- Infectious Diseases of Livestock
- Part 2
- Jaagsiekte
- GENERAL INTRODUCTION: PARAMYXOVIRIDAE AND PNEUMOVIRIDAE
- Rinderpest
- Peste des petits ruminants
- Parainfluenza type 3 infection
- Bovine respiratory syncytial virus infection
- Hendra virus infection
- Paramyxovirus-induced reproductive failure and congenital defects in pigs
- Nipah virus disease
- GENERAL INTRODUCTION: CALICIVIRIDAE AND ASTROVIRIDAE
- Vesicular exanthema
- Enteric caliciviruses of pigs and cattle
- GENERAL INTRODUCTION: RETROVIRIDAE
- Enzootic bovine leukosis
- Jaagsiekte
- Visna-maedi
- Caprine arthritis-encephalitis
- Equine infectious anaemia
- GENERAL INTRODUCTION: PAPILLOMAVIRIDAE
- Papillomavirus infection of ruminants
- Papillomavirus infection of equids
- GENERAL INTRODUCTION: ORTHOMYXOVIRIDAE
- Equine influenza
- Swine influenza
- GENERAL INTRODUCTION: CORONAVIRIDAE
- Porcine transmissible gastroenteritis
- Porcine respiratory coronavirus infection
- Porcine epidemic diarrhoea
- Porcine haemagglutinating encephalomyelitis virus infection
- Porcine deltacoronavirus infection
- Bovine coronavirus infection
- Ovine coronavirus infection
- Equine coronavirus infection
- GENERAL INTRODUCTION: PARVOVIRIDAE
- Porcine parvovirus infection
- Bovine parvovirus infection
- GENERAL INTRODUCTION: ADENOVIRIDAE
- Adenovirus infections
- GENERAL INTRODUCTION: HERPESVIRIDAE
- Equid herpesvirus 1 and equid herpesvirus 4 infections
- Equid gammaherpesvirus 2 and equid gammaherpesvirus 5 infections
- Equine coital exanthema
- Infectious bovine rhinotracheitis/infectious pustular vulvovaginitis and infectious pustular balanoposthitis
- Bovine alphaherpesvirus 2 infections
- Malignant catarrhal fever
- Pseudorabies
- Suid herpesvirus 2 infection
- GENERAL INTRODUCTION: ARTERIVIRIDAE
- Equine viral arteritis
- Porcine reproductive and respiratory syndrome
- GENERAL INTRODUCTION: FLAVIVIRIDAE
- Bovine viral diarrhoea and mucosal disease
- Border disease
- Hog cholera
- Wesselsbron disease
- Louping ill
- West nile virus infection
- GENERAL INTRODUCTION: TOGAVIRIDAE
- Equine encephalitides caused by alphaviruses in the Western Hemisphere
- Old World alphavirus infections in animals
- Getah virus infection
- GENERAL INTRODUCTION: BUNYAVIRIDAE
- Diseases caused by Akabane and related Simbu-group viruses
- Rift Valley fever
- Nairobi sheep disease
- Crimean-Congo haemorrhagic fever
- GENERAL INTRODUCTION: ASFARVIRIDAE
- African swine fever
- GENERAL INTRODUCTION: RHABDOVIRIDAE
- Rabies
- Bovine ephemeral fever
- Vesicular stomatitis and other vesiculovirus infections
- GENERAL INTRODUCTION: REOVIRIDAE
- Bluetongue
- Ibaraki disease in cattle
- Epizootic haemorrhagic disease
- African horse sickness
- Equine encephalosis
- Palyam serogroup orbivirus infections
- Rotavirus infections
- GENERAL INTRODUCTION: POXVIRIDAE
- Lumpy skin disease
- Sheeppox and goatpox
- Orf
- Ulcerative dermatosis
- Bovine papular stomatitis
- Pseudocowpox
- Swinepox
- Cowpox
- Horsepox
- Camelpox
- Buffalopox
- GENERAL INTRODUCTION: PICORNAVIRIDAE
- Teschen, Talfan and reproductive diseases caused by porcine enteroviruses
- Encephalomyocarditis virus infection
- Swine vesicular disease
- Equine picornavirus infection
- Bovine rhinovirus infection
- Foot-and-mouth disease
- GENERAL INTRODUCTION: BORNAVIRIDAE
- Borna disease
- GENERAL INTRODUCTION: CIRCOVIRIDAE AND ANELLOVIRIDAE
- Post-weaning multi-systemic wasting syndrome in swine
- GENERAL INTRODUCTION: PRION DISEASES
- Scrapie
- Bovine spongiform encephalopathy
- Transmissible spongiform encephalopathies related to bovine spongiform encephalopathy in other domestic and captive wild species
Jaagsiekte
This content is distributed under the following licence: Attribution-NonCommercial CC BY-NC  View Creative Commons Licence details here
View Creative Commons Licence details here
Jaagsiekte
D W VERWOERD, R C TUSTIN, C V HALLWIRTH AND D F YORK
Introduction
Jaagsiekte (JS) is a contagious bronchiolo-alveolar carcinoma of the lungs of sheep caused by a retrovirus. It was first recognized in South Africa more than a century ago,84, 89 when a detailed description of its clinical signs and macroscopic pathology was published by Hutcheon in 1891, along with evidence of its infectivity and an account of attempts to control it.32 This was followed by reports from England,34 Germany22 and France.1 The disease is now known to have an almost worldwide distribution and to have considerable economic impact in countries with substantial sheep populations. In South Africa, a survey over a 40-year period indicated that JS is by far the most common neoplasm reported in sheep (64 per cent).7
In the early literature, the disease was often confused with other lung conditions of sheep. The most important of these is maedi, which is caused by a lentivirus and characterized by chronic, non-neoplastic inflammatory lesions.58 Today it is known that the two diseases are closely associated in many countries and are often present as co-infections in the same animal, which adds to the existing confusion.16, 32, 39, 64, 79 A list of the diseases of livestock caused by retroviruses is given in Table 59.1.
Table 59.1 Diseases of livestock caused by members of the family Retroviridae
| SUBFAMILY | HOST ANIMAL* | DISEASE |
|---|---|---|
| Oncovirinae | Sheep, (goat) Cattle, (sheep) | Jaagsiekte Enzootic bovine leukosis |
| Lentivirinae | Sheep, (goat) Goat, (sheep) Horse, donkey | Maedi-visna Caprine arthritis-encephalitis Equine infectious anaemia |
| Spumavirinae | Various animals | Non-pathogenic, ‘foamy viruses’ in cell culture |
*the animal species given in the brackets are uncommon hosts
Aetiology
Even though JS was known to be contagious long before its neoplastic nature had been elucidated, many years of research in various countries failed to reveal its causal agent, although filtration experiments suggested a viral aetiology. The first successful experimental transmission of the disease by co-habitation was reported by De Kock,13 yielding results that were to be confirmed in Iceland nine years later.20 An ovine herpesvirus was the first candidate virus isolated,12, 17, 35, 37 but transmission and molecular hybridization experiments proved that this virus is a passenger and not directly involved in the aetiology of the disease.18
The possibility of a retroviral aetiology was first suggested by the observation in Israel of typical retrovirus particles in adenomatous lesions.68 Retroviruses were also found in cell cultures established from JS-affected lungs37 and reverse transcriptase (RT) activity in extracts of tumour tissue.70 However, no transmission attempts were made in these studies and the results were difficult to interpret in view of the possible presence of maedi-visna lentivirus in the material studied.
Several lines of evidence cumulatively implicated a retrovirus called Jaagsiekte Sheep Retrovirus (JSRV) in the aetiology of JS:14, 25, 56, 88
- Transmission studies showed that the disease could be reproduced by inoculation with cell-free extracts of tumours or cultured tumour cells, or with concentrated lung fluid containing RT activity characteristic of retroviruses. 27, 41, 65, 75, 90
- Serial transmission in new-born lambs with semi-purified lung-rinse material containing RT activity with a magnesium ion preference resulted in incubation periods inversely proportional to the RT level in the inocula.93
- Serological studies showed that both lung fluid and tumour homogenate contained proteins that cross-react with antisera to capsids and nucleocapsids of type D and type B retroviruses.77
- Retrovirus-like particles were observed by electron microscopy in lung tumours and lung fluid of affected sheep.62, 68
- The full genome of a JSRV isolated from the lungs of a JS affected sheep was cloned and sequenced, and genomic organization typical of typeDand B retroviruses shown.97, 98
- JSRV was found to be consistently, specifically and absolutely associated with JS.53
The above evidence did not prove unequivocally that infection with JSRV is sufficient to induce JS, and the possibility existed that JSRV was acting as a helper virus, complementing a hitherto undiscovered acutely transforming retrovirus. Further definition of the role played by JSRV in JS was hampered by the lack of an in vitro culture system for the virus. The precise nature of the association of JSRV with the disease was not known until Palmarini and co-workers constructed an infectious molecular clone derived from an integrated proviral exogenous JSRV sequence (called JSRV21) isolated from a spontaneous case of JS.55 The JSRV21 genome was placed under the transcriptional control of the human cytomegalovirus (CMV) immediate early promoter. The promoter was positioned in such a way that the resulting RNA transcript would be very similar to wild-type exogenous JSRV RNA, giving rise to the plasmid pCMV2JS21. The latter was transferred into the highly transfectable human 293T cell line, from which substantial amounts of JSRV21 virus were released into the supernatant. Concentrated stocks of JSRV21, obtained from transfected 293T cells, were used for intratracheal inoculation of new-born lambs. Two of the lambs developed JS (confirmed at the histological, immunological, and molecular level), thus proving conclusively that JSRV is both necessary and sufficient to induce the disease.57
Jaagsiekte sheep retrovirus exists in two closely related but molecularly distinct forms. The first is an infectious exogenous retrovirus (henceforth referred to as exJSRV) which is transmitted horizontally from one animal to another and is the aetiological agent of JS. The second is a group of endogenous retroviral loci that resemble exJSRV at the nucleotide level, but are transmitted/inherited vertically through the germ line.99 These endogenous sequences will be referred to as enJSRV. The latter are widely distributed in ungulates, with 15 to 20 copies of closely related sequences being found in the genomes of all domestic sheep and goats.26 Three different loci have been cloned and completely sequenced.54 Jaagsiekte is associated absolutely with its aetiological agent exJSRV,53 whereas enJSRVs by themselves are not associated with disease. No sheep population that lacks enJSRVs has been identified, and the possible role of these sequences in disease, as well as their importance to the host organism, has yet to be elucidated.
