- Infectious Diseases of Livestock
- Part 1
- Vectors: Tabanidae
- Vectors: Ticks
- Vectors: Tsetse flies
- Vectors: Muscidae
- Vectors: Tabanidae
- Vectors: Culicoides spp.
- Vectors: Mosquitoes
- Classification, epidemiology and control of arthropod-borne viruses
- Special factors affecting the control of livestock diseases in sub-Saharan Africa
- The control of infectious diseases of livestock: Making appropriate decisions in different epidemiological and socioeconomic conditions
- Infectious diseases of animals in sub-Saharan Africa: The wildlife⁄livestock interface
- Vaccination: An approach to the control of infectious diseases
- African animal trypanosomoses
- Dourine
- Trichomonosis
- Amoebic infections
- GENERAL INTRODUCTION: COCCIDIA
- Coccidiosis
- Cryptosporidiosis
- Toxoplasmosis
- Besnoitiosis
- Sarcocystosis
- Balantidiosis
- Leishmaniosis
- Neosporosis
- Equine protozoal myeloencephalitis
- GENERAL INTRODUCTION: BABESIOSES
- Bovine babesiosis
- Equine piroplasmosis
- Porcine babesiosis
- Ovine babesiosis
- GENERAL INTRODUCTION: THEILERIOSES OF CATTLE
- East Coast fever
- Corridor disease
- Zimbabwe theileriosis
- Turning sickness
- Theileria taurotragi infection
- Theileria mutans infection
- Theileria annulata theileriosis
- Theileriosis of sheep and goats
- Theileria buffeli⁄orientalis infection
- Non-pathogenic Theileria species in cattle
- GENERAL INTRODUCTION: RICKETTSIAL, CHLAMYDIAL AND HAEMOTROPIC MYCOPLASMAL DISEASES
- Heartwater
- Lesser known rickettsial infections in animals and humans
- Chlamydiosis
- Q fever
- Eperythrozoonosis
- Bovine Haemobartonellosis
- Potomac horse fever
- GENERAL INTRODUCTION: ANAPLASMOSES
- Bovine anaplasmosis
- Ovine and caprine anaplasmosis
Vectors: Tabanidae
This content is distributed under the following licence: Attribution-NonCommercial CC BY-NC  View Creative Commons Licence details here
View Creative Commons Licence details here
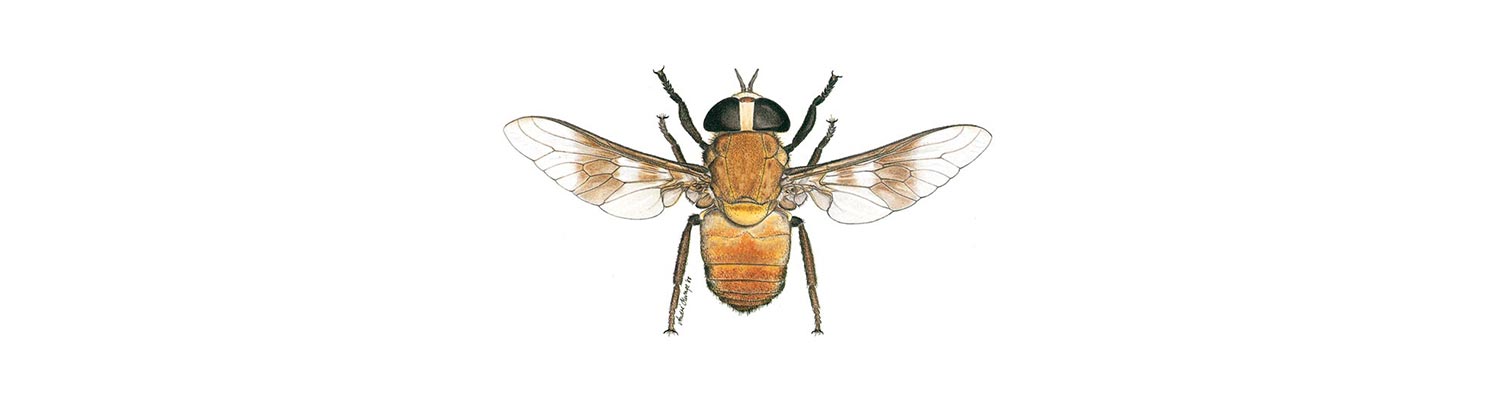
Vectors: Tabanidae
E M NEVILL, R J PHELPS AND B R STUCKENBERG
Introduction
The reason for the inclusion of a chapter on flies of the family Tabanidae in this book is their great potential as mechanical transmitters of infectious disease agents. Tabanidae are commonly known as horse flies, deer flies, clegs, and, in South Africa, as blindevlieë (Afrik.) (blind flies). The large size and variety of genera and species of Tabanidae in southern Africa should have made them an obvious choice for study by biologists, but studies on them have been mostly restricted to their taxonomy while those on their biology, role in disease transmission and control are limited.
