- Infectious Diseases of Livestock
- Part 2
- Swinepox
- GENERAL INTRODUCTION: PARAMYXOVIRIDAE AND PNEUMOVIRIDAE
- Rinderpest
- Peste des petits ruminants
- Parainfluenza type 3 infection
- Bovine respiratory syncytial virus infection
- Hendra virus infection
- Paramyxovirus-induced reproductive failure and congenital defects in pigs
- Nipah virus disease
- GENERAL INTRODUCTION: CALICIVIRIDAE AND ASTROVIRIDAE
- Vesicular exanthema
- Enteric caliciviruses of pigs and cattle
- GENERAL INTRODUCTION: RETROVIRIDAE
- Enzootic bovine leukosis
- Jaagsiekte
- Visna-maedi
- Caprine arthritis-encephalitis
- Equine infectious anaemia
- GENERAL INTRODUCTION: PAPILLOMAVIRIDAE
- Papillomavirus infection of ruminants
- Papillomavirus infection of equids
- GENERAL INTRODUCTION: ORTHOMYXOVIRIDAE
- Equine influenza
- Swine influenza
- GENERAL INTRODUCTION: CORONAVIRIDAE
- Porcine transmissible gastroenteritis
- Porcine respiratory coronavirus infection
- Porcine epidemic diarrhoea
- Porcine haemagglutinating encephalomyelitis virus infection
- Porcine deltacoronavirus infection
- Bovine coronavirus infection
- Ovine coronavirus infection
- Equine coronavirus infection
- GENERAL INTRODUCTION: PARVOVIRIDAE
- Porcine parvovirus infection
- Bovine parvovirus infection
- GENERAL INTRODUCTION: ADENOVIRIDAE
- Adenovirus infections
- GENERAL INTRODUCTION: HERPESVIRIDAE
- Equid herpesvirus 1 and equid herpesvirus 4 infections
- Equid gammaherpesvirus 2 and equid gammaherpesvirus 5 infections
- Equine coital exanthema
- Infectious bovine rhinotracheitis/infectious pustular vulvovaginitis and infectious pustular balanoposthitis
- Bovine alphaherpesvirus 2 infections
- Malignant catarrhal fever
- Pseudorabies
- Suid herpesvirus 2 infection
- GENERAL INTRODUCTION: ARTERIVIRIDAE
- Equine viral arteritis
- Porcine reproductive and respiratory syndrome
- GENERAL INTRODUCTION: FLAVIVIRIDAE
- Bovine viral diarrhoea and mucosal disease
- Border disease
- Hog cholera
- Wesselsbron disease
- Louping ill
- West nile virus infection
- GENERAL INTRODUCTION: TOGAVIRIDAE
- Equine encephalitides caused by alphaviruses in the Western Hemisphere
- Old World alphavirus infections in animals
- Getah virus infection
- GENERAL INTRODUCTION: BUNYAVIRIDAE
- Diseases caused by Akabane and related Simbu-group viruses
- Rift Valley fever
- Nairobi sheep disease
- Crimean-Congo haemorrhagic fever
- GENERAL INTRODUCTION: ASFARVIRIDAE
- African swine fever
- GENERAL INTRODUCTION: RHABDOVIRIDAE
- Rabies
- Bovine ephemeral fever
- Vesicular stomatitis and other vesiculovirus infections
- GENERAL INTRODUCTION: REOVIRIDAE
- Bluetongue
- Ibaraki disease in cattle
- Epizootic haemorrhagic disease
- African horse sickness
- Equine encephalosis
- Palyam serogroup orbivirus infections
- Rotavirus infections
- GENERAL INTRODUCTION: POXVIRIDAE
- Lumpy skin disease
- Sheeppox and goatpox
- Orf
- Ulcerative dermatosis
- Bovine papular stomatitis
- Pseudocowpox
- Swinepox
- Cowpox
- Horsepox
- Camelpox
- Buffalopox
- GENERAL INTRODUCTION: PICORNAVIRIDAE
- Teschen, Talfan and reproductive diseases caused by porcine enteroviruses
- Encephalomyocarditis virus infection
- Swine vesicular disease
- Equine picornavirus infection
- Bovine rhinovirus infection
- Foot-and-mouth disease
- GENERAL INTRODUCTION: BORNAVIRIDAE
- Borna disease
- GENERAL INTRODUCTION: CIRCOVIRIDAE AND ANELLOVIRIDAE
- Post-weaning multi-systemic wasting syndrome in swine
- GENERAL INTRODUCTION: PRION DISEASES
- Scrapie
- Bovine spongiform encephalopathy
- Transmissible spongiform encephalopathies related to bovine spongiform encephalopathy in other domestic and captive wild species
Swinepox
This content is distributed under the following licence: Attribution-NonCommercial CC BY-NC  View Creative Commons Licence details here
View Creative Commons Licence details here
NJ Maclachlan and M-L Penrith (Editors). S Babiuk, Swinepox, 2018.