Morphology and morphogenesis of the virion
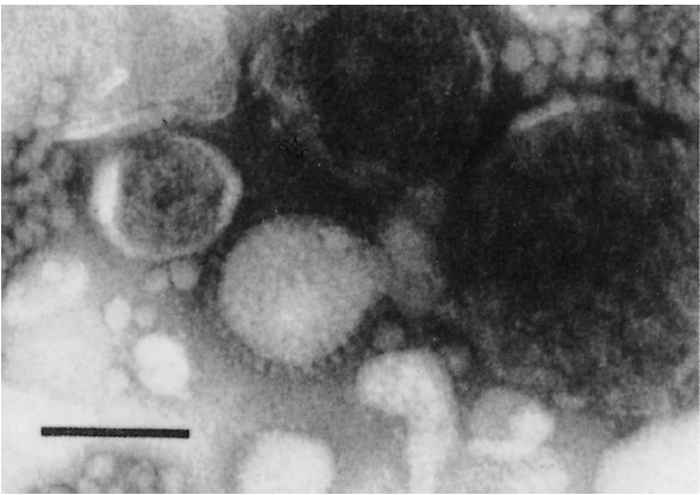
Mature virions have an average diameter of 110nm and, when negatively stained (Figure 59.1), appear as pleomorphic, enveloped particles with spikes on the surface, typical of most retroviruses. The morphology of positively stained JSRV is distinct from that of any other retrovirus (Figure 59.2). It possesses a slightly eccentric nucleoid with an electrondense perinucleoidal space.62 The virus has a density of 1,17 to 1,18 g/ml, similar to that of mouse mammary tumour virus (MMTV) and Mason-Pfizer monkey virus (MPMV), respectively type B and type D retroviruses.
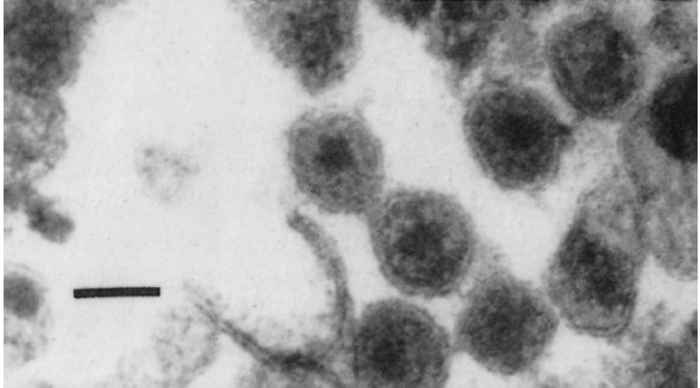
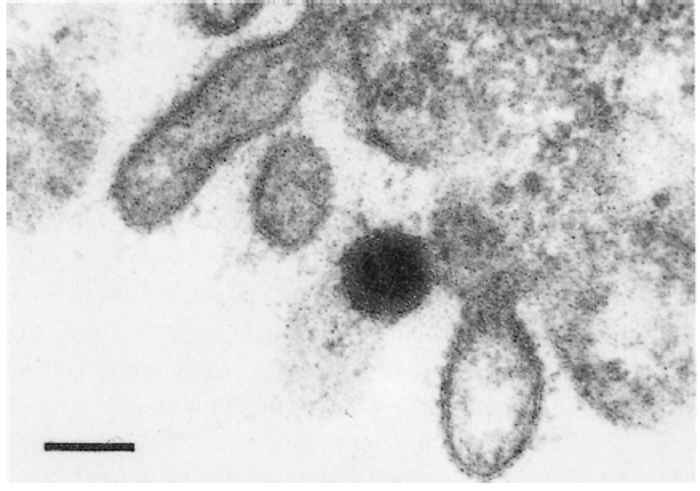
Replicating JSRV particles are occasionally seen in tumour cells. Immature particles, 75 nm in diameter, assemble in the cytoplasm (Figure 59.3) and are then transported to the cell membrane, where budding of mature particles takes place (Figure 59.4). The morphogenesis is similar to that of types B and D retroviruses, but is quite distinct from that of type C oncoviruses (e.g. bovine leukaemia virus) and lentiviruses (e.g. maedi-visna virus), where the virions are assembled at the cell membrane in a crescent shape before budding as an immature particle.62
In addition to the similarities in morphology, morphogenesis and density, JSRV also shares a common antigenic determinant77 and a preference by its RT for Mg++ ions93 with MPMV (type D) and MMTV (type B) oncoviruses. In contrast, both type C oncovirus and lentivirus enzymes prefer Mn++ and no serological cross-reactions can be demonstrated with these viruses. Anti-MPMV p27 serum has been used successfully to localize the related JSRV p29 antigen by using immunocytochemical techniques in sections of JS lung lesions.33, 63
Molecular characterization of exJSRV
The viral genome of exJSRV consists of single-stranded RNA. Arguably, the most significant advancement in the study of JSRV during the past decades has been the elucidation and publication of the complete genomic nucleotide sequence of exJSRV.98, 99 The sequence was obtained from a South African isolate of exJSRV, JSRV-SA. The JSRV-SA genome was found to be 7462 nucleotides in length, measured from the 5’R (‘repeat’) to the 3’R region. JSRV-SA exhibits a genomic organization typical of types D and B retroviruses, having overlapping open reading frames (ORFs) encoding the structural genes gag, pro, pol and env, with a separate pro reading frame (Figure 59.5). In addition, JSRV has an accessory ORF overlapping pol, the function and significance of which has not, as yet, been determined. This reading frame was termed ‘orf- X’, and seems to be conserved among all exJSRVs sequenced thus far.2, 57, 73
The inability to culture the virus in vitro has always been a limitation. However, the recent isolation/construction of a full length infectious clone of the exJSRV (JSRV21) solved a number of problems and it will be used in the future to answer many questions relating to this virus.57 For one, the protein profile of the virus in SDS-PAGE remained an uncertainty for many years. Whilst the sequenced genome provided the theoretical protein profile based on the computer- translated open frames, it was only recently that it was possible to confirm the SDS-PAGE profile of the virus by analysing a purified extract of JSRV21 (Figure 59.6). The JSp26 group-specific antigen was one of the few proteins that were identified with certainty when purified JSRV fractions from infected lung lavages were analysed.91, 98 The JSp26 protein has immunologically shared epitopes with MPMV and MMTV, prototypes of the D and B oncoviruses, respectively.77
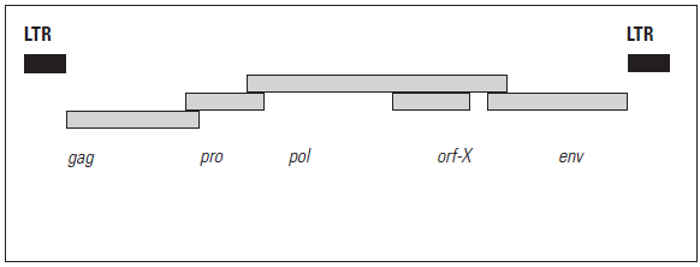
Figure 59.5 Schematic representation of the proviral genome of the exogenous infectious JSRV with the classical gag, pro, pol and env open reading frames. Open reading frame orf-X has no known function. The proviral genome is close to 7 848 base pairs long (including both LTRs) (adapted from York, Vigne, Verwoerd & Querat, 199299)
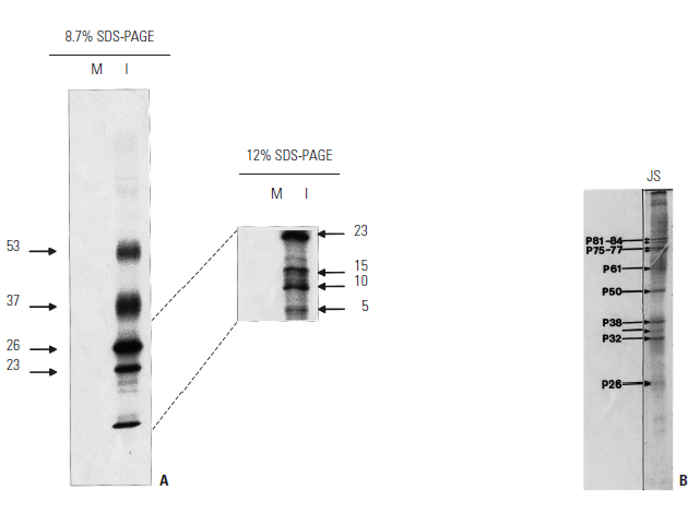
Figure 59.6 (A) SDS-PAGE of the proteins of the purified infectious clone (JSRV21) labelled with 35 methionine.57 The proteins were analysed on 8,7 per cent (left) and 12 per cent (right) polyacrylamide gels to resolve the lower molecular weight proteins. (B) The protein profile of the virus ‘purified’ from the lung lavage of a JS affected sheep were stained with Coomassie Blue, which is less sensitive than radioactive labelling. Note the apparent lack of the lower molecular weight proteins in this profile (from York 198797)
Analysis of the exJSRV-SA sequence revealed that it contains a primer binding site complementary to the 3’ end of a tRNALys 1,2 immediately downstream of the U5 region.99 This suggests a relationship to a group of human endogenous retroviruses (HERVs) that utilize a lysine (K) tRNA recognition sequence as their primer binding site for second strand synthesis during reverse transcription, hence the designation ‘HERV-K’.23, 36 The orf-X region is considered rather unusual. Its predicted polypeptide sequence of 166 amino acids is very hydrophobic and exhibits a codon usage that is significantly different from that of the pol gene. However, it does not appear to impose any constraints of the codon usage of pol. Recently, there have been three independent reports of the conservation of orf-X in all published exJSRV sequences, as well as in a number of enJSRVs.2, 57, 73 The studies concluded that orf-X must be a functional gene and that it was not a chance generation in JSRV-SA. Only one group of researchers has thus far reported any meaningful sequence homology between the deduced amino acid sequence of orf-X and other known proteins, namely mammalian adenosine receptor subtype 3.2 This is a G-proteincoupled receptor that regulates diverse biological events, including cell growth and differentiation. The reported amino acid identity and similarity are 28 per cent and 50 per cent, respectively, over a stretch of 90 amino acids (out of the total of 166 amino acids). Structural analysis of the putative mRNA transcript of orf-X placed it in a category of transcripts which include those that code for oncogenic growth factors, transcriptional factors, and regulatory proteins. These transcripts are predicted to be poorly translated.2 It has been conceded that the high degree of conservation of orf-X may result from the fact that it overlaps pol, which needs to be conserved to maintain biological function.73 It was suggested that orf-X encodes a novel, hydrophobic and membrane-bound protein of unkown function. A potential role of orf-X in cellular transformation has been argued against, the assumption being that enzootic nasal tumour virus (ENTV), a retrovirus that exhibits 95 per cent amino acid similarity with JSRV-SA and that is associated with intranasal tumours of sheep and goats, utilizes the same oncogenic mechanism as exJSRV.9 Enzootic nasal tumour virus has two stop codons in the region homologous to orf-X, which therefore does not represent an accessory ORF. It is a possibility that the two stop codons observed in the orf-X region of the published ENTV sequence might merely be an artefact of the sequencing strategy employed by the authors concerned, and that a possibility exists of ENTV exhibiting an uninterrupted orf-X region. It should be possible to investigate a possible pathogenic role for orf-X by sitedirected mutagenesis of that gene in the infectious JSRV21 clone.
