- Infectious Diseases of Livestock
- Part 2
- Bovine spongiform encephalopathy
- GENERAL INTRODUCTION: PARAMYXOVIRIDAE AND PNEUMOVIRIDAE
- Rinderpest
- Peste des petits ruminants
- Parainfluenza type 3 infection
- Bovine respiratory syncytial virus infection
- Hendra virus infection
- Paramyxovirus-induced reproductive failure and congenital defects in pigs
- Nipah virus disease
- GENERAL INTRODUCTION: CALICIVIRIDAE AND ASTROVIRIDAE
- Vesicular exanthema
- Enteric caliciviruses of pigs and cattle
- GENERAL INTRODUCTION: RETROVIRIDAE
- Enzootic bovine leukosis
- Jaagsiekte
- Visna-maedi
- Caprine arthritis-encephalitis
- Equine infectious anaemia
- GENERAL INTRODUCTION: PAPILLOMAVIRIDAE
- Papillomavirus infection of ruminants
- Papillomavirus infection of equids
- GENERAL INTRODUCTION: ORTHOMYXOVIRIDAE
- Equine influenza
- Swine influenza
- GENERAL INTRODUCTION: CORONAVIRIDAE
- Porcine transmissible gastroenteritis
- Porcine respiratory coronavirus infection
- Porcine epidemic diarrhoea
- Porcine haemagglutinating encephalomyelitis virus infection
- Porcine deltacoronavirus infection
- Bovine coronavirus infection
- Ovine coronavirus infection
- Equine coronavirus infection
- GENERAL INTRODUCTION: PARVOVIRIDAE
- Porcine parvovirus infection
- Bovine parvovirus infection
- GENERAL INTRODUCTION: ADENOVIRIDAE
- Adenovirus infections
- GENERAL INTRODUCTION: HERPESVIRIDAE
- Equid herpesvirus 1 and equid herpesvirus 4 infections
- Equid gammaherpesvirus 2 and equid gammaherpesvirus 5 infections
- Equine coital exanthema
- Infectious bovine rhinotracheitis/infectious pustular vulvovaginitis and infectious pustular balanoposthitis
- Bovine alphaherpesvirus 2 infections
- Malignant catarrhal fever
- Pseudorabies
- Suid herpesvirus 2 infection
- GENERAL INTRODUCTION: ARTERIVIRIDAE
- Equine viral arteritis
- Porcine reproductive and respiratory syndrome
- GENERAL INTRODUCTION: FLAVIVIRIDAE
- Bovine viral diarrhoea and mucosal disease
- Border disease
- Hog cholera
- Wesselsbron disease
- Louping ill
- West nile virus infection
- GENERAL INTRODUCTION: TOGAVIRIDAE
- Equine encephalitides caused by alphaviruses in the Western Hemisphere
- Old World alphavirus infections in animals
- Getah virus infection
- GENERAL INTRODUCTION: BUNYAVIRIDAE
- Diseases caused by Akabane and related Simbu-group viruses
- Rift Valley fever
- Nairobi sheep disease
- Crimean-Congo haemorrhagic fever
- GENERAL INTRODUCTION: ASFARVIRIDAE
- African swine fever
- GENERAL INTRODUCTION: RHABDOVIRIDAE
- Rabies
- Bovine ephemeral fever
- Vesicular stomatitis and other vesiculovirus infections
- GENERAL INTRODUCTION: REOVIRIDAE
- Bluetongue
- Ibaraki disease in cattle
- Epizootic haemorrhagic disease
- African horse sickness
- Equine encephalosis
- Palyam serogroup orbivirus infections
- Rotavirus infections
- GENERAL INTRODUCTION: POXVIRIDAE
- Lumpy skin disease
- Sheeppox and goatpox
- Orf
- Ulcerative dermatosis
- Bovine papular stomatitis
- Pseudocowpox
- Swinepox
- Cowpox
- Horsepox
- Camelpox
- Buffalopox
- GENERAL INTRODUCTION: PICORNAVIRIDAE
- Teschen, Talfan and reproductive diseases caused by porcine enteroviruses
- Encephalomyocarditis virus infection
- Swine vesicular disease
- Equine picornavirus infection
- Bovine rhinovirus infection
- Foot-and-mouth disease
- GENERAL INTRODUCTION: BORNAVIRIDAE
- Borna disease
- GENERAL INTRODUCTION: CIRCOVIRIDAE AND ANELLOVIRIDAE
- Post-weaning multi-systemic wasting syndrome in swine
- GENERAL INTRODUCTION: PRION DISEASES
- Scrapie
- Bovine spongiform encephalopathy
- Transmissible spongiform encephalopathies related to bovine spongiform encephalopathy in other domestic and captive wild species
Bovine spongiform encephalopathy
This content is distributed under the following licence: Attribution-NonCommercial CC BY-NC  View Creative Commons Licence details here
View Creative Commons Licence details here
NJ Maclachlan and M-L Penrith (Editors). T Konold and E Vallino-Costassa, Bovine spongiform encephalopathy, 2018.

Bovine spongiform encephalopathy
Previous authors: R BRADLEY AND D W VERWOERD
Current authors:
T KONOLD - Veterinary Surgeon, Information Officer & Veterinary Research Officer, DrMedVet, PhD, MRCVS, ASU, Pathology Department, APHA Weybridge, Woodham Lane, New Haw, Addlestone, Surrey, KT15 3NB, United Kingdom
E VALLINO COSTASSA - Doctor in Veterinary Medicine, DVM, PhD, Centre for Animal Encephalopathies (CEA), Istituto Zooprofilattico Sperimentale Piemonte Liguria e Valle d’Aosta, Turin, Italy
Introduction
A new disease in cattle that did not respond to any treatment was first observed on a farm in England in April 1985, which was initially termed chronic hypersensitivity and incoordination syndrome in the absence of any pathological diagnosis.120 The farm experienced a further nine cases over a two year period, of which three were submitted to the Central Veterinary Laboratory for further investigation. These cases and others submitted at around the same time resulted in the discovery of a novel progressive disease, bovine spongiform encephalopathy (BSE), in cattle,114 characterized by grey matter vacuolation and presence of fibrils in the brain. Similar pathological changes had previously been known to occur naturally only in sheep and goats with scrapie and deer and elk as a result of chronic wasting disease, caused by unconventional infectious agents. Epidemiological investigations soon traced the source of infection to the feeding of contaminated meat and bone meal (MBM),125 which was subsequently prohibited to eradicate the disease. More than 170,000 cattle have been affected in the United Kingdom (UK) alone but the disease was also detected in other European countries as well as the USA, Canada and Japan. In 1996, a new human spongiform encephalopathy (variant Creutzfeldt-Jakob disease, vCJD) was reported in the UK,126 which was clinically and pathologically distinct from the sporadic form that has an incidence of approximately 1.5-2 per million people across the world. The similarities in the biological properties of the infectious agent when brain from affected humans and cattle were transmitted into mice18 suggested that the agent was the same and that BSE, or popularly named “mad cow disease”, was a zoonosis. Over 170 vCJD cases have been diagnosed in the UK to date, with considerably fewer cases in other countries,78 some with a history of residence in the UK.
The BSE epidemic had a huge economic impact on the UK and other countries because of restrictions on trade and export of animals and derived products. The European Union introduced legislation in 2001 that required the testing of healthy slaughtered and fallen cattle for BSE. This testing regime identified two additional types of BSE in Europe,13, 23 which were distinct from the common form of BSE, now termed classical BSE (C-type BSE), and separated into H-type and L-type BSE based on the lower and higher molecular mass of the unglycosylated protein fragment detectable when brain samples were subjected to proteinase digestion and Western immune-blotting. The aetiology of these atypical BSE forms remains unknown but their rare occurrence (less than 1 per 1,000,000 tested adult cattle) in cattle aged predominantly 8 years or older, is suggestive of a spontaneous disease.112 It is not yet known whether these atypical BSE strains are also zoonotic but transmissions of L-type BSE to non-human primates26, 91 have been reported although they did not resemble any known form of CJD in humans based on studies in human transgenic mice.60
Aetiology
Bovine spongiform encephalopathy is associated with the presence of an abnormal isoform of a naturally occurring host protein (cellular prion protein, PrPC), which is resistant to proteolysis. It is now commonly accepted that the infectious agent is a proteinaceous infectious particle or prion,95 which induces a switch from an alpha-helical PrPC to a proteinase-resistant beta-sheet conformation. Multiple conformations may exist, resulting in different strains. These strains are usually identified by inoculating tissues from animals affected by transmissible spongiform encephalopathies (TSEs) into mice. The incubation time and the degree of vacuolation (vacuolation score) in specific areas of the brain in a group of diseased mice are then used to characterize the strain.17 The BSE strain responsible for the BSE epidemic appears to be unique and stable, even when transmitted to other susceptible species44 and resembles that of vCJD in mice.96
Questions still remain about the origin of the BSE strain causing the BSE epidemic. It may have been a specific ovine scrapie strain that was transmitted from sheep to cattle, a scrapie-derived strain that was modified in cattle during recycling in MBM, a spontaneous genetic mutation in cattle or strain originating from a non-ruminant mammalian species.94, 125 Intracerebral inoculation of cattle with pools of scrapie-affected sheep brains have failed to reproduce a BSE-like disease24, 29, 72, 97 and oral challenge did not produce disease at all28, 76 but other untested scrapie strains may exist that are pathogenic for cattle.
The discovery of atypical BSE has led to the identification of two additional BSE strains that are naturally occurring in cattle and distinct in their pathological, molecular and biological phenotype, which were termed H-type and L-type BSE. Transmission studies in mice confirmed that these represented new strains.19 In fact, recent research suggested that the origin of classical BSE may be a sporadically occurring atypical BSE strain passaged through other species because a phenotype change from the atypical BSE agent to classical BSE was demonstrated in transmissions to mice.12, 21, 111 However, this has not yet been demonstrated in a farm animal model, such as sheep.82, 101
Epidemiology
Epidemiological studies indicated that classical BSE is a food-borne disease. In Great Britain, the vehicle of infection is believed to be MBM, which is produced from rendering waste animal tissues, including those from ruminants that are not fit for human consumption. The resulting end-product was added to concentrate rations as a high-protein supplement. Changes in rendering practices (shorter processing time, lower temperature and reduction of the hydrocarbon solvent extraction of fat) led to the recycling of the BSE agent, which was not inactivated, and subsequent infection of cattle.122, 123 Trade of infected cattle or contaminated MBM contributed to the international spread of BSE. Other sources of infection outside Great Britain were considered to be cross-contaminated ruminant concentrate feeds and animal fat in milk replacers.25, 59, 129
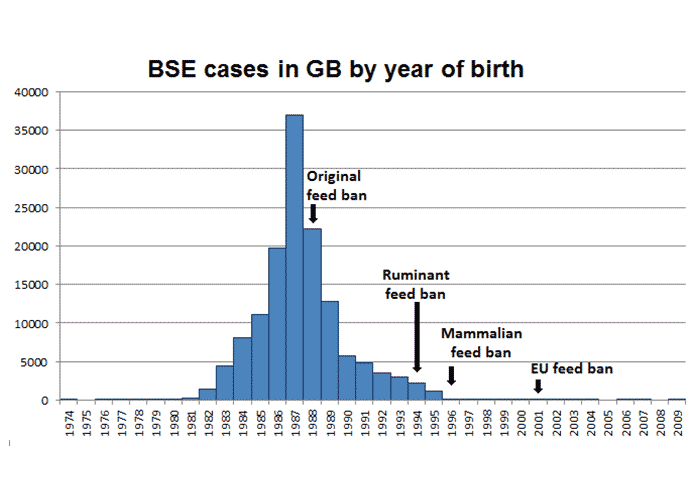
The first legislative measures introduced in Great Britain in July 1988 prohibited the feeding of ruminant derived MBM to ruminants. This lead to a considerable reduction in BSE cases (Figure 1) but due to the long incubation periods it took several years to see this effect. As further BSE cases occurred in cattle born after 1988, the feeding of ruminants with any form of mammalian protein was made illegal in November 1994, which was subsequently extended to any farmed livestock, including fish and horses, in July 1996 because accidental cross-contamination of ruminant diets with MBM intended for pigs and poultry in feed mills was considered a risk factor for cases that continued to occur after the ban.54 The fact that extremely low doses of infected material (1 mg of BSE brainstem) can produce disease in cattle by the oral route117 highlights the risk of cross-contamination. This reinforced ban was effective Europe-wide from January 2001.
