- Infectious Diseases of Livestock
- Part 2
- Orf
- GENERAL INTRODUCTION: PARAMYXOVIRIDAE AND PNEUMOVIRIDAE
- Rinderpest
- Peste des petits ruminants
- Parainfluenza type 3 infection
- Bovine respiratory syncytial virus infection
- Hendra virus infection
- Paramyxovirus-induced reproductive failure and congenital defects in pigs
- Nipah virus disease
- GENERAL INTRODUCTION: CALICIVIRIDAE AND ASTROVIRIDAE
- Vesicular exanthema
- Enteric caliciviruses of pigs and cattle
- GENERAL INTRODUCTION: RETROVIRIDAE
- Enzootic bovine leukosis
- Jaagsiekte
- Visna-maedi
- Caprine arthritis-encephalitis
- Equine infectious anaemia
- GENERAL INTRODUCTION: PAPILLOMAVIRIDAE
- Papillomavirus infection of ruminants
- Papillomavirus infection of equids
- GENERAL INTRODUCTION: ORTHOMYXOVIRIDAE
- Equine influenza
- Swine influenza
- GENERAL INTRODUCTION: CORONAVIRIDAE
- Porcine transmissible gastroenteritis
- Porcine respiratory coronavirus infection
- Porcine epidemic diarrhoea
- Porcine haemagglutinating encephalomyelitis virus infection
- Porcine deltacoronavirus infection
- Bovine coronavirus infection
- Ovine coronavirus infection
- Equine coronavirus infection
- GENERAL INTRODUCTION: PARVOVIRIDAE
- Porcine parvovirus infection
- Bovine parvovirus infection
- GENERAL INTRODUCTION: ADENOVIRIDAE
- Adenovirus infections
- GENERAL INTRODUCTION: HERPESVIRIDAE
- Equid herpesvirus 1 and equid herpesvirus 4 infections
- Equid gammaherpesvirus 2 and equid gammaherpesvirus 5 infections
- Equine coital exanthema
- Infectious bovine rhinotracheitis/infectious pustular vulvovaginitis and infectious pustular balanoposthitis
- Bovine alphaherpesvirus 2 infections
- Malignant catarrhal fever
- Pseudorabies
- Suid herpesvirus 2 infection
- GENERAL INTRODUCTION: ARTERIVIRIDAE
- Equine viral arteritis
- Porcine reproductive and respiratory syndrome
- GENERAL INTRODUCTION: FLAVIVIRIDAE
- Bovine viral diarrhoea and mucosal disease
- Border disease
- Hog cholera
- Wesselsbron disease
- Louping ill
- West nile virus infection
- GENERAL INTRODUCTION: TOGAVIRIDAE
- Equine encephalitides caused by alphaviruses in the Western Hemisphere
- Old World alphavirus infections in animals
- Getah virus infection
- GENERAL INTRODUCTION: BUNYAVIRIDAE
- Diseases caused by Akabane and related Simbu-group viruses
- Rift Valley fever
- Nairobi sheep disease
- Crimean-Congo haemorrhagic fever
- GENERAL INTRODUCTION: ASFARVIRIDAE
- African swine fever
- GENERAL INTRODUCTION: RHABDOVIRIDAE
- Rabies
- Bovine ephemeral fever
- Vesicular stomatitis and other vesiculovirus infections
- GENERAL INTRODUCTION: REOVIRIDAE
- Bluetongue
- Ibaraki disease in cattle
- Epizootic haemorrhagic disease
- African horse sickness
- Equine encephalosis
- Palyam serogroup orbivirus infections
- Rotavirus infections
- GENERAL INTRODUCTION: POXVIRIDAE
- Lumpy skin disease
- Sheeppox and goatpox
- Orf
- Ulcerative dermatosis
- Bovine papular stomatitis
- Pseudocowpox
- Swinepox
- Cowpox
- Horsepox
- Camelpox
- Buffalopox
- GENERAL INTRODUCTION: PICORNAVIRIDAE
- Teschen, Talfan and reproductive diseases caused by porcine enteroviruses
- Encephalomyocarditis virus infection
- Swine vesicular disease
- Equine picornavirus infection
- Bovine rhinovirus infection
- Foot-and-mouth disease
- GENERAL INTRODUCTION: BORNAVIRIDAE
- Borna disease
- GENERAL INTRODUCTION: CIRCOVIRIDAE AND ANELLOVIRIDAE
- Post-weaning multi-systemic wasting syndrome in swine
- GENERAL INTRODUCTION: PRION DISEASES
- Scrapie
- Bovine spongiform encephalopathy
- Transmissible spongiform encephalopathies related to bovine spongiform encephalopathy in other domestic and captive wild species
Orf
This content is distributed under the following licence: Attribution-NonCommercial CC BY-NC  View Creative Commons Licence details here
View Creative Commons Licence details here

Introduction
Orf is a contagious disease of sheep and goats characterized by vesicles, pustules and scabs, particularly around the mouth, caused by strains of parapoxvirus.9 The disease is infectious to humans. Orf is found in all sheep-rearing countries of the world, and the morbidity rate in all but well-isolated flocks approaches 100 per cent in yearling sheep and goats; in severe outbreaks the mortality rate can be greater than 10 per cent,2 particularly when associated with secondaryinfections such as those due to staphylococcus and fly strike. The disease tends to have a seasonal appearance, being particularly destructive when it appears around lambing. Depending on the husbandry practised, it is also common when weaned lambs and kids are put out to graze on coarse pasture, from which the term ‘thistle disease’ is derived.
