of something (of a disease, for example), whereas studies involve the comparison of the occurrence of the event of interest in different groups of animals, usually undertaken to elucidate causal associations. The major characteristics and useful features of different survey and study types are outlined in Table 9.8.
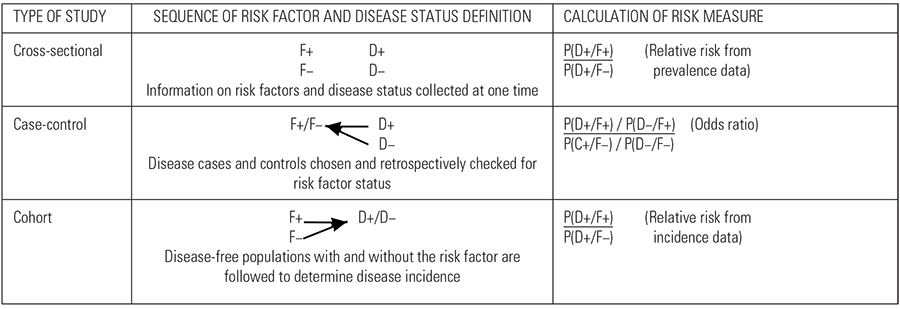
Observational studies are structured comparisons of the risk or rates (depending on the study) of disease (or of other parameters indicative of disease, such as infection, or decreased productivity) between different subsets of the animal population being studied. Successful studies depend on the accurate identification of 1) the presence of disease (or infection) in the study group, and 2) the presence or absence of attributes that may be determinants of the disease (or infection, or deviation from expected performance). Thus in its simplest form, this results in the generation of a 2 × 2 contingency table, as illustrated in Table 9.9.
Cross-sectional studies
In the process of better understanding diseases, their causes, their impacts and methods to optimally control them, a sensible starting place is to describe disease prevalence and what factors or attributes may be associated with it. For common diseases, crosssectional studies are the logical, quickest and easiest first step. For rare diseases, when disease records are available, case-control studies to compare cases with different noncase groups are commonly employed. After preliminary information on a disease has been collected, more powerful but costly study designs, such as cohort studies or clinical trials, need to be employed to assess the impact of risk factors on the incidence of the disease and to assess the impact of control factors on direct and indirect losses associated with the disease.
Thus, the commonest study used in the field in sub- Saharan Africa has been the cross-sectional. This has been the starting point for almost all efforts to study infectious disease occurrence in the field. It can be used to establish the prevalence of a given disease or infection, and the association with different natural (such as agro-ecological) or human-made variables (such as management or feeding). It has been quite extensively used to study the epidemiology of ECF over the last decade or so.47, 75, 138 At the commencement of a cross-sectional study, the only number that is known is the total animal population under study (or the number of the random or otherwise selected sample of that population). The number with and without the disease, with and without the attributes under investigation that might be associated with the disease, and any quantities to be estimated in the study, are unknown. Importantly, it will usually not be possible to differentiate between old and new cases (of disease or incidents of infection in the case of antibody prevalence being taken as the indicator of disease) in this type of study. The crosssectional study therefore measures point prevalence. The attributes being studied as influencing disease risk may have been acquired at different times than the disease itself, and thus causal relationships can be difficult to establish. However, causal relationships are somewhat easier to establish for attributes that are relatively unchanging and characteristic of the production system in which the animals are found. On balance, the principal benefits of crosssectional studies are their ability to estimate prevalence of disease (which may be a rough guide to its impact) and how prevalence cases are distributed by potential risk groups.
Case-control studies are also relatively easy and cheap to perform and have been used as a first step in trying to assess disease impact and factors associated with disease occurrence. The initial step is to identify ‘cases’ of the disease within a study population. Strict diagnostic guidelines must be established to define the case; the value of this type of study hinges on the quality of the diagnostic criteria, particularly with respect to their specificity. The same guidelines are then used to select non-cases, or controls, from the same population. Depending on the rarity of the disease, multiple controls per case may be selected. Casecontrol designs were initially responsible for linking smoking to lung cancer in humans,53 and have been used in the veterinary field to study a variety of conditions including feline urolithiasis,253 respiratory disease outbreaks in cattle200 and an acute and highly fatal intestinal syndrome in horses.185 While often very useful, case-control studies are susceptible to multiple biases, which has led them to be dismissed by many.61 Some main drawbacks include trying to extrapolate results from a specific control population (based on hospital or slaughterhouse records) and changes in the case population after their detection (e.g. dairy farmers with antibiotic residue violations who subsequently are better informed of sound antibiotic administration practices). These biases can be reduced to some extent, for example by investigating multiple control groups (often a control group in which the disease has been definitively excluded and a control group from the general population).
Table 9.8 A summary of the main features of surveys, observational studies and clinical trials in veterinary epidemiology
| RETROSPECTIVE | PROSPECTIVE | |||||
| TYPE | SURVEY | CROSS-SECTIONAL STUDY | CASE-CONTROL STUDY | LONGITUDINAL OBSERVATION STUDY | COHORT STUDY | CLINICAL TRIAL |
| Disease or infection frequency unit measured | Prevalence (sometimes incidence) | Prevalence (sometimes incidence) | Prevalence | Prevalence | Prevalence and incidence | Incidence |
| Comparison group | None (counting cases) | Non-affected animals in the study | Selected controls, matched with ‘cases’ for factors (e.g. breed, age) | Usually none | Selected animals not exposed to potential causal determinant | Matched animals exposed to infection or disease, but not to treatment |
| Primary data analysis procedure | Sum, mean, median | Relative risk, attributable risk | Odds ratio, attributable risk | Depends on parameter(s) being monitored | Relative risk, attributable risk | Relative risk, attributable risk |
| Ease of conduct | Very easy and cheap | Easy and cheap | Moderately easy and cheap | Hard and expensive | Hard and expensive | Hard and expensive |
| Level of causal proof | Low | Low | Low to moderate | High | High | High |
| Degree of investigator control | None | Very low | Low | Moderate | Moderate | High |
| Relevance to the field situation | Moderate | High but poor for impact measures | Moderate and poor for impact measures | High and good for impact if study population is representative | High and good for impact if study population is representative | High and good for impact if study population is representative |
Table 9.9 Basic data analysis for assessing the association between a two-category disease risk factor and disease or infection occurrence in different types of observational studies.
| DISEASE (OR INFECTION) STATUS | |||||
| PRESENT (D+) | ABSENT (D−) | TOTAL | |||
| RISK | PRESENT (F+) | a | b | a + b | |
| FACTOR | ABSENT (F−) | c | d | c + d | |
| STATUS | TOTAL | a + c | b + d | N = a + b + c + d | |
Longitudinal studies
Both cross-sectional and case-control studies provide a ‘snapshot’ of disease in a population, providing useful spatial data but lacking in temporal information. Investigation of what causal factors precede infection or disease incidence and the sequence of disease and control impacts are best investigated in longitudinal studies, in which study populations are monitored over time. There are three main variants of longitudinal study design. The first does not (necessarily) involve a comparison group, and is simply the monitoring of particular disease features over time. This may be simply to determine the dynamics of a particular infectious disease under defined field conditions over time, or to determine the relationship between levels of infection challenge and host response (in terms of immune response, morbidity, mortality and other productivity parameters). An important feature is the ability to measure infection and disease incidence, and disease- specific morbidity and mortality, all crucial components of impact assessment studies. However, longitudinal studies are expensive to set up and are logistically demanding, particularly if the study size is large. In addition, because of their expense and complexity, they are usually carried out under very defined local conditions, and it may not be appropriate to extrapolate the results to other circumstances.
Two other study types using a longitudinal structure, beginning with a disease-free population and monitoring the incidence of disease over time, are cohort studies (an observation study type) and clinical trials (an experimental study type). In cohort studies, the starting point is identifying a group of animals exposed to a particular suspect causal determinant, and then the identification of a matched group of animals not exposed. Both groups are monitored for the development of disease. This design of study is commonly used in human medicine with registered members of the population with certain characteristics (e.g. located in a high-risk radiation area, or with diabetes) to monitor their subsequent health compared to groups without those characteristics. In veterinary medicine, this design is primarily used where detailed herd monitoring schemes are in place. One example of a cohort study used to assess the incidence of ECF in different risk cohorts of female calves was conducted by Gitau et al.77
Clinical and field trials
In assessing the impact of diseases and their control, the most important aspect is not what the total losses associated with a disease are but what benefits can be obtained using specific strategies to control the disease.183 Clinical and field trials provide the optimal method for assessing the impact of control strategies on disease incidence, livestock production, reduction in control costs and other direct and indirect disease impacts (see above).
The key advantage of these trials is that they are conducted in real populations of interest, either individual animals, farms or disease control areas. The more the study groups resemble the diseased populations of interest, the greater the ability to extrapolate results widely. Another useful feature is that new control strategies can be compared to the current standard strategy, thus avoiding the difficult situation of trying to force farmers to apply no control in the face of disease risk. Finally, because their design requires the random allocation of animals or farms into treatment groups, clinical or field trials are extremely powerful, providing for statistical comparisons to be made without the worry of confounding, which is a constant problem in observational studies. Essentially, clinical trials have a very basic design. A study population, representing the reference population of interest, is assembled and randomly allocated into different treatment groups, depending on the treatment or control comparisons to be made. Treatments are made according to a standard protocol and, when possible, some degree of blinding, in which those who assess the disease and productivity outcomes are unaware of the treatment status. Following treatment, standard follow-up protocols are implemented to record loss-to-follow-up and clinical and production parameters of interest. Because individuals or herds are randomly assigned to treatment groups, the statistical comparison to assess differences between groups is usually straightforward unless there has been differential loss-to-follow-up. Further details on clinical and field trials can be found in standard veterinary epidemiology texts, as well as in Nordhuizen et al.164 and Meinert.148
Important considerations in conducting epidemiological studies in the field
Standard survey and study designs invariably need to be adapted to field conditions. The challenge is to maintain the essential features of observational or experimental designs that will lead to valid, meaningful and interpretable results. In Table 9.10 a checklist of key generic steps that need to be followed when planning a survey or study is provided.
Proper sampling is crucial to designing a study that provides maximum information for minimum cost and to obtaining valid results. Given its importance, there are surprisingly few comprehensive textbooks on sampling that can be read by non-mathematicians. Exceptions include Cochran,41 Levy and Lemeshow,121 and Yates 258 However, there are a number of papers, reports and books that describe useful methods for important sampling issues in veterinary field studies.29, 30, 31, 117, 143 The important principle in all sampling is that every animal has an equal or measurable probability of selection.
As part of the different field studies discussed above, the following practical questions usually need to be addressed:
- estimation of the prevalence or incidence of infection or disease;
- estimation of the distribution of risk factors, implementation of control methods and other factors associated with disease;
- comparison of disease rates, survival, mortality and productivity effects for different risk groups or under different control strategies; and
- detection of disease presence.
For all of these questions, the sample size will depend on the level of precision desired for the statistical estimate of interest, the level of confidence required that the estimate of interest lies within a given bound, the independence of sampling units from each other, and the performance (sensitivity and specificity) of tests used to measure disease. For small (finite) animal populations, the sample size may be reduced as a function of the total population size.143 Standard sample size calculations assume sampling from an infinite (10 000) population. Often in complex surveys or studies with multiple risk factors, it is difficult, a priori, to estimate the size of important comparison groups. In the end, as with most things in life, practical and logistical questions will play an important role in arriving at a final sample size.
A starting point for estimating sample sizes in cross-sectional studies is to estimate the likely prevalence of the disease or infection in the study population. Well, you might say, if we knew that, we wouldn’t be doing the study! The largest sample sizes are required when the estimated prevalence is around 50 per cent, and the smallest when the estimated prevalence is very high or very low. There is a reason for this apparently paradoxical situation. The estimation of prevalence requires an estimation of the proportion of positive to negative animals, rather than the absolute number of positives. Thus a larger number is required to demonstrate a 50:50 relationship than one of 90:10 (or 10:90). Thus, if no estimate of prevalence can be obtained, the sample size needed to determine a prevalence of 50 per cent will define the upper limit.
Depending on the distribution of disease, simple random sampling procedures may not be very efficient and more complex sampling methodologies, such as stratified, cluster and multi-stage sampling, may greatly decrease costs, increase sampling efficiency and provide estimates of disease occurrence at varying levels of interest. For example, a well designed study to determine the prevalence of an infectious disease in a district may be of little value in determining the impact, or in deciding on an intervention, if there are significant differences within the district. These differences may depend on factors such as agro-ecological zone, management and feeding practices and different cattle types, and it may be more effective to stratify the district on the basis of these variables. Standard statistical texts41 and epidemiology texts143 describe the sampling options available under different circumstances.
For infectious diseases, it is usual that the risks of disease for animals in a herd, agro-ecological zone or other grouping are not independent. While non-independence requires a modification of standard sample size formulae (an increase in sample size if the risk in a group is positively correlated), and is thus considered an extreme nuisance by most, it actually provides key information on the variability of disease risk and transmission. Highly contagious diseases, for example, tend to infect almost all animals of the susceptible species on a farm.
The clustering patterns of other diseases can provide information on whether within- or between herd risk factors are more important or whether factors associated with ecological areas are most influential (as happens with many vector-borne diseases.128 Methods to account for disease clustering, both in time and space, have been an active research area in recent years and there are a variety of techniques that can be used, ranging from simple to complex.
Table 9.10 Steps in planning a field survey or study
|
A simple and usually adequate procedure to apply if disease risk varies between herds is to inflate the sample size estimate by a factor of 1 + (m – 1)ρ, where m is the median herd size and ρ is the intra-herd-correlation coefficient (which measures the non-independence of cluster mates). A practical example of the use of this method in a study of antibody prevalence to tick-borne pathogens in three different agro-ecological zones of coastal Kenya can be found in the publication of Deem et al.47 More complex methods for two (such as animal and herd) and multiple (such as animal, herd, community, ecological area) levels of potential correlation have been described and applied to a variety of veterinary and medical situations.122, 126, 127, 129
Another important disease sampling application is to assess the presence of disease (or vectors) in large-scale control and eradication campaigns.125 Cameron and Baldock29, 30 have developed very useful methods for assessing the probability of disease presence using tests of varying sensitivity and specificity. These programmes are available and easy to use.65
As noted in Table 9.10, the most important and first task in developing any plan for a field survey or study is to carefully define the study objectives and from these identify important details such as the parameters to be measured, unit of concern and target population. Invariably, good studies result from appropriate and well-defined questions being asked and poor studies result when the questions to be addressed by the study are not correctly formulated.
Evidence from the published and unpublished literature
In assessing the impact of diseases and their control, much of the impact information required may be available from previously conducted studies. Unfortunately, study validity and quality vary greatly. Some studies can be relied on to provide good quality impact information while others are best left unread. Deciding on study quality is a daunting task for the uninitiated; however, with common sense and some basic guidelines on how to review papers, most readers can become very discriminating as to the value of most published studies. In (Table 9.11) an outline of the criteria that need to be considered in evaluating observational studies, and in (Table 9.12) an outline for the evaluation of clinical or field trials, are given. These checklists have been developed and refined over many years with a number of colleagues at the University of California, Davis, USA, and the University of Guelph, Canada.
Given that most information is imperfect, persons embarking on a large-scale review of evidence on the impacts of a disease and its control will probably wish to consider information from as many sources as possible, but to weight the information sources by the strength of the evidence provided. Meta-analysis of multiple study outcomes has been widely applied in human medicine to synthesize the results of multiple studies.50, 213 There are only a few examples of formal meta-analyses in veterinary studies.24 More common are impact and scenario development studies that informally evaluate and combine data from previous studies.183 In both sets of circumstances, an objective evaluation of the evidence presented by different studies is an important step in impact assessment.
Models
While evidence based on empirical field studies is critical to assessing the impact of a disease and its control, the wide range of field situations, control scenarios and other unique disease circumstances mean that all the empirical impact data required are unlikely to be available. Because of the wide variety of disease scenarios possible, models have been used to build a plausible framework that can be evaluated and validated with empirical field data, and if reasonable, used to make impact predictions under different disease and control scenarios. A wide selection of models has been used for different purposes to assess livestock populations, disease dynamics or combinations thereof. Based on their mathematical formulation, models are either:
- deterministic — in which a system of equations with an equilibrium solution is solved;
- stochastic — in which random probability elements are involved that can lead to extinction or outbreaks at various points; or
- simulation — in which computational algorithms are used to incorporate a wide variety of influences on different outcomes or states.
Livestock population dynamics models have been used to predict herd growth and productivity for different systems in Africa. One of the earliest models was developed by Texas A & M University215, 216 and subsequently applied to beef cattle production in Botswana.98 The model accounted for the interactive effects of genotype, breeding system, nutrition, reproduction, growth, milk production and nutrient intake. Animal performance was simulated by various sub-routines, the most important of which were animal growth, cow fertility and mortality. The model was used to predict the productivity of beef production systems in Botswana. Although it permitted a very detailed analysis, it was correspondingly very demanding in its data requirements. The model has not been used to assess the effects of disease on productivity.
A herd simulation model was developed by ILCA to provide the user with an array of potential policy options, such as for weaning, breeding, milking, buying and selling livestock, so that herd performance could be evaluated under different production regimes.110 A further model at ILCA, originally designed to assess interventions of fodder cultivation and feed supplementation in Mali and Niger, has been developed246 and subsequently modified to assess animal health interventions, specifically the economic impacts of chemoprophylaxis to control trypanosomosis in the Coast Province of Kenya.100
Other approaches to herd dynamics modelling have been taken20, 144 and models of the herd structure and dynamics of cattle in the traditional sector of Zambia have been developed.144 These models were used to assess the effects of varying calving rates, and calf and adult mortality on herd structure and dynamics.
