- Infectious Diseases of Livestock
- Part 2
- Rotavirus infections
- GENERAL INTRODUCTION: PARAMYXOVIRIDAE AND PNEUMOVIRIDAE
- Rinderpest
- Peste des petits ruminants
- Parainfluenza type 3 infection
- Bovine respiratory syncytial virus infection
- Hendra virus infection
- Paramyxovirus-induced reproductive failure and congenital defects in pigs
- Nipah virus disease
- GENERAL INTRODUCTION: CALICIVIRIDAE AND ASTROVIRIDAE
- Vesicular exanthema
- Enteric caliciviruses of pigs and cattle
- GENERAL INTRODUCTION: RETROVIRIDAE
- Enzootic bovine leukosis
- Jaagsiekte
- Visna-maedi
- Caprine arthritis-encephalitis
- Equine infectious anaemia
- GENERAL INTRODUCTION: PAPILLOMAVIRIDAE
- Papillomavirus infection of ruminants
- Papillomavirus infection of equids
- GENERAL INTRODUCTION: ORTHOMYXOVIRIDAE
- Equine influenza
- Swine influenza
- GENERAL INTRODUCTION: CORONAVIRIDAE
- Porcine transmissible gastroenteritis
- Porcine respiratory coronavirus infection
- Porcine epidemic diarrhoea
- Porcine haemagglutinating encephalomyelitis virus infection
- Porcine deltacoronavirus infection
- Bovine coronavirus infection
- Ovine coronavirus infection
- Equine coronavirus infection
- GENERAL INTRODUCTION: PARVOVIRIDAE
- Porcine parvovirus infection
- Bovine parvovirus infection
- GENERAL INTRODUCTION: ADENOVIRIDAE
- Adenovirus infections
- GENERAL INTRODUCTION: HERPESVIRIDAE
- Equid herpesvirus 1 and equid herpesvirus 4 infections
- Equid gammaherpesvirus 2 and equid gammaherpesvirus 5 infections
- Equine coital exanthema
- Infectious bovine rhinotracheitis/infectious pustular vulvovaginitis and infectious pustular balanoposthitis
- Bovine alphaherpesvirus 2 infections
- Malignant catarrhal fever
- Pseudorabies
- Suid herpesvirus 2 infection
- GENERAL INTRODUCTION: ARTERIVIRIDAE
- Equine viral arteritis
- Porcine reproductive and respiratory syndrome
- GENERAL INTRODUCTION: FLAVIVIRIDAE
- Bovine viral diarrhoea and mucosal disease
- Border disease
- Hog cholera
- Wesselsbron disease
- Louping ill
- West nile virus infection
- GENERAL INTRODUCTION: TOGAVIRIDAE
- Equine encephalitides caused by alphaviruses in the Western Hemisphere
- Old World alphavirus infections in animals
- Getah virus infection
- GENERAL INTRODUCTION: BUNYAVIRIDAE
- Diseases caused by Akabane and related Simbu-group viruses
- Rift Valley fever
- Nairobi sheep disease
- Crimean-Congo haemorrhagic fever
- GENERAL INTRODUCTION: ASFARVIRIDAE
- African swine fever
- GENERAL INTRODUCTION: RHABDOVIRIDAE
- Rabies
- Bovine ephemeral fever
- Vesicular stomatitis and other vesiculovirus infections
- GENERAL INTRODUCTION: REOVIRIDAE
- Bluetongue
- Ibaraki disease in cattle
- Epizootic haemorrhagic disease
- African horse sickness
- Equine encephalosis
- Palyam serogroup orbivirus infections
- Rotavirus infections
- GENERAL INTRODUCTION: POXVIRIDAE
- Lumpy skin disease
- Sheeppox and goatpox
- Orf
- Ulcerative dermatosis
- Bovine papular stomatitis
- Pseudocowpox
- Swinepox
- Cowpox
- Horsepox
- Camelpox
- Buffalopox
- GENERAL INTRODUCTION: PICORNAVIRIDAE
- Teschen, Talfan and reproductive diseases caused by porcine enteroviruses
- Encephalomyocarditis virus infection
- Swine vesicular disease
- Equine picornavirus infection
- Bovine rhinovirus infection
- Foot-and-mouth disease
- GENERAL INTRODUCTION: BORNAVIRIDAE
- Borna disease
- GENERAL INTRODUCTION: CIRCOVIRIDAE AND ANELLOVIRIDAE
- Post-weaning multi-systemic wasting syndrome in swine
- GENERAL INTRODUCTION: PRION DISEASES
- Scrapie
- Bovine spongiform encephalopathy
- Transmissible spongiform encephalopathies related to bovine spongiform encephalopathy in other domestic and captive wild species
Rotavirus infections
This content is distributed under the following licence: Attribution-NonCommercial CC BY-NC  View Creative Commons Licence details here
View Creative Commons Licence details here
NJ Maclachlan and M-L Penrith (Editors). J O Amimo, A N Vlasova and L J Saif, Rotavirus infections, 2018.

Rotavirus infections
Previous authors: A D STEELE, A GEYER AND G H GERDES
Current authors:
J O AMIMO - Lecturer, DVM, MSc, PhD, Animal Production, University of Nairobi, P.O. Box 29053, Nairobi, 00625, Kenya
A N VLASOVA - Assistant Professor, PhD, DVM, The Ohio State University, 1680 Madison Ave, Wooster, Ohio, 44691, USA
L J SAIF - Professor, MS, PhD, Food Animal Health Research Program, CFAES and CVM, OARDC, The Ohio State University, 1680 Madison Ave, Wooster, Ohio, 44691, USA
Introduction
Rotaviruses are ubiquitous in nature and generally cause infection of the small intestine in the young of many mammal and bird species.50 The disease occurs predominantly in intensively reared animals and is characterized by acute disease, a short incubation period, anorexia and diarrhoea.
Rotavirus infection was first recognized in 1963 in mice,2 followed by isolation of the SA11 (simian agent 11) in vervet monkey cell culture.18, 96 In 1969 virus particles (70 nm in diameter) were detected in bovine faeces and were shown to be associated with diarrhoea in calves.112 These murine,119 simian and bovine agents were later found to share a common group antigen and to be morphologically indistinguishable by transmission electron microscopy (TEM). Bovine rotaviruses (BRV) were among the earliest rotaviruses to be successfully adapted to cell culture110 after which research efforts were directed toward their characterization.
Rotaviruses have a world-wide distribution and commonly affect calves,111, 169 lambs,107, 156 piglets,14, 137, 144 goat kids41 and foals.39, 41
Aetiology
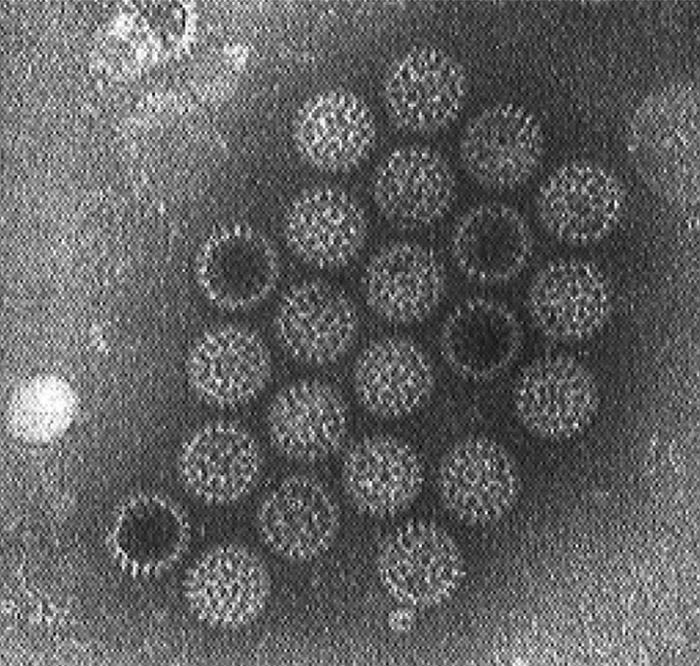
Rotaviruses are now classified in the family Reoviridae, genus Rotavirus,44, 50, 102 and each rotavirus is named after the species in which it occurs.They have a distinct morphology, revealed by TEM of negatively-stained virus in faeces (Figure 1). Those from different species and serogroups are morphologically indistinguishable, i.e. a non-enveloped virion with icosahedral symmetry.59 Three types of particles are evident: viral cores, double-layered and the triple-layered, with the latter constituting the complete infectious particle.
The name rotavirus derives from the Latin ‘rota’ meaning wheel59 because the outer capsid has a sharply defined outline resembling the rim of a wheel approximately 70 nm in diameter.24 The double-layered particle or inner capsid particle is approximately 55 nm in diameter50 while the electron-dense core is hexagonal and approximately 37 nm in diameter.58
The rotavirus genome is composed of eleven segments of double-stranded RNA encoding six structural (VP1–VP6) and six nonstructural (NSP1–NSP6) proteins.50 The segments can be readily separated by polyacrylamide gel electrophoresis (PAGE) producing a characteristic pattern known as an RNA electropherotype.145
The dsRNA segments can be divided into four class sizes (I to IV) based on migration through PAGE. Group A rotaviruses have a characteristic 4-2-3-2 PAGE pattern which is generally conserved among these viruses. However, groups B and C rotaviruses, which are also found in most domesticated livestock, have distinct patterns that allow rapid differentiation of strains from different serogroups.7, 132, 158 The group B rotaviruses have a characteristic RNA electropherotype where the segments cluster in a 4-2-2-3 pattern, and the group C rotaviruses exhibit an electrophoretic pattern of 4-3-2-2.132, 158 The viruses from the different serogroups of A, B and C rotaviruses also have distinct group antigens (see below) that induce non-cross-reactive antibody spectra.
Among livestock species, group A rotaviruses have been implicated in acute gastroenteritis in piglets, lambs, calves, foals, chickens and turkeys.21, 31, 132, 158 Additionally, rotaviruses from groups B and C have been recovered from the faeces of piglets, lambs, calves and foals.32, 158
Considerable antigenic diversity is found among rotaviruses and, historically, isolates were classified into groups based on the viral protein 6 (VP6) antigenic characteristics. They are further subdivided into dual serotype specificities (serotypes) and genotypes on the basis of outer capsid antigens/genes VP7 and VP4 (Table 1). To date, rotaviruses have been classified into at least nine groups/species (designated as RVA–RVI) on the basis of differences in the antigenicity of their middle capsid VP6 protein and nucleotide sequence identities of the VP6-encoding gene.100, 106, 117 A tentative tenth group (RVJ) was reported in bats.9 Among the nine RV groups, RVA, RVB, RVC and RVH are found in both humans and animals, while RVD–RVG have been detected only in animals, including birds, so far (Table 1).
