- Infectious Diseases of Livestock
- Part 1
- Q fever
- Vectors: Ticks
- Vectors: Tsetse flies
- Vectors: Muscidae
- Vectors: Tabanidae
- Vectors: Culicoides spp.
- Vectors: Mosquitoes
- Classification, epidemiology and control of arthropod-borne viruses
- Special factors affecting the control of livestock diseases in sub-Saharan Africa
- The control of infectious diseases of livestock: Making appropriate decisions in different epidemiological and socioeconomic conditions
- Infectious diseases of animals in sub-Saharan Africa: The wildlife⁄livestock interface
- Vaccination: An approach to the control of infectious diseases
- African animal trypanosomoses
- Dourine
- Trichomonosis
- Amoebic infections
- GENERAL INTRODUCTION: COCCIDIA
- Coccidiosis
- Cryptosporidiosis
- Toxoplasmosis
- Besnoitiosis
- Sarcocystosis
- Balantidiosis
- Leishmaniosis
- Neosporosis
- Equine protozoal myeloencephalitis
- GENERAL INTRODUCTION: BABESIOSES
- Bovine babesiosis
- Equine piroplasmosis
- Porcine babesiosis
- Ovine babesiosis
- GENERAL INTRODUCTION: THEILERIOSES OF CATTLE
- East Coast fever
- Corridor disease
- Zimbabwe theileriosis
- Turning sickness
- Theileria taurotragi infection
- Theileria mutans infection
- Theileria annulata theileriosis
- Theileriosis of sheep and goats
- Theileria buffeli⁄orientalis infection
- Non-pathogenic Theileria species in cattle
- GENERAL INTRODUCTION: RICKETTSIAL, CHLAMYDIAL AND HAEMOTROPIC MYCOPLASMAL DISEASES
- Heartwater
- Lesser known rickettsial infections in animals and humans
- Chlamydiosis
- Q fever
- Eperythrozoonosis
- Bovine Haemobartonellosis
- Potomac horse fever
- GENERAL INTRODUCTION: ANAPLASMOSES
- Bovine anaplasmosis
- Ovine and caprine anaplasmosis
Q fever
This content is distributed under the following licence: Attribution-NonCommercial CC BY-NC  View Creative Commons Licence details here
View Creative Commons Licence details here
Introduction
Q fever is a zoonosis caused by Coxiella burnetii, a bacterium that is found worldwide in domestic ruminants which are the main reservoirs for human infections. While most infections are asymptomatic, abortions occur occasionally in domestic ruminants, and humans may develop severe life threatening forms of the disease.
In 1937, ‘Q fever’ (for query fever) was proposed by Edward Holbrook Derrick to describe a febrile illness in abattoir workers in Brisbane, Australia.24 Macfarlane Burnet and Mavis Freeman reproduced the disease in guinea pigs, mice and monkeys and observed numerous intracellular small rods in smears from spleens of infected mice.20 Independently, Gordon Davis at the Rocky Mountain Laboratory in the USA found that ticks collected in Nine Mile, Montana, produced a febrile illness in guinea pigs on which they were allowed to feed. Davis and Herald Rea Cox found the aetiological agent was filterable and displayed properties of both viruses and rickettsiae and, in 1938, Cox succeeded in propagating the infectious agent in embryonated eggs.22 Shortly thereafter, Rolla Eugene Dyer, Director of the National Institutes of Health, became infected with the organism while working in the Rocky Mountain Laboratory and a febrile illness was reproduced in guinea pigs inoculated with Dyer’s blood. Cross-immunity was demonstrated between microorganisms isolated from Dyer’s blood, the Nine Mile agent and the Australian Q fever agent obtained from Burnet.26 Although the agent of Q fever was initially called Rickettsia burnetii, a new genus Coxiella was proposed in 1938 and the agent was named Coxiella burnetii in recognition of the work of Cox and Burnet.45
Q fever was detected in Europe during the Second World War and has subsequently been diagnosed in 51 countries on five continents.41, 58 The first serological evidence for Q fever in southern Africa30 was soon followed by a description of a clinical case in a recently immigrated stockman who contracted the disease on the Witwatersrand in South Africa.69 A subsequent serological survey carried out by the South African Institute for Medical Research, and published in their 1953 Annual Report,4 claimed that Q fever was the commonest rickettsial infection in people in South Africa.
Aetiology
Coxiella burnetii is a Gram-negative, obligate, intracellular, bipolar rod measuring 0,2 × 1,0 μm. It is currently classified in the genus Coxiella, tribe Rickettsiae, family Rickettsiaceae, order Rickettsiales45 but recent phylogenetic studies, based mainly on 16S rRNA gene analysis, have shown the Coxiella genus belongs in the gamma subdivision of the Proteobacteria79, 91 along with the closely related genera Legionella, Francisella and Rickettsiella.
