- Infectious Diseases of Livestock
- Part 1
- Heartwater
- Vectors: Ticks
- Vectors: Tsetse flies
- Vectors: Muscidae
- Vectors: Tabanidae
- Vectors: Culicoides spp.
- Vectors: Mosquitoes
- Classification, epidemiology and control of arthropod-borne viruses
- Special factors affecting the control of livestock diseases in sub-Saharan Africa
- The control of infectious diseases of livestock: Making appropriate decisions in different epidemiological and socioeconomic conditions
- Infectious diseases of animals in sub-Saharan Africa: The wildlife⁄livestock interface
- Vaccination: An approach to the control of infectious diseases
- African animal trypanosomoses
- Dourine
- Trichomonosis
- Amoebic infections
- GENERAL INTRODUCTION: COCCIDIA
- Coccidiosis
- Cryptosporidiosis
- Toxoplasmosis
- Besnoitiosis
- Sarcocystosis
- Balantidiosis
- Leishmaniosis
- Neosporosis
- Equine protozoal myeloencephalitis
- GENERAL INTRODUCTION: BABESIOSES
- Bovine babesiosis
- Equine piroplasmosis
- Porcine babesiosis
- Ovine babesiosis
- GENERAL INTRODUCTION: THEILERIOSES OF CATTLE
- East Coast fever
- Corridor disease
- Zimbabwe theileriosis
- Turning sickness
- Theileria taurotragi infection
- Theileria mutans infection
- Theileria annulata theileriosis
- Theileriosis of sheep and goats
- Theileria buffeli⁄orientalis infection
- Non-pathogenic Theileria species in cattle
- GENERAL INTRODUCTION: RICKETTSIAL, CHLAMYDIAL AND HAEMOTROPIC MYCOPLASMAL DISEASES
- Heartwater
- Lesser known rickettsial infections in animals and humans
- Chlamydiosis
- Q fever
- Eperythrozoonosis
- Bovine Haemobartonellosis
- Potomac horse fever
- GENERAL INTRODUCTION: ANAPLASMOSES
- Bovine anaplasmosis
- Ovine and caprine anaplasmosis
Heartwater
This content is distributed under the following licence: Attribution-NonCommercial CC BY-NC  View Creative Commons Licence details here
View Creative Commons Licence details here
NJ Maclachlan and M-L Penrith (Editors). BA Allsopp, M van Kleef and A Pretorius, Heartwater, 2018.
Heartwater
Previous Authors: B A ALLSOPP, J D BEZUIDENHOUT AND L PROZESKY
Current Authors:
B A ALLSOPP - Emeritus Professor, PhD, DIC, ARCS, 104 rue du Bosc 34980 St. Gély du Fesc, Hérault, 34980, France
M VAN KLEEF - Specialist Researcher, PhD, Agricultural Research Council-Onderstepoort Veterinary Research, 100 Old Soutpan Road, Onderstepoort, Pretoria, Gauteng, 0110, South Africa
A PRETORIUS - Senior Researcher, PhD, Agricultural Research Council, Onderstepoort Veterinary Research, 100 Old Soutpan Road, Onderstepoort, Pretoria, Gauteng, 0110, South Africa
Introduction
Heartwater (cowdriosis) is a tick-borne disease of cattle, sheep, goats and some wild ruminants that is caused by the rickettsia Ehrlichia ruminantium. Typically, the disease is characterized by high fever, nervous signs, hydropericardium, hydrothorax and oedema of the lungs and brain, and death. It is one of the major causes of stock losses in sub-Saharan Africa.
The first reference to what may have been heartwater was made by the Voortrekker pioneer Louis Trichardt in 1838.224 While trekking through what is today the Limpopo Province of South Africa many of his sheep succumbed to a disease known locally as ‘nintas’ three weeks after they had suffered massive tick infestation. According to evidence given by a farmer, John Webb, to the Cattle and Sheep Disease Commission of 1876 in Grahamstown, heartwater was observed in 1858 in South Africa in the northern part of the Eastern Cape Province. Because of its confusion with other local diseases of unknown aetiology that were prevalent at that time some of the earlier information regarding the occurrence of heartwater is unreliable.144
The first important experimental findings came in 1898 when both Dixon87 and Edington113 showed that heartwater disease could be induced by blood passage from infected to susceptible animals. No organisms could be demonstrated in the blood or other tissues of diseased animals but it was concluded that heartwater was caused by a living microorganism,149 at that time believed to be a virus.296 In 1900 Lounsbury published his confirmation of the long-standing suspicion that the bont tick (Amblyomma hebraeum) was the vector of heartwater in South Africa,177 but another quarter of a century elapsed before Cowdry demonstrated that the infectious agent in the tissues of infected animals and ticks was a rickettsia which he named Rickettsia ruminantium.76, 77 The name was later changed to Cowdria ruminantium210 and more recently to Ehrlichia ruminantium.111
Heartwater occurs wherever ticks capable of transmitting the organism are present (see Vectors: Ticks). The endemic area encompasses most of sub-Saharan Africa, including the islands of Madagascar, Sao Tomé, Réunion, Mauritius, and Zanzibar.125, 136, 137, 265 The disease is absent from the Kalahari Desert and dry coastal areas of Namibia and South Africa. Heartwater also occurs on the islands of Guadeloupe, Marie-Galante, and Antigua in the Caribbean63, 327 to which infected Amblyomma variegatum ticks were introduced, probably with cattle from Senegal, during the eighteenth century.29, 190
The occurrence of heartwater is frequently taken for granted in the endemic areas and definitive diagnoses are usually only conducted for particularly valuable animals. This leads to the prevalence rates of the disease being under-reported.62 In the endemic area in South Africa mortalities from heartwater are three times greater than those from babesiosis and anaplasmosis combined.226, 322 From a livestock census performed by the Directorate of Animal Health, South Africa, in 1996, it was estimated that 17.5 million head of livestock were at risk in the endemic area of the country. Goats are especially threatened, and in some parts of the rural farming sector it is believed that up to 30 per cent of goats become infected with heartwater annually. It is not known how many of these die from the infection, but a substantial proportion must do so.
The economic impact of heartwater is difficult to quantify, both because of the under-reporting noted above and because the actual occurrence of the disease may be partially suppressed by a range of factors. These include the use of acaricides (in 1996 acaricides costing US$ 13 million were purchased in South Africa), antibiotic prophylaxis,246 immunization by infection and treatment,339 the resistance of certain animal breeds to the disease,66 and endemic stability.312 An estimate of the impact of heartwater was made by the Deputy Director of Veterinary Services of the Eastern Cape Province of South Africa during 1998.23 His assessment was that 10 per cent of all stock losses, costing up to US$ 30 million annually, were due to heartwater, and this was despite an annual expenditure of between US$ 1 and 5 million for prophylaxis and vaccination. There are estimates from Zimbabwe of annual losses over a 10-year period amounting to US$ 5.6 million per annum, which includes the cost of acaricides, milk losses and treatment.213
Heartwater is a major obstacle to the introduction of high-producing animals into sub-Saharan Africa to upgrade local stock160, 320 and is of particular importance when susceptible animals are moved from heartwater-free to heartwater-infected areas.226, 293 Whatever the actual costs may be it is certain that the economic importance of heartwater as a disease of domestic ruminants in Africa is comparable to that of East Coast fever, trypanosomosis, rinderpest, and dermatophilosis.33, 265
The possibility of heartwater spreading from the infected Caribbean islands to the American mainland, where a suitable tick vector (Am. maculatum) and a potential reservoir host (Odocoileus virginianus) are both present, is a potential threat to the livestock industry.85, 186, 284, 321, 324 The disease is also a potential danger to countries where the vectors may be introduced and become established.28, 242, 352 It will be of major importance until an effective and safe vaccine becomes available.
Aetiology and life cycle
Classification
Cowdria ruminantium was traditionally classified as the sole species of the genus Cowdria, tribe Ehrlichieae, family Rickettsiaceae, order Rickettsiales,280 but it had long been realized that the organism had a close relationship with certain Ehrlichia spp.9 With the advent of molecular phylogenetic studies based on the small subunit (16S) ribosomalRNA (rRNA) gene it became evident that the genus Ehrlichia did not constitute a monophyletic group, and that organisms classified as Anaplasma, Cowdria, and Neorickettsia were members of three separate clades, each of which included various organisms then classified as Ehrlichia spp.263, 346 These developments led to a revision of the taxonomy of the order Rickettsiales in which the genera Anaplasma, Ehrlichia and Neorickettsia were moved to the family Anaplasmataceae, and Cowdria ruminantium was reclassified as Ehrlichia ruminantium.111 The question as to whether there should be more than one species of E. ruminantium is addressed below.
The type specimen of E. ruminantium comb. nov. is the Welgevonden strain.97 This was obtained from an Am. hebraeum tick collected in the same geographical area as the original R. ruminantium, which originated close to the Onderstepoort Veterinary Institute north of Pretoria, in Gauteng Province, South Africa.76 The pathology, morphology267 and virulence97 in cattle, sheep and goats appears to be the same as that of the original R. ruminantium.76 The full annotated genome sequence (NCBI Reference Sequence NC_005295.2) is deposited in GenBank.72 The Welgevonden 16S genotype is common in the field13 and frozen stocks of the strain, held at the Onderstepoort Veterinary Institute, date back to 1985.
Cellular characteristics
Species in the family Anaplasmataceae, including E. ruminantium, grow in an intravacuolar compartment, bounded by a lipid bilayer membrane, within the cytoplasm of infected host cells.111 This distinguishes them from species in the family Rickettsiaceae which grow freely in the cytoplasm of their eukaryotic host cells. Ehrlichia ruminantium is a Gram-negative bacterium, and stains purplish-blue with Giemsa.78 Individual cells, in common with other Gram-negative bacterial cells,36 are bounded by two membranes, an inner (plasma) membrane and an outer membrane. Several other staining methods have been used or developed to demonstrate the organism in its vertebrate and invertebrate hosts.56, 253, 267, 357 In paraffin-sections prepared from tick midgut and stained with Mallory's phloxine-methylene blue stain, Ehrlichia colonies and host cell nuclei stain dark blue against a uniformly pink background of tick tissue. This staining method is useful for identifying Ehrlichia-infected ticks and is superior to the variations of Mallory's stain which render the colours of colonies and tick tissues various shades of blue.357
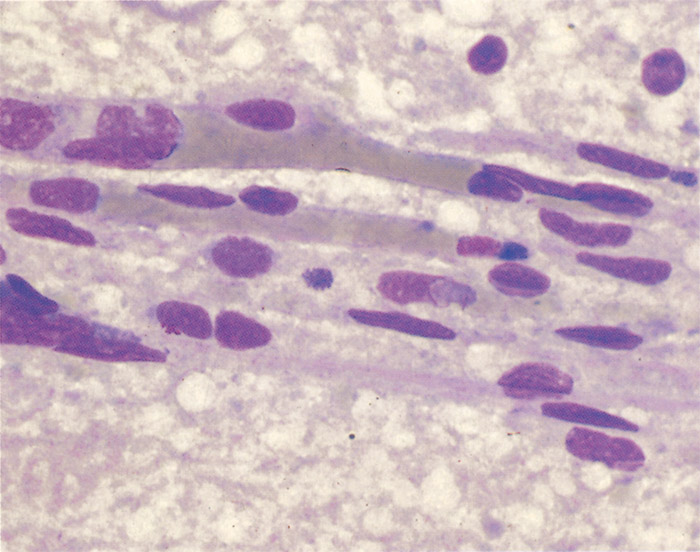
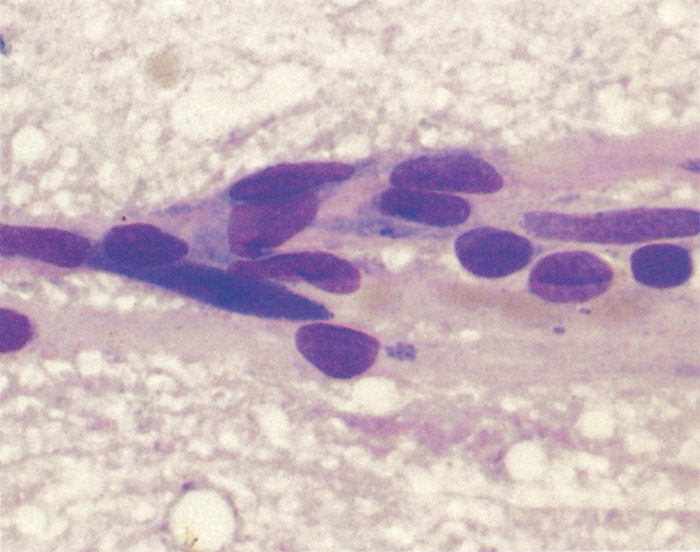
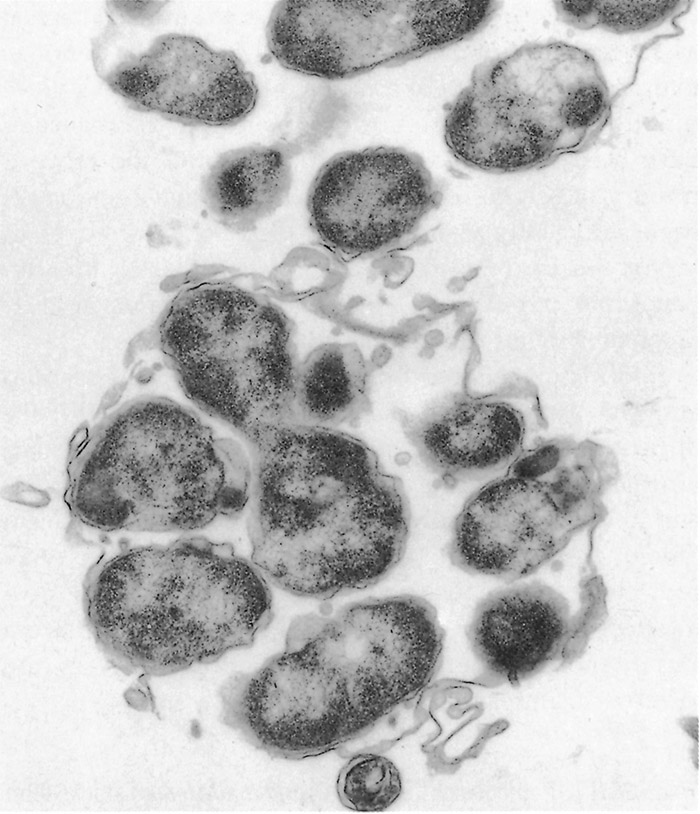
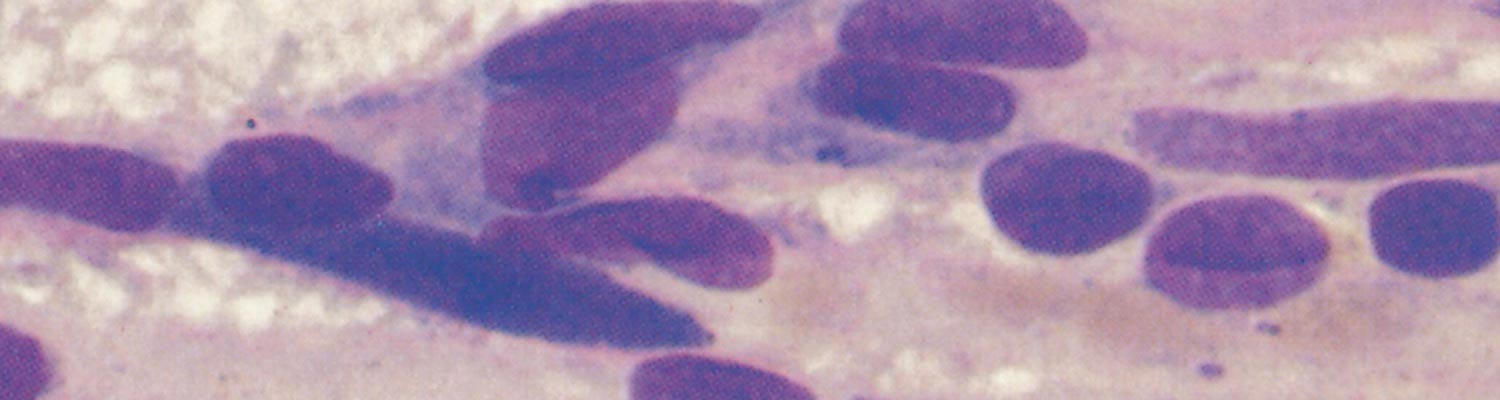
Ehrlichia ruminantium is a pleomorphic rickettsia, and colonies containing from one or two to several thousand individual organisms are found in the cytoplasm of endothelial cells (Figures 1 and 2).78, 253 In general the colonies consist predominantly of small (0.4 µm), medium (0.76 µm), large (1.04 µm) or very large (>1.04 µm) organisms253 but a number of smaller organisms are also found in colonies of larger organisms, and vice versa. Most organisms are coccoid, except for colonies containing very large organisms in which pleomorphic forms (horseshoe-, ring- and bacillary- shaped) may be seen.253
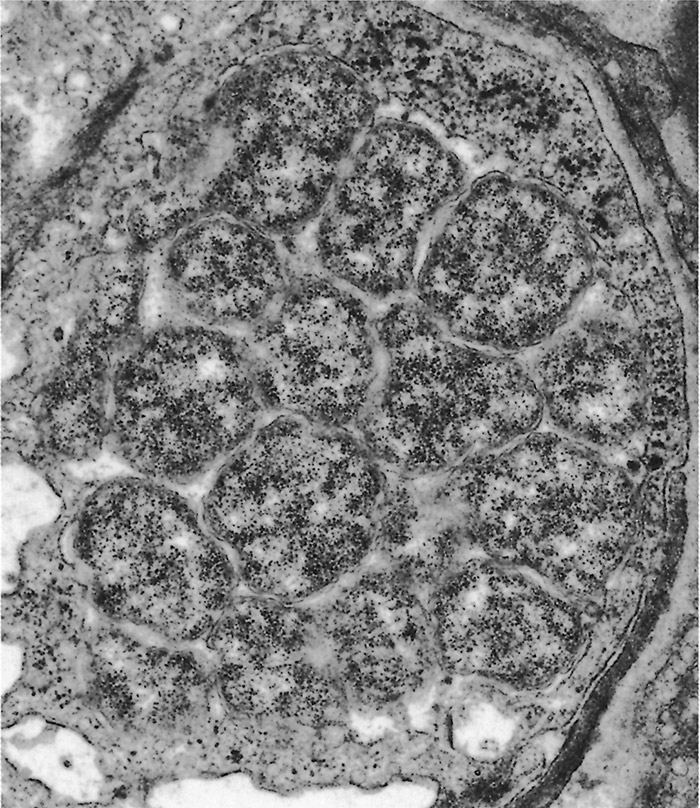
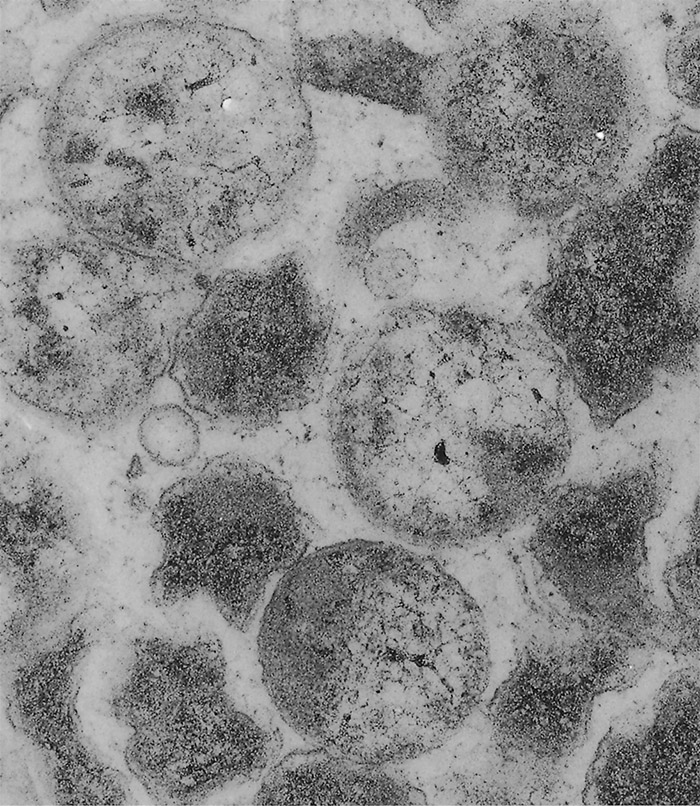
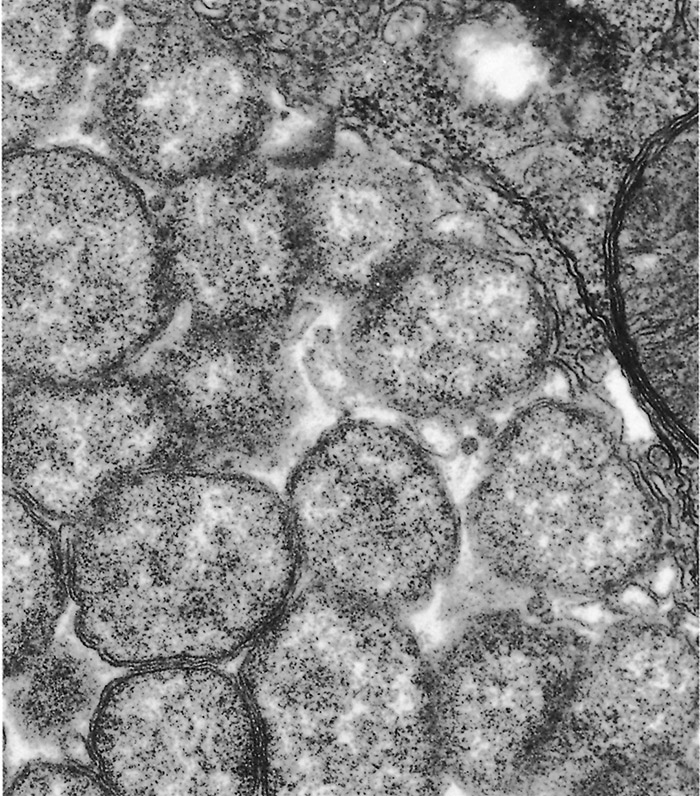
The ultrastructural morphology of E. ruminantium (Figures 3 to 6) is similar in endothelial cells of the choroid plexus of sheep, 253 endothelial cells in the lungs of mice, 272 endothelial cell cultures267, 269, and leukocytes, 176 and in the cells of ticks.38, 166, 167 Internally the cell contains electron-dense and electron-pale areas (Figure 4), with electron-dense material occupying the greater part of the inner structure of small and intermediate-sized cells (Figure 5), while electron-pale areas dominate the inner structure of large (Figure 3) and very large organisms (Figure 6). The smaller organisms are described as elementary (or electron-dense) bodies (EBs) and the larger cells are referred to as reticulated bodies (RBs). Organisms which do not readily fit into one or the other category are referred to as intermediate bodies.158, 253, 267
Life cycle
Ehrlichia ruminantium replicates mainly by binary fission, and possibly by endosporulation.253 It appears that the RBs are predominantly proliferative166, 253, 269, 272 while the EBs represent the infective stage.158 There is some evidence that E. ruminantium undergoes a sequential development in both the vertebrate96 and the invertebrate166 host. Transmission electron microscopic studies of in vitro cultures of E. ruminantium in endothelial cells have revealed the presence of intracellular reticulate bodies two to four days post-infection, and intermediate bodies four to five days post-infection.158 Large numbers of EBs are seen after rupture of the endothelial cells five to six days after culture initiation.158
Ehrlichia ruminantium was very early shown to be heat labile and to lose its viability within 12 to 38 hours at room temperature.6, 174 The first long-term cryopreservation of infective stabilates in liquid nitrogen was in media containing dimethyl sulphoxide (DMSO).152, 278 Subsequently a sucrose- potassium phosphate-glutamate medium (SPG: 0.218 M sucrose, 3.8 mM KH2PO4, 7.1 mM K2HPO4, 4.9 mM C5H8NO4K)48 was found to preserve the infectivity of the organism more effectively.51 The infective half-life of stabilate material frozen in SPG, and then thawed and kept on ice, is only 20 to 30 minutes.51 Large numbers of stabilates have been preserved from almost all the areas where the disease occurs, and the names and origins of important strains which are in current research use around the world are given in Table 1.
Table 1 Details of some well characterized E. ruminantium strains currently used for research
| Strain | Remarks | Origin | 16S genotype | Ref |
| Ball 3 | South African blood “vaccine” strain | Natural field infection, mammalian host not specified, Limpopo Province, South Africa | Ball 3 | 141 |
| Blaauwkrans | Highly pathogenic to goats in Eastern Cape, South Africa | A. hebraeum tick from an eland, Eastern Cape, South Africa | Welgevonden | 99 |
| Crystal Springs | - | Zimbabwe | Welgevonden | 58 |
| Gardel | Common Carribean genotype, annotated genome sequence 2005 NC_006831.1 | Guadeloupe | Gardel | 325 |
| Kerr Seringe | Contig assembly sequence 2016 | The Gambia | Not determined | 123 |
| Kűmm 1 | Two genotypes obtained from a single isolate | Naturally infected goat, Northern Province, South Africa | Senegal | 105 |
| Kűmm 2 | Omatjenne | |||
| Kwanyanga | Not highly pathogenic to sheep | Naturally infected sheep in the Eastern Cape, South Africa | Welgevonden | 180 |
| Mara 87/7 | Widespread South African genotype | A. hebraeum tick, Northern Province, South Africa | Mara 87/7 | 110 |
| Sankat 430 | Contig assembly sequence 2016 | Ghana | Not determined | 34 |
| Senegal | Attenuates readily in culture, scaffold assembly sequence 2013 | West Africa | Senegal | 156 |
| Welgevonden | Type specimen of | Onderstepoort, Gauteng Province, South Africa | Welgevonden | 97 |
Genetic variability
At one time it was thought that E. ruminantium was a relatively homogeneous entity, but as increasing amounts of genetic sequence data became available this notion changed profoundly and it is now known that there is a great deal of genetic variability among different E. ruminantium strains. Table 1 contains details of the origin (if known), and other important characteristics, of some E. ruminantium strains which are in widespread use for research. Currently eight distinct 16S genotypes are known which are considered to belong to the species E. ruminantium, each having a sequence identity of >99.4 per cent with respect to the others. These are: Ball 3, Gardel, Kiswani, Mara 87/7, Omatjenne, Pretoria North, Senegal, and Welgevonden. The genotypes are named from the strains from which their 16S sequences were first obtained, with the exception of Welgevonden. This sequence was first obtained from the Crystal Springs strain but is renamed here as it is the genotype of the E. ruminantium reference strain, which is widespread in southern Africa.13 Six of these genotypes are known to cause classical heartwater but two, Omatjenne and Pretoria North, apparently do not. This is not unusual; molecular genetic surveys have been carried out in areas of southern Africa which are free from clinical heartwater and where the known Amblyomma spp. tick vectors of heartwater do not occur. Examination of ticks and ruminants from these areas have detected sequences which appear to be from non-heartwater-causing genotypes of E. ruminantium.15, 16
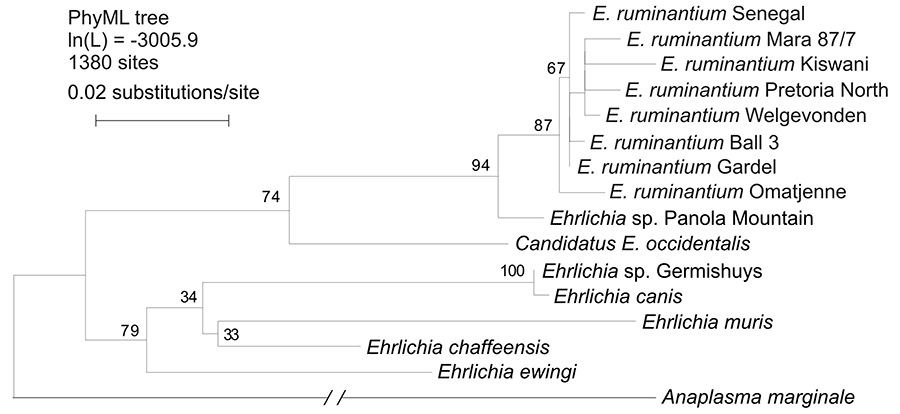
The eight different E. ruminantium 16S rRNA gene sequences noted above were compared with orthologs from five other named Ehrlichia spp., three temporarily designated Ehrlichia spp., and Anaplasma marginale. The sequences were aligned using MUSCLE112 and a maximum likelihood tree was inferred using PHYML138 running the HKY85 model. The result (Figure 7) shows that the E. ruminantium genotypes form a tight cluster, well distinguished from all the other Ehrlichia spp. Two enigmatic Ehrlichia spp., designated Ehrlichia sp. Panola Mountain and Candidatus E. occidentalis, also fall into the E. ruminantium clade, although they are more genetically distant. These organisms will be discussed below.
It has long been stated that E. ruminantium strains display differing degrees of pathogenicity in different hosts. Unfortunately much of the information in the early literature which relates to pathogenicity was obtained in the days before the organism could be propagated in tissue culture, before reliable methods for quantifying the infective dose were available, and long before molecular genetic methods of characterization had been developed. Thus there was no guarantee that individual genotypes of E. ruminantium were being investigated, nor that adequately infective challenge doses were being administered, nor even that the organisms could reliably be classified as E. ruminantium.