For infectious diseases, mathematical models assessing transmission and infection dynamics have proved very useful in assessing and predicting the impact of changes in key risk factors and the impact of vaccination and other control strategies. The main concepts and major applications for infectious disease modelling have been set out by Anderson and May7 in their authoritative text on the subject. The essential feature of mathematical transmission models is quite simple, particularly for microparasites (e.g. viruses and bacteria). Transmission persists if each case can produce one or more secondary cases. The rate (or ratio) at which primary cases become secondary cases is known as the reproductive rate of the disease, measured as either the effective reproductive rate (R) or the basic reproductive rate (R0), and transmission persists if this rate is greater than one. The basic reproductive ratio represents the reproductive potential of an infection while the effective reproductive rate, influenced by several factors, is less than or equal to the basic rate. If by circumstance or control effort the reproductive rate falls below one, then transmission ceases. The basic reproductive rate can be estimated in a number of ways, including:
- as a function of the number or density of susceptibles;5
- as a function of the birth rate, average age at infection and the duration of maternal immunity;7 or
- directly from outbreak data in closed populations when secondary cases due to a primary initial case can be determined. 42
A variety of influences on disease transmission can be estimated via R0 . Of key interest for control programmes is the critical proportion of the population to be immunized (pc = 1 – (1/R0).7 R0 can also be evaluated for other control options.
Mathematical modelling approaches have been applied to a number of infectious diseases, including brucellosis,96 tuberculosis, 196, 241 sheep blowfly infestation,66 FMD,124, 155, 188, 256, 257 rinderpest,102, 211 rabies,6, 18, 42, 199, 245 theileriosis,70, 146, 147 heartwater,168, 169 fascioliosis,71, 73, 172 ostertagiosis,72, 176 and trypanosomosis.86, 87, 88, 150, 208
In general, mathematical models have been very useful tools for assessing many infectious diseases, with most disease transmission and impact of major control strategies reasonably well predicted by relatively simple models.7
Key socioeconomic information required
In most cases, socioeconomic factors will be a critical component for understanding the impact of disease and its control. The epidemiological investigations described above generally incorporate some aspect of socioeconomic data collection, and this becomes increasingly important in smallholder production systems where livestock activities are less commercialized. While the emphasis has been on epidemiological methods, there also exist a number of design issues and methodologies specific to socioeconomic data collection and analysis. Refer to Poate and Daplyn197 for an overview.
Two practical points are worth stressing. First, deciding the sample size for socioeconomic data is more likely to be based on a rule of thumb conditioned by logistical constraints rather than on the standard formula. For epidemiological studies, the focus is often on a key parameter of interest, which drives the sample size. In socioeconomic studies, there is usually no single parameter of interest, but rather a number of potentially influential factors. The rule of thumb, therefore, is to try to have sufficient numbers of units of analysis (e.g. farmers, households and villages) for each major subgroup by which the sample has been stratified, to permit statistical comparisons across groups (e.g. t-tests). Social scientists tend to be comfortable, therefore, with a sample size of 25 to 30 units of analysis per subgroup.
Table 9.11 Outline for the evaluation of observational studies
| Were the objectives of the study clearly stated? | Y/?/N |
| GROUP FORMATION | |
| FOLLOW-UP/TRACEBACK | |
| OUTCOME and ANALYSES | |
| PRESENTATION and CONCLUSIONS | |
| The more ‘?’ and ‘N’s’, the less valid the study. |
Table 9.12 Outline for the evaluation of clinical trials
| OBJECTIVES AND EXPERIMENTAL POPULATION GROUP FORMATION FOLLOW-UP PERIOD OUTCOME and ANALYSES PRESENTATION and CONCLUSIONS The more ‘?’ and ‘N’s’, the less valid the study. | |
Second, researchers often underestimate the care that is needed during the design and implementation of instruments (questionnaires) for socioeconomic surveys. Questions must be appropriately formulated and asked if they are to solicit effectively the information sought by the researcher. The error that can be introduced simply in the translation process has been highlighted by Lee et al.116 Since many of the types of data collected are standard (e.g. farmer characteristics, livestock management practices, and expenditures on health inputs), it is important therefore to review and draw upon existing proven questionnaire strategies from standard socioeconomic surveys and borrow as needed rather than trying to reinvent the wheel and repeat past mistakes. Allowing adequate time for pre-testing questionnaires and thorough training of enumerators are both essential for ensuring quality data.
Measurement of other indirect impacts of disease and their control
As noted above, animal disease and its control generate a wide range of other outcomes beyond the obvious direct impacts on livestock and human health. Both productivity gains and expenditures on control have implications for farm and national-level income. Secondary economic outcomes such as these are discussed in the following section on economic approaches since their evaluation often serves as the basis for decision-making about control. Within the framework of such analyses, innovative approaches often must be developed to address specific indirect impacts. When evaluating a potential vaccine against bovine trypanosomosis, for example, Kristjanson et al.111 attempted to value the lost production potential—cattle production activities not undertaken due to the disease—that could be recovered by the introduction of a vaccine. They estimated this value by assuming that cattle densities in tsetse areas would rise to equal those found in non-tsetse areas, and in this manner predicted the added value of production.
Indirect impacts of epidemic diseases, in particular, may be much more severe than the direct impacts. Effective control of diseases that restrict international trade can offer access to new, higher-value export markets for livestock and their products, generating added value and employment not only within the livestock sector, but also through the rest of the national economy due to multiplier effects. The substantial impacts that an outbreak of FMD would create in Australia in terms of loss of exports and secondary effects beyond the livestock sector have been demonstrated.68 The 2001 outbreak of FMD in the UK highlights the magnitudes of these impacts, including revenues lost to tourism. Mullins et al.158 report a similar, more modest, effect of tsetse control on tourism in Botswana, though no attempt is made to value the impact. In each case, the measurement of indirect impacts will require the development of appropriate economic models adapted to the specific impact, once the impact has been properly identified.
Geographical information systems
The reporting and analysis of disease occurrence in a spatial context can be a useful tool in impact assessment. For years, large maps peppered with coloured pins denoting the sources of reports of diseases and vectors have adorned the offices of veterinary departments throughout Africa. With the advent of computer power, these have developed rapidly into geographical information systems (GIS). What do they do that the pins did not?
Geographical information systems have computerized procedures to allow input, storage, manipulation, analysis and display of spatial data.207 They have been applied to the reporting and study of many infectious diseases, particularly the vector-borne infections in Africa.91 We do not intend to cover this huge area in any detail in this chapter, but will summarize the main features of this useful tool.
Spatial data require that a location be assigned to the recording of a disease event, in the form of latitude/longitude for example. This then allows this piece of data to be aggregated with other data describing the environment in which that event occurred. Commonly, the first step is to express the disease occurrence in relation to administrative boundary data (such as province, district), and this is how diseases are generally reported. However, while this serves to locate where a disease incident has occurred, it does little to evaluate disease risk and impact on productivity and other parameters. Therefore, the next set of data commonly included in any spatial reporting of disease is usually agro-ecological zone, land use category, vegetation and communications infrastructure (such as roads). Remarkably, in most African countries, these types of data are now much easier to acquire, generally at quite high levels of resolution, than data on animal diseases or on the hosts they affect. It is also possible to access, through the Internet and other sources, high-quality satellite-derived data on climate and vegetation cover.
In many African countries, GIS units have been established within the cartography departments of government ministries and planning departments that have assembled much of the natural and infrastructural features of their countries. However, there are relatively few that have established the same degree of capacity in the livestock production or animal disease sectors. On a continental scale, the International Livestock Research Institute (ILRI) developed a map of cattle density for the continent of Africa (Figure 9.5). In doing this, ILRI used the most recently available data at the highest resolution from published sources. The result combines some densities that are at secondary administrative boundary level, and possibly 10 years old, with new data at a very high level of resolution (such as Zimbabwe, with cattle data down to the dip tank level, revised and published annually).
Not only are there difficulties with the quality of data on numbers and distribution in space for a given species, but when it comes to impact assessment, these livestock distributions do not differentiate between the different production systems in which any one species is used, and so the different impacts that animal diseases and their control will have. In the case of the cattle example, this will be particularly important in the differentiation of dairy, pastoral, mixed agropastoral and ranching systems, for example.
Given the inadequacies of available data on the distributions of host populations, some attempts have been made to model their distributions using GIS, and most notable is the work of Wint et al.255 These authors used a combination of available data, including those assembled by ILRI, and modelling techniques (Figure 9.6).
Similar to data on the distribution of host species, available spatial data on animal diseases in Africa are in the categories of recorded observations and model predictions. Given the inadequacies of many disease-reporting systems, there is no continental distribution of the main diseases based on recorded observations.
However, these do exist for several countries in the continent. A notable example is Zimbabwe, which developed a GIS capacity in the Department of Veterinary Services in the late 1980s. This unit produces distribution maps of disease outbreaks and of intervention programmes (such as vaccination campaigns against Newcastle disease) at regular intervals, which are used to keep stakeholders informed, but also as feedback to veterinary staff to encourage better disease reporting (Figure 9.7).
Considerable progress has been made using GIS for predicting the distributions of disease vectors. Most notable in this field is work on the distributions of ticks and tsetse flies. For the latter, pioneering work was done by Rogers and colleagues at Oxford.209 This has recently been followed up by McDermott et al.,131 who used models of predicted climate change on the length of the growing period to predict how the distribution of tsetse species might change over the next 50 years (Figure 9.8).
The use of GIS has also extended to mapping disease risk. Considerable effort was devoted to predicting the current and potential distribution of ECF and its vector, the brown ear tick (Rhipicephalus appendiculatus), in Africa through the use of climate matching models,120, 186, 187 but more recently the focus has been on predicting risk of the disease through the incorporation of a wider set of variables including the satellite-derived vegetation indices. This has been done both at a country level (in Zimbabwe)55 and at a continental level152 (Figure 9.9 and 9.10).
Combination of models, observational and experimental studies
In the above sections an array of tools that can be used to determine the impacts of diseases and of methods to control them have been described. More likely than not, no single tool will suffice in providing the answer. As a result, should resources be available, a combination of tools can be used, and the results normally render more than the sum of the component tools included. Epidemiological tools have played a particularly important role in studying the dynamics and impact of tick-borne diseases.182 A practice we have advocated in several investigations has been a combination of cross-sectional and longitudinal (usually cohort) studies. The cross-sectional is carried out initially, stratifying the population by key variables to determine major differences in prevalence within a parent population of animals, farms or households, followed by a longitudinal study to determine incidence and impact in the contrasting prevalence populations. Such was the design of two sets of studies on the impact of ECF in two different regions of Kenya: the central highlands75, 76, 77, 78 and the coastal region.138, 139, 140 Both of these series of studies ended with robust estimates of infection prevalence, disease incidence, case-morbidity and case-fatality in the different agro-ecological zones, cattle types and feeding management regimens prevalent in the study areas. The studies in the central highlands also derived impacts of infections on daily weight gains. Neither series progressed into evaluations of economic impacts.
In another series of studies, carried out on the impact of heartwater and its control in Zimbabwe, two additional components were added to this framework for impact assessment. The first was a mathematical model of the infection dynamics of the disease, which served as a starting point for the entire study (Figure 9.11).168 Using initially a conceptual model framework, the major parameters and pathways in the maintenance of the infection were proposed, and supplemented where possible by parameter values in an attempt at understanding the dynamics of infections. Major gaps in knowledge, and in parameter values, were identified, and these formed the basis for the objectives of a series of experimental and observational studies. Experimental studies were used to investigate principles and, in some cases, quantify infection processes under experimental conditions, and observational studies, principally longitudinal studies in clearly defined areas, were used to determine parameter values under different ecological and farming systems conditions in Zimbabwe. 191, 192, 193, 194, 195 Concurrently, surveys were set up to characterize the different livestock production systems in which heartwater occurs, and their economics.38, 39 Armed with the results from this wide range of studies, it was back to the models. The first task was to use the models to predict heartwater incidence and mortality under the variety of production systems in the country, based on the outputs of the cross-sectional and longitudinal studies. The second task was to use these estimates to calculate the economic impact of the disease, and of different control scenarios contemplated by the Department of Veterinary Services.157
These economic impact studies were subsequently expanded to include the other countries of the Southern African Development Community (SADC) region,151 although with the large region and multitude of production systems involved, considerable reliance was placed in this study on secondary data. Finally, it was back to the models again to evaluate how new vaccines under development might work under the different control scenarios contemplated.169
These examples illustrate the need for a good conceptual framework behind impact assessment studies, the important role of good epidemiological data in economic impact assessments, and the advantages of combining investigative tools for maximum effect.
Risk assessment
The introduction, spread and control of diseases all depend on a sequence of biological events that are uncertain. Rules regulating trade in animals and animal products (e.g. the sanitary and phyto-sanitary (SPS) regulations of the World Trade Organization (WTO)/OIE, and decisions regarding what disease control and food safety strategies should be adopted all depend on assessing and managing disease risks. Hazard Analysis and Critical Control Point (HACCP) methods used in industrial food safety is one example of risk analysis methods that have been widely adopted. Research and development in risk analysis, both technical and policy issues, have greatly expanded in recent years.90, 137, 165, 247, 249, 259
A risk-assessment process for potential hazards (such as disease introduction) and methods to control them needs to be considered by various categories of decision-makers in animal health. This should be a formal process but may be either qualitative or quantitative. A key initial step is to identify and characterize the potential hazard both in terms of its causes and consequences. The likelihood of a hazard (or hazards) occurring is then estimated. The impact or consequences of hazards then need to be assessed from a biological, economic and environmental perspective. Then an overall assessment of both risk and the uncertainty of the risk-assessment process is made by combining the likelihood and consequence analyses with judgements on the quality of data available.Once a risk assessment has been conducted, its results need to be communicated to and discussed with all stakeholders so that it can be incorporated into risk-management practices. As disease control and food safety become increasingly more complicated, formal and informal risk assessment and management will be an important feature of decision-making in animal disease control.
Measuring the impact of zoonotic diseases on the human population
Within human health, the disability adjusted life year (DALY) is an instrument recently developed to allow the relative burden imposed on humans to be determined. This was used as a common health measure in the first Global Burden of Disease study commissioned by the World Health Organization (WHO)159 that documented the human health status, worldwide, for the year 1990. It has subsequently been used in revised global disease burden estimates, most recently in the WHO World Health Report 2000, in which the health status for 1999 is described.252
As well as being used as a means of ranking the overall burden of individual disease, the DALY is also particularly useful as an outcome measure in economic evaluations of interventions, including cost-effectiveness analysis.80, 81 Consensus cost-effectiveness ratios of US $150 per DALY and US $25 per DALY averted are agreed thresholds for disease control interventions to be considered ‘attractive’ or ‘highly attractive’, respectively, in a developing country setting, and used to advocate one strategy over another.251
The DALY is a measure of health outcome that incorporates both premature death and morbidity/disability. The DALYs caused by a given disease consist of the years of life lost (YLLs) and the years of life lived with disability (YLDs). In quantifying the cost-effectiveness of an intervention, deaths averted due to the implementation of the intervention are converted to YLLs using the age-specific life expectancy found from appropriate life tables. In comparing the DALY burden across different settings a common life table is used (e.g. for the global burden of disease studies a Japanese life table with a life expectancy at birth of 82,5 years for females and 80 years for males). For cost-effectiveness studies, comparing different interventions in one setting, a local life table should be used.159
The years of life lived with disability are calculated based on the duration of disability/morbidity, and disability weights given to each condition. The weights ranged between 0 and 1, with 0 representing full health and 1 representing death. The weights for each condition used in the Global Burden of Disease study159 were derived using the person trade off method developed at an international meeting held in Geneva in 1995. For the vast majority of zoonotic diseases no such consensus of disability weights exists.
The YLLs and YLDs are summed to give the DALYs score. The DALYs may be discounted to account for time preference and the risk premium. The discounting of health benefits, which gives a lower weight to health benefits that occur further into the future, has been criticized as inequitable4 and has attracted considerable controversy and debate. Age weighting of DALYs is also highly controversial and should be avoided as no consensus on the use of age weights has yet emerged, and the impact of the age weighting function used in the Global Burden of Disease has been shown to be counter-intuitive.21, 175
The use of a generic measure such as the DALY, rather than intermediate measures of benefits such as cases prevented or cases cured, allows for comparison of preventive and treatment interventions. Moreover, as DALYs incorporate both mortality and morbidity/disability, it is possible to make comparisons with other interventions that improve the quality of life, as well as with those that save lives. Whilst several limitations of DALYs have been identified in recent critiques,4, 22, 58, 175, 242 they do have several important advantages, particularly in cost-effectiveness analysis,81 and their use facilitates comparison with the results of other recent cost-effectiveness studies in developing countries.103
Official global estimates exist for 27 infectious diseases of humans, with the most recent values calculated for 1999252 as shown in Table 9.13. Using a strict definition of documented natural transmission between animals and humans, 20 of the 27 diseases are classed as zoonoses.236 However, from a control perspective, targeting the animal reservoir is unlikely to have a significant impact on the burden of human disease in 13 of these 20 strictly defined zoonoses (Table 9.13), as the anthroponotic transmission cycle (from human to human) is relatively much more important in these than any zoonotic cycle. For the remaining seven zoonotic diseases — trypanosomosis,74, 208 schistosomosis,235 leishmaniosis, 17, 206 Chagas disease,83 Japanese B encephalitis,115 hookworm198 and hepatitis (caused by hepatitis E virus)226 —there is evidence suggesting that the animal transmission cycle is important and that veterinary interventions could have a profound effect on the global DALY burden of the disease. However, it is debatable whether any of these diseases falls in the most important category of zoonoses, in which the animal host is the sole maintenance reservoir of human disease, and where blocking the transmission between animals and humans will eliminate the disease in the human population. The conclusion is that DALYs are yet to be calculated for many of the important zoonotic diseases of impact in Africa.