Despite the reinforced ban, BSE cases have still been diagnosed in cattle born after 1996 in Great Britain. Investigation into these cases suggested that a food-borne source, possibly imported feed from EU member states prior to 2001, was still the most likely source of infection124 and subsequent cases are still mainly attributed to residual contamination in feed storage silos and in other parts of the feed production process and unlikely to occur spontaneously.5
Vertical and horizontal transmissions of BSE have been considered but are likely to occur at a very low rate if they occur at all, given the low within-herd incidence in BSE-affected herds.32 Embryos collected from cows at disease end-stage and placentae were not found to be infected,128 and bioassays in mice with ovaries, testes, amniotic fluid, milk/ colostrum and semen produced negative results.14, 20, 80, 109 Prion protein has also not been detected in milk from orally infected cattle.36
Bovine spongiform encephalopathy is a slowly progressive disease with a long incubation period. Cattle orally challenged with a range of different doses (ten-fold reductions from 100 g to 1 mg) of classical BSE brainstem homogenate developed disease after an incubation period of 31 to 127 months. Both attack rate and incubation period were dose-dependent.68 Incubation periods or survival times of naturally infected cattle in the UK, where most cases were found, ranged from 20 months to 22 years. The risk of infection is age-dependent, with calves having a higher risk of infection than adult cattle with the highest risk being in the first 6 months after birth.6 It was hypothesized that this age susceptibility was influenced by variation in the innervation of Peyer’s patches and subsequent neuroinvasion.30
Observed differences in risk between calves born in spring and autumn were most likely due to the feeding of higher levels of concentrates than in older cattle and less grazing opportunity in the autumn.6, 32
Epidemiological studies in the UK did not indicate a breed or sex predisposition for BSE and the relative over-representation of dairy cows compared to suckler cows and bulls was explained by the different concentrate feeding practices and number of animals in the country.125 A study in Germany, however, implied that Brown Swiss cattle may be more susceptible.99
Whilst susceptibility of small ruminants to TSEs is influenced by polymorphisms of the prion protein gene (see Scrapie and Transmissible spongiform encephalopathies related to bovine spongiform encephalopathy in other domestic and captive wild species), this is less obvious in cattle. Different forms of the bovine PrP gene with five or six copies of a short, G-C-rich element within the protein-coding exon have been reported43 but did not appear to influence susceptibility to BSE. More recently, two insertion/deletion (indel) polymorphisms, 23 and 12bp indels have been associated with BSE susceptibility. These are located within the promoter and intron 1 of the prion protein gene. Insertion variants were more protective against BSE47, 64 although breed differences exist.48 There was no evidence that this promoter genotype reduces susceptibility to experimental oral exposure to BSE based on small sample sizes,68 which suggests that genetic resistance is not as strong as in small ruminants. An atypical BSE case detected in the USA in 2006 was found to be polymorphic for glutamate (E) and lysine (K) codons at position 211 in the bovine PrP gene, which resembled the E200K mutation in humans with hereditary Creutzfeldt-Jakob disease but this polymorphism was estimated to be infrequent involving less than 1 in 2000 cattle.50 This polymorphism was not found in a study of atypical BSE cases in Poland.46
Pathogenesis
Multiple studies have been published to shed light into the pathogenesis of classical BSE: bioassays in mice or cattle inoculated with tissues from BSE-infected or inoculated animals were carried out or the pathology of natural or experimentally created disease was studied based on prion protein detection methods.
Initial studies confirmed the presence of disease-associated prion protein (PrPSc) in the Peyer’s patches of the distal ileum in cattle orally challenged with suspensions of BSE-infected brainstem, whilst it was not detected in naturally infected cattle.110 It was shown subsequently that the probability of detecting lymphoid follicles in the ileum decline with age and that the proportion of positive follicles increased with dose (100 g compared to 1 g of BSE brainstem) but decreased with age, whilst jejunum and duodenum contain considerably less detectable PrPSc 107 or infectivity.37 Transmission studies in cattle119 using wild-type3 and bovine transgenic mice20 confirmed that the distal ileum carries infectivity even in older cattle despite the absence of detectable PrPSc. Unlike the situation in small ruminants, infectivity in non-neural tissues is limited in cattle: infectivity has been confirmed in palatine tonsil based on cattle bioassay and bioassay in bovine transgenic mice with tissues from orally challenged cattle34, 119 but not in a naturally infected BSE case.20 Infectivity was also demonstrated in pooled nictitating membranes from BSE cases.74 With newer, more sensitive PrPSc detection methods like protein misfolding cyclic amplification, PrPSc was only inconsistently detected in gastrointestinal tissues (oesophagus, rumen, abomasum, colon and rectum) and mesenteric lymph node of orally challenged cattle.38 Bone marrow was considered to be infectious based on transmission studies in wild-type mice but this was neither confirmed in cattle105 nor by protein misfolding cyclic amplification (PMCA).38 Blood as the source of infected bone marrow is unlikely because of the absence of infectivity in blood or blood cells.20, 34, 74, 119 The limited distribution of PrPSc or infectivity in peripheral tissues whilst neural tissues (brain, spinal cord, sensory ganglia) are commonly affected led to the hypothesis that spread of prions to the brain occurs via neural pathways.116 Indeed, pathogenesis studies confirmed that initial PrP immunolabelling occurs in both brainstem and spinal cord suggestive of spread from the gut via splanchnic (sympathetic) nerves to the spinal cord and via the vagus (parasympathetic) nerve to the brainstem,52, 65 regardless of dose.103 There is some evidence that skeletal muscle contains prion protein or infectivity in cattle at the clinical stage81, 89 although not always confirmed by bioassay in cattle74, 119 or transgenic mice.20 This may be the result of centrifugal spread from the central nervous system through the somatic motor and/or sensory pathways to the muscle spindles.89 Striated muscle cells were not considered to harbor BSE infectivity according to a scientific opinion by the European Food Safety Authority.33 In the OIE’s Terrestrial Animal Health Code (Article 11.4.1), deboned skeletal muscle meat is also not associated with any conditions with regards to the BSE risk status of the cattle population where the meat was sourced.85
Less is known about the pathogenesis of atypical BSE due to the lack of naturally occurring clinical cases subject to a full post-mortem examination that enables collection of a wide range of tissues. Some information is available for cattle at the clinical end-stage following experimental, largely intracerebral, inoculation of cattle, assuming that this route of infection mimics natural disease. These studies showed that the tissue involvement is similar to classical BSE in cattle: disease-associated prion protein and/ or infectivity was detected in brain, neurohypophysis, retina, peripheral ganglia and nerves, adrenal gland and muscle.9, 57, 70, 86, 88, 90, 108 Oral transmission of L-type BSE was successful in one heifer challenged with 50 g of brain homogenate that died at 88 months post-challenge, which presented with PrPSc in brain, including brainstem, spinal cord, retina, and – to a lesser degree – in nerve ganglia, cauda equina, vagal nerve, optic nerve, neurohypophysis, ocular muscle, and adrenal gland but not in somatic nerve fibers, enteric nervous system and any lymphoid tissue.87
There are differences in the neuroanatomical PrPSc distribution in naturally occurring atypical BSE compared to classical BSE: prion protein accumulation in the obex is considerably less in atypical BSE, and most prominent in the cortex in L-type BSE and thalamus in H-type BSE,10 which may suggest a different pathogenesis for these atypical BSE types.
Clinical signs
Bovine spongiform encephalopathy has a long incubation period (mean incubation period of approximately five years for classical BSE2 so that clinical signs are only expected in adult cattle. The youngest clinical case of classical BSE was recorded in the UK at 20 months of age,32 whereas atypical BSE is usually found in cattle over 8 years of age.112 Clinical onset is usually insidious and the clinical course progressive, with an average duration of 1 to 2 months (range under 2 weeks to 1 year).67
Affected animals show a combination of non-specific and neurological signs; the latter can be further divided into changes in mental status and behaviour, changes in sensation and changes in posture and movement. There does not appear to be any difference in the clinical presentation between naturally and experimentally infected cattle, regardless of route of inoculation (orally or intracerebrally).
The clinical signs of classical BSE in cattle have been well documented and include, most frequently, apprehension, hyperaesthesia and ataxia.15, 121 Behavioural changes are usually the first clinical signs noticed and are best detected by people familiar with the ‘normal’ behaviour of the animal concerned. Cows may be restless during milking or the cow may enter the milking parlour in an altered position,127 may be reluctant to enter a crush and to pass through doorways (Figure 2) or to cross an obstacle.7, 16, 31
Animals may be offensively aggressive1 but aggression is most frequently defensive, as it occurs in most cases when the animals are confronted with no possibility of escape or confined in a crush, characterized by head butting, sometimes accompanied by vigorous vocalizations8, 127 (Figure 3).
Other behavioural signs include nose licking, nose wrinkling, sneezing, snorting, head tossing, and tooth grinding, which can occur singly or in combination.16, 53 Over-reactivity to external stimuli is a characteristic sensory change in affected cattle. They may startle at noises or movements that would not normally elicit any response. Touching of the lower limbs may result in powerful kicking (Figure 4), which in combination with an exaggerated response to sound, sudden light changes and/ or movement have been used as marker to aid in the clinical diagnosis.16, 69
Affected animals are often ataxic and may exhibit postural changes, such as wide-based stance and low head carriage later in the course of the disease, which eventually leads to recumbency.53, 121, 127 Tremors and myoclonus are also frequently observed.