Infectivity to mice was at one time an important method used to demonstrate variability between strains, and three different types of behaviour have been described.179 Firstly, there are fully pathogenic strains that kill mice (e.g. Welgevonden).97 Secondly, there are strains that are infective in mice without being pathogenic (e.g. Ball 3), they induce no clinical signs, although antibodies develop, and the organism can be recovered.141 Thirdly, there are strains that fail to establish themselves in mice (e.g. Gardel), and the animals remain serologically negative.325 The difficulty of quantitating infective E. ruminantium challenge material stems from its very short half-life after frozen stabilate material is thawed. This problem has now been addressed51 but an example of the uncertainties which used to arise is shown by the history of the Senegal strain, which was originally considered not to be pathogenic to mice.157 More recently it has been realized that this strain is pathogenic to mice if the infective dose is large enough.199
The existence of immunogenic variants, at one time hardly recognized, 105 is now known to be extensive. It is obviously of crucial importance for the development of vaccines to have an understanding of which strains can confer complete or partial cross immunity one to another in ruminants. Poor cross protection between strains, or even none at all, has been shown in several experiments, 110, 156, 157 but several of the experiments included at least one isolate now known to be genetically heterogeneous.367
Reliable cross immunity trials depend upon the use of molecularly characterized strains prepared as quantified challenge material. One such experiment has been carried out in sheep using six different E. ruminantium strains:73 Ball 3, Mara 87/7, Gardel, Welgevonden, Kwanyanga, and Blaauwkrans. All except Ball 3 grow well in tissue culture, and all except Gardel are of South African origin. Sheep were infected with the appropriate strain and treated when they became febrile. Homologous challenge was carried out on all treated animals to determine their immune status and they were then subjected to heterologous challenge using 10 × LD50 of titrated single-genotype stabilate prepared in sheep blood. The results are summarized in Table 2, and it was found that the Welgevonden strain was the only one which provided complete cross protection against challenge with any of the other strains. The Kwanyanga, Gardel and Blaauwkrans strains provided little cross protection against heterologous challenge, while Mara 87/7 and Ball 3 provided limited cross protection against heterologous challenge.
Table 2 Cross-immunity protection engendered in sheep between various E. ruminantium strains72
| Strain to which immune | Challenge strain | |||||
| Welgevonden | Ball 3 | Gardel | Mara 87/7 | Kwanyanga | Blaauwkrans | |
| Welgevonden | + | + | + | + | + | + |
| Ball 3 | - | + | - | +/- | + | +/- |
| Gardel | - | +/- | + | - | +/- | - |
| Mara 87/7 | - | + | - | + | +/- | +/- |
| Kwanyanga | - | +/- | +/- | - | + | - |
| Blaauwkrans | - | +/- | - | +/- | - | + |
| + | complete cross protection |
| - | no cross protection |
| +/- | partial cross protection |
The original Kümm isolate105 offers an example of the difficulty of ensuring, in the absence of an in vitro culture technique and methods for genetic characterization, that a strain contains only a single genotype. The Kümm isolate was obtained from a goat in the heartwater endemic Northern Province of South Africa and a lymph node suspension from the animal caused what appeared to be heartwater in sheep, but the isolate was found to behave anomalously in mice179 and cattle99 and it was at one time doubted that it was E. ruminantium.96 The isolate also resisted all attempts to establish it in tissue culture over a period of 15 years. When it was eventually established in culture and characterized genetically the original isolate material was shown to contain two different genotypes, designated as Kümm 1 and Kümm 2.367 Kümm 1 had the Senegal 16S genotype while Kümm 2 had the Omatjenne 16S genotype; each Kümm genotype had a distinct behaviour in culture, with Kümm 1 growing readily in bovine endothelial cells while Kümm 2 did not. To make matters more obscure, Kümm 2 did not behave identically to Omatjenne, the former being lethal in mice while the latter was not, and the former growing readily in sheep mononuclear cells while the latter did not. Kümm 2 also has a pCS20 sequence widely divergent from those of other heartwater-causing strains of E. ruminantium.341
The strain known as Germishuys offers an example of an organism previously thought to be E. ruminantium which should be reclassified. It was isolated from a naturally infected sheep, 110 long before any molecular genetic characterization methods were available, and the records indicate that it caused clinical disease symptoms normally associated with heartwater. The organism was assumed to be E. ruminantium until 16S gene sequence data showed it to be a previously uncharacterized species of Ehrlichia more closely related to E. canis than to E. ruminantium (Figure 7).17 This organism was provisionally named Ehrlichia sp. Germishuys and no classical heartwater-causing E. ruminantium 16S sequences have ever been recovered from it. It has never been grown in tissue culture, hence its ability to cause a disease that resembles heartwater has never been properly evaluated. More recently, however, a new ehrlichial agent was isolated from the peripheral blood mononuclear cells (PBMCs) of a dog in Venezuela, using a DH82 cell culture system. It was found to have a 16S sequence 99.6 per cent identical to the sequence of Germishuys, differing by a single base.326 It would be of interest to investigate this agent for pathogenicity in ruminants.
Two strains currently considered to be E. ruminantium but not yet grown in tissue culture offer a different classification problem, these are the Omatjenne and Pretoria North genotypes. Both are phylogenetically more closely related to classical heartwater-causing E. ruminantium at the 16S level than to any other currently known Ehrlichia spp., being two of the eight distinct 16S genotypes which form the tight clade considered to represent the species E. ruminantium (Figure 7). Despite this neither of these organisms has been shown to cause clinical heartwater in ruminants, so the classification question is more difficult than in the case of Ehrlichia sp. Germishuys. The Omatjenne genotype was originally derived from a Hyalomma tick in a heartwater-free area of Namibia98 where is did not appear to cause any disease in infected cattle. The same organism is widespread in the adjacent Northern Cape region of South Africa where it is carried by apparently healthy goats.17 The Pretoria North genotype was originally detected in a dog by DNA hybridization10 and it is not currently available either as an infective strain or in culture. Both of these unusual organisms must be grown in tissue culture and tested in vivo in ruminants before it can be said whether they cause classical heartwater or pose any disease threat in ruminants.
Two recently discovered Ehrlichia organisms further complicate the problem of defining what constitutes E. ruminantium: Ehrlichia sp. Panola Mountain was isolated from a goat in Georgia, USA173 and has a 16S sequence >99.2 per cent identical with each of the eight E. ruminantium 16S genotypes; Candidatus E. occidentalis, found in Am. triguttatum ticks in southwest Western Australia128 has a 16S sequence 98.1 per cent identical with each of Ehrlichia sp. Panola Mountain and E. ruminantium (Gardel). Both of these novel organisms fall into a clade with the eight genotypes of E. ruminantium, but they are well separated from the latter as well as from each other (Figure 7). The new organisms have not been shown to produce clinical heartwater and they have not been grown in culture. Their geographic origins are also widely separated, both from each other and from all the known heartwater-causing strains. The question as to whether they should be classified as strains of E. ruminantium depends upon what criteria are adopted to define this species.
It is evident that there are many E. ruminantium-like organisms in circulation which have not been characterised and whose disease-causing abilities have not been determined, and as more genetic surveys are performed more of these organisms come to light.218 It is likely that in the future a purely phylogenetic classification will be used to define E. ruminantium, and that the organisms so defined will not all have the same infectivity and pathogenicity in mammalian hosts, nor will they all be carried or transmitted by the same species of ticks. A more detailed description of the population genetics of classical heartwater-causing E. ruminantium will be given below in the section Diagnosis: Molecular Genetic Methods.
In vitro cultivation
The multiplication of E. ruminantium within the endothelial cells of infected animals was originally described by Cowdry in 192678 so it is not surprising that the first successful in vitro propagation of E. ruminantium was achieved using bovine umbilical cord endothelial cells as host cells.45 Almost all subsequent propagation studies of E. ruminantium used endothelial cells from various species and from various anatomical sites, such as bovine aorta (BA) and pulmonary artery, 58 ovine aorta, 59 sheep brain,53 bovine brain, 201 bovine brain microvasculature314 and caprine jugular vein.317 The first successful initiation of E. ruminantium tissue culture used a tick-derived stabilate, prepared from Am. hebraeum nymphs, as the source of infectious organisms.45 In subsequent developments freshly drawn heparinized infective blood and suspensions of liver and spleen prepared from infected mice were employed to initiate cultures.39 A technique using plasma from heparinized blood was later devised, which showed that infective E. ruminantium is present in the cell-free plasma of infected animals.59
Several procedures have been described to facilitate initial infection of the endothelial cell monolayer, such as irradiation and cycloheximide treatment of the endothelial cells45 or the use of a slow-rocking platform.39 Although rocking of the culture flasks is still widely employed it has been demonstrated that these treatments are unnecessary for the initiation or propagation of E. ruminantium.314, 356 It has also been shown that continuous propagation of E. ruminantium in DH82 (canine macrophage-monocyte) cells was only achievable when cycloheximide was added.361 Ehrlichia ruminantium cultures are normally incubated at around 37 °C and, depending upon the pH of the buffer system used in the medium, either in air or in a carbon dioxide enriched atmosphere.
The propagation of E. ruminantium in non-ruminant endothelial cells was first carried out using human umbilical and human microvascular endothelial cells.314 Subsequently propagation was achieved in endothelial cell lines from a range of wild African species, including those of a bushpig (Potamochoerus porcus).295 Short-duration cultures, using cells which are unsuitable for continuous in vitro propagation of the organism, have been achieved in caprine neutrophils,176 in mouse and ruminant macrophages,92 and in monocyte-macrophage cell lines from mice and dogs.155 Apart from endothelial cell and DH82 cell361 lines, continuous propagation of E. ruminantium has also been achieved in a tick cell line from Ixodes scapularis (IDE8).32, 35, 363
Various media, including commercially available synthetic culture media, have been used for the in vitro cultivation of E. ruminantium. Examples of the latter are the Glasgow modification of Eagle’s minimum essential medium (GMEM),39 Leibovitz L-15,58 Dulbecco’s minimal essential medium (DMEM)201 and Dulbecco’s modified Eagle’s medium nutrient mixture F-12 Ham (DME/F-12).368 Commonly used supplements for a complete culture medium are foetal bovine serum (FBS), new-born calf serum, and bovine serum at various concentrations, usually 10 per cent (v/v), although 3 per cent FBS has been used in one instance.356 Another supplement frequently used is tryptose phosphate broth. The media mentioned above also contain L-glutamine and antibiotics, and glucose (0.45 per cent) has been added to an L-15 based medium58 which otherwise only contains D-galactose.
Work to develop a chemically defined culture medium for E. ruminantium passed its first milestone when a serum free culture system was achieved, using a modified HL-1 medium.368 In subsequent experiments it was found that the serum could be eliminated when DME/F-12 medium supplemented with bovine lipoproteins and bovine transferrin was used.362 The final step was replacement of bovine lipoproteins by chemically defined lipids, and bovine transferrin by an inorganic source of iron; this resulted in the first formulation of a chemically defined, protein-free medium for the continuous culture of E. ruminantium.360
A method for bioreactor cultivation of E. ruminantium for vaccine production has been developed which uses teflon bags (VueLife®) on Cytodex 3 microcarriers, where bead-to-bead transfer of cells occurs.243 The study of E. ruminantium growth and release kinetics in this system revealed the optimum time points for harvesting and re-infection in order to facilitate the production of large batches.196 Using bovine aortic endothelial (BAE) cells the Gardel strain of E. ruminantium was cultivated with an exponential growth phase from 36 -108 h post infection (hpi). The pathogen was visible as morulae at 96 hpi and the host cells started to release EBs at 108 -180 hpi. The optimum harvest time for BAE cultures is 120 hpi while for bioreactor suspension cultures it is about 113 hpi.
Tick transmission
Heartwater is transmitted by ticks of the genus Amblyomma (see Epidemiology, below, and Vectors: Ticks). Most Amblyomma spp. are three-host ticks. Larvae and nymphs become infected when they feed on domestic and wild ruminants and possibly also on certain game birds and reptiles41 at a time when E. ruminantium is circulating in the blood of these hosts. The immature stages of the tick commonly feed on smaller species of domestic and wild ruminants and game birds, while the adults prefer cattle and the larger game animals, such as African buffalo (Syncerus caffer) and giraffe (Giraffa camelopardus), as hosts.252 Nymphs or adult ticks transmit E. ruminantium to susceptible hosts without losing the infection. Intrastadial transmission has been demonstrated, 18 and transovarial transmission was once demonstrated in very heavily infected ticks under laboratory conditions44 but it has always been thought unlikely that it occurs commonly in the field.
Of concern in this respect, therefore, are the results of a survey performed recently in West Africa which showed an E. ruminantium infection rate of 29 per cent in Rhiphicephalus microplus adult ticks.46 Most importantly, transovarial transmission was also shown, so if virulent E. ruminantium is adapting to these Rhiphicephalus ticks there would be serious epidemiological implications. The diagnostic test used was the pCS20 nested PCR test,203 which is described in more detail below. It should be noted that the pCS20 test detects the Kümm 2 strain, which does not cause classical heartwater, and that the pCS20 sequence of this strain is divergent from those of other heartwater-causing strains.341 The West African workers46 also found that the sequence of the ribonuclease III gene (which forms part of the pCS20 genomic region) of their newly detected E. ruminantium was divergent from that of Ball 3, Gardel, and Welgevonden. It is therefore essential to determine whether infected R. microplus ticks are able to transmit this newly discovered E. ruminantium to ruminants, and whether classical heartwater ensues. It is worth noting that Rhipicephalus evertsi evertsi and R. evertsi mimeticus are common in the Northern Cape where they could be responsible for spreading non-heartwater-causing genotypes of E. ruminantium, the vector(s) for which have never been identified.16 It is possible that heartwater-causing strains of E. ruminantium are only transmitted by certain Amblyomma tick species; in that case the discovery of E. ruminantium-like organisms in heartwater-free areas, and in ticks which are not known to be heartwater vectors, would be less of an immediate epidemiological concern.
The developmental cycle of E. ruminantium in the Amblyomma tick vectors, and the infectivity of successive stages of the tick, are poorly understood. It is thought that after an infected blood meal, initial replication of organisms takes place in the epithelium of the intestine of the tick and that the salivary glands eventually become parasitized.166 Colonies of E. ruminantium in tick haemolymph97 may be intermediate forms in transit from intestinal to salivary gland cells. Transmission of the parasites to the vertebrate host probably takes place either by regurgitation or through the saliva of the tick while feeding. The minimum period required for E. ruminantium to be transmitted after ticks have attached to susceptible animals is between 27 and 38 hours for nymphs and between 51 and 75 hours for adults.41
Epidemiology
The epidemiology of heartwater depends upon factors relating to the tick vector, the causative organism, and the vertebrate hosts. Important considerations relating to the tick vector are infection rates in the ticks, seasonal changes influencing tick abundance and activity, and the intensity of tick control. Significant factors concerning the parasite are differences between genotypes which may affect virulence or the stimulation of immunological cross protection in ruminants. As far as the vertebrate hosts are concerned, the availability of wild animal reservoirs and the age and genetic resistance of domestic ruminant populations are of importance.322 Some of these issues are summarized here, while immunological responses to heartwater are dealt with further on in the chapter.
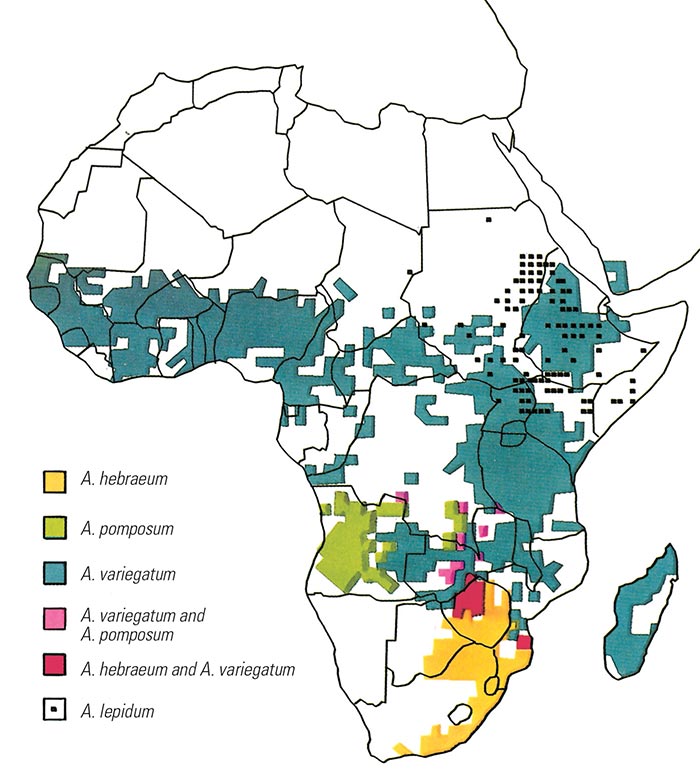
Heartwater occurs only where its vectors are present and several Amblyomma spp. capable of transmitting the organism occur widely in Africa (see Vectors: Ticks and Figure 8). The major vectors are Am. variegatum and Am. hebraeum, with the latter being the main vector in southern Africa while Am. variegatum has the widest distribution in the rest of the continent. Am. variegatum is also the only originally African Amblyomma species that has established itself successfully outside the continent, on three Caribbean islands.190, 327 The species Am. sparsum, Am. tholloni and Am. marmoreum were at one time considered to be ‘accidental’ vectors which do not normally feed on domestic stock, 252 but more recent evidence shows that this is not true for Am. marmoreum, and that this species can be an effective vector of heartwater.249 In 1999 Am. sparsum ticks, collected from leopard tortoises (Geochelone pardalis) imported into the USA from Zambia, were found to be positive for E. ruminantium by using the pCS20 genetic assay.57 However the genotype detected was not shown to be infective to ruminants and no further genetic characterization was performed. The pCS20 assay has been shown to detect non-heartwater-causing strains of E. ruminantium (see below).
Vectors of lesser importance include Am. pomposum which is prevalent in Angola; Am. lepidum which occurs in East Africa north of latitude 8°S; Am. astrion, which primarily parasitizes African buffalo in Central Africa; and Am. cohaerens and Am. gemma, which occur in East Africa.347
Two of the Amblyomma spp. native to the USA, Am. americanum and Am. cajennense, are only marginally susceptible to E. ruminantium infection and apparently do not transmit the parasite.186 Am. maculatum, on the other hand, has long been known to be capable of transmitting the disease321 and has a vector efficiency in sheep which is similar to that exhibited by Am. variegatum.186 A. maculatum and white tailed deer (Odocoileus virginianus) constitute a viable native sylvatic tick-host pair for the maintenance of E. ruminantium.
The importance of Amblyomma spp. as vectors of heartwater depends not only on their vector efficiency but also on their distribution and adaptation to domestic stock322 and on their activity and abundance, the latter being profoundly influenced by temperature and humidity.252 An increased prevalence of heartwater usually occurs when peak numbers of ticks are present, hence good rains are often followed by a transient increase in the occurrence of the disease. Its occurrence is not, however, strictly seasonal, a fact which is particularly true in regions like the Caribbean60 and in parts of Africa which have a temperate climate and a poorly defined rainy season.
Am. hebraeum ticks feeding on E. ruminantium-infected sheep have been shown to become infected during a period from two days after the commencement of the temperature reaction to two days after the animals have been treated for heartwater.41 The transmission of E. ruminantium by Am. variegatum feeding on Creole goats in Guadeloupe is apparently somewhat different, with the organism being transmitted to the ticks after a delay of two to three days for nymphs and four days for adults.65 Ostensibly healthy ruminant hosts have been shown to remain infective to ticks for long periods, at least 361 days in cattle18 and 11 months in Creole goats61. In the latter case the carrier status could only be detected intermittently during the 11-month period, demonstrating the danger which could be posed by the movement of apparently negative carrier animals to areas free from the disease.
As little as 2.7 to 5.5 ml of blood collected from a host during the febrile stage can infect Amblyomma larvae.60 Ticks retain their infectivity for life65, 150, 226 so a small number of infected ticks could presumably maintain the infection in a particular herd or area. The infection rates of vector ticks in heartwater-endemic areas are highly variable, changing according to the season, the year, and the locality in which they are collected. Surveys have been conducted in southern,14, 97, 109, 212, 230, 251 eastern214, 239, 308 and western115, 122 Africa, and on the three heartwater-infected Caribbean islands.327 The infection rates seen range from 1 per cent to 37 per cent with no clear overall pattern, and much of the variation could be due to the use of different detection assays having different sensitivities and specificities. When infected sheep are used experimentally to feed larval or nymphal Am. variegatum they subsequently show 100 per cent infection rates at the following instar.137 This suggests that the likely reason for some of the apparently low infection rates in the field may be that many ticks feed during their larval or nymphal stages on non-susceptible or non infected hosts.
Am. variegatum ticks have been seen to differ in their infection rates with different genotypes of E. ruminantium, having been shown to be less susceptible to two southern African strains than to a western African and a Caribbean strain.184 With Am. hebraeum, on the other hand, a similar level of susceptibility to infection has been demonstrated with all the strains tested. This could explain why heartwater is generally a more serious problem in those areas where Am. hebraeum is the principal vector. The phylogeographical structure of Am. variegatum is similar to that of E. ruminantium itself,30, 298 but there are no recorded phylogenetic studies of Am. hebraeum populations which could help to clarify this question.
When a pathogenic genotype of E. ruminantium infects a susceptible vertebrate host either inapparent or overt disease may develop, depending on the pathogenicity of the organism and on the species, breed, age, degree of natural resistance, and immune status of the host. Young calves, lambs and goat kids possess a reverse age resistance which is independent of the immune status of the dam.7, 108, 227, 319, 355 This resistance usually lasts for only the first four weeks of life in calves and for the first week in lambs and kids,227 although it has been seen to persist for six to eight months in calves.108 This age resistance is not absolute as infection of some calves less than three weeks of age, and of some lambs and kids less than one week old, can result in fatal disease.121, 227, 319 Although the reason for this resistance is unknown it could be that susceptible target cells for E. ruminantium infection are absent, or only present at low levels, in immature ruminant immune systems.
The susceptibility of different breeds of cattle to heartwater is variable, with Bos indicus (Zebu) breeds being in general more resistant than European (Bos taurus) breeds.322 The resistance of rural Zebu breeds, such as Nguni and Sanga, is probably due to an inherited resistance acquired through years of natural selection. This resistance does not prevent the establishment of infection but reduces the severity of clinical disease.322 The presence of conglutinin in the serum appears to be involved in non-specific resistance to heartwater in cattle.101 Vertical transmission of the disease from mixed-breed cows to calves has been shown to occur in a heartwater-endemic area of Zimbabwe,86 and has also been reported from sheep and goats to lambs and kids, respectively, in Gambia.121 It is speculated that this transmission may occur via infected cells in the colostrum or via the in utero route. If this is correct such transmission could presumably occur in other ruminant species. This resistance does not prevent the establishment of infection but reduces the severity of clinical disease.322 Conglutinin in the serum appears to be involved in non-specific resistance to heartwater in cattle.101 Vertical transmission of the disease from mixed-breed cows to their calves has been shown to occur in a heartwater-endemic area of Zimbabwe,86 and it is speculated that infected cells in the colostrum are at least one route by which this transmission may occur. If this is correct such transmission could presumably occur in other ruminant species.
Sheep are more susceptible to heartwater than are cattle, but variations in susceptibility between breeds of sheep are less than those in cattle breeds. Angora goats are highly susceptible to heartwater and their immunity is of short duration.104 Some natural resistance has been observed in Blackheaded Persian sheep6, 322 but no work appears to have been done on the genetic basis for this finding. Genetic resistance that is transmitted preferentially by dams66 has been demonstrated in goats on Guadeloupe.204, 283 Attempts are now being made to define the genetic loci involved in heartwater pathogenesis as a prelude to attempting to breed heartwater-resistant livestock.
A summary of published information on the susceptibility of wild ruminants to heartwater is presented in Table 3.235 From observations made on game animals infected in captivity it appears that antelope such as young black wildebeest (Connochaetes gnu), adult springbok (Antidorcas marsupialis), and water buffalo (Bubalus bubalis) with low levels of serum conglutinin, are susceptible, whereas adult black wildebeest, red hartebeest (Alcelaphus buselaphus) and scimitar horned oryx (Oryx dammah) with high levels of conglutinin proved to be highly resistant.108 Only the eland (Taurotragus oryx), blesbok (Damaliscus dorcas phillipsi), springbok, and black wildebeest have been reported to develop clinical heartwater disease.235 It appears that the most important natural ruminant reservoirs in southern Africa may be blesbok,221 black wildebeest,221 African buffalo,14, 18 and eland.350 Heartwater appears to have originated in Africa and African wild ruminants probably constitute the natural reservoir of the disease,225 however heartwater can maintain itself in the absence of a wild ruminant reservoir, as is seen in Madagascar, Guadeloupe and Sao Tomé.322
Helmeted guinea fowl (Numida meleagris), leopard tortoise (Geochelone pardalis) and scrub hare (Lepus saxitilis)41 have also been proven to harbour E. ruminantium subclinically after artificial infection, although little is known about their possible role as sources of infection. In addition, the multimammate mouse (Mastomys coucha)179 and the striped mouse (Rhabdomys pumilio) are susceptible to infection,147 but as wild rodents probably do not act as hosts of Amblyomma spp. in heartwater endemic areas they are unlikely to play a role in the epidemiology of the disease.146
Table 3 The susceptibility of various wild ruminants to E. ruminantium233
| Hosts (African) | Susceptibility | |||
| Common name | Scientific name | Clinical | Sub- clinical | Refractory |
| Giraffe | Giraffa camelopardalis | + | + | |
| Black wildebeest | Connochaetes gnu | + | + | + |
| Blue wildebeest | Connochaetes taurinus | + | ||
| Red hartebeest | Alcelaphus buselaphus | + | ||
| Blesbuck | Damaliscus dorcas phillipsi | + | + | |
| Duiker | Cephalophus sp. | + | ||
| Springbuck | Antidorcas marsupialis | + | ||
| Scimitar-horned oryx | Oryx dammah | + | ||
| African buffalo | Syncerus caffer | + | ||
| Bushbuck | Tragelaphus scriptus | + | ||
| Eland | Taurotragus oryx | + | + | |
| Hosts (Non - African) | ||||
| White tailed deer | Odocoileus virginianus | + | ||
| Fallow deer | Cervus dama | + | ||
| Timor deer | Cervus timorensis | + | ||
| Water buffalo | Bubalus bubalis | + | ||
| Barbary sheep | Ammotragus lervia | + | ||
| Himalayan tahr | Hemitragus jemlahicus | + | ||
| Nilgai | Boselaphus tragocamelus | + | ||
| Blackbuck | Antilope cervicapra | + | ||
| Mouflon | Ovis orientalis | + | ||
Pathogenesis
The pathogenesis of heartwater is not well understood. Vertebrate hosts are infected with E. ruminantium organisms through the saliva of attached ticks and/or by their regurgitated gut content.40, 166, 168 Initial replication of the organisms seems to take place in reticulo-endothelial cells and macrophages in the regional lymph nodes, after which they are disseminated via the bloodstream and invade endothelial cells of blood vessels in various organs and tissues, where further multiplication occurs.91 In domestic ruminants E. ruminantium most readily infects endothelial cells of the brain, and this coincides with the onset of the febrile reaction.91, 93 Genetic variation within both parasite and host populations plays a role in the degree of endothelial cell colonization in the brain and other organs.54
Increased vascular permeability with transudation is responsible for effusion into body cavities and tissue oedema, and this is particularly noticeable in the lungs, pericardial sac and pleural cavity. Oedema of the brain is responsible for the nervous signs, hydropericardium contributes to cardiac dysfunction during the terminal stages of the disease, and progressive pulmonary oedema and hydrothorax result in eventual asphyxiation.240 The effusion of fluid into tissues and body cavities also results in a drastic reduction in blood volume.69
The pathogenesis of the vascular permeability remains speculative, and in mice there is no apparent correlation between the number of parasitized endothelial cells in the pulmonary blood vessels and the severity of the pulmonary lesions.272 It has been suggested that endotoxin333 and increased cerebrospinal fluid pressure54 play a role in the development of lung oedema.
Clinical signs
Infected domestic ruminants may manifest a wide range of clinical signs. The incubation period, course, severity and outcome of artificially induced disease are influenced by the species, breed and age of animal affected, the route of infection, the virulence of the strain of E. ruminantium involved, and the amount and source of infective material administered.6, 226, 322 Peracute, acute, subacute, and clinically inapparent forms of the disease occur.
In the naturally acquired disease in a herd or flock, the morbidity and mortality rates are influenced by the species, breed and age of the animals affected, the virulence of the E. ruminantium strain, the effectiveness of immunization, tick control and specific chemotherapy programmes applied on the farm, and, in some instances, the season.37, 62, 108, 142 Death usually follows in animals which show clinical signs if they are not specifically treated for heartwater.
Cattle
The incubation period in naturally infected cattle ranges from 9 to 29 days, with an average of 18 days.6 Cows of B. taurus breeds, such as the Jersey, South Devon, Charolais, Limousin and Friesland, especially when in the advanced stages of pregnancy, are particularly prone to develop peracute heartwater, a form of the disease which may sometimes also occur in 6- to 18-month-old animals of different breeds.144, 338 Peracutely affected animals die within a few hours after the initial development of fever, either without any clinical signs having been manifested, or having shown terminal, paroxysmal convulsions and marked respiratory distress.6, 226
Acute heartwater, the most common form of the disease, mainly affects cattle between the ages of 3 and 18 months. It is characterized by a fever of 40°C or higher, which usually persists for three to six days, showing only small fluctuations before the body temperature falls to subnormal shortly before death.6, 108 Some observers consider that a mild mucoid diarrhoea is a regular occurrence,6, 80, 108 but others regard it as an infrequent sign.337 Certain breeds, such as the Friesland, Jersey and Simmentaler, and four- to eight-month-old animals of all breeds, apparently develop diarrhoea most commonly. A profuse, often haemorrhagic, diarrhoea may be the most prominent clinical sign in some cases of heartwater, particularly in Jerseys.6, 337
During the later stages of acute heartwater, nervous signs occur which range from mild incoordination to pronounced convulsions.6 The animals are hypersensitive when handled or exposed to sudden noise or bright light. Slight tapping with a finger on the forehead of the animal often evokes an exaggerated blinking reflex. They frequently show a peculiar high stepping gait that is usually more pronounced in the front limbs. Calves may wander around aimlessly and walk into fences, and some, previously unaccustomed to handling by humans, may be approached with ease. Animals may stand with their heads held low, make constant chewing movements, and push against objects. In the later stages they often fall down suddenly, assume a position of lateral recumbency, and show opisthotonus and either have frequent bouts of leg pedalling movements or the legs may be extended and stiff. In most cases the animals weaken rapidly and death usually follows soon after the commencement of a convulsive attack.
