- Infectious Diseases of Livestock
- Part 2
- Enteric caliciviruses of pigs and cattle
- GENERAL INTRODUCTION: PARAMYXOVIRIDAE AND PNEUMOVIRIDAE
- Rinderpest
- Peste des petits ruminants
- Parainfluenza type 3 infection
- Bovine respiratory syncytial virus infection
- Hendra virus infection
- Paramyxovirus-induced reproductive failure and congenital defects in pigs
- Nipah virus disease
- GENERAL INTRODUCTION: CALICIVIRIDAE AND ASTROVIRIDAE
- Vesicular exanthema
- Enteric caliciviruses of pigs and cattle
- GENERAL INTRODUCTION: RETROVIRIDAE
- Enzootic bovine leukosis
- Jaagsiekte
- Visna-maedi
- Caprine arthritis-encephalitis
- Equine infectious anaemia
- GENERAL INTRODUCTION: PAPILLOMAVIRIDAE
- Papillomavirus infection of ruminants
- Papillomavirus infection of equids
- GENERAL INTRODUCTION: ORTHOMYXOVIRIDAE
- Equine influenza
- Swine influenza
- GENERAL INTRODUCTION: CORONAVIRIDAE
- Porcine transmissible gastroenteritis
- Porcine respiratory coronavirus infection
- Porcine epidemic diarrhoea
- Porcine haemagglutinating encephalomyelitis virus infection
- Porcine deltacoronavirus infection
- Bovine coronavirus infection
- Ovine coronavirus infection
- Equine coronavirus infection
- GENERAL INTRODUCTION: PARVOVIRIDAE
- Porcine parvovirus infection
- Bovine parvovirus infection
- GENERAL INTRODUCTION: ADENOVIRIDAE
- Adenovirus infections
- GENERAL INTRODUCTION: HERPESVIRIDAE
- Equid herpesvirus 1 and equid herpesvirus 4 infections
- Equid gammaherpesvirus 2 and equid gammaherpesvirus 5 infections
- Equine coital exanthema
- Infectious bovine rhinotracheitis/infectious pustular vulvovaginitis and infectious pustular balanoposthitis
- Bovine alphaherpesvirus 2 infections
- Malignant catarrhal fever
- Pseudorabies
- Suid herpesvirus 2 infection
- GENERAL INTRODUCTION: ARTERIVIRIDAE
- Equine viral arteritis
- Porcine reproductive and respiratory syndrome
- GENERAL INTRODUCTION: FLAVIVIRIDAE
- Bovine viral diarrhoea and mucosal disease
- Border disease
- Hog cholera
- Wesselsbron disease
- Louping ill
- West nile virus infection
- GENERAL INTRODUCTION: TOGAVIRIDAE
- Equine encephalitides caused by alphaviruses in the Western Hemisphere
- Old World alphavirus infections in animals
- Getah virus infection
- GENERAL INTRODUCTION: BUNYAVIRIDAE
- Diseases caused by Akabane and related Simbu-group viruses
- Rift Valley fever
- Nairobi sheep disease
- Crimean-Congo haemorrhagic fever
- GENERAL INTRODUCTION: ASFARVIRIDAE
- African swine fever
- GENERAL INTRODUCTION: RHABDOVIRIDAE
- Rabies
- Bovine ephemeral fever
- Vesicular stomatitis and other vesiculovirus infections
- GENERAL INTRODUCTION: REOVIRIDAE
- Bluetongue
- Ibaraki disease in cattle
- Epizootic haemorrhagic disease
- African horse sickness
- Equine encephalosis
- Palyam serogroup orbivirus infections
- Rotavirus infections
- GENERAL INTRODUCTION: POXVIRIDAE
- Lumpy skin disease
- Sheeppox and goatpox
- Orf
- Ulcerative dermatosis
- Bovine papular stomatitis
- Pseudocowpox
- Swinepox
- Cowpox
- Horsepox
- Camelpox
- Buffalopox
- GENERAL INTRODUCTION: PICORNAVIRIDAE
- Teschen, Talfan and reproductive diseases caused by porcine enteroviruses
- Encephalomyocarditis virus infection
- Swine vesicular disease
- Equine picornavirus infection
- Bovine rhinovirus infection
- Foot-and-mouth disease
- GENERAL INTRODUCTION: BORNAVIRIDAE
- Borna disease
- GENERAL INTRODUCTION: CIRCOVIRIDAE AND ANELLOVIRIDAE
- Post-weaning multi-systemic wasting syndrome in swine
- GENERAL INTRODUCTION: PRION DISEASES
- Scrapie
- Bovine spongiform encephalopathy
- Transmissible spongiform encephalopathies related to bovine spongiform encephalopathy in other domestic and captive wild species
Enteric caliciviruses of pigs and cattle
This content is distributed under the following licence: Attribution-NonCommercial CC BY-NC  View Creative Commons Licence details here
View Creative Commons Licence details here
