- Infectious Diseases of Livestock
- Part 2
- Infectious bovine rhinotracheitis/infectious pustular vulvovaginitis and infectious pustular balanoposthitis
- GENERAL INTRODUCTION: PARAMYXOVIRIDAE AND PNEUMOVIRIDAE
- Rinderpest
- Peste des petits ruminants
- Parainfluenza type 3 infection
- Bovine respiratory syncytial virus infection
- Hendra virus infection
- Paramyxovirus-induced reproductive failure and congenital defects in pigs
- Nipah virus disease
- GENERAL INTRODUCTION: CALICIVIRIDAE AND ASTROVIRIDAE
- Vesicular exanthema
- Enteric caliciviruses of pigs and cattle
- GENERAL INTRODUCTION: RETROVIRIDAE
- Enzootic bovine leukosis
- Jaagsiekte
- Visna-maedi
- Caprine arthritis-encephalitis
- Equine infectious anaemia
- GENERAL INTRODUCTION: PAPILLOMAVIRIDAE
- Papillomavirus infection of ruminants
- Papillomavirus infection of equids
- GENERAL INTRODUCTION: ORTHOMYXOVIRIDAE
- Equine influenza
- Swine influenza
- GENERAL INTRODUCTION: CORONAVIRIDAE
- Porcine transmissible gastroenteritis
- Porcine respiratory coronavirus infection
- Porcine epidemic diarrhoea
- Porcine haemagglutinating encephalomyelitis virus infection
- Porcine deltacoronavirus infection
- Bovine coronavirus infection
- Ovine coronavirus infection
- Equine coronavirus infection
- GENERAL INTRODUCTION: PARVOVIRIDAE
- Porcine parvovirus infection
- Bovine parvovirus infection
- GENERAL INTRODUCTION: ADENOVIRIDAE
- Adenovirus infections
- GENERAL INTRODUCTION: HERPESVIRIDAE
- Equid herpesvirus 1 and equid herpesvirus 4 infections
- Equid gammaherpesvirus 2 and equid gammaherpesvirus 5 infections
- Equine coital exanthema
- Infectious bovine rhinotracheitis/infectious pustular vulvovaginitis and infectious pustular balanoposthitis
- Bovine alphaherpesvirus 2 infections
- Malignant catarrhal fever
- Pseudorabies
- Suid herpesvirus 2 infection
- GENERAL INTRODUCTION: ARTERIVIRIDAE
- Equine viral arteritis
- Porcine reproductive and respiratory syndrome
- GENERAL INTRODUCTION: FLAVIVIRIDAE
- Bovine viral diarrhoea and mucosal disease
- Border disease
- Hog cholera
- Wesselsbron disease
- Louping ill
- West nile virus infection
- GENERAL INTRODUCTION: TOGAVIRIDAE
- Equine encephalitides caused by alphaviruses in the Western Hemisphere
- Old World alphavirus infections in animals
- Getah virus infection
- GENERAL INTRODUCTION: BUNYAVIRIDAE
- Diseases caused by Akabane and related Simbu-group viruses
- Rift Valley fever
- Nairobi sheep disease
- Crimean-Congo haemorrhagic fever
- GENERAL INTRODUCTION: ASFARVIRIDAE
- African swine fever
- GENERAL INTRODUCTION: RHABDOVIRIDAE
- Rabies
- Bovine ephemeral fever
- Vesicular stomatitis and other vesiculovirus infections
- GENERAL INTRODUCTION: REOVIRIDAE
- Bluetongue
- Ibaraki disease in cattle
- Epizootic haemorrhagic disease
- African horse sickness
- Equine encephalosis
- Palyam serogroup orbivirus infections
- Rotavirus infections
- GENERAL INTRODUCTION: POXVIRIDAE
- Lumpy skin disease
- Sheeppox and goatpox
- Orf
- Ulcerative dermatosis
- Bovine papular stomatitis
- Pseudocowpox
- Swinepox
- Cowpox
- Horsepox
- Camelpox
- Buffalopox
- GENERAL INTRODUCTION: PICORNAVIRIDAE
- Teschen, Talfan and reproductive diseases caused by porcine enteroviruses
- Encephalomyocarditis virus infection
- Swine vesicular disease
- Equine picornavirus infection
- Bovine rhinovirus infection
- Foot-and-mouth disease
- GENERAL INTRODUCTION: BORNAVIRIDAE
- Borna disease
- GENERAL INTRODUCTION: CIRCOVIRIDAE AND ANELLOVIRIDAE
- Post-weaning multi-systemic wasting syndrome in swine
- GENERAL INTRODUCTION: PRION DISEASES
- Scrapie
- Bovine spongiform encephalopathy
- Transmissible spongiform encephalopathies related to bovine spongiform encephalopathy in other domestic and captive wild species
Infectious bovine rhinotracheitis/infectious pustular vulvovaginitis and infectious pustular balanoposthitis
This content is distributed under the following licence: Attribution-NonCommercial CC BY-NC  View Creative Commons Licence details here
View Creative Commons Licence details here
NJ Maclachlan and M-L Penrith (Editors). S Van Drunen Littel-Van Den Hurk, S K Tikoo and L A Babiuk, Infectious bovine rhinotracheitis/infectious pustular vulvovaginitis and infectious pustular balanoposthitis, 2019.

Infectious bovine rhinotracheitis/infectious pustular vulvovaginitis and infectious pustular balanoposthitis
Previous authors: L A BABIUK, S VAN DRUNEN LITTEL-VAN DEN HURK AND S K TIKOO
Current authors:
S VAN DRUNEN LITTEL-VAN DEN HURK - Professor and Graduate Chair, PhD,Microbiology & Immunology, College of Medicine, Program Chair, VIDO-Intervac, University of Saskatchewan, 120 Veterinary Road, Saskatoon, Saskatchewan, S7N 5E3, Canada
S K TIKOO - Professor and Research Fellow, BVSC&AH, MVSc (Veterinary Microbiology), PhD (Molecular Virology), VIDO, University of Saskatchewan, 120 Veterinary Road, Saskatoon, Saskatchewan, S7N 5E3, Canada
L A BABIUK - Professor, PhD, DSc, OC, FRSC, University of Alberta, 1030 Hume Ave Kelowna, British Columbia, V1P 1P2, Canada
Introduction
Although rhinotracheitis, pustular vulvovaginitis and pustular balanoposthitis are the most common manifestations of disease caused by bovine herpesvirus 1 (BoHV-1), the sites of infection of the virus are not strictly restricted to the respiratory or genital tracts; other syndromes caused by it include those associated with conjunctivitis, abortion or meningoencephalitis.38 It must, however, be borne in mind that BoHV-5 is more frequently associated with fatal meningoencephalitis in cattle than BoHV-1.
In 1913 a vulvovaginitis of cattle called Bläschenausschlag was first described in Europe.167 It was subsequently shown to be caused by a virus that was isolated from the genital tract of an affected cow.71, 161 In parallel, an acute contagious febrile infection of cattle characterized by an intense inflammatory reaction of the upper respiratory tract, which was accompanied by dyspnoea, nasal discharge and depression, was reported and called infectious bovine rhinotracheitis (IBR) to describe its infectious nature and hallmark which is rhinotracheitis.84 Although these two distinct syndromes have been referred to in the past as ‘red nose’, ‘dust pneumonia’, infectious necrotic rhinotracheitis, coital exanthema, vesicular venereal disease, or infectious pustular vulvovaginitis (IPV), it has been accepted that they all refer to infection with either bovine herpesvirus 1 (BoHV-1) subtype 1 or 2.38, 65, 66, 92, 137
Aetiology
Bovine herpesvirus 1 is a member of the order Herpesvirales, family Herpesviridae, subfamily Alphaherpesvirinae, genus Varicellovirus.122 Isolation and characterization of the infectious agent have clearly demonstrated that BoHV-1 can cause the clinical syndromes mentioned above. Although it is common that an animal suffers simultaneously from more than one syndrome, particularly respiratory disease and conjunctivitis, it is rare for an animal to suffer concurrently from both respiratory and genital infections.
Bovine herpesvirus 1 contains a double stranded genome of 135 kilobase pairs (kbp).40, 120 The genomic arrangement of BoHV-1 is typical of group D herpesviruses in that the genome is divided into a unique long (UL) segment of approximately 103 kbp and a unique short (US) segment of 32 kbp. While the UL region remains in its prototype orientation, the US region flanked by terminal repeat (TR) and inverted repeat (IR) regions can exist in two orientations relative to the UL region.93
A total of 73 open reading frames (ORFs) have identified in the BoHV-1 genome,131, 160 which encode 11 glycoproteins.120, 131 Of the 73 ORFs, only 33 appear to be essential for virus replication.120 The requirement of 2 genes (double copy) for virus replication is not clear.120 Similar to other alphaherpesviruses, the genome is transcribed in three different stages, viz. immediate early (α), early (β), and late (γ) genes.123 The late gene transcripts occur at the time of viral DNA synthesis. Recent evidence suggests that in addition to BICP0, BCIP4 and BCIP22, BoHV-1 encodes UL21, UL33 and UL34, which are expressed as immediate early genes.114 Interestingly, BoHV-1 also encodes four unique ORFs, UL0.5, UL3.5, Circ and US1.5.131 Proteomic analysis identified 33 proteins in purified BoHV-1 virions (including 9 envelope proteins, 5 capsid proteins and 19 tegument proteins) and 15 host proteins.9
In addition, the BoHV-1 genome encodes 10 microRNAs (miRNA), which are processed into 12 mature miRNAs, and may play a role in BHV-1 latency and post-transcriptional regulation of BoHV-1 transcripts.41, 69
There are three subtypes of BoHV-1: BoHV-1.1, BoHV1.2a, and BoHV1.2b101, 120 which have been differentiated by genomic DNA restriction endonuclease fragment polymorphisms.96, 97 Subtype 1.1 is more associated with respiratory infections; subtype 1.2a mostly causes respiratory and genital signs; and subtype 1.2b is predominantly associated with genital infections in Wentink et al., 1993.156 Although all the neurological strains were initially also grouped with BoHV-1, the one most frequently associated with fatal meningoencephalitis in cattle has been reclassified as BoHV-5 based on its unique genomic characteristics.124, 137 All isolates of BoHV-1 appear to be antigenically related and even after extensive in vitro or in vivo passage, the isolates retain their antigenicity.39 Studies have clearly indicated the genetic stability of the virus which has significant implications for vaccine development and diagnosis. However, there is some variation in restriction endonuclease DNA fragment patterns that may be useful for epidemiological studies in some situations.35, 96
Epidemiology
Bovine herpesvirus-1 infections occur worldwide. Although cattle are the principal host of BoHV-1, other ruminants such as goats, several members of the deer family (Cervidae), water buffalo (Bubalus bubalis), Cape or African buffalo (Syncerus caffer), and wildebeest (Connochaetes sp.) are susceptible and may be potential reservoirs for the virus.22, 47, 70, 100, 134, 164 Although BoHV-1 can infect cell lines of other animal species, the replication is either abortive or only occurs to a low level.
Outbreaks of disease are often preceded by introduction of an infected animal into herds. Spread to susceptible animals in adjacent pens or farms generally occurs quickly due to the highly contagious nature of the virus. An outbreak usually reaches its peak within two to three weeks from its commencement and is over in four to six weeks.
Bovine herpesvirus-1 can cause mild to severe clinical disease in cattle of all ages and all breeds, but the disease is most common in animals over six months of age. This is probably due to a decline in passive immunity acquired through the colostrum and increased chances of exposure to the virus in older animals.
Following a primary infection and recovery, the virus, like other herpesviruses, establishes a latent infection.30, 104, 112 Latency can be established regardless of whether the animal suffers from a clinical or a subclinical infection in ganglia and reactivation may occur following either a variety of natural stressors or artificially by immunomodulation with compounds such as dexamethasone.111 Since most animals do not show clinical signs following reactivation, it is impossible to predict when an outbreak might occur or which animal was responsible for the outbreak. Once the virus is reactivated from the latent state, it can be spread by aerosols or secretions to adjacent animals.