Biosystematics
The taxonomy of the Tabanidae of the Afrotropical Region embraces three subfamilies (Pangoniinae, Chrysopsinae and Tabaninae) containing 31 genera and 727 species.9, 10, 37–39 Four hundred and ten of these species occur in southern Africa, and of them about 80 per cent belong to only five genera, namely Haematopota, Tabanus, Philoliche, Rhigioglossa (= Mesomyia), and Chrysops.
Flies belonging to the Pangoniinae are usually of little known economic importance.30 Various species of Philoliche of the subfamily Pangoniinae are, however, persistent pursuers of blood meals from cattle and horses in localized situations.46 Of the Tabanidae recorded from southern Africa, three genera of the Chrysopsinae, namely Rhigioglossa (39 species), Chrysops (23 species) and Tabanocella (10 species); and five genera of the subfamily Tabaninae, namely Haematopota (137 species), Tabanus (68 species), Hybomitra (nine species), Atylotus (six species) and Ancala (four species) appear in a list of tabanid-transmitted disease agents.32
The taxonomy of tabanids of Angola,14, 16 Mozambique, 15 Zimbabwe24 and South Africa48 has been reviewed.
In general, the biology of the Tabanidae has been poorly studied throughout the world, but this is even more the case in southern Africa. Chainey and Oldroyd10 summarized the larval biology of these flies very succinctly as follows:
Tabanid larvae live in wet places but need to reach the atmosphere periodically as they breathe air through a posterior siphon. The usual habitat is the wet mud at the margins of ponds, lakes and streams, but those of some species live in sand on the seashore. Some Tabanus larvae inhabit pockets of damp soil in generally dry areas, and Haematopota larvae occur mainly in areas of wet soil. Tree-hole breeding occurs in a few Tabanidae; larvae of Thaumastocera for example living in the accumulated debris in rot-holes of palms and forest trees . . . larvae of Haematopota and Tabanus feed on other insects, crustacea, worms or their own kind, but many tabanid larvae, such as those of Chrysops, feed on vegetable debris.
Adult tabanid flies are generally large, usually with well-developed eyes. Their mouthparts are adapted for both sucking and lapping, with both sexes feeding on nectar. The females of most species also feed on blood, which is needed for the development of the ovaries. The mandibles and maxilla are serrated and are used to cut into the skin and lacerate the dermal tissues, and thus induce the formation of a pool of blood which is then lapped up. A small amount of blood is trapped within the component parts of the mouthparts when they are withdrawn. Mechanical transmission of disease organisms is thus facilitated by this action. The coarse structure of the mouthparts and the mode of feeding results in painful bites causing cattle to try to dislodge or kill the flies by striking them with their tongues.5, 30 Further details on the life cycles and bionomics of Tabanidae can be obtained from a chapter in the book by Kettle.30
The density of tabanid populations is closely associated with breeding sites. Populations around dams, vleis and natural wetlands may be high and composed of only a few species or perhaps even a single dominant one, which in South Africa is often a species of Haematopota.
