- Infectious Diseases of Livestock
- Part 2
- Rift Valley fever
- GENERAL INTRODUCTION: PARAMYXOVIRIDAE AND PNEUMOVIRIDAE
- Rinderpest
- Peste des petits ruminants
- Parainfluenza type 3 infection
- Bovine respiratory syncytial virus infection
- Hendra virus infection
- Paramyxovirus-induced reproductive failure and congenital defects in pigs
- Nipah virus disease
- GENERAL INTRODUCTION: CALICIVIRIDAE AND ASTROVIRIDAE
- Vesicular exanthema
- Enteric caliciviruses of pigs and cattle
- GENERAL INTRODUCTION: RETROVIRIDAE
- Enzootic bovine leukosis
- Jaagsiekte
- Visna-maedi
- Caprine arthritis-encephalitis
- Equine infectious anaemia
- GENERAL INTRODUCTION: PAPILLOMAVIRIDAE
- Papillomavirus infection of ruminants
- Papillomavirus infection of equids
- GENERAL INTRODUCTION: ORTHOMYXOVIRIDAE
- Equine influenza
- Swine influenza
- GENERAL INTRODUCTION: CORONAVIRIDAE
- Porcine transmissible gastroenteritis
- Porcine respiratory coronavirus infection
- Porcine epidemic diarrhoea
- Porcine haemagglutinating encephalomyelitis virus infection
- Porcine deltacoronavirus infection
- Bovine coronavirus infection
- Ovine coronavirus infection
- Equine coronavirus infection
- GENERAL INTRODUCTION: PARVOVIRIDAE
- Porcine parvovirus infection
- Bovine parvovirus infection
- GENERAL INTRODUCTION: ADENOVIRIDAE
- Adenovirus infections
- GENERAL INTRODUCTION: HERPESVIRIDAE
- Equid herpesvirus 1 and equid herpesvirus 4 infections
- Equid gammaherpesvirus 2 and equid gammaherpesvirus 5 infections
- Equine coital exanthema
- Infectious bovine rhinotracheitis/infectious pustular vulvovaginitis and infectious pustular balanoposthitis
- Bovine alphaherpesvirus 2 infections
- Malignant catarrhal fever
- Pseudorabies
- Suid herpesvirus 2 infection
- GENERAL INTRODUCTION: ARTERIVIRIDAE
- Equine viral arteritis
- Porcine reproductive and respiratory syndrome
- GENERAL INTRODUCTION: FLAVIVIRIDAE
- Bovine viral diarrhoea and mucosal disease
- Border disease
- Hog cholera
- Wesselsbron disease
- Louping ill
- West nile virus infection
- GENERAL INTRODUCTION: TOGAVIRIDAE
- Equine encephalitides caused by alphaviruses in the Western Hemisphere
- Old World alphavirus infections in animals
- Getah virus infection
- GENERAL INTRODUCTION: BUNYAVIRIDAE
- Diseases caused by Akabane and related Simbu-group viruses
- Rift Valley fever
- Nairobi sheep disease
- Crimean-Congo haemorrhagic fever
- GENERAL INTRODUCTION: ASFARVIRIDAE
- African swine fever
- GENERAL INTRODUCTION: RHABDOVIRIDAE
- Rabies
- Bovine ephemeral fever
- Vesicular stomatitis and other vesiculovirus infections
- GENERAL INTRODUCTION: REOVIRIDAE
- Bluetongue
- Ibaraki disease in cattle
- Epizootic haemorrhagic disease
- African horse sickness
- Equine encephalosis
- Palyam serogroup orbivirus infections
- Rotavirus infections
- GENERAL INTRODUCTION: POXVIRIDAE
- Lumpy skin disease
- Sheeppox and goatpox
- Orf
- Ulcerative dermatosis
- Bovine papular stomatitis
- Pseudocowpox
- Swinepox
- Cowpox
- Horsepox
- Camelpox
- Buffalopox
- GENERAL INTRODUCTION: PICORNAVIRIDAE
- Teschen, Talfan and reproductive diseases caused by porcine enteroviruses
- Encephalomyocarditis virus infection
- Swine vesicular disease
- Equine picornavirus infection
- Bovine rhinovirus infection
- Foot-and-mouth disease
- GENERAL INTRODUCTION: BORNAVIRIDAE
- Borna disease
- GENERAL INTRODUCTION: CIRCOVIRIDAE AND ANELLOVIRIDAE
- Post-weaning multi-systemic wasting syndrome in swine
- GENERAL INTRODUCTION: PRION DISEASES
- Scrapie
- Bovine spongiform encephalopathy
- Transmissible spongiform encephalopathies related to bovine spongiform encephalopathy in other domestic and captive wild species
Rift Valley fever
This content is distributed under the following licence: Attribution-NonCommercial CC BY-NC  View Creative Commons Licence details here
View Creative Commons Licence details here
Rift Valley fever
Previous authors: R SWANEPOEL AND JAW COETZER
Current authors:
JAW Coetzer - Professor Emeritus, Faculty of Veterinary Science, University of Pretoria, Private Bag X04, Onderstepoort, Pretoria, Gauteng, South Africa
JT Paweska - Head Centre for Emerging Zoonotic and Parasitic Diseases, National Institute for Communicable Diseases, South Africa
B Bird - Virologist, One Health Institute, School of Veterinary Medicine, University of California Davis, California, United States
R Swanepoel - Extraordinary Lecturer, BVSc, Department of Veterinary Tropical Diseases, Faculty of Veterinary Science, University of Pretoria Private Bag X04, Onderstepoort, Pretoria, Gauteng, South Africa
L Odendaal - Senior Lecturer, Faculty of Veterinary Science, University of Pretoria, Private Bag X04, Onderstepoort, Pretoria, Gauteng, South Africa
J Fafetine - Virologist, Faculty of Veterinary Medicine, Eduardo Mondlane University, Maputo, Mozambique
Introduction and history
Rift Valley fever (RVF) is a peracute or acute disease that mainly affects domestic ruminants and humans caused by a mosquito-borne RNA phlebovirus.346 In domestic animals, the disease is most severe in sheep and goats, resulting in high mortality in new-born lambs and kids and high percentage of abortion in pregnant sheep, goats and sometimes cattle. The infection in humans is usually associated with mild to moderate febrile illness (i.e. fever, myalgia, arthralgia, lethargy), but can progress to severe sequelae including ocular lesions and loss of vision, encephalitis, a haemorrhagic disease with necrotic hepatitis and a high case fatality rate in a minority of patients. Humans usually become infected from contact with virus-contaminated tissues and body fluids from infected animals, but mosquito bites can also transmit the virus. Outbreaks of the disease tend to occur when particularly heavy rains favour the breeding of competent mosquito vectors.
Since original isolation in Kenya,135 RVF virus (RVFV) has been contained for decades within the African continent, but in the last 30 years emerged outside its historic boundaries593 including crossing significant geographic barriers: the Sahara desert into Egypt in 1977,319 the Indian Ocean to Madagascar in 1991,439, 440 and the Comoros Islands in 2007,349 and the Red Sea to the Arabian Peninsula in 2000/01.557 The unpredictable and sudden emergence of RVFV outside traditional endemic areas, unavailability of safe and efficacious antiviral treatment and prophylactic immunization, led the World Health Organization (WHO) to identify RVF as a priority disease for development of effective therapeutics and vaccines.414
There have been multiple reviews including Henning,258 Weiss,649 Easterday,167 Peters and Meegan,499 Shimshony and Barzilai,556 and Meegan and Bailey.410 The renewed interest in RVF as a significant zoonotic threat has been a subject of numerous reviews covering various aspects of the disease, including epidemiology, pathogenesis, control/prevention, development of new diagnostic assays, therapeutics and vaccines,7, 35, 63, 66, 74, 165, 191, 271, 274, 287, 320, 331, 337, 361, 368, 383, 405, 416, 477, 485, 486, 490, 494, 503, 522, 593 molecular biology and genomics, 76, 209, 231, 270 viral and host determinants of virulence,274 surveillance, predictive models and control strategies emphasizing the One Health concept67, 203, 246, 331, 429 and ecological, climatic and anthropogenic factors that play a role in RVF emergence, re-emergence and spread.107, 384
An acute and highly fatal disease of lambs associated with heavy rains and accompanied by reports of illness in humans was first recognized in the Rift Valley in Kenya at the turn of the century, but the causative agent was not isolated until 1930.134, 431, 579 Major outbreaks were subsequently recorded in Kenya in 1930/31, 1968, 1978/79, 1997/98, 2006/07, 2014/15 and 2019 and lesser outbreaks at irregular intervals during the intervening years.145, 146, 410, 666
The disease was first recorded in southern Africa in the early 1950s when a large epidemic occurred in the western Free State, southern Gauteng and adjacent North West and Limpopo provinces of South Africa, although it was only recognized as RVF early in 1951 when humans became ill after assisting at a necropsy of a bull near Johannesburg.15, 453 Sheep farming dominates in some of the affected areas and it was estimated that 100 000 sheep died and 500 000 aborted in the epidemic, with smaller losses occurring in cattle.542 Lesser outbreaks of the disease or sporadic isolations of virus were recorded in South Africa in 1952/53, 1955/59, 1969/71, 1981, 1996, and 2018.25, 45, 283, 392, 395, 402, 629, 649 After more than a decade with minor reported RVF activity, large outbreaks affecting most of South Africa occurred in 2009/10.30, 82, 418, 657
The diversity of clinical disease presentation in humans was recognized first in Kenya, and during the 1950/51 epidemic in South Africa, when it was recognized that RVF could be accompanied by transient loss of visual acuity and serious ocular sequelae.217, 234, 539, 540 Human deaths following natural infection were first recorded in South Africa during the epidemic in 1975 when seven patients died of encephalitis and haemorrhagic fever associated with necrotic hepatitis.232, 233, 394, 403, 633 Human deaths were also recorded during the 1978 outbreak in Zimbabwe.592 Subsequently, human deaths were recognized during outbreaks in several countries, including a large epidemic which affected the normally arid north-eastern region of Kenya and adjacent part of Somalia after heavy rains in 1997/98.666
Since 1955 extensive outbreaks of the disease occurred at irregular intervals in southern Africa including Zimbabwe,109, 560, 585, 586 Mozambique,392, 393, 502, 626 and Zambia.12, 268
Major epidemics (1977/78, 1993 and 2003) were recorded in Egypt and are summarized by Kenawy, Abdel-Hamid and Beier.319 The first time that RVF was recognized outside eastern and southern Africa was during 1977/78, when a major epidemic occurred along the Nile Delta and Valley in Egypt, causing an unprecedented number of human infections and deaths, as well as numerous deaths and abortions in sheep and cattle and some losses in goats, water buffalo (Bubalus bubalis) and camels. Estimates of the number of human cases range from 18 000 to more than 200 000 with at least 598 deaths occurring from encephalitis and/or haemorrhagic fever.408, 409, 410, 499, 556 There was a marked decline in the occurrence of RVF in Egypt after 1978, but isolated foci of infection were detected up to 1981, with the virus being associated with meningitis in humans.17, 123, 124, 131, 132, 323, 379 It seems likely that the virus is endemic in these areas with isolated periodic reports of virus activity in livestock.1, 6, 33, 319, 418, 657
The first known excursion of RVFV beyond the continent of Africa occurred in Madagascar in 1979 when the virus was isolated from mosquitoes and a laboratory worker, but no naturally occurring livestock infections were recognized until 1990/91 and 2008/09 when widespread disease in livestock and humans was reported.211, 438, 439, 440, 442, 443
Rift Valley fever made dramatic impacts for the first time in West Africa in 1987, when a severe epidemic occurred in the Senegal River basin of southern Mauritania and northern Senegal. In Mauritania during this epidemic alone 232 human patients died of the disease, and there was a high rate of abortion in sheep and goats.291, 292, 293, 294, 335 After 1987, the prevalence of antibody to RVFV in livestock declined steadily in Mauritania, until an outbreak affecting sheep and goats was recorded in the south of the country in 1993, and a larger outbreak affecting livestock and humans occurred in the Hodh El Gharbi region of the south in 1998, with one human death.339, 457, 677 In northern Senegal, antibody prevalence in livestock also declined after the 1987 outbreak, although isolations of virus from mosquitoes and a low level of seroconversions were recorded in continuous monitoring, indicating that RVF is endemic in the region. Smaller outbreaks occurred in sheep and goats in Mauritania in 1994/95, 2010 and 2012 with the largest and most widespread and recent outbreak reported in 2015.108, 213, 243, 319, 577, 578, 600, 601, 602, 605, 658, 678
Severe outbreaks of RVF have also been reported in other African countries319 including Rwanda in 2012 /14 and 2016-2018, Senegal 2013/14, Niger 2016, Somalia 2006/07, Sudan 2007/08, 2010 and 2017-2019, Tanzania 2007, Uganda 2016 , Chad 2018, the Republic of Guinea 2006, Democratic Republic of Congo 2012, and The Central African Republic 2019.38
In September 2000, RVF was reported in the Jizan Province of southwest Saudi Arabia and in adjoining Yemen.557 These outbreaks lasted until early 2001, and resulted in 95 laboratory confirmed human deaths and the loss of thousands of sheep and goats.5, 42, 373, 557 There had been heavy rains in the inland mountain range which runs parallel to the coast, with drainage from the mountains resulting in the creation of ideal mosquito breeding habitats.303 The main vectors in the outbreaks were Aedes vexans arabiensis, which is a floodwater breeding aedine ideally suited to the flood irrigation farming practised in the affected area, and Culex tritaeniorrhynchus, which breeds in pools of water in the wadis and dams at the base of the mountains.303 There was speculation at the time that the virus may have been imported from Africa with animals, or carried from Africa by wind-borne mosquitoes in 2000, but there were no known epidemics in the Horn of Africa at the time. It is more likely that infected animals were imported during the large 1997/98 epidemic in East Africa, and that infection had gone unreported on the Arabian Peninsula until ideal circumstances for an epidemic occurred following heavy rains in 2000.557 Subsequent phylogenetic studies showed that RVFV that caused the outbreaks in Saudi Arabia was closely related to the virus of the 1997/98 epidemic in East Africa.557 Commercial trade of livestock from the Horn of Africa to the Arabian Peninsula consists of about 5-7 million live small ruminants, especially during religious festival periods.5, 38, 557 It remains to be determined whether the virus has become endemic on the Arabian Peninsula, but serological surveys indicate the potential for low-level ongoing transmission in the area.415
Between 2006 and 2011, a resurgence of severe outbreaks of RVF was reported from East Africa,428, 462, 555, 568 Madagascar,9 and South Africa,30, 657 and for the first time RVF was reported in livestock and humans in the Islands of Comoros from 2008/11 and Mayotte in 2008 and 2018/19.349 These islands form an archipelago of volcanic islands situated off the south-east coast of Africa, to the east of Mozambique and north-west of Madagascar.
In late 2017, the detection of RVFV antibodies in livestock were reported in Turkey, Iran and Iraq although with no confirmed cases of livestock deaths or human illnesses.196, 249, 451, 524, 673
Both legal and illegal trade of live livestock including camels from Sudan, Mauritania and other countries in that region may contribute to the spread of RVFV to North African countries.38, 75, 154, 179, 458 It raised concerns for further spread of RVFV to closest Asian (particularly Saud Arabia) and other parts of the world.10, 36, 38, 80, 107, 150, 163, 225, 253, 315, 351, 384, 494, 645, 675
Since the virus can be transmitted by a wide range of mosquitoes, and livestock circulate sufficiently high levels of virus to infect mosquitoes, many parts of the world would probably be receptive to the virus.411, 611, 617 A recent (2020) European Food Safety Agency (EFSA) Opinion publication38 stated that the risk of introduction of RVFV into the EU Member States through the movement of infected animals is very low, because of strict EU policies for the introduction of live animals from non-EU countries. Similarly, the risk of RVFV entry into the EU through infected vectors was considered very low or low, ,but the probability of the establishment of RVFV transmission, once introduced, was considered to vary from moderate to very high for all EU countries.
Aetiology
Taxonomy and molecular biology
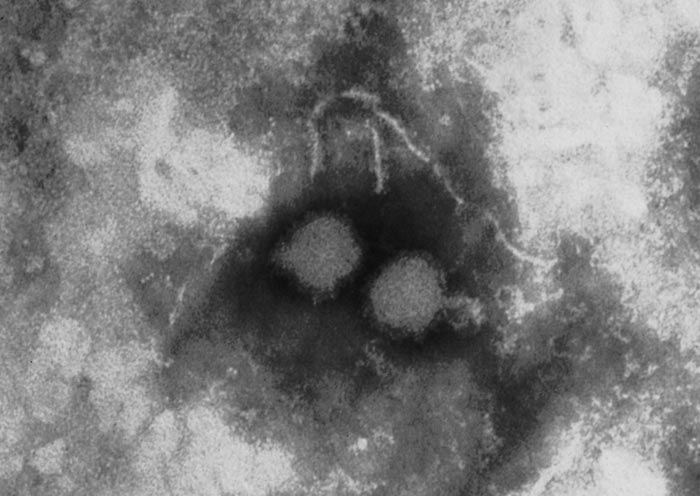
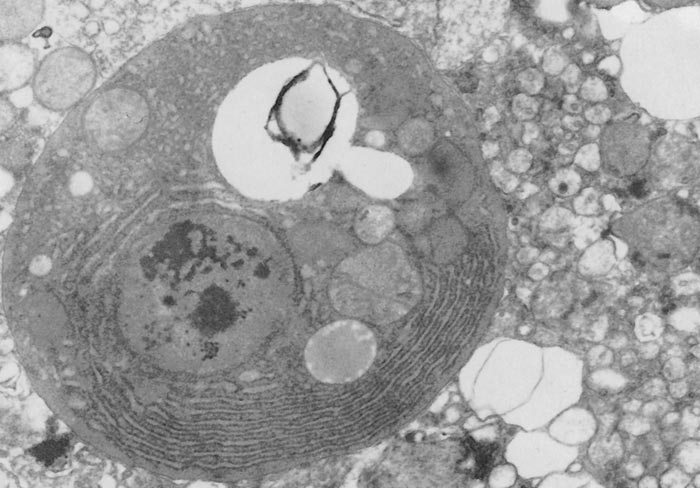
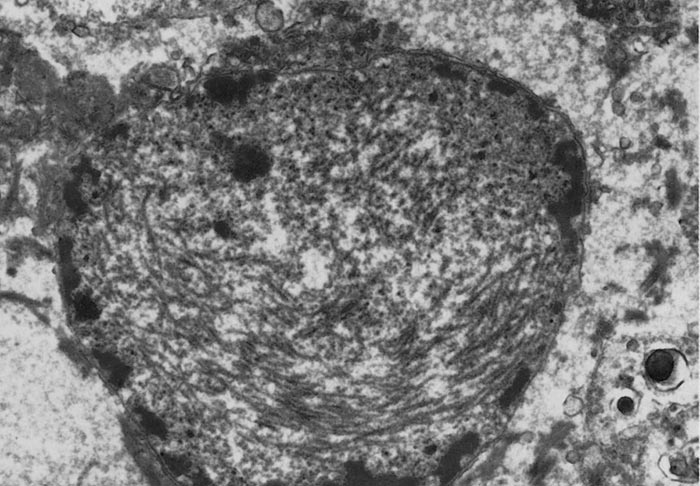
Rift Valley fever virus is a member of Phlebovirus genus, family Phenuiviridae of the order Bunyavirales.374 The viral particles are spherical, 80 to 120 nm in diameter (Figure 1), and consists of an envelope and a ribonucleocapsid (RNP). The envelope is composed of a host cell-derived lipid bilayer containing heterodimers of virus encoded glycoproteins, which are the building blocks of 122 capsomers118 (110 hexamers and 12 pentamers) arranged in T – 12 lattice.554 It has a three-segmented, single-stranded, negative-sense RNA genome of about 12 kb comprised of large (L, 6.4kb), medium (M, 3.8kb) and small (S, 1.6kb) segments. The L and M segments are of negative polarity. The L segment encodes the RNA-dependent RNA polymerase (L protein). The M segment encodes four proteins in a single open reading frame: the precursor to the structural glycoproteins Gn and Gc and two non-structural proteins designated NSm1 and NSm2. The S segment utilizes an ambisense orientation to encode two proteins, the nucleocapsid protein (N) and a non-structural protein (NSs).76, 238, 506 The NSs forms a ribbon-like filament in the nucleus 578, which is particular to RVFV and not shared with the NSs proteins of other bunyaviruses. Rift Valley fever virus replication cycle is similar to that of other negative-stranded RNA viruses76 and involves three major processes: 1. attachment, uptake and fusion, 2.primary transcription, transcription and replication, and 3.virus assembly and release.
All RVFV replication steps occur in the cytoplasm of infected cells and each segment is a template for replication and transcription.184 The virus enters host cells by receptor-mediated endocytosis employing a class II fusion mechanism.204, 367 After uptake, RVFV is trafficked along the endocytic pathway towards the perinuclear localized lysosomes.536 The low pH in endosomal compartments triggers fusion of the viral envelope and endosomal membrane204 followed by genome release into the cytosol, where primary transcription of the genomic-sense RNA (vRNA) is initiated by the ribonucleoprotein-associated RNA polymerase. Messenger RNA (mRNA) transcribed from all segments is translated and viral proteins accumulate. During the replication cycle each vRNA from L-, M-, and S-segments is transcribed into mRNA and is replicated through a process that involves the synthesis of the exact copy of the genome, called complementary RNA (cRNA). The latter serves as templates for vRNA and vice versa. Newly formed vRNAs are encapsidated by N protein and associate with the L protein to form RNPs in so-called viral factories composed of Golgi complex.210 Upon synthesis the Gn and Gc proteins exit the endoplasmic reticulum and accumulate in the membranes of the Golgi complex where virions are assembled through budding.274 Newly formed virions travel from Golgi complex to the plasma membrane where they are released. Systems to manipulate the genome of RVFV, including assembly of minigenomes, have been established and are advancing research on RVFV replication strategy and the development of new generation vaccines.76, 81
Functions and role of RVFV-encoded proteins
The glycoproteins mediate cell receptor recognition, virus entry by binding to specific cell surface receptors(s) and virions escape from infected cells.204 Both Gn and Gc likely contribute to virion assembly and interact with N protein.250 These glycoproteins induce the production of neutralizing antibodies, which play an important role in protection of animals against reinfection with the virus.266, 381
The N protein seems not be involved directly in pathogenesis, but it is essential for virion capsid formation, interacts with glycoproteins, and is involved in transcription and replication.362 The N protein is the most abundant protein in phlebovirus-infected cells and strongly immunogenic,376, 595 but it does not elicit neutralizing antibodies. However, immunization with recombinant N protein induces a partial immune protection in animals. The expression of type I IFN is upregulated in the liver and spleen of mice immunized with recombinant N shortly after RVFV challenge, compared to a delayed upregulation of the same gene in non-immunized mice. Furthermore, various genes with pro-apoptotic and pro-inflammatory effects are strongly upregulated, and anti-apoptotic genes downregulated in the liver of non-immunized mice.286
The M-segment encoded Nsm1 and Nsm2 proteins that were postulated to play a role in triggering apoptosis through the caspase 3, 8 and 9 pathways.663
The S-segment encoded NSs protein is the main virulence factor223, 236, 636, 662 inhibiting host innate viral defenses, primarily by counteracting the antiviral IFN system. This is achieved by a number of mechanisms at the transcriptional and translational levels, including suppression of general transcription, inhibition of IFN-β promoter and downregulation of double-stranded RNA-dependent protein kinase PKR early in infection.250, 275, 276, 311, 341, 597 Removal of the NSs gene results in attenuation of RVFV, which has been demonstrated in naturally attenuated452 and in recombinant viruses59 lacking this gene. NSs gene contains five cysteine residues,39, 40, 151, 181 which are highly conserved in all known isolates of RVFV.11, 527 Cysteine is a highly reactive, hydrophobic amino acid, capable of forming covalent disulphide bridges that stabilize the tertiary and quaternary structures of proteins. A genetic mutant of a highly virulent strain of RVFV was shown to be completely attenuated in a BALB/c mice model after a double cysteine-to-serine substitution at residues 39 and 40 of the NSs protein.430 Since the NSs protein is the major virulence factor by antagonizing IFN responses, the direct attenuation of NSs function potentially limits virus replication due to a lack of inhibition of the primary host immune response.
The NSs and NSm genes were shown to play a role in RVFV replication in mosquito vectors. RVFV lacking NSs and NSm failed to infect Aedes aegypti, and in Culex quinquefasciatus infection rates were lower than for wild-type virus. The double deleted viruses might represent an ideal safe vaccine due to their inability to efficiently infect and be transmitted by mosquitoes.126
The evolutionary history, dispersal, and genomics of RVFV are discussed below (see Epidemiology)
Epidemiology
Vector ecology and virus transmission
Multiple risk factors influence the extent (both in time and space) of re-emergence and expansion of RVF outbreaks in endemic regions and their emergence in previously RVF-free areas. These include, environmental conditions, susceptibility and immune status of host animals (herd immunity), vegetation density, climate change, trade of live livestock, animal movement and distribution of vectors .418. Current data suggest that more than 50 mosquito species, many of which with global distribution, can potentially act as vectors of RVFV.418
The outbreaks of RVF that occurred in North and West Africa (e.g. Egypt, Mauritania and Senegal) in recent years differed in many respects from the pattern of disease which occurred in sub-Saharan Africa: in the former, outbreaks occurred independently of rainfall in arid countries, apparently in association with vectors that breed in large rivers and dams214 It is possible that a proliferation in the construction of farm and river dams (e.g. along the Nile and Senegal rivers) facilitated the spread of RVFV. In contrast to the outbreaks of RVF in the drier parts of North Africa, epidemics in eastern, central and southern Africa have usually been associated with above average rainfall at irregular intervals of 5 to 15 years or longer.145, 397, 587 Meteorological conditions666 favouring the breeding of mosquito vectors of the virus usually prevail over large tracts of southern, eastern and central Africa so that there has been a tendency for outbreaks in adjacent territories such as Zimbabwe, Mozambique, Namibia and South Africa or Kenya, Uganda and Tanzania, to coincide.145, 335, 586 It is clear that the ecology of RVF is a prime example of how climate change and human-induced landscape alterations can impact the emergence and re-emergence of arbovirus diseases such as RVF.657
The flooding of dambos (also refer to as broad vleis) or pans in sub-Saharan Africa and the humid weather conditions prevailing in epidemics favour the breeding not only of the floodwater aedine mosquitoes but also of non-aedine mosquitoes that serve as epidemic vectors (see Vectors: Mosquitoes- a southern Africa perspective), as well as other biting insects that are potential mechanical transmitters of RVFV.93 Eggs of species that breed in water, other than aedine mosquitoes, cannot survive dry conditions and these insects re-colonize flooded dambos or pans from suitably close rivers or dams, so that a succession of vector species occurs once flooding takes place.356, 357, 358, 365 Infected livestock circulate high levels of virus and mechanical transmission of infection by mosquitoes, midges, phlebotomids, stomoxids, simulids and other biting flies is thought to play a significant role in epidemics.140, 161, 212, 307, 345, 618, 633 Infected animals appear to be more attractive to mosquitoes, and by implication other biting flies and midges, than non-infected animals and it has been shown that probing for blood and feeding proceed more rapidly and efficiently on viraemic hosts.40, 521, 612
Apart from mosquitoes, sandflies were shown experimentally to be capable of acting as biological vectors of RVFV, but this is not known to occur in nature.619 Experimentally inoculated Hyalomma truncatum ticks were able to transmit RVFV by bite, but transmission attempts with several species of ticks fed on viraemic hosts were unsuccessful, thus ticks are not thought to play a role in the epidemiology of the disease.134, 321, 360, 401
During times of heavy rain dambos in eastern, central and southern Africa and the panveld of the inland plateau in South Africa, may become flooded and the water may remain standing for several months. Dambos or broad vleis and pans constitute ideal breeding habitats for floodwater aedines. Under these conditions large populations of floodwater Aedes mosquitoes are produced initially, which are subsequently replaced by large populations of Culex mosquitoes. Infection rates in vector populations may be quite low even during epidemics, usually below 0,1 per cent, but enormous numbers of aedines emerge from flooded dambos or pans and vertebrates are subjected to high mosquito biting frequency.307, 353 Infected livestock and wild herbivores serve as a source of virus for mosquitoes and once infection is amplified in them, secondary or epidemic vectors such as Culex spp. and anopheline mosquitoes and other biting arthropods may become involved in transmission. It is notable that in southern Africa the onset of epidemics tends to be recognized late in summer following an initial increase in vector populations.
In southern Africa there is considerable arbovirus activity in the tropical region of Mozambique and the Zambezi Valley and in the subtropical, moist northern belt that extends westwards from Zimbabwe and embraces the Okavango swamps in northern Botswana, and the Zambezi Region (formerly Caprivi strip) and Etosha pan in north-eastern Namibia.70, 248, 329, 330 Unfortunately, a good understanding of the natural cycles of many arbovirus infections, and to some extent also RVFV, especially concerning the role of mosquito vectors and reservoir host vertebrates is lacking and often speculative. The mosquito catalogue444 website serves as a useful resource for determining the distributions of mosquito species and a review by Braack et al 201879 provides a comprehensive list of all mosquito species from which mosquito-borne arboviruses have been isolated from in Africa.
In contrast to the tropical and subtropical regions in southern Africa, the inland plateau in South Africa has virtually no rain from May to October, while dry winters from June to August are harsh and night temperatures are often near freezing. Dambos, pans and small dams may remain dry sometimes for many years at a time. Usually no major RVFV activity is recorded on the interior plateau of South Africa in the inter-epidemic period following major epidemics, such as the outbreaks of1974 to 1976, despite the occurrence of floods in many districts in the following 15 to 20 years. However, following heavy rains in 1996, in the northern and eastern parts of South Africa, a small outbreak of abortion caused by RVFV was reported among captive African buffalo (Syncerus caffer) in a boma in the Kruger National Park, but no spread of infection to the interior plateau was detected.25
The isolation of virus from a wild-caught mosquito is in itself not conclusive evidence for its role as a vector, especially if virus isolates have been made from whole mosquito bodies, as viruses may not be efficiently transmitted by the saliva despite being in other body parts. However, clues to potential vector species can be gained if multiple isolates of the same virus are made from a single species. Follow-up vector competence studies and virus isolations from salivary glands of wild-caught mosquitoes for those species should then be performed to confirm vector status. Inability to maintain colonies of many specific mosquito species and lack of expertise and facilities to conduct these follow-up studies, especially in Africa, add to the reasons for the paucity of confirmed African arboviral vector mosquito species.
Apart from virus isolation from wild-caught mosquitoes, the lines of evidence incriminating a mosquito species as a vector are its susceptibility to infection with, and its ability to transmit the virus concerned (vector competence) plus its relative density and ecological characteristics. Vector competence is assessed quantitatively in laboratory tests while data on density and ecology are obtained by field observations. Relevant aspects of bioecology are relative density, seasonal dynamics, feeding behaviour (including time of biting activity, host preferences, feeding frequencies), larval habitat associations and biology of the immature stages. Together, vector competence and aspects of adult bioecology that affect and condition transmission are usually expressed as vectorial capacity, a concept which was developed by Garrett-Jones230 after the mathematical modelling for malaria by MacDonald.371
Different species of the Aedes and Culex genera (see Vectors: Mosquitoes- a southern Africa perspective) are the main biological vectors of RVFV.300, 359, 361, 402, 403, 613 The order of RVF vectorial importance in southern Africa is Cx. theileri, Ae. circumluteolus, Cx. zombaensis, Ae. mcintoshi and Ae. juppi. Of the Aedes species, Ae. mcintoshi, Ae. circumluteolus and Ae.caballus have the widest distributions in southern African countries such as South Africa, Mozambique, Namibia, Botswana and Zimbabwe.
Aedes circumluteolus (a savannah group member of subgenus Neomelaniconion) is widespread in Africa.173, 263 It is often one of the most prevalent of all mosquitoes during the summer in southern Africa.152, 305, 391, 399, 400, 667 It feeds opportunistically on mammals such cattle, sheep, antelope and humans.305, 400, 482 Of all the “Savanna” group Neomelaniconion species, Ae. mcintoshi (formely named Ae. lineatopennis) is the most widespread in southern and eastern Africa. It would seem that Ae. juppi is adapted to temperate conditions and is endemic to the temperate regions of southern Africa. The development cycle of Ae. juppi and Ae. mcintoshi from egg to adult can be as short as five days after inundation.302
In 1982 and 1984 the virus was isolated from unfed male and female Ae.mcintoshi mosquitoes (reported as Ae. lineatopennis) hatched in dambos on a ranch in Kenya during inter-epidemic periods, indicating that it may be maintained by transovarial transmission in aedine mosquitoes. It is obligatory that the eggs of these floodwater aedine mosquitoes be subjected to a period of drying as water recedes before they will hatch when wetted again during the next flooding cycle. This biological requirement may provide a mechanism for seasonal survival (overwintering) of RVFV.353, 364, 365 It is thought that RVFV infected aedine eggs can survive for long periods in dried mud, possibly for several seasons if habitats remain dry.229, 299 Moreover, it appears that only a proportion of eggs hatch at each successive flooding, which clearly represents a survival mechanism to prevent the entire mosquito population from being lost when precipitation has been inadequate to sustain breeding.364 The finding furthermore suggests that sexual transmission could occur from transovarially infected males to female mosquitoes.359 It should be noted that Scott et al.548 had decades earlier found evidence of inter- epidemic transmission of RVF infection in cattle in Kenya. Moreover, Alexander in 1957 had made reference to the isolation of RVFV from unspecified mosquitoes reared from eggs collected from a pan in South Africa in a publication that was overlooked for almost three decades, and which pre-dated the demonstration of transovarial transmission of any other virus in mosquitoes by 16 years.68, 359, 364
Floodwater Aedes mosquitoes do not mate in insectary conditions, preventing them from forming colonies, and the few vector competence studies performed on them have been done on individuals reared from field collected larvae, which is not ideal. In South Africa, transovarial transmission tests and testing of mosquitoes reared from field-collected eggs have been undertaken to try to incriminate various floodwater Aedes spp. as reservoir vectors of RVFV.228, 300, 401 Such studies have so far been directed mainly at Ae. juppi and indicated that this species has a low vector potential, that it is only a subsidiary epidemic vector, and that it is not involved in vertical transmission of the virus.
In contrast to aedine mosquitoes, Culex spp. deposit their egg rafts on the water surface, where, depending on the temperature, they hatch within two or three days. These eggs cannot survive drying, hence the preferred types of larval habitat are permanent or semi-permanent ground pools. Culex theileri is widespread, occurring throughout the east and southern African region and the Mediterranean region, extending into the Northern Oriental region. (see Vectors: Mosquitoes- a southern Africa perspective).It is moderately ornithophilic, but also feeds on a wide range of other hosts such as goats, cattle, sheep and humans and serves as an arboviral bridge vector.18, 297, 304, 306 It has been implicated as a potential vector for several bunyaviruses.635 Theoretically it might be possible for some infected Culex spp. to overwinter and initiate transmission the next spring, especially as small numbers of Cx. theileri adults have been collected in the winter in Johannesburg, South Africa.296, 298
Field studies during RVF epidemics have shown only very low infection rates in the mosquito vectors.392, 401 In addition, high vector competence evidently requires high levels of viraemia in the vertebrate host.398, 401 Despite this, a striking feature of RVF epidemics among sheep on the inland plateau of South Africa and other parts of Africa is the rapidity with which infection spreads through flocks, causing heavy losses. To account for the high level of infection, biological infection by mosquitoes is almost certainly augmented by mechanical transmission by various haematophagous Diptera including Stomoxys spp., Culicoides spp. tabanid species, and several species of Aedes and Culex.261, 307 In view of the intense viraemia which occurs in RVF it is likely that virulent virus may be transferred when animals are vaccinated in succession with the same needle during an epidemic, and incidents where this appears to have happened were observed in South Africa during the 1974 epidemic.187
Although contagion has been demonstrated on occasion under artificial conditions, non-vectorial transmission is not considered to be important in livestock, as opposed to humans.639, 640, 649, 671
A high degree of herd immunity develops in locations where infection is most intense, as judged by high morbidity, mortality and antibody rates, and it can be surmised that this must be one of the factors that contributes to the abatement of epidemics. Nevertheless, the large numbers of animals that were on occasion vaccinated during outbreaks of RVF must ultimately have contributed significantly to the abatement of epidemics.
Apart from aedine and culicine mosquito vectors in southern eastern and central Africa Ae. caspius, Cx. pipiens, Cx antennatus and Cx. perexiguus have been reported as the main potential vectors during RVF outbreaks in Egypt319 while Ae. vexans and Aedes ochraceus serve as maintenance vectors and Ae. dalzieli and Cx poicilipes as possible secondary vectors in West Africa155, 212, 213, 605, 678 and Saudi Arabia.303, 420 Culex poicilipes and Cx. tritaeniorhynchus, were also important vectors of RVFV in the Saudi Arabia outbreaks.155, 303
According to EFSA Opinion and other publications.37, 107 the following potentially competent RVFV vectors are present in EU countries: Ae. vexans, Ochlerotatus caspius, Ochlerotatus detritus, Cx. pipiens, Cx. theileri, Cx. perexiguus, Cx. antennatus, Cx. ritaeniorhynchus, and Ae. albopictus. Of these Ochlerotatus caspius and Cx. pipiens are considered to be the most competent vectors.227, 446, 620 Culex pipiens (the main vector during the RVF outbreaks in Egypt in 1977/78) and Ae. albopictus are widespread in European countries89, 610 and competent to transmit RVFV.89 Few vector competence studies have been done on European mosquito species. Studies on field-collected mosquitoes in southern France and Tunisia found that Cx. pipiens was the most competent mosquito vector while among laboratory-established colonies of mosquitoes species, Ae. aegypti was most competent.446
Vector transmission is a complex process requiring the virus to enter and cross multiple intra-mosquito barriers and compartments. Briefly, following the ingestion of an infective blood meal by a susceptible mosquito there is an extrinsic incubation period of approximately one to two weeks before transmission can occur. During this time, RVFV replicates in the cells of the midgut, escapes to the haemocoel and is disseminated via the haemolymph to replicate in the salivary glands and other organs, but in a proportion of mosquitoes infection is confined to the midgut, implying that there is a mesenteronal barrier to the spread of infection.200, 201, 520, 615 In a small proportion of mosquitoes there is rapid dissemination of RVFV via a ring of cells at the junction of the foregut and midgut.347, 348, 519 Transmission rates increase to 100 per cent in some species of mosquito if the mesenteronal barrier is by-passed by intrathoracic inoculation of virus, but in other species some or all individuals remain unable to transmit the infection, indicating that there may also be a barrier to infection at the level of the salivary glands.609, 615 Infection of mosquito salivary glands by parasites, such as plasmodia, can facilitate penetration and transmission of RVFV.634
The proportions of mosquitoes in which infection, internal dissemination of infection and transmission of virus occur, and the duration of the extrinsic incubation period, tend to be characteristic for each vector species and do not appear to be influenced by strain of RVFV. However, increased doses of virus, and higher ambient temperatures during extrinsic incubation produce disseminated infection in a greater proportion of mosquitoes and result in higher transmission rates and shorter extrinsic incubation periods.88, 201, 608, 614, 615, 622 Thus, apart from favouring the breeding of vectors, warm weather may be an accessory factor in precipitating outbreaks of RVF through increasing vector efficiency. Different populations of the same mosquito species may vary in vector efficiency if there has been genetic selection, as could occur in the course of establishing a laboratory colony.227
Infection of mosquitoes with RVFV causes morbidity and mortality as evidenced by reduced fecundity, inefficient feeding and lowered survival rates, but the effects are not uniform in all species and vary with stress imposed on the mosquitoes.160, 202, 484, 607, 616 Inefficient feeding by infected mosquitoes manifests as repeated probing and partial engorgement, and may actually increase vector efficiency since attempts to feed may be made on a series of hosts in succession and transmission can occur each time a host is probed whether or not a blood meal is obtained. It appears that mosquitoes inoculate virus extravascularly.624 Feeding on immune animals does not appear to affect the subsequent ability of infected mosquitoes to transmit infection.483
Schreur et al.55, 483 showed that RVFV can be transmitted to lambs by laboratory-reared Ae.aegypti mosquitoes that were infected either by membrane feeding or by feeding on lambs that developed viraemia after intravenous inoculation of RVFV. These experimental models can be useful to investigate mosquito-mediated transmission of RVFV and to evaluate the efficacy of vaccines against mosquito mediated RVFV infection.55, 483 It should be noted that to date RVFV has not been detected in Ae. aegypti collected from the field. This may be due to differences in susceptibility between laboratory-reared mosquitoes and field-collected mosquitoes, as well as because of host preferences. Aedes aegypti is mostly found in urban areas, whereas RVFV is most prevalent in rural areas where domestic and wild ruminants reside.55, 483, 642 The efficient experimental transmission of RVFV to lambs by Ae. aegypti is of concern as this mosquito species is the primary vector of also of other arboviruses with major public health impacts, including chikungunya virus, dengue virus, yellow fever virus, and Zika virus.55, 241, 483 Growing populations of humans and livestock as well as peri-urban agriculture particularly in Africa may in future result in urban transmission cycles of RVFV.
Virus maintenance in the inter-epidemic period
While the ecological drivers of most RVF outbreaks are relatively well understood, the central enigma that remains in the epidemiology of the disease concerns the fate of the virus during the inter-epidemic periods (IEP).
