- Infectious Diseases of Livestock
- Part 2
- Ibaraki disease in cattle
- GENERAL INTRODUCTION: PARAMYXOVIRIDAE AND PNEUMOVIRIDAE
- Rinderpest
- Peste des petits ruminants
- Parainfluenza type 3 infection
- Bovine respiratory syncytial virus infection
- Hendra virus infection
- Paramyxovirus-induced reproductive failure and congenital defects in pigs
- Nipah virus disease
- GENERAL INTRODUCTION: CALICIVIRIDAE AND ASTROVIRIDAE
- Vesicular exanthema
- Enteric caliciviruses of pigs and cattle
- GENERAL INTRODUCTION: RETROVIRIDAE
- Enzootic bovine leukosis
- Jaagsiekte
- Visna-maedi
- Caprine arthritis-encephalitis
- Equine infectious anaemia
- GENERAL INTRODUCTION: PAPILLOMAVIRIDAE
- Papillomavirus infection of ruminants
- Papillomavirus infection of equids
- GENERAL INTRODUCTION: ORTHOMYXOVIRIDAE
- Equine influenza
- Swine influenza
- GENERAL INTRODUCTION: CORONAVIRIDAE
- Porcine transmissible gastroenteritis
- Porcine respiratory coronavirus infection
- Porcine epidemic diarrhoea
- Porcine haemagglutinating encephalomyelitis virus infection
- Porcine deltacoronavirus infection
- Bovine coronavirus infection
- Ovine coronavirus infection
- Equine coronavirus infection
- GENERAL INTRODUCTION: PARVOVIRIDAE
- Porcine parvovirus infection
- Bovine parvovirus infection
- GENERAL INTRODUCTION: ADENOVIRIDAE
- Adenovirus infections
- GENERAL INTRODUCTION: HERPESVIRIDAE
- Equid herpesvirus 1 and equid herpesvirus 4 infections
- Equid gammaherpesvirus 2 and equid gammaherpesvirus 5 infections
- Equine coital exanthema
- Infectious bovine rhinotracheitis/infectious pustular vulvovaginitis and infectious pustular balanoposthitis
- Bovine alphaherpesvirus 2 infections
- Malignant catarrhal fever
- Pseudorabies
- Suid herpesvirus 2 infection
- GENERAL INTRODUCTION: ARTERIVIRIDAE
- Equine viral arteritis
- Porcine reproductive and respiratory syndrome
- GENERAL INTRODUCTION: FLAVIVIRIDAE
- Bovine viral diarrhoea and mucosal disease
- Border disease
- Hog cholera
- Wesselsbron disease
- Louping ill
- West nile virus infection
- GENERAL INTRODUCTION: TOGAVIRIDAE
- Equine encephalitides caused by alphaviruses in the Western Hemisphere
- Old World alphavirus infections in animals
- Getah virus infection
- GENERAL INTRODUCTION: BUNYAVIRIDAE
- Diseases caused by Akabane and related Simbu-group viruses
- Rift Valley fever
- Nairobi sheep disease
- Crimean-Congo haemorrhagic fever
- GENERAL INTRODUCTION: ASFARVIRIDAE
- African swine fever
- GENERAL INTRODUCTION: RHABDOVIRIDAE
- Rabies
- Bovine ephemeral fever
- Vesicular stomatitis and other vesiculovirus infections
- GENERAL INTRODUCTION: REOVIRIDAE
- Bluetongue
- Ibaraki disease in cattle
- Epizootic haemorrhagic disease
- African horse sickness
- Equine encephalosis
- Palyam serogroup orbivirus infections
- Rotavirus infections
- GENERAL INTRODUCTION: POXVIRIDAE
- Lumpy skin disease
- Sheeppox and goatpox
- Orf
- Ulcerative dermatosis
- Bovine papular stomatitis
- Pseudocowpox
- Swinepox
- Cowpox
- Horsepox
- Camelpox
- Buffalopox
- GENERAL INTRODUCTION: PICORNAVIRIDAE
- Teschen, Talfan and reproductive diseases caused by porcine enteroviruses
- Encephalomyocarditis virus infection
- Swine vesicular disease
- Equine picornavirus infection
- Bovine rhinovirus infection
- Foot-and-mouth disease
- GENERAL INTRODUCTION: BORNAVIRIDAE
- Borna disease
- GENERAL INTRODUCTION: CIRCOVIRIDAE AND ANELLOVIRIDAE
- Post-weaning multi-systemic wasting syndrome in swine
- GENERAL INTRODUCTION: PRION DISEASES
- Scrapie
- Bovine spongiform encephalopathy
- Transmissible spongiform encephalopathies related to bovine spongiform encephalopathy in other domestic and captive wild species
Ibaraki disease in cattle
This content is distributed under the following licence: Attribution-NonCommercial CC BY-NC  View Creative Commons Licence details here
View Creative Commons Licence details here
NJ Maclachlan and M-L Penrith (Editors). H Shirafuji, Ibaraki disease in cattle, 2018.