Utilizing molecular probes, the transformed epithelial tumour cells in the alveoli of JS-affected sheep have been identified as the major sites of proviral exJSRV translation and virion assembly.52 A high level of viral expression is also observed in the mediastinal lymph nodes draining the lungs, and a lower level of expression has been demonstrated in various anatomically dispersed lymphoid tissues (e.g. the spleen, thymus and bone marrow) and peripheral blood mononuclear cells.55
More specifically, exJSRV was found to infect CD4+, CD8+ and B cells, as well as adherent lymphocytes, which exhibit a higher proviral load than the non-adherent cells.31 Macrophages in the lungs of affected animals contain the highest proviral burden of the non-lung tissues. This type of dispersed viral infection, of both lymphoid and non-lymphoid tissues, is similar to that observed with other type B and type D retroviruses.55
It has not yet been determined whether lymphoid infection with exJSRV precedes infection of the cells where transformation occurs, or if it is a consequence thereof. Proviral exJSRV DNA was detected in lymphoid cells of animals seven days post-inoculation, at which point there were no signs of JS.30 This suggests that lymphoid infection precedes transformation and tumour development. Systemic lymphatic dissemination could also precede transformation, but this has not yet been demonstrated.
Endogenous JSRV sequences
The existence of numerous endogenous sequences related to exJSRV in sheep genomes was first reported in 1992.99 This finding resulted from molecular probing of sheep genomic DNA with probes based on the sequence elucidated for JSRV-SA. Probes based on the capsid (CA, part of gag) region have been used to quantify the endogenous genomic sequences related to JSRV, and it was found that the probes hybridized to more than 20 bands, both in the tissues of JS-affected and unaffected sheep.24 These findings were subsequently extended to demonstrate that probes based on the envelope surface (SU) and CA regions hybridized to between 15 and 20 bands in all members of domestic sheep and goats tested.26 The banding patterns obtained were similar for the different genomes tested, indicating that the integration sites are probably similarly conserved. Furthermore, the SU and CA probes hybridized under high stringency conditions to samples obtained from six wild Ovis and Capra genera, implying that the enJSRV loci must have become fixed in the genomes of sheep and goats prior to the time of their domestication. The gag regions were found to be more conserved among sheep and goat enJSRVs than the env regions.
The consistent presence of a ScaI endonuclease restriction site in the gag region of exJSRVs that was absent in enJSRVs has been reported.53 These findings allowed a hemi-nested polymerase chain reaction (PCR) protocol to be designed that could distinguish between endogenous and exogenous JSRV transcripts on the basis of the differential ScaI restriction site in gag. The results of these tests showed that transcripts of endogenous JSRV origin are expressed in various sheep tissues, and that the exogenous form of JSRV is exclusively associated with JS.53 It was hypothesized that the loss of certain control regions in the long terminal repeats (LTRs) of enJSRV through insertions and mutations have reduced their potential for pathogenicity relative to exJSRV. It was also found that the U3 region in the LTRs of enJSRV proviral sequences contain insertions of 30 and 16 base pairs relative to exJSRV. This observation led to a far more sensitive hemi-nested PCR, based on the unique differences between the U3 regions of endogenous and exogenous sequences, facilitating better detection of exJSRV in a background of enJSRV sequences.55
Using PCR primers based on the sequence of exJSRV, a number of full-length enJSRV fragments from sheep genomes have been amplified and characterized, and attempts to classify the enJSRV loci based on the restriction profiles obtained for each isolate made.3 The complete nucleotide sequences of three proviral endogenous JSRV loci have now been determined.54 Owing to the fact that the endogenous clones were obtained subsequent to endonuclease digestion of sheep genomic DNA, as opposed to being generated in fragments by PCR amplification, these sequences are a faithful representation of those found within the genetic material of the animals they were derived from. The availability of full-length endogenous JSRV sequences is the primary requirement for sequence comparisons between pathogenic and non-pathogenic JSRVs, which are ultimately expected to lead to the elucidation of the mechanism of transformation employed by these viruses.
The sequence comparisons conducted thus far have identified three regions of conserved differentiation between enJSRVs and exexJSRVs, as has been previously reported, 3 but they also appear to diverge in terms of their sequences, based on the fact that no combination of theoretical nucleotide omissions in the enJSRV U3 regions will lead to homology with the corresponding sequences in exJSRVs. At least, the homology that can be achieved this way is far lower than that which is observed between regions in the U3 that are naturally conserved among the JSRVs. Considering the fact that the U3 regions of retroviruses encode numerous viral control regions, it is likely that there is some degree of biological significance attached to the diversification of enJSRVs and exJSRVs. Further detailed analysis of the differences between endogenous and exogenous U3 regions is required, as the features that distinguish pathogenic from non-pathogenic retroviruses can sometimes be subtle. A recently reported example of this is the demonstration that a single base pair change in the LTR of certain endogenous MMTV loci distinguishes those which are expressed in the mammary gland of mice from those that are not.74
The second conserved difference between enJSRVs and exJSRVs is part of the matrix region, which differs greatly between the two groups, and the associated loss of the prolinerich region in enJSRVs. Considering the high degree of diversification in this region and the homology in the surrounding sequences, it seems likely that the appearance of the proline-rich region in the exogenous viruses was the result of a homologous recombination with another related retrovirus. This is a common occurrence and a frequent source of mutations in retroviruses.38 A further variable region is encountered downstream of the proline-rich region; it is still within the first two-thirds of gag.
The fourth region which consistently differs when comparing exJSRVs to enJSRVs is part of the latter half of the transmembrane region in env. Although the generalized observation of a low degree of homology in this region has been previously reported,2, 3 it has never been described in detail, nor has the underlying reason for this difference been established. Using the comparisons which were made possible by the elucidation of the endogenous sequences, it was noted that the same region in ENTV also diverged from both enJSRVs and exJSRVs, although to a lesser extent from the latter group. It is possible that this region is involved in the determination of cellular host tropism, seeing that exJSRVs and ENTV infect and transform similar tissues in different parts of the respiratory system, whereas enJSRVs must once have had the ability to integrate into the DNA of germ line cells, otherwise they could not have become endogenous viruses that are passed from one generation to the next. Even though these findings do not as yet identify the region of the JSRV genome which is responsible for cellular transformation, they do indicate which regions should be studied in greater detail in order to elucidate the oncogenic nature of exJSRVs.
Phylogenetic relationships of exJSRV and enJSRV
Phylogenetic analysis shows that exJSRVs are closely related to Mason-Pfizer monkey virus (MPMV), the type D retroviral prototype, in the gag, pro and pol protein regions, but that the env proteins of these viruses are not related.99 Conversely, the env proteins encoded by exJSRVs are homologous in terms of their structural organization with those of MMTV (the type B retroviral prototype) and HERV-K (also a type B retrovirus), whereas the gag, pro and pol genes of these viruses are not closely related.23 exJSRVs are therefore thought to have diverged from the A-B-D evolutionary lineage of retroviruses between the divergence of the B prototype MMTV and the D prototype MPMV. exJSRVs thus represent a chimaera characterized by a type B morphology (as suggested by the spikes on the viral envelope, encoded by env) and a predominantly type D gag/pro/pol genome. Another possible explanation of the chimaeric nature of exJSRVs is that it could have arisen as a consequence of homologous recombination between type B and type D retroviruses.26
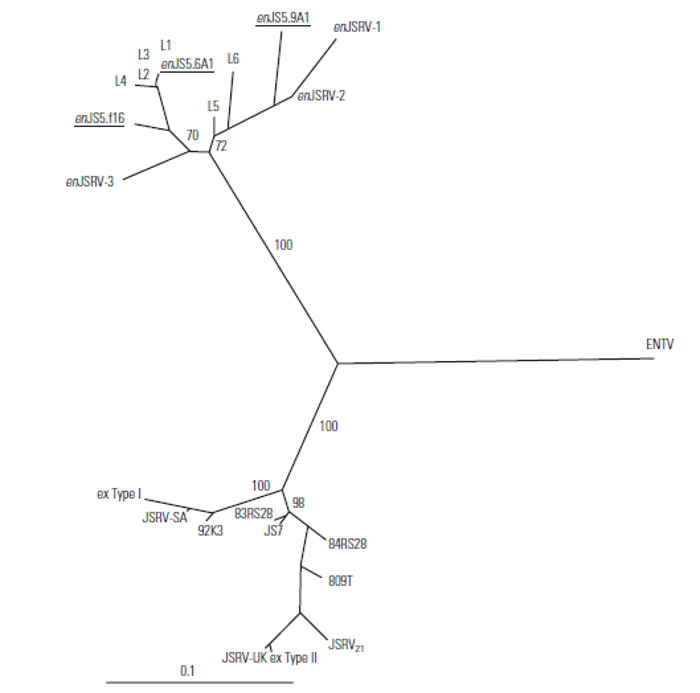
Based on phylogenetic analyses of the U3 region3, 54 as well as orf-X and env regions2 of various isolates of JSRV there are two exogenous genotypes of JSRV, the older of the two, being endemic to Africa, referred to as type I, and the more recent, being prevalent in the USA and UK, referred to as type II. Furthermore, the African JSRV genotypes group more closely with endogenous JSRV sequences than do the ones from the USA and UK.3
Phylogenetic analyses of the three complete endogenous JSRV genome sequences and the exJSRV sequences group them separately (Figure 59.7). Enzootic nasal tumour virus, a recently cloned and sequenced isolate regarded as the aetiological agent of enzootic nasal tumour, is equidistant from the endogenous and exogenous JSRV isolates.9
Oncogenic potential of exJSRV
The mechanism of transformation in JS has not yet been elucidated, but the possible means by which exJSRVs could cause neoplasia have been reviewed.25 Owing to the recent discovery that the JSRV21 provirus had integrated into a part of the sheep genome that does not align with any known cellular sequences (including proto-oncogenes), the possibility of insertional mutagenesis being the transforming mechanism is strongly indicated. 57 Furthermore, seeing that the JSRV21 clone was able to induce JS by itself, in the same time frame as field isolates, exJSRV is not a helper virus for an acutely transforming retrovirus. It was concluded that the genome of exJSRV itself harbours its oncogenic potential.
This leaves three possible alternatives suggested by Hecht and his co-workers.25 Firstly, even though exJSRV does not encode any sequences known to be associated with oncogenesis, one of its protein products may still lead to cellular transformation. Secondly, one of the viral products may act as a transcription factor to stimulate a cellular oncogene. This type of trans-activation is usually associated with a longer time course than the one observed in experimentally induced JS, but exJSRV may harbour a more potent activator than those found thus far. The third alternative is that the binding of the type II pulmonary epithelial cell receptors by their retroviral ligands could result in signal transduction, thereby stimulating the production of growth factors, followed by cell proliferation and subsequent neoplasia.
Epidemiology
Jaagsiekte occurs in sheep as a sporadic or endemic disease in all continents of the world where sheep occur, except in Australasia.6, 11, 15, 42, 49, 80, 81, 83, 84, 95 It displays an extremely variable prevalence between countries. For example, its prevalence can be as high as 20 per cent in certain flocks in South Africa, whereas it is infrequently diagnosed in the USA and Canada.14, 25
Natural disease with a low prevalence does occur in goats in some countries, but it has not been diagnosed in this species in southern Africa. It has, however, been experimentally transmitted to kids in South Africa.86 It would appear that goats are less susceptible to infection by JSRV than are sheep, because in one experiment a viral dose that produced extensive lesions in more than 90 per cent of new-born lambs after two to three months92 produced only small circumscribed lesions in two out of five kids after 9 to 13 months.86 Transmission studies in Europe have provided similar results.76 Apart from the low prevalence of the natural disease in goats, this view is also supported by the fact that in Iceland, during the epidemic of JS in the 1930s and 1940s, JS was not seen in goats despite close contact with sheep suffering from the disease.20
Jaagsiekte is endemic throughout South Africa and Namibia.84 Observations suggest that more animals die as a result of the disease in winter84 or after a sudden drop in environmental temperature,21 possibly as a result of secondary bacterial pneumonia. This gives a false impression of a greater prevalence during the colder months.
