- Infectious Diseases of Livestock
- Part 1
- Vectors: Culicoides spp.
- Vectors: Ticks
- Vectors: Tsetse flies
- Vectors: Muscidae
- Vectors: Tabanidae
- Vectors: Culicoides spp.
- Vectors: Mosquitoes
- Classification, epidemiology and control of arthropod-borne viruses
- Special factors affecting the control of livestock diseases in sub-Saharan Africa
- The control of infectious diseases of livestock: Making appropriate decisions in different epidemiological and socioeconomic conditions
- Infectious diseases of animals in sub-Saharan Africa: The wildlife⁄livestock interface
- Vaccination: An approach to the control of infectious diseases
- African animal trypanosomoses
- Dourine
- Trichomonosis
- Amoebic infections
- GENERAL INTRODUCTION: COCCIDIA
- Coccidiosis
- Cryptosporidiosis
- Toxoplasmosis
- Besnoitiosis
- Sarcocystosis
- Balantidiosis
- Leishmaniosis
- Neosporosis
- Equine protozoal myeloencephalitis
- GENERAL INTRODUCTION: BABESIOSES
- Bovine babesiosis
- Equine piroplasmosis
- Porcine babesiosis
- Ovine babesiosis
- GENERAL INTRODUCTION: THEILERIOSES OF CATTLE
- East Coast fever
- Corridor disease
- Zimbabwe theileriosis
- Turning sickness
- Theileria taurotragi infection
- Theileria mutans infection
- Theileria annulata theileriosis
- Theileriosis of sheep and goats
- Theileria buffeli⁄orientalis infection
- Non-pathogenic Theileria species in cattle
- GENERAL INTRODUCTION: RICKETTSIAL, CHLAMYDIAL AND HAEMOTROPIC MYCOPLASMAL DISEASES
- Heartwater
- Lesser known rickettsial infections in animals and humans
- Chlamydiosis
- Q fever
- Eperythrozoonosis
- Bovine Haemobartonellosis
- Potomac horse fever
- GENERAL INTRODUCTION: ANAPLASMOSES
- Bovine anaplasmosis
- Ovine and caprine anaplasmosis
Vectors: Culicoides spp.
This content is distributed under the following licence: Attribution-NonCommercial CC BY-NC  View Creative Commons Licence details here
View Creative Commons Licence details here
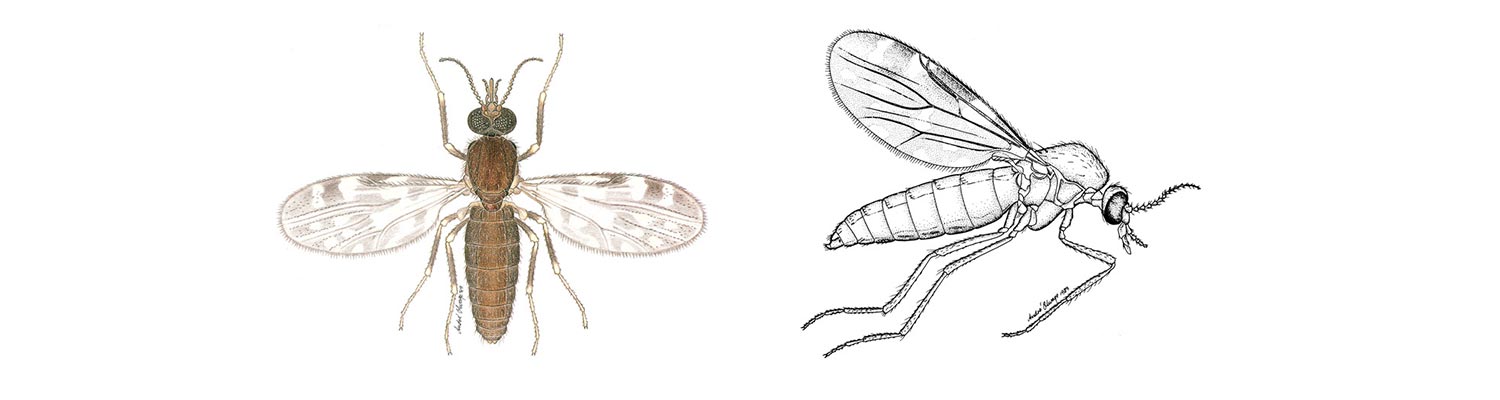
Vectors: Culicoides spp.
R MEISWINKEL, G J VENTER AND E M NEVILL
Introduction
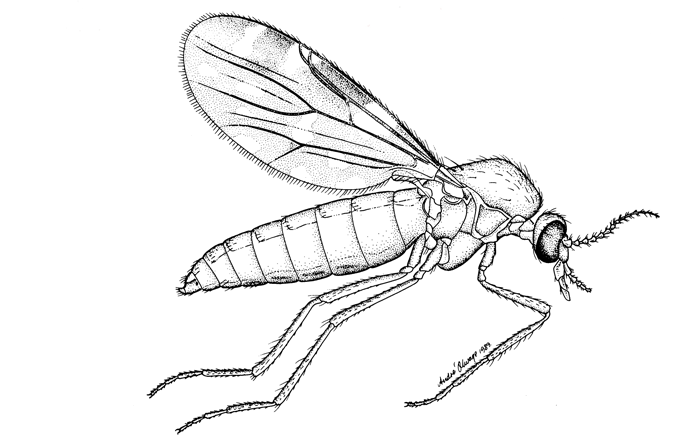
Culicoides biting midges are mosquito-like in their behaviour with the females of nearly all species being obligate blood-suckers (Figures 5.1 and 5.2) and being active only at night, especially when environmental conditions are calm and warm. The males feed on plant sap. Culicoides are 1 to 3 mm in size and are thus considerably smaller than mosquitoes but are more abundant and, on occasion, are found in enormous numbers in climates ranging from tropical to temperate. Most species possess grey- and white-patterned wings; these patterns being a very useful aid in their identification. To date 1 210 species of Culicoides have been described worldwide38 but it is certain that many more await discovery; for example, one third of the 112 species known to occur in South Africa are new to Science.216
Culicoides are well represented in the fossil record (39 species) with the lineage of the family Ceratopogonidae going back at least 130 million years.38 Midges clearly recognizable as Culicoides have been described from upper Cretaceous amber (88 to 93,5 million years ago), and it is therefore probable that Culicoides once fed on dinosaurs. A modern equivalent is the exclusive association between some Culicoides spp. and the African elephant (Loxodonta africana).223 At present 38 subgenera comprise the genus Culicoides but many more await delineation. This taxonomic diversity is testimony to a long and complex evolutionary history reflected in the fact that Culicoides today feed on a broad spectrum of hosts including reptiles, mammals, birds and humans. It has been shown that in southeast Asia some Culicoides spp. will even feed on bloodengorged mosquitoes. Their blood-thirsty attacks on humans in some parts of the world are legion, and have earned them scientific names such as damnosus, irritans, vexans and diabolicus. These attacks can be so intense as to retard development and production in industries such as forestry and tourism.155, 202 In Africa animals appear to be the hosts most intensively bitten. Under exceptional circumstances, such as prevail during overly wet periods, more than one million Culicoides can be captured in a single blacklight trap set at a horse stable on a warm summer night. Remarkably, such numbers probably represent only 1 per cent of the Culicoides population actively seeking blood, and illustrates the nightly levels of irritation, and possible risk of infection, that livestock may seasonally have to endure. 219
Certain species have a proven involvement in the transmission of at least two of the 15 OIE list ‘A’ diseases, namely bluetongue (BT) and African horse sickness (AHS). They also transmit epizootic haemorrhagic disease (EHD) of deer, and equine encephalosis (EE).116, 328 Thus, in sub-Saharan Africa, Culicoides are of great economic and veterinary significance. Indeed, it has been suggested that Culicoides midges were involved in two of the ten biblical plagues of ancient Egypt.210 This is not improbable, as the suppressant effect of Culicoides-transmitted disease must have manifested soon after the domestication of livestock some ten millennia ago. In support of this contention is that epidemics of devastating extent have occurred in the modern era, the most no table being when AHS swept through the Near and Middle East in 1959/60 causing an estimated 300 000 deaths.164 Another example is the AHS epidemic that decimated 70 000 equids in the then Cape of Good Hope Colony in southern Africa during the 1854/55 ‘season’.19 The unprecedented scale of the latter outbreak, which claimed 40 per cent of the colony’s total horse population in a mere eight months, attests not only to the efficiency of Culicoides as disseminators of infection but also to the modern-day necessity of protective annual vaccination. In eastern and southern Africa specifically this prophylactic regimen cannot be abandoned as it has long been known that there is a clear link between above average rainfall and outbreaks of Culicoides-borne orbiviral diseases.
The first studies on sub-Saharan Culicoides date back to 1908 when two species were described from Namibia122 but, whilst of a high standard, the subsequent continental research effort has been mostly short-term and fragmentary. The last decade, however, has brought significant advances especially in our basic understanding of Culicoides systematics, which includes the discovery that the most important Old World vector of BT and AHS, C. imicola, is but one member of a complex that comprises at least 10 species.
Not only does each species have a unique biology, but laboratory infection studies have shown that a recently described species within the complex, C. bolitinos, may in certain instances be a more efficient field vector of bluetongue virus (BTV) than C. imicola.
Further insights gained from field outbreaks strongly support the laboratory findings around C. bolitinos, and thus the ‘single-vector status quo’ that has stood since the time of du Toit’s seminal studies115 on C. imicola more than 50 years ago, is gradually being eroded. In addition, the longneglected study of the interaction between Culicoides and African game animals (which naturally cycle the abovementioned viruses) has finally received some attention, and yielded much that is of interest. This growth in basic knowledge was not only multi-disciplinary in its embrace but also formed part of an attempt to ‘modernize’ the study of Culicoides in Africa. Another consequence was the production of three satellite-based predictive risk maps for C. imicola, the first for any species of Culicoides in the world.
Also harnessed for the first time were molecular techniques such as the random-amplified polymorphic DNA-polymerase chain reaction (RAPD-PCR) and mitochondrial DNA (mtDNA) extraction. Not only have these studies resulted in a partial phylogeny of the Imicola complex but, importantly, have also reaffirmed the stability of ‘old-fashioned’ morphological identifications, and so reassure us that the steady advances being made on the unique life cycle of each species do indeed reflect ‘realities’ in nature. These advances are reviewed here, and because the research has focused principally on vectors of orbiviruses that affect livestock, this chapter concentrates on assembling much of the knowledge accumulated thus far on the two proven vectors C. imicola and C. bolitinos. Included is an introductory preamble of the standardized protocols used to collect, subsample and age-grade Culicoides, which were developed principally in South Africa over the last 30 years, and are now used in various parts of Africa and beyond. It is essential that data collected over a wide geographic area and during a number of seasons and years be comparable so that accurate distribution databases can be created for the subsequent development of disease risk-maps. At various junctures in this review an indication is given of the research still needed to refine our understanding of African Culicoides and the diseases they transmit.
Collecting adult Culicoides
The majority of investigations conducted on Culicoides worldwide deal primarily with the monitoring of disease vectors as their acknowledged role in epidemics of disease impacts directly on management practices, such as vaccination, and quarantine and export of live animals to international destinations. The primary, almost sole, monitoring tool used for the capture of Culicoides is the light trap. However, other trapping methods of some variety have been developed over many years for the capture of adult Culicoides. Those most commonly used in Africa include various models of white- or blacklight traps, emergence traps (for larval habitat studies), aspirators (for the collection of live midges off human and animal hosts), and truck traps (which involve the use of a large net mounted on top of a slow-moving vehicle). The trapping method employed depends upon the purpose of the research as each method enjoys some advantages not shared by the others. Their uses, and the merits and disadvantages of each, are briefly reviewed below.
Blacklight traps
These traps are preferred over those with whitelight as they apparently attract larger numbers and varieties of insects. This, and their flexibility in application, make the blacklight trap the tool of choice when biting midge abundance levels are to be determined during outbreaks of disease, and when large-scale faunistic surveys are being conducted. The Culicoides are captured in a bowl of liquid (suspended below a suction fan, which in turn is below the light source) in which they die. In a matter of hours these ‘wet’ captures make available large numbers of midges suitable for a number of purposes that include virus isolation, blood meal identification, age-grading, and taxonomic studies. When the light trap is set near livestock (usually at a height varying between 1 and 2m) a few small drops of detergent are added to the bowl of water to break the surface tension. This is necessary because midges are too small and light to drown and sink to the bottom, with the result that in the morning, when the trap is switched off, a number of them may recover and fly off. With minor modification the same trap can be used to catch large numbers of live midges for vector competence studies in the laboratory. In this instance a gauzed cage replaces the bowl of liquid. Alternatively, the bowl may be retained but is partially filled with loosely crumpled tissue paper so that the live, captured midges can creep into recesses away from the incessant wind created by the suction fan. Another advantage of the blacklight trap is that it is easy to use as it can be rigged in a short space of time and, furthermore, does not have to be monitored until morning, when it can be dismantled equally quickly. What is time-consuming, however, is the journey to disease outbreak points and choices to be made as to which sites will produce optimal catches. In addition, the journey has to be made twice as the traps must be retrieved the following morning. Light traps cannot be left in position for more than one night, and should not be allowed to hang in sunlight as the captured midges, especially those in a weak soapy detergent solution, start to decompose very quickly in tropical or subtropical daytime temperatures. Another disadvantage is that most light traps require electricity, which greatly restricts their use in rural situations.
Whilst it is possible to run these traps using a generator, the transport and movement of such heavy equipment means that very few geographic points can be sampled per night. Light traps can be adapted to run on 12-volt batteries but here, too, the transport and maintenance of heavy batteries can be burdensome and impractical. In this context it must also be borne in mind that the various modifications made to traps to capture Culicoides in out of the way places must not result in their lowered performance as this will provide data of limited comparative value. A further disadvantage is that the traps are equipped with a strong suction fan, which can damage Culicoides. This can be particularly vexing when rare or new species have been collected which must be in a suitable state for specialized taxonomic studies. Being powerful, these traps also capture large numbers of other insects including moths and, under certain conditions, this can result in collections that are extremely dirty and that take a long time to clean. Larger insects can also damage the smaller midges and is another reason why blacklight traps should be covered with a fine gauze mesh to exclude the capture of all insects larger than 2 mm. As mentioned, exceptionally large numbers of Culicoides can be captured in blacklight traps. To be able to determine this total, and the exact number of each species collected, it is essential to be able to subsample catches easily and rapidly as hundreds of collections may be made during a large survey. A method of subsampling Culicoides has been developed325 and is widely employed. Because of their strong attractant power, blacklight traps are considered biased samplers and therefore in more specialized studies, such as those concerned with host preference, other more ‘objective’ collecting techniques must be employed.
Vehicle-mounted traps
For obvious reasons the blacklight trap is useless for the study of Culicoides that may be active by day. For this, vehicle-mounted traps (commonly referred to as ‘truck traps’) can be used to capture flying Culicoides throughout the diel, and in this way hourly activity rates may be determined; the results can also be related to prevailing meteorological conditions such as temperature, relative humidity and windiness. Vehicle- mounted traps are particularly successful for the collection of large numbers of biting midges during the hours of dusk and dawn. In addition, male swarms, which may not be attracted to light traps, can be captured in this way, at times in large numbers. If collection periods are limited to 15 minutes, the midges captured will be in pristine condition and therefore suitable for the preparation of material on glass slides for taxonomic study. Because Culicoides are host-orientated, truck trapping greatly enhances the possibility of capturing Culicoides near large groups of wildlife that are either dangerous and/or scatter when approached on foot. It is, in addition, very difficult to set light traps near wildlife; this being compounded by the fact that herds are constantly on the move. Disadvantages of truck trapping are the cost of fuel for the vehicle and the time required for driving and trapping.
Aspirators and sweep nets
Aspirators (or ‘pooters’) and hand-operated sweep nets are used in specialized host preference studies when live Culicoides must be captured off tethered animals or humans. A hand-held pooter can be used but this requires that each individual midge be located, using a red torchlight, prior to capture, which is time-consuming. More commonly the ‘sweeping’ of marked areas of the host with a small handheld domestic vacuum cleaner is the preferred method. In this way more Culicoides may be captured more rapidly, which is necessary if it has to be established exactly which areas of the host are being attacked within a specified time slot. In life cycle studies in which the capture of gravid females for oviposition determinations is required, the midges are best obtained by pootering those attracted to a source of light.
Traps baited with carbon dioxide have also been used to collect Culicoides. The particular advantage of these traps is that they enable the collection of diurnal species. Limitations of carbon dioxide traps are that they require dry ice and that the unregulated release of carbon dioxide gas may provide concentrations that are attractive to some species and repellent to others.
Emergence traps
These are made of fine netting, are conical in shape, and have a collection bottle at the apex that is lined with a sticky substance or contains a liquid.276 An emergence trap (built to cover a specific unit area) is placed over a suspected Culicoides larval habitat and remains in situ where it can be monitored hourly, daily, or weekly. In this way emergence rates and species-association profiles can be obtained. The chemical composition and structure of the soil, and the amount of water it holds can, in addition, also be determined for a particular emergence site. Furthermore, during emergence, if larvae or pupae must be retrieved, samples of the substrate can be extracted and taken to the laboratory where a saturated sugar solution is added to them to alter the specific gravity in order to induce the larvae and pupae to float to the surface. The pupae are retrieved with a spatula and placed in individual vials for eclosion. The resultant adult and its associated pupal pelt can then be mounted on a single glass slide for identification. The rearing of individual larvae so obtained is more complicated but can be achieved on agarose media to which cultured micro-organisms are added. This, or a similar, rearing strategy is essential towards understanding more precisely the ecology of individual Culicoides spp. as it is the availability of the larval habitat that determines primarily the prevalence of a species in any given area.
It should be noted that much more research is still required towards more clearly defining the larval habitats and food requirements of the immature stages of the vector species of Culicoides, including C. imicola and C. bolitinos.
Table 5.1 Micro-organisms associated with the genus Culicoides worldwide
| CULICOIDES SP. | MICRO-ORGANISM | REFERENCE |
|---|---|---|
| C. amamiensis | Macacanema formosana | 27, 28 |
| C. arubae | Onchocerca sp. | 31, 347 |
| C. arakawae | Leucocytozoon (Akiba) caulleryi | 1, 2, 123, 170, 199, 246–250, 284, 317 |
| Lemdaninae | 318 | |
| Onchocerca gutturosa | 98, 113, 201, 318 | |
| C. arboricola | Haemoproteus meleagridis | 7 |
| C. adersi | Hepatocystis (Plasmodium) kochi | 137–139, 146, 147 |
| C. austeni | Dipetalonema perstans | 161, 162, 270 |
| C. circumscriptus | Leucocytozoon (Akiba) caulleryi | 146 |
| C. crepuscularis | Haemoproteus danilewskyi | 146 |
| H. fringillae | 146 | |
| Chandlerella quiscali | 166 | |
| C. downesi | Haemoproteus nettionis | 130, 146, 295 |
| C. edeni | Haemoproteus meleagridis | 7, 8, 10 |
| Vavraia sp. | 9 | |
| C. fulvithorax | Hepatocystis (Plasmodium) kochi | 137–139, 146, 147 |
| Onchocerca gutturosa | 98, 113, 201, 318 | |
| C. furens | Dipetalonema marmosetae | 204 |
| D. ozzardi | 58, 59, 132, 206 | |
| C. grahamii | Dipetalonema | 114, 156, 270, 271, 292, |
| perstans | 293 | |
| D. streptocercum | 68, 69, 156 | |
| C. haematopotus | Haemoproteus meleagridis | 7 |
| C. hinmani | Haemoproteus meleagridis | 7 |
| C. hollensis | Dipetalonema gracile | 119 |
| D. caudispina | 119 | |
| D. llewellyni | 350 | |
| D. marmosetae | 204 | |
| C. inornatipennis | Dipetalonema | 114, 156, 270, 271, 292, |
| perstans | 293 | |
| C. insinuatus | Dipetalonema ozzardi | 205, 232, 257, 312, 322 |
| C. kingi | Onchocerca gutturosa | 98, 113, 201, 318 |
| C. knowltoni | Haemoproteus meleagridis | 7 |
| C. krameri | Onchocerca gutturosa | 98, 113, 201, 318 |
| C. marksi | Onchocerca sp. | 31, 348 |
| O. gibsoni | 31, 60, 124, 201 | |
| C. multidenatus | Splendidofilaria californiensis | 341 |
| C. nubeculosus | Trypanosoma bakeri | 238 |
| T. chabaudi | 67 | |
| T. davidmolyneuxi | 67 | |
| T. everetti | 67 | |
| T. gentilinii | 67 | |
| Hepatocystis brayi | 239 | |
| H. levinei | 40, 195 | |
| Haemoproteus handai | 240 | |
| Eufilariella bartlettae | 11 | |
| E. delicata | 11 | |
| E. kalifai | 237 | |
| Onchocerca cervicalis | 133, 134, 157, 229, 230 | |
| O. gibsoni | 31, 60, 124, 201 | |
| O. gutturosa | 99, 113, 201, 318 | |
| O. reticulata | 244, 245 | |
| Dipetalonema ozzardi | 205, 232, 257, 312, 322 | |
| C. obsoletus | Onchocerca cervicalis | 76, 151, 157, 180, 201, 231, 301 |
| C. odibilis | Leucocytozoon (Akiba) caulleryi | 146 |
| C. oxystoma | Onchocerca gibsoni | 31, 60, 124, 201 |
| C. orientalis | Onchocerca gibsoni | 31, 60, 124, 201 |
| C. parroti | Onchocerca cervicalis | 76, 151, 157, 180, 201, 231, 301 |
| C. phlebotomus | Dipetalonema ozzardi | 205, 232, 257, 312, 322 |
| C. pulicaris | Francisella tularensis | 176, 273 |
| C. pungens | Onchocerca gibsoni | 31, 60, 124, 201 |
| C. riethi | Dipetalonema ozzardi | 205, 232, 257, 312, 322 |
| C. schultzei | Leucocytozoon (Akiba) caulleryi | 146 |
| C. shortti | Onchocerca gibsoni | 31, 60, 124, 201 |
| C. sphagnumensis | Trypanosoma avium | 25 |
| Haemoproteus canachites | 146 | |
| H. danilewskyi | 146 | |
| H. mansoni | 7 | |
| H. (Parahaemoproteus) velans | 146 | |
| C. stilobezzioides | Trypanosoma avium | 25, |
| Haemoproteus fringillae | 146 | |
| H. (Parahaemoproteus) velans | 146 | |
| Chandlerella chitwoodae | 17 | |
| C. travisi | Chandlerella chitwoodae | 17 |
| C. trifasciellus | Onchocerca gutturosa | 98, 113, 201, 318 |
| C. variipennis | Trypanosoma sp. | 161 |
| Hepatocystis brayi | 239 | |
| Onchocerca cervicalis | 133, 134, 157, 229, 230 | |
| Dipetalonema ozzardi | 205, 232, 257, 312, 322 | |
| Culicoides spp. | Leishmania sp. | 70 |
| Plasmodium malariae | 91 | |
| Leucocytozoon neavei | 129 | |
| L. schoutedeni | 129 | |
| L. (Akiba) sp. | 12, 126, 127, 187, 294 | |
| Haemoproteus | 147 | |
| canachites | ||
| H. sp. | 26, 79, 125–128, 130, 144, 147 | |
| Ornithofilaria fallisensis | 5 | |
| Onchocerca gibsoni | 31, 60, 124, 201 | |
| O. volvulus | 141, 326 |
Livestock and wildlife diseases/infections associated with Culicoides
Summer seasonal recurrent dermatitis
Summer seasonal recurrent dermatitis is most commonly referred to as sweet itch, but is also known as Queensland itch, ‘dhobie itch’, kasen disease, allergic dermatitis and sommerekzem. It is a chronic, seasonally superficial allergic dermatitis of horses that usually affects the skin of the mane, tail and withers resulting from the bites of Culicoides midges. Various species are involved. This condition was first recognized in France in 1840, but has since been investigated in many countries.55, 56, 78, 148, 188, 256, 323, 349 In Israel C. imicola was implicated as the principal species involved as 70 per cent of 195 specimens caught on a horse showed a clear preference for feeding on the dorsal ridge, which overlaps the itch zone.42 In the UK a significant and rapid reduction in the prevalence of sweet itch was demonstrated once affected horses were more regularly stabled.234 Virtually no studies have been conducted on the condition in Africa but it is suspected to occur in South Africa.324
Diseases associated with Nematoda: Filarioidea
Some 20 species of filarial nematodes are transmitted by Culicoides midges and include five species of Onchocerca in cattle, horses and water buffalo (Bubalis bubalus), and two species of Dipetalonema in monkeys (Table 5.1).201
Diseases associated with Protozoa
Worldwide, at least 33 species of protozoa in the orders Eucoccida and Kinetoplastida are known to be transmitted only by Culicoides (Table 5.1).201 Except for a species of Hepatocystis found in several species of monkeys in Africa and another in the Malaysian sciurid squirrels of the genera Callosciurus and Sundasciurus, the other 10 protozoa occur in birds. One of these, Leucocytozoon caulleryi, is an economically important disease of poultry in south-east Asia.
Diseases associated with viruses
Of all the pathogens transmitted by Culicoides, viruses are of the greatest veterinary importance, and, more especially, those that cause BT, AHS, EE and Akabane disease. Other viruses closely related to Akabane virus have also been isolated from Culicoides (Table 5.2). Although many of the viruses that are Culicoides-associated have been isolated from, but occur more commonly in, other arthropods such as mosquitoes, argasid and ixodid ticks, and phlebotomines, there remain 45 per cent which have been isolated only from Culicoides midges.
Worldwide, some 64 named and 11 unnamed arboviruses are Culicoides-associated. Of these, 25 are from Australia and 23 from Africa. Of the 75 viruses listed in Table 5.2, 46 have been isolated from pools of identified Culicoides spp. rather than from pools containing mixed species. Twenty-three of these viruses are from the Culicoides subgenus Avaritia alone, 15 from C. brevitarsis in Australia, and eight from C. imicola in Africa, with BTV being common to both continents. Although these figures may be biased in that they reflect both the specific directions taken in Culicoides research and the particular drive of the research team involved, they do show that the tropics and subtropics are particularly rich in both bunya- and orbiviruses.
