- Infectious Diseases of Livestock
- Part 2
- Pseudorabies
- GENERAL INTRODUCTION: PARAMYXOVIRIDAE AND PNEUMOVIRIDAE
- Rinderpest
- Peste des petits ruminants
- Parainfluenza type 3 infection
- Bovine respiratory syncytial virus infection
- Hendra virus infection
- Paramyxovirus-induced reproductive failure and congenital defects in pigs
- Nipah virus disease
- GENERAL INTRODUCTION: CALICIVIRIDAE AND ASTROVIRIDAE
- Vesicular exanthema
- Enteric caliciviruses of pigs and cattle
- GENERAL INTRODUCTION: RETROVIRIDAE
- Enzootic bovine leukosis
- Jaagsiekte
- Visna-maedi
- Caprine arthritis-encephalitis
- Equine infectious anaemia
- GENERAL INTRODUCTION: PAPILLOMAVIRIDAE
- Papillomavirus infection of ruminants
- Papillomavirus infection of equids
- GENERAL INTRODUCTION: ORTHOMYXOVIRIDAE
- Equine influenza
- Swine influenza
- GENERAL INTRODUCTION: CORONAVIRIDAE
- Porcine transmissible gastroenteritis
- Porcine respiratory coronavirus infection
- Porcine epidemic diarrhoea
- Porcine haemagglutinating encephalomyelitis virus infection
- Porcine deltacoronavirus infection
- Bovine coronavirus infection
- Ovine coronavirus infection
- Equine coronavirus infection
- GENERAL INTRODUCTION: PARVOVIRIDAE
- Porcine parvovirus infection
- Bovine parvovirus infection
- GENERAL INTRODUCTION: ADENOVIRIDAE
- Adenovirus infections
- GENERAL INTRODUCTION: HERPESVIRIDAE
- Equid herpesvirus 1 and equid herpesvirus 4 infections
- Equid gammaherpesvirus 2 and equid gammaherpesvirus 5 infections
- Equine coital exanthema
- Infectious bovine rhinotracheitis/infectious pustular vulvovaginitis and infectious pustular balanoposthitis
- Bovine alphaherpesvirus 2 infections
- Malignant catarrhal fever
- Pseudorabies
- Suid herpesvirus 2 infection
- GENERAL INTRODUCTION: ARTERIVIRIDAE
- Equine viral arteritis
- Porcine reproductive and respiratory syndrome
- GENERAL INTRODUCTION: FLAVIVIRIDAE
- Bovine viral diarrhoea and mucosal disease
- Border disease
- Hog cholera
- Wesselsbron disease
- Louping ill
- West nile virus infection
- GENERAL INTRODUCTION: TOGAVIRIDAE
- Equine encephalitides caused by alphaviruses in the Western Hemisphere
- Old World alphavirus infections in animals
- Getah virus infection
- GENERAL INTRODUCTION: BUNYAVIRIDAE
- Diseases caused by Akabane and related Simbu-group viruses
- Rift Valley fever
- Nairobi sheep disease
- Crimean-Congo haemorrhagic fever
- GENERAL INTRODUCTION: ASFARVIRIDAE
- African swine fever
- GENERAL INTRODUCTION: RHABDOVIRIDAE
- Rabies
- Bovine ephemeral fever
- Vesicular stomatitis and other vesiculovirus infections
- GENERAL INTRODUCTION: REOVIRIDAE
- Bluetongue
- Ibaraki disease in cattle
- Epizootic haemorrhagic disease
- African horse sickness
- Equine encephalosis
- Palyam serogroup orbivirus infections
- Rotavirus infections
- GENERAL INTRODUCTION: POXVIRIDAE
- Lumpy skin disease
- Sheeppox and goatpox
- Orf
- Ulcerative dermatosis
- Bovine papular stomatitis
- Pseudocowpox
- Swinepox
- Cowpox
- Horsepox
- Camelpox
- Buffalopox
- GENERAL INTRODUCTION: PICORNAVIRIDAE
- Teschen, Talfan and reproductive diseases caused by porcine enteroviruses
- Encephalomyocarditis virus infection
- Swine vesicular disease
- Equine picornavirus infection
- Bovine rhinovirus infection
- Foot-and-mouth disease
- GENERAL INTRODUCTION: BORNAVIRIDAE
- Borna disease
- GENERAL INTRODUCTION: CIRCOVIRIDAE AND ANELLOVIRIDAE
- Post-weaning multi-systemic wasting syndrome in swine
- GENERAL INTRODUCTION: PRION DISEASES
- Scrapie
- Bovine spongiform encephalopathy
- Transmissible spongiform encephalopathies related to bovine spongiform encephalopathy in other domestic and captive wild species
Pseudorabies
This content is distributed under the following licence: Attribution-NonCommercial CC BY-NC  View Creative Commons Licence details here
View Creative Commons Licence details here

Introduction
Pseudorabies (PR) is a disease caused by suid herpesvirus 1 and characterized by neurological signs in piglets, respiratory disease and lower growth performance in fattening pigs, and reproductive failures in sows. Older pigs generally survive the acute infection and carry the virus in a latent form for their entire life. Pseudorabies virus may be transmitted from pigs to other animal species, which invariably die from acute nervous disease.
The first to demonstrate that PR was caused by a virus different from rabies virus was Aladar Aujeszky in 1902.1 It is striking that pigs were not mentioned at all in his article, while at present it is well known that pigs are the natural host of the virus, and the only source of virus dissemination.
In the 1970s the economic importance of the disease increased as the pig industry intensified. Another reason for increased economic losses and spread of suid herpesvirus 1 [also known as pseudorabies virus (PRV)] might have been the emergence of more virulent strains.
The disease currently occurs virtually worldwide, particularly in countries where pigs are intensively raised. The UK (except Northern Ireland) and Denmark have succeeded in eradicating it; other free countries include Australia, Canada, Finland and Sweden. Eradication programmes are in progress in several countries, such as Belgium, France,Germany, Japan, the Netherlands, New Zealand and the USA.
Antibodies against PRV have been detected in wild boar (Sus scrofa) in Tunisia.38 No serological evidence of PRV infection in South Africa was found when sera from 5 337 pigs, obtained from slaughterhouses in the early 1990s, were examined. 100 Pseudorabies virus has been isolated from specimens from Zaïre. In spite of the importation of pigs from PR endemic areas, the disease does not appear to have become established in southern Africa.55
Aetiology
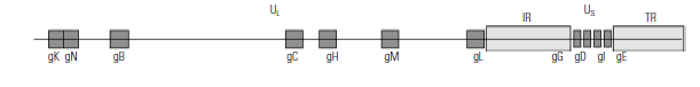
Suid herpesvirus 1 is classified in the family Herpesviridae, subfamily Alphaherpesvirinae, genus Varicellovirus. It has a size of 150 to 200 nm and contains double-stranded DNA of about 150 000 base pairs which encodes at least 70 proteins, of which more than 50 have been identified. The nucleoprotein is wrapped in a capsid consisting of 162 capsomeres. The capsid is surrounded by the tegument and the lipid bilayer envelope which harbours the glycoproteins. Eleven glycoproteins have been described: gB, gC, gD, gE, gG, gH, gI, gK, gL, gM and gN, of which gG is the only nonstructural one (Figure 82.1 ). The glycoproteins are involved in the complex interaction of PRV with its host and thus play a crucial role in the pathogenesis and immunology of the infection.
