- Infectious Diseases of Livestock
- Part 2
- West nile virus infection
- GENERAL INTRODUCTION: PARAMYXOVIRIDAE AND PNEUMOVIRIDAE
- Rinderpest
- Peste des petits ruminants
- Parainfluenza type 3 infection
- Bovine respiratory syncytial virus infection
- Hendra virus infection
- Paramyxovirus-induced reproductive failure and congenital defects in pigs
- Nipah virus disease
- GENERAL INTRODUCTION: CALICIVIRIDAE AND ASTROVIRIDAE
- Vesicular exanthema
- Enteric caliciviruses of pigs and cattle
- GENERAL INTRODUCTION: RETROVIRIDAE
- Enzootic bovine leukosis
- Jaagsiekte
- Visna-maedi
- Caprine arthritis-encephalitis
- Equine infectious anaemia
- GENERAL INTRODUCTION: PAPILLOMAVIRIDAE
- Papillomavirus infection of ruminants
- Papillomavirus infection of equids
- GENERAL INTRODUCTION: ORTHOMYXOVIRIDAE
- Equine influenza
- Swine influenza
- GENERAL INTRODUCTION: CORONAVIRIDAE
- Porcine transmissible gastroenteritis
- Porcine respiratory coronavirus infection
- Porcine epidemic diarrhoea
- Porcine haemagglutinating encephalomyelitis virus infection
- Porcine deltacoronavirus infection
- Bovine coronavirus infection
- Ovine coronavirus infection
- Equine coronavirus infection
- GENERAL INTRODUCTION: PARVOVIRIDAE
- Porcine parvovirus infection
- Bovine parvovirus infection
- GENERAL INTRODUCTION: ADENOVIRIDAE
- Adenovirus infections
- GENERAL INTRODUCTION: HERPESVIRIDAE
- Equid herpesvirus 1 and equid herpesvirus 4 infections
- Equid gammaherpesvirus 2 and equid gammaherpesvirus 5 infections
- Equine coital exanthema
- Infectious bovine rhinotracheitis/infectious pustular vulvovaginitis and infectious pustular balanoposthitis
- Bovine alphaherpesvirus 2 infections
- Malignant catarrhal fever
- Pseudorabies
- Suid herpesvirus 2 infection
- GENERAL INTRODUCTION: ARTERIVIRIDAE
- Equine viral arteritis
- Porcine reproductive and respiratory syndrome
- GENERAL INTRODUCTION: FLAVIVIRIDAE
- Bovine viral diarrhoea and mucosal disease
- Border disease
- Hog cholera
- Wesselsbron disease
- Louping ill
- West nile virus infection
- GENERAL INTRODUCTION: TOGAVIRIDAE
- Equine encephalitides caused by alphaviruses in the Western Hemisphere
- Old World alphavirus infections in animals
- Getah virus infection
- GENERAL INTRODUCTION: BUNYAVIRIDAE
- Diseases caused by Akabane and related Simbu-group viruses
- Rift Valley fever
- Nairobi sheep disease
- Crimean-Congo haemorrhagic fever
- GENERAL INTRODUCTION: ASFARVIRIDAE
- African swine fever
- GENERAL INTRODUCTION: RHABDOVIRIDAE
- Rabies
- Bovine ephemeral fever
- Vesicular stomatitis and other vesiculovirus infections
- GENERAL INTRODUCTION: REOVIRIDAE
- Bluetongue
- Ibaraki disease in cattle
- Epizootic haemorrhagic disease
- African horse sickness
- Equine encephalosis
- Palyam serogroup orbivirus infections
- Rotavirus infections
- GENERAL INTRODUCTION: POXVIRIDAE
- Lumpy skin disease
- Sheeppox and goatpox
- Orf
- Ulcerative dermatosis
- Bovine papular stomatitis
- Pseudocowpox
- Swinepox
- Cowpox
- Horsepox
- Camelpox
- Buffalopox
- GENERAL INTRODUCTION: PICORNAVIRIDAE
- Teschen, Talfan and reproductive diseases caused by porcine enteroviruses
- Encephalomyocarditis virus infection
- Swine vesicular disease
- Equine picornavirus infection
- Bovine rhinovirus infection
- Foot-and-mouth disease
- GENERAL INTRODUCTION: BORNAVIRIDAE
- Borna disease
- GENERAL INTRODUCTION: CIRCOVIRIDAE AND ANELLOVIRIDAE
- Post-weaning multi-systemic wasting syndrome in swine
- GENERAL INTRODUCTION: PRION DISEASES
- Scrapie
- Bovine spongiform encephalopathy
- Transmissible spongiform encephalopathies related to bovine spongiform encephalopathy in other domestic and captive wild species
West nile virus infection
This content is distributed under the following licence: Attribution-NonCommercial CC BY-NC  View Creative Commons Licence details here
View Creative Commons Licence details here
NJ Maclachlan and M-L Penrith (Editors). M Long, West nile virus infection, 2019.
West Nile virus infection
Previous authors: M L BUNNING, T M WILSON AND R A BOWEN
Current authors:
M T LONG - Fern Audette Chair and Associate Professor, DVM, PhD, Department of Comparative Diagnostic and Population Medicine, College of Veterinary Medicine, University of Florida, Veterinary Academic Building V3-157, 2015 SW 16th Avenue, Gainesville, Florida, FL 32608, USA
Introduction
West Nile virus (WNV) infection is mosquito-borne and may cause severe and fatal neurological disease in birds, humans, horses, and, more sporadically, a wide variety of other animals. The virus was first isolated in the West Nile district of Uganda in 1937.188 Prior to 1999, WNV caused only sporadic outbreaks of disease in humans and horses in Africa,77, 197 the Middle East69, 82 and Europe.79 Kunjin virus (KUN), a variant of WNV found in Australia, caused an extensive outbreak in 2011 in horses. In the late 1990s and early 2000s, severe concurrent disease outbreaks occurred in the United States (US), France, and Israel.69, 79, 218 At present, WNV is endemic in all continents except Antarctica.44 Following introduction to North America in 1999,64, 103 WNV was detected in Central and South America by 2005.46, 96, 134, 146 Expansion of all virus lineages with new subtypes has occurred in the US, Europe, and Eurasia.51 Finally, WNV was once considered a cause of subclinical or febrile disease in Africa, but reports of neurovirulent WNV have increased in Africa since 2007.206 The continued spread of WNV indicates that its associated disease will remain a threat to the health and well-being of livestock and humans worldwide.
Aetiology
West Nile virus is grouped within the genus Flavivirus of the family Flaviviridae (see General Introduction: Flaviviridae), an ever expanding group of viruses that as a family is one of the most pathogenic to animals and humans.86, 219 Flaviviruses, including WNV, are positive sense, single-stranded RNA viruses measuring approximately 45-50 nm in diameter. The virions are spherical, enveloped and contain a nucleocapsid.136 The genome is approximately 11kilobase (kb) and contains a single open reading frame (ORF) that is translated in its entirety and cleaved by both cell and viral proteases into 11 viral proteins, specifically three structural proteins, including capsid (C), premembrane (prM)/membrane (M), and envelope (E), and seven nonstructural (NS) proteins.57, 89, 102, 136, 157 The mature virion has 180 copies of the E protein arranged in a herringbone pattern covering a lipid bilayer.72, 99, 109, 132, 141, 145, 159, 193, 226 The E protein is dimerized, composed of two each of three domains (E-DI, E-DII, and E-DII) connected by hinges forming a type II viral fusion protein. E-DI is part of the central domain of the E protein and is an eight -barrel protein linked by two disulphide bonds with an N-link carbohydrate site. In vivo neurovirulence is altered with mutation of the amino acid residues 154-156, a N-Y-S glycosylation site of the E-DI.24, 76, 136, 157 A newly emerged neurovirulent strain of WNV in Australia (Kunjin virus [KUN]) is also glycosylated at this site whereas less virulent KUN viruses are not.70 The domain E-DII mediates dimerization and has the glycine rich fusion loop protein that inserts into the membranes of target cells. Located in a hydrophobic pocket between E-DI and E-DIII, mutation of E-DII renders the virus unable to fuse with synthetic membranes.142, 145 E-DIII forms as an immunoglobulin-like structure at the carboxy-terminus and projects from the surface of the virus.50, 142, 159, 177, 178 Since monoclonal antibodies to this site result in neutralization of the virus, this domain contains the main binding epitopes for attachment and entry into cells.42 The prM protein is essential for E protein folding, trafficking through the Golgi, and prevents interaction of the virus with futher cell membranes during egress.108, 110, 122, 158, 224 Maturation of the virus is not considered complete until this protein is cleaved by a cellular furin-like serine protease.225 However, immature viral particles can be released independent of furin maturation and have demonstrated cellular infectivity.158
The first step in viral infection occurs when domain III of the E protein binds to host cell receptors.111 Receptor mediated endocytosis is clathrin-mediated, and the virus enters the host cell within a low pH vesicle.15, 27, 41 This low pH is the signal for E protein re-arrangements (switch from a dimer to a trimer form) to occur, exposing domain II of the E protein and allowing it to fuse to the vesicle membrane. The virus is then released from the vesicle into the cell cytoplasm. At this stage, a single strand of positive sense RNA is present in the host cell cytoplasm.111 Translation occurs first, when host cell elongation initiation factors (eIF) bind to the 5’ untranslated region (UTR) of the viral genome. Host ribosomal subunits rRNA and tRNA with attached amino acids are then added in the traditional initiation, elongation, and termination steps of translation to form a polyprotein. Host cell proteases then cleave the polyprotein, allowing replication to occur. In subsequent translational events, the viral NS2B/NS3 protease carries out the cleavage of the polyprotein. Replication starts at the 3’ UTR of the viral RNA. The viral NS4B/NS5 RNA-dependent-RNA-polymerase (RdRp) binds to conserved stem loop structures of the 3’UTR along with a variety of host cell proteins. Other viral proteins involved in this replication complex include NS1 and NS4A. The viral NS3 helicase acts to unwind/stabilize the RNA genome. Replication of a negative-sense RNA strand then occurs in a 5’ to 3’ direction. These negative sense strands serve as the template for the creation of positive-sense viral RNA genomes. Replication of positive-sense RNA strands occurs when the same viral and host replication complex binds to the 3’ end of the negative-sense RNA (5’ UTR of the viral genome). There is no polyA tail on the viral RNA genome, but a 5’ cap is added by the NS5 viral protein. Viral packaging occurs once enough viral proteins and genomes have been created in the Golgi apparatus. The C protein is arranged around the positive-sense RNA with the assistance of NS2A. The prM and E proteins are then arranged around the viral capsid.
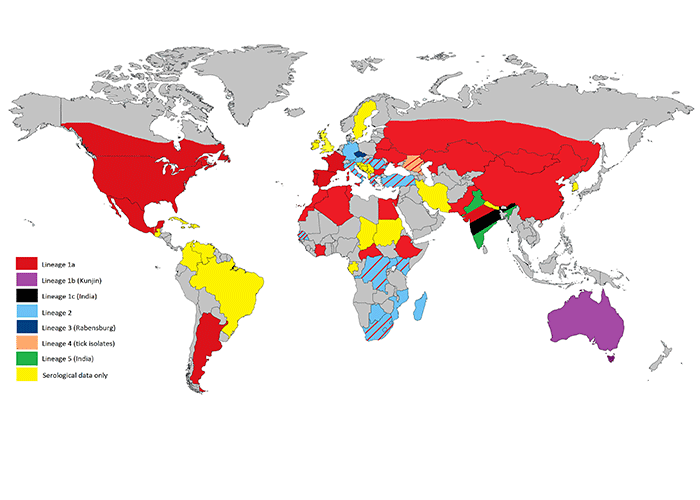
West Nile virus shares a close antigenic relationship with the other viruses of the Japanese encephalitis virus (JEV) sero-complex, as well as at least eight other viruses of the family Flaviviridae comprising the West Nile subgroup or complex of viruses.96, 118 Several of these viruses are responsible for causing disease in horses, including JEV, WNV (including KUN virus, which genetically is a lineage 1 WNV identified as WNVKUN), and Murray Valley encephalitis virus (MVE).86, 219 Based on phylogenetic analyses, strains of WNV typically segregate into five lineages.44 Lineage 1 (L1) is the most diverse and includes 3 sub lineages, 1a, 1b and 1c (Figure 1). Only lineage 1, 2 and 5 viruses have been documented to cause neuroinvasive disease in horses, and susceptibility of horses is likely similar to that of humans. There are significant differences in nucleotide sequences between WNV isolates from different parts of the world as well as marked antigenic variation among strains isolated within the same geographic region.47 This heterogeneity in WNV results in continued emergence of new dominant viruses in individual regions, leading to new outbreaks.47
Expansion and contraction of sub lineages of WNV have formed the basis of many new epidemics.55, 56, 123 Lineage 1a viruses are responsible for most North America epidemics45 with new subtypes emerging in 2005 and 2011.51, 54, 162 Neuroinvasive L1a viruses were also active during the late 1990s causing infections in humans and horses in Africa, Europe, Australia, Asia, Central and South America, and the Middle East.54, 162 The first WNV L1 case in India was detected in 2012.54, 123 Currently circulating Lineage 1b viruses are strains of WNVKUN that occur in Australia, some South-East Asian countries, and New Guinea.70, 78, 154 In 2011, some 900 horses in Australia developed disease following expansion of a new 1b sublineage virus, now designated as WNVNSW2011.70 This newly emergent virus is more neurovirulent than the prototype WNVKUN strain, possibly as a consequence of altered glycosylation of its E protein.70, 120 The WNV 1c viruses that occur in India are relatively avirulent to horses,55, 56, 123 although this sublineage may soon be redesignated as L5.
The surge in cases of neuroinvasive disease due to WNV L2 infections in Europe in approximately 2005 is noteworthy.9, 14, 223 Previously, WNV L2 was known to be endemic in Africa and typically responsible for only flu-like clinical disease in people and primarily subclinical disease in horses, but a new phenotype of L2 that appeared in Europe caused outbreaks of neuroinvasive disease in both people and horses in Northern Europe, Italy, and Greece.9, 36, 38, 43 Greece has experienced at least four human epidemics and neuroinvasive L2 has completely replaced L1 infections in Italy.211, 223 An increase in virulence of L2 with increase neuroinvasive disease in Africa mirrors this increase in WNV L2 in Europe.206, 207 People are considered at higher risk for neuroinvasive disease. Horses have been diagnosed with encephalitis from both L1 and L2 with increasing frequency in Africa.