Measuring environmental impacts of disease and its control
Livestock have been considered harmful to the environment and implicated in a host of environmental sins including desertification, deforestation and global warming. The control of livestock diseases, particularly large-scale disease control efforts, have been the subject of debate from an environmental impact perspective. The greatest environmental concerns have been in the large-scale control of tsetse fly and trypanosomosis in Africa, both the indirect effects on habitat and wildlife174 and the direct effects of chemicals used for tsetse fly control on non-target organisms (including people), water quality and nutrient cycling.161
A number of studies have assessed direct impacts of chemicals.54, 59, 109 In general, newer insecticides and the methods by which they are applied are considered to be less persistent and have no long-term, irreversible effects on nontarget species.222 However, risks always exist for personnel handling chemicals. Rola and Pingali210 provide a framework and examples for assessing the economic implications of such risk in a related application for crop pesticides. Another potential impact is the development of resistance to pesticides or drugs, such as in the case of trypanocides in sub- Saharan Africa.69 Fleischer and Waibel63 describe an approach based on natural resource economics for assessing the social value of resistance for crop protection in Germany; the approach has yet to be applied to the field of animal health. Various indirect environmental impacts of vector control of tsetse fly on wildlife, biodiversity and land use have been studied and found not to be invariably negative 204, 205, 254 but to depend on ecological circumstances and people’s response to reduced disease risk. Approaches have yet to be adapted from natural resource economics for valuing these types of impacts.
Table 9.13 Burden of infectious diseases in disability-adjusted life years (DALYs) by cause in WHO regions; estimates for 1999. The zoonotic status of each disease is indicated (from WHO 2000252)
| CAUSE | GLOBAL | AFRICA | THE AMERICAs | EASTERN MEDITERRANEAN | EUROPE | SOUTH-EAST ASIA | WESTERN PACIFIC | ZOONOTIC |
|---|---|---|---|---|---|---|---|---|
| HIV/AIDS | 89819 | 74449 | 2812 | 2172 | 725 | 8866 | 795 | No |
| Diarrhoeal diseases | 72063 | 24321 | 25141 | 10123 | 1131 | 30017 | 3955 | Yes |
| Malaria | 44998 | 36838 | 76 | 2774 | 2 | 3071 | 2235 | Yes |
| Tuberculosis | 33287 | 8721 | 1114 | 2260 | 1258 | 14101 | 5832 | Yes |
| Measles | 29838 | 17463 | 31 | 3293 | 148 | 8308 | 594 | Yes |
| Tetanus | 12020 | 3039 | 160 | 1821 | 131 | 6302 | 566 | Yes |
| Pertussis | 10905 | 4937 | 189 | 1415 | 197 | 3634 | 533 | No |
| Meningitis | 9824 | 3620 | 722 | 1472 | 438 | 2567 | 1005 | Yes |
| Chlamydia | 7969 | 2233 | 779 | 250 | 581 | 3978 | 145 | Yes |
| Syphilis | 6081 | 3189 | 113 | 648 | 6 | 2067 | 59 | No |
| Gonorrhoea | 5686 | 2425 | 313 | 101 | 126 | 675 | 45 | No |
| Lymphatic filariosis | 4918 | 1834 | 8 | 11 | 0 | 2788 | 278 | Yes |
| Hepatitis* | 2790 | 1172 | 212 | 109 | 57 | 706 | 535 | Yes |
| Trypanosomosis* | 2048 | 1991 | 1 | 56 | 0 | 0 | 0 | Yes |
| Leishmaniosis* | 1983 | 256 | 50 | 210 | 0 | 1467 | 0 | Yes |
| Schistosomosis* | 1932 | 1637 | 133 | 98 | 15 | 18 | 30 | Yes |
| Poliomyelitis | 1725 | 279 | 152 | 19 | 1 | 1095 | 178 | No |
| Hookworm* | 1699 | 406 | 173 | 93 | 0 | 862 | 166 | Yes |
| Trachoma | 1239 | 434 | 0 | 237 | 0 | 62 | 505 | No |
| Onchocercosis | 1085 | 1083 | 2 | 0 | 0 | 0 | 0 | Yes |
| Japanese encephalitis* | 1046 | 0 | 0 | 1 | 0 | 218 | 828 | Yes |
| Chagas disease* | 676 | 0 | 677 | 0 | 0 | 0 | 0 | Yes |
| Ascarosis | 505 | 202 | 43 | 43 | 0 | 186 | 31 | Yes |
| Trichurosis | 481 | 198 | 155 | 3 | 0 | 120 | 5 | Yes |
| Leprosy | 476 | 79 | 60 | 10 | 0 | 318 | 9 | Yes |
| Dengue | 465 | 24 | 0 | 1 | 0 | 440 | 0 | Yes |
| Diphtheria | 151 | 24 | 2 | 11 | 1 | 110 | 2 | No |
* Disease in which the zoonotic component is considered significant compared to anthroponotic transmission
Identifying and deciding between intervention options
In this section we focus on tools and methods for deciding between different control strategies for diseases judged to be of high priority. A much more extensive review of the relevant issues and methods, and from which much of the material in this section is drawn, can be found in the August 1999 OIE Revue Scientifique et Technique 181 The textbook on animal health economics by Dijkhuizen and Morris52 is the most up-to-date source for details on how to apply many of the standard analytical methods.
Steps in evaluating disease control interventions
Four major steps are generally involved in determining the most appropriate, technically feasible and economically sound control or intervention strategy. These are: scenario development; quantification of the intervention options and their effects in epidemiological and economic terms; development of clear decision criteria; and ex post evaluation of the results of the decision-making process.
Scenario development
In scenario development the framework for decision-making is constructed by listing the intervention options that could be considered and the different consequences of such options are described. The scenarios might be based on different geographical expansions of a disease (such as that described by Mukhebi et al., 1999,157 when considering the spread of heartwater in Zimbabwe), different technologies used for disease control (such as that of Minjauw et al., 2000151 when comparing acaricide-based and vaccine-based strategies for heartwater control in southern Africa), a combination of the two (such as in the Mukhebi et al., 1999157 example cited), or different lengths of time for a disease eradication programme to succeed (such as that of Perry et al., 1999,188 when considering future FMD eradication in Thailand).
There are many important features of scenario development, but the most important is that they be realistic. This requires that they not only take into consideration national policies and aspirations, but also that they are based on the best available information on disease occurrence, intervention effects, logistical considerations and resource availability.
Quantification of the intervention options and their effects
This step in the process is often the stumbling block to high-quality and credible decision-support aids due to inadequacies in data. It is necessary to articulate and quantify accurately the components and costs of the different intervention strategies under evaluation, and furthermore, what outcomes they will produce, in both epidemiological and economic terms. Invariably, there are data gaps and key assumptions have to be made but as long as they are reasonable, and are well articulated in the analysis, they need not be an impediment to developing quality decision-support aids.
Secondary data are often available from a broad range of sources, based on the types of information systems or studies described in the preceding section, with considerable variations in accuracy and precision. When the appropriate secondary data are lacking, primary data collection may be necessary to properly capture the specific epidemiological and socioeconomic consequences of the different control options under consideration.
It is often relatively straightforward to measure and quantify the direct impacts of disease control in terms of the market value of changes in production losses and control costs. Identifying and valuing additional indirect or secondary impacts may present much more of a challenge. For example, indirect, longer term impacts of improved reproductive performance may require use of a herd simulation model and a set of assumptions about how herd structures and off-take rates are expected to evolve over time. Off-site impacts, such as those on the environment or on other parts of the marketing chain and beyond (e.g. market prices, employment, human health), may require auxiliary analyses that draw on the economic literature. Still others may be ‘intangibles’ that are much more difficult to value, but clearly need to be recognized. How, for example, can the value of a control option better targeted to the poor be quantified compared to one targeted to commercial producers?
The challenge is to assemble information from disparate sources in a meaningful decision-making framework, combining both epidemiological and economic information.
Development of clear decision criteria
This is an area that is still poorly developed. The commonest criteria used are just biological, based on efficacy. Does the intervention successfully control the disease? However, as has been repeatedly demonstrated in empirical studies, what makes sense technically (the technical optimum) only on occasion makes sense economically. The decision criterion is therefore usually reframed in terms of: does the value of the impacts of disease control justify the effort (cost) invested? Or, if various options are being considered, which yields the best value for the effort or resources (cost) invested? Translating disease control interventions and their impacts into dollars and cents also provides the benefit of a common unit of measurement that can serve as the basis for comparing apples and oranges, e.g. control that improves reproductive performance versus another that improves weight gain in an animal
If the economic criterion is indeed the one adopted for decision-making, then several different measures of economic worth may be considered. The most popular include the benefit:cost ratio, the net present value and the internal rate of return-all measures from financial investment analysis. As noted above, however, these quantitative measures will often need to be qualified or supplemented by additional criteria for impacts more difficult to quantify or value, such as the degree to which benefits are distributed equitably among different segments of society.
Also affecting the criteria is the level at which the decision is to be made. Decision-makers work at all the different levels — farm, community, local authority, national, regional and international. Although certainly not the rule, as the decision moves from the farm to the national level, the criteria generally change from financial to economic. Financial analysis refers to costs and returns associated with cash transactions at nominal market prices directly incurred by the decision-maker, whereas economic analysis broadens the scope to include non-cash costs and returns valued at their ‘true’ prices (e.g. without distortions introduced by taxes and subsidies), and evaluates the value of control for society as a whole rather than the individual user. Financial analysis is therefore appropriate when trying to assess whether the control intervention will be feasible from the individual user’s perspective — can farmers or others handle its demands on their resources and cash flow, and is it sufficiently attractive in terms of financial benefits to merit their participation? Economic analysis, on the other hand, tends to be used when the decision-maker is interested in the public good, especially if public resources are used to support an intervention.
The choice of criteria, the perspective of the decisionmaker, and the particular context of the disease under consideration then inform the specific analytical approach or technique to be used to evaluate the disease control intervention.
Ex post evaluation of the decision-making process
It is clearly important that scenarios developed, data used, analyses conducted,and syntheses prepared, are appropriate for the decision-maker in question, in which case the adoption of the results should approach 100 per cent. However, they rarely do. One way to avoid the pitfalls of developing decision aids for farmers or national strategy development that are not used is to learn from each analysis carried out, and conduct an ex post impact assessment of the evaluation. This is rarely done, but is an area that needs developing if veterinary epidemiology and economics are to have greater impact on the quality of decision- making in the future.
The special case of eradication
In scenario development, the most common framework compares alternative control options. However, occasionally an eradication scenario is included, and this requires special consideration, for if eradication is to be a viable scenario, certain criteria must be met.62 These are summarized as follows:
- effective tools for breaking transmission are available;
- epidemiological and population ecological features of the disease make it amenable to eradication (e.g. clinical cases and/or infections are easily detectable, the size of the area affected is not too large and has a relatively stable population);
- the disease is of sufficient socioeconomic priority;
- there is compelling evidence that the cost to benefits of eradication outweigh those of control; and
- adequate managerial, operational and financial resources are available.
With these criteria, the feasibility, advantages and risks of eradication versus control can be carefully evaluated before a decision to embark on an eradication programme is made. The most powerful advantage of achieving eradication is that there may be no subsequent disease control costs, depending on whether eradication is global or local; in the latter case, costs continue to be incurred to avoid reintroduction of the disease. However, if eradication fails, continuing disease control costs remain and previous eradication programme costs (often substantial) are sunk and do not accrue the expected benefits. In addition, eradication programmes may divert funding from other more general public or animal health programmes of benefit.84
Evaluating other control strategies
If eradication is not being considered, the next set of decisions revolves around how best to live with the disease. This means whether to treat, to control, or to prevent (which usually infers the use of vaccines, but can also be through chemoprophylaxis and the use of genetic resistance). At the farm level, this decision will be influenced mainly by the type of infectious disease, the impact the disease is having or is likely to have, the (global) availability of different technical options, the local availability of those options, their probability of success, the impact of success, the relative costs of different intervention options, the availability of resources in the context of other constraints and obligations, and any social or regulatory responsibilities of the farmer to follow certain procedures with a given disease. At the national level, decisions will be influenced by the same factors, but the assembly of data and the evaluation of options will be infinitely more complex.
Approaches for financial and economic analysis
A wide range of analytical techniques has been successfully adapted from the economics literature for application to decision- making for animal disease control. Before briefly summarizing some of the most commonly used, it is useful to note three features of these applications that distinguish the field of animal health economics:
- The nature of livestock production, particularly for larger animals. Livestock are often a production asset and at the same time, the product. Control affects both, and its benefits may need to be valued from both perspectives. This requires taking into account the productive lifespan of the animal, and the multiple production cycles associated with off-take of livestock products. For this reason, economic analysis must often integrate some type of herd dynamics model that can describe how the production assets and off-take evolve over time.
- The nature of infectious disease. Risk of disease and the impact of control often are not limited to the individual animal; they may depend as well upon the epidemiological dynamics within the host population. Again, economic analysis must try to capture this dimension, and as a result, there have been a number of attempts to integrate epidemiological modelling with economic techniques. This also means that economic costs and benefits of disease control may extend well beyond the individual animal, herd, or farm.
- The multiple roles of livestock in smallholder farming systems. Economists like simple production processes where an added input results in an easily identifiable added output. In a commercial livestock production system, inputs and outputs are usually well defined and quantifiable, though complicated to some extent by the preceding two points. In smallholder farming systems, as described in preceding sections, livestock serve a number of different purposes, many unrelated to commercial sale. As a result, the benefits of disease control may need to be valued through their impact on draught power and improved crop production, or through other intangible impacts such as enhanced social status. Similarly, farmers have multiple objectives, so their decision-making may not be a function of the cash costs and benefits alone, which may violate the underlying assumption of many of the standard analytical methods.
Each of these features has considerably complicated the economic assessment of disease control, and the collaboration between veterinarians, economists, and livestock productionists to address these challenges has led to the development of a specialized field of animal health economics.52
In Table 9.14 the principal analytical approaches used for decision-making based on economic criteria are summarized. The first set refers to farm-level decision-making, and are used either to advise the farmer which control option will be most profitable to use by comparing costs and benefits, or to understand and predict what level of control the farmer is likely to adopt by evaluating the trade-offs in terms of overall farm resource allocation. The second set of approaches is better suited for the decision-maker managing larger-scale control interventions, often involving public resources.
Comparing costs and benefits is the basis underlying the majority of financial and economic analyses applied to animal disease control. Most may be broadly termed as ‘cost benefit analysis’ in spirit, though we must be careful to avoid confusion since this term is also the name of one of the specific techniques in this class.
Enterprise budgeting and gross margin analysis
These are standard tools for farm management in which a budget is developed for one or more livestock production activities. The budget details all inputs and their costs, together with the outputs and revenues, so that the net profit can be calculated. Enterprise budgets include both variable and fixed costs, whereas gross margin analysis only considers variable costs.
For disease-control decision-making, disease control is treated as an integral part of the production process, and so budgets are estimated for production activities based on different control options. Okello-Onen et al.,171 for example, treat three cattle herds under different tick control regimes (no control, once-a-month dipping, twice-a-week dipping) as different production activities and estimate gross margins for each. More typically, these approaches could be expected to be used in cases where the disease control intervention affects use of other inputs or the quality of outputs, thus fundamentally changing the production process, such as comparing the use of trypano-tolerant breeds of cattle versus regular chemotherapy with trypanosusceptible breeds.
Partial budget analysis
This is an abbreviated version of gross margin analysis. Rather than estimate a complete budget for a production activity, it considers only those costs and revenues that change due to the control intervention to determine if, and to what degree, incremental costs associated with the intervention (cost of the intervention plus any returns foregone due to the control) are rewarded by incremental gains (added output value plus any costs savings). The analysis is usually limited to a single production cycle or time period, and provides absolute (net return) and relative (benefit-cost ratio) indicators of investment profitability. With its modest data requirements, it is one of the easiest and most practical approaches. Pegram et al.177 used this approach to compare the value of intensive tick control versus no tick control in traditionally grazed cattle herds in Zambia. Gummow and Mapham82 provided a well-organized example of partial budget analysis enhanced with a stochastic dimension applied to experimental trial data from South Africa for evaluating a vaccine against Pasteurella haemolytica in cattle; while Kamau et al.105 reported a similar analysis to evaluate cypermethrin pour-ons to control trypanosomosis in Kenya.