Glycoprotein C (gC) is the major attachment protein and binds to heparin sulphate proteoglycans on the cell surface, and subsequently gD stably binds to an unidentified cellular receptor. Glycoproteins B, D, H and L are involved in the penetration process. The glycoproteins gC, gE, gG, gI, gK, gM and gN are not essential for the growth of PRV in cell cultures. For efficient cell-to-cell spread, the glycoproteins gB, gD, gE, gH, gI, gK and gL are important. Glycoproteins gE and gI, which form a complex, promote cell-to-cell spread and are involved in virion release.66
Although restriction endonuclease fingerprints can differentiate between isolates and have been used to divide strains in four major genome groups,30, 71 PRV consists of one genotype. Only slight sequence variation can exist, e.g. less than 3 per cent in the gC gene.35 There is one antigenic type of PRV, in that antibodies against one isolate invariably neutralize another isolate. With the use of monoclonal antibodies, antigenic variation4, 40, 75 and antigenic conservation have been described.68, 92, 101
Pseudorabies virus has a broad range of virulence. Virulence indicates to what degree a particular virus strain produces disease; it can only be determined in standardized experimental infections of pigs. Highly virulent strains induce severe disease and mortality, whereas strains of low virulence give rise to mild disease or subclinical infections. Many genes play a role in the expression of virulence:
- genes encoding glycoproteins gE, gI, gM.6, 19, 65 A gE-negative PRV strain can infect first-order neurons, but has a greatly reduced capacity to cross synapses between neurons of olfactory and trigeminal routes, which may explain its reduced virulence for piglets;36, 50, 69
- genes coding for enzymes, such as UL23 (thymidine kinase), 47 UL40 (ribonucleotide reductase),18 UL50 (dUTPase), 37 and US3 (a protein kinase);43
- a gene that codes for protein UL21 involved in capsid formation, as shown in the Bartha vaccine strain;49 and
- latency and early protein genes.99
The virus is relatively stable against environmental influences. In an atmosphere of 55 per cent relative humidity and a variety of droplet sizes, PRV had a half-life of 44 minutes at 4 °C and 36 minutes at 22 °C. At this latter temperature, the virus would decline from 107 to 104 TCID50 in 7,8 hours. Pseudorabies virus is rapidly inactivated after exposure to ultraviolet light or drying. In water it is most stable at pH 7, and a 99,99 per cent inactivation can be recorded in four days at 25 °C. Virus survival on fomites varies greatly: e.g. on clothing and boots between less than one and seven days, in swine feed from less than 1 to 36 days, and in bedding and manure from less than one to three days.5
Epidemiology
The pig is the natural host of PRV and is most commonly infected by the intranasal route. There may be an inverse relationship between the virulence of the virus and the dose required to initiate infection. Whereas as few as 30 to 40 plaque-forming units (PFU) of virulent virus may suffice to infect a pig intranasally, mild-virulent strains seem to need about a 10-fold more infectious virus to establish an infection; the infectious dose of avirulent PRV is much higher.57, 91 In addition, pigs infected with virulent virus appear to shed more virus than those given mild-virulent virus.42, 57 Virulence thus may partly determine the extent of spread of PRV among pigs.
The pig is the only source of PRV dissemination. Virus excretion starts before the onset of clinical signs. After infection, it is excreted for 10 to 20 days with peak titres of 106 to 108 PFU in nasal and oropharyngeal fluids on days three to six; virus can also be excreted in ocular and vaginal fluids, and milk, albeit at a much lower level, and possibly in faeces and urine. Vaginal excretions after abortion may contain high virus titres.73
After reactivation induced by administration of corticosteroids, latently infected pigs usually excrete less virus during a shorter period than after primary infection. Acetylcholine may also reactivate latent PRV.86 However, an experimentally induced anaphylactic shock and drastic diet change did not result in virus re-excretion.58 Reactivation and subsequent excretion of latent virus do not appear to play an important role in the epidemiology of the disease. Spontaneous reactivation was found in a sow three to eight days after farrowing, but the sow’s own piglets did not become infected.14 Field studies yielded circumstantial evidence that natural reactivation of PRV seldom occurs.21, 53 In addition, during the eradication campaign in the UK, there was epidemiological evidence that latency is not as crucial a factor in the control as originally thought.87
The virus is primarily transmitted among pigs through direct nose-to-nose contact or over a few metres by aerosols generated by coughing and sneezing; the virus titres in these aerosols are 100- to 1 000-fold lower than in nasal fluid.10 Strains of PRV that induced most sneezing in infected pigs also produced the highest airborne infectivity.16 Pseudorabies virus has been isolated from air samples on farms,52 and house flies may mechanically transmit it.61
Herds usually become infected after introduction of pigs that shed the virus. Manure stored below the stables may contain infectious PRV which can survive for weeks, and after spreading over land may be a source of infection.8, 52 There is growing evidence that the virus may be transmitted from one herd to another in aerosols over nine or more kilometres. 13, 27 Semen may contain the virus and thus may be a vehicle for transmission by artificial insemination.61 If embryos with intact zona pellucida are treated with trypsin, embryo transfer will not contribute to PRV transmission.63 Humans and their clothing and equipment may mechanically transmit the virus among pig herds. The risk of transmitting PRV to pigs by feeding them pig meat (pork) is insignificant.17 Feral pigs in the USA and wild boar in Europe can also harbour PRV. In feral pigs the virus may be transmitted through sexual contact.80
A wide range of domestic animals and wildlife is susceptible to PRV under natural conditions. They can become infected by close contact with pigs by breathing viruscontaining forced air currents in a stable, or by ingesting infected pig tissue, mainly throats. The infective dose for cattle appears to be much higher than for pigs and may depend on the virus strain: after intranasal infection cattle apparently need between 1 600 and 40 000 TCID50 to become infected.7, 94 Dogs may be more susceptible if they have abrasions or wounds in their oral mucosa. Horses can also contract PR.41 Humans are not susceptible to PRV. Experimentally, most if not all animal species can be infected with PRV, and subsequently die. Animal species other than pigs may occasionally excrete the virus, but do not transmit it to in-contact animals and are thus considered dead-end hosts.
The latent state can be defined as the inability to detect infectious virus, whereas at the same time viral DNA can be demonstrated. The DNA can be triggered to reactivate, i.e. to start replication leading to the production of infectious virus, by high doses of corticosteroids. The reactivated virus after anterograde axonal transport may be excreted. Highly virulent strains of PRV may be more easily reactivated than strains of mild or low virulence.91 The virus may also persist in cells of the haemopoietic system, particularly bone marrow and may replicate at a low level.2, 76
Monocytes appear to be more susceptible for PRV than lymphocytes and granulocytes seem to be resistant.70, 74 The low-level viraemia, which can be cell-free and cell-associated, may lead to infection of parenchymatous organs, to transplacental infection with subsequent death of foetuses, and to abortion.73, 74
Pseudorabies virus infects bronchial epithelial cells, pulmonary alveolar cells and alveolar macrophages which can impair their function and lead to cell death.33 This may give way to secondary bacterial infections and these combined infections can run a much more severe course than the viral or bacterial infection alone, as has been shown for Pasteurella multocida,25 Streptococcus suis,34 Haemophilus parasuis72 and Actinobacillus pleuropneumoniae.81 A PRV infection given after an experimental infection with Mycoplasma hyopneumoniae markedly increased the severity of the pneumonia. 82
The immune response against PRV infection is characterized by the appearance of a systemic IgM response six to eight days after infection, followed by IgA, IgG1 and IgG2 antibodies. The IgG1 and IgG2 antibodies persist. The IgM, IgG1 and IgG2 isotypes can neutralize PRV; these neutralizing antibodies are mostly directed against gB, gC and gD. Antibodies are directed against a wide variety of structural and non-structural proteins of PRV and have an antiviral activity in complement-dependent cytotoxicity and in antibody- dependent cellular cytotoxicity processes. IgM and IgA antibodies can also be transiently detected on mucosal surfaces. A cell-mediated immune response can be demonstrated after infection characterized by the appearance of Thelper and cytotoxic T cells, and by the activation of natural killer cells.44, 45 Glycoprotein gC is a major target for cytotoxic T cells.102 The immunity after infection protects against a subsequent infection during the first months, probably through a strong secondary T cell response.44 Piglets acquire maternal antibodies through ingesting colostrum. These antibodies reduce replication of PRV, and consequently strongly inhibit the invasion of the central nervous system and thus protect against severe disease,51 but they do not protect against the establishment of latent infections.56, 64, 93 Maternal antibodies have a half-life of approximately 14 days.
