- Infectious Diseases of Livestock
- Part 2
- Cowpox
- GENERAL INTRODUCTION: PARAMYXOVIRIDAE AND PNEUMOVIRIDAE
- Rinderpest
- Peste des petits ruminants
- Parainfluenza type 3 infection
- Bovine respiratory syncytial virus infection
- Hendra virus infection
- Paramyxovirus-induced reproductive failure and congenital defects in pigs
- Nipah virus disease
- GENERAL INTRODUCTION: CALICIVIRIDAE AND ASTROVIRIDAE
- Vesicular exanthema
- Enteric caliciviruses of pigs and cattle
- GENERAL INTRODUCTION: RETROVIRIDAE
- Enzootic bovine leukosis
- Jaagsiekte
- Visna-maedi
- Caprine arthritis-encephalitis
- Equine infectious anaemia
- GENERAL INTRODUCTION: PAPILLOMAVIRIDAE
- Papillomavirus infection of ruminants
- Papillomavirus infection of equids
- GENERAL INTRODUCTION: ORTHOMYXOVIRIDAE
- Equine influenza
- Swine influenza
- GENERAL INTRODUCTION: CORONAVIRIDAE
- Porcine transmissible gastroenteritis
- Porcine respiratory coronavirus infection
- Porcine epidemic diarrhoea
- Porcine haemagglutinating encephalomyelitis virus infection
- Porcine deltacoronavirus infection
- Bovine coronavirus infection
- Ovine coronavirus infection
- Equine coronavirus infection
- GENERAL INTRODUCTION: PARVOVIRIDAE
- Porcine parvovirus infection
- Bovine parvovirus infection
- GENERAL INTRODUCTION: ADENOVIRIDAE
- Adenovirus infections
- GENERAL INTRODUCTION: HERPESVIRIDAE
- Equid herpesvirus 1 and equid herpesvirus 4 infections
- Equid gammaherpesvirus 2 and equid gammaherpesvirus 5 infections
- Equine coital exanthema
- Infectious bovine rhinotracheitis/infectious pustular vulvovaginitis and infectious pustular balanoposthitis
- Bovine alphaherpesvirus 2 infections
- Malignant catarrhal fever
- Pseudorabies
- Suid herpesvirus 2 infection
- GENERAL INTRODUCTION: ARTERIVIRIDAE
- Equine viral arteritis
- Porcine reproductive and respiratory syndrome
- GENERAL INTRODUCTION: FLAVIVIRIDAE
- Bovine viral diarrhoea and mucosal disease
- Border disease
- Hog cholera
- Wesselsbron disease
- Louping ill
- West nile virus infection
- GENERAL INTRODUCTION: TOGAVIRIDAE
- Equine encephalitides caused by alphaviruses in the Western Hemisphere
- Old World alphavirus infections in animals
- Getah virus infection
- GENERAL INTRODUCTION: BUNYAVIRIDAE
- Diseases caused by Akabane and related Simbu-group viruses
- Rift Valley fever
- Nairobi sheep disease
- Crimean-Congo haemorrhagic fever
- GENERAL INTRODUCTION: ASFARVIRIDAE
- African swine fever
- GENERAL INTRODUCTION: RHABDOVIRIDAE
- Rabies
- Bovine ephemeral fever
- Vesicular stomatitis and other vesiculovirus infections
- GENERAL INTRODUCTION: REOVIRIDAE
- Bluetongue
- Ibaraki disease in cattle
- Epizootic haemorrhagic disease
- African horse sickness
- Equine encephalosis
- Palyam serogroup orbivirus infections
- Rotavirus infections
- GENERAL INTRODUCTION: POXVIRIDAE
- Lumpy skin disease
- Sheeppox and goatpox
- Orf
- Ulcerative dermatosis
- Bovine papular stomatitis
- Pseudocowpox
- Swinepox
- Cowpox
- Horsepox
- Camelpox
- Buffalopox
- GENERAL INTRODUCTION: PICORNAVIRIDAE
- Teschen, Talfan and reproductive diseases caused by porcine enteroviruses
- Encephalomyocarditis virus infection
- Swine vesicular disease
- Equine picornavirus infection
- Bovine rhinovirus infection
- Foot-and-mouth disease
- GENERAL INTRODUCTION: BORNAVIRIDAE
- Borna disease
- GENERAL INTRODUCTION: CIRCOVIRIDAE AND ANELLOVIRIDAE
- Post-weaning multi-systemic wasting syndrome in swine
- GENERAL INTRODUCTION: PRION DISEASES
- Scrapie
- Bovine spongiform encephalopathy
- Transmissible spongiform encephalopathies related to bovine spongiform encephalopathy in other domestic and captive wild species
Cowpox
This content is distributed under the following licence: Attribution-NonCommercial CC BY-NC  View Creative Commons Licence details here
View Creative Commons Licence details here
NJ Maclachlan and M-L Penrith (Editors). S Babiuk, Cowpox, 2018.
