- Infectious Diseases of Livestock
- Part 1
- Bovine anaplasmosis
- Vectors: Ticks
- Vectors: Tsetse flies
- Vectors: Muscidae
- Vectors: Tabanidae
- Vectors: Culicoides spp.
- Vectors: Mosquitoes
- Classification, epidemiology and control of arthropod-borne viruses
- Special factors affecting the control of livestock diseases in sub-Saharan Africa
- The control of infectious diseases of livestock: Making appropriate decisions in different epidemiological and socioeconomic conditions
- Infectious diseases of animals in sub-Saharan Africa: The wildlife⁄livestock interface
- Vaccination: An approach to the control of infectious diseases
- African animal trypanosomoses
- Dourine
- Trichomonosis
- Amoebic infections
- GENERAL INTRODUCTION: COCCIDIA
- Coccidiosis
- Cryptosporidiosis
- Toxoplasmosis
- Besnoitiosis
- Sarcocystosis
- Balantidiosis
- Leishmaniosis
- Neosporosis
- Equine protozoal myeloencephalitis
- GENERAL INTRODUCTION: BABESIOSES
- Bovine babesiosis
- Equine piroplasmosis
- Porcine babesiosis
- Ovine babesiosis
- GENERAL INTRODUCTION: THEILERIOSES OF CATTLE
- East Coast fever
- Corridor disease
- Zimbabwe theileriosis
- Turning sickness
- Theileria taurotragi infection
- Theileria mutans infection
- Theileria annulata theileriosis
- Theileriosis of sheep and goats
- Theileria buffeli⁄orientalis infection
- Non-pathogenic Theileria species in cattle
- GENERAL INTRODUCTION: RICKETTSIAL, CHLAMYDIAL AND HAEMOTROPIC MYCOPLASMAL DISEASES
- Heartwater
- Lesser known rickettsial infections in animals and humans
- Chlamydiosis
- Q fever
- Eperythrozoonosis
- Bovine Haemobartonellosis
- Potomac horse fever
- GENERAL INTRODUCTION: ANAPLASMOSES
- Bovine anaplasmosis
- Ovine and caprine anaplasmosis
Bovine anaplasmosis
This content is distributed under the following licence: Attribution-NonCommercial CC BY-NC  View Creative Commons Licence details here
View Creative Commons Licence details here

Bovine anaplasmosis
Previous authors:F T POTGIETER AND W H STOLTSZ
Current authors:
SM Noh - Research Veterinary Medical Officer, DVM, PhD, DACVP, USDA-ARS Animal Disease Research Unit, Washington State University, Pullman, Washington, USA
KA Brayton - USDA-ARS Animal Disease Research Unit, Washington State University, Pullman, Washington, USA
Introduction
Bovine anaplasmosis is a tick-borne disease caused predominantly by Anaplasma marginale, though a few reports indicate that Anaplasma centrale can also cause clinical disease. This pathogen has a worldwide distribution and is endemic in temperate, subtropical and tropical regions throughout the world. Bovine anaplasmosis is generally characterized by fever and progressive anaemia. In some instances, rapid destruction of erythrocytes by the reticuloendothelial system results in icterus, though this is not consistent. Infections can vary from clinically inapparent to severe, with clinical disease ranging from mild, self-limiting disease in small numbers of animals to larger herd outbreaks characterized by severe anaemia and unexpected death. Disease severity tends to increase with age and is typically inapparent in cattle less than nine months of age, although other, unknown factors, likely play a role in determining disease severity. Once infected, if animals survive initial infection, they become lifelong inapparent carriers and thus serve as a pathogen reservoir for ongoing transmission.
Historical perspective
‘Peripheral coccus-like bodies’ and ‘marginal points’ were observed in erythrocytes of cattle by Sir Arnold Theiler in 1910, which was long before he identified these inclusion bodies as being the causative organism of the specific disease known by local farmers as gallsickness168, 169, 170. Mixed infections of Babesia bigemina and A. marginale, which frequently occur, led earlier workers to believe that these ‘coccus-like bodies’ represented a stage in the life cycle of B. bigemina158. In 1911, Theiler171 referred to the following serendipitous discovery as a ‘lucky coincidence’. In 1908, cattle due to be imported from England to South Africa were vaccinated with a South African isolate of B. bigemina. On their arrival in the province then known as Transvaal, Theiler challenged them with blood from immune local cattle. Fifty per cent of these animals died, having shown fever, icterus, severe anaemia, ‘marginal bodies or points’ in the erythrocytes but no haemoglobinuria, and this led him to believe that these deaths were due to an agent other than B. bigemina. He concluded that the ‘marginal points’ were independent organisms responsible for the acute signs observed and called them anaplasmata because of their lack of cytoplasm, and the disease caused by them anaplasmosis. The specific organisms were called A. marginale because most resided near the margin (Figure 1) of the erythrocyte9.
In 1911 Arnold Theiler171 discovered a second, similar bacterium, which proved to be invaluable in the control of anaplasmosis. He named this related organism A. marginale variety centrale (Figure 2). By virtue of an erroneous report by Ristic who stated that Theiler had named this organism as a separate species, the name A. centrale was taxonomically validated, but it was only in 2018 that evidence was published establishing A. centrale was indeed a separate species from A. marginale67, 74, 143. Theiler based the distinction between A. marginale and A. centrale on: the different position taken up by the two organisms within the erythrocyte, the lower virulence of A. centrale, and the incomplete cross-immunity to A. marginale demonstrated in animals that recovered from A. centrale infections.
Theiler immediately realized the tremendous potential of A. centrale for use in a vaccine, and the difference in the lengths of the incubation periods of B. bigemina and A. centrale made it possible and practical to vaccinate imported cattle with a combined B. bigemina (redwater) and anaplasmosis vaccine, which was sold for a shilling172.
The A. centrale isolate that Theiler made has been used extensively in a live-blood based vaccine in southern Africa, Australia, Israel and South America. Therefore, the distinct possibility exists that any subsequent isolations of A. centrale in these countries may represent reisolation of the original vaccine strain. A more detailed account of the history of anaplasmosis in the southern African region and elsewhere is given by Henning61.
Aetiology and life cycle
Anaplasma marginale is a Gram negative, obligate intracellular bacteria in the family Anaplasmataceae, which includes the genera Anaplasma, Ehrlichia, Neorickettsia and Wolbachia44. Anaplasma spp. and Ehrlichia spp. are tick transmitted pathogens that cause disease in a variety of animals and humans. Less is known about Neorickettsia spp, which have a complex life cycle that involves multiple invertebrates. Disease in domestic animals or humans, when it occurs, tends to be due to inadvertent infection, rather than a natural component of the pathogen life cycle. Wolbachia are found in many arthropods, and have a complex and variable relationship with the host, often having an endosymbiotic relationship. Interestingly, organisms in this family are often associated with invertebrate hosts or vectors and are thought to have a long co-evolutionary relationship with these invertebrates.
The formal classification of the genus Anaplasma includes A. marginale, A. centrale, A. ovis, A. bovis, A. caudatum, A. phagocytophilum and A. platys. With the advent of deep sequencing new Anaplasma species are being detected, some being named prematurely, and their relevance indeterminant. One putative species with mounting evidence as a zoonotic agent is A. capra, but this name has not been validated yet. Anaplasma caudatum is thought to be a tailed strain of A. marginale, and not a distinct species. Anaplasma marginale and A. centrale are the two organisms that primarily infect bovine erythrocytes and cause bovine anaplasmosis. Anaplasma marginale is the primary pathogen, while A. centrale can cause mild disease, and is best known for its capacity to provide protective immunity against A. marginale as a live, blood-based vaccine.
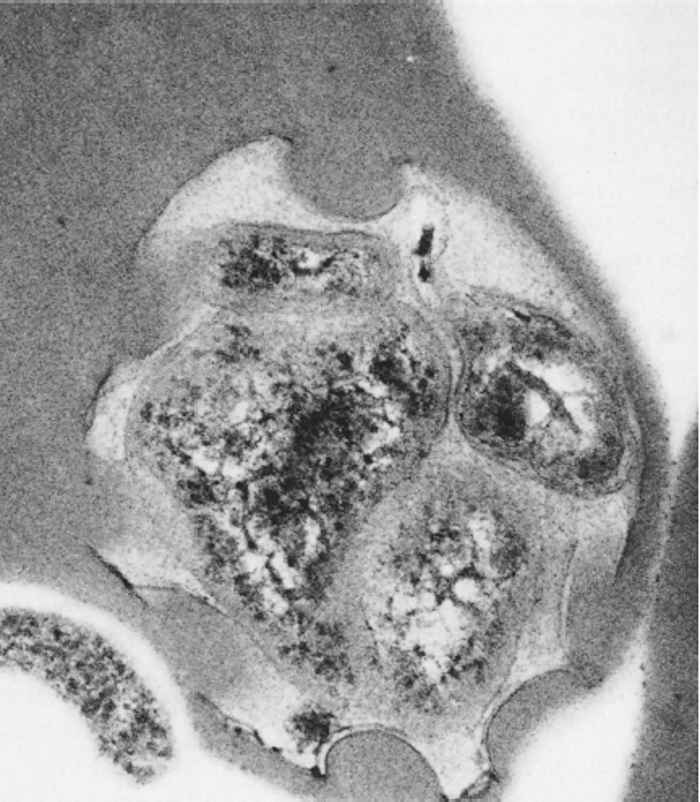
Upon tick transmission, A. marginale enters and replicates in erythrocytes, though endothelial cells may also be infected27. In Giemsa-stained blood smears, the intra-erythrocytic organisms are dense, deeply purple, roundish inclusions 0,3 to 1,0 μm in diameter (Figure 1) and (Figure 2). Anaplasma marginale and A. centrale are morphologically indistinguishable, though A. centrale is typically located in a more central position in the erythrocyte, while A. marginale tends to remain in the periphery as their names indicate. Ultrastructurally, Anaplasma inclusions are bound by a membrane and can contain up to eight bacteria. Typical of the Anaplasmataceae, the individual bacteria have outer and inner cell membranes50, 146 (Figure 3).
Ixodid ticks are the principal biological vectors of A. marginale, and primarily include Rhipicephalus, Dermacentor and Hyalomma species77, 131, 132, 156, 178. Amblyomma spp. are not competent vectors for A. marginale, nor are Argasid ticks. It is important to note that detection of pathogen DNA within a tick is insufficient evidence for vector competence.
The life cycle of A. marginale in adult D. andersoni ticks has been well-characterized78, 80, 81, 125, 160, 162. Within the midgut lumen, the blood meal is lysed and A. marginale enter the midgut epithelium using an as yet to be determined mechanism and replicates via binary fission to form large colonies. During ingestion of a second blood meal, the A. marginale travel to the salivary glands, where they enter the acinar cells, replicate to form large colonies and are released to the mammalian host along with the salivary gland secretions. Though not experimentally examined in all competent vectors, A. marginale is intrastadially and transtadially transmitted.
Only short-term erythrocyte cultures have been successful, and cell-to cell spread does not occur73. However, A. marginale can be maintained in vitro in several tick cell culture lines derived from embryonic cells including DAE100T (D. andersoni), DVE1 (Dermacentor variabilis), IDE8, IDE12 and ISE6 (I. scapularis), and BME26 (R. microplus). In addition, A. marginale can be propagated in Vero cells derived from kidney epithelial cells of an African Green monkey, Cercopithecus aethiops, as well as RF/6A cells derived from retina choroid endothelium of a rhesus monkey, Macaca mutatta31, 62, 111. Importantly the morphology and development cycle of A. marginale in tick cell culture are similar to those in naturally infected Dermacentor spp. ticks18, 76.
Epidemiology
Bovine anaplasmosis has a world-wide distribution and is endemic in many temperate, sub-tropical and tropical regions. Although bovine anaplasmosis is reportable to the World Organization for Animal Health (OIE), the amount of surveillance and reporting varies widely depending on national priorities of affected countries. Consequently, the availability of prevalence data is largely dependent on local research efforts. Data regarding incidence and economic impact of the disease in most regions is nearly absent.
The prevalence of A. marginale tends to be higher in tropical and subtropical regions than in temperate regions, but prevalence varies based on local and regional conditions and management practices. For example, Ghana is a tropical country, however, due to its north - south orientation, it has three vegetative zones: sub-humid dry, sub-humid humid, and humid. Accordingly, the highest within herd prevalence based on PCR to detect A. marginale is in the humid region of the coastal savannah with 75 per cent prevalence. The lowest prevalence (45 per cent) is in the sub-humid humid region of central Ghana15. Similarly, in Benin, also a tropical west African country, the more humid regions have a higher A. marginale prevalence of 77 per cent, based on PCR. While than the drier, more northern regions have a prevalence of approximately 35 per cent5.
