- Infectious Diseases of Livestock
- Part 2
- Borna disease
- GENERAL INTRODUCTION: PARAMYXOVIRIDAE AND PNEUMOVIRIDAE
- Rinderpest
- Peste des petits ruminants
- Parainfluenza type 3 infection
- Bovine respiratory syncytial virus infection
- Hendra virus infection
- Paramyxovirus-induced reproductive failure and congenital defects in pigs
- Nipah virus disease
- GENERAL INTRODUCTION: CALICIVIRIDAE AND ASTROVIRIDAE
- Vesicular exanthema
- Enteric caliciviruses of pigs and cattle
- GENERAL INTRODUCTION: RETROVIRIDAE
- Enzootic bovine leukosis
- Jaagsiekte
- Visna-maedi
- Caprine arthritis-encephalitis
- Equine infectious anaemia
- GENERAL INTRODUCTION: PAPILLOMAVIRIDAE
- Papillomavirus infection of ruminants
- Papillomavirus infection of equids
- GENERAL INTRODUCTION: ORTHOMYXOVIRIDAE
- Equine influenza
- Swine influenza
- GENERAL INTRODUCTION: CORONAVIRIDAE
- Porcine transmissible gastroenteritis
- Porcine respiratory coronavirus infection
- Porcine epidemic diarrhoea
- Porcine haemagglutinating encephalomyelitis virus infection
- Porcine deltacoronavirus infection
- Bovine coronavirus infection
- Ovine coronavirus infection
- Equine coronavirus infection
- GENERAL INTRODUCTION: PARVOVIRIDAE
- Porcine parvovirus infection
- Bovine parvovirus infection
- GENERAL INTRODUCTION: ADENOVIRIDAE
- Adenovirus infections
- GENERAL INTRODUCTION: HERPESVIRIDAE
- Equid herpesvirus 1 and equid herpesvirus 4 infections
- Equid gammaherpesvirus 2 and equid gammaherpesvirus 5 infections
- Equine coital exanthema
- Infectious bovine rhinotracheitis/infectious pustular vulvovaginitis and infectious pustular balanoposthitis
- Bovine alphaherpesvirus 2 infections
- Malignant catarrhal fever
- Pseudorabies
- Suid herpesvirus 2 infection
- GENERAL INTRODUCTION: ARTERIVIRIDAE
- Equine viral arteritis
- Porcine reproductive and respiratory syndrome
- GENERAL INTRODUCTION: FLAVIVIRIDAE
- Bovine viral diarrhoea and mucosal disease
- Border disease
- Hog cholera
- Wesselsbron disease
- Louping ill
- West nile virus infection
- GENERAL INTRODUCTION: TOGAVIRIDAE
- Equine encephalitides caused by alphaviruses in the Western Hemisphere
- Old World alphavirus infections in animals
- Getah virus infection
- GENERAL INTRODUCTION: BUNYAVIRIDAE
- Diseases caused by Akabane and related Simbu-group viruses
- Rift Valley fever
- Nairobi sheep disease
- Crimean-Congo haemorrhagic fever
- GENERAL INTRODUCTION: ASFARVIRIDAE
- African swine fever
- GENERAL INTRODUCTION: RHABDOVIRIDAE
- Rabies
- Bovine ephemeral fever
- Vesicular stomatitis and other vesiculovirus infections
- GENERAL INTRODUCTION: REOVIRIDAE
- Bluetongue
- Ibaraki disease in cattle
- Epizootic haemorrhagic disease
- African horse sickness
- Equine encephalosis
- Palyam serogroup orbivirus infections
- Rotavirus infections
- GENERAL INTRODUCTION: POXVIRIDAE
- Lumpy skin disease
- Sheeppox and goatpox
- Orf
- Ulcerative dermatosis
- Bovine papular stomatitis
- Pseudocowpox
- Swinepox
- Cowpox
- Horsepox
- Camelpox
- Buffalopox
- GENERAL INTRODUCTION: PICORNAVIRIDAE
- Teschen, Talfan and reproductive diseases caused by porcine enteroviruses
- Encephalomyocarditis virus infection
- Swine vesicular disease
- Equine picornavirus infection
- Bovine rhinovirus infection
- Foot-and-mouth disease
- GENERAL INTRODUCTION: BORNAVIRIDAE
- Borna disease
- GENERAL INTRODUCTION: CIRCOVIRIDAE AND ANELLOVIRIDAE
- Post-weaning multi-systemic wasting syndrome in swine
- GENERAL INTRODUCTION: PRION DISEASES
- Scrapie
- Bovine spongiform encephalopathy
- Transmissible spongiform encephalopathies related to bovine spongiform encephalopathy in other domestic and captive wild species
Borna disease
This content is distributed under the following licence: Attribution-NonCommercial CC BY-NC  View Creative Commons Licence details here
View Creative Commons Licence details here

Borna disease
R ROTT, S HERZOG AND J A RICHT
Introduction
