- Infectious Diseases of Livestock
- Part 2
- Bovine coronavirus infection
- GENERAL INTRODUCTION: PARAMYXOVIRIDAE AND PNEUMOVIRIDAE
- Rinderpest
- Peste des petits ruminants
- Parainfluenza type 3 infection
- Bovine respiratory syncytial virus infection
- Hendra virus infection
- Paramyxovirus-induced reproductive failure and congenital defects in pigs
- Nipah virus disease
- GENERAL INTRODUCTION: CALICIVIRIDAE AND ASTROVIRIDAE
- Vesicular exanthema
- Enteric caliciviruses of pigs and cattle
- GENERAL INTRODUCTION: RETROVIRIDAE
- Enzootic bovine leukosis
- Jaagsiekte
- Visna-maedi
- Caprine arthritis-encephalitis
- Equine infectious anaemia
- GENERAL INTRODUCTION: PAPILLOMAVIRIDAE
- Papillomavirus infection of ruminants
- Papillomavirus infection of equids
- GENERAL INTRODUCTION: ORTHOMYXOVIRIDAE
- Equine influenza
- Swine influenza
- GENERAL INTRODUCTION: CORONAVIRIDAE
- Porcine transmissible gastroenteritis
- Porcine respiratory coronavirus infection
- Porcine epidemic diarrhoea
- Porcine haemagglutinating encephalomyelitis virus infection
- Porcine deltacoronavirus infection
- Bovine coronavirus infection
- Ovine coronavirus infection
- Equine coronavirus infection
- GENERAL INTRODUCTION: PARVOVIRIDAE
- Porcine parvovirus infection
- Bovine parvovirus infection
- GENERAL INTRODUCTION: ADENOVIRIDAE
- Adenovirus infections
- GENERAL INTRODUCTION: HERPESVIRIDAE
- Equid herpesvirus 1 and equid herpesvirus 4 infections
- Equid gammaherpesvirus 2 and equid gammaherpesvirus 5 infections
- Equine coital exanthema
- Infectious bovine rhinotracheitis/infectious pustular vulvovaginitis and infectious pustular balanoposthitis
- Bovine alphaherpesvirus 2 infections
- Malignant catarrhal fever
- Pseudorabies
- Suid herpesvirus 2 infection
- GENERAL INTRODUCTION: ARTERIVIRIDAE
- Equine viral arteritis
- Porcine reproductive and respiratory syndrome
- GENERAL INTRODUCTION: FLAVIVIRIDAE
- Bovine viral diarrhoea and mucosal disease
- Border disease
- Hog cholera
- Wesselsbron disease
- Louping ill
- West nile virus infection
- GENERAL INTRODUCTION: TOGAVIRIDAE
- Equine encephalitides caused by alphaviruses in the Western Hemisphere
- Old World alphavirus infections in animals
- Getah virus infection
- GENERAL INTRODUCTION: BUNYAVIRIDAE
- Diseases caused by Akabane and related Simbu-group viruses
- Rift Valley fever
- Nairobi sheep disease
- Crimean-Congo haemorrhagic fever
- GENERAL INTRODUCTION: ASFARVIRIDAE
- African swine fever
- GENERAL INTRODUCTION: RHABDOVIRIDAE
- Rabies
- Bovine ephemeral fever
- Vesicular stomatitis and other vesiculovirus infections
- GENERAL INTRODUCTION: REOVIRIDAE
- Bluetongue
- Ibaraki disease in cattle
- Epizootic haemorrhagic disease
- African horse sickness
- Equine encephalosis
- Palyam serogroup orbivirus infections
- Rotavirus infections
- GENERAL INTRODUCTION: POXVIRIDAE
- Lumpy skin disease
- Sheeppox and goatpox
- Orf
- Ulcerative dermatosis
- Bovine papular stomatitis
- Pseudocowpox
- Swinepox
- Cowpox
- Horsepox
- Camelpox
- Buffalopox
- GENERAL INTRODUCTION: PICORNAVIRIDAE
- Teschen, Talfan and reproductive diseases caused by porcine enteroviruses
- Encephalomyocarditis virus infection
- Swine vesicular disease
- Equine picornavirus infection
- Bovine rhinovirus infection
- Foot-and-mouth disease
- GENERAL INTRODUCTION: BORNAVIRIDAE
- Borna disease
- GENERAL INTRODUCTION: CIRCOVIRIDAE AND ANELLOVIRIDAE
- Post-weaning multi-systemic wasting syndrome in swine
- GENERAL INTRODUCTION: PRION DISEASES
- Scrapie
- Bovine spongiform encephalopathy
- Transmissible spongiform encephalopathies related to bovine spongiform encephalopathy in other domestic and captive wild species
Bovine coronavirus infection
This content is distributed under the following licence: Attribution-NonCommercial CC BY-NC  View Creative Commons Licence details here
View Creative Commons Licence details here
Bovine coronavirus infection
Previous Authors: L J SAIF
Current Authors:
L J SAIF - Professor, MS, PhD, Food Animal Health Research Program, CFAES and CVM, OARDC, Ohio State University, 1680 Madison Ave, Wooster, Ohio, OH 44691, USA
M A ALHAMO - Research, DVM, MSc, OARDC Food Animal Health Research Program, Ohio State University, 1680 Madison Avenue, Wooster, Ohio, OH 44691, USA
Introduction
Bovine coronavirus (BCV) was first recognized in 1972 in the USA as a cause of neonatal calf diarrhoea.47 Since then, it has been implicated in winter dysentery in adult dairy cattle 2, 17, 57, 61, 62, 70, 73, 74 and in respiratory infections of cattle of various ages.11, 28, 34, 39, 41, 45, 52, 55, 56, 64, 65, 68, 79 In addition, coronaviruses (CoVs) antigenically indistinguishable from BCV have been isolated from wild ruminants,44, 71 including cases of bloody diarrhoea resembling winter dysentery of cattle.71 Bovine coronavirus enteric or respiratory infections are found in cattle worldwide,4, 8, 52, 53, 56, 58, 79 leading to major health and economic losses to beef and dairy farms. Among other domestic ruminants, BCVs have also been isolated from water buffaloes.14, 49 Studies of wild white-tailed deer (Odocoileus virginianus), mule deer (Odocoileus hemionus),71 and caribou (Rangifer tarandus caribou),18 in North America have indicated BCV antibody seroprevalence rates of 7 to 14 per cent, suggesting that BCV infections may be endemic in wild native ruminants.56, 71 Captive wild ruminants in the USA have been identified as a source of BCV infections, including sambar deer (Rusa unicolor), white-tailed deer, waterbuck (Kobus ellipsiprymnus), elk or wapiti (Cervus elephus), and giraffe (Giraffa camelopardalis).5, 26, 44, 71 In addition, betacoronaviruses (clade A, like BCVs), were detected from faeces or nasal swabs of camelids, namely alpacas (Vicugna pacos), llamas (Lama glama), and, in the United Arab Emirates (UAE), from dromedary camels (Camelus dromedarius).78 Analysis of a French calf BCV strain revealed 99.1 per cent nucleotide similarity in the orf1ab gene with the dromedary camel strain (HKU23–23-362F) from the UAE.35 To date, the coronaviruses isolated and characterized from both enteric and respiratory infections of cattle and enteric infections of wild ruminants are genetically, 1 biologically and antigenically similar and comprise a single serotype of BCV,4, 13, 16, 28, 52, 56, 71, 72 although two to three subtypes or subclades of BCV have been identified.21, 28, 72 However, experimental inoculation of calves with these different BCV strains cross-protected against disease, but not virus shedding.17
