- Infectious Diseases of Livestock
- Part 2
- Equine encephalitides caused by alphaviruses in the Western Hemisphere
- GENERAL INTRODUCTION: PARAMYXOVIRIDAE AND PNEUMOVIRIDAE
- Rinderpest
- Peste des petits ruminants
- Parainfluenza type 3 infection
- Bovine respiratory syncytial virus infection
- Hendra virus infection
- Paramyxovirus-induced reproductive failure and congenital defects in pigs
- Nipah virus disease
- GENERAL INTRODUCTION: CALICIVIRIDAE AND ASTROVIRIDAE
- Vesicular exanthema
- Enteric caliciviruses of pigs and cattle
- GENERAL INTRODUCTION: RETROVIRIDAE
- Enzootic bovine leukosis
- Jaagsiekte
- Visna-maedi
- Caprine arthritis-encephalitis
- Equine infectious anaemia
- GENERAL INTRODUCTION: PAPILLOMAVIRIDAE
- Papillomavirus infection of ruminants
- Papillomavirus infection of equids
- GENERAL INTRODUCTION: ORTHOMYXOVIRIDAE
- Equine influenza
- Swine influenza
- GENERAL INTRODUCTION: CORONAVIRIDAE
- Porcine transmissible gastroenteritis
- Porcine respiratory coronavirus infection
- Porcine epidemic diarrhoea
- Porcine haemagglutinating encephalomyelitis virus infection
- Porcine deltacoronavirus infection
- Bovine coronavirus infection
- Ovine coronavirus infection
- Equine coronavirus infection
- GENERAL INTRODUCTION: PARVOVIRIDAE
- Porcine parvovirus infection
- Bovine parvovirus infection
- GENERAL INTRODUCTION: ADENOVIRIDAE
- Adenovirus infections
- GENERAL INTRODUCTION: HERPESVIRIDAE
- Equid herpesvirus 1 and equid herpesvirus 4 infections
- Equid gammaherpesvirus 2 and equid gammaherpesvirus 5 infections
- Equine coital exanthema
- Infectious bovine rhinotracheitis/infectious pustular vulvovaginitis and infectious pustular balanoposthitis
- Bovine alphaherpesvirus 2 infections
- Malignant catarrhal fever
- Pseudorabies
- Suid herpesvirus 2 infection
- GENERAL INTRODUCTION: ARTERIVIRIDAE
- Equine viral arteritis
- Porcine reproductive and respiratory syndrome
- GENERAL INTRODUCTION: FLAVIVIRIDAE
- Bovine viral diarrhoea and mucosal disease
- Border disease
- Hog cholera
- Wesselsbron disease
- Louping ill
- West nile virus infection
- GENERAL INTRODUCTION: TOGAVIRIDAE
- Equine encephalitides caused by alphaviruses in the Western Hemisphere
- Old World alphavirus infections in animals
- Getah virus infection
- GENERAL INTRODUCTION: BUNYAVIRIDAE
- Diseases caused by Akabane and related Simbu-group viruses
- Rift Valley fever
- Nairobi sheep disease
- Crimean-Congo haemorrhagic fever
- GENERAL INTRODUCTION: ASFARVIRIDAE
- African swine fever
- GENERAL INTRODUCTION: RHABDOVIRIDAE
- Rabies
- Bovine ephemeral fever
- Vesicular stomatitis and other vesiculovirus infections
- GENERAL INTRODUCTION: REOVIRIDAE
- Bluetongue
- Ibaraki disease in cattle
- Epizootic haemorrhagic disease
- African horse sickness
- Equine encephalosis
- Palyam serogroup orbivirus infections
- Rotavirus infections
- GENERAL INTRODUCTION: POXVIRIDAE
- Lumpy skin disease
- Sheeppox and goatpox
- Orf
- Ulcerative dermatosis
- Bovine papular stomatitis
- Pseudocowpox
- Swinepox
- Cowpox
- Horsepox
- Camelpox
- Buffalopox
- GENERAL INTRODUCTION: PICORNAVIRIDAE
- Teschen, Talfan and reproductive diseases caused by porcine enteroviruses
- Encephalomyocarditis virus infection
- Swine vesicular disease
- Equine picornavirus infection
- Bovine rhinovirus infection
- Foot-and-mouth disease
- GENERAL INTRODUCTION: BORNAVIRIDAE
- Borna disease
- GENERAL INTRODUCTION: CIRCOVIRIDAE AND ANELLOVIRIDAE
- Post-weaning multi-systemic wasting syndrome in swine
- GENERAL INTRODUCTION: PRION DISEASES
- Scrapie
- Bovine spongiform encephalopathy
- Transmissible spongiform encephalopathies related to bovine spongiform encephalopathy in other domestic and captive wild species
Equine encephalitides caused by alphaviruses in the Western Hemisphere
This content is distributed under the following licence: Attribution-NonCommercial CC BY-NC  View Creative Commons Licence details here
View Creative Commons Licence details here

Equine encephalitides caused by alphaviruses in the Western Hemisphere
Previous author: E P J GIBBS
Current author:
E P J GIBBS - Retired Emeritus Professor, BVSc, PhD, FRCVS, College of Veterinary Medicine, University of Florida, Veterinary Academic Building V3-157, 2015 SW 16th Avenue, Gainesville, Florida, 32608, USA
Introduction
Several mosquito-borne alphaviruses, endemic to North, Central and South America, cause disease in horses, humans, and, on occasion, other domestic and wild animal species, particularly those that are not native to the New World. Prior to 1999, the principal viruses causing epidemics of encephalitis in horses and humans were eastern, western and Venezuelan equine encephalitis (EEE, WEE, VEE, respectively). The viruses of EEE, WEE and VEE are closely related and classified as alphaviruses within the Togaviridae family. In 1999, West Nile (WN) virus was introduced to North America, possibly from Israel by an unknown route. The virus has since spread throughout the Americas. It is considered endemic in North America and is recognized as a significant cause of encephalitis in horses, humans and other species. Since WN virus is classified within the Flaviridae family, it is discussed in a separate chapter (See West Nile virus infection).
Annual summaries of the incidence of disease caused by EEE,WEE and WN viruses in the USA are available on the web https://diseasemaps.usgs.gov/mapviewer/ Further information on infections caused by each of these viruses, particularly as it relates to human infection, can be found on the website of the Centers for Disease Control in the USA www.cdc.gov. The World Organization for Animal Health (also known as OIE) has information on the occurrence and diagnosis of these diseases worldwide. EEE, VEE and WN fever are “listed” diseases (notifiable). www.oie.int/wahis_2/public/wahid.php/Diseaseinformation/Diseaseoutbreakmaps
Before the recognition of EEE, WEE and VEE as antigenically distinct viruses, several epidemics of equine encephalitis were recorded in the western hemisphere. The first epidemic in horses was recognized in Massachusetts, USA, in 1831 and, in retrospect, was probably EEE. Once it had been established in 1933 that epidemics of encephalitis in horses in North America were mostly caused by either EEE or WEE viruses and were largely partitioned by geography, more accurate information became available. The largest recorded epidemic of EEE occurred in southern Louisiana and adjacent Texas in 1947. Over 14 000 horses* (*The term “horse” will be used to denote horses, mules, and donkeys except where qualified) were reported to have been affected, of which nearly 12 000 died. The case fatality rate of 83 per cent in this epidemic is typical of EEE in horses in North America.
The epidemic of encephalitis in horses in California that led to the recognition of WEE was estimated to have caused disease in 6 000 horses with a case fatality of 50 per cent. In another large epidemic in the 1940s in western Canada, the case fatality was lower, around 12 per cent, which is considered more typical of WEE.