In east Africa, reports from Ethiopia, Kenya and Tanzania, the prevalence based on either PCR or serology, is 5 per cent, 58 per cent, and 20-37 per cent, respectively1, 53, 165. In cattle rearing areas of South Africa, PCR or serology indicates prevalence between 26-98 per cent64, 101, 110 and 25 per cent in Zambia167.
There are a number of studies reporting the prevalence of A. marginale in Central and South America. The reported prevalence in Puerto Rico is 24 per cent179, while between 0-75 per cent of animals within herds in regions of Brazil35, 56 are seropositive for A. marginale.
In temperate regions of North America, herd prevalence in outbreaks in Manitoba, Canada, long considered free of A. marginale, was 0.04 to 66 per cent in a 2009 outbreak66. In one herd in California, USA, seroprevalence was between 90 to95 per cent in mature cows, and 31to37 per cent in cows in the high desert of south central Idaho98, 176. Recent studies indicate the seroprevalence in the Southeast United States, including Texas, Mississippi, Georgia and Kentucky varies from 4 per cent in Georgia to 88 per cent in some herds in Texas58, 121, 122, 123, 124. In the Midwest, 15 per cent of animals were seropositive in two feedlots in Iowa33. In Kansas herd prevalence was reported at 52.5 per cent159.
Anaplasma marginale can be transmitted by ticks, biting flies, iatrogenically via needles and equipment contaminated with blood, and transplacentally. Tick transmission is considered the most efficient means of transmission as A. marginale replicate within the tick and one to three ticks is sufficient to transmit the pathogen151, 153.
The ability of particular strains or isolates of A. marginale to be tick transmitted is a stable trait. For example, the St. Maries strain of A. marginale isolated in a temperate region of North America, is readily transmitted by R. microplus, a tropical tick. Similarly an isolate of A. marginale from Puerto Rico is readily transmissible by Dermacentor andersoni, a tick found in temperate regions of the western United States52. In contrast, a couple of strains of A. marginale have been isolated that are seemingly not tick transmissible. The best described are a strain from Florida and an isolate of A. marginale from bison in Saskatchewan, Canada152, 157. The A. centrale isolate contained in the commercial vaccine produced by the Onderstepoort Veterinary Institute can be transmitted by R. simus, but not by B. microplus or B. decoloratus135, whereas both the latter species are capable of transmitting A. marginale. D. andersoni is capable of transmission of both A. marginale and A. centrale, though transmission of A. centrale is markedly less efficient177.
Blood feeding diptera in the genera Tabanus and Stomoxys can also transmit A. marginale mechanically. Based on the repeated detection of non-tick transmissible strains of A. marginale, and the presence of large numbers of biting flies in some environments, biting flies can play an important role in the epidemiology of A. marginale. Instruments used in veterinary procedures, such as castration, dehorning and ear-tagging operations, which are contaminated with fresh blood, may also be responsible for the mechanical transfer of the infection42, 139. Circumstantial evidence indicates that some anaplasmosis outbreaks have followed vaccination programmes in South Africa, incriminating needle transmission between animals.
Transplacental transmission may also play a role in the maintenance of A. marginale within a herd. It has been estimated that 10 to 15 per cent of calves born to A. marginale infected cows will be infected with A. marginale though the development of clinical signs in neonates is unusual55, 134, 184. Overall the route of transmission that predominates in a particular herd or area depends on a variety of environmental conditions and management practices.
Once infected, animals typically remain infected for life and serve as reservoirs for ongoing transmission, though not all animals develop clinically apparent disease28, 29. Calves under six months old, irrespective of the immune status of their dams, tend to be resistant to severe clinical disease, but the risk of clinical anaplasmosis increases with age69, 113, 128. In endemic areas with high transmission rates, natural infection involving all animals in a herd during calfhood will help ensure minimal disease, often termed endemic stability.
The risk factors for the development of disease, including herd outbreaks, are poorly understood and likely multifactorial. Introduction of naïve, adult animals into an infected herd, variation in tick burdens through time leading to episodic periods of high transmission, variation in virulence of A. marginale strains, concurrent infections with other pathogens, and efficiency at which particular vector populations transmit the pathogen may all influence the likelihood that individual animals or groups of animals develop clinical disease.
Other domestic ruminants including sheep and goats can harbour A. marginale, though the ability to cause disease in small ruminants is unknown37. Additionally, little is known about the role wildlife species play in the epidemiology of bovine anaplasmosis. It is important to differentiate between the susceptibility of a particular species to infection in laboratory conditions, presence of the pathogen in wild populations, and likelihood of serving as a reservoir for domestic animals. An additional consideration is that the results of serologic surveys to detect Anaplasma spp. are difficult to interpret as antibody cross reactivity among all Anaplasma spp. and Ehrlichia spp. is common.
In South Africa, blesbok (Damaliscus dorcas phillipsi), common duiker (Sylvicapra grimmia L), and black wildebeest (Connochaetes gnou) are susceptible to experimental infection with A. marginale, but infections are subclinical112. In north America, Bison (Bison bison), elk (Cervus elaphus), pronghorn antelope (Antelocapra americana), and bighorn sheep (Ovis canadensis) are susceptible to infection.11, 60
In the Americas, A. marginale has been identified in a number of species of deer, including Andean deer (Hippocamelus antisensis) in Argentina and Pampas deer (Ozotoceros bezoarticus leucogaster) in the Brazilian Pantanal57, 129. Mule deer (Odocoileus hemionus) and black tailed deer (Odocoileus hemionus culumbianus) populations in the US are naturally infected with A. marginale, though white tailed deer (Odocoileus virginianus) seem to be resistant to infection11.
Pathogenesis
After primary infection, there is a prepatent period that terminates when the first infected erythrocytes are detected in blood smears. This is followed by a phase during which there is increasing parasitaemia and anaemia, and that ends either in death (preceded by evidence of severe anaemia and other clinical signs) or convalescence and the development of a carrier state.
The duration of the prepatent period seems to be directly related to the number of organisms in the inoculum, body weight of the host and route of infection. Generally, prepatent periods are shortest after intravenous inoculation, longer after intramuscular inoculation, and even longer following subcutaneous injection. The prepatent period can be as long as three to five weeks146. The course of the infection is not affected by the size of the inoculum82, but prepatency may vary even between animals which receive the same infective doses132. In spite of this, the number of organisms in live-blood vaccines is manipulated in such a way that the desired prepatency is achieved so that a reasonable synchronization of reactions results in those animals that will react clinically to the vaccine. Similarly, in the manufacture of frozen vaccine, adjustments can be made to the number of organisms per vaccine dose to compensate for loss in viability after freezing and thawing of the infected blood130. Prepatent periods may be prolonged after administration of tetracyclines68.
The intensity and duration of parasitaemia and the resulting anaemia are directly related to the age of the host69. Increasing anaemia may develop over 4 to 15 days, and animals may lose up to 70 per cent of their circulating erythrocytes. Maximal anaemia usually occurs one to six days after the peak parasitaemia. In animals that survive the patent phase, convalescence begins with evidence of erythropoiesis. The time of convalescence is related to the ability of the animal to produce new erythrocytes.
Although the manner by which erythrocytes become infected is not clear, an electron microscopic study has revealed how initial bodies penetrate the erythrocytic membrane147. It has been suggested that a direct transfer of initial bodies between erythrocytes takes place147. Increased osmotic fragility, preceded by a decrease in erythrocytic acetylcholinesterase has been observed in bovine anaplasmosis155, 180. This indicates that changes in the acetylcholinesterase activity may be associated with increased cell membrane permeability, which in turn facilitates the entry of A. marginale organisms into red blood cells155.
The pathogenesis of the anaemia in anaplasmosis was originally thought to be due to destruction of the erythrocytes by the parasites. However, the absence of haemoglobinaemia and haemoglobinuria indicates that intravascular haemolysis is not a feature of the disease68. The anaemia is caused by extensive erythrophagocytosis initiated by parasite- induced red blood cell damage12. Erythrocytes that become physically and chemically altered during the course of the disease43 are removed by the reticuloendothelial cells in the spleen, liver, lungs and associated lymph nodes154.
The rate of erythrocyte loss and the level of parasitaemia during the period of increasing anaemia prior to the detection of reticulocytes in the circulation are usually closely related. Evidence of a haemopoietic response in the peripheral circulation, which is a favourable prognostic sign, is typically first apparent just before the occurrence of maximum anaemia69.
Clinical signs
The prepatent or subclinical incubation period generally varies between 15 and 36 days (with an average of 26 days), although it may be longer. Disease manifestations can vary from from peracute disease leading to unexpected death to minimal disease.
Peracute anaplasmosis is the most severe form of the disease. It is rare but usually fatal. Animals may be found dead or succumb within a few hours of the onset of clinical signs. In addition to anaemia, milk production ceases, and there is excessive salivation, rapid respiration, irrational behaviour and signs of nervousness.
The most outstanding features of the acute bovine anaplasmosis are due to relatively rapid onset of anaemia and include pallor of mucous membranes, depression, inappetence, a decrease in milk production, general weakness, and a rapidly rising parasitaemia. In some animals, fever provides initial and persistent clinical evidence of anaplasmosis, but this is not consistent126, as in others the rectal temperature may remain below 40 °C26. As the disease progresses, constipation, rumen stasis, weight loss, dehydration, icterus, laboured respiration, a thumping heartbeat and muscle tremors are observed in severely affected animals. Diarrhoea may be manifested in the initial stages of the disease70. Abortions may occur in severe cases17, 34, 49, 127, 181. Abnormally aggressive behaviour, probably resulting from the anaemia and resultant cerebral hypoxia, is also manifested by some animals. The course of the disease may be protracted depending on the severity of anaemia and the capacity of the animal for erythropoiesis.
Once acute disease is controlled, most persistently infected animals are asymptomatic. However, chronic disease can occur and is manifested by slow recovery following acute disease. It may persist for a period of two weeks to several months144. It is characterized by poor appetite, loss of weight and varying degrees of dehydration, anaemia and icterus which are usually milder than in the acute disease.
Anaplasma marginale infections may induce temporary infertility in bulls70, anoestrus in heifers166, and abortions and neonatal anaplasmosis following foetal infection17, 119.
Pathology
The primary insult is extravascular haemolysis, which is the removal of A. marginale-infected RBCs by the reticuloendothelium system, particularly in the spleen, leading to anaemia. The rate at which the anaemia develops and the severity of anaemia will dictate the severity of disease and clinicopathological abnormalities. Initially, total and unconjugated bilirubin will be elevated if the capacity of the liver to take up and conjugate bilirubin is exceeded. If RBC destruction is rapid and severe, icterus may develop, which indicates a bilirubinaemia of >1.5 mg/dL. In some cases, particularly later in the course of infection, conjugated bilirubin can also be elevated suggesting impairment of the hepatocytes to excrete the conjugated bilirubin into the biliary system. Bilirubinuria may develop if sufficient amounts of conjugated bilirubin are released into circulation. Alkaline phosphatase (ALP), aspartate aminotransferase (AST) and serum urea nitrogen (SUN) can also be elevated, likely due to hypoxic injury to hepatocytes and the kidney8, 9, 10.
Gross and histopathologic lesions are primarily referrable to erythrophagocytosis and anaemia. On a post mortem examination, all tissues will be pale tan, indicating anaemia and icterus. The urine often has a brownish colour due to bilirubinuria. Splenomegaly is often marked. If hypoxia is severe and prolonged enough, the liver may be mottled due to centrilobular degeneration169.
There are few reports on the histopathology of anaplasmosis. Histologically there is evidence of haemosiderosis, erythrophagocytosis by the RE system and hyperplasia of erythropoietic elements in the bone marrow. Typically there is hyperplasia of the splenic red pulp, and an increase in the number of plasma cells and haemosiderin-bearing RE cells154, 175. Depending on severity, centribular hepatic necrosis and acute renal tubular necrosis, due to hypoxia may be apparent8, 68, 154. Bile stasis, and proliferation and haemosiderin pigmentation of Kupffer cells have also been reported. Haemosiderin may be present in macrophages of the lungs and lymph nodes175.
Diagnosis
It is important to understand the goal of the diagnostic testing for A. marginale to ensure selection of the appropriate test and accurate interpretation of results. In general, the primary purposes of diagnostic testing are to: 1) diagnosis of clinical cases of bovine anaplasmosis and; 2) to identify persistently infected animals. Robust laboratory-based diagnostic tests have been validated for both purposes. However, there remains a need for commercially available, low input or pen-side tests.