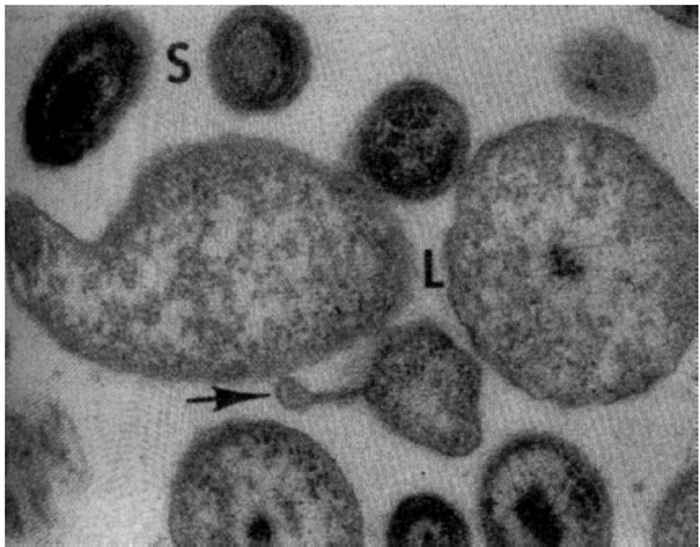
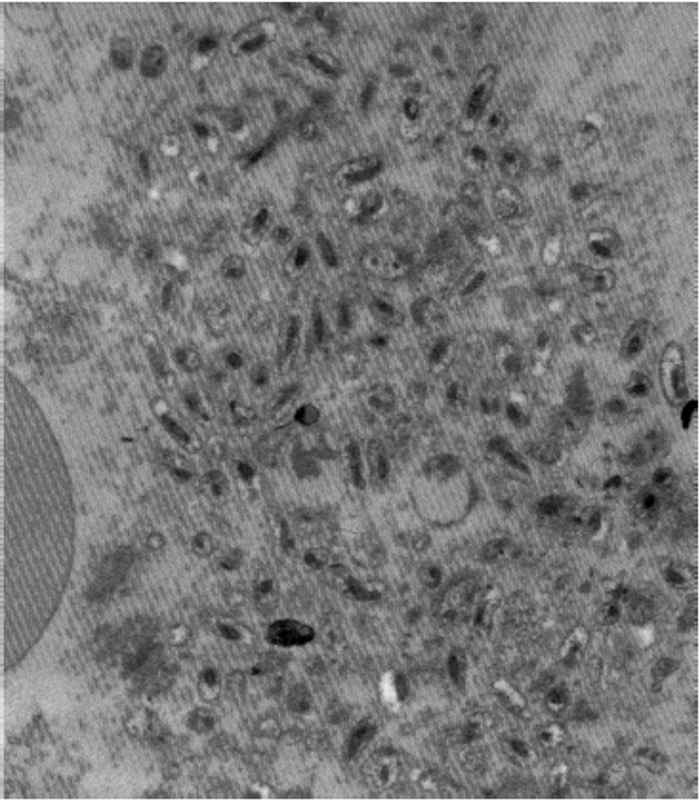
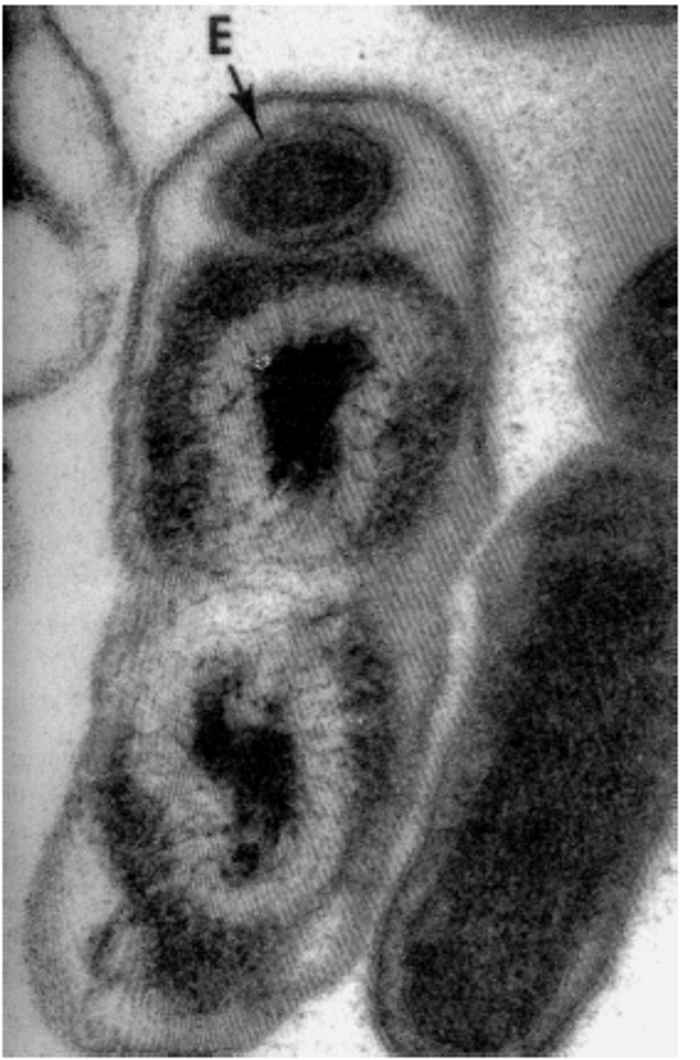
During infections, C. burnetii bind to receptors on monocytes/macrophages and are internalized in phagosomes which rapidly fuse with lysosomes to form phagolysosomes that appear as large vacuoles in infected cells.21 Coxiella burnetii is an acidophilic bacterium and its metabolism is enhanced by the acidic pH in the phagolysosome. 97 There are two distinct morphological forms of C. burnetii.53 The ‘small cell variant’ (SCV) is a compact, small rod with a very electron-dense centre of condensed nucleoid filaments while the ‘large cell variant’ (LCV) is a large, less dense, pleomorphic organism with a clear, periplasmic space between the outer and cytoplasmic membranes (Figure 43.1). A dense endospore-like body 130 to 170 nm in diameter might be found at one pole of some LCV (Figure 43.2). Both forms multiply by transverse binary fission. The SCVs are metabolically inactive and are the extracellular form of the organism. Once taken into the phagolysosome they are activated by the acidic environment and form LCVs, the metabolically active intracellular form of the bacterium. The cytoplasm of the host cell becomes filled with vacuoles loaded with organisms (Figure 43.3). ‘Large cell variants’ undergo sporogenic differentiation and form resistant, spore-like forms of the bacteria. These develop to SCVs that are released when the cells lyse.
Coxiella burnetii undergoes phase variation,81 mainly due to variations in the lipopolysaccharide of the organism. Phase I is the natural, highly infectious form of the organism which is found in infected animals, arthropods and humans.