The subacute form of heartwater is characterized by a fever which may remain high for 10 days or longer. The clinical signs are similar to those described for the acute form of heartwater, but less pronounced. During the course of the illness animals may die suddenly, or gradually recover within a few days, and death frequently results from complications such as hypostatic pneumonia.6
The prevalence of the clinically inapparent form of heartwater is difficult to determine under field conditions. Apart from fever, apathy and slight tachypnoea, the animals appear to be normal and most recover within a few days.62 Calves less than three weeks of age,108 cattle infected with a strain of low virulence,226 and infected but partially immune animals,6 are particularly prone to develop this mild form of the disease.
Sheep and goats
The incubation period in sheep and goats inoculated intravenously with 10 ml of infected blood varies from 5 to 35 days (average 9 to 10 days), and that of naturally infected animals from 7 to 35 days (average 14 days).6, 322
Exotic goat breeds, such as the Angora, and two- to six-month-old Boer goats, are commonly affected by the peracute form of the disease. Most animals collapse suddenly and die after a few paroxysmal convulsions without having been observed to be ill.322 Some, however, are anorectic, appear dull, exhibit nystagmus, bleat, and twitch their tails continuously. They may show forced respiration, frequent urination and defaecation, chewing movements and, when recumbent, leg-pedalling.161, 297
As is the case in cattle, acute heartwater is the most common form of the disease in sheep and goats. The majority of animals manifest nervous signs, but these are generally less pronounced than in cattle.6 Affected animals initially show fever, a progressive unsteady gait, and listlessness, and often stand with their legs wide apart with the head lowered and ears drooping. They eventually become prostrate, assume a position of lateral recumbency and show intermittent leg pedalling, chewing movements, opisthotonus, licking of the lips, and nystagmus.6
Black-headed Persian sheep possess some natural resistance to heartwater, and lambs and kids of all breeds under one week of age have a degree of innate resistance. Such animals, as well as those which are partially immune, may develop the mild form of heartwater and, apart from fever, no clinical signs are generally manifested in these cases.6, 322
Wild ungulates
Clinical signs of heartwater in susceptible wild ungulates (Table 3) have not been well studied but are generally similar to those reported in domestic ruminants suffering with the acute form of the disease.130, 132, 353
Laboratory mice
The incubation period in mice infected intravenously with the Welgevonden strain of E. ruminantium ranges from 10 to 14 days.272 No febrile reaction has been noted in mice experimentally infected with any of the strains to which they are susceptible.179 Twenty-four hours prior to death affected animals are lethargic, and develop tachypnoea and a ruffled coat. Incoordination is occasionally seen.180
Pathology
A slight reduction in haemoglobin and haematocrit values is observed in heartwater-infected sheep, goats and calves. The reductions coincide with, or start shortly after, the commencement of the febrile reaction and continue throughout the course of the disease, but neither value reaches critically low levels.1, 69, 240, 334 The anaemia, which is not clinically discernible, is usually normocytic and normochromic,334 although a microcytic, hypochromic anaemia has also been reported in goats.1 The pathogenesis of the anaemia is not known.
During the course of the disease the colour of the blood plasma of calves, sheep and goats may change progressively to a dark orange colour69, 133 as a result of increased levels of bilirubin and/or its conjugates.209 In calves the bilirubinaemia during the initial stages of the disease appears to be associated with anorexia rather than with haemolysis or liver damage.334
Mild leukopenia, mainly resulting from a decrease in the number of neutrophils, develops in calves and goats prior to the onset of fever and persists throughout the course of the acute form of the disease.151 An eosinopenia develops before or during the onset of the febrile reaction, and is accompanied by a lymphocytosis which commences shortly after the onset of the febrile reaction.1, 69, 240, 334
The total serum protein content is reduced in calves. The commencement of this reduction coincides with the start of the febrile reaction and persists throughout the acute stage of the disease.334 High concentrations of albumin and globulin occur in the oedematous fluid which accumulates in body cavities.334
Elevated levels of blood urea nitrogen69, 131 and creatinine have been detected in the blood shortly after the onset of the febrile reaction in sheep, whereas in calves these parameters are not higher than the upper normal levels. These observations suggest some interference in glomerular function during the acute stage of the disease.334 Blood glucose levels rise in the terminal stages of the disease which may cause glucosuria in sheep and goats.69, 131, 151
Terminal increases in blood pyruvate and lactate levels, and a simultaneous drop in bicarbonate, have been reported in sheep and goats.69, 151 During the acute stage of the disease in calves respiratory alkalosis and hypoxia occur, as evidenced by a lowered oxygen tension (pO2).334 Sheep infected with the Ball 3 strain show retention of carbon dioxide (increased pCO2) resulting in respiratory acidosis on the twelfth day post-infection. In addition there is a reduced oxygen diffusion which gives rise to hypoxia (lowered pO2) that persists until death.240 Other experiments in sheep showed changes in blood gas levels, including a decline in arterial pO2 combined with a respiratory alkalosis. Although the sheep became hypoxaemic blood-gas changes associated with respiratory failure were not observed.335
In sheep increases in both the respiratory rate and tidal volume result in increased respiratory minute volume and a simultaneous increase in ventilatory equivalent.240 The tendency towards increases in both physiological and alveolar dead space may be associated with lung oedema, and could account for the moderate reduction in pO2 which occurs during the acute stage of the disease.334 Also observed in sheep are changes in blood-clotting behaviour, blood calcium and protein levels, haematocrit, and white-cell counts. A marked decline in thrombocyte count is seen during the acute stage of the disease, associated with increases in both prothrombin time and activated partial thromboplastin time. Fibrinogen levels increased, while there was no detectable increase in fibrinogen degradation products, and total serum protein, albumin and globulin levels dropped very sharply. Total calcium showed a progressive drop, but ionized calcium rose initially and was then followed by a terminal decline. The total leukocyte count showed a terminal increase while the haematocrit dropped progressively.336
Lesions in cattle, sheep and goats are very similar and differ only in their severity and frequency.6, 108, 268, 300 Gross lesions may be inconspicuous or even virtually absent in fatal cases.268
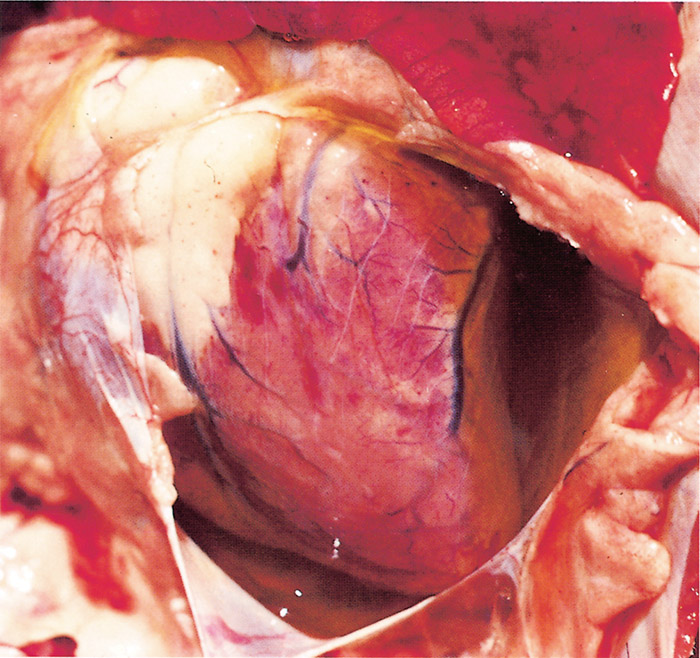
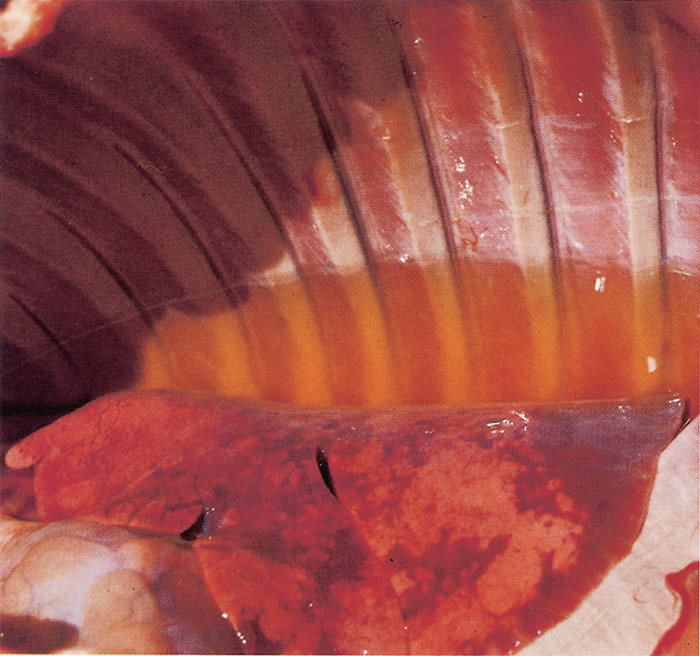
Severe hydropericardium (Figure 9) and hydrothorax (Figure 10), and in some cases a degree of ascites, are striking changes in most fatal cases of heartwater. However, hydropericardium is usually more pronounced in sheep and goats than in cattle.144 The transudate is a transparent to slightly turbid, light yellow fluid which may coagulate on exposure to air. Several litres of it may be present in the thorax in cattle, while in sheep up to 500 ml, and in goats rarely more than 20 ml, may be present.300
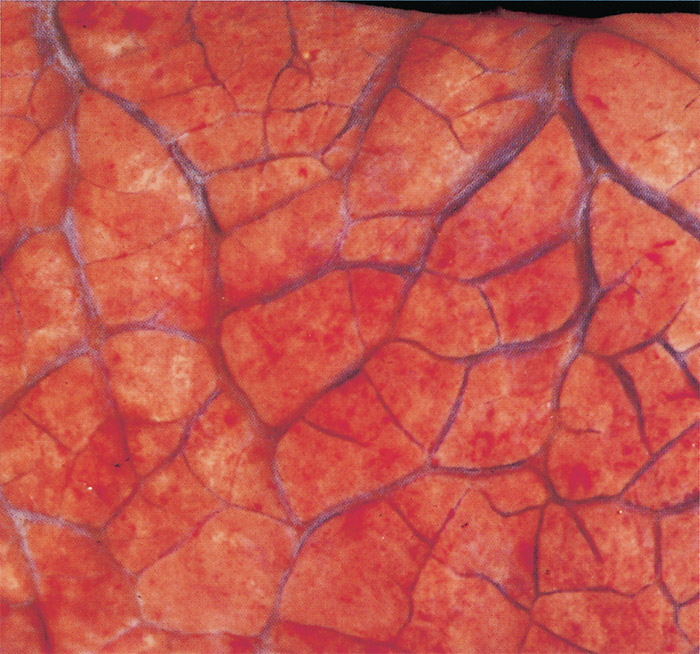
A moderate to severe oedema of the lungs occurs in most animals that die of the disease (Figure 11), but it is particularly severe in animals which have suffered from the peracute or acute form.337 Frothy oedematous fluid oozes from the cut surface of the lungs. The trachea and bronchi are often filled with a frothy serous foam occasionally accompanied by a fibrinous coagulum, and their mucous membranes are often congested and contain petechiae and ecchymoses. The mediastinum and bronchial lymph nodes may also be oedematous. A slight to moderate splenomegaly resulting from congestion and lymphoid hyperplasia is found in most animals, but in sheep and goats it is often not as severe as in cattle.
Oedema of the brain commonly occurs in animals suffering from the peracute and acute forms of heartwater.254, 337 Occasionally the entire brain, but particularly the gyri of the cerebrum, is prominently swollen, and this results in a partial herniation of the cerebellum through the foramen magnum. Most fatal cases show varied degrees of congestion and oedema of the meninges. The choroid plexus is swollen and dull greyish in appearance. In some animals, petechiae, ecchymoses and sometimes suggilations are evident in the brain substance, particularly of the midbrain, brain stem and cerebellum.254 Microscopic lesions in the brain are characterized by changes compatible with oedema, such as widened perivascular spaces which in tissue sections sometimes contain material representative of oedematous fluid or protein droplets, swollen and often necrotic astrocytes, swollen axons, and microcavitations, particularly in the white matter. Occasionally encountered are small haemorrhages in the neuropil, scant cellular perivascular accumulation of mainly macrophages and lymphocytes, fibrinoid vasculitis, fibrinous choroiditis and multifocal glial nodules, the latter particularly occurring in the white matter around small blood vessels.254 Widespread status spongiosus of varying severity, that mainly affects the larger tracts of white matter in the brain, may be found in animals that have been recumbent for days, and particularly in those that have been specifically but unsuccessfully treated a few days before death.266
Nephritis of varying degrees, sometimes accompanied by perirenal oedema and petechiae in the renal cortex, occurs in most fatal cases of heartwater.268 Renal tubular epithelial cells are swollen and occasional tubules may contain hyaline casts. Severe nephritis is particularly obvious in Angora goats that have been unsuccessfully treated for E. ruminantium after the first day of the febrile reaction, and killed in extremis four to seven days after the onset of the febrile reaction, or which have died three to four days after the first evidence of fever.270
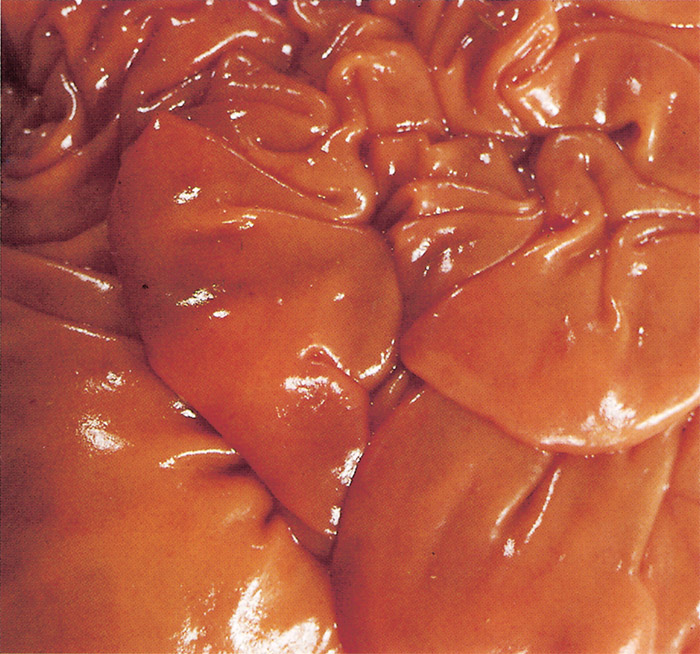
Congestion and/or oedema of the abomasal folds (Figure 12) are regular findings in cattle but are not as common in sheep and goats. Petechiae may also be present in the mucosa of the abomasum. Cattle which have shown some clinical evidence of diarrhoea often reveal the presence of a diffuse catarrhal enteritis of varying intensity. In others, particularly Jersey cattle breeds, severe diffuse enterorrhagia, together with intense congestion of the mucosa of particularly the small intestine, may be the most prominent of all the lesions.
Many lymph nodes are found to be moderately swollen, and cut surfaces are oedematous and congested, and may contain small haemorrhages.6 Petechiae, or even larger haemorrhages, may be present in serous and/or mucous membranes of tissues or organs such as the epi- and endocardium, urinary bladder, vagina and conjunctivas.
Variable numbers of E. ruminantium colonies are discernible in the cytoplasm of endothelial cells, and particularly those of the brain (Figures 1 and 2), lungs and kidneys, but generally they are difficult to find in haematoxylin and eosin stained tissue sections.
The lesions in game animals that die of heartwater are very similar to those reported in domestic ruminants.235, 268
In mice infected with the Welgevonden strain the lesions closely resemble those in cattle, sheep and goats that have died from heartwater.272 In tissue sections the endothelial cells of the lungs contain the highest concentration of organisms, followed by those of the myocardium. Organisms are only rarely detected in capillaries in tissue sections of the brain of mice.
Transmission electron microscopical studies of the lung lesions in sheep, goats and mice reveal the presence of minor cytopathic changes in endothelial cells. Apart from mild swelling of mitochondria and endoplasmic reticulum no other changes occur in most parasitized alveolar endothelial cells. Non-parasitized endothelial cells are sometimes swollen, or even necrotic, and are separated from their basement membranes. Oedema of blood vessel walls is infrequently seen.271, 272
Diagnosis
Clinical Signs
Nervous signs occur in most animals suffering from heartwater and they must be distinguished from a wide range of infectious and non-infectious conditions that manifest similar signs. In cattle nervous signs may be caused by other infections such as rabies, the nervous form of malignant catarrhal fever, cerebral babesiosis, cerebral theileriosis, chlamydiosis, meningitis and encephalitis caused by various bacteria, especially Streptococcus spp., Pasteurella spp., Arcanobacterium pyogenes, and Haemophilus spp. In sheep and goats meningitis and encephalitis are caused by a wide range of bacteria, and abscessation of the hypophysis (pituitary abscess) occurs, particularly in goats. In southern Africa nervous signs in cattle may be the result of poisoning with plants or fungi such as Albizia versicolor, Albizia tanganyicensis, Cynanchum spp., Euphorbia mauritanica, Sarcostemma viminale, Cynodon dactylon, Aspergillus clavatus and Claviceps paspali.163 Other poisons which may also induce nervous signs are pesticides (e.g. chlorinated hydrocarbons and organic phosphates) and heavy metals (such as lead and mercury).70, 306, 307 In sheep and goats plant poisoning (especially by Cynanchum spp., Euphorbia mauritanica, Sarcostemma viminale and Cynodon dactylon), and heavy metal and pesticide poisoning, induce nervous signs similar to those seen in cattle suffering from heartwater.
Lung oedema, hydropericardium and hydrothorax are common necropsy findings in cattle, sheep and goats that have died of heartwater, but they are also regular findings in the case of gousiekte (‘quick disease’) caused by the ingestion of the rubiaceous plants Pachystigma pygmaeum, Pachystigma thamnus, Pachystigma latifolius, Fadogia homblei, Pavetta harborii and Pavetta schumanniana.163 Lung oedema is also found in sheep that have succumbed to pulpy kidney disease or bluetongue, and in cattle suffering from Corridor disease or East Coast fever.
The diagnosis in cattle, particularly Jerseys, which have died from the form of heartwater in which intestinal changes are the most prominent, should be differentiated from poisonings by, for example, heavy metals (arsenic, mercury), plants (Homeria, Moraea, Urginea, Ornithoglossum and Ornithogalum spp.) and organophosphates.
Chlamydophila pecorum and E. ruminantium may be difficult to differentiate morphologically in brain smears or tissue sections made from animals which have died of the respective infections. Both organisms are pleomorphic, and elementary, intermediate and reticulated bodies are found in both of their multiplication cycles. As a rule, the organisms within a colony of E. ruminantium are of the same morphological form, either all EBs or all reticulated bodies.253 A colony of Chlamydophila, however, may contain EBs (0.24 to 0.4 μm in diameter) and reticulated bodies (0.6 to 1.5 μm in diameter).22 The histopathological lesions in the brain of the two diseases usually differ markedly: those caused by C. pecorum are generally characterized by a moderate to severe multifocal or diffuse lymphocytic meningoencephalitis, vasculitis and, in some instances, also by thrombosis and small foci of necrosis of the neuropil; on the other hand oedema of varying severity is usually the only lesion that is encountered in the brains of animals that have died of heartwater.
The traditional method of making a post-mortem diagnosis of heartwater is the demonstration by light microscopy of E. ruminantium in the cytoplasm of endothelial cells of blood vessels in stained smears of brain tissue.274 Organisms may also be found in tissue sections of the brain, or other organs such as the kidneys.
Brain smears are prepared in such a way that segments of capillaries remain more or less intact and can be examined after staining. A small piece (less than 5 × 5 × 5 mm) of hippocampus or cerebral grey matter is obtained either by opening the skull or by scooping some brain material through the foramen magnum.285 Smears are made by crushing the sample between two microscope glass slides until the tissue has a soft, pasty consistency. The material is then collected at the end of one of the slides, which is held firmly in a horizontal position. The other slide, angled at about 45°, is used to make the smear by drawing the tissue along the horizontal slide. While making the smear it is preferable to lift the angled slide slightly, about every 10 mm, so that the smear has alternating thick and thin areas. This procedure stretches the capillaries linearly and facilitates their microscopical detection. Smears should be air-dried before staining.274, 322
Various stains may be used to demonstrate heartwater organisms, such as Giemsa or CAM’s Quick Stain (C.A. Milsch (Pty) Ltd.),322 but Giemsa is the method of choice unless large numbers of organisms are present. For Giemsa staining smears are fixed for about one minute in absolute methanol or ethanol, and are then stained in aqueous Giemsa, either 30 min in 10 per cent or 10 min in 50 per cent cent solution. CAM’s Quick Stain yields acceptable results where a high concentration of organisms are present and requires only 2-3 min to complete. Fixed or unfixed smears are suitable for staining and diagnostic purposes for at least one month after they have been prepared.322
Colonies of E. ruminantium are generally easy to find in smears of untreated cases of heartwater, but their numbers may vary widely from one animal to another. Organisms can often still be demonstrated in brain smears prepared from animals in an advanced state of putrefaction. The examination of brain biopsies in live animals for the confirmation of a diagnosis of heartwater is useful in experimental animals, but is not practical under field conditions.64, 305, 331
For the histopathological diagnosis of heartwater in ruminants by the examination of tissue sections the kidneys and brain are the preferred organs, E. ruminantium particularly being sought in endothelial cells of the renal glomeruli or capillaries of the grey matter of the cerebral cortex.78 Because of the low numbers of organisms in these tissue sections it is a time-consuming method of confirming a diagnosis, and may even yield an inconclusive result. Various staining methods may be applied to demonstrate E. ruminantium in tissue sections.56 but those incorporating toluidine blue or Giemsa are preferred.266
The confirmation of a diagnosis of heartwater is often difficult in animals that have been treated. A small number of treated animals, particularly those that have received inadequate chemotherapy, do not recover fully, remain recumbent for days, and eventually have to be euthanased for humane reasons. Most of these cases reveal histologically a status spongiosus of varying degrees in the brains,266 but the typical macroscopical lesions of heartwater are usually absent at necropsy, and identification of E. ruminantium in brain smears 48 to 60 hours after an animal has been treated is often difficult. In these cases brain smears should be stained for 30 min in 10 per cent aqueous Giemsa. The electron-dense bodies are more severely affected by chemotherapy than are the reticulated bodies, and the organisms are poorly delineated and appear to fuse. This makes it difficult to distinguish them from phagosomes and chromatin in endothelial cells, and from groups of blood platelets and mast cell granules.266
Serological methods
The first serological test used for surveys for E. ruminantium, developed in 1981, was the indirect fluorescent antibody test (IFAT).94 The target antigens were peritoneal macrophages from mice infected with the Kümm isolate,106 or goat neutrophils or cells from endothelial cell cultures.202 An enzyme-linked immunosorbent assay (ELISA) was also developed.220 In both of these tests cross reactions with antibodies against related Ehrlichia and Anaplasma spp. occur, resulting in the common occurrence of false positive results.60, 109, 145, 175 This was particularly evident when the IFAT assay was recently used in South Africa, where it detected an unusually high incidence of positive goats from endemic areas as well as non endemic areas.206
A competitive ELISA (cELISA) test using a monoclonal anti-MAP1 antibody was developed ten years later,156 but this too gave false positive reactions24 with related Ehrlichia spp. This difficulty did not prevent the MAP1 cELISA assay being used to track the development of antibodies, presumed to be predominantly anti-E. ruminantium, in a rural area in Ghana where heartwater-vector Am. variegatum tick vectors were present on cattle, sheep and goats throughout the year.32 The results showed that seroconversion occurred in most livestock, and usually before 12 months of age; subsequently antibody levels declined to zero in most cattle, persisted in most sheep, and showed an intermediate pattern of persistence in goats.
The cELISA test was modified in 1995, in an attempt to improve its specificity for E. ruminantium, by the use of a fragment of MAP1, designated MAP 1B, in an indirect ELISA format.344 The improved test has been shown to have a higher specificity for E. ruminantium than any other serological test, and it does not cross react with antibodies against three other organisms in the order Rickettsiales which commonly infect ruminants, Anaplasma bovis, A. ovina and A. phagocytophila.344 Note, however, that the MAP 1B ELISA test does detect antibodies to the non-heartwater-causing Omatjenne strain of E. ruminantium,17 as well as to other Ehrlichia spp., notably E. canis, E. chaffeensis, and an unidentified Ehrlichia sp. infecting white-tailed deer (Odocoileus virginianus) in the south-eastern USA.162
Apart from this caveat the MAP 1B ELISA test is valuable for use in sheep and goats5, 84, 123, 159, 164, 205, 302 and cattle.164, 303 In cattle, however, antibody levels against E. ruminantium can be very low in heartwater endemic areas, even in cattle that have been vaccinated or are under continuous natural challenge by infected ticks.84, 288 Antibody levels decline in cattle to such an extent that the animals become seronegative 14 to 33 weeks after initial exposure, owing to a down regulation of MAP1-specific antibody responses post-recovery.288 Results confirming this effect in cattle have been reported using the IFA test109 and by immunoblotting.84, 288 Care must therefore be taken when using any serological test in cattle, especially if the animals are being tested in order to decide whether it is safe to move them to a non-endemic area, since it is known that they may be tick-infective subclinical carriers of heartwater.18
The reason for the false positive cross reactions which plague all the MAP1 antigen tests is that many other species of Anaplasmataceae have orthologous gene families coding for antigenic outer membrane proteins (OMPs) in the 28 to 30 kDa size range.229, 237, 241, 354, 358 In E. ruminantium the map gene family comprises 16 paralogs.340 Since one cannot rely on distinguishing between species closely related to E. ruminantium by serology, and since it is not possible to distinguish between different species of Ehrlichia by light microscopy, the only way reliably to characterize the heartwater parasite is to use molecular genetic methods.
Molecular Genetic Methods
The molecular genetic revolution which has taken place since the 1980s has transformed diagnostic techniques by making it possible to detect the presence of minute amounts of parasite genetic material amid a huge excess of host DNA. The technological developments behind this revolution are: (i) the specificity of DNA-DNA hybridization probes; (ii) the capability to amplify, by several orders of magnitude, specific target sequences using the polymerase chain reaction (PCR); (iii) the ease of access via the Internet of huge public databanks of genetic information; and (iv) the ready availability of enormous computer power to allow the searching for, and manipulation and analysis of, sequence data. For E. ruminantium diagnosis three families of probes are in use, targeting the pCS20 genomic region, the 16S rRNA gene, and the map1 gene.
The pCS20 genetic region was the first genetic target to be identified for E. ruminantium diagnosis345 and it was originally thought to be specific for E. ruminantium, giving no cross reactions with other Ehrlichia species tested.13 The original cloned genetic region was chimaeric and a redesign of primers and probes for the region resulted in a more sensitive test.341 Several different pCS20-based diagnostic assays have been developed, including a nested PCR,203 a quantitative real-time PCR Taqman™ probe assay,301 and a loop-mediated isothermal amplification (LAMP) assay.219 The various pCS20 assays have been widely used to detect E. ruminantium in domestic animals, wild game, and ticks.14, 185, 187, 189, 247, 248, 250, 292
Despite the successful use of pCS20-based assays it is known that there are sequence polymorphisms in the pCS20 region of different E. ruminantium strains,341 and the real-time Taqman™ modification of the test, designed for speed and convenience, also shows some cross reactions with E. chaffeensis and E. canis. The need to distinguish Ehrlichia sp. Panola Mountain (see above: Genetic Variability) from classical E. ruminantium led to the design of a dual-plex Taqman™ quantitative PCR (qPCR) assay targeting the groEL gene. This assay is capable of distinguishing 23 strains of E. ruminantium from Africa and the Caribbean from all other currently known species of Ehrlichia284 and it may well become the test of choice for the specific detection of E. ruminantium.
The 16S rRNA gene has been very widely used as a taxonomic and phylogenetic tool for classifying bacteria.238 Among the eight different 16S genotypes classified as E. ruminantium (Table 1, Figure 7) six are known to cause virulent heartwater in ruminants, which suggests that they are examples of what was traditionally understood to be E. ruminantium. The remaining two organisms are the Omatjenne genotype, which does not appear to be pathogenic to goats17 or cattle,98 and the Pretoria North genotype, which was found in dogs.10 The infectivity for ruminants of this latter organism is not known. All eight of these E. ruminantium genotypes give a positive reaction with the pCS20 probe and, because of the uncertainty which this may induce, it has been routine to use the 16S probes as well as the pCS20 probe on animals which are being examined to obtain permits for importation into non-endemic heartwater areas. The most recent development of the 16S assay for Ehrlichia spp. is a FRET-qPCR that can detect and differentiate four Ehrlichia groups: (E. canis, E. muris, E. ovinia), (E. chaffeensis, E. ewingii), and )Ehrlichia sp. Panola Mountain, E. ruminantium) based on melting point analysis in a single reaction.359
Despite the complicating existence of related OMPs in other Anaplasmatacea the map1 gene has been used as a target for the molecular genetic diagnosis of E. ruminantium.13, 203, 281 Because of its extensive polymorphism it was advocated for use as a marker for strains with differing antigenicities from different geographical areas,279 but as more data became available it was found that the variants were not geographically constrained.12
The aim of E. ruminantium molecular genetic characterization studies has progressed beyond just dissease diagnosis to the need to identify different antigenic types suitable for incorporation into future vaccines. With this in mind several workers have extended the map1 studies to other members of the map gene family, which comprises 16 paralogs.340 One such study sequenced the map1 genes from 80 strains and isolates, plus five other less variable map paralogs from a subset of the isolates.277 The different map paralogs were independently assorted, and there was no correlation between any overall map genotype and geographical origin, with all the different paralog assortments being found on all the different continents. Even more surprising was that the range of different overall map genotypes was the same whatever the scale of sampling - village, region, or continent.