Ideally, a diagnosis of clinical bovine anaplasmosis requires documentation of anaemia in conjunction with the presence of infected erythrocytes. Infected erythrocytes can be readily visualized on a Diff-Quick or 10 per cent Giemsa- stained blood smear using light microscopy. If only post mortem samples are available, air-dried thin smears from the liver, kidney, heart or lungs or peripheral blood vessels stained with Diff-Quick or 10 per cent Giemsa may be diagnostic120. Regardless, great care should be taken to ensure clean slides are used and all particulates are filtered from the stain because debris can easily be mistaken for A. marginale infected erythrocytes. Smears should be visualized under oil immersion at 100x magnification. The A. marginale colonies are approximately 1-2 µm in diameter, deeply stained, uniformly round, and typically located at the periphery of the RBC. Anaplasma centrale forms morphologically similar colonies that tend to be located in the centre of RBCs.
At the height of the anaemic crisis, A. marginale inclusion bodies may be difficult to find because of the removal of the infected erythrocytes from the circulation. In addition, the presence of a regenerative anaemia requires careful examination of blood smears to distinguish A. marginale inclusion bodies from Howell-Jolly bodies. PCR can be used in the place of, or in addition to, blood smears to identify A. marginale infected animals. In the absence of quantitation, a positive result with PCR indicates the presence of infection, not necessarily disease. Thus, in the absence of visualization of A. marginale-infected erythrocytes it is important to rule out other causes of anaemia such as bovine theileriosis, babesiosis, trypanosomosis before definitively diagnosing a case of bovine anaplasmosis.
PCR is generally a reliable method to identify A. marginale – infected animals. There are a number of platforms, and published targets for PCR-based assays to detect the pathogen in ruminant blood samples. Msp5, a highly conserved, single copy gene is the most commonly used target. The increased sensitivity of real-time PCR and nested PCR as compared to standard PCR is useful for identifying animals with low levels of circulating pathogen. Based on the specificity of the target gene, PCR is also well-suited for differentiating between A. marginale and other Anaplasma spp. or haemoparasites. Regardless of the platform and target gene, care should be taken to ensure the test has been validated for its intended purpose.
Serology is the most reliable method to detect persistently infected animals, which may be PCR negative due to low numbers of circulating organisms. However, serology should not be relied upon to diagnose clinical disease because seroconversion requires approximately 14-21 days and thus acutely infected animals may not yet have developed an antibody response. Additionally, for this same reason, identification of uninfected animals requires repeat testing to ensure recently infected animals that have not yet seroconverted are classified appropriately.
Historically, a number of serologic tests have been used to detect anti-A. marginale antibodies, including complement fixation, rapid card agglutination, and indirect immunofluorescent assays. A commercially available competitive ELISA (cELISA) using recombinant Msp5 and the monoclonal antibody AnaF16C1 is widely used. When validated with a population of cattle from the US, it had 99.7 per cent diagnostic specificity and 100 per cent diagnostic sensitivity32, 38, 120. However, this population of animals was unlikely to be infected with other Anaplasma spp. or other haemoparasites. Serologic cross-reaction among the Anaplasma spp. and Ehrlicha spp. is common. This cELISA will also detect antibodies directed against A. centrale, A. ovis, and possibly Ehrlichia spp.7, 102, but does not detect antibodies from A. phagocytophilum infected dogs and humans164. Serum from A. phagocytophilum infected animals will bind recombinant A. marginale Msp5 in other ELISA formats, such as the indirect ELISA164. In short, validation of serologic tests for the detection of anti-A. marginale antibodies, if applied to non-cattle species, is necessary to ensure accurate results.
Apart from blood or tissue smears and determination of haematocrit to diagnose clinically affected animals, there remains a great need for commercially available, low input tests that can be readily used at the pen-side or in regions without access to diagnostic laboratories. Latex agglutination to detect anti-A. marginale antibodies using Msp5 as the antigen has been experimentally validated in cattle populations in Brazil137. As low input thermocyclers become more available, reverse line blot, though potentially less sensitive than other PCR-based methods, may be useful30.
Differential diagnosis
The clinical signs of anaplasmosis should be differentiated from infectious and non-infectious conditions presenting with fever, anaemia, icterus, depression, inappetance, and reduced milk production. Similarly, on post-mortem examination, anaplasmosis should be differentiated from conditions which cause anaemia primarily due to erythrophagocytosis (extra vascular haemolysis).
In regions were both bovine babesiosis (redwater) caused by Babesia bigemina or B. bovis and bovine anaplasmosis are endemic, including Africa, Australia, and South and Central America, anaplasmosis can be confused clinically and pathologically with bovine babesiosis. The epidemiology of the two diseases can be quite similar as both diseases are tick-borne, have the same tick vectors, establish persistent infection in cattle, and young animals tend to be less susceptibility to severe disease. One of the main clinical features of babesiosis, but not bovine anaplasmosis, is haemoglobinuria. However, haemoglobinuria, is also not a constant feature of babesiosis, particularly during the early stage of B. bovis infection. Although diarrhoea, which commonly occurs in babesiosis, may be present in acute anaplasmosis, constipation and rumen stasis are more regular findings. The course of the clinical disease in anaplasmosis is often longer than that of babesiosis. Care should be taken as outbreaks of anaplasmosis and babesiosis may occur concurrently.
Some forms of theileriosis should be differentiated from bovine anaplasmosis. Theileria are apicomplexan, blood borne, intracellular tick transmitted parasites. Theileria orientalis, previously named T. buffeli in Australia, T. sargenti in Japan and East Asia, and T. orientalis in many other locations are now all classified as the T. orientalis complex and are separated into several genotypes54. Most genotypes are non-pathogenic, however, genotype Ikeda and Chitose do cause disease. Since 2010, the Ikeda genotype has been responsible for a number of outbreaks of clinical disease in Australia, New Zealand, and more recently, the United States45, 94, 118. Disease is due to the destruction of erythrocytes, and thus the clinical presentations are nearly identical to bovine anaplasmosis. In severe cases, intravascular haemolysis can occur leading to haemoglobinuria, which is not a clinical feature of bovine anaplasmosis. Detection of the pathogen is required to differentiate between these diseases in regions where they occur together.
Theileria parva and T. annulata are transforming Theileria sp. because they cause lymphoproliferation and subsequent lymphadenomegaly. All aspects of the diseases caused by T.parva and T annulata are described in detail in the respective chapters(see-East coast fever and Theileria annulata theileriosis)Disease due to these two pathogens is predominantly due to the effects of lymphoproliferation and clinical signs typically include fever, weight loss, lymphadenopathy and often death136. Animals infected with T. annulata can also develop progressive anaemia and icterus due to significant replication of the parasite in erythrocytes and thus may need to be differentiated from bovine anaplasmosis114. Additional clinical features of T. annulata which are not seen in bovine anaplasmosis include panleukopenia and marked thrombocytopenia. The later can lead to petechiae on the mucous membranes and serosal surfaces of many organs.
Anaemia and progressive weight loss are also clinical signs often associated with Trypanosoma spp. infections. Bovine trypanosomosis (nagana), however, generally occurs within well-defined tsetse-infested areas.
Anaemia and icterus may also be encountered in cattle suffering from leptospirosis, bacilliary haemoglobinuria72, chronic copper poisoning and intoxications with Brassica and Allium spp71, 72. In these conditions, haemoglobinaemia and haemoglobinuria are usually also present72.
Control
Control strategies vary greatly in different countries and geographic regions. Generally, the effectiveness of a strategy in a specific region depends on a thorough knowledge of the epidemiology of the disease, but it is frequently the case that the scientifically-based measures are constrained by such factors as legislation, available infrastructure, funding, and the level of management. For these reasons no universal control programme can be recommended.
Following the introduction of anaplasmosis, whether by infected ticks, cattle or other reservoir hosts, into regions free of the disease and where an epidemic amongst the susceptible resident cattle populations could be expected, eradication of the disease is indicated. This is achieved by identifying infected cattle by PCR, microscopic examination of blood smears, and serology. Chemosterilization, the use of drugs to clear infected animals of the pathogen, require long-term doses of and require follow-up testing to ensure success in each animal, and thus may be difficult to achieve on a herd level (see section on chemosterilization). Concurrent stringent vector control measures to ensure eradication of infected ticks and to prevent mechanical spread of the infection must be implemented. In order to eradicate the disease successfully, a sophisticated veterinary infrastructure, the support of relevant legislation and the availability of ample funding are essential. Regulatory measures should also be implemented to prevent the subsequent reintroduction of the disease.
In southern Africa, where anaplasmosis is endemic in many of the important cattle farming areas, its eradication followed by the implementation and long-term maintenance of strict tick control programmes is not generally recommended, because stringent tick control could render the cattle population susceptible to several other tick-borne diseases including babesiosis and heartwater with potentially catastrophic consequences (see Vectors: Ticks), as has been experienced in Zimbabwe93. Additionally, there are many financial and practical limitations regarding anaplasmosis eradication programmes. The development of acaricide resistance by ticks is common and the cost involved in high-frequency dipping programmes is too expensive for many farmers. Further complicating factors are that populations of wild ruminants sustain tick populations and some are reservoir hosts.
For these reasons, in endemic regions with high transmission rates, it is often advantageous to manage for a stable disease situation by allowing natural exposure of calves to tick-borne diseases, including anaplasmosis, during the period when they are naturally resistant to disease or protected by passively acquired maternal antibody. As far as anaplasmosis is concerned, the existence of epidemiological factors which inhibit the exposure of calves to infected ticks within the relevant period can be compensated for by the administration of the A. centrale live-blood vaccine. If this control strategy is followed, natural or artificial fluctuations in tick populations, or any other factor which may lead to episodic or low levels of pathogen transmission, are less likely to result in disease outbreaks.
Immunization
Anaplasma centrale, first identified in South Africa by Arnold Theiler, infects bovine erythrocytes, but is typically asymptomatic or causes milder disease than A. marginale. Anaplasma centrale circulates in South. Africa in non-vaccinated animals and is likely maintained in wildlife65, 75. Since its identification in 1912, intentional infection of animals with A. centrale is used as a means of vaccination to protect animals against bovine anaplasmosis150, 172, 183. This live vaccine has been used extensively in Israel, Australia, southern Africa, and many parts of South America. However, A. centrale, although generally a mild pathogen, can cause severe clinical disease following vaccination16, 83, 133, 183.
Use of A. centrale as a vaccine requires amplification of the organisms in splenectomized cattle and cryopreservation of the infected erythrocytes. This procedure results in a number of difficulties including ensuring safety, viability and quality control. Additionally, it requires maintenance of the cold chain and carries the risk of the spread of additional blood-borne pathogens. Consequently, it is not practical in many circumstances and cannot be licensed for use in some parts of the world.
The surface proteins of A. centrale share moderate to high identity with the surface proteins of A. marginale. Thus, it is likely that infection with A. centrale induces an immune response, including antibodies, that recognize A. marginale6. Anaplasma centrale generally provides protection against disease, but not infection. The degree of protection against disease is variable and likely depends on the challenge conditions and circulating strains of A. marginale. The highest recorded protection was in Kenya and Australia where parasitaemia upon challenge was 0.7 to 0.5 per cent in vaccinated animals and >7 to 10 per cent in nonvaccinated animals. In contrast, in studies conducted in Zimbabwe and South Africa, parasitaemia of 16 to 21 per cent developed in vaccinated animals following challenge, while, unvaccinated animals had parasitaemias of approximately 30 per cent. Intermediate, levels of protection have been recorded in Argentina and Paraguay with 2.3 to 5.7 per cent parasitaemia in vaccinated animals and 7.5 to 16 per cent parasitaemia in unvaccinated animals following challenge19.
The use of A. centrale as a vaccine requires careful management and administration. As young calves tend not to develop severe disease, it is generally advised that they are vaccinated with A. centrale before or at approximately six months of age. In older animals more, severe disease can be expected and close supervision is indicated. Pregnant cows should not be vaccinated as they may abort. The incubation period following vaccination usually varies from five to seven weeks. Vaccinated animals should be closely observed for clinical signs of anaplasmosis during this period, and should, if necessary, be specifically and symptomatically treated. In view of the possible reaction to vaccination in older animals, only a limited number of animals should be immunized at any given time.
The use of inactivated A. marginale vaccines prevents serious clinical disease20. However, little data exists concerning the efficacy of inactivated products. Repeated administration of products with residual proteins from bovine erythrocytes can result in fatal neonatal isoerythrolysis in calves born to vaccinated cows41. Additionally, cattle vaccinated with inactivated organisms are still highly susceptible to challenge with heterologous strains92.
