- Infectious Diseases of Livestock
- Part 2
- Nairobi sheep disease
- GENERAL INTRODUCTION: PARAMYXOVIRIDAE AND PNEUMOVIRIDAE
- Rinderpest
- Peste des petits ruminants
- Parainfluenza type 3 infection
- Bovine respiratory syncytial virus infection
- Hendra virus infection
- Paramyxovirus-induced reproductive failure and congenital defects in pigs
- Nipah virus disease
- GENERAL INTRODUCTION: CALICIVIRIDAE AND ASTROVIRIDAE
- Vesicular exanthema
- Enteric caliciviruses of pigs and cattle
- GENERAL INTRODUCTION: RETROVIRIDAE
- Enzootic bovine leukosis
- Jaagsiekte
- Visna-maedi
- Caprine arthritis-encephalitis
- Equine infectious anaemia
- GENERAL INTRODUCTION: PAPILLOMAVIRIDAE
- Papillomavirus infection of ruminants
- Papillomavirus infection of equids
- GENERAL INTRODUCTION: ORTHOMYXOVIRIDAE
- Equine influenza
- Swine influenza
- GENERAL INTRODUCTION: CORONAVIRIDAE
- Porcine transmissible gastroenteritis
- Porcine respiratory coronavirus infection
- Porcine epidemic diarrhoea
- Porcine haemagglutinating encephalomyelitis virus infection
- Porcine deltacoronavirus infection
- Bovine coronavirus infection
- Ovine coronavirus infection
- Equine coronavirus infection
- GENERAL INTRODUCTION: PARVOVIRIDAE
- Porcine parvovirus infection
- Bovine parvovirus infection
- GENERAL INTRODUCTION: ADENOVIRIDAE
- Adenovirus infections
- GENERAL INTRODUCTION: HERPESVIRIDAE
- Equid herpesvirus 1 and equid herpesvirus 4 infections
- Equid gammaherpesvirus 2 and equid gammaherpesvirus 5 infections
- Equine coital exanthema
- Infectious bovine rhinotracheitis/infectious pustular vulvovaginitis and infectious pustular balanoposthitis
- Bovine alphaherpesvirus 2 infections
- Malignant catarrhal fever
- Pseudorabies
- Suid herpesvirus 2 infection
- GENERAL INTRODUCTION: ARTERIVIRIDAE
- Equine viral arteritis
- Porcine reproductive and respiratory syndrome
- GENERAL INTRODUCTION: FLAVIVIRIDAE
- Bovine viral diarrhoea and mucosal disease
- Border disease
- Hog cholera
- Wesselsbron disease
- Louping ill
- West nile virus infection
- GENERAL INTRODUCTION: TOGAVIRIDAE
- Equine encephalitides caused by alphaviruses in the Western Hemisphere
- Old World alphavirus infections in animals
- Getah virus infection
- GENERAL INTRODUCTION: BUNYAVIRIDAE
- Diseases caused by Akabane and related Simbu-group viruses
- Rift Valley fever
- Nairobi sheep disease
- Crimean-Congo haemorrhagic fever
- GENERAL INTRODUCTION: ASFARVIRIDAE
- African swine fever
- GENERAL INTRODUCTION: RHABDOVIRIDAE
- Rabies
- Bovine ephemeral fever
- Vesicular stomatitis and other vesiculovirus infections
- GENERAL INTRODUCTION: REOVIRIDAE
- Bluetongue
- Ibaraki disease in cattle
- Epizootic haemorrhagic disease
- African horse sickness
- Equine encephalosis
- Palyam serogroup orbivirus infections
- Rotavirus infections
- GENERAL INTRODUCTION: POXVIRIDAE
- Lumpy skin disease
- Sheeppox and goatpox
- Orf
- Ulcerative dermatosis
- Bovine papular stomatitis
- Pseudocowpox
- Swinepox
- Cowpox
- Horsepox
- Camelpox
- Buffalopox
- GENERAL INTRODUCTION: PICORNAVIRIDAE
- Teschen, Talfan and reproductive diseases caused by porcine enteroviruses
- Encephalomyocarditis virus infection
- Swine vesicular disease
- Equine picornavirus infection
- Bovine rhinovirus infection
- Foot-and-mouth disease
- GENERAL INTRODUCTION: BORNAVIRIDAE
- Borna disease
- GENERAL INTRODUCTION: CIRCOVIRIDAE AND ANELLOVIRIDAE
- Post-weaning multi-systemic wasting syndrome in swine
- GENERAL INTRODUCTION: PRION DISEASES
- Scrapie
- Bovine spongiform encephalopathy
- Transmissible spongiform encephalopathies related to bovine spongiform encephalopathy in other domestic and captive wild species
Nairobi sheep disease
This content is distributed under the following licence: Attribution-NonCommercial CC BY-NC  View Creative Commons Licence details here
View Creative Commons Licence details here
Nairobi sheep disease