Non-specific signs include loss of milk yield and loss of weight or condition although the appetite is usually not affected. Animals with BSE often have an unexpectedly low heart rate despite their nervous behaviour or display cardiac arrhythmia suggestive of dysfunction of the autonomic nervous system although this has not been found useful for diagnostic purposes.71
Clinical diagnosis is often difficult in downer animals because thorough examination is not easy to perform. The clinical history is important in these cases to assess whether the animal had previously shown behavioural and/ or locomotor abnormalities. It has been suggested that any recumbent animal with abnormal positioning of the limbs should be considered a BSE suspect, in particular if over-reactivity to external stimuli is also present.75
Comparatively less is known about the clinical presentation of atypical BSE, which is almost exclusively based on experimental transmissions because clinical cases have so far not been reported. A natural case of H-type BSE in Sweden reportedly presented with difficulty in rising without obvious behavioural abnormalities or ataxia.41 A similar presentation was described for a heifer orally challenged with L-type BSE87 whereas intracerebral inoculation with H-type or L-type BSE usually produced a disease characterized by dullness and ataxia, resulting in difficulty rising from recumbency11, 40, 70, 73, 77 although some cattle also presented with over-reactivity, similar to classical BSE.11, 70 Difficulty in rising in the absence of other neurological signs may explain why atypical BSE is usually diagnosed retrospectively in downer cows and fallen stock.
Classical BSE in cattle
Experimental L-Type BSE in cattle
Diagnosis
Confirmatory diagnoses are made following a post-mortem examination, there being no specific gross lesions attributable to BSE. Microscopic lesions are restricted to the central nervous system (CNS), particularly the brain. That should therefore be the target organ for diagnostic purposes.
Histopathology
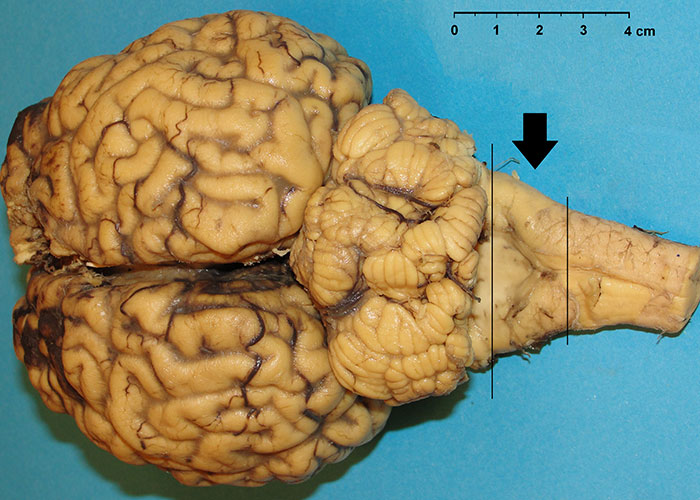
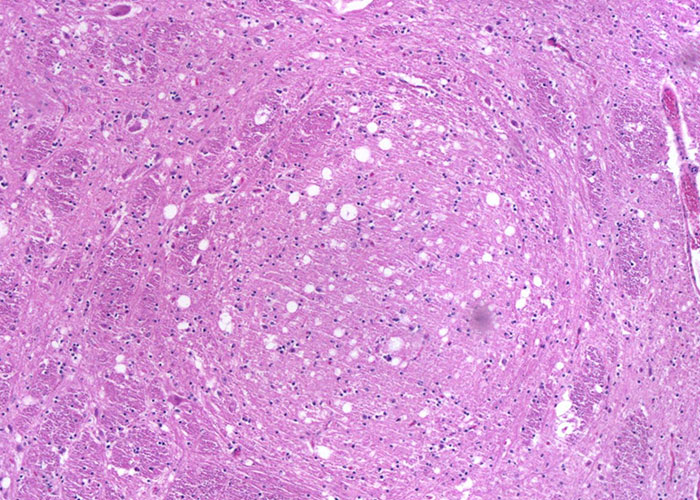
Histological examination needs to be performed on formalin-fixed, paraffin embedded brain sections that are stained with haematoxylin and eosin (H&E). The classic histological changes in the CNS that are visible microscopically are vacuolation of grey matter neuropil (spongiform change) and/or vacuolation of neurons, with a predilection for certain neuroanatomic locations; astrocytosis and neuronal degeneration may also be present.102 Vacuolation of the perikaryon and spongiform changes are usually bilaterally symmetrical.118 A single coronal section of the medulla oblongata at the level of the obex (Figure 5) was identified as appropriate for the histopathological diagnosis of BSE because the vast majority of cases have vacuolar changes in either the solitary tract nucleus NST, (Figure 6) or spinal tract nucleus of the trigeminal nerve (NSTV) although in a small proportion of cases vacuolar changes may be minimal.115 Cases detected by monitoring healthy slaughter cattle in abattoirs, however, may not present with any vacuolar changes at all in these nuclei.104
Moreover, vacuolation could be also present in central gray matter of the midbrain and mild spongiform changes of the neuropil could be observed in some cattle in the thalamus. In natural cases of L-type BSE, spongiosis is not consistently found in the brainstem, at the obex or in more rostral areas. The frontal, parietal, and occipital cortices are apparently spared, and no vacuolation is detected in the olfactory bulb, piriform cortex, and hippocampus.23 In experimental cases, a more severe involvement of central grey matter (periaqueductal grey) and rostral colliculus but not the vestibular nuclear complex is observed. Additional brain areas, including the olfactory areas, amygdalae, hippocampi and dorsal horns of spinal cords, are severely involved. Ventral and dorsal roots do not show major pathological changes. In experimental H-type BSE, vacuolar changes are generally observed in all the brain areas. The major vacuolation appears in the thalamic nuclei and neuropil of the central gray matter of the midbrain, and mild vacuolation is found in the caudal cerebral and cerebellar cortices. In the vestibular and pontine nuclei, spongy changes are not as prominent as in the other brainstem nuclei.86
More sensitive diagnostic tests have been developed since based on the detection of disease-associated prion protein (PrPSc) or its proteinase-resistant form with anti-PrP monoclonal or polyclonal antibodies. A variety of methods are available such as immunohistochemistry (IHC), Western blotting (WB), enzyme-linked immunosorbent assay (ELISA), and conformation-dependent immunoassay (CDI)42 applied to the same target area (obex). Tissues for examination are either formalin-fixed (for IHC) or fresh (for WB, ELISA, and CDI).
Immunohistochemistry (IHC)
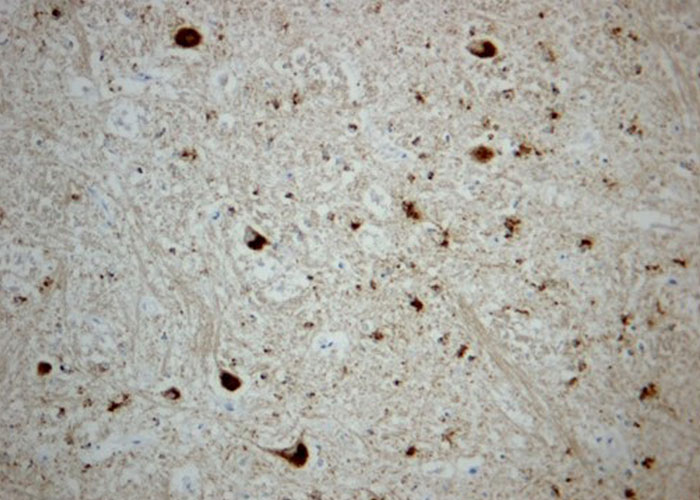
Immunohistochemical analysis should be performed on paraffin embedded brain tissues in order to detect the presence of PrPSc accumulations. The samples need to be deparaffinated, rehydrated, pretreated with 98 per cent formic acid and autoclaved at 121°C, then incubated at 4°C with a monoclonal primary antibody, incubated with avidin-biotin-peroxydase, reacted with chromogen 3-3’ diaminobenzidine (DAB), and counterstained with Mayer’s hemalum. The same neuroanatomical areas as for histopathological confirmation are assessed to confirm BSE: NST and NSTV (Figure 7). Different immunohistochemical types of PrPSc deposition can be observed in the brain of classical BSE affected cattle:22
- glial type labelling with PrPSc deposits branching out from the nucleus of glial cells on their processes conferring them a stellate appearance is predominantly in central grey matter and cerebral lamina and also within medial pontine nuclei in cerebral cortex, thalamus and obex;
- granular type that is characterized by granular PrPSc accumulations in the neuropil is commonly found in the neuropil of grey matter nuclei such as dorsal motor nucleus of vagus nerve (DMV), NST and in thalamic nuclei;
- intraneuronal type with PrPSc immunoreactivity throughout the neuronal cytoplasm is often observed in DMV, reticular formation, olivary nuclei, vestibular complex, pontine and thalamic nuclei and hypothalamus;
- perineuronal type, consisting in PrPSc deposits around individual neuronal perikarya and neuritis in caudate and putamen nuclei of basal ganglia and in DMV;
- linear tract characterized by PrPSc deposits along neuronal processes in particular at the level of reticular formation of the brainstem;
- coalescing type seemingly arising from the merging of granular PrPSc deposits to form amorphous or mesh-like masses and intraglial type with fine punctate PrPSc adjacent to glial nuclei (Figure 8).
In natural cases of L-type BSE, a distinctive feature is the presence of PrPSc deposition in the more rostral portions of the brain rather than occurs in classical BSE. At the level of the brainstem, the prevalent PrPSc deposition patterns are the punctate and granular types, which are mildly present in the hypoglossal and olivary nucleus and moderately present at the level of DMV nucleus, NST, NSTV and reticular formation. Glial, intraneuronal, perineuronal and linear tracts are also frequent in ‘bovine amyloidotic spongiform encephalopathy’(BASE) cases in different brain areas. Another characteristic of L-type BSE cases is the presence of PrP-positive amyloid plaques; they appear as dense, unicentric, or less frequently multicentric round structures up to 25 μm in diameter with a pale core and a dark radial periphery, which gave rise to the name ‘bovine amyloidotic spongiform encephalopathy’ (BASE).23 They are predominantly located in the thalamus, subcortical white matter, in deeper layers of cerebral cortexes and in the olfactory bulb (Figure 9). In experimental BASE/L-type BSE cattle, abundant amyloid PrP-plaques are observed in subcortical white matter and in deep grey nuclei, as observed in natural BASE cases. No PrP-plaques are seen in the olfactory glomeruli, the cerebellum or the spinal cord. Perineuronal pattern of PrPSc is also seen in ventral horn neurons of the spinal cord and in the dorsal root ganglion cells.