Outbreaks of JS occur when affected sheep are introduced into clean flocks. In Iceland, where the disease was eradicated in 1952,78 it was observed that approximately five-and-a-half to eight months elapsed between the introduction of affected sheep and the appearance of new cases14, 21 in a highly susceptible population where the animals are closely confined during winter. In South Africa, several years may elapse between the introduction of an infected animal into a ‘clean’ flock and the realization by the farmer that something is seriously amiss.84
It seems that there are breed differences in susceptibility. In a major epidemic of JS in Iceland it was found that Gottorp sheep were far more susceptible than animals of the Adalbol breed.19 In the UK, the fact that the disease has a higher prevalence in Scotland than in England also suggests that there are breed differences in susceptibility.77 In South Africa, Merino and Karakul sheep and their cross-breeds are apparently more susceptible than English breeds.87 In addition, it was found in Iceland that offspring of different rams varied greatly in their susceptibility to the disease.19 When these genetic differences were recognized, farmers were urged to breed from resistant families and breeds and this led to a decrease in the prevalence of the disease.
A survey in South Africa indicated that the annual mortality due to JS in infected flocks varied from less than 1 to 24 per cent, with an average of 3,6 per cent.84 The sex of the animal does not seem to play a role in its susceptibility.84
It is assumed that all sheep with lung lesions, however small, may excrete the virus. In most affected animals the tumour probably grows slowly and clinical signs are only manifested when the lesions are relatively well advanced and have therefore been present for some considerable time.84 Examination of the lungs of sheep from infected flocks which have been slaughtered out in toto suggests that some animals possess lung lesions which are static or, at most, progress very slowly.84
Figure 59.7 Phylogenetic analysis of the U3 region of a selection of exogenous and endogenous JSRV sequences. The exogenous JSRV sequences have been classified into two types: JSRV type I (African) and JSRV type II (North American and UK). The endogenous and exogenous retroviruses cluster together but each group is equally distant from ENTV (the goat enzootic nasal tumour virus). Accession numbers of the sequences used in the above analysis: JSRV-SA:M80216 end. locus 1, 2, 3, 4, 5 & 6 LTR; ex. type I LTR; ex type II LTR:X95445-X95452, ENTV:Y16627; JS7, 809T, 83RS28, 84RS28, 92K3:Y18301-Y18305; enJSRV-1, 2 & 3:Z66531-Z66533; JSRV-UK:Z71304:AF105220; enJS5.F16, 5.9A1 & 5.6A1:AF136224, AF136225 & AF153615
Although sheep of all ages are susceptible to experimental infection, neonatal lambs are more susceptible than are lambs more than a few days old.72, 92 It is likely, therefore, that affected sheep contract the disease at a very early age from infected dams, some of which are asymptomatic.
Experimental results suggest that intra-uterine transmission does not occur.59, 84 Therefore, embryo transfer has a potentially important role in creating ‘clean’ flocks, while enabling the introduction of new genes.59, 60
Pathogenesis
Experimental transmission of JS suggests that the respiratory system is the natural route of infection.84, 95
The primary lesion in JS is a well-differentiated multifocal tumour that originates from the transformation of type II pneumocytes and the non-ciliated bronchiolar (Clara) cells.48, 50, 68, 69, 84 Transformed cells, in which replication of the virus has been observed,62, 68 proliferate and form clusters that invade and eventually obliterate the alveolar lumen, resulting in death due to hypoxia. The transformed cells retain their secretory function, producing large amounts of surfactant-containing, clear, viscous fluid which accumulates in the air passages and aggravates respiratory distress. It also leads to coughing and the formation of aerosol droplets containing the virus or tumour cells, which can infect other animals.
Natural cases of JS are often complicated by secondary infections, most commonly pasteurellosis. In such cases the immediate cause of death is usually an acute or chronic pneumonia.
A characteristic histological feature of JS is the presence of large numbers of macrophages, usually in the areas adjacent to the adenomatous lesions. Various studies of their possible role in the pathogenesis of the disease have failed to provide a clear-cut answer. On the one hand, it has been clearly demonstrated that JS tumour cells produce a chemotactic factor which stimulates proliferation of macrophages, 45 while on the other hand, activated alveolar macrophages produce substances which stimulate the proliferation of type II cells. After chemotactic stimulation, macrophages have even been shown to produce both growth-stimulatory and inhibitory factors, depending on their interaction with other lymphoid cells.46 No evidence for activity of the alveolar macrophages against tumour cells has been found.
Another factor in the pathogenesis of JS about which little is known is the apparently close association of the disease with lentivirus infections.15, 64 Lentiviruses remain latent in alveolar macrophages and replicate only after these cells are activated.47 The presence of large numbers of activated macrophages in JS-affected lungs may therefore create ideal conditions for lentivirus replication, possibly aided by an immunosuppressive activity of the JSRV. It was recently demonstrated that a South African lentivirus isolate causes mild immunosuppression in sheep, the degree of which correlates well with the latent period before the appearance of JS signs in co-infected animals. It is possible that the lentivirus, in addition to causing a mild interstitial pneumonia, may also predispose animals to secondary infections, including JS.44
Clinical signs
In natural disease, the incubation period has been estimated at approximately five to six months, with two- to four-year-old sheep being most commonly affected.14 Clinical disease is therefore rarely seen in lambs under the age of six to nine months.19 Experimentally, however, because of the large number of lesions induced, the incubation period in lambs can be reduced to as short as three weeks by inoculation of concentrated semi-purified lung lavage material containing the aetiological agent in high concentrations. In these animals, the course of the disease is acute, with death often intervening within a few days of the first signs of the disease.75, 87
The onset of clinical signs is insidious in natural cases. Initially, while the habitus is normal and the condition of the animal is good, the respiratory rate is more rapid than normal after an animal has been driven. As the disease progresses, affected animals have reduced appetite, lose body weight, and lag behind the flock when it is driven. Marked respiratory distress is evident on exercise, the respiratory movements being short and jerky. Tachypnoea and dyspnoea eventually become evident even at rest. Spasmodic bouts of coughing occur and there is a great increase in the amount of secretion from the lungs. This is regarded as almost pathognomonic for JS.6, 43, 85 Moist râles are heard on auscultation of the thorax, sometimes even without the aid of a stethoscope.
There is generally no fever, but pyrexia may occur as a result of pneumonia following secondary bacterial infection.
The duration of the clinical disease varies considerably and depends largely on factors such as the presence, nature and extent of secondary bacterial pneumonia and on the environmental conditions under which the animal is kept. Sheep may survive for a few weeks or months, and cases are on record where affected animals have survived for more than a year.19 Results of a field survey indicate that the average duration of clinical disease is two months, with a range of a few days to six months.84 Once clinical signs manifest, the disease is irreversible.93
Pathology
No consistent haematological abnormalities have been reported in animals suffering from JS. However, increased blood levels of IgG occur in cases with advanced lesions,29, 65 and these have been ascribed to the presence of numerous plasma cells in the lungs.28
The extent and nature of the lung lesions depend to a large extent on the duration of the disease and whether or not secondary infection by organisms such as Trueperella (Actinomyces) pyogenes and Pasteurella spp. has occurred. The presence of secondary lesions in the lungs often obscures those due to the primary condition and renders the diagnosis of the disease tenuous on gross pathological evidence alone.
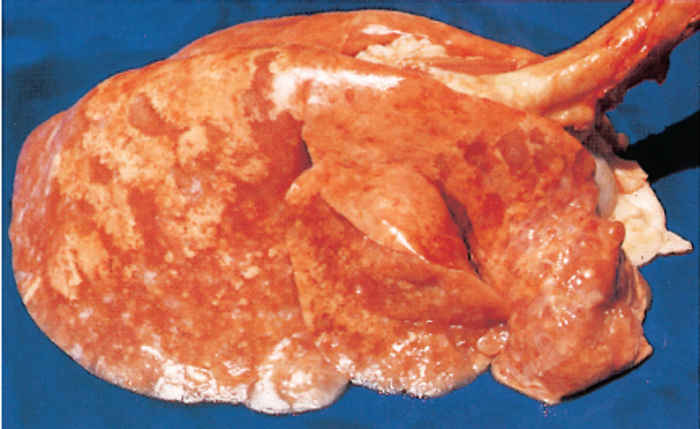
In advanced cases of JS, the lungs are three or more times their normal weight and almost fill the thoracic cavity. They do not collapse when the thorax is opened. Both lungs are usually involved, but not necessarily to the same extent. The cranial and middle lobes and the cranial part of the caudal lobes are commonly affected, but any part of the lung may be involved. Nodules and areas of diseased tissue of various sizes (the smallest being less than one millimetre in diameter) are sometimes scattered throughout the normal lung tissue. The lesions consist of very dense tumorous tissue and are greyish-white with a relatively firm consistency due to fibroplasia, which in some cases can be extensive (Figure 59.8).
The distribution of lesions suggests that the primary lesion or lesions grow by expansion. Intrapulmonary spread of the infection with the development of new foci probably occurs both aerogenously and via the lymph and bloodstream. Each new focus is seen grossly as a small greyishwhite, semi-transparent nodule which at first is barely visible to the naked eye. As it expands it may coalesce with neighbouring nodules and, in this way, large lesions eventually develop. The lesions of experimentally induced cases tend to be more multicentric than those resulting from natural infection.