F G DAVIES AND C TERPSTRA
Introduction
Nairobi sheep disease (NSD) is a tick-transmitted viral disease of small ruminants, mainly of sheep, for they are usually more numerous in the areas where the tick vectors, which are principally of the genus Rhipicephalus, are found. Goats are kept in the drier semi-arid zones. The disease is characterized by fever, debility and prostration, a haemorrhagic gastroenteritis, abortion and a high mortality. Nairobi sheep disease virus (NSDV) of the family Bunyaviridae is one of the most pathogenic viruses for small ruminants in East Africa.2, 5, 6, 18
Nairobi sheep disease was first recognized by Montgomery in Kenya in 191718 in sheep and goats after they had been moved from the dry semi-arid areas of the northern and southern parts of Kenya to the vicinity of Nairobi. It occurs in Kenya, Uganda, Tanzania and Ruanda,2 and has also been described from Northern Somalia and the haud (highland, Acacia-bushed grasslands), which is the contiguous ecozone of Region V of Ethiopia.6, 22, 24 Some NSD-positive sera have been found in sheep in Botswana and the Mozambique coastal plain, extending into South Africa’s northern KwaZulu-Natal Province.15, 28 The results of further studies have, however, suggested that the latter may have been cross-reactions with other Nairoviruses in the test systems used (mouse virus serum neutralization tests) and not be specific for NSD. In addition, many sheep and goat sera from Zambia and Botswana were subsequently tested and a few very low titre positives found, which also suggest crossreactions with other nairoviruses, and not specific responses to NSD infections.7 The clinical disease has not yet been diagnosed in these countries of southern Africa.
The identical or closely related Ganjam virus has been isolated from several species of Haemaphysalis ticks (intermedia and wellingtonii) in many Indian states and in Sri Lanka.
Aetiology
Nairobi sheep disease virus (NSDV) is classified in the family Bunyaviridae, genus Nairovirus22, 25(see Rift Valley fever). It is sensitive to lipid solvents and detergents and is rapidly inactivated at high and low pH values. Even in the range of optimal stability (pH 7,4 to 8,0) in the presence of 2 per cent serum, the virus is rapidly inactivated with a half-life of 6,8 days at 0 °C and 1,5 hours at 37 °C.25
Nairobi sheep disease virus is antigenically identical, or very closely related to Ganjam virus and more distantly related to Dugbe virus, which is responsible for a tick-transmitted infection of cattle in Nigeria.8There is no reported evidence for any immunological diversity between strains isolated from different geographical areas.
The virus replicates in vitro in primary or secondary cell cultures derived from sheep or goat testes and kidneys, in continuous cell lines of BHK, PS and Vero cells, and in cell lines prepared from Rhipicephalus appendiculatus ticks.1, 14, 20 Infected cells usually develop cytopathic effects, which become more obvious after one to two passages. Staining with haemotoxylin and eosin reveals typical eosinophilic intracytoplasmic inclusion bodies.1 These are ring- or spindle-shaped, polar or bipolar inclusions, which can also be observed by immuno- fluorescence. They contain large clusters of polyribosomes, which are revealed by electron microscopic examination.26 Electron microscopy shows that virus replication occurs in the cytoplasm by budding from the membranes of smooth surface intracellular vesicles associated with the Golgi complex. Virus particles are released by exocytosis and by cell lysis. The molecular basis for this is the retention of viral envelope glycoproteins G1 and G2 in the Golgi complex during replication. Virus particles are spherical in shape and approximately 80 to 100 nm in diameter, with a denser core and a double envelope of 5 nm thickness. They are typical of bunyavirus particles.