Table 9.14 Methods for the financial and economic analysis of disease and its control
| FARM LEVEL | ||
|---|---|---|
| METHOD | OBJECTIVE | FEATURES |
| Gross margin/enterprise budgets | Evaluate financial viability by comparing all costs and revenues for a given production activity incorporating a specific disease control strategy | – Suited to commercial production activities with market-valued inputs and outputs – Fails to incorporate impacts beyond the specific production activity |
| Partial budgets-Cost-benefit analysis (CBA) | Evaluate financial or economic viability by comparing incremental costs incurred and revenues generated by adopting a specific disease control intervention | – Single time period – Least demanding in data requirements – Multiple time periods – Cost and revenue flows discounted to account for time value of money – Indicators: NPV, BCR, IRR |
| Decision analysis | Compare the expected net returns from a process of sequential decision-making and probability-weighted outcomes | – Suited for analysis of response to epidemic disease occurrence that can be expressed as risk probability, or disease control having multiple potential outcomes – Mimics farmer decision-making – Fails to incorporate secondary impacts |
| Optimization models | Maximize an objective function (often profits), subject to resource and other constraints | – Useful for estimating optimal level of disease control, regardless of whether control is ‘lumpy’ or continuous variable – E.g.: linear programming, dynamic programming, econometric agricultural household model |
| Simulation models | Use a systems model to perform ‘what-if’ scenario analysis | – Typically based on biological models (esp., herd dynamics model, livestock/crop model) |
| REGIONAL/NATIONAL LEVEL | ||
|---|---|---|
| METHOD | OBJECTIVE | FEATURES |
| Cost-benefit analysis (CBA)-Economic surplus | Evaluate financial or economic viability by comparing incremental costs incurred and revenues generated by adopting a specific disease control intervention | – Multiple time periods – Cost and revenue flows discounted to account for time value of money – Indicators: NPV, BCR, IRR – Can be extended to include a wide range of impacts if they can be assigned a market value – Extension of CBA to capture impact on market prices and distributional impacts between producers and consumers – Typically used for evaluating returns to research investments |
| Decision analysis | Compare the expected net returns from a process of sequential decision-making and probability-weighted outcomes | – Suited for analysis of response to epidemic disease occurrence that can be expressed as risk probability, or disease control having multiple potential outcomes – Mimics veterinary service decision-making – Fails to incorporate secondary impacts |
| Optimization models | Maximize an objective function (often profits), subject to resource and other constraints | – Useful for estimating optimal level of disease control, regardless of whether control is ‘lumpy’ or continuous variable – E.g.: linear programming, dynamic programming |
| Simulation models and systems analysis | Use a systems model to perform ‘what-if’ scenario analysis | – Can integrate a variety of production and post-production systems |
Cost-benefit analysis (CBA)
This is essentially partial budget analysis extended over a longer period of time. Incremental costs and gains are identified for each time period, and discounting is introduced to capture the time value of money. The time value of money refers to the idea that $1 today is worth more than $1 a year from now, recognizing that today’s $1 can be invested and expected to earn interest (in a risk-free savings account, for instance), yielding $1,05 next year. In addition to profitability indicators of absolute returns (Net Present Value, the sum of discounted stream of net returns) and relative returns (Benefit-Cost Ratio), an Internal Rate of Return can be estimated that expresses the average annual interest rate yielded by the investment. This approach is appropriate when the costs or benefits extend beyond the current time period. Gittinger,79 an authority on CBA, offered an extensive explanation of the method and numerous examples.
Cost-benefit analysis tends to be used mainly for evaluating larger-scale interventions. Penne and D’Haese,178 for example, conducted a CBA of various ECF control options in Zambia. Benefits are estimated using a 30-year simulation model of traditionally managed cattle herds, and the results indicate that from an economic perspective, immunization makes more sense than chemotherapy or tick control. Stem231 applied this approach to evaluate a public vaccination campaign for peste des petits ruminants in Niger, and demonstrated the high economic return yielded by the intervention. Similarly, Perry et al.188 evaluated the returns to FMD eradication in Thailand, and found that such a programme would be justified if it led to export trade to highvalue markets elsewhere. Considering the case of a country with export trade already established, Berentsen et al.26 used CBA combined with simulation models to evaluate different control strategies if FMD were to be reintroduced into the Netherlands.
Economic surplus models
These are an extension of CBA in which the impact of intervention on supply and demand in markets for livestock and livestock products is captured. This can be important; if a given intervention has broad impact and results in avoiding mortality, the supply of meat will increase, lowering the relevant equilibrium market prices and therefore the estimated value of the benefits from the intervention. Changing prices and amounts marketed also translate into changes in consumer and producer surplus, and these can be quantified in the model to measure the welfare gains and losses to each group. Kristjanson et al.111 used this approach to evaluate the potential impact of a vaccine against trypanosomosis, and predicted that due to lower market prices, consumers will reap 55 per cent of the estimated US$ 1,3 billion in net benefits generated by the vaccine, versus 45 per cent for producers, who gain from lower production costs. The impact of the recently completed Pan-African Rinderpest Campaign was similarly analysed.234
Decision analysis
This is particularly useful for disease control interventions that involve diseases with variable risk of occurring, multiple possible outcomes, or a sequence of actions. This technique estimates the expected net returns for a set of actions (decisions) and outcomes by conditioning each associated cost and return by their probability of occurring.
Optimization models
These models use information on a household’s or firm’s resource base, market prices, and input–output relationships for its production activities to solve for the resource allocation and mix of production activities that will maximize some defined objective, typically farm or firm profits. One class of models is based on mathematical programming techniques (e.g. linear programming, dynamic programming).
Building a mathematical programming model is quite demanding in terms of data and research sophistication. For this reason, such models are used primarily for regional or national analyses in which models are developed for representative farms or herds; they are not practical tools for livestock producers themselves. Researchers at Wageningen University in the Netherlands have developed a number of such models for intensive commercial systems to advise on optimal timing for disease-related culling and stock replacement actions.95, 101 In a particularly innovative policy study looking at farmer incentives to participate in various components of bovine tuberculosis control in New Zealand, Bicknell et al.27 demonstrated how a mathematical epidemiological model can be transformed into a dynamic bio-economic optimization model. Applications for developing country situations are much less common. An exception is a study by Hall et al.85 in which a dynamic programming model was formulated to evaluate optimal ECF control strategies (tank dipping, immunization) in communal herds in Malawi over a five-year period.
The other main class of optimization models is based on econometric approaches, but these have been used little to date in the field of animal health economics. Chilonda and van Huylenbroeck,40 however, suggest how an agricultural household model225 might be adapted to examine smallholder farmer management behaviour for livestock health.
Simulation models
These models have been used extensively in animal health economics, but are usually limited to biological processes and applied to measure the impact of different control scenarios as an auxiliary component within an economic analytical technique, such as CBA or optimization modelling. As described above, analysts often include some type of herd population dynamics simulation model in order to capture benefits of disease control over time, and nearly all of the studies noted in the preceding paragraphs contain such a model. In some cases, though, simulation models have directly integrated financial or economic parameters as well. Such models are often packaged as highly specialized management software for intensive commercial production systems. Examples include PorkCHOP,51 PigORACLE and DairyORACLE,142 and DairyMAN.92 Similar models are rare for smallholder livestock production systems in developing countries, which can be attributed to a large degree to their lower commercial orientation and the difficulties of modelling the multiple roles of livestock in such systems. Exceptions are Nyangito et al.166 and Nyangito et al.,167 which model the complete farm household economy rather than the livestock production system alone to evaluate the financial and economic impacts of immunization against ECF on smallholder dairy farms in Kenya.
While certainly not exhaustive, this brief review of economic analytical techniques and examples from the animal health economics literature should give the reader an appreciation for the broad range of situations and types of decision- making to which economic analysis can be applied. Many relevant economic tools have yet to be applied or appropriately adapted to animal disease control problems, particularly in the developing world. A number of opportunities and some of the challenges that remain to make economic assessments more meaningful for effective decisionmaking are discussed in Perry and Randolph.183
Estimating the cost of a disease
Not mentioned above are a number of studies that have estimated the economic cost of a given disease, usually at national or regional level. Examples include Mukhebi et al.156 for ECF in eastern and southern Africa, Freyre et al.67 for toxoplasmosis in Uruguay, and Ngategize et al.163 for ovine faciolosis in Ethiopia. As cautioned by Perry and Randolph, 183 and earlier by McInerney,133, 134 such estimates may provide an indication of the relative importance of a disease, but will be misleading for decision-making about investing in its control unless this preliminary analysis is then supplemented by information and analysis of the costs and realizable benefits of the control itself. The point here is that the economic cost of the disease never equals the potential benefit to control, even in the case of eradication (e.g. does not capture changes in control costs post-eradication), and in any case, the cost of controlling disease also needs to be factored into the analysis. Estimates of the economic cost of a disease should be viewed as only a preliminary step towards a proper assessment of a control intervention.
Reliability of economic analysis
Economic criteria should be critical to responsible decisionmaking about disease control. But with what degree of confidence can we trust the results of economic analysis? How reliable are they? Schepers and Dijkhuizen218 describe, for example, the sometimes contradictory conclusions about mastitis control from different published studies. There are two key issues to consider in answering these concerns.
First, the quality of the analytical results is obviously largely a function of the quality of the data used, with key epidemiological parameters playing a particularly important role. ‘Garbage in, garbage out’ is the oft-quoted adage. Sufficiently high-quality data are often not available, however, and the needed primary data collection is not likely to be feasible due either to resource or time constraints. Welldesigned sensitivity analysis therefore is critical to evaluating the robustness of the quantitative results. This may be accomplished either by estimating results for a wide range of potential scenarios—rather than limiting the analysis to only one or two — or by changing values of individual key parameters that tend to drive the analysis. Stochastic models build this type of sensitivity analysis directly into the analytical model, and researchers have found the spreadsheetbased tools of @RISKTM (Palisade Corporation) particularly useful for this purpose since it allows the analyst to simultaneously represent multiple key parameters by different types of probability distributions. These types of sensitivity analyses can be very powerful in estimating notional confidence intervals for quantitative results.
Sensitivity analysis alone, however, does not ensure reliable results. Even if the data are accurate, the conclusions from the economic analysis may be flawed if the scope of the analysis is too narrow. Unless all potential impacts of disease control — both in terms of costs and benefits — have been captured, then the analytical results will be inaccurate. It is essential that the analysis be as comprehensive as possible to consider and quantify where possible the full range of impacts. Studies are usually good at identifying and measuring the obvious direct impacts, but are often less ambitious about addressing those that are indirect or secondary. Thus, while sensitivity analysis can compensate for certain data inadequacies, attention also needs to be paid to the comprehensiveness of the analysis of impacts.
How can control options be best implemented?
Once the impacts of an infectious disease and its potential control have been evaluated and the optimal control strategy decided, what remains is to implement the control measures. Despite improving knowledge of disease dynamics and the availability of better control technologies, the record of disease control has often been disappointing. This section explores a range of issues that need to be considered for effective disease control.
The historical context
In the introduction to this chapter, reference was made to the early efforts to eradicate ECF in southern Africa. At that time, how disease control should be implemented was clear. The priority was to address the threat of particularly contagious diseases and their devastating impact by imposing authoritarian measures on all livestock keepers for the public good. The need to control the major epidemic diseases was in fact the primary impetus for the creation of state veterinary services in Africa.99 For these diseases, the technical solution proposed by the veterinary services probably coincided fairly well with what was economically justified.
As the threat of epidemic disease was brought increasingly under control, public veterinary services continued to expand their bureaucracies by extending their mandates to protecting commercial livestock production from the reintroduction of epidemic diseases or the introduction of new exotic diseases, and by beginning to address zoonotic and endemic diseases. With the shift towards endemic diseases, disease control became less a question of ensuring compliance to imposed control interventions and increasingly one of encouraging farmer-adoption of control technologies. The combination of centrist state policies and the shortage of trained human resources meant, however, that disease control decision-making and implementation continued to be concentrated within state veterinary services, often discouraging the development of private sector provision. In this context, successful implementation of disease control was usually measured in terms of cost effectiveness.
The public fiscal crises experienced by African countries beginning in the 1970s led to pressure on state veterinary services to reduce costs and activities. This coincided with growing acceptance of the paradigm that the private sector can provide many types of disease control more efficiently and effectively than the public sector.94 As a result, efforts were encouraged to privatize livestock health services and shift the responsibility for much of the decision-making and cost for disease control to livestock keepers themselves.219 Such efforts have been undertaken to varying degrees and with varying levels of commitment in most, if not all, African countries.
The general consensus is that rather than improve the situation, the reduction of state services and privatization have instead reduced capacity to control diseases effectively in Africa.136, 219 These trends have prompted research and development efforts to better understand how to improve the design and performance of delivery systems for implementing disease control.
Delivery systems
A ‘delivery system’ is now commonly accepted as a generic term for describing how disease control is implemented, whether it involves a one-time public-managed diseaseeradication campaign or the sustainable provision of veterinary drugs for managing endemic diseases. In Figure 9.12 the basic components of a delivery system are presented.
Disease control involves two main inputs — veterinary products and knowledge — that are delivered or administered by livestock health services. The supply and distribution of the inputs, as well as the provision of services, may be the responsibility of various combinations of actors in the public, private, or civic sectors. A campaign to eradicate an epidemic disease may be predominantly public sector, with vaccine, manufactured by a parastatal agency, being administered free of charge by government veterinarians based on surveillance information and guidelines issued by publicly funded research institutes. Anthelmintics, on the other hand, are more likely to be produced and distributed primarily through commercial channels, although recommendations on their appropriate use may be disseminated through public extension services. The end-user is the livestock keeper, and which livestock keepers are concerned by a given disease control technology will depend not only on disease incidence, but also on the type of livestock production system as well as their perceptions of the disease and the utility of control. The institutional environment is also an integral component of a delivery system by conditioning the infrastructure and rules by which inputs and services can be provided.
For a specific disease control action, the appropriate composition of the generic delivery system described in Figure 9.12 will depend on the nature of both the disease and the control strategy:
- The nature of the disease
In Table 9.4 diseases are classified into six categories according to the type and scope of impacts they engender. The two key characteristics distinguishing these categories are: (1) contagiousness — to what degree the disease is limited to the affected animal or herd or is likely to spread to other animals and beyond; and (2) whether humans are affected. These characteristics have important implications for deciding who has responsibility for deciding the control response and its implementation. - The nature of the control strategy
Several different control options may be available for controlling a disease, each having its particular technical requirements and economic characteristics. As an example of a purely technical characteristic, the thermostability of a vaccine determines whether or not the vaccine requires delivery structured around a rigorous cold chain. Technical properties of a control option may also condition its economic characteristics. From the livestock keeper’s perspective, incentives for using a control option meant to protect his/her individual animal from a disease may be obvious, but it is less so for a control option that targets protection at the population level. Whether a control strategy is primarily curative rather than preventive is also important.
Due to the various technical and economic characteristics of a disease and control strategy, there is a range of approaches used, as illustrated by the dotted arrows in Figure 9.12. At one extreme, state veterinary services may try to impose control, say, to limit an outbreak of a highly contagious disease (a public good) that requires livestock keepers to respect a quarantine order. In this case, successful disease control is associated with ensuring compliance. At the other extreme, a livestock keeper’s willingness to pay a private veterinarian to care for an animal is an example in which successful disease control depends on the livestock keeper’s demand rather than compliance. Many disease control interventions represent a mix somewhere between these two extremes.
Improving delivery
Productivity remains well below potential in the majority of livestock production systems in the developing world, and disease continues to be a critical constraint despite the availability, in many cases, of appropriate control technologies. Inadequate health management is often attributed to poor delivery of control technologies. A number of research and development efforts have been devoted to identifying opportunities for improving delivery systems and effective use of disease control technologies. Many of these efforts target specific components of delivery systems as presented in Figure 9.12.
Better products
Certain characteristics of a control technology can constrain its delivery and adoption. The classic example is that of veterinary products that require a cold chain, i.e. that must be kept refrigerated at low temperatures to remain viable. Lack of infrastructure, especially in areas where livestock keepers are widely dispersed or mobile, has made delivery of such products problematic in much of Africa. One way to overcome these constraints is to adapt veterinary products accordingly. An important theme has been the development of thermostable products, particularly vaccines such as those against rinderpest, 141 Newcastle disease,2, 25, 230 and ECF.160 Another has been simplifying techniques of administration to make control more accessible or improve coverage, such as coating chicken feed with thermostable Newcastle disease vaccines or administering them orally using a simple eyedropper. 25 In the case of trypanosomosis control, the development of the Sterile Insect Technique, though very expensive to implement, avoids need for farmer involvement and many of the environmental drawbacks of alternative vector control options.3
Veterinary products can also be improved in terms of their economic characteristics, especially their price. Affordability can be enhanced by more efficient production and distribution that lower cost either through technical or institutional improvements. For products that provide significant benefits beyond the farm where they are used, public subsidies can directly reduce the nominal cost borne by end-users and thereby encourage uptake.57 Increasing the effectiveness, safety, or ease of application of veterinary products also has the effect of reducing economic risk by minimizing productivity losses and wasted investment when the products do not work to satisfaction.
Better information
As already noted in previous sections, epidemiological information is critical for evaluating when and how much disease control is appropriate.189 Improving the availability and quality of epidemiological information enables delivery systems to target their interventions better and hence become more effective and efficient in their provision of control. Sanson and Thornton,217 for example, estimated that diagnosis (and consequently control) of salmonellosis after its introduction into New Zealand would be delayed by eight months if the contemporary animal health surveillance activities were reduced. Efforts to improve animal health information, whether through more effective information systems and networks,46, 89, 170 improved diagnostic tools such as user-friendly pen-side tests, or soliciting information directly from farmers34 can therefore be expected to contribute to more effective implementation of disease control strategies.
Better information is needed not only to know when to implement control, but also how to implement it properly. Kenyan farmers, for example, report that the lack of printed information or advice on using anthelmintics contributes to under- or overdosing, and other inappropriate practices, including the use of substandard or counterfeit products.250 Both the private and public sectors are involved in generating and providing information using a variety of media, often through advertising or extension services, and targeted at both service providers and the farmers themselves. Research is now needed to explore such information channels and how their effectiveness can be improved.
Better services
Initially, improving the service component of disease control was largely concentrated on upgrading human resources through training and, more specifically, graduating adequate numbers of veterinarians. More recently, Kleeman and others have begun examining the opportunities for improving the quality of services through better management, and to this end Kleeman108 proposes a number of techniques as a ‘toolkit’ for analysing services and their efficiency.
Providing adequate livestock health services in areas where livestock production is not market-oriented, or production units are relatively few, dispersed or mobile—especially among pastoralists — has presented a particular challenge. Such areas often do not offer sufficient financial incentives to support private veterinary practice, and in the past depended largely on state-provided services. As state veterinary services have declined over the past three decades, a number of research-development efforts have experimented with community-based systems that could provide basic care in the absence of formal veterinary services. 33, 94 These schemes typically involve the training of a local community member as a Community-Based Animal Health Worker to provide limited paraveterinary services paid for by the community. This approach is being tested for example to provide veterinary care to backyard poultry producers.2
Under standing demand
As implementing disease control shifts from the public to the private sector, delivery systems need increasingly to take into account the particular requirements of livestock keepers and their production systems. This is important because failure to properly evaluate farmer demand risks undermining the financial viability and longer-term sustainability of livestock health services. Animal health information systems provide only one component of farmer demand for control: disease incidence and prevalence. How livestock keepers respond to disease risk will also depend on the role that livestock play in their household economy, their perceptions and understanding of both the disease and the control strategy, their resource levels, and the relative costs and benefits of using control. Evaluating costs and benefits to support decision-making have been discussed above. Various approaches are being developed and used to assess the other various components of demand.