Aetiology
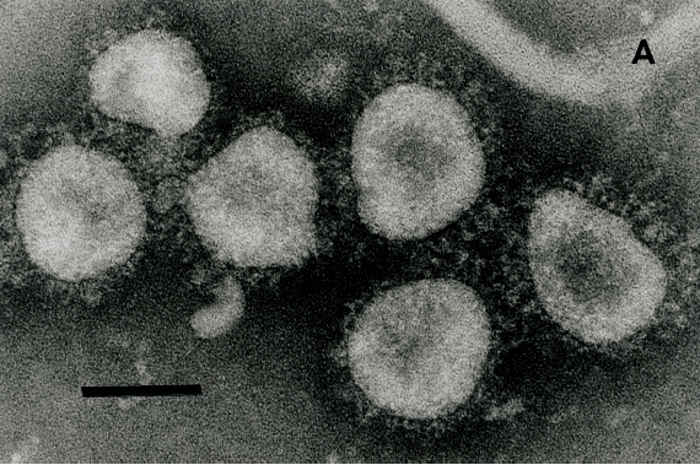
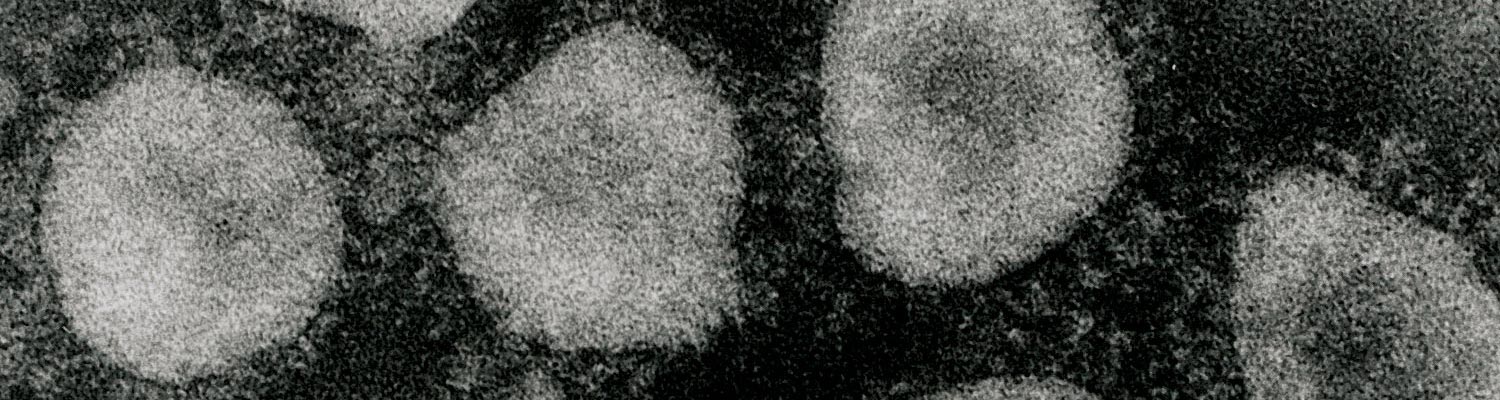
Bovine coronavirus (BCV) belongs to the Betacoronavirus genus (clade A) of the family Coronaviridae, in the order Nidovirales.33, 36, 78 Bovine coronavirus is an enveloped, pleomorphic virus ranging in diameter from 65 to 210 nm, with a double layer of surface projections, the shorter projections (~11 nm) consisting of the haemagglutinin and the longer projections (~25 nm) comprising the spike (S) glycoprotein.8, 56, 58 (Figure 1) The BCV contains a single-stranded, positive sense RNA genome of 27 to 32 kb. The virus is composed of four major structural proteins, three of which are glycosylated, whereas the fourth, the nucleocapsid (N) protein (50 kDa) that surrounds the viral RNA, is not. The glycoproteins include the 25 kDa integral membrane (M) protein, the 190 kDa S protein, which is cleaved into 90 and 100 kDa subunits (S1 and S2), and the haemagglutinin esterase (HE), which is a disulphide-linked dimer of 120 to 140 kDa.8, 58 These last two proteins are responsible for the haemagglutination (HA) of BCV with chicken, mouse, rat and hamster erythrocytes.28, 29 The HA protein also acts as a receptor-destroying enzyme (RDE), serving as an esterase to reverse haemagglutination. 8, 58 Although some variability exists among BCV strains in patterns of HA or RDE activity with erythrocytes from different species, these differences are unrelated to the clinical origin (enteric versus respiratory) of the BCV strains.28, 29, 72
Both the S and HA proteins are involved in viral attachment to host cells and induce the formation of neutralizing antibodies to BCV.8, 29, 58 Monoclonal antibodies (MAbs) to the S or HA proteins of BCV prevent villous atrophy in vivo in intestinal loops of calves,12 suggesting that either or both proteins are important for inducing immunity to BCV.58
As in other enveloped viruses, BCV is sensitive to detergents and lipid solvents such as ether and chloroform.8, 56, 58 It is also inactivated by formalin (0,02 per cent at 37 °C for 24 hours) and heating (50 °C for 1 hour), but is relatively stable at pH 3,0.
To date only one serotype of BCV is recognized.4, 8, 56, 58 Some antigenic variability exists among BCV isolates as demonstrated using two-way virus neutralization (VN) or haemagglutination inhibition (HI) assays with polyclonal antibodies or MAbs to BCV,13, 16, 28, 29, 72 and two to three subtypes of BCV have been described.28, 72 This variability, however, is unrelated to the age of the infected animal (calf diarrhoea versus winter dysentery in adults) or to the clinical source of the isolate (enteric versus respiratory).28, 72 The BCV is antigenically and genetically related to the betacoronaviruses, clade A, which include murine hepatitis virus, human coronavirus OC43, porcine haemagglutinating encephalomyelitis virus (PHEV), canine respiratory coronavirus (CRCoV) and a strain of turkey coronavirus.3, 35, 36, 58 In addition, a high nucleotide identity (96 per cent) was demonstrated between BCV and human coronavirus OC43 (HCoVC43), suggesting that HCoV-OC43 may have originated by zoonotic transmission of BCV to humans.36, 75 A high nucleotide identity was also detected between BCV and CRCoV.3
Field strains of BCV usually replicate poorly or are difficult to isolate in cell culture. Primary isolation of BCV has, however, been accomplished using primary calf kidney cells, cell lines including Vero, Madin-Darby bovine kidney (MDBK), and a human rectal-adenocarcinoma cell line (HRT-18).29, 58, 59, 64 The HRT-18 cell line has been the most reliable for primary isolation of BCV from the faeces of adult cases of winter dysentery,2, 72 faeces of wild ruminants 44, 71 and nasal swabs of cases of respiratory disease.11, 25, 28, 64, 65 Once adapted to cell culture, strains such as the original calf diarrhoea isolate (Mebus NCDV BCV) grow in a variety of primary and continuous bovine cell types and also in HRT-18 cells.28, 29, 58, 59, 64 The BCV produces cytopathic effects in cell culture consisting of rounded, detached cells and syncytia.2, 28, 29, 58, 59, 64, 71, 72 The addition of pancreatin or trypsin to the medium often enhances virus replication and plaque and syncytia formation, perhaps through proteolytic cleavage of the S protein.2, 28, 29, 58, 59, 64, 71, 72
Epidemiology
Calf diarrhoea
Based on a variety of diagnostic assays, BCV enteric infections in calves have been found with prevalence rates of 8 to 69 per cent in diarrhoeic calves and 0 to 46 per cent in subclinically infected calves.8, 23 Enteric infections occur in both beef and dairy herds.6, 7, 8, 9, 56, 57, 58 Outbreaks of diarrhoea are more common in the autumn and the winter months, presumably because the virus thrives better at lower temperatures, and often occur yearly on the same farm.8, 30, 58 The virus is transmitted via the faecal–oral or respiratory routes.8, 58 Within a herd, reservoirs of infection may be clinically infected calves or subclinically infected calves or cows. Based on increased antibody titres, recurrent infections 30, 32 or intermittent faecal or nasal shedding episodes have been observed in longitudinal studies of four- to nine month-old dairy calves 30, 32 Bovine coronavirus antigens have been found in the faeces of normal adult dairy cattle, suggesting that adults could be a reservoir for the persistence and transmission of BCV in a herd.9 Furthermore, 89 per cent of dairy herds in Sweden were seropositive for BCV antibodies in a bulk milk survey.69
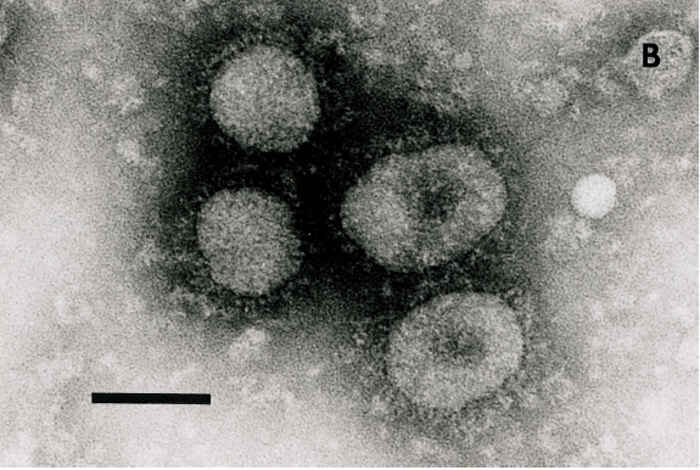
Figure 1 Electron micrograph of coronaviruses from the cell-culture passaged DBA strain of bovine coronavirus (BCV) (A) or waterbuck strain of wild ruminant coronavirus (B) incubated with hyperimmune antiserum to the Mebus calf strain of BCV. Bar = 100 nm
Winter dysentery
Winter dysentery occurs most commonly from November to March in the northern USA and it has also been reported in Europe, Australia and Asia.57 A dramatic agalactia with 25-95 per cent loss of milk production occurs in dairy herds during WD outbreaks,57 which last from a few days to several weeks according to herd size.6 The morbidity rate may range from 50 to 100 per cent.74 Van Kruiningen et al. 74 reported that lesions in cows with winter dysentery resembled those in BCV-infected neonatal calves.