Swinepox
Previous authors: E MUNZ AND K DUMBELL
Current authors:
S BABIUK - Research Scientific, PhD, National Centre for Foreign Animal Disease, 1015 Arlington Street, Manitoba, Canada, R3E 3MA
Introduction
Swinepox is an acute but usually mild disease particularly of young pigs characterized by the development of pox skin lesions.
In the past a similar disease was often caused by the adventitious infection of pigs with vaccinia virus, which was used to vaccinate humans against smallpox. Since the eradication of smallpox and the subsequent suspension of vaccination, swinepox is caused exclusively by a host-specific poxvirus. However, vaccinia- like viruses continue to circulate in Brazil and have the potential to infect pigs.21
A low prevalence of disease occurs in all countries where pigs are raised, although it may be high in individual herds.10, 12, 18, 24
Aetiology
The swine pox virus belongs to the genus Suipox and the family Poxviridae (see General Introduction Poxviridae).The genome of the swinepox virus is 146,454 bp and contains 150 putative genes of which 146 are poxvirus homologues.1 Morphologically, swinepox virus resembles vaccinia virus, virions measuring approximately 310 × 240 nm.7 Immunodiffusion and immunoelectrophoresis studies demonstrated common antigens between these two viruses.10 Different monoclonal antibodies that developed against swinepox virus were generally swinepox- specific, however, two monoclonal antibodies cross- reacted with vaccinia, cowpox, ectromelia and rabbit fibroma viruses.23
Swinepox virus is very resistant to adverse environmental influences and survives for 12 days at 37 °C. Scabs retain their infectivity for up to one year at room temperature.14, 17
Propagation of swinepox virus is difficult and serial passaging in swine cell cultures is usually necessary before cytopathic effects (intracytoplasmic inclusions one to two days after inoculation, nuclear vacuolation and margination of the chromatin) occur. Destruction of the monolayer occurs five to seven days after inoculation.8 Swinepox virus does not multiply on the chorioallantoic membrane of embryonated hen’s eggs.
Epidemiology
Pigs are the only animal susceptible to infection with swinepox virus.10, 16, 25
Transmission of the virus is usually by contact between acutely infected and susceptible animals. Lice (Haematopinus suis) are thought to transmit the disease mechanically and may influence the severity and spread of the disease by damaging the skin.18 Lice may be infectious for weeks or months, but vertical transmission of the virus within the louse population has not been proven. Other biting insects, especially flies, such as Stomoxys spp., may also transmit the virus mechanically.9, 18
Infected animals shed virus in saliva and lachrymal fluid towards the end of the incubation period. Virus is also shed from pox lesions in the later stages of the disease. Scabs and crusts on the animal or in the environment remain infectious for many months.
In older sows, only a few lesions are usually present and these may be overlooked as the animals do not show other clinical signs. Piglets may escape infection in some outbreaks of swinepox as a result of having acquired colostral antibody.2
Recovered pigs have a solid, usually life-long, immunity. A characteristic feature of swinepox is the slow rate at which antibody appears in the circulation. Virus neutralizing and precipitating antibodies are detectable from about Day 20 after infection and titres peak at four to six weeks.16 Cell- mediated immunity is detectable two weeks after infection.