For decades it was widely accepted that the virus was endemic in indigenous forests, where it circulated in mosquitoes and unknown vertebrates, and that it spread to livestock rearing areas when heavy rains favoured the breeding of epidemic mosquito vectors. Unfortunately, there is no scientific proof for this theory. A search was made for potential feral vertebrate hosts of RVFV in Zimbabwe with emphasis on rodents, which had been implicated since the early investigations in Kenya,133, 134 but neither in Zimbabwe nor elsewhere has definitive field or laboratory evidence been obtained to confirm rodents in cryptic cycling of the virus, despite the fact that several species develop transient high-titred viraemia following infection.136, 149, 234, 235, 257, 390, 423, 547, 590, 596, 646, 665 Rift Valley fever virus was isolated from a rat (Rattus rattus) during the Egyptian epidemic of 1977/78,277 but the finding was not considered to be epidemiologically significant.262, 499, 556 Viruses isolated from rodents subsequent to the Egyptian epidemic are thought to be uncharacterized phleboviruses.556 Antibody has been demonstrated in rodent sera in South Africa in areas where the disease has not been recorded for decades, and although the antibody tests are of dubious validity, the findings have led to renewed interest in rodents as reservoir hosts elsewhere. Similarly, surveys and laboratory studies have failed to prove that the virus is maintained in transmission cycles in birds, monkeys, baboons or other wild vertebrates, although it was felt at that time that wild ruminants could play a role similar to their domestic counterparts in areas where they predominate, as discussed below.66, 137, 138, 139, 141, 147, 195, 289, 382, 402, 522, 586, 590
In South Africa in particular, but also in other African countries, many private farmers rear wildlife for ecotourism, conservation, trophy hunting and game meat production. The game ranching industry in South Africa is now worth more than $700 million annually and game animals become infected with a range of mosquito-borne arboviral infections including RVFV. Mixed livestock and game animal farms/conservation areas create ideal opportunities for interactions between different arboviral reservoir host animal populations and bridge mosquito vectors.259, 570 This in turn creates added risks of viral transmission for farm workers and veterinarians, especially via exposure to infected carcasses.632
Although RVFV was isolated from unfed male and female Ae. mcintoshi mosquitoes (reported as Ae. lineatopennis) hatched in dambos on a ranch in Kenya during IEPs in 1982 and 1984, the role of vertical transmission in Aedes spp. in the maintenance of RVFV in the IEP has been poorly studied.368 Recently vertical transmission of RVFV in colonized Cx. tarsalis mosquitoes has been reported in the laboratory 55, 483, 537 Specifically, it is not known if this transmission mechanism alone is sufficient to maintain RVFV endemicity, or if a continuous low-level virus amplification in domestic animals and wildlife is required to maintain the virus in the environment during IEPs66 Numerous serological studies showed that there is continuous cryptic low-level virus transmission of RVFV during IEPs ,without noticeable outbreaks of disease or clinical cases, particularly in less susceptible domestic livestock such as cattle, goats, camels as well as wildlife in different African countries (e.g., South Africa, Mozambique, Sudan, Kenya, ) and would seem to be important in the maintenance of RVFV.32, 38, 77, 95, 106, 182, 189, 264, 282, 322, 359, 361, 385, 387, 449, 477, 494, 516, 522, 582, 583, 627, 628 Rift Valley fever virus transmission during IEPs has also been reported in humans in Tanzania,255 Kenya,336 Gabon510 and Botswana.533
While field studies have shown infection with RVFV of both male and female Aedes mosquitoes reared from field-collected larvae in Kenya, these original findings could not be subsequently confirmed, and demonstration of transovarial transmission is hampered by the difficulty in establishing laboratory colonies of Aedes mosquitoes. It has been demonstrated that the larvae of Cx. pipiens, Ae. mcintoshi and Ae. circumluteolus can become infected after feeding on liver homogenates from an experimentally inoculated hamster but the epidemiological importance of this finding is not clear.618
Rift Valley fever virus activity during the IEP in wildlife, certain domestic livestock and humans highlights the importance of a continuous cryptic endemic transmission cycle and virus evolution in suitable epidemiological/ecological settings. Clinical signs in these susceptible species are often mild or subclinical and are either underreported or misdiagnosed.490
The susceptibility of different livestock species and African wildlife species are discussed below (see Clinical signs).
Evolutionary history, dispersal, and genomics of RVFV
The evolutionary history of RVFV has been influenced by dramatic changes in the environment and land use throughout Africa in the past 150 years. Over this time span multiple levels of virus evolution have occurred, ranging from the macroscopic geographic translocations of virus genotypes across Africa to the molecular point-mutations and genome segment reassortment events.48, 61, 62, 71, 97, 218, 244, 525, 527, 529, 530, 557, 558, 576, 668 Genomic reassortment or homologous recombination are both potent mechanisms to generate genetic diversity and can eventually drive the emergence of novel RVFV variants. The reassortment of RNA genome segments within virus species such as RVFV and within other species of the order Bunyavirales and family Phenuiviridae has been reported frequently in both in vitro and in vivo studies; however, cross-species or cross-genera reassortants (e.g., Batai, Bunyamwera and Ngari viruses) occur less frequently.50, 51, 61, 73, 78, 84, 85, 103, 104, 237, 244, 529 In contrast, little evidence of homologous recombination among RVFV has been reported.61, 105, 244, 283, 338 Potential reassortant events have been identified involving each of the three RVFV genome segments (i.e., S segment: Lineage B viruses, M segment: Kenya 2006/07 strain #0608, L segment: CAR strain 73HB1230 and other examples).61, 62, 244, 283 The impact of these reassortment and potential recombination events on RVFV replication, fitness, and most importantly host pathogenicity is not fully known and requires further study.
As a consequence of the expanding genome sequence database, the number of identified lineages of RVFV has increased from three in early analysis by Sall et al.526 to 15 lineages (designated A-O) in the recent publication by Grobbelaar et al.244 Lineage A contains isolates from North Africa, lineages B-M and O contain isolates from east, central and southern Africa, and lineage N groups isolates from West Africa. The sequence database makes it possible to estimate the number of years prior to the present that the progenitor of the known RVF viruses was in circulation.128, 364 Recent Bayesian analysis suggests that the time of RVFV divergence from the most recent common ancestor (TMRCA) occurred relatively recently, with mean values of the TMRCA coalescing towards 90 to 140 years before the present, i.e., approximately 1880 to 1930.61, 62, 380 This time of origin and original dispersal from the ancestral homeland of the virus is broadly consistent with the early reports from Kenya at the beginning of the 1900s of a disease resembling RVF among animals. It also coincides with a time period of major ecological changes in eastern and southern Africa, with the establishment of colonial agriculture systems and the importation of large numbers of highly susceptible European breed livestock.251, 262
However, despite extensive geographic dispersion, wide range of susceptible arthropod vectors and vertebrate hosts, and natural occurrence of reassortment,48, 529, 531, 623 RVFV displays low genetic diversity. Irrespective of the genome segment analysed, the genetic diversity is approximately 4 per cent at the nucleotide and 1 per cent at the protein coding levels61, 62, 244, 380 The low genetic diversity of RVFV may reflect the evolutionary constraint imposed on arboviruses by their altering replication in mammalian and arthropod hosts.62 It has been shown that serial passages of RVFV in baby hamster kidney cells 21 (BHK-21) or Aedes aegypti cells (Aag2) results in large deletion of the NSs gene, while serial alternating passages between BHK21 cells and Aag2 did not induce this deletion. These findings indicate that host alternation is important to maintain stability of the NSs gene, thereby promoting the virus capacity in evasion of the innate immune response.447 Nevertheless, the minor nucleic acid and deduced amino acid differences that have been reported could account for differences in pathogenicity.19, 48, 98, 496
While no distinctive mutually exclusive correlation of virus genotype, virulence in livestock and humans, or geographic location can be observed with RVFV, virus isolates from one area tend to cluster together within each lineage, but virus genetic variants with distant origins are found within different lineages, providing evidence of widespread dispersal of RVFV throughout Africa. The magnitude of this long-distance and repeated translocation can be found by the monophyletic linkage of isolates from regions as distant as Egypt, Madagascar, and Zimbabwe or Kenya, Mauritania, Burkina Faso, Zimbabwe, and South Africa. The strong phylogenetic linkage of virus strains from distant geographic locations suggests that the movement of infected livestock and the dispersal of mosquitoes may account for the spread of RVFV throughout continental Africa, Madagascar, and the Arabian Peninsula.61, 244, 532 On the other hand, could it be that these virus strains have been present in these distant geographic locations and only now detected with the availability of improved molecular techniques?
During the 1977/78 epidemic in Egypt it was speculated that the transportation of infected sheep and cattle on the Nile river or overland from northern Sudan to markets in southern Egypt was the strongest possibility, and that movement of animals for slaughter by sea could account for the presence of antibody detected in the north and eastern coastal areas.222, 262, 409, 551 Although transportation on some routes could take a long time in relation to the course of the infection, RVFV antigen can persist particularly in the spleen, for up to 21 days after infection. Humans slaughtering or handling the tissues of such animals could have become infected and possibly served as the amplifying hosts for the infection of mosquitoes, since the putative vectors in Egypt include Ae. caspius, Cx. pipiens, Cx. attenuatus and Cx. perexiguus, which are known to feed on livestock and humans.221, 223, 224, 262, 288, 379, 620
Likewise, the results of phylogenetic studies demonstrated that the RVFV detected on the Arabian Peninsula in 2000 was closely related to the virus that had caused the 1997/98 epidemic in East Africa.557 As stated above it is likely that infected animals were imported during the large 1997/98 epidemic in East Africa, and that infection smouldered until heavy rains precipitated the epidemic in 2000. Travelling time for animals exported from infected areas can be as little as 48 hours, well within the usual incubation period of the disease.
In previously RVFV-free countries/regions, RVF outbreaks result from the spread of a single lineage62 Early detection of the large 2006/07 RVF outbreaks in East Africa provided an opportunity to conduct more detailed molecular epidemiology studies. Genome segment sequences obtained from domestic livestock and wildlife and representing all affected regions of Kenya were all monophyletic with a virus isolate from the previous 1997/98 outbreak in East Africa. However, among the 2006/07 viruses analyzed, two separate sub-lineages (Kenya-1 and Kenya-2) were identified with increased genomic diversity relative to that observed among RVFV isolates from the Egyptian 1977/78 and Mauritanian 1987 outbreaks.61 Results from a similar study analysing RVFV isolates from humans and mosquitoes in Kenya and Tanzania indicate that the sequential RVF epidemics in the region were caused by 3 distinct lineages (Kenya-1, Kenya-2, and Tanzania-1), sometimes independently activated or introduced in distinct outbreak foci.460 While the shared evolutionary history of the 1997/98 and 2006/07 outbreak viruses was apparent based on phylogeny, further population genetics studies on Kenya-1 and Kenya-2 lineages revealed that the two lineages were more closely related to the 1997/98 RVFV prototype than to each other, indicating ongoing and separate evolutionary patterns since the previous outbreak. Moreover, it appears that the Kenya-1 lineage viruses had recently undergone geographic or spatial expansion, whereas the Kenya-2 lineage viruses had likely not. Interestingly, the timing of the Kenya-1 expansion event was calculated to have occurred approximately 2-4 years prior to the detection of the 2006/07 outbreaks. These results indicate ongoing RVFV activity and evolution during the inter-epidemic period and highlight the importance of a cryptic endemic transmission cycle that allows for the establishment of RVFV endemicity and to precipitate explosive outbreaks.
Results of a molecular epidemiology study by Soumaré et al576 suggest that Barkedji was a “hub” associated with three distinct introductions of the virus in Senegal from where it was then spread to other localities in West Africa. Barkedji, situated in the semi-arid region of central Senegal, was previously postulated to play a role as an important gateway for RVFV into Senegal and Mauritania based on serological and entomological surveys.605, 677, 678 It is an important crossroad of migration movements of wildlife between the southern and northern regions of Senegal, and to a larger extent, to southern Mauritania. Dispersal distances of most RVFV vectors from their breeding sites is short, and is estimated to be about 1 km, varying from less than 150m for Aedes spp.to approximately 2 km for Cx. theileri.222, 553 Long-range dissemination of the virus in certain parts of Africa could be the result of infected members of roaming herds of livestock or wildlife.336
Mosquito bites constitute the principal infection mechanism of RVFV in animals, but the different dynamics of the virus spread during large epidemics suggest that other transmission mechanism may also exist. Results of space-time analyses of RVF outbreaks in South Africa in 2008 to2011 confirm the presence of an intense, short, initial transmission mechanism, which could be attributed to active vector dispersal, and highlight the presence of another transmission mechanism of lower intensity and over distances up to 40 to 90 km, within about 2 weeks. The appearance of long-distance spread could be explained by emergence of the virus at several foci as a result of hatching of infected Aedes eggs or multiple re-introductions of infected vectors, but as previously stated the importance of transovarial infection in aedine mosquitoes in the epidemiology of RVF remains speculative and must be further substantiated.419
Host susceptibility
The susceptibility of livestock, wildlife and humans is discussed below (see Clinical signs)
Pathogenesis
Differences in virulence and lethality of RVFV isolates have been observed during the experimental infection of BHK cells,659 mice,433 sheep,447 and cattle.659 Mice inoculated intraperitoneally with different wild-type strains (ZH501, Kenya 9800523, Kenya 90058, Saudi Arabia 200010911, OS1, OS7, SA75, Entebbe, or SA51) showed that RVFV strains of different genetic lineages display distinct virulence in outbred mice, although those strains are antigenically similar.272 Given the role of NSs as a major virulence factor for RVFV,57, 76, 250, 275 variability within its amino acid sequences could possibly account for the phenotypic differences between different virus strains.
The pathogenesis of RVF encompasses the spread of virus from the initial site of replication to critical organs such as spleen, liver and brain, which are either damaged by the lytic effects of the virus or immunopathological mechanisms, or else there is recovery mediated by non-specific and specific host responses.496 By analogy with the course of events believed to follow natural infection with other arthropod- borne viruses, it can be assumed that there may be an initial transient viraemia that of too low an intensity to be detectable or, more likely, that virus is conveyed from the inoculation site by lymphatic drainage to regional lymph nodes where there is replication and spill-over of virus into the circulation to produce primary viraemia. This in term leads to systemic infection with intense viraemia that results from release of virus following replication in target organs. Within this basic pattern there are individual and species-related variations in manifestation of disease, as exemplified by the different responses to experimental infection observed in mice, rats and rhesus monkeys.
In in vitro cultures, RVFV replicates in cells derived from virtually all tissues except primary macrophages and lymphoblastoid cell lines, yet in intact animals macrophages are infected. There is also selectivity for certain other tissues, as indicated by the demonstration of viral antigen in rats by immunofluorescence in littoral macrophages of lymph nodes, most areas of the spleen except T-dependent periarteriolar sheaths, foci of adrenocortical cells, virtually all cells of the liver, most renal glomeruli and some tubules, and scattered small vessel walls, as well as in necrotic foci in the brains of individuals with the encephalitic form of the disease.23, 496 In addition, the presence of diffuse virus antigen has been demonstrated in lung tissue in a rhesus monkey.497 The sites of virus replication inferred from the immunofluorescence studies correspond to the lymphoid necrosis in lymph nodes and spleen, hepatic necrosis and adrenal, lung and glomerular lesions seen in humans and livestock. Immunohistochemistry staining of tissues of naturally infected adult sheep with RVFV showed positive antigen staining in a variety of cells including hepatocytes, adrenocortical epithelial cells, renal tubular epithelial cells, macrophages, neutrophils, epidermal keratinocytes, microvascular endothelial cells and vascular smooth muscle.468 In lambs (< 1-month-old) and foetuses viral antigen was detected in the aforementioned cells but also in renal juxtaglomerular and extraglomerular mesangial cells, cardiomyocytes, Purkinje fibres and skeletal muscle cells.470, 471 The results of titration of infectivity in organ homogenates indicate that liver and spleen are the major sites of virus replication.23, 496, 497
Rift Valley fever virus, which attaches to receptors on susceptible cells, is internalized by endocytosis and replication occurs in the cytoplasm.24 The non-structural NSs protein synthesized during replication enters the cell nucleus and forms the filamentous intranuclear inclusions seen histologically in RVF-infected tissues.328, 580, 581, 589 The carboxy terminal domain of the NSs protein is essential for formation of the filamentous structures, but not for intranuclear localization of the protein.669
Viraemia may become demonstrable in lambs less than one week old within 16 hours of peripheral infection with small doses of RVFV, and persists for the duration of the illness, which may terminate fatally within 36 to 42 hours.167, 168, 171 In older sheep, goats and cattle viraemia becomes demonstrable one to two days after infection and persists for up to seven days, usually being most intense on the second to fifth days after infection.167, 170, 171, 398, 594 Maximum titres of viraemia recorded were 1010.1 MIPLD50/ml (mouse intraperitoneal 50 per cent dose /ml) in lambs, 107.6 in sheep, 107.5 in calves, 108.2 in kids, and 105.6 in goats,168, 170, 171 although individual animals may fail to develop demonstrable viraemia.594 Virus has been shown to persist in visceral organs of sheep, particularly spleen, for up to 21 days after infection.671 Viraemic titres of up to 108.6 MICLD50/ml have been recorded in human patients.499
Viraemia of similar intensity and duration to that in domestic ruminants has been demonstrated in hamsters, mice, some laboratory strains of rat and various wild rodents.169, 206, 390, 396, 423, 424, 425, 496, 590, 646 Viraemia of lower intensity and shorter duration has been detected in other animal species that have been studied, but quantitative tests were performed in a few instances only. Maximum intensities of viraemia recorded were 105,4 TCID50 (tissue culture 50 per cent infective doses/ml) in African buffalo, 104,9 MICLD50/ml in dogs, and 102,5 MIPLD50/ml in ponies.141, 639, 672
The peracute hepatic disease seen in mice given large doses of virus422, 423, 424, 425, 426, 496 most closely corresponds with that which occurs in other extremely susceptible hosts such as new-born lambs and kids.114, 117, 119, 168, 171 Other age groups and species of farm animals and humans commonly experience benign infection, but as with rhesus monkeys,125, 434, 497, 498 a variable proportion of adult sheep, cattle and goats and a small proportion of humans develop the fulminant hepatic form of the disease. In these hosts, however, hepatic necrosis may be a less dominant lesion than in mice, with evidence of lymphoid necrosis, vasculitis, nephrosis and haemorrhagic manifestations being relatively more prominent.118, 497, 633 The three genotypic responses of inbred rats to infection — highly susceptible in which there is rapidly fatal hepatic disease, moderately susceptible in which only a proportion of individuals die of encephalitis 7 to 28 days after infection, and resistant23, 90, 501 — mimic the disparate human phenotypic responses of fatal hepatic disease, encephalitis and benign infection.496, 501, 633 It is curious that the encephalitic form of the disease has not been reported in natural infection of ruminants, although it was described in a calf infected peripherally with wild virus.515 Factors contributing to fatal outcome in the hepatic form of the disease include anaemia, shock and hepatorenal failure, with the kidney lesions possibly being as important as shock in producing anuria.496
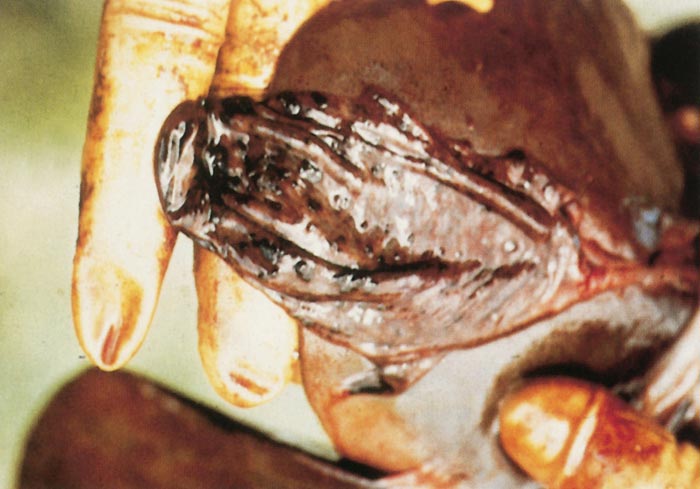
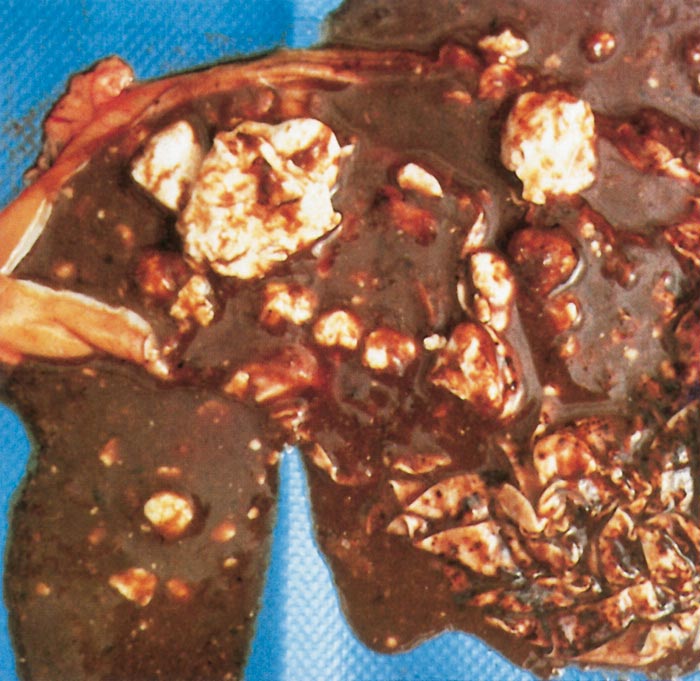
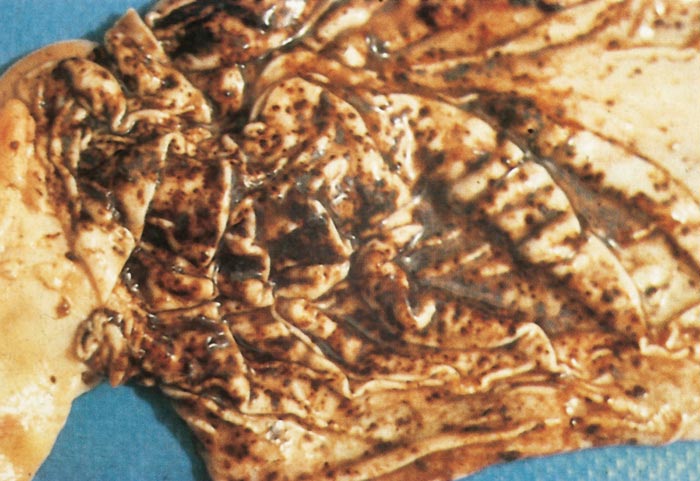
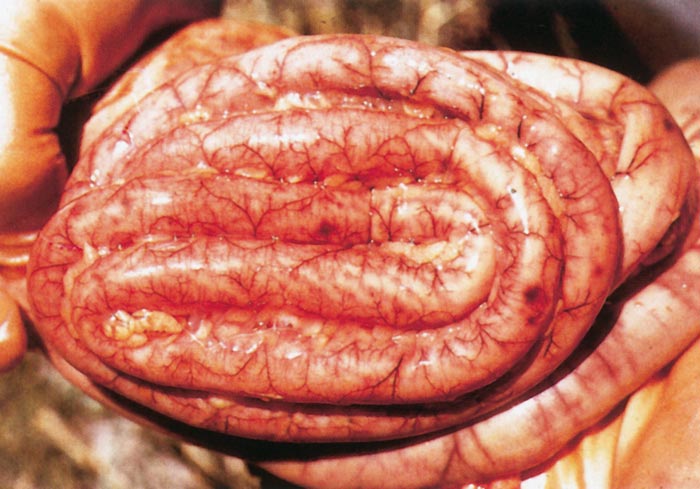
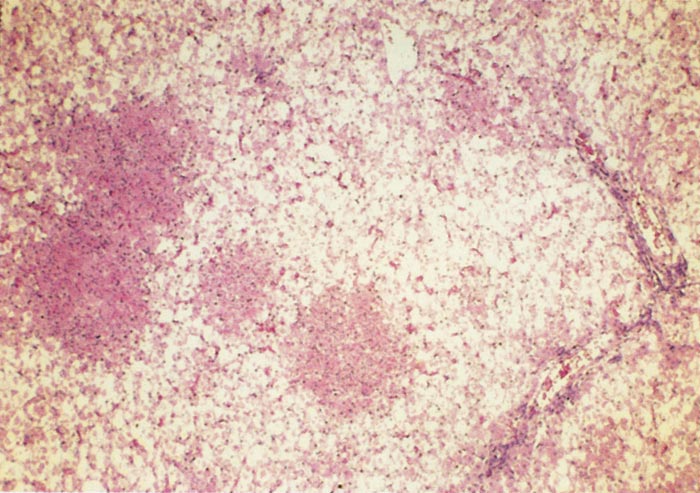
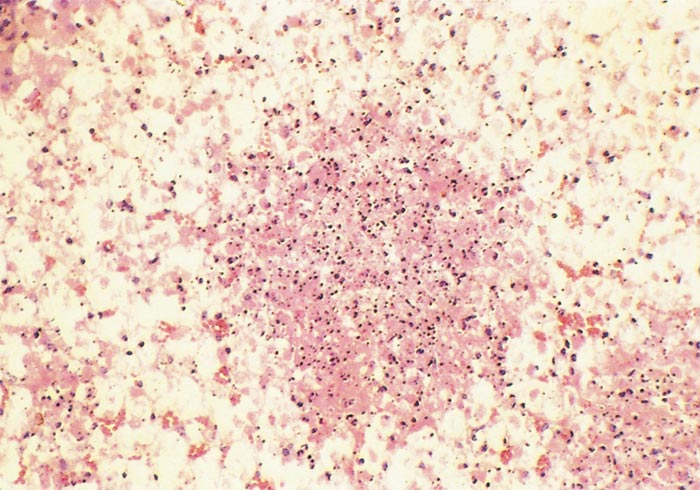
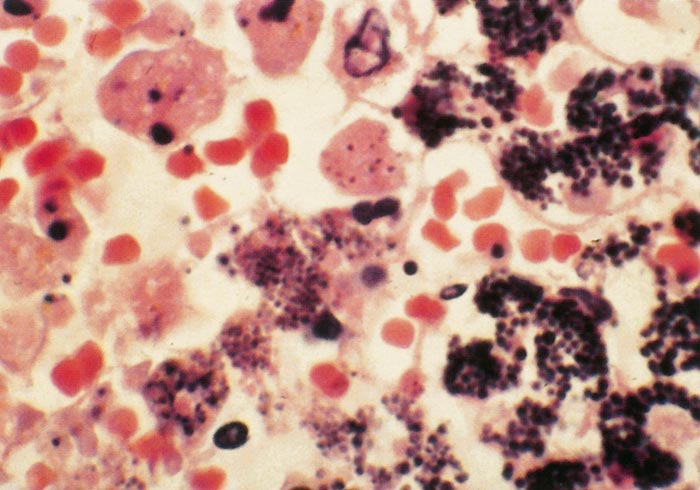
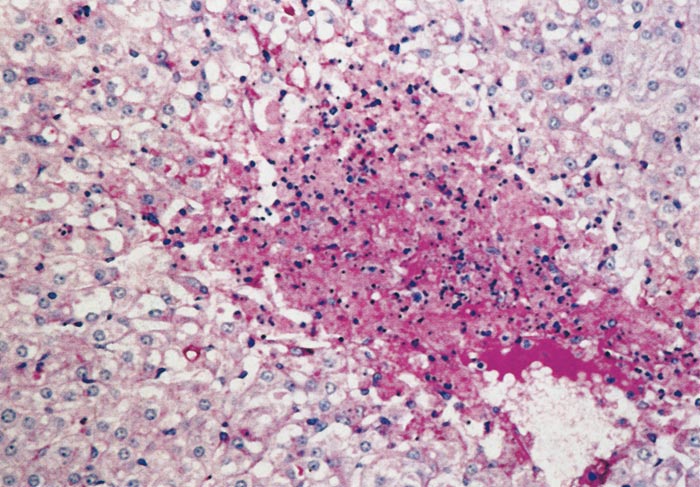
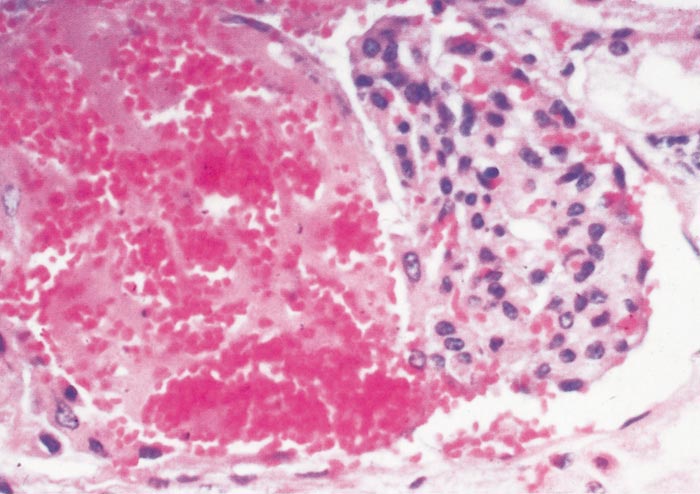
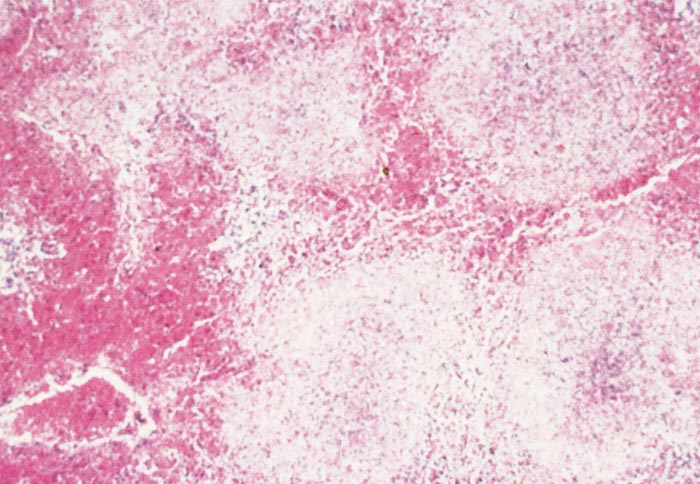
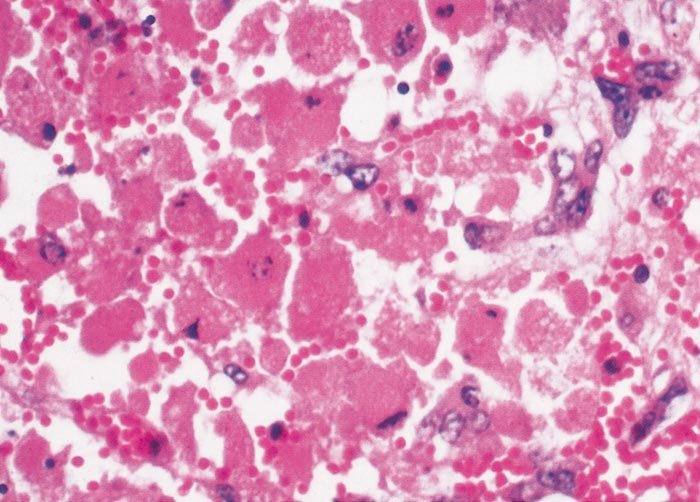
Lesions in target organs such as the liver in the acute disease are produced by the direct lytic effect of the virus on infected cells. In the liver of new-born lambs, for instance, there is initial cloudy swelling and hydropic degeneration of randomly scattered hepatocytes, which soon become necrotic as manifested by acidophilic cytoplasm and pyknotic nuclei. The lesions rapidly progress to form scattered primary necrotic foci of five to eight affected hepatocytes, with the presence of acidophilic cytoplasmic or apoptotic bodies resulting from cytolysis, and infiltration of neutrophils. As the primary lesions enlarge, numerous degenerated and necrotic hepatocytes and acidophilic bodies appear throughout the parenchyma. Ultimately there is massive necrotic hepatitis in which the residual primary foci can still be recognized as dense aggregates of cellular debris infiltrated by leukocytes.117, 168
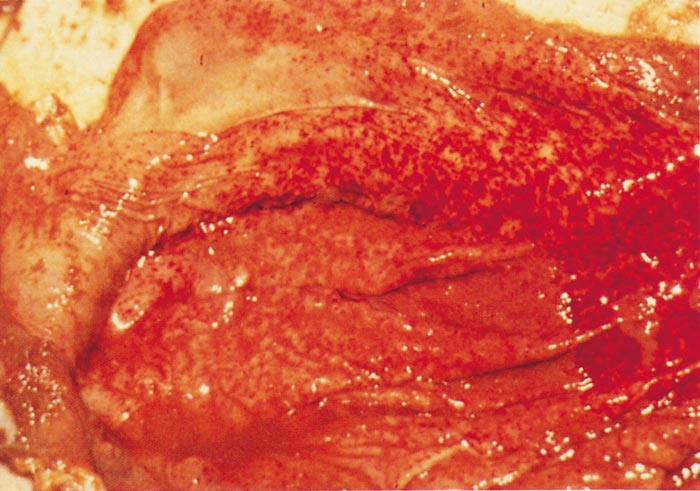
The haemostatic derangement that occurs in RVF has been investigated in detail only in rhesus monkeys, and the mechanisms involved remain speculative.125, 497 Impairment of coagulation occurs even in benign infection in monkeys,125, 497 and it is notable that moderate thrombocytopenia has been observed in benign infection in sheep.595 However, haemostatic derangement is most severe in the fatal hepatic syndrome, which manifests as a viral haemorrhagic fever with bleeding tendency and evidence of disseminated intravascular coagulopathy.125 Viraemia is intense and prolonged in individuals that develop the haemorrhagic syndrome, indicating that there is impaired clearance of viraemia and extensive dissemination of virus with attendant widespread tissue damage, and it is postulated that the critical lesions in the development of the haemorrhagic state are vasculitis and hepatic necrosis.125, 497, 498 Destruction of the antithrombotic properties of endothelial cells is thought to trigger intravascular coagulation, and the widespread necrosis of hepatocytes and other affected cells to result in the release of procoagulants into the circulation. Severe liver damage presumably limits or abolishes production of coagulation proteins and reduces clearance of activated coagulation factors, thereby further promoting the occurrence of disseminated intravascular coagulopathy, which in turn augments tissue injury by impairing blood flow. Vasculitis and haemostatic failure result in purpura and widespread haemorrhages.
Ocular lesions may occur as a complication to RVF infection in humans, occasionally appearing at the time of the acute febrile illness but usually up to four weeks later.122, 153, 217, 234, 377, 413, 539, 540, 564, 565 Reports of long term sequelae including permanent vision deficits and even blindness have increased the need for follow-up investigation of this potential outcome among human patients.13, 461 The essential lesion appears to be focal retinal ischaemia, generally in the macular or paramacular area, associated with thrombotic occlusion of arterioles and capillaries, and is characterized by retinal oedema and loss of transparency caused by dense white exudate and haemorrhages. Sometimes there is severe haemorrhage and detachment of the retina. Ocular lesions have not been recorded in limited experimental observations in infected rhesus monkeys or ruminants.114, 168, 435
Encephalitis occurs from one to four weeks after the acute febrile illness in a small proportion of human RVF patients.340, 370, 413 Virus only appears in the brain at a late stage in rats that succumb to encephalitis, by which time viraemia and infection and lesions of viscera are no longer demonstrable, and circulating antibody is already present; hence it is suggested that there is an immunopathological basis to the encephalitic syndrome in addition to the direct cytopathic effects of the virus.496 In the brain there are focal necrotic lesions with associated polymorphonuclear cell infiltration as well as mononuclear cell infiltration and perivascular cuffing, which suggest participation of T-cells specialized for delayed hypersensitivity or cytotoxicity.496, 633 Complete or partial protection of highly susceptible laboratory animals against the fatal disease can be achieved by the administration of antibody, interferon inducers, recombinant interferon, interferon-induced proteins, or chemotherapeutic agents, (e.g., ribavirin or T705), but sometimes suppression of the usual rapidly fatal hepatitis can lead to the development of the late encephalitic form of the disease.54, 219, 496, 534, 535, 566, 567, 680, 681
Work conducted using aerosol challenge in Sprague-Dawley rats and African green monkeys has established robust animal models of RVF encephalitic disease and revealed marked associations of survival with early increased proliferation of CD4+ and CD8+ lymphocytes and early induction of pro-inflammatory cytokines including gamma interferon (FN-γ), interleukin 6 (IL-6), IL-8, monocyte chemoattractant protein 1 (MCP-1) and antiviral (IFN-α) responses following infection, with late stage alterations after virus invasion of the central nervous system in vascular permeability mediated by matrix metalloprotease-9 (MMP-9) prior to death.254, 644, 664
Abortion is the typical, but not universal, outcome to infection of pregnant sheep, cattle and goats but it has also been reported in other animal species including camels and African and Asian buffalo.26, 350, 409, 670 Abortions which occur soon after infection may be related to the febrile illness of the dam since there may be no evidence of foetal infection, but in most instances abortion follows foetal death with lesions in the foetus resembling those in new-born lambs, including extensive hepatic necrosis.114, 118, 649, 670 However, the brain is also frequently infected and virus or antigen can usually be recovered from foetal viscera and brain.560, 585, 586 Virus can be isolated from the placenta and viral antigen was demonstrated in sheep in epithelial (syncytial) cells of the maternal villi and foetal trophoblasts.595, 678 Widespread necrosis of these cells may cause abortion.471, 480, 586, 670 Septic metritis can occur as a sequel to retention of the placenta, which commonly follows abortion. In a recently described pregnant rat model direct infection and crossing of the placental barrier appears to be a primary mechanism of foetal infection.404 This promising development could allow for the in-depth investigation of RVF-induced foetal infections,but structural differences between ruminant, human, and rodent placentation should be considered before direct extrapolation and interpretation of results. In humans, an attempt to relate the occurrence of abortion to evidence of RVF infection in Egypt produced inconclusive results,3, 413 but recently human miscarriage and vertical transmission events reported RVFV as the causative agent.8, 31, 49 (see Clinical signs).
Another potential foetal adverse event associated with RVFV has been demonstrated among foetuses of ewes inoculated with the live-attenuated Smithburn vaccine strain at 42 to 74 days of gestation may develop various brain and other anomalies such as porencephaly, hydranencephaly and micrencephaly as well as arthrogryposis, which in some cases is associated with hydrops amnii, prolonged gestation and dystocia.115 This vaccine strain was prepared by serial passage intracerbrally and peripherally in rodents and likely has retained high-level neurotropism in foetuses and should be used with caution in pregnant animals.16, 571
The congenic progeny produced by cross-breeding of susceptible and resistant strains of rat exhibit the fulminant hepatic and benign phenotypic responses to infection in approximately equal proportions, suggesting that resistance is inherited as a simple Mendelian gene of large effect, which does not appear to be linked to genes of the major histocompatibility complex.21, 496 Work with whole genome scanning of cross-bred rats used historically in studies of the RVFV stain ZH-501 (highly susceptible Wistar-Furth lineage with highly-resistant Lewis lineage rats) has revealed a genetic locus on chromosome three of as yet undetermined function that underlies this differential susceptibility.91 The same appears to be true for other vertebrate species. It has been found, for instance, that when gerbils (Meriones unguiculatus) are infected with minimal doses of virus about half will die, while the mortality is not increased significantly by administering high doses, i.e. half the gerbil population is susceptible to the lowest dose inoculated and half resist the highest dose.496 It has been estimated from figures generally reported for mortality during epidemics that up to one third of sheep may be susceptible to fulminant hepatic disease, and it is suggested that it may be possible to breed resistant flocks.496 This is a promising area of research, but complicated by the background of genetic diversity with respect to resistance to RVF even within well-defined inbred strains of laboratory animals.517
The understanding of the exact molecular mechanisms underlying virulence and genetic resistance in animal models and humans to RVFV remains an area of intense research.74, 91, 151, 366, 495, 643, 679 Immunosuppression of resistant of moderately susceptible rats with cyclophosphamide, which inhibits humoral response but not interferon production, reduces but does not entirely abolish resistance to RVF. This effect can be reversed by passive administration of antibody.23, 496 Moreover, viraemia and organ titres of infectivity are already very different in susceptible and resistant strains of rat by 24 hours post-infection, which suggests that factors which determine the outcome of infection are operative before humoral and cell-mediated immunity can be activated.496 Fibroblast or macrophage cultures derived from susceptible and resistant rats do not differ in their abilities to support or resist RVFV replication, but somewhat higher titres of virus are achieved in cultures of hepatocytes derived from susceptible as opposed to resistant rats.24, 496
Clinical signs
Despite large amounts of data regarding individual species susceptibility, it should be noted that the pattern of RVF can differ across individual herds and flocks, between separate epidemics, and during the course of a single epidemic. For instance, disease may predominate in either sheep or cattle at a particular location or stage of an epidemic, and outbreaks, which occur after long intervals or in new locations, may be characterized initially by abortions and disease of adults, and at a later stage of the outbreak, by disease of neonatal or immature animals.109, 135, 177, 240, 335, 542 It is understandable that abortion and disease of adult animals should be the major manifestations when an epidemic occurs in an immunologically susceptible herd or flock at a critical stage of the breeding cycle, or that disease of neonatal animals predominates after lambing or calving has taken place. Immune ewes confer colostral immunity on their lambs that is protective for up to five months,571but it was observed in the 1974 to 1976 epidemic in South Africa that lambs subjected to attack by large numbers of mosquitoes as soon as they were born could undergo irreversible infection before colostral immunity became effective.187
The clinical signs of RVF in livestock have been reviewed by several authors.135, 156, 167, 188, 206, 258, 477, 556, 649 Signs of the disease in domestic ruminants tend to be nonspecific, rendering it difficult to recognize individual cases of RVF. During epidemics, however, the simultaneous occurrence of numerous cases of abortion and disease in ruminants, together with disease of humans, tends to be characteristic of RVF. Figures for morbidity and mortality rates can be derived accurately for specific animals in the laboratory, but the perceived figures in the field vary with challenge rate, herd immunity and predisposing conditions. Factors that determine the morbidity and mortality associated with outbreaks of RVF include the virulence of the strain of virus and the susceptibility of the vertebrates involved.19, 21, 496, 501, 603, 606 Vertebrates are usually classified with respect to susceptibility to peripheral infection with RVFV on the basis of laboratory and field observations (see Table 1).19, 133, 134, 135, 139, 144, 147, 167, 168, 169, 170, 171, 205, 206, 208, 390, 410, 427, 496, 546, 556, 590, 639, 640, 646, 649, 660 In some instances extrapolations of mortality rates have been based on very few laboratory observations, while in other instances there are marked discrepancies in the patterns of disease observed in the field, which can partly be explained on the basis of differences in challenge rate and age, acquired immunity and reproductive status of the animals involved, as discussed below.