Perhaps the best known of more recent epidemics of encephalitis in horses was the one caused, not by EEE or WEE viruses, but by VEE virus. This epidemic began in northern South America in 1969 and, by 1972, had caused the death of hundreds of thousands of horses in Central America, Mexico and Texas, the cost being in excess of US $30 million. Many human cases were also recorded.
With the widespread availability of vaccines to protect horses, epidemics of these magnitudes have not occurred in recent years, but many horses still die annually in the USA of viral encephalitis. Further details of the history of EEE, WEE, and VEE are available in several comprehensive reviews.1, 10, 19, 23, 27, 29, 30, 41, 46
As mentioned above, West Nile virus was introduced to North America in 1999 and is now endemic in North America. Several thousand horses in the Americas, but mostly in North America, have died of encephalitis caused by this virus. Most reviews of the arboviruses that can cause encephalitis in horses now include West Nile virus.19
In contrast with WN virus, the life cycles of EEE, WEE and VEE viruses indicate that these alphaviruses have evolved in the western hemisphere and have established a balanced ecological relationship with indigenous vertebrate species. Those vertebrate species that develop clinical disease are, in terms of evolution, recent introductions to the continents of North and South America. While humans may have arrived from Asia as early as 30 000 years ago, it is believed that continental dispersion did not occur until approximately 13 000 years ago with the opening of the Canadian ice corridor. The modern horse arrived only after Columbus.
Recent molecular studies on the genetic conservation and evolution of individual alphaviruses have significantly advanced our knowledge of the epidemiology of alphaviruses in the western hemisphere.44 Contrary to the evolution of most RNA viruses, the alphaviruses of North America have evolved slowly. This slow evolution may be due to several factors, but it is most likely a result of the adaptation of the virus to specific species of mosquito vectors for their long-term perpetuation. While the situation with VEE, EEE and WEE in Central and South America is possibly more complex than that in North America (we know less about the life cycles), the concept of the slow evolution of alphaviruses in the western hemisphere (and, by corollary, the failure of the introduced mammalian and avian species that show clinical disease to have co-evolved to a successful host-parasite relationship) is generally accepted as the cornerstone for understanding these diseases in the Americas.
Since EEE, WEE and VEE viruses are exotic to the Old World it follows that, were any of these viruses from the western hemisphere ever to be introduced to tropical or subtropical regions, extensive epidemics could occur. The chances of long-term survival of these viruses would be good in view of the wide diversity of mosquitoes and potential vertebrate hosts present in such regions.
The life cycle of WN virus is similar to that of EEE, WEE and VEE. Its introduction to North America in 1999, its subsequent transmission by indigenous mosquitoes and, most importantly, the confirmation that WN virus now causes encephalitis annually in horses and disease in humans, viewed collectively, allow us to draw a parallel comparison to what might occur in the Old World were EEE, WEE or VEE viruses to be introduced.
Aetiology
Eastern, western and Venezuelan equine encephalitis viruses are classified in the family Togaviridae, genus Alphavirus; this genus also includes other viruses distributed around the world, some in southern Africa such as Chikungunya, Middelburg and Ndumu.44 However, the pathogenic potential of many of these viruses for domestic livestock is essentially unknown. A listing of the alphaviruses and the their associated clinical syndromes is presented as Table 1.44
Table 1 Viruses in he genus Alphavirus
| Antigenic complex | Species | Antigenic subtype | Antigenic variety | Clinical syndromes | Distribution |
| Barmah forest | Barmah forest (BFV) | Febrile illness, rash, arthritis* | Australia | ||
| Eastern equine encephalitis (EEE) | EEEV | Febrile illness, encephalitis* | North, Central, South America | ||
| Middelburg | Middelburg (MIDV) | None recognized* | Africa | ||
| Ndumu | Ndumu virus (NDUV) | None recognized* | Africa | ||
| Semliki forest | Semliki forest (SFV) | Febrile illness* | Africa | ||
| Chikungunya (CHIKV) | Febrile illness, arthralgia, rash* | Africa, Asia | |||
| O'nyong-nyong (ONNV) | Febrile illness, arthralgia, rash* | Africa | |||
| Getah (GETV) | None recognized* | Asia | |||
| Bebaru (BEBV) | None recognized* | Malaysia | |||
| Ross River (RRV) | Sagiyama | Febrile illness, arthralgia, rash* | Australia, Oceania | ||
| Mayaro (MAYV) | Febrile illness, arthralgia, rash* | South America, Trinidad | |||
| Una (UNAV) | None recognized* | South America | |||
| Venezuelan equine encephalitis (VEE) | VEEV | I | AB | Febrile illness, encephalitis* | North, Central, South America |
| C | Febrile illness, encephalitis* | South America | |||
| D | Febrile illness, encephalitis* | South America, Panama | |||
| E | Febrile illness, encephalitis* | Central America, Mexico | |||
| Mosso das Pedras (MDPV) | F | None recognized* | Brazil | ||
| Everglades (EVEV) | VEE-II | Febrile illness, encephalitis* | Florida (USA) | ||
| Mucambo (MUCV) | VEE-III | A | Febrile illness, encephalitis* | South America, Trinidad | |
| C (strain 71D1252) | Unknown* | Peru | |||
| D | Febrile illness* | Peru | |||
| Tonate (TONV) | VEE-IIIB | Febrile illness, encephalitis* | Brazil, Colorado (USA) | ||
| Pixuna (PIXV) | VEE-IV | Febrile illness, myalgia* | Brazil | ||
| Cabassou (CABV) | VEE-V | None recognized* | French Guiana | ||
| Rio Negro (RNV) | VEE-VI | Febrile illness, myalgia* | Argentina | ||
| Western equine encephalitis (WEE) | Sindbis (SINV) | Febrile illness, arthralgia, rash* | Africa, Europe, Asia, Australia | ||
| Babanki | Febrile illness, arthralgia, rash* | Africa | |||
| Ockelbo | Febrile illness, arthralgia, rash* | Europe | |||
| Kyzylagach | None recognized* | Azerbaijan, China | |||
| Whataroa (WHAV) | None recognized* | New Zealand | |||
| Aura (AURAV) | None recognized* | South America | |||
| WEEV | Several | Febrile illness, encephalitis* | Western North, South America | ||
| Highlands J (HJV) | Eastern North America | ||||
| Fort Morgan (FMV) | Buggy Creek | None recognized* | Western North Amreica | ||
| Trocara | Trocara (TROV) | South America | |||
| Salmon pancreas disease | Salmon pancreas disease (SPDV) | Pancreatic disease (salmon) | Atlantic Ocean and tributaries | ||
| Sleeping disease | Sleeping disease (trout) | Europe | |||
| Southern elephant seal | Southern elephant seal (SESV) | None recognized | Australia |
*Note: Clinical syndromes apply to humans
Alphaviruses have enveloped virions 60 to 70 nm in diameter with the envelope tightly surrounding an icosahedral nucleocapsid that has T= 4 symmetry. The viruses are not stable in the environment and are easily inactivated by common disinfectants.
The envelope of the virion is comprised of a lipid bilayer with fine peplomers surrounding a nucleocapsid that is 40 nm in diameter. The envelope is derived from the host cell membrane during budding. The peplomers are arranged as 80 trimers, each a heterodimer composed of two glycoproteins, El and E2. The virus attaches to host cell receptors though the E2 glycoprotein and the E1 protein includes a fusion peptide that mediates entry of nucleocapsids into the cytoplasm from endosomes. A single strand of positive-sense RNA makes up the genome that is approximately 12 kilobase pairs in length. The organization of the genome and the process of viral replication has been reviewed.44
All alphaviruses have common group-specific antigenic determinants identifiable by serological techniques, such as fluorescent antibody, complement fixation and ELISA. The glycoprotein E2 acts as a haemagglutinin, the activity of which is markedly pH dependent. This protein also induces neutralizing antibody responses, and most polyclonal as well as monoclonal antibodies appear to be directed at sites located on E2. Haemagglutination inhibition (HI), although cross-reactive to some extent, has been used to differentiate antigenic complexes. At one time, virus neutralization and kinetic HI reactions were used to differentiate antigenic complexes and species. These techniques led to significant advances in our understanding of the epidemiology of the alphaviruses in the western hemisphere, but have now been replaced by sequence analysis of the RNA genome. The current organization of the family is a composite of antigenic and genetic characterization.