During the past decade, evidence has accumulated implicating BCV as a cause of winter dysentery.2, 6, 57, 61, 62, 70, 72, 74 Epidemiological studies of winter dysentery have suggested that various host and environmental factors may contribute to the appearance of the disease.57, 61, 62, 73, 77 These include the age of the animal, with recently calved two- to six-year old cows being the most susceptible. Environmental risk factors include using manure-handling equipment to handle feed, close confinement (tie stall housing instead of free stall) and poor ventilation of barns.
Both in vitro and in vivo studies have indicated that winter dysentery and calf diarrhoea isolates of BCV are closely related. All winter dysentery and calf diarrhoea isolates tested in VN and HI tests cross-reacted, and the two subtypes of BCV observed were unrelated to the source (calf or adult cow diarrhoea) of the isolates.72 Similarly, cross-protective immunity between a calf diarrhoea and a winter dysentery BCV isolate was observed in gnotobiotic calves.17 A high degree of sequence conservation between winter dysentery and calf diarrhoea strains from Japan has been found.19 Thus, BCV strains from winter dysentery outbreaks are closely related biologically, antigenically and genetically to calf diarrhoea strains of BCV. Although BCV appears to be the major aetiological agent associated with winter dysentery, the host and environmental factors and interactions related to the manifestation of disease, especially the bloody diarrhoea often seen during outbreaks, have yet to be determined.
Calf respiratory infection
Bovine coronaviruses have also been implicated as a cause of respiratory infections in calves and young adult cattle. Field investigations of calfhood pneumonia (calves from two weeks to six months of age) in the UK and Austria resulted in the isolation of BCV from the lungs or nasal secretions of calves manifesting pneumonia.46, 66, 68 These respiratory BCV strains replicated in respiratory epithelial cell cultures including bovine tracheal organ cultures, nasal mucosal epithelial cell cultures and bovine embryonic lung cultures.46, 68
Additional studies documented that calves with diarrhoea in the field shed BCV both in faeces and in nasal secretions.30, 32, 55 A significant association was present between the simultaneous faecal and nasal shedding of BCV; most of the diarrhoeic calves necropsied had BCV antigen detected by immunofluorescence (IF) in both intestinal and respiratory tissues.55
In prospective longitudinal studies of calves from two closed dairy herds in the USA, it has been reported that both enteric and respiratory clinical BCV infections occurred in calves over one month of age, but that subclinical and recurrent infections were also common.30 There was a strong correlation between isotype antibody titres to BCV and the number of respiratory sick days. In another study by the same investigators of a large dairy herd (>800 head), BCV respiratory tract infections were also common, but most initial infections occurred at one to three weeks of age, often in conjunction with rhinitis.72 Recurrent respiratory infections with BCV were common.
As in field observations for conventional calves, experimental BCV challenge studies of gnotobiotic or colostrum-deprived calves established that both calf respiratory and enteric BCV isolates have pneumoenteric tropisms for both the intestinal and respiratory tracts as documented by faecal and nasal shedding detected by IF, immunoelectron microscopy (IEM) or ELISA.34, 46, 55, 60 Although signs of respiratory disease in calves in these studies were mild and variable, most calves inoculated with either the enteric or respiratory BCV isolates developed diarrhoea.46, 55, 60 The duration of nasal shedding of BCV averaged 5,3 days.55 Most calves necropsied had lesions and BCV antigens in the intestine, 34, 55 but lesions and BCV antigens in the lungs were less frequent (~20 to 30 per cent of calves examined) except in one study 34 in which two of three calves had lung lesions (interstitial pneumonia, congestion and haemorrhage) at death.
Respiratory infection and Bovine respiratory disease (BRD)
Bovine coronavirus is now established as a cause of respiratory infections in young adult cattle. As a component of the bovine respiratory disease complex (shipping fever) commonly seen in feedlot cattle in North America, it also contributes to respiratory disease and reduced growth performance.11, 22, 28, 39, 40, 41, 56, 64, 65, 67 Bovine coronavirus was isolated in HRT-18 cell cultures from nasal swabs from 38 per cent of cattle arriving at feedlots in Kansas and Arizona, and from 57 to 69 per cent of cattle with respiratory disease one day after arriving at feedlots in Texas.64, 65 In the Texas study, 25 of 26 cattle that died during the respiratory disease outbreak had BCV infections. In an Ohio feedlot,7 the overall proportion of cattle shedding BCV nasally, faecally or concurrently detected by ELISA was 46 per cent, 52 per cent and 44,6 per cent, respectively. In another study, application of nested RT-PCR detected higher respiratory BCoV nasal shedding rates of 84-97 per cent in feedlot calves.27, 79
Many investigators have shown an association between nasal shedding of BCV and respiratory disease.27, 39, 67 Feedlot calves shedding respiratory BCoV nasally were 1.5-2.7 times more likely to have respiratory disease than animals not shedding respiratory BCV.
A majority of cattle (58-95 per cent) seroresponded to BCV after 21-30 days in the feedlot.7, 39, 40, 41 Many investigators noted an association between higher BCV antibody titres of cattle entering feedlots and reduced aspects of BRD, including less likely to develop BRD, require treatment or show weight loss, shed BCV and develop increased BCV antibody titress.7, 40, 41, 45, 54, 67 Others reported that the mean antibody abundance (ELISA) in calves at weaning was inversely associated with the incidence of BRD after feedlot entry, but this was not the case when applied to individual calves.79 Where investigated, most studies have indicated that BCV causes respiratory and enteric infections in feedlot cattle and BCV infection is associated with increased risks for respiratory disease and pulmonary lesions that are detected in feedlot cattle at slaughter.
Only a few studies have addressed the relationships between respiratory and enteric strains of BCV. Cross-neutralization tests have confirmed that respiratory and enteric BCV strains are antigenically similar and comprise a single BCV serotype.28, 55 Only one respiratory BCV strain differed from the two enteric BCV subtypes identified previously by HI and VN tests and this strain may comprise a third subtype.28 In experimental trials, gnotobiotic or colostrum-deprived calves infected with calf or adult cow enteric BCV strains were resistant to challenge with certain calf or feedlot isolates of respiratory BCV strains.7, 38
Comparison of the nucleotide sequences of the S gene of a calf respiratory BCV strain (G95) and a calf enteropathogenic BCV isolate (LY138) revealed 98,7 per cent nucleotide and 98,3 per cent amino acid sequence homology.81 No insertions or deletions were found and nucleotide substitutions were random. Similarly, the BCV S1 genes of enteric and respiratory isolates from Austria and Slovakia revealed almost 99 per cent similarity at the nucleotide level and no differences between enteric and respiratory strains.76 Likewise, analysis of the complete genomes of BCV sequenced directly from nasal or rectal swabs of two feedlot calves failed to reveal any consistent sequence differences between the enteric and respiratory strains.80 The analysis confirmed presence of intra-host quasispecies of BCV. Thus, like calf diarrhoea and winter dysentery (adult cow) isolates of BCV, the respiratory BCV isolates examined to date appear biologically, antigenically and genetically similar to the contemporary enteric BCV isolates.
Wild ruminants
Coronaviruses were first detected in wild ruminants by transmission electron microscopy (TEM) of diarrhoeic faeces of sitatunga (Tragelaphus spekei), musk oxen (Ovibus moschatus) and waterbuck in a wildlife park in England in 1984.5 The investigators noted that these coronaviruses reacted to a BCV-antigen specific ELISA, but attempts to cultivate them in HRT-18 cell cultures or transmit them to gnotobiotic calves failed, precluding further studies of the relationship of these coronaviruses to BCV.
Since then, coronaviruses antigenically, biologically and genetically related to BCV have been isolated and characterized from captive exotic wild ruminants [sambar deer and waterbuck] and from native wild ruminants [white-tailed deer, mule deer, caribou, elk and wapiti] in North America.1, 18, 44 The affected captive exotic wild ruminants were in a wildlife park in Ohio that was experiencing an epidemic outbreak of diarrhoea.71
In a serological survey of coronavirus infections among wild deer, 8,7 per cent and 6,6 per cent of sera from mule deer in Wyoming and from white-tailed deer in Ohio, respectively, were seropositive against both the white-tailed deer isolate and the BCV strains tested by indirect IF tests.71 Others18 reported that 14 per cent of adult caribou had antibodies to BCV.