Most laboratory observations on the pathogenicity of RVFV for farm animals were made in breeds exotic to Africa and many of the experiments were conducted outside the continent. It has been suggested that exotic sheep and cattle are less resistant to RVF than indigenous African livestock,138 but it was shown that West African and Egyptian sheep are highly susceptible to experimental infection.475, 603, 618 Moreover, indigenous livestock were severely affected by the disease in the 1973, 1977/78 and 1987 RVF epidemics in the Sudan, Egypt and West Africa, respectively.176, 335, 386
Although new-born lambs are extremely susceptible and can be infected lethally with as little as 0,1 MIPLD50 of RVFV, it is frequently difficult to produce disease in non-pregnant sheep and cattle with high doses of ostensibly virulent virus.111, 171, 194, 252, 545, 595
Sheep and goats
In new-born lambs and goat kids the incubation period may be as short as 12 hours but is usually 24 to 36 hours. Onset of the disease is marked by the development of fever which may exceed 41 °C and which is often biphasic with a remission of 12 to 18 hours following the initial rise in rectal temperature. The fever subsides sharply a few hours prior to death. Affected animals are listless, disinclined to move or feed, and evidence of abdominal pain can be elicited. Respiration is rapid and often abdominal in terminal illness. The course of the disease is usually peracute and lambs rarely survive longer than 24 to 36 hours after the onset of the first signs of illness; many are simply found dead. In animals less than a week-old mortality is 90 per cent or greater.
Lambs and kids older than two weeks and mature sheep and goats are significantly less susceptible than new-born lambs to RVFV and may develop inapparent, acute or peracute disease. In the peracute disease animals die suddenly without exhibiting noteworthy signs of illness. Under field conditions most animals develop the acute disease. Following an incubation period of 24 to 72 hours there is fever of up to 42 °C that lasts for 24 to 96 hours, anorexia, weakness, listlessness and an increased respiratory rate. Some animals may regurgitate ingesta and develop melaena or foetid diarrhoea and blood-tinged, mucopurulent nasal discharge. Occasionally, animals may be icteric.
Reports on the progression of natural disease in goats are inconsistent. Although abortion in goats and mortality in kids were recorded in Kenya in 1930, the Sudan in 1973, South Africa and Namibia in 1974/75, and in West Africa in 1987, goats were considered to be more resistant to the disease in the Egyptian outbreak of 1977/78; goats infected experimentally developed high-levels of viraemia post-infection.135, 177, 277, 278, 334, 335, 379, 556, 648
Pregnant animals may abort at any stage of gestation as a result of the febrile reaction and/or placental necrosis.26, 471 Aborted foetuses are usually autolysed. Although there is no evidence that fertility is impaired after abortion, this is possible in instances where retained placenta and purulent metritis occur as complications to abortion. Mortality and abortion rates vary between and within epidemics, but in southern Africa mortality rates varying from 5 to 30 per cent and abortion rates of 40 to 100 per cent were estimated for sheep.46, 114, 542 In contrast, it was estimated that up to 60 per cent of sheep died and 80 to 100 per cent of ewes aborted on some farms during the Egyptian epidemic of 1977/78.409 Goats were said to be resistant to the disease in Egypt, whereas mortality in goats was reported to be 50 per cent in Sudan in 1973 and the abortion rate was estimated to be nearly 100 per cent in Mauritania in 1987.174, 294, 379
A range of anomalies of the central nervous system including porencephaly, hydranencephaly and micrencephaly, as well as arthrogryposis, and other defects in foetuses associated with hydrops amnii, prolonged gestation and dystocia115 may occur if ewes are inoculated with live Smithburn strain vaccine between about five and ten weeks of gestation.115 If ewes are inoculated before this stage there may be unnoticed early loss of the conceptus, while inoculation at a later stage may result in abortion, stillbirth or birth of immune or viraemic progeny,115, 116, 649, 650 Teratology following such vaccination has been recorded in the progeny of up to 15 per cent of pregnant ewes in flocks, but on average it appears to affect less than 2 per cent of ewes and abortion probably occurs in less than 10 per cent of pregnant ewes.115, 187, 316, 649, 650, 651
Cattle
The disease in calves resembles that in lambs and sheep, with occurrence of fever, inappetence, weakness and bloody or foetid diarrhoea, but a higher proportion of calves may develop icterus. In fatal disease, death generally occurs two to eight days after infection. Estimates of mortality during epidemics range from less than 10 per cent for all ages of cattle to 20 per cent for calves,114, 177, 542, 586 but a mortality rate of 70 per cent was reported in experimentally infected one-week-old calves.168
Infection is frequently inapparent in adult cattle,111, 118 but some animals develop acute disease characterized by a high fever of 24 to 96 hours’ duration, anorexia, staring coat, lachrymation, salivation, nasal discharge, dysgalactia and bloody or foetid diarrhoea.118, 135, 502 The death rate in cattle does not generally appear to exceed 10 per cent, but was reported to be 30 per cent among cattle that aborted in Egypt.114, 135, 156, 312, 542, 548, 586, 649 Illness tended to run a prolonged course of 10 to 20 days in cattle in Sudan in 1973, with severe icterus being a marked feature of the disease, although most animals recovered spontaneously.177 Sometimes abortion is the only manifestation of the disease in a herd. Average abortion rates of 15 to 40 per cent may occur in epidemics.114, 156, 316, 327, 560, 586, 591 As in sheep and goats, cows may abort at any stage of gestation, the aborted foetus usually being moderately autolysed.116, 541
Unusual manifestations, such as dermatitis crustosa of pigmented and unpigmented skin of the muzzle, face, perineum, udder and scrotum, catarrhal and erosive stomatitis, coronitis, laminitis, and exungulation of the hooves have been reported in association with outbreaks of RVF in cattle, but it is believed that these lesions may have been caused by concurrent infection with other agents, such as bluetongue virus.
Other domestic and wildlife animal species
Pigs, horses and donkeys
Data suggests that adult and young piglets are resistant to experimental infection but develop transient viraemia if a high dose of virus is administered.135, 170, 544 No isolations of RVFV have been made from pigs, and no antibodies were detected subsequent to the large scale Egyptian epidemic.278 However, more recently antibody positive pigs were reported among animals sampled at abattoirs in Egypt in 2009.674
Horses develop only low-grade viraemia following experimental infection,135, 672 but during the Egyptian epidemic, there was one isolation of virus from a horse, and four abortions in donkeys were ascribed to RVF, while a low prevalence of antibody to the virus was detected in the two species.278
Camels and camelids
Antibodies were detected in old-world camels (Camelus dromedarius) in a part of Kenya where abortions occurred during a RVF epidemic in 1962, and again during a survey following the 1978/79 epidemic.143, 546 However, while there was only one isolation of RVFV from a camel during the 1977/78 Egyptian epidemic, 56 deaths and one abortion were ascribed to the disease, albeit on circumstantial evidence.278 The full potential impact of RVFV on camelids became apparent when large scale die-offs and abortions in dromedary camels occurred in 2009/10 in Mauritania. New- world camelids (alpacas) that were imported for fibre production into South Africa developed lethal infection during the 2008/2010 RVF outbreaks.183, 282
Although infection in camels is generally subclinical in mature animals, pregnant camels may abort at any stage of pregnancy and neonatal deaths have been reported in Egypt and Sudan.2, 199, 203, 458, 473 During an unprecedented outbreak that occurred in 2010 in the northern Sahelian region of Mauritania, high mortality rates, haemorrhagic septicaemia and severe respiratory distress were reported.182
Antibodies to RVFV in camels have been detected in many African countries including Sudan, Tanzania, South Africa, Mauritania, Kenya, Niger and Nigeria, as well as Saudi Arabia; seroprevalence rates varied from 3 to 57 per cent.2, 86, 143, 175, 179, 183, 476, 584
Water or Asian buffalo
High seroprevalence of anti-RVFV antibodies, abortion rate of 12 per cent and mortality rate of 7 per cent were found in domesticated Asian water buffalo (Bubalus bubalis) during the 1977/78 epidemic in Egypt.
Wildlife species
Antibodies against RVFV have been detected in many African wildlife species including topi (Damaliscus korrigum), red fronted gazelle (Eudorcas rufifrons), dama gazelle (Nanger dama), scimitar-horned oryx (Oryx dammah), common reedbuck (Redunca redunca), African buffalo (Syncerus caffer), Dorcas gazelle (Gazella dorcas), Thomson’s gazelle (Gazella thomsonii), gerenuk (Litocranius walleri), lesser kudu (Tragelaphus strepsiceros), impala (Aepyceros melampus) sable antelope (Hippotragus niger), waterbuck (Kobus ellipsiprymnus), warthog (Phacochoerus aethiopicus), African lion (Panthera leo), giraffe (Giraffa camelopardalis), Burchell’s zebra (Equus burchellii), black rhinoceros (Diceros bicornis), springbok, (Antidorcas marsupialis), wildebeest (Connochaetes taurinus), African elephant (Loxodonta africana), kongoni (Alcelaphus buselaphus), bats (Micropteropus pusillus, Hipposideros caffer, Hipposideros abae), and waterbuck (Kobus ellipsiprymnus).66, 72, 86, 95, 143, 189, 290, 486
Several serological investigations of African buffalo in the Kruger National Park (KNP), South Africa found that older animals were more likely to be seropositive for anti-RVFV antibodies than younger animals and herds in the south of KNP were more likely to be seropositive than herds in the north.52, 336 Abortion as a result of RVF was reported in the late 1990s in African buffalo that were kept in a boma at Skukuza in the KNP.245
Experimental RVF infection of a seven-month-old African buffalo in Kenya produced fever and malaise,133 and in another experiment four out of five individuals exhibited transient viraemia and one of two pregnant cows aborted.141 It was noted on some properties involved in the 1950/51 epidemic in South Africa that abortion occurred in springbok (Antidorcas marsupialis) and blesbok (Damaliscus dorcas phillipsi) antelope, but this was not confirmed to be due to RVF.290, 295
To date, no experimental infections of any European wildlife have been reported.66 North American white-tailed deer (Odocoileus virginianus) were recently found to be highly susceptible to lethal disease characterized by severe clinical illness and high-titre viraemias. These animals are the most abundant large wild ungulate species in the US and this experimental study and computer modelling simulations suggest that this species could serve as an amplification host should RVFV be introduced.310, 660
Humans
Humans become infected from contact with infected tissues or from mosquito bites.102, 370, 403, 592. The majority of RVF infections in humans are inapparent or a transient influenza- like illness. It was recognized from the outset in Kenya in 1930 that the influenza- like illness in humans could be accompanied by transient loss of visual acuity, but the occurrence of serious ocular sequelae was first reported in the 1950/51 epidemic in South Africa.217, 234, 539, 540 A minority of patients develop ocular lesions, encephalitis or severe hepatic disease with haemorrhagic manifestations.242, 294, 340, 403, 410, 413, 499, 556, 565, 592, 633 Acute renal failure, with or without hepatic involvement, was described in patients in Sudan and Saudi Arabia.14, 181
After an incubation period of two to six days, the onset of the benign illness is usually very sudden and the disease is characterized by rigor, a fever that persists for several days and is often biphasic, headache, retro-orbital pain, photophobia, weakness, and muscle and joint pains. Sometimes there is nausea and vomiting, abdominal pain, vertigo, epistaxis and a petechial rash. Symptomatic improvement occurs in four to seven days in benign RVF and recovery is often complete in two weeks.
One of the most detailed descriptions of the clinical features associated with moderate to severe RVF was reported from patients admitted to the Gizan regional referral hospital during the first outbreak of the disease in Saudi Arabia in 2000. Among 165 patients who were treated and prospectively studied, the major clinical characteristics included hepatic failure (75.2 per cent of patients), acute renal failure (41.2 per cent), haemorrhagic manifestations (19.4 per cent), retinitis (9.7 per cent), and meningoencephalitis (4.2 per cent). Of those patients, 56 died (33.9 per cent), and there was no difference in case fatality rate with regard to the sex of the patients. Hepatorenal failure, shock, and severe anaemia were the major factors associated with death.14
Rare cases of miscarriage and stillbirth have been reported recently.8, 31, 49 Infected pregnant women had more severe bleeding, joint pain and malaise than their uninfected counterparts.49 Rift Valley fever virus appears to be capable of infecting both the syncytiotrophoblast and cytotrophoblast layers of the placenta, which may potentially result in the in utero infection of foetuses.404, 480 While recent experimental data highlight the threat of infection with RVFV to pregnant women and their unborn babies in endemic areas of Africa,406 more work is required to understand the underlying pathological mechanisms leading to miscarriages and/or abortions, epidemiological aspects and socio-economic burden of the disease in this group of at risk people.
There are numerous reports of humans becoming infected while investigating the disease in the field or laboratory.206, 215, 262, 295, 326, 386, 421, 453, 523, 543, 559, 572, 592, 647 Generally, persons who become affected are involved in the livestock industry, such as farmers who assist in dystocia of livestock, farm workers who salvage carcasses for human consumption, veterinarians and their assistants, and abattoir workers.82, 83, 102, 370, 403, 450, 466, 586, 592 The putative vectors in the outbreaks in Egypt in 1977/78 included Ae. caspius, Cx. pipiens, Cx. attenuatus and Cx. perexiguus, which are known to feed on livestock and humans,221, 223, 224, 262, 288, 379, 620 while the principal mosquito vectors of RVF in southern Africa tend to be zoophilic and sylvatic but it is thought that humans can become infected on occasion through transmission by Cx. theileri and Cx. zombaensis.
The results of an antibody survey following the 1974 to 1976 epidemic in South Africa revealed that 14.5 per cent of farm residents became infected, a remarkably similar proportion to the estimate of 10 to 15 per cent made for the 1950/51 epidemic.403, 542 Antibody was found in 9 per cent of farm residents in Zimbabwe following the 1978 epidemic.588 No outbreaks of the disease have been recognized in urban consumer populations and it is surmised that the fall in pH associated with the maturation of meat in abattoirs is deleterious to the virus.102, 586
Human infection presumably results from contact of virus with abraded skin, wounds or mucous membranes, but aerosol and intranasal infection have been demonstrated experimentally and circumstantial evidence suggests that aerosols have been involved in human infections in the laboratory, and in the field during epidemic.6, 30, 87, 167, 209, 215, 262, 317, 326, 403, 472, 479, 494, 543, 572, 658, 666 Low concentrations of virus have been found in milk and body fluids, such as saliva and nasal discharges of sheep and cattle, and although contact transmission is not considered important in livestock, it appears that there may have been a connection between human infection and consumption of raw milk.15, 44, 247, 252, 293, 336, 463, 466, 556, 649
Despite the intense viraemia that occurs in humans and the fact that virus has been isolated from throat washings, there are no records of person-to-person transmission of infection.215, 293, 556
In the minority of patients who develop ocular lesions these may occur at the time of the initial illness or anytime up to approximately four weeks later.122, 153, 217, 234, 274, 377, 413, 539, 540, 564, 565 It was estimated following the 1974 to 1976 epidemic in South Africa that ocular complications occur in up to 20 per cent of human infections,403 but estimates ranged from less than 5 per cent to less than 1 per cent for the Egyptian epidemic of 1977/78.340, 499 The ocular disease usually presents as a loss of acuity of central vision, sometimes with development of scotomas. The essential lesion appears to be focal retinal ischaemia, generally in the macular or paramacular area, associated with thrombotic occlusion of arterioles and capillaries, and is characterized by retinal oedema and loss of transparency caused by dense white exudate and haemorrhages. Sometimes there is severe haemorrhage and detachment of the retina. The loss of visual acuity generally resolves over a period of months with variable residual scarring of the retina, but in instances of severe haemorrhage and detachment of the retina there may be permanent uni or bilateral blindness.13, 461
Probably less than 1 per cent of human patients develop the haemorrhagic and/or encephalitic forms of the disease. The haemorrhagic syndrome starts with sudden onset of influenza-like illness similar to the benign disease, but within two to four days there may be development of petechial rash, purpura, ecchymoses and extensive subcutaneous haemorrhages, bleeding from needle puncture sites, epistaxis, haematemesis, diarrhoea and melaena, sore and inflamed throat, gingival bleeding, epigastric pain, hepatomegaly or hepatosplenomegaly and tenderness of the right upper quadrant of the abdomen and deep jaundice. This is followed by pneumonitis, anaemia, shock with racing pulse and low blood pressure, hepatorenal failure, coma and cardiorespiratory arrest.340, 413, 592, 633 A proportion of the less severely affected patients may make a protracted recovery without sequelae.
Signs of encephalitis in humans may supervene during the acute illness, or up to four weeks later and include severe headache, vertigo, confusion, disorientation, amnesia, meningismus, hallucinations, hypersalivation, grinding of teeth, choreiform movements, convulsions, hemiparesis, lethargy, decerebrate posturing, locked-in syndrome, coma and death. A proportion of patients may recover completely, but others may be left with sequelae, such as hemiparesis.340, 370, 413, 592, 633
The first known human fatality was recorded in 1934 in a laboratory worker in the USA soon after the initial isolation of the virus,543 but since the infection was complicated by thrombophlebitis and the patient died from pulmonary embolism the potential lethality of RVFV for humans was overlooked until seven fatal infections were recognized during the 1974 to 1976 epidemic in South Africa.403, 633 This was followed in short succession by reports of at least 598 human deaths in the Egyptian epidemic of 1977/78 and five deaths in the 1978 epidemic in Zimbabwe, and latterly by an estimate of at least 224 deaths in the 1987 epidemic in Mauritania.294, 408, 592 Since then fatalities in humans have been reported in many countries during outbreaks of RVF in livestock.340, 408, 412, 499
An antibody prevalence rate of 29.6 per cent was detected and the human population estimated at one to three million in the areas affected by the 1977/78 Egyptian epidemic.409, 556 Following the Mauritanian outbreak of 1987, 34.8 per cent of people in the town of Rosso were immune. It was estimated that there had been 9,320 infections in the town, of which only 1,013 had been symptomatic, with 47 deaths.292 Hence, the case fatality rate was 4.6 per cent, but the death rate in relation to total infections was only 0.5 per cent. An association between deaths in humans and increasing virus load and titre was found among patients hospitalized during the Saudi Arabia outbreak in 2000, suggesting that lack of immune control and rapid induction of innate immunity are potential determinants of poor patient outcome.373
Since the early 2000s, there have been reports of potentially increased case fatality among humans during outbreaks, sometimes exceeding 20 per cent.39 However, it is unclear if these reported increases are actually related to changes in virus virulence, underlying host-comorbidities, or surveillance bias due to more robust identification of severe cases during outbreaks.39, 66 Regardless, it is clear from large-scale serological surveys after epidemics and during inter-epidemic periods that the vast majority of RVFV infections in humans are self-limiting, and do not lead to lethal outcomes.
Table 1. Susceptibility of vertebrates to Rift Valley fever virus infection. (Information derived from sources cited in the text)
| EXTREMELY SUSCEPTIBLE (70–100 percent MORTALITY) | HIGHLY SUSCEPTIBLE (20–70 per cent MORTALITY) | MODERATELY SUSCEPTIBLE (LESS THAN 10 per cent MORTALITY) | RESISTANT (INAPPARENT INFECTION) | REFRACTORY (NOT SUSCEPTIBLE) |
|---|---|---|---|---|
| New-born lambs | Adult sheep | Cattle | Horses | Birds |
| New-born goat kids | New-born calves | Goats | Pigs | Reptiles |
| Puppies (experimental) | New- World camelids | Old- World camelids | Adult dogs (experimental) | Amphibians |
| Kittens (experimental) | New- World white tail deer | African buffalo | Adult cats (experimental) | |
| Mice | Asian buffalo | African green monkeys | ||
| Hamsters | South American monkeys | Baboons | ||
| Certain other rodents* | Rhesus macaques | Rabbits | ||
| Certain rodents* | Guinea pigs | |||
| Humans | Certain other rodents* |
Pathology
Even in benign disease in livestock there may be marked leukopenia during the first three to four days of infection, usually corresponding with peak fever and viraemia, and this may be followed by leukocytosis during early recovery.135, 167, 206, 595 At the same time there may be marked increases in serum levels of enzymes such as sorbitol dehydrogenase and glutamate dehydrogenase, which are indicative of hepatocyte necrosis, or aspartate aminotransferase, which is suggestive of hepatocyte degeneration, but gamma-glutamyltransferase levels remain normal, which is in keeping with the relative sparing of bile ducts in RVF, while alanine aminotransferase which is used as an indicator of liver damage in humans, is of no value in monitoring the disease in herbivores.
Prolonged clotting times have been recorded in mice,422 and thrombocytopenia in sheep with benign infection,595 but most information on haemostatic derangement in RVF comes from studies in rhesus monkeys.125, 497, 498 It was found that monkeys may have prolonged activated partial thromboplastin times and prothrombin times even in benign infection, and in severe liver disease there may be depletion of coagulation factors II, V, VII, IX, X and XII, thrombocytopenia and platelet dysfunction, increased schistocyte counts and depletion of fibrinogen (factor I) together with raised fibrin degradation product levels. Many of these findings suggest the occurrence of disseminated intravascular coagulopathy, a conclusion which is supported by the finding of fibrin deposits in glomeruli, intertubular vessels of the renal medulla and the spleen.125, 497 The findings in monkeys are compatible with the scant observations available on humans, except that leukocytosis and anaemia are marked in some patients.592, 633 The presence of fibrin thrombi in the liver, spleen, kidneys and lungs of sheep and cattle that succumb to RVF indicate that disseminated intravascular coagulopathy is also a feature of the disease in domestic ruminants.114, 116, 118, 119, 280
The hepatic lesions of RVF are essentially similar in all domestic animals and humans, varying mainly with the age of the affected individual.4, 114, 117, 118, 119, 135, 168, 170, 171, 206, 207, 280, 541, 633 The most severe lesions occur in aborted sheep foetuses and new-born lambs in which the liver is usually moderately to greatly enlarged, soft, friable and yellowish-brown to dark reddish-brown in colour with irregular congested patches and sometimes haemorrhages of varying size scattered throughout the parenchyma (Figures 2 and 3). Numerous greyish-white necrotic foci, 0,5 to 1,0mm in diameter, are invariably present in the parenchyma but may not be clearly discernible due to the discoloration of the organ. Fibrinous perihepatitis and subcapsular haematomas are seen occasionally. There may be oedema and haemorrhages in the wall of the gall bladder and hepatic lymph node. Icterus is evident in only about 10 per cent of affected new-born lambs because of the peracute course of the disease.
The hepatic lesions in adult sheep are generally not as severe or as widespread as in new-born lambs, but icterus may be more evident because of the less peracute nature of the disease. Pin-point reddish to greyish-white necrotic foci may be distributed throughout the parenchyma (Figure 4), and in a small proportion of sheep there are centrilobular haemorrhages and necrotic lesions which impart a mottled appearance (Figure 5) to the organ and render lobulation more distinct than normal.
Figure 4 Adult sheep: greyish-white foci of necrosis are scattered throughout the parenchyma of the liver
Haemorrhages and oedema of the wall of the gall bladder are common, and the lumen may contain a blood coagulum or blood-tinged bile (Figure 6).
The livers of aborted cattle foetuses resemble those of lambs, while the livers of calves and adult cattle are swollen, friable, discoloured orange-brown, and sometimes show congested areas and haemorrhages in the parenchyma with accentuation of the lobulation.118 While only scattered, greyish-white foci of necrosis, 0,5 to 1,0 mm in size, are seen in the livers of adult cattle, numerous foci are usually discernible in calves.
The hepatic lesions in new-born lambs are almost invariably accompanied by numerous petechiae and ecchymoses in the mucosa of the abomasum, and its contents are dark chocolate-brown as a result of the presence of partially digested blood (Figures 7 and 8). The contents of the small intestine may be similar in appearance. Most mature sheep and cattle have haemorrhages and oedema in the abomasal folds (Figure 9), and sometimes copious amounts of free blood in the lumen of the intestines. In most animals the spleen is slightly to moderately enlarged, with haemorrhages in the capsule. Infarcts are occasionally evident on the edges of the spleen in adult sheep as dark blueish-red, circumscribed areas, 10 to 20mm in diameter.116
In all animals the peripheral and visceral lymph nodes are enlarged, oedematous, and may have petechiae.
Other changes include widespread subcutaneous, serosal (Figure 10) and visceral haemorrhages, mild to moderate effusion of fluid, often blood-tinged, into body cavities, congestion and oedema of the lungs, enlargement of the adrenals with small haemorrhages in the cortex, and nephrosis which may be particularly severe in mature sheep. Haemoperitoneum is occasionally present in new-born lambs.
Hepatic necrosis is the most striking microscopic lesion of RVF in all domestic animals and humans.114, 117, 118, 119, 135, 167, 168, 206, 207, 265, 280, 468, 541, 633 In aborted sheep foetuses and neonatal lambs, primary foci of necrosis comprising dense aggregates of cytoplasmic and nuclear debris, some fibrin and a few neutrophils and macrophages can be discerned against a background of parenchyma reduced by nuclear pyknosis, karyorrhexis and cytolysis to scattered fragments of cytoplasm and chromatin, with only narrow rims of degenerated hepatocytes remaining reasonably intact close to portal triads (Figures 11 and 12). Destruction of hepatocytes may be so marked that most of the normal architecture of the organ is lost, giving the liver almost the appearance of lung tissue (Figure 11). Intensely acidophilic cytoplasmic bodies which resemble the Councilman bodies of yellow fever are common, and rod-shaped or oval eosinophilic intranuclear inclusions (Figure 13) are found in about 50 per cent of affected livers. Mineralization of necrotic hepatocytes may be evident as small purplish-blue cytoplasmic granules (Figure 14) in haematoxylin and eosin-stained sections in approximately 60 per cent of lambs, and bile casts are present in about 30 per cent of livers.114 Other noteworthy lesions in new-born lambs are pyknosis and karyorrhexis of lymphocytes in lymphoid tissues, cloudy swelling and hydropic degeneration of the epithelial cells of the convoluted tubules of the kidney and necrosis of some of the cellular elements in the glomeruli in ten per cent of lambs, and multifocal necrosis and haemorrhages in the adrenal cortex.114, 468
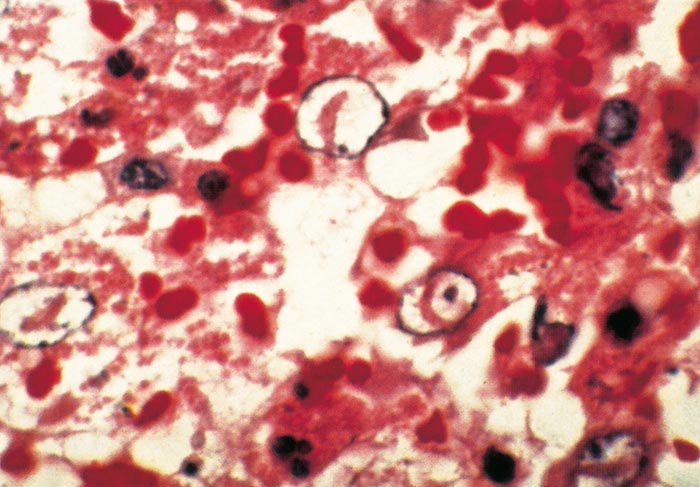
Figure 13 New-born lamb: note rod-shaped intranuclear inclusion bodies in necrotic hepatocytes. Haematoxylin and phloxin-stained section
In older sheep, liver necrosis is not usually as diffuse as in neonates and tends to be multifocally distributed (Figure 15) throughout the parenchyma and icterus is more common than in lambs. The hepatocytic changes in these foci are similar to those described for primary foci of necrosis in lambs. However, occasionally centrilobular necrosis and haemorrhage are also evident. Severe nephrosis which occasionally occurs in mature sheep is characterized by marked tubular degeneration and necrosis, intracytoplasmic accumulation of proteinaceous droplets in tubular epithelial cells and presence of hyaline casts in tubular lumens, while haemorrhages, necrosis and deposition of fibrin and haemorrhage may be present in glomeruli (Figure 16). A few acute subcapsular infarcts are occasionally present at the edges of the spleen and the sinuses may contain accumulations of excess fibrin.
Primary foci of necrosis are particularly numerous and conspicuous in the livers of affected calves (Figure 17) and aborted cattle foetuses, and are sometimes accompanied by massive hepatic necrosis, presenting a pattern similar to that described for new-born lambs.118, 168
Vasculitis, thrombosis and sinusoidal fibrin deposition are sometimes present in the livers of both adult cattle and calves. Apart from hepatic lesions, there is necrosis of lymphoid tissue, and numerous foci containing fibrin (fibrinoids) are sometimes evident in the red pulp of the spleen.
The extent of hepatocellular involvement in adult cattle varies greatly.118, 135, 168, 541
Marked centrilobular eosinophilic necrosis and haemorrhage, frequently extending to the middle of the lobule, occur in about 60 per cent of field cases of RVF, with individual or small groups of necrotic hepatocytes and acidophilic bodies being dispersed among the intact but degenerated hepatocytes in the rest of the lobule. Massive necrosis involving almost all hepatocytes (Figure 18) occurs in approximately 30 per cent of livers of field cases, while focal necrosis occurs in the remainder. The pathognomonic residual primary foci of necrosis, present in most new-born lamb livers,114 are sparse in the livers of mature cattle.118 Intranuclear inclusions, frequent in new-born lambs and aborted sheep and cattle foetuses, are less common in other animals and are only seen in about 20 per cent of livers of adult cattle. Other lesions in cattle are similar to those described for calves.
Many animals have lung congestion, alveolar and interstitial oedema, haemorrhages, a few fibrin thrombi in alveolar walls, scattered neutrophil infiltration, and necrosis of a few cells in alveolar walls, interlobular septa and peribronchial lymphoid tissue.
Varying degrees of lymphocyte necrosis have been described in the spleen and lymph nodes in sheep, cattle and humans.113, 118, 178, 190, 192, 469, 470, 471, 633 Necrosis of lymphocytes in the spleen of adult sheep is most apparent in the germinal centers of the white pulp. In young lambs and foetuses, the lymphocytes in the red pulp and the peripheral regions of the periarteriolar lymph sheaths are mainly involved. Karyorrhexis and karyolysis of the nuclei of lymphocytes in the spleen and lymph nodes as well as atrophy of the white pulp have been reported in fatal cases in humans.178, 633
In humans, encephalitis is characterized by focal necrosis with leukocyte infiltration and perivascular cuffing.633
Information on ultrastructural changes in RVF is scant and most reports deal with viral morphology and morphogenesis.119, 185, 186, 265, 344, 389, 455 The most conspicuous changes in hepatocytes include clumping and margination of chromatin, pyknosis, karyorrhexis, karyolysis, bizarre nuclear profiles, focal cytoplasmic degradation and sequestration with the formation of acidophilic or dark bodies (Figure 19), and cytoplasmic fragmentation or disintegration.119, 185, 186 Single or small groups of virions may be seen free among cytoplasmic organelles, inside smooth membrane-bound structures in a small proportion of hepatocytes, in cellular fragments and debris, or within sinusoids. Occasionally the viral particles may be associated with phagocytosed necrotic material within Kupffer cells, neutrophils or macrophages.119 Rod-shaped intranuclear inclusions (Figure 20), occupying up to half the length of the nucleus and composed of bundles of microfilaments or fibrils less than 10nm in diameter, occur in some infected cells.119, 185, 186, 265, 589 Sinusoidal endothelium is generally preserved but isolated endothelial cells may be necrotic late in the course of the infection.119
Diagnosis
Suspicion of RVF should be aroused when heavy rains are followed by the occurrence of abortions in sheep, cattle and goats together with fatal disease, particularly in young and pregnant animals, which is marked by necrotic hepatitis and widespread haemorrhages. Frequently, there is also an influenza-like illness in farm workers and others with contact to infected animal tissues.
The usually regional nature across national borders of RVF outbreaks and involvement of multiple species requires an integrated One Health approach to outbreak investigation, diagnostics, and counter-measure responses.313, 331, 429 Due to the severe potential economic impacts of RVF outbreaks in animals, animal health professionals should familiarize themselves with the specific guidelines and requirements listed in the World Organization for Animal Health (OIE) Manual for diagnostics and regulatory reporting requirements.474
Field biosafety and biosecurity concerns for veterinarians and other health personnel
Rift Valley fever virus has proven on multiple occasions to be easily transmitted to people from infected animal tissues and body fluids in farm settings. If RVFV is suspected among livestock, standard biosafety and biosecurity protocols and procedures should be followed to avoid infection of animal health workers and other personnel involved in the collection of diagnostic specimens. At a minimum for on-site field investigations, gloves, eye/face protection, and dedicated field clothing and boots should be worn and thoroughly disinfected with appropriate disinfectants labelled for virucidal activity against lipid-enveloped viruses. Ideally, when handling suspected RVFV infected animal carcasses or materials, personal-protective equipment (PPE) and practices should be supplemented by disposable coveralls and respiratory protection such as properly fitted N95 respiratory masks. Health workers should consult their national agricultural and public health authorities for proper guidance related to PPE and safe sampling procedures relevant for local conditions and practices.
Work with live infectious virus, non-inactivated clinical specimen materials, and experimentally infected animals should be conducted in laboratories (i.e., BSL-3 or higher) with appropriate biosafety facilities and PPE practices.197
Specimen collection
Specimens to be submitted for laboratory confirmation of the diagnosis include either EDTA, heparinized or clotted whole blood, plasma or serum of live affected animals or tissue samples, including liver, spleen, kidney, lymph nodes and heart blood of dead animals. Samples from aborted foetuses should include brain since this is usually less autolysed or putrified than viscera when obtainable.560, 586 Placenta specimens, if available, can also be collected.
Specimens should be securely packaged and submitted at 4 °C to a suitable laboratory for isolation or detection of virus or demonstration of antibody. Where delay in getting specimens to the laboratory is unavoidable or the material has to be transported at ambient temperature, tissue samples can be preserved in glycerol-saline solution. Tissue specimens from the liver, spleen, lymph nodes and kidney should also be collected in 10 per cent buffered formalin for histopathological and immunohistochemical examination. In animals that survive the disease, paired serum samples, one taken during the acute illness and the other two to three weeks later, should be submitted for antibody tests.
Diagnostic assays
Molecular detection assays: The frontline diagnostic assays routinely employed in centralized diagnostic laboratories are nucleic-acid amplification tests such as real-time reverse transcription polymerase chain reaction (RT-PCR), quantitative real-time PCR, or whole genome sequencing.60, 162, 456, 652, 661 These detection assays require moderate laboratory sophistication and technology, but are routinely available in most regional diagnostic centres. Multiple broadly reactive assays have been described and are routinely used to rapidly and accurately identify RVFV genome in a wide variety of submitted specimens (e.g. blood, plasma, serum, homogenized tissues, and other fluids). Viral nucleic acid can readily be detected in serum and other tissues of infected humans and livestock, as well as mosquitoes, by RT-qPCR.162, 226, 269, 301, 303, 528 and real-time reverse transcription-loop-mediated isothermal amplification assay.342
Antigen detection assays: RVFV antigens can be detected by immunofluorescence and immunocapture ELISA assays. Zaki et al.284, 676 reported an immunofluorescence assay, that utilizes a pool of mouse IgG monoclonal conjugates for the detection of RVFV-specific antigens. Although it was shown to be highly sensitive in detection of RVFV in patient sera, it’s use requires tissue culture amplification and handling of live virus. Jansen et al.284 developed a safe sandwich ELISA (sAg-ELISA) for the detection of RVFV NP antigen in specimens inactivated at 56 °C for 1 h in the presence of 0.5% Tween-20 (v/v) before testing. The test can be used for the detection of RVFV NP antigen in human and animal sera, homogenates of liver and spleen tissues of domestic and wild ruminants, and mosquito homogenates. The lateral flow immunochromatographic test for the detection of RVFV NP in animal sera and fluids from aborted foetuses is a valuable diagnostic tool for onsite rapid detection of RVFV.101, 284
Virus isolation and culture: Long considered the gold-standard of virus diagnosis, this technique requires relatively high levels of biosafety and biosecurity and has become less favoured than molecular diagnostic assays. However, obtaining high quality virus isolates from field specimens is essential for further research work and development and should be considered where adequate laboratory facilities and trained staff are present. Rift Valley fever virus can be isolated readily in a variety of cell cultures) including Vero, CER, BHK21, mosquito line cells, and primary calf, lamb and goat kidney or testis cells, or in suckling and weaned mice or hamsters inoculated intracerebrally or intraperitoneally.22, 136, 157, 167, 410, 499, 556, 586, 594, 649 Cytopathic effect (CPE) may be evident one to five days after inoculation of cell cultures, but virus can be identified and the diagnosis accelerated by performing immunofluorescent (IF) tests on cultures at 24 hours or even earlier and it is always necessary to perform IF tests on inoculated mosquito cells since CPE is not readily apparent in them. More commonly, virus isolated in cell cultures is identified with reference antiserum by performing neutralization tests.166, 594 Inoculation of suckling mice by the intracerebral route is used extensively for isolation of RVFV,586 and mice generally die two to five days after inoculation with field material.
The virus can be grown in and readily produces cytopathic effect and plaques in virtually all common continuous line and primary cell cultures, including Vero and BHK21 line cells, primary calf and lamb kidney or testis cells, the only major exceptions being primary macrophages and lymphoblastoid cell lines.496 Virulent virus readily gives rise to chronically infected cell cultures derived from a proportion of cells which escape cytopathic effect, the phenomenon being associated with production of defective RNA in all three segments of the genome.58 The virus can be grown in embryonated chicken eggs and a variety of laboratory animals including suckling or weaned mice and hamsters inoculated by intracerebral or intraperitoneal routes. Lambs are extremely susceptible, but some strains of rat are resistant, as are rabbits, guinea pigs, horses, and chickens.167, 410, 499, 556
Infectivity of virus stocks is highly sensitive to temperature and storage conditions. Controlled studies of the Clone 13 vaccine strain revealed that virus particles retain infectivity in serum and can be recovered after prolonged (weeks to months) storage at 4 °C; however increased temperature rapidly degrades virus particles and infectivity is lost within hours at 56°C.128 The virus is very stable at temperatures lower than –60°C or after freeze drying. It is inactivated by lipid solvents, low concentrations of formalin, and is very sensitive to acidic conditions with infectivity rapidly lost below pH 6.8.556
Microscopic pathology and immunohistochemistry: Histopathological lesions are very characteristic and, in particular, the liver lesions of new-born lambs and kids leave little room for doubt about the diagnosis of RVF.114, 117, 118, 119, 135, 167, 168, 206, 207, 265, 280, 541 Virus antigen can be detected in tissues of infected animals by immunohistochemistry.468, 630
Antibody detection assays: Antibody to RVFV can be demonstrated by different assays including ELISAs, multiplex-bead based arrays, indirect immunofluorescence, and virus plaque reduction neutralization test (PRNT).69, 110, 166, 175, 335, 465, 487, 504, 511, 549, 563, 574, 594, 631 All techniques except neutralization tests can be performed with inactivated antigens to enhance biosafety in the laboratory. Although regarded as a gold standard, virus neutralization assays are laborious, expensive, and require 5-7 days for completion. They can be performed only when a standardized stock of live virus and tissue cultures are available. Consequently, they are rarely used, and only in specialized laboratories equipped with high biocontainment facilities. Recently, a neutralization test based on an avirulent RVFV expressing an enhanced green fluorescent protein was developed and reported to be more sensitive than the classical neutralization test and safe to be used outside level 3 biocontainment facilities.538
Various ELISA formats have been developed and validated in recent years for specific detection of anti-RVFV antibodies in humans and animals, based on inactivated sucrose-acetone-extracted antigens derived from tissue culture or mouse brain.488, 489, 493 While they were shown to have high diagnostic specificity and diagnostic sensitivity compared to virus neutralization assays, the production of antigen for these assays also requires bio-containment facilities to limit the risk of exposure of laboratory personnel to infection. To address these problems, indirect ELISAs based on the recombinant NP and GP proteins have been developed for the detection of anti-RVFV antibodies in domestic ruminants, wildlife and humans.193, 281, 285, 491, 492 An ELISA platform based on recombinant NP and NSs proteins is able to distinguish infected from vaccinated animals. This diagnostic capacity is important for allowing moving of vaccinated animals, given the strict regulations for movement and export of animals.388
Since a wide range of domestic and wild animal species are susceptible to RVFV, the development of inhibition and competitive ELISAs provide an additional advantage of allowing multi-species RVFV antibody detection using the same diagnostic procedure without requirements for species-specific conjugates.493, 625
Recent or current infection must be distinguished from pre-existing immunity and, conventionally, serological diagnosis of recent disease is confirmed by demonstrating seroconversion or a four-fold or greater rise in titre of antibody in paired serum samples.563 The introduction of the IgM-capture ELISA for detection of antibody to RVFV in humans and domestic ruminants, however, allows diagnosis of recent infection to be made on a single serum sample.335, 465, 488, 489
Results obtained in experiments with sheep indicate that neutralizing antibody may become demonstrable as early as three days following infection and by the fourth to sixth day antibody is detectable by ELISA. Antibody titres detected by ELISA are maximal from two weeks to approximately six months after infection.594 Thereafter ELISA titres decline over a period of several years, while neutralizing antibody tends to maintain a plateau level after an initial post-convalescent decline and probably remains demonstrable for the life of the individual. Kinetics of IgM and IgG responses were studied in sheep experimentally infected with wild-type RVFV and in sheep vaccinated with live-attenuated Smithburn strain of RVFV. IgM and IgG seroconversion was detectable 4-6 days post-infection with IgG antibody reaching the highest level on Day 21 and IgM antibody on Day 11 after infection. Compared to sheep infected with wild-type virus, the IgG and IgM responses in vaccinated animals were less rapid and antibody levels and especially those of IgM were lower. IgM antibody could be detected only in 12.5 per cent of infected sheep 72 days post-infection.488 The time dependent detectability of anti-RVFV antibody, and especially IgM, may be of significance for epidemic situations, where the disease stage may affect the outcome of an assay result and interpretation of the data. The only information available on the duration of the IgM antibody response following natural infection relates to 195 cattle monitored after the 1991 epidemic in Madagascar; less than 30 per cent had IgM antibody two months after infection was diagnosed, and all were negative by six months.441
Theoretically, the antigenic cross-reactivity of the phleboviruses could present problems in making a serological diagnosis of RVF infection and for this reason neutralization tests, which are least affected by serological cross-reactivity, would seem to be most suitable for confirmatory diagnostic use.410, 561, 562, 598, 599
Establishing a definitive diagnosis in cases of teratology caused by the live modified RVF vaccine virus can be very difficult. Several viruses, other agents, and plant toxins can cause similar teratology. Foetuses may not be immunocompetent at the time that they undergo teratogenic infection and thus may not have virus or antibody at the time that the malformations become apparent, thus limiting diagnostic options for specific testing. Regarding serological diagnoses of new-born animals, those that are able to suckle acquire maternal IgG but not IgM antibody from milk ingestion. Compounding this, dams of affected foetuses will generally no longer have IgM antibody by the time that malformations become apparent. Identification of the teratogenic agent may need to rely heavily on risk-factor analysis and epidemiology to support a tentative aetiological diagnosis.