Ibaraki disease in cattle
Previous authors: Y KITANO
Current authors:
H SHIRAFUJI - Senior Researcher, Kyushu Research Station, National Institute of Animal Health (NIAH), 2702 Chuzan, Kagoshima, 891-0105, Japan
Introduction
Ibaraki disease is an acute Culicoides-borne orbiviral disease of cattle characterized by stomatitis and, in severe cases, difficulty in swallowing due to degenerative and necrotic changes in the musculature of the oesophagus, pharynx and/or tongue. The virus is not pathogenic for sheep.16, 20, 30, 37
It was first recognized in Japan in the late summer and autumn of 1959 and 1960, when it caused severe economic losses to cattle farmers. The causative agent was named after the prefecture of Ibaraki, in which the virus was first isolated from affected cattle.16 At that time, however, because the physicochemical and biological properties of Ibaraki virus were very similar to those of bluetongue virus (BTV) it was referred to as ‘bluetongue-like virus’ and ‘Kaeishi virus’, the latter because the first investigators were from the Japanese National Institute of Animal Health or ‘Kaeishi’ in Japanese. The disease was also known as laryngopharyngeal paralysis because of the characteristic clinical signs.16, 36
Aetiology
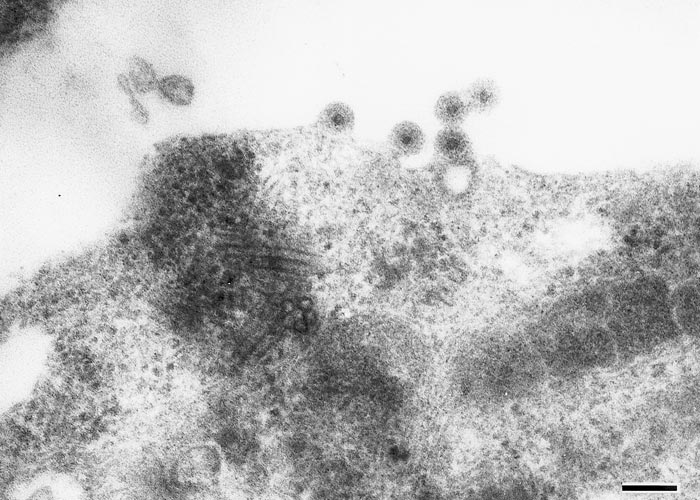
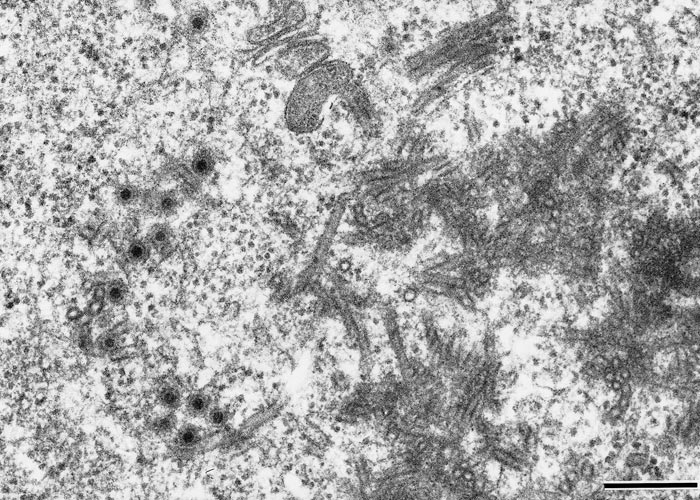
Ibaraki virus (Figures 1 and 2) is a member of the epizootic haemorrhagic disease of deer (EHD) serogroup (see Epizootic haemorrhagic disease ), genus Orbivirus of the family Reoviridae.10 It is a typical orbivirus (See Bluetongue Figure 2). It was initially classified as serotype 7 of the epizootic haemorrhagic disease virus (EHDV),11 but it is now classified as serotype 2 based on neutralization tests with isolates from North America, Japan, Nigeria and Australia.7, 8, 13, 34 The primary determinant of EHDV serotypes is VP2 encoded by genome segment 2 (Seg-2),28, 40 and the nucleotide and amino acid identities of complete Seg-2/VP2 sequences are 95.5 per cent and 96.8 per cent between Ibaraki virus and an EHDV serotype 2 strain of Australia (strain CSIRO 438).1 However, the identities are only 71.1 per cent and 73.6 per cent between Ibaraki virus and the prototype strain of EHDV serotype 2 (strain Alberta) that was isolated in Alberta, Canada.1
Ibaraki virus causes cytopathic effects in primary cell cultures of bovine, sheep and hamster kidney origin.38 It also replicates in cell cultures of BHK21-W11 of baby hamster kidney origin,18 HmLu-1 of suckling hamster lung origin, and mouse fibroblastic L cells, but not in primary cultures of horse and porcine kidney cells nor in HeLa cell cultures. When Ibaraki virus is injected into the yolk sac of chicken embryos, viral growth is as rapid in four-day-old embryos incubated at 33,5 °C as it is in those incubated at 37 °C. The virus also multiplies in the brain of suckling mice infected intracerebrally.39
The physicochemical properties of Ibaraki virus closely resemble those of BTV,30 but the neutralization, haemagglutination, complement fixation, and ferritin-tagging tests reveal that the two viruses possess different antigens.5, 6, 16, 36, 37 The agar gel precipitin and indirect fluorescent antibody tests show a two-way cross relationship between Ibaraki virus and EHDV serotypes 1 (New Jersey strain) and 2 (Alberta strain).5, 47