Alphaviruses replicate to high titre in the cytoplasm of infected cells and exit the cell by budding of preassembled nucleocapsids through the plasmalemma. They cause cytopathic effects in a wide range of vertebrate cells in vitro, particularly embryonic avian cells. Infection causes complete shutdown of host-cell protein and nucleic acid synthesis. Invertebrate cells, such as C6/36 derived from Aedes albopictus, are equally sensitive to infection with alphaviruses, but no cytopathic effect is apparent and cell division is unaffected.
Epidemiology
Since EEE, WEE and VEE are typical arbovirus infections, clinical disease in horses or humans usually occurs in midsummer or autumn, a time when mosquito activity is most active and reflective of warm and wet conditions. In the tropics and subtropics (and some adjacent regions that are technically not subtropical, such as Florida in the USA) cases may occur throughout the year.
Eastern equine encephalitis and encephalitis caused by Madariaga virus
The causative viruses are widely distributed in the Americas: from Quebec in the north, through the Caribbean and Central America to South America. In the USA, EEE is most commonly seen in the south-eastern states. It has been detected in all states east of the Mississippi river but also in a number of states west of the Mississippi river. For that reason, the adjective ‘eastern’ is to some extent misleading. There are no substantiated cases of EEE beyond the Americas.
Historically, disease attributable to EEE virus in horses and humans has been recorded throughout Central and South America. The first isolation of virus was in 1930 from a horse in Argentina, although it was not until 1953 that the virus was identified as EEE virus. However, as early as 1964, it was shown that North American (includes the Caribbean Region) and South American strains of EEE were antigenically distinct. More recent study has determined that there are 3 distinct genetic lineages encompassing isolates from Central and South America, but only one lineage representing North America.2 It is estimated that the North American and South American viruses diverged several thousand years ago. Perhaps reflective of this divergence, the epidemiology of the South American viruses differs from that of the North American viruses as they involve small rodents as the reservoir host. In light of these findings, the International Committee on the Taxonomy of Viruses has approved a proposal that the North American and South American EEE viruses be reclassified with Madariaga virus being the new species designation for the South American isolates of EEE virus.2, 44
To whom we are indebted for most of the recent molecular studies on the alphaviruses of the Americas, consider that these lineages may reflect ecological differences in the endemic transmission cycles. In North America, where passerine birds serve as the amplifying host, avian mobility may maintain the genetic homogeneity, with the virus population being frequently mixed following dispersal of viraemic hosts. This hypothesis is supported by the general observation that North American isolates of EEE virus are correlated more closely with the year of isolation rather than the geographic location. In contrast, in South America, where rodents are considered important in the transmission cycle, the limited geographic dispersal of these rodents (in comparison with birds) results in discrete genotypic foci of Madariaga viruses that evolve under the influence of genetic drift and the selective pressure of different hosts and vectors.
In the USA, most cases of EEE in horses occur in the northern parts of peninsular Florida. In some years, despite the availability of inexpensive vaccines, numerous cases may be recorded affecting all breeds of horses, but seldom does one see more than one or two cases on an individual farm in any one year.45 The majority of affected horses are unvaccinated. Disease is less commonly seen in humans; rarely are there more than 10 fatal human cases per year reported for the entire USA; the majority of these have historically occurred in Florida https://diseasemaps.usgs.gov/mapviewer/. Perhaps as a reflection of climate change, more cases of EEE in horses and humans have been recorded since 2000 in the North Eastern states of the USA and Eastern Canada. Occasionally, EEE virus is associated with clinical disease in species considered native to the USA, such as white tailed deer,18 various species of birds,8, 26 and on one occasion in a harbor seal in captivity.20 Eastern equine encephalitis virus was first shown to cause disease in species of birds that are not native to the USA when it was recognized in ring-necked pheasants in 1937 in Connecticut. Since that time, disease in sparrows, pigeons, Pekin ducks, and Chukar partridges (all old-world species) has also been reported. In 1991, EEE virus was shown to be the cause of death in commercial flocks of emus dying of haemorrhagic colitis in Louisiana; similar outbreaks have been reported in emus, ostriches, cassowaries and penguins.16, 37, 39 Outbreaks of EEE have been recorded in intensive pig herds in southern Georgia, USA.9 Cases attributable to EEE virus have been reported in cattle,21 sheep,3 and camelids.24 The records of veterinary diagnostic laboratories in the USA indicate that other mammalian species, such as dogs, moose, and, surprisingly, even manatees, are apparently susceptible to natural infection as assessed by the presence of serum antibodies.
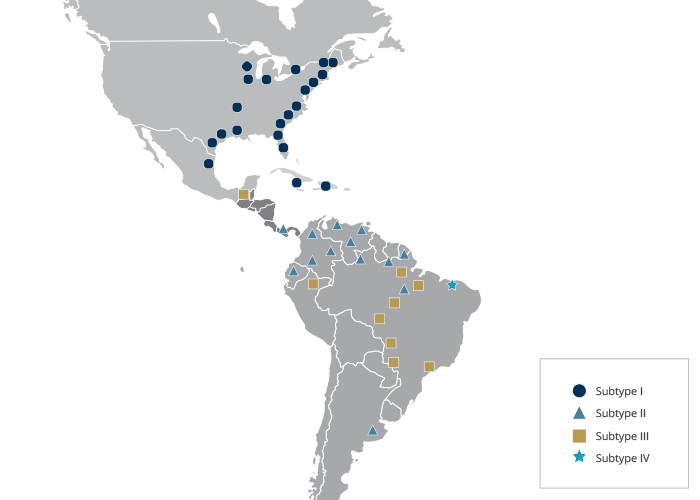
Until recently there were only isolated cases of encephalitis caused by Madariaga virus in horses and humans in Central and South America, but recently outbreaks in both horses and humans have been reported.7, 25, 35 The first reported outbreak of Madariaga virus causing encephalitis in humans was not until 2010 in Panama.7 The outbreak was characterized by the co circulation of both Madariaga virus and VEE virus with over 100 suspected cases and 19 hospitalizations for encephalitis. Madariaga virus was confirmed in 13 cases, VEE virus in 11, and one case of dual infection was detected. Figure 1 illustrates the distribution of EEE and Madariaga viruses in the Americas.
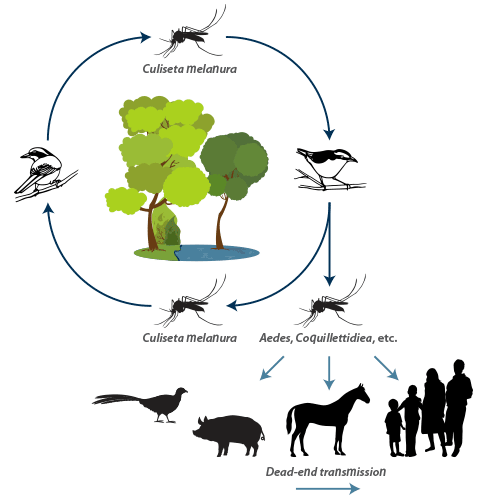
On the North American continent, the perpetuation of EEE virus is maintained in a ‘silent’ bird/mosquito cycle (i.e. no clinical disease is apparent) involving passerine birds of freshwater swamps and one species of ornithophilic mosquito, Culiseta melanura (the primary vector or endemic vector). Several species of secondary vector or epidemic vector mosquitoes are capable of biologically transmitting the virus to horses, humans, and other vertebrate species in which clinical disease develops. Generally, the level of viraemia in these clinically affected species is insufficiently high to allow further transmission of the virus by mosquitoes; i.e. in evolutionary terms these vertebrate species are “dead-end” hosts.