Differential diagnosis
Rift Valley fever, Wesselsbron (WSL) disease and other arthropod- borne virus diseases such as Schmallenberg virus tend to occur under the same climatic conditions (see Wesselsbron disease). Both RVF and WSL viruses can cause mortality in lambs, kids and calves and occasionally abortion in ewes, but RVF is associated with much higher mortality and abortion rates.114, 120, 121, 649 In experimental infection, RVF can produce a mortality rate of 90 per cent or greater in neonatal lambs, while WSL disease is lethal in about 30 per cent of lambs. Moreover, RVF infection produces severe disease and deaths in adult sheep and goats while WSL disease is usually inapparent in these animals. In the field, outbreaks of disease in sheep were ascribed to WSL disease virus shortly after its original isolation in South Africa in the 1950s, but some of the outbreaks were complicated by plant or copper poisoning,53, 343 and since then isolations of the virus from livestock have been few and sporadic. It is pertinent that WSL virus has never been isolated from an aborted foetus from the field. On the other hand, misuse of live modified WSL disease vaccine in pregnant animals during the 1974–76 RVF epidemic in South Africa and Namibia is believed to have caused foetal and neonatal teratology on a large scale.115
Definitive diagnosis of RVF and WSL disease ultimately depends on virological and serological examination of appropriate specimens but the diseases can also be distinguished on the basis of the hepatic changes found in each.114, 121 Lesions in the livers of young animals that succumb to WSL disease are usually less extensive than those in RVF, and tend to be less acute as evidenced by the occurrence of bile stasis, bile ductular and Kupffer cell proliferation, and hepatocellular regeneration. Hence moderate to severe icterus is a regular finding in WSL disease, whereas it is absent in most neonates which succumb to peracute massive hepatic necrosis in RVF infection. Although there may be extensive hepatic necrosis in WSL disease, neither the parenchymal hemorrhages nor the primary necrotic foci characteristic of RVF are evident macroscopically or microscopically in WSL disease. In both diseases, mucosal haemorrhages and free partially digested, chocolate-brown blood are found in the abomasum.
Agents causing mortality associated with hepatic lesions, haemorrhages and/or icterus which may superficially resemble RVF in domestic ruminants include poisonous plants affecting the liver in southern Africa such as Senecio spp., Crotalaria spp., Lasiospermum bipinnatum, Cestrum spp., Pteronia pallens and Hertia pallens, and an alga, Microcystis aeruginosa,318 as well as bacterial septicaemias such as pasteurellosis, salmonellosis (calf paratyphoid) and anthrax. Generally, domestic ruminants only eat toxic plants if forced to do so by scarcity of feed. Beyond southern Africa, peste des petits ruminants, Nairobi sheep disease, Schmallenberg virus, and possibly Thogoto virus infection148 could be confused with RVF. Diagnoses can be established in poisonings by instituting appropriate epidemiological and toxicological investigations, and in infectious diseases by means of appropriate microbiological and serological investigations.
Sometimes abortion is the only sign of RVF in cattle and diseases, which must be eliminated by appropriate laboratory investigations, include brucellosis, leptospirosis, chlamydiosis, Q fever, and salmonellosis. In instances where teratology is involved, investigations should include Schmallenberg virus, bluetongue virus, Simbu-serogroup bunyaviruses, Palyam serogroup orbiviruses, WSL and other flaviviruses such as West Nile and Banzi.46, 591 (see Bluetongue, Palyam serogroup orbivirus infections, and Diseases caused by Akabane and related Simbu-group viruses).
Rift Valley fever belongs to a group of viral haemorrhagic fever(VHF) diseases with worldwide distribution, but their specific aetiological agent is usually restricted to a known endemic region depending on the presence of natural reservoirs or/and competent arthropod vectors. As for most VHFs, the non-specific clinical manifestations of RVF makes it difficult to diagnose clinically. Therefore, the differential diagnoses of RVF in humans include a broad spectrum of conditions especially when first cases are encountered during a yet unrecognized outbreak. These include malaria, rickettsial infections, Q fever, typhoid fever, dysentery, plaque, brucellosis, leptospirosis, meningitis, sepsis from bacterial infections, viral hepatitis, other VHFs such as Lassa fever, Crimean Congo haemorrhagic fever, Marburg disease, Ebola fever, and the haemorrhagic fever associated with the renal syndrome caused by hantavirus infections. Non-infectious causes of disseminated intravascular coagulopathy and acute leukaemia should also be considered. Availability of laboratory results and epidemiological information usually help to narrow the spectrum of differential diagnoses. A provisional diagnosis can sometimes be made based on recent travel and exposure history (e.g., mosquito bites, or contact with animals or their tissues or products in endemic regions. An aetiological diagnosis is usually possible by considering all available laboratory results, and clinical, pathological, and epidemiological data. Rift Valley fever in humans should be suspected when there is a sudden outbreak of febrile illness with headache and myalgia in humans, in association with the occurrence of abortions in domestic ruminants and high mortality of young animals such as lambs and kids. Cases of RVF are sometimes only recognized weeks after infection by the occurrence of ocular complications. Haemorrhagic or encephalitic clinical manifestations may be present occasionally in residents of RVF-free countries following a visit to endemic areas.485, 593
Control
Vector control
The large number of mosquito species that can transmit RVFV and the vast number of breeding sites, particularly after heavy rains, makes it difficult to control mosquitoes on a large scale.38, 41 Mosquito control during outbreaks is in most instances thus not practical and is almost never applied. Strategically-timed application of larvicides can be used to suppress mosquito breeding. Other measures, such as chemical or biological control of adult vectors (see Vectors: Mosquitoes- a southern Africa perspective), movement of stock from low-lying areas to well-drained and wind-swept pastures at higher altitudes, or confining of animals to mosquito-proof stables, can be challenging to implement in the face of a RVF epidemic.7, 159, 375, 448, 518 In the case of dairy farms, the milking sheds should be made mosquito proof, and perhaps Culicoides-proof, and monthly spraying of their interior walls should be carried out with a residual insecticide other than an organochlorine. Organophosphate insecticides, such as malathion, fenthion (Baytex), fenitrothion (Sumithion), or carbamates such as carbaryl (Sevin) and propoxur (Arprocarb), can be used.552
Recent advances in genetic manipulation of mosquito populations through so called “gene drives” are potential mechanisms that could in the future be utilized to reduce the population level of mosquito vectors for a variety of arboviruses and other pathogens (e.g. malaria) if technical and bioethical hurdles can be overcome to allow for widespread use.372, 481
One Health approaches, vaccination and therapeutics
The central role of particularly viraemic livestock as a risk factor for human infection highlights the need for comprehensive One Health control strategies.65, 66, 67, 203, 331 For the successful long-term control and management of RVF in endemic areas, a multifaceted approach is needed that can reduce the level of virus circulation and amplification among mosquito vectors, livestock, and ultimately humans. Targeting one of the key amplification components, namely livestock, of the RVFV epidemic cycle could eliminate some of the greatest risk factors for human infection while at the same time greatly improving the economic and nutritional well-being of livestock producers and animal product consumers. Critical for this control approach to be successful will be the ready availability of inexpensive and effective vaccines that can be used safely in livestock or humans regardless of pregnancy and age status. Fortunately, the past 10 years have witnessed a resurgence in the development of a wide variety of novel vaccine candidates and therapeutics.35, 67, 165, 191, 279, 331, 383, 505, 535
Importantly, many of these candidate vaccines allow for the differentiation of naturally infected from vaccinated animals (DIVA) on the basis of serological testing. The capacity for pen-side DIVA testing of animals would allow compliance with international trade restrictions that are routinely imposed during RVF outbreaks and that further exacerbate the negative economic consequences of the disease. The key challenge ahead is moving these prototype vaccines from the laboratory into field trials, then past regulatory approval and eventual commercial viability.
Vaccines – historical and recent approaches
Vaccines have been an important component of routine management and emergency outbreak responses to RVF since the 1950s.63, 67, 191, 260, 273, 279, 494 The impact and cost-effectiveness of livestock vaccination have been recently assessed in RVF high-risk areas or epidemics using simulation modelling that demonstrated that animal vaccination is the most effective control measure, preventing both human and livestock cases.220, 325, 417 The renewed interest in RVF since its emergence in the Arabian Peninsula in 2000 and re-emergence of large RVF outbreaks in Africa in the last two decades have resulted in significant advances in our basic knowledge of RVFV replication strategy and virulence factors. This progress benefits and aids the development of novel vaccines both for humans and animals, including genetically modified-live vaccines, recombinant protein vaccines, DNA vaccines, virus-like particles (VLPs), virus replicons and virus-vectored vaccines165, 191, 198, 271 and the development antiviral drugs.459, 550 Considerable efforts have also been made in the establishment of reliable RVF challenge models for testing of potential vaccines and therapeutics.333
Wild-type RVFV, also referred to as hepato-, viscero-, or pantropic virus, has been attenuated to produce vaccine or candidate vaccine strains by serial passage in highly permissive systems such as suckling mice or cell cultures, by selection of small plaque variants and by chemical mutagenesis with 5-fluorouracil. The 5-fluorouracil94, 445, 571 mutagenized MP12 candidate vaccine virus has mutations in all three segments of the genome.527, 637 The Smithburn vaccine strain was developed by serial intracerebral passage of a Ugandan wild-type virus in suckling mice until it lost its tropism for the liver and became neuroadapted, i.e. until it no longer killed mice following peripheral inoculation yet retained its intracerebral lethality and its immunogenicity.571 Clone 13, a naturally occurring and highly attenuated mutant obtained by plaque selection of clones from a human isolate of RVFV from the Central African Republic, proved to have a 549 nucleotide deletion comprising 69 per cent of the NSs protein gene in the S RNA segment of the genome.379 The thermostable RVFV Clone 13 vaccine (CL13T) has been developed via a selection of viable virus populations at 56⁰C from the culture supernatant of Vero cells infected with the original Clone 13 strain of the virus, and then lyophilized in the presence of a stabilizer.128
Attenuated Smithburn strain and formalin- inactivated vaccines: The mouse neuroadapted Smithburn strain of RVFV is used at slightly different passage levels in laboratory host systems for production of livestock vaccines in South Africa and Kenya, while wild strains of RVFV are used for preparation of formalin-inactivated cell culture vaccines in South Africa and Egypt.15, 43, 45, 96, 112, 180, 316, 571, 649, 650 The Smithburn strain, received from Uganda after 82 intracerebral passes in suckling mice, was subjected to further passaging in mice and embryonated eggs in South Africa before being issued as an experimental vaccine in 1951. After minor adjustments in passage level were made during the following few years the strain was used until 1958 in a form which had undergone 102 intracerebral passages in suckling mice, followed by 50 egg passages and a further 16 mouse passages,and then provided as freeze-dried 10 per cent mouse brain vaccine. In 1958 reversion was made to seed virus of lower mouse passage level and since 1971 the same virus has been propagated in BHK21 cells for preparation of freeze-dried vaccine.43, 45, 316, 649, 650 The Smithburn strain has been used at the 106 intracerebral mouse passage level to produce vaccine for sheep and cattle in Kenya since 1960.96, 112
In contrast to the Smithburn vaccine, formalin-inactivated vaccines are safe for use even in pregnant animals, but they are expensive to produce and induce short-lived immunity, so that the administration of annual booster doses is necessary to ensure adequate protection.
The modified live Smithburn vaccine can readily be produced in large quantities, is inexpensive, and induces durable immunity in sheep six to seven days after a single inoculation, but in a proportion of pregnant animals it may cause abortions or teratology of the central nervous system of the foetus and hydrops amnii and prolonged gestation in the dam.115 The virus is, therefore, only partially attenuated and its use in pregnant animals should only be contemplated in the face of an epidemic when its adverse effects may be outweighed by the dangers of allowing the disease to take its natural course.137 It is considered theoretically possible, although not proven, that the virus could revert to full virulence if passaged through hosts such as mosquitoes which become infected as a result of feeding on animals in the viraemic stage following administration of the vaccine, and hence it is advised that inactivated vaccines should be used in situations where it is considered necessary to immunize animals in countries where the presence of RVF has not been proven.34, 621
The Smithburn RVF vaccine induces a poor antibody response in cattle,43, 45, 112 and they should preferably be immunized with formalin-inactivated vaccine to ensure that cows are able to confer colostral immunity to their offspring. Cattle should receive a booster dose three to six months after initial vaccination, followed by annual boosters before the rains are due, since immunity only lasts about one year.45 Formalin-inactivated vaccine can also be used in pregnant sheep to avoid abortion and foetal teratology.
Inactivated vaccines are normally used in non-endemic RVF countries.38, 467
MP12: The mutagen-derived MP12 candidate vaccine virus generated after 12 passages of the parental ZH548 strain (Egypt 1978) in the presence of the mutagen 5’-flurouracil was shown to be safe and effective for use in pregnant sheep and cattle, and has even been inoculated directly into bovine foetuses without apparent deleterious effects.47, 265, 432, 436, 437 However, the experiments were conducted with animals beyond the first trimester of pregnancy, after organogenesis has occurred when foetuses are less susceptible to the teratogenic effects of attenuated viruses. Teratology occurred in 14 per cent of lambs of sheep inoculated at an earlier stage of pregnancy.267 However, the MP12 vaccine has proven to be a safe and effective animal and human vaccine among non-pregnant individuals and recently was successfully tested in small scale Phase I/II human clinical trials.508, 509 Further iterations of the MP12 vaccine include recombinant- based deletions of the NSm nonstructural protein and codon usage deoptimization strategies.271, 369
Clone 13 vaccine: It is a live-attenuated vaccine based on a naturally attenuated strain (RVFV 74HB59) that was isolated in the Central African Republic from a RVF patient. It lacks 69 per cent of the NSs gene, a major virulence factor of the virus.452, 597 Experimental and field trials have shown that a single dose of the vaccine was highly immunogenic in domestic ruminants and did not cause abortion or foetal teratology in pregnant animals164, 363, 452, 466, 597 and conferred protection in calves against abortion on challenge with wild virus.638 Available data indicate that the Clone 13 vaccine did not cause detectable viraemia in vaccinated ruminants, thus minimizing the risk of virus transmission to the foetus or to mosquitoes and genome segment reassortment events with wild- type viruses. Since 2010, Clone 13 vaccine has been registered in South Africa, Namibia, Botswana, Zambia and Mozambique. More than 28 million doses of the vaccine have been used in South Africa by 2018, with more than 10 million doses used during the 2009-2010 RVF outbreaks.638 However, a recent report indicated that Clone 13 virus could cross the ovine placental barrier and be associated with foetal infections, malformations, and stillbirths when administered in an overdose to pregnant ewes at 50 days of gestation.378 Nevertheless, the Clone 13 vaccine at the recommended dose is considered to be safe and efficacious in domestic ruminants.129, 130
DDvax: The recombinant DDvax candidate was generated by the targeted deletion of the NSs and NSm virulence factors on the S and M genome segments of the parental ZH501 strain (Egypt 1978), respectively. This construct has proven safe and effective in a wide variety of animal species including sheep, cattle, non-human primates, and pregnant ewes vaccinated in the first trimester of gestation.59, 64, 569 While highly attenuated in mammals, this vaccine also was found not to be transmitted by mosquito vectors due to an inability to cross the midgut barrier caused by the deletion of the NSm gene and the associated gene products.126, 309
Four-segmented RVFV (RVFV-4s) – Bunyavax: An alternative approach to attenuation was pursued by splitting the RVFV M segment that contains the Gn and Gc glycoprotein coding regions into two separate genomic fragments using a recombinant plasmid-based system.653, 655, 656 Initial results were promising with marked attenuation and safety in a variety of animal species including rodents and livestock.653, 656 Later generation versions of this construct also contain a deletion of the major virulence factors the NSs gene on the S segment coding region likely further enhancing the safety of this vaccine candidate.
Virus-like particles and non-spreading replicon particles (multiple approaches): While virus-like particles (VLPs) are essentially virus-like shells comprised of virus glycoproteins and nucleoprotein, the more advanced concept of non-spread replicon particles seeks to capitalize on the safety advantages of killed vaccines with the inherent immunological boost derived from de novo virus genome replication and protein synthesis. Multiple groups of scientists have established VLP and replicon systems, some with the ability to undergo multiple rounds of genomic synthesis and replication, but without the ability to produce progeny virus-like particles.56, 158, 204, 256, 279, 332, 337, 454, 478, 654 These non-spreading virus-like or replication competent particles have proven to be safe and immunogenic in limited animal model studies, but could provide a mechanism for robust and rapid protection especially in non-endemic or at-risk countries.
Challenges to vaccination acceptance
It is advisable in African countries with large sheep and goat populations to immunize the offspring of vaccinated ewes and nannies on a regular basis at six months of age, when colostral immunity has waned, with a single dose of the modified live Smithburn vaccine. This should afford life-long protection.34 Lambs and kids of susceptible unvaccinated dams can be immunized at any age.650
Epidemics of RVF tend to occur at irregular intervals of many years, and it is usually difficult to persuade farmers to vaccinate livestock during long inter-epidemic periods when no apparent losses are occurring to justify vaccination costs. Compounding the sometimes long inter-epidemic periods is that the occurrence of epidemics is difficult to predict and outbreaks typically have a very sudden onset. Ideally, in African countries where RVF is endemic, large sheep and goat populations should be immunized routinely. The offspring of previously vaccinated livestock could be vaccinated on a regular basis at six months of age, when colostral immunity has waned, with a single dose of a modified live vaccine. Typically, this class of vaccines should provide life-long protection.34
Theoretically, the timing of livestock breeding programmes could be altered to avoid the worst effects of the disease on pregnant or young stock,135 but RVF epidemics occur too irregularly to make this feasible and other considerations such as the state of pastures in various seasons possibly have a more important bearing on breeding policy.
Due to the sporadic nature of RVF outbreaks, it is difficult to obtain the necessary political or donor support for routine or strategic RVF vaccinations for livestock. The burden of RVF in endemic countries has also not been well determined. To gain political support, research that demonstrates the long-term benefits of routine RVF vaccinations of livestock for public health, economic benefits, and threat reduction is critical. Economics likely remain a key determinant for RVF vaccine development and use. The infrequent nature of RVF disease outbreaks jeopardizes the incentive for manufacturing companies to produce and stockpile vaccines. The vaccine price could be the single most important constraint to livestock producers in developing countries to adopt routine or strategic livestock vaccination.165, 191, 271 The use of multivalent vaccines that could provide immunity against a prevalent disease (preferably for which vaccination is mandatory) while immunizing against other diseases of more sporadic nature, such as RVF would be a potentially acceptable strategy to overcome problems related to vaccine production costs. Bivalent recombinant virus-vectored vaccines designed to protect against RVF and lumpy skin disease573, 641 and RVF and bluetongue92 have been developed and were shown to be effective in mice and sheep.
No RVF vaccine has been authorised for use in the EU Member States: only emergency vaccination is permitted after authorization and following proper EU procedures.38
Vaccination of at-risk humans
It is essential that humans living in endemic areas and especially veterinarians and others engaged in the livestock industry should be made aware of the potential dangers of exposure to zoonotic agents in handling tissues of diseased animals, and that biosafety and biosecurity precautions should be increased during RVF epidemics. Currently only one human vaccine is approved for emergency use in humans. It is a formalin-inactivated cell culture vaccine (TSI-GSD 200) produced in the USA that is used only to immunize laboratory and field workers who are regularly exposed to RVF infection such as during outbreaks.172, 314, 407, 464, 500, 512, 513, 514 The vaccine induces low antibody tires, but human volunteers immunized with three doses administered on Days 0, 7 and 28, and given one booster, remained seropositive for up to eight years, while rats had partial immunity to aerosol infection six months after immunization.20, 216, 507
Recently, efforts to promote the development of licensed RVFV vaccines for humans has gained momentum.100, 414 These efforts led by the World Health Organization and the Coalition for Epidemic Preparedness Innovations have united multiple international aid organizations to provide funding and organized efforts to enable several experimental RVFV vaccines to progress towards human safety and efficacy assessment. With this renewed prioritization it is highly likely that the further development of next-generation vaccines for humans will be achievable in the near future. Vaccines lacking infectious genetic material (e.g. recombinant protein-based, VLPs) and replication-deficient virus vectored or replicon vaccines with a high safety profile could be the most appealing in getting regulatory approval for use in humans. Due to the rigorous regulatory requirements and approval process required for human vaccines, the mass vaccination of human populations against RVF may not be a viable option. A more rational strategy would be to vaccinate at risk personnel or groups: farmers, herders, veterinarians, slaughterhouse workers, and laboratory and research staff, and military and non-military personnel for deployment in RVF endemic risk zones.191
Surveillance and forecasting of outbreaks
Surveillance can be planned with different objectives and approaches and can include forecasting, early warning, raising awareness, outbreak investigations, environmental surveillance, vector surveillance and sentinel herd monitoring.199, 203
Three surveillance methods for RVF, namely, syndromic surveillance, participatory surveillance and risk-based surveillance are recommended by the OIE. The focus of syndromic surveillance is on the detection of clinical disease such as abortions, mortality in livestock and wildlife particularly in newborn and young lambs, kids or calves especially during favourable weather conditions conducive for vector transmission of RVFV. Participatory surveillance focuses on how best animal owners can be helped to identify and solve their health problems and to better understand the risks of RVF. Risk-based surveillance is the surveillance of locations, populations, and periods with increased disease threats with the aim to improve disease detection rapidly and therefore better use of resources. Risk-based surveillance uses quantitative or qualitative information that helps in disease control.199, 203
Sentinel herds are particularly useful to detect inter-epidemic transmission of RVFV.199 The activities at sentinel sites should include clinical observations and serological monitoring of herds, measurement of rainfall and other climatological parameters and vector sampling and surveillance.199
Recently, epidemiological models have become available to investigate the various aspects of the epidemiology of RVF.127, 416, 575, 604 with the aim to get a better understanding of the factors at play among vectors, hosts and the environment. Predictive models have been developed to act as early warning systems for RVF outbreak occurrence so that surveillance systems can be set up to detect RVFV circulation in mosquitoes and livestock.28
Attempts have been made to utilize satellite imaging to predict RVF outbreaks through the development of green vegetation indices or through monitoring of southern Pacific and Indian Ocean surface temperatures.27, 142, 229, 352, 354, 355 A close association between epidemic outbreaks of RVF in East Africa and the occurrence of El Niño has been described.29 NASA developed an outbreak risk mapping system that records monthly RVF risk for Africa and the Middle East based on interpretation of satellite vegetation indices.308
New approaches for the establishment of early warning systems for RVF should include monitoring of climatic data of different environments.99, 239 The objective of early warning systems is to provide National Veterinary Services with risk information in order to better plan in advance the resources and the activities to be put in place and to respond promptly and thereby minimizes the impact of the disease on animal and human health. Similarly, the provision of vaccines, trained staff, and suitable diagnostic tests is of paramount importance in infected countries.253, 260
Climate change and extreme weather events during the last decades may support the expansion of the geographical range of RVF northwards and into the Mediterranean countries.239 Forecasting models and livestock trade/movement data239, 324 may be useful in future to predict outbreaks of RVF outbreaks.239
References
- ABD EL-RAHIM, I. H., ABD EL-HAKIM, U. & HUSSEIN, M., 1999. An epizootic of Rift Valley fever in Egypt in 1997. Revue Scientifique et Technique, 18, 741-748.
- ABDALLAH, M. M. M., ADAM, I. A., ABDALLA, T. M., ABDELAZIZ, S. A., AHMED, M. E. & ARADAIB, I. E., 2016. A survey of rift valley fever and associated risk factors among the one-humped camel (Camelus dromedaries) in Sudan. Irish Veterinary Journal, 69, 6.
- ABDEL-AZIZ, A. A., MEGAN, J. M. & LAUGHLIN, L. W., 1980. Rift Valley fever as a possible cause of human abortions. Transactions of the Royal Society of Tropical Medicine and Hygiene, 74, 685-686.
- ABDEL-WAHAB, K. S. & EL BAZ, L. M., 1978. Rift Valley fever virus infections in Egypt: pathological and virological findings in man. Transactions of the Royal Society of Tropical Medicine and Hygiene,72, 392-396.
- ABDO-SALEM, S., WARET-SZKUTA, A., ROGER, F., OLIVE, M., SAEED, K. & CHEVALIER, V., 2011. Risk assessment of the introduction of Rift Valley fever from the Horn of Africa to Yemen via legal trade of small ruminants. Tropical Animal Health and Production, 43, 471-480.
- ABU-ELYAZEED, R., EL-SHARKAWY, S., OLSON, J., BOTROS, B., SOLIMAN, A., SALIB, A., CUMMINGS, C. & ARTHUR, R. R., 1996. Prevalence of anti-Rift-Valley-fever IgM antibody in abattoir workers in the Nile delta during the 1993 outbreak in Egypt. Bulletin of the World Health Organization, 74, 155-158.
- ACHEE, N. L., GRIECO, J. P., VATANDOOST, H., SEIXAS, G., PINTO, J., CHING-NG, L., MARTINS, A. J., JUNTARAJUMNONG, W., CORBEL, V., GOUAGNA, C., DAVID, J. P., LOGAN, J. G., ORSBORNE, J., MAROIS, E., DEVINE, G. J. & VONTAS, J., 2019. Alternative strategies for mosquito-borne arbovirus control. PLoS Neglected Tropical Diseases, 13, e0006822.
- ADAM, I. & KARSANY, M. S., 2008. Case report: Rift Valley Fever with vertical transmission in a pregnant Sudanese woman. Journal of Medical Virology, 80, 929.
- ADRIAMANDIMBY, S., RANDRIANARIVO-SOLOFONIAINA, A., JEANMAIRE, E., RAVOLOMANANA, L., RAZAFIMANANTSOA, L., RAKOTOJOELINANDRASANA, T., RAZAINIRINA, J., HOFFMAN, J., RAVALOHERY, J.-P., RAFISANDRATANTSOA, J.-T., ROLLIN, P. E. & REYNES, J.-M., 2010. Rift Valley fever during rainy seasons, Madagascar, 2008 and 2009. Emerging Infectious. Diseases, 16, 963-970.
- AFFSA., 2005. Rapport sur l’evaluation du risque d’apparition et de developpement des maladies animales compte tenu d’un eventuel rechauffement climatique, 8 février 2005 www.afssa.fr, 1-78.
- AITKEN, S. C., 2008. Variation in the S Segment of Rift Valley Fever Virus with Special Reference to the Nonstructural NSs Coding Region. University of Witwatersrand, 18-40 (MSc dissertation).
- AKAFEKWA, G. I., 1975. Sanitary position and methods of control in Zambia. Bulletin de l’Office International des Épizooties,83, 843-863.
- AL-HAZMI, A., AL-RAJHI, A. A., ABBOUD, E. B., AYOOLA, E. A., AL-HAZMI, M., SAADI, R. & AHMED, N., 2005. Ocular complications of Rift Valley fever outbreak in Saudi Arabia. Ophthalmology, 112, 313-318.
- AL-HAZMI, M., AYOOLA, E. A., ABDURAHMAN, M., BANZAL, S., ASHRAF, J., EL-BUSHRA, A., HAZMI, A., ABDULLAH, M., ABBO, H., ELAMIN, A., AL-SAMMANI, E. T., GADOUR, M., MENON, C., HAMZA, M., RAHIM, I., HAFEZ, M., JAMBAVALIKAR, M., ARISHI, H. & AQEEL, A., 2003. Epidemic Rift Vally fever in Saudi Arabia; a clinical study of severe illness in humans. Clinical Infectious Diseases, 36, 245-252.
- ALEXANDER, R. A., 1951. Rift Valley fever in the Union. Journal of the South African Veterinary Medical Association,22, 105-109.
- ALHAJ, M., 2016. Safety and Efficacy Profile of Commercial Veterinary Vaccines against Rift Valley Fever: A Review Study. Journal of Immunology Research, 7346294.
- ALLAM, I. H., FEINSOD, F. M., SCOTT, R. M., PETERS, C. J., SAAH, A. J., GHAFFAR, S. A., EL SAID, S. & DARWISH, M. A., 1986. Rift Valley fever surveillance in mobile sheep flocks in the Nile Delta.American Journal of Tropical Medicine and Hygiene, 35, 1055-1060.
- ANDERSON, D., 1967. Ecological studies on Sindbis and West Nile viruses in South Africa. III. Host preferences of mosquitoes as determined by the precipitin test. South African Journal of Medical Sciences, 32, 34-39.
- ANDERSON, G. W. & PETERS, C. J., 1988. Viral determinants of virulence for Rift Valley fever (RVF) in rats. Microbial Pathogenesis, 5, 241-250.
- ANDERSON, G. W. J., LEE, J. O., ANDERSON, A. O., POWELL, N., MANGIAFICO, J. A. & MEADORS, G., 1991. Efficacy of a Rift Valley fever virus vaccine against an aerosol infection in rats. Vaccine, 9, 710-714.
- ANDERSON, G. W. J., ROSEBROCK, J. A., JOHNSON, A. J., JENNINGS, G. B. & PETERS, C. J., 1991. Infection of inbred rat strains with Rift Valley fever virus: Development of a congenic resistant strain and observations on age-dependence of resistance. American Journal of Tropical Medicine and Hygiene,44, 475-480.
- ANDERSON, G. W. J., SALUZZO, J. F., KSIAZEK, T. G., SMITH, J. F., ENNIS, W., THUREEN, D., PETERS, C. J. & DIGOUTTE, J. P., 1989. Comparison of in vitroand in vivo systems of propagation of Rift Valley fever virus from clinical specimens. Research in Virology, 140, 129-138.
- ANDERSON, G. W. J., SLONE, T. W. J. & PETERS, C. J., 1987. Pathogenesis of Rift Valley fever virus (RVFV) in inbred rats. Microbial Pathogenesis,2, 283-293.
- ANDERSON, G. W. J. & SMITH, J. F., 1987. Immunoelectron microscopy of Rift Valley fever viral morphogenesis in primary rat hepatoytes. Virology,161, 91-100.
- ANONYMOUS., 1996. ARC Onderstepoort Veterinary Institute, Onderstepoort 0110, South Africa. Unpublished laboratory records.
- ANTONIS, A. F. G., KORTEKAAS, J., KANT, J., VLOET, R. P. M., VOGEL-BRINK, A., STOCKHOFE, N. & MOORMANN, R. J. M., 2013. Vertical transmission of Rift Valley fever virus without detectable maternal viremia. Vector-Borne and Zoonotic Diseases, 13, 601-606.
- ANYAMBA, A., -P., C. J., BRITCH, S. C., SOEBIYANTO, R. P., SMALL, J. L., JEPSEN, R., FORSHEY, B. M., SANCHEZ, J. L., SMITH, R. D., HARRIS, R., TUCKER, C. J., KARESH, W. B. & LINTHICUM, K. J., 2019. Global disease outbreaks associated with the 2015–2016 El Niño event. Scientific Reports, 9, 1930.
- ANYAMBA, A., LINTHICUM, K. J., SMALL, J., BRITCH, S. C., PAK, E., DE LA ROCQUE, S., FORMENTY, P., HIGHTOWER, A. W., BREIMAN, R. F., CHRETIEN, J. P., TUCKER, C. J., SCHNABEL, D., SANG, R., HAAGSMA, K., LATHAM, M., LEWANDOWSKI, H. B., MAGDI, S. O., MOHAMED, M. A., NGUKU, P. M., REYNES, J. M. & SWANEPOEL, R., 2010. Prediction, assessment of the Rift Valley fever activity in East and Southern Africa 2006-2008 and possible vector control strategies. American Journal of Tropical Medicine and Hygiene, 83, 43-51.
- ANYAMBA, A., LINTHICUM, K. J. & TUCKER, C. J., 2001. Climate-disease connections: Rift Valley fever in Kenya. Cadernos de Saúde Pública, 17, 133-140.
- ARCHER, B. N., WEYER, J., PAWESKA, J., NKOSI, D., LEMAN, P., TINT, K. S. & BLUMBERG, L., 2011. Outbreak of Rift Valley Fever affecting veterinarians and farmers in South Africa, 2008. South African Medical Journal, 101, 263-266.
- ARISHI, H. M., AQEEL, A. Y. & AL HAZMI, M. M., 2006. Vertical transmission of fatal Rift Valley fever in a newborn. Annals of Tropical Paediatrics, 26, 251-253.
- ARSEVSKA, E., HELLAL, J., MEJRI, S., HAMMAMI, S., MARIANNEAU, P., CALAVAS, D. & HÉNAUX, V., 2016. Identifying Areas Suitable for the Occurrence of Rift Valley Fever in North Africa: Implications for Surveillance. Transboundary and Emerging Diseases, 63, 658-674
- ARTHUR, R. R., EL SHARKAWY, M. S., COPE, S. E., BOTROS, B. A., OUN, S., MORRILL, J. C., SHOPE, R. E., HIBBS, R. G., DARWISH, M. A. & IMAM, I. Z., 1993. Recurrence of Rift Valley fever in Egypt. Lancet,342, 1149-1150.
- ASSAD, F., DAVIES, F. G. & EDDY, G. A., 1983. The use of veterinary vaccines for prevention and control of Rift Valley fever. Bulletin of the World Health Organization,61, 261-268.
- ATKINS, C. & FREIBERG, A. N., 2017. Recent advances in the development of antiviral therapeutics for Rift Valley fever virus infection. Future Virology, 12, 651-665.
- AUTHORITY., E. F. S., 2005. AHAW Panel Members, Opinion of the Scientific Panel on Animal Health and Welfare (AHAW). The risk of a Rift Valley fever incursion and its persistence within the Community. European Food Safety Authority Journal, 238, 1-128
- AUTHORITY., E. F. S., 2013. Panel on Animal Health and Welfare (AHAW); Scientific Opinion on Rift Valley Fever. European Food Safety Authority Journal, 11, 3180.
- AUTHORITY., E. F. S., 2020a. Rift Valley Fever–epidemiological update and risk of introduction into Europe. European Food Safety Authority Journal, 18, 6041.
- BABA, M., MASIGA, D. K., SANG, R. & VILLINGER, J., 2016. Has Rift Valley fever virus evolved with increasing severity in human populations in East Africa? Emerging Microbes and Infections, 5, e58
- BAILEY, C. L., 1981. U.S. Army Medical Research Institute for Infectious Diseases, Fort Detrick, Maryland. Personal communication.
- BALENGHIEN, T., CARDINALE, E., CHEVALIER, V., ELISSA, N., FAILLOUX, A. B., JEAN JOSE NIPOMICHENE, T. N., NICOLAS, G., RAKOTOHARINOME, V. M., ROGER, M. & ZUMBO, B., 2013. Towards a better understanding of Rift Valley fever epidemiology in the south-west of the Indian Ocean. Veterinary Research, 44, 78.
- BALKHY, H. H. & MEMISH, Z. A., 2003. Rift Valley Fever: An uninvited zoonosis in the Arabian Peninsula. International Journal of Antimicrobial Agents, 21,153-157.
- BARNARD, B. J. H., 1979. Rift Valley fever vaccine—antibody and immune response in cattle to a live and an inactivated vaccine. Journal of the South African Veterinary Association, 50, 155-157.
- BARNARD, B. J. H., 1981. Rift Valley fever in South Africa. Proceedings of the 49th General Session of the Office International des Epizooties, Paris, 25-30 May 1981.
- BARNARD, B. J. H. & BOTHA, M. J., 1977. An inactivated Rift Valley fever vaccine. Journal of the South African Veterinary Association, 48, 45-48.
- BARNARD, B. J. H. & VOGES, S. F., 1986. Flaviviruses in South Africa: Pathogenicity for sheep. Onderstepoort Journal of Veterinary Research, 53, 235–238.
- BASKERVILLE, A., HUBBARD, K. A. & STEPHENSON, J. R., 1992. Comparison of the pathogenicity for pregnant sheep of Rift Valley fever virus and a live attenuated vaccine. Research in Veterinary Science,52, 307-311.
- BATTLES, J. K. & DALRYMPLE, J. M., 1988. Genetic variation among geographic isolates of Rift Valley fever virus. American Journal of Tropical Medicine and Hygiene, 39, 617-631.
- BAUDIN, M., JUMAA, A. M., JOMMA, H. J. E., KARSANY, M. S., BUCHT, G., NÄSLUND, J., AHLM, C., EVANDER, M. & MOHAMED, N., 2016. Association of Rift Valley fever virus infection with miscarriage in Sudanese women: a cross-sectional study. Lancet Global Health, 4, e864-e871.
- BEATY, B. J., HOLTERMAN, M., TABACHNICK, W., SHOPE, R. E., ROZHON, E. J. & BISHOP, D. H., 1981. Molecular basis of bunyavirus transmission by mosquitoes: role of the middle-sized RNA segment. Science, 211, 1433-1435.
- BEATY, B. J., ROZHON, E. J., GENSEMER, P. & BISHOP, D. H., 1981. Formation of reassortant bunyaviruses in dually infected mosquitoes. Virology, 111, 662-665.
- BEECHLER, B. R., BENGIS, R., SWANEPOEL, R., PAWESKA, J. T., KEMP, A., VAN VUREN, P. J., JOUBERT, J., EZENWA, V. O. & JOLLES, A. E., 2015. Rift Valley Fever in Kruger National Park: Do buffalo play a role in Inter-epizootic circulation of virus. Transboundary and Emerging Disease, 62, 24-32.
- BELONJE, C. W. A., 1958. Field observations on Wesselsbron disease. Journal of the South African Veterinary Medical Association, 29, 1-12.
- BENNETT, D. G. & GERONE, P. J., 1962. Protection of mice from pantropic Rift Valley fever virus with immune calf serum. Bacteriological Proceedings, 62, 152.
- BERGREN, N. A., BORLAND, E. M., HARTMAN, D. A. & KADING, R. C., 2021. Laboratory demonstration of the vertical transmission of Rift Valley fever virus by Culex tarsalis mosquitoes. PLoS Neglected Tropical Diseases, 15(3), e0009273.
- BHARDWAJ, N., HEISE, M. T. & ROSS, T. M., 2010. Vaccination with DNA plasmids expressing Gn coupled to C3d or alphavirus replicons expressing gn protects mice against Rift Valley fever virus. PLoS Neglected Tropical Diseases, 4, e725.
- BILLECOCQ, A., SPIEGEL, M., VIALAT, P., KOHL, A., WEBER, F., BOULOY, M. & HALLER, O., 2004. NSs protein of Rift Valley fever virus blocks interferon production by inhibiting host gene transcription. Journal of Virology, 78, 9798-9806.
- BILLECOCQ, A., VIALAT, P. & BOULOY, M., 1996. Persistent infection of mammalian cells by Rift Valley fever virus. Journal of General Virology,77, 3053-3062.
- BIRD, B. H., ALBARIÑO, C. G., HARTMAN, A. L., ERICKSON, B. R., KSIAZEK, T. G. & NICHOL, S. T., 2008. Rift valley fever virus lacking the NSs and NSm genes is highly attenuated, confers protective immunity from virulent virus challenge, and allows for differential identification of infected and vaccinated animals. Journal of Virology, 82, 2681-2691.