Intra- and extrathoracic metastasis of the tumour may occur.1, 84 In South Africa extrapulmonary metastasis is rare, whereas it frequently occurs in sheep of the Awassi breed in Israel, where metastases both within and outside the thorax may be found in 30 to 50 per cent of cases.50, 66 This variation may also be linked to different viral strains.14 The most frequent metastatic sites are the bronchial and mediastinal lymph nodes, peritoneum, skeletal musculature, liver, spleen, kidneys, heart, and mesenteric lymph nodes.50, 84, 85, 95 Spread to the visceral and parietal pleura may also occur.84, 85
Apparently healthy sheep may, when slaughtered, reveal the presence of one, or perhaps more, isolated lesions, some of which are associated with fairly extensive fibroplasia. These lesions appear to be long-standing and give the impression that they grow very slowly.84
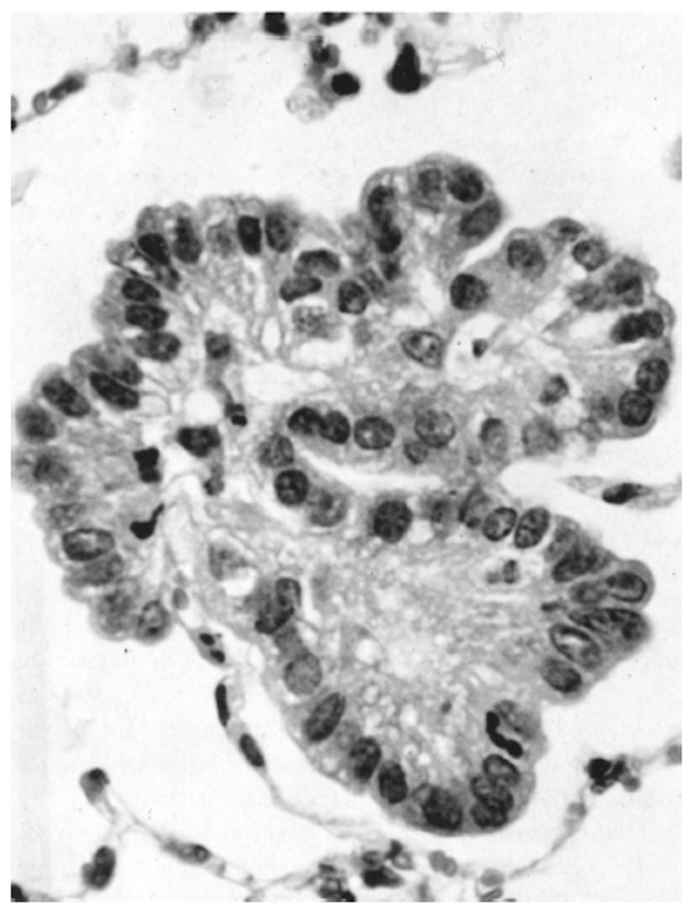
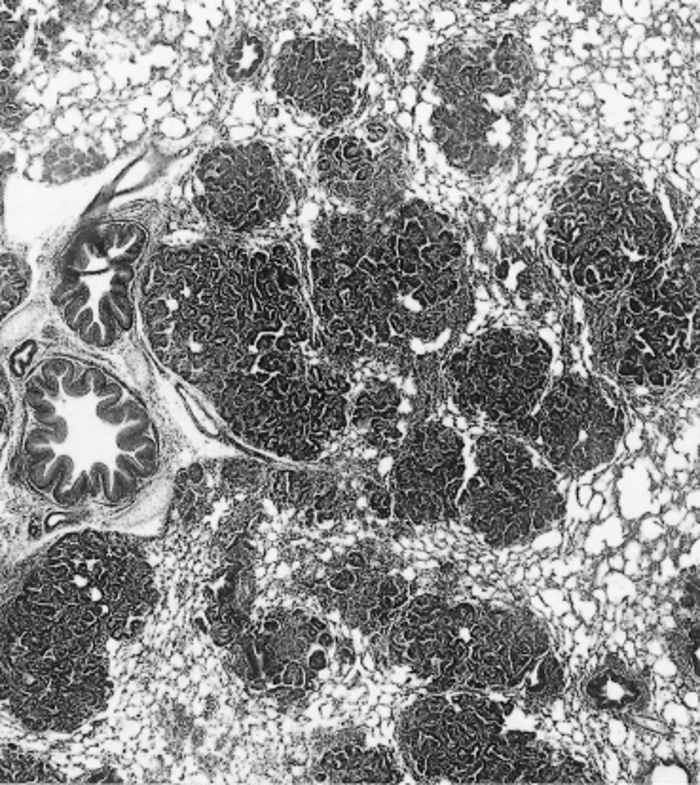
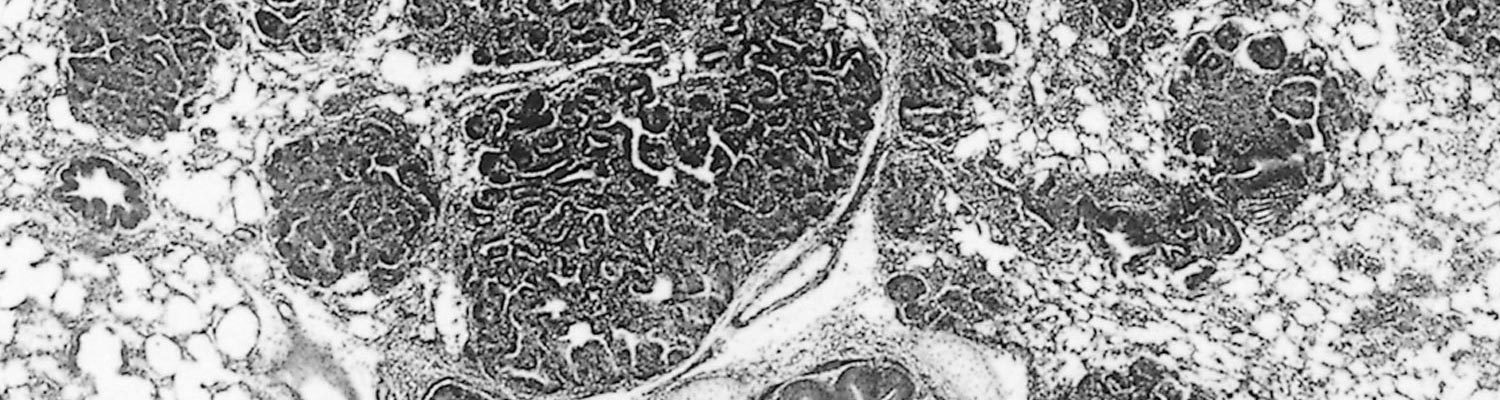
Histopathologically, the JS lesion has been classified as a bronchiolo-alveolar adenocarcinoma.82 The earliest lesions are found particularly in the epithelium of the alveoli, but also in the terminal and respiratory bronchioles. Very early lesions consist of a small number of neoplastic cuboidal to columnar epithelial cells which proliferate to form acinuslike structures that resemble glandular tissue (Figure 59.9). The acinus-like structures are generally lined by a single layer of well-differentiated tumour cells which are mainly cuboidal in shape, have large nuclei with several nucleoli, and often have a vacuolated cytoplasm. These cells stain intensely with the periodic-acid Schiff reagent before, but not after, treatment with diastase, indicating the presence of glycogen.40 In some acini, the tumour cells form papilliform projections into the lumina, particularly when bronchioles are involved. Neoplastic foci are scattered throughout the parenchyma (Figure 59.10).
In some cases, the connective tissue stroma is very prominent and may proliferate to form thick layers between the tumour acini or beneath the pleura. It generally consists of mature collagen, but occasionally may appear myxomatous (Figure 59.11).
A conspicuous feature of the lesions in animals with extensive disease is the alveolar macrophage response. These cells may be present within the lumens of the tumour acini, but are more prominent in the alveoli of the lung tissue adjacent to the neoplasm(Figure 59.12). Macrophages are not prominent in early lesions. Also conspicuous in some animals are perivascular, peribronchial and peribronchiolar lymphoid hyperplasia and increased numbers of plasma cells.
Evidence of exudative pneumonia due to secondary invaders is almost invariably present to a greater or lesser degree in and around the neoplastic lesions. This is generally represented by varying amounts of a fibrinous or purulent inflammatory reaction which may obscure the underlying primary lesion. Abscesses and pleuritis may be present.
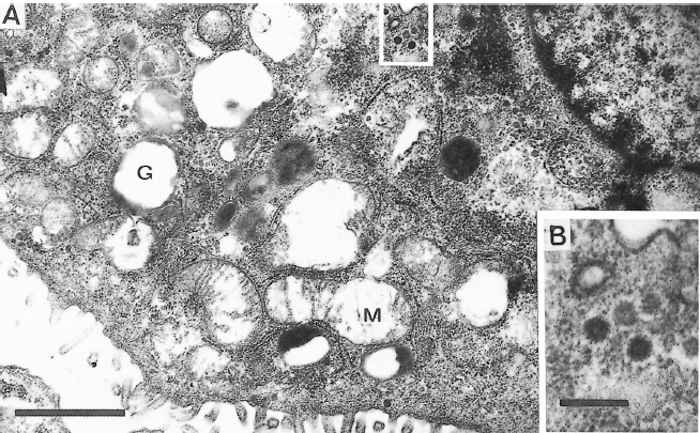
Ultrastructural studies of JS lesions11, 28, 48, 61, 68, 96 indicate that most neoplastic cells originate from granular (type II) pneumocytes. The cells on the periphery of the lesion are usually well differentiated, contain many free polysomes and show nucleolar margination, indicating that they are in a state of rapid protein synthesis and cell division. Intracytoplasmic JSRV particles, when seen, are often associated with centrioles, suggesting that the virus replicates in actively dividing cells.62 As is the case in most malignant cells, the nuclei and mitochondria are often abnormal. Numerous secretory granules, containing varying amounts of osmiophilic material and myelinoid bodies, are present in the cytoplasm. These granules are associated with surfactant production and are responsible for the large amount of lung secretion characteristic of JS.
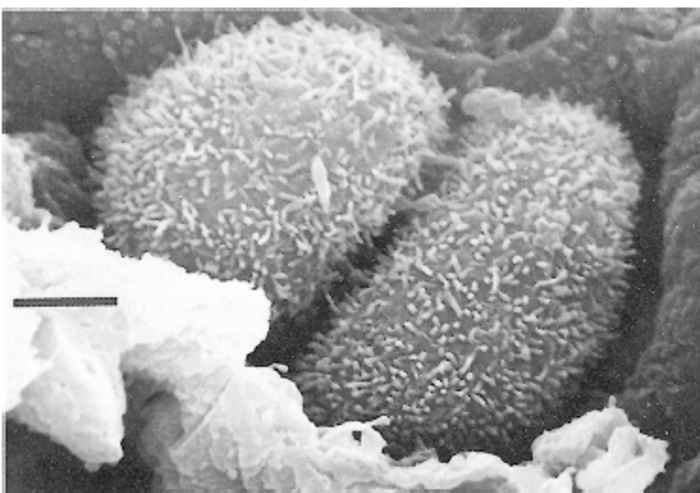
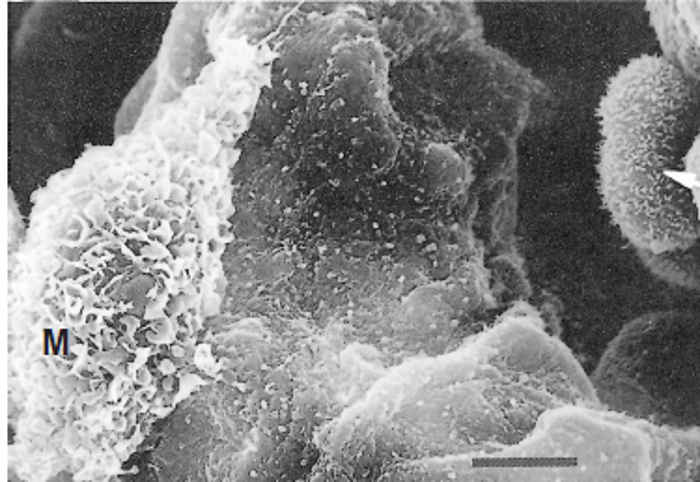
Scanning electron microscopy reveals that the surface of the neoplastic cells is usually covered by abundant microvilli, in stark contrast to the smooth surface of normal alveolar epithelial cells (Figure 59.13) . A single transformed cell first proliferates to form a single layer lining the alveolar lumen, which then develops into a grape-like cluster with papilliform projections eventually filling the alveolar space and spreading to adjacent alveoli (Figure 59.14) . The most common non-malignant cells, found mainly in advanced lesions, are macrophages. They have an ‘angry’ appearance, being large and possessing a highly ruffled surface(Figure 59.15) , indicating that they are in an activated state.