In southern Africa, the breeding sites of many tabanids in Malawi33–35 and the larval feeding habits of Tabanus biguttatus Wied. and Amanella emergens Oldroyd in South Africa8 have been recorded, but published information on the biology of adults of species of the Afrotropical Region is rather sparse and deals mainly with flight seasons, abundance and nocturnal activity.10, 48 It is considered that certain physical adaptations aimed at distracting the attentions of visually orientated biting flies have evolved in some African wildlife species.45 It has been demonstrated in the north-eastern KwaZulu-Natal Province of South Africa that the striping of zebra makes them virtually undetectable to tsetse flies (Glossina spp.) and tabanids.25Field observations also indicate that antelope species with a countershaded haircoat pattern, characterized particularly by a pale belly that eliminates the rounded appearance of the abdomen which is enhanced by shadowing in sunlight, are evidently unattractive to tabanids.45 In contrast, mammals with uniform colouring, such as elephant, rhinoceros and hippopotamus, which present a strongly rounded appearance in sunlight, are highly attractive to large tabanid species such as Tabanus taeniola P. de B. and Philoliche aethiopica Thunberg. Experimental evidence has shown that a dark, single colour, horizontal cylinder is a most attractive shape for tabanids, and indeed the Harris Fly Trap represents essentially such a form.26.
Tabanidae are unequally distributed in South Africa.48 In general, many species of the rich savannah and tropical woodland tabanid fauna of Africa occur in the eastern coastal lowlands as far south as the Eastern Cape Province, and also range to a much more limited extent into the subarid Limpopo Valley and onto the Highveld. In the southern and south-western parts of the Western Cape Province a taxonomically very distinct tabanid fauna is characteristic of the macchia (fynbos) and the fold mountains, as well as the adjacent Karroid areas. These two major faunal units have few species in common.48
Regions with a high rainfall have the highest number of species. Topographically diversified areas also have a more diverse tabanid fauna. Consequently the plateau slopes, the Mpumalanga–Limpopo Lowveld, the coastal plain of Kwa- Zulu-Natal, the Eastern Cape Province, and the southern and south-western part of the Western Cape Province have a much richer tabanid fauna than do the interior plateau areas and the arid western and northern regions of South Africa. 48
A large proportion of species occurring in the northern and eastern lowlands of South Africa is shared with Mozambique, Zimbabwe, Zambia and Angola.
Certain species dominate in particular areas in southern Africa. In South African sand-dune areas along the southern and western coast of the Western Cape Province, a distinctive tabanid fauna is evident, with Rhigioglossa edentula Wied. being particularly evident.46 The grassland plains of the Highveld support a limited fauna, though some species (e.g. Haematopota spp.) may achieve high densities where there are suitable breeding sites.46 Wooded areas, or patches of indigenous forest, favour a greater diversity of tabanid species and may harbour dense populations of these flies. A common assemblage of flies may consist, for example, of species of the genera Philoliche, Haematopota, Tabanocella and perhaps Amanella, and the component species will vary from locality to locality. Members of such a guild of species aggregate around hosts that are potential sources of blood meals; such hosts may range in size from small antelope to cattle, and the flies will follow them with a marked degree of persistence. Movement of the host outside the forest or woodland results in selective withdrawal of those tabanid species that are unwilling to remain in open areas in full sunlight.
Many tabanids have preferred feeding sites on animals; the smaller tabanid species tend to settle on the legs, whereas the large species usually seek blood meals on the sides of the body or on the flanks. This behaviour has been interpreted as a form of resource partitioning through which interspecific competition for feeding sites may be reduced. This aspect has not been studied extensively in Africa, apart from observations made in Uganda29 and Zimbabwe41
Most tabanid species are active as adult flies only during the rainy season. Mass emergence of the flies may occur with the advent of the first rains, or in meteorological conditions in which such rain is possible. Consequently, in the winter-rainfall area of South Africa tabanids occur commonly from late winter to early summer, whereas in summer- rainfall regions their occurrence may be highly correlated with the rainy season of November to March. Some species may persist in low numbers throughout the year in lowland areas without frost, and a few fly only in the dry season. In hot climates, some species can be seen drinking at water sources such as the edges of natural pools; it has been observed in the Kruger National Park in South Africa that cemented water troughs provided for game are constantly visited by species of Tabanidae in drought years when natural water sources are scarce. Rearing records show that males and females occur in equal numbers, but the males may seldom be seen as they do not take blood meals and so are not attracted to mammals and only rarely to traps.11, 43, 46
Large tabanids mainly attack large mammals (cattle, horses, large antelope, elephant) and their presence may affect or modify behavioural patterns of animals. The blood-sucking activity of three common large species of Tabanidae (Tabanus biguttatus, T. taeniola and Ancala africana Gray) (Figure 4.1) have an important influence on the summer diurnal activity of hippopotami in north-eastern KwaZulu- Natal.47 Because of the presence of large numbers of these flies in conjunction with high solar radiation, hippopotami are obliged to remain more or less submerged in the water during daylight hours.