Improved methods to culture A. marginale could result in a safer, easier to produce live vaccine. Currently, A. marginale can be readily cultured in tick cell lines, but not bovine erythrocytes111. However, protective immunity in cattle immunized with whole, killed A. marginale derived from cell culture is overall poor. In one study, protection was better with A. marginale derived from erythrocytes as compared to tick cells, though there were no statistically significant differences between any of the groups39. In a second trial, A. marginale derived from tick cell culture provided protection from anaemia though not parasitaemia79. There was no comparison of the protection provided with A. marginale from RBC in this study. While the major surface proteins of A. marginale are expressed during growth in cell culture, many other less immunogenic outer membrane proteins are greatly down-regulated in tick cells13, 116. Thus the antigenic profile of A. marginale derived from tick cells is likely inadequate to provide protective immunity.
A recombinant vaccine offers a safe, efficacious, and low-cost method to protect cattle against bovine anaplasmosis. Purified outer membrane proteins of A. marginale consistently protect nearly all animals from high parasitaemia and disease, though protection from infection is inconsistent25, 115, 117. The current model for vaccine-induced immunity against A. marginale proposes that clearance of the organism is affected by antibody against surface epitopes in combination with macrophage activation for enhanced phagocytosis and killing. Central to this model is the CD4+ T-cell expression of interferon gamma (IFN-γ), which enhances synthesis of the predominant opsonizing bovine IgG subclass, IgG2, concomitantly activating macrophages to increase receptor expression, phagocytosis, phagolysosomal fusion and release of rickettsiacidal nitric oxide.
The protective outer membrane proteins is a complex mixture, with over 25 immunogenic proteins97. Within this mixture, a subset of outer membrane proteins have been identified as promising vaccine candidates115. A number of those candidates are highly conserved among widely geographically separated A. marginale strains. Thus, development of a single broadly protective vaccine, rather than a number of strain-specific vaccines, is possible51, 63.
Several obstacles in the development of a recombinant vaccine remain, including: 1) Identification of a specific set of outer membrane proteins that provide protective immunity to homologous and heterologous challenge; 2) Development of an expression system that maintains the immunogenicity of the proteins, which is not the case for proteins expressed in E. coli; 3) Identification of an effective adjuvant.
Treatment
The treatment of clinical anaplasmosis in cattle requires, first, the suppression of the parasite (specific treatment) and, second, the alleviation or prevention of secondary complications and the hastening of recovery (symptomatic or supportive treatment)68.
Secondary complications, generally the result of hypoxia, are a frequent sequelae to anaemia. During acute anaplasmosis the severity of this anaemia is proportional to the extent of the parasitaemia23, 68, 169. Specific treatment early in the course of infection while the PCV remains above 15 per cent is desirable24, 36, 48, 68, 89, 107, 108, 109, 145, 146, and such treatment prior to the development of a high parasitaemia or the onset of severe anaemia is considered essential for a favourable outcome89, 182. Often, however, by the time that acute anaplasmosis is diagnosed the level of parasitaemia is already declining as a result of the phagocytic removal of parasitized erythrocytes, and the animals are suffering from severe anaemia23. Nevertheless, even when treatment is slightly delayed, treatment with tetracyclines or imidocarb is more beneficial than supportive treatment108.
Specific treatment
Tetracyclines are the only effective chemotherapeutic agents approved for the treatment of anaplasmosis88, 89, 100, 145, 146. Of these, chlortetracycline, tetracycline and oxytetracycline are the most commonly used drugs for the treatment of acute and chronic carrier infections87, 88, 89, 150. Anaplasma marginale infections in cattle are not cleared at the usual recommended therapeutic doses of the tetracyclines90, 108, 161.
There is some controversy about the desirability of treating acute anaplasmosis at the time of maximum anaemia23. Much of this controversy is related to whether or not an animal will recover spontaneously, and the conflicting opinions which have been expressed range from those who consider treatment to be beneficial at this time to those who feel that the excitement and exertion incident to treatment are more likely to be detrimental to the animal. At the time when increased erythropoiesis becomes evident, specific treatment probably has little, if any, effect on the course of the disease, since at this stage the parasitaemia is rapidly decreasing as a result of the phagocytic removal of infected erythrocytes, and spontaneous recovery is frequent23.
Tetracycline is the preferred treatment for clinical bovine anaplasmosis. Tetracyclines are bacteriostatic, inhibit replication by reversible binding to the 30S ribosomal subunit, and have been widely used for the treatment of anaplasmosis21, 22, 23, 36, 48, 103, 108, 145, 146.
Short acting oxytetracycline at 10mg/kg/day until 24 to 48 hours after symptoms subside for no more than 4 days will alleviate symptoms, if treatment is not initiated too late. Alternatively, a single intramuscular administrations of long-acting (LA) formulations of oxytetracycline at 20 mg/kg body weight also effectively reduces disease severity90, 161, 173. The use of a single treatment of long-acting oxytetracycline for acute anaplasmosis has an obvious practical advantage in that cattle receiving multiple injections of short acting oxytetracycline, or any of the other tetracycline drugs, need to be restrained repeatedly.
Imidocarb [3,3’bis-(2-imidazolin- 2-yl)-carbanilide dihydrochloride (or dipropionate)] has specific chemotherapeutic effects on A. marginale infections, and is approved for commercial sale in some countries14, 24, 40, 84, 85, 86, 87, 91, 105, 106, 148. This drug is primarily used as an antibabesial3, 4, 89, 158, 174. At normally recommended therapeutic dosage levels imidocarb does not clear A. marginale infections in cattle3, 4, 24, 40, 85, 87, 105.
Imidocarb hydrochloride59 and its dipropionate salt105both have a marked inhibitory effect on A. marginale. Therapeutic dosage levels of imidocarb for the treatment of acute anaplasmosis in cattle vary from 2 to 5 mg/kg body weight150 administered subcutaneously or intramuscularly87, 88. A single subcutaneous dose of imidocarb at 2,5 mg/kg body weight is as effective as higher rates varying from 5 to 10 mg/kg in the control of acute anaplasmosis,148 and that a dosage rate of 2,5 mg/kg is the most efficacious under field conditions3, 106.
Imidocarb is carcinogenic, has a long withdrawal time and is not approved for use in the USA or Europe. Administration of imidocarb at recommended therapeutic dosage rates usually causes transient side-effects3, 4, 106, 148. These include a localized swelling at the injection site (following subcutaneous administration) and systemic effects, characterized by increased salivation, lachrymation and nasal discharge3, 104, 148. The severity of these signs is dose-related, and when doses are repeated or increased to eliminate infections (see Control: Chemosterilization), more severe clinical signs of toxicity including diarrhoea, dyspnoea and depression frequently occur2. Since intravenous inoculation similarly results in more severe signs of toxicity87, 88, 106, 174 and even mortality at rates of 3 mg/kg or higher87, 174, this route of administration is not recommended.
Following successful treatment of a primary A. marginale infection with any of the above-mentioned drugs, the parasitaemia decreases to 1 per cent or lower within four to seven days148, 182, but there are frequent relapses (recrudescences of parasitaemia) of varying intensity at variable intervals thereafter3, 24, 40, 87, 106, 161. Because treatment does not clear the bacteria, cyclical rises in parasitemia, as occurs in untreated animals is likely. Typically, these increases in parasitaemia are well controlled by the immune system and animals do not develop clinical disease.
Supportive treatment
Following treatment with an effective chemotherapeutic compound, parasitaemia declines over a period of several days, reaching levels of 1 per cent or lower only after approximately one week. The decline in parasitaemia is usually accompanied by a reduction in packed cell volume over a similar period of time. It is therefore important to bear in mind that the clinical condition of an animal may deteriorate considerably as a result of progressive anaemia notwithstanding successful specific chemotherapy, and that supportive symptomatic treatment may be required to prevent mortality23.
Because of the risk of mechanical transmission of A. marginale, affected cattle (especially those with high parasitaemias) are a major source of infection to uninfected susceptible cattle in the herd and should preferably be separated from the healthy animals. When isolating diseased animals, consideration must be given to the provision of adequate protection against the elements, easy access to palatable feed and water, as well as facilities for continuing observation of the animals and for the administration of any additional chemotherapy, which would require the minimum amount of exertion on the part of the animals61.
Additional symptomatic treatment may be beneficial and is usually recommended. Furthermore, correction of any gastrointestinal disturbances such as ruminal stasis, constipation or lack of appetite by the oral or parenteral administration of rumenotorics, ruminal fluid, mild laxatives and appetite stimulants, may help prevent additional secondary complications and promote a more rapid recovery.
Control of vaccine reactions
Following the administration of live-blood vaccines, some animals may develop clinical anaplasmosis. For all practical purposes these cases should be appropriately treated specifically and symptomatically as though they were naturally acquired clinical infections.
Chemoprophylaxis
The chemoprophylaxis of anaplasmosis is largely based on the constant or repeated administration of tetracyclines to control A. marginale parasitaemias in naturally infected cattle. This practice, in the absence of clinical disease in a herd, is strongly discouraged and in some countries not allowed, as the judicious use of antibiotics is required to ameliorate the growing problem of antibiotic resistant bacterial populations in general.
Generally, chemotherapy during the prepatent period merely results in the prolongation of the prepatent period and delays the onset of clinical signs46, 48, 95. With repeated administrations of long-acting oxytetracycline (at a rate of 20 mg/kg at seven-day intervals), commencing seven days after infection, the prepatent period is extended by approximately the same time-span as the duration of treatment46, 95. Although all animals thus treated remain infected, fewer animals develop clinical signs of anaplasmosis as the number of treatments increases.
For animals exposed to natural infection, a treatment regimen consisting of intramuscular administrations of short-acting oxytetracycline (at a rate of 6,6 to 11 mg/kg or long-acting (LA), at a rate of 20 mg/kg at 21- to 28-day intervals) to all susceptible cattle throughout the season when vectors are most active and abundant, and continued for a further one or two months after its end, have been employed68, 88, 96, 140. This treatment regimen should prevent clinical disease in most animals that become infected, but will not eliminate A. marginale from carrier animals.
When tetracyclines have been used to prevent clinical anaplasmosis, animals should continue to be closely observed for clinical signs of the disease after withdrawal of medication.
Chemosterilization
Anaplasma infections in cattle can be cleared, though typically long treatment regimens are required. The best method to determine if the pathogen was eliminated is via subinoculation of blood into a splenectomized animal. Additionally, a loss of the anti-Msp5 antibody response, as determined through cELISA can serve as a proxy. Real-time PCR should not be relied upon to confirm elimination of the pathogen.
Treatment to eliminate A. marginale infections in endemic areas is contraindicated because of the high risk of reinfection and the relative susceptibility of the cattle population in the absence of any long-term immunity which accompanies low-level persistent infections88.
Sensitive serological tests such as the cELISA can be used to identify persistently infected cattle in a herd. Segregation or removal of these animals from the rest of the herd, or elimination of infection in these animals, if removal from the herd is undesirable, can be used to establish an A. marginale-free herd. This approach has successfully been used in parts of the USA and Canada, but is usually only feasible in areas with a low or moderate prevalence of anaplasmosis88, 145.
Because of the possibility of transplacental infection, calves should also be screened serologically, and positive cases treated or eliminated in order to prevent them from becoming a future source of infection to adult cattle that have been cleared of infection. It must, nevertheless, be borne in mind that the serum of uninfected calves born of infected dams may also react positively in serological tests for periods of up to six months after birth, due to the presence of colostral antibodies134.
Imidocarb has been used for chemosterilization, however there is considerable individual variation in tolerance to imidocarb87 and severe toxicity and even mortality may result at the dosages recommended for chemosterilization85, 163.
Oral administration of chlortetracycline for the elimination of A. marginale may have advantages over parenteral administration in that treatment on a herd basis in the form of feed premixes incorporating the drug is possible140, but the consistent elimination of carrier infections by this method is often difficult to attain because of differences in amounts of medicated feed or salt-mineral mixes consumed by individual animals87, 145, 146.
Chemosterilization can be achieved in persistently infected animals using chlortetracycline as top dressing provided twice daily for 80 days. Animals receiving 4.4mg/kg/day, 11 mg/kg/day, and 22 mg/kg/day were cleared of A. marginale between 46-49 days of treatment based on real-time PCR, depending on the dose. Importantly, clearance of the bacteria was confirmed by subinoculation into a splenectomized animal. The animals became seronegative approximately 110 days following a PCR negative results138.
Alternatively, a single injection of long acting oxytetracycline at 20mg/kg followed by 30 days of treatment of chlortetracycline at 4.4mg/kg/day as top dressing on feed also resulted in chemosterilization138. Following clearance of infection, animals can be re-infected, although these re-infected animals show resistance to clinical anaplasmosis for periods ranging from a few months up to 30 months following parasite elimination47, 99, 140, 141, 142, 149.