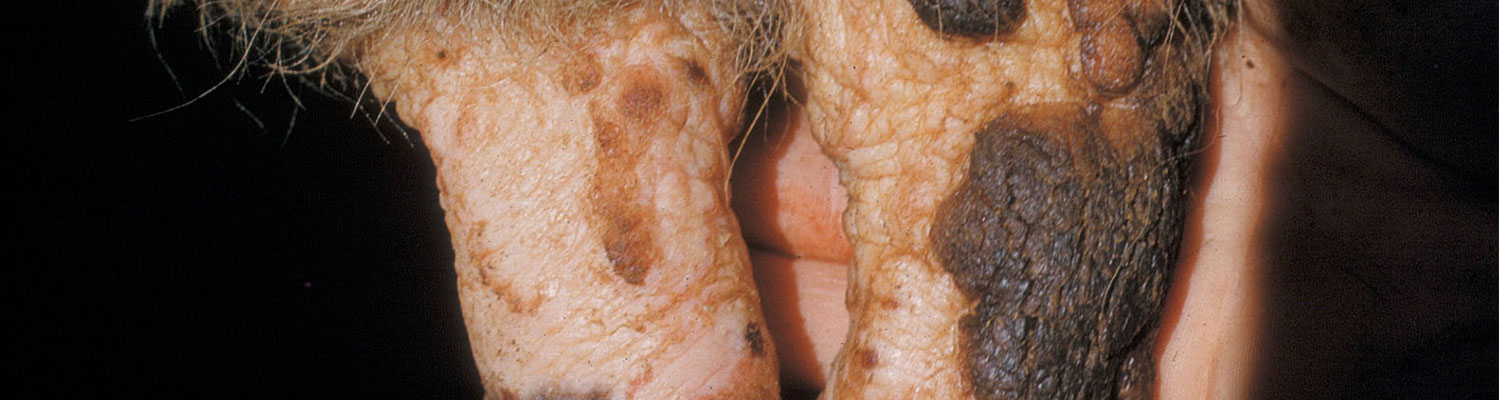
Cowpox
Previous authors: R P KITCHING
Current authors:
S BABIUK - Research Scientific, PhD, National Centre for Foreign Animal Disease, 1015 Arlington Street, Manitoba, Canada, R3E 3MA
Introduction
Cowpox is a rare disease of cattle caused by an orthopox virus and characterized by vesicles, pustules and scabs on the teats and udder. It was the observation by Jenner in 1798 that milkmaids who had been infected with cowpox virus were resistant to smallpox that led to the science of vaccinology.22, 45 Cowpox is a zoonotic disease and humans can be infected by contact with infected animals including the teat lesions during milking, cats, pet rats and when hunting rodents.10, 44, 46 The lack of orthopox virus immunity in humans since vaccination against smallpox ceased, has led to more human infections with cowpox virus, from contact with infected animals predominantly cats.
Aetiology
Orthopox viruses have historically been named for the animal where the initial isolates were recovered from, hence the naming of cowpox. However, further research showed that the name is a misrepresentation since the cow is an accidental host.