Disease transmission
The infectious diseases or agents with which horseflies have been implicated as playing a role in their transmission are summarized in (Table 4.1). The method of transmission maybe either biological, which is uncommon, or mechanical.
Only two disease-producing micro-organisms are known to be transmitted biologically by tabanid flies (Table 4.1). Salivary gland infections of Haemoproteus metchnikovi are known to occur in the deerfly (Chrysops callidus O.S.) after feeding on turtles.17 The other is Trypanosoma theileri which causes a benign infection in cattle. The results of carefully conducted trials in Germany suggest that transmission in the field takes place by contamination of the oral mucosa of cattle with metacyclid stages from gut contents or faeces of tabanids, when the cattle defend themselves and kill feeding tabanids with their tongues.5
Of interest is that although both salivarian and stercorarian trypanosomes undergo development in certain insects, infection rates of salivarian trypanosomes in the vector are often low, whereas stercorarian trypanosome infection rates may approach 100 per cent.30
In addition to infectious agents, various filarial worms develop in, and are transmitted by, species of tabanids throughout the world (Table 4.1).
Tabanids are the insect group most frequently associated with the mechanical transmission of pathogens—a result of a fly feeding successively on an infective host and then on a susceptible animal.17–22, 28, 53–56 Transmission is facilitated by the fact that not only are they generally large insects and their presence worrying to animals, but also that their bites are painful.
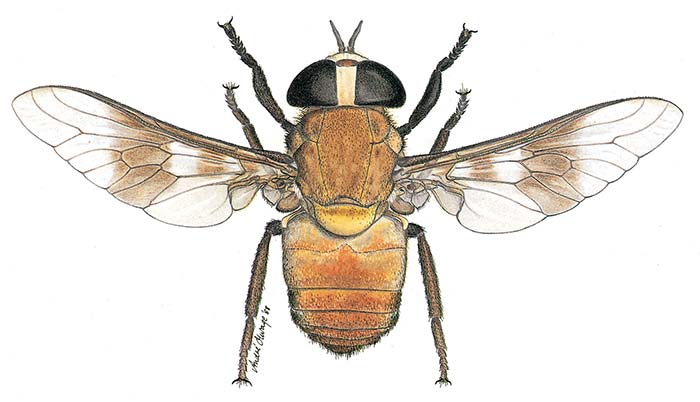
Figure 4.1 Ancala africana
Feeding is therefore often interrupted and dispersal to alternate hosts frequent. The factors which influence mechanical transmission are:17
- the survival time of the pathogen outside the host;
- the proximity of infected and susceptible hosts;
- vector mobility;
- persistence of feeding (the less persistent feeders are probably most important in mechanical transmission);
- vector size (the larger tabanids would appear to be better transmitters of pathogens);
- the total tabanid population (this appears to be the most consistent phenomenon associated with peaks of infection);
- painful bites (this can be directly correlated with disease transmission);
- the host response to the bite of tabanids may be diminished during the acute stage of some infectious diseases and therefore the early febrile periods of chronic and acute infections may be the critical periods for transmission as pathogen titres in the blood of the host are often highest then;
- the quantity of infectious material transported by individual vectors between hosts;
- the amount of infectious material deposited at a wound/ portal of entry;
- the titre of the pathogen in the blood of the infected host;
- seasonal changes and the persistence of infectious agents in the circulation of the mammalian host (e.g. under constant vector pressure an animal with a 1 per cent parasitaemia of Anaplasma marginale for 50 days may be equivalent, as a source of infection, to an animal with a parasitaemia of 50 per cent for one day);
- the density and age of the host; and
- prevalence of infection and degree of immunity in a host population.
