- Infectious Diseases of Livestock
- Part 2
- Diseases caused by Akabane and related Simbu-group viruses
- GENERAL INTRODUCTION: PARAMYXOVIRIDAE AND PNEUMOVIRIDAE
- Rinderpest
- Peste des petits ruminants
- Parainfluenza type 3 infection
- Bovine respiratory syncytial virus infection
- Hendra virus infection
- Paramyxovirus-induced reproductive failure and congenital defects in pigs
- Nipah virus disease
- GENERAL INTRODUCTION: CALICIVIRIDAE AND ASTROVIRIDAE
- Vesicular exanthema
- Enteric caliciviruses of pigs and cattle
- GENERAL INTRODUCTION: RETROVIRIDAE
- Enzootic bovine leukosis
- Jaagsiekte
- Visna-maedi
- Caprine arthritis-encephalitis
- Equine infectious anaemia
- GENERAL INTRODUCTION: PAPILLOMAVIRIDAE
- Papillomavirus infection of ruminants
- Papillomavirus infection of equids
- GENERAL INTRODUCTION: ORTHOMYXOVIRIDAE
- Equine influenza
- Swine influenza
- GENERAL INTRODUCTION: CORONAVIRIDAE
- Porcine transmissible gastroenteritis
- Porcine respiratory coronavirus infection
- Porcine epidemic diarrhoea
- Porcine haemagglutinating encephalomyelitis virus infection
- Porcine deltacoronavirus infection
- Bovine coronavirus infection
- Ovine coronavirus infection
- Equine coronavirus infection
- GENERAL INTRODUCTION: PARVOVIRIDAE
- Porcine parvovirus infection
- Bovine parvovirus infection
- GENERAL INTRODUCTION: ADENOVIRIDAE
- Adenovirus infections
- GENERAL INTRODUCTION: HERPESVIRIDAE
- Equid herpesvirus 1 and equid herpesvirus 4 infections
- Equid gammaherpesvirus 2 and equid gammaherpesvirus 5 infections
- Equine coital exanthema
- Infectious bovine rhinotracheitis/infectious pustular vulvovaginitis and infectious pustular balanoposthitis
- Bovine alphaherpesvirus 2 infections
- Malignant catarrhal fever
- Pseudorabies
- Suid herpesvirus 2 infection
- GENERAL INTRODUCTION: ARTERIVIRIDAE
- Equine viral arteritis
- Porcine reproductive and respiratory syndrome
- GENERAL INTRODUCTION: FLAVIVIRIDAE
- Bovine viral diarrhoea and mucosal disease
- Border disease
- Hog cholera
- Wesselsbron disease
- Louping ill
- West nile virus infection
- GENERAL INTRODUCTION: TOGAVIRIDAE
- Equine encephalitides caused by alphaviruses in the Western Hemisphere
- Old World alphavirus infections in animals
- Getah virus infection
- GENERAL INTRODUCTION: BUNYAVIRIDAE
- Diseases caused by Akabane and related Simbu-group viruses
- Rift Valley fever
- Nairobi sheep disease
- Crimean-Congo haemorrhagic fever
- GENERAL INTRODUCTION: ASFARVIRIDAE
- African swine fever
- GENERAL INTRODUCTION: RHABDOVIRIDAE
- Rabies
- Bovine ephemeral fever
- Vesicular stomatitis and other vesiculovirus infections
- GENERAL INTRODUCTION: REOVIRIDAE
- Bluetongue
- Ibaraki disease in cattle
- Epizootic haemorrhagic disease
- African horse sickness
- Equine encephalosis
- Palyam serogroup orbivirus infections
- Rotavirus infections
- GENERAL INTRODUCTION: POXVIRIDAE
- Lumpy skin disease
- Sheeppox and goatpox
- Orf
- Ulcerative dermatosis
- Bovine papular stomatitis
- Pseudocowpox
- Swinepox
- Cowpox
- Horsepox
- Camelpox
- Buffalopox
- GENERAL INTRODUCTION: PICORNAVIRIDAE
- Teschen, Talfan and reproductive diseases caused by porcine enteroviruses
- Encephalomyocarditis virus infection
- Swine vesicular disease
- Equine picornavirus infection
- Bovine rhinovirus infection
- Foot-and-mouth disease
- GENERAL INTRODUCTION: BORNAVIRIDAE
- Borna disease
- GENERAL INTRODUCTION: CIRCOVIRIDAE AND ANELLOVIRIDAE
- Post-weaning multi-systemic wasting syndrome in swine
- GENERAL INTRODUCTION: PRION DISEASES
- Scrapie
- Bovine spongiform encephalopathy
- Transmissible spongiform encephalopathies related to bovine spongiform encephalopathy in other domestic and captive wild species
Diseases caused by Akabane and related Simbu-group viruses
This content is distributed under the following licence: Attribution-NonCommercial CC BY-NC  View Creative Commons Licence details here
View Creative Commons Licence details here
NJ Maclachlan and M-L Penrith (Editors). PD Kirkland and TD St George, Diseases caused by Akabane and related Simbu-group viruses, 2018.
Diseases caused by Akabane and related Simbu-group viruses
Previous authors: T D ST GEORGE AND P D KIRKLAND
Current authors:
P D KIRKLAND - Senior Principal Researcher Scientist, Manager Virology Laboratory, BVSc, PhD, FAMS, Elizabeth Macarthur Agriculture Institute, Woodbrigde Road, Menangle, New South Wales, 2568, Australia
T D ST GEORGE - Retired from Commonwealth Scientific & Industrial Research Organisation, DVM, MVSc, BVSc, Virus Consultants International, 44/260 Cliveden Avenue, Corinda, Queensland, 4075, Australia
Introduction
Many viruses in the Simbu-group of the genus Orthobunyavirus family Peribunyaviridae, order Bunyavirales1 have not been identified as pathogens but those that are pathogenic usually cause congenital defects after infection of a susceptible pregnant small stock and cattle. The defects are principally arthrogryposis (AG) and hydranencephaly (HE) and can be associated with abortion in cattle, sheep and goats. These viruses usually produce clinically inapparent infections in non-pregnant animals. However, in pregnant cattle, sheep and goats, if the viruses reach the foetus at a critical stage of development, marked teratology, particularly of the central nervous system, may result. The defects are usually only apparent some months later with the birth of affected calves and lambs. Some Simbu-group viruses also occasionally cause post-natal encephalitis in a range of mammalian species. Antibodies to Akabane and related arboviruses are found in cattle, sheep, goats and wild and other domestic animals in Africa, Middle East and the Asian-Pacific region. In 2011 Schmallenberg virus, a previously unrecognised Simbu virus, has emerged and spread throughout most of Europe.30
The name ‘Akabane disease’ has been used,35 but it is not apt as Akabane virus is only one member of the Simbu-group, several of which may produce similar defects, including the recently recognised Schmallenberg virus. Akabane virus is nevertheless by far the best studied and probably most pathogenic member of the group.