- BIRD, B. H., BAWIEC, D. A., KSIAZEK, T. G., SHOEMAKER, T. R. & NICHOL, S. T., 2007. Highly sensitive and broadly reactive quantitative reverse transcription-PCR assay for high-throughput detection of Rift Valley fever virus. Journal of Clinical Microbiology, 45, 3506-3513.
- BIRD, B. H., GITHINJI, J. W., MACHARIA, J. M., KASIITI, J. L., MURIITHI, R. M., GACHERU, S. G., MUSAA, J. O., TOWNER, J. S., REEDER, S. A., OLIVER, J. B., STEVENS, T. L., ERICKSON, B. R., MORGAN, L. T., KHRISTOVA, M. L., HARTMAN, A. L., COMER, J. A., ROLLIN, P. E., KSIAZEK, T. G. & NICHOL, S. T., 2008. Multiple virus lineages sharing recent common ancestry were associated with a Large Rift Valley fever outbreak among livestock in Kenya during 2006-2007. Journal of Virology, 82,11152-11166.
- BIRD, B. H., KHRISTOVA, M. L., ROLLIN, P. E., KSIAZEK, T. G. & NICHOL, S. T., 2007. Complete genome analysis of 33 ecologically and biologically diverse Rift Valley fever virus strains reveals widespread virus movement and low genetic diversity due to recent common ancestry. Journal of Virology, 81, 2805-2816.
- BIRD, B. H., KSIAZEK, T. G., NICHOL, S. T. & MACLACHLAN, N. J., 2009. Rift Valley fever virus. Journal of the American Veterinary Medical Association, 234, 883-893.
- BIRD, B. H., MAARTENS, L. H., CAMPBELL, S., ERASMUS, B. J., ERICKSON, B. R., DODD, K. A., SPIROPOULOU, C. F., CANNON, D., DREW, C. P., KNUST, B., MCELROY, A. K., KHRISTOVA, M. L., ALBARIÑO, C. G. & NICHOL, S. T., 2011. Rift Valley fever virus vaccine lacking the NSs and NSm genes is safe, nonteratogenic, and confers protection from viremia, pyrexia, and abortion following challenge in adult and pregnant sheep. Journal of Virology, 85, 12901-12909.
- BIRD, B. H. & MAZET, J. A. K., 2018. Detection of Emerging Zoonotic Pathogens: An Integrated One Health Approach. Annual Review of Animal Biosciences, 6, 121-139.
- BIRD, B. H. & MCELROY, A. K., 2016. Rift Valley fever virus: Unanswered questions. Antiviral Research, 132, 274-280.
- BIRD, B. H. & NICHOL, S. T., 2012. Breaking the chain: Rift Valley fever virus control via livestock vaccination. Current Opinion in Virology, 2, 315-323.
- BITEYE, B., FALL, A. G., CISS, M., SECK, M. T., APOLLONI, A., FALL, M., TRAN, A. & GIMONNEAU, G., 2018. Ecological distribution and population dynamics of Rift Valley fever virus mosquito vectors (Diptera, Culicidae) in Senegal. Parasit Vectors, 11, 27.
- BLACKBURN, N. K. & BESSELAAR, T. G., 1991. A study of the effect of chemical inactivants on the epitopes of Rift Valley fever virus glycoproteins using monoclonal antibodies. Journal of Virological Methods,33, 367-374.
- BLACKBURN, N. K. & SWANEPOEL, R., 1980. An investigation of flavivirus infections of cattle in Zimbabwe Rhodesia with particular reference to Wesselsbron virus. Journal of Hygiene, 85, 1-33.
- BOB, N. S., BÂ, H., FALL, G., ISHAGH, E., DIALLO, M. Y., SOW, A., SEMBENE, P. M., FAYE, O., EL KOURI, B., SIDI, M. L. & SALL, A. A., 2017. Detection of the Northeastern African Rift Valley Fever Virus Lineage During the 2015 Outbreak in Mauritania. Open Forum Infectious Diseases, 4, ofx087.
- BOIRO I., K., K. O. & D., N. A., 1987. Isolation of Rift Valley Fever Virus from Bats in the Republic of Guinea. Bulletin de la Societe de pathologie exotique Filiales, 80, 62-67.
- BORUCKI, M. K., CHANDLER, L. J., PARKER, B. M., BLAIR, C. D. & BEATY, B. J., 1999. Bunyavirus superinfection and segment reassortment in transovarially infected mosquitoes. Journal of General Virology, 80(Pt 12), 3173-3179.
- BOSHRA, H., LORENZO, G., BUSQUETS, N. & BRUN, A., 2011. Rift valley fever: recent insights into pathogenesis and prevention. Journal of Virology, 85, 6098-6105.
- BOUGUEDOUR, R. & RIPANI, A., 2016. Review of the foot and mouth disease situation in North Africa and the risk of introducing the disease into Europe. Revue Scientifique et Technique, 35, 757-768.
- BOULOY, M. & WEBER, F., 2010. Molecular Biology of Rift Valley Fever Virus. Open Virology Journal, 4, 8-14.
- BOUSSINI, H., LAMIEN, C. E., NACOULMA, O. G., KABORÉ, A., PODA, G. & VILJOEN, G. J., 2014. Prevalence of Rift Valley fever in domestic ruminants in the central and northern regions of Burkina Faso. Revue Scientifique et Technique, 33, 893-901.
- BOWEN, M. D., TRAPPIER, S. G., SANCHEZ, A. J., MEYER, R. F., GOLDSMITH, C. S., ZAKI, S. R., DUNSTER, L. M., PETERS, C. J., KSIAZEK, T. G., NICHOL, S. T. & FORCE., R. T., 2001. A reassortant bunyavirus isolated from acute hemorrhagic fever cases in Kenya and Somalia. Virology, 291, 185-190.
- BRAACK, L. E. O., DE ALMEIDA, A. P. G., CORNEL, A. J., SWANEPOEL, R. & DE JAGER, C., 2018. Mosquito-borne arboviruses of African origin: review of key viruses and vectors. Parasite and Vectors, 11, 29. doi: 10.1186/s13071-017-2559-9.
- BREIMAN, R. F., NJENGA, M. K., CLEAVELAND, S., SHARIF, S. K., MBABU, M. & KING, L., 2008. Lessons from the 2006–2007 Rift Valley fever outbreak in East Africa: Implications for prevention of emerging infectious diseases. Future Virology, 3, 411-417.
- BRENNAN, B., WELCH, S. R., MCLEES, A. & ELLIOTT, R. M., 2011. Creation of a recombinant Rift Valley fever virus with a two-segmented genome. Journal of Virology, 85, 10310-10318.
- BRETT, N. A., THOMAS, J., WEYER, J., CENGIMBO, A., ESSOYA, L. D., JACOBS, C., NTULI, S., MODISE, M., MATHONSI, M., MASHISHI, M. S., LEMAN, P. A., LE ROUX, C., JANSEN VAN VUREN, P., KEMP, A., PAWESKA, J. T. & BLUMBERG, L., 2013. Epidemiological investigations into outbreaks of Rift Valley fever in humans, South Africa, 2008-2011. Emerging Infectious Diseases, 19(2), 1918-1925.
- BRETT, N. A., WEYER, J., PAWESKA, J., NKOSI, D., LEMAN, P., TINT, K. S. & BLUMBERG, L., 2011. Outbreak of Rift Valley fever virus affecting veterinarians and farmers in South Africa, 2008, South African Medical Journal, 101(4), 263-266.
- BRIESE, T., BIRD, B., KAPOOR, V., NICHOL, S. T. & LIPKIN, W. I., 2006. Batai and Ngari viruses: M segment reassortment and association with severe febrile disease outbreaks in East Africa. Journal of Virology, 80, 5627-5630.
- BRIESE, T., CALISHER, C. H. & HIGGS, S., 2013. Viruses of the family Bunyaviridae: are all available isolates reassortants? Virology, 446, 207-216.
- BRITCH, S. C., BINEPAL, Y. S., RUDER, M. G., KARIITHI, H. M., LINTHICUM, K. J., ANYAMBA, A., SMALL, J. L., TUCKER, C. J., ATEYA, L. O., ORIKO, A. A., GACHERU, S. & WILSON, W. C., 2013. Rift Valley fever risk map model and seroprevalence in selected wild ungulates and camels from Kenya. PLoS One, 8, e66626.
- BROWN, J. L., DOMINIK, J. W. & MORRISSEY, R. L., 1981. Respiratory infectivity of a recently isolated Egyptian strain of Rift Valley fever virus.Infection and Immunity, 33, 848-853.
- BRUBAKER, J. F. & TURELL, M. J., 1998. Effect of environmental temperature on the susceptibility of Culex pipiens (Diptera: Culicidae) to Rift Valley fever virus. Journal of Medical Entomology, 35, 918-921.
- BRUSTOLIN, M., TALAVERA, S., NUÑEZ, A., SANTAMARÍA, C., RIVAS, R., PUJOL, N., VALLE, M., VERDÚN, M., BRUN, A., PAGÈS, N. & BUSQUETS, N., 2017. Rift Valley fever virus and European mosquitoes: vector competence of Culex pipiens and Stegomyia albopicta (=Aedes albopictus). Medical and Veterinary Entomology, 31, 365-372.
- BUCCI, T. J., MOUSSA, I. M. & WOOD, O. L., 1981. Experimental Rift Valley fever encephalitis in ACI rats. Contributions to Epidemiology and Biostatistics,3: Rift Valley fever, 60-67.
- BUSCH, C. M., CALLICOTT, R. J., PETERS, C. J., MORRILL, J. C. & WOMACK, J. E., 2015. Mapping a Major Gene for Resistance to Rift Valley Fever Virus in Laboratory Rats. Journal of Heredity, 106, 728-733.
- CALVO-PINILLA, E., MARÍN-LÓPEZ, A., MORENO, S., LORENZO, G., UTRILLA-TRIGO, S., JIMÉNEZ-CABELLO, L., BENAVIDES, J., NOGALES, A., BLASCO, R., BRUN, A. & JAVIER ORTEGO, J., 2020. A protective bivalent vaccine against Rift Valley fever and bluetongue. Vaccines, 5, 70
- CAMPBELL, L. P. & ALEXANDER, A. M., 2017. Landscape Genetics of Aedes mcintoshi (Diptera: Culicidae), an Important Vector of Rift Valley Fever Virus in Northeastern Kenya. Journal of Medical Entomology, 54, 1258-1265.
- CAPLEN, H., PETERS, C. J. & BISHOP, D. H., 1985. Mutagen-directed attenuation of Rift Valley fever virus as a method for vaccine development. Journal of General Virology, 66 (Pt 10), 2271-2277.
- CAPOBIANCO DONDONA, A., ASCHENBORN, O., PINONI, C., DI GIALLEONARDO, L., MASEKE, A., BORTONE, G., POLCI, A., SCACCHIA, M., MOLINI, U. & MONACO, F., 2016. Rift Valley Fever Virus among Wild Ruminants, Etosha National Park, Namibia, 2011. Emerging Infectious Diseases, 22, 128-130.
- CAPSTICK, P. B. & GOSDEN, D., 1962. Neutralizing antibody response of sheep to pantropic and neurotropic Rift Valley fever virus. Nature,195, 583-584.
- CARROLL, S. A., REYNES, J. M., KHRISTOVA, M. L., ANDRIAMANDIMBY, S. F., ROLLIN, P. E. & NICHOL, S. T., 2011. Genetic evidence for Rift Valley fever outbreaks in Madagascar resulting from virus introductions from the East African mainland rather than enzootic maintenance. Journal of Virology, 85, 6162-6167.
- CASH, P., ROBESON, G., ERLICH, B. J. & BISHOP, D. H. L., 1981. Biochemical characterization of Rift Valley fever virus. Contributions to Microbiology and Immunology,3, 1-20.
- CECCATO, P., RAMIREZ, B., MANYANGADZE, T., GWAKISA, P. & THOMSON, M. C., 2018. Data and tools to integrate climate and environmental information into public health. Infectious Diseases of Poverty, 7, 126. https://doi.org/10.1186/s40249-018-0501-9.
- CEPI., 2017. New Vaccines For A Safer World. CEPI.
- CȆTYRE-SOSSAH, C., PE´DARRIEU, A., JUREMALM, M., JANSEN VAN VUREN, P., BRUN, A., OULD, E. L., MAMY, A. B., HÉRAUD, J. M., FILIPPONE, C., RAVALOHERY, J. P., CHAABIHI, H., ALBINA, E., DOMMERGUES, L., PAWESKA, J. & CARDINALE, E., 2019. Development and validation of a pen side test for Rift Valley fever. PLoS Neglected Tropical Diseases, 13(9), e0007700.
- CHAMBERS, P. G. & SWANEPOEL, R., 1980. Rift Valley fever in abattoir workers. Central African Journal of Medicine,26, 122-126.
- CHANDLER, L. J., BEATY, B. J., BALDRIDGE, G. D., BISHOP, D. H. & HEWLETT, M. J., 1990. Heterologous reassortment of bunyaviruses in Aedes triseriatus mosquitoes and transovarial and oral transmission of newly evolved genotypes. Journal of General Virology, 71(Pt 5), 1045-1050.
- CHANDLER, L. J., HOGGE, G., ENDRES, M., JACOBY, D. R., NATHANSON, N. & BEATY, B. J., 1991. Reassortment of La Crosse and Tahyna bunyaviruses in Aedes triseriatus mosquitoes. Virus Research, 20, 181-191.
- CHARE, E. R., GOULD, E. A. & HOLMES, E. C., 2003. Phylogenetic analysis reveals a low rate of homologous recombination in negative-sense RNA viruses. Journal of General Virology, 84, 2691-2703.
- CHEVALIER, V., LANCELOT, R., THIONGANE, Y., SALL, B., DIAITÉ, A. & MONDET, B., 2005. Rift Valley Fever in small ruminants, Senegal, 2003. Emerging Infectious Diseases, 11, 1693-1700.
- CHEVALIER, V., PEPIN, M., PLEE, L. & LANCELOT, R., 2010. Rift Valley Fever—A threat for Europe? Euro Surveillance, 15, 19506.
- CHEVALIER, V., THIONGANE, Y. & LANCELOT, R., 2009. Endemic transmission of Rift Valley Fever in Senegal. Transboundary Emerging Diseases, 56, 372-374.
- CHRISTIE, G. J., 1969. Rift Valley fever. Rhodesia Science News, 3, 238-240.
- CLARKE, D. H. & CASALS, J., 1958. Techniques for hemagglutination and hemagglutination-inhibition with arthropod-borne viruses. American Journal of Tropical Medicine and Hygiene, 7, 561-573.
- COACKLEY, W., PINI, A. & GOSDEN, D., 1967. Experimental infection of cattle with pantropic Rift Valley fever virus. Research in Veterinary Science, 8, 399-405.
- COACKLEY, W., PINI, A. & GOSDEN, D., 1967. The immunity induced in cattle and sheep in inoculation of neurotropic or pantropic Rift Valley fever viruses. Research in Veterinary Science,8, 406-414.
- COETZER, J. A. W., 1977. The pathology of Rift Valley fever. I. Lesions occurring in natural cases in new-born lambs. Onderstepoort Journal of Veterinary Research, 44, 205-211.
- COETZER, J. A. W., 1977. The pathology of Rift Valley fever. I. Lesions occurring in natural cases in new-born lambs. Onderstepoort Journal of Veterinary Research,44, 205-212.
- COETZER, J. A. W. & BARNARD, B. J. H., 1977. Hydrops amnii in sheep associated with hydranencephaly and arthrogryposis with Wesselsbron disease and Rift Valley fever viruses as aetiological agents. Onderstepoort Journal of Veterinary Research, 44, 119-126.
- COETZER, J. A. W. & IMES, G. D., 1975. Department of Veterinary Tropical Diseases, Faculty of Veterinary Science, University of Pretoria, Onderstepoort 0110. Unpublished observations.
- COETZER, J. A. W. & ISHAK, K. G., 1982. Sequential development of the liver lesions in new-born lambs infected with Rift Valley fever virus. I. Macroscopic and microscopic pathology. Onderstepoort Journal of Veterinary Research,49, 103-108.
- COETZER, J. A. W., ISHAK, K. G. & CALVERT, R. C., 1982. The pathology of Rift Valley fever. II. Lesions occurring in field cases in adult cattle, calves and aborted fetuses. Onderstepoort Journal of Veterinary Research, 49, 11-17.
- COETZER, J. A. W., ISHAK, K. G. & CALVERT, R. C., 1982. Sequential development of the liver lesions in new-born lambs infected with Rift Valley fever virus. II. Ultrastructural findings. Onderstepoort Journal of Veterinary Research,49, 109-122.
- COETZER, J. A. W. & THEODORIDIS, A., 1982. Clinical and pathological studies in adult sheep and goats experimentally infected with Wesselsbron disease virus. Onderstepoort Journal of Veterinary Research, 49, 19-22.
- COETZER, J. A. W., THEODORIDIS, A. & VAN HEERDEN, A., 1978. Wesselsbron disease. Pathological haematological and clinical studies in natural cases and experimentally infected new-born lambs. Onderstepoort Journal of Veterinary Research, 45, 93-106.
- COHEN, C. & LUNTZ, M. H., 1976. Rift-Valley-Fieber und Rickettsianretinitis einschliesslich fluoresceinangiographie. Klinische Monatsblatter fur Augenheilkunde,169, 685-699.
- CORWIN, A., HABIB, M., WATTS, D., DARWISH, M., OLSON, J., BOTROS, B., HIBBS, R., KLEINOSKY, M., LEE, H. W., SHOPE, R. & KILPATRICK, M., 1993. Community-based prevalence profile of arboviral, rickettsial, and Hantaan-like viral antibody in the Nile River Delta of Egypt. American Journal of Tropical Medicine and Hygiene,48, 776-783.
- CORWIN, A., HABIB, M., WATTS, D., OLSON, J., DARWISH, M., HIBBS, R., BOTROS, B., KLEINOSKY, M., SHOPE, R. & KILPATRICK, M., 1993. Prevalence of antibody to Rift Valley fever virus in the Nile River Delta of Egypt, 13 years after a major outbreak. Transactions of the Royal Society of Tropical Medicine and Hygiene,87, 161.
- COSGRIFF, T. M., MORRILL, J. C., JENNINGS, G. B., HODGSON, L. A., SLAYTER, M. V., GIBBS, P. H. & PETERS, C. J., 1989. Hemostatic derangement produced by Rift Valley fever virus in rhesus monkeys. Reviews of Infectious Diseases,11, 807-814.
- CRABTREE, M. B., KENT CROCKETT, R. J., BIRD, B. H., NICHOL, S. T., ERICKSON, B. R., BIGGERSTAFF, B. J., HORIUCHI, K. & MILLER, B. R., 2012. Infection and transmission of Rift Valley fever viruses lacking the NSs and/or NSm genes in mosquitoes: potential role for NSm in mosquito infection. PLoS Neglected Tropical Diseases, 6, e1639.
- DANZETTA, M. L., BRUNO, R., SAURO, F., SAVINI, L. & CALISTRI, P., 2016. Rift Valley fever transmission dynamics described by compartmental models. Preventive Veterinary Medicine, 134, 197-210.
- DAOUAM, S., FAKRI, F. Z., ENNAJI, M. M., EL ARKAM, A., TADLAOUI, K. O., OURA, C. & EL HARRAK, M., 2014. Heat stability of the Rift Valley fever Clone 13 live vaccines. Trials in Vaccinology, 3, 61-64.
- DAOUAM, S., GHZAL, F., ARKAM, A. E., Y., N., JAZOULI, M., ENNAJI, M. M., TADLAOUI, K. O., OURA, C. & ELHARRAK, M., 2015. Evaluation of the safety and efficacy of a live attenuated thermostable Rift Valley fever vaccine in sheep, goats and cattle. Journal of Vaccines and Vaccination, 6, 295.
- DAOUAM, S., GHZAL, F., NAOULI, Y., TADLAOUI, K. O., ENNAJI, M. M., OURA, C. & EL HARRAK, M., 2016. Safety and immunogenecity of a live attenuated Rift Valley fever vaccine (CL13T) in camels. BMC Veterinary Research, 12, 154.
- DARWISH, M. A. & HOOGSTRAAL, H., 1981. Arboviruses infecting humans and lower animals in Egypt: reviews of thirty years of research. Journal of the Egyptian Public Health Association,56, 1-112.
- DARWISH, M. A., IMAM, I. Z. E. & OMAR, F., 1978. A seroepidemiological study for Rift Valley fever virus in humans and domestic animals of Egypt. Journal of the Egyptian Public Health Association, 53, 153-162.
- DAUBNEY, R. & HUDSON, J. R., 1932. Rift Valley fever. Lancet, 1, 611-612.
- DAUBNEY, R. & HUDSON, J. R., 1933. Rift Valley fever. East African Medical Journal,10, 2-19.
- DAUBNEY, R., HUDSON, J. R. & GARNHAM, P. C., 1931. Enzootic hepatitis or Rift Valley fever. An undescribed virus disease of sheep, cattle and man from East Africa. Journal of Pathology and Bacteriology, 34, 545-579.
- DAVIES, F. G., 1975. Observations on the epidemiology of Rift Valley fever in Kenya.Journal of Hygiene, 75, 219-230.
- DAVIES, F. G., 1981. Rift Valley fever in Kenya. Proceedings of the 49th General Session of the Office International des Epizooties, Paris, 25-30 May.
- DAVIES, F. G. & ADDY, P. A. K., 1979. Rift Valley fever. A survey for antibody to the virus in bird species commonly found in situations considered to be enzootic. Transactions of the Royal Society of Tropical Medicine and Hygiene,73, 584-585.
- DAVIES, F. G., CLAUSEN, B. & LUND, L. J., 1972. The pathogenicity of Rift Valley fever virus for the baboon. Transactions of the Royal Society of Tropical Medicine and Hygiene,66, 363-365.
- DAVIES, F. G. & HIGHTON, R. B., 1980. Possible vectors of Rift Valley fever in Kenya. Transactions of The Royal Society of Tropical Medicine and Hygiene, 74, 815-816.
- DAVIES, F. G. & KARSTAD, L., 1981. Experimental infection of the African buffalo with the virus of Rift Valley fever. Tropical Animal Health and Production, 13, 185-188.
- DAVIES, F. G., KILELU, E., LINTHICUM, K. J. & PEGRAM, R. G., 1992. Patterns of Rift Valley fever activity in Zambia.Epidemiology and Infection, 108, 185-191.
- DAVIES, F. G., KOROS, J. & MBUGUA, H., 1985. Rift Valley fever in Kenya: the presence of antibody to the virus in camels (Camelus dromedarius). Journal of Hygiene (London), 94, 241-244.
- DAVIES, F. G. & LINTHICUM, K. J., 1986. The Sudan dioch (Quelea quelea aethiopica) and Rift Valley fever. Transactions of the Royal Society of Tropical Medicine and Hygiene,80, 171-171.
- DAVIES, F. G., LINTHICUM, K. J. & JAMES, A. D., 1985. Rainfall and epizootic Rift Valley fever. Bulletin of the World Health Organization,63, 941-943.
- DAVIES, F. G., LOGAN, T. M., BINEPAL, Y. & JESSEN, P., 1992. Rift Valley fever virus activity in East Africa in 1989.Veterinary Record, 130, 247-249.
- DAVIES, F. G. & ONYANGO, E., 1978. Rift Valley fever: The role of the vervet monkey as a reservoir or maintenance host for this virus. Transactions of the Royal Society of Tropical Medicine and Hygiene,72, 213-214.
- DAVIES, F. G., SOI, R. K. & WARIRU, B. N., 1984. Abortion in sheep caused by Thogoto virus. Veterinary Record,115, 654-654.
- DAVIS, D. H. S., 1957. The small wild mammals in relation to the virus studies. South African Journal of Medical Sciences, 22, 55-61.
- DAVOUST, B., MARIE, J. L. & BONI, M., 2008. Prevention of zoonoses: creation of a unit for early detection of animal infections, Bull. Bulletin de l'Académie Nationale de Médecine, 192, 541-554 (in French).
- DE LA FUENTE, C., PINKHAM, C., DABBAGH, D., BEITZEL, B., GARRISON, A., PALACIOS, G., HODGE, K. A., PETRICOIN, E. F., SCHMALJOHN, C., CAMPBELL, C. E., NARAYANAN, A. & KEHN-HALL, K., 2018. Phosphoproteomic analysis reveals Smad protein family activation following Rift Valley fever virus infection. PLoS One, 13, e0191983.
- DE MEILLON, B., PATERSON, H. E. & MUSPRATT, J., 1957. Studies on the arthropod-borne viruses of Tongaland. II. Notes on the more common mosquitoes. South African Journal of Medical Sciences, 22, 47-53.
- DEUTMAN, A. F. & KLOMP, H. J., 1981. Rift Valley fever retinitis. American Journal of Ophthalmology, 92, 38-42.
- DI NARDO, A., ROSSI, D., SALEH, S. M. L., LEJLIFA, S. M., HAMDI, S. J., DI GENNARO, A., SAVINI, G. & THRUSFIELD, M. V., 2014. Evidence of Rift Valley fever seroprevalence in the Sahrawi semi-nomadic pastoralist system, Western Sahara. BMC Veterinary Research, 10, 92.
- DIALLO, M., LOCHOUARN, L., BA, K., SALL, A. A., MONDO, M., GIRAULT, L. & MATHIOT, C., 2000. First isolation of the Rift Valley fever virus from Culex poicilipes (Diptera: Culicidae) in nature. American Journal of Tropical Medicine and Hygiene,62, 702-704.
- DICKSON, J. L., 1951. Rift Valley fever in the Union. Journal of the South African Veterinary Medical Association,22, 110-111.
- DIGOUTTE, J. P., JOUAN, A., LE GUENNO, B., RIOU, O., PHILIPPE, B., MEEGAN, J., KSIAZEK, T. G. & PETERS, C. J., 1989. Isolation of the Rift Valley fever virus by inoculation into Aedes pseudoscutellariscells: Comparison with other diagnostic methods. Research in Virology, 140, 31-41.
- DODD, K. A., BIRD, B. H., METCALFE, M. G., NICHOL, S. T. & ALBARIÑO, C. G., 2012. Single-dose immunization with virus replicon particles confers rapid robust protection against Rift Valley fever virus challenge. Journal of Virology, 86, 4204-4212.
- DODSON, B. L., ANDREWS, E. S., TURELL, M. J. & RASGON, J. L., 2017. Wolbachia effects on Rift Valley fever virus infection in Culex tarsalis mosquitoes. PLoS Neglected Tropical Diseases, 11, e0006050.
- DOHM, D. J., ROMOSER, W. S., TURELL, M. J. & LINTHICUM, K. J., 1991. Impact of stressful conditions on the survival of Culex pipiens exposed to Rift Valley fever virus. Journal of the American Mosquito Control Association, 7, 621-623.
- DOHM, D. J., ROWTON, E. D., LAWYER, P. G., O’GUINN, M. & TURELL, M. J., 2000. Laboratory transmission of Rift Valley fever virus by Phlebotomus duboscqi, Phlebotomus papatasi, Phlebotomus sergenti, and Sergentomyia schwetzi (Diptera: Psychodidae). Journal of Medical Entomology, 37, 435-438.
- DROSTEN, C., GÖTTIG, S., SCHILLING, S., ASPER, M., PANNING, M., SCHMITZ, H. & GÜNTHER, S., 2002. Rapid detection and quantification of RNA of Ebola and Marburg viruses, Lassa virus, Crimean-Congo hemorrhagic fever virus, Rift Valley fever virus, dengue virus, and yellow fever virus by real-time reverse transcription-PCR. Journal of Clinical Microbiology, 40, 2323-2330.
- DUFOUR, B., MOUTOU, F., HATTENBERGER, A. M. & RODHAIN, F., 2008. Global change: impact, management, risk approach and health measures – the case of Europe. Revue scientifique et technique (International Office of Epizootics), 27, 529-550.
- DUNGU, B., LOUW, I., LUBISI, A., HUNTER, P., VON TEICHMAN, B. F. & BOULOY, M., 2010. Evaluation of the efficacy and safety of the Rift Valley Fever Clone 13 vaccine in sheep. Vaccine, 28, 4581-4587.
- DUNGU, B., LUBISI, B. A. & IKEGAMI, T., 2018. Rift Valley fever vaccines: current and future needs. Current Opinion in Virology, 29, 8-15.
- EARLEY, E., PERALTA, P. H. & JOHNSON, K. M., 1967. A plaque reduction method for arboviruses. Proceedings of the Society for Experimental Biology and Medicine, 125, 741-747.
- EASTERDAY, B. C., 1965. Rift Valley fever. Advances in Veterinary Science,10, 65-127.
- EASTERDAY, B. C., MCGAVRAN, M. H., ROONEY, J. R. & MURPHY, L. C., 1962. The pathogenesis of Rift Valley fever in lambs. American Journal of Veterinary Research,23, 470-478.
- EASTERDAY, B. C. & MURPHY, L. C., 1963. Studies on Rift Valley fever in laboratory animals. Cornell Veterinarian,53, 423-433.
- EASTERDAY, B. C., MURPHY, L. C. & BENNETT, D. G., 1962. Experimental Rift Valley fever in calves, goats and pigs. American Journal of Veterinary Research,23, 1224-1230.
- EASTERDAY, B. C., MURPHY, L. C. & BENNETT, D. G., 1962. Experimental Rift Valley fever in lambs and sheep. American Journal of Veterinary Research,23, 1231-1240.
- EDDY, G. A., PETERS, C. J., MEADORS, G. & COLE, F. E. J., 1981. Rift Valley fever vaccine for humans. Contributions to Epidemiology and Biostatistics,3: Rift Valley fever, 124-141.
- EDWARDS, F. W., 1941. Mosquitoes of the Ethiopian region. III Culicine adults and pupae. British Museum (Natural History), London, 499
- EISA, M., 1981. A note on Rift Valley fever in the Sudan. Proceedings of the 49th General Session of the Office International des Epizooties, Paris, 25-30 May.
- EISA, M., 1984. Preliminary survey of domestic animals of the Sudan for precipitating antibodies to Rift Valley fever virus. Journal of Hygiene (London), 93,629-637.
- EISA, M. & OBEID, H. M. A., 1977. Rift Valley fever in the Sudan. II. Isolation and identification of the virus from a recent epizootic in Kosti District, 1973. Bulletin of Animal Health and Production in Africa,24, 349-355.
- EISA, M., OBEID, H. M. S. & EL SAEI, A. S. A., 1977. Rift Valley fever in the Sudan. I. Results of field investigations of the first epizootic in Kosti District, 1973. Bulletin of Animal Health and Production in Africa, 24, 343-347.
- EL-DIN ABDEL-WAHAB, K. S. & EL BAZ, L. M., 1978. Rift Valley fever virus infections in Egypt: pathological and virological findings in man. Transactions of the Royal Society of Tropical Medicine and Hygiene,72, 392-396.
- EL-HARRAK, M., MARTÍN-FOLGAR, R., LLORENTE, F., FERNÁNDEZ-PACHECO, P., BRUN, A., FIGUEROLA, J. & JIMÉNEZ-CLAVERO, M., 2011. Rift Valley and West Nile Virus Antibodies in Camels, North Africa. Emerging Infectious Diseases, 17, 2372-2374.
- EL-KARAMANY, R., IMAN, I. Z. E., FARID, A. H., SABER, M. S., MOUSTAFA, A. F., ZAKI, M. & BASHANDY, A., 1981. Production of inactivated RVF vaccine. Journal of the Egyptian Public Health Association,56, 495-525.
- EL IMAM, M., EL SABIQ, M., OMRAN, M., ABDALKAREEM, A., EL GAILI MOHAMED, M. A., ELBASHIR, A. & KHALAFALA, O., 2009. Acute renal failure associated with the Rift Valley fever: a single center study. Saudi Journal of Kidney Diseases and Transplantation, 20(6), 1047-1052.
- EL MAMY, A. B., BABA, M. O., BARRY, Y., ISSELMOU, K., DIA, M. L., EL KORY, M. O., DIOP, M., LO, M. M., THIONGANE, Y., BENGOUMI, M., PUECH, L., PLEE, L., CLAES, F., DE LA ROCQUE, S. & DOUMBIA, B., 2011. Unexpected Rift Valley fever outbreak, northern Mauritania. Emerging Infectious Diseases, 17, 1894-1896.
- EL MAMY, A. B., LO, M. M., THIONGANE, Y., DIOP, M., ISSELMOU, K., DOUMBIA, D., BABA, M. O., EL ARBI, A. S., LANCELOT, R., KANE, Y., ALBINA, E. & CÊTRE-SOSSAH, C., 2014. Comprehensive phylogenetic reconstructions of Rift Valley fever virus: the 2010 northern Mauritania outbreak in the Camelus Dromedarius species. Vector-Borne Diseases, 14, 856-861.
- ELLIOT, R. M., 1990. Molecular biology of the bunyaviridae. Journal of General Virology, 71, 501-522.
- ELLIS, D. S., SHIRODARIA, P. V., FLEMMING, E. & SIMPSON, D. I. H., 1988. Morphology and development of Rift Valley fever virus in Vero cell cultures. Journal of Medical Virology,24, 161-174.
- ELLIS, D. S., SIMPSON, D. I. H., STAMFORD, S. & ABDEL WAHAB, K. S. E., 1979. Rift Valley fever: Some ultrastructural observations on material from the outbreaks in Egypt.Journal of General Virology, 42, 329-337.
- ERASMUS, B. J., 1981. Onderstepoort Veterinary Institute, Onderstepoort 0110, South Africa. Personal communication.
- ERASMUS, B. J. & COETZER, J. A. W., 1981. Symptomatology and pathology of Rift Valley fever in domestic animals. Contributions to Epidemiology and Biostatistics,3: Rift Valley fever, 77-82.
- EVANS, A., GAKUYA, F., PAWESKA, J. T., ROSTAL, M., AKOOLO, L., VAN VUREN, P. J., MANYIBE, T., MACHARIA, J. M., KSIAZEK, T. G., FEIKIN, D. R., BREIMAN, R. F. & KARIUKI NJENGA, M., 2008. Prevalence of antibodies against Rift Valley fever virus in Kenyan wildlife. Epidemiology and Infection, 136, 1261-1269.
- FABURAY, B., GAUDREAULT, N. N., LIU, Q., DAVIS, A. S., SHIVANNA, V., SUNWOO, S. Y., LANG, Y., MOROZOV, I., RUDER, M., DROLET, B., SCOTT MCVEY, D., MA, W., WILSON, W. & RICHT, J. A., 2016a. Development of a sheep challenge model for Rift Valley fever. Virology, 489, 128-140.
- FABURAY, B., LABEAUD, A. D., MCVEY, D. S., WILSON, W. C. & RICHT, J. A., 2017. Current Status of Rift Valley Fever Vaccine Development. Vaccines, 5(3), 29.
- FABURAY, B., WILSON, W. C., GAUDREAULT, N. N., DAVIS, A. S., SHIVANNA, V., BAWA, B., SUNWOO, S. Y., MA, W., DROLET, B. S., MOROZOV, I., MCVEY, D. S. & RICHT, J. A., 2016b. A Recombinant Rift Valley Fever Virus Glycoprotein Subunit Vaccine Confers Full Protection against Rift Valley Fever Challenge in Sheep. Science Reports, 6, srep27719.
- FAFETINE, J., TIJHAAR, E., PAWESKA, J. T., NEVES, L. C. B. G., HENDRIKS, J., SWANEPOEL, R., COETZER, J. A. W., EGBERING, H. F. & RUTTEN, V. P. M. G., 2007. Cloning and expression of Rift Valley fever virus nucleocapsid (N) protein and evaluation of a N-protein based indirect ELISA for the detection of specific IgG and IgM antibodies in domestic ruminants. Veterinary Microbiology, 121, 29-38.
- FAGBAMI, A. H., TOMORI, O. & FABIYI, A., 1975. Experimental Rift Valley fever in West African dwarf sheep. Research in Veterinary Science, 18, 334-335.
- FAGBAMI, A. J., TOMORI, O. & KEMP, G. E., 1973. A survey of Nigerian domestic and wild animals for serum neutralizing antibody to indigenous Rift Valley fever virus. Nigerian Veterinary Journal,2, 45-48.
- FAKOUR, S., NASERABADI, S. & AHMADI, E., 2017. The first positive serological study on rift valley fever in ruminants of Iran. Journal of Vector Borne Diseases, 54, 348-352.
- FAO., 2002. GEERING, W. A. & DAVIES, F. G. with additions by MARTIN, V. Preparation of Rift Valley Fever Contingency Plans. FAO Animal Health Manual No. 15. Published by Food and Agriculture Organization of the United Nations, Rome 2002. ISBN: 92-5-104821-5.
- FAO., 2011. Rift Valley fever vaccine development, progress and constrains. Proceedings of the GF-TADs meeting, January 2011. Rome, Italy. FAO Animal Production and Health Proceedings, No.12. Rome Italy.
- FAO., 2018. MARINER, J. Rift Valley Fever Surveillance. FAO Animal Production and Health. Manual No. 21.
- FARAN, M. E., ROMOSER, W. S., ROUTIER, R. G. & BAILEY, C. J., 1986. Use of the avidin-biotin-peroxidase complex immunocytochemical procedure for detection of Rift Valley fever virus in paraffin sections of mosquitoes. American Journal of Tropical Medicine and Hygiene,35, 1061-1067.
- FARAN, M. E., ROMOSER, W. S., ROUTIER, R. G. & BAILEY, C. L., 1988. The distribution of Rift Valley fever virus in the mosquito Culex pipiensas revealed by viral titration of dissected organs and tissues. American Journal of Tropical Medicine and Hygiene, 39, 206-213.
- FARAN, M. E., TURELL, M. J., ROMOSER, W. S., ROUTIER, R. G., GIBBS, P. H., CANNON, T. L. & BAILEY, C. L., 1987. Reduced survival of adult Culex pipiensinfected with Rift Valley fever virus. American Journal of Tropical Medicine and Hygiene, 37, 403-409.
- FAWZY, M. & HELMY, Y. A., 2019. The One Health Approach is necessary for the control of rift valley fever infections in Egypt: A comprehensive Review. Viruses, 11, 139.
- FILONE, C. M., HEISE, M., DOMS, R. W. & BERTOLOTTI-CIARLET, A., 2006. Development and characterization of a Rift Valley fever virus cell-cell fusion assay using alphavirus replicon vectors. Virology, 356, 155-164.
- FINDLAY, G. M., 1932. The infectivity of Rift Valley fever for monkeys. Transactions of the Royal Society of Tropical Medicine and Hygiene,26, 161-168.
- FINDLAY, G. M., 1932. Rift Valley fever or enzootic hepatitis. Transactions of the Royal Society of Tropical Medicine and Hygiene,25, 229-265.
- FINDLAY, G. M., 1933. Cytological changes in the liver of Rift Valley fever, with special reference to the nuclear inclusions. British Journal for Experimental Pathology, 14, 207-219.
- FINDLAY, G. M., MACKENZIE, R. D. & STERN, R. O., 1936. Studies on neurotropic Rift Valley fever virus: The susceptibility of sheep and monkeys. British Journal for Experimental Pathology,17, 431-441.
- FLICK, R. & BOULOY, M., 2005. Rift Valley fever virus. Current Molecular Medicine, 5, 827-834.
- FONTANA, J., LOPEZ-MONTERO, N., ELLIOTT, R. M., FERNANDEZ, J. J. & RISCO, C., 2008. The unique architecture of Bunyamwera virus factories around the Golgi complex. Cell Microbiology, 10, 2012-2028.
- FONTENILLE, D., 1989. Transmission cycles of arboviruses in Madagascar. Archives de l'Institut Pasteur de Madagascar, 55, 7-317.
- FONTENILLE, D., TRAORE-LAMIZANA, M., DIALLO, M., THONNON, J., DIGOUTTE, J. P. & ZELLER, H. G., 1998. New vectors of Rift Valley fever in West Africa. Emerging Infectious Diseases, 4, 289-293.
- FONTENILLE, D., TRAORE-LAMIZANA, M., ZELLER, H., MONDO, M., DIALLO, M. & DIGOUTTE, J. P., 1995. Short report: Rift Valley fever in Western Africa: Isolations from Aedes mosquitoes during an inter epizootic period. American Journal of Tropical Medicine and Hygiene, 52, 403-404.
- FORMENTY, P., DOMENECH, J. & ZELLER, H. G., 1992. [Serological survey of Rift Valley fever in sheep on the Ivory Coast]. Revue d’Élevage et de Médécine Veterinaire des Pays Tropicaux, 45, 221-226.
- FRANCIS, T. J. & MAGILL, T. P., 1935. Rift Valley fever. A report of three cases of laboratory infection and the experimental transmission of the disease to ferrets. Journal of Experimental Medicine,62, 433-448.
- FRANK-PETERSIDE, N., 2000. Response of laboratory staff to vaccination with an inactivated Rift Valley fever vaccine—TSI-GSD 200. African Journal of Medicine and Medical Sciences,29, 89-92.
- FREED, I., 1951. Rift Valley fever in man complicated by retinal changes and loss of vision. South African Medical Journal, 25, 930-932.
- FREIRE, C. C. M., IAMARINO, A., SOUMARÉ, P. O. L., FAYE, O., SALL, A. A. & ZANOTTO, P. M. A., 2015. Reassortment and distinct evolutionary dynamics of Rift Valley Fever virus genomic segments. Scientific Reports, 5.
- FRESE, M., KOCHS, G., FELDMANN, H., HERTKORN, C. & HALLER, O., 1996. Inhibition of bunyaviruses, phleboviruses, and hantaviruses by human MxA protein. Journal of Virology,70, 915-923.