Epidemiology
The first five years of WNV emergence in the US consisted of an explosive epidemic affecting thousands of humans, horses, and birds followed by a steep drop in activity.66 Since then cyclical but local reintensification has characterized US activity47, 119 All provinces of Canada have reported WNV activity including the Northwest Territories, Yukon, and Nunavut.54, 58, 217 The incidence of equine and human disease appears low for Central and South America and the Caribbean compared to the US and many countries reported evidence of the virus between 2005 and 2006.12, 15, 60, 96, 121 France, Greece, Italy, the United Kingdom, and Austria have reported the most activity in recent years.10, 13, 14, 32, 170, 192, 210, 211, 223 In the United Kingdom, positive birds are relatively frequent, but equine cases reported are considered to have been exposed to the disease elsewhere. In South Africa, seroconversion rates to WNV are higher than previous years for horses along with reports of high mortality in horses.206, 207
In the horse, sex, age, and breed predilection for WNV encephalomyelitis have been examined. In some studies, female horses more commonly developed severe neuroinvasive disease or tested positive at the time of neurological disease in stablemates.147, 162, 174, 196 In another study, factors found to positively contribute to mortality in horses included light coat color, vaccine status, pasture-only management, and lack of solid stall walls if housed.65 Vaccination was the most important factor for prevention of disease, demonstrating 96 per cent efficacy in prevention of clinical WNV under field conditions.65, 66 Experimental as well as field studies have unequivocally demonstrated that vaccination reduces mortality due to WNV.114, 174, 216 Horses that were not given the required second injection during the first vaccination had increased risk of disease and mortality if vaccinated in the face of WNV activity.70 However, in horses that received partial vaccination well before the WNV season, the risk for neuroinvasive disease and mortality was significantly lower.65, 169
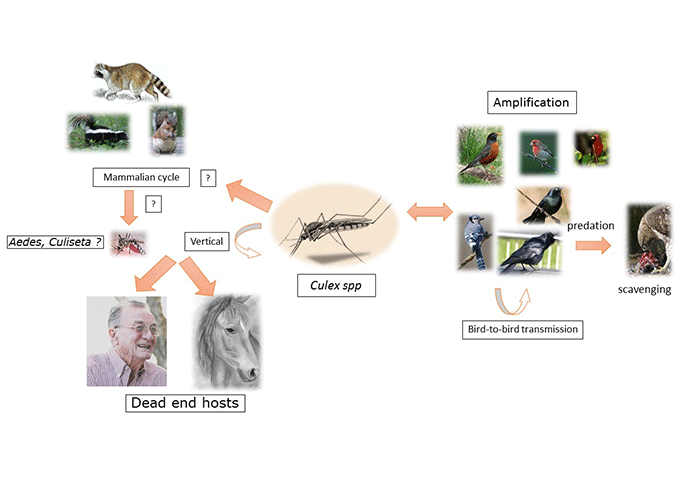
West Nile virus cycles in nature primarily in mosquitoes and wild birds, the specific species of which vary between geographic areas (Figure 2). Since 2000, new modes of transmission and hosts have also been identified. Blood meal-seeking mosquitoes within the Culex pipiens complex are the most significant vectors of WNV worldwide.2, 5, 91-93, 133, 187, 201 In the eastern US, approximately half of the WNV-positive mosquito pools are Cx. pipiens.2, 3, 129 Based on numbers of positive pools, Cx. nigripalpus and Cx. quinquefasciatus are the common vectors in the southern US.79 Culex tarsalis is a major vector west of the Mississippi river.12, 74, 209 In Europe, Cx. modestus and Cx. pipiens are principal vectors, and Cx. modestus has been shown to be a major amplifier of outbreaks in summer.11, 135, 199 Culex pipiens complex mosquitoes have been implicated as the main vector of WNV in the Middle East63, 151, 180, 202 and Cx. univittatus the principal vector in Africa.85, 126, 131 Other mammal-feeding mosquitoes that potentially contribute to transmission of WNV in the US include Anopheles quadrimaculatus, Coquillettidia perturbans, and Aedes albopictus.3, 7, 201
Migrating birds serve as the principal hosts for introducing WNV into new areas. Historic outbreaks across North America paralleled all of the US continental flyways, and over 300 species of mortally affected birds were described during years of high bird mortality.6, 155, 165 Although WNV infection produces very high mortality amongst birds of the family Corvidae (e.g. crows) in both the US and Europe, other species of birds that develop significant viraemia but without high mortality are likely primary drivers of the transmission cycle.91, 93, 94 For example, in the US, the American robin (Turdus migratorius) and house sparrow (Passer domesticus) are considered “super spreaders”.91, 205, 220 In Europe, virus has been commonly isolated from crows, pigeons, various native and migrant aquatics, and terrestrial birds.81 However, more recent introductions of new WNV lineages have caused die offs of sparrows and the Eurasian magpies (Pica pica), domestic geese, and white storks (Ciconia ciconia).80, 117, 118, 200 Migrant passerines and waterfowl introduce WNV from the Mediterranean and northern Africa and include the European turtle dove (Streptopelia turtur), mallard duck (Anas platyrhynchos), Eurasian teal (Anas crecca), Eurasian woodcock (Scolopax rusticola), and song thrush (Turdus phylomelos). The garden warbler (Sylvia boron), common blackbird (Turdus merula), the common redstart (Pheonicurius pheonicurius), the woodchat shrike (Lanius senator) and the European pied flycatcher (Ficedula hypoleuca) are incriminated for introduction from central Africa17, 117, 200, 204 Once resident in Europe, WNV likely is then transmitted via Cx. modestus and Cx pipiens to resident birds such as house sparrows, the Eurasian magpie , C. cornix and C. menudula.31, 39, 204
Natural WNV infection has been identified in many animal species before the emergence of WNV. Historically, serosurveys had identified WNV-specific antibodies in wild mammals such as lemurs, chimpanzees, rodents, bats, and domestic mammals such as camels, horses, mules, donkeys, goats, cattle, water buffalo, sheep, and pigs in Africa and the Middle East.18, 69, 77, 144 With severe and widespread outbreaks of WNV in the US, bats (Eptisecus fuscus and Myotis lucifugus), eastern chipmunks (Tamias striatus), squirrels (Sciurus carolinensis and S. niger), skunks (Mephitis mephitis), deer (Odocoileus virginianus), bears (Ursus arctos, U. maritimus), rabbits (Oryctolagus spp.), raccoons (Procyon lotor), alpacas, llamas, and goats have either tested seropositive or have had reported clinical disease in North or South America.59, 62, 67, 98, 102, 129, 148, 160, 194, 198, 221 Alligators (Alligator mississippiensis) develop extremely high viraemias and young alligators experience high mortality rates.95, 128
Seroconversion rates in dogs are high in areas of WNV activity.17, 37, 68, 90, 127 In fact, domestic dogs have been investigated epidemiologically in several studies in Europe, Canada, and the US. It is clear that with high seroconversion rates and earlier conversion in stray dogs compared to humans, dogs could be used as sentinels since horses are widely vaccinated and limited disease occurs in this species.68, 90 Wild canids demonstrate high levels of exposure serologically.17, 37, 68, 90, 127 Domestic cats are susceptible to infection orally and can develop high enough viraemias to transmit disease.8, 90 Potential other reservoirs include the raccoon and striped skunk because of high seroconversions rates.17, 48, 53
Pathogenesis
West Nile virus infection of animals occurs after the bite of an infected mosquito. Infection of mosquitoes occurs if a blood meal from a viraemic host contains between 104 and 105 plaque forming units (PFU)/ml of virus.73 Whereas many species of birds develop very high-titre viraemia following infection, humans and horses are “dead-end” hosts that develop only a low-titre viraemia. Unlike the restricted neurotropism exhibited by this virus in mammalian infection, WNV has an unrestricted tissue tropism in birds.71 Upon experimental infection, many species of birds develop viraemia capable of transmitting virus for 4 days or longer and depending on species, those titres have ranged between 102 PFU/ml and 108 PFU/ml.71, 138, 139 Upon infection, there is local replication with rapid dissemination of virus to all organs and even feathers.97, 99 Onset of viraemia is variable and depending on species can be within 1 day, but in general passerines develop a peak viraemia around 6 days post- infection (DPI) while raptors can develop peak viraemia around 10 DPI. If a bird does not succumb, viraemia usually decreases over the next several days. However, in some passerines, virus can persist for 4 – 6 months. Infection of the CNS occurs rapidly in highly susceptible species such as the crow.139 In larger birds, CNS infection can take between 4 and 6 DPI.138 West Nile virus is detected often in endothelial cells of the brain, which may indicate direct invasion of the CNS from the peripheral circulation.116, 222 Viral antigen is detected in mononuclear cells within tissues.116, 222 With initial widespread dissemination to the systemic organs including the kidneys, spleen, and heart, virus persistence has been demonstrated in these organs as well as eyes, brain, and skin.71, 220 Given the difference in species susceptible to WNV, which includes 100 per cent mortality in the American crow, work has been done to determine how the strain of virus contributes to the pathogenesis in birds. The first work demonstrated that a single substitution in the NS3 protein (T–P) of WNV NY99 strain resulted in increased virulence for this species; however, this same substitution did not account for increased disease in European birds.23, 24, 190 When WNV L2 caused increased mortality in birds in Greece, a change in this amino acid (H-P) was detected, but not all birds that succumbed to WNV L2 had this amino acid change during the encroachment of L2.149 The outcome of WNV infection in birds is also more complex, as nutrition, age, and co-morbidity are important. Finally, natural infection in birds may be highly variable depending on route of infection. Raptors and insectivorous birds may acquire WNV orally. In experimental infection when orally infected, birds generally develop less severe disease.97
Upon infection of horses, virus titres usually range between 101,0 PFU/ml and 103,0 PFU/ml of serum for 12-36 hours, which is below the minimum threshold to infect mosquitoes.30 Based on both epidemiological and peripheral challenge experiments, the approximate symptomatic:asymptomatic ratio is approximately 1:11.30 In horses with neuroinvasive disease, titres in the brain vary from undetected up to 106,8 PFU/g tissue. Distribution of the virus is variable and multifocal in the brain and spinal cord, but for diagnostic purposes is most consistently present in the thalamus, pons and medulla.30, 114 In the spinal cord, virus is most likely detected in the areas of the cord where lesions are identified.
Understanding the pathogenesis of clinical disease in horses depends on the neuroanatomical localization of the virus and its associated pathology. Clinically, neurological disease in horses consist of changes in mentation, spinal disease, and cranial nerve aberrations.189 Change in behaviour likely results from infection of the thalamus, medulla and pons with limited viral load in the cerebrum.35, 114 Pathology and virus localization are most abundant in the midbrain and rostral pons and cause disruption of nervous conduction of the reticular formation leading to changes in consciousness and sensorium.35, 114, 189 Clinical signs commonly attributed to forebrain involvement are probably due to infection of the midbrain that controls cholinergic stimulation to the entire cerebral cortex since limited virus is found in the cortex. Thus, what could be diagnosed as coma (common with forebrain disease) may be the result of combined sympathetic, cholinergic and neurotransmitter disruption in the large nuclei of the midbrain.20, 189 In terms of the motor deficits, these are multifocal, asymmetric and have components of weakness ranging from episodic hemiparesis to complete paralysis. Since WNV infects neurons, especially large cell bodies, loss of fine motor and coarse control occurs due to high viral loads and severe pathology of the thalamus and basal ganglia.114 Disruption of motor tracts in the hindbrain leads to loss of proprioceptive function resulting, in some cases, in marked ataxia. When infections occur in the large nerve cell bodies of the spinal cord, flaccid paralysis of one or more limbs is common.166 Experimental rodent and hamster models indicate that transaxonal infection occurs, thus localized paralysis may be due to this mode of CNS invasion.176
In mammals, two mechanims of WNV neuroinvasion are proposed, either via the blood brain barrier (BBB) or via retrograde transaxonal movement of virus. Theoretically, neuroinvasion across the BBB would be more likely to result in generalized CNS infection presenting as meningitis/encephalomyelitis. Transaxonal invasion would present as localized flaccid paralysis. In support of this, footpad WNV challenge models demonstrate localization to the olfactory bulbs after infection.195 Studies in hamsters showed that injection of virus in the vicinity of the sciatic nerve results in transaxonal movement of virus into the spinal cord with flaccid paralyais. Experimental data support both transcellular and paracellular pathways across the BBB.171, 195, 208 Tight junction proteins (TJP), and other molecules that maintain endothelial integrity such as occludin, claudin-1, -catenin, and adherin are downregulated in mouse infection with WNV.171In vitro, WNV infects endothelial cells readily although infection results in few cytopathic effects and is highly variable in individual cells.51 This may indicate that transendothelial infection is highly efficient leading to CNS infection without significant damage to the endothelium.208 West Nile virus infection of the brain, irrespective of neurovirulence properties of viral strain, upregulates adhesion molecules, intracellular adhesions moledule (ICAM)-1, VCAM-1 and endothelial-leukocyte adhesion (ELAM)-1 enhances transmigration of leukocytes, which implicates white blood cells as vehicles for invasion.172The most studied component of cellular injury and death in WNV infection is apoptosis. West Nile virus infection activates pro-apoptotic and pro-survival pathways.143 The C protein of flaviviruses is both anti-apoptotic and pro-apoptotic, blocking caspase (CAS)-3/8 and inducing caspase (CAS)-9, respectively.16, 175, 203 Conversely, NS3 interacts with NS2B to induce the CAS-3/8 pathways through induction of IFN α/.164 NS2A protein has been shown to decrease cellular injury through blockade of the IFN-/ pathway as well as independently of the interferon pathway.113, 124 Other mechanisms of apoptotic injury include the Bcl-2-like protein 4 (BAX)-dependent and stress pathways.1, 26, 150
In experiments,with susceptible mice, infection is almost always lethal with high amounts of virus especially in the cerebellum. CD8+ T cells are associated with decreased viral load and less pathology.184, 185 In horses, undergoing grave WNV infection there is low viral load. These horses also have a mixture of CD4+ and CD8+ T cells, but the dominant cellular phenotype consists of glial cells.49 Thus, resident cells may be more important in the pathogenesis of disease in the outbred host. Since horses have low viral load with profound disease, the main pathogenesis in the horse may be due to indirect or bystander injury, where neighboring cells adjacent to cells undergoing apoptosis are also damaged or killed.125 Evidence has been documented in horses20 and supported in the mouse model that excitatory neurotransmitters are highly dysregulated.19, 45 In horses, differential expression of mRNA expression indicates that almost all glutamate transporters and receptors are downregulated post-infection, which is highly indicative of an excess of the excitatory neurotransmitters in the synaptic cleft.19 Over excitation, if calcium dependent, then leads to apoptosis of neurons.
Autonomic dysfunction occurs with changes in electrocardiograms.213 Electromyographs performed on intestinal smooth muscle were reduced in a rodent model, which may account for gastrointestinal disturbances observed in horses and humans.213 However, autonomic dysfunction was not incriminated as a cause of death in this model.214 In rodent models phrenic nerve amplitude is reduced without corresponding changes in brainstem responses, indicating lower motor neuron disease may drive the pathogenesis of respiratory failure.227
The pathogenesis and outcome of WNV infection is a complex interplay of antiviral innate immune responses and viral proteins that induce and alter the response.105 Several pattern recognition receptors induce IFN-α/β, which are crucial for successful control of flavivirus infections.107 Induction of IFN signalling results in upregulation of several IFN-stimulated genes (ISG), which in turn induce expression of multiple antiviral responses through Janus kinase (JAK)-(signal transducer of activator of transcription proteins) STAT signalling.25, 106 These in turn upregulate G-protein signalling pathways to form adhesins that act to enhance tight junctions, thereby blocking WNV infection in the brain.104, 175 Direct antiviral mechanisms within the cells primarily driven by RNAse-L and 2-5-oligadenylate synthetase (OAS) responses destroy intracellular “foreign” RNAs. Upon induction with type 1 IFN, both RNAsel and elongation initiation factor (eIF)-2 degrade viral RNA and inhibit viral transcription, respectively.153, 158 Genetic predisposition giving rise to differences in the innate immune response is implicated. Initial studies on the pathogenesis in knock-out mice indicated a genetic component, which led to the identification of the “flavivirus susceptibility” gene in mice.26, 153 In horses, research examining polymorphisms in IFN response pathways, OAS-1 and RNase-L, demonstrated association with survival.168 A number of viral proteins, in turn, block the host cell response; NS4A and NS4B are effective at blocking the JAK-STAT translational pathway.52 The inhibition of the type I interferons by viral proteins leads to a block in the transcription of 2’5’ OAS and PKR kinase.
While much of the mechanistic investigation of the role of adaptive immunity in protection against WNV has been performed in mice, the role of adaptive immunity has been unequivocally demonstrated in equine WNV vaccine studies.21, 114, 140, 182, 186 Furthermore, studies in mice and hamsters demonstrate that serum from vaccinated horses protects mice against WNV challenge.140, 182 Humoral immunity in the form of both neutralizing antibody to the E protein and the NS1 have been shown to confer protection against lethal WNV challenge in mice by adoptive transfer experiments and studies using B cell knock out mice.184 Isotype IgG1 has been shown to increase consistently with induction of neutralizing activity in the horse.177, 178 In the only study examining neutralizing epitopes in the horse, the same epitope that has neutralizing activity in the mouse was not the dominant epitope in the mouse.177
Clinical signs
Birds as the most important reservoir species exhibit variation in susceptibility to disease, duration of viraemia, and level of viraemia.97 Futhermore, level of viraemia may not correlate to susceptibility to disease. Both North American and European corvid species have high fatality rates with high viraemia and succumb rapidly after infection and demonstrate widespread organ and vascular infection.97, 139 Often these animals are found dead but when presented alive are extremely emaciated, depressed and have a history of exhibiting abnormal behaviours, imbalance, and inability to fly.97 In experimental infection, birds become lethargic, ataxic, and develop abnormal pupillary responses.97, 138, 139 Loss of moytactic, postural, and proprioceptive deficits can occur.97, 138, 139 Birds also exhibit poor beak strength and laboured breathing.97, 138, 139 In other passerines, susceptibility to disease, die offs, and drops in population have occured in birds such as the Amerian robin, eastern bluebird and tufted titmouse, while birds such as the house wren show little disease or population declines.96 Raptors are often affected with clinical disease due to WNV. Experimental challenge studies revealed limited disease, but sample sizes tended to be small. In analyses of birds admitted to rehabilitation facilities and in captive collections, WNV infected birds were more likely to have longer rehabilitation, higher mortality, and higher incidence of neurological disease. Multiple species of raptors are susceptible to WNV infection, but susceptibility to neuroinvasive disease varies across species.137 Owl species of the Strigidae family often present with neurological signs that include change in mentation and disorientation, weakness, ataxia, limited ability to fly, head tilt, torticollis, nystagmus, seizures, recumbency, and sudden death.138 Like horses, these birds exhibit tremors and tetraparesis.137, 138 Large hawks, buteos, and eagles all have a higher frequency of disease while smaller hawks such as Swainson’s hawks, red tailed hawks, and kestrels tend to present with emaciation, depression, and anorexia.137 Feather abormalities are common in these latter species. Waterfowl can exhibit more prolonged disease including multiorgan disease, neurological signs, and cardiac arrthymias.183 In experimental infection of wild and domestic ducks, severity of disease was age dependent with higher mortality in young domestic and wild ducks; however, ducks develop high viraemia with oral and cloacal shedding.183
Clinical disease in the horse, primarily manifesting as outbreaks of encephalomyelitis, was first reported in Egypt in 1963180, 185 and France in the early 1970s.79 Multiple outbreaks of WNV L1 associated disease then occurred in the Rhone River Delta of France,61 Morocco,197 Israel,191 Italy,34, 35, 163 and the US.181, 189 Both WNV L 1 and 2 are now recognized to cause neuroinvasive disease in the horse with neuroinvasive L2 emerging after 2005.10, 75 Finally, this increase in virulence is a risk for African horses and other mammals. Since around 2007, WNV L2 has also demonstrated neuroinvasive disease in African horses with 35 per cent mortality.