Pathogenesis
The pathogenesis primarily depends on the virulence of the virus strain and age of the pigs. The virus enters the pig through the nasal mucosa and primarily replicates in epithelial cells of nasopharyngeal and olfactory mucosae (Figure 82.2), and tonsils. It spreads to the regional lymph nodes and the lungs, and is transported to the central nervous system through retrograde axonal transport via the sensory olfactorial and trigeminal nerves (Figure 82.3). The spread of PRV to the central nervous system is particularly abundant in young piglets. It infects the neurons of trigeminal ganglia and throughout the brain and invariably persists in a latent state in the trigeminal ganglia, probably in the olfactory bulbs,2 as well as in the tonsils. This neurotropism of PRV is currently exploited for tracing neural circuitry in living animals.22
The virus may use mechanisms to evade the immune response, such as the binding of complement factor C3 by glycoprotein gC32 and redistribution followed by endocytosis of viral glycoproteins on the cell membrane by viral antibodies, which may make the cell less prone to immune attack.24 Whether these processes operate in vivo is not yet known.
In non-porcine hosts, PRV is taken up by peripheral sensory nerves, and is transported to the related ganglia from where it spreads to the brain and spinal cord.
Clinical signs
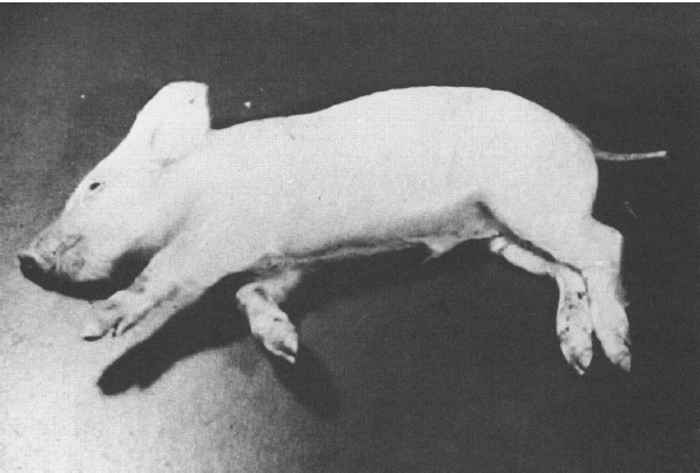
The age and immune status of the pig, and the virulence of the virus primarily determine the severity of clinical signs. The incubation period in neonatal pigs is one to three days. Piglets show signs of general illness: fever, anorexia, depression and occasional vomiting, as well as those associated with infection of the central nervous system, such as tremor, ataxia, convulsions, opisthotonus, paddling movements (Figure 82.4 ), and eventually paralysis with loss of consciousness. Mortality, which can occur one to three days after onset of disease signs, can approach 100 per cent. Maternally derived antibodies protect piglets from developing severe PR.
In older piglets, the same clinical signs can occur, but the disease usually runs a protracted and less severe course and consequently mortality is lower. Mortality rates decline progressively as age increases. In fattening pigs, respiratory signs such as nasal discharge, sneezing, coughing and dyspnoea are usually prominent, and are accompanied by fever and growth retardation. Occasionally, neurological signsmaybe observed. Pruritis is rare among pigs suffering from the disease.
In pregnant sows, which usually show no or very mild signs of disease, congenital infection may be followed by resorption of embryos in the early stage of development, abortion and farrowing of mummified foetuses.
Subclinical infections are quite common among pigs. It is not uncommon, in such cases, that the infection is traced to the death of a dog, cat or calf on the farm.
After an incubation period of a few days to at most one week, animals other than pigs generally show very severe neurological signs of disease for one to three days, and then invariably die from PR. The route of infection may partly determine the nature of the clinical signs of which the most prominent are anorexia, depression, fever, tremors, hyperexcitability, hypersalivation, severe pruritis resulting in automutilation, recumbency, ataxia and paralysis.
Pathology
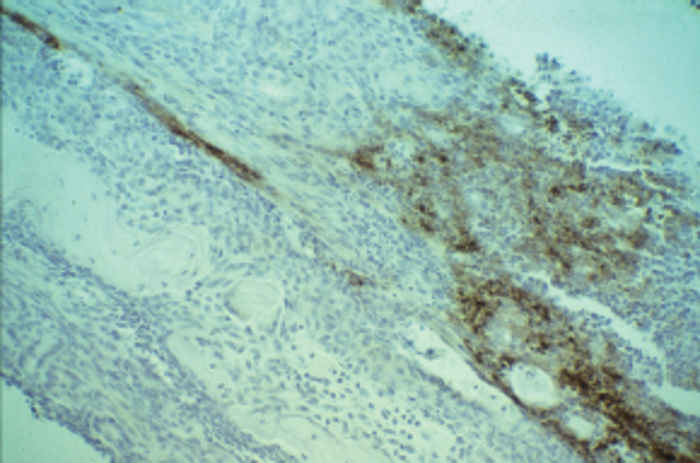
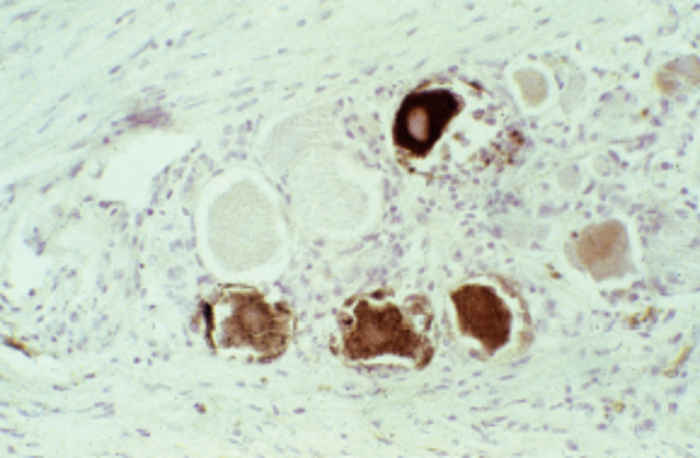
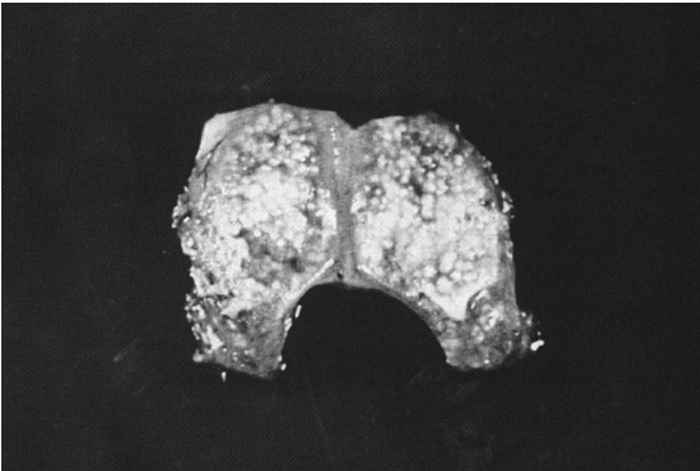
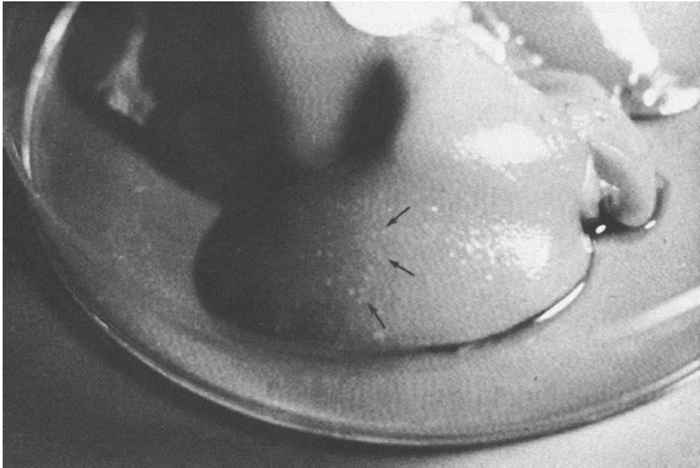
At necropsy there are usually few macroscopic lesions. Disseminated multifocal necrosis predominates. In pigs, necrotic mucosal surfaces of the upper respiratory tract with exudates ranging from serous to fibrinopurulent and necrotic tonsillitis can be observed (Figure 82.5 ). The regional lymph nodes of the upper respiratory tract may be swollen and haemorrhagic. Necrotic bronchitis, bronchopneumonia and pulmonary oedema may occur. Ganglioneuritis and non-suppurative meningoencephalitis with marked perivascular cuffing of mononuclear cells are predominant microscopic lesions. These inflammatory cells often exhibit pyknosis and karyorrhexis. Neuronal necrosis is widespread. Acidophilic intranuclear inclusion bodies in neurons and astrocytes develop in all animal species; fixation of nervous tissue in a mercurial fixative assists in their demonstration. 39 These inclusions in pigs are amphophilic, resembling those seen in other herpesvirus infections, but in other animal species they are granular and often small and multiple in an affected nucleus.39 Focal necrotic lesions, mostly surrounded by inflammatory cells, are frequently present in various tissues. Sows that have aborted may show endometritis and necrotic placentitis. In aborted foetuses and neonatal pigs, small foci of necrosis are evident in the liver and the spleen (Figure 82.6 ).