Nairobi sheep disease virus produces a fatal infection in infant mice when inoculated intracerebrally or intraperitoneally. Adult mice are only susceptible by the intracerebral route.29 The virulence of the virus decreases rapidly after serial passage in the brains of infant mice. A statistically significant difference was observed in the duration of illness and mortality between Persian fat-tailed sheep infected with the fifth mouse brain passage of the Entebbe strain and those infected with the progeny of the fifth brain passage after re-passage through ticks.26
A mouse brain attenuated strain has been propagated in embryonated hens’ eggs following chorioallantoic inoculation and modification of virulence has also followed passage in adult mouse brain.6
Epidemiology
Sheep and goats are the natural vertebrate hosts of NSDV. Cattle, African buffalo (Syncerus caffer), horses and pigs are refractory to infection. Although natural and even fatal infections have been reported in blue duikers (Cephalophus monticola), and viraemia and an immune response demonstrated in experimentally infected field rats (Arvicanthis abyssinicus nubilans), serological studies have not supported a suggestion that small antelopes or wild rodents are significantly involved in the maintenance cycle of NSDV.4 The waterbuck (Kobus ellipsiprymnus) and other wild ruminants, which are heavily infested with Rhipicephalus appendiculatus, do not have a high prevalence of NSD antibody.4 Antibodies have been found in human sera27 and accidental infections acquired in the laboratory resulting in fever, joint aches and general malaise have been reported.30 Nairobi sheep disease is not a common cause of laboratory infections even after long exposure and even accidental needle inoculation, which is surprising. Ganjam virus, however, has produced many accidental laboratory infections.
Ticks, mainly of the genus Rhipicephalus, serve as invertebrate hosts and vectors of the virus. The common vector of NSDV in East Africa is the brown ear tick (R. appendiculatus), but other species, including Rhipicephalus pulchellus, Rhipicephalus simus, and the bont tick (Amblyomma variegatum), can transmit the virus, albeit much less efficiently.3, 17 Rhipicephalus appendiculatus, in which transovarial and transstadial transmission has been shown to occur, is by far the most efficient vector.3, 12, 16 In the haud of northern Somalia and Ethiopia, R. pulchellus appears to be the principal tick vector. Transovarial and transstadial transmission of NSDV has been demonstrated to occur in the Somali strain of this tick species22 but it was not transmitted transovarially by the Kenyan strain of this tick.7 Within the same generation of infected R. appendiculatus ticks, all stages remain infective, irrespective of the natural susceptibility of subsequent hosts or their immune status.12 For example, the virus is passed through successive generations of ticks, regardless of the character of the feeding animal hosts, i.e., whether they are susceptible and generate a viraemia, or are not susceptible or immune. The tick does not require an infective feed to transmit the virus to successive generations, as was once thought.12 In one trial unfed larvae of R. appendiculatus were able to infect sheep six months after hatching, whilst unfed nymphae and adults remained infective for 12 and 28 months, respectively.16
The distribution and prevalence of NSDV antibodies in sheep and goats in Kenya and Uganda closely corresponds with the geographic distribution of R. appendiculatus. 5, 24 In such endemic areas, there is a situation of enzootic stability where clinical disease is rarely, if ever, seen in the indigenous sheep and goats. Most sheep and goats in endemic areas have antibody to the virus. The high frequency of antibody in different age groups, reflects a high exposure rate and infection at an early age when the lambs are still protected by maternal immunity.4 Outbreaks of NSD may occur in endemic areas following the introduction of susceptible animals or whenever tick control measures, which had been strictly applied, are allowed to lapse or fail for some reason.
Extensions in the range of the tick vectors occur beyond the usual endemic areas, whenever there are periodic cycles of years with excessive or prolonged rains. These result in vegetational and microclimatical changes that favour marked increases in R. appendiculatus populations. Where previously only one brown ear tick might be found after examining the ears of 20 cattle, after five to nine months some 100 may be found on each ear. The periodicity of NSD epidemics has varied in Kenya from 7 to 15 years, but the effects that the El Nino and Southern Oscillation phenomena have on weather patterns may alter this in future and epidemics may become more frequent.