Phase II organisms are obtained following serial passage of Phase I organisms in laboratory animals or cell culture and are minimally infective. Phase II organisms revert rapidly to Phase I organisms during a single passage in a vertebrate host. Monoclonal antibodies can be used to distinguish the phase variants.99
The genome of strains of C. burnetii varies from 1,5 to 2,4 Mb and isolates can be placed in genomic groups based on analysis by restriction fragment length polymorphism (RFLP) and pulsed-field gel electrophoresis.62, 96, 97 Eleven chromosomal genes have been cloned and expressed in Escherichia coli 58 and plasmids of 36 to 42 kb have been identified in the genome.97 Recent studies have indicated that genomic strain variation may be less important than host factors in explaining the occurrence of acute and chronic Q fever in humans.47
Epidemiology
Coxiella burnetii has been found worldwide, with the notable exception of New Zealand.,39 It occurs in a large variety of ticks, rodents, birds, and wild and domestic mammals and anti-C. burnetii antibodies have been found in snakes and tortoises in India.6 Although pigs, horses, camels, Asian buffaloes (Bubalus bubalis), dogs, cats, chickens, ducks, geese and turkeys may be infected, domestic ruminants are the major reservoirs for C. burnetii infections in humans.6
In southern Africa, serosurveys in cattle have shown that C. burnetii is endemic. Antibodies have been reported in cattle in Malawi (2 per cent),77 South Africa (8 per cent),35 Zambia (1 to 12 per cent)31, 63 and Zimbabwe (33 to 75 per cent).42, 68 Seropositive goats (10 per cent) and dogs (15 per cent) were also found in Zimbabwe and seropositive dogs have also been reported in Senegal (12 per cent) and Ivory Coast (8 per cent).16
Prevalences of antibodies to C. burnetii in humans have been reported from the Central African Republic (16 per cent),10 Egypt (16 to 25 per cent),17 Ethiopia (7 per cent),1 Sudan (10 per cent),17 Somalia (37 per cent),17 Tanzania (5 per cent),5 Tunisia (26 per cent)48 and Zimbabwe (37 per cent).42
Although cattle, sheep and goats may remain chronically infected for years, the majority of infections are subclinical. 46 Infected animals, however, may shed large numbers of organisms in the amniotic fluid, placenta (>109/g), foetal membranes, faeces and urine.82 A major risk factor for humans developing Q fever is contact with infected domestic livestock. Those at particularly high risk are farmers, abattoir workers, hide and fleece processors, and animal health workers. Infections of humans and domestic animals occur principally by inhalation of infected aerosols of the periparturient fluids with only a single organism being sufficient to cause infection.86 The extracellular C. burnetii are extremely resistant to desiccation, low or high pH, disinfectants and ultraviolet radiation.74 The organisms may remain infective in aerosols for up to two weeks and in the soil for as long as five months.49 Recent studies have shown that amoebae may be infected with C. burnetii and this could further promote their survival in the environment.58 Humans coming into contact with an infected farm environment are also at high risk of developing Q fever 85 outbreaks have occurred with exposure to manure.69 In some outbreaks, however, there is no direct contact with infected farms or animals and their products. In these outbreaks the source has been ascribed to windborne spread of C. burnetii from infected areas in the vicinity.37
Over 40 tick species have been found to be naturally infected with C. burnetii,6, 58 and transmission of the organism may occur during feeding of ticks. Although tick bites may play a role in the transmission of C. burnetii in wild rodents, lagomorphs and birds6, 46 there is little to indicate they are important in the maintenance of infections in livestock or humans. The organism does, however, multiply in the gut cells of ticks and large numbers of C. burnetii are shed in tick faeces. Contaminated hides and wool may be a source of infection for humans80 either by direct contact or after the faeces have dried and been inhaled as airborne dust particles.7
Large numbers of organisms are shed in the milk of dairy cows, particularly at the beginning of lactation. Up to 23 per cent of dairy cows may be shedding organisms in their milk14 and 21 per cent of bulk milk tanks in England and Wales have been reported to have serological evidence of infection. 64 Suspended in distilled water or sterile milk, the organism survives at room temperature for months and sometimes years.93 Although people may develop Q fever by drinking infected raw milk40 or eating infected milk products,92 in most cases ingestion of infected milk results in seroconversion without disease.11 Current recommendations for the pasteurization of milk (62,8 °C for 30 minutes or 71,7 °C for 15 seconds) are based on temperatures required to inactivate C. burnetii.27
In North America, outbreaks of Q fever have resulted from direct and indirect contact with parturient cats.,54 Seropositive cats have been found in Zimbabwe (13 per cent) and South Africa (2 per cent) but they have yet to be implicated in human infections.57
Outbreaks have also been reported in humans following exposure to infected pigeon faeces.78 Person-to-person transmission of C. burnetii is rare but occurs transplacentally and during blood transfusions and autopsies.58 A recent report describes sexual transmission in humans.60
Pathogenesis
The target cells of C. burnetii are monocytes/macrophages. Most infections occur via the respiratory route and involve the alveolar macrophages.85 The Kupffer cells of the liver are the primary cells infected in infections that take place via the digestive tract. Subsequently there is a bacteraemia which, in overt clinical cases in humans, coincides with fever.7 Calves in endemic areas may become infected in the first years of life and either recover completely or become latently infected. Hormonal changes in the final stages of pregnancy in domestic ruminants are thought to trigger massive multiplication of C. burnetii in the uterus, foetal tissues and udder. In the pregnant uterus, a mild to severe placentitis may follow the massive multiplication of organisms.56, 65 About 50 per cent of seropositive heifers shed C. burnetii in the placenta, birth fluids and milk at calving, but the percentage drops as they get older. About 50 per cent of chronically infected cows continue to shed organisms intermittently in their milk in successive lactations. After parturition, most infected ewes shed C. burnetii in vaginal secretions for up to eight days and some (c. 12 per cent) for about two months.13 They only occasionally shed organisms at subsequent parturitions.94 Although infected dairy cows may excrete organisms in their milk for many years,34 ewes only shed organisms in their milk for a few days.34 It is not clear where C. burnetii persists in domestic animals between pregnancies,14 but in humans the DNA of C. burnetii can be detected in the bone marrow for several years after acute infection.36