Ibaraki virus grown in HmLu-1 cell cultures haemagglutinates a wide variety of erythrocytes including bovine, goat, sheep, horse and rabbit, a property shared with other orbiviruses, such as African horse sickness (AHS) virus and BTV.48-50
Epidemiology
Ibaraki disease was first recognized in the southern and central parts of Japan from August to December 1959, when it affected 30 076 cattle and caused 4 023 deaths. In 1960 a similar outbreak occurred from the end of September to December, involving parts of the Chubu and Kinki districts in the central part of Japan. In this outbreak a total of 4 717 cases were reported.16, 36, 37
Since the first outbreaks of Ibaraki disease in Japan no cases were recorded until 1982 and 1987 when further outbreaks occurred. In both of these outbreaks stillbirths and abortions were also reported.23
In the summer and autumn of 1997, many clinically affected cattle were observed on Kyushu Island, to the west of Japan. During the same period many pregnant cows aborted or had stillbirths. The serum of affected cows and some of the foetuses contained antibody against Ibaraki virus, and virus was isolated from some of the foetuses although the isolates differed genetically from strains isolated previously.51 Later, the isolate was further analysed and identified as EHDV serotype 7.42
In October to December 2015, 46 cattle in Hyogo Prefecture, Japan showed clinical signs similar to those of Ibaraki disease including fever, anorexia, cessation of rumination, swollen eyelids, salivation, paralysis of the tongue, and difficulty in swallowing. The pathological findings were also similar to those observed in Ibaraki disease, but the causative agent was EHDV serotype 6.21
Serological surveys conducted from 1972 to 1975 to determine the possible involvement of arbovirus infections in the occurrence of congenital abnormalities in cattle in Japan, revealed that 16,4 per cent of the samples c antibody against Ibaraki virus.32
Outside Japan, 73 cases of Ibaraki disease occurred in Korea in 1982.2 The disease has not been reported from any other country despite the fact that cattle with neutralizing antibodies against Ibaraki virus have been found in Taiwan, Australia,16, 36 Trinidad, Tobago, Guyana,14 and the Bali islands of Indonesia.31 Both in Australia and the tropical parts of the Americas,15 while disease has not been reported, different serotypes of EHDV, including Ibaraki virus, have been isolated from wild-caught Culicoides spp.10, 11, 46 and sentinel43 or affected cattle, and antibodies against the viruses detected.3, 9, 35
One of the epidemiological features of Ibaraki disease is its seasonal prevalence particularly during the late summer and autumn months and hence the possibility that it is transmitted byan arthropod vector.37 The peaks of the 1959 and 1960 outbreaks were reached in October or November, but further south the prevalence of the disease peaked in September. An additional reason supporting the possibility that the disease may be arthropod-borne is that the other orbiviral diseases such as bluetongue (BT) and AHS are both transmitted by Culicoides spp.9, 17, 25, 41, 44, 45