The relationship between Akabane virus and congenital AG/HE was first recognized in Japan between 1972 and 1975.35 The studies in Japan and Australia, which in 1974 confirmed the link between infection with Akabane virus and AG/HE, have been reviewed.35, 63 Disease caused by Simbu-group viruses in Africa is poorly defined, though AG/HE has been observed in cattle in KwaZulu-Natal, South Africa, and in sheep in Zimbabwe.69
Although the association of Akabane and related viruses with epidemics of AG/HE is comparatively recent (1974), the syndrome was recognized much earlier. There are reliable records of its occurrence in Australia in the early 1930s29 when it was attributed to plant poisoning, in Japan in 194935 and in Israel in 1969/70.50
Other viruses of the Simbu-group which have been associated with similar disease are Aino,12 Peaton, Schmallenberg,6 Shamonda31 Shuni81 and Tinaroo, although clinical cases in the field due to Aino are not common and are rare with Peaton52 and Tinaroo.43 Schmallenberg virus has been responsible for an extensive outbreak of congenital defects in cattle, sheep and goats following its emergence in fully susceptible livestock populations in western Europe in 2011.6, 17 Shuni virus has been associated with encephalitis in horses and a range of wildlife species in South Africa81 and congenital defects in ruminants in Israel.22 Cache Valley virus, which belongs to a separate serogroup within the family Peribunyaviridae, is widely distributed in North America and has been reported to produce AG/HE in sheep in Texas.19
Aetiology
Viruses within the Simbu-group, one of the 36 antigenic clusters within the family Peribunyaviridae, have spherical, enveloped virions, 90 to 100 nm in diameter. They have tripartite segmented single-stranded RNA and are readily inactivated by chloroform, ether and trypsin. Akabane virus degrades moderately quickly at 37 °C,35 remains viable in blood samples kept at 4 °C for several months, and can be stored indefinitely at −70 °C or lower. For more information on the properties of the viruses which belong to the Peribunyaviridae, see General Introduction: Bunyaviridae and Rift Valley fever.
Epidemiology
Most of Africa, Asia (excluding Russia), and Australia may be regarded as endemic for Akabane virus and, in all probability, many of its antigenic relatives. Schmallenberg virus is the only Simbu virus that has been identified in Europe and England. The American continent (except for the presence of Cache Valley virus of the Bunyamwera serogroup), Papua-New Guinea and the island countries of the Pacific are believed to be free of infection with Simbu-group bunyaviruses.74 However, the distribution of Akabane and related viruses within each country is affected by the distribution, seasonal activity and abundance of insect vectors.
The isolation of Akabane virus from mosquitoes62 and the biting midge, Culicoides brevitarsis,18 preceded its incrimination as a cause of disease in sheep or cattle and was a necessary step in unravelling the aetiology. Despite the association between Akabane virus and biting arthropods, the virus has not yet been transmitted experimentally to any vertebrate by any species of insect. The virus has, however, been shown to multiply in experimentally infected C. brevitarsis and to reach the salivary glands in 10 days.59 Transovarial transmission was not demonstrated in a study involving 11 600 C. brevitarsis, indicating that transovarial transmission is not a frequent event if it occurs at all.3 The effects of local climate on the relative densities of Culicoides spp. and on the transmission of Akabane virus have been described.60 Since its recognition in Europe in 2011, extensive research of the biology and epidemiology of Schmallenberg virus infection has also been undertaken.6, 17, 78
The majority of Simbu-group virus isolations in Africa were made in the course of arbovirus surveys and little is known of their veterinary significance. Akabane virus has been isolated from mosquitoes in Kenya56 and Culicoides midges in Zimbabwe.48 It is not known whether the virus occurs in South Africa.5 Simbu virus, originally isolated from mosquitoes in South Africa,54, 83, 85 has since been isolated from mosquitoes in Cameroon, Senegal and the Central African Republic.36 Ingwavuma virus, originally isolated from the viscera of a weaver bird and from mosquitoes in South Africa,53, 55 has been isolated from birds and mosquitoes in the Central African Republic and Nigeria.36 Sabo, Sango, Shamonda and Shuni viruses were all originally isolated from cattle or goat sera, and subsequently from midges,79 in the course of surveys in Nigeria.9, 36 Sango virus has also been isolated from mosquitoes in Kenya36 and Shuni virus from mosquitoes in South Africa.54 A serological survey of horses in various regions in South Africa failed to detected antibodies to Simbu, Ingwavuma or Sathuperi viruses, a few animals had titres to Akabane and Shuni viruses, and antibody titres to Shamonda and Sabo viruses were prodominant.32 A serological study of cattle in Tanzania51 showed a similar distribution of a number of Simbu viruses, including Sabo, Sathuperi and Shamonda viruses. Interestingly, results suggest that Aino, Douglas, Peaton and Tinaroo viruses may also be present. None of these viruses had been detected previously in Africa.
Other than in Japan, Indonesia, Malaysia and Australia, where serological surveys have demonstrated their presence, information on infection with closely related viruses (Aino, Peaton, Tinaroo and Douglas viruses) is patchy. In Japan, Sathuperi and Shamonda viruses have been identified31 while Akabane virus infection has been found in pigs.86
Virus identification and characterization may be complicated by the occurrence of reassortant viruses or dual infections.13
Akabane virus has been isolated from two species of mosquitoes and Culicoides oxystoma in Japan46 and from C. brevitarsis and C. wadai in Australia.74 In Australia, the principal Culicoides vector species are dependent on cattle dung for breeding sites and their highest density is in areas of moderate to high rainfall. In Africa, the probable vectors are not so restricted and can breed in pastures rich in organic matter (see Vectors: Culicoides spp.). In Europe, Schmallenberg virus has been detected in several Culicoides species.82
The presence of infection in wildlife species complicates the epidemiology in Africa — in contrast to Australia where marsupials are not infected and Asian countries where there are few susceptible wildlife species. Neutralizing antibodies have been found in a wide range of African species including several antelope species, elephants, giraffe, impala, kudu, warthog and wildebeest.2, 15 In Europe, antibodies to Schmallenberg virus have been detected in a wide range of wildlife species including bison, roe deer, llama and wild boar.58 No disease has been linked to the infection of wildlife with Akabane virus and related Simbu-group viruses, but most new-born animals with congenital defects would be taken by predators.
In Australia the relative simplicity of the vector system and effective surveillance using sentinel cattle has resulted in the ability to forecast epidemics of AG/HE in the most severely affected area.42 Major epidemics of AG/HE disease in Japan, involving 30 000 calves, and in Australia, involving approximately 3 000 to 5 000 calves, have been described.26, 27, 34 Waves of abortion were detected in both Australia and Japan about two months before the birth of affected calves at full term.35, 70 It has been estimated that in Australia there is an ongoing economic loss within the tropical endemic areas with at least a quarter of a million cows at risk each year.77 In Europe, when Schmallenberg virus entered an extremely large, naive livestock population, a large number of cases were reported in cattle, goats and sheep but the within herd prevalence was mostly low compared to what is seen with Akabane virus. As a result of having a large number of foetuses at critical stages of gestation, higher losses were sometimes seen when synchronised breeding flocks of ewes were infected.