In summary it appears that map1 is the best single gene for identifying different genotypes of E. ruminantium across all the regions where the organism is found, but the different map1 genotypes do not correlate with geographical origin or virulence,12 or cross protective immunogenicity.2 It is in fact highly unlikely that any single gene will be found to be suitable for virulence or immunogenicity characterization, which has stimulated workers to search for other regions within the E. ruminantium genome as potential targets for better tests to characterize different strains. One such genomic target was identified by comparing three whole genome sequences of E. ruminantium to identify genomic regions which appeared to be mutational hotspots.329 Within these regions six complete coding sequences (CDSs) and four truncated CDSs were identified as suitable targets for differential diagnosis, and PCR amplification of these targets yielded strain-specific patterns of differently-sized DNA fragments visualized by electrophoresis.
Another technique, based on detecting the polymorphism of variable number tandem repeat (VNTR) regions, is named multi-locus VNTR analysis (MLVA).255 PCR amplification of these targets yielded different patterns of DNA fragments, but not all the strains examined showed a unique pattern, with 10 different profiles being obtained from 13 different strains. In particular a virulent and an attenuated strain of the same original isolate could not be distinguished. The MLVA scheme does have advantages, however, allowing typing of the pathogen directly from field samples without preliminary culture.
Some studies have used multilocus sequence typing (MLST), usually targeting multiple core function genes. In one case 12 different strains of E. ruminantium isolated in different areas of Africa and the Caribbean were examined for 8 genes: 16S rRNA, gltA, groEL, ftsZ, sodB, nuoB, rnc, and ctaG.11 Phylogenies inferred from the individual sequences of the different genes showed different branching orders for the different strains. Concatenation of the sequences followed by phylogenetic analysis,139 indicated an overall segregation of the genotypes into two main clades, one from west Africa and the other from eastern plus southern Africa. The one Caribbean strain fell in the eastern/southern clade with low bootstrap support, and one strain of southern African origin fell in the west African clade with solid bootstrap support. It was concluded that, contrary to all expectation for an obligately intracellular organism, extensive recombination takes place between different E. ruminantium genotypes in the field.
Two other MLST studies used a slightly different range of target genes: gltA, groEL, lepA, lipA, lipB, secY, sodB, and sucA3, 218 and in each case evidence was found of recombination between different E. ruminantium genotypes in the field.
The most recent MLST study uses the largest dataset so far examined, looking at 194 strains for 5 core function genes: lipA, lipB, secY, sodB, and sucA.67 Concatenation of the sequences showed 97 unique genotypes, which were analysed with a wide range of phylogenetic and population structure tools.55, 89, 119, 138, 264 The data divided into two main population groups, genetically well separated: Group 1, strains of mostly western African origin; Group 2, strains from the rest of the world. Group 2 was divided into 5 subgroups, more closely related to each other than to Group 1, and included most of the southern African, eastern African, Caribbean and Indian Ocean island strains, and some of western African origin. The separation between Groups 1 and 2 appears to predate the introduction of domestic ruminants into sub-Saharan Africa, with the two ancestral E. ruminantium populations originating in western Africa, and southern and/or eastern Africa, respectively. The mixed phylogeographical structure of Group 2 correlates with relatively recent historically documented cattle movements. Finally, and most importantly, there was statistically well supported evidence of extensive genetic recombination within and between all the groups.
Horizontal gene transfer (HGT) is a mechanism used by many free-living bacteria to acquire genes from other species of bacteria, thereby increasing their genetic capabilities and thus their evolutionary fitness.208 In contrast no HGT events from other species have been confirmed in any genome of E. ruminantium. Multiple E. ruminantium 16S genotypes are often seen in blood samples from animals in the field, but ticks removed from the same animals rarely harbour more than one genotype,16 and it seems that this may be due to infection exclusion.82 Hence possibly the only time in its life cycle when E. ruminantium could acquire new genes would be the 15 day period which elapses in the tick after full engorgement and before infection of the gut epithelial cells becomes established.165 This possibility requires investigation, but whatever the mechanism may be it is now certain that the phenomenon is commonplace for E. ruminantium, and must be a major factor enabling the organism to mutate and flourish despite attacks from its hosts’ immune systems.
There are many E. ruminantium strains circulating in the field which differ in their infectivities,367 pathogenicities,16 and cross protective immunogenicities.4, 73, 156 The consequences of widespread recombination between strains will make the development of a durable vaccination strategy quite difficult even when an effective vaccine becomes available. Preventing the organism from spreading, insofar as this may be possible, will need to be a component of any control strategy.
Table 4 Complete and draft genome sequences of Ehrlichia ruminantium19
| Strain | Assembly Level | Date | Size (Mbp) | per cent GC | Accession No. | Genes | Proteins |
| Welgevonden | Complete annotated genome | 2003 | 1.516355 | 27.50 | NC_005295.2 CR_767821.1 | 990 | 936 |
| Gardel | Complete annotated genome | 2005 | 1.499920 | 27.50 | NC_006831.1 CR_925677.1 | 986 | 925 |
| Welgevonden | Complete annotated genome | 2005 | 1.512977 | 27.50 | NC_006832.1 CR_925678.1 | 994 | 930 |
| Senegal virulent | Single scaffold | 2017 | 1.45538 | 27.50 | WGS - MQUJ01 | 972 | 867 |
| Senegal p63 | Single scaffold | 2017 | 1.45538 | 27.50 | WGS - MRDC01 | 973 | 867 |
| Palm River | 368 contigs | 2016 | 1.48588 | 28.10 | WGS - LUFS01 | 997 | 923 |
| Crystal Springs | 34 contigs | 2016 | 1.48117 | 27.50 | WGS - BDDK01 | 992 | 915 |
| Kerr Seringe | 118 contigs | 2016 | 1.45366 | 27.50 | WGS - BDDL01 | 996 | 867 |
| Pokoase 417 | 390 contigs | 2016 | 1.46506 | 27.50 | WGS - BDDM01 | 1049 | 819 |
| Sankat 430 | 183 contigs | 2016 | 1.4578 | 27.50 | WGS - BDDN01 | 1005 | 846 |
Notes: Downloaded from GenBank 22/08/2017, subject to regular updates.
WGS numbers are ongoing whole genome projects.
Genomics
The genome of the Welgevonden strain, the type specimen of E. ruminantium, was the first to be completely sequenced and annotated72 and the data are available from GenBank (NCBI Reference Sequence NC_005295.2). There are two additional completely sequenced and annotated E. ruminantium genomes,126, 127 and seven further genomes in various stages of sequencing.217 Some features of these genome sequencing projects are summarised in Table 4.19 The second and third genomes to be completed are those of the Gardel strain, isolated on Guadeloupe325 (Table 1), and a sample of the Welgevonden strain which was transferred to Guadeloupe from South Africa in 1985 and then maintained for 18 years in tissue culture.126 These two genomes are both somewhat smaller than that of the type strain (1,516,355 bp): Gardel is the smallest (1,499,920 bp), while the Guadeloupe-cultured Welgevonden is intermediate in size between the other two (1,512,977 bp). All three genomes have the relatively low G+C content of 27.5 per cent, a feature which is common for endosymbionts and intracellular pathogens.
A striking feature of all three E. ruminantium genomes72, 126 is the large number of tandemly repeated and duplicated sequences, amounting to 8.3 per cent of the genome. This contributes to the unusually low proportion (62 per cent) of coding sequence for a small intra-cellular parasite. In the course of the sequencing projects it was observed that the copy numbers of many of the repeats were actively variable, with the addition or deletion of repeats occurring primarily in noncoding regions, but also within CDSs. The repeats have evidently mediated numerous translocation and inversion events which have resulted in the duplication of some genes, and the truncation of others. Most of the pseudogenes in the genome appear to be the products of sequence duplication events, some of which have also given rise to new genes. Some of the small repeats are in or near promoter regions, and may be involved in promoter regulation. Twenty seven genes which contain long repeats are predicted to encode for membrane-associated proteins, or are genes unique to E. ruminantium, which suggests that they play important roles in the biology of the organism.
A detailed comparison made between the three complete genomes127 shows that they are very similar to each other, with a total of 888 orthologous CDSs present in all three genomes. Different annotation criteria were used for Gardel and the Guadeloupe-cultured Welgevonden, as compared to the type strain Welgevonden, so it was difficult to make entirely accurate comparisons of all the CDSs. However there appear to be no substantive differences in the open reading frames (ORFs) of the two Welgevonden strains, which each contain 35 CDSs which have no orthologs in Gardel. Twenty two Gardel CDSs have no orthologs in the two Welgevonden genomes. Four CDSs were found to be mutated in the Guadeloupe-cultured Welgevonden, as compared to the original Welgevonden, but these mutations did not appear in Gardel, suggesting that the mutations had occurred during the 18 years (11–13 passages) spent under cell culture conditions which differed from those of its parent strain. An analysis of the synonymous versus non-synonymous (dS/dN) substitutions per site in Gardel, as compared to the two Welgevonden strains, showed only three CDSs with a dS/dN ratio biased towards non-synonymous substitutions; two of the three are annotated as pseudogenes. The dS/dN ratios for the rest of the homologous CDSs were biased towards synonymous substitutions, indicative of mutation constrained by the need to conserve protein function. In summary the authors described the E. ruminantium genome as being unexpectedly ‘plastic’.
A separate comparative genomics study of the three E. ruminantium genomes148 calculated their divergence times, using an estimate of synonymous substitution rates for intracellular bacteria, as being 26,500–28,500 years ago (ya) for the two Welgevonden genomes, and 2.2 Mya for the last common ancestor of all three. Considering that the two Welgevonden genomes were only separated 18 years before sequencing,126 and that heartwater was transferred from Africa to the Caribbean not earlier than the 18th century,29, 190 it appears that E. ruminantium undergoes accelerated mutation rates following exposure to changes of environment. Experimental evidence for this was obtained quite incidentally during a transcription analysis of the map1 gene family.31 Two different Gardel samples were examined: one had been sent from Guadeloupe to Portugal and had undergone 45-66 passages in bovine pulmonary artery endothelial cells; the other had been sent from Guadeloupe to the UK and had undergone 12-15 passages in bovine aorta endothelial cells. Sequence analysis showed that the sample which had passed via the UK, had undergone a rearrangement of part of the map1 genomic region in which the map1-2 and map1-3 genes had recombined at a 14 bp site common to the two genes, resulting in a single hybrid gene and deletion of the original map1-2 gene. Recalling also that four genes were found to be mutated in the Guadeloupe-cultured Welgevonden as compared to the original Welgevonden126 it appears certain that E. ruminantium is prone to an unusually high mutation rate when in culture. In addition to the unexpected genomic plasticity is the readiness with which different genomes undergo recombination, revealing a parasite capable of unexpectedly rapid evolution in order to remain competitive in its struggle to survive.
Transcriptomics and proteomics
The first studies to look at transcription in E. ruminantium investigated the map gene family, which consists of 16 paralogs bounded by a hypothetical transcriptional regulator upstream and secA downstream.340 Purified mRNA, prepared from a Welgevonden-infected bovine endothelial cell line, was subjected to reverse transcription using PCR primers specific for all 16 paralogs, and all were found to be transcribed.
Since E. ruminantium survives and grows in mammalian and acarine hosts it would be expected that the required adaptations should be reflected in differential gene transcription and expression in the different environments. Work to investigate this possibility used three strains of E. ruminantium (Gardel, Senegal and Welgevonden) cultured in cells of bovine, heartwater-vector tick, and non-heartwater-vector tick, origin.31 Again transcripts for all 16 paralogs were detected in bovine cells, but the situation was different in tick cells. In the Am. variegatum vector cell line 10 transcripts were seen, with map1-1 being the most predominant. The number of transcripts detected in non-vector cell lines ranged from 4 in Rhipicephalus appendiculatus to 11 in Ixodes scapularis, and again map1-1 predominated. Over all the tick cell lines map1-11 and map1-3 transcripts were never detected and map1-1 and map1+1) were always found to be transcribed.
Extending these studies to live Am. variegatum ticks showed different patterns of map transcription in feeding and unfed ticks. In unfed ticks only map1-1 transcripts were detected, and then only in midguts and not in salivary glands. In feeding ticks, on the other hand, map1-1 transcripts were detected more abundantly, although still only in the mid gut, while map1 transcripts were now abundant, but only in the salivary glands.259 This work was confirmed using two-dimensional gel electrophoresis and mass spectrometry to identify MAP proteins in cell extracts from E. ruminantium-infected cells of bovine and Am. variegatum origin: only MAP1 was found in the bovine cells, and only MAP1-1 in the tick cells.258 It is evident that different map genes and MAP proteins are important to E. ruminantium during different stages of the life cycle, although their exact functions are not yet known.
A more comprehensive transcription study used microarrays to investigate differential gene expression at each life cycle stage of E. ruminantium grown in bovine cells.273 E. ruminantium mRNA was obtained free from contaminating host RNA and rRNA by selective capture of transcribed sequences.114 Microarray analysis and estimation of transcript levels by quantitative reverse transcriptase PCR revealed a total of 54 genes which were differentially expressed between the EB and RB growth stages. In RBs transcripts essential for energy production and for the exchange of nutrients between the host and E. ruminantium were up-regulated, confirming high metabolic activity in this predominantly proliferative stage. In EBs, which is the infective stage, up-regulated transcripts included those which would be expected to be important in host-pathogen interaction. These included genes involved in metabolism, nutrient exchange, and resistance to oxidative stress, which would enhance survival under oxidative stress and nutrient starvation conditions. These are known to occur in E. canis-containing vacuoles81 and could well occur during E. ruminantium infection also. Also up-regulated in EBs are genes involved in defence mechanisms, and genes for outer membrane proteins and virulence factors.
A partial proteome map of EBs of the Gardel strain of E. ruminantium in endothelial cell culture was constructed using two dimensional gel electrophoresis combined with mass spectrometry.191 Among the forty-eight different EB proteins identified only four were from MAP gene family, with MAP1 being the most abundant. Other proteins detected were related to energy metabolism, general metabolism, amino acid synthesis, post-translational modification, protein turnover, chaperone synthesis, and virulence factor translocation. A comparison of the proteomes of virulent and attenuated versions of the Gardel strain in cell culture showed that they shared 80 per cent of their proteins.195 As might be expected, the virulent strain showed up-regulation of proteins involved in pathogenesis, while the attenuated strain was geared for maximum replication, evident from increased expression of proteins involved in metabolism and protein and nucleic acid biosynthesis.
A proteomic study concentrating on the OMPs of E. ruminantium EBs has been carried out in view of their possible importance for vaccine design.211 Cell extracts were processed to enrich the OMP fraction, the proteins were separated by electrophoresis, digested, and the peptides were analysed by liquid chromatography coupled with mass spectrometry. Forty six unique proteins were identified, of which 18 were OMPs, including 4 MAP proteins not identified previously (MAP1-6, MAP1-13, MAP1-14 and MAP1+1). There were also eight proteins involved in cell structure and biogenesis, four in transport and virulence, including three components of a type IV secretion system (VirB9-1, VirB9-2, and VirB10), one was a porin, and five were proteins of unknown function. The most likely sub cellular location of these proteins was assessed from a consensus of the results from three different localisation prediction algorithms: it appeared that 18/46 were OMPs, 5/46 were inner membrane proteins, and 23/46 were cytoplasmic.
The genomics, transcriptomics and proteomics studies on E. ruminantium are the most likely means whereby the identification of previously unknown proteins involved in pathogenesis may be identified. Such proteins should be good candidates for inclusion in vaccines, a topic which will be dealt with in more detail below under Vaccines.
Control
Four methods are currently available for the control of heartwater: tick control, antibiotic treatment of clinical cases, prophylactic use of antibiotics, and immunization.
Tick control
Tick control has been advocated as a means of controlling heartwater from very early days.6, 88, 309 Even after the infected blood-based vaccine was developed, tick control was still advocated as a supplementary or alternative means of control.144, 231, 299
Tick control can be either intensive or strategic (see Vectors: Ticks).43 The objective of intensive was to control all stages of ticks throughout the year, and it was a system advocated for marginal areas where Amblyomma spp. were only found occasionally. The logistics and expense of such operations are formidable if they are applied over large areas, and acaricide resistance is a widespread problem.323 The main disadvantage, however, is that animals may lose all immunity to tick-borne diseases because of the lack of a natural challenge. Any breakdown of the intensive control regimen then results in heavy losses from heartwater and other tick-borne diseases. For these reasons intensive tick control has largely fallen into disuse.
Strategic tick control implies the control of tick numbers so that natural infection of livestock occurs and high levels of immunity are maintained. The aim is to achieve an epidemiologically stable situation with respect to heartwater by the regulation of the numbers of ticks present so as to prevent the debilitating effects of severe tick infestations. It is usually recommended that animals should be dipped only if they are carrying, on average, more than 10 adult Amblyomma ticks each. The animals should be monitored weekly in summer and autumn immediately after or during periods of good rains, and every two to three weeks in winter. It has been shown that this approach can lead to endemic (or enzootic) stability, even when the strategy is somewhat erratically applied.312 Economic studies have demonstrated that strategic tick control is both a more economical and a more practical option for limiting losses from heartwater and other tick-borne diseases.207 In fact, the counter-intuitive observation has been made that direct tick-borne disease losses increase with increasing use of acaricides.68
Antibiotic treatment
Several drugs have been used to treat animals suffering from heartwater, but the tetracyclines, especially oxytetracycline, are the most widely used.332 During the early 1950s the minimum recommended therapeutic dose was 5 mg/kg live weight, given either as a single dose or in a divided dose with a 24-hour interval.143, 349 Some years later it became apparent that this dosage rate was no longer effective and a rate of 10 to 20 mg/kg was recommended. Despite this it is said that there is apparently no resistance of E. ruminantium to tetracyclines.
Short-acting formulations of oxytetracycline are most commonly used at a dosage rate of 10 to 20 mg/kg body weight, either administered intramuscularly as a single dose, or half the calculated dose is given intravenously and the other half intramuscularly. This treatment is usually repeated 24 hours later. A long-acting oxytetracycline preparation has been shown to be equally effective.322 Doxycycline has been used successfully at a dose rate of 2 mg/kg body weight in the treatment of experimentally induced heartwater in sheep.153 This formulation is more lipid-soluble and has a longer serum half-life than has oxytetracycline, and smaller therapeutic doses with longer intervals between treatments can thus be used.
When treatment with tetracycline is instituted during the incubation period of heartwater in cattle (before approximately eight days after infection) the course of the disease is usually altered; animals usually develop no fever or other clinical signs, except that, in a few, a low-grade transient febrile response will result. In these animals no, or at best only a partial, immunity develops.107 Treatment of sheep and goats during the incubation period may also give rise to a delayed febrile reaction (it may be as long as 20 to 25 days after infection compared to the more usual 9 to 10 days in untreated animals), and the reaction may be so severe that additional treatment is required.256, 257
A wide variety of sulphonamides has been used in the treatment of heartwater,7, 142 and in fact, the treatment of heartwater with uleron by Neitz in 1940223 was the first use of any sulphonamide against a rickettsial infection. Sulphadimidine gives good results in severe reactions to heartwater that persist in spite of repeated oxytetracycline treatments338 but because tetracyclines are normally so effective sulphonamides are not commonly used. Rifamycin has also been used to treat E. ruminantium infections in mice and sheep at a dosage rate of 0.2 mg/kg.240
Supportive therapy in clinical cases of heartwater is often inefficient because of the poor understanding of the pathogenesis of the disease. Various anti-inflammatory agents have been used but the only published information concerns the use of dimethyl sulphoxide50 and prednisolone.275 Calves develop halitosis and dark yellowish-brown urine as a consequence of the use of dimethyl sulphoxide but no real improvement in the recovery rates occurs.140 Betamethasone can be given at a dose of 0.1mg/kg body weight and treatment can be repeated after 12 hours if necessary.
In addition to specific therapy, diuretics have been administered to counteract oedema, and in the past furosemide was used for this purpose.129 However, the drastic reduction in blood volume which occurs as a consequence of heartwater is exacerbated by the use of diuretics, and the administration of furosemide as a supportive treatment is no longer recommended.289
Prophylactic use of antibiotics
Routine oxytetracycline injections may be used to protect susceptible animals against heartwater when they are introduced into an endemic area.134, 275 In goats it is advocated that short-acting oxytetracyclines be administered at a dosage rate of 3 mg/kg body weight on days 10, 20, 30, 45 and 60 after their introduction, and that the animals should not be dipped until day 60.134 Injections of a long-acting oxytetracycline in cattle are sufficient to protect them from contracting heartwater, while at the same time allowing them to develop a natural immunity.275, 276 The dosage is 10 to 20 mg/kg body weight given on days 7, 14 and 21, or days 7, 12 and 17, or even on days 7 and 14. The animals should be kept under close scrutiny and given appropriate treatment if they do develop overt disease. The success of this regimen depends upon the animals becoming naturally infected during the time that they are protected by the drug.
Slow-release treatment with doxycycline has also been advocated, in the form of a tablet implanted behind the ear. In practice this is most often used as part of an immunization and treatment regimen.170 The dosage is critical so the body weight of the treated animals must be known, since an inadequate amount of antibiotic will not prevent fatal heartwater.
Immunization
The economic need for a control system based on vaccination, and the steps being taken to develop a suitable vaccine, are described in more detail below (see Immunity, Vaccines).
The only ‘vaccine’ currently commercially available is a cryopreserved preparation of blood from sheep infected with virulent E. ruminantium organisms of the Ball 3 strain.236 The blood is injected intravenously into animals to be immunized, the rectal temperature is monitored daily, and antibiotic treatment is administered at the proper time. The infective blood must be preserved on dry ice or in liquid nitrogen and thawed shortly before inoculation, and the whole procedure must be supervised by trained staff. The duration of immunity is uncertain, and because live organisms are involved the procedure cannot be used in non endemic areas. The procedure is, however, successfully used to protect susceptible animals against the disease, especially when they are first introduced into endemic areas, or if they are particularly valuable.
The Ball 3 strain was originally isolated in the Limpopo Province of South Africa141 and was chosen as the vaccine strain because it produces an early temperature rise several days before any other serious clinical signs appear. This makes it relatively easy to decide when to treat. Some strains, such as the highly virulent Welgevonden strain, can cause death very shortly after a rapid temperature rise and are therefore not usable for the infection and treatment method of immunization. Unfortunately the Ball 3 vaccine does not protect against all the strains which circulate in the field. It confers only limited or no protection against challenge with the Welgevonden,110 Senegal,157 and Gardel strains.73 It is noted above (see Genetic Variability) that the Welgevonden strain offers a wider spectrum of cross protection than other strains (Table 2),73 but its virulence makes it difficult to control and therefore unsuitable for an infection and treatment immunization procedure.
Immunization of pregnant cows, especially if their offspring are valuable, is not advisable as it may result in the peracute form of the disease, or abortion.338 Uncomplicated immunization of ewes that were two to four months pregnant has been reported42 but in such instances it is very important to monitor the rectal temperatures carefully and to treat the animals at the first rise in temperature.
Two to five per cent or more of animals, especially cattle that supposedly should have lost their age-related resistance, fail to react after immunization.101 This could be due to persistence of the innate age resistance in some animals102 or to high conglutinin levels. Reimmunization of non-reactors sometimes produces the desired effect, but the risk of anaphylactic shock increases with repeated inoculation of blood or tick suspensions.27
In the absence of periodic stimulation of the immune system, resulting from the bites of infected ticks, the duration of immunity after immunization varies greatly between different domestic animal species, and also perhaps between individuals within species. In sheep, the immunity may wane after six months95, 222 but in some cases it may remain sufficient to protect animals against a fatal outcome for at least four years.228
The duration of immunity in goats following immunization is poorly documented. In Angora goats the degree of immunity seems to depend largely on the time at which the animals are treated therapeutically during the reaction subsequent to the immunization. In one experiment, treatment shortly before the commencement of the febrile reaction resulted in the development of a poor immunity, while animals treated on the second and third days of the reaction were immune to challenge 107 to 205 days later.104
The duration of immunity in cattle after immunization in the absence of challenge appears to be approximately two years.101 It has been suggested that some animals may be susceptible again as soon as two months after immunization21 but this observation could have been the result of infection with an immunotype against which Ball 3 does not offer protection.
Immunization of very young animals
This procedure takes advantage of the age-related innate resistance of young animals to heartwater.107, 227 Immunization of calves under the age of one month and of lambs and kids less than seven days old does not generally result in clinical disease, but the animals do develop immunity. While it is advisable to monitor their rectal temperatures twice daily for a period after immunization, as is advised in the case of older animals, this is still the method with the lowest risk of losses due to fatal heartwater reactions after inoculation.102
Practical aspects of infection and treatment method
After the blood vaccine is administered to an animal a definite fever usually develops and if this is not treated it may be fatal. Inoculated animals are treated early in the course of the disease, but if treatment is effected too early no immunity will result. There is no indication that the administration of tetracyclines interferes with the development of immunity to heartwater294 as long as they are not given too early in the incubation period.107 It is advisable to immunize individuals or small groups of animals because large groups present serious management difficulties.
Starting on the day following vaccination, the rectal temperature of each animal should be recorded daily, preferably early in the morning and before the intake of food. In order to identify the first day of the temperature reaction the average normal temperature preceding the reaction must also be determined over a period of about six days for both small stock and cattle. The first day of the temperature reaction can be expected 9 to 14 days after inoculation in the case of small stock, and after 14 to 18 days in cattle, when the rectal temperature will usually be 1°C or more above the average. In exceptional cases this could occur as early as seven days in small stock and as long as 25 to 30 days in cattle. A rise in the early morning rectal temperature to above 39.5°C in cattle and goats, and 40°C in sheep, is usually regarded as an immunization reaction.339 The general condition and behaviour of all animals should, in addition to the temperature reaction, be closely monitored after vaccination.
It is unnecessary to treat as early as the first day of the reaction, except in rare cases where, after a sudden rise of 1.5°C or more, a temperature of 41.5°C is reached. Treatment should be given only if the temperature on the second day of the reaction equals or surpasses that on the first day. Should the temperature on the second day be lower than the previous day the animal is either resistant or partially immune, in which case the temperature on the third day will again be lower than on the first day and treatment would be unnecessary. Occasionally the temperature on the third day is much higher than on the second day and even higher than on the first day. If this occurs the animals should be treated immediately.
Treatment consists of an intramuscular injection of tetracycline at 10 mg/kg body weight, and short- or long-acting formulations can be used. The recording of temperatures must be continued after treatment, and if the temperature 48 hours after treatment equals or surpasses that on the day of treatment a second treatment should be given. A high temperature within the first 24 hours after treatment can be ignored, unless the animal shows other clinical signs such as listlessness and lack of appetite, in which case it should be treated a second time without delay.
Relapses of vaccine-induced heartwater may occur after treatment. It is therefore advisable to monitor the rectal temperatures, especially of valuable animals, for an additional period of two weeks after the last treatment.20 A small number of animals may react severely despite the lack of a fever, and other diseases, such as babesiosis, may complicate matters.227 Blood smears (and possibly other examinations) should be performed when animals do not respond satisfactorily to specific heartwater therapy.
The block method of immunization
The block method of immunization,107, 124, 256, 257 also known as ‘systematic treatment’,20, 322 is widely practised in South Africa, especially when immunizing large numbers of kids and lambs. In this method the vaccine is administered normally but is followed up by treatment, as described above, on a predetermined day, without recording daily rectal temperatures. Treatment is recommended on the following days, assuming that vaccination has taken place on day 0: exotic Bos taurus cattle breeds and their crosses, day 14; indigenous Bos indicus cattle breeds and their crosses, day 16; sheep and Angora goats, day 11; Boer and crossbreed goats, day 12.107
This procedure makes immunization on a large scale possible and it is particularly suitable for the immunization of large groups of animals being moved from a heartwater free area into an endemic area. The animals can be immunized on the farm of origin or immediately after their arrival in the heartwater area. Care must be taken, however, as the procedure has some disadvantages. In older animals durable immunity only follows after the animal has developed a febrile reaction.6, 107 Treatment performed too early in the incubation period results in failure to develop immunity,107 and if treatment is given too late heavy mortalities can result.104 In exceptional cases some animals may be more susceptible than the rest of the herd and may show clinical signs before they are treated.
A prudent modification which may be applied to the block method is to select 10 animals, representative of the breed and age of the herd, and to record their daily rectal temperatures in the usual way. As soon as the temperature of five or more of them is above the average for two consecutive days, the whole herd is treated.
The doxycycline implant method
In this method animals are inoculated and at the same time doxycycline tablets are implanted under the skin, usually behind the ear, using a special applicator. Slow release of the active ingredient means that the animal does not become clinically ill, but at the same time develops an immunity.170 Since the animal is handled only once, and daily temperatures are not recorded, this method is suitable for the immunization of large numbers of animals at one time. The dosage is critical, so the body weight of the treated animals must be known, since an inadequate amount of antibiotic will not prevent fatal heartwater, while an excess will prevent the subsequent development of immunity.