Diagnosis
The clinical diagnosis of JS cannot be made with certainty, especially in the first case to be encountered in a flock. A history of long-standing, progressively worsening respiratory distress syndrome in a flock, coupled with the results of clinical examination of affected animals, should alert one to the possible presence of JS. Up-ending an affected animal by its hind legs (often called the ‘wheel-barrow’ test) may, in cases with extensive lung pathology, induce the flow of a clear viscous fluid from the nostrils, supporting a diagnosis of JS.19, 43 In considering the diagnosis, it must be borne in mind that the course and clinical signs may be complicated by secondary pneumonia.
The diagnosis of JS must, at least initially, be confirmed by histopathological examination of the lung lesions. Lung neoplasia may be obscured, both macroscopically and microscopically, by secondary pneumonia. For this reason, several specimens of diseased tissue from different locations should be collected for histopathological examination, and reliance should not be placed on the results of the examination of a single specimen.84
All attempts at demonstrating circulating antibodies against exJSRV in natural or experimental cases of JS have failed, even in animals from which relatively large amounts of virus have been isolated from lung washes. In contrast, JSRV-specific IgA was found in lung fluid, suggesting an epithelial local immune response.91 The absence of a humoral response is possibly due to immune tolerance caused by the presence of enJSRV in all sheep. Expression of enJSRV antigens, closely related to those of exJSRV, in the neonatal period during ontogeny would lead to a depletion of JSRVantigen- reactive T- and B-lymphocyte populations. This would induce a state of immune tolerance with respect to JSRV antigens, both of endogenous and exogenous origin, so that no antibody immune response is launched upon infection with exJSRV.
In view of the recently demonstrated disseminated infection of lymphoid tissues by exJSRV,55 an alternative explanation for the lack of a humoral immune response might be interference with antibody formation by the immune system. 51 Whatever the explanation, the absence of circulating antibodies means that classical serological methods used as an indirect means to confirm infection cannot be used in the case of JS.
The use of alternative antigens such as MPMV-p2733 and a recombinant capsid protein of JSRV,51 both coupled to glutathione-S-transferase, enabled detection of a positive reaction in the sera of JS-affected sheep. However, these reactions were later found to be non-specific and of limited use.51
Various attempts have been made to produce both polyclonal and monoclonal antibodies to the JSRV proteins. The polyclonal antibodies prepared against semi-purified extracts of JS-affected lung fluid were relatively non-specific even after exhaustive absorption against normal lung proteins. 92 Three monoclonal antibodies selected using a duplicate screening assay (screened in parallel against both normal lung lavage antigens and antigens from JS-affected lungs) were extensively evaluated for their ability to specifically detect the exogenous infectious JSRV. One monoclonal antibody (Mab 4E10) was shown to react exclusively with lung biopsies from JS-affected sheep lungs as well as cells in a cell line established from a lung tumour of a JSaffected sheep.14, 100 Following an extensive investigation into this Mab, it was concluded that Mab 4E10 is not specific to a viral epitope but more likely against an epitope that is present on, or secreted in response to, or as a result of, the viral infection. Mab 4E10 also reacts against biopsies from human bronchiolo-alveolar carcinoma in a similar manner to that in JS-affected lungs.94 However, Mab 4E10 also reacts with the bronchioles in normal human lung sections derived from persons with other lung lesions due to other causes and some human lung cancers, thus supporting the view that the Mab is directed against an antigen that is not viral but rather one that is secreted in response to lung pathology. However, its value as a diagnostic tool to identify JS-affected sheep has been evaluated using an enzyme immunoassay (EIA) microtitre plate format and shown to be of value in screening nasal fluid from sheep showing clinical signs of the disease.100
The availability of nucleic acid sequence information from both the exogenous and endogenous viruses made it possible to search for unique sequences and restriction sites specific for either form of the virus. These differences were used to design molecular assays that could distinguish the exogenous from endogenous JSRV sequences and thereby identify those sheep infected with the exogenous infectious form of the virus.
One of the first molecular assays to be used for the specific detection of the exJSRV was a technique referred to as the ‘self-sustained sequence replication assay’ (3SR). This isothermic assay can be designed to amplify only singlestranded genomic material. The assay is both sensitive and specific, and theoretically would not amplify the endogenous viral sequences, which at the time were believed to exist only as endogenous proviral double-stranded DNA. An assay was developed to target the exJSRV gag gene and was shown to be able to detect the exJSRV in both lung lavage and nasal exudate material from JS-positive sheep but not from equivalent material obtained from non-infected sheep lungs.14, 97 The recent finding that endogenous viral transcripts were being produced and detected in noninfected (normal) lungs meant that these single-stranded transcripts would also be detected by the assay.
An improvement in the molecular detection of the exJSRV was the observation that the exJSRV had an additional ScaI restriction site that was present in the exJSRV but not in any enJSRV loci that had been sequenced to date.53 A PCR assay was developed that amplified a fragment that flanked the exJSRV-specific ScaI site. Digestion of the amplicon with ScaI followed by analysis on ethidium bromide stained agarose gels consistently distinguished the endogenous and exogenous viruses.3 Using this assay it was possible to identify the exJSRV in 100 per cent of lung biopsy samples analysed. It was therefore possible to use this assay to look for the distribution of the exJSRV in various cell populations. The molecular diagnosis of JSRV was further improved by the development of a hemi-nested PCR which targeted a unique section of the U3 region of the exJSRV that had mutations and deletions that were conserved in the exJSRV genomes and were not present in the enJSRV sequence. This assay was 105-fold more sensitive than the ScaI assay.55 Proviral DNA and JSRV transcripts were found in all tumours and lung secretions in all of 22 JS-affected sheep and in several lymphoid tissues. The virus was also detected in the mediastinal lymph nodes draining the lungs in all cases investigated as well as in other cells, although to a lesser degree. Unfortunately, from a diagnostic screening perspective, the assay has not yet been assessed extensively. The virus was only detected in peripheral blood monocytes from three out of seven whole blood samples, which would be the preferred material to be used to screen for the virus in asymptomatic sheep.
Differential diagnosis
Clinically and pathologically, JS can be confused with various forms of chronic pneumonia. In South Africa, pasteurellosis is the most commonly encountered lung condition in sheep, and is also the most common secondary complication in cases of JS. In countries where it occurs, maedi must also be differentiated from JS. Other organisms causing lung lesions include Trueperella (Actinomyces) pyogenes, Corynebacterium pseudotuberculosis, Mycoplasma spp., and lungworm.
Histologically, advanced JS lesions are so characteristic that a typical case should not be confused with any other disease. However, very early lesions may be difficult to distinguish from epithelialization of the alveoli, which tends to occur in chronic interstitial pneumonias caused by lungworm infestations and lentivirus infections. Co-infection with JSRV and lentivirus may give rise to ‘mixed lesions’. These lesions consist of the proliferative changes typical of JS as well as interstitial cell infiltration and collagen deposition characteristic of maedi.85, 87
Control
Jaagsiekte has been successfully eradicated from Iceland but this entailed the slaughter of the entire sheep population except in one isolated area.78 However, unless a sensitive diagnostic test for detecting carriers is developed, eradication will not be economically feasible in other countries. The alternative option of establishing and maintaining closed flocks free from infection is also hampered by the lack of techniques for determining whether or not an animal is free from infection. Under southern African conditions the prevalence in a flock can be reduced to less than 1 per cent by strict isolation and removal of animals showing early clinical signs. Furthermore, susceptibility to infection decreases rapidly after birth and lambs of four to six months of age are almost completely resistant to experimental infection. Therefore, if the lambs of infected ewes are eliminated together with their dams, the prevalence of JS can be further reduced.
The probability that the aetiological agent does not cross the placental barrier enhances the possibility of building up a ‘clean’ flock by either embryo transfer60 or by removing lambs from their dams by caesarian section or immediately after birth and subsequently raising them in a disease-free environment.59, 84 As natural infection by droplet inhalation requires close contact and retroviruses are relatively unstable when the atmosphere is dry and temperatures high, good management practices can contribute considerably to the control of the disease.
Apart from an age resistance in sheep to JS, circumstantial evidence exists for acquired resistance to infection, and for differences in breed susceptibility. In Iceland, initial losses in flocks first exposed to JS were high (50 to 60 per cent), but gradually declined to 5 to 10 per cent. English sheep breeds were found to be less susceptible in South Africa than were the Merino, Karakul and cross-breeds which make up the bulk of the sheep population. There is no evidence that this resistance is immunological.
The absence of a humoral response to exJSRV probably precludes the possibility of effective vaccination. However, progress that has been made in the past few years has opened up numerous research possibilities that did not exist before. Tools such as the complete exogenous and endogenous clones, their complete nucleic acid sequence and an infectious clone are now available. The regions of the exJSRV genome that are responsible for transformation and pathogenesis should be identifiable now that an infectious clone has been isolated and constructed. Understanding the mechanisms of oncogenesis should facilitate the development of counter-measures. With the complete genome sequence of both the exogenous and endogenous viruses known, it should be a formality to identify those genes/antigens that can be used to screen specifically for the presence of the exJSRV. The use of these antigens in sensitive and specific diagnostic assays will facilitate the early diagnosis and thereby control and possibly eradicate the disease from countries where JS causes significant economic losses.
In conclusion, from the research viewpoint, JS remains an excellent natural outbred model to study contagious epithelial cancers in other species.67 Results obtained in JS research will certainly impact on the progress made in understanding the equivalent human diseases, where aetiological agents have not yet been identified.4, 5, 8, 10
References
- AYNAUD, M., 1926. Origin vermineuse du cancer pulmonaire de la brebis. Comptus Rendus des Séances de la Société de Biologie, Paris, 95, 1540.
- BAI, J., BISHOP, J.V., CARLSON, J.O. & DEMARTINI, J.C., 1999. Sequence comparison of JSRV with endogenous proviruses: Envelope genotypes and a novel ORF with similarity to a G-protein-coupled receptor. Virology, 258, 333–343.
- BAI, J., ZHU, R-Y., STEDMAN, K., COUSENS, C., CARLSON, J., SHARP, J.M. & DEMARTINI, J.C., 1996. Unique long terminal repeat U3 sequences distinguish exogenous JS sheep retrovirus associated with ovine pulmonary carcinoma from endogenous loci in the sheep genome. Journal of Virology, 70, 3159–3168.
- BARKLEY, J.E. & GREEN, M.R., 1996. Bronchioloalveolar carcinoma. Journal of Clinical Oncology, 14, 2377–2386.
- BARSKY, S.H., CAMERON, R., OSANN, K.E., TOMITA, D. & HOLMES, E.C., 1994. Rising incidence of bronchiolo-alveolar lung carcinoma and its unique clinicopathologic features. Cancer, 73, 1163–1170.