Rotaviruses belonging to the RVA group are the most common cause of viral diarrhoea in a wide variety of animal species and birds and the most diverse group both genetically and antigenically due to point mutations and reassortment of cognate genes.105 However, most recent data also show a significant diversity of RVB and RVC in pigs.6, 97, 101 Thus group A rotaviruses from calves, lambs, piglets and human infants share common or related antigens that are cross-reactive amongst these strains. These are distinct antigenically and by gene sequence from the VP2 and VP6 proteins of the group B or C rotavirus strains.
The major dual classification of rotaviruses (serotypes) is based on the VP7 and VP4 outer capsid antigens. Viral protein 7 (VP7) contains the major neutralizing antigen of the virus and is an important consideration in the selection of viruses for inclusion as vaccine candidates. The VP7 serotypes are distinguished by various neutralization assays such as plaque neutralization79 and monoclonal antibody based enzyme-linked immunosorbent assay (ELISA).155 Molecular techniques based on the viral genomic RNA such as reverse transcription-polymerase chain reaction (RT-PCR) and hybridization are used to detect and differentiate the VP4 and VP7 genotypes within each RV group.
Thus far, at least 28 VP7 genotypes (G-types) and 39 VP4 P genotypes have been reported in the highly genetically diverse group A rotaviruses (Table 2), representing a continuous challenge for vaccine development.141
The dual (G/P) typing system was extended in 2008 to a full-genome sequence classification system, with nucleotide per cent identity cut-off values established for all 11 gene segments , with the notations Gx-P[x]-Ix-Rx-Cx-Mx-Ax-Nx-Tx-Ex-Hx used for the VP7-VP4-VP6-VP1-VP2-VP3-NSP1-NSP2-NSP3-NSP4-NSP5/6 encoding genes, respectively,104 with "x" indicating the numb,/er of the genotype. For example, Wa human RVA is classified as G1-P[8]-I1-R1-C1-M1-A1-N1-T1-E1-H1, OSU porcine RVA is classified as G4-P[6]-I1-R1-C1-M1-A8-N1-T1-E1-H1, while UK (tc) bovine RVA strain is G6-P[5]-I2-R2-C2-M2-A3-N2-T7-E2-H3.
The discovery that viral infectivity was enhanced by the addition of proteolytic enzymes such as pancreatin and trypsin to the cell culture growth medium aided in the isolation of these viruses.7 Use of an embryonic African green monkey kidney cell line (MA-104) with the incorporation of trypsin to the maintenance medium and the use of roller cultures has enabled the routine isolation of most animal RVA but only a few RVB170 and RVC.168
Rotaviruses are stable within a pH range of 3 to 9, in the presence of ether and chloroform and are not easily inactivated. They are resistant to iodophor and quarternary ammonium disinfectants and show an unusually high resistance to inactivation by products containing chlorine and sodium hypochlorite. Ethanol, phenol, formalin and lysol are useful disinfectants.157, 166 In practice 37 per cent formaldehyde diluted 1:10, 0,75 per cent hexachlorophene diluted 1:3, and 67 per cent chloramine T with 16,4 per cent chlorine diluted 1:5 are effective in destroying the virus.
Epidemiology
In calves infection usually occurs from week two to 11 of life but is rare in the first week after birth.23, 54 Although the infection rate may be as high as 79 per cent in calves,42 the proportion of animals that actually develop disease varies.42, 108 The role of non-group A rotavirus infections in calf diarrhoea is unclear although a high proportion of suckled calves have antibodies to group B and C rotaviruses.20, 26
Table 1 Genetic diversity of rotaviruses
| VP6 SUBGROUP | Host species | VP7 GENOTYPE | VP4 GENOTYPE | |
| Group A | 18 genotypes: I1 - I18 | All species including humans | 28 genotypes:designated G1–G28 | 39 genotypes: designated P[1]–[39] |
| Group B | 13 genotypes: I1 - I13 | Cattle, pigs, goats, sheep, rats, humans | 10 genotypes: designated G1 - G20 | Unknown |
| Group C | 10 genotypes: I1-I10 | Cattle, pigs, dogs, ferrets, humans | 10 genotypes: designated G1-G10 | 8 genotypes: designated P[1] - P[8] |
| Groups D, F & G | Unknown | Avian | ||
| Group E | Unknown | Only detected in pigs | ||
| Group H | Unknown | Humans and pigs | ||
| Group I* | Unknown | Only detected in dogs | Unknown | Unknown |
| Group J* | Unknown | Only detected in bats | Unknown | Unknown |
*Provisional classification
Table 2 VP7 serotype specificity of group A rotaviruses and the species of origin and prevalence
| VP7 SEROTYPE | HUMAN STRAINS | ANIMAL SPECIES |
| G1 | Common | Isolated from cattle & pigs |
| G2 | Common | Isolated from cattle |
| G3 | Common | Isolated from pigs, cats & dogs |
| G4 | Isolated | Common pigs |
| G5 | Isolated | Common pigs |
| G6 | Isolated | Common cattle & sheep |
| G7 | Emerging | Birds |
| G8 | Emerging | Isolated from cattle |
| G9 | Isolated | Pigs |
| G10 | Isolated | Common cattle & sheep |
| G11 | Rare | Common pigs |
| G12 | Emerging | – |
| G13 | – | Horses |
| G14 | – | Horses |
| G15 | - | Cattle |
| G16 | - | Horses |
| G17 | - | Birds |
| G18 | - | Birds |
| G19 | - | Birds |
| G20 | Rare | - |
| G21 | - | Isolated from cattle |
| G22 | - | birds |
| G23 | - | Birds |
| G24 | - | Cattle |
| G25 | - | Bat |
| G26 | Emerging/rare | Common in pigs |
| G28 | - | Birds/rodents |
Rotavirus infections cause important economic losses related to deaths, reduction in weight gain and treatment costs of affected animals. Neonatal calf diarrhoea virus (NCDV) and UK bovine prototype strains have been established as G6, whereas B223 and KK-3 are G10.86, 169 G6, G8 or G10 RVAs possessing VP4 genotype P[1], P[5] or P[11] are commonly found in cattle, with G6P[5] being the dominant genotype combination among bovine RVAs.103, 127 In addition, investigators have reported that the G/P types may vary among beef and dairy cattle, with G6 dominant in beef and G10 dominant in dairy cattle.94 Other VP7 genotypes (G1–G4, G5, G11, G12, G15, G17, G21 and G24) and VP4 genotypes (P[3], P[6], P[7], P[14], P[17], P[21], P[29] and P[33]) have been reported occasionally in cattle. Detection of rotavirus serotypes G1, G2, G3, and G11 in faeces of diarrhoeic calves by using polymerase chain reaction-derived cDNA probes has been reported.80 Among the bovine RVAs with novel genotypes, the G21P[29] and G24P[33] strains were isolated from asymptomatic cows.1 Strains RVA/Cow-wt/JPN/Azuk-1/2006/G21P[29] and RVA/Cow-wt/JPN/Dai-10/2007/G24P[33] appeared to be more closely related to simian or canine/feline rotaviruses than bovine rotaviruses, pointing toward possible interspecies transmission and multiple reassortment events involving bovine, simian and canine/feline RVAs.
Ovine RVs are members of either RVA or RVB, but the epidemiology of lamb RVs is still largely uncertain. The occurrence of ovine RVAs has been reported in various countries worldwide with detection rates up to 60 per cent, often with estimated 10 to15 per cent mortality rates. Limited information is available on genotypes of ovine RV strains globally. In India the most prevalent RVA VP7 genotype is G6 followed by G10, while the prevalent VP4 genotype is P[11].61 In China only G10P[15] RVA has been detected in sheep, while G10, G3 and G6 were reported to be prevalent in Spain. In the United Kingdom four lamb RVA strains were characterized, each one showing different specificities, namely G3P[1]; G6P[11]; G9P[8] and G10P[14].56
The occurrence of caprine RVA and RVB has been reported from various countries worldwide. The prevalence of RVA in goats reported in different countries was ~60 per cent in Italy, and 8 per cent and 50 per cent in Spain. Very few epidemiologic studies of RV in goats have been reported in Africa or Asia.
Information on the genotypes of caprine RVA strains globally is limited. Genotypes G6P[1] were reported in Italy and Bangladesh, G6P[14] in South Africa, G3P[3] in Korea, G10P[15] in China, G8 in India and G6P[1] or G8P[1] in Turkey.5, 63, 89, 136 Older lambs are less susceptible than new-born animals to ovine rotavirus but the disease is clinically mild even in lambs less than 10 days old unless aggravated by stress or secondary bacterial infections.159 Non-group A rotaviruses, which may be more common than other rotavirus types, may also be associated with acute fatal enteritis of young lambs.31
Rotaviruses of the RVA group are the main cause of diarrhoea in foals up to three months of age, causing severe economic loss due to morbidity and mortality. Equine RVA strains were first detected in foals with diarrhoea in England in 1975.57 Within the equine RVA, G3 and G14 are the most common VP7 genotypes worldwide and are always associated with the P[12] VP4 genotype.37, 49, 124, 128 In addition, a limited number of porcine-, bovine- and feline-like RVA viruses have been detected in horses36, 57, 85, 167. In a survey in the USA, rotavirus was found in the faeces of 30 per cent of diarrhoea cases in foals,39 but the majority of infections were probably subclinical and were not diagnosed. Seroprevalence studies have shown that almost 100 per cent of animals develop antibodies to rotavirus.20 In foals infection occurs from three days to three months of age.39 Rotavirus antibodies disappear from mares’ milk within seven to 16 days of parturition, and loss of maternal antibodies influences susceptibility of the foal to subsequent infection.39 Large fluctuations in ambient day and night temperatures and crowding of calves are important factors that predispose to outbreaks of the disease.120 Stress and secondary infections with a variety of agents contribute to the severity of the disease.51 Rotavirus infection in calves and piglets may potentiate the effects of Escherichia coli and vice versa.173
Porcine rotavirus (PRV) infection is endemic in pig herds worldwide.176 Antigen detection studies showed that two-thirds of herds are infected with PRV,14 while sero-prevalence studies indicated that almost 100 per cent of animals have been exposed.16 Of the nine RV groups (A-I), RVA, RVB, and RVC are associated with diarrhoea in piglets. Although discovered decades ago, porcine group E RVs (RVE) are uncommon and their pathogenesis has not been well studied. Many rotavirus infections in piglets are subclinical or result in mild diarrhoea lasting one to three days only,10 and diarrhoea caused by RVA is uncommon in the first week of life. Clinical disease is usually observed at one to two weeks of age and up to three to five days post-weaning.14, 88 Rotavirus C and A shedding and disease occur in nursing (one to two weeks of age) and weaned piglets, respectively.14, 40, 177 Reports of mortality are variable and range from seven to 20 per cent in nursing piglets and three to 50 per cent in weaned pigs.14 Recent studies have shown a high and increasing prevalence (up to 78 per cent) of RVC infection in the USA, Ireland, South Korea and Italy.6, 38, 82, 97, 99 Furthermore, in the USA the prevalence of RVC infection was higher than for RVAs in diarrhoeic piglets over three weeks of age.6, 99 In piglets, the most common rotavirus serotypes are G3, G4, G5 and G11.16, 79, 123, 146 Porcine rotavirus serotypes G4 and G5 strains have been isolated globally with G3 strains from Australia123 and Latin America.35 The porcine serotype G3 and G4 strains are antigenically related to the human rotaviruses of the same VP7 serotype. There have been reports of human-like RVA genotype G1 strains in calves,12 and G1 and G2 strains in piglets,131 although the significance of these strains in nature is unclear.