Experimental infection and natural challenge by infective ticks have revealed no difference in susceptibility between indigenous Persian fat-tailed and European breeds of sheep, although the latter show a milder disease syndrome with lower mortality.24, 26 The 50 per cent infective dose of the fully virulent Entebbe strain of NSDV was found to be close to 0,01 mouse LD50 for both local and exotic breeds.24, 26 Indigenous breeds of sheep may die from NSD within seven or eight days of experimental infection before they can mount an antibody response, whereas in exotic breeds, the disease often runs a more protracted course, allowing the immune system to respond, and the fatality rates are much lower.24
The mortality rate in field outbreaks has been reported to be as high as 70 to 90 per cent for indigenous breeds of sheep and 30 to 40 per cent for exotic and cross-bred animals.18 In endemic areas exposure to the infection early in life when lambs and kids possess passive (colostrum-derived) immunity protects them from the lethal effects of infection. They develop an active immunity, which enables them to survive in heavily tick-infested areas.
Goats are generally regarded as being less susceptible than sheep, and whilst this is usually the case, the mortality in indigenous goat breeds may be as high as 90 per cent.26
Pathogenesis
The pathogenesis of the disease after intravenous inoculation has been studied19 but the sequence of events after vector exposure remains to be determined. Regional lymph nodes draining the area infected by the tick vector first become enlarged and oedematous; this lymphadenopathy may be detected before the febrile reaction. The virus replicates to high titres in the liver, lungs, spleen and other organs of the reticulo-endothelial system, and appears to have a predilection for the vascular endothelium. The primary lesions consist of endothelial swelling, and oedema and necrosis of the capillary walls in the mucous membranes of the abomasum, small and large intestine, gall bladder, and female genital tract, and kidneys. The vascular lesions in the mucous membranes are accompanied by congestion, haemorrhage and catarrhal inflammation, followed by necrosis and desquamation of the epithelium. Oedema of the submucosae is prominent.
Clinical signs
In natural outbreaks disease usually occurs five to six days after susceptible animals have been transferred to paddocks infested with R. appendiculatus. This time interval agrees with experimental observations which suggest an incubation period of three to six days after tick attachment.24 In the case of needle infection, the incubation period generally varies from one to four days, with a maximum of six days, depending on the virus dose, route of infection, virulence of the strain and individual resistance of the animals.
Clinical signs begin with a steep rise in rectal temperature to above 41 °C. Fever persists for one to seven days and may reach 42 °C. The febrile response is accompanied by leukopenia and viraemia, the latter disappearing within 24 hours of the temperature returning to normal. Diarrhoea usually appears one to three days after the onset of fever and worsens during the following few days. The faeces become watery and foetid and often contain mucus and blood. The animals are dull and inappetent, standing with drooping heads while straining continuously, and sometimes groaning at each expiration. In about 50 per cent of cases, a mucopurulent, often blood-tinged, nasal discharge occurs. A lachrymal discharge with conjunctivitis may also be present. Pregnant ewes frequently abort. In peracute and acute cases the time between the appearance of disease and death is usually between two and seven days, but in less acute cases the course of the disease may be as long as 11 days.
The clinical signs of NSD in goats are similar to those in sheep but may be less severe.
Pathology
The hindquarters, soiled with faeces or a mixture of blood and faeces, is usually the most striking feature on external examination of the carcass. There is often conjunctivitis, and dried crusts may be seen around the nostrils as a result of nasal discharge. The carcass may be dehydrated, especially in animals that manifested prolonged scouring.