In animal species other than pigs, similar pathological lesions are present in sensory ganglia and central nervous system, mainly in the brain stem. Haemorrhages in the lungs and heart have been reported in some cattle, sheep and dogs.
Diagnosis
A presumptive diagnosis of PR in pigs can be made in animals manifesting the above clinical signs and/or lesions. To make a definite diagnosis, a number of pigs should be sampled for detection of virus and specific rises in antibody titres in paired sera. From living pigs, oropharyngeal or nasal fluids and acute and convalescent blood samples, and from dead pigs, samples of nasal mucosa, tonsil, brain, and lung tissues, are collected. In other animal species, portions of brain and spinal cord are the tissues of choice. The samples must be submitted to the laboratory under appropriate cooling conditions. Usually, virus isolation on porcine cell cultures with subsequent identification by monospecific antisera or monoclonal antibody is performed.76 Antigen detection tests in secretions and frozen tissue sections are not commonly employed in the laboratory diagnosis of PR. The polymerase chain reaction (PCR) is being increasingly used to detect PRV genome portions in clinical or cell culture samples. It is a rapid and highly sensitive test. Because of a chance that interfering substances in clinical samples may produce a false negative result, it is recommended that the sample be spiked before amplification with an internal control template. Furthermore, many test controls must be used to exclude contamination of samples before or during the reaction. The PCR harbours the risk of a false positive diagnosis, in the sense that the genome is found in case the PRV infection is not the (primary) cause of the disease. An advantage of the PCR is that the product can be sequenced, which may provide insight into strain differences of PRV, and thereby may help in epidemiological investigations of outbreaks. Most PRV vaccine strains can be easily differentiated from field strains by specific monoclonal antibodies or differential PCR.
When laboratory facilities are not available, rabbits can be inoculated with suspensions of tissue samples. A severe pruritis at the inoculation site developing within a few days followed rapidly by death, is strongly indicative for the presence of PRV. However, for humane reasons the use of the latter diagnostic method should be minimized as much as possible.
For a serological diagnosis, paired serum samples are examined for seroconversion (from seronegative to seropositive) or significant (four-fold) antibody increases against PRV. Usually, neutralization tests or enzyme-linked immunosorbent assays (ELISAs) are recommended and used for this purpose.77 Enzyme-linked immunosorbent assays have been developed that detect antibodies against whole virus antigen or specific glycoproteins whether or not produced as recombinant antigen in various expression systems. Irrespective of the test of choice, it should give a positive result with the OIE international standard serum.77 A definite diagnosis that PRV is the cause of disease is made when the virus is detected in secretions or tissues coupled to significant rises in antibody titres in several pigs. In other animal species, PRV detection in brain samples is sufficient for a definite diagnosis.
The serological diagnosis in countries where vaccination with so-called diva (to differentiate infected from vaccinated individuals) vaccines (also called marker vaccines) is widely practised, can be improved by the use of diva serological tests such as ELISAs to detect antibodies against gE, gG or gC.
Differential diagnosis
Neurological signs in piglets can also be caused by hog cholera, Teschen/Talfan disease, haemagglutinating encephalomyelitis virus infection, congenital tremors, or streptococcal or other bacterial forms of meningitis. Finishing pigs with respiratory disease may suffer from influenza, porcine reproductive and respiratory syndrome virus infection, and bacterial infections such as those caused by A. pleuropneumoniae, P. multocida, S. suis or M. hyopneumoniae. Reproductive failures in sows may have various infectious causes, e.g. parvovirus, encephalomyocarditis virus, pestiviruses, and leptospirosis, which thus can be of differential diagnostic importance.
In non-porcine hosts, diseases and intoxications that induce severe neurological signs should be taken into account as differential diagnoses, e.g. rabies.
Control
Introduction of PRV into a herd can best be prevented by only purchasing PRV-free pigs or by establishing a threeweek quarantine period for incoming pigs with unknown PR status. At the end of the quarantine period the pigs must be tested for antibodies to PRV and only groups of seronegative pigs may then be admitted to the herd. A second important control measure is strict hygiene. Cleaning and disinfecting of pens, vehicles, boots, and other objects are too often neglected. Disinfection can best be performed with 3 per cent formaldehyde or chlorine-releasing disinfectants, such as a 3 per cent chloramine solution. The presence of a sanitary room, where visitors must wash their hands, change boots and coveralls before entering the farm, can result in reduced introduction of PRV into the herd.84
Additional control strategies, e.g. the decision to eliminate PRV from a herd, are dependent on various factors, among which are its prevalence in the herd and region, costbenefit estimations and the existence of a regional or nationwide control programme. States or countries in which there is a low prevalence rate of antibodies against PRV in their swine herds may consider adopting a test-andremoval programme. Such a programme basically encompasses the detection, and subsequent slaughter, of seropositive herds or pigs. Repopulation is then established with seronegative pigs. Offspring segregation has also been advocated as an adequate control measure.88 Denmark and the UK, except Northern Ireland, had a low prevalence when they started to implement ‘test-and-removal’ eradication programmes in the beginning of the 1980s, and successfully effected these before 1990.