Borna disease (BD) is a sporadically occurring, progressive viral polioencephalomyelitis predominantly affecting horses and sheep, more rarely other Equidae, cattle, goats, cats, dogs and rabbits, and, exceptionally, a variety of other species. After an incubation period of several weeks to many months locomotor and sensory dysfunctions occur followed by paralysis and death. Natural infections seem to be subclinical in most cases.11, 43 The causative agent, Borna disease virus (BDV), has recently been classified in a new family Bornaviridae.27
In Germany, where BD is endemic, it has been known for more than 250 years. The commonly used name ‘Bornasche Krankheit’ originated from the frequent outbreaks of the disease in the district of Borna in Saxony between 1894 and 1896. Borna disease has since been described in Switzerland in 1979,24 Japan, in 199710 and Austria in 199842 for the first time. It is uncertain whether clinically similar cases of equine encephalitides in France, Romania, Libya, and the Near East were in fact BD. The disease has not been diagnosed in southern Africa, but there is reason to believe that it is more widely distributed since virus-specific antibodies have been demonstrated in horses from several continents. 14
Detailed studies of the pathology of BD were conducted during the first decade of the twentieth century.17 In the late 1920s Zwick and co-workers44 established its viral aetiology by reproducing the disease with bacteria-free filtrates. More recently, a soluble, virus-specific intranuclear antigen has been shown to exist in brain material of infected animals.9, 37, 40 The causative virus has been propagated in cell cultures, 13, 23 its molecular structure characterized and its replication strategies determined,6, 35 while aspects of the pathogenesis of the disease have been elucidated.26, 38
Aetiology
Borna disease virus has been characterized as a spherical enveloped virus particle with a diameter of about 100 to 130 nm.19The viral genome consists of a non-segmented single-stranded, 8,9 kb large RNA of negative polarity with complementary 3’ and 5’ termini. The genomic organization with six major open reading frames (ORFs) is similar to that of other members of the order Mononegavirales. Therefore, BDV is classified by the international nomenclature commission of International Committee for the Taxonomy of Viruses (ICTV) as a prototype of the new family Bornaviridae containing only one genus, Bornavirus.27 Expression of the identified ORFs permitted identification and characterization of five BDV-specific proteins. The first open reading frame (ORF1) encodes a protein with a molecular weight of 38/39 kDa (p38, also known as p40), the putative nucleoprotein (N). The second open reading fram (ORF2) codes for a phosphorylated 24 kd protein (p24, also called p23), representing the putative phosphoprotein (P). The products of ORF1 and ORF2 (N and P) form a heterodimer as shown by coprecipitation with monoclonal antibodies and hybrid interaction studies. Recently, an additional open reading frame (ORFX1) has been identified which overlaps with ORF2, and encodes a non-glycosylated BDV protein designated p10 (also called X). The third open reading frame (ORF3) encodes a 16 kDa protein (p16) which is post-translationally glycosylated, resulting in a molecular weight of 18 kDa (gp18), which represents the putative membrane protein (M). The fourth open reading frame (ORF4) encodes a 57 kd protein (p57) which is highly N-glycosylated resulting in a molecular weight of 84 or 94 kDa (gp84 and gp94), representing the putative precursor glycoprotein (GP) of the virus; cleavage of this GP by the subtilisin-like cellular protease furin results in two fragments with molecular masses of approximately 48 kDa and 51 kDa.28 Finally, the gene product of ORF5 which is localized at the 3’ end of the cRNA has been identified very recently as a 190 kDa protein.41 Sequence homology with RNA-dependent RNA polymerases of other members of the order Mononegavirales or its nuclear localization in infected cells and its interaction with the RNA phosphoprotein indicate that ORF5 encodes for the BDV polymerase.41
Antigenic variants have been found using monoclonal antibodies.14, 30 Neutralizing antibodies are demonstrable, if at all, only in the late phase of infection,35, 39 and antigenic variants within one individual animal have not yet been observed.