These studies confirm the existence of coronaviruses antigenically and genetically closely related to BCV in captive and native wild ruminants. Thus the possibility exists that native wild ruminants may transmit coronavirus infections to cattle or vice versa. Because the other coronaviruses isolated were from exotic wild ruminants maintained in closed herds in wildlife parks, further studies are needed to confirm the existence and significance of coronavirus infections among native exotic ruminant species in their natural habitats.
Pathogenesis, clinical signs and pathology
Calf diarrhoea
Bovine coronavirus usually causes diarrhoea in 3- to 30-day old calves, but clinical disease may also occur in calves up to three months of age.8, 30, 58 It infects the absorptive epithelial cells of the distal small intestine (distal jejunum, and ileum) and large intestine, and the superficial and crypt enterocytes of the colon, leading to villous atrophy and crypt hyperplasia.37, 58, 74 The villous cells undergo pyknosis, karyorrhexis and lysis and they are replaced by immature cuboidal cells from the crypts. The crypt epithelium becomes hyperplastic and the lamina propria is infiltrated by lymphocytes. Young calves develop severe, watery, malabsorptive and potentially fatal diarrhoea resulting in progressive dehydration, acidosis, hyperkalaemia and hypoglycaemia. Gross lesions consist of distended thin-walled intestines and petechiae in the mucosa of the ileum and colon. Faeces are usually greenish or yellowish, liquid and sometimes haemorrhagic.17, 30 Diarrhoea usually persists for three to eight days. Faecal and nasal BCV shedding are detected for five to eight and two to four days, respectively, in BCV-inoculated colostrum- deprived or gnotobiotic calves.17, 30
Winter dysentery
The presence of BCV in adults is not always subclinical or benign as evidenced in the case of winter dysentery. Winter dysentery is an acute diarrhoeic disease of beef and dairy cattle characterized by semi-liquid, often bloody diarrhoea sometimes accompanied by a dry cough or other respiratory signs, with a marked reduction in milk production in dairy herds.57, 61, 63, 70, 74, 77 Intestinal lesions observed in field cases of winter dysentery resemble those described above for calf coronavirus enterocolitis.74 Affected animals also showed signs of body mass loss.53
Respiratory infection and BRD
As in the BRD complex in young adult cattle in feedlots, a respiratory disease complex also occurs in younger calves.30, 31, 32, 34, 38, 46, 55, 60, 68 Clinical signs including coughing, fever, serous to mucopurulent nasal discharge and inappetence were observed in calves under six months of age. Besides other viruses also associated with the BRD complex (adenovirus, parainfluenza-3 [PI-3], bovine herpesvirus [BHV], bovine viral diarrhoea virus [BVDV] and bovine respiratory syncytial virus [BRSV]), BCV has also been isolated from nasal swabs and pharyngeal or lung wash samples of sick calves. Many sick calves also had concurrent diarrhoea and shed BCV in faeces. Experimental inoculation of one- to nine-week-old calves with these respiratory BCV isolates resulted in the development of diarrhoea in most of them, accompanied by BCV shedding in their faeces and in nasal secretions for an average of five days. However, signs of respiratory disease (coughs, nasal discharge, and fever) occurred in only a few of them. Histological evidence of BCV antigen in the lung and focal interstitial emphysema 60 or interstitial pneumonia 34 were present in only a few of the calves. Thus, most respiratory BCV isolates from calves appear to be pneumoenteric strains capable of inducing diarrhoea, but with the infrequent manifestation of respiratory disease. However, in the field, respiratory BCV infections in calves are probably exacerbated by stress or co-infections with other respiratory pathogens.30, 31, 32, 46, 56, 68
The BRD complex also occurs in young adult cattle after commingling at auction or buyer barns and transport to feedlots.7, 22, 38, 39, 40, 41, 45, 65, 67 It is characterized by fever, dyspnoea and inflammatory and necrotizing lung lesions leading to bronchopneumonia, weight loss and death. Surveys of feedlot cattle in the USA suggest that respiratory disease accounts for 67 to 82 per cent of total morbidity and 44 per cent of deaths among feedlot cattle.38 Thus it was of interest to survey feedlot cattle for respiratory BCV infections in addition to screening for the established respiratory viral pathogens (PI-3, BHV, BVDV, BRSV) and secondary bacterial pathogens (Mannheimia and Pasteurella spp.). In doing so, we and others have detected BCV or BCV antibodies in such animals that were suffering from acute respiratory tract disease.7, 11, 22, 28, 38, 39, 40, 41, 45, 64, 65, 67 In these studies, BCV nasal shedding (0 to 76 per cent of cattle tested positive in eight feedlots) was detected most frequently shortly before and within the first week after transport.7, 38, 39, 40, 41, 64, 65 In one study,7 some feedlot cattle also had concurrent diarrhoea and 44,6 per cent were shedding BCV both nasally and in the faeces. The cattle shedding BCV in their faeces, but not those shedding BCV nasally, had significantly reduced weight gains. Cattle shedding BCV nasally or seroconverting to BCV were 1,6 times more likely to require treatment for respiratory disease and 2,2 times more likely to have pulmonary lesions when slaughtered.39, 40 Previous data documented that BCV-exposed cattle gained 26 per cent less weight than the healthy group during the first three weeks after arrival at the feedlot.27 Feedlot calves in New Mexico and Arkansas that shed BCV in faeces, nasal secretions, or both, gained on average 8 kg less weight than calves antigenically free from BCV over a five-week period following feedlot entry.67 In another investigation,64 it was found that 11 percent of affected cattle died of pneumonia within 5 to 36 days after onset of the epidemic and from 25 of these cattle, BCV or respiratory bacteria (Mannheimia haemolytica and/or Pasteurella multocida) were isolated from lung tissues of 72 per cent and 97 per cent, respectively. Clinical signs in the affected cattle included high fevers (44 °C) and severe respiratory distress. Gross lung lesions revealed that 50 to 80 per cent of their total lung volumes had subacute exudative and necrotizing lobar pneumonia affecting the cranioventral regions of the lung. No gross lesions were seen in other organs. Histological lung lesions were characteristically those of a fibrinous, necrotizing lobar pneumonia typical of pneumonic pasteurellosis. However, in other areas of the lung, moderate to severe bronchitis and bronchiolitis manifested by degenerating and necrotic respiratory epithelium and a non-suppurative inflammatory cell infiltration into the mucosa were observed. Fused epithelial cells were present in the airway epithelium adjacent to areas of necrosis and mucosal infiltration. In cattle that died within seven days of the outbreak, BCV antigen was detected by IF in the epithelial cells of the bronchi, bronchioli and alveolar ducts. Thus these studies confirm that BCV is an initiating cause of respiratory infections among feedlot cattle and is associated with an increased risk for cattle to develop respiratory disease, pulmonary lesions, and death following subsequent Mannheimia haemolytica and/or Pasteurella multocida infections.
Wild ruminants
The disease in exotic wild ruminants in a wildlife park in Ohio was characterized by an acute onset of diarrhoea persisting for several days, with bloody faeces and high morbidity in adults.71 Three of ten affected sambar deer died several days after the onset of diarrhoea. In the captive adult white-tailed deer, the disease was sporadic and characterized by acute onset of diarrhoea, with liquid faeces persisting for several days, and a low mortality rate.71 The disease in elk manifested as sporadic diarrhoea in which the faeces were bloody or soft.44
Diagnosis and differential diagnosis
As described in a detailed review, BCV infections can be diagnosed by detection of virus, viral antigen or viral RNA in the tissues, excretions or secretions of infected animals.29 The definitive diagnosis of infectious causes of diarrhoea in calves and wild ruminants or adult cattle with winter dysentery requires submission of faeces, intestinal contents, sections of intestine, or swabs of rectal fluid collected within one to three days of the onset of diarrhoea. Antigens are detected in enterocytes of the distal small intestine (distal jejunum and ileum) and colon in frozen sections or acetone-fixed intestinal impression smears by direct or indirect IF staining with hyperimmune antiserum or MAbs to BCV.8, 29, 58 The detection of BCV or BCV antigens in the faeces or rectal swab fluids from live animals (calves, wild ruminants, adult cattle) is accomplished using IEM 30, 31, 57, 58, 71 or an antigen detection ELISA.9, 10, 24, 29, 30, 63 The BCV particles in faeces can be visualized directly by negative staining followed by TEM. However, because faeces often contain pleomorphic membranous particles that resemble coronaviruses, the use of IEM increases the sensitivity, specificity and reliability of TEM.8, 31, 58 This technique involves incubation of 1 to 10 per cent filtered faecal suspensions with appropriately diluted BCV antibodies, resulting in large clusters of antibody-aggregated BCV particles (Figure 1). The IEM procedure has been further refined and the virions made more distinctive by labelling of the immune complexes with protein-A gold particles.31
An ELISA using a pool of MAbs to BCV was found to be sensitive and specific for the detection of BCV in faeces or rectal swab fluids.9, 10, 24, 29, 30, 63 A commercial ELISA kit using a similar MAb pool for detection of BCV in faeces was licensed in the USA.24 ELISA has the advantage of being applicable for testing large numbers of samples, but in one study62 it was less sensitive than IEM for detection of BCV from winter dysentery cases, possibly due to interference by the widespread presence of BCV antibodies in adult cattle.