Countries with high prevalence rates of PRV in their pig herds obviously cannot follow the above strategies. If they do desire to control or eradicate PR the best option, also from an economic aspect, is first to lower the prevalence by intensive vaccination and, when a near-to-zero prevalence rate is reached, then to decide upon how to complete its eradication.60
The vaccines to be used in such a programme are the socalled diva vaccines. Although several types of these vaccines have been recognized or developed, e.g. gC-, gE-, and gG-negative, the gE-negative vaccines are the most widely applied. Live and killed gE-negative vaccines are available. The most well-known example of a classical vaccine contains the Bartha strain, which was developed by serially passaging a virus strain in cell cultures.3 The absence of fragment 7 in the restriction enzyme Bam HI cleavage profile of the Bartha strain,26 indicating a deletion in its genome compared to those of field strains, has been embraced to generate and select monoclonal antibodies that did not react with the Bartha strain, but did so with field strains of PRV. These monoclonal antibodies, which later proved to be directed against gE, were successfully employed in tests to differentiate antibodies after infection from those after vaccination. 95 This concept of diva vaccines and companion diagnostic tests has thus first been utilized in the field of PRV.
In the 1980s more insight was gradually gained in genes that play a role in the virulence of PRV and, by the advent of recombinant DNA technology, it became possible to delete or inactivate such non-essential genes and thus develop avirulent deletion mutants of PRV that could serve as vaccines. These deletion mutants usually carry an inactivated thymidine kinase gene and, in addition, have a glycoprotein gene deleted, either in gE, gG or gC.6, 47, 48, 54, 59, 65, 67, 79, 96 A glycoproteinsubunit vaccine devoid of gE has also been developed.12
The efficacy of these live and killed diva vaccines has been evaluated in vaccination-challenge experiments. Marked differences may exist in vaccine efficacy with respect to prevention of disease signs, reduction of challenge virus excretion and establishment of challenge virus latency. 90, 97, 98 Dissolving live vaccines in an adjuvant enhanced their efficacy.78 In general, live PRV diva vaccines are more efficacious than killed ones. In so-called transmission experiments in specified-pathogen-free pigs, it was demonstrated that gE-negative vaccines were able to significantly reduce transmission of field virus in small populations of specified-pathogen-free pigs under laboratory conditions.9, 15 In field trials, it was shown that in vaccinated breeding herds no major outbreaks occurred, whereas in vaccinated finishing herds major outbreaks may still be possible.83, 89
The safety of live and killed PRV diva vaccines has been demonstrated by numerous experiments. Although live vaccines have the potential to recombine,29 this is generally considered to be of no importance in the control of PR, particularly when only one type of diva vaccine is applied in a country. The most important disadvantage of live vaccines is the risk of contamination with pestiviruses.11 Live PRV vaccines must not be used in non-porcine animals, because this may result in the death of the vaccinated animal.
Companion diagnostic diva tests that can detect infected pigs in vaccinated populations are crucial in control/eradication programmes.91, 96 Most ELISAs to detect antibodies to gE are reliable and accurate,46, 85 and gE-antibodies appear to persist for years after infection.31, 91 In this regard, it is also essential to know that so far no field isolates have been found that lack the gE-protein.92
Results of eradication programmes based on intensive vaccination with gE-negative vaccines and determining the incidence of PRV infection with the gE-ELISA, indicate a marked decrease or virtual eradication of PRV from pig-dense areas.20, 23, 28 Once a country is free from PRV it should endeavour to prevent its introduction. Should the virus be introduced, a rapid diagnosis followed by ‘stamping out’ measures or the use of emergency diva vaccines is indicated.
References
- AUJESZKY, A., 1902. Ueber eine neue Infektionskrankheit bei Haustieren. Zentralblatt für Bakteriologie, Parasitenkunde und Infektionskrankheiten, 32, 353–357.
- BALASCH, M., PUJOLS. J., SEGALÉS, J., PLANA-DURAN, J. & PUMAROLA, M., 1998. Study of the persistence of Aujeszky’s disease (pseudorabies) virus in peripheral blood mononuclear cells and tissues of experimentally infected pigs. Veterinary Microbiology, 62, 171–183.
- BARTHA, A., 1961. Experiments to reduce the virulence of Aujeszky’s virus. Magyar Allatorvosok Lapja, 16, 42–25.
- BEN-PORAT, T., DEMARCHI, J.M., LOMNICZI, B. & KAPLAN, A.S., 1986. Role of glycoproteins of pseudorabies virus in eliciting neutralizing antibodies. Virology, 154, 325–334.
- BERAN, G.W., 1991. Transmission of Aujeszky’s disease virus. Proceedings First International Symposium on the Eradication of Pseudorabies (Aujeszky’s) Virus, St. Paul, Minnesota, USA, May 19–22, 1991. pp. 93–111.
- BERNS, A., VAN DEN OUWELAND, A., QUINT, W., VAN OIRSCHOT, J.T. & GIELKENS, A.L.J., 1985. Presence of markers for virulence in the unique short region or repeat region or both of pseudorabies hybrid viruses. Journal of Virology, 53, 89–93.
- BIRONT, P., VANDEPUTTE, J., PENSAERT, M.B. & LEUNEN, J., 1982. Vaccination of cattle against pseudorabies (Aujeszky’s disease) with homologous virus (herpes suis) and heterologous virus (herpes bovis 1). American Journal of Veterinary Research, 43, 760–763.
- BØTNER, A., 1991. Survival of Aujeszky’s disease virus in slurry at various temperatures. Veterinary Microbiology, 29, 225–235.
- BOUMA, A., DE JONG, M.C.M. & KIMMAN, T.G., 1997. Comparison of two pseudorabies virus vaccines, that differ in capacity to reduce virus excretion after a challenge infection, in their capacity of reducing transmission of pseudorabies virus. Veterinary Microbiology, 54, 113–122.
- BOURGUEIL, E., HUTET, E., CARIOLET, R. & VANNIER, P., 1992. Air sampling procedure for evaluation of viral excretion level by vaccinated pigs infected with Aujeszky’s disease (pseudorabies) virus. Research in Veterinary Science, 52, 182–186./li>
- CARNERO, R. & VANNIER, P., 1985. Effects pour le porc d’un virus propagé par un vaccin contre la maladie d’Aujeszky. Le Point Véterinaire, 17, 325–331.
- CHAPPUIS, G., FARGEAUD, D. & BRUN, A., 1989. Industrial production and control of a subunit vaccine against Aujeszky’s disease. In: van oirschot, j.t., (ed.). Vaccination and Control of Aujeszky’s disease. Dordrecht, The Netherlands: Kluwer Academic Publishers, pp. 67–78.
- CHRISTENSEN, L.S., MORTENSEN, S., BOTNER, A., STRANDBYGAARD, B.S., RONSHOLT, L., HENRIKSEN, C.A. & ANDERSEN, J.B., 1993. Further evidence of long distance airborne transmission of Aujeszky’s disease (pseudorabies) virus. The Veterinary Record, 132, 317–321.
- DAVIES, E.B. & BERAN, G.W., 1980. Spontaneous shedding of pseudorabies virus from a clinically recovered postparturient sow. Journal of the American Veterinary Medical Association, 176, 1345–1347.
- DE JONG, M.C.M. & KIMMAN, T.G., 1994. Experimental quantification of vaccine-induced reduction in virus transmission. Vaccine, 12, 761–766.
- DONALDSON, A.I., WARDLEY, R.C., MARTIN, S. & FERRIS, N.P., 1983. Experimental Aujeszky’s disease in pigs: Excretion, survival and transmission of the virus. The Veterinary Record, 113, 490–494.
- DONALDSON, A.I., WARDLEY, R.C., MARTIN, S. & HARKNESS, J.W., 1984. Influence of vaccination on Aujeszky’s disease virus and disease transmission. The Veterinary Record, 115, 121–124.
- WIND, N., BERNS, A., GIELKENS, A.L.J. & KIMMAN, T.G., 1993. Ribonucleotide reducaste-deficient mutants of pseudorabies virus are avirulent for pigs and induce partial protective immunity. Journal of General Virology, 74, 351–359.