Immunity
It is known that E. ruminantium causes down regulation of interferon (IFN)-γ production and inhibition of major histocompatibility complex (MHC) expression.330 This would be expected to slow the development of immunocompetent cells and, although no E. ruminantium specific cytotoxic CD8+ T cells were demonstrated, it was known that CD8+ T cells proliferate in animals vaccinated with inactivated E. ruminantium and then challenged with virulent organisms.197, 316 It is possible, therefore, that if T-cell proliferation is slowed until many endothelial cells are infected the subsequent specific killing of these cells could lead to blood vessel permeability. Against this hypothesis, however, is the observation that both infected and uninfected endothelial cells appear to be severely damaged in ruminants which die from the disease.253
The earliest work on the immune response to E. ruminantium consisted of attempts to transfer immunity from heartwater immune cattle to naive animals by transferring serum.6, 90, 91 These attempts failed, indicating that antibodies do not control the course of the disease, although antibodies may still play a role in opsonization, complement-mediated killing or antibody-dependent cell-mediated cytotoxicity.
The antigen of E. ruminantium which is currently the best characterized is MAP1, an immunodominant OMP156 of about 32 kDa,343 a size which varies from strain to strain.25 The protein is strongly identified by hyperimmune antiserum on Western blots of total E. ruminantium proteins282 and is also recognized by CD4+ T lymphocytes from immune animals.216 MAP1 has been used to develop serological assays (see Diagnosis: Serological Methods), and has been tested as a potential vaccine (see below). A second immunodominant OMP gene (map2) has also been identified183 and MAP2 was evaluated for use in serological diagnostic assays.49
As E. ruminantium is an intracellular parasite it would be expected that both CD8+ cytotoxic T cells and CD4+ helper T cells would be important in the development of protective immunity. The first confirmation that CD8+ T cells play a major role in protection came from adoptive transfer studies in a mouse model.96, 100, 103 It has now been shown that cattle immune to heartwater generate lymphocyte responses characterized by a mixture of CD4+, CD8+ and γδ T cells, and the expression of T helper type 1 cytokines.197, 215, 316 When PBMCs from heartwater-immune cattle were exposed in vitro, to either autologous infected endothelial cells or infected monocytes, PBMC proliferation was observed.215 Under the same conditions, however, E. ruminantium EBs did not induce PBMC proliferation.215 The endothelial cells required pre-treatment with T-cell growth factors to induce class II MHC expression prior to infection and their subsequent use as stimulators of PBMC.215 Cells from immune cattle have also been shown to respond specifically to recombinant antigens of E. ruminantium, CD4+ T cells responding to rMAP1, and γδ T cells responding to rMAP2; both proteins induced a T-helper type 1 immune response.216
Cattle immunized against heartwater with an inactivated lysate of E. ruminantium in Freund’s adjuvant were resistant to a virulent challenge 3 and 10 months later. Cell lines were generated by in vitro re-stimulation of PBMC from the cattle with E. ruminantium lysates. These cell lines were found to be MHC class II restricted IFN-γ-producing CD4+ T lymphocytes, and they responded strongly to whole E. ruminantium lysates but not to rMAP1 or rMAP2 proteins.316
Cells which did respond specifically to rMAP1 and rMAP2 were found to be γδ T-cell lines. When E. ruminantium-specific CD4+ T cell lines were stimulated with soluble E. ruminantium proteins, purified by fast performance liquid chromatography, a single peak, which included proteins between 20 and 32 kDa, induced proliferation.116, 313 The size ranges of the proteins causing this response were further defined in a separate study; PBMC were taken from cattle immunized either by infection and treatment or with inactivated organisms and exposed to E. ruminantium proteins size-fractionated by continuous-flow electrophoresis. Cells from animals immunized by either procedure proliferated specifically to protein peaks of 11, 12, 14-17 and 19-23 kDa.342
While flow cytometric analysis of PBMC has not shown any significant change in the immune cell population after vaccination with inactivated organisms, significant changes do occur when the animals are challenged. Initially there is a depletion of CD4+, CD8+ and γδ T-cell subsets, and a rise in numbers and activation of monocytes, and only later, when the animal is fighting the infection, is there an increase in CD8+ T lymphocytes.197
Immune cattle produce IFN-α when subjected to a lethal E. ruminantium challenge, while non-immune animals, similarly challenged, die without producing this cytokine.315 Even so IFN-α alone could not completely inhibit the in vitro growth of E. ruminantium, suggesting that other factors are required for complete inhibition. The type 2 cytokine IFN-γ has been shown to be a very potent inhibitor of E. ruminantium growth in endothelial cells, but the mechanism of this inhibition is unknown.313 Another important role played by IFN-γ is the upregulation of MHC class I and II expression on various cell types75 and, when monocytes are affected, this will lead to enhanced presentation of E. ruminantium antigens to the immune system. A study using PBMC from goats immunized with killed E. ruminantium showed that both the CD4+ and CD8+ subsets produced IFN-γ after stimulation with crude E. ruminantium antigen.116, 117, 118 Blocking experiments have also confirmed that CD8+ T cells require the help of CD4+ T cells in order to produce IFN-γ.118 There have, however, been mixed results concerning the production of IFN-γ in vaccinated animals; it has been reported that in goats IFN-γ could only be detected in the vaccinated animals after antigenic recall in vitro, while the control animals did not produce the cytokine.118 Other studies have shown that the production of IFN-γ is variable in both immunized and control animals and cannot be used on its own as an predictor of host survival after challenge or to measure vaccine potency.260, 262, 328
Ehrlichia ruminantium has been shown to elicit de novo synthesis of interleukin mRNA (IL-1β, IL-6 and IL-8) in bovine brain endothelial cells in vitro, an effect which was potentiated by IFN-γ.47 Since IL-1 and IL-6 can act as co-stimulatory signals for T- and B cell activation348 it is possible that, by secreting these cytokines, brain endothelial cells may contribute to protective immune responses. Antigens of E. ruminantium capable of inducing the production of these cytokines by immunocompetent cells should be investigated as potential vaccine antigens.
Vaccines
An economic model for the prevalence of heartwater and its impact under various farming systems has been developed as a result of a 10-year study in Zimbabwe.213 The estimated annual losses in that country amounted to US$ 5,6 million, with economic losses per animal in the commercial production sector being 25 times greater than the losses in the communal and traditional farming sectors, largely as a result of the cost of acaricides. The conclusion was reached that a control strategy based on a vaccine would be the most cost effective way of combatting the disease.213
Attenuated vaccines
During the last two decades molecular genetic research has made enormous contributions to a better understanding of E. ruminantium and heartwater, with a major goal being the development of a defined DNA- or protein-based vaccine. These research developments have not yet yielded any viable products and the only commercially available procedure for heartwater immunization is an infection and treatment technique, the practical details of which are described in detail above (see Control: Immunization). This technique was developed more than 40 years ago at the Onderstepoort Veterinary Institute, South Africa236, 339 and it has a number of drawbacks. The logistics of the procedure are particularly inappropriate for use in rural areas in Africa or the Caribbean because of the need for continual frozen storage facilities339 and both the production of the infective material used as antigen, and the methods required for its application, are expensive. As always with a live vaccine there is the concern about a possible increase in virulence, made more likely by the propensity of E. ruminantium for genetic recombination (see Diagnosis: Molecular Genetic Methods). Finally the use of attenuated vaccines is always deprecated for use in areas where the genotype used does not already occur. Yet despite all this, vaccination with live attenuated organisms against heartwater is still a cost-effective control strategy.290
The first E. ruminantium organism to be attenuated was the Senegal strain, which became attenuated after 64 serial in vitro passages organisms in bovine endothelial cell cultures.154 In sheep the attenuated Senegal vaccine confers 100 per cent immunity to homologous needle challenge,154 while in field trials against a heterologous natural tick challenge the protection levels are lower. In Senegal 57 per cent of vaccinated sheep were protected against natural challenge,135 and in the Gambia 75 per cent of vaccinated sheep were immune to a natural tick challenge.120 The Gardel strain became attenuated after >200 passages in bovine endothelial cell cultures,198 but there are no records of it being used as an experimental attenuated vaccine. There have been investigations into the proteomic differences between the attenuated and virulent strains, which show that they are optimized for maximum replicative capacity and maximum virulence, respectively, but no prominent antigenic differences came to light.195
The lack of cross immunity between different strains of E. ruminantium is discussed above (see Genetic Variability) and is assumed to be at least part of the reason for the poor protection seen in field trials of experimental vaccines. The Welgevonden strain is known to provide cross-protection against a needle challenge with a range of other virulent strains (Table 2) and this could make it a better candidate for use in an attenuated vaccine. Unfortunately this strain was resistant to attenuation during hundreds of bovine cell culture passages over several years135, 368 but it was eventually attenuated by propagation in a canine macrophage-monocyte cell line (DH82).361 Adult Am. hebraeum ticks fed as nymphs on sheep immunized with these organisms at passage 111 transmitted the attenuated strain to a naive sheep which was subsequently protected against a lethal homologous needle challenge.366 DH82 cells are cancerous and not suitable for vaccine use, hence the attenuated strain was re-adapted to bovine endothelial cells before being used at passage 44-64 as an experimental vaccine.366 Merino sheep and Boer goats, vaccinated intravenously, were 100 per cent protected from a lethal needle challenge with the homologous strain or with one of four different heterologous strains (Ball 3, Gardel, Mara 87/7, Blaauwkrans). The protection persisted at 100 per cent for at least 6 months, but had fallen to 80 per cent after one year,364 although under normal field conditions with a continuous natural challenge it is probable that the 100 per cent immunity level would have been retained. When used to vaccinate Angora goats, which are exceptionally susceptible to heartwater,104 the vaccine induced mild heartwater symptoms, and 90 per cent of the animals were subsequently immune to homologous challenge.364 The same vaccine was used to immunize Friesian cattle by intravenous injection and they were subsequently needle challenged with the Gardel strain; 5/6 cattle recovered without treatment, 1/6 had to be treated due to a deteriorating health status.365 The challenge was performed with the heterologous Gardel strain, which is highly virulent in cattle, since the Welgevonden strain is of low virulence in cattle.8 This experimental vaccine appears to be a highly promising candidate for commercial distribution in southern Africa, where the Welgevonden strain, and closely related strains, are in wide circulation.
Inactivated vaccines
Inactivated vaccines consist of an extract of whole disease-causing organisms, usually derived from tissue culture, which have been rendered non-viable by chemical treatment. The first inactivated heartwater vaccine trial was in goats using the Gardel strain, and 50-80 per cent of the animals were protected against a homologous needle challenge which killed 100 per cent of the negative controls.200 The next report used the Crystal Springs strain in sheep, and in this case 50-100 per cent of the animals were protected against a homologous needle challenge which killed 60 per cent of the negative controls.181 There have been more recent trials of inactivated vaccines designed to optimize the numerous operative parameters: one study was carried out to evaluate the minimal necessary protective dose of vaccine;328 another investigated the effect of the purification process and storage conditions on the efficacy of the vaccine;194 and another looked at the effectiveness of inactivated Gardel vaccination in the field in Burkina Faso, where the protection rate was relatively poor (65-72 per cent); this appeared to be the result of the extensive genetic diversity of the strains in the natural field challenge.4
Inactivated heartwater vaccines can normally be stored at ambient temperature, which is a great practical advantage in tropical areas. Two doses of vaccine are needed, which doubles the total cost of administration since, apart from the cost of the vaccine itself, the herds have to be corralled and handled twice. Large amounts of tissue culture material have to be prepared to manufacture the vaccine, and much research has gone into the optimization of industrial-scale culture techniques in order to reduce the overall cost of production. Currently the most cost effective method includes culturing of E. ruminantium in two-litre stirred tanks using serum–free media,193 followed by purification using membrane filtration.244 The concentration of E. ruminantium protein in the vaccine is estimated by qPCR targeting the Gardel map1 sequence245 and the required dose is then mixed with Montanide™ ISA 70 or 70M. This method yielded the maximum uniformity of particle size and induced 80-100 per cent protection when evaluated against homologous challenge in goats.192
At present the most important problem is that the protection levels of inactivated vaccines in field situations have been disappointing and this appears to be due, at least in part, to the large numbers of E. ruminantium of differing immunogenicities which are in circulation.4, 120, 188 A summary of all the field trials conducted by one research group over a period of several years shows that overall mortality levels of 71 per cent in naive animals can be reduced to 36 per cent by vaccination.182 A further problem factor is that ticks in a field situation present a much more virulent challenge than an experimental needle challenge.73, 262 This is a technical problem with all heartwater vaccine development, not just with the inactivated vaccines.
In theory it would be feasible to include as many immunotypes into an inactivated vaccine as would be required for effectiveness in a particular area. It would be necessary, however, to isolate ‘breakthrough’ organisms from any natural E. ruminantium infection appearing in animals immunized with an existing vaccine. The new strains would then need to be cultured and added to the cocktail, prior to re-testing in the field situation. This would be costly, although it could be a useful ‘stopgap’ measure until a more effective vaccine is developed. In summary the results suggest that at present the formulations for inactivated vaccines are not viable as a commercial proposition. This situation could change with more research, but eventually defined molecular vaccines may prove to be more effective.
Recombinant vaccines
The fact that immunization with inactivated E. ruminantium organisms can stimulate protective immunity implies that the development of a subunit vaccine is feasible. In principle all that is required is to identify those E. ruminantium genes which code for antigens which stimulate a protective response. A recombinant vaccine should be cheaper to manufacture, and easier to store, transport and administer, than an inactivated or attenuated vaccine. There would also be no reason to restrict its use outside the heartwater endemic area as a preventative measure, should the need arise. The most difficult problem is that no directed strategy has been devised to identify the relevant genes.
Because of its serological immunodominance MAP1 was the first E. ruminantium antigen to receive molecular genetic attention and several studies were conducted in an attempt to use the gene or the antigen as vaccines. A DNA vaccine vector expressing map1was shown to protect mice against a lethal homologous challenge, and splenocytes from the immunized mice were shown to proliferate in vitro when treated with rMAP1 protein or a whole protein extract of E. ruminantium.233 The proliferating cells also secreted enhanced amounts of IFN-γ and IL-2 as compared to cells from control mice. There was one serious concern with this study, however; the level of protection was not reproducible, varying from 23 per cent to 88 per cent in different experiments. In order to try and improve the induction of protective CD8+ T cells the team did further experiments in which they primed the mice with the map1 vaccine vector and then boosted with rMAP1. This significantly augmented protection to homologous challenge from 13-27 per cent without boosting to 53-67 per cent with boosting.232 It was disappointing to note that the 88 per cent protection level obtained using only the map1 vaccine vector in the original experiments could not be repeated in the prime boost experiments, which casts doubt on the usefulness of the map1 gene as a heartwater vaccine candidate.
One of the first attempts at a more directed approach used E. ruminantium genomic mini-libraries cloned into a Salmonella vaccine delivery system.79 Recombinants containing each mini-library were used to immunize groups of outbred mice which were subjected to a lethal needle challenge after two weeks.52 In one group of five mice one mouse lived much longer than the others. Each of the clones from the corresponding mini-library was then tested individually in 10 mice, and one mouse survived a lethal challenge. The E. ruminantium clone yielding this protection was tested in larger groups of mice and shown to protect 14 per cent of outbred mice against a lethal challenge.52 The protective clone was sequenced and found to include 11 ORFs,71 the functions of which were not known at the time. The 11 ORFs were cloned individually into a vaccine vector plasmid304 and tested in groups of mice for their ability to induce protective immunity; the survival rates after challenge of the mice in four groups were significantly higher than the controls.74 An equimolar cocktail of the vaccine plasmids containing the four corresponding ORFs was used to immunize five sheep; on subsequent lethal challenge all five sheep inoculated with the vaccine cocktail developed mildly elevated temperatures, but they continued to eat and behave normally, and all recovered without intervention.74 When the complete E. ruminantium genome sequence was annotated these ORFs were predicted to be genes belonging to an ABC transporter system and were given gene identities: Erum2540, Erum2550, Erum2580 and Erum2590.72
Another series of experiments searched through E. ruminantium genomic expression libraries by screening with sera from heartwater-immune sheep.26 Thirty four unique clones which reacted positively were sequenced, and after sequence analysis selected ORFs were cloned into an expression vector and recombinant proteins were generated. These were evaluated for their ability to react with heartwater-immune antisera and to stimulate proliferation of PBMC from cattle immunized by an infection and treatment method. Recombinants which had reacted positively in both tests were used, both individually and in pools, to immunize mice which were subsequently challenged. Significant protection (58-89 per cent survival) was only obtained for two pools of recombinants, but not for any individual recombinant, not even those from the protective pools. In a follow-up study, five of these individual DNA constructs induced partial protection in mice and a cell mediated immune response could be detected.291
At this stage in the search for candidate heartwater vaccine genes two fundamental technical problems were becoming evident: the mouse model, and the needle challenge procedure used to determine protection levels, were both unreliable. The lack of reproducibility in the map1-based vaccine trials in mice has been mentioned above,232 and while searching for protective genes in large E. ruminantium genomic clones it was found that the levels of protection in mice and ruminants were widely different.74, 178 The need for a method of delivering a quantifiable and reproducible E. ruminantium challenge was also evident, and while addressing this latter requirement it was also discovered that Balb/c mice are approximately 10x more sensitive to E. ruminantium challenge than C57BL/6J mice.51 The problems were addressed by only conducting heartwater vaccine development work in ruminants, and by developing at least two different techniques for preparing quantitated challenge material.51, 328
The four ABC transporter genes described above were tested as vaccine candidates with the aim of targeting the cytotoxic T cell (CTL) response in sheep. Four different immunization strategies were investigated: 1) plasmid DNA (pDNA) only, in an equimolar cocktail of the four genes,74, 260 2) pDNA only, but using the individual genes,260 3) pDNA prime followed by recombinant protein boost,262 and 4) a pDNA prime followed by recombinant lumpy skin disease virus (rLSDV) vectored boost.262 The four-gene pDNA cocktail gave repeatable 100 per cent protection against homologous needle challenge (Welgevonden strain) and heterologous needle challenge with four other virulent strains (Blaauwkrans, Ball 3, Gardel, Kwanyanga, and Mara 87/7). In addition, all four strategies were able to fully protect sheep against homologous needle challenge, except the LSDV boost strategy where only 4/5 animals survived. Natural tick challenge experiments, however, were not successful, with only 20 per cent of animals surviving following tick exposure after immunization using the pDNA prime/recombinant protein boost strategy.
The reduced protection against natural challenge is probably the result of immunological suppression induced by the salivary gland proteins of ticks351 plus the fact that only the CTL arm of the immune response was targeted. The existence of non cross protective immunotypes of E. ruminantium in the infecting ticks is unlikely because the four ABC transporter genes are conserved at the amino acid level. Some of the infectious organisms which broke through the immunization had the relevant genes amplified and sequenced; two genes were identical to those in the vaccine, and the other two each had a single conservative or semi-conservative amino acid substitution.262 It appears that a broad spectrum protection is required that does not only target one specific arm of the host immune system, but that the recombinant vaccine should be able to induce innate and adaptive immunity (CD4+ and CD8+ T cells). To achieve this will require the identification of antigens that induce cellular immunity and to determine the cell types producing the appropriate cytokines.
Some individual E. ruminantium genes have also been selected on an empirical basis and investigated as vaccine candidates. The gene originally known as Cowdria polymorphic gene 1 (cpg1), and later given the identification Erum2510,72 was used to immunize sheep by DNA immunization and by DNA prime/recombinant protein boost.178, 261 DNA immunization alone gave variable results and weak protection was observed. In contrast, the DNA prime/recombinant protein boost gave full protection in five sheep against E. ruminantium Welgevonden needle challenge, which suggests that Erum2510 could be a good candidate for inclusion in a recombinant against heartwater vaccine. There is of course a drawback for using any polymorphic genes as a vaccine, which is that they would be likely only to be effective in areas where the specific immunotype predominated. Vaccines with a wider range of effectiveness would require the inclusion of representative copies of each polymorphic variant gene to be found in the area of use.
An outer membrane protein identified as Erum1110,72 and with an ortholog in the Gardel genome126 contains multiple tandem repeats. This protein was shown to act as an adhesin for tick cells,83 but its role, if any, in mediating mammalian cell infection has not been determined. The gene has been identified during the NCBI’s prokaryotic RefSeq genome re-annotation project234 as a TRP47 family tandem repeat effector protein. Several tandem repeat proteins (TRPs) of Ehrlichia spp. have been found to be recognised by immune sera from mammalian hosts, and polyclonal antibodies directed against the tandem repeats from E. chafeensis TRP47 have been shown to provide protection in mice.169. It has now been found that an ortholog of TRP47 occurs in E. muris, identified as P29, but this gene does not contain tandem repeats. Despite this, mice immunized with recombinant E. muris P29 developed a strong antibody response and were protected against virulent E. muris challenge.311 This suggests that E. ruminantium TRP47 family genes should be considered for screening as possible vaccine candidates.
Since complete genome sequences of some E. ruminantium strains have become available there is another alternative to the pragmatic screening of available genes, or the performance of experimental searches through E. ruminantium genomic clones. The process called reverse vaccinology (RV) uses bioinformatic algorithms to search the genome sequence and selects possible vaccine candidate genes based on selected criteria.171, 286 The selection of the criteria, and the efficiency of the algorithms searching for genes having those criteria, are the critical issues with this rapidly evolving technology.
One RV study has so far been conducted for heartwater172 and the work is still in progress. The Welgevonden genome was screened in silico for genes fulfilling the following predicted criteria: surface-associated proteins; secreted proteins; transporters; environmental stress adaptation proteins; proteins containing tandem, tetratricopeptide or ankyrin repeats; adhesins; proteases; iron-binding proteins; methyltransferases; GTPases; and homologs of proteins identified as vaccine candidates in other pathogens. The initial identification of 419 genes was reduced to 272 by eliminating patented genes, genes tested previously, and genes with more than four predicted transmembrane helices. These genes were expressed in E. coli and the recombinant proteins were screened for their ability to stimulate PBMC from heartwater-immune cattle and sheep using several immunological techniques.172, 287, 310 Five low molecular weight proteins were identified that induced cellular immunity and these were tested as a cocktail pDNA vaccine in sheep,287 but only 20 per cent protection was observed. Antigens that induced the desired responses were further investigated to determine the epitopes that produce these immune responses,310 and an alternative delivery system using biodegradable microparticles was also investigated and shows promise in vitro.318 Continuing work is focussing on identifying more protection-stimulating epitopes which will be combined to produce a multi-epitope DNA vaccine.
References
- ABDEL RAHIM, A. I. & SHOMMEIN, A. M. 1978. Haematological studies in goats experimentally infected with Rickettsia ruminantium. Bulletin of Animal Health and Production in Africa, 26, 232-235.
- ADAKAL, H., GAVOTTE, L., STACHURSKI, F., KONKOBO, M., HENRI, H., ZOUNGRANA, S., HUBER, K., VACHIERY, N., MARTINEZ, D., MORAND, S., & FRUTOS, R. 2010. Clonal origin of emerging populations of Ehrlichia ruminantium in Burkina Faso. Infection, Genetics and Evolution, 10, 903-912.
- ADAKAL, H., MEYER, D. F., CARASCO-LACOMBE, C., PINARELLO, V., ALLEGRE, F., HUBER, K., STACHURSKI, F., MORAND, S., MARTINEZ, D., LEFRANCOIS, T., VACHIERY, N., & FRUTOS, R. 2009. MLST scheme of Ehrlichia ruminantium: genomic stasis and recombination in strains from Burkina-Faso. Infection, Genetics and Evolution, 9, 1320-1328.
- ADAKAL, H., STACHURSKI, F., KONKOBO, M., ZOUNGRANA, S., MEYER, D. F., PINARELLO, V., APRELON, R., MARCELINO, I., ALVES, P. M., MARTINEZ, D., LEFRANCOIS, T., & VACHIERY, N. 2010. Efficiency of inactivated vaccines against heartwater in Burkina Faso: impact of Ehrlichia ruminantium genetic diversity. Vaccine, 28, 4573-4580.
- AHMADU, B., LOVELACE, C. E., SAMUI, K. L., & MAHAN, S. 2004. Some observations on the sero-prevalence of heartwater and tick infestation in Zambian goats. Onderstepoort Journal of Veterinary Research, 71, 161-164.
- ALEXANDER, R. A. 1931. Heartwater. The present state of our knowledge of the disease. 17th Report of the Director of Veterinary Services and Animal Industry, Union of South Africa, 17, 89-150.
- ALEXANDER, R. A, NEITZ, W. O., & ADELAAR, T. F. 1946. Heartwater. Farming in South Africa, 21, 548-552.
- ALLSOPP, B. A. 2009. Trends in the control of heartwater. Onderstepoort Journal of Veterinary Research, 76, 81-88.
- ALLSOPP, B. A., ALLSOPP, M. T., DU PLESSIS, J. H., & VISSER, E. S. 1996. Uncharacterized Ehrlichia spp. may contribute to clinical heartwater. Annals of the New York Academy of Sciences, 791, 17-23.
- ALLSOPP, M. T. & ALLSOPP, B. A. 2001. A novel Ehrlichia genotype detected in dogs in South Africa. Journal of Clinical Microbiology, 39, 4204-4207.
- ALLSOPP, M. T. & ALLSOPP, B. A. 2007. Extensive genetic recombination occurs in the field between different genotypes of Ehrlichia ruminantium. Veterinary Microbiology, 124, 58-65.
- ALLSOPP, M. T., DORFLING, C. M., MAILLARD, J. C., BENSAID, A., HAYDON, D. T., VAN HEERDEN, H., & ALLSOPP, B. A. 2001. Ehrlichia ruminantium major antigenic protein gene (map1) variants are not geographically constrained and show no evidence of having evolved under positive selection pressure. Journal of Clinical Microbiology, 39, 4200-4203.
- ALLSOPP, M. T., HATTINGH, C. M., VOGEL, S. W., & ALLSOPP, B. A. 1999. Evaluation of 16S, map1 and pCS20 probes for detection of Cowdria and Ehrlichia species. Epidemiology and Infection, 122, 323-328.
- ALLSOPP, M. T., THERON, J., COETZEE, M. L., DUNSTERVILLE, M. T., & ALLSOPP, B. A. 1999. The occurrence of Theileria and Cowdria parasites in African buffalo (Syncerus caffer) and their associated Amblyomma hebraeum ticks. Onderstepoort Journal of Veterinary Research, 66, 245-249.
- ALLSOPP, M. T., VAN HEERDEN, H., STEYN, H. C., & ALLSOPP, B. A. 2003. Phylogenetic relationships among Ehrlichia ruminantium isolates. Annals of the New York Academy of Sciences, 990, 685-691.
- ALLSOPP, M. T., VAN STRIJP, M. F., FABER, E., JOSEMANS, A. I., & ALLSOPP, B. A. 2007. Ehrlichia ruminantium variants which do not cause heartwater found in South Africa. Veterinary Microbiology, 120, 158-166.
- ALLSOPP, M. T., VISSER, E. S., DU PLESSIS, J. L., VOGEL, S. W., & ALLSOPP, B. A. 1997. Different organisms associated with heartwater as shown by analysis of 16S ribosomal RNA gene sequences. Veterinary Parasitology, 71, 283-300.
- ANDREW, H. R. & NORVAL, R. A. 1989. The carrier status of sheep, cattle and African buffalo recovered from heartwater. Veterinary Parasitology, 34, 261-266.
- ANON, NCBI Resources (2017). – Ehrlichia ruminantium: Organism Overview; Genome Assembly and Annotation report. https://www.ncbi.nlm.nih.gov/genome/genomes/436 Accessed 2017
- ANONYMOUS. 1984. Chapter 14: Methods of immunization against Cowdria ruminantium, p. 564-576. In FAO (ed.), Ticks and Tick-Borne Diseases Control. A Practical Field Manual - Tick-Borne Disease Control. FAO, Rome.
- ARNOLD, R. M. & ASSELBERGS, M. 1981. Tick-borne diseases - Immunization of cattle imported into Mozambique. World Animal Review, 40, 23-29.
- AVAKYAN, A. A. & POPOV, V. L. 1984. Rickettsiaceae and Chlamydiaceae: comparative electron microscopic studies. Acta Virologica, 28, 159-173.
- AXSELL, G. 1998. Personal Communication.
- BARBET, A., TEBELE, N., SEMU, S., PETER, T., WASSINK, L., & MAHAN, S. 1993. Serological diagnosis of heartwater in Zimbabwe - Problems and perspectives. Revue d'Élevage et de Médecine Vétérinaire des Pays Tropicaux , 46, 121.
- BARBET, A. F., SEMU, S. M., CHIGAGURE, N., KELLY, P. J., JONGEJAN, F., & MAHAN, S. M. 1994. Size variation of the major immunodominant protein of Cowdria ruminantium. Clinical and Diagnostic Laboratory Immunology, 1, 744-746.
- BARBET, A. F., WHITMIRE, W. M., KAMPER, S. M., SIMBI, B. H., GANTA, R. R., MORELAND, A. L., MWANGI, D. M., MCGUIRE, T. C., & MAHAN, S. M. 2001. A subset of Cowdria ruminantium genes important for immune recognition and protection. Gene, 275, 287-298.
- BARNARD, W. G. 1953. Heartwater immunization under field conditions in Swaziland. Bulletin of Epizootic Diseases of Africa, 1, 300-313.
- BARRÉ, N., UILENBERG, G., MOREL, P. C., & CAMUS, E. 1987. Danger of introducing heartwater onto the American mainland: potential role of indigenous and exotic Amblyomma ticks. Onderstepoort Journal of Veterinary Research, 54, 405-17.
- BARRÉ, N., GARRIS, G., & CAMUS, E. 1995. Propagation of the tick Amblyomma variegatum in the Caribbean. Revue Scientifique et Technique, 14, 841-855.
- BEATI, L., PATEL, J., LUCAS-WILLIAMS, H., ADAKAL, H., KANDUMA, E. G., TEMBO-MWASE, E., KRECEK, R., MERTINS, J. W., ALFRED, J. T., KELLY, S., & KELLY, P. 2012. Phylogeography and demographic history of Amblyomma variegatum (Fabricius) (Acari: Ixodidae), the tropical bont tick. Vector Borne Zoonotic Diseases, 12, 514-525.