Infection occurs via the faecal–oral route by exposure of susceptible animals to faeces of infected individuals or to fomites. Transmission of the disease is favoured by the presence of large amounts of virus in diarrhoeic faeces (1011/g) and resistance of the virus to environmental conditions. These factors ensure rapid and heavy contamination of the surroundings as well as survival of the virus for prolonged periods.
Cows and sows may excrete virus in the faeces at the time of parturition and provide a source of infection for their offspring.11 However, the usual mode of spread is from neonate to neonate. Experimentally infected calves excrete the virus by the second day of infection and continue to do so for seven to eight days. Under natural conditions rotavirus may be present in the faeces of young animals from three days after birth.11, 42
Pathogenesis
The outcome of infection in all species depends on the virulence of the viral strain, the quantity of virus ingested, the presence of maternally derived antibodies in the lumen of the gut at the time of exposure,14, 30 age-related resistance to the disease.22, 143, 172 and animal management practices. The pathogenesis of RV infections also depends on virus-host interactions and viral gene products (VP3, VP4, VP7, NSP2, NSP3, NSP4).8, 25, 28, 71, 77, 78, 139 Infection occurs shortly after birth but is usually subclinical in the presence of colostral antibodies in the gut.
The virus has an affinity for mature enterocytes at the tips of the villi of the small intestine. As a result of infection, these cells, which have an absorptive function, are desquamated more rapidly than usual and are rapidly replaced by undifferentiated epithelial crypt cells, which have a secretory function.45, 118 Crypt cells lack sucrase or disaccharide enzyme function, leading to inability to utilize lactose. D-xylose malabsorption also occurs as a result of the loss of differentiated enterocytes.183 Undigested carbohydrates in the lumen of the colon are broken down by fermentation bacteria to short-chain fatty acids, giving rise to a hypertonic solution and subsequent osmotic fluid loss.
As a result of damage to the epithelium, the cellular sodium transport system is also disturbed, resulting in a net flow of fluid from the extracellular space into the lumen of the gut. The total faecal output is thus markedly increased with a concurrent loss of Na+ and Cl− ions.115 Animals may die as a result of fluid loss, electrolyte imbalance and concomitant acidosis if appropriate therapy is not instituted.179 Inflammatory changes in the intestine may cause hypermotility and aggravate the diarrhoea.40, 179 Colonization of the bowel by other infectious agents such as E. coli and Salmonella serovars increases the severity of the condition.51, 173, 182 Extra-intestinal spread of RV also occurs frequently as shown by detection of RV dsRNA, RV antigen or infectious RV in serum and other body sites.13, 83, 95, 153 However, the significance of the extra-intestinal occurrence of RV in causation of disease is controversial.52, 138, 140 It has been shown that RV can replicate in the liver, the biliary system and the pancreas, leading to biliary atresia and pancreatitis in immunocompromised murine hosts.53, 64 More recently, vomiting in RV-infected human hosts was explained by the fact that RV can infect the enterochromaffin cells in the gut, stimulating production of serotonin, which activates the afferent fibres of the vagus nerve and stimulates the brain stem sites controlling vomiting.72, 73
An immune evasion mechanism by rotaviruses, mediated by down regulation of interferons and other cytokines, was suggested based on gene expression profiling using microarrays4
Clinical signs
Rotaviral diarrhoea is usually sporadic in occurrence since most infections are in suckling animals with maternal immunity and thus most infections are subclinical. However, in situations with limited or no transfer of maternal immunity or after loss of maternal immunity (weaning) and where other predisposing factors occur simultaneously, the prevalence of disease may reach epidemic proportions.
The incubation period is generally 18 to 96 hours. Affected animals are initially depressed, reluctant to move and anorectic.181, 182 This is followed by profuse diarrhoea, dehydration and loss of body weight. Diarrhoea may be prolonged, up to 14 days in piglets, and severe if the animal recovers sufficiently rapidly to recommence feeding.14, 90, 181 The colour of the faeces depends on the diet and varies from yellow or brownish-grey to light green and seldom contains blood or mucus, unless secondary bacterial infection occurs.159, 182 It is usually an afebrile disease unless complicated by secondary bacterial infection. Affected animals may die as a result of dehydration, electrolyte imbalance and secondary infections.179, 182
Pathology
The only macroscopic changes in animals that have succumbed to this infection are dehydration, an increase in the fluid content of the gut, and presence of diarrhoeic faeces accompanied by contamination of the perineum and hind legs by faeces. Upon necropsy, intestinal walls are thin and filled with yellow fluid. The stomach may be full of undigested milk.
Microscopic changes are largely confined to the villi of the small intestine.111, 133 The cranial portion of the duodenum is generally not affected and there is a patchy distribution of affected areas throughout the rest of the small intestine. Villi appear blunt, short and fused, giving the mucosa an almost avillous appearance.133 Shortening of the villi is due to the loss of brush-border columnar epithelial cells, which are replaced by cuboidal or squamous cells lacking a brush border from the crypts. Increased numbers of mononuclear inflammatory cells may occur in the lamina propria.111, 164
Immunofluorescence studies showed that viral antigens are confined to the cells at the tips of villi in the middle and posterior small intestine of calves and piglets.15, 135 These cells are shed for 18 to 96 hours after infection.40, 154 In lambs, antigen is also present in the enterocytes of the large intestine, but less abundantly than in the small intestine.159 Within infected cells, the virus is associated with the cisternae of the rough endoplasmic reticulum.129
Diagnosis
Diagnosis of rotavirus infection is based on the detection of large numbers of viral particles in the intestinal contents or faeces76, 148, 165 by TEM. Virus isolation was once considered the 'gold standard' for detecting viral pathogens in samples; however, more rapid and sensitive methods are currently available including ELISA and RT-PCR. Cell culture is used to isolate viruses for diagnostic purposes as well as virus propagation for vaccine development or virus genetic characterization. Moreover, it is notable that field strains of RV may fail to replicate in vitro, and especially RVB and RVC. Many cell lines (e.g. MDBK, MA104, TF and PK-15 cells) have been used to isolate RV from animal faecal samples. Viral isolation has three advantages including:
- confirmation of the presence of the virus in a clinical sample,
- availability of the isolated virus for further genetic characterization and development of diagnostic kits and vaccine, and
- does not require virus/strain specific reagents except for confirmation of the virus isolate.
However, besides overall failure to grow in vitro, there are also several downsides to viral isolation including low sensitivity, variable permissiveness of cells, dependence on proper collection and handling of samples for virus viability and non-applicability for cytotoxic specimens.
Transmission electron microscopy applied to negatively- stained faeces or intestinal contents is commonly used for diagnosis because of high concentration of virus in clinical cases.10 This technique has the added advantage of demonstrating other infectious agents in cases of mixed enteric infections. There are two types of TEM: direct TEM and immune electron microscopy (IEM).17, 148 Two different staining techniques (positive and negative staining) can be performed to visualize the target. Immuno-electron microscopy has greater sensitivity than direct TEM since the specimen is incubated with antibody specific for the target virus in order to agglutinate the virus before staining. Most rotavirus geno-groups are difficult to isolate or propagate in cell culture (except group A rotaviruses) but can be differentiated easily from other pathogens according to their unique morphology by TEM.
Direct or indirect fluorescent antibody tests can be used to demonstrate antigen in cell culture, faecal smears and histological sections of the intestine.23, 112
The occurrence of shared, group-specific antigens enables human rotavirus enzyme-linked immunosorbent assay (ELISA) kits, which are available commercially, to be applied to the screening of large numbers of faecal specimens for viral antigen.70, 122 However, generally these kits have not been validated for detection of animal RVs so their sensitivity/specificity are generally undefined. These assays may also be used for serological screening, although high antibody prevalence in most populations negates the diagnostic value of this approach. A rapid, highly specific, and sensitive antigen-capture enzyme-linked immunosorbent assay (AC-ELISA) has been developed for detection of porcine RVA, by using VP6 (a highly conserved and antigenic protein of group-A rotavirus)-directed rabbit polyclonal antibodies (capture antibody) and murine monoclonal antibodies (detector antibody),114 which is advantageous in large-scale surveillance, timely detection and preventive control of rotavirus infections in swine farms. Similar but also G type (VP7) -specific ELISA was developed for detection and G typing of bovine RVA from beef and dairy calves.94
Other tests that were used historically for antigen detection include complement fixation, counter immuno-electrophoresis, radio-immunoassay and agar-gel diffusion; however, their use is less common nowdays.
Polyacrylamide gel electrophoresis (PAGE) of viral RNA extracted from faeces or virus propagated in cell culture is commonly used in epidemiological studies, particularly for differentiating between viral isolates and as a rapid means of detecting atypical rotaviruses in faecal specimens.34, 132, 145, 163
Several molecular detection tools are available. Hybridization tests have been developed using labelled cDNA probes based on hypervariable regions of outer capsid genes of RV that could characterize animal rotavirus strains.125, 130 RT-PCR using validated primers designed from RV genes is currently the most widely used assay for detection of RVs in animals.6, 62, 66-68, 81, 98, 126 Additionally, semi-nested or multiplex RT-PCR has been developed and used for the same purpose.92, 93, 116 RT-PCR is highly sensitive and specific and is suitable for genotyping RV and it has become the gold standard for RV diagnostics.81 Methods like sequencing and oligonucleotide microarray hybridization that are sensitive and capable of discriminating mixed rotavirus infections are also available.