- DIJKSTRA, J.M., GERDTS, V., KLUPP, B.G. & METTENLEITER, T.C., 1997. Deletion of glycoproteinMof pseudorabies virus results in attenuation for the natural host. Journal of General Virology, 78, 2147–2151.
- ELBERS, A.R.W. & STEGEMAN, A. 1996. Marked reduction of the prevalence of pseudorabies virus-infected pigs in pig dense regions of the Netherlands during the first year of a nation-wide vaccination campaign. Veterinary Quarterly, 18, 65–67.
- ENGEL, M., HIRD, D.W., CARPENTER, T.E. & WIERUP, M., 1995. Long-term lack of transmission of Aujeszky’s disease virus (ADV) in a chronically infected Swedish weaner pig producing herd. Preventive Veterinary Medicine, 24, 245–252.
- ENQUIST, L.W., HUSAK, P.J., BANFIELD, B.W. & SMITH, G.A., 1998. Infection and spread of alphaherpesviruses in the nervous system. Advances in Virus Research, 51, 237–347.
- EWALD, C., SCHMATZ, H.D., RUNGE, C., BAHNSEN, C., HEER, A. & WUTHE, H.H., 1994. Bekämpfung der Aujeszkyschen Krankheit in Schleswig-Holstein. 2. Mitteilung: Sanierung eines Gebietes durch Ausmerzung gI-positiver Zuchtschweine nach dreijahriger Flächenimpfung mit gI-deletierten Impfstoffen. Tierärtzliche Umschau, 49, 751–755.
- FAVOREEL, H.W., NAUWYNCK, H.J., VAN OOSTVELDT, P., METTENLEITER, T.C. & PENSAERT, M.B., 1997. Antibody-induced and cytoskeleton-mediated redistribution and shedding of viral glycoproteins, expressed on pseudorabies virus-infected cells. Journal of Virology, 71, 8254–8261.
- FUENTES, M.C. & PIJOAN, C., 1987. Pneumonia in pigs induced by intranasal challenge exposure with pseudorabies virus and Pasteurella multocida. American Journal of Veterinary Research 48, 1446–1448.
- GIELKENS, A.L.J., VAN OIRSCHOT, J.T., VAN SCHIE, F.W. & TOMA, B., 1984. Restriction endonuclease analysis of Aujeszky’s disease virus DNA. Current Topics in Veterinary Medicine and Animal Science, 29. In: Recent Advances in Virus Diagnosis, Boston: Martinus Nijhoff Publishers. pp. 101–114.
- GLOSTER, J., DONALDSON, A.I. & HOUGH, M.N., 1984. Analysis of a series of outbreaks of Aujeszky’s disease in Yorkshire in 1981–1982: The possibility of airborne disease spread. The Veterinary Record, 114, 234–239.
- GROSSE BEILAGE, E., FRIEDEL, K., BRUHN, F. & BOLLWAHN, R., 1997. Die Bekämpfung der Aujeszkyschen Krankheit mit Hilfe der Flächenimpfung und der Ausmerzung infizierter Zuchtschweine. III. Mitteilung: Eradikation der Aujeszkyschen Krankheit in infizierten Schweineherden am Beispiel des Sanierungsverfahrens im Landkreis Osnabrück/Niedersachsen. Deutsche Tierärtzliche Wochenschrift, 104, 393–400.
- HENDERSON, L., LEVINGS, R.L., DAVIS, A.J. & STURTZ, D.R., 1991. Recombination of pseudorabies virus vaccine strains in swine. American Journal of Veterinary Research, 52, 820–825.
- HERMANN, S-C., HEPPNER, B. & LUDWIG H., 1984. Pseudorabies viruses from clinical outbreaks and latent infections grouped into four major genotypes. In: wittmann, g., gaskell, r.m. & rziha, h-j., (eds). Latent Herpesvirus Infections in Veterinary Medicine. Current Topics in Veterinary Medicine and Animal Science, 27, 387–401.
- HOPP, W. & JUNGBLUT, R., 1994. Constancy of Aujeszky’s field virus antibody titres in sows repeatedly vaccinated with a gI-negative vaccine. Acta Veterinaria Hungarica, 42, 409–411.
- HUEMER, H.P., LARCHER, C. & COE, N.E., 1992. Pseudorabies virus glycoprotein III derived from virions and infected cells binds to the third component of complement. Virus Research, 23, 271–280.
- IGLESIAS, G.J., PIJOAN, C. & MOLITOR, T. 1989. Interactions of pseudorabies virus with swine alveolar macrophages. I. Virus replication. Archives of Virology, 104, 107–115.
- IGLESIAS, G.J. & TRUJANO, M. 1992. Inoculation of pigs with Streptococcus suis type 2 alone or in combination with pseudorabies virus. American Journal of Veterinary Research, 53, 364–367.
- ISHIKAWA, K., TSUTSUI, M., TAGUCHI, K., SAITOH, A. & MURAMATSU, M., 1996. Sequence variation of the gC gene among pseudorabies virus strains. Veterinary Microbiology, 49, 267–272.
- JACOBS, C.E., MULDER, W.A.M., VAN OIRSCHOT, J.T., GIELKENS, A.L.J. & KIMMAN, T.G., 1993. Deleting two amino-acids in glycoprotein I of pseudorabies virus decreases virulence and neurotropism for pigs, but does not affect immunogenicity. Journal of General Virology, 74,2201–2206.
- JONS, A., GERDTS, V., LANGE, E., KADEN, V. & METTENLEITER, T.C., 1997. Attenuation of dUTPase-deficient pseudorabies virus for the natural host. Veterinary Microbiology, 56, 47–54.
- JRIDI, M., BOUZGHAIA, H. & TOMA, B., 1996. Aujeszky’s disease in wild boar in Tunisia. Epidemiologie et Santé Animale, 30, 99–105.
- JUBB, K.V.F. & HUXTABLE, C.R., 1993. The nervous system. In: jubb, k.v.f., kennedy, p.c. & palmer, n., (eds). Pathology of Domestic Animals. 4th edn. San Diego: Academic Press, Inc.
- KATZ, J.B. & PEDERSON, J.C., 1992. Analysis of glycoprotein I (gI) negative and aberrant pseudorabies viral diagnostic isolates. American Journal of Veterinary Research, 53, 2259–2263.
- KIMMAN, T.G., BINKHORST, G.J., VAN DEN INGH, T.S.G.A.M., POL, J.M.A., GIELKENS, A.L.J. & ROELVINK, M.E., 1991. Aujeszky’s disease in horses fulfills Koch’s postulates. The Veterinary Record, 128, 103–106.
- KIMMAN, T.G., 1993. Characterization of the pseudorabies virus-specific immunoglobulinMresponse and evaluation of its diagnostic use in pigs with pre-existing immunity to the virus. Journal of Clinical Microbiology, 31, 2309–2314.
- KIMMAN, T.G., DE WIND, N., DE BRUIN, T., DE VISSER, Y. & VOERMANS, J., 1994. Inactivation of glycoprotein gE and thymidine kinase or the US3-encoded protein kinase synergistically decreases in vivo replication of pseudorabies virus and the induction of protective immunity. Virology, 205, 511–518.
- KIMMAN, T.G., DE BRUIN, T.M.G., VOERMANS, J.J.M. & BIANCHI, A.T.J., 1995. Development and antigen specificity of the lymphoproliferation response of pigs to pseudorabies virus: Dichotomy between secondary B- and T-cell responses. Immunology, 86, 372–378./li>
- KIMMAN, T.G., DE BRUIN, T.M.G, VOERMANS, J.J.M. & BIANCHI, A.T.J., 1996. Cell-mediated immunity to pseudorabies virus: Cytolytic effector cells with characteristics of lymphokine-activated killer cells lyse virus-infected and glycoprotein gB- and gC-transfected L14 cells. Journal of General Virology, 77, 987–990.