- BEKKER, C. P., POSTIGO, M., TAOUFIK, A., BELL-SAKYI, L., FERRAZ, C., MARTINEZ, D., & JONGEJAN, F. 2005. Transcription analysis of the major antigenic protein 1 multigene family of three in vitro-cultured Ehrlichia ruminantium isolates. Journal of Bacteriology, 187, 4782-4791.
- BELL-SAKYI, L. 2004. Ehrlichia ruminantium grows in cell lines from four ixodid tick genera. Journal of Comparative Pathology, 130, 285-93.
- BELL-SAKYI, L., KONEY, E. B., DOGBEY, O., & SUMPTION, K. J. 1996. Heartwater in Ghana: implications for control of ticks. Tropical Animal Health and Production, 28, 59S-64S; discussion 74S-86S.
- BELL-SAKYI, L., KONEY, E. B. M., DOGBEY, O., ABBAM, J. A., & ANING, K. G. 1997. Isolation and in vitro cultivation in Ghana of Cowdria ruminantium, the causative agent of heartwater. In Proceedings of the 22nd Annual Congress of the Ghana Veterinary Medical Association., Koney, E. B. M. & Aning, K. G. (eds.), 46-51
- BELL-SAKYI, L., PAXTON, E. A., MUNDERLOH, U. G., & SUMPTION, K. J. 2000. Growth of Cowdria ruminantium, the causative agent of heartwater, in a tick cell line. Journal of Clinical Microbiology, 38, 1238-40.
- BEVERIDGE, T. J. & DAVIES, J. A. 1983. Cellular responses of Bacillus subtilis and Escherichia coli to the Gram stain. Journal of Bacteriology, 156, 846-858.
- BEZUIDENHOUT, J. D. 1982. Diseases of the central nervous system of cattle under tropical conditions. In Proceedings of the 12th World Congress on Diseases of Cattle, The Netherlands, Amsterdam. 991-993
- BEZUIDENHOUT, J. D. 1984. Demonstration of Cowdria ruminantium in Amblyomma hebraeum by fluorescent antibody techniques, light and electron microscopy. Onderstepoort Journal of Veterinary Research, 51, 213-215.
- BEZUIDENHOUT, J. D. 1987. The present state of Cowdria ruminantium cultivation in cell lines. Onderstepoort Journal of Veterinary Research, 54, 205-210.
- BEZUIDENHOUT, J. D. 1987. Natural transmission of heartwater. Onderstepoort Journal of Veterinary Research, 54, 349-351.
- BEZUIDENHOUT, J. D. 1988. Certain aspects of the transmission of heartwater, the occurrence of the organism in ticks and in vitro culture. D.V.Sc. Thesis, University of Pretoria, South Africa.
- BEZUIDENHOUT, J. D. 1989. Chapter 2: Cowdria vaccines, p. 31-42. In Veterinary Protozoan and Hemoparasite Vaccines. CRC Press, Inc., Boca Raton, Florida.
- BEZUIDENHOUT, J. D. & BIGALKE, R. D. 1987. The control of heartwater by means of tick control. Onderstepoort Journal of Veterinary Research, 54, 525-528.
- BEZUIDENHOUT, J. D. & JACOBSZ, C. J. 1986. Proof of transovarial transmission of Cowdria ruminantium by Amblyomma herbraeum. Onderstepoort Journal of Veterinary Research, 53, 31-34.
- BEZUIDENHOUT, J. D., PATERSON, C. L., & BARNARD, B. J. 1985. In vitro cultivation of Cowdria ruminantium. Onderstepoort Journal of Veterinary Research, 52, 113-120.
- BIGUEZOTON, A., NOEL, V., ADEHAN, S., ADAKAL, H., DAYO, G. K., ZOUNGRANA, S., FAROUGOU, S., & CHEVILLON, C. 2016. Ehrlichia ruminantium infects Rhipicephalus microplus in West Africa. Parasites and Vectors, 9, 354.
- BOURDOULOUS, S., BENSAID, A., MARTINEZ, D., SHEIKBOUDOU, C., TRAP, I., STROSBERG, A. D., & COURAUD, P. O. 1995. Infection of bovine brain microvessel endothelial cells with Cowdria ruminantium elicits IL-1 beta, -6, and -8 mRNA production and expression of an unusual MHC class II DQ alpha transcript. Journal of Immunology, 154, 4032-4038.
- BOVARNICK, M. R., MILLER, J. C., & SNYDER, J. C. 1950. The influence of certain salts, amino acids, sugars and proteins on the stability of rickettsiae. Journal of Bacteriology, 59, 509-522.
- BOWIE, M. V., REDDY, G. R., SEMU, S. M., MAHAN, S. M., & BARBET, A. F. 1999. Potential value of major antigenic protein 2 for serological diagnosis of heartwater and related ehrlichial infections. Clinical & Diagnostic Laboratory Immunology, 6, 209-215.
- BRAYTON, C. F. 1986. Dimethyl sulfoxide (DMSO): A review. Cornell Veterinarian, 76, 61-90.
- BRAYTON, K. A., COLLINS, N. E., VAN STRIJP, F., & ALLSOPP, B. A. 2003. Preparation of Ehrlichia ruminantium challenge material for quantifiable and reproducible challenge in mice and sheep. Veterinary Parasitology, 112, 63-73.
- BRAYTON, K. A., VAN DER WALT, M., VOGEL, S. W., & ALLSOPP, B. A. 1998. A partially protective clone from Cowdria ruminantium identified by using a Salmonella vaccine delivery system. Annals of the New York Academy of Sciences, 849, 247-252.
- BRETT, M. S., BEZUIDENHOUT, J. D., & DE WAAL, D. T. 1991. The establishment of an ovine cerebral endothelial cell line that supports the growth of Cowdria ruminantium. In Programme and Abstracts of Papers and Posters for Congress on Parasitology of Wildlife, Kruger National Park. p. 72
- BROWN, C. C. & SKOWRONEK, A. J. 1990. Histologic and immunochemical study of the pathogenesis of heartwater (Cowdria ruminantium infection) in goats and mice. American Journal of Veterinary Research, 51, 1476-1480.
- BRUEN, T. C., PHILIPPE, H., & BRYANT, D. 2006. A simple and robust statistical test for detecting the presence of recombination. Genetics, 172, 2665-2681.
- BURDIN, M. L. 1962. Selective staining of Rickettsia ruminantium in tissue sections. The Veterinary Record, 74, 1371-1372.
- BURRIDGE, M. J., SIMMONS, L. A., SIMBI, B. H., PETER, T. F., & MAHAN, S. M. 2000. Evidence of Cowdria ruminantium infection (heartwater) in Amblyomma sparsum ticks found on tortoises imported into Florida. Journal of Parasitology, 86, 1135-1136.
- BYROM, B. & YUNKER, C. E. 1990. Improved culture conditions for Cowdria ruminantium (Rickettsiales), the agent of heartwater disease of domestic ruminants. Cytotechnology, 4, 285-290.
- BYROM, B., YUNKER, C. E., DONOVAN, P. L., & SMITH, G. E. 1991. In vitro isolation of Cowdria ruminantium from plasma of infected ruminants. Veterinary Microbiology, 26, 263-268.
- CAMUS, E. 1987. Contribution à l'étude épidémiologique de la cowdriose (Cowdria ruminantium) en Guadeloupe. D.Sc. Thesis, Université de Paris-Sud, Paris.
- CAMUS, E. 1992. Le portage asymptomatique de bovins et chèvres Créole guéris de la cowdriose en Guadeloupe. Revue d’Élevage et de Médecine Vétérinaire des Pays Tropicaux, 45, 133-135.
- CAMUS, E. AND BARRÉ, N. La cowdriose (heartwater) - Revue générale des connaissances. 1982. Institut d'Élevage et de Médecine Vétérinaire des Pays Tropicaux, 10 Rue Pierre-Curie, F-94704 Maisons-Alfort, France.
- CAMUS, E. & BARRÉ, N. 1995. Vector situation of tick-borne diseases in the Caribbean Islands. Veterinary Parasitology, 57, 167-176.
- CAMUS, E. & BARRÉ, N. 1987. Diagnosis of heartwater in the live animal: experiences with goats in Guadeloupe. Onderstepoort Journal of Veterinary Research, 54, 291-294.
- CAMUS, E. & BARRÉ, N. 1992. The role of Amblyomma variegatum in the transmission of heartwater with special reference to Guadeloupe. Annals of the New York Academy of Sciences, 653, 33-41.
- CAMUS, E., MAILLARD, J. C., RUFF, G., PEPIN, L., NAVES, M., & MATHERON, G. 1996. Genetic resistance of Creole goats to cowdriosis in Guadeloupe. Status in 1995. Annals of the New York Academy of Sciences, 791, 46-53.
- CANGI, N., GORDON, J. L., BOURNEZ, L., PINARELLO, V., APRELON, R., HUBER, K., LEFRANCOIS, T., NEVES, L., MEYER, D. F., & VACHIERY, N. 2016. Recombination Is a Major Driving Force of Genetic Diversity in the Anaplasmataceae Ehrlichia ruminantium. Frontiers in Cellular and Infection Microbiology, 6, 111.
- CHAMBOKO, T., MUKHEBI, A. W., CALLAGHAN, C. J., PETER, T. F., KRUSKA, R. L., MEDLEY, G. F., MAHAN, S. M., & PERRY, B. D. 1999. The control of heartwater on large-scale commercial and smallholder farms in Zimbabwe. Preventive Veterinary Medicine, 39, 191-210.
- CLARK, R. 1962. The pathological physiology of heartwater Cowdria (Rickettsia) ruminantium - Cowdry, 1926. Onderstepoort Journal of Veterinary Research, 29, 25-33.
- CLARKE, E. G. C. & CLARKE M. L. 1970. Garner's Veterinary Toxicology. Baillière Tindall & Cassell., London.
- COLLINS, N. E., DE VILLIERS, E. P., BRAYTON, K. A., & ALLSOPP, B. A. 1998. DNA sequence of a cosmid clone of Cowdria ruminantium. Annals of the New York Academy of Sciences, 849, 365-368.
- COLLINS, N. E., LIEBENBERG, J., DE VILLIERS, E. P., BRAYTON, K. A., LOUW, E., PRETORIUS, A., FABER, F. E., VAN HEERDEN, H., JOSEMANS, A., VAN KLEEF, M., STEYN, H. C., VAN STRIJP, M. F., ZWEYGARTH, E., JONGEJAN, F., MAILLARD, J. C., BERTHIER, D., BOTHA, M., JOUBERT, F., CORTON, C. H., THOMSON, N. R., ALLSOPP, M. T., & ALLSOPP, B. A. 2005. The genome of the heartwater agent Ehrlichia ruminantium contains multiple tandem repeats of actively variable copy number. Proceedings of the National Academy of Sciences of the United States of America, 102, 838-843.
- COLLINS, N. E., PRETORIUS, A., VAN KLEEF, M., BRAYTON, K. A., ALLSOPP, M. T., ZWEYGARTH, E., & ALLSOPP, B. A. 2003. Development of improved attenuated and nucleic acid vaccines for heartwater. Developments in Biologicals, 114, 121-136.
- COLLINS, N. E., PRETORIUS, A., VAN KLEEF, M., BRAYTON, K. A., ZWEYGARTH, E., & ALLSOPP, B. A. 2003. Development of improved vaccines for heartwater. Annals of the New York Academy of Sciences, 990, 474-484.
- COUTINHO, G. C., DURIEU-TRAUTMANN, O., STROSBERG, A. D., & COURAUD, P. O. 1991. Catecholamines stimulate the IFN-gamma-induced class II MHC expression on bovine brain capillary endothelial cells. Journal of Immunology, 147, 2525-9.
- COWDRY, E. V. 1925. Studies on the etiology of heartwater 1. Observation of a rickettsia, Rickettsia ruminantium (n. sp.), in the tissues of infected animals. Journal of Experimental Medicine, 42, 231-252.
- COWDRY, E. V. 1925. Studies on the etiology of heartwater 2. Rickettsia ruminantium (n. sp.) in the tissues of ticks transmitting the disease. Journal of Experimental Medicine, 42, 253-274.
- COWDRY, E V. 1926. Studies on the etiology of heartwater 3. The multiplication of Rickettsia ruminantium within the endothelial cells of infected animals and their discharge into the circulation. Journal of Experimental Medicine, 44, 803-814.
- CURTISS, R. & KELLY, S. M. 1987. Salmonella typhimurium deletion mutants lacking adenylate cyclase and cyclic AMP receptor protein are avirulent and immunogenic. Infection and Immunity, 55, 3035-3043.
- DA GRACA, H. M. 1964. Some aspects of heartwater in Angola. Bulletin de l'Office International des Epizooties, 62, 963-969.
- DA SILVA, A. S., MUNHOZ, T. D., FARIA, J. L., VARGAS-HERNANDEZ, G., MACHADO, R. Z., ALMEIDA, T. C., MORESCO, R. N., STEFANI, L. M., & TINUCCI-COSTA, M. 2013. Increase nitric oxide and oxidative stress in dogs experimentally infected by Ehrlichia canis: effect on the pathogenesis of the disease. Veterinary Microbiology, 164, 366-369.
- DE LA FUENTE, J., BLOUIN, E. F., & KOCAN, K. M. 2003. Infection exclusion of the rickettsial pathogen Anaplasma marginale in the tick vector Dermacentor variabilis. Clinical and Diagnostic Laboratory Immunology, 10, 182-184.
- DE LA FUENTE, J., GARCIA-GARCIA, J. C., BARBET, A. F., BLOUIN, E. F., & KOCAN, K. M. 2004. Adhesion of outer membrane proteins containing tandem repeats of Anaplasma and Ehrlichia species (Rickettsiales: Anaplasmataceae) to tick cells. Veterinary Microbiology, 98, 313-322.
- DE WAAL, D. T., MATTHEE, O., & JONGEJAN, F. 2000. Evaluation of the MAP1B ELISA for the diagnosis of heartwater in South Africa. Annals of the New York Academy of Sciences, 916, 622-627.
- DEEM, S. L. 1998. A review of heartwater and the threat of introduction of Cowdria ruminantium and Amblyomma spp. ticks to the American mainland. Journal of Zoo and Wildlife Medicine, 29, 109-113.
- DEEM, S. L., NORVAL, R. A., DONACHIE, P. L., & MAHAN, S. M. 1996. Demonstration of vertical transmission of Cowdria ruminantium, the causative agent of heartwater, from cows to their calves. Veterinary Parasitology, 61, 119-132.
- DIXON, R. W. 1898. Heartwater experiments. Agricultural Journal of the Cape of Good Hope, 12, 754-760.
- DIXON, R. W. 1899. Heartwater experiments. Agricultural Journal of the Cape of Good Hope, 14, 205-207.
- DRUMMOND, A. J., SUCHARD, M. A., XIE, D., & RAMBAUT, A. 2012. Bayesian phylogenetics with BEAUti and the BEAST 1.7. Molecular Biology and Evolution, 29, 1969-73.
- DU PLESSIS, J. L. 1970. Immunity in heartwater. I. A preliminary note on the role of serum antibodies. Onderstepoort Journal of Veterinary Research, 37, 147-9.
- DU PLESSIS, J. L. 1970. Pathogenesis of heartwater. I. Cowdria ruminantium in the lymph nodes of domestic ruminants. Onderstepoort Journal of Veterinary Research, 37, 89-95.
- DU PLESSIS, J. L. 1975. Electron microscopy of Cowdria ruminantium infected reticulo-endothelial cells of the mammalian host. Onderstepoort Journal of Veterinary Research, 42, 1-13.
- DU PLESSIS, J. L. 1975. Histopathological studies on the pathogenesis of heartwater as manifested in mice infected with a strain of Cowdria ruminantium. M.Med.Vet. thesis. University of Pretoria.
- DU PLESSIS, J. L. 1981. The application of the indirect fluorescent antibody test to the serology of heartwater. In "Tick Biology and Control", Proceedings of an International Conference, Whitehead, G. B. & Gibson, J. D. (eds.), Tick Research Unit, Rhodes University, Grahamstown, South Africa. 47-52
- DU PLESSIS, J. L. 1981. The influence of dithiosemicarbazone on the immunity of sheep to heartwater. Onderstepoort Journal of Veterinary Research, 48, 175-176.
- DU PLESSIS, J. L. 1982. Mice infected with a Cowdria ruminantium-like agent as a model in the study of heartwater. D.V.Sc. Thesis, University of Pretoria, South Africa.
- DU PLESSIS, J. L. 1985. A method for determining the Cowdria ruminantium infection rate of Amblyomma hebraeum: effects in mice injected with tick homogenates. Onderstepoort Journal of Veterinary Research, 52, 55-61.
- DU PLESSIS, J. L. 1990. Increased pathogenicity of an Ehrlichia-like agent after passage through Amblyomma hebraeum: a preliminary report. Onderstepoort Journal of Veterinary Research, 57, 233-237.
- DU PLESSIS, J. L. 1999. Personal Communication.
- DU PLESSIS, J. L., Berche, P., & Van Gas, L. 1991. T cell-mediated immunity to Cowdria ruminantium in mice: the protective role of Lyt-2+ T cells. Onderstepoort Journal of Veterinary Research, 58, 171-179.
- DU PLESSIS, J. L. & BEZUIDENHOUT, J. D. 1979. Investigations on the natural and acquired resistance of cattle to artificial infection with Cowdria ruminantium. Journal of the South African Veterinary Association, 50, 334-338.
- DU PLESSIS, J. L., BEZUIDENHOUT, J. D., & LUDEMANN, C. J. 1984. The immunization of calves against heartwater: subsequent immunity both in the absence and presence of natural tick challenge. Onderstepoort Journal of Veterinary Research, 51, 193-196.
- DU PLESSIS, J. L., GRAY, C., & VAN STRIJP, M. F. 1992. Flow cytometric analysis of T cell response in mice infected with Cowdria ruminantium. Onderstepoort Journal of Veterinary Research, 59, 337-338.
- DU PLESSIS, J. L., JANSEN, B. C., & PROZESKY, L. 1983. Heartwater in Angora goats. I. Immunity subsequent to artificial infection and treatment. Onderstepoort Journal of Veterinary Research, 50, 137-143.
- DU PLESSIS, J. L. & KUMM, N. A. 1971. The passage of Cowdria ruminantium in mice. Journal of the South African Veterinary Medical Association, 42, 217-221.
- DU PLESSIS, J. L. & MALAN, L. 1987. The application of the indirect fluorescent antibody test in research on heartwater. Onderstepoort Journal of Veterinary Research, 54, 319-325.
- DU PLESSIS, J. L. & MALAN, L. 1987. The block method of vaccination against heartwater. Onderstepoort Journal of Veterinary Research, 54, 493-495.
- DU PLESSIS, J. L. & MALAN, L. 1987. The non-specific resistance of cattle to heartwater. Onderstepoort Journal of Veterinary Research, 54, 333-336.
- DU PLESSIS, J. L. & MALAN, L. 1987. Problems with the interpretation of epidemiological data in heartwater: a study on 23 farms. Onderstepoort Journal of Veterinary Research, 54, 427-433.
- DU PLESSIS, J. L., VAN GAS, L., OLIVIER, J. A., & BEZUIDENHOUT, J. D. 1989. The heterogenicity of Cowdria ruminantium stocks: cross-immunity and serology in sheep and pathogenicity to mice. Onderstepoort Journal of Veterinary Research, 56, 195-201.
- DUMLER, J. S., BARBET, A. F., BEKKER, C. P., DASCH, G. A., PALMER, G. H., RAY, S. C., RIKIHISA, Y., & RURANGIRWA, F. R. 2001. Reorganization of genera in the families Rickettsiaceae and Anaplasmataceae in the order Rickettsiales: unification of some species of Ehrlichia with Anaplasma, Cowdria with Ehrlichia and Ehrlichia with Neorickettsia, descriptions of six new species combinations and designation of Ehrlichia equi and 'HGE agent' as subjective synonyms of Ehrlichia phagocytophila. International Journal of Systematic and Evolutionary Microbiology, 51, 2145-2165.
- EDGAR, R. C. 2004. MUSCLE: multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Research, 32, 1792-1797.
- EDINGTON, A. 1898. Heartwater. Agricultural Journal of the Cape of Good Hope, 12, 748-754.
- EMBOULE, L., DAIGLE, F., MEYER, D. F., MARI, B., PINARELLO, V., SHEIKBOUDOU, C., MAGNONE, V., FRUTOS, R., VIARI, A., BARBRY, P., MARTINEZ, D., LEFRANCOIS, T., & VACHIERY, N. 2009. Innovative approach for transcriptomic analysis of obligate intracellular pathogen: selective capture of transcribed sequences of Ehrlichia ruminantium. BMC Molecular Biology, 10, 111.
- ESEMU, S. N., BESONG, W. O., NDIP, R. N., & NDIP, L. M. 2013. Prevalence of Ehrlichia ruminantium in adult Amblyomma variegatum collected from cattle in Cameroon. Experimental and Applied Acarology, 59, 377-387.
- ESTEVES, I., MARTINEZ, D., & TOTTE, P. 2004. Identification of Ehrlichia ruminantium (Gardel strain) IFN-gamma inducing proteins after vaccination with a killed vaccine. Veterinary Microbiology, 100, 233-240.
- ESTEVES, I., VACHIERY, N., MARTINEZ, D., & TOTTE, P. 2004. Analysis of Ehrlichia ruminantium-specific T1/T2 responses during vaccination with a protective killed vaccine and challenge of goats. Parasite Immunology, 26, 95-103.
- ESTEVES, I., WALRAVENS, K., VACHIERY, N., MARTINEZ, D., LETESSON, J. J., & TOTTE, P. 2004. Protective killed Ehrlichia ruminantium vaccine elicits IFN-gamma responses by CD4+ and CD8+ T lymphocytes in goats. Veterinary Immunology and Immunopathology, 98, 49-57.
- EVANNO, G., REGNAUT, S., & GOUDET, J. 2005. Detecting the number of clusters of individuals using the software STRUCTURE: a simulation study. Molecular Ecology, 14, 2611-2620.
- FABURAY, B., GEYSEN, D., CEESAY, A., MARCELINO, I., ALVES, P. M., TAOUFIK, A., POSTIGO, M., BELL-SAKYI, L., & JONGEJAN, F. 2007. Immunisation of sheep against heartwater in The Gambia using inactivated and attenuated Ehrlichia ruminantium vaccines. Vaccine, 25, 7939-7947.
- FABURAY, B., GEYSEN, D., MUNSTERMANN, S., BELL-SAKYI, L., & JONGEJAN, F. 2007. Longitudinal monitoring of Ehrlichia ruminantium infection in Gambian lambs and kids by pCS20 PCR and MAP1-B ELISA. BMC Infectious Diseases, 7, 85.
- FABURAY, B., GEYSEN, D., MUNSTERMANN, S., TAOUFIK, A., POSTIGO, M., & JONGEJAN, F. 2007. Molecular detection of Ehrlichia ruminantium infection in Amblyomma variegatum ticks in The Gambia. Experimental and Applied Acarology, 42, 61-74.
- FABURAY, B., MUNSTERMANN, S., GEYSEN, D., BELL-SAKYI, L., CEESAY, A., BODAAN, C., & JONGEJAN, F. 2005. Point seroprevalence survey of Ehrlichia ruminantium infection in small ruminants in The Gambia. Clinical and Diagnostic Laboratory Immunology, 12, 508-512.
- FICK, J. F. & SCHUSS, J. 1952. Heartwater immunisation under field conditions in Swaziland. Journal of the South African Veterinary Medical Association, 23, 9-14.
- FLACH, E. J., WOODFORD, J. D., MORZARIA, S. P., DOLAN, T. T., & SHAMBWANA, I. 1990. Identification of Babesia bovis and Cowdria ruminantium on the island of Unguja, Zanzibar. Veterinary Record, 126, 57-59.
- FRUTOS, R., VIARI, A., FERRAZ, C., MORGAT, A., EYCHENIE, S., KANDASSAMY, Y., CHANTAL, I., BENSAID, A., COISSAC, E., VACHIERY, N., DEMAILLE, J., & MARTINEZ, D. 2006. Comparative genomic analysis of three strains of Ehrlichia ruminantium reveals an active process of genome size plasticity. Journal of Bacteriology, 188, 2533-2542.
- FRUTOS, R., VIARI, A., VACHIERY, N., BOYER, F., & MARTINEZ, D. 2007. Ehrlichia ruminantium: genomic and evolutionary features. Trends in Parasitology, 23, 414-419.
- GOFTON, A. W., WAUDBY, H. P., PETIT, S., GREAY, T. L., RYAN, U. M., & IRWIN, P. J. 2017. Detection and phylogenetic characterisation of novel Anaplasma and Ehrlichia species in Amblyomma triguttatum subsp. from four allopatric populations in Australia. Ticks and Tick Borne Diseases, 8, 749-756.
- GOODMAN, L. S. AND GILMAN, A. The Pharmacological Basis of Therapeutics. 12th. Edition. 2011. BRUNTON, L., CHABNER, B. A., & KNOLLMANN, B. C. Editors. McGraw-Hill, New York.
- GRADWELL, D. V., VAN NIEKERK, C. A., & JOUBERT, D. C. 1976. Attempted artificaial infection or impala, blue wildebeest, buffalo, kudu, giraffe and warthog with heartwater. Journal of the South African Veterinary Association, 47, 209-10.
- GRAF, H. 1933. Chemical blood studies. 3. Comparative studies on "laked" and "unlaked" blood filtrates of sheep in health and during "heartwater" (Rickettsia ruminantium infection) and bluetongue (catarrhal fever). Onderstepoort Journal of Veterinary Science and Animal Industry, 1, 285-334.
- GROSSKOPF, J. F. W. 1958. Heartwater immunization of eland (Taurotrachus oryx oryx, Pallas). Journal of the South African Veterinary Medical Association, 29, 329-330.
- GRUSS, B. 1981. Pigmentation of plasma in heartwater. Journal of the South African Veterinary Association, 52, 338.
- GRUSS, B. 1981. A practical approach to the control of heartwater in the Angora goat and certain sheep breeds in the Eastern Cape Coastal region. In "Tick Biology and Control", Proceedings of an International Conference, Whitehead, G. B. & Gibson, J. D. (eds.), Tick Research Unit, Rhodes University, Grahamstown, South Africa. 135-136
- GUEYE, A., JONGEJAN, F., MBENGUE, M., DIOUF, A., & UILENBERG, G. 1994. Essai sur le terrain d'un vaccin atténué contre la cowdriose. Revue d’Élevage et de Médecine Vétérinaire des Pays Tropicaux, 47, 401-404.
- GUEYE, A., MARTINEZ, D., MBENGUE, M. B., DIEYE, T. H., & DIOUF, A. 1993. Épidémiologie de la cowdriose au Sénégal. 2. Résultats de suivis séro-épidémiologiques. Revue d’Élevage et de Médecine Vétérinaire des Pays Tropicaux, 46, 449-454.
- GUEYE, A., MBENGUE, M., & DIOUF, A. 1993. Épidémiologie de la cowdriose au Sénégal. 1. Étude de la transmission et du taux d'infection d'Amblyomma variegatum (Fabricius, 1794) dans la région des Niayes. Revue d’Élevage et de Médecine Vétérinaire des Pays Tropicaux, 46, 441-447.
- GUINDON, S., DUFAYARD, J. F., LEFORT, V., ANISIMOVA, M., HORDIJK, W., & GASCUEL, O. 2010. New algorithms and methods to estimate maximum-likelihood phylogenies: assessing the performance of PhyML 3.0. Syst Biol, 59, 307-321.
- GUINDON, S. & GASCUEL, O. 2003. A simple, fast, and accurate algorithm to estimate large phylogenies by maximum likelihood. Syst Biol, 52, 696-704.
- GUMMOW, B., OBEREM, P. T., & VAN AMSTEL, S. 1988. New ideas in the symptomatic treatment of heartwater. In Proceedings of the South African Veterinary Association Biennial Congress, Pietermaritzburg, South Africa. 89-97
- HAIG, D. A. 1952. Note on the use of the white mouse for the transport of strains of heartwater. Journal of the South African Veterinary Medical Association, 23, 167-170.
- HAIG, D. A. 1955. Tickborne rickettsioses in South Africa. Advances in Veterinary Sciences, 2, 307-325.
- HAIG, D. A, ALEXANDER, R A, & WEISS, K E. 1954. Treatment of heartwater with terramycin. Journal of the South African Veterinary Medical Association, 25, 45-48.
- HENNING, M. W. Heartwater, hartwater. 3rd Edition. 1956. HENNING, M. W. Author. Central News Agency Ltd., South Africa.
- HOLLAND, C. J, LOGAN, L. L., MEBUS, C. A, & RISTIC, M. 1987. The serological relationship between Cowdria ruminantium and certain members of the genus Ehrlichia. Onderstepoort Journal of Veterinary Research, 54, 331.
- HOWELL, D. J., PETNEY, T. N., & HORAK, I. G. 1989. The host status of the striped mouse, Rhabdomys pumilio, in relation to the tick vectors of heartwater in South Africa. Onderstepoort Journal of Veterinary Research, 56, 289-291.
- HUDSON, J. R. & HENDERSON, R. M. 1941. Some preliminary experiments on the survival of heartwater "virus" in rats. Journal of the South African Veterinary Medical Association, 12, 39-49.
- HUGHES, A. L. & FRENCH, J. O. 2007. Homologous recombination and the pattern of nucleotide substitution in Ehrlichia ruminantium. Gene, 387, 31-37.
- HUTCHEON, D. 1900. History of heartwater. Agricultural Journal of the Cape of Good Hope, 17, 410-417.