Bunyaviridae
The Simbu group viruses (see Diseases caused by Akabane and related Simbu-group viruses) that are found in southern Africa90 include Akabane virus, the cause of abortion and congenital deformities in domestic ruminants. Akabane virus is widespread in southern Africa occurring in at least 25 species of wildlife,3 and the prevalence of antibodies to it in African buffalo (Syncerus caffer), blue wildebeest (Connochaetes taurinus) (as high as 69 per cent in Namibia) and African elephant (Loxodonta africana) strongly implicates them in its maintenance in nature. Sabo virus,90, 321 closely related to Akabane virus, also occurs in cattle and Culicoides midges in South Africa, whilst Shamonda virus has been isolated from C. imicola collected near cattle at the Onderstepoort Veterinary Institute in South Africa.211 In addition, in South Africa a strain of Shuni virus has been recovered from the brain of an adult mare that died after showing nervous signs. Subsequent serological surveys revealed positive antibody titres in horses to Akabane, Shuni, Shamonda and Sabo viruses, with the latter two predominant. Their presence may explain some of the febrile reactions and other vague clinical signs observed in horses seasonally.165 Although they have not yet been tested for pathogenicity in ruminants, South African strains of Sabo, Shamonda and Shuni viruses induce marked teratogenic effects in chicken embryos.90 Aino virus, which is closely related antigenically to Shuni virus, has, in Japan and Australia, been implicated as the cause of congenital anomalies in cattle and sheep.110, 316
Reoviridae
Within this family, the orbiviruses of BT, AHS, and EE are commonly associated with causes of livestock diseases in southern Africa. Other members of this family include EHD, Nyabira, Gweru and Kasba (= Chuzan) viruses. Both Nyabira and Gweru are little understood Palyam-serogroup viruses (see Palyam serogroup orbivirus infections) that have been found only in southern Africa but, like Akabane virus, are linked to abortions and teratology in cattle, goats and sheep.314, 344 Viruses of the Palyam serogroup have a particular association with cattle, and the many isolations made from Culicoides suggest that they are competent vectors for Palyam viruses in southern Africa.345 Nyabira virus, for instance, replicates well in C. imicola and C. zuluensis, but transmission trials have proved inconclusive. 54 There is serological evidence for the widespread distribution of Palyam-serogroup viruses throughout southern Africa except for the cooler winter rainfall region of the Western Cape Province of South Africa. Since they may be abortigenic in cattle, there is clearly a need for pathogenicity trials in livestock.345
Table 5.2 Viruses isolated from the genus Culicoides worldwide
| CULICOIDES SP. | SUBGENUS | VIRUS | ABBREVIATION | REFERENCE |
|---|---|---|---|---|
| C. actoni | Avaritia | Warrego | WAR | 149 |
| C. agathensis | Unassigned | Alphavirus sp. | 53 | |
| C. algecirensis | Monoculicoides | Bovine ephemeral fever | BEF | 296 |
| C. austropalpalis | Similis grp. | Kununurra | KNA | 149 |
| Wongabel | 225 | |||
| C. bedfordi | Unidentified | 266 | ||
| C. bolitinos | Avaritia | African horse sickness | AHS | 226 |
| Bluetongue | 16 | |||
| *Letsitele | 266 | |||
| C. brevitarsis | Avaritia | Aino | AINO | 111 |
| Akabane | AKA | 87, 99, 108, 111, 308 | ||
| Bluetongue | BT | 302, 307 | ||
| Bovine ephemeral fever | BEF | 87 | ||
| Bunyip Creek | BC | 87, 88 | ||
| CSIRO Village | CVG | 298 | ||
| D’Aguilar | DAG | 110, 111 | ||
| Douglas | DOU | 304 | ||
| Epizootic haemorrhagic disease of deer | EHD | 277, 306 | ||
| Kimberley | KIM | 351 | ||
| Ngaingan | NGA | 110 | ||
| Peaton | PEA | 310 | ||
| Tibrogargan | TIB | 89 | ||
| Tinaroo | TIN | 86, 304 | ||
| Wallal | WAL | 175 | ||
| Unidentified | 111, 305 | |||
| C. brevitarsis + C. schultzei** | Mixed | Unidentified | 305 | |
| C. bundyensis | Unassigned | Belmont | BEL | 149 |
| C. coarctatus | Unassigned | Bovine ephemeral fever | BEF | 34 |
| C. cockerellii | Sylvicola | Bluetongue | BT | 190 |
| C. cornutus | Monoculicoides | Epizootic haemorrhagic disease of deer | EHD | 16 |
| C. dycei | Unassigned | Wallal | WAL | 110, 111 |
| Warrego | WAR | 110 | ||
| C. exspectator | Similis grp. | Bluetongue | BT | 266 |
| C. filarifer | Unassigned | Bluetongue | BT | 242 |
| C. filarifer + C. pusillus | Mixed | Bluetongue | BT | 149 |
| C. fulvus | Avaritia | Bluetongue | BT | 298 |
| C. fulvus + C. orientalis | Mixed | Bluetongue | BT | 291 |
| C. gulbenkiani | Avaritia | Letsitele | 266 | |
| C. histrio | Meijerehelea | Thimiri | TIM | 298 |
| C. imicola | Avaritia | Akabane | AKA | 4, 34 |
| African horse sickness | AHS | 34, 266 | ||
| Bluetongue | BT | 16, 34, 53, 143, 235, 266, 339 | ||
| Bovine ephemeral fever | BEF | 34 | ||
| Equine encephalosis | EE | 266 | ||
| *Letsitele | 266 | |||
| *Nyabira | 34 | |||
| Sabo | SABO | 66, 175, 197 | ||
| Shamonda | SHA | 175, 197 | ||
| Simbu | 266 | |||
| Unidentified | 97 | |||
| C. imicola + C. schultzei** | Mixed | Unidentified | 97 | |
| C. insignis | Hoffmania | *Bivens Arm | 140 | |
| Bluetongue | BT | 77, 145, 242 | ||
| Sweetwater Branch | 225 | |||
| C. kingi | Remmia | Epizootic haemorrhagic disease of deer | EHD | 235 |
| Unidentified | 235 | |||
| C. magnus | Culicoides | *Letsitele | 266 | |
| C. marksi | Unassigned | Belmont | BEL | 298 |
| Barmah | BF | 298 | ||
| Eubenangee | EUB | 109, 298 | ||
| *Leanyer | 298 | |||
| *Mudjinbarry | 112 | |||
| *Parker’s Farm | 298 | |||
| Wallal | WAL | 110, 298 | ||
| Warrego | WAR | 110, 175 | ||
| Unidentified | 300 | |||
| C. milnei | Hoffmania | Akabane | AKA | 34 |
| Bluetongue | BT | 339 | ||
| C. nevilli | Remmia | Epizootic haemorrhagic disease of deer | EHD | 16 |
| C. newsteadi | Culicoides | Alphavirus sp. | 53 | |
| C. nivosus | Meijerehelea | Thimiri | TIM | 225 |
| C. nivosus | Meijerehelea | Thimiri | TIM | 225 |
| C. obsoletus | Avaritia | Bluetongue | BT | 236 |
| C. oxystoma | Remmia | Akabane | AKA | 192, 193 |
| Bunyip Creek | BC | 225 | ||
| Kasba | KAS | 194 | ||
| C. pallidothorax | Unassigned | Wongorr | WGR | 298 |
| C. paraensis | Unassigned | Ananindeua | ANU | 175 |
| Oropouche | ORO | 196 | ||
| C. peregrinus | Hoffmania | *Beatrice Hill | 298 | |
| C. peregrinus + C. schultzei** | Mixed | Unidentified | 88, 305 | |
| C. punctatus | Culicoides | Barur | BAR | 174 |
| C. pusillus | Avaritia | Bluetongue | BT | 242 |
| C. pycnostictus | Meijerehelea | Bluetongue | BT | 266 |
| C. schultzei** | Remmia | Bovine ephemeral fever | BEF | 97 |
| Bunyip Creek | BC | 298 | ||
| D’Aguilar | DAG | 111 | ||
| Epizootic haemorrhagic disease of deer | EHD | 196, 198 | ||
| Kasba | KAS | 194, 197, 198, 241 | ||
| Keterah | KTR | 259 | ||
| Marrakai | MAR | 298 | ||
| C. schultzei** + C. imicola | Mixed | Kasba | KAS | 97 |
| C. schultzei** + C. peregrinus | Mixed | Marrakai | MAR | 88 |
| C. schultzei grp | Remmia | Unidentified | 266 | |
| C. stellifer | Unassigned | Vesicular stomatitis New Jersey | VSNJ | 190, 340 |
| C. tororoensis | Avaritia | Nairobi sheep disease | NSD | 97, 339 |
| Bluetongue | BT | 340 | ||
| C. variipennis | Monoculicoides | Bluetongue | BT | 135, 136, 190, 203, 285, 346 |
| Bunyavirus sp. | 154 | |||
| Buttonwillow | BUT | 153, 154, 175, 190, 258, 287 | ||
| Epizootic haemorrhagic disease of deer | EHD | 145, 172, 354 | ||
| Lokern | LOK | 85, 190, 258 | ||
| Main Drain | MD | 85, 121, 258 | ||
| Vesicular stomatitis New Jersey | VSNJ | 175, 190, 340 | ||
| C. wadai | Avaritia | Akabane | AKA | 311 |
| C. zuluensis | Hoffmania | Letsitele | 266 | |
| Unidentified | 97 | |||
| C. zuluensis + C. imicola | Mixed | Unidentified | 97 | |
| Culicoides spp. | Selfia | Lokern | LOK | 190 |
| Main Drain | MD | 190 | ||
| Vesicular stomatitis New Jersey | VSNJ | 190, 340 | ||
| Culicoides spp. | Mixed | African horse sickness | AHS | 97, 175, 233, 266 |
| Aino | AINO | 175 | ||
| Akabane | AKA | 175, 321 | ||
| Bluetongue | BT | 32, 44, 197, 198, 243, 266, 283, 309 | ||
| Bovine ephemeral fever | BEF | 95, 97 | ||
| *Buritirana | 150 | |||
| Buttonwillow | BUT | 175, 190 | ||
| Congo-Crimean haemorrhagic fever | CCHF | 197 | ||
| CSIRO Village | CVG | 88 | ||
| D’Aguilar | DAG | 300 | ||
| Douglas | DOU | 298 | ||
| Dugbe | DUG | 197 | ||
| Eastern equine encephalomyelitis | EEE | 177 | ||
| Epizootic haemorrhagic disease of deer | EHD | 197, 198, 266 | ||
| Equine encephalosis | EE | 266 | ||
| *Gweru meningoencephalitis | 345 | |||
| *Itacaiunas | 149 | |||
| Kasba | KAS | 197, 198 | ||
| Kotonkan | KOT | 179, 197 | ||
| *Letsitele | 266 | |||
| Marrakai | MAR | 298 | ||
| Mitchell River | MR | 110 | ||
| Nairobi sheep disease | NSD | 197 | ||
| Ngaingan | NGA | 110, 178 | ||
| *Nyabira | 345 | |||
| Palyam | PAL | 266 | ||
| Rift Valley fever | RVF | 92, 94, 96 | ||
| Sabo | SABO | 66, 175, 197 | ||
| Sango | SAN | 66, 197 | ||
| Sathuperi | SAT | 66, 175, 197 | ||
| Shamonda | SHA | 175, 197 | ||
| Shuni | SHU | 66, 197 | ||
| Simbu | 266 | |||
| Tahyna | TAH | 150 | ||
| Utinga | UTI | 186 | ||
| Wallal | WAL | 110 | ||
| Warrego | WAR | 111 | ||
| *Weldona | 63 | |||
| Unidentified | 197, 266, 300, 321 |
A third Palyam-serogroup virus (Kasba virus) has been associated with congenital abnormalities in calves in Japan and has been isolated from C. oxystoma.241 This Culicoides is a member of the subgenus Remmia and is widely and abundantly found in south-east Asia and the eastern Palaearctic. Similarly, in Africa, the Mediterranean region and the Near and Middle East seven or eight species of Remmia can also be found in some abundance, particularly in hotter, drier regions; these therefore deserve consideration as potential vectors of various reoviruses. It was from a mixed pool of two Remmia spp. (including C. kingi) that an EHD virus was isolated in the Sudan.235 The first isolation of equine encephalosis virus (EEV) was made from a mixed pool of Culicoides consisting mainly of C. imicola collected at the Onderstepoort Veterinary Institute.321
Rhabdoviridae
In this family is included bovine ephemeral fever virus (BEFV). The seasonal occurrence of BEF (see Bovine ephemeral fever) suggested insect transmission to early investigators in Africa29, 30 although firm evidence of insect involvement was not provided until the causative virus was isolated from a pool of several species of Culicoides in Kenya in 1971.95, 97 It has been recovered from Culicoides only twice in southern Africa, namely from C. imicola and C. coarctatus in Zimbabwe.34 Its low isolation rate is possibly due to it being particularly difficult to isolate using standard cell culture techniques, and is further complicated by the fact that there are many other rhabdo-viruses closely related antigenically to BEFV.303 In Australia BEFV has been isolated from C. brevitarsis,253 the sister species to the African C. bolitinos. Both species breed in cattle dung, with C. bolitinos also being found in the dung of the African buffalo and blue wildebeest.213
Two other rhabdoviruses, Bivens Arm and Sweetwater Branch, have been isolated from C. (Hoffmania) insignis collected near a water buffalo imported into North America from Trinidad140 but have not been linked to naturally occurring or experimentally induced disease. These viruses are related to Tibrogargan virus, which is associated with water buffalo, cattle and horses in Australia and has also not, as yet, been shown to be pathogenic.90 The Culicoides milnei complex, which, in Africa, is represented by at least 16 species, is very closely related to species of the subgenus Hoffmania. They can occur in large numbers and must therefore be considered as potential vectors of rhabdo-viruses.
Viral maintenance and transmission
Vector capacity
Vector capacity is defined as the average number of infective bites that will be delivered by a Culicoides midge feeding on a single host animal in one day, and is a combination of all of the following:
- midge density in relation to the host animal,
- host preference,
- midge biting frequency,
- vector competence (reviewed below), and
- life span of the infected midge and duration of infectivity.
The successful transmission of an insect-borne virus from an infected to a susceptible host is dependent upon the complex relationship that exists between the virus, its insect vector and the vertebrate host, with each being influenced by particular environmental conditions.152 Four criteria determine the competence of a particular vector species:6
- the virus must be recoverable from field-collected arthropods whose abdomens are free of fresh blood,
- the ability of the arthropod to become systemically infected by feeding on a viraemic host or an artificial substitute must be demonstrated,
- similarly, its ability to transmit the infection biologically by bite must be demonstrated, and
- there should be field evidence confirming the association of infected arthropods with diseased vertebrates.
Vector competence
Vector competence is a measure of the number of midges that actually become infective after feeding on a viraemic host. This competence is dependent upon the genetic makeup of the vector midge and external environmental influences. 315, 342, 348
The virus vector competency of a Culicoides sp. can be experimentally assessed by first allowing a number of midges to feed on a viraemic animal or, in the laboratory, on a blood-virus suspension through a membrane. The engorged midges are then kept alive for the extrinsic incubation period, i.e. the period between feeding on infected blood and the appearance of virus in the saliva of the midge and is in the order of one to two weeks. Following ingestion by a susceptible arthropod, most arboviruses infect and replicate in cells of the mesenteron before penetrating the basal lamina to be released into the haemolymph to set up more cycles of infection and replication. Further barriers to infection appear to exist at the levels of organs such as the ovaries and salivary glands. A virus must infect and replicate in the salivary glands before the extrinsic incubation period of the virus is successfully concluded and the arthropod is able to transmit the virus by bite. It is possible for arthropods to obtain an infection with great efficiency by ingestion of a viraemic blood meal, yet they may still fail to transmit virus because the infection has not spread to the salivary glands. Hence it is important in vector studies in the laboratory to distinguish between arthropod infection rates and transmission rates.313 The ability of these midges to transmit virus is then assessed by allowing them to feed on susceptible animal hosts or on suitable substitutes.
After a series of failed experiments involving mosquitoes, 272 Du Toit in 1943 conducted the first successful Culicoides vector competence studies at the Onderstepoort Veterinary Institute.115 He fed field-collected Culicoides on BTV-infected sheep, and after an extrinsic incubation period of 10 days, was able to transmit the disease to susceptible sheep, but he did not specifically identify the species of Culicoides used in his studies. It is most likely to have been C. imicola as it is by far the most abundant species collected around Onderstepoort. He is reported to have similarly infected a horse with African horse sickness virus (AHSV) by Culicoides bite.334
These seminal findings by Du Toit that Culicoides are involved in the transmission of orbiviruses were later confirmed elsewhere in the world, i.e. in North America, Australia, and the UK, and then again in South Africa.329 The latter study involved C. imicola. In the laboratory,329 C. imicola was fed, through latex and chicken skin membranes, on blood containing BTV serotypes 3 and 6 and AHSV serotype 1. After an incubation period of 10 days at 25 to 27 °C, the infection prevalences for C. imicola for BTV serotypes 3 and 6 were established at 31 and 24 per cent respectively, but no AHSV could be recovered.
In 1998, in the laboratory, a second species of the Imicola complex, C. bolitinos, was incriminated as a vector of BTV.335 Significantly, for the three serotypes of BTV tested, higher infection prevalences, and higher virus titres per midge, were found in C. bolitinos than in C. imicola. In addition, it was shown that these higher infection prevalences and mean virus concentrations per midge in C. bolitinos were not influenced by the incubation period (2 to 20 days), nor by incubation temperature (10 to 30 °C) at which the midges were held.281 These findings suggest that C. bolitinos may be a more competent vector of BTV than C. imicola. In a subsequent study282, 334 three more species of Culicoides were shown to replicate BTV, and so broadens our understanding of the epidemiology of BT. In the same trial the workers were unable to demonstrate virus infection, after incubation, in 14 of the 22 Culicoides spp. examined.
Artificial feeding of 17 species of field-collected Culicoides on blood containing three serotypes of AHSV succeeded in infecting only C. imicola and C. bolitinos.327 The findings suggest species-specific serotype dependence. It appears cogent that no AHSV replication could be demonstrated in any of the remaining 15 species assayed, but for most of them only low numbers of midges were available. It was shown also that of seven species (but not including C. bolitinos) fed artificially on blood infected with the Bryanston serotype of EEV, only C. imicola (22,3 per cent) became infected.328
Available data clearly indicate that considerable variation, in terms of susceptibility and prevalence of infection with different orbivirus species and serotypes, appears to exist amongst the various Culicoides spp. Species of the Imicola complex appear to be the Culicoides most susceptible to infection in the laboratory, and are therefore the likely vectors of these orbiviruses in the field. Other data, such as high levels of abundance, seasonal and geographic prevalence, and field isolations of virus, support this contention (see below).
Biosystematics of Culicoides
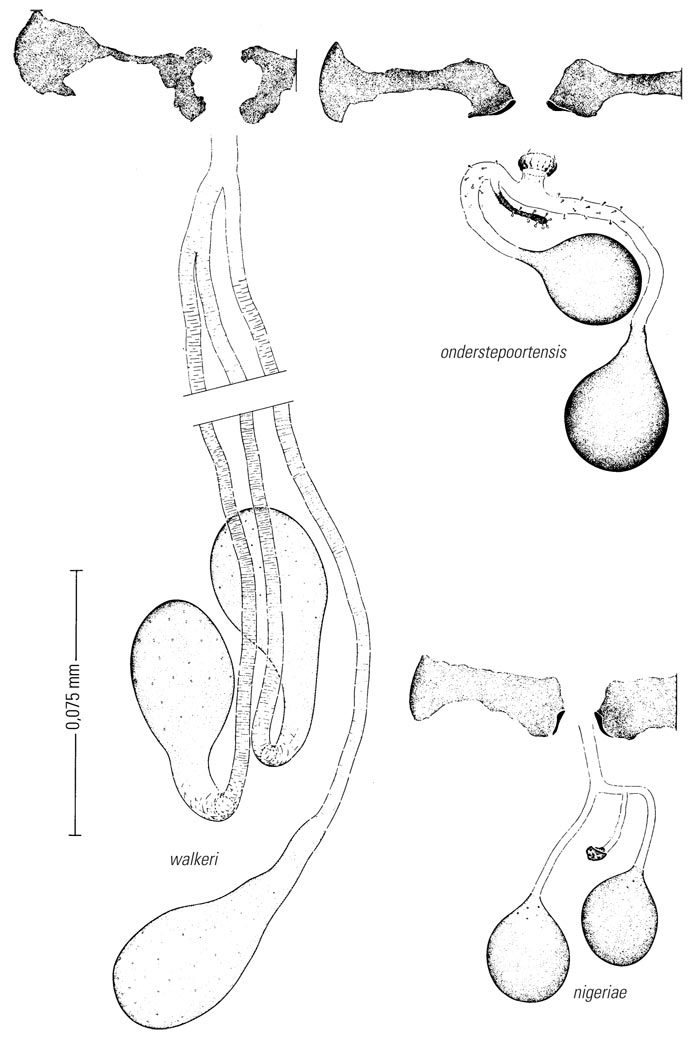
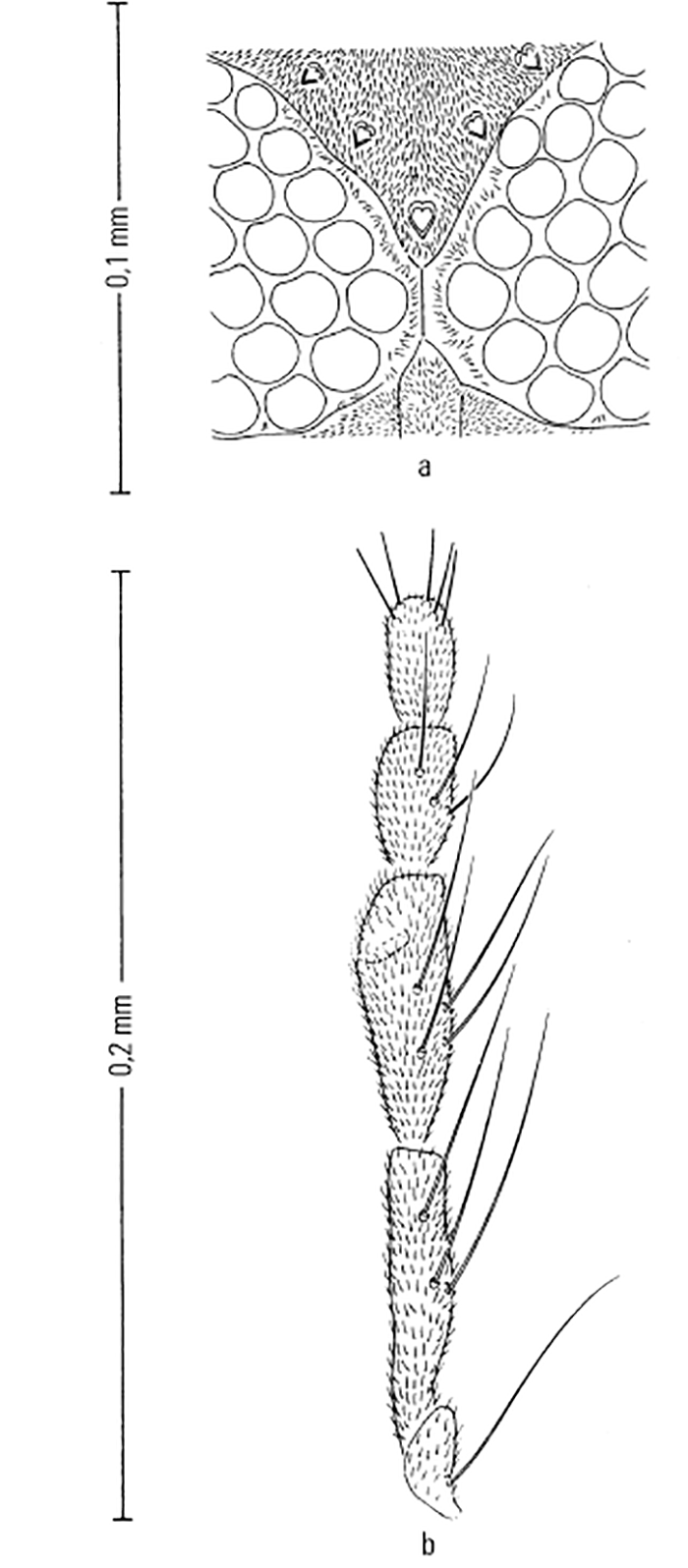
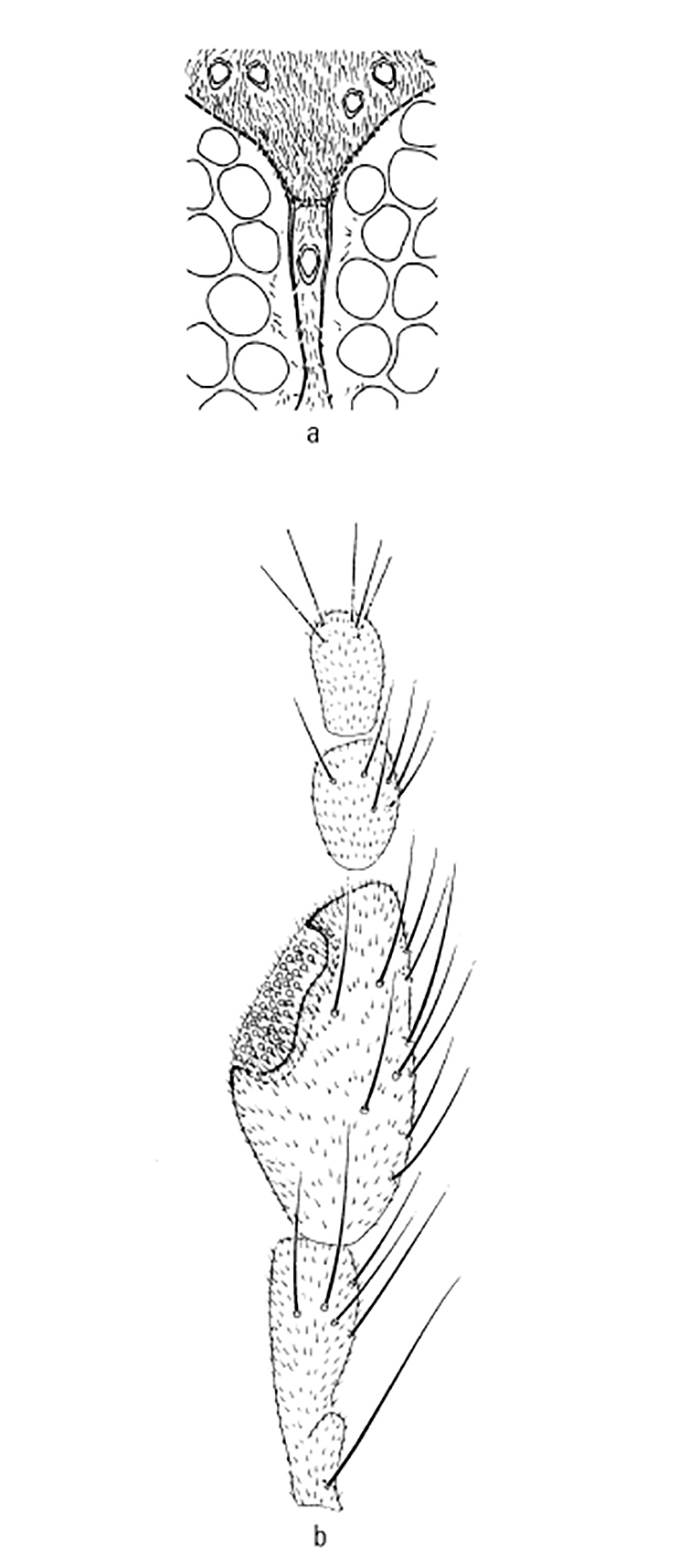
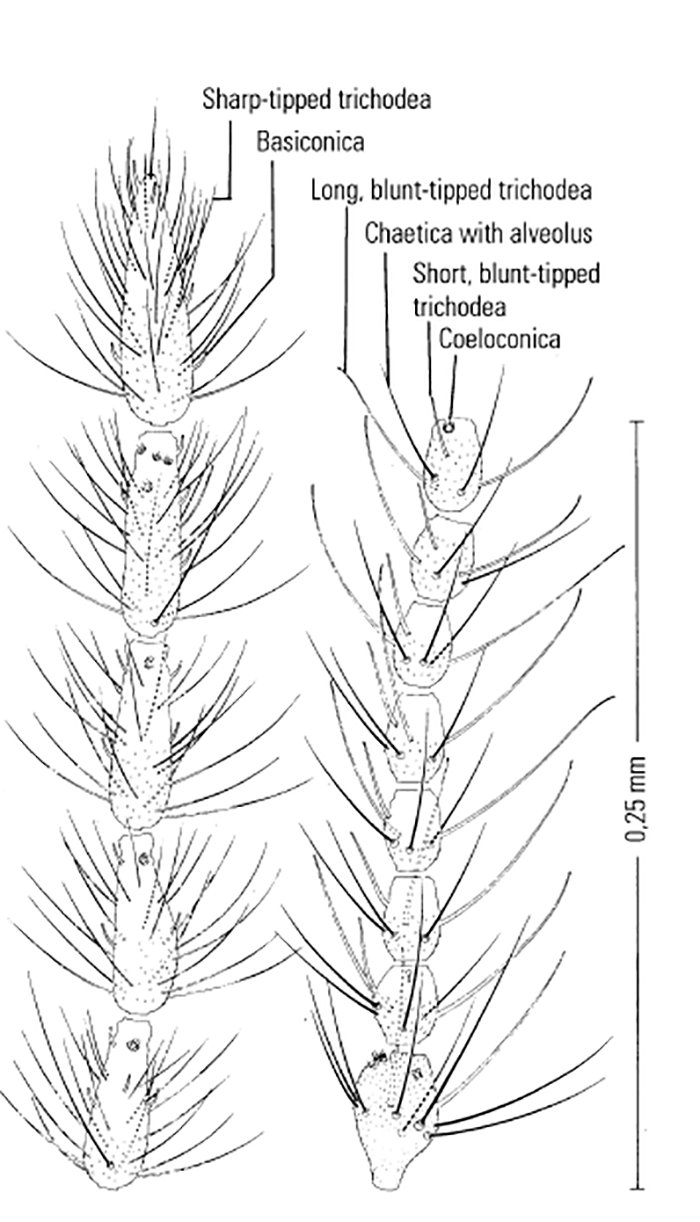
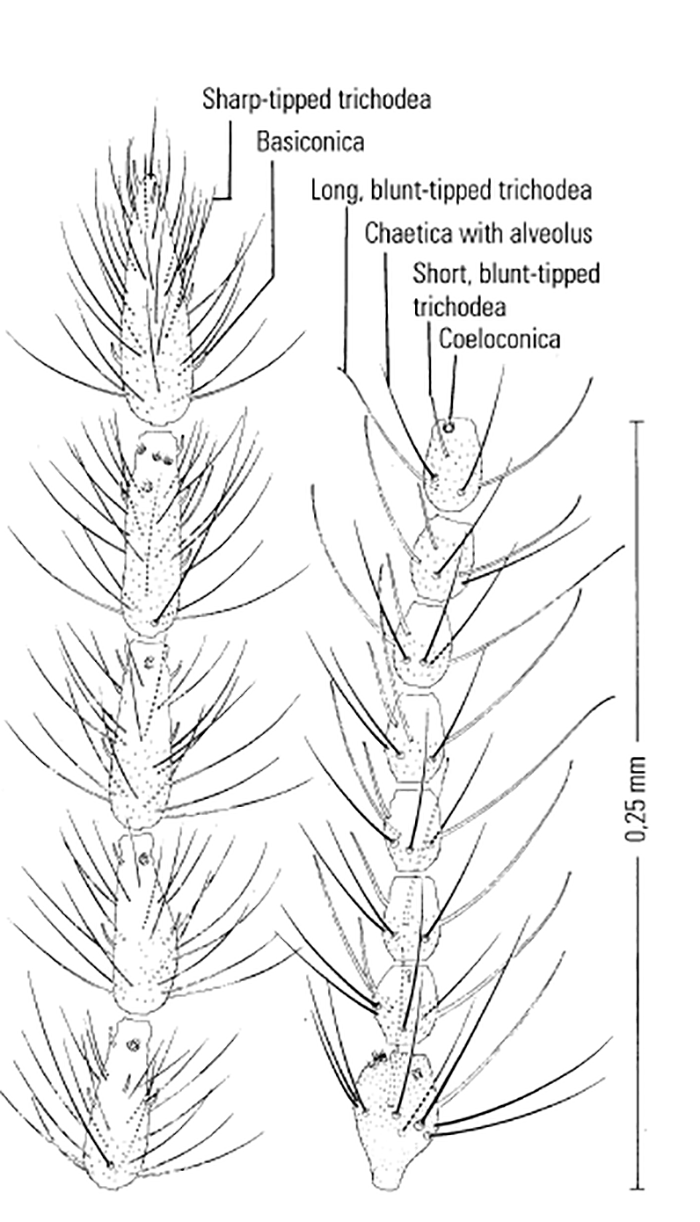
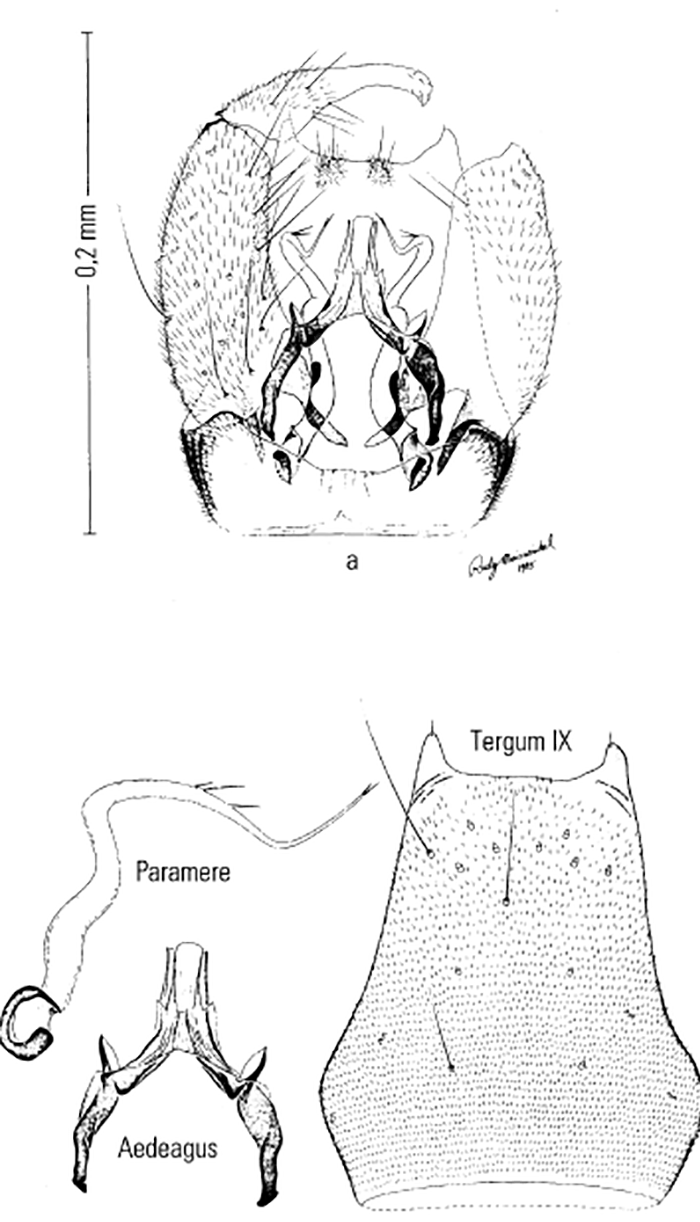
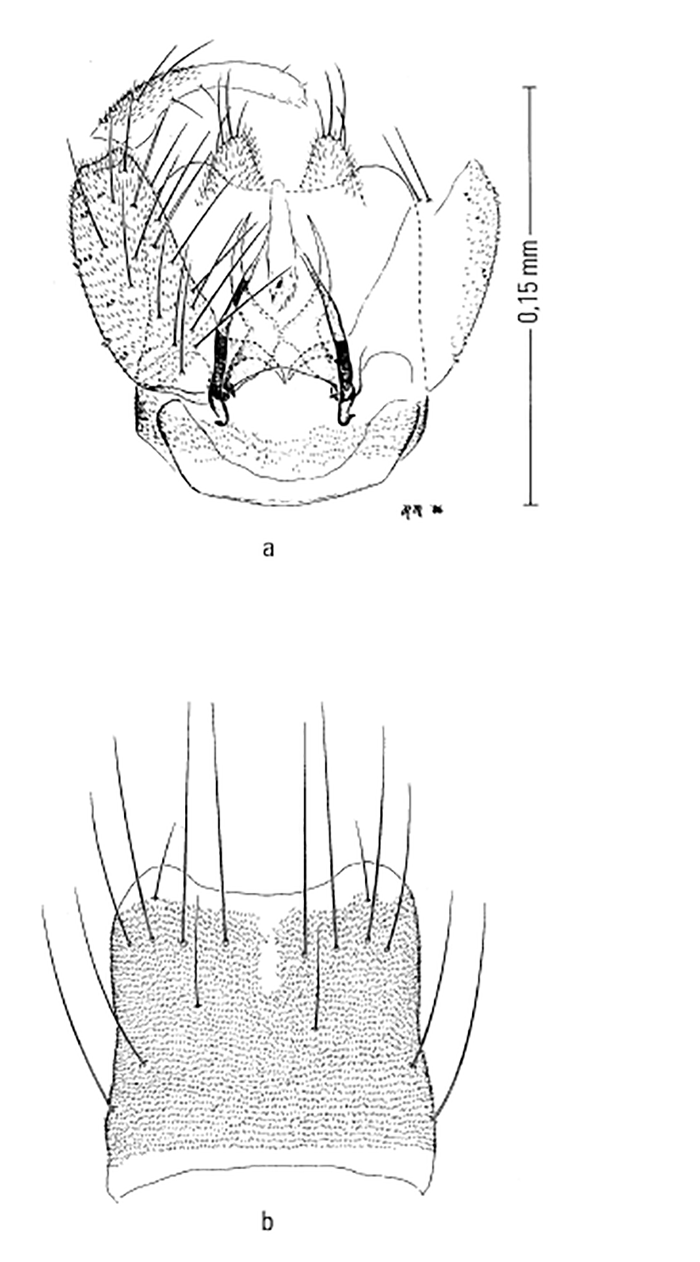
Most Culicoides have a wing pattern that is composed of grey and white spots that is unique to each species. Although this pattern may vary only subtly between species, and so complicates the delineation of intraspecific variation, it remains an extremely useful aid to the identification of the midge. The patterns can be fairly easily observed under a dissecting microscope; those of the 14 species of Culicoides most commonly found associated with livestock in Africa appear in Figure 5.3. As is evident in the figure, the species are quite easily separable on wing pattern but this is because they are representative of six subgenera. It is within subgenera that the patterns become more broadly similar, but what hampers rapid, easy identification is that subgenera can often comprise five or more species complexes. It is within complexes that identifications based on wing patterns alone are unreliable, even in the hands of the most experienced person. Furthermore, 10 per cent of African Culicoides spp., e.g. C. ravus, lack a wing pattern, and for reliable identification such species (indeed for all species in all complexes) must be dissected and mounted on glass slides and examined by light microscopy at 100 to 400 x magnification. In slide-mounted specimens identification of the female Culicoides to species level is based on the precise shape and number of the spermathecae (Figure 5.4),and the shape of the third palpal segment and the distribution of the sensillae on it (Figure 5.5), the conformation of the space between the eyes, and whether the chitinous areas between the ocelli are, or are not, adorned with hairs (Figure 5.6). Perhaps the most useful taxonomic aid for the identification of females to species level is the precise number and arrangement of each of the seven types of sensillae on the antenna (Figure 5.7), but unfortunately, in the majority of taxonomic studies published on Culicoides worldwide, these arrangements are not completely enumerated or described. This arrangement and number of sensillae on the antenna are also very useful for identification of the males to a species level but they are also a neglected source of taxonomic information (Figure 5.8). The shapes of the various parts of the male genitalia are, in addition, highly species-specific and are always used in identification (Figures 5.9 and 5.10). Unfortunately the preparation of such material on glass slides for taxonomic study requires extensive dissection, which is both laborious and time-consuming but is essential if the small differences that exist between species are to be discovered and used in descriptions. Whilst the more modern approaches, such as mtDNA extraction and RAPD-PCR, have recently been employed to separate species of the Imicola complex,290 it is not foreseen that they will ultimately replace the cheaper and more rapid morphological method of identification. Rather, they will be harnessed to solve the more elusive issues surrounding the identification of particular species within specific vector complexes.