Pathogenesis
The pathogenesis of disease caused by Akabane virus has been reviewed.35, 38, 67, 74 Akabane virus is present in the blood of naturally infected cattle for three to four days.75 Using sentinel cattle herds to estimate the time of transmission, it has been possible to accurately relate the time of infection to stage of pregnancy and to anticipate the spectrum of tissue damage in foetuses.16, 41, 48 The lesions characteristic of natural disease have been reproduced by infecting pregnant cattle, sheep and goats experimentally with Akabane virus and by allowing them to progress to term.45, 49, 65
The relationship between infection and developmental stage of the foetus determines whether foetal death and abortion, or a range of teratogenic defects will result. During pregnancy, the virus crosses from the maternal to the foetal circulation, infecting the placenta in the process.68 The virus then multiplies in the rapidly dividing cells of the foetus, especially in the brain and spinal cord.63 Whether the resultant damage following infection is entirely due to damage to the foetal cells, limiting capacity to divide and develop into mature tissues, or whether it is partially due to necrosis of formed tissues, is uncertain. In cattle, maximal damage occurs at approximately 12 to 16 weeks of gestation, when neural tissues, especially in the brain, are differentiating. Infection of sheep and goats at the most critical stage of gestation (28-36 days) results in extensive damage to the central nervous system and severe defects.
The duration of infection in the foetus is related to the immunocompetence of individual foetuses to develop antibodies which can neutralize the virus. Immunocompetence develops in calves from about 70 days and in lambs between 65 and 70 days of gestation64, 67 (see Bovine viral diarrhoea and mucosal disease, and Border disease).
Comparable observations have been made in ruminants infected with Schmallenberg virus, although the impact has been greater in sheep than in cattle.21, 80
There is little information available on the effects of the other Simbu-group viruses on mammalian hosts, but their mode of action and effects are probably similar to those of Akabane virus.
Clinical signs
Typically, infection of ruminants with Akabane virus after birth results in no clinical signs. However, there are strains of Akabane virus in Japan that have been shown to be a cause of acute encephalitis in both young calves and mature cattle. Aino, Peaton, Douglas and Tinaroo viruses have all been isolated on several occasions from sentinel cattle under close daily observation without the detection of any overt clinical signs.71-73, 76 When Schmallenberg virus first emerged in Europe, some cattle were observed with a fever and acute diarrhoea30 while Shuni virus in Africa has been associated with meningoencephalitis in horses and wildlife species including African buffalo, warthogs, sable antelope and white rhinoceros.10, 14, 20, 81
When pregnant ruminants are infected with Akabane virus, except for some abortions in the third trimester of pregnancy, there are no other clinical signs until progeny are born.75
Calves and lambs that develop HE or AG, or both, as a result of intrauterine infection with Akabane virus may be aborted, stillborn or delivered alive at term. Gestation periods, particularly when sheep are affected, may be prolonged and birth-weights reduced.28
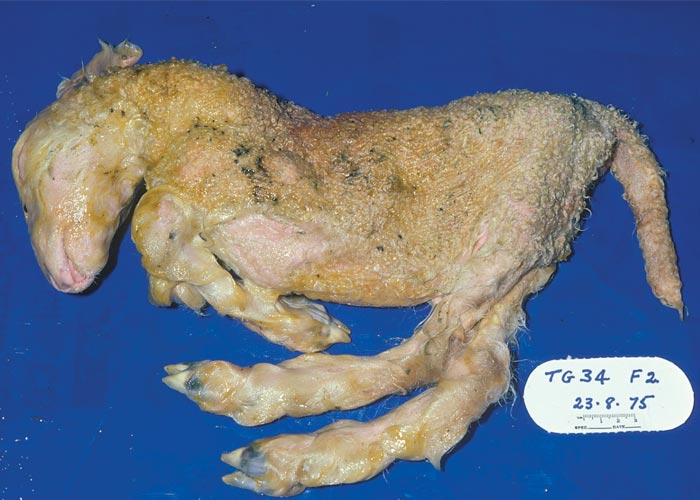
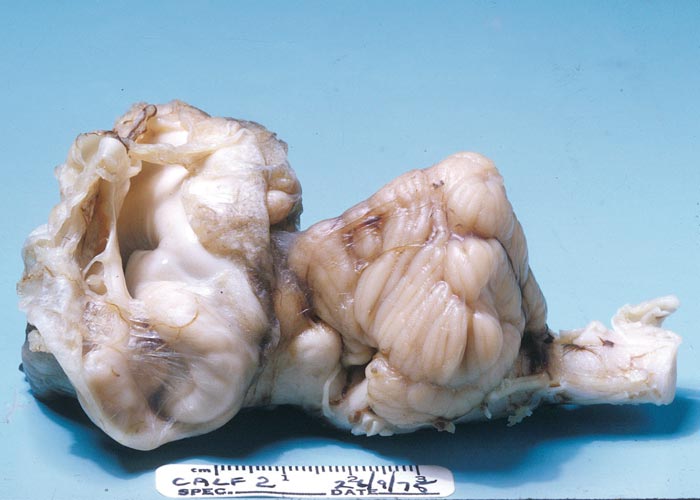
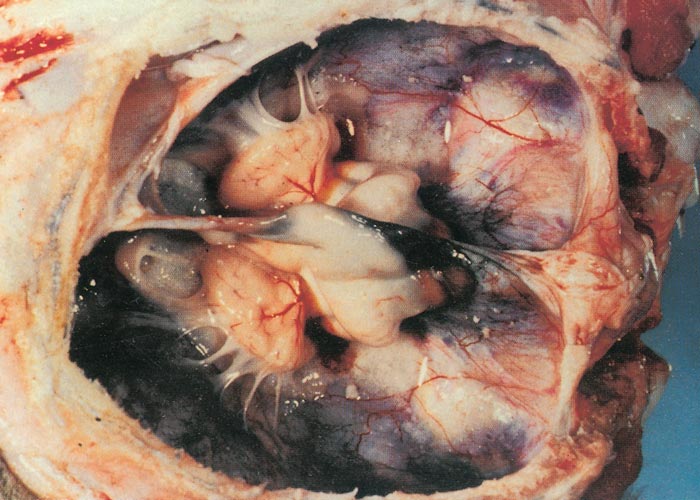
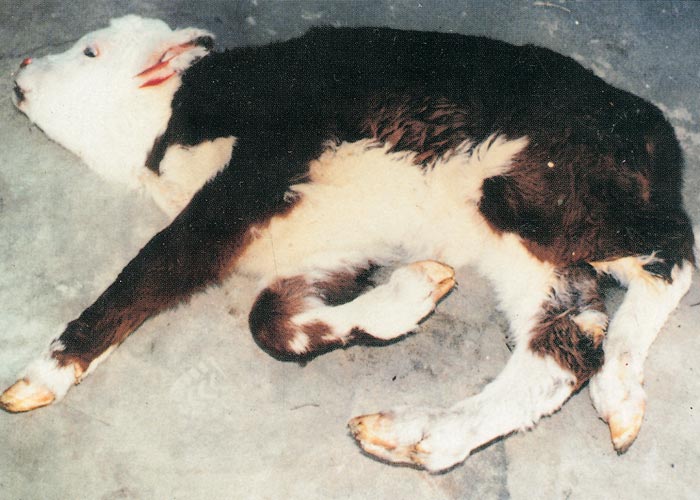
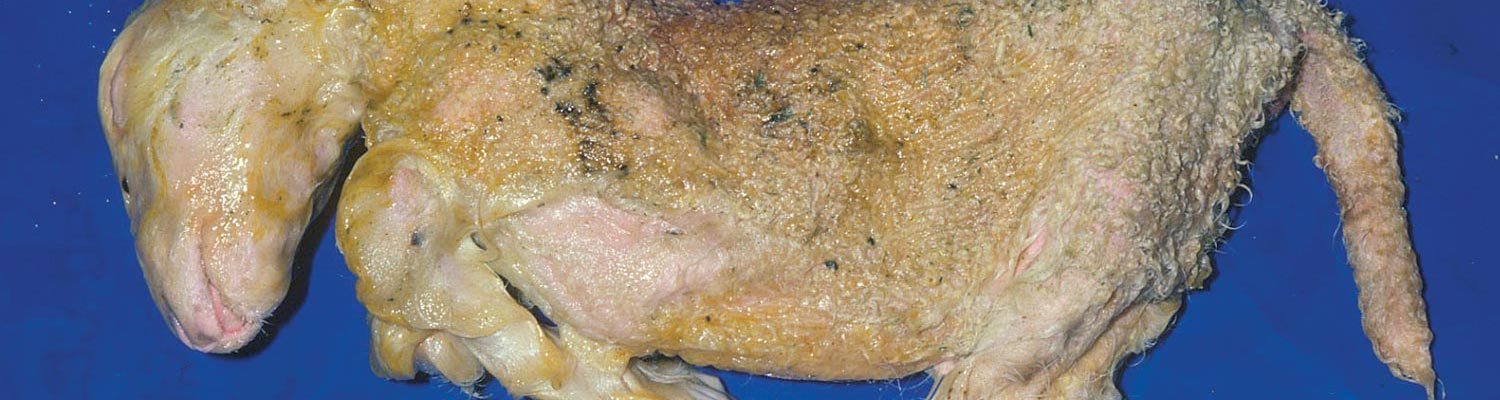
In cattle, the species in which the greatest losses usually occur, the range and severity of congenital abnormalities varies depending on the stage of gestation at which the foetus is infected. In herds with either ‘year round’ or extended calving periods, the full spectrum of abnormalities may be seen. Intensive study of cattle exposed to Akabane virus at different stages throughout gestation has demonstrated the sequential occurrence of defects in calves ranging from AG through to severe HE.41 Naturally, these conditions are observed in a ‘reverse’ sequence. Those that are the result of infection late in gestation are seen in the first calves born (eg. polioencephalomyelitis followed by AG) while those that