Clinical neurological disease in horses, whether from natural infection or experimentally reproduced, follows the same course and progression of clinical signs.30, 114, 162, 174, 182, 216, 217 A mild to moderate increase in rectal temperature (38.6-39.4C), anorexia, and depression are the most common initial signs and this usually occurs between 7 and 9 days post experimental inoculation.162 Abdominal pain is also a regular initial complaint.162, 181, 189 Gait abnormalities including lameness or dragging of a limb has been reported before development of an obvious neurological syndrome.162 One of the initial signs of motor abnormality is a short, slow, stilted gait that appears as lameness, and laminitis is a frequent differential at this stage.162 However, in human WNV disease, bradykinesia or slow, deliberate movement is described, and this may be the equine corollary.156 With full progression to neurological disease, both spinal cord disease and mental aberrations occur.
The major neurological features consist of changes in personality and muscle fasciculations.181, 189 Many horses have periods of hyperexcitability and apprehension, sometimes to the point of aggression. Fine and coarse fasciculations of the face and neck muscles are very common.160 Fasciculations can be quite severe and involve all four limbs and trunk, affecting normal activities such as walking, eating, and interaction with handlers and other horses. The fasciculations are most notable at the muzzle and eyelids. Some horses have periods of sudden sleep-like behaviour resembling narcolepsy and, when prolonged, horses are cataplectic. During these episodes horses can collapse suddenly and remain recumbent. Seizures and coma have been reported.160, 189 Eyelid activity is enhanced with light and at times it appears that horses are photophobic.173 Spinal abnormalities are characterized by ataxia and paresis that can be highly asymmetrical or involve only one or both of the front or hind limbs.162 Horses can become recumbent due to profound weakness. Cranial nerves are frequently abnormal with weakness of the tongue, muzzle deviation, and head tilt the most common abnormalities reported (Figure 3).162 Dysphagia has been reported with choking as a sequela.189 A cauda equina syndrome characterized by stranguria and rectal impaction is infrequently reported.162

Figure 3 Horse naturally infected with WNV exhibiting multiple cranial nerve (CN) abnormalities. This is Day 3 of clinical signs and the horse is leaning to the left while muzzle is deviated to the left. This horse exhibits involvement of CN 7 and 8. The paresis of the muzzle involves the right CN 7, while the leaning (and head tilt) involves the left CN 8 denoting probable central vestibular disease, which is consistent with a lesion in the central pons/medulla, a common site where WNV is found in diseased horses. (Courtesy of M.T. Long)
Overall, the combination, severity and duration of clinical signs can be highly variable. After initial signs abate, about one-third of clinically affected horses experience an increase in severity of clinical signs within the first seven to 10 days of onset. About 30 per cent of the horses progress to complete paralysis of one or more limbs. Most of these horses are euthanized for humane reasons or die spontaneously.162, 174, 181, 182, 189 Horses that become recumbent often need aggressive supportive care. Once horses demonstrate significant improvement, full recovery within one to six months can be expected in 90 per cent of patients.2, 162, 169 Residual weakness and ataxia appear to be the main problems with long-term loss of the use of one or more limbs infrequently described. In addition, mild to moderate persistent fatigue upon exercise has also been noted.162
Neurological disease due to WNV has been reported in camels, alpacas, and llamas with limited extraneural disease.59, 221 Animals present with signs consistent with diffuse or multifocal disease involving the brainstem and/or spinal cord. Most of the clinical symptomology is similar to that in horses, with change in mentation, behaviour, tremors, ataxia, and weakness all being prominent features. Cranial nerve deficits reflect hindbrain involvement. Opisthotonus and torticollis are more commonly reported features. In the limited camelids reported in the literature, recovery is not described; however, antemortem testing is limited in availability and mainly relies on neutralizaton testing. Subclinical infection does occur based on seroprevalence studies in North Africa, the Canary Islands of Spain, and the Middle East with seropositivity ranging from 10-50 per cent depending on the region sampled.63, 82, 180
Dogs develop relatively low viraemias and are not considered potential reservoirs based on infections performed with both L1 and L2 viruses.8, 18, 166 In a mosquito inoculation study, virus was detected sporadically over the first 1 through 5 days of infection. Treatment with glucocorticoids for one month resulted in significantly higher titres (peak mean titre of 4.76) for five days, which could result in theorectical mosquito transmission.22, 166 In this latter study, no dog developed clinical signs or pathological changes associated with challenge. While experimental infection did not result in significant disease, there are case reports of naturally infected dogs presenting with fatal encephalitis, severe myocarditis and polyarthritis.8, 33, 109 Direct infection of the heart and particularly the kidney has been demonstrated by real-time polymerase chain reaction (PCR) and culture.28, 109 Gastrointestinal signs consisting of pain and diarrhoea have been reported and associated with hepatopathy.109 In this former report, the animal also developed immune-mediated thrombocytopenia and anaemia during the course of clinical disease.109
Cats are likely infected by mosquitoes; however, experimental infection demonstrates oral infection is possible. Thus predation on infected birds and small mammals likely is a source of infection and may account for why outdoor cats have higher seroprevalence rates.8, 90 Cats develop viraemia of approximately 3 to 5 days duration.8 Clinical signs have varied depending on reports; experimentally infected animals became lethargic, however, small numbers of animals were infected.8, 179 In one report that examined four cats with either encephalitis or menigitis, WNV antigen was detected by immunohistochemistry (IHC), but this finding could not be substantiated by real-time PCR.179
Reptiles have demonstrated natural exposure, but the most prominently affected species is the alligator.95, 128 Not only does this species demonstrate viraemias consistently above the threshold for virus transmission, the viraemia is prolonged up to two weeks. In naturally infected farmed alligators, clinical disease consists of oral fibronecrotic mucosal lesions, anorexia, depression, and some CNS signs consisting of tremors. In farms of high intensity breeding, young alligators are highly susceptible and most likely to succumb to infection. Snakes are the next most investigated reptile, and garter snakes (Thamnophis sirtalis) experience sudden death and widespread virus dissemination to mulitple organs with experimental infection.192 If infected animals do not succumb acutely, chronic wasting and weakness are the main clinical features.
Pathology
The pathological changes in birds have been extensively and well reviewed,71 and the most consistent changes described in the literature across species occur in the brain, heart, kidneys, spleen, and liver.71, 94, 97, 138, 139, 183 In general, histopathological changes in the brain consist primarily of lymphoplasmacytic and histiocytic meningoencephalitis composed of gliosis, glial nodules, and perivascular cuffing. Virus and inflammation are consistently found in the cerebellum, followed by the cerebrum, midbrain, and hindbrain. Raptors and owls can have more neuronal necrosis, and inflammation of the peripheral nerves has been documented.137 In birds with high viraemia, such as corvids, vasculitis is a hallmark of infection in the brain.138 Cardiac involvement is primarily composed of myocardial necrosis and degeneration with widespread haemorrhage.71, 94, 97, 138, 139, 183 With time, mineralization and fibrosis are common. A lymphoplasmacytic and histiocytic interstitial nephritis with tubular epithelial cell necrosis or degeneration occurs in the kidneys.71, 94, 97, 138, 139, 183 Some degree of vasculitis is present depending on species of birds. Antigen is present in high amounts in epithelial cells, glomerular cells, inflammatory cells, endothelial cells, and even fibroblasts. Virus is commonly found in these lesions as well as myocytes, endothelial cells, and smooth muscle cells of arteries. Necrosis of hepatocytes and Kupffer cells of the liver, enterocytes and crypt cells of the intestine, mononuclear cells of the spleen, thymus, and bone marrow is also common.71, 94, 97, 138, 139, 183 In all of these organs, haemorrhages and vasculitis can be observed. There can be widespread necrosis of cells of the endocrine system with lesions in the pancreas, thyroid, and adrenal glands.71, 94, 97, 138, 139, 183 Virus is also detected in muscles and skin.71, 94, 97, 138, 139, 183 The quill and pulp of feathers are infected and abnormalities of the pulp can cause abnormal moults. Less commonly described are lesions of the eyes that consist of inflammatory infiltrates of the retina, vasculitis, and retinal necrosis.71, 94, 97, 138, 139, 183
Complete blood count (CBC) and serum biochemistry profiles of horses with clinical signs of WNV are usually normal. Low serum sodium is a frequent finding, which may be centrally mediated by hypothalamic disfunction since the thalamus and hypothalamus are common sites of infection.162 Cerobrospinal fluid (CSF) findings may be equivocal or be nonspecific for viral infection. Differential cell counts in CSF in affected horses usually have increased mononuclear cell populations (Figure 4).212 Protein concentrations are frequently elevated (N 70 mg/dL) and the colour of the fluid can be mildly xanthochromic.
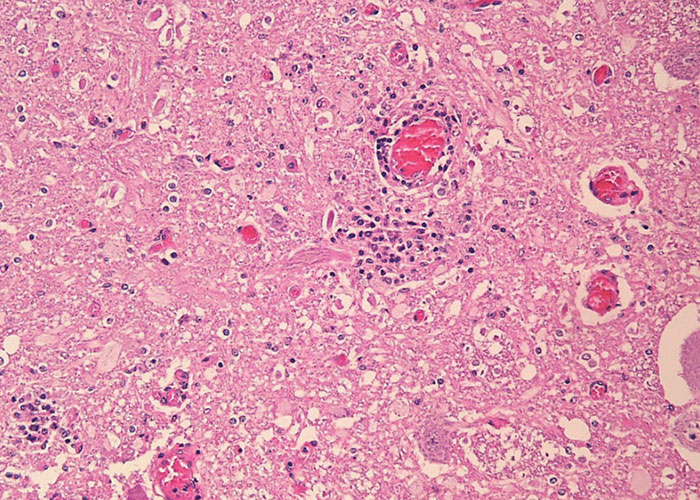
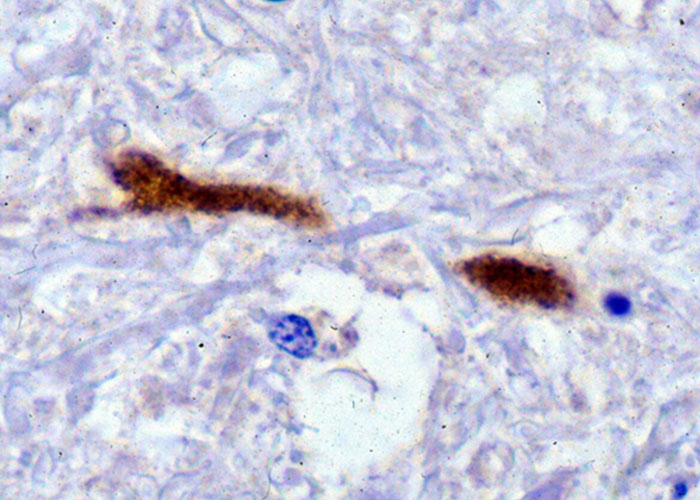
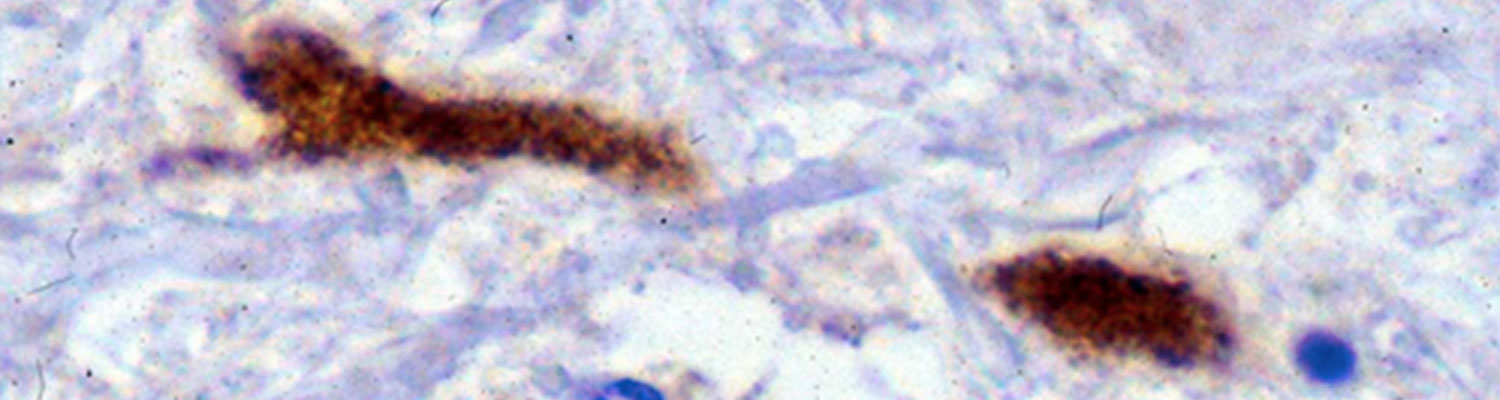
In horses, gross lesions are limited to the CNS and are characterized by mild to moderate meningeal hyperaemia, subdural exudates with fibrin tags, and areas of haemorrhage and malacia on cut sections of the brain stem and spinal cord (Figure 5).34, 35 These haemorrhages can be confluent over a segment of spinal cord and are noticeable mostly in the thalamus, pons, and medulla of the brain.29 Histologically, there is mild to severe non-suppurative polioencephalomyelitis characterized by multifocal, moderate to severe perivascular glial cell proliferation, infiltration of lymphocytes, some neutrophils, and macrophages with red blood cells. Gliosis and glial nodule formation occur with neuronal degeneration and necrosis (Figure 6). A prominent feature in horses is the presence of low amounts of virus in the face of life-ending clinical signs (Figure 7). Extensive populations of glial cells compared to lower numbers of infiltrating T cells are present in the CNS lesions. T cell phenotypes are highly mixed consisting of both CD4+ and CD8+ T cells.49 Axonal swelling and spheroid formation are frequently observed.29 Because WNV is frequently co-circulating with alphaviruses in the Americas (see Equine encephalitides caused by alphaviruses in the Western Hemisphere), close attention to localization of lesions is important for differential diagnosis. The pathology in the CNS of alphavirus infections is quite different in its localization (cerebrum to hindbrain) and cellular infiltration of the neuropil with numerous neutrophils that are non-degenerate. In other large hoof stock, WNV exhibits a tropism for the CNS, causing encephalitic changes similar to those in horses.59, 63, 82, 167, 180-182, 189 Lymphoplasmacytic meningoencephalitis is usually described for goats, sheep, and camelids. The most striking difference between infection of these latter species and horses is the high amount of virus detected in these animals at post mortem. An abundance of virus can be found in absence of inflammation in the Purkinje cells of the cerebellum in sheep.167 In dogs, CNS infection consists in general of lymphoplasmacytic polioencephalomyelitis or nonsuppurative meningoencephalitis. Cardiac lesions are not uncommonly reported and lymphohistiocytic necrotizing myocarditis is the primary feature. Renal and hepatic lesions consist mainly of necrosis and less frequently necrosis of the pancreas and intestinal epithelium has been documented. Polyarthritis has also been reported.33 Like birds, alligators and other reptiles can have inflammatory infiltrates and necrosis in many organs.