Clinical signs
The clinical signs of Ibaraki disease are similar to those of EHD and closely resemble those of BT (see Bluetongue).19, 33
Results of serological surveys indicate that most infections are inapparent.29 In experimentally induced cases the incubation period varies from 4 to 11 days, but it is usually four to seven days. The first sign of disease is a rise in temperature that may reach 41,4 °C. At the onset of the disease there is anorexia, depression, lachrymation, congestion of the conjunctival and nasal mucous membranes, profuse serous nasal discharges (Figure 3) and ruminal atony and stasis. The eyelids and tongue of severely affected cattle are markedly oedematous. The disease is usually mild and affected cattle recover completely in two to three days.4, 17, 24, 36, 37 In more severe cases, the skin of the muzzle and lips, and the nasal and oral mucous membranes become severely congested and cyanotic, and may become necrotic. The affected epidermis of the muzzle sloughs, while lesions in the mouth which may involve the tongue, gingiva and hard palate, develop into erosions or ulcers. Erosions or ulcers may also occur in the skin of the coronets and be responsible for severe lameness.17, 19, 36, 37 The joints of the legs may be swollen and painful.
Dysphagia is a prominent clinical feature in 20 to 30 per cent of affected cattle. It usually occurs four to 11 days after the onset of disease, when pyrexia and other initial signs have subsided. In some animals, however, it may be observed in the absence of prodromal signs. Difficulty in deglutition occurs as a result of severe haemorrhagic, degenerative and necrotic lesions in the musculature of the oesophagus, larynx, pharynx and/or tongue. When the head of an animal suffering from severe lesions in the oesophageal and pharyngeal musculature is held high the animal is able to swallow ingested food and water, but as soon as it is lowered, the contents of the oesophagus, sometimes accompanied by ruminal contents, flow from the mouth and nose. The difficulty in swallowing results in dehydration, emaciation and, occasionally, acute aspiration pneumonia, the latter constituting the major cause of death. Thirty to 40 per cent of cattle which develop difficulty in deglutition die or are slaughtered for humane reasons as there is little hope for their recovery.17, 36, 37
Pathology
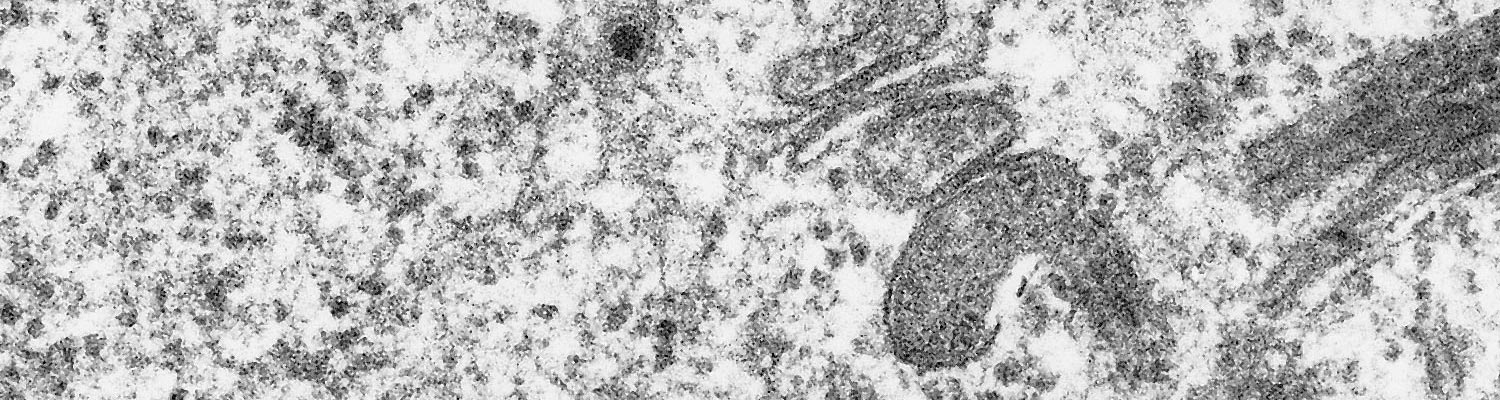
In addition to the lesions observed clinically, erosions or ulcers of the mucous membranes may be present in the oesophagus, rumen, reticulum, omasum and abomasum. Those in the abomasum are sometimes associated with marked congestion, haemorrhage and oedema.17, 19, 36, 37 In severely affected cases dilatation and mucosal haemorrhages of the oesophagus and focal degenerative and necrotic changes of its musculature may be present (Figure 4). Similar lesions may be present in the muscles of the tongue and pharynx but are absent in other striated muscles. Petechial haemorrhages may be present in the epi-, myo- and endocardium, liver, spleen, bladder, uterus, intestines and lymph nodes. Evidence of a focal interstitial nephritis may be present.