Recent studies have also shown the feasibility of using RT-PCR assays for the detection of BCV RNA directly from faecal specimens of experimentally BCV-inoculated calves and adult cattle.6, 29, 73 Specific primers for RT-PCR were based on conserved regions of the N protein, so that the assay was broadly reactive for divergent strains of BCV, including those from calf diarrhoea, winter dysentery and wild ruminants. The RT-PCR assay was more sensitive than IEM or ELISA in detecting BCV in faeces of experimentally inoculated animals.
The recognition that BCV is also associated with both clinical and subclinical respiratory infections in calves and young adult cattle in feedlots 11, 28, 34, 39, 40, 41, 45, 55, 64, 65, 68 has necessitated the development of diagnostic assays for detection of BCV from respiratory tract samples. This has been accomplished by adaptation of ELISA,11, 29, 30, 31, 39, 40, 41 IEM and RT-PCR 7, 29, 79 to detect BCV directly from nasal swab fluids of infected cattle. In comparisons of nasal swabs (NS), guarded nasopharyngeal swabs (NPS), bronchoalveolar lavage (BAL), and transtracheal wash (TTW) from calves in California, BCV was detected by RT-PCR in 15.6 per cent, 20.9 per cent, 14.3 per cent, and 6.6 per cent of NS, NPS, BAL, and TTW samples, respectively; the difference between the NS and NPS results and the TTW result (used as a standard in the experiment) was reported to be significant.15 In addition, BCV antigens can be detected by IF in nasal epithelial cells collected from nasal swab specimens of BCV-infected cattle.30, 31 HRT-18 cell cultures have been used to isolate BCV from nasal swabs or respiratory tissues of cattle with respiratory disease,11, 28, 64, 65 but the failure of some BCV strains to grow in cell culture and the time involved limit the usefulness of this approach in most diagnostic laboratories.
Because numerous other enteric viruses are associated with diarrhoea in calves and also induce atrophy of the intestinal villi (e.g. Bredavirus, rotaviruses, enteric calicivirus), an aetiological diagnosis is required for BCV enteric infections. Similarly, a number of other viruses and bacteria can cause respiratory infections of cattle (e.g. PI-3, BHV, BVDV, BRSC, Mannheimia haemolytica, Pasteurella multocida, certain mycoplasmas) necessitating their differentiation from BCV by diagnostic assays.
Antibodies to BCV can be quantified by several methods.29 Virus neutralization tests, using the reference Mebus NCDV BCV strain and sera from field cases, can be conducted using MDBK or HRT-18 cell cultures.2, 13, 59, 71, 72 Antibodies to all BCV strains tested cross-reacted with the Mebus strain although some strains had several-fold higher VN antibody titres against the homologous strain.13, 28, 71, 72 ELISAs have also been developed to detect BCV antibodies and antibody isotypes in serum and their sensitivity and specificity have been confirmed in several studies.30, 32, 39, 40, 41, 62 The HI test can be performed on sera using mouse or chicken erythrocytes. Bovine coronavirus strains are also cross-reactive in this test, but with some diversity in HI antibody titres.13, 28, 49, 72, 73 Because antibodies to BCV are widespread in cattle, examination of paired serum samples (acute and after two to four weeks’ convalescence) is necessary for serological diagnosis. Calves with high titres of maternally derived antibodies or acute phase serum samples collected later than three to four days after disease onset may not show unequivocal antibody increases, limiting the usefulness of serology in these cases, unless isotype-specific antibody ELISAs are used to detect increases in IgM and IgA antibody titres to BCV.8, 30, 32, 43
Control
Control of BCV currently depends on management strategies aimed at prevention, because although there are products that help to prevent diarrhoea in calves, the role of vaccination in managing the BCV disease complex remains limited. Multiple issues need to be addressed for development of effective BCV vaccines for enteric and respiratory BCV infections associated with the various BCV syndromes highlighted. These include: 1) The target cattle age groups (passive versus active immunity; 2) The subtypes or clades of BCV strains to be included; and 3) The induction of mucosal immunity in the respiratory and intestinal tracts to prevent severe disease and weight loss (but not necessarily virus shedding).
Calf diarrhoea
Control of neonatal enteric BCV infections in calves within the first few weeks of life is complicated by the frequent association of BCV diarrhoea with other enteropathogens. A commercial BCV vaccine used in the USA was not highly efficacious, either when given orally to calves at birth to stimulate active immunity 12 or when administered parenterally to cows to provide colostrum-derived passive immunity to calves. 51, 58 Although oral inoculation of newborn colostrum-deprived calves with an attenuated BCV vaccine induced active immunity to experimental challenge, the presence of antibodies to BCV in colostrum interfered with the efficacy of the oral BCV vaccine in colostrum-fed calves.12, 58
A more efficient method for controlling BCV infections in calves appears to be immunization of cows with inactivated, adjuvanted BCV vaccines to increase antibody titres in colostrum and milk over levels normally present in field exposed cows.48, 50, 58 Frequent feeding of the BCV immune colostrum or artificial colostrum containing gammaglobulins that include antibodies to BCV reduced the severity of BCV diarrhoea in calves in experimental challenge studies,50, 58 but lactogenic immunity was less effective in providing passive protection to field-exposed calves in the presence of a high degree of BCV exposure.48 Thus, continued feeding of colostrum from BCV-vaccinated cows (from frozen or acidified colostrum banks) during the first week of life may be helpful in preventing BCV diarrhoea and mortality in highly susceptible suckling calves. In addition, feeding of at least three litres of colostrum to calves within a few hours of birth via an oesophageal feeder should ensure adequate levels of serum immunoglobulins, which may contribute to passive immunity against BCV infections of the respiratory and intestinal tracts.
Respiratory infection and BRD
At present, there is little information on the prevention or control of respiratory or enteric BCV infections in weaned and adult cattle, as well as on the impact of existing BCV vaccines on such infections. No BCV vaccines have been licensed to prevent BCV-associated pneumonia in young calves or in feedlot cattle, despite their economic impacts. Also the correlates of immunity to respiratory BCV infections remain undefined. In one study, intranasal vaccination of feedlot calves with the historic modified live enteric BCV calf vaccine (NCDV strain) on entry to a feedlot reduced the risk of treatment for BRD in the calves by 36 per cent.54 Likewise, from calf challenge studies, virulent enteric and respiratory BCV strains given oronasally, even ones from different subtypes or subclades, cross-protected against diarrhoea, but not necessarily shedding (for heterotypic strains).7, 17, 38, 55 Thus both natural and experimental BCV infections may prevent disease upon reinfection, but may fail to prevent virus shedding. An important observation regarding BCV transmission was that although prolonged faecal or nasal shedding of BCV RNA detected by RT-PCR can occur (28-35 days), naïve sentinal contact calves were not infected when exposed to the shedding calves at 21 days after infection.52
The role of serum antibodies to BCV in protection against respiratory BCV infections is unclear. In several studies, cattle with adequate serum BCV antibody titres (HI, VN or ELISA IgA, IgG1 and IgG2 isotypes) on arrival in feedlots appeared to be protected against respiratory disease, pneumonia or BCV shedding.7, 22, 42, 43, 45, 67 Similarly, in a study of young calves, there was a strong correlation between IgG1, IgG2, and IgA antibody titres to BCV in serum and the number of respiratory sick days.32 These data suggest that if serum BCV antibodies are a correlate of immunity to respiratory BCV infection, then parenteral vaccines to boost levels of existing BCV antibodies may be protective. Such vaccines may be best implemented by vaccinating weaned calves on farm before shipping to auction barns or feedlots. This is because BCV infections occur after commingling of calves from different farms at the auction barn or shortly pre- or post- arrival at feedlots, necessitating rapid onset of immunity.7, 22, 67
The correlates of protective immunity associated with BCV infections in winter dysentery outbreaks in adult cattle are unclear, including the role of pre-existing serum antibodies to BCV in the affected adult cattle.31, 62, 73, 77 In the context of the three diverse clinical syndromes associated with BCV and the 2-3 subtypes or subclades reported,21, 28 especially ones divergent from the historical BCV NCDV calf vaccine strain, use of a mixture of such strains is one strategy to develop a broad spectrum BCV vaccine. Such a vaccine should be effective against each of the BCV clinical syndromes highlighted.