Additionally, programmes aimed at the elimination of the carrier state are best conducted during the period of lowest vector activity and should be accompanied by the implementation or maintenance of vector control measures88, 89.
References
- ABDELA, N., IBRAHIM, N. & BEGNA, F., 2018. Prevalence, risk factors and vectors identification of bovine anaplasmosis and babesiosis in and around Jimma town, Southwestern Ethiopia. Acta Tropica, 177, 9-18.
- ADAMS, L. G., CORRIER, D. E. & WILLIAMS, J. D., 1980. A study of the toxicity of imidocarb dipropionate in cattle. Research in Veterinary Science, 28, 172-177.
- ADAMS, L. G. & TODOROVIC, R. A., 1974. The chemotherapeutic efficacy of imidocarb dihydrochloride on concurrent bovine anaplasmosis and babesiosis. I. The effects of a single treatment. Tropical Animal Health and Production, 6, 71-78.
- ADAMS, L. G. & TODOROVIC, R. A., 1974. The chemotherapeutic efficacy of imidocarb dihydrochloride on concurrent bovine anaplasmosis and babesiosis. II. The effects of multiple treatments. Tropical Animal Health and Production, 6, 79-84.
- ADJOU MOUMOUNI, P. F., APLOGAN, G. L., KATAHIRA, H., GAO, Y., GUO, H., EFSTRATIOU, A., JIRAPATTHARASATE, C., WANG, G., LIU, M., RINGO, A. E., UMEMIYA-SHIRAFUJI, R., SUZUKI, H. & XUAN, X., 2018. Prevalence, risk factors, and genetic diversity of veterinary important tick-borne pathogens in cattle from Rhipicephalus microplus-invaded and non-invaded areas of Benin. Ticks and Tick-borne Diseases, 9, 450-464.
- AGNES, J. T., BRAYTON, K. A., LAFOLLETT, M., NORIMINE, J., BROWN, W. C. & PALMER, G. H., 2011. Identification of Anaplasma marginale outer membrane protein antigens conserved between A. marginale sensu stricto strains and the live A. marginale subsp. centrale vaccine. Infection and Immunity, 79, 1311-1318.
- AL-ADHAMI, B., SCANDRETT, W. B., LOBANOV, V. A. & GAJADHAR, A. A., 2011. Serological cross-reactivity between Anaplasma marginale and an Ehrlichia species in naturally and experimentally infected cattle. Journal of Veterinary Diagnostic Investigation, 23, 1181-1188.
- ALLBRITTON, A. R. & SEGER, C. L., 1962. The transport and excretion of bile pigments in anaplasmosis. American Journal of Veterinary Research, 23, 1011-1018.
- ALLEN, P. C., KUTTLER, K. L. & AMERAULT, T. E., 1981. Clinical chemistry of anaplasmosis: Blood chemical changes in infected mature cows. American Journal of Veterinary Research, 42, 323-325.
- ALLEN, P. C., KUTTLER, K. L. & AMERAULT, T. E., 1981. Clinical chemistry of anaplasmosis: Comparative serum protein changes elicited by attenuated and virulent Anaplasma marginale isolates. American Journal of Veterinary Research, 42, 326-328.
- AUBRY, P. & GEALE, D., 2011. A review of bovine anaplasmosis. Transboundary and Emerging Diseases, 58, 1-30.
- BAKER, N. F., OSEBOLD, J. W. & CHRISTENSEN, J. F., 1961. Erythrocyte survival in experimental anaplasmosis. American Journal of Veterinary Research, 22, 590-596.
- BARBET, A. F., BLENTLINGER, R., YI, J., LUNDGREN, A. M., BLOUIN, E. F. & KOCAN, K. M., 1999. Comparison of surface proteins of Anaplasma marginale grown in tick cell culture, tick salivary glands, and cattle. Infection and Immunity, 67, 102-107.
- BARRETT, P. A., BEVERIDGE, E., BRADLEY, P. L., BROWN, C. G. D., BUSHBY, S. R. M., CLARKE, M. L., NEAL, R. A., SMITH, R. & WILDE, J. K. H., 1965. Biological activity of some a-dithiosemicarbazones. Nature, 206, 1340-1341.
- BECKLEY, C. S., SHABAN, S., PALMER, G. H., HUDAK, A. T., NOH, S. M. & FUTSE, J. E., 2016. Disaggregating tropical disease prevalence by climatic and vegetative zones within tropical west Africa. PLoS One, 11, e0152560.
- BIGALKE, R. D., 1980. Laboratory and field observations on the use of Anaplasma centrale as a vaccine against anaplasmosis. Zimbabwe Veterinary Journal, 11, 21-22.
- BIRD, J. E., 1973. Neonatal anaplasmosis in a calf. Journal of the South African Veterinary Association, 44, 69-70.
- BLOUIN, E. F. & KOCAN, K. M., 1998. Morphology and development of Anaplasma marginale (Rickettsiales: Anaplasmataceae) in cultured Ixodes scapularis (Acari: Ixodidae) cells. Journal of Medical Entomology, 35, 788-797.
- BOCK, R. E. & DE VOS, A. J., 2001. Immunity following use of Australian tick fever vaccine: a review of the evidence. Australian Veterinary Journal, 79, 832-839.
- BROCK, W. E., KLIEWER, I. O. & PEARSON, C. C., 1965. A vaccine for anaplasmosis. Journal of the American Veterinary Medical Association, 147, 948-951.
- BROCK, W. E., PEARSON, C. C. & KLIEWER, I. O., 1953. High-level aureomycin (chlortetracycline) dosage in anaplasmosis. American Journal of Veterinary Research, 14, 510-513.
- BROCK, W. E., PEARSON, C. C. & KLIEWER, I. O., 1955. An experiment in the treatment of acute anaplasmosis with tetracycline hydrochloride. North American Veterinarian, 36, 547-550.
- BROCK, W. E., PEARSON, C. C., KLIEWER, I. O. & JONES, E. W., 1959. The relation of treatment to hematological changes in anaplasmosis. In: Proceedings of the United States Livestock Sanitary Association, 15-18 December 1959. San Francisco, California.
- BROWN, C. G. D., WILDE, J. K. H. & BERGER, J., 1968. Chemotherapy of experimental Anaplasma marginale infections. Trials with an experimental dithiosemicarbazone (a-ethoxyethylglyoxal dithiosemicarbazone). British Veterinary Journal, 124, 325-334.
- BROWN, W. C., SHKAP, V., ZHU, D., MCGUIRE, T. C., TUO, W., MCELWAIN, T. F. & PALMER, G. H., 1998. CD4(+) T-lymphocyte and immunoglobulin G2 responses in calves immunized with Anaplasma marginale outer membranes and protected against homologous challenge. Infection and Immunity, 66, 5406-5413.
- CALLOW, L. L., 1984. Protozoal and rickettsial diseases. Animal Health in Australia, 5. Canberra: Australian Government Publishing Service.
- CARREÑO, A. D., ALLEMAN, A. R., BARBET, A. F., PALMER, G. H., NOH, S. M. & JOHNSON, C. M., 2007. In vivo endothelial cell infection by Anaplasma marginale. Veterinary Pathology, 44, 116-118.
- CARSON, C. A., SELLS, D. M. & RISTIC, M., 1977. Cell-mediated immune response to virulent and attenuated Anaplasma marginale administered to cattle in live and inactivated forms. American Journal of Veterinary Research, 38, 173-179.
- CARSON, C. A., SELLS, D. M. & RISTIC, M., 1977. Cell-mediated immunity related to challenge exposure of cattle inoculated with virulent and attenuated strains of Anaplasma marginale. American Journal of Veterinary Research, 38, 1167-1172.
- CHAISI, M. E., BAXTER, J. R., HOVE, P., CHOOPA, C. N., OOSTHUIZEN, M. C., BRAYTON, K. A., KHUMALO, Z. T. H., MUTSHEMBELE, A. M., MTSHALI, M. S. & COLLINS, N. E., 2017. Comparison of three nucleic acid-based tests for detecting Anaplasma marginale and Anaplasma centrale in cattle. Onderstepoort Journal of Veterinary Research, 84, e1-e9.
- CHÁVEZ, A. S. O., FELSHEIM, R. F., KURTTI, T. J., KU, P.-S., BRAYTON, K. A. & MUNDERLOH, U. G., 2012. Expression patterns of Anaplasma marginale Msp2 variants change in response to growth in cattle, and tick cells versus mammalian cells. PLoS One, 7, e36012.
- CHUNG, C., WILSON, C., BANDARANAYAKA-MUDIYANSELAGE, C.-B., KANG, E., ADAMS, D. S., KAPPMEYER, L. S., KNOWLES, D. P., MCELWAIN, T. F., EVERMANN, J. F., UETI, M. W., SCOLES, G. A., LEE, S. S. & MCGUIRE, T. C., 2014. Improved diagnostic performance of a commercial Anaplasma antibody competitive enzyme-linked immunosorbent assay using recombinant major surface protein 5-glutathione S-transferase fusion protein as antigen. Journal of Veterinary Diagnostic Investigation, 26, 61-71.
- COETZEE, J. F., SCHMIDT, P. L., O’CONNOR, A. M. & APLEY, M. D., 2010. Seroprevalence of Anaplasma marginale in 2 Iowa feedlots and its association with morbidity, mortality, production parameters, and carcass traits. Canadian Veterinary Journal, 51, 862-868.
- CORREA, W. M., CORREA, C. N. M. & GOTTSCHALK, A. F., 1978. Bovine abortion associated with Anaplasma marginale. Canadian Journal of Comparative Medicine, 42, 227-228.
- COSTA, V. M. M., RIBEIRO, M. F. B., DUARTE, A. L. L., MANGUEIRA, J. M., PESSOA, A. F. A., AZEVEDO, S. S., DE BARROS, A. T. M., RIET-CORREA, F. & LABRUNA, M. B., 2013. Seroprevalence and risk factors for cattle anaplasmosis, babesiosis, and trypanosomiasis in a Brazilian semiarid region. Brazilian Journal of Veterinary Parasitology, 22, 207-213.
- CRISTENSEN, J. F. & HARROLD, J. B., 1956. Inhibition of Anaplasma marginale infection in cattle with oxytetracycline hydrochloride. In: Proceedings of the 60th Annual Meeting of the United States Animal Health Association. 28-30 November 1956. Chicago, Illinois.
- DA SILVA, N. B., TAUS, N. S., JOHNSON, W. C., MIRA, A., SCHNITTGER, L., VALENTE, J. D. M., VIDOTTO, O., MASTERSON, H. E., VIEIRA, T. S. W. J., UETI, M. W. & VIEIRA, R. F. C., 2018. First report of Anaplasma marginale infection in goats, Brazil. PLoS One, 13, e0202140.
- DE ECHAIDE, S. T., KNOWLES, D. P., MCGUIRE, T. C., PALMER, G. H., SUAREZ, C. E. & MCELWAIN, T. F., 1988. Detection of cattle naturally infected with Anaplasma marginale in a region of endemicity by nested PCR and a competitive enzyme-linked immunosorbent assay using recombinant major surface protein 5. Journal of Clinical Microbiology, 36, 777-782.
- DE LA FUENTE, J., KOCAN, K. M., GARCIA-GARCIA, J. C., BLOUIN, E. F., CLAYPOOL, P. L. & SALIKI, J. T., 2002. Vaccination of cattle with Anaplasma marginale derived from tick cell culture and bovine erythrocytes followed by challenge-exposure with infected ticks. Veterinary Microbiology, 89, 239-251.
- DE VOS, A. J., BARROWMAN, P. R., COETZER, J. A. W. & KELLERMAN, T. S., 1978. Amicarbalide: A therapeutic agent for anaplasmosis. Onderstepoort Journal of Veterinary Research, 45, 203-208.
- DENNIS, R. A., O’HARA, P. J., YOUNG, M. F. & DORRIS, K. D., 1970. Neonatal immunohemolytic anemia and icterus of calves. Journal of the American Veterinary Medical Association, 165, 1861-1869.
- DIKMANS, G., 1950. Transmission of anaplasmosis. American Journal of Veterinary Research, 11, 5-16.
- DIMOPOULLOS, G. T. & BEDELL, D. M., 1962. Studies of bovine erythrocytes in anaplasmosis. III. Role of chemical and physical changes in erythrocytes in the mechanism of anaemia in splenectomized calves. American Journal of Veterinary Research, 23, 813-820.