Histologically, in severe cases, lesions in the musculature of the oesophagus, larynx, pharynx and tongue are characterized by hyaline degeneration and necrosis and mineralization accompanied by an infiltration of neutrophils, lymphocytes and histiocytes (Figure 5).12, 17, 19, 36, 37 Perimysial cells are swollen and hyperplastic. A marked proliferation of fibroblasts occurs in more chronic lesions. These lesions are frequently accompanied by haemorrhages, in which case some associated blood vessels show oedema of their walls and hyaline thrombi in their lumens. In a few cases, degenerative and necrotic changes or fibroplasia may also be present in the myocardium.22
Diagnosis and differential diagnosis
A tentative diagnosis of Ibaraki disease can be made on epidemiological, clinical and pathological evidence, but it must be confirmed by isolation and identification of the virus17, 36, 37 from the blood or lymph nodes. Viral isolation can be improved if the erythrocytes in blood samples are first rinsed in phosphate buffered saline, and tissue specimens are frozen and then thawed. Virus can be isolated in tissue culture or by intracerebral inoculation of suckling mice. The tissue culture medium should be changed every seven to ten days; cytopathic effects may be observed after about 20 days of primary culture, or seven days of the second or third passage. After intracerebral inoculation baby mice often manifest paralysis of the legs. Virus in tissue culture cells is identified using an immunofluorescent test, and in the brains of suckling mice39 by the complement fixation test. Virus can also be identified by RT-PCR. Conventional and real-time RT-PCR assays have been developed for group-specific and serotype-specific detection of EHDV.26, 27
For the serological diagnosis of Ibaraki disease, it is essential to examine paired serum samples, a rise in antibody titre indicating a positive diagnosis. For this purpose either the neutralization or haemagglutination test,49, 50 is used. Neutralizing antibodies appear in the blood of affected cattle about one week after infection.
Ibaraki disease should be differentiated from diseases such as BT, EHD, foot-and-mouth disease, vesicular stomatitis, bovine herpesvirus type 1 infection, bovine virus diarrhoea/mucosal disease, bovine ephemeral fever, and bovine malignant catarrhal fever.
Control
Of primary importance in the treatment of Ibaraki disease is to take measures against the dehydration and aspiration pneumonia, which occur in severe cases due to the difficulties in deglutition.
A killed and a live virus vaccine for the control of Ibaraki disease are available commercially and both provide an effective immunity. They should be administered before summer when outbreaks usually occur.
References
- ANTHONY, S. J., MAAN, S., MAAN, N., KGOSANA, L., BACHANEK-BANKOWSKA, K., BATTEN, C., DARPEL, K.E., SUTTON, G., ATTOUI, H. & MERTENS, P.P.C, 2009. Genetic and phylogenetic analysis of the outer-coat proteins VP2 and VP5 of epizootic haemorrhagic disease virus (EHDV): Comparison of genetic and serological data to characterise the EHDV serogroup. Virus Research, 145, 200–210.