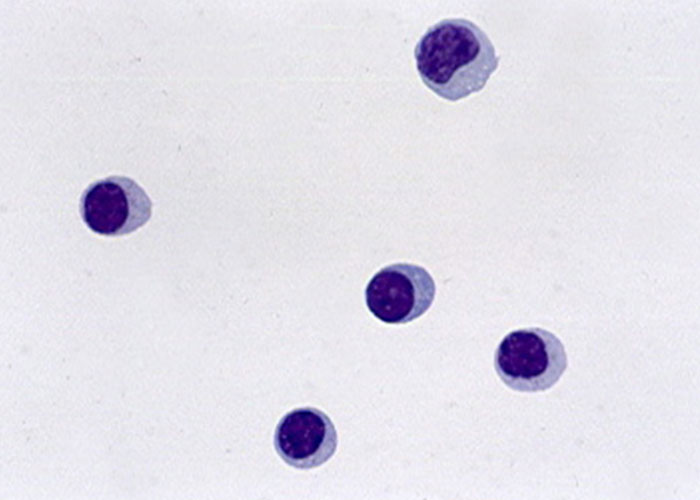
Figure 4 Wright-Giemsa staining of the cerebrospinal fluid of a horse with WNV infection. Cells are typically mononuclear and composed of both monocytes and lymphocytes.
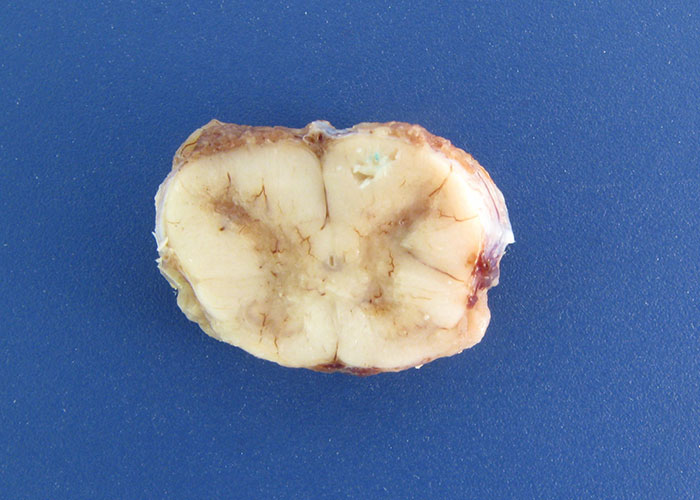
Figure 5 Cross section of the cervical spinal cord of a horse that was euthanized due to confirmed West Nile virus infection. Areas of congestion and haemorrhage are present with most of the lesions in the grey matter. There is loss of distinction between white and grey matter indicating some degree of malacia.
Diagnosis and differential diagnoses
Premortem diagnosis and differential conditions are confined in this discussion to horses, since in general, the diagnosis in most other species is at post-mortem. Consideration of WNV infection in a horse begins with assessment of whether the horse meets the case definition based on clinical signs and whether the horse resides in an area in which WNV has been confirmed in the current calendar year in mosquitoes, birds, humans, or horses, and vaccination history. Most horses that are clinically apparent have an IgM titre that remains detectable for four to six weeks with the capture ELISA, which has high sensitivity and specificity (> 88-90 per cent). In horses, the IgM response rises sharply during the first few days of clinical neurological signs.115 Measurement of IgM is performed at a single dilution of 1:400 and the detection of IgM antibody in CSF (if available) at a dilution of 1:2.147 Early work done after the US WNV encroachment on vaccinated horses that were tested on a weekly basis indicated limited detection of IgM at 1:400161, but work performed in Europe demonstrated positive IgM in 13/66 horses post-vaccination.40, 84 Thus, recent vaccination within the window of exposure will confound this test for diagnosis of WNV. Although ELISA is a highly specific and sensitive method for diagnosis, there is cross reactivity of the IgM between flaviviruses (WNV, Japanese encephalitis virus, tick-borne encephalitis virus, and Usutu virus).88 In non-vaccinated horses, a four-fold rise in paired neutralizing antibody titres is confirmatory of a diagnosis of WNV infection if there is no recent vaccination. Vaccination induces formation of neutralizing antibody to the E protein of the virus, which likely confounds interpretation of the plaque reduction virus neutralization test (PRNT); thus, the reliance on the PRNT for confirmatory serological diagnosis of WNV in horses has diminished. When used in vaccinated populations, comparison of side-by-side measured endpoint dilutions is essential and a four-fold change between the first and second sample indicates recent exposure. Serum neutralizing antibody has been measured in one of two formats, as a plaque reduction (large well) or microwell. A commonly used screening test is the haemagglutination inhibition (HI) test, which uses red blood cells coated with virus to detect antibody. This test should not be used for diagnosis in horses since it detects both IgM and IgG and can give false positive reactions due to nonspecific haemagglutinins.
Because WNV is usually a reportable disease to agricultural authorities and ministries in many countries, formal case definitions exist and practitioners should strive to test sera and perform necropsies on horses suspected of viral encephalitis to confirm disease. Therefore, the equine case definitions for WNV in the Veterinary Services of the United States Department of Agriculture (USDA-VS) and the European Union are based on similar test formats; however, for surveillance purposes, USDA-VS differentiates between presumptive and confirmed WNV diseased horses. At present, a horse with clinical signs (located in or recently visited an area of activity) and is only positive for serum neutralizing antibody is deemed a presumptive case since exposure may not have been concurrent with clinical signs, and thus a different aetiologic agent may be present. A confirmed case of WNV is defined as a horse with clinical signs and 1) detection of WNV IgM in serum or cerebrospinal fluid (CSF), or 2) an associated four-fold or greater rise in PRNT or IgG antibody, or 3) detection of WNV by virus isolation or molecular methods in brain, blood, or CSF.
Post-mortem confirmatory testing relies on detection of antigen by immunohistochemistry (IHC) or viral RNA by reverse transcription (rt)PCR. Unfortunately, almost all viral detection methods for WNV infection of horses can be unreliable. This is due to the relatively limited viral load of WNV, even in the face of mortal disease. This virus has high predilection for the grey matter of the brain of horses including the thalamus, hypothalamus, pons, and medulla (Figure 7).34, 152 Testing of these areas of the brain is recommended to increase the probability of detecting virus. These methods have excellent sensitivity in most species other than the horse and antigen or viral RNA is readily detected in histologically lesions. Since virus load is high in most birds that succumb to WNV, virus is detected in multiple organ tissues by PCR and IHC. The most common tissue requested and tested in US public health laboratories is kidney, although WNV can be readily detected in heart, spleen, and brain. Kidney is a commonly requested organ due to safety reasons; most of the occupational exposures from birds have been from dissection and penetration of skin by bones during necropsy. In the more uncommon species that succumb to WNV such as alpaca, dogs, and small ruminants, usually the disease is fulminating and virus is detected by either rtPCR or IHC. In the US, the CDC protocol should be utilized for consistent reporting purposes and this utilizes two primer sets.101 These primers work well on formalin-fixed and paraffin-embedded (FFPE) tissue, allowing for detection of virus directly where histopathological changes are most pronounced.152 West Nile virus can be isolated or detected in brain tissue by the use of cell culture.
The differential diagnoses in horses include rabies, alphavirus infection (i.e. Eastern, Western and Venezuelan equine encephalomyelitis), other flaviviruses including tick-borne encephalitis virus and Usutu virus, equid herpesvirus 1 infection, equine encephalosis virus, Borna disease, tetanus, and botulism. Non-infectious differentials include leukoencephalomalacia caused by Fusarium verticillioides (moniliforme), various plant poisonings such as Zygadenus and Solanum spp., and hepatoencephalopathy. Trauma especially that induces ataxia or dysphagia should also be considered and these injuries specifically include fractures of the basisphenoid and vertebrae. Additionally, when clinical signs are limited to the 7th and 8th cranial nerves, endoscopy and radiographs are recommended to rule out temporohyoid osteopathy.
Control
Vaccines have been developed for protection against WNV in horses. While not the focus of this chapter, vaccine efficacy has been examined in multiple avian species ranging from minimal to over 85 per cent protection achieved depending on bird species and vaccine candidate.83 Several vaccines have been developed, tested, licensed, and are currently marketed for use in horses. There are two whole virus-killed vaccines, one recombinant canary pox vaccine, and one inactivated flavivirus chimera vaccine, and several of these formulations are available globally, depending on the market.21, 130, 140, 182, 186 Several professional, public, and academic electronic websites update the marketed vaccines and because of the constant reorganization of animal vaccine producers, referral to these sites is recommended and local agricultural authorities and pharmaceutical agencies should be consulted for various in-country information regarding what vaccines are licensed locally (Table 1). The recommendations for vaccination are similar. After six months of age, all of the vaccines require an initial two dose series given three to four weeks apart. Any horse with doubtful history of WNV vaccination should undergo a full vaccination series if moved to a WNV endemic area. The current vaccines are labelled somewhat differently for foals, but all foals should receive a three-dose series irrespective of labelling. The first two doses should be administered four weeks apart and the third is best given 60 days after the second dose. However, foals from multiparous, fully vaccinated mares should be immunized at six months of age. For foals from partially vaccinated mares, or those with an unknown vaccine history, vaccination should commence at four months of age. All young horses should receive three doses of vaccines during the first three years of life in areas where the mosquito season is active for more than six months annually. All yearlings should receive their first injection for the year before March and a second injection by May in the Northern Hemisphere. This schedule should be followed in the Southern Hemisphere at times in the year corresponding to the appropriate onset of seasonal arbovirus activity in each respective location. All vaccines are labelled for one annual booster; however, in tropical and subtropical climates, where seasonal activity is throughout the year, six-month intervals are recommended for all horses over four years of age. The canary pox vaccine prevented viraemia in WNV experimental infection of domestic dogs and cats.87 This vaccine and the killed vaccine are also considered efficacious in camelids.100 Similar vaccination schedules are recommended in these species.
Many horses can become intolerant of frequent vaccination. Alternative approaches may be required, such as measurement of serum neutralizing antibody yearly before determining vaccination interval. There are no known data that indicate how long horses are protected past one year when fully immunized. New vaccines may be on the horizon, an example being a recombinant vaccine that immunizes against the EIII which, combined with equine CD40L, demonstrates high antigenicity.112 Another promising candidate is the Newcastle disease virus-vectored WNV vaccine. This vaccine elicited humoral immune responses in horses as well as birds, thus allowing for a multispecies vaccine platform deployable during epidemics.215 “Vaccine breaks” have been difficult to fully substantiate due to difficulty in obtaining an authentic vaccine history. Anecdotally, based on a decade of arboviral consulting by the author, new ownership without previous adequate vaccination, incomplete primary immunization, and lack of annual consistency of WNV vaccination have been commonly found as an underlying issue when WNV disease manifests in the “vaccinated” horse.
Table 1 Useful electronic resources for vaccination guidelines against WNV
| Title | Organization | URL |
|---|---|---|
| Vaccination Guidelines | American Association of Equine Practitioners | |
| Vaccination Equi-Planner Healthcare Tool | University of Guelph | |
| West Nile Virus Risk Assessment Tool | European Centre for Disease Prevention and Control | |
| EPAR Summary for the Public | European Medicines Agency: Equilis West Nile | https://www.ema.europa.eu/en/documents/overview/equilis-west-nile-epar-summary-public_en.pdf |
Measures that can be taken to lessen mosquito activity on farms include limitation of the use of lights, especially in the evening and at sunrise, and reducing breeding sites (e.g. removal of standing water containers, used tires, burn piles with cans and containers, and manure storage pits). Other methods include minimizing stagnation of water through aeration of ponds and provision of mosquito abatement to standing water such as stocking with fish and using natural antilarvicides, such as Bacillus thuringiensis. Ultimately, areas and buildings may require periodic application of adulticides through spraying and fogging. Local mosquito control experts can be consulted and can provide a comprehensive programme of abatement to decrease mosquito substantially.
References
- AMBROSE, R. L. & MACKENZIE, J. M., 2013. ATF6 signaling is required for efficient West Nile virus replication by promoting cell survival and inhibition of innate immune responses. Journal of Virology, 87, 2206-2214.
- ANDREADIS, T. G., 2012. The contribution of Culex pipiens complex mosquitoes to transmission and persistence of West Nile virus in North America. Journal of the American Mosquito Control Association, 28, 137-151.
- ANDREADIS, T. G., ANDERSON, J. F., VOSSBRINCK, C. R. & MAIN, A. J., 2004. Epidemiology of West Nile virus in Connecticut, a five-year analysis of mosquito data 1999-2003. Vector-Borne and Zoonotic Diseases, 4, 360-378.
- ANEZ, G., GRINEV, A., CHANCEY, C., BALL, C., AKOLKAR, N., LAND, K. J., WINKELMAN, V., STRAMER, S. L., KRAMER, L. D. & RIOS, M., 2013. Evolutionary dynamics of West Nile virus in the United States, 1999-2011, phylogeny, selection pressure and evolutionary time-scale analysis. PLoS Neglected Tropical Diseases, 7, e2245.
- ANONYMOUS., 2016. List of all mosquito species in which West Nile virus has been detected 1999-2016. In, DoV-BD National Center for Emerging and Zoonotic Infectious Diseases editor. Fort Collins, CO, Centers for Disease Control.
- ANONYMOUS., 2016. Species of dead birds in which West Nile has been detected, United States, 1999-2016. In: DoV-BD National Center for Emerging and Zoonotic Infectious Diseases editor. Fort Collins, CO, ArboNet Centers for Disease Control.
- APPERSON, C. S., HASSAN, H. K., HARRISON, B. A., SAVAGE, H. M., ASPEN, S. E., FARAJOLLAHI, A., CRANS, W., DANIELS, T. J., FALCO, R. C., BENEDICT, M., ANDERSON, M., MCMILLEN, L. & UNNASCH, T. R., 2004. Host feeding patterns of established and potential mosquito vectors of West Nile virus in the eastern United States. Vector-Borne and Zoonotic Diseases, 4, 71-82.
- AUSTGEN, L. E., BOWEN, R. A., BUNNING, M. L., DAVIS, B. S., MITCHELL, C. J. & CHANG, G. J., 2004. Experimental infection of cats and dogs with West Nile virus. Emerging Infectious Diseases, 10, 82-86.
- BAKONYI, T., HUBÁLEK, Z., RUDOLF, I. & NOWOTNY, N., 2005. Novel flavivirus or new lineage of West Nile virus, central Europe. Emerging Infectious Diseases, 11, 225-231.
- BAKONYI, T., IVANICS, E., ERDÉLYI, K., URSU, K., FERENCZI, E., WEISSENBÖCK, H. & NOWOTNY, N., 2006. Lineage 1 and 2 strains of encephalitic West Nile virus, central Europe. Emerging Infectious Diseases, 12, 618-623.
- BALENGHIEN, T., VAZEILLE, M., GRANDADAM, M., SCHAFFNER, F., ZELLER, H., REITER, P., SABATIER, P., FOUQUE, F. & BICOUT, D. J., 2008. Vector competence of some French Culex and Aedes mosquitoes for West Nile virus. Vector-Borne and Zoonotic Diseases, 8, 589-595.
- BARRERA, R., HUNSPERGER, E., MUÑOZ-JORDÁN, J. L., AMADOR, M., DIAZ, A., SMITH, J., BESSOFF, K., BELTRAN, M., VERGNE, E., VERDUIN, M., LAMBERT, A. & SUN, W., 2008. First isolation of West Nile virus in the Caribbean. American Journal of Tropical Medicine and Hygiene, 78, 666-668.
- BARZON, L., PACENTI, M., FRANCHIN, E., SQUARZON, L., LAVEZZO, E., TOPPO, S., MARTELLO, T., CATTAI, M., CUSINATO, R. & PALU, G., 2012. Novel West Nile virus lineage 1a full genome sequences from human cases of infection in north-eastern Italy, 2011. Clinical Microbiology and Infection, 18, e541-e544.