Embryonic brain cells of various provenance can be infected with BDV and co-cultivation of infected brain cells with permanent cell lines of diverse, non-central nervous system (CNS) origin may result in persistent infection.13Virus produced in such cultures is cell-associated, spreads by cell-to-cell contact without the production of cytopathic effects. Borna disease virus obtained from persistently infected cells (e.g. BDV/MDCK cells) is capable of infecting ‘uninfected cultures’ of the same cell line without further co-cultivation.13 Infectious virus particles, virus-like structures and virus-specific proteins can be detected in such persistently infected MDCK cells.4, 9, 19, 30 Borna disease virus- specific proteins can be demonstrated in the nuclei or cytoplasm of infected cells by immunofluorescence with antibodies obtained from infected animals.13
Little is known about the consecutive steps involved in virus replication. There is some evidence that the major glycoprotein BDV-GP (gp94) mediates penetration by membrane fusion after the fusion peptide becomes exposed by proteolytic cleavage by subtilisin-like cellular proteases.7, 28 In contrast to other nonsegmented RNA viruses, the BDV genome is replicated and transcribed in the nucleus of the infected cell. The cellular splicing machinery is required to process some of the primary BDV RNA transcripts. A variety of mechanisms are used to regulate gene expression, including the use of overlapping reading frames, overlapping transcription units, alternate RNA splicing, and leaky scanning of ribosomes during protein translation.6, 35 The replication cycle seems to be completed within 24 hours. Only small amounts, if any, of infectious virus are released from infected cells.
Epidemiology
The large spectrum of animals and types of cell cultures that can be infected experimentally with BDV suggests that natural infections occur more frequently and in a wider range of animal species than has previously been assumed. In addition to equids and sheep, natural infections have also been reported in cattle, goats, various species of captive wild animals, cats, dogs and rabbits.33 Recently, BDV-specific antibodies have been demonstrated in serum and cerebrospinal fluid from human patients suffering from neurological and psychiatric disorders in Germany, Japan, and the USA.2, 30, 34
Many species of animals, ranging from chickens to rhesus monkeys, can be infected experimentally by intracerebral or intranasal inoculation of brain homogenates or cerebrospinal fluid from diseased animals.11, 43 Rabbits are the most sensitive laboratory species. The manifestation of the experimental disease differs between species and is dependent on age, immune status, and the virus strain used. The disease may not be apparent (hamster, adult mouse), or it may produce predominantly alterations in the behaviour of the animal (tree shrew, rat), or cause obesity (rat), disturbances in fertility (rat), blindness or severe neurological signs with paralysis followed by death (rat, horse).33, 39
Borna disease was originally described as a disease restricted to certain endemic areas in eastern and southern Germany and recently also in the eastern part of Switzerland. Recent seroepidemiological studies, conducted mainly in horses, however, have shown that natural infection is much more widely distributed than previously realized. These studies have revealed the presence of seropositive horses not only throughout the entire Germany, but also in other European countries, such as the Netherlands, France and Poland, North America, Iran and Japan;1, 10, 14, 18 this strongly supports the notion that BD might have a worldwide distribution. The discrepancy between the number of infected horses and those that show clinical signs may be due to the fact that infection with BDV is largely subclinical in horses.11, 14 Detailed studies on the epidemiology of BD in other animal species are not available.