Enteric caliciviruses of pigs and cattle
N J KNOWLES AND I N CLARKE
Introduction and aetiology
A range of viruses has been associated with enteric disease in farm animals. Viruses resembling caliciviruses were first described in cattle in 19781, 31 and in pigs in 1980.23 Woode and Bridger (1978)31 in pioneering work examined diarrhoeic faecal samples from three independent UK cattle herds: viruses were found in faecal samples from a calf in each herd. Each sample contained small round viruses (SRVs) based on their appearance under the transmission electron microscope (TEM), but a number of other different viruses including rotaviruses (sample 1) and coronaviruses (sample 2) were also present. The presence of several viruses in calf diarrhoeic faecal samples is not uncommon but it does cause significant problems with establishing an aetiological link between the presence of the virus and the cause of the disease.
During the course of experiments in calves with the samples collected it was also realized that all three original faecal specimens harboured two morphologically distinct SRVs; in each case they contained both calici-like viruses and astroviruses. Attempts to grow the calici-like virus in cell culture were unsuccessful, and in order to separate the different SRVs, the calici-like virus in sample 1 (now known as Newbury agent 1, or NA1) was separated from the astrovirus by infecting a gnotobiotic calf with astrovirus and then by challenging the previously infected animal orally with a faecal filtrate containing the mixture of SRVs. Faeces from the challenged calf were used in oral serial passage of another four calves. Only the calici-like virus particles (NA1) were observed in the faeces of these calves. Further passage of sample 2 in gnotobiotic calves lost the astrovirus, leaving a small round virus which was identified as SRV2a as the sole cause of disease. It is now renamed Newbury agent 2 (NA2).
There were some subtle differences in the morphology of NA1 and NA2, but neither virus exhibited the clear morphological characteristics of the ‘classic’ caliciviruses seen in other animal species. It was later realized that the Newbury agents most closely resemble the human Norwalk virus.2 In a separate study of diarrhoeal disease in calves, viruses similar to the human prototype Norwalk virus were observed in the faeces of new-born calves in Thuringia, Germany.12 Other German workers also observed Norwalk-like viruses (NLVs) in the faeces of calves.9 Whilst all these observations were made in the late 1970s and 1980s it was not until the late 1990s that the first complete genome sequence was published for a German virus — the Jena virus (JV).18 This showed unequivocally that JV belonged to the Caliciviridae. Phylogenetic analysis indicated that JV is most closely related to the human NLVs. Jena virus has a similar genome organization to Norwalk virus, and the JV genome encodes three open reading frames (ORFs), a large ORF (ORF 1) that encodes the polyprotein, ORF 2 the capsid protein, and a 3’ terminal ORF (ORF 3) that encodes a small basic protein that has been speculated in other caliciviruses to have a structural role. Partial sequence of the 3’ terminal third of NA2 revealed a similar genome organization.5
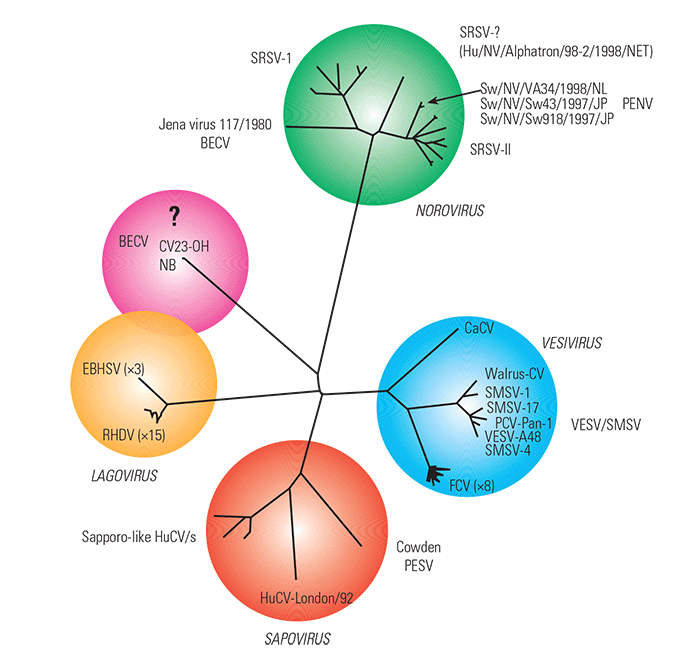