Because of the lack of highly effective vaccines for BCV, good management procedures are especially important in reducing exposure of susceptible young calves to infected faecal material or aerosols. These include thorough cleaning and disinfection of calving or nursery units and grounds, adequate ventilation to reduce airborne transmission, reducing direct contact between calves by use of clean, dry calf hutches, and minimizing mechanical spread of virus by thorough cleaning and disinfection of common equipment, including boots and coveralls. Symptomatic treatment of severely affected calves may be needed to prevent dehydration. This requires either oral or, in advanced cases, parenteral administration of electrolyte fluids. Reservoirs of BCV infections in a herd may be subclinically infected adult cows or older calves in which recurrent infections are common.9, 30, 32 Therefore, removing calves from their dams shortly after calving and avoiding the mixing of younger with older cattle are particularly important management strategies. Similar applicable management strategies to control BCV infections associated with winter dysentery 61, 63, 77 and BRD 20 have been reviewed.
References
- ALEKSEEV, K. P., VLASOVA, A.N., JUNG, K., HASOKSUZ, M., ZHANG, X., HALPIN, R., WANG, S., GHEDIN, E., SPIRO, D. & SAIF, L.J., 2008. Bovine-like coronaviruses isolated from four species of captive wild ruminants are homologous to bovine coronaviruses, based on complete genomic sequences. Journal of Virology, 82, 12422-12431.
- BENFIELD, D. A. & SAIF, L.J., 1990. Cell culture propagation of a coronavirus isolated from cows with winter dysentery. Journal of Clinical Microbiology, 28, 1454-1457.
- BIDOKHTI, M. R., TRAVEN, M., KRISHNA, N.K., MUNIR, M., BELAK, S., ALENIUS, S. & CORTEY, M., 2013. Evolutionary dynamics of bovine coronaviruses: natural selection pattern of the spike gene implies adaptive evolution of the strains. The Journal of General Virology, 94, 2036-2049.
- BOK, M., MINO, S., RODRIGUEZ, D., BADARACCO, A., NUNES, I., SOUZA, S.P., BILBAO, G., LOUGE URIARTE, E., GALARZA, R., VEGA, C., ODEON, A., SAIF, L.J. & PARRENO, V., 2015. Molecular and antigenic characterization of bovine Coronavirus circulating in Argentinean cattle during 1994-2010. Veterinary Microbiology, 181, 221-229.
- CHASEY, D., REYNOLDS, D.J., BRIDGER, J.C., DEBNEY, T.G. & SCOTT, A.C., 1984. Identification of coronaviruses in exotic species of Bovidae. The Veterinary Record, 115, 602-603.
- CHO, K. O., HALBUR, P.G., BRUNA, J.D., SORDEN, S.D., YOON, K.J., JANKE, B.H., CHANG, K.O. & SAIF, L.J., 2000. Detection and isolation of coronavirus from feces of three herds of feedlot cattle during outbreaks of winter dysentery-like disease. Journal of the American Veterinary Medical Association, 217, 1191-1194.
- CHO, K. O., HOET, A.E., LOERCH, S.C., WITTUM, T.E. & SAIF, L.J., 2001. Evaluation of concurrent shedding of bovine coronavirus via the respiratory tract and enteric route in feedlot cattle. American Journal of Veterinary Research, 62, 1436-1441.
- CLARK, M. A. 1993. Bovine coronavirus. The British Veterinary Journal, 149, 51-70.
- CROUCH, C. F. & ACRES, S.D., 1984. Prevalence of rotavirus and coronavirus antigens in the feces of normal cows. Canadian journal of comparative medicine : Revue Canadienne de Medecine Comparee, 48, 340-342.
- CROUCH, C. F., RAYBOULD, T.J. & ACRES, S.D., 1984. Monoclonal antibody capture enzyme-linked immunosorbent assay for detection of bovine enteric coronavirus. Journal of Clinical Microbiology, 19, 388-393.
- DA SILVA, M. R., O'REILLY, K.L., LIN, X., STINE, L. & STORZ, J., 1999. Sensitivity comparison for detection of respiratory bovine coronaviruses in nasal samples from feedlot cattle by ELISA and isolation with the G clone of HRT-18 cells. Journal of veterinary diagnostic investigation : official publication of the American Association of Veterinary Laboratory Diagnosticians, Inc, 11, 15-19.
- DE LEEUW, P. W. & TIESSINK, J.W., 1985. Laboratory experiments on oral vaccination of calves against rotavirus or coronavirus induced diarrhoea. Zentralblatt fur Veterinarmedizin. Reihe B. Journal of Veterinary Medicine, Series B, 32, 55-64.
- DEA, S., MICHAUD, L. & MILANE, G., 1995. Comparison of bovine coronavirus isolates associated with neonatal calf diarrhoea and winter dysentery in adult dairy cattle in Quebec. The Journal of General Virology, 76 ( Pt 5), 1263-1270.
- DECARO, N., MARTELLA, V., ELIA, G., CAMPOLO, M., MARI, V., DESARIO, C., LUCENTE, M.S., LORUSSO, A., GRECO, G., CORRENTE, M., TEMPESTA, M. & BUONAVOGLIA, C., 2008. Biological and genetic analysis of a bovine-like coronavirus isolated from water buffalo (Bubalus bubalis) calves. Virology, 370, 213-222.
- DOYLE, D., CREDILLE, B., LEHENBAUER, T.W., BERGHAUS, R., ALY, S.S., CHAMPAGNE, J., BLANCHARD, P., CROSSLEY, B., BERGHAUS, L., COCHRAN, S. & WOOLUMS, A., 2017. Agreement Among 4 Sampling Methods to Identify Respiratory Pathogens in Dairy Calves with Acute Bovine Respiratory Disease. Journal of Veterinary Internal Medicine, 31, 954-959.
- EL-GHORR, A. A., SNODGRASS, D.R., SCOTT, F.M. & CAMPBELL, I., 1989. A serological comparison of bovine coronavirus strains. Archives of Virology, 104, 241-248.
- EL-KANAWATI, Z. R., TSUNEMITSU, H., SMITH, D.R. & SAIF, L.J., 1998. Infection and cross-protection studies of winter dysentery and calf diarrhea bovine coronavirus astrains in colostrum-deprived and gnotobiotic calves. American Jounal of Veterinary Research, 57, 48-53.
- ELAZHARY, M. A., FRECHETTE, J.L., SILIM, A. & ROY, R.S., 1981. Serological evidence of some bovine viruses in the caribou (Rangifer tarandus caribou) in Quebec. Journal of wildlife diseases, 17, 609-612.
- FUKOTOMI, T., TSUNEMITSU, H., AKASHI, H. & SAIF, L.J., 1998. Antigenic diversity within bovine coronaviruses isolated in Japan. Conference Proceedings, Second International Rushmore Conference on Mechanisms in the Pathogenesis of Enteric Diseases, Abstract. Rapid City, S.D., 30 September-3 October 1998 (43).
- FULTON, R. W., 2009. Bovine respiratory disease research (1983-2009). Animal health research reviews, 10, 131-139.
- FULTON, R. W., RIDPATH, J.F. & BURGE, L.J., 2013. Bovine coronaviruses from the respiratory tract: antigenic and genetic diversity. Vaccine, 31, 886-892.
- FULTON, R. W., STEP, D.L., WAHRMUND, J., BURGE, L.J., PAYTON, M.E., COOK, B.J., BURKEN, D., RICHARDS, C.J. & CONFER, A.W., 2011. Bovine coronavirus (BCV) infections in transported commingled beef cattle and sole-source ranch calves. Canadian Journal of Veterinary Research = Revue Canadienne de Recherche Veterinaire, 75, 191-199.
- GOMEZ, D. E., ARROYO, L.G., POLJAK, Z., VIEL, L. & WEESE, J.S., 2017. Detection of Bovine Coronavirus in Healthy and Diarrheic Dairy Calves. Journal of Veterinary Internal Medicine, 15, 1-8.