The mode of transmission is unknown, but the presence of virus in nasal secretions, saliva and urine suggests that dissemination occurs either by direct contact or through contaminated fomites and food. Vertical transmission has not been convincingly demonstrated. No free-living animal reservoirs of the virus have been identified yet, although it is suspected that rats or other rodents might be virus carriers. The high prevalence of subclinically infected horses, which also shed virus in various secretions,29 is indicative that they represent a major virus reservoir. Arthropod vectors have not been convincingly incriminated.11
Pathogenesis
The understanding of the pathogenesis of BD is based mainly on studies of experimentally infected rats. In natural infections, the virus apparently enters the body through the olfactory nerve endings from where it migrates intraaxonally via the olfactory bulb to other parts of the brain.3, 25 Viral replication in adult animals takes place exclusively in cells of the nervous system. The virus migrates along axons and disseminates from the CNS to the peripheral nervous system. In contrast to adult rats, viral antigen and infectious virus can be found in many organs of infected new-born rats and, although these animals do not manifest clinical signs, they shed the virus persistently.15, 25, 39 Antibody titres in naturally infected animals are relatively low and are found in sera and cerebrospinal fluids.14
The mechanism of cell damage produced by BDV has not been determined. However, since experimental infection of immunocompetent Lewis rats invariably results in clinical signs whereas immunoincompetent Lewis rats do not show pathological alterations despite high levels of virus in the CNS, it is probable that BD is the result of an immunopathological reaction.16, 26
Although antibody transfer to immunosuppressed animals does not initiate the development of the disease, adoptive transfer of immune cells from the spleen or from lymph nodes derived from infected animals has been shown to be effective in the induction of encephalitis and clinical manifestation in immunosuppressed virus carriers.26, 39 There is strong evidence that BDV-specific Thelper/ inducer (CD4+) cells play a dominant role in the immunopathology of BD.32 It therefore seems that the pathogenesis is based on a delayed hypersensitivity reaction. Remarkably, when the same type of BDV-specific CD4+ T cells were passively transferred into naive rats before virus infection, animals were protected against clinical BD after challenge.31 Thus, in BDV infections of the CNS the same virus- specific immune cells that cause disease can also prevent disease depending on the dynamics of virus replication and the time and intensity of antiviral T-cell response.
Clinical signs
Clinical manifestations may vary among individual animals and various animal species. They do not always occur in the same sequence and not all signs are manifested in milder cases of BD.
Typical cases of BD show clinical signs as a result of the meningoencephalitis; they become apparent after a long incubation period of several weeks to months. Frequently, an elevated body temperature, moderate icterus, digestive disturbances (colic) and general uneasiness accompany the nervous signs. Variable, and often unusual, clinical signs such as reduced appetite, difficulty in swallowing, a fluctuating body temperature, and disturbed sensory functions, such as decreased reactions to stimulation of the skin, may, however, occur during the early stage of the disease.11, 43 As the disease progresses, animals show hyperaesthesia, incoordinated movements, unnatural body positions (Figure126.1), periods of excitement or depression, and ataxia. Epilepsy and amaurosis often occur. Increasing paresis and paralysis develop during the final stages of the disease with death usually occurring one to three weeks after the onset of clinical signs. The mortality rate varies from 37 to 94 per cent. In surviving animals recurrent episodes may occur, especially after stressful situations.11, 43
In sheep, disturbances of sensory functions are more pronounced than neurological signs such as paralysis, which develops preferentially in horses. In addition, ocular disorders and delayed pupillary reflexes are frequently observed.