Clinical signs
Infections of animals with C. burnetii seldom result in clinical signs. Following experimental infections of cattle with C. burnetii low-grade fevers have been reported25 and early reports indicated there may be pneumonia.66 Despite the presence of numerous organisms in the milk of infected cows, milk yields do not decrease.51 Reproductive disorders may,72 or may not,50 occur in infected cattle herds but, despite the predilection of C. burnetii for the trophoblast or foetus, abortion is uncommon.73, 83 Although still rare, abortions are more common in sheep and goats. 23, 56, 90 Up to 50 per cent of pregnant animals in a single flock may abort.22 New-born animals may be weak in herds in which abortions occur.65 During the years 1981 to 1987, the Section of Reproduction at the Onderstepoort Veterinary Institute found C. burnetii in eight out of 910 abortions in cattle (1 per cent); in seven out of 236 abortions in sheep (3 per cent), and in two out of 115 abortions in goats (2 per cent).38
Acute infections in humans are most often asymptomatic but can resemble a mild flu-like illness with fever, headache and myalgia that resolves spontaneously in a week. In some cases there may be atypical pneumonia which is sometimes fatal, granulomatous hepatitis, or more rarely meningoencephalitis, myocarditis or pericarditis.58 In most cases infections are controlled by T-cell immune responses, 59 but these do not eliminate C. burnetii and chronic infections may occur. This occurs particularly in humans with pre-existing heart valve disease, immunocompromised patients and pregnant women. The most common clinical presentation of chronic Q fever, which might occur months to years after infection, is endocarditis; vascular infections, chronic hepatitis and bone infections occur infrequently.58
Pathology
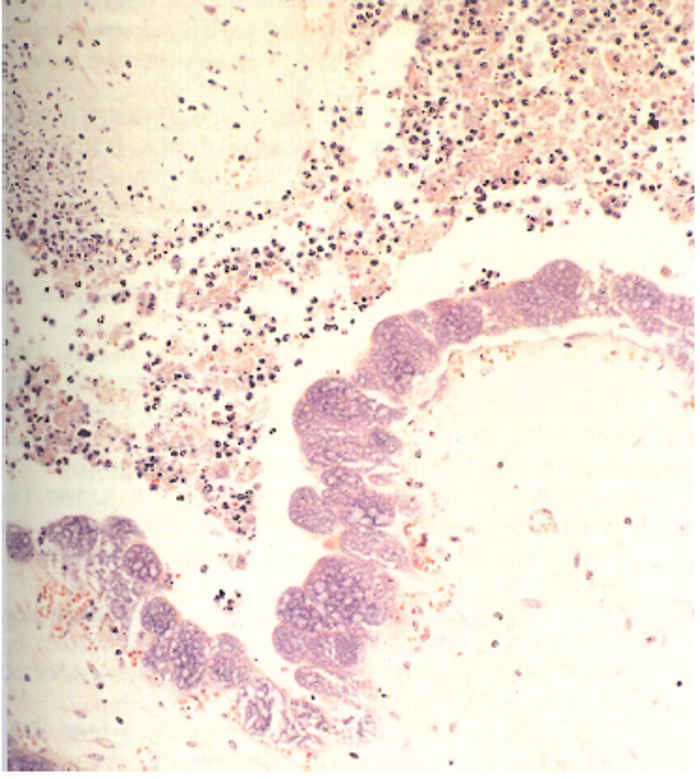
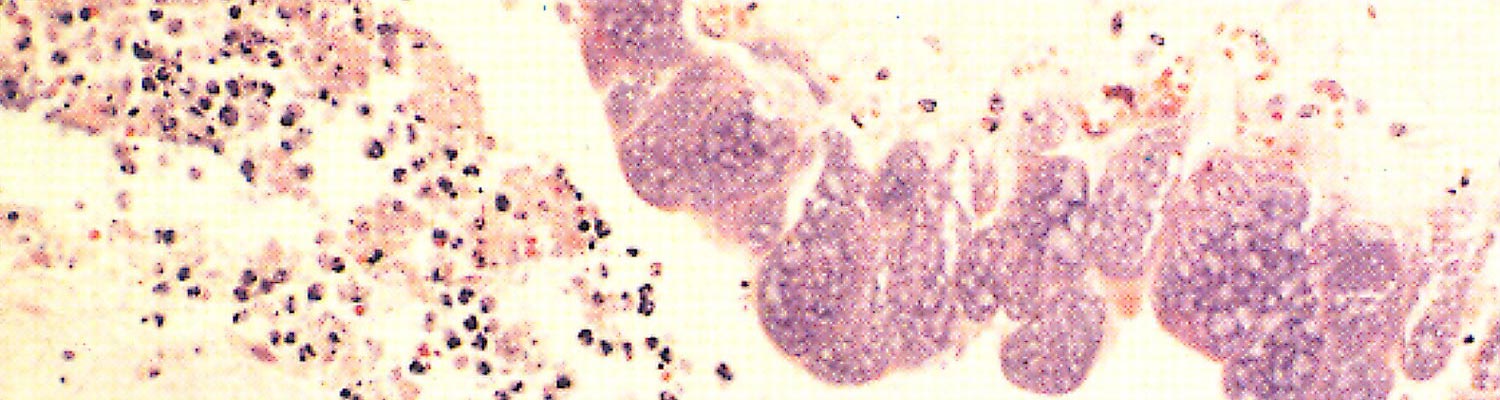
Few lesions have been reported in domesticated animals. Varying degrees of inflammation and necrosis of the cotyledons65, 76 occur in sheep and goats that have aborted as a result of C. burnetii infection. The placental lesions are most conspicuous in the intercotyledonary zone and are covered by abundant, inspissated exudate. The foetal membranes beneath the exudate are thickened, leathery and diffusely red to brownish. Multifocal areas of calcification, which tend to become confluent, also occur. The cotyledonary villi are necrotic, a prominent neutrophil infiltration occurs, and many C. burnetii are present in the trophoblast cells of the allantochorion (Figure 43.4). No gross lesions have been recorded in the foetus.61 In Coxiella- positive placentas from cattle that have aborted, placentitis occurs with the chorionic stroma infiltrated with mononuclear cells, necrosis of the chorionic trophoblast and focal exudation of fibrin and neutrophils.15 The placentitis in cattle is usually milder than that in sheep and fewer C. burnetii can be demonstrated immunocytochemically. 89
Diagnosis and differential diagnosis
Samples suspected to contain C. burnetii should be handled with extreme care as it is highly infectious and laboratory infections are not infrequent. Ideally, samples should be handled by experienced personnel in a biosafety level-three laboratory. Coxiella burnetii does not grow on artificial media but may be isolated in embryonated eggs, laboratory animals such as guinea pigs and mice, and in a number of tissue culture cell lines including mouse macrophage-like cells, fibroblasts and Vero cells 58 Diagnosis of C. burnetii infections by isolation of organisms is only available in very few laboratories because of the high risk of transmission to laboratory staff and the safety equipment and facilities required.
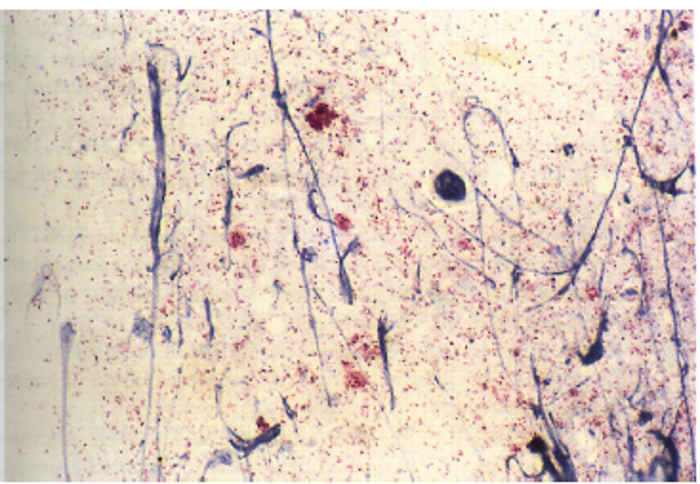
Staining with Giménez32 or the modified Ziehl-Neelsen stain (Figure 43.5)15 may be used to demonstrate organisms in impression smears made from affected cotyledons and chorion of placentas from animals that have aborted. Gram staining is unreliable, with C. burnetii appearing Gram positive under some conditions.33 Differentiating C. burnetii from other agents of abortion, for example Chlamydophila and Brucella, is often difficult in stained smears and immunofluorescence with mono- or polyclonal antibodies is recommended. This technique may be used on fresh and paraffin-embedded tissues. Organisms may also be detected with immunoperoxidase assays19, 89 and capture enzyme-linked immunosorbent assay (ELISA) or ELIFA84 but these techniques require fresh tissue. Care should be taken in interpreting results as C. burnetii may also be found in placentas of healthy sheep and goats.95
Recent studies have shown that C. burnetii can be detected by DNA probes29 and a number of polymerase chain reaction (PCR) assays have been described. A PCR with primers derived from the htpAB-associated sequence can be used to diagnose Q fever in humans and has been used to detect C. burnetii in cow’s milk,52, 88, 98 genital swabs, milk and faecal samples from ewes,12 cattle sera88 and dust from barns.100
In humans, Q fever is generally diagnosed using serology which enables differentiation of acute and chronic infections based on antibody responses to phase I and phase II organisms.87 Of the serological techniques available, the indirect fluorescent antibody test is the reference technique for diagnosis in humans; an ELISA has been reported to be useful for seroepidemiological studies.8, 67 The characteristics and value of other serological techniques such as the microagglutination, complement fixation, radio-immuno assay, indirect haemolysis, ELISA, dot immunoblotting and Western blotting, have been reviewed recently.43, 58 Serology may be used to detect infected animals but false negative results may occur.13
Control
The development of effective communication channels between public health officials and animal health workers is essential for the early diagnosis and control of Q fever outbreaks.