Taxonomy
Historical perspective and problems
Nearly 100 years have passed since the first Culicoides was described from Africa and still no regional monograph has appeared that treats the Afrotropical fauna as a whole. There are, however, a number of taxonomic publications from South Africa,74, 131 Angola,62 Kenya142, 181 and Nigeria.37 There are also landmark studies that deal with significant parts of the fauna such as those by Carter, Ingram and Macfie,65 Clastrier, 71, 73 Clastrier and Wirth,74 Cornet,80 Cornet and Chateau, 82 Cornet and Brunhes,81 Cornet and Nevill,83 Cornet, Nevill and Walker,84 de Meillon,100, 107 Itoua and Cornet,171 Meiswinkel and Dyce,224 Meiswinkel,213–215, 217 and Nevill and Dyce.269
The taxonomy of South African Culicoides for the larval, pupal, and adult male and female stages is shown in the diagram in (Figure 5.11). Only where the relevant areas are coloured in the diagram has the particular life stage been described and published.

Figure 5.3 A wing of each of the 14 species of Culicoides most commonly found associated with livestock in southern Africa
a = C. imicola (Zimb. 5)
b = C. bolitinos, (Afr. OP32)
c = C. zuluensis (OP69)
d = C. magnus (Zimb. 11)
e = C. gulbenkiani (Afr. OP37)
f = C. fulvithorax (Zimb. 8)
g = C. similis (Afr. OP34)
h = C. tropicalis (S.Afr. 6)
i = C. neavei (OP17)
j = C. schultzei (Afr. OP27)
k = C. engubandei (Zimb. 14)
l = C. bedfordi (S.Afr. 21)
m= C. pycnostictus (Afr. 9)
n = C. leucostictus (S.Afr. 10)
Yet undescribed species are numbered in the diagram; they comprise a third of the 112 species collected over the last 20 years in South Africa. The diagram shows too that the immature stages of at least 70 per cent of the species still await discovery and description. Annually, two or three species are added to this list,216 and if this is representative of the situation in the rest of Africa, it illustrates the amount of taxonomic research still to be done on Afrotropical Culicoides.
The first Afrotropical species described were C. schultzei (Figure 5.3) and C. herero from Namibia in 1908;122 by 1925 nearly a third of the 156 Afrotropical species recognised today had been described. An unavoidable result of the disparate collecting by early French, German and English workers is that the holotypes of many of their species are today either lost or are scattered amongst a number of European museums or research institutions; of the holotypes that are extant most are in a poor state of preservation. Exacerbating matters for practising systematists today is that most early descriptions were too brief, were based on a single specimen (usually female), and were often not accompanied by illustrations. This means that many character traits, crucial to identification and assignation of a species to the correct subgenus or species complex, are mostly lacking from these early descriptions.
One of the most prolific students of African Culicoides was the French abbé, J.J. Kieffer, who described 20 species between 1911 and 1921 from diverse localities that included the Seychelles, Kenya, the Cameroon (most species), Sudan and South Africa. To a great extent the research of Kieffer182– 185 has remained esoteric. For example, 14 of the 20 Culicoides names made available by him are not in use today even though the species he described are probably common. His ‘floating’ species are albosparsus, bisignatus, dentatus, guineensis, kribiensis, nilogenus, nilophilus, octosignatus, quadrisignatus, remotus, signatus, silvestrii, trisignatus and xanthogaster. Indeed, the names of a further three species, tropicalis, imicola and alticola, had suffered a similar fate, but were resurrected when their respective holotypes were discovered in the Paris Museum 60 years later.191
Contemporaneously (1919 to 1925, 1947), 21 species of Culicoides were described by Carter,64 Carter, Ingram and Macfie,65 Ingram and Macfie,167–169 and Macfie.207, 208 Most of their material emanated from Ghana but also included new species from the Sudan and Malawi. The taxonomic studies of Carter, Ingram and Macfie were of a high quality and, importantly, included the first descriptions of Culicoides larval and pupal stages, and their habitats. Influenced by Ingram, De Meillon (1936 and 1937)100, 101 commenced the first biological studies on South African Culicoides that culminated in the description of new species and their pupae, and the first observations on breeding sites.
Unlike the situation with Kieffer, many of the Carter, Ingram and Macfie species names, such as similis, accraensis, brucei, pycnostictus, punctithorax, nigeriae and bedfordi, are in use today. However, it must be noted that, like some of those of Kieffer, a number of their species are also both rarely recorded and poorly understood. These include adersi, austeni, ochrothorax, eriodendroni, inornatipennis, nigripennis, citroneus, clarkei, confusus, rutilus, arenarius, corsoni and lamborni. One explanation is that the countries from which most species were originally described have since only been rarely sampled for Culicoides. More cogent is that these authors collected mostly in the high-forested tropics, an area harbouring only a particular subset of the Afrotropical Culicoides fauna, and one that by all accounts appears to be relatively depauperate. It is pertinent that Kieffer and Carter, and Ingram and Macfie described most of their species from the equatorial coastal towns of Kribi (Cameroons) and Accra (Ghana) respectively. It is thus likely that these authors, who seldom referred to each other’s work, have to some extent described the same species.
An obvious and significant example is C. pallidipennis, which was subsequently shown to be a synonym of C. imicola Kieffer, 1913.
Besides the possibility of further synonyms the most vexing taxonomic problem to remain is that most of the species mentioned above represent species complexes, the most notable being the Accraensis, Grahamii and Nigripennis complexes. Each complex is represented by 10 or more species that are rarely collected and are very difficult to identify. One consequence is that precise delineation of the nominate species of each complex eludes us to this day, which means that the description of new members of these complexes are either held in abeyance or are being published as C. sp. # A, B, etc.37 or are given numbers.142, 223 Ultimately these systematic problems require resolution as each species within a complex fills a niche that should be defined as a first step towards establishing whether it includes the transmission of pathogens harmful to livestock. The range of niches is almost infinite and includes differences in geographic distribution, seasonal prevalence and abundance, host preferences and vector capacity.
Evolutionary radiation into a particular subset of ecological conditions is a hallmark of species comprising the recently discovered Imicola complex, and because this complex contains the most important transmitters of orbiviruses known today in Africa and beyond, it is discussed below in greater detail with special emphasis on the two vector species C. imicola and C. bolitinos.
Vector species
Of the more than 1 210 species of Culicoides that have been described, about 70 belong in the subgenus Avaritia. At least 30 species of this subgenus occur in Africa; of these nearly half remain undescribed and so would bring the world total to 85. Worldwide a mere 10 species of Culicoides are involved in the transmission of orbiviral diseases that affect livestock; fully half of these belong in Avaritia. The most important vectors in Africa are Avaritia spp., namely C. imicola and C. bolitinos. Both transmit BT and AHS (Table 5.2),115 and are the suspected vectors of EE, but less is known about their possible involvement in the transmission of Akabane and BEF.
Bluetongue virus has also been isolated from C. gulbenkiani, another species of Avaritia, which, like C. bolitinos, breeds in the dung of cattle and is therefore closely associated with livestock. However, it probably plays only a minor role as it seldom becomes abundant and is restricted in its geographic distribution.
In addition to the three livestock-associated species mentioned above, there are seven species of Avaritia particularly closely associated with larger African herbivores such as the elephant, both rhinoceros species (Ceratotherium simum and Diceros bicornis), buffalo, blue wildebeest and the plains zebra (Equus burchellii). Not only do these Avaritia spp. feed on these hosts but also use their dung as a larval habitat.41, 118, 212, 213, 223, 261, 267 In the case of C. bolitinos, it has broadened its resource range to now use not only buffalo but also cattle dung as a breeding medium. This has enabled it to maintain a widespread prevalence in Africa, which is in contrast to the elephant-associated C. loxodontis, which remains restricted in its distribution presumably due to its inability to invade other dung types.
With a burgeoning game farming industry in southern Africa, the translocation of wild herbivores can be accompanied by the establishment of pathogens to livestock in hitherto ‘clean’ farming areas. For example, zebra exported from Namibia were indubitably linked to the outbreaks of AHS that occurred in Spain between 1987 and 1991, in which several hundred horses died or were destroyed as a result.288 In this instance C. imicola played the principal vectorial role.233 Subsequently, it was confirmed in South Africa that cycles of AHSV occur in zebra and that this can proceed in the driest of environments when Culicoides populations are depressed.13, 217 There is also evidence that where new populations of wildlife are established on game farms, the associated Culicoides spp. will also become established in the long term, and in this way new vector foci are created.216
It may not be circumspect to single out species of the subgenus Avaritia as the sole vectors of orbiviruses in Africa as potential candidates from other subgenera have been discussed and listed in the literature.330 As noted above there have been sporadic isolations of viruses from a number of other species (Table 5.2). In addition, in laboratory infection studies,335 evidence has emerged that more than two species of Culicoides may be capable of replicating BTV; this is the first indication that there may be other vectors of greater or lesser importance in the field. One of the strongest candidates is C. magnus, which is a member of the subgenus Culicoides and which can at times become quite abundant in the cooler central and southern regions of South Africa. It has been recorded widely from the continent, and is closely related to the European Pulicaris complex, which is also suspected to play a role in the transmission of orbiviruses.233
Below is summarized what is known about C. imicola and C. bolitinos as regards their taxonomy, geographic distribution and seasonality, vectorial competence, host preferences, gonotrophic cycle, dispersal, and association with outbreaks of disease. In (Table 5.3) the broadscale larval habitat differences between these two vector species are compared, and the resultant impact on adult distribution, prevalence and abundance indicated.
Culicoides imicola Kieffer, 1913 (Figure 5.3a)
Taxonomy
As a taxon, C. imicola is most closely related to C. nudipalpis Delfinado, 1961, the latter known only from south-east Asian islands east of the Wallace line. Culicoides imicola is a member of the Imicola complex, which comprises 10 species, all restricted to the Old World; eight of these species occur in Africa. Detailed studies have done much towards clarifying their taxonomy, biology and distribution. 213–215, 220 The complex was reviewed in toto in 1995.217 The taxonomy of C. imicola in the Mediterranean region was comprehensively reviewed in 1991 by Boorman, 36 who noted only one difference between Mediterranean and South African populations.
This difference was due to an error that appeared in a publication of Meiswinkel,213 and so confirms that C. imicola sensu stricto is morphologically homogeneous across its large geographic range; this homogenicity being supported subsequently.220 The independent species status of the various Imicola complex taxa sensu Meiswinkel217 was recently validated by RAPD–PCR analyses;289 DNA (RAPD) patterns for seven species of the complex are shown in (Figure 5.12.) In (Figure 5.13) an unrooted neighbour-joining consensus tree shows the relationship and discrete clustering of 19 populations of these seven species collected in South Africa, Botswana, Madagascar and the Ivory Coast. Until 1972 studies on the taxonomy and biology of C. imicola sensu stricto appeared under a variety of names that include C. pallidipennis, C. iraqensis, C. minutus and C. pseudoturgidus.
Vector competence
Incriminated in 1944115 as a vector for BT and AHS, C. imicola is still regarded as the most important Old World vector of these livestock diseases. It is also considered a potential vector for EE.328 Depending on the environmental temperature, the extrinsic incubation period of BTV in C. imicola is 7 to 12 days.50, 115 More detailed information on serotype-specific infection rates and virogenesis is given above in the section on ‘Vector competence’.
Host preferences
Culicoides imicola has been found to feed on cattle, sheep, horses, pigs, goats and poultry.42, 45, 52, 57, 163, 265, 267, 338, 339 In Israel it is inclined to bite cattle and horses along the backline; in cattle it prefers feeding in the darker coloured areas of the animal.43 There are indications that C. imicola is somewhat exophilic in its feeding habits showing some reluctance to enter buildings221 but these require validation in situations where very large populations abound. As regards wildlife, eight females have been collected off a darted African buffalo.213
Gonotrophic cycle
It has been estimated338 that, with a gonotrophic cycle of four days, C. imicola females might take five blood meals during their lifetimes; it was similarly calculated subsequently50 that the mean period between blood meals was 3,3 to 4,6 days. In Israel49 C. imicola was found to complete 11 generations per annum with each generation being approximately 23 days in duration. It is generally accepted that under warmer environmental conditions the life cycle is shortened, and thus there is a greater number of generations and adults produced per annum.
Larval habitat
Using emergence traps, hundreds of C. imicola were first reared at the Onderstepoort Veterinary Institute in 1950 by du Toit and Fiedler260 in a swampy kikuyu grass (Pennisetum clandistenum)-covered area next to a leaking cement dam. Nevill260 was the first to show that the pupae are susceptible to drowning as they are unable to float on the surface of water as do many Culicoides spp. pupae.
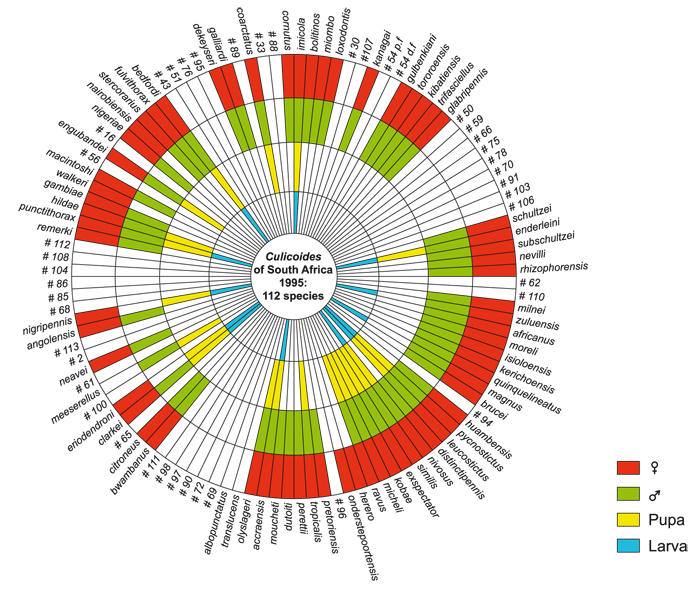
Figure 5.11 A diagram of the taxonomy of South African Culicoides for the larval, pupal, and adult male and female stages
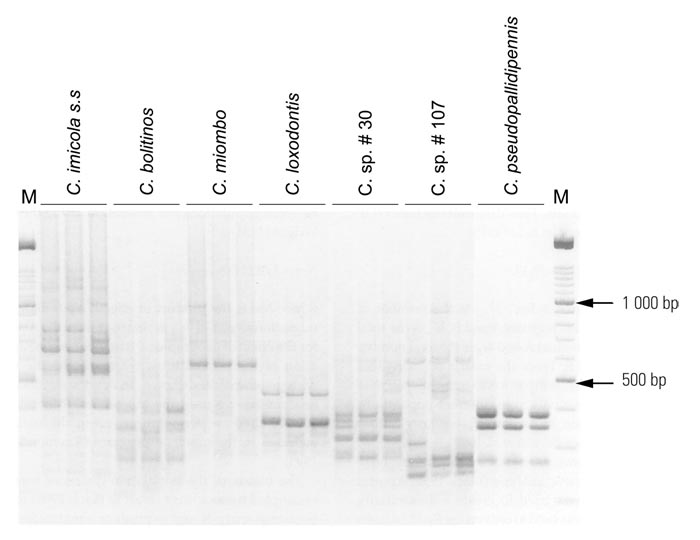
Figure 5.12 Representative individual random amplified polymorphic DNA (RAPD) patterns for the seven morphological species of the Culicoides imicola complex, using the primer D17: C. imicola ss from Onderstepoort, C. bolitinos from Rhinocerosfontein, C. miombo from Maun, C. loxodontis from Phalaborwa, C. sp. #30 from Port Edward, C. sp. #107 from Mabula Lodge, and C. pseudopallidipennis from Abokouamekro, Ivory Coast
Table 5.3 Broad-scale larval habitat differences between Culicoides imicola and C. bolitinos, and resultant impact on adult distribution, prevalence and abundance216
| CULICOIDES IMICOLA | CULICOIDES BOLITINOS |
|---|---|
| IMMATURE STAGES
ADULTS
| IMMATURE STAGES
ADULTS
|
The requirement for a semi-moist larval habitat by C. imicola appears to be a characteristic shared by all species of the subgenus Avaritia and, for C. imicola, was partly confirmed in Israel47 where it was reared in rich mixtures of organic matter and water-saturated soils, but despite this it was also the species adapted to the driest breeding conditions. The breeding habitat of C. imicola in Israel has been summarized as being ‘in and around animal pens in water trough overflow, at the margins of animal sewage and drainage canals and puddles created by leakage from water pipes.’33 In Kenya340 high concentrations of larvae were found in mud and dung mixtures surrounding cattle pens and in associated effluent ditches. In Cyprus236 C. imicola was found breeding where there were leaks from irrigation pipes. These were small seepages with little free surface water. They were generally drier than the type of habitat preferred by other species of Culicoides and were usually covered by a growth of fresh grass. There was almost no contamination by animal droppings, but organic matter was present in the soil in the form of decaying vegetation. In Laos163 it was reared in mud mixed with dung. In an unpublished study conducted at the Onderstepoort Veterinary Institute up to 382 C. imicola/m2 were reared in islands of short kikuyu grass in the middle of effluent-enriched drainage canals.275
They were, in addition, reared throughout the year with their numbers peaking in late summer and autumn precisely when the numbers of adult C. imicola are at their highest, and when diseases such as BT and AHS are most prevalent. In another study267 low numbers of C. imicola were reared in dry as well as in wet kikuyu paddocks especially where the soil was clay-like and medium moist. Midges were also reared in continuously moist areas where the substrate consisted of sandy soil covered by vegetation and a layer of organic matter, and where the moist but silty soil of irrigated pastures had very little surface organic matter except for some rotting plant particles and/or diluted manure. It is now fairly well established that C. imicola is most abundantly encountered where the soils are clayey, such as the black cotton soils at the Onderstepoort Veterinary Institute and those of the Laikipia plateau and the Masai Mara in Kenya, but it is virtually absent where the soils are overly sandy.222 It is postulated that sandy soils, in draining too quickly of moisture and being nutrient poor (especially those found along the coast), are simply unable to sustain the larval stages of C. imicola for the required 7- to 10-day developmental period.218, 219 The proposed relationships between soil type, prevalence and abundance of C. imicola, and the prevalence of AHS during the 1996 South African outbreak of the disease is illustrated graphically in (Figure 5.14). It is probable that the rearing records74, 161 of C. imicola, documented under the name C. pallidipennis, from rotting banana and plantain stems do not refer to C. imicola.