The family Caliciviridae comprises four genera, Vesivirus, Lagovirus, Norovirus (Norwalk-like viruses) and Sapovirus (Sapporo-like viruses).10 The human Norwalk-like viruses (often referred to as Small Round Structured Viruses, or SRSVs) have been divided into two genotypes, I and II. The bovine enteric caliciviruses (BECVs) which have so far been characterized by nucleotide sequencing are JV,18 NA25, NB and CV23-OH24 and 14 sequences of a calicivirus amplified from faecal samples in the Netherlands.27 With the exception of NB and CV23-OH, which appear to belong to a previously undescribed novel genus 24 (Figure 57.1), all are closely related to each other and belong to the genus Norovirus. They are more closely related to the genotype I SRSVs than to those belonging to genotype II (Figure 57.1). Porcine (or swine) enteric caliciviruses also fall into two distinct groups, one belonging to the Norovirus genus25, 27 and the other to the Sapovirus genus.13, 27, 28 To distinguish more easily between the porcine enteric caliciviruses belonging to the two genera, they are referred to below as porcine enteric noroviruses (PENVs) and porcine enteric sapoviruses (PESVs).
Figure 57.1 Unrooted neighbour-joining tree based on a comparison of the complete capsid sequences of members of the four calicivirus genera, Vesivirus, Lagovirus, Norovirus and Sapovirus, and a newly described distinct bovine enteric calicivirus. (NB: Amino acid sequences were obtained from the EMBL/GenBank databases, aligned and the tree constructed using Clustal X.26 The tree was drawn using TreeView.19) Abbreviations: BECV, bovine enteric calicivirus; CaCV, canine calicivirus; EBHSV, European brown hare syndrome virus; FCV, feline calicivirus; HuCV, human calicivirus; PCV, primate calicivirus; PENV, porcine enteric norovirus; PESV, porcine enteric sapovirus; RHDV, rabbit haemorrhagic disease virus; SMSV, San Miguel sealion virus; SRSV, small round structured virus; VESV, vesicular exanthema of swine virus
It is now clear that BECVs cause enteric disease in calves, the ensuing disease being of similar severity to that caused by rotaviruses. However, the role of PENV and PESV as aetiological agents of disease in pigs is less clear. In contrast to the rotaviruses, the low numbers of these viruses that are shed during infection and the lack of a cell culture system for growing them have severely hampered progress in understanding the disease they cause.
Epidemiology
Diarrhoeal disease in calves is economically important and losses occur due to mortality, poor growth and veterinary costs.29, 30 The role of enteric caliciviruses in calf diarrhoea has not been evaluated and our knowledge is therefore restricted to only a few studies. New-born calves are often simultaneously infected by a number of pathogens (bacteria, viruses and protozoa), making it difficult to establish a causal relationship between clinical signs and the presence of the pathogen in the faeces. In the case of BECVs the primary route of infection has been shown experimentally to be faecal–oral.31 From the limited surveys that have been conducted it is clear that BECVs are commonly found in new-born calves. The most detailed study involved TEM examination of faecal specimens from calves with diarrhoea from 43 outbreaks, 11 (26 per cent) of which were positive for calicivirus.22 Whilst other viruses were detected with higher frequency (rotavirus, 93 per cent; coronavirus, 56 per cent) it is likely that the number of calicivirus infections was under-reported because of the low numbers of viruses shed, the short shedding period, and the difficulty of making a definitive diagnosis by TEM based on virus morphology.
It seems likely that there are multiple ‘types’ of BECVs. Cross-challenge experiments with NA1 and NA2 indicated that prior infection with either virus protects from later challenge with homologous virus but does not protect against challenge with heterologous virus.3 Phylogenetic analysis of the NA2 and JV capsid protein suggests that these viruses are as different from each other as are individual human NLVs. Furthermore, the recent biochemical characterization of NA1 confirmed that it is antigenically unrelated to NA2.6 Further studies are required using recombinant antigens to establish seroprevalence and to determine the nature and extent of virus diversity by reverse transcription polymerase chain reaction (RT-PCR).