- BAK, U. B., CHEONG, C.K., CHOI, H.I., LEES, C.W., OH, H.S., RHEE, Y.O., CHO, M.R. & LIM, Y.I, 1983. An outbreak of Ibaraki disease in Korea. Korean Journal of Veterinary Research, 23, 81-89.
- BARNARD, B. J. H., GERDES, G. H. & MEISWINKEL, R., 1998. Some epidemiological and economic aspects of a bluetongue-like disease in cattle in South Africa — 1995/96 and 1997. Onderstepoort Journal of Veterinary Research, 65, 145-151.
- CAMPBELL, C. H., BARBER, T. L. & JOCHIM, M. M., 1978. Antigenic relationship of Ibaraki, bluetongue and epizootic haemorrhagic disease viruses. Veterinary Microbiology, 3, 14-21.
- CAMPBELL, C. H., BARBER, T.L. & JOCHIM, M.M., , 1978. Antigenic relationship of Ibaraki, bluetongue and epizootic haemorrhagic disease viruses. Veterinary Microbiology, 3, 14-21.
- CAMPBELL, C. H., BREESE, S. S., JR. & MCKERCHER, P. D., 1975. Antigenic and morphological comparisons of Ibaraki and bluetongue viruses. Canadian Journal of Microbiology, 21, 1098–2102.
- CAMPBELL, C. H. & ST. GEORGE, T. D., 1986. A preliminary report of a comparison of epizootic haemorrhagic disease viruses from Australia with others from North America, Japan and Nigeria. Australian Veterinary Journal, 63, 233.
- DANIELS, P. W., SENDOW, I., SOLEHA, E., SUKARSIH, HUNT, N.T. & BAHRI, S, 1995. Australian–Indonesian collaboration in veterinary arbovirology — a review. Veterinary Microbiology, 46, 141–174.
- FOSTER, N. M., METCALF, F. E., BARBER, T. L., JONES, R. H. & LEUDKE, A. J., 1980. Bluetongue and epizootic haemorrhagic disease virus isolations from vertebrate and invertebrate hosts at common geographic sites. Journal of the American Veterinary Medical Association, 176, 116-118.
- GORMAN, B. M., 1979. Variation in orbiviruses. Journal of General Virology, 44, 1-14.
- GORMAN, B. M., RAYLOR, J. & J., W. P., 1983. Orbiviruses. In: JOKLIK, W. K., (ed.). The Reoviridae, New York: Plenum Publishing Corporation.
- GOTO, M., 1959. Pathological studies on the so-called bovine influenza. II. On the changes of striated muscles and central nervous system. Japan Journal of Veterinary Science, 21, 149–160.
- GOULD, A. R. P., L.I., , 1991. Phylogenetic analyses of the complete necleotide sequence of the capsid protein (VP3) of Australian epizootic haemorrhagic disease of deer virus (serotype 2) and cognate genes from other orbiviruses. Virus Research, 21, 1-18.
- GUMM, I. D., TAYLOR, W.P., ROACH, C.J., ALEXANDER, F.C.M., GREINER, E.C. & GIBBS, E.P.J, 1984. Serological survey of ruminants in some Caribbean and South American countries of type-specific antibody to bluetongue and epizootic haemorrhagic disease of deer viruses. Veterinary Record, 114, 635–638.
- HOMAN, E. J., TAYLOR, W. P., LORBACHER DE RUIZ, H. & YUILL, T. M., 1985. Bluetongue virus and epizootic haemorrhagic disease of deer virus serotypes in northern Colombian cattle. Journal of Hygiene, Cambridge University Press, 95, 165–172.
- INABA, Y., 1975. Ibaraki disease and its relationship to bluetongue. Australian Veterinary Journal, 51, 178–185.
- INABA, Y., 1975. Ibaraki disease and its relationship to bluetongue. Australian veterinary journal, 51, 178–185.
- INABA, Y., TANAKA, Y., SATO, K., ITO, H., OMORI, T. & MATUMOTO, M. , 1968. Bovine epizootic fever. I. Propagation of the virus in suckling hamster, mouse and rat, and hamster kidney BHK21-W11 cells. Japan Journal of Microbiology, 11, 457–469.
- ISHITANI, R., 1967. Lesions and pathological differential diagnosis of bluetongue-like disease in cattle. Journal of the Japan Veterinary Medical Association, 20,
- ITO, Y., TANAKA, Y., INABA, T. & OMORI, T., 1973. Electron microscopy of Ibaraki virus. Archiv für die Gesamte Virusforschung, 40, 28–46.