Differential diagnosis
The aetiological diagnosis of neonatal calf diarrhoea is difficult; a reliable diagnosis is impossible in up to 27 per cent of cases.171 A variety of infectious agents, including rotaviruses, coronaviruses, enterotoxigenic E. coli, and cryptosporidia, may cause diarrhoea in neonatal calves.142, 161 Laboratory assistance is thus necessary to arrive at a diagnosis.
Variation in the frequency of rota- and coronavirus detection in beef and dairy calves has been demonstrated, coronavirus being more commonly found in beef calves and rotavirus in dairy animals.27
The differential diagnoses of enteritis in lambs include colibacillosis, salmonellosis, coccidiosis, cryptosporidiosis and adenovirus infections.
Diarrhoea as a result of rotavirus infection in foals should be differentiated from that caused by other infections such as E. coli, Salmonella serovars, Rhodococcus equi, Actinobacillus equuli and Clostridium spp., as well as foal-heat diarrhoea, nutritional factors and internal parasites.
The lesions and pathogenesis of porcine rotavirus diarrhoea closely resemble those of porcine transmissible gastroenteritis (TGE), porcine epidemic diarrhoea (PED) or porcine deltacoronavirus infection caused by coronaviruses.133 Porcine rotavirus diarrhoea should be differentiated from other causes of diarrhoea such as E. coli, internal parasites and nutritional factors. A multiplex RT-PCR has been developed that is reportedly able to differentiate TGEV, PEDV and porcine RVA.163
Control
As death is due to dehydration, loss of electrolytes, acidosis and shock, treatment should primarily be aimed at rehydration and the correction of electrolyte imbalances. Depending on the degree of dehydration, fluid may be given orally or parenterally. Antibiotics may be administered to control secondary bacterial infection. Because of the stability of rotaviruses and the high concentration of virus in the faeces of infected animals, cases should be segregated from non-affected animals, cold-stress and indoor crowding should be avoided and calving/farrowing areas cleaned and rotated if possible. Rotavirus build-up may also occur in the vicinity of outdoor water troughs.
Measures should be taken to enable timely and sufficient ingestion of colostrum, the principal mode of protection. The presence of colostral antibodies in the lumen of the gut (not circulating antibody) is necessary for the protection of neonates.113 Levels of colostral IgG decline rapidly in the transition from colostrum to milk and, in cows, the amounts present by the second week after parturition are no longer protective.48, 162, 178 In sows secretory immunoglobulin (IgA) in milk provides protection and the level remains constant during lactation, but may not be protective if the RV infectious dose is high.147 This differs from the situation in cattle, sheep and horses where colostral immunity is only protective for a short period (~5 days).
During outbreaks of neonatal diarrhoea in calves, the feeding of milk with 10 per cent colostrum from a colostrum pool provides passive protection for the two- to three-week period of risk.3, 47, 150 Thereafter the age-related resistance to clinical infection may provide sufficient protection of older animals. The feeding of artificial colostrum containing serum immunoglobulins, whey and vegetable oils or of IgY egg yolk antibodies175 has been used with good results.121 This strategy is obviously only applicable in bucket-fed dairy calves. If insufficient colostrum is ingested by neonatal lambs, immune serum fed twice daily at 2,5 ml/kg will afford specific protection.162 To boost maternal antibody levels, the dams (cows and ewes) need to be immunized with RV vaccines a few weeks before parturition to enhance passive protection in neonates.3, 150, 165
Oral immunization of calves less than one day old with an attenuated live virus vaccine elicits local cellular resistance.113, 174 Field trials with this vaccine have, nevertheless, been disappointing, as neutralization of vaccine virus by colostral antibodies occurs in the lumen of the gut, making the timing of administration crucial.43, 150
An alternative approach is maternal vaccination of pregnant animals. This has been reviewed by Saif and Fernandez, 1996149 and Chattha et al., 2015.33 Intramuscular immunization of pregnant animals results in increased levels of IgG in the serum and consequently colostrum.29, 134 Studies using a combined inactivated rotavirus-enterotoxigenic E. coli vaccine with an adjuvant resulted in increased immunoglobulin content of colostrum and milk and prolonged passive lactogenic immunity in calves.151, 160 Similarly, the use of a commercial vaccine one to three months before calving significantly reduced the prevalence of rotaviral diarrhoea in an affected herd.109 Although a number of VP7 serotypes of bovine rotavirus have been identified, vaccination of cows usually results in broad immunity with respect to serotype because of the heterotypic response of adult animals, which have usually been previously exposed to a variety of different serotypes. Supplementation with probiotics can prevent rotavirus-induced diarrhoea in young animals.65
If available, a commercial vaccine containing inactivated rotavirus, coronavirus, Clostridium perfringens type C toxoid and K99 E. coli bacterin can be used in pregnant cattle. Two doses are administered two weeks apart, with the second administration two to three weeks before calving. Vaccination should always be accompanied by change in management incorporating attention to factors causing stress and, more particularly, good hygiene.
High immunoglobulin levels in cows’ milk lasting at least 30 days after calving were obtained by the intramuscular or intramammary vaccination of pregnant dairy cows nine weeks prior to calving and by the inoculation of modified live virus together with an adjuvant into the mammary gland two weeks later.151, 152
Bovine colostrum has been used to protect piglets against rotavirus infection,19, 121, 160 although it was found that a vaccine prepared for cattle did not afford protection.91 A commercial porcine rotavirus vaccine has undergone field trials in the USA with variable results.60, 75, 87
Past studies using recombinant DNA and subunit vaccines, plant-based edible vaccines and virus-like particle (VLP) vaccines demonstrated modest to good (2/4/6/7 VLP or combined virus and 2/6VLP vaccines) immunogenicity.46, 55, 69, 74, 84, 180
References
- ABE, M., ITO, N., MASATANI, T., NAKAGAWA, K., YAMAOKA, S., KANAMARU, Y., SUZUKI, H., SHIBANO, K., ARASHI, Y. & SUGIYAMA, M., 2011. Whole genome characterization of new bovine rotavirus G21P[29] and G24P[33] strains provides evidence for interspecies transmission. Journal of General Virology, 92, 952-60.
- ADAMS, W. R. & KRAFT, L. M., 1963. Epizootic diarrhoea of infant mice: Identification of the aetiological agents. Science, 141, 359-360.
- AGRAWAL, D. K., SINGH, N. P. & CHAUHAN, R. S., 2002. Colostral antibodies against rotavirus infection in neonatal calves. Journal of Immunology and Immunopathology, 4, 107-109.
- AICH, P., WILSON, H. L., KAUSHIK, R. S., POTTER, A. A., BABIUK, L. A. & GRIEBEL, P., 2007. Comparative analysis of innate immune responses following infection of newborn calves with bovine rotavirus and bovine coronavirus. Journal of General Virology, 88, 2749-61.
- ALKAN, F., GULYAZ, V., OZKAN TIMURKAN, M., IYISAN, S., OZDEMIR, S., TURAN, N., BUONAVOGLIA, C. & MARTELLA, V., 2012. A large outbreak of enteritis in goat flocks in Marmara, Turkey, by G8P[1] group A rotaviruses. Archives of Virology, 157, 1183-7.
- AMIMO, J. O., VLASOVA, A. N. & SAIF, L. J., 2013. Prevalence and genetic heterogeneity of porcine group C rotaviruses in nursing and weaned piglets in Ohio, USA and identification of a potential new VP4 genotype. Veterinary Microbiology, 164, 27-38.
- BABIUK, L. A., MOHAMMED, K., SPENCE, L., FAUVEL, M. & PETRO, R., 1977. Rotavirus isolation and cultivation in the presence of trypsin. Journal of Clinical Microbiology, 6, 610-617.
- BALL, J. M., TIAN, P., ZENG, C. Q., MORRIS, A. P. & ESTES, M. K., 1996. Age-dependent diarrhea induced by a rotaviral nonstructural glycoprotein. Science, 272, 101-104.
- BANYAI, K., KEMENESI, G., BUDINSKI, I., FOLDES, F., ZANA, B., MARTON, S., VARGA-KUGLER, R., OLDAL, M., KURUCZ, K. & JAKAB, F., 2017. Candidate new rotavirus species in Schreiber's bats, Serbia. Infection, Genetics and Evolution, 48, 19-26.
- BENFIELD, D. A., SCHOLTZ, I. J., NELSON, E. A. & GROON, K. S., 1984. Comparison of a commercial enzyme-linked immunosorbent assay with electron microscopy, fluorescent antibody, and virus isolation for the detection of bovine and porcine rotavirus. American Journal of Veterinary Research, 45, 1998-2002.
- BENFIELD, D. A., STOTZ, I., MOORE, R. & MCADARAGH, J. P., 1982. Shedding of rotavirus in feces of sows before and after farrowing. Journal of Clinical Microbiology, 16, 186-90.
- BLACKHALL, J., BELLIZONI, R. C., MATTION, N., ESTES, M. K., LA TORRE, J. L. & MAGNUSSEN, G., 1992. A bovine rotavirus serotype 1: Serological characterisation of the virus and nucleotide sequence determination of the structural glycoprotein VP7 gene. Virology, 189, 833-837.
- BLUTT, S. E. & CONNER, M. E., 2007. Rotavirus: to the gut and beyond! Current Opinion in Gastroenterology, 23, 39-43.
- BOHL, E. H., 1979. Rotaviral diarrhoea in pigs: Brief review. Journal of the American Veterinary Medical Association, 174, 613-61.
- BOHL, E. H., KOHLER, E. M., SAIF, L. J., CROSS, R. F., AGNES, A. G. & THEIL, K. W., 1978. Rotavirus as a cause of diarrhea in pigs. Journal of the American Veterinary Medical Association, 172, 458-63.
- BOHL, E. H., THEIL, K. W. & SAIF, L. J., 1984. Isolation and serotyping of porcine rotaviruses and antigenic comparison with other rotaviruses. Journal of Clinical Microbiology, 19, 105-111.
- BRANDT, C. D., KIM, H. W., RODRIGUEZ, W. J., THOMAS, L., YOLKEN, R. H., ARROBIO, J. O., KAPIKIAN, A. Z., PARROTT, R. H. & CHANOCK, R. M., 1981. Comparison of direct electron microscopy, immune electron microscopy, and rotavirus enzyme-linked immunosorbent assay for detection of gastroenteritis viruses in children. Journal of Clinical Microbiology, 13, 976-81.
- BRIDGER, J. C., 1986. Novel rotaviruses in animals and man. Ciba Foundation Symposium, 128, 5-23.
- BRIDGER, J. C. & BROWN, J. F., 1981. Development of immunity to porcine rotavirus in piglets protected from disease by bovine colostrums. Infection and Immunity, 31, 906-910.