- GACHOHI, J. M., NJENGA, M. K., KITALA, P. & BETT, B., 2016. Modelling Vaccination Strategies against Rift Valley Fever in Livestock in Kenya. PLoS Neglected Tropical Diseases, 10(12), e0005049.
- GAD, A. M., FARID, H. A., RAMZY, R. R., RIAD, M. B., PRESLEY, S. M., COPE, S. E., HASSAN, M. M. & HASSAN, A. N., 1999. Host feeding of mosquitoes (Diptera: Culicidae) associated with the recurrence of Rift Valley fever in Egypt. Journal of Medical Entomology,36, 709-714.
- GAD, A. M., FEINSOD, F. M., ALLAN, I. H., EISA, M., HASSAN, A. N. & SOLIMAN, B. A., 1986. A possible route for the introduction of Rift Valley fever virus into Egypt during 1977. Journal of Tropical Medicine and Hygiene,89, 233-236.
- GAD, A. M., HASSAN, M. M., EL SAID, S., MOUSSA, M. I. & WOOD, O. L., 1987. Rift Valley fever virus transmission by different Egyptian mosquito species. Transactions of the Royal Society of Tropical Medicine and Hygiene,81, 694-698.
- GAD, A. M., RIAD, I. B. & FARID, H. A., 1995. Host-feeding patterns of Culex pipiens andCx. antennatus (Diptera: Culicidae) from a village in Sharqiya Governorate, Egypt. Journal of Medical Entomology, 32, 573-577.
- GALE, P., BROUWER, A., RAMNIAL, V., KELLY, L., KOSMIDER, R., FOOKS, A. R. & SNARY, E. L., 2010. Assessing the impact of climate change on vector-borne viruses in the EU through the elicitation of expert opinion. Epidemiology and Infection, 138, 214-225.
- GARCIA, S., CRANCE, J. M., BILLECOCQ, A., PEINNEQUIN, A., JOUAN, A., BOULOY, M. & GARIN, D., 2001. Quantitative real-time PCR detection of Rift Valley fever virus and its application to evaluation of antiviral compounds. Journal of Clinical Microbiology, 39, 4456-4461.
- GARGAN, T. P., BAILEY, C. L., HIGBEE, G. A., GAD, A. & EL SAID, S., 1983. The effect of laboratory colonization on the vector-pathogen interactions of Egyptian Culex Pipiens and Rift Valley fever virus. American Journal of Tropical Medicine and Hygiene, 3, 1154-1163.
- GARGAN, T. P., JUPP, P. G. & NOVAK, R. J., 1988. Rift Valley Fever outbreak in livestock, Mozambique, 2014. Emerging Infectious Diseases, 22, 2165-2167.
- GARGAN, T. P. I., JUPP, P. G. & NOVAK, R. J., 1988. Panveld oviposition sites of floodwater Aedesmosquitoes and attempts to detect transovarial transmission of Rift Valley fever virus in South Africa.Medical and Veterinary Entomology, 2, 231-236.
- GARRET-JONES, C., 1964. Prognosis for the interruption of malaria transmission through assessment of a mosquito’s vectorial capacity.Nature, 204, 1173-1175.
- GAUDREAULT, N. N., INDRAN, S. V., BALARAMAN, V., WILSON, W. C. & RICHT, J. A., 2019. Molecular aspects of Rift Valley fever virus and the emergence of reassortants. Virus Genes, 55, 1-11.
- GEAR, J. H. S., 1977. Hemorrhagic fevers of Africa: an account of two recent outbreaks. Journal of the South African Veterinary Association,48, 5-8.
- GEAR, J. H. S., 1978. National Institute for Virology, Sandringham 2131, South Africa. Personal communication.
- GEAR, J. H. S., DE MEILLON, B., LE ROUX, A. F. & KOFSKY, R., 1955. Rift Valley fever in South Africa; a study on the 1953 outbreak in the Orange Free State with special reference to the vectors and possible reservoir hosts. South African Medical Journal, 29, 514-518.
- GEAR, J. H. S., DE MEILLON, B., MEASROCH, V., HARWIN, R. & DAVIES, D. H. S., 1951. Rift Valley fever in South Africa: the occurrence of human cases in the Orange Free State, the north western Cape Province, the western and southern Transvaal. B. Field and laboratory investigations. South African Medical Journal,25, 980-912.
- GERRARD, S. R., BIRD, B. H., ALBARIÑO, C. G. & NICHOL, S. T., 2007. The NSm proteins of Rift Valley fever virus are dispensable for maturation, replication and infection. Virology, 359, 459-465.
- GERRARD, S. R., LI, L., BARRETT, A. D. & NICHOL, S. T., 2004. Ngari virus is a Bunyamwera virus reassortant that can be associated with large outbreaks of hemorrhagic fever in Africa. Journal of Virology, 78, 8922-8926.
- GERRARD, S. R. & NICHOL, S. T., 2007. Synthesis, proteolytic processing and complex formation of N-terminally nested precursor proteins of the Rift Valley fever virus glycoproteins. Virology, 357, 124-133.
- GF-TAD’S., 2012. Inter – regional conference Middle East & Horn of Africa. “RVF challenges, prevention and control”. Mombasa, Kenya held from12 to 15 November 2012. Available at: https://www.oie.int/doc/ged/D12326.PDF
- GHAFFAR, S. A., 1981. The epidemiological situation of RVF in Egypt up to December 1980. Journal of the Egyptian Public Health Association,56, 463-467.
- GIRARD, M., NELSON, C. B., PICOT, V. & GUBLER, D. J., 2020. Arboviruses: A global public health threat. Vaccine, 38(24), 3989-3994.
- GONZALEZ, J. P., BOUQUETY, J. C., LESBORDES, J. L., MADELON, M. C., MATHIOT, C. C., MEUNIER, D. M. Y. & GEORGES, A. J., 1987. Rift Valley fever virus and hemorrhagic fever in the Central African Republic. Annales de l’Institut Pasteur Virologie,138, 385-390.
- GORDON, S. W., TAMMARIELLO, R. F., LINTHICUM, K. J., DOHM, D. J., DIGOUTTE, J. P. & CALVO-WILSON, M. A., 1992. Arbovirus isolations from mosquitoes collected during 1988 in the Senegal River basin.American Journal of Tropical Medicine and Hygiene, 47, 742-748.
- GROBBELAAR, A. A., WEYER, J., LEMAN, P. A., KEMP, A., PAWESKA, J. T. & SWANEPOEL, R., 2011. Molecular epidemiology of Rift Valley fever virus. Emerging Infectious Diseases, 17, 2270-2276.
- GROBLER, D. G. & BENGIS, R., 1999. National Parks Board and Department of Veterinary Services, Kruger National Park, Skukuza 1350. Unpublished data
- GROSSI-SOYSTER, E. N. & LABEUD, A. D., 2020. Rift Valley fever: important consideration for risk mitigation and future outbreaks. Tropical Medicine and Infectious Diseases, 5, 89.
- GROSSI-SOYSTER, E. N., LEE, J., KING, C. H. & LA BEAUD, A. D., 2019. The influence of raw milk exposures on Rift Valley fever virus transmission. PLoS Neglected Tropical Diseases, 13, e0007258.
- GUDO, E. S., FALK, K. & CLIFF, J., 2018. Historical Perspective of Arboviruses in Mozambique and its implication for Current and Future Epidemics. Advances in Experimental Medical Biology, 1062, 11-18. doi: 10.1007/978-981-10-8727-1.
- GÜR, S., KALE, M., EROL, N., YAPICI, O., MAMAK, N. & YAVRU, S., 2017. The first serological evidence for Rift Valley fever infection in the camel, goitered gazelle and Anatolian water buffaloes in Turkey. Tropical Animal Health and Production, 49, 1531-1535.
- HABJAN, M., PICHLMAIR, A., ELLIOTT, R. M., OVERBY, A. K., GLATTER, T., GSTAIGER, M., SUPERTI-FURGA, G., UNGER, H. & WEBER, F., 2009. NSs protein of rift valley fever virus induces the specific degradation of the double-stranded RNA-dependent protein kinase. Journal of Virology, 83, 4365-4375.
- HANOTTE, O., BRADLEY, D. G., OCHIENG, J. W., VERJEE, Y., HILL, E. W. & REGE, J. E. O., 2002. African pastoralism: genetic imprints of origins and migrations. Science, 296, 336-339.
- HARRINGTON, D. G., LUPTON, H. W., CRABBS, C. L., PETERS, C. J., REYNOLDS, J. A. & SLONE, T. W. J., 1980. Evaluation of a formalin-inactivated Rift Valley fever vaccine in sheep. American Journal of Veterinary Research,41, 1559-1564.
- HARTLEY, D. M., RINDERKNECHT, J. L., NIPP, T. L., CLARKE, N. P. & SNOWDER, G. D., 2011. National Center for Foreign Animal and Zoonotic Disease Defense Advisory Group on Rift Valley Fever. Potential effects of Rift Valley fever in the United States. Emerging Infectious Diseases, 17, e1.
- HARTMAN, A. L., POWELL, D. S., BETHEL, L. M., CAROLINE, A. L., SCHMID, R. J., OURY, T. & REED, D. S., 2014. Aerosolized rift valley fever virus causes fatal encephalitis in african green monkeys and common marmosets. Journal of Virology, 88, 2235-2245.
- HEINRICH, N., SAATHOFF, E., WELLER, N., CLOWES, P., KROIDL, I., NTINGINYA, E., MACHIBYA, H., MABOKO, L., LOSCHER, T., DOBLER, G. & HOELSCHER, M., 2012. High Seroprevalence of Rift Valley Fever and Evidence for Endemic Circulation in Mbeya Region, Tanzania, in a Cross-Sectional Study. PLoS Neglected Tropical Diseases, 6, e1557.
- HEISE, M. T., WHITMORE, A., THOMPSON, J., PARSONS, M., GROBBELAAR, A. A., KEMP, A., PAWESKA, J. T., MADRIC, K., WHITE, L. J., SWANEPOEL, R. & BURT, F. J., 2009. An alphavirus replicon-derived candidate vaccine against Rift Valley fever virus. Epidemiology and Infection, 137, 1309-1318.
- HENDERSON, B. E., MCCRAE, A. W. R., KIRYA, B. G., SSENKUBUGE, Y. & SEMPALA, S. D. K., 1972. Arbovirus epizootics involving man, mosquitoes and vertebrates at Lunyo, Uganda 1968. Annals of Tropical Medicine and Parasitology, 66, 343-355.
- HENNING, M. W., 1956. Animal Diseases in South Africa. 3rd edn. Pretoria, South Africa: Central News Agency Ltd.
- HENRICHS, B., OOSTHUIZEN, M. C., TROSKIE, M., GORSICH, E., GONDHALEKAR, C., BEECHLER, B. R., EZENWA, V. O. & JOLLES, A. E., 2016. Within guild co-infections influence parasite community membership: a longitudinal study in African Buffalo. Journal of Animal Ecology, 85(4), 1025-1034.
- HIMEIDAN, Y. E., 2016. Rift Valley fever: current challenges and future prospects. Research and Reports in Tropical Medicine, 7, 1-9.
- HOCH, A. L., GARGAN, T. P. I. & BAILEY, C. L., 1985. Mechanical transmission of Rift Valley fever virus by hematophagous diptera. American Journal of Tropical Medicine and Hygiene, 34, 188-193.
- HOOGSTRAAL, H., MEEGAN, J. M., KHALIL, G. M. & ADHAM, F. K., 1979. The Rift Valley fever epizootic in Egypt 1977–78. 2. Ecological and entomological studies. Transactions of the Royal Society of Tropical Medicine and Hygiene, 73, 624-629.
- HOPKINS, G. H. E., 1952. Mosquitoes of the Ethiopian Region. I. Larval bionomics of mosquitoes and taxonomy of culicine larvae. 2nd edn. London: British Museum (Natural History).
- HOWELL, P. G., 1999. Faculty of Veterinary Science, University of Pretoria, Onderstepoort 0110. Unpublished data.
- HUBBARD, K. A., BASKERVILLE, A. & STEPHENSON, J. R., 1991. Ability of a mutagenized virus variant to protect young lambs from Rift Valley fever. American Journal of Veterinary Research,52, 50-55.
- HUISKONEN, J. T., OVERBY, A. K., WEBER, F. & GRÜNEWALD, K., 2009. Electron cryo-microscopy and single-particle averaging of Rift Valley fever virus: evidence for GN-GC glycoprotein heterodimers. Journal of Virology, 83, 3762-3769.
- HUNTER, P., ERASMUS, B. J. & VORSTER, J. H., 2001. Teratogenicity of a mutagenized Rift Valley fever virus (MVP12) in sheep. Onderstepoort Journal of Veterinary Research, 69, 95-98.
- HUSSEIN, N. A., CHIZYUKA, R. Z., KSIAZEK, T. G., SCOTT, R. M. & BOULOS, B. A. M., 1987. Epizootic of Rift Valley fever in Zambia, 1985. Veterinary Record,121, 111.
- IBRAHIM, M. S., TURELL, M. J., KNAUERT, F. K. & LOFTS, R. S., 1997. Detection of Rift Valley fever virus in mosquitoes by RT-PCR. Molecular and Cellular Probes,11, 49-53.
- IKEGAMI, T., 2012. Molecular biology and genetic diversity of Rift Valley fever virus. Antiviral Research, 95, 293-310.
- IKEGAMI, T., 2017. Rift Valley fever vaccines: an overview of the safety and efficacy of the live-attenuated MP-12 vaccine candidate. Expert Review of Vaccines, 16, 601-611.
- IKEGAMI, T., BALOGH, A., NISHIYAMA, S., LOKUGAMAGE, N., SAITO, T. B., MORRILL, J. C., SHIVANNA, V., INDRAN, S. V., ZHANG, L., SMITH, J. K., PEREZ, D., JUELICH, T. L., MOROZOV, I., WILSON, W. C., FREIBERG, A. N. & RICHT, J. A., 2017. Distinct virulence of Rift Valley fever Phlebovirus strains from different genetic lineages in a mouse model. PLoS One, 12, e0189250.
- IKEGAMI, T. & MAKINO, S., 2009. Rift valley fever vaccines. Vaccine, 27(4), D69-D72.
- IKEGAMI, T. & MAKINO, S., 2011. The pathogenesis of Rift Valley Fever. Viruses, 3, 493-519.
- IKEGAMI, T., NARAYANAN, K., WON, S., KAMITANI, W., PETERS, C. J. & MAKINO, S., 2009. Dual functions of Rift Valley fever virus NSs protein: inhibition of host mRNA transcription and post-transcriptional downregulation of protein kinase PKR. Annals of the New York Academy of Sciences, 1171, e75-e85.
- IKEGAMI, T., NARAYANAN, K., WON, S., KAMITANI, W., PETERS, C. J. & MAKINO, S., 2009. Rift Valley fever virus NSs protein promotes post-transcriptional downregulation of protein kinase PKR and inhibits eIF2alpha phosphorylation. PLoS Pathogens. 5, e1000287.
- IMAM, I. Z. E., EL KARAMANY, R. & DARWISH, M. A., 1979. An epidemic of Rift Valley fever in Egypt. II. Isolation of the virus from animals. Bulletin of the World Health Organization,57, 441-443.
- IMAM, I. Z. E., EL KARAMANY, R., OMAR, F. M. & EL KAFRAWY, O., 1981. Rift Valley fever in Egypt. Journal of the Egyptian Public Health Association,56, 356-383.
- INDRAN, S. V. & IKEGAMI, T., 2012. Novel approaches to develop Rift Valley fever vaccines. Frontiers in Cellular and Infection Microbiology, 2, 131.
- ISHAK, K. G., WALKER, D. H., COETZER, J. A. W., GARDNER, J. J. & GORELKIN, L., 1982. Viral hemorrhagic fevers with hepatic involvement: Pathologic aspects with clinical correlations. In:POPPER, H. & SCHAFFNER, F., (eds.). Progress in Liver Diseases, New York: Grune & Stratton, Inc, 3, 495-515.
- JÄCKEL, S., EIDEN, M., BALKEMA-BUSCHMANN, A., ZILLER, M., JANSEN VAN VUREN, P., PAWESKA, J. T. & GROSCHUP, M. H., 2013. A novel indirect ELISA based on glycoprotein Gn for the detection of IgG antibodies against Rift Valley fever virus in small ruminants. Research in Veterinary Science, 95(2), 725-730.
- JÄCKEL, S., EIDEN, M., EL MAMY, B. O., ISSELMOU, K., VINA-RODRIGUEZ, A., DOUMBIA, B. & GROSCHUP, M. H., 2013. Molecular and serological studies on the Rift Valley fever outbreak in Mauritania in 2010. Transboundary and Emerging Diseases, 60(2), 31-39.
- JANSEN VAN VUREN, P., KGALADI, J., MSIMANG, V. & PAWESKA, J. T., 2019. Rift Valley Fever Reemergence after 7 Years of Quiescence, South Africa, May 2018. Emerging Infectious Diseases, 25, 338-341.
- JANSEN VAN VUREN, P. & PAWESKA, J. T., 2009. Laboratory safe detection of nucleocapsid protein of Rift Valley fever virus in human and animal specimens by a sandwich ELISA, Journal of Virological Methods, 157, 15-24.
- JANSEN VAN VUREN, P., POTGIETER, A. C., PAWESKA, J. T. & VAN DIJK, A. A., 2007. Preparation and evaluation of a recombinant Rift Valley fever virus N protein for the detection of IgG and IgM antibodies in humans and animals. Journal of Virological Methods, 140, 106-114.
- JANSEN VAN VUREN, P., TIEMESSEN, C. T. & PAWESKA, J. T., 2011. Anti-nucleocapsid immune responses counteract pathogenic effects if Rift Valley fever virus infection in mice. PLoS One, 6, e2507.
- JAVELLE, E., LESUEUR, A., DE SANTI, V. P., DE LAVAL, F., LEFEBVRE, T., HOLWECK, G., DURAND, G. A., LEPARC-GOFFART, I. & TEXIER, G., 2020. The challenging management of Rift Valley fever in humans; literature revies of the clinical and algorithm proposal. Annals of Clinical Microbiology and Antimicrobials, 19, 4.
- JOHNSON, B. K., CHANAS, A. C., EL TAYEB, E., ABDEL-WAHAB, K. S. E., CHEHETA, F. A. & MOHAMED, A. E., 1978. Rift Valley fever in Egypt. Lancet,2, 745-745.
- JOHNSON, B. K., GITAU, L. G., GICHOGO, A., TUKEI, P. M., ELSE, J. G., SULEMAN, M. A., KIMANI, R. & SAYER, P. D., 1982. Marburg, Ebola and Rift Valley fever virus antibodies in East African primates. Transactions of the Royal Society of Tropical Medicine and Hygiene,76, 307-310.
- JORI, F., ALEXANDER, K. A., MOKOPASETSO, M., MUNSTERMANN, S., MOAGABO, K. & PAWESKA, J. T., 2015. Serological evidence of Rift Valley Fever virus circulation in domestic cattle and African buffalo in Northern Botswana (2010-2011). Froniters in Veterinary Science, 2, 63. doi:10.3389/fvets.2015.00063.
- JOUAN, A., ADAM, F., COULIBALY, I., RIOU, O., PHILIPPE, B., LEDRU, E., LEJAN, C., MERZOUG, N. O., KSIAZEK, T. & LEGUENNO, B., 1990. [Epidemic of Rift Valley fever in the Islamic republic of Mauritania. Geographic and ecological data]. Bulletin de la Societe de Pathologie Exotique,83, 611-620.
- JOUAN, A., ADAM, F., RIOU, O., PHILIPPE, B., MERZOUG, N. O., KSIAZEK, T., LEGUENNO, B. & DIGOUTTE, J. P., 1990. [Evaluation of indicators of health in the area of Trarza during the epidemic of Rift Valley fever in 1987]. Bulletin de la Societe de Pathologie Exotique,83, 621-627.
- JOUAN, A., COULIBALY, I., ADAM, F., PHILIPPE, B., RIOU, O., LEGUENNO, B., CHRISTIE, R., MERZOUG, N. O., KSIAZEK, T. & DIGOUTTE, J. P., 1989. Analytical study of a Rift Valley fever epidemic. Research in Virology,40, 175-186.
- JOUAN, A., LEGUENNO, B., DIGOUTTE, J. P., PHILLIPE, B., RIOU, O. & ADAM, F., 1988. An RVF epidemic in southern Mauritania. Annales de l’Institut Pasteur Virologie,139, 307-308.
- JOUBERT, J. D. S., FERGUSON, A. L. & GEAR, J., 1951. Rift Valley fever in South Africa. II. The occurrence of human cases in the Orange Free State the North-western Cape Province, the Western and Southern Transvaal. A. Epidemiological and clinical findings. South African Medical Journal, 25, 890-891.
- JUPP, P. G., 1969. Preliminary studies on the overwintering stages of Culex mosquitoes (Diptera: Culicidae) in the Highveld region of South Africa. Journal of the Entomological Society of Southern Africa, 32, 91-98.
- JUPP, P. G., 1973. Field studies on the feeding habits of mosquitoes in the Highveld region of South Africa. South African Journal of Medical Sciences, 38, 69-83.
- JUPP, P. G., 1975. Further studies on the overwintering stages of Culex mosquitoes (Diptera: Culicidae) in the Highveld region of South Africa. Journal of the Entomological Society of Southern Africa, 38, 89-97.
- JUPP, P. G., 1990. National Institute for Virology, Sandringham 2131, South Africa. Personal communication.
- JUPP, P. G. & CORNEL, A. J., 1988. Vector competence tests with Rift Valley fever virus and five South African species of mosquitoes. Journal of the American Mosquito Control Association, 4, 4-8.
- JUPP, P. G., GROBBELAAR, A. A., LEMAN, P. A., KEMP, A., DUNTON, R. F., BURKOT, T. R., KSIAZEK, T. G. & SWANEPOEL, R., 2000. Experimental detection of Rift Valley fever virus by reverse transcription-polymerase chain reaction assay in large samples of mosquitoes.Journal of Medical Entomology, 37, 467-471.
- JUPP, P. G. & KEMP, A., 1998. Studies on an outbreak of Wesselsbron virus in the Free State Province, South Africa. Journal of the American Mosquito Control Association, 14, 40-45
- JUPP, P. G., KEMP, A., GROBBELAAR, A., LEMAN, P., BURT, F. J., ALAHMED, A. M., AL MUJALLI, D., AL KHAMEES, M. & SWANEPOEL, R., 2002. The 2000 epidemic of Rift Valley fever in Saudi Arabia: Mosquito vector studies.Medical and Veterinary Entomology, 16, 245-252.
- JUPP, P. G. & MCINTOSH, B. M., 1967. Ecological studies on Sindbis and West Nile viruses in South Africa. II. Mosquito bionomics. South African Journal of Medical Sciences, 32, 15-33.
- JUPP, P. G. & MCINTOSH, B. M., 1987. A bionomic study of adult Aedes (Neomelaniconion) circumluteolus in northern Kwazulu, South Africa. Journal of the American Mosquito Control Association, 3, 131-136.
- JUPP, P. G., MCINTOSH, B. M. & NEVILL, E. M., 1980. A survey of the mosquito and Culicoides faunas at two localities in the Karoo region of South Africa with some observations on bionomics. Onderstepoort Journal of Veterinary Research, 47, 1-6.
- JUPP, P. G., MCINTOSH, B. M. & THOMPSON, O. L., 1984. Mechanical transmission of Rift Valley fever virus by mosquitoes. South African Journal of Science, 80, 276.
- KADING, R. C., ABWORO, E. O. & HAMER, G. L., 2019. Rift Valley Fever Virus, Japanese Encephalitis Virus, and African Swine Fever Virus: Three Transboundary, Vector-Borne, Veterinary Biothreats With Diverse Surveillance, and Response Capacity Needs. Frontiers in Veterinary Science, 6, 458.
- KADING, R. C., CRABTREE, M. B., BIRD, B. H., NICHOL, S. T., ERICKSON, B. R., HORIUCHI, K., BIGGERSTAFF, B. J. & MILLER, B. R., 2014. Deletion of the NSm virulence gene of Rift Valley fever virus inhibits virus replication in and dissemination from the midgut of Aedes aegypti mosquitoes. PLoS Neglected Tropical Diseases, 8, e2670.
- KAKANI, S., LABEAUD, A. D. & KING, C. H., 2010. Planning for Rift Valley fever virus: use of geographical information systems to estimate the human health threat of white-tailed deer (Odocoileus virginianus)-related transmission. Geospat Health, 5, 33-43.
- KALVERAM, B., LIHORADOVA, O. & IKEGAMI, T., 2011. NSs protein of Rift Valley fever virus promotes postranslational downregulation of the TFIIH subunit p62. Journal of Virology, 85, 6234-6243.
- KAMEL, S., 1981. Epidemiology of RVF in domestic animals in Egypt. Journal of the Egyptian Public Health Association,56, 474-476.
- KARIMURIBO, E. D., MBOERA, L. E. G., MBUGI, E., SIMBA, A., KIVARIA, F. M., MMBUJI, P. & RWEYEMAMU, M. M., 2011. Are we prepared for emerging and re-emerging diseases? Experience and lessons from epidemics that occurred in Tanzania during the last five decades. Tanzania Journal of Health Research, 13, 387-398.
- KARK, J. D., AYNOR, Y. & PETERS, C. J., 1982. A Rift Valley fever vaccine trial. I. Side effects and serologic response over a six-month folow-up. American Journal of Epidemiology,116, 808-820.
- KASARI, T. R., CARR, D. A., LYNN, T. V. & WEAVER, J. T., 2008. Evaluation of pathways for release of Rift Valley fever virus into domestic ruminant livestock, ruminant wildlife, and human populations in the continental United States. Journal of the American Veterinary Medical Association, 232, 514-529.
- KASCHULA, V. R., 1953. The propogation and modification of strains of Rift Valley fever viruses in embryonated eggs and their use as immunizing agents for domestic ruminants. D.V.Sc. thesis, University of Pretoria.
- KEEFER, G. V., ZEBRATH, G. L. & ALLEN, W. P., 1972. Susceptibility of dogs and cats to Rift Valley fever by inhalation or ingestion of virus. Journal of infectious Diseases,125, 307-309.
- KELLERMAN, T. S., W., C. J. A. & NAUDÉ, T. W., 1988. Plant Poisonings and Mycotoxicoses of Livestock in Southern Africa. Cape Town: Oxford University Press Southern Africa.
- KENAWY, M. A., ABDEL-HAMID, Y. M. & BEIER, J. C., 2018. Rift Valley fever in Egypt and other African countries: Historical review, recent outbreaks and possibility of disease occurrence in Egypt. Acta Tropica, 181, 40-49.
- KENNETH, J., LINTHICUM, K. J., SETH, C., BRITCH, S. C. & ANYAMBA, A., 2016. Rift Valley Fever: An Emerging Mosquito-Borne Disease. Annual Review of Entomology, 61, 395-415.
- KHALIL, G. M., 1981. Laboratory investigation of Rift Valley fever virus vectors. Journal of the Egyptian Public Health Association,56, 468-473.
- KIFARO, E. G., NKANGAGA, J., JOSHUA, G., SALLU, R., YONGOLO, M., DAUTU, G. & KASANGA, C. J., 2014. Epidemiological study of Rift Valley fever virus in Kigoma, Tanzania. Onderstepoort Journal of Veterinary Research, 81, e1-e5.
- KILPATRICK, M. E., GIRGIS, N. I., FARID, Z. & SIPPEL, J. E., 1991. Epidemiology, prevalence and clinical diagnosis of meningitis at Abbassia Fever Hospital, Cairo, 1966–1989. Transactions of the Royal Society of Tropical Medicine and Hygiene, 85(1), 4-5.
- KIM, Y., DOMMERGUES, L., M’SA, A. B., MÉROT, P., CARDINALE, E., EDMUNDS, J., PFEIFFER, D., FOURNIÉ, G. & MÉTRAS, R., 2018. Livestock trade network: potential for disease transmission and implications for risk-based surveillance on the island of Mayotte. Scientific Reports, 8, 11550. https://doi.org/10.1038/s41598-018-29999-y.
- KIMANI, T., SCHELLING, E. & BETT, B., 2016. Public health benefits from livestock Rift Valley fever control: a simulation of two epidemics in Kenya. EcoHealth, 13, 729-742.
- KITCHEN, S. F., 1934. Laboratory infections with the virus of Rift Valley fever. American Journal of Tropical Medicine and Hygiene, 1, 547-564.
- KLASTRUP, N. O. & HALLIWELL, R. W., 1977. Infectious causes of infertility/ abortion of cattle in Malawi. Nordisk Veterinaer Medicine,29, 325-330.
- KOHL, A., DI, B. & BOULOY, M., 1999. The Rift Valley fever virus nonstructural protein NSs is phosphorylated at serine residues located in casein kinase II consensus motifs in the carboxy-terminus. Virology, 263, 517-525.
- KOKERNOT, R. H., SMITHBURN, K. C., GANDARA, A. F., MCINTOSH, B. M. & HEYMANN, C. S., 1960. Provas de neutralizaçao com soros de indivíduos residentes em Moçambique contra determinados vírus isolados em Africa transmitidos por artrópodes. Anals Do Instituto De Medicino Tropical, 17, 201-230.
- KOKERNOT, R. H., SZLAMP, E. L., LEVITT, J. & MCINTOSH, B. M., 1965. Surveys for antibodies against arthropod-borne viruses in the sera of indigenous residents of the Caprivi Strip and Bechuanaland Protectorate. Transactions of the Royal Society of Tropical Medicine and Hygiene, 59, 553-562.
- KORTEKAAS, J., 2014. One Health approach to Rift Valley fever vaccine development. Antiviral Research, 106, 24-32.
- KORTEKAAS, J., ORESHKOVA, N., COBOS-JIMÉNEZ, V., VLOET, R. P. M., POTGIETER, C. A. & MOORMANN, R. J. M., 2011. Creation of a nonspreading Rift Valley fever virus. Journal of Virology, 85, 12622-12630.
- KROEKER, A. L., BABIUK, S., PICKERING, B. S., RICHT, J. A. & WILSON, W. C., 2020. Livestock challenge models of Rift Valley fever for agricultural vaccine testing. Frontiers in Veterinary Science, 7, 238.
- KROEKER, A. L., SMID, V., EMBURY-HYATT, C., MOFFAT, E., COLLIGNON, B., LUNG, O., LINDSAY, R. & WEINGARTL, H., 2018. RVFV Infection in Goats by Different Routes of Inoculation. Viruses, 10.
- KSIAZEK, T. G., JOUAN, A., MEEGAN, J. M., LEGUENNO, B., WILSON, M. L., PETERS, C. J., DIGOUTTE, J. P., GUILLAUD, M., MERZOUG, N. O. & TOURAY, E. M., 1989. Rift Valley fever among domestic animals in the recent West African outbreak. Research in Virology,140, 67-77.
- LA BEAUD, A. D., MUIRURI, S., SUTHERLAND, L. J., DAHIR, S., GILDENGORIN, G., MORRILL, J., PETERS, C. J., HISE, A. G., KAZURA, J. W. & KING, C. H., 2011. Post epidemic analysis of Rift Valley fever virus transmission in north eastern Kenya: a village cohort study. PLoS Neglected Tropical Diseases, 5, e1265.
- LABEAUD, D., 2010. Towards a safe, effective vaccine for Rift Valley fever virus. Future Virology, 5, 675-678.
- LAM, T. T. Y., LIU, W., BOWDEN, T. A., CUI, N., ZHUANG, L., LIU, K., ZHANG, Y. Y., CAO, W. C. & PYBUS, O. G., 2013. Evolutionary and molecular analysis of the emergent severe fever with thrombocytopenia syndrome virus. Epidemics, 5, 1-10.
- LANCELOT, R., GONZALEZ, J. P., LE GUENNO, B., DIALLO, B. C., GANDEGA, Y. & GUILLAUD, M., 1990. [Descriptive epidemiology of Rift Valley fever in small ruminants in Southern Mauritania after the 1988 rainy season]. Revue d’Élevage et de Médécine Veterinaire des Pays Tropicaux, 42, 485-491.
- LAUGHLIN, L. W., MEEGAN, J. M., STRAUSBAUGH, L. J., MORENS, D. M. & WATTEN, H., 1979. Epidemic Rift Valley fever in Egypt: Observations of the spectrum of human illness. Transactions of the Royal Society of Tropical Medicine and Hygiene,73, 630-633.
- LE MAY, N., MANSUROGLU, Z., LEGER, P., JOSSE, T., BLOT, G., BILLECOCQ, A., FLICK, R., JACOB, Y., BONNEFOY, E. & BOULOY, M., 2008. A SAP30 complex inhibits IFN-beta expression in Rift Valley fever virus infected cells. PLoS Pathogens, 4, e13.
- LE ROUX, C. A., KUBO, T., GROBBELAAR, A. A., JANSEN VAN VUREN, P., WEYER, J., NEL, L. H., SWANEPOEL, R., MORITA, K. & PAWESKA, J. T., 2009. Development and evaluation of a real-time reverse transcription-loop-mediated isothermal amplification assay for rapid detection of Rift Valley fever virus in clinical specimens. Journal of Clinical Microbiology, 47, 645-651.
- LE ROUX, J. M. W., 1959. The histopathology of Wesselsbron disease in sheep. Onderstepoort Journal of Veterinary Research, 28, 237-243.
- LECATSAS, G. & WEISS, K. E., 1968. Electron microscopic studies on BHK21 cells infected with Rift Valley fever. Archiv fu¨r die Gesamte Virusforschung., 25, 58-64.
- LEE, V. H., 1979. Isolation of viruses from field populations of Culicoides (Diptera: Ceratopogonidae) in Nigeria. Journal of Medical Entomology, 16, 76-79.
- LEFKOWITZ, E. J., DEMPSEY, D. M., HENDRICKSON, R. C., ORTON, R. J., SIDDELL, S. G. & SMITH, D. B., 2018. Virus taxonomy: the database of the International Committee on Taxonomy of Viruses (ICTV). Nucleic Acids Research, 46, D708-D717.
- LERDTHUSNEE, K. & ROMOSER, W. S., 1988. Sites of Rift Valley fever virus infection in the proventriculus of adult Culex pipiens. Abstracts of the 37th Annual Meeting of the American Society of Tropical Medicine and Hygiene,Washington DC.
- LERDTHUSNEE, K., ROMOSER, W. S., FARAN, M. E. & DOHM, D. J., 1995. Rift Valley fever virus in the cardia of Culex pipiens:an immunocytochemical and ultrastructural study. American Journal of Tropical Medicine and Hygiene, 53, 331-337.
- LERNOUT, T., CARDINALE, E., JEGO, M., DESPRES, P., COLLET, L., ZUMBO, B., TILLARD, E., GIRARD, S. & FILLEUL, L., 2013. Rift Valley Fever in Humans and Animals in Mayotte, an Endemic Situation? PLoS One, 8, e74192.
- LEWIS, J. C., BOTROS, B. A. M. & MEEGAN, J. M., 1978. Studies of Rift Valley fever in the domestic animals of Egypt. Journal of the Egyptian Public Health Association,53, 271-272.
- LINTHICUM, K. J., ANYAMBA, A., BRITCH, S. C., CHRETIEN, J. P., ERICKSON, R. L., SMALL, J., TUCKER, C. J., BENNETT, K. E., MAYER, R. T., SCHMIDTMANN, E. T., ANDREADIS, T. G., ANDERSON, J. F., WILSON, W. C., FREIER, J. E., JAMES, A. M., MILLER, R. S., DROLET, B. S., MILLER, S. N., TEDROW, C. A., BAILEY, C. L., STRICKMAN, D. A., BARNARD, D. R., CLARK, G. G. & ZOU, L., 2007. A Rift Valley fever risk surveillance system for Africa using remotely sensed data: potential for use on other continents. Veterinaria Italiana, 43, 663-674.
- LINTHICUM, K. J., ANYAMBA, A., TUCKER, C. J., KELLEY, P. W., MYERS, M. F. & PETERS, C. J., 1999. Climate and satellite indicators to forecast Rift Valley fever epidemics in Kenya. Science,285, 397-400.
- LINTHICUM, K. J. & BAILEY, C. L., 1985. Observations on the dispersal and survival of a population of Aedes lineatopennis(Ludlow) (Diptera: Culicidae) in Kenya. Bulletin of Entomological Research, 75, 661.
- LINTHICUM, K. J., BAILEY, C. L., DAVIES, F. G. & TUCKER, C. J., 1987. Detection of Rift Valley fever viral activity in Kenya by satellite remote sensing imagery. Science,235, 1656-1659.
- LINTHICUM, K. J., BAILEY, C. L., TUCKER, C. J., MITCHELL, K. D., LOGAN, T. M., DAVIES, F. G., KAMAU, C. W., THANDE, P. C. & WAGATEH, J. N., 1990. Application of polar-orbiting meteorological satellite data to detect flooding of Rift Valley fever virus vector mosquito habitats in Kenya. Medical and Veterinary Entomology, 4, 433-438.
- LINTHICUM, K. J., DAVIES, F. G., BAILEY, C. L. & KAIRO, A., 1983. Mosquito species succession in a dambo in an East African forest. Mosquito News,43, 464-470.
- LINTHICUM, K. J., DAVIES, F. G., BAILEY, C. L. & KAIRO, A., 1984. Mosquito species encountered in a flooded grassland dambo in Kenya. Mosquito News, 44, 228-232.
- LINTHICUM, K. J., DAVIES, F. G. & KAIRO, A., 1984. Observations of the biting activity of mosquitoes at a flooded dambo in Kenya. Mosquito News, 44, 589-595.
- LINTHICUM, K. J., DAVIES, F. G., KAIRO, A. & BAILEY, C. L., 1985. Rift Valley fever virus (family Bunyaviridae, genus Phlebovirus). Isolations from Diptera collected during an inter-epizootic period in Kenya. Journal of Hygiene, Cambridge, 95, 197-209.
- LINTHICUM, K. J., LOGAN, T. M., BAILEY, C. L., DOHM, D. J. & MOULTON, J. R., 1989. Transstadial and horizontal transmission of Rift Valley fever virus in Hyalomma truncatum. American Journal of Tropical Medicine and Hygiene, 41, 491-496.
- LINTHICUM, K. J., SETH, C., BRITCH, S. C. & ANYAMBA, A., 2016. Rift Valley Fever: An Emerging Mosquito-Borne Disease. Annual Review of Entomology, 61, 395-415.
- LIU, L., CELMA, C. C. P. & ROY, P., 2008. Rift Valley fever virus structural proteins: Expression, characterization and assembly of recombinant proteins. Virology Journal, 5, 82.
- LO, M. M., MBAO, V., SIERRA, P., THIONGANE, Y., DIP, M., DONADEU, M. & DUNGU, B., 2015. Safety and immunogenicity of Onderstepoort Biological Products’ Rift Valley fever Clone 13 vaccine. Onderstepoort Journal of Veterinary Research, 82, 857.
- LOGAN, T. M., LINTHICUM, K. J., THANDE, P. C., WAGATEH, J. N., NELSON, G. O. & ROBERTS, C. R., 1991. Egg hatching of Aedes mosquitoes during successive floodings in a Rift Valley fever endemic area in Kenya. Journal of the American Mosquito Control Association, 7, 109-112.
- LOGAN, T. M., LINTHICUM, K. J., THANDE, P. C., WAGATEH, J. N. & ROBERTS, C. R., 1991. Mosquito species collected from a marsh in western Kenya during the long rains. Journal of the American Mosquito Control Association, 7, 395-399.
- LORENZO, G., LÓPEZ-GIL, E., WARIMWE, G. M. & BRUN, A., 2015. Understanding Rift Valley fever: contributions of animal models to disease characterization and control. Molecular Immunology, 66, 78-88.
- LOZACH, P. Y., KUHBACHER, A., MEIER, R., MANCINI, R., BITTO, D., BOULOY, M. & HELENIUS, A., 2011. DC-SIGN as a receptor for phleboviruses. Cell Host Microbe, 10, 75-88.
- LUMLEY, S., HORTON, D. L., HERNANDEZ-TRIANA, L. L. M., JOHNSON, N., FOOKS, A. R. & HEWSON, R., 2017. Rift Valley fever virus: strategies for maintenance, survival and vertical transmission in mosquitoes. Journal of General Virology, 98, 875-887.
- LY, H. J., NISHIYAMA, S., LOKUGAMAGE, N., SMITH, J. K., ZHANG, L., PEREZ, D., JUELICH, T. L., FREIBERG, A. N. & IKEGAMI, T., 2017. Attenuation and protective efficacy of Rift Valley fever phlebovirus rMP12-GM50 strain. Vaccine, 35, 6634-6642.
- MAAR, S. A., SWANEPOEL, R. & GELFAND, M., 1979. Rift Valley fever encephalitis: a description of a case. Central African Journal of Medicine,5, 8-11.
- MACDONALD, G., 1957. The epidemiology and Control of Malaria. London: Oxford University Press.
- MACIAS, V. M., OHM, J. R. & RASGON, J. L., 2017. Gene Drive for Mosquito Control: Where Did It Come from and Where Are We Headed? International Journal of Environmental Research and Public Health, 14.
- MADANI, T. A., AL-MAZROU, Y. Y., AL-JEFFRI, M. H., MISHKHAS, A. A., AL-RABEAH, A. M., TURKISTANI, A. M., AL-SAYED, M. O., ABODAHISH, A. A., KHAN, A. S., KSIAZEK, T. G. & SHOBOKSHI, O., 2003. Rift Valley fever epidemic in Saudi Arabia: epidemiological, clinical, and laboratory characteristics. Clinical Infectious Diseases, 37, 1084-1092.