With regards to natural H-type BSE, in the brainstem, granular, intraneuronal, linear, intraglial and punctate PrPSc deposits are the most characteristic types, mainly detected at the level of the DMNV, NST, NSTV and in the reticular formation; however there is some variability in PrPSc distribution among different H-BSE cases93 (Figure 10). Regarding experimental H-type BSE, large amounts of PrPSc are diffusely deposited in the cerebral cortex, basal ganglia, thalamus, hypothalamus, brainstem, and spinal cord. The most conspicuous type of PrPSc deposition is fine or coarse particulate-type deposition in the neuropil of the gray matter throughout the brain and spinal cord. Linear, perineuronal, and intraneuronal types of PrPSc staining are observed in the cerebral cortex, basal ganglia, thalamus, and brainstem. Glial type PrPSc deposition is predominantly identified in the cerebral cortex, basal ganglia, thalamus, hypothalamus, and hippocampus and often in the cerebellar cortex, but is not visible in the brainstem and spinal cord. Intraglial-type PrPSc deposition is very consistent throughout the white matter of the CNS and spinal cord. Some animals show the presence of PrPSc -positive plaques scattered throughout the cerebral white matter.56
Western Immunoblot
Discriminatory Western blots are used to distinguish the different types of BSE by comparing the migration pattern and the reactions to different antibodies directed towards the core and N-terminal region of the prion protein after digestion of the sample with proteinase K.58 H-type BSE is characterized by the migration of the proteinase-treated prion protein (PrPres) to a higher position compared to classical BSE whereas PrPres of L-type BSE migrates to a lower position. In addition, the proportion of diglycosylated PrPres may be smaller in atypical BSE compared to classical BSE isolates. However, this may be less obvious in samples with a high total PrPres content.70
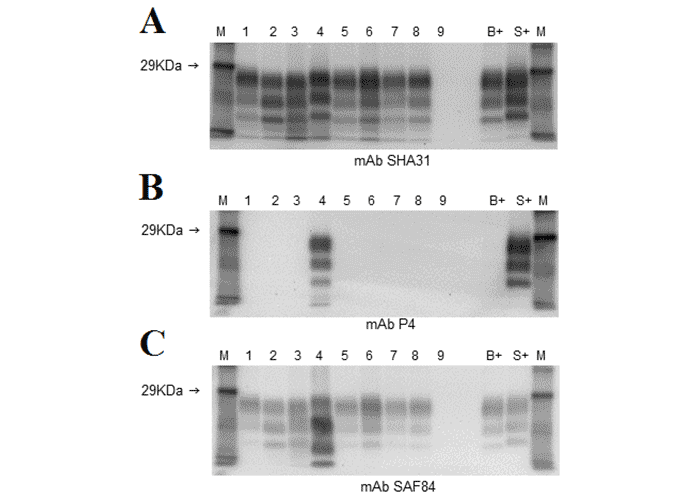
Figure 11 Discriminatory Western blots using three different monoclonal antibodies (mAb) to detect PrPres after digestion of the sample with proteinase K. Lanes 1, 5-8: classical BSE brain pool, lanes 2 and 3: L-type BSE (single case, diluted for lane 2), lane 4: H-type BSE (single case), lane 9: BSE-negative sample; B+ classical BSE positive control, S+: classical scrapie control (sheep), M: molecular mass marker. At least three bands should normally be visible, corresponding to a diglycosylated (top), monoglycosylated (second from top) and unglycosylated (third from top) peptide band (Figure 11A). H-type BSE is detected as characteristic four banded profile with mAb SAF84 (Figure 11C, lane 4) and is also detected with the N-terminal mAb P4 (Figure 11B, lane 4), similar to classical scrapie in sheep but unlike the other BSE types. L-type BSE produces a lower molecular mass than classical BSE for the unglycosylated band, which is more obvious when diluted (Figure 11A, lane 2) whilst H-type BSE produces a higher molecular mass for the unglycosylated band (Figure 11A, lane 4). (Courtesy by Sharon Everitt, APHA Weybridge)
More recently, ultrasensitive tests (protein misfolding cyclic amplification [PMCA] and real time quaking induced conversion [RT-QuIC]) have been developed that detect PrPSc in tissues at much lower concentrations. Both in vitro methods make use of the ability of the prion to convert the physiological (cellular) form of PrP (PrPC) into the abnormal form using alternate cycles of incubating and shaking. These are more sensitive than the other PrPSc -based tests.55, 106 They have also been successfully applied to brain samples from atypical BSE cases79, 83, 92 but are technically more challenging and spontaneous generation of prions may occur. This poses a problem for future use in BSE surveillance.
Biological strain-typing is still the gold-standard of aetiological diagnosis. Historically, inbred strains (RIII, C57Bl, VM) of mice were used, which presented with uniform brain spongiform lesion profiles when inoculated with classical BSE isolates.39, 44 More recently, transgenic mouse models have been created, which over-express bovine PrPC. These are more sensitive than wild-type mice and may even be more sensitive than the original species.45 However, a mouse bioassay is only available in specialist laboratories, is expensive, time-consuming and is thus not practical for routine use. It is usually reserved to studying new BSE types, such as atypical BSE,21, 111 confirming a potentially new BSE type100 or characterizing strains when BSE is detected for the first time in a country.
Wherever practical, some fresh brain, ideally from the brainstem, should be collected and frozen at -80˚C for possible subsequent transmission studies.
BSE Surveillance
Cattle populations can be monitored through passive and active surveillance programs. Passive surveillance relies on reporting of clinical suspects by the farmer or veterinarian, which depends on the willingness to report the disease and familiarisation with the clinical presentation. It is thus not sufficient to determine the true disease prevalence (Ducrot and others 2008). Suspect cattle are tested for the disease using confirmatory tests: histopathology (haematoxylin and eosin [H&E]), IHC, WB or demonstration of characteristic fibrils (SAF) by electron microscopy. If a brainstem sample tests negative, the OIE’s Manual of Diagnostic Tests and Vaccines for Terrestrial Animals84 recommends that the entire brain of the animal be tested to establish a differential diagnosis.
For active surveillance, healthy slaughter cattle or cattle at risk, i.e. cattle that have died or been killed on farm or during transport, fallen stock, and sick cattle sent for emergency slaughter, should be tested for BSE. Samples collected during active BSE surveillance are screened in Europe with approved rapid tests, in accordance with Regulation (EC) No 999/2001 on the prevention, control and eradication of certain TSEs35 and its amendments. These immunoassays are generally highly sensitive with a quick turnaround time, which is why they are used as initial screening tests. In inconclusive or positive cases, the sample needs to be tested using confirmatory tests, such as IHC and WB, although testing requirements vary outside Europe. The WB is generally used for confirmatory testing of autolysed brain material.
The first stage of all the current TSE diagnostic or screening tests involves the sampling of the brain and the subsequent examination of the sampled tissue. In particular, the minimum sampling requirement is the brainstem, at the level of the obex. This area can be accessed through the foramen magnum using a proprietary sampling spoon.
The quantity of tissue taken for testing should be sufficient to provide a hemisection of fresh obex for the initial rapid test immunoassay and confirmatory testing by WB if needed and a fixed cross-section, or hemi-section of obex, for confirmatory IHC and haematoxylin and eosin (H&E) staining. In general, correct diagnosis is dependent on several factors, such as sample quality, appropriate anatomical location, expertise in the tests and quality of the test kits. It is considered good practice to use two different tests to detect and confirm disease and to apply a third test such as WB or IHC if the initial two tests produce discrepant results.
For a more detailed description of the tests, their use, advantages or disadvantages and sampling techniques the reader is referred to the OIE manual’s chapter 2.4.5. on BSE.84
Differential diagnosis
The clinical signs of BSE, particularly over-reactivity, are distinctive but not pathognomonic. Progression over a period of weeks or months may serve to eliminate those nervous diseases with a short clinical phase like hypomagnesaemia or the nervous form of ketosis (nervous acetonemia). They can also be eliminated by serum analysis or response to treatment. Listeriosis is often associated with silage feeding and often presents with vestibular system dysfunction and other cranial nerve deficits (see Listeriosis). It is also usually febrile. In rare cases, animals may suffer from BSE and another neurological disease,69 which complicates the diagnosis.
Suspect clinical cases of BSE need to be confirmed by examination of the brain in a suitable laboratory. Neuropathological findings of suspect cases have included focal spongiosis of white matter (possibly related to hepatic dysfunction), listeriosis, suppurative thrombo-embolic encephalitis caused by Histophilus somni and other encephalitides, polioencephalomalacia (Vitamin B1 deficiency) and tumours.61, 113 An overview of neurological diseases occurring in western European cattle relevant to BSE was provided by Saegerman et al.98 Another disease in adult cattle characterized by BSE-like signs, termed ‘idiopathic brain stem neuronal necrosis and hippocampal sclerosis’ was described in Scotland63 but whether this is a further prion disease remains uncertain.62 Various surveys carried out in different countries revealed that the majority of suspect animals had no significant lesions in the brain. It is possible that some of these suffered from neurological diseases that do not cause changes visible by light microscopy, e.g. ketosis, hypomagnesaemia, spinal cord diseases, or suffered from BSE that was undetectable prior to the use of PrPSc markers for diagnosis. However, it is likely that these cases were erroneously attributed to BSE.69
Diagnosis of BSE in downer cows, a syndrome that has a multiple aetiology27 is more difficult than if cows are standing and able to move. Nevertheless, careful observation, in combination with the clinical history and supplemented where necessary by clinical biochemistry, can still enable a tentative alternative diagnosis to BSE.
Chemical and plant toxins create a more difficult diagnostic scenario, but groups of animals are more likely to be affected rather than singletons. Hyperammonaemia caused by feeding excessive non-protein nitrogen or ammoniated feed produces excitation and muscle tremors that may be confused with BSE although this is an acute disease.49
In other parts of the world, southern Africa for example, more specific differential diagnoses should be considered. These may include heartwater and rabies although, unlike BSE, these tend to be diseases with an acute course. In regard to plant poisonings and mycotoxicoses, the following should be considered: diploidiosis, albiziosis, cynanchosis and poisoning by Melica decumbens, Solanum (kwebense) tettense, Cotula (Matricaria) nigellifolia (atypical BSE dull syndrome-like), Cestrum laevigatum and mycotoxins (e.g. Aspergillus clavatus), which cause nervous signs.66
Control
Bovine spongiform encephalopathy is a notifiable or reportable disease and there is no prophylaxis or treatment for the disease. Animals suspected of suffering from BSE should be killed and the brain collected promptly for laboratory examination according to the methods recommended in the OIE Manual.84 All parts of the carcass not required for confirmation of diagnosis or authorized research in an approved laboratory should be destroyed, ideally by incineration, so they can enter no food or feed chain.
Control of BSE is the responsibility of the competent veterinary authority (usually the State Veterinary Service) according to national law and international recommendations. The latter are provided by the OIE’s Terrestrial Animal Health Code.85 This Code also lists the criteria recommended to minimize trade-risk posed by live cattle or commodities derived from cattle. Three categories for BSE disease status have been defined by the OIE:
- Negligible risk
- Controlled risk
- Undetermined risk
To be officially recognized as having a BSE risk status by the OIE, the member state has to submit a questionnaire as outlined in Chapter 1.6. of the OIE Terrestrial Animal Health Code and comply with all requirements specified in chapter 11.4 of the code.85 An application for official recognition by the OIE of free status is not possible for BSE, unlike for other diseases. BSE risk status of the cattle population is determined based on the outcome of a risk assessment on the likelihood that the BSE agent has been introduced or been present in a country (e.g. by importing cattle, cattle by-products or meat and bone meal) and about the likelihood of exposure (e.g. by looking at the processes of carcass removal and feed production and the level of BSE surveillance). A points system is used and countries have to reach certain points depending on the cattle population within a country to determine or maintain the BSE risk status. Point values are then assigned to each sample according to the probability of detecting infection in the subpopulation from which the sample was collected, with the highest point values given to clinical suspects, followed by casualty slaughter, fallen stock and lastly routine slaughter cattle examined.