Cowpox virus belongs to the genus Orthopoxvirus and the family Poxviridae (see General Introduction: Poxviridae). Orthopox viruses are large, brick-shaped, double-stranded DNA viruses, morphologically indistinguishable from capripox viruses. As with capripox viruses it is probable that under conditions of dual infection with more than one orthopox virus recombination events occur; such is one theory for the origin of vaccinia virus in the smallpox wards of London, as it replaced cowpox virus as the vaccine used against smallpox. Since the animal reservoir for cowpox are rodents, the virus can circulate and possibly recombine with other poxviruses. For example recombination with ectromelia virus resulted in a novel orthopox virus infection in Tonkean macaques that died in Italy. This orthopox virus is a novel clade lying between cowpox and ectromelia viruses.4 Further genetic characterization of this orthopox virus, named abatino, was most closely related to ectromelia with additional genes showing the highest similarity with cowpox indicating that it could be the result of complex evolutionary events.20
Cowpox virus has the largest genome of all the orthopox viruses (more than 220 000 base pairs). It is is also closely related genomically and antigenically to camelpox and buffalopox viruses.27 Phylogenetic analysis showed that the CPXV strains sequenced clearly cluster into several distinct clades, some of which are closely related to vaccinia viruses while others represent different clades in a CPXV cluster.8, 14 Using genomic data available on cowpox, phylogenetic analysis identified five monophyletic clades of cowpox viruses.31
Cowpox virus is sensitive to lipid solvents as found in detergents and to most disinfectants such as those containing hypochlorite, quaternary ammonium compounds or phenolics. It is also inactivated by direct sunlight. However, it can persist for many months in the dark of an infected cowshed.
Epidemiology
The natural and experimental host range for cowpox is very broad. The primary hosts of the virus are small rodents such as wild ground squirrels, gerbils, common voles and wood mice2, 12, 24, 34 in Europe and Asia. Rats are also susceptible to cowpox infection.26 Cowpox has also been reported in wild and domestic felids and in a variety of animal species in zoological collections including the larger members of the feline family, such as the cheetah, panther, lion, puma, jaguar, ocelot and lynx,as well as anteater, elephant, rhinoceros and banded mongoose.5, 21, 37 Dogs and related species are more resistant,40 but there is one reported case in which a dog spread the virus to a human.1, 7 Cowpox virus can also infect a variety of monkey species including squirrel monkeys (leading to death),19 new world monkeys (including Callithrix jacchus, C. penicillata, C. geoffroyi, Saimiri sciureus, Callimico goeldii), and tamarin species (Saguinus oedipus, S. fuscicollis, S. nigricollis, S. midas, and S. labiatus),30 Barbary macaques, pig- tailed macaques, Japanese macaques, cynomolgus macaques and rhesus macaques.29 Recently cowpox outbreaks occurred in alpaca herds in Germany35 and llamas in Italy.3, 38 Cowpox has been reported also in an aborted foal.13
Cowpox is endemic in Europe and Northern and Central Asia.11 It was probably rare, even in the time of Jenner. Now it is very rarely reported as a disease of cattle, and is more commonly found in cats and zoo animals. The virus can spread between cattle by contaminated milking machines, the hands of milkers and udder cloths. Teat injuries, which allow the virus to penetrate the skin, predispose to outbreaks. Biting flies may also mechanically transmit the virus.
Outbreaks of a vaccinia virus (Cantagalo virus) infections in cattle and humans have been reported in 1999 in Brazil and have been recurring in agricultural areas throughout the country9 and the Amazon region of Colombia.43 The reservoir of these outbreaks of vaccinia virus infections was likely rodents. Cats in Brazil have tested positive for vaccinia virus by PCR, but theydid not develop clinical disease as cats do following infection with cowpox virus.6 The virus may be introduced into a cattle herd by rodents, the natural hosts of the cowpox virus, or by infected bought-in cattle. However, the virus does not cause a persistent infection, and recovery results in solid immunity. Following the introduction of an infected cow into a susceptible dairy herd, depending on the level of hygiene and pre-existent teat injuries, the virus may spread through the herd. Subsequently only the first-calf heifers will develop disease. The presence of cowpox can reduce milk yields by making milking difficult, and predispose to secondary mastitis. The milk from affected cows is safe to drink following pasteurization.