Two different management situations influence the route of transmission. In intensive livestock environments, the close contact between animals plays a pivotal role in the rapid spread of infection. The other mode of transmission is via the genital route.158
Although the virus is an enveloped virus, thereby being susceptible to disinfectants which destroy the lipid envelope, it is generally accepted that it is relatively stable and can persist in the environment for several days under appropriate temperature and humidity conditions. Thus, it may be transmitted between animals by nasal secretions or saliva in feed bunks. In vitro, BoHV-1 can remain viable for a number of days at room temperature in cell culture medium. Due to its stability, the virus can also be transmitted mechanically by, for example, nose tongs. Flies may transmit the disease mechanically.26 The virus has been isolated from the tick Ornithodoros coriaceus.138 However, transmission by arthropods has not been accepted as an important method of spread.
For more information on the endogenous and exogenous factors that play a role in the epidemiology, the section on the pathogenesis and clinical signs should be consulted.
Pathogenesis and clinical signs
Regardless of the species or primary site of infection, BoHV-1 typically causes local lesions in mucous membranes characterized by the production of vesicles which progress to pustules and eventually erosions or ulcers that heal in 12 to 14 days. Since the clinical consequences of transmission via the respiratory tract or the genital tract are slightly different, they are described separately.
Respiratory infections
Natural transmission occurs by direct contact with droplets containing the virus. The levels of virus in nasal secretions can exceed 107 plaque-forming units/ml of nasal secretion at the peak of virus shedding.5 Viral shedding generally peaks at three to six days post-infection and is cleared by day 12 to 14 after infection.83
Since as little as 103 to 104 infectious virus particles are sufficient to cause infection, it is not surprising that the virus spreads rapidly between animals in close proximity.128
Although many factors can influence the severity of respiratory disease, especially the immune status of the animal, it is believed that the dose of virus that animals are exposed to, as well as the route of exposure, are critical. Generally the higher the dose animals are exposed to, the more severe the infection. Many animals infected with BoHV-1 are also infected simultaneously with other viruses, such as bovine respiratory syncytial virus (BRSV), bovine parainfluenza virus 3 (BPIV3) and bovine viral diarrhoea virus (BVDV) and in concert, these agents play a key role in initiating and exacerbating secondary bacterial infections.6 The respiratory complex caused by BoHV-1, BPIV3, BRSV and BVDV in combination with bacterial infections is referred to as “shipping fever” as it often occurs after moving calves to the feedlots in North America (see pneumonic pasteurellosis in cattle). Thus, the viral and bacterial microflora will ultimately influence the eventual outcome of the disease and whether antibiotic therapy will be helpful or not (see below).
In uncomplicated BoHV-1 infections, the virus enters the respiratory tract and multiplies in the epithelial cells of the upper respiratory tract and then spreads to the lower respiratory tract. As the virus replicates it causes destruction of the epithelial cells and alteration of the mucociliary clearance mechanisms. In addition to causing respiratory infection, the virus can also cause conjunctivitis. Indeed, in some animals the only real sign of infection is conjunctivitis. Whether conjunctivitis is due to initial infection of the respiratory tract, and subsequent spread to the conjunctiva or vice versa is not known.133 Although viraemia is difficult to detect, it must occur since the virus can spread to other parts of the body including the foetus, leading to abortion.38, 65, 107, 136, 159 This spread is probably as a result of infected monocytes, which then carry the virus to all parts of the body.36 Although the virus can spread systemically, systemic disease is rare except in foetuses and in neonatal animals.10, 38
In the case of neonatal calves, the level of maternal-derived immunity will markedly influence the clinical signs and extent of systemic spread. In animals possessing no such immunity, the virus can spread to all tissues, including the liver and gastrointestinal tract, and cause death.95 However, once animals are a few weeks old, they rarely die solely from BoHV-1 infection. Initially, there were reports of a neurovirulent form of BoHV-158 which could cause neurological infection in calves and even kill adult animals. Subsequently it has been shown that this condition is caused by BoHV-5.
In uncomplicated cases of IBR infections, regardless of the age of the animal, characteristic signs include fever (41 to 42 °C), increased respiratory rate, varying degrees of inappetence, occasional coughing, and serous to mucopurulent nasal discharge. In dairy cattle, an additional sign is a precipitous drop in milk production. Upon close observation of the nasal passages, hyperaemia and small foci of necrosis can be observed. Excessive salivation occurs in some cattle due to mouth breathing and a reluctance to swallow. The course of disease is variable among individual animals and secondary bacterial infection will both increase the severity of disease and duration of clinical signs. If râles are heard on auscultation of the thorax, complications due to secondary infection must be considered.
The severity and nature of lesions observed in the respiratory tract vary depending upon the time of examination and extent of secondary infection. The lesions include necrotizing rhinitis or rhinotracheitis, pharyngitis, and laryngotracheobronchitis.38, 164 As the disease progresses, there is epithelial necrosis with cellular debris and exudate on the mucosal surface or within the lumens of airways and congestion, oedema, neutrophil infiltration and mononuclear cell accumulation in the lamina propria and submucosa. The nasal plaques seen grossly are a result of coalesced pustules and necrotic epithelial cells that slough off to leave erosions and ulcers.
Following infection with BoHV-1, there is a high probability of colonization of the respiratory tract with bacteria, especially Mannheimia haemolytica, an organism that is often present in the nasopharynx of cattle6 (see pneumonic pasteurellosis in cattle). This superinfection may significantly increase mortality rates. Virus-induced lesions in the mucosa of the upper respiratory tract result in changes to the local physiological environment as well as the animal’s defences against infection that allow a large number of bacteria to adhere to the surface of epithelial cells as well as to replicate at a faster rate.6 Furthermore, BoHV-1 can impair alveolar macrophages and polymorphonuclear neutrophils (PMNs)15, 155 These factors permit multiplication of the bacteria and their inhalation into the lower respiratory tract. Mannheimia haemolytica produces a leukotoxin that is able to kill leukocytes or to impair the destruction of bacteria by leukocytes.86, 90, 164 Thus, both BoHV-1 and M. haemolytica have an effect on the host’s ability to clear the bacteria. A more severe infection develops which often results in serofibrinous bronchopneumonia and pleuritis. The bronchopneumonia occurs primarily in the ventral aspects of the lobes of the lungs and produces loud bronchial tones and moists râles that are audible on auscultation of the thoracic cavity. The pleuritis aggrevates the thoracic pain and produces audible rubbing sounds. If chemotherapy is not initiated early in the course of the disease, immunopathological events cannot be halted and the animal will then often die from anoxia and toxaemia.
Genital infections
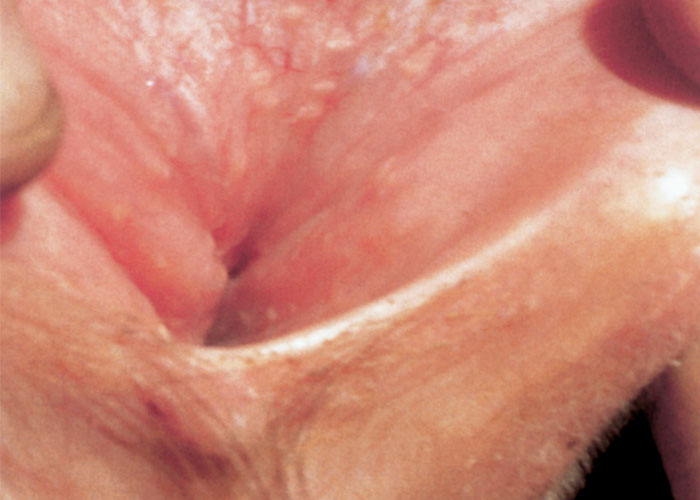
Genital infection with BoHV-1 subtype 2 occurs in both sexes. In cows and heifers acute IPV usually occurs about one to three days after mating with an infected bull. Some may, however, become infected and shed virus without showing genital lesions.26, 54, 68, 89 Genital infection may result in the development of vesicles, pustules and erosions or ulcers in the mucosa of the vulva and vagina (Figure 1). Following healing of the acute lesions depigmented areas may be present in the mucosa. The first clinical signs in females are frequent urination, and elevation of the tail and a mild vaginal discharge. Examination of the vulva usually reveals oedema with small pustules 1 to 2 mm in diameter on the mucosal surface of the vagina. In uncomplicated cases the animals generally recover in 10 to 14 days. If bacterial infection occurs, metritis may develop accompanied by a transient infertility and purulent vaginal discharge for several weeks.
The disease in bulls is known as infectious pustular balanoposthitis (IPB). As in cows, the incubation period is one to three days after which pustules appear on the mucosal surface of the penis and prepuce. The presence of the disease is seldom obvious externally although the prepuce may be swollen and a discharge may be present. In severe cases, the bull may refuse to mate due to the associated pain during breeding. Consequent secondary bacterial infections may occur and aggravate the disease.
In such cases scar tissue may develop, especially if the bull is allowed to continue mating, resulting in adhesions, anular constrictions, and distortion or bending of the penis. If severe, these might permanently impair the bull’s ability to breed.53
The use of artificial insemination does not totally eliminate the possibility of spreading the disease, since semen collected from subclinically affected bulls may contain the virus. It is for this reason that many artificial insemination units do not admit seropositive animals as semen donors.20, 72 This problem may be alleviated by the use of marker vaccines, also called DIVA (differentiating infected from vaccinated animals) vaccines, with accompanying diagnostic tests, which would allow differentiation of vaccinated from infected animals.
Abortions
Abortion is a common sequel following infection in a herd and may occur regardless of the severity of the primary syndrome, i.e. respiratory or genital.159 Some animals do not exhibit any evidence of impending abortion up to the time of the event. Thus, a farmer may not even be aware that his herd is infected with BoHV-1 until an abortion storm occurs.
Since abortions can occur as long as 90 days after infection, it might be difficult to relate the abortions to BoHV-1 infection, especially if the infection was mild or subclinical.38, 65, 136 Although the bovine foetus is susceptible at all stages of development, most abortions occur after the fifth month of pregnancy. Abortion can also be caused by intramuscular inoculation of pregnant cows with inadequately modified live virus vaccines.99
Foetuses are invariably dead when expelled and large amounts of blood-tinged pleural and peritoneal fluids are frequently present. In some cases, there is little relaxation of the vagina and vulva, thus, manual removal of the foetus might be necessary. The afterbirth might be temporarily retained, but infertility usually does not result. For accurate diagnosis, the entire foetus and any membranes shed by the cow should be submitted to a diagnostic laboratory as soon as possible.
Enteric infections
Bovine herpesvirus 1 occasionally causes enteritis in calves.121 Although it is able to replicate in the gastrointestinal mucosa of adults, no clinical consequences have been reported.
Latency
In animals that survive primary infection, the virus enters the peripheral nervous system via cell-to-cell spread and invariably establishes a latent state in the trigeminal (TG) or sacral (SG) ganglia following respiratory or genital infection, respectively.1, 51, 103, 164 Viral gene expression and infectious virus can be detected from 1-6 days in the TG,55, 126 while later only viral genomes can be detected.61 However, the latency-related (LR) gene is abundantly transcribed;59 the LR gene products promote establishment of latency by inhibiting apoptosis25 and viral gene expression.37 As in other herpesvirus infections, reactivation can and does occur due to stress (elevated corticosteroid levels) and/or immune suppression. The shipping of calves from the ranches to the feedlot can trigger reactivation from latency contributing to the bovine respiratory disease complex. During reactivation from latency LR gene expression decreases, while overall viral gene expression increases in the sensory neurons, and infectious virus is secreted from the ocular and nasal cavities.62
Episodes of reactivation can play a role in maintaining the virus in a herd. Reactivation of virus by treatment of animals with dexamethasone leads to viral shedding at levels equivalent to those seen following primary infection,111 but the duration of shedding is generally thought to be shorter during the former. The possible reason for this is that the secondary immune response rapidly clears the virus. However, the data supporting this contention should be interpreted cautiously since the virus is complexed with antibody, which neutralizes the virus, giving the impression that there is no virus present in nasal secretions. This is supported by the observation that even after multiple reactivations, when nasal antibodies are high, the reactivated virus can be detected in nasal secretions by electron microscopy or PCR, but no infectious virus is detected by conventional virus isolation techniques.110 These results suggest that after multiple reactivations, an animal would be less able to spread the virus to its cohorts due to the presence of mucosal antibodies. Whether a similar reduction in transmission of reactivated virus occurs during genital transmission is not known.