One approach is to solicit needs directly from livestock keepers using participatory techniques. Many of the tools developed to date including diagramming, mapping, scoring, ranking, and interviewing techniques, often used in conjunction with establishing community-based livestock health services have been summarized by Catley.36
The other principal approach is based on demand analysis techniques from the field of economics. Several of these techniques and their application to livestock health services in India, including assessing farmers’ willingness to pay (WTP) for specific services and products have been described.1 As pressure has mounted to ensure cost recovery for disease control, it has become evident that better techniques are needed to measure WTP. One of these techniques draws upon WTP methodologies used in market research and contingent valuation adapted to livestock health services.1 This technique has been used to evaluate the willingness of livestock keepers in Kenya to contribute their labour and money to maintaining tsetse traps to control animal trypanosomosis.56 Data collection and econometric analyses for other factors such as farmer and community-level characteristics that influence farmer demand have also been reported.1 In a similar analysis, Swallow et al.233 evaluated demand for pour-ons for trypanosomosis control in Ethiopia. Another technique adapted from marketing research is conjoint analysis used to assess consumer preferences regarding specific attributes of a disease control strategy.202 Each technique is likely only to capture one dimension of farmer demand, so several may be needed in combination.
Better institutional environment
The institutional environment refers to policies that dictate or influence which actors are involved in disease control, the roles they play, and the rules they work under. As noted, the current institutional environment governing veterinary delivery systems in developing countries has been conditioned to a large extent by external macroeconomic trends over the past three decades (especially public fiscal stabilization programmes) that have reduced capacity of state veterinary services and encouraged privatization and decentralization of livestock health services. These changes have occurred at a variable pace across Africa, and have created considerable confusion regarding public and private sector roles in disease control and the appropriate accompanying adjustments in the regulatory environment. The result has been considerable research devoted to developing a framework for evaluating and improving the institutional environment for delivery systems.
The evolving framework draws upon economic theory, particularly the New Institutional Economics, to characterize the underlying economic characteristics and incentives inherent in disease control products and services. Central to this type of analysis are the concepts of externalities and public goods. An externality occurs when one person’s use of goods or a service generates impacts — either negative or positive — on other people. If these impacts are not properly captured in the market cost of the good or service, then the consumer is likely to use either too little or too much from a societal welfare perspective, leading to what economists term as market failure. Market failure occurs when markets are unable to provide certain goods or services in the desired quantity or quality. Public institutions have evolved primarily in response to such market failures, and to take corrective action by providing public goods. For example, smaller livestock keepers are likely to ‘under- invest’ in vaccination against a contagious disease, and so public veterinary services may be justified in undertaking a vaccination campaign to ensure adequate population coverage. Externalities related to disease control are generally a function of the contagiousness of the disease and its zoonotic impacts. In the absence of externalities, economic theory dictates that markets (i.e. the private sector) are more efficient and cost-effective in providing disease control products and services than the public sector.
Evaluating the degree to which a given disease control action is associated with externalities thereby provides the basis for judging whether it is best left to the private or public sector to deliver. Two criteria are often used: excludability and rivalry (see definition in Figure 9.13). As shown in Figure 9.13, veterinary products and services can then be classified as public, toll, common pool, and private goods, each having implications for the appropriate actor to implement, or at least finance, the disease control action.94 A government supporting a vaccination campaign, for example, may find it more efficient to contract services from private veterinarians rather than setting up and managing its own vaccination teams.
The manner in which this framework could be applied to veterinary services was demonstrated in Kenya118 and then in Africa more generally.119 It was also developed as a methodology for classifying individual livestock services.243 In addition, an excellent overview of progress to date, including a number of examples of how this approach is being used to address specific issues and suggest solutions, has been published.94 It is not clear yet what impact this research has had on public veterinary policy.
Assessing performance
A general approach for assessing the overall performance of delivery systems for disease control is still lacking. Researchers at Reading University in the UK are in the initial stages of developing an approach based on the following four criteria:136
- accessibility, referring to the willingness of providers to supply disease control and time taken to find a service provider or product when needed;
- affordability, the actual cost as well as ability and willingness of livestock keepers to pay;
- acceptability, the form and manner in which the product or service is presented; and
- sustainability, both financial and institutional.
These criteria capture a number of the elements discussed in the preceding paragraphs. The challenge now remains to identify indicators that can be used to evaluate these criteria.
Research at the ILRI is currently exploring two additional techniques for assessing delivery systems.162 The first draws upon the Structure-Conduct-Performance paradigm from agricultural markets research, which compares delivery systems to what might be expected in a perfectly competitive market, especially in terms of the nominal cost of the final product. Using this technique, delivery systems are examined for evidence, for example, of monopoly power, barriers to entry, or inflated marketing margins that would likely contribute to inefficiency.
The second technique, based on another theme from the New Institutional Economics called Transaction Costs Analysis, shifts the focus to non-price factors that may contribute to poor delivery. Asymmetric information between different actors in a delivery system, for example, gives rise to a number of potential transaction costs. A livestock keeper has difficulty judging whether a veterinarian is treating his/her animal correctly, andtherefore faces an added risk—andadded cost—of losing the animal plus the value of the treatment. An effective delivery system is one that minimizes such transaction costs to the extent possible.
Institutionalizing impact assessment: the role of epidemiology and economics
The effective and optimal use of epidemiology and economics in impact assessment, national disease control planning and implementation depends very much on how the use of the discipline is ‘operationalized’. There has been a fairly wide range of modalities tried in different countries, some of which have been highly successful and have endured, some have struggled, and some have been quite transient. As epidemiologists and economists, it is important to evaluate what has contributed to these different outcomes, and learn from the successes and failures in the adoption of our discipline in the past.
Figure 9.13 Two key economic characteristics of disease control (source: Holden, 199994)
* Excludability: the ability of the user to exclude others who have not paid from benefiting from the user’s investment in a good or service
Rivalry: consumption of a good or service by one user precludes its use by other users
Possibly the greatest success in the field of epidemiology and economics has been in research. During the last two decades there has been a steady improvement in the focus, relevance and scientific quality of publications in the field of veterinary epidemiology and economics. This has been particularly important in the development, testing, validation and application of a variety of methodologies, which have very much consolidated the acceptance of veterinary epidemiology and economics as a bona fide discipline.
It is important to acknowledge the pivotal role that has been played in this process by the journal Preventive Veterinary Medicine. This progress has been made by individuals and groups in many countries of the world. During 1998, for example, the distribution of manuscript submissions to this journal was roughly one-third from Europe, one-third from the Americas, and one-third from the rest of the world.149 But what impact has this scientific success had beyond the research community? In many countries of the developed world, it had a significant impact. There is evidence that it has contributed, for example, to the development of several national disease surveillance programmes, such as the National Animal Health Monitoring System (NAHMS) in the USA, and to the teasing out of health and management constraints at the farm level that has probably had major impacts on national livestock productivity, particularly in the more intensive dairy, feedlot beef, poultry and pig production systems. It has also had an important effect on policy and strategy development for national disease control, for example in the work of Dijkhuizen and his colleagues in the Netherlands on FMD and classical swine fever. In most countries of the developing world, however, the impact has been quite different, and much more modest. There is definitely a raised awareness, and the value of epidemiology units as advisers to directors of veterinary services has been recognized by many donors who have supported training programmes. As a result, there has been a significant development of human capacity. The major obstacles to a more active adoption of the epidemiological and economic tools developed have been a lack of political understanding of the potential returns from their use, and lack of resources to allow the discipline to be effectively institutionalized and practised by its new disciples.
What are the necessary conditions for the effective adoption and use of the research products of veterinary epidemiology and economics, from journals such as, Preventive Veterinary Medicine, among many others? First, there have to be trained epidemiologists and animal health economists in a given country. In many developing countries, such people, if they exist, do not exceed a handful at most. There is probably a highly skewed geographical distribution of trained epidemiologists, depending in many cases on the previous existence of technical co-operation projects in which capacity building in the field of veterinary epidemiology was a priority. Next, there need to be positions earmarked for epidemiologists and economists in animal health research and development institutions. In most developing countries these would be unlikely to exceed one, if any. Thirdly is the condition that there must be adequate funding for this person to operate effectively, including access to journals, e-mail and Internet. Public sector funding for veterinary epidemiology and economics has had to compete with more operational aspects of veterinary services, and during the structural adjustment process undertaken by many countries, they have suffered badly. As far as access to electronic media is concerned, we were all made very aware of what was labelled at a recent G8 summit in Okinawa as the ‘digital divide’. It is not only the high cost of, and thus poor access to, computers, but also the inadequate telecommunications infrastructures, and the high cost of access to them. The bottom line is that the adoption and impact of scientific developments in our field leaves much to be desired in regions where this impact is needed most. This is particularly the case in Africa.
On the developmental side in which epidemiology and economics function as a component of a national veterinary service, various modalities have been used to integrate the discipline into national disease-control bodies. These include institutionalizing it in departments of veterinary services or in veterinary research institutes (or both), and the development and use of university departments, particularly those with a mandatory ‘extension’ function. There are a few ‘classics’ around the world that may serve as role models for some sets of circumstances (but not all), and the Veterinary Epidemiology Department at the Veterinary Laboratories Agency (formerly the Central Veterinary Laboratory) in Weybridge, UK, is certainly one of those national epidemiology units that has endured and gained an international reputation for focus and functionality. In the developing world, epidemiology and economics have been the ‘flavour of the moment’ in many countries, but while there are several success stories, epidemiology units in many developing countries have generally suffered from a lack of sustainability. Thus, in Africa, where epidemiology has been recognized as playing an important role, given the history of a population, rather than individual animal, approach to disease control, national units have been set up in several countries such as Zambia, Botswana, Nigeria, Zimbabwe, Tanzania, Uganda and Kenya, but their functionality and impact have ebbed and flowed due to their heavy reliance on donor agencies, whose support has varied over the years. However, on the bright side, there has been a considerable development of human capacity during this time, which bodes well for the future. In other parts of the developing world there are some examples of new national indigenous units, supported by funds from their own governments, that appear to be increasing in their impact; these include groups in Thailand, Colombia and Chile.
In all of the developing, as well as much of the developed, world, units or groups have tended to be led by epidemiologists, who have been more comfortable developing the technical dimensions. As a result, there has been a reluctance on the part of both research and development groups to employ economists, so severely limiting potential impact of the discipline. The Veterinary Epidemiology and Economics Research Unit at Reading University in the UK is one of the few academic institutions that truly incorporates both disciplines. Public sector funding of staff positions is in short supply, and economists are considered a lower priority human resource that cannot be afforded.
International organizations also have epidemiology capacity that may play a role in national animal disease control. The FAO has a small epidemiology group within its animal health service in the Rome headquarters, and individual epidemiologists and animal health economists in its many field projects. In addition, the OIE has an epidemiologist at the headquarters in Paris. Somewhat surprisingly for such prominent international organizations, neither has an effective integration of epidemiology and economics, nor do they employ agricultural economists within their animal health staff groupings. The ILRI, an International Agricultural Research Centre of the Consultative Group on International Agricultural Research, has an epidemiology and economics group, the mandate of which is entirely research. At a regional level, some epidemiology and economics capacity exists in the African Union Inter- African Bureau for Animal Resources, and this capacity is currently being enhanced with support from the European Union.
Disappointingly, most of these international and regional units are driven, in the traditional disease-control manner, by technical issues, and the small economics components, if they exist at all, are more of an add-on to provide the necessary figures to justify programmes. Here is a real opportunity for more integration to provide better support to strategic decision-making.
References
- AHUJA, V., GEORGE P.S., RAY, S., MCCONNELL, K.E., KURUP, M.P.G., GANDHI, V., UMALI-DEININGER, D. & DE HAAN, C., 2000. Agricultural services and the poor: case of livestock health and breeding services in India. Indian Institute of Management, Ahmedabad.
- ALDERS, R.G., 2001. Sustainable control of Newcastle disease in rural areas. In: ALDERS, R.G. & SPRADBROW, P.B., (eds). SADC planning workshop on Newcastle disease control in village chickens. Proceedings of an International Workshop, Maputo, Mozambique, 6–9 March 2000. ACIAR Proceedings. No. 103. pp. 80–90.
- ALLSOPP, R., 2001. Options for vector control against trypanosomiasis in Africa. Trends in Parasitology, 17, 15–19.
- ANAND, S. & HANSON, K., 1997. Disability adjusted life years: a critical review. Journal of Health Economics, 16, 685–702.
- ANDERSON, R.M., 1982. Population Dynamics of Infectious Diseases. London: Chapman and Hal.
- ANDERSON, R.M., 1982. Transmission dynamics and control of infectious disease agents. In: ANDERSON, R.M. & MAY, R.M., (eds). Population Biology of Infectious Diseases. Berlin: Springer-Verlag.
- ANDERSON, R.M. & MAY, R.M., 1991. Infectious Diseases of Humans— Dynamics and Control. Oxford: Oxford University Press.
- ANON., 1993. Botswana Agricultural Census Report, 1993. pp. 185.
- ANON., 1995. 1995 Swaziland Livestock Census. Data Processing Unit, Ministry of Agriculture and Co-operatives, Kingdom of Swaziland. Mbabane. pp. 44..
- ANON., 1996. Angola Livestock Population Data. Computer files. National Institute of Statistics (INE). Luanda.
- ANON., 1996. National Sample Census of Agriculture 1994/95, Tanzania Mainland. Volumes I and II. Statistics Unit, Ministry of Agriculture and Bureau of Statistics, Planning Commission, The United Republic of Tanzania. Dar es Salaam. pp. 33.
- ANON., 1997. Zambia Livestock Population Data. Electronic data files. Epidemiology Unit, Department of Animal Production and Health, Government of Zambia. Lusaka.
- ANON., 1997. Relatório Annual, 1996. Direcçao Nacional de Pecuária, República de Moçambique. Maputo. pp. 28 plus appendices.
- ANON., 1997. Abstract of Agricultural Statistics. Directorate, Agricultural Statistics and Management Information, National Department of Agriculture, Republic of South Africa, Pretoria. pp. 117.
- ANON., 1998. National Livestock Development Master Plan. National Livestock Development Master Plan Task Force, Department of Animal Health and Industry, Ministry of Agriculture and Irrigation, Government of Malawi. Lilongwe. pp. 71.
- ANON., 2000. Zimbabwe Livestock Population Data. Computer files. Department of Veterinary Services, Harare.
- ASHFORD, R.W., 1998. The leishmanises. In: PALMER, S.R., LORD SOULSBY & SIMPSON, D.I.H., (eds). Zoonoses: Biological, Clinical Practice and Public Health Control. Oxford: Oxford University Press: pp. 527–544.
- BACON, P.J. & MACDONALD, D.W., 1980. To control rabies: vaccinate foxes. New Scientist, 87, 640–645.
- BALTENWECK , I. & STAAL, S., 2001. Unpublished research report. ILRI, Nairobi.
- BAPTIST, R., 1988. Herd and flock productivity assessment using standard offtake and the demogram. Agricultural Systems, 28, 67–78.
- BARENDREGT, J.J., BONNEUX, L. & VAN DER MAAS, P.J., 1996. DALYS: the age-weights on balance. Bulletin of the World Health Organisation, 74, 439–43.
- BARKER, C. & GREEN, A., 1996. Opening the debate on DALYs. Health Policy and Planning, 11, 179–183.
- BASSETT, T.J., 1993. Land use conflicts in pastoral development in northern Côte d’Ivoire. In: BASSETT, T.J. & CRUMMEY, D.E., (eds). Land in African Agrarian Systems. Madison, Wisconsin, USA: University of Wisconsin Press. pp. 131–154.
- BECKETT, S., 1997. An approach to meta-analysis: post-partum GnRH and the reproductive performance of dairy cows. Epidemiologie et Sante Animale, 31–32:13.03.1–3.
- BELL, J.G., 2001. Comparison of the different vaccines available for the control of Newcastle disease in village chickens. In: ALDERS, R.G. & SPRADBROW, P.B., (eds). SADC planning workshop on Newcastle disease control in village chickens. Proceedings of an International Workshop, Maputo, Mozambique, 6–9 March 2000. ACIAR Proceedings. No. 103. pp. 56–60.
- BERENTSEN, P.B.M., DIJKHUIZEN, A.A. & OSKAM, A.J., 1992. A dynamic model for cost-benefit analysis of foot-and-mouth disease control strategies. Preventive Veterinary Medicine, 12, 229–243.
- BICKNELL, K.B., WILEN, J.E. & HOWITT, R.E., 1999. Public policy and private incentives for livestock disease control. Australian Journal of Agriculture and Resource Economy, 32, 501–521.
- BOKELLA, J.G., 1997. Beef, Sheep and Goats Marketing Review, 1996/97. Planning and Marketing Department, Marketing Development Bureau, Ministry of Agriculture and Co-operatives. Dar es Salaam. pp. 25.
- CAMERON, A.R. & BALDOCK, F.C., 1998A. A new probability formula for surveys to substantiate freedom from disease. Preventive Veterinary Medicine, 34, 1–17.
- CAMERON, A.R. & BALDOCK, F.C., 1998B. Two-stage sampling in surveys to substantiate freedom from disease. Preventive Veterinary Medicine, 34, 19–30.
- CANNON, R.M. & ROE, R.T., 1982. Livestock Disease Surveys: A Field Manual for Veterinarians. Canberra: Australian Government Publishing Service.
- CARNEY, D., 1998. Sustainable rural livelihoods: What contribution can we make? London: Department for International Development.
- CATLEY, A., 1996. Pastoralists, paravets and privatization: experiences in the Sanaag region of Somaliland. Pastoral Development Network Paper. No. 39. London: Overseas Development Institute.