Culiseta melanura frequents wetlands, particularly those with peat-muck soil dominated by hardwood trees. These wet and murky habitats and hardwood root systems favour oviposition by Cs. melanura and its larval development; larvae require water with a high content of organic matter and protection from sunlight. Female Cs. melanura feed almost exclusively on birds, particularly passerines, and a cycle of infection between birds and mosquitoes is maintained by the development of high viraemias in birds. Because Cs. melanura is highly ornithophilic, other vectors are considered to be responsible for transmitting EEE virus from the avian/Cs. melanura cycle. Eastern equine encephalitis virus has been isolated from over 20 different mosquito species representing six genera. Coquillettidia perturbans is suspected as a major epidemic or secondary vector because it feeds on mammals (including humans and horses) and birds with equal facility. Cases of EEE tend to cluster within 10 km of swampy habitats, which is within the flight range of this mosquito. Figure 2 illustrates the transmission cycle of EEE virus in North America.
The overwintering mechanism of EEE virus has been a matter of conjecture for many years and there are many interesting, but unproven theories. Transovarial transmission of EEE virus is not considered to be a significant overwintering mechanism, although there are reports that it occurs. In tropical and subtropical climates, the mosquito/avian cycle is presumably a continuous process, but in North America (excluding Florida) there are long winters during which no adult mosquitoes are present on the wing. Field studies in the state of Alabama using both serology and quantitative reverse transcription polymerase chain reaction to detect viral RNA, support the earlier hypothesis that snakes (more specifically cottonmouths Agkistrogon piscivorus at this study site), provide an overwintering mechanism for EEE virus in North America.4, 14
Culiseta melanura is a temperate breeding species of mosquito and does not readily breed in areas such as southern Florida and the Caribbean. It is therefore not surprising that EEE is not an endemic disease in these areas. However, sporadic cases of EEE in horses and small epidemics have been recorded in this region, presumably through the migratory flight of viraemic birds providing an occasional source of infection for secondary vectors. These secondary vectors can initiate a short-term epidemic, but, in the absence of a primary vector, such as Cs. melanura, endemicity is not established.
Isolation from mosquitoes of the subgenus Culex (Melanoconion) in Central and South America implicate these mosquitoes as the endemic vectors of Madariaga virus. Antibody prevalence studies of birds and small rodents indicate that, in contrast with North America, small rodents are involved in the primary cycle. The geographic scales defining the distribution of the different lineages of Madariaga virus are highly focal, on the order of a few hundred miles or less. This pattern suggests a mode of transmission that limits dispersal of these viruses and is consistent with the use of mammalian vertebrate hosts as reservoirs and amplifiers.2
Western equine encephalitis
Following the recognition of WEE virus in the 1930s, there was a period when it was thought that WEE virus was distributed throughout North and South America – the ‘western’ merely indicating the region in the USA where the disease was first and subsequently predominantly recognized. However, it is now believed that most infections that occur east of the Mississippi River are caused by a related, but distinct virus, known as Highlands J or HJ virus. Highlands J virus is less virulent for mammals than WEE virus, but has caused natural cases of encephalitis in horses and is pathogenic for introduced species of poultry.
Cases of WEE in horses and humans are now uncommon. No human cases of WEE have been recorded by CDC in the USA for the last 15 years and the last reported veterinary case was in 2004 https://diseasemaps.usgs.gov/mapviewer/. While equine vaccination against WEE is widespread and the human population is better protected against biting mosquitoes, (for example through improved housing [window screens, air conditioning etc.] and the use of, mosquito repellents when outdoors), there is no complete explanation for this change in the incidence of WEE.
The primary vectors of WEE virus are culicine mosquitoes, particularly Culex tarsalis that, at least in North America, maintain WEE virus in a cycle with birds, especially nestling passerines. Horses, even those that are obviously clinically affected, do not produce viraemias high enough to infect mosquitoes. Culex tarsalis population abundance is favoured by a rapid increase in temperature following a cool, wet spring, resulting in the rapid melting of snow and flooding of rivers. This species of mosquito also has a predilection for irrigated lands as breeding sites. Other ornithophilic mosquitoes become infected as the summer progresses and the infection eventually spills over to other types of birds, mammals and possibly even reptiles and amphibians. Most, if not all, of these infections are inapparent. This in turn results in the virus becoming established in species of mosquitoes with host preferences other than birds.
Despite intensive attempts to demonstrate transovarial transmission by mosquitoes, including Cx. tarsalis, this has not been proven, and interepidemic maintenance in temperate areas by mosquitoes is therefore in doubt. In South America, where Cx. tarsalis does not occur, antibody prevalence rates in birds are lower than in North America and the species (both vertebrate and invertebrate) responsible for maintaining the virus on that continent have not been identified.
In western North America, at least two variants of WEE virus (Fort Morgan and Buggy Creek) that are transmitted between birds by swallow nest bugs (Oeciacus vicarius) have been isolated. Neither variant is considered to be pathogenic for humans or horses. The closely related virus, Highlands J, causes occasional encephalitis in horses in Florida and possibly other eastern states of the USA. This virus is also recognized as an important cause of disease in domestic turkeys, pheasants, and some exotic species of birds. Western encephalitis virus has been reported in several countries in South America (Argentina, Guyana, Ecuador, Brazil and Uruguay), but only in Argentina has it been associated with human disease and significant epidemics in horses. The species of mosquitoes, from which WEE virus has been mostly isolated, feed principally on mammals rather than on birds.
Understanding the antigenic and genetic relationships between the viruses in the WEE complex has proved more challenging than the viruses in the EEE and VEE virus complexes. Western equine encephalitis virus is a member of the WEE antigenic complex that includes several Old World viruses in addition to the New World viruses described above (Table 1). Phylogenetic analyses of isolates from North and South America indicate that regional WEE lineages appear to evolve independently for several years to a few decades (for example, genotypes in South America are apparently absent from North America). However, common genotypes of WEE are readily dispersed across both North and South America. This contrasts with the situation within collections of EEE and VEE viruses where certain virus genotypes appear to be restricted to either North or South America.43
Venezuelan equine encephalitis
The first recognized outbreak of VEE occurred initially in Colombia and then in Venezuela in 1935 to 1936, although it is almost certain that outbreaks had occurred earlier in the region. A virus isolate from a case in Venezuela was subsequently shown to be different from WEE and EEE; VEE had entered the lexicon of virology. In contrast with EEE and WEE, VEE epidemics are more sporadic, but when they have occurred they have often had a wide geographic impact and caused disease in large numbers of horses and humans. In an epidemic that spread from the border between Peru and Equador, through Central America, to North America between 1969 and 1972 it is estimated that between 50 000 and 100 000 horses, mules and donkeys died and 250 000 humans were affected (mainly an influenza- like disease, but some cases of encephalitis and death). This epidemic revealed the potential for VEE to spread rapidly with an equine case fatality rate often as high as 90 per cent in some areas. Until this epidemic, clinical disease had never been recorded in Central or North America. Since the involvement of parts of southern Texas in 1971, clinical disease in horses due to VEE viruses has not occurred in the USA. It has never occurred in Canada.