Immunity
Innate responses
As is the case for all infections, the innate response is first activated, followed by the adaptive response. The magnitude and quality of the innate response is a determining factor for the robustness of the adaptive response. The first response to infection is mediated by recognition of pathogen associated molecular patterns (PAMPs) by pathogen recognition receptors (PRRs). The PRRs include toll-like receptors (TLRs), and RIG-I-like receptors (RLRs). Ten bovine TLRs have been identified with unique and overlapping specificities;2 they have 66-88 per cent nucleotide/amino acid similarity to the corresponding human/murine TLRs.157 It has been shown that several human herpesvirus-1 (HHV-1) PAMPs are recognized, for instance glycoproteins by TLR2 with TL1/6, ds DNA by TLR9 and ds and stem-loop RNA by TLR3.21, 109 Since BoHV-1 has a very similar composition and replication cycle, the same TLRs may recognize PAMPs in response to BoHV-1 infection. Once activated after recognition of viral PAMPs, the PRRs initiate a signalling cascade ultimately converging on the activation and nuclear translocation of transcription factors, NF-κB and IFN-regulatory factor (IRF)-3. Double-stranded RNA produced during infection can also be recognized by RIG-I and MDA-5 in the cytoplasm,109 leading to activation and nuclear translocation of NF-κB and IRF-3 and-7. The ultimate result is the production of type I IFNs (IFN-α/β) and several pro-inflammatory cytokines, including TNF-α, IL-12 and IL-6.
The type I IFNs play a critical role early after infection, by inducing an antiviral state in cells adjacent to the infected cells.2 They can be detected as early as 5 h post-infection and peak at 36-72 h in the nasal secretions and blood.8 It must be pointed out that BoHV-1 is relatively resistant to the direct antiviral effects of interferon. Type I IFNs and pro-inflammatory cytokines also recruit and activate macrophages, polymorphonuclear neutrophils and large granular lymphocytes (natural killer (NK) cells in cattle).
Polymorphonuclear neutrophils migrate to the site of infection within 24 to 48 hours post-infection. Early pro-inflammatory cytokines are generated by epithelial cells and alveolar macrophages leading to the expression of adhesion molecules which retain leukocytes at the site of infection.11, 80 Increased vascular permeability and adhesion brings cells to the site of infection. Bovine neutrophils are important in the defense against BoHV-1. Neutrophils have PRRs and receptors for complement and they are a major source of α and β defensins and cathelocidins.2 Polymorphonuclear neutrophils were found to be most effective in antibody-dependent cell cytotoxicity (ADDC).42 Bovine herpesvirus 1 infected cells can also be lysed directly by C-mediated neutrophil cytotoxicity (CDNC).6 Macrophages also have PRRs and play an important role in the responses to BoHV-1 infection, being able to produce IFN-α. Later during infection macrophages can kill BoHV-1-infected cells once activated by IFN-γ produced by T cells.18
Early during viral infection, NK cells are activated by type I IFNs. In cattle NK cells with homologs of human receptors have been identified.34 There are two sub-populations of NK cells (CD2+ and CD2-),14 both of which produce IFN-γ and are able to kill virus-infected cells unrestricted by MHC compatibility.19, 57, 108 Dendritic cells (DCs) bridge the innate and adaptive responses, and in fact are the only antigen-presenting cells (APCs) responsible for activation of resting T cells and the primary immune responses to infection. Both plasmacytoid and myeloid DCs express several PRRs. Plasmacytoid dendritic cells (pDCs) including bovine pDCs,118 produce by far the most IFN-α of all immune cells. However, pDCs have not been studied in context of BoHV-1 infection.
Adaptive responses
The adaptive immune response to infection, including BoHV-1, is initiated by the DCs, which take up and process antigen, followed by migration to secondary lymphoid organs and stimulation of T lymphocytes. Follicular dendritic cells (FDCs) play a major role in the presentation of antigen to B cells. Once activated, the DCs produce cytokines which influence the polarization of the immune response to development of T helper (Th)1 cells, which drive cell-mediated immune responses or Th2 cells, which promote antibody responses. There is evidence that ruminants develop Th1/Th2 response to infections similar to mice and humans.85 Thus, cytokines are pivotal in differentiation, amplification, and fine-tuning of the immune response to BoHV-1.17
Infection with BoHV-1 generally induces a balanced immune response with both antibody and cell-mediated immunity being present. This is critical since both are important in ameliorating the disease. Antibodies are important in preventing infection and assisting in recovery from secondary infections, and cellular immunity is primarily involved in recovery once an infection is established. The relative importance of each effector mechanism will clearly depend on whether the animal is exposed to the virus for the first time, or it is a secondary exposure following vaccination or infection with a field virus.
The cell-mediated immune response is very important, specifically early during infection, as BoHV-1 can spread from cell to cell. The CD8 T cell response depends on recognition of BoHV-1 glycoproteins on the cell surface, which in vitro was observed as early as 3-4 hour after infection and peaked 7-10 days.8 Bovine CD8 T cells express perforin50 and are capable of MHC I restricted killing in vitro.44, 132 Glycoproteins C and D were found to be targets for CD8 T cells.31 In addition, γδ T cells, which are found in higher numbers in cattle than humans or mice, can contribute to control of BoHV-1.113
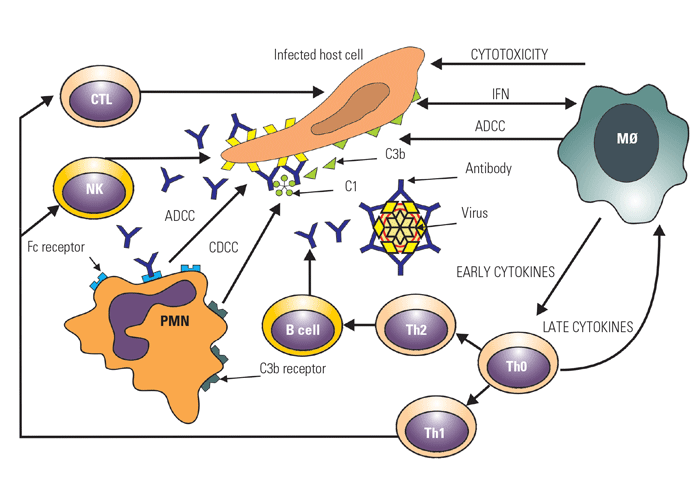
Since it has been shown that the viral glycoproteins are critical in attachment and penetration of BoHV-1 into cells, antibodies directed at these glycoproteins will generally prevent infection. Although 11 glycoproteins have been reported to be encoded by BoHV-1, the major focus has been on the role of gC and gB in virus attachment and gD in penetration of the virus into cells.8 Antibodies to any of these three glycoproteins can prevent infection and thus these glycoproteins are the basis for subunit vaccine development or DNA immunization.5, 7 Once the virus enters the cell, it can spread intracellularly without ever exiting the cell where it could be neutralized by antibody. Intracellular virus spread begins approximately 7 to 9 hours post-infection. Recent studies have shown the role of various glycoproteins in intracellular spread.88, 98, 116, 127 Thus, if antibody could target or modulate these glycoproteins on the cell surface, the spread of virus might be reduced. In addition to preventing the attachment or penetration of virus into host cells, antibodies can interact with complement to lyse virus-infected cells or to lyse virus-infected cells with the aid of Fc receptor-bearing cells (monocytes or polymorphonuclear cells) (Figure 2).
| CTL | = | cytotoxic T lymphocyte |
| NK | = | natural killer cell |
| PMN | = | polymorphonucleur granulocyte |
| B cell | = | B lymphocyte |
| Th 1 | = | T-helper 1 lymphocyte |
| Th 2 | = | T-helper 2 lymphocyte |
| Th 0 | = | T-helper lymphocyte |
| MØ | = | macrophage |
| IFN | = | interferon |
| ADCC | = | antibody dependent cell cytotoxicity |
| CDCC | = | complement dependent cell cytoxicity |
Figure 2 Immunological events in recovery from BoHV-1 infection. Upon initial infection with the virus, there is induction of new antigens on the infected cell surface and production of interferon, which can directly reduce virus replication and activate leukocytes. These activated leukocytes produce early cytokines, which activate and focus the immune response to produce antibodies, late cytokines, and cytotoxic cells, which can kill virus-infected cells via various mechanisms
Immune evasion
Bovine herpesvirus 1 has developed several strategies to evade or down-regulate the immune response. Bovine herpesvirus 1 bICP0 can degrade IRF3125 and thus inhibits IFN transcription48 or action. bICP0 also inhibits the ability of IRF-7 to activate IFN-β promoter activity.60, 61 The major tegument protein of BoHV-1, VP8, prevents nuclear translocation of STAT1, thereby preventing type I IFN activity,3 and interferes with the DNA damage repair pathway leading to apoptosis.4 VP8 also re-distributes PML in the nucleus, thereby modulating the anti-viral response.165 Glycoprotein C, expressed on the surface of BoHV-1 infected cells, can function as receptor for the cleavage product C3b, which may mediate cross-linking between C3b and gC on BoHV-1 infected cells and the PMN receptor.6 Alternatively, the C3b receptor may prevent complement action on the infected cells.101 Furthermore, PMNs from BoHV-1 infected calves have reduced chemotactic and phagocytic capacity.141 Glycoprotein G interacts with chemokines thereby interfering with their action.16 Bovine herpesvirus 1 also down regulates MHC class I. UL49.5 blocks transporter associated with antigen processing (TAP) and UL41 (vhs) reduces antigen presentation by MHC I74-76 and MHC II.49 Direct spread from cell to cell is mediated by gB, gD, gH/gL, and gE/gI allows BoHV-1 to avoid inactivation by neutralizing antibodies.144
Diagnosis
Several approaches to the diagnosis of BoHV-1 can be used. These include direct identification of the virus, detection of genomic DNA by PCR, nucleic acid hybridization and sequencing, and detection of BoHV-1-specific serum antibodies by ELISA and virus neutralization.
The direct approach can be used because at the peak of virus replication, large quantities of virus are present in nasal, ocular, or genital secretions. Thus, swabbing the suspected lesion area will generally yield high concentrations of the virus which is easy to culture in vitro in cells of bovine origin. In cases of abortion, foetal lung, liver, spleen, kidneys, and placental tissues are all good sources of virus. Although the virus is relatively stable, care should be taken to transport the samples properly (4 °C) and process them as quickly as possible. If the samples must be frozen before assaying, they should be stored at −70 °C or lower, not in a regular freezer since this will inactivate the virus. In some instances, especially for semen samples, care must be taken to avoid non-specific cellular toxicity.
Virus detection
Bovine herpesvirus 1 can be easily detected in primary cell cultures of bovine kidney, lung, testes, turbinate or trachea or established cell lines such as Madin Darby bovine kidney (MDBK) cells based on its characteristic cytopathic effect (CPE).102 At 2-3 days after inoculation grape-like clusters of rounded cells appear, followed by sloughing of the cell monolayer.135 Once the virus has caused cytopathology in vitro, the agent can be further identified as BoHV-1 by immunofluorescence or immunoperoxidase using a mono-specific BoHV-1 antiserum or monoclonal antibodies.33, 139 Specific monoclonal antibodies are available to further differentiate BoHV-1 subtypes 1 and 2.96 Restriction endonucleases can also be used to differentiate between BoHV-1 isolates and subtypes. Although restriction endonuclease tests are of limited value for diagnosis, they are useful for epidemiological studies.