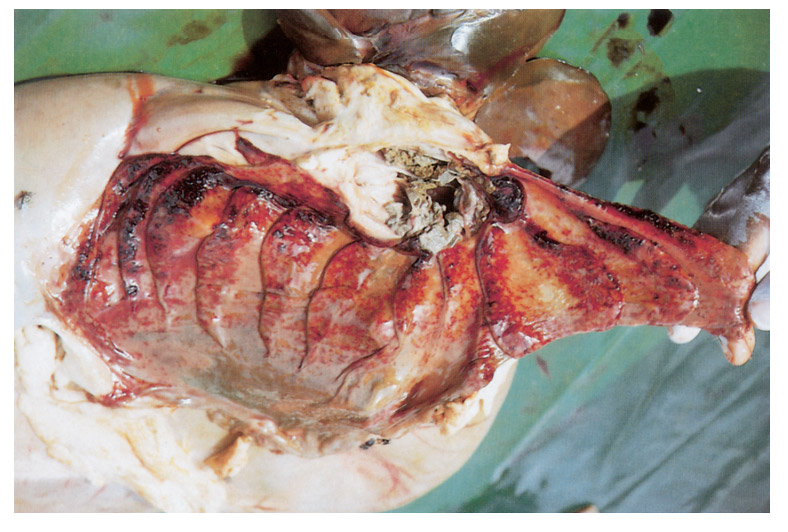
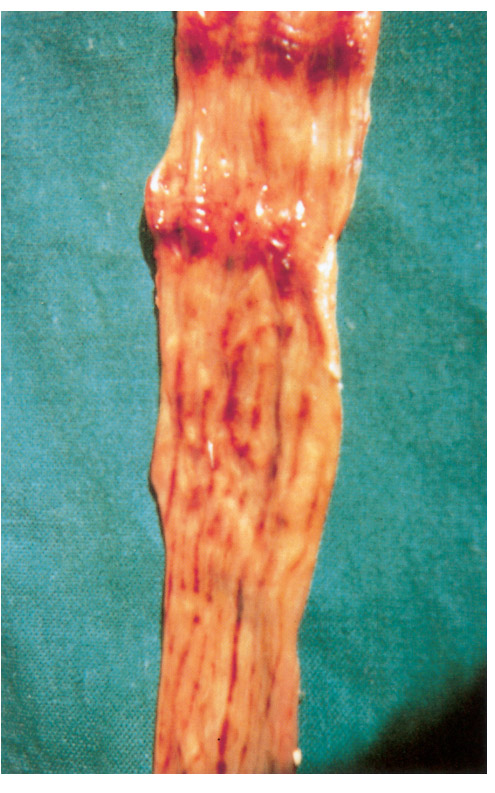
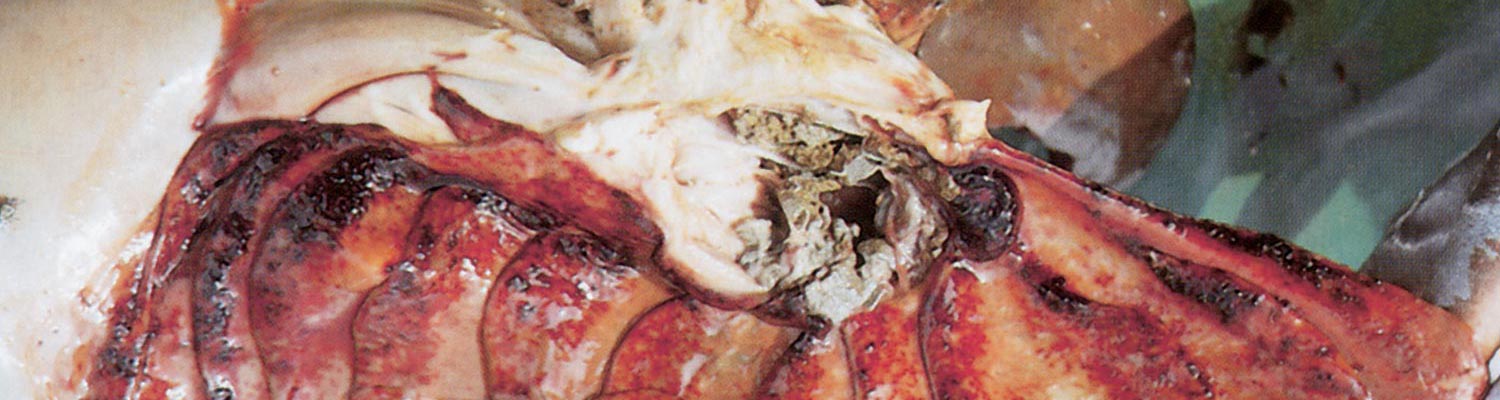
The necropsy picture is typical of a haemorrhagic diathesis accompanied by catarrhal or haemorrhagic inflammation of the gastrointestinal tract and, in ewes, the genital tract.2 The most prominent changes are found in the gastrointestinal tract and the kidneys. The lesions in the gastrointestinal tract consist of multiple haemorrhages of different size, located mainly in the longitudinal folds of the mucosa of the abomasum (Figures 96.1), distal part of the ileum and ileocaecal valve, caecum and proximal colon (Figures 96.2). The haemorrhages in the mucosa of the caecum and colon frequently appear as longitudinal striations while, in severe cases, lines of ecchymoses extend from the cranial caecum and the entire colon to the rectum. These mucosal striations are the most outstanding feature of the necropsy and in some the only evident lesion. They may, however, be absent in a small number of cases. The small intestine is often inflamed and the contents of colon and rectum, if present, are liquid.