- DUMLER, J. S., BARBET, A. F., BEKKER, C. P., DASCH, G. A., PALMER, G. H., RAY, S. C., RIKIHISA, Y. & RURANGIRWA, F. R., 2001. Reorganization of genera in the families Rickettsiaceae and Anaplasmataceae in the order Rickettsiales: unification of some species of Ehrlichia with Anaplasma, Cowdria with Ehrlichia and Ehrlichia with Neorickettsia, descriptions of six new species combinations and designation of Ehrlichia equi and'HGE agent'as subjective synonyms of Ehrlichia phagocytophila. International Journal of Systematic and Evolutionary Microbiology, 51, 2145-2165.
- EAMENS, G. J., GONSALVES, J. R., JENKINS, C., COLLINS, D. & BAILEY, G., 2013. Theileria orientalis MPSP types in Australian cattle herds associated with outbreaks of clinical disease and their association with clinical pathology findings. Veterinary Parasitology, 191, 209-217.
- ECKBLAD, W. P., LINCOLN, S. D. & MAGONIGLE, R. A., 1979. Efficacy of terramycin/LA-200 administered during the prepatent period of anaplasmosis. In: Proceedings of the 83rd Annual Meeting of the United States Animal Health Association, 28 October-2 November 1979. San Diego, California.
- ECKBLAD, W. P. & MAGONIGLE, R. A., 1983. Acquired cellular responsiveness in cattle cleared of Anaplasma marginale 28 months later. Veterinary Immunology and Immunopathology, 4, 659-663.
- FOOTE, L. E., FARLEY, H. & GALLAGHER, B., 1951. The use of aureomycin in anaplasmosis. North American Veterinarian, 32, 547-549.
- FOWLER, D. & SWIFT, B. L., 1975. Abortion in cows inoculated with Anaplasma marginale. Theriogenology, 4, 59-67.
- FRANCIS, D. H., KINDEN, D. A. & BUENING, G. M., 1979. Characterization of the inclusion limiting membrane of Anaplasma marginale by immunoferriten labelling. American Journal of Veterinary Research, 40, 777-782.
- FUTSE, J. E., BUAMI, G., KAYANG, B. B., KOKU, R., PALMER, G. H., GRAÇA, T. & NOH, S. M., 2019. Sequence and immunologic conservation of Anaplasma marginale OmpA within strains from Ghana as compared to the predominant OmpA variant. PLoS One, 14, e0217661.
- FUTSE, J. E., UETI, M. W., KNOWLES, D. P. J. & PALMER, G. H., 2003. Transmission of Anaplasma marginale by Boophilus microplus: retention of vector competence in the absence of vector-pathogen interaction. Journal of Clinical Microbiology, 41, 3829-3834.
- GACHOHI, J. M., NGUMI, P. N., KITALA, P. M. & SKILTON, R. A., 2010. Estimating seroprevalence and variation to four tick-borne infections and determination of associated risk factors in cattle under traditional mixed farming system in Mbeere District, Kenya. Preventive Veterinary Medicine, 95, 208-223.
- GEBREKIDAN, H., PERERA, P. K., GHAFAR, A., ABBAS, T., GASSER, R. B. & JABBAR, A., 2020. An appraisal of oriental theileriosis and the Theileria orientalis complex, with an emphasis on diagnosis and genetic characterisation. Parasitology Research, 119, 11-22.
- GRAU, H. E. G., DA CUNHA FILHO, N. A., PAPPEN, F. G. & DA ROSA FARIAS, N. A., 2013. Transplacental transmission of Anaplasma marginale in beef cattle chronically infected in southern Brazil. Brazilian Journal of Veterinary Parasitology, 22, 189-193.
- GUEDES, D. S. J., ARAÚJO, F. R., SILVA, F. J. M., RANGEL, C. P., BARBOSA NETO, J. D. & FONSECA, A. H., 2008. Frequency of antibodies to Babesia bigemina, B. bovis, Anaplasma marginale, Trypanosoma vivax and Borrelia burgdorferi in cattle from the Northeastern region of the State of Pará, Brazil. Brazilian Journal of Veterinary Parasitology, 17, 105-109.
- GUILLEMI, E. C., DE LA FOURNIERE, S., OROZCO, M., PEÑA MARTINEZ, J., CORREA, E., FERNANDEZ, J., LOPEZ ARIAS, L., PAOLETTA, M., CORONA, B., PINARELLO, V., WILKOWSKY, S. E. & FARBER, M. D., 2016. Molecular identification of Anaplasma marginale in two autochthonous South American wild species revealed an identical new genotype and its phylogenetic relationship with those of bovines. Parasites and Vectors, 9, 305.
- HAIRGROVE, T., SCHROEDER, M. E., BUDKE, C. M., RODGERS, S., CHUNG, C., UETI, M. W. & BOUNPHENG, M. A., 2015. Molecular and serological in-herd prevalence of Anaplasma marginale infection in Texas cattle. Preventive Veterinary Medicine, 119, 1-9.
- HART, C. B., ROY-SMITH, F., BERGER, J., SIMPSON, R. M. & MCHARDY, N., 1971. Imidocarb for the control of babesiosis and anaplasmosis. In: Proceedings of the 19th World Veterinary Congress.
- HENDRIX, G. K., BRAYTON, K. A. & BURCHAM, G. N., 2019. Anaplasma ovis as the suspected cause of mortality in a neonatal elk calf. Journal of Veterinary Diagnostic Investigation, 31, 267-270.
- HENNING, M. W., 1956. Animal Diseases in South Africa. 3rd edn. Pretoria, South Africa: Central News Agency Ltd.
- HIDALGO, R. J., JONES, E. W., BROWN, J. E. & AINSWORTH, A. J., 1989. Anaplasma marginale in tick cell culture. American Journal of Veterinary Research, 50, 2028-2032.
- HOVE, P., BRAYTON, K. A., LIEBENBERG, J., PRETORIUS, A., OOSTHUIZEN, M. C., NOH, S. M. & COLLINS, N. E., 2020. Anaplasma marginale outer membrane protein vaccine candidates are conserved in North American and South African strains. Ticks and Tick-Borne Diseases, 11, 101444.
- HOVE, P., CHAISI, M. E., BRAYTON, K. A., GANESAN, H., CATANESE, H. N., MTSHALI, M. S., MUTSHEMBELE, A. M., OOSTHUIZEN, M. C. & COLLINS, N. E., 2018a. Co-infections with multiple genotypes of Anaplasma marginale in cattle indicate pathogen diversity. Parasites and Vectors, 11, 5.
- HOVE, P., KHUMALO, Z. T. H., CHAISI, M. E., OOSTHUIZEN, M. C., BRAYTON, K. A. & COLLINS, N. E., 2018b. Detection and characterisation of Anaplasma marginale and A. centrale in South Africa. Veterinary Sciences, 5.
- HOWDEN, K. J., GEALE, D. W., PARÉ, J., GOLSTEYN-THOMAS, E. J. & GAJADHAR, A. A., 2010. An update on bovine anaplasmosis (Anaplasma marginale) in Canada. Canadian Veterinary Journal, 51, 837-840.
- IJSB., 1984. Validation of the publication of new names and new combinations previously effectively published outside the IJSB: List No. 15, 1984. International Journal of Systematic Bacteriology, 34, 355-357.
- JONES, E. W. & BROCK, W. E., 1966. Bovine anaplasmosis: Its diagnosis, treatment and control. Journal of the American Veterinary Medical Association, 119, 1624-1633.
- JONES, E. W., KLIEWER, I. O., NORMAN, B. B. & BROCK, W. E., 1968. Anaplasma marginale infection in young and aged cattle. American Journal of Veterinary Research, 29, 535-544.
- JONES, E. W. & NORMAN, B. B., 1962. Bovine anaplasmosis: The disease, its clinical diagnosis and prognosis. In: Proceedings of the Fourth National Anaplasmosis Conference, 26-27 April 1962. Reno, Nevada.
- KELLERMAN, T. S., COETZER, J. A. W. & NAUDÉ, T. W., 1988. Plant Poisonings and Mycotoxicoses of Livestock in Southern Africa. Cape Town: Oxford University Press Southern Africa.
- KELLY, W. R., 1974. Veterinary Clinical Diagnosis, 3rd edn. London: Baillière Tindall.
- KESSLER, R. H. & RISTIC, M., 1979. In vitro cultivation of Anaplasma marginale: Invasion of and development in non-infected erythrocytes. American Journal of Veterinary Research, 40, 1774-1776.
- KHUMALO, Z. T., BRAYTON, K. A., COLLINS, N. E., CHAISI, M. E., QUAN, M. & OOSTHUIZEN, M. C., 2018. Evidence confirming the phylogenetic position of Anaplasma centrale (ex Theiler 1911) Ristic and Kreier 1984. International Journal of Systematic and Evolutionary Microbiology, 68, 2682-2691.
- KHUMALO, Z. T. H., CATANESE, H. N., LIESCHING, N., HOVE, P., COLLINS, N. E., CHAISI, M. E., GEBREMEDHIN, A. H., OOSTHUIZEN, M. C. & BRAYTON, K. A., 2016. Characterization of Anaplasma marginale subsp. centrale strains by use of msp1aS genotyping reveals a wildlife reservoir. Journal of Clinical Microbiology, 54, 2503-2512.
- KOCAN, K. M., 1986. Development of Anaplasma marginale in ixodid ticks: Coordinated development of a rickettsial organism and its host tick. In: SAUER, J. & HAIR, J.A., (eds). Morphology, Physiology and Behavioural Ecology of Ticks. England: Ellis Horwood Ltd,
- KOCAN, K. M., DE LA FUENTE, J., BLOUIN, E. F. & GARCIA-GARCIA, J. C., 2004. Anaplasma marginale (Rickettsiales: Anaplasmataceae): Recent advances in defining host-pathogen adaptations of a tick-borne rickettsia. Parasitology, 129, S285-S300.
- KOCAN, K. M., HAIR, J. A. & EWING, S. A., 1980. Ultrastructure of Anaplasma marginale Theiler in Dermacentor andersoni Stiles and Dermacentor variabilis (Say). American Journal of Veterinary Research, 41, 1966-1976.
- KOCAN, K. M., HALBUR, T., BLOUIN, E. F., ONET, V., DE LA FUENTE, J., GARCIA-GARCIA, J. C. & SALIKI, J. T., 2001. Immunization of cattle with Anaplasma marginale derived from tick cell culture. Veterinary Parasitology, 102, 151-161.
- KOCAN, K. M., TEEL, K. D. & HAIR, J. A., 1980. Demonstration of Anaplasma marginale Theiler in ticks by tick transmission, animal inoculation and fluorescent antibody studies. American Journal of Veterinary Research, 141, 183-186.
- KOCAN, K. M., WICKWIRE, K. B., EWING, S. A., HAIR, J. A. & BARRON, S. J., 1988. Preliminary studies of the development of Anaplasma marginale in salivary glands of adult feeding Dermacentor andersoni ticks. American Journal of Veterinary Research, 49, 1010-1013.
- KUTTLER, K. L., 1966. Clinical and hematological comparison of Anaplasma marginale and Anaplasma centrale. American Journal of Veterinary Research, 27, 941-946.
- KUTTLER, K. L., 1967. A study of the immunological relationship of Anaplasma marginale and Anaplasma centrale. Research in Veterinary Science, 8, 467-471.
- KUTTLER, K. L., 1971. Efficacy of oxytetracycline and a dithiosemicarbazone in the treatment of bovine anaplasmosis. American Journal of Veterinary Research, 32, 1349-1352.
- KUTTLER, K. L., 1971. Promising therapeutic agents for the elimination of Anaplasma marginale in the carrier animal. In: Proceedings of the 75th Annual Meeting of the United States Animal Health Association, 24-29 October 1971. Oklahoma City, Oklahoma.
- KUTTLER, K. L., 1972. Combined treatment with a dithiosemicarbazone and oxytetracycline to eliminate Anaplasma marginale infections in splenectomized calves. Research in Veterinary Science, 13, 536-539.
- KUTTLER, K. L., 1973. Review: Current status of control and treatment with drugs. In: Proceedings of the Sixth National Anaplasmosis Conference, 19-20 March 1973. Las Vegas, Nevada.
- KUTTLER, K. L., 1979. Current anaplasmosis control techniques in the United States. Journal of the South African Veterinary Association, 50, 314-320.
- KUTTLER, K. L., 1980. Pharmacotherapeutics of drugs used in treatment of anaplasmosis and babesiosis. Journal of the American Veterinary Medical Association, 176, 1103-1108.
- KUTTLER, K. L. & SIMPSON, J. E., 1978. Relative efficacy of two oxytetracycline formulations and doxycycline in the treatment of acute anaplasmosis in splenectomized calves. American Journal of Veterinary Research, 39, 347-349.