Aetiology
The orf virion, as seen under the electron microscope, is oval in shape and covered in what appears to be a continuous filament (see the introduction, Poxviridae, and Figure 111.1), unlike the virions of other pox genera which are brick-shaped and covered in short tubular elements. The external dimensions measure 260 by 160 nm. The orf virion is morphologically and antigenically indistinguishable from other members of the parapoxvirus genus which cause pseudocowpox, bovine papular stomatitis and camel orf, and certain other parapox infections of wildlife. The genome is double-stranded DNA. Individual strains of orf virus can be characterized by the pattern produced on agarose gel by the restriction enzyme digests of the DNA. These distinguish orf virus from the parapoxviruses of cattle and other species, but do not exclude the possibility of some strains having the ability to infect both cattle and small ruminants. Monoclonal antibodies have been prepared against orf virus which identify a major neutralizing site, and may be of value in distinguishing between strains of parapoxvirus derived from different species.1
Traditionally, it was considered that orf virus was extremely resistant to inactivation in the environment, and this was used to explain how it persisted in a flock to infect each year’s lamb crop. Certainly the virus is more robust than most other viruses. It can retain some infectivity after 30 minutes at 55 °C, and scab material stored dry and in the dark at 7 °C remains infective for many years.
However, when exposed to sunlight or temperate environmental conditions, scab material loses infectivity in a few weeks, and the significance of orf virus persisting on pasture or on gateposts has probably been overstated. The virus can be destroyed by detergents containing lipid solvents, which destroy the virus membrane.
Epidemiology
Although the infection occurs mainly in young animals, both sexes and all breeds and ages of sheep and goats are susceptible to infection.8Wild sheep and goats may also become infected. Natural transmission occurs by direct contact or indirectly from the environment or fomites.8 Transmission is effected by the orf virus entering the host through abrasions in the skin or mucosa. The presence of persistently infected animals in a flock can explain its annual reappearance, and its introduction to a flock or herd often coincides with that of new, apparently uninfected stock.
Spread may also occur from wild ruminants accessing common pasture. Transmission between ewes and suckling lambs is common and results in the greatest losses, and is often associated with inability of the lambs to feed due to the presence of labial and/or buccal lesions and secondary mastitis in the ewes. Lambs at pasture may also be exposed to infection via buccal mucosa damaged by rough or sharp plant material, or skin softened by damp and muddy conditions. Other bacterial or viral infections which compromise the mucosal integrity can also predispose to orf infection, as does skin damage following shearing or rough handling. Vitamin A de- ficiency may contribute to susceptibility to infection. Poor hygiene, particularly in hand-milked flocks or herds, ensures the rapid spread of the virus.
In southern Africa it is believed that abrasions and other superficial injuries caused when feeding on dry stemmy or spiny feed and grass awns are the usual portal of entry of the virus. Goats tend to browse on thorny shrubs and trees, and this habit seems to be responsible for the severity of outbreaks of orf in Boergoats in Namibia and other semi-arid areas. Epidemics of orf in lambs sometimes occur during good rainy seasons when lambs are allowed on pastures with tall, wet grass or shrubs. The wet conditions give rise to softening and small wounds of the skin, particularly of the feet, with the result that the lesions are mostly confined to the coronet of the hooves.