- ILEMOBADE, A. A. 1977. Study of heartwater and the causative agent, Cowdria ruminantium (Cowdry 1925), in Nigeria. Dissertation Abstracts International, 38 B, p. 541.
- ILEMOBADE, A. A. & BLOTKAMP, C. 1978. Clinico-pathological study of heartwater in goats. Tropenmedizin und Parasitologie, 29, 71-76.
- ILEMOBADE, A. A. & BLOTKAMP, J. 1975. Preservation of Cowdria ruminantium at low termperatures. Research in Veterinary Science, 19, 337-338.
- IMMELMAN, A. & DREYER, G. 1982. The use of doxycycline to control heartwater in sheep. Journal of the South African Veterinary Association, 53, 23-24.
- JONGEJAN, F. 1991. Protective immunity to heartwater (Cowdria ruminantium infection) is acquired after vaccination with in vitro-attenuated rickettsiae. Infection and Immunity, 59, 729-731.
- JONGEJAN, F. & BEKKER, C. P. J. 1999. Cowdria ruminantium: recent developments in diagnostic methods, molecular characterization and vaccines, p. 373-386. In Raoult, D. & Brouqui, P. (eds.), Rickettsia and rickettsial diseases at the turn of the third millenium. Elsevier, Paris.
- JONGEJAN, F., THIELEMANS, M. J., BRIÈRE, C., & UILENBERG, G. 1991. Antigenic diversity of Cowdria ruminantium isolates determined by cross- immunity. Research in Veterinary Science, 51, 24-28.
- JONGEJAN, F., UILENBERG, G., FRANSSEN, F. F., GUEYE, A., & NIEUWENHUIJS, J. 1988. Antigenic differences between stocks of Cowdria ruminantium. Research in Veterinary Science, 44, 186-189.
- JONGEJAN, F., ZANDBERGEN, T. A., VAN DE WIEL, P. A., DE GROOT, M., & UILENBERG, G. 1991. The tick-borne rickettsia Cowdria ruminantium has a Chlamydia-like developmental cycle. Onderstepoort Journal of Veterinary Research, 58, 227-237.
- KAKONO, O., HOVE, T., GEYSEN, D., & MAHAN, S. 2003. Detection of antibodies to the Ehrlichia ruminantium MAP1-B antigen in goat sera from three communal land areas of Zimbabwe by an indirect enzyme-linked immunosorbent assay. Onderstepoort Journal of Veterinary Research, 70, 243-249.
- KANYARI, P. W. & KAGIRA, J. 2000. The role of parasitic diseases as causes of mortality in cattle in a high potential area of central Kenya: a quantitative analysis. Onderstepoort Journal of Veterinary Research, 67, 157-161.
- KARRAR, G. 1960. Rickettsial infection (heartwater) in sheep and goats in the Sudan. British Veterinary Journal, 116, 105-114.
- KATZ, J. B., BARBET, A. F., MAHAN, S. M., KUMBULA, D., LOCKHART, J. M., KEEL, M. K., DAWSON, J. E., OLSON, J. G., & EWING, S. A. 1996. A recombinant antigen from the heartwater agent (Cowdria ruminatium) reactive with antibodies in some southeastern United States white-tailed deer (Odocoileus virginianus), but not cattle, sera. Journal of Wildlife Diseases, 32, 424-430.
- KELLERMAN, T. S., COETZER, J. A. W., & NAUDÉ, T. W. 1988. Chapter 2: Central nervous system., In Kellerman, T. S., Coetzer, J. A. W., & Naudé, T. W. (eds.), Plant Poisonings and Mycotoxicoses of Livestock in Southern Africa. Oxford University Press, Cape Town.
- KELLY, P. J., LUCAS, H., YOWELL, C., BEATI, L., DAME, J., URDAZ-RODRIGUEZ, J., & MAHAN, S. 2011. Ehrlichia ruminantium in Amblyomma variegatum and domestic ruminants in the Caribbean. Journal of Medical Entomology, 48, 485-488.
- KOCAN, K. M. & BEZUIDENHOUT, J. D. 1987. Morphology and development of Cowdria ruminantium in Amblyomma ticks. Onderstepoort Journal of Veterinary Research, 54, 177-182.
- KOCAN, K. M., BEZUIDENHOUT, J. D., & HART, A. 1987. Ultrastructural features of Cowdria ruminantium in midgut epithelial cells and salivary glands of nymphal Amblyomma hebraeum. Onderstepoort Journal of Veterinary Research, 54, 87-92.
- KOCAN, K. M., MORZARIA, S. P., VOIGT, W. P., KIARIE, J., & IRVIN, A. D. 1987. Demonstration of colonies of Cowdria ruminantium in midgut epithelial cells of Amblyomma variegatum. American Journal of Veterinary Research, 48, 356-360.
- KOCAN, K. M., NORVAL, R. A., & DONOVAN, P. L. 1993. Development and transmission of Cowdria ruminantium by Amblyomma males transferred from infected to susceptible sheep. Revue d’Élevage et de Médecine Vétérinaire des Pays Tropicaux, 46, 183-188.
- KURIAKOSE, J. A., ZHANG, X., LUO, T., & MCBRIDE, J. W. 2012. Molecular basis of antibody mediated immunity against Ehrlichia chaffeensis involves species-specific linear epitopes in tandem repeat proteins. Microbes Infect, 14, 1054-1063.
- LAWRENCE, J. A., TJORNEHOJ, K., WHITELAND, A. P., & KAFUWA, P. T. 1995. The serological response to heartwater immunization in cattle is an indicator of protective immunity. Revue d’Élevage et de Médecine Vétérinaire des Pays Tropicaux, 48, 63-65.
- LEW-TABOR, A. E. & RODRIGUEZ VALLE, M. 2016. A review of reverse vaccinology approaches for the development of vaccines against ticks and tick borne diseases. Ticks and Tick Borne Diseases, 7, 573-585.
- LIEBENBERG, J., PRETORIUS, A., FABER, F. E., COLLINS, N. E., ALLSOPP, B. A., & VAN KLEEF, M. 2012. Identification of Ehrlichia ruminantium proteins that activate cellular immune responses using a reverse vaccinology strategy. Veterinary Immunology and Immunopathology, 145, 340-349.
- LOFTIS, D. A., REEVES, W. K., SPURLOCK, J. P., MAHAN, S. M., TROUGHTON, D. R., DASCH, G. A., & LEVIN, M. L. 2006. Infection of a goat with a tick-transmitted Ehrlichia from Georgia, U.S.A., that is closely related to Ehrlichia ruminantium. Journal of Vector Ecology, 31, 213-223.
- LOGAN, L. L. 1987. Cowdria ruminantium: stability and preservation of the organism. Onderstepoort Journal of Veterinary Research, 54, 187-91.
- LOGAN, L. L., HOLLAND, C. J., MEBUS, C. A., & RISTIC, M. 1986. Serological relationship between Cowdria ruminantium and certain Ehrlichia. Veterinary Record, 119, 458-459.
- LOGAN, L. L., WHYARD, T. C., QUINTERO, J. C., & MEBUS, C. A. 1987. The development of Cowdria ruminantium in neutrophils. Onderstepoort Journal of Veterinary Research, 54, 197-204.
- LOUNSBURY, C P. 1900. Tick heartwater experiments. Agricultural Journal of the Cape of Good Hope, 16, 682-687.
- LOUW, E., BRAYTON, K. A., COLLINS, N. E., PRETORIUS, A., VAN STRIJP, F., & ALLSOPP, B. A. 2002. Sequencing of a 15-kb Ehrlichia ruminantium clone and evaluation of the cpg1 open reading frame for protection against heartwater. Annals of the New York Academy of Sciences, 969, 147-150.
- MACKENZIE, P. K. & MCHARDY, N. 1987. Cowdria ruminantium infection in the mouse: a review. Onderstepoort Journal of Veterinary Research, 54, 267-269.
- MACKENZIE, P. K. I. & VAN ROOYEN, R. E. 1981. The isolation and culture of Cowdria ruminantium in albino mice. In "Tick Biology and Control"; Proceedings of an International Conference, Whitehead, G. B. & Gibson, J. D. (eds.), Tick Research Unit, Rhodes University, Grahamstown, South Africa. 33-39
- MAHAN, S. M., ANDREW, H. R., TEBELE, N., BURRIDGE, M. J., & BARBET, A. F. 1995. Immunisation of sheep against heartwater with inactivated Cowdria ruminantium. Research in Veterinary Science, 58, 46-49.
- MAHAN, S. M., BARBET, A. F., & BURRIDGE, M. J. 2003. Development of improved vaccines for heartwater. Developments in Biologicals, 114, 137-145.
- MAHAN, S. M., MCGUIRE, T. C., SEMU, S. M., BOWIE, M. V., JONGEJAN, F., RURANGIRWA, F. R., & BARBET, A. F. 1994. Molecular cloning of a gene encoding the immunogenic 21 kDa protein of Cowdria ruminantium. Microbiology, 140 (Pt 8), 2135-2142.
- MAHAN, S. M., PETER, T. F., SEMU, S. M., SIMBI, B. H., NORVAL, R. A., & BARBET, A. F. 1995. Laboratory reared Amblyomma hebraeum and Amblyomma variegatum ticks differ in their susceptibility to infection with Cowdria ruminantium. Epidemiology and Infection, 115, 345-353.
- MAHAN, S. M., PETER, T. F., SIMBI, B. H., & BURRIDGE, M. J. 1998. PCR detection of Cowdria ruminantium infection in ticks and animals from heartwater-endemic regions of Zimbabwe. Annals of the New York Academy of Sciences, 849, 85-87.
- MAHAN, S. M., PETER, T. F., SIMBI, B. H., KOCAN, K., CAMUS, E., BARBET, A. F., & BURRIDGE, M. J. 2000. Comparison of efficacy of American and African Amblyomma ticks as vectors of heartwater (Cowdria ruminantium) infection by molecular analyses and transmission trials. Journal of Parasitology, 86, 44-49.
- MAHAN, S. M., SIMBI, B. H., & BURRIDGE, M. J. 2004. The pCS20 PCR assay for Ehrlichia ruminantium does not cross-react with the novel deer ehrlichial agent found in white-tailed deer in the United States of America. Onderstepoort Journal of Veterinary Research, 71, 99-105.
- MAHAN, S. M., SMITH, G. E., KUMBULA, D., BURRIDGE, M. J., & BARBET, A. F. 2001. Reduction in mortality from heartwater in cattle, sheep and goats exposed to field challenge using an inactivated vaccine. Veterinary Parasitology, 97, 295-308.
- MAHAN, S. M., WAGHELA, S. D., MCGUIRE, T. C., RURANGIRWA, F. R., WASSINK, L. A., & BARBET, A. F. 1992. A cloned DNA probe for Cowdria ruminantium hybridizes with eight heartwater strains and detects infected sheep. Journal of Clinical Microbiology, 30, 981-986.
- MAILLARD, J. C. & MAILLARD, N. 1998. Historique du peuplement bovin et de l'introduction de la tique Amblyomma variegatum dans les îles françaises des Antilles: Synthèse bibliographique. Ethnozootechnie, 1, 19-36.
- MARCELINO, I., DE ALMEIDA, A. M., BRITO, C., MEYER, D. F., BARRETO, M., SHEIKBOUDOU, C., FRANCO, C. F., MARTINEZ, D., LEFRANCOIS, T., VACHIERY, N., CARRONDO, M. J., COELHO, A. V., & ALVES, P. M. 2012. Proteomic analyses of Ehrlichia ruminantium highlight differential expression of MAP1-family proteins. Veterinary Microbiology, 156, 305-314.
- MARCELINO, I., LEFRANCOIS, T., MARTINEZ, D., GIRAUD-GIRARD, K., APRELON, R., MANDONNET, N., GAUCHERON, J., BERTRAND, F., & VACHIERY, N. 2015. A user-friendly and scalable process to prepare a ready-to-use inactivated vaccine: the example of heartwater in ruminants under tropical conditions. Vaccine, 33, 678-685.
- MARCELINO, I., SOUSA, M. F., VERISSIMO, C., CUNHA, A. E., CARRONDO, M. J., & ALVES, P. M. 2006. Process development for the mass production of Ehrlichia ruminantium. Vaccine, 24, 1716-1725. Epub 2005 Oct 14.
- MARCELINO, I., VACHIERY, N., AMARAL, A. I., ROLDAO, A., LEFRANCOIS, T., CARRONDO, M. J., ALVES, P. M., & MARTINEZ, D. 2007. Effect of the purification process and the storage conditions on the efficacy of an inactivated vaccine against heartwater. Vaccine, 25, 4903-4913.
- MARCELINO, I., VENTOSA, M., PIRES, E., MULLER, M., LISACEK, F., LEFRANCOIS, T., VACHIERY, N., & COELHO, A. V. 2015. Comparative Proteomic Profiling of Ehrlichia ruminantium Pathogenic Strain and Its High-Passaged Attenuated Strain Reveals Virulence and Attenuation-Associated Proteins. PLoS One, 10, e0145328.
- MARCELINO, I., VERISSIMO, C., SOUSA, M. F., CARRONDO, M. J., & ALVES, P. M. 2005. Characterization of Ehrlichia ruminantium replication and release kinetics in endothelial cell cultures. Veterinary Microbiology, 110, 87-96.
- MARTINEZ, D. 1997. Analysis of the immune response of ruminants to Cowdria ruminantium infection. University of Utrecht.
- MARTINEZ, D. 1997. Unpublished data. Cited In: Analysis of the immune response of ruminants to Cowdria ruminantium infection. University of Utrecht.
- MARTINEZ, D. 2001. Personal Communication.
- MARTINEZ, D., MAILLARD, J. C., COISNE, S., SHEIKBOUDOU, C., & BENSAID, A. 1994. Protection of goats against heartwater acquired by immunisation with inactivated elementary bodies of Cowdria ruminantium. Veterinary Immunology and Immunopathology, 41, 153-163.
- MARTINEZ, D., SHEIKBOUDOU, C., COURAUD, P. O., & BENSAID, A. 1993. In vitro infection of bovine brain endothelial cells by Cowdria ruminantium. Research in Veterinary Science, 55, 258-260.
- MARTINEZ, D., SWINKELS, J., CAMUS, E., & JONGEJAN, F. 1990. Comparaison de trois antigènes pour le sérodiagnostic de la cowdriose par immunofluorescence indirecte. Revue d’Élevage et de Médecine Vétérinaire des Pays Tropicaux, 43, 159-166.
- MARTINEZ, D., VACHIERY, N., STACHURSKI, F., KANDASSAMY, Y., RALINIAINA, M., APRELON, R., & GUEYE, A. 2004. Nested PCR for detection and genotyping of Ehrlichia ruminantium: use in genetic diversity analysis. Annals of the New York Academy of Sciences, 1026, 106-113.
- MATHERON, G., BARRÉ, N., CAMUS, E., & GOGUE, J. 1987. Genetic resistance of Guadeloupe native goats to heartwater. Onderstepoort Journal of Veterinary Research, 54, 337-340.
- MBOLOI, M. M., BEKKER, C. P., KRUITWAGEN, C., GREINER, M., & JONGEJAN, F. 1999. Validation of the indirect MAP1B enzyme-linked immunosorbent assay for diagnosis of experimental Cowdria ruminantium infection in small ruminants. Clinical and Diagnostic Laboratory Immunology, 6, 66-72.
- MDLADLA, K., DZOMBA, E. F., & MUCHADEYI, F. C. 2016. Seroprevalence of Ehrlichia ruminantium antibodies and its associated risk factors in indigenous goats of South Africa. Preventive Veterinary Medicine, 125, 99-105.
- MELTZER, M. I., NORVAL, R. A., & DONACHIE, P. L. 1995. Effects of tick infestation and tick-borne disease infections (heartwater, anaplasmosis and babesiosis) on the lactation and weight gain of Mashona cattle in south-eastern Zimbabwe. Tropical Animal Health and Production, 27, 129-144.
- MERKL, R. 2004. SIGI: score-based identification of genomic islands. BMC Bioinformatics, 5, 22.
- METELERKAMP, N. C., LITTHAUER, D., NAUDE, R. J., OELOFSEN, W., & GRUSS, B. 1986. The orange-yellow pigment of heartwater-infected angora goats. Comparative Biochemistry and Physiology. B: Comparative Biochemistry, 84, 299-304.
- MOSHKOVSKI, S. D. 1947. Comments by readers. Science, 106, 62.
- MOUMENE, A., MARCELINO, I., VENTOSA, M., GROS, O., LEFRANCOIS, T., VACHIERY, N., MEYER, D. F., & COELHO, A. V. 2015. Proteomic Profiling of the Outer Membrane Fraction of the Obligate Intracellular Bacterial Pathogen Ehrlichia ruminantium. PLoS One, 10, e0116758.
- MTSHALI, K., KHUMALO, Z., NAKAO, R., GRAB, D. J., SUGIMOTO, C., & THEKISOE, O. 2016. Molecular detection of zoonotic tick-borne pathogens from ticks collected from ruminants in four South African provinces. Journal of Veterinary Medical Science, 77, 1573-9.
- MUKHEBI, A. W., CHAMBOKO, T., O'CALLAGHAN, C. J., PETER, T. F., KRUSKA, R. L., MEDLEY, G. F., MAHAN, S. M., & PERRY, B. D. 1999. An assessment of the economic impact of heartwater (Cowdria ruminantium infection) and its control in Zimbabwe. Preventive Veterinary Medicine, 39, 173-189.
- MURAMATSU, Y., UKEGAWA, S. Y., RAHIM, A., EL HUSSEIN, M., ABDEL RAHMAN, M. B., ABDEL GABBAR, K. M., CHITAMBO, A. M., KOMIYA, T., MWASE, E. T., MORITA, C., & TAMURA, Y. 2005. Ehrlichia ruminantium, Sudan. Emerging Infectious Diseases, 11, 1792-1793.
- MWANGI, D. M., MAHAN, S. M., NYANJUI, J. K., TARACHA, E. L., & MCKEEVER, D. J. 1998. Immunization of cattle by infection with Cowdria ruminantium elicits T lymphocytes that recognize autologous, infected endothelial cells and monocytes. Infection and Immunity, 66, 1855-1860.
- MWANGI, D. M., MCKEEVER, D. J., NYANJUI, J. K., BARBET, A. F., & MAHAN, S. M. 1998. Major antigenic proteins 1 and 2 of Cowdria ruminantium are targets for T-lymphocyte responses of immune cattle. Annals of the New York Academy of Sciences, 849, 372-374.
- NAKAO, R., JONGEJAN, F., & SUGIMOTO, C. 2016. Draft Genome Sequences of Three Strains of Ehrlichia ruminantium, a Tick-Borne Pathogen of Ruminants, Isolated from Zimbabwe, The Gambia, and Ghana. Genome Announcements, 4, e00453-16.
- NAKAO, R., MAGONA, J. W., ZHOU, L., JONGEJAN, F., & SUGIMOTO, C. 2011. Multi-locus sequence typing of Ehrlichia ruminantium strains from geographically diverse origins and collected in Amblyomma variegatum from Uganda. Parasites and Vectors, 4, 137.
- NAKAO, R., STROMDAHL, E. Y., MAGONA, J. W., FABURAY, B., NAMANGALA, B., MALELE, I., INOUE, N., GEYSEN, D., KAJINO, K., JONGEJAN, F., & SUGIMOTO, C. 2010. Development of loop-mediated isothermal amplification (LAMP) assays for rapid detection of Ehrlichia ruminantium. BMC Microbiology, 10, 296.
- NEITZ, A. W. H., VILJOEN, G. J., BEZUIDENHOUT, J. D., OBEREM, P. T., VISSER, L., & VERMEULEN, N. M. J. 1986. Detection of Cowdria ruminantium antigen and antibody during the course of hertwater disease in sheep by means of an enzyme-linked immunosorbent assay. Onderstepoort Journal of Veterinary Research, 53, 205-207.
- NEITZ, W. O. 1935. The blesbuck (Damaliscus albifrons) and the Black-Wilde Beest (Conochaetes gnu) as carriers of heartwater. Onderstepoort Journal of Veterinary Science and Animal Industry, 5, 35-40.
- NEITZ, W. O. 1939. The immunity in heartwater. Onderstepoort Journal of Veterinary Science and Animal Industry, 13, 245-283.
- NEITZ, W. O. 1940. Uleron in the treatment of heartwater. Journal of the South African Veterinary Medical Association, 11, 15.
- NEITZ, W. O. 1947. The transmission of heartwater by Amblyomma pomposum, Dönitz, 1909. South African Science, 1, 83.
- NEITZ, W. O. 1967. The epidemiological pattern of viral, protophytal and protozoal zoonoses in relation to game preservation in South Africa. Journal of the South African Veterinary Medical Association, 38, 129-141.
- NEITZ, W. O. 1968. Heartwater. Bulletin de l'Office International des Epizooties, 70, 329-336.
- NEITZ, W. O. & ALEXANDER, R A. 1941. The immunisation of calves against heartwater. Journal of the South African Veterinary Medical Association, 12, 103-111.
- NEITZ, W. O., ALEXANDER, R. A., & ADELAAR, T. F. 1947. Studies on immunity in heartwater. Onderstepoort Journal of Veterinary Science and Animal Industry, 21, 243-252.
- NOH, S. M., BRAYTON, K. A., BROWN, W. C., NORIMINE, J., MUNSKE, G. R., DAVITT, C. M., & PALMER, G. H. 2008. Composition of the surface proteome of Anaplasma marginale and its role in protective immunity induced by outer membrane immunization. Infection and Immunity, 76, 2219-2226.
- NORVAL, R. A., ANDREW, H. R., & YUNKER, C. E. 1990. Infection rates with Cowdria ruminantium of nymphs and adults of the bont tick Amblyomma hebraeum collected in the field in Zimbabwe. Veterinary Parasitology, 36, 277-283.
- NORVAL, R. A. I. & LAWRENCE, J. A. 1979. The control of heartwater in Zimbabwe, Rhodesia. Zimbabwe Rhodesia Agricultural Journal, 76, 161-165.
- NYIKA, A., BARBET, A. F., BURRIDGE, M. J., & MAHAN, S. M. 2002. DNA vaccination with map1 gene followed by protein boost augments protection against challenge with Cowdria ruminantium, the agent of heartwater. Vaccine, 20, 1215-1225.
- NYIKA, A., MAHAN, S. M., BURRIDGE, M. J., MCGUIRE, T. C., RURANGIRWA, F., & BARBET, A. F. 1998. A DNA vaccine protects mice against the rickettsial agent Cowdria ruminantium. Parasite Immunology, 20, 111-119.
- O'LEARY, N. A., WRIGHT, M. W., BRISTER, J. R., CIUFO, S., HADDAD, D., MCVEIGH, R., RAJPUT, B., ROBBERTSE, B., SMITH-WHITE, B., AKO-ADJEI, D., ASTASHYN, A., BADRETDIN, A., BAO, Y., BLINKOVA, O., BROVER, V., CHETVERNIN, V., CHOI, J., COX, E., ERMOLAEVA, O., FARRELL, C. M., GOLDFARB, T., GUPTA, T., HAFT, D., HATCHER, E., HLAVINA, W., JOARDAR, V. S., KODALI, V. K., LI, W., MAGLOTT, D., MASTERSON, P., MCGARVEY, K. M., MURPHY, M. R., O'NEILL, K., PUJAR, S., RANGWALA, S. H., RAUSCH, D., RIDDICK, L. D., SCHOCH, C., SHKEDA, A., STORZ, S. S., SUN, H., THIBAUD-NISSEN, F., TOLSTOY, I., TULLY, R. E., VATSAN, A. R., WALLIN, C., WEBB, D., WU, W., LANDRUM, M. J., KIMCHI, A., TATUSOVA, T., DICUCCIO, M., KITTS, P., MURPHY, T. D., & PRUITT, K. D. 2016. Reference sequence (RefSeq) database at NCBI: current status, taxonomic expansion, and functional annotation. Nucleic Acids Research, 44, D733-745.
- OBEREM, P. T. & BEZUIDENHOUT, J. D. 1987. Heartwater in hosts other than domestic ruminants. Onderstepoort Journal of Veterinary Research, 54, 271-275.
- OBEREM, P. T. & BEZUIDENHOUT, J. D. 1987. The production of heartwater vaccine. Onderstepoort Journal of Veterinary Research, 54, 485-488.
- OHASHI, N., RIKIHISA, Y., & UNVER, A. 2001. Analysis of transcriptionally active gene clusters of major outer membrane protein multigene family in Ehrlichia canis and E. chaffeensis. Infection and Immunity, 69, 2083-2091.
- OLSEN, G. J. & WOESE, C. R. 1993. Ribosomal RNA: a key to phylogeny. FASEB Journal, 7, 113-123.
- OMONDI, D., MASIGA, D. K., FIELDING, B. C., KARIUKI, E., AJAMMA, Y. U., MWAMUYE, M. M., OUSO, D. O., & VILLINGER, J. 2017. Molecular Detection of Tick-Borne Pathogen Diversities in Ticks from Livestock and Reptiles along the Shores and Adjacent Islands of Lake Victoria and Lake Baringo, Kenya. Frontiers in Veterinary Science, 4, 73.
- OWEN, N. C., LITTLEJOHN, A., KRUGER, J. M., & ERASMUS, B. J. 1973. Physiopathological features of heartwater in sheep. Journal of the South African Veterinary Association, 44, 397-403.
- PALMER, G. H., EID, G., BARBET, A. F., MCGUIRE, T. C., & MCELWAIN, T. F. 1994. The immunoprotective Anaplasma marginale major surface protein 2 is encoded by a polymorphic multigene family. Infection and Immunity, 62, 3808-3816.
- PASCUCCI, I., CONTE, A., & SCACCHIA, M. 2007. Use of geographic information systems to identify areas at risk of introducing Amblyomma variegatum and A. hebraeum to Italy. Veterinaria Italiana, 43, 655-661.
- PEDREGAL, A., DE SOUSA, D. R., NGUYEN, H. N., DAS NEVES, E. A., LOWAGIE, S., MARIQUE, T., KAGYE, N., GUERRA, I. T., KAMBA, Y., TOTTE, P., VACHIERY, N., LEFRANCOIS, T., MARTINEZ, D., YOURASSOWSKY, C., CALLENS, N., MONNOM, O., DUBOIS, F., & WERENNE, J. 2008. Toward prevention of cowdriosis. Annals of the New York Academy of Sciences, 1149, 286-91.
- PEIXOTO, C., MARCELINO, I., AMARAL, A. I., CARRONDO, M. J. T., & ALVES, P. M. 2007. Purification by membrane technology of an intracellular Ehrlichia ruminantium candidate vaccine against heartwater. Process Biochemistry, 42, 1084-1089.
- PEIXOTO, C. C., MARCELINO, I., VACHIERY, N., BENSAID, A., MARTINEZ, D., CARRONDO, M. J., & ALVES, P. M. 2005. Quantification of Ehrlichia ruminantium by real time PCR. Veterinary Microbiology, 107, 273-278. Epub 2005 Feb 24.
- PEREGRINE, A. S. 1994. Chemotherapy and delivery systems: haemoparasites. Veterinary Parasitology, 54, 223-248.
- PETER, T. F., BARBET, A. F., ALLEMAN, A. R., SIMBI, B. H., BURRIDGE, M. J., & MAHAN, S. M. 2000. Detection of the agent of heartwater, Cowdria ruminantium, in Amblyomma ticks by PCR: validation and application of the assay to field ticks. Journal of Clinical Microbiology, 38, 1539-1544.
- Peter, T. F., Bryson, N. R., Perry, B. D., O'Callaghan, C. J., Medley, G. F., Smith, G. E., Mlambo, G., Horak, I. G., Burridge, M. J., & Mahan, S. M. 1999. Cowdria ruminantium infection in ticks in the Kruger National Park. Veterinary Record, 145, 304-307.
- PETER, T. F., BURRIDGE, M. J., & MAHAN, S. M. 2000. Competence of the African tortoise tick, Amblyomma marmoreum (Acari: Ixodidae), as a vector of the agent of heartwater (Cowdria ruminantium). Journal of Parasitology, 86, 438-441.
- PETER, T. F., DEEM, S. L., BARBET, A. F., NORVAL, R. A., SIMBI, B. H., KELLY, P. J., & MAHAN, S. M. 1995. Development and evaluation of PCR assay for detection of low levels of Cowdria ruminantium infection in Amblyomma ticks not detected by DNA probe. Journal of Clinical Microbiology, 33, 166-172.
- PETER, T. F., PERRY, B. D., O'CALLAGHAN, C. J., MEDLEY, G. F., MLAMBO, G., BARBET, A. F., & MAHAN, S. M. 1999. Prevalence of Cowdria ruminantium infection in Amblyomma hebraeum ticks from heartwater-endemic areas of Zimbabwe. Epidemiology and Infection, 123, 309-316.
- PETNEY, T. N., HORAK, I. G., & RECHAV, Y. 1987. The ecology of the African vectors of heartwater, with particular reference to Amblyomma hebraeum and Amblyomma variegatum. Onderstepoort Journal of Veterinary Research, 54, 381-395.
- PIENAAR, J. G. 1970. Electron microscopy of Cowdria (Rickettsia) ruminantium (Cowdry, 1926) in the endothelial cells of the vertebrate host. Onderstepoort Journal of Veterinary Research, 37, 67-78.
- PIENAAR, J. G., BASSON, P. A., & VAN DER MERWE, J. L. DE B. 1966. Studies on the pathology of Heartwater Cowdria (Rickettsia) ruminantium, COWDRY 1926. 1. Neuropathological changes. Onderstepoort Journal of Veterinary Research, 33, 115-138.
- PILET, H., VACHIERY, N., BERRICH, M., BOUCHOUICHA, R., DURAND, B., PRUNEAU, L., PINARELLO, V., SALDANA, A., CARASCO-LACOMBE, C., LEFRANCOIS, T., MEYER, D. F., MARTINEZ, D., BOULOUIS, H. J., & HADDAD, N. 2012. A new typing technique for the Rickettsiales Ehrlichia ruminantium: multiple-locus variable number tandem repeat analysis. Journal of Microbiological Methods, 88, 205-211.