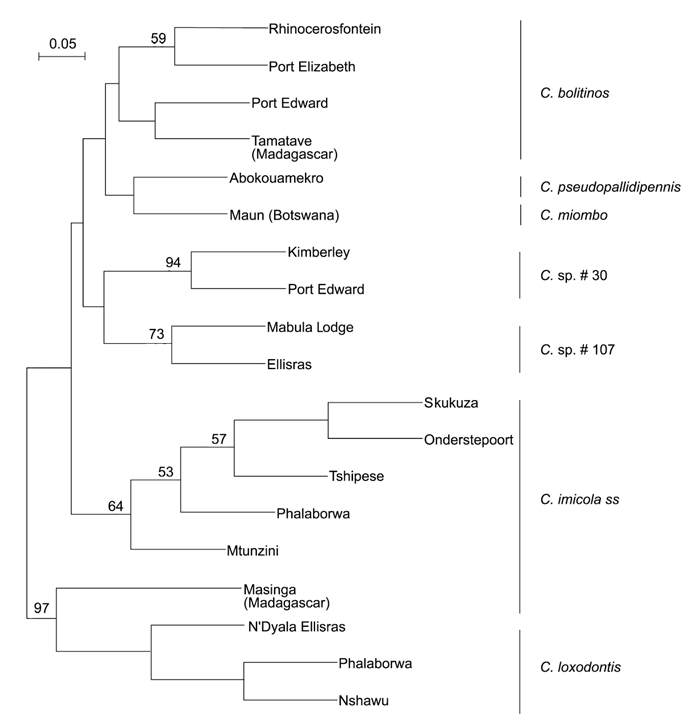
Figure 5.13 Unrooted neighbour-joining (NJ) consensus tree of 19 populations of the seven morphological species of the Culicoides imicola complex, based on the dissimilarity values inferred from 196 RAPD fragments, amplified using four arbitrary primers. Only bootstrap values greater than 50 per cent obtained from 100 bootstrap resamplings of the original dataset are shown
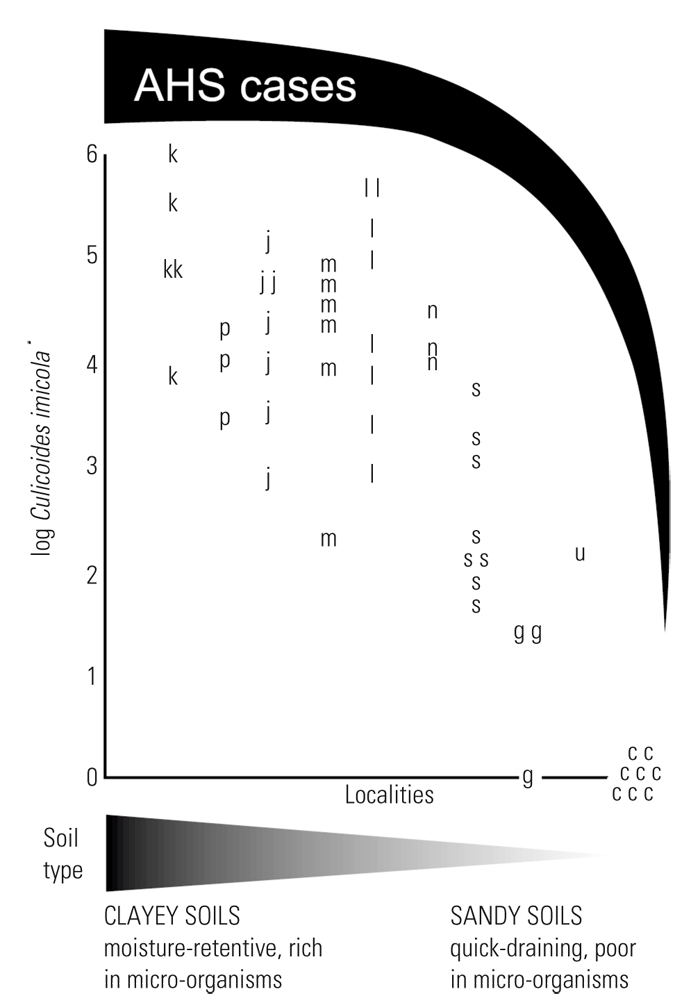
Figure 5.14 A schematic representation of the proposed relationships between soil type, numbers of Culicoides imicola and the number of cases of AHS during the 1996 outbreak in South Africa based on 52 insect collections at 47 sites. Sites were allocated to ten region-groups which, moving from left to right, show a decrease in AHS cases and an increase in soil sand content. Region-groups are: Kaalplaas farm, Onderstepoort Veterinary Institute (k); Pretoria (p); Johannesburg (j); south-central Mpumalanga (m); eastern Mpumalanga lowveld (l); KwaZulu-Natal (n); Free State and northern Eastern Cape (s); Graaff-Reinet area of Eastern Cape (g); Uitenhage, 30 km inland from the southern coast (u); and south-eastern Cape coast (c)
Immature stages
Contrary to a published statement142 and the questioning of their possible identity,36 the descriptions260, 262 of the larva and pupa of C. pallidipennis are indeed those of C. imicola and not of C. bolitinos. A study of the pupae of five taxa of the Imicola complex has revealed that they differ markedly in morphology from one another.268
Geographic distribution
Culicoides imicola was originally described from a single female specimen collected at Tiwi on the southern coast of Kenya. Today, C. imicola is known to be one of the most widely distributed Culicoides in the world and is found in at least 35 countries throughout Africa, in most countries surrounding the Mediterranean Sea, and in the Near, Middle and Far East as far as southern China, Laos and Vietnam. Only in South Africa, Morocco and the Iberian Peninsula has its distribution been mapped in some detail.274, 286, 332
Seasonal abundance and prevalence
Only in South Africa, Morocco and Spain has the abundance and prevalence of C. imicola been studied and mapped on a national scale.20, 23, 332, 333 On a smaller, but more intensive scale are the area-based studies in South Africa, of the Western Cape Province,267 eastern Free State Province,330 the Kruger National Park and adjoining areas,217 the greater Onderstepoort area in Gauteng Province331 and the Port Elizabeth area in the Eastern Cape Province,218 and in Lesotho.336 In addition, its abundance and prevalence were detailed during an outbreak of AHS in the central and eastern regions of South Africa.219 These studies all took into account the existence of sibling species and in this way do not conflate data from various Imicola complex taxa. They also demonstrate the extreme variability in the prevalence and abundance of C. imicola in the vicinity of livestock ranging from it being totally absent218 to widespread and superabundant.219 In each study an attempt was made to explain this variation in terms of climatic or edaphic factors. It is emerging from these studies that other than extreme cold and aridity, the degree of topographic slope (inducing water run-off), soil type (whether drainage is slow or rapid) and soil fertility (availability of micro-organisms) are additional important factors affecting the distribution and abundance of this species.
As to its detailed seasonal prevalence at various sites throughout South Africa reference should be made to two studies332, 333 whose data were used in conjunction with various climate variables and satellite imagery, to model the prevalence and abundance of C. imicola.21 Whilst the model explained 67 per cent of the observed variances it also shows some anomalies which are probably the result of too heavy reliance on the effects of rain and temperature. This was further demonstrated in an unpublished preliminary African model in which C. imicola was predicted to be most abundantly encountered in equatorial Africa, i.e. precisely where it is known that C. imicola is virtually absent.171, 214
In a separate section (see below) the more precise seasonal and geographic distribution of C. imicola and of C. bolitinos, as revealed by weekly collections made over a full year at 40 sites spread throughout South Africa, are discussed in greater detail, and the various implications of the findings assessed. At the periphery of its range in the Middle and the Far East the distribution and abundance of C. imicola are mostly unknown.
Dispersal
Under normal conditions the flight range of C. imicola probably varies in the range of a few hundred metres to a few kilometres. Clearly this is dependent upon the local availability of hosts for feeding and larval substrates for breeding. It is generally accepted that Culicoides are reluctant to fly if the wind is above a certain speed, which is when numbers caught in light traps diminish, but it is apparent too that light traps become increasingly inefficient as windspeeds increase and do not then accurately reflect biting midge activity rates.221, 319 There is compelling evidence that C. imicola can be transported upon prevailing winds for hundreds of kilometres. This is considered the most likely way in which outbreaks of BT are being precipitated in countries around the Mediterranean Sea,290 but it is difficult to prove as the clandestine movement of infected animals also merits consideration. Similarly, can the occurrence of C. imicola on islands off the African east and west coasts (Madagascar, Reunion, Mauritius and the Cape Verde) be explained by wind transport or were they moved together with livestock? Nevertheless, the evidence to unequivocally support wind dispersal remains largely unsubstantiated as the capture of large numbers of Culicoides at various altitudes over sea or land has yet to be made. Another, as yet unsubstantiated, mode of dispersal for C. imicola, is the postulation that Culicoides may be moved over long distances in enclosed horse transport floats.219
Association with outbreaks of disease
Since Du Toit’s incrimination of C. imicola as a vector of both BT and AHS in 1944, it is remarkable how few attempts have been made to isolate one or the other virus from this species during field outbreaks of these diseases. The various isolations made from C. imicola (Table 5.2) were invariably the result of random surveys for Culicoides-associated orbiviruses and were often not linked to an outbreak of a disease. A number of these isolations cannot be linked to a specific species but emanated rather from mixed Culicoides spp. pools. As far as can be established, the first isolations of AHSV from identified pools of C. imicola not containing freshly bloodengorged females were made during the 1987 to 1991 outbreak of the disease in Spain.233 Four serotypes of BTV were isolated from C. imicola in Israel44 before these dates. In Africa it is equally remarkable to note that only in 1996 was the first investigation made to establish which species of Culicoides (Table 5.4) were most abundant at stables in which losses during an extensive outbreak of AHS were occurring. 219 It was clear that C. imicola was the most abundant and prevalent species and thus, for the first time in Africa, it was positively linked geographically and temporally to an outbreak of AHS. Even in this investigation no attempt was made to isolate virus from identified pools as the age-grading and selection of non-engorged females from the millions that were captured were just too overwhelming. More recently, a single isolate of AHSV serotype 7 was made from pools of C. imicola collected during the 1999 outbreak of the disease in the Western Cape Province.189 A recent analysis of the 14 largest epidemics of AHS to affect southern Africa since 1719 were shown in 13 instances to be linked to warmphase El Niño weather events.22 These usually bring above average precipitation and it is this factor that strongly points to C. imicola having been principally involved in past epidemics of the disease. As shown in (Table 5.4) C. imicola was the only species of Culicoides found to increase explosively in numbers during, and following, the heavy rains of 1996. This pattern of outbreaks and the occurrence of AHS as a plague following excessive rains was noted as long ago as 1863278 and was subsequently corroborated in 1921.320
Table 5.4 Prevalence and ranked abundance of the 13 Culicoides species most commonly encountered in 66 light trap collections made at 47 sites during the 1996 outbreak of African horse sickness in South Africa.219 The last column is the abundance ranking of these species according to an earlier study332
| ABUNDANCE RANK219 | CULICOIDES SP. | % OF ALL CULICOIDES | PREVALENCE IN 66 LIGHT TRAPS | ABUNDANCE RANK 332 |
|---|---|---|---|---|
| 1 | C. imicola | 94,2 | 60 | 1 |
| 2 | C. zuluensis | 3,0 | 44 | 5 |
| 3 | C. magnus | 0,7 | 25 | 10 |
| 4 | C. bolitinos | 0,6 | 34 | 15 |
| 5 | C. leucostictus | 0,4 | 33 | 7 |
| 6 | C. pycnostictus | 0,1 | 28 | 6 |
| 7 | C. nivosus | < 0,1 | 15 | 8 |
| 8 | C. milnei | < 0,1 | 13 | 17 |
| 9 | C. neavei | < 0,1 | 11 | 16 |
| 10 | C. enderleini | < 0,1 | 10 | 2 |
| 11 | C. subschultzei | < 0,1 | 9 | 2 |
| 12 | C. onderstepoortensis | < 0,1 | 9 | 20 |
| 13 | C. gulbenkiani | < 0,1 | 9 | 11 |
Table 5.5 Summary of blood meal identifications for Culicoides species in South Africa and Zimbabwe
| CULICOIDES SP. | CATTLE Ref. 57, 225, 265, 267 | SHEEP Ref. 57, 225, 265, 267 | PIGS Ref. 51, 57, 265, 267 | HORSES Ref. 51, 57, 265, 267 | CATTLE/ SHEEP Ref. 57, 225, 265, 267 | BOVIDS Ref. 51 | HORSES/ CATTLE Ref. 57 | HORSES/ SHEEP Ref. 57 | HORSES/ SHEEP/ CATTLE Ref. 57 | MAMMALS Ref. 51 | AVIANS/ CHICKENS Ref. 51, 57, 225, 265, 267 | TOTAL |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| C. imicola | 247 | 459 | 270 | 371 | 111 | 11 | 1 | 16 | 126 | 9 | 1 621 | |
| C. milnei | 4 | 7 | 120 | 2 | 16 | 1 | 18 | 273 | 441 | |||
| C. sp. near glabripennis | 16 | 139 | 58 | 77 | 8 | 38 | 336 | |||||
| C. fulvithorax | 28 | 167 | 5 | 5 | 205 | |||||||
| C. zuluensis | 11 | 15 | 34 | 74 | 61 | 61 | ||||||
| C. leucostictus | 40 | |||||||||||
| C. bedfordi | 40 | 40 | ||||||||||
| C. magnus | 17 | 14 | 6 | 1 | 1 | 39 | ||||||
| C. brucei | 1 | 2 | 20 | 4 | 3 | 1 | 31 | |||||
| C. pycnostictus | 1 | 1 | 2 | 2 | 16 | 22 | ||||||
| C. gulbenkiani | 10 | 5 | 2 | 2 | 2 | 21 | ||||||
| C. bolitinos | 10 | 5 | 5 | 1 | 21 | |||||||
| Schultzei group | 16 | 2 | 1 | 19 | ||||||||
| C. coarctatus | 1 | 1 | 2 | 4 | ||||||||
| C. ravus | 2 | 2 | ||||||||||
| C. nivosus | 1 | 1 | 1 | 1 | 3 | |||||||
| C. engubandei | 1 | 1 | 2 | |||||||||
| C. neavei | 1 | 1 | 2 | |||||||||
| C. similis | 1 | 1 | 2 | |||||||||
| C. enderleini | 1 | 1 | ||||||||||
| Total | 332 | 687 | 428 | 828 | 116 | 42 | 2 | 16 | 1 | 199 | 402 | 3 053 |
Culicoides bolitinos Meiswinkel, 1989 (Figure 5.3b)
Taxonomy
Culicoides bolitinos was only recognized as a species separate from C. imicola in 1989 when the type series was reared from African buffalo dung in the Kruger National Park, South Africa. It is most closely related taxonomically to the Oriental/Australasian C. brevitarsis Kieffer, 1917, which is regarded as the most important vector of BTV in Australia. In the past C. bolitinos has been confused with both C. imicola and C. brevitarsis; these misidentifications have been corrected.213 Before 1989 C. bolitinos was recorded in the South African literature as C. sp. # 49. Culicoides bolitinos is a member of the Imicola complex but, although it is morphologically very similar to C. imicola, the two species differ markedly in various aspects of their respective ecologies. These differences are listed in Table 5.3.
Vector competence
Culicoides bolitinos was first incriminated as a vector of AHS in an outbreak of the disease in South Africa in 1998.226 Almost simultaneously it was shown in the laboratory to be a competent replicator of BTV and is therefore also a likely vector of the disease in the field.335 In addition, it is considered to be the likely principal vector of EEV in parts of the Eastern Cape Province, South Africa, where C. imicola is absent.280
Host preferences
Blood meal analyses show that C. bolitinos feeds on cattle, horses and sheep (Table 5.5). It appears to be more endophilic than C. imicola as it can be captured in large numbers in horse stables,221 thus disputing the long-held belief that Culicoides do not enter buildings.
Gonotrophic cycle
This is unknown but is probably very similar to that of C. imicola.
Larval habitat
Culicoides bolitinos was first reared from cattle dung in 1968 but at the time was mistakenly identified as C. pallidipennis (= C. imicola).261 The larval habitat of C. bolitinos was described as dry cattle dung pats with a very hard crust, a dry sponge-like centre, and a moist lower layer about 25 mm thick in direct contact with moist soil underneath. This describes a pat that has aged to some extent. It is probable that C. bolitinos oviposits only in very fresh dung still emitting the necessary volatile attractants, but this supposition remains to be substantiated. It has been speculated that in Australia the closely related C. brevitarsis oviposits in ageing cattle dung over a number of days. Culicoides bolitinos has also been reared from the dung of African buffalo and blue wildebeest.118, 213 On occasion more than 1 000 of these midges can be reared from a single buffalo dung pat.216 The duration of the larval and pupal stages in dung has been estimated to vary between 8 and 30 days.213 In South Africa, buffalo dung can produce C. bolitinos for up to 90 days especially during the cooler months when it does not desiccate as rapidly as during the hot summer months.216
Immature stages
The pupa has been retrieved from cattle dung, and morphological studies have confirmed that it differs from that of C. imicola.268
Geographic distribution
Culicoides bolitinos occurs in African tropical and subtropical regions south of the Sahara Desert. It has been recorded from 11 countries, namely South Africa, Botswana, Zimbabwe, Lesotho, Malawi, Kenya, Nigeria, the Ivory Coast, Gambia, Madagascar, and Mauritius (misidentified as C. brevitarsis). It is likely that it will be found to occur throughout Africa where cattle are kept. In South Africa it is almost as widespread as C. imicola, and has been found in all of the area-intensive Culicoides surveys referred to above under C. imicola. Further details of its distribution, as determined between 1996 and 1997, are given below (see Detailed distribution and seasonality in South Africa). Its occurrence on offshore African islands begs the question as to why C. bolitinos has not also penetrated into the Mediterranean region where cattle are found in abundance, or has it been overlooked being taxonomically very closely related to C. imicola?
Seasonal abundance and prevalence
Only in South Africa has the seasonal abundance and prevalence of C. bolitinos been studied.222 Before 1989 most studies on Culicoides in South Africa failed to recognize the existence of C. bolitinos with the result that the data on these two species are conflated. This is also partially true for another study conducted through much of South Africa in 1992.332 However, the area intensive studies mentioned above under C. imicola, and published between 1988 and 1998, are reliable as regards the information on C. bolitinos.
Dispersal
It is presumed that the dispersal of C. bolitinos is similar to that of C. imicola. Of interest is the record of thousands of females of C. bolitinos captured at dusk in the presence of a slow-moving herd of 60 grazing African buffalo.217 The specimens consisted of nulliparous, parous and gravid females, which implies that the first two categories were accompanying the buffalo in search of a blood meal for the development of their eggs and that the gravid females were seeking fresh dung for oviposition. In this way C. bolitinos may disperse a number of kilometres in a short space of time. The results of an investigation concerning the protective stabling of horses against the attacks of C. bolitinos implied that, in the absence of cattle dung in a 500-m radius of the stable, C. bolitinos dispersed nightly from neighbouring farms over distances greater than 500m to attack the stabled horses.221 The question remains as to how C. bolitinos arrived on the islands of Madagascar and Mauritius. A molecular study comparing a Madagascan population of C. bolitinos with three populations from mainland Africa failed to reveal any degree of genetic differentiation,289 and therefore suggests the invasion of Madagascar by C. bolitinos to be relatively recent. Whether the introduction of the midge occurred at the time cattle were introduced to the island or whether it predated this, cannot be answered.
Association with outbreaks of disease
The recent outbreak of AHS in the eastern Free State Province of South Africa involved C. bolitinos, and is the first substantive proof of its involvement in field outbreaks of disease.226, 227 In this outbreak five isolations of AHSV serotype 6 were made from identified pools of C. bolitinos as were four isolations of an unidentified serotype of BTV. Furthermore, C. bolitinos was the dominant Culicoides captured and was the only species to be found at all affected stables. In the same outbreak C. imicola was rare in numbers, and had a lowered prevalence rate of 71 per cent. Large outbreaks of AHS have in the past been reported from Lesotho, which immediately adjoins the eastern Free State, such as the one in 1953 when an estimated 5 000 horses died. It has been shown that C. imicola is rare in Lesotho, and it can only be assumed that C. bolitinos could have been the species involved. Culicoides bolitinos is also to be the likely vector of EEV in the Eastern Cape Province,280 specifically in the Port Elizabeth area, where it comprises up to 90 per cent of all Culicoides captured.218
Detailed distribution and seasonality of Culicoides imicola and C. bolitinos in South Africa222
From September 1996 to June 1999 the distribution and seasonality of these two vectors was determined by weekly collections made at 40 livestock holdings spread almost equidistantly across South Africa and the results are listed vertically in (Figures 5.15) and (5.16.) The data show that:
- Culicoides imicola is most abundant in the warmer northern and eastern parts of South Africa where it can be captured throughout the year, including winter. Sites include Tshipese, Polokwane (formerly Pietersburg), Phalaborwa, Komatipoort, Lydenburg, Middelburg and Onderstepoort Veterinary Institute. These areas have long been considered endemic and so may act as source areas for the southward spread of Culicoides-borne orbiviral diseases. However, the data also show clearly that C. imicola can be found in some abundance, and also for prolonged periods, in the southern half of the country (e.g. Kimberley, Joubertina, Laingsburg and Porterville); in the past these areas have also experienced devastating outbreaks of disease, those of AHS being especially well documented.19, 120 The figures also reveal that C. imicola can be found in most, but not all, parts of South Africa, which means that there is probably constant cycling of BTV, AHSV and EEV in many areas, and that when circumstances become particularly favourable for C. imicola, outbreaks will occur.
- As regards C. bolitinos the data in (Figure 5.16) reveal it to be an order of magnitude less abundant than C. imicola, and with the most numbers captured in cooler, higher rainfall areas (e.g. Middelburg, Ficksburg, Port Elizabeth and George). This indication that C. bolitinos is a wet- and cool-adapted species must be treated with caution as cattle, not present at all sites sampled, determine primarily the prevalence of C. bolitinos. Nevertheless, it is clear that significant populations of C. bolitinos are encountered in areas somewhat depauperate in C. imicola, a fact that increases the total area at risk to orbivirus transmission in South Africa. As revealed in (Figure 5.16) (Thabazimbi) it is clear that C. bolitinos can also occur abundantly in warmer, drier areas.
- The lowest annual abundances of both C. imicola and C. bolitinos occur in winter and spring (or weeks 22 to 48 of the year) (Figures 5.15 and 5.16). Their abundances increase gradually in the early summer to peak eventually during late summer and autumn (weeks 10 to 20). During this period the number of older, parous midges is also at its greatest, and so increases further the risk of virus transmission. This perfectly mirrors the late seasonal appearance of BT and AHS, which corroborates the maxim that the geographical and seasonal occurrence of an insect vector must correlate with that of the disease.
- It is generally believed that areas experiencing the coldest winters, including snow, are also those with the smallest populations of C. imicola. This holds true for the sites of Giant’s Castle in the Drakensberg, and Elliot but, even here, C. imicola will appear briefly in late summer (weeks 5 to 10). Unexpectedly, it was revealed (Figure5.15) that C. imicola is rarer at, or absent from, three sites which have a far milder, frost-free climate (Port Elizabeth, Struisbaai and Alexander Bay). These sites, situated on the sandy southern and western coastline, demonstrate that within certain limits, climate is secondary in determining the abundance and distribution of C. imicola, and that the availability of a suitable breeding habitat is therefore paramount. Where known, it has been found that Culicoides larvae inhabit the upper layers immediately below the surface of the soil,35 where they require adequate levels of moisture and nutrients to complete their 7- to 14-day developmental period. However, if these upper layers of the soil drain too rapidly of moisture, as in sandy coastal soils, or in sloping topography, the larvae desiccate and die. This means that overly dry soils most effectively control the numbers of C. imicola. Similarly, it is assumed that soils too poor in micro-organisms will also impede larval development. Such poor soils are commonly found in coastal regions as well as in areas where nutrients are leached by higher rainfall. Such barriers do not apply to the replication of C. bolitinos. The fact that it breeds in cattle dung ensures adequate levels of moisture and an abundance of micro-organisms. Thus soil type and quality play no role in determining its prevalence, which, in turn, means that C. bolitinos can invade areas free of C. imicola, but only if cattle are present.
- It has been argued that winds (such as prevail along the coastline) induce high mortality rates in C. imicola;20 this may be another reason to explain its absence along parts of the South African coastline. However, the persistent prevalence of C. bolitinos in an area such as Port Elizabeth, which is known to be the second windiest point in South Africa and where some of the largest captures of C. bolitinos have been made,228 reveals that windiness may only affect midge activity on particular nights, and does not impact upon overall feeding and breeding success. Presumably the same could be said for its sibling species, C. imicola; this therefore points to factors other than windiness inhibiting its establishment in the Port Elizabeth area.
- Of significance is that these data clearly show that both vectors of AHSV occur in some abundance in the AHS control zone of the Western Cape Province of South Africa. This means that the zone is always at risk to incursions by the virus, as was amply demonstrated by the death of some 30 horses from the disease in the Stellenbosch area in 1999.24 The prospect that this can recur at irregular and unpredictable intervals is real. Vector-free points (Figures 5.15 and 5.16) occur along the southern and western coastline of South Africa.
As noted, an earlier data set332 similar to the one above was used to model the distribution and abundance of C. imicola in South Africa.21 Whilst the model satisfactorily explained 67 per cent of the observed variance, significant anomalies appeared as regards the geographic distribution of C. imicola and its predicted abundances. This may be due to the data being representative of climatically ‘average’ years, but which fail to model for the superabundance of C. imicola following excessive rains.219 This would seem essential when one considers that its superabundance at such times correlates perfectly with the statement by Theiler320 ‘that horse sickness appears as a plague following heavy rains’. Interestingly this observation appears to be contradicted by the central clumping of abundance data in Figure 5.15 where it is clear that the largest numbers of C. imicola appear in those areas experiencing a more moderate annual average rainfall varying between 300 and 700 mm per annum. It remains to be demonstrated whether this apparent contradiction is explicable in terms of soil fertility, i.e. in areas experiencing a persistent annual average rainfall of above 800 mm, do the soils become too leached of nutrients to sustain the larvae of C. imicola? If so this may explain the virtual absence of C. imicola in the equatorial regions of Africa where suitably high environmental temperatures prevail, but where rainfall is excessive and soil fertility is notoriously poor. Another factor difficult to incorporate into any model is the extent to which humans aid the establishment of large populations of C. imicola by maintaining livestock in confined spaces and by irrigating pastures. Whilst a similar model has yet to be developed for C. bolitinos it would require the inclusion of other parameters such as the presence or absence of cattle.