- BARZON, L., PAPA, A., LAVEZZO, E., FRANCHIN, E., PACENTI, M., SINIGAGLIA, A., MASI, G., TREVISAN, M., SQUARZON, L., TOPPO, S., PAPADOPOULOU, E., NOWOTNY, N., ULBERT, S., PIRALLA, A., ROVIDA, F., BALDANTI, F., PERCIVALLE, E. & PALU, G., 2015. Phylogenetic characterization of Central/Southern European lineage 2 West Nile virus, analysis of human outbreaks in Italy and Greece, 2013-2014. Clinical Microbiology and Infection, 21(12).
- BEASLEY, D. W., 2005. Recent advances in the molecular biology of west nile virus. Current Molecular Medicine, 5, 835-850.
- BHUVANAKANTHAM, R. & CHEONG YK, N. M. L., 2010. West Nile virus capsid protein interaction with importin and HDM2 protein is regulated by protein kinase C-mediated phosphorylation. Microbes and Infection, 12(8-9), 615-625.
- BISCHOF, R. & ROGERS, D., 2005. Serologic survey of select infectious diseases in coyotes and raccoons in Nebraska. Journal of Wildlife Diseases, 41, 787-791.
- BLACKBURN, N. K., REYERS, F., L., B. W. & SHEPHERD, A. J., 1989. Susceptibility of dogs to West Nile virus, a survey and pathogenicity trial. Journal of Comparative Pathology, 100, 59-66.
- BLAKELY, P. K., KLEINSCHMIDT-DEMASTERS, B. K., TYLER, K. L. & IRANI, D. N., 2009. Disrupted glutamate transporter expression in the spinal cord with acute flaccid paralysis caused by West Nile virus infection. Journal of Neuropathology & Experimental Neurology, 68, 1061-1072.
- BOURGEOIS, M. A., DENSLOW, N. D., SEINO, K. S., BARBER, D. S. & LONG, M. T., 2011. Gene Expression Analysis in the Thalamus and Cerebrum of Horses Experimentally Infected with West Nile Virus. PLoS One, 6(10).
- BOWEN, R. A., BOSCO-LAUTH, A., SYVRUD, K., THOMAS, A., MEINERT, T. R., LUDLOW, D. R., COOK, C., SALT, J. & ONS, E., 2014. Protection of horses from West Nile virus Lineage 2 challenge following immunization with a whole, inactivated WNV lineage 1 vaccine. Vaccine, 32, 5455-5459.
- BOWEN, R. A., ROUGE, M. M., SIGER, L., MINKE, J. M., NORDGREN, R., KARACA, K. & JOHNSON, J., 2006. Pathogenesis of West Nile virus infection in dogs treated with glucocorticoids. American Journal of Tropical Medicine and Hygiene, 74, 670-674.
- BRAULT, A. C., HUANG, C. Y. H., LANGEVIN, S. A., KINNEY, R. M., BOWEN, R. A., RAMEY, W. N., PANELLA, N. A., HOLMES, E. C., POWERS, A. M. & MILLER, B. R., 2007. A single positively selected West Nile viral mutation confers increased virogenesis in American crows. Nature Genetics, 39, 1162-1166.
- BRAULT, A. C., LANGEVIN, S. A., BOWEN, R. A., PANELLA, N. A., BIGGERSTAFF, B. J., MILLER, B. R. & KOMAR, N., 2004. Differential virulence of West Nile strains for American crows. Emerging Infectious Diseases, 10, 2161-2168.
- BRIEN, J. D., DAFFIS, S., LAZEAR, H. M., CHO, H., SUTHAR, M. S., GALE, M. & DIAMOND, M. S., 2011. Interferon regulatory factor-1 (IRF-1) shapes both innate and CD8(+) T cell immune responses against West Nile virus infection. PLoS Pathogens, 7, e1002230.
- BRINTON, M. A., 1981. Genetically controlled resistance to flavivirus and lactate-dehydrogenase-elevating virus-induced disease. Current Topics in Microbiology and Immunology, 92, 1-14.
- BRINTON, M. A., 2002. The molecular biology of West Nile Virus, a new invader of the western hemisphere. Annual Review of Microbiology, 56, 371-402.
- BUCKWEITZ, S., KLEIBOEKER, S., MARIONI, K., RAMOS-VARA, J., ROTTINGHAUS, A., SCHWABENTON, B. & JOHNSON, G., 2003. Serological, reverse tanscriptase-polymerase chain reaction, and immunohistochemical detection of West Nile virus in a clinicaly affected dog. Journal of Veterinary Diagnostic Investigation, 15, 324-329
- BUERGELT, C. & DEL PIERO, F., 2014. Diseases of the Nervous System and Peripheral Nerves. In: BUERGELT, C. & DEL PIERO, F., (eds). Color Atlas of Equine Pathology. Hoboken, NJ, Wiley Blackwell, John Wiley & Sons, Inc, 63-100.
- BUNNING, M. L., BOWEN, R. A., CROPP, C. B., SULLIVAN, K. G., DAVIS, B. S., KOMAR, N., GODSEY, M. S., BAKER, D., HETTLER, D. L., HOLMES, D. A., BIGGERSTAFF, B. J. & MITCHELL, C. J., 2002. Experimental infection of horses with West Nile virus. Emerging Infectious Diseases, 8, 380-386.
- CAILLY, P., BALENGHIEN, T., EZANNO, P., FONTENILLE, D., TOTY, C. & TRAN, A., 2011. Role of the repartition of wetland breeding sites on the spatial distribution of Anopheles and Culex, human disease vectors in southern France. Parasites and Vectors, 4, 65.
- CALISTRI, P., GIOVANNINI, A., HUBALEK, Z., IONESCU, A., MONACO, F., SAVINI, G. & LELLI, R., 2010. Epidemiology of west nile in europe and in the mediterranean basin. Open Virology, 4, 29-37.
- CANNON, A. B., LUFF, J. A., BRAULT, A. C., MACLACHLAN, N. J., CASE, J. B., GREEN, E. N. G. & SYKES, J. E., 2006. Acute encephalitis, polyarthritis, and myocarditis associated with West Nile virus infection in a dog. Journal of Veterinary Internal Medicine, 20, 1219-1223.
- CANTILE, C., DEL PIERO, F., DI GUARDO, G. & ARISPICI, M., 2001. Pathologic and immunohistochemical findings in naturally occuring West Nile virus infection in horses. Veterinary Pathology, 38, 414-421.
- CANTILE, C., DI GUARDO, G., ELENI, C. & ARISPICI, M., 2000. Clinical and neuropathological features of West Nile virus equine encephalomyelitis in Italy. Equine Veterinary Journal, 32, 31-35.
- CAPELLI, G., RAVAGNAN, S., MONTARSI, F., FABRIZIO, S., CAZZIN, S., BONFANTI, L., DI, G., PORTANTI, O., MULATTI, P., MONNE, I., CATTOLI, G., CESTER, G., RUSSO, F., SAVINI, G. & MARANGON, S., 2013. Further evidence of lineage 2 West Nile Virus in Culex pipiens of North-Eastern Italy. Veterinaria Italiana, 49, 263-268.
- CARSTENSEN, M., GIUDICE, J., HILDEBRAND, E., DUBEY, J., ERB, J., STARK, D., HART, J., BARBER-MEYER, S., MECH, L., WINDELS, S. & EDWARDS, A., 2017. A serosurvey of diseases of free-ranging gray wolves (canis lupus) in minnesota, USA. Journal of Wildlife Diseases, 53, 459-471.
- CHASKOPOULOU, A., DOVAS, C., CHAINTOUTIS, S., BOUZALAS, I., ARA, G. & PAPANASTASSOPOULOU, M., 2011. Evidence of enzootic circulation of West Nile virus (Nea Santa-Greece-2010, lineage 2), Greece, May to July 2011. Eurosurveillance, 16(31).
- CHEVALIER, V., TRAN, A. & DURAND, B., 2013. Predictive modeling of West Nile virus transmission risk in the Mediterranean Basin, how far from landing? International Journal of Environmental Research and Public Health, 11, 67-90.
- CHOI, K. S., KO, Y. J., NAH, J. J., KIM, Y. J., KANG, S. Y., YOON, K. J. & JOO, Y. S., 2007. Monoclonal antibody-based competitive enzyme-linked immunosorbent assay for detecting and quantifying West Nile virus-neutralizing antibodies in horse sera. Clinical and Vaccine Immunology, 14, 134-138.
- CHU, J. J. & NG, M. L., 2004. Infectious entry of West Nile virus occurs through a clathrin-mediated endocytic pathway. Journal of Virology, 78, 10543-10555.
- CHU, J. J., RAJAMANONMANI, R., LI, J., BHUVANAKANTHAM, R., LESCAR, J. & NG, M. L., 2005. Inhibition of West Nile virus entry by using a recombinant domain III from the envelope glycoprotein. Journal of General Virology, 86(2), 405-412.
- CICCOZZI, M., PELETTO, S., CELLA, E., GIOVANETTI, M., LAI, A., GABANELLI, E., ACUTIS, P. L., MODESTO, P., REZZA, G., PLATONOV, A. E., LO, P. A. & ZEHENDER, G., 2013. Epidemiological history and phylogeography of West Nile virus lineage 2. Infection, Genetics and Evolution, 17, 46-50.
- CIOTA, A. T. & KRAMER, L. D., 2013. Vector-virus interactions and transmission dynamics of West Nile virus. Viruses, 5, 3021-3047.
- CLARKE, P., LESER, J. S., BOWEN, R. A. & TYLER, K. L., 2014. Virus-induced transcriptional changes in the brain include the differential expression of genes associated with interferon, apoptosis, interleukin 17 receptor A, and glutamate signaling as well as flavivirus-specific upregulation of tRNA synthetases. MBio, 5, e00902-00914.
- CRUZ, L., CARDENAS, V. M., ABARCA, M., RODRIGUEZ, T., REYNA, R. F., SERPAS, M. V., FONTAINE, R. E., BEASLEY, D. W., DA ROSA, A. P., WEAVER, S. C., TESH, R. B., POWERS, A. M. & SUAREZ-RANGEL, G., 2005. Short report, serological evidence of West Nile virus activity in El Salvador. American Journal of Tropical Medicine and Hygiene, 72, 612-615.
- DAVIS, C. T., EBEL, G. D., LANCIOTTI, R. S., BRAULT, A. C., GUZMAN, H., SIIRIN, M., LAMBERT, A., PARSONS, R. E., BEASLEY, D. W. C., NOVAK, R. J., ELIZONDO-QUIROGA, D., GREEN, E. N., YOUNG, D. S., STARK, L. M., DREBOT, M. A., ARTSOB, H., TESH, R. B., KRAMER, L. D. & BARRETT, A. D. T., 2005. Phylogenetic analysis of North American West Nile virus isolates, 2001-2004, Evidence for the emergence of a dominant genotype. Virology, 342, 252-265.
- DECARLO, C., OMAR, A., HAROUN, M., BIGLER, L., BIN RAIS, M., ABU, J., OMAR, A. & MOHAMMED, H., 2017. Potential Reservoir and Associated Factors for West Nile Virus in Three Distinct Climatological Zones. Vector-Borne and Zoonotic Diseases, 17, 709-713.
- DELCAMBRE, G. H., LIU, J., STREIT, W. J., SHAW, G. P. J., VALLARIO, K., HERRINGTON, J., WENZLOW, N., BARR, K. L. & LONG, M. T., 2017. Phenotypic characterisation of cell populations in the brains of horses experimentally infected with West Nile virus. Equine Veterinary Journal, 49, 815-820.
- DIAMOND, M., PIERSON, T. & FREMONT, D., 2008. The structural immunology of antibody protection against West Nile virus. Immunological Reviews, 225, 212-225.
- DIAMOND, M. S. & KLEIN, R. S., 2004. West Nile virus, crossing the blood-brain barrier. Nature Medicine, Vol. 10. United States, 1294-1295.
- DIAZ-BADILLO, A., BOLLING, B. G., PEREZ-RAMIREZ, G., MOORE, C. G., MARTINEZ-MUNOZ, J. P., PADILLA-VIVEROS, A. A., CAMACHO-NUEZ, M., DIAZ-PEREZ, A., BEATY, B. J. & MUNOZ, M. L., 2011. The distribution of potential West Nile virus vectors, Culex pipiens pipiens and Culex pipiens quinquefasciatus (Diptera, Culicidae), in Mexico City 39. Parasites and Vectors, 4, 70.
- DOCHERTY, D., SAMUEL, M., EGSTAD, K., GRIFFIN, K., NOLDEN, C., KARWAL, L. & IP, H., 2009. Short Report, Changes in West Nile Virus Seroprevalence and Antibody Titers among Wisconsin Mesopredators 2003-2006. American Journal of Tropical Medicine and Hygiene, 81, 177-179.
- DOHERTY, J. A., 2006. Final report and recommendations from the National Notifiable Diseases Working Group. Canada Communicable Disease Report, 32, 211-225.
- DONADIEU, E., BAHUON, C., LOWENSKI, S., ZIENTARA, S., COULPIER, M. & LECOLLINET, S., 2013. Differential virulence and pathogenesis of West Nile viruses. Viruses, 5, 2856-2880.
- DONADIEU, E., LOWENSKI, S., SERVELY, J. L., LALOY, E., LILIN, T., NOWOTNY, N., RICHARDONS, J., ZIENTARA, S., LECOLLINET, S. & COULPIER, M., 2013. Comparison of neuropathology induced by two West Nile virus strains. PLoS One, 8, e84473.
- DOSSANTOS, C. N. D., POST, P. R., CARVALHO, R., FERREIRA, I. I., RICE, C. M. & GALLER, R., 1995. Complete nucleotide-sequence of yellow-fever virus-vaccine strains 17dd and 17d-213. Virus Research, 35, 35-41.
- DREBOT, M. A., LINDSAY, R., BARKER, I. K., BUCK, P. A., FEARON, M., HUNTER, F., SOCKETT, P. & ARTSOB, H., 2003. West Nile virus surveillance and diagnostics, A Canadian perspective. Canadian Journal of Infectious Diseases, 14, 105-114.
- DUNKEL, B., DEL PIERO, E., WOTMAN, K., JOHNS, I., BEECH, J. & WILKINS, P., 2004. Encephalomyelitis from West Nile flavivirus in 3 alpacas. Journal of Veterinary Internal Medicine, 18, 365-367.
- DUPUIS, A. P., MARRA, P. P., REITSMA, R., JONES, M. J., LOUIE, K. L. & KRAMER, L. D., 2005. Serologic evidence for West Nile virus transmission in Puerto Rico and Cuba. American Journal of Tropical Medicine and Hygiene, 73, 474-476.
- DURAND, B., CHEVALIER, V., POUILLOT, R., LABIE, J., MARENDAT, I., MURGUE, B., ZELLER, H. & ZIENTARA, S., 2002. West Nile virus outbreak in horses, southern France, 2000, results of a serosurvey. Emerging Infectious Diseases, 8, 777-782.
- DUTTON, C. J., QUINNELL, M., LINDSAY, R., DELAY, J. & BARKER, I. K., 2009. Paraparesis in a polar bear (Ursus maritimus) associated with West Nile virus infection. Journal of Zoo and Wildlife Medicine, 40, 568-571.
- EL-BAHNASAWY, M. M., KHATER, M. K. & MORSY, T. A., 2013. The mosquito borne West Nile virus infection, is it threating to Egypt or a neglected endemic disease? Journal of the Egyptian Society of Parasitology, 43, 87-102.
- ENSERINK, M., 1999. Groups race to sequence and identify New York virus. Science, 286(5438), 206-207.
- EPP, T., WALDNER, C., LEIGHTON, F. A., BERKE, O. & TOWNSEND, H. G., 2007. Seroprevalence and risk factors for infection with West Nile virus in Saskatchewan horses, 2003. Canadian Journal of Veterinary Research, 71, 256-263.
- EPP, T., WALDNER, C. & TOWNSEND, H. G., 2007. A case-control study of factors associated with development of clinical disease due to West Nile virus, Saskatchewan 2003. Equine Veterinary Journal, 39, 498-503.
- FARAJOLLAHI, A., GATES, R., CRANS, W. & KOMAR, N., 2004. Serologic evidence of West Nile virus and St. Louis encephalitis virus infections in white-tailed deer (Odocoileus virginianus) from New Jersey, 2001. Vector-Borne and Zoonotic Diseases, 4, 379-383.