Pathogenesis, clinical signs and pathology
Primary skin lesions develop two to six days after infection at the portal of entry of the virus to the body.8 Secondary lesions are formed by local extension or implantation of infective material at other sites of the body. There is no detectable viraemia. Labial, pedal, genital and generalized forms of the disease may occur, but the labial form is most common.8
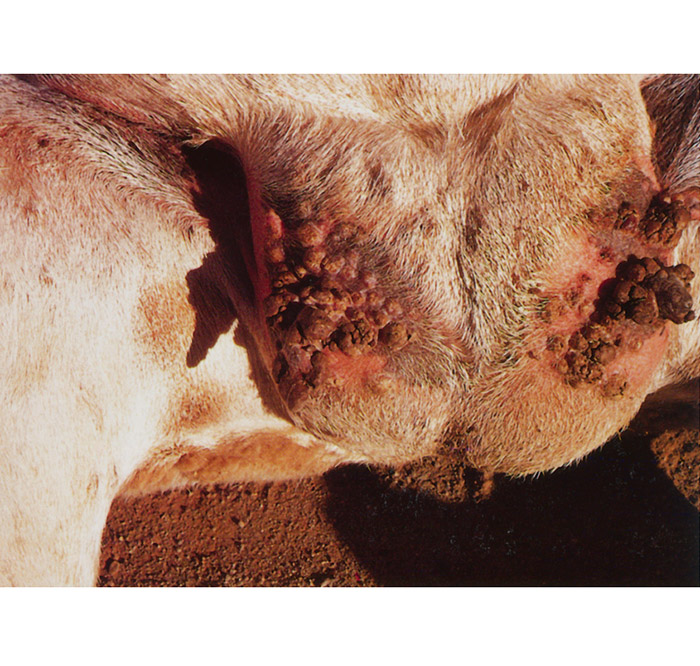
A macule develops at the site of virus entry, usually the lips, nostrils or udder, which enlarges to become a papule and, by the fourth to sixth day after infection, a transient vesicle. Twenty-four hours later this has changed to a pustule which soon ruptures, leaving an ulcer over which, by seven to eleven days after infection, a brown scab forms. Secondary lesions develop around the primary lesions or on other parts of the body where there has been skin damage. Lesions tend to commence at the commisures of the lips but are also common in the mouth (Figure 111.2 to 111.5), particularly on the gums around erupting teeth, and on the dental pad, palate and tongue. The mucosa of the oesophagus (Figure 111.6), rumen (Figure 111.7) and omasum may also be involved. Lesions may occur on the inner thigh, axilla, lower limbs, udder, teats, ventral aspect of the tail, and perineum.
Secondary bacterial mastitis is a common sequel. The pedal form is characterized by lesions in the skin of the coronet and interdigital space, and may even extend up to the carpus and tarsus. This results in varying degrees of lameness and, in severe cases, sloughing of the hooves. Secondary infections may aggravate the condition. Infection is by direct contact with the virus rather than by systemic spread. In uncomplicated cases all evidence of disease has disappeared within five weeks. In some animals large proliferative growths develop which persist for many months (Figure 111.8), a manifestation of which is strawberry footrot in older lambs, affecting the lower limbs, often associated with Dermatophilus congolensis infection; this may result in permanent lameness.
The histological changes10 seen with orf infection are confined to the epithelium, with ballooning degeneration and swelling of keratinocytes in the stratum spinosum, reticular degeneration, marked epidermal proliferation, intraepidermal micro-abscesses and accumulation of scale crust, vesicle formation and polymorphonuclear cell infiltration. Short-lived eosinophilic intracytoplasmic inclusions may be seen in the keratinocytes. Under the scab that forms after rupture of a pustule, there is marked epithelial hyperplasia and granulation tissue formation, which usually regresses between 22 and 40 days after infection. However, this proliferative lesion may persist, particularly on the polls and ears, but also on the udder and scrotum, and remain a rich source of orf virus. The involvement of a coinfecting papillomavirus is probable in some of these persistent lesions, and is consistent with the observation that they can spontaneously regress.
Secondary infections are common, particularly myiasis and staphylococcal mastitis in ewes resulting in loss of half the udder or death. Lambs may be unable or prevented from feeding, and those with internal lesions may develop a suppurative pleuropneumonia.
Humans, particularly farmers, herders, slaughterers and veterinarians, are occasionally affected (shepherd’s disease). The lesions are similar to those of pseudocowpox or milker’s nodule and are more proliferative than in sheep.4 Humans become infected through small abrasions of the skin when handling infected animals.
Diagnosis and differential diagnosis
A provisional diagnosis of orf is usually on the basis of clinical signs alone, but confirmation by electron microscopy and the demonstration of the typical orf virions is conclusive. An ELISA has been developed for detection of specific orf antigen. Virus can be isolated by tissue culture and neutralized by specific antibody. The value of antibody detection by ELISA, Western blotting or immunofluorescence is of doubtful diagnostic value because of the widespread distribution of the virus, unless a rising titre is shown in infected animals in paired sera collected two weeks apart.