Host preferences
Understandably, nearly all studies on African Culicoides are strongly biased towards the welfare of exotic breeds of horses, cattle and sheep. As several of the orbiviral diseases affecting domesticated livestock have their origin in African wildlife, it would seem appropriate to study the relationship between African Culicoides and game. As has been reviewed, 223 nine studies in Africa have purported to deal with this relationship but nearly all were conducted in unnatural systems in which wildlife had been removed from their habitats and maintained in subverted conditions. The relationship between some species of Culicoides and the African elephant, zebra and buffalo are briefly reviewed below. These herbivores serve as hosts for three species of the Imicola complex, and for other species of Avaritia whose role as vectors remains unknown.
Culicoides and the African elephant
There is only one study in which an attempt to clarify the life cycle of Culicoides found associated with African wildlife was made.223 This dealt with five species of the subgenus Avaritia, which were discovered feeding in considerable numbers behind the ears of the African elephant (Figure 5.18) and to breed in its dung. The midges were collected off culled elephants in broad daylight and included specimens that were fully and freshly engorged; this clearly shows that certain species of Culicoides feed throughout the diel, and not only at night as is commonly believed. It was learned too that all the Culicoides spp. collected on the ears of elephant were also those that breed in elephant dung, indicating their co-evolvement, a fact that is further substantiated by the observation that these mostly new species are exceptionally rarely collected outside the range of elephants. It was observed that gravid females of these species oviposit in fresh dung in broad daylight, and that Culicoides are among the first insects to arrive at freshly evacuated dung. Gravid Culicoides females must therefore track elephants in small swarms, and when dung is evacuated they immediately descend upon it to lay their eggs. Oviposition is rapid and is a likely part of an evolutionary adaptation enabling the Culicoides to relocate the elephant, which will have proceeded little more than 50 m from the original point of dung deposition. By once again attaching themselves behind the ear to obtain a fresh blood meal these elephant-associated Culicoides repeat the feeding cycle needed for the development of their next batch of eggs.
It appears that, after blood-engorgement, the Culicoides do not stay on the elephant, but rather abandon the host to reside on vegetation during the two or three days required for egg development. Here they will await the return of elephant to once again track them but only if they are fully gravid. It is well known that elephant herds, led by matriarchs, move in well-defined home ranges, and will daily traverse the same well-worn paths for feeding and watering.
Figure 5.15 Culicoides imicola: log (n+1) numbers captured weekly at 39 livestock holdings spread across South Africa between September 1996 (week 37) and September 1997 (week 36); sites are ranked from highest to lowest rainfall (1 200 mm to 250 mm/annum). The weekly Culicoides imicola totals are colour-coded: white (no collection made), green (0 C. imicola), yellow (1 to 9), orange (10 to 99), red (100 to 999), pink (1 000 to 9 999) and dark blue (10 000 to 99 999)
Figure 5.16 Culicoides bolitinos: log (n+1) numbers captured weekly at 39 livestock holdings spread across South Africa between September 1996 (week 37) and September 1997 (week 36); sites are ranked from highest to lowest rainfall (1 200 mm to 250 mm/annum). The weekly Culicoides bolitinos totals are colour-coded: white (no collection made), green (0 C. imicola), yellow (1 to 9), orange (10 to 99), red (100 to 999), pink (1 000 to 9 999) and dark blue (10 000 to 99 999)
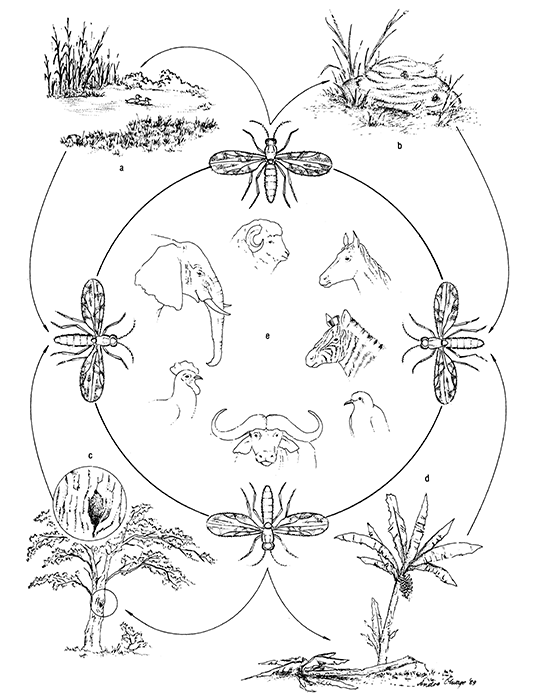
Figure 5.17 Diagrammatic representation of the interaction between Culicoides spp., their hosts and preferred habitat types. Each Culicoides sp. requires a specific substrate for the development of its larvae; these larval habitats may be divided into four main types:
| a b c d | surface water and soil interface (organically enriched or not) dung pats from large game or livestock tree-holes, plant and rock cavities rotting plant stems, fruit and fungi |
Culicoides spp. require the blood of specific host animals (e) in order to develop their eggs

Figure 5.18 The elephant possesses no sweat glands and so cools its blood by pumping large amounts (5 to 12 0/min.) through the ears. The skin at the back of these heavily veined ‘radiators’ is smooth, and is where hundreds of Culicoides feed. This photograph of eight Culicoides on the ear of an elephant was taken at 15:00 and includes a freshly blood-fed female
This would facilitate the development of sizeable populations of elephant-associated Culicoides in the immediate vicinity of these routes. An elephant bull daily consumes 300 kg of vegetation and evacuates 100 to 150 kg of dung.223 More than 3 500 Culicoides have been found to hatch from 5,75 kg of elephant dung.216 Thus the daily accrual in numbers of elephant-associated Culcoides amongst the 7 500 elephants of the Kruger National Park, in South Africa, could total roughly 500 million individuals.
One of the species associated with elephant is C. loxodontis, a member of the Imicola complex.214 Because it is closely allied to both C. imicola and C. bolitinos the question arises as to whether C. loxodontis is not also involved in the transmission of orbiviruses. It was suggested that the elephant may be a natural host for AHSV93 but subsequent studies have failed to bear this out.15 It would seem that in South Africa at least the elephant plays no part in the epidemiology of AHSV as the disease appears in large areas from which elephants are absent. Elephant-associated Culicoides are easily identifiable and they occur exceptionally rarely elsewhere in South Africa. This indicates that their mass movements, if any, over large distances, either do not occur or have yet to be observed.217, 222 Furthermore, an intensive two-year survey of the Culicoides that occur in and around the Kruger National Park which yielded over five million midges, failed to reveal the presence of significant numbers of elephant-associated Culicoides on livestock farms immediately adjoining the Park.217
Culicoides and the plains zebra
Recently, another new species of the Imicola complex, C. sp. # 107, was discovered, 217 almost solely restricted to the warmer and drier central-northern, northern and eastern parts of South Africa. In the Kruger National Park it enjoys a close association with the plains zebra (Figure 5.19) which is a host of AHSV.13 Significantly C. sp. # 107 breeds in the dung of zebra and, unusually, is most prevalent during the cooler seasons but only in frost-free areas.
Whilst C. sp. # 107 never becomes abundant, no doubt due to the limited availability of its breeding medium, its occurrence during winter raises the question as to whether or not C. sp. # 107 is capable of transmitting AHS at a time of the year when the disease is not considered a problem but has been shown still to be circulating in zebra.13
The question is potentially significant as C. sp. # 107 can be found in some abundance around horse stables where it in all probability breeds in the dung of horses.217 It also breeds abundantly in the dung of the white (square-lipped) rhinoceros (C. simum).217
Culicoides and the African buffalo
Both C. imicola and C. bolitinos have been collected live off a darted African buffalo (Figure 5.20).213 There is little doubt that C. bolitinos especially is closely associated with this host; in the Kruger National Park more than 1 000 specimens of C. bolitinos have been reared from a single buffalo dung pat.216 Of veterinary importance is that C. bolitinos also uses the dung of domestic cattle for breeding. This ensures that this seemingly efficient vector of BTV occurs almost throughout South Africa, and probably plays a major role in the infection of cattle with the virus. In support of this is the report that a 90 per cent BTV-infection rate has been found in some buffalo herds in the Kruger National Park,279 but this does not exclude the involvement of other Culicoides spp. in their infection. Of the hundreds of light trap collections made in the Kruger National Park the largest numbers of C. bolitinos, numbering in their thousands, were captured in the immediate vicinity of grazing buffalo herds. In these collections C. bolitinos was the dominant species, comprising more than 80 per cent of the captures; C. imicola comprised less than 3 per cent.217 The only other species of Avaritia that is likely to be associated with the buffalo is C. gulbenkiani but this has yet to be demonstrated.
Culicoides and domesticated livestock
Because of their minute size and generally nocturnal habits it is difficult to collect or identify Culicoides on any particular host. Virtually no bait-trapping has been done in Africa, and therefore the presence of large numbers of a particular species near specific livestock species is generally assumed to indicate that the Culicoides feed on these hosts.265 The wing patterns of the 14 species of Culicoides most commonly encountered near livestock in South Africa are shown in Figure 5.3.
These, ranked in order of abundance, are C. imicola, C. subschultzei, C. magnus, C. zuluensis, C. bolitinos, C. pycnostictus, C. leucostictus (= C. distinctipennis in earlier South African studies), C. nivosus, C. schultzei, and C. enderleini. These species made up 90 per cent of the 86 Culicoides spp. collected countrywide.222 Although close on 4 000 collections were made in the particular survey only 75 per cent of the Culicoides known to occur in South Africa were captured, indicating that many species either have highly specialized feeding preferences or larval habitats. A similar pattern of species prevalence and abundance was recently reported from Zimbabwe and Botswana.254, 255
Another approach used to determine the host preferences of Culicoides spp. is to count the number of sensilla coeloconica (‘pits’) found on 13 of the 15 antennal flagellar segments (‘antennomeres’) of the female.48 It is considered that a preference for mammals is indicated by six or fewer pit-bearing ‘antennomeres’, while those with 12 or 13 show a preference for birds. Although this approach may have much merit, reliance for its verification is still placed on blood meal identification.
In southern Africa Culicoides blood meal identification using various types of precipitin tests and an enzyme-linked immunosorbent assay (ELISA) has been limited to studies conducted in Zimbabwe and South Africa (Table 5.5).57, 265, 267 Blood meal identification can sometimes give a biased impression of the potential vector importance of certain Culicoides spp., especially if these species are collected in large numbers at isolated sites. For example, 215 Culicoides spp. near glabripennis tested positive for the blood of four host animals at one site, yet this species was rare in catches made at the other 23 sites in the Onderstepoort Veterinary Institute area (Table 5.5).
Culicoides spp. which usually feed on mammals will occasionally feed on birds if large numbers of them are present in an area and their primary source of blood is scarce. For example, in Zimbabwe, a small proportion of the large numbers of C. imicola, C. zuluensis and C. milnei collected on a poultry farm of 75 000 birds had fed on the poultry (Table 5.5), which indicates that under specific circumstances they may become opportunistic feeders.50
Conversely, known bird-feeders, such as C. leucostictus (= C. distinctipennis in early studies on South African Culicoides) and C. pycnostictus, must occasionally feed on mammals (Table 5.5), as suggested by the fact that in South Africa two BTV serotypes have been isolated from C. pycnostictus (Table 5.2). Epidemiologically this could be significant in the transmission of virus from birds to mammals and vice versa. Culicoides pycnostictus was recently also infected in the laboratory with BTV.282 However, the data remain confusing as in a more recent study221 it was found that at least five bird-associated species (C. leucostictus, C. pycnostictus, C. neavei, C. bedfordi and the Onderstepoortensis complex) would not enter horse stables whereas seven mammalophilic species did so readily. Whether this reflects a strong exophilic tendency, or whether mammals are truly unattractive to C. pycnostictus and thus very seldom attacked, remains to be clarified.
Life cycle
The life cycle of Culicoides spp. worldwide was reviewed in 1984 by Kettle.180 In South Africa an early attempt to clarify the life cycle of some species was made when C. imicola, C. pycnostictus, C. nivosus, C. leucostictus (as C. distinctipennis), C. enderleini (as C. schultzei), C. zuluensis, C. bedfordi and C. magnus were maintained in the laboratory under controlled conditions.260 After a blood meal had been taken, it was found that the maturation of eggs in C. imicola females took two days at 27 °C and three to four days at 22 °C. The ‘sausageshaped’ brown eggs were 400 μm long by 50 μm wide and were laid in a staggered double-rowed pattern somewhat resembling footprints. At 21 to 24 °C the eggs of five South African Culicoides spp. took three days to hatch. The number of eggs per batch varied from a mean of 69 for C. imicola to 162 for C. nivosus, but the number of batches was not determined. In C. variipennis, a North American species, up to four batches may be produced in a life-time.251 A blood meal must be taken by the female in between each batch of eggs.
Larvae of Culicoides go through four stages, are eel-like in their movements and burrow in and out of their breeding medium. As is shown by the variations in the structure of the epipharynges of different species,262 the food preferences of larvae vary; many are carnivorous and feed on protozoa, rotifers and nematodes.200 The fourth stage larvae of C. zuluensis and C. nivosus have been reported to be cannibalistic on second stage larvae.260 In the laboratory at 21 to 24 °C, larvae of C. nivosus, C. leucostictus (= C. distinctipennis in earlier South African studies), C. pycnostictus and C. bedfordi developed readily in a mixture of moist soil and cow dung and reached the pupal stage in 10 to 20 days. The following developmental times for six South African Culicoides spp. were: larval period 7 to 25 days, pupal period four days, oviposition to first adult 20 days. A generation (egg to egg) requires a minimum of 25 days. Adult Culicoides survive for between 14 and 21 days if fed on water containing 10 per cent honey.260 Further details on the life cycle of five species of Avaritia found in association with the African elephant have been reported.223
In attempts to explain the possible effect of various climatic factors on the survival and seasonal prevalence of Culicoides spp., especially C. imicola, the effect of desiccation, submersion in water and exposure to low temperatures on different life stages have been investigated.260 Eggs showed some resistance to desiccation, for example, 1 per cent of eggs allowed to dry at 85 per cent relative humidity did not collapse and later hatched on immersion in water. Low temperatures adversely affected egg viability. If the eggs were kept at 6,5 °C, viability started to decrease after seven days while none hatched after 37 days. The immersion in water of thousands of eggs, mostly of C. imicola, had no adverse effect on hatching — the larvae of seven species, including C. imicola, survived immersion in water at 22 °C for 6 to 13 days. Fourth instar larvae of C. nivosus, C. pycnostictus and C. zuluensis present in breeding medium survived refrigeration at 6,5 °C for 10 to 14 days, and completed their development normally on return to room temperature. On immersion, the pupae of all species, except C. imicola, wriggled free of the breeding medium and floated to the surface. Culicoides imicola pupae, however, lay on the substrate below the water surface and drowned within two days at room temperature. At 6,5 °C they survived for at least six days under water. The effect of low temperatures on adult survival and longevity was also tested.263 This showed that at temperatures as low as −1,5 °C, 15 per cent of C. imicola adults lived beyond 15 days, whereas the last C. pycnostictus died on the 54th day.
The results of these laboratory studies indicate that low winter temperatures do not kill any stage of Culicoides but merely slow down development or induce diapause. It can also be deduced that soaking rains have no adverse effect on the eggs, larvae and pupae of most species, but that they do drown C. imicola pupae; the larvae of C. imicola will not pupate until conditions are sufficiently dry. A seven-year study of the seasonal abundance of C. imicola at the Onderstepoort Veterinary Institute showed a drop in adult numbers during sustained rainy periods followed by a sharp increase in populations during the drier periods that followed.263 The greater tolerance of C. pycnostictus adults to low temperatures may partially explain why it was the most common species found in two studies in the Free State Province of South Africa, and why C. imicola comprised less than 1 per cent of the total Culicoides collected.173
Larval habitats
Knowledge about the habitats of Culicoides biting midges has increased greatly since the early 1960s as a result of studies involving the collection of live pupae from moist situations in the field and rearing them through to the adult stage.262, 268 Breeding material suspected to contain immature stages has also been collected and placed in special emergence boxes or cages,118, 212, 213, 261 or allowed to emerge in situ in specially constructed tent-type emergence traps placed over breeding grounds or materials.276
There is a common tendency to believe that Culicoides breed only in moist low-lying areas but, although this is true for some species, others have more specialized larval habitats. The basic requirements for Culicoides larvae are moisture and a medium containing organic matter. Thus, Culicoides may breed in situations varying from those which are almost aquatic in nature (such as pond margins) to those where no free water is present but the humidity is close to 100 per cent (such as the interior of dung pats). The various larval habitats can be grouped into four main types (Figure 5.17).
- Surface water and soil interface situations (Figure5.17a). About half the known Culicoides spp. in southern Africa make use of various combinations of soil and water as a medium in which to lay their eggs. Soil may vary from coarse sand to the finest clay, and the basic medium may be enriched to a greater or lesser extent with decomposing plant matter, varying from intact material to humus, or with fresh to well-decomposed dung, such as is often found on irrigated pastures. The water may range from fresh flowing streams to polluted stagnant pools with varying degrees of acidity, alkalinity or salinity. The degree of light and the presence or absence of plant cover, which may be either tall or kept short by grazing animals, are additional important factors that govern the larval habitat of certain Culicoides spp. In southern Africa most of the major stock-associated species (C. imicola, C. zuluensis, C. magnus, C. schultzei group, C. pycnostictus, C. leucostictus, and C. nivosus) use one or another of the above combinations as their larval habitat.275 Similarly, in Zimbabwe,41 thousands of specimens of 16 species of Culicoides were reared from mud samples taken at the edge of water bodies. Short notes on the larval habitats of six South African ‘groundwater’ species have been published.269
- Dung pats of large animals (Figure 5.17b). In Africa these larval habitats were only recognized in 1968, and their importance appreciated since 1985.118, 212, 213, 261 At least 10 Culicoides spp., all in the subgenus Avaritia, require the fresh dung of certain animals to complete their life cycles. For example, the dung of the African buffalo, cattle and the blue wildebeest is used by C. bolitinos, which apparently also feeds on these hosts.213 Other species, most of them still to be described, breed in the dung of the elephant, the black rhinoceros (Diceros bicornis) and white rhinoceros (Ceratotherium simum), and the plains zebra.212
- Tree-holes, plant and rock cavities (Figure 5.17c). These larval habitats vary from deep, dark, water-filled holes to shallow, exposed but moist hollows which may contain various amounts of water, decomposing leaf litter and sediment. Tree-holes are not restricted to dense forests, as even the more sparsely treed savannahs are rich in treehole- associated Culicoides. About 15 per cent of African Culicoides spp. (including C. accraensis, C. clarkei, C. olyslageri olyslageri, C. eriodendroni, C. punctithorax and C. nigripennis) are known or suspected to breed in these habitats,269 but owing to the restricted size and availability of such rain-dependent habitats these species never become abundant and are rarely collected. Birds are thought to be their primary source of blood. From the wet boles or crotches of 11 genera of trees in Zimbabwe more than 500 specimens of Culicoides representing the Nigripennis, Accraensis and Eriodendroni species groups were reared.41 As noted above species from these complexes are very difficult to identify.
- Rotting fruits and plants (Figure 5.17d). These larval habitats have still to be investigated thoroughly. In South Africa a new Avaritia sp., closely related to C. pseudopallidipennis from West Africa, has been reared from the rotting fallen fruits of the sausage tree (Kigelia africana) and the marula tree (Sclerocarya birrea).117 In West Africa the larvae of C. grahamii (also of the subgenus Avaritia) have been found in the rotting stems of the banana plant.209 The cultivation of bananas by humans may thus lead to the creation of larval habitats for this vector of the human filarial worm, Mansonella perstans. A concerted effort to rear Culicoides from rotting fruits, flower bracts, plant stems, and large fungal fruiting bodies should lead to the discovery of the breeding habits of several known and, perhaps, even new species. Some of these habitats, and many others, were sampled in Zimbabwe but without success.41
Control of Culicoides
Around livestock, Culicoides can occur in stupendous numbers especially on warmer nights and during periods of above average rainfall. At such times more than a million C. imicola can be captured in a single light trap in one night (Figure 5.21),219 and, if the estimate that this may represent a mere 1 per cent of the number of midges active on a particular night in the area concerned is correct, it graphically illustrates the intensity of attacks that must on occasion be endured nightly by exposed animals. It is impossible to eradicate such numbers of Culicoides and in Africa specifically, the first line of defence against Culicoides-borne orbiviruses must remain vaccination. However, even in the case of a notifiable disease such as the highly fatal AHS, and despite the social and commercial value of horses, it appears that less than half the horse population in southern Africa is being vaccinated annually. Recourse to additional methods of disease control, such as the elimination of the larval habitat of C. imicola, has received no attention in Africa perhaps because little success has been achieved with other species of Culicoides elsewhere in the world. Under restricted situations it may be feasible to reduce Culicoides adult numbers by treatment of their breeding sites with compounds such as Temephos.158 However, C. imicola’s potential for an explosive increase in numbers and its rapid radiation over large areas as soils become suitably moist under continuous rains, would make the widespread application of Temephos not only expensive but impracticable. For the same reasons, the biological control of C. imicola larvae would also not be feasible even if such a measure were available. Similarly, the daily treatment of animals with pyrethroids to reduce adult biting rates is labour intensive and expensive. Despite limited successes reported worldwide,252 and reviewed in some detail below, they merit further study, particularly in Africa. In Israel certain extracts of plants from the families Meliaceae and the Labiatae (including oils derived from sage, rosemary and oregano) were found to have a weak to negligible repellent effect against C. imicola.46
Recourse to more traditional or esoteric remedies such as the smoking of stables and the feeding of garlic would appear equally ineffective, but such control strategies have received little serious attention from researchers.
In the early years of animal husbandry, farmers soon noted that distancing their animals from low-lying moist areas reduced the prevalence of disease, and it became customary to move stock to higher-lying terrain at night or even for entire seasons.61 However, the modern era has seen a reduction in farm size and a concomitant increase in stocking rates. Sustained irrigation further increases the numbers of Culicoides breeding sites and, in the case of the moist soil-loving C. imicola and the cattle-dung breeding C. bolitinos, it is obvious that few areas will escape being colonized by one, or both, of these vector species.
Besides vaccination only stabling appears to have the potential of more reliably and permanently protecting animals from the attacks of Culicoides. More than 100 years ago horse owners in South Africa noted that stabled horses rarely succumbed to AHS, but despite these early and persistent observations only in two studies in Africa,14, 221 and not many more elsewhere in the world, have the merits of protective housing been explored. The results of the African studies revealed that both C. imicola and C. bolitinos readily enter stables, and indicated that stables were only of value if well sealed. The extent of sealing required was assessed:221 an 80 per cent shade-cloth induced a 14-fold reduction in the numbers of midges entering stables baited with horses. Since Culicoides midges of most species are active mainly during twilight hours and at night, valuable or young animals, whose immunity is not established, should be stabled at night.
Because it is now evident that Culicoides will enter stables, it is essential that stables be well built and as many of the small openings as possible closed. Thousands of C. bolitinos (and C. imicola but in lesser numbers) have been captured at night in an open stable in the Clarens district of the eastern Free State Province of South Africa. The exclusion of Culicoides can be more successfully achieved by screening stables with insect gauze or shade-cloth,221 and will be made more effective if their surfaces are treated with a pyrethroid insecticide. For exceptionally valuable animals a stable can be made virtually insect-free by the installation of extractor fans which maintain a negative pressure so that odours from the animals that remain inside go largely undetected by the Culicoides outside. It does not seem that light traps operated outside a stable at night will collect Culicoides in large enough numbers to significantly reduce the attack rate on animals inside.
Another strategy for disease control mooted 40 years ago is to maintain decoy hosts, after it was suggested that the presence of cattle protected nearby sheep from contracting BT.116, 264 It must be pointed out, however, that because C. bolitinos is dependent upon cattle dung its numbers are likely to increase wherever cattle occur, and therefore the presence of cattle may actually increase risk. Indeed the implication of cattle in the epidemiology of AHS only recently became obvious after an outbreak of the disease in the Clarens area of the eastern Free State Province in 1998, when it was revealed that the maintenance of unvaccinated horses in the presence of cattle is risky. Decoy animals are also an added expense and consideration must be given to the fact that any increase in animal biomass on a farm will result in a concomitant increase in local Culicoides numbers and in viral transmission.
Since many Culicoides spp. breed in moist, organically enriched soil, large concentrations of them can be expected in wet low-lying areas. Such areas should, if possible, be avoided by animals between dusk and dawn, i.e. during the hours of midge activity, and especially in mid- to late summer. However, it must be borne in mind that Culicoides in search of a blood meal are quite capable of dispersing over considerable distances. In addition, for the tracking of hosts they are equipped with acute sensory apparatuses (on the antennae) and therefore will locate animals wherever they may be kept on a farm. In the case of C. bolitinos the presence and amount of cattle dung will determine its presence and relative abundance.
Therefore, the removal of dung should lower the numbers of C. bolitinos but this is only practically feasible where cattle do not range extensively. Furthermore, such hygienic practices would have to be implemented over an entire farming community as C. bolitinos would be able to disperse from neighbouring farms that might harbour C. bolitinos if some owners are less careful about the removal of dung. In the case of C. imicola, its capacity to breed explosively under rainy conditions makes it an even more formidable insect to control.
No publications exist concerning the efficacy of repellents against Culicoides in the African situation. The chemical control of adult biting midges by direct treatment of livestock with pesticides is not usually practical under extensive farming conditions, but it may be justifiable for valuable animals such as racehorses. Many pyrethroid insecticides are effective against the Diptera, and, depending on the formulation used, have a reasonably long residual effect. Sprays can be used weekly and, in cattle, insecticide impregnated eartags may be effective for four to six weeks,18 or even as long as 10 weeks against Culicoides.159 In Australia it has been found that, after Hereford yearling cattle had been given a single subcutaneous injection of ivermectin at a dose of 200 mcg/kg, the mean mortality of engorged Culicoides females 48 hours after feeding was 99 per cent for 10 days after treatment.300 An unfortunate adverse effect is the lethal effect of ivermectin on the dung beetle fauna which are so important for sanitation.
In a recent and thorough study252 very limited success was reported against C. sonorensis in North America with formulations of 5 per cent permethrin or 27 per cent pirimiphos- methyl applied to the dorsum of calves. It was found that engorged Culicoides exhibiting sublethal intoxication recovered and subsequently matured average-sized batches of eggs. However, a belly spray of 0,2 per cent permethrin substantially reduced numbers of engorged females, and lowered engorgement levels on the third and seventh day post-treatment, but by the tenth day little effect was noted. In the case of C. sonorensis it was shown that most feeding was done on the belly of the calf, but in Israel41 C. imicola was found to feed preferentially on the dorsum (70 per cent) of a horse, with less (28 per cent) feeding on the belly. These studies on the chemical control of Culicoides show mixed promise and so merit additional research, and particularly in Africa where attack rates appear to be high.
References
- AKIBA, K., 1960. Studies on the Leucocytozoon found in the chicken in Japan. II. On the transmission of L. caulleryi by Culicoides arakawae. Japanese Journal of Veterinary Science, 22, 309–317.
- AKIBA, K., KITAOKA, S., YAJIMA, T., 1959. Studies on the Leucocytozoon found in the chicken in Japan. III. Transmission of Leucocytozoon caulleryi by Culicoides arakawae. Japanese Journal of Veterinary Science, 21, 127–128.
- AL-BUSAIDY, S., HAMBLIN, C. & TAYLOR, W.P., 1987. Neutralizing antibodies to Akabane virus in free-living wild animals in Africa. Tropical Animal Health and Production, 19, 197–202.