result following infection early in gestation HE are seen towards the end of calving. With very restricted seasonal breeding periods or limited periods of exposure to infected vectors, abnormalities may be limited to one type, for example only arthrogryposis. Calves that are infected very late in gestation may develop polioencephalomyelitis. At birth they are alert but unable to stand and have flaccid paralysis or incoordination of the limbs. Infections that occur in cattle from late in the first and throughout the second trimester of gestation (3-6 months of pregnancy) give rise to the conditions shown in Figures 1 to Figures 4. Animals with AG have fixed joints and muscular atrophy and may not be able to rise. Such defects often give rise to dystocia, and intervention to save the dam may be required. One or more joints on up to all four limbs may be involved to a greater or lesser extent. As calves are infected earlier in gestation (4-5 months), there is a tendency for more severe AG with more joints and more limbs to be involved and abnormalities of the spinal column (eg. torticollis and scoliosis) may also be observed.40 Some new-born animals with only one limb affected can rise and may improve their mobility with time. Hydranencephaly is the outcome of infection during the third and fourth months of gestation, with more severe defects following infection at the earliest stages of pregnancy. In severe cases of HE, the cerebral hemispheres are reduced to membranous fluid-filled sacs with only the brainstem and cerebellum remaining. In lambs and goat kids infected at 28-36 days of gestation, lesions of the brain and spinal cord are even more severe.
Depending on the extent of the defects, calves with brain lesions may have higher functions impaired. Those with some cerebral function may suck without assistance and learn to follow their dam or the group of cattle to which they belong. At the other extreme, calves may be blind, ‘dopey’ or unable to respond to their dam and find her udder and so are eventually abandoned. Usually the sucking reflex is intact and they can be hand-fed. Even when the cerebrum is more or less entirely absent, all vital functions are usually intact and the calf can stand and may be able to walk but may die from misadventure. Some have been reared to maturity and have even calved normally.
An essentially similar pattern occurs in sheep although the successive progession of lesions noted in cattle is less distinct. For example, lambs are often born with both severe AG and HE. The congenital abnormalities described for Schmallenberg virus are indistinguishable from those induced by Akabane virus. However, Schmallenberg is less teratogenic11 and on a flock or herd basis the incidence of abnormal offspring is generally lower.
Pathology
The gross and histological changes observed in both field and experimental cases resulting from Akabane virus infection of cattle and sheep have been described in detail.24, 25, 44, 45, 65, 66, 84 An outline of the sequential occurrence of gross pathological changes has also been described under clinical signs (see Clinical signs). When cows are infected at different stages of pregnancy, the longer gestational length results in less severe lesions in the CNS. The neurological signs that are observed when calves are infected around the time of birth, and occasionally in older animals86 are the result of an acute polioencephalomyelitis. The fixation of limbs associated with AG is primarily due to muscular atrophy arising mainly from destruction of motor horn neurons. Arthrogryposis will often involve a single joint following infection in the sixth month of gestation. When infection occurs at early stages of pregnancy, in the fifth or fourth months of gestation, the lesions become more severe, and multiple joints may be affected on all limbs and the spinal cord may also be involved. Similarly, defects of the brain is first observed as small focal cyst-like cavities (porencephaly) that progressively become more severe and after infection at the end of the third month of gestation, there can be complete destruction of the cerebral hemispheres leaving only fluid-filled sacs (hydranencephaly): The only structures that can be recognised are parts of the brainstem, and in the case of Akabane virus, a relatively normal cerebellum.