- BASSETT, H.F. & SHEEHAN, P., 1989. Outbreak of sheep adenomatosis in the Irish Republic. The Veterinary Record, 124, 46–47.
- BASTIANELLO, S.S., 1982. A survey on neoplasia in domestic species over a 40-year period from 1935 to 1974 in the Republic of South Africa. II. Tumours occurring in sheep. Onderstepoort Journal of Veterinary Research, 49, 205–209.
- CLAYTON, F., 1986. Bronchioloalveolar carcinomas: Cell types, patterns of growth, and prognostic correlates. Cancer, 57, 1555–1564.
- COUSENS, C., MINGUIJON, E., DALZIEL, R.G., ORTIN, A., GARCIA, M., PARK, L., GONZALES, L., SHARP, J.M. & DE LAS HERAS, M., 1999. Complete sequence of enzootic nasal tumour virus, a retrovirus associated with transmissible intranasal tumors of sheep. Journal of Virology, 73, 3986–3993.
- CREMER, K.J. & GRUBER, J., 1992. Animal models of retrovirus-associated malignancies. Veterinary Pathology, 29, 572–578.
- CUTLIP, R.C. & YOUNG, S., 1982. Sheep pulmonary adenomatosis (jaagsiekte) in the United States. American Journal of Veterinary Research, 43, 2108–2113.
- CVJETANOVIC, V., FORSEK, Z., NEVJESTIC, A. & RUKANVIA, L.J., 1972. Isolation of the sheep pulmonary adenomatosis (SPA) virus. Veterinaria, 21, 493–496.
- DE KOCK, G., 1929. Further observations on the aetiology of jaagsiekte in sheep. Fifteenth Annual Report, Director of Veterinary Services, Union of South Africa, 1169–1183.
- DEMARTINI, J.C. & YORK, D.F., 1997. Retrovirus-associated neoplasms of the respiratory system of sheep and goats: Ovine pulmonary carcinoma and enzootic nasal tumor. Food Animal Retroviruses, 13, 55–70.
- DEMARTINI, J.C., ROSADIO, R.H. & LAIRMORE, M.D., 1988. The etiology and pathogenesis of ovine pulmonary carcinoma (sheep pulmonary adenomatosis). Veterinary Microbiology, 17, 219–236.
- DEMARTINI, J.C., ROSADIO, R.H., SHARP, J.M., RUSSEL, H.I. & LAIRMORE, M.D., 1987. Experimental co-induction of type D retrovirus-associated pulmonary carcinoma and lentivirus-associated lymphoid interstitial pneumonia in lambs. Journal of the National Cancer Institute, 79, 167–177.
- DE VILLIERS, E.-M., ELS, H.J. & VERWOERD, D.W., 1975. Characteristics of an ovine herpes virus associated with pulmonary adenomatosis (jaagsiekte) in sheep. South African Journal of Medical Science, 40, 165–175.
- DE VILLIERS, E.-M. & VERWOERD, D.W., 1980. Presence of Herpesvirus ovis DNA sequences in cellular DNA from sheep lungs affected with jaagsiekte (pulmonary adenomatosis). Onderstepoort Journal of Veterinary Research, 47, 109–112.
- DUNGAL, N., 1938. Epizootic adenomatosis of the lungs of sheep in relation to verminous pneumonia and jaagsiekte. Proceedings of the Royal Society of Medicine, 31, 497–505.
- DUNGAL, N., 1946. Experiments with jaagsiekte. American Journal of Pathology, 22, 737–759.
- DUNGAL, N., GISLASON, G. & TAYLOR, E.L., 1938. Epizootic adenomatosis in the lungs of sheep—comparisons with jaagsiekte, verminous pneumonia and progressive pneumonia. Journal of Comparative Pathology and Therapeutics, 51, 46–68.
- EBER, A., 1899. Über multiple adenombildung in den Lungen der Schafe. Zeitschrift für Tiermedizin, 3, 161–165.
- HARRIS, J.M., HAYNES, R.H. & MCINTOSH, E.M., 1997. A consensus sequence for a functional human endogenous retrovirus K (HER-K) dUTPase. Biochemistry & Cell Biology, 75, 143–151.
- HECHT, S.J., CARLSON, J.O. & DEMARTINI, J.C., 1994. Analysis of a type D retroviral capsid gene expressed in ovine pulmonary carcinoma and present in both affected and unaffected sheep genomes. Virology, 202, 480–484.
- HECHT, S.J., SHARP, J.M. & DEMARTINI, J.C., 1996. Retroviral aetiopathogenesis of ovine pulmonary carcinoma: A critical appraisal. British Veterinary Journal, 152, 395–409.
- HECHT, S.J., STEDMAN, K.E., CARLSON, J.O. & DEMARTINI, J.C., 1996. Distribution of endogenous type B and type D sheep retrovirus sequences in ungulates and other mammals. Proceedings of the National Academy of Science, US, 93, 3297–3302.
- HERRING, A.J., SHARP, J.M., SCOTT, F.M.M. & ANGUS, K.W., 1983. Further evidence for a retrovirus as the aetiological agent of sheep pulmonary adenomatosis (jaagsiekte). Veterinary Microbiology, 8, 237–249.
- HOD, I., HERZ, A. & ZIMBER, A., 1977. Pulmonary carcinoma (jaagsiekte) of sheep. Ultrastructural study of early and advanced tumour lesions. American Journal of Pathology, 86, 545–558.
- HOD, I., ZIMBER, A., KLOPFER, U., HELDER, A.W., NOBEL, T.A. & PERK, K., 1974. Pulmonary carcinoma (jaagsiekte) of sheep: Pathologic findings and comparison in multiple-case and case-free herds. Journal of the National Cancer Institute, 53, 103–110.
- HOLLAND, M.J., PALMARINI, M., GARCIA-GOTI, M., GONZALES, L. MCKENDRICK, I., DE LAS HERAS, M. & SHARP, J.M., 1999. Jaagsiekte retrovirus is widely distributed both in T and B lymphocytes and in mononuclear macrophages of sheep with naturally and experimentally acquired pulmonary adenomatosis. Journal of Virology, 73, 4004–4008.
- HOUWERS, D.J. & TERPSTRA, C., 1984. Sheep pulmonary adenomatosis. The Veterinary Record, 114, 23.
- HUTCHEON, D., 1891. Reply to query no. 191 about jagziekte or chronic catarrhal pneumonia. Agricultural Journal of the Cape of Good Hope, 4, 87–89.
- KWANG, J., KEEN, J., ROSATI, S. & TOLARI, F., 1995. Development and application of an antibody ELISA for the marker protein of ovine pulmonary carcinoma. Veterinary Immunology and Immunopathology, 47, 323–331.
- MACFADYEAN, J., 1894. Verminous pneumonia in the sheep. Journal of Comparative Pathology and Therapeutics, 7, 31–39.
- MACKAY, J.M.K., 1969. Tissue culture studies of sheep pulmonary adenomatosis (jaagsiekte). II. Transmission of cytopathic effects to normal cultures. Journal of Comparative Pathology, 79, 147–154.
- MAGER, D.L. & FREEMAN, J.D., 1995. HERV-H endogenous retroviruses: Presence in the New World branch but amplification in the Old World primate lineage. Virology, 213, 395–404.
- MALMQUIST, W.A., KRAUSS, H.H., MOULTON, J.E. & WANDERA, J.G., 1972. Morphologic study of virus-infected lung cell cultures from sheep pulmonary adenomatosis (jaagsiekte). Laboratory Investigation, 26, 528–533.
- MANSKY, L.M., 1998. Retrovirus mutation rates and their role in genetic variation. Journal of General Virology, 79, 1337–1345.
- MARKSON, L.M., SPENCE, J.B. & DAWSON, M., 1983. Investigation of a flock heavily infected with maedi-visna virus. The Veterinary Record, 112, 267–271.
- MARKSON, L.M. & TERLECKI, S., 1964. The experimental transmission of ovine pulmonary adenomatosis. Pathologica Veterinaria, 1, 269–288.
- MARTIN, W.B., SCOTT, M.M., SHARP, G.M., ANGUS, K.W. & NORVAL, M., 1976. Experimental production of sheep pulmonary adenomatosis (jaagsiekte). Nature, 264, 1976–77.
- MCCULLOUGH, S.J., MALONE, F.E. & GREENE, J.A., 1985. Sheep pulmonary adenomatosis in Ireland. The Veterinary Record, 116, 470.
- MITCHELL, D.T., 1915. Investigation into jagziekte or chronic catarrhal-pneumonia of sheep. Third and Fourth Reports of the Director Veterinary Research, Union of South Africa, 314, 585–614.
- MYER, M.S., HUCHZERMEYER, H.F.A.K., YORK, D.F., HUNTER, P., VERWOERD, D.W. & GARNETT, H.M., 1988. The possible involvement of immunosuppression caused by a lentivirus in the aetiology of jaagsiekte and pasteurellosis in sheep. Onderstepoort Journal of Veterinary Research, 55, 127–133.
- MYER, M.S., VERWOERD, D.W. & GARNETT, H.M., 1987. Production of a macrophage chemotactic factor by cultured jaagsiekte tumour cells. Onderstepoort Journal of Veterinary Research, 54, 9–15.
- MYER, M.S., VERWOERD, D.W. & GARNETT, H.M., 1987. Demonstration of growth-inhibitory as well as growth-stimulatory factors in medium conditioned by lung lavage cells stimulated with a chemotactic factor secreted by jaagsiekte tumour cells. Onderstepoort Journal of Veterinary Research, 54, 123–130.
- NARAYAN, O., WOLINSKY, J.S., CLEMENTS, J.E., STRANDBERG, J.D., GRIFFIN, D.E. & CORK, L.C., 1982. Slow virus replication: The role of macrophages in the persistence and expression of visna viruses of sheep and goats. Journal of General Virology, 59, 345–356.
- NISBET, D.I., MACKAY, J.M.K., SMITH, N. & GRAY, E.W., 1971. Ultrastructure of sheep pulmonary adenomatosis (jaagsiekte). Journal of Pathology, 103, 157–162.
- NOBEL, T.A., 1958. Pulmonary adenomatosis (jaagsiekte) in sheep with special reference to its occurrence in Israel. Refuah Veterinarith (Israel), 15, 98–101.
- NOBEL, T.A., NEUMANN, F. & KLOPFER, U., 1969. Histological patterns of the metastases in pulmonary adenomatosis of sheep (jaagsiekte). Journal of Comparative Pathology, 79, 537–540.
- ORTÍN, A., MINGUIJÓN, E., DWAR, P., GARCÍA, M., FERRER, L.M., PALMARINI, M., GONZALES, L., SHARP, J.M. & DE LAS HERAS, M., 1998. Lack of a specific immune response against a recombinant capsid protein of Jaagsiekte sheep retrovirus in sheep and goats naturally affected by enzootic nasal tumour or sheep pulmonary adenomatosis. Veterinary Immunology and Immunopathology, 61, 229–237.
- PALMARINI, M., DWAR, P. DE LAS HERAS, M., INGLES, N.F., DALZIEL, R.G. & SHARP, J.M., 1995. Epithelial tumour cells in the lungs of sheep with pulmonary adenomatosis are major sites of replication for Jaagsiekte retrovirus. Journal of General Virology, 76, 2731–2737.