- AL-BUSAIDY, S.M. & MELLOR, P.S., 1991. Isolation and identification of arboviruses from the Sultanate of Oman. Epidemiology and Infection, 106, 403–413.
- ANDERSON, R.C., 1956. The life cycle and seasonal transmission of Ornithofilaria fallisensis Anderson, a parasite of domestic and wild ducks. Canadian Journal of Zoology, 34, 485–525.
- ANON., 1967. Arboviruses and Human Disease. World Health Organization. Technical Report. Series no. 369.
- ATKINSON, C.T., 1986. Ultrastructure of the ookinetes of Haemoproteus meleagridis (Haemosporina: Haemoproteidae). Journal of Parasitology, 75, 135–141.
- ATKINSON, C.T., 1988. Epizootiology of Haemoproteus meleagridis (Protozoa, Haemosporina) in Florida: potential vectors and prevalence in naturally infected Culicoides (Diptera: Ceratopogonidae). Journal of Medical Entomology, 25, 39–44.
- ATKINSON, C.T., 1990. Fine structure and sporogonic development of a Vavraia sp. (Microsporidia: Pleistophoridae) in the biting midge Culicoides edeni (Diptera: Ceratopogonidae). Journal of Invertebrate Pathology, 55, 105–111.
- ATKINSON, C.T., 1991. Vectors, epizootiology, and pathogenicity of avian species of Haemoproteus (Haemosporina: Haemoproteidae). Canadian Journal of Zoology, 69, 1880–1888.
- BAIN, O., 1980. Two filariae of the genus Eufilaria in Turdus merula: development in Culicoides nubeculosus. Annales de Parasitologie Humaine et Comparee, 55, 583–590.
- BAKER, J.R., 1970. Leucocytozoon in Europe. Journal of Parasitology, 56 (Supplement), 16.
- BARNARD, B.J.H., 1993. Circulation of African horsesickness virus in zebra (Equus burchelli) in the Kruger National Park, South Africa, as measured by the prevalence of type specific antibodies. Onderstepoort Journal of Veterinary Research, 60, 111–117.
- BARNARD, B.J.H., 1997. Some factors governing the entry of Culicoides spp. (Diptera: Ceratopogonidae) into stables. Onderstepoort Journal of Veterinary Research, 64, 227–233.
- BARNARD, B.J.H., BENGIS, R., KEET, D. & DEKKER, E., 1994. Epidemiology of African horsesickness: antibodies in free-living elephants (Loxodonta africana) and their response to experimental infection. Onderstepoort Journal of Veterinary Research, 62, 271–275.
- BARNARD, B.J.H., GERDES, G.H. & MEISWINKEL, R., 1998. Some epidemiological aspects of a bluetongue-like disease in South Africa – 1995/96 and 1997. Onderstepoort Journal of Veterinary Research, 65, 145–151.
- BARTLETT, C.M. & ANDERSON, R.C., 1980. Development of Chandlerella chitwoodae (Filarioidea: Onchocercidae) in Culicoides stilobezzioides and Culicoides travisi (Diptera: Ceratopogonidae). Canadian Journal of Zoology, 58, 1002–1006.
- BATH, G.F., 1989. Bluetongue. Proceedings of the Second International Congress for Sheep Veterinarians, New Zealand Veterinary Association, Massey University, Palmerston North, 12–16 February 1989, 349–357.
- BAYLEY, T.B., 1856. Notes on the horse-sickness at the Cape of Good Hope, in 1854–’55. Cape Town: Saul Solomon & Co., Steam Printing Office, Longmarket Street.
- BAYLIS, M., BOUAYOUNE, H., TOUTI, J. & EL HASNAOUI, H., 1998. Use of climatic data and satellite imagery to model the abundance of Culicoides imicola, the vector of African horse sickness virus, in Morocco. Veterinary and Medical Entomology, 12, 255–266.
- BAYLIS, M., MEISWINKEL, R. & VENTER, G. J., 1999. A preliminary attempt to use climate data and satellite imagery to model the abundance and distribution of Culicoides imicola (Diptera: Ceratopogonidae) in southern Africa. Journal of the South African Veterinary Association, 70, 80–89.
- BAYLIS, M., MELLOR, P.S. & MEISWINKEL, R., 1999. Horse sickness and ENSO in South Africa. Nature, 397, 574.
- BAYLIS, M. & RAWLINGS, P., 1998. Modelling the distribution and abundance of Culicoides imicola in Morocco and Iberia using climatic data and satellite imagery. Archives of Virology (Supplement 14), 137–153.
- BELL, R.A., 1999. Outbreak of African horse sickness in the Cape Province of South Africa. The Veterinary Record, 144, 483.
- BENNET, G.F., 1970. Development of trypanosomes of the T. avium complex in the invertebrate host. Canadian Journal of Zoology, 48, 945–957.
- BENNET, G.F. & COOMBS, R.F., 1975. Ornithophilic vectors of avian hematozoa in insular Newfoundland. Canadian Journal of Zoology, 53, 1241–1246.
- BERGNER, J.F., 1968. The parasites (with emphasis on the filaria Macacanema formosana) and the ecology of the Taiwan monkey, Macaca cyclopis. Dissertation Abstracts, 28, 2921–2922.
- BERGNER, J.F. & JACHOWSKI, L.A., 1968. The filarial parasite Macacanema formosana, from the Taiwan monkey and its development in various arthropods. Formosan Science, 22, 1–68.
- BEVAN, L.E.W., 1907. Preliminary work on the so-called ‘stiffness’ or ‘three-day-sickness’ of cattle in Rhodesia.Journal of Comparative Pathology and Therapeutics, 20, 104–113.
- BEVAN, L.E.W., 1912. Ephemeral fever or three days’ sickness of cattle. Veterinary Journal, 68, 458–461.
- BEVERIDGE, I., KUMMEROW, E.L., WILKINSON, P. & COPEMAN, D.B., 1981. An investigation of biting midges in relation to their potential as vectors of bovine onchocerciasis in north Queensland. Journal of the Australian Entomological Society, 20, 39–45.
- BHATNAGAR, P., PRASAD, G. & SRIVASTAVA, R.N., 1994. Culicoides (Ceratopogonidae) as vectors of bluetongue virus. Annals of Biology (Ludhiana), 10, 179–180.
- BIRLEY, M.H., BRAVERMAN, Y. & FRISH, K., 1984. Towards a survey of the survival rate and periodicity of some Culicoides species in Israel. Environmental Entomology, 13, 424–429.
- BLACKBURN, N.K., SEARLE, L. & PHELPS, R.J., 1985. Viruses isolated from Culicoides (Diptera: Ceratopogonidae) caught at the veterinary research farm, Mazowe, Zimbabwe. Journal of the Entomological Society of Southern Africa, 48, 331–336.
- BLACKWELL, A., YOUNG, M.R., & MORDUE, W., 1994. The microhabitat of Culicoides impunctatus (Diptera: Ceratopogonidae) larvae in Scotland. Bulletin of Entomological Research, 84, 295–301.
- BOORMAN, J., 1991. A review of Culicoides subgenus Avaritia species (Insecta, Diptera, Ceratopogonidae) vectors of viruses of sheep, cattle and horses, with particular reference to Culicoides imicola in Europe and the Mediterranean region. A report developed for The Overseas Development Administration. pp. 54.
- BOORMAN, J. & DIPEOLU, O. O., 1979. A taxonomic study of adult Nigerian Culicoides Latreille (Diptera: Ceratopogonidae species). Entomological Society of Nigeria Occasional Publication, 22. pp. 121.
- BORKENT, A., 1996. Biting midges from Upper Cretaceous New Jersey Amber (Ceratopogonidae: Diptera). American Museum Novitates, 3159, 1–29.
- BORKENT, A. & WIRTH, W.W., 1997. World species of biting midges (Diptera: Ceratopogonidae). Bulletin of the American Museum of Natural History, 233, 1–257.
- BOULARD, Y., LANDAU, I., MILTGEN, F., BACCAM, D. & HUMPHREY-SMITH, I., 1985. Ultrastructural observations on the sporogony in Hepatocystis levinei, hemoproteidan parasite of Australian Chiroptera, in Culicoides nubeculosus. Protistologica, 21, 93–105.
- BRAVERMAN, Y., 1978. Characteristics of Culicoides (Diptera: Ceratopogonidae) breeding places near Salisbury, Rhodesia. Ecological Entomology, 3, 163–170.
- BRAVERMAN, Y., 1988. Preferred landing sites of Culicoides species (Diptera: Ceratopogonidae) on a horse in Israel and its relevance to summer seasonal recurrent dermatitis (sweet itch). Equine Veterinary Journal, 20, 426–429.
- BRAVERMAN, Y., 1992. Host detection, hourly activity, and preferred biting sites of Culicoides imicola (Diptera: Ceratopogonidae) on a calf in Israel, In: WALTON, T.E. & OSBURN, B.I. (eds.) Bluetongue, African horse sickness, and related orbiviruses. Proceedings of the second international symposium. Boca Raton, Florida, U.S.A.: CRC Press.
- BRAVERMAN, Y., BARZILAI, E., FRISH, K. & RUBINA, M., 1985. Bluetongue virus isolations from pools of Culicoides spp. in Israel during 1981 to 1983. Progress in Clinical Biological Research, 178, 191–193.
- BRAVERMAN, Y., BOREHAM, P.F.L. & GALUN, R., 1971. The origin of bloodmeals of female Culicoides pallidipennis trapped in a sheepfold in Israel. Journal of Medical Entomology, 8, 379–381.
- bRAVERMAN, Y. & CHIZOV-GINZBURG, A., 1997. Repellency of synthetic and plant-derived preparations for Culicoides imicola. Medical and Veterinary Entomology, 11, 355–360.
- BRAVERMAN, Y., GALUN, R. & ZIV, M., 1974. Breeding sites of some Culicoides species (Diptera: Ceratopogonidae) in Israel. Mosquito News, 34, 303–308.
- BRAVERMAN, Y. & HULLEY, P.E., 1979. The relationship between the numbers of some antennal and palpal sense organs and host preference in some Culicoides (Diptera: Ceratopogonidae) from southern Africa. Journal of Medical Entomology, 15, 419–424.
- BRAVERMAN, Y. & LINLEY, J.R., 1988. Parity and voltinism of several Culicoides spp. (Diptera: Ceratopogonidae) in Israel, as determined by two trapping methods. Journal of Medical Entomology, 25, 121–126.
- BRAVERMAN, Y., LINLEY, J.R., MARCUS, R. & FRISH, K., 1985. Seasonal survival and expectation of infective life of Culicoides spp. (Diptera: Ceratopogonidae) in Israel, with implications for bluetongue virus transmission and a comparison of the parous rate in C. imicola from Israel and Zimbabwe. Journal of Medical Entomology, 22, 476–484.
- BRAVERMAN, Y. & PHELPS, R.J., 1981. Species composition and blood-meal identification in samples of Culicoides (Diptera: Ceratopogonidae) collected near Salisbury, Zimbabwe, in 1976-77. Journal of the Entomological Society of Southern Africa, 44, 315–323.
- BRAVERMAN, Y. & RUBINA, M., 1976. Light trapping of biting insects in poultry houses in Israel. Israel Journal of Zoology, 3, 95–101.
- BRAVERMAN, Y., RUBINA, M. & FRISH, K. 1981. Pathogens of veterinary importance isolated from mosquitoes and biting midges in Israel. Insect Science Application, 2, 157–161.
- BRAVERMAN, Y. & SWANEPOEL, R., 1981. Infection and transmission trials with Nyabira virus in Aedes aegypti (Diptera: Culicidae) and two species of Culicoides (Diptera: Ceratopogonidae). Zimbabwe Veterinary Journal, 12, 13–17.
- BRAVERMAN, Y., UNGAR-WARON, H., FRISH, K., ADLER, H., DANIELI, Y., BAKER, K.P. & QUINN, P.J., 1983. Epidemiological and immunological studies of sweet itch in horses in Israel. The Veterinary Record, 112, 521–524.
- BROSTRÖMM, H. & LARSSON, A., 1987. Allergic dermatitis (sweet itch) of Icelandic horses in Sweden: An epidemiological study. Equine Veterinary Journal, 19, 229–236.
- BROWN, M.A., MORDUE (LUNTZ), A.J., HAMBLIN, C., VENTER, G.J., & MORDUE, W., 2001. Host preference studies of Culicoidesbiting midges. The Royal Entomological Society International Symposium on Insects and Disease, 10–12 September 2001.
- BUCKLEY, J.J.C., 1933. A note on the development of Filaria ozzardi in Culicoides furens Poey. Journal of Helminthology, 11, 257–258.
- BUCKLEY, J.J.C., 1934. On the development in Culicoides furens Poey of Filaria (Mansonella) ozzardiManson, 1897. Journal of Helminthology, 12, 99–118.
- BUCKLEY, J.J.C., 1938. On Culicoides as a vector of Onchocerca gibsoni (Cleland and Johnson, 1910). Journal of Helminthology, 16, 121–158.
- BURCHELL, W.J., 1822. Travels in the Interior of South Africa. Vol. 1, London: Longman. Hurst, Orme, Brown, and Green.
- CAEIRO, V.M.P., 1961. Contribuiçao para o estudo das espécies Angolanas do género Culicoides Latreille, 1809 (Diptera: Ceratopogonidae). Junta de Investigaçoes Cientifias do Ultramar, Estudos, Ensaios, e Documentos, 86, 1–359, 58 pls.
- CALISHER, C.H., MCLEAN, L.E., ZELLER. H.G., FRANCY, D.B., KARABATSOS, N. & BOWEN, R.A., 1990. Isolation of Tete serogroup bunyaviruses from Ceratopogonidae collected in Colorado. The American Journal of Tropical Medicine and Hygiene, 53, 314–318.
- CARTER, H.F. 1919. New West African Ceratopogonidae. Annals of Tropical Medicine and Parasitology, 10, 131–138.
- CARTER, H.F., INGRAM, A. & MACFIE, J.W.S., 1920. Observations on the ceratopogonine midges of the Gold Coast with descriptions of new species. Part II. Annals of Tropical Medicine and Parasitology, 14, 211–274, pls 7–8.
- CAUSEY, O.R., KEMP, G.E., CAUSEY C.E. & LEE, V.H., 1972. Isolation of Simbu-group viruses in Ibadan, Nigeria 1964–1969, including the new types Sango, Shamonda, Sabo and Shuni. Annals of Tropical Medicine and Parasitology, 66, 357–362.
- CHANDENIER, J., LANDAU, I. & BACCAM, D., 1988. Sur les trypanosomes d’oiseaux estrildidae. II. Etudes biologiques. Annales de Parasitologie Humaine et Comparee, 63, 243–252.
- CHARDOME, M. & PEEL, E., 1949. The occurrence of filaria in the region of Coquillatville and the transmission of Dipetalonema streptocerca by Culicoides grahamii. Annales Societe Belge de Medicine Tropicale, 29, 99–119.
- CHARDOME, M. & PEEL, E., 1951. Research on the occurrence of filaria in the region of Coquillatville and the transmission of Dipetalonema streptocerca by Culicoides grahamii Austen. Memoires Institut Royal Colonial Belge. Section des Sciences Naturelles et Medicales, 19, 3–83.
- CHRISTOPHERS, S.R., SHORTT, H.E. & BARRAUD, P.J., 1925. Culicoides and Kala-Azar in Assam. Indian Journal of Medical Research, 13, 175–176.
- CLASTRIER, J., 1958. Notes sur les Cératopogonidés. IV. Cératopogonidés d’Afrique occidentale francaise. Archives de l’Institut Pasteur d’Algérie, 36, 192–258.
- CLASTRIER, J., 1959. Notes sur les Cératopogonidés. VI. Cératopogonidés d’Afrique occidentale francaise (3). Archives de l’Institut Pasteur d’Algérie, 37, 167–197.
- CLASTRIER, J., 1960. Notes sur les Cératopogonidés. IX. Cératopogonidés de la République du Congo. Archives de l’Institut Pasteur d’Algérie, 38, 79–105.
- CLASTRIER, J. & WIRTH, W.W., 1961. Notes sur les Cératopogonidés. XIV. Cératopogonidés de la Region éthiopienne (2). Archives de l’Institut Pasteur d’Algérie, 39, 302–337.
- COLAÇO, A.T.F., 1946. Alguns Culicoides do Transvaal. Annais do Instituto de Medicina Tropical, 3, 217–266.
- COLLINS, R.C. & JONES, R.H., 1978. Laboratory transmission of Onchocerca cervicalis with Culicoides variipennis. American Journal of Tropical Medicine and Hygiene, 27, 46–50.
- COLLISON, E.W. & BARBER, T.L., 1985. Isolation and identification of bluetongue virus: a serotype new to the U.S. Progress in Clinical Research, 178, 319–327.
- CONNAN, R.M. & LLOYD, S., 1988. Seasonal allergic dermatitis in sheep. The Veterinary Record, 123, 335–337.
- COOK, R.S., 1971. Haemoproteus Kruse 1890. In: DAVIS, J.W., (ed.). Infectious and Parasitic Diseases of Wild Birds. Iowa: Iowa State University Press. pp. 300–308.
- CORNET, M., 1969. Les Culicoïdes (Diptera Ceratopogonidae) de l’Ouest africain (1ère note). Cahiers de l’Office de la Recherche Scientifique et Technique Outre-Mer, série Entomologie médicale et Parasitologie, 7, 341–364.
- CORNET, M. & BRUNHES, J., 1994. Revision des espèces de Culicoides apparentées a C. schultzei (Enderlein, 1908) dans la region afrotropicale (Diptera, Ceratopogonidae). Bulletin de la Societe Entomologique de France, 99, 149–164.
- CORNET, M. & CHATEAU, R., 1970. Les Culicoides de l’Ouest africain (2e note). Espèces apparentées à C. similis Carter, Ingram et Macfie, 1920 (Diptera, Ceratopogonidae). Cahiers de l’Office de la Recherche Scientifique et Technique Outre-Mer, série Entomologie médicale et Parasitologie, 8, 141–173.
- CORNET, M. & NEVILL, E.M., 1980. Culicoides macintoshi n. sp., une nouvelle espèece d’Afrique du Sud (Diptera, Ceratopogonidae), avec une note sur la taxonomie des espèces éthiopiennes à ailes sans taches. Cahiers de l’Office de la Recherche Scientifique et Technique Outre-Mer, série Entomologie médicale et Parasitologie, 18, 383–389.
- CORNET, M., NEVILL, E.M & WALKER, A.R., 1974. Note sur les Culicoides (Diptera, Ceratopogonidae) du groupe de C. milnei Austen, 1909, en Afrique orientale et australe. Cahiers de l’Office de la Recherche Scientifique et Technique Outre-Mer, série Entomologie médicale et Parasitologie, 12, 231–243.
- CRANE, G.R., ELBEL, R.E., FRANCY, D.B. & CALISHER, C.H., 1983. Arboviruses from western Utah, 1967–1976. Journal of Medical Entomology, 20, 294–300.
- CYBINSKI, D.H. 1984. Douglas and Tinaroo viruses: two Simbu group arboviruses infecting Culicoides brevitarsis and livestock in Australia. Australian Journal of Biological Science, 37, 91–97.
- CYBINSKI, D.H. & MULLER, M.J., 1990. Isolation of arboviruses from cattle and insects at two sentinel sites in Queensland, Australia 1979–85. Australian Journal of Zoology, 38, 25–32.
- CYBINSKI, D.H. & ST. GEORGE, T.D., 1982. Preliminary characterisation of D’Aguilar virus and three Palyam group viruses new to Australia. Australian Journal of Biological Science, 35, 343–351.
- CYBINSKI, D.H., ST. GEORGE, T.D., STANDFAST, H.A. & MCGREGOR, A., 1980. Isolation of Tibrogargan virus, a new Australian rhabdovirus, from Culicoides brevitarsis. Veterinary Microbiology, 5, 301–308.
- DA COSTA MENDES, V.M., 1984. The isolation and importance of Simbu group viruses in South Africa. M.Med. Vet. (Vir.) Thesis. University of Pretoria.
- DAS GUPTA, B. & PAL, N., 1976. Malarial oocysts in Culicoides sp. in Darjeeling. Transactions of the Royal Society of Tropical Medicine and Hygiene, 70, 89–90.
- DAVIES, F.G. & HIGHTON, R.B., 1980. Possible vectors of Rift Valley Fever in Kenya. Transactions of the Royal Society of Tropical Medicine and Hygiene, 74, 815–816.
- DAVIES, F.G. & OTIENO, S., 1977. Elephants and zebras as possible reservoir hosts for African horsesickness virus. The Veterinary Record, 100, 291–292.
- DAVIES, F.G., SOI, R.K. & BINEPAL, V.S., 1993. African horse sickness viruses isolated in Kenya. The Veterinary Record, 132, 440.
- DAVIES, F.G. & WALKER, A.R., 1974. The isolation of ephemeral fever virus from cattle and Culicoides midges in Kenya. The Veterinary Record, 95, 63–64.
- DAVIES, F.G. & WALKER, A.R., 1974. The distribution in Kenya of bluetongue virus and antibody, and the Culicoides vector. Journal of Hygiene, Cambridge, 72, 265–272.
- DAVIES, F.G., WALKER, A.R., OCHIENG, P. & SHAW, T., 1979. Arboviruses isolated from Culicoides midges in Kenya. Journal of Comparative Pathology, 89, 587–595.
- DAVIES, J.B., TREES, A.J., MCCALL, P.J., BOCKARIE, M.J., THOMPSON, M.C., MCKELLAR, S.B. & MATTURI, A.S., 1989. On the possibility of bovine Onchocerca species infecting Simulium damnosum sensu lato in the forest zone of Sierra Leone. II. Biting densities and filarial infections in Similium spp. and Culicoides spp. Annals of Tropical Medicine and Parasitology, 83, 603–614.
- DELLA-PORTA, A.J., MURRAY, M.D., HARTLEY, W.J., STANDFAST, H.A. & ST. GEORGE, T.D., 1975. Akabane virus. Report of the CSIRO Division of Animal Health, 19–24.
- DE MEILLON, B., 1936. Entomological studies. Studies on insects of medical importance in South Africa – Part III. South African Ceratopogonidae. Part II. Some new and unrecorded species. Publications of the South African Institute for Medical Research, 7, 141–207.
- DE MEILLON, B., 1937. Entomological studies. Studies on insects of medical importance in South Africa and adjacent territories (Part IV). 2. Records and species from South Africa. Publications of the South African Institute for Medical Research, 7, 323–385.
- DE MEILLON, B., 1942. New Nematocera from the Ethiopian Region. Journal of the Entomological Society of Southern Africa, 5, 87–98.
- DE MEILLON, B., 1942. Ceratopogonidae (Dipt. Nematocera) from southern Rhodesia. Transactions of the Rhodesian Scientific Association, 39, 113–119.
- DE MEILLON, B., 1942. Simuliidae and Ceratopogonidae (Dipt. Nematocera) from the colony of Mocambique. Estacao Anti-malarica de Lourenco Marques. 26 pp.
- DE MEILLON, B., 1943. New records and new species of Nematocera (Diptera) from the Ethiopian Region. Journal of the Entomological Society of Southern Africa, 6, 90–113.
- DE MEILLON, B., 1947. New records and species of biting insects from the Ethiopian Region. II. Journal of the Entomological Society of Southern Africa, 10, 110–124.
- DE MEILLON, B., 1952. A new species of Culicoides (Diptera: Ceratopogonidae) from Uganda. Proceedings of the Royal Entomological Society of London (B), 21, 173.
- DOHERTY, R.L., CARLEY, J.G., FILIPPICH, C., BARROW, G.J., WILSON, R. & BROWN, K., 1975. Epidemiological studies of arboviruses. Report of the Queensland Institute of Medical Research, 30, 6–13.
- DOHERTY, R.L., CARLEY, J.G., FILIPPICH, C., GRAVATT, D.J., BARROW, G.J. & SMITH, D., 1976. Epidemiological studies of arboviruses. Report of the Queensland Institute of Medical Research, 31, 7–14.
- DOHERTY, R.L., CARLEY, J.G., STANDFAST, H.A., DYCE, A.L., KAY, B.H. & SNOWDON, W.A., 1972. Virus strains isolated from arthropods during an epizootic of bovine ephemeral fever in Queensland. Australian Veterinary Journal, 48, 81–86.
- DOHERTY, R.L., CARLEY, J.G., STANDFAST, H.A., DYCE, A.L., KAY, B.H. & SNOWDON, W.A., 1973. Isolations of arboviruses from mosquitoes, biting midges, sandflies and vertebrates collected in Queensland, 1969 and 1970. Transactions of the Royal Society of Tropical Medicine and Hygiene, 67, 536–543.
- DOHERTY, R.L., STANDFAST, H.A., DYCE, A.L., CARLEY, J.G., GORMAN, B.M., FILIPPICH, C. & KAY, B.H., 1978. Mudjinbarry virus, an orbivirus related to Wallal virus isolated from midges from the Northern territory of Australia. Australian Journal of Biological Science, 31, 97–103.
- DOHNAL, J., BLINN, J., WAHL, G. & SCHULTZ-KEY, H., 1990. Distribution of microfilariae of Onchocerca lienalis and Onchocerca gutturosa in the skin of cattle in Germany and their development in Simulium ornatum and Culicoides nubeculosus following artificial infestation. Veterinary Parasitology, 36, 325–332.
- DUKE, B.O.L., 1956. The intake of the microfilariae of Acanthocheilonema perstans by Culicoides grahamii and their subsequent development. Annals of Tropical Medicine and Parasitology, 48, 416–420.
- DU TOIT, R.M., 1944. The transmission of bluetongue and horsesickness by Culicoides. Onderstepoort Journal of Veterinary Research, 19, 7–16.
- DU TOIT, R.M., 1962. The role played by bovines in the transmission of bluetongue to sheep. Journal of the South African Veterinary Medical Association, 33, 483–490.
- DYCE, A.L., 1988. 48 Queens Rd., Asquith, N.S.W. 2077. Unpublished data.
- DYCE, A.L. & MARSHALL, B.D., 1989. An early record of Culicoides species (Diptera: Ceratopogonidae) developing in the dung of game animals in southern Africa. Onderstepoort Journal of Veterinary Research, 56, 85–86.
- EBERHARD, M.L. & LOWRIE, R.C., 1979. Development of Dipetalonema gracile and D. caudispina to the infective stages in Culicoides hollensis. Journal of Parasitology, 65, 89–95.
- EDINGTON, A., 1895. Cape Horse-Sickness.The Veterinarian, 68, 595–607.
- ELBEL, R.F., CRANE., G.T. & CALISHER, C.H., 1977. Arbovirus isolation from southwestern Utah and northwestern Arizona insects 1972–1975. Mosquito News, 37, 497–505.
- ENDERLEIN, G., 1908. 2. Ceratopogonidae. Neue Ceratopogoninen aus Südafrika. In: Zoologische und anthropologische Ergebnisse einer Forschungsreise im Westlichen unde zentralen Südafrika ausgeführt in dem Jahren 1903–1905. Erster Band: Systematik und Tiergeographie, zweite Lieferung. Insecte E. Diptera. Denkschriften der Medizinisch-Naturwissenschaftlichen Gesellschaft zu Jena, 13, 459–461.
- FADZIL, M. & CHEAH, T.S., 1975. Culicoides arakawae (Arakawa, 1910): a possible vector of Akiba caulleryi in peninsular Malaysia (preliminary report). Kajian Veterinaire (Singapore), 7, 32–36.
- FAHRNER, J., VANKAN, D. & SCHULZ-KEY, H., 1988. Ingestion of microfilariae from different sources by Culicoides nubeculosus (Diptera: Ceratopogonidae) through an artificial membrane. Veterinary Parasitology, 28, 315–320.
- FALLIS, A.M., 1958. Biting midges (Ceratopogonidae) as intermediate hosts for Haemoproteus of ducks. Proceedings of the Tenth International Congress of Entomology (1956), 3, 681.
- FALLIS, A.M., 1964. On the development and transmission of Leucocytozoon and Haemoproteus. Proceedings of the First International Congress of Parasitology (Romea), 1, 239–240.