Tabanids possibly played a role in the mechanical transmission of nagana (trypanosomosis) during the major epidemics in north-eastern KwaZulu-Natal.3, 56 Sporadic outbreaks of the disease still occur in this part of KwaZulu-Natal,6 and biting flies such as tabanids and horn flies (Haematobia spp.) may well play a role in its transmission.36
In southern Africa the only recorded experimental work on the mechanical transmission by tabanids of a pathogen is that done on, Besnoitia besnoiti.4 The flies that successfully transmitted the parasite by bite from cattle to rabbit and from cattle to cattle were Atylotus nigromaculatus Ricardo, Tabanocella denticornis Wied. and Haematopota albihirta Karsch, the only tabanid species which could be captured in sufficient numbers at the Onderstepoort Veterinary Institute. In three instances only a single fly was necessary to transmit B. besnoiti. Flies remained infective for up to 24 hours.4
Certain large species, notably Tabanus taeniola and Philoliche aethiopica, are uninhibited by large expanses of open terrain and are capable of sustained, powerful flight. Their often noisy flight, persistent attendance in large numbers and painful bites may induce restlessness in domestic stock.46
In southern Africa no work has been done on the evaluation of economic losses due to tabanid bites. This would be difficult as the appearance of a large number of these flies is often localized, sporadic and can occur simultaneously with other fly genera. In 1965 the bites of horse flies led to cattle losses in the USA amounting to 30 million dollars from reduced weight gain and 10 million dollars from reduced milk production.44 Cows treated with synergized pyrethrins showed a significant increase in butter fat production, and similarly treated beef cattle gained 9 to 14 kg more than untreated cattle, over a 38-day period.7
Control
Methods of control generally encompass either the use of baited or unbaited traps or insecticide-treated animals. However, no concerted effort is made to apply these methods in southern Africa and there is also no published work on the control of tabanids in the subcontinent.
Studies in Zimbabwe on the reactions of tabanids to mobile and stationary baits, with and without human and ox odour, showed that, although odour did entice more flies to stationary baits, the main attraction was visual.51 It has been shown in Zimbabwe that 1-octen-3-ol and 4-methylphenol, which are known attractants for some Glossina spp.49, 50 are strongly attractive to Tabanus pullulus Austen and T. copemani Austen, and possibly attractive to several other species. 40 The indications are that it may be possible to develop odour-baited traps to control some tabanids.
Control of adult tabanids in other parts of the world is not commonly practised. Various approaches have, however, been tested. Because of their visual attraction to baits, use has been made of Manning or box traps, Manitoba or canopy traps, and sticky traps,1, 42, 52 with or without the addition of carbon dioxide. In Georgia, USA, the effectiveness of the canopy trap in capturing female Tabanidae increased significantly when 1-octen-3-ol was used as an attractant.23 The success of these traps often depends on their being placed in unobstructed flyways such as footpaths or the edge of a forest.42
Another approach to tabanid control is the use of residual insecticides on animals. Permethrin is lethal to horse flies and persists on treated animals for about two weeks.2, 27 It has been found that 10 ppm permethrin is needed to kill tabanids compared with only 2 ppm for stable flies.27
Use of trapping or insecticides for the control of tabanids may be warranted under fairly intensive farming situations but, under extensive conditions, their control may not be successful due to large and mobile populations of one or more host species. In Oklahoma, USA, it has been shown that Tabanus abactor Philip is capable of flying at least 2,4 km and that the estimated number of host-seeking females per hectare can exceed 2 000 flies per day.12, 13 However, a noticeable decrease in horse fly numbers, mostly Tabanus nigrovittatus Macq., resulted when 300 to 730 box traps were placed around the perimeters of Cape Cod salt marshes in the USA.52
Since both male and female tabanids visit flowers for nectar, they will pick up pollen and so may be pollinators for some plants. A potential side-effect of the widespread use of odour-baited, insecticide-treated targets and traps to control tsetse flies might be to reduce tabanid populations, as it has been shown that at least some species of tabanids are strongly attracted to such targets and traps.40 This topic was investigated at Rekomitjie Research Station in Zimbabwe.31 By comparing pollen taken from tabanids with that from plants available at the same time, it was demonstrated that, at this locality, tabanids were not specializing on any particular plant species. In addition, direct observation revealed that plants visited by tabanids attracted many other insect species not known to be attracted to anti-tsetse devices. At Rekomitjie, any reduction in tabanid numbers would not have an impact on pollination.