Pathology
There are no gross lesions in the brain. The histopathology of BD is characterized by a non-purulent polioencephalomyelitis comprising perivascular and parenchymal infiltrations of mononuclear cells.17, 36The lesions are mostly located in the olfactory bulbs, basal cortex, nucleus caudatus, and hippocampus, which implies that the limbic system is primarily involved. Degeneration of nerve cells and neuronophagia are not prominent, but there is a distinct astrocytosis. There is no evidence of demyelination. In neurons and, less frequently, in glial cells, eosinophilic intranuclear and, less numerous, intracytoplasmic inclusion bodies (known as ‘Joest-Degen bodies’) may occur. Inflammatory changes are also present in the periventricular areas of the diencephalon and the midbrain and, to a lesser extent, in the medulla oblongata. The cerebellum, spinal cord, spinal nerves and the retina are in general rarely involved, while the peripheral nervous system is unaffected.8 A characteristic of experimentally infected rats and rabbits are histopathological lesions in the eye which manifest as a loss of rods and cones and ultimately in a progressive disappearance of neurons from the inner and outer nuclear layers. The complete loss of these neurons is the reason for the blindness occurring in experimentally infected rats and rabbits.20, 26
Diagnosis and differential diagnosis
The clinical signs and the histopathological lesions with or without the presence of intranuclear Joest-Degen inclusion bodies, assist in the making of a provisional diagnosis of BD. The diagnosis can only be confirmed by serological and/or virological methods. Borna disease virus-specific antibodies are present in the serum and cerebrospinal fluid of diseased animals12, 14, 21, 22 and can be detected by Western blot analysis, enzyme-linked immunosorbent assay (ELISA), and indirect immunofluorescence using persistently infected cell cultures.33 Monoclonal antibodies are available for the demonstration of viral antigen in formalin-fixed paraffin embedded brain sections or in brain homogenates by Western blot analysis (Figure126.2). With the advent of viral sequence information, new diagnostic techniques for BDV infections have been introduced, such as in situ hybridization and the RT-polymerase chain reaction.29 Infectious virus can also be isolated from homogenates of infected brain or from cerebrospinal fluid by inoculation onto susceptible cell cultures derived from embryonic rabbit or rat brain, or by the intracerebral inoculation of rabbits. The latter is the most sensitive method for the detection of small amounts of infectious virus; in positive cases rabbits become ill within three to four weeks after inoculation.
Neurological signs of BD in horses should be distinguished from those resulting from rabies, equid herpesvirus 1 and 4 infections, Venezuelan, Eastern and Western equine encephalomyelitis virus infections, and equine protozoal myeloencephalitis as well as equine leukoencephalomalacia caused by the ingestion of maize contaminated by Fusarium moniliforme. The extended incubation period of BD distinguishes it from other forms of viral encephalitides in horses.
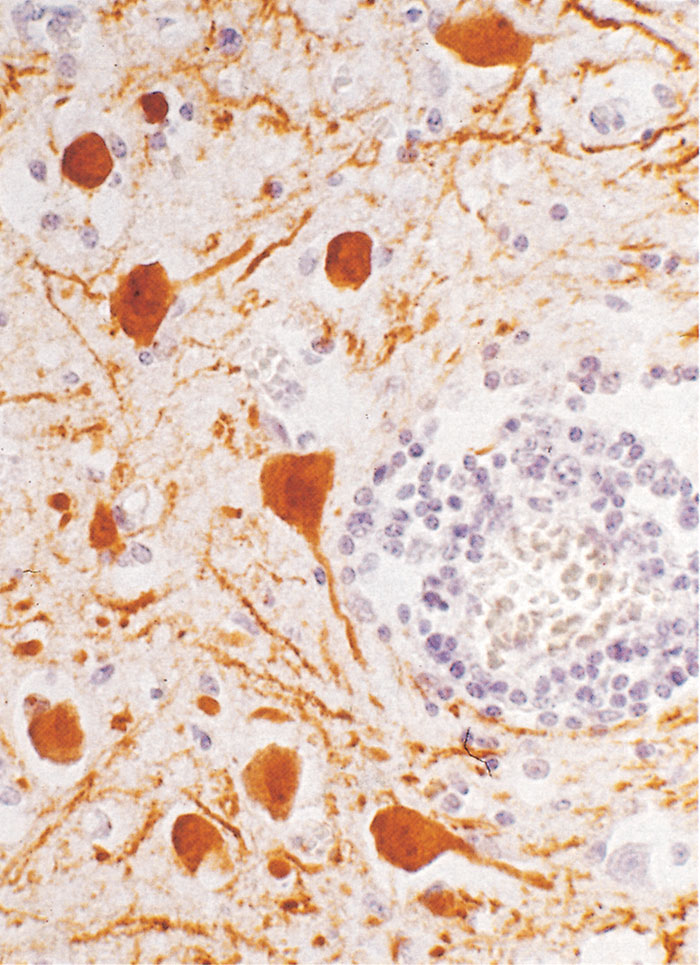