- CATLEY, A., 1997. Adapting participatory appraisal (PA) for the veterinary epidemiologist: PA tools for use in livestock disease data collection. In: GOODALE, A. & THRUSFIELD, M.V., (eds). Society for Veterinary Epidemiology and Preventive Medicine. Proceedings of a Meeting held at University College, Chester. 9–11 April 1997. pp. 246–257.
- CATLEY, A., 1998. Non-governmental organizations (NGOs) and the delivery of animal health services in developing countries. A Discussion Paper for the Department for International Development. Edinburgh: United Kingdom. Vetwork UK.
- CATLEY, A., 1999. Methods on the move: a review of veterinary uses of participatory approaches and methods focusing on experiences in dryland Africa. London: International Institute for Environment and Development.
- CATLEY, A., 1999. The herd instinct—Children and livestock in the Horn of Africa. London: Save the Children.
- CHAMBOKO, T., MUKHEBI, A.W., O’CALLAGHAN, C.J., PETER, T.F., KRUSKA, R.L., MEDLEY, G.F., MAHAN, S.M. & PERRY, B.D., 1999. The control of heartwater on large scale commercial and smallholder farms in Zimbabwe. Preventive Veterinary Medicine 39, 191–210.
- CHAMBOKO, T., MUKHEBI, A.W., O’CALLAGHAN, C.J., PETER, T.F., KRUSKA, R.L., MEDLEY, G.F., MAHAN, S.M. & PERRY, B.B., 1999. Heartwater and the economics of livestock production on large scale commercial and smallholder farms in Zimbabwe. Revue D’Élevage et de Médicine Vétérinaire des Pays Tropicaux, 52, 313–323.
- CHILONDA, P. & VAN HUYLENBROECK, G.A., 2001. A conceptual framework for the economic analysis of factors influencing decision-making of small-scale farmers in animal health management. OIE Scientific Technical Revue. 20, 687–700.
- COCHRAN, W.R., 1977. Sampling techniques. 3rd edition. New York: John Wiley.
- COLEMAN, P.G. & DYE, C., 1996. Immunization coverage required to prevent outbreaks of dog rabies. Vaccine, 14, 185–186.
- CUMMING, G.S., 1999. The evolutionary ecology of African ticks. Unpublished D. Phil. Thesis. University of Oxford, UK.
- CURRY, J.J., HUSS-ASHMORE, R.A., PERRY, B.D. & MUKHEBI, A.W., 1996. A framework for the analysis of gender, intra-household dynamics and livestock disease control, with examples from Uasin Gishu District, Kenya. Journal of Human Ecology, 24, 161–189.
- DE HAAN, C., 1995. Development support and livestock services. In: WILSON, R.T., EHUI, S. & MACK, S., (eds). Livestock Development Strategies for Low Income Countries. Nairobi: FAO/ILRI. pp. 23–27.
- DE LEEUW, P.N., MCDERMOTT, J.J. & LEBBIE, S.H.B., 1995. Monitoring of livestock health and production in sub-Saharan Africa. Preventive Veterinary Medicine, 25, 195–212.
- DEEM, S.L., PERRY, B.D., KATENDE, J.M., MCDERMOTT, J.J., MAHAN, S.M., MALOO, S.H., MORZARIA, S.P., MUSOKE, A.J. & ROWLANDS, G.J., 1993. Variations in prevalence rates of tick-borne diseases in Zebu cattle by agroecological zone: implications for East Coast fever immunisation. Preventive Veterinary Medicine, 16, 171–187.
- DEPARTMENT FOR INTERNATIONAL DEVELOPMENT (DFID), UK, 2000. Sustainable livelihood guidance sheets. (livelihoods@dfid.gov.uk).
- DELGADO, C., ROSEGRANT, M., STEINFELD, H., EHUI, S. & COURBOIS, C., 1999. Livestock to 2020: the next food revolution. Food, Agriculture and the Environment Discussion Paper 28. Washington D.C.: IFPRI/ILRI/ FAO.
- DICKERSIN, K. & BERLIN, J.A., 1992. Meta-analysis: state of the science. Epidemiology Reviews, 14, 154–160.
- DIJKHUIZEN, A.A., MORROW, M. & MORRIS, R.S., 1986. PorkCHOP user manual. University of Minnesota: Agricultural Extension Service.
- DIJKHUIZEN, A.A. & MORRIS, R.S., 1997. Animal Health Economics: Principles and Applications. Post Graduation Foundation in Veterinary Science. University of Sydney: Sydney South.
- DOLL, R. & HILL, A.B., 1964. Mortality in relation to smoking. Ten years observations of British doctors. British Medical Journal, 1, 1399–1410.
- DOUTHWAITE, R.J. & TINGLE, C.C.D., 1994. DDT in the Tropics: The Impact on Wildlife in Zimbabwe of Ground Spraying for Tsetse Fly Control. Chatham UK: Natural Resources Institute.
- DUCHATEAU, L., KRUSKA, R.L. & PERRY, B.D., 1997. Reducing a spatial database to its effective dimensionality for logistic regression analysis of livestock disease distribution data. Preventive Veterinary Medicine, 32, 207–218.
- ECHESSAH, P.N., SWALLOW, B.M., KAMARA, D.W. & CURRY, J.J., 1997. Willingness to contribute labor and money to tsetse control: application of contingent valuation in Busia District, Kenya. World Development, 25, 239–253.
- EKBOIR, J.M., 1999. The role of the public sector in the development and implemenation of animal health policies. Preventive Veterinary Medicine, 40, 101–115.
- EVANS, D.B. & HURLEY, S.F., 1995.The application of economic evaluation techniques in the health sector: the state of the art. Journal of International Development, 7, 503–524.
- EVERTS, J. W. & KOEMAN, J.H., 1987. The ecological impact of insecticides in connection to the control of tsetse flies in Africa: a review. In: CAVALLORO, R., (ed.). Integrated Tsetse Fly Control: Methods and Strategies. Proceedings of the CEC International Symposium/Ispra, 4–6 March 1986. Rotterdam: A.A. Balkema. pp. 49–56.
- FAO, 1994. Report of expert consultation on the need for information systems to strengthen veterinary services in developing countries. Rome: AGA-817, FAO.
- FEINSTEIN, A.R., 1985. Clinical epidemiology: the architecture of clinical research. Philadelphia: WB Saunders.
- FENNER, F., 1982. Global eradication of smallpox. Review of Infectious Diseases,4, 916–922.
- FLEISCHER, G. & WAIBEL, H., 2001. Elements of economic resistance management strategies—some empirical evidence from case studies in Germany. Economics of Resistance Conference held by Resources for the Future, Airlie House, Virginia, 5 April 2001.
- FEWS NET, 2000. Horn of Africa Food Security Update: October 20, 2000. http://www.fews.net/.
- FREECALC. 2001. Downloadable from http://epiweb.massey.ac.nz/ software.htm.
- FRENCH, N.P., WALL, R. & MORGAN, K.L., 1994. A simulation model of sheep blowfly strike caused by Lucilia sericata (Diptera: Calliphoridae). Kenya Veterinarian, 18, 379–381.
- FREYRE, A., BONINO, J., FALCON, J., CASTELLS, D., CORREA, O. & CASARETTO, A., 1997. The incidence and economic signifcance of ovine toxoplasmosis in Uruguay. Veterinary Parasitology, 73, 13–15.
- GARNER, M.G. & LACK, M.B., 1995.An evaluation of alternative control strategies for foot and mouth disease in Australia: a regional approach. Preventive Veterinary Medicine, 23, 9–32.
- GEERTS, S. & HOLMES, P.H., 1998. Drug management and parasite resistance in trypanosomes in Africa. PAAT Technical and Scientific Series 1. Rome: Food and Agriculture Organization.
- GETTINBY, G. & BYROM, W., 1989.The dynamics of East Coast fever: A modelling perspective for the integration of knowledge. Parasitology Today, 5, 68–73.
- GETTINBY, G. & MCCLEAN, S., 1979. A matrix formulation of the life cycle of liver fluke. Proceedings of the Irish Academy, 79B, 155–167.
- GETTINBY, G., BAIRDEN, K., ARMOUR, J. & BENITEZ-USHER, C., 1979. A prediction model for bovine ostertagiasis. The Veterinary Record, 105, 57–59.
- GIBSON, T.E., 1978.The ‘Mt’ system for forecasting the prevalence of fascioliasis. In: GIBSON, t.e., (ed.). Weather and Parasitic Animal Disease. World Meteorological Oraganization, Technical Note No. 159. Geneva, pp. 3–5.
- GIBSON, W., 1998. African trypanosomosis In: PALMER, S.R., LORD SOULSBY & SIMPSON, D.I.H., (eds). Zoonoses: Biological, Clinical Practice and Public Health Control. Oxford: Oxford University Press. pp. 501–512.
- GITAU, G.K., PERRY, B.D., KATENDE, J.M., MCDERMOTT, J.J., MORZARIA, S.P. & YOUNG, A.S., 1997. The prevalence of tick-borne infections in smallholder dairy farms in Murang’a District, Kenya: a cross-sectional study. Preventive Veterinary Medicine, 30, 95–107.
- GITAU, G.K., PERRY, B.D. & MCDERMOTT, J.J., 1999. The incidence, calf mortality and calf morbidity due to ,Theileria parva infections in smallholder dairy farms in Murang’a District, Kenya. Preventive Veterinary Medicine, 39, 65–79.
- GITAU, G.K., MCDERMOTT, J.J., KATENDE, J.M., O’CALLAGHAN, C.J., BROWN, R.N. & PERRY, B.D., 2000.Differences in the epidemiology of theileriosis on smallholder dairy farms in contrasting agro-ecological and grazing strata of highland Kenya. Epidemiology and Infection, 124, 325–335.
- GITAU, G.K., MCDERMOTT, J.J., MCDERMOTT, B. & PERRY, B.D., 2001. The impact of Theileria parva infection and other factors on calf mean daily weight gains in smallholder dairy farms in Murang’a District, Kenya. Preventive Veterinary Medicine, 51, 149–160.
- GITTINGER, R.P., 1982. Economic Analysis of Agricultural Projects. Baltimore, MD: Johns Hopkins University Press.
- GOODMAN, C., COLEMAN, P. & MILLS, A., 1999. Cost-effectiveness of malaria control in sub-Saharan Africa. Lancet, 354, 378–385.
- GOODMAN, C., COLEMAN, P. & MILLS, A., 2000. Economic Analysis of Malaria Control in Sub-Saharan Africa. Strategic Research Series, Global Forum for Health Research, WHO.
- GUMMOW, B. & MAPHAM, P.H., 2000. A stochastic partial-budget analysis of an experimental Pasteurella haemolytica feedlot vaccine trial. Preventive Veterinary Medicine, 43, 29–42.
- GÜRTLER, R.E., COHEN, J.E., CECERE, M.C., LAURICELLA, M.A., CHUIT, R. & SEGURA, E.L., 1988. Influence of humans and domestic animals on the household prevalence of Trypanosoma cruzi in Triatoma infestans populations in northwest Argentina. American Journal of Tropical Medicine and Hygiene, 58, 748–58.
- HALL, A.J., 1997. Editorial: Eradication or control? Tropical Medicine and International Health, 2, 413–414.
- HALL, D.C., KAISER, H.M. & BLAKE, R.W., 1998. Modelling the economics of animal health control programs using dynamic programming. Agricultural Systems, 56. 125–144.
- HABTEMARIAM, T.M., RUPPANNER, R., RIEMANN, H.P. & THEIS, J.H., 1983. An epidemiologic systems analysis model for African trypanosomiasis. Preventive Veterinary Medicine, 1, 125–136.
- HABTEMARIAM, T.M., RUPPANNER, R., RIEMANN, H.P. & THEIS, J.H., 1983. Epidemic and endemic characteristics of trypanosomiasis in cattle: A simulation model. Preventive Veterinary Medicine, 1, 137–145.
- HABTEMARIAM, T.M., RUPPANNER, R. , RIEMANN, H.P. & THEIS, J.H., 1983C. Evaluation of typanosomiais control alternatives using an epidemiologic simulation model. Preventive Veterinary Medicine, 1, 147–156.
- HARE, K.M. & BIGGS, H.C., 1996. Design and evaluation of a veterinary information system for Namibia. Preventive Veterinary Medicine, 26, 239–251.
- HATHAWAY, S.C., 1993. Risk analysis and meat hygiene. O.I.E. Scientific and Technical Revue, 12, 1265–1290.
- HAY, S.I., RANDOLPH, S.E. & ROGERS, D.J., 2000. Remote sensing and geographical information systems in epidemiology. Advances in Parasitology, Vol. 47. London: Academic Press.
- HAYES, D.P., PFEIFFER, D.U. & MORRIS, R.S., 1998. Production and reproductive response to use of DairyMAN: A management information system for New Zealand dairy herds. Journal of Dairy Science, 81, 2362–2368.
- HEFFERNAN, C. & MISTURELLI, F., 2000. The delivery of veterinary services to the poor: preliminary findings from Kenya. Report of DFID project R7359. Reading, UK: VEERU.
- HOLDEN, S., 1999. The economics of the delivery of veterinary services. In: PERRY, B.D., (ed.). The Economics of Animal Disease Control. OIE Scientific and Technical Review, 18, 425–439.
- HOUBEN, E.H.P., 1995. Economic optimization of decisions with respect to dairy cow health management. PhD Thesis, Wageningen Agricultural University.
- HUGH-JONES, M.E., ELLIS, R.R. & FELTON, M.R., 1975. An Assessment of the Eradication of Bovine Brucellosis in England and Wales. Study No. 19, University of Reading.
- HUSS ASHMORE, R. & CURRY, J.J., 1992. Impact of improved livestock disease control on household diet and welfare: a study in Uasin Gishu District, Kenya. ILRAD Technical Report No. 2. Nairobi, Kenya: International Laboratory for Animal Diseases
- ILCA, 1978. Mathematical Modelling of Livestock Production Systems: Application of The TexasA&MUniversity Beef Cattle Production Model to Botswana. ILCA Systems Study No.1. Addis Ababa: International Livestock Centre for Africa (ILCA), pp. 102.
- ILCA, 1979. Towards an economic assessment of veterinary inputs in tropical Africa. ILCA Working Document 1. Addis Ababa. International Livestock Centre for Africa (ILCA).
- ITTY, P., CHEMA, S., D’IETEREN, G.D.M., DURKIN, J., LEAK, S.G.A., MAEHL, J.H.H., MALOO, S.H., MUKENDI, F., NAGDA, S.M., RARIEYA, J.M., THORPE, W. & TRAIL, J.C.M., 1988. Economic aspects of cattle production and of chemoprophylaxis for control of trypanosomiasis in village East African Zebu cattle in Kenya. In: Livestock Production in Tsetse-Affected Areas of Africa. Proceedings of a Meeting of the African Trypanotolerant Livestock Network, Nairobi. Nairobi: International Livestock Centre for Africa and International Laboratory for Research on Animal Diseases (ILRAD), pp. 360–376.
- JALVINGH, A.W., 1993. Dynamic livestock modelling for on-farm decision support. PhD Thesis, Wageningen Agricultural University, The Netherlands.
- JAMES, A.D. & ROSSITER, P.B., 1989. An epidemiological model of rinderpest. 1. Description of the model. Tropical Animal Health and Production, 21, 59–68.
- JAMISON, D.T., MOSLEY, W.H., MEASHAM, A.R. & BOBADILLA, J.L., 1993. Disease Control Priorities in Developing Countries. New York: Published for the World Bank by Oxford University Press, Oxford.
- JOHNSTON, J., 1990. Health and productivity of village poultry in Southeast Asia. Economic impact of developing techniques to vaccinate birds orally against Newcastle disease. ACIAR Working Paper No. 31. Canberra, Australia.
- KAMAU, S.W., OMUKUBA, J., KIRAGU, J., MASIKA, P., NDUNG’U, J.M., WACHIRA, P. & MEHLITZ, D., 2000. Financial analysis of animal trypanosomiosis control using cypermethrin pour-on in Kenya. Preventive Veterinary Medicine, 44, 231–246.
- KITALA, P.M., MCDERMOTT, J.J., KYULE, M.N. & GATHUMA, J.M., 2000. Community-based active surveillance for rabies in Machakos District, Kenya. Preventive Veterinary Medicine, 44, 73–85.
- KITALA, P., MCDERMOTT, J.J., KYULE, M.N., GATHUMA, J.M., PERRY, B.D. & WANDELER, A., 2001. Dog ecology and demography information to support the planning of rabies control in Machakos District, Kenya. ,em>Acta Tropica, 78, 217–230.
- KLEEMAN, G., 1999. Service Management in Livestock Development: Concepts and Elements. Wiesbaden: Deutsche Gesellschaft für Technische Zusammenarbeit (GTZ) GmbH.
- KOEMAN, J.H., BALK, F. & TAKKEN, W., 1980. The environmental impact of tsetse control operations. A report on present knowledge. FAO Animal Production Health Paper 7, FAO, Rome, pp. 71.
- KONANDREAS, P.A. & ANDERSON, F.M., 1982. Cattle Herd Dynamics: An Integer and Stochastic Model for Evaluating Production Alternatives. ILCA Research Report No. 2, Addis Ababa: International Livestock Centre for Africa.
- KRISTJANSON, P.M., SWALLOW, B.M., ROWLANDS, G.J., KRUSKA, R.L. & DE LEEUW, P.N., 1999. Measuring the costs of African animal trypanosomiasis, the potential benefits of control and returns to research. Agricultural Systems, 59, 79–98.
- KRUSKA, R.L., PERRY, B.D., REID, R.S., 1995. Recent progress in the development of decision support systems for improved animal health.In: Integrated Geographic Information Systems Useful for a Sustainable Management of Natural Resources in Africa. Proceeding of the Africa GIS 1995 meeting, Abidjan, Côte d’Ivoire, March 6-9. UNITAR/95/2. United Nations, Geneva.