- BRIDGER, J. C. & BROWN, J. F., 1985. Prevalence of antibodies to typical and atypical rotaviruses in pigs. The Veterinary Record, 116, 50.
- BRIDGER, J. C., CLARK, J. N. & MCCRAE, M. A., 1982. Characterization of an antigenically distinct porcine rotavirus. Infection and Immunity, 35, 1058-1062.
- BRIDGER, J. C. & POCOCK, D. H., 1986. Variation in virulence of bovine rotaviruses. Journal of Hygiene, Cambridge. 96, 257-264.
- BRIDGER, J. C. & WOODE, G. H., 1975. Neonatal calf diarrhoea: Identification of a reovirus-like (rotavirus) agent in faeces by immunofluorescence and immune electron microscopy. British Veterinary Journal, 131, 528-535.
- BRIDGER, J. N. & WOODE, G. N., 1976. Characterisation of two particle types of calf rotavirus. Journal of General Virology, 31, 245-250.
- BROOME, R. L., VO, P. T., WARD, R. L., CLARK, H. F. & GREENBERG, H. B., 1993. Murine rotavirus genes encoding outer capsid proteins VP4 and VP7 are not major determinants of host range restriction and virulence. Journal of Virology, 67, 2448-55.
- BROWN, D. W. G., BEARDS, G. M., CHEN, G. M. & FLEWETT, T. H., 1987. Prevalence of antibody to group B rotavirus in humans and animals. Journal of Clinical Microbiology, 25, 316-319.
- BULGIN, M. S., ANDERSON, B. C., WARD, A. C. S. & EVERMANN, J. F., 1982. Infectious agents associated with neonatal calf diarrhoea in south western Idaho and eastern Oregon. Journal of the American Veterinary Medical Association, 180, 1222-1226.
- BURKE, B. & DESSELBERGER, U., 1996. Rotavirus pathogenicity. Virology, 218, 299-305.
- BUTLER, J. E., 1983. Bovine immunoglobulins: An augmented review. Veterinary Immunology and Immunopathology, 4, 143-152.
- CARPIO, M., BELLAMY, J. E. C. & BABIUK, L. A., 1981. Comparative virulence of different bovine rotavirus isolates. Canadian Journal of Comparative Medicine, 45, 38-42.
- CHASEY, D. & BANKS, J., 1984. The commonest rotaviruses from neonatal lamb diarrhoea in England and Wales have atypical electropherotypes. The Veterinary Record, 115, 326-237.
- CHASEY, D. & DAVIES, P., 1984. A typical rotaviruses in pigs and cattle. The Veterinary Record, 114, 16-17.
- CHATTHA, K. S., ROTH, J. A. & SAIF, L. J., 2015. Strategies for design and application of enteric viral vaccines. Annual Review of Animal Biosciences, 3, 375-395.
- CHAUHAN, R. S. & SINGH, N. P., 1992. Rapid diagnosis of rotavirus infection in calves by dot immunobinding assay. Veterinary Record, 130, 381.
- CIARLET, M., LUDERT, J. E. & LIPRANDI, F., 1995. Comparative amino acid sequence analysis of the major outer capsid protein (VP7) of porcine rotaviruses with G3 and G5 serotype specificities isolated in Venezuela and Argentina. Archives of Virology, 140, 437-451.
- CIARLET, M., P, I. A., CONNER, M. E. & LIPRANDI, F., 2001. Antigenic and molecular analyses reveal that the equine rotavirus strain H-1 is closely related to porcine, but not equine, rotaviruses: interspecies transmission from pigs to horses? Virus Genes, 22, 5-20.
- COLLINS, P. J., CULLINANE, A., MARTELLA, V. & O'SHEA, H., 2008. Molecular Characterization of Equine Rotavirus in Ireland. Journal of Clinical Microbiology, 46, 3346-3354.
- COLLINS, P. J., MARTELLA, V. & O'SHEA, H., 2008. Detection and characterization of group C rotaviruses in asymptomatic piglets in Ireland. Journal of Clinical Microbiology, 46, 2973-9.
- CONNER, M. E. & DARLINGTON, R. W., 1980. Rotavirus infection in foals. American Journal of Veterinary Research, 41, 1699-1703.
- CROUCH, C. F. & WOODE, G. N., 1978. Serial studies of virus multiplication and intestinal damage in gnotobiotic piglets infected with rotavirus. Journal of Medical Microbiology, 11, 325-334.
- DA COSTA MENDES, V. M., DE BEER, M. C., GOOSEN, G. H., ELS, H., THERON, J. & STEELE, A. D., 1994. Rotavirus in Saanen goats. Journal of the South African Veterinary Association, 65, 132-133.
- DE LEEUW, P. W., ELLENS, D. J., STRAVER, P. J., VAN BALKEN, J. A. M., MOERMAN, A. & BAANVINGER, T., 1980. Rotavirus infection in calves in dairy herds. Research in Veterinary Science, 29, 135-141.
- DE LEEUW, P. W., ELLENS, D. J., TALMAN, P. P. & ZIMMER, G. H., 1980. Rotavirus infection in calves: Efficacy of oral vaccination in endemically infected herds. Research in Veterinary Science, 29, 142-147.
- DERBYSHIRE, J. B. & WOODE, G. N., 1978. Classification of rotaviruses: Report from the World Health Organization/Food and Agriculture Organization Comparative Virology Program. Journal of the American Veterinary Medical Association, 173, 519-521.
- DHAMA, K., CHAUHAN, R. S., MAHENDRAN, M. & MALIK, S. V., 2009. Rotavirus diarrhea in bovines and other domestic animals. Veterinary Research Communications, 33, 1-23.
- DHAMA, K., MAHENDRAN, M., GUPTA, P. K. & RAI, A., 2008. DNA vaccines and their applications in veterinary practice: current perspectives. Veterinary Research Communications, 32, 341-56.
- DODET, B., HESELTINE, E., MARY, C. & SALIOU, P., 1997. Rotaviruses in human and veterinary medicine. Trends in Microbiology, 7, 195-199.
- ELLENS, D. J., DE LEEUW, P. W. & STRAVER, P. J., 1978. The detection of rotavirus specific antibody in colostrums and milk by ELISA. Annales de Recherches Vétérinaires, 9, 337-342.
- ELSCHNER, M., SCHRADER, C., HOTZEL, H., PRUDLO, J., SACHSE, K., EICHHORN, W., HERBST, W. & OTTO, P., 2005. Isolation and molecular characterisation of equine rotaviruses from Germany. Veterinary Microbiology, 105, 123-9.
- ESTES, M. K. & KAPIKIAN, A. Z., 2010. Rotaviruses Ovid : Fields Virology. Virology, 1-106.
- EUGESTER, A. K., WHITEFORD, H. W. & MEHR, L. E., 1978. Concurrent rotavirus and salmonella infection in foals. Journal of the American Veterinary Medical Association, 173, 857-858.
- FENAUX, M., CUADRAS, M. A., FENG, N., JAIMES, M. & GREENBERG, H. B., 2006. Extraintestinal spread and replication of a homologous EC rotavirus strain and a heterologous rhesus rotavirus in BALB/c mice. Journal of Virology, 80, 5219-32.
- FENG, N., KIM, B., FENAUX, M., NGUYEN, H., VO, P., OMARY, M. B. & GREENBERG, H. B., 2008. Role of interferon in homologous and heterologous rotavirus infection in the intestines and extraintestinal organs of suckling mice. Journal of Virology, 82, 7578-90.
- FENNER, F., BACHMANN, P. A., GIBBS, E. P. J., MURPHY, F. A., STUDDERT, M. J. & WHITE, D. O., 1987. Veterinary Virology, New York, London, Sydney, Tokyo, Toronto: Academic Press, Inc.
- FERNANDEZ, F. M., CONNER, M. E., HODGINS, D. C., PARWANI, A. V., NIELSEN, P. R., CRAWFORD, S. E., ESTES, M. K. & SAIF, L. J., 1998. Passive immunity to bovine rotavirus in newborn calves fed colostrum supplements from cows immunized with recombinant SA11 rotavirus core-like particle (CLP) or virus-like particle (VLP) vaccines. Vaccine, 16, 507-16.
- FITZGERALD, T. A., MUNOZ, M., WOOD, A. R. & SNODGRASS, D. R., 1995. Serological and genomic characterisation of group A rotaviruses from lambs. Archives of Virology, 140, 1541-8.
- FLEWETT, T. H., BRYDEN, A. S. & DAVIES, H., 1975. Letter: Virus diarrhoea in foals and other animals. Veterinary Record, 96, JMM.
- FLEWETT, T. H., BRYDEN, A. S., DAVIES, H., WOODE, G. H., BRIDGER, J. C. & DERRICK, J. M., 1974. Relationship between virus from acute gastroenteritis of children and newborn calves. Lancet, 2, 61-63.
- FLEWETT, T. H. & WOODE, G. H., 1978. The rotaviruses. A brief review. Archives of Virology, 57, 1-23.
- GARCIA-DIAZ, A., LOPEZ-ANDUJAR, P., RODRIGUEZ DIAZ, J., MONTAVA, R., TORRES BARCELO, C., RIBES, J. M. & BUESA, J., 2004. Nasal immunization of mice with a rotavirus DNA vaccine that induces protective intestinal IgA antibodies. Vaccine, 23, 489-98.
- GAZAL, S., TAKU, A. K. & KUMAR, B., 2012. Predominance of rotavirus genotype G6P[11] in diarrhoeic lambs. Veterinary Journal, 193(1), 299-300. doi: 10.1016/j.tvjl.2011.11.018.
- GENTSCH, J. R., GLASS, R. I., WOODS, P., GOUVEA, V., GORZIGLIA, M., FLORES, J., DAS, B. K. & BHAN, M. K., 1992. Identification of group A rotavirus gene 4 types by polymerase chain reaction. Journal of Clinical Microbiology, 30, 1365-73.
- GHOSH, S., ALAM, M. M., AHMED, M. U., TALUKDAR, R. I., PAUL, S. K. & KOBAYASHI, N., 2010. Complete genome constellation of a caprine group A rotavirus strain reveals common evolution with ruminant and human rotavirus strains. Journal of General Virology, 91, 2367-73.
- GILGER, M. A., MATSON, D. O., CONNER, M. E., ROSENBLATT, H. M., FINEGOLD, M. J. & ESTES, M. K., 1992. Extraintestinal rotavirus infections in children with immunodeficiency. The Journal of Pediatrics, 120, 912-7.