Even less is known about the prevalence of either PENV or PESV. In one RT-PCR study in veal calves on Dutch farms, 44 per cent of the specimens yielded bovine enteric calicivirus sequences, while only 2 per cent of specimens from pig farms were found to contain PENV sequences.27
Pathogenesis, clinical signs and pathology
Our knowledge of the disease caused by BECVs comes primarily from the infection experiments conducted with NA1 and NA2 in gnotobiotic calves11, 31 and from a preliminary study on JV in new-born calves.2, 3, 4, 16 From these studies it is clear that the pathology of BECV infection is as severe as that caused by rotaviruses. Oral inoculation of gnotobiotic calves frequently gave rise to disease and all animals displayed some clinical signs as measured by increased faecal output (greater than 500 g per day), changes in faecal colour (compared to pre-inoculation faeces), anorexia, reduced xylose absorption and the presence of caliciviruses in the faeces.3 All new-born calves infected with JV contracted diarrhoea and every calf excreted the virus in its faeces.11
The lesions caused by NA1 were investigated in detail in a study conducted in 1984.16 Fourteen gnotobiotic calves were infected and then killed and necropsied at intervals between 0,5 and 10 days post-infection. There was no evidence of infection in the abomasum or duodenum. Nine short regions of small intestine were taken at equally spaced locations for further investigation of the infection. A variety of changes were observed, the severity of the cellular damage varying considerably from calf to calf. Lesions were observed in the cranial half of the small intestine. The first change was separation of microvilli caused by enterocyte swelling. By 0,75 day post-infection degeneration and exfoliation of the enterocytes on the sides of villi were seen. At one day post-infection the lesions were most severe with entire villi exhibiting a range of pathological changes. No viruses were observed in any of the cells examined by TEM. However, immuno-staining with convalescent serum to NA1, whilst faint, was intracytoplasmic and limited to individual enterocytes. Infected cells were most frequently detected in sites 1 to 3 of the cranial small intestine and were present from 0,5 to 2 days post-infection. Virus first appeared in the intestinal contents at 0,75 days and could be detected until day three.
The small intestine crypts were deepened throughout. In the cranial part of the small intestine villi were severely stunted by day one but had returned to their normal size by seven to ten days post-infection. It also appeared that the villus height increased in the caudal small intestine and this was postulated to be a compensation for the loss of enterocytes in the more cranial part of the small intestine.
The PESV strain, Cowden, is unique among the enteric caliciviruses in having been adapted to grow in primary porcine kidney cells7 and continuous porcine kidney cell lines20 by incorporating intestinal contents of uninfected gnotobiotic pigs into the cell culture medium. In one study experimental infection of pigs with a wild-type (WT) Cowden strain resulted in profuse diarrhoea, anorexia, and intestinal lesions,8 while in another only moderate diarrhoea was produced.14
However, the tissue culture-adapted (TC) Cowden strain failed to produce diarrhoea.14 Differences in the degree of villous atrophy in the duodenum, jejenum and ileum of the WT and TC Cowden strains suggested that adaptation to cell cultures had resulted in a reduction in virulence.14 Differences in the capsid amino acid sequences of the WT and TC strains were postulated as being potentially responsible for the observed phenotypic changes.13, 14
Diagnosis
Transmission electron microscopy remains the only reliable method for the detection of enteric viruses. However, caliciviruses can be difficult to detect because they are shed in very low numbers and for only relatively short periods. Moreover, the lack of the distinctive surface structures of the classic caliciviruses gives the BECVs an amorphous appearance and makes them difficult to identify.
The close phylogenetic relationship between some of the BECVs and the human NLVs means that it has been possible to use ‘generic’ RT-PCR primers based on the human NLV sequences to amplify fragments of theBECVgenome. Initially, a small DNA fragment from the JV RNA polymerase gene was amplified and by using this information it was possible to obtain the primary sequence data for this bovine enteric calicivirus. 18 Thus RT-PCR is a useful alternative to TEM although little is known about sequence diversity in the bovine and porcine enteric caliciviruses and primers may have to be perfected as more sequence data become available. Serological methods to detect bovine and porcine enteric caliciviruses in faecal specimens have yet to be developed.