- KAMOMAE, Y., KAMOMAE, M., OHTA, Y., NABE, M., KAGAWA, Y., OGURA, Y., KATO, T., TANAKA, S., YANASE, T. & SHIRAFUJI, H, 2018. Epizootic hemorrhagic disease virus serotype 6 infection in cattle, Japan, 2015. Emerging Infectious Diseases, 24, 902-905.
- KITANO, Y., YAMASHITA, S. & FUKUYAMA, T., 1988. Pathological observations on cattle that died of Ibaraki disease. Journal of the Japan Veterinary Medical Association, 41, 884–888
- KITANO, Y., YAMASHITA, S. & FUKUYAMA, T, 1988. Pathological observations on cattle that died of Ibaraki disease. Journal of the Japan Veterinary Medical Association, 41, 884–888.
- KIUCHI, M. & INABA, Y., 1952. Study on so-called ‘Bovine influenza’. Bulletin of the National Institute of Animal Health, 25, 37-42.
- LUEDKE, A. J., JONES, R. H. & JOCHIM, M. M., 1967. Transmission of bluetongue between sheep and cattle by Culicoides variipennis. American Journal of Veterinary Research, 27, 457–460
- MAAN, N. S., MAAN, S., NOMIKOU, K., JOHNSON, D. J., EL HARRAK, M., MADANI, H., YADIN, H., INCOGLU, S., YESILBAG, K., ALLISON, A. B., STALLKNECHT, D. E., BATTEN, C., ANTHONY, S. J. & MERTENS, P. P. C., 2010. RT-PCR assays for seven serotypes of epizootic haemorrhagic disease virus & their use to type strains from the Mediterranean region and North America. PloS One, 5, e12782.
- MAAN, N. S., MAAN, S., POTGIETER, A.C., WRIGHT, I. M., BELAGANAHALLI, M. & MERTENS, P.P.C., , 2016. Development of real-time RT-PCR assays for detection and typing of epizootic haemorrhagic disease virus. Transboundary and Emerging Diseases, 64, 1120–1132.
- MACLACHLAN N.J., Z., S., SAVINI, G. & DANIELS, P.W, 2015. Revue Scientifique et Technique (International Office of Epizootics). 34, 341–351.
- MATUMOTO, M., INABA, Y., TANAKA, Y., MORIMOTO, T. & OMORI, T., 1970. Ibaraki virus, an agent of epizootic disease of cattle resembling bluetongue. III. Experimental inoculation of cell culture, embryonated eggs and laboratory animals. Japanese Journal of Microbiology, 14, 99–109.
- MATUMOTO, M., INABA, Y., TANAKA, Y., MORIMOTO, T. & OMORI, T, 1970. Ibaraki virus, an agent of epizootic disease of cattle resembling bluetongue. III. Experimental inoculation of cell culture, embryonated eggs and laboratory animals. Japanese Journal of Microbiology, 14, 99–109.
- MIURA, Y., INABA, Y., TSUDA, T., TOKUHISA, S., SATO, K., AKASHI, H. & MATUMOTO, M., 1982. A survey of antibodies to arthropod-borne viruses in Indonesian cattle. Japan Journal of Veterinary Science, 44, 857-863.
- MIURA, Y., INABA, Y., HAYASHI, S., TAKAHASHI, E. & MATUMOTO, M.,, 1980. A survey of antibodies to arthropod-borne viruses in Japanese cattle. Veterinary Microbiology, 5, 277-282.
- MOULTON, J. E., 1961. Pathology of bluetongue of sheep in California. Journal of the American Veterinary Medical Association, 138, 493–498.
- NANA PEREIRA, E. M., IWATA, H. & INOUE, T, 2000. The complete nucleotide sequence of segment L2 of Ibaraki virus encoding for the antigen recognized by neutralizing antibodies. Journal of Veterinary Medical Science, 62, 317-321.
- ODIAWA, G., BLUE, J. L., TYLER, D. E. & SHOTTS, E. B., 1985. Bluetongue and epizootic haemorrhagic disease in ruminants in Georgia: Survey by serotest and virologic isolation. American Journal of Veterinary Research, 46, 2193–2196.