- GILL H. & PRASAD, J., 2008. Probiotics, Immunomodulation, and Health Benefits. In: Bösze Z. (eds). Bioactive Components of Milk, Advances in Experimental Medicine and Biology, 606. Springer, New York, NY.
- GOUVEA, V., ALLEN, J. R., GLASS, R. I., FANG, Z. Y., BREMONT, M., COHEN, J., MCCRAE, M. A., SAIF, L. J., SINARACHATANANT, P. & CAUL, E. O., 1991. Detection of group B and C rotaviruses by polymerase chain reaction. Journal of Clinical Microbiology, 29, 519-23.
- GOUVEA, V., GLASS, R. I., WOODS, P., TANIGUCHI, K., CLARK, H. F., FORRESTER, B. & FANG, Z. Y., 1990. Polymerase chain reaction amplification and typing of rotavirus nucleic acid from stool specimens. Journal of Clinical Microbiology, 28, 276-82.
- GOUVEA, V., SANTOS, N. & TIMENETSKY, M. D. C., 1994. VP4 typing of bovine and porcine group A rotaviruses by PCR. Journal of Clinical Microbiology, 32, 1333-7.
- GRAHAM, K. L., TAKADA, Y. & COULSON, B. S., 2006. Rotavirus spike protein VP5* binds alpha2beta1 integrin on the cell surface and competes with virus for cell binding and infectivity. Journal of General Virology, 87, 1275-83.
- GRAUBALLE, P. C., WESTERGAARD, B. F., MEYLING, A. & GENNER, J., 1981. Optimized ELISA for detection of human and bovine rotavirus in stools. Journal of Medical Virology, 7, 29-40.
- GREENBERG, H. B. & ESTES, M. K., 2009. Rotaviruses: From pathogenesis to vaccination. Gastroenterology, 136, 1939-1951.
- HAGBOM, M., ISTRATE, C., ENGBLOM, D., KARLSSON, T., RODRIGUEZ-DIAZ, J., BUESA, J., TAYLOR, J. A., LOITTO, V. M., MAGNUSSON, K. E., AHLMAN, H., LUNDGREN, O. & SVENSSON, L., 2011. Rotavirus stimulates release of serotonin (5-HT) from human enterochromaffin cells and activates brain structures involved in nausea and vomiting. PLoS Pathogen, 7, e1002115.
- HAGBOM, M., SHARMA, S., LUNDGREN, O. & SVENSSON, L., 2012. Towards a human rotavirus disease model. Current Opinion in Virology, 2, 408-18.
- HERRMANN, J. E., 2006. DNA vaccines against enteric infections. Vaccine, 24, 3705-8.
- HOBLET, K. H., SAIF, L. J., KOHLER, E. M., THEIL, K. W., BECH-NIELSEN, S. & STIZLEIN, G. A., 1986. Efficacy of an orally administered modified live porcine origin rotavirus vaccine against post-weaning diarrhea in pigs. American Journal of Veterinary Research, 47, 1697-1703.
- HOLLAND, R. E., 1990. Some infectious causes of diarrhea in young farm animals. Linical Microbiology Reviews, 3, 345-75.
- HOSHINO, Y. & KAPIKIAN, A. Z., 1996. Classification of rotavirus VP4 and VP7 serotypes. Archives of Virology, Supplementum 12, 99-111.
- HOSHINO, Y., SAIF, L. J., KANG, S. Y., SERENO, M. M., CHEN, W. K. & KAPIKIAN, A. Z., 1995. Identification of group A rotavirus genes associated with virulence of a porcine rotavirus and host range restriction of a human rotavirus in the gnotobiotic piglet model. Virology, 209, 274-80.
- HOSHINO, Y., WYATT, R. G., GREENBERG, H. B., FLORES, J. & KAPIKIAN, A. Z., 1984. Serotypic similarity and diversity of rotaviruses of mammalian and avian origin as studied by plaque neutralization. Journal of Infectious Diseases, 149, 694-702.
- HUSSEIN, H. A., PARWANI, A. V., ROSEN, B. I., LUCCHELLI, A. & SAIF, L. J., 1993. Detection of rotavirus serotypes G1, G2, G3, and G11 in feces of diarrheic calves by using polymerase chain reaction-derived cDNA probes. Journal of Clinical Microbiology, 31, 2491-2496.
- ITURRIZA-GOMARA, M., KANG, G. & GRAY, J., 2004. Rotavirus genotyping: keeping up with an evolving population of human rotaviruses. Journal of Clinical Virology, 31, 259-65.
- JEONG, Y., PARK, S., HOSMILLO, M., SHIN, D., CHUN, Y., KIM, H., KWON, H., KANG, S., WOO, S., PARK, S., KIM, G., KANG, M. & CHO, K., 2009. Detection and molecular characterization of porcine group C rotaviruses in South Korea. Veterinary Microbiology, 138, 217-224.
- KIM, H. J., PARK, J. G., MATTHIJNSSENS, J., LEE, J. H., BAE, Y. C., ALFAJARO, M. M., PARK, S. I., KANG, M. I. & CHO, K. O., 2011. Intestinal and extra-intestinal pathogenicity of a bovine reassortant rotavirus in calves and piglets. Veterinary Microbiology, 152, 291-303.
- KIM, Y., CHANG, K. O., KIM, W. Y. & SAIF, L. J., 2002. Production of hybrid double- or triple-layered virus-like particles of group A and C rotaviruses using a baculovirus expression system. Virology, 302, 1-8.
- KOJIMA, K., TANIGUCHI, K. & KOBAYASHI, N., 1996. Species-specific and interspecies relatedness of NSP1 sequences in human, porcine, bovine, feline, and equine rotavirus strains. Archives of Virology, 141, 1-12.
- KOMOTO, S., PONGSUWANNA, Y., TACHAROENMUANG, R., GUNTAPONG, R., IDE, T., HIGO-MORIGUCHI, K., TSUJI, T., YOSHIKAWA, T. & TANIGUCHI, K., 2016. Whole genomic analysis of bovine group A rotavirus strains A5-10 and A5-13 provides evidence for close evolutionary relationship with human rotaviruses. Veterinary Microbiology, 195, 37-57.
- KOMOTO, S., SASAKI, J. & TANIGUCHI, K., 2006. Reverse genetics system for introduction of site-specific mutations into the double-stranded RNA genome of infectious rotavirus. Proceeding of Natitonal Academy of Science, USA, 103, 4646-51.
- LECCE, J. G. & KING, M. W., 1978. Role of rotavirus (reo-like) in weanling diarrhoea of pigs. Journal of Clinical Microbiology, 8, 454-458.
- LEE, J. B., YOUN, S. J., NAKAGOMI, T., PARK, S. Y., KIM, T. J., SONG, C. S., JANG, H. K., KIM, B. S. & NAKAGOMI, O., 2003. Isolation, serologic and molecular characterization of the first G3 caprine rotavirus. Archives of Virology, 148, 643-57.
- LEECE, J. G., BALSCAUGH, R. K., CLARKE, D. A. & KING, M. W., 1982. Rotavirus and haemolytic enteropathogenic E. coli in weaning diarrhoea of pigs. Journal of Clinical Microbiology, 16, 715-723.
- LEECE, J. G. & KING, M. W., 1979. The calf reo-like virus (rotavirus) vaccine: An ineffective immunization agent for rotaviral diarrhea of piglets. Canadian Journal of Comparative Medicine, 43, 90-93.
- LIU, J., KIBIKI, G., MARO, V., MARO, A., KUMBURU, H., SWAI, N., TANIUCHI, M., GRATZ, J., TONEY, D., KANG, G. & HOUPT, E., 2011. Multiplex reverse transcription PCR Luminexassay for detection and quantitation of viral agents of gastroenteritis. Journal of Clinical Virology, 50 (April (4)), 308–313.
- LUAN, J. J., YANG, S. H., ZHANG, W. J., GAO, Y. D., ZHONG, J. F. & ZHAO, H. K., 2006. Rapid detection of bovine rotavirus with semi-nested RT-PCR assay. Chinese Journal of Zoonoses, 22, 671–673.
- LUCCHELLI, A., KANG, S. Y., JAYASEKERA, M. K., PARWANI, A. V., ZEMAN, D. H. & SAIF, L. J., 1994. A survey of G6 and G10 serotypes of group A bovine rotaviruses from diarrheic beef and dairy calves using monoclonal antibodies in ELISA. Journal of Veterinary Diagnostic Investigation, 6, 175-181.
- LUNDGREN, O. & SVENSSON, L., 2001. Pathogenesis of rotavirus diarrhea. Microbes and Infection, 3, 1145-56.
- MALHERBE, H. H. & STRICKLAND-CHOLMLEY, M., 1967. Simian virus SA II and the related ‘O’ Agent. Archiv für die Gesamte Virusforschung, 22, 235-245.
- MARTELLA, V., BÁNYAI, K., LORUSSO, E., BELLACICCO, A. L., DECARO, N., CAMERO, M., BOZZO, G., MOSCHIDOU, P., ARISTA, S., PEZZOTTI, G., LAVAZZA, A. & BUONAVOGLIA, C., 2007. Prevalence of group C rotaviruses in weaning and post-weaning pigs with enteritis. Veterinary Microbiology, 123, 26-33.
- MARTHALER, D., HOMWONG, N., ROSSOW, K., CULHANE, M., GOYAL, S., COLLINS, J., MATTHIJNSSENS, J. & CIARLET, M., 2014. Rapid detection and high occurrence of porcine rotavirus A, B, and C by RT-qPCR in diagnostic samples. Journal of Virology Methods, 209, 30-34.
- MARTHALER, D., ROSSOW, K., CULHANE, M., COLLINS, J., GOYAL, S., CIARLET, M. & MATTHIJNSSENS, J., 2013. Identification, phylogenetic analysis and classification of porcine group C rotavirus VP7 sequences from the United States and Canada. Virology, 446, 189-198.
- MARTHALER, D., ROSSOW, K., CULHANE, M., GOYAL, S., COLLINS, J., MATTHIJNSSENS, J., NELSON, M. & CIARLET, M., 2014. Widespread rotavirus H in commercially raised pigs, United States. Emerging Infectious Diseases, 20, 1195-8.
- MARTHALER, D., ROSSOW, K., GRAMER, M., COLLINS, J., GOYAL, S., TSUNEMITSU, H., KUGA, K., SUZUKI, T., CIARLET, M. & MATTHIJNSSENS, J., 2012. Detection of substantial porcine group B rotavirus genetic diversity in the United States, resulting in a modified classification proposal for G genotypes. Virology, 433, 85-96.
- MATTHEWS, R. E. F., 1979. The classification and nomenclature of viruses: Summary of results of meetings of the international committee on taxonomy of viruses in the Hague. September 1978. Intervirology, 11, 133-135.