Figure 126.2 Section of hippocampus of a horse with spontaneous Borna disease. Note the monunuclear cells in the perivascular area and viral antigen in neurons using monoclonal antibodies against the viral nucleoprotein (PAP method, using monoclonal antibodies against a 38/39 virus-specific antigen). (By courtesy of Dr Frese)
Control
There is no rational basis for therapy and no satisfactory vaccine exists. A protective immunity can only be induced experimentally by inoculating animals with live virus,5 but such a practice poses the unacceptable risk of viral dissemination. The control of the disease, therefore, must focus on the identification of potential carrier animals which are responsible for the spread of the virus in a given population. After their identification they should be eliminated. In Germany BD is a notifiable disease.
Data from studies in humans with neuropsychiatric disorders have provided an increasing body of evidence that BDV is capable of infecting humans and that it may be a human pathogen. It is not yet known how humans become infected as no convincing correlation between human patients and contact with infected individual animals or herds has been found. The potential role of BDV as a human pathogen and therefore its role as a zoonotic agent requires further substantiation.2, 30
References
- bahmani, m.k., nowrouzian, i., nakaya, t., nakamura, y., hagiwara, k., takahashi, h., rad, m.a. & ikuda, k., 1996. Varied prevalence of Borna disease virus infection in Arabic, thoroughbred and cross-bred horses in Iran. Virus Research, 45, 1–3.
- bechter, k (ed.), 1998. Borna Disease Virus: Mögliche Ursache neurologischer und psychiatrischer Störungen des Menschen. Darmstadt: Steinkopf.
- carbone, k., duchala, c.s., griffin, j.w., kincaid, h.l. & narayan, o., 1987. Pathogenesis of Borna disease in rats: Evidence that intra-axonal spread is the major route of virus dissemination and for disease incubation. Journal of Virology, 61, 3431–3440.
- compans, r.w., melsen, l.r. & de la torre, j.c., 1994. Virus-like particles in MDCK cells persistently infected with Borna disease virus. Virus Research, 33, 261–268.
- danner, k., (ed.), 1982. Borna-Virus und Borna Infektion: Vom Miasma zum Modell. Stuttgart: Ferdinand Enke Verlag.
- de la torre, j.c., 1994. Molecular biology of Borna disease virus: Prototype of a new group of animal viruses. Journal of Virology, 68, 7669–7675.
- gonzalez-dunia, d., cubitt, b. & de la torre, j.c., 1998. Mechanism of Borna disease virus entry into cells. Journal of Virology, 72, 783–788.
- gosztonyi, g. & ludwig, h., 1984. Neurotransmitter receptors and viral neurotropism. Neuropsychiatria Clinical, 3, 107–114.
- haas, b., becht, h. & rott, r., 1986. Purification and properties of an intranuclear virus-specific antigen from tissue infected with Borna disease virus. Journal of General Virology, 67, 235–241.
- hagiwara, k., momiyama, n., taniyama, h., nakaya, t., tsunda, n., ishihara, c. & ikuta, k., 1997. Demonstration of Borna disease virus (BDV) in specific regions of the brain from horses positive for serum antibodies to BDV but negative for BDV RNA in the blood and internal organs. Medical Microbiology and Immunology, 86, 19–24.