In sheep and goats the gross defects of the brain is much more severe, with little normal CNS tissue recognisable. The lesions and defects described for Akabane are similar for other recognised pathogenic Simbu viruses such as Aino, Peaton, Shamonda and Schmallenberg but the lesions may vary in distribution and symmetry.12, 52
Diagnosis and differential diagnosis
Sporadic cases of AG/HE due to Akabane or related viruses often remain undiagnosed. When a cluster of cases occurs, the presence of AG or brain deformities in aborted or new-born calves or lambs should suggest a teratogenic virus as a possible cause (see Pathology). Serum should be collected, prior to suckling, from the affected calf or lamb to enable the detection of antibodies to potentially teratogenic viruses. Pericardial or pleural fluids are also suitable for the detection of antibodies and are often of higher quality than serum in stillborn animals. Both IgG and IgM antibodies to Akabane and other Simbu viruses are produced in foetuses after they become immunocompetent. Thus, antibodies to a Simbu virus in precolostral serum is strong evidence that the calf or lamb was infected in utero.24 An examination of the abomasum for milk curd should be made as confirmatory evidence that the calf or lamb has not ingested milk. As a further check, a serum sample should be taken from the dam and the two sera tested in parallel for antibodies to the suspect virus, as well as to one or more ubiquitous viruses. If the serum from the new-born contains antibodies to the same range of viruses as that of its dam, the conclusion can be drawn that it has sucked. If antibodies to only a single virus are in the serum of the new-born in contrast to several in its dam’s serum, then the evidence is strong that it has been infected in utero.
Although culture of tissues from affected calves or lambs for viruses usually gives negative results, residual RNA may sometimes be detected by qRT-PCR in samples of CNS tissue, placenta, spleen and lymph nodes.7 Virus is also more likely to be detected in the tissues of aborted foetuses.
Despite some cross- reactivity, virus neutralization tests are the most specific serological tests available.57 ELISAs have been evaluated in Japan and Australia for Akabane.33 In Europe there are several ELISAs available to detect antibodies to Schmallenberg.4, 8, 61 Some assays are Simbu-group reactive, while others are virus (eg. Akabane, Schmallenberg) specific.
Although the brain defects are suggestive of a Simbu virus infection, laboratory confirmation is essential because these lesions can be caused by a number of other viral infections, most notably the pestiviruses (see Bovine viral diarrhoea and Border disease) and some orbiviruses (see Palyam serogroup orbivirus infections) such as Chuzan as well as bluetongue (see Bluetongue) and Wesselsbron disease (see Wesselsbron disease).23
Control
Control of winged vectors (Culicoides and mosquitoes) is difficult and in most instances impractical. The most practical option for disease control is to immunize the potential target host before pregnancy occurs. Inactivated virus vaccines against Akabane virus has been produced in Japan and Australia,39, 47 a combination Akabane and Aino vaccine in Japan37 and there are several vaccines against Schmallenberg virus available in Europe.
References
- ADAMS, M. J., LEFKOWITZ, E. J., KING, A. M. Q., HARRACH, B., HARRISON, R. L., KNOWLES, N. J., KROPINSKI, A. M., KRUPOVIC, M., KUHN, J. H., MUSHEGIAN, A. R., NIBERT, M., SABANADZOVIC, S., SANFAÇON, H., SIDDELL, S. G., SIMMONDS, P., VARSANI, A., ZERBINI, F. M., GORBALENYA, A. E. & DAVISON, A. J., 2016. Ratification vote on taxonomic proposals to the International Committee on Taxonomy of Viruses. Archives of Virology, 161, 2921-2949.
- AL-BUSAIDY, S. M., HAMBLIN, C. & TAYLOR, W. P., 1987. Neutralising antibodies to Akabane virus in free-living wild animals in Africa. Tropical Animal Health and Production, 19, 197-202.
- ALLINGHAM, P. G. & STANDFAST, H. A., 1990. An investigation of transovarial transmission of Akabane virus in Culicoides brevitarsis. Australian Veterinary Journal, 67, 273-274.
- ANON, 2012. Serological ELISA for SBV currently being evaluated. Veterinary Record, 170, 453.
- BARNARD, B. J. H., 1977. Akabane virus in Suid-Afrika. Journal of the South African Veterinary Association, 48, 161.
- BEER, M. C. F. J. & VAN DER POEL, W. H. M., 2013. ‘Schmallenberg virus’- a novel orthobunyavirus emerging in Europe. Epidemiology and infection, 141, 1-8.
- BILK, S., ORG, S., SCHULZE, C., FISCHER, M., BEER, M., HLINAK, A. & HOFFMANN, B., 2012. Organ distribution of Schmallenberg virus RNA in malformed newborns. Veterinary Microbiology, 159, 236-8.
- BREARD, E., LARA, E., COMTET, L., VIAROUGE, C., DOCEUL, V., DESPRAT, A., VITOUR, D., POZZI, N., CAY, A. B., DE REGGE, N., POURQUIER, P., SCHIRRMEIER, H., HOFFMANN, B., BEER, M., SAILLEAU, C. & ZIENTARA, S., 2013. Validation of a Commercially Available Indirect Elisa Using a Nucleocapside Recombinant Protein for Detection of Schmallenberg Virus Antibodies. PLOS ONE, 8, e53446.
- CAUSEY, O. R., KEMP, G. E., CAUSEY, C. E. & LEE, V. H., 1972. Isolations of Simbu-group viruses in Ibadan, Nigeria 1964-69, including the new types Sango, Shamonda, Sabo and Shuni. Annals of Tropical Medicine and Parasitology, 66, 357-362.
- COETZER, J. A. W. & HOWELL, P. G., 1999. Viral encephalitides affecting horses in South Africa. In: WERNERY, U., WADE, J.F., MUMFORD, J.A. & KAADEN, O-R., (eds). Equine infectious diseases. VIII. Proceedings of the Eighth International Conference, Dubai, 23-26 March 1998. 269-273.
- COLLINS, A. B., MEE, J. F. & KIRKLAND, P. D., 2018. Pathogenicity and teratogenicity of Schmallenberg virus and Akabane virus in experimentally infected chicken embryos. Veterinary Microbiology, 216, 31-37.
- COVERDALE, O. R., CYBINSKI, D. H. & ST. GEORGE, T. D., 1979. A study of the involvement of three Simbu group arboviruses in bovine congenital arthrogryposis and hydranencephaly in the New England area of New South Wales. In: ST. GEORGE, T.D. & FRENCH, E.L., (eds). Arbovirus Research in Australia. Commonwealth Scientific and Industrial Research Organization and Queensland Institute of Medical Research, Brisbane. 130-136,
- CYBINSKI, D. H. & ZAKRZEWSKI, H., 1983. A dual infection of a bull with Akabane and Aino viruses. Australian Veterinary Journal, 60, 283.