Pathogenesis, clinical signs and pathology
After an incubation period of three to six days, a small red macule is seen in the skin, usually of a teat, at the site of viral entry. This develops into a solid, light-coloured papule surrounded by a red, hyperaemic zone, and then into a yellow vesicle with a depressed centre which develops into a pustule, and eventually a thick, red scab, approximately 10 to 20mm diameter, forms. In milking cows, the vesicle quickly ruptures, leaving a deep ulcer. Secondary lesions may develop on the same and other teats and in the skin at the base of the udder. Occasionally it spreads to the inside of the thighs and perineum, and onto the muzzles of calves suckling infected cows. In the absence of secondary bacterial infections, lesions heal in two weeks.
Cowpox virus infection in wild and domestic cats usually presents as an infected bite wound. However, a secondary nodular body rash appears ten days later and the nodules may ulcerate. These are replaced by scabs, which resolve in six to eight weeks. Secondary pneumonia may complicate the clinical picture,23 particularly in the larger wild feline species. A fatal form has been reported in a domestic cat simultaneously infected with feline immunodeficiency virus, but this may have been coincidental, as many nonfatal dual infections have been encountered.
Human infection with cowpox virus was rare in the past when vaccination against smallpox was performed. However, since the cessation of smallpox vaccination, orthopox virus immunity is not present in most of the populations with the result that orthopox virus infections are becoming more common.11 Clinical disease caused by cowpox is usually localized in the skin of the hands and forearms at the site of introduction. In some cases, however, it may spread to the mucous membranes of the eyes and cause mild systemic symptoms including lymphadenopathy. Those suffering from infection with the human immunodeficiency virus may be at greater risk.1, 7, 38 Cowpox virus infection can be fatal in immunosuppressed individuals.17 A laboratory-aquired human cowpox virus infection occurred in the United States.32
Histopathology of cowpox infection in 3D human skin models revealed necrosis, ballooning degeneration, intracytoplasmic inclusion bodies, and immunohistochemistry staining of antigen in keratinocytes and some fibroblasts.41
Diagnosis
When vaccinia virus was still being used to protect humans against smallpox it was not unusual to find that it had spread from a milker to the udder of a cow.
A rapid diagnosis can be achieved by demonstrating the orthopox virions by transmission electron microscopy (TEM) in specimens from skin lesions. Vaccinia virus is morphologically indistinguishable from cowpox virus andparapox virus is morphologically distinct from orthopox viruses by TEM (see Orf Figure).
Cowpox virus can be grown in tissue cultures of rabbit kidney and Vero cells, and in the chorioallantoic membrane of embryonated hen’s eggs causing red haemorrhagic pocks lesions. It can infect laboratory rodents and rabbits. Unlike vaccinia it will not grow on chicken-feather follicle cell cultures. In stained histological sections of skin lesions and in tissue culture cells intracytoplasmic inclusion bodies are present. Orthopox viruses can be detected using immunofluorescence or enzyme-linked immunosorbent assay21, 25 and identified using restriction endonuclease digest patterns.27, 33, 36, 42
Molecular methods can be applied to detect and identify cowpox virus. Real-time PCR assays for cowpox virus16, 28 and a multiplex real-time PCR for cowpox-, variola-, monkeypox- and vaccinia viruses have been developed.39 Since the skin lesions caused by cowpox are similar to those caused by other poxvirus infections a novel HRM assay has been developed for simultaneous detection and differentiation of cowpox-, camelpox-, capripox-, orf-, pseudocowpox- and bovine papular stomatitis viruses.18 Full genome sequencing can also be performed on an isolate to confirm the monophyletic clade that of the virus belongs to.