Haemorrhages may occur in the subserosa of the caecum, colon and gall bladder, below the capsule of the kidneys, in the mucous membranes of the female genital tract, especially after abortion, and lower respiratory tract. There may be pulmonary congestion with haemorrhage and alveolar oedema. The mucosa of the nasal cavity is often congested and manifests a catarrhal inflammation. The heart contains unclotted blood, its musculature is pale and flaccid, and petechiae and ecchymotic haemorrhages in the epi- and endocardium are present.
The spleen and superficial lymph nodes are usually slightly swollen, and the mesenteric nodes may be grossly enlarged and oedematous. The lymph nodes regional to the commonest site of attachment of the tick are generally more obviously enlarged than any others. The marrow of the long bones frequently has a gelatinous, bright cherry-red appearance.
Microscopically, there is a glomerulo-tubular nephritis with haemorrhages and necrotic changes of the glomerular and tubular epithelium. The lumens of the kidney tubules contain hyaline and cellular casts as early as 48 hours after infection. The kidney lesions are a constant feature of NSD and are important from a diagnostic viewpoint. Chronic interstitial fibrosis of the kidneys is found in recovered animals.19 There is often necrosis of cardiac myocytes and severe2, 19 coagulative necrosis of the mucosa of the gall bladder is usually evident.
Diagnosis and differential diagnosis
The occurrence of an acute febrile and highly fatal disease of sheep and goats, which are obviously infested with the brown ear or other tick species that can transmit the disease, especially after movement to a new area, should raise suspicion of NSD. Flocks can be decimated within a period of a few weeks. The clinical signs and lesions may be sufficiently distinct to make a tentative diagnosis. It must be noted, however, that many of the animals, which die during the acute stage of the disease, do not manifest signs and lesions suggestive of NSD, but merely show petechial and ecchymotic haemorrhages on the serous surfaces of many organs and tissues, and little else. These signs and lesions are not peculiar to NSD, but are found in many other acute diseases such as rinderpest, peste des petits ruminants, Rift Valley fever, heartwater and salmonellosis, and therefore laboratory confirmation is necessary.
Laboratory methods for the diagnosis of NSD are aimed at the identification of the causative virus or viral antigens and specific antibodies to the virus. High virus titres are present in the lungs, liver, spleen and mesenteric lymph nodes and can be recovered from whole blood or serum during the febrile stage of the disease.11, 29 Specimens for virus isolation should be sent on ice without preservatives and must reach the laboratory by the quickest possible means. Nairobi sheep disease virus replicates in a variety of cell cultures and in infant mouse brains. On primary isolation in cell cultures, however, it may not cause cytopathic effects, and immunofluorescence (IF) or immuno-peroxidase staining are useful tools for the detection and rapid identification of isolates.11 Primary isolation of the virus in infant mouse brain and identification either by IF or complement fixation (CF) tests is slightly more sensitive than isolation by the use of cell cultures.11 The inoculation of susceptible sheep with suspensions prepared from infected organs or blood is the most sensitive method for the detection of NSDV.
Viral antigen can be detected in the spleen, lung tissue and mesenteric lymph nodes of sheep that have suffered an acute fatal disease by simple immunodiffusion tests in which NSDV-specific hyperimmune serum is used as a source of antibody.24 The method is simple and rapid and can be applied in field laboratories or diagnostic centres without facilities for cell culture and immunofluorescence.