- heinig, h., 1969. Die Borna’sche Krankheit der Pferde und Schafe. In: röhrer, h., (ed.). Handbuch der Virusinfektionen bei Tieren, Vol. IV. Jena: VEB Gustav Fischer Verlag.
- herden, c., herzog, s., wehner, t., richt, j.a. & frese, k., 1996. Postmortem diagnosis of Borna disease in horses: Comparison of different methods. European Journal of Veterinary Pathology (Supplement), 2, 9–10.
- herzog, s. & rott, r., 1980. Replication of Borna disease virus in cell cultures. Medical Microbiology and Immunology, 168, 153–158.
- herzog, s., frese, k., richt, j.a & rott, r., 1994. Ein Beitrag zur Epizootiologie der Bornaschen Krankheit beim Pferd. Wiener Tierärztliche Monatsschrift, 81, 374–379.
- herzog, s., kompter, s.c., frese, k. & rott, r., 1984. Replication of Borna disease virus in rats: Age-dependent differences in tissue distribution. Medical Microbiology and Immunology, 173, 171–177.
- herzog, s., wonigeit, k., frese, k., hedrich, h.j. & rott, r., 1985. Effect of Borna disease virus infection on athymic rats. Journal of General Virology, 66, 503–508.
- joest, e. & degen, h., 1911. Untersuchungen u¨ber die pathologische Histologie, Pathogenese und postmortale Diagnose der seuchenhaften Gehirnru¨cken-Marksentzu¨ndung (Borna’sche Krankheit) der Pferde. Zeitschrift fu¨r Infektions-krankheiten, Parasitäre Krankheiten und Hygiene der Haustiere, 9, 1–98.
- kao, m., hamir, a.n., rupprecht, c.e., fu, z.f., shankar, v., koprowski, h. & dietzschold, b., 1993. Detection of antibodies against Borna disease virus in sera and cerebrospinal fluid of horses in the USA. The Veterinary Record, 132, 241–244.
- kohno, t., goto, t., takasaki, t., morita, c., nakaya, t., ikuta, k., kurane, i., sano, k. & nakai, m., 1999. Fine structure and morphogenesis of Borna disease virus. Journal of Virology, 73, 760–766.
- krey, h., ludwig, h. & rott, r., 1979. Spread of infectious virus along the optic nerve into the retina in Borna disease virus-infected rabbits. Archives of Virology, 61, 283–288.
- ludwig, h., koester, u., pauli, g. & rott, r., 1977. The cerebrospinal fluid of rabbits infected with Borna disease virus. Archives of Virology, 55, 209–223.
- ludwig, h. & thein, p., 1977. Demonstration of specific antibodies in the central nervous system of horses naturally infected with Borna disease virus. Medical Microbiology and Immunology, 163, 215–226.
- mayr, a. & danner, k., 1972. In vitro-Kultivierung von Bornavirus u¨ber Gehirn-Explantate infizierter Tiere. Zentralblatt fu¨r Veterinärmedizin, B19, 785–800.
- metzler, a., minder, h.-p., wegemann, ch. & zindel, w., 1979. Die Borna’sche Krankheit, ein veterinärmedizinisches Problem von regionaler Bedeutung. Schweizer Archiv fu¨r Tierheilkunde, 121, 207–213.
- morales, j.a., herzog, s., kompter, c., frese, k. & rott, r., 1988. Axonal transport of Borna disease virus along olfactory pathways in spontaneously and experimentally infected rats. Medical Microbiology and Immunology, 177, 51–68.
- narayan, o., herzog, s., frese, k., scheefers, h. & rott, r., 1983. Pathogenesis of Borna disease in rats: Immune-mediated viral ophthalmoencephalopathy causing blindness and behavioural abnormalities. Journal of Infectious Diseases, 148, 305–315.
- regenmortel, v.m.h., fauquet, c.m., bislan, d.h.c. et al., 2000. Virus Taxonomy, Seventh Report of the ICTV. San Diego: Academic Press.