Although all these surveillance streams contribute to the detection of BSE, surveillance of healthy slaughter cattle is the least effective and has been largely abandoned in the EU. It has been shown from experimental studies that the time point at which PrPSc was detectable in the obex in 50 per cent of cattle challenged orally with 1g of BSE-infected brainstem was only 1.7 months before clinical onset.4 This suggests that a large proportion of infected but clinically healthy cattle may be missed.
Additional measures to prevent exposure to BSE are the removal of specified risk material (e.g. brain, spinal column) during the dressing of carcasses and the prohibition of feeding specified risk material or processed animal protein, such as mammalian MBM, to ruminants or to the same species it was derived from.
Following an outbreak of classical BSE, different eradication measures can be applied depending on the country where the case was diagnosed, viz. culling of the index case only, whole-herd cull on the source farm and/ or the farm where the index case was kept. This may be extended to other animals on the farm, culling of the “feed cohort” that could have been exposed to the same feed as the index case or culling of the “birth cohort”, which are cattle born one year prior and one year after the index case.51 Even within countries these eradication measures may change given the decline in the number of BSE cases. For example in the UK initially only the index case was culled, subsequently replaced by feed and cohort culls. This obviously requires a robust livestock identification and movement recording system so that animals and relevant farms can be traced.
References
- AGERHOLM, J. S., KROGH, H. V., NIELSEN, T. K., AMMENDRUP, S. & DALSGAARD, H., 1993. A case of bovine spongiform encephalopathy in Denmark. Acta Veterinaria Scandinavica, 34, 99-100.
- ANDERSON, R. M., DONNELLY, C. A., FERGUSON, N. M., WOOLHOUSE, M. E. J., WATT, C. J., UDY, H. J., MAWHINNEY, S., DUNSTAN, S. P., SOUTHWOOD, T. R. E., WILESMITH, J. W., RYAN, J. B. M., HOINVILLE, L. J., HILLERTON, J. E., AUSTIN, A. R. & WELLS, G. A. H., 1996. Transmission dynamics and epidemiology of BSE in British cattle. Nature, 382, 779-788.
- ARNOLD, M. E., HAWKINS, S. A., GREEN, R., DEXTER, I. & WELLS, G. A., 2009. Pathogenesis of experimental bovine spongiform encephalopathy (BSE): estimation of tissue infectivity according to incubation period. Veterinary Research, 40, 8.
- ARNOLD, M. E., RYAN, J. B. M., KONOLD, T., SIMMONS, M. M., SPENCER, Y. I., WEAR, A., CHAPLIN, M., STACK, M., CZUB, S., MUELLER, R., WEBB, P. R., DAVIS, A., SPIROPOULOS, J., HOLDAWAY, J., HAWKINS, S. A. C., AUSTIN, A. R. & WELLS, G. A. H., 2007. Estimating the temporal relationship between PrPSc detection and incubation period in experimental bovine spongiform encephalopathy of cattle. Journal of General Virology, 88, 3198-3208.
- ARNOLD, M. E., SIMONS, R. R. L., HOPE, J., GIBBENS, N. & ADKIN, A. L., 2017. Is there a decline in bovine spongiform encephalopathy cases born after reinforced feed bans? A modelling study in EU member states. Epidemiology and Infection, 145, 2280-2286.
- ARNOLD, M. E. & WILESMITH, J. W., 2004. Estimation of the age-dependent risk of infection to BSE of dairy cattle in Great Britain. Preventive Veterinary Medicine, 66, 35-47.
- AUSTIN, A. R., HAWKINS, S. A. C., KELAY, N. S. & SIMMONS, M. M., 1994. New observations on the clinical signs of BSE and scrapie. Transmissible spongiform encephalopathies. Proceedings of a consultation on BSE with the Scientific Veterinary Committee of the Commission of the European Communities: 14-15 September 1993; Brussels. R. BRADLEY and B. MARCHANT. Brussels, European Commission, Agriculture. 277-287.
- AUSTIN, A. R., SIMMONS, M. M. & WELLS, G. A. H., 1997. Pathological temperament changes in bovines. Irish Veterinary Journal, 50, 304-309.
- BALKEMA-BUSCHMANN, A., FAST, C., KAATZ, M., EIDEN, M., ZIEGLER, U., MCINTYRE, L., KELLER, M., HILLS, B. & GROSCHUP, M. H., 2011. Pathogenesis of classical and atypical BSE in cattle. Preventive Veterinary Medicine, 102, 112-117.
- BALKEMA-BUSCHMANN, A., PRIEMER, G., KELLER, M., MAZZA, M., HILLS, B. & GROSCHUP, M. H., 2016. Intra- and interspecies transmission of atypical BSE − what can we learn from it? Food Safety, 4, 121-129.
- BALKEMA-BUSCHMANN, A., ZIEGLER, U., MCINTYRE, L., KELLER, M., HOFFMANN, C., ROGERS, R., HILLS, B. & GROSCHUP, M. H., 2011. Experimental challenge of cattle with German atypical bovine spongiform encephalopathy (BSE) isolates. Journal of Toxicology and Environmental Health - Part A, 74, 103-109.
- BÉRINGUE, V., ANDRÉOLETTI, O., LE DUR, A., ESSALMANI, R., VILOTTE, J.-L., LACROUX, C., REINE, F., HERZOG, L., BIACABÉ, A.-G., BARON, T., CARAMELLI, M., CASALONE, C. & LAUDE, H., 2007. A bovine prion acquires an epidemic bovine spongiform encephalopathy strain-like phenotype on interspecies transmission. Journal of Neuroscience, 27, 6965-6971.
- BIACABE, A. G., LAPLANCHE, J. L., RYDER, S. & BARON, T., 2004. Distinct molecular phenotypes in bovine prion diseases. EMBO Reports, 5, 110-114.
- BRADLEY, R., 1996. Experimental transmission of bovine spongiform encephalopathy. Transmissible subacute spongiform encephalopathies: prion diseases L. COURT and B. DODET. Paris, Elsevier. 51-56.
- BRAUN, U., SCHICKER, E. & HÖRNLIMANN, B., 1998. Diagnostic reliability of clinical signs in cows with suspected bovine spongiform encephalopathy. Veterinary Record, 143, 101-105.
- BRAUN, U., SCHICKER, E., PUSTERLA, N. & SCHÖNMANN, M., 1998. Klinische Befunde bei 50 Kühen mit boviner spongiformer Enzephalopathie (BSE) [Clinical findings in 50 cows with bovine spongiform encephalopathy (BSE)]. Berliner und Münchener Tierärztliche Wochenschrift, 111, 27-32.
- BRUCE, M. E., BOYLE, A. & MCCONNELL, I., 2004. TSE strain typing in mice. Techniques in prion research. S. LEHMANN and J. GRASSI. Basel, Birkhäuser Verlag. 132-146.
- BRUCE, M. E., WILL, R. G., IRONSIDE, J. W., MCCONNELL, I., DRUMMOND, D., SUTTIE, A., MCCARDLE, L., CHREE, A., HOPE, J., BIRKETT, C., COUSENS, S., FRASER, H. & BOSTOCK, C. J., 1997. Transmissions to mice indicate that 'new variant' CJD is caused by the BSE agent. Nature, 389, 498-501.
- BUSCHMANN, A., GRETZSCHEL, A., BIACABÉ, A. G., SCHIEBEL, K., CORONA, C., HOFFMANN, C., EIDEN, M., BARON, T., CASALONE, C. & GROSCHUP, M. H., 2006. Atypical BSE in Germany - Proof of transmissibility and biochemical characterization. Veterinary Microbiology, 117, 103-116.
- BUSCHMANN, A. & GROSCHUP, M. H., 2005. Highly bovine spongiform encephalopathy-sensitive transgenic mice confirm the essential restriction of infectivity to the nervous system in clinically diseased cattle. Journal of Infectious Diseases, 192, 934-942.
- CAPOBIANCO, R., CASALONE, C., SUARDI, S., MANGIERI, M., MICCOLO, C., LIMIDO, L., CATANIA, M., ROSSI, G., DI, F. G., GIACCONE, G., BRUZZONE, M. G., MINATI, L., CORONA, C., ACUTIS, P., GELMETTI, D., LOMBARDI, G., GROSCHUP, M. H., BUSCHMANN, A., ZANUSSO, G., MONACO, S., CARAMELLI, M. & TAGLIAVINI, F., 2007. Conversion of the BASE prion strain into the BSE strain: the origin of BSE? PLoS Pathogens, 3, e31.
- CASALONE, C., CARAMELLI, M., CRESCIO, M. I., SPENCER, Y. I. & SIMMONS, M. M., 2006. BSE immunohistochemical patterns in the brainstem: a comparison between UK and Italian cases. Acta Neuropathologica, 111, 444-449.
- CASALONE, C., ZANUSSO, G., ACUTIS, P., FERRARI, S., CAPUCCI, L., TAGLIAVINI, F., MONACO, S. & CARAMELLI, M., 2004. Identification of a second bovine amyloidotic spongiform encephalopathy: Molecular similarities with sporadic Creutzfeldt-Jakob disease. Proceedings of the National Academy of Sciences of the United States of America, 101, 3065-3070.
- CLARK, W. W., HOURRIGAN, J. L. & HADLOW, W. J., 1995. Encephalopathy in cattle experimentally infected with the scrapie agent. American Journal of Veterinary Research, 56, 606-612.
- CLAUSS, M., SAUTER-LOUIS, C., CHAHER, E., POTTGIESSER, C., GOEBEL, S., SELHORST, T., WICHMANN, H. E., KLEE, W. & KIENZLE, E., 2006. Investigations of the potential risk factors associated with cases of bovine spongiform encephalopathy in Bavaria, Germany. Veterinary Record, 158, 509-13.
- COMOY, E. E., CASALONE, C., LESCOUTRA-ETCHEGARAY, N., ZANUSSO, G., FREIRE, S., MARCÉ, D., AUVRÉ, F., RUCHOUX, M. M., FERRARI, S., MONACO, S., SALÈS, N., CARAMELLI, M., LEBOULCH, P., BROWN, P., LASMÉZAS, C. I. & DESLYS, J. P., 2008. Atypical BSE (BASE) transmitted from asymptomatic aging cattle to a primate. PLoS One, 3, e3017.
- CORREA, M. T., ERB, H. N. & SCARLETT, J. M., 1993. Risk factors for downer cow syndrome. Journal of Dairy Science, 76, 3460-3.
- CUTLIP, R. C., MILLER, J. M., HAMIR, A. N., PETERS, J., ROBINSON, M. M., JENNY, A. L., LEHMKUHL, H. D., TAYLOR, W. D. & BISPLINGHOFF, F. D., 2001. Resistance of cattle to scrapie by the oral route. Canadian Journal of Veterinary Research, 65, 131-132.
- CUTLIP, R. C., MILLER, J. M., RACE, R. E., JENNY, A. L., KATZ, J. B., LEHMKUHL, H. D., DEBEY, B. M. & ROBINSON, M. M., 1994. Intracerebral transmission of scrapie to cattle. Journal of Infectious Diseases, 169, 814-820.