Immuno-capture enzyme-linked immunosorbent analysis (ELISA) methods may shortly be available for this purpose.21
Antibodies to NSDV may be detected by CF, virus neutralization, agar-gel immuno-diffusion, indirect immunofluorescence and indirect haemagglutination tests.9, 24 Complement-fixing antibody can be detected for only six to nine months after infection. Virus neutralizing antibody levels to NSD are often difficult to demonstrate and it seems probable that neutralization may require the presence of complement. With the other tests, antibodies can be detected within a few days of the body temperature returning to normal and they persist for at least 15 months.11 An ELISA for the detection of NSDV antibody has been described21 but its application in the field has as yet to be assessed.
Control
Efforts to control and prevent NSD have been made by means of vector control and by vaccination in the enzootic areas. In East Africa, it has been found that the control of the vector ticks may be successful for periods of time, but invariably the control systems break down and outbreaks of the disease occur. In addition, tick control measures are very expensive to maintain and may be environmentally undesirable. Long-term tick control strategies are not recommended for NSD. There may be a role for a strategic tick control policy in epidemic situations, however.13
The best strategy for NSD control in endemic areas is to allow a stable endemic state to develop throughout the areas. Justification for interventions can only be made when periodic extensions in the range of the tick vectors occur. These could be predicted by the use of remote sensing satellite data to give adequate prior warning for vaccination and/or the use of, preferably, a ‘pour-on’ synthetic pyrethroid acaricide for tick control.
Incidental losses from NSD in endemic areas do not warrant the application of either tick control measures, nor prophylactic vaccination aimed specifically at this disease. Control of NSD by vaccination should therefore be directed toward individual animals or special groups at risk. The introduction of stud animals is an example, or whenever sheep and goats are moved from ‘clean’ into NSD endemic areas.5
Two types of vaccine have been developed — a modified live virus (MLV) vaccine containing virus attenuated by serial mouse brain passage24 and an inactivated oil-adjuvant vaccine.10 They have been used experimentally and for various reasons have not been used extensively in the field. Although a single dose of MLV vaccine rapidly induces immunity, this strain of virus has lost some immunogenicity and annual revaccination is necessary to maintain full protection of sheep. The MLV strain induces viraemia, but reversion to virulence is not possible as the strain cannot be transmitted by R. appendiculatus. Two doses of the inactivated vaccine given at a one-month interval are required for optimal stimulation of antibody production, and the duration of immunity appears to be limited. This vaccine was developed to vaccinate animals being moved into endemic areas.
References
- COACKLEY, W. & PINNI, A., 1965. The effect of Nairobi sheep disease virus on tissue culture systems. Journal of Pathology and Bacteriology, 90, 672–675.
- DAUBNEY, R. & HUDSON, J.R., 1931. Nairobi sheep disease. Parasitology, 23, 507–524
- DAUBNEY, R. & HUDSON, J.R., 1934. Nairobi sheep disease: Natural and experimental transmission by ticks other than Rhipicephalus appendiculatus. Parasitology, 26, 496–509
- DAVIES, F.G., 1978. A survey of Nairobi sheep disease antibody in sheep and goats, wild ruminants and rodents within Kenya. Journal of Hygiene, 81, 251–258
- DAVIES, F.G., 1978. Nairobi sheep disease in Kenya. The isolation of virus from sheep and goats, ticks and possible maintenance hosts. Journal of Hygiene, 81, 259–265.
- DAVIES, F.G., 1988. Nairobi Sheep Disease. In: MONATH, T.P., (ed.). The Arboviruses: Epidemiology and Ecology. Vol III. Boca Raton, Florida: CRC Press, Inc.
- DAVIES, F.G., 2002. Unpublished data
- DAVIES, F.G., CASALS, J., JESSETT, D.M. & OCHIENG, P., 1978. The serological relationships of Nairobi sheep disease virus. Journal of Comparative Pathology, 88, 519–523.
- DAVIES, F.G., JESSETT, D.M. & OTIENO, S., 1976. The antibody response of sheep following infection with Nairobi sheep disease virus. Journal of Comparative Pathology, 86, 497–502
- DAVIES, F.G., MUNGAI, J. & SHAW, T., 1974. A Nairobi sheep disease vaccine. The Veterinary Record, 94, 128.
- DAVIES, F.G., MUNGAI, J.N. & TAYLOR, M., 1977. The diagnosis of Nairobi sheep disease. Tropical Animal Health and Production, 9, 75–80.