- OMORI, T., 1970. Ibaraki disease: A bovine epizootic disease resembling bluetongue. National Institute of Animal Health Quarterly, 10, 45-55.
- OMORI, T., INABA, Y., MORIMOTO, T., TANAKA, Y., ISHITANI, R., KUROGI, H., MUNAKATA, K., MATSUDA, K. & MATUMOTO, M., 1969. Ibaraki disease, an agent of epizootic disease of cattle resembling bluetongue. I. Epidemiologic, clinical and pathologic observations and experimental transmission to calves. Japan Journal of Microbiology, 13, 139–147.
- OMORI, T., INABA, Y., MORIMOTO, T., TANAKA, Y., KONO, M., KUROGI, H. & MATUMOTO, M., 1969. Ibaraki disease, an agent of epizootic disease of cattle resembling bluetongue. II. Isolation of the virus in bovine cell culture. Japan Journal of Microbiology, 13, 149–168.
- SASAKI, N., KODAMA, K., IWAMOTO, I., IZUMIDA, A. & MATSUBARA, T., 1968. Serial transmission in suckling mice of a virus from cattle with bovine epizootic fever. Japan Journal of Microbiology, 11, 151–152.
- SAVINI, G., AFONSO, A., MELLOR, P., ARADAIB, I., YADIN, H., SANAA, M., WILSON, W., MONACO, F. & DOMINGO, M., 2011. Epizootic heamorragic disease. Research in Veterinary Science, 91, 1-17.
- SELLERS, R. F., 1980. Weather, host and vector — their interplay in the spread of insect-borne animal virus disease. Journal of Hygiene, Cambridge University Press, 85, 65–102.
- SHIRAFUJI, H., KATO, T., YAMAKAWA, M., TANAKA, T., MINEMORI, Y. & YANASE, T., 2017. Characterization of genome segments 2, 3 and 6 of epizootic hemorrhagic disease virus strains isolated in Japan in 1985–2013: Identification of their serotypes and geographical genetic types. Infection, Genetics and Evolution, 53, 38-46.
- ST GEORGE, T. D., 1985. Studies on the pathogenesis of bovine ephemeral fever in sentinel cattle. I. Virology and serology. Veterinary Microbiology, 10, 493–504.
- ST GEORGE, T. D., CYBINSKI, I. H. & STANDFAST, H. A., 1983. The isolation of five different viruses of the epizootic haemorrhagic disease of deer. Australian veterinary journal, 60, 216-217.
- ST GEORGE, T. D. & MULLER, M. J., 1984. The isolation of a bluetongue virus from Culicoides brevitarsis. Australian Veterinary Journal, 61, 95.
- ST. GEORGE, T. D., CYBINSKI, I. H. & STANDFAST, H. A., 1983. The isolation of five different viruses of the epizootic haemorrhagic disease of deer. Australian Veterinary Journal, 60, 216-217.
- SUGIYAMA, M., HIRAYAMA, N., SASAKI, H., SUGIYAMA, T., MINAMOTO, T. & KINJO, T., 1989. Antigenic relationship among strains of Ibaraki virus and epizootic haemorrhagic disease virus studied with monoclonal antibodies. Research in Veterinary Science, 46, 273–275.
- TOKUHISA, S., INABA, Y., MIURA, Y., SATO, K. & MATUMOTO, M., 1983. Salt-dependent haemagglutination with Ibaraki virus and its inhibition by specific antisera. Japan Journal of Veterinary Science, 45, 14–21
- TOKUHISA, S., INABA, Y., SATO, K., MIURA, Y., KANEKO, N. & MATUMOTO, M., 1983. Hemagglutination-inhibiting antibody sensitive to 2-mercaptoethanol in cattle infected with Ibaraki virus. Japan Journal of Veterinary Science, 45, 687–689.
- TOKUHISA, S., INABA, Y., SATO, K., MIURA, Y., KANEKO, N. & MATUMOTO, M., 1983. Physicochemical properties of Ibaraki virus haemagglutinin. Japan Journal of Veterinary Science, 45, 637–646.
- TSUDA, T., 1998. Outbreak of Ibaraki disease and related stillbirth-abortion of cattle in 1997. Journal of Veterinary Clinic, 45, 251–255.