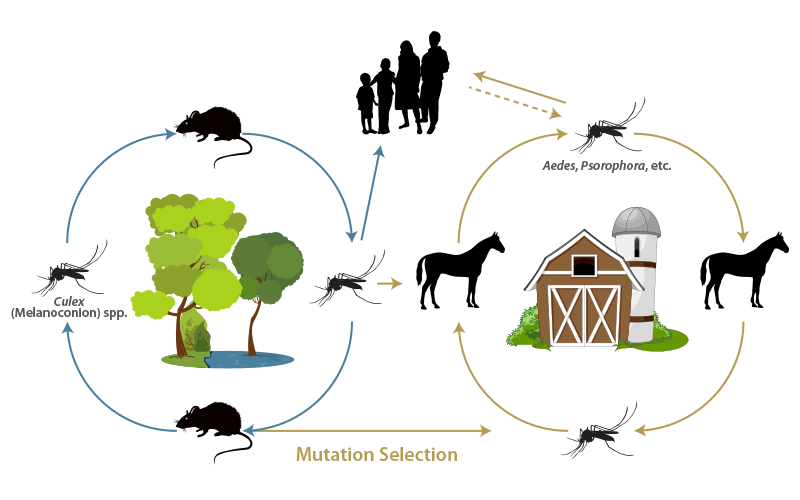
Before further discussion, it is important to note that, in contrast with EEE and WEE, where horses are not considered to be a major source of virus for the vector, in VEE epidemics, horses are the most important amplifiers of virus activity. Horses infected with VEE viruses develop viraemias between 105.5 and 108.0 TCID50/ml, which persist for up to five days.36
It was not until the 1990’s that epidemic VEE re-emerged in horses. In 1995, an epidemic of VEE occurred in Venezuela and Colombia that had all the initial hallmarks of the 1969-1972 epidemic. Not only did large numbers of horses, mules and donkeys die in this 1995 epidemic, but there was also extensive involvement of the human population, with an estimated 75 000 to 100 000 cases being recorded. VEE viruses isolated in 1995 were genetically very similar to those associated with disease in the 1960s.
It is important to note that a significantly smaller outbreak of VEE had been recorded two years earlier in 1993 in the Mexican state of Chiapas, in southern Mexico and a similar outbreak subsequently occurred in the neighbouring state of Oaxaca in 1996. In contrast with the 1995 epidemic in Venezuela and Colombia, the 1993 and 1996 outbreaks in Mexico involved comparatively few horses. Importantly, these outbreaks represented the first confirmed equine cases attributed to a different type of VEE virus, namely subtype IE.
Based on the use of kinetic haemagglutination inhibition assays in the late 1960s, distinct subtypes and varieties of viruses were identified, within what has now become known as the VEE virus complex. In these early serological studies, it was found that those viruses that had a history of being associated with epidemics in horses could be grouped together. This identification of the complex was a major advance in understanding the epidemiology of VEE infections. The alphaviruses within the VEE complex are currently classified into six subtypes, designated I to VI, and consist of 9 species, of which subtype I contains viruses of veterinary and medical importance (Table 144). With minor exceptions, molecular analyses of the genomes of a wide selection of geographically dispersed VEE viruses have supported the antigenic classification. Molecular analysis appears to have distinct advantages over serological methods for more precisely determining epidemiological relatedness and patterns of VEE virus evolution. Thus, ‘phylogenetic trees’ of the nucleotide sequences of the viral genome are progressively supplanting the serological classification denoting endemic and epidemic types. However, the serological classification of VEE viruses still features in the international regulations controlling the movement of horses travelling between countries. Many still prefer to review the molecular analyses within the more familiar, albeit to some, confusing, antigenic classification presented in Table 1.
Molecular analyses have also been used to address the central enigma of the epidemiology of VEE, namely ‘what is the source of the epidemic strains of VEE virus and how do they persist between epidemics?’ While we do not have a comprehensive understanding of how epidemics in horses originate, a clearer picture is emerging. Isolates that are virulent for horses do not appear to be transmitted in the inter-epidemic period. It is generally accepted that endemic viruses within antigenic subtype I appear to have the greatest potential to mutate to create the epidemic variants. Studies on the viruses isolated from horses during the 1995 epidemic in Venezuela and Colombia demonstrated that the emergence of VEE virus into an epidemic form was associated with specific mutations arising in the envelope glycoprotein 2 (E2) gene of endemic subtype ID.42 These mutations resulted in changes on the surface of the virion spikes that gave rise to increased virulence and viremia in horses and sometimes enhanced infection of epidemic vector mosquitoes, such as Aedes (Ochlerotatus) taeniorhynchus. These higher viremia levels in horses can lead to infection of mosquitoes that are not normally involved in endemic circulation which can then result in spillover infections of humans.11
But the epidemiology of how endemic strains of VEE emerge to cause disease outbreaks is not uniform. As mentioned earlier, the outbreaks of VEE in horses in Mexico in 1993 and 1995 were associated with mutations to IE strains, which hitherto had not been associated with disease in horses. The isolates of IE from these outbreaks exhibited significantly greater infectivity for Aedes (Ochlerotatus) taeniorhynchus when compared with closely related endemic strains of IE viruses, supporting the hypothesis that adaptation to an efficient epidemic vector may have contributed to the emergence of disease.5 While the mutation led to clinical disease in horses, experimental infection of horses established that the level of viremia associated with these viruses was low and probably insufficient to infect mosquitoes,13, 34 which may, in part, explain the limited nature of the epidemics as infected horses were not acting as amplifiers of infection.
Applying such knowledge, the prevailing opinion is that VEE viruses classified in other subtypes of the complex other than subtype I do not appear to constitute a significant disease risk to horses. The situation in the USA, whereby VEE viruses have been found over many years to be endemic in areas such as southern Florida (Everglades virus) and Colorado (Bijou Bridge virus) in the USA without ever causing clinical disease in horses, illustrates the point.
There is a wide range of mosquitoes capable of biologically transmitting the virus to horses and humans. At least 41 species of mosquitoes from 11 genera have been found to be naturally infected with epidemic strains of VEE virus. Twenty of the mosquito species are in the genus Culex and 13 in the subgenus Melanoconion. The dominant sylvatic (endemic) virus vectors are rodent-feeding species of Culex. Virus has also been isolated from Culicoides spp. (Ceratopogonidae) and blackflies (Simuliidae), but it is not known whether insects in these families are capable of biological transmission of VEE virus. Rodents appear to be the most important vertebrate hosts for endemic viruses, but over a 100 species of birds have been either virologically or serologically associated with transmission of epidemic VEE virus. Shore birds in general, and herons in particular, appear to be capable of serving as amplifier hosts. Birds may develop viraemias as high as 108.0 TCID50/ml of blood. Other animals such as bats, marsupials, rats, sloths, rabbits, racoons and non-human primates may also contribute to virus maintenance. Dogs regularly become infected and may be capable of virus amplification. Figure 3 illustrates the transmission cycles of VEE viruses in the Americas.
Pathogenesis
The regional lymph node is presumed to be the site of primary viral replication following cutaneous inoculation of EEE, WEE or VEE viruses by mosquito bite. The reticuloendothelial system, in general, is a major target in epidemic VEE infections.36 It may be of interest to know that VEE and, to a lesser extent, EEE viruses have been intensively studied as biological weapons, and strains have been adapted to be infective as aerosols. Although one might expect little published information on such viruses, there are in fact extensive reports on the pathogenesis of these viruses in laboratory animals and how model systems have been developed to evaluate vaccines and antiviral drugs.36
In humans, naturally infected by mosquitoes, the common symptoms (fever, headache and myalgia accompanied by leukopenia) of infection with alphaviruses have been ascribed to circulating interferon and other soluble mediators. In horses, brain lesions are thought to be the direct result of viral replication and are characterized by necrotizing encephalitis with neuronal dysfunction. Immunity following both inapparent infection and frank disease caused by EEE, WEE and VEE viruses is long lasting.