- LATHAM, M., 1997. Human nutrition in the developing worlds. FAO Food and Nutrition Paper 29. Rome: FAO.
- LAWRENCE, J.A., 1992. History of bovine theileriosis in southern Africa. In: NORVAL, R.A.I., PERRY, B.D. & YOUNG, A.S.,The Epidemiology of Theileriosis in Africa. London: Academic Press. pp. 1–39.
- LEAK, C., 1998. Mosquito-borne arboviruses. In: PALMER, S.R., LORD SOULSBY & SIMPSON, D.I.H., (eds). Zoonoses: Biological, Clinical Practice and Public Health Control. Oxford: Oxford University Press. pp.401–415.
- LEE, J-A.L.M., MORE, S.J. & COTIWAN, B.S., 1999. Problems translating a questionnaire in a cross-cultural setting. Preventive Veterinary Medicine, 41, 187–194.
- LEECH, F.B. & SELLERS, K.C., 1979. Statistical Epidemiology in Veterinary Science. London: Charles Griffin and Company Ltd.
- LEONARD, D.K., 1987. The supply of veterinary services: Kenyan lessons. Journal of Agricultural Administration and Extension, 26, 219–236.
- LEONARD, D.K., 1993. Structural reform of the veterinary profession in Africa and the New Institutional Economics. Development and Change, 24, 227–267.
- LESSARD, P., L’EPLATTENIER, R.L., NORVAL, R.A.I., KUNDERT, K., DOLAN, T.T., CROZE, H, WALKER, J.B., IRVIN, A.D. & PERRY, B.D., 1990. Geographical information systems for studying the epidemiology of cattle diseases caused by Theileria parva. The Veterinary Record, 126, 255–262.
- LEVY, P.S. & LEMESHOW, S., 1991. Sampling of populations: methods and applications. New York: John Wiley.
- LIANG, K.-Y. & ZEGER, S.L., 1993. Regression analysis for correlated data. Annual Review of Public Health, 14, 43–68.
- LIVESTOCK IN DEVELOPMENT (LID), 1999. Livestock on Poverty-Focused Development. Livestock in Development: Crewkerne, Somerset, UK.
- MCCAULEY, E.H., AULAQI, N.A., NEW, J.C., SUNDQUIST, W.B. & MILLER, W.M., 1979. A Study of the Potential Impact of Foot and Mouth Disease in the United States. Washington D.C.: United States Government Printing Office.
- MCDERMOTT, J.J., 2000. Evidence required for establishing the absence of tsetse and trypanosomosis associated with tsetse eradication programmes. In: DWINGER, R., (ed.). Animal Trypanosomosis: Diagnosis and Epidemiology. Vienna: International Atomic Energy Agency. pp. 156–164.
- MCDERMOTT, J.J. & SCHUKKEN, Y.H., 1994. A review of methods to adjust for cluster effects in explanatory epidemiological studies of animal populations. ,Preventive Veterinary Medicine, 18, 155–173.
- MCDERMOTT. J.J., SCHUKEN, Y.H. & SHOUKRI, M.M., 1994. Study design and analytic methods for data collected from clusters of animals. Preventive Veterinary Medicine, 18, 175–191.
- MCDERMOTT, J., O’CALLAGHAN, C. & KADOHIRA, M., 1997. Experiences in applying multi-level models for assessing the distribution of animal diseases. Epidemiologie et Sante Animale, 31–32:13.23.1–3.
- MCDERMOTT, J.J., KADOHIRA, M., O’CALLAGHAN, C.J. & SHOUKRI, M.M., 1997. A comparison of different models for assessing variations in the sero-prevalence of infectious bovine rhinotracheitis by farm, area and district in Kenya. Preventive Veterinary Medicine, 32, 219–234.
- MCDERMOTT, J.J., RANDOLPH, T.F. & STAAL, S.J., 1999. The economics of optimal health and productivity in smallholder livestock systems in developing countries. In: PERRY, B.D., (ed.). The Economics of Animal Disease Control. O.I.E. Scientific and Technical Revue, 18, 399–419.
- MCDERMOTT, J.J., KRISTJANSON, P.M., KRUSKA. R.L., REID, R.S., ROBINSON, T.P., COLEMAN, P.G., JONES, P.G. & THORNTON, P.K., 2001. Effects of climate, human population and socio-economic changes on tsetse-transmitted trypanosomiasis to 2050. In: SEED, R. & BLACK, S., (eds). World Class Parasites. Vol. 1. The African Trypanosomes. Boston: Kluwer. pp. 25–38.
- MACHILA, N., EISLER, M.C., WANYANGU, S.W., MCDERMOTT, J.J., WELBURN, S.C. & MAUDLIN, I., 2000. Perceptions of cattle owners in the control of African bovine trypanosomiasis. Proceedings of the Ninth International Symposium on Veterinary Epidemiology and Economics, 7–11 August 2000. Breckenridge, Colorado.
- MCINERNEY, J.P., 1988. The economic analysis of livestock disease: the developing framework. In: WILLEBERG, P., AGGER, J.F. & RIEMANN, H.P., (eds). Proceedings of the 5th International Symposium on Veterinary Epidemiology and Economics, Acta Veterinaria Scandinavica, Supplementum, 84, 68–74.
- MCINERNEY, J., 1996. Old economics for new problems-livestock disease. Journal of Agricultural Economics, 47, 315–337.
- MCLEOD, A. & LESLIE, J., 2000. Socio-economic impacts of freedom from livestock disease and export promotion in developing countries. Livestock Information and Policy Discussion. Paper No. 3. Rome: FAO.
- MCLEOD, A. & WILSMORE, T., 2002. Delivery of animal health care to the poor: a review. In: PERRY B.D., RANDOLPH, T.F., MCDERMOTT, J.J., SONES, K.R. & THORNTON, P.K., Investing in Animal Health Research to Alleviate Poverty. Nairobi: International Livestock Research Institute (ILRI).
- MCNAB, W.B., 1998. A general framework illustrating an approach to quantitative microbial food safety risk assessment. Journal of Food Protection, 61, 1216–1228.
- MALOO, S.H., THORPE, W., KIOO, G., NGUMI, P., ROWLANDS, G.J. & PERRY, B.D., 2001. Seroprevalences of vector-transmitted infections of small-holder dairy cattle in coastal Kenya. Preventive Veterinary Medicine, 52, 1–16.
- MALOO, S.H., ROWLANDS, G.J., THORPE, W., GETTINBY, G. & PERRY, B.D., 2001. A longitudinal study of disease incidence and case fatality risks on smallholder farms in coastal Kenya. Preventive Veterinary Medicine, 52, 17–29.
- MALOO, S.H., NGUMI, P., MBOGO, S., WILLIAMSON, S., THORPE, W, ROWLANDS, G.J. & PERRY, B.D., 2001. Identification of a target populations for immunisation against East Coast fever in coastal Kenya. Preventive Veterinary Medicine, 52, 31–41.
- MARINER, J.C., AKABWAI, D.M.O., TOYANG, J., ZOYEM, N. & NGANGYOU A. 1994.The Kenya Veterinarian, 18, 507–509.
- MARSH, W.E., 1987. Practical computer models for assessing productivity in pig production systems. In: THRUSFIELD, M.V., (ed.). Proceedings of the Society for Veterinary Epidemiology and Preventive Medicine, University of Edinburgh, pp. 95–106.
- MARTIN, S.W., MEEK, A.H. & WILLEBERG, P., 1987. Veterinary Epidemiology: Principles and Methods. Ames: Iowa State University Press.
- MATTHEWMAN, R.W. & PERRY, B.D., 1985. Measuring the benefits of disease control: the relationship between herd structure and herd productivity. Tropical Animal Health and Production, 17, 39–51.
- MBOGOH, S.G., 1984. Dairy development and internal marketing in sub-Saharan Africa: Performance, policies and options. Livestock Policy Unit Working Paper No. 5. Addis Ababa, Ethiopia: International Livestock Centre for Africa (ILCA).
- MEDLEY, G.F., 1994. The transmission dynamics of Theileria parva. In: Modelling vector-borne and other parasitic diseases. International Laboratory for Research on Animal Diseases (ILRAD), Nairobi. pp. 105–117.
- MEDLEY, G.F., PERRY, B.D. & YOUNG, A.S., 1993. Preliminary analysis of the transmission dynamics of theileriosis in eastern Africa. Parasitology, 106, 251–264.
- MEINERT, C.L., 1986. Clinical trials-design, conduct and analysis. Oxford: Oxford University Press.
- MIGCHIELSEN, S. 2000. Elsevier publications, Amsterdam. Personal communication.
- MILLIGAN, P.J.M. & BAKER, R.D., 1988. A model of tsetse-transmitted animal trypanosomiasis. Parasitology, 96, 211–239.
- MINJAUW, B., KRUSKA, R., ODERO, A., RANDOLPH, T., MCDERMOTT, J.J., MAHAN, S. & PERRY, B.D., 2000. Economic impact assessment of Cowdria ruminantium infection and its control in southern Africa. Proceedings of the Ninth International Symposium on Veterinary Epidemiology and Economics, 7–11 August 2000. Breckenridge CO, USA.
- MINJAUW, B., ELSWORTH, D., WELLS, C., MWAURA, S., KRUSKA, R., ROBINSON, T., NIJBROEK, R., 2001. East Coast fever in sub-Saharan Africa. Poster. International Livestock Research Institute (ILRI). Nairobi, Kenya.
- MORRIS, R.S., 1999. The application of economics in animal health programmes: a practical guide. In: PERRY, B.D., (ed.). The Economics of Animal Disease Control. O.I.E. Scientific and Technical Revue, 18, 305–314.
- MORRIS, R.S. & MARSH, W.E., 1994. The relationship between infections, diseases and their economic effects. In: PERRY, B.D. & HANSEN, J.W., (eds). Modelling Vector Borne and other Parasitic Diseases. Proceedings of a workshop organized by ILRI in collaboration with FAO, 23–27 November 1992, Nairobi, Kenya. International Laboratory for Research in Animal Diseases (ILRAD), pp. 192–213.
- MORRIS, R.S., WILESMITH, J.W., STERN, M.W., SANSON, R.L. & STEVENSON, M.A., 2001.Predictive spatial modelling of alternative control strategies for the foot-and-mouth disease epidemic in Great Britain.The Veterinary Record, 149, 137–144.
- MUKHEBI, A.W., PERRY, B.D. & KRUSKA, R.L., 1992. Estimated economics of theileriosis control in Africa. Preventive Veterinary Medicine, 12, 73–85.
- MUKHEBI, A.W., CHAMBOKO, T., O’CALLAGHAN, C.J., PETER, T.F., KRUSKA, R.L., MEDLEY, G.F., MAHAN, S.M. & PERRY, B.D., 1999. An assessment of the economic impact of heartwater (Cowdria ruminantium infection) and its control in Zimbabwe. Preventive Veterinary Medicine, 39, 173–189.
- MULLINS, G., ALLSOPP, R., NKHORI, P., KOLANYANE, M. & PHILLEMON-MOTSU, T., 1997. The effect of tsetse fly and tsetse control on tourism in the Okavango Delta of Botswana. Publication no. 119. Proceedings of the 24th meeting of the International Scientific Council for Trypanosomiasis Research and Control (ISCTRC), Maputo. pp. 555–562.
- MURRAY, C.J.L & LOPEZ, A.D., 1996. Global Burden of Disease. Boston: Harvard University Press.
- MUSOKE, A., MORZARIA, S. & NENE, V., 1999. Subunit vaccines for the control of Theileria parva. In: MORZARIA, S. & WILLIAMSON, S., (eds). Live vaccines for Theileria parva: Deployment in Eastern, Central and Southern Africa. Proceedings of an FAO, OAU-IBAR and ILRI Workshop held at International Livestock Research Institute, 10–12 March 1997. Nairobi, Kenya. pp. 125–130.
- NAGEL, P., 1995. Environmental Monitoring Handbook for Tsetse Control Operations. Edited by the Scientific Environmental Monitoring Group (SEMG). Regional Tsetse and Trypanosomiasis Control Programme, Harare, Zimbabwe.
- NDUNG’U, L.W., RANDOLPH, T.F., COETZEE, G., KRECEK, R.C. & PERRY, B.D., 2000. An economic assessment of current delivery pathways for the control of tick-borne diseases in Kenya. In: SALMAN, M.D., MORLEY, P.S. & RUCH-GALLIE, R., (eds). Proceedings of the Ninth Symposium of the International Society for Veterinary Epidemiology and Economics, 6–11 August 2000, Breckenridge, Colorado. Compact disk.
- NGATEGIZE, P.K., BEKELE, T. & TILAHUN, G., 1993. Financial losses caused by ovine fascioliasis in the Ethiopian highlands. Tropical Animal Health and Production, 25, 155–161.
- NORDHUIZEN, J.P.T.M., FRANKENA, K., PLOEGER, H. & NELL, T., 1993. Field trial and error. Proceeding of the International Seminar with Workshops on the Design, Conduct and Interpretation of Field Trials, Berg en Dal, The Netherlands, 27–28 April 1993. Wageningen: Epidecon.
- NOTERMANS, S., GALLHOF, G., ZWIETERING, M.H. & MEAD, G.C., 1995. The HACCP concept: specification of criteria using quantitative risk assessment. Food Microbiology, 12, 81–90.
- NYANGITO, H.O., RICHARDSON, J.W., MUKHEBI, A.W., MUNDY, D.S., ZIMMEL, P., NAMKEN, J. & PERRY, B.D., 1994. Whole farm economic analysis of East Coast fever immunization strategies in Kilifi District, Kenya. Preventive Veterinary Medicine, 21, 215–235.
- NYANGITO, H.O., RICHARDSON, J.W., MUKHEBI, A.W., ZIMMEL, P., NAMKEN, J. & PERRY, B.D., 1996. Whole farm simulation analysis of economic impacts of East Coast fever immunization strategies on mixed crop-livestock farms in Kenya. Agricultural Systems, 51, 1–27.
- O’CALLAGHAN, C.J., MEDLEY, G.F., PETER, T.F. & PERRY, B.D., 1998. Analysis of the epidemiology of heartwater (Cowdria ruminantium infection) in a transmission dynamics model. Parasitology, 117, 49–61.
- O’CALLAGHAN, C.J., MEDLEY, G.F., PETER, T.F., MAHAN, S.M. & PERRY, B.D., 1999. The effect of vaccination on the transmission dynamics of heartwater (Cowdria ruminantium infection). Preventive Veterinary Medicine, 42, 17–38.
- OGUNDIPE, G.A.T., OLUOKUN S.B. & ESURUOSO, G.O., 1989. The development and efficiency of the animal health information system in Nigeria. Preventive Veterinary Medicine, 7, 121–135.
- OKELLO-ONEN, J., MUKHEBI, A.W., TUKAHIRWA, E.M., MUSISI, G., BODE, E., HEINONEN, R., PERRY, B.D. & OPUDA-ASIBO, J., 1998. Financial analysis of dipping strategies for indigenous cattle under ranch conditions in Uganda. Preventive Veterinary Medicine, 33, 241–250.
- OLLERENSHAW, C.B., 1966. The approach to forecasting the incidence of fascioliasis over England and Wales, 1958–1962. Agricultural Meteorology, 3, 35–54.
- OMORE, A.O., MCDERMOTT, J.J., STAAL, S.,ARIMI, S.M., KANG’ETHE, E.K. & OUMA, E.A., 2000. Analysis of public health risks from consumption of informally marketed milk in sub-Saharan African countries. Proceedings of the Ninth International Symposium on Veterinary Epidemiology and Economics, Breckenridge, Colorado, 7–11 August 2000.
- ORMEROD, W.E., 1990. Africa with and without tsetse. Insect Science and Its Application, 11, 455–461.
- PAALMAN, M., BEKEDAM, H., HAWKEN, L. & NYHEIM, D., 1998. A critical review of priority setting in the health sector: the methodology of the 1993 World Development Report. Health Policy and Planning, 13, 13–31.
- PATON, G. & GETTINBY, G., 1983. The control of a parasitic nematode population in sheep represented by a discrete time network with stochastic inputs. Proceedings of the Irish Academy, 83B, 267–280.
- PEGRAM, R.G., JAMES, A.D., OOSTERWIJK, G.P.M., KILLORN, K.J., LEMCHE, J., GHIROTTI, M., TEKLE, Z., CHIZYUKA, H.G.B., MWASE, E.T. & CHIZHUKA, F., 1991. Studies on the economics of ticks in Zambia. Experimental and Applied Acarology, 12, 9–26.
- PENNE, K. & D’HAESE, L.D., 1999. Theileriosis control modelling (experiences from Southern Province, Zambia). Tropical Medicine and International Health, 4, A58–A70.
- PERMIN, A. & MADSEN, M., 2002. Literature review of disease control delivery, adoption and impact (small holder poultry)., In: PERRY, B.D., RANDOLPH, T.F., MCDERMOTT, J.J., SONES, K.R. & THORNTON, P.K., Investing in Animal Health Research to alleviate Poverty. Nairobi, Kenya: International Livestock Research Institute (ILRI).
- PERRY, B.D., 1988. The design and use of supportive epidemiological studies. In: LESSARD, P. & PERRY, B.D., (eds). The Investigation of Disease Outbreaks and Impaired Productivity. Veterinary Clinics of North America. Philadelphia: W.B. Saunders. pp. 97–108.
- PERRY, B.D. (ed.), 1999. The Economics of Animal Disease Control. OIE Scientific and Technical Revue, Special Edition, 18, (2).
- PERRY, B.D. & young, a.s., 1995. The past and future roles of epidemiology and economics in the control of tick-borne diseases of livestock in Africa: the case of theileriosis. Preventive Veterinary Medicine, 25, 107–120.