- DEFAWEUX, V., DORBAN, G., ANTOINE, N., PIRET, J., GABRIEL, A., JACQMOT, O., FALISSE-POIRIER, N., FLANDROY, S., ZORZI, D. & HEINEN, E., 2007. Neuroimmune connections in jejunal and ileal Peyer’s patches at various bovine ages: potential sites for prion neuroinvasion. Cell and Tissue Research, 329, 35-44.
- DENNY, G. O. & HUESTON, W. D., 1997. Epidemiology of bovine spongiform encephalopathy in Northern Ireland 1988 to 1995. Veterinary Record, 140, 302-306.
- DUCROT, C., ARNOLD, M., DE KOEIJER, A., HEIM, D. & CALAVAS, D., 2008. Review on the epidemiology and dynamics of BSE epidemics. Veterinary Research, 39, 15.
- EFSA, 2011. Scientific opinion on the revision of the quantitative risk assessment (QRA) of the BSE risk posed by processed animal proteins (PAPs). EFSA Journal, 9, 1947.
- ESPINOSA, J. C., MORALES, M., CASTILLA, J., ROGERS, M. & TORRES, J. M., 2007. Progression of prion infectivity in asymptomatic cattle after oral bovine spongiform encephalopathy challenge. Journal of General Virology, 88, 1379-1383.
- EUROPEAN PARLIAMENT & COUNCIL OF THE EUROPEAN UNION, 2001. Regulation (EC) No 999/2001 of the European Parliament and of the council of 22 May 2001 laying down rules for the prevention, control and eradication of certain transmissible spongiform encephalopathies. Brussels. https://publications.europa.eu/en/publication-detail/-/publication/cba1d322-3457-4545-a539-5b38936b3b15/language-en.
- EVEREST, S. J., THORNE, L. T., HAWTHORN, J. A., JENKINS, R., HAMMERSLEY, C., RAMSAY, A. M., HAWKINS, S. A., VENABLES, L., FLYNN, L., SAYERS, R., KILPATRICK, J., SACH, A., HOPE, J. & JACKMAN, R., 2006. No abnormal prion protein detected in the milk of cattle infected with the bovine spongiform encephalopathy agent. Journal of General Virology, 87, 2433-2441.
- FAST, C., KELLER, M., BALKEMA-BUSCHMANN, A., HILLS, B. & GROSCHUP, M. H., 2013. Complementary studies detecting classical bovine spongiform encephalopathy infectivity in jejunum, ileum and ileocaecal junction in incubating cattle. Veterinary Research, 44, 123-123.
- FRANZ, M., EIDEN, M., BALKEMA-BUSCHMANN, A., GREENLEE, J., SCHATZL, H., FAST, C., RICHT, J., HILDEBRANDT, J. P. & GROSCHUP, M. H., 2012. Detection of PrPSc in peripheral tissues of clinically affected cattle after oral challenge with bovine spongiform encephalopathy. Journal of General Virology, 93, 2740-2748.
- FRASER, H., BRUCE, M. E., CHREE, A., MCCONNELL, I. & WELLS, G. A. H., 1992. Transmission of bovine spongiform encephalopathy and scrapie to mice. Journal of General Virology, 73, 1891-1897.
- FUKUDA, S., IWAMARU, Y., IMAMURA, M., MASUJIN, K., SHIMIZU, Y., MATSUURA, Y., SHU, Y., KURACHI, M., KASAI, K., MURAYAMA, Y., ONOE, S., HAGIWARA, K., SATA, T., MOHRI, S., YOKOYAMA, T. & OKADA, H., 2009. Intraspecies transmission of L-type-like bovine spongiform encephalopathy detected in Japan. Microbiology and Immunology, 53, 704-707.
- GAVIER-WIDEN, D., NOREMARK, M., LANGEVELD, J. P. M., STACK, M., BIACABE, A. G., VULIN, J., CHAPLIN, M., RICHT, J. A., JACOBS, J., ACIN, C., MONLEON, E., RENSTROM, L., KLINGEBORN, B. & BARON, T. G. M., 2008. Bovine spongiform encephalopathy in Sweden: an H-type variant. Journal of Veterinary Diagnostic Investigation, 20, 2-10.
- GAVIER-WIDÉN, D., STACK, M. J., BARON, T., BALACHANDRAN, A. & SIMMONS, M., 2005. Diagnosis of transmissible spongiform encephalopathies in animals: a review. Journal of Veterinary Diagnostic Investigation, 17, 509-527.
- GOLDMANN, W., HUNTER, N., MARTIN, T., DAWSON, M. & HOPE, J., 1991. Different forms of the bovine PrP gene have five or six copies of a short, G-C-rich element within the protein-coding exon. Journal of General Virology, 72, 201-204.
- GREEN, R., HORROCKS, C., WILKINSON, A., HAWKINS, S. A. C. & RYDER, S. J., 2005. Primary isolation of the bovine spongiform encephalopathy agent in mice: agent definition based on a review of 150 transmissions. Journal of Comparative Pathology, 132, 117-131.
- GROSCHUP, M. H. & BUSCHMANN, A., 2008. Rodent models for prion diseases. Veterinary Research, 39, 32.
- GURGUL, A., POLAK, M. P., LARSKA, M. & SŁOTA, E., 2012. PRNP and SPRN genes polymorphism in atypical bovine spongiform encephalopathy cases diagnosed in Polish cattle. Journal of Applied Genetics, 53, 337-342.
- GURGUL, A. & SLOTA, E., 2007. Effect of bovine PRNP gene polymorphisms on BSE susceptibility in cattle. Folia biologica (Krakow), 55, 81-6.
- HAASE, B., DOHERR, M. G., SEUBERLICH, T., DRÖGEMÜLLER, C., DOLF, G., NICKEN, P., SCHIEBEL, K., ZIEGLER, U., GROSCHUP, M. H., ZURBRIGGEN, A. & LEEB, T., 2007. PRNP promoter polymorphisms are associated with BSE susceptibility in Swiss and German cattle. BMC Genetics, 8, 15.
- HALIBURTON, J. C. & MORGAN, S. E., 1989. Nonprotein nitrogen-induced ammonia toxicosis and ammoniated feed toxicity syndrome. Veterinary Clinics of North America. Food Animal Practice, 5, 237-49.
- HEATON, M. P., KEELE, J. W., HARHAY, G. P., RICHT, J. A., KOOHMARAIE, M., WHEELER, T. L., SHACKELFORD, S. D., CASAS, E., KING, D. A., SONSTEGARD, T. S., VAN TASSELL, C. P., NEIBERGS, H. L., CHASE, C. C., JR., KALBFLEISCH, T. S., SMITH, T. P., CLAWSON, M. L. & LAEGREID, W. W., 2008. Prevalence of the prion protein gene E211K variant in U.S. cattle. BMC Veterinary Research, 4, 25.
- HEIM, D. & MURRAY, N., 2004. Possibilities to manage the BSE epidemic: cohort culling versus herd culling – experiences in Switzerland. Prions. A challenge for science, medicine and the public health system. H. F. RABENAU, J. CINATL, and H. W. DOERR. Basel, Karger. 11, 186-192.
- HOFFMANN, C., ZIEGLER, U., BUSCHMANN, A., WEBER, A., KUPFER, L., OELSCHLEGEL, A., HAMMERSCHMIDT, B. & GROSCHUP, M. H., 2007. Prions spread via the autonomic nervous system from the gut to the central nervous system in cattle incubating bovine spongiform encephalopathy. Journal of General Virology, 88, 1048-1055.
- HOINVILLE, L., 1993. Clinical signs of reported cases of BSE and their analysis to aid in diagnosis. Cattle Practice, 1, 59-61.
- HOINVILLE, L. J., WILESMITH, J. W. & RICHARDS, M. S., 1995. An investigation of risk factors for cases of bovine spongiform encephalopathy born after the introduction of the 'feed ban'. Veterinary Record, 136, 312-318.
- HWANG, S., WEST GREENLEE, M. H., BALKEMA-BUSCHMANN, A., GROSCHUP, M. H., NICHOLSON, E. M. & GREENLEE, J. J., 2018. Real-time quaking-induced conversion detection of bovine spongiform encephalopathy prions in a subclinical steer. Frontiers in Veterinary Science, 4, 242.
- IULINI, B., COSTASSA, E. V., CORONA, C., MELONI, D., FAVOLE, A., MAZZA, M., CARAMELLI, M., MAURELLA, C. & CASALONE, C., 2017. Classical and atypical bovine spongiform encephalopathy: epidemiology, pathogenesis and diagnosis. Prion. Y. TUTAR. London, IntechOpen. 3-25.
- IWAMARU, Y., IMAMURA, M., MATSUURA, Y., MASUJIN, K., SHIMIZU, Y., SHU, Y., KURACHI, M., KASAI, K., MURAYAMA, Y., FUKUDA, S., ONOE, S., HAGIWARA, K., YAMAKAWA, Y., SATA, T., MOHRI, S., OKADA, H. & YOKOYAMA, T., 2010. Accumulation of L-type bovine prions in peripheral nerve tissues. Emerging Infectious Diseases, 16, 1151-1154.
- JACOBS, J. G., LANGEVELD, J. P. M., BIACABÉ, A.-G., ACUTIS, P. L., POLAK, M. P., GAVIER-WIDEN, D., BUSCHMANN, A., CARAMELLI, M., CASALONE, C., MAZZA, M., GROSCHUP, M., ERKENS, J. H. F., DAVIDSE, A., VAN ZIJDERVELD, F. G. & BARON, T., 2007. Molecular discrimination of atypical bovine spongiform encephalopathy strains from a geographical region spanning a wide area in Europe. Journal of Clinical Microbiology, 45, 1821-1829.
- JARRIGE, N., DUCROT, C., CAZEAU, G., MORIGNAT, E., LA BONNARDIÈRE, C. & CALAVAS, D., 2007. Case-control study on feed risk factors for BSE cases born after the feed ban in France. Veterinary Research, 38, 505-16.
- JAUMAIN, E., QUADRIO, I., HERZOG, L., REINE, F., REZAEI, H., ANDRÉOLETTI, O., LAUDE, H., PERRET-LIAUDET, A., HAÏK, S. & BÉRINGUE, V., 2016. Absence of evidence for a causal link between bovine spongiform encephalopathy strain variant L-BSE and known forms of sporadic Creutzfeldt-Jakob disease in human PrP transgenic mice. Journal of Virology, 90, 10867-10874.
- JEFFREY, M., 1992. A neuropathological survey of brains submitted under the Bovine Spongiform Encephalopathy Orders in Scotland. Veterinary Record, 131, 332-337.
- JEFFREY, M., PEREZ, B. B., MARTIN, S., TERRY, L. & GONZALEZ, L., 2008. Idiopathic Brainstem Neuronal Chromatolysis (IBNC): a novel prion protein related disorder of cattle? BMC Veterinary Research, 4, 38.
- JEFFREY, M. & WILESMITH, J. W., 1992. Idiopathic brainstem neuronal chromatolysis and hippocampal sclerosis: a novel encephalopathy in clinically suspect cases of bovine spongiform encephalopathy. Veterinary Record, 131, 359-362.