Direct detection of viral antigens in swabs of nasal, ocular and genital scrapings, and in tissue samples is performed by immunofluorescence or immunoperoxidase staining. It is best to perform these assays on fresh or frozen tissues, but it is also possible to detect BoHV-1 antigens in paraffin sections of tissues.45 In all these instances, it is important to select the antibody used for antigen detection to ensure that it recognizes conserved epitopes present on all BoHV-1 isolates. Furthermore, it is also important to select antibodies directed against epitopes that are not destroyed by fixation. The advantage of direct antigen identification over virus isolation is that the diagnosis can be made in a single day and there is no need for expensive cell culture facilities.
In most laboratories polymerase chain reaction (PCR) is now the method of choice to diagnose BoHV-1, being rapid, reproducible, specific and much more sensitive than virus isolation. With real-time PCR small amounts of virus can be detected in nasal swabs, bovine foetal serum, and semen samples.154 This technique is extremely sensitive and can even detect BoHV-1 sequences in latently infected ganglia.150, 151, 163 However, it is critical to standardize and optimize the PCR assay to preclude false-positives.102 A multiplex real-time PCR assay has been developed to detect BoHV-1, BRSV and BPIV3 in specimens from bovine respiratory disease cases.140
Serology
Detection of BoHV-1-specific antibodies in serum is important for IBR diagnosis and control. Bovine herpesvirus 1 specific antibodies are identified by a variety of serological tests. These include various modifications of ELISAs,27, 77 neutralization assays, and an indirect immunofluorescent assay. ELISAs are most commonly used. In conjunction with the use of marker (DIVA) vaccines, ELISAs are used to demonstrate absence of infection to support control programs and surveillance. In an indirect (i)-ELISA plates are coated with purified virus, followed by incubation with serum and then enzyme-labelled anti-bovine IgG. For detection a substrate is added, which then develops colour. A competition (c)-ELISA is based on blocking binding of monoclonal antibodies to gB or gE by serum samples, and used in conjunction with DIVA vaccines. Plates are coated with gB or gE, and then incubated with mixtures of serum samples and enzyme-labeled gB- or gE-specific monoclonal antibody. A blocking percentage is calculated for each serum sample.105
These assays are generally useful for epidemiological studies. To diagnose a specific outbreak as being caused by BoHV-1 requires an acute serum sample collected when using doubling dilutions as early as possible during the outbreak and a convalescent sample two to three weeks after the outbreak. An increase in specific antibody titres of four-fold or higher is indicative of a BoHV-1 infection. These serological assays are also extremely useful in diagnosing herds for the presence of latently infected animals and are routinely used by artificial insemination units for eradication of the disease from specific herds, and for international trade purposes. Any animal that is serologically positive should be considered a latent carrier of the virus unless they have been vaccinated with a marker vaccine and proven to be seronegative for the specific marker. Animals that have been vaccinated with a DIVA vaccine lacking gB or gE can be differentiated from infected animals by using an accompanying anti-gB or -gE blocking c-ELISA.
Control
One approach to control this disease is to prevent contact between infected animals and seronegative animals. Since the virus is latent in seropositive animals, the major strategy is to identify seropositive animals and eliminate them from the herd. This is the practice in artificial insemination units. In countries such as Switzerland where the number of seropositive animals was originally low, a test and slaughter programme was embarked upon resulting in that country being considered free of BoHV-1. To maintain the national herd BoHV-1 free, it is essential to monitor animal movements and test each animal arriving in the country for the presence of antibodies to BoHV-1. In countries where there is a high rate of infection in the national herd, which is usually the case, the best approach to controlling disease is immunization. The use of marker vaccines with an accompanying c-ELISA allows differentation of infected animals from vaccinated animals, which is a strategy applied to the control and eventual eradication of BoHV-1. Bovine herpesvirus 1 has been eradicated in several European regions, including Bavaria of Germany, Bolzano of Italy, Austria, Denmark, Finland, Switzerland, Sweden and Norway.115
Vaccines
Vaccination remains the most effective control method of reducing economic losses due to IBR and other clinical syndromes caused by BoHV-1 infections. However, due to the fact that BoHV-1 establishes latency vaccination reduces clinical disease and transmission, but does not prevent infection or eradicate the virus. This can only be achieved with strict management practices in conjunction with vaccination. According to a database of livestock veterinary vaccines153 there are 190 BoHV-1 vaccines. While by far the majority of these vaccines are still produced by conventional methods, among this list are 5 marker vaccines (four gE-deletion mutants and one double gE-TK- mutant), which are extensively used in Europe, in particular in those countries that are planning to initiate or already have BoHV-1 control/eradication programs.
Conventional vaccines
Conventional vaccines contain either modified live virus (MLV) or killed virus (KV).145 Modified live vaccines are developed by passage of BoHV-1 in various cell cultures and selection of mutants that retain their ability to replicate in vivo, but do not cause respiratory disease (subclinical infection),129, 130, 148, 166 while KV vaccines are generated by chemical treatments with for instance formaldehyde, β-propiolactone or binary ethyleneimine.
Modified live virus (MLV) vaccines
Live viral vaccines, especially those given intranasally, have a number of advantages. Since virus replication in the cells of the vaccinated animals leads to presentation of viral antigens on both MHC class I and II molecules, MLV vaccines generally induce both humoral and cell-mediated immune responses. When delivered intranasally, they also elicit mucosal immunity that resembles a natural infection. The immune response induced by live viruses is also generally rapid and of longer duration than that obtained with killed vaccines. Live vaccines also have several disadvantages. It is not known which virulence genes of the virus are altered during attenuation; therefore, it is possible that the vaccine virus could revert to virulence, especially after its reactivation. Unfortunately, MLV virus can still spread systemically and may induce abortions if not attenuated sufficiently, so these vaccines generally should not be used to vaccinate pregnant animals. Furthermore, since the virus may be shed from vaccinated animals, these vaccines should not even be used in animals that will be in contact with pregnant animals.79, 94, 99 Live vaccines also establish latency in vivo,29, 106, 111 which can lead to reactivation and shedding. However, a second generation of MLV has been developed by further attenuation of the virus142, 143 These highly attenuated virus vaccines can be administered intranasally and, as a result of their degree of attenuation, do not induce abortions. This is especially the case with the temperature-sensitive (ts) mutants which cannot replicate at 39 °C.168 Since the nasal passage of a cow is below 39 °C, these ts mutants can replicate and induce immunity without inducing abortions. Since many of the BoHV-1 vaccines are given in combination with other vaccines and BoHV-1 MLV vaccines can be immunosuppressive, there is a concern about interference between multiple vaccines.46 Vaccines containing live virus(es) must be maintained in a cold chain to prevent inactivation, which is not always possible under animal husbandry conditions. In addition, MLVs can be pathogenic in young calves because of their relatively immature immune system. Finally, it is not possible to effectively and consistently immunize young calves that possess maternally derived antibodies, which may interfere with induction of immunity.
Inactivated or killed virus vaccines
The major advantage of KV vaccines is their safety. However, there are several disadvantages. The antigens may be altered due to the inactivation process.32, 52, 63, 81 Since inactivated virus in KV vaccines cannot replicate, the antigens are provided exogenously and presented only on MHC class II molecules, resulting in a Th2-biased immune response and humoral immunity. To enhance the immune response potent adjuvants are required. Some of the modern adjuvants such as the TLR ligand poly I:C, can cross-present antigen resulting in cell-mediated immune responses. Unfortunately, adjuvants may induce a reaction at the injection site.149 These vaccines are all injected intramuscularly and, due to the general compartmentalization of the immune response, they do not induce mucosal immunity — the first line of defence in preventing infection. Generally, KV vaccines induce immunity of shorter duration than do live virus vaccines. However, the effectiveness of these vaccines could be increased by increasing the antigenic mass, by altering the inactivation process to ensure that all the important epitopes are retained and/or by developing better adjuvants. It is generally recommended that with inactivated vaccines a primary and secondary inoculation should be given four weeks apart followed by booster inoculations annually.
Genetically engineered vaccines
The limitations of conventional vaccines combined with the major advances in molecular biology and immunology have led to the development of novel genetically engineered live virus vaccines, subunit vaccines and, most recently, nucleic acid vaccines. In addition to being safer, such vaccines can also be used as ‘marker’ vaccines in conjunction with accompanying c-ELISAs to help eradicate BoHV-1.
Gene-deleted vaccines: Once the entire BoHV-1 genome was sequenced and a large number of non-essential genes identified, a number of these could be deleted resulting in attenuation and a dramatic decrease in the chances of reversion to virulence. These new genetically engineered vaccines can be much safer than conventionally produced live vaccines and serologically distinguished from wild type field strains,145 which is particularly useful for eradication programs. The basis for marker vaccines is that animals develop immune responses to the antigens that are present in the vaccine but not to antigens that have been excluded from the vaccine. Gene deleted vaccines if sufficiently attenuated are safe and yet can induce robust humoral and cell-mediated immune responses.
Several mutants lacking non-essential genes have been generated.23, 64 The gG-, gE-, gI- and gE-/gI- mutants had significantly reduced virulence in cattle; however, the gC mutant was as virulent as wild type virus.64 The gC-, gE- and gG- mutants elicited significant protection, whereas the gI- and gE-/gI- mutants were less protective. The gC- and gG- mutants also caused latency, whereas this was not established for the gE-, gI- and gE-/gI- mutants. It was further established that the gE- mutant is unlikely to be excreted after potential re-activation.91 Another advantage of the gE- BoHV-1 is the fact that BoHV-1 infection leads to the generation of gE-specific antibodies, and that the antigenic variability of gE between strains is low.119 The gE- mutant was therefore selected as DIVA vaccine and used as either MLV or KV vaccine, both of which are protective.13, 73 Companion gE-based c-ELISAs were developed, which differentiate BoHV-1 infected from gE- mutant-vaccinated animals.152 While less sensitive than a gB-based c-ELISA, it is sufficient at the herd level.105 In 2007 a study with this gE- mutant was performed in three European countries, which showed reduction in the rate of seroconversion observed in vaccinated animals, suggesting efficiency of this vaccination program in containing viral spread.87 The gE- live marker vaccine has been used in eradication programs in countries with a high prevalence of BoHV-1 infection.
While the gE- mutants were derived from a low-virulence strain and are further attenuated, there still was a concern that it might be able to recombine with wild type virus. To alleviate this concern, a double deletion mutant was generated with the TK gene deleted in addition to gE; this double deletion mutant reduced the establishment of latency after wild type BoHV-1 challenge.67 Recently, immunosuppressive genes have been deleted to generate a more immunogenic virus. For example, deletion of the gN gene (UL49.5), which down-regulates MHC-I presentation, might result in stronger cell-mediated immune responses. Another approach has been to generate a three-gene-mutated BoHV-1 vaccine virus missing the UL49.5 luminal domain (residues 30-32), the cytoplasmic tail (residues 80-96) of gE and the entire US9. The gE-deleted BoHV-1 and the triple BoHV-1 mutant elicited equal protection from clinical signs after challenge with virulent BoHV-1 . However, the group vaccinated with the triple mutant cleared the challenge virus three days earlier than the group vaccinated with the gE-deleted mutant.24
Subunit vaccines: Subunit vaccines are considered to be extremely safe since they do not contain any replicating virus, but contain purified proteins that are important in stimulating immune responses to the specific proteins involved in virus entry. The disadvantage is the fact that such vaccines are expensive, specifically when the proteins need to be produced in mammalian cells. In the case of BoHV-1, a number of experimental vaccines incorporating gB, gC, and gD have been tested, and found to induce protective immunity against BoHV-1 challenge.5, 149 For a subunit vaccine it is critical to formulate them with appropriate adjuvants. A Quil A-adjuvanted gD subunit vaccine was tested in parallel with the inactivated gE- mutant in the Netherlands, and found to be as effective.12 In contrast to many commercial adjuvants, such as alum and Emulsigen, which only promote humoral immune responses, TLR agonists have the ability to promote balanced immune responses when formulated with recombinant antigens. For instance CpG oligodeoxynuclotides (CpG ODN) and polyI:C are strong Th1 promoting adjuvants as they induce secretion of Th1 cytokines such as IL-12 and IFN-γ.78 A subunit vaccine consisting of gD formulated with CpG ODN and an oil-based adjuvant, Emulsigen, elicited both VN antibodies and activated T cells, as well as excellent protection against BoHV-1 challenge,56 while formulation with Emulsigen alone or with a non-CpG ODN induced partial protection. In addition to being very safe, subunit vaccines are DIVA vaccines in combination with an accompanying c-ELISA; in the case of the gD vaccine, any other immunogenic protein that induces a strong antibody response during infection can be used, for example the more sensitive gB-specific c-ELISA.