- DA COSTA MENDES, V. M., 1984. The isolation and importance of Simbu group viruses in South Africa. M.Med.Vet. thesis, University of Pretoria.
- DAVIES, F. G. & JESSET, D. M., 1985. A study of the host range distribution of antibody to Akabane virus (genus Bunyavirus, family Bunyaviridae) in Kenya. Journal of Hygiene, 95, 191-196.
- DELLA-PORTA, A. J., O’HALLORAN, M. L., PARSONSON, I. M., SNOWDON, W. A., MURRAY, M. D., HARTLEY, W. J. & HAUGHEY, K. J., 1977. Akabane disease: Isolation of the virus from naturally infected ovine foetuses. Australian Veterinary Journal, 53, 51-52.
- DOCEUL, V., LARA, E., SAILLEAU, C., BELBIS, G., RICHARDSON, J., BRÉARD, E., VIAROUGE, C., DOMINGUEZ, M., HENDRIKX, P., CALAVAS, D., DESPRAT, A., LANGUILLE, J., COMTET, L., POURQUIER, P., ELÉOUËT, J. F., DELMAS, B., MARIANNEAU, P., VITOUR, D. F. & ZIENTARA, S., 2013. Epidemiology, molecular virology and diagnostics of Schmallenberg virus, an emerging orthobunyavirus in Europe. Veterinary research, 44:31.
- DOHERTY, R. L., CARLY, J. G., STANDFAST, H. A., DYCE, A. L. & SNOWDON, W. A., 1972. Virus strains isolated from arthropods during an epizootic of bovine ephemeral fever in Queensland. Australian Veterinary Journal, 48, 81-86.
- EDWARDS, J. F., LIVINGSTON, C. W., CHUNG, S. I. & COLLISON, E. C., 1989. Ovine arthrogryposis and central nervous system malformations associated with in utero Cache Valley virus infection: Spontaneous disease. Veterinary Pathology, 26, 33-39.
- FOGGIN, C. M. & SWANEPOEL, R., 1990. Veterinary Research Laboratory, P.O. Box 8101, Causeway, Harare, Zimbabwe. Unpublished data.
- GARIGLINANY, M. M., HOFFMANN, B., DIVE, M., SARTELET, A., BAYROU, C., CASSART, D., BEER, M. & DESCHMECT, D., 2012. Schmallenberg virus in calf born at term with porencephaly, Belgium. Emerging Infectious Diseases, 18, 1005-1006.
- GOLENDER, N., BRENNER, J., VALDMAN, M., KHINICH, Y., VELIZAR, BUMBAROV, V., PANSHIN, A., EDERY, N., PISMANIK, S. & BEHAR, A., 2015. Malformations Caused by Shuni Virus in Ruminants, Israel, 2014-2015. Emerging Infectious Diseases, 21, 2267-2268.
- GOTO, Y. M. Y. & KONO, Y., 1988. Serologic evidence for the etiologic role of Chuzan virus in an epizootic of congenital abnormalities with hydranencephaly-cerebellar hypoplasia syndrome of calves in Japan. American Journal of Veterinary Research, 49, 2026-2029.
- HARTLEY, W. J., DE SARAM, W. G., DELLA-PORTA, A. J., SNOWDON, W. A. & SHEPHERD, N. C., 1977. Pathology of congenital bovine epizootic arthrogryposis and hydranencephaly and its relationship to Akabane virus. Australian Veterinary Journal, 53, 319-325.
- HARTLEY, W. J. & HAUGHEY, K. G., 1974. An outbreak of microencephaly in lambs in New South Wales. Australian Veterinary Journal, 50, 55-58.
- HARTLEY, W. J. & WANNER, R. A., 1974. Bovine congenital arthrogryposis in New South Wales. Australian Veterinary Journal, 50, 185-188.
- HARTLEY, W. J., WANNER, R. A., DELLA-PORTA, A. J. & SNOWDON, W. A., 1975. Serological evidence for the association of Akabane virus with epizootic bovine congenital arthrogryposis and hydranencephaly syndromes in New South Wales. Australian Veterinary Journal, 51, 103-104.
- HAUGHEY, K. G., HARTLEY, W. J., DELLAPORTA, A. J. & MURRAY, M. D., 1988. Akabane disease in sheep. Australian Veterinary Journal, 65, 136-140.
- HENRY, M., 1937. Research — General. Livestock Disease Research New South Wales, 7, 58. Department of Agriculture, Sydney, New South Wales.
- HIRASHIMA, Y., KITAHARA, S., KATO, T., SHIRAFUJI, H., TANAKA, S. & YANASE, T., 2017. Congenital Malformations of Calves Infected with Shamonda Virus, Southern Japan. Emerging Infectious Diseases, 23, 993-996.
- HOFFMANN, B., SCHEUCH, M., HOPER, D., JUNGBLUT, R., HOLSTEG, M., SCHIRRMEIER, H., ESCHBAUMER, M., GOLLER, K. V., WERNIKE, K., M., F., BREITHAUPT, A., METTENLEITER, T. C. & BEER, M., 2012. Novel orthobunyavirus in cattle, Europe, 2011. Emerging Infectious Diseases, 18, 469-472.
- HOWELL, P. G. & COETZER, J. A. W., 1999. Serological evidence of infection of horses by viruses of the Simbu-serogroup in South Africa. In: WERNERY, U., WADE, J.F., MUMFORD, J.A. & KAADEN, O-R., (eds). Equine infectious diseases. VIII. Proceedings of the Eighth International Conference, Dubai, 23-26 March 1998. 549.
- IDE, S., BABA, K., TSUCHIMOTO, M., NAGANO, H., EIGUCHI, Y., YAMAGAMA, T., YAMAGISHI, H., TANAKA, Y., FUJISAKI, T., HOHDATSU, T. & MATUMOTO, M., 1989. Detection of antibodies against Akabane virus in bovine sera by enzyme-linked immunoabsorbent assay. Veterinary Microbiology, 20, 275-280.
- INABA, Y., KUROGI, H. & OMORI, T., 1975. Akabane disease: Epizootic abortion, premature birth, stillbirth and congenital arthrogryposis/ hydranencephaly in cattle, sheep and goats caused by Akabane virus. Australian Veterinary Journal, 51, 584-585.
- INABA, Y. & MATUMOTO, M., 1981. Congenital arthrogryposis hydranencephaly syndrome. In: GIBBS, E.P.J., (ed.). Virus Diseases of Food Animals. Vol. II. New York: Academic Press.
- KARABATSOS, N., 1985. International Catalogue of Arboviruses Including Certain Other Viruses of Vertebrates. San Antonio, Texas: American Society of Tropical Medicine and Hygiene.