- KUTTLER, K. L. & TODOROVIC, R. A., 1973. Techniques of premunization for the control of anaplasmosis. In: Proceedings of the Sixth National Anaplasmosis Conference, 19-20 March 1973. Las Vegas, Nevada.
- KUTTLER, K. L., ZAUGG, J. L. & JOHNSON, L. W., 1984. Serologic and clinical responses of pre-immunized, vaccinated, and previously infected cattle to challenge exposure by two different Anaplasma isolates. American Journal of Veterinary Research, 45, 2223-2226.
- LAWRENCE, J. A., FOGGIN, C. M. & NORVAL, R. A. I., 1980. The effect of war on the control of diseases of livestock in Rhodesia (Zimbabwe). Veterinary Record, 107, 82-85.
- LAWRENCE, K., MCFADDEN, A., GIAS, E., PULFORD, D. J. & POMROY, W. E., 2016. Epidemiology of the epidemic of bovine anaemia associated with Theileria orientalis (Ikeda) between August 2012 and March 2014. New Zealand Veterinary Journal, 64, 38-47.
- LINCOLN, S. D., ECKBLAD, W. P. & MAGONIGLE, R. A., 1982. Bovine anaplasmosis: Clinical, hematologic, and serologic manifestations in cows given a long-acting oxytetracycline formulation in the prepatent period. American Journal of Veterinary Research, 43, 1360-1362.
- LINCOLN, S. D. & MAGONIGLE, R. A., 1981. The prophylactic efficacy of long-acting oxytetracycline (Terramycin/LA-200) in experimental bovine anaplasmosis. In: Proceedings of the Seventh National Anaplasmosis Conference, 21-23 October 1981. Starkville, Mississippi.
- LOPEZ, J. E., SIEMS, W. F., PALMER, G. H., BRAYTON, K. A., MCGUIRE, T. C., NORIMINE, J. & BROWN, W. C., 2005. Identification of novel antigenic proteins in a complex Anaplasma marginale outer membrane immunogen by mass spectrometry and genomic mapping. Infection and Immunity, 73, 8109-8118.
- MAAS, J., LINCOLN, S. D., COAN, M. E., KUTTLER, K. L., ZAUGG, J. L. & STILLER, D., 1986. Epidemiologic aspects of bovine anaplasmosis in semiarid range conditions of south central Idaho. American Journal of Veterinary Research, 47, 528-533.
- MAGONIGLE, R. A. & NEWBY, T. J., 1984. Response of cattle upon re-exposure to Anaplasma marginale after elimination of chronic carrier infections. American Journal of Veterinary Research, 45, 695-697.
- MAGONIGLE, R. A., RENSHAW, H. W., VAUGHN, H. W., STAUBER, E. H. & FRANK, F. W., 1975. Effect of five daily intravenous treatments with oxytetracycline hydrochloride on the carrier status of bovine anaplasmosis. Journal of the American Veterinary Medical Association, 167, 1080-1083.
- MARUFU, M. C., CHIMONYO, M., DZAMA, K. & MAPIYE, C., 2010. Seroprevalence of tick-borne diseases in communal cattle reared on sweet and sour rangelands in a semi-arid area of South Africa. Veterinary Journal, 184, 71-76.
- MASON, K. L., GONZALEZ, M. V., CHUNG, C., MOUSEL, M. R., WHITE, S. N., TAYLOR, J. B. & SCOLES, G. A., 2017. Validation of an improved Anaplasma antibody competitive ELISA for detection of Anaplasma ovis antibody in domestic sheep. Journal of Veterinary Diagnostic Investigation, 29, 763-766.
- MAZZOLA, V. & KUTTLER, K. L., 1981. The effect of minocycline on the acute and carrier phases of Anaplasma marginale in experimentally infected, splenectomized cattle. In: Proceedings of the Seventh National Anaplasmosis Conference, Missiiippi State University.
- MCHARDY, N., BERGER, J., TAYLOR, R. J., FAREBROTHER, D. & JAMES, J. A., 1980. Comparison of gloxazone, an effective but toxic anaplasmacide, with imidocarb hydrochloride. Research in Veterinary Science, 29, 198-202.
- MCHARDY, N. & SIMPSON, R. M., 1974. Imidocarb dipropionate therapy in Kenyan anaplasmosis and babesiosis. Tropical Animal Health and Production, 6, 63-70.
- MCHARDY, N. & SIMPSON, R. M., 1975. Repeated dosing in the treatment of Kenyan anaplasmosis, using imidocarb dihydrochloride. Tropical Animal Health and Production, 7, 139-148.
- MILLER, J. G., 1953. Indications and limitations of antibiotic therapy in anaplasmosis. em>. In: Proceedings of the 57th Annual Meeting of the United States Animal Health Association.
- MILLER, J. G., 1956. The prevention and treatment of anaplasmosis. Annals of the New York Academy of Science, 64, 49-55.
- MILLER, J. G., LEVY, H. E., TORBENT, B. J. & OGLESBY, W. T., 1953. The treatment of anaplasmosis in Louisiana with aureomycin and terramycin. Journal of the American Veterinary Medical Association, 122, 390-392.
- MTSHALI, M. S., DE LA FUENTE, J., RUYBAL, P., KOCAN, K. M., VICENTE, J., MBATI, P. A., SHKAP, V., BLOUIN, E. F., MOHALE, N. E., MOLOI, T. P., SPICKETT, A. M. & LATIF, A. A., 2007. Prevalence and genetic diversity of Anaplasma marginale strains in cattle in South Africa. Zoonoses and Public Health, 54, 23-30.
- MUNDERLOH, U. G., BLOUIN, E. F., KOCAN, K. M., GE, N. L., EDWARDS, W. L. & KURTTI, T. J., 1996. Establishment of the tick (Acari: Ixodidae)-borne cattle pathogen Anaplasma marginale (Rickettsiales: Anaplasmataceae) in tick cell culture. Journal of Medical Entomology, 33, 656-664.
- NEITZ, W. O., 1935. Bovine anaplasmosis: The transmission of Anaplasma marginale to a black-wildebeest (Connochaetes gnu). Onderstepoort Journal of Veterinary Science and Animal Industry, 5, 9-20.
- NEITZ, W. O. & ALEXANDER, R. A., 1945. Immunization of cattle against heartwater and the control of tick-borne diseases, redwater, gallsickness and heartwater. Onderstepoort Journal of Veterinary Science and Animal Industry, 20, 138-158.
- NENE, V., KIARA, H., LACASTA, A., PELLE, R., SVITEK, N. & STEINAA, L., 2016. The biology of Theileria parva and control of East Coast fever – Current status and future trends. Ticks and Tick-Borne Diseases, 7, 549–564.
- NOH, S. M., BRAYTON, K. A., BROWN, W. C., NORIMINE, J., MUNSKE, G. R., DAVITT, C. M. & PALMER, G. H., 2008. Composition of the surface proteome of Anaplasma marginale and its role in protective immunity induced by outer membrane immunization. Infection and Immunity, 76, 2219-2226.
- NOH, S. M., BRAYTON, K. A., KNOWLES, D. P., AGNES, J. T., DARK, M. J., BROWN, W. C., BASZLER, T. V. & PALMER, G. H., 2006. Differential expression and sequence conservation of the Anaplasma marginale msp2 gene superfamily outer membrane proteins. Infection and Immunity, 74, 3471-3479.
- NOH, S. M., TURSE, J. E., BROWN, W. C., NORIMINE, J. & PALMER, G. H., 2013. Linkage between Anaplasma marginale outer membrane proteins enhances immunogenicity but is not required for protection from challenge. Clinical and Vaccine Immunology, 20, 651-656.
- OAKES, V. J., YABSLEY, M. J., SCHWARTZ, D., LEROITH, T., BISSETT, C., BROADDUS, C., SCHLATER, J. L., TODD, S. M., BOES, K. M., BROOKHART, M. & LAHMERS, K. K., 2019. Theileria orientalis Ikeda genotype in Cattle, Virginia, USA. Emerging Infectious Diseases, 25, 1653-1659.
- OBERST, R. D., KOCAN, K. M., HAIR, J. A. & EWING, S. A., 1981. Staining characteristics of colonies of Anaplasma marginale Theiler in Dermacentor andersoni Stiles. American Journal of Veterinary Research, 42, 2006-2009.
- OIE., 2015. Bovine Anaplasmosis, in: OIE Terrestrial Manual, 999-1013.
- OKAFOR, C. C., COLLINS, S. L., DANIEL, J. A., COETZEE, J. F. & WHITLOCK, B. K., 2019a. Seroprevalence of bovine Anaplasmosis in Georgia. Veterinary Parasitology: Regional Studies and Reports, 15, 1-5.
- OKAFOR, C. C., COLLINS, S. L., DANIEL, J. A., COETZEE, J. F. & WHITLOCK, B. K., 2019b. Factors associated with seroprevalence of bovine anaplasmosis in Mississippi, USA. Veterinary Parasitology: Regional Studies and Reports, 17, 1-10.
- OKAFOR, C. C., COLLINS, S. L., DANIEL, J. A., HARVEY, B., COETZEE, J. F. & WHITLOCK, B. K., 2018a. Factors associated with Seroprevalence of bovine anaplasmosis in Texas. Veterinary Parasitology: Regional Studies and Reports, 14, 32-40.
- OKAFOR, C. C., COLLINS, S. L., DANIEL, J. A., HARVEY, B., SUN, X., COETZEE, J. F. & WHITLOCK, B. K., 2018b. Factors associated with Seroprevalence of Anaplasma marginale in Kentucky cattle. Veterinary Parasitology: Regional Studies and Reports, 13, 212-219.
- OSORNO, M. B., SOLANA, P. M., PEREZ, J. M. & LOPEZ, T. R., 1975. Study of an attenuated Anaplasma marginale vaccine in Mexico—natural challenge of immunity in an enzootic area. American Journal of Veterinary Research, 36, 631-633.
- PAIN, G. D. & MILLER, A. S., 1977. Anaplasmosis in a newborn calf. Veterinary Record, 100, 58.
- PALMER, G. H., BARBET, A. F., DAVIS, W. C. & MCGUIRE, T. C., 1986. Immunization with an isolate-common surface protein protects cattle against anaplasmosis. Science, 231, 1299-1302.
- PAUL, N. I., PARKER, R. J., WILSON, A. J. & CAMBELL, R. S. F., 1980. Epidemiology of bovine anaplasmosis in beef calves in northern Queensland. Australian Veterinary Journal, 56, 267-271.
- PICOLOTO, G., LIMA, R. F. D., OLEGÁRIO, L. A. O., CARVALHO, C. M. E., LACERDA, A. C. R., TOMÁS, W. M., BORGES, P. A. L., PELLEGRIN, A. O. & MADRUGA, C. R., 2010. Real time polymerase chain reaction to diagnose Anaplasma marginale in cattle and deer (Ozotoceros bezoarticus leucogaster) of the Brazilian Pantanal. Brazilian Journal of Veterinary Parasitology, 19, 186-188.
- PIPANO, E., KRIGEL, Y., FRANK, M., MARKOVICS, A. & MAYER, E., 1986. Frozen Anaplasma centrale vaccine against anaplasmosis in cattle. British Veterinary Journal, 142, 553-556.
- POTGIETER, F. T., 1979. Epizootiology and control of anaplasmosis in South Africa. Journal of the South African Veterinary Association, 50, 367-372.
- POTGIETER, F. T., 1981. Tick transmission of anaplasmosis in South Africa. In: Proceedings of the International Conference on Tick Biology and Control, 27-29 January 1981. Grahamstown.
- POTGIETER, F. T. & VAN RENSBURG, L. J., 1983. Infectivity, virulence and immunogenicity of Anaplasma centrale live blood vaccine. Onderstepoort Journal of Veterinary Research, 50, 29-31.
- POTGIETER, F. T. & VAN RENSBURG, L. J., 1987. The persistence of colostral Anaplasma antibodies and incidence of in utero transmission of Anaplasma infections in calves, under laboratory conditions. Onderstepoort Journal of Veterinary Research, 54, 557-560.
- POTGIETER, F. T. & VAN RENSBURG, L. J., 1987. Tick transmission of Anaplasma centrale. Onderstepoort Journal of Veterinary Research, 54, 5-7.
- PRESTON, P. M., BROWN, C. G., BELL-SAKYI, L., RICHARDSON, W. & SANDERSON, A., 1992. Tropical theileriosis in Bos taurus and Bos taurus cross Bos indicus calves: response to infection with graded doses of sporozoites of Theileria annulata. Research in Veterinary Science, 53, 230-243.