Vectored vaccines: An E3-deleted bovine adenovirus has been used as vector for BoHV-1 gD. A replication-defective version elicited systemic immune responses, while a replication competent version elicited serum and nasal antibodies to gD, as well as partial protection.117
A bacterial plasmid has also been used as non-replicating vector for gD.28 In this case the gene encoding a protective antigen, for example gD, is cloned into the plasmid, which then expresses the gene within the transfected cells.82, 162 The attractiveness of DNA immunization lies in some of the advantages of this technology. This type of vaccine is safe, does not cause injection site reactions, and is simple to develop and inexpensive to produce. Since the antigens are produced endogenously, balanced immune responses are induced. Long duration of immunity has been reported after immunization with DNA vaccines. When delivered intradermally, the plasmid encoding a truncated version of gD elicited excellent cell-mediated immune responses, moderate humoral responses, and significant protection from BoHV-1 challenge.146
When gC was used as a DNA vaccine, calves developed gC-specific antibody and lymphoproliferative responses; after BoHV-1 challenge they had lower temperatures and cleared the virus more rapidly than the control animals.43 A major advantage of DNA immunization is the potential to vaccinate new-born animals with maternal antibodies; lambs immunized with plasmid encoding gD developed strong cell-mediated and humoral immune responses, equivalent to lambs without maternal antibodies.145 This approach would dramatically alter our ability to induce immunity at an early age and thus, close the ‘window of susceptibility’ to BoHV-1 under different management conditions. However, even though it has been shown to be possible to immunize cattle against BoHV-1 using plasmids, one of the major impediments to adopting this approach for routine use in the field was the need for improving the efficiency of delivery of plasmids into cells and expression of sufficient levels of antigen for induction of humoral immune responses. Recently improved delivery systems for DNA vaccines dramatically improved their efficiency. For example, delivery by injection combined with electroporation significantly enhanced immune responses in cattle.148 This technology is currently widely used to delivery DNA vaccines in human clinical trials, supporting its non-invasiveness.147 The DNA immunization approach also is compatible with marker diagnostic tests to differentiate vaccinated animals from latent carriers of BoHV-1.146
References
- ACKERMANN, M. & WYLER, R., 1984. The DNA of an IPV strain of bovine herpesvirus 1 in sacral ganglia during latency after intravaginal infection. Veterinary Microbiology, 9, 53–63.
- ACKERMANN, M. R., DERSCHEID, R. & ROTH, J. A., 2010. Innate immunology of bovine respiratory disease. Veterinary Clinics of North America: Food Animal Practice, 26(2), 215-228.
- AFROZ, S., BROWNLIE, R., FODJE, M. & VAN DRUNEN LITTEL-VAN DEN HURK, S., 2016. VP8, the Major Tegument Protein of Bovine Herpesvirus 1, Interacts with Cellular STAT1 and Inhibits Interferon Beta Signaling. Journal of Virology, 90(10), 4889-4904.
- AFROZ, S., GARG, R., FODJE, M. & VAN DRUNEN LITTEL-VAN DEN HURK, S., 2018. The Major Tegument Protein of Bovine Herpesvirus 1, VP8, Interacts with DNA Damage Response Proteins and Induces Apoptosis. Journal of Virology, 92(15),
- BABIUK, L. A., L’ITALIEN, J., VAN DRUNEN LITTEL-VAN DEN HURK, S., ZAMB, T., LAWMAN, M. J. P., HUGHES, G. & GIFFORD, G. A., 1987. Protection of cattle from bovine herpesvirus type I (BoHV-1) infection by immunization with individual viral glycoproteins. Virology, 159, 57-66.
- BABIUK, L. A., LAWMAN, M. J. P. & BIELEFELDT OHMANN, H., 1988. Viral bacterial syngeristic interactions in respiratory disease. Advances in Virus Research, 35, 219-249.
- BABIUK, L. A., LEWIS, P. J., VAN DRUNEN LITTEL-VAN DEN HURK, S. & BRAUN, R., 1999. DNA Immunization: Present and Future. Advances in Veterinary Science, 41, 163–179.
- BABIUK, L. A., VAN DRUNEN LITTEL-VAN DEN HURK, S. & TIKOO, S. K., 1996. Immunology of bovine herpesvirus-1 infection. Veterinary Microbiology, 53, 31-42.
- BARBER, K. A., DAUGHERTY, H. C., ANDER, S. C., JEFFERSON, V. A., SHACK, L. A., PECHAN, T., NANDURI, B. & MEYER, F., 2017. Protein Composition of the Bovine Herpesvirus 1.1 Virion. Veterinary Science, 4, 11.
- BAXTER, G. M., 1984. Neonatal meningoencephalitis associated with infectious bovine rhinotracheitis virus. Bovine Practitioner, 19, 41-44.
- BOCHNER, B. S., LANDY, S. D., PLAUT, M., DINARELLO, C. A. & SCHLEIMER, R. P., 1987. Interleukin 1 production by human lung tissue. I. Identification and characterization. Journal of Immunology, 139, 2297–2302.
- BOSCH, J. C., DE JONG, M. C., FRANKEN, P., FRANKENA, K., HAGE, J. J., KAASHOEK, M. J., MARIS-VELDHUIS, M. A., NOORDHUIZEN, J. P., VAN DER POEL, W. H., VERHOEFF, J., WEERDMEESTER, K., ZIMMER, G. M. & VAN OIRSCHOT, J. T., 1998. An inactivated gE-negative marker vaccine and an experimental gD-subunit vaccine reduce the incidence of bovine herpesvirus 1 infections in the field. Vaccine, 16, 265–271.
- BOSCH, J. C., KAASHOEK, M. J. & VAN OIRSCHOT, J. T., 1997. Inactivated bovine herpesvirus 1 marker vaccines are more efficacious in reducing virus excretion after reactivation than a live marker vaccine. Vaccine, 15(14), 1512-1517.
- BOYSEN, P., OLSEN, I., BERG, I., KULBERG, S., JOHANSEN, G. M. & STORSET, A. K., 2006. Bovine CD2-/NKp46+ cells are fully functional natural killer cells with a high activation status. BMC Immunology, 7, 10.
- BROWN, T. T. J. & ANANABA, G., 1988. Effect of respiratory infections caused by bovine herpesvirus-1 or parainfluenza-3 virus on bovine alveolar macrophage functions. American Journal of Veterinary Research, 49(9), 1447-1451.
- BRYANT, N. A., DAVIS-POYNTER, N., VAN DER PLASSCHEN, A. & ALCAMI, A., 2003. Glycoprotein G isoforms from some alphaherpesviruses function as broad-spectrum chemokine binding proteins. EMBO Journal, 22(4), 833-846.
- CAMPOS, M., GODSON, D. L., HUGHES, H. P. A. & BABIUK, L. A., 1994. Cytokine applications in infectious diseases., In: MORRISONS, B. G. A. I., (ed.). Cell-Mediated Immunity in Ruminants. Boca Raton: CRC Press, 229–240.
- CAMPOS, M., OHMANN, H. B., HUTCHINGS, D., RAPIN, N., BABIUK, L. A. & LAWMAN, M. J. P., 1989. Role of interferon-gamma in inducing cytotoxicity of peripheral blood mononuclear leukocytes to bovine herpesvirus type 1 (BHV-1)-infected cells. Cell Immunology, 120, 259–269.
- CAMPOS, M. & ROSSI, C. R., 1986. Cytotoxicity of bovine lymphocytes after treatment with lymphokines. American Journal of Veterinary Research, 47, 1524–1528.
- CHAPMAN, M. S., LUCAS, M. H., HERBERT, C. N. & GOODEY, R. G., 1979. Survival of infectious bovine rhinotracheitis virus in stored bovine semen. Veterinary Science Communications, 3, 137–139.
- CHEW, T., TAYLOR, K. E. & MOSSMAN, K. L., 2009. Innate and adaptive immune responses to herpes simplex virus. Viruses, 1(3), 979-1002.
- CHOW, T. L. & DAVIS, R. W., 1964. The susceptibility of mule deer to infectious bovine rhinotracheitis. American Journal of Veterinary Research, 25, 518-519.
- CHOWDHURY, S. I., 1996. Construction and characterization of an attenuated bovine herpesvirus type 1 (BHV-1) recombinant virus. Veterinary Biology, 52(1-2), 13-23.
- CHOWDHURY, S. I., WEI H, W., M., PANNHORST, K. & PAULSEN, D. B., 2014. A triple gene mutant of BoHV-1 administered intranasally is significantly more efficacious than a BoHV-1 glycoprotein E-deleted virus against a virulent BoHV-1 challenge. Vaccine, 32(39), 4909-4915.
- CIACCI-ZANELLA, J., STONE, M., HENDERSON, G. & JONES, C., 1999. The latency-related gene of bovine herpesvirus 1 inhibits programmed cell death. Journal of Virology, 73(12), 9734-9740.
- COLLINGS, D. F., GIBBS, E. P. J. & STAFFORD, L. D., 1972. Concurrent respiratory and genital disease associated with infectious bovine rhinotracheitis/ infectious pustular vulvovaginitis (IBR/IPV) virus in a dairy herd in the United Kingdom. The Veterinary Record, 91, 214-218.
- COLLINS, J. K., AYERS, V. K. & CARMAN, J., 1988. Evaluation of an antigen-capture ELISA for the detection of bovine herpesvirus type 1 shedding from feedlot cattle. Veterinary Microbiology, 16, 101-107.
- COX, G., ZAMB, T. & BABIUK, L. A., 1993. Bovine herpesvirus-1: Immune responses in mice and cattle injected with plasmid DNA. Journal of Virology, 67, 5664–5667.
- DARCEL, C. L. Q. & DORWARD, W. J., 1975. Recovery of infectious bovine rhinotracheitis virus following corticosteroid treatment of vaccinated animals. Canadian Veterinary Journal, 16, 87-88.
- DAVIES, D. H. & DUNCAN, J. H., 1974. The pathogenesis of recurrent infections with infectious bovine rhinotracheitis virus induced in calves by treatment with corticosteroids. Cornell Veterinarian, 64, 340–366.
- DENIS, M., SLAOUI, M., KEIL, G., BABIUK, L. A., ERNST, E., PASTORET, P. P. & THIRY, E., 1993. Identification of different target glycoproteins for bovine herpesvirus-1 specific cytotoxic T lymphocytes depending on the method of in vitro stimulation. Immunology, 78, 7-13.
- DUQUE, H., MARSHALL, R. L., ISRAEL, B. A. & LETCHWORTH, G. J., 1989. Effects of formalin inactivation on bovine herpes virus-1 glycoproteins and antibody response elicited by formalin-inactivated vaccines in rabbits. Vaccine, 7, 513–520.
- EDWARDS, S., CHASEY, D. & WHITE, H., 1983. Experimental infectious bovine rhinotracheitis: Comparison of four antigen detection methods. Research in Veterinary Science, 34, 42-45.
- ENDSLEY, J. J., ENDSLEY, M. A. & ESTES, D. M., 2006. Bovine natural killer cells acquire cytotoxic/effector activity following activation with IL-12/15 and reduce Mycobacterium bovis BCG in infected macrophages. Journal of Leukocyte Biology, 79(1), 71-79.
- ENGELS, M., STECK, F. & WYLER, R., 1981. Comparison of the genomes of infectious bovine rhinotracheitis and infectious pustular vulvovaginitis virus strains by restriction endonuclease analysis. Archives of Virology, 67, 169–174.