Table 4.1 Disease agents associated with tabanids17, 32
| AGENT | ASSOCIATION |
|---|---|
| VIRUSES BACTERIA AND RICKETTSIAS PROTOZOA HELMINTHS |
|
B = biological transmission
D = development of organism observed in tabanid
EE = epidemiological evidence
ET = experimental transmission in unnatural manner
NT = natural mechanical transmission
(exp.) = in experimentally infected flies
I = isolation from wild tabanids
References
- bauer, b. & wetzel, h., 1974. Versuche mit Fliegenfallen zur Tabanidenbekämpfung auf der Weide. Tierärztliche Umschau, 29, 206–210.
- bay, d.e., ronald, n.c. & harris, r.l., 1976. Evaluation of a synthetic pyrethroid for tabanid control on horses and cattle. The Southwestern Entomologist, 1, 198–203.
- bedford, g.a.h., 1927. Report on the transmission of Nagana in the Ntabanana and Mhlatuze Settlements, Zululand. Part I. Eleventh & Twelfth Reports of the Director of Veterinary Education and Research, 275–300.
- bigalke, r.d., 1968. New concepts on the epidemiological features of bovine besnoitiosis as determined by laboratory and field investigations. Onderstepoort Journal of Veterinary Research, 35, 3–138.
- böse, r., friedhoff, k.t., olbrich, s., bu¨scher, g. & domeyer, i., 1987. Transmission of Trypanosoma theileri to cattle by Tabanidae. Parasitology Research, 73, 421–424.
- bosman, p.p., 1990. Trypanosomiasis (T. congolense) in South Africa. Office de L’International des Epizooties: Disease Information, 3, 79–80.
- bruce, w.n. & decker, g.c., 1951. Tabanid control on dairy and beef cattle with synergized pyrethrins. Journal of Economic Entomology, 44, 154–159.
- callan, e.mcc., 1980. Larval feeding habits of Tabanus biguttatus and Amanella emergens in South Africa (Diptera: Tabanidae). Revue de Zoologie Africaine, 94, 791–794.
- chainey, j.e., 1987. Afrotropical Tabanidae (Diptera): The genus Rhigioglossa Wiedemann 1828 (including Mesomyia Macquart, 1850, as a subgenus). Annals of the Natal Museum, 28, 137–159.
- chainey, j.e. & oldroyd, h., 1980. Family Tabanidae. In: crosskey, r.w., (ed.). Catalogue of the Diptera of the Afrotropical Region. London: British Museum (Natural History).
- clarke, j.e., 1968. Seasonal abundance of Tabanidae at Mazabuka, Zambia. Proceedings of the Royal Entomological Society, London [A], 43, 108–121.
- cooksey, l.m. & wright, r.e., 1987. Flight range and dispersal activity of the host-seeking horse fly, Tabanus abactor (Diptera: Tabanidae), in northcentral Oklahoma. Environmental Entomology, 16, 211–217.
- cooksey, l.m. & wright, r.e., 1989. Population estimation of the horse fly, Tabanus abactor (Diptera: Tabanidae), in northcentral Oklahoma. Journal of Medical Entomology, 26, 167–172.
- dias, j.a. travassos santos, 1960. Nova contribuicao ao estudo dos tabanídeos (Diptera: Tabanidae) de Angola. Publicacoes culturais Companhia de Diamantes de Angola, 53, 1–125.
- dias, j.a. travassos santos, 1966. Tabanídeos (Diptera-Tabanidae) de Mocambique. Contribuicao pard o seu conhecimento. XVI + 1283PP. Lourenco Marques [= Maputo].