- RAMOS, C. A. N., ARAÚJO, F. R., SANTOS, R. C., MELO, E. S. P., SOUSA, L. C., VIDAL, C. E. S., GUERRA, N. R. & RAMOS, R. A. N., 2014. Development and assessment of a latex agglutination test based on recombinant MSP5 to detect antibodies against Anaplasma marginale in cattle. Brazilian Journal of Microbiology, 45, 199-204.
- REINBOLD, J. B., COETZEE, J. F., HOLLIS, L. C., NICKELL, J. S., RIEGEL, C., OLSON, K. C. & GANTA, R. R., 2010a. The efficacy of three chlortetracycline regimens in the treatment of persistent Anaplasma marginale infection. Veterinary Microbiology, 145, 69-75.
- REINBOLD, J. B., COETZEE, J. F., HOLLIS, L. C., NICKELL, J. S., RIEGEL, C. M., CHRISTOPHER, J. A. & GANTA, R. R., 2010b. Comparison of iatrogenic transmission of Anaplasma marginale in Holstein steers via needle and needle-free injection techniques. American Journal of Veterinary Research, 71, 1178-1188.
- RICHEY, E. J., 1981. Management of anaplasmosis in beef cattle. In: Proceedings of the Seventh National Anaplasmosis Conference, 21-23 October 1981. Starkville, Mississippi.
- RICHEY, E. J., BROCK, W. E., KLIEWER, I. O. & JONES, E. W., 1976. The effect of feeding low levels of chlortetracycline for extended periods on the carrier state of anaplasmosis. Bovine Practitioner, 11, 73-75.
- RICHEY, E. J., BROCK, W. E., KLIEWER, I. O. & JONES, E. W., 1977. Resistance to anaplasmosis after elimination of latent Anaplasma marginale infections. American Journal of Veterinary Research, 38, 169-170.
- RISTIC, M., 1968. Anaplasmosis, Infectious diseases of man and animals. Academic Press, New York, NY, USA.
- RISTIC, M., 1968. Anaplasmosis. In: WEINMAN, D. & RISTIC, M., (eds). Infectious Blood Diseases of Man and Animals, 2. New York, London: Academic Press.
- RISTIC, M., 1980. Anaplasmosis. In: AMSTUTZ, H. E., (ed.). Bovine Medicine and Surgery, 1. Santa Barbara: American Veterinary Publications.
- RISTIC, M., 1981. Anaplasmosis. In: RISTIC, M. & MCINTYRE, I., (eds). Diseases of Cattle in the Tropics. The Hague, Boston and London: Martinus Nijhoff.
- RISTIC, M. & WATRACH, A. M., 1963. Anaplasmosis. VI. Studies and a hypothesis concerning the cycle of development of the causative agent. American Journal of Veterinary Research,24, 267-277.
- ROBY, T. O., 1972. The inhibitory effect of imidocarb on experimental anaplasmosis in splenectomized calves. Research in Veterinary Science, 13, 519-522.
- ROBY, T. O., AMERAULT, T. E., MAZZOLA, V., ROSE, J. E. & ILEMOBADE, A., 1974. Immunity in bovine anaplasmosis after elimination of Anaplasma marginale infections with imidocarb. American Journal of Veterinary Research, 35, 993-995.
- ROGERS, R. J. & SHIELS, I. A., 1979. Epidemiology and control of anaplasmosis. Journal of the South African Veterinary Association, 50, 363-366.
- SCOLES, G. A., BROCE, A. B., LYSYK, T. J. & PALMER, G. H., 2005. Relative efficiency of biological transmission of Anaplasma marginale (Rickettsiales: Anaplasmataceae) by Dermacentor andersoni (Acari: Ixodidae) compared with mechanical transmission by Stomoxys calcitrans (Diptera: Muscidae). Journal of medical entomology, 42, 668-675.
- SCOLES, G. A., MCELWAIN, T. F., RURANGIRWA, F. R., KNOWLES, D. P. & LYSYK, T. J., 2006. A Canadian bison isolate of Anaplasma marginale (Rickettsiales: Anaplasmataceae) is not transmissible by Dermacentor andersoni (Acari: Ixodidae), whereas ticks from two Canadian D. andersoni populations are competent vectors of a U.S. Journal of Medical Entomology, 43, 971-975.
- SCOLES, G. A., MILLER, J. A. & FOIL, L. D., 2008. Comparison of the efficiency of biological transmission of Anaplasma marginale (Rickettsiales: Anaplasmataceae) by Dermacentor andersoni Stiles (Acari: Ixodidae) with mechanical transmission by the Horse Fly, Tabanus fuscicostatus Hine. Journal of Medical Entomology, 45, 109-114.
- SEGER, C. L. & WHITE, D., 1962. The histopathology of anaplasmosis. In: Proceedings of the Fourth National Anaplasmosis Conference, 26-27 April 1962. Reno, Nevada.
- SHARMA, S. P. & TRIPATHI, H. C., 1985. Observations on erythrocytic acetylcholinesterase in experimentally induced anaplasmosis in calves. Veterinary Parasitology, 17, 251-254.
- SHKAP, V., KOCAN, K., MOLAD, T., MAZUZ, M., LEIBOVICH, B., KRIGEL, Y., MICHOYTCHENKO, A., BLOUIN, E., DE LA FUENTE, J., SAMISH, M., MTSHALI, M., ZWEYGARTH, E., FLEIDEROVICH, E. L. & FISH, L., 2009. Experimental transmission of field Anaplasma marginale and the A. centrale vaccine strain by Hyalomma excavatum, Rhipicephalus sanguineus and Rhipicephalus (Boophilus) annulatus ticks. Veterinary Microbiology, 134, 254-260.
- SMITH, R. D., LEVY, M. G., KUHLENSCHMIDT, M. S., ADAMS, J. H., RZECHULA, D. L., HARDT, T. A. & KOCAN, K. M., 1986. Isolate of Anaplasma marginale not transmitted by ticks. American Journal of Veterinary Research, 47, 127-129.
- SMITH, T. & KILBOURNE, F. L., 1893. Investigations into the nature, causation and prevention of Texas or southern cattle fever. United States Department of Agriculture. Bureau of Animal Industry. Bulletin No. 1, 1-156. Washington: Government Printing Office.
- SPARE, M. R., HANZLICEK, G. A., WOOTTEN, K. L., ANDERSON, G. A., THOMSON, D. U., SANDERSON, M. W., GANTA, R. R., REIF, K. E. & RAGHAVAN, R. K., 2020. Bovine anaplasmosis herd prevalence and management practices as risk-factors associated with herd disease status. Veterinary Parasitology X, 3(1), 100021.
- STAATS, J. J., KOCAN, K. M., HAIR, J. A. & EWING, S. A., 1982. Immunocytochemical labelling of Anaplasma marginale Theiler in Dermacentor andersoni Stiles with peroxidase-antiperoxidase technique. American Journal of Veterinary Research, 43, 979-983.
- STEWART, C. G., IMMELMAN, A., GRIMBEEK, P. & GRIB, D., 1979. The use of a short and long acting oxytetracycline for the treatment of Anaplasma marginale in splenectomized calves. Journal of the South African Veterinary Association, 50, 83-85.
- STILLER, D., KOCAN, K. M., EDWARDS, W., EWING, S. A., HAIR, J. A. & BARRON, S. J., 1989. Detection of colonies of Anaplasma marginale in salivary glands of three Dermacentor spp. infected as nymphs or adults. American Journal of Veterinary Research, 50, 1381-1385.
- STOLTSZ, W. H., 1990. Onderstepoort Veterinary Institute. Unpublished data.
- STRIK, N. I., ALLEMAN, A. R., BARBET, A. F., SORENSON, H. L., WAMSLEY, H. L., GASCHEN, F. P., LUCKSCHANDER, N., WONG, S., CHU, F., FOLEY, J. E., BJOERSDORFF, A., STUEN, S. & KNOWLES, D. P., 2007. Characterization of Anaplasma phagocytophilum major surface protein 5 and the extent of its cross-reactivity with A. marginale. Clinical and Vaccine Immunology, 14, 262-268.
- SWAI, E. S., KARIMURIBO, E. D., OGDEN, N. H., FRENCH, N. P., FITZPATRICK, J. L., BRYANT, M. J. & KAMBARAGE, D. M., 2005. Seroprevalence estimation and risk factors for A. marginale on smallholder dairy farms in Tanzania. Tropical Animal Health and Production, 37, 599-610.
- SWIFT, B. L., MURDOCH, W. J. & DAHLGEN, R. R., 1984. Anemia associated with anestrus in beef heifers inoculated with Anaplasma marginale: Endocrinology and ovarian changes. Theriogenology, 22, 643-649.
- TEMBO, S., COLLINS, N. E., SIBEKO-MATJILA, K. P., TROSKIE, M., VORSTER, I., BYARUHANGA, C. & OOSTHUIZEN, M. C., 2018. Occurrence of tick-borne haemoparasites in cattle in the Mungwi District, Northern Province, Zambia. Ticks and Tick-borne Diseases, 9, 707-717.
- THEILER, A., 1910. Anaplasma marginale (Gen. and spec. nov.). A protozoan of cattle: A cause of the so-called gallsickness. Transvaal Medical Journal, 5, 110-111.
- THEILER, A., 1910. Anaplasma marginale (Gen. spec. Nova): The marginal points in the blood of cattle suffering from a specific disease. Report of the Government Veterinary Bacteriologist of the Transvaal, 1908–1909: 7-64.
- THEILER, A., 1910. ‘Anaplasma marginale’. A new genus and species of the Protozoa. Transactions of the Royal Society of South Africa, II, 69-72.
- THEILER, A., 1911. Further investigations into anaplasmosis of South African cattle. First Report of the Director of Veterinary Research, Union of South Africa, 7-46.
- THEILER, A., 1912. Gallsickness of imported cattle and the protective inoculation against this disease. Agricultural Journal of the Union of South Africa, 3, 1-2.
- TODOROVIC, R. A., GONZALEZ, E. F. & GARCIA, O., 1979. Evaluation of a new long-acting oxytetracycline formulation against anaplasmosis in Colombian cattle. Tropenmedizin und Parasitologie, 30, 236-238.
- TODOROVIC, R. A., VIZCAINO, O. G., GONZALEZ, E. F. & ADAMS, L. G., 1973. Chemoprophylaxis against Babesia bigemina and Babesia argentina infections. American Journal of Veterinary Research, 34, 1153-1161.
- TRUEMAN, K. F. & WILSON, A. J., 1979. Observation on the pathology of Anaplasma marginale infections in cattle. Australian Advances in Veterinary Science, 5, 75.
- TUCKER, T. R., ALY, S. S., MAAS, J., DAVY, J. S. & FOLEY, J. E., 2016. Investigation of Anaplasma marginale seroprevalence in a traditionally managed large California beef herd. Veterinary Medicine International, 6186078.
- UETI, M. W., REAGAN, J. O., KNOWLES, D. P., SCOLES, G. A., SHKAP, V. & PALMER, G. H., 2007. Identification of midgut and salivary glands as specific and distinct barriers to efficient tick-borne transmission of Anaplasma marginale. Infection and Immunity, 75, 2959-2964.
- UILENBERG, G., 1995. International collaborative research: significance of tick-borne hemoparasitic diseases to world animal health. Veterinary Parasitology, 57, 19-41.
- URDAZ-RODRÍGUEZ, J. H., FOSGATE, G. T., ALLEMAN, A. R., RAE, D. O., DONOVAN, G. A. & MELENDEZ, P., 2009. Seroprevalence estimation and management factors associated with high herd seropositivity for Anaplasma marginale in commercial dairy farms of Puerto Rico. Tropical Animal Health and Production, 41, 1439-1448.
- WALLACE, W. R., 1967. Loss of erythrocytic acetylcholinesterase activity and its relationship to osmotic fragility of erythrocytes in bovine anaplasmosis. American Journal of Veterinary Research, 28, 55-61.
- WANDERA, J. G. & MUNYUA, W. K., 1971. Severe anaplasmosis in a 4-day-old calf. Bulletin of Epizootic Diseases in Africa, 19, 219-211.
- WIESENHÜTTER, E., 1970. Experience with reverin in the treatment of anaplasmosis in dairy cattle. The Blue Book for the Veterinary Profession, 17, 9-12.
- WILSON, A. J., PARKER, R. & TRUEMAN, K. F., 1980. Experimental immunization of calves against Anaplasma marginale infection: Observation on the use of living A. centrale and A. marginale. Veterinary Parasitology, 7, 305-311.
- ZAUGG, J. L., 1985. Bovine anaplasmosis: transplacental transmission as it relates to stage of gestation. American Journal of Veterinary Research, 46, 570-572.