- FORMAN, A. J., BABIUK, L. A., MISRA, V. & BALDWIN, F., 1982. The responsibility of bovine macrophages to infectious bovine rhinotracheitis virus infection. Infection and Immunity, 35, 1048–1057.
- GEISER, V., INMAN, M., ZHANG, Y. & JONES, C., 2002. The latency-related gene of bovine herpesvirus-1 can inhibit the ability of bICP0 to activate productive infection. Journal of General Virology, 83(12), 2965-2971.
- GIBBS, E. P. J. & RWEYEMAMU, M. M., 1977. Bovine herpesviruses, part 1. Veterinary Bulletin, 47, 317–343.
- GILLESPIE, M. H., MCENTEE, K., KENDRICK, J. W. & WAGNER, W. C., 1959. Comparison of infectious pustular vaginitis virus with infectious bovine rhinotracheitis virus. Cornell Veterinarian, 49, 228–297.
- GLAZOV, E. A., HORWOOD, P. F., ASSAVALAPSAKUL, W., KONGSUWAN, K., MITCHELL, R. W., MITTER, N. & MAHONY, T. J., 2010. Characterization of microRNAs encoded by the bovine herpesvirus 1 genome. Journal of General Virology, 91, 32–41.
- GLAZOV, E. A., HORWOOD, P. F., ASSAVALAPSAKUL, W., KONGSUWAN, K., MITCHELL, R. W., MITTER, N. & MAHONY, T. J., 2010. Characterization of microRNAs encoded by the bovine herpesvirus 1 genome. Journal of General Virology, 91(Pt 1), 32-41.
- GREWAL, A. S., ROUSE, B. T. & BABIUK, L. A., 1977. Mechanisms of resistant of herpesviruses: comparison of the effectiveness of different cell types in mediating antibody-dependent cell-mediated cytotoxicity. Infection and Immunity, 15(3), 698-703.
- GUPTA, P. K., SAINI, M., GUPTA, L. K., RAO, V. D., BANDYOPADHYAY, S. K., BUTCHAIAH, G., GARG, G. K. & GARG, S. K., 2001. Induction of immune responses in cattle with a DNA vaccine encoding glycoprotein C of bovine herpesvirus-1. Veterinary Microbiology, 78(4), 293-305.
- GUZMAN, E., TAYLOR, G., CHARLESTON, B., SKINNER, M. A. & ELLIS, S. A., 2008. An MHC-restricted CD8+ T-cell response is induced in cattle by foot-and-mouth disease virus (FMDV) infection and also following vaccination with inactivated FMDV. Journal of General Virology, 89(Pt 3), 667-675.
- HAINES, D. M. & ELLIS, J. A., 1994. Special tests for the diagnosis of infectious causes of reproductive failure in ruminants. Veterinary Clinics of North America for the Food Animal Practitioner, 10, 561–585.
- HARLAND, R. J., POTTER, A. A., VAN DRUNEN LITTEL-VAN DEN HURK, S., VAN DONKERSGOED, J., PARKER, M. D., ZAMB, T. J. & JANZEN, E. D., 1992. The effect of subunit or modified live bovine herpesvirus-1 vaccines on the efficacy of a recombinant Pasteurella haemolytica vaccine for the prevention of respiratory disease in feedlot calves. Canadian Veterinary Journal, 33, 734–741.
- HEDGER, R. S. & HAMBLIN, C., 1978. Neutralizing antibodies to bovine herpesvirus-1 (infectious bovine rhinotracheitis/infectious pustular vulvovaginitis) in African wildlife with special reference to the Cape Buffalo (Syncerus caffer). Journal of Comparative Pathology, 88, 211–218.
- HENDERSON, G., ZHANG, Y. & JONES, C., 2005. The Bovine herpesvirus 1 gene encoding infected cell protein 0 (bICP0) can inhibit interferon-dependent transcription in the absence of other viral genes. Journal of General Virology, 86(10), 2697-2702.
- HINKLEY, S., AMBAGALA, A. P., JONES, C. J. & SRIKUMARAN, S., 2000. A vhs-like activity of bovine herpesvirus-1. Archives of Virology, 145(10), 2027-2046.
- HOGG, A. E., PARSONS, K., TAYLOR, G., WORTH, A., BEVERLEY, P., HOWARD, C. J. & VILLARREAL-RAMOS, B., 2011. Characterization of age-related changes in bovine CD8+ T-cells. Veterinary Immunology and Immunopathology, 140(1-2), 47-54.
- HOMAN, E. J. & EASTERDAY, B. C., 1980. Isolation of bovine herpesvirus-1 from trigeminal ganglia of clinically normal cattle. American Journal of Veterinary Research, 41, 1212–1213.
- HRISTOV, S. & KARADJOV, I., 1975. Study on the immunogenic properties of inactivated vaccines produced with the virus of the infectious bovine rhinotracheitis. Veterinarnomeditsinski Nauki, 13, 8–13.
- HUCK, R. A., MILLAR, P. G., EVANS, D. H., STABLES, J. W. & ROSS, A., 1971. Penoposthitis associated with infectious bovine rhinotracheitisinfectious pustular vulvovaginitis (I.B.R.-I.P.V.) virus in a stud of bulls. The Veterinary Record, 88, 292–297.
- HYNE, R. H. J. & JOHNSTON, K. G., 1964. An outbreak of infectious pustular vulvovaginitis in dairy cattle in New South Wales. Australian Veterinary Journal, 40, 385–386.
- INMAN, M., LOVATO, L., DOSTER, A. & JONES, C., 2002. A mutation in the latency-related gene of bovine herpesvirus 1 disrupts the latency reactivation cycle in calves. Journal of Virology, 76(13), 6771-6779.
- IOANNOU, X. P., GRIEBEL, P., HECKER, R., BABIUK, L. A. & VAN DRUNEN LITTEL-VAN DEN HURK, S., 2002. The immunogenicity and protective efficacy of bovine herpesvirus 1 glycoprotein D plus Emulsigen are increased by formulation with CpG oligodeoxynucleotides. Journal of Virology, 76(18), 9002-9010.
- JENSEN, J. & SCHULTZ, R. D., 1990. Bovine natural cell-mediated cytotoxicity (NCMC): Activation by cytokines. Veterinary Immunology and Immunopathology, 24, 113–124.
- JOHNSTON, L. A. Y., SIMMONS, G. C. & MCGAVIN, M. D., 1962. A viral meningoencephalitis of calves. Australian Veterinary Journal, 38, 207–215.
- JONES, C., 1998. Alphaherpesvirus latency: its role in disease and survival of the virus in nature. Advances in Virus Research, 51, 81-133.
- JONES, C. & CHOWDHURY, S., 2007. A review of the biology of bovine herpesvirus type 1 (BHV-1), its role as a cofactor in the bovine respiratory disease complex and development of improved vaccines. Animal Health Research Reviews, 8(2), 187-205.
- JONES, C. & CHOWDHURY, S., 2010. Bovine herpesvirus type 1 (BHV-1) is an important cofactor in the bovine respiratory disease complex. Veterinary Clinics of North America: Food Animal Practice, 26(2), 303-321.
- JONES, C., GEISER, V., HENDERSON, G., JIANG, Y., MEYER, F., PEREZ, S. & ZHANG, Y., 2006. Functional analysis of bovine herpesvirus 1 (BHV-1) genes expressed during latency. Veterinary Microbiology, 113(3-4), 199-210.
- JUHASZ, M., PALATKA, Z. & TOTH, B., 1974. Experiments on the effectiveness of inactivated infectious bovine rhinotracheitis vaccine. Developments in Biological Standardization, 26, 1-4.
- KAASHOEK, M. J., RIJSEWIJK, F. A., RUULS, R. C., KEIL, G. M., THIRY, E., PASTORET, P. P. & VAN OIRSCHOT, J. T., 1998. Virulence, immunogenicity and reactivation of bovine herpesvirus 1 mutants with a deletion in the gC, gG, gI, gE, or in both the gI and gE gene. Vaccine, 16(8), 802-809.
- KAHRS, R. F., 1977. Infectious bovine rhinotracheitis: A review and update. Journal of the American Veterinary Medical Association, 171, 1055–1064.
- KAHRS, R. F. & SMITH, R. S., 1965. Infectious bovine rhinotracheitis, infectious pustular vulvovaginitis and abortion in a New York dairy herd. Journal of American Veterinary Medical Association, 146, 217–220.
- KALTHOFF, D., KONIG, P., TRAPP, S. & BEER, M., 2010. Immunization and challenge experiments with a new modified live bovine herpesvirus type 1 marker vaccine prototype adjuvanted with a co-polymer. Vaccine, 28(36), 5871-5877.
- KAMINJOLO, J. S., JR., NYAGA, P. N., OMUSE, J. K. & MUTIGA, E. R., 1975. Infectious bovine rhinotracheitis-infectious pustular vulvovaginitis viral isolates from cattle with epididymitis and vaginitis. American Journal of Veterinary Research, 36, 123–125.
- KANOKUDOM, S., MAHONY, T. J., SMITH, D. R. & ASSAVALAPSAKUL, W., 2018. Modulation of bovine herpesvirus 1 infection by virally encoded microRNAs. Virus Research, 15(257), 1-6.
- KARSTAD, L., JESSETT, D. M., OTEMA, J. C. & DREVEMO, S., 1974. Vulvovaginitis in wildebeests caused by the virus of infectious bovine rhinotracheitis. Journal of Wildlife Diseases, 10, 392–396.
- KENDRICK, J. W., GILLESPIE, J. H. & MCENTEE, K., 1958. Infectious pustular vulvovaginitis of cattle. Cornell Veterinarian, 48, 458–495.
- KENDRICK, J. W. & STRAUB, O. C., 1967. Infectious bovine rhinotracheitis-infectious pustular vulvovaginitis infection in pregnant cows. American Journal of Veterinary Research, 28, 1269–1282.
- KERKHOFS, P., RENJIFO, X., TOUSSAINT, J. F., LETELLIER, C., VANOPDENBOSCH, E. & WELLEMANS, G., 2003. Enhancement of the immune response and virological protection of calves against bovine herpesvirus type 1 with an inactivated gE-deleted vaccine. The Veterinary Record, 152(22), 681-686.
- KOPPERS-LALIC, D., REITS, E. A., RESSING, M. E., LIPINSKA, A. D., ABELE, R., KOCH, J., MARCONDES REZENDE, M., ADMIRAAL, P., VAN LEEUWEN, D., BIENKOWSKA-SZEWCZYK, K., METTENLEITER, T. C., RIJSEWIJK, F. A., TAMPE, R., NEEFJES, J. & WIERTZ, E. J., 2005. Varicelloviruses avoid T cell recognition by UL49.5-mediated inactivation of the transporter associated with antigen processing. Proceedings of the National Academy of Sciences of the United States of America, 102(14), 5144-5149.
- KOPPERS-LALIC, D., RIJSEWIJK, F. A., VERSCHUREN, S. B., VAN GAANS-VAN DEN BRINK, J. A., NEISIG, A., RESSING, M. E., NEEFJES, J. & WIERTZ, E. J., 2001. The UL41-encoded virion host shutoff (vhs) protein and vhs-independent mechanisms are responsible for down-regulation of MHC class I molecules by bovine herpesvirus 1. Journal of General Virology, 82(9), 2071-2081.
- KOPPERS-LALIC, D., RYCHLOWSKI, M., VAN LEEUWEN, D., RIJSEWIJK, F. A., RESSING, M. E., NEEFJES, J. J., BIENKOWSKA-SZEWCZYK, K. & WIERTZ, E. J., 2003. Bovine herpesvirus 1 interferes with TAP-dependent peptide transport and intracellular trafficking of MHC class I molecules in human cells. Archives of Virology, 148(10), 2023-2037.
- KRAMPS, J. A., QUAK, S., WEERDMEESTER, K. & VAN OIRSCHOT, J. T., 1993. Comparative study on sixteen enzyme-linked immunosorbent assays for the detection of antibodies to bovine herpesvirus 1 in cattle. Veterinary Microbiology, 35, 11-21.