- richt, j.a., fu¨rbringer, t., koch, a., pfeuffer, i., herden, c., bause-niedrig, i. & garten, w., 1998. Processing of the Borna disease virus glycoprotein gp 94 by the subtilisin-like endoprotease furin. Journal of Virology, 72, 4528–4533.
- richt, j.a., herzog, s., haberzettl, k. & rott, r., 1993. Demonstration of Borna disease virus-specific RNA in secretions of naturally infected horses by the polymerase chain reaction. Medical Microbiology and Immunology, 182, 293–304.
- richt, j.a., pfeuffer; i., christ, m., frese, k., bechter, k. & herzog, s., 1997. Borna disease virus infection in animals and man. Emerging Infectious Diseases, 3, 343–352.
- richt, j.a., schmeel, a., frese, k., carbone, k.m., narayan, o. & rott, r., 1994. Borna disease virus-specific T cells protect against or cause immunopathological Borna disease. Journal of Experimental Medicine, 179, 1467–1473.
- richt, j.a., stitz, l., wekerle, h. & rott, r., 1988. Borna disease, a progressive meningoencephalitis as a model for CD4+ T cell-mediated immunopathology in the brain. Journal of Experimental Medicine, 170, 1045–1050.
- rott, r. & becht, h., 1995. Natural and experimental Borna disease in animals. Current Topics in Microbiology and Immunology, 190, 17–30.
- rott, r., herzog, s., fleischer, b., winokur, a., amsterdam, j., dyson, w. & koprowski, h., 1985. Detection of serum antibodies to Borna disease virus in patients with psychiatric disorders. Science, 228, 755–756. 1
- schneemann, a., schneider, p.a., lamb, r.a. & lipkin, w.i., 1995. The remarkable coding strategy of Borna disease virus: A new member of the nonsegmented negative RNA virus. Virology, 210, 1–8.
- seifried, o. & spatz, h., 1930. Die Ausbreitung der encephalitischen Reaktion bei der Bornaschen Krankheit der Pferde und deren Beziehungen zur Encephalitis Epidemica, der Heine-Medinschen Krankheit und der Lyssa des Menschen. Eine vergleichende pathologische Studie. Zeitschrift fu¨r Neurologie, 124, 317–382.
- sprockhoff, h.v. & nitzschke, e., 1955. Untersuchungen u¨ber das komplementbindende Antigen in Gehirnen Bornavirus-infizierter Kaninchen. 1. Nachweis eines löslichen Antigens. Zentralblatt fu¨r Veterinärmedizin, 2, 185–192.
- stitz, l. & rott, r., 1999. Borna disease virus (Borna viridae). In: webster, r.g. & granoff, f., (eds). Encylopedia in Virology, Vol. I. London: Academic Press.
- stitz, l., dietzschold, b. & carbone, k.m., 1995. Immunopathogenesis of Borna disease. Current Topics in Microbiology and Immunology, 190, 75–92.
- wagner, k., ludwig, h. & paulsen, j., 1968. Fluoreszenzserologischer Nachweis von Borna-Virus Antigen. Berliner und Mu¨nchener Tierärztliche Wochenschrift, 81, 395–396.
- walker, m.p., jordan, i., briese, t., fischer, n. & lipkin, w., 2000. Expression and characterization of the Borna disease virus polymerase. Journal of Virology, 74, 4425–4428.
- weissenbock, h., suchy, a., caplazi, p., herzog, s. & nowotny, n., 1998. Borna disease in Austrian horses. The Veterinary Record, 143, 21–22.
- zwick, w., 1939. Borna’sche Krankheit und Encephalomyelitis der Tiere. In: gildemeister, e., haagen, e. & waldmann, o., (eds). Handbuch der Viruskrankheiten. Vol. II, Jena: Gustav Fischer Verlag.
- zwick, w., seifried, o. & witte, j., 1927. Experimentelle Untersuchungen u¨ber die Gehirnru¨ckenmarksentzu¨ndung der Pferde (Borna’sche Krankheit). Zeitschrift fu¨r Infektionskrankheiten, Parasitäre Krankheiten und Hygiene der Haustiere, 30, 42–136.