- MATTHIJNSSENS, J., CIARLET, M., MCDONALD, S. M., ATTOUI, H., BÁNYAI, K., BRISTER, J. R., BUESA, J., ESONA, M. D., ESTES, M. K., GENTSCH, J. R., ITURRIZA-GÓMARA, M., JOHNE, R., KIRKWOOD, C. D., MARTELLA, V., MERTENS, P. P., NAKAGOMI, O., PARREÑO, V., RAHMAN, M., RUGGERI, F. M., SAIF, L. J., SANTOS, N., STEYER, A., TANIGUCHI, K., PATTON, J. T., DESSELBERGER, U. & VAN RANST, M., 2011. Uniformity of rotavirus strain nomenclature proposed by the Rotavirus Classification Working Group (RCWG). Archives of Virology, 156, 1397-413.
- MATTHIJNSSENS, J., CIARLET, M., RAHMAN, M., ATTOUI, H., BANYAI, K., ESTES, M. K., GENTSCH, J. R., ITURRIZA-GOMARA, M., KIRKWOOD, C. D., MARTELLA, V., MERTENS, P. P., NAKAGOMI, O., PATTON, J. T., RUGGERI, F. M., SAIF, L. J., SANTOS, N., STEYER, A., TANIGUCHI, K., DESSELBERGER, U. & VAN RANST, M., 2008. Recommendations for the classification of group A rotaviruses using all 11 genomic RNA segments. Archives of Virology, 153, 1621-9.
- MATTHIJNSSENS, J., DESSELBERGER, U., HACKER, J., DOBRINDT, U. & KURTH, R., 2012. Genome Diversity and Evolution of Rotaviruses. Genome Plasticity and Infectious Diseases, Washington, DC, ASM Press, 214-241. DOI 10.1128/9781555817213.ch13.
- MATTHIJNSSENS, J., OTTO, P. H., CIARLET, M., DESSELBERGER, U., VAN RANST, M. & JOHNE, R., 2012 VP6-sequence-based cutoff values as a criterion for rotavirus species demarcation. Archives of Virology, 157, 1177-82.
- MCNULTY, M. S., ALLAN, G. M., PEARSON, G. R., MCFERRAN, J. B., CURRAN, W. L. & MCCRACKEN, R. M., 1976. Reovirus-like agent (rotavirus) from lambs. Infection and Immunity, 14, 1332-1338.
- MCNULTY, M. S. & LOGAN, E. F., 1983. Longitudinal survey of rotavirus infection in calves. The Veterinary Record, 113, 333-335.
- MCNULTY, M. S. & LOGAN, E. F., 1987. Effect of vaccination of the dam on rotavirus infection in young calves. The Veterinary Record, 120, 250-252.
- MEBUS, C. A., KONO, M., UNDERDAHL, N. R. & TWIEHAUS, M. J., 1971. Cell culture propagation of neonatal calf diarrhea (scours) virus. Canadian Veterinary Journal, 12, 69-72.
- MEBUS, C. A., STAIR, E. L., UNDERDAHL, N. R. & TWIEHAUS, M. J., 1971. Pathology of neonatal calf diarrhea induced by a reo-like virus. Veterinary Pathology, 8, 490-505.
- MEBUS, C. A., UNDERDAHL, N. R., RHODES, M. B. & TWIEHAUS, M. J., 1969. Calf diarrhea (scours) reproduced with a virus from a field outbreak. University of Nebraska Agricultural Experimental Station Research Bulletin, 233, 1-16.
- MEBUS, C. A., WHITE, R. G., BASS, E. P. & TWIEHAUS, M. J., 1973. Immunity to neonatal calf diarrhea virus. Journal of the American Veterinary Medical Association, 173, 544-546.
- MEMON, A. M., BHUYAN, A. A., CHEN, F., GUO, X., MENGHWAR, H., ZHU, Y., KU, X., CHEN, S., LI, Z. & HE, Q., 2017. Development and Validation of Monoclonal Antibody-Based Antigen Capture ELISA for Detection of Group A Porcine Rotavirus. Viral Immunology, 30, 264-270.
- MIDDLETON, P. J., 1978. Pathogenesis of rotavirus infection. Journal of the American Veterinary Medical Association, 173, 544-546.
- MIDGLEY, S. E., BÁNYAI, K., BUESA, J., HALAIHEL, N., HJULSAGER, C. K., JAKAB, F., KAPLON, J., LARSEN, L. E., MONINI, M., POLJŠAK-PRIJATELJ, M., POTHIER, P., RUGGERI, F. M., STEYER, A., KOOPMANS, M. & BÖTTIGER, B., 2012. Diversity and zoonotic potential of rotaviruses in swine and cattle across Europe. Veterinary Microbiology, 156, 238-45.
- MIHALOV-KOVÁCS, E., GELLÉRT, Á., MARTON, S., FARKAS, S. L., FEHÉR, E., OLDAL, M., JAKAB, F., MARTELLA, V. & BÁNYAI, K., 2015. Candidate new rotavirus species in sheltered dogs, Hungary. Emerging Infectious Diseases, 21, 660-3.
- MOON, H. W. & JOEL, D. D., 1975. Epithelial cell migration in the small intestine of sheep and calves. American Journal of Veterinary Research, 36, 187-189.
- MUCH, D. H. & ZAJAC, I., 1972. Purification and characterization of epizootic diarrhoea of infant mice virus. Infection and Immunity, 6, 1019-1024.
- MURAKAMI, T., HIRANO, N., CHITOSE, K., TSUCHIYA, K., ONO, K., SATO, F., SUZUKI, Y. & MURUKAMI, Y., 1987. A survey on bovine rotavirus type 1 associated neonatal calf diarrhoea in a beef herd. Japanese Journal of Veterinary Science, 49, 23-30.
- MURAKAMI, T., HIRANO, N., INQUE, A., CHITOSE, K., TSUSHIYA, K., ONO, K., NAITO, Y. & YANAGIHARA, T., 1986. Protective effects of orally administered immunoglobulins against experimental calf diarrhoea. Japanese Journal of Veterinary Science, 48, 237-245.
- MURPHY, F. A., GIBBS, E. P. J., HORZINEK, M. C. & STUDDERT, M. J., 1999. Veterinary Virology.
- NAGESHA, H. S. & HOLMES, I. H., 1988. New porcine rotavirus serotype antigenically related to human rotavirus 3. Journal of Clinical Microbiology, 26, 171-174.
- NEMOTO, M., RYAN, E., LYONS, P. & CULLINANE, A., 2017. Molecular characterisation of equine group A rotaviruses in Ireland (2011-2015). Journal of Veterinary Science, 226, 12-14.
- PALOMBO, E. A., 2002. Genetic analysis of group A rotaviruses: evidence for interspecies transmission of rotavirus genes. Virus Genes, 24, 11-20.
- PANG, X. L., LEE, B., BOROUMAND, N., LEBLANC, B., PREIKSAITIS, J. K. & YU IP, C. C., 2004. Increased detection of rotavirus using a real time reverse transcription-polymerase chain reaction (RT-PCR) assay in stool specimens from children with diarrhea. Journal of Medical Virology, 72, 496-501.
- PAPP, H., LÁSZLÓ, B., JAKAB, F., GANESH, B., DE GRAZIA, S., MATTHIJNSSENS, J., CIARLET, M., MARTELLA, V. & BÁNYAI, K., 2013. Review of group A rotavirus strains reported in swine and cattle. Veterinary Microbiology, 165, 190-9.
- PAPP, H., MATTHIJNSSENS, J., MARTELLA, V., CIARLET, M. & BANYAI, K., 2013. Global distribution of group A rotavirus strains in horses: a systematic review. Vaccine, 31, 5627-33.
- PARSON, G. R. & MCNULTY, M. S., 1979. Ultrastructural changes in small intestinal epithelium of neonatal pigs infected with pig rotavirus. Archives of Virology, 59, 127-136.
- PARWANI, A. V., HUSSEIN, H. A., ROSEN, B. I., LUCCHELLI, A., NAVARRO, L. & SAIF, L. J., 1993. Characterization of field strains of group A bovine rotaviruses by using polymerase chain reaction-generated G and P type-specific cDNA probes. Journal of Clinical Microbiology, 31, 2010-2015.
- PAUL, P. S., LYOO, Y. S., ANDREWS, J. J. & HILL, H. T., 1988. Isolation of two new serotypes of porcine rotavirus from pigs with diarrhoea. Archives of Virology, 100, 139-143.
- PEDLEY, S., BRIDGER, J. C., BROWN, J. F. & MCCRAE, M. A., 1983. Molecular characterization of rotavirus with distinct group antigens. Journal of General Virology, 64, 2093-2120.
- PENSAERT, M. B., HAELTERMAN, E. O. & BURNSTEIN, T., 1970. Transmissible gastroenteritis of swine. Virus intestinal cell reactions. I. Immunofluorescence, histopathology and virus production in the small intestine through the course of infection. Archiv für die Gesamte Virusforschung, 31, 321-334.
- PIERCE, A. E. & FEINSTEIN, A., 1965. Biophysical and immunological studies on bovine immunoglobulins with evidence for selective transport within the mammary gland from maternal plasma to colostrum. Immunology, 8, 106-123.
- POSPISCHIL, A., HESS, R. G. & BACHMANN, P. A., 1981. Light Microscopy and Ultrahistology of Intestinal Changes in Pigs Infected with Epizootic Diarrhoea Virus (EVD): Comparison with Transmissible Gastroenteritis (TGE) Virus and Porcine Rotavirus Infections. Zentralblatt für Veterinärmedizin Reihe B, 28, 564-577.
- PRATELLI, A., MARTELLA, V., TEMPESTA, M. & BUONAVOGLIA, C., 1999. Characterization by polymerase chain reaction of ruminant rotaviruses isolated in Italy. New Microbiologica, 22, 105-9.
- PROSEZKY, L. & THEODORIDIS, A., 1977. Diarrhoea in pigs induced by rotavirus. Onderstepoort Journal of Veterinary Research, 44, 275-278.
- RAMANI, S., SANKARAN, P., ARUMUGAM, R., SARKAR, R., BANERJEE, I., MOHANTY, I., JANA, A. K., KURUVILLA, K. A. & KANG, G., 2010. Comparison of viral load and duration of virus shedding in symptomatic and asymptomatic neonatal rotavirus infections. Journal of Medical Virology, 82, 1803-7.
- RAMIG, R. F., 2004. Pathogenesis of intestinal and systemic rotavirus infection. Journal of Virology, 78, 10213-20.