- JULING, K., SCHWARZENBACHER, H., WILLIAMS, J. L. & FRIES, R., 2006. A major genetic component of BSE susceptibility. BMC Biology, 4, 33.
- KAATZ, M., FAST, C., ZIEGLER, U., BALKEMA-BUSCHMANN, A., HAMMERSCHMIDT, B., KELLER, M., OELSCHLEGEL, A., MCINTYRE, L. & GROSCHUP, M. H., 2012. Spread of classic BSE prions from the gut via the peripheral nervous system to the brain. American Journal of Pathology, 181, 515-524.
- KELLERMAN, T. S., COETZER, J. A. W., NAUDÉ, T. W. & BOTHA, C. J., 2005. Plant poisonings and mycotoxicoses of livestock in southern Africa.
- KIMBERLIN, R. H., 1993. Clinical signs. Bovine spongiform encephalopathy. Rome, FAO. 31-32.
- KONOLD, T., ARNOLD, M. E., AUSTIN, A. R., CAWTHRAW, S., HAWKINS, S. A., STACK, M. J., SIMMONS, M. M., SAYERS, A. R., DAWSON, M., WILESMITH, J. W. & WELLS, G. A., 2012. Bovine spongiform encephalopathy: the effect of oral exposure dose on attack rate and incubation period in cattle - an update. BMC Research Notes, 5, 674.
- KONOLD, T., BONE, G., RYDER, S., HAWKINS, S. A. C., COURTIN, F. & BERTHELIN-BAKER, C., 2004. Clinical findings in 78 suspected cases of bovine spongiform encephalopathy in Great Britain. Veterinary Record, 155, 659-666.
- KONOLD, T., BONE, G. E., CLIFFORD, D., CHAPLIN, M. J., CAWTHRAW, S., STACK, M. J. & SIMMONS, M. M., 2012. Experimental H-type and L-type bovine spongiform encephalopathy in cattle: observation of two clinical syndromes and diagnostic challenges. BMC Veterinary Research, 8, 22.
- KONOLD, T., BONE, G. E. & SIMMONS, M. M., 2011. Time and frequency domain analysis of heart rate variability in cattle affected by bovine spongiform encephalopathy. BMC Research Notes, 4, 259.
- KONOLD, T., NONNO, R., SPIROPOULOS, J., CHAPLIN, M. J., STACK, M. J., HAWKINS, S. A., CAWTHRAW, S., WILESMITH, J. W., WELLS, G. A., AGRIMI, U., DI BARI, M. A., ANDREOLETTI, O., ESPINOSA, J. C., AGUILAR-CALVO, P. & TORRES, J. M., 2015. Further characterisation of transmissible spongiform encephalopathy phenotypes after inoculation of cattle with two temporally separated sources of sheep scrapie from Great Britain. BMC Research Notes, 8, 312.
- KONOLD, T., PHELAN, L. J., CLIFFORD, D., CHAPLIN, M. J., CAWTHRAW, S., STACK, M. J. & SIMMONS, M. M., 2014. The pathological and molecular but not clinical phenotypes are maintained after second passage of experimental atypical bovine spongiform encephalopathy in cattle. BMC Veterinary Research, 10, 243.
- KONOLD, T., SAYERS, A. R., SACH, A., BONE, G., VAN WINDEN, S., WELLS, G., SIMMONS, M., STACK, M., WEAR, A. & HAWKINS, S., 2010. Relationship between clinical signs and postmortem test status in cattle experimentally infected with the bovine spongiform encephalopathy agent. BMC Veterinary Research, 6, 53.
- KONOLD, T., SIVAM, S. K., RYAN, J., GUBBINS, S., LAVEN, R. & HOWE, M. J., 2006. Analysis of clinical signs associated with bovine spongiform encephalopathy in casualty slaughter cattle. Veterinary Journal, 171, 438-444.
- KONOLD, T., SPIROPOULOS, J., CHAPLIN, M. J., STACK, M. J., HAWKINS, S. A., WILESMITH, J. W. & WELLS, G. A., 2013. Unsuccessful oral transmission of scrapie from British sheep to cattle. Veterinary Record, 173, 118.
- LOMBARDI, G., CASALONE, C., D' ANGELO, A., GELMETTI, D., TORCOLI, G., BARBIERI, I., CORONA, C., FASOLI, E., FARINAZZO, A., FIORINI, M., GELATI, M., IULINI, B., TAGLIAVINI, F., FERRARI, S., CARAMELLI, M., MONACO, S., CAPUCCI, L. & ZANUSSO, G., 2008. Intraspecies transmission of BASE induces clinical dullness and amyotrophic changes. PLoS Pathogens, 4, e1000075.
- MACKENZIE, G. & WILL, R., 2017. Creutzfeldt-Jakob disease: recent developments. F1000Research, 6, 2053.
- MASUJIN, K., ORRU, C. D., MIYAZAWA, K., GROVEMAN, B. R., RAYMOND, L. D., HUGHSON, A. G. & CAUGHEY, B., 2016. Detection of atypical H-type bovine spongiform encephalopathy and discrimination of bovine prion strains by real-time quaking-induced conversion. Journal of Clinical Microbiology, 54, 676-86.
- MIDDLETON, D. J. & BARLOW, R. M., 1993. Failure to transmit bovine spongiform encephalopathy to mice by feeding them with extraneural tissues of affected cattle. Veterinary Record, 132, 545-7.
- MURAYAMA, Y., YOSHIOKA, M., MASUJIN, K., OKADA, H., IWAMARU, Y., IMAMURA, M., MATSUURA, Y., FUKUDA, S., ONOE, S., YOKOYAMA, T. & MOHRI, S., 2010. Sulfated dextrans enhance in vitro amplification of bovine spongiform encephalopathy PrPSc and enable ultrasensitive detection of bovine PrPSc. PLoS One, 5, e13152.
- NICOT, S., BENCSIK, A., MIGLIORE, S., CANAL, D., LEBOIDRE, M., AGRIMI, U., NONNO, R. & BARON, T., 2014. L-type bovine spongiform encephalopathy in genetically susceptible and resistant sheep: changes in prion strain or phenotypic plasticity of the disease-associated prion protein? Journal of Infectious Diseases, 209, 950-9.
- O'CONNOR, M. J., BISHOP, K., WORKMAN, R. G., MADDISON, B. C. & GOUGH, K. C., 2017. In vitro amplification of H-type atypical bovine spongiform encephalopathy by protein misfolding cyclic amplification. Prion, 11, 54-64.
- OIE, 2016. Chapter 2.4.5. http://www.oie.int/fileadmin/Home/eng/Health_standards/tahm/2.04.05_BSE.pdf. Bovine spongiform encephalopathy. Manual of diagnostic tests and vaccines for terrestrial animals. Paris, World Organisation for Animal Health. 1-14.
- OIE, 2018. Chapter 11.4. http://www.oie.int/fileadmin/Home/eng/Health_standards/tahc/current/chapitre_bse.pdf. Bovine spongiform encephalopathy. Terrestrial Animal Health Code. Paris, World Organisation for Animal Health. 1-16.
- OKADA, H., IWAMARU, Y., IMAMURA, M., MASUJIN, K., MATSUURA, Y., SHIMIZU, Y., KASAI, K., MOHRI, S., YOKOYAMA, T. & CZUB, S., 2011. Experimental H-type bovine spongiform encephalopathy characterized by plaques and glial- and stellate-type prion protein deposits. Veterinary Research, 42, 79.
- OKADA, H., IWAMARU, Y., IMAMURA, M., MIYAZAWA, K., MATSUURA, Y., MASUJIN, K., MURAYAMA, Y. & YOKOYAMA, T., 2017. Oral Transmission of L-Type Bovine Spongiform Encephalopathy Agent among Cattle. Emerging Infectious Diseases, 23, 284-287.
- OKADA, H., IWAMARU, Y., YOKOYAMA, T. & MOHRI, S., 2013. Immunohistochemical detection of disease-associated prion protein in the peripheral nervous system in experimental H-type bovine spongiform encephalopathy. Veterinary Pathology, 50, 659-63.
- OKADA, H., MIYAZAWA, K., FUKUDA, S., IWAMARU, Y., IMAMURA, M., MASUJIN, K., MATSUURA, Y., FUJII, T., FUJII, K., KAGEYAMA, S., YOSHIOKA, M., MURAYAMA, Y. & YOKOYAMA, T., 2014. The presence of disease-associated prion protein in skeletal muscle of cattle infected with classical bovine spongiform encephalopathy. Journal of Veterinary Medical Science, 76, 103-7.
- OKADA, H., MIYAZAWA, K., MASUJIN, K. & YOKOYAMA, T., 2016. Coexistence of two forms of disease-associated prion protein in extracerebral tissues of cattle infected with H-type bovine spongiform encephalopathy. Journal of Veterinary Medical Science, 78, 1189-93.
- ONO, F., TASE, N., KUROSAWA, A., HIYAOKA, A., OHYAMA, A., TEZUKA, Y., WADA, N., SATO, Y., TOBIUME, M., HAGIWARA, K., YAMAKAWA, Y., TERAO, K. & SATA, T., 2011. Atypical L-type bovine spongiform encephalopathy (L-BSE) transmission to cynomolgus macaques, a non-human primate. Japanese Journal of Infectious Diseases, 64, 81-4.
- ORRÚ, C. D., FAVOLE, A., CORONA, C., MAZZA, M., MANCA, M., GROVEMAN, B. R., HUGHSON, A. G., ACUTIS, P. L., CARAMELLI, M., ZANUSSO, G., CASALONE, C. & CAUGHEY, B., 2015. Detection and discrimination of classical and atypical L-type bovine spongiform encephalopathy by real-time quaking-induced conversion. Journal of Clinical Microbiology, 53, 1115-20.
- PORCARIO, C., HALL, S. M., MARTUCCI, F., CORONA, C., IULINI, B., PERAZZINI, A. Z., ACUTIS, P., HAMIR, A. N., LOIACONO, C. M., GREENLEE, J. J., RICHT, J. A., CARAMELLI, M. & CASALONE, C., 2011. Evaluation of two sets of immunohistochemical and Western blot confirmatory methods in the detection of typical and atypical BSE cases. BMC Research Notes, 4, 376.
- PRINCE, M. J., BAILEY, J. A., BARROWMAN, P. R., BISHOP, K. J., CAMPBELL, G. R. & WOOD, J. M., 2003. Bovine spongiform encephalopathy. Revue Scientifique et Technique - Office International des Epizooties, 22, 37-60.
- PRUSINER, S. B., 1995. The prion diseases. Scientific American, 272, 48-57.
- RITCHIE, D. L., BOYLE, A., MCCONNELL, I., HEAD, M. W., IRONSIDE, J. W. & BRUCE, M. E., 2009. Transmissions of variant Creutzfeldt-Jakob disease from brain and lymphoreticular tissue show uniform and conserved bovine spongiform encephalopathy-related phenotypic properties on primary and secondary passage in wild-type mice. Journal of General Virology, 90, 3075-3082.