- KINKER, D.R., SWENSON, S.L., WU, L.L. & ZIMMERMAN, J.J., 1997. Evaluation of serological tests for the detection of pseudorabies gE antibodies during early infection. Veterinary Microbiology, 55, 99–106.
- KIT, S., KIT, M. & PIRTLE, E.C., 1985. Attenuated properties of thymidine-kinase-negative deletion mutant of pseudorabies virus. American Journal of Veterinary Research, 46, 1359–1367.
- KIT, S., SHEPPARD, M., ICHIMURA, J. & KIT, M., 1987. Second generation pseudorabies virus vaccine with deletions in the thymidine kinase and glycoprotein genes. American Journal of Veterinary Research, 35, 911–915.
- KLUPP, B.G., LOMNICZI, B., VISSER, N., FUCHS, W. & METTENLEITER, T.C., 1995. Mutations affecting the UL21 gene contribute to avirulence of pseudorabies virus vaccine strain Bartha. Virology, 212, 466–473.
- KRITAS, S.K., PENSAERT, M.B. & METTENLEITER, T.C., 1994. Role of gE, gI and gC in the invasion and spread of Aujeszky’s disease virus (ADV) in the olfactory pathway of the pig. Journal of General Virology, 75, 2319–2327.
- KRITAS, S.K., NAUWYNCK, H.J., PENSAERT, M.B. & KYRIAKIS, S.C., 1997. Effect of the concentration of maternal antibodies on the neural invasion of Aujeszky’s disease virus in neonatal pigs. Veterinary Microbiology, 55, 29–36.
- MACK, H., WEKERLE, J. & STRAUCH, D., 1986. Vorläufige Mitteilung über die Isolierung von Aujeszky virus as Fest- und Flüssigmist von Schweinen sowie aus Stalluft. Tierärztliche Umschau, 41, 32–38.
- MAES, L. & PENSAERT, M.B., 1984. Examination for virus persistence on swine fattening and breeding farms after an outbreak of Aujeszky’s disease. Tijdschrift voor Diergeneeskunde, 109, 439–445.
- MARCHIOLI, C.C., YANCEY, R.J., WARDLEY, R.C., THOMSEN, D.R. & POST, L.E., 1987. A vaccine strain of pseudorabies virus with deletions in the thymidine kinase and glycoprotein X genes. American Journal of Veterinary Research, 48, 1577–1583.
- MARÉ, C.J., 1994. Aujeszky’s disease. In: coetzer, j.a.w., thomson, g.r. & tustin, r.c., (eds). Infectious Diseases of Livestock with Special Reference to Southern Africa. Cape Town: Oxford University Press Southern Africa.
- MCCAW, M.B., OSORIO, F.A., WHEELER, J., XU, J. & ERICKSON, G.A., 1997. Effect of maternally acquired Aujeszky’s disease (pseudorabies) virus-specific antibody in pigs on establishment of latency and seroconversion to differential glycoproteins after low dose challenge. Veterinary Microbiology, 55, 91–98.
- MCCULLOUGH, S.J., 1989. Vaccination and control of Aujeszky’s disease in Northern Ireland. In: van oirschot, j.t., (ed.). Vaccination and Control of Aujeszky’s Disease. Dordrecht, The Netherlands: Kluwer Academic Publishers. pp. 231–238.
- MCFERRAN, J.B. & DOW, C., 1964. The excretion of Aujeszky’s disease virus by experimentally-infected pigs. Research Veterinary Science, 5, 405– 410./li>
- MCGREGOR, S., EASTERDAY, B.C., KAPLAN, A.S. & BERN-PORAT. T., 1985. Vaccination of swine with thymidine kinase-deficient mutants of pseudorabies virus. American Journal of Veterinary Research, 46, 1494–1497.
- MCINERNEY, J. & KOOIJ, D., 1997. Economic analysis of alternative AD control programmes. Veterinary Microbiology, 55, 112–121.
- MEDVECZKY, I. & SZABO, I., 1981. Isolation of Aujeszky’s disease virus from boar semen. Acta Veterinari Hungarica, 29, 29–35.
- MEDVECZKY, I., KOVACS, L., KOVACS, F. & PAPP, L., 1988. The role of the housefly, Musca domestica, in the spread of Aujeszky’s disease (pseudorabies). Medical and Veterinary Entomology, 2, 81–86.
- MEDVECZKY, I., SOLTI, L., HARASZTI, J., RONAY, G., EKES, K., BELAK, S., TURY, E., SEREGI, J. & VARGA, J., 1996. Transmission of Aujeszky’s disease (pseudorabies) virus is blocked by trypsin treatment of transferred embryos. Theriogenology, 46, 1357–1365.
- MENGELING, W.L., 1989. Latent infection and subsequent reactivation of pseudorabies virus in swine exposed to pseudorabies while nursing immune dams. American Journal of Veterinary Research, 50, 1658–1666.
- METTENLEITER, T.C., LUKACS, N. & RZIHA, H.-J., 1985. Pseudorabies virus avirulent strains fail to express a major glycoprotein. Journal of Virology, 56, 307–311.
- METTENLEITER, T.C., 1996. Immunobiology of pseudorabies (Aujeszky’s disease). Veterinary Immunology and Immunopathology, 54, 221–229.
- MOORMANN, R.J.M., DE ROVER, T., BRIAIRE, J., PEETERS, B.P.H., GIELKENS, A.L.J. AND VAN OIRSCHOT, J.T., 1990. Inactivation of the thymidine kinase gene of a gI deletion mutant of pseudorabies virus generates a safe but still highly immunogenic vaccine strain. Journal of General Virology, 71, 1591–1595.
- MORENKOV, O.S., FODOR, N., SOBKO, Y.A. & FODOR, I., 1997. Immunological characterisation of glycoprotein E of Aujeszky’s disease virus. Virus Research, 51, 65–79.
- MULDER, W.A.M., JACOBS, C.E., PRIEM, J., KOK, G.L., WAGENAAR, F., KIMMAN, T.G. & POL, J.A.M., 1994. Glycoprotein E-negative pseudorabies virus has a reduced capability to infect second- and third-order neurons of the olfactory and trigeminal routes in the porcine central nervous system. Journal of General Virology, 75, 3095–3106.
- MULDER, W.A.M., PRIEM, J., POL, J.M.A. & KIMMAN, T.G., 1995. Role of viral proteins and concanavalin A in in vitro replication of pseudorabies virus in porcine peripheral blood mononuclear cells. Journal of General Virology, 76, 1433–1442.
- MÜLLER, T., KLUPP, B., ZELLMER, R., TEUFFERT, J., ZIEDLER, K., POSSARDT, C., MEWES, L., DRESENKAMP, B., CONRATHS, F.J. & METTENLEITER, T.C., 1998. Characterisation of pseudorabies virus isolated from wild boar. The Veterinary Record, 143, 337–340.
- NARITA, M., KAWASHIMA, K., MATSUURA, S., UCHIMURA, A. & MIURA, Y., 1994. Pneumonia in pigs with pseudorabies virus and Haemophilus parasuis Serovar 4. Journal of Comparative Pathology, 110, 329–339.
- NAUWYNCK, H.J. & PENSAERT M.B., 1992. Abortions induced in vaccinated sows by cell-associated pseudorabies (Aujeszky’s disease) virus. American Journal of Veterinary Research, 53, 489–493.
- NAUWYNCK, H.J. & PENSAERT, M.B., 1994. Cell-free and cell-associated viremia in pigs after oronasal infection with Aujeszky’s disease virus. Veterinary Microbiology, 43, 307–314.