The control of C. burnetii infections is difficult as its extreme resistance to physical factors and chemical disinfection makes elimination of the organism from the environment problematic.3 Appropriate tick control strategies and good hygiene practices can decrease environmental contamination. Infected foetal fluids and membranes, aborted foetuses and contaminated bedding should be incinerated or buried. Infected animals should be removed from herds or provided with separate containment facilities in which to give birth. Detection of infected animals may be difficult, however, as serology is not reliable and the use of PCR is generally impractical.
Inactivated vaccines for domestic animals have been developed. A vaccine containing phase II organisms44 may be of economic benefit by reducing infertility, abortions and low birth weights. Outbreaks of Q fever in humans have, however, been associated with vaccinated animals,28 as this vaccine does not prevent shedding of C. burnetii, particularly in animals naturally infected before vaccination.18, 71 In Slovakia, large-scale cattle vaccination programmes with a single dose of vaccine containing phase I organisms have been associated with a decrease in the incidence of Q fever in humans.75
The need for treatment of infected domesticated animals seldom arises but, if necessary, tetracyclines are the drugs of choice. However, antibiotic treatment of cattle chronically infected with C. burnetii does not appear to prevent shedding of organisms.9 Doxycycline, 100 mg twice a day for 14 to 21 days, is the drug of choice for humans with acute Q fever.58 Chronic Q fever in humans is difficult to treat: it is recommended that patients be tested with doxycycline (100 mg twice a day) and chloroquine (200 mg three times a day) for 18 months or with doxycycline (100 mg twice a day) and ofloxacin (200 mg three times a day) for three years.58
Infections in humans can be minimized by ensuring that workers in the animal industry are fully informed about the risk factors of acquiring Q fever. Laboratories should be provided with appropriate safety facilities and equipment. Vaccination of humans in high-risk occupations should be considered: in Australia, a formalin-inactivated whole cell C. burnetii (Henzerling strain, phase 1) vaccine (Q-Vax, CSL) has proven highly effective in reducing Q fever in abattoir workers.2 Reducing exposure to raw milk and promoting the use of pasteurized milk and its products will also contribute to lowering the prevalence of Q fever.
References
- ABEBE, A., 1990. Prevalence of Q fever infection in the Addis Ababa abattoir. Ethiopian Medical Journal, 28, 119–122.
- ACKLAND, J.R., WORSWICK, D.A. & MARMION, B.P., 1994. Vaccine prophylaxis of Q fever. A follow-up study of the efficacy of Q-Vax (CSL) 1985–1990. Medical Journal of Australia, 160, 704–708.
- AITKEN, I.D., BÖGEL, K., CRACEA, E., EDLINGER, E., HOUWERS, K.H., RADY, M., REHACEK, J., SCHIEFER, H.G., SCHMEER, N., TARASEVICH, I.V. & TRINGALI, G., 1987. Q fever in Europe: Current aspects of aetiology, epidemiology, human infection, diagnosis and therapy. Infection, 15, 323–327.
- ANON., 1953. Annual Report, South African Institute of Medical Research.
- ANSTEY, N.M., TISSOT DUPONT, H., HAHN, C.G., MWAIKAMBO, E.D., MCDONALD, M.I., RAOULT, D. & SEXTON, D.J., 1997. Seroepidemiology of Rickettsia typhi, spotted fever group rickettsiae, and Coxiella burnetii infection in pregnant women from urban Tanzania. American Journal of Tropical Medicine and Hygiene, 57, 187–189.
- BABUDIERI, B., 1959. Q fever; a zoonosis. Advances in Veterinary Science, 5, 81–182.
- BACA, O.G. & PARETSKY, D., 1983. Q fever and Coxiella burnetii: A model for host-parasite interactions. Microbiological Reviews, 47, 127–149.
- BEHYMER, D.E., RUPPANER, R., BROOKS, D., WILLIAMS, J.C. & FRANTI, C.E., 1985. Enzyme immunoassay for surveillance of Q fever. American Journal of Veterinary Research, 46, 2413–2417.
- BEHYMER, D., RUPPANNER, R., RIEMANN, H.P., BIBERSTEIN, E.L. & FRANTI, C.E., 1977. Observation on chemotherapy in cows chronically infected with Coxiella burnetii (Q fever). Folia Veterinaria Latina, 7, 64–70.
- BELEC, L., GRESENGUET, G., EKALA, M.T., JACOB, A., VOHITO, M.D., COTIGNY, S. & PAYAN, C., 1993. Coxiella burnetii infection among subjects infected with HIV type 1 in the Central African Republic. European Journal of Clinical Microbiology and Infectious Diseases, 12, 775–778.
- BENSON, W.W., BROCK, D.W. & MATHER, J., 1963. Serological analysis of a penitentiary group using raw milk froma Q fever-infected herd. United States Public Health Service Reports, 78, 707–710.
- BERRI, M., LAROUCAU, K. & RODOLAKIS, A., 2000. The detection of Coxiella burnetii from ovine genital swabs, milk and fecal samples by the use of a single touchdown polymerase chain reaction. Veterinary Microbiology, 72, 285–293.
- BERRI, M., SOURIAU, A., CROSBY, M., CROCHET, D., LECHOPIER, P. & RODOLAKIS, A., 2001. Relationships between the shedding of Coxiella burnetii, clinical signs and serological responses of 34 sheep. The Veterinary Record, 148, 502–505.
- BIBERSTEIN, E.L., BEHYMER, D.E., BUSHNELL, R., CRENSHAW, G., RIEMANN, H.P. & FRANTI, C.E., 1974. A survey of Q fever (Coxiella burnetii) in California dairy cows. American Journal of Veterinary Research, 35, 1577–1582.