- ROBINSON, M. M., HADLOW, W. J., KNOWLES, D. P., HUFF, T. P., LACY, P. A., MARSH, R. F. & GORHAM, J. R., 1995. Experimental infection of cattle with the agents of transmissible mink encephalopathy and scrapie. Journal of Comparative Pathology, 113, 241-251.
- SAEGERMAN, C., CLAES, L., DEWAELE, A., DESMECHT, D., ROLLIN, F., HAMOIR, J., GUSTIN, P., CZAPLICKI, G., BUGHIN, J., WULLEPIT, J., LAUREYNS, J., ROELS, S., BERKVENS, D., VANOPDENBOSCH, E. & THIRY, E., 2003. Differential diagnosis of neurologically expressed disorders in Western European cattle. Revue Scientifique et Technique - Office International des Epizooties, 22, 83-82.
- SAUTER-LOUIS, C., CLAUSS, M., CHAHER, E., KLEE, W., WICHMANN, H. E. & KIENZLE, E., 2006. Breed predisposition for BSE: epidemiological evidence in Bavarian cattle. Schweizer Archiv für Tierheilkunde, 148, 245-250.
- SERRA, F., DUDAS, S., TORRES, J. M., ANDERSON, R., OEVERMANN, A., ESPINOSA, J. C., CZUB, S. & SEUBERLICH, T., 2018. Presumptive BSE cases with an aberrant prion protein phenotype in Switzerland, 2011: Lack of prion disease in experimentally inoculated cattle and bovine prion protein transgenic mice. Transboundary and Emerging Diseases.
- SIMMONS, M. M., CHAPLIN, M. J., KONOLD, T., CASALONE, C., BECK, K. E., THORNE, L., EVERITT, S., FLOYD, T., CLIFFORD, D. & SPIROPOULOS, J., 2016. L-BSE experimentally transmitted to sheep presents as a unique disease phenotype. Veterinary Research, 47, 112.
- SIMMONS, M. M., HARRIS, P., JEFFREY, M., MEEK, S. C., BLAMIRE, I. W. H. & WELLS, G. A. H., 1996. BSE in Great Britain: Consistency of the neurohistopathological findings in two random annual samples of clinically suspect cases. Veterinary Record, 138, 175-177.
- SIMMONS, M. M., SPIROPOULOS, J., WEBB, P. R., SPENCER, Y. I., CZUB, S., MUELLER, R., DAVIS, A., ARNOLD, M. E., MARSH, S., HAWKINS, S. A. C., COOPER, J. A., KONOLD, T. & WELLS, G. A. H., 2011. Experimental classical bovine spongiform encephalopathy: definition and progression of neural PrP immunolabeling in relation to diagnosis and disease controls. Veterinary Pathology, 48, 948-963.
- SISÓ, S., ORDÓÑEZ, M., CORDON, I., VIDAL, E. & PUMAROLA, M., 2004. Distribution of PrPres in the brains of BSE-affected cows detected by active surveillance in Catalonia, Spain. Veterinary Record, 155, 524-525.
- SOHN, H. J., LEE, Y. H., GREEN, R. B., SPENCER, Y. I., HAWKINS, S. A. C., STACK, M. J., KONOLD, T., WELLS, G. A. H., MATTHEWS, D., CHO, I. S. & JOO, Y. S., 2009. Bone marrow infectivity in cattle exposed to the bovine spongiform encephalopathy agent. Veterinary Record, 164, 272-273.
- SOTO, C., ANDERES, L., SUARDI, S., CARDONE, F., CASTILLA, J., FROSSARD, M. J., PEANO, S., SAÁ, P., LIMIDO, L., CARBONATTO, M., IRONSIDE, J., TORRES, J. M., POCCHIARI, M. & TAGLIAVINI, F., 2005. Pre-symptomatic detection of prions by cyclic amplification of protein misfolding. FEBS Letters, 579, 638.
- STACK, M. J., MOORE, S. J., VIDAL-DIEZ, A., ARNOLD, M. E., JONES, E. M., SPENCER, Y. I., WEBB, P., SPIROPOULOS, J., POWELL, L., BELLERBY, P., THURSTON, L., COOPER, J., CHAPLIN, M. J., DAVIS, L. A., EVERITT, S., FOCOSI-SNYMAN, R., HAWKINS, S. A., SIMMONS, M. M. & WELLS, G. A., 2011. Experimental bovine spongiform encephalopathy: detection of PrP(Sc) in the small intestine relative to exposure dose and age. Journal of Comparative Pathology, 145, 289-301.
- SUARDI, S., VIMERCATI, C., CASALONE, C., GELMETTI, D., CORONA, C., IULINI, B., MAZZA, M., LOMBARDI, G., MODA, F., RUGGERONE, M., CAMPAGNANI, I., PICCOLI, E., CATANIA, M., GROSCHUP, M. H., BALKEMA-BUSCHMANN, A., CARAMELLI, M., MONACO, S., ZANUSSO, G. & TAGLIAVINI, F., 2012. Infectivity in skeletal muscle of cattle with atypical bovine spongiform encephalopathy. PLoS One, 7, e31449.
- TAYLOR, D. M., FERGUSON, C. E., BOSTOCK, C. J. & DAWSON, M., 1995. Absence of disease in mice receiving milk from cows with bovine spongiform encephalopathy. Veterinary Record, 136, 592.
- TERRY, L. A., MARSH, S., RYDER, S. J., HAWKINS, S. A. C., WELLS, G. A. H. & SPENCER, Y. I., 2003. Detection of disease-specific PrP in the distal ileum of cattle exposed orally to the agent of bovine spongiform encephalopathy. Veterinary Record, 152, 387-392.
- TORRES, J. M., ANDRÉOLETTI, O., LACROUX, C., PRIETO, I., LORENZO, P., LARSKA, M., BARON, T. & ESPINOSA, J. C., 2011. Classical bovine spongiform encephalopathy by transmission of H-type prion in homologous prion protein context. Emerging Infectious Diseases, 17, 1636-1644.
- TRANULIS, M. A., BENESTAD, S. L., BARON, T. & KRETZSCHMAR, H., 2011. Atypical prion diseases in humans and animals. Topics in Current Chemistry, 305, 23-50.
- WELLS, G. A., SAYERS, A. R. & WILESMITH, J. W., 1995. Clinical and epidemiological correlates of the neurohistology of cases of histologically unconfirmed, clinically suspect bovine spongiform encephalopathy. Veterinary Record, 136, 211-216.
- WELLS, G. A., SCOTT, A. C., JOHNSON, C. T., GUNNING, R. F., HANCOCK, R. D., JEFFREY, M., DAWSON, M. & BRADLEY, R., 1987. A novel progressive spongiform encephalopathy in cattle. Veterinary Record, 121, 419-420.
- WELLS, G. A. H., HANCOCK, R. D., COOLEY, W. A., RICHARDS, M. S., HIGGINS, R. J. & DAVID, G. P., 1989. Bovine spongiform encephalopathy: Diagnostic significance of vacuolar changes in selected nuclei of the medulla oblongata. Veterinary Record, 125, 521-524.
- WELLS, G. A. H., HAWKINS, S. A. C., GREEN, R. B., AUSTIN, A. R., DEXTER, I., SPENCER, Y. I., CHAPLIN, M. J., STACK, M. J. & DAWSON, M., 1998. Preliminary observations on the pathogenesis of experimental bovine spongiform encephalopathy (BSE): an update. Veterinary Record, 142, 103-106.
- WELLS, G. A. H., KONOLD, T., ARNOLD, M. E., AUSTIN, A. R., HAWKINS, S. A. C., STACK, M., SIMMONS, M. M., LEE, Y. H., GAVIER-WIDÉN, D., DAWSON, M. & WILESMITH, J. W., 2007. Bovine spongiform encephalopathy: the effect of oral exposure dose on attack rate and incubation period in cattle. Journal of General Virology, 88, 1363-1373.
- WELLS, G. A. H. & MCGILL, I. S., 1992. Recently described scrapie-like encephalopathies of animals: case definitions. Research in Veterinary Science, 53, 1-10.
- WELLS, G. A. H., SPIROPOULOS, J., HAWKINS, S. A. C. & RYDER, S. J., 2005. Pathogenesis of experimental bovine spongiform encephalopathy: preclinical infectivity in tonsil and observations on the distribution of lingual tonsil in slaughtered cattle. Veterinary Record, 156, 401-407.
- WHITAKER, C. J. & JOHNSON, C., 2017. A neurological syndrome. Cattle Practice, 25, 32-37.
- WILESMITH, J. W., HOINVILLE, L. J., RYAN, J. B. & SAYERS, A. R., 1992. Bovine spongiform encephalopathy: aspects of the clinical picture and analyses of possible changes 1986-1990. Veterinary Record, 130, 197-201.
- WILESMITH, J. W., RYAN, J. B. & ATKINSON, M. J., 1991. Bovine spongiform encephalopathy: epidemiological studies on the origin. Veterinary Record, 128, 199-203.
- WILESMITH, J. W., RYAN, J. B., STEVENSON, M. A., MORRIS, R. S., PFEIFFER, D. U., LIN, D., JACKSON, R. & SANSON, R. L., 2000. Temporal aspects of the epidemic of bovine spongiform encephalopathy in Great Britain: holding-associated risk factors for the disease. Veterinary Record, 147, 319-325.
- WILESMITH, J. W., RYAN, J. B. M., ARNOLD, M. E., STEVENSON, M. A. & BURKE, P. J., 2010. Descriptive epidemiological features of cases of bovine spongiform encephalopathy born after July 31, 1996 in Great Britain. Veterinary Record, 167, 279-286.
- WILESMITH, J. W., WELLS, G. A., CRANWELL, M. P. & RYAN, J. B., 1988. Bovine spongiform encephalopathy: epidemiological studies. Veterinary Record, 123, 638-644.
- WILL, R. G., IRONSIDE, J. W., ZEIDLER, M., COUSENS, S. N., ESTIBEIRO, K., ALPEROVITCH, A., POSER, S., POCCHIARI, M., HOFMAN, A. & SMITH, P. G., 1996. A new variant of Creutzfeldt-Jakob disease in the UK. Lancet, 347, 921-925.
- WINTER, M. H., ALDRIDGE, B. M., SCOTT, P. R. & CLARKE, M., 1989. Occurrence of 14 cases of bovine spongiform encephalopathy in a closed dairy herd. British Veterinary Journal, 145, 191-194.
- WRATHALL, A. E., BROWN, K. F. D., SAYERS, A. R., WELLS, G. A. H., SIMMONS, M. M., FARRELLY, S. S. J., BELLERBY, P., SQUIRRELL, J., SPENCER, Y. I., WELLS, M., STACK, M. J., BASTIMAN, B., PULLAR, D., SCATCHERD, J., HEASMAN, L., PARKER, J., HANNAM, D. A. R., HELLIWELL, D. W., CHREE, A. & FRASER, H., 2002. Studies of embryo transfer from cattle clinically affected by bovine spongiform encephalopathy (BSE). Veterinary Record, 150, 365-378.
- YOSHIKAWA, Y., 2008. Epidemiological study on BSE outbreak in Japan. Journal of Veterinary Medical Science, 70, 325-36.