- PERRY, B.D. & RANDOLPH, T.F., 1999. Improving the assessment of the economic impact of parasitic diseases in production animals. Veterinary Parasitology, 84, 143–166.
- PERRY, B.D., MWANAUMO, B., SCHELS, H.F., EICHER, E. & ZAMAN, M.R., 1984. A study of health and productivity of traditionally managed cattle in Zambia. Preventive Veterinary Medicine, 2, 633–653.
- PERRY, B.D., PALMER, J.E., TROUTT, H.F., BIRCH, J.B., MORRIS, D., EHRICH, M. & RIKIHISA, Y., 1986. A case-control study of Potomac horse fever. Preventive Veterinary Medicine, 4, 69–82.
- PERRY, B.D., LESSARD, P., NORVAL, R.A.I., KUNDERT, K. & KRUSKA, R., 1990. Climate, vegetation and the distribution of Rhipicephalus appendiculatus in Africa. Parasitology Today, 6, 100–104.
- PERRY, B.D., KRUSKA, R., LESSARD, P., NORVAL, R.A.I. & KUNDERT, K., 1991. Estimating the distribution and abundance of Rhipicephalus appendiculatus in Africa. Preventive Veterinary Medicine, 11, 261–268.
- PERRY, B.P., KALPRAVIDH, W., COLEMAN, P.G., HORST, H.S., MCDERMOTT, J.J., RANDOLPH, T.F. & GLEESON, L.J., 1999. The economic impact of foot-and-mouth disease and its control in southeast Asia: a preliminary assessment with special reference to Thailand. In: PERRY, B.D., (ed.). The Economics of Animal Disease Control. OIE Scientific and Technical Review. 18, 478–495.
- PERRY, B., MCDERMOTT, J. & RANDOLPH, T., 2001. Can epidemiology and economics make a meaningful contribution to national animal-disease control? Preventive Veterinary Medicine, 48, 231–260.
- PERRY, B.D., RANDOLPH, T.F., MCDERMOTT, J.J., SONES, K.R. & THORNTON, P.K., 2002. Investing in animal health research to alleviate poverty. International Livestock Research Institute (ILRI), Nairobi, Kenya.
- PETER, T.F., PERRY, B.D., O’CALLAGHAN, C.J., MEDLEY, G.F., SHUMBA, W., MADZIMA, W., BURRIDGE, M.J. & MAHAN, S.M., 1998.The distribution of heartwater in the highveld of Zimbabwe, 1980–1977. Onderstepoort Journal of Veterinary Research, 65, 177–187.
- PETER, T.F., PERRY, B.D., O’ CALLAGHAN, C.J., MEDLEY, G.F., SHUMBA, W., MADZIMA, W., BURRIDGE, M.J. & MAHAN, S.M., 1998.Distribution of the vectors of heartwater, Amblyomma hebraeum and Amblyomma variegatum (Acari; Ixodidae) in Zimbabwe. Experimental & Applied Acarology , 22, 1–16.
- PETER, T.F., ANDERSON, E.C., BURRIDGE, M.J., PERRY, B.D. & MAHAN, S.M., 1999. Susceptibility and carrier status of impala, sable and tsessebe for Cowdria ruminantium infection (heartwater). Journal of Parasitology , 85, 31–35.
- PETER, T.F., BRYSON, N.J., PERRY, B.D., O’CALLAGHAN, C.J., MEDLEY, G.F., MLAMBO, G., HORAK, I.G., BURRIDGE, M.J. & MAHAN, S.M., 1999. Cowdria ruminantium infection in an African Nature reserve. The Veterinary Record. 145, 304–307.
- PETER, T.F., PERRY, B.D., O’CALLAGHAN, C.J., MEDLEY, G.F., MLAMBO, G., BARBET, A.F. & MAHAN, S.M., 1999. Prevalence of Cowdria ruminantium infection in Amblyomma hebraeum ticks from heartwater endemic areas of Zimbabwe. Epidemiology and Infection, 123, 309–316.
- PFEIFFER, D.U., STERN, M.W. & MORRIS, R.S., 1994. POSSPOP: A geographical simulation model of bovine tuberculosis infection in a wildlife population. The Kenyan Veterinarian, 18, 313–315.
- POATE, C.D. & DAPLYN, P.F., 1993. Data for Agrarian Development. Cambridge: Cambridge University Press.
- PROVIC, P., 1998. Zoonotic hookworm infections (ancylostomosis). In: PALMER, S.R., LORD SOULSBY & SIMPSON, D.I.H., (eds). Zoonoses: Biological, Clinical Practice and Public Health Control. Oxford: Oxford University Press. pp.803–822.
- POND, B.A., BROEKHOVEN, L.H., TINLINE, R.R. & VOIGT, D.R., 1984. Users Guide to the Ontario Rabies Model. Ontario: Queens University.
- PRITCHARD, D.G., EDWARDS, S. & MORZARIA, S.P., 1983. Case-control studies on the evaluation of serological data from respiratory disease outbreaks in cattle. Proceedings of the Society for Veterinary Epidemiology and Preventive Medicine, Edinburgh, pp. 131–138.
- PUTT, S.N.H., SHAW, A.P.M., WOODS, A.J., TYLER, L. & JAMES, A.D., 1987. Veterinary Epidemiology and Economics in Africa. Addis Ababa: International Livestock Centre for Africa.
- RANDOLPH, T. & NDUNG’U, L., 2000. Gender and transaction cost: a conjoint analysis of choice of livestock health service among smallholder dairy farmers in Kenya. Background paper for a poster presented at the Twenty-fourth Congress of the International Association of Agricultural Economists, Berlin, 18–24 August 2000. Nairobi: International Livestock Research Institute.
- RANDOLPH, T.F., KRISTJANSON, P.M., OMAMO, W.S., ODERO, A.N., THORNTON, P.K., REID, R.S., ROBINSON, T. & RYAN, J.G., 2001. A framework for priority setting in international livestock research. Research Evaluation, 10, 142–160.
- REID, R.S., 1997. Impacts of controlling trypanosomosis on land-use and the environment: state of our knowledge and future directions. Proceedings of the Twenty-fourth Meeting of the International Scientific Council for Trypanosomiasis Research and Control, Maputo, Mozambique, 29 September – 3 October 1997.
- REID, R.S., KRUSKA, R.L., MUTHUI, N., TAYE, A., WOTTON, S., WILSON, C. & MULATU, W., 2000. Land use and land cover dynamics in response to changes in climatic, biological and sociopolitical forces: the case of southwestern Ethiopia. Landscape Ecology, 15, 330–355.
- REITHINGER, R. & DAVIES, C.R., 1999. Is the domestic dog (Canis familiaris) a reservoir host of American cutaneous leishmaniasis? A critical review of current evidence. American Journal of Tropical Medicine and Hygiene, 61, 530–541.
- ROBINSON, T., 2000. Spatial statistics and geographical information systems in epidemiology and public health. In: hay, s.i., randolph, s.e. & rogers, d.j., (eds). Remote Sensing and Geographical Information Systems in Epidemiology. Advances in Parasitology, Vol. 47. London: Academic Press. pp. 81–128.
- ROGERS, D.J., 1988. A general model for the African trypanosomiases. Parasitology, 97, 193–212.
- ROGERS, D.J., 2000. Satellites, space, time and African trypanosomes. In: HAY, S.I., RANDOLPH, S.E. & ROGERS, D.J., (eds). Remote Sensing and Geographical Information Systems in Epidemiology. Advances in Parasitology. Vol. 47. London: Academic Press. pp. 129–171.
- ROLA, A.C. & PINGALI, P.L., 1993. Pesticides, rice productivity, and farmers’ health: an economic assessment. Manila: International Rice Research Institute.
- ROSSITER, P.B. & JAMES, A.D., 1989. An epidemiological model of rinderpest. 2. Simulations of the behaviour of rinderpest virus in populations. Tropical Animal Health and Production, 21, 69–84.
- ROTH, F. & ZINSSTAG, J., 2001. Economic analysis of brucellosis control in Mongolia-improvement of human health through the veterinary sector —the case of animal brucellosis control. Basle: Swiss Tropical Institute.
- ROTHMAN, K.J. & GREENLAND, S., 1998. Modern Epidemiology. 2nd edn. Philadelphia: Lippincott-Raven.
- SALEQUE, A., 2000. Scaling-up: critical factors in leadership, management, human resource development and institution building in going from pilot project to large scale implementation: the BRAC poultry model in Bangladesh. In: DOLBERG, F. & PETERSEN, P.H., (eds). Poultry as a Tool in Poverty Eradication and Promotion of Gender Equality. Proceedings of a Workshop held on 22–26 March 1999, at Tune, Denmark. Frederiksberg: DSR Publishers. pp. 51-71.
- SANDERS, J.O. & CARTWRIGHT, T.C., 1979. A general cattle production systems model. I. Agricultural Systems, 4, 217–227.
- SANDERS, J.O. & CARTWRIGHT, T.C., 1979. A general cattle production systems model II. Proceedings used for simulating animal performance. Agricultural Systems, 4, 289–309.
- SANSON, R.L. & THORNTON, R.N., 1997. A modelling approach to the quantification of the benefits of a national surveillance programme. Preventive Veterinary Medicine, 30, 37–47.
- SCHEPERS, J.A. & DIJKHUIZEN, A.A., 1991. The economics of mastitis and mastitis control in dairy cattle: a critical analysis of estimates published since 1970. Preventive Veterinary Medicine, 10, 213–224.
- SCHILLHORN VAN VEEN, T.W. & DE HAAN, C., 1995. Trends in the organization and financing of livestock and animal health services. Preventive Veterinary Medicine, 25, 225–240.
- SCHWABE, C.W., 1984. Veterinary Medicine and Human Health. 3rd edn. Baltimore USA: Williams and Wilkins.
- SCHWABE, C.W., RIEMANN, H.P. & FRANTI, C.E., 1977. Epidemiology in Veterinary Practice. Philadelphia: Lea and Febiger.
- SCIENTIFIC ENVIRONMENTAL MONITORING GROUP, 1993. Impact of aerial spraying and odour-baited targets on ecosystems. Report 1987–1990. Monitoring of tsetse control operations in Zambia and Zimbabwe. EC Delegation, Harare, Zimbabwe.
- SERE, C. & STEINFELD, H., 1996. World livestock production systems: current status, issues and trends. FAO Animal Production and Health Paper No. 127. Rome: Food and Agriculture Organisation of the United Nations.
- SHAW, A.P.M. & HOSTE, C.H., 1987. Trypanotolerant cattle and livestock development in West and Central Africa. Vol. 1. International Supply and Demand for Breeding Stock. FAO Animal Health and Production Paper 67/1. Rome: FAO.
- SINGH, I., SQUIRE, L. & STRAUSS, J., 1986. Agricultural Household Models. Baltimore: Johns Hopkins University Press.
- SKIDMORE, S.J., 1999. Factors in the spread of hepatitis E. Lancet, 354, 1049–1050.
- SMITH, R.D., 1991. Veterinary Clinical Epidemiology: A Problem-Oriented Approach. Stoneham: Butterworth-Heinemann.
- SONAIYA, E.B., BRANCKAERT, R.D.S. & GUEYE, E.F., 1999. Research and Development Options for Family Poultry. Introductory paper—First INFPD/FAO Electronic Conference on Family Poultry, GUEYE, E.F., (ed.). Rome: FAO.
- SPRADBROW, P.B., 1993. Newcastle disease in village chickens. In: Poultry Science Review 5. pp. 57–96.
- SPRADBROW, P.B. & COPLAND, J.W., 1996. Production of thermostable Newcastle disease virus in developing countries. Preventive Veterinary Medicine, 29, 157–159.
- STEM, C., 1993. An economic analysis of the prevention of peste des petits ruminants in Nigerian goats. Preventive Veterinary Medicine, 16, 141–150.
- SWALLOW, B.M., 2000. Impacts of Trypanosomiasis on African Agriculture. PAAT Technical and Scientific Series No. 2. Rome: FAO.
- SWALLOW, B.M., MULATU, W. & LEAK, S.G.A., 1995. Potential demand for a mixed public-private animal health input: evaluation of a pour-on insecticide for controlling tsetse-tranmitted trypanosomiasis in Ethiopia. Preventive Veterinary Medicine, 24, 265–275.
- TAMBI, E.N., MAINA, O.W., MUKHEBI, A.W. & RANDOLPH, T.F., 1999. Economic impact assessment of rinderpest control in Africa. In: PERRY, B.D., (ed.). The Economics of Animal Disease Control. OIE Scientific and Technical Review, 18, 458–477.
- TAYLOR, M.G., 1998. Schistosomosis. In: PALMER, S.R., LORD SOULSBY & SIMPSON, D.I.H., (eds). Zoonoses: Biological, Clinical Practice and Public Health Control. Oxford: Oxford University Press. pp.717–730.
- TAYLOR, L.H., LATHAM, S.M. & WOOLHOUSE, M.E.J., 2001. Risk factors for human disease emergence. Philosophical Transaction of the Royal Society: (Series B: Biological Sciences), 356, 983–989.
- THORNTON, P.K., KRUSKA, R.L., HENNINGER, N., KRISTJANSON, P.M., REID, R.S., ATIENO, F., ODERO, A. & NDEGWA, T., 2002. Mapping poverty and livestock in the developing world. International Livestock Research Institute (ILRI), Nairobi, Kenya.
- THORNTON, P.K., RANDOLPH, T.F., KRISTJANSON, P.M., OMAMO, W.S., ODERO, A.N. & RYAN, J.G., 2000. Priority assessment for the International Livestock Research Institute 2000 to 2010. ILRI Impact Assessment Series No. 6, Nairobi.
- THRUSFIELD, M., 1995. Veterinary Epidemiology. 2nd edn. Oxford: Blackwell Science.
- TOMA, B., BÉNET, J.J., DUFOUR, B., ELOIT, M., MOUTOU, F. & SANAA, M., 1991. Glossaire d’epidemiologie animale. Maisons-Alfort: Editions du Point Vétérinaire.
- TREWHELLA, W.J. & ANDERSON, R.M., 1983. Modelling bovine tuberculosis in badgers. In: THRUSFIELD, M.V., (ed.). Proceedings of the Society for Veterinary Epidemiology and Preventive Medicine. Edinburgh: University of Edinburgh. pp. 78–84.
- UGALDE, A. & JACKSON, J.T., 1995. The World Bank and international health policy: a critical review. Journal of International Development, 7, 525–541.
- UMALI, D.L., FEDER, G. & DE HAAN, C., 1992. The balance between public and private sector activities in the delivery of livestock services. World Bank Discussion Paper 163. Washington D.C.
- UNDP, 1997. HUMAN DEVELOPMENT REPORT, 1997. Oxford: Oxford University Press.
- VOIGT, D.R., TINLINE, R.R. & BROEKHOVEN, L.H., 1985. A spatial simulation model for rabies control. In: BACON, P.J., (ed.). Population Dynamics of Rabies in Wildlife. London: Academic Press. pp. 311–349.
- VON KAUFMANN, R., MCINTIRE, J. & ITTY, P., 1990. ILCA Bio-Economic Herd Model for the Microcomputer. Addis Ababa: International Livestock Centre for Africa.
- VOSE, D., 1996. Quantitative Risk Analysis: A Guide to Monte Carlo Simulation Modeling. Chichester, UK: John Wiley and Sons.
- WACHER, T.J., MILLIGAN, P.M., RAWLINGS, P. & SNOW, W.F., 1994. Spatial factors in the assessment of trypanosomosis challenge. In: PERRY, B.D. & HANSEN, J.E., (eds). Modelling Vector-borne and Parasitic Diseases. Nairobi: ILRAD and FAO. pp. 119–130.
- WALTNER-TOEWS, D. & MCEWEN, S.A., 1994. Chemical residues in foods of animal origin: overview and risk assessment. Preventive Veterinary Medicine, 20, 161–178.
- WANYANGU, S.W., BAIN, R.K., RUGUTT, M.K., NGINYI, J.M. & MUGAMBI, J.M., 1996. Anthelmintic resistance amongst sheep and goats in Kenya. Preventive Veterinary Medicine, 25, 285–290.
- WHO, 1996. Investing in Health Research and Development: Report of the Ad Hoc Committee on Health Research Relating to Future Intervention Options. Geneva: TDR/Gen/96.1.
- WHO, 2000. World Health Report 2000. Geneva: World Health Organization.
- WILLEBERG, P., 1975. A case control study of some fundamental determinants in the epidemiology of the feline urology syndrome. Nordisk Veterinaermedecin, 27, 15–19.
- WILSON, C.J., REID, R.S., STANTON, N.L. & PERRY, B.D., 1997. Ecological consequences of controlling the tsetse fly in southwestern Ethiopia: effects of land-use on bird species diversity. Conservation Biology, 11, 435–447.
- WINT, W., SLINGENBERGH, J. & ROGERS, D., 2000. Livestock Distribution, Production and Diseases: towards a Global Livestock Atlas. Consultant’s report. Food and Agriculture Organization of the United Nations (FAO), Rome.
- WOOLHOUSE, M.E., HAYDON, D.T., PEARSON, A. & KITCHING, R.P., 1996. Failure of vaccination to prevent foot-and-mouth disease. Epidemiology and Infection, 116, 363–371.
- WOOLHOUSE, M.E. & DONALDSON, A., 2000. The science of controlling disease outbreaks. Nature, 410, 515–516.
- YATES f., 1981. Sampling Methods for Censuses and Surveys. 4th edn. London: Charles Griffin.
- ZEPEDA, C., SALMAN, M. & RUPPANER, R., 2001. International trade, animal health and veterinary epidemiology: challenges and opportunities. Preventive Veterinary Medicine, 48, 261–271.
- ZIMBABWE DEPARTMENT OF VETERINARY SERVICES, 2001. Foot-and-Mouth Disease Outbreak: August 2001. Map downloaded from http://www.africaonline.co.zw/vet/fmd.htm on 19 November 2001.