- FARFAN-ALE, J. A., BLITVICH, B. J., MARLENEE, N. L., LORONO-PINO, M. A., PUERTO-MANZANO, F., GARCIA-REJON, J. E., ROSADO-PAREDES, E. P., FLORES-FLORES, L. F., ORTEGA-SALAZAR, A., CHAVEZ-MEDINA, J., CREMIEUX-GRIMALDI, J. C., CORREA-MORALES, F., HERNANDEZ-GAONA, G., MENDEZ-GALVAN, J. F. & BEATY, B. J., 2006. Antibodies to West Nile virus in asymptomatic mammals, birds, and reptiles in the Yucatan Peninsula of Mexico. American Journal of Tropical Medicine and Hygiene, 74, 908-914.
- FILIPE, A. & DEANDRADE, H., 1990. Arboviruses in the iberian peninsula. Acta Virologica, 34, 582-591.
- FROST, M. J., ZHANG, J., EDMONDS, J. H., PROW, N. A., GU, X., DAVIS, R., HORNITZKY, C., ARZEY, K. E., FINLAISON, D., HICK, P., READ, A., HOBSON-PETERS, J., MAY, F. J., DOGGETT, S. L., HANIOTIS, J., RUSSELL, R. C., HALL, R. A., KHROMYKH, A. A. & KIRKLAND, P. D., 2012. Characterization of virulent West Nile virus Kunjin strain, Australia, 2011. Emerging Infectious Diseases, 18, 792-800. doi: 10.1186/1297-9716-44-39.
- GAMINO, V. & HOFLE, U., 2013. Pathology and tissue tropism of natural West Nile viurs infection in birds: a review. Veterinary Record, 44, 30.
- GIBBONS, D., ERK, I., REILLY, B., NAVAZA, J., KIELIAN, M., REY, F. & LEPAULT, J., 2003. Visualization of the target-membrane-inserted fusion protein of Semliki Forest virus by combined electron microscopy and crystallography. Cell, 114, 573-583.
- GODDARD, L. B., ROTH, A. E., REISEN, W. K. & SCOTT, T. W., 2003. Vertical transmission of West Nile virus by three California Culex (Diptera , Culicidae) species. Journal of Medical Entomology, 40, 743-746.
- GODSEY, M. S., BLACKMORE, M. S., PANELLA, N. A., BURKHALTER, K., GOTTFRIED, K., HALSEY, L. A., RUTLEDGE, R., LANGEVIN, S. A., GATES, R., LAMONTE, K. M., LAMBERT, A., LANCIOTTI, R. S., BLACKMORE, C. G., LOYLESS, T., STARK, L., OLIVERI, R., CONTI, L. & KOMAR, N., 2005. West Nile virus epizootiology in the southeastern United States, 2001. Vector-Borne and Zoonotic Diseases, 5, 82-89.
- GUTHRIE, A., HOWELL, P., GARDNER, I., SWANEPOEL, R., NURTON, J., HARPER, C., PARDINI, A., GROENEWALD, D., VISAGE, C., HEDGES, J., BALASURIYA, U., CORNEL, A. & MACLACHLAN, N., 2003. West nile virus infection of Thoroughbred horses in South Africa (2000-2001). Equine Veterinary Journal, 35, 601-605.
- HANNA, S., PIERSON, T., SANCHEZ, M., AHMED, A., MURTADHA, M. & DOMS, R., 2005. N-linked glycosylation of west nile virus envelope proteins influences particle assembly and infectivity. Journal of Virology, 79, 13262-13274.
- HAYES, C. G., 2001. West Nile virus, Uganda, 1937, to New York City, 1999. Annals of the New York Academy of Sciences, 951, 25-37.
- HERNANDEZ-JOVER, M., ROCHE, S. & WARD, M. P., 2013. The human and animal health impacts of introduction and spread of an exotic strain of West Nile virus in Australia. Preventive Veterinary Medicine, 109(3-4), 186-204.
- HOFFMANN, L., MOUCHET, J., RAGEAU, J., HANNOUN, C., JOUBERT, L., OUDAR, J. & BEYTOUT, D., 1968. Epidemiology of the West Nile virus, study of an outbreak in Camargue. II. Outline of the physical, biological and human environment. Annales de l'Institut Pasteur (Paris), 114, 521-538.
- HUBÁLEK, Z., 2008. Mosquito-borne viruses in Europe. Parasitology Research, 103(1), S29-S43.
- HUBÁLEK, Z. & HALOUZKA, J., 1999. West Nile fever - a re-emerging mosquito-borne viral disease in Europe. Emerging Infectious Diseases, 5, 643-650.
- HURLBUT, H. S., RIZK, F., TAYLOR, R. M. & WORK, T. H., 1956. A study of the ecology of West Nile virus in Egypt. American Journal of Tropical Medicine and Hygiene, 5, 579-620.
- IYER, A. V. & KOUSOULAS, K. G., 2013. A revew of vaccine approaches for West Nile virus. International Journal of Environmental Research and Public Health, 10, 4200-4223.
- JOÓ, K., BAKONYI, T., SZENCI, O., SÁRDI, S., FERENCZI, E., BARNA, M., MALIK, P., HUBALEK, Z., FEHÉR, O. & KUTASI, O., 2017. Comparison of assays for the detection of West Nile virus antibodies in equine serum after natural infection or vaccination. Veterinary Immunology and Immunopathology, 183, 1-6.
- JUPP, P. G., 2001. The ecology of West Nile virus in South Africa and the occurrence of outbreaks in humans. Annals of the New York Academy of Sciences, 951, 143-152.
- KARABATSOS, L., 1985. International catalogue of arboviruses. San Antonio, Texas, American Society of Tropical Medicine and Hygiene.
- KARACA, K., BOWEN, R., AUSTGEN, L. E., TEEHEE, M., SIGER, L., GROSENBAUGH, D., LOOSEMORE, L., AUDONNET, J. C., NORDGREN, R. & MINKE, J. M., 2005. Recombinant canarypox vectored West Nile virus (WNV) vaccine protects dogs and cats against a mosquito WNV challenge. Vaccine, 23, 3808-3813.
- KHAN, E., BARR, K. L., FAROOQI, J. Q., PRAKOSO, D., ABBAS, A., KHAN, Z. Y., ASHI, S., IMTIAZ, K., AZIZ, Z., MALIK, F., LEDNICKY, J. A. & LONG, M. T., 2018. Human West Nile Virus Disease Outbreak in Pakistan, 2015-2016. Front Public Health, 6, 20.
- KHROMYKH, A. A. & WESTAWAY, E. G., 1994. COMPLETION OF KUNJIN VIRUS-RNA SEQUENCE AND RECOVERY OF AN INFECTIOUS RNA TRANSCRIBED FROM STABLY CLONED FULL-LENGTH CDNA. Journal of Virology, 68, 4580-4588.
- KILE, J., PANELLA, N., KOMAR, N., CHOW, C., MACNEIL, A., ROBBINS, B. & BUNNING, M., 2005. Serologic survey of cats and dogs during an epidemic of West Nile virus infection in humans. Journal of the American Veterinary Medical Association, 226, 1349-1353.
- KILPATRICK, A. M., DASZAK, P., JONES, M. J., MARRA, P. P. & KRAMER, L. D., 2006. Host heterogeneity dominates West Nile virus transmission. Biological Sciences, 273(1599), 2327-2333.
- KILPATRICK, A. M., KRAMER, L. D., CAMPBELL, S. R., ALLEYNE, E. O., DOBSON, A. P. & DASZAK, P., 2005. West Nile virus risk assessment and the bridge vector paradigm. Emerging Infectious Diseases, 11, 425-429.
- KILPATRICK, A. M., KRAMER, L. D., JONES, M. J., MARRA, P. P. & DASZAK, P., 2006. West Nile virus epidemics in North America are driven by shifts in mosquito feeding behavior. PLoS Biology, 4, e82.
- KILPATRICK, A. M., PETERS, R. J., DUPUIS, A. P., JONES, M. J., MARRA, P. P. & KRAMER, L. D., 2013. Predicted and observed mortality from vector-borne disease in small songbirds. Biological Conservation, 165, 79-85.
- KLENK, K., SNOW, J., MORGAN, K., BOWEN, R., STEPHENS, M., FOSTER, F., GORDY, P., BECKETT, S., KOMAR, N., GUBLER, D. & BUNNING, M., 2004. Alligators as West Nile virus amplifiers. Emerging Infectious Diseases, 10, 2150-2155.
- KOMAR, N. & CLARK, G. G., 2006. West Nile virus activity in Latin America and the Caribbean. Revista Panamericana de Salud Pública, 19, 112-117.
- KOMAR, N., LANGEVIN, S., HINTEN, S., NEMETH, N., EDWARDS, E., HETTLER, D., DAVIS, B., BOWEN, R. & BUNNING, M., 2003. Experimental infection of North American birds with the New York 1999 strain of West Nile virus. Emerging Infectious Diseases, 9, 311-322.
- KRAMER, L. D. & BERNARD, K. A., 2001. West Nile virus infection in birds and mammals. Annals of the New York Academy of Sciences, 951, 84-93.
- KUHN, R. J., ZHANG, W., ROSSMANN, M. G., PLETNEV, S. V., CORVER, J., LENCHES, E., JONES, C. T., MUKHOPADHYAY, S., CHIPMAN, P. R., STRAUSS, E. G., BAKER, T. S. & STRAUSS, J. H., 2002. Structure of dengue virus, implications for flavivirus organization, maturation, and fusion. Cell , 108, 717-725.
- KUTZLER, M. A., BAKER, R. J. & MATTSON, D. E., 2004. Humoral response to West Nile virus vaccination in alpacas and llamas. Journal of the American Veterinary Medical Association, 225, 414-416.
- LANCIOTTI, R. S., 2003. Molecular amplification assays for the detection of flaviviruses. Advances in Virus Research, 61, 67-99.
- LANCIOTTI, R. S., EBEL, G. D., DEUBEL, V., KERST, A. J., MURRI, S., MEYER, R., BOWEN, M., MCKINNEY, N., MORRILL, W. E., CRABTREE, M. B., KRAMER, L. D. & ROEHRIG, J. T., 2002. Complete genome sequences and phylogenetic analysis of West Nile virus strains isolated from the United States, Europe, and the Middle East. Virology, 298, 96-105.
- LANCIOTTI, R. S., ROEHRIG, J. T., DEUBEL, V., SMITH, J., PARKER, M., STEELE, K., CRISE, B., VOLPE, K. E., CRABTREE, M. B., SCHERRET, J. H., HALL, R. A., MACKENZIE, J. S., CROPP, C. B., PANIGRAHY, B., OSTLUND, E., SCHMITT, B., MALKINSON, M., BANET, C., WEISSMAN, J., KOMAR, N., SAVAGE, H. M., STONE, W., MCNAMARA, T. & GUBLER, D. J., 1999. Origin of the West Nile virus responsible for an outbreak of encephalitis in the northeastern United States. Science, 286(5448), 2333-2337.
- LAZEAR, H. M., DANIELS, B. P., PINTO, A. K., HUANG, A. C., VICK, S. C., DOYLE, S. E., GALE, M., KLEIN, R. S. & DIAMOND, M. S., 2015. Interferon-lambda restricts West Nile virus neuroinvasion by tightening the blood-brain barrier. Science Translational Medicine, 7, 284ra259.
- LAZEAR, H. M. & DIAMOND, M. S., 2015. New insights into innate immune restriction of West Nile virus infection. Current Opinion in Virology, 11, 1-6.
- LAZEAR, H. M., LANCASTER, A., WILKINS, C., SUTHAR, M. S., HUANG, A., VICK, S. C., CLEPPER, L., THACKRAY, L., BRASSIL, M. M., VIRGIN, H. W., NIKOLICH-ZUGICH, J., MOSES, A. V., GALE, M., FRÜH, K. & DIAMOND, M. S., 2013. IRF-3, IRF-5, and IRF-7 coordinately regulate the type I IFN response in myeloid dendritic cells downstream of MAVS signaling. PLoS Pathogens, 9, e1003118.
- LAZEAR, H. M., PINTO, A. K., RAMOS, H. J., VICK, S. C., SHRESTHA, B., SUTHAR, M. S., GALE, M. & DIAMOND, M. S., 2013. Pattern recognition receptor MDA5 modulates CD8+ T cell-dependent clearance of West Nile virus from the central nervous system. Journal of Virology, 87, 11401-11415.
- LEE, P., MUKHERJEE, S., EDELING, M., DOWD, K., AUSTIN, S., MANHART, C., DIAMOND, M., FREMONT, D. & PIERSON, T., 2013. The Fc Region of an Antibody Impacts the Neutralization of West Nile Viruses in Different Maturation States. Journal of Virology, 87, 13729-13740.
- LICHTENSTEIGER, C. A., HEINZ-TAHENY, K., OSBORNE, T. S., NOVAK, R. J., LEWIS, B. A. & FIRTH, M. L., 2003. West Nile virus encephalitis and myocarditis in a wolf and dog. Emerging Infectious Diseases, 9, 1303-1306.
- LIN, T., DOWD, K., MANHART, C., NELSON, S., WHITEHEAD, S. & PIERSON, T., 2012. A Novel Approach for the Rapid Mutagenesis and Directed Evolution of the Structural Genes of West Nile Virus. Journal of Virology, 86, 3501-3512.
- LINDENBACH, B. D., MURRAY, C. L., THIEL, H. J. & RICE, C. M., 2013. Flaviviridae. In: HOWLEY, P. M., COHEN, J. I. & GRIFFEN, D. E., (eds). Field's Virology. Philadelphia, Wolters Kluwer-Lippincott Williams & Wilkins.
- LIU, S. A., HAQUE, M., STANFIELD, B., ANDREWS, F. M., ROY, A. A. & KOUSOULAS, K. G., 2017. A recombinant fusion protein consisting of West Nile virus envelope domain III fused in-frame with equine CD40 ligand induces antiviral immune responses in horses. Veterinary Microbiology, 198, 51-58.
- LIU, W. J., WANG, X. J., CLARK, D. C., LOBIGS, M., HALL, R. A. & KHROMYKH, A. A., 2006. A single amino acid substitution in the West Nile virus nonstructural protein NS2A disables its ability to inhibit alpha/beta interferon induction and attenuates virus virulence in mice. Journal of Virology, 80, 2396-2404.
- LONG, M. T., GIBBS, E. P., MELLENCAMP, M. W., BOWEN, R. A., SEINO, K. K., ZHANG, S., BEACHBOARD, S. E. & HUMPHREY, P. P., 2007. Efficacy, duration, and onset of immunogenicity of a West Nile virus vaccine, live Flavivirus chimera, in horses with a clinical disease challenge model. Equine Veterinary Journal, 39, 491-497.
- LONG, M. T., JETER, W., HERNANDEZ, J., SELLON, D. C., GOSCHE, D., GILLIS, K., BILLE, E. & GIBBS, E. P., 2006. Diagnostic performance of the equine IgM capture ELISA for serodiagnosis of West Nile virus infection. Journal of Veterinary Internal Medicine, 20, 608-613.
- LOPES, H., REDIG, P., GLASER, A., ARMIEN, A. & WUNSCHMANN, A., 2007. Clinical findings, lesions, and viral antigen distribution in great gray owls (Strix nebulosa) and barred owls (Strix varia) with spontaneous West Nile virus infection. Avian Diseases, 51, 140-145.
- MALKINSON, M. & BANET, C., 2002. The role of birds in the ecology of West Nile virus in Europe and Africa. Current Topics in Microbiology and Immunology, 267, 309-322.
- MALKINSON, M., WEISMAN, Y., POKAMONSKI, S., KING, R. & DEUBEL, V., 2001. Intercontinental transmission of West Nile virus by migrating white storks. Emerging Infectious Diseases, 7(3), 540.
- MANN, B. R., MCMULLEN, A. R., SWETNAM, D. M., SALVATO, V., REYNA, M., GUZMAN, H., BUENO, R., DENNETT, J. A., TESH, R. B. & BARRETT, A. D., 2013. Continued evolution of West Nile virus, Houston, Texas, USA, 2002-2012. Emerging Infectious Diseases, 19, 1418-1427.
- MANN, R. A., FEGAN, M., O'RILEY, K., MOTHA, J. & WARNER, S., 2013. Molecular characterization and phylogenetic analysis of Murray Valley encephalitis virus and West Nile virus (Kunjin subtype) from an arbovirus disease outbreak in horses in Victoria, Australia, in 2011. Journal of Veterinary Diagnostic Investigation, 25, 35-44.
- MARKA, A., et al., 2013. West Nile virus state of the art report of MALWEST Project 12. International Journal of Environmental Research and Public Health, 10, 6534-6610.