Clinical signs
Different strains of EEE, WEE and VEE viruses may differ in their pathogenicity not only for horses but also for humans, certain domestic and wild animals, birds and laboratory animals. Clinical observations during epidemics of VEE indicate that the disease is generally less severe in donkeys (burros) and mules. However, no experimental studies have been reported confirming the field reports. None of these viruses appears to cause clinical disease in their reservoir hosts, i.e. species indigenous to North and South America. Although there are broad differences in the clinical manifestations that are produced by the three virus complexes in horses and humans, many infections, apart from those caused by highly virulent strains, are subclinical. Eastern equine encephalitis and epidemic VEE viruses are generally more neuroinvasive than WEE and endemic VEE viruses. Young animals of all susceptible species, as well as children, are more liable than adults to develop signs referable to infection of the central nervous system. The incubation periods of EEE, WEE and VEE vary between two to three days and three weeks. Inapparent infections in horses may or may not be accompanied by fever. In clinical cases, pyrexia is the first clinical manifestation of infection. It has usually abated by the time signs of encephalitis become evident. Neurological signs are variable, but depression, ataxia, paralysis, anorexia and ultimately stupor all occur in clinical cases. Irregular gait, grinding of teeth, incoordination, circling, staggering, head pressing and hyperexcitability are also observed. Severely ataxic animals may stand by leaning against walls or other objects and sometimes stand with their hind legs crossed. Partial or even total blindness may be evident. In severely depressed horses, the head hangs low with drooping ears, and the eyelids may be slightly swollen and partly closed, while the lips are flaccid and the tongue may protrude from the mouth. The profound depression associated with these virus infections give rise to the common name of ‘sleeping sickness’. Oesophageal paralysis manifested by repeated unsuccessful attempts to drink has also been described.
The course of the disease in severe cases varies between 2 and 14 days. Nearly all horses with EEE encephalitis die irrespective of the quality and intensity of clinical care. Cases of VEE and WEE are more likely to survive. Terminally ill horses can no longer stand, become comatose and frequently thrash their feet (paddling), resulting in marked disturbance of the soil and vegetation around the animal.
Encephalitis in humans caused by EEE virus is also associated with a poor prognosis; the presentation of WEE is generally milder. Human infections with VEE virus are more commonly seen as an ‘influenza-like’ disease rather than encephalitis, but encephalitis does occur, particularly in young children. The neurologic sequelae resulting from encephalitic alphavirus infections has been reviewed.31 Several of the reviews mentioned above also describe the disease in humans.10, 27, 29, 32
Most other animal species infected with the viruses of EEE, WEE and VEE generally develop subclinical infections. Such subclinical infections, however, may be associated with viraemias that are sufficiently high to infect mosquitoes with a low threshold for infection; for example dogs infected with VEE virus.27 Pigs that are less than two months of age when infected with EEE virus can develop encephalitis; the mortality rate may be high.9
Non-indigenous birds, such as emus and ostriches, when infected with EEE virus may show signs of encephalitis, but more commonly they exhibit haemorrhagic diarrhoea and prostration.37
Pathology
There are no consistent gross lesions in the central nervous system or elsewhere in horses that die of EEE, WEE or VEE. Histologically, neuronal necrosis with neurophagia, marked perivascular cuffing, the infiltrating cells comprising both mono- and polymorphonuclear leukocytes, and focal and diffuse microglial proliferations are evident. The lesions are more pronounced in the grey than in the white matter of the brain. Lesions are most marked in the cerebral cortex, thalamus and hypothalamus, while the spinal cord is mildly affected. The pathology in humans is similar.12, 27, 29, 41
The pathology of EEE in emus and other ratites is characterized by hepatitis and diffuse lymphoid necrosis.40
Diagnosis and differential diagnosis
Even in endemic areas it is difficult for an experienced clinician to differentiate the various types of encephalitis with any certainty. Long19 has provided an illustrated description of the diagnostic procedures used in North America for the differential diagnosis of encephalitis in the horse. While the focus is on EEE and West Nile the principles apply to WEE and VEE. When only an individual case is presented, the differential diagnosis should include rabies, equine protozoal myeloencephalitis (EPM), equine herpesvirus (EHV)-1, verminous encephalitis (VE), hepatoencephalopathy, mouldy corn poisoning, botulism, trauma, cervical vertebral myelopathy, and neoplasia.
For antemortem diagnosis, cerebrospinal fluid (CSF) analysis can be particularly helpful in helping to differentiate EEE from encephalitis caused by West Nile virus; in acute infections with EEE virus, the pleocytosis is predominately neutrophilic, whereas in acute encephalitis caused by West Nile virus, if pleocytosis is present, mononuclear cells predominate. Detection of IgM in serum can also be helpful for antemortem diagnosis. Virus isolation from blood is rarely possible as the viraemia has frequently ceased by the time encephalitis develops. However, in a VEE epidemic it may be possible to isolate the virus from non-encephalitic horses in the group, particularly if they have elevated body temperatures.
Following postmortem examination examination, the viruses of EEE, WEE, VEE, and West Nile encephalitis can usually be isolated in cell culture (e.g. Vero cells) from brain tissue. Historically, intracerebral inoculation of suckling mice was successfully used to isolate these viruses. Virus isolation has now been supplanted by the use of reverse transcription polymerase chain reaction to detect characteristic gene sequences of the causal virus.47 Immunochemistry can also be used to detect viral antigen in the brain and spinal cord.19
For horses that survive, paired sera can be examined using a virus neutralization test, but must be interpreted within the context of prior vaccination.
When disease in avian species is suspected as being caused by either EEE or WEE viruses, samples of liver, spleen and intestine should be collected in addition to brain tissue. OIE has information on the tests approved for the diagnosis of EEE, WEE, VEE and West Nile infections, http://www.oie.int/international-standard-setting/terrestrial-manual/access-online.
Control
The treatment of human patients with encephalitis caused by arboviruses relies upon several different strategies often involving combinations of intravenous immunoglobulins, corticosteroids, and interferon. Sadly, the outcome is seldom favourable and for those that survive the sequelae may be severe and life changing.
The welfare of the horse must always be considered when treating cases of encephalitis. Recumbent animals have a poor prognosis and euthanasia is appropriate. Corticosteroids can be used to lessen the brain oedema in horses, but the results are inconsistent.
Vaccination is critical for the prevention of EEE, WEE, VEE and West Nile encephalitis in the horse. Inactivated vaccines for EEE and WEE became available shortly after the discovery of the two viruses. Bivalent vaccines (consisting of formaldehyde-inactivated virus) against EEE and WEE are commercially available in North America. The vaccine requires a primary and secondary immunization schedule about a month apart followed by biannual boosters.19 The vaccines are not particularly effective in protecting foals and yearling horses for reasons that are not understood.10 The vaccines are inexpensive, but even in communities where cost should not be an issue, not all horses in endemic areas get vaccinated.
With EEE and WEE vaccination is directed to protecting the horse from disease. With VEE, vaccination of horses not only protects them from disease but it also helps to control the outbreak spreading further by removing the horse from the epidemic cycle as a source of infection for mosquitoes; vaccination removes the horse as the viral amplifier.
The 1969 to 1972 VEE epidemic in Central America and the southern USA was controlled partially by immunizing large numbers of horses with an attenuated VEE virus strain, TC-83. This vaccine strain was produced by serial passage of an epidemic variant in guinea-pig cell cultures. The vaccine was originally developed to protect military personnel against biological warfare and was fortuitously available to control the VEE epidemic in horses. Because of concerns over the presence of low-level viraemias in some horses and the possible transmission of vaccine virus between horses by mosquitoes and reversion in virulence, inactivated vaccines against VEE are now available for use in horses. These vaccines are not widely used in North America as they compromise the international movement of horses for competition and breeding.