Immunohistochemistry can be used to identify orthopox virus. The virus neutralization assay can be improved by using a fluorescently labelled anti-vaccinia antibody.15
Differential diagnosis
The differential diagnosis for teat lesions in cattle includes foot-and-mouth disease, vaccinia virus infection, pseudocowpox, bovine ulcerative mammillitis, staphylococcal and corynebacterial infections (black pox, udder impetigo) and papillomavirus infection (wart). Pseudocowpox and bovine ulcerative mammillitis are relatively common, caused by parapox virus and herpesvirus infection respectively. Because secondary infection and milking machine trauma quickly modify the appearance of teat lesions, laboratory support for definitive diagnosis is required.
Control
There are no specific vaccines, although vaccinia virus could be used. Treatment is directed at preventing secondary infections and inactivating the virus in the environment and on the skin with antiseptics, particularly those incorporating a mild detergent. Milking machines and the hands of those using them or milking manually should be disinfected between cows. Flies must be controlled to prevent spread. Infected animals should be milked separately or, at least, last.
References
- BAXBY, D. & BENNETT, M., 1994. Human cowpox 1969–93: A review based on 54 cases. British Journal of Dermatology, 131, 598–607.
- BENNETT, M., CROUCH, A. J., BEGON, M., DUFFY, B., FEORE, S., GASKELL, R. M., KELLY, D. F., MCCRACKEN, C. M., VICARY, L. & BAXBY, D., 1997. Cowpox in British voles and mice. Journal of Comparative Pathology, 116, 35-44.
- CARDETI, G., BROZZI, A., ELENI, C., POLICI, N., D'ALTERIO, G., CARLETTI, F., SCICLUNA, M. T., CASTILLETTI, C., CAPOBIANCHI, M. R., DI CARO, A., AUTORINO, G. L. & AMADDEO, D., 2011. Cowpox virus in llama, Italy. Emerging Infectious Diseases, 17, 1513-5.
- CARDETI, G., GRUBER, C. E. M., ELENI, C., CARLETTI, F., CASTILLETTI, C., MANNA, G., ROSONE, F., GIOMBINI, E., SELLERI, M., LAPA, D., PURO, V., DI CARO, A., LORENZETTI, R., SCICLUNA, M. T., GRIFONI, G., RIZZOLI, A., TAGLIAPIETRA, V., DE MARCO, L., CAPOBIANCHI, M. R. & AUTORINO, G. L., 2017. Fatal Outbreak in Tonkean Macaques Caused by Possibly Novel Orthopoxvirus, Italy, January 2015. Emerging Infectious Diseases, 23, 1941–1949.
- CHANTREY, J., MEYER, H., BAXBY, D., BEGON, M., BOWN, K. J., HAZEL, S. M., JINES, T., MONTGOMERY, W. I. & BENNETT, M., 1999. Cowpox: Reservoir hosts and geographic range. Epidemiology and Infection, 122, 455-460.
- COSTA, G. B., MIRANDA, J. B., ALMEIDA, G. G., SILVA DE OLIVEIRA, J., PINHEIRO, M. S., GONÇALVES, S. A., PIMENTA DOS REIS, J. K., GONÇALVES, R., FERREIRA, P. C., BONJARDIM, C. A., ABRAHÃO, J. S., KROON, E. G. & TRINDADE, G. S., 2017. Detection of Vaccinia Virus in Urban Domestic Cats, Brazil. Emerging Infectious Diseases, 23, 360-362.
- CZERNY, C. P., ZELLER-LUE, C., EIS-HUEBINGER, A. M., KAADEN, O. R. & MEYER, H., 1997. Characterization of a cowpox-like orthopox virus which had caused a lethal infection in man. Archives of Virology Supplement, 0 (13), 13–24.
- DABROWSKI, P., RADONIC, A., KURTH, A. & NITSCHE, A., 2013. Genome-wide comparison of cowpox viruses reveals a new clade related to Variola virus. PloS One, 8:e79953.