- PALMARINI, M., COUSENS, C., DALZIEL, R.D., BAI, J., STEDMAN, K., DEMARTINI, J.C. & SHARP, J.M., 1996A. The exogenous form of jaagsiekte retrovirus is specifically associated with a contagious lung cancer of sheep. Journal of Virology, 70, 1618–1623.
- PALMARINI, M., HALLWIRTH, C., YORK, D., MURGIA, C., DE OLIVIERA, T., SPENCER, T. & FAN, H., 2000. Molecular cloning and functional analysis of three type D endogenous retroviruses of sheep reveal a different cell tropism from that of the highly related exogenous jaagsiekte sheep retrovirus. Journal of Virology, 74, 8065–8076.
- PALMARINI, M., HOLLAND, M.J., COUSENS, C., DALZIEL, R.G. & SHARP, J.M., 1996b. Jaagsiekte retrovirus establishes a disseminated infection of the lymphoid tissues of sheep affected by pulmonary adenomatosis. Journal of General Virology, 77, 2991–2998.
- PALMARINI, M., FAN, H. & SHARP, J.M., 1997. Sheep pulmonary adenomatosis: A unique model of retrovirus-associated lung cancer. Trends in Microbiology, 5, 478–483.
- PALMARINI, M., SHARP, M.J., DE LAS HERAS, M. & FAN, H., 1999. Jaagsiekte sheep retrovirus is necessary and sufficient to induce a contagious lung cancer in sheep. Journal of Virology, 73, 6964–6972.
- PALSSON, P.A., 1976. Maedi and visna in sheep. In: kimberlin, r.h., (ed.). Slow Virus Diseases of Animals and Man. Amsterdam: North-Holland Publishing Company.
- PARKER, B.W.J., WRATHALL, A.E., SAUNDERS, R.W., DAWSON, M., DONE, S.H. & BRADLEY, R., 1989. Outbreak of sheep pulmonary adenomatosis in the Irish Republic. The Veterinary Record, 177.
- PARKER, B.N.J., WRATHALL, A.E., SAUNDERS, R.W., DAWSON, M., DONE, S.H., FRANCIS, P.G., DEXTER, I. & BRADLEY, R., 1998. Prevention of transmission of sheep pulmonary adenomatosis by embryo transfer. The Veterinary Record, 142(25), 687–689.
- PAYNE, A.-L. & VERWOERD, D.W., 1984. A scanning and transmission electron microscopy study of jaagsiekte lesions. Onderstepoort Journal of Veterinary Research, 51, 1–13.
- PAYNE, A.-L., VERWOERD, D.W. & GARNETT, H.M., 1983. The morphology and morphogenesis of jaagsiekte retrovirus (JSRV). Onderstepoort Journal of Veterinary Research, 50, 317–322.
- PAYNE, A.-L., VERWOERD, D.W. & GARNETT, H.M., 1986. The localization of a Mason-Pfizer monkey virus-related antigen in jaagsiekte tumour tissue and cell lines. Onderstepoort Journal of Veterinary Research, 53, 87–91.
- PAYNE, A.-L., YORK, D.F., DE VILLIERS, E.-M., VERWOERD, D.W., QUERAT, G., BARBAN, V., SAUZE, N. & VIGNE, R., 1986. Isolation and identification of a South African lentivirus from jaagsiekte lungs. Onderstepoort Journal of Veterinary Research, 53, 55–62.
- PERK, K., 1982. Slow virus infections of ovine lung. Advances in Veterinary Science and Comparative Medicine, 26, 267–287.
- PERK, K., 1988. Koret School of Veterinary Medicine. The Hebrew University of Jerusalem. Personal communication.
- PERK, K. & HOD, I., 1982. Sheep lung carcinoma: An endemic analogue of a sporadic human neoplasm. Journal of the National Cancer Institute, 69, 747–749.
- PERK, K., HOD, I. & NOBEL, T.A., 1971. Pulmonary adenomatosis of sheep (jaagsiekte). I. Ultrastructure of the tumour. Journal of the National Cancer Institute, 46, 525–537.
- PERK, K., HOD, I., PRESENTEY, B. & NOBEL, T.A., 1971. Pulmonary adenomatosis of sheep (Jaagsiekte). II. Histogenesis of the tumor. Journal of the National Cancer Institute, 47, 197–205.
- PERK, K., MICHALIDES, R., SPIEGELMAN, S. & SCHLOM, J., 1974. Biochemical and morphologic evidence for the presence of an RNA tumour virus in pulmonary carcinoma of sheep (jaagsiekte). Journal of the National Cancer Institute, 53, 131–135.
- RAJYA, B.S. & SINGH, C.M., 1964. The pathology of pneumonia and associated respiratory disease of sheep and goats. I. Occurrence of jaagsiekte and maedi in sheep and goats in India. American Journal of Veterinary Research, 25, 61–67.
- ROSADIO, R.H., LAIRMORE, M.D., RUSSEL, H.I. & DEMARTINI, J.C., 1988. Retrovirus-associated ovine pulmonary carcinoma (sheep pulmonary adenomatosis) and lymphoid interstitial pneumonia. I. Lesion development and age susceptibility. Veterinary Pathology, 25, 475–483.
- ROSATI, S., PITTAU, M., ALBERTI, A., POZZI, S., YORK D.F., SHARP, J.M. & PALMARINI, M. (in preparation). An accessory open reading frame (orf-x) of jaagsiekte sheep retrovirus is conserved between different virus isolates.
- ROSS, S.R., 1998. Mouse mammary tumour virus and its interaction with the immune system. Immunologic Research, 17, 209–216.
- SHARP, J.M., ANGUS, K.W., GRAY, E.W. & SCOTT, F.M., 1983. Rapid transmission of sheep pulmonary adenomatosis (jaagsiekte) in young lambs. Archives of Virology, 78, 89–95.
- SHARP, J.M., ANGUS, K.W., JASSIM, F.A. & SCOTT, F.M.M., 1986. Experimental transmission of sheep pulmonary adenomatosis to a goat. The Veterinary Record, 119, 245.
- SHARP, J.M. & HERRING, A.J., 1983. Sheep pulmonary adenomatosis: Demonstration of a protein which cross reacts with the major core proteins of Mason-Pfizer monkey virus and mouse mammary tumour virus. Journal of General Virology, 64, 2323–2327.
- SIGURDSSON, B., 1958. Adenomatosis of sheep’s lungs. Experimental transmission. Archiv der Gesamte Virusforschung, 8, 51–58.
- SNYDER, S.P., DEMARTINI, J.C., AMEGHINO, E. & CALETTI, E., 1983. Coexistence of pulmonary adenomatosis and progressive pneumonia in sheep in the central sierra of Peru. American Journal of Veterinary Research, 44, 1334–1338.
- STEFANOU, D., TSANGARIS, T.H. & LEKKAS, S.T., 1975. Pulmonary adenomatosis in the goats in the District of Pieria (Greece). In: Twentieth World Veterinary Congress Summaries, Vol. 1, Thessaloniki, Greece.
- STEVENSON, R.G. & REHMTULLA, A.J., 1980. Pulmonary adenomatosis (jaagsiekte) in sheep in Canada. Canadian Veterinary Journal, 21, 267.
- STUNZI, H., HEAD, K.W. & NIELSEN, S.W., 1974. Tumours of the lung. Bulletin of the World Health Organization, 50, 9–19.
- TOUMAZOS, P., 1989. First report of sheep pulmonary adenomatosis in Cyprus. British Veterinary Journal, 145, 299–290.
- TUSTIN, R.C., 1969. Ovine jaagsiekte. Journal of the South African Veterinary Medical Association, 40, 3–23.
- TUSTIN, R.C., 1989. Faculty of Veterinary Science, University of Pretoria. Personal observation.
- TUSTIN, R.C., WILLIAMSON, A.L., YORK, D.F. & VERWOERD, D.W., 1988. Experimental transmission of jaagsiekte (ovine pulmonary adenomatosis) to goats. Onderstepoort Journal of Veterinary Research, 55, 27–32.
- VERWOERD, D.W., 1985–1989. Veterinary Research Institute, Onderstepoort. Unpublished data.
- VERWOERD, D.W., 1996. Ovine pulmonary adenomatosis (jaagsiekte). British Veterinary Journal, 152, 369–372.
- VERWOERD, D.W., 1990. Jaagsiekte (Ovine Pulmonary Adenomatosis). In: dinter, z. & morein, b., (eds). Virus Infections of Ruminants. Amsterdam: Elsevier.
- VERWOERD, D.W., DE VILLIERS, E-M. & TUSTIN, R., 1980. Aetiology of jaagsiekte: Experimental transmission to lambs by means of cultured cells and cell homogenates. Onderstepoort Journal of Veterinary Research, 47, 13–18.
- VERWOERD, D.W., PAYNE, A.-L., YORK, D.F. & MYER, M.S., 1983. Isolation and preliminary characterization of the jaagsiekte retrovirus (JSRV). Onderstepoort Journal of Veterinary Research, 50, 309–316.
- VERWOERD, D.W., TUSTIN, R.C. & PAYNE, A-L., 1985. Jaagsiekte: An infectious pulmonary adenomatosis of sheep. In: olsen, g., krakowka, s. & blakeslee, j.r., (eds). Comparative Pathobiology of Viral Diseases. Vol. II. Boca Raton, Florida: CRC Press Inc.
- VERWOERD, D.W., WILLIAMSON, A.L. & DE VILLIERS, E.-M., 1980. Aetiology of jaagsiekte: Transmission by means of subcellular fractions and evidence for the involvement of a retrovirus. Onderstepoort Journal of Veterinary Research, 47, 275–280.
- VINSEN, C., 2000. (In press)
- WANDERA, J.G., 1971. Sheep pulmonary adenomatosis. In: brandley, c.a. & cornelius, c.e., (eds). Advances in Veterinary Science and Comparative Medicine, 15, 251–283. New York, London: Academic Press.
- WANDERA, J.G. & KRAUSS, H.,1971. The ultrastructure of sheep pulmonary adenomatosis. Zentralblatt für Veterinärmedizin, 18, 325–334.
- YORK, D.F., 1987. A biochemical and immunological comparison of the jaagsiekte and two related retroviruses (PhD thesis). University of Natal, Pietermaritzburg, South Africa.
- YORK, D.F., VIGNE, R., VERWOERD, D.W. & QUERAT, G., 1991. Isolation, identification, and partial cDNA cloning of genomic RNA of jaagsiekte retrovirus, the etiological agent of sheep pulmonary adenomatosis. Journal of Virology, 65, 5061–5067.
- YORK, D.F., VIGNE, R., VERWOERD, D.W. & QUERAT, G., 1992. Nucleotide sequence of jaagsiekte sheep retrovirus, an exogenous and endogenous type D and B retrovirus of sheep and goats. Journal of Virology, 66, 4630–4939.
- YORK, D.F., VIGNE, R., VERWOERD, D.W., & QUERAT, G., 1993. The use of a self-sustained sequence replication assay (3SR) to detect the Jaagsiekte sheep retroviral genome. In: Proceedings of the Ninth International Congress of Virology, Glasgow, Scotland.