- KIM, Y. H., KWEON, C. H., TARK, D. S., LIM, S. I., YANG, D. K., HYUN, B. H., SONG, J. Y., HUR, W. & PARK, S. C., 2011. Development of inactivated trivalent vaccine for the teratogenic Aino, Akabane and Chuzan viruses. Biologicals, 39, 152-157.
- KIRKLAND, P. D., 2015. Akabane virus infection. OIE Scientific and Technical Review. 34, 403-410.
- KIRKLAND, P. D. & BARRY, R. D., 1984. The epidemiology and control of Akabane disease. In: DELLA-PORTA, A.J., (ed.). Veterinary Viral Diseases. Their Significance in Southeast Asia and the Western Pacific. Sydney: Academic Press.
- KIRKLAND, P. D. & BARRY, R. D., 1986. The economic impact of Akabane virus and cost effectiveness of Akabane vaccine in New South Wales. In: ST. GEORGE, T.D., KAY, B.H. & BLOK, J., (eds). Arbovirus Research in Australia. Commonwealth Scientific and Industrial Research Organisation and Queensland Institute of Medical Research, Brisbane. 229-232.
- KIRKLAND, P. D., BARRY, R. D., HARPER, P. A. W. & ZELSKI, R. Z., 1988. The development of Akabane virus-induced congenital abnormalities in cattle. The Veterinary Record, 122, 582-586.
- KIRKLAND, P. D., BARRY , R. D. & MACADAM, J. F., 1983. An impending epidemic of bovine congenital deformities. Australian Veterinary Journal, 60, 221-223.
- KIRKLAND, P. D., CYBINSKI, D. H. & WALKER, K. H., 1989. Congenital defects in animals in Australia — the role of arboviruses. In: UREN, M.F., BLOK, J. & MANDERSON, L., (eds). Arbovirus Research in Australia. Proceedings of the Fifth Symposium. Commonwealth Scientific and Industrial Research Organization and Queensland Institute of Medical Research, Brisbane.
- KONNO, S., MORIWAKI, M. & NAKAGAWA, M., 1982. Akabane disease in cattle: Congenital abnormalities caused by viral infection. Spontaneous disease. Veterinary Pathology, 19, 246-266.
- KONNO, S. & NAKAGAWA, M., 1982. Akabane disease in cattle: Congenital abnormalities caused by viral infection. Experimental disease. Veterinary Pathology, 19, 267-279.
- KUROGI, H., AKIBA, Y., INABA, Y. & MATUMOTO, M., 1987. Isolation of Akabane virus from the biting midge Culicoides oxystoma in Japan. Veterinary Microbiology, 15, 243-248.
- KUROGI, H., INABA, Y., TAKAHASHI, E., SATO, K., GOTO, Y., SATODA, K., OMORI, T. & HATAKEYAMA, H., 1978. Development of inactivated vaccine for Akabane disease. National Institute of Animal Health Quarterly, 18, 97-108.
- KUROGI, H., INABA, Y., TAKAHASHI, E., SATO, K., OMORI, T., MIURA, Y., GOTO, Y., FUJIWARA , Y., HATANO, Y., KODAMA, K., FUKUYAMA, S., SASAKI, N. & MATUMOTO, M., 1976. Epizootic congenital arthrogryposis hydranencephaly syndrome in cattle: Isolation of Akabane virus from affected fetuses. Archives of Virology, 51, 56-74.
- KUROGI, H., Y., I., TAKAHASHI, E., SATO, K., SATODA, K., GOTO, Y., OMORI, T. & MATUMOTO, M., 1977. Congenital abnormalities in newborn calves after inoculation of pregnant cows with Akabane virus. Infection and Immunity, 17, 338-343.
- MARKUSFELD, O. & MAYER, E., 1971. An arthrogryposis and hydranencephaly syndrome in calves in Israel, 1969/70. Epidemiological and clinical aspects. Refuah Veterinarith, 28, 51-61.
- MATHEW, C., KLEVAR, S., ELBERS, A. R. W., VAN, D., POEL, W. H. M., KIRKLAND, P. D., GODFROID, J., MDEGELA, R. H., MWAMENGELE, G. & STOKSTAD, M., 2015. Detection of serum neutralizing antibodies to Simbu sero-group viruses in cattle in Tanzania. Veterinary research, 11, 208.
- MATSUMORI, Y., AIZAWA, M., SAKAI, Y., INOUE, D., KODANI, M., TSUHA, O., BEPPU, A., HIRASHIMA, Y., KONO, R., OHTANI, A., YANASE, T., SHIRAFUJI, H., KATO, T., TANAKA, S. & YAMAKAWA, M., 2018. Congenital abnormalities in calves associated with Peaton virus in Japan. Journal of Veterinary Diagnostic Investigation, doi.org/10.1177/1040638718796269.
- MCINTOSH, B. M., 1980. The epidemiology of arthropod-borne viruses in southern Africa. D.Sc. thesis, University of Pretoria.
- MCINTOSH, B. M., JUPP, P. G. & DE SOUSA, J., 1972. Further isolations of arboviruses from mosquitoes collected in Tongaland, South Africa, 1960-1968. Journal of medical entomology, 9, 155-159.
- MCINTOSH, B. M., MCGILLIVRAY, G. M. & DICKENSON, D. B., 1965. Ingwavuma virus: An arbovirus isolated in South Africa. South African Journal of Medicine, 30, 67-70.
- METSELAAR, D. & ROBIN, Y., 1976. Akabane virus isolated in Kenya. The Veterinary Record, 99, 86.
- MIURA, Y., HAYASHI, S., ISHIHARA, T., INABA, Y., OMORI, T. & MATUMOTO, M., 1974. Neutralizing antibody against Akabane virus in precolostral sera from calves with congenital arthrogryposis-hydranencephaly syndrome. Archiv für die Gesamte Virusforschung, 46, 377-380.
- MOUCHANTAT, S., WERNIKE, K., LUTZ, W., HOFFMANN, B., ULRICH, R. G., BÖRNER, K., WITTSTATT, U. & BEER, M., 2015. A broad spectrum screening of Schmallenberg virus antibodies in wildlife animals in Germany. Veterinary research, 46, 99.
- MULLER, M. J., 1990. CSIRO Division of Tropical Animal Production, Indooroopilly, Queensland, Australia. Unpublished data.
- MURRAY, M. D., 1987. Akabane epizootics in New South Wales: Evidence for long-distance dispersal of the biting midge Culicoides brevitarsis. Australian Veterinary Journal, 64, 305-308.
- NÄSLUND, K., BLOMQVIST, G., VERNERSSON, C., ZIENTARA, S., BRÉARD, E. & VALARCHER, J. F., 2014. Development and evaluation of an indirect enzyme-linked immunosorbent assay for serological detection of Schmallenberg virus antibodies in ruminants using whole virus antigen. Acta veterinaria Scandinavica, 56, 71.