- MARTIN-ACEBES, M. A., BLAZQUEZ, A. B., DE OYA, N. J., ESCRIBANO-ROMERO, E., SHI, P. Y. & SAIZ, J. C., 2013. A single amino acid substitution in the core protein of West Nile virus increases resistance to acidotropic compounds 49. PLoS One, 8, e69479.
- MAY, F. J., DAVIS, C. T., TESH, R. B. & BARRETT, A. D., 2011. Phylogeography of West Nile virus, from the cradle of evolution in Africa to Eurasia, Australia, and the Americas. Journal of Virology, 85, 2964-2974.
- MELIAN, E. B., EDMONDS, J. H., NAGASAKI, T. K., HINZMAN, E., FLODEN, N. & KHROMYKH, A. A., 2013. West Nile virus NS2A protein facilitates virus-induced apoptosis independently of interferon response. Journal of General Virology, 94(2), 308-313.
- MESNIL, M., PICCOLI, C., TIRABY, G., WILLECKE, K. & YAMASAKI, H., 1996. Bystander killing of cancer cells by herpes simplex virus thymidine kinase gene is mediated by connexins. Proceedings of the National Academy of Sciences of the United States of America (Journal), 93,1831-1835.
- MILLER, B. R., NASCI, R. S., GODSEY, M. S., SAVAGE, H. M., LUTWAMA, J. J., LANCIOTTI, R. S. & PETERS, C. J., 2000. First field evidence for natural vertical transmission of West Nile virus in Culex univittatus complex mosquitoes from Rift Valley province, Kenya. American Journal of Tropical Medicine and Hygiene, 62, 240-246.
- MILLER, D., SCHRECENGOST, J., MERRILL, A., KILGO, J., RAY, H., MILLER, K. & BALDWIN, C., 2009. Hematology, Parasitology, and Serology of Free-Ranging Coyotes (Canis latrans) from South Carolina. Journal of Wildlife Diseases, 45, 863-869.
- MILLER, D. L., MAUEL, M. J., BALDWIN, C., BURTLE, G., INGRAM, D., HINES, M. E. & FRAZIER, K. S., 2003. West Nile virus in farmed alligators. Emerging Infectious Diseases, 9, 794-799.
- MILLER, D. L., RADI, Z. A., BALDWIN, C. & INGRAM, D., 2005. Fatal West Nile virus infection in a white-tailed deer (Odocoileus virginianus). Journal of Wildlife Diseases, 41, 246-249.
- MINKE, J. M., SIGER, L., KARACA, K., AUSTGEN, L., GORDY, P., BOWEN, R., RENSHAW, R. W., LOOSMORE, S., AUDONNET, J. C. & NORDGREN, B., 2004. Recombinant canarypoxvirus vaccine carrying the prM/E genes of West Nile virus protects horses against a West Nile virus-mosquito challenge. Archives of Virology, 221-230.
- MIXÃO, V., BRAVO BARRIGA, D., PARREIRA, R., NOVO, M. T., SOUSA, C. A., FRONTERA, E., VENTER, M., BRAACK, L. & ALMEIDA, A. P., 2016. Comparative morphological and molecular analysis confirms the presence of the West Nile virus mosquito vector, Culex univittatus, in the Iberian Peninsula. Parasites and Vectors, 9, 601.
- MODIS, Y., OGATA, S., CLEMENTS, D. & HARRISON, S. C., 2003. A ligand-binding pocket in the dengue virus envelope glycoprotein. Proceedings of the National Academy of Sciences of the United States of America (Journal), 100, 6986-6991.
- MOLAEI, G., ANDREADIS, T. G., ARMSTRONG, P. M., ANDERSON, J. F. & VOSSBRINCK, C. R., 2006. Host feeding patterns of Culex mosquitoes and West Nile virus transmission, northeastern United States. Emerging Infectious Diseases, 12, 468-474.
- MORALES, M. A., BARRANDEGUY, M., FABBRI, C., GARCIA, J. B., VISSANI, A., TRONO, K., GUTIERREZ, G., PIGRETTI, S., MENCHACA, H., GARRIDO, N., TAYLOR, N., FERNANDEZ, F., LEVIS, S. & ENRÍA, D., 2006. West Nile virus isolation from equines in Argentina, 2006. Emerging Infectious Diseases, 12,1559-1561.
- MUGHINI-GRAS, L., MULATTI, P., SEVERINI, F., BOCCOLINI, D., ROMI, R., BONGIORNO, G., KHOURY, C., BIANCHI, R., MONTARSI, F., PATREGNANI, T., BONFANTI, L., REZZA, G., CAPELLI, G. & BUSANI, L., 2014. Ecological niche modelling of potential west nile virus vector mosquito species and their geographical association with equine epizootics in Italy 1. EcoHealth, 11, 120-132.
- MUKHOPADHYAY, S., KIM, B. S., CHIPMAN, P. R., ROSSMANN, M. G. & KUHN, R. J., 2003. Structure of West Nile virus. Science, 302(5643), 248.
- NEMETH, N. M., GOULD, D. H., BOWEN, R. A. & KOMAR, N., 2006. Natural and experimental West Nile virus infection in five species of raptors. Journal of Wildlife Diseases, 42, 1-13.
- NEMETH, N. M., HAHN, D. C., GOULD, D. H. & BOWEN, R. A., 2006. Experimental West Nile virus infection in Eastern Screech Owls (Megascops asio). Avian Diseases, 50, 252-258.
- NEMETH, N. M., THOMSEN, B. V., SPRAKER, T. R., BENSON, J. M., BOSCO-LAUTH, A. M., OESTERLE, P. T., BRIGHT, J. M., MUTH, J. P., CAMPBELL, T. W., GIDLEWSKI, T. L. & BOWEN, R. A., 2011. Clinical and pathologic responses of American Crows (Corvus brachyrhynchos) and fish crows (C ossifragus) to experimental West Nile vurs infection. Veterinary Pathology, 48, 1061-1074.
- NG, T., HATHAWAY, D., JENNINGS, N., CHAMP, D., CHIANG, Y. W. & CHU, H. J., 2003. Equine vaccine for West Nile virus. Developmental Biology (Basel), 114, 221-227.
- NYBAKKEN, G., NELSON, C., CHEN, B., DIAMOND, M. & FREMONT, D., 2006. Crystal structure of the West Nile virus envelope glycoprotein. Journal of Virology, 80,11467-11474.
- NYBAKKEN, G., OLIPHANT, T., JOHNSON, S., BURKE, S., DIAMOND, M. & FREMONT, D., 2005. Structural basis of West Nile virus neutralization by a therapeutic antibody. Nature, 437(7059), 764-768.
- OKAMOTO, T., SUZUKI, T., KUSAKABE, S., TOKUNAGA, M., HIRANO, J., MIYATA, Y. & MATSUURA, Y., 2017. Regulation of Apoptosis during Flavivirus Infection. Viruses, 9(9).
- OLALEYE, O., OMILABU, S., ILOMECHINA, E. & FAGBAMI, A., 1990. A survey for hemagglutination-inhibiting antibody to west nile virus in human and animal sera in nigeria. Comparative Immunology Microbiology and Infectious Diseases, 13, 35-39.
- OLIPHANT, T., NYBAKKEN, G., ENGLE, M., XU, Q., NELSON, C., SUKUPOLVI-PETTY, S., MARRI, A., LACHMI, B., OLSHEVSKY, U., FREMONT, D., PIERSON, T. & DIAMOND, M., 2006. Antibody recognition and neutralization determinants on domains I and II of West Nile virus envelope protein. Journal of Virology, 80, 12149-12159.
- OSORIO, J. E., CIUODERIS, K. A., LOPERA, J. G., PIEDRAHITA, L. D., MURPHY, D., LEVASSEUR, J., CARRILLO, L., OCAMPO, M. C. & HOFMEISTER, E., 2012. Characterization of West Nile Viruses Isolated from Captive American Flamingoes (Phoenicopterus ruber) in Medellin, Colombia. American Journal of Tropical Medicine and Hygiene, 87, 565-572.
- OSTLUND, E. N., ANDRESEN, J. E. & ANDRESEN, M., 2000. West Nile encephalitis. Veterinary Clinics of North America: Equine Practice, 16, 427-441.
- PADGETT, K. A., REISEN, W. K., KAHL-PURCELL, N., FANG, Y., CAHOON-YOUNG, B., CARNEY, R., ANDERSON, N., ZUCCA, L., WOODS, L., HUSTED, S. & KRAMER, V. L., 2007. West Nile virus infection in tree squirrels (Rodentia, Sciuridae) in California, 2004-2005. American Journal of Tropical Medicine and Hygiene, 76, 810-813.
- PAPA, A., BAKONYI, T., XANTHOPOULOU, K., VAZQUEZ, A., TENORIO, A. & NOWOTNY, N., 2011. Genetic characterization of West Nile virus lineage 2, Greece, 2010. Emerging Infectious Diseases, 17, 920-922.
- PARQUET, M. C., KUMATORI, A., HASEBE, F., MORITA, K. & IGARASHI, A., 2001. West Nile virus-induced bax-dependent apoptosis. FEBS Letters, 500(1-2), 17-24.
- PAZ, S. & SEMENZA, J. C., 2013. Environmental drivers of West Nile fever epidemiology in Europe and Western Asia--a review. International Journal of Environmental Research and Public Health, 10, 3543-3562.
- PENNICK, K., MCKNIGHT, C., PATTERSON, J., LATIMER, K., MAES, R., WISE, A. & KIUPEL, M., 2012. Diagnostic sensitivity and specificity of in situ hybridization and immunohistochemistry for Eastern equine encephalitis virus and West Nile virus in formalin-fixed, paraffin-embedded brain tissue of horses. Journal of Veterinary Diagnostic Investigation, 24, 333-338.
- PERELYGIN, A. A., SCHERBIK, S. V., ZHULIN, I. B., STOCKMAN, B. M., LI, Y. & BRINTON, M. A., 2002. Positional cloning of the murine flavivirus resistance gene. Proceedings of the National Academy of Sciences of the United States of America, 99, 9322-9327.
- PÉREZ-RAMÍREZ, E., LLORENTE, F., DEL AMO, J., FALL, G., SALL, A. A., LUBISI, A., LECOLLINET, S., VÁZQUEZ, A. & JIMÉNEZ-CLAVERO, M., 2017. Pathogenicity evaluation of twelve West Nile virus strains belonging to four lineages from five continents in a mouse model, discrimination between three pathogenicity categories. Journal of General Virology, 98, 662-670.
- PETERSEN, L. R., BRAULT, A. C. & NASCI, R. S., 2013. West Nile virus, review of the literature. Journal of the American Medical Association, 310, 308-315.
- PETERSEN, L. R., ROEHRIG, J. T. & HUGHES, J. M., 2002. West Nile virus encephalitis. New England Journal of Medicine, 347, 1225-1226.
- PIERRE, V., DROUET, M. T. & DEUBEL, V., 1994. Identification of mosquito-borne flavivirus sequences using universal primers and reverse transcription-polymerase chain-reaction. Research in Virology, 145, 93-104.
- PIERSON, T. & DIAMOND, M., 2012. Degrees of maturity, the complex structure and biology of flaviviruses. Current Opinion in Virology, 2, 168-175.
- PIERSON, T., XU, Q., NELSON, S., OLIPHANT, T., NYBAKKEN, G., FREMONT, D. & DIAMOND, M., 2007. The stoichiometry of antibody-mediated neutralization and enhancement of west nile virus infection. Cell Host and Microbe, 1, 135-145.
- PLATT, K., TUCKER, B., HALBUR, P., TIAWSIRISUP, S., BLITVICH, B., FABIOSA, F., BARTHOLOMAY, L. & ROWLEY, W., 2007. West Nile virus viremia in eastern Chipmunks (Tamias striatus) sufficient for infecting different mosquitoes. Emerging Infectious Diseases, 13, 831-837.
- PORTER, M. B., LONG, M., GOSCHE, D. G., SCHOTT, H. M., HINES, M. T., ROSSANO, M. & SELLON, D. C., 2004. Immunoglobulin M-capture enzyme-linked Immunosorbent assay testing of cerebrospinal fluid and serum from horses exposed to West Nile virus by vaccination or natural infection. Journal of Veterinary Internal Medicine, 18, 866-870.
- PORTER, M. B., LONG, M. T., GETMAN, L. M., GIGUÈRE, S., MACKAY, R. J., LESTER, G. D., ALLEMAN, A. R., WAMSLEY, H. L., FRANKLIN, R. P., JACKS, S., BUERGELT, C. D. & DETRISAC, C. J., 2003. West Nile virus encephalomyelitis in horses, 46 cases (2001). New England Journal of Medicine, 222, 1241-1247.
- PORTER, R. S., LEBLOND, A., LECOLLINET, S., TRITZ, P., CANTILE, C., KUTASI, O., ZIENTARA, S., PRADIER, S., VAN GALEN, G., SPEYBROEK, N. & SAEGERMAN, C., 2011. Clinical diagnosis of West Nile Fever in Equids by classification and regression tree (CART) analysis and comparative study of clinical appearance in three European countries. Transboundary and Emerging Diseases, 58, 197-205.
- RAMANATHAN, M., PANKHONG, P., KUTZLER, M., CHAMBERS, C., KIM, J., GUNASEKARAN, G., PIERSON, T. & WEINER, D., 2006. Protein kinase CK2, A potential target in the development of novel therapeutics against West Nile virus. Journal of Neurovirology, 12, 69-69.
- RAPPOLE, J. H., DERRICKSON, S. R. & HUBÁLEK, Z., 2000. Migratory birds and spread of West Nile virus in the Western Hemisphere. Emerging Infectious Diseases, 6, 319-328.
- REAGAN, R. L., ORLOWSKI, M. & BRUECKNER, A. L., 1953. Response of young dogs to West Nile virus. Proceedings of the Society for Experimental Biology and Medicine, 82, 261-262.
- RIMOLDI, G., METE, A., ADASKA, J. M., ANDERSON, M. L., SYMMES, K. P. & DIAB, S., 2017. West Nile virus infection in sheep. Veterinary Pathology, 54, 155-158.
- RIOS, J. J., FLEMING, J. G. W., BRYANT, U. K., CARTER, C. N., HUBER, J. C. J., LONG, M. T., SPENCER, T. E. & ADELSON, D. L., 2010. OAS1 Polymorphisms Are Associated with Susceptibility to West Nile Encephalitis in Horses. PLoS One, 5(5).
- RIOS, L. M., SHEU, J. J., DAY, J. F., MARUNIAK, J. E., SEINO, K., ZARETSKY, H. & LONG, M. T., 2009. Environmental risk factors associated with West Nile virus clinical disease in Florida horses. Medical and Veterinary Entomology, 23, 357-366.
- RIZZO, C., VESCIO, F., DECLICH, S., FINARELLI, A., MACINI, P., MATTIVI, A., ROSSINI, G., PIOVESAN, C., BARZON, L., PALU, G., GOBBI, F., MACCHI, L., PAVAN, A., MAGURANO, F., CIUFOLINI, M., NICOLETTI, L., SALMASO, S. & REZZA, G., 2009. West Nile virus transmission with human cases in italy, August-September 2009. Eurosurveillance, 14, 5-8.
- ROE, K., KUMAR, M., LUM, S., ORILLO, B., NERURKAR, V. R. & VERMA, S., 2012. West Nile virus-induced disruption of the blood-brain barrier in mice is characterized by the degradation of the junctional complex proteins and increase in multiple matrix metalloproteinases. Journal of General Virology, 93(6), 1193-1203.
- ROE, K., ORILLO, B. & VERMA, S., 2014. West Nile virus-induced cell adhesion molecules on human brain microvascular endothelial cells regulate leukocyte adhesion and modulate permeability of the in vitro blood-brain barrier model. PLoS One, 9, e102598.
- SAEGERMAN, C., ALBA-CASALS, A., GARCÍA-BOCANEGRA, I., DAL POZZO, F. & VAN GALEN, G., 2016. Clinical Sentinel Surveillance of Equine West Nile Fever, Spain. Transboundary Emerging Diseases, 63, 184-193.
- SALAZAR, P., TRAUB-DARGATZ, J. L., MORLEY, P. S., WILMOT, D. D., STEFFEN, D. J., CUNNINGHAM, W. E. & SALMAN, M. D., 2004. Outcome of equids with clinical signs of West Nile virus infection and factors associated with death. New England Journal of Medicine, 225, 267-274.
- SAMUEL, M. A., MORREY, J. D. & DIAMOND, M. S., 2007. Caspase 3-dependent cell death of neurons contributes to the pathogenesis of West Nile virus encephalitis. Journal of Virology, 81, 2614-2623.