- DAMASO, C., ESPOSITO, J. J., CONDIT, R. C. & MOUSSATCHÉ, N., 2000. An emergent poxvirus from humans and cattle in Rio de Janeiro State: Cantagalo virus may derive from Brazilian smallpox vaccine. Virology, 277, 439-49.
- ELSENDOORN, A., AGIUS, G., LE MOAL, G., AAJAJI, F., FAVIER, A. L., WIERZBICKA-HAINAULT, E., BÉRAUD, G., FLUSIN, O., CRANCE, J. M. & ROBLOT, F., 2011. Severe ear chondritis due to cowpox virus transmitted by a pet rat. Journal of Infection, 63, 391-3.
- ESSBAUER, S., M, P. & MEYER, H., 2010. Zoonotic poxviruses. Veterinary Microbiology, 140, 229-36.
- FEORE, S., BENNETT, M., CHANTREY, J., JONES, T., BAXBY, D. & BEGON, M., 1997. The effect of cowpox virus infection on fecundity in bank voles and wood mice. Proceedings. Biological sciences, 264, 1457-61.
- FRANKE, A., KERSHAW, O., JENCKEL, M., KÖNIG, L., BEER, M., HOFFMANN, B. & HOFFMANN, D., 2016. Fatal Cowpox Virus Infection in an Aborted Foal. Vector-Borne and Zoonotic Diseases, 16, 431-3.
- FRANKE, A., PFAFF, F., JENCKEL, M., HOFFMANN, B., HÖPER, D., ANTWERPEN, M., MEYER, H., BEER, M. & HOFFMANN, D., 2017. Classification of Cowpox Viruses into Several Distinct Clades and Identification of a Novel Lineage. Viruses, 9. pii: E142.
- GATES, I., OLSON, V., SMITH, S., PATEL, N., DAMON, I. & KAREM, K., 2015. Development of a High-Content Orthopoxvirus Infectivity and Neutralization Assays. PloS One, 10:e0138836.
- GAVRILOVA, E. V., SHCHERBAKOV, D. N., MAKSYUTOV, R. A. & SHCHELKUNOV, S. N., 2010. Development of real-time PCR assay for specific detection of cowpox virus. Journal of Clinical Virology, 49, 37-40.
- GAZZANI, P., GACH, J. E., COLMENERO, I., MARTIN, J., MORTON, H., BROWN, K. & MILFORD, D. V., 2017. Fatal disseminated cowpox virus infection in an adolescent renal transplant recipient. Pediatric Nephrology, 32, 533-536.
- GELAYE, E., MACH, L., KOLODZIEJEK, J., GRABHERR, R., LOITSCH, A., ACHENBACH, J. E., NOWOTNY, N., DIALLO, A. & LAMIEN, C. E., 2017. A novel HRM assay for the simultaneous detection and differentiation of eight poxviruses of medical and veterinary importance. Scientific Reports, 7, 42892.
- GIRLING, S., PIZZI, R., COX, A. & BEARD, P. M., 2011. Fatal cowpox virus infection in two squirrel monkeys (Saimiri sciureus). The Veterinary Record, 169, 156.
- GRUBER, C., GIOMBINI, E., SELLERI, M., TAUSCH, S. H., ANDRUSCH, A., TYSHAIEVA, A., CARDETI, G., LORENZETTI, R., DE MARCO, L., CARLETTI, F., NITSCHE, A., CAPOBIANCHI, M. R., IPPOLITO, G., AUTORINO, G. L. & CASTILLETTI, C., 2018. Whole Genome Characterization of Orthopoxvirus (OPV) Abatino, a Zoonotic Virus Representing a Putative Novel Clade of Old World Orthopoxviruses. Viruses, 10. pii: E546.
- HAZEL, S. M., BENNETT, M., CHANTREY, J., BOWN, K., CAVANAGH, R., JONES, T. R., BAXBY, D. & BEGON, M., 2000. A longitudinal study of an endemic disease in its wildlife reservoir: Cowpox and wild rodents. Epidemiology and Infection, 124, 551–562.