- KRIEG, A. M., 2006. Therapeutic potential of Toll-like receptor 9 activation. Nature Reviews, 5(6), 471-484.
- KUCERA, C. J., WHITE, R. G. & BECKENHAUER, W. H., 1978. Evaluation of the safety and efficacy of an intranasal vaccine containing a temperature-sensitive strain of infectious bovine rhinotracheitis virus. American Journal of Veterinary Research, 39, 607–610.
- LAMONTAGNE, L., GAULDIE, J., STADNYK, A., RICHARDS, C. & JENKINS, E., 1985. In vivo initiation of unstimulated in vitro interleukin-1 release by alveolar macrophages. American Review of Respiratory Diseases, 131, 326-330.
- LEVINGS, R. L., KAEBERLE, M. L. & REED, D. E., 1984. The effect of some common inactivation procedures on the antigens of bovine herpesvirus 1. Veterinary Microbiology, 9, 313–328.
- LEWIS, P. J., COX, G. J. M., VAN DRUNEN LITTEL-VAN DEN HURK, S. & BABIUK, L., 1997. Polynucleotide vaccines in animals: Enhancing and modulating responses. Vaccine, 15, 861–864.
- LUPTON, H. W. & REED, D. E., 1980. Clearance and shedding of infectious bovine rhinotracheitis virus from the nasal mucosa of immune and non-immune calves. American Journal of Veterinary Research, 124, 721–722.
- MADIN, S. H., YORK, C. J. & MCKERCHER, D. G., 1956. Isolation of the IBR virus. Science. Science, 124, 721–722.
- MAGOMBEDZE, G., EDA, S. & GANUSOV, V. V., 2014. Competition for antigen between Th1 and Th2 responses determines the timing of the immune response switch during Mycobaterium avium subspecies paratuberulosis infection in ruminants. PLoS Computational Biology, 10(1), e1003414.
- MAHESWARAN, S. K., BERGGREN, K. A., SIMONSON, R. R., WARD, G. E. & MUSCOPLAT, C. C., 1980. Kinetics of interaction and fate of Pasteurella hemolytica in bovine alveolar macrophages. Infection and Immunity, 30, 254–262.
- MAKOSCHEY, B., ZEHLE, H. H., BUSSACCHINI, M., VALLA, G., PALFI, V. & FOLDI, J., 2007. Efficacy of a live bovine herpesvirus type 1 marker vaccine under field conditions in three countries. The Veterinary Record, 161(9), 295-298.
- MALLORY, S., SOMMER, M. & ARVIN, A. M., 1998. Analysis of the glycoproteins I and E of varicella-zoster virus (VZV) using deletional mutations of VZV cosmids. Journal of Infectious Diseases, 178(1), S22–S26.
- MARE, C. J. & VAN RENSBURG, S. J., 1961. The isolation of viruses associated with infertility in cattle: A preliminary report. Journal of the South African Veterinary Medical Association,, 32, 201-210.
- MARKHAM, R. J. F. & WILKIE, B. N., 1980. Interaction between Pasteurella haemolytica and bovine alveolar macrophages: Cytotoxic effect on macrophages and impaired phagocytosis. American Journal of Veterinary Research, 41, 18-22.
- MARS, M. H., DE JONG, M. C. & VAN OIRSCHOT, J. T., 2000. A gE-negative BHV1 vaccine virus strain cannot perpetuate in cattle populations. Vaccine, 18(20), 2120-2124.
- MATHEWS, R. E. F., 1982. Classification and nomenclature of viruses. Intervirology, 17, 28–199.
- MAYFIELD, J. E., GOOD, P. J., VANOORT, H. J., CAMPBELL, A. R. & REED, D. E., 1983. Cloning and cleavage site mapping of DNA from bovine herpesvirus 1 (Cooper strain). Journal of Virology, 47, 259–264.
- MCKERCHER, D. G. & CRENSHAW, G. L., 1971. Comparative efficacy of intranasally and parenterally administered infectious bovine rhinotracheitis vaccines. Journal of the American Veterinary Medical Association, 159, 1362–1369.
- MECHOR, G. D., ROUSSEAUX, C. G., RADOSTITS, O. M., BABIUK, L. A. & PETRIE, L., 1987. Protection of newborn calves against fatal multisystemic infectious bovine rhinotracheitis by feeding colostrum from vaccinated cows. Canadian Journal of Veterinary Research, 51, 452–459.
- METZLER, A. E., MATILE, H., GASSMANN, U., ENGELS, M. & WYLER, R., 1985. European isolates of bovine herpesvirus 1: A comparison of restriction endonuclease sites, polypeptides, and reactivity with monoclonal antibodies. Archives of Virology, 85, 57–69.
- METZLER, A. E., SCHUDEL, A. A. & ENGELS, M., 1986. Bovine herpesvirus 1: molecular and antigenic characteristics of variant viruses isolated from calves with neurological disease. Archives of Virology, 87(3-4), 205-217.
- MEYER, G., HANON, E., GEORLETTE, D., PASTORET, P. P. & THIRY, E., 1998. Bovine herpesvirus type 1 glycoprotein H is essential for penetration and propagation in cell culture. Journal of General Virology, 79, 1983– 1987.
- MITCHELL, D. H., 1974. An outbreak of abortion in a dairy herd following inoculation with an intramuscular infectious bovine rhinotracheitis virus vaccine. Canadian Veterinary Journal, 15, 148–151.
- MOHANTY, S. B., LILLIE, M. G., CORSELIUS, N. P. & BECK, J. D., 1972. Natural infection with infectious bovine rhinotracheitis virus in goats. Journal of the American Veterinary Medical Association, 160, 879–880.
- MUYLKENS, B., THIRY, J., KIRTEN, P., SCHYNTS, F. & THIRY, E., 2007. Bovine herpesvirus 1 infection and infectious bovine rhinotracheitis. Veterinary Research, 38(2), 181-209.
- NANDI, S., KUMAR, M., MANOHAR, M. & CHAUHAN, R. S., 2009. Bovine herpes virus infections in cattle. Animal Health Research Reviews, 10(1), 85-98.
- NARITA, M., INUI, S., NAMBA, K. & SHIMIZU, Y., 1976. Trigeminal ganglionitis and encephalitis in calves intranasally inoculated with infectious bovine rhinotracheitis virus. Journal of Comparative Pathology, 86, 93–100.
- NARITA, M., INUI, S., NANBA, K. & SHIMIZU, Y., 1981. Recrudescence of infectious bovine rhinotracheitis virus and associated neural changes in calves treated with Dexamethasone. American Journal of Veterinary Research, 42, 1192–1197.
- NETTLETON, P. & RUSSELL, G. M., 2017. Update on infectious bovine rhinotrachetis. In Practice, 39, 255-272.
- NETTLETON, P. F., SHARP, J. M. & HERRING, A. J., 1984. Infectious bovine rhinotracheitis virus excretion after vaccination, challenge, and immunosuppression., In: WITTMAN, R. M. G. & RHIZA, H. J., (eds.). Latent Herpesvirus Infections in Veterinary Medicine. Boston: Martinus Nijhoff, 191–209.
- NYAGA, P. N. & MCKERCHER, D. G., 1979. Pathogenesis of bovine herpesvirus-1 (BOHV-1) infections: Interactions of the virus with peripheral bovine blood cellular components. Comparative Immunology, Microbiology and Infectious Diseases, 24, 682.
- PALMER, L. D., LEARY, T. P., WILSON, D. M. & SPLITTER, G. A., 1990. Bovine natural killer-like cell responses against cell lines expressing recombinant bovine herpesvirus type 1 glycoproteins. Journal of Immunology, 145, 1009–1014.
- PALUDAN, S. R., BOWIE, A. G., HORAN, K. A. & FITZGERALD, K. A., 2011. Recognition of herpesviruses by the innate immune system. Nature reviews. Immunology, 11(2), 143-154.
- PASTORET, P. P., AGUILAR-SETIEU, A., BURTONBOY, G., MAYER, J., JETTEUR, P. & SCHOENAERS, F., 1979. Effect of repeated treatment with Dexamethasone on the re-excretion pattern of infectious bovine rhinotracheitis virus and humoral immune response. Veterinary Microbiology, 4, 149–156.
- PASTORET, P. P., BABIUK, L. A., MISRA, V. & GRIEBEL, P., 1980. Reactivation of temperature-sensitive and non-temperature-sensitive infectious bovine rhinotracheitis vaccine virus with dexamethasone. Infection and Immunity, 29, 483–488.
- PASTORET, P. P., THIRY, E., BROCHIER, B. & DERBOVEN, G., 1982. Bovid herpesvirus 1 infection of cattle: Pathogenesis, latency, consequences of latency. Annales de Recherches Vétérinaires, 13, 221–235.
- PLATT, R., BURDETT, W. & ROTH, J. A., 2006. Induction of antigen-specific T-cell subset activation to bovine respiratory disease viruses by a modified-live virus vaccine. American Journal of Veterinary Research, 67(7), 1179-1184.
- POKHRIYAL, M., RATTA, B., SINGH YADAV, B., KUMAR, A., SAXENA, M., PRAKASH VERMA, O. & SHARMA, B., 2018. Three newly identified Immediate Early Genes of Bovine herpesvirus 1 lack the characteristic Octamer binding motif-1. Scientific Reports, 8, 11441.
- RAAPERI, K., ORRO, T. & VILTROP, A., 2014. Epidemiology and control of bovine herpesvirus 1 infection in Europe. Veterinary Journal, 201(3), 249-256.
- RAJCANI, J. & VOJVODOVA, A., 1998. The role of herpes simplex virus glycoproteins in the virus replication cycle. Acta Virologica, 42, 103–118.
- REDDY, P. S., IDAMAKANTI, N., PYNE, C., ZAKHARTCHOUK, A. N., GODSON, D. L., PAPP, Z., BACA-ESTRADA, M. E., BABIUK, L. A., MUTWIRI, G. K. & TIKOO, S. K., 2000. The immunogenicity and efficacy of replication-defective and replication-competent bovine adenovirus-3 expressing bovine herpesvirus- 1 glycoprotein gD in cattle. Veterinary Immunology and Immunopathology, 76(3-4), 257-268.
- REID, E., JULEFF, N., GUBBINS, S., PRENTICE, H., SEAGO, J. & CHARLESTON, B., 2011. Bovine plasmacytoid dendritic cells are the major source of type I interferon in response to foot-and-mouth disease virus in vitro and in vivo. Journal of Virology, 85(9), 4297-4308.
- RIJSEWIJK, F. A., KAASHOEK, M. J., LANGEVELD, J. P., MARIS-VELDHUIS, M. A., MAGDALENA, J., VERSCHUREN, S. B., MELOEN, R. H. & VAN OIRSCHOT, J. T., 2000. Epitopes on glycoprotein E and on the glycoprotein E/glycoprotein I complex of bovine herpesvirus 1 are expressed by all of 222 isolates and 11 vaccine strains. Archives of Virology, 145(5), 921-936.
- ROBINSON, K. E., MEERS, J., GRAVEL, J. L., MCCARTHY, F. M. & MAHONY, T. J., 2008. The essential and non-essential genes of bovine herpesvirus 1. Journal of General Virology, 89, 2851–2863.
- ROGERS, R. J., MCCUBBIN, I. K., MCKENZIE, R. A., CHUNG, Y. S. & ROSENFELD, L. E., 1978. Bovine herpesvirus-1: Infection of the upper alimentary tract of cattle and its association with a severe mortality. Australian Veterinary Journal, 54, 562–565.
- ROIZMAN, B., CARMICHAEL, L. E. & DEINHART, F., 1982. Herpesviridae: Definition, provisional nomenclature and taxonomy. Intervirology, 16, 201-217.
- ROIZMAN, B. & SEARS, A. C., 1996. Herpes simplex viruses and their replication., In: FIELDS, B. N., KNIPE, D., HOWLEY, P. M., CHANOCK, R. M., MELNICK, J. L., MONATH, T. P., ROIZMAN, B. & STRAUSS, S., (eds.). Fields Virology. New York: Lipponcott, Raven Press, 2, 2231–2295.