- GRAY, M., HUCHZERMEIER, R., SAIF, L.J., ZEMAN, D. & OSORIO, F., 1999. Evaluation of a new commercial antigen capture ELISA for detection of bovine coronavirus in fecal samples. Veterinary Diagnosticians Meeting, 8-11 October 1999, San Diego, CA, Abstract (11).
- HAJER, I. & STORZ, J., 1978. Antigens of bovine coronavirus strain LY-138 and their diagnostic properties. American Journal of Veterinary Research, 39, 441-444.
- HASOKSUZ, M., ALEKSEEV, K., VLASOVA, A., ZHANG, X., SPIRO, D., HALPIN, R., WANG, S., GHEDIN, E. & SAIF, L.J., 2007. Biologic, antigenic, and full-length genomic characterization of a bovine-like coronavirus isolated from a giraffe. Journal of Virology, 81, 4981-4990.
- HASOKSUZ, M., HOET, A.E., LOERCH, S.C., WITTUM, T.E., NIELSEN, P.R. & SAIF, L.J., 2002. Detection of respiratory and enteric shedding of bovine coronaviruses in cattle in an Ohio feedlot. Journal of Veterinary Diagnostic Investigation : Official Publication of The American Association of Veterinary Laboratory Diagnosticians, Inc, 14, 308-313.
- HASOKSUZ, M., LATHROP, S.L., GADFIELD, K.L. & SAIF, L.J., 1999. Isolation of bovine respiratory coronaviruses from feedlot cattle and comparison of their biological and antigenic properties with bovine enteric coronaviruses. American Journal of Veterinary Research, 60, 1227-1233.
- HASOKSUZ, M., VLASOVA, A. & SAIF, L.J., 2008. Detection of group 2a coronaviruses with emphasis on bovine and wild ruminant strains. Virus isolation and detection of antibody, antigen, and nucleic acid. Methods in Molecular Biology, 454, 43-59.
- HECKERT, R. A., SAIF, L.J., HOBLET, K.H. & AGNES, A.G., 1990. A longitudinal study of bovine coronavirus enteric and respiratory infections in dairy calves in two herds in Ohio. Veterinary Microbiology, 22, 187-201.
- HECKERT, R. A., SAIF, L.J. & MYERS, G.W., 1989. Development of protein A-gold immunoelectron microscopy for detection of bovine coronavirus in calves: comparison with ELISA and direct immunofluorescence of nasal epithelial cells. Veterinary Microbiology, 19, 217-231.
- HECKERT, R. A., SAIF, L.J., MYERS, G.W. & AGNES, A.G., 1991. Epidemiologic factors and isotype-specific antibody responses in serum and mucosal secretions of dairy calves with bovine coronavirus respiratory tract and enteric tract infections. American Journal of Veterinary Research, 52, 845-851.
- ICTV., 2017. INTERNATIONAL COMMITTEE ON TAXONOMY OF VIRUSES (ICTV). Taxonomy.⟨https:// talk.ictvonline.org/taxonomy⟩.
- KAPIL, S., POMEROY, K.A., GOYAL, S.M. & TRENT, A.M., 1991. Experimental infection with a virulent pneumoenteric isolate of bovine coronavirus. Journal of veterinary diagnostic investigation : Official Publication of the American Association of Veterinary Laboratory Diagnosticians, Inc, 3, 88-89.
- KIN, N., GUERARD, P., DIANCOURT, L., CARO, V., VABRET, A. & AND AR GOUILH, M., 2017. First Complete Genome Sequence of a French Bovine coronavirus Strain. Genome Announcements, 5.
- KIN, N., MISZCZAK, F., DIANCOURT, L., CARO, V., MOUTOU, F., VABRET, A. & AR GOUILH, M., 2016. Comparative molecular epidemiology of two closely related coronaviruses, bovine coronavirus (BCoV) and human coronavirus OC43 (HCoV-OC43), reveals a different evolutionary pattern. Infection, Genetics and Evolution : Journal of Molecular Epidemiology and Evolutionary Genetics in Infectious Diseases, 40, 186-191.
- LANGPAP, T. J., BERGELAND, M.E. & REED, D.E., 1979. Coronaviral enteritis of young calves: virologic and pathologic findings in naturally occurring infections. American Journal of Veterinary Research, 40, 1476-1478.
- LATHROP, S. L., 1999. The epidemiology of bovine respiratory coronavirus infections in feedlot cattle. Food Animal Health Research Program, Veterinary Preventive Medicine, Ohio Agricultural Research and Development Center, The Ohio State University, Wooster, Ohio, PhD thesis.
- LATHROP, S. L., WITTUM, T.E., BROCK, K.V., LOERCH, S.C., PERINO, L.J., BINGHAM, H.R., MCCOLLUM, F.T. & SAIF, L.J., 2000. Association between infection of the respiratory tract attributable to bovine coronavirus and health and growth performance of cattle in feedlots. American Journal of Veterinary Research, 61, 1062-1066.
- LATHROP, S. L., WITTUM, T.E., LOERCH, S.C., PERINO, L.J. & SAIF, L.J., 2000. Antibody titers against bovine coronavirus and shedding of the virus via the respiratory tract in feedlot cattle. American Journal of Veterinary Research, 61, 1057-1061.
- LATHROP, S. L., WITTUM, T.E., MORLEY, P.S., SMITH, D.R., BINGHAM, H.R. & SAIF, L.J., 1996. Bovine coronavirus respiratory infections in feedlot cattle. Proceedings of the Conference for Research Workers in Animal Disease, 11-12 November 1996, Abstract (200).
- LIN, X., O'REILLY, K.L., BURRELL, M.L. & STORZ, J., 2001. Infectivity-neutralizing and hemagglutinin-inhibiting antibody responses to respiratory coronavirus infections of cattle in pathogenesis of shipping fever pneumonia. Clinical and Diagnostic Laboratory Immunology, 8, 357-362.
- LIN, X. Q., KL, O.E., STORZ, J., PURDY, C.W. & LOAN, R.W., 2000. Antibody responses to respiratory coronavirus infections of cattle during shipping fever pathogenesis. Archives of Virology, 145, 2335-2349.
- MAJHDI, F., MINOCHA, H.C., & KAPIL, S., 1997. Isolation and characterization of a coronavirus from elk calves with diarrhea. Journal of Clinical Microbiology, 35, 2937-2942.
- MARTIN, S. W., NAGY, E., SHEWEN, P.E. & HARLAND, R.J., 1998. The association of titers to bovine coronavirus with treatment for bovine respiratory disease and weight gain in feedlot calves. Canadian Journal of Veterinary Research = Revue Canadienne De Recherche Veterinaire, 62, 257-261.
- MCNULTY, M. S., BRYSON, D.G., ALLAN, G.M. & LOGAN, E.F., 1984. Coronavirus infection of the bovine respiratory tract. Veterinary Microbiology, 9, 425-434.
- MEBUS, C. A., STAIR, E,L., RHODES, M.B. & TWIEHAUS, M.J., 1973. Pathology of neonatal calf diarrhea induced by a coronavirus-like agent. Veterinary Pathology, 10, 45-64.
- MOSTL, K. & BURKI, F., 1988. Incidence of diarrhoea and of rotavirus- and coronavirus-shedding in calves, whose dams had been vaccinated with an experimental oil-adjuvanted vaccine containing bovine rotavirus and bovine coronavirus. Zentralblatt fur Veterinarmedizin. Reihe B. Journal of Veterinary Medicine, Series B, 35, 186-196.
- MUNIIAPPA, L., MITOV, B.K. & KHARALAMBIEV, K.H.E., 1985. Demonstration of coronavirus infection in buffaloes. Veterinarno-Meditsinski Nauki, 22, 27-32.
- MURAKAMI, T., HIRANO, N., INOUE, A., CHITOSE, K., TSUCHIYA, K., ONO, K. & NAITO, Y., 1985. Transfer of antibodies against viruses of calf diarrhea from cows to their offspring via colostrum. Nihon juigaku zasshi. The Japanese Journal of Veterinary Science, 47, 507-510.
- MYERS, L. L. & SNODGRASS, D.R., 1982. Colostral and milk antibody titers in cows vaccinated with a modified live-rotavirus-coronavirus vaccine. Journal of the American Veterinary Medical Association, 181, 486-488.
- OMA, V. S., TRAVEN, M., ALENIUS, S., MYRMEL, M. & STOKSTAD, M., 2016. Bovine coronavirus in naturally and experimentally exposed calves; viral shedding and the potential for transmission. Virology Journal, 13, 100.