Swinepox in a herd may increase the hazard of failure of classical swine fever (hog cholera) vaccination,4 and it is believed that other diseases (mycoplasmal pneumonia, salmonellosis) are more severe in affected animals.14
Pathogenesis, clinical signs and pathology
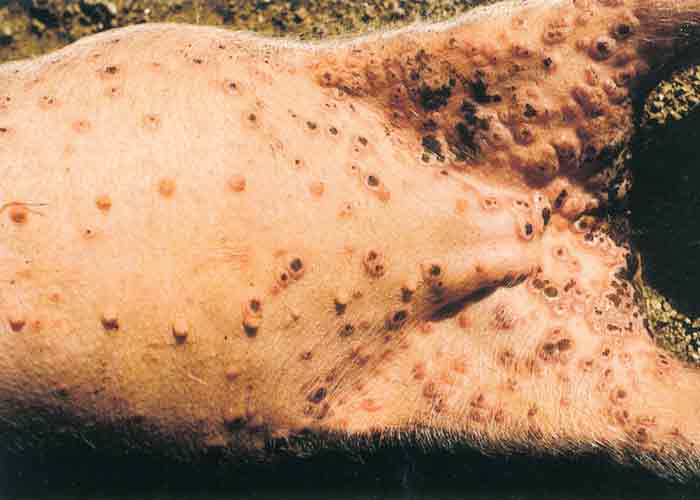
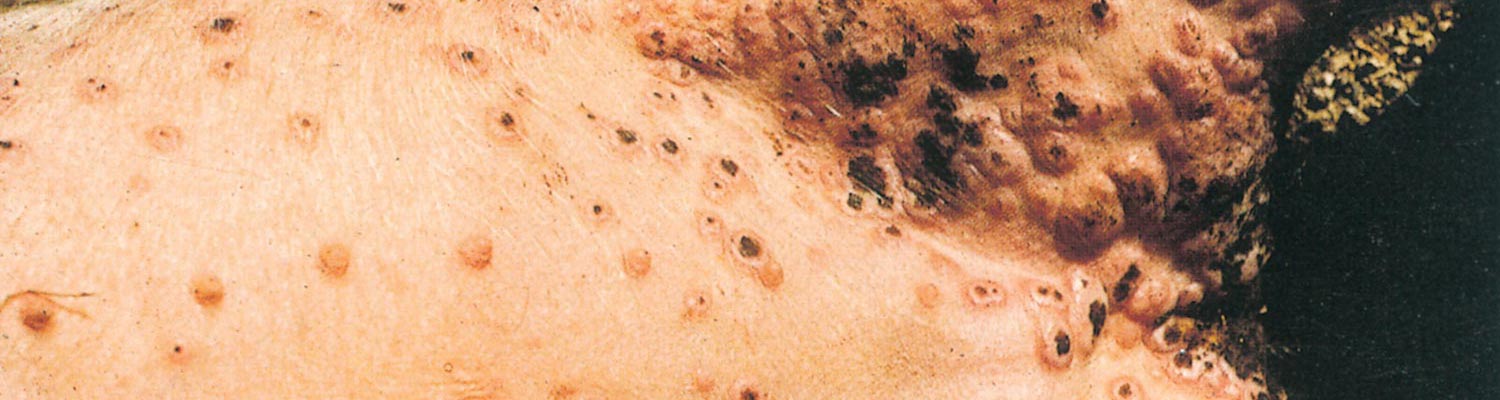
Swinepox virus has a particular affinity for cells of the epidermis, and in severe cases epithelial cells of the upper digestive and respiratory tracts may also be affected. The incubation period varies from 4 to 14 days. Initial signs include low-grade fever, anorexia and dullness, after which flat macules appear on the skin, predominantly on the inner aspects of the proximal part of the limbs (Figure 1), ears, snout and ventral abdomen. These reddish areas develop into papules and then into umbilicated pustules.
The vesicular stage is usually not seen. After six to eight days the pustules dry and scabs with depressed centres are formed, still surrounded by a raised and inflamed zone. Dark- brown scabs, 10 to 20 mm in diameter, give affected animals a spotted appearance after about 10 to 14 days. Pox lesions develop successively so that not all are at the same stage of development.18 After about three weeks the crusts are shed without leaving a scar. The number of lesions on individual animals varies a great deal and seems to be directly related to the severity of louse infestation.
Sows suckling affected piglets may suffer from teat and udder lesions so that nursing becomes painful, and piglets may even die as a result of starvation. Congenital cases of swinepox have been reported.5
Few pigs die of uncomplicated swinepox but the morbidity rate in individual herds may be high and young animals may have poor growth rates. Secondary bacterial infections occasionally cause cellulitis or abscess formation, which may result in high mortality rates.22 Self-inflicted wounds may develop as the result of irritation and subsequent scratching and rubbing.