Because of concerns that arboviruses can be used for bioterrorism, research is supported by various countries on new vaccines against EEE and VEE. As with the history of TC-83, new vaccines that have been developed for use in humans may, in turn, prove safe and effective in protecting horses. Several new alphavirus vaccine constructs have been shown to provide protection in laboratory animals against EEE and VEE infection, but no studies in horses have been reported.28, 33
OIE has information on the availability of vaccines for EEE, WEE, VEE and West Nile infections, http://www.oie.int/international-standard-setting/terrestrial-manual/access-online.
It has been shown through experience with both EEE and epidemic VEE, that mosquito control and immunization of horses are both important in preventing and controlling disease. Vector control can be achieved through reducing the breeding activities of mosquitoes by implementing appropriate water management systems, although this can be difficult in extensive areas. The widespread dispersal (usually achieved by aerial spraying) of insecticides was used successfully to control the 1969-1972 epidemic of VEE. There are a number of environmental factors that would need to be considered before embarking on such an approach were another epidemic to occur. Concern over widespread spraying of insecticides can be mitigated in some circumstances if the biology of the mosquito vector is well known. For example, in northern Florida, where EEE is endemic, treating the pools of water in which Cs. melanura breeds with a larvicide is often practical and economically feasible on those farms with valuable horses.10 Swamps with the soil types that support the breeding of Cs. melanura can often be recognized by the non-entomologist by the presence of the loblolly bay tree (Gordonia lasianthus); this broadleaf evergreen tree grows to a height of about 10m and can easily be recognized by its white magnolia- like flowers and/or serrated leaves. Risk assessment can be assisted by geographic mapping of large areas where mosquitoes breed by using thematic mappers, such as those on orbiting satellites.22
References
- ARECHIGA-CEBALLOS, N. & AGUILAR-SETIEN, A., 2015. Alphaviral equine encephalomyelitis (Eastern, Western and Venezuelan). Revue Scientifique et Technique-Office International Des Epizooties, 34, 491-501.
- ARRIGO, N.C., ADAMS, A.P. & WEAVER, S.C., 2010. Evolutionary patterns of Eastern equine encephalitis virus in north versus South America suggest ecological differences and taxonomic revision. Journal of Virology, 84, 1014-1025.
- BAUER, R.W., GILL, M.S., POSTON, R.P. & KIM, D.Y., 2005. Naturally occurring eastern equine encephalitis in a Hampshire wether. Journal of Veterinary Diagnostic Investigation, 17, 281-285.
- BINGHAM, A.M., GRAHAM, S.P., BURKETT-CADENA, N.D., WHITE, G.S., HASSAN, H.K. & UNNASCH, T.R., 2012. Detection of Eastern Equine Encephalomyelitis Virus RNA in North American Snakes. American Journal of Tropical Medicine and Hygiene, 87, 1140-1144.
- BRAULT, A.C., POWERS, A.M., ORTIZ, D., ESTRADA-FRANCO, J.G., NAVARRO-LOPEZ, R. & WEAVER, S.C., 2004. Venezuelan equine encephalitis emergence: Enhanced vector infection from a single amino acid substitution in the envelope glycoprotein. Proceedings of the National Academy of Sciences of the United States of America, 101, 11344-11349.
- BROWN, T.P., ROBERTS, W. & PAGE, R.K., 1993. Acute hemorrhagic enterocolitis in ratites: Isolation of eastern equine encephalomyelitis virus and reproduction of the disease in ostriches and turkey poults. Avian Diseases, 37, 602–605.
- CARRERA, J.P., FORRESTER, N., WANG, E.Y., VITTOR, A.Y., HADDOW, A.D., LOPEZ-VERGES, S., ABADIA, I., CASTANO, E., SOSA, N., BAEZ, C., ESTRIPEAUT, D., DIAZ, Y., BELTRAN, D., CISNEROS, J., CEDENO, H.G., DA ROSA, A.P.T., HERNANDEZ, H., MARTINEZ-TORRES, A.O., TESH, R.B. & WEAVER, S.C., 2013. Eastern equine encephalitis in Latin America. New England Journal of Medicine, 369, 732-744.
- DAY, J.F. & STARK, L.M., 1996. Eastern equine encephalitis transmission to emus (Dromaius novaehollandiae) in Volusia County, Florida: 1992 through 1994. Journal of the American Mosquito Control Association, 12, 429–436.
- DEIN, F.J., CARPENTER, J.W., CLARK, G.G., MONTALI, R.J., CRABBS, C.L., TSAI, T.F. & DOCHERTY, D.E., 1986. Mortality of captive whooping cranes caused by eastern equine encephalitis virus. Journal of the American Veterinary Medical Association, 189, 1006–1010.
- ELVINGER, F., LIGGETT, A.D., TANG, K.N., HARRISON, L.R., COLE, J.R.J., BALDWIN, C.A. & NESSMITH, W.B., 1994. Eastern equine encephalomyelitis virus infection in swine. Journal of the American Veterinary Medical Association, 205, 1014–1016.
- FORRESTER, N.L., WERTHEIM, J.O., DUGAN, V.G., AUGUSTE, A.J., LIN, D., ADAMS, A.P., CHEN, R.B., GORCHAKOV, R., LEAL, G., ESTRADA-FRANCO, J.G., PANDYA, J., HALPIN, R.A., HARI, K., JAIN, R., STOCKWELL, T.B., DAS, S.R., WENTWORTH, D.E., SMITH, M.D., POND, S.L.K. & WEAVER, S.C., 2017. Evolution and spread of Venezuelan equine encephalitis complex alphavirus in the Americas. PLoS Neglected Tropical Diseases, 11, E0005693.
- GIBBS, E.P.J. & TSAI, T.F., 1994. Eastern equine encephalitis. In: BERAN, G.W., (ed.). Handbook of Zoonoses. 2nd Edition. Boca Raton: CRC Press Inc, 11–24.
- GONZALEZ-SALAZAR, D., ESTRADA-FRANCO, J.G., CARRARA, A.S., ARONSON, J.F. & WEAVER, S.C., 2003. Equine amplification and virulence of subtype IE Venezuelan equine encephalitis viruses isolated during the 1993 and 1996 Mexican epizootics. Emerging Infectious Diseases, 9, 161-168.
- GRAHAM, S.P., HASSAN, H.K., CHAPMAN, T., WHITE, G., GUYER, C. & UNNASCH, T.R., 2012. Serosurveillance of Eastern Equine Encephalitis Virus in Amphibians and Reptiles from Alabama, USA. American Journal of Tropical Medicine and Hygiene, 86, 540-544.
- GREENWAY, T.E., ELDRIDGE, J.H., LUDWIG, G., STAAS, J.K., SMITH, J.F., GILLEY, R.M. & MICHALEK, S.M., 1998. Induction of protective immune responses against Venezuelan equine encephalitis (VEE) virus aerosol challenge with microencapsulated VEE virus vaccine. Vaccine, 16, 1314–1323.
- GUTHRIE, A., CITINO, S., ROOKER, L., ZELAZO-KESSLER, A., LIM, A., MYERS, C., BOLIN, S.R. & TRAINOR, K., 2016. Eastern equine encephalomyelitis virus infection in six captive southern cassowaries (Casuarius casuarius). Journal of the American Veterinary Medical Association, 249, 319-324.
- IVERSEN, J.O., 1994. Western equine encephalitis. In: BERAN, G.W., (ed.). Handbook of Zoonoses. 2nd Edition. Boca Raton: CRC Press Inc, 25–31
- KIUPEL, M., FITZGERALD, S.D., PENNICK, K.E., COOLEY, T.M., O'BRIEN, D.J., BOLIN, S.R., MAES, R.K. & DEL PIERO, F., 2013. Distribution of Eastern equine encephalomyelitis viral protein and nucleic acid within central nervous tissue lesions in White-tailed deer (Odocoileus virginianus). Veterinary Pathology, 50, 1058-1062.