- ROIZMANN, B., DESROSIERS, R. C., FLECKENSTEIN, B., LOPEZ, C., MINSON, A. C. & STUDDERT, M. J., 1992. The family Herpesviridae: An update. The Herpesvirus Study Group of the International Committee on Taxonomy of Viruses. Archives of Virology, 123, 425–449.
- SAIRA, K., ZHOU, Y. & JONES, C., 2007. The infected cell protein 0 encoded by bovine herpesvirus 1 (bICP0) induces degradation of interferon response factor 3 and, consequently, inhibits beta interferon promoter activity. Journal of Virology, 81(7), 3077-3086.
- SCHANG, L. M. & JONES, C., 1997. Analysis of bovine herpesvirus 1 transcripts during a primary infection of trigeminal ganglia of cattle. Journal of Virology, 79(9), 6786-6795.
- SCHRODER, C. & KEIL, G. M., 1999. Bovine herpesvirus 1 requires glycoprotein H for infectivity and direct spreading and glycoproteins gH(W450) and gB for glycoprotein D-independent cell-to-cell spread. Journal of General Virology, 80, 57-61.
- SCHROYER, E. I. & EASTERDAY, B. C., 1968. Studies on the pathogenesis of infectious bovine rhinotracheitis following aerosol exposure. Cornell Veterinarian, 58, 442–461.
- SCHWARTZ, A. J. F., YORK, C. J., ZIRBEL, L. W. & ESTELA, L. A., 1957. Modification of infectious bovine rhinotracheitis (IBR) virus in tissue culture and development of a vaccine. Proceedings of the Society for Experimental Biology and Medicine, 96, 453–458.
- SCHWARTZ, A. J. F., ZIRBEL, L. W., ESTADA, L. A. & YORK, C. Y., 1958. Propagation and modification of infectious bovine rhinotracheitis (IBR) virus in porcine kidney tissue culture. Proceedings of the Society for Experimental Biology and Medicine, 97, 680–683.
- SCHWYZER, M. & ACKERMANN, M., 1996. Molecular virology of ruminant herpesviruses. Veterinary Microbiology, 53, 17–29.
- SPLITTER, G. A., ESKRA, L. & ABRUZZINI, A. F., 1988. Cloned bovine cytolytic T cells recognize bovine herpes virus-1 in a genetically restricted, antigen-specific manner. Immunology, 63, 145–150.
- ST GEORGE, T. D., 1965. Keratoconjunctivitis associated with infectious bovine rhinotracheitis infection in Victorian cattle. Australian Veterinary Journal, 41, 222–223.
- ST GEORGE, T. D. & PHILPOT, M., 1972. Isolation of IBR virus from the prepuce of water buffalo bulls in Australia. Australian Veterinary Journal, 48, 126.
- STRAUB, O. C., BENGELSDORFF, H. J. & WIZIGMANN, G., 1990. The detection of bovine herpesvirus type 1 (BHV 1) using an intradermal test. II. Experimental studies. Zentralbl Veterinarmed [B], 37(1), 35-46.
- STUBBINGS, D. P. & CAMERON, I. R. D., 1981. Bovine abortion associated with infectious bovine rhinotracheitis virus infection. The Veterinary Record, 108, 101-102.
- STUDDERT, M. J., 1994. Bovine herpesviruses, In: WEBSTER, R. G. & GRANOFF, A., (eds.). Encyclopedia of Virology. New York, NY: Academic Press, 1, 155–158.
- TAYLOR, R. E., SEAL, B. S. & ST JEOR, S., 1982. Isolation of infectious bovine rhinotracheitis virus from the soft-shelled tick, Ornithodoros coriaceus. Science, 216, 300–301.
- TERPSTRA, C., 1979. Diagnosis of infectious bovine rhinotracheitis by direct immunofluorescence. Tijdschrift voor Diergeneeskunde, 104, 138-144.
- THONUR, L., MALEY, M., GILRAY, J., CROOK, T., LAMING, E., TURNBULL, D., NATH, M. & WILLOUGHBY, K., 2012. One-step multiplex real time RT-PCR for the detection of bovine respiratory syncytial virus, bovine herpesvirus 1 and bovine parainfluenza virus 3. BMC Veterinary Research, 8, 37.
- TIKOO, S. K., CAMPOS, M. & BABIUK, L. A., 1995. Bovine herpesvirus 1 (BHV-1): biology, pathogenesis, and control. Advances in Virus Research, 45, 191-223.
- TODD, J. D., 1974. Development of intranasal vaccination for the immunization of cattle against infectious bovine rhinotracheitis. Canadian Veterinary Journal, 15, 257–259.
- TODD, J. D., VOLENEC, F. J. & PATON, I. M., 1971. Intranasal vaccination against infectious bovine rhinotracheitis: Studies on early onset of protection and use of the vaccine in pregnant cows. Journal of the American Veterinary Medical Association, 159, 1370–1374.
- TRAPP, S., OSTERRIEDER, N., KEIL, G. M. & BEER, M., 2003. Mutagenesis of a bovine herpesvirus type 1 genome cloned as an infectious bacterial artificial chromosome: analysis of glycoprotein E and G double deletion mutants. Journal of General Virology, 84(2), 301-306.
- VAN DRUNEN LITTEL-VAN DEN HURK, S., BABIUK, L. A., BRAUN, R., LEWIS, P. J., KARVONEN, B., BACA-ESTRADA, M. E., SNIDER, M., MCCARTNEY, D. & WATTS, T., 1998. Intradermal immunization with a bovine herpesvirus-1 DNA vaccine induces protective immunity in cattle. Journal of General Virology, 79, 831–839.
- VAN DRUNEN LITTEL-VAN DEN HURK, S., BRAUN, R. P., LEWIS, P. J., KARVONEN, B. C., BABIUK, L. A. & GRIEBEL, P. J., 1999. Immunization of neonates with DNA encoding a bovine herpesvirus glycoprotein is effective in the presence of maternal antibodies. Viral Immunology, 12(1), 67-77.
- VAN DRUNEN LITTEL-VAN DEN HURK, S. & HANNAMAN, D., 2010. Electroporation for DNA immunization: clinical application. Expert Review of Vaccines, 9(5), 503-517.
- VAN DRUNEN LITTEL-VAN DEN HURK, S., LAWMAN, Z., WILSON, D., LUXEMBOURG, A., ELLEFSEN, B., VAN DEN HURK, J. V. & HANNAMAN, D., 2010. Electroporation enhances immune responses and protection induced by a bovine viral diarrhea virus DNA vaccine in newborn calves with maternal antibodies. Vaccine, 28(39), 6445-6454.
- VAN DRUNEN LITTEL-VAN DEN HURK, S., TIKOO, S. K., VAN DEN HURK, J. V., BABIUK, L. A. & VAN DONKERSGOED, J., 1997. Protective immunity in cattle following vaccination with conventional and marker bovine herpesvirus-1 (BHV1) vaccines. Vaccine, 15, 36–44.
- VAN ENGELENBURG, F. A., VAN SCHIE, F. W., RIJSEWIJK, F. A. M. & VAN OIRSCHOT, J. T., 1995. Excretion of bovine herpesvirus-1 in semen is detected much longer by PCR than by virus isolation. Journal of Clinical Microbiology, 33, 308–312.
- VAN ENGELENBURG, F. A. C., MAES, R. K., VAN OIRSCHOT, J. T. & RIJSEWIJK, F. A. M., 1993. Rapid and sensitive detection of bovine herpesvirus type 1 in bovine semen by a polymerase chain reaction-based assay. Journal of Clinical Microbiology, 31, 3129–3135.
- VAN OIRSCHOT, J. T., KAASHOEK, M. J., MARIS-VELDHUIS, M. A., WEERDMEESTER, K. & RIJSEWIJK, F. A., 1997. An enzyme-linked immunosorbent assay to detect antibodies against glycoprotein gE of bovine herpesvirus 1 allows differentiation between infected and vaccinated cattle. Journal of Virological Method, 67(1), 23-34.
- VETVAC., 2017. A free online database of livestock veterinary vaccines. Bovine herpesvirus 1. www.vetvac.org/process.php?textIni=bovine+herpesvirus&submitSimple=submit&submitSimple=submit.
- WANG, J., O'KEEFE, J., ORR, D., LOTH, L., BANKS, M., WAKELEY, P., WEST, D., CARD, R., IBATA, G., VAN MAANEN, K., THOREN, P., ISAKSSON, M. & KERKHOFS, P., 2008. An international inter-laboratory ring trial to evaluate a real-time PCR assay for the detection of bovine herpesvirus 1 in extended bovine semen. Veterinary Microbiology, 126(1-3), 11-19.
- WARREN, L. M., BABIUK, L. A. & CAMPOS, M., 1996. Effects of BHV-1 on PMN adhesion to bovine lung endothelial cells. Veterinary Immunology and Immunopathology, 55(1-3), 73-82.
- WENTINK, G. H., VAN OIRSCHOT, J. T. & VERHOEFF, J., 1993. Risk of infection with bovine herpes virus 1 (BHV1): a review. Veterinary Quarterly, 15(1), 30-33.
- WERLING, D., PIERCY, J. & COFFEY, T. J., 2006. Expression of TOLL-like receptors (TLR) by bovine antigen-presenting cells-potential role in pathogen discrimination? Veterinary Immunology and Immunopathology, 112(1-2), 2-11.
- WHITE, M. B. & SNOWDON, W. A., 1973. The breeding record of cows inseminated with a batch of semen contaminated with infectious bovine rhinotrachetitis virus. Australian Veterinary Journal, 49, 501-506.
- WILSON, T. E., 1974. Observations and comments on two outbreaks of abortion associated with IBR virus infection. Canadian Veterinary Journal, 15, 227-229.
- WIRTH, U. V., GUNKEL, K., ENGELS, M. & SCHWYZER, M., 1989. Spatial and temporal distribution of bovine herpesvirus 1 transcripts. Journal of Virology, 63, 4882–4889.
- WITTE, J., 1933. Untersuchungen über den Bläschenausschlag (Exanthema pustulosum coitale) des Rindes. Zeitschrift für Infektionskrankheit Parasitäre Krankheiten und Hygiene der Haustiere, 44, 163.
- WOLFF, J. A., LUDTKE, J. J., ASCADI, G., WILLIAMS, P. & JANI, A., 1992. Long-term persistence of plasmid DNA and foreign gene expression in mouse muscle. Human Molecular Genetics, 1, 363-369.
- XIA, J. C., YASON, C. V. & KIBENGE, F. S. B., 1995. Comparison of dot blot hybridization, polymerase chain reaction, and virus isolation for detection of bovine herpesvirus-1 (BoHV-1) in artificially infected semen. Canadian Journal of Veterinary Research, 59, 102-109.
- YATES, W. D. G., 1982. A review of infectious rhinotracheitus, shipping fever pneumonia, and viral-bacterial synergism in respiratory diseases of cattle. Canadian Journal of Comparative Medicine, 46, 225–263.
- ZHANG, K., BROWNLIE, R., SNIDER, M. & VAN DRUNEN LITTEL-VAN DEN HURK, S., 2016. Phosphorylation of Bovine Herpesvirus 1 VP8 Plays a Role in Viral DNA Encapsidation and Is Essential for Its Cytoplasmic Localization and Optimal Virion Incorporation. Journal of Virology, 90(9), 4427-4440.
- ZUSCHEK, F. & CHOW, T. L., 1961. Immunogenicity of two infectious bovine rhinotracheitis vaccines. Journal of the American Veterinary Medical Association, 139, 236–237.
- ZWICK, P. & GMINDER, D., 1913. Untersuchungen über den Bläschenausschlag (Exanthema visiculosum coitale) der Rinder. Berliner Tierärztliche Wochenschrift, 29, 637–640.
- ZYGRAICH, N., LOBMANN, M., VASCOBOINIC, E., BERGE, E. & HUYGELEN, C., 1974. In vivo and in vitro properties of a temperature sensitive mutant of infectious bovine Rhinotracheitis virus. Research in Veterinary Science, 16, 328–335.