Differential diagnosis includes sheep- and goatpox (capripox), dermatophilosis, bluetongue, foot rot, footand-mouth disease (FMD), papillomatosis, herpesvirus infection, staphylococcal infections, actinobacillosis, and ulcerative dermatosis, any of which may be present in addition to orf.
Control
Antibiotic treatment, particularly high doses of penicillin, can reduce the secondary infections, but have no effect on the virus itself. Topical sprays and ointments containing gentian violet may be helpful, and cautery or diathermy can be used on the proliferative lesions. Hand-feeding badly affected lambs will prevent death from nutritional deficiency.
Immunity following recovery is only partial, and animals affected a second time show an accelerated immune response in which the progress of the infection is reduced to 10 to 14 days, during which live virus can still be recovered.3 A variety of live vaccines derived from attenuated strains is available, but the protection provided cannot be expected to be better than that induced following natural infection with a virulent strain. The only value of vaccination is that it allows better timing for the disease to occur. There is evidence that lambs from vaccinated ewes may show milder clinical signs following challenge than those from unvaccinated ewes, suggesting that colostrumderived antibody does provide some protection.
Vaccine is applied by rubbing a small amount of vaccine into a skin scarification in the hairless areas of the axilla or groin. The axilla is the preferred site for ewes, as suckling lambs could become infected with vaccine virus applied to the groin.5 Ideally the ewes should be vaccinated eight weeks before lambing, to allow for full recovery and shedding of the scabs, and then moved to the lambing yards. Lambs can be vaccinated in the first two days after birth, also in the axilla to avoid the lamb spreading the virus to other sites on its body by being able to nibble at the developing vaccinal lesions.
A second dose administered six to eight weeks after the first has been recommended for lambs. The time of peak infection varies between flocks, countries and husbandry systems, and once identified, vaccination is best carried out six to eight weeks before this time. Vaccine can also be used in the face of an outbreak.
Orf vaccines should not be used in flocks that have not shown evidence of infection, as once introduced, the vaccine virus will persist in the flock.The effectiveness of the vaccine is poor compared with most other viral vaccines, but there are efforts to develop improved vaccines using new technology, such as vaccinia recombinants carrying inserts of orf genes which code for neutralizing epitopes.6
References
- CZERNY, C.-P., WALDMANN, R. & SCHEUBECK, T., 1997. Identification of three distinct antigenic sites in parapoxvirus. Archives of Virology, 142, 807–821.
- GUMBRELL, R.C. & MCGREGOR, D.A., 1997. Outbreak of severe fatal orf in lambs. The Veterinary Record, 141, 150–151..
- HAIG, D.MCK., HUTCHINSON, G., THOMSON, J., YIRRELL, D. & REID, H.W., 1996. Cytolytic activity and associated serine protease expression by skin and afferent lymph CD8+ T cells during orf infection. Journal of General Virology, 77, 953–961.
- LEAVELL, U.W., MCNAMARA, M.J., MUELLING, R., TALBERT, W.M., RUCKER, R.C. & DALTON, A.J., 1968. Orf. Report of 19 human cases with clinical and pathological observation. Journal of the American Medical Association, 204, 657–664.
- LEWIS, C., 1996. Update on orf. Practice, 18, 376–381.
- MERCER, A.A., YIRRELL, D.L., WHELAN, E.M., NETTLETON, P.F., POW, I., GILRAY, J.A., REID, H.W. & ROBINSON, A.J., 1997. A novel strategy for determining protective antigens of the parapoxvirus, orf virus. Virology, 229, 193– 200.
- MOORE, D.M., MACKENZIE, W.F., DOEPEL, F. & HANSEN, T.N., 1983. Contagious ecthyma in lambs and laboratory personnel. Laboratory Animal Science, 33, 473–475.
- MUNZ, E. & DUMBELL, K., 1994. Orf. In COETZER, J.A.W., THOMSON, G.R. & TUSTIN, R.C., (eds). Infectious Diseases of Livestock with Special Reference to Southern Africa. Cape Town: Oxford University Press
- ROBINSON, A.J. & BALASSAU, T.C., 1981. Contagious pustular dermatitis (orf). The Veterinary Bulletin, 51, 771–782.
- YAYER, J.A. & SCOTT, D.A., 1993. The skin and appendages. In: JUBB, K.V.F., KENNEDY, P.C. & PALMER, K., (eds). Pathology of Domestic Animals. San Diego: Academic Press.