- POOLE, J. D. H. 1962. Flock immunisation of sheep and goats against heartwater. Part 1. Investigations regarding routine flock immunisation of sheep. Journal of the South African Veterinary Medical Association, 33, 35-41.
- POOLE, J. D. H. 1962. Flock immunisation of sheep and goats against heartwater. Part 2. Preliminary experiments on flock immunisation of goats. Journal of the South African Veterinary Medical Association, 33, 357-362.
- POSTIGO, M., TAOUFIK, A., BELL-SAKYI, L., BEKKER, C. P., DE VRIES, E., MORRISON, W. I., & JONGEJAN, F. 2008. Host cell-specific protein expression in vitro in Ehrlichia ruminantium. Veterinary Microbiology, 128, 136-147.
- POSTIGO, M., TAOUFIK, A., BELL-SAKYI, L., DE VRIES, E., MORRISON, W. I., & JONGEJAN, F. 2007. Differential transcription of the major antigenic protein 1 multigene family of Ehrlichia ruminantium in Amblyomma variegatum ticks. Veterinary Microbiology, 122, 298-305.
- PRETORIUS, A., COLLINS, N. E., STEYN, H. C., VAN STRIJP, F., VAN KLEEF, M., & ALLSOPP, B. A. 2007. Protection against heartwater by DNA immunisation with four Ehrlichia ruminantium open reading frames. Vaccine, 25, 2316-2324.
- PRETORIUS, A., LIEBENBERG, J., LOUW, E., COLLINS, N. E., & ALLSOPP, B. A. 2010. Studies of a polymorphic Ehrlichia ruminantium gene for use as a component of a recombinant vaccine against heartwater. Vaccine, 28, 3531-3539.
- PRETORIUS, A., VAN KLEEF, M., COLLINS, N. E., TSHIKUDO, N., LOUW, E., FABER, F. E., VAN STRIJP, M. F., & ALLSOPP, B. A. 2008. A heterologous prime/boost immunisation strategy protects against virulent Ehrlichia ruminantium Welgevonden needle challenge but not against tick challenge. Vaccine, 26, 4363-4371.
- PRETZMAN, C., RALPH, D., STOTHARD, D. R., FUERST, P. A., & RIKIHISA, Y. 1995. 16S rRNA gene sequence of Neorickettsia helminthoeca and its phylogenetic alignment with members of the genus Ehrlichia. International Journal of Systematic Bacteriology, 45, 207-211.
- PRITCHARD, J. K., STEPHENS, M., & DONNELLY, P. 2000. Inference of population structure using multilocus genotype data. Genetics, 155, 945-959.
- PROVOST, A. & BEZUIDENHOUT, J. D. 1987. The historical background and global importance of heartwater. Onderstepoort Journal of Veterinary Research, 54, 165-169.
- PROZESKY, L. 1987. Diagnosis of heartwater at post-mortem in ruminants and the confirmation of Cowdria ruminantium in mice. Onderstepoort Journal of Veterinary Research, 54, 301-303.
- PROZESKY, L. 1987. Heartwater. The morphology of Cowdria ruminantium and its staining characteristics in the vertebrate host and in vitro. Onderstepoort Journal of Veterinary Research, 54, 173-176.
- PROZESKY, L. 1987. The pathology of heartwater. III. A review. Onderstepoort Journal of Veterinary Research, 54, 281-286.
- PROZESKY, L., BEZUIDENHOUT, J. D., & PATERSON, C. L. 1986. Heartwater: an in vitro study of the ultrastructure of Cowdria ruminatium. Onderstepoort Journal of Veterinary Research, 53, 153-159.
- PROZESKY, L & DU PLESSIS, J L. 1985. Heartwater in Angora goats. 2. A pathological study of artificially infected, treated and untreated goats. Onderstepoort Journal of Veterinary Research, 52, 13-19.
- PROZESKY, L & DU PLESSIS, J L. 1985. The pathology of heartwater. 2. A study of the lung lesions in sheep and goats infected with the Ball3, strain of Cowdria ruminantium. Onderstepoort Journal of Veterinary Research, 52, 81-85.
- PROZESKY, L. & DU PLESSIS, J. L. 1985. The pathology of heartwater. I. A study of mice infected with the Welgevonden strain of Cowdria ruminantium. Onderstepoort Journal of Veterinary Research, 52, 71-79.
- PRUNEAU, L., EMBOULE, L., GELY, P., MARCELINO, I., MARI, B., PINARELLO, V., SHEIKBOUDOU, C., MARTINEZ, D., DAIGLE, F., LEFRANCOIS, T., MEYER, D. F., & VACHIERY, N. 2012. Global gene expression profiling of Ehrlichia ruminantium at different stages of development. FEMS Immunology and Medical Microbiology, 64, 66-73.
- PURCHASE, H. S. 1945. A simple and rapid method for demonstrating Rickettsia ruminantium (COWDRY, 1925) in heartwater brains. Veterinary Record, 57, 413-414.
- PURNELL, R. E. 1987. Development of a prophylactic regime using Terramycin/LA to assist in the introduction of susceptible cattle into heartwater endemic areas of Africa. Onderstepoort Journal of Veterinary Research, 54, 509-512.
- PURNELL, R. E., GUNTER, T. D., & SCHRODER, J. 1989. Development of a prophylactic regime using long-acting tetracycline for the control of redwater and heartwater in susceptible cattle moved into an endemic area. Tropical Animal Health and Production, 21, 11-19.
- RALINIAINA, M., MEYER, D. F., PINARELLO, V., SHEIKBOUDOU, C., EMBOULE, L., KANDASSAMY, Y., ADAKAL, H., STACHURSKI, F., MARTINEZ, D., LEFRANCOIS, T., & VACHIERY, N. 2010. Mining the genetic diversity of Ehrlichia ruminantium using map genes family. Veterinary Parasitology, 167, 187-195.
- RAMISSE, J. & UILENBERG, G. 1970. Preservation of a Cowdria ruminantium strain by freezing. Revue d’Élevage et de Médecine Vétérinaire des Pays Tropicaux, 23, 313-316.
- REDDY, G. R., SULSONA, C. R., HARRISON, R. H., MAHAN, S. M., BURRIDGE, M. J., & BARBET, A. F. 1996. Sequence heterogeneity of the major antigenic protein 1 genes from Cowdria ruminantium isolates from different geographical areas. Clinical and Diagnostic Laboratory Immunology, 3, 417-422.
- RISTIC, M. & HUXSOLL, D. L. 1984. Genus 5. Cowdria Moshkovski, p. 709-710. In Bergey's Manual of Systematic Bacteriology. Williams & Wilkins, Baltimore.
- ROBINSON, J. B., EREMEEVA, M. E., OLSON, P. E., THORNTON, S. A., MEDINA, M. J., SUMNER, J. W., & DASCH, G. A. 2009. New approaches to detection and identification of Rickettsia africae and Ehrlichia ruminantium in Amblyomma variegatum (Acari: Ixodidae) ticks from the Caribbean. Journal of Medical Entomology, 46, 942-951.
- ROSSOUW, M., NEITZ, A. W., DE WAAL, D. T., DU PLESSIS, J. L., VAN GAS, L., & BRETT, S. 1990. Identification of the antigenic proteins of Cowdria ruminantium. Onderstepoort Journal of Veterinary Research, 57, 215-221.
- RUFF, G., MAILLARD, J. C., CAMUS, E., DEPRES, E., & MATHERON, G. 1993. Occurrence of caprine leucocyte antigens (CLA) in Creole goats susceptible/resistant to heartwater. Revue d’Élevage et de Médecine Vétérinaire des Pays Tropicaux, 46, 205-207.
- SAYLER, K. A., LOFTIS, A. D., MAHAN, S. M., & BARBET, A. F. 2016. Development of a Quantitative PCR Assay for Differentiating the Agent of Heartwater Disease, Ehrlichia ruminantium, from the Panola Mountain Ehrlichia. Transboundary and Emerging Diseases, 63, e260-e269.
- SCHREUDER, B. E. 1980. A simple technique for the collection of brain samples for the diagnosis of heartwater. Tropical Animal Health and Production, 12, 25-29.
- SCHUBERT-UNKMEIR, A. & CHRISTODOULIDES, M. 2013. Genome-based bacterial vaccines: current state and future outlook. BioDrugs, 27, 419-430.
- SEBATJANE, S. I., PRETORIUS, A., LIEBENBERG, J., STEYN, H., & VAN KLEEF, M. 2010. In vitro and in vivo evaluation of five low molecular weight proteins of Ehrlichia ruminantium as potential vaccine components. Veterinary Immunology and Immunopathology, 137, 217-225.
- SEMU, S. M., PETER, T. F., MUKWEDEYA, D., BARBET, A. F., JONGEJAN, F., & MAHAN, S. M. 2001. Antibody responses to MAP 1B and other Cowdria ruminantium antigens are down regulated in cattle challenged with tick-transmitted heartwater. Clinical and Diagnostic Laboratory Immunology, 8, 388-396.
- SHAKESPEARE, A. S., REYERS, F., VAN AMSTEL, S. R., SWAN, G. E., & VAN DEN BERG, J. S. 1998. Treatment of heartwater: potential adverse effects of furosemide administration on certain homeostatic parameters in normal sheep. Journal of the South African Veterinary Association, 69, 129-136.
- SHKAP, V., DE VOS, A. J., ZWEYGARTH, E., & JONGEJAN, F. 2007. Attenuated vaccines for tropical theileriosis, babesiosis and heartwater: the continuing necessity. Trends in Parasitology, 23, 420-426.
- SIMBI, B. H., BOWIE, M. V., MCGUIRE, T. C., BARBET, A. F., & MAHAN, S. M. 2006. Evaluation of E. ruminantium Genes in DBA/2 Mice as Potential DNA Vaccine Candidates for Control of Heartwater. Annals of the New York Academy of Sciences, 1078, 424-437.
- SIMBI, B. H., PETER, T. F., BURRIDGE, M. J., & MAHAN, S. M. 2003. Comparing the detection of exposure to Ehrlichia ruminantium infection on a heartwater-endemic farm by the pCS20 polymerase chain reaction assay and an indirect MAP1-B enzyme linked immunosorbent assay. Onderstepoort Journal of Veterinary Research, 70, 231-235.
- SIMPSON, B. C., LINDSAY, M. S., MORRIS, J. R., MUIRHEAD, F. S., POLLOCK, A., PRICHARD, S. G., STANLEY, H. G., THIRLWELL, G. R., HUNTER, A. G., & BRADLEY, J. 1987. Protection of cattle against heartwater in Botswana: comparative efficacy of different methods against natural and blood-derived challenges. Veterinary Record, 120, 135-138.
- SIMPSON, R. M. & WILEY, A. J. 1951. Experiments with aureomycin hydrocloride - The use of aureomycin in the treatment of heartwater. Annual Report of the Veterinary Department of Kenya for 1949, 23-27.
- SMITH, G. E., ANDERSON, E. C., BURRIDGE, M. J., PETER, T. F., & MAHAN, S. M. 1998. Growth of Cowdria ruminantium in tissue culture endothelial cell lines from wild African mammals. Journal of Wildlife Diseases, 34, 297-304.
- SPREULL, J. 1904. Heartwater inoculation experiments. Agricultural Journal of the Cape of Good Hope, 24, 433-442.
- SPREULL, J. 1922. Heartwater. Journal of the Department of Agriculture; Union of South Africa, 4, 236-245.
- STACHURSKI, F., TORTOSA, P., RAHAJARISON, P., JACQUET, S., YSSOUF, A., & HUBER, K. 2013. New data regarding distribution of cattle ticks in the south-western Indian Ocean islands. Veterinary Research, 44, 79.
- STAMPA, S. 1969. Experiences with heartwater eradication. In Proceedings of the Symposium on Biology and Control of ticks in southern Africa, Rhodes University, Grahamstown, South Africa. 133-150
- STECK, W. 1928. Pathological studies on heartwater. 13th and 14th Report of the Director of Veterinary Education and Research, 283-305 and 2 plates.
- STEYN, H. C., PRETORIUS, A., MCCRINDLE, C. M., STEINMANN, C. M., & VAN KLEEF, M. 2008. A quantitative real-time PCR assay for Ehrlichia ruminantium using pCS20. Veterinary Microbiology, 131, 258-265.
- SWAI, E. S., MOSHY, W., MTUI, P. F., BWANGA, S., MACHANGE, G., & SANKA, P. 2009. Serological survey of antibodies to Ehrlichia ruminantium in small ruminants in Tanzania. Tropical Animal Health and Production, 41, 959-967.
- SWAI, E. S., MTUI, P. F., CHANG'A, A. K., & MACHANGE, G. E. 2008. The prevalence of serum antibodies to Ehrlichia ruminantium infection in ranch cattle in Tanzania: a cross-sectional study. Journal of the South African Veterinary Association, 79, 71-75.
- SYKES, K. F. & JOHNSTON, S. A. 1999. Genetic live vaccines mimic the antigenicity but not pathogenicity of live viruses. DNA and Cell Biology, 18, 521-531.
- SYNGE, B. A. 1978. Brain biopsy for the diagnosis of heartwater. Tropical Animal Health and Production, 10, 45-48.
- TERBLANCHE, H. J. J. 1968. Organophosphate poisoning in farm stock in southern Africa. Journal of the South African Veterinary Medical Association, 39, 69-72.
- TERBLANCHE, H. J. J. & MINNE, J. A. 1968. Thiodan poisoning of cattle. A case report. Journal of the South African Veterinary Medical Association, 39, 91-92.
- TESHALE, S., KUMSA, B., MENANDRO, M. L., CASSINI, R., & MARTINI, M. 2016. Anaplasma, Ehrlichia and rickettsial pathogens in ixodid ticks infesting cattle and sheep in western Oromia, Ethiopia. Experimental and Applied Acarology, 70, 231-237.
- THEILER, A. 1905. The advance of our knowledge respecting the stock diseases of South Africa. Transvaal Agricultural Journal, 4, 69-80.
- THEMA, N., PRETORIUS, A., TSHILWANE, S. I., LIEBENBERG, J., STEYN, H., & VAN KLEEF, M. 2016. Cellular immune responses induced in vitro by Ehrlichia ruminantium secreted proteins and identification of vaccine candidate peptides. Onderstepoort Journal of Veterinary Research, 83, e1-e11.
- THIRUMALAPURA, N. R., CROCQUET-VALDES, P. A., SAITO, T. B., THOMAS, S., MCBRIDE, J. W., & WALKER, D. H. 2013. Recombinant Ehrlichia P29 protein induces a protective immune response in a mouse model of ehrlichiosis. Vaccine, 31, 5960-5967.
- TICE, G. A., BRYSON, N. R., STEWART, C. G., DU PLESSIS, B., & DE WAAL, D. T. 1998. The absence of clinical disease in cattle in communal grazing areas where farmers are changing from an intensive dipping programme to one of endemic stability to tick-borne diseases. Onderstepoort Journal of Veterinary Research, 65, 169-175.
- TOTTÉ, P., BENSAID, A., MAHAN, S. M., MARTINEZ, D., & MCKEEVER, D. J. 1999. Immune responses to Cowdria ruminantium infections. Parasitology Today, 15, 286-290.
- TOTTÉ, P., BLANKAERT, D., ZILIMWABAGABO, P., & WERENNE, J. 1993. Inhibition of Cowdria ruminantium infectious yield by interferons alpha and gamma in endothelial cells. Revue d’Élevage et de Médecine Vétérinaire des Pays Tropicaux, 46, 189-194.
- TOTTÉ, P., JONGEJAN, F., DE GEE, A. L., & WERENNE, J. 1994. Production of alpha interferon in Cowdria ruminantium-infected cattle and its effect on infected endothelial cell cultures. Infection and Immunity, 62, 2600-2604.
- TOTTÉ, P., MCKEEVER, D., MARTINEZ, D., & BENSAID, A. 1997. Analysis of T-cell responses in cattle immunized against heartwater by vaccination with killed elementary bodies of Cowdria ruminantium. Infection and Immunity, 65, 236-241.
- TOTTÉ, P., VACHIERY, N., MARTINEZ, D., TRAP, I., BALLINGALL, K. T., MACHUGH, N. D., BENSAID, A., & WERENNE, J. 1996. Recombinant bovine interferon gamma inhibits the growth of Cowdria ruminantium but fails to induce major histocompatibility complex class II following infection of endothelial cells. Veterinary Immunology and Immunopathology, 53, 61-71.
- TSHIKHUDO, N., PRETORIUS, A., PUTTERILL, J., & VAN KLEEF, M. 2010. Preparation and in vitro characterisation of Ehrlichia ruminantium plasmid DNA and proteins encapsulated into and DNA adsorbed onto biodegradable microparticles. Ticks and Tick Borne Diseases, 1, 186-193.
- UILENBERG, G. 1971. Studies on cowdriosis in Madagascar. I. Revue d’Élevage et de Médecine Vétérinaire des Pays Tropicaux, 24, 239-249.
- UILENBERG, G. 1982. Disease problems associated with the introduction of European cattle in the tropics. In Proceedings of the 12th World Congress on Diseases of Cattle, The Netherlands. 1025-1032
- UILENBERG, G. 1982. Experimental transmission of Cowdria ruminantium by the Gulf coast tick Amblyomma maculatum: danger of introducing heartwater and benign African theileriasis onto the American mainland. American Journal of Veterinary Research, 43, 1279-1282.
- UILENBERG, G. 1983. Heartwater (Cowdria ruminantium infection): current status. Advances in Veterinary Science and Comparative Medicine, 27, 427-480.
- UILENBERG, G. 1990. Methods currently used for the control of heartwater: their validity and proposals for future control strategies. Parassitologia, 32, 55-62.
- UILENBERG, G, BARRÉ, N, CAMUS, E, BURRIDGE, M J, & GARRIS, G I. 1984. Heartwater in the Caribbean. Preventive Veterinary Medicine, 2, 255-267.
- UILENBERG, G., CAMUS, E., & BARRÉ, N. 1985. Quelques observations sur une souche de Cowdria ruminantium isolée en Guadeloupe (Antilles Françaises). Revue d’Élevage et de Médecine Vétérinaire des Pays Tropicaux, 38, 34-42.
- UNVER, A., PEREZ, M., ORELLANA, N., HUANG, H., & RIKIHISA, Y. 2001. Molecular and Antigenic Comparison of Ehrlichia canis Isolates from Dogs, Ticks, and a Human in Venezuela. Journal of Clinical Microbiology, 39, 2788-93.
- VACHIÉRY, N., JEFFERY, H., PEGRAM, R., APRELON, R., PINARELLO, V., KANDASSAMY, R. L., RALINIAINA, M., MOLIA, S., SAVAGE, H., ALEXANDER, R., FREBLING, M., MARTINEZ, D., & LEFRANCOIS, T. 2008. Amblyomma variegatum ticks and heartwater on three Caribbean Islands. Annals of the New York Academy of Sciences, 1149, 191-195.
- VACHIÉRY, N., LEFRANCOIS, T., ESTEVES, I., MOLIA, S., SHEIKBOUDOU, C., KANDASSAMY, Y., & MARTINEZ, D. 2006. Optimisation of the inactivated vaccine dose against heartwater and in vitro quantification of Ehrlichia ruminantium challenge material. Vaccine, 24, 4747-4756.
- VACHIÉRY, N., MAGANGA, G., LEFRANCOIS, T., KANDASSAMY, Y., STACHURSKI, F., ADAKAL, H., FERRAZ, C., MORGAT, A., BENSAID, A., COISSAC, E., BOYER, F., DEMAILLE, J., VIARI, A., MARTINEZ, D., & FRUTOS, R. 2008. Differential strain-specific diagnosis of the heartwater agent: Ehrlichia ruminantium. Infection, Genetics and Evolution, 8, 459-466.
- VACHIÉRY, N., TRAP, I., TOTTE, P., MARTINEZ, D., & BENSAID, A. 1998. Inhibition of MHC class I and class II cell surface expression on bovine endothelial cells upon infection with Cowdria ruminantium. Veterinary Immunology and Immunopathology, 61, 37-48.
- VAN AMSTEL, S. R. 1987. The use of electro-encephalography and brain biopsy in the clinical diagnosis of heartwater. Onderstepoort Journal of Veterinary Research, 54, 295-299.
- VAN AMSTEL, S. R. & OBEREM, P. T. 1987. The treatment of heartwater. Onderstepoort Journal of Veterinary Research, 54, 475-479.
- VAN AMSTEL, S. R., OBEREM, P. T., DIDOMENICO, M., KIRKPATRICK, R. D., & MATHEE, J. 1988. The presence of endotoxin activity in cases of experimentally-induced heartwater in sheep. Onderstepoort Journal of Veterinary Research, 55, 217-220.
- VAN AMSTEL, S. R., REYERS, F., GUTHRIE, A. J., OBEREM, P. T., & BERTSCHINGER, H. 1988. The clinical pathology of heartwater. I. Haematology and blood chemistry. Onderstepoort Journal of Veterinary Research, 55, 37-45.
- VAN AMSTEL, S. R., REYERS, F., & MYBURGH, E. 1994. Changes in the blood-gas status of sheep with experimentally induced heartwater. Onderstepoort Journal of Veterinary Research, 61, 45-49.
- VAN AMSTEL, S. R., REYERS, F., MYBURGH, E., PRETORIUS, G., & SACKS, P. 1994. The clinical pathology of heartwater. III. Changes in blood clotting, blood calcium, blood protein, haematocrit and white-cell counts in sheep with experimentally induced heartwater. Onderstepoort Journal of Veterinary Research, 61, 21-27.
- VAN DE PYPEKAMP, H. E. & PROZESKY, L. 1987. Heartwater. An overview of the clinical signs, susceptibility and differential diagnoses of the disease in domestic ruminants. Onderstepoort Journal of Veterinary Research, 54, 263-266.
- VAN DER MERWE, L. 1979. Field experience with heartwater (Cowdria ruminantium) in cattle. Journal of the South African Veterinary Association, 50, 323-325.
- VAN DER MERWE, L. 1987. The infection and treatment method of vaccination against heartwater. Onderstepoort Journal of Veterinary Research, 54, 489-491.
- VAN HEERDEN, H., COLLINS, N. E., BRAYTON, K. A., RADEMEYER, C., & ALLSOPP, B. A. 2004. Characterization of a major outer membrane protein multigene family in Ehrlichia ruminantium. Gene, 330, 159-168.
- VAN HEERDEN, H., STEYN, H. C., ALLSOPP, M. T., ZWEYGARTH, E., JOSEMANS, A. I., & ALLSOPP, B. A. 2004. Characterization of the pCS20 region of different Ehrlichia ruminantium isolates. Veterinary Microbiology, 101, 279-291.
- VAN KLEEF, M., GUNTER, N. J., MACMILLAN, H., ALLSOPP, B. A., SHKAP, V., & BROWN, W. C. 2000. Identification of Cowdria ruminantium antigens that stimulate proliferation of lymphocytes from cattle immunized by infection and treatment or with inactivated organisms. Infection and Immunity, 68, 603-614.
- VAN VLIET, A. H., JONGEJAN, F., VAN KLEEF, M., & VAN DER ZEIJST, B. A. 1994. Molecular cloning, sequence analysis, and expression of the gene encoding the immunodominant 32-kilodalton protein of Cowdria ruminantium. Infection and Immunity, 62, 1451-1456.
- VAN VLIET, A. H., VAN DER ZEIJST, B. A., CAMUS, E., MAHAN, S. M., MARTINEZ, D., & JONGEJAN, F. 1995. Use of a specific immunogenic region on the Cowdria ruminantium MAP1 protein in a serological assay. Journal of Clinical Microbiology, 33, 2405-2410.
- WAGHELA, S. D., RURANGIRWA, F. R., MAHAN, S. M., YUNKER, C. E., CRAWFORD, T. B., BARBET, A. F., BURRIDGE, M. J., & MCGUIRE, T. C. 1991. A cloned DNA probe identifies Cowdria ruminantium in Amblyomma variegatum ticks. Journal of Clinical Microbiology, 29, 2571-2577.
- WALKER, D. H. & DUMLER, J. S. 1996. Emergence of the ehrlichioses as human health problems. Emerging Infectious Diseases, 2, 18-29.
- WALKER, J. B. & OLWAGE, A. 1987. The tick vectors of Cowdria ruminantium (Ixodoidea, Ixodidae, genus Amblyomma) and their distribution. Onderstepoort Journal of Veterinary Research, 54, 353-379.
- WEAVER, C. T. & UNANUE, E. R. 1990. The co-stimulatory function of antigen-presenting cells. Immunology Today, 11, 49-55.
- WEISS, K. E., HAIG, D. A., & ALEXANDER, R. A. 1952. Aureomycin in the treatment of heartwater. Onderstepoort Journal of Veterinary Research, 25, 41-50.
- WESONGA, F. D., MUKOLWE, S. W., & GROOTENHUIS, J. 2001. Transmission of Cowdria ruminantium by Amblyomma gemma from infected African buffalo (Syncerus caffer) and eland (Taurotragus oryx) to sheep. Tropical Animal Health and Production, 33, 379-390.
- WIKEL, S. K. & ALARCON-CHAIDEZ, F. J. 2001. Progress toward molecular characterization of ectoparasite modulation of host immunity. Veterinary Parasitology, 101, 275-287.
- WILSON, D. D. & RICHARD, R. D. 1984. Interception of a vector of heartwater, Amblyomma hebraeum Koch (Acari: Ixodidae) on black rhinoceroses imported into the United States. In Proceedings of the Eighty eigth Annual Meeting of the United States Animal Health Association, The Hyatt Regency Fort Worth Hotel, Fort Worth, Texas. 303-311.
- YOUNG, E. & BASSON, P. A. 1973. Heartwater in the eland. Journal of the South African Veterinary Association, 44, 185-186.
- YU, X., MCBRIDE, J. W., ZHANG, X., & WALKER, D. H. 2000. Characterization of the complete transcriptionally active Ehrlichia chaffeensis 28 kDa outer membrane protein multigene family. Gene, 248, 59-68.
- YUNKER, C. E. 1996. Heartwater in sheep and goats: a review. Onderstepoort Journal of Veterinary Research, 63, 159-170.
- YUNKER, C. E., BYROM, B., & SEMU, S. 1988. Cultivation of Cowdria ruminantium in bovine vascular endothelial cells. The Kenya Veterinarian, 12, 12-16.
- YUNKER, C. E., KOCAN, K. M., NORVAL, R. A., & BURRIDGE, M. J. 1987. Distinctive staining of colonies of Cowdria ruminantium in midguts of Amblyomma hebraeum. Onderstepoort Journal of Veterinary Research, 54, 183-185.
- ZHANG, C., XIONG, Q., KIKUCHI, T., & RIKIHISA, Y. 2008. Identification of 19 polymorphic major outer membrane protein genes and their immunogenic peptides in Ehrlichia ewingii for use in a serodiagnostic assay. Clinical and Vaccine Immunology, 15, 402-411.
- ZHANG, J., KELLY, P., GUO, W., XU, C., WEI, L., JONGEJAN, F., LOFTIS, A., & WANG, C. 2015. Development of a generic Ehrlichia FRET-qPCR and investigation of ehrlichioses in domestic ruminants on five Caribbean islands. Parasites and Vectors, 8, 506.
- ZWEYGARTH, E. & JOSEMANS, A. I. 2001. A chemically defined medium for the growth of Cowdria ruminantium. Onderstepoort Journal of Veterinary Research, 68, 37-40.
- ZWEYGARTH, E. & JOSEMANS, A. I. 2001. Continuous in vitro propagation of Cowdria ruminantium (Welgevonden stock) in a canine macrophage-monocyte cell line. Onderstepoort Journal of Veterinary Research, 68, 155-157.
- ZWEYGARTH, E., JOSEMANS, A. I., & HORN, E. 1998. Serum-free media for the in vitro cultivation of Cowdria ruminantium. Annals of the New York Academy of Sciences, 849, 307-312.
- ZWEYGARTH, E., JOSEMANS, A. I., & STEYN, H. C. 2008. In vitro isolation of Ehrlichia ruminantium in ovine blood into Ixodes scapularis (IDE8) cell cultures. Onderstepoort Journal of Veterinary Research, 75, 121-126.
- ZWEYGARTH, E., JOSEMANS, A. I., & STEYN, H. C. 2008. Experimental use of the attenuated Ehrlichia ruminantium (Welgevonden) vaccine in Merino sheep and Angora goats. Vaccine, 26, G34-G39.
- ZWEYGARTH, E., JOSEMANS, A. I., & TROSKIE, P. C. 2009. Experimental use of the attenuated Ehrlichia ruminantium (Welgevonden) vaccine in Friesian cattle: A pilot study. In Abstracts of 10th Biennial Conference, Lübeck, Germany, June 28-July 3, 2009, 73
- ZWEYGARTH, E., JOSEMANS, A. I., VAN STRIJP, M. F., LOPEZ-REBOLLAR, L., VAN KLEEF, M., & ALLSOPP, B. A. 2005. An attenuated Ehrlichia ruminantium (Welgevonden stock) vaccine protects small ruminants against virulent heartwater challenge. Vaccine, 23, 1695-1702.
- ZWEYGARTH, E., JOSEMANS, A. I., VAN STRIJP, M. F., VAN HEERDEN, H., ALLSOPP, M. T., & ALLSOPP, B. A. 2002. The Kümm isolate of Ehrlichia ruminantium: in vitro isolation, propagation and characterization. Onderstepoort Journal of Veterinary Research, 69, 147-153.
- ZWEYGARTH, E., VOGEL, S. W., JOSEMANS, A. I., & HORN, E. 1997. In vitro isolation and cultivation of Cowdria ruminantium under serum-free culture conditions. Research in Veterinary Science, 63, 161-164.