- SAMUEL, M. A., WANG, H., SIDDHARTHAN, V., MORREY, J. D. & DIAMOND, M. S., 2007. Axonal transport mediates West Nile virus entry into the central nervous system and induces acute flaccid paralysis. Proceedings of the National Academy of Sciences of the United States of America, 104, 17140-17145.
- SANCHEZ, M., PIERSON, T., DEL PIERO, F., DAVIDSON, A., TRAUB-DARGATZ, J., HANNA, S., HOXIE, J. & DOMS, R., 2005. Neutralization of West Nile virus by monoclonal antibodies and horse sera. American Journal of Tropical Medicine and Hygiene, 73, 90.
- SANCHEZ, M., PIERSON, T., MCALLISTER, D., HANNA, S., PUFFER, B., VALENTINE, L., MURTADHA, M., HOXIE, J. & DOMS, R., 2005. Characterization of neutralizing antibodies to West Nile virus. Virology, 336, 70-82.
- SCHAUDIN, D., SCHWAB, D., LINKE, S., SEELIGER, F., PAULI, G., BAUMGARTNER, W. & HERDEN, C., 2008. Lack of detectable West Nile virus RNA in brains of kidneys of dogs and cats with immunohistological precipitates using virus-specific antibodies. Veterinary Microbiology, 25, 171-176.
- SCHMIDT, J. R. & ELMANSOURY, H. K., 1963. Natural and experimental infection of egyptian equines with west nile virus. Annals of Tropical Medicine and Parasitology, 57, 415-427.
- SCHULER, L. A., KHAITSA, M. L., DYER, N. W. & STOLTENOW, C. L., 2004. Evaluation of an outbreak of West Nile virus infection in horses, 569 cases (2002). New England Journal of Medicine, 225, 1084-1089.
- SEINO, K. K., LONG, M. T., GIBBS, E. P., BOWEN, R. A., BEACHBOARD, S. E., HUMPHREY, P. P., DIXON, M. A. & BOURGEOIS, M. A., 2007. Comparative efficacies of three commercially available vaccines against West Nile Virus (WNV) in a short-duration challenge trial involving an equine WNV encephalitis model. Clinical and Vaccine Immunology, 14, 1465-1471.
- SHIRAFUJI, H., KANEHIRA, K., KUBO, M., SHIBAHARA, T. & KAMIO, T., 2009. Experimental West Nile virus infection in aigamo ducks, a cross between wild ducks (Anas platyrhynchos) and domestic ducks (Anas platyrhynchos var. domesticus). Avian Diseases, 53, 239-244.
- SHRESTHA, B. & DIAMOND, M. S., 2004. Role of CD8+ T cells in control of West Nile virus infection. Journal of Virology, 78, 8312-8321.
- SHRESTHA, B., NG, T., CHU, H. J., NOLL, M. & DIAMOND, M. S., 2008. The relative contribution of antibody and CD8+ T cells to vaccine immunity against West Nile encephalitis virus. Vaccine, 26, 2020-2033.
- SIGER, L., BOWEN, R., KARACA, K., MURRAY, M., JAGANNATHA, S., ECHOLS, B., NORDGREN, R. & MINKE, J. M., 2006. Evaluation of the efficacy provided by a Recombinant Canarypox-Vectored Equine West Nile Virus vaccine against an experimental West Nile Virus intrathecal challenge in horses. Veterinary Therapeutics, 7, 249-256.
- SIMPSON, J. E., FOLSOM-O'KEEFE, C. M., CHILDS, J. E., SIMONS, L. E., ANDREADIS, T. G. & DIUK-WASSER, M. A., 2009. Avian host-selection by Culex pipiens in experimental trials 71. PLoS One, 4, e7861.
- SMITHBURN, K. C., HUGHES, T. P., BURKE, A. W. & PAUL, J. H., 1940. A neurotropic virus isolated from the blood of a native of Uganda. American Journal of Tropical Medicine, 20, 471-472.
- SNOOK, C., HYMAN, S., DEL PIERO, F., PALMER, J., OSTLUND, E., BARR, B., DESROCHERS, A. & REILLY, L., 2001. West Nile virus encephalomyelitis in eight horses. Journal of the American Veterinary Medical Association, 218, 1576-1579.
- SOTELO, E., GUTIERREZ-GUZMAN, A. V., DEL AMO, J., LLORENTE, F., EL-HARRAK, M., PEREZRAMIREZ, E., MANUEL BLANCO, J., HOEFLE, U. & ANGEL JIMENEZ-CLAVERO, M., 2011. Pathogenicity of two recent Western Mediterranean West Nile virus isolates in a wild bird species indigenous to Southern Europe: the redlegged partridge. Veterinary Research, 42, 11.
- STEINMAN, A., BANET, C., SUTTON, G. A., YADIN, H., HADAR, S. & BRILL, A., 2002. Clinical signs of West Nile virus encephalomyelitis in horses during the outbreak in Israel in 2000. Veterinary Record, 151, 47-49.
- STEINMAN, A., BANET-NOACH, C., SIMANOV, L., GRINFELD, N., AIZENBERG, Z., LEVI, O., LAHAV, D., MALKINSON, M., PERK, S. & SHPIGEL, N. Y., 2006. Experimental infection of common garter snakes (Thamnophis sirtalis) with West Nile virus. Vector-Borne and Zoonotic Diseases, 6, 361-368.
- STURA, E., TAUSSIG, M., SUTTON, B., DUQUERROY, S., BRESSANELLI, S., MINSON, A. & REY, F., 2002. Scaffolds for protein crystallisation. Acta Crystallographica Section D-Biological Crystallography, 58, 1715-1721.
- SUEN, W., UDDIN, M., WANG, W., BROWN, V., ADNEY, D., BROAD, N., PROW, N., BOWEN, R., HALL, R. & BIELEFELDT-OHMANN, H., 2015. Experimental West Nile Virus Infection in Rabbits, An Alternative Model for Studying Induction of Disease and Virus Control. Pathogens, 4, 529-558.
- SUEN, W. W., PROW, N. A., HALL, R. A. & BIELEFELDT-OHMANN, H., 2014. Mechanism of West Nile virus neuroinvasion, a critical appraisal. Viruses, 6, 2796-2825.
- TANNER, J. M., TRAUB-DARGATZ, J. L., HILL, A. E., VAN CAMPEN, H., KNIGHT, A. P., CUNNINGHAM, W. E. & SALMAN, M. D., 2006. Evaluation of factors associated with positive IgM capture ELISA results in equids with clinical signs compatible with West Nile virus infection, 1,017 cases (2003). England Journal of Medicine, 228, 414-421.
- TBER, A., 1996. West Nile fever in horses in Morocco. Bulletin De L'Office International Des Epizooties, 11, 867-869.
- TIAWSIRISUP, S., PLATT, K., TUCKER, B. & ROWLEY, W., 2005. Eastern cottontail rabbits (Sylvilagus floridanus) develop West Nile virus viremias sufficient for infecting select mosquito species. Vector-Borne and Zoonotic Diseases, 5, 342-350.
- TOMA, L., CIPRIANI, M., GOFFREDO, M., ROMI, R. & LELLI, R., 2008. First report on entomological field activities for the surveillance of West Nile disease in Italy. Veterinaria Italiana, 44, 483-512.
- TRAN, A., L'AMBERT, G., BALANCA, G., PRADIER, S., GROSBOIS, V., BALENGHIEN, T., BALDET, T., LECOLLINET, S., LEBLOND, A. & GAIDET-DRAPIER, N., 2017. An Integrative Eco-Epidemiological Analysis of West Nile Virus Transmission. EcoHealth, 14, 474-489.
- TURELL, M. J., DOHM, D. J., SARDELIS, M. R., OGUINN, M. L., ANDREADIS, T. G. & BLOW, J. A., 2005. An update on the potential of north American mosquitoes (Diptera, Culicidae) to transmit West Nile Virus 189. Journal of Medical Entomology, 42, 57-62.
- TURELL, M. J., MORRILL, J. C., ROSSI, C. A., GAD, A. M., COPE, S. E., CLEMENTS, T. L., ARTHUR, R. R., WASIELOSKI, L. P., DOHM, D. J., NASH, D., HASSAN, M. M., HASSAN, A. N., MORSY, Z. S. & PRESLEY, S. M., 2002. Isolation of west nile and sindbis viruses from mosquitoes collected in the Nile Valley of Egypt during an outbreak of Rift Valley fever. Journal of Medical Entomology, 39, 248-250.
- URBANOWSKI, M. D. & HOBMAN, T. C., 2013. The West Nile virus capsid protein blocks apoptosis through a phosphatidylinositol 3-kinase-dependent mechanism. Journal of Virology, 87, 872-881.
- VALIAKOS, G., TOULOUDI, A., ATHANASIOU, L. V., GIANNAKOPOULOS, A., IACOVAKIS, C., BIRTSAS, P., SPYROU, V., DALABIRAS, Z., PETROVSKA, L. & BILLINIS, C., 2012. Serological and molecular investigation into the role of wild birds in the epidemiology of West Nile virus in Greece. Journal of Virology, 9, 266.
- VANDALEN, K. K., HALL, J. S., CLARK, L., MCLEAN, R. G. & SMERASKI, C., 2013. West Nile virus infection in American Robins, new insights on dose response. PLoS One, 8, e68537.
- VENTER, M., HUMAN, S., ZAAYMAN, D., GERDES, G. H., WILLIAMS, J., STEYL, J., LEMAN, P. A., PAWESKA, J. T., SETZKORN, H., ROUS, G., MURRAY, S., PARKER, R., DONNELLAN, C. & SWANEPOEL, R., 2009. Lineage 2 West Nile virus as casue of fatal neurologic disease in horses, South Africa. Emerging Infectious Diseases, 15, 877-884. doi. 10.3201/eid1506.081515.
- VENTER, M., PRETORIUS, M., FULL, J. A., BOTHA, E., RAKGOTHO, M., STIVAKTAS, V., WEYER, C., ROMITO, M. & WILLIAMS, J., 2017. West Nile virus lineage 2 in horeses and other animals with neurologic disease, South Africa, 2008-2015. Emerging Infectious Diseases, 23, 2060-2064.
- VERMA, S., LO, Y., CHAPAGAIN, M., LUM, S., KUMAR, M., GURJAV, U., LUO, H., NAKATSUKA, A. & NERURKAR, V. R., 2009. West Nile virus infection modulates human brain microvascular endothelial cells tight junction proteins and cell adhesion molecules, Transmigration across the in vitro blood-brain barrier. Virology, 385, 425-433.
- VITEK, C. J., RICHARDS, S. L., MORES, C. N., DAY, J. F. & LORD, C. C., 2008. Arbovirus transmission by Culex nigripalpus in Florida, 2005. Journal of Medical Entomology, 45, 483-493.
- VITTECOQ, M., LECOLLINET, S., JOURDAIN, E., THOMAS, F., BLANCHON, T., ARNAL, A., LOWENSKI, S. & GAUTHIER-CLERC, M., 2013. Recent circulation of west nile virus and potentially other closely related flaviviruses in Southern France. Vector-Borne and Zoonotic Diseases, 13, 610-613.
- VRIONI, G., MAVROULI, M., KAPSIMALI, V., STAVROPOULOU, A., DETSIS, M., DANIS, K. & TSAKRIS, A., 2014. Laboratory and clinical characteristics of human West Nile virus infections during 2011 outbreak in southern Greece. Vector-Borne and Zoonotic Diseases, 14, 52-58.
- WAMSLEY, H. L., ALLEMAN, A. R., PORTER, M. B. & LONG, M. T., 2002. Findings in cerebrospinal fluids of horses infected with West Nile virus, 30 cases (2001). New England Journal of Medicine, 221, 1303-1305.
- WANG, H., SIDDHARTHAN, V., HALL, J. O. & MORREY, J. D., 2011. Autonomic nervous dysfunction in hamsters infected with West Nile virus. PLoS One, 6, e19575.
- WANG, H., SIDDHARTHAN, V., HALL, J. O. & MORREY, J. D., 2014. Autonomic deficit not the cause of death in West Nile virus neurological disease. Clinical Autonomic Research, 24, 15-23.
- WANG, J., YANG, J., GE, J., HUA, R., LIU, R., LI, X., WANG, X., SHAO, Y., SUN, E., WU, D., QIN, C., WEN, Z. & BU, Z., 2016. Newcastle disease virus-vectored West Nile fever vaccine is immunogenic in mammals and poultry. Journal of Virology, 13,109.
- WARD, M. P., SCHUERMANN, J. A., HIGHFIELD, L. D. & MURRAY, K. O., 2006. Characteristics of an outbreak of West Nile virus encephalomyelitis in a previously uninfected population of horses. Veterinary Microbiology, 118(3-4), 255-259.
- WEESE, J. S., BAIRD, J. D., DELAY, J., KENNEY, D. G., STAEMPFLI, H. R., VIEL, L., PARENT, J., SMITH-MAXIE, L. & POMA, R., 2003. West Nile virus encephalomyelitis in horses in Ontario, 28 cases. Canadian Veterinary Journal, 44, 469-473.
- WEINBERGER, M., PITLIK, S. D., GANDACU, D., LANG, R., NASSAR, F., BEN DAVID, D., RUBINSTEIN, E., IZTHAKI, A., MISHAL, J., KITZES, R., SIEGMAN-IGRA, Y., GILADI, M., PICK, N., MENDELSON, E., BIN, H. & SHOHAT, T., 2001. West Nile fever outbreak, Israel, 2000, epidemiologic aspects. Emerging Infectious Disease, 7, 686-691.
- WESTAWAY, E., BRINTON, M., GAIDAMOVICH, S., HORZINEK, M., IGARASHI, A., KAARIAINEN, L., LVOV, D., PORTERFIELD, J., RUSSELL, P. & TRENT, D., 1985. Togaviridae. Intervirology, 24, 125-139.
- WHEELER, S. S., VINEYARD, M. P., WOODS, L. W. & REISEN, W. K., 2012. Dynamics of West Nile Virus Persistence in House Sparrows (Passer domesticus). PLoS Neglected Tropical Diseases,6(10).
- WHITEHEAD, C. & BEDENICE, D., 2009. Neurologic Diseases in Llamas and Alpacas. Veterinary Clinics of North America-Food Animal Practice, 25, 385-389.
- WUNSCHMANN, A., SHIVERS, J., CARROLL, L. & BENDER, J., 2004. Pathological and immunohistochemical findings in American crows (Corvus brachyrhynchos) naturally infected with West Nile virus. Journal of Veterinary Diagnostic Investigation, 16, 329-333.
- ZEHENDER, G., VEO, C., EBRANATI, E., CARTA, V., ROVIDA, F., PERCIVALLE, E., MORENO, A., LELLI, D., CALZOLARI, M., LAVAZZA, A., CHIAPPONI, C., BAIONI, L., CAPELLI, G., RAVAGNAN, S., DA ROLD, G., LAVEZZE, E., PALU, G., BALDANTI, F., BARZON, L. & GALLI, M., 2017. Reconstructing the recent West Nile virus lineage 2 epidemic in Europe and Italy using discrete and continuous phylogeography. PLoS One, 12(7).
- ZHANG, R., MINER, J., GORMAN, M., RAUSCH, K., RAMAGE, H., WHITE, J., ZUIANI, A., ZHANG, P., FERNANDEZ, E., ZHANG, Q., DOWD, K., PIERSON, T., CHERRY, S. & DIAMOND, M., 2016. A CRISPR screen defines a signal peptide processing pathway required by flaviviruses. Nature, 535(7610), 164-169.
- ZHANG, Y., CORVER, J., CHIPMAN, P., ZHANG, W., PLETNEV, S., SEDLAK, D., BAKER, T., STRAUSS, J., KUHN, R. & ROSSMANN, M., 2003. Structures of immature flavivirus particles. Embo Journal, 22, 2604-2613.
- ZHANG, Y., ZHANG, W., OGATA, S., CLEMENTS, D., STRAUSS, J., BAKER, T., KUHN, R. & ROSSMANN, M., 2004. Conformational changes of the flavivirus E glycoprotein. Structure, 12,1607-1618.
- ZUKOR, K., WANG, H., HURST, B. L., SIDDHARTHAN, V., VAN WETTERE, A., PILOWSKY, P. M. & MORREY, J. D., 2017. Phrenic nerve deficits and neurological immunopathology associated with acute West Nile virus infection in mice and hamsters. Journal of NeuroVirology, 23, 186-204.