- MAES, P., et al., 2018. Taxonomy of the family Arenaviridae and the order Bunyavirales: update 2018. Archives of Virology, 163(8), 2295-2310.
- MAES, P., HARRIES, A. D., VAN DEN BERGH, R., NOOR, A., SNOW, R. W., TAYLER-SMITH, K., HINDERAKER, S. G., ZACHARIAH, R. & ALLAN, R., 2014. Can timely vector control interventions triggered by atypical environmental conditions prevent malaria epidemics? A case-study from Wajir County, Kenya. PLoS One, 9, e92386.
- MAGURANO, F. & NICOLETTI, L., 1999. Humoral response in Toscana virus acute neurological diseases investigated by viral-protein-specific immunoassays. Clinical and Diagnostic Laboratory Immunology, 6, 55-60.
- MAHDY, M. S., BANSEN, E., JOSHUA, J. M., PARKER, J. A. & STUART, P. F., 1979. A case report of Rift Valley fever with retinopathy. Canada Diseases Weekly Report, 5, 189-192.
- MAKOSCHEY, B., VAN KILSDONK, E., HUBERS, W. R., VRIJENHOEK, M. P., SMIT, M., WICHGERS SCHREUR, P. J., KORTEKAAS, J. & MOULIN, V., 2016. Rift Valley Fever Vaccine Virus Clone 13 Is Able to Cross the Ovine Placental Barrier Associated with Foetal Infections, Malformations, and Stillbirths. PLoS Neglected Tropical Diseases, 10, e0004550.
- MALIK, S. K. A., 1981. Epidemiology of Rift Valley fever in domestic animals in Egypt. Proceedings of the 49th General Session of the Office International des Epizooties, Paris, 25-30 May.
- MALULEKE, M. R., PHOSIWA, M., VAN SCHALKWYK, A., MICHUKI, G., LUBISI, B. A., KEGAKILWE, P. S., KEMP, S. J. & MAJIWA, P. A. O., 2019. A comparative genome analysis of Rift Valley Fever virus isolates from foci of the disease outbreak in South Africa in 2008-2010. PLoS Neglected Tropical Diseases, 13, e0006576.
- MANDEL, R. B., KOUKUNTLA, R., MOGLER, L. J., CARZOLI, A. K., FREIBERG, A. N., HOLBROOK, M. R., MARTIN, B. K., STAPLIN, W. R., VAHANIAN, N. N., LINK, C. J. & FLICK, R., 2010. A replication-incompetent Rift Valley fever vacine:chimeric virus-like particles protect mice and rats agaist lethal challenge. Virology, 397, 187-198.
- MANORE, C. A. & BEECHLER, B. R., 2015. Inter-epidemic and between-season persistence of rift valley fever: vertical transmission or cryptic cycling? Transboundary and Emerging Diseases, 62, 13-23.
- MANSFIELD, K. L., BANYARD, A. C., MCELHINNEY, L., JOHNSON, N., HORTON, D. L., HERNÁNDEZ-TRIANA, L. M. & FOOKS, A. R., 2015. Rift Valley fever virus: A review of diagnosis and vaccination, and implications for emergence in Europe.Vaccine, 33, 5520-5531.
- MARTIN, V., CHEVALIER, V., CECCATO, P., ANYAMBA, A., DE SIMONE, L., LUBROTH, J., DE LA ROCQUE, S. & DOMENECH, J., 2008. The impact of climate change on the epidemiology and control of Rift Valley fever. Revue Scientifique et Technique (International Office of Epizootics), 27, 413-426.
- MATIKO, M. K., SALEKWA, L. P., KASANGA, C. J., KIMERA, S. I., EVANDER, M. & NYANGI, W. P., 2018. Serological evidence of inter-epizootic/inter-epidemic circulation of Rift Valley fever virus in domestic cattle in Kyela and Morogoro, Tanzania. PLoS Neglected Tropical Diseases, 12, e0006931.
- MATUMOTO, M., IWASA, S. & ENDO, M., 1950. Complement fixation reaction of Rift Valley fever virus. Japanese Journal of Experimental Medicine,20, 501-508.
- MBOTHA, D., BETT, B., KAIRU-WANYOIKE, S., GRACE, D., KIHARA, A., WAINAINA, M., HOPPENHEIT, A., CLAUSEN, P.-H. & LINDAHL, J., 2018. Inter-epidemic Rift Valley fever virus seroconversions in an irrigation scheme in Bura, south-east Kenya. Transboundary Emerging Diseases, 65, e55-e62.
- MCELROY, A. K., ALBARINO, C. G. & NICHOL, S. T., 2009. Development of a RVFV ELISA that can distinguish infected from vaccinated animals. Virology Journal, 6, 125.
- MCGAVRAN, M. H. & EASTERDAY, B. C., 1963. Rift Valley fever virus hepatitis: Light and electron microscopic studies in the mouse. American Journal of Pathology,42, 587-607.
- MCINTOSH, B. M., 1961. Susceptibility of some African wild rodents to infection with various arthropod-borne viruses. Transactions of the Royal Society of Tropical Medicine and Hygiene, 55, 63-68.
- MCINTOSH, B. M., 1971. The aedine subgenus Neomelaniconion Newstead (Culicidae, Diptera) in southern Africa with descriptions of two new species. Journal of the Entomological Society of Southern Africa, 34, 319-333.
- MCINTOSH, B. M., 1972. Rift Valley fever. I. Vector studies in the field. Journal of the South African Veterinary Association, 43, 391-395.
- MCINTOSH, B. M., 1975. Mosquitoes as vectors of viruses in Southern Africa. Entomology Memoir, Department of Agricultural Technical Services of the Republic of South Africa, 1-19.
- MCINTOSH, B. M., 1976. National Institute for Virology, Sandringham 2131, South Africa. Personal communication.
- MCINTOSH, B. M., 1980. The epidemiology of arthropod-borne viruses in southern Africa. D.Sc. Thesis, University of Pretoria.
- MCINTOSH, B. M., DICKINSON, D. B. & DOS SANTOS, I., 1973. Rift Valley fever. IV. The susceptibility of mice and hamsters in relation to transmission of virus by mosquitoes. Journal of the South African Veterinary Association,44, 167-169.
- MCINTOSH, B. M. & JUPP, P. G., 1981. Epidemiological aspects of Rift Valley fever in South Africa with reference to vectors. Contributions to Epidemiology and Biostatistics, 3: Rift Valley fever, 92-99.
- MCINTOSH, B. M., JUPP, P. G., ANDERSON, D. & DICKINSON, D. B., 1973. Rift Valley fever. II. Attempts to transmit virus with seven species of mosquitoes. Journal of the South Africa Veterinary Association, 44, 57-60.
- MCINTOSH, B. M., JUPP, P. G. & DE SOUSA, J., 1972. Further isolations of arboviruses from mosquitoes collected in Tongaland, South Africa, 1960–1968. Journal of Medical Entomology, 9, 155-159.
- MCINTOSH, B. M., JUPP, P. G. & DE SOUSA, J., 1972. Mosquitoes feeding at two horizontal levels in gallery forest in Natal, South Africa, with reference to possible vectors of chikungunya virus. Journal of the Entomological Society of Southern Africa, 35, 81-90.
- MCINTOSH, B. M., JUPP, P. G., DOS SANTOS, I. & BARNARD, B. J. H., 1980. Vector studies on Rift Valley fever in South Africa. South African Medical Journal, 58, 127-132.
- MCINTOSH, B. M., JUPP, P. G., DOS SANTOS, I. & ROWE, A. C., 1983. Field and laboratory evidence implicating Culex zombaensis and Aedes circumluteolus as vectors of Rift Valley fever virus in coastal South Africa. South African Medical Journal of Science, 79, 61-64.
- MCINTOSH, B. M., RUSSELL, D., DOS SANTOS, I. & GEAR, J. H. S., 1980. Rift Valley Fever in humans in South Africa. South African Medical Journal, 58, 803-806.
- MCMILLEN, C. M., ARORA, N., BOYLES, D. A., ALBE, J. R., KUJAWA, M. R., BONADIO, J. F., COYNE, C. B. & HARTMAN, A. L., 2018. Rift Valley fever virus induces fetal demise in Sprague-Dawley rats through direct placental infection. Science Advances, 4, eaau9812.
- MCMILLEN, C. M. & HARTMAN, A. L., 2018. Rift Valley fever in animals and humans: Current perspectives. Antiviral Research, 156, 29-37.
- MCMILLEN, C. M. & HARTMAN, A. L., 2021. Rift Valley fever: a threat to pregnant women hiding in plain sight? Journal of Virology. doi: 10.1128/JVI.01394-19.
- MEADORS, G. F. I., GIBBS, P. H. & PETERS, C. J., 1986. Evaluations of a new Rift Valley fever vaccine: Safety and immunogenicity trials. Vaccine, 4, 179-184.
- MEEGAN, J. M., 1979. The Rift Valley fever epizootic in Egypt 1977–1978. I. Description of the epizootic and virological studies. Transactions of the Royal Society of Tropical Medicine and Hygiene, 73, 618-623.
- MEEGAN, J. M., 1981. Rift Valley fever in Egypt: An overview of the epizootics in 1977 and 1978. Contributions to Epidemiology and Biostatistics, 3: Rift Valley fever, 100-113.
- MEEGAN, J. M. & BAILEY, C. L., 1989. Rift Valley fever. In:MONATH, T. P., (ed.). The Arboviruses: Epidemiology and Ecology. Vol. IV. Boca Raton, Florida: CRC Press Inc, 51-57.
- MEEGAN, J. M., KHALIL, G. M., HOOGSTRAAL, H. & ADHAM, F. K., 1980. Experimental transmission and field isolation studies implicating Culex pipiens as a vector of Rift Valley fever virus in Egypt. American Journal of Tropical Medicine and Hygiene, 29, 1405-1410.
- MEEGAN, J. M. & SHOPE, R. E., 1981. Emerging concepts on Rift Valley fever. Perspectives in Virology, 11, 267-287.
- MEEGAN, J. M., WATTEN, R. H. & LAUGHLIN, L. W., 1981. Clinical experience with Rift Valley fever in humans during the 1977 Egyptian epizootic.Contributions to Epidemiology and Biostatistics, 3, 114-1223.
- MEHAND, M. S., AL-SHORBAJI, F., MILLETT, P. & MURGUE, B., 2018. The WHO R&D Blueprint: 2018 review of emerging infectious diseases requiring urgent research and development efforts. Antiviral Research, 159, 63-67.
- MEMISH, Z. A., MASRI, M. A., ANDERSON, B. D., HEIL, G. L., MERRILL, H. R., KHAN, S. U., ALSAHLY, A. & GRAY, G. C., 2015. Elevated antibodies against Rift Valley fever virus among humans with exposure to ruminants in Saudi Arabia. American Journal of Tropical Medicine and Hygiene, 92, 739-743.
- MÉTRAS, R., COLLINS, L. M., WHITE, R. G., ALONSO, S., CHEVALIER, V., THURANIRA-MCKEEVER, C. & PFEIFFER, D. U., 2011. Rift Valley fever epidemiology, surveillance, and control: what have models contributed? Vector-Borne and Zoonotic Diseases, 11, 761-771.
- MÉTRAS, R., EDMUNDS, W. J., YOUSSOUFFI, C., DOMMERGUES, L., FOURNIÉ, G., CAMACHO, A., FUNK, S., CARDINALE, E., LE GODAIS, G., COMBO, S., FILLEUL, L., YOUSSOUF, H. & SUBIROS, M., 2020. Estimation of Rift Valley fever virus spillover to humans during the Mayotte 2018–2019 epidemic. Proceedings of the National Academy of Sciences of the United States of America, 117(39), 24567-24574.
- METRAS, R., JEWELL, C., PORPHYRE, T., THOMPSON, P. N., PFEIFFER, D. U., COLLINS, L. M. & WHITE, R. G., 2015. Risk factors associated with Rift Valley fever epidemics in South Africa in 2008-11. Scientific Reports, 6, 9492, doi: 10.1038/srep09492.
- MÉTRAS, R., PORPHYRE, T., PFEIFFER, D. U., KEMP, A., THOMSON, P., COLLINS, L. M. & WHITE, R. G., 2012. Exploratory space-time analyses of Rift Valley fever in South Africa in 2008-2011. PLoS Neglected Tropical Diseases, 6, e1808.
- MILLER, B. R., GODSEY, M. S., CRABTREE, M. B., SAVAGE, H. M., AL-MAZRAO, Y., AL-JEFFRI, M. H., ABDOON, A. M., AL-SEGHAYER, S. M., AL-SHAHRANI, A. M. & KSIAZEK, T. G., 2002. Isolation and genetic characterization of Rift Valley fever virus from Aedes vexans arabiensis, Kingdom of Saudi Arabia. Emerging Infectious Diseases, 8, 1492-1494.
- MIMS, C. A., 1955. Rift Valley fever in mice. East African Virus Research Institute Annual Report.
- MIMS, C. A., 1956. The coagulation defect in Rift Valley fever and yellow fever virus infections. Annals of Tropical Medicine and Parasitology,50, 147-149.
- MIMS, C. A., 1956. Rift Valley fever in mice. I. General features of the infection. British Journal of Experimental Pathology, 37, 99-109.
- MIMS, C. A., 1956. Rift Valley fever virus in mice. II. Absorption and multiplication of virus. British Journal of Experimental Pathology,37, 110-119.
- MIMS, C. A., 1956. Rift Valley fever virus in mice. III. Further quantitative features of the infective process. British Journal of Experimental Pathology, 37, 120-128.
- MIMS, C. A., 1982. The Pathogenesis of Infectious Diseases. 2nd edn. London: Academic Press.
- MITTEN, J. O., REMMELE, N. S., WALKER, J. S., CARTER, R. C., STEPHEN, E. L. & KLEIN, F., 1970. The clinical aspects of Rift Valley fever virus in household pets. III. Pathological changes in the dog and cat. Journal of Infectious Diseases,121, 25-31.
- MOHAMED, M., MOSHA, F., MGHAMBA, J., ZAKI, S. R., SHIEH, W.-J., PAWESKA, J., OMULO, S., GIKUNDI, S., MMBUJI, P., BLOLAND, P., ZEIDNER, N., KALINGA, R., BREIMAN, R. F. & NJENGA, M. K., 2010. Epidemiologic and clinical aspects of a Rift Valley fever outbreak in humans in Tanzania, 2007. American Jopurnal of Tropical Medicine, 83(2), 22-27.
- MONATH, T. P., 2013. Vaccines against diseases transmitted from animals to humans: a one health paradigm. Vaccine, 31, 5321-5338.
- MONTEIRO, G. E. R., JANSEN VAN VUREN, P., WICHGERS SCHREUR, P. J., ODENDAAL, L., CLIFT, S. J., KORTEKAAS, J. & PAWESKA, J. T., 2018. Mutation of adjacent cysteine residues in the NSs protein of Rift Valley fever virus results in loss of virulence in mice. Virus Research, 249, 31-44.
- MONTGOMERY, R. E., 1912. Nairobi sheep disease. Annual Report, Department of Agriculture, British East Africa, 1910-1911.
- MORRILL, J. C., CARPENTER, L., TAYLOR, D., RAMSBURG, H. H., QUANCE, J. & PETERS, C. J., 1991. Further evaluation of a mutagen-attenuated Rift Valley fever vaccine in sheep. Vaccine,9, 35-41.
- MORRILL, J. C., IKEGAMI, T., YOSHIKAWA-IWATA, N., LOKUGAMAGE, N., WON, S., TERASAKI, K., ZAMOTO-NIIKURA, A., PETERS, C. J. & MAKINO, S., 2010. Rapid accumulation of virulent Rift Valley fever virus in mice from an attenuated virus carrying a single nucleotide substitution in the mRNA. PLoS One, 5, e9986.
- MORRILL, J. C., JENNINGS, G. B., COSGRIFF, T. M., GIBBS, P. H. & PETERS, C. J., 1989. Prevention of Rift Valley fever in rhesus monkeys with interferon-a. Reviews of Infectious Diseases,11, 807-814.
- MORRILL, J. C., JENNINGS, G. B., JOHNSON, A. J., COSGRIFF, T. M., GIBBS, P. H. & PETERS, C. J., 1990. Pathogenesis of Rift Valley fever in rhesus monkeys: role of interferon response. Archives of Virology, 110, 195-212.
- MORRILL, J. C., MEBUS, C. A. & PETERS, C. J., 1997. Safety and efficacy of a mutagen-attenuated Rift Valley fever virus vaccine in cattle. American Journal of Veterinary Research,58, 1104-1109.
- MORRILL, J. C., MEBUS, C. A. & PETERS, C. J., 1997. Safety of a mutagen-attenuated Rift Valley fever virus vaccine in fetal and neonatal bovids.American Journal of Veterinary Research, 58, 1110-1114.
- MORVAN, J., FONTENILLE, D., SALUZZO, J. F. & COULANGES, P., 1991. Possible Rift Valley fever outbreak in man and cattle in Madagascar. Transactions of the Royal Society of Tropical Medicine and Hygiene, 85, 108.
- MORVAN, J., LESBORDES, J. L., ROLLIN, P. E., MOUDEN, J. C. & ROUX, J., 1992. First fatal human case of Rift Valley fever in Madagascar. Transactions of the Royal Society of Tropical Medicine and Hygiene,86, 320.
- MORVAN, J., ROLLIN, P. E., LAVENTURE, S., RAKOTOARIVONY, I. & ROUX, J., 1992. Rift Valley fever epizootic in the central highlands of Madagascar. Research in Virology, 143, 407-415.
- MORVAN, J., ROLLIN, P. E., LAVENTURE, S. & ROUX, J., 1992. Duration of immunoglobulinMantibodies against Rift Valley fever virus in cattle after natural infection. Transactions of the Royal Society of Tropical Medicine and Hygiene, 86, 675.
- MORVAN, J., ROLLIN, P. E. & ROUX, J., 1992. [Sero-epidemiological studies in cattle]. Revue d’Élevage et de Médécine Veterinaire des Pays Tropicaux,45, 121-127.
- MORVAN, J., SALUZZO, J. F., FONTENILLE, D., ROLLIN, P. E. & COULANGES, P., 1991. Rift Valley fever on the east coast of Madagascar. Research in Virology, 142, 475-482.
- MOSQUITO CATALOGUE, http://www.mosquitocatalog.org/. (Accessed 2 January 2019)
- MOUSSA, M. I., WOOD, O. L. & ABDEL-WAHAB, K. S., 1982. Reduced pathogenicity associated with a small plaque variant of the Egyptian strain of Rift Valley fever virus (ZH501). Transactions of the Royal Society of Tropical Medicine and Hygiene,76, 482-486.
- MOUTAILLER, S., KRIDA, G., SCHAFFNER, F., VAZEILLE, M. & FAILLOUX, A. B., 2008. Potential vectors of Rift Valley Fever Virus in the Mediterranean region. Vector-Borne and Zoonotic Diseases, 8, 6.
- MOUTAILLER, S., ROCHE, B., THIBERGE, J. M., CARO, V., ROUGEON, F. & FAILLOUX, A. B., 2011. Host alternation is necessary to maintain the genome stability of rift valley fever virus. PLoS Neglected Tropical Diseases. 5, e1156.
- MOYES, C. L., VONTAS, J., MARTINS, A. J., NG, L. C., KOOU, S. Y., DUSFOUR, I., RAGHAVENDRA, K., PINTO, J., CORBEL, V., DAVID, J. P. & WEETMAN, D., 2017. Contemporary status of insecticide resistance in the major Aedes vectors of arboviruses infecting humans. PLoS Neglected Tropical Diseases, 11, e0005625.
- MROZ, C., GWIDA, M., EL-ASHKER, M., ZIEGLER, U., HOMEIER-BACHMANN, T., EIDEN, M. & GROSCHUP, M. H., 2017. Rift Valley fever virus infections in Egyptian cattle and their prevention. Transboundary Emerging Diseases, 64, 2049-2058.
- MSIMANG, V., THOMPSON, P. N., JANSEN VAN VUREN, P., TEMPIA, S., CORDEL, C., KGALADI, J., KHOSA, J., BURT, F. J., LIANG, J., ROSTAL, M. K., KARESH, W. B. & PAWESKA, J. T., 2019. Rift Valley fever virus exposure amongst farmers, farm workers, and veterinary professionals in central South Africa. Viruses, 11, 140.
- MUHSEN, R. K., 2012. Seroepidemiology of Rift Valley Fever in Basrah. Journal of Veterinary Medical Science, 3, 91-95.
- MULLER, R., SALUZZO, J. F., LOPEZ, N., DREIER, T., TURELL, M., SMITH, J. & BOULOY, M., 1995. Characterization of clone13, a naturally attenuated avirulent isolate of Rift Valley fever virus, which is altered in the small segment. American Journal of Tropical Medicine and Hygiene, 53, 405-411.
- MUNDEL, B. & GEAR, J., 1951. Rift valley fever. I. The occurrence of human cases in Johannesburg. South African Medical Journal,25, 797-800.
- MURAKAMI, S., TERASAKI, K., RAMIREZ, S. I., MORRILL, J. C. & MAKINO, S., 2014. Development of a novel, single-cycle replicable rift valley Fever vaccine. PLoS Neglected Tropical Diseases, 8, e2746.
- MURPHY, F. A., HARRISON, A. K. & WHITFIELD, S. G., 1973. Bunyaviridae: morphological and morphogenetic similarities of Banyawera serological supergroup viruses and several other arthropod-borne viruses.Intervirology, 1, 297-316.
- MWAENGO, D., LORENZO, G., IGLESIAS, J., WARIGIA, M., SANG, R., BISHOP, R. P. & BRUN, A., 2012. Detection and identification of Rift Valley fever virus in mosquito vectors by quantitative real-time PCR. Virus Research, 169, 137-143.
- NABETH, P., KANE, Y., ABDALAHI, M. O., DIALLO, M., NDIAYE, K., BA, K., SCHNEEGANS, F., SALL, A. A. & MATHIOT, C., 2001. Rift Valley fever outbreak, Mauritania, 1998: seroepidemiologic, virologic, entomologic, and zoologic investigations. Emerging Infectious Diseases, 7, 1052-1054.
- NAPP, S., CHEVALIER, V., BUSQUETS, N., CALISTRI, P., CASAL, J., ATTIA, M., ELBASSAL, R., HOSNI, H., FARRAG, H., HASSAN, N., TAWFIK, R., ABD ELKADER, S. & BAYOMY, S., 2018. Understanding the legal trade of cattle and camels and the derived risk of Rift Valley Fever introduction into and transmission within Egypt. PLoS Neglected Tropical Diseases, 12, e0006143.
- NARAYANAN, A., KEHN-HALL, K., SENINA, S., HILL, L., VAN DUYNE, R., GUENDEL, I., DAS, R., BAER, A., BETHEL, L., TURELL, M., HARTMAN, A. L., DAS, B., BAILEY, C. & KASHANCHI, F., 2012. Curcumin inhibits Rift Valley fever replication in human cells. Journal of Biological Chemistry, 40, 33198-33214.
- NDERITU, L., LEE, J. S., OMOLO, J., OMULO, S., O’GUINN, M. L., HIGHTOWER, A., MOSHA, F., MOHAMED, M., MUNYUA, P., NGANGA, Z., HIETT, K., SEAL, B., FEIKIN, D. R., BREIMAN, R. F. & NJENGA, M. K., 2011. Sequential Rift Valley fever outbreaks in Eastern Africa caused by multiple lineages of the virus. Journal of Infectious Diseases, 203, 655-665.
- NEWMAN-GERHARDT, S., MUIRURI, S., MUCHIRI, E., PETERS, C. J., MORRILL, J., LUCAS, A. H., KING, C. H., KAZURA, J. & LABEAUD, A. D., 2013. Potential for autoimmune pathogenesis of Rift Valley Fever virus retinitis. American Journal of Tropical Medicine and Hygiene, 89, 495-497.
- NGUKU, P., SHARIF, S. K., MUTONGA, D., AMWAYI, S., OMOLO, J., MOHAMED, O., FARNON, E. C., GOULD, L. H., LEDERMAN, E., RAO, C., SANG, R., SCHNABEL, D., FEIKIN, D. R., HIGHTOWER, A., NJENGA, M. K. & BREIMAN, R. F., 2010. An investigation of a major outbreak of Rift Valley fever in Kenya: 2006-2007. American Jopurnal of Tropical Medicine, 83, 5-13.
- NICHOLAS, D. E., JACOBSEN, K. H. & WATERS, N. M., 2014. Risk factors associated with human Rift Valley fever infection: systematic review and meta-analysis. Tropical Medicine and International Health, 19, 1420-1429.
- NIKLASSON, B., PETERS, C. J., BENGTSSON, E. & NORRBY, E., 1985. Rift Valley fever virus vaccine trial: study of neutralizing antibody response in humans. Vaccine,3, 123-127.
- NIKLASSON, B., PETERS, C. J., GRANDIEN, M. & WOOD, O., 1984. Detection of human immunoglobulins G andMantibodies to Rift Valley fever virus by enzyme-linked immunosorbent assay. Journal of Clinical Microbiology, 19, 225-229.
- NYAKARAHUKA, L., DE ST. MAURICE, A., PURPURA, L., ERVIN, E., BALINANDI, S., TUMUSIIME, A., KYONDO, J., MULEI, S., TUSIIME, P., LUTWAMA, J., KLENA, J. D., BROWN, S., KNUST, B., ROLLIN, P. E., NICHOL, S. T. & SHOEMAKER, T. R., 2018. Prevalence and Risk Factors of Rift Valley Fever in Humans and Animals from Kabale District in Southwestern Uganda, 2016. PLoS Neglected Tropical Diseases, 12, e0006412.
- O’BRIEN, D., SCUDAMORE, J., CHARLIER, J. & DELAVERGNE, M., 2016. DISCONTOOLS: a database to identify research gaps on vaccines, pharmaceuticals and diagnostics for the control of infectious diseases of animals. Veterinary Research, 13, 1.
- ODENDAAL, L., CLIFT, S. & FOSGATE, G. T., 2018. Lesions and cellular tropism of natural Rift Valley fever in adult sheep. Veterinary Pathology, 1-17.
- ODENDAAL, L., CLIFT, S. J., FOSGATE, G. T. & DAVIS, A. S., 2019. Lesions and cellular tropism of natural Rift Valley fever virus infection in adult sheep. Veterinary Pathology, 56, 61-77.
- ODENDAAL, L., CLIFT, S. J., FOSGATE, G. T. & DAVIS, A. S., 2020a. Ovine fetal and placental lesions and cellular tropism in natural Rift Valley fever virus infections. Veterinary Pathology, 57(6), 791-806.
- ODENDAAL, L., DAVIS, A. S., FOSGATE, G. T. & CLIFT, S. J., 2020b. Lesions and cellular tropism of natural Rift Valley fever virus infection in young lambs. Veterinary Pathology, 57(1), 66-81.
- OIE., 2009. Rift Valley Fever. OIE Technical Disease Cards, OIE Scientific and Technical Department, 1-5.
- OIE., 2018. Rift Valley fever (infection with Rift Valley fever virus). OIE Terrestrial manual 2018. http://www.oie.int/fileadmin/Home/eng/Health_standards/tahm/2.01.18_RVF.pdf.
- OIE., 2019. Terrestrial Animal Health Code. Infection with Rift Valley fever virus. https://www.oie.int/fileadmin/Home/eng/Health_standards/tahc/current/chapitre_rvf.pdf.
- OLALEYE, O. D., TOMORI, O., FAJIMI, J. L. & SCHMITZ, H., 1996. Experimental infection of three Nigerian breeds of sheep with the Zinga strain of the Rift Valley fever virus.Revue d’Élevage et de Médécine Veterinaire des Pays Tropicaux, 49, 6-16.
- OLALEYE, O. D., TOMORI, O. & SCHMITZ, H., 1996. Rift Valley fever in Nigeria: infections in domestic animals. Revue Scientifique et Technique, 15, 937-946.
- OLIVE, M. M., GOODMAN, S. M. & REYNES, J. M., 2012. The role of wild mammals in the maintenance of Rift Valley fever virus. Journal of Wildlife Diseases, 48, 241-266.
- ORESHKOVA, N., CORNELISSEN, L. A. H. M., DE HAAN, C. A. M., MOORMANN, R. J. M. & KORTEKAAS, J. A., 2014. Evaluation of nonspreading Rift Valley fever virus as a vaccine vector using influenza virus hemagglutinin as a model antigen. Vaccine, 32, 5323-5329.
- ORGANIZATION)., W. W. H., 2008. Anthrax in humans and animals. 4th edItion. Turnbull, P. C. B., (ed.). Geneva, Switzerland: World Health Organization, World Organisation for Animal Health. Food and Agriculture Organization of the United Nations.
- OYMANS, J., WICHGERS SCHREUR, P. J., VAN KEULEN, L., KANT, J. & KORTEKAAS, J., 2020. Rift Valley fever virus targets the maternal-foetal interface in ovine and human placentas. PLoS Neglected Tropical Diseases, 14, e0007898.
- PAPATHANOS, P. A., BOURTZIS, K., TRIPET, F., BOSSIN, H., VIRGINIO, J. F., CAPURRO, M. L., PEDROSA, M. C., GUINDO, A., SYLLA, L., COULIBALY, M. B., YAO, F. A., EPOPA, P. S. & DIABATE, A., 2018. A perspective on the need and current status of efficient sex separation methods for mosquito genetic control. Parasit Vectors, 11, 654.
- PATERSON, H. E., BRONSDEN, P., LEVITT, J. & WORTH, C. B., 1964. Some culicine mosquitoes (Diptera: Culicidae) at Ndumu, Republic of South Africa; a study of their host preferences and host range. Medical Proceedings, 10, 188-192.
- PATRICAN, L. A. & BAILEY, C. L., 1989. Ingestion of immune bloodmeals and infection of Aedes fowleri, Aedes mcintoshi and Culex pipienswith Rift Valley fever virus. American Journal of Tropical Medicine and Hygiene, 40, 534-540.
- PATRICAN, L. A., LOGAN, T. M. & BAILEY, C. L., 1988. Influence of Rift Valley fever viral infection on the daily survivorship of Aedes mcintoshiand Aedes fowleri mosquitoes. Abstracts, 37th Annual Meeting of the American Society of Tropical Medicine and Hygiene. Washington DC, December.
- PAWESKA, J. T., 2014. Rift Valley fever. In: Clinical Case Study of Emerging Infectious Diseases. ERGONUL, O., CAN, F., AKOVA, M. & MADOFF, L., (eds), Elsevier, Academic Press, 73-93. ISBN: 978-0-12-416975-3.
- PAWESKA, J. T., 2015. Rift Valley fever. Revue scientifique et technique (International Office of Epizootics), 34, 375-389.
- PAWESKA, J. T., BARNARD, B. J. & WILLIAMS, R., 1995. The use of sucrose-acetone-extracted Rift Valley fever virus antigen derived from cell culture in an indirect enzyme-linked immunosorbent assay and haemagglutination-inhibition test. Onderstepoort Journal of Veterinary Research,62, 227-233.
- PAWESKA, J. T., BURT, F. J., ANTHONY, F., SMITH, S. J., GROBBLEAAR, A. A., CROFT, J. E., KSIAZEK, T. & SWANEPOEL, R., 2003. IgG-sandwich and IgM-capture enzyme-linked immunosorbent assay for detection of antibody to Rift Valley fever in domestic ruminants. Journal of Virological Methods, 113, 103-112.
- PAWESKA, J. T., BURT, F. J. & SWANEPOEL, R., 2005a. Validation of IgG-sandwich and IgM-capture ELISA for the detection of antibody to Rift Valley fever virus in humans. Journal of Virological Methods, 124, 173-181.
- PAWESKA, J. T. & JANSEN VAN VUREN, P., 2013. Rift Valley fever virus: a virus with potential for global emergence. In: The role of Animals in emerging viral diseases, JOHNSON, N., (ed.). Elsevier, Academic Press, 169-200. ISBN: 978-0-12-405191-1.
- PAWESKA, J. T., JANSEN VAN VUREN, P., KEMP, A., BUSS, P., BENGIS, R. G., GAKUYA, F., BREIMAN, R. F., KARIUKI, M. N. & SWANEPOEL, R., 2008. Recombinant nucleocapsid-based ELISA for detection of IgG antibody to Rift Valley fever virus in African buffalo. Veterinary Microbiology, 127, 21-28.
- PAWESKA, J. T., JANSEN VAN VUREN, P., KEMP, A., SWANEPOEL, R., BUSS, P., ROY, G., BENGIS, R. G., GAKUYA, F., BREMEN, R. F. & KARIUKI, N. M., 2010. A recombinant nucleocapsid-based indirect ELISA for serodiagnosis of Rift Valley fever in African wildlife. In: Proceedings of Sustainable Improvement of Animal Production and Health. ODONGO, N. E., GARCIA, M. & VILJOEN, G.J., (eds.). FAO, Rome, 309-312. ISBN 978-92-5-106697-3.
- PAWESKA, J. T., MORTIMER, E., LEMAN, P. A. & SWANEPOEL, R., 2005b. An inhibition enzyme-linked immunosorbent assay for the detection of antibody to Rift Valley fever virus in humans, domestic and wild ruminants. Journal of Virological Methods, 127, 10-18.
- PEPIN, M., BOULOY, M., BIRD, B. H., KEMP, A. & PAWESKA, J., 2010. Rift Valley fever virus(Bunyaviridae: Phlebovirus): an update on pathogenesis, molecular epidemiology, vectors, diagnostics and prevention. Veterinary Research, 41, 61.
- PERREIRA, J. M., CHIN, C. R., FEELEY, E. M. & BRASS, A. L., 2013. IFITMs restrict the replication of multiple pathogenic viruses. Journal of Molecular Biology, 425, 4937-4955.
- PETERS, C. J. & ANDERSON, G. W., 1981. Pathogenesis of Rift Valley fever. Contributions to Epidemiology and Biostatistics, 3, 21-41.
- PETERS, C. J., JONES, D., TROTTER, R., DONALDSON, J., WHITE, J., STEPHEN, S. & SLONE, T. W. J., 1988. Experimental Rift Valley fever in rhesus macaques. Archives of Virology, 99, 31-44.
- PETERS, C. J., LIU, C. T., ANDERSON, G. W., MORRILL, J. C. & JAHRLING, P. B., 1989. Pathogenesis of viral hemorrhagic fevers: Rift Valley fever and Lassa fever contrasted. Reviews of Infectious Diseases,11, 743-749.
- PETERS, C. J. & MEEGAN, J. M., 1981. Rift Valley fever. In:BERAN, G., (ed.). CRC Handbook Series in Zoonoses. Boca Raton, Florida: CRC Press Inc, Sect B1, 403-420.
- PETERS, C. J. & SHELOKOV, A., 1990. Viral hemorrhagic fever. Current Therapy in Infectious Diseases, 3, 355-360.
- PETERS, C. J. & SLONE, T. W., 1982. Inbred rat strains mimic the disparate human response to Rift Valley fever virus infection. Journal of Medical Virology, 10, 45-54.
- PETISCA, J. L. N. & SERRA, J. J. B. L., 1971. Anatomia patóligica de algumas doencas dos animais domésticos. VI Febre do Vale de Rift. Veterinária Moçambicana,4, 69-73.
- PETROVA, V., KRISTIANSEN, P., NORHEIM, G. & YIMER, A. A., 2020. Rift Valley fever: diagnostic challenges and investment need for vaccine development. BMJ Global Health, 5, e002694.
- PINI, A., LUND, L. J. & DAVIES, F. G., 1973. Fluorescent and neutralizing antibody response to infections by Rift Valley fever virus. Journal of the South African Veterinary Association,44, 161-165.
- PINKHAM, C., AHMED, A., BRACCI, N., NARAYANAN, A. & KEHN-HALL, K., 2018. Host-based processes as therapeutic targets for Rift Valley fever virus. Antiviral Research, 160, 64-78.
- PIPER, C. J., SORENSON, D. R. & GERRARD, S. R., 2011. Efficient celluar release of Rift Valley fever virus requires genomic RNA. PLoS One, 6, e18070.
- PITTMAN, P. R., LIU, C. T., CANNON, T. L., MAKUCH, R. S., MANGIAFICO, J. A., GIBBS, P. H. & PETERS, C. J., 1999. Immunogenicity of an inactivated Rift Valley fever vaccine in humans: a 12-year experience. Vaccine, 18, 181-189.
- PITTMAN, P. R., MCCLAIN, D., QUINN, X., COONAN, K. M., MANGIAFICO, J., MAKUCH, R. S., MORRILL, J. & PETERS, C. J., 2016. Safety and immunogenicity of a mutagenized, live attenuated Rift Valley fever vaccine, MP-12, in a Phase 1 dose escalation and route comparison study in humans. Vaccine, 34, 424-429.
- PITTMAN, P. R., NORRIS, S. L., BROWN, E. S., RANADIVE, M. V., SCHIBLY, B. A., BETTINGER, G. E., LOKUGAMAGE, N., KORMAN, L., MORRILL, J. C. & PETERS, C. J., 2016. Rift Valley fever MP-12 vaccine Phase 2 clinical trial: Safety, immunogenicity, and genetic characterization of virus isolates. Vaccine, 34, 523-530.
- POOURRUT, X., NKOGHE, D., SOURIS, M., PAUPY, C., PAWESKA, J., PADILLA, C., MOUSSAVOU, G. & LEROY, R. M., 2010. Rift Valley fever seroprevalence in human rural populations of Gabon. PLoS Neglected Tropical Diseases, 4, e763.
- RAGAN, I. K., DAVIS, A. S., MCVEY, D. S., RICHT, J. A., ROWLAND, R. R. & WILSON, W. C., 2018. Evaluation of Fluorescence Microsphere Immunoassay for Detection of Antibodies to Rift Valley Fever Virus Nucleocapsid Protein and Glycoproteins. Journal of Clinical Microbiology, 56.
- RANDALL, R., BINN, L. M. & HARRISON, V. R., 1963. Rift Valley fever virus vaccine. American Journal of Tropical Medicine and Hygiene,12, 611-615.
- RANDALL, R., BINN, L. M. & HARRISON, V. R., 1964. Immunization against Rift Valley fever virus. Studies on the immunogenicity of lyophilized formalin-inactivated vaccine. Journal of Immunology, 93, 293-299.
- RANDALL, R., GIBBS, C. J. J., AULISIO, C. G., BINN, L. N. & HARRISON, V. R., 1962. The development of a formalin-killed Rift Valley fever virus vaccine for use in man. Journal of Immunology,89, 660-671.
- RIPPY, M. K., TOPPER, M. J., MEBUS, C. A. & MORRILL, J. C., 1992. Rift Valley fever virus-induced encephalomyelitis and hepatitis in calves. Veterinary Pathology,29, 495-502.
- RISSMANN, M., EIDEN, M., EL MAMY, B., ISSELMOU, K., DOUMBIA, B., ZIEGLER, U., HOMEIER-BACHMANN, T., YAHYA, B. & GROSCHUP, M. H., 2017. Serological and genomic evidence of Rift Valley fever virus during inter-epidemic periods in Mauritania. Epidemiology and Infection, 145, 1058-1068.
- RITTER, M., BOULOY, M., VIALAT, P., JANZEN, C., HALLER, O. & FRESE, M., 2000. Resistance to Rift Valley fever virus in Rattus norvegicus:genetic variability within certain ‘inbred’ strains. Journal of General Virology, 81, 2683-2688.
- ROIZ, D., WILSON, A. L., SCOTT, T. W., FONSECA, D. M., JOURDAIN, F., MÜLLER, P., VELAYUDHAN, R. & CORBEL, V., 2018. Integrated Aedes management for the control of Aedes-borne diseases. PLoS Neglected Tropical Diseases, 12, e0006845.
- ROMOSER, W. S., FARAN, M. E. & BAILEY, C. L., 1987. Newly recognized route of arbovirus dissemination from the mosquito (Diptera: Culicidae) midgut. Journal of Medical Entomology, 24, 431-432.
- ROMOSER, W. S., FARAN, M. E., BAILEY, C. L. & LERDTHUSNEE, K., 1992. An immunocytochemical study of the distribution of Rift Valley fever virus in the mosquito Culex pipiens. American Journal of Tropical Medicine and Hygiene,46, 489-501.
- ROSSIGNOL, P. A., RIBEIRO, J. M. C., JUNGERY, M., TURELL, M. J., SPIELMAN, A. & BAILEY, C. L., 1985. Enhanced mosquito blood-finding success on parasitemic hosts: Evidence for vector-parasite mutualism.Proceedings of the National Academy of Sciences of the United States of America, 82, 7725-7727.
- ROSTAL, M. K., LIANG, J. E., ZIMMERMANN, D., BENGIS, R., PAWESKA, J. & KARESH, W. B., 2017. Rift Valley Fever: Does Wildlife Play a Role? ILAR Journal, 58, 359-370.
- SABIN, A. B. & BLUMBERG, R. W., 1947. Human infection with Rift Valley fever virus and immunity twelve years after single attack. Proceedings of the Society for Experimental Biology and Medicine,64, 385-389.
- SALEH AGHAA, O. B. & RHAYMAH, M. S., 2013. Seroprevelance study of Rift Valley fever antibody in sheep and goats in Ninevah governorate. Iraqi. Journal of Veterinary Medical Science, 27, 53-61.
- SALL, A. A., DE A ZANOTTO, P. M., VIALAT, P., SENE, O. K. & BOULOY, M., 1998. Origin of 1997-98 Rift Valley fever outbreak in East Africa. Lancet, 352, 1596-1597.
- SALL, A. A., DE A ZANOTTO, P. M., ZELLER, H. G., DIGOUTTE, J. P., THIONGANE, Y. & BOULOY, M., 1997. Variability of the NS(S) protein among Rift Valley fever virus isolates. Journal of General Virology, 78(Pt 11), 2853-2858.