It is not known whether enteric caliciviruses from animals can infect humans. Experimental infections of a number of different animal species with the human Norwalk virus have failed to induce disease.17
Control
The faecal–oral spread of bovine and porcine enteric caliciviruses together with other enteric viruses and the apparent endemic nature of these infections offers little hope of control through improved farmyard hygiene. The fact that prior infection offers protection from infection with the same virus is in contrast to the situation in humans and suggests that a vaccine may be a possibility. It is not known, however, how many different ‘types’ of virus exist and in the production of any vaccine this would have to be taken into account. It should be possible to obtain expression of the capsid proteins of BECV and PENV as virus-like particles (VLPs), as has been achieved with the NLVs21 and PESV,15 and these could be produced in plants for incorporation into animal feed.
References
- ALMEIDA, J.D., CRAIG, C.R. & HALL, T.E., 1978. Multiple viruses present in the faeces of a scouring calf. The Veterinary Record, 102, 170–171.
- BRIDGER, J.C. & HALL, G.A., 1979. Effects of a calicivirus-like agent (The Newbury Agent) on gnotobiotic calves. Inserm, 90, 233–236.
- BRIDGER, J.C., HALL, G.A. & BROWN, J.F., 1984. Characterization of a calici-like virus (Newbury agent) found in association with astrovirus in bovine diarrhea. Infection and Immunity, 43, 133–138.
- BRIDGER, J.C., HALL, G.A., REYNOLDS, D.J. & BROWN, J.F., 1983. Calici-like viruses in calf diarrhoea. Proceedings of the Fourth International Symposium on Neonatal Diarrhoea. pp. 155–159.
- DASTJERDI, A.M., GREEN, J., GALLIMORE, C.I., BROWN, D.W.G. & BRIDGER, J.C., 1999. The bovine Newbury agent-2 is genetically more closely related to human SRSVs than to animal caliciviruses. Virology, 254, 1–5.
- DASTJERDI, A.M., SNODGRASS, D.R. & BRIDGER, J.C., 2000. Characterisation of the bovine enteric calici-like virus, Newbury agent 1. FEMS Microbiology Letters, 192, 125–131.
- FLYNN, W.T. & SAIF, L.J., 1988. Serial propagation of porcine enteric calicivirus-like virus in primary porcine kidney cell cultures. Journal of Clinical Microbiology, 26, 206–212.
- FLYNN, W.T., SAIF, L.J. & MOORHEAD, P.D., 1988. Pathogenesis of porcine enteric calicivirus-like virus in four-day-old gnotobiotic pigs. American Journal of Veterinary Research, 49, 819–825.
- GRANZOW, H. & SCHIRRMEIER, H., 1985. Identification of 32 nm viruses in faeces of diarrhoeic calves by electron microscopy. Monatshefte Veterinarmed, 40, 228–229.
- GREEN, K.Y., ANDO, T., BALAYAN, M.S., BERKE, T., CLARKE, I.N., ESTES, M.K., MATSON, D.O., NAKATA, S., NEILL, J.D., STUDDERT, M.J. & THIEL, H.J., 2000. Taxonomy of the caliciviruses. Journal of Infectious Diseases, 181 Supplement 2, S322–330.
- GÜNTHER, H. & OTTO, P., 1987. Studies into diarrhoea of young calves. Seventh Communication: ‘Zackenvirus’ (Jena-Agens 117/80) — A new diarrhoea pathogen to calf. Archives of Experimental Veterinary Medicine, Leipzig, 41, 934–938.
- GÜNTHER, H., OTTO, P. & HEILMAN, P., 1984. Studies into diarrhoea of young calves. Sixth communication: Detection and determination of pathogenicity of a bovine Corona virus and an undefined icosahedric virus. Archives of Experimental Veterinary Medicine, Leipzig, 38, 781–792.
- GUO, M., CHANG, K.O., HARDY, M.E., ZHANG, Q., PARWANI, A.V. & SAIF, L.J., 1999. Molecular characterization of a porcine enteric calicivirus genetically related to Sapporo-like human caliciviruses. Journal of Virology, 73, 9625–9631.
- GUO, M., HAYES, J., CHO, K.O., PARWANI, A.V., LUCAS, L.M. & SAIF, L.J., 2001. Comparative pathogenesis of tissue culture-adapted and wild-type Cowden porcine enteric calicivirus (PEC) in gnotobiotic pigs and induction of diarrhea by intravenous inoculation of wild-type PEC. Journal of Virology, 75, 9239–9251.