Microscopically, hyperplasia, ballooning and swelling of keratinocytes in the Malpighian layer and large eosinophilic cytoplasmic inclusion bodies occur in affected cells. Cell nuclei show marked vacuolation. Transmission electron microscopy (TEM) reveals virions and the formation of fibrillar structures in the cytoplasm and nuclear vacuoles.13
Diagnosis and differential diagnosis
Typical pox lesions together with louse infestation are indicative of the disease. Histopathologically cytoplasmic inclusion bodies can be detected in the keratinocytes in skin lesions. Swinepox virions are easily detectable by TEM of negatively- stained preparations of material of the lesions but cannot be differentiated morphologically from vaccinia virions.
Swinepox must be differentiated from skin infections caused by vaccinia virus, Staphylococcus and Streptococcus spp. Lesions caused by mites (especially Demodex) and fungi, such as dermatophytes and ptyriasis rosea, should also be taken into consideration.
Isolation of swinepox can be performed using skin lesion homogenates on PK-15 cells although several passages are required to observe cytopathic effects.19
A PCR targeting the DNA polymerase and DNA topoisomerase I genes of members of the subfamily Chordopoxvirinae followed by DNA sequencing to identify the specific poxvirus can be used to identify swinepox virus.6, 20 The P42 gene of swinepox virus which is the homologue of the vaccinia virus P37 gene3 can also be used to identify swinepox using PCR to amplify the gene followed by sequencing the PCR product to confirm the identity of swinepox virus.19 A duplex PCR has been developed to detect swinepox and vaccinia viruses in skin lesions of pigs.21
Control
There is no specific treatment or prophylactic immunization available, although attempts at immunizing pigs with culture-adapted virus have produced promising results.14
Swinepox virus has been experimentally evaluated as a vaccine vector for several different swine diseases including porcine epidemic diarrhoea27 classical swine fever15 and swine influenza.26
Infected sows and their litters should be isolated to prevent further spread and their pens should be disinfected with, for example, 3 to 4 per cent iodophor solutions.11 Control of lice is the single most important prophylactic measure.
References
- AFONSO, C., TULMAN, E. R., LU, Z., ZSAK, L., OSORIO, F. A., BALINSKY, C., KUTISH, G. F. & ROCK, D. L., 2002. The genome of swinepox virus. Journal of Virology, 76, 783-90.
- BACHMANN, P. A., GEDEK, B., MAHNEL, H., MAYR, A. & SCHELS, H., 1984. Poxviridae. In: MAYR, A., (ed.). Rolle/ Mayr: Medizinische Mikrobiologie, Infektions- und Seuchenlehre, 5th edn. Stuttgart: Enke-Verlag.
- BÁRCENA, J., LORENZO, M. M., SÁNCHEZ-PUIG, J. M. & BLASCO, R., 2000. Sequence and analysis of a swinepox virus homologue of the vaccinia virus major envelope protein P37 (F13L). Journal of General Virology, 81, 1073-85.
- BLOOD, D. C., RADOSTITS, O. M. & HENDERSON, J. A., 1985. Viral diseases characterized by skin lesions. In: Veterinary Medicine, 6th edn, London: Bailliere Tindall.
- BORST, G., KIMMAN, T. G., GIELKENS, A. L. & VAN DER KAMP, J. S., 1990. Four sporadic cases of congenital swinepox. The Veterinary Record, 127, 61-3.
- BRACHT, A., BRUDEK, R. L., EWING, R. Y., MANIRE, C. A., BUREK, K. A., ROSA, C., BECKMEN, K. B., MARUNIAK, J. E. & ROMERO, C. H., 2006. Genetic identification of novel poxviruses of cetaceans and pinnipeds. Archives of Virology, 151, 423-38.
- CHEVILLE, N. F., 1966. Immunofluorescent and morphological studies on swinepox. Pathologia Veterinaria, 3, 556–564.
- CONROY, J. D. & MEYER, R. C., 1971. Electron microscopy of swinepox virus in germfree pigs and in cell culture. American Journal of Veterinary Research, 32, 2021–2032.
- COPLAND, J. W., 1974. Swinepox in Papua New Guinea. Tropical Animal Health Production, 6, 153–157.