- RAMIG, R. F., 2007. Systemic rotavirus infection. Expert Review of Anti-infective Therapy, 5, 591-612.
- RCWG, R. C. W. G., 2015. Minutes of the 7th RCWG meeting. https://rega.kuleuven.be/cev/viralmetagenomics/virus-classification/minutes-of-the-7th-rcwg-meeting.
- REYNOLDS, D. J., MORGAN, J. H., CHANTER, N., JONES, P. W., BRIDGER, J. C., DEBNEY, T. G. & BUNCH, K. J., 1986. Microbiology of calf diarrhoea in southern Britain. The Veterinary Record, 119, 34-39.
- RIEPENHOFF-TALTY, M., LEE, P., CARMODY, P. J., BARRET, H. J. & OGRA, P. L., 1982. Age-dependent rotavirus enterocyte interactions. Proceedings of the Society for Experimental Biology and Medicine, 170, 146-154.
- RODGER, S. M., CRAVEN, J. A. & WILLIAMS, I., 1975. Demonstration of virus-like particles in the intestinal contents of piglets with diarrhoea. Australian Veterinary Journal, 51, 536.
- RODGER, S. M. & HOLMES, I. H., 1979. Comparison of the genomes of simian, bovine and human rotaviruses by gel electrophoresis and detection of genomic variation among bovine isolates. Journal of Virology, 30, 839-846.
- RUIZ, A. M., LOPEZ, I. V., LOPEZ, S., ESPEJO, R. T. & ARIAS, C. F., 1988. Molecular and antigenic characterization of porcine rotavirus YM, a possible new rotavirus serotype. Journal of Virology, 62, 4331-4336.
- SAIF, L. J. & BOHL, E. H., 1980. Passive immunity against enteric viral infections. Proceedings of the Third International Symposium on Neonatal Diarrhoea, 6-8 October 1980, University of Saskatchewan, Canada.
- SAIF, L. J., BOHL, E. H., KOHLER, E. M. & HUGHES, J. H., 1977. Immune electron microscopy of transmissible gastroenteritis virus and rotavirus (reovirus-like agent) of swine. American Journal of Veterinary Research, 38, 13-20.
- SAIF, L. J. & FERNANDEZ, F. M., 1996. Group A rotavirus veterinary vaccines. The Journal of Infectious Diseases, 74 Supplement 1, 98-106.
- SAIF, L. J. & FERNANDEZ, F. M., 1996. Group A rotavirus veterinary vaccines. The Journal of Infectious Diseases, 174 Suppl 1, S98-106.
- SAIF, L. J. & SMITH, K. L., 1985. Enteric viral infections of calves and passive immunity. Journal of Dairy Science, 68, 206-228.
- SAIF, L. J., SMITH, K. L., LANDMEIER, B. J., BOHL, E. H., THEIL, K. W. & TODHUNTER, D. A., 1984. Immune response of pregnant cows to bovine rotavirus immunization. The American Journal of Veterinary Research, 45, 49-58.
- SHAO, L., FISCHER, D. D., KANDASAMY, S., RAUF, A., LANGEL, S. N., WENTWORTH, D. E., STUCKER, K. M., HALPIN, R. A., LAM, H. C., MARTHALER, D., SAIF, L. J. & VLASOVA, A. N., 2016. Comparative In Vitro and In Vivo Studies of Porcine Rotavirus G9P[13] and Human Rotavirus Wa G1P[8]. Journal of Virology, 90, 142-51.
- SNODGRASS, D. R., ANGUS, K. W. & GRAY, E. W., 1977. Rotavirus infection in lambs: Pathogenesis and pathology. Archives of Virology, 55, 263-274.
- SNODGRASS, D. R., FITZGERALD, T., CAMPBELL, I., SCOTT, F. M., BROWNING, G. F., MILLER, D. L., HERRING, A. J. & GREENBERG, H. B., 1990. Rotavirus serotypes 6 and 10 predominate in cattle. Journal of Clinical Microbiology, 28, 504-507.
- SNODGRASS, D. R., GRAY, E. W. & HERRING, J. A., 1976. A rotavirus in lambs with diarrhoea. Research in Veterinary Science, 20, 113-114.
- SNODGRASS, D. R. & HERRING, J. A., 1977. The activity of disinfectants on lamb rotavirus. The Veterinary Record, 101, 81.
- SNODGRASS, D. R., HERRING, J. A., CAMPBELL, I., INGLIS, J. M. & HARGREAVES, F. D., 1984. Comparison of a typical rotavirus from calves, piglets, lambs and man. Journal of General Virology, 65, 909-914.
- SNODGRASS, D. R., HERRING, J. A. & GRAY, E. W., 1976. Experimental rotavirus infection in lambs. Journal of Comparative Pathology, 86, 637-642.
- SNODGRASS, D. R., STEWART, J., TAYLOR, J., KRAUTIL, F. L. & SMITH, M. L., 1982. Diarrhoea in dairy calves reduced by feeding colostrum from cows vaccinated with rotavirus. Research in Veterinary Science, 32, 70-73.
- SNODGRASS, D. R., TERZOLO, H. R., SHERWOOD, D., CAMPBELL, I., MENZIES, J. D. & SYNGE, B. A., 1986. Aetiology of diarrhoea in young calves. The Veterinary Record, 119, 31-34.
- SNODGRASS, D. R. & WELLS, P. W., 1978. The immunoprophylaxis of rotavirus infection in lambs. The Veterinary Record, 102, 146-148.
- SONG, D. S., KANG, B. K., OH, J. S., HA, G. W., YANG, J. S., MOON, H. J., JANG, Y.-S. & PARK, B. K., 2006. Multiplex reverse transcription-PCR for rapid differential detection of porcine epidemic diarrhea virus, transmissible gastroenteritis virus, and porcine group A rotavirus. Journal of veterinary diagnostic investigation, 18, 278-281.
- STAIR, E. L., MEBUS, C. A., TWIEHAUS, M. J. & UNDERDAHL, N. R., 1973. Neonatal calf diarrhoea: electron microscopy of intestines infected with a reo-like agent. Veterinary Pathology, 10, 155.
- STEELE, A. D., DE BEER, M. C., GEYER, A., PEENZE, I., BOS, P. & ALEXANDER, J. J., 2004. Epidemiology of rotavirus infection in South Africa. South African Medical Journal, 94, 465.
- TAN, J. A. & SCHNAGL, R., 1981. Inactivation of a rotavirus by disinfectants. Medical Journal of Australia, 1, 19-32.
- TANIGUCHI, K., URASAWA, T. & URASAWA, S., 1993. Independent Segregation Of The Vp4 And The Vp7 Genes In Bovine Rotaviruses As Confirmed By Vp4 Sequence-Analysis Of G8 And G10 Bovine Rotavirus Strains. Journal of General Virology, 74, 1215-1221.
- TERRETT, L. A. & SAIF, L. J., 1987. Serial propagation of porcine group C rotavirus (pararotavirus) in primary porcine kidney cell cultures. Journal of Clinical Microbiology, 25, 1316-9.
- THEODORIDIS, A., PROSEZKY, L. & ELS, H. J., 1979. The isolation and cultivation of calf rotavirus in the Republic of South Africa. Onderstepoort Journal of Veterinary Research, 46, 65-69.
- TSUNEMITSU, H., MORITA, D., TAKAKU, H., NISHIMORI, T., IMAI, K. & SAIF, L. J., 1999. First detection of bovine group B rotavirus in Japan and sequence of its VP7 gene. Archives of Virology, 144, 805-15.
- TZIPORI, S. R., 1985. The relative importance of enteric pathogens affecting neonates of domestic animals. Advances in Veterinary Science and Comparative Medicine, 29, 103-106.
- TZIPORI, S. R., MAKIN, T. J., SMITH, M. L. & KRAUTIL, F. L., 1981. Clinical manifestations of diarrhoea in calves infected with rotavirus and enterotoxigenic E. coli. Journal of Clinical Microbiology, 13, 1011-1016.
- VAN OPDENBOSCH, E., WELLEMANS, G., DEKEGEL, D. & STROBBE, R., 1979. Neonatal calf diarrhoea: A complex viral aetiology. Vlaams Diergeneeskundige Tijdschrift, 48, 512-526.
- VAN ZAANDE, D. I. J. & DE LEEUW, P. W., 1986. Intestinal antibody response after vaccination and infection with rotavirus of calves fed colostrum with or without rotavirus antibody. Veterinary Immunology and Immunopathology, 11, 45-63.
- VEGA, C., BOK, M., CHACANA, P., SAIF, L., FERNANDEZ, F. & PARRENO, V., 2011. Egg yolk IgY: Protection against rotavirus induced diarrhea and modulatory effect on the systemic and mucosal antibody responses in newborn calves. Veterinary Immunology and Immunopathology, 142, 156-69.
- VLASOVA, A., AMIMO, J. & SAIF, L., 2017. Porcine Rotaviruses: Epidemiology, Immune Responses and Control Strategies. Viruses, 9, 48.
- VLASOVA, A., AMIMO, J. & SAIF, L., 2017. Porcine Rotaviruses: Epidemiology, Immune Responses and Control Strategies. Viruses, 9, 48.
- WELLEMANS, G. & VAN OPDENBOSCH, E., 1981. Postpartum antibody levels for rota-, corona- and BVD virus in the cow’s milk. Vlaams Diergeneeskundige Tijdschrift, 50, 46-52.
- WHIPP, S. C., 1978. Physiology of diarrhea — small intestine. Journal of the American Veterinary Medical Association, 173, 662-666.
- WIGDOROVITZ, A., MOZGOVOJ, M., SANTOS, M. J., PARRENO, V., GOMEZ, C., PEREZ-FILGUEIRA, D. M., TRONO, K. G., RIOS, R. D., FRANZONE, P. M., FERNANDEZ, F., CARRILLO, C., BABIUK, L. A., ESCRIBANO, J. M. & BORCA, M. V., 2004. Protective lactogenic immunity conferred by an edible peptide vaccine to bovine rotavirus produced in transgenic plants. Journal of General Virology, 85, 1825-32.
- WOODE, G. H. & BRIDGER, J. C., 1975. Viral enteritis of calves. The Veterinary Record, 96, 85-88.
- WOODE, G. H. & CROUCH, C. F., 1978. Naturally occurring and experimentally induced rotavirus infection of domestic and laboratory animals. Journal of the American Veterinary Medical Association, 173, 522-526.
- WOODE, G. H., SMITH, C. & DENNIS, M. J., 1978. Intestinal damage in rotavirus infected calves assessed by D-xylose malabsorption. The Veterinary Record, 102, 340-341.