- BILDFELL, R.J., THOMSON, G.W., HAINES, D.M., MCEWEN, B.J. & SMART, N., 2000. Coxiella burnetii infection is associated with placentitis in cases of bovine abortion. Journal of Veterinary Diagnostic Investigation, 12, 419–425
- BONI, M., DAVOUST, B., TISSOT-DUPONT, H. & RAOULT, D., 1998. Survey of seroprevalence of Q fever in dogs in the southeast of France, French Guyana, Martinique, Senegal and the Ivory Coast. Veterinary Microbiology, 64, 1–5.
- BOTROS, B.A., SOLIMAN, A.K., SALIB, A.W., OLSON, J., HIBBS, R.G., WILLIAMS, J.C., DARWISH, M., EL TIGANI, A. & WATTS, D.M., 1995. Coxiella burnetii antibody prevalences among human populations in north-east Africa determined by enzyme immunoassay. Journal of Tropical Medicine and Hygiene, 98, 173–178.
- BROOKS, D.L., ERMEL, R.W., FRANTI, C.E., RUPPANER, R., BEHYMER, D.E., WILLIAMS, J.C. & STEPHENSON, J.C., 1986. Q fever vaccination of sheep: Challenge of immunity in ewes. American Journal of Veterinary Research, 47, 1235–1238.
- BROUQUI, P., DUMLER, J. S. & RAOULT, D., 1994. Immunohistologic demonstration of Coxiella burnetii in the valves of patients with Q fever endocarditis, American Journal of Medicine, 97, 451–458.
- BURNET, F.M. & FREEMAN, M., 1937. Experimental studies on the virus of ‘Q’ fever. Medical Journal of Australia, 2, 299–305.
- BURTON, P.R., KORDOVA, N. & PARETSKY, D., 1971. Electron microscopic studies of the rickettsia Coxiella burnetii: Entry, lysosomal response, and fate of rickettsial DNA in L-cells. Canadian Journal of Microbiology, 17, 143–150.
- COX, H.R. & BELL, E.J., 1939. The cultivation of Rickettsia diaporica in tissue culture and in the tissues of developing chicken embryos, Public Health Reports, 54, 2171–2175.
- CROWTHER, R.W. & SPICER, A.J., 1976. Abortion in sheep and goats in Cyprus caused by Coxiella burnetii. The Veterinary Record, 99, 29–30.
- DERRICK, E.H., 1937. ‘Q’ fever, a new fever entity; clinical features, diagnosis and laboratory investigation. Medical Journal of Australia, 2, 281–299.
- DERRICK, E.H., SMITH, D.J.W. & BROWN, H.E., 1942. Studies in the epidemiology of Q fever. IX. The role of the cow in the transmission of human infection. Australian Journal of Experimental Biology and Medicine, 20, 105–110.
- DYER, R.E., 1939. Similarity of Australian ‘Q fever’ and a disease caused by an infectious agent isolated from ticks. Public Health Reports, 53, 2277–2282.
- ENRIGHT, J.B., SADLER, W.W. & THOMAS, R.C., 1957. Thermal inactivation of Coxiella burnetii in milk pasteurization. Public health monograph no.47. U.S. Public Health Service publication no.517. U.S. Government Printing Office, Washington, D.C.
- FISHBEIN, D.B. & RAOULT, D., 1992. A cluster of Coxiella burnetii infections associated with exposure to vaccinated goats and their unpasteurized products. American Journal of Tropical Medicine and Hygiene, 47, 35–40.
- FRAZIER, M.E., HEINZEN, R.A., MALLAVIA, I.P. & BACA, O.G., 1992. DNA probes detecting Coxiella burnetii strains. Acta Virologica, 36, 83–89.
- GEAR, J.H.S., WOLSTENHOLME, B. & CART, A., 1950. Q fever: Serological evidence of the occurrence of a case in South Africa. South African Medical Journal, 24, 409–411.
- GHIROTTI, M., SEMPRONI, G., DE MENEGHI, D., MUNGABA, F.N., NANNINI, D., CALZETTA, G. & PAGANICO, G., 1991. Sero-prevalences of selected cattle diseases in the Kafue flats of Zambia. Veterinary Research Communications, 15, 25–36.
- GIMÉNEZ, D.F., 1964. Staining rickettsiae in yolk-sac cultures. Stain Technology, 39, 135–140.
- GIMÉNEZ, D. F., 1965. Gram staining of C. burnetii. Journal of Bacteriology, 110, 368–377.
- GRIST, N.R., 1959. The persistence of Q fever in a dairy herd. The Veterinary Record, 71, 839–841.
- GUMMOW, B., POERSTAMPER, N. & HERR, S., 1987. The incidence of Coxiella burnetii antibodies in cattle in the Transvaal. Onderstepoort Journal of Veterinary Research, 54, 569–571.
- HARRIS, R.J., STORM, P.A., LLOYD, A., ARENS, M. & MARMION, B.P., 2000. Long-term persistence of Coxiella burnetii in the host after primary Q fever. Epidemiology and Infection, 124, 543–549.
- HAWKER, J.I., AYRES, J.G., BLAIR, I., EVANS, M.R., SMITH, D.L., SMITH, E.G., BURGE, P.S., CARPENTER, M.J., CAUL, E.O., COUPLAND, B., DESSELBERGER, U., FARRELL, I.D., SAUNDERS, P.J. & WOOD, M.J., 1998. A large outbreak of Q fever in the West Midlands: windborne spread into a metropolitan area? Communicable Diseases and Public Health, 1, 180–187.
- HERR, S., 1988. Onderstepoort Veterinary Institute, Onderstepoort, unpublished data.
- HILBINK, F., PENROSE, M., KOVACOVA, E. & KAZAR, J., 1993. Q fever is absent from New Zealand. International Journal of Epidemiology, 22, 945–949.
- HUEBNER, R.J., JELLISON, W.L., BECK, M.D., PORTER, R.R. & SHEPARD, C.C., 1948. Q fever studies in southern California. I. Recovery of Rickettsia burnetii from raw milk. United States Public Health Reports, 63, 214–222.
- KAPLAN, M.M. & BERTAGNA, P., 1955. The geographical distribution of Q fever. Bulletin of the World Health Organisation, 13, 829–860.
- KELLY, P.J., MATTHEWMAN, L.A., MASON, P.R. & RAOULT, D. 1993. Q fever in Zimbabwe. A review of the disease and the results of a serosurvey of humans, cattle, goats and dogs. South African Medical Journal, 83, 21–25.
- KOVACOVA, E., KAZAR, J. & SPANELOVA, D., 1998. Suitability of various Coxiella burnetii antigen preparations for detection of serum antibodies by various tests. Acta Virologica, 42, 365–368.
- KRAUSS, H., 1989. Clinical aspects and prevention of Q fever in animals. European Journal of Epidemiology, 5, 454–455.
- KRIEG, N.R. & HOLT, J.G., 1984. Bergey’s Manual of Systematic Bacteriology. Vol. I. Baltimore: Williams & Wilkins.