- FALLIS, A.M. & BENNET, G.F., 1960. Sporogony of Leucocytozoon and Haemoproteus in simuliids and ceratopogonidae and a revised classification of the Haemosporidiida. Canadian Journal of Zoology, 39, 215–228.
- FALLIS, A.M. & BENNET, G.F., 1961. Ceratopogonidae as intermediate hosts for Haemoproteus and other parasites. Mosquito News, 21, 21–28.
- FALLIS, A.M., JACOBSON, R.L., & RAYBOULD, J.N., 1973. Haematozoa in domestic chickens and guinea fowl in Tanzania and transmission of Leucocytozoon neavei and Leucocytozoon schoutedeni. Journal of Protozoology, 20, 438–442.
- FALLIS, A.M. & WOOD, D., 1957. Biting midges (Diptera: Ceratopogonidae) as intermediate hosts for Haemoproteus of ducks. Canadian Journal of Zoology, 35, 425–435.
- FIEDLER, O.G.H., 1951. The South African biting midges of the genus Culicoides (Ceratopogonid: Dipt.). Onderstepoort Journal of Veterinary Research, 25, 3–33.
- FLOCH, H. & ABONNENC, E., 1949. Mansonella ozzardi and Culicoides furens in the French territories of the American continent. Comptes Rendus des Seances Societe de Biologie et des Ses Filiales, 143, 1343.
- FOIL, L.D., KLEI, T.R., MILLER, R.I., CHURCH, G.E., FRENCH, D.D. & SMITH, J.N., 1987. Seasonal changes in density and tissue distribution of Onchocerca cervicalismicrofilariae in ponies and related changes in Culicoides variipennis in Louisiana (USA). Journal of Protozoology, 73, 320–326.
- FOIL, L., STAGE, D., & KLEI, T.R., 1984. Assessment of wild-caught Culicoides (Ceratopogonidae) species as natural vectors of Onchocerca cervicalis in Louisiana (USA). Mosquito News, 44, 204–206.
- FOSTER, N.M., JONES., R.H., & MCCRORY, B.R., 1963. Preliminary investigation on insect transmission of bluetongue virus in sheep. American Journal of Veterinary Research, 24, 1195–1200.
- FOSTER, N.M., METCALF, H.E., BARBER, T.L., JONES, R.H. & LUEDKE, A.J., 1980. Bluetongue and epizootic hemorrhagic disease virus isolations from vertebrate and invertebrate hosts at a common geographic site. Journal of the American Veterinary Association, 176, 126–129.
- GARNHAM, P.C.C., HEISCH, R.B., MINTER, D.M., PHILIPPS, J.D. & IKATA, M., 1961. Culicoides adersi Ingram and Macfie, 1923, a presumed vector of Hepatocystis (= Plasmodium) kochi (Laveran, 1899). Nature, 190, 739–741.
- GARNHAM, P.C.C., HEISCH, R.B. & MINTER, D.M., 1961. The vector of Hepatocystis (Plasmodium) kochi, the successful conclusion of observations in many parts of tropical Africa. Transactions of the Royal Society of Tropical Medicine and Hygiene, 55, 497–502.
- GARNHAM, P.C.C., HEISCH, R.B., WIRTH, W.W. & IKATA, M., 1961. Hepatocystis (Plasmodium) kochi in Culicoides adersi. Transactions of the Royal Society of Tropical Medicine and Hygiene, 55, 304.
- GIBBS, E.P.J., CALISHER, C.H., TESH, R.B., LAZUICK, J.S., BOWEN, R. & GREINER, E.C., 1989. Bivens Arm virus: A new rhabdovirus isolated from Culicoides insignis in Florida and related to Tibrogargan virus of Australia. Veterinary Microbiology, 19, 141–150.
- GIBSON, C.J. & ASCOLI, W.F., 1952. The relationship of Culicoides (Diptera: Heleidae) to the transmission of Onchocerca volvulus. Journal of Parasitology, 38, 315–320.
- GLICK, J.I., 1990. Culicoides biting midges (Diptera: Ceratopogonidae) of Kenya. Journal of Medical Entomology, 27, 85–195.
- GOLDSMIT, L. & BARZILAI, E., 1985. Bluetongue in Israel. In: barber, t.l. & jochim, m.m., (eds.). Bluetongue and Related Orbiviruses. New York: Alan R. Liss.
- GREINER, E.C. 1975. Prevalence and potential vectors of Haemoproteus in Nebraska mourning doves. Journal of Wildlife Diseases, 11, 150–156.
- GREINER, E.C., BARBER, T.L., PEARSON, J.E., KREMER, W.L. & GIBBS, E.P.J., 1985. Orbiviruses from Culicoides in Florida. Progress in Clinical Biological Research, 178, 195–200.
- GREINER, E.C. & BENNET, G.F., 1977. Culicoides as vectors of Haemosporidia. Mosquito News, 37, 286.
- GREINER, E.C., EVELEIGH, E.S. & WAYNE, M.B., 1978. Ornithophilic Culicoides spp. (Diptera: Ceratopogonidae) from New Brunswick, Canada, and implications of their involvement in haemoproteid transmission. Journal of Medical Entomology, 14, 701–704.
- GREINER, E.C., FADOK, V.A. & RABIN, E.B., 1988. Equine Culicoides hypersensitivity in Florida: biting midges collected in lighttraps near horses. Medical and Veterinary Entomology, 2, 129–135.
- HALOUZKA, J. & HUBALEK, Z., 1996. Biting midges (Ceratopogonidae) of medical and veterinary importance (a review). Acta Scientiarum Naturalium Academiae Scientiarum Bohemicae-Brno, 30, pp. 56.
- HALOUZKA, J., PEJC´OCH, M., HUBALEK, Z. & KNOZ, J., 1991. Isolations of Tahyna virus from biting midges (Diptera: Ceratopogonidae) in Czecho-Slovakia. Acta Virologica, 35, 247–251.
- HAM, P.J., JAMES, E.R., & BIANCO, A.E., 1979. Onchocerca spp.: cryopreservation of microfilariae and subsequent development in the insect host. Experimental Parasitology, 47, 384–391.
- HARDY, J.L., HOUK, E.J., KRAMER, L.D. & REEVES, W.C., 1983. Intrinsic factors affecting vector competence of mosquitoes for arboviruses. Annual Review of Entomology, 28, 229–262.
- HARDY, J.L., SCRIVANI, R.P., LYNESS, R.N., NELSON, R.I. & ROBERTS, D.R., 1970. Ecological studies on Buttonwillow virus in Kern County, California, 1961–1968. American Journal of Tropical Medicine and Hygiene, 19, 552–563.
- HAYES, R.O., FRANCY, D.B., LAZUIK, J.S., SMITH, G.C. & JONES, R.H., 1976. Arbovirus surveillance in six states during 1972, USA. American Journal of Tropical Medicine and Hygiene, 25, 463–476.
- HENDRY, G., 1989. Midges in Scotland. Aberdeen: Aberdeen University Press. 83 pp.
- HENRARD, C. & PEEL, E., 1949. Culicoides grahamii Austen, vector of Dipetalonema streptocerca and not of Acanthocheilonema. Annales Societe Belge de Medecine Tropicale, 29, 127–143.
- HIGGINS, J.A. KLEI, T.R. & FOIL, L.D., 1988. Factors influencing the ingestion of Onchocerca cervicalis microfilariae by Culicoides variipennis. Journal of the Mosquito Control Association, 4, 242–247.
- HOLBROOK, F.R., 1982. Evaluations of three insecticides against colonized and field-collected larvae of Culicoides variipennis (Diptera: Ceratopogonidae). Journal of Economic Entomology, 75, 736–737.
- HOLBROOK, F.R., 1986. Exposure of Culicoides variipennis (Diptera: Ceratopogonidae) to hair clippings to evaluate insecticide-impregnated ear tags in cattle. Journal of Economic Entomology, 79, 1127–1129.
- HOMMEL, M. & CROFT, A.L., 1975. A trypanosomatid parasite of laboratory-bred Culicoides variipennis. Transactions of the Royal Society of Tropical Medicine and Hygiene, 69, 431.
- HOPKINS, C.A., 1952. Notes on the biology of certain Culicoides studied in the British Cameroons, West Africa, together with observations on their possible role as vectors of Acanthocheilonema perstans. Annals of Tropical Medicine and Parasitology, 46, 165–172.
- HOPKINS, C.A. & NICHOLAS, W.L., 1952. Culicoides austeni, the vector of a Acanthocheilonema perstans. Annals of Tropical Medicine and Parasitology, 46, 276–283.
- HOWARTH, F.G., 1985. Biosystematics of the Culicoides of Laos (Diptera: Ceratopogonidae). International Journal of Entomology, 27, 1–96.
- HOWELL, P.G., 1963. African horsesickness. In: Emerging Diseases of Animals. Rome: Food and Agriculture Organization of the United Nations. pp. 71–108.
- HOWELL, P.G. & COETZER, J.A.W., 1999. Serological evidence of infection of horses by viruses of the Simbu serogroup in South Africa. In: WERNERY, U., WADE, J.F., MUMFORD, J.A. & KAADEN, O.-R., (eds). Equine Infectious Diseases. VIII. Proceedings of the Eighth International Conference Dubai, 23–26 March 1998. R & W Publications (Newmarket) Limited.
- HUIZINGA, H.W. & GRANATH, W.O., 1984. Seasonal prevalence of Chandlerella quiscali (Onchocercidae: Filarioidea) in the brain of the common crackle (Quiscalus quiscala versicolor). Proceedings of the Helminthology Society of Washington, 51, 191–195.
- INGRAM, A. & MACFIE, J.W.S., 1921. West African Ceratopogonidae. Annals of Tropical Medicine and Parasitology, 15, 313–376.
- INGRAM, A. & MACFIE, J.W.S., 1923. Notes of some African Ceratopogonidae. Bulletin of Entomological Research, 14, 41–47.
- INGRAM, A. & MACFIE, J.W.S., 1925. New Ceratopogonidae from Nyasaland (Dipt.). Bulletin of Entomological Research, 1, 283–288.
- ITO, K., SHIHARA, T., MORI, T. & KITAOKA, S., 1975. Drug efficiency for the prevention of Leucocytozoon caulleryi transmitted to chickens by Culicoides. Animal Husbandry (Tokyo), 29, 543–544. (in Japanese).
- ITOUA, A. & CORNET, M., 1986. Les Cératopogonidés (Diptera) du Mayombe Congolais. III. Revue taxonomique des espèces du genera CulicoidesLatreille 1809. Cahiers del’office de la Recherche Scientifique et Technique Outre-Mer, Série Entomologie médicale et Parasitologie, 24, 233–250.
- JONES, R.H., ROUGHTON, R.D., FOSTER, M.M. & BANDO, B.M., 1977. Culicoides Journal of Wildlife Diseases, 13, 2–8.
- JUPP, P.G., MCINTOSH, B.M. & NEVILL, E.M., 1980. A survey of the mosquito and Culicoides faunas at two localities in the Karoo region of South Africa with some observations on bionomics. Onderstepoort Journal of Veterinary Research, 47, 1–6.
- KANEKO, N., INABA, Y., AKASHI, H., MIURA, Y., SHORTHOSE, J. & KURASHIGE, K., 1986. Isolation of a new bovine ephemeral fever group virus. Australian Veterinary Journal, 63, 29.
- KARABATSOS N. (ed.), 1985. International Catalogue of Arboviruses including certain other viruses of invertebrates. American Society of Tropical Medicine and Hygiene, San Antonio. pp. 1147.
- KARPOV, S.P., POPOV, V.M., SLINKINA, A.G., CHERNYSHEV, F.I. & RYAZANTSEV, M.I., 1943. Epidemiology of a transmissive outbreak of tularemia. Zhurnal Mikrobiologii, Epidemiologii, i Immunobiologii, 7(8), 24–28 (in Russian).
- KARSTAD, L.H., FLETCHER, O.K., SPALATIN, J., ROBERTS, R. & HANSON, R.P., 1957. Eastern equine encephalomyelitis virus isolated from three species of Diptera from Georgia. Science, 125, 395–396.
- KAY, B.H., BOREHAM, P.F.L., DYCE, A.L. & STANDFAST, H.A., 1978. Blood feeding patterns of biting midges (Diptera: Ceratopogonidae) at Kowanyama, Cape York Peninsula, northern Queensland. Bulletin of Entomological Research, 17, 145–149.
- KEMP, G.E., LEE, V.H., MOORE, D.L., SHOPE, R.E., CAUSEY, O.R. & MURPHY, E.A., 1973. Kotonkan virus, a new Rhabdovirus related to Mokola virus of the Rabies serogroup. American Journal of Epidemiology, 98, 43–49.
- KETTLE, D.S., 1984. Medical and Veterinary Entomology. London and Sydney: Croom Helm.
- KHAMALA, C.P.M. & KETTLE, D.S., 1971. The Culicoides Latreille (Diptera: Ceratopogonidae) of East Africa. Transactions of the Royal Entomological Society of London, 123, 1–95.
- KIEFFER, J.J., 1911. The Percy Sladen Trust Expedition to the Indian Ocean in 1905. Under the leadership of Mr. J. Stanley Gardiner. Vol. 3. No. XV. Diptera, Chironomidae der Seychellen-inseln, aus der Sammlung von Mr. H. Scott. Transactions of the Linnean Society of London (2nd Ser.) 14, 331–366.
- KIEFFER, J.J., 1913. Chironomidae et Cecidomyidae. In: alluaud, c.a. & jeannel, r., (eds). Voyage de Ch. Alluad et R. Jeannel en Afrique orientale (1911–1912). Resultats scientifiques. Diptera. (= Vol. 5), 1-43, Paris. 351 pp.
- KIEFFER, J.J., 1918. Chironomides d’Afrique et d’Asie conserves au Museum National Hongrois de Budapest. Annales Historico-Naturales Musei Nationalis Hungarici 16, 31–136.
- KIEFFER, J.J., 1921. Chironomides de l’Afrique Équatoriale. Annales de la Societe Entomologique de France, 90, 1–56.
- KINNEY, R.M. & CALISHER, C.H., 1981. Antigenic relationship among Simbu serogroup (Bunyaviridae) viruses. American Journal of Tropical Medicine and Hygiene, 30, 1307–1318.
- KITAOKA, S., 1972. Recent progress in research on chicken leucocytozoons in Japan. Abstract of the Fourteenth International Congress of Entomology, Canberra, 278.
- KLEIDER, N. & LEES, M.J., 1984. Culicoides hypersensitivity in the horse: 15 cases in southwestern British Columbia. Canadian Veterinary Journal, 25, 26–32.
- KOEKEMOER, J.J.O., PAWESKA, J.T., & VAN DIJK, A.A., 2001. Molecular analysis of the VP2-gene of African horsesickness virus isolates from field cases in South Africa. World Association of Veterinary Laboratory Diagnosticians. Tenth International Symposium of Veterinary Laboratory Diagnosticians and OIE Seminar on Biotechnology, Salsomaggiore, Parma, Italy. 4–7 July 2001. pp. 288–289.
- KRAMER, W.L., JONES, R.H., HOLBROOK, F.R., WALTON, T.E. & CALISHER, C.H., 1990. Isolations of arboviruses from Culicoides midges (Diptera: Ceratopogonidae) in Colorado during an epizootic of vesicular stomatitis New Jersey virus. Journal of Medical Entomology, 27, 487–493.
- KREMER, M., 1972. Redescription deCulicoides imicola, C. alticola et C. tropicalis Kieffer sur des exemplaires déterminer par l’auteur (Diptera, Ceratopogonidae). Bulletin du Museum National d’Historie Naturelle, 58, 645–655.
- KUROGI, H., AKIBA, K., INABA, Y. & MATUMOTO, M., 1987. Isolations of Akabane virus from the biting midge Culicoides oxystoma in Japan. Veterinary Microbiology, 15, 243–248.
- KUROGI, H., AKIBA, K. & KUBO. M., 1986. Isolations of Akabane virus from the biting midge Culicoides oxystoma in Kagoshima, Japan in 1984. Journal of Japanese Veterinary Medical Association, 39, 166–170.
- KUROGI, H., SUZUKI, T., AKASHI, H., ITO, T., INABA, Y. & MATUMOTO, M., 1989. Isolations and preliminary characterisation of an orbivirus of the Palyam serogroup from the biting midge Culicoides oxystoma in Japan. Veterinary Microbiology, 19, 1–11.
- LANDAU, I., HUMPHREY-SMITH, I., CHABAUD, A.G., MILTGEN, F., COPEMAN, B. & BOULARD, Y., 1985. Description of Hepatocystis levinei, new species, a parasite of Pteropus poliocephalus from Queensland (Australia). Annales de Parasitologie Humaine et Comparee, 60, 373–382.
- LE DUC, J.W., HOCH, A.L. & PINHEIRO, F.P., 1980. Ecology of Oropouche virus, a Simbu group arbovirus from northern South America. In: LABUDA, M. & CALISHER, C.H., (eds). New Aspects in Ecology of Arboviruses. Institute of Virology, Bratislava: 103–113.
- LEE, V.H., 1979. Isolations of viruses from field populations of Culicoides (Diptera: Ceratopogonidae) in Nigeria. Journal of Medical Entomology, 16, 76–79.
- LEE, V.H., CAUSEY, O.R. & MOORE, D.L., 1974. Bluetongue and related viruses in Ibadan, Nigeria: Isolation and preliminary identification of viruses. American Journal of Veterinary Research, 35, 1105–1108.
- LEE, Y.C., CHENG, C.H. & FUH, T.H., 1968. Studies on leucocytozoonosis of chickens. IV. The vector of L. caulleryi in Taiwan. Taiwan Journal of Veterinary Medicine and Animal Husbandry, 12, 41–47.
- LINLEY, J.R., 1979. Nutrient agar mixtures for culture of food organisms for predaceous Culicoides larvae (Diptera: Ceratopogonidae). Journal of Medical Entomology, 16, 171–172.
- LINLEY, J.R., 1985. Biting midges (Diptera: Ceratopogonidae) as vectors of nonviral animal pathogens. Journal of Medical Entomology, 22, 589–599.
- LINLEY, J.R. & DAVIES, J.B., 1971. Sandflies and tourism in Florida and the Bahamas and Caribbean area. Journal of Economic Entomology, 64, 264–278.
- LOOMIS, E.C., BUSSCHNELL, R.B. & OSBURN, B.I., 1985. Epidemiologic studies of bluetongue virus on the Tejo ranch, California: vector, host, virus relationships. In: barber, t.l. & jochim, m.m., (eds). Bluetongue and Related Orbiviruses. New York: Alan R. Liss.
- LOWRIE, R.C., EBERHARD, M.L. & ORIHEL, T.C., 1978. Development of Tetrapetalonema marmosetae to the infective stage in Culicoides hollensis and C. furens. Journal of Parasitology, 64, 1003–1007.
- LOWRIE, R.C., ORIHEL, T.C. & EBERHARD, M.L., 1982. Culicoides variipennis, a laboratory vector for the Amazon form of Mansonella ozzardi. American Journal of Tropical Medicine and Hygiene, 31, 166–167.
- LOWRIE, R.C., RACCURT, C.P., EBERHARDT, M.L. & KATZ, S.P., 1983. Assessment of Leptoconops bequaerti as a potential vector of Mansonella ozzardi in Haiti. American Journal of Tropical Medicine and Hygiene, 32, 1013–1015.
- MACFIE, J.W.S., 1924. On some Egyptian Ceratopogonidae. Bulletin of Entomological Research, 15, 61–67.
- MACFIE, J.W.S., 1947. Ceratopogonidae from the Anglo-Egyptian Sudan. Proceedings of the Royal Entomological Society of London (B), 14, 1–2.
- MACFIE, J.W.S. & INGRAM, A., 1923. Certain nurseries of insect life in West Africa. Bulletin of Entomological Research, 13, 291–294.
- MARR, J.S. & MALLOY, C.D., 1996. An epidemiologic analysis of the ten plagues of Egypt. Cladeus, 12, 7–24.
- MCINTOSH, B.M., 1980. The epidemiology of arthropod-borne viruses in southern Africa. D.Sc. Thesis. University of Pretoria.
- MEISWINKEL, R., 1987. Afrotropical Culicoides: a redescription of C. (Avaritia) kanagai Khamala & Kettle 1971, reared from elephant dung in the Kruger National Park, South Africa. Onderstepoort Journal of Veterinary Research, 54, 585–590.
- MEISWINKEL, R., 1989. Afrotropical Culicoides: a redescription of C. (Avaritia) imicola Kieffer, 1913 (Diptera: Ceratopogonidae) with description of the closely allied C. (A.) bolitinos sp. nov. reared from the dung of the African buffalo, blue wildebeest and cattle in South Africa. Onderstepoort Journal of Veterinary Research, 56, 23–39.
- MEISWINKEL, R., 1991. Afrotropical Culicoides: C. (Avaritia) miombo sp. nov., a widespread species closely allied to C. (A.) imicola Kieffer, 1913 (Diptera: Ceratopogonidae). Onderstepoort Journal of Veterinary Research, 58, 155–170.
- MEISWINKEL, R., 1992. Afrotropical Culicoides: C. (Avaritia) loxodontis sp. nov., a new member of the Imicola group (Diptera: Ceratopogonidae) associated with the African elephant in the Kruger National Park, South Africa. Onderstepoort Journal of Veterinary Research, 59, 145–159.
- MEISWINKEL, R., 1993. Onderstepoort Veterinary Institute, Onderstepoort, 0110 South Africa. Unpublished data.
- MEISWINKEL, R., 1995. Afrotropical Culicoides: biosystematics of the Imicola group, subgenus Avaritia (Diptera: Ceratopogonidae), with special reference to the epidemiology of African horse sickness. M.Sc. (Agric.) Thesis, University of Pretoria, Pretoria, RSA.
- MEISWINKEL, R., 1997. Discovery of a Culicoides imicola-free zone in South Africa: preliminary notes and potential significance. Onderstepoort Journal of Veterinary Research, 64, 81–86.
- MEISWINKEL, R., 1998. The 1996 outbreak of African horse sickness in South Africa—the entomological perspective. Archives of Virology, (Supplement 14), 69–83.
- MEISWINKEL, R. & BAYLIS, M., 1998. Morphological confirmation of the separate species status of Culicoides (Avaritia) nudipalpis Delfinado, 1961 and C. (A.) imicola Kieffer, 1913 (Diptera: Ceratopogonidae). Onderstepoort Journal of Veterinary Research, 65, 9–16.
- MEISWINKEL, R., BAYLIS, M. & LABUSCHAGNE, K., 2000. Stabling and the protection of horses from Culicoides bolitinos (Diptera: Ceratopogonidae), a recently identified vector of African horse sickness. Bulletin of Entomological Research, 90, 509–515.
- MEISWINKEL, R., BAYLIS, M., LABUSCHAGNE, K. & MELLOR, P.S., 1999. Unpublished data.
- MEISWINKEL, R. & BRAACK, L.E.O., 1994. African horsesickness epidemiology: five species of Culicoides (Diptera: Ceratopogonidae) collected live behind the ears and at the dung of the African elephant in the Kruger National Park, South Africa. Onderstepoort Journal of Veterinary Research, 61, 151–170.
- MEISWINKEL, R. & DYCE, A.L., 1989. Afrotropical Culicoides: Synhelea Kieffer, 1925, resurrected as subgenus to embrace 10 species (Diptera: Ceratopogonidae). Onderstepoort Journal of Veterinary Research, 56, 147–163.
- MEISWINKEL, R., NEVILL, E.M. & VENTER, G.J., 1994. Vectors: Culicoides spp. In: coetzer, j.a.w., thomson, g.r. & tustin, r.c., (eds). Infectious Diseases of Livestock with Special Reference to Southern Africa. Cape Town: Oxford University Press Southern Africa. pp. 68–89.
- MEISWINKEL, R. & PAWESKA, J.T., 1998. The 1998 outbreak of horse sickness in South Africa: A new Culicoides Latreille (Ceratopogonidae) vector? Abstract, Fourth International Congress of Dipterology, Keble College, Oxford, UK, 6–13 September 1998. pp. 145–146.
- MEISWINKEL, R. & PAWESKA, J.T., 1999. The 1998 outbreak of African horse sickness in the eastern Free State, South Africa: New insights into the epidemiology of the disease. Proceedings of the Ninth International Symposium of the World Association of Veterinary Laboratory Diagnosticians and OIE Biotechnology Seminar, College Station, Texas, USA. pp. 123.
- MEISWINKEL, R. & VENTER, G.J., 1996. Onderstepoort Veterinary Institute, Onderstepoort, 0110 South Africa. Unpublished data.
- MELLOR, P.S, 1973. Studies of Onchocerca cervicalis Railliet et Henry 1910. I. Onchocerca cervicalis larvae in British horses. Journal of Helminthology, 47, 97–110.
- MELLOR, P.S., 1974. Studies of Onchocerca cervicalis Railliet et Henry 1910. IV. Behaviour of the vector Culicoides nubeculosus in relation to the transmission of Onchocerca cervicalis. Journal of Helminthology, 48, 283–284.
- MELLOR, P.S., 1975. Studies of Onchocerca cervicalis Railliet et Henry 1910. V. The development of Onchocerca cervicalis larvae in the vectors. Journal of Helminthology, 49, 33–42.
- MELLOR, P.S., 1976. Infection of Culicoides nubeculosus, Culicoides riethi and Aedes aegypti with Mansonella ozzardi. Transactions of the Royal Society of Tropical Medicine and Hygiene, 70, 353.
- MELLOR, P.S., BONED, J., HAMBLIN, C. & GRAHAM, S., 1990. Isolations of African horse sickness virus from vector insects made during the 1988 epizootic in Spain. Epidemiology and Infection, 105, 447–454.
- MELLOR, P.S. & MCCAIG, J., 1974. The probable cause of ‘sweet itch’ in England. The Veterinary Record, 95, 411–415.
- MELLOR, P.S., OSBORNE, R. & JENNINGS, D.M., 1984. Isolation of bluetongue and related viruses from Culicoides spp. in Sudan. Journal of Hygiene, Cambridge, 93, 621–628.
- MELLOR, P.S. & PITZOLIS, G., 1979. Observations on breeding sites and light-trap collections of Culicoides during an outbreak of bluetongue in Cyprus. Bulletin of Entomological Research, 69, 229–234.
- MILLET, P. & BAIN, O., 1984. Une nouvelle filarea de la pie, Eufilaria kalifai n. sp. (Lemdaninae) et son développement chez Culicoides nubeculosus. Annales de Parasitologie humaine et comparee, 59, 177–178.
- MILTGEN, F. & LANDAU, I., 1982. Culicoides nubeculosus, an experimental vector of a new trypanosome from a psittaciform (Psittacula roseata): Trypanosoma bakeri, new species. Annales de Parasitologie humaine et comparee, 57, 423–428.
- MILTGEN, F., LANDAU, I., CANNING, E.U., BOORMAN, J. & KREMER, M., 1976. Hepatocystis de Malaise. III. Développement d’Hepatocystis brayi chez Culicoides nubeculosus et C. variipennis. Annales de Parasitologie humaine et comparee, 51, 299–302.
- MILTGEN, F., LANDAU, I., RATANAWORABHAN, N., & YENBUTRA, S., 1981. Parahaemoproteus desseri n. sp.; gametogonie et schizogonie chez l’hote natural, Psittacula roseata de Thailande, et sporogonie experimentale chez Culicoides nubeculosus. Annales de Parasitologie humaine et comparee, 56, 123–130.
- MIURA, Y., GOTO, Y., KUBO, M. & KONO, Y., 1988. Isolation of Chuzan virus, a new member of the Palyam subgroup of the genus Orbivirus, from cattle and Culicoides oxystoma in Japan. American Journal of Veterinary Research, 49, 2022–2025.
- MO, C.L., THOMPSON, L.H., HOMAN, J., OVIEDO, M.T., GREINER, E.C., GONZÃLES, J. & SÃENZ, M.R., 1994. Bluetongue virus isolations from vectors and ruminants in Central America and the Carribbean. American Journal of Veterinary Research, 55, 211–215.