- PARK, S. J., KIM, G.Y., CHOY, H.E., HONG, Y.J., SAIF, L.J., JEONG, J.H., PARK, S.I., KIM, H.H., KIM, S.K., SHIN, S.S., KANG, M.I. & CHO, K.O., 2007. Dual enteric and respiratory tropisms of winter dysentery bovine coronavirus in calves. Archives of Virology, 152, 1885-1900.
- PLUMMER, P. J., ROHRBACH, B.W., DAUGHERTY, R.A., DAUGHERTY, R.A., THOMAS, K.V., WILKES, R.P., DUGGAN, F.E. & KENNEDY, M.A., 2004. Effect of intranasal vaccination against bovine enteric coronavirus on the occurrence of respiratory tract disease in a commercial backgrounding feedlot. Journal of the American Veterinary Medical Association, 225, 726-731.
- REYNOLDS, D. J., DEBNEY, T.G., HALL, G.A., THOMAS, L.H. & PARSONS, K.R., 1985. Studies on the relationship between coronaviruses from the intestinal and respiratory tracts of calves. Archives of Virology, 85, 71-83.
- SAIF, L. J., 2010. Bovine respiratory coronavirus. The Veterinary clinics of North America. Food Animal Practice, 26, 349-364.
- SAIF, L. J., 1990. A review of evidence implicating bovine coronavirus in the etiology of winter dysentery in cows: an enigma resolved? The Cornell Veterinarian, 80, 303-311.
- SAIF, L. J. & HECKERT, R.H., 1990. Enteropathogenic Coronaviruses. In: SAIF, L.J. & THEIL, K.W., (eds). Viral Diarrheas of Man and Animals. Boca Raton, FL: CRC Press, 185-252.
- SAIF, L. J., HECKERT, R.A., MILLER, K.L. & TAREK, M.M., 1988. Cell Culture Propagation of Rotaviruses. Journal of Tissue Culture Methods, 11, 147-156.
- SAIF, L. J., REDMAN, D.R. & THEIL, K.W., 1986. Experimental coronavirus infections in calves: iral replication in the respiratory and intestinal tracts. American Journal of Veterinary Research, 47, 1426-1432.
- SMITH, D. R., FEDORKA-CRAY, P.J., MOHAN, R., BROCK, K.V., WITTUM, T.E., MORLEY, P.S., HOBLET, K.H. & SAIF, L.J.,1998. Epidemiologic herd-level assessment of causative agents and risk factors for winter dysentery in dairy cattle. American Journal of Veterinary Research, 59, 994-1001.
- SMITH, D. R., FEDORKA-CRAY, P.J., MOHAN, R., BROCK, K.V., WITTUM, T.E., MORLEY, P.S., HOBLET, K.H. & SAIF, L.J., 1998. Evaluation of cow-level risk factors for the development of winter dysentery in dairy cattle. American Journal of Veterinary Research, 59, 986-993.
- SMITH, D. R., TSUNEMITSU, H., HECKERT, R.A. & SAIF, L.J., 1996. Evaluation of two antigen-capture ELISAs using polyclonal or monoclonal antibodies for the detection of bovine coronavirus. Journal of Veterinary Diagnostic Investigation : Official Publication of The American Association Of Veterinary Laboratory Diagnosticians, Inc, 8, 99-105.
- STORZ, J., LIN, X., PURDY, C.W., CHOULJENKO, V.N., KOUSOULAS, K.G., ENRIGHT, F.M., GILMORE, W.C., BRIGGS, R.E. & LOAN, R.W., 2000. Coronavirus and Pasteurella infections in bovine shipping fever pneumonia and Evans' criteria for causation. Journal of Clinical Microbiology, 38, 3291-3298.
- STORZ, J., STINE, L., LIEM, A. & ANDERSON, G.A., 1996. Coronavirus isolation from nasal swab samploes in catttle with signs of respiratory tract disease after shipping. Journal of American Veterinary Medical Association, 208, 1452-1455.
- STRAUB, O. C., 1995. Viral respiratory infections of cattle. Bovine Practitioner, 29, 66-70.
- THOMAS, C. J., HOET, A.E., SREEVATSAN, S., WITTUM, T.E., BRIGGS, R.E., DUFF, G.C. & SAIF, L.J., 2006. Transmission of bovine coronavirus and serologic responses in feedlot calves under field conditions. American Journal of Veterinary Research, 67, 1412-1420.
- THOMAS, L. H., GOURLAY, R.N., STOTT, E.J., HOWARD, C.J. & BRIDGER, J.C., 1982. A search for new microorganisms in calf pneumonia by the inoculation of gnotobiotic calves. Research in Veterinary Science, 33, 170-182.
- TRAVEN, M., BJORNEROT, L. & LARSSON, B., 1999. Nationwide survey of antibodies to bovine coronavirus in bulk milk from Swedish dairy herds. The Veterinary Record, 144, 527-529.
- TRAVEN, M., NASLUND, K., LINDE, N., LINDE, B., SILVAN, A., FOSSUM, C., HEDLUND, K.O. & LARSSON, B., 2001. Experimental reproduction of winter dysentery in lactating cows using BCV -- comparison with BCV infection in milk-fed calves. Veterinary Microbiology, 81, 127-151.
- TSUNEMITSU, H., EL-KANAWATI, Z.R., SMITH, D.R., REED, H.H. & SAIF, L.J., 1995. Isolation of coronaviruses antigenically indistinguishable from bovine coronavirus from wild ruminants with diarrhea. Journal of Clinical Microbiology, 33, 3264-3269.
- TSUNEMITSU, H. & SAIF, L.J., 1995. Antigenic and biological comparisons of bovine coronaviruses derived from neonatal calf diarrhea and winter dysentery of adult cattle. Archives of Virology, 140, 1303-1311.
- TSUNEMITSU, H., SMITH, D.R. & SAIF, L.J., 1999. Experimental inoculation of adult dairy cows with bovine coronavirus and detection of coronavirus in feces by RT-PCR. Archives of Virology, 144, 167-175.
- VAN KRUININGEN, H. J., KHAIRALLAH, L.H., SASSEVILLE, V.G., WYAND, M.S. & POST, J.E., 1987. Calfhood coronavirus enterocolitis: a clue to the etiology of winter dysentery. Veterinary Pathology, 24, 564-567.
- VIJGEN, L., KEYAERTS, E., MOES, E., THOELEN, I., WOLLANTS, E., LEMEY, P., VANDAMME, A.M. & VAN RANST, M., 2005. Complete genomic sequence of human coronavirus OC43: molecular clock analysis suggests a relatively recent zoonotic coronavirus transmission event. Journal of Virology, 79, 1595-1604.
- VILCEK, S., JACKOVA, A., KOLESAROVA, M. & VLASAKOVA, M., 2017. Genetic variability of the S1 subunit of enteric and respiratory bovine coronavirus isolates. Acta Virologica, 61, 212-216.
- WHITE, M. E., SCHUKKEN, Y.H. & TANKSLEY, B., 1989. Space-time clustering of, and risk factors for, farmer-diagnosed winter dysentery in dairy cattle. The Canadian Veterinary Journal = La Revue Veterinaire Canadienne, 30, 948-951.
- WOO, P. C., LAU, S.K., WERNERY, U., WONG, E.Y., TSANG, A.K., JOHNSON, B., YIP, C.C., LAU, C.C., SIVAKUMAR, S., CAI, J.P., FAN, R.Y., CHAN, K.H., MAREENA, R. & YUEN, K.Y., 2014. Novel betacoronavirus in dromedaries of the Middle East, 2013. Emerging Infectious Diseases, 20, 560-572.
- WORKMAN, A. M., KUEHN, L.A., MCDANELD, T.G., CLAWSON, M.L., CHITKO-MCKOWN, C.G. & LOY, J,D., 2017. Evaluation of the effect of serum antibody abundance against bovine coronavirus on bovine coronavirus shedding and risk of respiratory tract disease in beef calves from birth through the first five weeks in a feedlot. American Journal of Veterinary Research, 78, 1065-1076.
- ZHANG, X., HASOKSUZ, M., SPIRO, D., HALPIN, R., WANG, S., VLASOVA, A., JANIES, D., JONES, L.R., GHEDIN, E. & SAIF, L.J., 2007. Quasispecies of bovine enteric and respiratory coronaviruses based on complete genome sequences and genetic changes after tissue culture adaptation. Virology, 363, 1-10.
- ZHANG, X., HERBST, W., KOUSOULAS, K.G. & STORZ, J., 1994. Comparison of the S genes and the biological properties of respiratory and enteropathogenic bovine coronaviruses. Archives of Virology, 134, 421-426.