- LANG, G.H., 1990. Coxiellosis (Q fever) in animals. In: MARIE, T.J., (ed.). Q fever. The Disease. Vol. I. Boca Raton, Florida: CRC Press, Inc.
- A SCOLA, B., LEPIDI, H., MAURIN, M. & RAOULT, D., 1998. A guinea pig model for Q fever endocarditis. Journal of Infectious Diseases, 178, 278–281.
- LETAIEF, A.O., YACOUB, S., DUPONT, H.T., LE CAM, C., GHACHEM, L., JEMNI, L. & RAOULT, D., 1995. Seroepidemiological survey of rickettsial infections among blood donors in central Tunisia. Transactions of the Royal Society of Tropical Medicine and Hygiene, 89, 266–268.
- LENETTE, E.H. & WELSH, H.H., 1951. Q fever in California. X. Recovery of Coxiella burnetii from the air of premises harboring infected goats. American Journal of Hygiene, 54: 44–49.
- LITERAK, I. & KROUPA L., 1998. Herd-level Coxiella burnetii seroprevalence was not associated with herd-level breeding performance in Czech dairy herds. Preventative Veterinary Medicine, 33, 261–265.
- LITTLE, T.W.A., 1983. Q fever—an enigma. British Veterinary Journal, 139, 277–283.
- LORENZ, H., JAGER, C., WILLEMS, H. & BALJER, G., 1998. PCR detection of Coxiella burnetii from different clinical specimens, especially bovine milk, on the basis of DNA preparation with a silica matrix. Applied and Environmental Microbiology, 64, 4234–4237.
- MCCAUL, T.F. & WILLIAMS, J.C., 1981. Development cycle of Coxiella burnetii: structure and morphogenesis of vegetative and sporogenic differentiations. Journal of Bacteriology, 147, 1063–1076.
- MARIE, T.J., DURANT, H., WILLIAMS, J.C., MINTZ, E. & WANG, D.M., 1988. exposure to parturient cats is a risk factor for acquisition of Q fever in Maritime Canada. Journal of Infectious Diseases, 158, 101–108.
- MARMION, B.P., ORMSBEE, R.A., KYRKOU, M., WRIGHT, J., WORSWICK, D.A., IZZO, A.A., ESTERMAN, A., FEERY, B. & SHAPIRO, R.A., 1990. Vaccine prophylaxis of abattoir-associated Q fever: eight years’ experience in Australian abattoirs. Epidemiology and Infection, 104, 275–287.
- MARMION, B.P. & WATSON, W.A., 1961. Q fever and ovine abortion. Journal of Comparative Pathology and Therapeutics, 71, 360–369.
- MATTHEWMAN, L., KELLY, P.J., HAYTER, D., DOWNIE, S., WRAY, K., BRYSON, N., RYCROFT, A. & RAOULT, D., 1997. Exposure of cats in southern Africa to Coxiella burnetii, the agent of Q fever. European Journal of Epidemiology, 13, 477–479.
- MAURIN, M. & RAOULT, D., 1999. Q fever. Clinical Microbiology Reviews, 12, 518–553.
- MEGE, J.L., MAURIN, M., CAPO, C. & RAOULT. D., 1997. Coxiella burnetii: the ‘query’ fever bacterium. A model of immune subversion by a strictly intracellular microorganism. FEMS Microbiology Reviews, 19, 209–217.
- MILAZZO, A., HALL, R., STORM, P.A., HARRIS, R.J., WINSLOW, W. & MARMION, B.P., 2001. Sexually transmitted Q fever. Clinical Infectious Diseases, 33, 399–402.
- MILLER, R.B., PALMER, N.C. & KIERSTAD, M., 1986. Coxiella burnetii infection in goats. In: MORROW, D.A. (ed.). Current Therapy in Theriogenology. II. Obstetrics-Veterinary. Philadelphia: W.B. Saunders.
- NGUYEN, S.V. & HIRAI, K., 1999. Differentiation of Coxiella burnetii isolates by sequence determination and PCR-restriction fragment length polymorphism analysis of isocitrate dehydrogenase gene. FEMS Microbiology Letters, 180, 249–254.
- OKABAYASHI, T., HASEBE, F., SAMUI, K.L., MWEENE, A.S., PANDEY, S.G., YANASE, T., MURAMATSU, Y., UENO, H. & MORITA C., 1999. Short report: prevalence of antibodies against spotted fever, murine typhus, and Q fever rickettsiae in humans living in Zambia. American Journal of Tropical Medicine and Hygiene, 61, 70–72.
- PAIBA, G.A., GREEN, L.E., LLOYD, G., PATEL, D. & MORGAN, K.L., 1999. Prevalence of antibodies to Coxiella burnetii (Q fever) in bulk tank milk in England and Wales. The Veterinary Record, 144, 519–522.
- PALMER, N.C., KIERSTAD, M., KEY, D.W., WILLIAMS, J.C., PEACOCK, M.G. & VELLEND, H., 1983. Placentitis and abortion in goats and sheep in Ontario caused by Coxiella burnetii. Canadian Veterinary Journal, 24, 60–61.
- PAYZIN, S., 1953. Epidemiological investigations on Q fever in Turkey. Bulletin of the World Health Organization, 9, 553–558.
- PETER, O., DUPUIS G., BEE D., LUTHY R., NICOLET J. & BURGDORFER. W., 1988. Enzyme-linked immunosorbent assay for diagnosis of chronic Q fever. Journal of Clinical Microbiology, 26, 1978–1982.
- RHODE, C., KELLY, P.J. & RAOULT, D., 1993. Dairy cows as reservoirs of Coxiella burnetii in Zimbabwe. Central African Journal of Medicine, 39, 208–210.
- SALMON, M.M., HOWELLS, B., GLENCROSS, E.J.G., EVANS, A.D. & PALMER, S.R., 1982. Q fever in an urban area. Lancet, I,1002–1004.
- SANER, R.G. & FEHLER, B.M., 1950. A case of Q fever contracted on the Witwatersrand. South African Medical Journal, 24, 1000–1002.
- SCHMEER, N., MULLER, P., LANGEL, J., KRAUSS, H., FROST, J. W. & WIEDA, J., 1987. Q fever vaccines for animals. Zentralblatt furBakteriologie, Microbioogie und Hygiene, A 267, 79–88.
- SCHMEER, N., WIEDA, J., FROST, J.W., HERBERT, W., WEISS, R. & KRAUSS, H., 1987. Diagnose, Differentialdiagnose und Bekämpfung des bovinen Q-Fiebers in einem Vorzugsmilchbestand mit Fruchbarkeitertorungen. Tierärztliche Umschau, 42, 287, 290–296.