- SALL, A. A., DE, A. Z. P., ZELLER, H. G., DIGOUTTE, J. P., THIONGANE, Y. & BOULOY, M., 1997. Variability of the NS(S) protein among Rift Valley fever virus isolates.Journal of General Virology, 78, 2853-2858.
- SALL, A. A., MACONDO, E. A., SENE, O. K., DIAGNE, M., SYLLA, R., MONDO, M., GIRAULT, L., MARRAMA, L., SPIEGEL, A., DIALLO, M., BOULOY, M. & MATHIOT, C., 2002. Use of reverse transcriptase PCR in early diagnosis of Rift Valley fever. Clinical and Diagnostic Laboratory Immunology,9, 713-715.
- SALL, A. A., ZANOTTO, P. M., SENE, O. K., ZELLER, H. G., DIGOUTTE, J. P., THIONGANE, Y. & BOULOY, M. J., 1999. Genetic reassortment of Rift Valley fever virus in nature. Virology, 73, 8196-8200.
- SALL, A. A., ZANOTTO, P. M., VIALAT, P., SÈNE, O. K. & BOULOY, M., 1998. Molecular epidemiology and emergence of Rift Valley fever. Memórias do Instituto Oswaldo Cruz, 93, 609-614.
- SALUZZO, J. F., TURELL, M., RAMMARIELLO, R. & SMITH, J., 1988. Genetic reassortment of Rift Valley fever virus strains in cell culture and in the mosquito, Culex pipiens. Abstracts, 37th Annual Meeting of the American Society of Tropical Medicine and Hygiene.Washington DC: December.
- SAMY, A. M., PETERSON, A. T. & HALL, M., 2017. Phylogeography of Rift Valley Fever Virus in Africa and the Arabian Peninsula. PLoS Neglected Tropical Diseases, 11, e0005226.
- SANDERSON, C. E., JORI, F., MOOLLA, N., PAWESKA, J. T., OUMER, N. & ALEXANDER, K. A., 2020. Silent circulation of Rift Valley Fever in humans, Botswana, 2013–2014. Emerging Infectious Diseases, 26 (10), 2453-2456.
- SANDROCK, M., FRESE, M., HALLER, O. & KOCHS, G., 2001. Interferon-induced rat Mx proteins confer resistance to Rift Valley fever virus and other arthropod-borne viruses. Journal of Interferon and Cytokine Research,21, 663-668.
- SCHARTON, D., BAILEY, K. W., VEST, Z., WESTOVER, J. B., KUMAKI, Y., VAN WETTERE, A., FURUTA, Y. & GOWEN, B. B., 2014. Favipiravir (T-705) protects against peracute Rift Valley fever virus infection and reduces delayed-onset neurologic disease observed with ribavirin treatment. Antiviral Research, 104, 84-92.
- SCHMALJOHN, C. S. & NICHOL, S. T., 2007. Bunyaviridae. In: KNIPE, D. M., HOWLEY, P. M., GRIFFIN, D. E., LAMB, R. A., MARTIN, M. A., ROIZMAN, B. & STRAUS, S. E., (eds.). Fields Virology. 5th edition. Lippincott, Williams & Wilkins, Philadelphia, PA, USA, 1741-1789.
- SCHREUR, P. J. Q., VLOET, R. P. M., KANT, J., VAN KEULEN, L., GONZALES, J. L., VISSER, T. M., KOERAADT, C. J. M., VOGELS, C. B. F. & KORTEKAAS, J., 2021. Reproducing the Rift Valley fever virus mosquito‑lamb‑mosquito transmission cycle. Scientific Reports, 11, 1477.
- SCHREUR, P. W., PAWESKA, J. T., KANT, J. & KORTEKAAS, J., 2017. A novel highly sensitive, rapid and safe Rift Valley fever virus neutralization test. Journal of Virological Methods, 248, 26-30.
- SCHRIRE, I., 1951. Macular changes in Rift Valley fever. South African Medical Journal,25, 926-930.
- SCHRIRE, I. & GEAR, J. H. S., 1956. The fevers of Africa. 3. Rift Valley fever.Central African Journal of Medicine, 2, 237-240.
- SCHULZ, K. C. A., 1951. The pathology of Rift Valley fever or enzootic hepatitis in South Africa. Journal of the South African Veterinary Medical Association, 22, 113-120.
- SCHULZ, K. H., 1951. Rift Valley fever in South Africa. Special Report 5/51. Pretoria: Union Department of Health.
- SCHWENTKER, F. F. & RIVERS, T. M., 1934. Rift Valley fever in man: report of a fatal laboratory infection complicated by thrombophlebitis. Journal of Experimental Medicine, 59, 305-313.
- SCOTT, G. R., 1963. Pigs and Rift Valley fever. Nature,200, 919-920.
- SCOTT, G. R., 1981. Fever and death in sheep infected with the viruses of Rift Valley fever and Nairobi sheep disease. Transactions of the Royal Society of Tropical Medicine and Hygiene,75, 329-329.
- SCOTT, G. R., COACKLEY, W., ROACH, R. W. & DOWDY, N. R., 1963. Rift Valley fever in camels. Journal of Pathology and Bacteriology, 86, 229-231.
- SCOTT, G. R. & HEISCH, R. B., 1959. Rift Valley fever and Rift Valley rodents.East African Journal of Medicine, 36, 665-667.
- SCOTT, G. R., WEDDELL, W. & REID, D., 1956. Preliminary findings on the prevalence of Rift Valley fever in Kenya cattle. Bulletin of Epizootic Diseases of Africa, 4, 17-25.
- SCOTT, R. M., FEINSOD, F. M., ALLAM, I. H., KSIAZEK, T. G., PETERS, C. J., BOTROS, B. A. M. & DARWISH, M. A., 1986. Serological tests for detecting Rift Valley fever viral antibodies in sheep from the Nile Delta. Journal of Clinical Microbiology,24, 612-614.
- SCOTT, T., PAWESKA, J. T., ARBUTHNOT, P. & WEINBERG, M. S., 2012. Pathogenic effects of Rift Vally fever virus NSs gene are alleviated in cultured cells by expressed antiviral short hairpin RNAs. Antiviral Therapie, 17, 64356.
- SELLERS, R. F., PEDGLEY, D. E. & TUCKER, M. R., 1982. Rift Valley fever, Egypt —1977: disease spread by wind-borne insect vectors? Veterinary Record,110, 73-77.
- SERVICE, M. W., 1980. A Guide to Medical Entomology. London: Macmillan.
- SERVICE, M. W., 1997. Mosquito (Diptera:Culicidae) dispersal - the long and short of it. Journal of Medical Entomology, 34, 579-588.
- SHERMAN, M. B., FREIBERG, A. N., HOLBROOK, M. R. & WATOWICH, S. J., 2009. Single-particle cryo-electron microscopy of Rift Valley fever virus. Virology, 387, 11-15.
- SHIEH, W.-J., PADDOCK, C. D., LEDERMAN, E., RAO, C. Y., GOULD, L. H., MOHAMED, M., MOSHA, F., MGHAMBA, J., BLOLAND, P., NJENGA, M. K., MUTONGA, D., SAMUEL, A. A., GUARNER, J., BREIMAN, R. F. & ZAKI, S. R., 2010. Pathologic studies on suspect animal and human cases of Rift Valley fever from an outbreak in Eastern Africa, 2006-2007. American Jopurnal of Tropical Medicine, 83, 38-42.
- SHIMSHONY, A. & BARZILAI, R., 1983. Rift Valley fever. Advances in Veterinary Science and Comparative Medicine,27, 347-425.
- SHOEMAKER, T. R., BOULIANNE, C., VINCENT, M. J., PEZZANITE, L., AL-QAHTANI, M. M., AL-MAZROU, Y., KHAN, A. S., ROLLIN, P. E., SWANEPOEL, R., KSIAZEK, T. G. & NICHOL, S. T., 2002. Genetic analysis of viruses associated with emergence of Rift Valley fever in Saudi Arabia and Yemen, 2000–01. Emerging Infectious Diseases, 8, 1415-1420.
- SHOEMAKER, T. R., NYAKARAHUKA, L., BALINANDI, S., OJWANG, J., TUMUSIIME, A., MULEI, S., KYONDO, J., LUBWAMA, B., SEKAMATTE, M., NAMUTEBI, A., TUSIIME, P., MONJE, F., MAYANJA, M., SSENDAGIRE, S., DAHLKE, M., KYAZZE, S., WETAKA, M., MAKUMBI, I., BORCHERT, J., ZUFAN, S., PATEL, K., WHITMER, S., BROWN, S., DAVIS, W. G., KLENA, J. D., NICHOL, S. T., ROLLIN, P. E. & LUTWAMA, J. J., 2019. First Laboratory-Confirmed Outbreak of Human and Animal Rift Valley Fever Virus in Uganda in 48 Years. American Journal of Tropical Medicine and Hygiene, 100, 659-671.
- SHONE, D. K., 1958. Rift Valley fever in Southern Rhodesia. Central African Journal of Medicine,4, 284-286.
- SHONE, D. K., R., P. J., M., R. R. & CHRISTIE, G. J., 1958. Some aetiological agents of bovine abortions in Southern Rhodesia. Journal of the South African Veterinary Medical Association, 29, 55-62.
- SHOPE, R. E., MEEGAN, J. M., PETERS, C. J., TESH, R. B. & TRAVASSOS DA ROSA, A. A., 1981. Immunologic status of Rift Valley fever virus. Contributions to Epidemiology and Biostatistics, 3, 42-52.
- SHOPE, R. E., PETERS, C. J. & WALKER, J. S., 1980. Serological relation between Rift Valley fever virus and viruses of phlebotomus fever serogroup. Lancet, 1, 886-887.
- SHOPE, R. E. & SATHER, G. E., 1979. Arboviruses. In: LENNETTE, E. H. & SCHMIDT, N. J., (eds.). Diagnostic Procedures for Viral, Rickettsial and Chlamydial Infections. 5th edn. Washington D.C.: Public Health Association.
- SIAM, A. L. & MEEGAN, J. M., 1980. Ocular disease resulting from infection with Rift Valley fever virus. Transactions of the Royal Society of Tropical Medicine and Hygiene,74, 539-541.
- SIAM, A. L., MEEGAN, J. M. & GHARBAWI, K. F., 1980. Rift Valley fever ocular manifestation: observations during the 1977 epidemic in the Arab Republic of Egypt.British Journal of Ophthalmology, 64, 366-374.
- SIDWELL, R. W., HUFFMAN, J. H., BARNARD, D. L., SMEE, D. F., WARREN, R. P., CHIRIGOS, M. A., KENDE, M. & HUGGINS, J., 1994. Antiviral and immunomodulating inhibitors of experimentally-induced Punta Toro virus infections.Antiviral Research, 25, 105-122.
- SIDWELL, R. W., HUFFMAN, J. H., SMEE, D. F., GILBERT, J., GESSAMAN, A., PEASE, A., P., W. R., J., H. & KENDE, M., 1992. Potential role of immunomodulators for treatment of phlebovirus infections of animals. Annals of the New York Academy of Sciences, 653, 344-355.
- SINDATO, C., KARIMURIBO, E. D., PFEIFFER, D. U., KIVARIA, F., DAUTU, G., MBOERA, L. E. G. & PAWESKA, J. T., 2014. Spatial and temporal pattern of Rift Valley fever outbreaks in Tanzania 1930 to 2007. PLoS One, 9(2), e88897.
- SMITH, D. R., JOHNSTON, S. C., PIPER, A., BOTTO, M., DONNELLY, G., SHAMBLIN, J., ALBARIÑO, C. G., HENSLEY, L. E., SCHMALJOHN, C., NICHOL, S. T. & BIRD, B. H., 2018. Attenuation and efficacy of live-attenuated Rift Valley fever virus vaccine candidates in non-human primates. PLoS Neglected Tropical Diseases, 12, e0006474.
- SMITH, E. R. & PARKER, D. M., 2010. Tick communities at the expanding wildlife/cattle interface in the Eastern Cape Province, South Africa: implications for Corridor disease. Journal of the South African Veterinary Association, 81, 237-240.
- SMITHBURN, K. C., 1949. Rift Valley fever; the neurotropic adaptation of the virus and the experimental use of this modified virus as a vaccine. British Journal of Experimental Pathology, 30, 1-16.
- SMITHBURN, K. C., MAHAFFY, A. F., HADDOW, A. J., KITCHEN, S. F. & SMITH, J. F., 1949. Rift Valley fever: accidental infections among laboratory workers. Journal of Immunology, 62, 213-227.
- SOI, R. K., RURANGIRWA, F. R., MCGUIRE, T. C., RWAMBO, P. M., DEMARTINI, J. C., B., T. & CRAWFORD, T. B., 2010. Protection of Sheep against Rift Valley Fever Virus and Sheep Poxvirus with a Recombinant Capripoxvirus Vaccine. Clinical and Vaccine Immunology, 17 (12), 1842-1849.
- SOLIMAN, A. K., WATTS, D. M., SALIB, A. W., SHEHATA, A. E., ARTHUR, R. R. & BOTROS, B. A., 1997. Application of an immunoperoxidase monolayer assay for the detection of arboviral antibodies. Journal of Virological Methods,65, 147-151.
- SOTI, V., TRAN, A., DEGENNE, P., CHEVALIER, V., LO SEEN, D., THIONGANE, Y., DIALLO, M., GUÉGAN, J. & FONTENILLE, D., 2012. Combining Hydrology and Mosquito Population Models to Identify the Drivers of Rift Valley Fever Emergence in Semi-Arid Regions of West Africa. PLoS Neglected Tropical Diseases, 6(8), e1795. doi:10.1371/journal.pntd.0001795.
- SOUMARÉ, P. O. L., FREIRE, C. C. M., FAYE, O., DIALLO, M., DE OLIVEIRA, J. V. C., ZANOTTO, P. M. A. & SALL, A. A., 2012. Phylogeography of Rift Valley Fever virus in Africa reveals multiple introductions in Senegal and Mauritania. PLoS One, 7, e35216.
- SOW, A., FAYE, O., BA, Y., DIALLO, D., FALL, G., FAYE, O., BOB, N. S., LOUCOUBAR, C., RICHARD, V., DIA, A. T., DIALLO, M., MALVY, D. & SALL, A. A., 2016. Widespread Rift Valley Fever Emergence in Senegal in 2013-2014. Open Forum Infectious Diseases, 3, ofw149.
- SOW, A., FAYE, O., FAYE, O., DIALLO, D., SADIO, B. D., WEAVER, S. C., DIALLO, M. & SALL, A. A., 2014. Rift Valley fever in Kedougou, southeastern Senegal, 2012. Emerging Infectious Diseases, 20, 504-506.
- STORDY, R. J., 1913. Mortality among lambs. Annual Report, Department of Agriculture, British East Africa, 1912–1913.
- STRUTHERS, J. K. & SWANEPOEL, R., 1982. Identification of a major nonstructural protein in the nuclei of Rift Valley fever virus-infected cells. Journal of General Virology, 60, 381-384.
- STRUTHERS, J. K., SWANEPOEL, R. & SHEPHERD, S. P., 1984. Protein synthesis in Rift Valley fever virus-infected cells. Virology, 134, 118-124.
- SUMAYE, R. D., ABATIH, E. N., THIRY, E., AMURI, M., BERKVENS, D. & GEUBBELS, E., 2015. Inter-epidemic acquisition of Rift Valley fever virus in humans in Tanzania. PLoS Neglected Tropical Diseases, 9, e0003536.
- SUMAYE, R. D., GEUBBELS, E., MBEYELA, E. & BERKVENS, D., 2013. Inter-epidemic transmission of Rift Valley fever in livestock in the Kilombero River Valley, Tanzania: a cross-sectional survey. PLoS Neglected Tropical Diseases, 7, e2356.
- SWAI, E. S. & SINDATO, C., 2015. Seroprevalence of Rift Valley fever virus infection in camels (dromedaries) in northern Tanzania. Tropical Animal Health and Production, 47, 347-352.
- SWANEPOEL, R., 1976. Studies on the epidemiology of Rift Valley fever. Journal of the South African Veterinary Medical Association, 47, 93-94.
- SWANEPOEL, R., 1981. Observations on Rift Valley fever in Zimbabwe. Contributions to Epidemiology and Biostatistics,3: Rift Valley fever, 83-91.
- SWANEPOEL, R., 1981-2002. National Institute for Virology, Private Bag X4, Sandringham 2131, Johannesburg, South Africa. Unpublished observations.
- SWANEPOEL, R., 1990. National Institute for Virology, Sandringham 2131. Unpublished observations, 1969-1990.
- SWANEPOEL, R. & BLACKBURN, N. K., 1977. Demonstration of nuclear immunofluorescence in Rift Valley fever infected cells. Journal of General Virology,34, 557-561.
- SWANEPOEL, R., BLACKBURN, N. K., EFSTRATIOU, S. & CONDY, J. B., 1978. Studies of Rift Valley fever in some African murids (Rodentia: Muridae). Journal of Hygiene, Cambridge, 70, 183-196.
- SWANEPOEL, R., K., B. N., P., L. K., B., V. D. & LEWIS, A. R., 1975. An investigation of infectious infertility and abortion of cattle. Rhodesian Veterinary Journal, 6, 42-55.
- SWANEPOEL, R., MANNING, B. & WATT, J. A., 1979. Fatal Rift Valley fever affecting humans in Southern Africa: a clinicopathological study. Central African Journal of Medicine,25, 1-8.
- SWANEPOEL, R. & PAWESKA, J. T., 2011. Rift Valley fever. In: Oxford Textbook of Zoonoses: Biology, Clinical Practise, and Public Health Control, 2nd edition. PALMER, S. R., SOULSBY, L., TORGERSON, P. R. & BROWN, D. W. G., (eds.). Oxford: Oxford University Press, 421-431.
- SWANEPOEL, R., STRUTHERS, J. K., ERASMUS, M. J., SHEPHERD, S. P. & MCGILLIVRAY, G. M., 1986. Comparison of techniques for demonstrating antibodies to Rift Valley fever virus. Journal of Hygiene, Cambridge, 97, 317-329.
- SWANEPOEL, R., STRUTHERS, J. K., ERASMUS, M. J., SHEPHERD, S. P., MCGILLIVRAY, G. M., SHEPHERD, A. J., HUMMITZSCH, D. E., ERASMUS, B. J. & BARNARD, B. J., 1986. Comparative pathogenicity and antigenic cross-reactivity of Rift Valley fever and other African phleboviruses in sheep. Journal of Hygiene, Cambridge, 97, 331-346.
- TAYLOR, P. & SWANEPOEL, R., 1980. Observations on mosquitoes, midges and rodents during the 1978 Rift Valley fever epizootic in Zimbabwe. Zimbabwe Veterinary Journal,11, 44-49.
- TERASAKI, K., RAMIREZ, S. I. & MAKINO, S., 2016. Mechanistic Insight into the Host Transcription Inhibition Function of Rift Valley Fever Virus NSs and Its Importance in Virulence. PLoS Neglected Tropical Diseases, 10, e0005047.
- TESH, R. B. & DUBOISE, S. M., 1987. Viremia and immune response with sequential phlebovirus infections. American Journal of Tropical Medicine and Hygiene,36, 662-668.
- TESH, R. B., PETERS, C. J. & MEEGAN, J. M., 1982. Studies on the antigenic relationship among phleboviruses.American Journal of Tropical Medicine and Hygiene, 31, 149-155.
- THIONGANE, Y., GONZALEZ, J. P., FATI, A. & AKAKPO, J. A., 1991. Changes in Rift Valley fever neutralizing antibody prevalence among small domestic ruminants following the 1987 outbreak in the Senegal River basin. Research in Virology,142, 67-70.
- THIONGANE, Y., ZELLER, H., LO, M. M., FATI, N. A., AKAKPO, J. A. & GONZALEZ, J. P., 1994. [Decrease of natural immunity against Rift Valley fever in domestic ruminants of the Senegal River basin after the epizootic outbreak of 1987]. Bulletin de la Societe de Pathologie Exotique, 87, 5-6.
- THONNON, J., PICQUET, M., THIONGANE, Y., LO, M., SYLLA, R. & VERCRUYSSE, J., 1999. Rift valley fever surveillance in the lower Senegal river basin: Update 10 years after the epidemic. Tropical Medicine and International Health, 4, 580-585.
- TOMORI, O., 1979. Clinical, virological and serological response of the West African dwarf sheep to experimental infection with different strains of Rift Valley fever virus.Research in Veterinary Science, 26, 152-159.
- TRAN, A., FALL, A. G., BITEYE, B., CISS, M., GIMONNEAU, G., CASTETS, M., SECK, M. T. & CHEVALIER, V., 2019. Spatial Modeling of Mosquito Vectors for Rift Valley Fever Virus in Northern Senegal: Integrating Satellite-Derived Meteorological Estimates in Population Dynamics Models. Remote Sensing, 11, 1024. https://doi.org/10.3390/rs11091024.
- TRAORE-LAMIZANA, M., FONTENILLE, D., DIALLO, M., BA, Y., ZELLER, H. G., MONDO, M., ADAM, F., THONON, J. & MAÏGA, A., 2001. Arbovirus surveillance from 1990 to 1995 in the Barkedji area (Ferlo) of Senegal, a possible natural focus of Rift Valley fever virus. Journal of Medical Entomology, 38, 480-492.
- TURELL, M. J., 1988. Effect of the developmental stage at infection on the ability of Anopheles albimanusto transmit Rift Valley fever virus. Abstracts, 37th Annual Meeting of the American Society of Tropical Medicine and Hygiene, Washington DC, December.
- TURELL, M. J., 1992. Virus-dependent mortality in Rift Valley fever, eastern equine encephalomyelitis, and chikungunya virus-inoculated mosquito (Diptera: Culicidae) larvae. Journal of Medical Entomology,29, 792-795.
- TURELL, M. J., 1993. Effect of environmental temperature on the vector competence of Aedes taeniorhynchusfor Rift Valley fever and Venezuelan equine encephalitis viruses. American Journal of Tropical Medicine and Hygiene, 49, 672-676.
- TURELL, M. J. & BAILEY, C. L., 1987. Transmission studies in mosquitoes (Diptera: Culicidae) with disseminated Rift Valley fever virus infections. Journal of Medical Entomology, 24, 11-18.
- TURELL, M. J., BAILEY, C. L. & BEAMAN, J. R., 1988. Vector Competence of a Houston, Texas Strain of Aedes Albopictus for Rift Valley Fever Virus. Journal of the American Mosquito Control Association, 4, 94-96.
- TURELL, M. J., BAILEY, C. L. & BEAMAN, J. R., 1988. Vector competence of a Houston, Texas strain of Aedes albopictusfor Rift Valley fever virus. Journal of the American Mosquito Control Association, 4, 94-98.
- TURELL, M. J., BAILEY, C. L. & ROSSI, C. A., 1984. Increased mosquito feeding on Rift Valley fever virus-infected lambs. American Journal of Tropical Medicine and Hygiene,33, 1232-1238.
- TURELL, M. J., DOHM, D. J., MORES, C. N., TERRACINA, L., WALLETTE, D. L. J., HRIBAR, L. J., PECOR, J. E. & BLOW, J. A., 2008. Potential for North American mosquitoes to transmit Rift Valley fever virus. Journal of the American Mosquito Control Association, 24, 502-507.
- TURELL, M. J., FARAN, M. E., CORNET, M. & BAILEY, C. L., 1988. Vector competence of Senegalese Aedes fowleri(Diptera: Culicidae) for Rift Valley fever virus. Journal of Medical Entomology, 25, 262-266.
- TURELL, M. J., GARGAN, I. & BAILEY, C. L., 1984. Replication and dissemination of Rift Valley fever virus in Culex pipiens. American Journal of Tropical Medicine and Hygiene,33, 176-181.
- TURELL, M. J., GARGAN, T. P. & BAILEY, C. L., 1985. Culex pipiens (Diptera: Culicidae) morbidity and mortality associated with Rift Valley fever viral infection. Journal of Medical Entomology, 22, 332-337.
- TURELL, M. J. & KAY, B. H., 1998. Susceptibility of selected strains of Australian mosquitoes (Diptera: Culicidae) to Rift Valley fever virus. Journal of Medical Entomology,35, 132-135.
- TURELL, M. J., LINTHICUM, K. J. & BEAMAN, J. R., 1990. Transmission of Rift Valley fever virus by adult mosquitoes after ingestion of virus as larvae. American Journal of Tropical Medicine and Hygiene, 43, 677-680.
- TURELL, M. J. & PERKINS, P. V., 1990. Transmission of Rift Valley fever virus by the sand fly,Phlebotomus duboscqi (Diptera: Psychodidae). American Journal of Tropical Medicine and Hygiene, 42, 185-188.
- TURELL, M. J., PRESLEY, S. M., GAD, A. M., COPE, S. E., DOHM, D. J., MORRILL, J. C. & ARTHUR, R. R., 1996. Vector competence of Egyptian mosquitoes for Rift Valley fever virus. American Journal of Tropical Medicine and Hygiene, 54, 136-139.
- TURELL, M. J. & ROSSI, C. A., 1991. Potential for mosquito transmission of attenuated strains of Rift Valley fever virus. American Journal of Tropical Medicine and Hygiene,44, 278-282.
- TURELL, M. J., ROSSI, C. A. & BAILEY, C. L., 1985. Effect of extrinsic incubation temperature on the ability of Aedes taeniorhynchusand Culex pipiens to transmit Rift Valley fever virus. American Journal of Tropical Medicine and Hygiene, 34, 1211-1218.
- TURELL, M. J., SALUZZO, J. F., TAMMARIELLO, R. F. & SMITH, J. F., 1990. Generation and transmission of Rift Valley fever viral reassortants by the mosquito Culex pipiens. Journal of General Virology, 71, 2307-2312.
- TURELL, M. J. & SPIELMAN, A., 1992. Nonvascular delivery of Rift Valley fever virus by infected mosquitoes. American Journal of Tropical Medicine and Hygiene,47, 190-194.
- UPRETI, D., CERNICCHIARO, N., RICHT, J. A., WILSON, W. C., CLAVIJO, A. & DAVIS, A. S., 2018. Preliminary evaluation of diagnostic accuracy and precision of a competitive ELISA for detection of antibodies to Rift Valley fever virus in cattle and sheep sera. Journal of Virological Methods, 262, 6-11.
- VALADAO, F. G., 1969. Nota prévia sobre a ocorréncia de uma nova doenca em Mocambique-a febre do Vale de Rift. Veterinária Moçambicana,2, 13-20.
- VAN DER BERGH, C., VENTER, E. H., SWANEPEOL, R. & THOMSON, P. N., 2019. High seroconversion rate to Rift Valley fever virus in cattle and goats in far northern KwaZulu-Natal, South Africa, in the absence of reported outbreaks. PLoS Neglected Tropical Diseases, 13(5), e0007296.
- VAN DER BERGH, C., VENTER, E. H., SWANEPOEL, R., HANEKOM, C. C. & THOMSON, P. N., 2020. Neutralizing antibodies against Rift Valley fever virus in wild antelope in far northern KwaZulu-Natal, South Africa, indicate recent virus circulation. Transboundary Emerging Diseases, 67, 1356-1363.
- VAN DER LINDE, N. T., 1953. A recent epidemic of Rift Valley fever in the Orange Free State. Journal of the South African Veterinary Medical Association, 24, 145-148.
- VAN DER LUGT, J. J., COETZER, J. A. & SMIT, M. M., 1996. Distribution of viral antigen in tissues of new-born lambs infected with Rift Valley fever virus. Onderstepoort Journal of Veterinary Research,63, 341-347.
- VAN DER WAL, F. J., ACHTERBERG, R. P., DE BOER, S. M., BOSHRA, H., BRUN, A., MAASSEN, C. B. M. & KORTEKAAS, J., 2012. Bead-based suspension array for simultaneous detection of antibodies against the Rift Valley fever virus nucleocapsid and Gn glycoprotein. Journal of Virological Methods, 183, 99-105.
- VAN EEDEN, C., SWANEPOEL, R. & VENTER, M., 2014. Antibodies against West Nile and Shuni Viruses in Veterinarians, South Africa. Emerging Infectious Diseases, 20, 1409-1411. doi:10.3201/eid2008.131724.
- VAN VELDEN, D. J. J., MEYER, J. D. & OLIVIER, J. A., 1977. Rift Valley fever affecting humans in South Africa. A clinicopathological study. South African Medical Journal, 51, 867-871.
- VAUGHAN, J. A. & TURELL, M. J., 1996. Facilitation of Rift Valley fever virus transmission by Plasmodium bergheisporozoites in Anopheles stephensi mosquitoes. American Journal of Tropical Medicine and Hygiene, 55, 407-409.
- VENTER, M., 2018. Assessing the zoonotic potential of arboviruses of African origin. Current Opinion in Virology, 26, 74-84.
- VIALAT, P., BILLECOCQ, A., KOHL, A. & BOULOY, M., 2000. The S segment of rift valley fever phlebovirus (Bunyaviridae) carries determinants for attenuation and virulence in mice. Journal of Virology, 74, 1538-1543.
- VIALAT, P., MULLER, R., VU, T. H., PREHAUD, C. & BOULOY, M., 1997. Mapping of the mutations present in the genome of the Rift Valley fever virus attenuated MP12 strain and their putative role in attenuation.Virus Research, 52, 43-50.
- VON TEICHMAN, B., ENGELBRECHT, A., ZULU, G., DUNGU, B., PARDINI, A. & BOULOY, M., 2011. Safety and efficacy of Rift Valley fever Smithburn and Clone 13 vaccines in calves. Vaccine, 29, 5771-5777.
- WALKER, J. S., REMMELE, N. S., CARTER, R. C., MITTEN, J. Q., SCHUH, L. G., STEPHEN, E. L. & KLEIN, F., 1970. The clinical aspects of Rift Valley fever virus in household pets. I. Susceptibility of the dog.Journal of Infectious Diseases, 121, 9-18.
- WALKER, J. S., STEPHEN, E. L., REMMELE, N. S., CARTER, R. C., MITTEN, J. Q., SCHUH, L. G. & KLEIN, F., 1970. The clinical aspects of Rift Valley fever in household pets. II. Susceptibility of the cat. Journal of Infectious Diseases, 121, 19-24.
- WALLACE, D. B., ELLIS, C. E., ESPACH, A., SMITH, S. J., GREYLING, R. R. & VILJOEN, G. J., 2006. Protective immune responses induced by different recombinant vaccine regimes to Rift Valley fever. Vaccine, 24, 7181-7189.
- WALSH, M. G., DE WILLEM, S. A. & MOR, S. M., 2017. Wetlands wild Bovidae species richness and sheep density delineate risk of Rift Valley fever outbreaks in the African continent and Arabian Peninsula. PLoS Neglected Tropical Diseases, 11, e0005756.
- WALTER, C. T. & BARR, J. N., 2011. Recent advances in the molecular and cellular biology of bunyaviruses. Journal of General Virology, 92, 2467-2484.
- WALTERS, A. W., KUJAWA, M. R., ALBE, J. R., REED, D. S., KLIMSTRA, W. B. & HARTMAN, A. L., 2019. Vascular permeability in the brain is a late pathogenic event during Rift Valley fever virus encephalitis in rats. Virology, 526, 173-179.
- WEAVER, S. C. & REISEN, W. K., 2010. Present and future arboviral threats. Antiviral Research, 85, 328-345.
- WEINBREN, M. P. & MASON, P. J., 1957. Rift Valley fever in wild field rat (Arvicanthis abyssinicus): A possible natural host. South African Medical Journal,31, 427-430.
- WEINBREN, M. P., WILLIAMS, M. C. & HADDOW, A. J., 1957. A variant of Rift Valley fever virus. South African Medical Journal,31, 951-957.
- WEINGARTL, H. M., MILLER, M., NFON, C. & WILSON, W. C., 2014. Development of a Rift Valley fever virus viremia challenge model in sheep and goats. Vaccine, 32, 2337-2344.
- WEISS, K. E., 1957. Rift Valley fever—a review. Bulletin of Epizootic Diseases of Africa, 5, 431-458.
- WEISS, K. E., 1962. Studies on Rift Valley fever—passive and active immunity in lambs. Onderstepoort Journal of Veterinary Research,29, 3-9.
- WEISS, K. E., 1978. Onderstepoort Veterinary Institute, Onderstepoort 0110, South Africa. Personal communication.
- WERNIKE, K., HOFFMANN, B. & BEER, M., 2015. Simultaneous detection of five notifiable viral diseases of cattle by single-tube multiplex real-time RT-PCR. Journal of Virological Methods, 217, 28-35.
- WICHGERS SCHREUR, P. J., KANT, J., VAN KEULEN, L., MOORMANN, R. J. M. & KORTEKAAS, J., 2015. Four-segmented Rift Valley fever virus induces sterile immunity in sheep after a single vaccination. Vaccine, 33, 1459-1464.
- WICHGERS SCHREUR, P. J., ORESHKOVA, N., HARDERS, F., BOSSERS, A., MOORMANN, R. J. M. & KORTEKAAS, J., 2014. Paramyxovirus-based production of Rift Valley fever virus replicon particles. Journal of General Virology, 95, 2638-2648.
- WICHGERS SCHREUR, P. J., ORESHKOVA, N., MOORMANN, R. J. M. & KORTEKAAS, J., 2014. Creation of Rift Valley fever viruses with four-segmented genomes reveals flexibility in bunyavirus genome packaging. Journal of Virology, 88, 10883-10893.
- WICHGERS SCHREUR, P. J., VAN KEULEN, L., KANT, J. & KORTEKAAS, J., 2017. Four-segmented Rift Valley fever virus-based vaccines can be applied safely in ewes during pregnancy. Vaccine, 35, 3123-3128.
- WILLIAMS, R., MALHERBE, J., WEEPENER, H., MAJIWA, P. & SWANEPOEL, R., 2016. Anomalous High Rainfall and Soil Saturation as Combined Risk Indicator of Rift Valley Fever Outbreaks, South Africa, 2008-2011. Emerging Infectious Diseases, 22, 2054-2062.
- WILSON, M. L., CHAPMAN, L. E., HALL, D. B., DYKSTRA, E. A., BA, K., ZELLER, H. G., TRAORE-LAMIZANA, M., HERVY, J. P., LINTHICUM, K. J. & PETERS, C. J., 1994. Rift Valley fever in rural Northern Senegal: human risk factors and potential vectors. American Journal of Tropical Medicine and Hygiene, 50, 663-675.
- WILSON, W. C., DAVIS, A. S., GAUDREAULT, N. N., FABURAY, B., TRUJILLO, J. D., SHIVANNA, V., SUNWOO, S. Y., BALOGH, A., ENDALEW, A., MA, W., DROLET, B. S., RUDER, M. G., MOROZOV, I., MCVEY, D. S. & RICHT, J. A., 2016. Experimental Infection of Calves by Two Genetically Distinct Strains of Rift Valley Fever Virus. Viruses, 8, e145.
- WILSON, W. C., KIM, I. J., TRUJILLO, J. D., SUNWOO, S. Y., NORONHA, L. E., URBANIAK, K., MCVEY, D. S., DROLET, B. S., MOROZOV, I., FABURAY, B., SCHIRTZINGER, E. E., KOOPMAN, T., INDRAN, S. V., BALARAMAN, V. & RICHT, J. A., 2018. Susceptibility of White-Tailed Deer to Rift Valley Fever Virus. Emerging Infectious Diseases, 24, 1717-1719.
- WILSON, W. C., ROMITO, M., JASPERSON, D. C., WEINGARTL, H., BINEPAL, Y. S., MALULEKE, M. R., WALLACE, D. B., VAN VUREN, P. J. & PAWESKA, J. T., 2013. Development of a Rift Valley fever real-time RT-PCR assay that can detect all three genome segments. Journal of Virological Methods, 193, 426-431.
- WON, S., IKEGAMI, T., PETERS, C. J. & MAKINO, S., 2006. NSm and 78-kilodalton proteins of Rift Valley fever virus are nonessential for viral replication in cell culture. Journal of Virology, 80, 8274-8278.
- WON, S., IKEGAMI, T., PETERS, C. J. & S., M., 2007. NSm protein of Rift Valley fever virus suppresses virus-induced apoptosis. Journal of Virology, 81, 13335-13345.
- WONDERLICH, E. R., CAROLINE, A. L., MCMILLEN, C. M., WALTERS, A. W., REED, D. S., BARRATT-BOYES, S. M. & HARTMAN, A. L., 2018. Peripheral Blood Biomarkers of Disease Outcome in a Monkey Model of Rift Valley Fever Encephalitis. Journal of Virology, 92.
- WOODALL, J. P. & WILLIAMS, M. C., 1960. Work with rodents. Annual Report, East African Virus Research Institute, Entebbe.
- WOODS, C. W., KARPATI, A. M., GREIN, T., MCCARTHY, N., GATURUKU, P., MUCHIRI, E., DUNSTER, L., HENDERSON, A., KHAN, A. S., SWANEPOEL, R., BONMARIN, I., MARTIN, L., MANN, P., SMOAK, B. L., RYAN, M., KSIAZEK, T. G., ARTHUR, R. R., NDIKUYEZE, A., AGATA, N. N., PETERS, C. J. & FORCE, W. H. O. H. F. T., 2002. An outbreak of Rift Valley fever in Northeastern Kenya, 1997-98. Emerging Infectious Diseases, 8, 138-144.
- WORTH, C. B., PATERSON, H. E. & DE MEILLON, B., 1961. The incidence of arthropod-borne viruses in a population of culicine mosquitoes in Tongaland, Union of South Africa (January, 1956, through April, 1960). American Journal of Tropical Medicine and Hygiene, 10, 583-592.
- XU, F., LIU, D., NUNES, M. R. T., DA ROSA, A. P. A. T., TESH, R. B. & XIAO, S.-Y., 2007. Antigenic and genetic relationships among Rift Valley fever virus and other selected members of the genus Phlebovirus (Bunyaviridae). American Journal of Tropical Medicine and Hygiene, 76, 1194-1200.
- YADANI, F. Z., KOHL, A., PREHAUD, C., BILLECOCQ, A. & BOULOY, M., 1999. The carboxy-terminal acidic domain of Rift Valley Fever virus NSs protein is essential for the formation of filamentous structures but not for the nuclear localization of the protein. Journal of Virology, 73, 5018-5025.
- YEDLOUTSCHNIG, R. J., DARDIRI, A. H., MEBUS, C. A. & WALKER, J. S., 1981. Abortion in vaccinated sheep and cattle after challenge with Rift Valley fever virus. Veterinary Record,109, 383-384.
- YEDLOUTSCHNIG, R. J., DARDIRI, A. H. & WALKER, J. S., 1981. Persistence of Rift Valley fever virus in the spleen, liver and brain of sheep after experimental infection. Contributions to Epidemiology and Biostatics,3, 53-59.
- YEDLOUTSCHNIG, R. J., DARDIRI, A. H. & WALKER, J. S., 1981. The response of ponies to inoculation with Rift Valley fever virus. Contributions to Epidemiology and Biostatistics,3, 68-71.
- YILMAZ, A., YILMAZ, H., FABURAY, B., KARAKULLUKCU, A., KASAPCOPUR, A., BARUT, K., CIZMECIGIL, U. Y., AYDIN, O., TEKELIOGLU, B. K., KASAPCOPUR, O., OZKUL, A. A., LABEAUD, D., KOCAZEYBEK, B., RICHT, J. A. & TURAN, N., 2017. Presence of antibodies to Rift Valley fever virus in children, cattle and sheep in Turkey. Journal of Virology and Antiviral Research, 6, 29.
- YOUSSEF, B. Z., 2009. The potential role of pigs in the enzootic cycle of rift valley Fever at alexandria governorate, egyp. Egyptian Public Health Association, 84, 331-344.
- ZABRANSKY, R. J., 2005. Is Rift Valley fever a threat in the United States? Clinical Microbiology Newsletter, 27,41.
- ZAKI, A., COUDRIER, D., YOUSEF, A. I., FAKEEH, M., BOULOY, M. & BILECOCQ, A. D., 2006. Production of monoclonal antibodies against Rift Valley fever virus: application for rapid diagnosis tests (virus detection and ELISA) in human sera. Journal of Virological Methods, 131(1), 34-40.
- ZELLER, H. G., AKAKPO, A. J. & BA, M. M., 1995. Rift Valley fever epizootic in small ruminants in southern Mauritania (October 1993): risk of extensive outbreaks. Annales de la Societe Belge de Medecine Tropicale, 75, 135-140.
- ZELLER, H. G., FONTENILLE, D., TRAORE-LAMIZANA, M., THIONGANE, Y. & DIGOUTTE, J. P., 1997. Enzootic activity of Rift Valley fever virus in Senegal. American Journal of Tropical Medicine and Hygiene, 56(3), 265-272.
- ZINZULA, L. & TRAMONTANO, E., 2013. Strategies of highly pathogenic RNA viruses to block dsRNA detection by RIG-I-like receptors: hide, mask, hit. Antiviral Research, 100, 615-635.
- ZVILICH, M., WILLIAMS, J. C., WAAG, D., RILL, W. R., BELL, P. & KENDE, M., 1995. Efficacy of Coxiella burnetiiand its chloroform-methanol residue (CMR) fraction against Rift Valley fever virus infection in mice. Antiviral Research, 27, 137-149.
- ZVILICH, M., WILLIAMS, J. C., WAAG, D., RILL, W. R., MALLI, R. J., BELL, P. & KENDE, M., 1995. Characterization of the non-specific humoral and cellular antiviral immunity stimulated by the chloroform-methanol residue (CMR) fraction of Coxiella burnetii. Antiviral Research,27, 389-404.