- HILLEMAN, M. R., 2000. Vaccines in historic evolution and perspective: A narrative of vaccine discoveries. Journal of Human Virology, 3, 63–76.
- HINRICHS, U., VAN DE POEL, H. & VAN DEN INGH, T., 2000. Necrotizing pneumonia in a cat caused by an orthopox virus. Journal of Comparative Pathology, 121, 191–196.
- HOFFMANN, D., FRANKE, A., JENCKEL, M., TAMOŠIUNAITE, A., SCHLUCKEBIER, J., GRANZOW, H., HOFFMANN, B., FISCHER, S., ULRICH, R. G., HÖPER, D., GOLLER, K., OSTERRIEDER, N. & BEER, M., 2015. Out of the Reservoir: Phenotypic and Genotypic Characterization of a Novel Cowpox Virus Isolated from a Common Vole. Journal of Virology, 89, 10959-69.
- JANA, D. & MEHROTRA, M. L., 2000. Standardization of a dot enzyme linked immunosorbent assay for the serodiagnosis of experimental cowpox virus infection and humoral immunity in rabbits. Indian Veterinary Journal, 77, 281–284.
- KALTHOFF, D., KÖNIG, P., MEYER, H., BEER, M. & HOFFMANN, B., 2011. Experimental cowpox virus infection in rats. Veterinary Microbiology, 153, 382-6.
- LOPAREV, V. N., MASSUNG, R. F., ESPOSITO, J. J. & MEYER, H., 2001. Detection and differentiation of Old World orthopoxviruses: Restriction fragment length polymorphism of the crmB gene region. Journal of Clinical Microbiology, 39, 94–100.
- MAKSYUTOV, R. A., GAVRILOVA, E. V., MEYER, H. & SHCHELKUNOV, S. N., 2015. Real-time PCR assay for specific detection of cowpox virus. Journal of Virological Methods, 211, 8-11.
- MARTINA, B., VAN DOORNUM, G., DORRESTEIN, G. M., NIESTERS, H. G., STITTELAAR, K. J., WOLTERS, M. A., VAN BOLHUIS, H. G. & OSTERHAUS, A. D., 2006. Cowpox virus transmission from rats to monkeys, the Netherlands. Emerging Infectious Diseases, 12, 1005-7.
- MÄTZ-RENSING, K., ELLERBROK, H., EHLERS, B., PAULI, G., FLOTO, A., ALEX, M., CZERNY, C. P. & KAUP, F. J., 2006. Fatal poxvirus outbreak in a colony of New World monkeys. Veterinary Pathology, 43, 212-8.
- MAULDIN, M., ANTWERPEN, M., EMERSON, G. L., Y, L., ZOELLER, G., CARROLL, D. S. & MEYER, H., 2017. Cowpox virus: What's in a Name? Viruses, 9. pii: E101.
- MCCOLLUM, A., AUSTIN, C., NAWROCKI, J., HOWLAND, J., PRYDE, J., VAID, A., HOLMES, D., WEIL, M. R., LI, Y., WILKINS, K., ZHAO, H., K, S. S., KAREM, K., REYNOLDS, M. G. & DAMON, I. K., 2012. Investigation of the first laboratory-acquired human cowpox virus infection in the United States. The Journal of Infectious Diseases, 206, 63-8.
- MEYER, H., SCHAY, C., MAHNEL, H. & PFEFFER, M., 1999. Characterization of orthopoxviruses isolated from man and animals in Germany. Archives of Virology, 144, 491–501.
- OLDAL, M., SIRONEN, T., HENTTONEN, H., VAPALAHTI, O., MADAI, M., HORVÁTH, G., DALLOS, B., KUTAS, A., FÖLDES, F., KEMENESI, G., NÉMETH, V., BÁNYAI, K. & JAKAB, F., 2015. Serologic survey of orthopoxvirus infection among rodents in hungary. Vector-Borne and Zoonotic Diseases, 15, 317-22.