- MOHAMMED, M.E.H. & MELLOR, P.S., 1990. Further studies on bluetongue and bluetongue-related orbiviruses in the Sudan. Epidemiology and Infection, 105, 619–632.
- MOIGNOUX, J.B., 1951. Culicoides nubeculosus (Meig.) (Diptera: Ceratopogonidae), possible intermediate host of the filaria Onchocerca reticulata Dies. in the Camargue. Comptes Rendus Academie des Sciences (Paris), 233, 102–103.
- MOIGNOUX, J.B., 1951. The Onchocerca of equidae. Acta Tropica, 9, 125–150.
- MORII, T. & KITAOKA, S., 1968. Influence of temperature on the sporogony of Akiba caulleryi in three Culicoides species. National Institute of Animal Health Quarterly (Tokyo), 8, 210–216.
- MORII, T. & KITAOKA, S., 1968. Relationship between the course of gametocyte appearance of Akiba caulleryi in the chicken and sporozoite production in Culicoides arakawae. National Institute of Animal Health Quarterly (Tokyo), 8, 204–209.
- MORII, T., KITAOKA, S. & AKIBA, K., 1965. Some investigations on the sporogony of Leucocytozoon caulleryi in laboratory-reared biting midges of four Culicoides species. National Institute of Animal Health Quarterly (Tokyo), 5, 109–110.
- MORII, T., MATSUI, T., IIJIMA, T., FUJINAGA, F., 1984. Infectivity of Leucocytozoon caulleryi sporozoites developed in vitro and in vivo. International Journal of Parasitology, 14, 135–140.
- MORII, T., NAKAMURA, K., LEE, Y.C. IIJIMA, T. & HOJI, K., 1986. Observations on the Taiwanese strain of Leucocytozoon caulleryi (Haemosporina) in chickens. Journal of Protozoology, 33, 231–234.
- MULLENS, B.A. & RUTZ, D.A., 1984. Age structure and survivorship of Culicoides variipennis (Diptera: Ceratopogonidae) in central New York State, USA. Journal of Medical Entomology, 21, 194–203.
- MULLENS, B.A., VELTEN, R.K., GERRY, A.C., BRAVERMAN, Y. & ENDRIS, R.G., 2000. Feeding and survival of Culicoides sonorensis on cattle treated with permethrin or pirimiphos-methyl. Medical and Veterinary Entomology, 14, 313–320.
- MULLER, M.J. & STANDFAST, H.A., 1986. Vectors of ephemeral fever group viruses. In: st.george, t.d., kay, b.h. & blok, j., (eds). Arbovirus Research in Australia. Proceedings Fourth Symposium, 6–9 May 1986. Brisbane. pp. 295–298.
- MUSHI, E.Z., ISA, J.F.W., CHABO, R.G., BINTA, M.G., SAKIA, R.M. & KAPAATA, R.W., 1998. Culicoides associated with dairy cows at Sebele, Gabarone, Botswana. Tropical Animal Health and Production, 30, 305–307.
- MUSUKA, G.N., MEISWINKEL, R., BAYLIS, M., KELLY, P.J. & MELLOR, P.S., 2001. Prevalence of Culicoides imicola and other species (Diptera: Ceratopogonidae) at eight sites in Zimbabwe. Journal of the South African Veterinary Association, 72, 62–63.
- NAKAMURA, R., MATSUHASHI, A., YAMASHITA, N. & YAMAMOTO, T., 1956. Studies on ‘Kasen’ of horses in Hokkaido. III. Research on the actual state of the disease (in Japanese). Japanese Journal of Veterinary Research, 4, 81–88.
- NATHAN, M.B., 1981. Transmission of the human filarial parasite Mansonella ozzardi by Culicoides phlebotomus (Diptera: Ceratopogonidae) in coastal north Trinidad. Bulletin of Entomological Research, 71, 97–106.
- NELSON, R.L. & SCRIVANI, R.P. 1972. Isolations of arboviruses from parous midges of the Culicoides variipennis complex, and parous rates in biting populations. Journal of Medical Entomology, 9, 277–281.
- NEMOVA, N.V., BULYCHEV, V.P., GORDEEVA, Z.E. & KOSTYUKOV, M.A., 1989. Isolation of Issyk-Kul virus from blood-sucking biting flies Culicoides schultzei Enderlein in southern Tadzhikistan (in Russian). Medicinskaya Parazitologiya i Parazitarnye Bolezni, 6, 25–26.
- NEVILL, E.M., 1967. Biological studies on some South African Culicoides species (Diptera: Ceratopogonidae) and the morphology of their immature stages. M.Sc. (Agric.) Thesis. University of Pretoria.
- NEVILL, E.M., 1968. A significant new breeding site of Culicoides pallidipennis Carter, Ingram and Macfie (Diptera: Ceratopogonidae). Journal of the South African Veterinary Medical Association, 39, 61.
- NEVILL, E.M., 1969. The morphology of the immature stages of some South African Culicoides species (Diptera: Ceratopogonidae). Onderstepoort Journal of Veterinary Research, 36, 265–284.
- NEVILL, E.M., 1971. Cattle and Culicoides biting midges as possible overwintering hosts of bluetongue virus. Onderstepoort Journal of Veterinary Research, 38, 65–72.
- NEVILL, E.M., 1978. The use of cattle to protect sheep from bluetongue infection. Journal of the South African Veterinary Association, 49, 129–130.
- NEVILL, E.M. & ANDERSON, DORA, 1972. Host preference of Culicoides midges (Diptera: Ceratopogonidae) in South Africa as determined by precipitin test and light trap catches. Onderstepoort Journal of Veterinary Research, 39, 147–152.
- NEVILL, E.M., ERASMUS, B.J. & VENTER, G.J., 1992. A six-year survey of viruses associated with Culicoides biting midges throughout South Africa (Diptera: Ceratopogonidae). In: walton, t.e. & osburn, b.i., (eds). Bluetongue, African Horse Sickness and Related Orbiviruses. Boca Raton, Florida: CRC Press. pp. 314–319.
- NEVILL, E.M., VENTER, G.J., EDWARDES, M., PAJOR, I.T.P., MEISWINKEL, R. & VAN GAS, J.H., 1988. Culicoides species associated with livestock in the Stellenbosch area of the western Cape Province, Republic of South Africa (Diptera: Ceratopogonidae). Onderstepoort Journal of Veterinary Research, 55, 101–106.
- NEVILL, H., 1995. Onderstepoort Veterinary Institute, Onderstepoort, 0110 South Africa. Unpublished data.
- NEVILL, H. & DYCE, A.L., 1995. Afrotropical Culicoides: Description and comparision of the pupae of seven species of the Similis supergroup (Diptera: Ceratopogonidae). Onderstepoort Journal of Veterinary Research, 61, 85–106.
- NICHOLAS, W.L., 1953. The bionomics of Culicoides austeni, vector of Acanthocheilonema perstans in the rain-forest of the British Cameroons and other species which may be vectors in the same area. Annals of Tropical Medicine and Parasitology, 47, 187–206.
- NICHOLAS, W.L & KERSHAW, W.E., 1954. Studies on the intake of mocrofilariae by their vectors, their survival, and their effect on the survival of their vectors. III. The intake of the microfilariae of Acanthocheilonema perstans by Culicoides austeni and Culicoides grahamii. Annals of Tropical Medicine and Parasitology, 48, 201–205.
- NIESCHULZ, O. & DU TOIT, R.M., 1937. Investigations into the transmission of horsesickness at Onderstepoort during the season 1932–1933. Onderstepoort Journal of Veterinary Science and Animal Industry, 8, 213–269.
- OLSUFIEV, N.G. & RUDNEV, G.P., (eds.) 1960. Tularemia (in Russian). Medgiz, Moskva, 459 pp.
- ORTEGA, M.D., MELLOR, P.S., RAWLINGS, P. & PRO, M.J., 1998. The seasonal and geographic distribution of Culicoides imicola, C. pulicaris group andC. obsoletus group biting midges in central and southern Spain. Archives of Virology (Supplement 14), 85–91.
- PAJOR, I.T.P., 1983-1985. Onderstepoort Veterinary Institute. Onderstepoort, 0110 South Africa. Unpublished data.
- PAJOR, I.T.P., 1987. A collapsible, semi-automatic tent-type, emergence trap, suitable for sampling Culicoides (Diptera: Ceratopogonidae) from a wide range of habitats. Onderstepoort Journal of Veterinary Research, 54, 99–101.
- PARSONSON, I.M. & SNOWDON, W.A., 1985. Bluetongue, epizootic haemorrhagic disease of deer and related viruses: current situation in Australia. In : BARBER, T.L. & JOCHIM, M.M., (eds).Bluetongue and Related Orbiviruses. New York: Alan R. Liss. pp. 27–35.
- PATON, T., 1863. The ‘horse sickness’ of the Cape of Good Hope. Veterinarian, 36, 489–494.
- PAWESKA, J.T., 2001. Onderstepoort Veterinary Institute. Onderstepoort, 0110 South Africa. Unpublished data.
- PAWESKA, J.T., GERDES, G.H. & MEISWINKEL, R., 1999. Onderstepoort Veterinary Institute. Onderstepoort, 0110 South Africa. Unpublished data.
- PAWESKA, J.T. & VENTER, G.J., 1999. The effect of temperature and incubation period on the replication of bluetongue virus in Culicoides imicola and C. bolitinos (Diptera: Ceratopogonidae). Proceedings of the Twelth Congress of the Entomological Society of Southern Africa, Potchefstroom, 12–15 July 1999. p. 91.
- PAWESKA, J.T., VENTER, G.J. & MELLOR, P.S., 2002. Vector competence of South African Culicoides species for bluetongue virus serotype 1 (BTV-1) with special reference to the effect of temperature on the rate of virus replication in C. imicola and C. bolitinos. Medical and Veterinary Entomology, 16, 10–21.
- PHELPS, R.J., BLACKBURN, N.K. & SEARLE, L., 1982. Culicoides (Diptera: Ceratopogonidae) catches and virus isolations from them in the Mukwadzi Valley, Zimbabwe. Journal of the Entomological Society of Southern Africa, 45, 195–200.
- RAHMAT, S.M.S. & PARAMESWARAN, S., 1991. Isolation and maintenance of Leucocytozoon caulleryi: VRI strain. Malaysian Veterinary Journal, 3, 65–69.
- RAO, V.D. & ROY, P., 1983. Genetic variation of bluetongue virus serotype 11 isolated from host (sheep) and vector (Culicoides variipennis) at the same site. American Journal of Veterinary Research, 44, 911–914.
- RAWLINGS P., CAPELA, R., PRO, M.J., ORTEGA, M.D., PENA, I., RUBIO, C., GASCA, A. & MELLOR, P.S., 1998. The relationship between climate and the distribution of Culicoides imicola in Iberia. Archives of Virology (Supplement 14), 93–102.
- REEVES, W.C., SCRIVANI, R.P., NELSON, R.P., HARDY, J.L., ROBERTS, D.R. & NELSON, R.L., 1970. Buttonwillow virus, a new arbovirus isolated from mammals and Culicoides midges in Kern County, California. American Journal of Tropical Medicine and Hygiene, 19, 544–551.
- RODRIGEUZ, J.L., HOOGHUIS, H. & CASTAÑO, MA., 1992. African horse sickness in Spain. Veterinary Microbiology, 33, 129–142.
- SEBASTIANI, F., MEISWINKEL, R., GOMULSKI, L.M., GUGLIELMINO, C.R., MELLOR, P.S., MALACRIDA, A.R. & GASPERI, G., 2001. Molecular differentiation of the old world Culicoides imicola species complex (Diptera: Ceratopogonidae), inferred using random amplified polymorphic DNA markers. Molecular Ecology, 10, 1773–1786.
- SELLERS, R.F., PEDGLEY, D.E. & TUCKER, M.R., 1977. Possible spread of African horse sickness on the wind. Journal of Hygiene, Cambridge, 79, 279–297.
- SENDOW, I., SUKARSIH, E., ERASMUS, B.J. & DANIELS, P.W., 1993. Isolation of bluetongue virus serotype 21 from Culicoides spp. in Indonesia. Veterinary Microbiology, 36, 349–353.
- SHARP, N.A.D., 1927. Development of Microfilaria perstans in Culcoides grahamii, a preliminary note. Transactions of the Royal Society of Tropical Medicine and Hygiene, 21, 70.
- SHARP, N.A.D., 1928. Filaria perstans, its development in Culicoides austeni. Transactions of the Royal Society of Tropical Medicine and Hygiene, 21, 371–396.
- SHIMOSAWA, T., AMANO, A., SUZUKI, K., MUROGA, N., KURIHARA, T., INOUE, I., NOSHINO, M. & OHMI, T., 1986. Epizootiological survey of chicken leucocytozoonosis and Culicoides arakawae. Bulletin of the College of Agriculture and Veterinary Medicine, Nihon University, 43, 202–209.
- SIBLEY, L.D. & WERNER, J.K., 1984. Susceptibility of peking (Anas platyrhynchos) and muscovy (Cairina moschata) ducks to Haemoproteus nettionis. Journal of Wildlife Diseases, 20, 108–133.
- SKVORTSOVA, T.M., GROMASHEVSKII, V.L., SIDORVA, G.A., KHUTORETSKAYA, N.U., ARISTOVA, V.A., KONDRASHINA, N.G., POLYAKOVA, A.N., MURADOV, SH.M., BELOUSOV, E.N. & KURCHENKO, F., 1982. Results of a virological survey of the arthropod vectors on the territory of Turkmenia. Ekologia Virusov (Moscow), 192, 139–144.
- STANDFAST, H.A. & DYCE, A.L., 1982. Isolation of Thimiri virus from Culicoides histrio (Diptera: Ceratopogonidae) collected in northern Australia. Journal of Medical Entomology, 19, 212.
- STANDFAST, H.A., DYCE, A.L., ST. GEORGE, T.D., MULLER, M.J., DOHERTY, R.L., CHARLEY, J.G. & FILIPPICH, C., 1984. Isolation of arboviruses from insects collected at Beatrice Hill, Northern Territory of Australia, 1974–1976. Australian Journal of Biological Science, 37, 351–366.
- STANDFAST, H.A., MULLER, M.J. & WILSON, D.D., 1985. Mortality of Culicoides brevitarsis fed on cattle treated with Ivermectin. In: BARBER,T.L. & JOCHIM, M.M., (eds). Bluetongue and Related Orbiviruses. New York: Alan R. Liss. pp. 611–616.
- STANDFAST, H.A., ST. GEORGE, T.D., DOHERTY, R.L. & DYCE, A.L., 1975. Beatrice Hill project—virus isolations. Report of the CSIRO Division of Animal Health (Canberra), 62, 82.
- STEWARD, J.S., 1933. Onchocerca cervicalis (Railliet et Henry, 1910) and its development in Culicoides nubeculosus Mg. Report of the Institute of Animal Pathology, University of Cambridge, 3, 272–284.
- ST. GEORGE, T.D., 1985. The search for bluetongue viruses in Australia. In: BARBER, T.L. & JOCHIM, M.M., (eds).Bluetongue and Related Orbiviruses. New York: Alan R. Liss. pp. 295–305.
- ST. GEORGE, T.D., 1994. Bovine ephemeral fever. In: COETZER, J.A.W., THOMSON, G.R. & TUSTIN, R.C., (eds). Infectious Diseases of Livestock with Special Reference to Southern Africa. Cape Town: Oxford University Press Southern Africa. pp. 553–562.
- ST. GEORGE, T.D., CYBINSKI, D.H., FILIPPICHI, C. & CARLEY, J.G., 1979. The isolation of three Simbu group viruses new to Australia. Australian Journal of Experimental Biology and Medical Science, 57, 581–582.
- ST. GEORGE, T.D., CYBINSKI, D.H. & STANDFAST, H.A., 1982. The continued search for bluetongue related viruses in Australia. In: ST. GEORGE, T.D. & KAY, B.H. (eds). Arbovirus Research in Australia. Proceedings of the Third Symposium, 15–17 February 1982. pp. 188–193.
- ST. GEORGE, T.D., CYBINSKI, D.H., STANDFAST, H.A., GARD, G.P. & DELLA-PORTA, A.J. 1983. The isolation of five different viruses of the epizootic haemorrhagic disease of deer serogroup. Australian Veterinary Journal, 60, 216–217.
- ST. GEORGE, T.D. & MULLER, M.J., 1984. The isolation of a bluetongue virus from Culicoides brevitarsis. Australian Veterinary Journal, 61, 95.
- ST. GEORGE, T.D., STANDFAST, H.A. & CYBINSKI, D.H., 1978. Isolation of Akabane virus from sentinal cattle and Culicoides brevitarsis. Australian Veterinary Journal, 54, 558–561.
- ST. GEORGE, T.D., STANDFAST, H.A., CYBINSKI, D.H., DYCE, A.L., MULLER, H.J., DOHERTY, R.L., CARLEY, J.G., FILIPPICH, C. & FRAZIER, C.L., 1978. The isolation of a bluetongue virus from Culicoides collected in the Northern territory of Australia. Australian Veterinary Journal, 3, 153–154.
- ST. GEORGE, T.D., STANDFAST, H.A., CYBINSKI, D.H., FILIPPICH, C. & CARLEY, J.G., 1980. Peaton virus a new Simbu group arbovirus isolated from cattle and Culicoides brevitarsis in Australia. Australian Journal of Biological Science, 33, 235–244.
- ST. GEORGE, T.D., STANDFAST, H.A., MULLER, H.J., ZAKRZEWSKI, H., CYBINSKI, D.H. & GIBSON, D.S., 1978. Isolation of arboviruses from cattle and insects at sentinel sites in Queensland, Australia, 1979–1985. Arbovirus Information Exchange, June, 159–167.
- SUSWILLO, R.R., NELSON, G.S., MULLER, R., MCGREEVY, P.B., DUKE, B.O.L. & DENHAM, D.A., 1977. Attempts to infect jirds (Meriones unguiculatus) with Wuchereria bancrofti, Onchocerca volvulus, Loa loa and Mansonella ozzardi. Journal of Helmintology, 51, 132–134.
- SWANEPOEL, R., 1994. Classification, epidemiology and control of arthropod-borne viruses. In: COETZER, J.A.W., THOMSON, G.R. & TUSTIN, R.C., (eds). Infectious Diseases of Livestock with Special Reference to Southern Africa. Cape Town: Oxford University Press Southern Africa. pp. 103– 120.
- SWANEPOEL, R. & BLACKBURN, N.K., 1976. A new member of the Palyam serogroup of orbiviruses. The Veterinary Record, 99, 360.
- TABACHNICK, W.J., 1991. Genetic control of oral susceptibility of Culicoides variipennis with bluetongue virus. American Journal of Tropical Medicine and Hygiene, 45, 666–671.
- TAKAHASHI, K., OYA, A., OKADA, T., MATSUO, R., KUMA, M. & NOGUCHI, H., 1968. Aino virus, a new member of the Simbu group of arboviruses from mosquitoes in Japan. Japanese Journal of Medical Science and Biology, 21, 95–101.
- TAKAMATSU, H., FUJISAKI, K., KITAOKA, S., ISHI, T. & FUJITA, J., 1984. Cultivation of Leucocytozoon caulleryi: purification method of sporozoites for inoculum in ovo. Bulletin of the Nippon Veterinary and Zootechnical College, 33, 71–75.
- TAKAOKA, H., AOKI, C., BAIN, O., OGATA, K. & BABA, M., 1995. Investigation of Culicoides (Diptera: Ceratopogonidae) in relation to the transmission of bovine onchocerca and other filariae in central Kyushu, Japan. Parasite —Journal soc. Franc. Parasitology, 2, 367–371.
- TAYLOR, L.R., 1962. The absolute efficiency of insect suction traps. Annals of Applied Biology, 50, 405–421.
- THEILER, A., 1921. African Horse Sickness (Pestis Equorum). Science Bulletin. No 19. Department of Agriculture Union of South Africa. pp. 1–32.
- THEODORIDIS, A., NEVILL, E.M., ELS, H.J. & BOSHOFF, S.T., 1979. Viruses isolated from Culicoides midges in South Africa during unsuccessful attempts to isolate bovine ephemeral fever virus. Onderstepoort Journal of Veterinary Research, 46, 191–198.
- TIDWELL, M.A. & TIDWELL, M.A., 1982. Development of Mansonella ozzardi in Simulium amazonicum, Similium argentiscutum and Culicoides insinuatus from Amazonas, Colombia. American Journal of Tropical Medicine and Hygiene, 31, 1137–1141.
- UNKEL, M. 1984. Epidemiological studies of summer eczema in Iceland ponies. Praktische Tierarzt, 65, 659–663.
- VAN AMSTEL, S., 1990. University of Pretoria, Onderstepoort 0110, South Africa. Unpublished data.
- VAN ARK, H. & MEISWINKEL, R., 1992. Subsampling of large light trap catches of Culicoides (Diptera: Ceratopogonidae). Onderstepoort Journal of Veterinary Research, 59, 183–189.
- VARGAS, L., 1969. Culicoides as transmitters of diseases. Gaceta Medica de Mexico, 99, 782–787.
- VENTER, G.J., GRAHAM, S.D. & HAMBLIN, C. 2000. African horse sickness epidemiology: vector competence of South African Culicoides species for African horse sickness virus serotypes 3, 5 and 8. Medical and Veterinary Entomology, 14, 245–250.
- VENTER, G.J., GROENEWALD, D.M., PAWESKA, J.T., VENTER, E.H. & HOWELL, P.G., 1999. Vector competence of selected South African Culicoides species for the Bryanston serotype of equine encephalosis virus. Medical and Veterinary Entomology, 13, 393–400.
- VENTER, G.J., HILL, ELAINE, PAJOR, I.T.P. & NEVILL, E.M., 1991. The use of a membrane feeding technique to determine the infection rate of Culicoides imicola (Diptera: Ceratopogonidae) for two bluetongue virus serotypes in South Africa. Onderstepoort Journal of Veterinary Research, 58, 5–9.
- VENTER, G.J. & MEISWINKEL, R., 1994. The virtual absence of Culicoides imicola (Diptera: Ceratopogonidae) in a light-trap survey of the colder, high-lying area of the eastern Orange Free State, South Africa, and implications for the transmission of arboviruses.Onderstepoort Journal of Veterinary Research, 61, 327–340.
- VENTER G.J., MEISWINKEL, R., NEVILL, E.M. & EDWARDES, M., 1996. Culicoides (Diptera: Ceratopogonidae) associated with livestock in the Onderstepoort area, Gauteng Province, South Africa, as determined by light-trap collections. Onderstepoort Journal of Veterinary Research, 63, 315–325.
- VENTER, G.J., NEVILL, E.M. & VAN DER LINDE T.C. DE K., 1996. Geographical distribution and relative abundance of stock-associated Culicoides species (Diptera: Ceratopogonidae) in southern Africa, in relation to their potential as viral vectors. Onderstepoort Journal of Veterinary Research, 63, 25–38.
- VENTER, G.J., NEVILL, E.M. & VAN DER LINDE T.C. DE K., 1997. Seasonal abundance and parity of stock-associated Culicoides species (Diptera: Ceratopogonidae) in different climatic regions in southern Africa in relation to their viral vector potential. Onderstepoort Journal of Veterinary Research, 64, 259–271.
- VENTER, G.J. & PAWESKA, J.T., 1999. Bluetongue, a multi-vector disease in South Africa. Proceedings of the 12th Congress of the Entomological Society of Southern Africa. Potchefstroom, 12–15 July 1999. p. 130.
- VENTER, G.J., PAWESKA, J.T. VAN DIJK A.A., MELLOR, P.S. & TABACHNICK, W.J., 1998. Vector competence of Culicoides bolitinos and C. imicola (Diptera: Ceratopogonidae) for South African bluetongue virus serotypes 1, 3 and 4. Medical and Veterinary Entomology, 12, 101–108.
- VENTER, G.J. & SWEATMAN, G.K., 1989. Seasonal abundance and parity of Culicoides biting midges associated with livestock at Roma, Lesotho (Diptera: Ceratopogonidae). Onderstepoort Journal of Veterinary Research, 56, 173–177.
- WALKER, A.R., 1977. Adult lifespan and reproductive status of Culicoides (Diptera: Ceratopogonidae) in Kenya, with reference to virus transmission. Bulletin of Entomological Research
- WALKER, A.R. & BOREHAM, P.F.L., 1976. Blood feeding of Culicoides (Diptera: Ceratopogonidae) in Kenya in relation to the epidemiology of bluetongue and ephemeral fever. Bulletin of Entomological Research, 66, 181–188.
- WALKER, A.R. & DAVIES, F.G., 1971. A preliminary survey of the epidemiology of bluetongue in Kenya. Journal of Hygiene, Cambridge, 69, 47–60.
- WALTON, T.W., WEBB, P.A., KRAMER, W.L., SMITH, G.C., DAVIS, C., HOLBROOK, F.R., MOORE, C.G., SCHIEFER, T.J., JONES, R.H. & JANNEY, G.C., 1987. Epizootic vesicular stomatitis in Colorado (USA) 1982: epidemiologic and entomologic studies. American Journal of Tropical Medicine and Hygiene, 36, 166–176.
- WEINMANN, J.R. MURPHY, K., ANDERSON, J.R., DEMARTINI, J.C., LONGHURST, W.M. & CONNOLLY, G., 1979. Seasonal prevalence, pathology and transmission of the quail heartworm, Splendidofilaria californiensis, in northern California, U.S.A. (Nematoda: Filaroidea). Canadian Journal of Zoology, 57, 1871–1877.
- WELLBY, M.P., BAYLIS, M., RAWLINGS, P. & MELLOR, P.S., 1996. Effects of temperature on survival and rate of virogenesis of African horse sickness virus in Culicoides variipennis sonorensis (Diptera: Ceratopogonidae) and its significance in relation to the epidemiology of disease. Bulletin of Entomological Research, 86, 715–720.
- WETZEL, H., NEVILL, E.M. & ERASMUS, B.J., 1970. Studies on the transmission of African horsesickness. Onderstepoort Journal of Veterinary Research, 37, 165–168.
- WHISTLER, T. & SWANEPOEL, R., 1988. Characterization of potentially foetotropic Palyam serogroup orbiviruses isolated in Zimbabwe. Journal of General Virology, 69, 2221–2227.
- WHISTLER, T., SWANEPOEL, R. & ERASMUS, B.J., 1989. Characterization of Palyam serogroup orbiviruses isolated in South Africa and serologic evidence for their widespread distribution in the country. Epidemiological Information, 102, 317–324.
- WIESERSCHIMPF, L., WILSON, W.C., FRENCH, D.D., BAHAM, A. & FOIL, L.D., 1993. Bluetongue virus in sheep and cattle and Culicoides variipennis and C. stellifer (Diptera: Ceratopogonidae) in Louisiana. Journal of Medical Entomology, 30, 719–724.
- WILLIAMS, R.E., HALL, R.D. BROCE, A.B. & SCHOLL, P.J., 1985. Livestock Entomology. New York: J. Wiley. 335 pp.
- WITTMANN, E.J., MELLOR, P.S. & BAYLIS, M., 2001. Using climate data to map the potential distribution of Culicoides imicola (Diptera: Ceratopogonidae) in Europe. Revue scienfique et technique, Office International des Epizooties, 20, 731–740.
- YAMASHITA, J., KITAMURA, U. & NAKAMURA, R., 1975. Studies on ‘Kasen’ of horses in Hokkaido IV. Researches on the punkies in Hokkaido with description of a new species. Japanese Journal of Veterinary Research, 5, 89–96.
- YATES, J,A., LOWRIE, R.C. & EBERHARD, M.L., 1982. Development of Tetrapetalonema llewellyni to the infective stage in Culicoides hollensis. Journal of Parasitology, 68, 293–352.
- ZAKREWSKI, H. & CYBINSKI, D.H., 1984. Isolation of Kimberley virus, a rhabdovirus, from Culicoides brevitarsis. Australian Journal of Experimental Biology and Medical Science, 62, 779–780.