- LONG, M.T., 2014. West Nile virus and equine encephalitis viruses: new perspectives. Veterinary Clinics of North America-Equine Practice, 30, 523-542.
- MCBRIDE, M.P., SIMS, M.A., COOPER, R.W., NYAOKE, A.C., CULLION, C., KIUPEL, M., FRASCA, S., FORRESTER, N., WEAVER, S.C., WEBER, E.S. & AQUATIC VET, S., 2008. Eastern equine encephalitis in a captive harbor seal (Phoca vitulina). Journal of Zoo and Wildlife Medicine, 39, 631-637.
- MCGEE, E.D., LITTLETON, C.H., MAPP, J.B. & BROWN, R.J., 1992. Eastern equine encephalomyelitis in an adult cow. Veterinary Pathology, 29, 361-363.
- MONCAYO, A.C., EDMAN, J.D. & FINN, J.T., 2000. Application of geographic information technology in determining risk of eastern equine encephalomyelitis virus transmission. Journal of the American Mosquito Control Association, 16, 28–35.
- MORRIS, C.D., 1989. Eastern equine encephalitis. In: MONATH, T.P., (ed.). The Arboviruses: Epidemiology and Ecology. Vol. 3. Boca Raton: CRC Press Inc, 1–20.
- NOLEN-WALSTON, R., BEDENICE, D., RODRIGUEZ, C., RUSHTON, S., BRIGHT, A., FECTEAU, M.E., SHORT, D., MAJDALANY, R., TEWARI, D., PEDERSEN, D., KIUPEL, M., MAES, R. & DEL PIERO, F., 2007. Eastern equine encephalitis in 9 South American Camelids. Journal of Veterinary Internal Medicine, 21, 846-852.
- OLIVEIRA, R.D., IAMAMOTO, K., SILVA, M., ACHKAR, S.M., CASTILHO, J.G., ONO, E.D., LOBO, R.S.V., BRANDAO, P.E., CARNIELI, P., CARRIERI, M.L., KOTAIT, I. & MACEDO, C.I., 2014. Eastern equine encephalitis cases among horses in Brazil between 2005 and 2009. Archives of Virology, 159, 2615-2620.
- OLIVER, J., LUKACIK, G., KRAMER, L.D., BACKENSON, P.B., SHERWOOD, J.A. & HOWARD, J.J., 2016. Geography and timing of cases of Eastern equine encephalitis in New York State from 1992 to 2012. Vector-Borne and Zoonotic Diseases, 16, 283-289.
- OSORIO, J.E. & YUILL, T.M., 1994. Venezuelan equine encephalitis In: BERAN, G.W., (ed.) Handbook of Zoonoses. 2nd Edition. Boca Raton: CRC Press Inc, 33–46.
- PAESSLER, S. & WEAVER, S.C., 2009. Vaccines for Venezuelan equine encephalitis. Vaccine 27 Suppl 4, D80-D85.
- REISEN, W.K. & MONATH, T.P., 1989. Western equine encephalitis. In: MONATH, T.P., (ed.). The Arboviruses: Epidemiology and Ecology. Vol. 5. Boca Raton: CRC Press Inc, 89–137
- RICO-HESSE, R., 2000. Venezuelan equine encephalitis. Veterinary Clinics of North America: Equine Practice, 16, 553-563.
- RONCA, S.E., DINELEY, K.T. & PAESSLER, S., 2016. Neurological Sequelae Resulting from Encephalitic Alphavirus Infection. Frontiers in Microbiology, 7, 959 doi: 10.3389/fmicb.2016.00959.
- ROOS, K.L., 1999. Encephalitis. Neurologic Clinics, 17, 813.
- ROY, C.J., ADAMS, A.P., WANG, E.Y., LEAL, G., SEYMOUR, R.L., SIVASUBRAMANI, S.K., MEGA, W., FROLOV, I., DIDIER, P.J. & WEAVER, S.C., 2013. A chimeric Sindbis-based vaccine protects cynomolgus macaques against a lethal aerosol challenge of eastern equine encephalitis virus. Vaccine, 31, 1464-1470.
- SAHU, S.P., PEDERSEN, D.D., JENNY, A.L., SCHMITT, B.J. & ALSTAD, A.D., 2003. Pathogenicity of a Venezuelan equine encephalomyelitis serotype IE virus isolate for ponies. American Journal of Tropical Medicine and Hygiene, 68, 485-494.
- SILVA, M., GALIZA, G.J.N., DANTAS, A.F.M., OLIVEIRA, R.N., IAMAMOTO, K., ACHKAR, S.M. & RIET-CORREA, F., 2011. Outbreaks of Eastern equine encephalitis in northeastern Brazil. Journal of Veterinary Diagnostic Investigation, 23, 570-575.
- TAYLOR, K.G. & PAESSLER, S., 2013. Pathogenesis of Venezuelan equine encephalitis. Veterinary Microbiology, 167, 145-150.
- TULLY, T.N.J., SHANE, S.M., POSTON, R.P., ENGLAND, J.J., VICE, C.C., CHO, D.Y. & PANIGRAHY, B., 1992. Eastern equine encephalitis in a flock of emus (Dromaius novaehollandiae). Avian Diseases, 36, 808–812.
- TURELL, M.J., LUDWIG, G.V., KONDIG, J. & SMITH, J.F., 1999. Limited potential for mosquito transmission of genetically engineered, live-attenuated Venezuelan equine encephalitis virus vaccine candidates. American Journal of Tropical Medicine and Hygiene, 60, 1041–1044.
- TUTTLE, A.D., ANDREADIS, T.G., FRASCA, S. & DUNN, J.L., 2005. Eastern equine encephalitis in a flock of African penguins maintained at an aquarium. Journal of the American Veterinary Medical Association, 226, 2059-2062.
- VEAZEY, R.S., VICE, C.C., CHO, D.Y., TULLY, T.N.J. & SHANE, S.M., 1994. Pathology of Eastern equine encephalitis in emus (Dromaius novaehollandiae). Veterinary Pathology, 31, 109–111.
- WALTON, T.E. & GRAYSON, M.A., 1989. Venezuelan equine encephalomyelitis. In: MONATH, T.P., (ed.). The Arboviruses: Epidemiology and Ecology. Vol. 4. Boca Raton: CRC Press Inc, 203–231.
- WEAVER, S.C., FERRO, C., BARRERA, R., BOSHELL, J. & NAVARRO, J.C., 2004. Venezuelan equine encephalitis. Annual Review of Entomology, 49, 141-174.
- WEAVER, S.C., POWERS, A.M., BRAULT, A.C. & BARRETT, A.D.T., 1999. Molecular epidemiological studies of veterinary arboviral encephalitides. Veterinary Journal, 157, 123–138.
- WEAVER, S.C., WINEGAR, R., MANGER, I.D. & FORRESTER, N.L., 2012. Alphaviruses: Population genetics and determinants of emergence. Antiviral Research, 94, 242-257.
- WILSON, J.H., RUBIN, H.L., LANE, T.J. & GIBBS, E.P.J., 1986. A survey of Eastern equine encephalitis in Florida horses: prevalence, economic impact, and management practices, 1982–1983. Preventive Veterinary Medicine, 4, 261–271.
- ZACKS, M.A. & PAESSLER, S., 2010. Encephalitic alphaviruses. Veterinary Microbiology, 140, 281-286.
- ZINK, S.D., JONES, S.A., MAFFEI, J.G. & KRAMER, L.D., 2013. Quadraplex qRT-PCR assay for the simultaneous detection of Eastern equine encephalitis virus and West Nile virus. Diagnostic Microbiology and Infectious Disease, 77, 129-132.