- DAVIES, F.G. & MWAKIMA, F., 1982. Qualitative studies of the transmission of Nairobi sheep disease virus by Rhipicephalus appendiculatus (Ixodoidea, Ixodidae). Journal of Comparative Pathology, 92, 15–20.
- EDELSTEN, R.M., 1975. The distribution and prevalence of Nairobi sheep disease and other tick-borne infections of sheep and goats in northern Somalia. Tropical Animal Health and Production, 7, 29–34.
- HOWARTH, J.A. & TERPSTRA, C., 1965. The propagation of Nairobi sheep disease virus in tissue culture. Journal of Comparative Pathology, 75, 437–441.
- KARABATSOS, N., 1985. International Catalogue of Arboviruses. 3rd edn. San Antonio, Texas: American Society of Tropical Medicine & Hygiene
- LEWIS, E.A., 1946. Nairobi sheep disease. The survival of the virus in the tick Rhipicephalus appendiculatus. Parasitology, 37, 55–59.
- LEWIS, E.A., 1947. Nairobi sheep disease. Annual Report of the Department of Veterinary Services, Kenya, 45 & 51.
- MONTGOMERY, R.E., 1917. On a tick-borne gastroenteritis of sheep and goats occurring in British East Africa. Journal of Comparative Pathology and Therapeutics, 30, 28–58.
- MUGERA, G.M. & CHEMA, S., 1967. Nairobi sheep disease: A study of its pathogenesis in sheep, goats and suckling mice. Bulletin of Epizootic Diseases in Africa, 15, 337–354.
- MUNDERLOH, U., 1977. Die Nairobi sheep disease — Eine synoptische Darstelling der Erkrankung sowie Untersuchungen zum Verhalten des Erregers in vivo und in vitro. Veterinary Medicine Dissertation, University of München
- MUNZ, E., REIMANN, M. & MEIER, K., 1983. An enzyme-linked immunosorbent assay (ELISA) for the detection of antibodies to Nairobi sheep disease virus in comparison with an indirect immunofluorescent and haemagglutination test. Zentralblat für Veterinärmedizin, B, 30, 473–479.
- PELLEGRINI, D., 1959; La gastro enterite emorragica delle pecore. Experimenti di transmissione con Rhipicephalus pulchellus. Bolletin Soceitie Italien Medicine Igiene Tropicale, 10, 164.
- PEGRUM, R.G., 1976, Ticks (Acarina, Ixodidea) of the Northern regions of the Somalia Democratic Republic. Bulletin Entomological Research, 66, 345..
- PORTERFIELD, J.S., (ed.) 1989. Andrew’s Viruses of Vertebrates. London, Philadelphia: Baillière Tindall..
- TERPSTRA, C., 1967. Serial passaging of attenuated NSD virus in eggs. Annual Report of the Animal Health Research Centre, Entebbe.
- TERPSTRA, C., 1969. Nairobi sheep disease. Studies on virus properties, epizootiology and vaccination in Uganda. Thesis, University of Utrecht
- TERPSTRA, C., 1983. Physical and biological properties of Nairobi sheep disease virus. Veterinary Microbiology, 8, 531–541.
- TERPSTRA, C. & VREESWIJK, H., 1983. Centraal Diergeneeskundig Instituut, Houtrivweg 39, 8221 RA, Lelystad, Holland. Unpublished data.
- WEINBREN, M.P., 1959. Virus neutralizing antibodies in human sera from East Africa. Annual Report of the East African Virus Research Institute, 1958–59, 9, 12–13..
- WEINBREN, M.P., 1967. Personal communication; also in: Catalogue of Arthropod-borne Viruses. No. 110. University of California, Berkeley, USA
- WEINBREN, M.P., GOURLAY, R.N., LUMSDEN, W.H.R. & WEINBREN, B.M., 1958. An epizootic of Nairobi sheep disease in Uganda. The Journal of Comparative Pathology and Therapeutics, 68, 174–187.
- WOODALL, J.P., WILLIAMS, M.C., SANTOS, D.F. & ELLICE, J.M., 1962. Nairobi sheep disease SE 2175. Annual Report of the East African Virus Research Institute, 1961–62, 12, 24.