- dias, j.a. travassos santos & serrano, f.m.h., 1967. Alguns novos dados sobre os tabanídeos (Diptera-Tabanidae) de Angola. Revista Estudos gerais Universitarios de Mocambique, 4, 443–530.
- foil, l.d., 1989. Tabanids as vectors of disease agents. Parasitology Today, 5, 88–96.
- foil, l.d., adams, w.v., issel, c.j. & pierce, r., 1984. Tabanid (Diptera) populations associated with an equine infectious anemia outbreak in an inapparently infected herd of horses. Journal of Medical Entomology, 21, 28–30.
- foil, l.d., adams, w.v., mcmanus, j.m. & issel, c.j., 1987. Bloodmeal residues on mouthparts of Tabanus fuscicostatus (Diptera: Tabanidae) and the potential for mechanical transmission of pathogens. Journal of Medical Entomology, 24, 613–616.
- foil, l.d., french, d.d., hoyt, p.g., issel, c.j., leprince, d.j., mcmanus, j.m. & seger, c.l., 1989. Transmission of bovine leukemia virus by Tabanus fuscicostatus. American Journal of Veterinary Research, 50, 1771–1773.
- foil, l.d., meek, c.l., adams, m.s. & issel, c.j., 1983. Mechanical transmission of equine infectious anemia virus by deer flies (Chrysops flavidus) and stable flies (Stomoxys calcitrans). American Journal of Veterinary Research, 44, 155–156.
- foil, l.d., seger, c.l., french, d.d., issel, c.j., mcmanus, j.m., ohrberg, c.j. & ramsey, r.t., 1988. Mechanical transmission of bovine leukemia virus by horse flies (Diptera: Tabanidae). Journal of Medical Entomology, 25, 374–376.
- french, f.e. & kline, d.l., 1989. 1-octen-3-ol, an effective attractant for Tabanidae (Diptera). Journal of Medical Entomology, 26, 459–461.
- goodier, r., 1967. A list of Southern Rhodesian horseflies (Diptera: Tabanidae). Entomologist’s Monthly Magazine, 102, 99–105.
- harris, r.h.t.p., 1930. Report on the bionomics of the tsetse fly (Glossina pallidipes Aust.). Provincial Administration of Natal, Pietermaritzburg.
- harris, r.h.t.p., 1930. Report on the trapping of tsetse flies (with illustrations). Pietermaritzburg: The Natal Witness, Ltd.
- harris, r.l. & oehler, d.d., 1976. Control of tabanids on horses. The Southwestern Entomologist, 1, 194–197.
- issel, c.j. & foil, l.d., 1984. Studies on equine infectious anemia virus transmission by insects. Journal of the American Veterinary Medical Association, 184, 293–297.
- kangwagye, t.n., 1978. Reactions of large mammals to biting flies in Rwenzori National Park, Uganda. Proceedings of the First East African Conference of Entomology and Pest Control, 32–44.
- kettle, d.s., 1984. Medical and Veterinary Entomology. London and Sydney: Croom Helm.
- krämer, a., 1991. Die Bedeutung von Tabaniden als Pollinatoren und ihre potentielle Gefährdung durch Tsetsefliegenbekämfungsaktionen in Simbabwe. Diplomarbeit, Institut fu¨r Biogeographie, Universität Saarlandes, Saarbru¨cken.
- krinsky, w.l., 1976. Animal disease agents transmitted by horse flies and deer flies (Diptera: Tabanidae). Journal of Medical Entomology, 13, 225–275.
- neave, s.a., 1910. Report on a journey to the Luangwa Valley, North-Eastern Rhodesia from July to September, 1910. Bulletin of Entomological Research, 1, 303–317.
- neave, s.a., 1912. Notes on the blood sucking insects of Eastern Tropical Africa. Bulletin of Entomological Research, 3, 275–323.
- neave, s.a., 1915. The Tabanidae of Southern Nyasaland with notes on their life histories. Bulletin of Entomological Research, 5, 287–320.
- nevill, e.m., 2001. Onderdstepoort Veterinary Institute, Onderstepoort, 0110 South Africa. Unpublished observations.