- NOSETTO, E.O., ECHEVERRIA, M.G., HORIMOTO, T., PECORARO, M.R., GALOSI, C.M., TOHYA, Y., NORIMINE, J., TAKAHASHI, E., ETCHEVER-RIGARAY, M.E. & MIKAMI, T., 1997. Characterization of several pseudorabies viral strains by virus-neutralization test using monoclonal antibodies. Viral Immunology, 10, 159–164.
- OHLINGER., V.F., HECK, BEHRENS, RZIHA, H.-J., WEILAND & WITTMANN, G., 1987. Die Infektion von Zellen des hämatopoetischen Systems durch das Aujeszkyvirus. Tierärztliche Umschau, 42, 210–219.
- OFFICE INTERNATIONAL DES EPIZOOTIES, 1996. Manual of Standards for Diagnostic Tests and Vaccines. pp. 181–191.
- PENSAERT, M.B., DE SMET, K. & DE WAELE, K., 1990. Extent and duration of virulent virus excretion upon challenge of pigs vaccinated with different glycoprotein-deleted Aujeszky’s disease vaccines. Veterinary Microbiology, 22, 107–117.
- QUINT, W.G.V., GIELKENS, A.L.J., VAN OIRSCHOT, J.T., BERNS, A.L.M. & CUYPERS, H.T., 1987. Construction and characterization of deletion mutants of pseudorabies virus: a new generation of ‘live’ vaccines. Journal of General Virology, 68, 523–534.
- romero, c.h., meade, p., santagata, j., gillis, k., lollis, g., hahn, e.c. & gibbs, e.p.j., 1997. Genital infection and transmission of pseudorabies virus in feral swine in Florida, USA. Veterinary Microbiology, 55, 131– 139.
- sakano, t., shibata, i., samegai, y., taneda, a., okada, m., irisawa, t. & sato, s., 1993. Experimental pneumonia in pigs infected with Aujeszky’s disease virus and Actinobacillus pleuropneumoniae. Journal of Veterinary Medical Science, 55, 575–579.
- shibata, i., okada, m., uruno, k., samegai, y., ono, m., sakano, t. & sato, s., 1998. Experimental dual infection of cesarean-derived, colostrum-deprived pigs with Mycoplasma hyopneumoniae and pseudorabies virus. Journal of Veterinary Medical Science, 60, 295–300.
- stegeman, j.a., van nes, a., de jong, m.c.m. & bolder, f.w.m., 1995. Assessment of the effectiveness of vaccination against pseudorabies finishing pigs. American Journal of Veterinary Research, 56, 573–578.
- stegeman, j.a., de jong, m.c.m., van nes, a. & bouma, a., 1997a. Dynamics of pseudorabies virus infections in vaccinated pigs populations: A review. Veterinary Quarterly, 19, 117–122.
- stegeman, j.a., de jong, m.c.m., van der heijden, h.m.j.f., elbers, a.r.w. & kimman, t.g., 1997b. Evaluation of tests for detection of antibodies to Aujeszky’s disease (pseudorabies) virus glycoprotein E in the target population. Veterinary Microbiology, 55, 107–111.
- tanaka, s., imamura, t., sakaguchi, m., mannen, k. & matsuo, k., 1996. Acetylcholine activates latent pseudorabies virus in pigs. Archives of Virology, 141, 161–166.
- taylor, k.c., 1989. Epidemiological aspects of Aujeszky’s disease control in Great Britain. In: van oirschot, j.t., (ed.). Vaccination and Control of Aujeszky’s Disease. Current Topics in Veterinary Medicine and Animal Science, 49, 185–196.
- thawley, d.g., gustafson, d.p. & beran, g.w., 1982. Procedures for the elimination of pseudorabies virus from herds of swine. Journal of the American Veterinary Medical Association, 181, 1513–1518.
- van nes, a., stegeman, j.a., de jong, m.c.m., loeffen, w.l.a., kimman, t.g. & verheijden, j.h.m., 1996. No major outbreaks of pseudorabies virus in well-immunised sow herds. Vaccine, 14, 1042–1044.
- vannier, p., huet, e., bourgueuil, e. & cariolet, r., 1991. Level of virulent virus excreted by infected pigs previously vaccinated with different glycoprotein deleted Aujeszky’s disease vaccines. Veterinary Microbiology, 29, 213–223.
- van oirschot, j.t., 1988. Induction of antibodies to gI in pigs exposed to different doses of a mildly virulent strain of Aujeszky’s disease virus. The Veterinary Record, 122, 599–603.
- VAN OIRSCHOT, J.T., 1989. The antibody response to glycoprotein I and the control of Aujeszky’s disease virus. In: van oirschot, j.t., (ed.). Vaccination and Control of Aujeszky’s Disease. Dordrecht, The Netherlands: Kluwer Academic Publishers. pp. 129–138.
- VAN OIRSCHOT, J.T. & GIELKENS, A.L.J., 1984. In vivo and in vitro reactivation of latent pseudorabies virus in pigs born to vaccinated sows. American Journal of Veterinary Research, 45, 567–571.
- VAN OIRSCHOT, J.T., DE LEEUW, P.W. & TIESSINK, J.W.A., 1985. Vaccination confers protection against Aujeszky’s disease in cattle. Zentralblatt für Veterinärmedizin, B, 32, 173–180.
- VAN OIRSCHOT, J.T., RZIHA, H.J., MOONEN, P.L.J.M., POL, J.M.A. & VAN ZAANE, D., 1986. Differentiation of serum antibodies from pigs vaccinated or infected with Aujeszky’s disease virus by a competitive enzyme immunoassay. Journal of General Virology, 67, 1179–1182.
- VAN OIRSCHOT, J.T., GIELKENS, A.L.J., MOORMANN, R.J.M. & BERNS, A.J.M., 1990. Marker vaccines, virus protein-specific antibody assay and the control of Aujeszky’s disease. Veterinary Microbiology, 23, 85–101.
- VAN OIRSCHOT, J.T., MOORMANN, R.J.M., BERNS, A.J.M. & GIELKENS, A.L.J., 1991. Efficacy of a pseudorabies virus vaccine based on deletion mutant strain 783 that does not express thymidine kinase and glycoprotein I. American Journal of Veterinary Research, 52, 1056–1060.
- VILNIS, A., SUSSMAN, M.D., THACKER, B.J., SENN, M. & MAES, R.K., 1998. Vaccine genotype and route of administration affect pseudorabies field virus latency load after challenge. Veterinary Microbiology, 62: 2, 81–96.
- WESLEY, R.D. & CHEUNG, A.K., 1996. A pseudorabies virus mutant with deletions in the latency and early protein O genes: Replication, virulence, and immunity in neonatal piglets. Journal of Veterinary Diagnostic Investigation, 8: 1, 21–24.
- WILLIAMS, R., ESTERHUYSEN, J.J. & ROBINSON, J.T.R., 1994. Pseudorabies and transmissible gastroenteritis: A serological survey in South Africa. Onderstepoort Journal of Veterinary Research, 61, 67–70.
- ZARIPOV, M.M., MORENKOV, O.S., SIKLODI, B., BARNAVETRO, I., GYONGYOSI HORVATH, A. & FODOR, I., 1998. Glycoprotein B of Aujeszky’s disease virus: Topographical epitope mapping and epitope-specific antibody response. Research in Virology, 149, 29–41.
- ZUCKERMANN, F.A., ZSAK, L., METTENLEITER, T.C. & BEN-PORAT, T., 1990. Pseudorabies virus glycoprotein gIII is a major target antigen for murine and swine-specific cytotoxic T lymphocytes. Journal of Virology, 64, 802–812.