- SCHUTTE, A.P., KURZ, J., BARNARD, B.J.H. & ROUX, D.J., 1976. Q fever in cattle and sheep in southern Africa. A preliminary report. Onderstepoort Journal of Veterinary Research, 43, 129–132.
- SCOTT, G.H. & WILLIAMS, J.C., 1990. Susceptibility of Coxiella burnetii to chemical disinfectants. Annals of the New York Academy of Science, 590, 291–296.
- SERBEZOV, V.S., KAZAR, J., NOVKIRISHKI, V., GATCHEVA, N., KOVACOVA, E. & VOYNOVA, V., 1999. Q fever in Bulgaria and Slovakia. Emerging Infectious Diseases, 5, 388–394.
- SPICER, A.J., CROWTHER, R.W., VELLA, E.E., BENGTSSON, E., MILES, R. & PITZOLIS, G., 1977. Q fever and animal abortion in Cyprus. Transactions of the Royal Society of Tropical Medicine and Hygiene, 71, 16–20.
- STALEY, G.P., MYBURGH, J.G. & CHAPARRO, F., 1989. Serological evidence of Q fever in cattle in Malawi. Onderstepoort Journal of Veterinary Research, 56, 205–206.
- STEIN, A. & RAOULT, D., 1999. Pigeon pneumonia in Provence: a bird-borne Q fever outbreak, Clinical Infectious Diseases, 29, 617–620.
- STEIN, A., SAUNDERS, N.A., TAYLOR, A.G. & RAOULT, D., 1993. Phylogenic homogeneity of Coxiella burnetii strains as determinated by 16S ribosomal RNA sequencing. FEMS Microbiology Letters, 113, 339–344.
- STOKER, M.G.P., BROWN, R.D., KETT, F.J.L., COLLINGS, P.C. & MARMION, B.P., 1955. Q fever in Britain. Isolation of Rickettsia burnetii from placenta and wool of sheep in an endemic area. Journal of Hygiene, 53, 313–321.
- STOKER, M.G.P. & FISET, P., 1956. Phase variation of the Nine Mile and other strains of Rickettsia burnetii. Canadian Journal of Microbiology, 2, 310–321.
- STOCKER, M.G.P. & MORRISON, B.P. 1955. The spread of Q fever from animals to man: the natural history of rickettsial disease. Bulletin of the WHO.
- TAINTURIER, D., 1987. Métrites en série chez la vache provoquées par la fièvre Q. Recueil de Médecine Vétérinaire, 163, 195–198.
- THIELE, D., KARO, M. & KRAUSS, H., 1992. Monoclonal antibody based capture ELISA/ELIFA for detection of Coxiella burnetii in clinical specimens, European Journal of Epidemiology, 8, 568–574.
- THOMAS, D.R., TREWEEK, L., SALMON, R.L., KENCH, S.M., COLEMAN, T.J., MEADOWS, D., MORGAN-CAPNER, P. & CAUL, E.O., 1995. The risk of acquiring Q fever on farms: a seroepidemiological study. Occupational and Environmental Medicine, 52, 644–647.
- TIGERTT, W.D., BENENSON, A.S. & GOCHENOUR, W.S., 1961. Airborne Q fever. Bacteriological Reviews, 25, 285–293.
- TISSOT DUPONT, H., THIRION, X. & RAOULT, D., 1994. Q fever serology: cutoff determination for microimmunofluorescence. Clinical and Diagnostic Laboratory Immunology, 1, 189–196.
- TO, H., HTWE, K.K., KAKO, N., KIM, H.J., YAMAGUCHI, T., FUKUSHI, H. & HIRAI, K. 1998. Prevalence of Coxiella burnetii infection in dairy cattle with reproductive disorders. Journal of Veterinary Medical Science, 60, 859–861.
- VAN MOLL, P., BAUMGARTNER, W., ESKENS, U. & HANICHEN, T., 1993. Immunocytochemical demonstration of Coxiella burnetii antigen in the fetal placenta of naturally infected sheep and cattle. Journal of Comparative Pathology, 109, 295–301.
- WALDHALM, D.G., STOENNER, H.G., SIMMONS, R.E. & THOMAS, L.A., 1978. Abortion associated with Coxiella burnetii infection in goats. Journal of the American Veterinary Medical Association, 173, 1580–1581.
- WEISBURG, W.G., DOBSON, M.E., SAMUEL, J.E., DASCH, G.A., MALLAVIA, L.P., BACA, O., MANDELCO, L., SECHREST, J.E., WEISS, E. & WOESE, C.R., 1989. Phylogenetic diversity of the Rickettsiae. Journal of Bacteriology, 171, 4202–4206.
- WEISMANN, E., 1952. Die Q-fever Forschung in der Schweiz in den Jahren 1947–1951. Zeitschrift für Tropenmedizin und Parasitologie, 3, 297–301.
- WEISS, E., 1973. Growth and physiology of rickettsiae. Bacteriological Reviews, 37, 259–283.
- WELSH, H.H., JENSEN, F.W. & LENETTE, E.H., 1959. Q fever studies. XX. Comparison of four serological techniques for the detection and measurement of antibody to Coxiella burnetii in naturally exposed sheep. American Journal of Hygiene, 70, 1–13.
- WELSH, H.H., LENNETTE, E.H., ABINANTI, F.R. & WINN, J.F., 1951. Q fever in California. IV. Occurrence of Coxiella burnetii in the placenta of naturally infected sheep. United States Public Health Reports, 66, 1473–1477.
- WILLEMS, H., JAGER, C. & BALJER, G., 1998. Physical and genetic map of the obligate intracellular bacterium Coxiella burnetii. Journal of Bacteriology, 180, 3816–3822.
- WILLEMS, H., THIELE, D., BURGER, C., RITTER, M., OSWALD, W. & KRAUSS, H., 1996. Molecular biology of Coxiella burnetii. In: KAZAR, J. & TOMAN, R., (eds). Rickettsiae and Rickettsial Diseases. Bratislava, Slovakia: Slovak Academy of Sciences. pp. 363–378.
- WILLEMS, H., THIELE, D., FRÖLICH-RITTER, R. & KRAUSS. H., 1994. Detection of Coxiella burnetii in cow’s milk using the polymerase chain reaction. Zentralblatt fur Veterinärmedicine, 41, 580–587.
- WILLIAMS, J.C., JOHNSTON, M.R., PEACOCK, M.G., THOMAS, L.A., STEWART, S. & PORTIS, J.L., 1984. Monoclonal antibodies distinguish phase variants of Coxiella burnetii. Infection and Immunity, 43, 421–428.
- YANASE, T., MURAMATSU, Y., INOUYE, I., OKABAYASHI, T., UENO, H. & MORITA, C., 1998. Detection of Coxiella burnetii from dust in a barn housing dairy cattle. Microbiology and Immunology, 42, 51–53.