- oldroyd, h., 1952. I. Haematopota and Hippocentrum. The Horse-flies (Diptera: Tabanidae) of the Ethiopian Region. London: British Museum (Natural History).
- oldroyd, h., 1954. II. Tabanus and related genera. The Horse-flies (Diptera: Tabanidae) of the Ethiopian Region. London: British Museum (Natural History).
- oldroyd, h., 1957. III. Subfamilies Chrysopinae, Scepsidinae and Pangoniinae and a revised classification. The Horse-flies (Diptera: Tabanidae) of the Ethiopian Region. London: British Museum (Natural History).
- phelps, r.j. & holloway, m.t.p., 1992. Catches of Tabanidae in response to visual and odour attractants in Zimbabwe. Journal of African Zoology, 106, 371–380.
- phelps, r.j. & holloway, m.t.p., 1990. Alighting sites of female Tabanidae (Diptera) at Rekomitjie, Zimbabwe. Medical and Veterinary Entomology, 4, 349–356.
- roberts, r.h., 1978. Horse flies and deer flies (Family Tabanidae). In: bram, r.a., (ed.). Surveillance and Collection of Arthropods of Veterinary Importance. USDA Agricultural Handbook No. 518.
- robertson, a.g., 1982. Observations on the seasonal incidence and abundance of haematophagus higher Diptera other than tsetse (Diptera: Glossinidae) in the Sabi Valley of Zimbabwe, and comparisons with tabanid catches from elsewhere in Zimbabwe. Transactions of the Zimbabwe Scientific Association, 61, 41–55.
- steelman, c.d., 1976. Effects of external and internal arthropod parasites on domestic livestock production. Annual Review of Entomology, 21, 155–178.
- stuckenberg, b.r., 1981. The striping of zebras: A new look at old data. Antenna, 5, 145–149.
- stuckenberg, r.b., 2001. Natal Museum, Private Bag 9070, Pietermaritzburg, South Africa. Unpublished data.
- tinley, k.l., 1964. Some observations on certain tabanid flies in north eastern Zululand (Diptera: Tabanidae). Proceedings of the Royal Entomological Society of London (A), 39, 73–75.
- usher, p.j., 1972. A review of the South African horsefly fauna (Diptera: Tabanidae). Annals of the Natal Museum, 21, 459–507.
- vale, g.a. & hall, d.r., 1985. The role of 1-octenol-3-ol, acetone and carbon dioxide in the attraction of tsetse flies, Glossina spp. (Diptera: Glossinidae) to ox odour. Bulletin of Entomological Research, 75, 209–217.
- vale, g.a., hall, d.r. & gough, a.j.e., 1988. The olfactory responses of tsetse flies Glossina spp. (Diptera: Glossinidae) to phenols and urine in the field. Bulletin of Entomological Research, 78, 293–300.
- vale, g.a. & phelps, r.j., 1974. Notes on the host-finding behaviour of Tabanidae (Diptera). Arnoldia Rhodesia, 6, 1–6.
- wall, w.j. & doane, o.w., 1980. Large scale use of box traps to study and control saltmarsh greenhead flies (Diptera: Tabanidae) on Cape Cod, Massachusetts. Environmental Entomology, 9, 371–375.
- wiesenhu¨tter, e., 1975. Research into the relative importance of Tabanidae (Diptera) in mechanical disease transmission. II. Investigation of the behaviour and feeding habits of Tabanidae in relation to cattle. Journal of Natural History, 9, 385–392.
- wiesenhu¨tter, e., 1975. Research into the relative importance of Tabanidae (Diptera) in mechanical disease transmission. III. The epidemiology of anaplasmosis in a Dar-es-Salaam dairy farm. Tropical Animal Health and Production, 7, 14–22.
- wiesenhu¨tter, e., 1976. Research into the relative importance of Tabanidae (Diptera) in mechanical disease transmission. IV. A contribution to the epidemiology of bovine trypanosomiasis in Africa. Tropical Animal Health and Production, 8, 195–201.
- zumpt, f., 1949. Medical and veterinary importance of horse-flies. South African Medical Journal, 23, 359–362.