- GUO, M., QIAN, Y., CHANG, K.O. & SAIF, L.J., 2001. Expression and self-assembly in baculovirus of porcine enteric calicivirus capsids into virus-like particles and their use in an enzyme-linked immunosorbent assay for antibody detection in swine. Journal of Clinical Microbiology, 39, 1487–1493.
- HALL, G.A., BRIDGER, J.C., BROOKER, B.E., PARSONS, K.R. & ORMEROD, E., 1984. Lesions of gnotobiotic calves experimentally infected with a calicivirus-like (Newbury) agent. Veterinary Pathology, 21, 208–215.
- KAPIKIAN, A.Z., 1994. Norwalk and Norwalk-like Viruses. In: kapikian, a.z. (ed.). Viral Infections of the Gastrointestinal Tract. New York: Marcel Dekker, Vol. 10, pp. 471–518.
- LIU, B.L., LAMBDEN, P.R., GÜNTHER, H., OTTO, P., ELSCHNER, M. & CLARKE, I.N., 1999. Molecular characterization of a bovine enteric calicivirus: Relationship to the Norwalk-like viruses. Journal of Virology, 73, 819–825.
- PAGE, R.D.M., 1996. Treeview: An application to display phylogenetic trees on personal computers. Computer Applications in the Biosciences, 12, 357–358.
- PARWANI, A.V., FLYNN, W.T., GADFIELD, K.L. & SAIF, L.J., 1991. Serial propagation of porcine enteric calicivirus in a continuous cell line. Effect of medium supplementation with intestinal contents or enzymes. Archives of Virology, 120, 115–122.
- PRASAD, B.V.V., HARDY, M.E. & ESTES M.K., 2000. Structural studies of recombinant Norwalk capsids. Journal of Infectious Diseases, 181 (Supplement 2), S317–321.
- REYNOLDS, D.J., MORGAN, J.H., CHANTER, N., JONES, P.W., BRIDGER, J.C., DEBNEY, T.G. & BUNCH, K.J., 1986. Microbiology of calf diarrhoea in southern Britain. The Veterinary Record, 119, 34–39.
- SAIF, L.J., BOHL, E.H., THEIL, K.W., CROSS, R.F. & HOUSE, J.A., 1980. Rotavirus-like, calicivirus-like, and 23-nm virus-like particles associated with diarrhoea in young pigs. Journal of Clinical Microbiology, 12, 105–111.
- SMILEY, J.R., CHANG, K.O., HAYES, J., VINJE, J. & SAIF, L.J., 2002. Characterization of an enteropathogenic bovine calicivirus representing a potentially new calicivirus genus. Journal of Virology, 76, 10089–10098.
- SUGIEDA, M., NAGAOKA, H., KAKISHIMA, Y., OHSHITA, T., NAKAMURA, S. & NAKAJIMA, S., 1998. Detection of Norwalk-like virus genes in the caecum contents of pigs. Archives of Virology, 143, 1215–1221.
- THOMPSON, J.D., GIBSON, T.J., PLEWNIAK, F., JEANMOUGIN, F. & HIGGINS, D.G., 1997. The Clustal-X windows interface: Flexible strategies for multiple sequence alignment aided by quality analysis tools. Nucleic Acids Research, 25, 4876–4882.
- VAN DER POEL, W.H., VINJE, J., VAN DER HEIDE, R., HERRERA, M.I., VIVO, A. & KOOPMANS, M.P., 2000. Norwalk-like calicivirus genes in farm animals. Emerging Infectious Diseases, 6, 36–41.
- VINJE, J. & KOOPMANS, M.P.G., 2000. Simultaneous detection and genotyping of ‘Norwalk-like viruses’ by oligonucleotide array in a reverse line blot hybridization format. Journal of Clinical Microbiology, 38, 2595–2601.
- WOODE, G.N., 1976. Viral diarrhoea in calves. Veterinary Annual, 16, 30–34.
- WOODE, G.N. & BRIDGER, J.C., 1975. Viral enteritis of calves. The Veterinary Record, 96, 85–88.
- WOODE, G.N. & BRIDGER, J.C., 1978. Isolation of small viruses resembling astroviruses and caliciviruses from acute enteritis of calves. Journal of Medical Microbiology, 11, 441–452.