- DE BOER, G. F., 1975. Swinepox. Virus isolation, experimental infections and the differentiation from vaccinia virus infections. Archives of Virology, 49, 141–150.
- EVANS, D. H., STUART, P. & ROBERTS, D. H., 1977. Disinfection of animal viruses. British Veterinary Journal, 133, 356–359.
- FALLON, G. R., 1992. Swinepox in pigs in northern Western Australia. The Veterinary Journal, 69, 233.
- KIM, U. H., MUKHAJONPAN, V., NII, S. & KATO, S., 1977. Ultrastructural study of swinepox and orf viruses. Biken Journal, 20, 57–67.
- KUBIN, G., 1972. Immunisierung der Schweine gegen Schweinepocken mit kulturadaptiertem virus. Wiener Tierarztliche Monatsschrift, 59, 317–319.
- LIN, H., MA, Z., CHEN, L. & FAN, H., 2017. Recombinant Swinepox Virus Expressing Glycoprotein E2 of Classical Swine Fever Virus Confers Complete Protection in Pigs upon Viral Challenge. Frontiers in Veterinary Science, 4:81.
- MAYR, A., 1959. Experimentelle Untersuchungen über das Virus der originaren Schweinepocken. Archiv für die Gesamte Virusforschung, IX, 156–192.
- MAYR, A., EISSNER, G. & MAYR-BIBRACK, B., 1984. Handbuch der Schutzimpfungen in der Tiermedizin. Berlin, Hamburg: Verlag Paul Parey.
- MAYR, A. N., NEUBRAND, K. & MAHNEL, H., 1966. Seuchenhaftes Auftreten von originaren Schweinepocken in Bayern. Tierarztliche Umschau, 21F, 1–6.
- MECH, P., BORA, D. P., NEHER, S., BARMAN, N. N., BORAH, P., TAMULY, S., DUTTA, L. J. & DAS, S. K., 2018. Identification of swinepox virus from natural outbreaks in pig population of Assam. Virus Disease, 29, 395-399.
- MEDAGLIA, M., PEREIRA ADE, C., FREITAS, T. R. & DAMASO, C. R., 2011. Swinepox virus outbreak, Brazil, 2011. Emerging Infectious Diseases, 17, 1976-8.
- MEDAGLIA, M., SÁ, N. M. B., CORREA, I. A., COSTA, L. J. & DAMASO, C. R., 2015. One-step duplex polymerase chain reaction for the detection of swinepox and vaccinia viruses in skin lesions of swine with poxvirus-related disease. Journal of Virological Methods, 219, 10-13.
- MILLER, R. B. & OLSON, L. D., 1978. Epizootic of concurrent cutaneous streptococcal abscesses and swinepox in a herd of swine. Journal of the American Veterinary Medical Association, 172, 672–680.
- OUCHI, M., FUJIWARA, M., HATANO, Y., YAMADA, M. & NII, S., 1992. Analysis of swinepox virus antigens using monoclonal antibodies. Journal of Veterinary Medical Science, 54, 731-7.
- RIYESH, T., BARUA, S., KUMAR, N., JINDAL, N., BERA, B. C., NARANG, G., MAHAJAN, N. K., ARORA, D., ANAND, T., VAID, R. K., YADAV, M., CHANDEL, S. S., MALIK, P., TRIPATHI, B. N. & SINGH, R. K., 2016. Isolation and genetic characterization of swinepox virus from pigs in India. Comparative Immunology, Microbiology and Infectious Diseases, 46, 60-5.
- SHOPE, R. E., 1940. Swine pox. Archiv für die Gesamte Virusforschung, 1, 457–467.
- XU, J., HUANG, D., XU, J., LIU, S., LIN, H., ZHU, H., LIU, B., CHEN, W. & LU, C., 2013. Immune responses and protective efficacy of a recombinant swinepox virus co-expressing HA1 genes of H3N2 and H1N1 swine influenza virus in mice and pigs. Veterinary Microbiology, 162, 259-64.
- YUAN, X., LIN, H. L. B., HE, K. & FAN, H., 2017. Efficacy and immunogenicity of recombinant swinepox virus expressing the truncated S protein of a novel isolate of porcine epidemic diarrhea virus. Archives of Virology, 162, 3779-3789.