- PRKNO, A., HOFFMANN, D., GOERIGK, D., KAISER, M., VAN MAANEN, A. C. F., JESKE, K., JENCKEL, M., PFAFF, F., VAHLENKAMP, T. W., BEER, M., ULRICH, R. G., STARKE, A. & PFEFFER, M., 2017. Epidemiological Investigations of Four Cowpox Virus Outbreaks in Alpaca Herds, Germany. Viruses, 9. pii: E344.
- ROPP, S., JIN, Q., KNIGHT, J. C., MASSUNG, R. F. & ESPOSITO, J. J., 1995. PCR strategy for identification and differentiation of small pox and other orthopoxviruses. Journal of Clinical Microbiology, 33, 2069-76.
- SCHMIEDEKNECHT, G., EICKMANN, M., KÖHLER, K., HERDEN, C. E., KOLESNIKOVA, L., FÖRSTER, C., BURKHARDT, E. H., KÖNIG, M., THIEL, M. & REINACHER, M., 2010. Fatal cowpox virus infection in captive banded mongooses (Mungos mungo). Veterinary Pathology, 47, 547-52.
- SCHUPP, P., PFEFFER, M., MEYER, H., BURCK, G., KOLMEL, K. & NEUMANN, C., 2001. Cowpox virus in a 12-year-old boy: Rapid identification by an orthopoxvirus-specific polymerase chain reaction. British Journal of Dermatology, 145, 146–150.
- SHCHELKUNOV, S. N., SHCHERBAKOV, D. N., MAKSYUTOV, R. A. & GAVRILOVA, E. V., 2011. Species-specific identification of variola, monkeypox, cowpox, and vaccinia viruses by multiplex real-time PCR assay. Journal of Virological Methods, 175, 163-9.
- SMITH, K. C., BENNETT, M. & GARRETT, D. C., 1999. Skin lesions caused by orthopoxvirus in a dog. Journal of Small Animal Practice, 40, 495–497.
- TAMOSIUNAITE, A., HOFFMANN, D., FRANKE, A., SCHLUCKEBIER, J., TAUSCHER, K., TISCHER, B. K., BEER, M. & KLOPFLEISCH OSTERRIEDER, N., 2016. Histopathological and Immunohistochemical Studies of Cowpox Virus Replication in a Three-Dimensional Skin Model. Journal of Comparative Pathology, 155, 55-61.
- TRYLAND, M., MYRMEL, H., HOLTET, L., HAUKENES, G. & TRAAVIK, T., 1998. Clinical cowpox cases in Norway. Scandinavian Journal of Infectious Diseases, 30, 301–303.
- USME-CIRO, J., PAREDES, A., WALTEROS, D. M., TOLOSA-PÉREZ, E. N., LAITON-DONATO, K., PINZÓN, M. D., PETERSEN, B. W., GALLARDO-ROMERO, N. F. L. Y., WILKINS, K., DAVIDSON, W., GAO, J., PATEL, N., NAKAZAWA, Y., REYNOLDS, M. G., SATHESHKUMA, R. P. S., EMERSON, G. L. & PÁEZ-MARTÍNEZ, A., 2017. A. Detection and Molecular Characterization of Zoonotic Poxviruses Circulating in the Amazon Region of Colombia, 2014. Emerging Infectious Diseases, 23, 649-653.
- VOGEL, S., SÁRDY, M., GLOS, K., KORTING, H. C., RUZICKA, T. & WOLLENBERG, A., 2012. The Munich outbreak of cutaneous cowpox infection: transmission by infected pet rats. Acta Dermato-Venereologica, 92, 126-31.
- WILLIS, J. N., 1997. Edward Jenner and the eradication of smallpox. Scottish Medical Journal, 42, 118–121.
- ZABA, R., JALOWSKA, M., KOWALCZYK, M. J., BOWSZYC-DMOCHOWSKA, M., ADAMSKI, Z. & SZKARADKIEWICZ, A., 2017. Cowpox virus infection in a child after contact with a domestic cat: a case report. New Microbiologica, 40, 148-150.