- OYA, A., OKUNO, T., OGATA, T., KOBAYASHI, I. & MATSUYAMA, T., 1961. Akabane, a new arbovirus isolated in Japan. Japanese Journal of Medical Sciences and Biology, 14, 101-108.
- PARSONSON, I. M., DELLA-PORTA, A. J. & MCPHEE, D. A., 1982. Pathogenesis and virulence studies of Australian Simbu serogroup Bunyaviruses. In: MACKENZIE, J., (ed.). Viral Diseases in South-East Asia and the Western Pacific. Sydney: Academic Press.
- PARSONSON, I. M., DELLA-PORTA, A. J., O’HALLORAN, M. L., SNOWDON, W. A., FAHEY, K. J. & STANDFAST, H. A., 1981. Akabane virus infection in the pregnant ewe. I. Growth of virus in the foetus and the development of the foetal immune response. Veterinary Microbiology, 6, 197-207.
- PARSONSON, I. M., DELLA-PORTA, A. J. & SNOWDON, W. A., 1977. Congenital abnormalities in newborn lambs after infection of pregnant sheep with Akabane virus. Infection and Immunity, 15, 254-262.
- PARSONSON, I. M., DELLAPORTA, A. J. & SNOWDON, W. A., 1981. Akabane virus infection in the pregnant ewe. II. Pathology of the foetus. Veterinary Microbiology, 6, 209-224.
- PARSONSON, I. M. & MCPHEE, D. A., 1985. Bunyavirus pathogenesis. Advances in Virus Research, 40, 279-316.
- PARSONSON, I. M., MCPHEE, D. A., DELLAPORTA, A. J., MCLURE, S. & MCCULLAGH, P., 1988. Transmission of Akabane virus from the ewe to the early fetus (32-53 days). Journal of Comparative Pathology, 98, 215-227.
- RUDERT, C. P., LAWRENCE, J. A., FOGGIN, C. & BARLOW, R. M., 1978. A rigid lamb syndrome in sheep in Rhodesia. The Veterinary Record, 102, 374.
- SHEPHERD, N. C., GEE, C. D., JESSEP, T., TIMMINS, G., CARROLL, S. N. & BONNER, R. B., 1978. Congenital bovine epizootic arthrogryposis and hydranencephaly. Australian Veterinary Journal, 54, 171-177.
- ST. GEORGE, T. D., 1985. Studies on the pathogenesis of bovine ephemeral fever in sentinel cattle. I. Virology and Serology. Veterinary Microbiology, 10, 493-504.
- ST. GEORGE, T. D., 1990. CSIRO Division of Tropical Animal Production, Indooroopilly, Queensland, Australia. Unpublished data.
- ST. GEORGE, T. D., CYBINSKI, D. H., FILIPPICH, C. & CARLEY, J. G., 1979. The isolation of three Simbu group viruses new to Australia. Australian Journal of Experimental Biology and Medical Science, 57, 581-582.
- ST. GEORGE, T. D. & STANDFAST, H. A., 1989. Simbu group viruses with teratogenic potential. In: MONATH, T.P., (ed.). The Arboviruses: Epidemiology and Ecology. Vol. IV. Florida: CRC Press.
- ST. GEORGE, T. D., STANDFAST, H. A. & CYBINSKI, D. H., 1978. Isolations of Akabane virus from sentinel cattle and Culicoides brevitarsis. Australian Veterinary Journal, 54, 558-561.
- ST. GEORGE, T. D., STANDFAST, H. A., CYBINSKI, D. H., FILIPPICH, C. & CARLEY, J. G., 1980. Peaton virus: A new Simbu group arbovirus isolated from cattle and Culicoides brevitarsis in Australia. Australian Journal of Biological Sciences, 33, 235-243.
- STANDFAST, H. A., ST GEORGE, T. D. & CYBINSKI, D. H., 1986. Economics and epidemiology of Simbu group viruses in Australia. In: ST. GEORGE, T.D., KAY, B.H. & BLOK, J., (eds). Arbovirus Research in Australia. Commonwealth Scientific and Industrial Research Organisation and Queensland Institute of Medical Research, Brisbane.
- TARLINTON, R., DALY DUNHAM, S. & KYDD, J., 2012. The challenge of Schmallenberg virus emergence in Europe. The Veterinary Journal, 194, 10-18.
- THEODORIDIS, A., NEVILL, E. M., ELS, H. J. & BOSHOFF, S. T., 1979. Viruses isolated from Culicoides midges in South Africa during unsuccessful attempts to isolate bovine ephemeral fever virus. Onderstepoort Journal of Veterinary Research, 46, 191-198.
- VAN DEN BROM, R., LUTTIKHOLT, S. J., LIEVAART-PETERSON, K., PEPERKAMP, N. H., MARS, M. H., VAN DER POEL, W. H. & VELLEMA, P., 2012. Epizootic of ovine congenital malformations associated with Schmallenberg virus infection. Tijdschrift voor Diergeneeskunde, 137, 106-111.
- VAN EEDEN, C., WILLIAMS, J. H., GERDES, T. G. H., VAN WILPE, E., VILJOEN, A., SWANEPOEL, R. & VENTER, M., 2012. Shuni virus as cause of neurologic disease in horses. Emerging Infectious Diseases, 18, 318-321.
- VERONESI, E., HENSTOCK, M., GUBBINS, S., BATTEN, C., MANLEY, R., BARBER, J., HOFFMANN, B., BEER, M., ATTOUI, H., MERTENS, P. P. C. & CARPENTER, S., 2013. Implicating Culicoides Biting Midges as Vectors of Schmallenberg Virus Using Semi-Quantitative RT-PCR. PLOS ONE, 8, e57747.
- WEINBREN, M. P., HEYMANN, C. S., KOKERNOT, R. H. & PATERSON, H. E., 1957. Studies on the arthropod-borne viruses of Tongaland. VII. Simbu virus, a hitherto unknown agent isolated from Aedes (Banksinella) circumluteolus. South African Journal of Medical Science, 22, 93-102.
- WHITTEM, J. H., 1957. Congenital abnormalities in calves: Arthrogryposis and hydranencephaly. Journal of Pathology and Bacteriology, 73, 375-387.
- WORTH, C. P., PATERSON, H. E. & DE MEILLON, B., 1961. The incidence of arthropod-borne viruses in a population of culicine mosquitoes in Tongaland, Union of South Africa. 1956-1960. American Journal of Tropical Medicine and Hygiene, 10, 583-592.
- YANASE, T., KATO, T., HAYAMA, Y., AKIYAMA, M., ITOH, N., HORIUCHI, S., HIRASHIMA, Y., SHIRAFUJI, H., YAMAKAWA, M., TANAKA, S. & TSUTSUI, T., 2017. Transition of Akabane virus genogroups and its association with changes in the nature of disease in Japan. Transboundary and Emerging Diseases, 65, e434-e443.